User login
First-line Canakinumab Without Steroids Shows Effectiveness for Systemic Juvenile Idiopathic Arthritis
VIENNA — The interleukin-1 receptor antagonist (IL-1RA) canakinumab provided control of systemic juvenile idiopathic arthritis (sJIA) without the use of glucocorticoids for up to a year in most study participants after three monthly injections.
In this study of 20 patients with newly diagnosed sJIA treated off glucocorticoids, fever was controlled after a single injection in all patients, and 16 patients reached the primary outcome of remission after three injections, said Gerd Horneff, MD, PhD, Asklepios Children’s Hospital, Sankt Augustin, Germany.
Results of this open-label study, called CANAKINUMAB FIRST, were presented as late-breaking findings at the European Alliance of Associations for Rheumatology (EULAR) 2024 Annual Meeting.
“Steroid-free, first-line treatment with canakinumab led to sustained responses in most patients, with a considerable number achieving remission,” said Dr. Horneff, adding that the observation in this group is ongoing.
Building on Earlier Data
The efficacy of canakinumab was previously reported in anecdotal experiences and one small patient series published 10 years ago. Dr. Horneff noted that he has offered this drug off label to patients with challenging cases.
The objective was to evaluate canakinumab as a first-line monotherapy administered in the absence of glucocorticoids. The study was open to children aged 2-18 years with active sJIA/juvenile Still disease confirmed with published criteria. All were naive to biologic or nonbiologic disease-modifying antirheumatic drugs as well as steroids.
The median age of the children was 8.4 years. A total of 60% were men. The median disease duration at the time of entry was 1.2 months. Most had fever (95%) and rash (80%) with high levels of inflammatory markers at baseline. The mean number of painful joints was 3.1, and the mean number of systemic manifestations was 2.8. No patient was without any systemic involvement, but four of the patients did not have any painful joints.
At enrollment, patients were scheduled to receive three injections of canakinumab at monthly intervals during an active treatment phase, after which they entered an observation phase lasting 40 weeks. In the event of nonresponse or flares in either phase, they were transitioned to usual care.
Symptoms Resolve After Single Injection
After the first injection, active joint disease and all systemic manifestations resolved in 16 (80%) of the 20 patients. Joint activity and systemic manifestations also remained controlled after the second and third injections in 16 of the 20 patients.
One patient in this series achieved inactive disease after a single injection but developed what appeared to be a treatment-related allergic reaction. He received no further treatment and was excluded from the study, although he is being followed separately.
“According to sJADAS [systemic JIA Disease Activity Score] criteria at month 3, 14 had inactive disease, three had minimal disease activity, and one patient had moderate disease activity,” Dr. Horneff said.
At week 24, or 3 months after the last injection, there was still no joint activity in 16 patients. Systemic manifestations remained controlled in 13 patients, but 1 patient by this point had a flare. Another flare occurred after this point, and other patients have not yet completed the 52-week observation period.
“Of the 10 patients who remained in the study and have completed the 52-week observation period, eight have had a drug-free remission,” Dr. Horneff said.
MAS Event Observed in One Patient
In addition to the allergic skin reaction, which was considered probably related to the study drug, there were three flares, one of which was a macrophage activation syndrome (MAS) event. The MAS occurred 8 weeks after the last injection, but it was managed successfully.
Of 30 infections that developed during the observation period, 18 involved the upper airway. All were treated successfully. There were also two injection-site reactions and one case of cytopenia.
Among the studies planned for follow-up, investigators will examine genomic and gene activation in relation to disease activity and the effect of canakinumab.
Comoderator of the abstract session and chair of the EULAR 2024 Abstract Selection Committee, Christian Dejaco, MD, PhD, a consultant rheumatologist and associate professor at the Medical University of Graz in Graz, Austria, suggested that these are highly encouraging data for a disease that does not currently have any approved therapies. Clearly, larger studies with a longer follow-up period are needed, but he pointed out that phase 3 trials in a rare disease like sJIA are challenging.
Because of the limited number of cases, “it will be difficult to conduct a placebo-controlled trial,” he pointed out. However, he hopes this study will provide the basis for larger studies and sufficient data to lead to an indication for this therapy.
In the meantime, he also believes that these data are likely to support empirical use in a difficult disease, even in advance of formal regulatory approval.
“We heard that canakinumab is already being used off label in JIA, and these data might encourage more of that,” he said.
Dr. Horneff reported financial relationships with AbbVie, Boehringer Ingelheim, Celgene, Chugai, GlaxoSmithKline, Janssen, Merck Sharpe & Dohme, Novartis, Pfizer, Roche, Sanofi, and Sobe. Dr. Dejaco reported no potential conflicts of interest.
A version of this article first appeared on Medscape.com.
VIENNA — The interleukin-1 receptor antagonist (IL-1RA) canakinumab provided control of systemic juvenile idiopathic arthritis (sJIA) without the use of glucocorticoids for up to a year in most study participants after three monthly injections.
In this study of 20 patients with newly diagnosed sJIA treated off glucocorticoids, fever was controlled after a single injection in all patients, and 16 patients reached the primary outcome of remission after three injections, said Gerd Horneff, MD, PhD, Asklepios Children’s Hospital, Sankt Augustin, Germany.
Results of this open-label study, called CANAKINUMAB FIRST, were presented as late-breaking findings at the European Alliance of Associations for Rheumatology (EULAR) 2024 Annual Meeting.
“Steroid-free, first-line treatment with canakinumab led to sustained responses in most patients, with a considerable number achieving remission,” said Dr. Horneff, adding that the observation in this group is ongoing.
Building on Earlier Data
The efficacy of canakinumab was previously reported in anecdotal experiences and one small patient series published 10 years ago. Dr. Horneff noted that he has offered this drug off label to patients with challenging cases.
The objective was to evaluate canakinumab as a first-line monotherapy administered in the absence of glucocorticoids. The study was open to children aged 2-18 years with active sJIA/juvenile Still disease confirmed with published criteria. All were naive to biologic or nonbiologic disease-modifying antirheumatic drugs as well as steroids.
The median age of the children was 8.4 years. A total of 60% were men. The median disease duration at the time of entry was 1.2 months. Most had fever (95%) and rash (80%) with high levels of inflammatory markers at baseline. The mean number of painful joints was 3.1, and the mean number of systemic manifestations was 2.8. No patient was without any systemic involvement, but four of the patients did not have any painful joints.
At enrollment, patients were scheduled to receive three injections of canakinumab at monthly intervals during an active treatment phase, after which they entered an observation phase lasting 40 weeks. In the event of nonresponse or flares in either phase, they were transitioned to usual care.
Symptoms Resolve After Single Injection
After the first injection, active joint disease and all systemic manifestations resolved in 16 (80%) of the 20 patients. Joint activity and systemic manifestations also remained controlled after the second and third injections in 16 of the 20 patients.
One patient in this series achieved inactive disease after a single injection but developed what appeared to be a treatment-related allergic reaction. He received no further treatment and was excluded from the study, although he is being followed separately.
“According to sJADAS [systemic JIA Disease Activity Score] criteria at month 3, 14 had inactive disease, three had minimal disease activity, and one patient had moderate disease activity,” Dr. Horneff said.
At week 24, or 3 months after the last injection, there was still no joint activity in 16 patients. Systemic manifestations remained controlled in 13 patients, but 1 patient by this point had a flare. Another flare occurred after this point, and other patients have not yet completed the 52-week observation period.
“Of the 10 patients who remained in the study and have completed the 52-week observation period, eight have had a drug-free remission,” Dr. Horneff said.
MAS Event Observed in One Patient
In addition to the allergic skin reaction, which was considered probably related to the study drug, there were three flares, one of which was a macrophage activation syndrome (MAS) event. The MAS occurred 8 weeks after the last injection, but it was managed successfully.
Of 30 infections that developed during the observation period, 18 involved the upper airway. All were treated successfully. There were also two injection-site reactions and one case of cytopenia.
Among the studies planned for follow-up, investigators will examine genomic and gene activation in relation to disease activity and the effect of canakinumab.
Comoderator of the abstract session and chair of the EULAR 2024 Abstract Selection Committee, Christian Dejaco, MD, PhD, a consultant rheumatologist and associate professor at the Medical University of Graz in Graz, Austria, suggested that these are highly encouraging data for a disease that does not currently have any approved therapies. Clearly, larger studies with a longer follow-up period are needed, but he pointed out that phase 3 trials in a rare disease like sJIA are challenging.
Because of the limited number of cases, “it will be difficult to conduct a placebo-controlled trial,” he pointed out. However, he hopes this study will provide the basis for larger studies and sufficient data to lead to an indication for this therapy.
In the meantime, he also believes that these data are likely to support empirical use in a difficult disease, even in advance of formal regulatory approval.
“We heard that canakinumab is already being used off label in JIA, and these data might encourage more of that,” he said.
Dr. Horneff reported financial relationships with AbbVie, Boehringer Ingelheim, Celgene, Chugai, GlaxoSmithKline, Janssen, Merck Sharpe & Dohme, Novartis, Pfizer, Roche, Sanofi, and Sobe. Dr. Dejaco reported no potential conflicts of interest.
A version of this article first appeared on Medscape.com.
VIENNA — The interleukin-1 receptor antagonist (IL-1RA) canakinumab provided control of systemic juvenile idiopathic arthritis (sJIA) without the use of glucocorticoids for up to a year in most study participants after three monthly injections.
In this study of 20 patients with newly diagnosed sJIA treated off glucocorticoids, fever was controlled after a single injection in all patients, and 16 patients reached the primary outcome of remission after three injections, said Gerd Horneff, MD, PhD, Asklepios Children’s Hospital, Sankt Augustin, Germany.
Results of this open-label study, called CANAKINUMAB FIRST, were presented as late-breaking findings at the European Alliance of Associations for Rheumatology (EULAR) 2024 Annual Meeting.
“Steroid-free, first-line treatment with canakinumab led to sustained responses in most patients, with a considerable number achieving remission,” said Dr. Horneff, adding that the observation in this group is ongoing.
Building on Earlier Data
The efficacy of canakinumab was previously reported in anecdotal experiences and one small patient series published 10 years ago. Dr. Horneff noted that he has offered this drug off label to patients with challenging cases.
The objective was to evaluate canakinumab as a first-line monotherapy administered in the absence of glucocorticoids. The study was open to children aged 2-18 years with active sJIA/juvenile Still disease confirmed with published criteria. All were naive to biologic or nonbiologic disease-modifying antirheumatic drugs as well as steroids.
The median age of the children was 8.4 years. A total of 60% were men. The median disease duration at the time of entry was 1.2 months. Most had fever (95%) and rash (80%) with high levels of inflammatory markers at baseline. The mean number of painful joints was 3.1, and the mean number of systemic manifestations was 2.8. No patient was without any systemic involvement, but four of the patients did not have any painful joints.
At enrollment, patients were scheduled to receive three injections of canakinumab at monthly intervals during an active treatment phase, after which they entered an observation phase lasting 40 weeks. In the event of nonresponse or flares in either phase, they were transitioned to usual care.
Symptoms Resolve After Single Injection
After the first injection, active joint disease and all systemic manifestations resolved in 16 (80%) of the 20 patients. Joint activity and systemic manifestations also remained controlled after the second and third injections in 16 of the 20 patients.
One patient in this series achieved inactive disease after a single injection but developed what appeared to be a treatment-related allergic reaction. He received no further treatment and was excluded from the study, although he is being followed separately.
“According to sJADAS [systemic JIA Disease Activity Score] criteria at month 3, 14 had inactive disease, three had minimal disease activity, and one patient had moderate disease activity,” Dr. Horneff said.
At week 24, or 3 months after the last injection, there was still no joint activity in 16 patients. Systemic manifestations remained controlled in 13 patients, but 1 patient by this point had a flare. Another flare occurred after this point, and other patients have not yet completed the 52-week observation period.
“Of the 10 patients who remained in the study and have completed the 52-week observation period, eight have had a drug-free remission,” Dr. Horneff said.
MAS Event Observed in One Patient
In addition to the allergic skin reaction, which was considered probably related to the study drug, there were three flares, one of which was a macrophage activation syndrome (MAS) event. The MAS occurred 8 weeks after the last injection, but it was managed successfully.
Of 30 infections that developed during the observation period, 18 involved the upper airway. All were treated successfully. There were also two injection-site reactions and one case of cytopenia.
Among the studies planned for follow-up, investigators will examine genomic and gene activation in relation to disease activity and the effect of canakinumab.
Comoderator of the abstract session and chair of the EULAR 2024 Abstract Selection Committee, Christian Dejaco, MD, PhD, a consultant rheumatologist and associate professor at the Medical University of Graz in Graz, Austria, suggested that these are highly encouraging data for a disease that does not currently have any approved therapies. Clearly, larger studies with a longer follow-up period are needed, but he pointed out that phase 3 trials in a rare disease like sJIA are challenging.
Because of the limited number of cases, “it will be difficult to conduct a placebo-controlled trial,” he pointed out. However, he hopes this study will provide the basis for larger studies and sufficient data to lead to an indication for this therapy.
In the meantime, he also believes that these data are likely to support empirical use in a difficult disease, even in advance of formal regulatory approval.
“We heard that canakinumab is already being used off label in JIA, and these data might encourage more of that,” he said.
Dr. Horneff reported financial relationships with AbbVie, Boehringer Ingelheim, Celgene, Chugai, GlaxoSmithKline, Janssen, Merck Sharpe & Dohme, Novartis, Pfizer, Roche, Sanofi, and Sobe. Dr. Dejaco reported no potential conflicts of interest.
A version of this article first appeared on Medscape.com.
FROM EULAR 2024
‘Critical Gaps’ Seen in Managing Moms’ Postpartum BP
TOPLINE:
Over 80% of women with new-onset hypertensive disorders during pregnancy experienced persistent hypertension in the 6 weeks after delivery.
METHODOLOGY:
- Researchers analyzed data from 2705 women in the University of Pittsburgh Medical Center system who developed new-onset hypertensive disorders during pregnancy and participated in a remote blood pressure (BP) monitoring program after discharge from the hospital.
- Nurses showed patients how to monitor their pressure at home, and patients had access to a call center that focused on BP management.
TAKEAWAY:
- Persistent hypertension postpartum — defined as an at-home BP measurement of 140/90 mmHg or greater or treatment with an antihypertensive medication — occurred in 81.8% of the participants.
- A total of 14.1% developed severe hypertension (BP of 160/110 mmHg or greater); 22.6% started an antihypertensive medication after discharge.
- Hospital readmission occurred for 13.4% of the women with severe hypertension, 4% of the women with less serious hypertension, and 2.7% of those who did not have persistent high BP.
IN PRACTICE:
Many of the patients had met criteria to initiate antihypertensive treatment during the delivery admission based on guidance from the American College of Cardiology/American Heart Association (67.9%) and the American College of Obstetricians and Gynecologists (38.7%), “yet only 23.5% were discharged with antihypertensive medications,” Sadiya S. Khan, MD, MSc, of Northwestern University Feinberg School of Medicine, in Chicago, wrote in an editor’s note accompanying the study. “These data highlight several critical gaps in evidence-based recommendations for the monitoring and management of BP following a pregnancy complicated by” hypertensive disorders of pregnancy.
SOURCE:
The study was led by Alisse Hauspurg, MD, MS, of Magee-Womens Research Institute in Pittsburgh, and appeared online in JAMA Cardiology.
LIMITATIONS:
The study was limited to data from one center, and the researchers relied on self-reported BP measurements.
DISCLOSURES:
The study was supported by the National Institutes of Health and the American Heart Association. A coauthor disclosed consulting for Organon and being a cofounder of Naima Health.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Over 80% of women with new-onset hypertensive disorders during pregnancy experienced persistent hypertension in the 6 weeks after delivery.
METHODOLOGY:
- Researchers analyzed data from 2705 women in the University of Pittsburgh Medical Center system who developed new-onset hypertensive disorders during pregnancy and participated in a remote blood pressure (BP) monitoring program after discharge from the hospital.
- Nurses showed patients how to monitor their pressure at home, and patients had access to a call center that focused on BP management.
TAKEAWAY:
- Persistent hypertension postpartum — defined as an at-home BP measurement of 140/90 mmHg or greater or treatment with an antihypertensive medication — occurred in 81.8% of the participants.
- A total of 14.1% developed severe hypertension (BP of 160/110 mmHg or greater); 22.6% started an antihypertensive medication after discharge.
- Hospital readmission occurred for 13.4% of the women with severe hypertension, 4% of the women with less serious hypertension, and 2.7% of those who did not have persistent high BP.
IN PRACTICE:
Many of the patients had met criteria to initiate antihypertensive treatment during the delivery admission based on guidance from the American College of Cardiology/American Heart Association (67.9%) and the American College of Obstetricians and Gynecologists (38.7%), “yet only 23.5% were discharged with antihypertensive medications,” Sadiya S. Khan, MD, MSc, of Northwestern University Feinberg School of Medicine, in Chicago, wrote in an editor’s note accompanying the study. “These data highlight several critical gaps in evidence-based recommendations for the monitoring and management of BP following a pregnancy complicated by” hypertensive disorders of pregnancy.
SOURCE:
The study was led by Alisse Hauspurg, MD, MS, of Magee-Womens Research Institute in Pittsburgh, and appeared online in JAMA Cardiology.
LIMITATIONS:
The study was limited to data from one center, and the researchers relied on self-reported BP measurements.
DISCLOSURES:
The study was supported by the National Institutes of Health and the American Heart Association. A coauthor disclosed consulting for Organon and being a cofounder of Naima Health.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Over 80% of women with new-onset hypertensive disorders during pregnancy experienced persistent hypertension in the 6 weeks after delivery.
METHODOLOGY:
- Researchers analyzed data from 2705 women in the University of Pittsburgh Medical Center system who developed new-onset hypertensive disorders during pregnancy and participated in a remote blood pressure (BP) monitoring program after discharge from the hospital.
- Nurses showed patients how to monitor their pressure at home, and patients had access to a call center that focused on BP management.
TAKEAWAY:
- Persistent hypertension postpartum — defined as an at-home BP measurement of 140/90 mmHg or greater or treatment with an antihypertensive medication — occurred in 81.8% of the participants.
- A total of 14.1% developed severe hypertension (BP of 160/110 mmHg or greater); 22.6% started an antihypertensive medication after discharge.
- Hospital readmission occurred for 13.4% of the women with severe hypertension, 4% of the women with less serious hypertension, and 2.7% of those who did not have persistent high BP.
IN PRACTICE:
Many of the patients had met criteria to initiate antihypertensive treatment during the delivery admission based on guidance from the American College of Cardiology/American Heart Association (67.9%) and the American College of Obstetricians and Gynecologists (38.7%), “yet only 23.5% were discharged with antihypertensive medications,” Sadiya S. Khan, MD, MSc, of Northwestern University Feinberg School of Medicine, in Chicago, wrote in an editor’s note accompanying the study. “These data highlight several critical gaps in evidence-based recommendations for the monitoring and management of BP following a pregnancy complicated by” hypertensive disorders of pregnancy.
SOURCE:
The study was led by Alisse Hauspurg, MD, MS, of Magee-Womens Research Institute in Pittsburgh, and appeared online in JAMA Cardiology.
LIMITATIONS:
The study was limited to data from one center, and the researchers relied on self-reported BP measurements.
DISCLOSURES:
The study was supported by the National Institutes of Health and the American Heart Association. A coauthor disclosed consulting for Organon and being a cofounder of Naima Health.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Which Surgery for Vaginal Vault Prolapse? No Clear Winner
TOPLINE:
Various surgical approaches to treat vaginal vault prolapse may be similarly safe and effective and can produce high rates of patient satisfaction.
METHODOLOGY:
- A randomized clinical trial at nine sites in the United States included 360 women with vaginal vault prolapse after hysterectomy (average age, 66 years).
- The women were randomly assigned to undergo native tissue repair (transvaginal repair using the sacrospinous or uterosacral ligament), sacrocolpopexy (mesh repair placed abdominally via open or minimally invasive surgery), or transvaginal mesh repair.
TAKEAWAY:
- At 36 months, a composite measure of treatment failure — based on the need for retreatment, the presence of symptoms, or prolapse beyond the hymen — had occurred in 28% of the women who received sacrocolpopexy, 29% who received transvaginal mesh, and 43% who underwent native tissue repair.
- Sacrocolpopexy was superior to native tissue repair for treatment success (adjusted hazard ratio, 0.57; P = .01), and transvaginal mesh was noninferior to sacrocolpopexy, the researchers found.
- All of the surgical approaches were associated with high rates of treatment satisfaction and improved quality of life and sexual function.
- Adverse events and mesh complications were uncommon.
IN PRACTICE:
“All approaches were associated with high treatment satisfaction; improved symptoms, quality of life, and sexual function; and low rates of regret,” the authors of the study wrote. “As such, clinicians counseling patients with prolapse can discuss the ramifications of each approach and engage in shared, individualized decision-making.”
SOURCE:
The study was led by Shawn A. Menefee, MD, Kaiser Permanente San Diego in San Diego, California. It was published online in JAMA Surgery.
LIMITATIONS:
The US Food and Drug Administration in April 2019 banned transvaginal mesh for pelvic organ prolapse because of concerns about complications such as exposure and erosion. Five trial participants who had been assigned to receive transvaginal mesh but had not yet received it at that time were rerandomized to one of the other surgical approaches.
DISCLOSURES:
The study was supported by grants from the Eunice Kennedy Shriver National Institute of Child Health and Human Development and the National Institutes of Health Office of Research on Women’s Health. Researchers disclosed consulting for companies that market medical devices.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Various surgical approaches to treat vaginal vault prolapse may be similarly safe and effective and can produce high rates of patient satisfaction.
METHODOLOGY:
- A randomized clinical trial at nine sites in the United States included 360 women with vaginal vault prolapse after hysterectomy (average age, 66 years).
- The women were randomly assigned to undergo native tissue repair (transvaginal repair using the sacrospinous or uterosacral ligament), sacrocolpopexy (mesh repair placed abdominally via open or minimally invasive surgery), or transvaginal mesh repair.
TAKEAWAY:
- At 36 months, a composite measure of treatment failure — based on the need for retreatment, the presence of symptoms, or prolapse beyond the hymen — had occurred in 28% of the women who received sacrocolpopexy, 29% who received transvaginal mesh, and 43% who underwent native tissue repair.
- Sacrocolpopexy was superior to native tissue repair for treatment success (adjusted hazard ratio, 0.57; P = .01), and transvaginal mesh was noninferior to sacrocolpopexy, the researchers found.
- All of the surgical approaches were associated with high rates of treatment satisfaction and improved quality of life and sexual function.
- Adverse events and mesh complications were uncommon.
IN PRACTICE:
“All approaches were associated with high treatment satisfaction; improved symptoms, quality of life, and sexual function; and low rates of regret,” the authors of the study wrote. “As such, clinicians counseling patients with prolapse can discuss the ramifications of each approach and engage in shared, individualized decision-making.”
SOURCE:
The study was led by Shawn A. Menefee, MD, Kaiser Permanente San Diego in San Diego, California. It was published online in JAMA Surgery.
LIMITATIONS:
The US Food and Drug Administration in April 2019 banned transvaginal mesh for pelvic organ prolapse because of concerns about complications such as exposure and erosion. Five trial participants who had been assigned to receive transvaginal mesh but had not yet received it at that time were rerandomized to one of the other surgical approaches.
DISCLOSURES:
The study was supported by grants from the Eunice Kennedy Shriver National Institute of Child Health and Human Development and the National Institutes of Health Office of Research on Women’s Health. Researchers disclosed consulting for companies that market medical devices.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Various surgical approaches to treat vaginal vault prolapse may be similarly safe and effective and can produce high rates of patient satisfaction.
METHODOLOGY:
- A randomized clinical trial at nine sites in the United States included 360 women with vaginal vault prolapse after hysterectomy (average age, 66 years).
- The women were randomly assigned to undergo native tissue repair (transvaginal repair using the sacrospinous or uterosacral ligament), sacrocolpopexy (mesh repair placed abdominally via open or minimally invasive surgery), or transvaginal mesh repair.
TAKEAWAY:
- At 36 months, a composite measure of treatment failure — based on the need for retreatment, the presence of symptoms, or prolapse beyond the hymen — had occurred in 28% of the women who received sacrocolpopexy, 29% who received transvaginal mesh, and 43% who underwent native tissue repair.
- Sacrocolpopexy was superior to native tissue repair for treatment success (adjusted hazard ratio, 0.57; P = .01), and transvaginal mesh was noninferior to sacrocolpopexy, the researchers found.
- All of the surgical approaches were associated with high rates of treatment satisfaction and improved quality of life and sexual function.
- Adverse events and mesh complications were uncommon.
IN PRACTICE:
“All approaches were associated with high treatment satisfaction; improved symptoms, quality of life, and sexual function; and low rates of regret,” the authors of the study wrote. “As such, clinicians counseling patients with prolapse can discuss the ramifications of each approach and engage in shared, individualized decision-making.”
SOURCE:
The study was led by Shawn A. Menefee, MD, Kaiser Permanente San Diego in San Diego, California. It was published online in JAMA Surgery.
LIMITATIONS:
The US Food and Drug Administration in April 2019 banned transvaginal mesh for pelvic organ prolapse because of concerns about complications such as exposure and erosion. Five trial participants who had been assigned to receive transvaginal mesh but had not yet received it at that time were rerandomized to one of the other surgical approaches.
DISCLOSURES:
The study was supported by grants from the Eunice Kennedy Shriver National Institute of Child Health and Human Development and the National Institutes of Health Office of Research on Women’s Health. Researchers disclosed consulting for companies that market medical devices.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Liver Resection Beats Out Alternatives in Early Multinodular HCC
TOPLINE:
METHODOLOGY:
- The presentation of HCC is often multinodular — meaning patients have two or three nodules measuring ≤ 3 cm each. Although liver resection is considered the gold standard curative treatment for early-stage disease, experts debate its efficacy in multinodular HCC, researchers explained.
- Using two large Italian registries with data from multiple centers, researchers compared the efficacy of liver resection, percutaneous radiofrequency ablation, and TACE in 720 patients with early multinodular HCC. Overall, 296 patients underwent liver resection, 240 had percutaneous radiofrequency ablation, and 184 underwent TACE.
- To avoid crossovers between groups, the researchers considered liver resection, percutaneous radiofrequency ablation, and TACE the main treatments in each population in a hierarchical order. That meant, in the liver resection group, researchers excluded patients undergoing a superior treatment during the follow-up, such as liver transplant. In the ablation group, patients undergoing surgery to treat HCC recurrences were excluded.
- The primary outcome was overall survival at 1, 3, and 5 years. The researchers used a matching-adjusted indirect comparison (MAIC) to balance data and control for confounding factors between the three treatment groups.
TAKEAWAY:
- After MAIC adjustment, the survival rate at 1 year was slightly lower in the liver resection group — 89% vs 94% in the ablation group and 91% in the TACE group. However, at 3 and 5 years, survival rates were better in the liver resection group — 71% at 3 years and 56% at 5 years vs 65% and 40%, respectively, in the ablation group and 49% and 29%, respectively, in the TACE group.
- Median overall survival was 69 months with liver resection, 54 months with ablation, and 34 months with TACE. Multivariable Cox survival analysis confirmed a significantly higher mortality risk with ablation (hazard ratio [HR], 1.41; P = .01) and TACE (HR, 1.86; P = .001) than with liver resection.
- In competing risk analyses, patients who underwent liver resection had a lower risk for HCC-related death than peers who had ablation (HR, 1.38; P = .07) or TACE (HR, 1.91; P = .006).
- In a subgroup survival analysis of patients with Child-Pugh class B cirrhosis, liver resection provided significantly better overall survival than TACE (HR, 2.79; P = .001) and higher overall survival than ablation (HR, 1.44; P = .21), but these findings were not statistically significant.
IN PRACTICE:
“The main result of the current study is the indisputable superiority” of liver resection over percutaneous radiofrequency ablation and TACE in patients with multinodular HCC, the researchers concluded. “For patients with early multinodular HCC who are ineligible for transplant, LR [liver resection] should be prioritized as the primary therapeutic option,” followed by percutaneous radiofrequency ablation and TACE, when resection is not feasible.
The authors of an invited commentary said the analysis provides “convincing” data that liver resection leads to superior 3- and 5-year survival. “All of our local therapies are getting better. Making each available under different clinical circumstances and combining these when appropriate provides patients with the best chance at cure with the least invasiveness,” the editorialists added.
SOURCE:
The study, with first author Alessandro Vitale, MD, PhD, with the Department of Surgical, Oncological and Gastroenterological Sciences, University of Padova, Padua, Italy, and the accompanying commentary were published online last month in JAMA Surgery.
LIMITATIONS:
Selection bias cannot be ruled out due to potential hidden variables that were not collected in the centers’ databases. Not all patients included in the study were potentially treatable with all three proposed approaches. The study population was derived from Italian centers, which may have limited the generalizability of the results.
DISCLOSURES:
The study reported no specific funding. The authors reported various disclosures during the conduct of the study, including ties to AstraZeneca, AbbVie, Bayer, MSD, Roche, and Eisai. An editorialist reported ties to Medtronic, Theromics, Vergent Bioscience, Imugene, Sovato Health, XDemics, and Imugene.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- The presentation of HCC is often multinodular — meaning patients have two or three nodules measuring ≤ 3 cm each. Although liver resection is considered the gold standard curative treatment for early-stage disease, experts debate its efficacy in multinodular HCC, researchers explained.
- Using two large Italian registries with data from multiple centers, researchers compared the efficacy of liver resection, percutaneous radiofrequency ablation, and TACE in 720 patients with early multinodular HCC. Overall, 296 patients underwent liver resection, 240 had percutaneous radiofrequency ablation, and 184 underwent TACE.
- To avoid crossovers between groups, the researchers considered liver resection, percutaneous radiofrequency ablation, and TACE the main treatments in each population in a hierarchical order. That meant, in the liver resection group, researchers excluded patients undergoing a superior treatment during the follow-up, such as liver transplant. In the ablation group, patients undergoing surgery to treat HCC recurrences were excluded.
- The primary outcome was overall survival at 1, 3, and 5 years. The researchers used a matching-adjusted indirect comparison (MAIC) to balance data and control for confounding factors between the three treatment groups.
TAKEAWAY:
- After MAIC adjustment, the survival rate at 1 year was slightly lower in the liver resection group — 89% vs 94% in the ablation group and 91% in the TACE group. However, at 3 and 5 years, survival rates were better in the liver resection group — 71% at 3 years and 56% at 5 years vs 65% and 40%, respectively, in the ablation group and 49% and 29%, respectively, in the TACE group.
- Median overall survival was 69 months with liver resection, 54 months with ablation, and 34 months with TACE. Multivariable Cox survival analysis confirmed a significantly higher mortality risk with ablation (hazard ratio [HR], 1.41; P = .01) and TACE (HR, 1.86; P = .001) than with liver resection.
- In competing risk analyses, patients who underwent liver resection had a lower risk for HCC-related death than peers who had ablation (HR, 1.38; P = .07) or TACE (HR, 1.91; P = .006).
- In a subgroup survival analysis of patients with Child-Pugh class B cirrhosis, liver resection provided significantly better overall survival than TACE (HR, 2.79; P = .001) and higher overall survival than ablation (HR, 1.44; P = .21), but these findings were not statistically significant.
IN PRACTICE:
“The main result of the current study is the indisputable superiority” of liver resection over percutaneous radiofrequency ablation and TACE in patients with multinodular HCC, the researchers concluded. “For patients with early multinodular HCC who are ineligible for transplant, LR [liver resection] should be prioritized as the primary therapeutic option,” followed by percutaneous radiofrequency ablation and TACE, when resection is not feasible.
The authors of an invited commentary said the analysis provides “convincing” data that liver resection leads to superior 3- and 5-year survival. “All of our local therapies are getting better. Making each available under different clinical circumstances and combining these when appropriate provides patients with the best chance at cure with the least invasiveness,” the editorialists added.
SOURCE:
The study, with first author Alessandro Vitale, MD, PhD, with the Department of Surgical, Oncological and Gastroenterological Sciences, University of Padova, Padua, Italy, and the accompanying commentary were published online last month in JAMA Surgery.
LIMITATIONS:
Selection bias cannot be ruled out due to potential hidden variables that were not collected in the centers’ databases. Not all patients included in the study were potentially treatable with all three proposed approaches. The study population was derived from Italian centers, which may have limited the generalizability of the results.
DISCLOSURES:
The study reported no specific funding. The authors reported various disclosures during the conduct of the study, including ties to AstraZeneca, AbbVie, Bayer, MSD, Roche, and Eisai. An editorialist reported ties to Medtronic, Theromics, Vergent Bioscience, Imugene, Sovato Health, XDemics, and Imugene.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- The presentation of HCC is often multinodular — meaning patients have two or three nodules measuring ≤ 3 cm each. Although liver resection is considered the gold standard curative treatment for early-stage disease, experts debate its efficacy in multinodular HCC, researchers explained.
- Using two large Italian registries with data from multiple centers, researchers compared the efficacy of liver resection, percutaneous radiofrequency ablation, and TACE in 720 patients with early multinodular HCC. Overall, 296 patients underwent liver resection, 240 had percutaneous radiofrequency ablation, and 184 underwent TACE.
- To avoid crossovers between groups, the researchers considered liver resection, percutaneous radiofrequency ablation, and TACE the main treatments in each population in a hierarchical order. That meant, in the liver resection group, researchers excluded patients undergoing a superior treatment during the follow-up, such as liver transplant. In the ablation group, patients undergoing surgery to treat HCC recurrences were excluded.
- The primary outcome was overall survival at 1, 3, and 5 years. The researchers used a matching-adjusted indirect comparison (MAIC) to balance data and control for confounding factors between the three treatment groups.
TAKEAWAY:
- After MAIC adjustment, the survival rate at 1 year was slightly lower in the liver resection group — 89% vs 94% in the ablation group and 91% in the TACE group. However, at 3 and 5 years, survival rates were better in the liver resection group — 71% at 3 years and 56% at 5 years vs 65% and 40%, respectively, in the ablation group and 49% and 29%, respectively, in the TACE group.
- Median overall survival was 69 months with liver resection, 54 months with ablation, and 34 months with TACE. Multivariable Cox survival analysis confirmed a significantly higher mortality risk with ablation (hazard ratio [HR], 1.41; P = .01) and TACE (HR, 1.86; P = .001) than with liver resection.
- In competing risk analyses, patients who underwent liver resection had a lower risk for HCC-related death than peers who had ablation (HR, 1.38; P = .07) or TACE (HR, 1.91; P = .006).
- In a subgroup survival analysis of patients with Child-Pugh class B cirrhosis, liver resection provided significantly better overall survival than TACE (HR, 2.79; P = .001) and higher overall survival than ablation (HR, 1.44; P = .21), but these findings were not statistically significant.
IN PRACTICE:
“The main result of the current study is the indisputable superiority” of liver resection over percutaneous radiofrequency ablation and TACE in patients with multinodular HCC, the researchers concluded. “For patients with early multinodular HCC who are ineligible for transplant, LR [liver resection] should be prioritized as the primary therapeutic option,” followed by percutaneous radiofrequency ablation and TACE, when resection is not feasible.
The authors of an invited commentary said the analysis provides “convincing” data that liver resection leads to superior 3- and 5-year survival. “All of our local therapies are getting better. Making each available under different clinical circumstances and combining these when appropriate provides patients with the best chance at cure with the least invasiveness,” the editorialists added.
SOURCE:
The study, with first author Alessandro Vitale, MD, PhD, with the Department of Surgical, Oncological and Gastroenterological Sciences, University of Padova, Padua, Italy, and the accompanying commentary were published online last month in JAMA Surgery.
LIMITATIONS:
Selection bias cannot be ruled out due to potential hidden variables that were not collected in the centers’ databases. Not all patients included in the study were potentially treatable with all three proposed approaches. The study population was derived from Italian centers, which may have limited the generalizability of the results.
DISCLOSURES:
The study reported no specific funding. The authors reported various disclosures during the conduct of the study, including ties to AstraZeneca, AbbVie, Bayer, MSD, Roche, and Eisai. An editorialist reported ties to Medtronic, Theromics, Vergent Bioscience, Imugene, Sovato Health, XDemics, and Imugene.
A version of this article first appeared on Medscape.com.
Extensive Multidrug-Resistant Dermatophytosis From Trichophyton indotineae
To the Editor:
Historically, commonly available antifungal medications have been effective for treating dermatophytosis (tinea). However, recent tinea outbreaks caused by Trichophyton indotineae—a dermatophyte often resistant to terbinafine and sometimes to other antifungals—have been reported in South Asia, Europe, the Middle East, Southeast Asia, and Australia.1-5
Three confirmed cases of T indotineae dermatophytosis in the United States were reported in 2023 in New York3,6; a fourth confirmed case was reported in 2024 in Pennsylvania.7 Post hoc laboratory testing of fungal isolates in New York in 2022 and 2023 identified an additional 11 cases.8 We present a case of extensive multidrug-resistant tinea caused by T indotineae in a man in California.
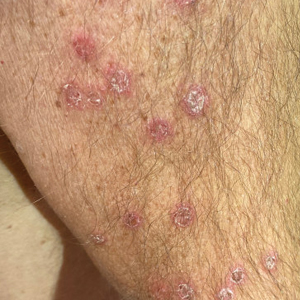
An otherwise healthy 65-year-old man who had traveled to Europe in the past 3 months presented to his primary care physician with a widespread pruritic rash (Figure 1). He was treated with 2 weeks of oral terbinafine 250 mg/d and topical medicines, including clotrimazole cream 1%, fluocinonide ointment 0.05%, and clobetasol ointment 0.05% without improvement. Subsequently, 2 weeks of oral griseofulvin microsize 500 mg/d also proved ineffective. An antibody test was negative for HIV. His hemoglobin A1c was 6.2% (reference range, ≤5.6%). The patient was referred to dermatology.
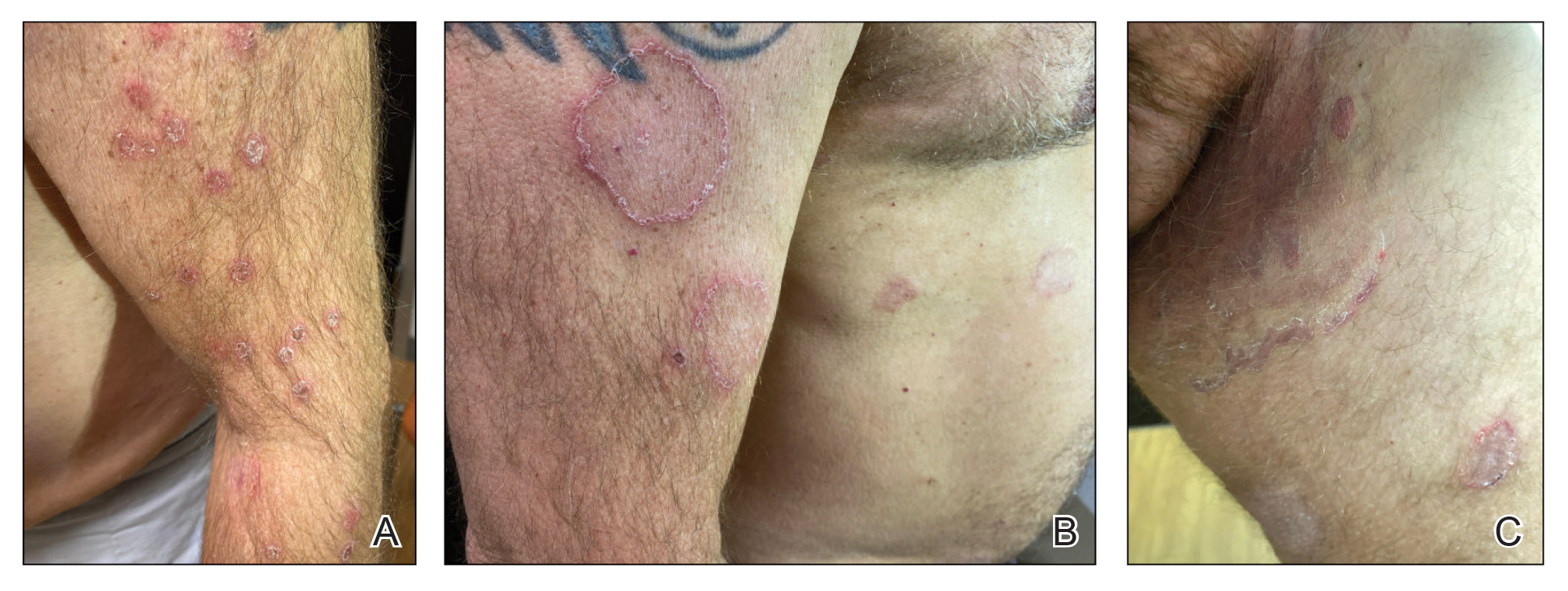
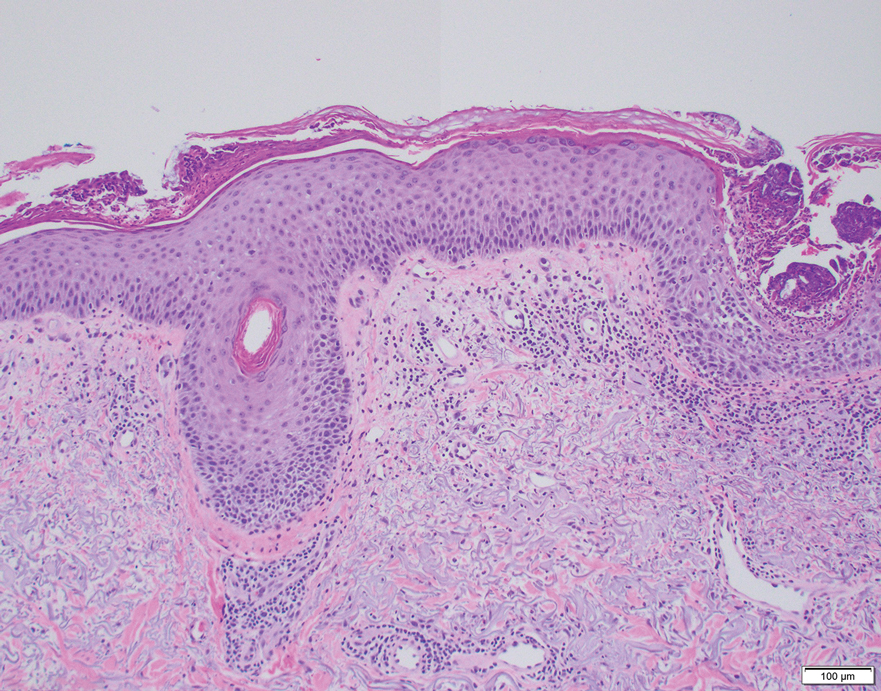
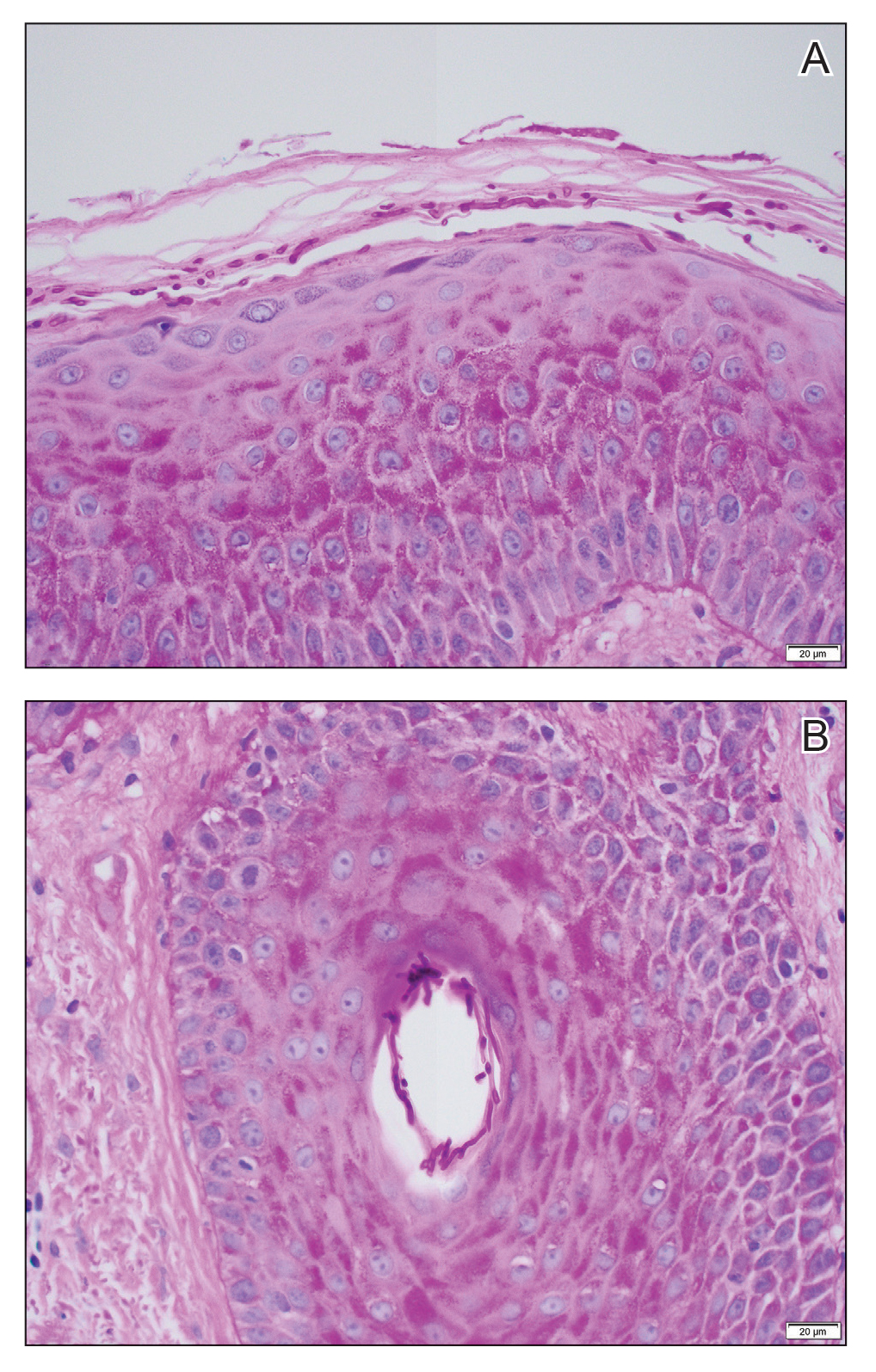
Erythematous plaques—many scaly throughout and some annular with central clearing—were present on the arms, legs, and torso as well as in the groin. Honey crust was present on some plaques on the leg. A potassium hydroxide preparation showed abundant fungal hyphae. Material for fungal and bacterial cultures was collected. The patient was treated again with oral terbinafine 250 mg/d, an oral prednisone taper starting at 60 mg/d for a presumed id reaction, and various oral antihistamines for pruritus; all were ineffective. A bacterial culture showed only mixed skin flora. Oral fluconazole 200 mg/d was prescribed. A skin biopsy specimen showed compact orthokeratosis and parakeratosis of the stratum corneum with few neutrophils and focal pustule formation (Figure 2). Superficial perivascular inflammation, including lymphocytes, histiocytes, and few neutrophils, was present. A periodic acid–Schiff stain showed fungal hyphae in the stratum corneum and a hair follicle (Figure 3). After approximately 2 weeks, mold was identified in the fungal culture. Approximately 2 weeks thereafter, the organism was reported as Trichophyton species.
The rash did not improve; resistance to terbinafine, griseofulvin, and fluconazole was suspected clinically. The fungal isolate was sent to a reference laboratory (University of Texas Health Science Center, San Antonio). Meanwhile, oral itraconazole 200 mg twice daily and ketoconazole cream 2% were prescribed; the rash began to improve. A serum itraconazole trough level obtained 4 days after treatment initiation was 0.5 μg/mL (reference range, ≥0.6 μg/mL). The evening itraconazole dose was increased to 300 mg; a subsequent trough level was 0.8 μg/mL.
Approximately 1 month after the fungal isolate was sent to the reference laboratory, T indotineae was confirmed based on polymerase chain reaction (PCR) testing of internal transcribed spacer region sequences. Minimum inhibitory concentrations (MICs) obtained through antifungal susceptibility testing (AFST) were reported for fluconazole (8 μg/mL), griseofulvin (2 μg/mL), itraconazole (≤0.03 μg/mL), posaconazole (≤0.03 μg/mL), terbinafine (≥2 μg/mL), and voriconazole (0.125 μg/mL).
Approximately 7 weeks after itraconazole and ketoconazole were started, the rash had completely resolved. Nearly 8 months later (at the time this article was written), the rash had not recurred.
We report a unique case of T indotineae in a patient residing in California. Post hoc laboratory testing of dermatophyte isolates sent to the University of Texas reference laboratory identified terbinafine-resistant T indotineae specimens from the United States and Canada dating to 2017; clinical characteristics of patients from whom those isolates were obtained were unavailable.9
Trichophyton indotineae dermatophytosis typically is more extensive, inflamed, and pruritic, as well as likely more contagious, than tinea caused by other dermatophytes.5 Previously called Trichophyton mentagrophytes genotype VIII when first isolated in 2017, the pathogen was renamed T indotineae in 2020 after important genetic differences were discovered between it and other T mentagrophytes species.5 The emergence of T indotineae has been attributed to concomitant use of topical steroids and antifungals,5,10 inappropriate prescribing of antifungals,5 and nonadherence to antifungal treatment.5
Likely risk factors for T indotineae infection include suboptimal hygiene, overcrowded conditions, hot and humid environments, and tight-fitting synthetic clothing.4 Transmission from family members appears common,5 especially when fomites are shared.4 A case reported in Pennsylvania likely was acquired through sexual contact.7 Travel to South Asia has been associated with acquisition of T indotineae infection,3,5-7 though our patient and some others had not traveled there.3,8 It is not clear whether immunosuppression and diabetes mellitus are associated with T indotineae infection.4,5,8 Trichophyton indotineae also can affect animals,11 though zoonotic transmission has not been reported.4
Not all T indotineae isolates are resistant to one or more antifungals; furthermore, antifungal resistance in other dermatophyte species has been reported.5 Terbinafine resistance in T indotineae is conferred by mutations in the gene encoding squalene epoxidase, which helps synthesize ergosterol—a component of the cell membrane in fungi.2,4,5,12 Although clinical cut-points for MIC obtained by AFST are not well established, T indotineae MICs for terbinafine of 0.5 μg/mL or more correlate with resistance.9 Resistance to azoles has been linked to overexpression of transporter genes, which increase azole efflux from cells, as well as to mutations in the gene encoding lanosterol 14α demethylase.4,12,13
Potassium hydroxide preparations and fungal cultures cannot differentiate T indotineae from other dermatophytes that typically cause tinea.5,14 Histopathologic findings in our case were no different than those of non–T indotineae dermatophytes. Only molecular testing using PCR assays to sequence internal transcribed spacer genes can confirm T indotineae infection. However, PCR assays and AFST are not available in many US laboratories.5 Matrix-assisted laser desorption ionization-time of flight (MALDI-TOF) mass spectrometry has shown promise in distinguishing T indotineae from other dermatophytes, though its clinical use is limited and it cannot assess terbinafine sensitivity.15,16 Clinicians in the United States who want to test specimens from cases suspicious for T indotineae infection should contact their local or state health department or the Centers for Disease Control and Prevention for assistance.3,5
Systemic treatment typically is necessary for T indotineae infection.5 Combinations of oral and topical azoles have been used, as well as topical ciclopirox, amorolfine (not available in the United States), and luliconazole.1,5,17-21
Itraconazole has emerged as the treatment of choice for T indotineae tinea, typically at 200 mg/d and often for courses of more than 3 months.5 Testing for serum itraconazole trough levels, as done for our patient, typically is not recommended. Clinicians should counsel patients to take itraconazole with high-fat foods and an acidic beverage to increase bioavailability.5 Potential adverse effects of itraconazole include heart failure and numerous drug-drug interactions.5,22 Patients with T indotineae dermatophytosis should avoid sharing personal belongings and having skin-to-skin contact of affected areas with others.4
Dermatologists who suspect T indotineae infection should work with public health agencies that can assist with testing and undertake infection surveillance, prevention, and control.5,23 Challenges to diagnosing and managing T indotineae infection include lack of awareness among dermatology providers, the need for specialized laboratory testing to confirm infection, lack of established clinical cut-points for MICs from AFST, the need for longer duration of treatment vs what is needed for typical tinea, and potential challenges with insurance coverage for testing and treatment. Empiric treatment with itraconazole should be considered when terbinafine-resistant dermatophytosis is suspected or when terbinafine-resistant T indotineae infection is confirmed.
Acknowledgments—Jeremy Gold, MD; Dallas J. Smith, PharmD; and Shawn Lockhart, PhD, all of the Centers for Disease Control and Prevention, Mycotic Diseases Branch (Atlanta, Georgia), provided helpful comments to the authors in preparing the manuscript of this article.
- Uhrlaß S, Verma SB, Gräser Y, al. Trichophyton indotineae—an emerging pathogen causing recalcitrant dermatophytoses in India and worldwide—a multidimensional perspective. J Fungi (Basel). 2022;8:757. doi:10.3390/jof8070757
- Jabet A, Brun S, Normand A-C, et al. Extensive dermatophytosis caused by terbinafine-resistant Trichophyton indotineae, France. Emerg Infect Dis. 2022;28:229-233. doi:10.3201/eid2801.210883
- Caplan AS, Chaturvedi S, Zhu Y, et al. Notes from the field. First reported U.S. cases of tinea caused by Trichophyton indotineae—New York City, December 2021-March 2023. MMWR Morb Mortal Wkly Rep. 2023;72:536-537. doi:10.15585/mmwr.mm7219a4
- Jabet A, Normand A-C, Brun S, et al. Trichophyton indotineae, from epidemiology to therapeutic. J Mycol Med. 2023;33:101383. doi:10.1016/j.mycmed.2023.101383
- Hill RC, Caplan AS, Elewski B, et al. Expert panel review of skin and hair dermatophytoses in an era of antifungal resistance. Am J Clin Dermatol. 2024;25:359-389. doi:10.1007/s40257-024-00848-1
- Caplan AS, Zakhem GA, Pomeranz MK. Trichophyton mentagrophytes internal transcribed spacer genotype VIII. JAMA Dermatol. 2023;159:1130. doi:10.1001/jamadermatol.2023.2645
- Spivack S, Gold JAW, Lockhart SR, et al. Potential sexual transmission of antifungal-resistant Trichophyton indotineae. Emerg Infect Dis. 2024;30:807-809. doi:10.3201/eid3004.240115
- Caplan AS, Todd GC, Zhu Y, et al. Clinical course, antifungal susceptibility, and genomic sequencing of Trichophyton indotineae. JAMA Dermatol. Published online May 15, 2024. doi:10.1001/jamadermatol.2024.1126
- Cañete-Gibas CF, Mele J, Patterson HP, et al. Terbinafine-resistant dermatophytes and the presence of Trichophyton indotineae in North America. J Clin Microbiol. 2023;61:e0056223. doi:10.1128/jcm.00562-23
- Gupta AK, Venkataraman M, Hall DC, et al. The emergence of Trichophyton indotineae: implications for clinical practice. Int J Dermatol. 2023;62:857-861.
- Oladzad V, Nasrollahi Omran A, Haghani I, et al. Multi-drug resistance Trichophyton indotineae in a stray dog. Res Vet Sci. 2024;166:105105. doi:10.1016/j.rvsc.2023.105105
- Martinez-Rossi NM, Bitencourt TA, Peres NTA, et al. Dermatophyte resistance to antifungal drugs: mechanisms and prospectus. Front Microbiol. 2018;9:1108. doi:10.3389/fmicb.2018.01108
- Sacheli R, Hayette MP. Antifungal resistance in dermatophytes: genetic considerations, clinical presentations and alternative therapies. J Fungi (Basel). 2021;711:983. doi:10.3390/jof7110983
- Gupta AK, Cooper EA. Dermatophytosis (tinea) and other superficial fungal infections. In: Hospenthal DR, Rinaldi MG, eds. Diagnosis and Treatment of Human Mycoses. Humana Press; 2008:355-381.
- Normand A-C, Moreno-Sabater A, Jabet A, et al. MALDI-TOF mass spectrometry online identification of Trichophyton indotineae using the MSI-2 application. J Fungi (Basel). 2022;8:1103. doi:10.3390/jof8101103
- De Paepe R, Normand A-C, Uhrlaß S, et al. Resistance profile, terbinafine resistance screening and MALDI-TOF MS identification of the emerging pathogen Trichophyton indotineae. Mycopathologia. 2024;189:29. doi:10.1007/s11046-024-00835-4
- Rajagopalan M, Inamadar A, Mittal A, et al. Expert consensus on the management of dermatophytosis in India (ECTODERM India). BMC Dermatol. 2018;18:6. doi:10.1186/s12895-018-0073-1
- Verma SB, Panda S, Nenoff P, et al. The unprecedented epidemic-like scenario of dermatophytosis in India: III. Antifungal resistance and treatment options. Indian J Dermatol Venereol Leprol. 2021;87:468-482. doi:10.25259/IJDVL_303_20
- Shaw D, Singh S, Dogra S, et al. MIC and upper limit of wild-type distribution for 13 antifungal agents against a Trichophyton mentagrophytes–Trichophyton interdigitale complex of Indian origin. Antimicrob Agents Chemother. 2020;64:E01964-19. doi:10.1128/AAC.01964-19
- Burmester A, Hipler U-C, Uhrlaß S, et al. Indian Trichophyton mentagrophytes squalene epoxidase erg1 double mutants show high proportion of combined fluconazole and terbinafine resistance. Mycoses. 2020;63:1175-1180. doi:10.1111/myc.13150
- Khurana A, Agarwal A, Agrawal D, et al. Effect of different itraconazole dosing regimens on cure rates, treatment duration, safety, and relapse rates in adult patients with tinea corporis/cruris: a randomized clinical trial. JAMA Dermatol. 2022;158:1269-1278. doi:10.1001/jamadermatol.2022.3745
- Itraconazole capsule. DailyMed [Internet]. Updated June 3, 2024. Accessed June 19, 2024. https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=2ab38a8a-3708-4b97-9f7f-8e554a15348d
- Bui TS, Katz KA. Resistant Trichophyton indotineae dermatophytosis—an emerging pandemic, now in the US. JAMA Dermatol. Published online May 15, 2024. doi:10.1001/jamadermatol.2024.1125
To the Editor:
Historically, commonly available antifungal medications have been effective for treating dermatophytosis (tinea). However, recent tinea outbreaks caused by Trichophyton indotineae—a dermatophyte often resistant to terbinafine and sometimes to other antifungals—have been reported in South Asia, Europe, the Middle East, Southeast Asia, and Australia.1-5
Three confirmed cases of T indotineae dermatophytosis in the United States were reported in 2023 in New York3,6; a fourth confirmed case was reported in 2024 in Pennsylvania.7 Post hoc laboratory testing of fungal isolates in New York in 2022 and 2023 identified an additional 11 cases.8 We present a case of extensive multidrug-resistant tinea caused by T indotineae in a man in California.
An otherwise healthy 65-year-old man who had traveled to Europe in the past 3 months presented to his primary care physician with a widespread pruritic rash (Figure 1). He was treated with 2 weeks of oral terbinafine 250 mg/d and topical medicines, including clotrimazole cream 1%, fluocinonide ointment 0.05%, and clobetasol ointment 0.05% without improvement. Subsequently, 2 weeks of oral griseofulvin microsize 500 mg/d also proved ineffective. An antibody test was negative for HIV. His hemoglobin A1c was 6.2% (reference range, ≤5.6%). The patient was referred to dermatology.
Erythematous plaques—many scaly throughout and some annular with central clearing—were present on the arms, legs, and torso as well as in the groin. Honey crust was present on some plaques on the leg. A potassium hydroxide preparation showed abundant fungal hyphae. Material for fungal and bacterial cultures was collected. The patient was treated again with oral terbinafine 250 mg/d, an oral prednisone taper starting at 60 mg/d for a presumed id reaction, and various oral antihistamines for pruritus; all were ineffective. A bacterial culture showed only mixed skin flora. Oral fluconazole 200 mg/d was prescribed. A skin biopsy specimen showed compact orthokeratosis and parakeratosis of the stratum corneum with few neutrophils and focal pustule formation (Figure 2). Superficial perivascular inflammation, including lymphocytes, histiocytes, and few neutrophils, was present. A periodic acid–Schiff stain showed fungal hyphae in the stratum corneum and a hair follicle (Figure 3). After approximately 2 weeks, mold was identified in the fungal culture. Approximately 2 weeks thereafter, the organism was reported as Trichophyton species.

The rash did not improve; resistance to terbinafine, griseofulvin, and fluconazole was suspected clinically. The fungal isolate was sent to a reference laboratory (University of Texas Health Science Center, San Antonio). Meanwhile, oral itraconazole 200 mg twice daily and ketoconazole cream 2% were prescribed; the rash began to improve. A serum itraconazole trough level obtained 4 days after treatment initiation was 0.5 μg/mL (reference range, ≥0.6 μg/mL). The evening itraconazole dose was increased to 300 mg; a subsequent trough level was 0.8 μg/mL.


Approximately 1 month after the fungal isolate was sent to the reference laboratory, T indotineae was confirmed based on polymerase chain reaction (PCR) testing of internal transcribed spacer region sequences. Minimum inhibitory concentrations (MICs) obtained through antifungal susceptibility testing (AFST) were reported for fluconazole (8 μg/mL), griseofulvin (2 μg/mL), itraconazole (≤0.03 μg/mL), posaconazole (≤0.03 μg/mL), terbinafine (≥2 μg/mL), and voriconazole (0.125 μg/mL).
Approximately 7 weeks after itraconazole and ketoconazole were started, the rash had completely resolved. Nearly 8 months later (at the time this article was written), the rash had not recurred.
We report a unique case of T indotineae in a patient residing in California. Post hoc laboratory testing of dermatophyte isolates sent to the University of Texas reference laboratory identified terbinafine-resistant T indotineae specimens from the United States and Canada dating to 2017; clinical characteristics of patients from whom those isolates were obtained were unavailable.9
Trichophyton indotineae dermatophytosis typically is more extensive, inflamed, and pruritic, as well as likely more contagious, than tinea caused by other dermatophytes.5 Previously called Trichophyton mentagrophytes genotype VIII when first isolated in 2017, the pathogen was renamed T indotineae in 2020 after important genetic differences were discovered between it and other T mentagrophytes species.5 The emergence of T indotineae has been attributed to concomitant use of topical steroids and antifungals,5,10 inappropriate prescribing of antifungals,5 and nonadherence to antifungal treatment.5
Likely risk factors for T indotineae infection include suboptimal hygiene, overcrowded conditions, hot and humid environments, and tight-fitting synthetic clothing.4 Transmission from family members appears common,5 especially when fomites are shared.4 A case reported in Pennsylvania likely was acquired through sexual contact.7 Travel to South Asia has been associated with acquisition of T indotineae infection,3,5-7 though our patient and some others had not traveled there.3,8 It is not clear whether immunosuppression and diabetes mellitus are associated with T indotineae infection.4,5,8 Trichophyton indotineae also can affect animals,11 though zoonotic transmission has not been reported.4
Not all T indotineae isolates are resistant to one or more antifungals; furthermore, antifungal resistance in other dermatophyte species has been reported.5 Terbinafine resistance in T indotineae is conferred by mutations in the gene encoding squalene epoxidase, which helps synthesize ergosterol—a component of the cell membrane in fungi.2,4,5,12 Although clinical cut-points for MIC obtained by AFST are not well established, T indotineae MICs for terbinafine of 0.5 μg/mL or more correlate with resistance.9 Resistance to azoles has been linked to overexpression of transporter genes, which increase azole efflux from cells, as well as to mutations in the gene encoding lanosterol 14α demethylase.4,12,13
Potassium hydroxide preparations and fungal cultures cannot differentiate T indotineae from other dermatophytes that typically cause tinea.5,14 Histopathologic findings in our case were no different than those of non–T indotineae dermatophytes. Only molecular testing using PCR assays to sequence internal transcribed spacer genes can confirm T indotineae infection. However, PCR assays and AFST are not available in many US laboratories.5 Matrix-assisted laser desorption ionization-time of flight (MALDI-TOF) mass spectrometry has shown promise in distinguishing T indotineae from other dermatophytes, though its clinical use is limited and it cannot assess terbinafine sensitivity.15,16 Clinicians in the United States who want to test specimens from cases suspicious for T indotineae infection should contact their local or state health department or the Centers for Disease Control and Prevention for assistance.3,5
Systemic treatment typically is necessary for T indotineae infection.5 Combinations of oral and topical azoles have been used, as well as topical ciclopirox, amorolfine (not available in the United States), and luliconazole.1,5,17-21
Itraconazole has emerged as the treatment of choice for T indotineae tinea, typically at 200 mg/d and often for courses of more than 3 months.5 Testing for serum itraconazole trough levels, as done for our patient, typically is not recommended. Clinicians should counsel patients to take itraconazole with high-fat foods and an acidic beverage to increase bioavailability.5 Potential adverse effects of itraconazole include heart failure and numerous drug-drug interactions.5,22 Patients with T indotineae dermatophytosis should avoid sharing personal belongings and having skin-to-skin contact of affected areas with others.4
Dermatologists who suspect T indotineae infection should work with public health agencies that can assist with testing and undertake infection surveillance, prevention, and control.5,23 Challenges to diagnosing and managing T indotineae infection include lack of awareness among dermatology providers, the need for specialized laboratory testing to confirm infection, lack of established clinical cut-points for MICs from AFST, the need for longer duration of treatment vs what is needed for typical tinea, and potential challenges with insurance coverage for testing and treatment. Empiric treatment with itraconazole should be considered when terbinafine-resistant dermatophytosis is suspected or when terbinafine-resistant T indotineae infection is confirmed.
Acknowledgments—Jeremy Gold, MD; Dallas J. Smith, PharmD; and Shawn Lockhart, PhD, all of the Centers for Disease Control and Prevention, Mycotic Diseases Branch (Atlanta, Georgia), provided helpful comments to the authors in preparing the manuscript of this article.
To the Editor:
Historically, commonly available antifungal medications have been effective for treating dermatophytosis (tinea). However, recent tinea outbreaks caused by Trichophyton indotineae—a dermatophyte often resistant to terbinafine and sometimes to other antifungals—have been reported in South Asia, Europe, the Middle East, Southeast Asia, and Australia.1-5
Three confirmed cases of T indotineae dermatophytosis in the United States were reported in 2023 in New York3,6; a fourth confirmed case was reported in 2024 in Pennsylvania.7 Post hoc laboratory testing of fungal isolates in New York in 2022 and 2023 identified an additional 11 cases.8 We present a case of extensive multidrug-resistant tinea caused by T indotineae in a man in California.
An otherwise healthy 65-year-old man who had traveled to Europe in the past 3 months presented to his primary care physician with a widespread pruritic rash (Figure 1). He was treated with 2 weeks of oral terbinafine 250 mg/d and topical medicines, including clotrimazole cream 1%, fluocinonide ointment 0.05%, and clobetasol ointment 0.05% without improvement. Subsequently, 2 weeks of oral griseofulvin microsize 500 mg/d also proved ineffective. An antibody test was negative for HIV. His hemoglobin A1c was 6.2% (reference range, ≤5.6%). The patient was referred to dermatology.
Erythematous plaques—many scaly throughout and some annular with central clearing—were present on the arms, legs, and torso as well as in the groin. Honey crust was present on some plaques on the leg. A potassium hydroxide preparation showed abundant fungal hyphae. Material for fungal and bacterial cultures was collected. The patient was treated again with oral terbinafine 250 mg/d, an oral prednisone taper starting at 60 mg/d for a presumed id reaction, and various oral antihistamines for pruritus; all were ineffective. A bacterial culture showed only mixed skin flora. Oral fluconazole 200 mg/d was prescribed. A skin biopsy specimen showed compact orthokeratosis and parakeratosis of the stratum corneum with few neutrophils and focal pustule formation (Figure 2). Superficial perivascular inflammation, including lymphocytes, histiocytes, and few neutrophils, was present. A periodic acid–Schiff stain showed fungal hyphae in the stratum corneum and a hair follicle (Figure 3). After approximately 2 weeks, mold was identified in the fungal culture. Approximately 2 weeks thereafter, the organism was reported as Trichophyton species.

The rash did not improve; resistance to terbinafine, griseofulvin, and fluconazole was suspected clinically. The fungal isolate was sent to a reference laboratory (University of Texas Health Science Center, San Antonio). Meanwhile, oral itraconazole 200 mg twice daily and ketoconazole cream 2% were prescribed; the rash began to improve. A serum itraconazole trough level obtained 4 days after treatment initiation was 0.5 μg/mL (reference range, ≥0.6 μg/mL). The evening itraconazole dose was increased to 300 mg; a subsequent trough level was 0.8 μg/mL.


Approximately 1 month after the fungal isolate was sent to the reference laboratory, T indotineae was confirmed based on polymerase chain reaction (PCR) testing of internal transcribed spacer region sequences. Minimum inhibitory concentrations (MICs) obtained through antifungal susceptibility testing (AFST) were reported for fluconazole (8 μg/mL), griseofulvin (2 μg/mL), itraconazole (≤0.03 μg/mL), posaconazole (≤0.03 μg/mL), terbinafine (≥2 μg/mL), and voriconazole (0.125 μg/mL).
Approximately 7 weeks after itraconazole and ketoconazole were started, the rash had completely resolved. Nearly 8 months later (at the time this article was written), the rash had not recurred.
We report a unique case of T indotineae in a patient residing in California. Post hoc laboratory testing of dermatophyte isolates sent to the University of Texas reference laboratory identified terbinafine-resistant T indotineae specimens from the United States and Canada dating to 2017; clinical characteristics of patients from whom those isolates were obtained were unavailable.9
Trichophyton indotineae dermatophytosis typically is more extensive, inflamed, and pruritic, as well as likely more contagious, than tinea caused by other dermatophytes.5 Previously called Trichophyton mentagrophytes genotype VIII when first isolated in 2017, the pathogen was renamed T indotineae in 2020 after important genetic differences were discovered between it and other T mentagrophytes species.5 The emergence of T indotineae has been attributed to concomitant use of topical steroids and antifungals,5,10 inappropriate prescribing of antifungals,5 and nonadherence to antifungal treatment.5
Likely risk factors for T indotineae infection include suboptimal hygiene, overcrowded conditions, hot and humid environments, and tight-fitting synthetic clothing.4 Transmission from family members appears common,5 especially when fomites are shared.4 A case reported in Pennsylvania likely was acquired through sexual contact.7 Travel to South Asia has been associated with acquisition of T indotineae infection,3,5-7 though our patient and some others had not traveled there.3,8 It is not clear whether immunosuppression and diabetes mellitus are associated with T indotineae infection.4,5,8 Trichophyton indotineae also can affect animals,11 though zoonotic transmission has not been reported.4
Not all T indotineae isolates are resistant to one or more antifungals; furthermore, antifungal resistance in other dermatophyte species has been reported.5 Terbinafine resistance in T indotineae is conferred by mutations in the gene encoding squalene epoxidase, which helps synthesize ergosterol—a component of the cell membrane in fungi.2,4,5,12 Although clinical cut-points for MIC obtained by AFST are not well established, T indotineae MICs for terbinafine of 0.5 μg/mL or more correlate with resistance.9 Resistance to azoles has been linked to overexpression of transporter genes, which increase azole efflux from cells, as well as to mutations in the gene encoding lanosterol 14α demethylase.4,12,13
Potassium hydroxide preparations and fungal cultures cannot differentiate T indotineae from other dermatophytes that typically cause tinea.5,14 Histopathologic findings in our case were no different than those of non–T indotineae dermatophytes. Only molecular testing using PCR assays to sequence internal transcribed spacer genes can confirm T indotineae infection. However, PCR assays and AFST are not available in many US laboratories.5 Matrix-assisted laser desorption ionization-time of flight (MALDI-TOF) mass spectrometry has shown promise in distinguishing T indotineae from other dermatophytes, though its clinical use is limited and it cannot assess terbinafine sensitivity.15,16 Clinicians in the United States who want to test specimens from cases suspicious for T indotineae infection should contact their local or state health department or the Centers for Disease Control and Prevention for assistance.3,5
Systemic treatment typically is necessary for T indotineae infection.5 Combinations of oral and topical azoles have been used, as well as topical ciclopirox, amorolfine (not available in the United States), and luliconazole.1,5,17-21
Itraconazole has emerged as the treatment of choice for T indotineae tinea, typically at 200 mg/d and often for courses of more than 3 months.5 Testing for serum itraconazole trough levels, as done for our patient, typically is not recommended. Clinicians should counsel patients to take itraconazole with high-fat foods and an acidic beverage to increase bioavailability.5 Potential adverse effects of itraconazole include heart failure and numerous drug-drug interactions.5,22 Patients with T indotineae dermatophytosis should avoid sharing personal belongings and having skin-to-skin contact of affected areas with others.4
Dermatologists who suspect T indotineae infection should work with public health agencies that can assist with testing and undertake infection surveillance, prevention, and control.5,23 Challenges to diagnosing and managing T indotineae infection include lack of awareness among dermatology providers, the need for specialized laboratory testing to confirm infection, lack of established clinical cut-points for MICs from AFST, the need for longer duration of treatment vs what is needed for typical tinea, and potential challenges with insurance coverage for testing and treatment. Empiric treatment with itraconazole should be considered when terbinafine-resistant dermatophytosis is suspected or when terbinafine-resistant T indotineae infection is confirmed.
Acknowledgments—Jeremy Gold, MD; Dallas J. Smith, PharmD; and Shawn Lockhart, PhD, all of the Centers for Disease Control and Prevention, Mycotic Diseases Branch (Atlanta, Georgia), provided helpful comments to the authors in preparing the manuscript of this article.
- Uhrlaß S, Verma SB, Gräser Y, al. Trichophyton indotineae—an emerging pathogen causing recalcitrant dermatophytoses in India and worldwide—a multidimensional perspective. J Fungi (Basel). 2022;8:757. doi:10.3390/jof8070757
- Jabet A, Brun S, Normand A-C, et al. Extensive dermatophytosis caused by terbinafine-resistant Trichophyton indotineae, France. Emerg Infect Dis. 2022;28:229-233. doi:10.3201/eid2801.210883
- Caplan AS, Chaturvedi S, Zhu Y, et al. Notes from the field. First reported U.S. cases of tinea caused by Trichophyton indotineae—New York City, December 2021-March 2023. MMWR Morb Mortal Wkly Rep. 2023;72:536-537. doi:10.15585/mmwr.mm7219a4
- Jabet A, Normand A-C, Brun S, et al. Trichophyton indotineae, from epidemiology to therapeutic. J Mycol Med. 2023;33:101383. doi:10.1016/j.mycmed.2023.101383
- Hill RC, Caplan AS, Elewski B, et al. Expert panel review of skin and hair dermatophytoses in an era of antifungal resistance. Am J Clin Dermatol. 2024;25:359-389. doi:10.1007/s40257-024-00848-1
- Caplan AS, Zakhem GA, Pomeranz MK. Trichophyton mentagrophytes internal transcribed spacer genotype VIII. JAMA Dermatol. 2023;159:1130. doi:10.1001/jamadermatol.2023.2645
- Spivack S, Gold JAW, Lockhart SR, et al. Potential sexual transmission of antifungal-resistant Trichophyton indotineae. Emerg Infect Dis. 2024;30:807-809. doi:10.3201/eid3004.240115
- Caplan AS, Todd GC, Zhu Y, et al. Clinical course, antifungal susceptibility, and genomic sequencing of Trichophyton indotineae. JAMA Dermatol. Published online May 15, 2024. doi:10.1001/jamadermatol.2024.1126
- Cañete-Gibas CF, Mele J, Patterson HP, et al. Terbinafine-resistant dermatophytes and the presence of Trichophyton indotineae in North America. J Clin Microbiol. 2023;61:e0056223. doi:10.1128/jcm.00562-23
- Gupta AK, Venkataraman M, Hall DC, et al. The emergence of Trichophyton indotineae: implications for clinical practice. Int J Dermatol. 2023;62:857-861.
- Oladzad V, Nasrollahi Omran A, Haghani I, et al. Multi-drug resistance Trichophyton indotineae in a stray dog. Res Vet Sci. 2024;166:105105. doi:10.1016/j.rvsc.2023.105105
- Martinez-Rossi NM, Bitencourt TA, Peres NTA, et al. Dermatophyte resistance to antifungal drugs: mechanisms and prospectus. Front Microbiol. 2018;9:1108. doi:10.3389/fmicb.2018.01108
- Sacheli R, Hayette MP. Antifungal resistance in dermatophytes: genetic considerations, clinical presentations and alternative therapies. J Fungi (Basel). 2021;711:983. doi:10.3390/jof7110983
- Gupta AK, Cooper EA. Dermatophytosis (tinea) and other superficial fungal infections. In: Hospenthal DR, Rinaldi MG, eds. Diagnosis and Treatment of Human Mycoses. Humana Press; 2008:355-381.
- Normand A-C, Moreno-Sabater A, Jabet A, et al. MALDI-TOF mass spectrometry online identification of Trichophyton indotineae using the MSI-2 application. J Fungi (Basel). 2022;8:1103. doi:10.3390/jof8101103
- De Paepe R, Normand A-C, Uhrlaß S, et al. Resistance profile, terbinafine resistance screening and MALDI-TOF MS identification of the emerging pathogen Trichophyton indotineae. Mycopathologia. 2024;189:29. doi:10.1007/s11046-024-00835-4
- Rajagopalan M, Inamadar A, Mittal A, et al. Expert consensus on the management of dermatophytosis in India (ECTODERM India). BMC Dermatol. 2018;18:6. doi:10.1186/s12895-018-0073-1
- Verma SB, Panda S, Nenoff P, et al. The unprecedented epidemic-like scenario of dermatophytosis in India: III. Antifungal resistance and treatment options. Indian J Dermatol Venereol Leprol. 2021;87:468-482. doi:10.25259/IJDVL_303_20
- Shaw D, Singh S, Dogra S, et al. MIC and upper limit of wild-type distribution for 13 antifungal agents against a Trichophyton mentagrophytes–Trichophyton interdigitale complex of Indian origin. Antimicrob Agents Chemother. 2020;64:E01964-19. doi:10.1128/AAC.01964-19
- Burmester A, Hipler U-C, Uhrlaß S, et al. Indian Trichophyton mentagrophytes squalene epoxidase erg1 double mutants show high proportion of combined fluconazole and terbinafine resistance. Mycoses. 2020;63:1175-1180. doi:10.1111/myc.13150
- Khurana A, Agarwal A, Agrawal D, et al. Effect of different itraconazole dosing regimens on cure rates, treatment duration, safety, and relapse rates in adult patients with tinea corporis/cruris: a randomized clinical trial. JAMA Dermatol. 2022;158:1269-1278. doi:10.1001/jamadermatol.2022.3745
- Itraconazole capsule. DailyMed [Internet]. Updated June 3, 2024. Accessed June 19, 2024. https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=2ab38a8a-3708-4b97-9f7f-8e554a15348d
- Bui TS, Katz KA. Resistant Trichophyton indotineae dermatophytosis—an emerging pandemic, now in the US. JAMA Dermatol. Published online May 15, 2024. doi:10.1001/jamadermatol.2024.1125
- Uhrlaß S, Verma SB, Gräser Y, al. Trichophyton indotineae—an emerging pathogen causing recalcitrant dermatophytoses in India and worldwide—a multidimensional perspective. J Fungi (Basel). 2022;8:757. doi:10.3390/jof8070757
- Jabet A, Brun S, Normand A-C, et al. Extensive dermatophytosis caused by terbinafine-resistant Trichophyton indotineae, France. Emerg Infect Dis. 2022;28:229-233. doi:10.3201/eid2801.210883
- Caplan AS, Chaturvedi S, Zhu Y, et al. Notes from the field. First reported U.S. cases of tinea caused by Trichophyton indotineae—New York City, December 2021-March 2023. MMWR Morb Mortal Wkly Rep. 2023;72:536-537. doi:10.15585/mmwr.mm7219a4
- Jabet A, Normand A-C, Brun S, et al. Trichophyton indotineae, from epidemiology to therapeutic. J Mycol Med. 2023;33:101383. doi:10.1016/j.mycmed.2023.101383
- Hill RC, Caplan AS, Elewski B, et al. Expert panel review of skin and hair dermatophytoses in an era of antifungal resistance. Am J Clin Dermatol. 2024;25:359-389. doi:10.1007/s40257-024-00848-1
- Caplan AS, Zakhem GA, Pomeranz MK. Trichophyton mentagrophytes internal transcribed spacer genotype VIII. JAMA Dermatol. 2023;159:1130. doi:10.1001/jamadermatol.2023.2645
- Spivack S, Gold JAW, Lockhart SR, et al. Potential sexual transmission of antifungal-resistant Trichophyton indotineae. Emerg Infect Dis. 2024;30:807-809. doi:10.3201/eid3004.240115
- Caplan AS, Todd GC, Zhu Y, et al. Clinical course, antifungal susceptibility, and genomic sequencing of Trichophyton indotineae. JAMA Dermatol. Published online May 15, 2024. doi:10.1001/jamadermatol.2024.1126
- Cañete-Gibas CF, Mele J, Patterson HP, et al. Terbinafine-resistant dermatophytes and the presence of Trichophyton indotineae in North America. J Clin Microbiol. 2023;61:e0056223. doi:10.1128/jcm.00562-23
- Gupta AK, Venkataraman M, Hall DC, et al. The emergence of Trichophyton indotineae: implications for clinical practice. Int J Dermatol. 2023;62:857-861.
- Oladzad V, Nasrollahi Omran A, Haghani I, et al. Multi-drug resistance Trichophyton indotineae in a stray dog. Res Vet Sci. 2024;166:105105. doi:10.1016/j.rvsc.2023.105105
- Martinez-Rossi NM, Bitencourt TA, Peres NTA, et al. Dermatophyte resistance to antifungal drugs: mechanisms and prospectus. Front Microbiol. 2018;9:1108. doi:10.3389/fmicb.2018.01108
- Sacheli R, Hayette MP. Antifungal resistance in dermatophytes: genetic considerations, clinical presentations and alternative therapies. J Fungi (Basel). 2021;711:983. doi:10.3390/jof7110983
- Gupta AK, Cooper EA. Dermatophytosis (tinea) and other superficial fungal infections. In: Hospenthal DR, Rinaldi MG, eds. Diagnosis and Treatment of Human Mycoses. Humana Press; 2008:355-381.
- Normand A-C, Moreno-Sabater A, Jabet A, et al. MALDI-TOF mass spectrometry online identification of Trichophyton indotineae using the MSI-2 application. J Fungi (Basel). 2022;8:1103. doi:10.3390/jof8101103
- De Paepe R, Normand A-C, Uhrlaß S, et al. Resistance profile, terbinafine resistance screening and MALDI-TOF MS identification of the emerging pathogen Trichophyton indotineae. Mycopathologia. 2024;189:29. doi:10.1007/s11046-024-00835-4
- Rajagopalan M, Inamadar A, Mittal A, et al. Expert consensus on the management of dermatophytosis in India (ECTODERM India). BMC Dermatol. 2018;18:6. doi:10.1186/s12895-018-0073-1
- Verma SB, Panda S, Nenoff P, et al. The unprecedented epidemic-like scenario of dermatophytosis in India: III. Antifungal resistance and treatment options. Indian J Dermatol Venereol Leprol. 2021;87:468-482. doi:10.25259/IJDVL_303_20
- Shaw D, Singh S, Dogra S, et al. MIC and upper limit of wild-type distribution for 13 antifungal agents against a Trichophyton mentagrophytes–Trichophyton interdigitale complex of Indian origin. Antimicrob Agents Chemother. 2020;64:E01964-19. doi:10.1128/AAC.01964-19
- Burmester A, Hipler U-C, Uhrlaß S, et al. Indian Trichophyton mentagrophytes squalene epoxidase erg1 double mutants show high proportion of combined fluconazole and terbinafine resistance. Mycoses. 2020;63:1175-1180. doi:10.1111/myc.13150
- Khurana A, Agarwal A, Agrawal D, et al. Effect of different itraconazole dosing regimens on cure rates, treatment duration, safety, and relapse rates in adult patients with tinea corporis/cruris: a randomized clinical trial. JAMA Dermatol. 2022;158:1269-1278. doi:10.1001/jamadermatol.2022.3745
- Itraconazole capsule. DailyMed [Internet]. Updated June 3, 2024. Accessed June 19, 2024. https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=2ab38a8a-3708-4b97-9f7f-8e554a15348d
- Bui TS, Katz KA. Resistant Trichophyton indotineae dermatophytosis—an emerging pandemic, now in the US. JAMA Dermatol. Published online May 15, 2024. doi:10.1001/jamadermatol.2024.1125
Practice Points
- Trichophyton indotineae can cause extensive dermatophytosis that often is resistant to terbinafine and in some cases to other antifungals.
- Only molecular testing, which is not widely available, can distinguish T indotineae from other dermatophytes.
- Suspected or confirmed cases of T indotineae dermatophytosis should be reported to public health agencies to provide assistance with testing, as well as surveillance, prevention, and control of infection.
New Trials in Lung Cancer: Could Your Patients Benefit?
Resected stage II, IIIA, or IIIB with nodal involvement non–small cell lung cancer (NSCLC). Adult patients with this type of cancer can join a randomized, controlled, phase 3 study assessing whether an investigational drug called V940 added to pembrolizumab (Keytruda) delays cancer recurrence better than pembrolizumab alone.
V940 is an individualized neoantigen therapy designed to generate T-cell antitumor responses targeted to a patient’s specific mutation profile.
V940 plus pembrolizumab showed a trend toward longer recurrence-free survival vs pembrolizumab alone in a recent phase 2 study in melanoma (hazard ratio, 0.561; P = .053).
In the current trial, one group of participants will receive intramuscular injections of V940 every 3 weeks plus intravenous (IV) pembrolizumab every 6 weeks for up to approximately 1 year or until disease recurrence or unacceptable toxicity, whichever happens first. The other people in the trial will be on the same schedule, with a placebo replacing V940.
Centers in Florida, Georgia, Kentucky, Montana, New Jersey, New York, North Dakota, and six other countries started recruiting for the trial’s 868 participants in December 2023. Disease-free survival is the primary endpoint. Overall survival over approximately 12 years and quality of life (QoL) are secondary endpoints. More details at clinicaltrials.gov.
Metastatic NSCLC with a programmed cell death ligand 1 (PD-L1)–tumor proportion score of > 50%. Adults in this clinical situation are eligible for a randomized, open-label, phase 3 trial to determine whether an experimental antibody-drug conjugate called MK-2870 added to standard pembrolizumab prolongs survival.
MK-2870 delivers a cytotoxin to cancer cells by binding to trophoblast cell-surface antigen 2, known to promote tumor cell growth and metastasis. For up to 2 years, half of participants will receive MK-2870 by IV every 2 weeks plus IV pembrolizumab every 6 weeks. The other group will receive only pembrolizumab.
In December 2023, study sites in Georgia, Minnesota, Mississippi, Nevada, Oregon, Australia, Denmark, Taiwan, and Turkey started seeking the trial’s 614 participants. Overall survival over approximately 4 years is the primary endpoint; QoL is a secondary endpoint. More details at clinicaltrials.gov.
Untreated locally advanced or metastatic NSCLC with KRAS G12C mutations. Individuals with this type of lung cancer may be interested in a randomized, controlled, phase 3 study examining whether an experimental oral KRAS G12C inhibitor called LY3537982 boosts the effectiveness of standard treatment and patients can tolerate the combination. Currently approved KRAS G12C inhibitors sotorasib (Lumakras, Lumykras) and adagrasib (Krazati) are indicated for second-line treatment; this trial may lead to a first-line approval for newcomer LY3537982.
The trial has three parts: dose optimization, safety, and efficacy. During dose optimization, each participant will take one of two oral doses of LY3537982 and receive IV pembrolizumab every 3 weeks. In the safety phase, all participants will receive oral LY3537982 at the chosen dose plus standard therapy of 3-times-weekly IV pembrolizumab, pemetrexed, and a platinum therapy (cisplatin or carboplatin). In the experimental phase, for up to about 1 year, participants will receive one of these four options: Pembrolizumab plus LY3537982, pembrolizumab plus a placebo, standard therapy plus LY3537982, or standard therapy plus a placebo.
The study, which is planning to recruit 1016 participants, opened across 16 US states and 12 countries worldwide in December 2023. Sites in 11 more US states, the District of Columbia, Brazil, Canada, China, India, and 11 more European countries are gearing up. Adverse events and progression-free survival are the primary endpoints. Overall survival over approximately 3 years and QoL are secondary endpoints. More details at clinicaltrials.gov.
Unresectable, untreated locally advanced or metastatic non-squamous NSCLC with human epidermal growth factor receptor 2 (HER2) mutations. People with this diagnosis who have HER2 mutations instead of KRAS G12C mutations can participate in a phase 3 study comparing an investigational oral first-line treatment with standard IV therapy. The drug in this study, zongertinib, is a HER2 tyrosine kinase inhibitor.
For up to approximately 4 years, one group of participants will take oral zongertinib only, and the other individuals will receive IV pembrolizumab, pemetrexed, and a platinum agent (cisplatin or carboplatin). Study sites in California, Missouri, South Carolina, Australia, China, Japan, South Korea, and Singapore opened in January ready to welcome 270 participants. Progression-free survival is the primary outcome. Overall survival over 53 months and QoL are secondary endpoints. More details at clinicaltrials.gov.
Completely resected stage IIB, IIIA, or select IIIB, PD-L1–positive NSCLC. Adults with this type of lung cancer who have received adjuvant platinum-based chemotherapy may be eligible for a randomized, controlled, phase 3 study to assess whether two immune checkpoint inhibitors are better than one at delaying cancer recurrence. In this trial, tiragolumab will be added to the approved PD-L1 inhibitor atezolizumab (Tecentriq).
A recent study, however, found that tiragolumab did not confer an additional benefit when added to atezolizumab, carboplatin, and etoposide in untreated extensive-stage small cell lung cancer.
In the current trial, one group of participants will receive IV atezolizumab and tiragolumab, while the other people will receive a placebo instead of tiragolumab. Centers in California, Georgia, Illinois, New Mexico, Australia, China, South Korea, and Taiwan started recruiting for the trial’s 1150 participants in March 2024. Disease-free survival is the primary endpoint. Overall survival over approximately 15 years and QoL are secondary outcomes. More details at clinicaltrials.gov.
Previously treated metastatic or non-operable non-squamous NSCLC. Adults in this position who have received no more than one platinum-based chemotherapy and one anti–PD-L1 drug are sought for a randomized, open-label, phase 3 trial comparing second-line standard docetaxel with experimental antibody-drug conjugate sigvotatug vedotin. Patients who have tumors with certain treatable genomic alterations must have received at least one drug targeted to that alteration, as well as a platinum-based agent.
Approximately half the participants will receive sigvotatug vedotin by IV every 2 weeks, and the other half will receive IV docetaxel every 3 weeks. The study opened in March across 13 US states, France, Hungary, Poland, and Spain seeking 600 people eligible to participate. The primary outcomes are overall survival over approximately 5 years and objective response rate. QoL is a secondary outcome. More details at clinicaltrials.gov.All trial information is from the National Institutes of Health US National Library of Medicine (online at clinicaltrials.gov).
A version of this article appeared on Medscape.com .
Resected stage II, IIIA, or IIIB with nodal involvement non–small cell lung cancer (NSCLC). Adult patients with this type of cancer can join a randomized, controlled, phase 3 study assessing whether an investigational drug called V940 added to pembrolizumab (Keytruda) delays cancer recurrence better than pembrolizumab alone.
V940 is an individualized neoantigen therapy designed to generate T-cell antitumor responses targeted to a patient’s specific mutation profile.
V940 plus pembrolizumab showed a trend toward longer recurrence-free survival vs pembrolizumab alone in a recent phase 2 study in melanoma (hazard ratio, 0.561; P = .053).
In the current trial, one group of participants will receive intramuscular injections of V940 every 3 weeks plus intravenous (IV) pembrolizumab every 6 weeks for up to approximately 1 year or until disease recurrence or unacceptable toxicity, whichever happens first. The other people in the trial will be on the same schedule, with a placebo replacing V940.
Centers in Florida, Georgia, Kentucky, Montana, New Jersey, New York, North Dakota, and six other countries started recruiting for the trial’s 868 participants in December 2023. Disease-free survival is the primary endpoint. Overall survival over approximately 12 years and quality of life (QoL) are secondary endpoints. More details at clinicaltrials.gov.
Metastatic NSCLC with a programmed cell death ligand 1 (PD-L1)–tumor proportion score of > 50%. Adults in this clinical situation are eligible for a randomized, open-label, phase 3 trial to determine whether an experimental antibody-drug conjugate called MK-2870 added to standard pembrolizumab prolongs survival.
MK-2870 delivers a cytotoxin to cancer cells by binding to trophoblast cell-surface antigen 2, known to promote tumor cell growth and metastasis. For up to 2 years, half of participants will receive MK-2870 by IV every 2 weeks plus IV pembrolizumab every 6 weeks. The other group will receive only pembrolizumab.
In December 2023, study sites in Georgia, Minnesota, Mississippi, Nevada, Oregon, Australia, Denmark, Taiwan, and Turkey started seeking the trial’s 614 participants. Overall survival over approximately 4 years is the primary endpoint; QoL is a secondary endpoint. More details at clinicaltrials.gov.
Untreated locally advanced or metastatic NSCLC with KRAS G12C mutations. Individuals with this type of lung cancer may be interested in a randomized, controlled, phase 3 study examining whether an experimental oral KRAS G12C inhibitor called LY3537982 boosts the effectiveness of standard treatment and patients can tolerate the combination. Currently approved KRAS G12C inhibitors sotorasib (Lumakras, Lumykras) and adagrasib (Krazati) are indicated for second-line treatment; this trial may lead to a first-line approval for newcomer LY3537982.
The trial has three parts: dose optimization, safety, and efficacy. During dose optimization, each participant will take one of two oral doses of LY3537982 and receive IV pembrolizumab every 3 weeks. In the safety phase, all participants will receive oral LY3537982 at the chosen dose plus standard therapy of 3-times-weekly IV pembrolizumab, pemetrexed, and a platinum therapy (cisplatin or carboplatin). In the experimental phase, for up to about 1 year, participants will receive one of these four options: Pembrolizumab plus LY3537982, pembrolizumab plus a placebo, standard therapy plus LY3537982, or standard therapy plus a placebo.
The study, which is planning to recruit 1016 participants, opened across 16 US states and 12 countries worldwide in December 2023. Sites in 11 more US states, the District of Columbia, Brazil, Canada, China, India, and 11 more European countries are gearing up. Adverse events and progression-free survival are the primary endpoints. Overall survival over approximately 3 years and QoL are secondary endpoints. More details at clinicaltrials.gov.
Unresectable, untreated locally advanced or metastatic non-squamous NSCLC with human epidermal growth factor receptor 2 (HER2) mutations. People with this diagnosis who have HER2 mutations instead of KRAS G12C mutations can participate in a phase 3 study comparing an investigational oral first-line treatment with standard IV therapy. The drug in this study, zongertinib, is a HER2 tyrosine kinase inhibitor.
For up to approximately 4 years, one group of participants will take oral zongertinib only, and the other individuals will receive IV pembrolizumab, pemetrexed, and a platinum agent (cisplatin or carboplatin). Study sites in California, Missouri, South Carolina, Australia, China, Japan, South Korea, and Singapore opened in January ready to welcome 270 participants. Progression-free survival is the primary outcome. Overall survival over 53 months and QoL are secondary endpoints. More details at clinicaltrials.gov.
Completely resected stage IIB, IIIA, or select IIIB, PD-L1–positive NSCLC. Adults with this type of lung cancer who have received adjuvant platinum-based chemotherapy may be eligible for a randomized, controlled, phase 3 study to assess whether two immune checkpoint inhibitors are better than one at delaying cancer recurrence. In this trial, tiragolumab will be added to the approved PD-L1 inhibitor atezolizumab (Tecentriq).
A recent study, however, found that tiragolumab did not confer an additional benefit when added to atezolizumab, carboplatin, and etoposide in untreated extensive-stage small cell lung cancer.
In the current trial, one group of participants will receive IV atezolizumab and tiragolumab, while the other people will receive a placebo instead of tiragolumab. Centers in California, Georgia, Illinois, New Mexico, Australia, China, South Korea, and Taiwan started recruiting for the trial’s 1150 participants in March 2024. Disease-free survival is the primary endpoint. Overall survival over approximately 15 years and QoL are secondary outcomes. More details at clinicaltrials.gov.
Previously treated metastatic or non-operable non-squamous NSCLC. Adults in this position who have received no more than one platinum-based chemotherapy and one anti–PD-L1 drug are sought for a randomized, open-label, phase 3 trial comparing second-line standard docetaxel with experimental antibody-drug conjugate sigvotatug vedotin. Patients who have tumors with certain treatable genomic alterations must have received at least one drug targeted to that alteration, as well as a platinum-based agent.
Approximately half the participants will receive sigvotatug vedotin by IV every 2 weeks, and the other half will receive IV docetaxel every 3 weeks. The study opened in March across 13 US states, France, Hungary, Poland, and Spain seeking 600 people eligible to participate. The primary outcomes are overall survival over approximately 5 years and objective response rate. QoL is a secondary outcome. More details at clinicaltrials.gov.All trial information is from the National Institutes of Health US National Library of Medicine (online at clinicaltrials.gov).
A version of this article appeared on Medscape.com .
Resected stage II, IIIA, or IIIB with nodal involvement non–small cell lung cancer (NSCLC). Adult patients with this type of cancer can join a randomized, controlled, phase 3 study assessing whether an investigational drug called V940 added to pembrolizumab (Keytruda) delays cancer recurrence better than pembrolizumab alone.
V940 is an individualized neoantigen therapy designed to generate T-cell antitumor responses targeted to a patient’s specific mutation profile.
V940 plus pembrolizumab showed a trend toward longer recurrence-free survival vs pembrolizumab alone in a recent phase 2 study in melanoma (hazard ratio, 0.561; P = .053).
In the current trial, one group of participants will receive intramuscular injections of V940 every 3 weeks plus intravenous (IV) pembrolizumab every 6 weeks for up to approximately 1 year or until disease recurrence or unacceptable toxicity, whichever happens first. The other people in the trial will be on the same schedule, with a placebo replacing V940.
Centers in Florida, Georgia, Kentucky, Montana, New Jersey, New York, North Dakota, and six other countries started recruiting for the trial’s 868 participants in December 2023. Disease-free survival is the primary endpoint. Overall survival over approximately 12 years and quality of life (QoL) are secondary endpoints. More details at clinicaltrials.gov.
Metastatic NSCLC with a programmed cell death ligand 1 (PD-L1)–tumor proportion score of > 50%. Adults in this clinical situation are eligible for a randomized, open-label, phase 3 trial to determine whether an experimental antibody-drug conjugate called MK-2870 added to standard pembrolizumab prolongs survival.
MK-2870 delivers a cytotoxin to cancer cells by binding to trophoblast cell-surface antigen 2, known to promote tumor cell growth and metastasis. For up to 2 years, half of participants will receive MK-2870 by IV every 2 weeks plus IV pembrolizumab every 6 weeks. The other group will receive only pembrolizumab.
In December 2023, study sites in Georgia, Minnesota, Mississippi, Nevada, Oregon, Australia, Denmark, Taiwan, and Turkey started seeking the trial’s 614 participants. Overall survival over approximately 4 years is the primary endpoint; QoL is a secondary endpoint. More details at clinicaltrials.gov.
Untreated locally advanced or metastatic NSCLC with KRAS G12C mutations. Individuals with this type of lung cancer may be interested in a randomized, controlled, phase 3 study examining whether an experimental oral KRAS G12C inhibitor called LY3537982 boosts the effectiveness of standard treatment and patients can tolerate the combination. Currently approved KRAS G12C inhibitors sotorasib (Lumakras, Lumykras) and adagrasib (Krazati) are indicated for second-line treatment; this trial may lead to a first-line approval for newcomer LY3537982.
The trial has three parts: dose optimization, safety, and efficacy. During dose optimization, each participant will take one of two oral doses of LY3537982 and receive IV pembrolizumab every 3 weeks. In the safety phase, all participants will receive oral LY3537982 at the chosen dose plus standard therapy of 3-times-weekly IV pembrolizumab, pemetrexed, and a platinum therapy (cisplatin or carboplatin). In the experimental phase, for up to about 1 year, participants will receive one of these four options: Pembrolizumab plus LY3537982, pembrolizumab plus a placebo, standard therapy plus LY3537982, or standard therapy plus a placebo.
The study, which is planning to recruit 1016 participants, opened across 16 US states and 12 countries worldwide in December 2023. Sites in 11 more US states, the District of Columbia, Brazil, Canada, China, India, and 11 more European countries are gearing up. Adverse events and progression-free survival are the primary endpoints. Overall survival over approximately 3 years and QoL are secondary endpoints. More details at clinicaltrials.gov.
Unresectable, untreated locally advanced or metastatic non-squamous NSCLC with human epidermal growth factor receptor 2 (HER2) mutations. People with this diagnosis who have HER2 mutations instead of KRAS G12C mutations can participate in a phase 3 study comparing an investigational oral first-line treatment with standard IV therapy. The drug in this study, zongertinib, is a HER2 tyrosine kinase inhibitor.
For up to approximately 4 years, one group of participants will take oral zongertinib only, and the other individuals will receive IV pembrolizumab, pemetrexed, and a platinum agent (cisplatin or carboplatin). Study sites in California, Missouri, South Carolina, Australia, China, Japan, South Korea, and Singapore opened in January ready to welcome 270 participants. Progression-free survival is the primary outcome. Overall survival over 53 months and QoL are secondary endpoints. More details at clinicaltrials.gov.
Completely resected stage IIB, IIIA, or select IIIB, PD-L1–positive NSCLC. Adults with this type of lung cancer who have received adjuvant platinum-based chemotherapy may be eligible for a randomized, controlled, phase 3 study to assess whether two immune checkpoint inhibitors are better than one at delaying cancer recurrence. In this trial, tiragolumab will be added to the approved PD-L1 inhibitor atezolizumab (Tecentriq).
A recent study, however, found that tiragolumab did not confer an additional benefit when added to atezolizumab, carboplatin, and etoposide in untreated extensive-stage small cell lung cancer.
In the current trial, one group of participants will receive IV atezolizumab and tiragolumab, while the other people will receive a placebo instead of tiragolumab. Centers in California, Georgia, Illinois, New Mexico, Australia, China, South Korea, and Taiwan started recruiting for the trial’s 1150 participants in March 2024. Disease-free survival is the primary endpoint. Overall survival over approximately 15 years and QoL are secondary outcomes. More details at clinicaltrials.gov.
Previously treated metastatic or non-operable non-squamous NSCLC. Adults in this position who have received no more than one platinum-based chemotherapy and one anti–PD-L1 drug are sought for a randomized, open-label, phase 3 trial comparing second-line standard docetaxel with experimental antibody-drug conjugate sigvotatug vedotin. Patients who have tumors with certain treatable genomic alterations must have received at least one drug targeted to that alteration, as well as a platinum-based agent.
Approximately half the participants will receive sigvotatug vedotin by IV every 2 weeks, and the other half will receive IV docetaxel every 3 weeks. The study opened in March across 13 US states, France, Hungary, Poland, and Spain seeking 600 people eligible to participate. The primary outcomes are overall survival over approximately 5 years and objective response rate. QoL is a secondary outcome. More details at clinicaltrials.gov.All trial information is from the National Institutes of Health US National Library of Medicine (online at clinicaltrials.gov).
A version of this article appeared on Medscape.com .
CMS Announces End to Cyberattack Relief Program
The Centers for Medicare & Medicaid Services (CMS) has announced the conclusion of a program that provided billions in early Medicare payments to those affected by the Change Healthcare/UnitedHealth Group cyberattack last winter.
CMS reported that the program advanced more than $2.55 billion in Medicare payments to > 4200 Part A providers, including hospitals, and more than $717.18 million in payments to Part B suppliers such as physicians, nonphysician practitioners, and durable medical equipment suppliers.
According to CMS, the Medicare billing system is now functioning properly, and 96% of the early payments have been recovered. The advances were to represent ≤ 30 days of typical claims payments in a 3-month period of 2023, with full repayment expected within 90 days through “automatic recoupment from Medicare claims” — no extensions allowed.
The agency took a victory lap regarding its response. “In the face of one of the most widespread cyberattacks on the US health care industry, CMS promptly took action to get providers and suppliers access to the funds they needed to continue providing patients with vital care,” CMS Administrator Chiquita Brooks-LaSure said in a statement. “Our efforts helped minimize the disruptive fallout from this incident, and we will remain vigilant to be ready to address future events.”
Ongoing Concerns from Health Care Organizations
Ben Teicher, an American Hospital Association spokesman, said that the organization hopes that CMS will be responsive if there’s more need for action after the advance payment program expires. The organization represents about 5000 hospitals, health care systems, and other providers.
“Our members report that the aftereffects of this event will likely be felt throughout the remainder of the year,” he said. According to Teicher, hospitals remain concerned about their ability to process claims and appeal denials, the safety of reconnecting to cyber services, and access to information needed to bill patients and reconcile payments.
In addition, hospitals are concerned about “financial support to mitigate the considerable costs incurred as a result of the cyberattack,” he said.
Charlene MacDonald, executive vice-president of public affairs at the Federation of American Hospitals, which represents more than 1000 for-profit hospitals, sent a statement to this news organization that said some providers “are still feeling the effects of care denials and delays caused by insurer inaction.
“We appreciate that the Administration acted within its authority to support providers during this unprecedented crisis and blunt these devastating impacts, especially because a vast majority of managed care companies failed to step up to the plate,” she said. “It is now time to shift our focus to holding plans accountable for using tactics to delay and deny needed patient care.”
Cyberattack Impact and Response
The ransom-based cyberattack against Change Healthcare/UnitedHealth Group targeted an electronic data interchange clearing house processing payer reimbursement systems, disrupting cash flows at hospitals and medical practices, and affecting patient access to prescriptions and life-saving therapy.
Change Healthcare — part of the UnitedHealth Group subsidiary Optum — processes half of all medical claims, according to a Department of Justice lawsuit. The American Hospital Association described the cyberattack as “the most significant and consequential incident of its kind” in US history.
By late March, UnitedHealth Group said nearly all medical and pharmacy claims were processing properly, while a deputy secretary of the US Department of Health & Human Services told clinicians that officials were focusing on the last group of clinicians who were facing cash-flow problems.
Still, a senior advisor with CMS told providers at that time that “we have heard from so many providers over the last several weeks who are really struggling to make ends meet right now or who are worried that they will not be able to make payroll in the weeks to come.”
Randy Dotinga is a freelance health/medical reporter and board member of the Association of Health Care Journalists.
A version of this article appeared on Medscape.com.
The Centers for Medicare & Medicaid Services (CMS) has announced the conclusion of a program that provided billions in early Medicare payments to those affected by the Change Healthcare/UnitedHealth Group cyberattack last winter.
CMS reported that the program advanced more than $2.55 billion in Medicare payments to > 4200 Part A providers, including hospitals, and more than $717.18 million in payments to Part B suppliers such as physicians, nonphysician practitioners, and durable medical equipment suppliers.
According to CMS, the Medicare billing system is now functioning properly, and 96% of the early payments have been recovered. The advances were to represent ≤ 30 days of typical claims payments in a 3-month period of 2023, with full repayment expected within 90 days through “automatic recoupment from Medicare claims” — no extensions allowed.
The agency took a victory lap regarding its response. “In the face of one of the most widespread cyberattacks on the US health care industry, CMS promptly took action to get providers and suppliers access to the funds they needed to continue providing patients with vital care,” CMS Administrator Chiquita Brooks-LaSure said in a statement. “Our efforts helped minimize the disruptive fallout from this incident, and we will remain vigilant to be ready to address future events.”
Ongoing Concerns from Health Care Organizations
Ben Teicher, an American Hospital Association spokesman, said that the organization hopes that CMS will be responsive if there’s more need for action after the advance payment program expires. The organization represents about 5000 hospitals, health care systems, and other providers.
“Our members report that the aftereffects of this event will likely be felt throughout the remainder of the year,” he said. According to Teicher, hospitals remain concerned about their ability to process claims and appeal denials, the safety of reconnecting to cyber services, and access to information needed to bill patients and reconcile payments.
In addition, hospitals are concerned about “financial support to mitigate the considerable costs incurred as a result of the cyberattack,” he said.
Charlene MacDonald, executive vice-president of public affairs at the Federation of American Hospitals, which represents more than 1000 for-profit hospitals, sent a statement to this news organization that said some providers “are still feeling the effects of care denials and delays caused by insurer inaction.
“We appreciate that the Administration acted within its authority to support providers during this unprecedented crisis and blunt these devastating impacts, especially because a vast majority of managed care companies failed to step up to the plate,” she said. “It is now time to shift our focus to holding plans accountable for using tactics to delay and deny needed patient care.”
Cyberattack Impact and Response
The ransom-based cyberattack against Change Healthcare/UnitedHealth Group targeted an electronic data interchange clearing house processing payer reimbursement systems, disrupting cash flows at hospitals and medical practices, and affecting patient access to prescriptions and life-saving therapy.
Change Healthcare — part of the UnitedHealth Group subsidiary Optum — processes half of all medical claims, according to a Department of Justice lawsuit. The American Hospital Association described the cyberattack as “the most significant and consequential incident of its kind” in US history.
By late March, UnitedHealth Group said nearly all medical and pharmacy claims were processing properly, while a deputy secretary of the US Department of Health & Human Services told clinicians that officials were focusing on the last group of clinicians who were facing cash-flow problems.
Still, a senior advisor with CMS told providers at that time that “we have heard from so many providers over the last several weeks who are really struggling to make ends meet right now or who are worried that they will not be able to make payroll in the weeks to come.”
Randy Dotinga is a freelance health/medical reporter and board member of the Association of Health Care Journalists.
A version of this article appeared on Medscape.com.
The Centers for Medicare & Medicaid Services (CMS) has announced the conclusion of a program that provided billions in early Medicare payments to those affected by the Change Healthcare/UnitedHealth Group cyberattack last winter.
CMS reported that the program advanced more than $2.55 billion in Medicare payments to > 4200 Part A providers, including hospitals, and more than $717.18 million in payments to Part B suppliers such as physicians, nonphysician practitioners, and durable medical equipment suppliers.
According to CMS, the Medicare billing system is now functioning properly, and 96% of the early payments have been recovered. The advances were to represent ≤ 30 days of typical claims payments in a 3-month period of 2023, with full repayment expected within 90 days through “automatic recoupment from Medicare claims” — no extensions allowed.
The agency took a victory lap regarding its response. “In the face of one of the most widespread cyberattacks on the US health care industry, CMS promptly took action to get providers and suppliers access to the funds they needed to continue providing patients with vital care,” CMS Administrator Chiquita Brooks-LaSure said in a statement. “Our efforts helped minimize the disruptive fallout from this incident, and we will remain vigilant to be ready to address future events.”
Ongoing Concerns from Health Care Organizations
Ben Teicher, an American Hospital Association spokesman, said that the organization hopes that CMS will be responsive if there’s more need for action after the advance payment program expires. The organization represents about 5000 hospitals, health care systems, and other providers.
“Our members report that the aftereffects of this event will likely be felt throughout the remainder of the year,” he said. According to Teicher, hospitals remain concerned about their ability to process claims and appeal denials, the safety of reconnecting to cyber services, and access to information needed to bill patients and reconcile payments.
In addition, hospitals are concerned about “financial support to mitigate the considerable costs incurred as a result of the cyberattack,” he said.
Charlene MacDonald, executive vice-president of public affairs at the Federation of American Hospitals, which represents more than 1000 for-profit hospitals, sent a statement to this news organization that said some providers “are still feeling the effects of care denials and delays caused by insurer inaction.
“We appreciate that the Administration acted within its authority to support providers during this unprecedented crisis and blunt these devastating impacts, especially because a vast majority of managed care companies failed to step up to the plate,” she said. “It is now time to shift our focus to holding plans accountable for using tactics to delay and deny needed patient care.”
Cyberattack Impact and Response
The ransom-based cyberattack against Change Healthcare/UnitedHealth Group targeted an electronic data interchange clearing house processing payer reimbursement systems, disrupting cash flows at hospitals and medical practices, and affecting patient access to prescriptions and life-saving therapy.
Change Healthcare — part of the UnitedHealth Group subsidiary Optum — processes half of all medical claims, according to a Department of Justice lawsuit. The American Hospital Association described the cyberattack as “the most significant and consequential incident of its kind” in US history.
By late March, UnitedHealth Group said nearly all medical and pharmacy claims were processing properly, while a deputy secretary of the US Department of Health & Human Services told clinicians that officials were focusing on the last group of clinicians who were facing cash-flow problems.
Still, a senior advisor with CMS told providers at that time that “we have heard from so many providers over the last several weeks who are really struggling to make ends meet right now or who are worried that they will not be able to make payroll in the weeks to come.”
Randy Dotinga is a freelance health/medical reporter and board member of the Association of Health Care Journalists.
A version of this article appeared on Medscape.com.
Online Tool Predicts Real-World Driving Ability of Older Drivers
An algorithm using two well-known tests has shown strong accuracy (91%) in predicting whether an older driver can pass an on-road driving evaluation according to a new study published in the Journal of the American Medical Directors Association .
The Fit2Drive algorithm combines the Mini-Mental State Exam (MMSE), a 30-point dementia screening tool that has been found in several studies to have an association with driving ability, and the Trails B test, which gauges cognitive flexibility and set-shifting (task switching), considered to be measures of executive functioning.
Algorithm Available for Providers
The algorithm is clinically available and providers can fill in patients’ information and results of the two tests at the Fit2Drive website. Results may help physicians with often-difficult conversations with older patients about driving when they present with cognitive concerns.
Families report it is one of the most difficult conversations they have with a loved one and doctors are often asked to be part of the conversation. This is particularly difficult when, often, little objective information is available. In the past, a clinical rule of thumb has been that people diagnosed with Alzheimer’s disease or related dementias (ADRD) will usually be able to drive for 3 years after diagnosis.
“[T]he anger, tears, and frustration on the part of the individual patient and the lack of objective data to guide provider recommendations are the driving forces behind our effort to develop a highly accurate, evidence-based predictor of the ability to pass an on-road driving test,” the authors write. They added that the goal of the study was to identify the smallest number of cognitive test results that could predict likelihood of passing an on-road driver evaluation.
A number of tests were evaluated for the algorithm, but the combination of Trails B in seconds and MMSE using the highest scores of the serial 7s (counting back from 100 by 7s) or WORLD spelled backward accounted for the highest correlation with passing the on-road driving test, according to the authors, led by Ruth Tappen, EdD, FN, with the Christine E. Lynn College of Nursing at Florida Atlantic University, in Boca Raton.
A receiver operator characteristic (ROC) analysis was conducted on the linear combination of the two assessments.
“Because an ROC of 0.70 is considered to be the minimal requirement [for predictive value], 0.80 is considered good, and higher than 0.90 is excellent, these findings [with 91% area under the curve] suggest excellent accuracy using these two cognitive tests in this population,” the authors write.
For this analysis, researchers included 412 older drivers (179 men and 233 women) with an average age of 80. T he study was conducted at the Florida Atlantic University’s Memory Center and Clinical Research Unit. Participants included those who received a driving evaluation at the Memory Center and agreed to have their results included in the Driving Repository, and community-based older drivers who volunteered to participate.
Limitations of the Study
There were marginal differences between sexes on the measures, but they were not significant. The sample was composed of relatively well-educated people, primarily of European American ethnic origin, which is a consideration in generalizing the results.
Among other limitations are that physical and sensory factors, in addition to cognitive issues, may affect an individual’s ability to drive safely and are not included in the algorithm. Sensory disabilities, including reduced visual acuity caused by binocular field vision loss, contrast sensitivity, glare sensitivity, and other conditions, may affect driving ability as well as the ability to fully rotate the head and neck. Medical conditions affecting the cardiovascular, neurological, and orthopedic systems can also influence driving ability.
“Future studies should involve more diverse samples and a greater variety of driving challenges, including school zones and multilane highways, which are not included in the study,” the authors write.
The study received grant support from the State of Florida Department of Health and the Ed and Ethel Moore Alzheimer’s Disease Research Program.
An algorithm using two well-known tests has shown strong accuracy (91%) in predicting whether an older driver can pass an on-road driving evaluation according to a new study published in the Journal of the American Medical Directors Association .
The Fit2Drive algorithm combines the Mini-Mental State Exam (MMSE), a 30-point dementia screening tool that has been found in several studies to have an association with driving ability, and the Trails B test, which gauges cognitive flexibility and set-shifting (task switching), considered to be measures of executive functioning.
Algorithm Available for Providers
The algorithm is clinically available and providers can fill in patients’ information and results of the two tests at the Fit2Drive website. Results may help physicians with often-difficult conversations with older patients about driving when they present with cognitive concerns.
Families report it is one of the most difficult conversations they have with a loved one and doctors are often asked to be part of the conversation. This is particularly difficult when, often, little objective information is available. In the past, a clinical rule of thumb has been that people diagnosed with Alzheimer’s disease or related dementias (ADRD) will usually be able to drive for 3 years after diagnosis.
“[T]he anger, tears, and frustration on the part of the individual patient and the lack of objective data to guide provider recommendations are the driving forces behind our effort to develop a highly accurate, evidence-based predictor of the ability to pass an on-road driving test,” the authors write. They added that the goal of the study was to identify the smallest number of cognitive test results that could predict likelihood of passing an on-road driver evaluation.
A number of tests were evaluated for the algorithm, but the combination of Trails B in seconds and MMSE using the highest scores of the serial 7s (counting back from 100 by 7s) or WORLD spelled backward accounted for the highest correlation with passing the on-road driving test, according to the authors, led by Ruth Tappen, EdD, FN, with the Christine E. Lynn College of Nursing at Florida Atlantic University, in Boca Raton.
A receiver operator characteristic (ROC) analysis was conducted on the linear combination of the two assessments.
“Because an ROC of 0.70 is considered to be the minimal requirement [for predictive value], 0.80 is considered good, and higher than 0.90 is excellent, these findings [with 91% area under the curve] suggest excellent accuracy using these two cognitive tests in this population,” the authors write.
For this analysis, researchers included 412 older drivers (179 men and 233 women) with an average age of 80. T he study was conducted at the Florida Atlantic University’s Memory Center and Clinical Research Unit. Participants included those who received a driving evaluation at the Memory Center and agreed to have their results included in the Driving Repository, and community-based older drivers who volunteered to participate.
Limitations of the Study
There were marginal differences between sexes on the measures, but they were not significant. The sample was composed of relatively well-educated people, primarily of European American ethnic origin, which is a consideration in generalizing the results.
Among other limitations are that physical and sensory factors, in addition to cognitive issues, may affect an individual’s ability to drive safely and are not included in the algorithm. Sensory disabilities, including reduced visual acuity caused by binocular field vision loss, contrast sensitivity, glare sensitivity, and other conditions, may affect driving ability as well as the ability to fully rotate the head and neck. Medical conditions affecting the cardiovascular, neurological, and orthopedic systems can also influence driving ability.
“Future studies should involve more diverse samples and a greater variety of driving challenges, including school zones and multilane highways, which are not included in the study,” the authors write.
The study received grant support from the State of Florida Department of Health and the Ed and Ethel Moore Alzheimer’s Disease Research Program.
An algorithm using two well-known tests has shown strong accuracy (91%) in predicting whether an older driver can pass an on-road driving evaluation according to a new study published in the Journal of the American Medical Directors Association .
The Fit2Drive algorithm combines the Mini-Mental State Exam (MMSE), a 30-point dementia screening tool that has been found in several studies to have an association with driving ability, and the Trails B test, which gauges cognitive flexibility and set-shifting (task switching), considered to be measures of executive functioning.
Algorithm Available for Providers
The algorithm is clinically available and providers can fill in patients’ information and results of the two tests at the Fit2Drive website. Results may help physicians with often-difficult conversations with older patients about driving when they present with cognitive concerns.
Families report it is one of the most difficult conversations they have with a loved one and doctors are often asked to be part of the conversation. This is particularly difficult when, often, little objective information is available. In the past, a clinical rule of thumb has been that people diagnosed with Alzheimer’s disease or related dementias (ADRD) will usually be able to drive for 3 years after diagnosis.
“[T]he anger, tears, and frustration on the part of the individual patient and the lack of objective data to guide provider recommendations are the driving forces behind our effort to develop a highly accurate, evidence-based predictor of the ability to pass an on-road driving test,” the authors write. They added that the goal of the study was to identify the smallest number of cognitive test results that could predict likelihood of passing an on-road driver evaluation.
A number of tests were evaluated for the algorithm, but the combination of Trails B in seconds and MMSE using the highest scores of the serial 7s (counting back from 100 by 7s) or WORLD spelled backward accounted for the highest correlation with passing the on-road driving test, according to the authors, led by Ruth Tappen, EdD, FN, with the Christine E. Lynn College of Nursing at Florida Atlantic University, in Boca Raton.
A receiver operator characteristic (ROC) analysis was conducted on the linear combination of the two assessments.
“Because an ROC of 0.70 is considered to be the minimal requirement [for predictive value], 0.80 is considered good, and higher than 0.90 is excellent, these findings [with 91% area under the curve] suggest excellent accuracy using these two cognitive tests in this population,” the authors write.
For this analysis, researchers included 412 older drivers (179 men and 233 women) with an average age of 80. T he study was conducted at the Florida Atlantic University’s Memory Center and Clinical Research Unit. Participants included those who received a driving evaluation at the Memory Center and agreed to have their results included in the Driving Repository, and community-based older drivers who volunteered to participate.
Limitations of the Study
There were marginal differences between sexes on the measures, but they were not significant. The sample was composed of relatively well-educated people, primarily of European American ethnic origin, which is a consideration in generalizing the results.
Among other limitations are that physical and sensory factors, in addition to cognitive issues, may affect an individual’s ability to drive safely and are not included in the algorithm. Sensory disabilities, including reduced visual acuity caused by binocular field vision loss, contrast sensitivity, glare sensitivity, and other conditions, may affect driving ability as well as the ability to fully rotate the head and neck. Medical conditions affecting the cardiovascular, neurological, and orthopedic systems can also influence driving ability.
“Future studies should involve more diverse samples and a greater variety of driving challenges, including school zones and multilane highways, which are not included in the study,” the authors write.
The study received grant support from the State of Florida Department of Health and the Ed and Ethel Moore Alzheimer’s Disease Research Program.
FROM JAMDA
Compounded Semaglutide: How to Better Ensure Its Safety
Glucagon-like peptide 1 (GLP-1) receptor agonists such as semaglutide (marketed as Ozempic and Rybelsus for type 2 diabetes and as Wegovy for obesity) slow down digestion and curb hunger by working on the brain’s dopamine reward center. They are prescribed to promote weight loss, metabolic health in type 2 diabetes, and heart health in coronary artery disease.
Semaglutide can be prescribed in two forms: the brand-name version, which is approved and confirmed as safe and effective by the US Food and Drug Administration (FDA), and the versions that can be obtained from a compounding pharmacy. Compounding pharmacies are permitted by the FDA to produce what is “ essentially a copy” of approved medications when there’s an official shortage, which is currently the case with semaglutide and other GLP-1 receptor agonists.
Patients are often drawn to compounding pharmacies for pricing-related reasons. If semaglutide is prescribed for a clear indication like diabetes and is covered by insurance, the brand-name version is commonly dispensed. However, if it’s not covered, patients need to pay out of pocket for branded versions, which carry a monthly cost of $1000 or more. Alternatively, their doctors can prescribe compounded semaglutide, which some telehealth companies advertise at costs of approximately $150-$300 per month.
Potential Issues With Compounded Semaglutide
Compounding pharmacies produce drugs from raw materials containing active pharmaceutical ingredients (APIs). Although compounders use many of the same ingredients found in brand-name medications, for drugs like semaglutide, they may opt for specific salts that are not identical to those involved in the production of the standard versions. These salts are typically reserved for research purposes and may not be suitable for general use.
In late 2023, the FDA issued a letter asking the public to exercise caution when using compounded products containing semaglutide or semaglutide salts. This was followed in January 2024 by an FDA communication citing adverse events reported with the use of compounded semaglutide and advising patients to avoid these versions if an approved form of the drug is available.
Compound Pharmacies: A Closer Look
Compounding pharmacies have exploded in popularity in the past several decades. The compounding pharmacy market is expected to grow at 7.8% per year over the next decade.
Historically, compounding pharmacies have filled a niche for specialty vitamins for intravenous administration as well as chemotherapy medications. They also offer controlled substances, such as ketamine lozenges and nasal sprays, which are unavailable or are in short supply from traditional manufacturers.
Compounding pharmacies fall into two categories. First are compounding pharmacies covered under Section 503A of the Federal Food, Drug and Cosmetic Act; these drugs are neither tested nor monitored. Such facilities do not have to report adverse events to the FDA. The second category is Section 503B outsourcing facilities. These pharmacies choose to be tested by, to be inspected by, and to report adverse events to the FDA.
The FDA’s Latest Update on This Issue
This news organization contacted the FDA for an update on the adverse events reported about compounded semaglutide. From August 8, 2021, to March 31, 2024, they received more than 20,000 adverse events reports for FDA-approved semaglutide. Comparatively, there were 210 adverse events reported on compounded semaglutide products.
The FDA went on to describe that many of the adverse events reported were consistent with known reactions in the labeling, like nausea, diarrhea, and headache. Yet, they added that, “the FDA is unable to determine how, or if, other factors may have contributed to these adverse events, such as differences in ingredients and formulation between FDA-approved and compounded semaglutide products.” They also noted there was variation in the data quality in the reports they have received, which came only from 503B compounding pharmacies.
In conclusion, given the concerns about compounded semaglutide, it is prudent for the prescribing physicians as well as the patients taking the medication to know that risks are “higher” according to the FDA. We eagerly await more specific information from the FDA to better understand reported adverse events.
How to Help Patients Receive Safe Compounded Semaglutide
For clinicians considering prescribing semaglutide from compounding pharmacies, there are several questions worth asking, according to the Alliance for Pharmacy Compounding. First, find out whether the pharmacy complies with United States Pharmacopeia compounding standards and whether they source their APIs from FDA-registered facilities, the latter being required by federal law. It’s also important to ensure that these facilities undergo periodic third-party testing to verify medication purity and dosing.
Ask whether the pharmacy is accredited by the Pharmacy Compounding Accreditation Board (PCAB). Accreditation from the PCAB means that pharmacies have been assessed for processes related to continuous quality improvement. In addition, ask whether the pharmacy is designated as a 503B compounder and if not, why.
Finally, interviewing the pharmacist themselves can provide useful information about staffing, training, and their methods of preparing medications. For example, if they are preparing a sterile eye drop, it is important to ask about sterility testing.
Jesse M. Pines, MD, MBA, MSCE, is a clinical professor of emergency medicine at George Washington University in Washington, and a professor in the department of emergency medicine at Drexel University College of Medicine in Philadelphia, Pennsylvania. Dr. Pines is also the chief of clinical innovation at US Acute Care Solutions in Canton, Ohio. Robert D. Glatter, MD, is an assistant professor of emergency medicine at Zucker School of Medicine at Hofstra/Northwell in Hempstead, New York. Dr. Pines reported conflicts of interest with CSL Behring and Abbott Point-of-Care. Dr. Glatter reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Glucagon-like peptide 1 (GLP-1) receptor agonists such as semaglutide (marketed as Ozempic and Rybelsus for type 2 diabetes and as Wegovy for obesity) slow down digestion and curb hunger by working on the brain’s dopamine reward center. They are prescribed to promote weight loss, metabolic health in type 2 diabetes, and heart health in coronary artery disease.
Semaglutide can be prescribed in two forms: the brand-name version, which is approved and confirmed as safe and effective by the US Food and Drug Administration (FDA), and the versions that can be obtained from a compounding pharmacy. Compounding pharmacies are permitted by the FDA to produce what is “ essentially a copy” of approved medications when there’s an official shortage, which is currently the case with semaglutide and other GLP-1 receptor agonists.
Patients are often drawn to compounding pharmacies for pricing-related reasons. If semaglutide is prescribed for a clear indication like diabetes and is covered by insurance, the brand-name version is commonly dispensed. However, if it’s not covered, patients need to pay out of pocket for branded versions, which carry a monthly cost of $1000 or more. Alternatively, their doctors can prescribe compounded semaglutide, which some telehealth companies advertise at costs of approximately $150-$300 per month.
Potential Issues With Compounded Semaglutide
Compounding pharmacies produce drugs from raw materials containing active pharmaceutical ingredients (APIs). Although compounders use many of the same ingredients found in brand-name medications, for drugs like semaglutide, they may opt for specific salts that are not identical to those involved in the production of the standard versions. These salts are typically reserved for research purposes and may not be suitable for general use.
In late 2023, the FDA issued a letter asking the public to exercise caution when using compounded products containing semaglutide or semaglutide salts. This was followed in January 2024 by an FDA communication citing adverse events reported with the use of compounded semaglutide and advising patients to avoid these versions if an approved form of the drug is available.
Compound Pharmacies: A Closer Look
Compounding pharmacies have exploded in popularity in the past several decades. The compounding pharmacy market is expected to grow at 7.8% per year over the next decade.
Historically, compounding pharmacies have filled a niche for specialty vitamins for intravenous administration as well as chemotherapy medications. They also offer controlled substances, such as ketamine lozenges and nasal sprays, which are unavailable or are in short supply from traditional manufacturers.
Compounding pharmacies fall into two categories. First are compounding pharmacies covered under Section 503A of the Federal Food, Drug and Cosmetic Act; these drugs are neither tested nor monitored. Such facilities do not have to report adverse events to the FDA. The second category is Section 503B outsourcing facilities. These pharmacies choose to be tested by, to be inspected by, and to report adverse events to the FDA.
The FDA’s Latest Update on This Issue
This news organization contacted the FDA for an update on the adverse events reported about compounded semaglutide. From August 8, 2021, to March 31, 2024, they received more than 20,000 adverse events reports for FDA-approved semaglutide. Comparatively, there were 210 adverse events reported on compounded semaglutide products.
The FDA went on to describe that many of the adverse events reported were consistent with known reactions in the labeling, like nausea, diarrhea, and headache. Yet, they added that, “the FDA is unable to determine how, or if, other factors may have contributed to these adverse events, such as differences in ingredients and formulation between FDA-approved and compounded semaglutide products.” They also noted there was variation in the data quality in the reports they have received, which came only from 503B compounding pharmacies.
In conclusion, given the concerns about compounded semaglutide, it is prudent for the prescribing physicians as well as the patients taking the medication to know that risks are “higher” according to the FDA. We eagerly await more specific information from the FDA to better understand reported adverse events.
How to Help Patients Receive Safe Compounded Semaglutide
For clinicians considering prescribing semaglutide from compounding pharmacies, there are several questions worth asking, according to the Alliance for Pharmacy Compounding. First, find out whether the pharmacy complies with United States Pharmacopeia compounding standards and whether they source their APIs from FDA-registered facilities, the latter being required by federal law. It’s also important to ensure that these facilities undergo periodic third-party testing to verify medication purity and dosing.
Ask whether the pharmacy is accredited by the Pharmacy Compounding Accreditation Board (PCAB). Accreditation from the PCAB means that pharmacies have been assessed for processes related to continuous quality improvement. In addition, ask whether the pharmacy is designated as a 503B compounder and if not, why.
Finally, interviewing the pharmacist themselves can provide useful information about staffing, training, and their methods of preparing medications. For example, if they are preparing a sterile eye drop, it is important to ask about sterility testing.
Jesse M. Pines, MD, MBA, MSCE, is a clinical professor of emergency medicine at George Washington University in Washington, and a professor in the department of emergency medicine at Drexel University College of Medicine in Philadelphia, Pennsylvania. Dr. Pines is also the chief of clinical innovation at US Acute Care Solutions in Canton, Ohio. Robert D. Glatter, MD, is an assistant professor of emergency medicine at Zucker School of Medicine at Hofstra/Northwell in Hempstead, New York. Dr. Pines reported conflicts of interest with CSL Behring and Abbott Point-of-Care. Dr. Glatter reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Glucagon-like peptide 1 (GLP-1) receptor agonists such as semaglutide (marketed as Ozempic and Rybelsus for type 2 diabetes and as Wegovy for obesity) slow down digestion and curb hunger by working on the brain’s dopamine reward center. They are prescribed to promote weight loss, metabolic health in type 2 diabetes, and heart health in coronary artery disease.
Semaglutide can be prescribed in two forms: the brand-name version, which is approved and confirmed as safe and effective by the US Food and Drug Administration (FDA), and the versions that can be obtained from a compounding pharmacy. Compounding pharmacies are permitted by the FDA to produce what is “ essentially a copy” of approved medications when there’s an official shortage, which is currently the case with semaglutide and other GLP-1 receptor agonists.
Patients are often drawn to compounding pharmacies for pricing-related reasons. If semaglutide is prescribed for a clear indication like diabetes and is covered by insurance, the brand-name version is commonly dispensed. However, if it’s not covered, patients need to pay out of pocket for branded versions, which carry a monthly cost of $1000 or more. Alternatively, their doctors can prescribe compounded semaglutide, which some telehealth companies advertise at costs of approximately $150-$300 per month.
Potential Issues With Compounded Semaglutide
Compounding pharmacies produce drugs from raw materials containing active pharmaceutical ingredients (APIs). Although compounders use many of the same ingredients found in brand-name medications, for drugs like semaglutide, they may opt for specific salts that are not identical to those involved in the production of the standard versions. These salts are typically reserved for research purposes and may not be suitable for general use.
In late 2023, the FDA issued a letter asking the public to exercise caution when using compounded products containing semaglutide or semaglutide salts. This was followed in January 2024 by an FDA communication citing adverse events reported with the use of compounded semaglutide and advising patients to avoid these versions if an approved form of the drug is available.
Compound Pharmacies: A Closer Look
Compounding pharmacies have exploded in popularity in the past several decades. The compounding pharmacy market is expected to grow at 7.8% per year over the next decade.
Historically, compounding pharmacies have filled a niche for specialty vitamins for intravenous administration as well as chemotherapy medications. They also offer controlled substances, such as ketamine lozenges and nasal sprays, which are unavailable or are in short supply from traditional manufacturers.
Compounding pharmacies fall into two categories. First are compounding pharmacies covered under Section 503A of the Federal Food, Drug and Cosmetic Act; these drugs are neither tested nor monitored. Such facilities do not have to report adverse events to the FDA. The second category is Section 503B outsourcing facilities. These pharmacies choose to be tested by, to be inspected by, and to report adverse events to the FDA.
The FDA’s Latest Update on This Issue
This news organization contacted the FDA for an update on the adverse events reported about compounded semaglutide. From August 8, 2021, to March 31, 2024, they received more than 20,000 adverse events reports for FDA-approved semaglutide. Comparatively, there were 210 adverse events reported on compounded semaglutide products.
The FDA went on to describe that many of the adverse events reported were consistent with known reactions in the labeling, like nausea, diarrhea, and headache. Yet, they added that, “the FDA is unable to determine how, or if, other factors may have contributed to these adverse events, such as differences in ingredients and formulation between FDA-approved and compounded semaglutide products.” They also noted there was variation in the data quality in the reports they have received, which came only from 503B compounding pharmacies.
In conclusion, given the concerns about compounded semaglutide, it is prudent for the prescribing physicians as well as the patients taking the medication to know that risks are “higher” according to the FDA. We eagerly await more specific information from the FDA to better understand reported adverse events.
How to Help Patients Receive Safe Compounded Semaglutide
For clinicians considering prescribing semaglutide from compounding pharmacies, there are several questions worth asking, according to the Alliance for Pharmacy Compounding. First, find out whether the pharmacy complies with United States Pharmacopeia compounding standards and whether they source their APIs from FDA-registered facilities, the latter being required by federal law. It’s also important to ensure that these facilities undergo periodic third-party testing to verify medication purity and dosing.
Ask whether the pharmacy is accredited by the Pharmacy Compounding Accreditation Board (PCAB). Accreditation from the PCAB means that pharmacies have been assessed for processes related to continuous quality improvement. In addition, ask whether the pharmacy is designated as a 503B compounder and if not, why.
Finally, interviewing the pharmacist themselves can provide useful information about staffing, training, and their methods of preparing medications. For example, if they are preparing a sterile eye drop, it is important to ask about sterility testing.
Jesse M. Pines, MD, MBA, MSCE, is a clinical professor of emergency medicine at George Washington University in Washington, and a professor in the department of emergency medicine at Drexel University College of Medicine in Philadelphia, Pennsylvania. Dr. Pines is also the chief of clinical innovation at US Acute Care Solutions in Canton, Ohio. Robert D. Glatter, MD, is an assistant professor of emergency medicine at Zucker School of Medicine at Hofstra/Northwell in Hempstead, New York. Dr. Pines reported conflicts of interest with CSL Behring and Abbott Point-of-Care. Dr. Glatter reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Is This Journal Legit? Predatory Publishers
This transcript has been edited for clarity.
Andrew N. Wilner, MD: My guest today is Dr. Jose Merino, editor in chief of the Neurology family of journals and professor of neurology and co-vice chair of education at Georgetown University in Washington, DC.
Our program today is a follow-up of Dr. Merino’s presentation at the recent American Academy of Neurology meeting in Denver, Colorado. Along with two other panelists, Dr. Merino discussed the role of open-access publication and the dangers of predatory journals.
Jose G. Merino, MD, MPhil: Thank you for having me here. It’s a pleasure.
Open Access Defined
Dr. Wilner: I remember when publication in neurology was pretty straightforward. It was either the green journal or the blue journal, but things have certainly changed. I think one topic that is not clear to everyone is this concept of open access. Could you define that for us?
Dr. Merino: Sure. Open access is a mode of publication that fosters more open or accessible science. The idea of open access is that it combines two main elements. One is that the papers that are published become immediately available to anybody with an internet connection anywhere in the world without any restrictions.
The second important element from open access, which makes it different from other models we can talk about, is the fact that the authors retain the copyright of their work, but they give the journal and readers a license to use, reproduce, and modify the content.
This is different, for example, from instances where we have funder mandates. For example, NIH papers have to become available 6 months after publication, so they’re available to everybody but not immediately.
Dr. Wilner: I remember that when a journal article was published, say, in Neurology, if you didn’t have a subscription to Neurology, you went to the library that hopefully had a subscription.
If they didn’t have it, you would write to the author and say, “Hey, I heard you have this great paper because the abstract was out there. Could you send me a reprint?” Has that whole universe evaporated?
Dr. Merino: It depends on how the paper is published. For example, in Neurology, some of the research we publish is open access. Basically, if you have an internet connection, you can access the paper.
That’s the case for papers published in our wholly open-access journals in the Neurology family like Neurology Neuroimmunology & Neuroinflammation, Neurology Genetics, or Neurology Education.
For other papers that are published in Neurology, not under open access, there is a paywall. For some of them, the paywall comes down after a few months based on funder mandates and so on. As I was mentioning, the NIH-funded papers are available 6 months later.
In the first 6 months, you may have to go to your library, and if your library has a subscription, you can download it directly. [This is also true for] those that always stay behind the paywall, where you have to have a subscription or your library has to have a subscription.
Is Pay to Publish a Red Flag?
Dr. Wilner: I’m a professional writer. With any luck, when I write something, I get paid to write it. There’s been a long tradition in academic medicine that when you submit an article to, say, Neurology, you don’t get paid as an author for the publication. Your reward is the honor of it being published.
Neurology supports itself in various ways, including advertising and so on. That’s been the contract: free publication for work that merits it, and the journal survives on its own.
With open access, one of the things that’s happened is that — and I’ve published open access myself — is that I get a notification that I need to pay to have my article that I’ve slaved over published. Explain that, please.
Dr. Merino: This is the issue with open access. As I mentioned, the paper gets published. You’re giving the journal a license to publish it. You’re retaining the copyright of your work. That means that the journal cannot make money or support itself by just publishing open access because they belong to you.
Typically, open-access journals are not in print and don’t have much in terms of advertising. The contract is you’re giving me a license to publish it, but it’s your journal, so you’re paying a fee for the journal expenses to basically produce your paper. That’s what’s happening with open access.
That’s been recognized with many funders, for example, with NIH funding or many of the European funders, they’re including open-access fees as part of their funding for research. Now, of course, this doesn’t help if you’re not a funded researcher or if you’re a fellow who’s doing work and so on.
Typically, most journals will have waived fees or lower fees for these situations. The reason for the open-access fee is the fact that you’re retaining the copyright. You’re not giving it to the journal who can then use it to generate its revenue for supporting itself, the editorial staff, and so on.
Dr. Wilner: This idea of charging for publication has created a satellite business of what are called predatory journals. How does one know if the open-access journal that I’m submitting to is really just in the business of wanting my $300 or my $900 to get published? How do I know if that’s a reasonable place to publish?
Predatory Journals
Dr. Merino: That’s a big challenge that has come with this whole idea of open access and the fact that now, many journals are online only, so you’re no longer seeing a physical copy. That has given rise to the predatory journals.
The predatory journal, by definition, is a journal that claims to be open access. They’ll take your paper and publish it, but they don’t provide all the other services that you would typically expect from the fact that you’re paying an open-access fee. This includes getting appropriate peer review, production of the manuscript, and long-term curation and storage of the manuscript.
Many predatory journals will take your open-access fee, accept any paper that you submit, regardless of the quality, because they’re charging the fees for that. They don’t send it to real peer review, and then in a few months, the journal disappears so there’s no way for anybody to actually find your paper anymore.
There are certain checklists. Dr. David Moher at the University of Toronto has produced some work trying to help us identify predatory journals.
One thing I typically suggest to people who ask me this question is: Have you ever heard of this journal before? Does the journal have a track record? How far back does the story of the journal go? Is it supported by a publisher that you know? Do you know anybody who has published there? Is it something you can easily access?
If in doubt, always ask your friendly medical librarian. There used to be lists that were kept in terms of predatory journals that were being constantly updated, but those had to be shut down. As far as I understand, there were legal issues in terms of how things got on that list.
I think that overall, if you’ve heard of it, if it’s relevant, if it’s known in your field, and if your librarian knows it, it’s probably a good legitimate open-access journal. There are many very good legitimate open-access journals.
I mentioned the two that we have in our family, but all the other major journals have their own open-access journal within their family. There are some, like BMC or PLOS, that are completely open-access and legitimate journals.
Impact Factor
Dr. Wilner: What about impact factor? Many journals boast about their impact factor. I’m not sure how to interpret that number.
Dr. Merino: Impact factor is very interesting. The impact factor was developed by medical librarians to try to identify the journals they should be subscribing to. It’s a measure of the average citations to an average paper in the journal.
It doesn’t tell you about specific papers. It tells you, on average, how many of the papers in this journal get cited so many times. It’s calculated by the number of articles that were cited divided by the number of articles that were published. Journals that publish many papers, like Neurology, have a hard time bringing up their impact factor beyond a certain level.
Similarly, very small journals with one or two very highly cited papers have a very high impact factor. It’s being used as a measure, perhaps inappropriately, of how good or how reputable a journal is. We all say we don’t care about journal impact factors, but we all know our journal impact factor and we used to know it to three decimals. Now, they changed the system, and there’s only one decimal point, which makes more sense.
This is more important, for example, for authors when deciding where to submit papers. I know that in some countries, particularly in Europe, the impact factor of the journal where you publish has an impact on your promotion decisions.
I would say what’s even more important than the impact factor, is to say, “Well, is this the journal that fits the scope of my paper? Is this the journal that reaches the audience that I want to reach when I write my paper?”
There are some papers, for example, that are very influential. The impact factor just captures citations. There are some papers that are very influential that may not get cited very often. There may be papers that change clinical practice.
If you read a paper that tells you that you should be changing how you treat your patients with myasthenia based on this paper, that may not get cited. It’s a very clinically focused paper, but it’s probably more impactful than one that gets cited very much in some respect, or they make it to public policy decisions, and so on.
I think it’s important to look more at the audience and the journal scope when you submit your papers.
Dr. Wilner: One other technical question. The journals also say they’re indexed in PubMed or Google Scholar. If I want to publish my paper and I want it indexed where the right people are going to find it, where does it need to be indexed?
Dr. Merino: I grew up using Index Medicus, MedlinePlus, and the Library of Science. I still do. If I need to find something, I go to PubMed. Ideally, papers are listed in MedlinePlus or can be found in PubMed. They’re not the same thing, but you can find them through them.
That would be an important thing. Nowadays, a lot more people are using Google Scholar or Google just to identify papers. It may be a little bit less relevant, but it’s still a measure of the quality of the journal before they get indexed in some of these. For example, if you get listed in MedlinePlus, it has gone through certain quality checks by the index itself to see whether they would accept the journal or not. That’s something you want to check.
Typically, most of the large journals or the journals you and I know about are listed in more than one place, right? They’re listed in Scopus and Web of Science. They’re listed in MedlinePlus and so on. Again, if you’re submitting your paper, go somewhere where you know the journal and you’ve heard about it.
Dr. Wilner: I’m not going to ask you about artificial intelligence. We can do that another time. I want to ask something closer to me, which is this question of publish or perish.
There seems to be, in academics, more emphasis on the number of papers that one has published rather than their quality. How does a younger academician or one who really needs to publish cope with that?
Dr. Merino: Many people are writing up research that may not be relevant or that may not be high quality just because you need to have a long list of papers to get promoted, for example, if you’re an academician.
Doug Altman, who was a very influential person in the field quality of not only medical statistics but also medical publishing, had the idea that we need less research, but we need better research.
We often receive papers where you say, well, what’s the rationale behind the question in this paper? It’s like they had a large amount of data and were trying to squeeze as much as they could out of that. I think, as a young academician, the important thing to think about is whether it is an important question that matters to you and to the field, from whatever perspective, whether it’s going to advance research, advance clinical care, or have public policy implications.
Is this one where the answer will be important no matter what the answer is? If you’re thinking of that, your work will be well recognized, people will know you, and you’ll get invited to collaborate. I think that’s the most important thing rather than just churning out a large number of papers.
The productivity will come from the fact that you start by saying, let me ask something that’s really meaningful to me and to the field, with a good question and using strong research methodology.
Dr. Wilner: Thanks for that, Dr. Merino. I think that’s very valuable for all of us. This has been a great discussion. Do you have any final comments before we wrap up?
Dr. Merino: I want to encourage people to continue reading medical journals all the time and submitting to us, again, good research and important questions with robust methodology. That’s what we’re looking for in Neurology and most serious medical journals.
Dr. Wilner is an associate professor of neurology at the University of Tennessee Health Science Center, Memphis. Dr. Merino is a professor in the department of neurology at Georgetown University Medical Center, Washington, DC. Dr. Wilner reported conflicts of interest with Accordant Health Services and Lulu Publishing. Dr. Merino reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Andrew N. Wilner, MD: My guest today is Dr. Jose Merino, editor in chief of the Neurology family of journals and professor of neurology and co-vice chair of education at Georgetown University in Washington, DC.
Our program today is a follow-up of Dr. Merino’s presentation at the recent American Academy of Neurology meeting in Denver, Colorado. Along with two other panelists, Dr. Merino discussed the role of open-access publication and the dangers of predatory journals.
Jose G. Merino, MD, MPhil: Thank you for having me here. It’s a pleasure.
Open Access Defined
Dr. Wilner: I remember when publication in neurology was pretty straightforward. It was either the green journal or the blue journal, but things have certainly changed. I think one topic that is not clear to everyone is this concept of open access. Could you define that for us?
Dr. Merino: Sure. Open access is a mode of publication that fosters more open or accessible science. The idea of open access is that it combines two main elements. One is that the papers that are published become immediately available to anybody with an internet connection anywhere in the world without any restrictions.
The second important element from open access, which makes it different from other models we can talk about, is the fact that the authors retain the copyright of their work, but they give the journal and readers a license to use, reproduce, and modify the content.
This is different, for example, from instances where we have funder mandates. For example, NIH papers have to become available 6 months after publication, so they’re available to everybody but not immediately.
Dr. Wilner: I remember that when a journal article was published, say, in Neurology, if you didn’t have a subscription to Neurology, you went to the library that hopefully had a subscription.
If they didn’t have it, you would write to the author and say, “Hey, I heard you have this great paper because the abstract was out there. Could you send me a reprint?” Has that whole universe evaporated?
Dr. Merino: It depends on how the paper is published. For example, in Neurology, some of the research we publish is open access. Basically, if you have an internet connection, you can access the paper.
That’s the case for papers published in our wholly open-access journals in the Neurology family like Neurology Neuroimmunology & Neuroinflammation, Neurology Genetics, or Neurology Education.
For other papers that are published in Neurology, not under open access, there is a paywall. For some of them, the paywall comes down after a few months based on funder mandates and so on. As I was mentioning, the NIH-funded papers are available 6 months later.
In the first 6 months, you may have to go to your library, and if your library has a subscription, you can download it directly. [This is also true for] those that always stay behind the paywall, where you have to have a subscription or your library has to have a subscription.
Is Pay to Publish a Red Flag?
Dr. Wilner: I’m a professional writer. With any luck, when I write something, I get paid to write it. There’s been a long tradition in academic medicine that when you submit an article to, say, Neurology, you don’t get paid as an author for the publication. Your reward is the honor of it being published.
Neurology supports itself in various ways, including advertising and so on. That’s been the contract: free publication for work that merits it, and the journal survives on its own.
With open access, one of the things that’s happened is that — and I’ve published open access myself — is that I get a notification that I need to pay to have my article that I’ve slaved over published. Explain that, please.
Dr. Merino: This is the issue with open access. As I mentioned, the paper gets published. You’re giving the journal a license to publish it. You’re retaining the copyright of your work. That means that the journal cannot make money or support itself by just publishing open access because they belong to you.
Typically, open-access journals are not in print and don’t have much in terms of advertising. The contract is you’re giving me a license to publish it, but it’s your journal, so you’re paying a fee for the journal expenses to basically produce your paper. That’s what’s happening with open access.
That’s been recognized with many funders, for example, with NIH funding or many of the European funders, they’re including open-access fees as part of their funding for research. Now, of course, this doesn’t help if you’re not a funded researcher or if you’re a fellow who’s doing work and so on.
Typically, most journals will have waived fees or lower fees for these situations. The reason for the open-access fee is the fact that you’re retaining the copyright. You’re not giving it to the journal who can then use it to generate its revenue for supporting itself, the editorial staff, and so on.
Dr. Wilner: This idea of charging for publication has created a satellite business of what are called predatory journals. How does one know if the open-access journal that I’m submitting to is really just in the business of wanting my $300 or my $900 to get published? How do I know if that’s a reasonable place to publish?
Predatory Journals
Dr. Merino: That’s a big challenge that has come with this whole idea of open access and the fact that now, many journals are online only, so you’re no longer seeing a physical copy. That has given rise to the predatory journals.
The predatory journal, by definition, is a journal that claims to be open access. They’ll take your paper and publish it, but they don’t provide all the other services that you would typically expect from the fact that you’re paying an open-access fee. This includes getting appropriate peer review, production of the manuscript, and long-term curation and storage of the manuscript.
Many predatory journals will take your open-access fee, accept any paper that you submit, regardless of the quality, because they’re charging the fees for that. They don’t send it to real peer review, and then in a few months, the journal disappears so there’s no way for anybody to actually find your paper anymore.
There are certain checklists. Dr. David Moher at the University of Toronto has produced some work trying to help us identify predatory journals.
One thing I typically suggest to people who ask me this question is: Have you ever heard of this journal before? Does the journal have a track record? How far back does the story of the journal go? Is it supported by a publisher that you know? Do you know anybody who has published there? Is it something you can easily access?
If in doubt, always ask your friendly medical librarian. There used to be lists that were kept in terms of predatory journals that were being constantly updated, but those had to be shut down. As far as I understand, there were legal issues in terms of how things got on that list.
I think that overall, if you’ve heard of it, if it’s relevant, if it’s known in your field, and if your librarian knows it, it’s probably a good legitimate open-access journal. There are many very good legitimate open-access journals.
I mentioned the two that we have in our family, but all the other major journals have their own open-access journal within their family. There are some, like BMC or PLOS, that are completely open-access and legitimate journals.
Impact Factor
Dr. Wilner: What about impact factor? Many journals boast about their impact factor. I’m not sure how to interpret that number.
Dr. Merino: Impact factor is very interesting. The impact factor was developed by medical librarians to try to identify the journals they should be subscribing to. It’s a measure of the average citations to an average paper in the journal.
It doesn’t tell you about specific papers. It tells you, on average, how many of the papers in this journal get cited so many times. It’s calculated by the number of articles that were cited divided by the number of articles that were published. Journals that publish many papers, like Neurology, have a hard time bringing up their impact factor beyond a certain level.
Similarly, very small journals with one or two very highly cited papers have a very high impact factor. It’s being used as a measure, perhaps inappropriately, of how good or how reputable a journal is. We all say we don’t care about journal impact factors, but we all know our journal impact factor and we used to know it to three decimals. Now, they changed the system, and there’s only one decimal point, which makes more sense.
This is more important, for example, for authors when deciding where to submit papers. I know that in some countries, particularly in Europe, the impact factor of the journal where you publish has an impact on your promotion decisions.
I would say what’s even more important than the impact factor, is to say, “Well, is this the journal that fits the scope of my paper? Is this the journal that reaches the audience that I want to reach when I write my paper?”
There are some papers, for example, that are very influential. The impact factor just captures citations. There are some papers that are very influential that may not get cited very often. There may be papers that change clinical practice.
If you read a paper that tells you that you should be changing how you treat your patients with myasthenia based on this paper, that may not get cited. It’s a very clinically focused paper, but it’s probably more impactful than one that gets cited very much in some respect, or they make it to public policy decisions, and so on.
I think it’s important to look more at the audience and the journal scope when you submit your papers.
Dr. Wilner: One other technical question. The journals also say they’re indexed in PubMed or Google Scholar. If I want to publish my paper and I want it indexed where the right people are going to find it, where does it need to be indexed?
Dr. Merino: I grew up using Index Medicus, MedlinePlus, and the Library of Science. I still do. If I need to find something, I go to PubMed. Ideally, papers are listed in MedlinePlus or can be found in PubMed. They’re not the same thing, but you can find them through them.
That would be an important thing. Nowadays, a lot more people are using Google Scholar or Google just to identify papers. It may be a little bit less relevant, but it’s still a measure of the quality of the journal before they get indexed in some of these. For example, if you get listed in MedlinePlus, it has gone through certain quality checks by the index itself to see whether they would accept the journal or not. That’s something you want to check.
Typically, most of the large journals or the journals you and I know about are listed in more than one place, right? They’re listed in Scopus and Web of Science. They’re listed in MedlinePlus and so on. Again, if you’re submitting your paper, go somewhere where you know the journal and you’ve heard about it.
Dr. Wilner: I’m not going to ask you about artificial intelligence. We can do that another time. I want to ask something closer to me, which is this question of publish or perish.
There seems to be, in academics, more emphasis on the number of papers that one has published rather than their quality. How does a younger academician or one who really needs to publish cope with that?
Dr. Merino: Many people are writing up research that may not be relevant or that may not be high quality just because you need to have a long list of papers to get promoted, for example, if you’re an academician.
Doug Altman, who was a very influential person in the field quality of not only medical statistics but also medical publishing, had the idea that we need less research, but we need better research.
We often receive papers where you say, well, what’s the rationale behind the question in this paper? It’s like they had a large amount of data and were trying to squeeze as much as they could out of that. I think, as a young academician, the important thing to think about is whether it is an important question that matters to you and to the field, from whatever perspective, whether it’s going to advance research, advance clinical care, or have public policy implications.
Is this one where the answer will be important no matter what the answer is? If you’re thinking of that, your work will be well recognized, people will know you, and you’ll get invited to collaborate. I think that’s the most important thing rather than just churning out a large number of papers.
The productivity will come from the fact that you start by saying, let me ask something that’s really meaningful to me and to the field, with a good question and using strong research methodology.
Dr. Wilner: Thanks for that, Dr. Merino. I think that’s very valuable for all of us. This has been a great discussion. Do you have any final comments before we wrap up?
Dr. Merino: I want to encourage people to continue reading medical journals all the time and submitting to us, again, good research and important questions with robust methodology. That’s what we’re looking for in Neurology and most serious medical journals.
Dr. Wilner is an associate professor of neurology at the University of Tennessee Health Science Center, Memphis. Dr. Merino is a professor in the department of neurology at Georgetown University Medical Center, Washington, DC. Dr. Wilner reported conflicts of interest with Accordant Health Services and Lulu Publishing. Dr. Merino reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Andrew N. Wilner, MD: My guest today is Dr. Jose Merino, editor in chief of the Neurology family of journals and professor of neurology and co-vice chair of education at Georgetown University in Washington, DC.
Our program today is a follow-up of Dr. Merino’s presentation at the recent American Academy of Neurology meeting in Denver, Colorado. Along with two other panelists, Dr. Merino discussed the role of open-access publication and the dangers of predatory journals.
Jose G. Merino, MD, MPhil: Thank you for having me here. It’s a pleasure.
Open Access Defined
Dr. Wilner: I remember when publication in neurology was pretty straightforward. It was either the green journal or the blue journal, but things have certainly changed. I think one topic that is not clear to everyone is this concept of open access. Could you define that for us?
Dr. Merino: Sure. Open access is a mode of publication that fosters more open or accessible science. The idea of open access is that it combines two main elements. One is that the papers that are published become immediately available to anybody with an internet connection anywhere in the world without any restrictions.
The second important element from open access, which makes it different from other models we can talk about, is the fact that the authors retain the copyright of their work, but they give the journal and readers a license to use, reproduce, and modify the content.
This is different, for example, from instances where we have funder mandates. For example, NIH papers have to become available 6 months after publication, so they’re available to everybody but not immediately.
Dr. Wilner: I remember that when a journal article was published, say, in Neurology, if you didn’t have a subscription to Neurology, you went to the library that hopefully had a subscription.
If they didn’t have it, you would write to the author and say, “Hey, I heard you have this great paper because the abstract was out there. Could you send me a reprint?” Has that whole universe evaporated?
Dr. Merino: It depends on how the paper is published. For example, in Neurology, some of the research we publish is open access. Basically, if you have an internet connection, you can access the paper.
That’s the case for papers published in our wholly open-access journals in the Neurology family like Neurology Neuroimmunology & Neuroinflammation, Neurology Genetics, or Neurology Education.
For other papers that are published in Neurology, not under open access, there is a paywall. For some of them, the paywall comes down after a few months based on funder mandates and so on. As I was mentioning, the NIH-funded papers are available 6 months later.
In the first 6 months, you may have to go to your library, and if your library has a subscription, you can download it directly. [This is also true for] those that always stay behind the paywall, where you have to have a subscription or your library has to have a subscription.
Is Pay to Publish a Red Flag?
Dr. Wilner: I’m a professional writer. With any luck, when I write something, I get paid to write it. There’s been a long tradition in academic medicine that when you submit an article to, say, Neurology, you don’t get paid as an author for the publication. Your reward is the honor of it being published.
Neurology supports itself in various ways, including advertising and so on. That’s been the contract: free publication for work that merits it, and the journal survives on its own.
With open access, one of the things that’s happened is that — and I’ve published open access myself — is that I get a notification that I need to pay to have my article that I’ve slaved over published. Explain that, please.
Dr. Merino: This is the issue with open access. As I mentioned, the paper gets published. You’re giving the journal a license to publish it. You’re retaining the copyright of your work. That means that the journal cannot make money or support itself by just publishing open access because they belong to you.
Typically, open-access journals are not in print and don’t have much in terms of advertising. The contract is you’re giving me a license to publish it, but it’s your journal, so you’re paying a fee for the journal expenses to basically produce your paper. That’s what’s happening with open access.
That’s been recognized with many funders, for example, with NIH funding or many of the European funders, they’re including open-access fees as part of their funding for research. Now, of course, this doesn’t help if you’re not a funded researcher or if you’re a fellow who’s doing work and so on.
Typically, most journals will have waived fees or lower fees for these situations. The reason for the open-access fee is the fact that you’re retaining the copyright. You’re not giving it to the journal who can then use it to generate its revenue for supporting itself, the editorial staff, and so on.
Dr. Wilner: This idea of charging for publication has created a satellite business of what are called predatory journals. How does one know if the open-access journal that I’m submitting to is really just in the business of wanting my $300 or my $900 to get published? How do I know if that’s a reasonable place to publish?
Predatory Journals
Dr. Merino: That’s a big challenge that has come with this whole idea of open access and the fact that now, many journals are online only, so you’re no longer seeing a physical copy. That has given rise to the predatory journals.
The predatory journal, by definition, is a journal that claims to be open access. They’ll take your paper and publish it, but they don’t provide all the other services that you would typically expect from the fact that you’re paying an open-access fee. This includes getting appropriate peer review, production of the manuscript, and long-term curation and storage of the manuscript.
Many predatory journals will take your open-access fee, accept any paper that you submit, regardless of the quality, because they’re charging the fees for that. They don’t send it to real peer review, and then in a few months, the journal disappears so there’s no way for anybody to actually find your paper anymore.
There are certain checklists. Dr. David Moher at the University of Toronto has produced some work trying to help us identify predatory journals.
One thing I typically suggest to people who ask me this question is: Have you ever heard of this journal before? Does the journal have a track record? How far back does the story of the journal go? Is it supported by a publisher that you know? Do you know anybody who has published there? Is it something you can easily access?
If in doubt, always ask your friendly medical librarian. There used to be lists that were kept in terms of predatory journals that were being constantly updated, but those had to be shut down. As far as I understand, there were legal issues in terms of how things got on that list.
I think that overall, if you’ve heard of it, if it’s relevant, if it’s known in your field, and if your librarian knows it, it’s probably a good legitimate open-access journal. There are many very good legitimate open-access journals.
I mentioned the two that we have in our family, but all the other major journals have their own open-access journal within their family. There are some, like BMC or PLOS, that are completely open-access and legitimate journals.
Impact Factor
Dr. Wilner: What about impact factor? Many journals boast about their impact factor. I’m not sure how to interpret that number.
Dr. Merino: Impact factor is very interesting. The impact factor was developed by medical librarians to try to identify the journals they should be subscribing to. It’s a measure of the average citations to an average paper in the journal.
It doesn’t tell you about specific papers. It tells you, on average, how many of the papers in this journal get cited so many times. It’s calculated by the number of articles that were cited divided by the number of articles that were published. Journals that publish many papers, like Neurology, have a hard time bringing up their impact factor beyond a certain level.
Similarly, very small journals with one or two very highly cited papers have a very high impact factor. It’s being used as a measure, perhaps inappropriately, of how good or how reputable a journal is. We all say we don’t care about journal impact factors, but we all know our journal impact factor and we used to know it to three decimals. Now, they changed the system, and there’s only one decimal point, which makes more sense.
This is more important, for example, for authors when deciding where to submit papers. I know that in some countries, particularly in Europe, the impact factor of the journal where you publish has an impact on your promotion decisions.
I would say what’s even more important than the impact factor, is to say, “Well, is this the journal that fits the scope of my paper? Is this the journal that reaches the audience that I want to reach when I write my paper?”
There are some papers, for example, that are very influential. The impact factor just captures citations. There are some papers that are very influential that may not get cited very often. There may be papers that change clinical practice.
If you read a paper that tells you that you should be changing how you treat your patients with myasthenia based on this paper, that may not get cited. It’s a very clinically focused paper, but it’s probably more impactful than one that gets cited very much in some respect, or they make it to public policy decisions, and so on.
I think it’s important to look more at the audience and the journal scope when you submit your papers.
Dr. Wilner: One other technical question. The journals also say they’re indexed in PubMed or Google Scholar. If I want to publish my paper and I want it indexed where the right people are going to find it, where does it need to be indexed?
Dr. Merino: I grew up using Index Medicus, MedlinePlus, and the Library of Science. I still do. If I need to find something, I go to PubMed. Ideally, papers are listed in MedlinePlus or can be found in PubMed. They’re not the same thing, but you can find them through them.
That would be an important thing. Nowadays, a lot more people are using Google Scholar or Google just to identify papers. It may be a little bit less relevant, but it’s still a measure of the quality of the journal before they get indexed in some of these. For example, if you get listed in MedlinePlus, it has gone through certain quality checks by the index itself to see whether they would accept the journal or not. That’s something you want to check.
Typically, most of the large journals or the journals you and I know about are listed in more than one place, right? They’re listed in Scopus and Web of Science. They’re listed in MedlinePlus and so on. Again, if you’re submitting your paper, go somewhere where you know the journal and you’ve heard about it.
Dr. Wilner: I’m not going to ask you about artificial intelligence. We can do that another time. I want to ask something closer to me, which is this question of publish or perish.
There seems to be, in academics, more emphasis on the number of papers that one has published rather than their quality. How does a younger academician or one who really needs to publish cope with that?
Dr. Merino: Many people are writing up research that may not be relevant or that may not be high quality just because you need to have a long list of papers to get promoted, for example, if you’re an academician.
Doug Altman, who was a very influential person in the field quality of not only medical statistics but also medical publishing, had the idea that we need less research, but we need better research.
We often receive papers where you say, well, what’s the rationale behind the question in this paper? It’s like they had a large amount of data and were trying to squeeze as much as they could out of that. I think, as a young academician, the important thing to think about is whether it is an important question that matters to you and to the field, from whatever perspective, whether it’s going to advance research, advance clinical care, or have public policy implications.
Is this one where the answer will be important no matter what the answer is? If you’re thinking of that, your work will be well recognized, people will know you, and you’ll get invited to collaborate. I think that’s the most important thing rather than just churning out a large number of papers.
The productivity will come from the fact that you start by saying, let me ask something that’s really meaningful to me and to the field, with a good question and using strong research methodology.
Dr. Wilner: Thanks for that, Dr. Merino. I think that’s very valuable for all of us. This has been a great discussion. Do you have any final comments before we wrap up?
Dr. Merino: I want to encourage people to continue reading medical journals all the time and submitting to us, again, good research and important questions with robust methodology. That’s what we’re looking for in Neurology and most serious medical journals.
Dr. Wilner is an associate professor of neurology at the University of Tennessee Health Science Center, Memphis. Dr. Merino is a professor in the department of neurology at Georgetown University Medical Center, Washington, DC. Dr. Wilner reported conflicts of interest with Accordant Health Services and Lulu Publishing. Dr. Merino reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.