User login
The US Food and Drug Administration (FDA) has granted orphan drug designation to DSP-7888, an investigational cancer peptide vaccine, for the treatment of myelodysplastic syndromes (MDS).
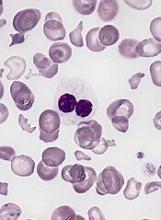
DSP-7888 contains peptides to induce Wilms’ tumor gene 1 (WT1)-specific cytotoxic T lymphocytes and helper T cells, which attack WT1-expressing cancerous cells found in various hematologic and solid tumor malignancies.
DSP-7888 is being developed by Boston Biomedical, Inc.
The first clinical data for DSP-7888, from a phase 1/2 study in patients with MDS who progressed on or after first-line azacitidine treatment, were presented at the 2016 ASH Annual Meeting.
Results were reported in 12 patients—7 with higher-risk MDS and 5 with lower-risk disease.
DSP-7888 was given at doses of 3.5 mg/body (n=6) or 10.5 mg/body (n=6) by intradermal injections every 2 to 4 weeks.
There were no dose-limiting toxicities. The most common adverse event was injection site reactions. Six patients had grade 3 injection site reactions.
There were 5 serious adverse events—3 injection site reactions, 1 case of pyrexia, and 1 case of myocarditis.
Eight patients had stable disease, 2 with hematological improvements.
Cytotoxic T lymphocyte induction was observed in 6 patients, and delayed type hypersensitivity response was observed in 10 patients.
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved. ![]()
The US Food and Drug Administration (FDA) has granted orphan drug designation to DSP-7888, an investigational cancer peptide vaccine, for the treatment of myelodysplastic syndromes (MDS).
DSP-7888 contains peptides to induce Wilms’ tumor gene 1 (WT1)-specific cytotoxic T lymphocytes and helper T cells, which attack WT1-expressing cancerous cells found in various hematologic and solid tumor malignancies.
DSP-7888 is being developed by Boston Biomedical, Inc.
The first clinical data for DSP-7888, from a phase 1/2 study in patients with MDS who progressed on or after first-line azacitidine treatment, were presented at the 2016 ASH Annual Meeting.
Results were reported in 12 patients—7 with higher-risk MDS and 5 with lower-risk disease.
DSP-7888 was given at doses of 3.5 mg/body (n=6) or 10.5 mg/body (n=6) by intradermal injections every 2 to 4 weeks.
There were no dose-limiting toxicities. The most common adverse event was injection site reactions. Six patients had grade 3 injection site reactions.
There were 5 serious adverse events—3 injection site reactions, 1 case of pyrexia, and 1 case of myocarditis.
Eight patients had stable disease, 2 with hematological improvements.
Cytotoxic T lymphocyte induction was observed in 6 patients, and delayed type hypersensitivity response was observed in 10 patients.
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved. ![]()
The US Food and Drug Administration (FDA) has granted orphan drug designation to DSP-7888, an investigational cancer peptide vaccine, for the treatment of myelodysplastic syndromes (MDS).
DSP-7888 contains peptides to induce Wilms’ tumor gene 1 (WT1)-specific cytotoxic T lymphocytes and helper T cells, which attack WT1-expressing cancerous cells found in various hematologic and solid tumor malignancies.
DSP-7888 is being developed by Boston Biomedical, Inc.
The first clinical data for DSP-7888, from a phase 1/2 study in patients with MDS who progressed on or after first-line azacitidine treatment, were presented at the 2016 ASH Annual Meeting.
Results were reported in 12 patients—7 with higher-risk MDS and 5 with lower-risk disease.
DSP-7888 was given at doses of 3.5 mg/body (n=6) or 10.5 mg/body (n=6) by intradermal injections every 2 to 4 weeks.
There were no dose-limiting toxicities. The most common adverse event was injection site reactions. Six patients had grade 3 injection site reactions.
There were 5 serious adverse events—3 injection site reactions, 1 case of pyrexia, and 1 case of myocarditis.
Eight patients had stable disease, 2 with hematological improvements.
Cytotoxic T lymphocyte induction was observed in 6 patients, and delayed type hypersensitivity response was observed in 10 patients.
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved. ![]()