User login
Article Type
Changed
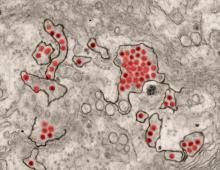
A first-in-human trial of a live, attenuated Zika virus vaccine has begun, according to an announcement by the National Institutes of Health.
Courtesy National Institute of Allergy and Infectious Diseases
The vaccine, developed by scientists at the National Institute of Allergy and Infectious Diseases will be tested in 28 healthy, nonpregnant adults aged 18-50 years at two centers, the Johns Hopkins Bloomberg School of Public Health Center for Immunization Research in Baltimore, and the Vaccine Testing Center at the University of Vermont in Burlington.
The challenge virus in the vaccine is an attenuated genetic chimera consisting of a dengue virus 4 backbone that expresses Zika virus surface proteins designed to elicit an immune response. The virus was previously tested in rhesus macaque monkeys.
Study participants for the phase 1 trial, Evaluation of the Safety and Immunogenicity of the Live Attenuated Zika Vaccine rZIKV/D4Δ30-713 in Flavivirus-Naive Adults, (NCT03611946) will be assessed based on local and general adverse events to the vaccine and peak neutralizing antibody titer to Zika virus as measured up to 90 days after vaccination. The trial is expected to be reach primary completion by Dec. 31, 2018.
If the phase 1 trial is successful, the goal is to integrate the vaccine with a live, attenuated dengue vaccine candidate called TV003, which is designed to elicit antibodies against all four dengue virus serotypes. The TV003 experimental vaccine is currently under evaluation in a phase 3 clinical trial (NCT02406729) underway in Brazil. Both Zika and dengue viruses frequently are endemic in the same regions and a single vaccine against both diseases would be valued. Stephen Whitehead, PhD of NIAID’s Laboratory of Viral Diseases led the efforts to develop both experimental vaccines.
SOURCE: NIH, August 16, 2018. News Release.
Sections
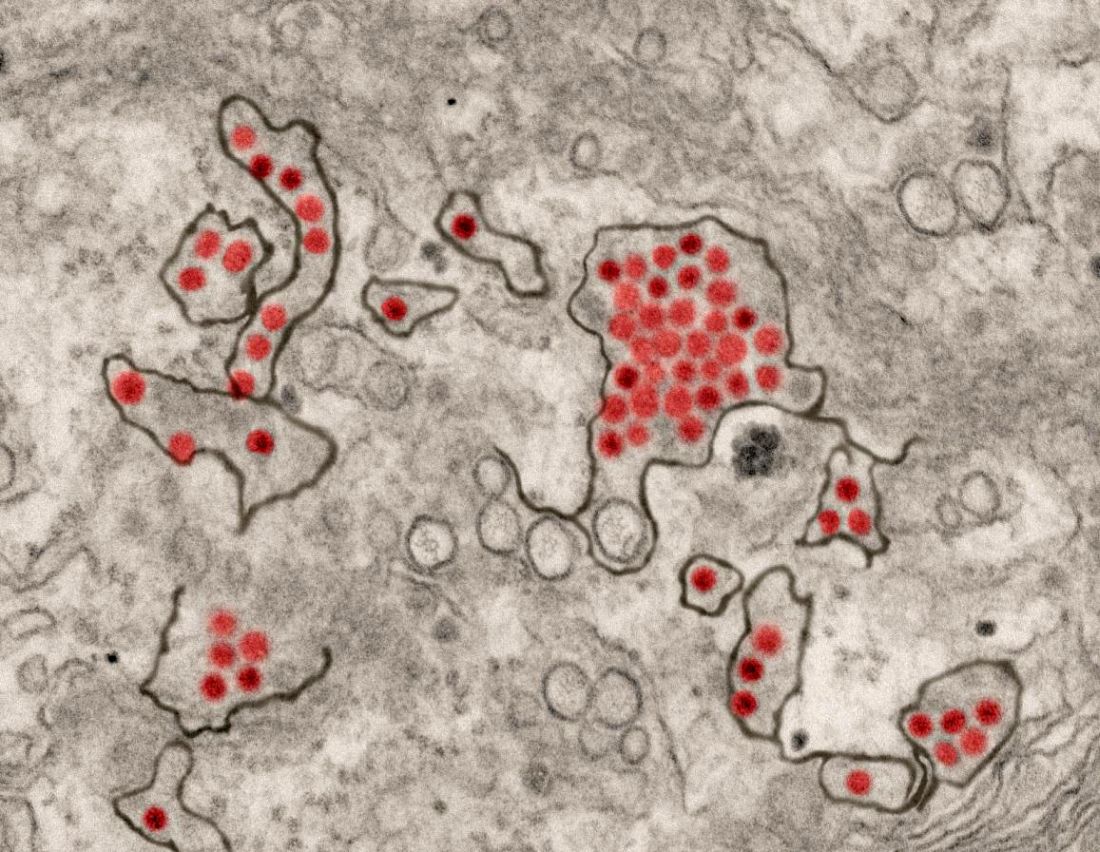
A first-in-human trial of a live, attenuated Zika virus vaccine has begun, according to an announcement by the National Institutes of Health.
Courtesy National Institute of Allergy and Infectious Diseases
The vaccine, developed by scientists at the National Institute of Allergy and Infectious Diseases will be tested in 28 healthy, nonpregnant adults aged 18-50 years at two centers, the Johns Hopkins Bloomberg School of Public Health Center for Immunization Research in Baltimore, and the Vaccine Testing Center at the University of Vermont in Burlington.
The challenge virus in the vaccine is an attenuated genetic chimera consisting of a dengue virus 4 backbone that expresses Zika virus surface proteins designed to elicit an immune response. The virus was previously tested in rhesus macaque monkeys.
Study participants for the phase 1 trial, Evaluation of the Safety and Immunogenicity of the Live Attenuated Zika Vaccine rZIKV/D4Δ30-713 in Flavivirus-Naive Adults, (NCT03611946) will be assessed based on local and general adverse events to the vaccine and peak neutralizing antibody titer to Zika virus as measured up to 90 days after vaccination. The trial is expected to be reach primary completion by Dec. 31, 2018.
If the phase 1 trial is successful, the goal is to integrate the vaccine with a live, attenuated dengue vaccine candidate called TV003, which is designed to elicit antibodies against all four dengue virus serotypes. The TV003 experimental vaccine is currently under evaluation in a phase 3 clinical trial (NCT02406729) underway in Brazil. Both Zika and dengue viruses frequently are endemic in the same regions and a single vaccine against both diseases would be valued. Stephen Whitehead, PhD of NIAID’s Laboratory of Viral Diseases led the efforts to develop both experimental vaccines.
SOURCE: NIH, August 16, 2018. News Release.
A first-in-human trial of a live, attenuated Zika virus vaccine has begun, according to an announcement by the National Institutes of Health.
Courtesy National Institute of Allergy and Infectious Diseases
The vaccine, developed by scientists at the National Institute of Allergy and Infectious Diseases will be tested in 28 healthy, nonpregnant adults aged 18-50 years at two centers, the Johns Hopkins Bloomberg School of Public Health Center for Immunization Research in Baltimore, and the Vaccine Testing Center at the University of Vermont in Burlington.
The challenge virus in the vaccine is an attenuated genetic chimera consisting of a dengue virus 4 backbone that expresses Zika virus surface proteins designed to elicit an immune response. The virus was previously tested in rhesus macaque monkeys.
Study participants for the phase 1 trial, Evaluation of the Safety and Immunogenicity of the Live Attenuated Zika Vaccine rZIKV/D4Δ30-713 in Flavivirus-Naive Adults, (NCT03611946) will be assessed based on local and general adverse events to the vaccine and peak neutralizing antibody titer to Zika virus as measured up to 90 days after vaccination. The trial is expected to be reach primary completion by Dec. 31, 2018.
If the phase 1 trial is successful, the goal is to integrate the vaccine with a live, attenuated dengue vaccine candidate called TV003, which is designed to elicit antibodies against all four dengue virus serotypes. The TV003 experimental vaccine is currently under evaluation in a phase 3 clinical trial (NCT02406729) underway in Brazil. Both Zika and dengue viruses frequently are endemic in the same regions and a single vaccine against both diseases would be valued. Stephen Whitehead, PhD of NIAID’s Laboratory of Viral Diseases led the efforts to develop both experimental vaccines.
SOURCE: NIH, August 16, 2018. News Release.
Article Type
Sections
Disallow All Ads
Content Gating
No Gating (article Unlocked/Free)
Alternative CME
Disqus Comments
Default
Consolidated Pubs: Do Not Show Source Publication Logo
Use ProPublica