User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
Powered by CHEST Physician, Clinician Reviews, MDedge Family Medicine, Internal Medicine News, and The Journal of Clinical Outcomes Management.
NELSON trial: CT Screening reduces lung cancer deaths
TORONTO – Computed tomography screening among asymptomatic men at high risk for lung cancer reduced lung cancer deaths by a highly statistically significant 26% at 10 years and appeared to reduce lung cancer mortality risk even more in women in the population-based, NELSON trial.
The findings from this large controlled trial encompassing more than 27,000 CT scans in 15,792 individuals support those from the National Lung Screening Trials (NLST), published in 2011, and should “inform and direct future CT screening programs worldwide,” according to Harry de Koning, MD, who presented the findings at the World Conference on Lung Cancer.
Participants were randomized to CT screening at baseline, 1, 3, and 5.5 years after randomization, or to a control group that received usual care. Overall 157 lung cancer deaths occurred in the screening arm vs. 250 in the control arm. Detection rates varied between 0.8% and 1.1% across screenings (0.9% overall), and the positive predictive value of screening was 41%, Dr. de Koning of Erasmus Medical Center, Rotterdam, the Netherlands, said at the meeting sponsored by the International Association for the Study of Lung Cancer.
Notably, 69% of the 243 lung cancers detected by screening were detected at stage 1A or 1B, compared with 10%-12% being detected at stage 4 in about 50% of control patients and based on registry data in the Netherlands.
“There’s huge importance of this early detection in the screening arm,” Dr. de Koning said.
Additionally, an analysis of a subset of those with lung cancer showed a significant threefold increase in surgical treatment among the screened patients vs. those in the control arm who developed lung cancer (67.7% vs. 24.5%), he said.
CT screening reduced the risk of death from lung cancer by 9% to 41% in men over the course of the study, with an overall reduction of 26% at 10 years, and in a smaller subset of women, the rate-ratio of dying from lung cancer varied from 0.39 to 0.61 at different years of follow-up, he said, noting that this suggests a “significant and even larger reduction” in women.
Study participants were individuals aged 50-74 years in the Netherlands and Leuven, Belgium, who were considered at high risk for lung cancer based on responses to a general questionnaire. Participants’ records were linked with national registries with 100% coverage regarding cancer diagnosis and date and cause of death, and medical records for deceased lung cancer patients were reviewed by a blinded expert panel through 2013, and for the remaining study years cause of death as reported by Statistics Netherlands was used. Compliance among those randomized to the screening group was 86%, Dr. de Koning said.
“These findings show that CT screenings are an effective way to assess lung nodules in people at high risk for lung cancer, often leading to detection of suspicious nodules and subsequent surgical intervention at relatively low rates and with few false positives, and can positively increase the chances of cure in this devastating disease,” Dr. de Koning said in a press statement. “It is the second-largest trial in the world, with an even more favorable outcome than the first trial, the NLST, showed. These results should be used to inform and direct future CT screening in the world.”
During a press briefing, in response to a question about whether lung cancer screening should be offered more widely, he said that yes, countries – including the United States – should take note that “now two large-scale trials show large benefit.”
Dr. de Koning reported having no disclosures.
TORONTO – Computed tomography screening among asymptomatic men at high risk for lung cancer reduced lung cancer deaths by a highly statistically significant 26% at 10 years and appeared to reduce lung cancer mortality risk even more in women in the population-based, NELSON trial.
The findings from this large controlled trial encompassing more than 27,000 CT scans in 15,792 individuals support those from the National Lung Screening Trials (NLST), published in 2011, and should “inform and direct future CT screening programs worldwide,” according to Harry de Koning, MD, who presented the findings at the World Conference on Lung Cancer.
Participants were randomized to CT screening at baseline, 1, 3, and 5.5 years after randomization, or to a control group that received usual care. Overall 157 lung cancer deaths occurred in the screening arm vs. 250 in the control arm. Detection rates varied between 0.8% and 1.1% across screenings (0.9% overall), and the positive predictive value of screening was 41%, Dr. de Koning of Erasmus Medical Center, Rotterdam, the Netherlands, said at the meeting sponsored by the International Association for the Study of Lung Cancer.
Notably, 69% of the 243 lung cancers detected by screening were detected at stage 1A or 1B, compared with 10%-12% being detected at stage 4 in about 50% of control patients and based on registry data in the Netherlands.
“There’s huge importance of this early detection in the screening arm,” Dr. de Koning said.
Additionally, an analysis of a subset of those with lung cancer showed a significant threefold increase in surgical treatment among the screened patients vs. those in the control arm who developed lung cancer (67.7% vs. 24.5%), he said.
CT screening reduced the risk of death from lung cancer by 9% to 41% in men over the course of the study, with an overall reduction of 26% at 10 years, and in a smaller subset of women, the rate-ratio of dying from lung cancer varied from 0.39 to 0.61 at different years of follow-up, he said, noting that this suggests a “significant and even larger reduction” in women.
Study participants were individuals aged 50-74 years in the Netherlands and Leuven, Belgium, who were considered at high risk for lung cancer based on responses to a general questionnaire. Participants’ records were linked with national registries with 100% coverage regarding cancer diagnosis and date and cause of death, and medical records for deceased lung cancer patients were reviewed by a blinded expert panel through 2013, and for the remaining study years cause of death as reported by Statistics Netherlands was used. Compliance among those randomized to the screening group was 86%, Dr. de Koning said.
“These findings show that CT screenings are an effective way to assess lung nodules in people at high risk for lung cancer, often leading to detection of suspicious nodules and subsequent surgical intervention at relatively low rates and with few false positives, and can positively increase the chances of cure in this devastating disease,” Dr. de Koning said in a press statement. “It is the second-largest trial in the world, with an even more favorable outcome than the first trial, the NLST, showed. These results should be used to inform and direct future CT screening in the world.”
During a press briefing, in response to a question about whether lung cancer screening should be offered more widely, he said that yes, countries – including the United States – should take note that “now two large-scale trials show large benefit.”
Dr. de Koning reported having no disclosures.
TORONTO – Computed tomography screening among asymptomatic men at high risk for lung cancer reduced lung cancer deaths by a highly statistically significant 26% at 10 years and appeared to reduce lung cancer mortality risk even more in women in the population-based, NELSON trial.
The findings from this large controlled trial encompassing more than 27,000 CT scans in 15,792 individuals support those from the National Lung Screening Trials (NLST), published in 2011, and should “inform and direct future CT screening programs worldwide,” according to Harry de Koning, MD, who presented the findings at the World Conference on Lung Cancer.
Participants were randomized to CT screening at baseline, 1, 3, and 5.5 years after randomization, or to a control group that received usual care. Overall 157 lung cancer deaths occurred in the screening arm vs. 250 in the control arm. Detection rates varied between 0.8% and 1.1% across screenings (0.9% overall), and the positive predictive value of screening was 41%, Dr. de Koning of Erasmus Medical Center, Rotterdam, the Netherlands, said at the meeting sponsored by the International Association for the Study of Lung Cancer.
Notably, 69% of the 243 lung cancers detected by screening were detected at stage 1A or 1B, compared with 10%-12% being detected at stage 4 in about 50% of control patients and based on registry data in the Netherlands.
“There’s huge importance of this early detection in the screening arm,” Dr. de Koning said.
Additionally, an analysis of a subset of those with lung cancer showed a significant threefold increase in surgical treatment among the screened patients vs. those in the control arm who developed lung cancer (67.7% vs. 24.5%), he said.
CT screening reduced the risk of death from lung cancer by 9% to 41% in men over the course of the study, with an overall reduction of 26% at 10 years, and in a smaller subset of women, the rate-ratio of dying from lung cancer varied from 0.39 to 0.61 at different years of follow-up, he said, noting that this suggests a “significant and even larger reduction” in women.
Study participants were individuals aged 50-74 years in the Netherlands and Leuven, Belgium, who were considered at high risk for lung cancer based on responses to a general questionnaire. Participants’ records were linked with national registries with 100% coverage regarding cancer diagnosis and date and cause of death, and medical records for deceased lung cancer patients were reviewed by a blinded expert panel through 2013, and for the remaining study years cause of death as reported by Statistics Netherlands was used. Compliance among those randomized to the screening group was 86%, Dr. de Koning said.
“These findings show that CT screenings are an effective way to assess lung nodules in people at high risk for lung cancer, often leading to detection of suspicious nodules and subsequent surgical intervention at relatively low rates and with few false positives, and can positively increase the chances of cure in this devastating disease,” Dr. de Koning said in a press statement. “It is the second-largest trial in the world, with an even more favorable outcome than the first trial, the NLST, showed. These results should be used to inform and direct future CT screening in the world.”
During a press briefing, in response to a question about whether lung cancer screening should be offered more widely, he said that yes, countries – including the United States – should take note that “now two large-scale trials show large benefit.”
Dr. de Koning reported having no disclosures.
REPORTING FROM WCLC 2018
Key clinical point: CT screening among high-risk patients significantly reduces lung cancer mortality.
Major finding: CT screening in high-risk patients reduced lung cancer deaths by 26% in men, 39%-61% in women.
Study details: A population-based controlled trial of 15,792 individuals.
Disclosures: Dr. de Koning reported having no disclosures.
Troponin I: Powerful all-cause mortality risk marker in COPD
PARIS – High relative even after researchers adjusted for all major cardiovascular and COPD prognostic indicators, according to a late-breaker presentation at the annual congress of the European Respiratory Society.
Troponin I is detectable in the plasma of most patients with COPD, but relative increases in troponin I correlate with greater relative increases in most cardiovascular and COPD risk factors, according to Benjamin Waschki, MD, Pulmonary Research Institute, LungenClinic, Grosshansdorf, Germany.
The relationship between increased troponin I and increased all-cause mortality was observed in an on-going prospective multicenter cohort of COPD patients followed at 31 centers in Germany. The cohort is called COSYCONET and it began in 2010. The current analysis evaluated 2,020 COPD patients without regard to stage of disease.
There were 136 deaths over the course of follow-up. Without adjustment, the hazard ratio (HR) for death was more than twofold higher in the highest quartile of troponin I (equal to or greater than 6.6 ng/mL), when compared with the lowest (under 2.5 ng/mL) (HR, 2.42; P less than .001). Graphically, the mortality curves for each of the quartiles began to separate at about 12 months, widening in a stepwise manner for greater likelihood of death from the lowest to highest quartiles.
The risk of death from any cause remained elevated for the highest relative to lowest troponin I quartiles after adjusting for cardiovascular risk factors and after adjusting for COPD severity. Again, there was a distinct stepwise separation of the mortality curves for each higher troponin quartile,
Of particular importance, troponin I remained predictive beyond the BODE index, which is a currently employed prognostic mortality predictor in COPD, according to Dr. Waschki. When defining elevated troponin as greater than 6 ng/ML and a high BODE score as greater than 4, mortality was higher for those with a high BODE and low troponin than a high troponin and low BODE, (P less than .001), but a high troponin I was associated with a higher risk of mortality when BODE was low (P less than .001). Moreover, when both troponin I and BODE were elevated, all-cause mortality was more than doubled, relative to those without either risk factor (HR, 2.56; P = .003), Dr. Waschki reported.
After researchers adjusted for major cardiovascular risk factors, such as history of MI and renal impairment, and for major COPD risk factors, such as 6-minute walk test and BODE index, those in the highest quartile had a more than 50% greater risk of death relative to those in the lower quartile over the 3 years of follow-up (HR, 1.69; P = .007), according to Dr. Waschki.
Although troponin I is best known for its diagnostic role in MI, it is now being evaluated as a risk stratifier for many chronic diseases, such as heart failure and chronic kidney disease, explained Dr. Waschki in providing background for this study. He reported that many groups are looking at this as a marker of risk in a variety of chronic diseases.
In fact, a group working independently published a study in COPD just weeks before the ERS Congress that was complementary to those presented by Dr. Waschki. In this study, the goal was to evaluate troponin I as a predictor of cardiovascular events and cardiovascular death (Adamson PD et al. J Am Coll Cardiol 2018;72:1126-37). Performed as a subgroup analysis of 1,599 COPD patients participating in a large treatment trial, there was an almost fourfold increase in the risk of cardiovascular events (HR, 3.7; P = .012) when those in the highest quintile of troponin I (greater than 7.7 ng/ML) were compared with those in the lowest quintile (less than 2.3 ng/mL).
When compared for cardiovascular death, the highest quintile, relative to the lowest quintile, had a more than 20-fold increased risk of cardiovascular death (HR 20.1; P = .005). In the Adamson et al. study, which evaluated inhaled therapies for COPD, treatment response had no impact on troponin I levels or on the risk of cardiovascular events or death.
Based on this study and his own data, Dr. Waschki believes troponin I, which is readily ordered laboratory value, appears to be a useful tool for identifying COPD patients at high risk of death.
“The major message is that after adjusting for all known COPD and cardiovascular risk factors, troponin I remains a significant independent predictor of mortality,” he said.
Dr. Waschki reports no relevant conflicts of interest.
PARIS – High relative even after researchers adjusted for all major cardiovascular and COPD prognostic indicators, according to a late-breaker presentation at the annual congress of the European Respiratory Society.
Troponin I is detectable in the plasma of most patients with COPD, but relative increases in troponin I correlate with greater relative increases in most cardiovascular and COPD risk factors, according to Benjamin Waschki, MD, Pulmonary Research Institute, LungenClinic, Grosshansdorf, Germany.
The relationship between increased troponin I and increased all-cause mortality was observed in an on-going prospective multicenter cohort of COPD patients followed at 31 centers in Germany. The cohort is called COSYCONET and it began in 2010. The current analysis evaluated 2,020 COPD patients without regard to stage of disease.
There were 136 deaths over the course of follow-up. Without adjustment, the hazard ratio (HR) for death was more than twofold higher in the highest quartile of troponin I (equal to or greater than 6.6 ng/mL), when compared with the lowest (under 2.5 ng/mL) (HR, 2.42; P less than .001). Graphically, the mortality curves for each of the quartiles began to separate at about 12 months, widening in a stepwise manner for greater likelihood of death from the lowest to highest quartiles.
The risk of death from any cause remained elevated for the highest relative to lowest troponin I quartiles after adjusting for cardiovascular risk factors and after adjusting for COPD severity. Again, there was a distinct stepwise separation of the mortality curves for each higher troponin quartile,
Of particular importance, troponin I remained predictive beyond the BODE index, which is a currently employed prognostic mortality predictor in COPD, according to Dr. Waschki. When defining elevated troponin as greater than 6 ng/ML and a high BODE score as greater than 4, mortality was higher for those with a high BODE and low troponin than a high troponin and low BODE, (P less than .001), but a high troponin I was associated with a higher risk of mortality when BODE was low (P less than .001). Moreover, when both troponin I and BODE were elevated, all-cause mortality was more than doubled, relative to those without either risk factor (HR, 2.56; P = .003), Dr. Waschki reported.
After researchers adjusted for major cardiovascular risk factors, such as history of MI and renal impairment, and for major COPD risk factors, such as 6-minute walk test and BODE index, those in the highest quartile had a more than 50% greater risk of death relative to those in the lower quartile over the 3 years of follow-up (HR, 1.69; P = .007), according to Dr. Waschki.
Although troponin I is best known for its diagnostic role in MI, it is now being evaluated as a risk stratifier for many chronic diseases, such as heart failure and chronic kidney disease, explained Dr. Waschki in providing background for this study. He reported that many groups are looking at this as a marker of risk in a variety of chronic diseases.
In fact, a group working independently published a study in COPD just weeks before the ERS Congress that was complementary to those presented by Dr. Waschki. In this study, the goal was to evaluate troponin I as a predictor of cardiovascular events and cardiovascular death (Adamson PD et al. J Am Coll Cardiol 2018;72:1126-37). Performed as a subgroup analysis of 1,599 COPD patients participating in a large treatment trial, there was an almost fourfold increase in the risk of cardiovascular events (HR, 3.7; P = .012) when those in the highest quintile of troponin I (greater than 7.7 ng/ML) were compared with those in the lowest quintile (less than 2.3 ng/mL).
When compared for cardiovascular death, the highest quintile, relative to the lowest quintile, had a more than 20-fold increased risk of cardiovascular death (HR 20.1; P = .005). In the Adamson et al. study, which evaluated inhaled therapies for COPD, treatment response had no impact on troponin I levels or on the risk of cardiovascular events or death.
Based on this study and his own data, Dr. Waschki believes troponin I, which is readily ordered laboratory value, appears to be a useful tool for identifying COPD patients at high risk of death.
“The major message is that after adjusting for all known COPD and cardiovascular risk factors, troponin I remains a significant independent predictor of mortality,” he said.
Dr. Waschki reports no relevant conflicts of interest.
PARIS – High relative even after researchers adjusted for all major cardiovascular and COPD prognostic indicators, according to a late-breaker presentation at the annual congress of the European Respiratory Society.
Troponin I is detectable in the plasma of most patients with COPD, but relative increases in troponin I correlate with greater relative increases in most cardiovascular and COPD risk factors, according to Benjamin Waschki, MD, Pulmonary Research Institute, LungenClinic, Grosshansdorf, Germany.
The relationship between increased troponin I and increased all-cause mortality was observed in an on-going prospective multicenter cohort of COPD patients followed at 31 centers in Germany. The cohort is called COSYCONET and it began in 2010. The current analysis evaluated 2,020 COPD patients without regard to stage of disease.
There were 136 deaths over the course of follow-up. Without adjustment, the hazard ratio (HR) for death was more than twofold higher in the highest quartile of troponin I (equal to or greater than 6.6 ng/mL), when compared with the lowest (under 2.5 ng/mL) (HR, 2.42; P less than .001). Graphically, the mortality curves for each of the quartiles began to separate at about 12 months, widening in a stepwise manner for greater likelihood of death from the lowest to highest quartiles.
The risk of death from any cause remained elevated for the highest relative to lowest troponin I quartiles after adjusting for cardiovascular risk factors and after adjusting for COPD severity. Again, there was a distinct stepwise separation of the mortality curves for each higher troponin quartile,
Of particular importance, troponin I remained predictive beyond the BODE index, which is a currently employed prognostic mortality predictor in COPD, according to Dr. Waschki. When defining elevated troponin as greater than 6 ng/ML and a high BODE score as greater than 4, mortality was higher for those with a high BODE and low troponin than a high troponin and low BODE, (P less than .001), but a high troponin I was associated with a higher risk of mortality when BODE was low (P less than .001). Moreover, when both troponin I and BODE were elevated, all-cause mortality was more than doubled, relative to those without either risk factor (HR, 2.56; P = .003), Dr. Waschki reported.
After researchers adjusted for major cardiovascular risk factors, such as history of MI and renal impairment, and for major COPD risk factors, such as 6-minute walk test and BODE index, those in the highest quartile had a more than 50% greater risk of death relative to those in the lower quartile over the 3 years of follow-up (HR, 1.69; P = .007), according to Dr. Waschki.
Although troponin I is best known for its diagnostic role in MI, it is now being evaluated as a risk stratifier for many chronic diseases, such as heart failure and chronic kidney disease, explained Dr. Waschki in providing background for this study. He reported that many groups are looking at this as a marker of risk in a variety of chronic diseases.
In fact, a group working independently published a study in COPD just weeks before the ERS Congress that was complementary to those presented by Dr. Waschki. In this study, the goal was to evaluate troponin I as a predictor of cardiovascular events and cardiovascular death (Adamson PD et al. J Am Coll Cardiol 2018;72:1126-37). Performed as a subgroup analysis of 1,599 COPD patients participating in a large treatment trial, there was an almost fourfold increase in the risk of cardiovascular events (HR, 3.7; P = .012) when those in the highest quintile of troponin I (greater than 7.7 ng/ML) were compared with those in the lowest quintile (less than 2.3 ng/mL).
When compared for cardiovascular death, the highest quintile, relative to the lowest quintile, had a more than 20-fold increased risk of cardiovascular death (HR 20.1; P = .005). In the Adamson et al. study, which evaluated inhaled therapies for COPD, treatment response had no impact on troponin I levels or on the risk of cardiovascular events or death.
Based on this study and his own data, Dr. Waschki believes troponin I, which is readily ordered laboratory value, appears to be a useful tool for identifying COPD patients at high risk of death.
“The major message is that after adjusting for all known COPD and cardiovascular risk factors, troponin I remains a significant independent predictor of mortality,” he said.
Dr. Waschki reports no relevant conflicts of interest.
REPORTING FROM ERS CONGRESS 2018
Key clinical point: Elevated troponin I identifies COPD patients with increased mortality risk independent of all other clinical risk markers.
Major finding: With high troponin I levels, all-cause mortality was increased 69% after researchers adjusted for other risk markers.
Study details: Analysis drawn from on-going multicenter cohort study
Disclosures: Dr. Waschki reports no relevant conflicts of interest.
Guideline-recommended tests’ prognostic ability affirmed in prostanoid-treated PAH
The results of guideline-recommended prognostic tests that measure mortality risk in patients with pulmonary arterial hypertension (PAH) are strongly associated with survival in those who are receiving a parenteral prostanoid, according to a recent study.
Patients with no lower-risk findings or at least two higher-risk findings had the worst outcomes, reported lead author Sonja Bartolome, MD, of the University of Texas Southwestern Medical Center in Dallas, and her colleagues.
Prostanoids are the most effective therapy for advanced PAH. However, patients may respond inadequately, so guidelines recommend lung transplant evaluation 3 months after starting a prostanoid.
The current study relied upon the 2015 European Society of Cardiology and European Respiratory Society (ESC/ERS) consensus guidelines. The guidelines recommend several tests to determine adequate response to therapy, including invasive hemodynamic measures, brain natriuretic peptide (BNP) level, N-terminal BNP (NT-proBNP) level, 6-minute walk distance (6MWD), and functional class (FC). Results of these tests are sorted into three hazard ratios for mortality: lower, intermediate, or higher risk.
It is commonly accepted that these risk categories can predict survival. For example, a patient with several higher-risk results and no lower-risk results would have a poor prognosis. However, the reliability of this method is poorly studied.
“In practicality the definition of an ‘inadequate response’ remains nebulous given that data on prognostic markers in patients on advanced therapy is limited,” the authors wrote. Their report was published in Chest®. “We therefore sought to evaluate whether consensus guidelines recommended prognostic measures associate with survival free from transplant in PAH patients initiating parenteral prostanoids.”
The retrospective study involved 195 patients with group 1 PAH at multiple treatment centers who received a parenteral prostanoid between 2007 and 2016. Diagnosis relied upon CT angiography, ventilation-perfusion scan, pulmonary function testing, cardiac catheterization, or echocardiogram. Eligible diagnoses were idiopathic PAH (n = 111), heritable PAH (n = 9), and PAH associated with connective tissue disease (n = 61), congenital heart disease (n = 12), and HIV (n = 2).
Patients received either IV epoprostenol (n = 132), SC treprostinil (n = 38), or IV treprostinil (n = 25). Routine prognostic testing was done prior to prostanoid therapy, and again at least 90 days later (with right heart catheterization). The investigators then analyzed the data for associations between test outcomes and survival.
Results showed that survival rates at 1, 2, and 3 years were 84%, 77%, and 67%, respectively. All major prognostic measures improved after patients started a prostanoid. Better SVO2, BNP, NT-proBNP, 6MWD, and FC were associated with survival, but cardiac index (CI) was not. Survival was least likely in patients who had at least two higher-risk measures or no lower-risk measures; of these patients, less than 50% were alive after 2 years.
“These findings are likely broadly applicable to PAH patients being treated with parenteral prostanoids,” the authors wrote, citing the fact that all patients in the study were newly started on either of the two parenteral prostanoids currently available in the United States (including patients who were treatment naive as well as those transitioning to parenteral therapy) and the study involved patients with multiple PAH subtypes. They also noted that 97% of patients were receiving combination therapy at first follow-up, “reflecting the more frequent use of combinations of medications in the modern era.”
However, not all of the prognostic measures were reliable, particularly CI.
Although CI is used as a major determinant for lung transplant, the authors noted that the lack of association between CI and survival suggests that “the strength and usefulness of some individual prognostic measures may differ for prostanoid-treated PAH patients.”
Some of the authors disclosed financial ties to United Therapeutics, which markets treprostinil for infusion (Remodulin), and Actelion, which markets epoprostenol for injection (Veletri), as well as other pharmaceutical companies.
Source: Bartolome S et al. Chest. 2018 Sep 1. doi: 10.1016/j.chest.2018.03.050
The results of guideline-recommended prognostic tests that measure mortality risk in patients with pulmonary arterial hypertension (PAH) are strongly associated with survival in those who are receiving a parenteral prostanoid, according to a recent study.
Patients with no lower-risk findings or at least two higher-risk findings had the worst outcomes, reported lead author Sonja Bartolome, MD, of the University of Texas Southwestern Medical Center in Dallas, and her colleagues.
Prostanoids are the most effective therapy for advanced PAH. However, patients may respond inadequately, so guidelines recommend lung transplant evaluation 3 months after starting a prostanoid.
The current study relied upon the 2015 European Society of Cardiology and European Respiratory Society (ESC/ERS) consensus guidelines. The guidelines recommend several tests to determine adequate response to therapy, including invasive hemodynamic measures, brain natriuretic peptide (BNP) level, N-terminal BNP (NT-proBNP) level, 6-minute walk distance (6MWD), and functional class (FC). Results of these tests are sorted into three hazard ratios for mortality: lower, intermediate, or higher risk.
It is commonly accepted that these risk categories can predict survival. For example, a patient with several higher-risk results and no lower-risk results would have a poor prognosis. However, the reliability of this method is poorly studied.
“In practicality the definition of an ‘inadequate response’ remains nebulous given that data on prognostic markers in patients on advanced therapy is limited,” the authors wrote. Their report was published in Chest®. “We therefore sought to evaluate whether consensus guidelines recommended prognostic measures associate with survival free from transplant in PAH patients initiating parenteral prostanoids.”
The retrospective study involved 195 patients with group 1 PAH at multiple treatment centers who received a parenteral prostanoid between 2007 and 2016. Diagnosis relied upon CT angiography, ventilation-perfusion scan, pulmonary function testing, cardiac catheterization, or echocardiogram. Eligible diagnoses were idiopathic PAH (n = 111), heritable PAH (n = 9), and PAH associated with connective tissue disease (n = 61), congenital heart disease (n = 12), and HIV (n = 2).
Patients received either IV epoprostenol (n = 132), SC treprostinil (n = 38), or IV treprostinil (n = 25). Routine prognostic testing was done prior to prostanoid therapy, and again at least 90 days later (with right heart catheterization). The investigators then analyzed the data for associations between test outcomes and survival.
Results showed that survival rates at 1, 2, and 3 years were 84%, 77%, and 67%, respectively. All major prognostic measures improved after patients started a prostanoid. Better SVO2, BNP, NT-proBNP, 6MWD, and FC were associated with survival, but cardiac index (CI) was not. Survival was least likely in patients who had at least two higher-risk measures or no lower-risk measures; of these patients, less than 50% were alive after 2 years.
“These findings are likely broadly applicable to PAH patients being treated with parenteral prostanoids,” the authors wrote, citing the fact that all patients in the study were newly started on either of the two parenteral prostanoids currently available in the United States (including patients who were treatment naive as well as those transitioning to parenteral therapy) and the study involved patients with multiple PAH subtypes. They also noted that 97% of patients were receiving combination therapy at first follow-up, “reflecting the more frequent use of combinations of medications in the modern era.”
However, not all of the prognostic measures were reliable, particularly CI.
Although CI is used as a major determinant for lung transplant, the authors noted that the lack of association between CI and survival suggests that “the strength and usefulness of some individual prognostic measures may differ for prostanoid-treated PAH patients.”
Some of the authors disclosed financial ties to United Therapeutics, which markets treprostinil for infusion (Remodulin), and Actelion, which markets epoprostenol for injection (Veletri), as well as other pharmaceutical companies.
Source: Bartolome S et al. Chest. 2018 Sep 1. doi: 10.1016/j.chest.2018.03.050
The results of guideline-recommended prognostic tests that measure mortality risk in patients with pulmonary arterial hypertension (PAH) are strongly associated with survival in those who are receiving a parenteral prostanoid, according to a recent study.
Patients with no lower-risk findings or at least two higher-risk findings had the worst outcomes, reported lead author Sonja Bartolome, MD, of the University of Texas Southwestern Medical Center in Dallas, and her colleagues.
Prostanoids are the most effective therapy for advanced PAH. However, patients may respond inadequately, so guidelines recommend lung transplant evaluation 3 months after starting a prostanoid.
The current study relied upon the 2015 European Society of Cardiology and European Respiratory Society (ESC/ERS) consensus guidelines. The guidelines recommend several tests to determine adequate response to therapy, including invasive hemodynamic measures, brain natriuretic peptide (BNP) level, N-terminal BNP (NT-proBNP) level, 6-minute walk distance (6MWD), and functional class (FC). Results of these tests are sorted into three hazard ratios for mortality: lower, intermediate, or higher risk.
It is commonly accepted that these risk categories can predict survival. For example, a patient with several higher-risk results and no lower-risk results would have a poor prognosis. However, the reliability of this method is poorly studied.
“In practicality the definition of an ‘inadequate response’ remains nebulous given that data on prognostic markers in patients on advanced therapy is limited,” the authors wrote. Their report was published in Chest®. “We therefore sought to evaluate whether consensus guidelines recommended prognostic measures associate with survival free from transplant in PAH patients initiating parenteral prostanoids.”
The retrospective study involved 195 patients with group 1 PAH at multiple treatment centers who received a parenteral prostanoid between 2007 and 2016. Diagnosis relied upon CT angiography, ventilation-perfusion scan, pulmonary function testing, cardiac catheterization, or echocardiogram. Eligible diagnoses were idiopathic PAH (n = 111), heritable PAH (n = 9), and PAH associated with connective tissue disease (n = 61), congenital heart disease (n = 12), and HIV (n = 2).
Patients received either IV epoprostenol (n = 132), SC treprostinil (n = 38), or IV treprostinil (n = 25). Routine prognostic testing was done prior to prostanoid therapy, and again at least 90 days later (with right heart catheterization). The investigators then analyzed the data for associations between test outcomes and survival.
Results showed that survival rates at 1, 2, and 3 years were 84%, 77%, and 67%, respectively. All major prognostic measures improved after patients started a prostanoid. Better SVO2, BNP, NT-proBNP, 6MWD, and FC were associated with survival, but cardiac index (CI) was not. Survival was least likely in patients who had at least two higher-risk measures or no lower-risk measures; of these patients, less than 50% were alive after 2 years.
“These findings are likely broadly applicable to PAH patients being treated with parenteral prostanoids,” the authors wrote, citing the fact that all patients in the study were newly started on either of the two parenteral prostanoids currently available in the United States (including patients who were treatment naive as well as those transitioning to parenteral therapy) and the study involved patients with multiple PAH subtypes. They also noted that 97% of patients were receiving combination therapy at first follow-up, “reflecting the more frequent use of combinations of medications in the modern era.”
However, not all of the prognostic measures were reliable, particularly CI.
Although CI is used as a major determinant for lung transplant, the authors noted that the lack of association between CI and survival suggests that “the strength and usefulness of some individual prognostic measures may differ for prostanoid-treated PAH patients.”
Some of the authors disclosed financial ties to United Therapeutics, which markets treprostinil for infusion (Remodulin), and Actelion, which markets epoprostenol for injection (Veletri), as well as other pharmaceutical companies.
Source: Bartolome S et al. Chest. 2018 Sep 1. doi: 10.1016/j.chest.2018.03.050
FROM CHEST®
Key clinical point: Survival in patients with pulmonary arterial hypertension (PAH) who are receiving a parenteral prostanoid is closely associated with the results of most guideline-recommended prognostic tests that measure mortality risk.
Major finding:
Study details: A retrospective study of 195 patients with PAH at multiple treatment centers who received a parenteral prostanoid between 2007 and 2016.
Disclosures: Some of the authors disclosed financial ties to United Therapeutics, which markets treprostinil for infusion (Remodulin), and Actelion, which markets epoprostenol for injection (Veletri), as well as other pharmaceutical companies.
Source: Bartolome S et al. Chest. 2018 Sep 1. doi: 10.1016/j.chest.2018.03.050.
Most in-hospital pneumonia deaths may not be preventable
Most in-hospital deaths from community-acquired pneumonia are not preventable with current medical therapy, according to an analysis of deaths at five U.S. hospitals with expertise in pneumonia care.
Adults who are hospitalized with community-acquired pneumonia (CAP) are at high risk for short-term mortality but it is unclear whether an improvement in care could lower this risk, noted the study authors led by Grant W. Waterer, MBBS, PhD, of Northwestern University, Chicago.
“Understanding the circumstances in which CAP patients die could facilitate improvements in the management of CAP by enabling future improvement efforts to focus on common preventable causes of death,” they wrote. Their report was published in CHEST®.
They therefore performed a secondary analysis of the Etiology of Pneumonia in the Community (EPIC) study involving adults hospitalized with CAP between January 2010 and June 2012 across five tertiary-care hospitals in the United States.
The clinical characteristics of patients who died in the hospital were compared with those of patients who survived to hospital discharge. Chronic heart failure, chronic obstructive pulmonary disease, coronary artery disease, chronic liver disease, cerebrovascular disease, cancer (excluding skin cancer), and diabetes were considered as severe chronic comorbidities based on their association with increased mortality and ICU admission in CAP severity scores.
Deaths caused by septic shock, respiratory failure, multisystem organ failure, cardiopulmonary arrest prior to stabilization of CAP, and endocarditis, were considered to be directly related to CAP.
Conversely, causes of death indirectly related to CAP included acute cardiovascular disease, stroke, acute renal failure, and secondary infections developed after hospitalization. Deaths caused by cancer, cirrhosis, and chronic neurologic conditions were considered unrelated to CAP.
Medical notes were assessed to determine whether the patient received management consistent with current recommendations; for example, antibiotics consistent with guidelines from the Infectious Diseases Society of America.
End-of-life limitations in care, such as patient/family decision not to proceed with full medical treatment, also were considered by the research team.
Results showed that among the 2,320 patients with radiographically confirmed CAP, 52 died during initial hospitalization, 33 of whom were aged 65 years or older, and 32 of whom had two or more chronic comorbidities.
Most of the in-hospital deaths occurred early in the hospitalization: 35 within the first 10 days of admission, and 5 after 30 days in hospital.
CAP was judged by an expert physician review panel to be the direct cause of death in 27 of the patients, 10 with CAP having an indirect role with major contribution, 9 with CAP having an indirect role with minor contribution, and 6 with CAP having no role in death.
Do-not-resuscitate orders were present at the time of death for 21 of the patients.
Forty-five of the patients were admitted to an ICU, with 37 dying in the ICU. The eight patients who died on the ward after transfer out of the ICU had end-of-life limitations of care in place.
The researchers noted that the number of patients dying in the ICU was greater in the United States, possibly because in Europe fewer patients are admitted to an ICU.
“This discrepancy likely reflects cultural differences between the U.S. and Europe in the role of intensive care for patients with advanced age and/or advanced comorbid conditions,” they noted.
Overall, the physician review panel identified nine patients who had a lapse in quality of in-hospital CAP care, with four of the deaths potentially linked to this lapse in care.
However, two of the patients had end-of-life limitations of care in place, which according to the authors meant that “only two patients undergoing full medical treatment without end-of-life limitations of care had an identified lapse in quality of in-hospital pneumonia care potentially contributing to in-hospital death, including one with a delay in antibiotics for over an hour in the presence of shock and one with initial antibiotics not consistent with IDSA/ATS guidelines.”
The research team concluded that most in-hospital deaths among adult patients admitted with CAP in their study would not have been preventable with higher quality in-hospital pneumonia care.
“Many of the in-hospital deaths among patients admitted with CAP occurred in older patients with severe comorbidities and end-of-life limitations in care,” they noted.
They said the influence of end-of-life limitations on care short of full palliation was an important finding, with all patients who died outside the ICU having end-of-life limitations in care.
“Current diagnostic related group (DRG) and international classification of diseases (ICD) coding systems do not have the necessary nuances to capture these limitations of care, yet they are clearly important factors in determining whether patients experience in-hospital death,” they added.
Dr. Waterer reported no conflicts. Two coauthors reported potential conflicts of interest in relation to consulting fees from several pharmaceutical companies.
SOURCE: Waterer G. et al. CHEST 2018;154(3):628-35. doi: 10.1016/j.chest.2018.05.021.
Most in-hospital deaths from community-acquired pneumonia are not preventable with current medical therapy, according to an analysis of deaths at five U.S. hospitals with expertise in pneumonia care.
Adults who are hospitalized with community-acquired pneumonia (CAP) are at high risk for short-term mortality but it is unclear whether an improvement in care could lower this risk, noted the study authors led by Grant W. Waterer, MBBS, PhD, of Northwestern University, Chicago.
“Understanding the circumstances in which CAP patients die could facilitate improvements in the management of CAP by enabling future improvement efforts to focus on common preventable causes of death,” they wrote. Their report was published in CHEST®.
They therefore performed a secondary analysis of the Etiology of Pneumonia in the Community (EPIC) study involving adults hospitalized with CAP between January 2010 and June 2012 across five tertiary-care hospitals in the United States.
The clinical characteristics of patients who died in the hospital were compared with those of patients who survived to hospital discharge. Chronic heart failure, chronic obstructive pulmonary disease, coronary artery disease, chronic liver disease, cerebrovascular disease, cancer (excluding skin cancer), and diabetes were considered as severe chronic comorbidities based on their association with increased mortality and ICU admission in CAP severity scores.
Deaths caused by septic shock, respiratory failure, multisystem organ failure, cardiopulmonary arrest prior to stabilization of CAP, and endocarditis, were considered to be directly related to CAP.
Conversely, causes of death indirectly related to CAP included acute cardiovascular disease, stroke, acute renal failure, and secondary infections developed after hospitalization. Deaths caused by cancer, cirrhosis, and chronic neurologic conditions were considered unrelated to CAP.
Medical notes were assessed to determine whether the patient received management consistent with current recommendations; for example, antibiotics consistent with guidelines from the Infectious Diseases Society of America.
End-of-life limitations in care, such as patient/family decision not to proceed with full medical treatment, also were considered by the research team.
Results showed that among the 2,320 patients with radiographically confirmed CAP, 52 died during initial hospitalization, 33 of whom were aged 65 years or older, and 32 of whom had two or more chronic comorbidities.
Most of the in-hospital deaths occurred early in the hospitalization: 35 within the first 10 days of admission, and 5 after 30 days in hospital.
CAP was judged by an expert physician review panel to be the direct cause of death in 27 of the patients, 10 with CAP having an indirect role with major contribution, 9 with CAP having an indirect role with minor contribution, and 6 with CAP having no role in death.
Do-not-resuscitate orders were present at the time of death for 21 of the patients.
Forty-five of the patients were admitted to an ICU, with 37 dying in the ICU. The eight patients who died on the ward after transfer out of the ICU had end-of-life limitations of care in place.
The researchers noted that the number of patients dying in the ICU was greater in the United States, possibly because in Europe fewer patients are admitted to an ICU.
“This discrepancy likely reflects cultural differences between the U.S. and Europe in the role of intensive care for patients with advanced age and/or advanced comorbid conditions,” they noted.
Overall, the physician review panel identified nine patients who had a lapse in quality of in-hospital CAP care, with four of the deaths potentially linked to this lapse in care.
However, two of the patients had end-of-life limitations of care in place, which according to the authors meant that “only two patients undergoing full medical treatment without end-of-life limitations of care had an identified lapse in quality of in-hospital pneumonia care potentially contributing to in-hospital death, including one with a delay in antibiotics for over an hour in the presence of shock and one with initial antibiotics not consistent with IDSA/ATS guidelines.”
The research team concluded that most in-hospital deaths among adult patients admitted with CAP in their study would not have been preventable with higher quality in-hospital pneumonia care.
“Many of the in-hospital deaths among patients admitted with CAP occurred in older patients with severe comorbidities and end-of-life limitations in care,” they noted.
They said the influence of end-of-life limitations on care short of full palliation was an important finding, with all patients who died outside the ICU having end-of-life limitations in care.
“Current diagnostic related group (DRG) and international classification of diseases (ICD) coding systems do not have the necessary nuances to capture these limitations of care, yet they are clearly important factors in determining whether patients experience in-hospital death,” they added.
Dr. Waterer reported no conflicts. Two coauthors reported potential conflicts of interest in relation to consulting fees from several pharmaceutical companies.
SOURCE: Waterer G. et al. CHEST 2018;154(3):628-35. doi: 10.1016/j.chest.2018.05.021.
Most in-hospital deaths from community-acquired pneumonia are not preventable with current medical therapy, according to an analysis of deaths at five U.S. hospitals with expertise in pneumonia care.
Adults who are hospitalized with community-acquired pneumonia (CAP) are at high risk for short-term mortality but it is unclear whether an improvement in care could lower this risk, noted the study authors led by Grant W. Waterer, MBBS, PhD, of Northwestern University, Chicago.
“Understanding the circumstances in which CAP patients die could facilitate improvements in the management of CAP by enabling future improvement efforts to focus on common preventable causes of death,” they wrote. Their report was published in CHEST®.
They therefore performed a secondary analysis of the Etiology of Pneumonia in the Community (EPIC) study involving adults hospitalized with CAP between January 2010 and June 2012 across five tertiary-care hospitals in the United States.
The clinical characteristics of patients who died in the hospital were compared with those of patients who survived to hospital discharge. Chronic heart failure, chronic obstructive pulmonary disease, coronary artery disease, chronic liver disease, cerebrovascular disease, cancer (excluding skin cancer), and diabetes were considered as severe chronic comorbidities based on their association with increased mortality and ICU admission in CAP severity scores.
Deaths caused by septic shock, respiratory failure, multisystem organ failure, cardiopulmonary arrest prior to stabilization of CAP, and endocarditis, were considered to be directly related to CAP.
Conversely, causes of death indirectly related to CAP included acute cardiovascular disease, stroke, acute renal failure, and secondary infections developed after hospitalization. Deaths caused by cancer, cirrhosis, and chronic neurologic conditions were considered unrelated to CAP.
Medical notes were assessed to determine whether the patient received management consistent with current recommendations; for example, antibiotics consistent with guidelines from the Infectious Diseases Society of America.
End-of-life limitations in care, such as patient/family decision not to proceed with full medical treatment, also were considered by the research team.
Results showed that among the 2,320 patients with radiographically confirmed CAP, 52 died during initial hospitalization, 33 of whom were aged 65 years or older, and 32 of whom had two or more chronic comorbidities.
Most of the in-hospital deaths occurred early in the hospitalization: 35 within the first 10 days of admission, and 5 after 30 days in hospital.
CAP was judged by an expert physician review panel to be the direct cause of death in 27 of the patients, 10 with CAP having an indirect role with major contribution, 9 with CAP having an indirect role with minor contribution, and 6 with CAP having no role in death.
Do-not-resuscitate orders were present at the time of death for 21 of the patients.
Forty-five of the patients were admitted to an ICU, with 37 dying in the ICU. The eight patients who died on the ward after transfer out of the ICU had end-of-life limitations of care in place.
The researchers noted that the number of patients dying in the ICU was greater in the United States, possibly because in Europe fewer patients are admitted to an ICU.
“This discrepancy likely reflects cultural differences between the U.S. and Europe in the role of intensive care for patients with advanced age and/or advanced comorbid conditions,” they noted.
Overall, the physician review panel identified nine patients who had a lapse in quality of in-hospital CAP care, with four of the deaths potentially linked to this lapse in care.
However, two of the patients had end-of-life limitations of care in place, which according to the authors meant that “only two patients undergoing full medical treatment without end-of-life limitations of care had an identified lapse in quality of in-hospital pneumonia care potentially contributing to in-hospital death, including one with a delay in antibiotics for over an hour in the presence of shock and one with initial antibiotics not consistent with IDSA/ATS guidelines.”
The research team concluded that most in-hospital deaths among adult patients admitted with CAP in their study would not have been preventable with higher quality in-hospital pneumonia care.
“Many of the in-hospital deaths among patients admitted with CAP occurred in older patients with severe comorbidities and end-of-life limitations in care,” they noted.
They said the influence of end-of-life limitations on care short of full palliation was an important finding, with all patients who died outside the ICU having end-of-life limitations in care.
“Current diagnostic related group (DRG) and international classification of diseases (ICD) coding systems do not have the necessary nuances to capture these limitations of care, yet they are clearly important factors in determining whether patients experience in-hospital death,” they added.
Dr. Waterer reported no conflicts. Two coauthors reported potential conflicts of interest in relation to consulting fees from several pharmaceutical companies.
SOURCE: Waterer G. et al. CHEST 2018;154(3):628-35. doi: 10.1016/j.chest.2018.05.021.
FROM CHEST
Key clinical point: Most in-hospital deaths from community-acquired pneumonia are not preventable with current medical therapy.
Major finding: Two out of 52 patients who died in-hospital from community-acquired pneumonia (CAP) who were undergoing full medical treatment without end-of-life limitations of care had an identified lapse in quality of in-hospital pneumonia care that potentially contributed to their death.
Study details: A secondary analysis of the prospective multicenter Etiology of Pneumonia in the Community (EPIC) study involving 2,320 adults with radiographically confirmed CAP.
Disclosures: Dr. Waterer reported no conflicts. Two coauthors reported potential conflicts of interest in relation to consulting fees from several pharmaceutical companies.
Source: Waterer G. et al. CHEST 2018;154(3):628-35.
Short sleep linked to elevated blood pressure
CHICAGO – Consider 24-hour ambulatory blood pressure monitoring when patients complain about not getting enough sleep. You might catch hypertension early, according to researchers from the University of Pennsylvania, Philadelphia, and elsewhere.
They found a less than 7 hours a night and a mean in the study of 5.5 hours. Every 2-2.5 minutes of lost sleep was associated with an increase of 1 mm Hg in 24-hour mean systolic blood pressure and a increase of 1 beat per minute in heart rate.
The relationship was independent of office BP, nocturnal dipping status, and BP variability. It held in both the obese and nonobese, and in patients with and without obstructive sleep apnea (OSA). However, the relationship was found only among subjects who were not on antihypertensive medications.
“Adults with shorter sleep duration may benefit from screening with 24-hour ambulatory BP monitoring to promote earlier detection of hypertension and potentially mitigate the” the risk of cardiovascular disease. “This may be particularly important in screening for masked hypertension,” meaning normal pressures in the office, but elevated pressures at home, said investigators led by Jordana Cohen, MD, of the department of medicine at the University of Pennsylvania in a presentation at the joint scientific sessions of AHA Council on Hypertension, AHA Council on Kidney in Cardiovascular Disease, and American Society of Hypertension.
Dr. Cohen suggested that perhaps the sympathetic and endothelial derangements that drive hypertension in OSA also affect people with insufficient sleep. It may be that the normal morning surge in blood pressure persists longer into the day, she suggested. The investigative team analyzed data from two studies. The first, LIMBS (Lifestyle Modification in BP Lowering Study), was a phase 2 trial assessing yoga for blood pressure lowering. It was conducted in West Philadelphia and excluded people with diabetes, hypertension, OSA, and kidney or cardiovascular disease. The new analysis included 66 LIMBS subjects who had 24-hour blood pressure monitoring and kept sleep diaries to record their sleep duration (J Clin Hypertens (Greenwich). 2016 Aug;18[8]:809-16).
The team also analyzed 153 subjects from the PISA (Penn Icelandic Sleep Apnea) cohort, an ongoing project assessing continuous positive airway pressure for OSA, among other things. PISA includes patients with OSA, diabetes, hypertension, and kidney or cardiovascular disease. Sleep duration in the 153 subjects was again self-reported, but corroborated by actigraphy (J Sleep Res. 2015 Jun;24[3]:328-38).
The new findings were driven mostly by higher daytime systolic BP among short sleepers in LIMBS, and higher systolic pressures during both day and night among short sleepers in PISA, compared with subjects who slept at least 7 hours, and a mean of 8.5 hours, with napping included in overall sleep duration assessment.
In LIMBS, the mean 24-hour systolic BP was 12.7 mm Hg higher and the average heart rate 8 bpm faster among short sleepers; in PISA, the mean 24-hour systolic BP was 4.7 mm Hg higher and the heart rate 2 bpm faster.
Every 2.57 minutes of sleep lost in LIMBS and every 1.99 minute of lost sleep in PISA was associated with a 1–mm Hg gain in mean systolic BP and about a 1-bpm increase in heart rate. The findings were statistically significant and adjusted for age, race, body mass index, nocturnal dipping status, and office systolic blood pressure.
Baseline characteristics were generally well matched between short and long sleepers in both studies. However, while mean office systolic BP in LIMBS was the same in both sleep groups at about 139 mm Hg, the mean office systolic BP among long sleepers in PISA was 130 mm Hg versus 136 mm Hg among short sleepers, a significant difference.
It’s unclear why some people slept less, Dr. Cohen said, and the use of sleeping pills wasn’t considered in the analysis. Patients were an average of about 50 years old, with a body mass index of about 30 mg/m2. The same model of 24-hour blood pressure monitor was used in both studies.
The work was funded by the National Institutes of Health. The investigators had no disclosures.
CHICAGO – Consider 24-hour ambulatory blood pressure monitoring when patients complain about not getting enough sleep. You might catch hypertension early, according to researchers from the University of Pennsylvania, Philadelphia, and elsewhere.
They found a less than 7 hours a night and a mean in the study of 5.5 hours. Every 2-2.5 minutes of lost sleep was associated with an increase of 1 mm Hg in 24-hour mean systolic blood pressure and a increase of 1 beat per minute in heart rate.
The relationship was independent of office BP, nocturnal dipping status, and BP variability. It held in both the obese and nonobese, and in patients with and without obstructive sleep apnea (OSA). However, the relationship was found only among subjects who were not on antihypertensive medications.
“Adults with shorter sleep duration may benefit from screening with 24-hour ambulatory BP monitoring to promote earlier detection of hypertension and potentially mitigate the” the risk of cardiovascular disease. “This may be particularly important in screening for masked hypertension,” meaning normal pressures in the office, but elevated pressures at home, said investigators led by Jordana Cohen, MD, of the department of medicine at the University of Pennsylvania in a presentation at the joint scientific sessions of AHA Council on Hypertension, AHA Council on Kidney in Cardiovascular Disease, and American Society of Hypertension.
Dr. Cohen suggested that perhaps the sympathetic and endothelial derangements that drive hypertension in OSA also affect people with insufficient sleep. It may be that the normal morning surge in blood pressure persists longer into the day, she suggested. The investigative team analyzed data from two studies. The first, LIMBS (Lifestyle Modification in BP Lowering Study), was a phase 2 trial assessing yoga for blood pressure lowering. It was conducted in West Philadelphia and excluded people with diabetes, hypertension, OSA, and kidney or cardiovascular disease. The new analysis included 66 LIMBS subjects who had 24-hour blood pressure monitoring and kept sleep diaries to record their sleep duration (J Clin Hypertens (Greenwich). 2016 Aug;18[8]:809-16).
The team also analyzed 153 subjects from the PISA (Penn Icelandic Sleep Apnea) cohort, an ongoing project assessing continuous positive airway pressure for OSA, among other things. PISA includes patients with OSA, diabetes, hypertension, and kidney or cardiovascular disease. Sleep duration in the 153 subjects was again self-reported, but corroborated by actigraphy (J Sleep Res. 2015 Jun;24[3]:328-38).
The new findings were driven mostly by higher daytime systolic BP among short sleepers in LIMBS, and higher systolic pressures during both day and night among short sleepers in PISA, compared with subjects who slept at least 7 hours, and a mean of 8.5 hours, with napping included in overall sleep duration assessment.
In LIMBS, the mean 24-hour systolic BP was 12.7 mm Hg higher and the average heart rate 8 bpm faster among short sleepers; in PISA, the mean 24-hour systolic BP was 4.7 mm Hg higher and the heart rate 2 bpm faster.
Every 2.57 minutes of sleep lost in LIMBS and every 1.99 minute of lost sleep in PISA was associated with a 1–mm Hg gain in mean systolic BP and about a 1-bpm increase in heart rate. The findings were statistically significant and adjusted for age, race, body mass index, nocturnal dipping status, and office systolic blood pressure.
Baseline characteristics were generally well matched between short and long sleepers in both studies. However, while mean office systolic BP in LIMBS was the same in both sleep groups at about 139 mm Hg, the mean office systolic BP among long sleepers in PISA was 130 mm Hg versus 136 mm Hg among short sleepers, a significant difference.
It’s unclear why some people slept less, Dr. Cohen said, and the use of sleeping pills wasn’t considered in the analysis. Patients were an average of about 50 years old, with a body mass index of about 30 mg/m2. The same model of 24-hour blood pressure monitor was used in both studies.
The work was funded by the National Institutes of Health. The investigators had no disclosures.
CHICAGO – Consider 24-hour ambulatory blood pressure monitoring when patients complain about not getting enough sleep. You might catch hypertension early, according to researchers from the University of Pennsylvania, Philadelphia, and elsewhere.
They found a less than 7 hours a night and a mean in the study of 5.5 hours. Every 2-2.5 minutes of lost sleep was associated with an increase of 1 mm Hg in 24-hour mean systolic blood pressure and a increase of 1 beat per minute in heart rate.
The relationship was independent of office BP, nocturnal dipping status, and BP variability. It held in both the obese and nonobese, and in patients with and without obstructive sleep apnea (OSA). However, the relationship was found only among subjects who were not on antihypertensive medications.
“Adults with shorter sleep duration may benefit from screening with 24-hour ambulatory BP monitoring to promote earlier detection of hypertension and potentially mitigate the” the risk of cardiovascular disease. “This may be particularly important in screening for masked hypertension,” meaning normal pressures in the office, but elevated pressures at home, said investigators led by Jordana Cohen, MD, of the department of medicine at the University of Pennsylvania in a presentation at the joint scientific sessions of AHA Council on Hypertension, AHA Council on Kidney in Cardiovascular Disease, and American Society of Hypertension.
Dr. Cohen suggested that perhaps the sympathetic and endothelial derangements that drive hypertension in OSA also affect people with insufficient sleep. It may be that the normal morning surge in blood pressure persists longer into the day, she suggested. The investigative team analyzed data from two studies. The first, LIMBS (Lifestyle Modification in BP Lowering Study), was a phase 2 trial assessing yoga for blood pressure lowering. It was conducted in West Philadelphia and excluded people with diabetes, hypertension, OSA, and kidney or cardiovascular disease. The new analysis included 66 LIMBS subjects who had 24-hour blood pressure monitoring and kept sleep diaries to record their sleep duration (J Clin Hypertens (Greenwich). 2016 Aug;18[8]:809-16).
The team also analyzed 153 subjects from the PISA (Penn Icelandic Sleep Apnea) cohort, an ongoing project assessing continuous positive airway pressure for OSA, among other things. PISA includes patients with OSA, diabetes, hypertension, and kidney or cardiovascular disease. Sleep duration in the 153 subjects was again self-reported, but corroborated by actigraphy (J Sleep Res. 2015 Jun;24[3]:328-38).
The new findings were driven mostly by higher daytime systolic BP among short sleepers in LIMBS, and higher systolic pressures during both day and night among short sleepers in PISA, compared with subjects who slept at least 7 hours, and a mean of 8.5 hours, with napping included in overall sleep duration assessment.
In LIMBS, the mean 24-hour systolic BP was 12.7 mm Hg higher and the average heart rate 8 bpm faster among short sleepers; in PISA, the mean 24-hour systolic BP was 4.7 mm Hg higher and the heart rate 2 bpm faster.
Every 2.57 minutes of sleep lost in LIMBS and every 1.99 minute of lost sleep in PISA was associated with a 1–mm Hg gain in mean systolic BP and about a 1-bpm increase in heart rate. The findings were statistically significant and adjusted for age, race, body mass index, nocturnal dipping status, and office systolic blood pressure.
Baseline characteristics were generally well matched between short and long sleepers in both studies. However, while mean office systolic BP in LIMBS was the same in both sleep groups at about 139 mm Hg, the mean office systolic BP among long sleepers in PISA was 130 mm Hg versus 136 mm Hg among short sleepers, a significant difference.
It’s unclear why some people slept less, Dr. Cohen said, and the use of sleeping pills wasn’t considered in the analysis. Patients were an average of about 50 years old, with a body mass index of about 30 mg/m2. The same model of 24-hour blood pressure monitor was used in both studies.
The work was funded by the National Institutes of Health. The investigators had no disclosures.
REPORTING FROM JOINT HYPERTENSION 2018
Key clinical point: Ambulatory blood pressure monitoring in patients complaining about lack of sleep could detect hypertension.
Major finding: Every 2-2.5 minutes of lost sleep was associated with a 1–mm Hg increase in 24-hour mean systolic blood pressure and a 1-bpm increase in heart rate.
Study details: Post-hoc review of 219 patients in two trials
Disclosures: The work was funded by the National Institutes of Health. The investigators didn’t have any disclosures.
UN aims to eradicate TB by 2030
A concerted a lethal disease affecting one-quarter of the world’s population by the year 2030.
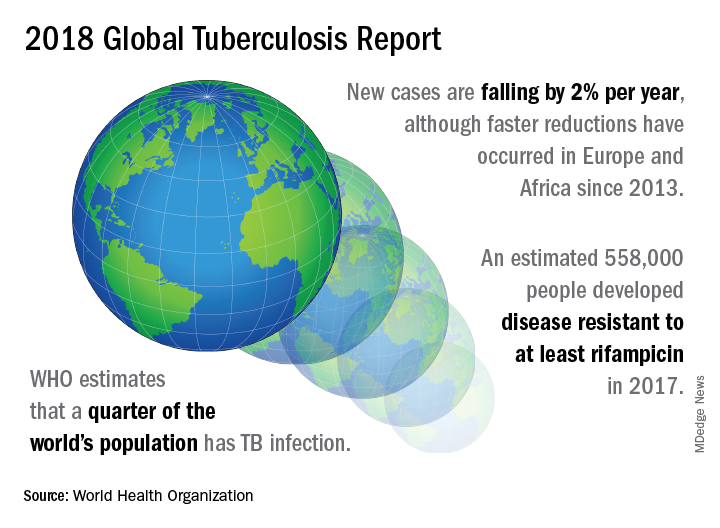
On September 26 the United Nations General Assembly will convene a high-level meeting of global stakeholders to solidify the eradication plan, addressing the global crisis of tuberculosis (TB), the world’s most deadly infectious disease.
“We must seize the moment,” said Tereza Kasaeva, MD, director of the World Health Organization’s global TB program, speaking at a telebriefing and press conference accompanying the release of the World Health Organization’s annual global tuberculosis report. “It’s unacceptable in the 21st century that millions lose their lives to this preventable and curable disease.”
TB caused 1.6 million deaths globally in 2017, and the World Health Organization (WHO) estimates that of the 10 million new cases of TB last year, 558,000 are multi-drug resistant (MDR) infections.
Though death rates and new cases are falling globally each year, significantly more resources are needed to boost access to preventive treatment for latent TB infection; “Most people needing it are not yet accessing care,” according to the press briefing accompanying the report.
A review and commentary on TB incubation and latency published in BMJ (2018;362:k2738 doi: 10.1136/bmj.k2738; e-pub 23 Aug 2018) has called into question the focus preventive treatment of latent cases at the expense of reaching those most likely to die from TB (e.g., HIV patients, children of individuals living with active TB). The authors state that “latent” TB is identified by indirect evidence of present or past infection with Mycobacterium tuberculosis as inferred by a detectable adaptive immune response to M tuberculosis antigens. Active TB infection is overwhelmingly the result of a primary infection and almost always occurs within two years.
In order to meet the ambitious goal of TB eradication by the year 2030, treatment coverage must rise to 90% globally from the current 64%, according to the report.
Progress in southern Africa and in the Russian Federation, where efforts have led to a 30% reduction in TB mortality and a decrease in incidence of 5% per year, show that steep reductions in TB are possible when resources are brought to bear on the problem, said Dr. Kasaeva. “We should acknowledge that actions in some countries and regions show that progress can accelerate,” she said. Still, she noted, “Four thousand lives per day are lost to TB. Tuberculosis is the leading killer of people living with HIV, and the major cause of deaths related to antimicrobial resistance” at a global level.
Two thirds of all TB cases occur in eight countries, with India, China, and Indonesia leading this group. About half of the cases of MDR TB occur in India, China, and Russia, said Dr. Kasaeva, and globally only one in four individuals with MDR TB who need access to treatment have received it. “We need to urgently tackle the multidrug resistant TB public health crisis,” she said.
Major impediments to successful public health efforts against TB are underdiagnosis and underreporting: It is estimated that 3.6 million of 2017’s 10 million new cases were not officially recorded or reported. Countries where these problems are most serious include India, Indonesia, and Nigeria. Fewer than half of the children with TB are reported globally, according to the report.
People living with HIV/AIDS who are also infected with TB number nearly 1,000,000, but only about half of these were officially reported in 2017.
In terms of prevention priorities, WHO has recommended targeting treatment of latent TB in two groups: People living with HIV/AIDS, and children under the age of 5 years who live in households with TB-infected individuals.
“To enable these actions,” said Dr. Kasaeva, “we need strengthened commitments not just for TB care, but for overall health services. So the aim for universal coverage is real.” Underreporting is particularly prevalent in lower income countries with large unregulated private sectors, she said, though India and Indonesia have taken corrective steps to increase reporting.
A meaningful global initiative will not come cheap: The current annual shortfall in funding for TB prevention, diagnosis, and treatment is about $3.5 billion. By the year 2022, the gap between funding and what’s needed to stay on track for the 2030 target will be over $6 billion, said Dr. Kasaeva.
The best use of increased resources for TB eradication will be in locally focused efforts, said Irene Koek, MD, the United States Agency for International Development’s deputy administrator for global health. “It is likely that each region requires a tailored response.” Further, “to improve quality of care we need to ensure that services are patient centered,” she said at the press conference.
To that end, Dr. Koek expects that at the upcoming high-level meeting, the United Nations member states will be called on to develop an open framework, with clear accountability for monitoring and reviewing progress. The road forward should “celebrate accomplishments and acknowledge shortcomings,” she said. Some recent studies have shown that treatment success rates above 80% for patients with MDR TB can be achieved.
“Lessons learned from these experiences should be documented and shared in order to replicate success globally,” said Dr. Koek.
The United States, said Dr. Koek, is the leading global investor in TB research and treatment. “We welcome increased partnerships, especially with countries with the highest burden, to end global suffering from this disease.”
Eric Goosby, MD, the United Nations special envoy on TB, used his speaking time to lend some perspective to the social framework around TB’s longtime lethality.
There are aspects of TB infection that differentiate it from HIV/AIDS, said Dr. Goosby, who has spent most of his clinical and public health career on HIV/AIDS treatment and prevention. In contrast to an infection that at present requires a lifetime of treatment, TB can ordinarily be treated in 6 months, making it an unpleasant episode that an individual may be eager to move past. Additionally, the fact that TB has had a “hold on the world since the time of the ancient Egyptians” may paradoxically have served to lessen urgency in research and treatment efforts, he noted.
Dr. Goosby also spoke of the stigma surrounding TB, whose sufferers are likely to be facing dire poverty, malnutrition, and other infectious disease burdens. Civil society concerned with TB, he said, has spoken up “for those without a voice, for those who have difficulty advocating for themselves.”
Dr. Kasaeva agreed, noting that TB “affects the poorest of the poor, which makes it extraordinarily difficult for activism to come from that population.”
However, others have spoken for those affected, said Dr. Goosby. “The TB civil society has put its heart and soul this last year into gathering political will from leaders around the world…. It’s not a passive effort; it involves a lot of work.” During the past year of concerted effort, he said, “All of us have known the difficulty of pushing a political leader up that learning curve.”
As the upcoming high-level meeting approaches, those who have been working on the effort can feel the momentum, said Dr. Goosby. Still, he noted, “While there’s a significant step forward, this is not the time for a victory dance. This is really the time for a reflection...Do we understand the burden in our respective countries, and has the response been adequate?”
The goal for the meeting is to have leaders “step up to commit, not for one day, or for one meeting, but for the duration of the effort,” said Dr. Goosby. “We must make sure that the words that we hear next week from our leaders translate into action...Next week the world will say, ‘No more. No longer. No one is immune to TB. Tuberculosis is preventable; tuberculosis is treatable; tuberculosis is curable.’”
The BMJ commentary, by Marcel A. Behr, MD, of McGill International TB Centre, Infectious Diseases and Immunity in Global Health Program, McGill University Health Centre Research Institute, and his colleagues, recommend caution when building a prevention strategy around treating many millions of individuals with “latent” TB. They wrote, “Immunoreactivity to TB does not necessarily indicate the presence of live bacteria, as reactivity can persist after infection has been cleared. Classifying two billion people with evidence of immunoreactivity as having latent TB infection may divert fundamental research and public health interventions away from transmissible active TB disease and newly infected people at highest risk of progression to disease.”
This story was updated on 09/24/2018
A concerted a lethal disease affecting one-quarter of the world’s population by the year 2030.

On September 26 the United Nations General Assembly will convene a high-level meeting of global stakeholders to solidify the eradication plan, addressing the global crisis of tuberculosis (TB), the world’s most deadly infectious disease.
“We must seize the moment,” said Tereza Kasaeva, MD, director of the World Health Organization’s global TB program, speaking at a telebriefing and press conference accompanying the release of the World Health Organization’s annual global tuberculosis report. “It’s unacceptable in the 21st century that millions lose their lives to this preventable and curable disease.”
TB caused 1.6 million deaths globally in 2017, and the World Health Organization (WHO) estimates that of the 10 million new cases of TB last year, 558,000 are multi-drug resistant (MDR) infections.
Though death rates and new cases are falling globally each year, significantly more resources are needed to boost access to preventive treatment for latent TB infection; “Most people needing it are not yet accessing care,” according to the press briefing accompanying the report.
A review and commentary on TB incubation and latency published in BMJ (2018;362:k2738 doi: 10.1136/bmj.k2738; e-pub 23 Aug 2018) has called into question the focus preventive treatment of latent cases at the expense of reaching those most likely to die from TB (e.g., HIV patients, children of individuals living with active TB). The authors state that “latent” TB is identified by indirect evidence of present or past infection with Mycobacterium tuberculosis as inferred by a detectable adaptive immune response to M tuberculosis antigens. Active TB infection is overwhelmingly the result of a primary infection and almost always occurs within two years.
In order to meet the ambitious goal of TB eradication by the year 2030, treatment coverage must rise to 90% globally from the current 64%, according to the report.
Progress in southern Africa and in the Russian Federation, where efforts have led to a 30% reduction in TB mortality and a decrease in incidence of 5% per year, show that steep reductions in TB are possible when resources are brought to bear on the problem, said Dr. Kasaeva. “We should acknowledge that actions in some countries and regions show that progress can accelerate,” she said. Still, she noted, “Four thousand lives per day are lost to TB. Tuberculosis is the leading killer of people living with HIV, and the major cause of deaths related to antimicrobial resistance” at a global level.
Two thirds of all TB cases occur in eight countries, with India, China, and Indonesia leading this group. About half of the cases of MDR TB occur in India, China, and Russia, said Dr. Kasaeva, and globally only one in four individuals with MDR TB who need access to treatment have received it. “We need to urgently tackle the multidrug resistant TB public health crisis,” she said.
Major impediments to successful public health efforts against TB are underdiagnosis and underreporting: It is estimated that 3.6 million of 2017’s 10 million new cases were not officially recorded or reported. Countries where these problems are most serious include India, Indonesia, and Nigeria. Fewer than half of the children with TB are reported globally, according to the report.
People living with HIV/AIDS who are also infected with TB number nearly 1,000,000, but only about half of these were officially reported in 2017.
In terms of prevention priorities, WHO has recommended targeting treatment of latent TB in two groups: People living with HIV/AIDS, and children under the age of 5 years who live in households with TB-infected individuals.
“To enable these actions,” said Dr. Kasaeva, “we need strengthened commitments not just for TB care, but for overall health services. So the aim for universal coverage is real.” Underreporting is particularly prevalent in lower income countries with large unregulated private sectors, she said, though India and Indonesia have taken corrective steps to increase reporting.
A meaningful global initiative will not come cheap: The current annual shortfall in funding for TB prevention, diagnosis, and treatment is about $3.5 billion. By the year 2022, the gap between funding and what’s needed to stay on track for the 2030 target will be over $6 billion, said Dr. Kasaeva.
The best use of increased resources for TB eradication will be in locally focused efforts, said Irene Koek, MD, the United States Agency for International Development’s deputy administrator for global health. “It is likely that each region requires a tailored response.” Further, “to improve quality of care we need to ensure that services are patient centered,” she said at the press conference.
To that end, Dr. Koek expects that at the upcoming high-level meeting, the United Nations member states will be called on to develop an open framework, with clear accountability for monitoring and reviewing progress. The road forward should “celebrate accomplishments and acknowledge shortcomings,” she said. Some recent studies have shown that treatment success rates above 80% for patients with MDR TB can be achieved.
“Lessons learned from these experiences should be documented and shared in order to replicate success globally,” said Dr. Koek.
The United States, said Dr. Koek, is the leading global investor in TB research and treatment. “We welcome increased partnerships, especially with countries with the highest burden, to end global suffering from this disease.”
Eric Goosby, MD, the United Nations special envoy on TB, used his speaking time to lend some perspective to the social framework around TB’s longtime lethality.
There are aspects of TB infection that differentiate it from HIV/AIDS, said Dr. Goosby, who has spent most of his clinical and public health career on HIV/AIDS treatment and prevention. In contrast to an infection that at present requires a lifetime of treatment, TB can ordinarily be treated in 6 months, making it an unpleasant episode that an individual may be eager to move past. Additionally, the fact that TB has had a “hold on the world since the time of the ancient Egyptians” may paradoxically have served to lessen urgency in research and treatment efforts, he noted.
Dr. Goosby also spoke of the stigma surrounding TB, whose sufferers are likely to be facing dire poverty, malnutrition, and other infectious disease burdens. Civil society concerned with TB, he said, has spoken up “for those without a voice, for those who have difficulty advocating for themselves.”
Dr. Kasaeva agreed, noting that TB “affects the poorest of the poor, which makes it extraordinarily difficult for activism to come from that population.”
However, others have spoken for those affected, said Dr. Goosby. “The TB civil society has put its heart and soul this last year into gathering political will from leaders around the world…. It’s not a passive effort; it involves a lot of work.” During the past year of concerted effort, he said, “All of us have known the difficulty of pushing a political leader up that learning curve.”
As the upcoming high-level meeting approaches, those who have been working on the effort can feel the momentum, said Dr. Goosby. Still, he noted, “While there’s a significant step forward, this is not the time for a victory dance. This is really the time for a reflection...Do we understand the burden in our respective countries, and has the response been adequate?”
The goal for the meeting is to have leaders “step up to commit, not for one day, or for one meeting, but for the duration of the effort,” said Dr. Goosby. “We must make sure that the words that we hear next week from our leaders translate into action...Next week the world will say, ‘No more. No longer. No one is immune to TB. Tuberculosis is preventable; tuberculosis is treatable; tuberculosis is curable.’”
The BMJ commentary, by Marcel A. Behr, MD, of McGill International TB Centre, Infectious Diseases and Immunity in Global Health Program, McGill University Health Centre Research Institute, and his colleagues, recommend caution when building a prevention strategy around treating many millions of individuals with “latent” TB. They wrote, “Immunoreactivity to TB does not necessarily indicate the presence of live bacteria, as reactivity can persist after infection has been cleared. Classifying two billion people with evidence of immunoreactivity as having latent TB infection may divert fundamental research and public health interventions away from transmissible active TB disease and newly infected people at highest risk of progression to disease.”
This story was updated on 09/24/2018
A concerted a lethal disease affecting one-quarter of the world’s population by the year 2030.

On September 26 the United Nations General Assembly will convene a high-level meeting of global stakeholders to solidify the eradication plan, addressing the global crisis of tuberculosis (TB), the world’s most deadly infectious disease.
“We must seize the moment,” said Tereza Kasaeva, MD, director of the World Health Organization’s global TB program, speaking at a telebriefing and press conference accompanying the release of the World Health Organization’s annual global tuberculosis report. “It’s unacceptable in the 21st century that millions lose their lives to this preventable and curable disease.”
TB caused 1.6 million deaths globally in 2017, and the World Health Organization (WHO) estimates that of the 10 million new cases of TB last year, 558,000 are multi-drug resistant (MDR) infections.
Though death rates and new cases are falling globally each year, significantly more resources are needed to boost access to preventive treatment for latent TB infection; “Most people needing it are not yet accessing care,” according to the press briefing accompanying the report.
A review and commentary on TB incubation and latency published in BMJ (2018;362:k2738 doi: 10.1136/bmj.k2738; e-pub 23 Aug 2018) has called into question the focus preventive treatment of latent cases at the expense of reaching those most likely to die from TB (e.g., HIV patients, children of individuals living with active TB). The authors state that “latent” TB is identified by indirect evidence of present or past infection with Mycobacterium tuberculosis as inferred by a detectable adaptive immune response to M tuberculosis antigens. Active TB infection is overwhelmingly the result of a primary infection and almost always occurs within two years.
In order to meet the ambitious goal of TB eradication by the year 2030, treatment coverage must rise to 90% globally from the current 64%, according to the report.
Progress in southern Africa and in the Russian Federation, where efforts have led to a 30% reduction in TB mortality and a decrease in incidence of 5% per year, show that steep reductions in TB are possible when resources are brought to bear on the problem, said Dr. Kasaeva. “We should acknowledge that actions in some countries and regions show that progress can accelerate,” she said. Still, she noted, “Four thousand lives per day are lost to TB. Tuberculosis is the leading killer of people living with HIV, and the major cause of deaths related to antimicrobial resistance” at a global level.
Two thirds of all TB cases occur in eight countries, with India, China, and Indonesia leading this group. About half of the cases of MDR TB occur in India, China, and Russia, said Dr. Kasaeva, and globally only one in four individuals with MDR TB who need access to treatment have received it. “We need to urgently tackle the multidrug resistant TB public health crisis,” she said.
Major impediments to successful public health efforts against TB are underdiagnosis and underreporting: It is estimated that 3.6 million of 2017’s 10 million new cases were not officially recorded or reported. Countries where these problems are most serious include India, Indonesia, and Nigeria. Fewer than half of the children with TB are reported globally, according to the report.
People living with HIV/AIDS who are also infected with TB number nearly 1,000,000, but only about half of these were officially reported in 2017.
In terms of prevention priorities, WHO has recommended targeting treatment of latent TB in two groups: People living with HIV/AIDS, and children under the age of 5 years who live in households with TB-infected individuals.
“To enable these actions,” said Dr. Kasaeva, “we need strengthened commitments not just for TB care, but for overall health services. So the aim for universal coverage is real.” Underreporting is particularly prevalent in lower income countries with large unregulated private sectors, she said, though India and Indonesia have taken corrective steps to increase reporting.
A meaningful global initiative will not come cheap: The current annual shortfall in funding for TB prevention, diagnosis, and treatment is about $3.5 billion. By the year 2022, the gap between funding and what’s needed to stay on track for the 2030 target will be over $6 billion, said Dr. Kasaeva.
The best use of increased resources for TB eradication will be in locally focused efforts, said Irene Koek, MD, the United States Agency for International Development’s deputy administrator for global health. “It is likely that each region requires a tailored response.” Further, “to improve quality of care we need to ensure that services are patient centered,” she said at the press conference.
To that end, Dr. Koek expects that at the upcoming high-level meeting, the United Nations member states will be called on to develop an open framework, with clear accountability for monitoring and reviewing progress. The road forward should “celebrate accomplishments and acknowledge shortcomings,” she said. Some recent studies have shown that treatment success rates above 80% for patients with MDR TB can be achieved.
“Lessons learned from these experiences should be documented and shared in order to replicate success globally,” said Dr. Koek.
The United States, said Dr. Koek, is the leading global investor in TB research and treatment. “We welcome increased partnerships, especially with countries with the highest burden, to end global suffering from this disease.”
Eric Goosby, MD, the United Nations special envoy on TB, used his speaking time to lend some perspective to the social framework around TB’s longtime lethality.
There are aspects of TB infection that differentiate it from HIV/AIDS, said Dr. Goosby, who has spent most of his clinical and public health career on HIV/AIDS treatment and prevention. In contrast to an infection that at present requires a lifetime of treatment, TB can ordinarily be treated in 6 months, making it an unpleasant episode that an individual may be eager to move past. Additionally, the fact that TB has had a “hold on the world since the time of the ancient Egyptians” may paradoxically have served to lessen urgency in research and treatment efforts, he noted.
Dr. Goosby also spoke of the stigma surrounding TB, whose sufferers are likely to be facing dire poverty, malnutrition, and other infectious disease burdens. Civil society concerned with TB, he said, has spoken up “for those without a voice, for those who have difficulty advocating for themselves.”
Dr. Kasaeva agreed, noting that TB “affects the poorest of the poor, which makes it extraordinarily difficult for activism to come from that population.”
However, others have spoken for those affected, said Dr. Goosby. “The TB civil society has put its heart and soul this last year into gathering political will from leaders around the world…. It’s not a passive effort; it involves a lot of work.” During the past year of concerted effort, he said, “All of us have known the difficulty of pushing a political leader up that learning curve.”
As the upcoming high-level meeting approaches, those who have been working on the effort can feel the momentum, said Dr. Goosby. Still, he noted, “While there’s a significant step forward, this is not the time for a victory dance. This is really the time for a reflection...Do we understand the burden in our respective countries, and has the response been adequate?”
The goal for the meeting is to have leaders “step up to commit, not for one day, or for one meeting, but for the duration of the effort,” said Dr. Goosby. “We must make sure that the words that we hear next week from our leaders translate into action...Next week the world will say, ‘No more. No longer. No one is immune to TB. Tuberculosis is preventable; tuberculosis is treatable; tuberculosis is curable.’”
The BMJ commentary, by Marcel A. Behr, MD, of McGill International TB Centre, Infectious Diseases and Immunity in Global Health Program, McGill University Health Centre Research Institute, and his colleagues, recommend caution when building a prevention strategy around treating many millions of individuals with “latent” TB. They wrote, “Immunoreactivity to TB does not necessarily indicate the presence of live bacteria, as reactivity can persist after infection has been cleared. Classifying two billion people with evidence of immunoreactivity as having latent TB infection may divert fundamental research and public health interventions away from transmissible active TB disease and newly infected people at highest risk of progression to disease.”
This story was updated on 09/24/2018
FROM A WORLD HEALTH ORGANIZATION PRESS CONFERENCE
Dupilumab efficacy extends to chronic rhinosinusitis/nasal polyposis
PARIS – Severe asthma patients with chronic rhinosinusitis (CRS), nasal polyposis (NP), or both derive more protection from severe exacerbations with the monoclonal antibody dupilumab than do those who do not have these comorbidities, according to a post hoc analysis of a phase 3 trial presented at the annual congress of the European Respiratory Society.
“Dupilumab reduced rates of severe exacerbations and improved FEV1 [forced expiratory volume in 1 second] in patients in asthma patients with or without CRS/NP. In those with CRS/NP, dupilumab reduced symptoms associated with these comorbidities,” reported Ian Pavord, MBBS, statutory chair in respiratory medicine at University of Oxford (England).
The data were drawn from the phase 3 Liberty Asthma Quest trial, which was published earlier this year in the New England Journal of Medicine (2018;378:2486-96). In that study, both the 200-mg and 300-mg dose of dupilumab (Dupixent) administered every 2 weeks was associated with about a 50% reduction in the annualized rate of severe exacerbations relative to placebo (P less than .001 for both doses).
In this new post hoc analysis, response in the 382 patients who entered the study with a history of CRS/NP was compared with the 1,520 without CRS/NP. In the CRS/NP patients, the reductions relative to placebo in the rates of severe exacerbations, defined as 3 or more days of systemic glucocorticoids or visit to an emergency department leading to treatment with systemic glucocorticoids, were 63% and 61% for the 200-mg and 300-mg doses of dupilumab, respectively (both P less than .001).
In the non-CRS/NP arms, the reductions relative to placebo were 42% and 40%, respectively (both P less than .001). The greater relative reductions in the CRS/NP patients were achieved even though they were older (mean age approximately 52 vs. 47 years for non-CRS/NP patients), had a significantly greater number of exacerbations in the past year (P = .027), and had higher baseline fractional exhaled nitric oxide and eosinophil levels (both P less than .001), Dr. Pavord reported.
“The greater asthma severity in the CRS/NP patients in this trial is consistent with that reported previously by others,” Dr. Pavord said.
Although the greater asthma severity may have provided a larger margin for benefit, Dr. Pavord also reported that there were improvements in CRS/NP-specific symptoms as measured with the 22-item Sino-Nasal Outcome Test (SNOT-22). By week 12, the total score reduction in SNOT-22 was approximately 15 points (P less than .05) from baseline for both the 200-mg and 300-mg dupilumab doses. This was significantly greater (P less than .05) relative to modest SNOT-22 reductions in the placebo arms (P less than .05). After 52 weeks, the reduction In SNOT-22 scores were sustained, providing an even greater statistical advantage over placebo (P less than .001).
In addition to greater protection against severe exacerbations and CRS/NP-specific symptoms, dupilumab may offer specific improvements on CRS/NP pathology, according to Dr. Pavord. Although imaging was not part of this study, he noted in particular that previous studies with dupilumab as well as other biologics have shown shrinkage of nasal polyps with treatment.
Dupilumab was similarly well tolerated in those with and without CRS/NP. The most common adverse event was injection site reactions in both groups, Dr. Pavord said.
Calling CRS and NP “important comorbidities” in severe asthma patients, Dr. Pavord said that this analysis should be reassuring for those who with CRS/NP who are being considered for dupilumab. Already approved for treatment of atopic dermatitis, dupilumab, which binds to interleukin-4 (IL-4) and IL-13 receptors, is currently under review for the treatment of moderate to severe asthma.
Dr. Pavord has financial relationships with Aerocrine, Almirall, AstraZeneca, Boehringer Ingelheim, Chiesi, GlaxoSmithKline, Knapp, Merck Sharpe, Novartis, Knapp Teva, RespiVert, and Schering-Plough.
PARIS – Severe asthma patients with chronic rhinosinusitis (CRS), nasal polyposis (NP), or both derive more protection from severe exacerbations with the monoclonal antibody dupilumab than do those who do not have these comorbidities, according to a post hoc analysis of a phase 3 trial presented at the annual congress of the European Respiratory Society.
“Dupilumab reduced rates of severe exacerbations and improved FEV1 [forced expiratory volume in 1 second] in patients in asthma patients with or without CRS/NP. In those with CRS/NP, dupilumab reduced symptoms associated with these comorbidities,” reported Ian Pavord, MBBS, statutory chair in respiratory medicine at University of Oxford (England).
The data were drawn from the phase 3 Liberty Asthma Quest trial, which was published earlier this year in the New England Journal of Medicine (2018;378:2486-96). In that study, both the 200-mg and 300-mg dose of dupilumab (Dupixent) administered every 2 weeks was associated with about a 50% reduction in the annualized rate of severe exacerbations relative to placebo (P less than .001 for both doses).
In this new post hoc analysis, response in the 382 patients who entered the study with a history of CRS/NP was compared with the 1,520 without CRS/NP. In the CRS/NP patients, the reductions relative to placebo in the rates of severe exacerbations, defined as 3 or more days of systemic glucocorticoids or visit to an emergency department leading to treatment with systemic glucocorticoids, were 63% and 61% for the 200-mg and 300-mg doses of dupilumab, respectively (both P less than .001).
In the non-CRS/NP arms, the reductions relative to placebo were 42% and 40%, respectively (both P less than .001). The greater relative reductions in the CRS/NP patients were achieved even though they were older (mean age approximately 52 vs. 47 years for non-CRS/NP patients), had a significantly greater number of exacerbations in the past year (P = .027), and had higher baseline fractional exhaled nitric oxide and eosinophil levels (both P less than .001), Dr. Pavord reported.
“The greater asthma severity in the CRS/NP patients in this trial is consistent with that reported previously by others,” Dr. Pavord said.
Although the greater asthma severity may have provided a larger margin for benefit, Dr. Pavord also reported that there were improvements in CRS/NP-specific symptoms as measured with the 22-item Sino-Nasal Outcome Test (SNOT-22). By week 12, the total score reduction in SNOT-22 was approximately 15 points (P less than .05) from baseline for both the 200-mg and 300-mg dupilumab doses. This was significantly greater (P less than .05) relative to modest SNOT-22 reductions in the placebo arms (P less than .05). After 52 weeks, the reduction In SNOT-22 scores were sustained, providing an even greater statistical advantage over placebo (P less than .001).
In addition to greater protection against severe exacerbations and CRS/NP-specific symptoms, dupilumab may offer specific improvements on CRS/NP pathology, according to Dr. Pavord. Although imaging was not part of this study, he noted in particular that previous studies with dupilumab as well as other biologics have shown shrinkage of nasal polyps with treatment.
Dupilumab was similarly well tolerated in those with and without CRS/NP. The most common adverse event was injection site reactions in both groups, Dr. Pavord said.
Calling CRS and NP “important comorbidities” in severe asthma patients, Dr. Pavord said that this analysis should be reassuring for those who with CRS/NP who are being considered for dupilumab. Already approved for treatment of atopic dermatitis, dupilumab, which binds to interleukin-4 (IL-4) and IL-13 receptors, is currently under review for the treatment of moderate to severe asthma.
Dr. Pavord has financial relationships with Aerocrine, Almirall, AstraZeneca, Boehringer Ingelheim, Chiesi, GlaxoSmithKline, Knapp, Merck Sharpe, Novartis, Knapp Teva, RespiVert, and Schering-Plough.
PARIS – Severe asthma patients with chronic rhinosinusitis (CRS), nasal polyposis (NP), or both derive more protection from severe exacerbations with the monoclonal antibody dupilumab than do those who do not have these comorbidities, according to a post hoc analysis of a phase 3 trial presented at the annual congress of the European Respiratory Society.
“Dupilumab reduced rates of severe exacerbations and improved FEV1 [forced expiratory volume in 1 second] in patients in asthma patients with or without CRS/NP. In those with CRS/NP, dupilumab reduced symptoms associated with these comorbidities,” reported Ian Pavord, MBBS, statutory chair in respiratory medicine at University of Oxford (England).
The data were drawn from the phase 3 Liberty Asthma Quest trial, which was published earlier this year in the New England Journal of Medicine (2018;378:2486-96). In that study, both the 200-mg and 300-mg dose of dupilumab (Dupixent) administered every 2 weeks was associated with about a 50% reduction in the annualized rate of severe exacerbations relative to placebo (P less than .001 for both doses).
In this new post hoc analysis, response in the 382 patients who entered the study with a history of CRS/NP was compared with the 1,520 without CRS/NP. In the CRS/NP patients, the reductions relative to placebo in the rates of severe exacerbations, defined as 3 or more days of systemic glucocorticoids or visit to an emergency department leading to treatment with systemic glucocorticoids, were 63% and 61% for the 200-mg and 300-mg doses of dupilumab, respectively (both P less than .001).
In the non-CRS/NP arms, the reductions relative to placebo were 42% and 40%, respectively (both P less than .001). The greater relative reductions in the CRS/NP patients were achieved even though they were older (mean age approximately 52 vs. 47 years for non-CRS/NP patients), had a significantly greater number of exacerbations in the past year (P = .027), and had higher baseline fractional exhaled nitric oxide and eosinophil levels (both P less than .001), Dr. Pavord reported.
“The greater asthma severity in the CRS/NP patients in this trial is consistent with that reported previously by others,” Dr. Pavord said.
Although the greater asthma severity may have provided a larger margin for benefit, Dr. Pavord also reported that there were improvements in CRS/NP-specific symptoms as measured with the 22-item Sino-Nasal Outcome Test (SNOT-22). By week 12, the total score reduction in SNOT-22 was approximately 15 points (P less than .05) from baseline for both the 200-mg and 300-mg dupilumab doses. This was significantly greater (P less than .05) relative to modest SNOT-22 reductions in the placebo arms (P less than .05). After 52 weeks, the reduction In SNOT-22 scores were sustained, providing an even greater statistical advantage over placebo (P less than .001).
In addition to greater protection against severe exacerbations and CRS/NP-specific symptoms, dupilumab may offer specific improvements on CRS/NP pathology, according to Dr. Pavord. Although imaging was not part of this study, he noted in particular that previous studies with dupilumab as well as other biologics have shown shrinkage of nasal polyps with treatment.
Dupilumab was similarly well tolerated in those with and without CRS/NP. The most common adverse event was injection site reactions in both groups, Dr. Pavord said.
Calling CRS and NP “important comorbidities” in severe asthma patients, Dr. Pavord said that this analysis should be reassuring for those who with CRS/NP who are being considered for dupilumab. Already approved for treatment of atopic dermatitis, dupilumab, which binds to interleukin-4 (IL-4) and IL-13 receptors, is currently under review for the treatment of moderate to severe asthma.
Dr. Pavord has financial relationships with Aerocrine, Almirall, AstraZeneca, Boehringer Ingelheim, Chiesi, GlaxoSmithKline, Knapp, Merck Sharpe, Novartis, Knapp Teva, RespiVert, and Schering-Plough.
REPORTING FROM THE ERS CONGRESS 2018
Key clinical point: In asthma patients with chronic rhinosinusitis and/or nasal polyposis (CRS/NP), dupilumab reduces exacerbations.
Major finding: At 52 weeks, severe exacerbations were reduced 61% in CRS/NP patients and 40% in non-CRS/NP patients (both P less than .001).
Study details: Post hoc analysis of phase 3 trial.
Disclosures: Dr. Pavord has financial relationships with Aerocrine, Almirall, AstraZeneca, Boehringer Ingelheim, Chiesi, GlaxoSmithKline, Knapp, Merck Sharpe, Novartis, Knapp Teva, RespiVert, and Schering-Plough.
Updated IPF guideline refines diagnostic criteria with HRCT
A recently updated guideline for idiopathic pulmonary fibrosis (IPF) provides refined diagnostic criteria in an effort to improve clinical application and diagnostic accuracy.
Of note, the guideline recommends that high-resolution CT (HRCT) patterns be used to dictate management course. The guideline also calls for detailed medical history, serological testing to exclude connective tissue disease, and multidisciplinary discussion. Serum biomarkers are recommended against as a means of distinguishing between IPF and other interstitial lung diseases (ILDs).
“Diagnosing IPF is challenging because these symptoms are nonspecific: They occur with all other interstitial lung diseases and with other respiratory problems,” Ganesh Raghu, MD, chair of the guideline committee and professor of medicine and director of the Center for Interstitial Lung Disease at the University of Washington, Seattle, said in a written statement. “Because drugs may slow the progression of IPF, an early and accurate diagnosis is essential for prompt and appropriate treatment for this fatal disease.”
The 2018 guideline represents a second collaborative effort from the American Thoracic Society, European Respiratory Society, Japanese Respiratory Society, and Latin American Thoracic Society. The guideline committee consisted of 29 clinicians, scientists, and a patient with IPF. They evaluated all IPF-related evidence and rated the quality of findings with the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) system. The first IPF guideline was published 7 years ago; the intervening time has revealed some clinical limitations that the 2018 guideline aims to fix.
“The 2011 guideline provided the first evidence-based, formal criteria for diagnosis of IPF and allowed patients with a well-defined diagnosis of IPF to participate in numerous clinical studies and randomized controlled trials that enhanced our understanding of the disease,” Dr. Raghu said. “However, it became clear that there were significant challenges in ascertaining the diagnosis per the 2011 criteria, and abundant evidence accumulated since then allowed the committee to refine the diagnostic criteria now.”
“This [updated] guideline is intended to help clinicians make an accurate diagnosis of IPF,” the authors wrote in the American Journal of Respiratory and Critical Care Medicine, “and to empower them to implement recommended courses of action in the context of individual patient values and preferences, particularly decisions regarding which diagnostic interventions to pursue.”
While the 2011 guideline did not distinguish between patients with different HRCT patterns, the 2018 guideline emphasizes the use of HRCT. It is now recommended that patients undergo HRCT to determine the pattern of usual interstitial pneumonia (UIP). Broadly, patients exhibit the UIP pattern or one of three possible non-UIP patterns (probable UIP, indeterminate UIP, or an alternative diagnosis). Recommendations are specific for UIP and non-UIP.
Patients with probable UIP, indeterminate UIP, or an alternative diagnosis should undergo bronchoalveolar lavage (BAL) and surgical lung biopsy (SLB). Transbronchial lung biopsy (TBBx) and lung cryobiopsy recommendations were not described in these patients because of a lack of evidence. However, patients with UIP should not undergo BAL, SLB, TBBx, or cryobiopsy.
For all patients, the 2018 guideline recommends that medical histories include environmental exposure and medication use, and that serological testing is performed to exclude connective tissue disease. Multidisciplinary discussion is conditionally recommended for diagnostic decision making, particularly when a patient has probable UIP, indeterminate UIP, or an alternative diagnosis.
Finally, the guideline recommends against the use of serum biomarkers as a means of distinguishing between IPF and other ILDs because of weak data on this point. “For the time being, the guideline panel dismissed serum biomarker measurement as an approach to distinguishing IPF from other ILDs because of the high false-positive and false-negative result rates,” the panel wrote.
“Our hope is that this new guideline will bridge the gap between the experienced IPF experts and general pulmonologists in making a prompt and accurate diagnosis of IPF for the individual unfortunately confronted with the disease,” Dr. Raghu said. “This will allow patients to make well-informed decisions about treatment options and participation in clinical trials.”
Looking to the future, the panel described an “urgent need to refine and validate diagnostic approaches to ILD. These needs can be roughly categorized as investigations into the roles of clinical observations, HRCT, bronchoscopy, histopathology, and biomarkers.” The authors also emphasized “the need to refine prognostic approaches, identify risk factors for the development of IPF, and determine the impact and approach to the diagnosis of comorbid illness in the patient with IPF.”
The authors reported funding from Bellerophon, Gilead, Roche, Sanofi, and others.
SOURCE: Raghu G et al. Am J Respir Crit Care Med. 2018 Sep 1. doi: 10.1164/rccm.201807-1255ST.
A recently updated guideline for idiopathic pulmonary fibrosis (IPF) provides refined diagnostic criteria in an effort to improve clinical application and diagnostic accuracy.
Of note, the guideline recommends that high-resolution CT (HRCT) patterns be used to dictate management course. The guideline also calls for detailed medical history, serological testing to exclude connective tissue disease, and multidisciplinary discussion. Serum biomarkers are recommended against as a means of distinguishing between IPF and other interstitial lung diseases (ILDs).
“Diagnosing IPF is challenging because these symptoms are nonspecific: They occur with all other interstitial lung diseases and with other respiratory problems,” Ganesh Raghu, MD, chair of the guideline committee and professor of medicine and director of the Center for Interstitial Lung Disease at the University of Washington, Seattle, said in a written statement. “Because drugs may slow the progression of IPF, an early and accurate diagnosis is essential for prompt and appropriate treatment for this fatal disease.”
The 2018 guideline represents a second collaborative effort from the American Thoracic Society, European Respiratory Society, Japanese Respiratory Society, and Latin American Thoracic Society. The guideline committee consisted of 29 clinicians, scientists, and a patient with IPF. They evaluated all IPF-related evidence and rated the quality of findings with the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) system. The first IPF guideline was published 7 years ago; the intervening time has revealed some clinical limitations that the 2018 guideline aims to fix.
“The 2011 guideline provided the first evidence-based, formal criteria for diagnosis of IPF and allowed patients with a well-defined diagnosis of IPF to participate in numerous clinical studies and randomized controlled trials that enhanced our understanding of the disease,” Dr. Raghu said. “However, it became clear that there were significant challenges in ascertaining the diagnosis per the 2011 criteria, and abundant evidence accumulated since then allowed the committee to refine the diagnostic criteria now.”
“This [updated] guideline is intended to help clinicians make an accurate diagnosis of IPF,” the authors wrote in the American Journal of Respiratory and Critical Care Medicine, “and to empower them to implement recommended courses of action in the context of individual patient values and preferences, particularly decisions regarding which diagnostic interventions to pursue.”
While the 2011 guideline did not distinguish between patients with different HRCT patterns, the 2018 guideline emphasizes the use of HRCT. It is now recommended that patients undergo HRCT to determine the pattern of usual interstitial pneumonia (UIP). Broadly, patients exhibit the UIP pattern or one of three possible non-UIP patterns (probable UIP, indeterminate UIP, or an alternative diagnosis). Recommendations are specific for UIP and non-UIP.
Patients with probable UIP, indeterminate UIP, or an alternative diagnosis should undergo bronchoalveolar lavage (BAL) and surgical lung biopsy (SLB). Transbronchial lung biopsy (TBBx) and lung cryobiopsy recommendations were not described in these patients because of a lack of evidence. However, patients with UIP should not undergo BAL, SLB, TBBx, or cryobiopsy.
For all patients, the 2018 guideline recommends that medical histories include environmental exposure and medication use, and that serological testing is performed to exclude connective tissue disease. Multidisciplinary discussion is conditionally recommended for diagnostic decision making, particularly when a patient has probable UIP, indeterminate UIP, or an alternative diagnosis.
Finally, the guideline recommends against the use of serum biomarkers as a means of distinguishing between IPF and other ILDs because of weak data on this point. “For the time being, the guideline panel dismissed serum biomarker measurement as an approach to distinguishing IPF from other ILDs because of the high false-positive and false-negative result rates,” the panel wrote.
“Our hope is that this new guideline will bridge the gap between the experienced IPF experts and general pulmonologists in making a prompt and accurate diagnosis of IPF for the individual unfortunately confronted with the disease,” Dr. Raghu said. “This will allow patients to make well-informed decisions about treatment options and participation in clinical trials.”
Looking to the future, the panel described an “urgent need to refine and validate diagnostic approaches to ILD. These needs can be roughly categorized as investigations into the roles of clinical observations, HRCT, bronchoscopy, histopathology, and biomarkers.” The authors also emphasized “the need to refine prognostic approaches, identify risk factors for the development of IPF, and determine the impact and approach to the diagnosis of comorbid illness in the patient with IPF.”
The authors reported funding from Bellerophon, Gilead, Roche, Sanofi, and others.
SOURCE: Raghu G et al. Am J Respir Crit Care Med. 2018 Sep 1. doi: 10.1164/rccm.201807-1255ST.
A recently updated guideline for idiopathic pulmonary fibrosis (IPF) provides refined diagnostic criteria in an effort to improve clinical application and diagnostic accuracy.
Of note, the guideline recommends that high-resolution CT (HRCT) patterns be used to dictate management course. The guideline also calls for detailed medical history, serological testing to exclude connective tissue disease, and multidisciplinary discussion. Serum biomarkers are recommended against as a means of distinguishing between IPF and other interstitial lung diseases (ILDs).
“Diagnosing IPF is challenging because these symptoms are nonspecific: They occur with all other interstitial lung diseases and with other respiratory problems,” Ganesh Raghu, MD, chair of the guideline committee and professor of medicine and director of the Center for Interstitial Lung Disease at the University of Washington, Seattle, said in a written statement. “Because drugs may slow the progression of IPF, an early and accurate diagnosis is essential for prompt and appropriate treatment for this fatal disease.”
The 2018 guideline represents a second collaborative effort from the American Thoracic Society, European Respiratory Society, Japanese Respiratory Society, and Latin American Thoracic Society. The guideline committee consisted of 29 clinicians, scientists, and a patient with IPF. They evaluated all IPF-related evidence and rated the quality of findings with the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) system. The first IPF guideline was published 7 years ago; the intervening time has revealed some clinical limitations that the 2018 guideline aims to fix.
“The 2011 guideline provided the first evidence-based, formal criteria for diagnosis of IPF and allowed patients with a well-defined diagnosis of IPF to participate in numerous clinical studies and randomized controlled trials that enhanced our understanding of the disease,” Dr. Raghu said. “However, it became clear that there were significant challenges in ascertaining the diagnosis per the 2011 criteria, and abundant evidence accumulated since then allowed the committee to refine the diagnostic criteria now.”
“This [updated] guideline is intended to help clinicians make an accurate diagnosis of IPF,” the authors wrote in the American Journal of Respiratory and Critical Care Medicine, “and to empower them to implement recommended courses of action in the context of individual patient values and preferences, particularly decisions regarding which diagnostic interventions to pursue.”
While the 2011 guideline did not distinguish between patients with different HRCT patterns, the 2018 guideline emphasizes the use of HRCT. It is now recommended that patients undergo HRCT to determine the pattern of usual interstitial pneumonia (UIP). Broadly, patients exhibit the UIP pattern or one of three possible non-UIP patterns (probable UIP, indeterminate UIP, or an alternative diagnosis). Recommendations are specific for UIP and non-UIP.
Patients with probable UIP, indeterminate UIP, or an alternative diagnosis should undergo bronchoalveolar lavage (BAL) and surgical lung biopsy (SLB). Transbronchial lung biopsy (TBBx) and lung cryobiopsy recommendations were not described in these patients because of a lack of evidence. However, patients with UIP should not undergo BAL, SLB, TBBx, or cryobiopsy.
For all patients, the 2018 guideline recommends that medical histories include environmental exposure and medication use, and that serological testing is performed to exclude connective tissue disease. Multidisciplinary discussion is conditionally recommended for diagnostic decision making, particularly when a patient has probable UIP, indeterminate UIP, or an alternative diagnosis.
Finally, the guideline recommends against the use of serum biomarkers as a means of distinguishing between IPF and other ILDs because of weak data on this point. “For the time being, the guideline panel dismissed serum biomarker measurement as an approach to distinguishing IPF from other ILDs because of the high false-positive and false-negative result rates,” the panel wrote.
“Our hope is that this new guideline will bridge the gap between the experienced IPF experts and general pulmonologists in making a prompt and accurate diagnosis of IPF for the individual unfortunately confronted with the disease,” Dr. Raghu said. “This will allow patients to make well-informed decisions about treatment options and participation in clinical trials.”
Looking to the future, the panel described an “urgent need to refine and validate diagnostic approaches to ILD. These needs can be roughly categorized as investigations into the roles of clinical observations, HRCT, bronchoscopy, histopathology, and biomarkers.” The authors also emphasized “the need to refine prognostic approaches, identify risk factors for the development of IPF, and determine the impact and approach to the diagnosis of comorbid illness in the patient with IPF.”
The authors reported funding from Bellerophon, Gilead, Roche, Sanofi, and others.
SOURCE: Raghu G et al. Am J Respir Crit Care Med. 2018 Sep 1. doi: 10.1164/rccm.201807-1255ST.
FROM THE AMERICAN JOURNAL OF RESPIRATORY AND CRITICAL CARE MEDICINE
Review protocols, follow reprocessing guidelines to cut device-related HAIs
ATLANTA – Ongoing vigilance regarding the role of and transmission of antimicrobial-resistant pathogen is needed, according to Isaac Benowitz, MD, of the Centers for Disease Control and Prevention’s Division of Healthcare Quality Promotion (DHQP).
A review of records from the DHQP, which investigates and responds to infections and related adverse events in health care settings upon invitation, showed that in 2017 environmental pathogens were most often the triggers for these investigations, said Dr. Benowitz, a medical epidemiologist.
He reviewed internal records for consultations with state and local health departments involving medical devices and collected data on health care setting, pathogen, investigation findings including possible exposure or transmission, and public health actions.
Of 285 consultations, 48 involved a specific medical device or general medical device reprocessing, he said, noting that most of those 48 were in an acute care hospital (63%) or clinic (19%).
“The most frequent pathogens noted in these consultations were nontuberculous mycobacteria at 21%, Candida species ... at 10%, and Burkholderia species ... at 8%,” he said, noting that a wide variety of devices were implicated.
In the inpatient setting these devices included ventilators, dialysis machines, breast pumps, central lines, and respiratory therapy equipment. In the outpatient setting they included glucometers and opthalmic equipment.
“In many settings we saw issues with endoscopes, including duodenoscopes, but also bronchoscopes,” he added.
Actions taken as part of the investigations included medical device recalls, improved infection control and reprocessing procedures, and patient notification, education, guidance, testing, and treatment.
In some cases there was disciplinary action or oversight for health care professionals, he added.
Investigations identified medical devices contaminated in manufacturing, incorrect reprocessing of endoscopes or ventilators, and inappropriate medical device use or reuse, he said.
A number of lessons can be learned from these and other investigations, he added.
“First, devices can be reservoirs and transmission vectors for health care–associated infections. Second, health care facilities, health care facility staff, and public health partners should take opportunities to review protocols and the practices within those protocol,” he said. “These are opportunities to strengthen infection control practices even in the absence of documented transmission.”
In fact, in most of the investigations he discussed, transmission was rarely confirmed to be associated with a medical device. This was largely because of a lack of “epidemiological rigor,” but associations between health care–associated infections and medical devices “are still quite meaningful and often actionable,” he said.
Dr. Benowitz stressed the importance of engaging public health partners to discuss findings and actions, explaining that “what may look like a single-facility issue may have a very different perspective when you realize that there’s a similar issue at another facility elsewhere.”
“For all devices, it’s important to ensure adherence to the device reprocessing guidelines, “ he added, noting that these include a combination of facility protocols, manufacturer instructions for use, and guidance from organizations like the Food and Drug Administration and the CDC.
Dr. Benowitz reported having no disclosures.
[email protected]
SOURCE: Benowitz I et al. ICEID 2018, Oral Abstract Presentation E2.
ATLANTA – Ongoing vigilance regarding the role of and transmission of antimicrobial-resistant pathogen is needed, according to Isaac Benowitz, MD, of the Centers for Disease Control and Prevention’s Division of Healthcare Quality Promotion (DHQP).
A review of records from the DHQP, which investigates and responds to infections and related adverse events in health care settings upon invitation, showed that in 2017 environmental pathogens were most often the triggers for these investigations, said Dr. Benowitz, a medical epidemiologist.
He reviewed internal records for consultations with state and local health departments involving medical devices and collected data on health care setting, pathogen, investigation findings including possible exposure or transmission, and public health actions.
Of 285 consultations, 48 involved a specific medical device or general medical device reprocessing, he said, noting that most of those 48 were in an acute care hospital (63%) or clinic (19%).
“The most frequent pathogens noted in these consultations were nontuberculous mycobacteria at 21%, Candida species ... at 10%, and Burkholderia species ... at 8%,” he said, noting that a wide variety of devices were implicated.
In the inpatient setting these devices included ventilators, dialysis machines, breast pumps, central lines, and respiratory therapy equipment. In the outpatient setting they included glucometers and opthalmic equipment.
“In many settings we saw issues with endoscopes, including duodenoscopes, but also bronchoscopes,” he added.
Actions taken as part of the investigations included medical device recalls, improved infection control and reprocessing procedures, and patient notification, education, guidance, testing, and treatment.
In some cases there was disciplinary action or oversight for health care professionals, he added.
Investigations identified medical devices contaminated in manufacturing, incorrect reprocessing of endoscopes or ventilators, and inappropriate medical device use or reuse, he said.
A number of lessons can be learned from these and other investigations, he added.
“First, devices can be reservoirs and transmission vectors for health care–associated infections. Second, health care facilities, health care facility staff, and public health partners should take opportunities to review protocols and the practices within those protocol,” he said. “These are opportunities to strengthen infection control practices even in the absence of documented transmission.”
In fact, in most of the investigations he discussed, transmission was rarely confirmed to be associated with a medical device. This was largely because of a lack of “epidemiological rigor,” but associations between health care–associated infections and medical devices “are still quite meaningful and often actionable,” he said.
Dr. Benowitz stressed the importance of engaging public health partners to discuss findings and actions, explaining that “what may look like a single-facility issue may have a very different perspective when you realize that there’s a similar issue at another facility elsewhere.”
“For all devices, it’s important to ensure adherence to the device reprocessing guidelines, “ he added, noting that these include a combination of facility protocols, manufacturer instructions for use, and guidance from organizations like the Food and Drug Administration and the CDC.
Dr. Benowitz reported having no disclosures.
[email protected]
SOURCE: Benowitz I et al. ICEID 2018, Oral Abstract Presentation E2.
ATLANTA – Ongoing vigilance regarding the role of and transmission of antimicrobial-resistant pathogen is needed, according to Isaac Benowitz, MD, of the Centers for Disease Control and Prevention’s Division of Healthcare Quality Promotion (DHQP).
A review of records from the DHQP, which investigates and responds to infections and related adverse events in health care settings upon invitation, showed that in 2017 environmental pathogens were most often the triggers for these investigations, said Dr. Benowitz, a medical epidemiologist.
He reviewed internal records for consultations with state and local health departments involving medical devices and collected data on health care setting, pathogen, investigation findings including possible exposure or transmission, and public health actions.
Of 285 consultations, 48 involved a specific medical device or general medical device reprocessing, he said, noting that most of those 48 were in an acute care hospital (63%) or clinic (19%).
“The most frequent pathogens noted in these consultations were nontuberculous mycobacteria at 21%, Candida species ... at 10%, and Burkholderia species ... at 8%,” he said, noting that a wide variety of devices were implicated.
In the inpatient setting these devices included ventilators, dialysis machines, breast pumps, central lines, and respiratory therapy equipment. In the outpatient setting they included glucometers and opthalmic equipment.
“In many settings we saw issues with endoscopes, including duodenoscopes, but also bronchoscopes,” he added.
Actions taken as part of the investigations included medical device recalls, improved infection control and reprocessing procedures, and patient notification, education, guidance, testing, and treatment.
In some cases there was disciplinary action or oversight for health care professionals, he added.
Investigations identified medical devices contaminated in manufacturing, incorrect reprocessing of endoscopes or ventilators, and inappropriate medical device use or reuse, he said.
A number of lessons can be learned from these and other investigations, he added.
“First, devices can be reservoirs and transmission vectors for health care–associated infections. Second, health care facilities, health care facility staff, and public health partners should take opportunities to review protocols and the practices within those protocol,” he said. “These are opportunities to strengthen infection control practices even in the absence of documented transmission.”
In fact, in most of the investigations he discussed, transmission was rarely confirmed to be associated with a medical device. This was largely because of a lack of “epidemiological rigor,” but associations between health care–associated infections and medical devices “are still quite meaningful and often actionable,” he said.
Dr. Benowitz stressed the importance of engaging public health partners to discuss findings and actions, explaining that “what may look like a single-facility issue may have a very different perspective when you realize that there’s a similar issue at another facility elsewhere.”
“For all devices, it’s important to ensure adherence to the device reprocessing guidelines, “ he added, noting that these include a combination of facility protocols, manufacturer instructions for use, and guidance from organizations like the Food and Drug Administration and the CDC.
Dr. Benowitz reported having no disclosures.
[email protected]
SOURCE: Benowitz I et al. ICEID 2018, Oral Abstract Presentation E2.
REPORTING FROM ICEID 2018
Key clinical point: Medical devices can be reservoirs and transmission vectors for health care–associated infections.
Major finding: Of 285 consultations, 48 involved medical devices or device reprocessing.
Study details: A review of records from 285 consultations
Disclosures: Dr. Benowitz reported having no disclosures
Source: Benowitz I et al. ICEID 2018, Oral Abstract Presentation E2.
Alabama, Oregon, and pediatric asthma
according to estimates from the American Lung Association.
Oregon’s rate comes in at 557 per 10,000 population under the age of 18 years, just ahead of Montana at 574 per 10,000 and Iowa at 577. The prevalence of pediatric asthma in Alabama is 1,315 per 10,000, with North Carolina (1,149), Connecticut (1,107), Hawaii (1,026), and New York (1,005) joining it as members of the over-1,000 club. (MDedge News used the ALA’s estimates for persons under age 18 years with asthma in each state and Census Bureau estimates for population to calculate an unadjusted rate for each state.)
The ALA analysis was based on data from the Behavioral Risk Factor Behavioral System surveys for 2016 (31 states), 2015 (District of Columbia, Louisiana, New Hampshire, Texas), 2014 (Alabama, Maryland, North Carolina, Tennessee, West Virginia), 2012 (North Dakota and Wyoming), and 2011 (Iowa). National data were used for eight states (Alaska, Arkansas, Colorado, Delaware, Idaho, South Carolina, South Dakota, Virginia) that had no data available.
according to estimates from the American Lung Association.

Oregon’s rate comes in at 557 per 10,000 population under the age of 18 years, just ahead of Montana at 574 per 10,000 and Iowa at 577. The prevalence of pediatric asthma in Alabama is 1,315 per 10,000, with North Carolina (1,149), Connecticut (1,107), Hawaii (1,026), and New York (1,005) joining it as members of the over-1,000 club. (MDedge News used the ALA’s estimates for persons under age 18 years with asthma in each state and Census Bureau estimates for population to calculate an unadjusted rate for each state.)
The ALA analysis was based on data from the Behavioral Risk Factor Behavioral System surveys for 2016 (31 states), 2015 (District of Columbia, Louisiana, New Hampshire, Texas), 2014 (Alabama, Maryland, North Carolina, Tennessee, West Virginia), 2012 (North Dakota and Wyoming), and 2011 (Iowa). National data were used for eight states (Alaska, Arkansas, Colorado, Delaware, Idaho, South Carolina, South Dakota, Virginia) that had no data available.
according to estimates from the American Lung Association.

Oregon’s rate comes in at 557 per 10,000 population under the age of 18 years, just ahead of Montana at 574 per 10,000 and Iowa at 577. The prevalence of pediatric asthma in Alabama is 1,315 per 10,000, with North Carolina (1,149), Connecticut (1,107), Hawaii (1,026), and New York (1,005) joining it as members of the over-1,000 club. (MDedge News used the ALA’s estimates for persons under age 18 years with asthma in each state and Census Bureau estimates for population to calculate an unadjusted rate for each state.)
The ALA analysis was based on data from the Behavioral Risk Factor Behavioral System surveys for 2016 (31 states), 2015 (District of Columbia, Louisiana, New Hampshire, Texas), 2014 (Alabama, Maryland, North Carolina, Tennessee, West Virginia), 2012 (North Dakota and Wyoming), and 2011 (Iowa). National data were used for eight states (Alaska, Arkansas, Colorado, Delaware, Idaho, South Carolina, South Dakota, Virginia) that had no data available.