User login
Article Type
Changed
Display Headline
Targeted Therapies and Surgical Resection for Lung Cancer: Evolving Treatment Options
References
- American Cancer Society. Key statistics for lung cancer. Revised January 29, 2024. Accessed June 10, 2024. https://www.cancer.org/cancer/types/lung-cancer/about/key-statistics.html
- Drilon A, Camidge DR, Lin JJ, et al; for the TRIDENT-1 Investigators. Repotrectinib in ROS1 fusion-positive non-small-cell lung cancer. N Engl J Med. 2024;390(2):118-131. doi:10.1056/NEJMoa2302299
- Wu YL, Dziadziuszko R, Ahn JS, et al; for the ALINA Investigators. Alectinib in resected ALK-positive non-small-cell lung cancer. N Engl J Med. 2024;390(14):1265-1276.
- Mulligan L. Selective RET kinase inhibitors and lung cancer. N Engl J Med. 2023;389(20):1913-1916. doi:10.1056/NEJMe2311295
- Zhou C, Soloman B, Loong HH, et al; for the LIBRETTO-432 Trial Investigators. First-line selpercatinib or chemotherapy and pembrolizumab in RET fusion-positive NSCLC. N Engl J Med. 2023:389(20):1839-1850. doi:10.1056/NEJMoa239457
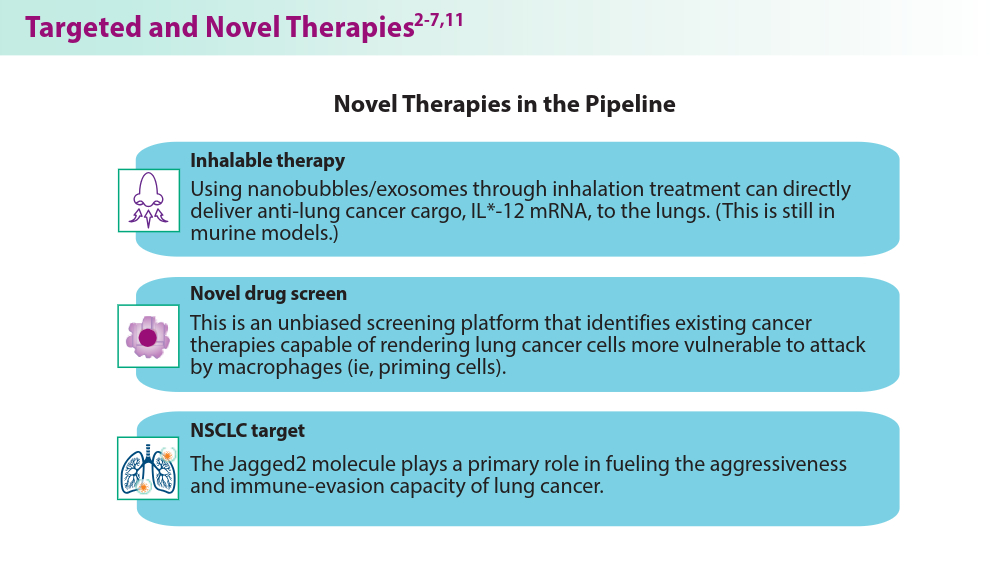
- Vaccaro K, Allen J, Whitfield TW, et al. Targeted therapies prime oncogene-driven lung cancers for macrophage-mediated destruction. bioRxiv. Preprint posted online March 6, 2023. doi:10.1101/2023.03.03.531059
- Liu M, Hu S, Yan N, Popowski KD, Cheng K. Inhalable extracellular vesicle delivery of IL-12 mRNA to treat lung cancer and promote systemic immunity. Nat Nanotechnol. 2024;19(4):565-575. doi:10.1038/s41565-023-01580-3
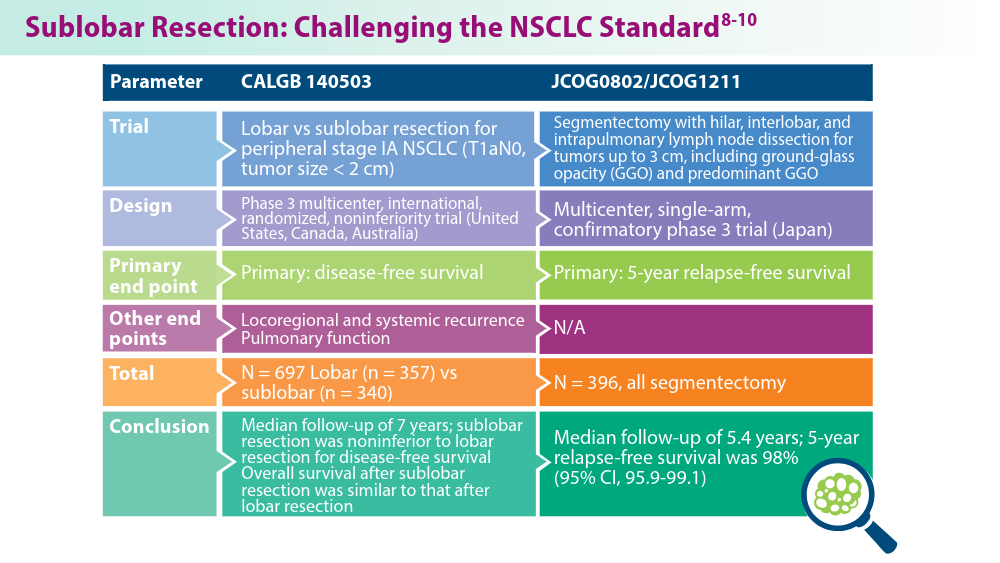
- Altorki N, Wang X, Kozono D, et al. Lobar or sublobar resection for peripheral stage IA non-small-cell lung cancer. N Engl J Med. 2023;388(6):489-498. doi:10.1056/NEJMoa2212083
- Koike T, Hasebe T, Nakamura M, Shimizu Y, Goto T, Tsuchida M. Towards better outcomes: segmentectomy for ground-glass opacity-dominant non-small cell lung cancer 3 cm or less─insights form JCOG1211 [editorial commentary]. AME Clin Trials Rev. 2023;1:5. doi:10.21037/actr-23-10
- Aokage K, Suzuki K, Saji H, et al; for the Japan Clinical Oncology Group. Segmentectomy for ground-glass-dominant lung cancer with a tumour diameter of 3 cm or less including groundglass opacity (JCOG1211): a multicentre, single-arm, confirmatory phase 3 trial. Lancet Respir Med. 2023;11(6):540-549. doi:10.1016/S2213-2600(23)00041-3
- Mandula JK, Sierra-Mondragon RA, Jimenez RV, et al. Jagged2 targeting in lung cancer activates anti-tumor immunity via Notch-induced functional reprogramming of tumor-associated macrophages. Immunity. 2024;57(5):1124-1140.e9. doi:10.1016/j.immuni.2024.03.020
Publications
Topics
References
- American Cancer Society. Key statistics for lung cancer. Revised January 29, 2024. Accessed June 10, 2024. https://www.cancer.org/cancer/types/lung-cancer/about/key-statistics.html
- Drilon A, Camidge DR, Lin JJ, et al; for the TRIDENT-1 Investigators. Repotrectinib in ROS1 fusion-positive non-small-cell lung cancer. N Engl J Med. 2024;390(2):118-131. doi:10.1056/NEJMoa2302299
- Wu YL, Dziadziuszko R, Ahn JS, et al; for the ALINA Investigators. Alectinib in resected ALK-positive non-small-cell lung cancer. N Engl J Med. 2024;390(14):1265-1276.
- Mulligan L. Selective RET kinase inhibitors and lung cancer. N Engl J Med. 2023;389(20):1913-1916. doi:10.1056/NEJMe2311295
- Zhou C, Soloman B, Loong HH, et al; for the LIBRETTO-432 Trial Investigators. First-line selpercatinib or chemotherapy and pembrolizumab in RET fusion-positive NSCLC. N Engl J Med. 2023:389(20):1839-1850. doi:10.1056/NEJMoa239457
- Vaccaro K, Allen J, Whitfield TW, et al. Targeted therapies prime oncogene-driven lung cancers for macrophage-mediated destruction. bioRxiv. Preprint posted online March 6, 2023. doi:10.1101/2023.03.03.531059
- Liu M, Hu S, Yan N, Popowski KD, Cheng K. Inhalable extracellular vesicle delivery of IL-12 mRNA to treat lung cancer and promote systemic immunity. Nat Nanotechnol. 2024;19(4):565-575. doi:10.1038/s41565-023-01580-3
- Altorki N, Wang X, Kozono D, et al. Lobar or sublobar resection for peripheral stage IA non-small-cell lung cancer. N Engl J Med. 2023;388(6):489-498. doi:10.1056/NEJMoa2212083
- Koike T, Hasebe T, Nakamura M, Shimizu Y, Goto T, Tsuchida M. Towards better outcomes: segmentectomy for ground-glass opacity-dominant non-small cell lung cancer 3 cm or less─insights form JCOG1211 [editorial commentary]. AME Clin Trials Rev. 2023;1:5. doi:10.21037/actr-23-10
- Aokage K, Suzuki K, Saji H, et al; for the Japan Clinical Oncology Group. Segmentectomy for ground-glass-dominant lung cancer with a tumour diameter of 3 cm or less including groundglass opacity (JCOG1211): a multicentre, single-arm, confirmatory phase 3 trial. Lancet Respir Med. 2023;11(6):540-549. doi:10.1016/S2213-2600(23)00041-3
- Mandula JK, Sierra-Mondragon RA, Jimenez RV, et al. Jagged2 targeting in lung cancer activates anti-tumor immunity via Notch-induced functional reprogramming of tumor-associated macrophages. Immunity. 2024;57(5):1124-1140.e9. doi:10.1016/j.immuni.2024.03.020
References
- American Cancer Society. Key statistics for lung cancer. Revised January 29, 2024. Accessed June 10, 2024. https://www.cancer.org/cancer/types/lung-cancer/about/key-statistics.html
- Drilon A, Camidge DR, Lin JJ, et al; for the TRIDENT-1 Investigators. Repotrectinib in ROS1 fusion-positive non-small-cell lung cancer. N Engl J Med. 2024;390(2):118-131. doi:10.1056/NEJMoa2302299
- Wu YL, Dziadziuszko R, Ahn JS, et al; for the ALINA Investigators. Alectinib in resected ALK-positive non-small-cell lung cancer. N Engl J Med. 2024;390(14):1265-1276.
- Mulligan L. Selective RET kinase inhibitors and lung cancer. N Engl J Med. 2023;389(20):1913-1916. doi:10.1056/NEJMe2311295
- Zhou C, Soloman B, Loong HH, et al; for the LIBRETTO-432 Trial Investigators. First-line selpercatinib or chemotherapy and pembrolizumab in RET fusion-positive NSCLC. N Engl J Med. 2023:389(20):1839-1850. doi:10.1056/NEJMoa239457
- Vaccaro K, Allen J, Whitfield TW, et al. Targeted therapies prime oncogene-driven lung cancers for macrophage-mediated destruction. bioRxiv. Preprint posted online March 6, 2023. doi:10.1101/2023.03.03.531059
- Liu M, Hu S, Yan N, Popowski KD, Cheng K. Inhalable extracellular vesicle delivery of IL-12 mRNA to treat lung cancer and promote systemic immunity. Nat Nanotechnol. 2024;19(4):565-575. doi:10.1038/s41565-023-01580-3
- Altorki N, Wang X, Kozono D, et al. Lobar or sublobar resection for peripheral stage IA non-small-cell lung cancer. N Engl J Med. 2023;388(6):489-498. doi:10.1056/NEJMoa2212083
- Koike T, Hasebe T, Nakamura M, Shimizu Y, Goto T, Tsuchida M. Towards better outcomes: segmentectomy for ground-glass opacity-dominant non-small cell lung cancer 3 cm or less─insights form JCOG1211 [editorial commentary]. AME Clin Trials Rev. 2023;1:5. doi:10.21037/actr-23-10
- Aokage K, Suzuki K, Saji H, et al; for the Japan Clinical Oncology Group. Segmentectomy for ground-glass-dominant lung cancer with a tumour diameter of 3 cm or less including groundglass opacity (JCOG1211): a multicentre, single-arm, confirmatory phase 3 trial. Lancet Respir Med. 2023;11(6):540-549. doi:10.1016/S2213-2600(23)00041-3
- Mandula JK, Sierra-Mondragon RA, Jimenez RV, et al. Jagged2 targeting in lung cancer activates anti-tumor immunity via Notch-induced functional reprogramming of tumor-associated macrophages. Immunity. 2024;57(5):1124-1140.e9. doi:10.1016/j.immuni.2024.03.020
Publications
Publications
Topics
Article Type
Display Headline
Targeted Therapies and Surgical Resection for Lung Cancer: Evolving Treatment Options
Display Headline
Targeted Therapies and Surgical Resection for Lung Cancer: Evolving Treatment Options
Disallow All Ads
Content Gating
Open Access (article Unlocked/Open Access)
Alternative CME
Disqus Comments
Default
Consolidated Pubs: Do Not Show Source Publication Logo
Use ProPublica
Conference Recap Checkbox
Not Conference Recap
Clinical Edge
Medscape Article
Display survey writer
Reuters content
Disable Inline Native ads
WebMD Article