User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
Powered by CHEST Physician, Clinician Reviews, MDedge Family Medicine, Internal Medicine News, and The Journal of Clinical Outcomes Management.
Flu outbreaks may be more intense in small cities
Influenza outbreaks in the United States tend to be concentrated and intense in small cities and more evenly spread throughout the season in large cities, results of a recent study show.
Swings in humidity further intensified the influenza spikes in small cities, but didn’t seem to have as much of an effect in large cities, the results suggest.
These findings help explain differences in influenza transmission patterns between cities that have similar climates and virus epidemiology, according to researcher Benjamin D. Dalziel, PhD, of the departments of integrative biology and mathematics at Oregon State University in Corvallis.
“City size and structure can play a role in determining how other factors such as climate affect and influence transmission,” Dr. Dalziel said in a press conference.
“Our results show how metropolises play a disproportionately important role in this process, as epidemic foci, and as potential sentinel hubs, where epidemiological observatories could integrate local strain dynamics to predict larger-scale patterns. As the growth and form of cities affect their function as climate-driven incubators of infectious disease, it may be possible to design smarter cities that better control epidemics in the face of accelerating global change,” the researchers wrote in their study.
Dr. Dalziel and his coauthors analyzed the weekly incidence of influenza-like illness across 603 U.S. ZIP codes using data obtained from medical claims from 2002 to 2008. They used epidemic intensity as a summary statistic to compare cities. By this variable, low epidemic intensity indicated a diffuse spread evenly across weeks of the flu season, whereas high epidemic intensity indicated intensively focused outbreaks on particular weeks.
In small cities, epidemics were more intensely focused on shorter periods at the peak of flu season, they found. In large cities, incidence was more diffuse, according to results published in Science.
Patterns of where people live and work in a city may account for the more diffuse and prolonged outbreaks seen in large cities, the authors wrote. Large cities have organized population movement patterns and crowding. In more highly established work locations, for example, the population density is pronounced during the day.
“We found the structure makes a difference for how the flu spreads at different times of year,” Dr. Dalziel said of the study, which used U.S. Census data to evaluate spatial population distributions. “In large cities with more highly organized patterns, conditions play a relatively smaller role in flu transmission.”
Humidity’s lower impact on outbreaks in large cities might also be explained by population effects: “If an infected person is sitting beside you, it matters less what the specific humidity is,” Dr. Dalziel said, adding that the proximity helps the virus find hosts even when climatic conditions are not at their most favorable.
The study findings may have implications for health care resources in small cities, which could be strained by intense outbreaks, said coinvestigator Cecile Viboud, PhD, of the Division of International Epidemiology and Population Studies, Fogarty International Center, National Institutes of Health, Bethesda, Md.
Intense outbreaks could overload the health care system, making it challenging to respond, especially around the peak of the epidemic. Pressure on the health care system may be less intense in cities such as Miami or New York, where flu epidemics are more diffuse and spread out during the year, she said.
Variations in vaccination coverage were not associated with variations in epidemic intensity at the state level. However, the data period that was analyzed ended in 2008, a time when flu vaccination rates were much lower than they are today, according to Dr. Viboud.
“It would be important to revisit the effect of city structure and humidity on flu transmission in a high vaccination regime in more recent years, especially if there is a lot of interest in developing broadly cross-protective flu vaccines, which might become available in the market in the future,” she said.
The researchers declared no competing interests related to their research, which was supported by a grant from the Bill & Melinda Gates Foundation, the RAPIDD program of the Science and Technology Directorate Department of Homeland Security, and the Fogarty International Center, National Institutes of Health.
SOURCE: Dalziel BD et al. Science. 2018 Oct 5;362(6410):75-9.
Public health policy makers may need to switch up their thinking about infection control during influenza outbreaks. Instead of targeting the population at large, it may make sense to focus on specific small towns or metropolitan areas for control.
Summary statistics, such as epidemic intensity, help to identify which places require more surge capacity to deal with peak health care demand. They also help to guide locations for active influenza surveillance where long transmission chains of influenza occur, and where new genetic variants of the influenza virus can be detected.
The findings of this study could foster the development of more accurate short-term, small-scale forecasts of the expected health care demand in a season. Most important, they could guide long-term projections that reveal how the shifting demography, growth of cities, and the changing climate alter infection dynamics and required control efforts.
Prof. Jacco Wallinga is with the Center for Infectious Disease Control, National Institute for Public Health and the Environment, Bilthoven, the Netherlands, and the Department of Biomedical Data Sciences, Leiden (the Netherlands) University Medical Center. These comments appeared in his editorial in Science (2018 Oct 5;362[6410]:29-30).
Public health policy makers may need to switch up their thinking about infection control during influenza outbreaks. Instead of targeting the population at large, it may make sense to focus on specific small towns or metropolitan areas for control.
Summary statistics, such as epidemic intensity, help to identify which places require more surge capacity to deal with peak health care demand. They also help to guide locations for active influenza surveillance where long transmission chains of influenza occur, and where new genetic variants of the influenza virus can be detected.
The findings of this study could foster the development of more accurate short-term, small-scale forecasts of the expected health care demand in a season. Most important, they could guide long-term projections that reveal how the shifting demography, growth of cities, and the changing climate alter infection dynamics and required control efforts.
Prof. Jacco Wallinga is with the Center for Infectious Disease Control, National Institute for Public Health and the Environment, Bilthoven, the Netherlands, and the Department of Biomedical Data Sciences, Leiden (the Netherlands) University Medical Center. These comments appeared in his editorial in Science (2018 Oct 5;362[6410]:29-30).
Public health policy makers may need to switch up their thinking about infection control during influenza outbreaks. Instead of targeting the population at large, it may make sense to focus on specific small towns or metropolitan areas for control.
Summary statistics, such as epidemic intensity, help to identify which places require more surge capacity to deal with peak health care demand. They also help to guide locations for active influenza surveillance where long transmission chains of influenza occur, and where new genetic variants of the influenza virus can be detected.
The findings of this study could foster the development of more accurate short-term, small-scale forecasts of the expected health care demand in a season. Most important, they could guide long-term projections that reveal how the shifting demography, growth of cities, and the changing climate alter infection dynamics and required control efforts.
Prof. Jacco Wallinga is with the Center for Infectious Disease Control, National Institute for Public Health and the Environment, Bilthoven, the Netherlands, and the Department of Biomedical Data Sciences, Leiden (the Netherlands) University Medical Center. These comments appeared in his editorial in Science (2018 Oct 5;362[6410]:29-30).
Influenza outbreaks in the United States tend to be concentrated and intense in small cities and more evenly spread throughout the season in large cities, results of a recent study show.
Swings in humidity further intensified the influenza spikes in small cities, but didn’t seem to have as much of an effect in large cities, the results suggest.
These findings help explain differences in influenza transmission patterns between cities that have similar climates and virus epidemiology, according to researcher Benjamin D. Dalziel, PhD, of the departments of integrative biology and mathematics at Oregon State University in Corvallis.
“City size and structure can play a role in determining how other factors such as climate affect and influence transmission,” Dr. Dalziel said in a press conference.
“Our results show how metropolises play a disproportionately important role in this process, as epidemic foci, and as potential sentinel hubs, where epidemiological observatories could integrate local strain dynamics to predict larger-scale patterns. As the growth and form of cities affect their function as climate-driven incubators of infectious disease, it may be possible to design smarter cities that better control epidemics in the face of accelerating global change,” the researchers wrote in their study.
Dr. Dalziel and his coauthors analyzed the weekly incidence of influenza-like illness across 603 U.S. ZIP codes using data obtained from medical claims from 2002 to 2008. They used epidemic intensity as a summary statistic to compare cities. By this variable, low epidemic intensity indicated a diffuse spread evenly across weeks of the flu season, whereas high epidemic intensity indicated intensively focused outbreaks on particular weeks.
In small cities, epidemics were more intensely focused on shorter periods at the peak of flu season, they found. In large cities, incidence was more diffuse, according to results published in Science.
Patterns of where people live and work in a city may account for the more diffuse and prolonged outbreaks seen in large cities, the authors wrote. Large cities have organized population movement patterns and crowding. In more highly established work locations, for example, the population density is pronounced during the day.
“We found the structure makes a difference for how the flu spreads at different times of year,” Dr. Dalziel said of the study, which used U.S. Census data to evaluate spatial population distributions. “In large cities with more highly organized patterns, conditions play a relatively smaller role in flu transmission.”
Humidity’s lower impact on outbreaks in large cities might also be explained by population effects: “If an infected person is sitting beside you, it matters less what the specific humidity is,” Dr. Dalziel said, adding that the proximity helps the virus find hosts even when climatic conditions are not at their most favorable.
The study findings may have implications for health care resources in small cities, which could be strained by intense outbreaks, said coinvestigator Cecile Viboud, PhD, of the Division of International Epidemiology and Population Studies, Fogarty International Center, National Institutes of Health, Bethesda, Md.
Intense outbreaks could overload the health care system, making it challenging to respond, especially around the peak of the epidemic. Pressure on the health care system may be less intense in cities such as Miami or New York, where flu epidemics are more diffuse and spread out during the year, she said.
Variations in vaccination coverage were not associated with variations in epidemic intensity at the state level. However, the data period that was analyzed ended in 2008, a time when flu vaccination rates were much lower than they are today, according to Dr. Viboud.
“It would be important to revisit the effect of city structure and humidity on flu transmission in a high vaccination regime in more recent years, especially if there is a lot of interest in developing broadly cross-protective flu vaccines, which might become available in the market in the future,” she said.
The researchers declared no competing interests related to their research, which was supported by a grant from the Bill & Melinda Gates Foundation, the RAPIDD program of the Science and Technology Directorate Department of Homeland Security, and the Fogarty International Center, National Institutes of Health.
SOURCE: Dalziel BD et al. Science. 2018 Oct 5;362(6410):75-9.
Influenza outbreaks in the United States tend to be concentrated and intense in small cities and more evenly spread throughout the season in large cities, results of a recent study show.
Swings in humidity further intensified the influenza spikes in small cities, but didn’t seem to have as much of an effect in large cities, the results suggest.
These findings help explain differences in influenza transmission patterns between cities that have similar climates and virus epidemiology, according to researcher Benjamin D. Dalziel, PhD, of the departments of integrative biology and mathematics at Oregon State University in Corvallis.
“City size and structure can play a role in determining how other factors such as climate affect and influence transmission,” Dr. Dalziel said in a press conference.
“Our results show how metropolises play a disproportionately important role in this process, as epidemic foci, and as potential sentinel hubs, where epidemiological observatories could integrate local strain dynamics to predict larger-scale patterns. As the growth and form of cities affect their function as climate-driven incubators of infectious disease, it may be possible to design smarter cities that better control epidemics in the face of accelerating global change,” the researchers wrote in their study.
Dr. Dalziel and his coauthors analyzed the weekly incidence of influenza-like illness across 603 U.S. ZIP codes using data obtained from medical claims from 2002 to 2008. They used epidemic intensity as a summary statistic to compare cities. By this variable, low epidemic intensity indicated a diffuse spread evenly across weeks of the flu season, whereas high epidemic intensity indicated intensively focused outbreaks on particular weeks.
In small cities, epidemics were more intensely focused on shorter periods at the peak of flu season, they found. In large cities, incidence was more diffuse, according to results published in Science.
Patterns of where people live and work in a city may account for the more diffuse and prolonged outbreaks seen in large cities, the authors wrote. Large cities have organized population movement patterns and crowding. In more highly established work locations, for example, the population density is pronounced during the day.
“We found the structure makes a difference for how the flu spreads at different times of year,” Dr. Dalziel said of the study, which used U.S. Census data to evaluate spatial population distributions. “In large cities with more highly organized patterns, conditions play a relatively smaller role in flu transmission.”
Humidity’s lower impact on outbreaks in large cities might also be explained by population effects: “If an infected person is sitting beside you, it matters less what the specific humidity is,” Dr. Dalziel said, adding that the proximity helps the virus find hosts even when climatic conditions are not at their most favorable.
The study findings may have implications for health care resources in small cities, which could be strained by intense outbreaks, said coinvestigator Cecile Viboud, PhD, of the Division of International Epidemiology and Population Studies, Fogarty International Center, National Institutes of Health, Bethesda, Md.
Intense outbreaks could overload the health care system, making it challenging to respond, especially around the peak of the epidemic. Pressure on the health care system may be less intense in cities such as Miami or New York, where flu epidemics are more diffuse and spread out during the year, she said.
Variations in vaccination coverage were not associated with variations in epidemic intensity at the state level. However, the data period that was analyzed ended in 2008, a time when flu vaccination rates were much lower than they are today, according to Dr. Viboud.
“It would be important to revisit the effect of city structure and humidity on flu transmission in a high vaccination regime in more recent years, especially if there is a lot of interest in developing broadly cross-protective flu vaccines, which might become available in the market in the future,” she said.
The researchers declared no competing interests related to their research, which was supported by a grant from the Bill & Melinda Gates Foundation, the RAPIDD program of the Science and Technology Directorate Department of Homeland Security, and the Fogarty International Center, National Institutes of Health.
SOURCE: Dalziel BD et al. Science. 2018 Oct 5;362(6410):75-9.
FROM SCIENCE
Key clinical point: The intensity of influenza epidemics in U.S. cities varies according to population.
Major finding: Smaller cities had more intense outbreaks concentrated around the peak of flu season, while larger cities had cases spread throughout the season.
Study details: Analysis of weekly influenza-like illness incidence for 603 U.S. ZIP codes in medical claims data from 2002 to 2008.
Disclosures: The authors declared no competing interests. Funding came from the Bill & Melinda Gates Foundation, the Science and Technology Directorate Department of Homeland Security, and the Fogarty International Center, National Institutes of Health.
Source: Dalziel BD et al. Science. 2018 Oct 5;362(6410):75-9.
Ultrasound can’t rule out pulmonary embolism in the ED
SAN DIEGO – In the ICU, ultrasound has been shown to reduce the need for CT to evaluate potential pulmonary embolism. But in the ED, this strategy hasn’t worked out so far, according to Joseph Brown, MD, of the department of emergency medicine at the University of California, San Francisco.

Based on the data so far, the ED patients were less likely than the ICU patients to have another etiology identified on ultrasound that explained their symptoms. Further, ultrasound alone missed small subsegmental pulmonary emboli that were detected on subsequent CT scans in 2 of 11 patients.
The study is continuing, and Dr. Brown explains in this interview how ultrasound might be combined with other risk stratification measures to safely achieve reductions in CT scans.
SAN DIEGO – In the ICU, ultrasound has been shown to reduce the need for CT to evaluate potential pulmonary embolism. But in the ED, this strategy hasn’t worked out so far, according to Joseph Brown, MD, of the department of emergency medicine at the University of California, San Francisco.

Based on the data so far, the ED patients were less likely than the ICU patients to have another etiology identified on ultrasound that explained their symptoms. Further, ultrasound alone missed small subsegmental pulmonary emboli that were detected on subsequent CT scans in 2 of 11 patients.
The study is continuing, and Dr. Brown explains in this interview how ultrasound might be combined with other risk stratification measures to safely achieve reductions in CT scans.
SAN DIEGO – In the ICU, ultrasound has been shown to reduce the need for CT to evaluate potential pulmonary embolism. But in the ED, this strategy hasn’t worked out so far, according to Joseph Brown, MD, of the department of emergency medicine at the University of California, San Francisco.

Based on the data so far, the ED patients were less likely than the ICU patients to have another etiology identified on ultrasound that explained their symptoms. Further, ultrasound alone missed small subsegmental pulmonary emboli that were detected on subsequent CT scans in 2 of 11 patients.
The study is continuing, and Dr. Brown explains in this interview how ultrasound might be combined with other risk stratification measures to safely achieve reductions in CT scans.
REPORTING FROM ACEP18
Teens who vape are likely to add cigarette smoking
instead of substituting one for the other, according to research published in Nicotine & Tobacco Research.
“Our work provides more evidence that young people who use e-cigarettes progress to smoking cigarettes in the future,” Michael S. Dunbar, PhD, a behavioral scientist at the RAND Corp. stated in a press release. “This study also suggests that teens don’t substitute vaping products for cigarettes. Instead, they go on to use both products more frequently as they get older.”
Dr. Dunbar and his colleagues followed 2,039 adolescents aged 16-20 years who were originally enrolled in a Los Angeles–based substance use prevention program in sixth and seventh grade (2008) and completed annual Web-based surveys during 2015-2017 on their use of e-cigarettes (EC), cigarettes, alcohol, and marijuana. They also answered questions about their mental health with questions about anxiety and depression. The researchers used two models to measures associations between different factors.
The first model showed an association between EC and cigarette use. Then other factors, such as alcohol use, were introduced. Alcohol use was associated with increased cigarette use, and cigarette use was associated with increased use of alcohol. When introducing marijuana as a factor, associations remained between cigarette use and EC use, with higher EC use associated with greater marijuana use and vice versa. Greater cigarette use, however, was not predictive of later marijuana use. There was no association between EC use and mental health, but more cigarette use was associated with poorer mental health.
Under the second model, there was a moderate to strong association between EC use and cigarette use, and participants with greater EC use and greater cigarette use also reported higher alcohol use. There also was a significant between-person association with higher EC use, cigarette use, and marijuana use. There was a small negative association between mental health and cigarette use, but not with mental health and EC use,the researchers said.
“For young people, using these products may actually lead to more harm in the long run,” Dr. Dunbar said in the press release. “This highlights the importance of taking steps to prevent youth from vaping in the first place. One way to do this could be to limit e-cigarette and other tobacco advertising in kid-accessible spaces.”
This study was funded by grants from the National Institute of Alcohol Abuse and Alcoholism. The authors reported no relevant conflicts of interest.
SOURCE: Dunbar MS et al. Nicotine Tob Res. 2018 Oct 3. doi: 10.1093/ntr/nty179.
instead of substituting one for the other, according to research published in Nicotine & Tobacco Research.
“Our work provides more evidence that young people who use e-cigarettes progress to smoking cigarettes in the future,” Michael S. Dunbar, PhD, a behavioral scientist at the RAND Corp. stated in a press release. “This study also suggests that teens don’t substitute vaping products for cigarettes. Instead, they go on to use both products more frequently as they get older.”
Dr. Dunbar and his colleagues followed 2,039 adolescents aged 16-20 years who were originally enrolled in a Los Angeles–based substance use prevention program in sixth and seventh grade (2008) and completed annual Web-based surveys during 2015-2017 on their use of e-cigarettes (EC), cigarettes, alcohol, and marijuana. They also answered questions about their mental health with questions about anxiety and depression. The researchers used two models to measures associations between different factors.
The first model showed an association between EC and cigarette use. Then other factors, such as alcohol use, were introduced. Alcohol use was associated with increased cigarette use, and cigarette use was associated with increased use of alcohol. When introducing marijuana as a factor, associations remained between cigarette use and EC use, with higher EC use associated with greater marijuana use and vice versa. Greater cigarette use, however, was not predictive of later marijuana use. There was no association between EC use and mental health, but more cigarette use was associated with poorer mental health.
Under the second model, there was a moderate to strong association between EC use and cigarette use, and participants with greater EC use and greater cigarette use also reported higher alcohol use. There also was a significant between-person association with higher EC use, cigarette use, and marijuana use. There was a small negative association between mental health and cigarette use, but not with mental health and EC use,the researchers said.
“For young people, using these products may actually lead to more harm in the long run,” Dr. Dunbar said in the press release. “This highlights the importance of taking steps to prevent youth from vaping in the first place. One way to do this could be to limit e-cigarette and other tobacco advertising in kid-accessible spaces.”
This study was funded by grants from the National Institute of Alcohol Abuse and Alcoholism. The authors reported no relevant conflicts of interest.
SOURCE: Dunbar MS et al. Nicotine Tob Res. 2018 Oct 3. doi: 10.1093/ntr/nty179.
instead of substituting one for the other, according to research published in Nicotine & Tobacco Research.
“Our work provides more evidence that young people who use e-cigarettes progress to smoking cigarettes in the future,” Michael S. Dunbar, PhD, a behavioral scientist at the RAND Corp. stated in a press release. “This study also suggests that teens don’t substitute vaping products for cigarettes. Instead, they go on to use both products more frequently as they get older.”
Dr. Dunbar and his colleagues followed 2,039 adolescents aged 16-20 years who were originally enrolled in a Los Angeles–based substance use prevention program in sixth and seventh grade (2008) and completed annual Web-based surveys during 2015-2017 on their use of e-cigarettes (EC), cigarettes, alcohol, and marijuana. They also answered questions about their mental health with questions about anxiety and depression. The researchers used two models to measures associations between different factors.
The first model showed an association between EC and cigarette use. Then other factors, such as alcohol use, were introduced. Alcohol use was associated with increased cigarette use, and cigarette use was associated with increased use of alcohol. When introducing marijuana as a factor, associations remained between cigarette use and EC use, with higher EC use associated with greater marijuana use and vice versa. Greater cigarette use, however, was not predictive of later marijuana use. There was no association between EC use and mental health, but more cigarette use was associated with poorer mental health.
Under the second model, there was a moderate to strong association between EC use and cigarette use, and participants with greater EC use and greater cigarette use also reported higher alcohol use. There also was a significant between-person association with higher EC use, cigarette use, and marijuana use. There was a small negative association between mental health and cigarette use, but not with mental health and EC use,the researchers said.
“For young people, using these products may actually lead to more harm in the long run,” Dr. Dunbar said in the press release. “This highlights the importance of taking steps to prevent youth from vaping in the first place. One way to do this could be to limit e-cigarette and other tobacco advertising in kid-accessible spaces.”
This study was funded by grants from the National Institute of Alcohol Abuse and Alcoholism. The authors reported no relevant conflicts of interest.
SOURCE: Dunbar MS et al. Nicotine Tob Res. 2018 Oct 3. doi: 10.1093/ntr/nty179.
FROM NICOTINE & TOBACCO RESEARCH
Key clinical point: Adolescents who use e-cigarettes are more likely to progress to cigarette use, while continuing to vape.
Major finding: There was a significant bidirectional association between e-cigarette use and cigarette use, and some third factors such as alcohol use, marijuana use, and mental health.
Study details: A longitudinal study of adolescents aged 16-20 years enrolled in a substance use prevention program.
Disclosures: This study was funded by grants from the National Institute of Alcohol Abuse and Alcoholism. The authors reported no relevant conflicts of interest.
Source: Dunbar MS et al. Nicotine Tob Res. 2018 Oct 3. doi: 10.1093/ntr/nty179.
Two distinct subtypes of SLE-linked pulmonary arterial hypertension described
Patients with pulmonary arterial hypertension as a consequence of systemic lupus erythematosus can be classified into two different subtypes, one of which shows significantly greater mortality, new research suggests.
Systemic lupus erythematosus–associated pulmonary arterial hypertension (SLE-PAH) has a poor prognosis with 3-year mortality ranging from 45% to 88%, leading the first author of the study Fangfang Sun of the School of Medicine at Shanghai (China) Jiaotong University and coauthors to seek “to further differentiate among SLE-PAH patterns to better understand the disease and optimize its management,
In a letter published online in Annals of the Rheumatic Diseases, the researchers presented data from a retrospective study of a derivation cohort of 108 Chinese patients with SLE-PAH and a validation cohort of 87 patients. Using clinical and laboratory characteristics, the researchers classified the derivation cohort into two clusters, which they labeled as the vasculitic and vasculopathic subtypes.
Patients with the vasculitic subtype of SLE-PAH had higher levels of SLE disease activity and manifestations, such as pericarditis, rash, arthritis, nephritis, and neuropsychiatric lupus, while those with the vasculopathic subtype showed “purer” PAH and lower lupus disease activity.
The researchers found that the vasculitic subtype had around a threefold higher 3-year mortality than did the vasculopathic subtype (34.5%-40.2% vs. 13.0%-18.6%; hazard ratio, 2.84-3.15; P less than .05), even after adjusting for differences in treatments.
Patients who developed PAH less than 2 years after being diagnosed with SLE were significantly more likely to have the vasculitic subtype of SLE-PAH (P less than .0001). A SLE disease activity index score greater than nine was also significantly associated with the vasculitic subtype (P = .001).
The prediction model developed based on these two factors had a sensitivity of 98.5% and a specificity of 74.4% (area under the curve = 0.94; P less than .0001), for a weighted score of two or more, in identifying patients with the vasculitic subtype of SLE-PAH.
Dr. Sun and colleagues suggested that the existence of two distinct clinical subtypes of SLE-PAH points to different underlying pathophysiologic mechanisms; one that is autoimmune mediated and one that involves noninflammatory vascular remodeling. However, the authors stressed that their findings needed confirmation in prospective studies.
“The next key question that remains unanswered is how to balance the utility of immunosuppressants and PAH-targeted drugs in patients with different phenotypes,” they wrote.
Two authors declared research funding from academic and government agencies. No conflicts of interest were declared.
SOURCE: Sun F et al. Ann Rheum Dis. 2018 Sep 19. doi: 10.1136/annrheumdis-2018-214197.
Patients with pulmonary arterial hypertension as a consequence of systemic lupus erythematosus can be classified into two different subtypes, one of which shows significantly greater mortality, new research suggests.
Systemic lupus erythematosus–associated pulmonary arterial hypertension (SLE-PAH) has a poor prognosis with 3-year mortality ranging from 45% to 88%, leading the first author of the study Fangfang Sun of the School of Medicine at Shanghai (China) Jiaotong University and coauthors to seek “to further differentiate among SLE-PAH patterns to better understand the disease and optimize its management,
In a letter published online in Annals of the Rheumatic Diseases, the researchers presented data from a retrospective study of a derivation cohort of 108 Chinese patients with SLE-PAH and a validation cohort of 87 patients. Using clinical and laboratory characteristics, the researchers classified the derivation cohort into two clusters, which they labeled as the vasculitic and vasculopathic subtypes.
Patients with the vasculitic subtype of SLE-PAH had higher levels of SLE disease activity and manifestations, such as pericarditis, rash, arthritis, nephritis, and neuropsychiatric lupus, while those with the vasculopathic subtype showed “purer” PAH and lower lupus disease activity.
The researchers found that the vasculitic subtype had around a threefold higher 3-year mortality than did the vasculopathic subtype (34.5%-40.2% vs. 13.0%-18.6%; hazard ratio, 2.84-3.15; P less than .05), even after adjusting for differences in treatments.
Patients who developed PAH less than 2 years after being diagnosed with SLE were significantly more likely to have the vasculitic subtype of SLE-PAH (P less than .0001). A SLE disease activity index score greater than nine was also significantly associated with the vasculitic subtype (P = .001).
The prediction model developed based on these two factors had a sensitivity of 98.5% and a specificity of 74.4% (area under the curve = 0.94; P less than .0001), for a weighted score of two or more, in identifying patients with the vasculitic subtype of SLE-PAH.
Dr. Sun and colleagues suggested that the existence of two distinct clinical subtypes of SLE-PAH points to different underlying pathophysiologic mechanisms; one that is autoimmune mediated and one that involves noninflammatory vascular remodeling. However, the authors stressed that their findings needed confirmation in prospective studies.
“The next key question that remains unanswered is how to balance the utility of immunosuppressants and PAH-targeted drugs in patients with different phenotypes,” they wrote.
Two authors declared research funding from academic and government agencies. No conflicts of interest were declared.
SOURCE: Sun F et al. Ann Rheum Dis. 2018 Sep 19. doi: 10.1136/annrheumdis-2018-214197.
Patients with pulmonary arterial hypertension as a consequence of systemic lupus erythematosus can be classified into two different subtypes, one of which shows significantly greater mortality, new research suggests.
Systemic lupus erythematosus–associated pulmonary arterial hypertension (SLE-PAH) has a poor prognosis with 3-year mortality ranging from 45% to 88%, leading the first author of the study Fangfang Sun of the School of Medicine at Shanghai (China) Jiaotong University and coauthors to seek “to further differentiate among SLE-PAH patterns to better understand the disease and optimize its management,
In a letter published online in Annals of the Rheumatic Diseases, the researchers presented data from a retrospective study of a derivation cohort of 108 Chinese patients with SLE-PAH and a validation cohort of 87 patients. Using clinical and laboratory characteristics, the researchers classified the derivation cohort into two clusters, which they labeled as the vasculitic and vasculopathic subtypes.
Patients with the vasculitic subtype of SLE-PAH had higher levels of SLE disease activity and manifestations, such as pericarditis, rash, arthritis, nephritis, and neuropsychiatric lupus, while those with the vasculopathic subtype showed “purer” PAH and lower lupus disease activity.
The researchers found that the vasculitic subtype had around a threefold higher 3-year mortality than did the vasculopathic subtype (34.5%-40.2% vs. 13.0%-18.6%; hazard ratio, 2.84-3.15; P less than .05), even after adjusting for differences in treatments.
Patients who developed PAH less than 2 years after being diagnosed with SLE were significantly more likely to have the vasculitic subtype of SLE-PAH (P less than .0001). A SLE disease activity index score greater than nine was also significantly associated with the vasculitic subtype (P = .001).
The prediction model developed based on these two factors had a sensitivity of 98.5% and a specificity of 74.4% (area under the curve = 0.94; P less than .0001), for a weighted score of two or more, in identifying patients with the vasculitic subtype of SLE-PAH.
Dr. Sun and colleagues suggested that the existence of two distinct clinical subtypes of SLE-PAH points to different underlying pathophysiologic mechanisms; one that is autoimmune mediated and one that involves noninflammatory vascular remodeling. However, the authors stressed that their findings needed confirmation in prospective studies.
“The next key question that remains unanswered is how to balance the utility of immunosuppressants and PAH-targeted drugs in patients with different phenotypes,” they wrote.
Two authors declared research funding from academic and government agencies. No conflicts of interest were declared.
SOURCE: Sun F et al. Ann Rheum Dis. 2018 Sep 19. doi: 10.1136/annrheumdis-2018-214197.
FROM ANNALS OF THE RHEUMATIC DISEASES
Key clinical point: A more vasculitic subtype of SLE-PAH is associated with higher mortality.
Major finding: The more severe vasculitic subtype of SLE-PAH shows threefold higher 3-year mortality than a vasculopathic subtype.
Study details: Retrospective cohort study in 195 patients with SLE-PAH
Disclosures: Two authors declared research funding from academic and government agencies. No conflicts of interest were declared.
Source: Sun F et al. Ann Rheum Dis. 2018 Sep 19. doi: 10.1136/annrheumdis-2018-214197.
FDA approves Arikayce for MAC lung diseases
that has been caused by members of the Mycobacterium avium complex and is refractory to other treatments.
In a randomized, controlled trial, patients with refractory M. avium complex infections were assigned to receive either Arikayce plus a multidrug antibacterial regimen or just the antibacterial regimen. By 6 months, sputum cultures for 29% of those treated with the combination had shown no mycobacterial growth for 3 consecutive months, whereas this was only true for the cultures for 9% of patients on the multidrug antibacterial regimen alone.
The Arikayce prescribing information includes a boxed warning regarding the increased risk of respiratory conditions, including hypersensitivity pneumonitis, bronchospasm, exacerbation of underlying lung disease, and hemoptysis, some of which have proven serious enough to lead to hospitalization. Other side effects include dysphonia, cough, musculoskeletal pain, nausea, and fatigue.
According to the press announcement from the FDA, this is the first approval under the Limited Population Pathway for Antibacterial and Antifungal Drugs, which was set up by Congress “to advance development and approval of antibacterial and antifungal drugs to treat serious or life-threatening infections in a limited population of patients with unmet need.” It does so by allowing a more streamlined clinical development program that may involve smaller, shorter, or fewer clinical trials.
More information can be found in the full press announcement.
that has been caused by members of the Mycobacterium avium complex and is refractory to other treatments.
In a randomized, controlled trial, patients with refractory M. avium complex infections were assigned to receive either Arikayce plus a multidrug antibacterial regimen or just the antibacterial regimen. By 6 months, sputum cultures for 29% of those treated with the combination had shown no mycobacterial growth for 3 consecutive months, whereas this was only true for the cultures for 9% of patients on the multidrug antibacterial regimen alone.
The Arikayce prescribing information includes a boxed warning regarding the increased risk of respiratory conditions, including hypersensitivity pneumonitis, bronchospasm, exacerbation of underlying lung disease, and hemoptysis, some of which have proven serious enough to lead to hospitalization. Other side effects include dysphonia, cough, musculoskeletal pain, nausea, and fatigue.
According to the press announcement from the FDA, this is the first approval under the Limited Population Pathway for Antibacterial and Antifungal Drugs, which was set up by Congress “to advance development and approval of antibacterial and antifungal drugs to treat serious or life-threatening infections in a limited population of patients with unmet need.” It does so by allowing a more streamlined clinical development program that may involve smaller, shorter, or fewer clinical trials.
More information can be found in the full press announcement.
that has been caused by members of the Mycobacterium avium complex and is refractory to other treatments.
In a randomized, controlled trial, patients with refractory M. avium complex infections were assigned to receive either Arikayce plus a multidrug antibacterial regimen or just the antibacterial regimen. By 6 months, sputum cultures for 29% of those treated with the combination had shown no mycobacterial growth for 3 consecutive months, whereas this was only true for the cultures for 9% of patients on the multidrug antibacterial regimen alone.
The Arikayce prescribing information includes a boxed warning regarding the increased risk of respiratory conditions, including hypersensitivity pneumonitis, bronchospasm, exacerbation of underlying lung disease, and hemoptysis, some of which have proven serious enough to lead to hospitalization. Other side effects include dysphonia, cough, musculoskeletal pain, nausea, and fatigue.
According to the press announcement from the FDA, this is the first approval under the Limited Population Pathway for Antibacterial and Antifungal Drugs, which was set up by Congress “to advance development and approval of antibacterial and antifungal drugs to treat serious or life-threatening infections in a limited population of patients with unmet need.” It does so by allowing a more streamlined clinical development program that may involve smaller, shorter, or fewer clinical trials.
More information can be found in the full press announcement.
CDC: Trivalent adjuvanted influenza vaccine aIIV3 safe in elderly adults
ATLANTA – according to an analysis of reports to the Vaccine Adverse Event Reporting System (VAERS) during July 2016 through March 2018.
VAERS received 630 reports related to the vaccine (aIIV3; FLUAD) during the study period, of which 521 involved adults aged 65 years and older.
“Eighteen (3%) were serious reports, including two death reports (0.4%), all in adults aged [at least] 65 years,” Penina Haber and her colleagues at the Immunization Safety Office at the Centers for Disease Control and Prevention reported in a poster at the International Conference on Emerging Infectious Diseases.
The deaths included a 75-year-old man who died from Sjögren’s syndrome and a 65-year-old man who died from a myocardial infarction. The other serious events included five neurologic disorders (two cases of Guillain-Barré syndrome and one each of Bell’s palsy, Bickerstaff encephalitis, and lower-extremity weakness), five musculoskeletal and connective tissue disorders (three with shoulder pain and two with arm pain), three general disorders and administration site conditions (two cases of fever/chills and one case of cellulitis/bursitis), and one case each of a gastrointestinal disorder (acute diarrhea/gastroenteritis), an injury (a fall), and a skin/subcutaneous tissue disorder (keratosis pilaris rubra), according to the investigators.
There were no reports of anaphylaxis.
For the sake of comparison, the investigators also looked at reports associated with IIV3-HD and IIV3/IIV4 vaccines in adults aged 65 years and older during the same time period; they found that patient characteristics and reported events were similar for all the vaccines. For example, the percentages of reports involving patients aged 65 years and older were 65% or 66% for each, and those involving patients aged 75-84 years were 27%-29%. Further, 0.2%-0.6% of reports for each vaccine involved death.
The most frequently reported events for aIIV3, IIV3-HD, and IIV3/IIV4, respectively, were extremity pain (21%, 17%, and 15%, respectively), injection site erythema (18%, 19%, and 15%), and injection site pain (15%, 16%, and 16%), they said.
The aIIV3 vaccine – the first seasonal inactivated trivalent influenza vaccine produced from three influenza virus strains (two subtype A strains and one type B strain) – was approved by the Food and Drug Administration in 2015 for adults aged 65 years and older. It was the first influenza vaccine containing the adjuvant MF59 – a purified oil-in-water emulsion of squalene oil added to boost immune response in that population. Its safety was assessed in 15 randomized, controlled clinical studies, and several trials in older adults supported its efficacy and safety over nonadjuvanted influenza vaccines, the investigators reported. They noted that the Advisory Committee on Immunization Practices (ACIP) recommended the vaccine as an option for routine use in adults aged 65 years and older during the 2016-2017 flu seasons.
For the 2018-2019 flu season, ACIP determined that “For persons aged ≥65 years, any age-appropriate IIV formulation (standard-dose or high-dose, trivalent or quadrivalent, unadjuvanted or adjuvanted) or RIV4 are acceptable options.”
The findings of the analysis of the 2017-2018 flu season data are consistent with prelicensure studies, Ms. Haber and her colleagues concluded, noting that data mining did not detect disproportional reporting of any unexpected adverse event.
“[There were] no safety concerns following aIIV3 when compared to the nonadjuvanted influenza vaccines (IIV3-HD or IIV3/IIV4),” they wrote, adding that the “CDC and FDA will continue to monitor and ensure the safety of aIIV3.”
Ms. Haber reported having no disclosures
SOURCE: Haber P et al. ICEID 2018, Board 320.
ATLANTA – according to an analysis of reports to the Vaccine Adverse Event Reporting System (VAERS) during July 2016 through March 2018.
VAERS received 630 reports related to the vaccine (aIIV3; FLUAD) during the study period, of which 521 involved adults aged 65 years and older.
“Eighteen (3%) were serious reports, including two death reports (0.4%), all in adults aged [at least] 65 years,” Penina Haber and her colleagues at the Immunization Safety Office at the Centers for Disease Control and Prevention reported in a poster at the International Conference on Emerging Infectious Diseases.
The deaths included a 75-year-old man who died from Sjögren’s syndrome and a 65-year-old man who died from a myocardial infarction. The other serious events included five neurologic disorders (two cases of Guillain-Barré syndrome and one each of Bell’s palsy, Bickerstaff encephalitis, and lower-extremity weakness), five musculoskeletal and connective tissue disorders (three with shoulder pain and two with arm pain), three general disorders and administration site conditions (two cases of fever/chills and one case of cellulitis/bursitis), and one case each of a gastrointestinal disorder (acute diarrhea/gastroenteritis), an injury (a fall), and a skin/subcutaneous tissue disorder (keratosis pilaris rubra), according to the investigators.
There were no reports of anaphylaxis.
For the sake of comparison, the investigators also looked at reports associated with IIV3-HD and IIV3/IIV4 vaccines in adults aged 65 years and older during the same time period; they found that patient characteristics and reported events were similar for all the vaccines. For example, the percentages of reports involving patients aged 65 years and older were 65% or 66% for each, and those involving patients aged 75-84 years were 27%-29%. Further, 0.2%-0.6% of reports for each vaccine involved death.
The most frequently reported events for aIIV3, IIV3-HD, and IIV3/IIV4, respectively, were extremity pain (21%, 17%, and 15%, respectively), injection site erythema (18%, 19%, and 15%), and injection site pain (15%, 16%, and 16%), they said.
The aIIV3 vaccine – the first seasonal inactivated trivalent influenza vaccine produced from three influenza virus strains (two subtype A strains and one type B strain) – was approved by the Food and Drug Administration in 2015 for adults aged 65 years and older. It was the first influenza vaccine containing the adjuvant MF59 – a purified oil-in-water emulsion of squalene oil added to boost immune response in that population. Its safety was assessed in 15 randomized, controlled clinical studies, and several trials in older adults supported its efficacy and safety over nonadjuvanted influenza vaccines, the investigators reported. They noted that the Advisory Committee on Immunization Practices (ACIP) recommended the vaccine as an option for routine use in adults aged 65 years and older during the 2016-2017 flu seasons.
For the 2018-2019 flu season, ACIP determined that “For persons aged ≥65 years, any age-appropriate IIV formulation (standard-dose or high-dose, trivalent or quadrivalent, unadjuvanted or adjuvanted) or RIV4 are acceptable options.”
The findings of the analysis of the 2017-2018 flu season data are consistent with prelicensure studies, Ms. Haber and her colleagues concluded, noting that data mining did not detect disproportional reporting of any unexpected adverse event.
“[There were] no safety concerns following aIIV3 when compared to the nonadjuvanted influenza vaccines (IIV3-HD or IIV3/IIV4),” they wrote, adding that the “CDC and FDA will continue to monitor and ensure the safety of aIIV3.”
Ms. Haber reported having no disclosures
SOURCE: Haber P et al. ICEID 2018, Board 320.
ATLANTA – according to an analysis of reports to the Vaccine Adverse Event Reporting System (VAERS) during July 2016 through March 2018.
VAERS received 630 reports related to the vaccine (aIIV3; FLUAD) during the study period, of which 521 involved adults aged 65 years and older.
“Eighteen (3%) were serious reports, including two death reports (0.4%), all in adults aged [at least] 65 years,” Penina Haber and her colleagues at the Immunization Safety Office at the Centers for Disease Control and Prevention reported in a poster at the International Conference on Emerging Infectious Diseases.
The deaths included a 75-year-old man who died from Sjögren’s syndrome and a 65-year-old man who died from a myocardial infarction. The other serious events included five neurologic disorders (two cases of Guillain-Barré syndrome and one each of Bell’s palsy, Bickerstaff encephalitis, and lower-extremity weakness), five musculoskeletal and connective tissue disorders (three with shoulder pain and two with arm pain), three general disorders and administration site conditions (two cases of fever/chills and one case of cellulitis/bursitis), and one case each of a gastrointestinal disorder (acute diarrhea/gastroenteritis), an injury (a fall), and a skin/subcutaneous tissue disorder (keratosis pilaris rubra), according to the investigators.
There were no reports of anaphylaxis.
For the sake of comparison, the investigators also looked at reports associated with IIV3-HD and IIV3/IIV4 vaccines in adults aged 65 years and older during the same time period; they found that patient characteristics and reported events were similar for all the vaccines. For example, the percentages of reports involving patients aged 65 years and older were 65% or 66% for each, and those involving patients aged 75-84 years were 27%-29%. Further, 0.2%-0.6% of reports for each vaccine involved death.
The most frequently reported events for aIIV3, IIV3-HD, and IIV3/IIV4, respectively, were extremity pain (21%, 17%, and 15%, respectively), injection site erythema (18%, 19%, and 15%), and injection site pain (15%, 16%, and 16%), they said.
The aIIV3 vaccine – the first seasonal inactivated trivalent influenza vaccine produced from three influenza virus strains (two subtype A strains and one type B strain) – was approved by the Food and Drug Administration in 2015 for adults aged 65 years and older. It was the first influenza vaccine containing the adjuvant MF59 – a purified oil-in-water emulsion of squalene oil added to boost immune response in that population. Its safety was assessed in 15 randomized, controlled clinical studies, and several trials in older adults supported its efficacy and safety over nonadjuvanted influenza vaccines, the investigators reported. They noted that the Advisory Committee on Immunization Practices (ACIP) recommended the vaccine as an option for routine use in adults aged 65 years and older during the 2016-2017 flu seasons.
For the 2018-2019 flu season, ACIP determined that “For persons aged ≥65 years, any age-appropriate IIV formulation (standard-dose or high-dose, trivalent or quadrivalent, unadjuvanted or adjuvanted) or RIV4 are acceptable options.”
The findings of the analysis of the 2017-2018 flu season data are consistent with prelicensure studies, Ms. Haber and her colleagues concluded, noting that data mining did not detect disproportional reporting of any unexpected adverse event.
“[There were] no safety concerns following aIIV3 when compared to the nonadjuvanted influenza vaccines (IIV3-HD or IIV3/IIV4),” they wrote, adding that the “CDC and FDA will continue to monitor and ensure the safety of aIIV3.”
Ms. Haber reported having no disclosures
SOURCE: Haber P et al. ICEID 2018, Board 320.
REPORTING FROM ICEID 2018
Key clinical point: No new or unexpected adverse events were reported among the 630 reports related to the vaccine during the study period, of which 521 involved adults aged 65 years and older.
Major finding: Of 521 reports, 18 were serious, and there were two deaths.
Study details: A review of 521 reports to the Vaccine Adverse Event Reporting System in 2017-2018.
Disclosures: Ms. Haber reported having no disclosures.
Source: Haber P et al. ICEID 2018, Board 320.
PCV13 moderately effective in older adults
ATLANTA – (IPD) caused by PCV13 vaccine serotypes in adults aged 65 years and older, according to a case-control study involving Medicare beneficiaries.
Conversely, the 23-valent pneumococcal polysaccharide vaccine (PPSV23) showed limited effectiveness against serotypes unique to that vaccine in the study, which included 699 cases and more than 10,000 controls, Olivia Almendares, an epidemiologist at the Centers for Disease Control and Prevention, Atlanta, and her colleagues reported in a poster at the International Conference on Emerging Infectious Diseases.
“Vaccine efficacy against PCV13 [plus 6C type, which has cross-reactivity with serotype 6A] was 47% in those who received PCV13 vaccine only,” Ms. Almendares said in an interview, noting that efficacy was 26% against serotype 3 and 67% against other PCV13 serotypes (plus 6C). “Vaccine efficacy against PPSV23-unique types was 36% for those who received only PPSV23.”
Neither vaccine showed effectiveness against serotypes not included in the respective vaccines, she said.
The findings are timely given that the Advisory Committee on Immunization Practices (ACIP) is reevaluating its PCV13 recommendation for adults aged 65 years and older, she added.
“Specifically, ACIP is addressing whether PCV13 should be recommended routinely for all immunocompetent adults aged 65 and older given sustained indirect effects,” she said, explaining that, in 2014 when ACIP recommended routine use of the vaccine in series with PPSV23 for adults aged 65 years and older, the committee recognized that herd immunity effects from PCV13 use in children might eventually limit the utility of this recommendation, and therefore it proposed reevaluation and revision as needed after 4 years.
For the current study, she and her colleagues linked IPD cases in persons aged 65 years and older, which were identified through Active Bacterial Core surveillance during 2015-2016, to records for Centers for Medicare & Medicaid Services (CMS) beneficiaries. Vaccination and medical histories were obtained through medical records, and vaccine effectiveness was estimated as one minus the odds ratio for vaccination with PCV13 only or PPSV23 only versus neither vaccine using conditional logistic regression, with adjustment for sex and underlying medical conditions.
Of 2,246 IPD cases, 1,017 (45%) were matched to Medicare beneficiaries, and 699 were included in the analysis after those with noncontinuous enrollment in Medicare, long-term care residence, and missing census tract data were excluded. The cases were matched based on age, census tract of residence, and length of Medicare enrollment to 10,152 matched controls identified through CMS.
IPD associated with PCV13 (plus type 6C) accounted for 164 (23% of cases), of which 88 (12% of cases) involved serotype 3, and invasive pneumococcal disease associated with PPSV23 accounted for 350 cases (50%), she said.
PCV13 vaccine was given alone in 14% and 18% of cases and controls, respectively; PPSV23 alone was given in 22% and 21% of case patients and controls, respectively; and both vaccines were given in 8% of cases and controls.
Compared with controls, case patients were more likely to be of nonwhite race (16% vs. 11%), to have more than one chronic medical condition (88% vs. 58%), and to have one or more immunocompromising conditions (54% vs. 32%), she and her colleagues reported.
“PCV13 showed moderate overall effectiveness in preventing IPD caused by PCV13 (including 6C), but effectiveness may be lower for serotype 3 than for other PCV13 types,” she said.
“These results are in agreement with those from CAPiTA – a large clinical trial conducted in the Netherlands, which showed PCV13 to be effective against IPD caused by vaccine serotypes among community-dwelling adults aged 65 and older,” she noted. “Additionally, data from CDC surveillance suggest that PCV13-serotype [invasive pneumococcal disease] among children and adults aged 65 and older has declined dramatically following PCV13 introduction for children in 2010, as predicted.”
In fact, among adults aged 65 years and older, PCV13-serotype invasive pneumococcal disease declined by 40% after the vaccine was introduced in children. This corresponds to a change in the annual PCV13-serotype incidence from 14 cases per 100,000 population in 2010 to five cases per 100,000 population in 2014, she said; she added that IPD incidence plateaued in 2014-2016 with vaccine serotypes contributing to a small proportion of overall IPD burden among adults aged 65 years and older.
ACIP’s reevaluation of the PCV13 recommendation is ongoing and will be addressed at upcoming meetings.
“As part of the review process, we look at changes in disease incidence focusing primarily on invasive pneumococcal disease and noninvasive pneumonia, vaccine efficacy and effectiveness, and vaccine safety,” she said. She noted that ACIP currently has no plans to consider revising PCV13 recommendations for adults who have immunocompromising conditions, for whom PCV13 has been recommended since 2012.
Ms. Almendares reported having no disclosures.
SOURCE: Almendares O et al. ICEID 2018, Board 376.
ATLANTA – (IPD) caused by PCV13 vaccine serotypes in adults aged 65 years and older, according to a case-control study involving Medicare beneficiaries.
Conversely, the 23-valent pneumococcal polysaccharide vaccine (PPSV23) showed limited effectiveness against serotypes unique to that vaccine in the study, which included 699 cases and more than 10,000 controls, Olivia Almendares, an epidemiologist at the Centers for Disease Control and Prevention, Atlanta, and her colleagues reported in a poster at the International Conference on Emerging Infectious Diseases.
“Vaccine efficacy against PCV13 [plus 6C type, which has cross-reactivity with serotype 6A] was 47% in those who received PCV13 vaccine only,” Ms. Almendares said in an interview, noting that efficacy was 26% against serotype 3 and 67% against other PCV13 serotypes (plus 6C). “Vaccine efficacy against PPSV23-unique types was 36% for those who received only PPSV23.”
Neither vaccine showed effectiveness against serotypes not included in the respective vaccines, she said.
The findings are timely given that the Advisory Committee on Immunization Practices (ACIP) is reevaluating its PCV13 recommendation for adults aged 65 years and older, she added.
“Specifically, ACIP is addressing whether PCV13 should be recommended routinely for all immunocompetent adults aged 65 and older given sustained indirect effects,” she said, explaining that, in 2014 when ACIP recommended routine use of the vaccine in series with PPSV23 for adults aged 65 years and older, the committee recognized that herd immunity effects from PCV13 use in children might eventually limit the utility of this recommendation, and therefore it proposed reevaluation and revision as needed after 4 years.
For the current study, she and her colleagues linked IPD cases in persons aged 65 years and older, which were identified through Active Bacterial Core surveillance during 2015-2016, to records for Centers for Medicare & Medicaid Services (CMS) beneficiaries. Vaccination and medical histories were obtained through medical records, and vaccine effectiveness was estimated as one minus the odds ratio for vaccination with PCV13 only or PPSV23 only versus neither vaccine using conditional logistic regression, with adjustment for sex and underlying medical conditions.
Of 2,246 IPD cases, 1,017 (45%) were matched to Medicare beneficiaries, and 699 were included in the analysis after those with noncontinuous enrollment in Medicare, long-term care residence, and missing census tract data were excluded. The cases were matched based on age, census tract of residence, and length of Medicare enrollment to 10,152 matched controls identified through CMS.
IPD associated with PCV13 (plus type 6C) accounted for 164 (23% of cases), of which 88 (12% of cases) involved serotype 3, and invasive pneumococcal disease associated with PPSV23 accounted for 350 cases (50%), she said.
PCV13 vaccine was given alone in 14% and 18% of cases and controls, respectively; PPSV23 alone was given in 22% and 21% of case patients and controls, respectively; and both vaccines were given in 8% of cases and controls.
Compared with controls, case patients were more likely to be of nonwhite race (16% vs. 11%), to have more than one chronic medical condition (88% vs. 58%), and to have one or more immunocompromising conditions (54% vs. 32%), she and her colleagues reported.
“PCV13 showed moderate overall effectiveness in preventing IPD caused by PCV13 (including 6C), but effectiveness may be lower for serotype 3 than for other PCV13 types,” she said.
“These results are in agreement with those from CAPiTA – a large clinical trial conducted in the Netherlands, which showed PCV13 to be effective against IPD caused by vaccine serotypes among community-dwelling adults aged 65 and older,” she noted. “Additionally, data from CDC surveillance suggest that PCV13-serotype [invasive pneumococcal disease] among children and adults aged 65 and older has declined dramatically following PCV13 introduction for children in 2010, as predicted.”
In fact, among adults aged 65 years and older, PCV13-serotype invasive pneumococcal disease declined by 40% after the vaccine was introduced in children. This corresponds to a change in the annual PCV13-serotype incidence from 14 cases per 100,000 population in 2010 to five cases per 100,000 population in 2014, she said; she added that IPD incidence plateaued in 2014-2016 with vaccine serotypes contributing to a small proportion of overall IPD burden among adults aged 65 years and older.
ACIP’s reevaluation of the PCV13 recommendation is ongoing and will be addressed at upcoming meetings.
“As part of the review process, we look at changes in disease incidence focusing primarily on invasive pneumococcal disease and noninvasive pneumonia, vaccine efficacy and effectiveness, and vaccine safety,” she said. She noted that ACIP currently has no plans to consider revising PCV13 recommendations for adults who have immunocompromising conditions, for whom PCV13 has been recommended since 2012.
Ms. Almendares reported having no disclosures.
SOURCE: Almendares O et al. ICEID 2018, Board 376.
ATLANTA – (IPD) caused by PCV13 vaccine serotypes in adults aged 65 years and older, according to a case-control study involving Medicare beneficiaries.
Conversely, the 23-valent pneumococcal polysaccharide vaccine (PPSV23) showed limited effectiveness against serotypes unique to that vaccine in the study, which included 699 cases and more than 10,000 controls, Olivia Almendares, an epidemiologist at the Centers for Disease Control and Prevention, Atlanta, and her colleagues reported in a poster at the International Conference on Emerging Infectious Diseases.
“Vaccine efficacy against PCV13 [plus 6C type, which has cross-reactivity with serotype 6A] was 47% in those who received PCV13 vaccine only,” Ms. Almendares said in an interview, noting that efficacy was 26% against serotype 3 and 67% against other PCV13 serotypes (plus 6C). “Vaccine efficacy against PPSV23-unique types was 36% for those who received only PPSV23.”
Neither vaccine showed effectiveness against serotypes not included in the respective vaccines, she said.
The findings are timely given that the Advisory Committee on Immunization Practices (ACIP) is reevaluating its PCV13 recommendation for adults aged 65 years and older, she added.
“Specifically, ACIP is addressing whether PCV13 should be recommended routinely for all immunocompetent adults aged 65 and older given sustained indirect effects,” she said, explaining that, in 2014 when ACIP recommended routine use of the vaccine in series with PPSV23 for adults aged 65 years and older, the committee recognized that herd immunity effects from PCV13 use in children might eventually limit the utility of this recommendation, and therefore it proposed reevaluation and revision as needed after 4 years.
For the current study, she and her colleagues linked IPD cases in persons aged 65 years and older, which were identified through Active Bacterial Core surveillance during 2015-2016, to records for Centers for Medicare & Medicaid Services (CMS) beneficiaries. Vaccination and medical histories were obtained through medical records, and vaccine effectiveness was estimated as one minus the odds ratio for vaccination with PCV13 only or PPSV23 only versus neither vaccine using conditional logistic regression, with adjustment for sex and underlying medical conditions.
Of 2,246 IPD cases, 1,017 (45%) were matched to Medicare beneficiaries, and 699 were included in the analysis after those with noncontinuous enrollment in Medicare, long-term care residence, and missing census tract data were excluded. The cases were matched based on age, census tract of residence, and length of Medicare enrollment to 10,152 matched controls identified through CMS.
IPD associated with PCV13 (plus type 6C) accounted for 164 (23% of cases), of which 88 (12% of cases) involved serotype 3, and invasive pneumococcal disease associated with PPSV23 accounted for 350 cases (50%), she said.
PCV13 vaccine was given alone in 14% and 18% of cases and controls, respectively; PPSV23 alone was given in 22% and 21% of case patients and controls, respectively; and both vaccines were given in 8% of cases and controls.
Compared with controls, case patients were more likely to be of nonwhite race (16% vs. 11%), to have more than one chronic medical condition (88% vs. 58%), and to have one or more immunocompromising conditions (54% vs. 32%), she and her colleagues reported.
“PCV13 showed moderate overall effectiveness in preventing IPD caused by PCV13 (including 6C), but effectiveness may be lower for serotype 3 than for other PCV13 types,” she said.
“These results are in agreement with those from CAPiTA – a large clinical trial conducted in the Netherlands, which showed PCV13 to be effective against IPD caused by vaccine serotypes among community-dwelling adults aged 65 and older,” she noted. “Additionally, data from CDC surveillance suggest that PCV13-serotype [invasive pneumococcal disease] among children and adults aged 65 and older has declined dramatically following PCV13 introduction for children in 2010, as predicted.”
In fact, among adults aged 65 years and older, PCV13-serotype invasive pneumococcal disease declined by 40% after the vaccine was introduced in children. This corresponds to a change in the annual PCV13-serotype incidence from 14 cases per 100,000 population in 2010 to five cases per 100,000 population in 2014, she said; she added that IPD incidence plateaued in 2014-2016 with vaccine serotypes contributing to a small proportion of overall IPD burden among adults aged 65 years and older.
ACIP’s reevaluation of the PCV13 recommendation is ongoing and will be addressed at upcoming meetings.
“As part of the review process, we look at changes in disease incidence focusing primarily on invasive pneumococcal disease and noninvasive pneumonia, vaccine efficacy and effectiveness, and vaccine safety,” she said. She noted that ACIP currently has no plans to consider revising PCV13 recommendations for adults who have immunocompromising conditions, for whom PCV13 has been recommended since 2012.
Ms. Almendares reported having no disclosures.
SOURCE: Almendares O et al. ICEID 2018, Board 376.
REPORTING FROM ICEID 2018
Pulmonary artery denervation boosts walking capacity in left heart failure
SAN DIEGO – Pulmonary artery denervation is efficacious for treating combined pre- and postcapillary pulmonary hypertension attributable to left heart failure, based on results of the Chinese PADN-5 trial reported at the Transcatheter Cardiovascular Therapeutics annual meeting.
This ablative treatment has been studied among patients with pulmonary hypertension attributable to other etiologies, but not in randomized fashion among this population, noted lead investigator Shao-Liang Chen, MD, of Nanjing (China) First Hospital, Nanjing Medical University. The treatment is an attractive one, as medications recommended for pulmonary arterial hypertension are not recommended for joint pre- and postcapillary pulmonary hypertension (group II pulmonary hypertension).
In PADN-5, 98 patients were randomized to pulmonary artery denervation or to sham denervation plus open-label sildenafil (Viagra), which at the time of trial initiation was thought to be safe and potentially beneficial.
The trial’s main outcome, 6-minute walk distance at 6 months, improved in both groups, according to data reported at the meeting and simultaneously published in JACC Cardiovascular Interventions. But the improvement was about four times greater in the pulmonary artery denervation group. Secondary efficacy outcomes also favored that group, and the rate of fatal pulmonary embolism did not differ for the two groups.
“The PADN-5 trial demonstrates the benefits of pulmonary artery denervation for patients with combined pre- and postcapillary pulmonary hypertension. Patients with preserved and with reduced ejection fraction equally benefited,” summarized Dr. Chen, who pioneered this procedure about 7 years ago. “There was no sign of any harm of sildenafil in patients with combined pre- and postcapillary pulmonary hypertension.”
Trial critique
“This is a very difficult study to conduct, being able to recruit patients and actually have these procedures done,” commented press conference moderator Ori Ben-Yehuda, MD, professor of clinical medicine and director, coronary care unit, UC San Diego Medical Center.
At the same time, he expressed some reservations about the trial. “Sildenafil in the control group might actually be expected to ... decrease your effect size. Also, particularly in men, perhaps even in women, it might unblind them to which group they are in and undermine your sham design,” he noted. In addition, some hemodynamic changes after pulmonary artery denervation – a decrease in wedge pressure and an increase in ejection fraction – were puzzling.
“We need a lot more data here. There are some issues with this trial in terms of design, and we haven’t even gotten into the issue of whether there were core labs, whether the echoes, the hemodynamics, were read blindly,” Dr. Ben-Yehuda maintained. “This issue of secondary or group II pulmonary hypertension due to left heart failure is one that has been very frustrating in terms of actual PA-specific therapies. So this is an important step further, but it needs confirmation in truly sham-controlled trials that have no potential for unblinding.”
The catheter used in PADN-5 is available in China but has not received clearance in the United States, he pointed out. “There are alternative or competing technologies, one using ultrasound, for example, that has a very similar approach. … We’ll have to see how it ends up [performing].”
Trial details
Patients in the PADN-5 pulmonary artery denervation group underwent ablation only in the periconjunctional area between the distal main trunk and the left ostial branch with a multifunction catheter having premounted electrodes. Those in the control group underwent a sham procedure, with catheter positioning at the target sites and connection to a generator but no ablation, and were given open-label sildenafil. All additionally received standard heart failure medical therapy. (No sildenafil placebo was used in the denervation group.)
Trial results reported at the meeting, which is sponsored by the Cardiovascular Research Foundation, showed that most echocardiographic and hemodynamic measures improved more in the pulmonary artery denervation group.
The greater improvement in 6-minute walk test with denervation versus sham sildenafil at 6 months was evident in a variety of measures: absolute median distance walked (432.5 m vs. 358 m) and mean distance walked (434.6 m vs. 359.4 m), and absolute increase (80 m vs. 17.5 m) and relative increase (21.4% vs. 4.9%) The difference was significant for all measures at P less than .001.
The denervation group had a comparatively greater reduction of pulmonary vascular resistance (29.8% vs. 3.4%; P less than .001) and were less likely to experience clinical worsening (16.7% vs. 40.0%; P = .014).
There was a single fatal pulmonary embolism in each treatment group. Of the seven total deaths, two occurred in the denervation group (one attributable to pump failure, one a sudden death) and five occurred in the sham sildenafil group (all but one attributable to pump failure).
Dr. Chen disclosed that he had no relevant conflicts of interest. The trial was sponsored by Nanjing First Hospital, Nanjing Medical University.
SOURCE: Chen S-J et al. TCT 2018. JACC Cardiovasc Interv. 2018 Sep 23.
SAN DIEGO – Pulmonary artery denervation is efficacious for treating combined pre- and postcapillary pulmonary hypertension attributable to left heart failure, based on results of the Chinese PADN-5 trial reported at the Transcatheter Cardiovascular Therapeutics annual meeting.
This ablative treatment has been studied among patients with pulmonary hypertension attributable to other etiologies, but not in randomized fashion among this population, noted lead investigator Shao-Liang Chen, MD, of Nanjing (China) First Hospital, Nanjing Medical University. The treatment is an attractive one, as medications recommended for pulmonary arterial hypertension are not recommended for joint pre- and postcapillary pulmonary hypertension (group II pulmonary hypertension).
In PADN-5, 98 patients were randomized to pulmonary artery denervation or to sham denervation plus open-label sildenafil (Viagra), which at the time of trial initiation was thought to be safe and potentially beneficial.
The trial’s main outcome, 6-minute walk distance at 6 months, improved in both groups, according to data reported at the meeting and simultaneously published in JACC Cardiovascular Interventions. But the improvement was about four times greater in the pulmonary artery denervation group. Secondary efficacy outcomes also favored that group, and the rate of fatal pulmonary embolism did not differ for the two groups.
“The PADN-5 trial demonstrates the benefits of pulmonary artery denervation for patients with combined pre- and postcapillary pulmonary hypertension. Patients with preserved and with reduced ejection fraction equally benefited,” summarized Dr. Chen, who pioneered this procedure about 7 years ago. “There was no sign of any harm of sildenafil in patients with combined pre- and postcapillary pulmonary hypertension.”
Trial critique
“This is a very difficult study to conduct, being able to recruit patients and actually have these procedures done,” commented press conference moderator Ori Ben-Yehuda, MD, professor of clinical medicine and director, coronary care unit, UC San Diego Medical Center.
At the same time, he expressed some reservations about the trial. “Sildenafil in the control group might actually be expected to ... decrease your effect size. Also, particularly in men, perhaps even in women, it might unblind them to which group they are in and undermine your sham design,” he noted. In addition, some hemodynamic changes after pulmonary artery denervation – a decrease in wedge pressure and an increase in ejection fraction – were puzzling.
“We need a lot more data here. There are some issues with this trial in terms of design, and we haven’t even gotten into the issue of whether there were core labs, whether the echoes, the hemodynamics, were read blindly,” Dr. Ben-Yehuda maintained. “This issue of secondary or group II pulmonary hypertension due to left heart failure is one that has been very frustrating in terms of actual PA-specific therapies. So this is an important step further, but it needs confirmation in truly sham-controlled trials that have no potential for unblinding.”
The catheter used in PADN-5 is available in China but has not received clearance in the United States, he pointed out. “There are alternative or competing technologies, one using ultrasound, for example, that has a very similar approach. … We’ll have to see how it ends up [performing].”
Trial details
Patients in the PADN-5 pulmonary artery denervation group underwent ablation only in the periconjunctional area between the distal main trunk and the left ostial branch with a multifunction catheter having premounted electrodes. Those in the control group underwent a sham procedure, with catheter positioning at the target sites and connection to a generator but no ablation, and were given open-label sildenafil. All additionally received standard heart failure medical therapy. (No sildenafil placebo was used in the denervation group.)
Trial results reported at the meeting, which is sponsored by the Cardiovascular Research Foundation, showed that most echocardiographic and hemodynamic measures improved more in the pulmonary artery denervation group.
The greater improvement in 6-minute walk test with denervation versus sham sildenafil at 6 months was evident in a variety of measures: absolute median distance walked (432.5 m vs. 358 m) and mean distance walked (434.6 m vs. 359.4 m), and absolute increase (80 m vs. 17.5 m) and relative increase (21.4% vs. 4.9%) The difference was significant for all measures at P less than .001.
The denervation group had a comparatively greater reduction of pulmonary vascular resistance (29.8% vs. 3.4%; P less than .001) and were less likely to experience clinical worsening (16.7% vs. 40.0%; P = .014).
There was a single fatal pulmonary embolism in each treatment group. Of the seven total deaths, two occurred in the denervation group (one attributable to pump failure, one a sudden death) and five occurred in the sham sildenafil group (all but one attributable to pump failure).
Dr. Chen disclosed that he had no relevant conflicts of interest. The trial was sponsored by Nanjing First Hospital, Nanjing Medical University.
SOURCE: Chen S-J et al. TCT 2018. JACC Cardiovasc Interv. 2018 Sep 23.
SAN DIEGO – Pulmonary artery denervation is efficacious for treating combined pre- and postcapillary pulmonary hypertension attributable to left heart failure, based on results of the Chinese PADN-5 trial reported at the Transcatheter Cardiovascular Therapeutics annual meeting.
This ablative treatment has been studied among patients with pulmonary hypertension attributable to other etiologies, but not in randomized fashion among this population, noted lead investigator Shao-Liang Chen, MD, of Nanjing (China) First Hospital, Nanjing Medical University. The treatment is an attractive one, as medications recommended for pulmonary arterial hypertension are not recommended for joint pre- and postcapillary pulmonary hypertension (group II pulmonary hypertension).
In PADN-5, 98 patients were randomized to pulmonary artery denervation or to sham denervation plus open-label sildenafil (Viagra), which at the time of trial initiation was thought to be safe and potentially beneficial.
The trial’s main outcome, 6-minute walk distance at 6 months, improved in both groups, according to data reported at the meeting and simultaneously published in JACC Cardiovascular Interventions. But the improvement was about four times greater in the pulmonary artery denervation group. Secondary efficacy outcomes also favored that group, and the rate of fatal pulmonary embolism did not differ for the two groups.
“The PADN-5 trial demonstrates the benefits of pulmonary artery denervation for patients with combined pre- and postcapillary pulmonary hypertension. Patients with preserved and with reduced ejection fraction equally benefited,” summarized Dr. Chen, who pioneered this procedure about 7 years ago. “There was no sign of any harm of sildenafil in patients with combined pre- and postcapillary pulmonary hypertension.”
Trial critique
“This is a very difficult study to conduct, being able to recruit patients and actually have these procedures done,” commented press conference moderator Ori Ben-Yehuda, MD, professor of clinical medicine and director, coronary care unit, UC San Diego Medical Center.
At the same time, he expressed some reservations about the trial. “Sildenafil in the control group might actually be expected to ... decrease your effect size. Also, particularly in men, perhaps even in women, it might unblind them to which group they are in and undermine your sham design,” he noted. In addition, some hemodynamic changes after pulmonary artery denervation – a decrease in wedge pressure and an increase in ejection fraction – were puzzling.
“We need a lot more data here. There are some issues with this trial in terms of design, and we haven’t even gotten into the issue of whether there were core labs, whether the echoes, the hemodynamics, were read blindly,” Dr. Ben-Yehuda maintained. “This issue of secondary or group II pulmonary hypertension due to left heart failure is one that has been very frustrating in terms of actual PA-specific therapies. So this is an important step further, but it needs confirmation in truly sham-controlled trials that have no potential for unblinding.”
The catheter used in PADN-5 is available in China but has not received clearance in the United States, he pointed out. “There are alternative or competing technologies, one using ultrasound, for example, that has a very similar approach. … We’ll have to see how it ends up [performing].”
Trial details
Patients in the PADN-5 pulmonary artery denervation group underwent ablation only in the periconjunctional area between the distal main trunk and the left ostial branch with a multifunction catheter having premounted electrodes. Those in the control group underwent a sham procedure, with catheter positioning at the target sites and connection to a generator but no ablation, and were given open-label sildenafil. All additionally received standard heart failure medical therapy. (No sildenafil placebo was used in the denervation group.)
Trial results reported at the meeting, which is sponsored by the Cardiovascular Research Foundation, showed that most echocardiographic and hemodynamic measures improved more in the pulmonary artery denervation group.
The greater improvement in 6-minute walk test with denervation versus sham sildenafil at 6 months was evident in a variety of measures: absolute median distance walked (432.5 m vs. 358 m) and mean distance walked (434.6 m vs. 359.4 m), and absolute increase (80 m vs. 17.5 m) and relative increase (21.4% vs. 4.9%) The difference was significant for all measures at P less than .001.
The denervation group had a comparatively greater reduction of pulmonary vascular resistance (29.8% vs. 3.4%; P less than .001) and were less likely to experience clinical worsening (16.7% vs. 40.0%; P = .014).
There was a single fatal pulmonary embolism in each treatment group. Of the seven total deaths, two occurred in the denervation group (one attributable to pump failure, one a sudden death) and five occurred in the sham sildenafil group (all but one attributable to pump failure).
Dr. Chen disclosed that he had no relevant conflicts of interest. The trial was sponsored by Nanjing First Hospital, Nanjing Medical University.
SOURCE: Chen S-J et al. TCT 2018. JACC Cardiovasc Interv. 2018 Sep 23.
REPORTING FROM TCT 2018
Key clinical point: Pulmonary artery denervation is efficacious for treating pulmonary hypertension related to heart failure.
Major finding: Improvement in 6-minute walk distance was greater with pulmonary artery denervation than with sham denervation plus sildenafil (21.4% vs. 4.9%; P less than .001).
Study details: PADN-5 is a randomized controlled trial among 98 patients with combined pre- and postcapillary pulmonary hypertension secondary to left heart failure (group II pulmonary hypertension).
Disclosures: Dr. Chen disclosed that he had no relevant conflicts of interest. The trial was sponsored by Nanjing First Hospital, Nanjing Medical University.
Source: Chen S-L et al. TCT 2018. JACC Cardiovasc Interv. 2018 Sep 23.
ID experts urge widespread flu vaccination for 2018-2019 season
WASHINGTON – The flu vaccine may not be perfect, but it can reduce the severity of illness and curb the risk of spreading the disease to others, William Schaffner, MD, emphasized at a press conference held by the National Foundation for Infectious Diseases.
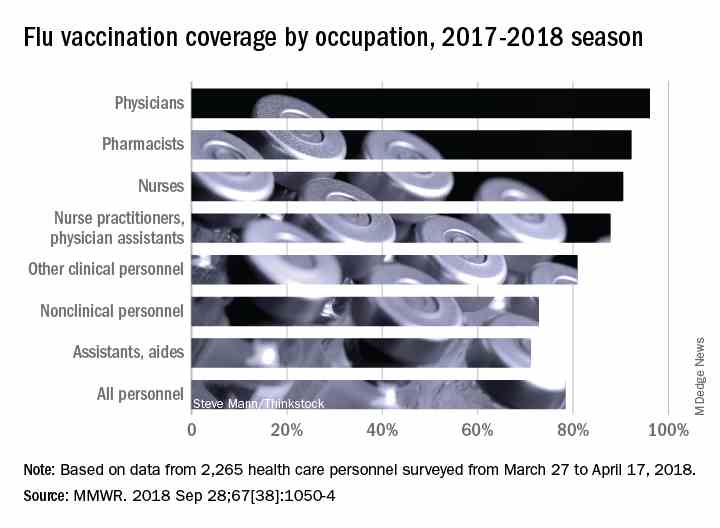
“Give the vaccine credit for softening the blow,” said Dr. Schaffner, medical director of NFID and a professor at Vanderbilt University in Nashville.
Dr. Schaffner and a panel of experts including U.S. Surgeon General Jerome M. Adams, MD, encouraged the public and the health care community to follow recommendation from the Centers for Disease Control & Prevention that everyone aged 6 months and older receive an influenza vaccine.
Dr. Schaffner shared recent data showing that complications from the flu don’t stop when the acute illness resolves. Acute influenza causes a whole-body inflammatory reaction, and consequently “there is an increased risk of heart attack and stroke during the 2-4 weeks of recovery from acute influenza,” he said. In addition, older adults who experience acute flu and are already frail may never regain their pre-flu level of function, as the flu can start a “domino effect of decline and disability.”
Despite last year’s severe flu season that included 180 deaths in children, vaccination remains the most effective protection against the flu, Dr. Adams said.
This year, between 163 million and 168 million doses of vaccine will be available in the United States. The vaccine is available in a range of settings including doctors’ offices, pharmacies, grocery stores, and workplaces, said Dr. Adams.
Flu vaccine choices this year include a return of the live-attenuated influenza vaccine (LAIV) given via nasal spray, along with the standard influenza vaccine that includes either three influenza viruses (trivalent, with two influenza A and one influenza B) or four influenza viruses (quadrivalent, with two influenza A and two influenza B). Other options are adjuvanted vaccine and high-dose vaccine for adults aged 65 years and older, and a cell-based and recombinant vaccine as alternatives to egg-based vaccines.
Dr. Adams emphasized the importance of healthy people getting vaccinated to protect the community. “All the people who died from the flu caught it from someone else,” he said.
The message to health care providers remains the same: Recommend the flu vaccine to patients at every opportunity, and lead by example and get vaccinated yourself, Dr. Adams said. He noted this year’s strategies to promote flu vaccination on social media, and encouraged clinicians to recommend the flu shot to their patients and to showcase their own shots via the #FightFlu hashtag.
Vaccination among health care personnel last year was approximately 78%, which is a plateau over the past several years (MMWR 2018; 67:1050-54).
Be prepared to offer antivirals to patients as appropriate, and to promote the pneumococcal vaccine to eligible older adults as well, to protect not only themselves, but their contacts and the community, Dr. Adams emphasized. Currently approved antiviral drugs recommended for the 2018-2019 flu season: oseltamivir, zanamivir, and peramivir.
Wendy Sue Swanson, MD, of Seattle Children’s Hospital, stressed the importance of flu vaccination for all children, given their ability to spread viral infections. She noted a concerning 2% drop in vaccinations for children aged 6 months to 4 years, although vaccination coverage in this group was highest among children overall, at approximately 68% last season.
Last year, approximately 80% of the child deaths from flu occurred in unvaccinated children, but the vaccine has been shown to reduce the likelihood of hospitalization or death even if a child does become ill, Dr. Swanson said.
Laura E. Riley, MD, of Weill Cornell Medical Center, noted that vaccination of pregnant women has plateaued in recent years, and was 49% last year. “Our goal is 80% plus,” she said. Data show that pregnant women who received flu vaccination were 40% less likely to be hospitalized for the flu, she noted. The American College of Obstetricians and Gynecologists recommends flu vaccination as safe during any trimester, and valuable to both mothers and newborns because it provides protective antibodies during the first 6 months of life before babies can receive their own vaccinations, Dr. Riley said.
More information about this year’s flu season is available from the CDC and NFID.
WASHINGTON – The flu vaccine may not be perfect, but it can reduce the severity of illness and curb the risk of spreading the disease to others, William Schaffner, MD, emphasized at a press conference held by the National Foundation for Infectious Diseases.

“Give the vaccine credit for softening the blow,” said Dr. Schaffner, medical director of NFID and a professor at Vanderbilt University in Nashville.
Dr. Schaffner and a panel of experts including U.S. Surgeon General Jerome M. Adams, MD, encouraged the public and the health care community to follow recommendation from the Centers for Disease Control & Prevention that everyone aged 6 months and older receive an influenza vaccine.
Dr. Schaffner shared recent data showing that complications from the flu don’t stop when the acute illness resolves. Acute influenza causes a whole-body inflammatory reaction, and consequently “there is an increased risk of heart attack and stroke during the 2-4 weeks of recovery from acute influenza,” he said. In addition, older adults who experience acute flu and are already frail may never regain their pre-flu level of function, as the flu can start a “domino effect of decline and disability.”
Despite last year’s severe flu season that included 180 deaths in children, vaccination remains the most effective protection against the flu, Dr. Adams said.
This year, between 163 million and 168 million doses of vaccine will be available in the United States. The vaccine is available in a range of settings including doctors’ offices, pharmacies, grocery stores, and workplaces, said Dr. Adams.
Flu vaccine choices this year include a return of the live-attenuated influenza vaccine (LAIV) given via nasal spray, along with the standard influenza vaccine that includes either three influenza viruses (trivalent, with two influenza A and one influenza B) or four influenza viruses (quadrivalent, with two influenza A and two influenza B). Other options are adjuvanted vaccine and high-dose vaccine for adults aged 65 years and older, and a cell-based and recombinant vaccine as alternatives to egg-based vaccines.
Dr. Adams emphasized the importance of healthy people getting vaccinated to protect the community. “All the people who died from the flu caught it from someone else,” he said.
The message to health care providers remains the same: Recommend the flu vaccine to patients at every opportunity, and lead by example and get vaccinated yourself, Dr. Adams said. He noted this year’s strategies to promote flu vaccination on social media, and encouraged clinicians to recommend the flu shot to their patients and to showcase their own shots via the #FightFlu hashtag.
Vaccination among health care personnel last year was approximately 78%, which is a plateau over the past several years (MMWR 2018; 67:1050-54).
Be prepared to offer antivirals to patients as appropriate, and to promote the pneumococcal vaccine to eligible older adults as well, to protect not only themselves, but their contacts and the community, Dr. Adams emphasized. Currently approved antiviral drugs recommended for the 2018-2019 flu season: oseltamivir, zanamivir, and peramivir.
Wendy Sue Swanson, MD, of Seattle Children’s Hospital, stressed the importance of flu vaccination for all children, given their ability to spread viral infections. She noted a concerning 2% drop in vaccinations for children aged 6 months to 4 years, although vaccination coverage in this group was highest among children overall, at approximately 68% last season.
Last year, approximately 80% of the child deaths from flu occurred in unvaccinated children, but the vaccine has been shown to reduce the likelihood of hospitalization or death even if a child does become ill, Dr. Swanson said.
Laura E. Riley, MD, of Weill Cornell Medical Center, noted that vaccination of pregnant women has plateaued in recent years, and was 49% last year. “Our goal is 80% plus,” she said. Data show that pregnant women who received flu vaccination were 40% less likely to be hospitalized for the flu, she noted. The American College of Obstetricians and Gynecologists recommends flu vaccination as safe during any trimester, and valuable to both mothers and newborns because it provides protective antibodies during the first 6 months of life before babies can receive their own vaccinations, Dr. Riley said.
More information about this year’s flu season is available from the CDC and NFID.
WASHINGTON – The flu vaccine may not be perfect, but it can reduce the severity of illness and curb the risk of spreading the disease to others, William Schaffner, MD, emphasized at a press conference held by the National Foundation for Infectious Diseases.

“Give the vaccine credit for softening the blow,” said Dr. Schaffner, medical director of NFID and a professor at Vanderbilt University in Nashville.
Dr. Schaffner and a panel of experts including U.S. Surgeon General Jerome M. Adams, MD, encouraged the public and the health care community to follow recommendation from the Centers for Disease Control & Prevention that everyone aged 6 months and older receive an influenza vaccine.
Dr. Schaffner shared recent data showing that complications from the flu don’t stop when the acute illness resolves. Acute influenza causes a whole-body inflammatory reaction, and consequently “there is an increased risk of heart attack and stroke during the 2-4 weeks of recovery from acute influenza,” he said. In addition, older adults who experience acute flu and are already frail may never regain their pre-flu level of function, as the flu can start a “domino effect of decline and disability.”
Despite last year’s severe flu season that included 180 deaths in children, vaccination remains the most effective protection against the flu, Dr. Adams said.
This year, between 163 million and 168 million doses of vaccine will be available in the United States. The vaccine is available in a range of settings including doctors’ offices, pharmacies, grocery stores, and workplaces, said Dr. Adams.
Flu vaccine choices this year include a return of the live-attenuated influenza vaccine (LAIV) given via nasal spray, along with the standard influenza vaccine that includes either three influenza viruses (trivalent, with two influenza A and one influenza B) or four influenza viruses (quadrivalent, with two influenza A and two influenza B). Other options are adjuvanted vaccine and high-dose vaccine for adults aged 65 years and older, and a cell-based and recombinant vaccine as alternatives to egg-based vaccines.
Dr. Adams emphasized the importance of healthy people getting vaccinated to protect the community. “All the people who died from the flu caught it from someone else,” he said.
The message to health care providers remains the same: Recommend the flu vaccine to patients at every opportunity, and lead by example and get vaccinated yourself, Dr. Adams said. He noted this year’s strategies to promote flu vaccination on social media, and encouraged clinicians to recommend the flu shot to their patients and to showcase their own shots via the #FightFlu hashtag.
Vaccination among health care personnel last year was approximately 78%, which is a plateau over the past several years (MMWR 2018; 67:1050-54).
Be prepared to offer antivirals to patients as appropriate, and to promote the pneumococcal vaccine to eligible older adults as well, to protect not only themselves, but their contacts and the community, Dr. Adams emphasized. Currently approved antiviral drugs recommended for the 2018-2019 flu season: oseltamivir, zanamivir, and peramivir.
Wendy Sue Swanson, MD, of Seattle Children’s Hospital, stressed the importance of flu vaccination for all children, given their ability to spread viral infections. She noted a concerning 2% drop in vaccinations for children aged 6 months to 4 years, although vaccination coverage in this group was highest among children overall, at approximately 68% last season.
Last year, approximately 80% of the child deaths from flu occurred in unvaccinated children, but the vaccine has been shown to reduce the likelihood of hospitalization or death even if a child does become ill, Dr. Swanson said.
Laura E. Riley, MD, of Weill Cornell Medical Center, noted that vaccination of pregnant women has plateaued in recent years, and was 49% last year. “Our goal is 80% plus,” she said. Data show that pregnant women who received flu vaccination were 40% less likely to be hospitalized for the flu, she noted. The American College of Obstetricians and Gynecologists recommends flu vaccination as safe during any trimester, and valuable to both mothers and newborns because it provides protective antibodies during the first 6 months of life before babies can receive their own vaccinations, Dr. Riley said.
More information about this year’s flu season is available from the CDC and NFID.
FROM AN NFID PRESS CONFERENCE
Point-of-care test for respiratory viruses lowers antibiotic use
Routine testing in the ED is advocated
PARIS – Using a point-of-care test for viral pathogens, hospital admissions were avoided in about a third of emergency department patients with suspected respiratory infection when other clinical signs also suggested a low risk of a bacterial pathogen, according to a single-center experience presented at the annual congress of the European Respiratory Society.
“We found that when patients had point-of-care respiratory viral testing soon after they were admitted to the emergency department, we were able to reduce unnecessary admission and improve bed flow in our center,” reported Kay Roy, MBBS, consultant physician in respiratory medicine, West Hertfordshire (England) Hospital NHS Trust.
In a protocol that was launched at Dr. Kay’s institution in January 2018, the point-of-care viral test was combined with other clinical factors, particularly chest x-rays and elevated C-reactive protein (CRP), to determine whether patients had a viral pathogen and whether they could be discharged without antibiotics.
“Clinical judgment will always be required in individual patient decisions regarding antibiotic avoidance and early discharge,” Dr. Roy maintained. “But the point-of-care viral assay can be integrated into a strategy that permits more informed and rapid decision-making.”
This assertion is supported by the experience using a protocol anchored with the point-of-care viral test over a 4-month period. During this time, 901 patients with respiratory symptoms suspected of having a viral etiology were evaluated with the proprietary point-of-care device called FilmArray (bioMérieux).
From a sample taken with a nasopharyngeal swab, the test can identify a broad array of viruses using polymerase chain reaction technology in less than 45 minutes. However, the ED protocol for considering discharge without antibiotics requires additional evidence that the pathogen is viral, including a normal chest x-ray and a CRP less than 50 mg/L.
Of the 901 patients tested, a substantial proportion of whom had chronic obstructive pulmonary disease (COPD) or asthma, 507 (56%) tested positive for at least one virus, including influenza, rhinoviruses, coronaviruses, and adenovirus. Of these, 239 had normal chest x-rays and CRPs less than 50 mg/L. Because of the severity of symptoms or other clinical considerations, 154 patients were admitted, but 85 (36% of those meeting protocol criteria) were discharged without an antibiotic prescription.
“Antibiotics were continued in 90% of the patients who had an abnormal chest x-ray and abnormal CRP,” Dr. Roy reported. However, an objective strategy that permits clinicians to discharge patients at very low risk of a bacterial infection has many advantages even if it applies to a relatively modest proportion of those tested, according to Dr. Roy.
“Each respiratory admission can cost around [2,000 pounds] at our center,” reported Dr. Kay, referring to a figure equivalent to more than $2,600. In addition, she said that avoiding hospitalization frees up hospital beds and facilitates improved antimicrobial stewardship, which is vital to stem resistance.
Avoiding antibiotic use in patients with viral respiratory infections also is relevant to improved antibiotic stewardship in the community. For this reason, a randomized trial with a similar protocol involving the point-of-care viral test is planned in the outpatient setting. According to Dr. Roy, this will involve a community hub to which patients can be referred for testing and clinical evaluation.
“We hope that the quality of care can be improved with the point-of-care test for respiratory viruses as well as helping to reduce antibiotic resistance,” Dr. Roy said.
This approach is promising, according to Tobias Welte, MD, of the department of respiratory medicine at Hannover (Germany) Medical School, but he cautioned that it is not a standard approach.
“The protocol described by Dr. Roy will have to be compared to guidelines and recommended best clinical practice to confirm its usefulness,” he said, while conceding that any strategy that reduces unnecessary hospitalizations deserves further evaluation.
Routine testing in the ED is advocated
Routine testing in the ED is advocated
PARIS – Using a point-of-care test for viral pathogens, hospital admissions were avoided in about a third of emergency department patients with suspected respiratory infection when other clinical signs also suggested a low risk of a bacterial pathogen, according to a single-center experience presented at the annual congress of the European Respiratory Society.
“We found that when patients had point-of-care respiratory viral testing soon after they were admitted to the emergency department, we were able to reduce unnecessary admission and improve bed flow in our center,” reported Kay Roy, MBBS, consultant physician in respiratory medicine, West Hertfordshire (England) Hospital NHS Trust.
In a protocol that was launched at Dr. Kay’s institution in January 2018, the point-of-care viral test was combined with other clinical factors, particularly chest x-rays and elevated C-reactive protein (CRP), to determine whether patients had a viral pathogen and whether they could be discharged without antibiotics.
“Clinical judgment will always be required in individual patient decisions regarding antibiotic avoidance and early discharge,” Dr. Roy maintained. “But the point-of-care viral assay can be integrated into a strategy that permits more informed and rapid decision-making.”
This assertion is supported by the experience using a protocol anchored with the point-of-care viral test over a 4-month period. During this time, 901 patients with respiratory symptoms suspected of having a viral etiology were evaluated with the proprietary point-of-care device called FilmArray (bioMérieux).
From a sample taken with a nasopharyngeal swab, the test can identify a broad array of viruses using polymerase chain reaction technology in less than 45 minutes. However, the ED protocol for considering discharge without antibiotics requires additional evidence that the pathogen is viral, including a normal chest x-ray and a CRP less than 50 mg/L.
Of the 901 patients tested, a substantial proportion of whom had chronic obstructive pulmonary disease (COPD) or asthma, 507 (56%) tested positive for at least one virus, including influenza, rhinoviruses, coronaviruses, and adenovirus. Of these, 239 had normal chest x-rays and CRPs less than 50 mg/L. Because of the severity of symptoms or other clinical considerations, 154 patients were admitted, but 85 (36% of those meeting protocol criteria) were discharged without an antibiotic prescription.
“Antibiotics were continued in 90% of the patients who had an abnormal chest x-ray and abnormal CRP,” Dr. Roy reported. However, an objective strategy that permits clinicians to discharge patients at very low risk of a bacterial infection has many advantages even if it applies to a relatively modest proportion of those tested, according to Dr. Roy.
“Each respiratory admission can cost around [2,000 pounds] at our center,” reported Dr. Kay, referring to a figure equivalent to more than $2,600. In addition, she said that avoiding hospitalization frees up hospital beds and facilitates improved antimicrobial stewardship, which is vital to stem resistance.
Avoiding antibiotic use in patients with viral respiratory infections also is relevant to improved antibiotic stewardship in the community. For this reason, a randomized trial with a similar protocol involving the point-of-care viral test is planned in the outpatient setting. According to Dr. Roy, this will involve a community hub to which patients can be referred for testing and clinical evaluation.
“We hope that the quality of care can be improved with the point-of-care test for respiratory viruses as well as helping to reduce antibiotic resistance,” Dr. Roy said.
This approach is promising, according to Tobias Welte, MD, of the department of respiratory medicine at Hannover (Germany) Medical School, but he cautioned that it is not a standard approach.
“The protocol described by Dr. Roy will have to be compared to guidelines and recommended best clinical practice to confirm its usefulness,” he said, while conceding that any strategy that reduces unnecessary hospitalizations deserves further evaluation.
PARIS – Using a point-of-care test for viral pathogens, hospital admissions were avoided in about a third of emergency department patients with suspected respiratory infection when other clinical signs also suggested a low risk of a bacterial pathogen, according to a single-center experience presented at the annual congress of the European Respiratory Society.
“We found that when patients had point-of-care respiratory viral testing soon after they were admitted to the emergency department, we were able to reduce unnecessary admission and improve bed flow in our center,” reported Kay Roy, MBBS, consultant physician in respiratory medicine, West Hertfordshire (England) Hospital NHS Trust.
In a protocol that was launched at Dr. Kay’s institution in January 2018, the point-of-care viral test was combined with other clinical factors, particularly chest x-rays and elevated C-reactive protein (CRP), to determine whether patients had a viral pathogen and whether they could be discharged without antibiotics.
“Clinical judgment will always be required in individual patient decisions regarding antibiotic avoidance and early discharge,” Dr. Roy maintained. “But the point-of-care viral assay can be integrated into a strategy that permits more informed and rapid decision-making.”
This assertion is supported by the experience using a protocol anchored with the point-of-care viral test over a 4-month period. During this time, 901 patients with respiratory symptoms suspected of having a viral etiology were evaluated with the proprietary point-of-care device called FilmArray (bioMérieux).
From a sample taken with a nasopharyngeal swab, the test can identify a broad array of viruses using polymerase chain reaction technology in less than 45 minutes. However, the ED protocol for considering discharge without antibiotics requires additional evidence that the pathogen is viral, including a normal chest x-ray and a CRP less than 50 mg/L.
Of the 901 patients tested, a substantial proportion of whom had chronic obstructive pulmonary disease (COPD) or asthma, 507 (56%) tested positive for at least one virus, including influenza, rhinoviruses, coronaviruses, and adenovirus. Of these, 239 had normal chest x-rays and CRPs less than 50 mg/L. Because of the severity of symptoms or other clinical considerations, 154 patients were admitted, but 85 (36% of those meeting protocol criteria) were discharged without an antibiotic prescription.
“Antibiotics were continued in 90% of the patients who had an abnormal chest x-ray and abnormal CRP,” Dr. Roy reported. However, an objective strategy that permits clinicians to discharge patients at very low risk of a bacterial infection has many advantages even if it applies to a relatively modest proportion of those tested, according to Dr. Roy.
“Each respiratory admission can cost around [2,000 pounds] at our center,” reported Dr. Kay, referring to a figure equivalent to more than $2,600. In addition, she said that avoiding hospitalization frees up hospital beds and facilitates improved antimicrobial stewardship, which is vital to stem resistance.
Avoiding antibiotic use in patients with viral respiratory infections also is relevant to improved antibiotic stewardship in the community. For this reason, a randomized trial with a similar protocol involving the point-of-care viral test is planned in the outpatient setting. According to Dr. Roy, this will involve a community hub to which patients can be referred for testing and clinical evaluation.
“We hope that the quality of care can be improved with the point-of-care test for respiratory viruses as well as helping to reduce antibiotic resistance,” Dr. Roy said.
This approach is promising, according to Tobias Welte, MD, of the department of respiratory medicine at Hannover (Germany) Medical School, but he cautioned that it is not a standard approach.
“The protocol described by Dr. Roy will have to be compared to guidelines and recommended best clinical practice to confirm its usefulness,” he said, while conceding that any strategy that reduces unnecessary hospitalizations deserves further evaluation.
REPORTING FROM THE ERS CONGRESS 2018
Key clinical point:
Major finding: Of patients with a negative chest x-ray and low CRP level, 36% avoided hospital admission due to a positive test for a virus.
Study details: A case series.
Disclosures: Dr. Roy reports no financial relationships relevant to this study.