User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
Powered by CHEST Physician, Clinician Reviews, MDedge Family Medicine, Internal Medicine News, and The Journal of Clinical Outcomes Management.
Children with poor cardiorespiratory fitness have a higher risk of type 2 diabetes and cardiovascular disease
according to the analysis of an ongoing Finnish study of physical activity and dietary intervention in school children.
“Our results are in agreement with previous findings that cardiorespiratory fitness measured in exercise test laboratories or using field tests and scaled by body mass (BM) using the ratio standard method had a strong inverse association with cardiometabolic risk in children,” lead author Andrew O. Agbaje, MD, MPH, and his coauthors wrote in the Scandinavian Journal of Medicine & Science in Sports.
The coauthors assessed the cardiorespiratory fitness of 352 primary school children – 186 boys and 166 girls – from Kuopio, Finland, all of whom were already participating in the ongoing PANIC (Physical Activity and Nutrition in Children) Study. The children were asked to perform a maximal exercise test, upon which fitness was assessed by measuring peak oxygen uptake (VO2 peak), noted Dr. Agbaje, a PhD student at the University of Eastern Finland’s Institute of Biomedicine in Kuopio, and his colleagues.
Body mass and lean mass were also measured by bioelectrical impedance and used to scale VO2 peak, while variables such as waist circumference, insulin, glucose, HDL cholesterol, and triglycerides were used to calculate a continuous cardiometabolic risk score. Upon analysis, VO2 peak less than 45.8 mL/kg BM-1 min-1 in boys and less than 44.1 mL/kg BM-1 min-1 in girls was associated with increased cardiometabolic risk.
The coauthors noted that cardiorespiratory fitness can be influenced by genetics and that adjustments for puberty had “no effect on the relationships between VO2 peak and cardiometabolic risk.” As such, they recommended that “longitudinal studies are needed to clarify the role of CRF in cardiometabolic health during growth and maturation.”
That said, despite advocating caution in regard to determining proper CRF thresholds, the coauthors suggested that CRF scaled by BM could be used to screen children and improve prevention efforts. “Cardiometabolic risk tracks from childhood into adulthood and the early identification of individuals at increased risk is essential in developing public health actions targeted at preventing cardiometabolic diseases,” they wrote.
The study was funded by grants from the Ministry of Education and Culture of Finland, Ministry of Social Affairs and Health of Finland, Research Committee of the Kuopio University Hospital Catchment Area (State Research Funding), Finnish Innovation Fund Sitra, Social Insurance Institution of Finland, Finnish Cultural Foundation, Foundation for Paediatric Research, Diabetes Research Foundation in Finland, Finnish Foundation for Cardiovascular Research, Juho Vainio Foundation, Paavo Nurmi Foundation, and the Yrjö Jahnsson Foundation. Dr. Agbaje reported grant support from the Olvi Foundation and the Urho Känkanen Foundation.
SOURCE: Agbaje AO et al. Scand J Med Sci Sports. 2018 Sep 19. doi: 10.1111/sms.13307.
according to the analysis of an ongoing Finnish study of physical activity and dietary intervention in school children.
“Our results are in agreement with previous findings that cardiorespiratory fitness measured in exercise test laboratories or using field tests and scaled by body mass (BM) using the ratio standard method had a strong inverse association with cardiometabolic risk in children,” lead author Andrew O. Agbaje, MD, MPH, and his coauthors wrote in the Scandinavian Journal of Medicine & Science in Sports.
The coauthors assessed the cardiorespiratory fitness of 352 primary school children – 186 boys and 166 girls – from Kuopio, Finland, all of whom were already participating in the ongoing PANIC (Physical Activity and Nutrition in Children) Study. The children were asked to perform a maximal exercise test, upon which fitness was assessed by measuring peak oxygen uptake (VO2 peak), noted Dr. Agbaje, a PhD student at the University of Eastern Finland’s Institute of Biomedicine in Kuopio, and his colleagues.
Body mass and lean mass were also measured by bioelectrical impedance and used to scale VO2 peak, while variables such as waist circumference, insulin, glucose, HDL cholesterol, and triglycerides were used to calculate a continuous cardiometabolic risk score. Upon analysis, VO2 peak less than 45.8 mL/kg BM-1 min-1 in boys and less than 44.1 mL/kg BM-1 min-1 in girls was associated with increased cardiometabolic risk.
The coauthors noted that cardiorespiratory fitness can be influenced by genetics and that adjustments for puberty had “no effect on the relationships between VO2 peak and cardiometabolic risk.” As such, they recommended that “longitudinal studies are needed to clarify the role of CRF in cardiometabolic health during growth and maturation.”
That said, despite advocating caution in regard to determining proper CRF thresholds, the coauthors suggested that CRF scaled by BM could be used to screen children and improve prevention efforts. “Cardiometabolic risk tracks from childhood into adulthood and the early identification of individuals at increased risk is essential in developing public health actions targeted at preventing cardiometabolic diseases,” they wrote.
The study was funded by grants from the Ministry of Education and Culture of Finland, Ministry of Social Affairs and Health of Finland, Research Committee of the Kuopio University Hospital Catchment Area (State Research Funding), Finnish Innovation Fund Sitra, Social Insurance Institution of Finland, Finnish Cultural Foundation, Foundation for Paediatric Research, Diabetes Research Foundation in Finland, Finnish Foundation for Cardiovascular Research, Juho Vainio Foundation, Paavo Nurmi Foundation, and the Yrjö Jahnsson Foundation. Dr. Agbaje reported grant support from the Olvi Foundation and the Urho Känkanen Foundation.
SOURCE: Agbaje AO et al. Scand J Med Sci Sports. 2018 Sep 19. doi: 10.1111/sms.13307.
according to the analysis of an ongoing Finnish study of physical activity and dietary intervention in school children.
“Our results are in agreement with previous findings that cardiorespiratory fitness measured in exercise test laboratories or using field tests and scaled by body mass (BM) using the ratio standard method had a strong inverse association with cardiometabolic risk in children,” lead author Andrew O. Agbaje, MD, MPH, and his coauthors wrote in the Scandinavian Journal of Medicine & Science in Sports.
The coauthors assessed the cardiorespiratory fitness of 352 primary school children – 186 boys and 166 girls – from Kuopio, Finland, all of whom were already participating in the ongoing PANIC (Physical Activity and Nutrition in Children) Study. The children were asked to perform a maximal exercise test, upon which fitness was assessed by measuring peak oxygen uptake (VO2 peak), noted Dr. Agbaje, a PhD student at the University of Eastern Finland’s Institute of Biomedicine in Kuopio, and his colleagues.
Body mass and lean mass were also measured by bioelectrical impedance and used to scale VO2 peak, while variables such as waist circumference, insulin, glucose, HDL cholesterol, and triglycerides were used to calculate a continuous cardiometabolic risk score. Upon analysis, VO2 peak less than 45.8 mL/kg BM-1 min-1 in boys and less than 44.1 mL/kg BM-1 min-1 in girls was associated with increased cardiometabolic risk.
The coauthors noted that cardiorespiratory fitness can be influenced by genetics and that adjustments for puberty had “no effect on the relationships between VO2 peak and cardiometabolic risk.” As such, they recommended that “longitudinal studies are needed to clarify the role of CRF in cardiometabolic health during growth and maturation.”
That said, despite advocating caution in regard to determining proper CRF thresholds, the coauthors suggested that CRF scaled by BM could be used to screen children and improve prevention efforts. “Cardiometabolic risk tracks from childhood into adulthood and the early identification of individuals at increased risk is essential in developing public health actions targeted at preventing cardiometabolic diseases,” they wrote.
The study was funded by grants from the Ministry of Education and Culture of Finland, Ministry of Social Affairs and Health of Finland, Research Committee of the Kuopio University Hospital Catchment Area (State Research Funding), Finnish Innovation Fund Sitra, Social Insurance Institution of Finland, Finnish Cultural Foundation, Foundation for Paediatric Research, Diabetes Research Foundation in Finland, Finnish Foundation for Cardiovascular Research, Juho Vainio Foundation, Paavo Nurmi Foundation, and the Yrjö Jahnsson Foundation. Dr. Agbaje reported grant support from the Olvi Foundation and the Urho Känkanen Foundation.
SOURCE: Agbaje AO et al. Scand J Med Sci Sports. 2018 Sep 19. doi: 10.1111/sms.13307.
FROM THE SCANDINAVIAN JOURNAL OF MEDICINE & SCIENCE IN SPORTS
Key clinical point: Peak oxygen uptake less than 45.8 mL/kg BM-1 min-1 in boys and less than 44.1 mL/kg BM-1 min-1 in girls was associated with increased cardiometabolic risk.
Major finding: Cardiorespiratory fitness scaled by body mass could be used to screen for cardiometabolic risk in children.
Study details: An analysis of 352 Finnish children, all aged 9-11 years, who took a maximal exercise test as part of an ongoing physical activity and dietary intervention study.
Disclosures: The study was funded by grants from the Ministry of Education and Culture of Finland, Ministry of Social Affairs and Health of Finland, Research Committee of the Kuopio University Hospital Catchment Area, Finnish Innovation Fund Sitra, Social Insurance Institution of Finland, Finnish Cultural Foundation, Foundation for Paediatric Research, Diabetes Research Foundation in Finland, Finnish Foundation for Cardiovascular Research, Juho Vainio Foundation, Paavo Nurmi Foundation, and the Yrjö Jahnsson Foundation. Dr. Agbaje reported grant support from the Olvi Foundation and the Urho Känkanen Foundation.
Source: Agbaje AO et al. Scand J Med Sci Sports. 2018 Sep 19. doi: 10.1111/sms.13307.
FDA aims to squash youth vaping, smoking
The Food and Drug Administration once again has upped the ante in its war on youth smoking and vaping.
“Today, I’m pursuing actions aimed at addressing the disturbing trend of youth nicotine use and continuing to advance the historic declines we’ve achieved in recent years in the rates of combustible cigarette use among kids,” FDA Commissioner Scott Gottlieb, MD, said in a statement.
First and foremost, the FDA wants to reduce the lure of e-cigarettes by limiting the variety of flavored products for sale in retail outlets. Under the proposal unveiled Nov. 15, only electronic nicotine delivery systems (ENDS) that are unflavored or have tobacco, mint, or menthol flavors would be widely available. Flavored products – think cherry, cotton candy, and mango – would be sold in age-restricted environments, such as stand-alone tobacco retailers like vape shops. The FDA also seeks more stringent enforcement of age verification on ENDS products sold online.
The proposal also would reexamine regulations governing flavored cigars, with the possible aim of banning them.
“These efforts to address flavors and protect youth would dramatically impact the ability of American kids to access tobacco products that we know are both appealing and addicting,” Dr. Gottlieb said in a statement. “This policy framework reflects a redoubling of the FDA’s efforts to protect kids from all nicotine-containing products.”
In a move that seems to be aimed at youth-oriented products like Juul, the FDA will be seeking to remove from the market any ENDS product that is marketed specifically to young people.
Finally, the FDA intends to pursue regulation that would ban menthol from combustible tobacco products.
“I believe these menthol-flavored products represent one of the most common and pernicious routes by which kids initiate on combustible cigarettes,” Dr. Gottlieb said. “The menthol serves to mask some of the unattractive features of smoking that might otherwise discourage a child from smoking. Moreover, I believe that menthol products disproportionately and adversely affect underserved communities. And as a matter of public health, they exacerbate troubling disparities in health related to race and socioeconomic status.”
The policy shift comes as the Centers for Disease Control and Prevention released data from the 2018 National Youth Tobacco Survey showing that use of e-cigarettes among high schoolers is on the rise, growing from 1.5% in 2011 to 20.8% in 2018. Middle schoolers saw use over the same time period increase from 0.6% to 4.9%.
The rise of current use of e-cigarettes was enough to reverse a declining trend in overall tobacco use in recent years between 2015 and 2017.
“FDA’s enforcement efforts and policy framework would restrict access to most flavored e-cigarettes and limit the chances of youth beginning to use these products, while ensuring the products are available to adult smokers as an alternative to combustible cigarettes,” Alex M. Azar II, secretary of the Department of Health & Human Services, said in a statement supporting the FDA’s efforts. “Our obligation at HHS is always to the public health, and we believe FDA’s goals strike the right public health balance in addressing the multifaceted challenge we have before us today.”
Under Dr. Gottlieb, the FDA has been aggressively pursuing ways to reduce tobacco consumption, targeting both ENDS and combustible tobacco regulations in an effort to limit nicotine exposure and reduce the number of people addicted to nicotine and the health issues that come with it.
The American College of Cardiology voiced its support of the FDA’s actions.
“The FDA’s announcement restricting the sale of flavored e-cigarettes and other tobacco products shows they are ready to do their part in making tobacco products less available to our children,” ACC President C. Michael Valentine, MD, said in a statement, adding that the medical community needs to continue to do its part to make sure tobacco use continues to decline, especially in the nonadult population.
The FDA proposals were published as part of an advance notice of proposed rulemaking in the Federal Register. Comments can be made at www.regulations.gov through June 19.
The Food and Drug Administration once again has upped the ante in its war on youth smoking and vaping.
“Today, I’m pursuing actions aimed at addressing the disturbing trend of youth nicotine use and continuing to advance the historic declines we’ve achieved in recent years in the rates of combustible cigarette use among kids,” FDA Commissioner Scott Gottlieb, MD, said in a statement.
First and foremost, the FDA wants to reduce the lure of e-cigarettes by limiting the variety of flavored products for sale in retail outlets. Under the proposal unveiled Nov. 15, only electronic nicotine delivery systems (ENDS) that are unflavored or have tobacco, mint, or menthol flavors would be widely available. Flavored products – think cherry, cotton candy, and mango – would be sold in age-restricted environments, such as stand-alone tobacco retailers like vape shops. The FDA also seeks more stringent enforcement of age verification on ENDS products sold online.
The proposal also would reexamine regulations governing flavored cigars, with the possible aim of banning them.
“These efforts to address flavors and protect youth would dramatically impact the ability of American kids to access tobacco products that we know are both appealing and addicting,” Dr. Gottlieb said in a statement. “This policy framework reflects a redoubling of the FDA’s efforts to protect kids from all nicotine-containing products.”
In a move that seems to be aimed at youth-oriented products like Juul, the FDA will be seeking to remove from the market any ENDS product that is marketed specifically to young people.
Finally, the FDA intends to pursue regulation that would ban menthol from combustible tobacco products.
“I believe these menthol-flavored products represent one of the most common and pernicious routes by which kids initiate on combustible cigarettes,” Dr. Gottlieb said. “The menthol serves to mask some of the unattractive features of smoking that might otherwise discourage a child from smoking. Moreover, I believe that menthol products disproportionately and adversely affect underserved communities. And as a matter of public health, they exacerbate troubling disparities in health related to race and socioeconomic status.”
The policy shift comes as the Centers for Disease Control and Prevention released data from the 2018 National Youth Tobacco Survey showing that use of e-cigarettes among high schoolers is on the rise, growing from 1.5% in 2011 to 20.8% in 2018. Middle schoolers saw use over the same time period increase from 0.6% to 4.9%.
The rise of current use of e-cigarettes was enough to reverse a declining trend in overall tobacco use in recent years between 2015 and 2017.
“FDA’s enforcement efforts and policy framework would restrict access to most flavored e-cigarettes and limit the chances of youth beginning to use these products, while ensuring the products are available to adult smokers as an alternative to combustible cigarettes,” Alex M. Azar II, secretary of the Department of Health & Human Services, said in a statement supporting the FDA’s efforts. “Our obligation at HHS is always to the public health, and we believe FDA’s goals strike the right public health balance in addressing the multifaceted challenge we have before us today.”
Under Dr. Gottlieb, the FDA has been aggressively pursuing ways to reduce tobacco consumption, targeting both ENDS and combustible tobacco regulations in an effort to limit nicotine exposure and reduce the number of people addicted to nicotine and the health issues that come with it.
The American College of Cardiology voiced its support of the FDA’s actions.
“The FDA’s announcement restricting the sale of flavored e-cigarettes and other tobacco products shows they are ready to do their part in making tobacco products less available to our children,” ACC President C. Michael Valentine, MD, said in a statement, adding that the medical community needs to continue to do its part to make sure tobacco use continues to decline, especially in the nonadult population.
The FDA proposals were published as part of an advance notice of proposed rulemaking in the Federal Register. Comments can be made at www.regulations.gov through June 19.
The Food and Drug Administration once again has upped the ante in its war on youth smoking and vaping.
“Today, I’m pursuing actions aimed at addressing the disturbing trend of youth nicotine use and continuing to advance the historic declines we’ve achieved in recent years in the rates of combustible cigarette use among kids,” FDA Commissioner Scott Gottlieb, MD, said in a statement.
First and foremost, the FDA wants to reduce the lure of e-cigarettes by limiting the variety of flavored products for sale in retail outlets. Under the proposal unveiled Nov. 15, only electronic nicotine delivery systems (ENDS) that are unflavored or have tobacco, mint, or menthol flavors would be widely available. Flavored products – think cherry, cotton candy, and mango – would be sold in age-restricted environments, such as stand-alone tobacco retailers like vape shops. The FDA also seeks more stringent enforcement of age verification on ENDS products sold online.
The proposal also would reexamine regulations governing flavored cigars, with the possible aim of banning them.
“These efforts to address flavors and protect youth would dramatically impact the ability of American kids to access tobacco products that we know are both appealing and addicting,” Dr. Gottlieb said in a statement. “This policy framework reflects a redoubling of the FDA’s efforts to protect kids from all nicotine-containing products.”
In a move that seems to be aimed at youth-oriented products like Juul, the FDA will be seeking to remove from the market any ENDS product that is marketed specifically to young people.
Finally, the FDA intends to pursue regulation that would ban menthol from combustible tobacco products.
“I believe these menthol-flavored products represent one of the most common and pernicious routes by which kids initiate on combustible cigarettes,” Dr. Gottlieb said. “The menthol serves to mask some of the unattractive features of smoking that might otherwise discourage a child from smoking. Moreover, I believe that menthol products disproportionately and adversely affect underserved communities. And as a matter of public health, they exacerbate troubling disparities in health related to race and socioeconomic status.”
The policy shift comes as the Centers for Disease Control and Prevention released data from the 2018 National Youth Tobacco Survey showing that use of e-cigarettes among high schoolers is on the rise, growing from 1.5% in 2011 to 20.8% in 2018. Middle schoolers saw use over the same time period increase from 0.6% to 4.9%.
The rise of current use of e-cigarettes was enough to reverse a declining trend in overall tobacco use in recent years between 2015 and 2017.
“FDA’s enforcement efforts and policy framework would restrict access to most flavored e-cigarettes and limit the chances of youth beginning to use these products, while ensuring the products are available to adult smokers as an alternative to combustible cigarettes,” Alex M. Azar II, secretary of the Department of Health & Human Services, said in a statement supporting the FDA’s efforts. “Our obligation at HHS is always to the public health, and we believe FDA’s goals strike the right public health balance in addressing the multifaceted challenge we have before us today.”
Under Dr. Gottlieb, the FDA has been aggressively pursuing ways to reduce tobacco consumption, targeting both ENDS and combustible tobacco regulations in an effort to limit nicotine exposure and reduce the number of people addicted to nicotine and the health issues that come with it.
The American College of Cardiology voiced its support of the FDA’s actions.
“The FDA’s announcement restricting the sale of flavored e-cigarettes and other tobacco products shows they are ready to do their part in making tobacco products less available to our children,” ACC President C. Michael Valentine, MD, said in a statement, adding that the medical community needs to continue to do its part to make sure tobacco use continues to decline, especially in the nonadult population.
The FDA proposals were published as part of an advance notice of proposed rulemaking in the Federal Register. Comments can be made at www.regulations.gov through June 19.
FDA approves Primatene Mist return
After a long absence, Primatene Mist, an over-the-counter asthma inhaler removed from the market in 2011, is being reintroduced in a metered-dose inhaler with a new, environmentally friendly propellant.
But the inhaler’s comeback may prove as controversial as its removal. Respiratory medicine associations have taken issue with the Food and Drug Administration’s decision, warning patients that asthma is not a “do-it-yourself” disease that can be managed with over-the-counter medications.
The American College of Allergy, Asthma, and Immunology, American College of Chest Physicians, American Lung Association, American Thoracic Society, and the American Association of Asthma Educators have each individually protested the decision, and together sent a joint resolution to FDA decrying it. At the core of their protest are the facts that epinephrine is a symptomatic, not therapeutic, asthma treatment and that racemic epinephrine is not a not a recommended asthma treatment under the National Institutes of Health’s “Guidelines for the Diagnosis and Management of Asthma.”
CHEST has published the following statement: “The American College of Chest Physicians (CHEST) is disappointed with the FDA’s decision to approve over-the counter epinephrine (Primatene® Mist HFA) for the treatment of asthma. CHEST is a nonprofit organization dedicated to advancing best patient outcomes. Our membership of more than 19,000 members from around the world provides patient care in pulmonary, critical care, and sleep medicine.
Asthma is a serious and chronic condition with associated high health-care burden. Care for ALL patients with asthma should be under the guidance of a health-care provider. The majority of asthma patients requires treatment with a controller medication, which is only available by prescription. Frequent rescue inhaler use has been associated with increased morbidity and mortality. Over the counter availability of a reliever medication like Primatene Mist can endanger a patient’s wellbeing by providing temporary relief in symptoms, resulting in delay in seeking medical care.”
The inhaler was pulled from sales as part of an international pact to reduce ozone-depleting substances. The 1989 Montreal Protocol of Substances that Deplete the Ozone Layer and the Clean Air Act of 1990 targeted chlorofluorocarbons among those substances, and epinephrine inhalers that contained CFCs were phased out.
The new Primatene Mist HFA (Amphastar Pharmaceuticals) contains hydrofluoroalkane (HFA) propellants, which are permitted under current international and U.S. law. This puts Primatene in the same category with other inhalers, including albuterol and levalbuterol, which also use HFAs as propellants. Each dose delivers 125 mcg of epinephrine.
The inhaler itself has also been redesigned, according to Theresa Michele, MD, director of the FDA’s Division of Nonprescription Drug Products in the Center for Drug Evaluation and Research. The active ingredient is still epinephrine, albeit a smaller dose than found in the original 200-mcg mist. However, the inhaler needs to be activated before first use and cleaned every day after use to prevent a medication buildup. Like other metered-dose inhalers, it requires a priming spray before the inhalation dose, Dr. Michele noted in her online column.
“The inhaler also needs to be shaken and then sprayed once into the air before each use. It may seem strange to shake and spray the inhaler into the air each time before using it. But these two steps are critical to ensure that the medicine is properly mixed before each dose,” Dr. Michele wrote.
A public statement by FDA Commissioner Scott Gottlieb, MD, and Janet Woodcock, MD, director of the Center for Drug Evaluation and Research, asserted that the inhaler fills a clinical gap for patients with mild to moderate intermittent asthma.
“The scientific information we reviewed to approve the new version of OTC Primatene Mist shows there is a narrow population of those diagnosed with asthma that may benefit from having access to this type of OTC asthma inhaler. But the product has certain cautions. Making sure that patients can understand and apply the instructions for use was a critical consideration for the FDA. The new product is only appropriate for those with a diagnosis of mild, intermittent asthma. Patients with more severe asthma should not rely on it. Instead, they should be working with their health care provider to ensure an appropriate treatment plan for their condition.”
Before this approval, Amphastar had unsuccessfully brought the reformulated Primatene before FDA several times. The move to finally reinstate it comes after a long, and sometimes contentious, debate among patients and FDA’s Nonprescription Drugs and Pulmonary-Allergy Drugs advisory committee. A quick Internet or Facebook search brings up dozens of stories from patients who say they effectively managed their mild to moderate asthma for years with Primatene. Typically, the stories describe changing to prescription asthma medications that, for some, run into the hundreds of dollars per month. Supporters often negatively compare decades of using the inexpensive Primatene with no ill effects to their recent experiences using prescription corticosteroid inhalers.
It was 4 years ago when Amphastar first appeared before the advisory committee with the reformulated inhaler and positive safety and efficacy data. Although agreeing with the efficacy data, the advisory committee voted against approval, because some felt that asthma should always be managed by a physician; an OTC bronchodilator encouraged self-medicating and discouraged patients from seeking medical care, they said.
“On the one hand, it has been stated that a quick-relief medication available OTC is needed for use in low-income, elderly, and uninsured individuals who might otherwise not have access to treatment or be able to see a health care practitioner,” FDA documents noted. “In contrast, there is also a concern that because asthma is a potentially life-threatening condition that should be diagnosed and treated by a health care professional, availability of an OTC bronchodilator product may discourage consumers from seeking appropriate care, resulting in worse asthma outcomes.”
Two years later, the company received another blow to Primatene program. FDA’s Complete Response Letter required Amphastar to make additional changes to the packaging and run a consumer product safety study, intended to show that people could learn to use the metered-dose inhaler correctly.
In Amphastar’s 2018 first-quarter report, however, company CEO Jack Zhang, PhD, finally shared some good news. “We are pleased to announce that we have resubmitted our NDA for Primatene Mist after receiving good results from our recent human factors study. While we don’t have a Prescription Free User Drug Act [PDUFA] date yet, we plan to begin producing inventory in preparation for a launch.”
That day arrived on Nov. 8, when the PDUFA was granted. In their public letter, Dr. Gottlieb and Dr. Woodcock acknowledged the long and difficult approval path and offered reassurance that Primatene is safe and effective.
“For the right patient, our analysis of the data, including new information that was developed since this product was previously on the market, shows that there are no serious safety concerns when Primatene Mist is used as directed. The product is appropriate for mild symptoms of intermittent asthma, however, even patients with mild asthma can have severe exacerbations – so it’s still important to consult a health care provider about appropriate care and have their condition reassessed. And, of course, all patients who experience severe exacerbations should go to the emergency room right away.”
Primatene Mist HFA is intended for the temporary relief of mild symptoms of intermittent asthma (wheezing, tightness of chest, shortness of breath) in patients aged 12 years and older. It should not be considered a replacement for prescription asthma medications. It should be available in stores early next year.
After a long absence, Primatene Mist, an over-the-counter asthma inhaler removed from the market in 2011, is being reintroduced in a metered-dose inhaler with a new, environmentally friendly propellant.
But the inhaler’s comeback may prove as controversial as its removal. Respiratory medicine associations have taken issue with the Food and Drug Administration’s decision, warning patients that asthma is not a “do-it-yourself” disease that can be managed with over-the-counter medications.
The American College of Allergy, Asthma, and Immunology, American College of Chest Physicians, American Lung Association, American Thoracic Society, and the American Association of Asthma Educators have each individually protested the decision, and together sent a joint resolution to FDA decrying it. At the core of their protest are the facts that epinephrine is a symptomatic, not therapeutic, asthma treatment and that racemic epinephrine is not a not a recommended asthma treatment under the National Institutes of Health’s “Guidelines for the Diagnosis and Management of Asthma.”
CHEST has published the following statement: “The American College of Chest Physicians (CHEST) is disappointed with the FDA’s decision to approve over-the counter epinephrine (Primatene® Mist HFA) for the treatment of asthma. CHEST is a nonprofit organization dedicated to advancing best patient outcomes. Our membership of more than 19,000 members from around the world provides patient care in pulmonary, critical care, and sleep medicine.
Asthma is a serious and chronic condition with associated high health-care burden. Care for ALL patients with asthma should be under the guidance of a health-care provider. The majority of asthma patients requires treatment with a controller medication, which is only available by prescription. Frequent rescue inhaler use has been associated with increased morbidity and mortality. Over the counter availability of a reliever medication like Primatene Mist can endanger a patient’s wellbeing by providing temporary relief in symptoms, resulting in delay in seeking medical care.”
The inhaler was pulled from sales as part of an international pact to reduce ozone-depleting substances. The 1989 Montreal Protocol of Substances that Deplete the Ozone Layer and the Clean Air Act of 1990 targeted chlorofluorocarbons among those substances, and epinephrine inhalers that contained CFCs were phased out.
The new Primatene Mist HFA (Amphastar Pharmaceuticals) contains hydrofluoroalkane (HFA) propellants, which are permitted under current international and U.S. law. This puts Primatene in the same category with other inhalers, including albuterol and levalbuterol, which also use HFAs as propellants. Each dose delivers 125 mcg of epinephrine.
The inhaler itself has also been redesigned, according to Theresa Michele, MD, director of the FDA’s Division of Nonprescription Drug Products in the Center for Drug Evaluation and Research. The active ingredient is still epinephrine, albeit a smaller dose than found in the original 200-mcg mist. However, the inhaler needs to be activated before first use and cleaned every day after use to prevent a medication buildup. Like other metered-dose inhalers, it requires a priming spray before the inhalation dose, Dr. Michele noted in her online column.
“The inhaler also needs to be shaken and then sprayed once into the air before each use. It may seem strange to shake and spray the inhaler into the air each time before using it. But these two steps are critical to ensure that the medicine is properly mixed before each dose,” Dr. Michele wrote.
A public statement by FDA Commissioner Scott Gottlieb, MD, and Janet Woodcock, MD, director of the Center for Drug Evaluation and Research, asserted that the inhaler fills a clinical gap for patients with mild to moderate intermittent asthma.
“The scientific information we reviewed to approve the new version of OTC Primatene Mist shows there is a narrow population of those diagnosed with asthma that may benefit from having access to this type of OTC asthma inhaler. But the product has certain cautions. Making sure that patients can understand and apply the instructions for use was a critical consideration for the FDA. The new product is only appropriate for those with a diagnosis of mild, intermittent asthma. Patients with more severe asthma should not rely on it. Instead, they should be working with their health care provider to ensure an appropriate treatment plan for their condition.”
Before this approval, Amphastar had unsuccessfully brought the reformulated Primatene before FDA several times. The move to finally reinstate it comes after a long, and sometimes contentious, debate among patients and FDA’s Nonprescription Drugs and Pulmonary-Allergy Drugs advisory committee. A quick Internet or Facebook search brings up dozens of stories from patients who say they effectively managed their mild to moderate asthma for years with Primatene. Typically, the stories describe changing to prescription asthma medications that, for some, run into the hundreds of dollars per month. Supporters often negatively compare decades of using the inexpensive Primatene with no ill effects to their recent experiences using prescription corticosteroid inhalers.
It was 4 years ago when Amphastar first appeared before the advisory committee with the reformulated inhaler and positive safety and efficacy data. Although agreeing with the efficacy data, the advisory committee voted against approval, because some felt that asthma should always be managed by a physician; an OTC bronchodilator encouraged self-medicating and discouraged patients from seeking medical care, they said.
“On the one hand, it has been stated that a quick-relief medication available OTC is needed for use in low-income, elderly, and uninsured individuals who might otherwise not have access to treatment or be able to see a health care practitioner,” FDA documents noted. “In contrast, there is also a concern that because asthma is a potentially life-threatening condition that should be diagnosed and treated by a health care professional, availability of an OTC bronchodilator product may discourage consumers from seeking appropriate care, resulting in worse asthma outcomes.”
Two years later, the company received another blow to Primatene program. FDA’s Complete Response Letter required Amphastar to make additional changes to the packaging and run a consumer product safety study, intended to show that people could learn to use the metered-dose inhaler correctly.
In Amphastar’s 2018 first-quarter report, however, company CEO Jack Zhang, PhD, finally shared some good news. “We are pleased to announce that we have resubmitted our NDA for Primatene Mist after receiving good results from our recent human factors study. While we don’t have a Prescription Free User Drug Act [PDUFA] date yet, we plan to begin producing inventory in preparation for a launch.”
That day arrived on Nov. 8, when the PDUFA was granted. In their public letter, Dr. Gottlieb and Dr. Woodcock acknowledged the long and difficult approval path and offered reassurance that Primatene is safe and effective.
“For the right patient, our analysis of the data, including new information that was developed since this product was previously on the market, shows that there are no serious safety concerns when Primatene Mist is used as directed. The product is appropriate for mild symptoms of intermittent asthma, however, even patients with mild asthma can have severe exacerbations – so it’s still important to consult a health care provider about appropriate care and have their condition reassessed. And, of course, all patients who experience severe exacerbations should go to the emergency room right away.”
Primatene Mist HFA is intended for the temporary relief of mild symptoms of intermittent asthma (wheezing, tightness of chest, shortness of breath) in patients aged 12 years and older. It should not be considered a replacement for prescription asthma medications. It should be available in stores early next year.
After a long absence, Primatene Mist, an over-the-counter asthma inhaler removed from the market in 2011, is being reintroduced in a metered-dose inhaler with a new, environmentally friendly propellant.
But the inhaler’s comeback may prove as controversial as its removal. Respiratory medicine associations have taken issue with the Food and Drug Administration’s decision, warning patients that asthma is not a “do-it-yourself” disease that can be managed with over-the-counter medications.
The American College of Allergy, Asthma, and Immunology, American College of Chest Physicians, American Lung Association, American Thoracic Society, and the American Association of Asthma Educators have each individually protested the decision, and together sent a joint resolution to FDA decrying it. At the core of their protest are the facts that epinephrine is a symptomatic, not therapeutic, asthma treatment and that racemic epinephrine is not a not a recommended asthma treatment under the National Institutes of Health’s “Guidelines for the Diagnosis and Management of Asthma.”
CHEST has published the following statement: “The American College of Chest Physicians (CHEST) is disappointed with the FDA’s decision to approve over-the counter epinephrine (Primatene® Mist HFA) for the treatment of asthma. CHEST is a nonprofit organization dedicated to advancing best patient outcomes. Our membership of more than 19,000 members from around the world provides patient care in pulmonary, critical care, and sleep medicine.
Asthma is a serious and chronic condition with associated high health-care burden. Care for ALL patients with asthma should be under the guidance of a health-care provider. The majority of asthma patients requires treatment with a controller medication, which is only available by prescription. Frequent rescue inhaler use has been associated with increased morbidity and mortality. Over the counter availability of a reliever medication like Primatene Mist can endanger a patient’s wellbeing by providing temporary relief in symptoms, resulting in delay in seeking medical care.”
The inhaler was pulled from sales as part of an international pact to reduce ozone-depleting substances. The 1989 Montreal Protocol of Substances that Deplete the Ozone Layer and the Clean Air Act of 1990 targeted chlorofluorocarbons among those substances, and epinephrine inhalers that contained CFCs were phased out.
The new Primatene Mist HFA (Amphastar Pharmaceuticals) contains hydrofluoroalkane (HFA) propellants, which are permitted under current international and U.S. law. This puts Primatene in the same category with other inhalers, including albuterol and levalbuterol, which also use HFAs as propellants. Each dose delivers 125 mcg of epinephrine.
The inhaler itself has also been redesigned, according to Theresa Michele, MD, director of the FDA’s Division of Nonprescription Drug Products in the Center for Drug Evaluation and Research. The active ingredient is still epinephrine, albeit a smaller dose than found in the original 200-mcg mist. However, the inhaler needs to be activated before first use and cleaned every day after use to prevent a medication buildup. Like other metered-dose inhalers, it requires a priming spray before the inhalation dose, Dr. Michele noted in her online column.
“The inhaler also needs to be shaken and then sprayed once into the air before each use. It may seem strange to shake and spray the inhaler into the air each time before using it. But these two steps are critical to ensure that the medicine is properly mixed before each dose,” Dr. Michele wrote.
A public statement by FDA Commissioner Scott Gottlieb, MD, and Janet Woodcock, MD, director of the Center for Drug Evaluation and Research, asserted that the inhaler fills a clinical gap for patients with mild to moderate intermittent asthma.
“The scientific information we reviewed to approve the new version of OTC Primatene Mist shows there is a narrow population of those diagnosed with asthma that may benefit from having access to this type of OTC asthma inhaler. But the product has certain cautions. Making sure that patients can understand and apply the instructions for use was a critical consideration for the FDA. The new product is only appropriate for those with a diagnosis of mild, intermittent asthma. Patients with more severe asthma should not rely on it. Instead, they should be working with their health care provider to ensure an appropriate treatment plan for their condition.”
Before this approval, Amphastar had unsuccessfully brought the reformulated Primatene before FDA several times. The move to finally reinstate it comes after a long, and sometimes contentious, debate among patients and FDA’s Nonprescription Drugs and Pulmonary-Allergy Drugs advisory committee. A quick Internet or Facebook search brings up dozens of stories from patients who say they effectively managed their mild to moderate asthma for years with Primatene. Typically, the stories describe changing to prescription asthma medications that, for some, run into the hundreds of dollars per month. Supporters often negatively compare decades of using the inexpensive Primatene with no ill effects to their recent experiences using prescription corticosteroid inhalers.
It was 4 years ago when Amphastar first appeared before the advisory committee with the reformulated inhaler and positive safety and efficacy data. Although agreeing with the efficacy data, the advisory committee voted against approval, because some felt that asthma should always be managed by a physician; an OTC bronchodilator encouraged self-medicating and discouraged patients from seeking medical care, they said.
“On the one hand, it has been stated that a quick-relief medication available OTC is needed for use in low-income, elderly, and uninsured individuals who might otherwise not have access to treatment or be able to see a health care practitioner,” FDA documents noted. “In contrast, there is also a concern that because asthma is a potentially life-threatening condition that should be diagnosed and treated by a health care professional, availability of an OTC bronchodilator product may discourage consumers from seeking appropriate care, resulting in worse asthma outcomes.”
Two years later, the company received another blow to Primatene program. FDA’s Complete Response Letter required Amphastar to make additional changes to the packaging and run a consumer product safety study, intended to show that people could learn to use the metered-dose inhaler correctly.
In Amphastar’s 2018 first-quarter report, however, company CEO Jack Zhang, PhD, finally shared some good news. “We are pleased to announce that we have resubmitted our NDA for Primatene Mist after receiving good results from our recent human factors study. While we don’t have a Prescription Free User Drug Act [PDUFA] date yet, we plan to begin producing inventory in preparation for a launch.”
That day arrived on Nov. 8, when the PDUFA was granted. In their public letter, Dr. Gottlieb and Dr. Woodcock acknowledged the long and difficult approval path and offered reassurance that Primatene is safe and effective.
“For the right patient, our analysis of the data, including new information that was developed since this product was previously on the market, shows that there are no serious safety concerns when Primatene Mist is used as directed. The product is appropriate for mild symptoms of intermittent asthma, however, even patients with mild asthma can have severe exacerbations – so it’s still important to consult a health care provider about appropriate care and have their condition reassessed. And, of course, all patients who experience severe exacerbations should go to the emergency room right away.”
Primatene Mist HFA is intended for the temporary relief of mild symptoms of intermittent asthma (wheezing, tightness of chest, shortness of breath) in patients aged 12 years and older. It should not be considered a replacement for prescription asthma medications. It should be available in stores early next year.
New pediatric therapies show promise for influenza, multidrug-resistant pathogens
ORLANDO – John S. Bradley, MD, said at the annual meeting of the American Academy of Pediatrics.
Dr. Bradley, director of the division of infectious diseases at Rady Children’s Hospital–San Diego, discussed a therapy for influenza, baloxavir, which was recently approved as a fast-acting single-dose medication and currently is under study in children. Also, a recent double-blind, phase 3 trial in the New England Journal of Medicine recruited patients as young as 12 years old. In the study, patients in the intervention group resolved their fever in median 25 hours, compared with 42 hours in the placebo group. Baloxavir better reduced viral load at day 2, compared with oseltamivir and placebo, but there was a similar alleviation of symptoms between both groups. There was a greater incidence of nausea and vomiting among the oseltamivir group, while the baloxavir group had a higher rate of diarrhea (N Engl J Med 2018;379:913-23).
However, Dr. Bradley noted baloxavir is much more expensive than oseltamivir, which may not justify the better tolerance of the drug for influenza treatment.
You don’t get better with it faster, so I’m not going to be recommending you all run to baloxavir this flu season for kids 12 years of age and older,” Dr. Bradley said. “I think oseltamivir is still fine, unless we end up with oseltamivir resistance.”
Solithromycin, an intravenous and oral fluoroketolide, has shown promising results against gram-positive and gram-negative pathogens for community-acquired pneumonia and other infections. During the drug’s study period, Cempra sold solithromycin to Melinta. However, one trial showed elevated liver functions in a higher number of patients than expected, and the Food and Drug Administration asked Melinta to conduct additional studies. Investigations on solithromycin have currently stopped until Melinta secures funding. “Until they get better resources, this particular drug is on hold, but you’ll see it again, I’m sure,” said Dr. Bradley, who also is professor and chief of the division of infectious diseases at the University of California, San Diego.
Dr. Bradley also discussed the efficacy of tedizolid, a protein synthesis inhibitor similar to linezolid approved in adults for the treatment of skin infections. He noted tedizolid is more active than linezolid, but the treatment course is a shorter dose for a shorter amount of time. Compared with linezolid, which can cause thrombocytopenia or neutropenia if taken for more than 10 days to 14 days, there also are fewer side effects.
“The tedizolid is much, much safer,” Dr. Bradley said, who added that trials for efficacy of tedizolid are currently underway in pediatric patients. “We’re hoping that will end up being the pediatric oxazolidinone.”
Other investigative therapies approved for adults and under study for use in children include ceftazidime/avibactam for treatment of urinary tract and complicated intra-abdominal infections, which is effective against meropenem-resistant Enterobacteriaceae and resistant Escherichia coli with extended-spectrum beta-lactamases (ESBL); ceftolozane/tazobactam has also been approved for adults, is pending approval in pediatric patients, and is active against ESBLs such as Pseudomonas; and meropenem/vaborbactam, which is active against Klebsiella pneumoniae carbapenemase (KPC)–producing isolates. Plazomicin, an aminoglycoside similar to gentamicin used to treat KPC-producing isolates, is stable against enzymes that degrade gentamicin and tobramycin.
Therapies currently under study for adults and being considered for children include imipenem/relebactam for treatment against E. coli, Enterobacter species, and KPC-producing isolates, and cefiderocol, a siderophore cephalosporin antibiotic – commonly described as a “Trojan horse” antibiotic because it binds to iron and is actively transported into the organism – is effective against Pseudomonas and has finished phase 2 trials in adults, with researchers looking to do single-dose trials in children, Dr. Bradley noted.
More experimentally, phage therapy for multidrug-resistant Acinetobacter baumannii proved effective in a 68-year-old patient with necrotizing pancreatitis who continued to deteriorate over a 4-month period despite multiple courses of antibiotics and attempted drainage of a pancreatic pseudocyst. Researchers selected a phage-specific bacterium with specificity for A. baumannii and cured him. “This is like science fiction,” Dr. Bradley said.
Dr. Bradley reported no relevant conflicts of interest.
ORLANDO – John S. Bradley, MD, said at the annual meeting of the American Academy of Pediatrics.
Dr. Bradley, director of the division of infectious diseases at Rady Children’s Hospital–San Diego, discussed a therapy for influenza, baloxavir, which was recently approved as a fast-acting single-dose medication and currently is under study in children. Also, a recent double-blind, phase 3 trial in the New England Journal of Medicine recruited patients as young as 12 years old. In the study, patients in the intervention group resolved their fever in median 25 hours, compared with 42 hours in the placebo group. Baloxavir better reduced viral load at day 2, compared with oseltamivir and placebo, but there was a similar alleviation of symptoms between both groups. There was a greater incidence of nausea and vomiting among the oseltamivir group, while the baloxavir group had a higher rate of diarrhea (N Engl J Med 2018;379:913-23).
However, Dr. Bradley noted baloxavir is much more expensive than oseltamivir, which may not justify the better tolerance of the drug for influenza treatment.
You don’t get better with it faster, so I’m not going to be recommending you all run to baloxavir this flu season for kids 12 years of age and older,” Dr. Bradley said. “I think oseltamivir is still fine, unless we end up with oseltamivir resistance.”
Solithromycin, an intravenous and oral fluoroketolide, has shown promising results against gram-positive and gram-negative pathogens for community-acquired pneumonia and other infections. During the drug’s study period, Cempra sold solithromycin to Melinta. However, one trial showed elevated liver functions in a higher number of patients than expected, and the Food and Drug Administration asked Melinta to conduct additional studies. Investigations on solithromycin have currently stopped until Melinta secures funding. “Until they get better resources, this particular drug is on hold, but you’ll see it again, I’m sure,” said Dr. Bradley, who also is professor and chief of the division of infectious diseases at the University of California, San Diego.
Dr. Bradley also discussed the efficacy of tedizolid, a protein synthesis inhibitor similar to linezolid approved in adults for the treatment of skin infections. He noted tedizolid is more active than linezolid, but the treatment course is a shorter dose for a shorter amount of time. Compared with linezolid, which can cause thrombocytopenia or neutropenia if taken for more than 10 days to 14 days, there also are fewer side effects.
“The tedizolid is much, much safer,” Dr. Bradley said, who added that trials for efficacy of tedizolid are currently underway in pediatric patients. “We’re hoping that will end up being the pediatric oxazolidinone.”
Other investigative therapies approved for adults and under study for use in children include ceftazidime/avibactam for treatment of urinary tract and complicated intra-abdominal infections, which is effective against meropenem-resistant Enterobacteriaceae and resistant Escherichia coli with extended-spectrum beta-lactamases (ESBL); ceftolozane/tazobactam has also been approved for adults, is pending approval in pediatric patients, and is active against ESBLs such as Pseudomonas; and meropenem/vaborbactam, which is active against Klebsiella pneumoniae carbapenemase (KPC)–producing isolates. Plazomicin, an aminoglycoside similar to gentamicin used to treat KPC-producing isolates, is stable against enzymes that degrade gentamicin and tobramycin.
Therapies currently under study for adults and being considered for children include imipenem/relebactam for treatment against E. coli, Enterobacter species, and KPC-producing isolates, and cefiderocol, a siderophore cephalosporin antibiotic – commonly described as a “Trojan horse” antibiotic because it binds to iron and is actively transported into the organism – is effective against Pseudomonas and has finished phase 2 trials in adults, with researchers looking to do single-dose trials in children, Dr. Bradley noted.
More experimentally, phage therapy for multidrug-resistant Acinetobacter baumannii proved effective in a 68-year-old patient with necrotizing pancreatitis who continued to deteriorate over a 4-month period despite multiple courses of antibiotics and attempted drainage of a pancreatic pseudocyst. Researchers selected a phage-specific bacterium with specificity for A. baumannii and cured him. “This is like science fiction,” Dr. Bradley said.
Dr. Bradley reported no relevant conflicts of interest.
ORLANDO – John S. Bradley, MD, said at the annual meeting of the American Academy of Pediatrics.
Dr. Bradley, director of the division of infectious diseases at Rady Children’s Hospital–San Diego, discussed a therapy for influenza, baloxavir, which was recently approved as a fast-acting single-dose medication and currently is under study in children. Also, a recent double-blind, phase 3 trial in the New England Journal of Medicine recruited patients as young as 12 years old. In the study, patients in the intervention group resolved their fever in median 25 hours, compared with 42 hours in the placebo group. Baloxavir better reduced viral load at day 2, compared with oseltamivir and placebo, but there was a similar alleviation of symptoms between both groups. There was a greater incidence of nausea and vomiting among the oseltamivir group, while the baloxavir group had a higher rate of diarrhea (N Engl J Med 2018;379:913-23).
However, Dr. Bradley noted baloxavir is much more expensive than oseltamivir, which may not justify the better tolerance of the drug for influenza treatment.
You don’t get better with it faster, so I’m not going to be recommending you all run to baloxavir this flu season for kids 12 years of age and older,” Dr. Bradley said. “I think oseltamivir is still fine, unless we end up with oseltamivir resistance.”
Solithromycin, an intravenous and oral fluoroketolide, has shown promising results against gram-positive and gram-negative pathogens for community-acquired pneumonia and other infections. During the drug’s study period, Cempra sold solithromycin to Melinta. However, one trial showed elevated liver functions in a higher number of patients than expected, and the Food and Drug Administration asked Melinta to conduct additional studies. Investigations on solithromycin have currently stopped until Melinta secures funding. “Until they get better resources, this particular drug is on hold, but you’ll see it again, I’m sure,” said Dr. Bradley, who also is professor and chief of the division of infectious diseases at the University of California, San Diego.
Dr. Bradley also discussed the efficacy of tedizolid, a protein synthesis inhibitor similar to linezolid approved in adults for the treatment of skin infections. He noted tedizolid is more active than linezolid, but the treatment course is a shorter dose for a shorter amount of time. Compared with linezolid, which can cause thrombocytopenia or neutropenia if taken for more than 10 days to 14 days, there also are fewer side effects.
“The tedizolid is much, much safer,” Dr. Bradley said, who added that trials for efficacy of tedizolid are currently underway in pediatric patients. “We’re hoping that will end up being the pediatric oxazolidinone.”
Other investigative therapies approved for adults and under study for use in children include ceftazidime/avibactam for treatment of urinary tract and complicated intra-abdominal infections, which is effective against meropenem-resistant Enterobacteriaceae and resistant Escherichia coli with extended-spectrum beta-lactamases (ESBL); ceftolozane/tazobactam has also been approved for adults, is pending approval in pediatric patients, and is active against ESBLs such as Pseudomonas; and meropenem/vaborbactam, which is active against Klebsiella pneumoniae carbapenemase (KPC)–producing isolates. Plazomicin, an aminoglycoside similar to gentamicin used to treat KPC-producing isolates, is stable against enzymes that degrade gentamicin and tobramycin.
Therapies currently under study for adults and being considered for children include imipenem/relebactam for treatment against E. coli, Enterobacter species, and KPC-producing isolates, and cefiderocol, a siderophore cephalosporin antibiotic – commonly described as a “Trojan horse” antibiotic because it binds to iron and is actively transported into the organism – is effective against Pseudomonas and has finished phase 2 trials in adults, with researchers looking to do single-dose trials in children, Dr. Bradley noted.
More experimentally, phage therapy for multidrug-resistant Acinetobacter baumannii proved effective in a 68-year-old patient with necrotizing pancreatitis who continued to deteriorate over a 4-month period despite multiple courses of antibiotics and attempted drainage of a pancreatic pseudocyst. Researchers selected a phage-specific bacterium with specificity for A. baumannii and cured him. “This is like science fiction,” Dr. Bradley said.
Dr. Bradley reported no relevant conflicts of interest.
EXPERT ANALYSIS FROM AAP 18
More acute flaccid myelitis cases confirmed by CDC
Acute flaccid myelitis (AFM) has stricken 90 patients in the United States this year and another 252 cases are being investigated, according to new data from the Centers for Disease Control and Prevention.
The number of confirmed cases is triple that seen in 2017.
Nearly all of the patients (90%) were children aged 2-8 years, and 99% experienced a fever and /or respiratory illness 7-10 days before the onset of symptoms. But although the prodrome and seasonality of AFM suggest an infective process, only 54% of the patients tested positive for the virus, Nancy Messonnier, MD, said during a briefing held by CDC officials. The most common findings were the enteroviruses EV-A71 (29%) and EV-D68 (37%); other viruses were recovered in the remaining pathogen-positive cases.
It’s not at all clear that these were causative agents, said Dr. Messonnier, director of the National Center for Immunization and Respiratory Diseases.
“At this time of year lots of children have a fever and respiratory infections,” she said. AFM may be caused by one of the identified viruses, a still-undetected pathogen, or a pathogen hiding in untested tissue. “Or, it could be an infection that’s kicking off an immune process,” attacking gray matter in the spinal cord.
The reported increase in cases must be viewed cautiously, Dr. Messonnier said. Physicians are becoming more aware of AFM, so the spike could represent an increase in reporting as well as actual incidence.
It’s not clear why the disease manifests almost exclusively in children, Dr. Messonnier said. Nor do health officials have much of a grasp on AFM’s long-term sequelae.
“We know that patients can recover fully, but at least half don’t, and some of those have serious sequelae. Unfortunately, we have not been following every patient, so this is a gap in our knowledge.”
A newly created national task force will examine AFM’s long-term effects, Dr. Messonnier said. The task force will also look at mortality; health departments across the country will examine mortality records to identify any past deaths preceded by AFM-like symptoms.
“One of the reasons we have convened this task force is to think about this hypothesis [of an autoimmune syndrome]. We have not backed off on the idea of an infectious organism causing it, but we are thinking more broadly,” Dr. Messonnier said.
Some anti-immunization groups are blaming vaccines for the disease, noting that several childhood vaccines list encephalomyelitis and transverse myelitis as possible adverse events.
“We are investigating every one of the cases in this and prior years and have a list of hypotheses based on the epidemiology,” Dr. Messonnier said. “I would say toxins are low on that list. Many of the children may have been vaccinated [before developing AFM] and that is something we will look at, but for now we recommend that all children should be vaccinated” according to the recommended schedule.
Additional details were published on 80 of the cases. Patients’ mean age was 4 years; 59% were male. Symptoms suggesting a viral illness occurred in 99%; these included fever (81%), cough, rhinorrhea, and congestion (78%), and vomiting and diarrhea (38%).
AFM symptoms varied; 47% had only upper limb involvement, 9% only lower limb, 15% two or three upper, and 29% all four limbs. All the patients with confirmed AFM were hospitalized, and 59% treated in intensive care units. There were no deaths (MMWR. 2018;ePub:13 November. DOI: http://dx.doi.org/10.15585/mmwr.mm6745e1).
AFM remains extremely rare, Dr. Messonnier said. But physicians should be alert for any signs of sudden limb weakness in children and report those immediately. The workup should include questions about recent fever with or without respiratory or gastrointestinal symptoms. Prompt collection of viral testing samples (cerebrospinal fluid, serum, respiratory, and stool specimens) is critical.
Additional information for health care professionals is available on the CDC AFM web page.
Acute flaccid myelitis (AFM) has stricken 90 patients in the United States this year and another 252 cases are being investigated, according to new data from the Centers for Disease Control and Prevention.
The number of confirmed cases is triple that seen in 2017.
Nearly all of the patients (90%) were children aged 2-8 years, and 99% experienced a fever and /or respiratory illness 7-10 days before the onset of symptoms. But although the prodrome and seasonality of AFM suggest an infective process, only 54% of the patients tested positive for the virus, Nancy Messonnier, MD, said during a briefing held by CDC officials. The most common findings were the enteroviruses EV-A71 (29%) and EV-D68 (37%); other viruses were recovered in the remaining pathogen-positive cases.
It’s not at all clear that these were causative agents, said Dr. Messonnier, director of the National Center for Immunization and Respiratory Diseases.
“At this time of year lots of children have a fever and respiratory infections,” she said. AFM may be caused by one of the identified viruses, a still-undetected pathogen, or a pathogen hiding in untested tissue. “Or, it could be an infection that’s kicking off an immune process,” attacking gray matter in the spinal cord.
The reported increase in cases must be viewed cautiously, Dr. Messonnier said. Physicians are becoming more aware of AFM, so the spike could represent an increase in reporting as well as actual incidence.
It’s not clear why the disease manifests almost exclusively in children, Dr. Messonnier said. Nor do health officials have much of a grasp on AFM’s long-term sequelae.
“We know that patients can recover fully, but at least half don’t, and some of those have serious sequelae. Unfortunately, we have not been following every patient, so this is a gap in our knowledge.”
A newly created national task force will examine AFM’s long-term effects, Dr. Messonnier said. The task force will also look at mortality; health departments across the country will examine mortality records to identify any past deaths preceded by AFM-like symptoms.
“One of the reasons we have convened this task force is to think about this hypothesis [of an autoimmune syndrome]. We have not backed off on the idea of an infectious organism causing it, but we are thinking more broadly,” Dr. Messonnier said.
Some anti-immunization groups are blaming vaccines for the disease, noting that several childhood vaccines list encephalomyelitis and transverse myelitis as possible adverse events.
“We are investigating every one of the cases in this and prior years and have a list of hypotheses based on the epidemiology,” Dr. Messonnier said. “I would say toxins are low on that list. Many of the children may have been vaccinated [before developing AFM] and that is something we will look at, but for now we recommend that all children should be vaccinated” according to the recommended schedule.
Additional details were published on 80 of the cases. Patients’ mean age was 4 years; 59% were male. Symptoms suggesting a viral illness occurred in 99%; these included fever (81%), cough, rhinorrhea, and congestion (78%), and vomiting and diarrhea (38%).
AFM symptoms varied; 47% had only upper limb involvement, 9% only lower limb, 15% two or three upper, and 29% all four limbs. All the patients with confirmed AFM were hospitalized, and 59% treated in intensive care units. There were no deaths (MMWR. 2018;ePub:13 November. DOI: http://dx.doi.org/10.15585/mmwr.mm6745e1).
AFM remains extremely rare, Dr. Messonnier said. But physicians should be alert for any signs of sudden limb weakness in children and report those immediately. The workup should include questions about recent fever with or without respiratory or gastrointestinal symptoms. Prompt collection of viral testing samples (cerebrospinal fluid, serum, respiratory, and stool specimens) is critical.
Additional information for health care professionals is available on the CDC AFM web page.
Acute flaccid myelitis (AFM) has stricken 90 patients in the United States this year and another 252 cases are being investigated, according to new data from the Centers for Disease Control and Prevention.
The number of confirmed cases is triple that seen in 2017.
Nearly all of the patients (90%) were children aged 2-8 years, and 99% experienced a fever and /or respiratory illness 7-10 days before the onset of symptoms. But although the prodrome and seasonality of AFM suggest an infective process, only 54% of the patients tested positive for the virus, Nancy Messonnier, MD, said during a briefing held by CDC officials. The most common findings were the enteroviruses EV-A71 (29%) and EV-D68 (37%); other viruses were recovered in the remaining pathogen-positive cases.
It’s not at all clear that these were causative agents, said Dr. Messonnier, director of the National Center for Immunization and Respiratory Diseases.
“At this time of year lots of children have a fever and respiratory infections,” she said. AFM may be caused by one of the identified viruses, a still-undetected pathogen, or a pathogen hiding in untested tissue. “Or, it could be an infection that’s kicking off an immune process,” attacking gray matter in the spinal cord.
The reported increase in cases must be viewed cautiously, Dr. Messonnier said. Physicians are becoming more aware of AFM, so the spike could represent an increase in reporting as well as actual incidence.
It’s not clear why the disease manifests almost exclusively in children, Dr. Messonnier said. Nor do health officials have much of a grasp on AFM’s long-term sequelae.
“We know that patients can recover fully, but at least half don’t, and some of those have serious sequelae. Unfortunately, we have not been following every patient, so this is a gap in our knowledge.”
A newly created national task force will examine AFM’s long-term effects, Dr. Messonnier said. The task force will also look at mortality; health departments across the country will examine mortality records to identify any past deaths preceded by AFM-like symptoms.
“One of the reasons we have convened this task force is to think about this hypothesis [of an autoimmune syndrome]. We have not backed off on the idea of an infectious organism causing it, but we are thinking more broadly,” Dr. Messonnier said.
Some anti-immunization groups are blaming vaccines for the disease, noting that several childhood vaccines list encephalomyelitis and transverse myelitis as possible adverse events.
“We are investigating every one of the cases in this and prior years and have a list of hypotheses based on the epidemiology,” Dr. Messonnier said. “I would say toxins are low on that list. Many of the children may have been vaccinated [before developing AFM] and that is something we will look at, but for now we recommend that all children should be vaccinated” according to the recommended schedule.
Additional details were published on 80 of the cases. Patients’ mean age was 4 years; 59% were male. Symptoms suggesting a viral illness occurred in 99%; these included fever (81%), cough, rhinorrhea, and congestion (78%), and vomiting and diarrhea (38%).
AFM symptoms varied; 47% had only upper limb involvement, 9% only lower limb, 15% two or three upper, and 29% all four limbs. All the patients with confirmed AFM were hospitalized, and 59% treated in intensive care units. There were no deaths (MMWR. 2018;ePub:13 November. DOI: http://dx.doi.org/10.15585/mmwr.mm6745e1).
AFM remains extremely rare, Dr. Messonnier said. But physicians should be alert for any signs of sudden limb weakness in children and report those immediately. The workup should include questions about recent fever with or without respiratory or gastrointestinal symptoms. Prompt collection of viral testing samples (cerebrospinal fluid, serum, respiratory, and stool specimens) is critical.
Additional information for health care professionals is available on the CDC AFM web page.
FROM A CDC BRIEFING
FDA approves Yupelri for COPD maintenance therapy
The Food and Drug Administration has approved Yupelri (revefenacin) for maintenance therapy of patients with chronic obstructive pulmonary disease (COPD).
Revefenacin is a long-acting muscarinic antagonist aimed at improving the lung function of patients with COPD. Yupelri is an inhalation solution administered once daily through a standard jet nebulizer.
The most common adverse events associated with Yupelri are cough, nasopharyngitis, upper respiratory tract infection, headache, and back pain. Patients receiving other anticholinergic-containing drugs or OATP1B1 and OATP1B3 inhibitors should not receive Yupelri.
“Patients should also be alert for signs and symptoms of acute narrow-angle glaucoma [e.g., eye pain or discomfort, blurred vision, visual changes]. Patients should consult a healthcare professional immediately if any of these signs or symptoms develop,” the FDA said in the press release.
The expanded label for Yupelri can be found on the FDA website.
The Food and Drug Administration has approved Yupelri (revefenacin) for maintenance therapy of patients with chronic obstructive pulmonary disease (COPD).
Revefenacin is a long-acting muscarinic antagonist aimed at improving the lung function of patients with COPD. Yupelri is an inhalation solution administered once daily through a standard jet nebulizer.
The most common adverse events associated with Yupelri are cough, nasopharyngitis, upper respiratory tract infection, headache, and back pain. Patients receiving other anticholinergic-containing drugs or OATP1B1 and OATP1B3 inhibitors should not receive Yupelri.
“Patients should also be alert for signs and symptoms of acute narrow-angle glaucoma [e.g., eye pain or discomfort, blurred vision, visual changes]. Patients should consult a healthcare professional immediately if any of these signs or symptoms develop,” the FDA said in the press release.
The expanded label for Yupelri can be found on the FDA website.
The Food and Drug Administration has approved Yupelri (revefenacin) for maintenance therapy of patients with chronic obstructive pulmonary disease (COPD).
Revefenacin is a long-acting muscarinic antagonist aimed at improving the lung function of patients with COPD. Yupelri is an inhalation solution administered once daily through a standard jet nebulizer.
The most common adverse events associated with Yupelri are cough, nasopharyngitis, upper respiratory tract infection, headache, and back pain. Patients receiving other anticholinergic-containing drugs or OATP1B1 and OATP1B3 inhibitors should not receive Yupelri.
“Patients should also be alert for signs and symptoms of acute narrow-angle glaucoma [e.g., eye pain or discomfort, blurred vision, visual changes]. Patients should consult a healthcare professional immediately if any of these signs or symptoms develop,” the FDA said in the press release.
The expanded label for Yupelri can be found on the FDA website.
Cigarette smoking at lowest level ever
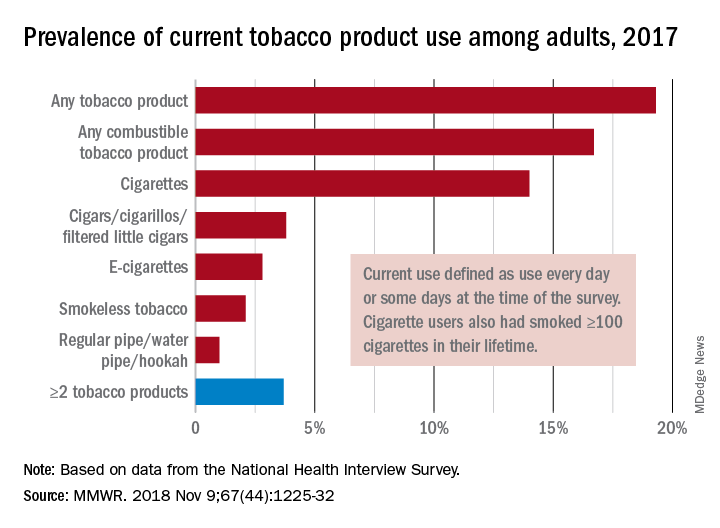
“This new all-time low in cigarette smoking among U.S. adults is a tremendous public health accomplishment, and it demonstrates the importance of continued proven strategies to reduce smoking,” CDC Director Robert Redfield said in a written statement.
In 2017, 19.3% of adults aged 18 years and older – approximately 47.4 million Americans – reported current use of some type of tobacco product, and current use of combustible tobacco was 16.7%, Teresa W. Wang, PhD, of the CDC’s National Center for Chronic Disease Prevention and Health Promotion, Atlanta, and her associates reported in the Morbidity and Mortality Weekly Report. Current use was defined as use every day or some days, with an added requirement of at least 100 cigarettes in a lifetime added for cigarette smokers.
Data from the National Health Interview Survey showed that from 2016 to 2017, current use declined for any tobacco product, any combustible tobacco product, cigarettes, smokeless tobacco, and the combination of two or more tobacco products. The most common combination in 2017 was cigarettes and e-cigarettes, which was reported by 30.1% of the 9 million adults who used more than one product, Dr. Wang and her associates said.
Prevalence of current tobacco use was higher among men than women (24.8% vs. 14.2%), and adults aged 25-44 years (22.5%) had the highest level by age, followed by those aged 45-64 years (21.3%), 18-24 years (18.3%), and 65 years or older (11%). Use by race/ethnicity was highest among American Indian/Alaska Natives (29.8%), with the Midwest putting up the highest prevalence by region at 23.5%, they said.
“Although cigarette smoking among U.S. adults has declined considerably, tobacco products have evolved in recent years to include various combustible, noncombustible, and electronic products,” Dr. Wang and her associates wrote. “Implementation of evidence-based tobacco control interventions that address the diversity of tobacco products used by U.S. adults, in coordination with regulation of tobacco product manufacturing, marketing, and sales, can reduce tobacco-related disease and death in the United States.”
SOURCE: Wang TW et al. MMWR. 2018 Nov 9;67[44]:1225-32.

“This new all-time low in cigarette smoking among U.S. adults is a tremendous public health accomplishment, and it demonstrates the importance of continued proven strategies to reduce smoking,” CDC Director Robert Redfield said in a written statement.
In 2017, 19.3% of adults aged 18 years and older – approximately 47.4 million Americans – reported current use of some type of tobacco product, and current use of combustible tobacco was 16.7%, Teresa W. Wang, PhD, of the CDC’s National Center for Chronic Disease Prevention and Health Promotion, Atlanta, and her associates reported in the Morbidity and Mortality Weekly Report. Current use was defined as use every day or some days, with an added requirement of at least 100 cigarettes in a lifetime added for cigarette smokers.
Data from the National Health Interview Survey showed that from 2016 to 2017, current use declined for any tobacco product, any combustible tobacco product, cigarettes, smokeless tobacco, and the combination of two or more tobacco products. The most common combination in 2017 was cigarettes and e-cigarettes, which was reported by 30.1% of the 9 million adults who used more than one product, Dr. Wang and her associates said.
Prevalence of current tobacco use was higher among men than women (24.8% vs. 14.2%), and adults aged 25-44 years (22.5%) had the highest level by age, followed by those aged 45-64 years (21.3%), 18-24 years (18.3%), and 65 years or older (11%). Use by race/ethnicity was highest among American Indian/Alaska Natives (29.8%), with the Midwest putting up the highest prevalence by region at 23.5%, they said.
“Although cigarette smoking among U.S. adults has declined considerably, tobacco products have evolved in recent years to include various combustible, noncombustible, and electronic products,” Dr. Wang and her associates wrote. “Implementation of evidence-based tobacco control interventions that address the diversity of tobacco products used by U.S. adults, in coordination with regulation of tobacco product manufacturing, marketing, and sales, can reduce tobacco-related disease and death in the United States.”
SOURCE: Wang TW et al. MMWR. 2018 Nov 9;67[44]:1225-32.

“This new all-time low in cigarette smoking among U.S. adults is a tremendous public health accomplishment, and it demonstrates the importance of continued proven strategies to reduce smoking,” CDC Director Robert Redfield said in a written statement.
In 2017, 19.3% of adults aged 18 years and older – approximately 47.4 million Americans – reported current use of some type of tobacco product, and current use of combustible tobacco was 16.7%, Teresa W. Wang, PhD, of the CDC’s National Center for Chronic Disease Prevention and Health Promotion, Atlanta, and her associates reported in the Morbidity and Mortality Weekly Report. Current use was defined as use every day or some days, with an added requirement of at least 100 cigarettes in a lifetime added for cigarette smokers.
Data from the National Health Interview Survey showed that from 2016 to 2017, current use declined for any tobacco product, any combustible tobacco product, cigarettes, smokeless tobacco, and the combination of two or more tobacco products. The most common combination in 2017 was cigarettes and e-cigarettes, which was reported by 30.1% of the 9 million adults who used more than one product, Dr. Wang and her associates said.
Prevalence of current tobacco use was higher among men than women (24.8% vs. 14.2%), and adults aged 25-44 years (22.5%) had the highest level by age, followed by those aged 45-64 years (21.3%), 18-24 years (18.3%), and 65 years or older (11%). Use by race/ethnicity was highest among American Indian/Alaska Natives (29.8%), with the Midwest putting up the highest prevalence by region at 23.5%, they said.
“Although cigarette smoking among U.S. adults has declined considerably, tobacco products have evolved in recent years to include various combustible, noncombustible, and electronic products,” Dr. Wang and her associates wrote. “Implementation of evidence-based tobacco control interventions that address the diversity of tobacco products used by U.S. adults, in coordination with regulation of tobacco product manufacturing, marketing, and sales, can reduce tobacco-related disease and death in the United States.”
SOURCE: Wang TW et al. MMWR. 2018 Nov 9;67[44]:1225-32.
FROM MMWR
Many teens don’t know e-cigarettes contain nicotine
ORLANDO – Flavoring and lack of Food and Drug Administration regulation of e-cigarettes has led to more children and adolescents using these devices, according to American Academy of Pediatrics President Colleen A. Kraft, MD.
In an interview, Dr. Kraft said the FDA should regulate these products and limit their purchase to adults who are at least 21 years old. E-cigarettes were initially intended as an aid for adults to reduce their cigarette use, but the addition of flavoring has attracted children and adolescents to the devices, Dr. Kraft noted.
“When you have these devices that have flavors like gummy bear and cotton candy and bubblegum, you are marketing to children, and we are calling out the FDA because they could actually stop this today,” she said. In fact, Dr. Kraft added, many children and adolescents don’t even realize that e-cigarettes contain nicotine.
Dr. Kraft reported no relevant conflicts of interest.
ORLANDO – Flavoring and lack of Food and Drug Administration regulation of e-cigarettes has led to more children and adolescents using these devices, according to American Academy of Pediatrics President Colleen A. Kraft, MD.
In an interview, Dr. Kraft said the FDA should regulate these products and limit their purchase to adults who are at least 21 years old. E-cigarettes were initially intended as an aid for adults to reduce their cigarette use, but the addition of flavoring has attracted children and adolescents to the devices, Dr. Kraft noted.
“When you have these devices that have flavors like gummy bear and cotton candy and bubblegum, you are marketing to children, and we are calling out the FDA because they could actually stop this today,” she said. In fact, Dr. Kraft added, many children and adolescents don’t even realize that e-cigarettes contain nicotine.
Dr. Kraft reported no relevant conflicts of interest.
ORLANDO – Flavoring and lack of Food and Drug Administration regulation of e-cigarettes has led to more children and adolescents using these devices, according to American Academy of Pediatrics President Colleen A. Kraft, MD.
In an interview, Dr. Kraft said the FDA should regulate these products and limit their purchase to adults who are at least 21 years old. E-cigarettes were initially intended as an aid for adults to reduce their cigarette use, but the addition of flavoring has attracted children and adolescents to the devices, Dr. Kraft noted.
“When you have these devices that have flavors like gummy bear and cotton candy and bubblegum, you are marketing to children, and we are calling out the FDA because they could actually stop this today,” she said. In fact, Dr. Kraft added, many children and adolescents don’t even realize that e-cigarettes contain nicotine.
Dr. Kraft reported no relevant conflicts of interest.
REPORTING FROM AAP 2018
Pneumonia, COPD most common emergency care–sensitive conditions
SAN DIEGO – Emergency care–sensitive conditions – those for which timely access to high-quality emergency care impact morbidity and mortality—account for 14% of all ED visits, results from a large analysis of national data showed.
In previously published work, an eight-member expert panel identified 51 condition groups as emergency care–sensitive conditions (ECSCs), including asthma, cardiac arrest, cerebral infarction, and pneumonia. The purpose of the current study, published in Annals of Emergency Medicine and presented by Anita Vashi, MD, MPH, at the annual meeting of the American College of Emergency Physicians, was to provide the first national estimates of acute care utilization and the demographic characteristics of adults experiencing ECSCs, compare ECSC and non-ECSC ED visits, and assess patient- and hospital-level characteristics predictive of an ECSC-related ED visit.
Using the Nationwide Emergency Department Sample data set, Dr. Vashi, a physician investigator at the Center for Innovation to Implementation at the VA Palo Alto Health Care System, and her colleagues retrospectively evaluated all ED visits for patients aged 18 years and older from 2009 to 2014. The researchers used summary statistics to compare population characteristics across groups and multivariable logistic regression models to assess the odds of an ECSC-related ED visit with patient- and hospital-level characteristics.
Of the 622,725,542 estimated ED visits evaluated during the study period, 86,577,041 (14%) were ECSCs. Among these ECSC visits, 58% of patients were admitted for an average length of 3.2 days and an average charge of $2,240. The most frequent ECSC-related visits were for pneumonia (9%), chronic obstructive pulmonary disease (9%), asthma (7%), heart failure (7%), and sepsis (5%), but varied by age group.
Dr. Vashi and her colleagues found that ECSCs were more common among older adults, males, those who reside in low-income areas, those who reside in the South, and among metropolitan-based hospitals and nontrauma center hospitals. ECSCs also accounted for about 45% of all inpatient admissions.
Multivariate logistic regression analysis revealed that the odds of having an ECSC-related visit was highest among patients aged 65 years and older (odds ratio, 3.84), those on Medicare (OR, 1.37), those who resided in rural counties (OR, 1.21), and those who reside in the Western portion of the United States (OR, 1.11). Significant hospital-related factors related to ECSC visits included trauma centers (OR, 1.09), nonteaching hospitals (OR, 1.04), and EDs located in the wealthiest counties (OR, 1.02).
The researchers also found that 40% of patients who made ECSC-related ED visits were treated and discharged back to the community. “There is evidence of regional variability, suggesting the need for future research,” said Dr. Vashi, who also holds a faculty position in the department of emergency medicine at Stanford (Calif.) University. “We found no consistent relationship between insurance, income, and ED use for ECSC-related conditions. This suggests that ECSCs are not significantly influenced by socioeconomic factor and can serve as a reliable marker for acuity.”
The next steps in this research area, she added, are to create condition-specific measures related to morbidity, mortality, and posthospital events, as well as to analyze regional and hospital variations including correlation across conditions, and to compare performance across conditions and hospitals.
Dr. Vashi reported having no financial disclosures.
Source: Vashi A et al. Ann Emerg Med. 2018 Oct;72;4:S38. doi. 10.1016/j.annemergmed.2018.08.091.
SAN DIEGO – Emergency care–sensitive conditions – those for which timely access to high-quality emergency care impact morbidity and mortality—account for 14% of all ED visits, results from a large analysis of national data showed.
In previously published work, an eight-member expert panel identified 51 condition groups as emergency care–sensitive conditions (ECSCs), including asthma, cardiac arrest, cerebral infarction, and pneumonia. The purpose of the current study, published in Annals of Emergency Medicine and presented by Anita Vashi, MD, MPH, at the annual meeting of the American College of Emergency Physicians, was to provide the first national estimates of acute care utilization and the demographic characteristics of adults experiencing ECSCs, compare ECSC and non-ECSC ED visits, and assess patient- and hospital-level characteristics predictive of an ECSC-related ED visit.
Using the Nationwide Emergency Department Sample data set, Dr. Vashi, a physician investigator at the Center for Innovation to Implementation at the VA Palo Alto Health Care System, and her colleagues retrospectively evaluated all ED visits for patients aged 18 years and older from 2009 to 2014. The researchers used summary statistics to compare population characteristics across groups and multivariable logistic regression models to assess the odds of an ECSC-related ED visit with patient- and hospital-level characteristics.
Of the 622,725,542 estimated ED visits evaluated during the study period, 86,577,041 (14%) were ECSCs. Among these ECSC visits, 58% of patients were admitted for an average length of 3.2 days and an average charge of $2,240. The most frequent ECSC-related visits were for pneumonia (9%), chronic obstructive pulmonary disease (9%), asthma (7%), heart failure (7%), and sepsis (5%), but varied by age group.
Dr. Vashi and her colleagues found that ECSCs were more common among older adults, males, those who reside in low-income areas, those who reside in the South, and among metropolitan-based hospitals and nontrauma center hospitals. ECSCs also accounted for about 45% of all inpatient admissions.
Multivariate logistic regression analysis revealed that the odds of having an ECSC-related visit was highest among patients aged 65 years and older (odds ratio, 3.84), those on Medicare (OR, 1.37), those who resided in rural counties (OR, 1.21), and those who reside in the Western portion of the United States (OR, 1.11). Significant hospital-related factors related to ECSC visits included trauma centers (OR, 1.09), nonteaching hospitals (OR, 1.04), and EDs located in the wealthiest counties (OR, 1.02).
The researchers also found that 40% of patients who made ECSC-related ED visits were treated and discharged back to the community. “There is evidence of regional variability, suggesting the need for future research,” said Dr. Vashi, who also holds a faculty position in the department of emergency medicine at Stanford (Calif.) University. “We found no consistent relationship between insurance, income, and ED use for ECSC-related conditions. This suggests that ECSCs are not significantly influenced by socioeconomic factor and can serve as a reliable marker for acuity.”
The next steps in this research area, she added, are to create condition-specific measures related to morbidity, mortality, and posthospital events, as well as to analyze regional and hospital variations including correlation across conditions, and to compare performance across conditions and hospitals.
Dr. Vashi reported having no financial disclosures.
Source: Vashi A et al. Ann Emerg Med. 2018 Oct;72;4:S38. doi. 10.1016/j.annemergmed.2018.08.091.
SAN DIEGO – Emergency care–sensitive conditions – those for which timely access to high-quality emergency care impact morbidity and mortality—account for 14% of all ED visits, results from a large analysis of national data showed.
In previously published work, an eight-member expert panel identified 51 condition groups as emergency care–sensitive conditions (ECSCs), including asthma, cardiac arrest, cerebral infarction, and pneumonia. The purpose of the current study, published in Annals of Emergency Medicine and presented by Anita Vashi, MD, MPH, at the annual meeting of the American College of Emergency Physicians, was to provide the first national estimates of acute care utilization and the demographic characteristics of adults experiencing ECSCs, compare ECSC and non-ECSC ED visits, and assess patient- and hospital-level characteristics predictive of an ECSC-related ED visit.
Using the Nationwide Emergency Department Sample data set, Dr. Vashi, a physician investigator at the Center for Innovation to Implementation at the VA Palo Alto Health Care System, and her colleagues retrospectively evaluated all ED visits for patients aged 18 years and older from 2009 to 2014. The researchers used summary statistics to compare population characteristics across groups and multivariable logistic regression models to assess the odds of an ECSC-related ED visit with patient- and hospital-level characteristics.
Of the 622,725,542 estimated ED visits evaluated during the study period, 86,577,041 (14%) were ECSCs. Among these ECSC visits, 58% of patients were admitted for an average length of 3.2 days and an average charge of $2,240. The most frequent ECSC-related visits were for pneumonia (9%), chronic obstructive pulmonary disease (9%), asthma (7%), heart failure (7%), and sepsis (5%), but varied by age group.
Dr. Vashi and her colleagues found that ECSCs were more common among older adults, males, those who reside in low-income areas, those who reside in the South, and among metropolitan-based hospitals and nontrauma center hospitals. ECSCs also accounted for about 45% of all inpatient admissions.
Multivariate logistic regression analysis revealed that the odds of having an ECSC-related visit was highest among patients aged 65 years and older (odds ratio, 3.84), those on Medicare (OR, 1.37), those who resided in rural counties (OR, 1.21), and those who reside in the Western portion of the United States (OR, 1.11). Significant hospital-related factors related to ECSC visits included trauma centers (OR, 1.09), nonteaching hospitals (OR, 1.04), and EDs located in the wealthiest counties (OR, 1.02).
The researchers also found that 40% of patients who made ECSC-related ED visits were treated and discharged back to the community. “There is evidence of regional variability, suggesting the need for future research,” said Dr. Vashi, who also holds a faculty position in the department of emergency medicine at Stanford (Calif.) University. “We found no consistent relationship between insurance, income, and ED use for ECSC-related conditions. This suggests that ECSCs are not significantly influenced by socioeconomic factor and can serve as a reliable marker for acuity.”
The next steps in this research area, she added, are to create condition-specific measures related to morbidity, mortality, and posthospital events, as well as to analyze regional and hospital variations including correlation across conditions, and to compare performance across conditions and hospitals.
Dr. Vashi reported having no financial disclosures.
Source: Vashi A et al. Ann Emerg Med. 2018 Oct;72;4:S38. doi. 10.1016/j.annemergmed.2018.08.091.
REPORTING FROM ACEP18
Key clinical point: Emergency care–sensitive conditions (ECSCs) make up a significant proportion of ED visits.
Major finding: The most common ECSC-related visits were for pneumonia (9%), chronic obstructive pulmonary disease (9%), and asthma (7%).
Study details: A retrospective cohort study of more than 86.5 million ECSC-related ED visits.
Disclosures: Dr. Vashi reported having no financial disclosures.
Source: Vashi A et al. Ann Emerg Med. 2018 Oct;72;4:S38. doi. 10.1016/j.annemergmed.2018.08.091.
Opioids negatively affect breathing during sleep
SAN ANTONIO – Opioids do not mix well with sleep, interfering with breathing and increasing the risk of central sleep apnea, explained Anita Rajagopal, MD, a pulmonologist in private practice in Indianapolis.
“The chronic respiratory suppressant effects of opioids are well described,” Dr. Rajagopal told attendees at the annual meeting of the American College of Chest Physicians. “The most characteristic signs of chronic opioid effects are irregular central apneas, ataxic breathing, Biot’s respiration and hypoxemia, mainly during NREM sleep.”
Dr. Rajagopal reviewed the research on the effects of opioid use, primarily for therapeutic use, during sleep, especially highlighting the adverse respiratory effects.
In one small study of 24 patients, ages 18-75, who were taking long-term opioids for chronic pain, 46% had severe sleep-disordered breathing, defined as an apnea-hypopnea index greater than 30/hour (J Clin Sleep Med. 2014 Aug 15;10[8]:847-52).
When compared to sleep clinic patients referred for sleep disordered breathing, the participants taking opioids had a higher frequency of central apneas and a lower arousal index. Further, the researchers found that “morphine equivalent doses correlated with the severity of sleep-disordered breathing.”
In another study, a systematic review from 2015, researchers sought to characterize the clinical features of sleep-disordered breathing associated with chronic opioid therapy (Anesth Analg. 2015 Jun;120[6]:1273-85). They identified eight studies with 560 patients, about a quarter of whom (24%) had central sleep apnea.
Once again, “The morphine equivalent daily dose was strongly associated with the severity of the sleep disordered breathing, predominantly central sleep apnea, with a morphine equivalent daily dose of more than 200 mg being a threshold of particular concern,” the researchers reported.
Patients receiving methadone therapy for heroin addiction are not spared the respiratory risks of opioids during sleep. Dr. Rajagopal shared research revealing that patients receiving methadone treatment for at least two months had a blunted hypercapnic respiratory response and increased hypoxemic ventilatory response, changes related to respiratory rate but not tidal volume.
“All mu-opioid receptor agonists can cause complex and potentially lethal effects on respiration during sleep,” Dr. Rajagopal said as she shared evidence from a 2007 study that compared breathing patterns during sleep between 60 patients taking chronic opioids and 60 matched patients not taking opioids (J Clin Sleep Med. 2007 Aug 15;3[5]:455-61).
That study found chronic opioid use to be associated with increased central apneas and reduced arterial oxygen saturation during wakefulness and NREM sleep. Again, a dose-response relationship emerged between morphine dose equivalent and the apnea-hypopnea, obstructive apnea, hypopnea and central apnea indices (P less than .001).
Patients who took opioids long-term were also more likely to have ataxic or irregular breathing during NREM sleep, compared with patients not taking opioids.
In yet another meta-analysis and systematic review she related, researchers found across 803 patients in seven studies that long-term opioids users had a modestly increased risk for central sleep apnea but no similar increased risk for obstructive sleep apnea (J Clin Sleep Med. 2016 Apr 15;12[4]:617-25).
“REM and slow-wave sleep are decreased across all categories of opioid use — intravenous morphine, oral morphine, or methadone and heroin,” she said.
Since some patients are still going to need opioids, such as methadone therapy for those recovering from opioid use disorder, it’s important to understand appropriate effective treatments for central sleep apnea.
“CPAP [continuous positive airway pressure] is generally ineffective for opioid-induced sleep apnea and may augment central events,” Dr. Rajagopal explained, but adaptive servo ventilation (ASV) is effective for opioid-induced central apneas.
In one study of 20 patients receiving opioid therapy and referred for obstructive apnea, for example, the participants were diagnosed instead with central sleep apnea (J Clin Sleep Med. 2014 Jun 15;10[6]:637-43). The 16 patients who received CPAP continued to show central sleep apnea, with an AHI of 34 events/hour and central-apnea index (CAI) of 20 events/hour. Even after a four-week break before restarting CPAP, patients’ apnea did not resolve.
After receiving ASV, however, the average AHI dropped to 11 events/hour and CAI dropped to 0 events/hour. Those changes were accompanied by improvements in oxygen saturation, with the oxyhemoglobin saturation nadir increasing from 83% to 90%.
Similarly, a prospective multi-center observational trial assessed 27 patients with central apnea after they used ASV at home for three months (Chest. 2015 Dec;148[6]:1454-1461). The participants began with an average AHI of 55 and CAI of 23 at baseline. CPAP dropped these values only to an AHI of 33 and CAI of 10, but treatment with ASV dropped them to an AHI of 4 and CAI of 0 (P less than .001).
SAN ANTONIO – Opioids do not mix well with sleep, interfering with breathing and increasing the risk of central sleep apnea, explained Anita Rajagopal, MD, a pulmonologist in private practice in Indianapolis.
“The chronic respiratory suppressant effects of opioids are well described,” Dr. Rajagopal told attendees at the annual meeting of the American College of Chest Physicians. “The most characteristic signs of chronic opioid effects are irregular central apneas, ataxic breathing, Biot’s respiration and hypoxemia, mainly during NREM sleep.”
Dr. Rajagopal reviewed the research on the effects of opioid use, primarily for therapeutic use, during sleep, especially highlighting the adverse respiratory effects.
In one small study of 24 patients, ages 18-75, who were taking long-term opioids for chronic pain, 46% had severe sleep-disordered breathing, defined as an apnea-hypopnea index greater than 30/hour (J Clin Sleep Med. 2014 Aug 15;10[8]:847-52).
When compared to sleep clinic patients referred for sleep disordered breathing, the participants taking opioids had a higher frequency of central apneas and a lower arousal index. Further, the researchers found that “morphine equivalent doses correlated with the severity of sleep-disordered breathing.”
In another study, a systematic review from 2015, researchers sought to characterize the clinical features of sleep-disordered breathing associated with chronic opioid therapy (Anesth Analg. 2015 Jun;120[6]:1273-85). They identified eight studies with 560 patients, about a quarter of whom (24%) had central sleep apnea.
Once again, “The morphine equivalent daily dose was strongly associated with the severity of the sleep disordered breathing, predominantly central sleep apnea, with a morphine equivalent daily dose of more than 200 mg being a threshold of particular concern,” the researchers reported.
Patients receiving methadone therapy for heroin addiction are not spared the respiratory risks of opioids during sleep. Dr. Rajagopal shared research revealing that patients receiving methadone treatment for at least two months had a blunted hypercapnic respiratory response and increased hypoxemic ventilatory response, changes related to respiratory rate but not tidal volume.
“All mu-opioid receptor agonists can cause complex and potentially lethal effects on respiration during sleep,” Dr. Rajagopal said as she shared evidence from a 2007 study that compared breathing patterns during sleep between 60 patients taking chronic opioids and 60 matched patients not taking opioids (J Clin Sleep Med. 2007 Aug 15;3[5]:455-61).
That study found chronic opioid use to be associated with increased central apneas and reduced arterial oxygen saturation during wakefulness and NREM sleep. Again, a dose-response relationship emerged between morphine dose equivalent and the apnea-hypopnea, obstructive apnea, hypopnea and central apnea indices (P less than .001).
Patients who took opioids long-term were also more likely to have ataxic or irregular breathing during NREM sleep, compared with patients not taking opioids.
In yet another meta-analysis and systematic review she related, researchers found across 803 patients in seven studies that long-term opioids users had a modestly increased risk for central sleep apnea but no similar increased risk for obstructive sleep apnea (J Clin Sleep Med. 2016 Apr 15;12[4]:617-25).
“REM and slow-wave sleep are decreased across all categories of opioid use — intravenous morphine, oral morphine, or methadone and heroin,” she said.
Since some patients are still going to need opioids, such as methadone therapy for those recovering from opioid use disorder, it’s important to understand appropriate effective treatments for central sleep apnea.
“CPAP [continuous positive airway pressure] is generally ineffective for opioid-induced sleep apnea and may augment central events,” Dr. Rajagopal explained, but adaptive servo ventilation (ASV) is effective for opioid-induced central apneas.
In one study of 20 patients receiving opioid therapy and referred for obstructive apnea, for example, the participants were diagnosed instead with central sleep apnea (J Clin Sleep Med. 2014 Jun 15;10[6]:637-43). The 16 patients who received CPAP continued to show central sleep apnea, with an AHI of 34 events/hour and central-apnea index (CAI) of 20 events/hour. Even after a four-week break before restarting CPAP, patients’ apnea did not resolve.
After receiving ASV, however, the average AHI dropped to 11 events/hour and CAI dropped to 0 events/hour. Those changes were accompanied by improvements in oxygen saturation, with the oxyhemoglobin saturation nadir increasing from 83% to 90%.
Similarly, a prospective multi-center observational trial assessed 27 patients with central apnea after they used ASV at home for three months (Chest. 2015 Dec;148[6]:1454-1461). The participants began with an average AHI of 55 and CAI of 23 at baseline. CPAP dropped these values only to an AHI of 33 and CAI of 10, but treatment with ASV dropped them to an AHI of 4 and CAI of 0 (P less than .001).
SAN ANTONIO – Opioids do not mix well with sleep, interfering with breathing and increasing the risk of central sleep apnea, explained Anita Rajagopal, MD, a pulmonologist in private practice in Indianapolis.
“The chronic respiratory suppressant effects of opioids are well described,” Dr. Rajagopal told attendees at the annual meeting of the American College of Chest Physicians. “The most characteristic signs of chronic opioid effects are irregular central apneas, ataxic breathing, Biot’s respiration and hypoxemia, mainly during NREM sleep.”
Dr. Rajagopal reviewed the research on the effects of opioid use, primarily for therapeutic use, during sleep, especially highlighting the adverse respiratory effects.
In one small study of 24 patients, ages 18-75, who were taking long-term opioids for chronic pain, 46% had severe sleep-disordered breathing, defined as an apnea-hypopnea index greater than 30/hour (J Clin Sleep Med. 2014 Aug 15;10[8]:847-52).
When compared to sleep clinic patients referred for sleep disordered breathing, the participants taking opioids had a higher frequency of central apneas and a lower arousal index. Further, the researchers found that “morphine equivalent doses correlated with the severity of sleep-disordered breathing.”
In another study, a systematic review from 2015, researchers sought to characterize the clinical features of sleep-disordered breathing associated with chronic opioid therapy (Anesth Analg. 2015 Jun;120[6]:1273-85). They identified eight studies with 560 patients, about a quarter of whom (24%) had central sleep apnea.
Once again, “The morphine equivalent daily dose was strongly associated with the severity of the sleep disordered breathing, predominantly central sleep apnea, with a morphine equivalent daily dose of more than 200 mg being a threshold of particular concern,” the researchers reported.
Patients receiving methadone therapy for heroin addiction are not spared the respiratory risks of opioids during sleep. Dr. Rajagopal shared research revealing that patients receiving methadone treatment for at least two months had a blunted hypercapnic respiratory response and increased hypoxemic ventilatory response, changes related to respiratory rate but not tidal volume.
“All mu-opioid receptor agonists can cause complex and potentially lethal effects on respiration during sleep,” Dr. Rajagopal said as she shared evidence from a 2007 study that compared breathing patterns during sleep between 60 patients taking chronic opioids and 60 matched patients not taking opioids (J Clin Sleep Med. 2007 Aug 15;3[5]:455-61).
That study found chronic opioid use to be associated with increased central apneas and reduced arterial oxygen saturation during wakefulness and NREM sleep. Again, a dose-response relationship emerged between morphine dose equivalent and the apnea-hypopnea, obstructive apnea, hypopnea and central apnea indices (P less than .001).
Patients who took opioids long-term were also more likely to have ataxic or irregular breathing during NREM sleep, compared with patients not taking opioids.
In yet another meta-analysis and systematic review she related, researchers found across 803 patients in seven studies that long-term opioids users had a modestly increased risk for central sleep apnea but no similar increased risk for obstructive sleep apnea (J Clin Sleep Med. 2016 Apr 15;12[4]:617-25).
“REM and slow-wave sleep are decreased across all categories of opioid use — intravenous morphine, oral morphine, or methadone and heroin,” she said.
Since some patients are still going to need opioids, such as methadone therapy for those recovering from opioid use disorder, it’s important to understand appropriate effective treatments for central sleep apnea.
“CPAP [continuous positive airway pressure] is generally ineffective for opioid-induced sleep apnea and may augment central events,” Dr. Rajagopal explained, but adaptive servo ventilation (ASV) is effective for opioid-induced central apneas.
In one study of 20 patients receiving opioid therapy and referred for obstructive apnea, for example, the participants were diagnosed instead with central sleep apnea (J Clin Sleep Med. 2014 Jun 15;10[6]:637-43). The 16 patients who received CPAP continued to show central sleep apnea, with an AHI of 34 events/hour and central-apnea index (CAI) of 20 events/hour. Even after a four-week break before restarting CPAP, patients’ apnea did not resolve.
After receiving ASV, however, the average AHI dropped to 11 events/hour and CAI dropped to 0 events/hour. Those changes were accompanied by improvements in oxygen saturation, with the oxyhemoglobin saturation nadir increasing from 83% to 90%.
Similarly, a prospective multi-center observational trial assessed 27 patients with central apnea after they used ASV at home for three months (Chest. 2015 Dec;148[6]:1454-1461). The participants began with an average AHI of 55 and CAI of 23 at baseline. CPAP dropped these values only to an AHI of 33 and CAI of 10, but treatment with ASV dropped them to an AHI of 4 and CAI of 0 (P less than .001).
REPORTING FROM CHEST 2018