User login
Preclinical research suggests that targeting JAK2 can reduce the risk of graft-versus-host disease (GVHD) in transplant recipients.
Researchers found that genetic ablation of JAK2 on donor T cells or inhibition of JAK2 via treatment with pacritinib reduced GVHD in mice without compromising graft-versus-leukemia (GVL) activity.
“An effort to identify selective immune suppression whereby GVHD is reduced and the antitumor activity of the graft is preserved is key to improving the success of blood and marrow transplantation,” said Brian C. Betts, MD, of Moffitt Cancer Center in Tampa, Florida.
Dr Betts and his colleagues conducted this research and recounted their findings in PNAS.
In experiments with mice, the researchers found that donor T cells with JAK2 deletion were less likely than wild-type donor T cells to induce GVHD. However, JAK2 deletion did not impair the GVL effect.
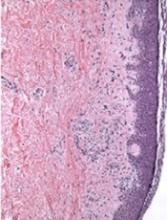
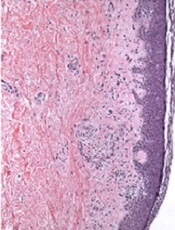
Mice that received JAK2−/− T cells had longer survival, higher body weights, and less GVHD than control mice.
The researchers said targeting JAK2 may reduce GVHD, in part, by limiting Th1 differentiation and the migratory capacity of alloreactive T cells. However, targeting JAK2 promotes beneficial regulatory-T-cell and Th2 differentiation as well.
The researchers also tested the effects of JAK2 inhibition with pacritinib. Mice received allografts and were treated with pacritinib or vehicle control for 3 weeks.
As before, JAK2 inhibition reduced acute GVHD mortality while preserving the GVL effect.
The team also discovered that pacritinib could protect mice from tissue graft rejection, suggesting the drug could be used to prevent kidney or liver transplant rejection.
Now, the researchers are working on a phase 1/2 trial designed to determine if pacritinib and standard immune suppression can prevent acute GVHD after hematopoietic stem cell transplant (NCT02891603). ![]()
Preclinical research suggests that targeting JAK2 can reduce the risk of graft-versus-host disease (GVHD) in transplant recipients.
Researchers found that genetic ablation of JAK2 on donor T cells or inhibition of JAK2 via treatment with pacritinib reduced GVHD in mice without compromising graft-versus-leukemia (GVL) activity.
“An effort to identify selective immune suppression whereby GVHD is reduced and the antitumor activity of the graft is preserved is key to improving the success of blood and marrow transplantation,” said Brian C. Betts, MD, of Moffitt Cancer Center in Tampa, Florida.
Dr Betts and his colleagues conducted this research and recounted their findings in PNAS.
In experiments with mice, the researchers found that donor T cells with JAK2 deletion were less likely than wild-type donor T cells to induce GVHD. However, JAK2 deletion did not impair the GVL effect.
Mice that received JAK2−/− T cells had longer survival, higher body weights, and less GVHD than control mice.
The researchers said targeting JAK2 may reduce GVHD, in part, by limiting Th1 differentiation and the migratory capacity of alloreactive T cells. However, targeting JAK2 promotes beneficial regulatory-T-cell and Th2 differentiation as well.
The researchers also tested the effects of JAK2 inhibition with pacritinib. Mice received allografts and were treated with pacritinib or vehicle control for 3 weeks.
As before, JAK2 inhibition reduced acute GVHD mortality while preserving the GVL effect.
The team also discovered that pacritinib could protect mice from tissue graft rejection, suggesting the drug could be used to prevent kidney or liver transplant rejection.
Now, the researchers are working on a phase 1/2 trial designed to determine if pacritinib and standard immune suppression can prevent acute GVHD after hematopoietic stem cell transplant (NCT02891603). ![]()
Preclinical research suggests that targeting JAK2 can reduce the risk of graft-versus-host disease (GVHD) in transplant recipients.
Researchers found that genetic ablation of JAK2 on donor T cells or inhibition of JAK2 via treatment with pacritinib reduced GVHD in mice without compromising graft-versus-leukemia (GVL) activity.
“An effort to identify selective immune suppression whereby GVHD is reduced and the antitumor activity of the graft is preserved is key to improving the success of blood and marrow transplantation,” said Brian C. Betts, MD, of Moffitt Cancer Center in Tampa, Florida.
Dr Betts and his colleagues conducted this research and recounted their findings in PNAS.
In experiments with mice, the researchers found that donor T cells with JAK2 deletion were less likely than wild-type donor T cells to induce GVHD. However, JAK2 deletion did not impair the GVL effect.
Mice that received JAK2−/− T cells had longer survival, higher body weights, and less GVHD than control mice.
The researchers said targeting JAK2 may reduce GVHD, in part, by limiting Th1 differentiation and the migratory capacity of alloreactive T cells. However, targeting JAK2 promotes beneficial regulatory-T-cell and Th2 differentiation as well.
The researchers also tested the effects of JAK2 inhibition with pacritinib. Mice received allografts and were treated with pacritinib or vehicle control for 3 weeks.
As before, JAK2 inhibition reduced acute GVHD mortality while preserving the GVL effect.
The team also discovered that pacritinib could protect mice from tissue graft rejection, suggesting the drug could be used to prevent kidney or liver transplant rejection.
Now, the researchers are working on a phase 1/2 trial designed to determine if pacritinib and standard immune suppression can prevent acute GVHD after hematopoietic stem cell transplant (NCT02891603). ![]()