User login
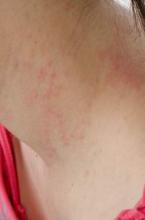
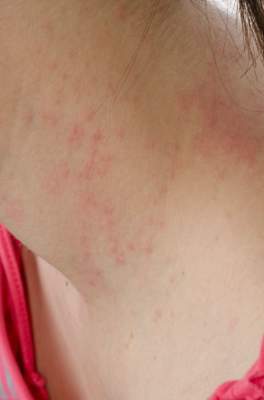
SCOTTSDALE, ARIZ. – Treatment with ustekinumab did not result in significant improvements in atopic dermatitis after 16 weeks, compared with placebo in a phase II study of adults with moderate to severe atopic dermatitis, Dr. Patrick Brunner reported at the annual meeting of the Society for Investigative Dermatology.
However, molecular studies revealed robust modulation of relevant transcriptomic genes after 1 month of ustekinumab, compared with placebo, said Dr. Brunner, who is with the Laboratory for Investigative Dermatology, Rockefeller University, New York. Since the crossover design of this trial and the use of topical corticosteroids by patients probably diluted the differences in clinical outcomes between the intervention and placebo groups, “another study with a different design and dosing is mandated,” he said in an oral presentation at the meeting.
Ustekinumab (Stelara) is a long-acting injectable human interleukin-12 and interleukin-23 antagonist that suppresses Th1, Th17, and Th22 activation. It was approved in 2009 in the United States for moderate to severe plaque psoriasis, and in 2013 for active psoriatic arthritis.
To investigate ustekinumab as therapy for atopic dermatitis, Dr. Brunner and his associates randomly assigned 33 adults aged 18-75 years with refractory atopic dermatitis and baseline scores on the Scoring Atopic Dermatitis (SCORAD) severity scale above 15 to subcutaneous injections of either placebo (17 patients) or ustekinumab (16) at weeks 0, 4, and 16. Ustekinumab was dosed the same way as in psoriasis: 45 mg per injection for patients at or under 100 kg, and 90 mg per injection for heavier patients. At week 16, all patients crossed over to the other treatment for another 16 weeks.
The groups were similar in terms of baseline demographics, intrinsic versus extrinsic IgE status, mean SCORAD, and average levels of IgE and eosinophils. To increase enrollment and retention, both groups were given triamcinolone acetonide 0.025%.
The proportion of SCORAD50 responders (patients with at least a 50% drop from the baseline SCORAD) was greater for ustekinumab than placebo at weeks 12, 16, and 20, but the differences never reached statistical significance, Dr. Brunner reported. At week 16, five (31%) ustekinumab-treated patients had achieved SCORAD50 (the primary endpoint), compared with three (18%) of those on placebo. The ustekinumab SCORAD50 response reached 50% by week 20, but by then patients had crossed over and the two groups began to resemble each other, he said.
“The lessons learned for designing atopic dermatitis trials are to avoid a crossover design with a long-lasting drug, and to keep in mind that even mild background topical steroids can tremendously confound detection of drug effects,” Dr. Brunner commented.
Studies of the atopic dermatitis molecular profile, or transcriptome, revealed similar gene expression levels for both trial arms at baseline, but substantially more gene modulation after 4 weeks of ustekinumab, compared with placebo, he reported. This molecular response involved the Th1, Th17, Th22, but also Th2-related atopic dermatitis genes, and in all cases the differences from placebo were statistically significant (P less than .05). Furthermore, gene modulation became more pronounced through the end of the trial and correlated with clinical response, he added.
The most common adverse effect associated with ustekinumab was respiratory infection, which affected two patients. There were no serious adverse effects reported and none of the patients stopped treatment because of adverse effects.
“Ustekinumab had clear clinical and molecular effects, but clinical outcomes might have been obscured by a profound placebo effect, most likely due to background topical glucocorticosteroids and possibly insufficient dosing for atopic dermatitis,” Dr. Brunner and his associates concluded in a report of their findings, which was published online after the meeting in Experimental Dermatology (Exp Dermatol. 2016 Jun 15. doi: 10.1111/exd.13112).
The study was supported by Janssen, the manufacturer of ustekinumab, and by the National Institutes of Health. Dr. Brunner had no disclosures.
SCOTTSDALE, ARIZ. – Treatment with ustekinumab did not result in significant improvements in atopic dermatitis after 16 weeks, compared with placebo in a phase II study of adults with moderate to severe atopic dermatitis, Dr. Patrick Brunner reported at the annual meeting of the Society for Investigative Dermatology.
However, molecular studies revealed robust modulation of relevant transcriptomic genes after 1 month of ustekinumab, compared with placebo, said Dr. Brunner, who is with the Laboratory for Investigative Dermatology, Rockefeller University, New York. Since the crossover design of this trial and the use of topical corticosteroids by patients probably diluted the differences in clinical outcomes between the intervention and placebo groups, “another study with a different design and dosing is mandated,” he said in an oral presentation at the meeting.
Ustekinumab (Stelara) is a long-acting injectable human interleukin-12 and interleukin-23 antagonist that suppresses Th1, Th17, and Th22 activation. It was approved in 2009 in the United States for moderate to severe plaque psoriasis, and in 2013 for active psoriatic arthritis.
To investigate ustekinumab as therapy for atopic dermatitis, Dr. Brunner and his associates randomly assigned 33 adults aged 18-75 years with refractory atopic dermatitis and baseline scores on the Scoring Atopic Dermatitis (SCORAD) severity scale above 15 to subcutaneous injections of either placebo (17 patients) or ustekinumab (16) at weeks 0, 4, and 16. Ustekinumab was dosed the same way as in psoriasis: 45 mg per injection for patients at or under 100 kg, and 90 mg per injection for heavier patients. At week 16, all patients crossed over to the other treatment for another 16 weeks.
The groups were similar in terms of baseline demographics, intrinsic versus extrinsic IgE status, mean SCORAD, and average levels of IgE and eosinophils. To increase enrollment and retention, both groups were given triamcinolone acetonide 0.025%.
The proportion of SCORAD50 responders (patients with at least a 50% drop from the baseline SCORAD) was greater for ustekinumab than placebo at weeks 12, 16, and 20, but the differences never reached statistical significance, Dr. Brunner reported. At week 16, five (31%) ustekinumab-treated patients had achieved SCORAD50 (the primary endpoint), compared with three (18%) of those on placebo. The ustekinumab SCORAD50 response reached 50% by week 20, but by then patients had crossed over and the two groups began to resemble each other, he said.
“The lessons learned for designing atopic dermatitis trials are to avoid a crossover design with a long-lasting drug, and to keep in mind that even mild background topical steroids can tremendously confound detection of drug effects,” Dr. Brunner commented.
Studies of the atopic dermatitis molecular profile, or transcriptome, revealed similar gene expression levels for both trial arms at baseline, but substantially more gene modulation after 4 weeks of ustekinumab, compared with placebo, he reported. This molecular response involved the Th1, Th17, Th22, but also Th2-related atopic dermatitis genes, and in all cases the differences from placebo were statistically significant (P less than .05). Furthermore, gene modulation became more pronounced through the end of the trial and correlated with clinical response, he added.
The most common adverse effect associated with ustekinumab was respiratory infection, which affected two patients. There were no serious adverse effects reported and none of the patients stopped treatment because of adverse effects.
“Ustekinumab had clear clinical and molecular effects, but clinical outcomes might have been obscured by a profound placebo effect, most likely due to background topical glucocorticosteroids and possibly insufficient dosing for atopic dermatitis,” Dr. Brunner and his associates concluded in a report of their findings, which was published online after the meeting in Experimental Dermatology (Exp Dermatol. 2016 Jun 15. doi: 10.1111/exd.13112).
The study was supported by Janssen, the manufacturer of ustekinumab, and by the National Institutes of Health. Dr. Brunner had no disclosures.
SCOTTSDALE, ARIZ. – Treatment with ustekinumab did not result in significant improvements in atopic dermatitis after 16 weeks, compared with placebo in a phase II study of adults with moderate to severe atopic dermatitis, Dr. Patrick Brunner reported at the annual meeting of the Society for Investigative Dermatology.
However, molecular studies revealed robust modulation of relevant transcriptomic genes after 1 month of ustekinumab, compared with placebo, said Dr. Brunner, who is with the Laboratory for Investigative Dermatology, Rockefeller University, New York. Since the crossover design of this trial and the use of topical corticosteroids by patients probably diluted the differences in clinical outcomes between the intervention and placebo groups, “another study with a different design and dosing is mandated,” he said in an oral presentation at the meeting.
Ustekinumab (Stelara) is a long-acting injectable human interleukin-12 and interleukin-23 antagonist that suppresses Th1, Th17, and Th22 activation. It was approved in 2009 in the United States for moderate to severe plaque psoriasis, and in 2013 for active psoriatic arthritis.
To investigate ustekinumab as therapy for atopic dermatitis, Dr. Brunner and his associates randomly assigned 33 adults aged 18-75 years with refractory atopic dermatitis and baseline scores on the Scoring Atopic Dermatitis (SCORAD) severity scale above 15 to subcutaneous injections of either placebo (17 patients) or ustekinumab (16) at weeks 0, 4, and 16. Ustekinumab was dosed the same way as in psoriasis: 45 mg per injection for patients at or under 100 kg, and 90 mg per injection for heavier patients. At week 16, all patients crossed over to the other treatment for another 16 weeks.
The groups were similar in terms of baseline demographics, intrinsic versus extrinsic IgE status, mean SCORAD, and average levels of IgE and eosinophils. To increase enrollment and retention, both groups were given triamcinolone acetonide 0.025%.
The proportion of SCORAD50 responders (patients with at least a 50% drop from the baseline SCORAD) was greater for ustekinumab than placebo at weeks 12, 16, and 20, but the differences never reached statistical significance, Dr. Brunner reported. At week 16, five (31%) ustekinumab-treated patients had achieved SCORAD50 (the primary endpoint), compared with three (18%) of those on placebo. The ustekinumab SCORAD50 response reached 50% by week 20, but by then patients had crossed over and the two groups began to resemble each other, he said.
“The lessons learned for designing atopic dermatitis trials are to avoid a crossover design with a long-lasting drug, and to keep in mind that even mild background topical steroids can tremendously confound detection of drug effects,” Dr. Brunner commented.
Studies of the atopic dermatitis molecular profile, or transcriptome, revealed similar gene expression levels for both trial arms at baseline, but substantially more gene modulation after 4 weeks of ustekinumab, compared with placebo, he reported. This molecular response involved the Th1, Th17, Th22, but also Th2-related atopic dermatitis genes, and in all cases the differences from placebo were statistically significant (P less than .05). Furthermore, gene modulation became more pronounced through the end of the trial and correlated with clinical response, he added.
The most common adverse effect associated with ustekinumab was respiratory infection, which affected two patients. There were no serious adverse effects reported and none of the patients stopped treatment because of adverse effects.
“Ustekinumab had clear clinical and molecular effects, but clinical outcomes might have been obscured by a profound placebo effect, most likely due to background topical glucocorticosteroids and possibly insufficient dosing for atopic dermatitis,” Dr. Brunner and his associates concluded in a report of their findings, which was published online after the meeting in Experimental Dermatology (Exp Dermatol. 2016 Jun 15. doi: 10.1111/exd.13112).
The study was supported by Janssen, the manufacturer of ustekinumab, and by the National Institutes of Health. Dr. Brunner had no disclosures.
AT THE 2016 SID ANNUAL MEETING
Key clinical point: Ustekinumab missed its primary endpoint in a phase II trial of adults with moderate to severe atopic dermatitis.
Major finding: At 16 weeks, five (31%) ustekinumab patients had achieved SCORAD50, compared with three (19%) placebo patients, a difference that was not statistically significant.
Data source: A randomized, double-blind study evaluated ustekinumab in 33 patients with moderate to severe atopic dermatitis.
Disclosures: The study was supported by Janssen, the maker of ustekinumab (Stelara), and by the National Institutes of Health. Dr. Brunner had no disclosures.