User login
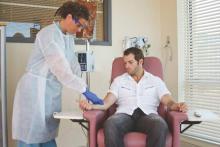
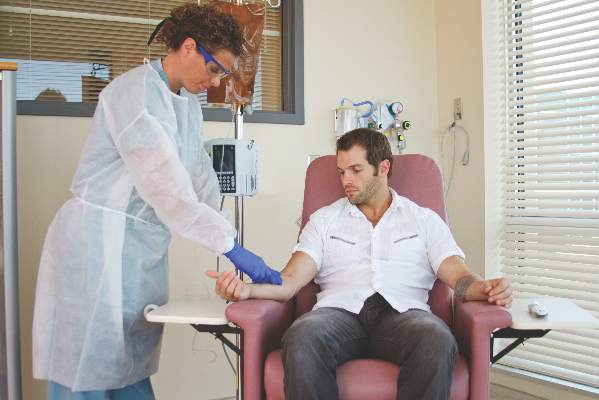
Proper administration of intravenous biologics should take place under the close supervision of a physician in a physician’s office, infusion center, or hospital rather than in a patient’s home in order to address potential infusion reactions that can range from mild to life threatening, according to a position statement issued by the American College of Rheumatology’s Committee on Rheumatologic Care.
The “Patient Safety and Site of Service for Infusible Biologics” statement, issued in late February, comes in opposition to “policies that require home infusion” that appear to seek potential cost savings with home infusions rather than meet the standard of care with on-site physician supervision.
“One observation made by some but not all payers is that infusible biologics are about twice as expensive when infused in a hospital-based infusion center as compared to other locations, such as a clinic-based infusion center or the patient’s home. Thus, some payers are rolling out policies designed to shift patients from hospital-based infusion centers to less expensive sites. The ACR is opposed to policies that would force patients, solely for the purpose of cost containment, to receive infusible biologics in an improperly supervised setting. The purpose of the position statement is to outline that stance,” Dr. Douglas W. White, chair of the ACR’s Committee on Rheumatologic Care, said in an interview.
He noted that he’s “been in on conversations with two payers who are implementing policies to move patients away from hospital-based infusions, but we are aware that others are in various stages of implementing such policies, too. It’s not so much an issue of critical mass for us, rather we’re just trying to keep ahead of the trends, and we think this will be a big trend.”
The potential for adverse reactions is not uncommon during intravenous administration of biologics, the committee wrote, noting, for example, that 10% of patients given infliximab have acute infusion reactions. On-site physicians such as rheumatologists who have experience with the “tremendous heterogeneity of patients with autoimmune disease and the diversity of conditions treated with biologics” can determine the severity of infusion reactions and decide whether or not it is safe to continue a particular biologic agent, in addition to providing reassurance to patients during acute and potentially severe reactions, according to the ACR statement.
Infusion reactions can range in severity from a mild rash to life-threatening anaphylaxis that can involve multiple organ systems leading to respiratory and cardiovascular collapse and requiring immediate treatment with medications such as epinephrine or intravenous glucocorticoids.
The position statement recognizes unusual situations in which home infusion is necessary for a patient to receive treatment because of transportation problems to a medical facility or comorbid conditions in which the risk of no treatment may outweigh the risk of home infusion. In these circumstances, the ACR “encourages providers in such unusual and difficult situations to make the best medical decision based on the individual needs of the patient. Routine home infusion of biologics is considered an unnecessary and dangerous risk to patients and violates our current clinical standards of practice.”
Requirements for using home infusion also threaten “to reduce access to” intravenous biologics, the ACR contends, because “specially trained physicians are less likely to prescribe treatments that are not properly administered in the safest clinical setting [and] patient fear of biologic therapy may lead to noncompliance and inadequate control of disease.”
The ACR noted that home administration of subcutaneous biologics is medically appropriate and the injection site reactions that can occur with their use are often easily managed.
Proper administration of intravenous biologics should take place under the close supervision of a physician in a physician’s office, infusion center, or hospital rather than in a patient’s home in order to address potential infusion reactions that can range from mild to life threatening, according to a position statement issued by the American College of Rheumatology’s Committee on Rheumatologic Care.
The “Patient Safety and Site of Service for Infusible Biologics” statement, issued in late February, comes in opposition to “policies that require home infusion” that appear to seek potential cost savings with home infusions rather than meet the standard of care with on-site physician supervision.
“One observation made by some but not all payers is that infusible biologics are about twice as expensive when infused in a hospital-based infusion center as compared to other locations, such as a clinic-based infusion center or the patient’s home. Thus, some payers are rolling out policies designed to shift patients from hospital-based infusion centers to less expensive sites. The ACR is opposed to policies that would force patients, solely for the purpose of cost containment, to receive infusible biologics in an improperly supervised setting. The purpose of the position statement is to outline that stance,” Dr. Douglas W. White, chair of the ACR’s Committee on Rheumatologic Care, said in an interview.
He noted that he’s “been in on conversations with two payers who are implementing policies to move patients away from hospital-based infusions, but we are aware that others are in various stages of implementing such policies, too. It’s not so much an issue of critical mass for us, rather we’re just trying to keep ahead of the trends, and we think this will be a big trend.”
The potential for adverse reactions is not uncommon during intravenous administration of biologics, the committee wrote, noting, for example, that 10% of patients given infliximab have acute infusion reactions. On-site physicians such as rheumatologists who have experience with the “tremendous heterogeneity of patients with autoimmune disease and the diversity of conditions treated with biologics” can determine the severity of infusion reactions and decide whether or not it is safe to continue a particular biologic agent, in addition to providing reassurance to patients during acute and potentially severe reactions, according to the ACR statement.
Infusion reactions can range in severity from a mild rash to life-threatening anaphylaxis that can involve multiple organ systems leading to respiratory and cardiovascular collapse and requiring immediate treatment with medications such as epinephrine or intravenous glucocorticoids.
The position statement recognizes unusual situations in which home infusion is necessary for a patient to receive treatment because of transportation problems to a medical facility or comorbid conditions in which the risk of no treatment may outweigh the risk of home infusion. In these circumstances, the ACR “encourages providers in such unusual and difficult situations to make the best medical decision based on the individual needs of the patient. Routine home infusion of biologics is considered an unnecessary and dangerous risk to patients and violates our current clinical standards of practice.”
Requirements for using home infusion also threaten “to reduce access to” intravenous biologics, the ACR contends, because “specially trained physicians are less likely to prescribe treatments that are not properly administered in the safest clinical setting [and] patient fear of biologic therapy may lead to noncompliance and inadequate control of disease.”
The ACR noted that home administration of subcutaneous biologics is medically appropriate and the injection site reactions that can occur with their use are often easily managed.
Proper administration of intravenous biologics should take place under the close supervision of a physician in a physician’s office, infusion center, or hospital rather than in a patient’s home in order to address potential infusion reactions that can range from mild to life threatening, according to a position statement issued by the American College of Rheumatology’s Committee on Rheumatologic Care.
The “Patient Safety and Site of Service for Infusible Biologics” statement, issued in late February, comes in opposition to “policies that require home infusion” that appear to seek potential cost savings with home infusions rather than meet the standard of care with on-site physician supervision.
“One observation made by some but not all payers is that infusible biologics are about twice as expensive when infused in a hospital-based infusion center as compared to other locations, such as a clinic-based infusion center or the patient’s home. Thus, some payers are rolling out policies designed to shift patients from hospital-based infusion centers to less expensive sites. The ACR is opposed to policies that would force patients, solely for the purpose of cost containment, to receive infusible biologics in an improperly supervised setting. The purpose of the position statement is to outline that stance,” Dr. Douglas W. White, chair of the ACR’s Committee on Rheumatologic Care, said in an interview.
He noted that he’s “been in on conversations with two payers who are implementing policies to move patients away from hospital-based infusions, but we are aware that others are in various stages of implementing such policies, too. It’s not so much an issue of critical mass for us, rather we’re just trying to keep ahead of the trends, and we think this will be a big trend.”
The potential for adverse reactions is not uncommon during intravenous administration of biologics, the committee wrote, noting, for example, that 10% of patients given infliximab have acute infusion reactions. On-site physicians such as rheumatologists who have experience with the “tremendous heterogeneity of patients with autoimmune disease and the diversity of conditions treated with biologics” can determine the severity of infusion reactions and decide whether or not it is safe to continue a particular biologic agent, in addition to providing reassurance to patients during acute and potentially severe reactions, according to the ACR statement.
Infusion reactions can range in severity from a mild rash to life-threatening anaphylaxis that can involve multiple organ systems leading to respiratory and cardiovascular collapse and requiring immediate treatment with medications such as epinephrine or intravenous glucocorticoids.
The position statement recognizes unusual situations in which home infusion is necessary for a patient to receive treatment because of transportation problems to a medical facility or comorbid conditions in which the risk of no treatment may outweigh the risk of home infusion. In these circumstances, the ACR “encourages providers in such unusual and difficult situations to make the best medical decision based on the individual needs of the patient. Routine home infusion of biologics is considered an unnecessary and dangerous risk to patients and violates our current clinical standards of practice.”
Requirements for using home infusion also threaten “to reduce access to” intravenous biologics, the ACR contends, because “specially trained physicians are less likely to prescribe treatments that are not properly administered in the safest clinical setting [and] patient fear of biologic therapy may lead to noncompliance and inadequate control of disease.”
The ACR noted that home administration of subcutaneous biologics is medically appropriate and the injection site reactions that can occur with their use are often easily managed.