User login
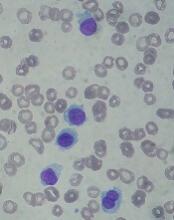
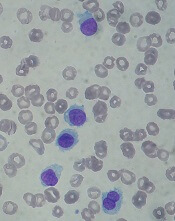
The U.S. Food and Drug Administration (FDA) has approved moxetumomab pasudotox-tdfk (Lumoxiti), a CD22-directed cytotoxin, to treat hairy cell leukemia (HCL).
Moxetumomab pasudotox is approved to treat adults with relapsed or refractory HCL who have received at least two prior systemic therapies, including treatment with a purine nucleoside analog.
The prescribing information for moxetumomab pasudotox includes a Boxed Warning noting that the drug poses risks of capillary leak syndrome (CLS) and hemolytic uremic syndrome (HUS). Treatment with moxetumomab pasudotox should be delayed or discontinued in patients who develop CLS and discontinued in patients with HUS.
The FDA granted the application for moxetumomab pasudotox fast track and priority review designations, and the drug received orphan drug designation from the FDA.
The agency granted the approval of moxetumomab pasudotox to AstraZeneca Pharmaceuticals based on results from a phase 3 trial (NCT01829711).
Data from this study were presented at the 2018 ASCO Annual Meeting (abstract 7004).
The trial included 80 patients with relapsed or refractory HCL who had received at least two prior lines of therapy.
At a median of 16.7 months of follow-up, the objective response rate was 75% (60/80), the complete response (CR) rate was 41% (33/80), and the durable CR rate was 30% (24/80). Durable CR was defined as CR with hematologic remission for more than 180 days.
Most patients with a CR achieved minimal residual disease negativity (82%; 27/33).
The median duration of response was not reached, nor was the median progression-free survival.
The most frequent treatment-related adverse events (AEs) were nausea (28%), peripheral edema (26%), headache (21%), and pyrexia (20%). Other treatment-related AEs included infections (8%) and neutropenia (3%).
Treatment-related AEs that led to discontinuation included HUS (5%), CLS (3%), and increased blood creatinine (3%).
In all, seven patients (9%) had CLS, and seven (9%) had HUS. This includes four (5%) patients who had both. CLS and HUS proved manageable and reversible.
There were three deaths in this trial, but none of them were considered treatment-related.
The U.S. Food and Drug Administration (FDA) has approved moxetumomab pasudotox-tdfk (Lumoxiti), a CD22-directed cytotoxin, to treat hairy cell leukemia (HCL).
Moxetumomab pasudotox is approved to treat adults with relapsed or refractory HCL who have received at least two prior systemic therapies, including treatment with a purine nucleoside analog.
The prescribing information for moxetumomab pasudotox includes a Boxed Warning noting that the drug poses risks of capillary leak syndrome (CLS) and hemolytic uremic syndrome (HUS). Treatment with moxetumomab pasudotox should be delayed or discontinued in patients who develop CLS and discontinued in patients with HUS.
The FDA granted the application for moxetumomab pasudotox fast track and priority review designations, and the drug received orphan drug designation from the FDA.
The agency granted the approval of moxetumomab pasudotox to AstraZeneca Pharmaceuticals based on results from a phase 3 trial (NCT01829711).
Data from this study were presented at the 2018 ASCO Annual Meeting (abstract 7004).
The trial included 80 patients with relapsed or refractory HCL who had received at least two prior lines of therapy.
At a median of 16.7 months of follow-up, the objective response rate was 75% (60/80), the complete response (CR) rate was 41% (33/80), and the durable CR rate was 30% (24/80). Durable CR was defined as CR with hematologic remission for more than 180 days.
Most patients with a CR achieved minimal residual disease negativity (82%; 27/33).
The median duration of response was not reached, nor was the median progression-free survival.
The most frequent treatment-related adverse events (AEs) were nausea (28%), peripheral edema (26%), headache (21%), and pyrexia (20%). Other treatment-related AEs included infections (8%) and neutropenia (3%).
Treatment-related AEs that led to discontinuation included HUS (5%), CLS (3%), and increased blood creatinine (3%).
In all, seven patients (9%) had CLS, and seven (9%) had HUS. This includes four (5%) patients who had both. CLS and HUS proved manageable and reversible.
There were three deaths in this trial, but none of them were considered treatment-related.
The U.S. Food and Drug Administration (FDA) has approved moxetumomab pasudotox-tdfk (Lumoxiti), a CD22-directed cytotoxin, to treat hairy cell leukemia (HCL).
Moxetumomab pasudotox is approved to treat adults with relapsed or refractory HCL who have received at least two prior systemic therapies, including treatment with a purine nucleoside analog.
The prescribing information for moxetumomab pasudotox includes a Boxed Warning noting that the drug poses risks of capillary leak syndrome (CLS) and hemolytic uremic syndrome (HUS). Treatment with moxetumomab pasudotox should be delayed or discontinued in patients who develop CLS and discontinued in patients with HUS.
The FDA granted the application for moxetumomab pasudotox fast track and priority review designations, and the drug received orphan drug designation from the FDA.
The agency granted the approval of moxetumomab pasudotox to AstraZeneca Pharmaceuticals based on results from a phase 3 trial (NCT01829711).
Data from this study were presented at the 2018 ASCO Annual Meeting (abstract 7004).
The trial included 80 patients with relapsed or refractory HCL who had received at least two prior lines of therapy.
At a median of 16.7 months of follow-up, the objective response rate was 75% (60/80), the complete response (CR) rate was 41% (33/80), and the durable CR rate was 30% (24/80). Durable CR was defined as CR with hematologic remission for more than 180 days.
Most patients with a CR achieved minimal residual disease negativity (82%; 27/33).
The median duration of response was not reached, nor was the median progression-free survival.
The most frequent treatment-related adverse events (AEs) were nausea (28%), peripheral edema (26%), headache (21%), and pyrexia (20%). Other treatment-related AEs included infections (8%) and neutropenia (3%).
Treatment-related AEs that led to discontinuation included HUS (5%), CLS (3%), and increased blood creatinine (3%).
In all, seven patients (9%) had CLS, and seven (9%) had HUS. This includes four (5%) patients who had both. CLS and HUS proved manageable and reversible.
There were three deaths in this trial, but none of them were considered treatment-related.