User login
The year 2014 marked a sea change in our approach to tissue extraction during minimally invasive surgery. The US Food and Drug Administration (FDA) initiated this transformation in April, when it issued a safety warning on the use of open power morcellation.1 A flurry of statements on the practice followed from professional societies, capped, in late November, with another statement from the FDA.2–4 The new bottom line: The use of open electromechanical (“power”) morcellation is contraindicated in perimenopausal and postmenopausal women, as well as in those who are known or suspected to have a malignancy.4
Most of the concern to date has centered on the risk that an occult leiomyosarcoma could be morcellated inadvertently during uterine surgery, an event that may worsen the prognosis for the patient. To get a gynecologic oncologist’s take on the controversy, OBG Management caught up with Amanda Nickles Fader, MD, director of the Kelly Gynecologic Oncology Service at Johns Hopkins University. Dr. Fader’s perspective is unique in that she treats a relatively high number of patients who have leiomyosarcoma and other uterine cancers.
In this Q&A, we discuss the patient population at Johns Hopkins; why Dr. Fader is especially qualified to speak to the future of electromechanical morcellation in gynecologic surgery; the benefits and risks of minimally invasive surgery, including tissue extraction; her recommendations for preoperative evaluation and counseling of patients undergoing uterine surgery; and guidance on how the specialty of gynecologic surgery should proceed in the future.
OBG Management: Dr. Fader, by way of introduction, could you characterize your patient population?
Amanda Nickles Fader, MD: Like most gynecologic oncologists, I primarily treat women with cancers of the uterus, ovary, cervix, and vulva. Many of us also have the opportunity to treat a number of women each year with complex benign gynecologic conditions that require surgery. As someone who is extremely interested in rare gynecologic tumors, I also treat a relatively high volume of women diagnosed with uterine sarcoma.
Approximately 75% of the women I see in my practice have preinvasive or invasive cancer, and 25% have a benign condition, such as enlarged fibroids or advanced-stage endometriosis.
OBG Management: How many cases of uterine sarcoma do you encounter on an annual basis?
Dr. Fader: Uterine sarcomas are very rare. They represent only 2% of all uterine cancers. Put into perspective, that means that about 0.4 cases of leiomyosarcoma occur in every 100,000 US women—most commonly postmenopausal women. Leiomyosarcoma is a biologically aggressive, high-grade malignancy that often is lethal.5
Endometrial stroma sarcoma is even less common—only 0.3 cases occur in every 100,000 US women. However, this tumor type is more indolent, often diagnosed at an earlier stage, and potentially curable with surgery (with or without hormonal therapy).6
As a referral center for rare uterine tumors, the Kelly Gynecologic Oncology Service and the Sarcoma Center at Johns Hopkins see approximately 35 to 40 new cases of uterine sarcoma annually for treatment of primary disease or recurrence. An additional 15 to 20 consult cases are reviewed from outside hospitals each year by our gynecologic pathology department.
Why a minimally invasive approach is vital
OBG Management: When it comes to uterine surgery for presumed benign conditions, why is a minimally invasive approach important?
Dr. Fader: Minimally invasive surgery clearly benefits women and is one of the greatest advances of the past half-century within our field. Randomized controlled trials and meta-analyses have demonstrated without question that women who undergo minimally invasive surgery for benign conditions or early-stage cancerous gynecologic conditions have superior clinical outcomes, compared with women who undergo surgery via laparotomy.7,8 These outcomes include fewer perioperative complications (including fewer cases of surgical site infection, venous thromboembolism, wound dehiscence, and hospital readmission), shorter hospital stays, less pain, faster recovery, and fewer adhesions. And when women with early-stage cancers undergo minimally invasive surgery, randomized controlled trials show, they have a stage-specific survival rate similar to that observed in women treated with laparotomy.9
Benefits and risks of tissue extraction in minimally invasive surgery
OBG Management: What are the main benefits of tissue extraction, including morcellation?
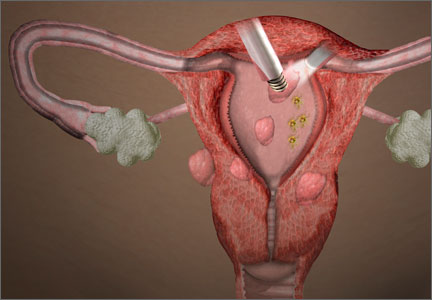
Dr. Fader: Tissue extraction is a practice that has allowed us to offer minimally invasive surgery to countless more women than we could have in the recent past. It is a technique in which a large specimen (typically a uterus or fibroid) is fragmented into smaller parts in order to remove it through a small laparoscopic incision or orifice (vagina, umbilicus). Without tissue-extraction practices, thousands of women who undergo myomectomy each year to conserve their fertility and hundreds of thousands of women who require hysterectomy potentially would have to undergo a more painful and risky surgery through a larger abdominal incision. That would not be desirable, as we know conclusively that laparotomy is associated with worse outcomes—and even an increased risk of mortality—compared with minimally invasive surgery performed by experienced surgeons.10
Tissue extraction can be approached in a variety of ways. It can be performed with a scalpel, with a resectoscope, or with an electromechanical morcellator. Tissue extraction can be performed within the uterine cavity, through the vagina, within the abdominal compartment, or within a containment system in any of those compartments.
OBG Management: What are the risks of tissue extraction?
Dr. Fader: The risks of tissue extraction with electromechanical morcellation include potential injury to intra-abdominal organs and vasculature and risk of dissemination of an occult (ie, undiagnosed) uterine cancer. A report by our research group also demonstrated the risks of dissemination of benign uterine tissue requiring subsequent surgery in the setting of open electromechanical morcellation.11 The risk of these events occurring in the hands of a thoughtful and experienced surgeon who conducts comprehensive preoperative patient evaluations is extremely low. However, recent evidence demonstrates that a handful of women worldwide are diagnosed with an occult uterine cancer each year during a morcellation procedure.12–14 Although it is a very rare event (given that most women undergoing hysterectomy and myomectomy procedures are of reproductive age and unlikely to have uterine cancer), this risk is a serious issue. There is an exigent need for the gynecologic surgical community to develop better approaches to tissue extraction that minimize preventable harm in women.
Needed: high-quality data on the risks of morcellation
OBG Management: How does the recent FDA statement urging caution with the use of open power morcellation factor into this equation? The most recent FDA statement noted that open power morcellation is contraindicated in perimenopausal and postmenopausal women.4
Dr. Fader: The FDA’s concern is legitimate. However, the magnitude of the risk of occult uterine sarcoma in women undergoing presumed benign gynecologic surgery has been scrutinized and debated. The FDA panel quoted a risk that roughly 1 in 350 women undergoing presumed benign gynecologic surgery for fibroids will have an occult leiomyosarcoma diagnosed. However, more recent systematic reviews and a review of the prospective published literature demonstrate that the risk is more likely on the order of 1 in 1,700 to 1 in 8,333 women.15 The risk may be even lower in gynecologic surgery practices that see a high volume of hysterectomy/myomectomy cases and utilize meticulous preoperative patient selection criteria to establish a woman’s candidacy for tissue-extraction procedures.
I am concerned with how “occult sarcoma” discovered during surgery for “presumed benign gynecologic disease is being defined in the literature. There is no uniform definition being used. A cancer in this setting is only truly occult or undiagnosed if the physician was thinking about it and made every effort to rule out cancer preoperatively, and the morcellation procedure was performed in a low-risk population (but cancer was still diagnosed on final pathology in this population). However, in the majority of the morcellation studies in the literature, it is not clear that thorough preoperative evaluations occurred in patients to rule out uterine malignancy—in fact, there is a paucity of published information regarding establishing appropriate patient candidacy for morcellation procedures. So we can’t derive any conclusions regarding whether “occult” sarcomas were truly undetectable or not in the published literature.
In addition, the literature is very clear that advancing age and postmenopausal status are risk factors for uterine malignancy.16 The vast majority of uterine sarcoma cases are diagnosed in postmenopausal women. Yet, in one large US population-based hysterectomy study, 20% of the morcellated cases (and the overwhelming majority of the “occult” morcellated uterine cancers) occurred in postmenopausal women!17
Further, in a more recent study by the same authors, again the risks of morcellating a uterine cancer in a population undergoing myomectomy was significantly higher in a postmenopausal population and occurred only rarely in women younger than age 40. But it should come as no surprise that a greater incidence of uterine cancer was identified in these older cohorts—cancer risk increases precipitously with age. That’s not “occult”; that’s basic cancer epidemiology.
In other words, we cannot assume from population-based administrative claims data that morcellation performed in inappropriate populations at higher risk of uterine malignancy (in which we do not know whether patients were properly screened for the procedure preoperatively or whether they had risk factors for uterine cancer but were presumably poor candidates for morcellation due to age alone) helps us define the true incidence of “occult” sarcoma or cancer in a population.
These studies are provocative, however, and do inform us that, as women get older, we are apt to see a greater incidence of uterine cancer. We cannot safely assume that a postmenopausal or elderly woman with symptomatic or enlarging fibroids has “presumed benign disease”—it is cancer until proven otherwise, and we need to be looking for it preoperatively. Therefore, we need to be particularly careful with our surgical practices in this population—ie, the basis for the FDA’s recommendation to avoid morcellation in older women. And I agree with the FDA that open electromechanical morcellation generally is contraindicated in postmenopausal women. However, we need better data from large prospective studies to inform our understanding of the true incidence of undiagnosed or “occult” uterine sarcoma in women undergoing surgery for presumed benign disease. These future studies are likely to demonstrate what we already know—that in young, well-screened, well-selected candidates for minimally invasive hysterectomy or myomectomy, the risk of occult cancer is going to be exceptionally low.
OBG Management: Which is greater—the risks or benefits of tissue extraction?
Dr. Fader: Assessment of risks and benefits in medicine has everything to do with the intervention in question as applied to an individual patient. At the end of the day, there are risks and benefits to every medical or surgical treatment offered to patients in every medical and surgical discipline. But the risk of an occult uterine sarcoma is extremely low in a woman of reproductive age who has been properly selected and comprehensively evaluated for minimally invasive surgery and tissue extraction prior to surgery. And this small—though not negligible—risk must be weighed against the much higher risk of harm that may be incurred with an open abdominal procedure, compared with minimally invasive surgery.
However, in many elderly women, the risks of tissue extraction with an electromechanical morcellator may outweigh the benefits. Even so, there are exceptions in which tissue extraction may be acceptable in postmenopausal women (ie, using alternative tissue-extraction methods in those undergoing minimally invasive supracervical hysterectomy and sacral colpopexy for pelvic organ prolapse).
Few of the data published on the risks of morcellation are of very high quality in terms of scientific rigor or methodology. The best thing we can do as a gynecologic surgical community is conduct sound quality-improvement (QI) programs, disseminate our QI results, publish our data, establish guidelines for best practices in uterine tissue extraction, and collaborate readily to increase the scholarly output on this issue so that national societies and government regulatory agencies have better-quality data to inform future policy on this issue.
A case-based approach
OBG Management: How would you approach tissue extraction in the following case?
CASE: A desire for myomectomy
A 35-year-old woman (G1P1) who delivered by cesarean has an 8-cm symptomatic intramural fibroid. She has regular heavy periods that have led to anemia (hemoglobin level = 10 mg/dL). Her medical history is negative for malignancy, pelvic radiation, or tamoxifen use, and she wants to preserve her fertility. Magnetic resonance imaging (MRI) confirms an 8-cm fibroid. Endometrial biopsy results are negative.
Dr. Fader: At Johns Hopkins, as at many other centers, we use well-defined criteria to determine whether a minimally invasive approach (and tissue extraction) might be appropriate. We also individualize treatment and surgical decision-making on a case-by-case basis. Any candidate for minimally invasive surgery and tissue extraction for uterine fibroids must undergo a thorough preoperative assessment.
Johns Hopkins preoperative assessment criteria include:
- endometrial biopsy
- imaging (often MRI)
- a detailed history and physical, with a comprehensive review of risk factors for malignancy, including family and genetic history or a personal history of malignancy, pelvic radiation, tamoxifen use, or BRCA or hereditary nonpolyposis colorectal cancer (HNPCC) deleterious mutation carrier status, among other things.
- In addition, all cases are discussed at a peer-reviewed, preoperative conference to ensure that a thorough work-up has been conducted and to verify the patient’s candidacy for a minimally invasive procedure with tissue extraction. As the FDA recommends, we conduct an enhanced informed consent process and counsel patients being considered for tissue extraction about the risk of occult sarcoma.
Our top priority is patient safety, so until more data are available, we no longer perform open electromechanical morcellation. We perform all tissue extraction within a containment system and under institutional review board protocol. We primarily perform tissue extraction via scalpel morcellation.
OBG Management: How do the patient’s wishes factor into the decision to perform minimally invasive surgery with morcellation?
Dr. Fader: Our patients make their own decisions regarding surgical approach and procedure after undergoing extensive counseling about the risks and benefits of the proposed procedures. I certainly would offer a patient like the one described in this case the opportunity for a minimally invasive approach (if, after thorough preoperative evaluation, she were deemed to be at very low risk for uterine malignancy). In my experience, most women opt for the minimally invasive approach in this setting; however, if a patient declines minimally invasive surgery, I respect her decision.
Tissue extraction in perimenopause
CASE: A desire for myomectomy at age 48
OBG Management: How would you approach the same case if the patient were a 48-year-old perimenopausal woman?
Dr. Fader: In perimenopausal women, we are more selective about performing morcellation, given the recent FDA safety statement, and because the incidence of occult cancer starts to increase slightly in this patient cohort (although it doesn’t precipitously increase until well into the postmenopausal period).
In addition, myomectomy may have less value in a 48-year-old, given the lower likelihood of achieving successful fertility, although there are exceptions. US cancer statistics and studies on morcellation demonstrate that the vast majority of women in their 30s and 40s have an extremely low risk for sarcomas and other uterine malignancies.2
In select cases in which a woman has undergone a comprehensive preoperative work-up, has a stable-appearing fibroid(s), and is well-educated and counseled about the pros and cons of morcellation, we would consider performing a procedure with contained tissue extraction. As a general matter, however, I would be more inclined to offer a 48-year-old in this situation a uterine artery embolization or minimally invasive hysterectomy than a myomectomy procedure, especially given the recent study by Wright and colleagues demonstrating the significantly increased risks of uterine cancer at myomectomy surgery for a woman in her late 40s or early 50s.18
Preoperative assessment should be comprehensive
OBG Management: What preoperative evaluation do you perform when tissue extraction, including morcellation, is an issue?
Dr. Fader: It is the policy at Johns Hopkins that all women being considered for minimally invasive surgery and tissue extraction must undergo a rigorous preoperative work-up that includes:
- a comprehensive history and physical (to exclude malignancy and risk for occult malignancy)
- an endometrial evaluation (most commonly, an endometrial biopsy)
- uterine imaging (with longitudinal evaluation of imaging findings if performed previously)
- discussion of each patient case at peer-reviewed, preoperative department conferences.
We have separate conferences for the gynecology and gynecologic oncology services and have employed this practice for many years at Hopkins. We are also studying the role of serum lactate dehydrogenase (LDH) isoenzyme levels in stratifying women with uterine fibroids versus cancer/sarcoma.19
OBG Management: Which patients would you exclude from the electromechanical morcellation option?
Dr. Fader: As the American College of Obstetricians and Gynecologists, the AAGL, the Society of Gynecologic Oncology, and the Society of Gynecologic Surgeons appear to agree:
- When utilized in select patients of reproductive age, minimally invasive surgery and morcellation are beneficial.
- Morcellation should categorically not be performed in any woman who has a known or suspected uterine (or other gynecologic) malignancy.2,3
At our institution, we have significantly curtailed the use of electromechanical morcellation at this time and especially do not perform it in women aged 50 or older or in those with confirmed postmenopausal status. We also do not perform electromechanical morcellation in women with a personal history of uterine, cervical, or ovarian preinvasive or invasive cancer, or in women with a strong family history of gynecologic malignancy.
Other populations we exclude are women with:
- a history of mitotically active or atypical fibroids (as determined at previous myomectomy)
- known BRCA or HNPCC deleterious mutation, hereditary leiomyomatosis and renal cell carcinoma (HLRCC) syndrome, hereditary childhood retinoblastoma, or other genetic predisposition to uterine or ovarian cancer
- a history of pelvic radiation
- a history of tamoxifen use.
Counseling the patient
OBG Management: If tissue extraction is an issue, and morcellation will be necessary for a minimally invasive approach, how do you counsel the patient?
Dr. Fader: We have an informed consent protocol we use at Hopkins in this regard. We speak extensively to our patients about the fact that every procedure or intervention performed in medicine carries a benefit/risk ratio. We inform patients of the FDA morcellation safety statement—that their fibroid or fibroids may contain unexpected cancerous tissue and that laparoscopic electromechanical morcellation may spread the cancer and possibly worsen their prognosis. We also explain that, while the FDA quotes a risk of approximately 1 in 350 for occult sarcoma, that this data review was somewhat limited in scope and included postmenopausal women in the estimates. Based on the best available published systematic reviews and internal Hopkins data, we believe the risk of morcellating an occult uterine malignancy in a woman of reproductive age is more likely on the order of approximately 1 in 1,700 to 1 in 8,333.
We discuss our institution’s approach to tissue extraction (ie, that we no longer perform open electromechanical morcellation but instead perform contained tissue extraction on an institutional review board protocol). We tell patients that contained tissue-extraction practices are still experimental, although there is published literature preliminarily supporting the safety of the practice. In addition, we discuss the fact that contained tissue extraction has been used for years to safely remove large intra-abdominal specimens from laparoscopic incisions, from adnexal tissue to gallbladder, spleen, kidney, intestinal, and appendiceal specimens.
We also explain that, as physicians, we do our best to ensure that risks are minimized and reasonable in relation to anticipated benefits but that, even when we use the very best diagnostic measures, no test is 100% sensitive or specific to rule out malignancy in this setting (or in any setting, actually).
Finally, we discuss the fact that minimally invasive surgery and tissue extraction practices performed in appropriate patients by skilled surgeons conclusively benefit hundreds of thousands of women each year around the world. That is a narrative that has been somewhat lacking in the recent dialogue about tissue-extraction practices.
OBG Management: What do we know about outcomes when a leiomyosarcoma is inadvertently morcellated?
Dr. Fader: This is considered a “cut-through” procedure, in that a cancerous tumor that is potentially contained to an organ is not removed intact or with clean margins. The morcellation procedure effectively cuts through the occult cancer, which is not desirable. Intact surgical removal of uterine cancers or sarcomas is the mainstay of therapy for these malignancies, based on NCCN guidelines.22
OBG Management: Does a morcellated leiomyosarcoma carry a worse prognosis than an unmorcellated leiomyosarcoma?
Dr. Fader: When it comes to morcellated versus “intact” uterine leiomyosarcoma, we enter a largely data-free zone. We don’t know with certainty whether the outcome is worsened. If we’re being intellectually honest, we must admit the possibility that a morcellated cancer is more likely to be disseminated, rendering it potentially more difficult to treat. However, sarcomas primarily spread by hematogenous dissemination. It is quite possible that even the act of incising an intact fibroid in an open abdominal procedure or performing a supracervical or total hysterectomy without morcellation may still result in hematogenous cancer dissemination. So there is no indication yet that electromechanical morcellation poses a unique and higher risk of cancer upstaging or worse prognosis compared with techniques such as open myomectomy, supracervical hysterectomy, or hysteroscopic myomectomy.
In addition, the prognosis associated with even early-stage uterine leiomyosarcoma is uniformly poor. A published study from Hopkins that included 108 patients with uterine leiomyosarcoma suggests that the recurrence rate and survival of patients with early-stage, “intact” leiomyosarcoma are very poor and comparable to the survival rate documented in the literature for women with morcellated sarcomas.23
The few retrospective studies that exist suggesting worse outcomes with morcellation have limitations that preclude any definitive conclusions.2 These studies are marred by small numbers, heterogeneity in morcellation practices, poor follow-up times, and a lack of detail regarding how patients with morcellated versus unmorcellated sarcomas were treated. Nevertheless, a couple of these small retrospective reports indicate the possibility of worse outcomes in women with morcellated uterine sarcomas, compared with historical controls with intact sarcoma removal.
Is the morcellator at fault—or the user?
OBG Management: Some would argue that even one case of a morcellated uterine sarcoma is too many. How would you respond?
Dr. Fader: There is no doubt that a handful of women each year have been harmed by morcellation practices. Those women deserve our dedication and best efforts to learn how to better treat morcellated sarcomas—and more importantly—how to mitigate the risks associated with morcellation practices and reduce the risk of preventable harm for all women undergoing minimally invasive fibroid procedures. I think the single best thing we can do to mitigate risk is to be more conscientious about selecting our patients for tissue-extraction procedures (ie, strict selection criteria, appropriate preoperative work-ups). If we did this, we likely would reduce the number of oncologic morcellator mishaps by 50% to 80% without changing anything else.
When we closely scrutinize the literature, and when I reflect on the women with morcellated cancers that we have cared for at Hopkins, we observe that some (but not all) “occult” uterine cancers were not that hidden after all and may have been detected preoperatively if an effort had been made.
We have noted a number of patients in this setting who experienced harm not because a specific device was used to cut up their uterus but because they were never appropriate candidates for the procedure in the first place. For instance, I recently cared for an elderly woman with a morcellated uterine cancer who underwent a laparoscopic supracervical hysterectomy without an appropriate preoperative work-up (ie, no endometrial biopsy or imaging) or informed consent about the possibility that she might have cancer. If an elderly woman presents with concerning symptoms related to her uterus (ie, enlargement, bleeding), she must be evaluated and counseled regarding the considerable risk of potential malignancy. Even in the setting of a normal work-up, I don’t believe it is a good idea to perform electromechanical morcellation in higher-risk women, including elderly women. That does not mean that select, well-screened women cannot be considered for alternative tissue-extraction techniques, but the risks and benefits must be carefully weighed in each patient, and informed consent must be obtained.
By continuing to refine the safety features of the electromechanical morcellator devices and choosing patients more carefully for minimally invasive procedures and tissue extraction, we likely will reduce the risk of preventable harm in women undergoing gynecologic surgery.
Can leiomyosarcoma be detected preoperatively?
OBG Management: How can we improve preoperative detection of uterine malignancy, particularly leiomyosarcoma?
Dr. Fader: For starters, we can improve detection of uterine cancers by simply looking for them. Almost all epithelial uterine cancers will be detectable by endometrial sampling. A comprehensive history and physical and uterine imaging also may be helpful. And although they are more difficult to detect than epithelial uterine cancers, it is a myth that sarcomas cannot be diagnosed preoperatively. Investigators from Columbia University retrospectively evaluated the ability to preoperatively detect epithelial uterine cancers and uterine sarcomas on final pathology. In 72 women who were ultimately diagnosed with a sarcoma, preoperative endometrial sampling suggested an invasive tumor in 86% and predicted the correct histology in 64%. In fact, the rate of detection of invasive cancer by preoperative sampling was not statistically different among sarcomas than it was among epithelial uterine cancers, although there was less of a correlation with appropriate histology seen with endometrial biopsy.20
That being said, sarcomas can be missed, especially in younger women who have extremely large, degenerated, or necrotic-appearing fibroids. Improvements in diagnostic testing are desperately needed to help distinguish benign fibroids from sarcomas, as there are no reliable modalities to exclude a sarcoma at this time. MRI appears to be the most useful imaging modality, although it cannot definitively distinguish a fibroid from a sarcoma. However, a fairly constant finding in leiomyosarcomas is the absence of calcifications. Further, some studies also suggest that ill-defined margins are consistent with a sarcoma. Finally, several centers, including our own, are studying novel biologic markers and revisiting the utility of previously described markers such as LDH in the preoperative detection of uterine sarcoma.21
The way forward
OBG Management: You have said, “Keep patients informed and safe but avoid being too reactionary.” Could you expand on this statement?
Dr. Fader: Certainly. There are many examples throughout the history of medicine in which treatments have brought benefit to thousands or millions of individuals but may cause harm in a select few. We know that when controversial medical issues have arisen in the past, the pendulum has swung widely in terms of societal response.
For example, the landmark Women’s Health Initiative had an immediate and adverse impact on hormone replacement therapy (HRT) administration. The increased risk of cardiovascular disease and breast cancer observed with use of long-term combination therapy with conjugated equine estrogen and medroxyprogesterone acetate prompted many US health-care providers to abandon use of HRT—until more contemporary data demonstrated that, in younger, healthier postmenopausal women, these adverse events were very rare and the benefits of HRT outweighed the risks.
Can we avoid stroke, heart attack, and breast cancer in all younger women taking HRT? Of course not. But we counsel women about the benefit/risk ratio of the therapy and advise them that the likelihood of these events is rare.
Similarly, as with the HRT analogy, younger women have lower risks associated with surgery and morcellation, compared with older women, and are more likely to derive benefit from the procedure (after ensuring appropriate candidacy with a comprehensive preoperative evaluation and informed consent).
However, if the expectation is that no cases of harm will ever occur with a surgical device or procedure in order for it to be deemed acceptable and safe to use in practice, then that is simply an impossible standard to uphold. There is no device, medication, or intervention I know of in medicine that is completely risk-free.
OBG Management: Are there other examples of this type of benefit/risk assessment?
Dr. Fader: Yes. For instance, tamoxifen is a nonsteroidal anti-estrogen agent approved by the FDA for adjuvant treatment of breast cancer, treatment of metastatic breast cancer, and reduction of breast cancer incidence in high-risk women. Tamoxifen has effectively reduced breast cancer rates and significantly improved survival in select breast cancer patients. Yet, it is well known that long-term use of tamoxifen is associated with a twofold increased risk of uterine cancer and uterine sarcoma—a likely far more commonly occurring adverse event than an occult, morcellated uterine sarcoma during a minimally invasive gynecologic procedure.
Should the FDA ban or significantly curtail the use of tamoxifen? No, not likely, because the benefits far outweigh the risks in previvors and hormone-positive breast cancer survivors. “Keep patients informed and safe but avoid being too reactionary” means that we must do our due diligence as physicians by comprehensively counseling and obtaining informed consent from our patients before performing medical interventions. We also must closely scrutinize and improve upon practices that may cause harm.
However, what this also means is that while it may be prudent to restrict or limit a surgical practice in select higher-risk populations or modify it in some way to make it safer, we shouldn’t necessarily completely abandon or ban a practice that has benefited hundreds of thousands of patients at low risk of harm until we have objectively reviewed all of the available science and fully understand the implications of a practice change (ie, would the risks of preventable harm be even greater for women if more of them had to undergo open abdominal surgery?). We need continued cool heads and sound scientific reasoning to decide upon health-care policy changes or treatment paradigm shifts.
At the end of the day, however, it is paramount that we mitigate patient harm. The subject of tissue extraction during minimally invasive surgery is a complex and nuanced issue that merits continued study and open-minded and intelligent dialogue between patient stakeholders, clinicians, scientists, industry, ethicists, regulatory agencies, and the press. I think we can all appreciate how humbling and challenging the morcellation issue has been for many of us, especially our patients—particularly the unfortunate women who have been diagnosed with a uterine sarcoma.
Like many of my colleagues, I have been privileged to care for a number of women with uterine leiomyosarcoma. It is a devastating disease, and the prognosis is very poor, whether it is morcellated or removed intact. We are fortunate that the vast majority of women who undergo a minimally invasive procedure for fibroids or other presumed benign indications will not be at risk for an occult malignancy. But what can we do now to continue to offer the benefits of minimally invasive surgery and tissue extraction to women while simultaneously reducing the risk to the select few who will develop a rare uterine cancer?
We can all make a greater effort to more carefully select our patients for minimally invasive surgery and tissue extraction, to limit the performance of open electromechanical morcellation and collaborate in studying the role of refined tissue-extraction techniques and containment systems, to enhance the informed consent process, to develop improved diagnostic tests for preoperative cancer detection, and to conduct higher-quality studies on minimally invasive tissue-extraction techniques for regulatory agencies to review in the near future. It also goes without saying that we need more federal funding to study rare tumors (and gynecologic cancers in general) and develop better sarcoma treatments.
Although there is no medical treatment or surgical procedure that is completely risk-free, interventions such as HRT, tamoxifen, and uterine morcellation—when used in appropriate patients and for appropriate indications—will allow preventable harm to be minimized and make it possible for countless women to continue to derive tremendous benefit.
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
1. US Food and Drug Administration. Laparoscopic uterine power morcellation in hysterectomy and myomectomy: FDA Safety Communication. http://www.fda.gov/MedicalDevices/Safety/AlertsandNotices/ucm393576.htm. Published April 17, 2014. Accessed February 3, 2014.
2. AAGL. Morcellation during uterine tissue extraction. http://www.aagl.org/wp-content/uploads/2014/05/Tissue_Extraction_TFR.pdf. Accessed February 3, 2015.
3. American College of Obstetricians and Gynecologists. Power morcellation and occult malignancy in gynecologic surgery: a special report. http://www.acog.org/Resources-And-Publications/Task-Force-and-Work-Group-Reports/Power-Morcellation-and-Occult-Malignancy-in-Gynecologic-Surgery. Published May 2014. Accessed February 3, 2015.
4. US Food and Drug Administration. Updated Laparoscopic uterine power morcellation in hysterectomy and myomectomy: FDA Safety Communication. http://www.fda.gov/MedicalDevices/Safety/AlertsandNotices/ucm424443.htm. Published November 24, 2014. Accessed February 2, 2015.
5. American Cancer Society. Uterine sarcoma: What is uterine sarcoma? http://www.cancer.org/cancer/uterinesarcoma/detailedguide/uterine-sarcoma-what-is-uterine-sarcoma. Updated January 12, 2015. Accessed February 3, 2015.
6. D’Angelo E, Prat J. Uterine sarcomas: a review. Gynecol Oncol. 2010;116(1):131–139.
7. Chittawar B, Franik S, Pouwer AW, Farquhar C. Minimally invasive surgical techniques versus open myomectomy for uterine fibroids. Cochrane Database Syst Rev. 2014;10:CD004638. doi:10.1002/14651858.CD004638.pub3.
8. Mori KM, Neubauer NL. Minimally invasive surgery in gynecologic oncology. ISRN Obstet Gynecol. 2013; article ID 312982. http://dx.doi.org/10.1155/2013/312982. Accessed February 2, 2015.
9. Li G, Yan X, Shang H, Wang G, Chen L, Han Y. A comparison of laparoscopic radical hysterectomy and pelvic lymphadenectomy and laparotomy in the treatment of Ib IIa cervical cancer. Gynecol Oncol. 2007;105(1):176–180.
10. Walker JL, Piedmonte MR, Spirtos NM, et al. Laparoscopy compared with laparotomy for comprehensive surgical staging of uterine cancer: Gynecologic Oncology Group Study Lap2. J Clin Oncol. 2009;27(32):5331–5336.
11. Ramos A, Fader AN, Long Roche K. Surgical cytoreduction for disseminated benign disease after open power uterine morcellation. Obstet Gynecol. 2014;125(1):99–102.
12. Theben J, Schellong A, Altgassen C, Kelling K, Schneider S, Grobe-Drieling D. Unexpected malignancies after laparoscopic-assisted supracervical hysterectomies (LASH): an analysis of 1,584 LASH cases. Arch Gynecol Obstet. 2013;287(3):455–462.
13. Parker WH, Fu YS, Berek JS. Uterine sarcoma in patients operated on for presumed leiomyoma and rapidly growing leiomyoma. Obstet Gynecol. 1994;83(3):414–418.
14. Seidman MA, Oduyebo T, Muto MG, Crum CP, Nucci MR, Quade BJ. Peritoneal dissemination complicating morcellation of uterine mesenchymal neoplasms. PLoS One. 2012;7(11):e50058.
15. Pritts EA, Parker WH, Brown J, Olive DL. Outcome of occult uterine leiomyosarcoma after surgery for presumed uterine fibroids: a systematic review. J Minim Invasive Gynecol. 2015;22(1):26–33.
16. Wright JD, Tergas AI, Burke WM, et al. Prevalence of uterine pathology in women undergoing minimally invasive hysterectomy employing electric power morcellation. JAMA. 2014;312(12):1253–1255.
17. Wright JD, Tergas AI, Burke WM, et al. Uterine pathology in women undergoing minimally invasive hysterectomy using morcellation. JAMA. 2014;312(12):1253–1255.
18. Wright JD, Tergas AI, Cul R, et al. Use of electric power morcellation and prevalence of underlying cancer in women who undergo myomectomy. JAMA Oncol. 2015; published online February 19, 2015. doi:10.1001/jamaoncol.2014.206.
19. Goto A, Takeuchi S, Sugimura K, Maruo T. Usefulness of Gd-DTPA contrast-enhanced dynamic MRI and serum determination of LDH and its isozymes in the differential diagnosis of leiomyosarcoma from degenerated leiomyoma of the uterus. Int J Gynecol Cancer. 2002; 12:354.
20. Ricci S, Giuntoli RL 2nd, Eisenhauer E, et al. Does adjuvant chemotherapy improve survival for women with early-stage uterine leiomyosarcoma? Gynecol Oncol. 2013;131(3):629–633.
21. Bansal N, Herzog TJ, Burke W, Cohen CJ, Wright JD. The utility of preoperative endometrial sampling for the detection of uterine sarcomas. Gynecol Oncol. 2008;110(1):43–48.
22. Koh WJ, Greer BE, Abu-Rustum NR, et al. Uterine neoplasms, version 1.2014. J Natl Compr Canc Netw. 2014;12(2):248–280.
23. Schwartz LB, Zawin M, Carcangiu ML, Lange R, McCarthy S. Does pelvic magnetic resonance imaging differentiate among the histologic subtypes of uterine leiomyomata? Fertil Steril. 1998;70(3):580–587.
The year 2014 marked a sea change in our approach to tissue extraction during minimally invasive surgery. The US Food and Drug Administration (FDA) initiated this transformation in April, when it issued a safety warning on the use of open power morcellation.1 A flurry of statements on the practice followed from professional societies, capped, in late November, with another statement from the FDA.2–4 The new bottom line: The use of open electromechanical (“power”) morcellation is contraindicated in perimenopausal and postmenopausal women, as well as in those who are known or suspected to have a malignancy.4
Most of the concern to date has centered on the risk that an occult leiomyosarcoma could be morcellated inadvertently during uterine surgery, an event that may worsen the prognosis for the patient. To get a gynecologic oncologist’s take on the controversy, OBG Management caught up with Amanda Nickles Fader, MD, director of the Kelly Gynecologic Oncology Service at Johns Hopkins University. Dr. Fader’s perspective is unique in that she treats a relatively high number of patients who have leiomyosarcoma and other uterine cancers.
In this Q&A, we discuss the patient population at Johns Hopkins; why Dr. Fader is especially qualified to speak to the future of electromechanical morcellation in gynecologic surgery; the benefits and risks of minimally invasive surgery, including tissue extraction; her recommendations for preoperative evaluation and counseling of patients undergoing uterine surgery; and guidance on how the specialty of gynecologic surgery should proceed in the future.
OBG Management: Dr. Fader, by way of introduction, could you characterize your patient population?
Amanda Nickles Fader, MD: Like most gynecologic oncologists, I primarily treat women with cancers of the uterus, ovary, cervix, and vulva. Many of us also have the opportunity to treat a number of women each year with complex benign gynecologic conditions that require surgery. As someone who is extremely interested in rare gynecologic tumors, I also treat a relatively high volume of women diagnosed with uterine sarcoma.
Approximately 75% of the women I see in my practice have preinvasive or invasive cancer, and 25% have a benign condition, such as enlarged fibroids or advanced-stage endometriosis.
OBG Management: How many cases of uterine sarcoma do you encounter on an annual basis?
Dr. Fader: Uterine sarcomas are very rare. They represent only 2% of all uterine cancers. Put into perspective, that means that about 0.4 cases of leiomyosarcoma occur in every 100,000 US women—most commonly postmenopausal women. Leiomyosarcoma is a biologically aggressive, high-grade malignancy that often is lethal.5
Endometrial stroma sarcoma is even less common—only 0.3 cases occur in every 100,000 US women. However, this tumor type is more indolent, often diagnosed at an earlier stage, and potentially curable with surgery (with or without hormonal therapy).6
As a referral center for rare uterine tumors, the Kelly Gynecologic Oncology Service and the Sarcoma Center at Johns Hopkins see approximately 35 to 40 new cases of uterine sarcoma annually for treatment of primary disease or recurrence. An additional 15 to 20 consult cases are reviewed from outside hospitals each year by our gynecologic pathology department.
Why a minimally invasive approach is vital
OBG Management: When it comes to uterine surgery for presumed benign conditions, why is a minimally invasive approach important?
Dr. Fader: Minimally invasive surgery clearly benefits women and is one of the greatest advances of the past half-century within our field. Randomized controlled trials and meta-analyses have demonstrated without question that women who undergo minimally invasive surgery for benign conditions or early-stage cancerous gynecologic conditions have superior clinical outcomes, compared with women who undergo surgery via laparotomy.7,8 These outcomes include fewer perioperative complications (including fewer cases of surgical site infection, venous thromboembolism, wound dehiscence, and hospital readmission), shorter hospital stays, less pain, faster recovery, and fewer adhesions. And when women with early-stage cancers undergo minimally invasive surgery, randomized controlled trials show, they have a stage-specific survival rate similar to that observed in women treated with laparotomy.9
Benefits and risks of tissue extraction in minimally invasive surgery
OBG Management: What are the main benefits of tissue extraction, including morcellation?
Dr. Fader: Tissue extraction is a practice that has allowed us to offer minimally invasive surgery to countless more women than we could have in the recent past. It is a technique in which a large specimen (typically a uterus or fibroid) is fragmented into smaller parts in order to remove it through a small laparoscopic incision or orifice (vagina, umbilicus). Without tissue-extraction practices, thousands of women who undergo myomectomy each year to conserve their fertility and hundreds of thousands of women who require hysterectomy potentially would have to undergo a more painful and risky surgery through a larger abdominal incision. That would not be desirable, as we know conclusively that laparotomy is associated with worse outcomes—and even an increased risk of mortality—compared with minimally invasive surgery performed by experienced surgeons.10
Tissue extraction can be approached in a variety of ways. It can be performed with a scalpel, with a resectoscope, or with an electromechanical morcellator. Tissue extraction can be performed within the uterine cavity, through the vagina, within the abdominal compartment, or within a containment system in any of those compartments.
OBG Management: What are the risks of tissue extraction?
Dr. Fader: The risks of tissue extraction with electromechanical morcellation include potential injury to intra-abdominal organs and vasculature and risk of dissemination of an occult (ie, undiagnosed) uterine cancer. A report by our research group also demonstrated the risks of dissemination of benign uterine tissue requiring subsequent surgery in the setting of open electromechanical morcellation.11 The risk of these events occurring in the hands of a thoughtful and experienced surgeon who conducts comprehensive preoperative patient evaluations is extremely low. However, recent evidence demonstrates that a handful of women worldwide are diagnosed with an occult uterine cancer each year during a morcellation procedure.12–14 Although it is a very rare event (given that most women undergoing hysterectomy and myomectomy procedures are of reproductive age and unlikely to have uterine cancer), this risk is a serious issue. There is an exigent need for the gynecologic surgical community to develop better approaches to tissue extraction that minimize preventable harm in women.
Needed: high-quality data on the risks of morcellation
OBG Management: How does the recent FDA statement urging caution with the use of open power morcellation factor into this equation? The most recent FDA statement noted that open power morcellation is contraindicated in perimenopausal and postmenopausal women.4
Dr. Fader: The FDA’s concern is legitimate. However, the magnitude of the risk of occult uterine sarcoma in women undergoing presumed benign gynecologic surgery has been scrutinized and debated. The FDA panel quoted a risk that roughly 1 in 350 women undergoing presumed benign gynecologic surgery for fibroids will have an occult leiomyosarcoma diagnosed. However, more recent systematic reviews and a review of the prospective published literature demonstrate that the risk is more likely on the order of 1 in 1,700 to 1 in 8,333 women.15 The risk may be even lower in gynecologic surgery practices that see a high volume of hysterectomy/myomectomy cases and utilize meticulous preoperative patient selection criteria to establish a woman’s candidacy for tissue-extraction procedures.
I am concerned with how “occult sarcoma” discovered during surgery for “presumed benign gynecologic disease is being defined in the literature. There is no uniform definition being used. A cancer in this setting is only truly occult or undiagnosed if the physician was thinking about it and made every effort to rule out cancer preoperatively, and the morcellation procedure was performed in a low-risk population (but cancer was still diagnosed on final pathology in this population). However, in the majority of the morcellation studies in the literature, it is not clear that thorough preoperative evaluations occurred in patients to rule out uterine malignancy—in fact, there is a paucity of published information regarding establishing appropriate patient candidacy for morcellation procedures. So we can’t derive any conclusions regarding whether “occult” sarcomas were truly undetectable or not in the published literature.
In addition, the literature is very clear that advancing age and postmenopausal status are risk factors for uterine malignancy.16 The vast majority of uterine sarcoma cases are diagnosed in postmenopausal women. Yet, in one large US population-based hysterectomy study, 20% of the morcellated cases (and the overwhelming majority of the “occult” morcellated uterine cancers) occurred in postmenopausal women!17
Further, in a more recent study by the same authors, again the risks of morcellating a uterine cancer in a population undergoing myomectomy was significantly higher in a postmenopausal population and occurred only rarely in women younger than age 40. But it should come as no surprise that a greater incidence of uterine cancer was identified in these older cohorts—cancer risk increases precipitously with age. That’s not “occult”; that’s basic cancer epidemiology.
In other words, we cannot assume from population-based administrative claims data that morcellation performed in inappropriate populations at higher risk of uterine malignancy (in which we do not know whether patients were properly screened for the procedure preoperatively or whether they had risk factors for uterine cancer but were presumably poor candidates for morcellation due to age alone) helps us define the true incidence of “occult” sarcoma or cancer in a population.
These studies are provocative, however, and do inform us that, as women get older, we are apt to see a greater incidence of uterine cancer. We cannot safely assume that a postmenopausal or elderly woman with symptomatic or enlarging fibroids has “presumed benign disease”—it is cancer until proven otherwise, and we need to be looking for it preoperatively. Therefore, we need to be particularly careful with our surgical practices in this population—ie, the basis for the FDA’s recommendation to avoid morcellation in older women. And I agree with the FDA that open electromechanical morcellation generally is contraindicated in postmenopausal women. However, we need better data from large prospective studies to inform our understanding of the true incidence of undiagnosed or “occult” uterine sarcoma in women undergoing surgery for presumed benign disease. These future studies are likely to demonstrate what we already know—that in young, well-screened, well-selected candidates for minimally invasive hysterectomy or myomectomy, the risk of occult cancer is going to be exceptionally low.
OBG Management: Which is greater—the risks or benefits of tissue extraction?
Dr. Fader: Assessment of risks and benefits in medicine has everything to do with the intervention in question as applied to an individual patient. At the end of the day, there are risks and benefits to every medical or surgical treatment offered to patients in every medical and surgical discipline. But the risk of an occult uterine sarcoma is extremely low in a woman of reproductive age who has been properly selected and comprehensively evaluated for minimally invasive surgery and tissue extraction prior to surgery. And this small—though not negligible—risk must be weighed against the much higher risk of harm that may be incurred with an open abdominal procedure, compared with minimally invasive surgery.
However, in many elderly women, the risks of tissue extraction with an electromechanical morcellator may outweigh the benefits. Even so, there are exceptions in which tissue extraction may be acceptable in postmenopausal women (ie, using alternative tissue-extraction methods in those undergoing minimally invasive supracervical hysterectomy and sacral colpopexy for pelvic organ prolapse).
Few of the data published on the risks of morcellation are of very high quality in terms of scientific rigor or methodology. The best thing we can do as a gynecologic surgical community is conduct sound quality-improvement (QI) programs, disseminate our QI results, publish our data, establish guidelines for best practices in uterine tissue extraction, and collaborate readily to increase the scholarly output on this issue so that national societies and government regulatory agencies have better-quality data to inform future policy on this issue.
A case-based approach
OBG Management: How would you approach tissue extraction in the following case?
CASE: A desire for myomectomy
A 35-year-old woman (G1P1) who delivered by cesarean has an 8-cm symptomatic intramural fibroid. She has regular heavy periods that have led to anemia (hemoglobin level = 10 mg/dL). Her medical history is negative for malignancy, pelvic radiation, or tamoxifen use, and she wants to preserve her fertility. Magnetic resonance imaging (MRI) confirms an 8-cm fibroid. Endometrial biopsy results are negative.
Dr. Fader: At Johns Hopkins, as at many other centers, we use well-defined criteria to determine whether a minimally invasive approach (and tissue extraction) might be appropriate. We also individualize treatment and surgical decision-making on a case-by-case basis. Any candidate for minimally invasive surgery and tissue extraction for uterine fibroids must undergo a thorough preoperative assessment.
Johns Hopkins preoperative assessment criteria include:
- endometrial biopsy
- imaging (often MRI)
- a detailed history and physical, with a comprehensive review of risk factors for malignancy, including family and genetic history or a personal history of malignancy, pelvic radiation, tamoxifen use, or BRCA or hereditary nonpolyposis colorectal cancer (HNPCC) deleterious mutation carrier status, among other things.
- In addition, all cases are discussed at a peer-reviewed, preoperative conference to ensure that a thorough work-up has been conducted and to verify the patient’s candidacy for a minimally invasive procedure with tissue extraction. As the FDA recommends, we conduct an enhanced informed consent process and counsel patients being considered for tissue extraction about the risk of occult sarcoma.
Our top priority is patient safety, so until more data are available, we no longer perform open electromechanical morcellation. We perform all tissue extraction within a containment system and under institutional review board protocol. We primarily perform tissue extraction via scalpel morcellation.
OBG Management: How do the patient’s wishes factor into the decision to perform minimally invasive surgery with morcellation?
Dr. Fader: Our patients make their own decisions regarding surgical approach and procedure after undergoing extensive counseling about the risks and benefits of the proposed procedures. I certainly would offer a patient like the one described in this case the opportunity for a minimally invasive approach (if, after thorough preoperative evaluation, she were deemed to be at very low risk for uterine malignancy). In my experience, most women opt for the minimally invasive approach in this setting; however, if a patient declines minimally invasive surgery, I respect her decision.
Tissue extraction in perimenopause
CASE: A desire for myomectomy at age 48
OBG Management: How would you approach the same case if the patient were a 48-year-old perimenopausal woman?
Dr. Fader: In perimenopausal women, we are more selective about performing morcellation, given the recent FDA safety statement, and because the incidence of occult cancer starts to increase slightly in this patient cohort (although it doesn’t precipitously increase until well into the postmenopausal period).
In addition, myomectomy may have less value in a 48-year-old, given the lower likelihood of achieving successful fertility, although there are exceptions. US cancer statistics and studies on morcellation demonstrate that the vast majority of women in their 30s and 40s have an extremely low risk for sarcomas and other uterine malignancies.2
In select cases in which a woman has undergone a comprehensive preoperative work-up, has a stable-appearing fibroid(s), and is well-educated and counseled about the pros and cons of morcellation, we would consider performing a procedure with contained tissue extraction. As a general matter, however, I would be more inclined to offer a 48-year-old in this situation a uterine artery embolization or minimally invasive hysterectomy than a myomectomy procedure, especially given the recent study by Wright and colleagues demonstrating the significantly increased risks of uterine cancer at myomectomy surgery for a woman in her late 40s or early 50s.18
Preoperative assessment should be comprehensive
OBG Management: What preoperative evaluation do you perform when tissue extraction, including morcellation, is an issue?
Dr. Fader: It is the policy at Johns Hopkins that all women being considered for minimally invasive surgery and tissue extraction must undergo a rigorous preoperative work-up that includes:
- a comprehensive history and physical (to exclude malignancy and risk for occult malignancy)
- an endometrial evaluation (most commonly, an endometrial biopsy)
- uterine imaging (with longitudinal evaluation of imaging findings if performed previously)
- discussion of each patient case at peer-reviewed, preoperative department conferences.
We have separate conferences for the gynecology and gynecologic oncology services and have employed this practice for many years at Hopkins. We are also studying the role of serum lactate dehydrogenase (LDH) isoenzyme levels in stratifying women with uterine fibroids versus cancer/sarcoma.19
OBG Management: Which patients would you exclude from the electromechanical morcellation option?
Dr. Fader: As the American College of Obstetricians and Gynecologists, the AAGL, the Society of Gynecologic Oncology, and the Society of Gynecologic Surgeons appear to agree:
- When utilized in select patients of reproductive age, minimally invasive surgery and morcellation are beneficial.
- Morcellation should categorically not be performed in any woman who has a known or suspected uterine (or other gynecologic) malignancy.2,3
At our institution, we have significantly curtailed the use of electromechanical morcellation at this time and especially do not perform it in women aged 50 or older or in those with confirmed postmenopausal status. We also do not perform electromechanical morcellation in women with a personal history of uterine, cervical, or ovarian preinvasive or invasive cancer, or in women with a strong family history of gynecologic malignancy.
Other populations we exclude are women with:
- a history of mitotically active or atypical fibroids (as determined at previous myomectomy)
- known BRCA or HNPCC deleterious mutation, hereditary leiomyomatosis and renal cell carcinoma (HLRCC) syndrome, hereditary childhood retinoblastoma, or other genetic predisposition to uterine or ovarian cancer
- a history of pelvic radiation
- a history of tamoxifen use.
Counseling the patient
OBG Management: If tissue extraction is an issue, and morcellation will be necessary for a minimally invasive approach, how do you counsel the patient?
Dr. Fader: We have an informed consent protocol we use at Hopkins in this regard. We speak extensively to our patients about the fact that every procedure or intervention performed in medicine carries a benefit/risk ratio. We inform patients of the FDA morcellation safety statement—that their fibroid or fibroids may contain unexpected cancerous tissue and that laparoscopic electromechanical morcellation may spread the cancer and possibly worsen their prognosis. We also explain that, while the FDA quotes a risk of approximately 1 in 350 for occult sarcoma, that this data review was somewhat limited in scope and included postmenopausal women in the estimates. Based on the best available published systematic reviews and internal Hopkins data, we believe the risk of morcellating an occult uterine malignancy in a woman of reproductive age is more likely on the order of approximately 1 in 1,700 to 1 in 8,333.
We discuss our institution’s approach to tissue extraction (ie, that we no longer perform open electromechanical morcellation but instead perform contained tissue extraction on an institutional review board protocol). We tell patients that contained tissue-extraction practices are still experimental, although there is published literature preliminarily supporting the safety of the practice. In addition, we discuss the fact that contained tissue extraction has been used for years to safely remove large intra-abdominal specimens from laparoscopic incisions, from adnexal tissue to gallbladder, spleen, kidney, intestinal, and appendiceal specimens.
We also explain that, as physicians, we do our best to ensure that risks are minimized and reasonable in relation to anticipated benefits but that, even when we use the very best diagnostic measures, no test is 100% sensitive or specific to rule out malignancy in this setting (or in any setting, actually).
Finally, we discuss the fact that minimally invasive surgery and tissue extraction practices performed in appropriate patients by skilled surgeons conclusively benefit hundreds of thousands of women each year around the world. That is a narrative that has been somewhat lacking in the recent dialogue about tissue-extraction practices.
OBG Management: What do we know about outcomes when a leiomyosarcoma is inadvertently morcellated?
Dr. Fader: This is considered a “cut-through” procedure, in that a cancerous tumor that is potentially contained to an organ is not removed intact or with clean margins. The morcellation procedure effectively cuts through the occult cancer, which is not desirable. Intact surgical removal of uterine cancers or sarcomas is the mainstay of therapy for these malignancies, based on NCCN guidelines.22
OBG Management: Does a morcellated leiomyosarcoma carry a worse prognosis than an unmorcellated leiomyosarcoma?
Dr. Fader: When it comes to morcellated versus “intact” uterine leiomyosarcoma, we enter a largely data-free zone. We don’t know with certainty whether the outcome is worsened. If we’re being intellectually honest, we must admit the possibility that a morcellated cancer is more likely to be disseminated, rendering it potentially more difficult to treat. However, sarcomas primarily spread by hematogenous dissemination. It is quite possible that even the act of incising an intact fibroid in an open abdominal procedure or performing a supracervical or total hysterectomy without morcellation may still result in hematogenous cancer dissemination. So there is no indication yet that electromechanical morcellation poses a unique and higher risk of cancer upstaging or worse prognosis compared with techniques such as open myomectomy, supracervical hysterectomy, or hysteroscopic myomectomy.
In addition, the prognosis associated with even early-stage uterine leiomyosarcoma is uniformly poor. A published study from Hopkins that included 108 patients with uterine leiomyosarcoma suggests that the recurrence rate and survival of patients with early-stage, “intact” leiomyosarcoma are very poor and comparable to the survival rate documented in the literature for women with morcellated sarcomas.23
The few retrospective studies that exist suggesting worse outcomes with morcellation have limitations that preclude any definitive conclusions.2 These studies are marred by small numbers, heterogeneity in morcellation practices, poor follow-up times, and a lack of detail regarding how patients with morcellated versus unmorcellated sarcomas were treated. Nevertheless, a couple of these small retrospective reports indicate the possibility of worse outcomes in women with morcellated uterine sarcomas, compared with historical controls with intact sarcoma removal.
Is the morcellator at fault—or the user?
OBG Management: Some would argue that even one case of a morcellated uterine sarcoma is too many. How would you respond?
Dr. Fader: There is no doubt that a handful of women each year have been harmed by morcellation practices. Those women deserve our dedication and best efforts to learn how to better treat morcellated sarcomas—and more importantly—how to mitigate the risks associated with morcellation practices and reduce the risk of preventable harm for all women undergoing minimally invasive fibroid procedures. I think the single best thing we can do to mitigate risk is to be more conscientious about selecting our patients for tissue-extraction procedures (ie, strict selection criteria, appropriate preoperative work-ups). If we did this, we likely would reduce the number of oncologic morcellator mishaps by 50% to 80% without changing anything else.
When we closely scrutinize the literature, and when I reflect on the women with morcellated cancers that we have cared for at Hopkins, we observe that some (but not all) “occult” uterine cancers were not that hidden after all and may have been detected preoperatively if an effort had been made.
We have noted a number of patients in this setting who experienced harm not because a specific device was used to cut up their uterus but because they were never appropriate candidates for the procedure in the first place. For instance, I recently cared for an elderly woman with a morcellated uterine cancer who underwent a laparoscopic supracervical hysterectomy without an appropriate preoperative work-up (ie, no endometrial biopsy or imaging) or informed consent about the possibility that she might have cancer. If an elderly woman presents with concerning symptoms related to her uterus (ie, enlargement, bleeding), she must be evaluated and counseled regarding the considerable risk of potential malignancy. Even in the setting of a normal work-up, I don’t believe it is a good idea to perform electromechanical morcellation in higher-risk women, including elderly women. That does not mean that select, well-screened women cannot be considered for alternative tissue-extraction techniques, but the risks and benefits must be carefully weighed in each patient, and informed consent must be obtained.
By continuing to refine the safety features of the electromechanical morcellator devices and choosing patients more carefully for minimally invasive procedures and tissue extraction, we likely will reduce the risk of preventable harm in women undergoing gynecologic surgery.
Can leiomyosarcoma be detected preoperatively?
OBG Management: How can we improve preoperative detection of uterine malignancy, particularly leiomyosarcoma?
Dr. Fader: For starters, we can improve detection of uterine cancers by simply looking for them. Almost all epithelial uterine cancers will be detectable by endometrial sampling. A comprehensive history and physical and uterine imaging also may be helpful. And although they are more difficult to detect than epithelial uterine cancers, it is a myth that sarcomas cannot be diagnosed preoperatively. Investigators from Columbia University retrospectively evaluated the ability to preoperatively detect epithelial uterine cancers and uterine sarcomas on final pathology. In 72 women who were ultimately diagnosed with a sarcoma, preoperative endometrial sampling suggested an invasive tumor in 86% and predicted the correct histology in 64%. In fact, the rate of detection of invasive cancer by preoperative sampling was not statistically different among sarcomas than it was among epithelial uterine cancers, although there was less of a correlation with appropriate histology seen with endometrial biopsy.20
That being said, sarcomas can be missed, especially in younger women who have extremely large, degenerated, or necrotic-appearing fibroids. Improvements in diagnostic testing are desperately needed to help distinguish benign fibroids from sarcomas, as there are no reliable modalities to exclude a sarcoma at this time. MRI appears to be the most useful imaging modality, although it cannot definitively distinguish a fibroid from a sarcoma. However, a fairly constant finding in leiomyosarcomas is the absence of calcifications. Further, some studies also suggest that ill-defined margins are consistent with a sarcoma. Finally, several centers, including our own, are studying novel biologic markers and revisiting the utility of previously described markers such as LDH in the preoperative detection of uterine sarcoma.21
The way forward
OBG Management: You have said, “Keep patients informed and safe but avoid being too reactionary.” Could you expand on this statement?
Dr. Fader: Certainly. There are many examples throughout the history of medicine in which treatments have brought benefit to thousands or millions of individuals but may cause harm in a select few. We know that when controversial medical issues have arisen in the past, the pendulum has swung widely in terms of societal response.
For example, the landmark Women’s Health Initiative had an immediate and adverse impact on hormone replacement therapy (HRT) administration. The increased risk of cardiovascular disease and breast cancer observed with use of long-term combination therapy with conjugated equine estrogen and medroxyprogesterone acetate prompted many US health-care providers to abandon use of HRT—until more contemporary data demonstrated that, in younger, healthier postmenopausal women, these adverse events were very rare and the benefits of HRT outweighed the risks.
Can we avoid stroke, heart attack, and breast cancer in all younger women taking HRT? Of course not. But we counsel women about the benefit/risk ratio of the therapy and advise them that the likelihood of these events is rare.
Similarly, as with the HRT analogy, younger women have lower risks associated with surgery and morcellation, compared with older women, and are more likely to derive benefit from the procedure (after ensuring appropriate candidacy with a comprehensive preoperative evaluation and informed consent).
However, if the expectation is that no cases of harm will ever occur with a surgical device or procedure in order for it to be deemed acceptable and safe to use in practice, then that is simply an impossible standard to uphold. There is no device, medication, or intervention I know of in medicine that is completely risk-free.
OBG Management: Are there other examples of this type of benefit/risk assessment?
Dr. Fader: Yes. For instance, tamoxifen is a nonsteroidal anti-estrogen agent approved by the FDA for adjuvant treatment of breast cancer, treatment of metastatic breast cancer, and reduction of breast cancer incidence in high-risk women. Tamoxifen has effectively reduced breast cancer rates and significantly improved survival in select breast cancer patients. Yet, it is well known that long-term use of tamoxifen is associated with a twofold increased risk of uterine cancer and uterine sarcoma—a likely far more commonly occurring adverse event than an occult, morcellated uterine sarcoma during a minimally invasive gynecologic procedure.
Should the FDA ban or significantly curtail the use of tamoxifen? No, not likely, because the benefits far outweigh the risks in previvors and hormone-positive breast cancer survivors. “Keep patients informed and safe but avoid being too reactionary” means that we must do our due diligence as physicians by comprehensively counseling and obtaining informed consent from our patients before performing medical interventions. We also must closely scrutinize and improve upon practices that may cause harm.
However, what this also means is that while it may be prudent to restrict or limit a surgical practice in select higher-risk populations or modify it in some way to make it safer, we shouldn’t necessarily completely abandon or ban a practice that has benefited hundreds of thousands of patients at low risk of harm until we have objectively reviewed all of the available science and fully understand the implications of a practice change (ie, would the risks of preventable harm be even greater for women if more of them had to undergo open abdominal surgery?). We need continued cool heads and sound scientific reasoning to decide upon health-care policy changes or treatment paradigm shifts.
At the end of the day, however, it is paramount that we mitigate patient harm. The subject of tissue extraction during minimally invasive surgery is a complex and nuanced issue that merits continued study and open-minded and intelligent dialogue between patient stakeholders, clinicians, scientists, industry, ethicists, regulatory agencies, and the press. I think we can all appreciate how humbling and challenging the morcellation issue has been for many of us, especially our patients—particularly the unfortunate women who have been diagnosed with a uterine sarcoma.
Like many of my colleagues, I have been privileged to care for a number of women with uterine leiomyosarcoma. It is a devastating disease, and the prognosis is very poor, whether it is morcellated or removed intact. We are fortunate that the vast majority of women who undergo a minimally invasive procedure for fibroids or other presumed benign indications will not be at risk for an occult malignancy. But what can we do now to continue to offer the benefits of minimally invasive surgery and tissue extraction to women while simultaneously reducing the risk to the select few who will develop a rare uterine cancer?
We can all make a greater effort to more carefully select our patients for minimally invasive surgery and tissue extraction, to limit the performance of open electromechanical morcellation and collaborate in studying the role of refined tissue-extraction techniques and containment systems, to enhance the informed consent process, to develop improved diagnostic tests for preoperative cancer detection, and to conduct higher-quality studies on minimally invasive tissue-extraction techniques for regulatory agencies to review in the near future. It also goes without saying that we need more federal funding to study rare tumors (and gynecologic cancers in general) and develop better sarcoma treatments.
Although there is no medical treatment or surgical procedure that is completely risk-free, interventions such as HRT, tamoxifen, and uterine morcellation—when used in appropriate patients and for appropriate indications—will allow preventable harm to be minimized and make it possible for countless women to continue to derive tremendous benefit.
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
The year 2014 marked a sea change in our approach to tissue extraction during minimally invasive surgery. The US Food and Drug Administration (FDA) initiated this transformation in April, when it issued a safety warning on the use of open power morcellation.1 A flurry of statements on the practice followed from professional societies, capped, in late November, with another statement from the FDA.2–4 The new bottom line: The use of open electromechanical (“power”) morcellation is contraindicated in perimenopausal and postmenopausal women, as well as in those who are known or suspected to have a malignancy.4
Most of the concern to date has centered on the risk that an occult leiomyosarcoma could be morcellated inadvertently during uterine surgery, an event that may worsen the prognosis for the patient. To get a gynecologic oncologist’s take on the controversy, OBG Management caught up with Amanda Nickles Fader, MD, director of the Kelly Gynecologic Oncology Service at Johns Hopkins University. Dr. Fader’s perspective is unique in that she treats a relatively high number of patients who have leiomyosarcoma and other uterine cancers.
In this Q&A, we discuss the patient population at Johns Hopkins; why Dr. Fader is especially qualified to speak to the future of electromechanical morcellation in gynecologic surgery; the benefits and risks of minimally invasive surgery, including tissue extraction; her recommendations for preoperative evaluation and counseling of patients undergoing uterine surgery; and guidance on how the specialty of gynecologic surgery should proceed in the future.
OBG Management: Dr. Fader, by way of introduction, could you characterize your patient population?
Amanda Nickles Fader, MD: Like most gynecologic oncologists, I primarily treat women with cancers of the uterus, ovary, cervix, and vulva. Many of us also have the opportunity to treat a number of women each year with complex benign gynecologic conditions that require surgery. As someone who is extremely interested in rare gynecologic tumors, I also treat a relatively high volume of women diagnosed with uterine sarcoma.
Approximately 75% of the women I see in my practice have preinvasive or invasive cancer, and 25% have a benign condition, such as enlarged fibroids or advanced-stage endometriosis.
OBG Management: How many cases of uterine sarcoma do you encounter on an annual basis?
Dr. Fader: Uterine sarcomas are very rare. They represent only 2% of all uterine cancers. Put into perspective, that means that about 0.4 cases of leiomyosarcoma occur in every 100,000 US women—most commonly postmenopausal women. Leiomyosarcoma is a biologically aggressive, high-grade malignancy that often is lethal.5
Endometrial stroma sarcoma is even less common—only 0.3 cases occur in every 100,000 US women. However, this tumor type is more indolent, often diagnosed at an earlier stage, and potentially curable with surgery (with or without hormonal therapy).6
As a referral center for rare uterine tumors, the Kelly Gynecologic Oncology Service and the Sarcoma Center at Johns Hopkins see approximately 35 to 40 new cases of uterine sarcoma annually for treatment of primary disease or recurrence. An additional 15 to 20 consult cases are reviewed from outside hospitals each year by our gynecologic pathology department.
Why a minimally invasive approach is vital
OBG Management: When it comes to uterine surgery for presumed benign conditions, why is a minimally invasive approach important?
Dr. Fader: Minimally invasive surgery clearly benefits women and is one of the greatest advances of the past half-century within our field. Randomized controlled trials and meta-analyses have demonstrated without question that women who undergo minimally invasive surgery for benign conditions or early-stage cancerous gynecologic conditions have superior clinical outcomes, compared with women who undergo surgery via laparotomy.7,8 These outcomes include fewer perioperative complications (including fewer cases of surgical site infection, venous thromboembolism, wound dehiscence, and hospital readmission), shorter hospital stays, less pain, faster recovery, and fewer adhesions. And when women with early-stage cancers undergo minimally invasive surgery, randomized controlled trials show, they have a stage-specific survival rate similar to that observed in women treated with laparotomy.9
Benefits and risks of tissue extraction in minimally invasive surgery
OBG Management: What are the main benefits of tissue extraction, including morcellation?
Dr. Fader: Tissue extraction is a practice that has allowed us to offer minimally invasive surgery to countless more women than we could have in the recent past. It is a technique in which a large specimen (typically a uterus or fibroid) is fragmented into smaller parts in order to remove it through a small laparoscopic incision or orifice (vagina, umbilicus). Without tissue-extraction practices, thousands of women who undergo myomectomy each year to conserve their fertility and hundreds of thousands of women who require hysterectomy potentially would have to undergo a more painful and risky surgery through a larger abdominal incision. That would not be desirable, as we know conclusively that laparotomy is associated with worse outcomes—and even an increased risk of mortality—compared with minimally invasive surgery performed by experienced surgeons.10
Tissue extraction can be approached in a variety of ways. It can be performed with a scalpel, with a resectoscope, or with an electromechanical morcellator. Tissue extraction can be performed within the uterine cavity, through the vagina, within the abdominal compartment, or within a containment system in any of those compartments.
OBG Management: What are the risks of tissue extraction?
Dr. Fader: The risks of tissue extraction with electromechanical morcellation include potential injury to intra-abdominal organs and vasculature and risk of dissemination of an occult (ie, undiagnosed) uterine cancer. A report by our research group also demonstrated the risks of dissemination of benign uterine tissue requiring subsequent surgery in the setting of open electromechanical morcellation.11 The risk of these events occurring in the hands of a thoughtful and experienced surgeon who conducts comprehensive preoperative patient evaluations is extremely low. However, recent evidence demonstrates that a handful of women worldwide are diagnosed with an occult uterine cancer each year during a morcellation procedure.12–14 Although it is a very rare event (given that most women undergoing hysterectomy and myomectomy procedures are of reproductive age and unlikely to have uterine cancer), this risk is a serious issue. There is an exigent need for the gynecologic surgical community to develop better approaches to tissue extraction that minimize preventable harm in women.
Needed: high-quality data on the risks of morcellation
OBG Management: How does the recent FDA statement urging caution with the use of open power morcellation factor into this equation? The most recent FDA statement noted that open power morcellation is contraindicated in perimenopausal and postmenopausal women.4
Dr. Fader: The FDA’s concern is legitimate. However, the magnitude of the risk of occult uterine sarcoma in women undergoing presumed benign gynecologic surgery has been scrutinized and debated. The FDA panel quoted a risk that roughly 1 in 350 women undergoing presumed benign gynecologic surgery for fibroids will have an occult leiomyosarcoma diagnosed. However, more recent systematic reviews and a review of the prospective published literature demonstrate that the risk is more likely on the order of 1 in 1,700 to 1 in 8,333 women.15 The risk may be even lower in gynecologic surgery practices that see a high volume of hysterectomy/myomectomy cases and utilize meticulous preoperative patient selection criteria to establish a woman’s candidacy for tissue-extraction procedures.
I am concerned with how “occult sarcoma” discovered during surgery for “presumed benign gynecologic disease is being defined in the literature. There is no uniform definition being used. A cancer in this setting is only truly occult or undiagnosed if the physician was thinking about it and made every effort to rule out cancer preoperatively, and the morcellation procedure was performed in a low-risk population (but cancer was still diagnosed on final pathology in this population). However, in the majority of the morcellation studies in the literature, it is not clear that thorough preoperative evaluations occurred in patients to rule out uterine malignancy—in fact, there is a paucity of published information regarding establishing appropriate patient candidacy for morcellation procedures. So we can’t derive any conclusions regarding whether “occult” sarcomas were truly undetectable or not in the published literature.
In addition, the literature is very clear that advancing age and postmenopausal status are risk factors for uterine malignancy.16 The vast majority of uterine sarcoma cases are diagnosed in postmenopausal women. Yet, in one large US population-based hysterectomy study, 20% of the morcellated cases (and the overwhelming majority of the “occult” morcellated uterine cancers) occurred in postmenopausal women!17
Further, in a more recent study by the same authors, again the risks of morcellating a uterine cancer in a population undergoing myomectomy was significantly higher in a postmenopausal population and occurred only rarely in women younger than age 40. But it should come as no surprise that a greater incidence of uterine cancer was identified in these older cohorts—cancer risk increases precipitously with age. That’s not “occult”; that’s basic cancer epidemiology.
In other words, we cannot assume from population-based administrative claims data that morcellation performed in inappropriate populations at higher risk of uterine malignancy (in which we do not know whether patients were properly screened for the procedure preoperatively or whether they had risk factors for uterine cancer but were presumably poor candidates for morcellation due to age alone) helps us define the true incidence of “occult” sarcoma or cancer in a population.
These studies are provocative, however, and do inform us that, as women get older, we are apt to see a greater incidence of uterine cancer. We cannot safely assume that a postmenopausal or elderly woman with symptomatic or enlarging fibroids has “presumed benign disease”—it is cancer until proven otherwise, and we need to be looking for it preoperatively. Therefore, we need to be particularly careful with our surgical practices in this population—ie, the basis for the FDA’s recommendation to avoid morcellation in older women. And I agree with the FDA that open electromechanical morcellation generally is contraindicated in postmenopausal women. However, we need better data from large prospective studies to inform our understanding of the true incidence of undiagnosed or “occult” uterine sarcoma in women undergoing surgery for presumed benign disease. These future studies are likely to demonstrate what we already know—that in young, well-screened, well-selected candidates for minimally invasive hysterectomy or myomectomy, the risk of occult cancer is going to be exceptionally low.
OBG Management: Which is greater—the risks or benefits of tissue extraction?
Dr. Fader: Assessment of risks and benefits in medicine has everything to do with the intervention in question as applied to an individual patient. At the end of the day, there are risks and benefits to every medical or surgical treatment offered to patients in every medical and surgical discipline. But the risk of an occult uterine sarcoma is extremely low in a woman of reproductive age who has been properly selected and comprehensively evaluated for minimally invasive surgery and tissue extraction prior to surgery. And this small—though not negligible—risk must be weighed against the much higher risk of harm that may be incurred with an open abdominal procedure, compared with minimally invasive surgery.
However, in many elderly women, the risks of tissue extraction with an electromechanical morcellator may outweigh the benefits. Even so, there are exceptions in which tissue extraction may be acceptable in postmenopausal women (ie, using alternative tissue-extraction methods in those undergoing minimally invasive supracervical hysterectomy and sacral colpopexy for pelvic organ prolapse).
Few of the data published on the risks of morcellation are of very high quality in terms of scientific rigor or methodology. The best thing we can do as a gynecologic surgical community is conduct sound quality-improvement (QI) programs, disseminate our QI results, publish our data, establish guidelines for best practices in uterine tissue extraction, and collaborate readily to increase the scholarly output on this issue so that national societies and government regulatory agencies have better-quality data to inform future policy on this issue.
A case-based approach
OBG Management: How would you approach tissue extraction in the following case?
CASE: A desire for myomectomy
A 35-year-old woman (G1P1) who delivered by cesarean has an 8-cm symptomatic intramural fibroid. She has regular heavy periods that have led to anemia (hemoglobin level = 10 mg/dL). Her medical history is negative for malignancy, pelvic radiation, or tamoxifen use, and she wants to preserve her fertility. Magnetic resonance imaging (MRI) confirms an 8-cm fibroid. Endometrial biopsy results are negative.
Dr. Fader: At Johns Hopkins, as at many other centers, we use well-defined criteria to determine whether a minimally invasive approach (and tissue extraction) might be appropriate. We also individualize treatment and surgical decision-making on a case-by-case basis. Any candidate for minimally invasive surgery and tissue extraction for uterine fibroids must undergo a thorough preoperative assessment.
Johns Hopkins preoperative assessment criteria include:
- endometrial biopsy
- imaging (often MRI)
- a detailed history and physical, with a comprehensive review of risk factors for malignancy, including family and genetic history or a personal history of malignancy, pelvic radiation, tamoxifen use, or BRCA or hereditary nonpolyposis colorectal cancer (HNPCC) deleterious mutation carrier status, among other things.
- In addition, all cases are discussed at a peer-reviewed, preoperative conference to ensure that a thorough work-up has been conducted and to verify the patient’s candidacy for a minimally invasive procedure with tissue extraction. As the FDA recommends, we conduct an enhanced informed consent process and counsel patients being considered for tissue extraction about the risk of occult sarcoma.
Our top priority is patient safety, so until more data are available, we no longer perform open electromechanical morcellation. We perform all tissue extraction within a containment system and under institutional review board protocol. We primarily perform tissue extraction via scalpel morcellation.
OBG Management: How do the patient’s wishes factor into the decision to perform minimally invasive surgery with morcellation?
Dr. Fader: Our patients make their own decisions regarding surgical approach and procedure after undergoing extensive counseling about the risks and benefits of the proposed procedures. I certainly would offer a patient like the one described in this case the opportunity for a minimally invasive approach (if, after thorough preoperative evaluation, she were deemed to be at very low risk for uterine malignancy). In my experience, most women opt for the minimally invasive approach in this setting; however, if a patient declines minimally invasive surgery, I respect her decision.
Tissue extraction in perimenopause
CASE: A desire for myomectomy at age 48
OBG Management: How would you approach the same case if the patient were a 48-year-old perimenopausal woman?
Dr. Fader: In perimenopausal women, we are more selective about performing morcellation, given the recent FDA safety statement, and because the incidence of occult cancer starts to increase slightly in this patient cohort (although it doesn’t precipitously increase until well into the postmenopausal period).
In addition, myomectomy may have less value in a 48-year-old, given the lower likelihood of achieving successful fertility, although there are exceptions. US cancer statistics and studies on morcellation demonstrate that the vast majority of women in their 30s and 40s have an extremely low risk for sarcomas and other uterine malignancies.2
In select cases in which a woman has undergone a comprehensive preoperative work-up, has a stable-appearing fibroid(s), and is well-educated and counseled about the pros and cons of morcellation, we would consider performing a procedure with contained tissue extraction. As a general matter, however, I would be more inclined to offer a 48-year-old in this situation a uterine artery embolization or minimally invasive hysterectomy than a myomectomy procedure, especially given the recent study by Wright and colleagues demonstrating the significantly increased risks of uterine cancer at myomectomy surgery for a woman in her late 40s or early 50s.18
Preoperative assessment should be comprehensive
OBG Management: What preoperative evaluation do you perform when tissue extraction, including morcellation, is an issue?
Dr. Fader: It is the policy at Johns Hopkins that all women being considered for minimally invasive surgery and tissue extraction must undergo a rigorous preoperative work-up that includes:
- a comprehensive history and physical (to exclude malignancy and risk for occult malignancy)
- an endometrial evaluation (most commonly, an endometrial biopsy)
- uterine imaging (with longitudinal evaluation of imaging findings if performed previously)
- discussion of each patient case at peer-reviewed, preoperative department conferences.
We have separate conferences for the gynecology and gynecologic oncology services and have employed this practice for many years at Hopkins. We are also studying the role of serum lactate dehydrogenase (LDH) isoenzyme levels in stratifying women with uterine fibroids versus cancer/sarcoma.19
OBG Management: Which patients would you exclude from the electromechanical morcellation option?
Dr. Fader: As the American College of Obstetricians and Gynecologists, the AAGL, the Society of Gynecologic Oncology, and the Society of Gynecologic Surgeons appear to agree:
- When utilized in select patients of reproductive age, minimally invasive surgery and morcellation are beneficial.
- Morcellation should categorically not be performed in any woman who has a known or suspected uterine (or other gynecologic) malignancy.2,3
At our institution, we have significantly curtailed the use of electromechanical morcellation at this time and especially do not perform it in women aged 50 or older or in those with confirmed postmenopausal status. We also do not perform electromechanical morcellation in women with a personal history of uterine, cervical, or ovarian preinvasive or invasive cancer, or in women with a strong family history of gynecologic malignancy.
Other populations we exclude are women with:
- a history of mitotically active or atypical fibroids (as determined at previous myomectomy)
- known BRCA or HNPCC deleterious mutation, hereditary leiomyomatosis and renal cell carcinoma (HLRCC) syndrome, hereditary childhood retinoblastoma, or other genetic predisposition to uterine or ovarian cancer
- a history of pelvic radiation
- a history of tamoxifen use.
Counseling the patient
OBG Management: If tissue extraction is an issue, and morcellation will be necessary for a minimally invasive approach, how do you counsel the patient?
Dr. Fader: We have an informed consent protocol we use at Hopkins in this regard. We speak extensively to our patients about the fact that every procedure or intervention performed in medicine carries a benefit/risk ratio. We inform patients of the FDA morcellation safety statement—that their fibroid or fibroids may contain unexpected cancerous tissue and that laparoscopic electromechanical morcellation may spread the cancer and possibly worsen their prognosis. We also explain that, while the FDA quotes a risk of approximately 1 in 350 for occult sarcoma, that this data review was somewhat limited in scope and included postmenopausal women in the estimates. Based on the best available published systematic reviews and internal Hopkins data, we believe the risk of morcellating an occult uterine malignancy in a woman of reproductive age is more likely on the order of approximately 1 in 1,700 to 1 in 8,333.
We discuss our institution’s approach to tissue extraction (ie, that we no longer perform open electromechanical morcellation but instead perform contained tissue extraction on an institutional review board protocol). We tell patients that contained tissue-extraction practices are still experimental, although there is published literature preliminarily supporting the safety of the practice. In addition, we discuss the fact that contained tissue extraction has been used for years to safely remove large intra-abdominal specimens from laparoscopic incisions, from adnexal tissue to gallbladder, spleen, kidney, intestinal, and appendiceal specimens.
We also explain that, as physicians, we do our best to ensure that risks are minimized and reasonable in relation to anticipated benefits but that, even when we use the very best diagnostic measures, no test is 100% sensitive or specific to rule out malignancy in this setting (or in any setting, actually).
Finally, we discuss the fact that minimally invasive surgery and tissue extraction practices performed in appropriate patients by skilled surgeons conclusively benefit hundreds of thousands of women each year around the world. That is a narrative that has been somewhat lacking in the recent dialogue about tissue-extraction practices.
OBG Management: What do we know about outcomes when a leiomyosarcoma is inadvertently morcellated?
Dr. Fader: This is considered a “cut-through” procedure, in that a cancerous tumor that is potentially contained to an organ is not removed intact or with clean margins. The morcellation procedure effectively cuts through the occult cancer, which is not desirable. Intact surgical removal of uterine cancers or sarcomas is the mainstay of therapy for these malignancies, based on NCCN guidelines.22
OBG Management: Does a morcellated leiomyosarcoma carry a worse prognosis than an unmorcellated leiomyosarcoma?
Dr. Fader: When it comes to morcellated versus “intact” uterine leiomyosarcoma, we enter a largely data-free zone. We don’t know with certainty whether the outcome is worsened. If we’re being intellectually honest, we must admit the possibility that a morcellated cancer is more likely to be disseminated, rendering it potentially more difficult to treat. However, sarcomas primarily spread by hematogenous dissemination. It is quite possible that even the act of incising an intact fibroid in an open abdominal procedure or performing a supracervical or total hysterectomy without morcellation may still result in hematogenous cancer dissemination. So there is no indication yet that electromechanical morcellation poses a unique and higher risk of cancer upstaging or worse prognosis compared with techniques such as open myomectomy, supracervical hysterectomy, or hysteroscopic myomectomy.
In addition, the prognosis associated with even early-stage uterine leiomyosarcoma is uniformly poor. A published study from Hopkins that included 108 patients with uterine leiomyosarcoma suggests that the recurrence rate and survival of patients with early-stage, “intact” leiomyosarcoma are very poor and comparable to the survival rate documented in the literature for women with morcellated sarcomas.23
The few retrospective studies that exist suggesting worse outcomes with morcellation have limitations that preclude any definitive conclusions.2 These studies are marred by small numbers, heterogeneity in morcellation practices, poor follow-up times, and a lack of detail regarding how patients with morcellated versus unmorcellated sarcomas were treated. Nevertheless, a couple of these small retrospective reports indicate the possibility of worse outcomes in women with morcellated uterine sarcomas, compared with historical controls with intact sarcoma removal.
Is the morcellator at fault—or the user?
OBG Management: Some would argue that even one case of a morcellated uterine sarcoma is too many. How would you respond?
Dr. Fader: There is no doubt that a handful of women each year have been harmed by morcellation practices. Those women deserve our dedication and best efforts to learn how to better treat morcellated sarcomas—and more importantly—how to mitigate the risks associated with morcellation practices and reduce the risk of preventable harm for all women undergoing minimally invasive fibroid procedures. I think the single best thing we can do to mitigate risk is to be more conscientious about selecting our patients for tissue-extraction procedures (ie, strict selection criteria, appropriate preoperative work-ups). If we did this, we likely would reduce the number of oncologic morcellator mishaps by 50% to 80% without changing anything else.
When we closely scrutinize the literature, and when I reflect on the women with morcellated cancers that we have cared for at Hopkins, we observe that some (but not all) “occult” uterine cancers were not that hidden after all and may have been detected preoperatively if an effort had been made.
We have noted a number of patients in this setting who experienced harm not because a specific device was used to cut up their uterus but because they were never appropriate candidates for the procedure in the first place. For instance, I recently cared for an elderly woman with a morcellated uterine cancer who underwent a laparoscopic supracervical hysterectomy without an appropriate preoperative work-up (ie, no endometrial biopsy or imaging) or informed consent about the possibility that she might have cancer. If an elderly woman presents with concerning symptoms related to her uterus (ie, enlargement, bleeding), she must be evaluated and counseled regarding the considerable risk of potential malignancy. Even in the setting of a normal work-up, I don’t believe it is a good idea to perform electromechanical morcellation in higher-risk women, including elderly women. That does not mean that select, well-screened women cannot be considered for alternative tissue-extraction techniques, but the risks and benefits must be carefully weighed in each patient, and informed consent must be obtained.
By continuing to refine the safety features of the electromechanical morcellator devices and choosing patients more carefully for minimally invasive procedures and tissue extraction, we likely will reduce the risk of preventable harm in women undergoing gynecologic surgery.
Can leiomyosarcoma be detected preoperatively?
OBG Management: How can we improve preoperative detection of uterine malignancy, particularly leiomyosarcoma?
Dr. Fader: For starters, we can improve detection of uterine cancers by simply looking for them. Almost all epithelial uterine cancers will be detectable by endometrial sampling. A comprehensive history and physical and uterine imaging also may be helpful. And although they are more difficult to detect than epithelial uterine cancers, it is a myth that sarcomas cannot be diagnosed preoperatively. Investigators from Columbia University retrospectively evaluated the ability to preoperatively detect epithelial uterine cancers and uterine sarcomas on final pathology. In 72 women who were ultimately diagnosed with a sarcoma, preoperative endometrial sampling suggested an invasive tumor in 86% and predicted the correct histology in 64%. In fact, the rate of detection of invasive cancer by preoperative sampling was not statistically different among sarcomas than it was among epithelial uterine cancers, although there was less of a correlation with appropriate histology seen with endometrial biopsy.20
That being said, sarcomas can be missed, especially in younger women who have extremely large, degenerated, or necrotic-appearing fibroids. Improvements in diagnostic testing are desperately needed to help distinguish benign fibroids from sarcomas, as there are no reliable modalities to exclude a sarcoma at this time. MRI appears to be the most useful imaging modality, although it cannot definitively distinguish a fibroid from a sarcoma. However, a fairly constant finding in leiomyosarcomas is the absence of calcifications. Further, some studies also suggest that ill-defined margins are consistent with a sarcoma. Finally, several centers, including our own, are studying novel biologic markers and revisiting the utility of previously described markers such as LDH in the preoperative detection of uterine sarcoma.21
The way forward
OBG Management: You have said, “Keep patients informed and safe but avoid being too reactionary.” Could you expand on this statement?
Dr. Fader: Certainly. There are many examples throughout the history of medicine in which treatments have brought benefit to thousands or millions of individuals but may cause harm in a select few. We know that when controversial medical issues have arisen in the past, the pendulum has swung widely in terms of societal response.
For example, the landmark Women’s Health Initiative had an immediate and adverse impact on hormone replacement therapy (HRT) administration. The increased risk of cardiovascular disease and breast cancer observed with use of long-term combination therapy with conjugated equine estrogen and medroxyprogesterone acetate prompted many US health-care providers to abandon use of HRT—until more contemporary data demonstrated that, in younger, healthier postmenopausal women, these adverse events were very rare and the benefits of HRT outweighed the risks.
Can we avoid stroke, heart attack, and breast cancer in all younger women taking HRT? Of course not. But we counsel women about the benefit/risk ratio of the therapy and advise them that the likelihood of these events is rare.
Similarly, as with the HRT analogy, younger women have lower risks associated with surgery and morcellation, compared with older women, and are more likely to derive benefit from the procedure (after ensuring appropriate candidacy with a comprehensive preoperative evaluation and informed consent).
However, if the expectation is that no cases of harm will ever occur with a surgical device or procedure in order for it to be deemed acceptable and safe to use in practice, then that is simply an impossible standard to uphold. There is no device, medication, or intervention I know of in medicine that is completely risk-free.
OBG Management: Are there other examples of this type of benefit/risk assessment?
Dr. Fader: Yes. For instance, tamoxifen is a nonsteroidal anti-estrogen agent approved by the FDA for adjuvant treatment of breast cancer, treatment of metastatic breast cancer, and reduction of breast cancer incidence in high-risk women. Tamoxifen has effectively reduced breast cancer rates and significantly improved survival in select breast cancer patients. Yet, it is well known that long-term use of tamoxifen is associated with a twofold increased risk of uterine cancer and uterine sarcoma—a likely far more commonly occurring adverse event than an occult, morcellated uterine sarcoma during a minimally invasive gynecologic procedure.
Should the FDA ban or significantly curtail the use of tamoxifen? No, not likely, because the benefits far outweigh the risks in previvors and hormone-positive breast cancer survivors. “Keep patients informed and safe but avoid being too reactionary” means that we must do our due diligence as physicians by comprehensively counseling and obtaining informed consent from our patients before performing medical interventions. We also must closely scrutinize and improve upon practices that may cause harm.
However, what this also means is that while it may be prudent to restrict or limit a surgical practice in select higher-risk populations or modify it in some way to make it safer, we shouldn’t necessarily completely abandon or ban a practice that has benefited hundreds of thousands of patients at low risk of harm until we have objectively reviewed all of the available science and fully understand the implications of a practice change (ie, would the risks of preventable harm be even greater for women if more of them had to undergo open abdominal surgery?). We need continued cool heads and sound scientific reasoning to decide upon health-care policy changes or treatment paradigm shifts.
At the end of the day, however, it is paramount that we mitigate patient harm. The subject of tissue extraction during minimally invasive surgery is a complex and nuanced issue that merits continued study and open-minded and intelligent dialogue between patient stakeholders, clinicians, scientists, industry, ethicists, regulatory agencies, and the press. I think we can all appreciate how humbling and challenging the morcellation issue has been for many of us, especially our patients—particularly the unfortunate women who have been diagnosed with a uterine sarcoma.
Like many of my colleagues, I have been privileged to care for a number of women with uterine leiomyosarcoma. It is a devastating disease, and the prognosis is very poor, whether it is morcellated or removed intact. We are fortunate that the vast majority of women who undergo a minimally invasive procedure for fibroids or other presumed benign indications will not be at risk for an occult malignancy. But what can we do now to continue to offer the benefits of minimally invasive surgery and tissue extraction to women while simultaneously reducing the risk to the select few who will develop a rare uterine cancer?
We can all make a greater effort to more carefully select our patients for minimally invasive surgery and tissue extraction, to limit the performance of open electromechanical morcellation and collaborate in studying the role of refined tissue-extraction techniques and containment systems, to enhance the informed consent process, to develop improved diagnostic tests for preoperative cancer detection, and to conduct higher-quality studies on minimally invasive tissue-extraction techniques for regulatory agencies to review in the near future. It also goes without saying that we need more federal funding to study rare tumors (and gynecologic cancers in general) and develop better sarcoma treatments.
Although there is no medical treatment or surgical procedure that is completely risk-free, interventions such as HRT, tamoxifen, and uterine morcellation—when used in appropriate patients and for appropriate indications—will allow preventable harm to be minimized and make it possible for countless women to continue to derive tremendous benefit.
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
1. US Food and Drug Administration. Laparoscopic uterine power morcellation in hysterectomy and myomectomy: FDA Safety Communication. http://www.fda.gov/MedicalDevices/Safety/AlertsandNotices/ucm393576.htm. Published April 17, 2014. Accessed February 3, 2014.
2. AAGL. Morcellation during uterine tissue extraction. http://www.aagl.org/wp-content/uploads/2014/05/Tissue_Extraction_TFR.pdf. Accessed February 3, 2015.
3. American College of Obstetricians and Gynecologists. Power morcellation and occult malignancy in gynecologic surgery: a special report. http://www.acog.org/Resources-And-Publications/Task-Force-and-Work-Group-Reports/Power-Morcellation-and-Occult-Malignancy-in-Gynecologic-Surgery. Published May 2014. Accessed February 3, 2015.
4. US Food and Drug Administration. Updated Laparoscopic uterine power morcellation in hysterectomy and myomectomy: FDA Safety Communication. http://www.fda.gov/MedicalDevices/Safety/AlertsandNotices/ucm424443.htm. Published November 24, 2014. Accessed February 2, 2015.
5. American Cancer Society. Uterine sarcoma: What is uterine sarcoma? http://www.cancer.org/cancer/uterinesarcoma/detailedguide/uterine-sarcoma-what-is-uterine-sarcoma. Updated January 12, 2015. Accessed February 3, 2015.
6. D’Angelo E, Prat J. Uterine sarcomas: a review. Gynecol Oncol. 2010;116(1):131–139.
7. Chittawar B, Franik S, Pouwer AW, Farquhar C. Minimally invasive surgical techniques versus open myomectomy for uterine fibroids. Cochrane Database Syst Rev. 2014;10:CD004638. doi:10.1002/14651858.CD004638.pub3.
8. Mori KM, Neubauer NL. Minimally invasive surgery in gynecologic oncology. ISRN Obstet Gynecol. 2013; article ID 312982. http://dx.doi.org/10.1155/2013/312982. Accessed February 2, 2015.
9. Li G, Yan X, Shang H, Wang G, Chen L, Han Y. A comparison of laparoscopic radical hysterectomy and pelvic lymphadenectomy and laparotomy in the treatment of Ib IIa cervical cancer. Gynecol Oncol. 2007;105(1):176–180.
10. Walker JL, Piedmonte MR, Spirtos NM, et al. Laparoscopy compared with laparotomy for comprehensive surgical staging of uterine cancer: Gynecologic Oncology Group Study Lap2. J Clin Oncol. 2009;27(32):5331–5336.
11. Ramos A, Fader AN, Long Roche K. Surgical cytoreduction for disseminated benign disease after open power uterine morcellation. Obstet Gynecol. 2014;125(1):99–102.
12. Theben J, Schellong A, Altgassen C, Kelling K, Schneider S, Grobe-Drieling D. Unexpected malignancies after laparoscopic-assisted supracervical hysterectomies (LASH): an analysis of 1,584 LASH cases. Arch Gynecol Obstet. 2013;287(3):455–462.
13. Parker WH, Fu YS, Berek JS. Uterine sarcoma in patients operated on for presumed leiomyoma and rapidly growing leiomyoma. Obstet Gynecol. 1994;83(3):414–418.
14. Seidman MA, Oduyebo T, Muto MG, Crum CP, Nucci MR, Quade BJ. Peritoneal dissemination complicating morcellation of uterine mesenchymal neoplasms. PLoS One. 2012;7(11):e50058.
15. Pritts EA, Parker WH, Brown J, Olive DL. Outcome of occult uterine leiomyosarcoma after surgery for presumed uterine fibroids: a systematic review. J Minim Invasive Gynecol. 2015;22(1):26–33.
16. Wright JD, Tergas AI, Burke WM, et al. Prevalence of uterine pathology in women undergoing minimally invasive hysterectomy employing electric power morcellation. JAMA. 2014;312(12):1253–1255.
17. Wright JD, Tergas AI, Burke WM, et al. Uterine pathology in women undergoing minimally invasive hysterectomy using morcellation. JAMA. 2014;312(12):1253–1255.
18. Wright JD, Tergas AI, Cul R, et al. Use of electric power morcellation and prevalence of underlying cancer in women who undergo myomectomy. JAMA Oncol. 2015; published online February 19, 2015. doi:10.1001/jamaoncol.2014.206.
19. Goto A, Takeuchi S, Sugimura K, Maruo T. Usefulness of Gd-DTPA contrast-enhanced dynamic MRI and serum determination of LDH and its isozymes in the differential diagnosis of leiomyosarcoma from degenerated leiomyoma of the uterus. Int J Gynecol Cancer. 2002; 12:354.
20. Ricci S, Giuntoli RL 2nd, Eisenhauer E, et al. Does adjuvant chemotherapy improve survival for women with early-stage uterine leiomyosarcoma? Gynecol Oncol. 2013;131(3):629–633.
21. Bansal N, Herzog TJ, Burke W, Cohen CJ, Wright JD. The utility of preoperative endometrial sampling for the detection of uterine sarcomas. Gynecol Oncol. 2008;110(1):43–48.
22. Koh WJ, Greer BE, Abu-Rustum NR, et al. Uterine neoplasms, version 1.2014. J Natl Compr Canc Netw. 2014;12(2):248–280.
23. Schwartz LB, Zawin M, Carcangiu ML, Lange R, McCarthy S. Does pelvic magnetic resonance imaging differentiate among the histologic subtypes of uterine leiomyomata? Fertil Steril. 1998;70(3):580–587.
1. US Food and Drug Administration. Laparoscopic uterine power morcellation in hysterectomy and myomectomy: FDA Safety Communication. http://www.fda.gov/MedicalDevices/Safety/AlertsandNotices/ucm393576.htm. Published April 17, 2014. Accessed February 3, 2014.
2. AAGL. Morcellation during uterine tissue extraction. http://www.aagl.org/wp-content/uploads/2014/05/Tissue_Extraction_TFR.pdf. Accessed February 3, 2015.
3. American College of Obstetricians and Gynecologists. Power morcellation and occult malignancy in gynecologic surgery: a special report. http://www.acog.org/Resources-And-Publications/Task-Force-and-Work-Group-Reports/Power-Morcellation-and-Occult-Malignancy-in-Gynecologic-Surgery. Published May 2014. Accessed February 3, 2015.
4. US Food and Drug Administration. Updated Laparoscopic uterine power morcellation in hysterectomy and myomectomy: FDA Safety Communication. http://www.fda.gov/MedicalDevices/Safety/AlertsandNotices/ucm424443.htm. Published November 24, 2014. Accessed February 2, 2015.
5. American Cancer Society. Uterine sarcoma: What is uterine sarcoma? http://www.cancer.org/cancer/uterinesarcoma/detailedguide/uterine-sarcoma-what-is-uterine-sarcoma. Updated January 12, 2015. Accessed February 3, 2015.
6. D’Angelo E, Prat J. Uterine sarcomas: a review. Gynecol Oncol. 2010;116(1):131–139.
7. Chittawar B, Franik S, Pouwer AW, Farquhar C. Minimally invasive surgical techniques versus open myomectomy for uterine fibroids. Cochrane Database Syst Rev. 2014;10:CD004638. doi:10.1002/14651858.CD004638.pub3.
8. Mori KM, Neubauer NL. Minimally invasive surgery in gynecologic oncology. ISRN Obstet Gynecol. 2013; article ID 312982. http://dx.doi.org/10.1155/2013/312982. Accessed February 2, 2015.
9. Li G, Yan X, Shang H, Wang G, Chen L, Han Y. A comparison of laparoscopic radical hysterectomy and pelvic lymphadenectomy and laparotomy in the treatment of Ib IIa cervical cancer. Gynecol Oncol. 2007;105(1):176–180.
10. Walker JL, Piedmonte MR, Spirtos NM, et al. Laparoscopy compared with laparotomy for comprehensive surgical staging of uterine cancer: Gynecologic Oncology Group Study Lap2. J Clin Oncol. 2009;27(32):5331–5336.
11. Ramos A, Fader AN, Long Roche K. Surgical cytoreduction for disseminated benign disease after open power uterine morcellation. Obstet Gynecol. 2014;125(1):99–102.
12. Theben J, Schellong A, Altgassen C, Kelling K, Schneider S, Grobe-Drieling D. Unexpected malignancies after laparoscopic-assisted supracervical hysterectomies (LASH): an analysis of 1,584 LASH cases. Arch Gynecol Obstet. 2013;287(3):455–462.
13. Parker WH, Fu YS, Berek JS. Uterine sarcoma in patients operated on for presumed leiomyoma and rapidly growing leiomyoma. Obstet Gynecol. 1994;83(3):414–418.
14. Seidman MA, Oduyebo T, Muto MG, Crum CP, Nucci MR, Quade BJ. Peritoneal dissemination complicating morcellation of uterine mesenchymal neoplasms. PLoS One. 2012;7(11):e50058.
15. Pritts EA, Parker WH, Brown J, Olive DL. Outcome of occult uterine leiomyosarcoma after surgery for presumed uterine fibroids: a systematic review. J Minim Invasive Gynecol. 2015;22(1):26–33.
16. Wright JD, Tergas AI, Burke WM, et al. Prevalence of uterine pathology in women undergoing minimally invasive hysterectomy employing electric power morcellation. JAMA. 2014;312(12):1253–1255.
17. Wright JD, Tergas AI, Burke WM, et al. Uterine pathology in women undergoing minimally invasive hysterectomy using morcellation. JAMA. 2014;312(12):1253–1255.
18. Wright JD, Tergas AI, Cul R, et al. Use of electric power morcellation and prevalence of underlying cancer in women who undergo myomectomy. JAMA Oncol. 2015; published online February 19, 2015. doi:10.1001/jamaoncol.2014.206.
19. Goto A, Takeuchi S, Sugimura K, Maruo T. Usefulness of Gd-DTPA contrast-enhanced dynamic MRI and serum determination of LDH and its isozymes in the differential diagnosis of leiomyosarcoma from degenerated leiomyoma of the uterus. Int J Gynecol Cancer. 2002; 12:354.
20. Ricci S, Giuntoli RL 2nd, Eisenhauer E, et al. Does adjuvant chemotherapy improve survival for women with early-stage uterine leiomyosarcoma? Gynecol Oncol. 2013;131(3):629–633.
21. Bansal N, Herzog TJ, Burke W, Cohen CJ, Wright JD. The utility of preoperative endometrial sampling for the detection of uterine sarcomas. Gynecol Oncol. 2008;110(1):43–48.
22. Koh WJ, Greer BE, Abu-Rustum NR, et al. Uterine neoplasms, version 1.2014. J Natl Compr Canc Netw. 2014;12(2):248–280.
23. Schwartz LB, Zawin M, Carcangiu ML, Lange R, McCarthy S. Does pelvic magnetic resonance imaging differentiate among the histologic subtypes of uterine leiomyomata? Fertil Steril. 1998;70(3):580–587.
IN THIS ARTICLE
– A case-based approach
– Comprehensive preoperative assessment
– What is the prognosis when a leiomyosarcoma is morcellated?