User login
Needs of Veterans With Personality Disorder Diagnoses in Community-Based Mental Health Care
Needs of Veterans With Personality Disorder Diagnoses in Community-Based Mental Health Care
Personality disorders (PDs) are enduring patterns of internal experience and behavior that differ from cultural norms and expectations, are inflexible and pervasive, have their onset in adolescence or early adulthood, and lead to distress or impairment. Ten PDs are included in the Diagnostic and Statistical Manual of Mental Disorders (Fifth Edition): paranoid, schizoid, schizotypal, borderline, antisocial, histrionic, narcissistic, avoidant, dependent, and obsessive-compulsive.1 These disorders impose a high burden on patients, families, health care systems, and broader economic systems.2,3 Up to 1 in 7 persons in the community and 50% of those receiving outpatient mental health treatment experience a PD.4,5 These conditions are associated with an increased risk of adverse events, including suicide attempt and death by suicide, criminal-legal involvement, homelessness, substance use, underemployment, relational issues, and high utilization of psychiatric services.6-9 PDs are routinely underassessed, underdocumented, and undertreated in clinical settings, and consistently receive less research funding than other, less prevalent forms of psychopathology. 10-12 As a result, there is limited understanding of clinical needs of individuals experiencing PDs.
MILITARY VETERANS WITH PERSONALITY DISORDERS
Underacknowledgment of PDs and their associated difficulties may be especially pronounced in veteran populations. Due to longstanding etiological theories that implicate childhood trauma and adolescent onset in pathology development, PDs are traditionally considered pre-existing conditions or developmental abnormalities by the US Department of Defense and US Department of Veterans Affairs (VA). As a result, PDs are therefore deemed incompatible with military service and ineligible for service-connected disability benefits.13-15 Such determinations allowed PD pathology to be used as grounds for discharge for 26,000 service members from 2001 to 2007, or 2.6% of total enlisted discharges during that period.13,15,16
Despite this structural discrimination, recent research suggests veterans may be more likely to experience PD pathology than the general population.17 For example, a 2021 epidemiological survey in a community-based veteran sample found elevated rates of borderline, antisocial, and schizotypal PDs (6%-13%).6 In contrast, only 0.8% to 5.0% of veteran electronic health records (EHRs) have a documented PD diagnosis.8,18,19 Such elevations in PD pathology within veteran samples imply either a disproportionately high prevalence among enlistees (and therefore missed during recruitment procedures) or onset following military service, possibly due to exposure to traumatic events and/ or occupational stress.17 Due to the relative infancy of research in this area and a lack of longitudinal studies, etiology and course of illness for personality pathology in veterans remains largely unclear.
Structural underacknowledgment of PDs among military personnel has contributed to their underrepresentation in research on veteran populations. PD-focused research with veterans is rare, despite a rapid increase in broader empirical attention paid to these conditions in nonveteran samples.20 A recent meta-analysis of veterans with PDs identified 27 studies that included basic prevalence statistics. PDs were rarely a primary focus for these studies, and most were limited to veterans seen in Veterans Health Administration (VHA) settings.17 The literature also paints a bleak picture, suggesting veterans who experience PDs are at higher risk for suicide attempt and death by suicide, criminal-legal involvement, and homelessness. They also tend to experience more severe comorbid psychopathological symptoms and more often use high-intensity mental health services (eg, care within emergency departments or psychiatric inpatient settings) than veterans without PD pathology.6,8,18,19,21 However, PD pathology does not appear to impede the effectiveness of treatment for veterans.22-24 The implications of PD pathology on broader psychosocial functioning and health care needs certify a need for additional research that examines patterns of personality pathology, particularly in veterans outside the VHA.
METHODS
This study aims to enhance understanding of veterans affected by PDs and offer insight and guidance for treatment of these conditions in federal and nonfederal treatment settings. Previous research has been largely limited to VHA care-receiving samples; the longstanding stigma against PDs by the US military and VA may contribute to biased diagnosis and documentation of PDs in these settings. A large sample of veterans receiving community-based mental health care was therefore used to explore aims of the current study. This study specifically examined demographic patterns, diagnostic comorbidity, psychosocial outcomes, and treatment care settings among veterans with and without a PD diagnosis. Consistent with previous research, we hypothesized that veterans with a PD diagnosis would have more severe mental health comorbidities, poorer psychosocial outcomes, and receive care in higher intensity settings relative to veterans without a diagnosis.
Data for the sample were drawn from the Mental Health Client-Level Data, a publicly available national dataset of nearly 7 million patients who received mental health treatment services provided or funded through state mental health agencies in 2022.25 The analytic sample included about 2.5 million patients for whom veteran status and data around the presence or absence of a PD diagnosis were available. Of these patients, 104,198 were identified as veterans. Veteran patients were identified as predominantly male (63%), White (71%), non-Hispanic (90%), and never married (54%).
Measures
The parent dataset included demographic, clinical, and psychosocial outcome information reported by treatment facilities to individual state administrative systems for each patient who received services. To protect patient privacy, only nonprotected health information is included, and efforts were made throughout compilation of the parent dataset to ensure patient privacy (eg, limiting detail of information disseminated for public access). Because the parent dataset does not include protected health information, studies using these data are considered exempt from institutional review board oversight.
Demographic information. This study reviewed veteran status, sex, race, ethnicity, age, education, and marital status. Veteran status was defined by whether the patient was aged ≥ 18 years and had previously served (but was not currently serving) in the military. Patients with a history of service in the National Guard or Military Reserves were only classified as veterans if they had been called or ordered to active duty while serving. Sex was operationalized dichotomously as male or female; no patients were identified as intersex, transgender, or other gender identities.
Clinical information. Up to 3 mental health diagnoses were reported for each patient and included the following disorders: personality, trauma and attention-deficit/hyperactivity, stressor, anxiety, conduct, delirium/dementia, bipolar, depressive, oppositional defiant, pervasive developmental, schizophrenia or other psychotic, and alcohol or substance use. Mental health diagnosis categories were generated for the parent dataset by grouping diagnostic codes corresponding to each category. To protect patient privacy, more detailed diagnostic information was not available as part of the parent dataset. Although the American Psychiatric Association recognizes 10 distinct PDs, the exact nature of PD diagnoses was not included within the parent dataset. PD diagnoses were coded to reflect the presence or absence of any such diagnosis.
A substance use problem designation was also provided for patients according to various identification methods, including substance use disorder (SUD) diagnosis, substance use screening results, enrollment in a substance use program, substance use survey, service claims information, and other related sources of information. A severe mental illness or serious emotional disturbance designation was provided for patients meeting state definitions of these designations. Context(s) of service provision were coded as inpatient state psychiatric hospital, community-based program, residential treatment center, judicial institution, or other psychiatric inpatient setting.
Psychosocial outcome information. Patient employment and residential status were also included in analyses. Each reflected status at the time of discharge from services or end of reporting period; employment status was only provided for patients receiving treatment in community-based programs.
Data Analysis
Descriptive statistics and X2 analyses were used to compare demographic, clinical, and psychosocial outcome variables between patients with and without PD diagnoses. These analyses were calculated for both the 104,198 veterans and the 2,222,306 nonveterans aged ≥ 18 years in the dataset. Given the sample size, a conservative α of .01 was used to determine statistical significance.
RESULTS
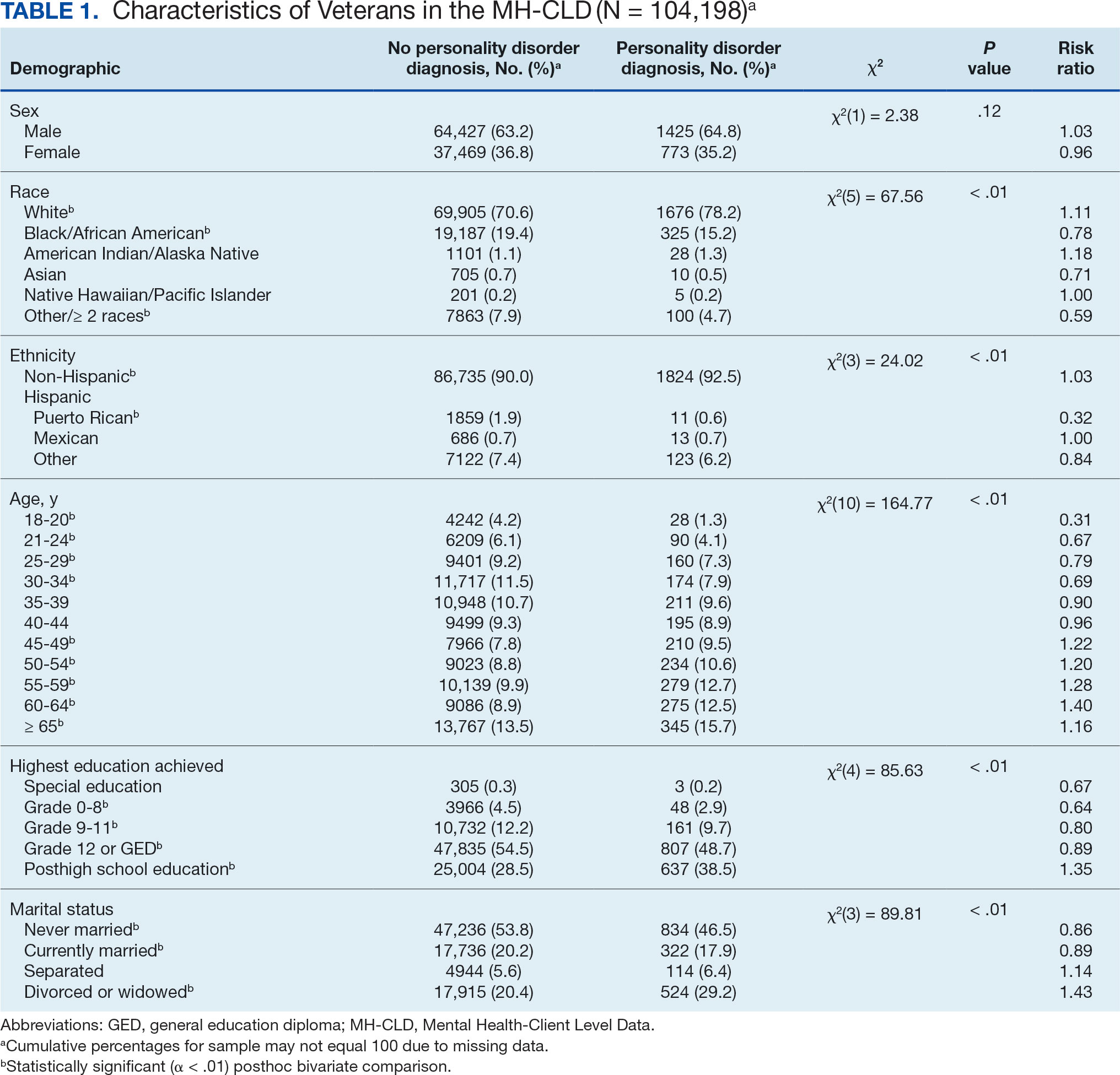
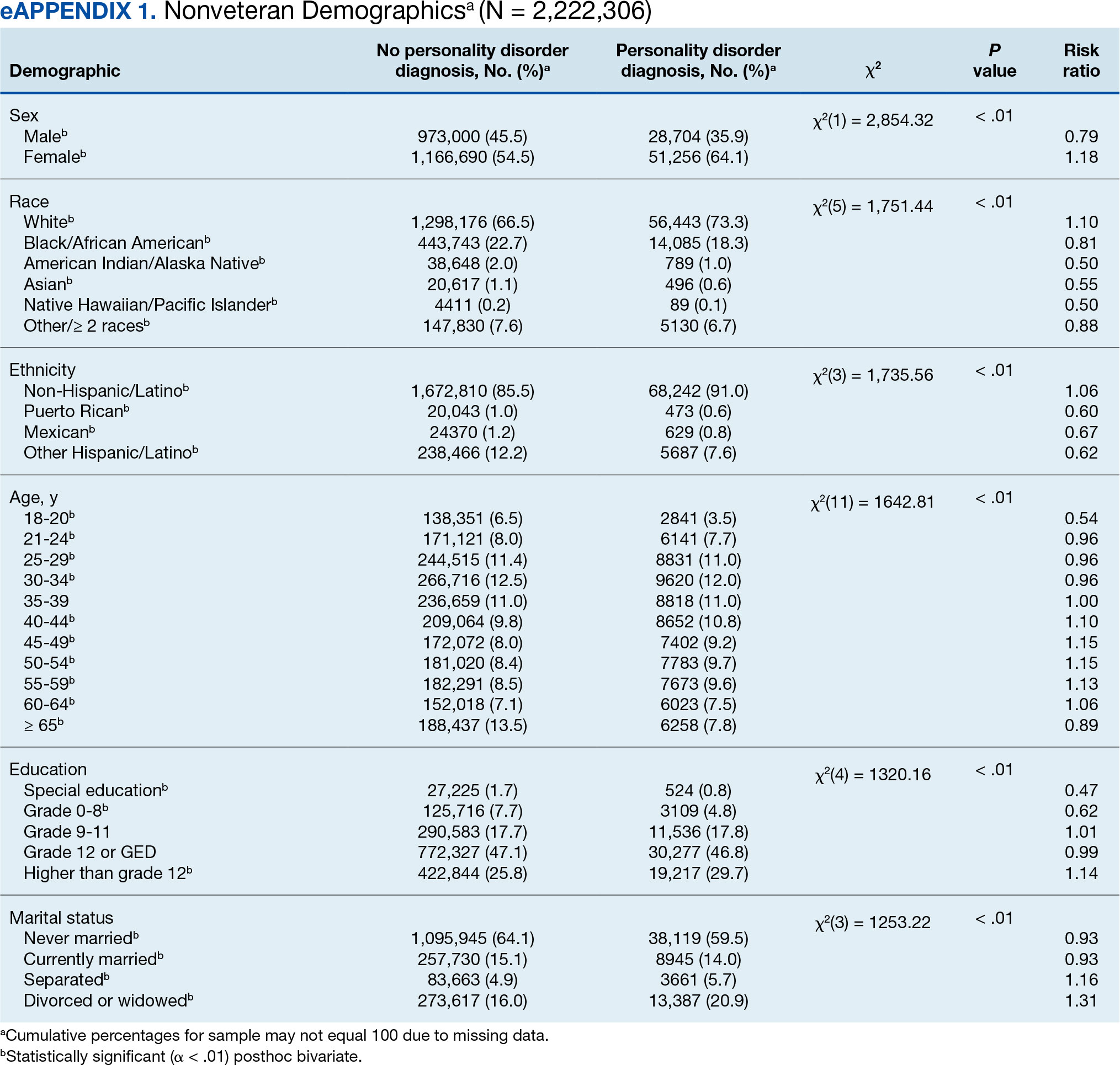
In this sample of persons receiving state-funded mental health care, veterans were significantly less likely than nonveterans to have a documented PD diagnosis (2.1% vs 3.6%, X2 [1] = 647.49; P < .01). PD diagnoses were more common among White (risk ratio [RR], 1.11), non-Hispanic (RR, 1.03) veterans who were in middle to late adulthood (RR, 1.16-1.40), more educated (RR, 1.35), and divorced or widowed (RR, 1.43), and less common among Black/African American (RR, 0.78) or Puerto Rican (RR, 0.32) veterans who were in early adulthood (RR, 0.31-0.79), less educated (RR, 0.64-0.89), and currently married (RR, 0.89) or never married (RR, 0.86). Veteran men and women were equally likely to have a PD diagnosis (RR, 1.03) (Table 1). Among nonveterans, men were less likely than women to have a PD diagnosis (RR, 0.79), and PD diagnoses were most common among persons in middle adulthood (RR, 1.06-1.15) (eAppendix 1).
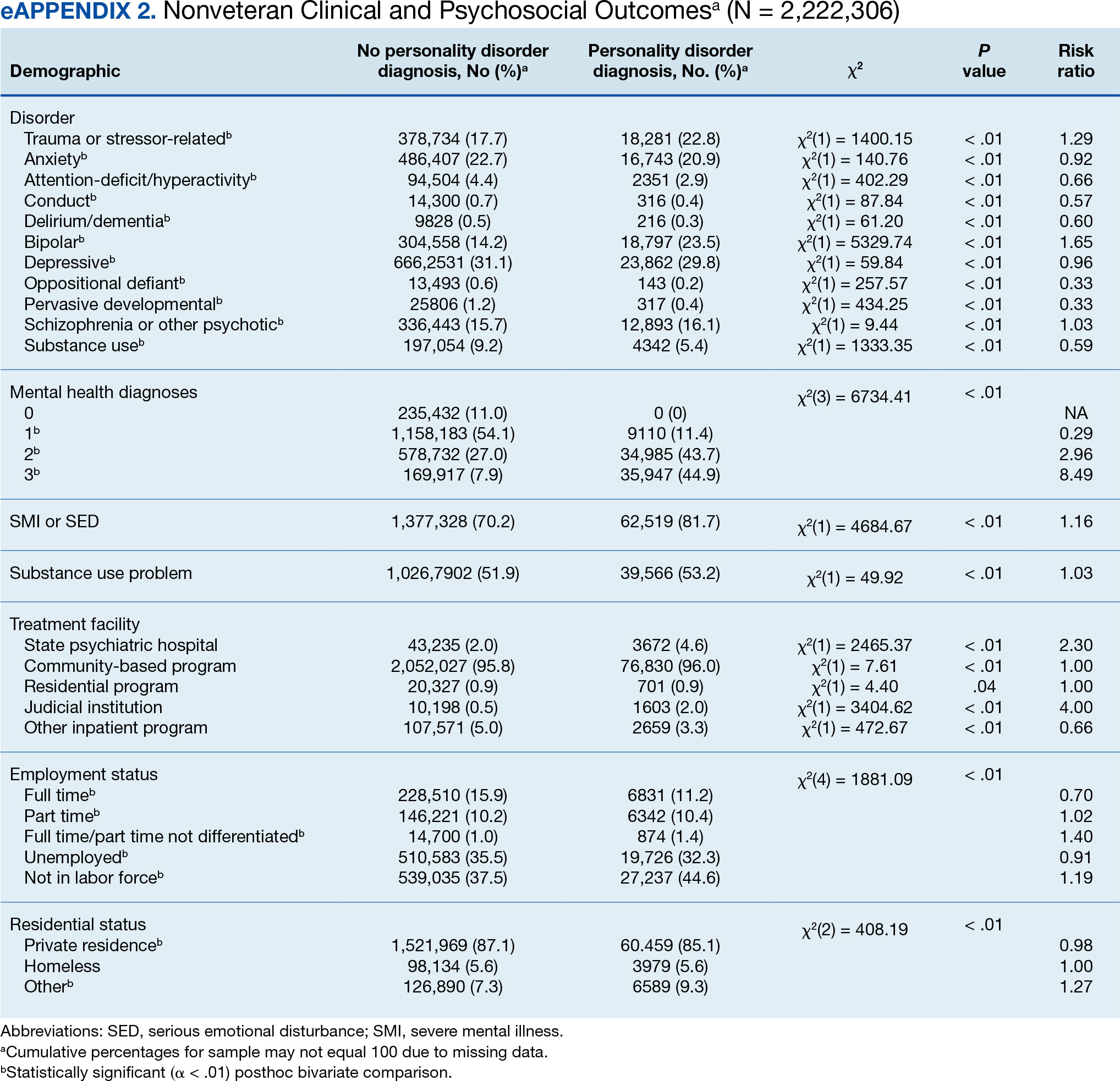
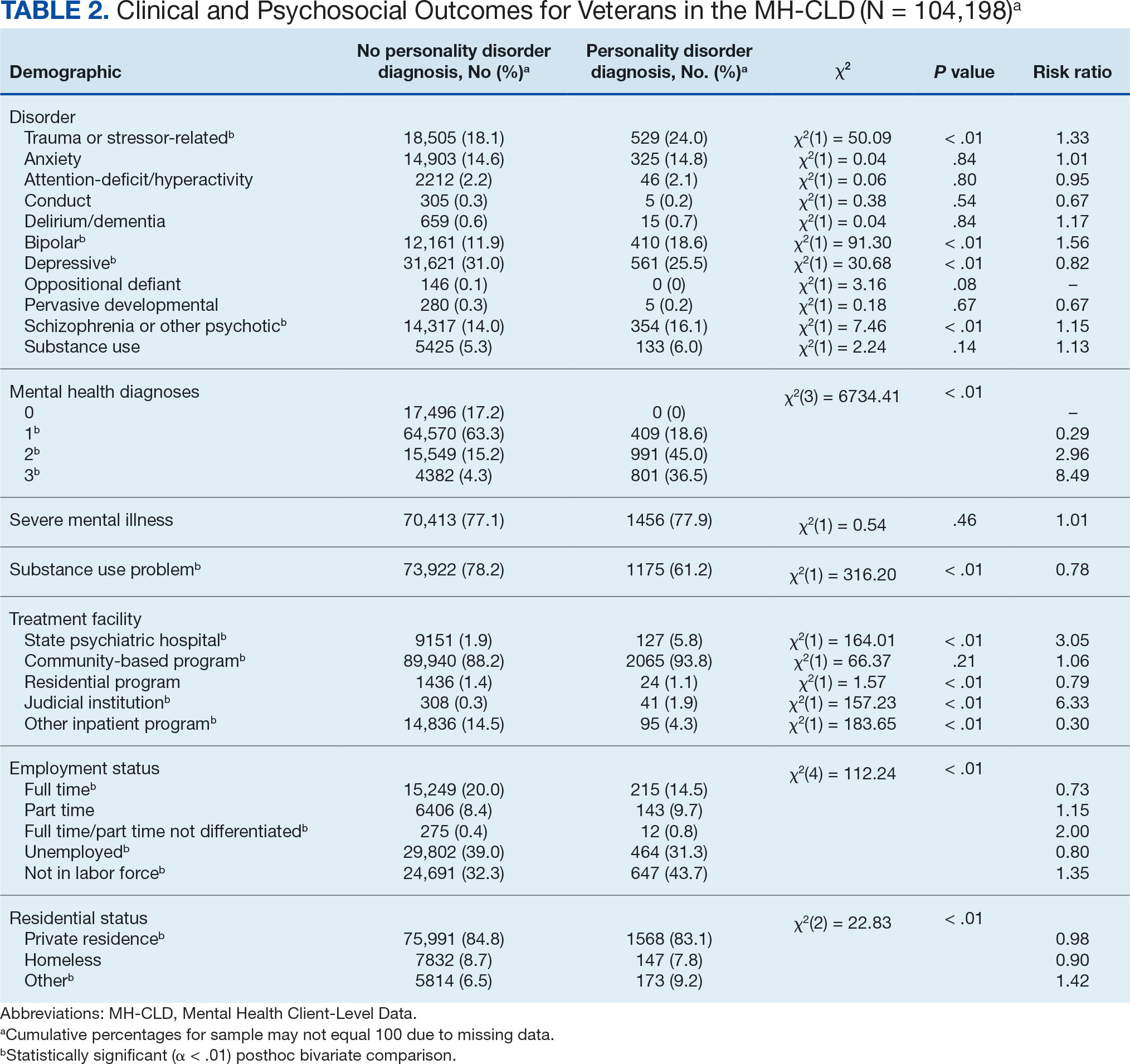
Veterans with a PD diagnosis were more likely than those without a diagnosis to have more diagnoses (RR, 2.96-8.49) and to have comorbid trauma or related stressor (RR, 1.33), or bipolar (RR, 1.56) or psychotic (RR, 1.15) disorder diagnoses, but less likely to have comorbid depressive disorder (RR, 0.82). Although veterans with and without a PD diagnosis were similarly likely to have a comorbid SUD (RR, 1.13), those with a PD diagnosis were significantly less likely to be assigned a substance use problem designation (RR, 0.78). PD diagnosis was also more common among veterans who received services in state psychiatric hospitals (RR, 3.05), community-based clinics (RR, 1.06), and judicial institutions (RR, 6.33) and less common among those who received services in other psychiatric inpatient settings (RR, 0.30). No differences were observed for residential treatment settings (RR, 0.79). Among nonveterans, a PD diagnosis was associated with slightly greater odds of a substance use designation (RR, 1.03) (eAppendix 2).
Veterans with a PD diagnosis were also less likely to have full-time employment (RR, 0.73) and more likely to have undifferentiated employment (RR, 2.00) or to be removed from the labor force (RR, 1.35). Veterans with a PD diagnosis were also more likely to reside in nontraditional living conditions (RR, 1.42) and less likely to be residing in a private residence (RR, 0.98), compared with those without PD diagnosis. The rates of homelessness were similar for veterans with and without a PD diagnosis (RR, 0.90) (Table 2). These patterns were similar among nonveterans.
DISCUSSION
This study examined the rate and correlates of PD diagnosis among a large, community-based sample of veterans receiving state-funded mental health care. About 2% of veterans in this sample had a PD diagnosis, with diagnoses more common among veterans who were White, non-Hispanic, aged ≥ 45 years, with higher education, divorced or widowed, also diagnosed with trauma-related, bipolar, and/or psychotic disorders, underemployed, nontraditionally housed, and receiving treatment in state psychiatric hospital, community-based clinic, or judicial system settings.
The observed rate of PD diagnosis in this study aligns with what is typically observed in VHA EHRs.8,18,19 However, the rate is notably lower than prevalence estimates for psychiatric outpatient settings (about 50%) and in meta-analyses of prevalence among veterans (0.8%-23% for each of the 10 PDs).4,17,26 Longstanding stigma against PDs may contribute to underdiagnosis. For example, many clinicians are concerned that documentation or disclosure of a PD will interfere with the patient’s ability to access treatment due to stigma and discrimination.27,28 These fears are not unfounded; even among clinicians, PDs are commonly considered untreatable, and many individuals with PDs are denied access to evidence-based treatments due to the diagnosis.29 In a 2016 survey of community psychiatrists, nearly 1 in 4 reported that they avoid taking patients with a borderline PD diagnosis in their caseloads.28 To date, no studies have been conducted to explore clinicians’ willingness to accept patients with other PDs or, specifically, among veterans.
Despite such widespread stigma, research suggests clinicians' negative attitudes toward PDs can be decreased through antistigma campaigns.30 However, it remains unclear if such efforts also contribute to an increase in clinicians’ willingness to document PD diagnoses. Without accurate identification and documentation, the field’s understanding of PDs will remain limited.
In the current study, veterans with PD diagnoses tended to present with more complex and severe psychiatric comorbidities compared to veterans without such diagnoses. Observed comorbidity of PDs (particularly borderline PD) with trauma-related and bipolar disorders is well established.8 Conversely, co-occurring personality and psychotic disorders—which comprise 16% of veterans with a PD diagnosis in the sample in this study—are not consistently examined in the literature. A 2022 examination of veterans receiving VHA care suggested 12% and 13% of those with a PD diagnosis documented in their EHR also had documented schizophrenia or another psychotic disorder, respectively. PD diagnoses were associated with 6.88- and 9.80-fold increases in risk for comorbid schizophrenia and other psychotic disorder diagnoses, respectively.8 Similarly, a recent longitudinal study of nearly 2 million Swedish individuals suggested borderline PD is specifically associated with a > 24-times greater risk of having a comorbid psychotic disorder.31 It is therefore possible that the comorbidity between personality and psychotic disorders is quite common despite its relative lack of attention in empirical research.
Veterans with PD diagnoses in this study were also more likely to experience substandard housing, employment challenges, and receive treatment through judicial institutions than those without a PD diagnosis. Such findings are consistent with previous research demonstrating the substantial psychosocial challenges associated with PD diagnosis, even after controlling for comorbid conditions.7,9 Veterans with PDs may benefit from specialized case management and support to facilitate stable housing and employment and to mitigate the risk of judicial involvement. Some research suggests veterans with PDs may be less likely to gain competitive employment after participating in VA therapeutic and supportive employment services programs, suggesting standard programming may be less suitable for this population.32 Similarly, other research suggests individuals with PDs may benefit more from specialized, intensive services than standard clinical case management.33 Future research may therefore benefit from clarifying the degree to which adaptations to standard programming could yield beneficial effects for persons with PD diagnoses.
Implications
Cumulatively, the results of this study attest to the necessity for transdiagnostic treatment planning that includes close collaboration between psychotherapeutic, pharmacological, and case management services. Some psychotherapy models for PDs, such as dialectical behavior therapy (DBT), which includes a combination of group skills training, individual therapy, as-needed phone coaching, and therapist consultation, may be successfully adapted to include this collaboration.34-36 However, implementation of such comprehensive programming often requires extensive clinician training and coordination of resources, which poses implementation challenges.37-39 In 2021, the VHA began large-scale implementation of PD-specific psychotherapy for veterans with recent suicidal self-directed violence and borderline PD, including DBT, though to date results remain unclear.40 Generalist approaches, such as good psychiatric management (GPM), which emphasizes emotional validation, practical problem solving, realistic goal setting, and relationship functioning within the context of standard care appointments, may be more easily implemented in community care settings due to lesser training and resource requirements and can also be adapted to include needed elements of care coordination.41,42 Both DBT and GPM were initially developed for the treatment of borderline PD. Although DBT has also demonstrated some effectiveness in the treatment of antisocial PD, potential applications of DBT and GPM to other PDs remain largely underdeveloped.43-46
There are no widely accepted medications for the treatment of PDs. Pharmacotherapy for these conditions typically consists of individualized approaches informed by personal experience that attempt to balance targeting of specific symptoms while minimizing polypharmacy and potential risks (eg, overdose or addiction).47,48 Despite this, pharmacotherapy is often considered a necessary component in the treatment of bipolar and psychotic disorders, both common comorbidities of PDs found in veterans in this study.49,50 Careful consideration of complex comorbidities and pharmacotherapy needs is warranted in the treatment of veterans with PDs. Future research may benefit from clarifying clinical guidelines around pharmacotherapy, particularly for observed comorbidities of PDs to trauma, bipolar, and psychotic disorders.
It is important to note the discrepancies in the results of this study surrounding patient substance use. The results suggest a negligible or inverse association between the likelihood of a PD diagnosis and difficulties with substance use among the veterans in this study. However, the unexpectedly low rate of SUD diagnoses (< 6%) suggests that they were likely underdocumented. Research suggests a strong association between personality and SUDs in both veteran and civilian samples.6,51 Results suggesting a lower prevalence of substance use difficulties among treatment-seeking veterans with PDs should be interpreted with great caution.
Demographically, PD diagnoses were more common among veterans who were White, non-Hispanic, and aged ≥ 45 years, and less common among veterans who were Black/ African American, mixed/unspecified race, Puerto Rican or other non-Mexican Hispanic ethnicity, or aged < 35 years. No significant sex-based differences were observed. These patterns are consistent with research suggesting individuals who identify as Black may be less likely than individuals who identify as White to report PD symptoms, meet criteria for a PD, and have a PD diagnosed even when it is warranted.52
The findings observed in this study with respect to age, however, are notably inconsistent with the literature. Previous research typically suggests a negative association between age and PD pathology; however, a 2020 review of PDs in older adults by Penders et al suggests a prevalence of 11% to 15% in this population.53,54 Research into PDs most often focuses on adolescent and early adulthood developmental periods, limiting insight into the phenomenology of PDs in middle to late adulthood.55 Further, most research into PDs among geriatric populations has focused on psychometric assessment rather than practical treatment guidance.54 However, in this study, elevated risk for PD diagnoses was salient throughout middle to late adulthood among veterans; similar, albeit less pronounced patterns were also observed for elevated risk of PD diagnosis in middle adulthood among nonveterans. Such findings suggest clarifying the phenomenology and treatment needs of individuals with PDs in middle to late adulthood may have particularly salient implications for the mental health care of veterans affected by these conditions. As the veteran population advances in age, these needs will present unique challenges if health care systems are unprepared to effectively address them.
Limitations
This study is characterized by several strengths, most notably its use of a large dataset recently collected on a national scale. Few studies outside of the VHA system include samples of > 100,000 treatment-seeking veterans collected on a national scale. Nevertheless, results should be understood within the context of several methodological limitations. However, the dataset was limited to the first 3 diagnoses documented in patients’ EHRs, and many patients had no listed diagnoses. Patients with complex comorbidities may have > 3 diagnoses; for these individuals, data provided an incomplete picture of clinical presentation. This is especially relevant for individuals with PDs, who tend to meet criteria for a range of comorbid conditions.8,10 The now dated practice of listing PDs on Axis II also increases the chance of clinicians listing PDs after conditions traditionally listed on Axis I (eg, major depressive disorder) in patient charts.56 This study’s inclusion of only the first 3 listed diagnoses likely underestimated true PD diagnosis prevalence.
The results of this study must be interpreted as reflecting the prevalence and correlates of receiving a PD diagnosis rather than meeting diagnostic criteria for a PD. Relatedly, PD diagnoses were reported as a single construct, limiting insight into prevalence and correlates of individual PD diagnoses (eg, borderline vs paranoid PDs). Meta-analyses estimates suggest PD prevalence among veterans is likely much higher than observed in this study.17 Stigma continues to discourage clinicians from documenting and disclosing PD diagnoses even when warranted.27,28 Continued research should aim to clarify conditions (eg, patient presentation, stigma, or institutional culture) contributing to documentation of PD diagnoses. Given the cross-sectional nature of this study, results cannot speak to longitudinal treatment outcomes or prognosis of persons receiving a PD diagnosis.
Despite its large sample size and national representation, the sampling strategy of this study could have contributed to idiosyncrasies in the dataset. Restriction of data to the persons receiving state-funded mental health services introduces a notable bias to the composition of the sample, which is likely comprised of a disproportionately high number of Medicaid recipients, students, and individuals with chronic illnesses and underrepresentation of persons who pay for mental health services using private insurance or private pay arrangements. As such, although socioeconomic information was not provided within this dataset, one can presume a generally lower socioeconomic status among study participants compared to the community at large. This study also included a proportionally small sample of veterans (3.6% compared to about 6.2% in the broader US population), suggesting veterans may have been underrepresented or underidentified in surveyed mental health care settings.57 This study also did not include data around service in active-duty military, national guard, or military reserves; a greater proportion of the sample likely had a history of military service than was represented by veteran status designation. Further, the proportionally high sample of individuals with severe mental illness suggests a likely overrepresentation of such conditions in surveyed settings.
Institutional differences in the practice of assigning diagnoses likely limited statistical power to detect potentially meaningful associations and effects. Structural influences, such as stigma and institutional culture, may have notable effects on documentation practices, particularly for PDs. Future research should aim to replicate observed associations using more controlled diagnostic procedures.
Lastly, even with the use of a more conservative α and a focus on effect sizes to guide interpretation of results, use of multiple bivariate analyses can be presumed to have increased the likelihood of type I error. Given the limited prior research in this area, an exploratory approach to statistical analysis was considered warranted to maximize opportunity for identifying areas in need of additional empirical attention. Continued research using more conservative statistical approaches (eg, multivariate analyses) is needed to determine replicability and generalizability of observed results.
CONCLUSIONS
This study examined the prevalence and correlates of PD diagnoses in a national sample of veterans receiving community-based, state-funded mental health care. About 2% received a PD diagnosis, with diagnoses most common among veterans who were White, non-Hispanic, aged ≥ 45 years, also diagnosed with trauma-based, bipolar, and/or psychotic disorders, underemployed, nontraditionally housed, and receiving treatment in a state psychiatric hospital or judicial system setting. The results attest to a necessity for transdiagnostic treatment planning and care coordination for this population, with particular attention to psychosocial stressors.
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed, text revision. American Psychiatric Association; 2022.
- Hastrup LH, Jennum P, Ibsen R, Kjellberg J, Simonsen E. Societal costs of borderline personality disorders: a matched-controlled nationwide study of patients and spouses. Acta Psychiatr Scand. 2019;140(5):458-467. doi:10.1111/acps.13094
- Sveen CA, Pedersen G, Ulvestad DA, Zahl KE, Wilberg T, Kvarstein EH. Societal costs of personality disorders: a cross-sectional multicenter study of treatment-seeking patients in mental health services in Norway. J Clin Psychol. 2023;79(8):1752-1769. doi:10.1002/jclp.23504
- Beckwith H, Moran PF, Reilly J. Personality disorder prevalence in psychiatric outpatients: a systematic literature review. Personal Ment Health. 2014;8(2):91-101. doi:10.1002/pmh.1252
- Eaton NR, Greene AL. Personality disorders: community prevalence and socio-demographic correlates. Curr Opin Psychol. 2018;21:28-32. doi:10.1016/j.copsyc. 2017.09.001
- Edwards ER, Barnes S, Govindarajulu U, Geraci J, Tsai J. Mental health and substance use patterns associated with lifetime suicide attempt, incarceration, and homelessness: a latent class analysis of a nationally representative sample of U.S. veterans. Psychol Serv. 2021;18(4):619-631. doi:10.1037/ser0000488
- Moran P, Romaniuk H, Coffey C, et al. The influence of personality disorder on the future mental health and social adjustment of young adults: a population-based cohort study. Lancet Psychiatry. 2016;3(7):636-645. doi:10.1016/S2215-0366(16)30029-3
- Nelson SM, Griffin CA, Hein TC, Bowersox N, McCarthy JF. Personality disorder and suicide risk among patients in the Veterans Affairs health system. Personal Disord. 2022;13(6):563-571. doi:10.1037/per0000521
- Skodol AE. Impact of personality pathology on psychosocial functioning. Curr Opin Psychol. 2018;21;33-38. doi:10.1016/j.copsyc.2017.09.006
- Tyrer P, Reed GM, Crawford MJ. Classification, assessment, prevalence, and effect of personality disorder. Lancet. 2015;385(9969):717-726. doi:10.1016/S0140-6736(14)61995-4
- Fitzpatrick S, Goss S, Di Bartolomeo A, Varma S, Tissera T, Earle E. Follow the money: is borderline personality disorder research underfunded in Canada? Can Psychol. 2024;65(1):46-57. doi:10.1037/cap0000375
- Zimmerman M, Gazarian D. Is research on borderline personality disorder underfunded by the National Institute of Health? Psychiatry Res. 2014;220(3):941-944. doi:10.1016/j.psychres.2014.09.021
- Leroux TC. U.S. military discharges and pre-existing personality disorders: a health policy review. Adm Policy Ment Health. 2015;42(6):748-755. doi:10.1007/s10488-014-0611-z
- Monahan MC, Keener JK. Fitness-for-duty evaluations. In Kennedy CH, Zillmer EA, eds. Military Psychology: Clinical and Operational Applications. 2nd ed. Guilford Publications; 2012:25-49.
- Hearing Before the Committee on Veterans’ Affairs, 111th Congress 2nd Sess (2010). Personality disorder discharges: impact on veterans benefits. Accessed March 4, 2025. https://www.govinfo.gov/content/pkg/CHRG-111hhrg61755/html/CHRG-111hhrg61755.htm
- Ader M, Cuthbert R, Hoechst K, Simon EH, Strassburger Z, Wishnie M. Casting troops aside: the United States military’s illegal personality disorder discharge problem. Vietnam Veterans of America. March 2012. Accessed February 28, 2025. https://law.yale.edu/sites/default/files/documents/pdf/Clinics/VLSC_CastingTroopsAside.pdf
- Edwards ER, Tran H, Wrobleski J, Rabhan Y, Yin J, Chiodi C, Goodman M, Geraci J. Prevalence of personality disorders across veteran samples: A meta-analysis. J Pers Disord. 2022;36(3):339-358. doi:10.1521/ pedi.2022.36.3.339
- Holliday R, Desai A, Edwards E, Borges L. Personal i ty disorder diagnosis among just ice -involved veterans: an investigation of VA-using veterans. J Nerv Ment Dis. 2023;211(5):402-406 doi:10.1097/ NMD.0000000000001627
- McCarthy JF, Bossarte RM, Katz IR, et al. Predictive modeling and concentration of the risk of suicide: implications for preventive interventions in the US Department of Veterans Affairs. Am J Public Health. 2015;105(9):1935-1942. doi:10.2105/AJPH.2015.302737
- Liu Y, Chen C, Zhou Y, Zhang N, Liu S. Twenty years of research on borderline personality disorder: a scientometric analysis of hotspots, bursts, and research trends. Front Psych. 2024;15:1361535. doi:10.3389/ fpsyt.2024.1361535
- Williams R, Holliday R, Clem M, Anderson E, Morris EE, Surís A. Borderline personality disorder and military sexual trauma: analysis of previous traumatization and current psychiatric presentation. J Interpers Violence. 2017;32(15):2223-2236. doi:10.1177/0886260515596149
- Holder N, Holliday R, Pai A, Surís A. Role of borderline personality disorder in the treatment of military sexual trauma-related posttraumatic stress disorder with cognitive processing therapy. Behav Med. 2017;43(3):184-190. doi:10.1080/08964289.2016.1276430
- Ralevski E, Ball S, Nich C, Limoncelli D, Petrakis I. The impact of personality disorders on alcohol-use outcomes in a pharmacotherapy trial for alcohol dependence and comorbid Axis I disorders. Am J Addict. 2007;16(6):443- 449. doi:10.1080/10550490701643336
- Walter KH, Bolte TA, Owens GP, Chard KM. The impact of personality disorders on treatment outcome for veterans in a posttraumatic stress disorder residential treatment program. Cognit Ther Res. 2012;36(5):576-584. doi:10.1007/s10608-011-9393-8
- Substance Abuse and Mental Health Services. Mental health client-level data (MH-CLD), 2022. Accessed February 28, 2025. https://www.datafiles.samhsa.gov/dataset/mental-health-client-level-data-2022-mh-cld-2022-ds0001
- Zimmerman M, Rothschild L, Chelminski I. The prevalence of DSM-IV personality disorders in psychiatric outpatients. Am J Psychiatry. 2005;162(10):1911-1918. doi:10.1176/appi.ajp.162.10.1911
- Campbell K, Clarke KA, Massey D, Lakeman R. Borderline personality disorder: To diagnose or not to diagnose? That is the question. Int J Mental Health Nurs. 2020;29(5):972-981. doi:10.1111/inm.12737
- Sisti D, Segal AG, Siegel AM, Johnson R, Gunderson J. Diagnosing, disclosing, and documenting borderline personality disorder: a survey of psychiatrists’ practices. J Pers Disord. 2016;30(6):848-856. doi:10.1521/ pedi_2015_29_228
- Klein P, Fairweather AK, Lawn S. Structural stigma and its impact on healthcare for borderline personality disorder: a scoping review. Int J Ment Health Syst. 2022;16(1):48. doi:10.1186/s13033-022-00558-3
- Knaak S, Szeto AC, Fitch K, Modgill G, Patten S. Stigma towards borderline personality disorder: effectiveness and generalizability of an anti-stigma program for healthcare providers using a pre-post randomized design. Borderline Personal Disord Emot Dysregul. 2015;2:9. doi:10.1186/s40479-015-0030-0
- Tate AE, Sahlin H, Liu S, et al. Borderline personality disorder: associations with psychiatric disorders, somatic illnesses, trauma, and adverse behaviors. Mol Psychiatry. 2022;27:2514-2521. doi:10.1038/s41380- 022-01503-z
- Abraham KM, Yosef M, Resnick SG, Zivin K. Competitive employment outcomes among veterans in VHA Therapeutic and Supported Employment Services programs. Psychiatr Serv. 2017;68(9)938-946. doi:10.1176/appi. ps201600412
- Frisman LK, Mueser KT, Covell NH, et al. Use of integrated dual disorder treatment via Assertive Community Treatment versus clinical case management for persons with co-occurring disorders and antisocial personality disorder. J Nerv Ment Dis. 2009;197(11):822-828. doi:10.1097/NMD.0b013e3181beac52
- Edwards ER, Kober H, Rinne GR, Griffin SA, Axelrod S, Cooney EB. Skills]homework completion and phone coaching as predictors of therapeutic change and outcomes in completers of a DBT intensive outpatient programme. Psychol Psychother. 2021;94(3):504-522. doi:10.1111/papt.12325
- Linehan MM, Dimeff LA, Reynolds SK, et al. Dialectical behavior therapy versus comprehensive validation therapy plus 12-step for the treatment of opioid dependent women meeting criteria for borderline personality disorder. Drug Alcohol Depend. 2002;67(1):13-26. doi:10.1016/s0376-8716(02)00011-x
- Linehan MM, Korslund KE, Harned MS, et al. Dialectical behavior therapy for high suicide risk in individuals with borderline personality disorder: a randomized clinical trial and component analysis. JAMA Psychiatry. 2015;72(5):475-482.doi:10.1001 /jamapsychiatry.2014.3039
- Carmel A, Rose ML, Fruzzetti AE. Barriers and solutions to implementing dialectical behavior therapy in a public behavioral health system. Adm Policy Ment Health. 2014;41(5):608-614. doi:10.1007/s10488-013-0504-6
- Decker SE, Matthieu MM, Smith BN, Landes SJ. Barriers and facilitators to dialectical behavior therapy skills groups in the Veterans Health Administration. Mil Med. 2024;189(5-6):1055-1063. doi:10.1093/milmed/ usad123
- Landes SJ, Rodriguez AL, Smith BN, et al. Barriers, facilitators, and benefits of implementation of dialectical behavior therapy in routine care: results from a national program evaluation survey in the Veterans Health Administration. Transl Behav Med. 2017;7(4):832-844. doi:10.1007/s13142-017-0465-5
- Walker J, Betthauser LM, Green K, Landes SJ, Stacy M. Suicide Prevention 2.0 Clinical Telehealth Program: Evidence- Based Treatment in the Veterans Health Administration. April 28, 2024. Accessed February 28, 2025. https://www.youtube.com/watch?v=fFsDzkg0SR0
- Gunderson J, Masland S, Choi-Kain L. Good psychiatric management: a review. Curr Opin Psychol. 2018;21:127- 131. doi:10.1016/j.copsyc.2017.12.006
- Kramer U. Good-enough therapy: a review of the empirical basis of good psychiatric management. Am J Psychother. 2025;78(1): 11-15. doi:10.1176/appi .psychotherapy.20230041
- Visdómine-Lozano JC. Contextualist perspectives in the treatment of antisocial behaviors and offending: a comparative review of FAP, ACT, DBT, and MDT. Trauma Violence Abuse. 2022;23(1):241-254. doi:10.1177/1524838020939509
- Drago A, Marogna C, Jørgen Søgaard H. A review of characteristics and treatments of the avoidant personality disorder. Could the DBT be an option? Int J Psychol Psychoanal. 2016;2(1):013.
- Finch EF, Choi-Kain LW, Iliakis EA, Eisen JL, Pinto A. Good psychiatric management for obsessive–compulsive personality disorder. Curr Behav Neurosci Rep. 2021;8:160-171. doi:10.1007/s40473-021-00239-4
- Miller TW, Kraus RF. Modified dialectical behavior therapy and problem solving for obsessive-compulsive personality disorder. Journal Contemp Psychother. 2007;37:79-85. doi:10.1007/s10879-006-9039-4
- Bozzatello P, Rocca P, De Rosa ML, Bellino S. Current and emerging medications for borderline personality disorder: is pharmacotherapy alone enough? Expert Opin Pharmacother. 2020;21(1):47-61.doi:10.1080/14656566 .2019.1686482
- Sand P, Derviososki E, Kollia S, Strand J, Di Leone F. Psychiatrists’ perspectives on prescription decisions for patients with personality disorders. J Pers Disord. 2024;38(3):225-240. doi:10.1521/pedi.2024.38.3.225
- Kane JM, Leucht S, Carpenter D, Docherty JP; Expert Consensus Panel for Optimizing Pharmacologic Treatment of Psychotic Disorders. The expert consensus guideline series. Optimizing pharmacologic treatment of psychotic disorders. Introduction: Methods, commentary, and summary. J Clin Psychiatry. 2003;64 Suppl 12:5-19.
- Nierenberg AA, Agustini B, Köhler-Forsberg O, et al. Diagnosis and treatment of bipolar disorder: a review. JAMA. 2023;330(14):1370-1380. doi:10.1001 /jama.2023.18588
- Köck P, Walter M. Personality disorder and substance use disorder–an update. Ment Health Prev. 2018;12:82- 89. doi:10.1016/J.MHP.2018.10.003
- Garb HN. Race bias and gender bias in the diagnosis of psychological disorders. Clin Psych Rev. 2021;90:102087. doi:10.1016/j.cpr.2021.102087
- Debast I, van Alphen SPJ, Rossi G, et al. Personality traits and personality disorders in late middle and old age: do they remain stable? A literature review. Clin Gerontol. 2014;37(3):253-271.doi:10.1080/07317115 .2014.885917
- Penders KAP, Peeters IGP, Metsemakers JFM, van Alphen SPJ. Personality disorders in older adults: a review of epidemiology, assessment, and treatment. Curr Psychiatry Rep. 2020;22(3):1-14. doi:10.1007/s11920-020- 1133-x
- Videler AC, Hutsebaut J, Schulkens JEM, Sobczak S, van Alphen SPJ. A life span perspective on borderline personality disorder. Curr Psychiatry Rep. 2019;21(7) :1-8. doi:10.1007/s11920-019-1040-1
- Wakefield JC. DSM-5 and the general definition of personality disorder. Clin Soc Work J. 2013;41(2):168-183. doi:10.1007/s10615-012-0402-5
- US Census Bureau. 2022 American Community Survey 1-year. Accessed February 28, 2025. https://data.census.gov/table/ACSST1Y2022.S2101?q=Veterans&y=2022comparison
Personality disorders (PDs) are enduring patterns of internal experience and behavior that differ from cultural norms and expectations, are inflexible and pervasive, have their onset in adolescence or early adulthood, and lead to distress or impairment. Ten PDs are included in the Diagnostic and Statistical Manual of Mental Disorders (Fifth Edition): paranoid, schizoid, schizotypal, borderline, antisocial, histrionic, narcissistic, avoidant, dependent, and obsessive-compulsive.1 These disorders impose a high burden on patients, families, health care systems, and broader economic systems.2,3 Up to 1 in 7 persons in the community and 50% of those receiving outpatient mental health treatment experience a PD.4,5 These conditions are associated with an increased risk of adverse events, including suicide attempt and death by suicide, criminal-legal involvement, homelessness, substance use, underemployment, relational issues, and high utilization of psychiatric services.6-9 PDs are routinely underassessed, underdocumented, and undertreated in clinical settings, and consistently receive less research funding than other, less prevalent forms of psychopathology. 10-12 As a result, there is limited understanding of clinical needs of individuals experiencing PDs.
MILITARY VETERANS WITH PERSONALITY DISORDERS
Underacknowledgment of PDs and their associated difficulties may be especially pronounced in veteran populations. Due to longstanding etiological theories that implicate childhood trauma and adolescent onset in pathology development, PDs are traditionally considered pre-existing conditions or developmental abnormalities by the US Department of Defense and US Department of Veterans Affairs (VA). As a result, PDs are therefore deemed incompatible with military service and ineligible for service-connected disability benefits.13-15 Such determinations allowed PD pathology to be used as grounds for discharge for 26,000 service members from 2001 to 2007, or 2.6% of total enlisted discharges during that period.13,15,16
Despite this structural discrimination, recent research suggests veterans may be more likely to experience PD pathology than the general population.17 For example, a 2021 epidemiological survey in a community-based veteran sample found elevated rates of borderline, antisocial, and schizotypal PDs (6%-13%).6 In contrast, only 0.8% to 5.0% of veteran electronic health records (EHRs) have a documented PD diagnosis.8,18,19 Such elevations in PD pathology within veteran samples imply either a disproportionately high prevalence among enlistees (and therefore missed during recruitment procedures) or onset following military service, possibly due to exposure to traumatic events and/ or occupational stress.17 Due to the relative infancy of research in this area and a lack of longitudinal studies, etiology and course of illness for personality pathology in veterans remains largely unclear.
Structural underacknowledgment of PDs among military personnel has contributed to their underrepresentation in research on veteran populations. PD-focused research with veterans is rare, despite a rapid increase in broader empirical attention paid to these conditions in nonveteran samples.20 A recent meta-analysis of veterans with PDs identified 27 studies that included basic prevalence statistics. PDs were rarely a primary focus for these studies, and most were limited to veterans seen in Veterans Health Administration (VHA) settings.17 The literature also paints a bleak picture, suggesting veterans who experience PDs are at higher risk for suicide attempt and death by suicide, criminal-legal involvement, and homelessness. They also tend to experience more severe comorbid psychopathological symptoms and more often use high-intensity mental health services (eg, care within emergency departments or psychiatric inpatient settings) than veterans without PD pathology.6,8,18,19,21 However, PD pathology does not appear to impede the effectiveness of treatment for veterans.22-24 The implications of PD pathology on broader psychosocial functioning and health care needs certify a need for additional research that examines patterns of personality pathology, particularly in veterans outside the VHA.
METHODS
This study aims to enhance understanding of veterans affected by PDs and offer insight and guidance for treatment of these conditions in federal and nonfederal treatment settings. Previous research has been largely limited to VHA care-receiving samples; the longstanding stigma against PDs by the US military and VA may contribute to biased diagnosis and documentation of PDs in these settings. A large sample of veterans receiving community-based mental health care was therefore used to explore aims of the current study. This study specifically examined demographic patterns, diagnostic comorbidity, psychosocial outcomes, and treatment care settings among veterans with and without a PD diagnosis. Consistent with previous research, we hypothesized that veterans with a PD diagnosis would have more severe mental health comorbidities, poorer psychosocial outcomes, and receive care in higher intensity settings relative to veterans without a diagnosis.
Data for the sample were drawn from the Mental Health Client-Level Data, a publicly available national dataset of nearly 7 million patients who received mental health treatment services provided or funded through state mental health agencies in 2022.25 The analytic sample included about 2.5 million patients for whom veteran status and data around the presence or absence of a PD diagnosis were available. Of these patients, 104,198 were identified as veterans. Veteran patients were identified as predominantly male (63%), White (71%), non-Hispanic (90%), and never married (54%).
Measures
The parent dataset included demographic, clinical, and psychosocial outcome information reported by treatment facilities to individual state administrative systems for each patient who received services. To protect patient privacy, only nonprotected health information is included, and efforts were made throughout compilation of the parent dataset to ensure patient privacy (eg, limiting detail of information disseminated for public access). Because the parent dataset does not include protected health information, studies using these data are considered exempt from institutional review board oversight.
Demographic information. This study reviewed veteran status, sex, race, ethnicity, age, education, and marital status. Veteran status was defined by whether the patient was aged ≥ 18 years and had previously served (but was not currently serving) in the military. Patients with a history of service in the National Guard or Military Reserves were only classified as veterans if they had been called or ordered to active duty while serving. Sex was operationalized dichotomously as male or female; no patients were identified as intersex, transgender, or other gender identities.
Clinical information. Up to 3 mental health diagnoses were reported for each patient and included the following disorders: personality, trauma and attention-deficit/hyperactivity, stressor, anxiety, conduct, delirium/dementia, bipolar, depressive, oppositional defiant, pervasive developmental, schizophrenia or other psychotic, and alcohol or substance use. Mental health diagnosis categories were generated for the parent dataset by grouping diagnostic codes corresponding to each category. To protect patient privacy, more detailed diagnostic information was not available as part of the parent dataset. Although the American Psychiatric Association recognizes 10 distinct PDs, the exact nature of PD diagnoses was not included within the parent dataset. PD diagnoses were coded to reflect the presence or absence of any such diagnosis.
A substance use problem designation was also provided for patients according to various identification methods, including substance use disorder (SUD) diagnosis, substance use screening results, enrollment in a substance use program, substance use survey, service claims information, and other related sources of information. A severe mental illness or serious emotional disturbance designation was provided for patients meeting state definitions of these designations. Context(s) of service provision were coded as inpatient state psychiatric hospital, community-based program, residential treatment center, judicial institution, or other psychiatric inpatient setting.
Psychosocial outcome information. Patient employment and residential status were also included in analyses. Each reflected status at the time of discharge from services or end of reporting period; employment status was only provided for patients receiving treatment in community-based programs.
Data Analysis
Descriptive statistics and X2 analyses were used to compare demographic, clinical, and psychosocial outcome variables between patients with and without PD diagnoses. These analyses were calculated for both the 104,198 veterans and the 2,222,306 nonveterans aged ≥ 18 years in the dataset. Given the sample size, a conservative α of .01 was used to determine statistical significance.
RESULTS
In this sample of persons receiving state-funded mental health care, veterans were significantly less likely than nonveterans to have a documented PD diagnosis (2.1% vs 3.6%, X2 [1] = 647.49; P < .01). PD diagnoses were more common among White (risk ratio [RR], 1.11), non-Hispanic (RR, 1.03) veterans who were in middle to late adulthood (RR, 1.16-1.40), more educated (RR, 1.35), and divorced or widowed (RR, 1.43), and less common among Black/African American (RR, 0.78) or Puerto Rican (RR, 0.32) veterans who were in early adulthood (RR, 0.31-0.79), less educated (RR, 0.64-0.89), and currently married (RR, 0.89) or never married (RR, 0.86). Veteran men and women were equally likely to have a PD diagnosis (RR, 1.03) (Table 1). Among nonveterans, men were less likely than women to have a PD diagnosis (RR, 0.79), and PD diagnoses were most common among persons in middle adulthood (RR, 1.06-1.15) (eAppendix 1).


Veterans with a PD diagnosis were more likely than those without a diagnosis to have more diagnoses (RR, 2.96-8.49) and to have comorbid trauma or related stressor (RR, 1.33), or bipolar (RR, 1.56) or psychotic (RR, 1.15) disorder diagnoses, but less likely to have comorbid depressive disorder (RR, 0.82). Although veterans with and without a PD diagnosis were similarly likely to have a comorbid SUD (RR, 1.13), those with a PD diagnosis were significantly less likely to be assigned a substance use problem designation (RR, 0.78). PD diagnosis was also more common among veterans who received services in state psychiatric hospitals (RR, 3.05), community-based clinics (RR, 1.06), and judicial institutions (RR, 6.33) and less common among those who received services in other psychiatric inpatient settings (RR, 0.30). No differences were observed for residential treatment settings (RR, 0.79). Among nonveterans, a PD diagnosis was associated with slightly greater odds of a substance use designation (RR, 1.03) (eAppendix 2).

Veterans with a PD diagnosis were also less likely to have full-time employment (RR, 0.73) and more likely to have undifferentiated employment (RR, 2.00) or to be removed from the labor force (RR, 1.35). Veterans with a PD diagnosis were also more likely to reside in nontraditional living conditions (RR, 1.42) and less likely to be residing in a private residence (RR, 0.98), compared with those without PD diagnosis. The rates of homelessness were similar for veterans with and without a PD diagnosis (RR, 0.90) (Table 2). These patterns were similar among nonveterans.

DISCUSSION
This study examined the rate and correlates of PD diagnosis among a large, community-based sample of veterans receiving state-funded mental health care. About 2% of veterans in this sample had a PD diagnosis, with diagnoses more common among veterans who were White, non-Hispanic, aged ≥ 45 years, with higher education, divorced or widowed, also diagnosed with trauma-related, bipolar, and/or psychotic disorders, underemployed, nontraditionally housed, and receiving treatment in state psychiatric hospital, community-based clinic, or judicial system settings.
The observed rate of PD diagnosis in this study aligns with what is typically observed in VHA EHRs.8,18,19 However, the rate is notably lower than prevalence estimates for psychiatric outpatient settings (about 50%) and in meta-analyses of prevalence among veterans (0.8%-23% for each of the 10 PDs).4,17,26 Longstanding stigma against PDs may contribute to underdiagnosis. For example, many clinicians are concerned that documentation or disclosure of a PD will interfere with the patient’s ability to access treatment due to stigma and discrimination.27,28 These fears are not unfounded; even among clinicians, PDs are commonly considered untreatable, and many individuals with PDs are denied access to evidence-based treatments due to the diagnosis.29 In a 2016 survey of community psychiatrists, nearly 1 in 4 reported that they avoid taking patients with a borderline PD diagnosis in their caseloads.28 To date, no studies have been conducted to explore clinicians’ willingness to accept patients with other PDs or, specifically, among veterans.
Despite such widespread stigma, research suggests clinicians' negative attitudes toward PDs can be decreased through antistigma campaigns.30 However, it remains unclear if such efforts also contribute to an increase in clinicians’ willingness to document PD diagnoses. Without accurate identification and documentation, the field’s understanding of PDs will remain limited.
In the current study, veterans with PD diagnoses tended to present with more complex and severe psychiatric comorbidities compared to veterans without such diagnoses. Observed comorbidity of PDs (particularly borderline PD) with trauma-related and bipolar disorders is well established.8 Conversely, co-occurring personality and psychotic disorders—which comprise 16% of veterans with a PD diagnosis in the sample in this study—are not consistently examined in the literature. A 2022 examination of veterans receiving VHA care suggested 12% and 13% of those with a PD diagnosis documented in their EHR also had documented schizophrenia or another psychotic disorder, respectively. PD diagnoses were associated with 6.88- and 9.80-fold increases in risk for comorbid schizophrenia and other psychotic disorder diagnoses, respectively.8 Similarly, a recent longitudinal study of nearly 2 million Swedish individuals suggested borderline PD is specifically associated with a > 24-times greater risk of having a comorbid psychotic disorder.31 It is therefore possible that the comorbidity between personality and psychotic disorders is quite common despite its relative lack of attention in empirical research.
Veterans with PD diagnoses in this study were also more likely to experience substandard housing, employment challenges, and receive treatment through judicial institutions than those without a PD diagnosis. Such findings are consistent with previous research demonstrating the substantial psychosocial challenges associated with PD diagnosis, even after controlling for comorbid conditions.7,9 Veterans with PDs may benefit from specialized case management and support to facilitate stable housing and employment and to mitigate the risk of judicial involvement. Some research suggests veterans with PDs may be less likely to gain competitive employment after participating in VA therapeutic and supportive employment services programs, suggesting standard programming may be less suitable for this population.32 Similarly, other research suggests individuals with PDs may benefit more from specialized, intensive services than standard clinical case management.33 Future research may therefore benefit from clarifying the degree to which adaptations to standard programming could yield beneficial effects for persons with PD diagnoses.
Implications
Cumulatively, the results of this study attest to the necessity for transdiagnostic treatment planning that includes close collaboration between psychotherapeutic, pharmacological, and case management services. Some psychotherapy models for PDs, such as dialectical behavior therapy (DBT), which includes a combination of group skills training, individual therapy, as-needed phone coaching, and therapist consultation, may be successfully adapted to include this collaboration.34-36 However, implementation of such comprehensive programming often requires extensive clinician training and coordination of resources, which poses implementation challenges.37-39 In 2021, the VHA began large-scale implementation of PD-specific psychotherapy for veterans with recent suicidal self-directed violence and borderline PD, including DBT, though to date results remain unclear.40 Generalist approaches, such as good psychiatric management (GPM), which emphasizes emotional validation, practical problem solving, realistic goal setting, and relationship functioning within the context of standard care appointments, may be more easily implemented in community care settings due to lesser training and resource requirements and can also be adapted to include needed elements of care coordination.41,42 Both DBT and GPM were initially developed for the treatment of borderline PD. Although DBT has also demonstrated some effectiveness in the treatment of antisocial PD, potential applications of DBT and GPM to other PDs remain largely underdeveloped.43-46
There are no widely accepted medications for the treatment of PDs. Pharmacotherapy for these conditions typically consists of individualized approaches informed by personal experience that attempt to balance targeting of specific symptoms while minimizing polypharmacy and potential risks (eg, overdose or addiction).47,48 Despite this, pharmacotherapy is often considered a necessary component in the treatment of bipolar and psychotic disorders, both common comorbidities of PDs found in veterans in this study.49,50 Careful consideration of complex comorbidities and pharmacotherapy needs is warranted in the treatment of veterans with PDs. Future research may benefit from clarifying clinical guidelines around pharmacotherapy, particularly for observed comorbidities of PDs to trauma, bipolar, and psychotic disorders.
It is important to note the discrepancies in the results of this study surrounding patient substance use. The results suggest a negligible or inverse association between the likelihood of a PD diagnosis and difficulties with substance use among the veterans in this study. However, the unexpectedly low rate of SUD diagnoses (< 6%) suggests that they were likely underdocumented. Research suggests a strong association between personality and SUDs in both veteran and civilian samples.6,51 Results suggesting a lower prevalence of substance use difficulties among treatment-seeking veterans with PDs should be interpreted with great caution.
Demographically, PD diagnoses were more common among veterans who were White, non-Hispanic, and aged ≥ 45 years, and less common among veterans who were Black/ African American, mixed/unspecified race, Puerto Rican or other non-Mexican Hispanic ethnicity, or aged < 35 years. No significant sex-based differences were observed. These patterns are consistent with research suggesting individuals who identify as Black may be less likely than individuals who identify as White to report PD symptoms, meet criteria for a PD, and have a PD diagnosed even when it is warranted.52
The findings observed in this study with respect to age, however, are notably inconsistent with the literature. Previous research typically suggests a negative association between age and PD pathology; however, a 2020 review of PDs in older adults by Penders et al suggests a prevalence of 11% to 15% in this population.53,54 Research into PDs most often focuses on adolescent and early adulthood developmental periods, limiting insight into the phenomenology of PDs in middle to late adulthood.55 Further, most research into PDs among geriatric populations has focused on psychometric assessment rather than practical treatment guidance.54 However, in this study, elevated risk for PD diagnoses was salient throughout middle to late adulthood among veterans; similar, albeit less pronounced patterns were also observed for elevated risk of PD diagnosis in middle adulthood among nonveterans. Such findings suggest clarifying the phenomenology and treatment needs of individuals with PDs in middle to late adulthood may have particularly salient implications for the mental health care of veterans affected by these conditions. As the veteran population advances in age, these needs will present unique challenges if health care systems are unprepared to effectively address them.
Limitations
This study is characterized by several strengths, most notably its use of a large dataset recently collected on a national scale. Few studies outside of the VHA system include samples of > 100,000 treatment-seeking veterans collected on a national scale. Nevertheless, results should be understood within the context of several methodological limitations. However, the dataset was limited to the first 3 diagnoses documented in patients’ EHRs, and many patients had no listed diagnoses. Patients with complex comorbidities may have > 3 diagnoses; for these individuals, data provided an incomplete picture of clinical presentation. This is especially relevant for individuals with PDs, who tend to meet criteria for a range of comorbid conditions.8,10 The now dated practice of listing PDs on Axis II also increases the chance of clinicians listing PDs after conditions traditionally listed on Axis I (eg, major depressive disorder) in patient charts.56 This study’s inclusion of only the first 3 listed diagnoses likely underestimated true PD diagnosis prevalence.
The results of this study must be interpreted as reflecting the prevalence and correlates of receiving a PD diagnosis rather than meeting diagnostic criteria for a PD. Relatedly, PD diagnoses were reported as a single construct, limiting insight into prevalence and correlates of individual PD diagnoses (eg, borderline vs paranoid PDs). Meta-analyses estimates suggest PD prevalence among veterans is likely much higher than observed in this study.17 Stigma continues to discourage clinicians from documenting and disclosing PD diagnoses even when warranted.27,28 Continued research should aim to clarify conditions (eg, patient presentation, stigma, or institutional culture) contributing to documentation of PD diagnoses. Given the cross-sectional nature of this study, results cannot speak to longitudinal treatment outcomes or prognosis of persons receiving a PD diagnosis.
Despite its large sample size and national representation, the sampling strategy of this study could have contributed to idiosyncrasies in the dataset. Restriction of data to the persons receiving state-funded mental health services introduces a notable bias to the composition of the sample, which is likely comprised of a disproportionately high number of Medicaid recipients, students, and individuals with chronic illnesses and underrepresentation of persons who pay for mental health services using private insurance or private pay arrangements. As such, although socioeconomic information was not provided within this dataset, one can presume a generally lower socioeconomic status among study participants compared to the community at large. This study also included a proportionally small sample of veterans (3.6% compared to about 6.2% in the broader US population), suggesting veterans may have been underrepresented or underidentified in surveyed mental health care settings.57 This study also did not include data around service in active-duty military, national guard, or military reserves; a greater proportion of the sample likely had a history of military service than was represented by veteran status designation. Further, the proportionally high sample of individuals with severe mental illness suggests a likely overrepresentation of such conditions in surveyed settings.
Institutional differences in the practice of assigning diagnoses likely limited statistical power to detect potentially meaningful associations and effects. Structural influences, such as stigma and institutional culture, may have notable effects on documentation practices, particularly for PDs. Future research should aim to replicate observed associations using more controlled diagnostic procedures.
Lastly, even with the use of a more conservative α and a focus on effect sizes to guide interpretation of results, use of multiple bivariate analyses can be presumed to have increased the likelihood of type I error. Given the limited prior research in this area, an exploratory approach to statistical analysis was considered warranted to maximize opportunity for identifying areas in need of additional empirical attention. Continued research using more conservative statistical approaches (eg, multivariate analyses) is needed to determine replicability and generalizability of observed results.
CONCLUSIONS
This study examined the prevalence and correlates of PD diagnoses in a national sample of veterans receiving community-based, state-funded mental health care. About 2% received a PD diagnosis, with diagnoses most common among veterans who were White, non-Hispanic, aged ≥ 45 years, also diagnosed with trauma-based, bipolar, and/or psychotic disorders, underemployed, nontraditionally housed, and receiving treatment in a state psychiatric hospital or judicial system setting. The results attest to a necessity for transdiagnostic treatment planning and care coordination for this population, with particular attention to psychosocial stressors.
Personality disorders (PDs) are enduring patterns of internal experience and behavior that differ from cultural norms and expectations, are inflexible and pervasive, have their onset in adolescence or early adulthood, and lead to distress or impairment. Ten PDs are included in the Diagnostic and Statistical Manual of Mental Disorders (Fifth Edition): paranoid, schizoid, schizotypal, borderline, antisocial, histrionic, narcissistic, avoidant, dependent, and obsessive-compulsive.1 These disorders impose a high burden on patients, families, health care systems, and broader economic systems.2,3 Up to 1 in 7 persons in the community and 50% of those receiving outpatient mental health treatment experience a PD.4,5 These conditions are associated with an increased risk of adverse events, including suicide attempt and death by suicide, criminal-legal involvement, homelessness, substance use, underemployment, relational issues, and high utilization of psychiatric services.6-9 PDs are routinely underassessed, underdocumented, and undertreated in clinical settings, and consistently receive less research funding than other, less prevalent forms of psychopathology. 10-12 As a result, there is limited understanding of clinical needs of individuals experiencing PDs.
MILITARY VETERANS WITH PERSONALITY DISORDERS
Underacknowledgment of PDs and their associated difficulties may be especially pronounced in veteran populations. Due to longstanding etiological theories that implicate childhood trauma and adolescent onset in pathology development, PDs are traditionally considered pre-existing conditions or developmental abnormalities by the US Department of Defense and US Department of Veterans Affairs (VA). As a result, PDs are therefore deemed incompatible with military service and ineligible for service-connected disability benefits.13-15 Such determinations allowed PD pathology to be used as grounds for discharge for 26,000 service members from 2001 to 2007, or 2.6% of total enlisted discharges during that period.13,15,16
Despite this structural discrimination, recent research suggests veterans may be more likely to experience PD pathology than the general population.17 For example, a 2021 epidemiological survey in a community-based veteran sample found elevated rates of borderline, antisocial, and schizotypal PDs (6%-13%).6 In contrast, only 0.8% to 5.0% of veteran electronic health records (EHRs) have a documented PD diagnosis.8,18,19 Such elevations in PD pathology within veteran samples imply either a disproportionately high prevalence among enlistees (and therefore missed during recruitment procedures) or onset following military service, possibly due to exposure to traumatic events and/ or occupational stress.17 Due to the relative infancy of research in this area and a lack of longitudinal studies, etiology and course of illness for personality pathology in veterans remains largely unclear.
Structural underacknowledgment of PDs among military personnel has contributed to their underrepresentation in research on veteran populations. PD-focused research with veterans is rare, despite a rapid increase in broader empirical attention paid to these conditions in nonveteran samples.20 A recent meta-analysis of veterans with PDs identified 27 studies that included basic prevalence statistics. PDs were rarely a primary focus for these studies, and most were limited to veterans seen in Veterans Health Administration (VHA) settings.17 The literature also paints a bleak picture, suggesting veterans who experience PDs are at higher risk for suicide attempt and death by suicide, criminal-legal involvement, and homelessness. They also tend to experience more severe comorbid psychopathological symptoms and more often use high-intensity mental health services (eg, care within emergency departments or psychiatric inpatient settings) than veterans without PD pathology.6,8,18,19,21 However, PD pathology does not appear to impede the effectiveness of treatment for veterans.22-24 The implications of PD pathology on broader psychosocial functioning and health care needs certify a need for additional research that examines patterns of personality pathology, particularly in veterans outside the VHA.
METHODS
This study aims to enhance understanding of veterans affected by PDs and offer insight and guidance for treatment of these conditions in federal and nonfederal treatment settings. Previous research has been largely limited to VHA care-receiving samples; the longstanding stigma against PDs by the US military and VA may contribute to biased diagnosis and documentation of PDs in these settings. A large sample of veterans receiving community-based mental health care was therefore used to explore aims of the current study. This study specifically examined demographic patterns, diagnostic comorbidity, psychosocial outcomes, and treatment care settings among veterans with and without a PD diagnosis. Consistent with previous research, we hypothesized that veterans with a PD diagnosis would have more severe mental health comorbidities, poorer psychosocial outcomes, and receive care in higher intensity settings relative to veterans without a diagnosis.
Data for the sample were drawn from the Mental Health Client-Level Data, a publicly available national dataset of nearly 7 million patients who received mental health treatment services provided or funded through state mental health agencies in 2022.25 The analytic sample included about 2.5 million patients for whom veteran status and data around the presence or absence of a PD diagnosis were available. Of these patients, 104,198 were identified as veterans. Veteran patients were identified as predominantly male (63%), White (71%), non-Hispanic (90%), and never married (54%).
Measures
The parent dataset included demographic, clinical, and psychosocial outcome information reported by treatment facilities to individual state administrative systems for each patient who received services. To protect patient privacy, only nonprotected health information is included, and efforts were made throughout compilation of the parent dataset to ensure patient privacy (eg, limiting detail of information disseminated for public access). Because the parent dataset does not include protected health information, studies using these data are considered exempt from institutional review board oversight.
Demographic information. This study reviewed veteran status, sex, race, ethnicity, age, education, and marital status. Veteran status was defined by whether the patient was aged ≥ 18 years and had previously served (but was not currently serving) in the military. Patients with a history of service in the National Guard or Military Reserves were only classified as veterans if they had been called or ordered to active duty while serving. Sex was operationalized dichotomously as male or female; no patients were identified as intersex, transgender, or other gender identities.
Clinical information. Up to 3 mental health diagnoses were reported for each patient and included the following disorders: personality, trauma and attention-deficit/hyperactivity, stressor, anxiety, conduct, delirium/dementia, bipolar, depressive, oppositional defiant, pervasive developmental, schizophrenia or other psychotic, and alcohol or substance use. Mental health diagnosis categories were generated for the parent dataset by grouping diagnostic codes corresponding to each category. To protect patient privacy, more detailed diagnostic information was not available as part of the parent dataset. Although the American Psychiatric Association recognizes 10 distinct PDs, the exact nature of PD diagnoses was not included within the parent dataset. PD diagnoses were coded to reflect the presence or absence of any such diagnosis.
A substance use problem designation was also provided for patients according to various identification methods, including substance use disorder (SUD) diagnosis, substance use screening results, enrollment in a substance use program, substance use survey, service claims information, and other related sources of information. A severe mental illness or serious emotional disturbance designation was provided for patients meeting state definitions of these designations. Context(s) of service provision were coded as inpatient state psychiatric hospital, community-based program, residential treatment center, judicial institution, or other psychiatric inpatient setting.
Psychosocial outcome information. Patient employment and residential status were also included in analyses. Each reflected status at the time of discharge from services or end of reporting period; employment status was only provided for patients receiving treatment in community-based programs.
Data Analysis
Descriptive statistics and X2 analyses were used to compare demographic, clinical, and psychosocial outcome variables between patients with and without PD diagnoses. These analyses were calculated for both the 104,198 veterans and the 2,222,306 nonveterans aged ≥ 18 years in the dataset. Given the sample size, a conservative α of .01 was used to determine statistical significance.
RESULTS
In this sample of persons receiving state-funded mental health care, veterans were significantly less likely than nonveterans to have a documented PD diagnosis (2.1% vs 3.6%, X2 [1] = 647.49; P < .01). PD diagnoses were more common among White (risk ratio [RR], 1.11), non-Hispanic (RR, 1.03) veterans who were in middle to late adulthood (RR, 1.16-1.40), more educated (RR, 1.35), and divorced or widowed (RR, 1.43), and less common among Black/African American (RR, 0.78) or Puerto Rican (RR, 0.32) veterans who were in early adulthood (RR, 0.31-0.79), less educated (RR, 0.64-0.89), and currently married (RR, 0.89) or never married (RR, 0.86). Veteran men and women were equally likely to have a PD diagnosis (RR, 1.03) (Table 1). Among nonveterans, men were less likely than women to have a PD diagnosis (RR, 0.79), and PD diagnoses were most common among persons in middle adulthood (RR, 1.06-1.15) (eAppendix 1).


Veterans with a PD diagnosis were more likely than those without a diagnosis to have more diagnoses (RR, 2.96-8.49) and to have comorbid trauma or related stressor (RR, 1.33), or bipolar (RR, 1.56) or psychotic (RR, 1.15) disorder diagnoses, but less likely to have comorbid depressive disorder (RR, 0.82). Although veterans with and without a PD diagnosis were similarly likely to have a comorbid SUD (RR, 1.13), those with a PD diagnosis were significantly less likely to be assigned a substance use problem designation (RR, 0.78). PD diagnosis was also more common among veterans who received services in state psychiatric hospitals (RR, 3.05), community-based clinics (RR, 1.06), and judicial institutions (RR, 6.33) and less common among those who received services in other psychiatric inpatient settings (RR, 0.30). No differences were observed for residential treatment settings (RR, 0.79). Among nonveterans, a PD diagnosis was associated with slightly greater odds of a substance use designation (RR, 1.03) (eAppendix 2).

Veterans with a PD diagnosis were also less likely to have full-time employment (RR, 0.73) and more likely to have undifferentiated employment (RR, 2.00) or to be removed from the labor force (RR, 1.35). Veterans with a PD diagnosis were also more likely to reside in nontraditional living conditions (RR, 1.42) and less likely to be residing in a private residence (RR, 0.98), compared with those without PD diagnosis. The rates of homelessness were similar for veterans with and without a PD diagnosis (RR, 0.90) (Table 2). These patterns were similar among nonveterans.

DISCUSSION
This study examined the rate and correlates of PD diagnosis among a large, community-based sample of veterans receiving state-funded mental health care. About 2% of veterans in this sample had a PD diagnosis, with diagnoses more common among veterans who were White, non-Hispanic, aged ≥ 45 years, with higher education, divorced or widowed, also diagnosed with trauma-related, bipolar, and/or psychotic disorders, underemployed, nontraditionally housed, and receiving treatment in state psychiatric hospital, community-based clinic, or judicial system settings.
The observed rate of PD diagnosis in this study aligns with what is typically observed in VHA EHRs.8,18,19 However, the rate is notably lower than prevalence estimates for psychiatric outpatient settings (about 50%) and in meta-analyses of prevalence among veterans (0.8%-23% for each of the 10 PDs).4,17,26 Longstanding stigma against PDs may contribute to underdiagnosis. For example, many clinicians are concerned that documentation or disclosure of a PD will interfere with the patient’s ability to access treatment due to stigma and discrimination.27,28 These fears are not unfounded; even among clinicians, PDs are commonly considered untreatable, and many individuals with PDs are denied access to evidence-based treatments due to the diagnosis.29 In a 2016 survey of community psychiatrists, nearly 1 in 4 reported that they avoid taking patients with a borderline PD diagnosis in their caseloads.28 To date, no studies have been conducted to explore clinicians’ willingness to accept patients with other PDs or, specifically, among veterans.
Despite such widespread stigma, research suggests clinicians' negative attitudes toward PDs can be decreased through antistigma campaigns.30 However, it remains unclear if such efforts also contribute to an increase in clinicians’ willingness to document PD diagnoses. Without accurate identification and documentation, the field’s understanding of PDs will remain limited.
In the current study, veterans with PD diagnoses tended to present with more complex and severe psychiatric comorbidities compared to veterans without such diagnoses. Observed comorbidity of PDs (particularly borderline PD) with trauma-related and bipolar disorders is well established.8 Conversely, co-occurring personality and psychotic disorders—which comprise 16% of veterans with a PD diagnosis in the sample in this study—are not consistently examined in the literature. A 2022 examination of veterans receiving VHA care suggested 12% and 13% of those with a PD diagnosis documented in their EHR also had documented schizophrenia or another psychotic disorder, respectively. PD diagnoses were associated with 6.88- and 9.80-fold increases in risk for comorbid schizophrenia and other psychotic disorder diagnoses, respectively.8 Similarly, a recent longitudinal study of nearly 2 million Swedish individuals suggested borderline PD is specifically associated with a > 24-times greater risk of having a comorbid psychotic disorder.31 It is therefore possible that the comorbidity between personality and psychotic disorders is quite common despite its relative lack of attention in empirical research.
Veterans with PD diagnoses in this study were also more likely to experience substandard housing, employment challenges, and receive treatment through judicial institutions than those without a PD diagnosis. Such findings are consistent with previous research demonstrating the substantial psychosocial challenges associated with PD diagnosis, even after controlling for comorbid conditions.7,9 Veterans with PDs may benefit from specialized case management and support to facilitate stable housing and employment and to mitigate the risk of judicial involvement. Some research suggests veterans with PDs may be less likely to gain competitive employment after participating in VA therapeutic and supportive employment services programs, suggesting standard programming may be less suitable for this population.32 Similarly, other research suggests individuals with PDs may benefit more from specialized, intensive services than standard clinical case management.33 Future research may therefore benefit from clarifying the degree to which adaptations to standard programming could yield beneficial effects for persons with PD diagnoses.
Implications
Cumulatively, the results of this study attest to the necessity for transdiagnostic treatment planning that includes close collaboration between psychotherapeutic, pharmacological, and case management services. Some psychotherapy models for PDs, such as dialectical behavior therapy (DBT), which includes a combination of group skills training, individual therapy, as-needed phone coaching, and therapist consultation, may be successfully adapted to include this collaboration.34-36 However, implementation of such comprehensive programming often requires extensive clinician training and coordination of resources, which poses implementation challenges.37-39 In 2021, the VHA began large-scale implementation of PD-specific psychotherapy for veterans with recent suicidal self-directed violence and borderline PD, including DBT, though to date results remain unclear.40 Generalist approaches, such as good psychiatric management (GPM), which emphasizes emotional validation, practical problem solving, realistic goal setting, and relationship functioning within the context of standard care appointments, may be more easily implemented in community care settings due to lesser training and resource requirements and can also be adapted to include needed elements of care coordination.41,42 Both DBT and GPM were initially developed for the treatment of borderline PD. Although DBT has also demonstrated some effectiveness in the treatment of antisocial PD, potential applications of DBT and GPM to other PDs remain largely underdeveloped.43-46
There are no widely accepted medications for the treatment of PDs. Pharmacotherapy for these conditions typically consists of individualized approaches informed by personal experience that attempt to balance targeting of specific symptoms while minimizing polypharmacy and potential risks (eg, overdose or addiction).47,48 Despite this, pharmacotherapy is often considered a necessary component in the treatment of bipolar and psychotic disorders, both common comorbidities of PDs found in veterans in this study.49,50 Careful consideration of complex comorbidities and pharmacotherapy needs is warranted in the treatment of veterans with PDs. Future research may benefit from clarifying clinical guidelines around pharmacotherapy, particularly for observed comorbidities of PDs to trauma, bipolar, and psychotic disorders.
It is important to note the discrepancies in the results of this study surrounding patient substance use. The results suggest a negligible or inverse association between the likelihood of a PD diagnosis and difficulties with substance use among the veterans in this study. However, the unexpectedly low rate of SUD diagnoses (< 6%) suggests that they were likely underdocumented. Research suggests a strong association between personality and SUDs in both veteran and civilian samples.6,51 Results suggesting a lower prevalence of substance use difficulties among treatment-seeking veterans with PDs should be interpreted with great caution.
Demographically, PD diagnoses were more common among veterans who were White, non-Hispanic, and aged ≥ 45 years, and less common among veterans who were Black/ African American, mixed/unspecified race, Puerto Rican or other non-Mexican Hispanic ethnicity, or aged < 35 years. No significant sex-based differences were observed. These patterns are consistent with research suggesting individuals who identify as Black may be less likely than individuals who identify as White to report PD symptoms, meet criteria for a PD, and have a PD diagnosed even when it is warranted.52
The findings observed in this study with respect to age, however, are notably inconsistent with the literature. Previous research typically suggests a negative association between age and PD pathology; however, a 2020 review of PDs in older adults by Penders et al suggests a prevalence of 11% to 15% in this population.53,54 Research into PDs most often focuses on adolescent and early adulthood developmental periods, limiting insight into the phenomenology of PDs in middle to late adulthood.55 Further, most research into PDs among geriatric populations has focused on psychometric assessment rather than practical treatment guidance.54 However, in this study, elevated risk for PD diagnoses was salient throughout middle to late adulthood among veterans; similar, albeit less pronounced patterns were also observed for elevated risk of PD diagnosis in middle adulthood among nonveterans. Such findings suggest clarifying the phenomenology and treatment needs of individuals with PDs in middle to late adulthood may have particularly salient implications for the mental health care of veterans affected by these conditions. As the veteran population advances in age, these needs will present unique challenges if health care systems are unprepared to effectively address them.
Limitations
This study is characterized by several strengths, most notably its use of a large dataset recently collected on a national scale. Few studies outside of the VHA system include samples of > 100,000 treatment-seeking veterans collected on a national scale. Nevertheless, results should be understood within the context of several methodological limitations. However, the dataset was limited to the first 3 diagnoses documented in patients’ EHRs, and many patients had no listed diagnoses. Patients with complex comorbidities may have > 3 diagnoses; for these individuals, data provided an incomplete picture of clinical presentation. This is especially relevant for individuals with PDs, who tend to meet criteria for a range of comorbid conditions.8,10 The now dated practice of listing PDs on Axis II also increases the chance of clinicians listing PDs after conditions traditionally listed on Axis I (eg, major depressive disorder) in patient charts.56 This study’s inclusion of only the first 3 listed diagnoses likely underestimated true PD diagnosis prevalence.
The results of this study must be interpreted as reflecting the prevalence and correlates of receiving a PD diagnosis rather than meeting diagnostic criteria for a PD. Relatedly, PD diagnoses were reported as a single construct, limiting insight into prevalence and correlates of individual PD diagnoses (eg, borderline vs paranoid PDs). Meta-analyses estimates suggest PD prevalence among veterans is likely much higher than observed in this study.17 Stigma continues to discourage clinicians from documenting and disclosing PD diagnoses even when warranted.27,28 Continued research should aim to clarify conditions (eg, patient presentation, stigma, or institutional culture) contributing to documentation of PD diagnoses. Given the cross-sectional nature of this study, results cannot speak to longitudinal treatment outcomes or prognosis of persons receiving a PD diagnosis.
Despite its large sample size and national representation, the sampling strategy of this study could have contributed to idiosyncrasies in the dataset. Restriction of data to the persons receiving state-funded mental health services introduces a notable bias to the composition of the sample, which is likely comprised of a disproportionately high number of Medicaid recipients, students, and individuals with chronic illnesses and underrepresentation of persons who pay for mental health services using private insurance or private pay arrangements. As such, although socioeconomic information was not provided within this dataset, one can presume a generally lower socioeconomic status among study participants compared to the community at large. This study also included a proportionally small sample of veterans (3.6% compared to about 6.2% in the broader US population), suggesting veterans may have been underrepresented or underidentified in surveyed mental health care settings.57 This study also did not include data around service in active-duty military, national guard, or military reserves; a greater proportion of the sample likely had a history of military service than was represented by veteran status designation. Further, the proportionally high sample of individuals with severe mental illness suggests a likely overrepresentation of such conditions in surveyed settings.
Institutional differences in the practice of assigning diagnoses likely limited statistical power to detect potentially meaningful associations and effects. Structural influences, such as stigma and institutional culture, may have notable effects on documentation practices, particularly for PDs. Future research should aim to replicate observed associations using more controlled diagnostic procedures.
Lastly, even with the use of a more conservative α and a focus on effect sizes to guide interpretation of results, use of multiple bivariate analyses can be presumed to have increased the likelihood of type I error. Given the limited prior research in this area, an exploratory approach to statistical analysis was considered warranted to maximize opportunity for identifying areas in need of additional empirical attention. Continued research using more conservative statistical approaches (eg, multivariate analyses) is needed to determine replicability and generalizability of observed results.
CONCLUSIONS
This study examined the prevalence and correlates of PD diagnoses in a national sample of veterans receiving community-based, state-funded mental health care. About 2% received a PD diagnosis, with diagnoses most common among veterans who were White, non-Hispanic, aged ≥ 45 years, also diagnosed with trauma-based, bipolar, and/or psychotic disorders, underemployed, nontraditionally housed, and receiving treatment in a state psychiatric hospital or judicial system setting. The results attest to a necessity for transdiagnostic treatment planning and care coordination for this population, with particular attention to psychosocial stressors.
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed, text revision. American Psychiatric Association; 2022.
- Hastrup LH, Jennum P, Ibsen R, Kjellberg J, Simonsen E. Societal costs of borderline personality disorders: a matched-controlled nationwide study of patients and spouses. Acta Psychiatr Scand. 2019;140(5):458-467. doi:10.1111/acps.13094
- Sveen CA, Pedersen G, Ulvestad DA, Zahl KE, Wilberg T, Kvarstein EH. Societal costs of personality disorders: a cross-sectional multicenter study of treatment-seeking patients in mental health services in Norway. J Clin Psychol. 2023;79(8):1752-1769. doi:10.1002/jclp.23504
- Beckwith H, Moran PF, Reilly J. Personality disorder prevalence in psychiatric outpatients: a systematic literature review. Personal Ment Health. 2014;8(2):91-101. doi:10.1002/pmh.1252
- Eaton NR, Greene AL. Personality disorders: community prevalence and socio-demographic correlates. Curr Opin Psychol. 2018;21:28-32. doi:10.1016/j.copsyc. 2017.09.001
- Edwards ER, Barnes S, Govindarajulu U, Geraci J, Tsai J. Mental health and substance use patterns associated with lifetime suicide attempt, incarceration, and homelessness: a latent class analysis of a nationally representative sample of U.S. veterans. Psychol Serv. 2021;18(4):619-631. doi:10.1037/ser0000488
- Moran P, Romaniuk H, Coffey C, et al. The influence of personality disorder on the future mental health and social adjustment of young adults: a population-based cohort study. Lancet Psychiatry. 2016;3(7):636-645. doi:10.1016/S2215-0366(16)30029-3
- Nelson SM, Griffin CA, Hein TC, Bowersox N, McCarthy JF. Personality disorder and suicide risk among patients in the Veterans Affairs health system. Personal Disord. 2022;13(6):563-571. doi:10.1037/per0000521
- Skodol AE. Impact of personality pathology on psychosocial functioning. Curr Opin Psychol. 2018;21;33-38. doi:10.1016/j.copsyc.2017.09.006
- Tyrer P, Reed GM, Crawford MJ. Classification, assessment, prevalence, and effect of personality disorder. Lancet. 2015;385(9969):717-726. doi:10.1016/S0140-6736(14)61995-4
- Fitzpatrick S, Goss S, Di Bartolomeo A, Varma S, Tissera T, Earle E. Follow the money: is borderline personality disorder research underfunded in Canada? Can Psychol. 2024;65(1):46-57. doi:10.1037/cap0000375
- Zimmerman M, Gazarian D. Is research on borderline personality disorder underfunded by the National Institute of Health? Psychiatry Res. 2014;220(3):941-944. doi:10.1016/j.psychres.2014.09.021
- Leroux TC. U.S. military discharges and pre-existing personality disorders: a health policy review. Adm Policy Ment Health. 2015;42(6):748-755. doi:10.1007/s10488-014-0611-z
- Monahan MC, Keener JK. Fitness-for-duty evaluations. In Kennedy CH, Zillmer EA, eds. Military Psychology: Clinical and Operational Applications. 2nd ed. Guilford Publications; 2012:25-49.
- Hearing Before the Committee on Veterans’ Affairs, 111th Congress 2nd Sess (2010). Personality disorder discharges: impact on veterans benefits. Accessed March 4, 2025. https://www.govinfo.gov/content/pkg/CHRG-111hhrg61755/html/CHRG-111hhrg61755.htm
- Ader M, Cuthbert R, Hoechst K, Simon EH, Strassburger Z, Wishnie M. Casting troops aside: the United States military’s illegal personality disorder discharge problem. Vietnam Veterans of America. March 2012. Accessed February 28, 2025. https://law.yale.edu/sites/default/files/documents/pdf/Clinics/VLSC_CastingTroopsAside.pdf
- Edwards ER, Tran H, Wrobleski J, Rabhan Y, Yin J, Chiodi C, Goodman M, Geraci J. Prevalence of personality disorders across veteran samples: A meta-analysis. J Pers Disord. 2022;36(3):339-358. doi:10.1521/ pedi.2022.36.3.339
- Holliday R, Desai A, Edwards E, Borges L. Personal i ty disorder diagnosis among just ice -involved veterans: an investigation of VA-using veterans. J Nerv Ment Dis. 2023;211(5):402-406 doi:10.1097/ NMD.0000000000001627
- McCarthy JF, Bossarte RM, Katz IR, et al. Predictive modeling and concentration of the risk of suicide: implications for preventive interventions in the US Department of Veterans Affairs. Am J Public Health. 2015;105(9):1935-1942. doi:10.2105/AJPH.2015.302737
- Liu Y, Chen C, Zhou Y, Zhang N, Liu S. Twenty years of research on borderline personality disorder: a scientometric analysis of hotspots, bursts, and research trends. Front Psych. 2024;15:1361535. doi:10.3389/ fpsyt.2024.1361535
- Williams R, Holliday R, Clem M, Anderson E, Morris EE, Surís A. Borderline personality disorder and military sexual trauma: analysis of previous traumatization and current psychiatric presentation. J Interpers Violence. 2017;32(15):2223-2236. doi:10.1177/0886260515596149
- Holder N, Holliday R, Pai A, Surís A. Role of borderline personality disorder in the treatment of military sexual trauma-related posttraumatic stress disorder with cognitive processing therapy. Behav Med. 2017;43(3):184-190. doi:10.1080/08964289.2016.1276430
- Ralevski E, Ball S, Nich C, Limoncelli D, Petrakis I. The impact of personality disorders on alcohol-use outcomes in a pharmacotherapy trial for alcohol dependence and comorbid Axis I disorders. Am J Addict. 2007;16(6):443- 449. doi:10.1080/10550490701643336
- Walter KH, Bolte TA, Owens GP, Chard KM. The impact of personality disorders on treatment outcome for veterans in a posttraumatic stress disorder residential treatment program. Cognit Ther Res. 2012;36(5):576-584. doi:10.1007/s10608-011-9393-8
- Substance Abuse and Mental Health Services. Mental health client-level data (MH-CLD), 2022. Accessed February 28, 2025. https://www.datafiles.samhsa.gov/dataset/mental-health-client-level-data-2022-mh-cld-2022-ds0001
- Zimmerman M, Rothschild L, Chelminski I. The prevalence of DSM-IV personality disorders in psychiatric outpatients. Am J Psychiatry. 2005;162(10):1911-1918. doi:10.1176/appi.ajp.162.10.1911
- Campbell K, Clarke KA, Massey D, Lakeman R. Borderline personality disorder: To diagnose or not to diagnose? That is the question. Int J Mental Health Nurs. 2020;29(5):972-981. doi:10.1111/inm.12737
- Sisti D, Segal AG, Siegel AM, Johnson R, Gunderson J. Diagnosing, disclosing, and documenting borderline personality disorder: a survey of psychiatrists’ practices. J Pers Disord. 2016;30(6):848-856. doi:10.1521/ pedi_2015_29_228
- Klein P, Fairweather AK, Lawn S. Structural stigma and its impact on healthcare for borderline personality disorder: a scoping review. Int J Ment Health Syst. 2022;16(1):48. doi:10.1186/s13033-022-00558-3
- Knaak S, Szeto AC, Fitch K, Modgill G, Patten S. Stigma towards borderline personality disorder: effectiveness and generalizability of an anti-stigma program for healthcare providers using a pre-post randomized design. Borderline Personal Disord Emot Dysregul. 2015;2:9. doi:10.1186/s40479-015-0030-0
- Tate AE, Sahlin H, Liu S, et al. Borderline personality disorder: associations with psychiatric disorders, somatic illnesses, trauma, and adverse behaviors. Mol Psychiatry. 2022;27:2514-2521. doi:10.1038/s41380- 022-01503-z
- Abraham KM, Yosef M, Resnick SG, Zivin K. Competitive employment outcomes among veterans in VHA Therapeutic and Supported Employment Services programs. Psychiatr Serv. 2017;68(9)938-946. doi:10.1176/appi. ps201600412
- Frisman LK, Mueser KT, Covell NH, et al. Use of integrated dual disorder treatment via Assertive Community Treatment versus clinical case management for persons with co-occurring disorders and antisocial personality disorder. J Nerv Ment Dis. 2009;197(11):822-828. doi:10.1097/NMD.0b013e3181beac52
- Edwards ER, Kober H, Rinne GR, Griffin SA, Axelrod S, Cooney EB. Skills]homework completion and phone coaching as predictors of therapeutic change and outcomes in completers of a DBT intensive outpatient programme. Psychol Psychother. 2021;94(3):504-522. doi:10.1111/papt.12325
- Linehan MM, Dimeff LA, Reynolds SK, et al. Dialectical behavior therapy versus comprehensive validation therapy plus 12-step for the treatment of opioid dependent women meeting criteria for borderline personality disorder. Drug Alcohol Depend. 2002;67(1):13-26. doi:10.1016/s0376-8716(02)00011-x
- Linehan MM, Korslund KE, Harned MS, et al. Dialectical behavior therapy for high suicide risk in individuals with borderline personality disorder: a randomized clinical trial and component analysis. JAMA Psychiatry. 2015;72(5):475-482.doi:10.1001 /jamapsychiatry.2014.3039
- Carmel A, Rose ML, Fruzzetti AE. Barriers and solutions to implementing dialectical behavior therapy in a public behavioral health system. Adm Policy Ment Health. 2014;41(5):608-614. doi:10.1007/s10488-013-0504-6
- Decker SE, Matthieu MM, Smith BN, Landes SJ. Barriers and facilitators to dialectical behavior therapy skills groups in the Veterans Health Administration. Mil Med. 2024;189(5-6):1055-1063. doi:10.1093/milmed/ usad123
- Landes SJ, Rodriguez AL, Smith BN, et al. Barriers, facilitators, and benefits of implementation of dialectical behavior therapy in routine care: results from a national program evaluation survey in the Veterans Health Administration. Transl Behav Med. 2017;7(4):832-844. doi:10.1007/s13142-017-0465-5
- Walker J, Betthauser LM, Green K, Landes SJ, Stacy M. Suicide Prevention 2.0 Clinical Telehealth Program: Evidence- Based Treatment in the Veterans Health Administration. April 28, 2024. Accessed February 28, 2025. https://www.youtube.com/watch?v=fFsDzkg0SR0
- Gunderson J, Masland S, Choi-Kain L. Good psychiatric management: a review. Curr Opin Psychol. 2018;21:127- 131. doi:10.1016/j.copsyc.2017.12.006
- Kramer U. Good-enough therapy: a review of the empirical basis of good psychiatric management. Am J Psychother. 2025;78(1): 11-15. doi:10.1176/appi .psychotherapy.20230041
- Visdómine-Lozano JC. Contextualist perspectives in the treatment of antisocial behaviors and offending: a comparative review of FAP, ACT, DBT, and MDT. Trauma Violence Abuse. 2022;23(1):241-254. doi:10.1177/1524838020939509
- Drago A, Marogna C, Jørgen Søgaard H. A review of characteristics and treatments of the avoidant personality disorder. Could the DBT be an option? Int J Psychol Psychoanal. 2016;2(1):013.
- Finch EF, Choi-Kain LW, Iliakis EA, Eisen JL, Pinto A. Good psychiatric management for obsessive–compulsive personality disorder. Curr Behav Neurosci Rep. 2021;8:160-171. doi:10.1007/s40473-021-00239-4
- Miller TW, Kraus RF. Modified dialectical behavior therapy and problem solving for obsessive-compulsive personality disorder. Journal Contemp Psychother. 2007;37:79-85. doi:10.1007/s10879-006-9039-4
- Bozzatello P, Rocca P, De Rosa ML, Bellino S. Current and emerging medications for borderline personality disorder: is pharmacotherapy alone enough? Expert Opin Pharmacother. 2020;21(1):47-61.doi:10.1080/14656566 .2019.1686482
- Sand P, Derviososki E, Kollia S, Strand J, Di Leone F. Psychiatrists’ perspectives on prescription decisions for patients with personality disorders. J Pers Disord. 2024;38(3):225-240. doi:10.1521/pedi.2024.38.3.225
- Kane JM, Leucht S, Carpenter D, Docherty JP; Expert Consensus Panel for Optimizing Pharmacologic Treatment of Psychotic Disorders. The expert consensus guideline series. Optimizing pharmacologic treatment of psychotic disorders. Introduction: Methods, commentary, and summary. J Clin Psychiatry. 2003;64 Suppl 12:5-19.
- Nierenberg AA, Agustini B, Köhler-Forsberg O, et al. Diagnosis and treatment of bipolar disorder: a review. JAMA. 2023;330(14):1370-1380. doi:10.1001 /jama.2023.18588
- Köck P, Walter M. Personality disorder and substance use disorder–an update. Ment Health Prev. 2018;12:82- 89. doi:10.1016/J.MHP.2018.10.003
- Garb HN. Race bias and gender bias in the diagnosis of psychological disorders. Clin Psych Rev. 2021;90:102087. doi:10.1016/j.cpr.2021.102087
- Debast I, van Alphen SPJ, Rossi G, et al. Personality traits and personality disorders in late middle and old age: do they remain stable? A literature review. Clin Gerontol. 2014;37(3):253-271.doi:10.1080/07317115 .2014.885917
- Penders KAP, Peeters IGP, Metsemakers JFM, van Alphen SPJ. Personality disorders in older adults: a review of epidemiology, assessment, and treatment. Curr Psychiatry Rep. 2020;22(3):1-14. doi:10.1007/s11920-020- 1133-x
- Videler AC, Hutsebaut J, Schulkens JEM, Sobczak S, van Alphen SPJ. A life span perspective on borderline personality disorder. Curr Psychiatry Rep. 2019;21(7) :1-8. doi:10.1007/s11920-019-1040-1
- Wakefield JC. DSM-5 and the general definition of personality disorder. Clin Soc Work J. 2013;41(2):168-183. doi:10.1007/s10615-012-0402-5
- US Census Bureau. 2022 American Community Survey 1-year. Accessed February 28, 2025. https://data.census.gov/table/ACSST1Y2022.S2101?q=Veterans&y=2022comparison
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed, text revision. American Psychiatric Association; 2022.
- Hastrup LH, Jennum P, Ibsen R, Kjellberg J, Simonsen E. Societal costs of borderline personality disorders: a matched-controlled nationwide study of patients and spouses. Acta Psychiatr Scand. 2019;140(5):458-467. doi:10.1111/acps.13094
- Sveen CA, Pedersen G, Ulvestad DA, Zahl KE, Wilberg T, Kvarstein EH. Societal costs of personality disorders: a cross-sectional multicenter study of treatment-seeking patients in mental health services in Norway. J Clin Psychol. 2023;79(8):1752-1769. doi:10.1002/jclp.23504
- Beckwith H, Moran PF, Reilly J. Personality disorder prevalence in psychiatric outpatients: a systematic literature review. Personal Ment Health. 2014;8(2):91-101. doi:10.1002/pmh.1252
- Eaton NR, Greene AL. Personality disorders: community prevalence and socio-demographic correlates. Curr Opin Psychol. 2018;21:28-32. doi:10.1016/j.copsyc. 2017.09.001
- Edwards ER, Barnes S, Govindarajulu U, Geraci J, Tsai J. Mental health and substance use patterns associated with lifetime suicide attempt, incarceration, and homelessness: a latent class analysis of a nationally representative sample of U.S. veterans. Psychol Serv. 2021;18(4):619-631. doi:10.1037/ser0000488
- Moran P, Romaniuk H, Coffey C, et al. The influence of personality disorder on the future mental health and social adjustment of young adults: a population-based cohort study. Lancet Psychiatry. 2016;3(7):636-645. doi:10.1016/S2215-0366(16)30029-3
- Nelson SM, Griffin CA, Hein TC, Bowersox N, McCarthy JF. Personality disorder and suicide risk among patients in the Veterans Affairs health system. Personal Disord. 2022;13(6):563-571. doi:10.1037/per0000521
- Skodol AE. Impact of personality pathology on psychosocial functioning. Curr Opin Psychol. 2018;21;33-38. doi:10.1016/j.copsyc.2017.09.006
- Tyrer P, Reed GM, Crawford MJ. Classification, assessment, prevalence, and effect of personality disorder. Lancet. 2015;385(9969):717-726. doi:10.1016/S0140-6736(14)61995-4
- Fitzpatrick S, Goss S, Di Bartolomeo A, Varma S, Tissera T, Earle E. Follow the money: is borderline personality disorder research underfunded in Canada? Can Psychol. 2024;65(1):46-57. doi:10.1037/cap0000375
- Zimmerman M, Gazarian D. Is research on borderline personality disorder underfunded by the National Institute of Health? Psychiatry Res. 2014;220(3):941-944. doi:10.1016/j.psychres.2014.09.021
- Leroux TC. U.S. military discharges and pre-existing personality disorders: a health policy review. Adm Policy Ment Health. 2015;42(6):748-755. doi:10.1007/s10488-014-0611-z
- Monahan MC, Keener JK. Fitness-for-duty evaluations. In Kennedy CH, Zillmer EA, eds. Military Psychology: Clinical and Operational Applications. 2nd ed. Guilford Publications; 2012:25-49.
- Hearing Before the Committee on Veterans’ Affairs, 111th Congress 2nd Sess (2010). Personality disorder discharges: impact on veterans benefits. Accessed March 4, 2025. https://www.govinfo.gov/content/pkg/CHRG-111hhrg61755/html/CHRG-111hhrg61755.htm
- Ader M, Cuthbert R, Hoechst K, Simon EH, Strassburger Z, Wishnie M. Casting troops aside: the United States military’s illegal personality disorder discharge problem. Vietnam Veterans of America. March 2012. Accessed February 28, 2025. https://law.yale.edu/sites/default/files/documents/pdf/Clinics/VLSC_CastingTroopsAside.pdf
- Edwards ER, Tran H, Wrobleski J, Rabhan Y, Yin J, Chiodi C, Goodman M, Geraci J. Prevalence of personality disorders across veteran samples: A meta-analysis. J Pers Disord. 2022;36(3):339-358. doi:10.1521/ pedi.2022.36.3.339
- Holliday R, Desai A, Edwards E, Borges L. Personal i ty disorder diagnosis among just ice -involved veterans: an investigation of VA-using veterans. J Nerv Ment Dis. 2023;211(5):402-406 doi:10.1097/ NMD.0000000000001627
- McCarthy JF, Bossarte RM, Katz IR, et al. Predictive modeling and concentration of the risk of suicide: implications for preventive interventions in the US Department of Veterans Affairs. Am J Public Health. 2015;105(9):1935-1942. doi:10.2105/AJPH.2015.302737
- Liu Y, Chen C, Zhou Y, Zhang N, Liu S. Twenty years of research on borderline personality disorder: a scientometric analysis of hotspots, bursts, and research trends. Front Psych. 2024;15:1361535. doi:10.3389/ fpsyt.2024.1361535
- Williams R, Holliday R, Clem M, Anderson E, Morris EE, Surís A. Borderline personality disorder and military sexual trauma: analysis of previous traumatization and current psychiatric presentation. J Interpers Violence. 2017;32(15):2223-2236. doi:10.1177/0886260515596149
- Holder N, Holliday R, Pai A, Surís A. Role of borderline personality disorder in the treatment of military sexual trauma-related posttraumatic stress disorder with cognitive processing therapy. Behav Med. 2017;43(3):184-190. doi:10.1080/08964289.2016.1276430
- Ralevski E, Ball S, Nich C, Limoncelli D, Petrakis I. The impact of personality disorders on alcohol-use outcomes in a pharmacotherapy trial for alcohol dependence and comorbid Axis I disorders. Am J Addict. 2007;16(6):443- 449. doi:10.1080/10550490701643336
- Walter KH, Bolte TA, Owens GP, Chard KM. The impact of personality disorders on treatment outcome for veterans in a posttraumatic stress disorder residential treatment program. Cognit Ther Res. 2012;36(5):576-584. doi:10.1007/s10608-011-9393-8
- Substance Abuse and Mental Health Services. Mental health client-level data (MH-CLD), 2022. Accessed February 28, 2025. https://www.datafiles.samhsa.gov/dataset/mental-health-client-level-data-2022-mh-cld-2022-ds0001
- Zimmerman M, Rothschild L, Chelminski I. The prevalence of DSM-IV personality disorders in psychiatric outpatients. Am J Psychiatry. 2005;162(10):1911-1918. doi:10.1176/appi.ajp.162.10.1911
- Campbell K, Clarke KA, Massey D, Lakeman R. Borderline personality disorder: To diagnose or not to diagnose? That is the question. Int J Mental Health Nurs. 2020;29(5):972-981. doi:10.1111/inm.12737
- Sisti D, Segal AG, Siegel AM, Johnson R, Gunderson J. Diagnosing, disclosing, and documenting borderline personality disorder: a survey of psychiatrists’ practices. J Pers Disord. 2016;30(6):848-856. doi:10.1521/ pedi_2015_29_228
- Klein P, Fairweather AK, Lawn S. Structural stigma and its impact on healthcare for borderline personality disorder: a scoping review. Int J Ment Health Syst. 2022;16(1):48. doi:10.1186/s13033-022-00558-3
- Knaak S, Szeto AC, Fitch K, Modgill G, Patten S. Stigma towards borderline personality disorder: effectiveness and generalizability of an anti-stigma program for healthcare providers using a pre-post randomized design. Borderline Personal Disord Emot Dysregul. 2015;2:9. doi:10.1186/s40479-015-0030-0
- Tate AE, Sahlin H, Liu S, et al. Borderline personality disorder: associations with psychiatric disorders, somatic illnesses, trauma, and adverse behaviors. Mol Psychiatry. 2022;27:2514-2521. doi:10.1038/s41380- 022-01503-z
- Abraham KM, Yosef M, Resnick SG, Zivin K. Competitive employment outcomes among veterans in VHA Therapeutic and Supported Employment Services programs. Psychiatr Serv. 2017;68(9)938-946. doi:10.1176/appi. ps201600412
- Frisman LK, Mueser KT, Covell NH, et al. Use of integrated dual disorder treatment via Assertive Community Treatment versus clinical case management for persons with co-occurring disorders and antisocial personality disorder. J Nerv Ment Dis. 2009;197(11):822-828. doi:10.1097/NMD.0b013e3181beac52
- Edwards ER, Kober H, Rinne GR, Griffin SA, Axelrod S, Cooney EB. Skills]homework completion and phone coaching as predictors of therapeutic change and outcomes in completers of a DBT intensive outpatient programme. Psychol Psychother. 2021;94(3):504-522. doi:10.1111/papt.12325
- Linehan MM, Dimeff LA, Reynolds SK, et al. Dialectical behavior therapy versus comprehensive validation therapy plus 12-step for the treatment of opioid dependent women meeting criteria for borderline personality disorder. Drug Alcohol Depend. 2002;67(1):13-26. doi:10.1016/s0376-8716(02)00011-x
- Linehan MM, Korslund KE, Harned MS, et al. Dialectical behavior therapy for high suicide risk in individuals with borderline personality disorder: a randomized clinical trial and component analysis. JAMA Psychiatry. 2015;72(5):475-482.doi:10.1001 /jamapsychiatry.2014.3039
- Carmel A, Rose ML, Fruzzetti AE. Barriers and solutions to implementing dialectical behavior therapy in a public behavioral health system. Adm Policy Ment Health. 2014;41(5):608-614. doi:10.1007/s10488-013-0504-6
- Decker SE, Matthieu MM, Smith BN, Landes SJ. Barriers and facilitators to dialectical behavior therapy skills groups in the Veterans Health Administration. Mil Med. 2024;189(5-6):1055-1063. doi:10.1093/milmed/ usad123
- Landes SJ, Rodriguez AL, Smith BN, et al. Barriers, facilitators, and benefits of implementation of dialectical behavior therapy in routine care: results from a national program evaluation survey in the Veterans Health Administration. Transl Behav Med. 2017;7(4):832-844. doi:10.1007/s13142-017-0465-5
- Walker J, Betthauser LM, Green K, Landes SJ, Stacy M. Suicide Prevention 2.0 Clinical Telehealth Program: Evidence- Based Treatment in the Veterans Health Administration. April 28, 2024. Accessed February 28, 2025. https://www.youtube.com/watch?v=fFsDzkg0SR0
- Gunderson J, Masland S, Choi-Kain L. Good psychiatric management: a review. Curr Opin Psychol. 2018;21:127- 131. doi:10.1016/j.copsyc.2017.12.006
- Kramer U. Good-enough therapy: a review of the empirical basis of good psychiatric management. Am J Psychother. 2025;78(1): 11-15. doi:10.1176/appi .psychotherapy.20230041
- Visdómine-Lozano JC. Contextualist perspectives in the treatment of antisocial behaviors and offending: a comparative review of FAP, ACT, DBT, and MDT. Trauma Violence Abuse. 2022;23(1):241-254. doi:10.1177/1524838020939509
- Drago A, Marogna C, Jørgen Søgaard H. A review of characteristics and treatments of the avoidant personality disorder. Could the DBT be an option? Int J Psychol Psychoanal. 2016;2(1):013.
- Finch EF, Choi-Kain LW, Iliakis EA, Eisen JL, Pinto A. Good psychiatric management for obsessive–compulsive personality disorder. Curr Behav Neurosci Rep. 2021;8:160-171. doi:10.1007/s40473-021-00239-4
- Miller TW, Kraus RF. Modified dialectical behavior therapy and problem solving for obsessive-compulsive personality disorder. Journal Contemp Psychother. 2007;37:79-85. doi:10.1007/s10879-006-9039-4
- Bozzatello P, Rocca P, De Rosa ML, Bellino S. Current and emerging medications for borderline personality disorder: is pharmacotherapy alone enough? Expert Opin Pharmacother. 2020;21(1):47-61.doi:10.1080/14656566 .2019.1686482
- Sand P, Derviososki E, Kollia S, Strand J, Di Leone F. Psychiatrists’ perspectives on prescription decisions for patients with personality disorders. J Pers Disord. 2024;38(3):225-240. doi:10.1521/pedi.2024.38.3.225
- Kane JM, Leucht S, Carpenter D, Docherty JP; Expert Consensus Panel for Optimizing Pharmacologic Treatment of Psychotic Disorders. The expert consensus guideline series. Optimizing pharmacologic treatment of psychotic disorders. Introduction: Methods, commentary, and summary. J Clin Psychiatry. 2003;64 Suppl 12:5-19.
- Nierenberg AA, Agustini B, Köhler-Forsberg O, et al. Diagnosis and treatment of bipolar disorder: a review. JAMA. 2023;330(14):1370-1380. doi:10.1001 /jama.2023.18588
- Köck P, Walter M. Personality disorder and substance use disorder–an update. Ment Health Prev. 2018;12:82- 89. doi:10.1016/J.MHP.2018.10.003
- Garb HN. Race bias and gender bias in the diagnosis of psychological disorders. Clin Psych Rev. 2021;90:102087. doi:10.1016/j.cpr.2021.102087
- Debast I, van Alphen SPJ, Rossi G, et al. Personality traits and personality disorders in late middle and old age: do they remain stable? A literature review. Clin Gerontol. 2014;37(3):253-271.doi:10.1080/07317115 .2014.885917
- Penders KAP, Peeters IGP, Metsemakers JFM, van Alphen SPJ. Personality disorders in older adults: a review of epidemiology, assessment, and treatment. Curr Psychiatry Rep. 2020;22(3):1-14. doi:10.1007/s11920-020- 1133-x
- Videler AC, Hutsebaut J, Schulkens JEM, Sobczak S, van Alphen SPJ. A life span perspective on borderline personality disorder. Curr Psychiatry Rep. 2019;21(7) :1-8. doi:10.1007/s11920-019-1040-1
- Wakefield JC. DSM-5 and the general definition of personality disorder. Clin Soc Work J. 2013;41(2):168-183. doi:10.1007/s10615-012-0402-5
- US Census Bureau. 2022 American Community Survey 1-year. Accessed February 28, 2025. https://data.census.gov/table/ACSST1Y2022.S2101?q=Veterans&y=2022comparison
Needs of Veterans With Personality Disorder Diagnoses in Community-Based Mental Health Care
Needs of Veterans With Personality Disorder Diagnoses in Community-Based Mental Health Care
Gut Microbiome Influences Multiple Neurodegenerative Disorders
WASHINGTON, DC — Age-related neurodegenerative disorders — motor neuron diseases, demyelinating diseases, Alzheimer’s disease, and other proteinopathies — are at an “inflection point,” said researcher Andrea R. Merchak, PhD, with a fuller understanding of disease pathophysiology but an overall dearth of effective disease-modifying treatments.
And this, Merchak said at the Gut Microbiota for Health (GMFH) World Summit 2025, is where the gut microbiome comes in. “The gut-brain axis is important to take into consideration,” she urged, both for gut microbiome researchers — whose collaboration with neurologists and neuroscientists is essential — and for practicing gastroenterologists.
“We are the sum of our environmental exposures,” said Merchak, assistant research professor of neurology at the Indiana University School of Medicine, in Indianapolis. “So for your patient populations, remember you’re not only treating the diseases they’re coming to you with, you’re also treating them for a lifetime of healthy [brain] aging.”
At the center of a healthy aging brain are the brain-residing microglia and peripheral monocytes, she said. These immune cell populations are directly influenced by blood-brain barrier breakdown, inflammation, and gut permeability — and indirectly influenced by microbial products, gastrointestinal (GI) function, and bacterial diversity, Merchak said at the meeting, which was convened by AGA and the European Society of Neurogastroenterology and Motility.
“Many of us grew up learning that the brain is an immune-privileged site, but we’ve been establishing that this is fundamentally not true,” she said. “While the brain does have a privileged status, there are interactions with the blood, with the peripheral immune cells.”
Merchak coauthored a 2024 review in Neurotherapeutics in which she and her colleagues explained that the brain is “heavily connected with peripheral immune dynamics,” and that the gut — as the largest immune organ in the body — is a critical place for peripheral immune development, “thus influencing brain health.”
Gut microbiota interact with the brain via several mechanisms including microbiota-derived metabolites that enter circulation, direct communication via the vagus nerve, and modulation of the immune system, Merchak and her coauthors wrote. Leaky gut, they noted, can lead to an accumulation of inflammatory signals and cells that can exacerbate or induce the onset of neurodegenerative conditions.
As researchers better understand the role that GI dysfunction plays in neurodegenerative disease — as they identify microbiome signatures for predicting risk, for instance — there will be “opportunities to target the microbiome to prevent or reverse dysbiosis as a way to delay, arrest or prevent the onset and progression of neurodegenerative diseases,” they wrote.
At the GMFH meeting, Merchak described both ongoing preclinical research that is dissecting gut-brain communication, and preliminary clinical evidence for the use of gut microbiota-modulating therapies in neurodegenerative disease.
Support for a Gut-Focused Approach
Research on bile acid metabolism in multiple sclerosis (MS) and on peripheral inflammation in dementia exemplify the ongoing preclinical research uncovering the mechanisms of gut-brain communication, Merchak said.
The finding that bile acid metabolism modulates MS autoimmunity comes from research done by Merchak and a team at the University of Virginia, Charlottesville, Virginia, several years ago in which mice with experimental autoimmune encephalomyelitis (EAE) — an animal model of MS — were engineered for T cell specific knockout of the aryl hydrocarbon receptor (AHR). The AHR has been directly tied to MS, and T lymphocytes are known to play a central role in MS pathophysiology.
Blocking the activity of AHR in CD4-positive T cells significantly affected the production of bile acids and other metabolites in the microbiome — and the outcome of central nervous system autoimmunity. “Mice with high levels of bile acids, both primary and secondary, actually recovered from this EAE” and regained motor function, Merchak said at the GMFH meeting.
The potential impact of genetic manipulation on recovery was ruled out — and the role of bile acids confirmed — when, using the EAE model, gut bacteria from mice without AHR were transplanted into mice with AHR. The mice with AHR were able to recover, confirming that AHR can reprogram the gut microbiome and that “high levels of bile acid can lead to reduced autoimmunity in an MS model,” she said.
Other elements and stages of the research, which was published in PLOS Biology in 2023, showed increased apoptosis of CD4-positive immune cells in AHR-deficient mice and the ability of oral taurocholic acid — a bile acid that was especially high in mice without AHR — to reduce the severity of EAE, Merchak said.
Evidence for the role of gut and peripheral inflammation on neurodegeneration is building on numerous fronts, Merchak said. Unpublished research using spatial transcriptomics of colon biopsies from patients with inflammatory bowel disease (IBD), patients with Parkinson’s disease (PD), and neurologically healthy control individuals, for instance, showed similar cell communication patterns in patients with IBD or PD (and no history of IBD) compared with healthy control individuals.
And in research using a single-cell genomics approach and a mouse model of lipopolysaccharide (LPS)-induced system neuroinflammation, microglia were found to preferentially communicate with peripheral myeloid cells rather than other microglia after peripheral LPS exposure.
“In saline-treated mice, the microglia are talking primarily to microglia, but in LPS-treated mice, microglia spend more time communicating with monocytes and T cells,” Merchak explained. “We see communication going from inside the brain to cells coming in from the periphery.”
In another experiment, 2 months of a high-fat, high-sugar diet in mice with an engineered predisposition to frontotemporal dementia led to significant upregulation of major histocompatibility complex class II (MHC II) expression on monocytes in the brain, she said, describing unpublished research. Because MHC II handles antigen presentation in the brain, the change signals increased central-peripheral immune crosstalk and increased brain inflammation.
State of Clinical Research
On the clinical side, Merchak said studies of gut microbiome-modulating therapies are currently not longitudinal enough to accurately study neurodegenerative diseases that may develop over decades. Still, her review of the literature — part of her 2024 article — suggests there is at least some preliminary clinical evidence for the use of probiotics/prebiotics/diet and fecal microbiota transplant (FMT) in several diseases.
- Parkinson’s Disease: “There has been some evidence,” Merchak said at the meeting, “for the treatment [with probiotics, prebiotics and diet] of nonmotor symptoms — things like gastrointestinal distress and mood changes — but no real evidence that such treatments can help with the motor symptoms we see in Parkinson’s.” Over 60 patients with PD have been treated with FMT, she said, with reduced GI distress and mixed results with motor symptoms.
- Alzheimer’s and related dementias: “Diet shows promise for cognitive outcomes, but there hasn’t been much evidence for probiotics,” she said. Her review found 17 patients diagnosed with dementia who were treated with FMT, “and for many of them, maintenance of cognitive function was reported — so no further decline — which is excellent.”
- Multiple Sclerosis: “We see higher quality-of-life measures in patients getting probiotics, prebiotics, and changes in diet,” Merchak said. “Again, most of this [relates to] mood and digestion, but some studies show a slowing of neurological damage as measured by MRI.”
There are reports of 15 patients treated with FMT, and “three of these document full functional recovery,” she said, noting that longer follow-up is necessary as MS is characterized by relapsed and periods of recovery.
Merchak reported no financial disclosures.
A version of this article appeared on Medscape.com.
WASHINGTON, DC — Age-related neurodegenerative disorders — motor neuron diseases, demyelinating diseases, Alzheimer’s disease, and other proteinopathies — are at an “inflection point,” said researcher Andrea R. Merchak, PhD, with a fuller understanding of disease pathophysiology but an overall dearth of effective disease-modifying treatments.
And this, Merchak said at the Gut Microbiota for Health (GMFH) World Summit 2025, is where the gut microbiome comes in. “The gut-brain axis is important to take into consideration,” she urged, both for gut microbiome researchers — whose collaboration with neurologists and neuroscientists is essential — and for practicing gastroenterologists.
“We are the sum of our environmental exposures,” said Merchak, assistant research professor of neurology at the Indiana University School of Medicine, in Indianapolis. “So for your patient populations, remember you’re not only treating the diseases they’re coming to you with, you’re also treating them for a lifetime of healthy [brain] aging.”
At the center of a healthy aging brain are the brain-residing microglia and peripheral monocytes, she said. These immune cell populations are directly influenced by blood-brain barrier breakdown, inflammation, and gut permeability — and indirectly influenced by microbial products, gastrointestinal (GI) function, and bacterial diversity, Merchak said at the meeting, which was convened by AGA and the European Society of Neurogastroenterology and Motility.
“Many of us grew up learning that the brain is an immune-privileged site, but we’ve been establishing that this is fundamentally not true,” she said. “While the brain does have a privileged status, there are interactions with the blood, with the peripheral immune cells.”
Merchak coauthored a 2024 review in Neurotherapeutics in which she and her colleagues explained that the brain is “heavily connected with peripheral immune dynamics,” and that the gut — as the largest immune organ in the body — is a critical place for peripheral immune development, “thus influencing brain health.”
Gut microbiota interact with the brain via several mechanisms including microbiota-derived metabolites that enter circulation, direct communication via the vagus nerve, and modulation of the immune system, Merchak and her coauthors wrote. Leaky gut, they noted, can lead to an accumulation of inflammatory signals and cells that can exacerbate or induce the onset of neurodegenerative conditions.
As researchers better understand the role that GI dysfunction plays in neurodegenerative disease — as they identify microbiome signatures for predicting risk, for instance — there will be “opportunities to target the microbiome to prevent or reverse dysbiosis as a way to delay, arrest or prevent the onset and progression of neurodegenerative diseases,” they wrote.
At the GMFH meeting, Merchak described both ongoing preclinical research that is dissecting gut-brain communication, and preliminary clinical evidence for the use of gut microbiota-modulating therapies in neurodegenerative disease.
Support for a Gut-Focused Approach
Research on bile acid metabolism in multiple sclerosis (MS) and on peripheral inflammation in dementia exemplify the ongoing preclinical research uncovering the mechanisms of gut-brain communication, Merchak said.
The finding that bile acid metabolism modulates MS autoimmunity comes from research done by Merchak and a team at the University of Virginia, Charlottesville, Virginia, several years ago in which mice with experimental autoimmune encephalomyelitis (EAE) — an animal model of MS — were engineered for T cell specific knockout of the aryl hydrocarbon receptor (AHR). The AHR has been directly tied to MS, and T lymphocytes are known to play a central role in MS pathophysiology.
Blocking the activity of AHR in CD4-positive T cells significantly affected the production of bile acids and other metabolites in the microbiome — and the outcome of central nervous system autoimmunity. “Mice with high levels of bile acids, both primary and secondary, actually recovered from this EAE” and regained motor function, Merchak said at the GMFH meeting.
The potential impact of genetic manipulation on recovery was ruled out — and the role of bile acids confirmed — when, using the EAE model, gut bacteria from mice without AHR were transplanted into mice with AHR. The mice with AHR were able to recover, confirming that AHR can reprogram the gut microbiome and that “high levels of bile acid can lead to reduced autoimmunity in an MS model,” she said.
Other elements and stages of the research, which was published in PLOS Biology in 2023, showed increased apoptosis of CD4-positive immune cells in AHR-deficient mice and the ability of oral taurocholic acid — a bile acid that was especially high in mice without AHR — to reduce the severity of EAE, Merchak said.
Evidence for the role of gut and peripheral inflammation on neurodegeneration is building on numerous fronts, Merchak said. Unpublished research using spatial transcriptomics of colon biopsies from patients with inflammatory bowel disease (IBD), patients with Parkinson’s disease (PD), and neurologically healthy control individuals, for instance, showed similar cell communication patterns in patients with IBD or PD (and no history of IBD) compared with healthy control individuals.
And in research using a single-cell genomics approach and a mouse model of lipopolysaccharide (LPS)-induced system neuroinflammation, microglia were found to preferentially communicate with peripheral myeloid cells rather than other microglia after peripheral LPS exposure.
“In saline-treated mice, the microglia are talking primarily to microglia, but in LPS-treated mice, microglia spend more time communicating with monocytes and T cells,” Merchak explained. “We see communication going from inside the brain to cells coming in from the periphery.”
In another experiment, 2 months of a high-fat, high-sugar diet in mice with an engineered predisposition to frontotemporal dementia led to significant upregulation of major histocompatibility complex class II (MHC II) expression on monocytes in the brain, she said, describing unpublished research. Because MHC II handles antigen presentation in the brain, the change signals increased central-peripheral immune crosstalk and increased brain inflammation.
State of Clinical Research
On the clinical side, Merchak said studies of gut microbiome-modulating therapies are currently not longitudinal enough to accurately study neurodegenerative diseases that may develop over decades. Still, her review of the literature — part of her 2024 article — suggests there is at least some preliminary clinical evidence for the use of probiotics/prebiotics/diet and fecal microbiota transplant (FMT) in several diseases.
- Parkinson’s Disease: “There has been some evidence,” Merchak said at the meeting, “for the treatment [with probiotics, prebiotics and diet] of nonmotor symptoms — things like gastrointestinal distress and mood changes — but no real evidence that such treatments can help with the motor symptoms we see in Parkinson’s.” Over 60 patients with PD have been treated with FMT, she said, with reduced GI distress and mixed results with motor symptoms.
- Alzheimer’s and related dementias: “Diet shows promise for cognitive outcomes, but there hasn’t been much evidence for probiotics,” she said. Her review found 17 patients diagnosed with dementia who were treated with FMT, “and for many of them, maintenance of cognitive function was reported — so no further decline — which is excellent.”
- Multiple Sclerosis: “We see higher quality-of-life measures in patients getting probiotics, prebiotics, and changes in diet,” Merchak said. “Again, most of this [relates to] mood and digestion, but some studies show a slowing of neurological damage as measured by MRI.”
There are reports of 15 patients treated with FMT, and “three of these document full functional recovery,” she said, noting that longer follow-up is necessary as MS is characterized by relapsed and periods of recovery.
Merchak reported no financial disclosures.
A version of this article appeared on Medscape.com.
WASHINGTON, DC — Age-related neurodegenerative disorders — motor neuron diseases, demyelinating diseases, Alzheimer’s disease, and other proteinopathies — are at an “inflection point,” said researcher Andrea R. Merchak, PhD, with a fuller understanding of disease pathophysiology but an overall dearth of effective disease-modifying treatments.
And this, Merchak said at the Gut Microbiota for Health (GMFH) World Summit 2025, is where the gut microbiome comes in. “The gut-brain axis is important to take into consideration,” she urged, both for gut microbiome researchers — whose collaboration with neurologists and neuroscientists is essential — and for practicing gastroenterologists.
“We are the sum of our environmental exposures,” said Merchak, assistant research professor of neurology at the Indiana University School of Medicine, in Indianapolis. “So for your patient populations, remember you’re not only treating the diseases they’re coming to you with, you’re also treating them for a lifetime of healthy [brain] aging.”
At the center of a healthy aging brain are the brain-residing microglia and peripheral monocytes, she said. These immune cell populations are directly influenced by blood-brain barrier breakdown, inflammation, and gut permeability — and indirectly influenced by microbial products, gastrointestinal (GI) function, and bacterial diversity, Merchak said at the meeting, which was convened by AGA and the European Society of Neurogastroenterology and Motility.
“Many of us grew up learning that the brain is an immune-privileged site, but we’ve been establishing that this is fundamentally not true,” she said. “While the brain does have a privileged status, there are interactions with the blood, with the peripheral immune cells.”
Merchak coauthored a 2024 review in Neurotherapeutics in which she and her colleagues explained that the brain is “heavily connected with peripheral immune dynamics,” and that the gut — as the largest immune organ in the body — is a critical place for peripheral immune development, “thus influencing brain health.”
Gut microbiota interact with the brain via several mechanisms including microbiota-derived metabolites that enter circulation, direct communication via the vagus nerve, and modulation of the immune system, Merchak and her coauthors wrote. Leaky gut, they noted, can lead to an accumulation of inflammatory signals and cells that can exacerbate or induce the onset of neurodegenerative conditions.
As researchers better understand the role that GI dysfunction plays in neurodegenerative disease — as they identify microbiome signatures for predicting risk, for instance — there will be “opportunities to target the microbiome to prevent or reverse dysbiosis as a way to delay, arrest or prevent the onset and progression of neurodegenerative diseases,” they wrote.
At the GMFH meeting, Merchak described both ongoing preclinical research that is dissecting gut-brain communication, and preliminary clinical evidence for the use of gut microbiota-modulating therapies in neurodegenerative disease.
Support for a Gut-Focused Approach
Research on bile acid metabolism in multiple sclerosis (MS) and on peripheral inflammation in dementia exemplify the ongoing preclinical research uncovering the mechanisms of gut-brain communication, Merchak said.
The finding that bile acid metabolism modulates MS autoimmunity comes from research done by Merchak and a team at the University of Virginia, Charlottesville, Virginia, several years ago in which mice with experimental autoimmune encephalomyelitis (EAE) — an animal model of MS — were engineered for T cell specific knockout of the aryl hydrocarbon receptor (AHR). The AHR has been directly tied to MS, and T lymphocytes are known to play a central role in MS pathophysiology.
Blocking the activity of AHR in CD4-positive T cells significantly affected the production of bile acids and other metabolites in the microbiome — and the outcome of central nervous system autoimmunity. “Mice with high levels of bile acids, both primary and secondary, actually recovered from this EAE” and regained motor function, Merchak said at the GMFH meeting.
The potential impact of genetic manipulation on recovery was ruled out — and the role of bile acids confirmed — when, using the EAE model, gut bacteria from mice without AHR were transplanted into mice with AHR. The mice with AHR were able to recover, confirming that AHR can reprogram the gut microbiome and that “high levels of bile acid can lead to reduced autoimmunity in an MS model,” she said.
Other elements and stages of the research, which was published in PLOS Biology in 2023, showed increased apoptosis of CD4-positive immune cells in AHR-deficient mice and the ability of oral taurocholic acid — a bile acid that was especially high in mice without AHR — to reduce the severity of EAE, Merchak said.
Evidence for the role of gut and peripheral inflammation on neurodegeneration is building on numerous fronts, Merchak said. Unpublished research using spatial transcriptomics of colon biopsies from patients with inflammatory bowel disease (IBD), patients with Parkinson’s disease (PD), and neurologically healthy control individuals, for instance, showed similar cell communication patterns in patients with IBD or PD (and no history of IBD) compared with healthy control individuals.
And in research using a single-cell genomics approach and a mouse model of lipopolysaccharide (LPS)-induced system neuroinflammation, microglia were found to preferentially communicate with peripheral myeloid cells rather than other microglia after peripheral LPS exposure.
“In saline-treated mice, the microglia are talking primarily to microglia, but in LPS-treated mice, microglia spend more time communicating with monocytes and T cells,” Merchak explained. “We see communication going from inside the brain to cells coming in from the periphery.”
In another experiment, 2 months of a high-fat, high-sugar diet in mice with an engineered predisposition to frontotemporal dementia led to significant upregulation of major histocompatibility complex class II (MHC II) expression on monocytes in the brain, she said, describing unpublished research. Because MHC II handles antigen presentation in the brain, the change signals increased central-peripheral immune crosstalk and increased brain inflammation.
State of Clinical Research
On the clinical side, Merchak said studies of gut microbiome-modulating therapies are currently not longitudinal enough to accurately study neurodegenerative diseases that may develop over decades. Still, her review of the literature — part of her 2024 article — suggests there is at least some preliminary clinical evidence for the use of probiotics/prebiotics/diet and fecal microbiota transplant (FMT) in several diseases.
- Parkinson’s Disease: “There has been some evidence,” Merchak said at the meeting, “for the treatment [with probiotics, prebiotics and diet] of nonmotor symptoms — things like gastrointestinal distress and mood changes — but no real evidence that such treatments can help with the motor symptoms we see in Parkinson’s.” Over 60 patients with PD have been treated with FMT, she said, with reduced GI distress and mixed results with motor symptoms.
- Alzheimer’s and related dementias: “Diet shows promise for cognitive outcomes, but there hasn’t been much evidence for probiotics,” she said. Her review found 17 patients diagnosed with dementia who were treated with FMT, “and for many of them, maintenance of cognitive function was reported — so no further decline — which is excellent.”
- Multiple Sclerosis: “We see higher quality-of-life measures in patients getting probiotics, prebiotics, and changes in diet,” Merchak said. “Again, most of this [relates to] mood and digestion, but some studies show a slowing of neurological damage as measured by MRI.”
There are reports of 15 patients treated with FMT, and “three of these document full functional recovery,” she said, noting that longer follow-up is necessary as MS is characterized by relapsed and periods of recovery.
Merchak reported no financial disclosures.
A version of this article appeared on Medscape.com.
FROM GMFH 2025
Five Reasons to Update Your Will
You have a will, so you can rest easy, right? Not necessarily. If your will is outdated, it can cause more harm than good.
Even though it can provide for some contingencies, an old will can’t cover every change that may have occurred since it was first drawn. Professionals advise that you review your will every few years and more often if situations such as the following five have occurred since you last updated your will.
1. Family Changes
If you’ve had any changes in your family situation, you will probably need to update your will. Events such as marriage, divorce, death, birth, adoption, or a falling out with a loved one may affect how your estate will be distributed, who should act as guardian for your dependents, and who should be named as executor of your estate.
2. Relocating to a New State
The laws among the states vary. Moving to a new state or purchasing property in another state can affect your estate plan and how property in that state will be taxed and distributed.
3. Tax Law Changes
Federal and state legislatures are continually tinkering with federal estate and state inheritance tax laws. An old will may fail to take advantage of strategies that will minimize estate taxes.
4. You Want to Support a Favorite Cause
If you have developed a connection to a cause, you may want to benefit a particular charity with a gift in your estate. Contact us for sample language you can share with your attorney to include a gift to us in your will.
5. Changes in Your Estate’s Value
When you made your will, your assets may have been relatively modest. Now the value may be larger and your will no longer reflects how you would like your estate divided.
You will help spark future discoveries in GI. Visit our website at https://gastro.planmylegacy.org or contact us at [email protected].
You have a will, so you can rest easy, right? Not necessarily. If your will is outdated, it can cause more harm than good.
Even though it can provide for some contingencies, an old will can’t cover every change that may have occurred since it was first drawn. Professionals advise that you review your will every few years and more often if situations such as the following five have occurred since you last updated your will.
1. Family Changes
If you’ve had any changes in your family situation, you will probably need to update your will. Events such as marriage, divorce, death, birth, adoption, or a falling out with a loved one may affect how your estate will be distributed, who should act as guardian for your dependents, and who should be named as executor of your estate.
2. Relocating to a New State
The laws among the states vary. Moving to a new state or purchasing property in another state can affect your estate plan and how property in that state will be taxed and distributed.
3. Tax Law Changes
Federal and state legislatures are continually tinkering with federal estate and state inheritance tax laws. An old will may fail to take advantage of strategies that will minimize estate taxes.
4. You Want to Support a Favorite Cause
If you have developed a connection to a cause, you may want to benefit a particular charity with a gift in your estate. Contact us for sample language you can share with your attorney to include a gift to us in your will.
5. Changes in Your Estate’s Value
When you made your will, your assets may have been relatively modest. Now the value may be larger and your will no longer reflects how you would like your estate divided.
You will help spark future discoveries in GI. Visit our website at https://gastro.planmylegacy.org or contact us at [email protected].
You have a will, so you can rest easy, right? Not necessarily. If your will is outdated, it can cause more harm than good.
Even though it can provide for some contingencies, an old will can’t cover every change that may have occurred since it was first drawn. Professionals advise that you review your will every few years and more often if situations such as the following five have occurred since you last updated your will.
1. Family Changes
If you’ve had any changes in your family situation, you will probably need to update your will. Events such as marriage, divorce, death, birth, adoption, or a falling out with a loved one may affect how your estate will be distributed, who should act as guardian for your dependents, and who should be named as executor of your estate.
2. Relocating to a New State
The laws among the states vary. Moving to a new state or purchasing property in another state can affect your estate plan and how property in that state will be taxed and distributed.
3. Tax Law Changes
Federal and state legislatures are continually tinkering with federal estate and state inheritance tax laws. An old will may fail to take advantage of strategies that will minimize estate taxes.
4. You Want to Support a Favorite Cause
If you have developed a connection to a cause, you may want to benefit a particular charity with a gift in your estate. Contact us for sample language you can share with your attorney to include a gift to us in your will.
5. Changes in Your Estate’s Value
When you made your will, your assets may have been relatively modest. Now the value may be larger and your will no longer reflects how you would like your estate divided.
You will help spark future discoveries in GI. Visit our website at https://gastro.planmylegacy.org or contact us at [email protected].
Simple Score Predicts Advanced Colorectal Neoplasia in Young Adults
While colorectal cancer (CRC) incidence has declined overall due to screening, early-onset CRC is on the rise, particularly in individuals younger than 45 years — an age group not currently recommended for CRC screening.
Studies have shown that the risk for early-onset advanced neoplasia varies based on several factors, including sex, race, family history of CRC, smoking, alcohol consumption, diabetes, hyperlipidemia, obesity, and diet.
A score that incorporates some of these factors to identify which younger adults are at higher risk for advanced neoplasia, a precursor to CRC, could support earlier, more targeted screening interventions.
The simple clinical score can be easily calculated by primary care providers in the office, Carole Macaron, MD, lead author of the study and a gastroenterologist at Cleveland Clinic, told GI & Hepatology News. “Patients with a high-risk score would be referred for colorectal cancer screening.”
The study was published in Digestive Diseases and Sciences.
To develop and validate their risk score, Macaron and colleagues did a retrospective cross-sectional analysis of 9446 individuals aged 18-44 years (mean age, 36.8 years; 61% women) who underwent colonoscopy at their center.
Advanced neoplasia was defined as a tubular adenoma ≥ 10 mm or any adenoma with villous features or high-grade dysplasia, sessile serrated polyp ≥ 10 mm, sessile serrated polyp with dysplasia, traditional serrated adenoma, or invasive adenocarcinoma.
The 346 (3.7%) individuals found to have advanced neoplasia served as the case group, and the remainder with normal colonoscopy or non-advanced neoplasia served as controls.
A multivariate logistic regression model identified three independent risk factors significantly associated with advanced neoplasia: Higher body mass index (BMI; P = .0157), former and current tobacco use (P = .0009 and P = .0015, respectively), and having a first-degree relative with CRC < 60 years (P < .0001) or other family history of CRC (P = .0117).
The researchers used these risk factors to develop a risk prediction score to estimate the likelihood of detecting advanced neoplasia, which ranged from a risk of 1.8% for patients with a score of 1 to 22.2% for those with a score of 12. Individuals with a score of ≥ 9 had a 14% or higher risk for advanced neoplasia.
Based on the risk model, the likelihood of detecting advanced neoplasia in an asymptomatic 32-year-old overweight individual, with a history of previous tobacco use and a first-degree relative younger than age 60 with CRC would be 20.3%, Macaron and colleagues noted.
The model demonstrated “moderate” discriminatory power in the validation set (C-statistic: 0.645), indicating that it can effectively differentiate between individuals at a higher and lower risk for advanced neoplasia.
Additionally, the authors are exploring ways to improve the discriminatory power of the score, possibly by including additional risk factors.
Given the score is calculated using easily obtainable risk factors for individuals younger than 45 who are at risk for early-onset colorectal neoplasia, it could help guide individualized screening decisions for those in whom screening is not currently offered, Macaron said. It could also serve as a tool for risk communication and shared decision-making.
Integration into electronic health records or online calculators may enhance its accessibility and clinical utility.
The authors noted that this retrospective study was conducted at a single center caring mainly for White non-Hispanic adults, limiting generalizability to the general population and to other races and ethnicities.
Validation in Real-World Setting Needed
“There are no currently accepted advanced colorectal neoplasia risk scores that are used in general practice,” said Steven H. Itzkowitz, MD, AGAF, professor of medicine, oncological sciences, and medical education, Icahn School of Medicine at Mount Sinai in New York City. “If these lesions can be predicted, it would enable these young individuals to undergo screening colonoscopy, which could detect and remove these lesions, thereby preventing CRC.”
Many of the known risk factors (family history, high BMI, or smoking) for CRC development at any age are incorporated within this tool, so it should be feasible to collect these data,” said Itzkowitz, who was not involved with the study.
But he cautioned that accurate and adequate family histories are not always performed. Clinicians also may not have considered combining these factors into an actionable risk score.
“If this score can be externally validated in a real-world setting, it could be a useful addition in our efforts to lower CRC rates among young individuals,” Itzkowitz said.
The study did not receive any funding. Macaron and Itzkowitz reported no competing interests.
A version of this article first appeared on Medscape.com.
While colorectal cancer (CRC) incidence has declined overall due to screening, early-onset CRC is on the rise, particularly in individuals younger than 45 years — an age group not currently recommended for CRC screening.
Studies have shown that the risk for early-onset advanced neoplasia varies based on several factors, including sex, race, family history of CRC, smoking, alcohol consumption, diabetes, hyperlipidemia, obesity, and diet.
A score that incorporates some of these factors to identify which younger adults are at higher risk for advanced neoplasia, a precursor to CRC, could support earlier, more targeted screening interventions.
The simple clinical score can be easily calculated by primary care providers in the office, Carole Macaron, MD, lead author of the study and a gastroenterologist at Cleveland Clinic, told GI & Hepatology News. “Patients with a high-risk score would be referred for colorectal cancer screening.”
The study was published in Digestive Diseases and Sciences.
To develop and validate their risk score, Macaron and colleagues did a retrospective cross-sectional analysis of 9446 individuals aged 18-44 years (mean age, 36.8 years; 61% women) who underwent colonoscopy at their center.
Advanced neoplasia was defined as a tubular adenoma ≥ 10 mm or any adenoma with villous features or high-grade dysplasia, sessile serrated polyp ≥ 10 mm, sessile serrated polyp with dysplasia, traditional serrated adenoma, or invasive adenocarcinoma.
The 346 (3.7%) individuals found to have advanced neoplasia served as the case group, and the remainder with normal colonoscopy or non-advanced neoplasia served as controls.
A multivariate logistic regression model identified three independent risk factors significantly associated with advanced neoplasia: Higher body mass index (BMI; P = .0157), former and current tobacco use (P = .0009 and P = .0015, respectively), and having a first-degree relative with CRC < 60 years (P < .0001) or other family history of CRC (P = .0117).
The researchers used these risk factors to develop a risk prediction score to estimate the likelihood of detecting advanced neoplasia, which ranged from a risk of 1.8% for patients with a score of 1 to 22.2% for those with a score of 12. Individuals with a score of ≥ 9 had a 14% or higher risk for advanced neoplasia.
Based on the risk model, the likelihood of detecting advanced neoplasia in an asymptomatic 32-year-old overweight individual, with a history of previous tobacco use and a first-degree relative younger than age 60 with CRC would be 20.3%, Macaron and colleagues noted.
The model demonstrated “moderate” discriminatory power in the validation set (C-statistic: 0.645), indicating that it can effectively differentiate between individuals at a higher and lower risk for advanced neoplasia.
Additionally, the authors are exploring ways to improve the discriminatory power of the score, possibly by including additional risk factors.
Given the score is calculated using easily obtainable risk factors for individuals younger than 45 who are at risk for early-onset colorectal neoplasia, it could help guide individualized screening decisions for those in whom screening is not currently offered, Macaron said. It could also serve as a tool for risk communication and shared decision-making.
Integration into electronic health records or online calculators may enhance its accessibility and clinical utility.
The authors noted that this retrospective study was conducted at a single center caring mainly for White non-Hispanic adults, limiting generalizability to the general population and to other races and ethnicities.
Validation in Real-World Setting Needed
“There are no currently accepted advanced colorectal neoplasia risk scores that are used in general practice,” said Steven H. Itzkowitz, MD, AGAF, professor of medicine, oncological sciences, and medical education, Icahn School of Medicine at Mount Sinai in New York City. “If these lesions can be predicted, it would enable these young individuals to undergo screening colonoscopy, which could detect and remove these lesions, thereby preventing CRC.”
Many of the known risk factors (family history, high BMI, or smoking) for CRC development at any age are incorporated within this tool, so it should be feasible to collect these data,” said Itzkowitz, who was not involved with the study.
But he cautioned that accurate and adequate family histories are not always performed. Clinicians also may not have considered combining these factors into an actionable risk score.
“If this score can be externally validated in a real-world setting, it could be a useful addition in our efforts to lower CRC rates among young individuals,” Itzkowitz said.
The study did not receive any funding. Macaron and Itzkowitz reported no competing interests.
A version of this article first appeared on Medscape.com.
While colorectal cancer (CRC) incidence has declined overall due to screening, early-onset CRC is on the rise, particularly in individuals younger than 45 years — an age group not currently recommended for CRC screening.
Studies have shown that the risk for early-onset advanced neoplasia varies based on several factors, including sex, race, family history of CRC, smoking, alcohol consumption, diabetes, hyperlipidemia, obesity, and diet.
A score that incorporates some of these factors to identify which younger adults are at higher risk for advanced neoplasia, a precursor to CRC, could support earlier, more targeted screening interventions.
The simple clinical score can be easily calculated by primary care providers in the office, Carole Macaron, MD, lead author of the study and a gastroenterologist at Cleveland Clinic, told GI & Hepatology News. “Patients with a high-risk score would be referred for colorectal cancer screening.”
The study was published in Digestive Diseases and Sciences.
To develop and validate their risk score, Macaron and colleagues did a retrospective cross-sectional analysis of 9446 individuals aged 18-44 years (mean age, 36.8 years; 61% women) who underwent colonoscopy at their center.
Advanced neoplasia was defined as a tubular adenoma ≥ 10 mm or any adenoma with villous features or high-grade dysplasia, sessile serrated polyp ≥ 10 mm, sessile serrated polyp with dysplasia, traditional serrated adenoma, or invasive adenocarcinoma.
The 346 (3.7%) individuals found to have advanced neoplasia served as the case group, and the remainder with normal colonoscopy or non-advanced neoplasia served as controls.
A multivariate logistic regression model identified three independent risk factors significantly associated with advanced neoplasia: Higher body mass index (BMI; P = .0157), former and current tobacco use (P = .0009 and P = .0015, respectively), and having a first-degree relative with CRC < 60 years (P < .0001) or other family history of CRC (P = .0117).
The researchers used these risk factors to develop a risk prediction score to estimate the likelihood of detecting advanced neoplasia, which ranged from a risk of 1.8% for patients with a score of 1 to 22.2% for those with a score of 12. Individuals with a score of ≥ 9 had a 14% or higher risk for advanced neoplasia.
Based on the risk model, the likelihood of detecting advanced neoplasia in an asymptomatic 32-year-old overweight individual, with a history of previous tobacco use and a first-degree relative younger than age 60 with CRC would be 20.3%, Macaron and colleagues noted.
The model demonstrated “moderate” discriminatory power in the validation set (C-statistic: 0.645), indicating that it can effectively differentiate between individuals at a higher and lower risk for advanced neoplasia.
Additionally, the authors are exploring ways to improve the discriminatory power of the score, possibly by including additional risk factors.
Given the score is calculated using easily obtainable risk factors for individuals younger than 45 who are at risk for early-onset colorectal neoplasia, it could help guide individualized screening decisions for those in whom screening is not currently offered, Macaron said. It could also serve as a tool for risk communication and shared decision-making.
Integration into electronic health records or online calculators may enhance its accessibility and clinical utility.
The authors noted that this retrospective study was conducted at a single center caring mainly for White non-Hispanic adults, limiting generalizability to the general population and to other races and ethnicities.
Validation in Real-World Setting Needed
“There are no currently accepted advanced colorectal neoplasia risk scores that are used in general practice,” said Steven H. Itzkowitz, MD, AGAF, professor of medicine, oncological sciences, and medical education, Icahn School of Medicine at Mount Sinai in New York City. “If these lesions can be predicted, it would enable these young individuals to undergo screening colonoscopy, which could detect and remove these lesions, thereby preventing CRC.”
Many of the known risk factors (family history, high BMI, or smoking) for CRC development at any age are incorporated within this tool, so it should be feasible to collect these data,” said Itzkowitz, who was not involved with the study.
But he cautioned that accurate and adequate family histories are not always performed. Clinicians also may not have considered combining these factors into an actionable risk score.
“If this score can be externally validated in a real-world setting, it could be a useful addition in our efforts to lower CRC rates among young individuals,” Itzkowitz said.
The study did not receive any funding. Macaron and Itzkowitz reported no competing interests.
A version of this article first appeared on Medscape.com.
A ‘Fool’s Errand’? Picking a Winner for Treating Early-Stage NSCLC
For years, the default definitive treatment for patients with early-stage I non–small cell lung cancer (NSCLC) has been surgical resection, typically minimally invasive lobectomy with systematic lymph node dissection.
Guidelines from the National Comprehensive Cancer Network (NCCN), the American Society of Clinical Oncology, and the European Society for Medical Oncology all list surgery (in particular, lobectomy) as the primary local therapy for fit, operable patients with stage I NSCLC.
More recently, however, stereotactic body radiotherapy (SBRT), also called stereotactic ablative radiotherapy, has emerged as a definitive treatment option for stage I NSCLC, especially for older, less fit patients who are unsuitable or deemed high-risk for surgery.
“We see patients in our practice who cannot undergo surgery or who may not have adequate lung function to be able to tolerate surgery, and for these patients who are medically inoperable or surgically unresectable, radiation therapy may be a reasonable option,” Charu Aggarwal, MD, MPH, professor and lung cancer specialist, University of Pennsylvania, Philadelphia, told this news organization.
Given some encouraging data suggesting that SBRT could provide similar survival outcomes and be an alternative to surgery for operable disease, SBRT is also increasingly being considered in these early-stage patients. However, other evidence indicates that SBRT may be associated with higher rates of regional and distant recurrences and worse long-term survival, particularly in operable patients.
What may ultimately matter most is carefully selecting operable patients who undergo SBRT.
Aggarwal has encountered certain patients who are fit for surgery but would rather have radiation therapy. “This is an individual decision, and these patients are usually discussed at tumor board and in multidisciplinary discussions to really make sure that they’re making the right decision for themselves,” she explained.
The Pros and Cons of SBRT
SBRT is a nonsurgical approach in which precision high-dose radiation is delivered in just a few fractions — typically, 3, 5, or 8, depending on institutional protocols and tumor characteristics.
SBRT is performed on an outpatient basis, usually over 1-2 weeks, with most patients able to resume usual activities with minimal to no delay. Surgery, on the other hand, requires a hospital stay and takes most people about 2-6 weeks to return to regular activities. SBRT also avoids anesthesia and surgical incisions, both of which come with risks.
The data on SBRT in early-stage NSCLC are mixed. While some studies indicate that SBRT comes with promising survival outcomes, other research has reported worse survival and recurrence rates.
One potential reason for higher recurrence rates with SBRT is the lack of pathologic nodal staging, which only happens after surgery, as well as lower rates of nodal evaluation with endobronchial ultrasound or mediastinoscopy before surgery or SBRT. Without nodal assessments, clinicians may miss a more aggressive histology or more advanced nodal stage, which would go undertreated if patients received SBRT.
Latest Data in Large Cohort
A recent study published in Lung Cancer indicated that, when carefully selected, operable patients with early NSCLC have comparable survival with lobectomy or SBRT.
In the study, Dutch researchers took an in-depth look at survival and recurrence patterns in a retrospective cohort study of 2183 patients with clinical stage I NSCLC treated with minimally invasive lobectomy or SBRT. The study includes one of the largest cohorts to date, with robust data collection on baseline characteristics, comorbidities, tumor size, performance status, and follow-up.
Patients receiving SBRT were typically older (median age, 74 vs 67 years), had higher comorbidity burdens (Charlson index ≥ 5 in 57% of SBRT patients vs 23% of surgical patients), worse performance status, and lower lung function. To adjust for these differences, the researchers used propensity score weighting so the SBRT group’s baseline characteristics were comparable with those in the surgery group.
The surgery cohort had more invasive nodal evaluation: 21% underwent endobronchial ultrasound or mediastinoscopy vs only 12% in the SBRT group. The vast majority in both groups had PET-CT staging, reflecting modern imaging-based workups.
While 5-year local recurrence rates between the two groups were similar (13.1% for SBRT vs 12.1% for surgery), the 5-year regional recurrence rate was significantly higher after SBRT than lobectomy (18.1% vs 14.2%; hazard ratio [HR], 0.74), as was the distant metastasis rate (26.2% vs 20.2%; HR, 0.72).
Mortality at 30 days was higher after surgery than SBRT (1.0% vs 0.2%). And in the unadjusted analysis, 5-year overall survival was significantly better with lobectomy than SBRT (70.2% vs 40.3%).
However, when the analysis only included patients with similar baseline characteristics, overall survival was no longer significantly different in the two groups (HR, 0.89; 95% CI, 0.65-1.20). Lung cancer–specific mortality was also not significantly different between the two treatments (HR, 1.08), but the study was underpowered to detect significant differences in this outcome on the basis of a relatively low number of deaths from NSCLC.
Still, even after comparing similar patients, recurrence-free survival was notably better with surgery (HR, 0.70), due to fewer regional recurrences and distant metastases. Overall, 13% of the surgical cohort had nodal upstaging at pathology, meaning that even in clinically “node-negative” stage I disease, a subset of patients had unsuspected nodal involvement.
Patients receiving SBRT did not have pathologic nodal staging, raising the possibility of occult micrometastases. The authors noted that the proportion of SBRT patients with occult lymph node metastases is likely at least equal to that in the surgery group, but these metastases would go undetected without pathologic assessment.
Missing potential occult micrometastases in the SBRT group likely contributed to higher regional recurrence rates over time. By improving nodal staging, more patients with occult lymph node metastases who would be undertreated with SBRT may be identified before treatment, the authors said.
What Do Experts Say?
So, is SBRT an option for patients with stage I NSCLC?
Opinions vary.
“If you got one shot for a cure, then you want to do the surgery because that’s what results in a cure,” said Raja Flores, MD, chairman of Thoracic Surgery, Mount Sinai Health System, New York City.
Flores noted that the survival rate with surgery is high in this population. “There’s really nothing out there that can compare,” he said.
In his view, surgery “remains the gold standard.” However, “radiation could be considered in nonsurgical candidates,” he said.
The most recent NCCN guidelines align with Flores’ take. The guidelines say that SBRT is indicated for stage IA-IIA (N0) NSCLC in patients who are deemed “medically inoperable, high surgical risk as determined by thoracic surgeon, and those who decline surgery after thoracic surgical consultation.”
Clifford G. Robinson, MD, agreed. “In the United States, we largely treat patients with SBRT who are medically inoperable or high-risk operable and a much smaller proportion who decline surgery,” said Robinson, professor of radiation oncology and chief of SBRT at Washington University in St. Louis, St. Louis. “Many patients who are deemed operable are not offered SBRT.”
Still, for Robinson, determining which patients are best suited for surgery or SBRT remains unclear.
“Retrospective comparisons are fraught with problems because of confounding,” Robinson told this news organization. “That is, the healthier patients get surgery, and the less healthy ones get SBRT. No manner of fancy statistical manipulation can remove that fact.”
In fact, a recent meta-analysis found that the most significant variable predicting whether surgery or SBRT was superior in retrospective studies was whether the author was a surgeon or radiation oncologist.
Robinson noted that multiple randomized trials have attempted to randomize patients with medically operable early-stage NSCLC to surgery or SBRT and failed to accrue, largely due to patients’ “understandable unwillingness to be randomized between operative vs nonoperative interventions when most already prefer one or the other approach.”
Flores highlighted another point of caution about interpreting trial results: Not all early-stage NSCLC behaves similarly. “Some are slow-growing ‘turtles,’ and others are aggressive ‘rabbits’ — and the turtles are usually the ones that have been included in these radiotherapy trials, and that’s the danger,” he said.
While medical operability is the primary factor for deciding the treatment modality for early-stage NSCLC, there are other more subtle factors that can play into the decision.
These include prior surgery or radiotherapy to the chest, prior cancers, and social issues, such as the patient being a primary caregiver for another person and job insecurity, that might make recovery from surgery more challenging. And in rare instances, a patient may be medically fit to undergo surgery, but the cancer is technically challenging to resect due to anatomic issues or prior surgery to the chest, Robinson added.
A Winner?
Results from two ongoing, highly anticipated randomized trials expected in the next several years will hopefully provide additional insights and clarify ongoing uncertainties about the optimal treatment strategies for operable patients with stage I NSCLC.
STABLE-MATES is comparing outcomes after sublobar resection and SBRT in high-risk operable stage I NSCLC, and VALOR is evaluating outcomes after anatomic pulmonary resections and SBRT in patients with stage I NSCLC who have a long life expectancy and are fit enough to tolerate surgery.
But Robinson said his group believes that trying to decide on a winner is a “fool’s errand” and is instead running a pragmatic study across multiple academic and community centers around the United States and Canada where patients choose therapy based on their personal preferences and guidance from their physicians. The researchers will carefully track baseline comorbidity and frailty and assess serial quality-of-life changes over time.
“The goal is to create a calculator that a given patient might use in the future to determine what patients like them would have received, complete with expected outcomes and side effects,” Robinson said.
Robinson cautioned, however, that it “seems unlikely, given the existing literature, that one of the treatments will be truly ‘superior’ to the other one and lead to the ‘losing’ treatment fading away since both are excellent options with pros and cons.”
Aggarwal, Robinson, and Flores had no relevant disclosures.
A version of this article first appeared on Medscape.com.
For years, the default definitive treatment for patients with early-stage I non–small cell lung cancer (NSCLC) has been surgical resection, typically minimally invasive lobectomy with systematic lymph node dissection.
Guidelines from the National Comprehensive Cancer Network (NCCN), the American Society of Clinical Oncology, and the European Society for Medical Oncology all list surgery (in particular, lobectomy) as the primary local therapy for fit, operable patients with stage I NSCLC.
More recently, however, stereotactic body radiotherapy (SBRT), also called stereotactic ablative radiotherapy, has emerged as a definitive treatment option for stage I NSCLC, especially for older, less fit patients who are unsuitable or deemed high-risk for surgery.
“We see patients in our practice who cannot undergo surgery or who may not have adequate lung function to be able to tolerate surgery, and for these patients who are medically inoperable or surgically unresectable, radiation therapy may be a reasonable option,” Charu Aggarwal, MD, MPH, professor and lung cancer specialist, University of Pennsylvania, Philadelphia, told this news organization.
Given some encouraging data suggesting that SBRT could provide similar survival outcomes and be an alternative to surgery for operable disease, SBRT is also increasingly being considered in these early-stage patients. However, other evidence indicates that SBRT may be associated with higher rates of regional and distant recurrences and worse long-term survival, particularly in operable patients.
What may ultimately matter most is carefully selecting operable patients who undergo SBRT.
Aggarwal has encountered certain patients who are fit for surgery but would rather have radiation therapy. “This is an individual decision, and these patients are usually discussed at tumor board and in multidisciplinary discussions to really make sure that they’re making the right decision for themselves,” she explained.
The Pros and Cons of SBRT
SBRT is a nonsurgical approach in which precision high-dose radiation is delivered in just a few fractions — typically, 3, 5, or 8, depending on institutional protocols and tumor characteristics.
SBRT is performed on an outpatient basis, usually over 1-2 weeks, with most patients able to resume usual activities with minimal to no delay. Surgery, on the other hand, requires a hospital stay and takes most people about 2-6 weeks to return to regular activities. SBRT also avoids anesthesia and surgical incisions, both of which come with risks.
The data on SBRT in early-stage NSCLC are mixed. While some studies indicate that SBRT comes with promising survival outcomes, other research has reported worse survival and recurrence rates.
One potential reason for higher recurrence rates with SBRT is the lack of pathologic nodal staging, which only happens after surgery, as well as lower rates of nodal evaluation with endobronchial ultrasound or mediastinoscopy before surgery or SBRT. Without nodal assessments, clinicians may miss a more aggressive histology or more advanced nodal stage, which would go undertreated if patients received SBRT.
Latest Data in Large Cohort
A recent study published in Lung Cancer indicated that, when carefully selected, operable patients with early NSCLC have comparable survival with lobectomy or SBRT.
In the study, Dutch researchers took an in-depth look at survival and recurrence patterns in a retrospective cohort study of 2183 patients with clinical stage I NSCLC treated with minimally invasive lobectomy or SBRT. The study includes one of the largest cohorts to date, with robust data collection on baseline characteristics, comorbidities, tumor size, performance status, and follow-up.
Patients receiving SBRT were typically older (median age, 74 vs 67 years), had higher comorbidity burdens (Charlson index ≥ 5 in 57% of SBRT patients vs 23% of surgical patients), worse performance status, and lower lung function. To adjust for these differences, the researchers used propensity score weighting so the SBRT group’s baseline characteristics were comparable with those in the surgery group.
The surgery cohort had more invasive nodal evaluation: 21% underwent endobronchial ultrasound or mediastinoscopy vs only 12% in the SBRT group. The vast majority in both groups had PET-CT staging, reflecting modern imaging-based workups.
While 5-year local recurrence rates between the two groups were similar (13.1% for SBRT vs 12.1% for surgery), the 5-year regional recurrence rate was significantly higher after SBRT than lobectomy (18.1% vs 14.2%; hazard ratio [HR], 0.74), as was the distant metastasis rate (26.2% vs 20.2%; HR, 0.72).
Mortality at 30 days was higher after surgery than SBRT (1.0% vs 0.2%). And in the unadjusted analysis, 5-year overall survival was significantly better with lobectomy than SBRT (70.2% vs 40.3%).
However, when the analysis only included patients with similar baseline characteristics, overall survival was no longer significantly different in the two groups (HR, 0.89; 95% CI, 0.65-1.20). Lung cancer–specific mortality was also not significantly different between the two treatments (HR, 1.08), but the study was underpowered to detect significant differences in this outcome on the basis of a relatively low number of deaths from NSCLC.
Still, even after comparing similar patients, recurrence-free survival was notably better with surgery (HR, 0.70), due to fewer regional recurrences and distant metastases. Overall, 13% of the surgical cohort had nodal upstaging at pathology, meaning that even in clinically “node-negative” stage I disease, a subset of patients had unsuspected nodal involvement.
Patients receiving SBRT did not have pathologic nodal staging, raising the possibility of occult micrometastases. The authors noted that the proportion of SBRT patients with occult lymph node metastases is likely at least equal to that in the surgery group, but these metastases would go undetected without pathologic assessment.
Missing potential occult micrometastases in the SBRT group likely contributed to higher regional recurrence rates over time. By improving nodal staging, more patients with occult lymph node metastases who would be undertreated with SBRT may be identified before treatment, the authors said.
What Do Experts Say?
So, is SBRT an option for patients with stage I NSCLC?
Opinions vary.
“If you got one shot for a cure, then you want to do the surgery because that’s what results in a cure,” said Raja Flores, MD, chairman of Thoracic Surgery, Mount Sinai Health System, New York City.
Flores noted that the survival rate with surgery is high in this population. “There’s really nothing out there that can compare,” he said.
In his view, surgery “remains the gold standard.” However, “radiation could be considered in nonsurgical candidates,” he said.
The most recent NCCN guidelines align with Flores’ take. The guidelines say that SBRT is indicated for stage IA-IIA (N0) NSCLC in patients who are deemed “medically inoperable, high surgical risk as determined by thoracic surgeon, and those who decline surgery after thoracic surgical consultation.”
Clifford G. Robinson, MD, agreed. “In the United States, we largely treat patients with SBRT who are medically inoperable or high-risk operable and a much smaller proportion who decline surgery,” said Robinson, professor of radiation oncology and chief of SBRT at Washington University in St. Louis, St. Louis. “Many patients who are deemed operable are not offered SBRT.”
Still, for Robinson, determining which patients are best suited for surgery or SBRT remains unclear.
“Retrospective comparisons are fraught with problems because of confounding,” Robinson told this news organization. “That is, the healthier patients get surgery, and the less healthy ones get SBRT. No manner of fancy statistical manipulation can remove that fact.”
In fact, a recent meta-analysis found that the most significant variable predicting whether surgery or SBRT was superior in retrospective studies was whether the author was a surgeon or radiation oncologist.
Robinson noted that multiple randomized trials have attempted to randomize patients with medically operable early-stage NSCLC to surgery or SBRT and failed to accrue, largely due to patients’ “understandable unwillingness to be randomized between operative vs nonoperative interventions when most already prefer one or the other approach.”
Flores highlighted another point of caution about interpreting trial results: Not all early-stage NSCLC behaves similarly. “Some are slow-growing ‘turtles,’ and others are aggressive ‘rabbits’ — and the turtles are usually the ones that have been included in these radiotherapy trials, and that’s the danger,” he said.
While medical operability is the primary factor for deciding the treatment modality for early-stage NSCLC, there are other more subtle factors that can play into the decision.
These include prior surgery or radiotherapy to the chest, prior cancers, and social issues, such as the patient being a primary caregiver for another person and job insecurity, that might make recovery from surgery more challenging. And in rare instances, a patient may be medically fit to undergo surgery, but the cancer is technically challenging to resect due to anatomic issues or prior surgery to the chest, Robinson added.
A Winner?
Results from two ongoing, highly anticipated randomized trials expected in the next several years will hopefully provide additional insights and clarify ongoing uncertainties about the optimal treatment strategies for operable patients with stage I NSCLC.
STABLE-MATES is comparing outcomes after sublobar resection and SBRT in high-risk operable stage I NSCLC, and VALOR is evaluating outcomes after anatomic pulmonary resections and SBRT in patients with stage I NSCLC who have a long life expectancy and are fit enough to tolerate surgery.
But Robinson said his group believes that trying to decide on a winner is a “fool’s errand” and is instead running a pragmatic study across multiple academic and community centers around the United States and Canada where patients choose therapy based on their personal preferences and guidance from their physicians. The researchers will carefully track baseline comorbidity and frailty and assess serial quality-of-life changes over time.
“The goal is to create a calculator that a given patient might use in the future to determine what patients like them would have received, complete with expected outcomes and side effects,” Robinson said.
Robinson cautioned, however, that it “seems unlikely, given the existing literature, that one of the treatments will be truly ‘superior’ to the other one and lead to the ‘losing’ treatment fading away since both are excellent options with pros and cons.”
Aggarwal, Robinson, and Flores had no relevant disclosures.
A version of this article first appeared on Medscape.com.
For years, the default definitive treatment for patients with early-stage I non–small cell lung cancer (NSCLC) has been surgical resection, typically minimally invasive lobectomy with systematic lymph node dissection.
Guidelines from the National Comprehensive Cancer Network (NCCN), the American Society of Clinical Oncology, and the European Society for Medical Oncology all list surgery (in particular, lobectomy) as the primary local therapy for fit, operable patients with stage I NSCLC.
More recently, however, stereotactic body radiotherapy (SBRT), also called stereotactic ablative radiotherapy, has emerged as a definitive treatment option for stage I NSCLC, especially for older, less fit patients who are unsuitable or deemed high-risk for surgery.
“We see patients in our practice who cannot undergo surgery or who may not have adequate lung function to be able to tolerate surgery, and for these patients who are medically inoperable or surgically unresectable, radiation therapy may be a reasonable option,” Charu Aggarwal, MD, MPH, professor and lung cancer specialist, University of Pennsylvania, Philadelphia, told this news organization.
Given some encouraging data suggesting that SBRT could provide similar survival outcomes and be an alternative to surgery for operable disease, SBRT is also increasingly being considered in these early-stage patients. However, other evidence indicates that SBRT may be associated with higher rates of regional and distant recurrences and worse long-term survival, particularly in operable patients.
What may ultimately matter most is carefully selecting operable patients who undergo SBRT.
Aggarwal has encountered certain patients who are fit for surgery but would rather have radiation therapy. “This is an individual decision, and these patients are usually discussed at tumor board and in multidisciplinary discussions to really make sure that they’re making the right decision for themselves,” she explained.
The Pros and Cons of SBRT
SBRT is a nonsurgical approach in which precision high-dose radiation is delivered in just a few fractions — typically, 3, 5, or 8, depending on institutional protocols and tumor characteristics.
SBRT is performed on an outpatient basis, usually over 1-2 weeks, with most patients able to resume usual activities with minimal to no delay. Surgery, on the other hand, requires a hospital stay and takes most people about 2-6 weeks to return to regular activities. SBRT also avoids anesthesia and surgical incisions, both of which come with risks.
The data on SBRT in early-stage NSCLC are mixed. While some studies indicate that SBRT comes with promising survival outcomes, other research has reported worse survival and recurrence rates.
One potential reason for higher recurrence rates with SBRT is the lack of pathologic nodal staging, which only happens after surgery, as well as lower rates of nodal evaluation with endobronchial ultrasound or mediastinoscopy before surgery or SBRT. Without nodal assessments, clinicians may miss a more aggressive histology or more advanced nodal stage, which would go undertreated if patients received SBRT.
Latest Data in Large Cohort
A recent study published in Lung Cancer indicated that, when carefully selected, operable patients with early NSCLC have comparable survival with lobectomy or SBRT.
In the study, Dutch researchers took an in-depth look at survival and recurrence patterns in a retrospective cohort study of 2183 patients with clinical stage I NSCLC treated with minimally invasive lobectomy or SBRT. The study includes one of the largest cohorts to date, with robust data collection on baseline characteristics, comorbidities, tumor size, performance status, and follow-up.
Patients receiving SBRT were typically older (median age, 74 vs 67 years), had higher comorbidity burdens (Charlson index ≥ 5 in 57% of SBRT patients vs 23% of surgical patients), worse performance status, and lower lung function. To adjust for these differences, the researchers used propensity score weighting so the SBRT group’s baseline characteristics were comparable with those in the surgery group.
The surgery cohort had more invasive nodal evaluation: 21% underwent endobronchial ultrasound or mediastinoscopy vs only 12% in the SBRT group. The vast majority in both groups had PET-CT staging, reflecting modern imaging-based workups.
While 5-year local recurrence rates between the two groups were similar (13.1% for SBRT vs 12.1% for surgery), the 5-year regional recurrence rate was significantly higher after SBRT than lobectomy (18.1% vs 14.2%; hazard ratio [HR], 0.74), as was the distant metastasis rate (26.2% vs 20.2%; HR, 0.72).
Mortality at 30 days was higher after surgery than SBRT (1.0% vs 0.2%). And in the unadjusted analysis, 5-year overall survival was significantly better with lobectomy than SBRT (70.2% vs 40.3%).
However, when the analysis only included patients with similar baseline characteristics, overall survival was no longer significantly different in the two groups (HR, 0.89; 95% CI, 0.65-1.20). Lung cancer–specific mortality was also not significantly different between the two treatments (HR, 1.08), but the study was underpowered to detect significant differences in this outcome on the basis of a relatively low number of deaths from NSCLC.
Still, even after comparing similar patients, recurrence-free survival was notably better with surgery (HR, 0.70), due to fewer regional recurrences and distant metastases. Overall, 13% of the surgical cohort had nodal upstaging at pathology, meaning that even in clinically “node-negative” stage I disease, a subset of patients had unsuspected nodal involvement.
Patients receiving SBRT did not have pathologic nodal staging, raising the possibility of occult micrometastases. The authors noted that the proportion of SBRT patients with occult lymph node metastases is likely at least equal to that in the surgery group, but these metastases would go undetected without pathologic assessment.
Missing potential occult micrometastases in the SBRT group likely contributed to higher regional recurrence rates over time. By improving nodal staging, more patients with occult lymph node metastases who would be undertreated with SBRT may be identified before treatment, the authors said.
What Do Experts Say?
So, is SBRT an option for patients with stage I NSCLC?
Opinions vary.
“If you got one shot for a cure, then you want to do the surgery because that’s what results in a cure,” said Raja Flores, MD, chairman of Thoracic Surgery, Mount Sinai Health System, New York City.
Flores noted that the survival rate with surgery is high in this population. “There’s really nothing out there that can compare,” he said.
In his view, surgery “remains the gold standard.” However, “radiation could be considered in nonsurgical candidates,” he said.
The most recent NCCN guidelines align with Flores’ take. The guidelines say that SBRT is indicated for stage IA-IIA (N0) NSCLC in patients who are deemed “medically inoperable, high surgical risk as determined by thoracic surgeon, and those who decline surgery after thoracic surgical consultation.”
Clifford G. Robinson, MD, agreed. “In the United States, we largely treat patients with SBRT who are medically inoperable or high-risk operable and a much smaller proportion who decline surgery,” said Robinson, professor of radiation oncology and chief of SBRT at Washington University in St. Louis, St. Louis. “Many patients who are deemed operable are not offered SBRT.”
Still, for Robinson, determining which patients are best suited for surgery or SBRT remains unclear.
“Retrospective comparisons are fraught with problems because of confounding,” Robinson told this news organization. “That is, the healthier patients get surgery, and the less healthy ones get SBRT. No manner of fancy statistical manipulation can remove that fact.”
In fact, a recent meta-analysis found that the most significant variable predicting whether surgery or SBRT was superior in retrospective studies was whether the author was a surgeon or radiation oncologist.
Robinson noted that multiple randomized trials have attempted to randomize patients with medically operable early-stage NSCLC to surgery or SBRT and failed to accrue, largely due to patients’ “understandable unwillingness to be randomized between operative vs nonoperative interventions when most already prefer one or the other approach.”
Flores highlighted another point of caution about interpreting trial results: Not all early-stage NSCLC behaves similarly. “Some are slow-growing ‘turtles,’ and others are aggressive ‘rabbits’ — and the turtles are usually the ones that have been included in these radiotherapy trials, and that’s the danger,” he said.
While medical operability is the primary factor for deciding the treatment modality for early-stage NSCLC, there are other more subtle factors that can play into the decision.
These include prior surgery or radiotherapy to the chest, prior cancers, and social issues, such as the patient being a primary caregiver for another person and job insecurity, that might make recovery from surgery more challenging. And in rare instances, a patient may be medically fit to undergo surgery, but the cancer is technically challenging to resect due to anatomic issues or prior surgery to the chest, Robinson added.
A Winner?
Results from two ongoing, highly anticipated randomized trials expected in the next several years will hopefully provide additional insights and clarify ongoing uncertainties about the optimal treatment strategies for operable patients with stage I NSCLC.
STABLE-MATES is comparing outcomes after sublobar resection and SBRT in high-risk operable stage I NSCLC, and VALOR is evaluating outcomes after anatomic pulmonary resections and SBRT in patients with stage I NSCLC who have a long life expectancy and are fit enough to tolerate surgery.
But Robinson said his group believes that trying to decide on a winner is a “fool’s errand” and is instead running a pragmatic study across multiple academic and community centers around the United States and Canada where patients choose therapy based on their personal preferences and guidance from their physicians. The researchers will carefully track baseline comorbidity and frailty and assess serial quality-of-life changes over time.
“The goal is to create a calculator that a given patient might use in the future to determine what patients like them would have received, complete with expected outcomes and side effects,” Robinson said.
Robinson cautioned, however, that it “seems unlikely, given the existing literature, that one of the treatments will be truly ‘superior’ to the other one and lead to the ‘losing’ treatment fading away since both are excellent options with pros and cons.”
Aggarwal, Robinson, and Flores had no relevant disclosures.
A version of this article first appeared on Medscape.com.
FDA OKs Guselkumab for Crohn’s Disease
The approval marks the fourth indication for guselkumab, which was approved for moderate to severe plaque psoriasis in 2017, active psoriatic arthritis in 2020, and moderately to severely active ulcerative colitis in 2024.
Guselkumab is the first and only interleukin-23 (IL-23) inhibitor that offers both subcutaneous (SC) and intravenous (IV) induction options for CD, the company said in a news release.
“Despite the progress in the management of Crohn’s disease, many patients experience debilitating symptoms and are in need of new treatment options,” Remo Panaccione, MD, director of the Inflammatory Bowel Disease Unit at the University of Calgary, Calgary, Alberta, Canada, said in the release.
“The approval of Tremfya offers an IL-23 inhibitor that has shown robust rates of endoscopic remission with both subcutaneous and intravenous induction regimens. Importantly, the fully subcutaneous regimen offers choice and flexibility for patients and providers not available before,” said Panaccione.
The FDA nod in CD was based on positive results from three phase 3 trials evaluating guselkumab in more than 1300 patients with moderately to severely active CD who failed or were intolerant to corticosteroids, immunomodulators, or biologics.
The GRAVITI trial showed that guselkumab as SC induction and maintenance therapy was superior to placebo in clinical remission as well as endoscopic response and remission and deep remission.
Results from GALAXI 2 and GALAXI 3 showed that guselkumab was superior to ustekinumab (Stelara) on all pooled endoscopic endpoints.
Guselkumab is the only IL-23 inhibitor to demonstrate “clinical remission and endoscopic response, both at 1 year, with a fully subcutaneous induction regimen,” the company said.
The recommended SC induction dose of guselkumab is 400 mg (given as two consecutive injections of 200 mg each, dispensed in one induction pack) at weeks 0, 4 and 8. The drug is also available in a 200 mg prefilled syringe. For the IV induction option, 200 mg IV infusions are administered at weeks 0, 4, and 8.
The recommended maintenance dosage is 100 mg administered by SC injection at week 16, and every 8 weeks thereafter, or 200 mg administered by SC injection at week 12, and every 4 weeks thereafter.
Use of the lowest effective recommended dosage to maintain therapeutic response is recommended.
Full prescribing information and medication guide are available online.
A version of this article first appeared on Medscape.com.
The approval marks the fourth indication for guselkumab, which was approved for moderate to severe plaque psoriasis in 2017, active psoriatic arthritis in 2020, and moderately to severely active ulcerative colitis in 2024.
Guselkumab is the first and only interleukin-23 (IL-23) inhibitor that offers both subcutaneous (SC) and intravenous (IV) induction options for CD, the company said in a news release.
“Despite the progress in the management of Crohn’s disease, many patients experience debilitating symptoms and are in need of new treatment options,” Remo Panaccione, MD, director of the Inflammatory Bowel Disease Unit at the University of Calgary, Calgary, Alberta, Canada, said in the release.
“The approval of Tremfya offers an IL-23 inhibitor that has shown robust rates of endoscopic remission with both subcutaneous and intravenous induction regimens. Importantly, the fully subcutaneous regimen offers choice and flexibility for patients and providers not available before,” said Panaccione.
The FDA nod in CD was based on positive results from three phase 3 trials evaluating guselkumab in more than 1300 patients with moderately to severely active CD who failed or were intolerant to corticosteroids, immunomodulators, or biologics.
The GRAVITI trial showed that guselkumab as SC induction and maintenance therapy was superior to placebo in clinical remission as well as endoscopic response and remission and deep remission.
Results from GALAXI 2 and GALAXI 3 showed that guselkumab was superior to ustekinumab (Stelara) on all pooled endoscopic endpoints.
Guselkumab is the only IL-23 inhibitor to demonstrate “clinical remission and endoscopic response, both at 1 year, with a fully subcutaneous induction regimen,” the company said.
The recommended SC induction dose of guselkumab is 400 mg (given as two consecutive injections of 200 mg each, dispensed in one induction pack) at weeks 0, 4 and 8. The drug is also available in a 200 mg prefilled syringe. For the IV induction option, 200 mg IV infusions are administered at weeks 0, 4, and 8.
The recommended maintenance dosage is 100 mg administered by SC injection at week 16, and every 8 weeks thereafter, or 200 mg administered by SC injection at week 12, and every 4 weeks thereafter.
Use of the lowest effective recommended dosage to maintain therapeutic response is recommended.
Full prescribing information and medication guide are available online.
A version of this article first appeared on Medscape.com.
The approval marks the fourth indication for guselkumab, which was approved for moderate to severe plaque psoriasis in 2017, active psoriatic arthritis in 2020, and moderately to severely active ulcerative colitis in 2024.
Guselkumab is the first and only interleukin-23 (IL-23) inhibitor that offers both subcutaneous (SC) and intravenous (IV) induction options for CD, the company said in a news release.
“Despite the progress in the management of Crohn’s disease, many patients experience debilitating symptoms and are in need of new treatment options,” Remo Panaccione, MD, director of the Inflammatory Bowel Disease Unit at the University of Calgary, Calgary, Alberta, Canada, said in the release.
“The approval of Tremfya offers an IL-23 inhibitor that has shown robust rates of endoscopic remission with both subcutaneous and intravenous induction regimens. Importantly, the fully subcutaneous regimen offers choice and flexibility for patients and providers not available before,” said Panaccione.
The FDA nod in CD was based on positive results from three phase 3 trials evaluating guselkumab in more than 1300 patients with moderately to severely active CD who failed or were intolerant to corticosteroids, immunomodulators, or biologics.
The GRAVITI trial showed that guselkumab as SC induction and maintenance therapy was superior to placebo in clinical remission as well as endoscopic response and remission and deep remission.
Results from GALAXI 2 and GALAXI 3 showed that guselkumab was superior to ustekinumab (Stelara) on all pooled endoscopic endpoints.
Guselkumab is the only IL-23 inhibitor to demonstrate “clinical remission and endoscopic response, both at 1 year, with a fully subcutaneous induction regimen,” the company said.
The recommended SC induction dose of guselkumab is 400 mg (given as two consecutive injections of 200 mg each, dispensed in one induction pack) at weeks 0, 4 and 8. The drug is also available in a 200 mg prefilled syringe. For the IV induction option, 200 mg IV infusions are administered at weeks 0, 4, and 8.
The recommended maintenance dosage is 100 mg administered by SC injection at week 16, and every 8 weeks thereafter, or 200 mg administered by SC injection at week 12, and every 4 weeks thereafter.
Use of the lowest effective recommended dosage to maintain therapeutic response is recommended.
Full prescribing information and medication guide are available online.
A version of this article first appeared on Medscape.com.
Helping to Protect Our Children From Invasive Pneumococcal Disease
Invasive pneumococcal disease (IPD) remains a serious health threat for infants and can result in hospitalizations, serious complications, or even death.1-3 IPD rates peak at a critical stage in a child’s immune development, when maternal antibody protection wanes and the child has not yet received or is in the process of receiving their primary vaccination series.4 Pneumococcal vaccination is especially important during this vulnerable period to help protect against potentially severe consequences from IPD.2,4,5
Over the last 25 years, the widespread adoption of pneumococcal conjugate vaccines (PCVs) in children has led to a reduction in the spread of many different types of pneumococcal bacteria – referred to as serotypes.2 Although these vaccines have helped reduce the burden of disease, pneumococcal disease remains an issue, with specific serotypes presenting a greater threat to children’s health.6-10
Understanding the burden of IPD in children
According to the Centers for Disease Control and Prevention (CDC), the incidence of IPD is highest in the first year of life,3,* and the death rate due to IPD is higher in infants than in any other pediatric age group.11,† Infants' immune systems are still developing in the first year of life; therefore, protection during this time is critical.3,4,11
The CDC recommends routine pediatric pneumococcal vaccination as a four-dose series at months two, four, and six with a booster administered between 12-15 months.12 Despite the risks associated with invasive pneumococcal disease, some children do not receive all four doses.1-3,13 Many factors can contribute to incomplete childhood immunization coverage, including ethnicity, geographic location, and socioeconomic status.14 In fact, up to one in five babies within the Vaccines for Children Program have received only three of the four recommended PCV doses by two years of age, according to a CDC Morbidity and Mortality Weekly Report from 2021-2023.12,13 The immune response generated after the third dose of a pneumococcal conjugate vaccine is important when evaluating protection against IPD, especially for the children who don't receive their fourth dose.12,15,16
Additionally, certain serotypes, like Serotype 3, are responsible for more IPD cases and are associated with higher morbidity and mortality rates in children.7-10,a Despite being included in PCVs for over a decade, Serotype 3 continues to be a leading cause of IPD in children under five, as shown in a pooled analysis of national-level CDC data from 2018-2022.7,17 This particular serotype has resisted antibody-mediated clearance and continues to be associated with adverse effects.18
What should pediatricians consider when it comes to protecting children from IPD?
When it comes to protecting against IPD, it's important to consider factors in addition to the number of serotypes covered by a vaccine, such as early and robust protection against key serotypes that cause pediatric IPD in the first year of life.2,7,10,19
VAXNEUVANCE® (Pneumococcal 15-valent Conjugate Vaccine) is a pediatric pneumococcal conjugate vaccine that can help deliver strong protection against key disease-causing serotypes during infancy, when the threat of IPD is the highest.2,3,7,10,19-21
Indications and Usage
VAXNEUVANCE is indicated for active immunization for the prevention of invasive disease caused by Streptococcus pneumoniae serotypes 1, 3, 4, 5, 6A, 6B, 7F, 9V, 14, 18C, 19A, 19F, 22F, 23F, and 33F in individuals 6 weeks of age and older.
Select Safety Information
Do not administer VAXNEUVANCE to individuals with a severe allergic reaction (eg, anaphylaxis) to any component of VAXNEUVANCE or to diphtheria toxoid.
Some individuals with altered immunocompetence, including those receiving immunosuppressive therapy, may have a reduced immune response to VAXNEUVANCE.
Apnea following intramuscular vaccination has been observed in some infants born prematurely. Vaccination of premature infants should be based on the infant’s medical status and the potential benefits and possible risks.
(Select Safety Information for VAXNEUVANCE continues below.)
VAXNEUVANCE delivers robust immune responses at seven months, following the third dose, for three key disease-causing serotypes: 3, 22F and 33F.7,10,19,b,c Clinical data showed that immune responses for VAXNEUVANCE were superior to PCV13 (pneumococcal 13-valent conjugate vaccine) for those three critical serotypes2,7,10,19,d and were comparable for the 12 shared serotypes between the vaccines.19
Further, VAXNEUVANCE showcased superior immune responses against Serotype 3 after the third dose with an immunoglobulin G (IgG) geometric mean concentrations (GMCs) response rate of 93.1% compared to PCV13, which demonstrated a 74% response rate.19,b
Although completing the full recommended immunization series remains the best way to help maximize protection,12,22 many children still receive fewer than the recommended four doses of a PCV.12,13 It is important to consider a vaccine that targets problematic serotypes and provides robust immune responses after three doses – of the four dose series – to help protect this vulnerable population from IPD.3,4,7,10,11,19
VAXNEUVANCE can help prevent pediatric IPD in the first year of life and beyond and is an important option for pediatricians to consider for their appropriate patients.7,19
###
Select Safety Information (continued)
The most commonly reported solicited adverse reactions in children vaccinated at 2, 4, 6, and 12 through 15 months of age, provided as a range across the 4-dose series, were: irritability (57.3% to 63.4%), somnolence (24.2% to 47.5%), injection-site pain (25.9% to 40.3%), fever ≥38.0°C (13.3% to 20.4%), decreased appetite (14.1% to 19.0%), injection-site induration (13.2% to 15.4%), injection-site erythema (13.7% to 21.4%) and injection-site swelling (11.3% to 13.4%).
The most commonly reported solicited adverse reactions in children 2 through 17 years of age vaccinated with a single dose were: injection-site pain (54.8%), myalgia (23.7%), injection-site swelling (20.9%), injection-site erythema (19.2%), fatigue (15.8%), headache (11.9%) and injection-site induration (6.8%).
Vaccination with VAXNEUVANCE may not protect all vaccine recipients.
Before administering VAXNEUVANCE, please read the accompanying Prescribing Information. The Patient Information also is available.
* Based on pooled analysis of national-level CDC ABC surveillance data from 2018–2022, representing ~35 million people surveyed annually in 10 states across the US. IPD incidence rates were 10.3 in <1 year, 8.2 in 1 year, 4.0 in 2–4 years, 5.0 in 1–4 years, and 1.3 in 5–17 years (Regional variations may exist).3
† Based on national-level CDC ABC surveillance data from 2022, representing ~35 million people in 10 states across the US (Regional variations may exist).11
Key Study Details
GMC Ratios Postdose 3c
Primary endpoint: VAXNEUVANCE delivered comparable immune responses for 12 of the 13 shared serotypes found in PCV13. Shared Serotype 6A was just below the noninferiority criteria by a small margin, with the lower bound of the 2-sided 95% CI for the GMC ratio being 0.48 vs >0.5.19,23
Study Design
Study 8 was a pivotal, double-blind, active comparator-controlled study in which participants were randomized to receive VAXNEUVANCE (N=860) or PCV13 (N=860) in a 4-dose series. The first 3 doses were administered to infants at 2, 4, and 6 months of age and the fourth dose was administered to children at 12 through 15 months of age. Participants also received other licensed pediatric vaccines concomitantly. Immune responses were measured by IgG response rates, IgG GMCs, and OPA GMTs for all 15 serotypes contained in VAXNEUVANCE.19
aBased on a pooled analysis of national-level CDC data from 2018–2021, the top 6 IPD-causing serotypes in children under 5 years of age were 15C, 33F, 19F, 3, 23B, and 22F. Serotypes 15C and 23B are not included in any recommended pediatric PCV in the US.7,17,19,22,24
bPostdose 3 superiority was demonstrated based on measurements taken 30 days after the 6-month dose (at 7 months).19
cMeasurements were taken 30 days postdose specified.19
dSecondary endpoint: Postdose 3 IgG response rate percentage point difference vs PCV13 (95% CI): for Serotype 3, 19.1 (14.4, 24.0); for Serotype 22F, 8.1 (5.1, 11.5); for Serotype 33F, -5.1 (-9.5, -0.7).19,23
Randomized controlled trials assessing the clinical efficacy of VAXNEUVANCE compared to PCV13 have not been conducted.19
References:
1Dalton M. Pneumoccal disease. National Foundation for Infectious Diseases. Published July 2024. https://www.nfid.org/infectious-disease/pneumococcal/
2Gierke R, Wodi P, Kobayashi M. Epidemiology and Prevention of Vaccine-Preventable Diseases (Pink Book). 14th edition. Chapter 17: Pneumococcal disease. Epidemiology and Prevention of Vaccine-Preventable Diseases. Published May 1, 2024. Accessed December 10, 2024. https://www.cdc.gov/pinkbook/hcp/table-of-contents/chapter-17-pneumococcal-disease.html
3Data available on request from the Merck National Service Center via email at [email protected]. Please specify information package US-PVC-02072.
4Mohanty S, Done N, Liu Q, et al. Incidence of pneumococcal disease in children ≤48 months old in the United States: 1998–2019. Vaccine. Published online March 1, 2024. doi: 10.1016/j.vaccine.2024.03.013
5Clinical overview of pneumococcal disease. Centers for Disease Control and Prevention. February 6, 2024. Accessed May 22, 2024. https://www.cdc.gov/pneumococcal/hcp/clinical-overview/
6Wasserman MD, Perdrizet J, Grant L, et al. Clinical and economic burden of pneumococcal disease due to serotypes contained in current and investigational pneumococcal conjugate vaccines in children under five years of age. Infect Dis Ther. 2021;10(4):2701-2720. doi:10.1007/s40121-021-00544-1
7Centers for Disease Control and Prevention (CDC). Visualization – Based on 1998-2022 serotype data for invasive pneumococcal disease cases by age group from Active Bacterial Core surveillance (ABCs). Updated July 22, 2024. Accessed August 30, 2024. https://data.cdc.gov/Public-Health-Surveillance/1998-2022-Serotype-Data-for-Invasive-Pneumococcal-/qvzb-qs6p/about_data
8Varghese J, Chochua S, Tran T, et al. Multistate population and whole genome sequence-based strain surveillance of invasive pneumococci recovered in the USA during 2017. Clin Microbiol Infect. 2020;26(4):512.e1-512.e10. doi:10.1016/j.cmi.2019.09.008
9Azarian T, Mitchell PK, Georgieva M, et al. Global emergence and population dynamics of divergent serotype 3 CC180 pneumococci. PLoS Pathog. 2018;14(11):e1007438. doi:10.1371/journal.ppat.1007438
10Hu T, Weiss T, Owusu-Edusei K, Petigara T. Health and economic burden associated with 15-valent pneumococcal conjugate vaccine serotypes in children in the United States. J Med Econ. 2020;23(12):1653-1660. doi:10.1080/13696998.2020.184021613
11Active Bacterial Core surveillance (ABCs) report, Emerging Infections Program network, Streptococcus pneumoniae, 2022. Centers for Disease Control and Prevention. Updated July 5, 2024. Accessed October 15, 2024. https://www.cdc.gov/abcs/downloads/SPN_Surveillance_Report_2022.pdf
12Recommended child and adolescent immunization schedule for ages 18 years or younger, United States, 2025. Centers for Disease Control and Prevention. Addendum updated November 21, 2024. Accessed November 25, 2024. https://www.cdc.gov/vaccines/hcp/imz-schedules/downloads/child/0-18yrs-child-combined-schedule.pdf
13Hill HA, et al. Decline in Vaccination Coverage by Age 24 Months and Vaccination Inequities Among Children Born in 2020 and 2021 — National Immunization Survey-Child, United States, 2021–2023. MMWR Morb Mortal Wkly Rep, pages 844–853.
14Feemster K, Weaver J, Buchwald U, Banniettis N, Cox KS, McIntosh ED, Spoulou V. Pneumococcal Vaccine Breakthrough and Failure in Infants and Children: A Narrative Review. Vaccines (Basel). 2023 Nov 24;11(12):1750. doi:10.3390/vaccines11121750. PMID: 38140155; PMCID: PMC10747311.
15Recommendations to assure the quality, safety and efficacy of pneumoccoccal conjugate vaccines. Annex 3. TRS no 977. World Health Organization. October 19, 2013. Accessed October 31, 2024. https://www.who.int/publications/m/item/pneumococcal-conjugate-vaccines-annex3-trs-977
16Guidelines on clinical evaluation of vaccines: regulatory expectations. Annex 9. TRA No 924.World Health Organization. Last reviewed October 21, 2020. Accessed October 31, 2024. https://www.who.int/publications/m/item/WHO-TRS-1004-web-annex-9
17Prevnar 13. Prescribing Information. Pfizer; 2019.
18Luck JN, Tettelin H, Orihuela CJ. Sugar-Coated Killer: Serotype 3 Pneumococcal Disease. Front Cell Infect Microbiol. 2020;10:613287. Published 2020 Dec 23. doi:10.3389/fcimb.2020.613287
19VAXNEUVANCE. Prescribing Information. Merck & Co., Inc., 2024.
20Moraes-Pinto MI, Suano-Souza F, Aranda CS. Immune system: development and acquisition of immunological competence. J Pediatr (Rio J). 2021;97(S1):S59-S66. doi:10.1016/j.jped.2020.10.006
21Wodi AP, Morelli V. Epidemiology and Prevention of Vaccine-Preventable Diseases (Pink Book). 14th edition. Chapter 1: Principles of vaccination. Centers for Disease Control and Prevention. Last reviewed March 2024. Accessed May 9, 2024. https://www.cdc.gov/pinkbook/hcp/table-of-contents/chapter-1-principles-of-vaccination.html
22Pneumococcal vaccination. Centers for Disease Control and Prevention. Last reviewed September 12, 2024. Accessed September 30, 2024. https://www.cdc.gov/pneumococcal/vaccines/index.html
23Lupinacci R, Rupp R, Wittawatmongkol O, et al. A phase 3, multicenter, randomized, double-blind, active-comparator-controlled study to evaluate the safety, tolerability, and immunogenicity of a 4-dose regimen of V114, a 15-valent pneumococcal conjugate vaccine, in healthy infants (PNEU-PED). Vaccine. 2023;41(5):1142-1152. doi:10.1016/j.vaccine.2022.12.054
24Prevnar 20. Prescribing Information. Pfizer; 2023.
Copyright © 2025 Frontline Medical Communications Inc. All rights reserved. No part of this publication may be reproduced or transmitted in any form, by any means, without prior written permission of the Publisher. Frontline Medical Communications Inc. will not assume responsibility for damages, loss, or claims of any kind arising from or related to the information contained in this publication, including any claims related to the products, drugs, or services mentioned herein. The opinions expressed in this publication do not necessarily reflect the views of the Publisher. All other trademarks are property of their respective owners.
Neither the editors of Pediatric News nor the Editorial Advisory Board nor the reporting staff contributed to this content.
US-PVC-01998 03/25
Invasive pneumococcal disease (IPD) remains a serious health threat for infants and can result in hospitalizations, serious complications, or even death.1-3 IPD rates peak at a critical stage in a child’s immune development, when maternal antibody protection wanes and the child has not yet received or is in the process of receiving their primary vaccination series.4 Pneumococcal vaccination is especially important during this vulnerable period to help protect against potentially severe consequences from IPD.2,4,5
Over the last 25 years, the widespread adoption of pneumococcal conjugate vaccines (PCVs) in children has led to a reduction in the spread of many different types of pneumococcal bacteria – referred to as serotypes.2 Although these vaccines have helped reduce the burden of disease, pneumococcal disease remains an issue, with specific serotypes presenting a greater threat to children’s health.6-10
Understanding the burden of IPD in children
According to the Centers for Disease Control and Prevention (CDC), the incidence of IPD is highest in the first year of life,3,* and the death rate due to IPD is higher in infants than in any other pediatric age group.11,† Infants' immune systems are still developing in the first year of life; therefore, protection during this time is critical.3,4,11
The CDC recommends routine pediatric pneumococcal vaccination as a four-dose series at months two, four, and six with a booster administered between 12-15 months.12 Despite the risks associated with invasive pneumococcal disease, some children do not receive all four doses.1-3,13 Many factors can contribute to incomplete childhood immunization coverage, including ethnicity, geographic location, and socioeconomic status.14 In fact, up to one in five babies within the Vaccines for Children Program have received only three of the four recommended PCV doses by two years of age, according to a CDC Morbidity and Mortality Weekly Report from 2021-2023.12,13 The immune response generated after the third dose of a pneumococcal conjugate vaccine is important when evaluating protection against IPD, especially for the children who don't receive their fourth dose.12,15,16
Additionally, certain serotypes, like Serotype 3, are responsible for more IPD cases and are associated with higher morbidity and mortality rates in children.7-10,a Despite being included in PCVs for over a decade, Serotype 3 continues to be a leading cause of IPD in children under five, as shown in a pooled analysis of national-level CDC data from 2018-2022.7,17 This particular serotype has resisted antibody-mediated clearance and continues to be associated with adverse effects.18
What should pediatricians consider when it comes to protecting children from IPD?
When it comes to protecting against IPD, it's important to consider factors in addition to the number of serotypes covered by a vaccine, such as early and robust protection against key serotypes that cause pediatric IPD in the first year of life.2,7,10,19
VAXNEUVANCE® (Pneumococcal 15-valent Conjugate Vaccine) is a pediatric pneumococcal conjugate vaccine that can help deliver strong protection against key disease-causing serotypes during infancy, when the threat of IPD is the highest.2,3,7,10,19-21
Indications and Usage
VAXNEUVANCE is indicated for active immunization for the prevention of invasive disease caused by Streptococcus pneumoniae serotypes 1, 3, 4, 5, 6A, 6B, 7F, 9V, 14, 18C, 19A, 19F, 22F, 23F, and 33F in individuals 6 weeks of age and older.
Select Safety Information
Do not administer VAXNEUVANCE to individuals with a severe allergic reaction (eg, anaphylaxis) to any component of VAXNEUVANCE or to diphtheria toxoid.
Some individuals with altered immunocompetence, including those receiving immunosuppressive therapy, may have a reduced immune response to VAXNEUVANCE.
Apnea following intramuscular vaccination has been observed in some infants born prematurely. Vaccination of premature infants should be based on the infant’s medical status and the potential benefits and possible risks.
(Select Safety Information for VAXNEUVANCE continues below.)
VAXNEUVANCE delivers robust immune responses at seven months, following the third dose, for three key disease-causing serotypes: 3, 22F and 33F.7,10,19,b,c Clinical data showed that immune responses for VAXNEUVANCE were superior to PCV13 (pneumococcal 13-valent conjugate vaccine) for those three critical serotypes2,7,10,19,d and were comparable for the 12 shared serotypes between the vaccines.19
Further, VAXNEUVANCE showcased superior immune responses against Serotype 3 after the third dose with an immunoglobulin G (IgG) geometric mean concentrations (GMCs) response rate of 93.1% compared to PCV13, which demonstrated a 74% response rate.19,b
Although completing the full recommended immunization series remains the best way to help maximize protection,12,22 many children still receive fewer than the recommended four doses of a PCV.12,13 It is important to consider a vaccine that targets problematic serotypes and provides robust immune responses after three doses – of the four dose series – to help protect this vulnerable population from IPD.3,4,7,10,11,19
VAXNEUVANCE can help prevent pediatric IPD in the first year of life and beyond and is an important option for pediatricians to consider for their appropriate patients.7,19
###
Select Safety Information (continued)
The most commonly reported solicited adverse reactions in children vaccinated at 2, 4, 6, and 12 through 15 months of age, provided as a range across the 4-dose series, were: irritability (57.3% to 63.4%), somnolence (24.2% to 47.5%), injection-site pain (25.9% to 40.3%), fever ≥38.0°C (13.3% to 20.4%), decreased appetite (14.1% to 19.0%), injection-site induration (13.2% to 15.4%), injection-site erythema (13.7% to 21.4%) and injection-site swelling (11.3% to 13.4%).
The most commonly reported solicited adverse reactions in children 2 through 17 years of age vaccinated with a single dose were: injection-site pain (54.8%), myalgia (23.7%), injection-site swelling (20.9%), injection-site erythema (19.2%), fatigue (15.8%), headache (11.9%) and injection-site induration (6.8%).
Vaccination with VAXNEUVANCE may not protect all vaccine recipients.
Before administering VAXNEUVANCE, please read the accompanying Prescribing Information. The Patient Information also is available.
* Based on pooled analysis of national-level CDC ABC surveillance data from 2018–2022, representing ~35 million people surveyed annually in 10 states across the US. IPD incidence rates were 10.3 in <1 year, 8.2 in 1 year, 4.0 in 2–4 years, 5.0 in 1–4 years, and 1.3 in 5–17 years (Regional variations may exist).3
† Based on national-level CDC ABC surveillance data from 2022, representing ~35 million people in 10 states across the US (Regional variations may exist).11
Key Study Details
GMC Ratios Postdose 3c
Primary endpoint: VAXNEUVANCE delivered comparable immune responses for 12 of the 13 shared serotypes found in PCV13. Shared Serotype 6A was just below the noninferiority criteria by a small margin, with the lower bound of the 2-sided 95% CI for the GMC ratio being 0.48 vs >0.5.19,23
Study Design
Study 8 was a pivotal, double-blind, active comparator-controlled study in which participants were randomized to receive VAXNEUVANCE (N=860) or PCV13 (N=860) in a 4-dose series. The first 3 doses were administered to infants at 2, 4, and 6 months of age and the fourth dose was administered to children at 12 through 15 months of age. Participants also received other licensed pediatric vaccines concomitantly. Immune responses were measured by IgG response rates, IgG GMCs, and OPA GMTs for all 15 serotypes contained in VAXNEUVANCE.19
aBased on a pooled analysis of national-level CDC data from 2018–2021, the top 6 IPD-causing serotypes in children under 5 years of age were 15C, 33F, 19F, 3, 23B, and 22F. Serotypes 15C and 23B are not included in any recommended pediatric PCV in the US.7,17,19,22,24
bPostdose 3 superiority was demonstrated based on measurements taken 30 days after the 6-month dose (at 7 months).19
cMeasurements were taken 30 days postdose specified.19
dSecondary endpoint: Postdose 3 IgG response rate percentage point difference vs PCV13 (95% CI): for Serotype 3, 19.1 (14.4, 24.0); for Serotype 22F, 8.1 (5.1, 11.5); for Serotype 33F, -5.1 (-9.5, -0.7).19,23
Randomized controlled trials assessing the clinical efficacy of VAXNEUVANCE compared to PCV13 have not been conducted.19
References:
1Dalton M. Pneumoccal disease. National Foundation for Infectious Diseases. Published July 2024. https://www.nfid.org/infectious-disease/pneumococcal/
2Gierke R, Wodi P, Kobayashi M. Epidemiology and Prevention of Vaccine-Preventable Diseases (Pink Book). 14th edition. Chapter 17: Pneumococcal disease. Epidemiology and Prevention of Vaccine-Preventable Diseases. Published May 1, 2024. Accessed December 10, 2024. https://www.cdc.gov/pinkbook/hcp/table-of-contents/chapter-17-pneumococcal-disease.html
3Data available on request from the Merck National Service Center via email at [email protected]. Please specify information package US-PVC-02072.
4Mohanty S, Done N, Liu Q, et al. Incidence of pneumococcal disease in children ≤48 months old in the United States: 1998–2019. Vaccine. Published online March 1, 2024. doi: 10.1016/j.vaccine.2024.03.013
5Clinical overview of pneumococcal disease. Centers for Disease Control and Prevention. February 6, 2024. Accessed May 22, 2024. https://www.cdc.gov/pneumococcal/hcp/clinical-overview/
6Wasserman MD, Perdrizet J, Grant L, et al. Clinical and economic burden of pneumococcal disease due to serotypes contained in current and investigational pneumococcal conjugate vaccines in children under five years of age. Infect Dis Ther. 2021;10(4):2701-2720. doi:10.1007/s40121-021-00544-1
7Centers for Disease Control and Prevention (CDC). Visualization – Based on 1998-2022 serotype data for invasive pneumococcal disease cases by age group from Active Bacterial Core surveillance (ABCs). Updated July 22, 2024. Accessed August 30, 2024. https://data.cdc.gov/Public-Health-Surveillance/1998-2022-Serotype-Data-for-Invasive-Pneumococcal-/qvzb-qs6p/about_data
8Varghese J, Chochua S, Tran T, et al. Multistate population and whole genome sequence-based strain surveillance of invasive pneumococci recovered in the USA during 2017. Clin Microbiol Infect. 2020;26(4):512.e1-512.e10. doi:10.1016/j.cmi.2019.09.008
9Azarian T, Mitchell PK, Georgieva M, et al. Global emergence and population dynamics of divergent serotype 3 CC180 pneumococci. PLoS Pathog. 2018;14(11):e1007438. doi:10.1371/journal.ppat.1007438
10Hu T, Weiss T, Owusu-Edusei K, Petigara T. Health and economic burden associated with 15-valent pneumococcal conjugate vaccine serotypes in children in the United States. J Med Econ. 2020;23(12):1653-1660. doi:10.1080/13696998.2020.184021613
11Active Bacterial Core surveillance (ABCs) report, Emerging Infections Program network, Streptococcus pneumoniae, 2022. Centers for Disease Control and Prevention. Updated July 5, 2024. Accessed October 15, 2024. https://www.cdc.gov/abcs/downloads/SPN_Surveillance_Report_2022.pdf
12Recommended child and adolescent immunization schedule for ages 18 years or younger, United States, 2025. Centers for Disease Control and Prevention. Addendum updated November 21, 2024. Accessed November 25, 2024. https://www.cdc.gov/vaccines/hcp/imz-schedules/downloads/child/0-18yrs-child-combined-schedule.pdf
13Hill HA, et al. Decline in Vaccination Coverage by Age 24 Months and Vaccination Inequities Among Children Born in 2020 and 2021 — National Immunization Survey-Child, United States, 2021–2023. MMWR Morb Mortal Wkly Rep, pages 844–853.
14Feemster K, Weaver J, Buchwald U, Banniettis N, Cox KS, McIntosh ED, Spoulou V. Pneumococcal Vaccine Breakthrough and Failure in Infants and Children: A Narrative Review. Vaccines (Basel). 2023 Nov 24;11(12):1750. doi:10.3390/vaccines11121750. PMID: 38140155; PMCID: PMC10747311.
15Recommendations to assure the quality, safety and efficacy of pneumoccoccal conjugate vaccines. Annex 3. TRS no 977. World Health Organization. October 19, 2013. Accessed October 31, 2024. https://www.who.int/publications/m/item/pneumococcal-conjugate-vaccines-annex3-trs-977
16Guidelines on clinical evaluation of vaccines: regulatory expectations. Annex 9. TRA No 924.World Health Organization. Last reviewed October 21, 2020. Accessed October 31, 2024. https://www.who.int/publications/m/item/WHO-TRS-1004-web-annex-9
17Prevnar 13. Prescribing Information. Pfizer; 2019.
18Luck JN, Tettelin H, Orihuela CJ. Sugar-Coated Killer: Serotype 3 Pneumococcal Disease. Front Cell Infect Microbiol. 2020;10:613287. Published 2020 Dec 23. doi:10.3389/fcimb.2020.613287
19VAXNEUVANCE. Prescribing Information. Merck & Co., Inc., 2024.
20Moraes-Pinto MI, Suano-Souza F, Aranda CS. Immune system: development and acquisition of immunological competence. J Pediatr (Rio J). 2021;97(S1):S59-S66. doi:10.1016/j.jped.2020.10.006
21Wodi AP, Morelli V. Epidemiology and Prevention of Vaccine-Preventable Diseases (Pink Book). 14th edition. Chapter 1: Principles of vaccination. Centers for Disease Control and Prevention. Last reviewed March 2024. Accessed May 9, 2024. https://www.cdc.gov/pinkbook/hcp/table-of-contents/chapter-1-principles-of-vaccination.html
22Pneumococcal vaccination. Centers for Disease Control and Prevention. Last reviewed September 12, 2024. Accessed September 30, 2024. https://www.cdc.gov/pneumococcal/vaccines/index.html
23Lupinacci R, Rupp R, Wittawatmongkol O, et al. A phase 3, multicenter, randomized, double-blind, active-comparator-controlled study to evaluate the safety, tolerability, and immunogenicity of a 4-dose regimen of V114, a 15-valent pneumococcal conjugate vaccine, in healthy infants (PNEU-PED). Vaccine. 2023;41(5):1142-1152. doi:10.1016/j.vaccine.2022.12.054
24Prevnar 20. Prescribing Information. Pfizer; 2023.
Copyright © 2025 Frontline Medical Communications Inc. All rights reserved. No part of this publication may be reproduced or transmitted in any form, by any means, without prior written permission of the Publisher. Frontline Medical Communications Inc. will not assume responsibility for damages, loss, or claims of any kind arising from or related to the information contained in this publication, including any claims related to the products, drugs, or services mentioned herein. The opinions expressed in this publication do not necessarily reflect the views of the Publisher. All other trademarks are property of their respective owners.
Neither the editors of Pediatric News nor the Editorial Advisory Board nor the reporting staff contributed to this content.
US-PVC-01998 03/25
Invasive pneumococcal disease (IPD) remains a serious health threat for infants and can result in hospitalizations, serious complications, or even death.1-3 IPD rates peak at a critical stage in a child’s immune development, when maternal antibody protection wanes and the child has not yet received or is in the process of receiving their primary vaccination series.4 Pneumococcal vaccination is especially important during this vulnerable period to help protect against potentially severe consequences from IPD.2,4,5
Over the last 25 years, the widespread adoption of pneumococcal conjugate vaccines (PCVs) in children has led to a reduction in the spread of many different types of pneumococcal bacteria – referred to as serotypes.2 Although these vaccines have helped reduce the burden of disease, pneumococcal disease remains an issue, with specific serotypes presenting a greater threat to children’s health.6-10
Understanding the burden of IPD in children
According to the Centers for Disease Control and Prevention (CDC), the incidence of IPD is highest in the first year of life,3,* and the death rate due to IPD is higher in infants than in any other pediatric age group.11,† Infants' immune systems are still developing in the first year of life; therefore, protection during this time is critical.3,4,11
The CDC recommends routine pediatric pneumococcal vaccination as a four-dose series at months two, four, and six with a booster administered between 12-15 months.12 Despite the risks associated with invasive pneumococcal disease, some children do not receive all four doses.1-3,13 Many factors can contribute to incomplete childhood immunization coverage, including ethnicity, geographic location, and socioeconomic status.14 In fact, up to one in five babies within the Vaccines for Children Program have received only three of the four recommended PCV doses by two years of age, according to a CDC Morbidity and Mortality Weekly Report from 2021-2023.12,13 The immune response generated after the third dose of a pneumococcal conjugate vaccine is important when evaluating protection against IPD, especially for the children who don't receive their fourth dose.12,15,16
Additionally, certain serotypes, like Serotype 3, are responsible for more IPD cases and are associated with higher morbidity and mortality rates in children.7-10,a Despite being included in PCVs for over a decade, Serotype 3 continues to be a leading cause of IPD in children under five, as shown in a pooled analysis of national-level CDC data from 2018-2022.7,17 This particular serotype has resisted antibody-mediated clearance and continues to be associated with adverse effects.18
What should pediatricians consider when it comes to protecting children from IPD?
When it comes to protecting against IPD, it's important to consider factors in addition to the number of serotypes covered by a vaccine, such as early and robust protection against key serotypes that cause pediatric IPD in the first year of life.2,7,10,19
VAXNEUVANCE® (Pneumococcal 15-valent Conjugate Vaccine) is a pediatric pneumococcal conjugate vaccine that can help deliver strong protection against key disease-causing serotypes during infancy, when the threat of IPD is the highest.2,3,7,10,19-21
Indications and Usage
VAXNEUVANCE is indicated for active immunization for the prevention of invasive disease caused by Streptococcus pneumoniae serotypes 1, 3, 4, 5, 6A, 6B, 7F, 9V, 14, 18C, 19A, 19F, 22F, 23F, and 33F in individuals 6 weeks of age and older.
Select Safety Information
Do not administer VAXNEUVANCE to individuals with a severe allergic reaction (eg, anaphylaxis) to any component of VAXNEUVANCE or to diphtheria toxoid.
Some individuals with altered immunocompetence, including those receiving immunosuppressive therapy, may have a reduced immune response to VAXNEUVANCE.
Apnea following intramuscular vaccination has been observed in some infants born prematurely. Vaccination of premature infants should be based on the infant’s medical status and the potential benefits and possible risks.
(Select Safety Information for VAXNEUVANCE continues below.)
VAXNEUVANCE delivers robust immune responses at seven months, following the third dose, for three key disease-causing serotypes: 3, 22F and 33F.7,10,19,b,c Clinical data showed that immune responses for VAXNEUVANCE were superior to PCV13 (pneumococcal 13-valent conjugate vaccine) for those three critical serotypes2,7,10,19,d and were comparable for the 12 shared serotypes between the vaccines.19
Further, VAXNEUVANCE showcased superior immune responses against Serotype 3 after the third dose with an immunoglobulin G (IgG) geometric mean concentrations (GMCs) response rate of 93.1% compared to PCV13, which demonstrated a 74% response rate.19,b
Although completing the full recommended immunization series remains the best way to help maximize protection,12,22 many children still receive fewer than the recommended four doses of a PCV.12,13 It is important to consider a vaccine that targets problematic serotypes and provides robust immune responses after three doses – of the four dose series – to help protect this vulnerable population from IPD.3,4,7,10,11,19
VAXNEUVANCE can help prevent pediatric IPD in the first year of life and beyond and is an important option for pediatricians to consider for their appropriate patients.7,19
###
Select Safety Information (continued)
The most commonly reported solicited adverse reactions in children vaccinated at 2, 4, 6, and 12 through 15 months of age, provided as a range across the 4-dose series, were: irritability (57.3% to 63.4%), somnolence (24.2% to 47.5%), injection-site pain (25.9% to 40.3%), fever ≥38.0°C (13.3% to 20.4%), decreased appetite (14.1% to 19.0%), injection-site induration (13.2% to 15.4%), injection-site erythema (13.7% to 21.4%) and injection-site swelling (11.3% to 13.4%).
The most commonly reported solicited adverse reactions in children 2 through 17 years of age vaccinated with a single dose were: injection-site pain (54.8%), myalgia (23.7%), injection-site swelling (20.9%), injection-site erythema (19.2%), fatigue (15.8%), headache (11.9%) and injection-site induration (6.8%).
Vaccination with VAXNEUVANCE may not protect all vaccine recipients.
Before administering VAXNEUVANCE, please read the accompanying Prescribing Information. The Patient Information also is available.
* Based on pooled analysis of national-level CDC ABC surveillance data from 2018–2022, representing ~35 million people surveyed annually in 10 states across the US. IPD incidence rates were 10.3 in <1 year, 8.2 in 1 year, 4.0 in 2–4 years, 5.0 in 1–4 years, and 1.3 in 5–17 years (Regional variations may exist).3
† Based on national-level CDC ABC surveillance data from 2022, representing ~35 million people in 10 states across the US (Regional variations may exist).11
Key Study Details
GMC Ratios Postdose 3c
Primary endpoint: VAXNEUVANCE delivered comparable immune responses for 12 of the 13 shared serotypes found in PCV13. Shared Serotype 6A was just below the noninferiority criteria by a small margin, with the lower bound of the 2-sided 95% CI for the GMC ratio being 0.48 vs >0.5.19,23
Study Design
Study 8 was a pivotal, double-blind, active comparator-controlled study in which participants were randomized to receive VAXNEUVANCE (N=860) or PCV13 (N=860) in a 4-dose series. The first 3 doses were administered to infants at 2, 4, and 6 months of age and the fourth dose was administered to children at 12 through 15 months of age. Participants also received other licensed pediatric vaccines concomitantly. Immune responses were measured by IgG response rates, IgG GMCs, and OPA GMTs for all 15 serotypes contained in VAXNEUVANCE.19
aBased on a pooled analysis of national-level CDC data from 2018–2021, the top 6 IPD-causing serotypes in children under 5 years of age were 15C, 33F, 19F, 3, 23B, and 22F. Serotypes 15C and 23B are not included in any recommended pediatric PCV in the US.7,17,19,22,24
bPostdose 3 superiority was demonstrated based on measurements taken 30 days after the 6-month dose (at 7 months).19
cMeasurements were taken 30 days postdose specified.19
dSecondary endpoint: Postdose 3 IgG response rate percentage point difference vs PCV13 (95% CI): for Serotype 3, 19.1 (14.4, 24.0); for Serotype 22F, 8.1 (5.1, 11.5); for Serotype 33F, -5.1 (-9.5, -0.7).19,23
Randomized controlled trials assessing the clinical efficacy of VAXNEUVANCE compared to PCV13 have not been conducted.19
References:
1Dalton M. Pneumoccal disease. National Foundation for Infectious Diseases. Published July 2024. https://www.nfid.org/infectious-disease/pneumococcal/
2Gierke R, Wodi P, Kobayashi M. Epidemiology and Prevention of Vaccine-Preventable Diseases (Pink Book). 14th edition. Chapter 17: Pneumococcal disease. Epidemiology and Prevention of Vaccine-Preventable Diseases. Published May 1, 2024. Accessed December 10, 2024. https://www.cdc.gov/pinkbook/hcp/table-of-contents/chapter-17-pneumococcal-disease.html
3Data available on request from the Merck National Service Center via email at [email protected]. Please specify information package US-PVC-02072.
4Mohanty S, Done N, Liu Q, et al. Incidence of pneumococcal disease in children ≤48 months old in the United States: 1998–2019. Vaccine. Published online March 1, 2024. doi: 10.1016/j.vaccine.2024.03.013
5Clinical overview of pneumococcal disease. Centers for Disease Control and Prevention. February 6, 2024. Accessed May 22, 2024. https://www.cdc.gov/pneumococcal/hcp/clinical-overview/
6Wasserman MD, Perdrizet J, Grant L, et al. Clinical and economic burden of pneumococcal disease due to serotypes contained in current and investigational pneumococcal conjugate vaccines in children under five years of age. Infect Dis Ther. 2021;10(4):2701-2720. doi:10.1007/s40121-021-00544-1
7Centers for Disease Control and Prevention (CDC). Visualization – Based on 1998-2022 serotype data for invasive pneumococcal disease cases by age group from Active Bacterial Core surveillance (ABCs). Updated July 22, 2024. Accessed August 30, 2024. https://data.cdc.gov/Public-Health-Surveillance/1998-2022-Serotype-Data-for-Invasive-Pneumococcal-/qvzb-qs6p/about_data
8Varghese J, Chochua S, Tran T, et al. Multistate population and whole genome sequence-based strain surveillance of invasive pneumococci recovered in the USA during 2017. Clin Microbiol Infect. 2020;26(4):512.e1-512.e10. doi:10.1016/j.cmi.2019.09.008
9Azarian T, Mitchell PK, Georgieva M, et al. Global emergence and population dynamics of divergent serotype 3 CC180 pneumococci. PLoS Pathog. 2018;14(11):e1007438. doi:10.1371/journal.ppat.1007438
10Hu T, Weiss T, Owusu-Edusei K, Petigara T. Health and economic burden associated with 15-valent pneumococcal conjugate vaccine serotypes in children in the United States. J Med Econ. 2020;23(12):1653-1660. doi:10.1080/13696998.2020.184021613
11Active Bacterial Core surveillance (ABCs) report, Emerging Infections Program network, Streptococcus pneumoniae, 2022. Centers for Disease Control and Prevention. Updated July 5, 2024. Accessed October 15, 2024. https://www.cdc.gov/abcs/downloads/SPN_Surveillance_Report_2022.pdf
12Recommended child and adolescent immunization schedule for ages 18 years or younger, United States, 2025. Centers for Disease Control and Prevention. Addendum updated November 21, 2024. Accessed November 25, 2024. https://www.cdc.gov/vaccines/hcp/imz-schedules/downloads/child/0-18yrs-child-combined-schedule.pdf
13Hill HA, et al. Decline in Vaccination Coverage by Age 24 Months and Vaccination Inequities Among Children Born in 2020 and 2021 — National Immunization Survey-Child, United States, 2021–2023. MMWR Morb Mortal Wkly Rep, pages 844–853.
14Feemster K, Weaver J, Buchwald U, Banniettis N, Cox KS, McIntosh ED, Spoulou V. Pneumococcal Vaccine Breakthrough and Failure in Infants and Children: A Narrative Review. Vaccines (Basel). 2023 Nov 24;11(12):1750. doi:10.3390/vaccines11121750. PMID: 38140155; PMCID: PMC10747311.
15Recommendations to assure the quality, safety and efficacy of pneumoccoccal conjugate vaccines. Annex 3. TRS no 977. World Health Organization. October 19, 2013. Accessed October 31, 2024. https://www.who.int/publications/m/item/pneumococcal-conjugate-vaccines-annex3-trs-977
16Guidelines on clinical evaluation of vaccines: regulatory expectations. Annex 9. TRA No 924.World Health Organization. Last reviewed October 21, 2020. Accessed October 31, 2024. https://www.who.int/publications/m/item/WHO-TRS-1004-web-annex-9
17Prevnar 13. Prescribing Information. Pfizer; 2019.
18Luck JN, Tettelin H, Orihuela CJ. Sugar-Coated Killer: Serotype 3 Pneumococcal Disease. Front Cell Infect Microbiol. 2020;10:613287. Published 2020 Dec 23. doi:10.3389/fcimb.2020.613287
19VAXNEUVANCE. Prescribing Information. Merck & Co., Inc., 2024.
20Moraes-Pinto MI, Suano-Souza F, Aranda CS. Immune system: development and acquisition of immunological competence. J Pediatr (Rio J). 2021;97(S1):S59-S66. doi:10.1016/j.jped.2020.10.006
21Wodi AP, Morelli V. Epidemiology and Prevention of Vaccine-Preventable Diseases (Pink Book). 14th edition. Chapter 1: Principles of vaccination. Centers for Disease Control and Prevention. Last reviewed March 2024. Accessed May 9, 2024. https://www.cdc.gov/pinkbook/hcp/table-of-contents/chapter-1-principles-of-vaccination.html
22Pneumococcal vaccination. Centers for Disease Control and Prevention. Last reviewed September 12, 2024. Accessed September 30, 2024. https://www.cdc.gov/pneumococcal/vaccines/index.html
23Lupinacci R, Rupp R, Wittawatmongkol O, et al. A phase 3, multicenter, randomized, double-blind, active-comparator-controlled study to evaluate the safety, tolerability, and immunogenicity of a 4-dose regimen of V114, a 15-valent pneumococcal conjugate vaccine, in healthy infants (PNEU-PED). Vaccine. 2023;41(5):1142-1152. doi:10.1016/j.vaccine.2022.12.054
24Prevnar 20. Prescribing Information. Pfizer; 2023.
Copyright © 2025 Frontline Medical Communications Inc. All rights reserved. No part of this publication may be reproduced or transmitted in any form, by any means, without prior written permission of the Publisher. Frontline Medical Communications Inc. will not assume responsibility for damages, loss, or claims of any kind arising from or related to the information contained in this publication, including any claims related to the products, drugs, or services mentioned herein. The opinions expressed in this publication do not necessarily reflect the views of the Publisher. All other trademarks are property of their respective owners.
Neither the editors of Pediatric News nor the Editorial Advisory Board nor the reporting staff contributed to this content.
US-PVC-01998 03/25
WATS-3D Biopsy Increases Detection of Barrett’s Esophagus in GERD
, new research showed.
Compared with forceps biopsies (FB) alone, the addition of WATS-3D led to confirmation of BE in an additional one fifth of patients, roughly doubled dysplasia diagnoses, and influenced clinical management in the majority of patients.
“The big take-home point here is that the use of WATS-3D brushing along with conventional biopsies increases the likelihood that intestinal metaplasia will be identified,” first author Nicholas Shaheen, MD, MPH, AGAF, with the Center for Esophageal Diseases and Swallowing, University of North Carolina School of Medicine at Chapel Hill, North Carolina, told GI & Hepatology News.
“Almost 20% of patients who harbor BE were only identified by WATS-3D and might have otherwise gone undiagnosed had only forceps biopsies been performed,” Shaheen said.
The study was published in The American Journal of Gastroenterology.
Beyond Traditional Biopsies
BE develops as a complication of chronic GERD and is the chief precursor to esophageal adenocarcinoma. Early detection of BE and dysplasia is crucial to enable timely intervention.
The current gold standard for BE screening involves upper endoscopy with FB following the Seattle protocol, which consists of four-quadrant biopsies from every 1-2 cm of areas of columnar-lined epithelium (CLE) to confirm the presence of intestinal metaplasia. However, this protocol is prone to sampling errors and high false-negative rates, leading to repeat endoscopy, the study team pointed out.
WATS-3D (CDx Diagnostics) is a complementary technique designed to improve diagnostic yield by using brush biopsy to sample more tissue than routine biopsies.
WATS-3D has been shown to increase detection of dysplasia in patients with BE undergoing surveillance for BE, but less is known about the value of WATS-3D for BE screening in a community-based cohort of patients with GERD.
To investigate, Shaheen and colleagues studied 23,933 consecutive patients enrolled in a prospective observational registry assessing the utility of WATS-3D in the screening of symptomatic GERD patients for BE.
Patients had both WATS-3D and FB in the same endoscopic session. No patient had a history of BE, intestinal metaplasia or dysplasia in esophageal mucosa, or esophageal surgery, endoscopic ablation or endoscopic mucosal resection prior to enrollment.
Overall, 6829 patients (29%) met endoscopic criteria for BE (≥ 1 cm esophageal CLE with accompanying biopsies showing intestinal metaplasia).
Of these, 2878 (42%) had intestinal metaplasia identified by either FB or WATS-3D, but 19.3% had their BE diagnosis confirmed solely on the basis of WATS-3D findings.
Among patients who fulfilled the endoscopic criteria for BE, the adjunctive yield of WATS-3D was 76.5% and the absolute yield was 18.1%.
Of the 240 (1.0%) patients with dysplasia, 107 (45%) were found solely by WATS-3D.
‘Clinically Valuable Adjunct’
Among patients with positive WATS-3D but negative FB results, clinical management changed in 90.7% of cases, mostly involving initiation or modification of surveillance and proton pump inhibitor therapy.
These results suggest that WATS-3D is a “clinically valuable adjunct” to FB for the diagnosis of BE when used as a screening tool in symptomatic GERD patients and particularly in patients with endoscopic evidence of > 1 cm esophageal columnar-lined epithelium, the study team wrote.
Adjunctive use of WATS-3D when BE is suspected “may save endoscopies and lead to quicker, more accurate diagnoses,” they added.
The investigators said a limitation of the study is the lack of central pathology review, potentially leading to diagnostic variability. They also noted that over half of the detected dysplasia cases were crypt dysplasia or indefinite for dysplasia, raising concerns about clinical significance.
Reached for comment, Philip O. Katz, MD, AGAF, professor of medicine and director of the GI Function Laboratories, Weill Cornell Medicine in New York, said he’s been using WATS for more than a decade as an adjunct to standard biopsy in patients undergoing screening and surveillance for BE and finds it clinically helpful in managing his patients.
This new study provides “further information that WATS added to biopsy that has been traditionally done with the Seattle protocol increases the yield of intestinal metaplasia and likely dysplasia in patients being screened for Barrett’s,” Katz, who wasn’t involved in the study, told GI & Hepatology News.
Funding for the study was provided by CDx Diagnostics. Shaheen and several coauthors disclosed relationships with the company. Katz disclosed relationships (consultant/advisor) for Phathom Pharmaceuticals and Sebella.
A version of this article appeared on Medscape.com.
, new research showed.
Compared with forceps biopsies (FB) alone, the addition of WATS-3D led to confirmation of BE in an additional one fifth of patients, roughly doubled dysplasia diagnoses, and influenced clinical management in the majority of patients.
“The big take-home point here is that the use of WATS-3D brushing along with conventional biopsies increases the likelihood that intestinal metaplasia will be identified,” first author Nicholas Shaheen, MD, MPH, AGAF, with the Center for Esophageal Diseases and Swallowing, University of North Carolina School of Medicine at Chapel Hill, North Carolina, told GI & Hepatology News.
“Almost 20% of patients who harbor BE were only identified by WATS-3D and might have otherwise gone undiagnosed had only forceps biopsies been performed,” Shaheen said.
The study was published in The American Journal of Gastroenterology.
Beyond Traditional Biopsies
BE develops as a complication of chronic GERD and is the chief precursor to esophageal adenocarcinoma. Early detection of BE and dysplasia is crucial to enable timely intervention.
The current gold standard for BE screening involves upper endoscopy with FB following the Seattle protocol, which consists of four-quadrant biopsies from every 1-2 cm of areas of columnar-lined epithelium (CLE) to confirm the presence of intestinal metaplasia. However, this protocol is prone to sampling errors and high false-negative rates, leading to repeat endoscopy, the study team pointed out.
WATS-3D (CDx Diagnostics) is a complementary technique designed to improve diagnostic yield by using brush biopsy to sample more tissue than routine biopsies.
WATS-3D has been shown to increase detection of dysplasia in patients with BE undergoing surveillance for BE, but less is known about the value of WATS-3D for BE screening in a community-based cohort of patients with GERD.
To investigate, Shaheen and colleagues studied 23,933 consecutive patients enrolled in a prospective observational registry assessing the utility of WATS-3D in the screening of symptomatic GERD patients for BE.
Patients had both WATS-3D and FB in the same endoscopic session. No patient had a history of BE, intestinal metaplasia or dysplasia in esophageal mucosa, or esophageal surgery, endoscopic ablation or endoscopic mucosal resection prior to enrollment.
Overall, 6829 patients (29%) met endoscopic criteria for BE (≥ 1 cm esophageal CLE with accompanying biopsies showing intestinal metaplasia).
Of these, 2878 (42%) had intestinal metaplasia identified by either FB or WATS-3D, but 19.3% had their BE diagnosis confirmed solely on the basis of WATS-3D findings.
Among patients who fulfilled the endoscopic criteria for BE, the adjunctive yield of WATS-3D was 76.5% and the absolute yield was 18.1%.
Of the 240 (1.0%) patients with dysplasia, 107 (45%) were found solely by WATS-3D.
‘Clinically Valuable Adjunct’
Among patients with positive WATS-3D but negative FB results, clinical management changed in 90.7% of cases, mostly involving initiation or modification of surveillance and proton pump inhibitor therapy.
These results suggest that WATS-3D is a “clinically valuable adjunct” to FB for the diagnosis of BE when used as a screening tool in symptomatic GERD patients and particularly in patients with endoscopic evidence of > 1 cm esophageal columnar-lined epithelium, the study team wrote.
Adjunctive use of WATS-3D when BE is suspected “may save endoscopies and lead to quicker, more accurate diagnoses,” they added.
The investigators said a limitation of the study is the lack of central pathology review, potentially leading to diagnostic variability. They also noted that over half of the detected dysplasia cases were crypt dysplasia or indefinite for dysplasia, raising concerns about clinical significance.
Reached for comment, Philip O. Katz, MD, AGAF, professor of medicine and director of the GI Function Laboratories, Weill Cornell Medicine in New York, said he’s been using WATS for more than a decade as an adjunct to standard biopsy in patients undergoing screening and surveillance for BE and finds it clinically helpful in managing his patients.
This new study provides “further information that WATS added to biopsy that has been traditionally done with the Seattle protocol increases the yield of intestinal metaplasia and likely dysplasia in patients being screened for Barrett’s,” Katz, who wasn’t involved in the study, told GI & Hepatology News.
Funding for the study was provided by CDx Diagnostics. Shaheen and several coauthors disclosed relationships with the company. Katz disclosed relationships (consultant/advisor) for Phathom Pharmaceuticals and Sebella.
A version of this article appeared on Medscape.com.
, new research showed.
Compared with forceps biopsies (FB) alone, the addition of WATS-3D led to confirmation of BE in an additional one fifth of patients, roughly doubled dysplasia diagnoses, and influenced clinical management in the majority of patients.
“The big take-home point here is that the use of WATS-3D brushing along with conventional biopsies increases the likelihood that intestinal metaplasia will be identified,” first author Nicholas Shaheen, MD, MPH, AGAF, with the Center for Esophageal Diseases and Swallowing, University of North Carolina School of Medicine at Chapel Hill, North Carolina, told GI & Hepatology News.
“Almost 20% of patients who harbor BE were only identified by WATS-3D and might have otherwise gone undiagnosed had only forceps biopsies been performed,” Shaheen said.
The study was published in The American Journal of Gastroenterology.
Beyond Traditional Biopsies
BE develops as a complication of chronic GERD and is the chief precursor to esophageal adenocarcinoma. Early detection of BE and dysplasia is crucial to enable timely intervention.
The current gold standard for BE screening involves upper endoscopy with FB following the Seattle protocol, which consists of four-quadrant biopsies from every 1-2 cm of areas of columnar-lined epithelium (CLE) to confirm the presence of intestinal metaplasia. However, this protocol is prone to sampling errors and high false-negative rates, leading to repeat endoscopy, the study team pointed out.
WATS-3D (CDx Diagnostics) is a complementary technique designed to improve diagnostic yield by using brush biopsy to sample more tissue than routine biopsies.
WATS-3D has been shown to increase detection of dysplasia in patients with BE undergoing surveillance for BE, but less is known about the value of WATS-3D for BE screening in a community-based cohort of patients with GERD.
To investigate, Shaheen and colleagues studied 23,933 consecutive patients enrolled in a prospective observational registry assessing the utility of WATS-3D in the screening of symptomatic GERD patients for BE.
Patients had both WATS-3D and FB in the same endoscopic session. No patient had a history of BE, intestinal metaplasia or dysplasia in esophageal mucosa, or esophageal surgery, endoscopic ablation or endoscopic mucosal resection prior to enrollment.
Overall, 6829 patients (29%) met endoscopic criteria for BE (≥ 1 cm esophageal CLE with accompanying biopsies showing intestinal metaplasia).
Of these, 2878 (42%) had intestinal metaplasia identified by either FB or WATS-3D, but 19.3% had their BE diagnosis confirmed solely on the basis of WATS-3D findings.
Among patients who fulfilled the endoscopic criteria for BE, the adjunctive yield of WATS-3D was 76.5% and the absolute yield was 18.1%.
Of the 240 (1.0%) patients with dysplasia, 107 (45%) were found solely by WATS-3D.
‘Clinically Valuable Adjunct’
Among patients with positive WATS-3D but negative FB results, clinical management changed in 90.7% of cases, mostly involving initiation or modification of surveillance and proton pump inhibitor therapy.
These results suggest that WATS-3D is a “clinically valuable adjunct” to FB for the diagnosis of BE when used as a screening tool in symptomatic GERD patients and particularly in patients with endoscopic evidence of > 1 cm esophageal columnar-lined epithelium, the study team wrote.
Adjunctive use of WATS-3D when BE is suspected “may save endoscopies and lead to quicker, more accurate diagnoses,” they added.
The investigators said a limitation of the study is the lack of central pathology review, potentially leading to diagnostic variability. They also noted that over half of the detected dysplasia cases were crypt dysplasia or indefinite for dysplasia, raising concerns about clinical significance.
Reached for comment, Philip O. Katz, MD, AGAF, professor of medicine and director of the GI Function Laboratories, Weill Cornell Medicine in New York, said he’s been using WATS for more than a decade as an adjunct to standard biopsy in patients undergoing screening and surveillance for BE and finds it clinically helpful in managing his patients.
This new study provides “further information that WATS added to biopsy that has been traditionally done with the Seattle protocol increases the yield of intestinal metaplasia and likely dysplasia in patients being screened for Barrett’s,” Katz, who wasn’t involved in the study, told GI & Hepatology News.
Funding for the study was provided by CDx Diagnostics. Shaheen and several coauthors disclosed relationships with the company. Katz disclosed relationships (consultant/advisor) for Phathom Pharmaceuticals and Sebella.
A version of this article appeared on Medscape.com.
Intensive Nutrition Therapy Improves Outcomes in Alcohol-Related ACLF
In a randomized controlled trial, compared with standard care, dietitian-supported, intensive nutritional therapy improved survival, reduced frailty, and lowered hospitalization rates in men with alcohol-related ACLF.
The study, performed by a team from the Postgraduate Institute of Medical Education and Research, Chandigarh, India, was published in Clinical Gastroenterology and Hepatology.
ACLF related to alcohol use is associated with poor outcomes due to poor nutritional intake and frailty. Frail patients with ACLF face higher morbidity, mortality, and hospitalization rates than their nonfrail counterparts. However, research on the role of structured nutritional interventions in improving these outcomes is limited.
Patal Giri, MBBS, MD, and colleagues enrolled 70 men with alcohol-related ACLF and frailty (liver frailty index [LFI] > 4.5) in a single-center, open-label study. Half were randomly allocated to an intervention group receiving outpatient intensive nutrition therapy (OINT) plus standard medical treatment (SMT) and half to a control group receiving SMT alone for 3 months.
The intervention group received a monitored high-calorie, high-protein, and salt-restricted diet as prescribed by a dedicated senior liver dietitian. The control group received regular nutritional recommendations and were managed for the ACLF-associated complications, without intervention or guidance by the study team.
After 3 months follow-up, overall survival (the primary outcome) was significantly improved in the OINT group compared with the control group (91.4% vs 57.1%), “suggesting that the improvement in nutrition status is associated with better survival,” the study team noted. Three patients died in the OINT group vs 15 in the SMT group.
OINT also led to a significant improvement in frailty, with LFI scores decreasing by an average of 0.93 in the intervention group vs 0.33 in the control group; 97% of patients improved from frail to prefrail status in the OINT group, whereas only 20% of patients improved in the SMT group.
The mean change in LFI of 0.93 with OINT is “well above the substantially clinically important difference” (change of 0.8) established in a previous study, the authors noted.
Significant improvements in weight and body mass index were also observed in the OINT group relative to the control group.
Liver disease severity, including model for end-stage liver disease (MELD) scores, showed greater improvement in the OINT group than in the control group (−8.7 vs −6.3 points from baseline to 3 months).
During the follow-up period, fewer patients in the intervention group than in the control group required a hospital stay (17% vs 45.7%).
Limitations of the study include the single-center design and the short follow-up period of 3 months, which limits long-term outcome assessment. Further, the study only included patients meeting Asia Pacific Association for Study of Liver criteria for ACLF, which does not include the patients with organ failure as defined by European Association for the Study of the Liver-Chronic Liver Failure Consortium criteria. Patients with ACLF who had more severe disease (MELD score > 30 or AARC > 10) were also not included.
Despite these limitations, the authors said their study showed that “dietician-monitored goal-directed nutrition therapy is very important in the management of patients with alcohol-related ACLF along with SMT.”
Confirmatory Data
Reached for comment, Katherine Patton, MEd, RD, a registered dietitian with the Center for Human Nutrition at Cleveland Clinic, Cleveland, Ohio, said it’s well known that the ACLF patient population has a “very high rate of morbidity and mortality and their quality of life tends to be poor due to their frailty. It is also fairly well-known that proper nutrition therapy can improve outcomes, however barriers to adequate nutrition include decreased appetite, nausea, pain, altered taste, and early satiety from ascites.”
“Hepatologists are likely stressing the importance of adequate protein energy intake and doctors may refer patients to an outpatient dietitian, but it is up to the patient to make that appointment and act on the recommendations,” Patton told GI & Hepatology News.
“If a dietitian works in the same clinic as the hepatologist and patients can be referred and seen the same day, this is ideal. During a hospital admission, protein/calorie intake can be more closely monitored and encouraged by a multi-disciplinary team,” Patton explained.
She cautioned that “the average patient is not familiar with how to apply general calorie and protein goals to their everyday eating habits. This study amplifies the role of a dietitian and what consistent education and resources can do to improve a patient’s quality of life and survival.”
This study had no specific funding. The authors have declared no relevant conflicts of interest. Patton had no relevant disclosures.
A version of this article appeared on Medscape.com.
In a randomized controlled trial, compared with standard care, dietitian-supported, intensive nutritional therapy improved survival, reduced frailty, and lowered hospitalization rates in men with alcohol-related ACLF.
The study, performed by a team from the Postgraduate Institute of Medical Education and Research, Chandigarh, India, was published in Clinical Gastroenterology and Hepatology.
ACLF related to alcohol use is associated with poor outcomes due to poor nutritional intake and frailty. Frail patients with ACLF face higher morbidity, mortality, and hospitalization rates than their nonfrail counterparts. However, research on the role of structured nutritional interventions in improving these outcomes is limited.
Patal Giri, MBBS, MD, and colleagues enrolled 70 men with alcohol-related ACLF and frailty (liver frailty index [LFI] > 4.5) in a single-center, open-label study. Half were randomly allocated to an intervention group receiving outpatient intensive nutrition therapy (OINT) plus standard medical treatment (SMT) and half to a control group receiving SMT alone for 3 months.
The intervention group received a monitored high-calorie, high-protein, and salt-restricted diet as prescribed by a dedicated senior liver dietitian. The control group received regular nutritional recommendations and were managed for the ACLF-associated complications, without intervention or guidance by the study team.
After 3 months follow-up, overall survival (the primary outcome) was significantly improved in the OINT group compared with the control group (91.4% vs 57.1%), “suggesting that the improvement in nutrition status is associated with better survival,” the study team noted. Three patients died in the OINT group vs 15 in the SMT group.
OINT also led to a significant improvement in frailty, with LFI scores decreasing by an average of 0.93 in the intervention group vs 0.33 in the control group; 97% of patients improved from frail to prefrail status in the OINT group, whereas only 20% of patients improved in the SMT group.
The mean change in LFI of 0.93 with OINT is “well above the substantially clinically important difference” (change of 0.8) established in a previous study, the authors noted.
Significant improvements in weight and body mass index were also observed in the OINT group relative to the control group.
Liver disease severity, including model for end-stage liver disease (MELD) scores, showed greater improvement in the OINT group than in the control group (−8.7 vs −6.3 points from baseline to 3 months).
During the follow-up period, fewer patients in the intervention group than in the control group required a hospital stay (17% vs 45.7%).
Limitations of the study include the single-center design and the short follow-up period of 3 months, which limits long-term outcome assessment. Further, the study only included patients meeting Asia Pacific Association for Study of Liver criteria for ACLF, which does not include the patients with organ failure as defined by European Association for the Study of the Liver-Chronic Liver Failure Consortium criteria. Patients with ACLF who had more severe disease (MELD score > 30 or AARC > 10) were also not included.
Despite these limitations, the authors said their study showed that “dietician-monitored goal-directed nutrition therapy is very important in the management of patients with alcohol-related ACLF along with SMT.”
Confirmatory Data
Reached for comment, Katherine Patton, MEd, RD, a registered dietitian with the Center for Human Nutrition at Cleveland Clinic, Cleveland, Ohio, said it’s well known that the ACLF patient population has a “very high rate of morbidity and mortality and their quality of life tends to be poor due to their frailty. It is also fairly well-known that proper nutrition therapy can improve outcomes, however barriers to adequate nutrition include decreased appetite, nausea, pain, altered taste, and early satiety from ascites.”
“Hepatologists are likely stressing the importance of adequate protein energy intake and doctors may refer patients to an outpatient dietitian, but it is up to the patient to make that appointment and act on the recommendations,” Patton told GI & Hepatology News.
“If a dietitian works in the same clinic as the hepatologist and patients can be referred and seen the same day, this is ideal. During a hospital admission, protein/calorie intake can be more closely monitored and encouraged by a multi-disciplinary team,” Patton explained.
She cautioned that “the average patient is not familiar with how to apply general calorie and protein goals to their everyday eating habits. This study amplifies the role of a dietitian and what consistent education and resources can do to improve a patient’s quality of life and survival.”
This study had no specific funding. The authors have declared no relevant conflicts of interest. Patton had no relevant disclosures.
A version of this article appeared on Medscape.com.
In a randomized controlled trial, compared with standard care, dietitian-supported, intensive nutritional therapy improved survival, reduced frailty, and lowered hospitalization rates in men with alcohol-related ACLF.
The study, performed by a team from the Postgraduate Institute of Medical Education and Research, Chandigarh, India, was published in Clinical Gastroenterology and Hepatology.
ACLF related to alcohol use is associated with poor outcomes due to poor nutritional intake and frailty. Frail patients with ACLF face higher morbidity, mortality, and hospitalization rates than their nonfrail counterparts. However, research on the role of structured nutritional interventions in improving these outcomes is limited.
Patal Giri, MBBS, MD, and colleagues enrolled 70 men with alcohol-related ACLF and frailty (liver frailty index [LFI] > 4.5) in a single-center, open-label study. Half were randomly allocated to an intervention group receiving outpatient intensive nutrition therapy (OINT) plus standard medical treatment (SMT) and half to a control group receiving SMT alone for 3 months.
The intervention group received a monitored high-calorie, high-protein, and salt-restricted diet as prescribed by a dedicated senior liver dietitian. The control group received regular nutritional recommendations and were managed for the ACLF-associated complications, without intervention or guidance by the study team.
After 3 months follow-up, overall survival (the primary outcome) was significantly improved in the OINT group compared with the control group (91.4% vs 57.1%), “suggesting that the improvement in nutrition status is associated with better survival,” the study team noted. Three patients died in the OINT group vs 15 in the SMT group.
OINT also led to a significant improvement in frailty, with LFI scores decreasing by an average of 0.93 in the intervention group vs 0.33 in the control group; 97% of patients improved from frail to prefrail status in the OINT group, whereas only 20% of patients improved in the SMT group.
The mean change in LFI of 0.93 with OINT is “well above the substantially clinically important difference” (change of 0.8) established in a previous study, the authors noted.
Significant improvements in weight and body mass index were also observed in the OINT group relative to the control group.
Liver disease severity, including model for end-stage liver disease (MELD) scores, showed greater improvement in the OINT group than in the control group (−8.7 vs −6.3 points from baseline to 3 months).
During the follow-up period, fewer patients in the intervention group than in the control group required a hospital stay (17% vs 45.7%).
Limitations of the study include the single-center design and the short follow-up period of 3 months, which limits long-term outcome assessment. Further, the study only included patients meeting Asia Pacific Association for Study of Liver criteria for ACLF, which does not include the patients with organ failure as defined by European Association for the Study of the Liver-Chronic Liver Failure Consortium criteria. Patients with ACLF who had more severe disease (MELD score > 30 or AARC > 10) were also not included.
Despite these limitations, the authors said their study showed that “dietician-monitored goal-directed nutrition therapy is very important in the management of patients with alcohol-related ACLF along with SMT.”
Confirmatory Data
Reached for comment, Katherine Patton, MEd, RD, a registered dietitian with the Center for Human Nutrition at Cleveland Clinic, Cleveland, Ohio, said it’s well known that the ACLF patient population has a “very high rate of morbidity and mortality and their quality of life tends to be poor due to their frailty. It is also fairly well-known that proper nutrition therapy can improve outcomes, however barriers to adequate nutrition include decreased appetite, nausea, pain, altered taste, and early satiety from ascites.”
“Hepatologists are likely stressing the importance of adequate protein energy intake and doctors may refer patients to an outpatient dietitian, but it is up to the patient to make that appointment and act on the recommendations,” Patton told GI & Hepatology News.
“If a dietitian works in the same clinic as the hepatologist and patients can be referred and seen the same day, this is ideal. During a hospital admission, protein/calorie intake can be more closely monitored and encouraged by a multi-disciplinary team,” Patton explained.
She cautioned that “the average patient is not familiar with how to apply general calorie and protein goals to their everyday eating habits. This study amplifies the role of a dietitian and what consistent education and resources can do to improve a patient’s quality of life and survival.”
This study had no specific funding. The authors have declared no relevant conflicts of interest. Patton had no relevant disclosures.
A version of this article appeared on Medscape.com.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY