User login
Streamlined Testosterone Order Template to Improve the Diagnosis and Evaluation of Hypogonadism in Veterans
Streamlined Testosterone Order Template to Improve the Diagnosis and Evaluation of Hypogonadism in Veterans
Testosterone therapy is administered following pragmatic diagnostic evaluation and workup to assess whether an adult male is hypogonadal, based on symptoms consistent with androgen deficiency and low morning serum testosterone concentrations on ≥ 2 occasions. Effects of testosterone administration include the development or maintenance of secondary sexual characteristics and increases in libido, muscle strength, fat-free mass, and bone density.
Testosterone prescriptions have markedly increased in the past 20 years, including within the US Department of Veterans Affairs (VA) health care system.1-3 This trend may be influenced by various factors, including patient perceptions of benefit, an increase in marketing, and the availability of more user-friendly formulations.
Since 2006, evidence-based clinical practice guidelines have recommended specific clinical and laboratory evaluation and counseling prior to starting testosterone replacement therapy (TRT).4-8 However, research has shown poor adherence to these recommendations, including at the VA, which raises concerns about inappropriate TRT initiation without proper diagnostic evaluation.9,10 Observational research has suggested a possible link between testosterone therapy and increased risk of cardiovascular (CV) events. The US Food and Drug Administration prescribing information includes boxed warnings about potential risks of high blood pressure, myocardial infarction, stroke, and CV-related mortality with testosterone treatment, contact transfer of transdermal testosterone, and pulmonary oil microembolism with testosterone undecanoate injections.11-15
A VA Office of Inspector General (OIG) review of VA clinician adherence to clinical and laboratory evaluation guidelines for testosterone deficiency found poor adherence among VA practitioners and made recommendations for improvement.4,15 These focused on establishing clinical signs and symptoms consistent with testosterone deficiency, confirming hypogonadism by repeated testosterone testing, determining the etiology of hypogonadism by measuring gonadotropins, initiating a discussion of risks and benefits of TRT, and assessing clinical improvement and obtaining an updated hematocrit test within 3 to 6 months of initiation.
The VA Puget Sound Health Care System (VAPSHCS) developed a local prior authorization template to assist health care practitioners (HCPs) to address the OIG recommendations. This testosterone order template (TOT) aimed to improve the diagnosis, evaluation, and monitoring of TRT in males with hypogonadism, combined with existing VA pharmacy criteria for the use of testosterone based on Endocrine Society guidelines. A version of the VAPSHCS TOT was approved as the national VA Computerized Patient Record System (CPRS) template.
Preliminary evaluation of the TOT suggested improved short-term adherence to guideline recommendations following implementation.16 This quality improvement study sought to assess the long-term effectiveness of the TOT with respect to clinical practice guideline adherence. The OIG did not address prostate-specific antigen (PSA) monitoring because understanding of the relationship between TRT and the risks of elevated PSA levels remains incomplete.6,17 This project hypothesized that implementation of a pharmacy-managed TOT incorporated into CPRS would result in higher adherence rates to guideline-recommended clinical and laboratory evaluation, in addition to counseling of men with hypogonadism prior to initiation of TRT.
Methods
Eligible participants were cisgender males who received a new testosterone prescription, had ≥ 2 clinic visits at VAPSHCS, and no previous testosterone prescription in the previous 2 years. Individuals were excluded if they had testosterone administered at VAPSHCS; were prescribed testosterone at another facility (VA or community-based); pilot tested an initial version of the TOT prior to November 30, 2019; or had an International Classification of Diseases, Tenth Revision codes for hypopituitarism, gender identity disorder, history of sexual assignment, or Klinefelter syndrome for which testosterone therapy was already approved. Patients who met the inclusion criteria were identified by an algorithm developed by the VAPSHCS pharmacoeconomist.
This quality improvement project used a retrospective, pre-post experimental design. Electronic chart review and systematic manual review of all eligible patient charts were performed for the pretemplate period (December 1, 2018, to November 30, 2019) and after the template implementation, (December 1, 2021, to November 30, 2022).
An initial version of the TOT was implemented on July 1, 2019, but was not fully integrated into CPRS until early 2020; individuals in whom the TOT was used prior to November 30, 2019, were excluded. Data from the initial period of the COVID-19 pandemic were avoided because of alterations in clinic and prescribing practices. As a quality improvement project, the TOT evaluation was exempt from formal review by the VAPSHCS Institutional Review Board, as determined by the Director of the Office of Transformation/Quality/Safety/Value.
Interventions
Testosterone is a Schedule III controlled substance with potential risks and a propensity for varied prescribing practices. It was designated as a restricted drug requiring a prior authorization drug request (PADR) for which a specific TOT was developed, approved by the VAPSHCS Pharmacy and Therapeutics Committee, and incorporated into CPRS. A team of pharmacists, primary care physicians, geriatricians, endocrinologists, and health informatics experts created and developed the TOT. Pharmacists managed and monitored its completion.
The process for prescribing testosterone via the TOT is outlined in the eAppendix. When an HCP orders testosterone in CPRS, reminders prompt them to use the TOT and indicate required laboratory measurements (an order set is provided). Completion of TOT is not necessary to order testosterone for patients with an existing diagnosis of an organic cause of hypogonadism (eg, Klinefelter syndrome or hypopituitarism) or transgender women (assigned male at birth). In the TOT, the prescriber must also indicate signs and symptoms of testosterone deficiency; required laboratory tests; and counseling regarding potential risks and benefits of TRT. A pharmacist reviews the TOT and either approves or rejects the testosterone prescription and provides follow-up guidance to the prescriber. The completed TOT serves as documentation of guideline adherence in CPRS. The TOT also includes sections for first renewal testosterone prescriptions, addressing guideline recommendations for follow-up laboratory evaluation and clinical response to TRT. Due to limited completion of this section in the posttemplate period, evaluating adherence to follow-up recommendations was not feasible.
Measures
This project assessed the percentage of patients in the posttemplate period vs pretemplate period with an approved PADR. Documentation of specific guideline-recommended measures was assessed: signs and symptoms of testosterone deficiency; ≥ 2 serum testosterone measurements (≥ 2 total, free and total, or 2 free testosterone levels, and ≥ 1 testosterone level before 10
The project also assessed the proportion of patients in the posttemplate period vs pretemplate period who had all hormone tests (≥ 2 serum testosterone and LH and FSH concentrations), all laboratory tests (hormone tests and hematocrit), and all 5 guideline-recommended measures.
Analysis
Statistical comparisons between the proportions of patients in the pretemplate and posttemplate periods for each measure were performed using a χ2 test, without correction for multiple comparisons. All analyses were conducted using Stata version 10.0. A P value < .05 was considered significant for all comparisons.
Results
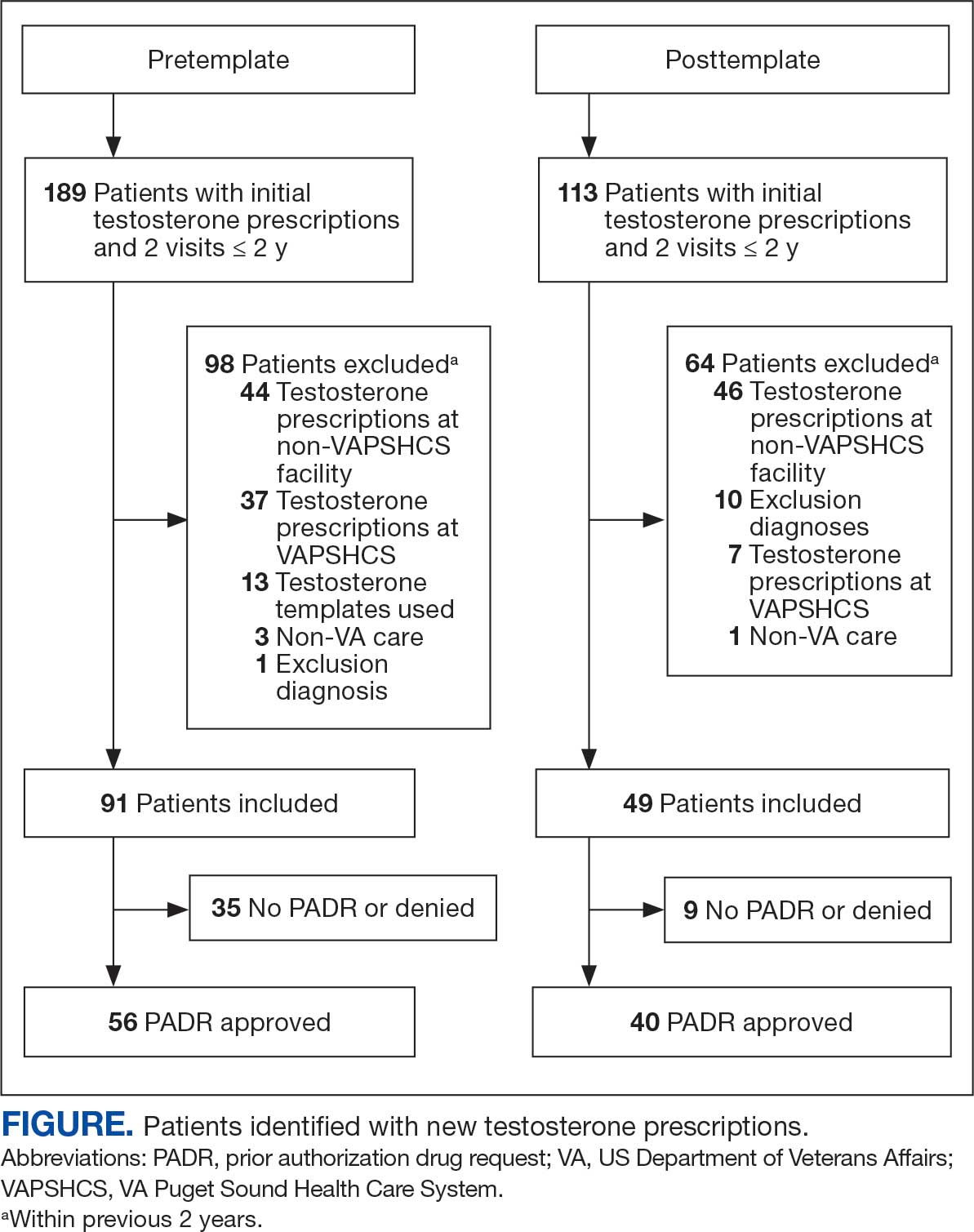
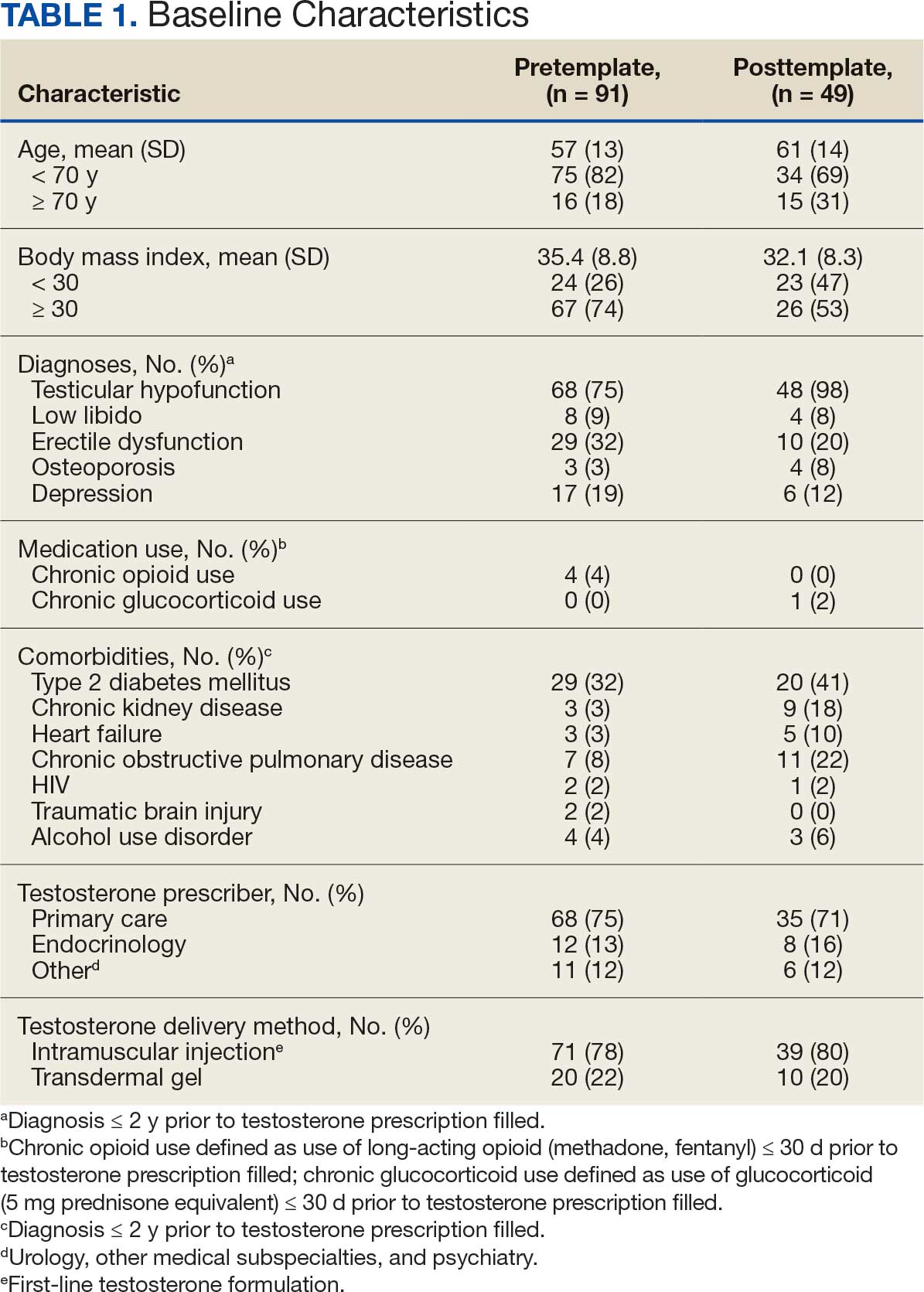
Chart review identified 189 patients in the pretemplate period and 113 patients in the posttemplate period with a new testosterone prescription (Figure). After exclusions, 91 and 49 patients, respectively, met eligibility criteria (Table 1). Fifty-six patients (62%) pretemplate and 40 patients (82%) posttemplate (P = .015) had approved PADRs and comprised the groups that were analyzed (Table 2).

The mean age and body mass index were similar in the pretemplate and posttemplate periods, but there was variation in the proportions of patients aged < 70 years and those with a body mass index < 30 between the groups. The most common diagnosis in both groups was testicular hypofunction, and the most common comorbidity was type 2 diabetes mellitus. Concomitant use of opioids or glucocorticoids that can lower testosterone levels was rare. Most testosterone prescriptions originated from primary care clinics in both periods: 68 (75%) in the pretemplate period and 35 (71%) in the posttemplate period. Most testosterone treatment was delivered by intramuscular injection.
In the posttemplate period vs pretemplate period, the proportion of patients with an approved PADR (82% vs 62%, P = .02), and documentation of signs and symptoms of hypogonadism (93% vs 71%, P = .002) prior to starting TRT were higher, while the percentage of patients having ≥ 2 testosterone measurements (85% vs 89%, P = .53), ≥ 1 testosterone level before 10 AM (78% vs 75%, P = .70), and hematocrit measured (95% vs 91%, P = .47) were similar. Rates of LH and FSH testing were higher in the posttemplate period (80%) vs the pretemplate period (63%) but did not achieve statistical significance (P = .07), and discussion of the risks and benefits of TRT was higher in the posttemplate period (58%) vs the pretemplate period (34%) (P = .02). The percentage of patients who had all hormone measurements (total and/or free testosterone, LH, and FSH) was higher in the posttemplate period (78%) vs the pretemplate period (59%) but did not achieve statistical significance (P = .06). The rates of all guideline-recommended laboratory test orders were higher in the posttemplate period (78%) vs the pretemplate period (55%) (P = .03), and all 5 guideline-recommended clinical and laboratory measures were higher in the posttemplate period (45%) vs the pretemplate period (18%) (P = .004).
Discussion
The implementation of a pharmacy-managed TOT in CPRS demonstrated higher adherence to evidence-based guidelines for diagnosing and evaluating hypogonadism before TRT. After TOT implementation, a higher proportion of patients had documented signs and symptoms of testosterone deficiency, underwent all recommended laboratory tests, and had discussions about the risks and benefits of TRT. Adherence to 5 clinical and laboratory measures recommended by Endocrine Society guidelines was higher after TOT implementation, indicating improved prescribing practices.4
The requirement for TOT completion before testosterone prescription and its management by trained pharmacists likely contributed to higher adherence to guideline recommendations than previously reported. Integration of the TOT into CPRS with pharmacy oversight may have enhanced adherence by summarizing and codifying evidence-based guideline recommendations for clinical and biochemical evaluation prior to TRT initiation, offering relevant education to clinicians and pharmacists, automatically importing pertinent clinical information and laboratory results, and generating CPRS documentation to reduce clinician burden during patient care.
The proportion of patients with documented signs and symptoms of testosterone deficiency before TRT increased from the pretemplate period (71%) to the posttemplate period (93%), indicating that most patients receiving TRT had clinical manifestations of hypogonadism. This aligns with Endocrine Society guidelines, which define hypogonadism as a clinical disorder characterized by clinical manifestations of testosterone deficiency and persistently low serum testosterone levels on ≥ 2 separate occasions.4,6 However, recent trends in direct-to-consumer advertising for testosterone and the rise of “low T” clinics may contribute to increased testing, varied practices, and inappropriate testosterone therapy initiation (eg, in men with low testosterone levels who lack symptoms of hypogonadism).18 Improved adherence in documenting clinical hypogonadism with implementation of the TOT reinforces the value of incorporating educational material, as previously reported.11
Adherence to guideline recommendations following implementation of the TOT in this project was higher than those previously reported. In a study of 111,631 outpatient veterans prescribed testosterone from 2009 to 2012, only 18.3% had ≥ 2 testosterone prescriptions, and 3.5% had ≥ 2 testosterone, LH, and FSH levels measured prior to the initiation of a TRT.9 In a report of 63,534 insured patients who received TRT from 2010 to 2012, 40.3% had ≥ 2 testosterone prescriptions, and 12% had LH and/or FSH measured prior to the initiation.8
Low rates of guideline-recommended laboratory tests prior to initiation of testosterone treatment were reported in prior non-VA studies.19,20 Poor guideline adherence reinforces the need for clinician education or other methods to improve TRT and ensure appropriate prescribing practices across health care systems. The TOT described in this project is a sustainable clinical tool with the potential to improve testosterone prescribing practices.
The high rates of adherence to guideline recommendations at VAPSHCS likely stem from local endocrine expertise and ongoing educational initiatives, as well as the requirement for template completion before testosterone prescription. However, most testosterone prescriptions were initiated by primary care and monitored by pharmacists with varying degrees of training and clinical experience in hypogonadism and TRT.
However, adherence to guideline recommendations was modest, suggesting there is still an opportunity for improvement. The decision to initiate therapy should be made only after appropriate counseling with patients regarding its potential benefits and risks. Reports on the CV risk of TRT have been mixed. The 2023 TRAVERSE study found no increase in major adverse CV events among older men with hypogonadism and pre-existing CV risks undergoing TRT, but noted higher instances of pulmonary embolism, atrial fibrillation, and acute kidney injury.21 This highlights the need for clinicians to continue to engage in informed decision-making with patients. Effective pretreatment counseling is important but time-consuming; future TOT monitoring and modifications could consider mandatory checkboxes to document counseling on TRT risks and benefits.
The TOT described in this study could be adapted and incorporated into the prescribing process and electronic health record of larger health care systems. Use of an electronic template allows for automatic real-time dashboard monitoring of organization performance. The TOT described could be modified or simplified for specialty or primary care clinics or individual practitioners to improve adherence to evidence-based guideline recommendations and quality of care.
Strengths
A strength of this study is the multidisciplinary team (composed of stakeholders with experience in VA health care system and subject matter experts in hypogonadism) that developed and incorporated a user-friendly template for testosterone prescriptions; the use of evidence-based guideline recommendations; and the use of a structured chart review permitted accurate assessment of adherence to recommendations to document signs and symptoms of testosterone deficiency and a discussion of potential risks and benefits prior to TRT. To our knowledge, these recommendations have not been assessed in previous reports.
Limitations
The retrospective pre-post design of this study precludes a conclusion that implementation of the TOT caused the increase in adherence to guideline recommendations. Improved adherence could have resulted from the ongoing development of the preauthorization process for testosterone prescriptions or other changes over time. However, the preauthorization process had already been established for many years prior to template implementation. Forty-nine patients had new prescriptions for testosterone in the posttemplate period compared to 91 in the pretemplate period, but TRT was initiated in accordance with guideline recommendations more appropriately in the posttemplate period. The study’s sample size was small, and many eligible patients were excluded; however, exclusions were necessary to evaluate men who had new testosterone prescriptions for which the template was designed. Most men excluded were already taking testosterone.
Conclusions
The implementation of a CPRS-based TOT improved adherence to evidence-based guidelines for the diagnosis, evaluation, and counseling of patients with hypogonadism before starting TRT. While there were improvements in adherence with the TOT, the relatively low proportion of patients with documentation of TRT risks and benefits and all guideline recommendations highlights the need for additional efforts to further strengthen adherence to guideline recommendations and ensure appropriate evaluation, counseling, and prescribing practices before initiating TRT.
- Layton JB, Li D, Meier CR, et al. Testosterone lab testing and initiation in the United Kingdom and the United States, 2000 to 2011. J Clin Endocrinol Metab. 2014;99:835-842. doi:10.1210/jc.2013-3570
- Baillargeon J, Kuo YF, Westra JR, et al. Testosterone prescribing in the United States, 2002-2016. JAMA. 2018;320:200-202. doi:10.1001/jama.2018.7999
- Jasuja GK, Bhasin S, Rose AJ. Patterns of testosterone prescription overuse. Curr Opin Endocrinol Diabetes Obes. 2017;24:240-245. doi:10.1097/MED.0000000000000336
- Bhasin S, Cunningham GR, Hayes FJ, et al. Testosterone therapy in adult men with androgen deficiency syndromes: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2006;91:1995-2010. doi:10.1210/jc.2005-2847
- Bhasin S, Cunningham GR, Hayes FJ, et al. Testosterone therapy in men with androgen deficiency syndromes: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2010;95:2536-2559. doi:10.1210/jc.2009-2354
- Bhasin S, Brito JP, Cunningham GR, et al. Testosterone therapy in men with hypogonadism: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2018;103:1715-1744. doi:10.1210/jc.2018-00229
- Mulhall JP, Trost LW, Brannigan RE, et al. Evaluation and management of testosterone deficiency: AUA guideline. J Urol. 2018;200:423-432. doi:10.1016/j.juro.2018.03.115
- Muram D, Zhang X, Cui Z, et al. Use of hormone testing for the diagnosis and evaluation of male hypogonadism and monitoring of testosterone therapy: application of hormone testing guideline recommendations in clinical practice. J Sex Med. 2015;12:1886-1894. doi:10.1111/jsm.12968
- Jasuja GK, Bhasin S, Reisman JI, et al. Ascertainment of testosterone prescribing practices in the VA. Med Care. 2015;53:746-752. doi:10.1097/MLR.0000000000000398?
- Jasuja GK, Bhasin S, Reisman JI, et al. Who gets testosterone? Patient characteristics associated with testosterone prescribing in the Veteran Affairs system: a cross-sectional study. J Gen Intern Med. 2017;32:304-311. doi:10.1007/s11606-016-3940-7
- Basaria S, Coviello AD, Travison TG, et al. Adverse events associated with testosterone administration. N Engl J Med. 2010;363:109-122. doi:10.1056/NEJMoa1000485
- Vigen R, O’Donnell CI, Barón AE, et al. Association of testosterone therapy with mortality, myocardial infarction, and stroke in men with low testosterone levels. JAMA. 2013;310:1829-1836. doi:10.1001/jama.2013.280386
- Finkle WD, Greenland S, Ridgeway GK, et al. Increased risk of non-fatal myocardial infarction following testosterone therapy prescription in men. PLoS One. 2014;9:e85805. doi:10.1371/journal.pone.0085805
- US Food and Drug Administration. FDA Drug Safety Communication: FDA cautions about using testosterone products for low testosterone due to aging; requires labeling change to inform of possible increased risk of heart attack and stroke with use. FDA.gov. March 3, 2015. Updated February 28, 2025. Accessed July 8, 2025. http://www.fda.gov/Drugs/DrugSafety/ucm436259.htm
- US Dept of Veterans Affairs, Office of Inspector General. Healthcare inspection – testosterone replacement therapy initiation and follow-up evaluation in VA male patients. April 11, 2018. Accessed July 8, 2025. https://www.vaoig.gov/reports/national-healthcare-review/healthcare-inspection-testosterone-replacement-therapy
- Narla R, Mobley D, Nguyen EHK, et al. Preliminary evaluation of an order template to improve diagnosis and testosterone therapy of hypogonadism in veterans. Fed Pract. 2021;38:121-127. doi:10.12788/fp.0103
- Bhasin S, Travison TG, Pencina KM, et al. Prostate safety events during testosterone replacement therapy in men with hypogonadism: a randomized clinical trial. JAMA Netw Open. 2023;6:e2348692. doi:10.1001/jamanetworkopen.2023.48692
- Dubin JM, Jesse E, Fantus RJ, et al. Guideline-discordant care among direct-to-consumer testosterone therapy platforms. JAMA Intern Med. 2022;182:1321-1323. doi:10.1001/jamainternmed.2022.4928
- Baillargeon J, Urban RJ, Ottenbacher KJ, et al. Trends in androgen prescribing in the United States, 2001 to 2011. JAMA Intern Med. 2013;173:1465-1466. doi:10.1001/jamainternmed.2013.6895
- Locke JA, Flannigan R, Günther OP, et al. Testosterone therapy: prescribing and monitoring patterns of practice in British Columbia. Can Urol Assoc J. 2021;15:e110-e117. doi:10.5489/cuaj.6586
- Lincoff AM, Bhasin S, Flevaris P, et al. Cardiovascular safety of testosterone-replacement therapy. N Engl J Med. 2023;389:107-117. doi:10.1056/NEJMoa2215025
Testosterone therapy is administered following pragmatic diagnostic evaluation and workup to assess whether an adult male is hypogonadal, based on symptoms consistent with androgen deficiency and low morning serum testosterone concentrations on ≥ 2 occasions. Effects of testosterone administration include the development or maintenance of secondary sexual characteristics and increases in libido, muscle strength, fat-free mass, and bone density.
Testosterone prescriptions have markedly increased in the past 20 years, including within the US Department of Veterans Affairs (VA) health care system.1-3 This trend may be influenced by various factors, including patient perceptions of benefit, an increase in marketing, and the availability of more user-friendly formulations.
Since 2006, evidence-based clinical practice guidelines have recommended specific clinical and laboratory evaluation and counseling prior to starting testosterone replacement therapy (TRT).4-8 However, research has shown poor adherence to these recommendations, including at the VA, which raises concerns about inappropriate TRT initiation without proper diagnostic evaluation.9,10 Observational research has suggested a possible link between testosterone therapy and increased risk of cardiovascular (CV) events. The US Food and Drug Administration prescribing information includes boxed warnings about potential risks of high blood pressure, myocardial infarction, stroke, and CV-related mortality with testosterone treatment, contact transfer of transdermal testosterone, and pulmonary oil microembolism with testosterone undecanoate injections.11-15
A VA Office of Inspector General (OIG) review of VA clinician adherence to clinical and laboratory evaluation guidelines for testosterone deficiency found poor adherence among VA practitioners and made recommendations for improvement.4,15 These focused on establishing clinical signs and symptoms consistent with testosterone deficiency, confirming hypogonadism by repeated testosterone testing, determining the etiology of hypogonadism by measuring gonadotropins, initiating a discussion of risks and benefits of TRT, and assessing clinical improvement and obtaining an updated hematocrit test within 3 to 6 months of initiation.
The VA Puget Sound Health Care System (VAPSHCS) developed a local prior authorization template to assist health care practitioners (HCPs) to address the OIG recommendations. This testosterone order template (TOT) aimed to improve the diagnosis, evaluation, and monitoring of TRT in males with hypogonadism, combined with existing VA pharmacy criteria for the use of testosterone based on Endocrine Society guidelines. A version of the VAPSHCS TOT was approved as the national VA Computerized Patient Record System (CPRS) template.
Preliminary evaluation of the TOT suggested improved short-term adherence to guideline recommendations following implementation.16 This quality improvement study sought to assess the long-term effectiveness of the TOT with respect to clinical practice guideline adherence. The OIG did not address prostate-specific antigen (PSA) monitoring because understanding of the relationship between TRT and the risks of elevated PSA levels remains incomplete.6,17 This project hypothesized that implementation of a pharmacy-managed TOT incorporated into CPRS would result in higher adherence rates to guideline-recommended clinical and laboratory evaluation, in addition to counseling of men with hypogonadism prior to initiation of TRT.
Methods
Eligible participants were cisgender males who received a new testosterone prescription, had ≥ 2 clinic visits at VAPSHCS, and no previous testosterone prescription in the previous 2 years. Individuals were excluded if they had testosterone administered at VAPSHCS; were prescribed testosterone at another facility (VA or community-based); pilot tested an initial version of the TOT prior to November 30, 2019; or had an International Classification of Diseases, Tenth Revision codes for hypopituitarism, gender identity disorder, history of sexual assignment, or Klinefelter syndrome for which testosterone therapy was already approved. Patients who met the inclusion criteria were identified by an algorithm developed by the VAPSHCS pharmacoeconomist.
This quality improvement project used a retrospective, pre-post experimental design. Electronic chart review and systematic manual review of all eligible patient charts were performed for the pretemplate period (December 1, 2018, to November 30, 2019) and after the template implementation, (December 1, 2021, to November 30, 2022).
An initial version of the TOT was implemented on July 1, 2019, but was not fully integrated into CPRS until early 2020; individuals in whom the TOT was used prior to November 30, 2019, were excluded. Data from the initial period of the COVID-19 pandemic were avoided because of alterations in clinic and prescribing practices. As a quality improvement project, the TOT evaluation was exempt from formal review by the VAPSHCS Institutional Review Board, as determined by the Director of the Office of Transformation/Quality/Safety/Value.
Interventions
Testosterone is a Schedule III controlled substance with potential risks and a propensity for varied prescribing practices. It was designated as a restricted drug requiring a prior authorization drug request (PADR) for which a specific TOT was developed, approved by the VAPSHCS Pharmacy and Therapeutics Committee, and incorporated into CPRS. A team of pharmacists, primary care physicians, geriatricians, endocrinologists, and health informatics experts created and developed the TOT. Pharmacists managed and monitored its completion.
The process for prescribing testosterone via the TOT is outlined in the eAppendix. When an HCP orders testosterone in CPRS, reminders prompt them to use the TOT and indicate required laboratory measurements (an order set is provided). Completion of TOT is not necessary to order testosterone for patients with an existing diagnosis of an organic cause of hypogonadism (eg, Klinefelter syndrome or hypopituitarism) or transgender women (assigned male at birth). In the TOT, the prescriber must also indicate signs and symptoms of testosterone deficiency; required laboratory tests; and counseling regarding potential risks and benefits of TRT. A pharmacist reviews the TOT and either approves or rejects the testosterone prescription and provides follow-up guidance to the prescriber. The completed TOT serves as documentation of guideline adherence in CPRS. The TOT also includes sections for first renewal testosterone prescriptions, addressing guideline recommendations for follow-up laboratory evaluation and clinical response to TRT. Due to limited completion of this section in the posttemplate period, evaluating adherence to follow-up recommendations was not feasible.
Measures
This project assessed the percentage of patients in the posttemplate period vs pretemplate period with an approved PADR. Documentation of specific guideline-recommended measures was assessed: signs and symptoms of testosterone deficiency; ≥ 2 serum testosterone measurements (≥ 2 total, free and total, or 2 free testosterone levels, and ≥ 1 testosterone level before 10
The project also assessed the proportion of patients in the posttemplate period vs pretemplate period who had all hormone tests (≥ 2 serum testosterone and LH and FSH concentrations), all laboratory tests (hormone tests and hematocrit), and all 5 guideline-recommended measures.
Analysis
Statistical comparisons between the proportions of patients in the pretemplate and posttemplate periods for each measure were performed using a χ2 test, without correction for multiple comparisons. All analyses were conducted using Stata version 10.0. A P value < .05 was considered significant for all comparisons.
Results
Chart review identified 189 patients in the pretemplate period and 113 patients in the posttemplate period with a new testosterone prescription (Figure). After exclusions, 91 and 49 patients, respectively, met eligibility criteria (Table 1). Fifty-six patients (62%) pretemplate and 40 patients (82%) posttemplate (P = .015) had approved PADRs and comprised the groups that were analyzed (Table 2).



The mean age and body mass index were similar in the pretemplate and posttemplate periods, but there was variation in the proportions of patients aged < 70 years and those with a body mass index < 30 between the groups. The most common diagnosis in both groups was testicular hypofunction, and the most common comorbidity was type 2 diabetes mellitus. Concomitant use of opioids or glucocorticoids that can lower testosterone levels was rare. Most testosterone prescriptions originated from primary care clinics in both periods: 68 (75%) in the pretemplate period and 35 (71%) in the posttemplate period. Most testosterone treatment was delivered by intramuscular injection.
In the posttemplate period vs pretemplate period, the proportion of patients with an approved PADR (82% vs 62%, P = .02), and documentation of signs and symptoms of hypogonadism (93% vs 71%, P = .002) prior to starting TRT were higher, while the percentage of patients having ≥ 2 testosterone measurements (85% vs 89%, P = .53), ≥ 1 testosterone level before 10 AM (78% vs 75%, P = .70), and hematocrit measured (95% vs 91%, P = .47) were similar. Rates of LH and FSH testing were higher in the posttemplate period (80%) vs the pretemplate period (63%) but did not achieve statistical significance (P = .07), and discussion of the risks and benefits of TRT was higher in the posttemplate period (58%) vs the pretemplate period (34%) (P = .02). The percentage of patients who had all hormone measurements (total and/or free testosterone, LH, and FSH) was higher in the posttemplate period (78%) vs the pretemplate period (59%) but did not achieve statistical significance (P = .06). The rates of all guideline-recommended laboratory test orders were higher in the posttemplate period (78%) vs the pretemplate period (55%) (P = .03), and all 5 guideline-recommended clinical and laboratory measures were higher in the posttemplate period (45%) vs the pretemplate period (18%) (P = .004).
Discussion
The implementation of a pharmacy-managed TOT in CPRS demonstrated higher adherence to evidence-based guidelines for diagnosing and evaluating hypogonadism before TRT. After TOT implementation, a higher proportion of patients had documented signs and symptoms of testosterone deficiency, underwent all recommended laboratory tests, and had discussions about the risks and benefits of TRT. Adherence to 5 clinical and laboratory measures recommended by Endocrine Society guidelines was higher after TOT implementation, indicating improved prescribing practices.4
The requirement for TOT completion before testosterone prescription and its management by trained pharmacists likely contributed to higher adherence to guideline recommendations than previously reported. Integration of the TOT into CPRS with pharmacy oversight may have enhanced adherence by summarizing and codifying evidence-based guideline recommendations for clinical and biochemical evaluation prior to TRT initiation, offering relevant education to clinicians and pharmacists, automatically importing pertinent clinical information and laboratory results, and generating CPRS documentation to reduce clinician burden during patient care.
The proportion of patients with documented signs and symptoms of testosterone deficiency before TRT increased from the pretemplate period (71%) to the posttemplate period (93%), indicating that most patients receiving TRT had clinical manifestations of hypogonadism. This aligns with Endocrine Society guidelines, which define hypogonadism as a clinical disorder characterized by clinical manifestations of testosterone deficiency and persistently low serum testosterone levels on ≥ 2 separate occasions.4,6 However, recent trends in direct-to-consumer advertising for testosterone and the rise of “low T” clinics may contribute to increased testing, varied practices, and inappropriate testosterone therapy initiation (eg, in men with low testosterone levels who lack symptoms of hypogonadism).18 Improved adherence in documenting clinical hypogonadism with implementation of the TOT reinforces the value of incorporating educational material, as previously reported.11
Adherence to guideline recommendations following implementation of the TOT in this project was higher than those previously reported. In a study of 111,631 outpatient veterans prescribed testosterone from 2009 to 2012, only 18.3% had ≥ 2 testosterone prescriptions, and 3.5% had ≥ 2 testosterone, LH, and FSH levels measured prior to the initiation of a TRT.9 In a report of 63,534 insured patients who received TRT from 2010 to 2012, 40.3% had ≥ 2 testosterone prescriptions, and 12% had LH and/or FSH measured prior to the initiation.8
Low rates of guideline-recommended laboratory tests prior to initiation of testosterone treatment were reported in prior non-VA studies.19,20 Poor guideline adherence reinforces the need for clinician education or other methods to improve TRT and ensure appropriate prescribing practices across health care systems. The TOT described in this project is a sustainable clinical tool with the potential to improve testosterone prescribing practices.
The high rates of adherence to guideline recommendations at VAPSHCS likely stem from local endocrine expertise and ongoing educational initiatives, as well as the requirement for template completion before testosterone prescription. However, most testosterone prescriptions were initiated by primary care and monitored by pharmacists with varying degrees of training and clinical experience in hypogonadism and TRT.
However, adherence to guideline recommendations was modest, suggesting there is still an opportunity for improvement. The decision to initiate therapy should be made only after appropriate counseling with patients regarding its potential benefits and risks. Reports on the CV risk of TRT have been mixed. The 2023 TRAVERSE study found no increase in major adverse CV events among older men with hypogonadism and pre-existing CV risks undergoing TRT, but noted higher instances of pulmonary embolism, atrial fibrillation, and acute kidney injury.21 This highlights the need for clinicians to continue to engage in informed decision-making with patients. Effective pretreatment counseling is important but time-consuming; future TOT monitoring and modifications could consider mandatory checkboxes to document counseling on TRT risks and benefits.
The TOT described in this study could be adapted and incorporated into the prescribing process and electronic health record of larger health care systems. Use of an electronic template allows for automatic real-time dashboard monitoring of organization performance. The TOT described could be modified or simplified for specialty or primary care clinics or individual practitioners to improve adherence to evidence-based guideline recommendations and quality of care.
Strengths
A strength of this study is the multidisciplinary team (composed of stakeholders with experience in VA health care system and subject matter experts in hypogonadism) that developed and incorporated a user-friendly template for testosterone prescriptions; the use of evidence-based guideline recommendations; and the use of a structured chart review permitted accurate assessment of adherence to recommendations to document signs and symptoms of testosterone deficiency and a discussion of potential risks and benefits prior to TRT. To our knowledge, these recommendations have not been assessed in previous reports.
Limitations
The retrospective pre-post design of this study precludes a conclusion that implementation of the TOT caused the increase in adherence to guideline recommendations. Improved adherence could have resulted from the ongoing development of the preauthorization process for testosterone prescriptions or other changes over time. However, the preauthorization process had already been established for many years prior to template implementation. Forty-nine patients had new prescriptions for testosterone in the posttemplate period compared to 91 in the pretemplate period, but TRT was initiated in accordance with guideline recommendations more appropriately in the posttemplate period. The study’s sample size was small, and many eligible patients were excluded; however, exclusions were necessary to evaluate men who had new testosterone prescriptions for which the template was designed. Most men excluded were already taking testosterone.
Conclusions
The implementation of a CPRS-based TOT improved adherence to evidence-based guidelines for the diagnosis, evaluation, and counseling of patients with hypogonadism before starting TRT. While there were improvements in adherence with the TOT, the relatively low proportion of patients with documentation of TRT risks and benefits and all guideline recommendations highlights the need for additional efforts to further strengthen adherence to guideline recommendations and ensure appropriate evaluation, counseling, and prescribing practices before initiating TRT.
Testosterone therapy is administered following pragmatic diagnostic evaluation and workup to assess whether an adult male is hypogonadal, based on symptoms consistent with androgen deficiency and low morning serum testosterone concentrations on ≥ 2 occasions. Effects of testosterone administration include the development or maintenance of secondary sexual characteristics and increases in libido, muscle strength, fat-free mass, and bone density.
Testosterone prescriptions have markedly increased in the past 20 years, including within the US Department of Veterans Affairs (VA) health care system.1-3 This trend may be influenced by various factors, including patient perceptions of benefit, an increase in marketing, and the availability of more user-friendly formulations.
Since 2006, evidence-based clinical practice guidelines have recommended specific clinical and laboratory evaluation and counseling prior to starting testosterone replacement therapy (TRT).4-8 However, research has shown poor adherence to these recommendations, including at the VA, which raises concerns about inappropriate TRT initiation without proper diagnostic evaluation.9,10 Observational research has suggested a possible link between testosterone therapy and increased risk of cardiovascular (CV) events. The US Food and Drug Administration prescribing information includes boxed warnings about potential risks of high blood pressure, myocardial infarction, stroke, and CV-related mortality with testosterone treatment, contact transfer of transdermal testosterone, and pulmonary oil microembolism with testosterone undecanoate injections.11-15
A VA Office of Inspector General (OIG) review of VA clinician adherence to clinical and laboratory evaluation guidelines for testosterone deficiency found poor adherence among VA practitioners and made recommendations for improvement.4,15 These focused on establishing clinical signs and symptoms consistent with testosterone deficiency, confirming hypogonadism by repeated testosterone testing, determining the etiology of hypogonadism by measuring gonadotropins, initiating a discussion of risks and benefits of TRT, and assessing clinical improvement and obtaining an updated hematocrit test within 3 to 6 months of initiation.
The VA Puget Sound Health Care System (VAPSHCS) developed a local prior authorization template to assist health care practitioners (HCPs) to address the OIG recommendations. This testosterone order template (TOT) aimed to improve the diagnosis, evaluation, and monitoring of TRT in males with hypogonadism, combined with existing VA pharmacy criteria for the use of testosterone based on Endocrine Society guidelines. A version of the VAPSHCS TOT was approved as the national VA Computerized Patient Record System (CPRS) template.
Preliminary evaluation of the TOT suggested improved short-term adherence to guideline recommendations following implementation.16 This quality improvement study sought to assess the long-term effectiveness of the TOT with respect to clinical practice guideline adherence. The OIG did not address prostate-specific antigen (PSA) monitoring because understanding of the relationship between TRT and the risks of elevated PSA levels remains incomplete.6,17 This project hypothesized that implementation of a pharmacy-managed TOT incorporated into CPRS would result in higher adherence rates to guideline-recommended clinical and laboratory evaluation, in addition to counseling of men with hypogonadism prior to initiation of TRT.
Methods
Eligible participants were cisgender males who received a new testosterone prescription, had ≥ 2 clinic visits at VAPSHCS, and no previous testosterone prescription in the previous 2 years. Individuals were excluded if they had testosterone administered at VAPSHCS; were prescribed testosterone at another facility (VA or community-based); pilot tested an initial version of the TOT prior to November 30, 2019; or had an International Classification of Diseases, Tenth Revision codes for hypopituitarism, gender identity disorder, history of sexual assignment, or Klinefelter syndrome for which testosterone therapy was already approved. Patients who met the inclusion criteria were identified by an algorithm developed by the VAPSHCS pharmacoeconomist.
This quality improvement project used a retrospective, pre-post experimental design. Electronic chart review and systematic manual review of all eligible patient charts were performed for the pretemplate period (December 1, 2018, to November 30, 2019) and after the template implementation, (December 1, 2021, to November 30, 2022).
An initial version of the TOT was implemented on July 1, 2019, but was not fully integrated into CPRS until early 2020; individuals in whom the TOT was used prior to November 30, 2019, were excluded. Data from the initial period of the COVID-19 pandemic were avoided because of alterations in clinic and prescribing practices. As a quality improvement project, the TOT evaluation was exempt from formal review by the VAPSHCS Institutional Review Board, as determined by the Director of the Office of Transformation/Quality/Safety/Value.
Interventions
Testosterone is a Schedule III controlled substance with potential risks and a propensity for varied prescribing practices. It was designated as a restricted drug requiring a prior authorization drug request (PADR) for which a specific TOT was developed, approved by the VAPSHCS Pharmacy and Therapeutics Committee, and incorporated into CPRS. A team of pharmacists, primary care physicians, geriatricians, endocrinologists, and health informatics experts created and developed the TOT. Pharmacists managed and monitored its completion.
The process for prescribing testosterone via the TOT is outlined in the eAppendix. When an HCP orders testosterone in CPRS, reminders prompt them to use the TOT and indicate required laboratory measurements (an order set is provided). Completion of TOT is not necessary to order testosterone for patients with an existing diagnosis of an organic cause of hypogonadism (eg, Klinefelter syndrome or hypopituitarism) or transgender women (assigned male at birth). In the TOT, the prescriber must also indicate signs and symptoms of testosterone deficiency; required laboratory tests; and counseling regarding potential risks and benefits of TRT. A pharmacist reviews the TOT and either approves or rejects the testosterone prescription and provides follow-up guidance to the prescriber. The completed TOT serves as documentation of guideline adherence in CPRS. The TOT also includes sections for first renewal testosterone prescriptions, addressing guideline recommendations for follow-up laboratory evaluation and clinical response to TRT. Due to limited completion of this section in the posttemplate period, evaluating adherence to follow-up recommendations was not feasible.
Measures
This project assessed the percentage of patients in the posttemplate period vs pretemplate period with an approved PADR. Documentation of specific guideline-recommended measures was assessed: signs and symptoms of testosterone deficiency; ≥ 2 serum testosterone measurements (≥ 2 total, free and total, or 2 free testosterone levels, and ≥ 1 testosterone level before 10
The project also assessed the proportion of patients in the posttemplate period vs pretemplate period who had all hormone tests (≥ 2 serum testosterone and LH and FSH concentrations), all laboratory tests (hormone tests and hematocrit), and all 5 guideline-recommended measures.
Analysis
Statistical comparisons between the proportions of patients in the pretemplate and posttemplate periods for each measure were performed using a χ2 test, without correction for multiple comparisons. All analyses were conducted using Stata version 10.0. A P value < .05 was considered significant for all comparisons.
Results
Chart review identified 189 patients in the pretemplate period and 113 patients in the posttemplate period with a new testosterone prescription (Figure). After exclusions, 91 and 49 patients, respectively, met eligibility criteria (Table 1). Fifty-six patients (62%) pretemplate and 40 patients (82%) posttemplate (P = .015) had approved PADRs and comprised the groups that were analyzed (Table 2).



The mean age and body mass index were similar in the pretemplate and posttemplate periods, but there was variation in the proportions of patients aged < 70 years and those with a body mass index < 30 between the groups. The most common diagnosis in both groups was testicular hypofunction, and the most common comorbidity was type 2 diabetes mellitus. Concomitant use of opioids or glucocorticoids that can lower testosterone levels was rare. Most testosterone prescriptions originated from primary care clinics in both periods: 68 (75%) in the pretemplate period and 35 (71%) in the posttemplate period. Most testosterone treatment was delivered by intramuscular injection.
In the posttemplate period vs pretemplate period, the proportion of patients with an approved PADR (82% vs 62%, P = .02), and documentation of signs and symptoms of hypogonadism (93% vs 71%, P = .002) prior to starting TRT were higher, while the percentage of patients having ≥ 2 testosterone measurements (85% vs 89%, P = .53), ≥ 1 testosterone level before 10 AM (78% vs 75%, P = .70), and hematocrit measured (95% vs 91%, P = .47) were similar. Rates of LH and FSH testing were higher in the posttemplate period (80%) vs the pretemplate period (63%) but did not achieve statistical significance (P = .07), and discussion of the risks and benefits of TRT was higher in the posttemplate period (58%) vs the pretemplate period (34%) (P = .02). The percentage of patients who had all hormone measurements (total and/or free testosterone, LH, and FSH) was higher in the posttemplate period (78%) vs the pretemplate period (59%) but did not achieve statistical significance (P = .06). The rates of all guideline-recommended laboratory test orders were higher in the posttemplate period (78%) vs the pretemplate period (55%) (P = .03), and all 5 guideline-recommended clinical and laboratory measures were higher in the posttemplate period (45%) vs the pretemplate period (18%) (P = .004).
Discussion
The implementation of a pharmacy-managed TOT in CPRS demonstrated higher adherence to evidence-based guidelines for diagnosing and evaluating hypogonadism before TRT. After TOT implementation, a higher proportion of patients had documented signs and symptoms of testosterone deficiency, underwent all recommended laboratory tests, and had discussions about the risks and benefits of TRT. Adherence to 5 clinical and laboratory measures recommended by Endocrine Society guidelines was higher after TOT implementation, indicating improved prescribing practices.4
The requirement for TOT completion before testosterone prescription and its management by trained pharmacists likely contributed to higher adherence to guideline recommendations than previously reported. Integration of the TOT into CPRS with pharmacy oversight may have enhanced adherence by summarizing and codifying evidence-based guideline recommendations for clinical and biochemical evaluation prior to TRT initiation, offering relevant education to clinicians and pharmacists, automatically importing pertinent clinical information and laboratory results, and generating CPRS documentation to reduce clinician burden during patient care.
The proportion of patients with documented signs and symptoms of testosterone deficiency before TRT increased from the pretemplate period (71%) to the posttemplate period (93%), indicating that most patients receiving TRT had clinical manifestations of hypogonadism. This aligns with Endocrine Society guidelines, which define hypogonadism as a clinical disorder characterized by clinical manifestations of testosterone deficiency and persistently low serum testosterone levels on ≥ 2 separate occasions.4,6 However, recent trends in direct-to-consumer advertising for testosterone and the rise of “low T” clinics may contribute to increased testing, varied practices, and inappropriate testosterone therapy initiation (eg, in men with low testosterone levels who lack symptoms of hypogonadism).18 Improved adherence in documenting clinical hypogonadism with implementation of the TOT reinforces the value of incorporating educational material, as previously reported.11
Adherence to guideline recommendations following implementation of the TOT in this project was higher than those previously reported. In a study of 111,631 outpatient veterans prescribed testosterone from 2009 to 2012, only 18.3% had ≥ 2 testosterone prescriptions, and 3.5% had ≥ 2 testosterone, LH, and FSH levels measured prior to the initiation of a TRT.9 In a report of 63,534 insured patients who received TRT from 2010 to 2012, 40.3% had ≥ 2 testosterone prescriptions, and 12% had LH and/or FSH measured prior to the initiation.8
Low rates of guideline-recommended laboratory tests prior to initiation of testosterone treatment were reported in prior non-VA studies.19,20 Poor guideline adherence reinforces the need for clinician education or other methods to improve TRT and ensure appropriate prescribing practices across health care systems. The TOT described in this project is a sustainable clinical tool with the potential to improve testosterone prescribing practices.
The high rates of adherence to guideline recommendations at VAPSHCS likely stem from local endocrine expertise and ongoing educational initiatives, as well as the requirement for template completion before testosterone prescription. However, most testosterone prescriptions were initiated by primary care and monitored by pharmacists with varying degrees of training and clinical experience in hypogonadism and TRT.
However, adherence to guideline recommendations was modest, suggesting there is still an opportunity for improvement. The decision to initiate therapy should be made only after appropriate counseling with patients regarding its potential benefits and risks. Reports on the CV risk of TRT have been mixed. The 2023 TRAVERSE study found no increase in major adverse CV events among older men with hypogonadism and pre-existing CV risks undergoing TRT, but noted higher instances of pulmonary embolism, atrial fibrillation, and acute kidney injury.21 This highlights the need for clinicians to continue to engage in informed decision-making with patients. Effective pretreatment counseling is important but time-consuming; future TOT monitoring and modifications could consider mandatory checkboxes to document counseling on TRT risks and benefits.
The TOT described in this study could be adapted and incorporated into the prescribing process and electronic health record of larger health care systems. Use of an electronic template allows for automatic real-time dashboard monitoring of organization performance. The TOT described could be modified or simplified for specialty or primary care clinics or individual practitioners to improve adherence to evidence-based guideline recommendations and quality of care.
Strengths
A strength of this study is the multidisciplinary team (composed of stakeholders with experience in VA health care system and subject matter experts in hypogonadism) that developed and incorporated a user-friendly template for testosterone prescriptions; the use of evidence-based guideline recommendations; and the use of a structured chart review permitted accurate assessment of adherence to recommendations to document signs and symptoms of testosterone deficiency and a discussion of potential risks and benefits prior to TRT. To our knowledge, these recommendations have not been assessed in previous reports.
Limitations
The retrospective pre-post design of this study precludes a conclusion that implementation of the TOT caused the increase in adherence to guideline recommendations. Improved adherence could have resulted from the ongoing development of the preauthorization process for testosterone prescriptions or other changes over time. However, the preauthorization process had already been established for many years prior to template implementation. Forty-nine patients had new prescriptions for testosterone in the posttemplate period compared to 91 in the pretemplate period, but TRT was initiated in accordance with guideline recommendations more appropriately in the posttemplate period. The study’s sample size was small, and many eligible patients were excluded; however, exclusions were necessary to evaluate men who had new testosterone prescriptions for which the template was designed. Most men excluded were already taking testosterone.
Conclusions
The implementation of a CPRS-based TOT improved adherence to evidence-based guidelines for the diagnosis, evaluation, and counseling of patients with hypogonadism before starting TRT. While there were improvements in adherence with the TOT, the relatively low proportion of patients with documentation of TRT risks and benefits and all guideline recommendations highlights the need for additional efforts to further strengthen adherence to guideline recommendations and ensure appropriate evaluation, counseling, and prescribing practices before initiating TRT.
- Layton JB, Li D, Meier CR, et al. Testosterone lab testing and initiation in the United Kingdom and the United States, 2000 to 2011. J Clin Endocrinol Metab. 2014;99:835-842. doi:10.1210/jc.2013-3570
- Baillargeon J, Kuo YF, Westra JR, et al. Testosterone prescribing in the United States, 2002-2016. JAMA. 2018;320:200-202. doi:10.1001/jama.2018.7999
- Jasuja GK, Bhasin S, Rose AJ. Patterns of testosterone prescription overuse. Curr Opin Endocrinol Diabetes Obes. 2017;24:240-245. doi:10.1097/MED.0000000000000336
- Bhasin S, Cunningham GR, Hayes FJ, et al. Testosterone therapy in adult men with androgen deficiency syndromes: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2006;91:1995-2010. doi:10.1210/jc.2005-2847
- Bhasin S, Cunningham GR, Hayes FJ, et al. Testosterone therapy in men with androgen deficiency syndromes: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2010;95:2536-2559. doi:10.1210/jc.2009-2354
- Bhasin S, Brito JP, Cunningham GR, et al. Testosterone therapy in men with hypogonadism: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2018;103:1715-1744. doi:10.1210/jc.2018-00229
- Mulhall JP, Trost LW, Brannigan RE, et al. Evaluation and management of testosterone deficiency: AUA guideline. J Urol. 2018;200:423-432. doi:10.1016/j.juro.2018.03.115
- Muram D, Zhang X, Cui Z, et al. Use of hormone testing for the diagnosis and evaluation of male hypogonadism and monitoring of testosterone therapy: application of hormone testing guideline recommendations in clinical practice. J Sex Med. 2015;12:1886-1894. doi:10.1111/jsm.12968
- Jasuja GK, Bhasin S, Reisman JI, et al. Ascertainment of testosterone prescribing practices in the VA. Med Care. 2015;53:746-752. doi:10.1097/MLR.0000000000000398?
- Jasuja GK, Bhasin S, Reisman JI, et al. Who gets testosterone? Patient characteristics associated with testosterone prescribing in the Veteran Affairs system: a cross-sectional study. J Gen Intern Med. 2017;32:304-311. doi:10.1007/s11606-016-3940-7
- Basaria S, Coviello AD, Travison TG, et al. Adverse events associated with testosterone administration. N Engl J Med. 2010;363:109-122. doi:10.1056/NEJMoa1000485
- Vigen R, O’Donnell CI, Barón AE, et al. Association of testosterone therapy with mortality, myocardial infarction, and stroke in men with low testosterone levels. JAMA. 2013;310:1829-1836. doi:10.1001/jama.2013.280386
- Finkle WD, Greenland S, Ridgeway GK, et al. Increased risk of non-fatal myocardial infarction following testosterone therapy prescription in men. PLoS One. 2014;9:e85805. doi:10.1371/journal.pone.0085805
- US Food and Drug Administration. FDA Drug Safety Communication: FDA cautions about using testosterone products for low testosterone due to aging; requires labeling change to inform of possible increased risk of heart attack and stroke with use. FDA.gov. March 3, 2015. Updated February 28, 2025. Accessed July 8, 2025. http://www.fda.gov/Drugs/DrugSafety/ucm436259.htm
- US Dept of Veterans Affairs, Office of Inspector General. Healthcare inspection – testosterone replacement therapy initiation and follow-up evaluation in VA male patients. April 11, 2018. Accessed July 8, 2025. https://www.vaoig.gov/reports/national-healthcare-review/healthcare-inspection-testosterone-replacement-therapy
- Narla R, Mobley D, Nguyen EHK, et al. Preliminary evaluation of an order template to improve diagnosis and testosterone therapy of hypogonadism in veterans. Fed Pract. 2021;38:121-127. doi:10.12788/fp.0103
- Bhasin S, Travison TG, Pencina KM, et al. Prostate safety events during testosterone replacement therapy in men with hypogonadism: a randomized clinical trial. JAMA Netw Open. 2023;6:e2348692. doi:10.1001/jamanetworkopen.2023.48692
- Dubin JM, Jesse E, Fantus RJ, et al. Guideline-discordant care among direct-to-consumer testosterone therapy platforms. JAMA Intern Med. 2022;182:1321-1323. doi:10.1001/jamainternmed.2022.4928
- Baillargeon J, Urban RJ, Ottenbacher KJ, et al. Trends in androgen prescribing in the United States, 2001 to 2011. JAMA Intern Med. 2013;173:1465-1466. doi:10.1001/jamainternmed.2013.6895
- Locke JA, Flannigan R, Günther OP, et al. Testosterone therapy: prescribing and monitoring patterns of practice in British Columbia. Can Urol Assoc J. 2021;15:e110-e117. doi:10.5489/cuaj.6586
- Lincoff AM, Bhasin S, Flevaris P, et al. Cardiovascular safety of testosterone-replacement therapy. N Engl J Med. 2023;389:107-117. doi:10.1056/NEJMoa2215025
- Layton JB, Li D, Meier CR, et al. Testosterone lab testing and initiation in the United Kingdom and the United States, 2000 to 2011. J Clin Endocrinol Metab. 2014;99:835-842. doi:10.1210/jc.2013-3570
- Baillargeon J, Kuo YF, Westra JR, et al. Testosterone prescribing in the United States, 2002-2016. JAMA. 2018;320:200-202. doi:10.1001/jama.2018.7999
- Jasuja GK, Bhasin S, Rose AJ. Patterns of testosterone prescription overuse. Curr Opin Endocrinol Diabetes Obes. 2017;24:240-245. doi:10.1097/MED.0000000000000336
- Bhasin S, Cunningham GR, Hayes FJ, et al. Testosterone therapy in adult men with androgen deficiency syndromes: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2006;91:1995-2010. doi:10.1210/jc.2005-2847
- Bhasin S, Cunningham GR, Hayes FJ, et al. Testosterone therapy in men with androgen deficiency syndromes: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2010;95:2536-2559. doi:10.1210/jc.2009-2354
- Bhasin S, Brito JP, Cunningham GR, et al. Testosterone therapy in men with hypogonadism: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2018;103:1715-1744. doi:10.1210/jc.2018-00229
- Mulhall JP, Trost LW, Brannigan RE, et al. Evaluation and management of testosterone deficiency: AUA guideline. J Urol. 2018;200:423-432. doi:10.1016/j.juro.2018.03.115
- Muram D, Zhang X, Cui Z, et al. Use of hormone testing for the diagnosis and evaluation of male hypogonadism and monitoring of testosterone therapy: application of hormone testing guideline recommendations in clinical practice. J Sex Med. 2015;12:1886-1894. doi:10.1111/jsm.12968
- Jasuja GK, Bhasin S, Reisman JI, et al. Ascertainment of testosterone prescribing practices in the VA. Med Care. 2015;53:746-752. doi:10.1097/MLR.0000000000000398?
- Jasuja GK, Bhasin S, Reisman JI, et al. Who gets testosterone? Patient characteristics associated with testosterone prescribing in the Veteran Affairs system: a cross-sectional study. J Gen Intern Med. 2017;32:304-311. doi:10.1007/s11606-016-3940-7
- Basaria S, Coviello AD, Travison TG, et al. Adverse events associated with testosterone administration. N Engl J Med. 2010;363:109-122. doi:10.1056/NEJMoa1000485
- Vigen R, O’Donnell CI, Barón AE, et al. Association of testosterone therapy with mortality, myocardial infarction, and stroke in men with low testosterone levels. JAMA. 2013;310:1829-1836. doi:10.1001/jama.2013.280386
- Finkle WD, Greenland S, Ridgeway GK, et al. Increased risk of non-fatal myocardial infarction following testosterone therapy prescription in men. PLoS One. 2014;9:e85805. doi:10.1371/journal.pone.0085805
- US Food and Drug Administration. FDA Drug Safety Communication: FDA cautions about using testosterone products for low testosterone due to aging; requires labeling change to inform of possible increased risk of heart attack and stroke with use. FDA.gov. March 3, 2015. Updated February 28, 2025. Accessed July 8, 2025. http://www.fda.gov/Drugs/DrugSafety/ucm436259.htm
- US Dept of Veterans Affairs, Office of Inspector General. Healthcare inspection – testosterone replacement therapy initiation and follow-up evaluation in VA male patients. April 11, 2018. Accessed July 8, 2025. https://www.vaoig.gov/reports/national-healthcare-review/healthcare-inspection-testosterone-replacement-therapy
- Narla R, Mobley D, Nguyen EHK, et al. Preliminary evaluation of an order template to improve diagnosis and testosterone therapy of hypogonadism in veterans. Fed Pract. 2021;38:121-127. doi:10.12788/fp.0103
- Bhasin S, Travison TG, Pencina KM, et al. Prostate safety events during testosterone replacement therapy in men with hypogonadism: a randomized clinical trial. JAMA Netw Open. 2023;6:e2348692. doi:10.1001/jamanetworkopen.2023.48692
- Dubin JM, Jesse E, Fantus RJ, et al. Guideline-discordant care among direct-to-consumer testosterone therapy platforms. JAMA Intern Med. 2022;182:1321-1323. doi:10.1001/jamainternmed.2022.4928
- Baillargeon J, Urban RJ, Ottenbacher KJ, et al. Trends in androgen prescribing in the United States, 2001 to 2011. JAMA Intern Med. 2013;173:1465-1466. doi:10.1001/jamainternmed.2013.6895
- Locke JA, Flannigan R, Günther OP, et al. Testosterone therapy: prescribing and monitoring patterns of practice in British Columbia. Can Urol Assoc J. 2021;15:e110-e117. doi:10.5489/cuaj.6586
- Lincoff AM, Bhasin S, Flevaris P, et al. Cardiovascular safety of testosterone-replacement therapy. N Engl J Med. 2023;389:107-117. doi:10.1056/NEJMoa2215025
Streamlined Testosterone Order Template to Improve the Diagnosis and Evaluation of Hypogonadism in Veterans
Streamlined Testosterone Order Template to Improve the Diagnosis and Evaluation of Hypogonadism in Veterans
Simpler Approach Increases Diagnostic Accuracy of Timed Barium Esophagram for Achalasia
, according to investigators.
The classification tree offers a practical alternative for evaluating esophagogastric junction (EGJ) outflow disorders when more advanced methods like high-resolution manometry (HRM) or functional lumen imaging probe (FLIP) panometry are unavailable, lead author Ofer Z. Fass, MD, of Northwestern University, Chicago, and colleagues reported.
“[T]here are limited data on normative TBE values,” the investigators wrote in Gastroenterology. “Furthermore, data supporting the accuracy of TBE as a screening test for esophageal motility disorders, as well as clinically relevant test thresholds, remains limited.”
TBE is conventionally interpreted using a handful of single measurements, most often the barium column height at 1, 2, or 5 minutes. Although these metrics are simple to obtain, variability in technique, cutoff values, and interpretation across centers limits reproducibility and weakens diagnostic accuracy, according to the investigators. The role of TBE has therefore been largely confined to adjudicating inconclusive manometry findings, but even in that setting, the absence of validated reference standards constrains its utility as a reliable screening tool.
To address this gap, Fass and colleagues conducted a prospective analysis of 290 patients who underwent TBE at Northwestern Memorial Hospital, Chicago, with HRM and FLIP panometry, interpreted according to the Chicago Classification version 4.0 (CCv4.0), serving as the diagnostic reference standards.
Patients were included if they had both TBE and manometry performed within a short interval, ensuring that the two tests could be meaningfully compared. The study population represented a broad spectrum of esophageal motility presentations, allowing the model to be trained on clinically relevant variation.
Beyond column height, the investigators measured barium height at multiple timepoints, maximal esophageal body width, maximum EGJ diameter, and tablet passage. These variables were incorporated into a recursive partitioning algorithm to build a multimetric classification tree aimed at distinguishing EGJ outflow obstruction from other motility disorders.
The optimal tree incorporated three sequential decision levels. At the top was maximum esophageal body width, followed by EGJ diameter and barium height at the second level, and tablet passage at the third. This stepwise structure allowed the model to refine diagnoses by combining simple, reproducible TBE metrics that are already collected in routine practice.
Among the 290 patients, 121 (42%) had EGJ outflow disorders, 151 (52%) had no outflow disorder, and 18 (6%) had inconclusive manometry findings. Using conventional interpretation with column height and tablet passage, TBE demonstrated a sensitivity of 77.8%, a specificity of 86.0%, and an accuracy of 82.2%. The multimetric classification tree improved diagnostic performance across all parameters, with a sensitivity of 84.2%, a specificity of 92.1%, and an accuracy of 88.3%.
The advantages of multimetric interpretation were most notable in patients with borderline column heights, which single-metric approaches often misclassify, underscoring the value of integrating multiple measurements into a unified model.
“[T]his study demonstrated that TBE can accurately identify achalasia when analyzed using multiple metrics in a classification tree model,” Fass and colleagues wrote. “Future studies should explore the use of TBE metrics and models to identify more specific esophageal motor disorders (such as esophageal spasm and absent contractility), as well as validation in a larger, multicenter cohort.”
Clinical Takeaways
Rishi Naik, MD, of the Center for Swallowing and Esophageal Disorders, Vanderbilt University Medical Center, Nashville, Tennessee, said the study represents a step forward in how clinicians can use a widely accessible esophageal imaging test.
“This study is important in that it has updated the way we use a very common, readily available imaging test and compared it to the current gold standard of HRM and FLIP,” he told GI & Hepatology News. “This provides a practical, standardized framework for clinicians evaluating patients with suspected esophageal motility disorders.”
Naik noted that while HRM and FLIP provide highly detailed information, both carry drawbacks that limit their universal adoption.
“Practically, HRM is a transnasal test that can be cumbersome, and FLIP is performed during a sedated procedure,” he said. “From a comfort and cost perspective, the esophagram outcompetes. What the TBE lacked was adequate sensitivity and specificity when just looking at column height, which is how the authors overcame this by leveraging the comparisons using CCv4.0.”
Implementation, however, requires discipline.
“A timed barium esophagram is a protocol, not a single esophagram,” Naik said. “Without proper measurements, you can’t follow the decision tree.”
Still, he pointed to radiology’s increasing adoption of artificial intelligence (AI) as a promising way forward.
“AI has already transformed radiological reads, and I’m optimistic it will eventually allow us to incorporate not only width, height, and tablet clearance but also 3D [three-dimensional] reconstructions of bolus retention and pressure to enhance predictive modeling,” Naik said.
This study was supported by the Public Health Service.
The investigators disclosed having relationships with Takeda, Phathom Pharmaceuticals, Medtronic, and others. Naik is a consultant for Sanofi/Regeneron, Eli Lilly and Company, and Renexxion.
A version of this article appeared on Medscape.com.
, according to investigators.
The classification tree offers a practical alternative for evaluating esophagogastric junction (EGJ) outflow disorders when more advanced methods like high-resolution manometry (HRM) or functional lumen imaging probe (FLIP) panometry are unavailable, lead author Ofer Z. Fass, MD, of Northwestern University, Chicago, and colleagues reported.
“[T]here are limited data on normative TBE values,” the investigators wrote in Gastroenterology. “Furthermore, data supporting the accuracy of TBE as a screening test for esophageal motility disorders, as well as clinically relevant test thresholds, remains limited.”
TBE is conventionally interpreted using a handful of single measurements, most often the barium column height at 1, 2, or 5 minutes. Although these metrics are simple to obtain, variability in technique, cutoff values, and interpretation across centers limits reproducibility and weakens diagnostic accuracy, according to the investigators. The role of TBE has therefore been largely confined to adjudicating inconclusive manometry findings, but even in that setting, the absence of validated reference standards constrains its utility as a reliable screening tool.
To address this gap, Fass and colleagues conducted a prospective analysis of 290 patients who underwent TBE at Northwestern Memorial Hospital, Chicago, with HRM and FLIP panometry, interpreted according to the Chicago Classification version 4.0 (CCv4.0), serving as the diagnostic reference standards.
Patients were included if they had both TBE and manometry performed within a short interval, ensuring that the two tests could be meaningfully compared. The study population represented a broad spectrum of esophageal motility presentations, allowing the model to be trained on clinically relevant variation.
Beyond column height, the investigators measured barium height at multiple timepoints, maximal esophageal body width, maximum EGJ diameter, and tablet passage. These variables were incorporated into a recursive partitioning algorithm to build a multimetric classification tree aimed at distinguishing EGJ outflow obstruction from other motility disorders.
The optimal tree incorporated three sequential decision levels. At the top was maximum esophageal body width, followed by EGJ diameter and barium height at the second level, and tablet passage at the third. This stepwise structure allowed the model to refine diagnoses by combining simple, reproducible TBE metrics that are already collected in routine practice.
Among the 290 patients, 121 (42%) had EGJ outflow disorders, 151 (52%) had no outflow disorder, and 18 (6%) had inconclusive manometry findings. Using conventional interpretation with column height and tablet passage, TBE demonstrated a sensitivity of 77.8%, a specificity of 86.0%, and an accuracy of 82.2%. The multimetric classification tree improved diagnostic performance across all parameters, with a sensitivity of 84.2%, a specificity of 92.1%, and an accuracy of 88.3%.
The advantages of multimetric interpretation were most notable in patients with borderline column heights, which single-metric approaches often misclassify, underscoring the value of integrating multiple measurements into a unified model.
“[T]his study demonstrated that TBE can accurately identify achalasia when analyzed using multiple metrics in a classification tree model,” Fass and colleagues wrote. “Future studies should explore the use of TBE metrics and models to identify more specific esophageal motor disorders (such as esophageal spasm and absent contractility), as well as validation in a larger, multicenter cohort.”
Clinical Takeaways
Rishi Naik, MD, of the Center for Swallowing and Esophageal Disorders, Vanderbilt University Medical Center, Nashville, Tennessee, said the study represents a step forward in how clinicians can use a widely accessible esophageal imaging test.
“This study is important in that it has updated the way we use a very common, readily available imaging test and compared it to the current gold standard of HRM and FLIP,” he told GI & Hepatology News. “This provides a practical, standardized framework for clinicians evaluating patients with suspected esophageal motility disorders.”
Naik noted that while HRM and FLIP provide highly detailed information, both carry drawbacks that limit their universal adoption.
“Practically, HRM is a transnasal test that can be cumbersome, and FLIP is performed during a sedated procedure,” he said. “From a comfort and cost perspective, the esophagram outcompetes. What the TBE lacked was adequate sensitivity and specificity when just looking at column height, which is how the authors overcame this by leveraging the comparisons using CCv4.0.”
Implementation, however, requires discipline.
“A timed barium esophagram is a protocol, not a single esophagram,” Naik said. “Without proper measurements, you can’t follow the decision tree.”
Still, he pointed to radiology’s increasing adoption of artificial intelligence (AI) as a promising way forward.
“AI has already transformed radiological reads, and I’m optimistic it will eventually allow us to incorporate not only width, height, and tablet clearance but also 3D [three-dimensional] reconstructions of bolus retention and pressure to enhance predictive modeling,” Naik said.
This study was supported by the Public Health Service.
The investigators disclosed having relationships with Takeda, Phathom Pharmaceuticals, Medtronic, and others. Naik is a consultant for Sanofi/Regeneron, Eli Lilly and Company, and Renexxion.
A version of this article appeared on Medscape.com.
, according to investigators.
The classification tree offers a practical alternative for evaluating esophagogastric junction (EGJ) outflow disorders when more advanced methods like high-resolution manometry (HRM) or functional lumen imaging probe (FLIP) panometry are unavailable, lead author Ofer Z. Fass, MD, of Northwestern University, Chicago, and colleagues reported.
“[T]here are limited data on normative TBE values,” the investigators wrote in Gastroenterology. “Furthermore, data supporting the accuracy of TBE as a screening test for esophageal motility disorders, as well as clinically relevant test thresholds, remains limited.”
TBE is conventionally interpreted using a handful of single measurements, most often the barium column height at 1, 2, or 5 minutes. Although these metrics are simple to obtain, variability in technique, cutoff values, and interpretation across centers limits reproducibility and weakens diagnostic accuracy, according to the investigators. The role of TBE has therefore been largely confined to adjudicating inconclusive manometry findings, but even in that setting, the absence of validated reference standards constrains its utility as a reliable screening tool.
To address this gap, Fass and colleagues conducted a prospective analysis of 290 patients who underwent TBE at Northwestern Memorial Hospital, Chicago, with HRM and FLIP panometry, interpreted according to the Chicago Classification version 4.0 (CCv4.0), serving as the diagnostic reference standards.
Patients were included if they had both TBE and manometry performed within a short interval, ensuring that the two tests could be meaningfully compared. The study population represented a broad spectrum of esophageal motility presentations, allowing the model to be trained on clinically relevant variation.
Beyond column height, the investigators measured barium height at multiple timepoints, maximal esophageal body width, maximum EGJ diameter, and tablet passage. These variables were incorporated into a recursive partitioning algorithm to build a multimetric classification tree aimed at distinguishing EGJ outflow obstruction from other motility disorders.
The optimal tree incorporated three sequential decision levels. At the top was maximum esophageal body width, followed by EGJ diameter and barium height at the second level, and tablet passage at the third. This stepwise structure allowed the model to refine diagnoses by combining simple, reproducible TBE metrics that are already collected in routine practice.
Among the 290 patients, 121 (42%) had EGJ outflow disorders, 151 (52%) had no outflow disorder, and 18 (6%) had inconclusive manometry findings. Using conventional interpretation with column height and tablet passage, TBE demonstrated a sensitivity of 77.8%, a specificity of 86.0%, and an accuracy of 82.2%. The multimetric classification tree improved diagnostic performance across all parameters, with a sensitivity of 84.2%, a specificity of 92.1%, and an accuracy of 88.3%.
The advantages of multimetric interpretation were most notable in patients with borderline column heights, which single-metric approaches often misclassify, underscoring the value of integrating multiple measurements into a unified model.
“[T]his study demonstrated that TBE can accurately identify achalasia when analyzed using multiple metrics in a classification tree model,” Fass and colleagues wrote. “Future studies should explore the use of TBE metrics and models to identify more specific esophageal motor disorders (such as esophageal spasm and absent contractility), as well as validation in a larger, multicenter cohort.”
Clinical Takeaways
Rishi Naik, MD, of the Center for Swallowing and Esophageal Disorders, Vanderbilt University Medical Center, Nashville, Tennessee, said the study represents a step forward in how clinicians can use a widely accessible esophageal imaging test.
“This study is important in that it has updated the way we use a very common, readily available imaging test and compared it to the current gold standard of HRM and FLIP,” he told GI & Hepatology News. “This provides a practical, standardized framework for clinicians evaluating patients with suspected esophageal motility disorders.”
Naik noted that while HRM and FLIP provide highly detailed information, both carry drawbacks that limit their universal adoption.
“Practically, HRM is a transnasal test that can be cumbersome, and FLIP is performed during a sedated procedure,” he said. “From a comfort and cost perspective, the esophagram outcompetes. What the TBE lacked was adequate sensitivity and specificity when just looking at column height, which is how the authors overcame this by leveraging the comparisons using CCv4.0.”
Implementation, however, requires discipline.
“A timed barium esophagram is a protocol, not a single esophagram,” Naik said. “Without proper measurements, you can’t follow the decision tree.”
Still, he pointed to radiology’s increasing adoption of artificial intelligence (AI) as a promising way forward.
“AI has already transformed radiological reads, and I’m optimistic it will eventually allow us to incorporate not only width, height, and tablet clearance but also 3D [three-dimensional] reconstructions of bolus retention and pressure to enhance predictive modeling,” Naik said.
This study was supported by the Public Health Service.
The investigators disclosed having relationships with Takeda, Phathom Pharmaceuticals, Medtronic, and others. Naik is a consultant for Sanofi/Regeneron, Eli Lilly and Company, and Renexxion.
A version of this article appeared on Medscape.com.
FROM GASTROENTEROLOGY
Medicolegal Concerns in Contemporary Private GI Practice
The need for gastroenterology (GI) services is on the rise in the US, with growing rates of colonoscopy, earlier-onset colon cancer, and inflammatory bowel disease. This increase is taking place in the context of a changing regulatory landscape.
, and to that end, a recent educational practice management update was published in Clinical Gastroenterology and Hepatology by Erin Smith Aebel, JD, a health law specialist with Trenam Law in Tampa, Florida. Aebel has been a speaker at several national GI conferences and has addressed GI trainees on these issues in medical schools.
“Healthcare regulation continues to evolve and it’s a complicated area,” Aebel told GI & Hepatology News. “Some physician investors in healthcare ventures see the potential profits but are not fully aware of how a physician’s license and livelihood could be affected by noncompliance.”
Aebel has seen some medical business owners and institutions pushing physicians to their limits in order to maximize profits. “They’re failing to allow them the meaningful things that allow for a long-term productive and successful practice that provides great patient care,” she said. “A current issue I’m dealing with is employers’ taking away physicians’ administrative time and not respecting the work that is necessary for the physician to be efficient and provide great care,” she said. “If too many physicians get squeezed in this manner, they will eventually walk away from big employers to something they can better control.”
Aebel noted that private-equity acquisitions of medical practices — a fast-growing US trend — are often targeted at quick profits and quick exits, which can be inconsistent with quality long-term patient care. “A question to be asked by physicians and patients is who is benefiting from this transaction?” she said. “Sometimes retired physicians can see a great benefit in private equity, but newer physicians can get tied up with a strong noncompete agreement. The best deals are ones that try to find wins for all involved, including patients.”
Many independent gastroenterologists focusing on the demands of daily practice are less aware than they should be of the legal and business administration sides. “I often get clients who come to me complaining about their contracts after they’ve signed them. I don’t have leverage to do as much for them,” she admitted.
From a business standpoint, gastroenterologists need to understand where they can negotiate for financial gain and control. These could relate to compensation and bonuses, as well as opportunities to invest in the practice, the practice management company, and possibly real estate or ambulatory surgery centers (ASCs).
Aebel’s overarching messages to gastroenterologists are as follows: “Be aware. Learn basic health law. Read your contracts before you sign them. And invest in good counsel before you sign agreements,” she said. “In addition, GI practitioners need to have a working knowledge of the federal Anti-Kickback Statute and the federal Stark Law and how they could be commonly applied in their practices.”
These are designed to protect government-funded patient care from monetary influence. The False Claims Act is another federal buttress against fraud and abuse, she said.
Update Details
Though not intended to be legal advice, Aebel’s update touches on several important medicolegal areas.
Stark Law on Self-Referrals
Gastroenterologists should be familiar with this federal law, a self-referral civil penalty statute regulating how physicians can pay themselves in practices that provide designated health services covered by federal healthcare programs such as Medicare or Medicaid.
For a Stark penalty to apply, there must be a physician referral to an entity (eg, lab, hospital, nutrition service, physiotherapy or radiotherapy center) in which the physician or a close family member has a financial interest.
Ambulatory Surgery Centers
Another common area vulnerable to federal fraud and abuse regulation is investment in ASCs. “Generally speaking, it is a felony to pay or be paid anything of value for Medicare or Medicaid business referrals,” Aebel wrote. This provision relates to the general restriction of the federal AKB statute.
A gastroenterologist referring Medicare patients to a center where that physician has an investment could technically violate this law because the physician will receive profit distributions from the referral. In addition to constituting a felony with potential jail time, violation of this statute is grounds for substantial civil monetary penalties and/or exclusion from the government coverage program.
Fortunately, Aebel noted, legal safe harbors cover many financial relationships, including investment in an ASC. The financial arrangement is protected from prosecution if it meets five safe harbor requirements, including nondiscriminatory treatment of government-insured patients and physician investment unrelated to a center’s volume or the value of referrals. If even one aspect is not met, that will automatically constitute a crime.
“However, the government will look at facts and circumstances to determine whether there was an intent to pay for a referral,” Aebel wrote.
The safe harbor designates requirements for four types of ASCs: surgeon-owned, single-specialty, multispecialty, and hospital/physician ASCs.
Private-Equity Investment
With mergers and acquisitions of US medical practices and networks by private-equity firms becoming more common, gastroenterologists need to be aware of the legal issues involved in such investment.
Most states abide by corporate practice of medicine doctrines, which prohibit unlicensed people from direct ownership in a medical practice. These doctrines vary by state, but their primary goal is to ensure that medical decisions are made solely based on patient care and not influenced by corporate interests. The aim is to shield the physician-patient relationship from commercial influence.
“Accordingly, this creates additional complicated structures necessary for private-equity investment in gastroenterology practices,” Aebel wrote. Usually, such investors will invest in a management services organization (MSO), which takes much of the practice’s value via management fees. Gastroenterologists may or may not have an opportunity to invest in the practice and the MSO in this scenario.
Under corporate practice of medicine doctrine, physicians must control the clinical aspects of patient care. Therefore, some states may have restrictions on private-equity companies’ control of the use of medical devices, pricing, medical protocols, or other issues of patient care.
“This needs to be considered when reviewing the investment documents and structural documents proposed by private equity companies,” the advisory stated. From a business standpoint, gastroenterologists need to understand where they can negotiate for financial gain and control over their clinical practice. “This could relate to their compensation, bonuses, and investment opportunities in the practice, the practice management company, and possibly real estate or ASCs.”
Offering a gastroenterologist’s perspective on the paper, Camille Thélin, MD, MSc, an associate professor in the Division of Digestive Diseases and Health at the University of South Florida, Tampa, Florida, who also practices privately, said, that “what Erin Aebel reminds us is that the business side of GI can be just as tricky as the clinical side. Ancillary services like capsule studies or office labs fall under strict Stark rules, ASC ownership has Anti-Kickback Law restrictions, and private-equity deals may affect both your paycheck and your autonomy.”
Thélin’s main takeaway advice is that business opportunities can be valuable but carry real legal risks if not structured correctly. “This isn’t just abstract compliance law — it’s about protecting one’s ability to practice medicine, earn fairly, and avoid devastating penalties,” she told GI & Hepatology News. “This article reinforces the need for proactive legal review and careful structuring of business arrangements so physicians can focus on patient care without stumbling into avoidable legal pitfalls. With the right legal structure, ancillaries, ASCs, and private equity can strengthen your GI practice without risking compliance.”
The bottom line, said Aebel, is that gastroenterologists already in private practice or considering entering one must navigate a complex landscape of compliance and regulatory requirements — particularly when providing ancillary services, investing in ASCs, or engaging with private equity.
Understanding the Stark law, the AKB statute, and the intricacies of private-equity investment is essential to mitigate risks and avoid severe penalties, the advisory stressed. By proactively seeking expert legal and business guidance, gastroenterologists can structure their financial and ownership arrangements in a compliant manner, safeguarding their practices while capitalizing on growth opportunities.
This paper listed no external funding. Neither Aebel nor Thélin had any relevant conflicts of interest.
A version of this article appeared on Medscape.com.
The need for gastroenterology (GI) services is on the rise in the US, with growing rates of colonoscopy, earlier-onset colon cancer, and inflammatory bowel disease. This increase is taking place in the context of a changing regulatory landscape.
, and to that end, a recent educational practice management update was published in Clinical Gastroenterology and Hepatology by Erin Smith Aebel, JD, a health law specialist with Trenam Law in Tampa, Florida. Aebel has been a speaker at several national GI conferences and has addressed GI trainees on these issues in medical schools.
“Healthcare regulation continues to evolve and it’s a complicated area,” Aebel told GI & Hepatology News. “Some physician investors in healthcare ventures see the potential profits but are not fully aware of how a physician’s license and livelihood could be affected by noncompliance.”
Aebel has seen some medical business owners and institutions pushing physicians to their limits in order to maximize profits. “They’re failing to allow them the meaningful things that allow for a long-term productive and successful practice that provides great patient care,” she said. “A current issue I’m dealing with is employers’ taking away physicians’ administrative time and not respecting the work that is necessary for the physician to be efficient and provide great care,” she said. “If too many physicians get squeezed in this manner, they will eventually walk away from big employers to something they can better control.”
Aebel noted that private-equity acquisitions of medical practices — a fast-growing US trend — are often targeted at quick profits and quick exits, which can be inconsistent with quality long-term patient care. “A question to be asked by physicians and patients is who is benefiting from this transaction?” she said. “Sometimes retired physicians can see a great benefit in private equity, but newer physicians can get tied up with a strong noncompete agreement. The best deals are ones that try to find wins for all involved, including patients.”
Many independent gastroenterologists focusing on the demands of daily practice are less aware than they should be of the legal and business administration sides. “I often get clients who come to me complaining about their contracts after they’ve signed them. I don’t have leverage to do as much for them,” she admitted.
From a business standpoint, gastroenterologists need to understand where they can negotiate for financial gain and control. These could relate to compensation and bonuses, as well as opportunities to invest in the practice, the practice management company, and possibly real estate or ambulatory surgery centers (ASCs).
Aebel’s overarching messages to gastroenterologists are as follows: “Be aware. Learn basic health law. Read your contracts before you sign them. And invest in good counsel before you sign agreements,” she said. “In addition, GI practitioners need to have a working knowledge of the federal Anti-Kickback Statute and the federal Stark Law and how they could be commonly applied in their practices.”
These are designed to protect government-funded patient care from monetary influence. The False Claims Act is another federal buttress against fraud and abuse, she said.
Update Details
Though not intended to be legal advice, Aebel’s update touches on several important medicolegal areas.
Stark Law on Self-Referrals
Gastroenterologists should be familiar with this federal law, a self-referral civil penalty statute regulating how physicians can pay themselves in practices that provide designated health services covered by federal healthcare programs such as Medicare or Medicaid.
For a Stark penalty to apply, there must be a physician referral to an entity (eg, lab, hospital, nutrition service, physiotherapy or radiotherapy center) in which the physician or a close family member has a financial interest.
Ambulatory Surgery Centers
Another common area vulnerable to federal fraud and abuse regulation is investment in ASCs. “Generally speaking, it is a felony to pay or be paid anything of value for Medicare or Medicaid business referrals,” Aebel wrote. This provision relates to the general restriction of the federal AKB statute.
A gastroenterologist referring Medicare patients to a center where that physician has an investment could technically violate this law because the physician will receive profit distributions from the referral. In addition to constituting a felony with potential jail time, violation of this statute is grounds for substantial civil monetary penalties and/or exclusion from the government coverage program.
Fortunately, Aebel noted, legal safe harbors cover many financial relationships, including investment in an ASC. The financial arrangement is protected from prosecution if it meets five safe harbor requirements, including nondiscriminatory treatment of government-insured patients and physician investment unrelated to a center’s volume or the value of referrals. If even one aspect is not met, that will automatically constitute a crime.
“However, the government will look at facts and circumstances to determine whether there was an intent to pay for a referral,” Aebel wrote.
The safe harbor designates requirements for four types of ASCs: surgeon-owned, single-specialty, multispecialty, and hospital/physician ASCs.
Private-Equity Investment
With mergers and acquisitions of US medical practices and networks by private-equity firms becoming more common, gastroenterologists need to be aware of the legal issues involved in such investment.
Most states abide by corporate practice of medicine doctrines, which prohibit unlicensed people from direct ownership in a medical practice. These doctrines vary by state, but their primary goal is to ensure that medical decisions are made solely based on patient care and not influenced by corporate interests. The aim is to shield the physician-patient relationship from commercial influence.
“Accordingly, this creates additional complicated structures necessary for private-equity investment in gastroenterology practices,” Aebel wrote. Usually, such investors will invest in a management services organization (MSO), which takes much of the practice’s value via management fees. Gastroenterologists may or may not have an opportunity to invest in the practice and the MSO in this scenario.
Under corporate practice of medicine doctrine, physicians must control the clinical aspects of patient care. Therefore, some states may have restrictions on private-equity companies’ control of the use of medical devices, pricing, medical protocols, or other issues of patient care.
“This needs to be considered when reviewing the investment documents and structural documents proposed by private equity companies,” the advisory stated. From a business standpoint, gastroenterologists need to understand where they can negotiate for financial gain and control over their clinical practice. “This could relate to their compensation, bonuses, and investment opportunities in the practice, the practice management company, and possibly real estate or ASCs.”
Offering a gastroenterologist’s perspective on the paper, Camille Thélin, MD, MSc, an associate professor in the Division of Digestive Diseases and Health at the University of South Florida, Tampa, Florida, who also practices privately, said, that “what Erin Aebel reminds us is that the business side of GI can be just as tricky as the clinical side. Ancillary services like capsule studies or office labs fall under strict Stark rules, ASC ownership has Anti-Kickback Law restrictions, and private-equity deals may affect both your paycheck and your autonomy.”
Thélin’s main takeaway advice is that business opportunities can be valuable but carry real legal risks if not structured correctly. “This isn’t just abstract compliance law — it’s about protecting one’s ability to practice medicine, earn fairly, and avoid devastating penalties,” she told GI & Hepatology News. “This article reinforces the need for proactive legal review and careful structuring of business arrangements so physicians can focus on patient care without stumbling into avoidable legal pitfalls. With the right legal structure, ancillaries, ASCs, and private equity can strengthen your GI practice without risking compliance.”
The bottom line, said Aebel, is that gastroenterologists already in private practice or considering entering one must navigate a complex landscape of compliance and regulatory requirements — particularly when providing ancillary services, investing in ASCs, or engaging with private equity.
Understanding the Stark law, the AKB statute, and the intricacies of private-equity investment is essential to mitigate risks and avoid severe penalties, the advisory stressed. By proactively seeking expert legal and business guidance, gastroenterologists can structure their financial and ownership arrangements in a compliant manner, safeguarding their practices while capitalizing on growth opportunities.
This paper listed no external funding. Neither Aebel nor Thélin had any relevant conflicts of interest.
A version of this article appeared on Medscape.com.
The need for gastroenterology (GI) services is on the rise in the US, with growing rates of colonoscopy, earlier-onset colon cancer, and inflammatory bowel disease. This increase is taking place in the context of a changing regulatory landscape.
, and to that end, a recent educational practice management update was published in Clinical Gastroenterology and Hepatology by Erin Smith Aebel, JD, a health law specialist with Trenam Law in Tampa, Florida. Aebel has been a speaker at several national GI conferences and has addressed GI trainees on these issues in medical schools.
“Healthcare regulation continues to evolve and it’s a complicated area,” Aebel told GI & Hepatology News. “Some physician investors in healthcare ventures see the potential profits but are not fully aware of how a physician’s license and livelihood could be affected by noncompliance.”
Aebel has seen some medical business owners and institutions pushing physicians to their limits in order to maximize profits. “They’re failing to allow them the meaningful things that allow for a long-term productive and successful practice that provides great patient care,” she said. “A current issue I’m dealing with is employers’ taking away physicians’ administrative time and not respecting the work that is necessary for the physician to be efficient and provide great care,” she said. “If too many physicians get squeezed in this manner, they will eventually walk away from big employers to something they can better control.”
Aebel noted that private-equity acquisitions of medical practices — a fast-growing US trend — are often targeted at quick profits and quick exits, which can be inconsistent with quality long-term patient care. “A question to be asked by physicians and patients is who is benefiting from this transaction?” she said. “Sometimes retired physicians can see a great benefit in private equity, but newer physicians can get tied up with a strong noncompete agreement. The best deals are ones that try to find wins for all involved, including patients.”
Many independent gastroenterologists focusing on the demands of daily practice are less aware than they should be of the legal and business administration sides. “I often get clients who come to me complaining about their contracts after they’ve signed them. I don’t have leverage to do as much for them,” she admitted.
From a business standpoint, gastroenterologists need to understand where they can negotiate for financial gain and control. These could relate to compensation and bonuses, as well as opportunities to invest in the practice, the practice management company, and possibly real estate or ambulatory surgery centers (ASCs).
Aebel’s overarching messages to gastroenterologists are as follows: “Be aware. Learn basic health law. Read your contracts before you sign them. And invest in good counsel before you sign agreements,” she said. “In addition, GI practitioners need to have a working knowledge of the federal Anti-Kickback Statute and the federal Stark Law and how they could be commonly applied in their practices.”
These are designed to protect government-funded patient care from monetary influence. The False Claims Act is another federal buttress against fraud and abuse, she said.
Update Details
Though not intended to be legal advice, Aebel’s update touches on several important medicolegal areas.
Stark Law on Self-Referrals
Gastroenterologists should be familiar with this federal law, a self-referral civil penalty statute regulating how physicians can pay themselves in practices that provide designated health services covered by federal healthcare programs such as Medicare or Medicaid.
For a Stark penalty to apply, there must be a physician referral to an entity (eg, lab, hospital, nutrition service, physiotherapy or radiotherapy center) in which the physician or a close family member has a financial interest.
Ambulatory Surgery Centers
Another common area vulnerable to federal fraud and abuse regulation is investment in ASCs. “Generally speaking, it is a felony to pay or be paid anything of value for Medicare or Medicaid business referrals,” Aebel wrote. This provision relates to the general restriction of the federal AKB statute.
A gastroenterologist referring Medicare patients to a center where that physician has an investment could technically violate this law because the physician will receive profit distributions from the referral. In addition to constituting a felony with potential jail time, violation of this statute is grounds for substantial civil monetary penalties and/or exclusion from the government coverage program.
Fortunately, Aebel noted, legal safe harbors cover many financial relationships, including investment in an ASC. The financial arrangement is protected from prosecution if it meets five safe harbor requirements, including nondiscriminatory treatment of government-insured patients and physician investment unrelated to a center’s volume or the value of referrals. If even one aspect is not met, that will automatically constitute a crime.
“However, the government will look at facts and circumstances to determine whether there was an intent to pay for a referral,” Aebel wrote.
The safe harbor designates requirements for four types of ASCs: surgeon-owned, single-specialty, multispecialty, and hospital/physician ASCs.
Private-Equity Investment
With mergers and acquisitions of US medical practices and networks by private-equity firms becoming more common, gastroenterologists need to be aware of the legal issues involved in such investment.
Most states abide by corporate practice of medicine doctrines, which prohibit unlicensed people from direct ownership in a medical practice. These doctrines vary by state, but their primary goal is to ensure that medical decisions are made solely based on patient care and not influenced by corporate interests. The aim is to shield the physician-patient relationship from commercial influence.
“Accordingly, this creates additional complicated structures necessary for private-equity investment in gastroenterology practices,” Aebel wrote. Usually, such investors will invest in a management services organization (MSO), which takes much of the practice’s value via management fees. Gastroenterologists may or may not have an opportunity to invest in the practice and the MSO in this scenario.
Under corporate practice of medicine doctrine, physicians must control the clinical aspects of patient care. Therefore, some states may have restrictions on private-equity companies’ control of the use of medical devices, pricing, medical protocols, or other issues of patient care.
“This needs to be considered when reviewing the investment documents and structural documents proposed by private equity companies,” the advisory stated. From a business standpoint, gastroenterologists need to understand where they can negotiate for financial gain and control over their clinical practice. “This could relate to their compensation, bonuses, and investment opportunities in the practice, the practice management company, and possibly real estate or ASCs.”
Offering a gastroenterologist’s perspective on the paper, Camille Thélin, MD, MSc, an associate professor in the Division of Digestive Diseases and Health at the University of South Florida, Tampa, Florida, who also practices privately, said, that “what Erin Aebel reminds us is that the business side of GI can be just as tricky as the clinical side. Ancillary services like capsule studies or office labs fall under strict Stark rules, ASC ownership has Anti-Kickback Law restrictions, and private-equity deals may affect both your paycheck and your autonomy.”
Thélin’s main takeaway advice is that business opportunities can be valuable but carry real legal risks if not structured correctly. “This isn’t just abstract compliance law — it’s about protecting one’s ability to practice medicine, earn fairly, and avoid devastating penalties,” she told GI & Hepatology News. “This article reinforces the need for proactive legal review and careful structuring of business arrangements so physicians can focus on patient care without stumbling into avoidable legal pitfalls. With the right legal structure, ancillaries, ASCs, and private equity can strengthen your GI practice without risking compliance.”
The bottom line, said Aebel, is that gastroenterologists already in private practice or considering entering one must navigate a complex landscape of compliance and regulatory requirements — particularly when providing ancillary services, investing in ASCs, or engaging with private equity.
Understanding the Stark law, the AKB statute, and the intricacies of private-equity investment is essential to mitigate risks and avoid severe penalties, the advisory stressed. By proactively seeking expert legal and business guidance, gastroenterologists can structure their financial and ownership arrangements in a compliant manner, safeguarding their practices while capitalizing on growth opportunities.
This paper listed no external funding. Neither Aebel nor Thélin had any relevant conflicts of interest.
A version of this article appeared on Medscape.com.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Fecal Transplant Benefits in Primary C Difficile Infection Similar to Vancomycin
, with efficacy that is comparable to the standard treatment of vancomycin, and in some measures, showing even stronger efficacy, new research showed.
“FMT, prepared and administered according to international guidelines, is an effective and safe treatment option for C difficile infections, which should be considered for all patients with the infection,” first author Frederik Emil Juul, MD, PhD, of the Clinical Effectiveness Research Group, University of Oslo, in Oslo, Norway, told GI & Hepatology News.
FMT even showed a numerical superiority to vancomycin, which, though not statistically significant, “indicates that FMT has the potential to change the current practice of antibiotic therapy and may establish FMT as a first-line treatment for primary CDI,” the authors further asserted in the study, published recently in the Annals of Internal Medicine.
In the treatment of antibiotic-associated colitis due to CDI, vancomycin or fidaxomicin are the standard therapies, yet up to 20% of patients experience one or more symptom recurrences following successful initial antibiotic treatment, prompting the need for continued antibiotic regimens, resulting in increased costs and potential adverse events, while contributing to antibiotic resistance.
FMT, designed to restore a normal functional colonic microenvironment with the transfer of a healthy person’s stool, though still somewhat controversial, has gained acceptance and favor in recent years in the treatment of recurrent CDI, however, research has been lacking on its efficacy in the treatment of primary CDI.
With a previous proof-of-concept trial and observational study showing promising results in primary CDI, Juul and colleagues conducted the current randomized, open-label noninferiority trial.
For the multi-center study, 100 adult patients with CDI, defined as C diff toxin in stool and at least three loose stools daily, and no previous CDI within 1 year prior to enrollment, were randomized at 20 hospitals in Norway to receive either FMT, administered as an enema, without antibiotic pretreatment, or oral vancomycin at a dose of 125 mg, four times daily for 10 days.
The patients had a median age of about 70 years; more than 40% of patients had a Charlson Comorbidity Index score of ≥ 4, indicating severe comorbidity, and a third had severe CDI.
With the trial showing favorable results, a data and safety monitoring board recommended stopping the trial for efficacy and noninferiority after about half of the planned enrollment was reached.
The primary endpoint of a clinical cure, defined as firm stools or less than three bowel movements daily and no disease recurrence within 60 days without additional treatment, was observed in 34 of 51 patients who received FMT (66.7%) compared with 30 of 49 of those receiving vancomycin (61.2%; difference, 5.4 percentage points; P for noninferiority < .001).
The results contradict the theory that response to FMT is 25 percentage points lower than response to vancomycin, the authors noted.
The proportion of patients with clinical cure at day 14 was 70.6% in the FMT group and 77.6% in the vancomycin group, and among those patients, two (5.6%) in the FMT group had disease recurrence compared with eight (21.1%) in the vancomycin group between days 15 and 60.
In the FMT group, 11 patients received additional treatment compared with four in the vancomycin group, predominantly oral vancomycin in both groups.
Despite the high rates of severe comorbidity among the patients at baseline, a subgroup analyses showed no significant differences in treatment effects based on factors including sex, age group, Charlson Comorbidity Index score, or CDI severity.
Importantly, there were also no significant differences in adverse events between the groups.
“Our results indicate that it is reasonable to treat patients with primary CDI with FMT and provide antibiotics only to patients with ongoing symptoms or recurrence after FMT,” the authors concluded.
FMT Faces Challenges in the US
FMT specifically consists of direct instillation of fecal matter to the upper gastrointestinal tract, via capsules or duodenal infusion, or the lower gastrointestinal tract via colonoscopy or enema.
While an AGA guideline issued in 2024 endorsed FMT for the prevention of recurrent, refractory, or fulminant CDI in select adults not responding to standard antibiotics, the association underscored important caveats, including a low quality of evidence, and concluded that FMT could not yet be recommended for other gastrointestinal conditions.
The treatment meanwhile has faced an uphill battle in the US. The provision of screened FMT inocula through the nonprofit OpenBiome, previously the country’s largest stool bank, was recently suspended amid FDA policy changes.
And while other commercial-grade biotherapeutic products Rebyota and Vowst, have received FDA approval, cost and insurance coverage can be significant barriers, said Elizabeth Hohmann, MD, of the Infectious Disease Division at Massachusetts General Hospital, Boston, in an editorial published with the study.
“Currently approved options are expensive and are not available to many who might benefit for various reasons, primarily cost,” she said.
Acceptance Higher in Europe
In Europe, and particularly Norway, acceptance of FMT for CDI and other indications has been more favorable, and while regulation of the treatment has varied among European countries, a new regulation to be implemented by the European Union in 2027 will improve standardization of the production, handling, storage, and other factors of FMT, Juul told GI & Hepatology News.
“I believe the new regulations will make the treatment more available to patients, and a standardization of the FMT production will make future trials more comparable and useful across countries,” he said.
Juul said he further expects that “our results will lower the threshold for choosing FMT as treatment in primary infections. I know that Denmark also gives FMT to patients with primary CDI.”
Quality of Life
Hohmann, who has treated many patients with recurrent CDI with FMT, noted that a key factor that should be underscored is how much better patients can feel after the treatment.
“Although there are no quality of life surveys in [the current study], had they been done, I suspect quality of life might have been higher in the FMT group; in my experience, people feel better after microbiome restoration.”
She added that her patients “report feeling much better, and that’s why I keep doing it,” she said. “I’ve had an 80-year-old patient tell me he’s going back to snow shoveling; another saying she can return to yoga classes.”
“When you have had bad gut microbiome dysbiosis that becomes normal, you feel a lot better,” Hohmann said.
In the treatment of primary CDI, however, Hohmann said the prospects, at least in the US, are likely slim.
“I do not believe that we in the United States will see FMT as a primary treatment of C difficile infection anytime soon,” she wrote in the editorial.
Nevertheless, Hohmann asserted that “FMT should remain available, with appropriate sources of carefully screened inocula for care and for further research into the many illnesses and therapies that are influenced by the health of the gut microbiome.”
This study received funding from the South-East Norway Health Trust. Hohmann had no disclosures to report.
A version of this article appeared on Medscape.com.
, with efficacy that is comparable to the standard treatment of vancomycin, and in some measures, showing even stronger efficacy, new research showed.
“FMT, prepared and administered according to international guidelines, is an effective and safe treatment option for C difficile infections, which should be considered for all patients with the infection,” first author Frederik Emil Juul, MD, PhD, of the Clinical Effectiveness Research Group, University of Oslo, in Oslo, Norway, told GI & Hepatology News.
FMT even showed a numerical superiority to vancomycin, which, though not statistically significant, “indicates that FMT has the potential to change the current practice of antibiotic therapy and may establish FMT as a first-line treatment for primary CDI,” the authors further asserted in the study, published recently in the Annals of Internal Medicine.
In the treatment of antibiotic-associated colitis due to CDI, vancomycin or fidaxomicin are the standard therapies, yet up to 20% of patients experience one or more symptom recurrences following successful initial antibiotic treatment, prompting the need for continued antibiotic regimens, resulting in increased costs and potential adverse events, while contributing to antibiotic resistance.
FMT, designed to restore a normal functional colonic microenvironment with the transfer of a healthy person’s stool, though still somewhat controversial, has gained acceptance and favor in recent years in the treatment of recurrent CDI, however, research has been lacking on its efficacy in the treatment of primary CDI.
With a previous proof-of-concept trial and observational study showing promising results in primary CDI, Juul and colleagues conducted the current randomized, open-label noninferiority trial.
For the multi-center study, 100 adult patients with CDI, defined as C diff toxin in stool and at least three loose stools daily, and no previous CDI within 1 year prior to enrollment, were randomized at 20 hospitals in Norway to receive either FMT, administered as an enema, without antibiotic pretreatment, or oral vancomycin at a dose of 125 mg, four times daily for 10 days.
The patients had a median age of about 70 years; more than 40% of patients had a Charlson Comorbidity Index score of ≥ 4, indicating severe comorbidity, and a third had severe CDI.
With the trial showing favorable results, a data and safety monitoring board recommended stopping the trial for efficacy and noninferiority after about half of the planned enrollment was reached.
The primary endpoint of a clinical cure, defined as firm stools or less than three bowel movements daily and no disease recurrence within 60 days without additional treatment, was observed in 34 of 51 patients who received FMT (66.7%) compared with 30 of 49 of those receiving vancomycin (61.2%; difference, 5.4 percentage points; P for noninferiority < .001).
The results contradict the theory that response to FMT is 25 percentage points lower than response to vancomycin, the authors noted.
The proportion of patients with clinical cure at day 14 was 70.6% in the FMT group and 77.6% in the vancomycin group, and among those patients, two (5.6%) in the FMT group had disease recurrence compared with eight (21.1%) in the vancomycin group between days 15 and 60.
In the FMT group, 11 patients received additional treatment compared with four in the vancomycin group, predominantly oral vancomycin in both groups.
Despite the high rates of severe comorbidity among the patients at baseline, a subgroup analyses showed no significant differences in treatment effects based on factors including sex, age group, Charlson Comorbidity Index score, or CDI severity.
Importantly, there were also no significant differences in adverse events between the groups.
“Our results indicate that it is reasonable to treat patients with primary CDI with FMT and provide antibiotics only to patients with ongoing symptoms or recurrence after FMT,” the authors concluded.
FMT Faces Challenges in the US
FMT specifically consists of direct instillation of fecal matter to the upper gastrointestinal tract, via capsules or duodenal infusion, or the lower gastrointestinal tract via colonoscopy or enema.
While an AGA guideline issued in 2024 endorsed FMT for the prevention of recurrent, refractory, or fulminant CDI in select adults not responding to standard antibiotics, the association underscored important caveats, including a low quality of evidence, and concluded that FMT could not yet be recommended for other gastrointestinal conditions.
The treatment meanwhile has faced an uphill battle in the US. The provision of screened FMT inocula through the nonprofit OpenBiome, previously the country’s largest stool bank, was recently suspended amid FDA policy changes.
And while other commercial-grade biotherapeutic products Rebyota and Vowst, have received FDA approval, cost and insurance coverage can be significant barriers, said Elizabeth Hohmann, MD, of the Infectious Disease Division at Massachusetts General Hospital, Boston, in an editorial published with the study.
“Currently approved options are expensive and are not available to many who might benefit for various reasons, primarily cost,” she said.
Acceptance Higher in Europe
In Europe, and particularly Norway, acceptance of FMT for CDI and other indications has been more favorable, and while regulation of the treatment has varied among European countries, a new regulation to be implemented by the European Union in 2027 will improve standardization of the production, handling, storage, and other factors of FMT, Juul told GI & Hepatology News.
“I believe the new regulations will make the treatment more available to patients, and a standardization of the FMT production will make future trials more comparable and useful across countries,” he said.
Juul said he further expects that “our results will lower the threshold for choosing FMT as treatment in primary infections. I know that Denmark also gives FMT to patients with primary CDI.”
Quality of Life
Hohmann, who has treated many patients with recurrent CDI with FMT, noted that a key factor that should be underscored is how much better patients can feel after the treatment.
“Although there are no quality of life surveys in [the current study], had they been done, I suspect quality of life might have been higher in the FMT group; in my experience, people feel better after microbiome restoration.”
She added that her patients “report feeling much better, and that’s why I keep doing it,” she said. “I’ve had an 80-year-old patient tell me he’s going back to snow shoveling; another saying she can return to yoga classes.”
“When you have had bad gut microbiome dysbiosis that becomes normal, you feel a lot better,” Hohmann said.
In the treatment of primary CDI, however, Hohmann said the prospects, at least in the US, are likely slim.
“I do not believe that we in the United States will see FMT as a primary treatment of C difficile infection anytime soon,” she wrote in the editorial.
Nevertheless, Hohmann asserted that “FMT should remain available, with appropriate sources of carefully screened inocula for care and for further research into the many illnesses and therapies that are influenced by the health of the gut microbiome.”
This study received funding from the South-East Norway Health Trust. Hohmann had no disclosures to report.
A version of this article appeared on Medscape.com.
, with efficacy that is comparable to the standard treatment of vancomycin, and in some measures, showing even stronger efficacy, new research showed.
“FMT, prepared and administered according to international guidelines, is an effective and safe treatment option for C difficile infections, which should be considered for all patients with the infection,” first author Frederik Emil Juul, MD, PhD, of the Clinical Effectiveness Research Group, University of Oslo, in Oslo, Norway, told GI & Hepatology News.
FMT even showed a numerical superiority to vancomycin, which, though not statistically significant, “indicates that FMT has the potential to change the current practice of antibiotic therapy and may establish FMT as a first-line treatment for primary CDI,” the authors further asserted in the study, published recently in the Annals of Internal Medicine.
In the treatment of antibiotic-associated colitis due to CDI, vancomycin or fidaxomicin are the standard therapies, yet up to 20% of patients experience one or more symptom recurrences following successful initial antibiotic treatment, prompting the need for continued antibiotic regimens, resulting in increased costs and potential adverse events, while contributing to antibiotic resistance.
FMT, designed to restore a normal functional colonic microenvironment with the transfer of a healthy person’s stool, though still somewhat controversial, has gained acceptance and favor in recent years in the treatment of recurrent CDI, however, research has been lacking on its efficacy in the treatment of primary CDI.
With a previous proof-of-concept trial and observational study showing promising results in primary CDI, Juul and colleagues conducted the current randomized, open-label noninferiority trial.
For the multi-center study, 100 adult patients with CDI, defined as C diff toxin in stool and at least three loose stools daily, and no previous CDI within 1 year prior to enrollment, were randomized at 20 hospitals in Norway to receive either FMT, administered as an enema, without antibiotic pretreatment, or oral vancomycin at a dose of 125 mg, four times daily for 10 days.
The patients had a median age of about 70 years; more than 40% of patients had a Charlson Comorbidity Index score of ≥ 4, indicating severe comorbidity, and a third had severe CDI.
With the trial showing favorable results, a data and safety monitoring board recommended stopping the trial for efficacy and noninferiority after about half of the planned enrollment was reached.
The primary endpoint of a clinical cure, defined as firm stools or less than three bowel movements daily and no disease recurrence within 60 days without additional treatment, was observed in 34 of 51 patients who received FMT (66.7%) compared with 30 of 49 of those receiving vancomycin (61.2%; difference, 5.4 percentage points; P for noninferiority < .001).
The results contradict the theory that response to FMT is 25 percentage points lower than response to vancomycin, the authors noted.
The proportion of patients with clinical cure at day 14 was 70.6% in the FMT group and 77.6% in the vancomycin group, and among those patients, two (5.6%) in the FMT group had disease recurrence compared with eight (21.1%) in the vancomycin group between days 15 and 60.
In the FMT group, 11 patients received additional treatment compared with four in the vancomycin group, predominantly oral vancomycin in both groups.
Despite the high rates of severe comorbidity among the patients at baseline, a subgroup analyses showed no significant differences in treatment effects based on factors including sex, age group, Charlson Comorbidity Index score, or CDI severity.
Importantly, there were also no significant differences in adverse events between the groups.
“Our results indicate that it is reasonable to treat patients with primary CDI with FMT and provide antibiotics only to patients with ongoing symptoms or recurrence after FMT,” the authors concluded.
FMT Faces Challenges in the US
FMT specifically consists of direct instillation of fecal matter to the upper gastrointestinal tract, via capsules or duodenal infusion, or the lower gastrointestinal tract via colonoscopy or enema.
While an AGA guideline issued in 2024 endorsed FMT for the prevention of recurrent, refractory, or fulminant CDI in select adults not responding to standard antibiotics, the association underscored important caveats, including a low quality of evidence, and concluded that FMT could not yet be recommended for other gastrointestinal conditions.
The treatment meanwhile has faced an uphill battle in the US. The provision of screened FMT inocula through the nonprofit OpenBiome, previously the country’s largest stool bank, was recently suspended amid FDA policy changes.
And while other commercial-grade biotherapeutic products Rebyota and Vowst, have received FDA approval, cost and insurance coverage can be significant barriers, said Elizabeth Hohmann, MD, of the Infectious Disease Division at Massachusetts General Hospital, Boston, in an editorial published with the study.
“Currently approved options are expensive and are not available to many who might benefit for various reasons, primarily cost,” she said.
Acceptance Higher in Europe
In Europe, and particularly Norway, acceptance of FMT for CDI and other indications has been more favorable, and while regulation of the treatment has varied among European countries, a new regulation to be implemented by the European Union in 2027 will improve standardization of the production, handling, storage, and other factors of FMT, Juul told GI & Hepatology News.
“I believe the new regulations will make the treatment more available to patients, and a standardization of the FMT production will make future trials more comparable and useful across countries,” he said.
Juul said he further expects that “our results will lower the threshold for choosing FMT as treatment in primary infections. I know that Denmark also gives FMT to patients with primary CDI.”
Quality of Life
Hohmann, who has treated many patients with recurrent CDI with FMT, noted that a key factor that should be underscored is how much better patients can feel after the treatment.
“Although there are no quality of life surveys in [the current study], had they been done, I suspect quality of life might have been higher in the FMT group; in my experience, people feel better after microbiome restoration.”
She added that her patients “report feeling much better, and that’s why I keep doing it,” she said. “I’ve had an 80-year-old patient tell me he’s going back to snow shoveling; another saying she can return to yoga classes.”
“When you have had bad gut microbiome dysbiosis that becomes normal, you feel a lot better,” Hohmann said.
In the treatment of primary CDI, however, Hohmann said the prospects, at least in the US, are likely slim.
“I do not believe that we in the United States will see FMT as a primary treatment of C difficile infection anytime soon,” she wrote in the editorial.
Nevertheless, Hohmann asserted that “FMT should remain available, with appropriate sources of carefully screened inocula for care and for further research into the many illnesses and therapies that are influenced by the health of the gut microbiome.”
This study received funding from the South-East Norway Health Trust. Hohmann had no disclosures to report.
A version of this article appeared on Medscape.com.
Withdrawing Anti-TNF in IBD Remission: New Data
In the Spanish EXIT study, anti-TNF withdrawal in selected patients with IBD in clinical, endoscopic, and radiological remission had no impact on sustained clinical remission at 1 year, although objective markers of activity were higher in patients who stopped treatment.
The discontinuation of anti-TNF treatment “could be considered as an option” for a selected group of patients, said the authors led by Javier Gisbert, MD, PhD, with Autonomous University of Madrid.
However, the higher proportion of patients with elevated fecal calprotectin and significant endoscopic lesions at the end of follow-up “calls for caution and should be considered when discontinuing treatment in patients,” Gisbert and colleagues concluded.
The EXIT study results were published in the journal Gut (2025 Feb. doi: 10.1136/gutjnl-2024-333385).
Risky Business?
Anti-TNF drugs have reshaped IBD treatment but bring infection risks and costs, prompting interest in planned withdrawal after stable remission.
Yet prior evidence has been mixed. A meta-analysis of 27 studies suggested higher relapse after stopping anti-TNF therapy. However, the results were heterogeneous and most of the studies were retrospective, with a low number of patients and without a control group to compare with.
Clinical trials that have assessed the risk for relapse after discontinuation of anti-TNF therapy generally favored maintenance but had notable limitations.
The EXIT trial was conducted at 33 IBD units across Spain. A total of 140 patients in steroid-free clinical remission for ≥ 6 months on standard-dose infliximab or adalimumab were randomized (1:1) to either continue anti-TNF or switch to placebo matched to the drug they had been taking. All patients continued on immunomodulator therapy.
At 1 year, the proportion of patients with sustained clinical remission (primary outcome) was similar between patients who continued anti-TNF therapy and peers who stopped the medication (76% and 84%, respectively).
However, the proportion of patients with significant endoscopic lesions at the end of follow-up was higher in those who withdrew anti-TNF therapy (19% vs 8.5%; P = .01). Elevated fecal calprotectin (> 250 µg/g) was more common after withdrawal (33% vs 13%; P = .01).
Fecal calprotectin > 250 µg/g at baseline predicted lower odds of sustained remission and higher risk for losing remission — and was the only factor associated with lower likelihood of sustained remission.
Common Clinical Question
“When a patient starts an advanced biologic therapy, they often ask — will I be able to stop it?” Jean-Frederic Colombel, MD, director of the Inflammatory Bowel Disease Clinical Center at the Icahn School of Medicine at Mount Sinai, New York City, who wasn’t involved in the study, told GI & Hepatology News.
Generally speaking, Colombel said he tells patients, “If the drug is working well and you are in deep remission, they should try to avoid stopping because there is a risk of relapse. And with relapse, we never know if the drug will work again and maybe we’ll have to switch to another medication.”
“It’s an individualized discussion and decision and patients who do opt to stop [anti-TNF therapy] need to be monitored closely,” Colombel said.
Colombel cautioned that the study had a relatively short 1-year follow-up and those that stopped anti-TNF therapy had evidence of recurrent inflammation.
“Even though it didn’t translate yet to clinical relapse, there were more patients with subclinical active disease in the group that stopped as compared to the group that continued,” Colombel said.
He also noted that in the SPARE trial of patients with Crohn’s disease in clinical remission, patients who stopped infliximab had a higher risk for relapse compared with patients who stopped azathioprine and those who continued the combination therapy.
The EXIT study was supported by grants from Instituto de Salud Carlos III, Grupo Español de Trabajo en Enfermedad de Crohn y Colitis Ulcerosa and AbbVie. Gisbert reported serving as speaker, consultant, and advisory member for or receiving research funding from MSD, AbbVie, Pfizer, Kern Pharma, Biogen, Mylan, Takeda, Janssen, Roche, Sandoz, Celgene/Bristol Myers, Gilead/Galapagos, Lilly, Ferring, Faes Farma, Shire Pharmaceuticals, Dr. Falk Pharma, Tillotts Pharma, Chiesi, Casen Fleet, Gebro Pharma, Otsuka Pharmaceutical, Norgine and Vifor Pharma. Colombel had no relevant disclosures.
A version of this article appeared on Medscape.com.
In the Spanish EXIT study, anti-TNF withdrawal in selected patients with IBD in clinical, endoscopic, and radiological remission had no impact on sustained clinical remission at 1 year, although objective markers of activity were higher in patients who stopped treatment.
The discontinuation of anti-TNF treatment “could be considered as an option” for a selected group of patients, said the authors led by Javier Gisbert, MD, PhD, with Autonomous University of Madrid.
However, the higher proportion of patients with elevated fecal calprotectin and significant endoscopic lesions at the end of follow-up “calls for caution and should be considered when discontinuing treatment in patients,” Gisbert and colleagues concluded.
The EXIT study results were published in the journal Gut (2025 Feb. doi: 10.1136/gutjnl-2024-333385).
Risky Business?
Anti-TNF drugs have reshaped IBD treatment but bring infection risks and costs, prompting interest in planned withdrawal after stable remission.
Yet prior evidence has been mixed. A meta-analysis of 27 studies suggested higher relapse after stopping anti-TNF therapy. However, the results were heterogeneous and most of the studies were retrospective, with a low number of patients and without a control group to compare with.
Clinical trials that have assessed the risk for relapse after discontinuation of anti-TNF therapy generally favored maintenance but had notable limitations.
The EXIT trial was conducted at 33 IBD units across Spain. A total of 140 patients in steroid-free clinical remission for ≥ 6 months on standard-dose infliximab or adalimumab were randomized (1:1) to either continue anti-TNF or switch to placebo matched to the drug they had been taking. All patients continued on immunomodulator therapy.
At 1 year, the proportion of patients with sustained clinical remission (primary outcome) was similar between patients who continued anti-TNF therapy and peers who stopped the medication (76% and 84%, respectively).
However, the proportion of patients with significant endoscopic lesions at the end of follow-up was higher in those who withdrew anti-TNF therapy (19% vs 8.5%; P = .01). Elevated fecal calprotectin (> 250 µg/g) was more common after withdrawal (33% vs 13%; P = .01).
Fecal calprotectin > 250 µg/g at baseline predicted lower odds of sustained remission and higher risk for losing remission — and was the only factor associated with lower likelihood of sustained remission.
Common Clinical Question
“When a patient starts an advanced biologic therapy, they often ask — will I be able to stop it?” Jean-Frederic Colombel, MD, director of the Inflammatory Bowel Disease Clinical Center at the Icahn School of Medicine at Mount Sinai, New York City, who wasn’t involved in the study, told GI & Hepatology News.
Generally speaking, Colombel said he tells patients, “If the drug is working well and you are in deep remission, they should try to avoid stopping because there is a risk of relapse. And with relapse, we never know if the drug will work again and maybe we’ll have to switch to another medication.”
“It’s an individualized discussion and decision and patients who do opt to stop [anti-TNF therapy] need to be monitored closely,” Colombel said.
Colombel cautioned that the study had a relatively short 1-year follow-up and those that stopped anti-TNF therapy had evidence of recurrent inflammation.
“Even though it didn’t translate yet to clinical relapse, there were more patients with subclinical active disease in the group that stopped as compared to the group that continued,” Colombel said.
He also noted that in the SPARE trial of patients with Crohn’s disease in clinical remission, patients who stopped infliximab had a higher risk for relapse compared with patients who stopped azathioprine and those who continued the combination therapy.
The EXIT study was supported by grants from Instituto de Salud Carlos III, Grupo Español de Trabajo en Enfermedad de Crohn y Colitis Ulcerosa and AbbVie. Gisbert reported serving as speaker, consultant, and advisory member for or receiving research funding from MSD, AbbVie, Pfizer, Kern Pharma, Biogen, Mylan, Takeda, Janssen, Roche, Sandoz, Celgene/Bristol Myers, Gilead/Galapagos, Lilly, Ferring, Faes Farma, Shire Pharmaceuticals, Dr. Falk Pharma, Tillotts Pharma, Chiesi, Casen Fleet, Gebro Pharma, Otsuka Pharmaceutical, Norgine and Vifor Pharma. Colombel had no relevant disclosures.
A version of this article appeared on Medscape.com.
In the Spanish EXIT study, anti-TNF withdrawal in selected patients with IBD in clinical, endoscopic, and radiological remission had no impact on sustained clinical remission at 1 year, although objective markers of activity were higher in patients who stopped treatment.
The discontinuation of anti-TNF treatment “could be considered as an option” for a selected group of patients, said the authors led by Javier Gisbert, MD, PhD, with Autonomous University of Madrid.
However, the higher proportion of patients with elevated fecal calprotectin and significant endoscopic lesions at the end of follow-up “calls for caution and should be considered when discontinuing treatment in patients,” Gisbert and colleagues concluded.
The EXIT study results were published in the journal Gut (2025 Feb. doi: 10.1136/gutjnl-2024-333385).
Risky Business?
Anti-TNF drugs have reshaped IBD treatment but bring infection risks and costs, prompting interest in planned withdrawal after stable remission.
Yet prior evidence has been mixed. A meta-analysis of 27 studies suggested higher relapse after stopping anti-TNF therapy. However, the results were heterogeneous and most of the studies were retrospective, with a low number of patients and without a control group to compare with.
Clinical trials that have assessed the risk for relapse after discontinuation of anti-TNF therapy generally favored maintenance but had notable limitations.
The EXIT trial was conducted at 33 IBD units across Spain. A total of 140 patients in steroid-free clinical remission for ≥ 6 months on standard-dose infliximab or adalimumab were randomized (1:1) to either continue anti-TNF or switch to placebo matched to the drug they had been taking. All patients continued on immunomodulator therapy.
At 1 year, the proportion of patients with sustained clinical remission (primary outcome) was similar between patients who continued anti-TNF therapy and peers who stopped the medication (76% and 84%, respectively).
However, the proportion of patients with significant endoscopic lesions at the end of follow-up was higher in those who withdrew anti-TNF therapy (19% vs 8.5%; P = .01). Elevated fecal calprotectin (> 250 µg/g) was more common after withdrawal (33% vs 13%; P = .01).
Fecal calprotectin > 250 µg/g at baseline predicted lower odds of sustained remission and higher risk for losing remission — and was the only factor associated with lower likelihood of sustained remission.
Common Clinical Question
“When a patient starts an advanced biologic therapy, they often ask — will I be able to stop it?” Jean-Frederic Colombel, MD, director of the Inflammatory Bowel Disease Clinical Center at the Icahn School of Medicine at Mount Sinai, New York City, who wasn’t involved in the study, told GI & Hepatology News.
Generally speaking, Colombel said he tells patients, “If the drug is working well and you are in deep remission, they should try to avoid stopping because there is a risk of relapse. And with relapse, we never know if the drug will work again and maybe we’ll have to switch to another medication.”
“It’s an individualized discussion and decision and patients who do opt to stop [anti-TNF therapy] need to be monitored closely,” Colombel said.
Colombel cautioned that the study had a relatively short 1-year follow-up and those that stopped anti-TNF therapy had evidence of recurrent inflammation.
“Even though it didn’t translate yet to clinical relapse, there were more patients with subclinical active disease in the group that stopped as compared to the group that continued,” Colombel said.
He also noted that in the SPARE trial of patients with Crohn’s disease in clinical remission, patients who stopped infliximab had a higher risk for relapse compared with patients who stopped azathioprine and those who continued the combination therapy.
The EXIT study was supported by grants from Instituto de Salud Carlos III, Grupo Español de Trabajo en Enfermedad de Crohn y Colitis Ulcerosa and AbbVie. Gisbert reported serving as speaker, consultant, and advisory member for or receiving research funding from MSD, AbbVie, Pfizer, Kern Pharma, Biogen, Mylan, Takeda, Janssen, Roche, Sandoz, Celgene/Bristol Myers, Gilead/Galapagos, Lilly, Ferring, Faes Farma, Shire Pharmaceuticals, Dr. Falk Pharma, Tillotts Pharma, Chiesi, Casen Fleet, Gebro Pharma, Otsuka Pharmaceutical, Norgine and Vifor Pharma. Colombel had no relevant disclosures.
A version of this article appeared on Medscape.com.
How Chronic Stress Disrupts the Gut Microbiome
Chronic psychological stress is common. A 2023 survey revealed that about one quarter of US adults reported high stress levels, and three quarters reported that chronic stress affects their daily lives.
Emerging evidence suggests that chronic stress not only exacts a high toll on mental health but also can wreak havoc on all levels of gastrointestinal (GI) functioning, all the way down to the microbiome.
Aasma Shaukat, MD, MPH, AGAF, gastroenterologist with NYU Langone Health and director of GI Outcomes Research, Gastroenterology at NYU Grossman School of Medicine in New York City, said in an interview with GI & Hepatology News.
“This basically means that the normal balance of microorganisms that essentially we think are beneficial gets reduced, and the colonies considered to be more harmful proliferate,” she explained.
What Does the Science Tell Us?
Numerous studies published in the past 5 years have linked chronic stress to modest but reproducible shifts in the composition of the microbiome.
A study of frontline healthcare workers during COVID-19 revealed that the pandemic was associated with significant depression, anxiety, and stress, as well as gut dysbiosis that persisted for at least half a year.
Notably, healthcare workers had low gut alpha diversity, indicating a less resilient and diverse microbiome, a state often associated with dysbiosis and increased risk for various diseases and negative health outcomes.
A two-cohort study of healthy adults found higher alpha diversity in those reporting low stress levels. It also found a link between stress and enriched levels of Escherichia/Shigella, an overgrowth of which has been linked to various conditions, including inflammatory bowel disease.
In addition, a 2023 systematic review of human studies concluded that stress is associated with changes in specific genera — namely reductions in gut-healthy Lachnospira/Lachnospiraceae and Phascolarctobacterium, which produce beneficial short-chain fatty acids that support the health of the intestinal lining and modulate the immune system.
Stress during specific life stages also appears to alter the gut microbiome.
For example, in a study of postpartum women, those at an increased risk for parenting stress showed lower alpha diversity on the Shannon diversity index.
Research involving mother-child pairs tied adversity — such as maltreatment of the mother during her childhood, prenatal anxiety, and hardship in the child’s early life — to distinct microbiome profiles in 2-year-olds, supporting a stress-microbiome pathway relevant to socioemotional outcomes, the authors said.
Emerging evidence indicates a link between the gut microbiome and posttraumatic stress disorder (PTSD).
A recent systematic review found differences in gut microbial taxa between individuals with PTSD and trauma-exposed controls without PTSD. A separate analysis pointed to a potential causal impact of gut microbiomes on the development of PTSD.
Mechanisms Behind the Link
Stress interferes with the brain’s production of neurotransmitters, such as serotonin, which controls anxiety, mood, sleep, and many other functions in the brain, Shaukat told GI & Hepatology News.
“But serotonin also crosses the blood-brain barrier, and actually, the gut has more serotonin receptors than the brain, so an imbalance of serotonin can actually affect the gut microbiome through signaling at the neurotransmitter level,” Shaukat explained.
Stress can also affect sleep, and sleep itself has regulatory properties for gut bacteria, Shaukat noted.
“Stress also lowers our immunity, and this can make the gut barrier susceptible or permeable to bacterial toxins that can pass through and breach the gut barrier and be released into the bloodstream, which can trigger inflammation,” Shaukat explained.
Implications for Patient Care
The gut-brain-microbiome axis remains an emerging field of study. “We’re learning more and more about this, and we need to because the microbial colonies are so diverse and we haven’t nailed it down yet,” Shaukat said.
In the meantime, what can clinicians tell patients?
Aside from managing stress, which “is easier said than done,” patients can improve their diet, Shaukat said.
“What we tell patients is to essentially increase their intake of gut-friendly foods that preferentially grow the bacterial colonies that are beneficial for us,” Shaukat said. This includes fermented foods, yogurt, kimchi, chia seeds, kombucha, pickled vegetables, and whole grains.
A recent randomized controlled trial of healthy adults found a “psychobiotic diet” — a diet high in prebiotic and fermented foods — was associated with less perceived stress and subtle beneficial shifts in microbial composition.
“These foods can help keep the gut in good health and may actually also reduce or mitigate some of the effects of stress,” Shaukat said.
“Eating well is something I think we should all think about and maybe prioritize when we’re going through a stressful situation or looking to kind of mitigate the effects of stress and the anxiety and depression it can cause,” she advised.
Shaukat said she also encourages patients to engage in regular physical activity, which benefits the gut microbiome by helping to regulate gut motility. Exercise can also boost mood and help relieve stress.
“A balanced Mediterranean diet and regular activity is truly the secret for gut health,” Shaukat said.
Patients may be tempted by the probiotic supplements lining drugstore shelves, but there “isn’t great evidence for probiotic supplements,” she said. “What we can get from dietary sources far outweighs what can be put in a pill.”
Shaukat disclosed having no relevant disclosures.
A version of this article appeared on Medscape.com.
Chronic psychological stress is common. A 2023 survey revealed that about one quarter of US adults reported high stress levels, and three quarters reported that chronic stress affects their daily lives.
Emerging evidence suggests that chronic stress not only exacts a high toll on mental health but also can wreak havoc on all levels of gastrointestinal (GI) functioning, all the way down to the microbiome.
Aasma Shaukat, MD, MPH, AGAF, gastroenterologist with NYU Langone Health and director of GI Outcomes Research, Gastroenterology at NYU Grossman School of Medicine in New York City, said in an interview with GI & Hepatology News.
“This basically means that the normal balance of microorganisms that essentially we think are beneficial gets reduced, and the colonies considered to be more harmful proliferate,” she explained.
What Does the Science Tell Us?
Numerous studies published in the past 5 years have linked chronic stress to modest but reproducible shifts in the composition of the microbiome.
A study of frontline healthcare workers during COVID-19 revealed that the pandemic was associated with significant depression, anxiety, and stress, as well as gut dysbiosis that persisted for at least half a year.
Notably, healthcare workers had low gut alpha diversity, indicating a less resilient and diverse microbiome, a state often associated with dysbiosis and increased risk for various diseases and negative health outcomes.
A two-cohort study of healthy adults found higher alpha diversity in those reporting low stress levels. It also found a link between stress and enriched levels of Escherichia/Shigella, an overgrowth of which has been linked to various conditions, including inflammatory bowel disease.
In addition, a 2023 systematic review of human studies concluded that stress is associated with changes in specific genera — namely reductions in gut-healthy Lachnospira/Lachnospiraceae and Phascolarctobacterium, which produce beneficial short-chain fatty acids that support the health of the intestinal lining and modulate the immune system.
Stress during specific life stages also appears to alter the gut microbiome.
For example, in a study of postpartum women, those at an increased risk for parenting stress showed lower alpha diversity on the Shannon diversity index.
Research involving mother-child pairs tied adversity — such as maltreatment of the mother during her childhood, prenatal anxiety, and hardship in the child’s early life — to distinct microbiome profiles in 2-year-olds, supporting a stress-microbiome pathway relevant to socioemotional outcomes, the authors said.
Emerging evidence indicates a link between the gut microbiome and posttraumatic stress disorder (PTSD).
A recent systematic review found differences in gut microbial taxa between individuals with PTSD and trauma-exposed controls without PTSD. A separate analysis pointed to a potential causal impact of gut microbiomes on the development of PTSD.
Mechanisms Behind the Link
Stress interferes with the brain’s production of neurotransmitters, such as serotonin, which controls anxiety, mood, sleep, and many other functions in the brain, Shaukat told GI & Hepatology News.
“But serotonin also crosses the blood-brain barrier, and actually, the gut has more serotonin receptors than the brain, so an imbalance of serotonin can actually affect the gut microbiome through signaling at the neurotransmitter level,” Shaukat explained.
Stress can also affect sleep, and sleep itself has regulatory properties for gut bacteria, Shaukat noted.
“Stress also lowers our immunity, and this can make the gut barrier susceptible or permeable to bacterial toxins that can pass through and breach the gut barrier and be released into the bloodstream, which can trigger inflammation,” Shaukat explained.
Implications for Patient Care
The gut-brain-microbiome axis remains an emerging field of study. “We’re learning more and more about this, and we need to because the microbial colonies are so diverse and we haven’t nailed it down yet,” Shaukat said.
In the meantime, what can clinicians tell patients?
Aside from managing stress, which “is easier said than done,” patients can improve their diet, Shaukat said.
“What we tell patients is to essentially increase their intake of gut-friendly foods that preferentially grow the bacterial colonies that are beneficial for us,” Shaukat said. This includes fermented foods, yogurt, kimchi, chia seeds, kombucha, pickled vegetables, and whole grains.
A recent randomized controlled trial of healthy adults found a “psychobiotic diet” — a diet high in prebiotic and fermented foods — was associated with less perceived stress and subtle beneficial shifts in microbial composition.
“These foods can help keep the gut in good health and may actually also reduce or mitigate some of the effects of stress,” Shaukat said.
“Eating well is something I think we should all think about and maybe prioritize when we’re going through a stressful situation or looking to kind of mitigate the effects of stress and the anxiety and depression it can cause,” she advised.
Shaukat said she also encourages patients to engage in regular physical activity, which benefits the gut microbiome by helping to regulate gut motility. Exercise can also boost mood and help relieve stress.
“A balanced Mediterranean diet and regular activity is truly the secret for gut health,” Shaukat said.
Patients may be tempted by the probiotic supplements lining drugstore shelves, but there “isn’t great evidence for probiotic supplements,” she said. “What we can get from dietary sources far outweighs what can be put in a pill.”
Shaukat disclosed having no relevant disclosures.
A version of this article appeared on Medscape.com.
Chronic psychological stress is common. A 2023 survey revealed that about one quarter of US adults reported high stress levels, and three quarters reported that chronic stress affects their daily lives.
Emerging evidence suggests that chronic stress not only exacts a high toll on mental health but also can wreak havoc on all levels of gastrointestinal (GI) functioning, all the way down to the microbiome.
Aasma Shaukat, MD, MPH, AGAF, gastroenterologist with NYU Langone Health and director of GI Outcomes Research, Gastroenterology at NYU Grossman School of Medicine in New York City, said in an interview with GI & Hepatology News.
“This basically means that the normal balance of microorganisms that essentially we think are beneficial gets reduced, and the colonies considered to be more harmful proliferate,” she explained.
What Does the Science Tell Us?
Numerous studies published in the past 5 years have linked chronic stress to modest but reproducible shifts in the composition of the microbiome.
A study of frontline healthcare workers during COVID-19 revealed that the pandemic was associated with significant depression, anxiety, and stress, as well as gut dysbiosis that persisted for at least half a year.
Notably, healthcare workers had low gut alpha diversity, indicating a less resilient and diverse microbiome, a state often associated with dysbiosis and increased risk for various diseases and negative health outcomes.
A two-cohort study of healthy adults found higher alpha diversity in those reporting low stress levels. It also found a link between stress and enriched levels of Escherichia/Shigella, an overgrowth of which has been linked to various conditions, including inflammatory bowel disease.
In addition, a 2023 systematic review of human studies concluded that stress is associated with changes in specific genera — namely reductions in gut-healthy Lachnospira/Lachnospiraceae and Phascolarctobacterium, which produce beneficial short-chain fatty acids that support the health of the intestinal lining and modulate the immune system.
Stress during specific life stages also appears to alter the gut microbiome.
For example, in a study of postpartum women, those at an increased risk for parenting stress showed lower alpha diversity on the Shannon diversity index.
Research involving mother-child pairs tied adversity — such as maltreatment of the mother during her childhood, prenatal anxiety, and hardship in the child’s early life — to distinct microbiome profiles in 2-year-olds, supporting a stress-microbiome pathway relevant to socioemotional outcomes, the authors said.
Emerging evidence indicates a link between the gut microbiome and posttraumatic stress disorder (PTSD).
A recent systematic review found differences in gut microbial taxa between individuals with PTSD and trauma-exposed controls without PTSD. A separate analysis pointed to a potential causal impact of gut microbiomes on the development of PTSD.
Mechanisms Behind the Link
Stress interferes with the brain’s production of neurotransmitters, such as serotonin, which controls anxiety, mood, sleep, and many other functions in the brain, Shaukat told GI & Hepatology News.
“But serotonin also crosses the blood-brain barrier, and actually, the gut has more serotonin receptors than the brain, so an imbalance of serotonin can actually affect the gut microbiome through signaling at the neurotransmitter level,” Shaukat explained.
Stress can also affect sleep, and sleep itself has regulatory properties for gut bacteria, Shaukat noted.
“Stress also lowers our immunity, and this can make the gut barrier susceptible or permeable to bacterial toxins that can pass through and breach the gut barrier and be released into the bloodstream, which can trigger inflammation,” Shaukat explained.
Implications for Patient Care
The gut-brain-microbiome axis remains an emerging field of study. “We’re learning more and more about this, and we need to because the microbial colonies are so diverse and we haven’t nailed it down yet,” Shaukat said.
In the meantime, what can clinicians tell patients?
Aside from managing stress, which “is easier said than done,” patients can improve their diet, Shaukat said.
“What we tell patients is to essentially increase their intake of gut-friendly foods that preferentially grow the bacterial colonies that are beneficial for us,” Shaukat said. This includes fermented foods, yogurt, kimchi, chia seeds, kombucha, pickled vegetables, and whole grains.
A recent randomized controlled trial of healthy adults found a “psychobiotic diet” — a diet high in prebiotic and fermented foods — was associated with less perceived stress and subtle beneficial shifts in microbial composition.
“These foods can help keep the gut in good health and may actually also reduce or mitigate some of the effects of stress,” Shaukat said.
“Eating well is something I think we should all think about and maybe prioritize when we’re going through a stressful situation or looking to kind of mitigate the effects of stress and the anxiety and depression it can cause,” she advised.
Shaukat said she also encourages patients to engage in regular physical activity, which benefits the gut microbiome by helping to regulate gut motility. Exercise can also boost mood and help relieve stress.
“A balanced Mediterranean diet and regular activity is truly the secret for gut health,” Shaukat said.
Patients may be tempted by the probiotic supplements lining drugstore shelves, but there “isn’t great evidence for probiotic supplements,” she said. “What we can get from dietary sources far outweighs what can be put in a pill.”
Shaukat disclosed having no relevant disclosures.
A version of this article appeared on Medscape.com.
PET and CPT Show Promise in Veteran PTSD Treatment
Posttraumatic stress disorder (PTSD) guidelines increasingly are recommending prolonged exposure therapy (PET) and cognitive processing therapy (CPT) as first-line treatments, including the 2023 US Department of Veterans Affairs (VA) and US Department of Defense clinical practice guideline.
Since 2006, > 6000 VA therapists have been trained in PET and CPT; the VA requires all veterans to have access to these treatments. However, despite strong clinical trial evidence supporting PET and CPT for the treatment of PTSD, a 2023 study found that only 11.6% of veterans who received a PTSD diagnosis between 2017 and 2019 initiated Trauma-Focused Evidence-Based Psychotherapy (TF-EBP) in their first year of treatment. Of those who initiated TF-EBP, 67% dropped out. Recent VA programs have attempted to expand the reach of PET with video telehealth to reach rural and remote veterans through virtual group programs.
Recent research has suggested ways to maximize the effectiveness of the programs and assist veterans in receiving the full benefits. Studies have found that swapping traditional longer-term treatments (usually spanning 8 to 15 weeks) for intensified, shorter versions (eg, 6 sessions) may enhance engagement and retention.
Intensive PET for PTSD is safe and highly effective. A study involving patients with chronic PTSD and complex trauma showed significant reductions in PTSD symptom severity, with large effect sizes and sustained improvements at 3 and 6 months. Multiple 90-minute sessions over consecutive days, supplemented with in vivo exposure or followed by weekly booster sessions, were found to minimize treatment disruptions.
PET is among the most extensively studied treatments for PTSD and is supported by dozens of clinical trials involving thousands of patients. The intervention was originally developed and validated in civilian samples and includes psychoeducation, relaxation through breathing retraining, and in vivo and imaginal exposure to traumatic memories.
A recent study compared treatment outcomes among military veterans and civilian patients receiving treatment in a community setting. Although some studies have compared PET outcomes for military veterans and civilian participants in community settings, none have directly compared outcomes across trauma type (combat, terror, or civilian trauma) and veteran status (military vs civilian) within the same framework. The study notes that combat-related trauma significantly differs from other forms of trauma exposure, as it is typically more prolonged and severe and therefore is more often resistant to treatment. Military personnel also often find themselves both victims and aggressors, a duality that can intensify guilt, shame, anger, disgust, and emotional reactions to moral injury, complicating treatment.
The study assessed the effects of 8 to 15 PET sessions on PTSD symptoms in 55 civilians and 43 veterans using the PTSD Symptom Scale–Interview Version (PSS-I). Participants showed significant symptom reductions across all trauma types and veteran statuses.
Although veterans and participants in the combat trauma subgroup showed higher levels of baseline symptom severity compared with civilians, all groups experienced similar symptom reductions. These findings differ from some meta-analyses, which have found that PET often produces smaller effect sizes in combat-related PTSD compared to civilian trauma samples.
The study compared treatment outcomes across different groups within the same treatment centers and under consistent supervision. The PET intervention was delivered in community mental health centers to all patients regardless of background. Only 2 prior studies have compared civilian and military veterans within the same locations.
Although the “traditional” number of PET sessions produce evidence-based outcomes, high dropout rates and relapses have catalyzed interest in approaches that boost the power of therapy, such as delivering PET in ever-shorter sequences.
A study in a Swedish psychiatric outpatient clinic compared the effect of an 8-day intensified treatment program with traditionally spaced treatments on 101 participants with PTSD or complex PTSD. The study reported a significant reduction in PTSD symptoms at posttreatment, with large effect sizes in both conditions. Moreover, symptom reduction was maintained at follow-up. Dropout rates were significantly different between treatment groups: 4.3% in the intensified treatment program and 24.1% in the traditional group.
Another study used VA administrative data to assess the impact of sequenced psychotherapy (≥ 8 sessions of not trauma-focused individual or group psychotherapy delivered before trauma-focused care) on initiation and retention in CPT and PET over 2 years. Roughly 13% of 490,097 veterans who entered care for PTSD between 2014 and 2020 initiated VA-disseminated evidence-based treatment within 21 months (9.5% CPT, 3.4% PE). Among those who initiated treatment, retention was 46% and 42%, respectively. Individual therapy was associated with increased CPT and PET retention of 8.0% and 8.2%. For group therapy, retention increases were 3.4% and 8.7%.
Another recent study examined the RESET (Reconsolidation, Exposure, and Short-term Emotional Transformation) clinical protocol, an intensive, structured trauma-focused intervention designed to treat PTSD within 6 daily sessions. The protocol includes psychoeducation, targeted exposure, dynamic case formulation, and guided trauma processing. This novel framework ensures therapy moves beyond symptom reduction, fostering a deep understanding of the patient’s core struggles and their broader psychological patterns, and integrates it with the reconsolidation of the index trauma narrative to form a more cohesive sense of self.”
Clinical studies are ongoing to refine and enhance PET and CPT. They may serve to make therapy more useful and effective in easing—maybe erasing—veterans’ traumatic memories.
Posttraumatic stress disorder (PTSD) guidelines increasingly are recommending prolonged exposure therapy (PET) and cognitive processing therapy (CPT) as first-line treatments, including the 2023 US Department of Veterans Affairs (VA) and US Department of Defense clinical practice guideline.
Since 2006, > 6000 VA therapists have been trained in PET and CPT; the VA requires all veterans to have access to these treatments. However, despite strong clinical trial evidence supporting PET and CPT for the treatment of PTSD, a 2023 study found that only 11.6% of veterans who received a PTSD diagnosis between 2017 and 2019 initiated Trauma-Focused Evidence-Based Psychotherapy (TF-EBP) in their first year of treatment. Of those who initiated TF-EBP, 67% dropped out. Recent VA programs have attempted to expand the reach of PET with video telehealth to reach rural and remote veterans through virtual group programs.
Recent research has suggested ways to maximize the effectiveness of the programs and assist veterans in receiving the full benefits. Studies have found that swapping traditional longer-term treatments (usually spanning 8 to 15 weeks) for intensified, shorter versions (eg, 6 sessions) may enhance engagement and retention.
Intensive PET for PTSD is safe and highly effective. A study involving patients with chronic PTSD and complex trauma showed significant reductions in PTSD symptom severity, with large effect sizes and sustained improvements at 3 and 6 months. Multiple 90-minute sessions over consecutive days, supplemented with in vivo exposure or followed by weekly booster sessions, were found to minimize treatment disruptions.
PET is among the most extensively studied treatments for PTSD and is supported by dozens of clinical trials involving thousands of patients. The intervention was originally developed and validated in civilian samples and includes psychoeducation, relaxation through breathing retraining, and in vivo and imaginal exposure to traumatic memories.
A recent study compared treatment outcomes among military veterans and civilian patients receiving treatment in a community setting. Although some studies have compared PET outcomes for military veterans and civilian participants in community settings, none have directly compared outcomes across trauma type (combat, terror, or civilian trauma) and veteran status (military vs civilian) within the same framework. The study notes that combat-related trauma significantly differs from other forms of trauma exposure, as it is typically more prolonged and severe and therefore is more often resistant to treatment. Military personnel also often find themselves both victims and aggressors, a duality that can intensify guilt, shame, anger, disgust, and emotional reactions to moral injury, complicating treatment.
The study assessed the effects of 8 to 15 PET sessions on PTSD symptoms in 55 civilians and 43 veterans using the PTSD Symptom Scale–Interview Version (PSS-I). Participants showed significant symptom reductions across all trauma types and veteran statuses.
Although veterans and participants in the combat trauma subgroup showed higher levels of baseline symptom severity compared with civilians, all groups experienced similar symptom reductions. These findings differ from some meta-analyses, which have found that PET often produces smaller effect sizes in combat-related PTSD compared to civilian trauma samples.
The study compared treatment outcomes across different groups within the same treatment centers and under consistent supervision. The PET intervention was delivered in community mental health centers to all patients regardless of background. Only 2 prior studies have compared civilian and military veterans within the same locations.
Although the “traditional” number of PET sessions produce evidence-based outcomes, high dropout rates and relapses have catalyzed interest in approaches that boost the power of therapy, such as delivering PET in ever-shorter sequences.
A study in a Swedish psychiatric outpatient clinic compared the effect of an 8-day intensified treatment program with traditionally spaced treatments on 101 participants with PTSD or complex PTSD. The study reported a significant reduction in PTSD symptoms at posttreatment, with large effect sizes in both conditions. Moreover, symptom reduction was maintained at follow-up. Dropout rates were significantly different between treatment groups: 4.3% in the intensified treatment program and 24.1% in the traditional group.
Another study used VA administrative data to assess the impact of sequenced psychotherapy (≥ 8 sessions of not trauma-focused individual or group psychotherapy delivered before trauma-focused care) on initiation and retention in CPT and PET over 2 years. Roughly 13% of 490,097 veterans who entered care for PTSD between 2014 and 2020 initiated VA-disseminated evidence-based treatment within 21 months (9.5% CPT, 3.4% PE). Among those who initiated treatment, retention was 46% and 42%, respectively. Individual therapy was associated with increased CPT and PET retention of 8.0% and 8.2%. For group therapy, retention increases were 3.4% and 8.7%.
Another recent study examined the RESET (Reconsolidation, Exposure, and Short-term Emotional Transformation) clinical protocol, an intensive, structured trauma-focused intervention designed to treat PTSD within 6 daily sessions. The protocol includes psychoeducation, targeted exposure, dynamic case formulation, and guided trauma processing. This novel framework ensures therapy moves beyond symptom reduction, fostering a deep understanding of the patient’s core struggles and their broader psychological patterns, and integrates it with the reconsolidation of the index trauma narrative to form a more cohesive sense of self.”
Clinical studies are ongoing to refine and enhance PET and CPT. They may serve to make therapy more useful and effective in easing—maybe erasing—veterans’ traumatic memories.
Posttraumatic stress disorder (PTSD) guidelines increasingly are recommending prolonged exposure therapy (PET) and cognitive processing therapy (CPT) as first-line treatments, including the 2023 US Department of Veterans Affairs (VA) and US Department of Defense clinical practice guideline.
Since 2006, > 6000 VA therapists have been trained in PET and CPT; the VA requires all veterans to have access to these treatments. However, despite strong clinical trial evidence supporting PET and CPT for the treatment of PTSD, a 2023 study found that only 11.6% of veterans who received a PTSD diagnosis between 2017 and 2019 initiated Trauma-Focused Evidence-Based Psychotherapy (TF-EBP) in their first year of treatment. Of those who initiated TF-EBP, 67% dropped out. Recent VA programs have attempted to expand the reach of PET with video telehealth to reach rural and remote veterans through virtual group programs.
Recent research has suggested ways to maximize the effectiveness of the programs and assist veterans in receiving the full benefits. Studies have found that swapping traditional longer-term treatments (usually spanning 8 to 15 weeks) for intensified, shorter versions (eg, 6 sessions) may enhance engagement and retention.
Intensive PET for PTSD is safe and highly effective. A study involving patients with chronic PTSD and complex trauma showed significant reductions in PTSD symptom severity, with large effect sizes and sustained improvements at 3 and 6 months. Multiple 90-minute sessions over consecutive days, supplemented with in vivo exposure or followed by weekly booster sessions, were found to minimize treatment disruptions.
PET is among the most extensively studied treatments for PTSD and is supported by dozens of clinical trials involving thousands of patients. The intervention was originally developed and validated in civilian samples and includes psychoeducation, relaxation through breathing retraining, and in vivo and imaginal exposure to traumatic memories.
A recent study compared treatment outcomes among military veterans and civilian patients receiving treatment in a community setting. Although some studies have compared PET outcomes for military veterans and civilian participants in community settings, none have directly compared outcomes across trauma type (combat, terror, or civilian trauma) and veteran status (military vs civilian) within the same framework. The study notes that combat-related trauma significantly differs from other forms of trauma exposure, as it is typically more prolonged and severe and therefore is more often resistant to treatment. Military personnel also often find themselves both victims and aggressors, a duality that can intensify guilt, shame, anger, disgust, and emotional reactions to moral injury, complicating treatment.
The study assessed the effects of 8 to 15 PET sessions on PTSD symptoms in 55 civilians and 43 veterans using the PTSD Symptom Scale–Interview Version (PSS-I). Participants showed significant symptom reductions across all trauma types and veteran statuses.
Although veterans and participants in the combat trauma subgroup showed higher levels of baseline symptom severity compared with civilians, all groups experienced similar symptom reductions. These findings differ from some meta-analyses, which have found that PET often produces smaller effect sizes in combat-related PTSD compared to civilian trauma samples.
The study compared treatment outcomes across different groups within the same treatment centers and under consistent supervision. The PET intervention was delivered in community mental health centers to all patients regardless of background. Only 2 prior studies have compared civilian and military veterans within the same locations.
Although the “traditional” number of PET sessions produce evidence-based outcomes, high dropout rates and relapses have catalyzed interest in approaches that boost the power of therapy, such as delivering PET in ever-shorter sequences.
A study in a Swedish psychiatric outpatient clinic compared the effect of an 8-day intensified treatment program with traditionally spaced treatments on 101 participants with PTSD or complex PTSD. The study reported a significant reduction in PTSD symptoms at posttreatment, with large effect sizes in both conditions. Moreover, symptom reduction was maintained at follow-up. Dropout rates were significantly different between treatment groups: 4.3% in the intensified treatment program and 24.1% in the traditional group.
Another study used VA administrative data to assess the impact of sequenced psychotherapy (≥ 8 sessions of not trauma-focused individual or group psychotherapy delivered before trauma-focused care) on initiation and retention in CPT and PET over 2 years. Roughly 13% of 490,097 veterans who entered care for PTSD between 2014 and 2020 initiated VA-disseminated evidence-based treatment within 21 months (9.5% CPT, 3.4% PE). Among those who initiated treatment, retention was 46% and 42%, respectively. Individual therapy was associated with increased CPT and PET retention of 8.0% and 8.2%. For group therapy, retention increases were 3.4% and 8.7%.
Another recent study examined the RESET (Reconsolidation, Exposure, and Short-term Emotional Transformation) clinical protocol, an intensive, structured trauma-focused intervention designed to treat PTSD within 6 daily sessions. The protocol includes psychoeducation, targeted exposure, dynamic case formulation, and guided trauma processing. This novel framework ensures therapy moves beyond symptom reduction, fostering a deep understanding of the patient’s core struggles and their broader psychological patterns, and integrates it with the reconsolidation of the index trauma narrative to form a more cohesive sense of self.”
Clinical studies are ongoing to refine and enhance PET and CPT. They may serve to make therapy more useful and effective in easing—maybe erasing—veterans’ traumatic memories.
Moral Injury-informed Interventions May Enhance Treatment for Combat Veterans
“Moral and mortal stressors may be intertwined in their contribution to the complex symptomatic outcomes” of combat exposure according to a recent study in the European Journal of Psychotraumatology. The study examined the effect moral injury has on Israel Defense Forces (IDF) combat veterans. The resulting trauma may be consolidated in a single category, such as posttraumatic stress disorder (PTSD), but stressors leading to that diagnosis may have been quite different. Properly defining the stressors to assist in better targeted treatment is a challenge.
Moral injury is the emotional distress of being involved in or witnessing actions that conflict with deeply held beliefs. Such experiences could be committing or failing to prevent a transgressive act or learning about or surviving a transgressive act.
The study defines moral injury outcomes as the psychological and emotional consequences that result from exposure to potentially morally injurious events (PMIEs): “This terminology is intended to distinguish the outcomes of moral injury from the broader and sometimes ambiguous use of ‘moral injury’ in the literature, which can refer to either the event, the experience, or the resulting symptoms.”
The study followed 374 male combat veterans for 5 years. Veterans served in the Israel Defense Forces (IDF) in 4 primary combat roles: infantry, armored corps, special forces, and combat engineering. Psychological characteristics were measured 12 months prior to enlistment. PMIE exposure was measured during the final month of military service using the Moral Injury Events Scale. Moral injury outcomes were assessed 6 months postdischarge using the Expressions of Moral Injury Scale-Military Version-Short Form. Posttraumatic stress symptom (PTSS) clusters were evaluated 1 year postdischarge using the PTSD Checklist for the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition.
Nearly half (49%) of participants reported exposure to PMIEs, while 8% met criteria for probable PTSD. The researchers say elevated PMIE rates observed in their sample may be attributed, in part, to participants’ extended deployments in densely populated urban areas, carrying out operations in close proximity to civilians, where it is difficult to distinguish between combatants and noncombatants. PTSD rates were somewhat lower than those reported in US studies (10% to 30%) among veterans; this may be attributed to the cohort not being engaged in a full-scale war, but deployed mostly in peacekeeping missions.
Longitudinal studies have described the effects of wartime atrocities on PTSD symptom severity. Studies have also linked moral injury outcomes and PTSS clusters (including negative alterations in cognition and mood [NACM]), depression, anxiety, and substance abuse. PMIEs can also include perceptions of betrayal from leaders, colleagues, or trusted others. The study of 374 male combat veterans found a direct effect of PMIE-betrayal on arousal and reactivity as well as NACM clusters. Results also showed indirect associations between exposure to all PMIE dimensions and PTSS clusters via moral injury outcomes. Combat exposure and experiencing PMIEs during military service significantly contributed to the emergence of PTSS during the first year after discharge. The study found 2 distinct paths PMIEs may lead to PTSS among veterans: experiencing acts of transgression and encountering betrayal.
Betrayal has been linked to feelings of anger and humiliation, emotions thought to have evolved to trigger adaptive behavioral responses, such as aggression and revenge, to threats or transgressions by others. PMIE-betrayal also demonstrated direct effects on the arousal and reactivity and NACM symptom clusters, suggesting partial mediation. Another study (also on IDF veterans) found significant positive correlations between PMIE-betrayal and the NACM cluster, suggesting PMIE-betrayal as a link between PTSD and moral injury. While the link between betrayal and NACM is readily apparent, its connection to arousal and reactivity, a fear-based physiological symptom, is less evident.
The findings of the study point to the need for assessment tools that separately measure exposure to PMIEs and individual reactions to them. A recent Federal Practitioner study of 100 veterans with a history of incarceration completed the Moral Injury Events Scale and an adapted version for legal-involved persons (MIES-LIP). The authors found that MIES-LIP demonstrated strong psychometric properties, including good reliability and convergent validity for legal-related moral injury.
The study cites a recent review of cognitive-behavioral psychotherapies for individuals experiencing moral injury that challenges the adequacy of existing evidence-based treatments for PTSD for addressing moral injury and its associated symptoms. It is important to evaluate individuals who express feelings of betrayal with tailored, evidence-based interventions such as adaptive disclosure or cognitive-processing therapy. Acceptance and commitment therapy may also help individuals experiencing emotions such as shame, humiliation, guilt, and anger following morally injurious events.
Newer therapy models like Multi-Modal Motion-Assisted Memory Desensitization and Reconsolidation allow clinicians to use personalized trauma cues to facilitate memory processing, reduce avoidance, and aid in emotional reconsolidation. Clinical research has demonstrated this model’s efficacy in reducing PTSD symptoms, depression, and anxiety, with high acceptability and low dropout rates among military personnel, veterans, and first responders.
Regardless of the treatment, the researchers encourage mental health professionals to approach veterans seeking help with the “utmost sensitivity and attentiveness to any expressions of (moral injury) outcomes.”
“Moral and mortal stressors may be intertwined in their contribution to the complex symptomatic outcomes” of combat exposure according to a recent study in the European Journal of Psychotraumatology. The study examined the effect moral injury has on Israel Defense Forces (IDF) combat veterans. The resulting trauma may be consolidated in a single category, such as posttraumatic stress disorder (PTSD), but stressors leading to that diagnosis may have been quite different. Properly defining the stressors to assist in better targeted treatment is a challenge.
Moral injury is the emotional distress of being involved in or witnessing actions that conflict with deeply held beliefs. Such experiences could be committing or failing to prevent a transgressive act or learning about or surviving a transgressive act.
The study defines moral injury outcomes as the psychological and emotional consequences that result from exposure to potentially morally injurious events (PMIEs): “This terminology is intended to distinguish the outcomes of moral injury from the broader and sometimes ambiguous use of ‘moral injury’ in the literature, which can refer to either the event, the experience, or the resulting symptoms.”
The study followed 374 male combat veterans for 5 years. Veterans served in the Israel Defense Forces (IDF) in 4 primary combat roles: infantry, armored corps, special forces, and combat engineering. Psychological characteristics were measured 12 months prior to enlistment. PMIE exposure was measured during the final month of military service using the Moral Injury Events Scale. Moral injury outcomes were assessed 6 months postdischarge using the Expressions of Moral Injury Scale-Military Version-Short Form. Posttraumatic stress symptom (PTSS) clusters were evaluated 1 year postdischarge using the PTSD Checklist for the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition.
Nearly half (49%) of participants reported exposure to PMIEs, while 8% met criteria for probable PTSD. The researchers say elevated PMIE rates observed in their sample may be attributed, in part, to participants’ extended deployments in densely populated urban areas, carrying out operations in close proximity to civilians, where it is difficult to distinguish between combatants and noncombatants. PTSD rates were somewhat lower than those reported in US studies (10% to 30%) among veterans; this may be attributed to the cohort not being engaged in a full-scale war, but deployed mostly in peacekeeping missions.
Longitudinal studies have described the effects of wartime atrocities on PTSD symptom severity. Studies have also linked moral injury outcomes and PTSS clusters (including negative alterations in cognition and mood [NACM]), depression, anxiety, and substance abuse. PMIEs can also include perceptions of betrayal from leaders, colleagues, or trusted others. The study of 374 male combat veterans found a direct effect of PMIE-betrayal on arousal and reactivity as well as NACM clusters. Results also showed indirect associations between exposure to all PMIE dimensions and PTSS clusters via moral injury outcomes. Combat exposure and experiencing PMIEs during military service significantly contributed to the emergence of PTSS during the first year after discharge. The study found 2 distinct paths PMIEs may lead to PTSS among veterans: experiencing acts of transgression and encountering betrayal.
Betrayal has been linked to feelings of anger and humiliation, emotions thought to have evolved to trigger adaptive behavioral responses, such as aggression and revenge, to threats or transgressions by others. PMIE-betrayal also demonstrated direct effects on the arousal and reactivity and NACM symptom clusters, suggesting partial mediation. Another study (also on IDF veterans) found significant positive correlations between PMIE-betrayal and the NACM cluster, suggesting PMIE-betrayal as a link between PTSD and moral injury. While the link between betrayal and NACM is readily apparent, its connection to arousal and reactivity, a fear-based physiological symptom, is less evident.
The findings of the study point to the need for assessment tools that separately measure exposure to PMIEs and individual reactions to them. A recent Federal Practitioner study of 100 veterans with a history of incarceration completed the Moral Injury Events Scale and an adapted version for legal-involved persons (MIES-LIP). The authors found that MIES-LIP demonstrated strong psychometric properties, including good reliability and convergent validity for legal-related moral injury.
The study cites a recent review of cognitive-behavioral psychotherapies for individuals experiencing moral injury that challenges the adequacy of existing evidence-based treatments for PTSD for addressing moral injury and its associated symptoms. It is important to evaluate individuals who express feelings of betrayal with tailored, evidence-based interventions such as adaptive disclosure or cognitive-processing therapy. Acceptance and commitment therapy may also help individuals experiencing emotions such as shame, humiliation, guilt, and anger following morally injurious events.
Newer therapy models like Multi-Modal Motion-Assisted Memory Desensitization and Reconsolidation allow clinicians to use personalized trauma cues to facilitate memory processing, reduce avoidance, and aid in emotional reconsolidation. Clinical research has demonstrated this model’s efficacy in reducing PTSD symptoms, depression, and anxiety, with high acceptability and low dropout rates among military personnel, veterans, and first responders.
Regardless of the treatment, the researchers encourage mental health professionals to approach veterans seeking help with the “utmost sensitivity and attentiveness to any expressions of (moral injury) outcomes.”
“Moral and mortal stressors may be intertwined in their contribution to the complex symptomatic outcomes” of combat exposure according to a recent study in the European Journal of Psychotraumatology. The study examined the effect moral injury has on Israel Defense Forces (IDF) combat veterans. The resulting trauma may be consolidated in a single category, such as posttraumatic stress disorder (PTSD), but stressors leading to that diagnosis may have been quite different. Properly defining the stressors to assist in better targeted treatment is a challenge.
Moral injury is the emotional distress of being involved in or witnessing actions that conflict with deeply held beliefs. Such experiences could be committing or failing to prevent a transgressive act or learning about or surviving a transgressive act.
The study defines moral injury outcomes as the psychological and emotional consequences that result from exposure to potentially morally injurious events (PMIEs): “This terminology is intended to distinguish the outcomes of moral injury from the broader and sometimes ambiguous use of ‘moral injury’ in the literature, which can refer to either the event, the experience, or the resulting symptoms.”
The study followed 374 male combat veterans for 5 years. Veterans served in the Israel Defense Forces (IDF) in 4 primary combat roles: infantry, armored corps, special forces, and combat engineering. Psychological characteristics were measured 12 months prior to enlistment. PMIE exposure was measured during the final month of military service using the Moral Injury Events Scale. Moral injury outcomes were assessed 6 months postdischarge using the Expressions of Moral Injury Scale-Military Version-Short Form. Posttraumatic stress symptom (PTSS) clusters were evaluated 1 year postdischarge using the PTSD Checklist for the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition.
Nearly half (49%) of participants reported exposure to PMIEs, while 8% met criteria for probable PTSD. The researchers say elevated PMIE rates observed in their sample may be attributed, in part, to participants’ extended deployments in densely populated urban areas, carrying out operations in close proximity to civilians, where it is difficult to distinguish between combatants and noncombatants. PTSD rates were somewhat lower than those reported in US studies (10% to 30%) among veterans; this may be attributed to the cohort not being engaged in a full-scale war, but deployed mostly in peacekeeping missions.
Longitudinal studies have described the effects of wartime atrocities on PTSD symptom severity. Studies have also linked moral injury outcomes and PTSS clusters (including negative alterations in cognition and mood [NACM]), depression, anxiety, and substance abuse. PMIEs can also include perceptions of betrayal from leaders, colleagues, or trusted others. The study of 374 male combat veterans found a direct effect of PMIE-betrayal on arousal and reactivity as well as NACM clusters. Results also showed indirect associations between exposure to all PMIE dimensions and PTSS clusters via moral injury outcomes. Combat exposure and experiencing PMIEs during military service significantly contributed to the emergence of PTSS during the first year after discharge. The study found 2 distinct paths PMIEs may lead to PTSS among veterans: experiencing acts of transgression and encountering betrayal.
Betrayal has been linked to feelings of anger and humiliation, emotions thought to have evolved to trigger adaptive behavioral responses, such as aggression and revenge, to threats or transgressions by others. PMIE-betrayal also demonstrated direct effects on the arousal and reactivity and NACM symptom clusters, suggesting partial mediation. Another study (also on IDF veterans) found significant positive correlations between PMIE-betrayal and the NACM cluster, suggesting PMIE-betrayal as a link between PTSD and moral injury. While the link between betrayal and NACM is readily apparent, its connection to arousal and reactivity, a fear-based physiological symptom, is less evident.
The findings of the study point to the need for assessment tools that separately measure exposure to PMIEs and individual reactions to them. A recent Federal Practitioner study of 100 veterans with a history of incarceration completed the Moral Injury Events Scale and an adapted version for legal-involved persons (MIES-LIP). The authors found that MIES-LIP demonstrated strong psychometric properties, including good reliability and convergent validity for legal-related moral injury.
The study cites a recent review of cognitive-behavioral psychotherapies for individuals experiencing moral injury that challenges the adequacy of existing evidence-based treatments for PTSD for addressing moral injury and its associated symptoms. It is important to evaluate individuals who express feelings of betrayal with tailored, evidence-based interventions such as adaptive disclosure or cognitive-processing therapy. Acceptance and commitment therapy may also help individuals experiencing emotions such as shame, humiliation, guilt, and anger following morally injurious events.
Newer therapy models like Multi-Modal Motion-Assisted Memory Desensitization and Reconsolidation allow clinicians to use personalized trauma cues to facilitate memory processing, reduce avoidance, and aid in emotional reconsolidation. Clinical research has demonstrated this model’s efficacy in reducing PTSD symptoms, depression, and anxiety, with high acceptability and low dropout rates among military personnel, veterans, and first responders.
Regardless of the treatment, the researchers encourage mental health professionals to approach veterans seeking help with the “utmost sensitivity and attentiveness to any expressions of (moral injury) outcomes.”






