User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
Powered by CHEST Physician, Clinician Reviews, MDedge Family Medicine, Internal Medicine News, and The Journal of Clinical Outcomes Management.
Pertussis vaccine at birth shows immune response, tolerability
compared with a group receiving only the hepatitis B vaccine, a randomized clinical trial from Australia has found.
“These results indicate that a birth dose of aP vaccine is immunogenic in newborns and significantly narrows the immunity gap between birth and 14 days after receipt of DTaP at 6 or 8 weeks of age, marking the critical period when infants are most vulnerable to severe pertussis infection,” reported Nicholas Wood, PhD, of the National Centre for Immunisation Research and Surveillance of Vaccine Preventable Diseases in New South Wales, Australia, and his colleagues.
“Administration of the acellular pertussis vaccine at birth has the potential to reduce severe morbidity from Bordetella pertussis infection in the first 3 months of life, especially for infants of mothers who have not received a pertussis vaccine during pregnancy,” the researchers concluded in JAMA Pediatrics.
The researchers enrolled 417 infants from Sydney, Melbourne, Adelaide, and Perth between June 2010 and March 2013 and randomized them to receive either the hepatitis B vaccine alone (n = 205) or the hepatitis B vaccine with a monovalent acellular pertussis vaccine (n = 212) within the first 5 days after birth. The randomization was stratified for mothers’ receipt of the Tdap before pregnancy.
The Centers for Disease Control and Prevention currently recommends all newborns receive the hepatitis B vaccine shortly after birth and that pregnant women receive the Tdap vaccine during each pregnancy. There is not currently a monovalent acellular pertussis vaccine licensed in the United States.
The study infants then received the hexavalent DTaP-Hib-hep B-polio vaccine and the 10-valent pneumococcal conjugate vaccine at 6 weeks, 4 months, and 6 months.
The primary outcome was detectable levels of IgG antibody to pertussis toxin and pertactin at 10 weeks old.
Of the 206 infants receiving the pertussis vaccine at birth, 93% had detectable antibodies to pertussis toxin and pertactin at 10 weeks, compared with 51% of the 193 infants who received only the hepatitis B shot (P less than .001). Geometric mean concentration for pertussis toxin IgG also was four times higher in infants who received the pertussis vaccine at birth.
Adverse events were similar in the two groups both at birth and at 32 weeks, demonstrating that the pertussis birth dose is safe and tolerable.
“More important, in this study, the prevalence of fever after receipt of the birth dose, which can mistakenly be associated with potential sepsis and result in additional investigations in the neonatal period, was similar in both the group that received the aP vaccine at birth and the control group,” the authors reported.
A remaining question is the potential impact of maternal antibodies on protection from pertussis.
“The presence of maternal pertussis antibodies at birth can negatively affect postprimary responses to pertussis, diphtheria, and diphtheria-related CRM197 conjugate vaccines with a variety of infant immunization schedules and vaccines,” the authors noted. “The clinical significance of reductions in pertussis antibody related to maternal interference will require ongoing clinical evaluation, because there are no accepted serologic correlates of protection.”
The research was funded by a Australian National Health and Medical Research Council (NHMRC) grant, and several authors received NHMRC grants. One author also was supported by a Murdoch Children’s Research Institute Career Development Award. GlaxoSmithKline provided the vaccine and conducted the serologic assays. The authors reported having no conflicts of interest.
SOURCE: Wood N et al, JAMA Pediatr. 2018 Sep 10. doi: 10.1001/jamapediatrics.2018.2349.
Pertussis is most likely to cause morbidity or kill neonates between birth and when they are given their first pertussis vaccine at 6-8 weeks of age. This is well known.
In the current study giving the acellular pertussis (aP) vaccine at birth led to “significantly higher antibody titers to pertussis antigens at 10 weeks of age,” compared with those who did not receive it. Those infants who received the birth dose of aP vaccine also had higher pertussis antibodies at 6 weeks, whether or not their mothers had received Tdap within 5 years prior to delivery.
When this study began in 2009, maternal immunization was not a well accepted concept, but this attitude has changed, in part due to the safe vaccination of pregnant women with the pandemic flu vaccine. Despite this, Centers for Disease Control and Prevention 2016 data showed that only 49% of pregnant women in the United Stated received Tdap. These rates need to increase.
Administering the aP vaccine with the existing hepatitis B vaccine at birth to infants whose mothers who did not receive Tdap during pregnancy would be a practical solution, if the aP vaccine were universally available.
But the aP vaccine currently is not available in the United States and many other countries as a standalone vaccine, and the administration of DTaP as a birth dose has been linked with “significant immune interference.” The aP vaccine could have a place in countries where it is available, and there is no maternal immunization program. Otherwise, boosting maternal immunization appears to be the primary approach for now.
Kathryn M. Edwards, MD, is the Sarah H. Sell and Cornelius Vanderbilt Chair in Pediatrics at Vanderbilt University, Nashville. She specializes in pediatric infectious diseases. These comments are a summary of her editorial accompanying the article by Wood et al. (Pediatrics. 2018 Sep 10. doi: 10.1001/jamapediatrics.2018.2363). Dr. Edwards said she had no conflicts of interest.
Pertussis is most likely to cause morbidity or kill neonates between birth and when they are given their first pertussis vaccine at 6-8 weeks of age. This is well known.
In the current study giving the acellular pertussis (aP) vaccine at birth led to “significantly higher antibody titers to pertussis antigens at 10 weeks of age,” compared with those who did not receive it. Those infants who received the birth dose of aP vaccine also had higher pertussis antibodies at 6 weeks, whether or not their mothers had received Tdap within 5 years prior to delivery.
When this study began in 2009, maternal immunization was not a well accepted concept, but this attitude has changed, in part due to the safe vaccination of pregnant women with the pandemic flu vaccine. Despite this, Centers for Disease Control and Prevention 2016 data showed that only 49% of pregnant women in the United Stated received Tdap. These rates need to increase.
Administering the aP vaccine with the existing hepatitis B vaccine at birth to infants whose mothers who did not receive Tdap during pregnancy would be a practical solution, if the aP vaccine were universally available.
But the aP vaccine currently is not available in the United States and many other countries as a standalone vaccine, and the administration of DTaP as a birth dose has been linked with “significant immune interference.” The aP vaccine could have a place in countries where it is available, and there is no maternal immunization program. Otherwise, boosting maternal immunization appears to be the primary approach for now.
Kathryn M. Edwards, MD, is the Sarah H. Sell and Cornelius Vanderbilt Chair in Pediatrics at Vanderbilt University, Nashville. She specializes in pediatric infectious diseases. These comments are a summary of her editorial accompanying the article by Wood et al. (Pediatrics. 2018 Sep 10. doi: 10.1001/jamapediatrics.2018.2363). Dr. Edwards said she had no conflicts of interest.
Pertussis is most likely to cause morbidity or kill neonates between birth and when they are given their first pertussis vaccine at 6-8 weeks of age. This is well known.
In the current study giving the acellular pertussis (aP) vaccine at birth led to “significantly higher antibody titers to pertussis antigens at 10 weeks of age,” compared with those who did not receive it. Those infants who received the birth dose of aP vaccine also had higher pertussis antibodies at 6 weeks, whether or not their mothers had received Tdap within 5 years prior to delivery.
When this study began in 2009, maternal immunization was not a well accepted concept, but this attitude has changed, in part due to the safe vaccination of pregnant women with the pandemic flu vaccine. Despite this, Centers for Disease Control and Prevention 2016 data showed that only 49% of pregnant women in the United Stated received Tdap. These rates need to increase.
Administering the aP vaccine with the existing hepatitis B vaccine at birth to infants whose mothers who did not receive Tdap during pregnancy would be a practical solution, if the aP vaccine were universally available.
But the aP vaccine currently is not available in the United States and many other countries as a standalone vaccine, and the administration of DTaP as a birth dose has been linked with “significant immune interference.” The aP vaccine could have a place in countries where it is available, and there is no maternal immunization program. Otherwise, boosting maternal immunization appears to be the primary approach for now.
Kathryn M. Edwards, MD, is the Sarah H. Sell and Cornelius Vanderbilt Chair in Pediatrics at Vanderbilt University, Nashville. She specializes in pediatric infectious diseases. These comments are a summary of her editorial accompanying the article by Wood et al. (Pediatrics. 2018 Sep 10. doi: 10.1001/jamapediatrics.2018.2363). Dr. Edwards said she had no conflicts of interest.
compared with a group receiving only the hepatitis B vaccine, a randomized clinical trial from Australia has found.
“These results indicate that a birth dose of aP vaccine is immunogenic in newborns and significantly narrows the immunity gap between birth and 14 days after receipt of DTaP at 6 or 8 weeks of age, marking the critical period when infants are most vulnerable to severe pertussis infection,” reported Nicholas Wood, PhD, of the National Centre for Immunisation Research and Surveillance of Vaccine Preventable Diseases in New South Wales, Australia, and his colleagues.
“Administration of the acellular pertussis vaccine at birth has the potential to reduce severe morbidity from Bordetella pertussis infection in the first 3 months of life, especially for infants of mothers who have not received a pertussis vaccine during pregnancy,” the researchers concluded in JAMA Pediatrics.
The researchers enrolled 417 infants from Sydney, Melbourne, Adelaide, and Perth between June 2010 and March 2013 and randomized them to receive either the hepatitis B vaccine alone (n = 205) or the hepatitis B vaccine with a monovalent acellular pertussis vaccine (n = 212) within the first 5 days after birth. The randomization was stratified for mothers’ receipt of the Tdap before pregnancy.
The Centers for Disease Control and Prevention currently recommends all newborns receive the hepatitis B vaccine shortly after birth and that pregnant women receive the Tdap vaccine during each pregnancy. There is not currently a monovalent acellular pertussis vaccine licensed in the United States.
The study infants then received the hexavalent DTaP-Hib-hep B-polio vaccine and the 10-valent pneumococcal conjugate vaccine at 6 weeks, 4 months, and 6 months.
The primary outcome was detectable levels of IgG antibody to pertussis toxin and pertactin at 10 weeks old.
Of the 206 infants receiving the pertussis vaccine at birth, 93% had detectable antibodies to pertussis toxin and pertactin at 10 weeks, compared with 51% of the 193 infants who received only the hepatitis B shot (P less than .001). Geometric mean concentration for pertussis toxin IgG also was four times higher in infants who received the pertussis vaccine at birth.
Adverse events were similar in the two groups both at birth and at 32 weeks, demonstrating that the pertussis birth dose is safe and tolerable.
“More important, in this study, the prevalence of fever after receipt of the birth dose, which can mistakenly be associated with potential sepsis and result in additional investigations in the neonatal period, was similar in both the group that received the aP vaccine at birth and the control group,” the authors reported.
A remaining question is the potential impact of maternal antibodies on protection from pertussis.
“The presence of maternal pertussis antibodies at birth can negatively affect postprimary responses to pertussis, diphtheria, and diphtheria-related CRM197 conjugate vaccines with a variety of infant immunization schedules and vaccines,” the authors noted. “The clinical significance of reductions in pertussis antibody related to maternal interference will require ongoing clinical evaluation, because there are no accepted serologic correlates of protection.”
The research was funded by a Australian National Health and Medical Research Council (NHMRC) grant, and several authors received NHMRC grants. One author also was supported by a Murdoch Children’s Research Institute Career Development Award. GlaxoSmithKline provided the vaccine and conducted the serologic assays. The authors reported having no conflicts of interest.
SOURCE: Wood N et al, JAMA Pediatr. 2018 Sep 10. doi: 10.1001/jamapediatrics.2018.2349.
compared with a group receiving only the hepatitis B vaccine, a randomized clinical trial from Australia has found.
“These results indicate that a birth dose of aP vaccine is immunogenic in newborns and significantly narrows the immunity gap between birth and 14 days after receipt of DTaP at 6 or 8 weeks of age, marking the critical period when infants are most vulnerable to severe pertussis infection,” reported Nicholas Wood, PhD, of the National Centre for Immunisation Research and Surveillance of Vaccine Preventable Diseases in New South Wales, Australia, and his colleagues.
“Administration of the acellular pertussis vaccine at birth has the potential to reduce severe morbidity from Bordetella pertussis infection in the first 3 months of life, especially for infants of mothers who have not received a pertussis vaccine during pregnancy,” the researchers concluded in JAMA Pediatrics.
The researchers enrolled 417 infants from Sydney, Melbourne, Adelaide, and Perth between June 2010 and March 2013 and randomized them to receive either the hepatitis B vaccine alone (n = 205) or the hepatitis B vaccine with a monovalent acellular pertussis vaccine (n = 212) within the first 5 days after birth. The randomization was stratified for mothers’ receipt of the Tdap before pregnancy.
The Centers for Disease Control and Prevention currently recommends all newborns receive the hepatitis B vaccine shortly after birth and that pregnant women receive the Tdap vaccine during each pregnancy. There is not currently a monovalent acellular pertussis vaccine licensed in the United States.
The study infants then received the hexavalent DTaP-Hib-hep B-polio vaccine and the 10-valent pneumococcal conjugate vaccine at 6 weeks, 4 months, and 6 months.
The primary outcome was detectable levels of IgG antibody to pertussis toxin and pertactin at 10 weeks old.
Of the 206 infants receiving the pertussis vaccine at birth, 93% had detectable antibodies to pertussis toxin and pertactin at 10 weeks, compared with 51% of the 193 infants who received only the hepatitis B shot (P less than .001). Geometric mean concentration for pertussis toxin IgG also was four times higher in infants who received the pertussis vaccine at birth.
Adverse events were similar in the two groups both at birth and at 32 weeks, demonstrating that the pertussis birth dose is safe and tolerable.
“More important, in this study, the prevalence of fever after receipt of the birth dose, which can mistakenly be associated with potential sepsis and result in additional investigations in the neonatal period, was similar in both the group that received the aP vaccine at birth and the control group,” the authors reported.
A remaining question is the potential impact of maternal antibodies on protection from pertussis.
“The presence of maternal pertussis antibodies at birth can negatively affect postprimary responses to pertussis, diphtheria, and diphtheria-related CRM197 conjugate vaccines with a variety of infant immunization schedules and vaccines,” the authors noted. “The clinical significance of reductions in pertussis antibody related to maternal interference will require ongoing clinical evaluation, because there are no accepted serologic correlates of protection.”
The research was funded by a Australian National Health and Medical Research Council (NHMRC) grant, and several authors received NHMRC grants. One author also was supported by a Murdoch Children’s Research Institute Career Development Award. GlaxoSmithKline provided the vaccine and conducted the serologic assays. The authors reported having no conflicts of interest.
SOURCE: Wood N et al, JAMA Pediatr. 2018 Sep 10. doi: 10.1001/jamapediatrics.2018.2349.
FROM JAMA PEDIATRICS
Key clinical point: A monovalent acellular pertussis vaccine dose at birth appears safe, tolerable, and effective.
Major finding: 93% of 212 newborns receiving an acellular pertussis vaccine at birth showed antibodies against pertussis toxin and pertactin at 10 weeks, compared with 51% of 205 newborns without the birth dose.
Study details: The findings are based on a randomized controlled trial involving 417 healthy term newborns in four Australian cities from June 2010 to March 2013.
Disclosures: The research was funded by an Australian National Health and Medical Research Council (NHMRC) grant, and several authors received NHMRC grants. One author also was supported by a Murdoch Children’s Research Institute Career Development Award. GlaxoSmithKline provided the vaccine and conducted the serologic assays. The authors reporting having no conflicts of interest.
Source: Wood N et al. JAMA Pediatr. 2018 Sep. 10. doi: 10.1001/jamapediatrics.2018.2349.
Single-dose influenza drug baloxavir similar to oseltamivir in efficacy
A new single-dose influenza antiviral drug appears significantly better than placebo at relieving the symptoms of infection, and reduces viral load faster than does oseltamivir, new research suggests.
Baloxavir marboxil – a selective inhibitor of influenza cap-dependent endonuclease – was tested in two randomized, double-blind, controlled trials. The first was a double-blind, placebo-controlled, dose-ranging, phase 2 randomized trial of 389 Japanese adults aged 20-64 years with acute uncomplicated influenza from December 2015 through March 2016. The second was a phase 3 randomized controlled trial of 1,366 patients comparing baloxavir with placebo and oseltamivir.
The phase 2 study showed patients treated with 10 mg, 20 mg or 40 mg oral dose of baloxavir experienced a significantly shorter median time to symptom alleviation compared with placebo (54.2, 51, 49.5, and 77.7 hours, respectively), according to a paper published in the Sept. 6 edition of the New England Journal of Medicine.
In addition, all three doses showed significantly greater reductions in influenza virus titers on days 2 and 3, compared with placebo.
The phase 3 trial CAPSTONE-1 (NCT02954354) was a double-blind, placebo- and oseltamivir-controlled, randomized trial that enrolled outpatients aged 12-64 years with influenza-like illness in the United States and Japan from December 2016 through March 2017. Patients aged 20-64 years received a single, weight-based oral dose of baloxavir (40 mg for patients weighing more than 80 kg, 80 mg for those weighing 80 kg or less) on day 1 only or oseltamivir at a dose of 75 mg twice daily or matching placebos on a 5-day regimen.
Patients aged 12-19 years were randomly assigned to receive either baloxavir or placebo on day 1 only, according to the researchers.
The median time to alleviation of symptoms was similar in the baloxavir (53.5 hours) and oseltamivir group (53.8 hours). However, patients taking baloxavir had significantly faster declines in infectious viral load compared with those taking oseltamivir, which was taken as a 75-mg dose twice daily for 5 days. In addition, patients who were treated with baloxavir within 24 hours of symptom onset showed significantly shorter time to alleviation of symptoms compared with placebo than did those who started treatment more than 24 hours after symptoms began.
Adverse events related to the study drug were more common among patients taking oseltamivir (8.4%) compared with those taking baloxavir (4.4%) or placebo (3.9%). In the phase 2 study, the adverse event rate was lower in the three baloxavir dosage groups compared with the placebo group. The study also showed a similar low frequency of complications requiring antibiotic treatment in both the baloxavir, oseltamivir, and placebo arms.
Some patients did show evidence of decreased susceptibility to baloxavir; for example, PA I38T/M amino acid substitutions were seen in 9.7% of the patients taking baloxavir but none of randomly selected patients in the placebo group of the phase 3 trial.
“These trials showed that single doses of the cap-dependent endonuclease inhibitor baloxavir were superior to placebo in alleviating influenza symptoms in patients with uncomplicated influenza, without clinically significant side effects,” wrote Dr. Frederick G. Hayden of the University of Virginia, Charlottesville, and his coauthors.
“The antiviral effects that were observed with baloxavir in patients with uncomplicated influenza provide encouragement with respect to its potential value in treating complicated or severe influenza infections,” they noted.
Because the treatment was inhibitory for influenza virus strains that were resistant to neuraminidase inhibitors or M2 ion-channel inhibitors, it could be a treatment option for patients infected with those viruses, the researchers added.
CAPSTONE-2, a randomized, controlled trial involving patients at high risk for influenza complications (NCT02949011) is in progress.
The study was supported by Shionogi, which developed baloxavir. Seven authors declared fees from the pharmaceutical industry, including Shionogi. Six authors were employees of Shionogi, one also holding stock. No other conflicts of interest were declared.
SOURCE: Hayden F et al. N Engl J Med. 2018;379:913-23. doi: 10.1056/NEJMoa1716197.
These two studies of baloxavir show that the drug has a clinical benefit similar to that of oseltamivir in individuals with uncomplicated influenza infection. As a single-dose treatment, baloxavir has the advantage in reducing concerns about adherence compared to the treatment regimen for oseltamivir, which requires 5 days of twice-daily dosing.
However, these studies should be viewed as the first step. While baloxavir showed significantly greater reductions in viral load at 24 hours and a shorter duration of infectious virus detection than did oseltamivir or placebo, it also induced the emergence of viral escape mutants with reduced susceptibility.
It’s not yet known whether these influenza viruses with reduced susceptibility are transmissible, and whether surveillance for I38T and other markers will be needed. We also need trials to identify which patients are most likely to benefit from baloxavir, and the timing for treatment.
Timothy M. Uyeki, MD, is with the Influenza Division at the National Center for Immunization and Respiratory Diseases, Centers for Disease Control and Prevention. These comments are taken from an editorial (N Engl J Med. 2018;397:975-7. doi: 10.1056/NEJMe1810815. No conflicts of interest were declared.
These two studies of baloxavir show that the drug has a clinical benefit similar to that of oseltamivir in individuals with uncomplicated influenza infection. As a single-dose treatment, baloxavir has the advantage in reducing concerns about adherence compared to the treatment regimen for oseltamivir, which requires 5 days of twice-daily dosing.
However, these studies should be viewed as the first step. While baloxavir showed significantly greater reductions in viral load at 24 hours and a shorter duration of infectious virus detection than did oseltamivir or placebo, it also induced the emergence of viral escape mutants with reduced susceptibility.
It’s not yet known whether these influenza viruses with reduced susceptibility are transmissible, and whether surveillance for I38T and other markers will be needed. We also need trials to identify which patients are most likely to benefit from baloxavir, and the timing for treatment.
Timothy M. Uyeki, MD, is with the Influenza Division at the National Center for Immunization and Respiratory Diseases, Centers for Disease Control and Prevention. These comments are taken from an editorial (N Engl J Med. 2018;397:975-7. doi: 10.1056/NEJMe1810815. No conflicts of interest were declared.
These two studies of baloxavir show that the drug has a clinical benefit similar to that of oseltamivir in individuals with uncomplicated influenza infection. As a single-dose treatment, baloxavir has the advantage in reducing concerns about adherence compared to the treatment regimen for oseltamivir, which requires 5 days of twice-daily dosing.
However, these studies should be viewed as the first step. While baloxavir showed significantly greater reductions in viral load at 24 hours and a shorter duration of infectious virus detection than did oseltamivir or placebo, it also induced the emergence of viral escape mutants with reduced susceptibility.
It’s not yet known whether these influenza viruses with reduced susceptibility are transmissible, and whether surveillance for I38T and other markers will be needed. We also need trials to identify which patients are most likely to benefit from baloxavir, and the timing for treatment.
Timothy M. Uyeki, MD, is with the Influenza Division at the National Center for Immunization and Respiratory Diseases, Centers for Disease Control and Prevention. These comments are taken from an editorial (N Engl J Med. 2018;397:975-7. doi: 10.1056/NEJMe1810815. No conflicts of interest were declared.
A new single-dose influenza antiviral drug appears significantly better than placebo at relieving the symptoms of infection, and reduces viral load faster than does oseltamivir, new research suggests.
Baloxavir marboxil – a selective inhibitor of influenza cap-dependent endonuclease – was tested in two randomized, double-blind, controlled trials. The first was a double-blind, placebo-controlled, dose-ranging, phase 2 randomized trial of 389 Japanese adults aged 20-64 years with acute uncomplicated influenza from December 2015 through March 2016. The second was a phase 3 randomized controlled trial of 1,366 patients comparing baloxavir with placebo and oseltamivir.
The phase 2 study showed patients treated with 10 mg, 20 mg or 40 mg oral dose of baloxavir experienced a significantly shorter median time to symptom alleviation compared with placebo (54.2, 51, 49.5, and 77.7 hours, respectively), according to a paper published in the Sept. 6 edition of the New England Journal of Medicine.
In addition, all three doses showed significantly greater reductions in influenza virus titers on days 2 and 3, compared with placebo.
The phase 3 trial CAPSTONE-1 (NCT02954354) was a double-blind, placebo- and oseltamivir-controlled, randomized trial that enrolled outpatients aged 12-64 years with influenza-like illness in the United States and Japan from December 2016 through March 2017. Patients aged 20-64 years received a single, weight-based oral dose of baloxavir (40 mg for patients weighing more than 80 kg, 80 mg for those weighing 80 kg or less) on day 1 only or oseltamivir at a dose of 75 mg twice daily or matching placebos on a 5-day regimen.
Patients aged 12-19 years were randomly assigned to receive either baloxavir or placebo on day 1 only, according to the researchers.
The median time to alleviation of symptoms was similar in the baloxavir (53.5 hours) and oseltamivir group (53.8 hours). However, patients taking baloxavir had significantly faster declines in infectious viral load compared with those taking oseltamivir, which was taken as a 75-mg dose twice daily for 5 days. In addition, patients who were treated with baloxavir within 24 hours of symptom onset showed significantly shorter time to alleviation of symptoms compared with placebo than did those who started treatment more than 24 hours after symptoms began.
Adverse events related to the study drug were more common among patients taking oseltamivir (8.4%) compared with those taking baloxavir (4.4%) or placebo (3.9%). In the phase 2 study, the adverse event rate was lower in the three baloxavir dosage groups compared with the placebo group. The study also showed a similar low frequency of complications requiring antibiotic treatment in both the baloxavir, oseltamivir, and placebo arms.
Some patients did show evidence of decreased susceptibility to baloxavir; for example, PA I38T/M amino acid substitutions were seen in 9.7% of the patients taking baloxavir but none of randomly selected patients in the placebo group of the phase 3 trial.
“These trials showed that single doses of the cap-dependent endonuclease inhibitor baloxavir were superior to placebo in alleviating influenza symptoms in patients with uncomplicated influenza, without clinically significant side effects,” wrote Dr. Frederick G. Hayden of the University of Virginia, Charlottesville, and his coauthors.
“The antiviral effects that were observed with baloxavir in patients with uncomplicated influenza provide encouragement with respect to its potential value in treating complicated or severe influenza infections,” they noted.
Because the treatment was inhibitory for influenza virus strains that were resistant to neuraminidase inhibitors or M2 ion-channel inhibitors, it could be a treatment option for patients infected with those viruses, the researchers added.
CAPSTONE-2, a randomized, controlled trial involving patients at high risk for influenza complications (NCT02949011) is in progress.
The study was supported by Shionogi, which developed baloxavir. Seven authors declared fees from the pharmaceutical industry, including Shionogi. Six authors were employees of Shionogi, one also holding stock. No other conflicts of interest were declared.
SOURCE: Hayden F et al. N Engl J Med. 2018;379:913-23. doi: 10.1056/NEJMoa1716197.
A new single-dose influenza antiviral drug appears significantly better than placebo at relieving the symptoms of infection, and reduces viral load faster than does oseltamivir, new research suggests.
Baloxavir marboxil – a selective inhibitor of influenza cap-dependent endonuclease – was tested in two randomized, double-blind, controlled trials. The first was a double-blind, placebo-controlled, dose-ranging, phase 2 randomized trial of 389 Japanese adults aged 20-64 years with acute uncomplicated influenza from December 2015 through March 2016. The second was a phase 3 randomized controlled trial of 1,366 patients comparing baloxavir with placebo and oseltamivir.
The phase 2 study showed patients treated with 10 mg, 20 mg or 40 mg oral dose of baloxavir experienced a significantly shorter median time to symptom alleviation compared with placebo (54.2, 51, 49.5, and 77.7 hours, respectively), according to a paper published in the Sept. 6 edition of the New England Journal of Medicine.
In addition, all three doses showed significantly greater reductions in influenza virus titers on days 2 and 3, compared with placebo.
The phase 3 trial CAPSTONE-1 (NCT02954354) was a double-blind, placebo- and oseltamivir-controlled, randomized trial that enrolled outpatients aged 12-64 years with influenza-like illness in the United States and Japan from December 2016 through March 2017. Patients aged 20-64 years received a single, weight-based oral dose of baloxavir (40 mg for patients weighing more than 80 kg, 80 mg for those weighing 80 kg or less) on day 1 only or oseltamivir at a dose of 75 mg twice daily or matching placebos on a 5-day regimen.
Patients aged 12-19 years were randomly assigned to receive either baloxavir or placebo on day 1 only, according to the researchers.
The median time to alleviation of symptoms was similar in the baloxavir (53.5 hours) and oseltamivir group (53.8 hours). However, patients taking baloxavir had significantly faster declines in infectious viral load compared with those taking oseltamivir, which was taken as a 75-mg dose twice daily for 5 days. In addition, patients who were treated with baloxavir within 24 hours of symptom onset showed significantly shorter time to alleviation of symptoms compared with placebo than did those who started treatment more than 24 hours after symptoms began.
Adverse events related to the study drug were more common among patients taking oseltamivir (8.4%) compared with those taking baloxavir (4.4%) or placebo (3.9%). In the phase 2 study, the adverse event rate was lower in the three baloxavir dosage groups compared with the placebo group. The study also showed a similar low frequency of complications requiring antibiotic treatment in both the baloxavir, oseltamivir, and placebo arms.
Some patients did show evidence of decreased susceptibility to baloxavir; for example, PA I38T/M amino acid substitutions were seen in 9.7% of the patients taking baloxavir but none of randomly selected patients in the placebo group of the phase 3 trial.
“These trials showed that single doses of the cap-dependent endonuclease inhibitor baloxavir were superior to placebo in alleviating influenza symptoms in patients with uncomplicated influenza, without clinically significant side effects,” wrote Dr. Frederick G. Hayden of the University of Virginia, Charlottesville, and his coauthors.
“The antiviral effects that were observed with baloxavir in patients with uncomplicated influenza provide encouragement with respect to its potential value in treating complicated or severe influenza infections,” they noted.
Because the treatment was inhibitory for influenza virus strains that were resistant to neuraminidase inhibitors or M2 ion-channel inhibitors, it could be a treatment option for patients infected with those viruses, the researchers added.
CAPSTONE-2, a randomized, controlled trial involving patients at high risk for influenza complications (NCT02949011) is in progress.
The study was supported by Shionogi, which developed baloxavir. Seven authors declared fees from the pharmaceutical industry, including Shionogi. Six authors were employees of Shionogi, one also holding stock. No other conflicts of interest were declared.
SOURCE: Hayden F et al. N Engl J Med. 2018;379:913-23. doi: 10.1056/NEJMoa1716197.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: Single-dose influenza antiviral baloxavir shows efficacy similar to that of oseltamivir.
Major finding: Baloxavir shows similar time to alleviation of influenza symptoms compared with oseltamivir, but greater reductions in viral load.
Study details: Phase 2 and phase 3 randomized controlled trials in 389 and 1,366 otherwise healthy patients with influenza.
Disclosures: The study was supported by Shionogi, which developed baloxavir. Seven authors declared fees from the pharmaceutical industry, including Shionogi. Six authors were employees of Shionogi, one also holding stock. No other conflicts of interest were declared.
Source: Hayden F et al. N Engl J Med 2018;379:913-23. doi: 10.1056/NEJMoa1716197.
Service, please: Hospital setting matters for pneumonia
the National Center for Health Statistics (NCHS) reported.
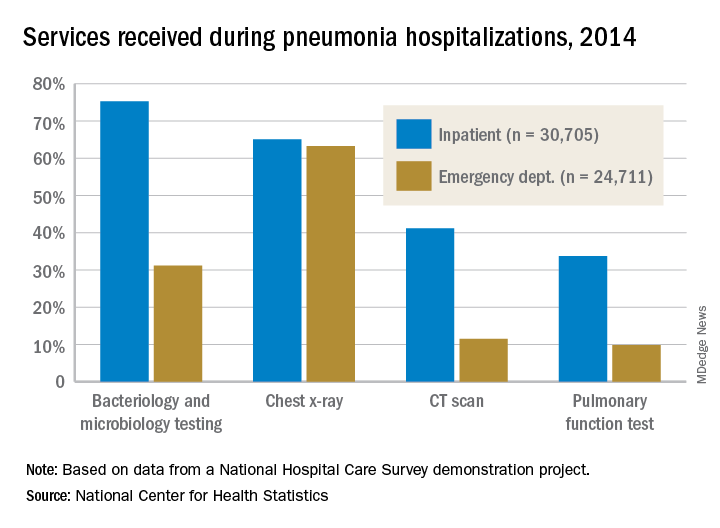
The percentages were not as close, however, for other diagnostic services. Inpatient stays were much more likely than ED encounters to involve bacteriology and microbiology testing (75.3% vs. 31.2%), CT scans (41.2% vs. 11.5%), and pulmonary function tests (33.7% vs. 9.8%), investigators from the NCHS said.
The age distribution of the two patient populations also were quite different, with those aged 65 years and older making up the largest share (46%) of pneumonia inpatients and the 15-and-under group representing the largest proportion (47%) of ED visits. For the inpatient setting, the smallest age group was those aged 15-44 years (10%), and for the ED it was those aged 65 years and older (14%), they reported.
The National Hospital Care Survey “is not yet nationally representative,” the NCHS investigators wrote – the overall sample for 2014 consisted of 581 hospitals – but “the number of encounters and the inclusion of [personally identifiable information] allow an example of analysis that was not previously possible.”
the National Center for Health Statistics (NCHS) reported.

The percentages were not as close, however, for other diagnostic services. Inpatient stays were much more likely than ED encounters to involve bacteriology and microbiology testing (75.3% vs. 31.2%), CT scans (41.2% vs. 11.5%), and pulmonary function tests (33.7% vs. 9.8%), investigators from the NCHS said.
The age distribution of the two patient populations also were quite different, with those aged 65 years and older making up the largest share (46%) of pneumonia inpatients and the 15-and-under group representing the largest proportion (47%) of ED visits. For the inpatient setting, the smallest age group was those aged 15-44 years (10%), and for the ED it was those aged 65 years and older (14%), they reported.
The National Hospital Care Survey “is not yet nationally representative,” the NCHS investigators wrote – the overall sample for 2014 consisted of 581 hospitals – but “the number of encounters and the inclusion of [personally identifiable information] allow an example of analysis that was not previously possible.”
the National Center for Health Statistics (NCHS) reported.

The percentages were not as close, however, for other diagnostic services. Inpatient stays were much more likely than ED encounters to involve bacteriology and microbiology testing (75.3% vs. 31.2%), CT scans (41.2% vs. 11.5%), and pulmonary function tests (33.7% vs. 9.8%), investigators from the NCHS said.
The age distribution of the two patient populations also were quite different, with those aged 65 years and older making up the largest share (46%) of pneumonia inpatients and the 15-and-under group representing the largest proportion (47%) of ED visits. For the inpatient setting, the smallest age group was those aged 15-44 years (10%), and for the ED it was those aged 65 years and older (14%), they reported.
The National Hospital Care Survey “is not yet nationally representative,” the NCHS investigators wrote – the overall sample for 2014 consisted of 581 hospitals – but “the number of encounters and the inclusion of [personally identifiable information] allow an example of analysis that was not previously possible.”
Community-based therapy improved asthma outcomes in African American teens
, according to results published in Pediatrics.
In a study of 167 African American patients aged 12-16 years, the 84 randomly assigned to Multisystemic Therapy–Health Care (MST-HC) had greater improvement in forced expiratory volume in 1 second (FEV1) over time, compared with the 83 patients randomly assigned to family support (FS) therapy (beta = 0.097, t[164.27] = 2.52; P = .01). Improvements in secondary outcomes also were observed in this group, reported Sylvie Naar, PhD, of Florida State University, Tallahassee, and her coauthors.
They studied African American adolescents with moderate to severe persistent asthma who resided in a home setting with a caregiver and were at high risk for poorly controlled asthma. Families were randomized to either MST-HC (84 patients) or FS (83 patients) based on severity of urgent care use, and follow-up was completed 7 and 12 months after baseline assessment. Families were paid $50 for each assessment.
FEV1 was the primary outcome. Secondary outcomes were medication adherence, symptom severity and frequency, inpatient hospitalizations, and ED visits. Medication adherence was evaluated via the Family Asthma Management System Scale (FAMSS) and the Daily Phone Diary (DPD). Other outcomes were confirmed via medical records.
Patients in the FS control group received weekly home-based counseling for up to 6 months. Patients in the MST-HC treatment group were first engaged in a motivational session with a therapist and evaluated for asthma management with interviews and observations within the home and community. Once possible contributing factors to poor asthma management (such as medication underuse or low parental monitoring) were identified, targeted interventions such as skills training, behavioral and family therapy, or communication training with school and medical staff were chosen, and treatment goals continually monitored and modified, the authors said.
The mean length of treatment until termination in the MST-HC group was 5 months, and the mean number of sessions was 27. In the FS group, mean length of treatment was 4 months, and the mean number of sessions was 11.
FEV1 for the MST-HC group improved from 2.05 at baseline to 2.25 at 7 months (a 10% improvement), and to 2.37 (a 16% improvement) at 12 months, compared with an improvement from 2.21 to 2.31 at 7 months (a 4% improvement) and 2.33 (a 5% improvement) at 12 months in the control group, the authors reported.
At 12 months, FAMSS adherence scores improved from 4.19 to 5.24 in the MST-HC group and from 4.61 to 4.72 in the control group.
DPD adherence scores improved from a mean of 0.33 at baseline to 0.69 for the MST-HC group, and from 0.43 to 0.46 in the FS group.
At 12 months, the mean frequency of asthma symptoms in the MST-HC group improved from 2.75 at baseline to 1.43, compared with a decline of 2.67 to 2.58 in the control group. The mean number of hospitalizations in the MST-HC group improved from 0.87 to 0.24, compared with a change from 0.66 to 0.34 in the control group.
The study results are “especially noteworthy because African American adolescents experience greater morbidity and mortality from asthma than white adolescents even when controlling for socioeconomic variables,” Dr. Naar and her associates wrote. Future research should focus on the “transportability” of MST-HC treatment to community settings, which is “ready to be studied in effectiveness and implementation trials.”
The study was supported by a National Institutes of Health grant. Coauthor Phillippe Cunningham, PhD, is a co-owner of Evidence-Based Services, a network partner organization that is licensed to disseminate Multisystemic Therapy for drug court and juvenile delinquency settings. The other authors said they have no potential conflicts of interest.
SOURCE: Naar S et al. Pediatrics. 2018. doi: 10.1542/peds.2017-3737.
, according to results published in Pediatrics.
In a study of 167 African American patients aged 12-16 years, the 84 randomly assigned to Multisystemic Therapy–Health Care (MST-HC) had greater improvement in forced expiratory volume in 1 second (FEV1) over time, compared with the 83 patients randomly assigned to family support (FS) therapy (beta = 0.097, t[164.27] = 2.52; P = .01). Improvements in secondary outcomes also were observed in this group, reported Sylvie Naar, PhD, of Florida State University, Tallahassee, and her coauthors.
They studied African American adolescents with moderate to severe persistent asthma who resided in a home setting with a caregiver and were at high risk for poorly controlled asthma. Families were randomized to either MST-HC (84 patients) or FS (83 patients) based on severity of urgent care use, and follow-up was completed 7 and 12 months after baseline assessment. Families were paid $50 for each assessment.
FEV1 was the primary outcome. Secondary outcomes were medication adherence, symptom severity and frequency, inpatient hospitalizations, and ED visits. Medication adherence was evaluated via the Family Asthma Management System Scale (FAMSS) and the Daily Phone Diary (DPD). Other outcomes were confirmed via medical records.
Patients in the FS control group received weekly home-based counseling for up to 6 months. Patients in the MST-HC treatment group were first engaged in a motivational session with a therapist and evaluated for asthma management with interviews and observations within the home and community. Once possible contributing factors to poor asthma management (such as medication underuse or low parental monitoring) were identified, targeted interventions such as skills training, behavioral and family therapy, or communication training with school and medical staff were chosen, and treatment goals continually monitored and modified, the authors said.
The mean length of treatment until termination in the MST-HC group was 5 months, and the mean number of sessions was 27. In the FS group, mean length of treatment was 4 months, and the mean number of sessions was 11.
FEV1 for the MST-HC group improved from 2.05 at baseline to 2.25 at 7 months (a 10% improvement), and to 2.37 (a 16% improvement) at 12 months, compared with an improvement from 2.21 to 2.31 at 7 months (a 4% improvement) and 2.33 (a 5% improvement) at 12 months in the control group, the authors reported.
At 12 months, FAMSS adherence scores improved from 4.19 to 5.24 in the MST-HC group and from 4.61 to 4.72 in the control group.
DPD adherence scores improved from a mean of 0.33 at baseline to 0.69 for the MST-HC group, and from 0.43 to 0.46 in the FS group.
At 12 months, the mean frequency of asthma symptoms in the MST-HC group improved from 2.75 at baseline to 1.43, compared with a decline of 2.67 to 2.58 in the control group. The mean number of hospitalizations in the MST-HC group improved from 0.87 to 0.24, compared with a change from 0.66 to 0.34 in the control group.
The study results are “especially noteworthy because African American adolescents experience greater morbidity and mortality from asthma than white adolescents even when controlling for socioeconomic variables,” Dr. Naar and her associates wrote. Future research should focus on the “transportability” of MST-HC treatment to community settings, which is “ready to be studied in effectiveness and implementation trials.”
The study was supported by a National Institutes of Health grant. Coauthor Phillippe Cunningham, PhD, is a co-owner of Evidence-Based Services, a network partner organization that is licensed to disseminate Multisystemic Therapy for drug court and juvenile delinquency settings. The other authors said they have no potential conflicts of interest.
SOURCE: Naar S et al. Pediatrics. 2018. doi: 10.1542/peds.2017-3737.
, according to results published in Pediatrics.
In a study of 167 African American patients aged 12-16 years, the 84 randomly assigned to Multisystemic Therapy–Health Care (MST-HC) had greater improvement in forced expiratory volume in 1 second (FEV1) over time, compared with the 83 patients randomly assigned to family support (FS) therapy (beta = 0.097, t[164.27] = 2.52; P = .01). Improvements in secondary outcomes also were observed in this group, reported Sylvie Naar, PhD, of Florida State University, Tallahassee, and her coauthors.
They studied African American adolescents with moderate to severe persistent asthma who resided in a home setting with a caregiver and were at high risk for poorly controlled asthma. Families were randomized to either MST-HC (84 patients) or FS (83 patients) based on severity of urgent care use, and follow-up was completed 7 and 12 months after baseline assessment. Families were paid $50 for each assessment.
FEV1 was the primary outcome. Secondary outcomes were medication adherence, symptom severity and frequency, inpatient hospitalizations, and ED visits. Medication adherence was evaluated via the Family Asthma Management System Scale (FAMSS) and the Daily Phone Diary (DPD). Other outcomes were confirmed via medical records.
Patients in the FS control group received weekly home-based counseling for up to 6 months. Patients in the MST-HC treatment group were first engaged in a motivational session with a therapist and evaluated for asthma management with interviews and observations within the home and community. Once possible contributing factors to poor asthma management (such as medication underuse or low parental monitoring) were identified, targeted interventions such as skills training, behavioral and family therapy, or communication training with school and medical staff were chosen, and treatment goals continually monitored and modified, the authors said.
The mean length of treatment until termination in the MST-HC group was 5 months, and the mean number of sessions was 27. In the FS group, mean length of treatment was 4 months, and the mean number of sessions was 11.
FEV1 for the MST-HC group improved from 2.05 at baseline to 2.25 at 7 months (a 10% improvement), and to 2.37 (a 16% improvement) at 12 months, compared with an improvement from 2.21 to 2.31 at 7 months (a 4% improvement) and 2.33 (a 5% improvement) at 12 months in the control group, the authors reported.
At 12 months, FAMSS adherence scores improved from 4.19 to 5.24 in the MST-HC group and from 4.61 to 4.72 in the control group.
DPD adherence scores improved from a mean of 0.33 at baseline to 0.69 for the MST-HC group, and from 0.43 to 0.46 in the FS group.
At 12 months, the mean frequency of asthma symptoms in the MST-HC group improved from 2.75 at baseline to 1.43, compared with a decline of 2.67 to 2.58 in the control group. The mean number of hospitalizations in the MST-HC group improved from 0.87 to 0.24, compared with a change from 0.66 to 0.34 in the control group.
The study results are “especially noteworthy because African American adolescents experience greater morbidity and mortality from asthma than white adolescents even when controlling for socioeconomic variables,” Dr. Naar and her associates wrote. Future research should focus on the “transportability” of MST-HC treatment to community settings, which is “ready to be studied in effectiveness and implementation trials.”
The study was supported by a National Institutes of Health grant. Coauthor Phillippe Cunningham, PhD, is a co-owner of Evidence-Based Services, a network partner organization that is licensed to disseminate Multisystemic Therapy for drug court and juvenile delinquency settings. The other authors said they have no potential conflicts of interest.
SOURCE: Naar S et al. Pediatrics. 2018. doi: 10.1542/peds.2017-3737.
FROM PEDIATRICS
Key clinical point: Multisystemic Therapy–Health Care (MST-HC) significantly improved outcomes in African American adolescents with moderate to severe asthma.
Major finding: Patients randomly assigned to MST-HC treatment had greater improvement in FEV1 over time, compared with controls (beta = 0.097; t(164.27) = 2.52; P = .01).
Study details: A study of 167 African American patients aged 12-16 years, randomly assigned to either MST-HC or FS.
Disclosures: The study was supported by a National Institutes of Health grant. Coauthor Phillippe Cunningham, PhD, is a co-owner of Evidence-Based Services, a network partner organization that is licensed to disseminate multisystemic therapy for drug court and juvenile delinquency settings. The other authors said they have no potential conflicts of interest.
Source: Naar S et al. Pediatrics. 2018. doi: 10.1542/peds.2017-3737.
E-cigarette use highest among adults aged under 35 years
Almost 11 million adults use e-cigarettes in the United States, and the majority are under the age of 35 years, according to the American Heart Association.
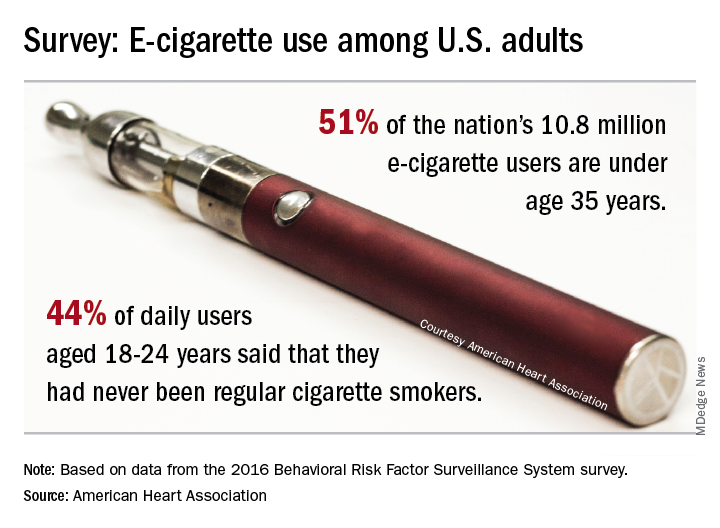
As of 2016, an estimated 4.5% of adults – more than 10.8 million individuals – used e-cigarettes every day or some days, which defined current use for the 466,842 people who responded to the Behavioral Risk Factor Surveillance System survey and were included in the study conducted by the AHA’s Tobacco Regulation and Addiction Center and published in the Annals of Internal Medicine.
Based on that survey data, an estimated 51% of current users were under the age of 35 years in 2016. Daily use was highest among those aged 18-24 years, and of those respondents, 44% said that they had never been regular cigarette users. “It’s particularly disturbing to see these younger people who have never been regular cigarette smokers taking up the use of e-cigarettes, perhaps with the assumption that this alternative nicotine delivery system has been proven to be safe,” said Rose Marie Robertson, MD, who is the AHA’s chief science and medical officer.
The analysis also showed that about 60% of e-cigarette users were men and that use was higher among LGBT people. The first-ever estimates of current use by state put the prevalence highest in Oklahoma at 7.0% and lowest in South Dakota (3.1%) and the District of Columbia (2.3%), the AHA said.
The study was funded through a grant from the National Institutes of Health and the Food and Drug Administration’s Center for Tobacco Products.
[email protected]
SOURCE: Mirbolouk M et al. Ann Intern Med. 2018 Aug 28. doi: 10.7326/M17-3440.
Almost 11 million adults use e-cigarettes in the United States, and the majority are under the age of 35 years, according to the American Heart Association.

As of 2016, an estimated 4.5% of adults – more than 10.8 million individuals – used e-cigarettes every day or some days, which defined current use for the 466,842 people who responded to the Behavioral Risk Factor Surveillance System survey and were included in the study conducted by the AHA’s Tobacco Regulation and Addiction Center and published in the Annals of Internal Medicine.
Based on that survey data, an estimated 51% of current users were under the age of 35 years in 2016. Daily use was highest among those aged 18-24 years, and of those respondents, 44% said that they had never been regular cigarette users. “It’s particularly disturbing to see these younger people who have never been regular cigarette smokers taking up the use of e-cigarettes, perhaps with the assumption that this alternative nicotine delivery system has been proven to be safe,” said Rose Marie Robertson, MD, who is the AHA’s chief science and medical officer.
The analysis also showed that about 60% of e-cigarette users were men and that use was higher among LGBT people. The first-ever estimates of current use by state put the prevalence highest in Oklahoma at 7.0% and lowest in South Dakota (3.1%) and the District of Columbia (2.3%), the AHA said.
The study was funded through a grant from the National Institutes of Health and the Food and Drug Administration’s Center for Tobacco Products.
[email protected]
SOURCE: Mirbolouk M et al. Ann Intern Med. 2018 Aug 28. doi: 10.7326/M17-3440.
Almost 11 million adults use e-cigarettes in the United States, and the majority are under the age of 35 years, according to the American Heart Association.

As of 2016, an estimated 4.5% of adults – more than 10.8 million individuals – used e-cigarettes every day or some days, which defined current use for the 466,842 people who responded to the Behavioral Risk Factor Surveillance System survey and were included in the study conducted by the AHA’s Tobacco Regulation and Addiction Center and published in the Annals of Internal Medicine.
Based on that survey data, an estimated 51% of current users were under the age of 35 years in 2016. Daily use was highest among those aged 18-24 years, and of those respondents, 44% said that they had never been regular cigarette users. “It’s particularly disturbing to see these younger people who have never been regular cigarette smokers taking up the use of e-cigarettes, perhaps with the assumption that this alternative nicotine delivery system has been proven to be safe,” said Rose Marie Robertson, MD, who is the AHA’s chief science and medical officer.
The analysis also showed that about 60% of e-cigarette users were men and that use was higher among LGBT people. The first-ever estimates of current use by state put the prevalence highest in Oklahoma at 7.0% and lowest in South Dakota (3.1%) and the District of Columbia (2.3%), the AHA said.
The study was funded through a grant from the National Institutes of Health and the Food and Drug Administration’s Center for Tobacco Products.
[email protected]
SOURCE: Mirbolouk M et al. Ann Intern Med. 2018 Aug 28. doi: 10.7326/M17-3440.
FROM ANNALS OF INTERNAL MEDICINE
Variation in bacterial drug susceptibility tied to TB relapse risk
Higher pretreatment drug concentrations close to a resistance breakpoint for susceptibility were associated with greater relapse risk in TB, based on data from 54 patients who relapsed and 63 who were treated and cured.
“We postulated that drug-susceptible Mycobacterium tuberculosis might have a graded spectrum of susceptibilities that could be used to determine the risk of relapse,” wrote Roberto Colangeli, PhD, of Rutgers University, Newark, N.J., and his colleagues.
In a study published in the New England Journal of Medicine, the researchers examined pretreatment bacterial isolates from adults with TB who had experienced relapse and those who were cured. Using these isolates, they identified the minimum inhibitory concentration (MIC) – the lowest concentration of the drug that prevents visible bacterial growth in culture – for isoniazid and rifampin.
Overall, after controlling for other potential relapse risk factors, higher pretreatment MIC values for both isoniazid and rifampin were associated with an increased relapse risk. For isoniazid, the average MIC below the breakpoint was 0.0334 mcg/mL for relapsed patients and 0.0286 mcg/mL for cured patients. For rifampin, the average MIC below the breakpoint was 0.0695 mcg/mL for relapsed patients and 0.0453 mcg/mL for cured patients. The higher values for the relapsed versus cured patients were represented by factors of 1.17 and 1.53 for isoniazid and rifampin, respectively.
The average age of the patients was 41 years; 83% were men, and 35% were non-Hispanic white.
The study findings were limited by several factors, including the small sample size, retrospective design, and inability to test MIC values from primary cultures versus subcultures, the researchers wrote. However, the results suggest an impact of MIC values on treatment outcomes, and “additional studies that are performed in larger, well-defined prospective cohorts and that include MIC testing of pretreatment culture isolates will be useful to better validate these findings,”
The study was funded by the National Institute of Allergy and Infectious Diseases. Dr. Colangeli reported no financial conflicts. Dr. Alland disclosed funding from Cepheid and several current and pending patents in the United States and Europe, with some royalties paid to Cepheid.
SOURCE: Colangeli R et al. N Engl J Med. 2018;379:823-33.
Although standard four-drug therapy has been shown to cure 90% of patients in several clinical trials, patients do relapse for reasons such as poor treatment adherence and “variations in the characteristics of the infected patients or the infecting pathogens,” wrote Eric J. Rubin, MD, in an accompanying editorial (N Engl J Med. 2018;379:882-3).
Current antibiotic susceptibility thresholds are often set by committees using models, said Dr. Rubin. “Given the uncertainties in modeling, as has been seen in clinical studies, these breakpoints can be imperfect predictors of treatment response.”
Dr. Rubin proposed that minimum inhibitory concentration (MIC) concentrations could be an alternative to in vitro testing as a predictor of treatment response.
“The clinical laboratory provides us not only with a breakpoint interpretation but also with raw data, a quantitative assessment of MIC values,” he noted. “These values can be thought of more as probabilities of successful therapy than as absolute thresholds, a change in attitude that may dispel a false sense of security about the choice of regimen in the treatment of patients with tuberculosis.”
Dr. Rubin is affiliated with the department of immunology and infectious diseases at the Harvard School of Public Health, Boston. He had no relevant financial conflicts to disclose.
Although standard four-drug therapy has been shown to cure 90% of patients in several clinical trials, patients do relapse for reasons such as poor treatment adherence and “variations in the characteristics of the infected patients or the infecting pathogens,” wrote Eric J. Rubin, MD, in an accompanying editorial (N Engl J Med. 2018;379:882-3).
Current antibiotic susceptibility thresholds are often set by committees using models, said Dr. Rubin. “Given the uncertainties in modeling, as has been seen in clinical studies, these breakpoints can be imperfect predictors of treatment response.”
Dr. Rubin proposed that minimum inhibitory concentration (MIC) concentrations could be an alternative to in vitro testing as a predictor of treatment response.
“The clinical laboratory provides us not only with a breakpoint interpretation but also with raw data, a quantitative assessment of MIC values,” he noted. “These values can be thought of more as probabilities of successful therapy than as absolute thresholds, a change in attitude that may dispel a false sense of security about the choice of regimen in the treatment of patients with tuberculosis.”
Dr. Rubin is affiliated with the department of immunology and infectious diseases at the Harvard School of Public Health, Boston. He had no relevant financial conflicts to disclose.
Although standard four-drug therapy has been shown to cure 90% of patients in several clinical trials, patients do relapse for reasons such as poor treatment adherence and “variations in the characteristics of the infected patients or the infecting pathogens,” wrote Eric J. Rubin, MD, in an accompanying editorial (N Engl J Med. 2018;379:882-3).
Current antibiotic susceptibility thresholds are often set by committees using models, said Dr. Rubin. “Given the uncertainties in modeling, as has been seen in clinical studies, these breakpoints can be imperfect predictors of treatment response.”
Dr. Rubin proposed that minimum inhibitory concentration (MIC) concentrations could be an alternative to in vitro testing as a predictor of treatment response.
“The clinical laboratory provides us not only with a breakpoint interpretation but also with raw data, a quantitative assessment of MIC values,” he noted. “These values can be thought of more as probabilities of successful therapy than as absolute thresholds, a change in attitude that may dispel a false sense of security about the choice of regimen in the treatment of patients with tuberculosis.”
Dr. Rubin is affiliated with the department of immunology and infectious diseases at the Harvard School of Public Health, Boston. He had no relevant financial conflicts to disclose.
Higher pretreatment drug concentrations close to a resistance breakpoint for susceptibility were associated with greater relapse risk in TB, based on data from 54 patients who relapsed and 63 who were treated and cured.
“We postulated that drug-susceptible Mycobacterium tuberculosis might have a graded spectrum of susceptibilities that could be used to determine the risk of relapse,” wrote Roberto Colangeli, PhD, of Rutgers University, Newark, N.J., and his colleagues.
In a study published in the New England Journal of Medicine, the researchers examined pretreatment bacterial isolates from adults with TB who had experienced relapse and those who were cured. Using these isolates, they identified the minimum inhibitory concentration (MIC) – the lowest concentration of the drug that prevents visible bacterial growth in culture – for isoniazid and rifampin.
Overall, after controlling for other potential relapse risk factors, higher pretreatment MIC values for both isoniazid and rifampin were associated with an increased relapse risk. For isoniazid, the average MIC below the breakpoint was 0.0334 mcg/mL for relapsed patients and 0.0286 mcg/mL for cured patients. For rifampin, the average MIC below the breakpoint was 0.0695 mcg/mL for relapsed patients and 0.0453 mcg/mL for cured patients. The higher values for the relapsed versus cured patients were represented by factors of 1.17 and 1.53 for isoniazid and rifampin, respectively.
The average age of the patients was 41 years; 83% were men, and 35% were non-Hispanic white.
The study findings were limited by several factors, including the small sample size, retrospective design, and inability to test MIC values from primary cultures versus subcultures, the researchers wrote. However, the results suggest an impact of MIC values on treatment outcomes, and “additional studies that are performed in larger, well-defined prospective cohorts and that include MIC testing of pretreatment culture isolates will be useful to better validate these findings,”
The study was funded by the National Institute of Allergy and Infectious Diseases. Dr. Colangeli reported no financial conflicts. Dr. Alland disclosed funding from Cepheid and several current and pending patents in the United States and Europe, with some royalties paid to Cepheid.
SOURCE: Colangeli R et al. N Engl J Med. 2018;379:823-33.
Higher pretreatment drug concentrations close to a resistance breakpoint for susceptibility were associated with greater relapse risk in TB, based on data from 54 patients who relapsed and 63 who were treated and cured.
“We postulated that drug-susceptible Mycobacterium tuberculosis might have a graded spectrum of susceptibilities that could be used to determine the risk of relapse,” wrote Roberto Colangeli, PhD, of Rutgers University, Newark, N.J., and his colleagues.
In a study published in the New England Journal of Medicine, the researchers examined pretreatment bacterial isolates from adults with TB who had experienced relapse and those who were cured. Using these isolates, they identified the minimum inhibitory concentration (MIC) – the lowest concentration of the drug that prevents visible bacterial growth in culture – for isoniazid and rifampin.
Overall, after controlling for other potential relapse risk factors, higher pretreatment MIC values for both isoniazid and rifampin were associated with an increased relapse risk. For isoniazid, the average MIC below the breakpoint was 0.0334 mcg/mL for relapsed patients and 0.0286 mcg/mL for cured patients. For rifampin, the average MIC below the breakpoint was 0.0695 mcg/mL for relapsed patients and 0.0453 mcg/mL for cured patients. The higher values for the relapsed versus cured patients were represented by factors of 1.17 and 1.53 for isoniazid and rifampin, respectively.
The average age of the patients was 41 years; 83% were men, and 35% were non-Hispanic white.
The study findings were limited by several factors, including the small sample size, retrospective design, and inability to test MIC values from primary cultures versus subcultures, the researchers wrote. However, the results suggest an impact of MIC values on treatment outcomes, and “additional studies that are performed in larger, well-defined prospective cohorts and that include MIC testing of pretreatment culture isolates will be useful to better validate these findings,”
The study was funded by the National Institute of Allergy and Infectious Diseases. Dr. Colangeli reported no financial conflicts. Dr. Alland disclosed funding from Cepheid and several current and pending patents in the United States and Europe, with some royalties paid to Cepheid.
SOURCE: Colangeli R et al. N Engl J Med. 2018;379:823-33.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: Risk of TB relapse increased with higher pretreatment minimum inhibitory concentration values for either isoniazid or rifampin.
Major finding: The higher values for the relapsed versus cured patients were represented by factors of 1.17 and 1.53 for isoniazid and rifampin, respectively.
Study details: The data come from a retrospective study of isolates from 54 patients with TB who relapsed and 63 who were treated and cured.
Disclosures: The study was funded by the National Institute of Allergy and Infectious Diseases. Dr. Colangeli reported no financial conflicts. Dr. Alland disclosed funding from Cepheid and several current and pending patents in the United States and Europe, with some royalties paid to Cepheid.
Source: Colangeli R et al. N Engl J Med. 2018;379:823-33.
Declining lung function linked to heart failure, stroke
Rapid declines in spirometric measures of lung function were associated with higher risks of cardiovascular disease, according to a recent analysis of a large, prospective cohort study.
Rapid declines in forced expiratory volume in 1 second (FEV1) were associated with increased incidence of heart failure, stroke, and death in the analysis of the ARIC (Atherosclerosis Risk in Communities) study.
The risk of incident heart failure among FEV1 rapid decliners was particularly high, with a fourfold increase within 12 months. That suggests clinicians should carefully consider incipient heart failure in patients with rapid changes in FEV1, investigators reported in the Journal of the American College of Cardiology.
Rapid declines in forced vital capacity (FVC) were also associated with higher incidences of heart failure and death in the analysis by Odilson M. Silvestre, MD, of the division of cardiovascular medicine at Brigham and Women’s Hospital, Boston, and colleagues.
The analysis included a total of 10,351 ARIC participants with a mean follow-up of 17 years. All had undergone spirometry at the first study visit between 1987 and 1989, and on the second visit between 1990 and 1992.
One-quarter of participants were classified as FEV1 rapid decliners, defined by an FEV1 decrease of at least 1.9% per year. Likewise, one-quarter of participants were classified as FVC rapid decliners, based on an FVC decrease of at least 2.1%.
Rapid decline in FEV1 was associated with a higher risk of incident heart failure (hazard ratio, 1.17; 95% confidence interval, 1.04-1.33; P = .010), and was most prognostic in the first year of follow-up (HR, 4.22; 95% CI, 1.34-13.26; P = .01), investigators said.
Rapid decline in FVC was likewise associated with a greater heart failure risk (HR, 1.27; 95% CI, 1.12-1.44; P less than .001).
Increased heart failure risk persisted after excluding patients with incident coronary heart disease in both the FEV1 and FVC rapid decliners, the investigators said.
A rapid decline in FEV1 was also associated with a higher stroke risk (HR, 1.25; 95% CI, 1.04-1.50; P = .015).
FEV1 rapid decliners had a higher overall rate of incident cardiovascular disease than those without rapid decline, even after adjustment for baseline variables such as age, sex, race, body mass index, and heart rate (HR, 1.15; 95% CI, 1.04-1.26; P = .004), and FVC rapid decliners likewise had a 19% greater risk of the composite endpoint (HR, 1.19; 95% CI, 1.08-1.32; P less than .001).
The National Heart, Lung, and Blood Institute, the American Heart Association, and other sources supported the study. Dr. Silvestre reported having no relevant conflicts.
SOURCE: Silvestre OM et al. J Am Coll Cardiol. 2018 Sep 4;72(10):1109-22.
Cardiologists and pulmonologists should closely collaborate to better identify the relationship between lung function decline and early cardiovascular disease detection, said the authors of an editorial accompanying the study.
Improved collaboration would help manage these conditions, stopping early disease progression and preventing overt cardiovascular disease, wrote Daniel A. Duprez, MD, PhD, and David R. Jacobs Jr., PhD.
“The resulting symptomatic and prognostic benefits outweigh those attainable by treating either condition alone,” noted Dr. Duprez and Dr. Jacobs.
The study by Dr. Silvestre and coauthors showed that rapid declines in forced expiratory volume in one second (FEV1) and forced vital capacity (FVC) among participants in the ARIC (Atherosclerosis Risk in Communities) study were associated with a higher incidence of composite cardiovascular disease, heart failure, and total death.
The association between FEV1 and new heart failure was substantially impacted by 22 heart failure events occurring in the first year, with a hazard ratio of 4.22 for predicting those cases, the editorial authors noted.
“We suggest that this association with early cases could be the result of reversed causality, reflecting heart failure undiagnosed at the second spirometry test,” they explained (J Am Coll Cardiol. 2018 Sep 4;72[10]:1123-5).
Dr. Duprez is with the cardiovascular division of the University of Minnesota, Minneapolis, and Dr. Jacobs is with the division of epidemiology and community health at the University of Minnesota, Minneapolis. Dr. Duprez and Dr. Jacobs reported they had no relevant disclosures.
Cardiologists and pulmonologists should closely collaborate to better identify the relationship between lung function decline and early cardiovascular disease detection, said the authors of an editorial accompanying the study.
Improved collaboration would help manage these conditions, stopping early disease progression and preventing overt cardiovascular disease, wrote Daniel A. Duprez, MD, PhD, and David R. Jacobs Jr., PhD.
“The resulting symptomatic and prognostic benefits outweigh those attainable by treating either condition alone,” noted Dr. Duprez and Dr. Jacobs.
The study by Dr. Silvestre and coauthors showed that rapid declines in forced expiratory volume in one second (FEV1) and forced vital capacity (FVC) among participants in the ARIC (Atherosclerosis Risk in Communities) study were associated with a higher incidence of composite cardiovascular disease, heart failure, and total death.
The association between FEV1 and new heart failure was substantially impacted by 22 heart failure events occurring in the first year, with a hazard ratio of 4.22 for predicting those cases, the editorial authors noted.
“We suggest that this association with early cases could be the result of reversed causality, reflecting heart failure undiagnosed at the second spirometry test,” they explained (J Am Coll Cardiol. 2018 Sep 4;72[10]:1123-5).
Dr. Duprez is with the cardiovascular division of the University of Minnesota, Minneapolis, and Dr. Jacobs is with the division of epidemiology and community health at the University of Minnesota, Minneapolis. Dr. Duprez and Dr. Jacobs reported they had no relevant disclosures.
Cardiologists and pulmonologists should closely collaborate to better identify the relationship between lung function decline and early cardiovascular disease detection, said the authors of an editorial accompanying the study.
Improved collaboration would help manage these conditions, stopping early disease progression and preventing overt cardiovascular disease, wrote Daniel A. Duprez, MD, PhD, and David R. Jacobs Jr., PhD.
“The resulting symptomatic and prognostic benefits outweigh those attainable by treating either condition alone,” noted Dr. Duprez and Dr. Jacobs.
The study by Dr. Silvestre and coauthors showed that rapid declines in forced expiratory volume in one second (FEV1) and forced vital capacity (FVC) among participants in the ARIC (Atherosclerosis Risk in Communities) study were associated with a higher incidence of composite cardiovascular disease, heart failure, and total death.
The association between FEV1 and new heart failure was substantially impacted by 22 heart failure events occurring in the first year, with a hazard ratio of 4.22 for predicting those cases, the editorial authors noted.
“We suggest that this association with early cases could be the result of reversed causality, reflecting heart failure undiagnosed at the second spirometry test,” they explained (J Am Coll Cardiol. 2018 Sep 4;72[10]:1123-5).
Dr. Duprez is with the cardiovascular division of the University of Minnesota, Minneapolis, and Dr. Jacobs is with the division of epidemiology and community health at the University of Minnesota, Minneapolis. Dr. Duprez and Dr. Jacobs reported they had no relevant disclosures.
Rapid declines in spirometric measures of lung function were associated with higher risks of cardiovascular disease, according to a recent analysis of a large, prospective cohort study.
Rapid declines in forced expiratory volume in 1 second (FEV1) were associated with increased incidence of heart failure, stroke, and death in the analysis of the ARIC (Atherosclerosis Risk in Communities) study.
The risk of incident heart failure among FEV1 rapid decliners was particularly high, with a fourfold increase within 12 months. That suggests clinicians should carefully consider incipient heart failure in patients with rapid changes in FEV1, investigators reported in the Journal of the American College of Cardiology.
Rapid declines in forced vital capacity (FVC) were also associated with higher incidences of heart failure and death in the analysis by Odilson M. Silvestre, MD, of the division of cardiovascular medicine at Brigham and Women’s Hospital, Boston, and colleagues.
The analysis included a total of 10,351 ARIC participants with a mean follow-up of 17 years. All had undergone spirometry at the first study visit between 1987 and 1989, and on the second visit between 1990 and 1992.
One-quarter of participants were classified as FEV1 rapid decliners, defined by an FEV1 decrease of at least 1.9% per year. Likewise, one-quarter of participants were classified as FVC rapid decliners, based on an FVC decrease of at least 2.1%.
Rapid decline in FEV1 was associated with a higher risk of incident heart failure (hazard ratio, 1.17; 95% confidence interval, 1.04-1.33; P = .010), and was most prognostic in the first year of follow-up (HR, 4.22; 95% CI, 1.34-13.26; P = .01), investigators said.
Rapid decline in FVC was likewise associated with a greater heart failure risk (HR, 1.27; 95% CI, 1.12-1.44; P less than .001).
Increased heart failure risk persisted after excluding patients with incident coronary heart disease in both the FEV1 and FVC rapid decliners, the investigators said.
A rapid decline in FEV1 was also associated with a higher stroke risk (HR, 1.25; 95% CI, 1.04-1.50; P = .015).
FEV1 rapid decliners had a higher overall rate of incident cardiovascular disease than those without rapid decline, even after adjustment for baseline variables such as age, sex, race, body mass index, and heart rate (HR, 1.15; 95% CI, 1.04-1.26; P = .004), and FVC rapid decliners likewise had a 19% greater risk of the composite endpoint (HR, 1.19; 95% CI, 1.08-1.32; P less than .001).
The National Heart, Lung, and Blood Institute, the American Heart Association, and other sources supported the study. Dr. Silvestre reported having no relevant conflicts.
SOURCE: Silvestre OM et al. J Am Coll Cardiol. 2018 Sep 4;72(10):1109-22.
Rapid declines in spirometric measures of lung function were associated with higher risks of cardiovascular disease, according to a recent analysis of a large, prospective cohort study.
Rapid declines in forced expiratory volume in 1 second (FEV1) were associated with increased incidence of heart failure, stroke, and death in the analysis of the ARIC (Atherosclerosis Risk in Communities) study.
The risk of incident heart failure among FEV1 rapid decliners was particularly high, with a fourfold increase within 12 months. That suggests clinicians should carefully consider incipient heart failure in patients with rapid changes in FEV1, investigators reported in the Journal of the American College of Cardiology.
Rapid declines in forced vital capacity (FVC) were also associated with higher incidences of heart failure and death in the analysis by Odilson M. Silvestre, MD, of the division of cardiovascular medicine at Brigham and Women’s Hospital, Boston, and colleagues.
The analysis included a total of 10,351 ARIC participants with a mean follow-up of 17 years. All had undergone spirometry at the first study visit between 1987 and 1989, and on the second visit between 1990 and 1992.
One-quarter of participants were classified as FEV1 rapid decliners, defined by an FEV1 decrease of at least 1.9% per year. Likewise, one-quarter of participants were classified as FVC rapid decliners, based on an FVC decrease of at least 2.1%.
Rapid decline in FEV1 was associated with a higher risk of incident heart failure (hazard ratio, 1.17; 95% confidence interval, 1.04-1.33; P = .010), and was most prognostic in the first year of follow-up (HR, 4.22; 95% CI, 1.34-13.26; P = .01), investigators said.
Rapid decline in FVC was likewise associated with a greater heart failure risk (HR, 1.27; 95% CI, 1.12-1.44; P less than .001).
Increased heart failure risk persisted after excluding patients with incident coronary heart disease in both the FEV1 and FVC rapid decliners, the investigators said.
A rapid decline in FEV1 was also associated with a higher stroke risk (HR, 1.25; 95% CI, 1.04-1.50; P = .015).
FEV1 rapid decliners had a higher overall rate of incident cardiovascular disease than those without rapid decline, even after adjustment for baseline variables such as age, sex, race, body mass index, and heart rate (HR, 1.15; 95% CI, 1.04-1.26; P = .004), and FVC rapid decliners likewise had a 19% greater risk of the composite endpoint (HR, 1.19; 95% CI, 1.08-1.32; P less than .001).
The National Heart, Lung, and Blood Institute, the American Heart Association, and other sources supported the study. Dr. Silvestre reported having no relevant conflicts.
SOURCE: Silvestre OM et al. J Am Coll Cardiol. 2018 Sep 4;72(10):1109-22.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Key clinical point: Rapid declines in spirometric measures of lung function were associated with higher risks of heart failure, among other adverse cardiovascular outcomes.
Major finding: Rapid decline in forced expiratory volume in 1 second was associated with higher risk of incident heart failure (HR, 1.17; 95% CI, 1.04-1.33; P = .010), and was most prognostic in the first year of follow-up (HR, 4.22; 95% CI, 1.34-13.26; P = .01).
Study details: An analysis including a total of 10,351 participants in a large, prospective cohort study with a mean follow-up of 17 years.
Disclosures: The National Heart, Lung, and Blood Institute, the American Heart Association, and other sources supported the study. Dr. Silvestre reported having no relevant conflicts.
Source: Silvestre OM et al. J Am Coll Cardiol. 2018 Sep 4;72(10):1109-22.
Live attenuated flu vaccine gets ACIP nod for 2018-2019 season
The latest seasonal influenza vaccine recommendations from the Advisory Committee on Immunization Practices provide several key updates that will impact clinical practice in the 2018-2019 influenza season.
Of note, , following two seasons in which the committee recommended it not be used.
ACIP also updated its recommendations for individuals with a history of egg allergy, described the vaccine strains chosen for 2018-2019 season, and detailed the changes in age indications for Afluria Quadrivalent and Fluarix Quadrivalent that have been made since publication of its previous guidelines.
Published in MMWR Recommendations and Reports, the updated ACIP recommendations reflect discussions and decisions from the three public meetings of ACIP that have taken place since the last annual update.
All individuals 6 months of age and older who have no contraindications to influenza vaccine should receive routine annual influenza vaccine, ACIP also said in its report, reinforcing a key recommendation that has been in place since 2010.
“To avoid missed opportunities for vaccination, providers should offer vaccination during routine health care visits and hospitalizations,” wrote authors of the report, including lead author Lisa A. Grohskopf, MD, of the National Center for Immunization and Respiratory Diseases, Centers for Disease Control and Prevention, Atlanta.
Dr. Grohskopf and coauthors made no specific recommendations on which vaccine to use. They said providers should choose licensed, age-appropriate recommended vaccines expected to be available for the 2018-2019 season, including inactivated influenza vaccines (IIV), a recombinant influenza vaccine (RIV4), and the LAIV option.
FluMist Quadrivalent, the one LAIV product expected to be available for the 2018-2019 season, is licensed for individuals aged 2-49 years.
In its deliberations over the updated LAIV recommendation, ACIP reviewed observational data from previous seasons suggesting that the vaccine was poorly effective, and significantly less effective than IIV, against influenza A(H1N1) pdm09 viruses.
The current formulation of FluMist includes a new H1N1pdm09-like vaccine virus. While no effectiveness estimates were available at the time of review, ACIP said it did consider manufacturer data on shedding and immunogenicity for the current vaccine in children between the ages of 24 months through less than 4 years.
“These data suggest that this new H1N1pdm09-like virus has improved replicative fitness over previous H1N1pdm09-like viruses included in LAIV,” Dr. Grohskopf and colleagues wrote.
Individuals with an egg allergy history also can receive any licensed, recommended, age-appropriate IIV, RIV, or LAIV vaccine, said ACIP. This updated recommendation was based in part on the committee’s review and discussion of three studies that showed no cases of anaphylaxis in egg-allergic children receiving LAIV.
The ACIP recommendation update also outlines the strains selected earlier this year for the 2018-2019 season. Trivalent influenza vaccines in the United States will include an A/Michigan/45/2015 (H1N1) pdm09–like virus, an A/Singapore/INFIMH-16-0019/2016 (H3N2)-like virus, and a B/Colorado/06/2017–like virus (Victoria lineage). Quadrivalent vaccines will include those strains plus a B/Phuket/3073/2013–like virus (Yamagata lineage).
The report also acknowledges the recent expansion of age indication for two vaccines that have occurred since the last ACIP recommendations.
Afluria Quadrivalent was previously licensed for individuals 18 years of age and older. In August 2017, the Food and Drug Administration approved expansion of the indication to individuals 5 years of age or older. In January 2018, FDA approved expansion of the Fluarix Quadrivalent indication, previously licensed for age 3 and older, to individuals 6 months and older.
Report coauthor Emmanuel B. Walter disclosed grants from Novavax and Merck. The remaining report authors reported no relevant financial disclosures.
SOURCE: Grohskopf LA et al. MMWR Recomm Rep. 2018 Aug 24;67(3):1-20.
The latest seasonal influenza vaccine recommendations from the Advisory Committee on Immunization Practices provide several key updates that will impact clinical practice in the 2018-2019 influenza season.
Of note, , following two seasons in which the committee recommended it not be used.
ACIP also updated its recommendations for individuals with a history of egg allergy, described the vaccine strains chosen for 2018-2019 season, and detailed the changes in age indications for Afluria Quadrivalent and Fluarix Quadrivalent that have been made since publication of its previous guidelines.
Published in MMWR Recommendations and Reports, the updated ACIP recommendations reflect discussions and decisions from the three public meetings of ACIP that have taken place since the last annual update.
All individuals 6 months of age and older who have no contraindications to influenza vaccine should receive routine annual influenza vaccine, ACIP also said in its report, reinforcing a key recommendation that has been in place since 2010.
“To avoid missed opportunities for vaccination, providers should offer vaccination during routine health care visits and hospitalizations,” wrote authors of the report, including lead author Lisa A. Grohskopf, MD, of the National Center for Immunization and Respiratory Diseases, Centers for Disease Control and Prevention, Atlanta.
Dr. Grohskopf and coauthors made no specific recommendations on which vaccine to use. They said providers should choose licensed, age-appropriate recommended vaccines expected to be available for the 2018-2019 season, including inactivated influenza vaccines (IIV), a recombinant influenza vaccine (RIV4), and the LAIV option.
FluMist Quadrivalent, the one LAIV product expected to be available for the 2018-2019 season, is licensed for individuals aged 2-49 years.
In its deliberations over the updated LAIV recommendation, ACIP reviewed observational data from previous seasons suggesting that the vaccine was poorly effective, and significantly less effective than IIV, against influenza A(H1N1) pdm09 viruses.
The current formulation of FluMist includes a new H1N1pdm09-like vaccine virus. While no effectiveness estimates were available at the time of review, ACIP said it did consider manufacturer data on shedding and immunogenicity for the current vaccine in children between the ages of 24 months through less than 4 years.
“These data suggest that this new H1N1pdm09-like virus has improved replicative fitness over previous H1N1pdm09-like viruses included in LAIV,” Dr. Grohskopf and colleagues wrote.
Individuals with an egg allergy history also can receive any licensed, recommended, age-appropriate IIV, RIV, or LAIV vaccine, said ACIP. This updated recommendation was based in part on the committee’s review and discussion of three studies that showed no cases of anaphylaxis in egg-allergic children receiving LAIV.
The ACIP recommendation update also outlines the strains selected earlier this year for the 2018-2019 season. Trivalent influenza vaccines in the United States will include an A/Michigan/45/2015 (H1N1) pdm09–like virus, an A/Singapore/INFIMH-16-0019/2016 (H3N2)-like virus, and a B/Colorado/06/2017–like virus (Victoria lineage). Quadrivalent vaccines will include those strains plus a B/Phuket/3073/2013–like virus (Yamagata lineage).
The report also acknowledges the recent expansion of age indication for two vaccines that have occurred since the last ACIP recommendations.
Afluria Quadrivalent was previously licensed for individuals 18 years of age and older. In August 2017, the Food and Drug Administration approved expansion of the indication to individuals 5 years of age or older. In January 2018, FDA approved expansion of the Fluarix Quadrivalent indication, previously licensed for age 3 and older, to individuals 6 months and older.
Report coauthor Emmanuel B. Walter disclosed grants from Novavax and Merck. The remaining report authors reported no relevant financial disclosures.
SOURCE: Grohskopf LA et al. MMWR Recomm Rep. 2018 Aug 24;67(3):1-20.
The latest seasonal influenza vaccine recommendations from the Advisory Committee on Immunization Practices provide several key updates that will impact clinical practice in the 2018-2019 influenza season.
Of note, , following two seasons in which the committee recommended it not be used.
ACIP also updated its recommendations for individuals with a history of egg allergy, described the vaccine strains chosen for 2018-2019 season, and detailed the changes in age indications for Afluria Quadrivalent and Fluarix Quadrivalent that have been made since publication of its previous guidelines.
Published in MMWR Recommendations and Reports, the updated ACIP recommendations reflect discussions and decisions from the three public meetings of ACIP that have taken place since the last annual update.
All individuals 6 months of age and older who have no contraindications to influenza vaccine should receive routine annual influenza vaccine, ACIP also said in its report, reinforcing a key recommendation that has been in place since 2010.
“To avoid missed opportunities for vaccination, providers should offer vaccination during routine health care visits and hospitalizations,” wrote authors of the report, including lead author Lisa A. Grohskopf, MD, of the National Center for Immunization and Respiratory Diseases, Centers for Disease Control and Prevention, Atlanta.
Dr. Grohskopf and coauthors made no specific recommendations on which vaccine to use. They said providers should choose licensed, age-appropriate recommended vaccines expected to be available for the 2018-2019 season, including inactivated influenza vaccines (IIV), a recombinant influenza vaccine (RIV4), and the LAIV option.
FluMist Quadrivalent, the one LAIV product expected to be available for the 2018-2019 season, is licensed for individuals aged 2-49 years.
In its deliberations over the updated LAIV recommendation, ACIP reviewed observational data from previous seasons suggesting that the vaccine was poorly effective, and significantly less effective than IIV, against influenza A(H1N1) pdm09 viruses.
The current formulation of FluMist includes a new H1N1pdm09-like vaccine virus. While no effectiveness estimates were available at the time of review, ACIP said it did consider manufacturer data on shedding and immunogenicity for the current vaccine in children between the ages of 24 months through less than 4 years.
“These data suggest that this new H1N1pdm09-like virus has improved replicative fitness over previous H1N1pdm09-like viruses included in LAIV,” Dr. Grohskopf and colleagues wrote.
Individuals with an egg allergy history also can receive any licensed, recommended, age-appropriate IIV, RIV, or LAIV vaccine, said ACIP. This updated recommendation was based in part on the committee’s review and discussion of three studies that showed no cases of anaphylaxis in egg-allergic children receiving LAIV.
The ACIP recommendation update also outlines the strains selected earlier this year for the 2018-2019 season. Trivalent influenza vaccines in the United States will include an A/Michigan/45/2015 (H1N1) pdm09–like virus, an A/Singapore/INFIMH-16-0019/2016 (H3N2)-like virus, and a B/Colorado/06/2017–like virus (Victoria lineage). Quadrivalent vaccines will include those strains plus a B/Phuket/3073/2013–like virus (Yamagata lineage).
The report also acknowledges the recent expansion of age indication for two vaccines that have occurred since the last ACIP recommendations.
Afluria Quadrivalent was previously licensed for individuals 18 years of age and older. In August 2017, the Food and Drug Administration approved expansion of the indication to individuals 5 years of age or older. In January 2018, FDA approved expansion of the Fluarix Quadrivalent indication, previously licensed for age 3 and older, to individuals 6 months and older.
Report coauthor Emmanuel B. Walter disclosed grants from Novavax and Merck. The remaining report authors reported no relevant financial disclosures.
SOURCE: Grohskopf LA et al. MMWR Recomm Rep. 2018 Aug 24;67(3):1-20.
FROM MMWR
Some PE patients don’t require hospitalization
A new study suggests that
Researchers tested outpatient anticoagulant therapy in 200 patients with PE with a low mortality risk. At 90 days of follow-up, there were no deaths or recurrences of venous thromboembolism (VTE), but one patient experienced major bleeding after a traumatic injury.
A majority of patients said they were satisfied with outpatient care.
Joseph R. Bledsoe, MD, of Intermountain Medical Center in Salt Lake City, and his colleagues reported these results in Chest.
The researchers tracked patients who were treated for acute PE in five Intermountain Healthcare emergency departments (EDs) from 2013 to 2016. The patients had to have a low mortality risk according to the Pulmonary Embolism Severity Index (score less than 86), echocardiography (no signs of right heart strain), and whole-leg compression ultrasound. Patients could not have deep vein thrombosis proximal to the popliteal vein, hypoxia, hypotension, hepatic failure, or renal failure. They had to be eligible for therapeutic anticoagulation and could not have any condition requiring hospitalization.
With these criteria, the researchers selected 200 patients. They were observed in the ED or hospital for 12-24 hours and then discharged with anticoagulant therapy. Patients received rivaroxaban (n = 149), enoxaparin transitioned to warfarin (n = 26), apixaban (n = 24), or enoxaparin alone (n = 1).
Results
The study’s primary outcome was the 90-day composite rate of all-cause mortality, recurrent symptomatic VTE, and major bleeding. There were no deaths and no cases of recurrent VTE, but one patient did experience major bleeding at day 61 because of a traumatic thigh injury.
Within 7 days of study enrollment, there were 19 patients (9.5%) who returned to the ED and 2 patients (1%) who were admitted to the hospital. One patient with pulmonary infarct was admitted for pain control (day 2); the other was admitted for an elective coronary intervention (day 7) because of a positive cardiac stress test.
Within 30 days, 32 patients (16%) returned to the ED, and 5 (3%) were admitted to the hospital for events unrelated to their PE.
The study also showed that patients were largely satisfied with outpatient care. Of the 146 patients who completed a satisfaction survey at 90 days, 89% said they would choose outpatient management if they had another PE in the future.
“We found a large subset of patients with blood clots who’d do well at home; in fact, who probably did better at home,” Dr. Bledsoe said. “When patients are sent home versus staying in the hospital, they’re at lower risk of getting another infection. It’s a lot less expensive, too.”
Currently, the standard of care in the United States for acute PE is hospitalization for all patients. That’s recommended, in part, because their overall mortality rate is 17%. However, the lower mortality rate among some appropriately risk-stratified patients suggests that at-home care, which has become the norm in some European countries, leads to better outcomes for those patients overall and less chance of a hospital-introduced infection, according to Dr. Bledsoe. “Our findings show that if you appropriately risk-stratify patients, there are a lot of people with blood clots who are safe to go home.”
He added that similar research should be conducted outside of the Intermountain Healthcare system to confirm the results of this study and that a larger group of patients should be studied.
The investigators reported no conflicts related to this study.
SOURCE: Bledsoe JR et al. Chest. 2018 Aug;154(2):249-56.
A new study suggests that
Researchers tested outpatient anticoagulant therapy in 200 patients with PE with a low mortality risk. At 90 days of follow-up, there were no deaths or recurrences of venous thromboembolism (VTE), but one patient experienced major bleeding after a traumatic injury.
A majority of patients said they were satisfied with outpatient care.
Joseph R. Bledsoe, MD, of Intermountain Medical Center in Salt Lake City, and his colleagues reported these results in Chest.
The researchers tracked patients who were treated for acute PE in five Intermountain Healthcare emergency departments (EDs) from 2013 to 2016. The patients had to have a low mortality risk according to the Pulmonary Embolism Severity Index (score less than 86), echocardiography (no signs of right heart strain), and whole-leg compression ultrasound. Patients could not have deep vein thrombosis proximal to the popliteal vein, hypoxia, hypotension, hepatic failure, or renal failure. They had to be eligible for therapeutic anticoagulation and could not have any condition requiring hospitalization.
With these criteria, the researchers selected 200 patients. They were observed in the ED or hospital for 12-24 hours and then discharged with anticoagulant therapy. Patients received rivaroxaban (n = 149), enoxaparin transitioned to warfarin (n = 26), apixaban (n = 24), or enoxaparin alone (n = 1).
Results
The study’s primary outcome was the 90-day composite rate of all-cause mortality, recurrent symptomatic VTE, and major bleeding. There were no deaths and no cases of recurrent VTE, but one patient did experience major bleeding at day 61 because of a traumatic thigh injury.
Within 7 days of study enrollment, there were 19 patients (9.5%) who returned to the ED and 2 patients (1%) who were admitted to the hospital. One patient with pulmonary infarct was admitted for pain control (day 2); the other was admitted for an elective coronary intervention (day 7) because of a positive cardiac stress test.
Within 30 days, 32 patients (16%) returned to the ED, and 5 (3%) were admitted to the hospital for events unrelated to their PE.
The study also showed that patients were largely satisfied with outpatient care. Of the 146 patients who completed a satisfaction survey at 90 days, 89% said they would choose outpatient management if they had another PE in the future.
“We found a large subset of patients with blood clots who’d do well at home; in fact, who probably did better at home,” Dr. Bledsoe said. “When patients are sent home versus staying in the hospital, they’re at lower risk of getting another infection. It’s a lot less expensive, too.”
Currently, the standard of care in the United States for acute PE is hospitalization for all patients. That’s recommended, in part, because their overall mortality rate is 17%. However, the lower mortality rate among some appropriately risk-stratified patients suggests that at-home care, which has become the norm in some European countries, leads to better outcomes for those patients overall and less chance of a hospital-introduced infection, according to Dr. Bledsoe. “Our findings show that if you appropriately risk-stratify patients, there are a lot of people with blood clots who are safe to go home.”
He added that similar research should be conducted outside of the Intermountain Healthcare system to confirm the results of this study and that a larger group of patients should be studied.
The investigators reported no conflicts related to this study.
SOURCE: Bledsoe JR et al. Chest. 2018 Aug;154(2):249-56.
A new study suggests that
Researchers tested outpatient anticoagulant therapy in 200 patients with PE with a low mortality risk. At 90 days of follow-up, there were no deaths or recurrences of venous thromboembolism (VTE), but one patient experienced major bleeding after a traumatic injury.
A majority of patients said they were satisfied with outpatient care.
Joseph R. Bledsoe, MD, of Intermountain Medical Center in Salt Lake City, and his colleagues reported these results in Chest.
The researchers tracked patients who were treated for acute PE in five Intermountain Healthcare emergency departments (EDs) from 2013 to 2016. The patients had to have a low mortality risk according to the Pulmonary Embolism Severity Index (score less than 86), echocardiography (no signs of right heart strain), and whole-leg compression ultrasound. Patients could not have deep vein thrombosis proximal to the popliteal vein, hypoxia, hypotension, hepatic failure, or renal failure. They had to be eligible for therapeutic anticoagulation and could not have any condition requiring hospitalization.
With these criteria, the researchers selected 200 patients. They were observed in the ED or hospital for 12-24 hours and then discharged with anticoagulant therapy. Patients received rivaroxaban (n = 149), enoxaparin transitioned to warfarin (n = 26), apixaban (n = 24), or enoxaparin alone (n = 1).
Results
The study’s primary outcome was the 90-day composite rate of all-cause mortality, recurrent symptomatic VTE, and major bleeding. There were no deaths and no cases of recurrent VTE, but one patient did experience major bleeding at day 61 because of a traumatic thigh injury.
Within 7 days of study enrollment, there were 19 patients (9.5%) who returned to the ED and 2 patients (1%) who were admitted to the hospital. One patient with pulmonary infarct was admitted for pain control (day 2); the other was admitted for an elective coronary intervention (day 7) because of a positive cardiac stress test.
Within 30 days, 32 patients (16%) returned to the ED, and 5 (3%) were admitted to the hospital for events unrelated to their PE.
The study also showed that patients were largely satisfied with outpatient care. Of the 146 patients who completed a satisfaction survey at 90 days, 89% said they would choose outpatient management if they had another PE in the future.
“We found a large subset of patients with blood clots who’d do well at home; in fact, who probably did better at home,” Dr. Bledsoe said. “When patients are sent home versus staying in the hospital, they’re at lower risk of getting another infection. It’s a lot less expensive, too.”
Currently, the standard of care in the United States for acute PE is hospitalization for all patients. That’s recommended, in part, because their overall mortality rate is 17%. However, the lower mortality rate among some appropriately risk-stratified patients suggests that at-home care, which has become the norm in some European countries, leads to better outcomes for those patients overall and less chance of a hospital-introduced infection, according to Dr. Bledsoe. “Our findings show that if you appropriately risk-stratify patients, there are a lot of people with blood clots who are safe to go home.”
He added that similar research should be conducted outside of the Intermountain Healthcare system to confirm the results of this study and that a larger group of patients should be studied.
The investigators reported no conflicts related to this study.
SOURCE: Bledsoe JR et al. Chest. 2018 Aug;154(2):249-56.
FROM CHEST
Key clinical point: There were no deaths or recurrences of pulmonary embolism at 90 days in a group of patients stratified by criteria for low risk.
Major finding: At 90 days of follow-up, there were no deaths or recurrences of venous thromboembolism.
Study details: Researchers tested outpatient anticoagulant therapy in 200 patients with pulmonary embolism with a low mortality risk.
Disclosures: The investigators reported no conflicts related to this study.
Source: Bledsoe JR et al. Chest. 2018 Aug;154(2):249-56.
Low-dose CT fails to improve small cell lung cancer survival
Early had no significant impact on patient survival, based on data from a randomized trial.
Although early detection of small-cell lung cancer might improve outcomes for patients given the metastatic nature of the disease, previous studies on the value and impact of low-dose CT have been divergent, wrote Anish Thomas, MD, of the National Cancer Institute and his colleagues.
In a study published in Chest, the researchers reviewed characteristics of low-dose CT–detected small cell lung cancer (SCLC) in the randomized National Lung Screening Trial (NLST). The trial compared low-dose CT (LDCT) with chest radiography to assess patients at high risk for developing lung cancer. The study population included adults aged 55-74 years with a history of smoking of at least 30 pack-years, and former smokers who had quit within the past 15 years.
A total of 26,722 participants were randomized to the LDCT treatment arm with a median follow-up of 6.5 years; 143 SCLC cases and 926 non–small cell lung cancer (NSCLC) cases were identified in this group.
Of the 143 SCLC cases, 49 (34.2%) were screen detected, 15 (10.5%) were interval detected and 79 (55.2%) were not screened or were identified post screening.
A cancer was defined as screen detected if it was diagnosed within 1 year of a positive screening or diagnosed after a longer period but with no time gap between diagnostic procedures of more than 1 year; an interval cancer was diagnosed within a year of a negative screen, and non-screened cancers were those found in patients who received no NLST screening or other post screening.
Significantly more of the SCLC cases were at stage III or IV than the NSCLC cases (86% vs. 36%). Unfavorable stage III or IV lung cancers were identified in 80% of screen detected, 86% of interval detected, and 90% of nonscreened or postscreened cases.
In addition, 31 (63%) of the 49 screen-detected SCLC cases had at least one noncalcified nodule in the cancer lobe and 24 (49%) had a single NCN in the cancer lobe.
The 3-year cancer-specific survival rates were not significantly different for screen-detected, interval-detected, and nonscreened or postscreened cases (15.3%, 20.0%, and 13.8%, respectively).
“As expected, the majority of SCLC were late-stage cancers, but surprisingly the unfavorable stage distribution was present regardless of whether the cancer was screen-, interval- or post-screen detected,” the researchers noted.
The findings differed from the results of previous smaller studies showing more favorable stage distribution for SCLC cases detected by LDCT, but this difference may be accounted for by various factors including study populations that were smaller, younger, and lacking well-defined risk factors, the researchers said.
“Our findings underscore and provide further granularity to the premise that SCLC is a very aggressive neoplasm, and contrary to smaller prior studies, indicate that LDCT is an ineffective tool to screen for SCLC,” they said. “If early detection of SCLC were to be realistic, it would need to be detected before [lesions] are visible on LDCT,” they emphasized.
The study was supported in part by the National Cancer Institute. The researchers had no financial conflicts to disclose.
SOURCE: Thomas A et al. Chest. 2018 Aug 3. doi: 10.1016/j.chest.2018.07.029.
Early had no significant impact on patient survival, based on data from a randomized trial.
Although early detection of small-cell lung cancer might improve outcomes for patients given the metastatic nature of the disease, previous studies on the value and impact of low-dose CT have been divergent, wrote Anish Thomas, MD, of the National Cancer Institute and his colleagues.
In a study published in Chest, the researchers reviewed characteristics of low-dose CT–detected small cell lung cancer (SCLC) in the randomized National Lung Screening Trial (NLST). The trial compared low-dose CT (LDCT) with chest radiography to assess patients at high risk for developing lung cancer. The study population included adults aged 55-74 years with a history of smoking of at least 30 pack-years, and former smokers who had quit within the past 15 years.
A total of 26,722 participants were randomized to the LDCT treatment arm with a median follow-up of 6.5 years; 143 SCLC cases and 926 non–small cell lung cancer (NSCLC) cases were identified in this group.
Of the 143 SCLC cases, 49 (34.2%) were screen detected, 15 (10.5%) were interval detected and 79 (55.2%) were not screened or were identified post screening.
A cancer was defined as screen detected if it was diagnosed within 1 year of a positive screening or diagnosed after a longer period but with no time gap between diagnostic procedures of more than 1 year; an interval cancer was diagnosed within a year of a negative screen, and non-screened cancers were those found in patients who received no NLST screening or other post screening.
Significantly more of the SCLC cases were at stage III or IV than the NSCLC cases (86% vs. 36%). Unfavorable stage III or IV lung cancers were identified in 80% of screen detected, 86% of interval detected, and 90% of nonscreened or postscreened cases.
In addition, 31 (63%) of the 49 screen-detected SCLC cases had at least one noncalcified nodule in the cancer lobe and 24 (49%) had a single NCN in the cancer lobe.
The 3-year cancer-specific survival rates were not significantly different for screen-detected, interval-detected, and nonscreened or postscreened cases (15.3%, 20.0%, and 13.8%, respectively).
“As expected, the majority of SCLC were late-stage cancers, but surprisingly the unfavorable stage distribution was present regardless of whether the cancer was screen-, interval- or post-screen detected,” the researchers noted.
The findings differed from the results of previous smaller studies showing more favorable stage distribution for SCLC cases detected by LDCT, but this difference may be accounted for by various factors including study populations that were smaller, younger, and lacking well-defined risk factors, the researchers said.
“Our findings underscore and provide further granularity to the premise that SCLC is a very aggressive neoplasm, and contrary to smaller prior studies, indicate that LDCT is an ineffective tool to screen for SCLC,” they said. “If early detection of SCLC were to be realistic, it would need to be detected before [lesions] are visible on LDCT,” they emphasized.
The study was supported in part by the National Cancer Institute. The researchers had no financial conflicts to disclose.
SOURCE: Thomas A et al. Chest. 2018 Aug 3. doi: 10.1016/j.chest.2018.07.029.
Early had no significant impact on patient survival, based on data from a randomized trial.
Although early detection of small-cell lung cancer might improve outcomes for patients given the metastatic nature of the disease, previous studies on the value and impact of low-dose CT have been divergent, wrote Anish Thomas, MD, of the National Cancer Institute and his colleagues.
In a study published in Chest, the researchers reviewed characteristics of low-dose CT–detected small cell lung cancer (SCLC) in the randomized National Lung Screening Trial (NLST). The trial compared low-dose CT (LDCT) with chest radiography to assess patients at high risk for developing lung cancer. The study population included adults aged 55-74 years with a history of smoking of at least 30 pack-years, and former smokers who had quit within the past 15 years.
A total of 26,722 participants were randomized to the LDCT treatment arm with a median follow-up of 6.5 years; 143 SCLC cases and 926 non–small cell lung cancer (NSCLC) cases were identified in this group.
Of the 143 SCLC cases, 49 (34.2%) were screen detected, 15 (10.5%) were interval detected and 79 (55.2%) were not screened or were identified post screening.
A cancer was defined as screen detected if it was diagnosed within 1 year of a positive screening or diagnosed after a longer period but with no time gap between diagnostic procedures of more than 1 year; an interval cancer was diagnosed within a year of a negative screen, and non-screened cancers were those found in patients who received no NLST screening or other post screening.
Significantly more of the SCLC cases were at stage III or IV than the NSCLC cases (86% vs. 36%). Unfavorable stage III or IV lung cancers were identified in 80% of screen detected, 86% of interval detected, and 90% of nonscreened or postscreened cases.
In addition, 31 (63%) of the 49 screen-detected SCLC cases had at least one noncalcified nodule in the cancer lobe and 24 (49%) had a single NCN in the cancer lobe.
The 3-year cancer-specific survival rates were not significantly different for screen-detected, interval-detected, and nonscreened or postscreened cases (15.3%, 20.0%, and 13.8%, respectively).
“As expected, the majority of SCLC were late-stage cancers, but surprisingly the unfavorable stage distribution was present regardless of whether the cancer was screen-, interval- or post-screen detected,” the researchers noted.
The findings differed from the results of previous smaller studies showing more favorable stage distribution for SCLC cases detected by LDCT, but this difference may be accounted for by various factors including study populations that were smaller, younger, and lacking well-defined risk factors, the researchers said.
“Our findings underscore and provide further granularity to the premise that SCLC is a very aggressive neoplasm, and contrary to smaller prior studies, indicate that LDCT is an ineffective tool to screen for SCLC,” they said. “If early detection of SCLC were to be realistic, it would need to be detected before [lesions] are visible on LDCT,” they emphasized.
The study was supported in part by the National Cancer Institute. The researchers had no financial conflicts to disclose.
SOURCE: Thomas A et al. Chest. 2018 Aug 3. doi: 10.1016/j.chest.2018.07.029.
FROM CHEST
Key clinical point: No significant difference in survival occurred in SCLC cancer patients identified early by low-dose CT vs. those identified at interval or post screening.
Major finding: Unfavorable stage III or IV lung cancers were identified in 80% of screen-detected, 86% of interval-detected, and 90% of nonscreened or postscreened cases.
Study details: The data come from 143 small cell lung cancer cases identified in the randomized National Lung Screening Trial.
Disclosures: The study was supported in part by the National Cancer Institute. The researchers had no financial conflicts to disclose.
Source: Thomas A et al. Chest. 2018 Aug 3. doi: 10.1016/j.chest.2018.07.029.