User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
Powered by CHEST Physician, Clinician Reviews, MDedge Family Medicine, Internal Medicine News, and The Journal of Clinical Outcomes Management.
Lung ultrasound predicts need for first-dose surfactant in neonates
Lung ultrasound score (LUS) is an effective means of predicting whether extremely preterm neonates undergoing continuous positive airway pressure (CPAP) treatment for respiratory distress syndrome (RDS) require surfactant, according to results of study published in Pediatrics.
Lucia De Martino, MD, of the division of pediatrics and neonatal critical care at the A. Béclère Medical Centre of the South Paris University Hospital and her associates enrolled 133 neonates of 30 weeks’ gestation or less born between 2015 and 2016. They designed the prospective diagnostic accuracy cohort study, which was conducted in an academic tertiary care referral neonatal ICU.
The first dose of surfactant was administered at a mean 4 hours of life. Those that required further treatment received a second dose of surfactant at a mean 28 hours of life. Each patient received a single lung ultrasound lasting an average of 3 minutes. In each case, the procedure was well tolerated.
In particular, the study demonstrated that LUS is able to accurately predict the need for a first dose and “reveals fair accuracy when it comes to predicting surfactant retreatment,” they observed. The authors speculate that using LUS to predict retreatment is somewhat less reliable because of either the lower number of patients requiring retreatment or reasons related to the biology of surfactant.
“A LUS cutoff value between 6 and 8 provides optimal sensitivity and specificity for predicting the need for the first surfactant dose, whereas a cutoff value of 10 predicts the need for surfactant retreatment,” Dr. De Martino and her colleagues advised.
Of key importance was the finding that LUS is of greatest value to preterm infants less than 34 weeks’ gestation; the authors observed that LUS had significantly lower diagnostic accuracy in infants who were either late term or term. They offered that this outcome was likely attributable to the homogeneous nature of preterm neonates, who are commonly affected by RDS and tend to present with a variety of respiratory disorders and surfactant injury to differing degrees.
At present, international guidelines only recommend surfactant replacement in cases where CPAP has failed, but administering surfactant within the first 2-3 hours of life is key to reducing bronchopulmonary dysplasia as well as the risk of death, they said.
Current surfactant replacement is determined solely by fraction of inspired oxygen cutoff levels, which can result in delayed or even unnecessary treatment. Because neonates who are extremely preterm benefit the most from treatment, “both situations are potentially harmful because late surfactant replacement is less efficacious and giving surfactant when it is not needed may be invasive and seems to increase lung inflammation in animal models,” Dr. De Martino and her associates cautioned.
The authors had no relevant financial disclosures.
SOURCE: De Martino L et al. Pediatrics. 2018. doi: 10.1542/peds.2018-0463.
Point-of-care ultrasound (POCUS) has been recognized for years for its value in assessing sick neonates, but a recent survey showed that less than one-third of U.S. neonatal-perinatal medicine programs actually use bedside ultrasound for health care diagnosis and management. Although its use historically has been confined to pediatric cardiology and radiology, it has gained more of a foothold in acute pediatric care settings, and its use in evaluating neonate lungs is a “relatively new and potentially revolutionary approach,” Maria V. Fraga, MD, and her associates wrote in an accompanying editorial.
A growing body of data over the past 2 decades is available to help radiologists and bedside providers to better understand the applications and limitations of POCUS. Findings in similar studies looking at the use of LUS in neonates “make the article by De Martino et al. so important,” Dr. Fraga and her associates emphasized. Dr. De Martino and her colleagues were able to use POCUS of the lung “to develop reliable predictive models for the need for surfactant treatment and re-dosing” in a group of preterm infants.
Although it would seem reasonable to expect the potential benefits of POCUS to have worldwide application, implementation is inconsistent. Clinicians in Australia, New Zealand, and Canada are trained and use POCUS daily, but this is not the case in other countries such as the United States. Concern over legal risks and training, as well as turf disputes with cardiology and radiology, the lack of clinicians actively using ultrasound, and scarce evidence showing benefit of its use could be to blame.
“The development of a POCUS program requires an accessible dedicated ultrasound machine kept in close proximity to clinical areas, a core group of interested clinicians, and a training and accreditation program with a commitment to continuing professional development,” advised Dr. Fraga and her associates.
“It is important to understand the limitation of bedside ultrasound, which should always be performed for a specific clinical purpose and to answer a clinical question and does not always mandate a full comprehensive study,” they added.
Dr. Fraga and her associates are affiliated with the department of pediatrics at the University of Pennsylvania, Philadelphia. There was no external funding and the authors had no relevant financial disclosures. These comments are adapted from an editorial accompanying the article by De Martino et al. (Pediatrics. 2018. doi: 10.1542/peds.2018-1621).
Point-of-care ultrasound (POCUS) has been recognized for years for its value in assessing sick neonates, but a recent survey showed that less than one-third of U.S. neonatal-perinatal medicine programs actually use bedside ultrasound for health care diagnosis and management. Although its use historically has been confined to pediatric cardiology and radiology, it has gained more of a foothold in acute pediatric care settings, and its use in evaluating neonate lungs is a “relatively new and potentially revolutionary approach,” Maria V. Fraga, MD, and her associates wrote in an accompanying editorial.
A growing body of data over the past 2 decades is available to help radiologists and bedside providers to better understand the applications and limitations of POCUS. Findings in similar studies looking at the use of LUS in neonates “make the article by De Martino et al. so important,” Dr. Fraga and her associates emphasized. Dr. De Martino and her colleagues were able to use POCUS of the lung “to develop reliable predictive models for the need for surfactant treatment and re-dosing” in a group of preterm infants.
Although it would seem reasonable to expect the potential benefits of POCUS to have worldwide application, implementation is inconsistent. Clinicians in Australia, New Zealand, and Canada are trained and use POCUS daily, but this is not the case in other countries such as the United States. Concern over legal risks and training, as well as turf disputes with cardiology and radiology, the lack of clinicians actively using ultrasound, and scarce evidence showing benefit of its use could be to blame.
“The development of a POCUS program requires an accessible dedicated ultrasound machine kept in close proximity to clinical areas, a core group of interested clinicians, and a training and accreditation program with a commitment to continuing professional development,” advised Dr. Fraga and her associates.
“It is important to understand the limitation of bedside ultrasound, which should always be performed for a specific clinical purpose and to answer a clinical question and does not always mandate a full comprehensive study,” they added.
Dr. Fraga and her associates are affiliated with the department of pediatrics at the University of Pennsylvania, Philadelphia. There was no external funding and the authors had no relevant financial disclosures. These comments are adapted from an editorial accompanying the article by De Martino et al. (Pediatrics. 2018. doi: 10.1542/peds.2018-1621).
Point-of-care ultrasound (POCUS) has been recognized for years for its value in assessing sick neonates, but a recent survey showed that less than one-third of U.S. neonatal-perinatal medicine programs actually use bedside ultrasound for health care diagnosis and management. Although its use historically has been confined to pediatric cardiology and radiology, it has gained more of a foothold in acute pediatric care settings, and its use in evaluating neonate lungs is a “relatively new and potentially revolutionary approach,” Maria V. Fraga, MD, and her associates wrote in an accompanying editorial.
A growing body of data over the past 2 decades is available to help radiologists and bedside providers to better understand the applications and limitations of POCUS. Findings in similar studies looking at the use of LUS in neonates “make the article by De Martino et al. so important,” Dr. Fraga and her associates emphasized. Dr. De Martino and her colleagues were able to use POCUS of the lung “to develop reliable predictive models for the need for surfactant treatment and re-dosing” in a group of preterm infants.
Although it would seem reasonable to expect the potential benefits of POCUS to have worldwide application, implementation is inconsistent. Clinicians in Australia, New Zealand, and Canada are trained and use POCUS daily, but this is not the case in other countries such as the United States. Concern over legal risks and training, as well as turf disputes with cardiology and radiology, the lack of clinicians actively using ultrasound, and scarce evidence showing benefit of its use could be to blame.
“The development of a POCUS program requires an accessible dedicated ultrasound machine kept in close proximity to clinical areas, a core group of interested clinicians, and a training and accreditation program with a commitment to continuing professional development,” advised Dr. Fraga and her associates.
“It is important to understand the limitation of bedside ultrasound, which should always be performed for a specific clinical purpose and to answer a clinical question and does not always mandate a full comprehensive study,” they added.
Dr. Fraga and her associates are affiliated with the department of pediatrics at the University of Pennsylvania, Philadelphia. There was no external funding and the authors had no relevant financial disclosures. These comments are adapted from an editorial accompanying the article by De Martino et al. (Pediatrics. 2018. doi: 10.1542/peds.2018-1621).
Lung ultrasound score (LUS) is an effective means of predicting whether extremely preterm neonates undergoing continuous positive airway pressure (CPAP) treatment for respiratory distress syndrome (RDS) require surfactant, according to results of study published in Pediatrics.
Lucia De Martino, MD, of the division of pediatrics and neonatal critical care at the A. Béclère Medical Centre of the South Paris University Hospital and her associates enrolled 133 neonates of 30 weeks’ gestation or less born between 2015 and 2016. They designed the prospective diagnostic accuracy cohort study, which was conducted in an academic tertiary care referral neonatal ICU.
The first dose of surfactant was administered at a mean 4 hours of life. Those that required further treatment received a second dose of surfactant at a mean 28 hours of life. Each patient received a single lung ultrasound lasting an average of 3 minutes. In each case, the procedure was well tolerated.
In particular, the study demonstrated that LUS is able to accurately predict the need for a first dose and “reveals fair accuracy when it comes to predicting surfactant retreatment,” they observed. The authors speculate that using LUS to predict retreatment is somewhat less reliable because of either the lower number of patients requiring retreatment or reasons related to the biology of surfactant.
“A LUS cutoff value between 6 and 8 provides optimal sensitivity and specificity for predicting the need for the first surfactant dose, whereas a cutoff value of 10 predicts the need for surfactant retreatment,” Dr. De Martino and her colleagues advised.
Of key importance was the finding that LUS is of greatest value to preterm infants less than 34 weeks’ gestation; the authors observed that LUS had significantly lower diagnostic accuracy in infants who were either late term or term. They offered that this outcome was likely attributable to the homogeneous nature of preterm neonates, who are commonly affected by RDS and tend to present with a variety of respiratory disorders and surfactant injury to differing degrees.
At present, international guidelines only recommend surfactant replacement in cases where CPAP has failed, but administering surfactant within the first 2-3 hours of life is key to reducing bronchopulmonary dysplasia as well as the risk of death, they said.
Current surfactant replacement is determined solely by fraction of inspired oxygen cutoff levels, which can result in delayed or even unnecessary treatment. Because neonates who are extremely preterm benefit the most from treatment, “both situations are potentially harmful because late surfactant replacement is less efficacious and giving surfactant when it is not needed may be invasive and seems to increase lung inflammation in animal models,” Dr. De Martino and her associates cautioned.
The authors had no relevant financial disclosures.
SOURCE: De Martino L et al. Pediatrics. 2018. doi: 10.1542/peds.2018-0463.
Lung ultrasound score (LUS) is an effective means of predicting whether extremely preterm neonates undergoing continuous positive airway pressure (CPAP) treatment for respiratory distress syndrome (RDS) require surfactant, according to results of study published in Pediatrics.
Lucia De Martino, MD, of the division of pediatrics and neonatal critical care at the A. Béclère Medical Centre of the South Paris University Hospital and her associates enrolled 133 neonates of 30 weeks’ gestation or less born between 2015 and 2016. They designed the prospective diagnostic accuracy cohort study, which was conducted in an academic tertiary care referral neonatal ICU.
The first dose of surfactant was administered at a mean 4 hours of life. Those that required further treatment received a second dose of surfactant at a mean 28 hours of life. Each patient received a single lung ultrasound lasting an average of 3 minutes. In each case, the procedure was well tolerated.
In particular, the study demonstrated that LUS is able to accurately predict the need for a first dose and “reveals fair accuracy when it comes to predicting surfactant retreatment,” they observed. The authors speculate that using LUS to predict retreatment is somewhat less reliable because of either the lower number of patients requiring retreatment or reasons related to the biology of surfactant.
“A LUS cutoff value between 6 and 8 provides optimal sensitivity and specificity for predicting the need for the first surfactant dose, whereas a cutoff value of 10 predicts the need for surfactant retreatment,” Dr. De Martino and her colleagues advised.
Of key importance was the finding that LUS is of greatest value to preterm infants less than 34 weeks’ gestation; the authors observed that LUS had significantly lower diagnostic accuracy in infants who were either late term or term. They offered that this outcome was likely attributable to the homogeneous nature of preterm neonates, who are commonly affected by RDS and tend to present with a variety of respiratory disorders and surfactant injury to differing degrees.
At present, international guidelines only recommend surfactant replacement in cases where CPAP has failed, but administering surfactant within the first 2-3 hours of life is key to reducing bronchopulmonary dysplasia as well as the risk of death, they said.
Current surfactant replacement is determined solely by fraction of inspired oxygen cutoff levels, which can result in delayed or even unnecessary treatment. Because neonates who are extremely preterm benefit the most from treatment, “both situations are potentially harmful because late surfactant replacement is less efficacious and giving surfactant when it is not needed may be invasive and seems to increase lung inflammation in animal models,” Dr. De Martino and her associates cautioned.
The authors had no relevant financial disclosures.
SOURCE: De Martino L et al. Pediatrics. 2018. doi: 10.1542/peds.2018-0463.
FROM PEDIATRICS
Key clinical point:
Major finding: A LUS cutoff value between 6 and 8 provides optimal sensitivity and specificity for predicting the need for the first surfactant dose.
Study details: Prospective cohort diagnostic accuracy study that included 133 infants.
Disclosures: The authors had no relevant financial disclosures.
Source: De Martino L et al. Pediatrics. 2018. doi: 10.1542/peds.2018-0463.
French warn of upsurge in pneumococcal meningitis
MALMO, SWEDEN – A French national study has documented a sharp increase in pneumococcal meningitis since 2015 in children under age 15 years.
The culprit has been identified as serotype 24F, which is not covered by the infant 13-valent conjugate pneumococcal vaccine (PCV13), Naim Ouldali, MD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
The rapid emergence of serotype 24F has been accompanied by a disturbing change in its penicillin susceptibility. Indeed, penicillin resistance was present in only 18% of serotype 24F isolates in France during 2000-2014, then jumped to 74% during 2015-2016, according to Dr. Ouldali of René Descartes University in Paris.
“PCV13 has strongly reduced the pneumococcal meningitis burden in children, but its benefit now seems to be jeopardized, at least in France. So serum 24F could become a major concern in the coming years because of its characteristics. And now the question is, is this emergence an epidemic phenomenon or not? And if it’s confirmed in future studies and in other countries, probably it should drive the development of next-generation PCV formulations,” he said.
Dr. Ouldali presented a population-based interrupted time-series analysis of a nationwide prospective survey conducted in France during 2001-2016. He noted that the Cochrane Collaboration has deemed this study design second only to the randomized controlled trial in terms of quality of evidence.
The study, which included 227 French pediatric wards and 168 microbiology departments, identified 1,778 children under age 15 years with pneumococcal meningitis. This is believed to be more than 60% of all cases that occurred in the country during the study years.
The purpose of the study was to determine the impact of implementation of routine PCV13 as part of the national vaccine strategy. Rates of PCV13 coverage in French children are very high: in excess of 90% during 2015 to 2016.
Implementation of PCV13 led to a dramatic 38% reduction in the monthly incidence of pneumococcal meningitis, from 0.12 cases per 100,000 children before PCV13 to a low of 0.07 cases per 100,000 in December 2014. But after that the rate rebounded sharply, by 2.3% per month during 2015-2016, to a high of 0.13 cases per 100,000 per month by the end of 2016. Drilling down into the data, Dr. Ouldali and his coinvestigators learned that the resurgence of pneumococcal meningitis was due largely to the emergence of serotype 24F.
“This serotype is of particular concern because of two characteristics: First, it is already known to have a high disease potential – one of the highest, along with serotype 12F – and second, this rapid emergence was accompanied by a change in its penicillin susceptibility,” he noted.
Most of the French rebound in pneumococcal meningitis has occurred in children under 2 years of age. Of note, German investigators also have recently reported a rebound in invasive pneumococcal disease in German children under 16 years of age. Non-PCV13 serotypes accounted for 84% of all invasive pneumococcal disease during 2015-2016, with serotypes 10A and 24F leading the way. As in France, most of the resurgence has involved children less than 2 years old. However, unlike in France, most of the German increase has been in nonmeningitis forms of invasive pneumococcal disease (Vaccine. 2018 Jan 25;36[4]:572-7).
In response to a question from a concerned audience member, Dr. Ouldali said that while the penicillin susceptibility of serotype 24F has taken a sharp turn for the worse, cephalosporin susceptibility has not.
“To date, we have not seen any cephalosporin-resistant strains. To date, there is no need to use vancomycin,” he said.
Dr. Ouldali said the next step he and his colleagues plan to take is to see if there is a clonal expansion or a particular underlying genetic pattern which could explain the explosive emergence of 24F.
The study was funded by a research grant from Pfizer and by the French Pediatric Infectious Diseases Group.
MALMO, SWEDEN – A French national study has documented a sharp increase in pneumococcal meningitis since 2015 in children under age 15 years.
The culprit has been identified as serotype 24F, which is not covered by the infant 13-valent conjugate pneumococcal vaccine (PCV13), Naim Ouldali, MD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
The rapid emergence of serotype 24F has been accompanied by a disturbing change in its penicillin susceptibility. Indeed, penicillin resistance was present in only 18% of serotype 24F isolates in France during 2000-2014, then jumped to 74% during 2015-2016, according to Dr. Ouldali of René Descartes University in Paris.
“PCV13 has strongly reduced the pneumococcal meningitis burden in children, but its benefit now seems to be jeopardized, at least in France. So serum 24F could become a major concern in the coming years because of its characteristics. And now the question is, is this emergence an epidemic phenomenon or not? And if it’s confirmed in future studies and in other countries, probably it should drive the development of next-generation PCV formulations,” he said.
Dr. Ouldali presented a population-based interrupted time-series analysis of a nationwide prospective survey conducted in France during 2001-2016. He noted that the Cochrane Collaboration has deemed this study design second only to the randomized controlled trial in terms of quality of evidence.
The study, which included 227 French pediatric wards and 168 microbiology departments, identified 1,778 children under age 15 years with pneumococcal meningitis. This is believed to be more than 60% of all cases that occurred in the country during the study years.
The purpose of the study was to determine the impact of implementation of routine PCV13 as part of the national vaccine strategy. Rates of PCV13 coverage in French children are very high: in excess of 90% during 2015 to 2016.
Implementation of PCV13 led to a dramatic 38% reduction in the monthly incidence of pneumococcal meningitis, from 0.12 cases per 100,000 children before PCV13 to a low of 0.07 cases per 100,000 in December 2014. But after that the rate rebounded sharply, by 2.3% per month during 2015-2016, to a high of 0.13 cases per 100,000 per month by the end of 2016. Drilling down into the data, Dr. Ouldali and his coinvestigators learned that the resurgence of pneumococcal meningitis was due largely to the emergence of serotype 24F.
“This serotype is of particular concern because of two characteristics: First, it is already known to have a high disease potential – one of the highest, along with serotype 12F – and second, this rapid emergence was accompanied by a change in its penicillin susceptibility,” he noted.
Most of the French rebound in pneumococcal meningitis has occurred in children under 2 years of age. Of note, German investigators also have recently reported a rebound in invasive pneumococcal disease in German children under 16 years of age. Non-PCV13 serotypes accounted for 84% of all invasive pneumococcal disease during 2015-2016, with serotypes 10A and 24F leading the way. As in France, most of the resurgence has involved children less than 2 years old. However, unlike in France, most of the German increase has been in nonmeningitis forms of invasive pneumococcal disease (Vaccine. 2018 Jan 25;36[4]:572-7).
In response to a question from a concerned audience member, Dr. Ouldali said that while the penicillin susceptibility of serotype 24F has taken a sharp turn for the worse, cephalosporin susceptibility has not.
“To date, we have not seen any cephalosporin-resistant strains. To date, there is no need to use vancomycin,” he said.
Dr. Ouldali said the next step he and his colleagues plan to take is to see if there is a clonal expansion or a particular underlying genetic pattern which could explain the explosive emergence of 24F.
The study was funded by a research grant from Pfizer and by the French Pediatric Infectious Diseases Group.
MALMO, SWEDEN – A French national study has documented a sharp increase in pneumococcal meningitis since 2015 in children under age 15 years.
The culprit has been identified as serotype 24F, which is not covered by the infant 13-valent conjugate pneumococcal vaccine (PCV13), Naim Ouldali, MD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
The rapid emergence of serotype 24F has been accompanied by a disturbing change in its penicillin susceptibility. Indeed, penicillin resistance was present in only 18% of serotype 24F isolates in France during 2000-2014, then jumped to 74% during 2015-2016, according to Dr. Ouldali of René Descartes University in Paris.
“PCV13 has strongly reduced the pneumococcal meningitis burden in children, but its benefit now seems to be jeopardized, at least in France. So serum 24F could become a major concern in the coming years because of its characteristics. And now the question is, is this emergence an epidemic phenomenon or not? And if it’s confirmed in future studies and in other countries, probably it should drive the development of next-generation PCV formulations,” he said.
Dr. Ouldali presented a population-based interrupted time-series analysis of a nationwide prospective survey conducted in France during 2001-2016. He noted that the Cochrane Collaboration has deemed this study design second only to the randomized controlled trial in terms of quality of evidence.
The study, which included 227 French pediatric wards and 168 microbiology departments, identified 1,778 children under age 15 years with pneumococcal meningitis. This is believed to be more than 60% of all cases that occurred in the country during the study years.
The purpose of the study was to determine the impact of implementation of routine PCV13 as part of the national vaccine strategy. Rates of PCV13 coverage in French children are very high: in excess of 90% during 2015 to 2016.
Implementation of PCV13 led to a dramatic 38% reduction in the monthly incidence of pneumococcal meningitis, from 0.12 cases per 100,000 children before PCV13 to a low of 0.07 cases per 100,000 in December 2014. But after that the rate rebounded sharply, by 2.3% per month during 2015-2016, to a high of 0.13 cases per 100,000 per month by the end of 2016. Drilling down into the data, Dr. Ouldali and his coinvestigators learned that the resurgence of pneumococcal meningitis was due largely to the emergence of serotype 24F.
“This serotype is of particular concern because of two characteristics: First, it is already known to have a high disease potential – one of the highest, along with serotype 12F – and second, this rapid emergence was accompanied by a change in its penicillin susceptibility,” he noted.
Most of the French rebound in pneumococcal meningitis has occurred in children under 2 years of age. Of note, German investigators also have recently reported a rebound in invasive pneumococcal disease in German children under 16 years of age. Non-PCV13 serotypes accounted for 84% of all invasive pneumococcal disease during 2015-2016, with serotypes 10A and 24F leading the way. As in France, most of the resurgence has involved children less than 2 years old. However, unlike in France, most of the German increase has been in nonmeningitis forms of invasive pneumococcal disease (Vaccine. 2018 Jan 25;36[4]:572-7).
In response to a question from a concerned audience member, Dr. Ouldali said that while the penicillin susceptibility of serotype 24F has taken a sharp turn for the worse, cephalosporin susceptibility has not.
“To date, we have not seen any cephalosporin-resistant strains. To date, there is no need to use vancomycin,” he said.
Dr. Ouldali said the next step he and his colleagues plan to take is to see if there is a clonal expansion or a particular underlying genetic pattern which could explain the explosive emergence of 24F.
The study was funded by a research grant from Pfizer and by the French Pediatric Infectious Diseases Group.
REPORTING FROM ESPID 2018
Key clinical point:
Major finding: The incidence of pneumococcal meningitis in French children jumped by 2.3% per month during 2015-2016.
Study details: This population-based interrupted time-series analysis included all 1,778 cases of pneumococcal meningitis in children under age 15 years during 2001-2016 in 227 French pediatric wards.
Disclosures: The study was funded by a grant from Pfizer and by the French Pediatric Infectious Diseases Group.
Shared decision making falls short for lung cancer screening
A small study of discussions between clinicians and patients about has highlighted a lack of shared decision making and information about potential harms.
“Our findings are consistent with increasingly robust evidence that patients, members of the public, and clinicians tend to overestimate the benefits and underestimate the harms of medical interventions, including treatments, tests, or screening tests,” wrote Alison T. Brenner, PhD, and her colleagues at the University of North Carolina at Chapel Hill, in a presentation of the findings in JAMA Internal Medicine.
The researchers transcribed conversations between 14 patients – who were eligible for lung cancer screening because of their age – and their primary care or pulmonary care physicians. They found that not one physician adequately explained false positives or their consequences, such as the possibility of additional imaging and invasive diagnostic procedures, nor did any discuss the potential for diagnosis and treatment of cancer that would not have affected the individual during his or her lifetime (overdiagnosis).
Researchers used a 12-item scoring system for physician behaviors, with 0-4 points allocated to each item. The items included telling patients there was more than one way to deal with the identified problem, explaining the pros and cons of the available options, exploring patients’ fears and concerns, and offering the patient clear opportunities to ask questions.
Mean scores for each item ranged from 0 to 0.79. Two conversations met the baseline skill criteria – a score of two points – for one item each, two other conversations met the baseline skill criteria for two items. But for 8 of the 12 items, not one conversation achieved even a baseline skill score. The mean total visit length was 13:07 minutes, and the mean time spent discussing LCS was 0:59 minute (range, 0:16-2:19 minutes).
“Although experts disagree on how well the existing evidence suggests an overall net benefit of LCS [lung cancer screening], consensus has emerged on the importance of shared decision making,” wrote the investigators. Current U.S. Preventive Services Task Force recommendations stress that lung cancer screening should not occur without a shared decision-making process, including a thorough discussion of benefits and harms.
The authors said that, while their study was small, it did raise concerns that shared decision making in practice is a long way from what is recommended by the guidelines.
“The fact that the main drivers of harms from LCS (false positives and their sequelae, as well as overdiagnosis) were not adequately explained by physicians is troubling,” they wrote. “However, these findings are consistent with other evidence that discussions between patients and physicians regarding preference-sensitive cancer screening decisions are imbalanced with respect to explaining the pros and cons.”
Based on these findings, the authors called for urgent discussions between clinical leaders, policy makers, and researchers about how to involve patients more meaningfully in discussions about lung cancer screening.
“Until more is known, we believe that guideline and policy makers should not assume that recommending SDM [shared decision-making] for cancer-screening decisions with a ‘tenuous balance of benefits and harms,’ like LCS, will protect patients who would value avoiding screening harms.”
The study was supported by the North Carolina Translational and Clinical Sciences Institute and the National Cancer Institute. No conflicts of interest were declared.
SOURCE: Brenner A et al. JAMA Intern Med. 2018; Aug 13. doi: 10.1001/jamainternmed.2018.3054.
The results of this first real-world study of the U.S. Preventive Services Task Force recommendations on lung cancer screening – which comes 4 years after the recommendations were made – are disappointing. Even the highest-scoring conversations made no mention of possible harms, such as a 98% false-positive rate, additional testing, and the small increased cancer risk from radiation.
Rita F. Redberg, MD, is from the department of medicine in the division of cardiology at the University of California, San Francisco, and the editor of JAMA Internal Medicine. These comments are taken from an accompanying editorial (JAMA Int Med. 2018 Aug 13. doi: 10.1001/jamainternmed.2018.3527). Dr. Redberg chaired the April 2014 Medicare Evidence Development & Coverage Advisory Committee meeting on lung cancer screening.
The results of this first real-world study of the U.S. Preventive Services Task Force recommendations on lung cancer screening – which comes 4 years after the recommendations were made – are disappointing. Even the highest-scoring conversations made no mention of possible harms, such as a 98% false-positive rate, additional testing, and the small increased cancer risk from radiation.
Rita F. Redberg, MD, is from the department of medicine in the division of cardiology at the University of California, San Francisco, and the editor of JAMA Internal Medicine. These comments are taken from an accompanying editorial (JAMA Int Med. 2018 Aug 13. doi: 10.1001/jamainternmed.2018.3527). Dr. Redberg chaired the April 2014 Medicare Evidence Development & Coverage Advisory Committee meeting on lung cancer screening.
The results of this first real-world study of the U.S. Preventive Services Task Force recommendations on lung cancer screening – which comes 4 years after the recommendations were made – are disappointing. Even the highest-scoring conversations made no mention of possible harms, such as a 98% false-positive rate, additional testing, and the small increased cancer risk from radiation.
Rita F. Redberg, MD, is from the department of medicine in the division of cardiology at the University of California, San Francisco, and the editor of JAMA Internal Medicine. These comments are taken from an accompanying editorial (JAMA Int Med. 2018 Aug 13. doi: 10.1001/jamainternmed.2018.3527). Dr. Redberg chaired the April 2014 Medicare Evidence Development & Coverage Advisory Committee meeting on lung cancer screening.
A small study of discussions between clinicians and patients about has highlighted a lack of shared decision making and information about potential harms.
“Our findings are consistent with increasingly robust evidence that patients, members of the public, and clinicians tend to overestimate the benefits and underestimate the harms of medical interventions, including treatments, tests, or screening tests,” wrote Alison T. Brenner, PhD, and her colleagues at the University of North Carolina at Chapel Hill, in a presentation of the findings in JAMA Internal Medicine.
The researchers transcribed conversations between 14 patients – who were eligible for lung cancer screening because of their age – and their primary care or pulmonary care physicians. They found that not one physician adequately explained false positives or their consequences, such as the possibility of additional imaging and invasive diagnostic procedures, nor did any discuss the potential for diagnosis and treatment of cancer that would not have affected the individual during his or her lifetime (overdiagnosis).
Researchers used a 12-item scoring system for physician behaviors, with 0-4 points allocated to each item. The items included telling patients there was more than one way to deal with the identified problem, explaining the pros and cons of the available options, exploring patients’ fears and concerns, and offering the patient clear opportunities to ask questions.
Mean scores for each item ranged from 0 to 0.79. Two conversations met the baseline skill criteria – a score of two points – for one item each, two other conversations met the baseline skill criteria for two items. But for 8 of the 12 items, not one conversation achieved even a baseline skill score. The mean total visit length was 13:07 minutes, and the mean time spent discussing LCS was 0:59 minute (range, 0:16-2:19 minutes).
“Although experts disagree on how well the existing evidence suggests an overall net benefit of LCS [lung cancer screening], consensus has emerged on the importance of shared decision making,” wrote the investigators. Current U.S. Preventive Services Task Force recommendations stress that lung cancer screening should not occur without a shared decision-making process, including a thorough discussion of benefits and harms.
The authors said that, while their study was small, it did raise concerns that shared decision making in practice is a long way from what is recommended by the guidelines.
“The fact that the main drivers of harms from LCS (false positives and their sequelae, as well as overdiagnosis) were not adequately explained by physicians is troubling,” they wrote. “However, these findings are consistent with other evidence that discussions between patients and physicians regarding preference-sensitive cancer screening decisions are imbalanced with respect to explaining the pros and cons.”
Based on these findings, the authors called for urgent discussions between clinical leaders, policy makers, and researchers about how to involve patients more meaningfully in discussions about lung cancer screening.
“Until more is known, we believe that guideline and policy makers should not assume that recommending SDM [shared decision-making] for cancer-screening decisions with a ‘tenuous balance of benefits and harms,’ like LCS, will protect patients who would value avoiding screening harms.”
The study was supported by the North Carolina Translational and Clinical Sciences Institute and the National Cancer Institute. No conflicts of interest were declared.
SOURCE: Brenner A et al. JAMA Intern Med. 2018; Aug 13. doi: 10.1001/jamainternmed.2018.3054.
A small study of discussions between clinicians and patients about has highlighted a lack of shared decision making and information about potential harms.
“Our findings are consistent with increasingly robust evidence that patients, members of the public, and clinicians tend to overestimate the benefits and underestimate the harms of medical interventions, including treatments, tests, or screening tests,” wrote Alison T. Brenner, PhD, and her colleagues at the University of North Carolina at Chapel Hill, in a presentation of the findings in JAMA Internal Medicine.
The researchers transcribed conversations between 14 patients – who were eligible for lung cancer screening because of their age – and their primary care or pulmonary care physicians. They found that not one physician adequately explained false positives or their consequences, such as the possibility of additional imaging and invasive diagnostic procedures, nor did any discuss the potential for diagnosis and treatment of cancer that would not have affected the individual during his or her lifetime (overdiagnosis).
Researchers used a 12-item scoring system for physician behaviors, with 0-4 points allocated to each item. The items included telling patients there was more than one way to deal with the identified problem, explaining the pros and cons of the available options, exploring patients’ fears and concerns, and offering the patient clear opportunities to ask questions.
Mean scores for each item ranged from 0 to 0.79. Two conversations met the baseline skill criteria – a score of two points – for one item each, two other conversations met the baseline skill criteria for two items. But for 8 of the 12 items, not one conversation achieved even a baseline skill score. The mean total visit length was 13:07 minutes, and the mean time spent discussing LCS was 0:59 minute (range, 0:16-2:19 minutes).
“Although experts disagree on how well the existing evidence suggests an overall net benefit of LCS [lung cancer screening], consensus has emerged on the importance of shared decision making,” wrote the investigators. Current U.S. Preventive Services Task Force recommendations stress that lung cancer screening should not occur without a shared decision-making process, including a thorough discussion of benefits and harms.
The authors said that, while their study was small, it did raise concerns that shared decision making in practice is a long way from what is recommended by the guidelines.
“The fact that the main drivers of harms from LCS (false positives and their sequelae, as well as overdiagnosis) were not adequately explained by physicians is troubling,” they wrote. “However, these findings are consistent with other evidence that discussions between patients and physicians regarding preference-sensitive cancer screening decisions are imbalanced with respect to explaining the pros and cons.”
Based on these findings, the authors called for urgent discussions between clinical leaders, policy makers, and researchers about how to involve patients more meaningfully in discussions about lung cancer screening.
“Until more is known, we believe that guideline and policy makers should not assume that recommending SDM [shared decision-making] for cancer-screening decisions with a ‘tenuous balance of benefits and harms,’ like LCS, will protect patients who would value avoiding screening harms.”
The study was supported by the North Carolina Translational and Clinical Sciences Institute and the National Cancer Institute. No conflicts of interest were declared.
SOURCE: Brenner A et al. JAMA Intern Med. 2018; Aug 13. doi: 10.1001/jamainternmed.2018.3054.
FROM JAMA INTERNAL MEDICINE
Key clinical point: Few clinicians are following recommendations about shared decision-making with patients regarding lung cancer screening.
Major finding: No physicians in the study explained false positives and their risks in discussions with patients about lung cancer screening.
Study details: Content analysis of 14 recorded consultations about lung cancer screening.
Disclosures: The study was supported by the North Carolina Translational and Clinical Sciences Institute and the National Cancer Institute. No conflicts of interest were declared.
Source: Brenner A et al. JAMA Intern Med. 2018; Aug 13. doi: 10.1001/jamainternmed.2018.3054.
No increase in autism risk with prenatal Tdap
A retrospective cohort study in more than 80,000 children has found no evidence of an increased risk of autism spectrum disorder associated with prenatal tetanus, diphtheria, and acellular pertussis (Tdap) immunization.
Of 81,993 children born between 2011 and 2014, 1,341 children (1.6%) were diagnosed with autism spectrum disorder. The incidence of autism spectrum disorder was 3.78 per 1,000 person-years in the Tdap-vaccinated group, and 4.05 per 1,000 person years in the unvaccinated group, representing an unadjusted hazard ratio of 0.98 and an adjusted hazard ratio of 0.85. This was consistent across all the birth cohorts.
Prenatal immunization rates with the prenatal Tdap vaccine ranged from 26% of the 2012 birth cohort to 79% of the 2014 birth cohort, and mean gestational age at vaccination was 28 weeks.
Tracy A. Becerra-Culqui, PhD, MPH, and colleagues of the department of research and evaluation at Kaiser Permanente Southern California, Pasadena, said this was the first study to look at the risk of autism spectrum disorder after maternal exposure to the Tdap vaccine, to their knowledge. “Our results potentially indicate that the maternal Tdap vaccine affects immune trajectories protecting infants against infections that would otherwise lead to neurodevelopmental alterations.”
They highlighted several strengths of their study. One was that maternal Tdap vaccination and information on autism spectrum disorder both were derived from EHRs and therefore not subject to recall bias. The study, published online in Pediatrics, also included children diagnosed with autism spectrum disorder from age 1 year onwards, reflecting the latest evidence on screening and diagnosis of autism.
“Our weighting procedures enabled us to balance the Tdap-exposed and -unexposed groups to compare two populations that were comparable in important measured confounding factors,” Dr. Becerra-Culqui and associates noted.
The investigators found that women who received the Tdap vaccine during pregnancy were more likely to be Asian American or Pacific Islander, to have a bachelor’s degree or higher, be nulliparous, to have also been vaccinated prenatally against influenza, and to deliver at term, compared with unvaccinated women.
However the authors did note that their follow-up was limited to 6.5 years for the earliest birth cohort, and 3.5 years for the latest cohort, so they may not have picked up children who received a later diagnosis of autism spectrum disorder.
The study was supported by Kaiser Permanente Southern California. Five authors declared funding from GlaxoSmithKline, Bayer AG, or the Centers for Disease Control and Prevention for unrelated or separate studies.
SOURCE: Becerra-Culqui T et al. Pediatrics. 2018;142(3):e20180120.
A retrospective cohort study in more than 80,000 children has found no evidence of an increased risk of autism spectrum disorder associated with prenatal tetanus, diphtheria, and acellular pertussis (Tdap) immunization.
Of 81,993 children born between 2011 and 2014, 1,341 children (1.6%) were diagnosed with autism spectrum disorder. The incidence of autism spectrum disorder was 3.78 per 1,000 person-years in the Tdap-vaccinated group, and 4.05 per 1,000 person years in the unvaccinated group, representing an unadjusted hazard ratio of 0.98 and an adjusted hazard ratio of 0.85. This was consistent across all the birth cohorts.
Prenatal immunization rates with the prenatal Tdap vaccine ranged from 26% of the 2012 birth cohort to 79% of the 2014 birth cohort, and mean gestational age at vaccination was 28 weeks.
Tracy A. Becerra-Culqui, PhD, MPH, and colleagues of the department of research and evaluation at Kaiser Permanente Southern California, Pasadena, said this was the first study to look at the risk of autism spectrum disorder after maternal exposure to the Tdap vaccine, to their knowledge. “Our results potentially indicate that the maternal Tdap vaccine affects immune trajectories protecting infants against infections that would otherwise lead to neurodevelopmental alterations.”
They highlighted several strengths of their study. One was that maternal Tdap vaccination and information on autism spectrum disorder both were derived from EHRs and therefore not subject to recall bias. The study, published online in Pediatrics, also included children diagnosed with autism spectrum disorder from age 1 year onwards, reflecting the latest evidence on screening and diagnosis of autism.
“Our weighting procedures enabled us to balance the Tdap-exposed and -unexposed groups to compare two populations that were comparable in important measured confounding factors,” Dr. Becerra-Culqui and associates noted.
The investigators found that women who received the Tdap vaccine during pregnancy were more likely to be Asian American or Pacific Islander, to have a bachelor’s degree or higher, be nulliparous, to have also been vaccinated prenatally against influenza, and to deliver at term, compared with unvaccinated women.
However the authors did note that their follow-up was limited to 6.5 years for the earliest birth cohort, and 3.5 years for the latest cohort, so they may not have picked up children who received a later diagnosis of autism spectrum disorder.
The study was supported by Kaiser Permanente Southern California. Five authors declared funding from GlaxoSmithKline, Bayer AG, or the Centers for Disease Control and Prevention for unrelated or separate studies.
SOURCE: Becerra-Culqui T et al. Pediatrics. 2018;142(3):e20180120.
A retrospective cohort study in more than 80,000 children has found no evidence of an increased risk of autism spectrum disorder associated with prenatal tetanus, diphtheria, and acellular pertussis (Tdap) immunization.
Of 81,993 children born between 2011 and 2014, 1,341 children (1.6%) were diagnosed with autism spectrum disorder. The incidence of autism spectrum disorder was 3.78 per 1,000 person-years in the Tdap-vaccinated group, and 4.05 per 1,000 person years in the unvaccinated group, representing an unadjusted hazard ratio of 0.98 and an adjusted hazard ratio of 0.85. This was consistent across all the birth cohorts.
Prenatal immunization rates with the prenatal Tdap vaccine ranged from 26% of the 2012 birth cohort to 79% of the 2014 birth cohort, and mean gestational age at vaccination was 28 weeks.
Tracy A. Becerra-Culqui, PhD, MPH, and colleagues of the department of research and evaluation at Kaiser Permanente Southern California, Pasadena, said this was the first study to look at the risk of autism spectrum disorder after maternal exposure to the Tdap vaccine, to their knowledge. “Our results potentially indicate that the maternal Tdap vaccine affects immune trajectories protecting infants against infections that would otherwise lead to neurodevelopmental alterations.”
They highlighted several strengths of their study. One was that maternal Tdap vaccination and information on autism spectrum disorder both were derived from EHRs and therefore not subject to recall bias. The study, published online in Pediatrics, also included children diagnosed with autism spectrum disorder from age 1 year onwards, reflecting the latest evidence on screening and diagnosis of autism.
“Our weighting procedures enabled us to balance the Tdap-exposed and -unexposed groups to compare two populations that were comparable in important measured confounding factors,” Dr. Becerra-Culqui and associates noted.
The investigators found that women who received the Tdap vaccine during pregnancy were more likely to be Asian American or Pacific Islander, to have a bachelor’s degree or higher, be nulliparous, to have also been vaccinated prenatally against influenza, and to deliver at term, compared with unvaccinated women.
However the authors did note that their follow-up was limited to 6.5 years for the earliest birth cohort, and 3.5 years for the latest cohort, so they may not have picked up children who received a later diagnosis of autism spectrum disorder.
The study was supported by Kaiser Permanente Southern California. Five authors declared funding from GlaxoSmithKline, Bayer AG, or the Centers for Disease Control and Prevention for unrelated or separate studies.
SOURCE: Becerra-Culqui T et al. Pediatrics. 2018;142(3):e20180120.
FROM PEDIATRICS
Key clinical point:
Major finding: The adjusted hazard ratio for autism spectrum disorder in children exposed to the prenatal Tdap vaccine is 0.98, compared with unvaccinated children.
Study details: A retrospective cohort study in 81,993 children exposed to the prenatal Tdap vaccine.
Disclosures: The study was supported by Kaiser Permanente Southern California. Five authors declared funding from GlaxoSmithKline, Bayer AG, or the Centers for Disease Control and Prevention for unrelated or separate studies.
Source: Becerra-Culqui T et al. Pediatrics. 2018;142(3):e20180120.
Secondhand smoke drives ED visits for teens
Teens who were exposed to any type of and to have more such visits, compared with unexposed controls, based on data from more than 7,000 adolescents.
Approximately 35% of U.S. teens spent more than an hour exposed to secondhand smoke in a given week, wrote Ashley L. Merianos, PhD, of the University of Cincinnati and her colleagues.
In a study published in Pediatrics, the researchers conducted a secondary analysis of nonsmoking adolescents aged 12-17 years who had not been diagnosed with asthma and who were part of the PATH (Population Assessment of Tobacco and Health) study, a longitudinal cohort trial of tobacco use behavior and related health outcomes in adolescents and adults in the United States. The data were collected between Oct. 3, 2014, and Oct. 30, 2015. The researchers reviewed three main measures of tobacco smoke exposure (TSE): living with a smoker, being exposed to secondhand smoke at home, and being exposed to secondhand smoke for an hour or more in the past 7 days.
Overall, teens who lived with a smoker, had secondary exposure at home, or had at least 1 hour of TSE had significantly more emergency department and/or urgent care visits (mean ranged from 1.62 to 1.65), compared with unexposed peers (mean visits ranged from 1.42 to 1.48). Those who both lived with a smoker and had at least 1 hour of TSE exposure were significantly more likely to visit an ED or urgent care center.
In addition, teens who lived with a smoker, had secondary exposure at home, and had at least 1 hour of TSE were significantly more likely than were unexposed peers to report shortness of breath, difficulty exercising, wheezing during and after exercise, and a dry cough at night (P less than .001).
The researchers also assessed other health indicators, and found that teens with TSE exposure were significantly less likely than were unexposed peers to report very good or excellent health and were approximately 1.50 times more likely than unexposed peers to report missing school because of poor health.
The results were limited by several factors including the use of self-reports of TSE and parent reports of emergency or urgent care visits, and by the inclusion only of other public use variables in the PATH database, the researchers noted. But they adjusted for potentially confounding factors such as household income level, parent education, and health insurance status. However, “Because adolescents are high users of EDs and/or [urgent care] for primary care reasons, these venues are high-volume settings that should be used to offer interventions to adolescents with TSE and their families,” they said.
The researchers had no financial conflicts to disclose. The study was funded by the National Institutes of Health via the National Institute on Drug Abuse and the Eunice Kennedy Shriver National Institute of Child Health and Human Development.
SOURCE: Merianos AL et al. Pediatrics 2018 Aug 6. doi: org/10.1542/peds.2018-0266.
Teens who were exposed to any type of and to have more such visits, compared with unexposed controls, based on data from more than 7,000 adolescents.
Approximately 35% of U.S. teens spent more than an hour exposed to secondhand smoke in a given week, wrote Ashley L. Merianos, PhD, of the University of Cincinnati and her colleagues.
In a study published in Pediatrics, the researchers conducted a secondary analysis of nonsmoking adolescents aged 12-17 years who had not been diagnosed with asthma and who were part of the PATH (Population Assessment of Tobacco and Health) study, a longitudinal cohort trial of tobacco use behavior and related health outcomes in adolescents and adults in the United States. The data were collected between Oct. 3, 2014, and Oct. 30, 2015. The researchers reviewed three main measures of tobacco smoke exposure (TSE): living with a smoker, being exposed to secondhand smoke at home, and being exposed to secondhand smoke for an hour or more in the past 7 days.
Overall, teens who lived with a smoker, had secondary exposure at home, or had at least 1 hour of TSE had significantly more emergency department and/or urgent care visits (mean ranged from 1.62 to 1.65), compared with unexposed peers (mean visits ranged from 1.42 to 1.48). Those who both lived with a smoker and had at least 1 hour of TSE exposure were significantly more likely to visit an ED or urgent care center.
In addition, teens who lived with a smoker, had secondary exposure at home, and had at least 1 hour of TSE were significantly more likely than were unexposed peers to report shortness of breath, difficulty exercising, wheezing during and after exercise, and a dry cough at night (P less than .001).
The researchers also assessed other health indicators, and found that teens with TSE exposure were significantly less likely than were unexposed peers to report very good or excellent health and were approximately 1.50 times more likely than unexposed peers to report missing school because of poor health.
The results were limited by several factors including the use of self-reports of TSE and parent reports of emergency or urgent care visits, and by the inclusion only of other public use variables in the PATH database, the researchers noted. But they adjusted for potentially confounding factors such as household income level, parent education, and health insurance status. However, “Because adolescents are high users of EDs and/or [urgent care] for primary care reasons, these venues are high-volume settings that should be used to offer interventions to adolescents with TSE and their families,” they said.
The researchers had no financial conflicts to disclose. The study was funded by the National Institutes of Health via the National Institute on Drug Abuse and the Eunice Kennedy Shriver National Institute of Child Health and Human Development.
SOURCE: Merianos AL et al. Pediatrics 2018 Aug 6. doi: org/10.1542/peds.2018-0266.
Teens who were exposed to any type of and to have more such visits, compared with unexposed controls, based on data from more than 7,000 adolescents.
Approximately 35% of U.S. teens spent more than an hour exposed to secondhand smoke in a given week, wrote Ashley L. Merianos, PhD, of the University of Cincinnati and her colleagues.
In a study published in Pediatrics, the researchers conducted a secondary analysis of nonsmoking adolescents aged 12-17 years who had not been diagnosed with asthma and who were part of the PATH (Population Assessment of Tobacco and Health) study, a longitudinal cohort trial of tobacco use behavior and related health outcomes in adolescents and adults in the United States. The data were collected between Oct. 3, 2014, and Oct. 30, 2015. The researchers reviewed three main measures of tobacco smoke exposure (TSE): living with a smoker, being exposed to secondhand smoke at home, and being exposed to secondhand smoke for an hour or more in the past 7 days.
Overall, teens who lived with a smoker, had secondary exposure at home, or had at least 1 hour of TSE had significantly more emergency department and/or urgent care visits (mean ranged from 1.62 to 1.65), compared with unexposed peers (mean visits ranged from 1.42 to 1.48). Those who both lived with a smoker and had at least 1 hour of TSE exposure were significantly more likely to visit an ED or urgent care center.
In addition, teens who lived with a smoker, had secondary exposure at home, and had at least 1 hour of TSE were significantly more likely than were unexposed peers to report shortness of breath, difficulty exercising, wheezing during and after exercise, and a dry cough at night (P less than .001).
The researchers also assessed other health indicators, and found that teens with TSE exposure were significantly less likely than were unexposed peers to report very good or excellent health and were approximately 1.50 times more likely than unexposed peers to report missing school because of poor health.
The results were limited by several factors including the use of self-reports of TSE and parent reports of emergency or urgent care visits, and by the inclusion only of other public use variables in the PATH database, the researchers noted. But they adjusted for potentially confounding factors such as household income level, parent education, and health insurance status. However, “Because adolescents are high users of EDs and/or [urgent care] for primary care reasons, these venues are high-volume settings that should be used to offer interventions to adolescents with TSE and their families,” they said.
The researchers had no financial conflicts to disclose. The study was funded by the National Institutes of Health via the National Institute on Drug Abuse and the Eunice Kennedy Shriver National Institute of Child Health and Human Development.
SOURCE: Merianos AL et al. Pediatrics 2018 Aug 6. doi: org/10.1542/peds.2018-0266.
FROM PEDIATRICS
Treating sleep disorders in chronic opioid users
BALTIMORE – Given the prevalence of opioid use in the general population, sleep specialists need to be alert to the effects of opioid use on sleep and the link between chronic use and sleep disorders, a pulmonologist recommended at the annual meeting of the Associated Professional Sleep Societies.
Chronic opioid use has multiple effects on sleep that render continuous positive airway pressure (CPAP) all but ineffective, said Bernardo J. Selim, MD, of Mayo Clinic, Rochester, Minn. Characteristic signs of the effects of chronic opioid use on sleep include ataxic central sleep apnea (CSA) and sustained hypoxemia, for which CPAP is generally not effective. Obtaining arterial blood gas measures in these patients is also important to rule out a hypoventilative condition, he added.
In his review of opioid-induced sleep disorders, Dr. Selim cited a small “landmark” study of 24 chronic pain patients on opioids that found 46% had sleep disordered breathing and that the risk rose with the morphine equivalent dose they were taking (J Clin Sleep Med. 2014 Aug 15; 10[8]:847-52).
A meta-analysis also found a dose-dependent relationship with the severity of CSA in patients on opioids, Dr. Selim noted (Anesth Analg. 2015 Jun;120[6]:1273-85). The prevalence of CSA was 24% in the study, which defined two risk factors for CSA severity: a morphine equivalent dose exceeding 200 mg/day and a low or normal body mass index.
Dr. Selim noted that opioids reduce respiration rate more than tidal volume and cause changes to respiratory rhythm. “[Opioids] decrease hypercapnia but increase hypoxic ventilatory response, decrease the arousal index, decrease upper-airway muscle tone, and decrease and also act on chest and abdominal wall compliance.”
Further, different opioids and injection methods can influence breathing. For example, REM and slow-wave sleep decreased across all three categories – intravenous morphine, oral morphine or methadone, and heroin use.
Sleep specialists should be aware that all opioid receptor agonists, whether legal or illegal, have respiratory side effects, Dr. Selim said. “They can present in any way, in any form – CSA, obstructive sleep apnea [OSA], ataxic breathing or sustained hypoxemia. Most of the time [respiratory side effects] present as a combination of complex respiratory patterns.”
In one meta-analysis, CSA was significantly more prevalent in OSA patients on opioids than it was in nonusers, Dr. Selim said, with increased sleep apnea severity as well (J Clin Sleep Med. 2016 Apr 15;12[4]:617-25). Another study found that ataxic breathing was more frequent in non-REM sleep in chronic opioid users (odds ratio, 15.4; P = .017; J Clin Sleep Med. 2007 Aug 15;3[5]:455-61).
The key rule for treating sleep disorders in opioid-dependent patients is to change to nonopioid analgesics, Dr. Selim said. In that regard, ampakines are experimental drugs which have been shown to improve opioid-induced ventilation without loss of the analgesic effect (Clin Pharmacol Ther. 2010 Feb;87[2]:204-11). “Ampakines modulate the action of the glutamate neurotransmitter, decreasing opiate-induced respiratory depression,” Dr. Selim said. An emerging technology, adaptive servo-ventilation (ASV), has been as effective in the treatment of central and complex sleep apnea in chronic opioid users as it is in patients with congestive heart failure, Dr. Selim said (J Clin Sleep Med. 2016 May 15;12[5]:757-61). “ASV can be very effective in these patients; lower body mass index being a predictor for ASV success,” he said.
Dr. Selim reported having no financial relationships.
BALTIMORE – Given the prevalence of opioid use in the general population, sleep specialists need to be alert to the effects of opioid use on sleep and the link between chronic use and sleep disorders, a pulmonologist recommended at the annual meeting of the Associated Professional Sleep Societies.
Chronic opioid use has multiple effects on sleep that render continuous positive airway pressure (CPAP) all but ineffective, said Bernardo J. Selim, MD, of Mayo Clinic, Rochester, Minn. Characteristic signs of the effects of chronic opioid use on sleep include ataxic central sleep apnea (CSA) and sustained hypoxemia, for which CPAP is generally not effective. Obtaining arterial blood gas measures in these patients is also important to rule out a hypoventilative condition, he added.
In his review of opioid-induced sleep disorders, Dr. Selim cited a small “landmark” study of 24 chronic pain patients on opioids that found 46% had sleep disordered breathing and that the risk rose with the morphine equivalent dose they were taking (J Clin Sleep Med. 2014 Aug 15; 10[8]:847-52).
A meta-analysis also found a dose-dependent relationship with the severity of CSA in patients on opioids, Dr. Selim noted (Anesth Analg. 2015 Jun;120[6]:1273-85). The prevalence of CSA was 24% in the study, which defined two risk factors for CSA severity: a morphine equivalent dose exceeding 200 mg/day and a low or normal body mass index.
Dr. Selim noted that opioids reduce respiration rate more than tidal volume and cause changes to respiratory rhythm. “[Opioids] decrease hypercapnia but increase hypoxic ventilatory response, decrease the arousal index, decrease upper-airway muscle tone, and decrease and also act on chest and abdominal wall compliance.”
Further, different opioids and injection methods can influence breathing. For example, REM and slow-wave sleep decreased across all three categories – intravenous morphine, oral morphine or methadone, and heroin use.
Sleep specialists should be aware that all opioid receptor agonists, whether legal or illegal, have respiratory side effects, Dr. Selim said. “They can present in any way, in any form – CSA, obstructive sleep apnea [OSA], ataxic breathing or sustained hypoxemia. Most of the time [respiratory side effects] present as a combination of complex respiratory patterns.”
In one meta-analysis, CSA was significantly more prevalent in OSA patients on opioids than it was in nonusers, Dr. Selim said, with increased sleep apnea severity as well (J Clin Sleep Med. 2016 Apr 15;12[4]:617-25). Another study found that ataxic breathing was more frequent in non-REM sleep in chronic opioid users (odds ratio, 15.4; P = .017; J Clin Sleep Med. 2007 Aug 15;3[5]:455-61).
The key rule for treating sleep disorders in opioid-dependent patients is to change to nonopioid analgesics, Dr. Selim said. In that regard, ampakines are experimental drugs which have been shown to improve opioid-induced ventilation without loss of the analgesic effect (Clin Pharmacol Ther. 2010 Feb;87[2]:204-11). “Ampakines modulate the action of the glutamate neurotransmitter, decreasing opiate-induced respiratory depression,” Dr. Selim said. An emerging technology, adaptive servo-ventilation (ASV), has been as effective in the treatment of central and complex sleep apnea in chronic opioid users as it is in patients with congestive heart failure, Dr. Selim said (J Clin Sleep Med. 2016 May 15;12[5]:757-61). “ASV can be very effective in these patients; lower body mass index being a predictor for ASV success,” he said.
Dr. Selim reported having no financial relationships.
BALTIMORE – Given the prevalence of opioid use in the general population, sleep specialists need to be alert to the effects of opioid use on sleep and the link between chronic use and sleep disorders, a pulmonologist recommended at the annual meeting of the Associated Professional Sleep Societies.
Chronic opioid use has multiple effects on sleep that render continuous positive airway pressure (CPAP) all but ineffective, said Bernardo J. Selim, MD, of Mayo Clinic, Rochester, Minn. Characteristic signs of the effects of chronic opioid use on sleep include ataxic central sleep apnea (CSA) and sustained hypoxemia, for which CPAP is generally not effective. Obtaining arterial blood gas measures in these patients is also important to rule out a hypoventilative condition, he added.
In his review of opioid-induced sleep disorders, Dr. Selim cited a small “landmark” study of 24 chronic pain patients on opioids that found 46% had sleep disordered breathing and that the risk rose with the morphine equivalent dose they were taking (J Clin Sleep Med. 2014 Aug 15; 10[8]:847-52).
A meta-analysis also found a dose-dependent relationship with the severity of CSA in patients on opioids, Dr. Selim noted (Anesth Analg. 2015 Jun;120[6]:1273-85). The prevalence of CSA was 24% in the study, which defined two risk factors for CSA severity: a morphine equivalent dose exceeding 200 mg/day and a low or normal body mass index.
Dr. Selim noted that opioids reduce respiration rate more than tidal volume and cause changes to respiratory rhythm. “[Opioids] decrease hypercapnia but increase hypoxic ventilatory response, decrease the arousal index, decrease upper-airway muscle tone, and decrease and also act on chest and abdominal wall compliance.”
Further, different opioids and injection methods can influence breathing. For example, REM and slow-wave sleep decreased across all three categories – intravenous morphine, oral morphine or methadone, and heroin use.
Sleep specialists should be aware that all opioid receptor agonists, whether legal or illegal, have respiratory side effects, Dr. Selim said. “They can present in any way, in any form – CSA, obstructive sleep apnea [OSA], ataxic breathing or sustained hypoxemia. Most of the time [respiratory side effects] present as a combination of complex respiratory patterns.”
In one meta-analysis, CSA was significantly more prevalent in OSA patients on opioids than it was in nonusers, Dr. Selim said, with increased sleep apnea severity as well (J Clin Sleep Med. 2016 Apr 15;12[4]:617-25). Another study found that ataxic breathing was more frequent in non-REM sleep in chronic opioid users (odds ratio, 15.4; P = .017; J Clin Sleep Med. 2007 Aug 15;3[5]:455-61).
The key rule for treating sleep disorders in opioid-dependent patients is to change to nonopioid analgesics, Dr. Selim said. In that regard, ampakines are experimental drugs which have been shown to improve opioid-induced ventilation without loss of the analgesic effect (Clin Pharmacol Ther. 2010 Feb;87[2]:204-11). “Ampakines modulate the action of the glutamate neurotransmitter, decreasing opiate-induced respiratory depression,” Dr. Selim said. An emerging technology, adaptive servo-ventilation (ASV), has been as effective in the treatment of central and complex sleep apnea in chronic opioid users as it is in patients with congestive heart failure, Dr. Selim said (J Clin Sleep Med. 2016 May 15;12[5]:757-61). “ASV can be very effective in these patients; lower body mass index being a predictor for ASV success,” he said.
Dr. Selim reported having no financial relationships.
EXPERT ANALYSIS FROM SLEEP 2018
Next-gen sputum PCR panel boosts CAP diagnostics
NEW ORLEANS – A next-generation lower respiratory tract sputum polymerase chain reaction (PCR) film array panel identified etiologic pathogens in 100% of a group of patients hospitalized for community-acquired pneumonia, Kathryn Hendrickson, MD, reported at the annual meeting of the American College of Physicians.
The investigational new diagnostic assay, the BioFire Pneumonia Panel, is now under Food and Drug Administration review for marketing clearance. (CAP), observed Dr. Hendrickson, an internal medicine resident at Providence Portland (Ore.) Medical Center. The new product is designed to complement the currently available respiratory panels from BioFire.
“Rapid-detection results in less empiric antibiotic use in hospitalized patients. When it’s FDA approved, this investigational sputum PCR panel will simplify the diagnostic bundle while improving antibiotic stewardship,” she observed.
She presented a prospective study of 63 patients with CAP hospitalized at the medical center, all of whom were evaluated by two laboratory methods: the hospital’s standard bundle of diagnostic tests and the new BioFire film array panel. The purpose was to determine if there was a difference between the two tests in the detection rate of viral and/or bacterial pathogens as well as the clinical significance of any such differences; that is, was there an impact on days of treatment and length of hospital stay?
Traditional diagnostic methods detect an etiologic pathogen in at best half of hospitalized CAP patients, and the results take too much time. So Providence Portland Medical Center adopted as its standard diagnostic bundle a nasopharyngeal swab and a BioFire film array PCR that’s currently on the market and can detect nine viruses and three bacteria, along with urine antigens for Legionella sp. and Streptococcus pneumoniae, nucleic acid amplification testing for S. pneumoniae and Staphylococcus aureus, and blood and sputum cultures. In contrast, the investigational panel probes for 17 viruses, 18 bacterial pathogens, and seven antibiotic-resistant genes; it also measures procalcitonin levels in order to distinguish between bacterial colonization and invasion.
The new BioFire Pneumonia Panel detected a mean of 1.4 species of pathogenic bacteria in 79% of patients, while the standard diagnostic bundle detected 0.7 species in 59% of patients. The investigational panel identified a mean of 1.0 species of viral pathogens in 86% of the CAP patients; the standard bundle detected a mean of 0.6 species in 56%.
All told, any CAP pathogen was detected in 100% of patients using the new panel, with a mean of 2.5 different pathogens identified. The standard bundle detected any pathogen in 84% of patients, with half as many different pathogens found, according to Dr. Hendrickson.
A peak procalcitonin level of 0.25 ng/mL or less, which was defined as bacterial colonization, was associated with a mean 7 days of treatment, while a level above that threshold was associated with 11.3 days of treatment. Patients with a peak procalcitonin of 0.25 ng/mL or less had an average hospital length of stay of 5.9 days, versus 7.8 days for those with a higher procalcitonin indicative of bacterial invasion.
The new biofilm assay reports information about the abundance of 15 of the 18 bacterial targets in the sample. However, in contrast to peak procalcitonin, Dr. Hendrickson and her coinvestigators didn’t find this bacterial quantitation feature to be substantially more useful than a coin flip in distinguishing bacterial colonization from invasion.
She reported having no financial conflicts regarding the head-to-head comparative study, which was supported by BioFire Diagnostics.
NEW ORLEANS – A next-generation lower respiratory tract sputum polymerase chain reaction (PCR) film array panel identified etiologic pathogens in 100% of a group of patients hospitalized for community-acquired pneumonia, Kathryn Hendrickson, MD, reported at the annual meeting of the American College of Physicians.
The investigational new diagnostic assay, the BioFire Pneumonia Panel, is now under Food and Drug Administration review for marketing clearance. (CAP), observed Dr. Hendrickson, an internal medicine resident at Providence Portland (Ore.) Medical Center. The new product is designed to complement the currently available respiratory panels from BioFire.
“Rapid-detection results in less empiric antibiotic use in hospitalized patients. When it’s FDA approved, this investigational sputum PCR panel will simplify the diagnostic bundle while improving antibiotic stewardship,” she observed.
She presented a prospective study of 63 patients with CAP hospitalized at the medical center, all of whom were evaluated by two laboratory methods: the hospital’s standard bundle of diagnostic tests and the new BioFire film array panel. The purpose was to determine if there was a difference between the two tests in the detection rate of viral and/or bacterial pathogens as well as the clinical significance of any such differences; that is, was there an impact on days of treatment and length of hospital stay?
Traditional diagnostic methods detect an etiologic pathogen in at best half of hospitalized CAP patients, and the results take too much time. So Providence Portland Medical Center adopted as its standard diagnostic bundle a nasopharyngeal swab and a BioFire film array PCR that’s currently on the market and can detect nine viruses and three bacteria, along with urine antigens for Legionella sp. and Streptococcus pneumoniae, nucleic acid amplification testing for S. pneumoniae and Staphylococcus aureus, and blood and sputum cultures. In contrast, the investigational panel probes for 17 viruses, 18 bacterial pathogens, and seven antibiotic-resistant genes; it also measures procalcitonin levels in order to distinguish between bacterial colonization and invasion.
The new BioFire Pneumonia Panel detected a mean of 1.4 species of pathogenic bacteria in 79% of patients, while the standard diagnostic bundle detected 0.7 species in 59% of patients. The investigational panel identified a mean of 1.0 species of viral pathogens in 86% of the CAP patients; the standard bundle detected a mean of 0.6 species in 56%.
All told, any CAP pathogen was detected in 100% of patients using the new panel, with a mean of 2.5 different pathogens identified. The standard bundle detected any pathogen in 84% of patients, with half as many different pathogens found, according to Dr. Hendrickson.
A peak procalcitonin level of 0.25 ng/mL or less, which was defined as bacterial colonization, was associated with a mean 7 days of treatment, while a level above that threshold was associated with 11.3 days of treatment. Patients with a peak procalcitonin of 0.25 ng/mL or less had an average hospital length of stay of 5.9 days, versus 7.8 days for those with a higher procalcitonin indicative of bacterial invasion.
The new biofilm assay reports information about the abundance of 15 of the 18 bacterial targets in the sample. However, in contrast to peak procalcitonin, Dr. Hendrickson and her coinvestigators didn’t find this bacterial quantitation feature to be substantially more useful than a coin flip in distinguishing bacterial colonization from invasion.
She reported having no financial conflicts regarding the head-to-head comparative study, which was supported by BioFire Diagnostics.
NEW ORLEANS – A next-generation lower respiratory tract sputum polymerase chain reaction (PCR) film array panel identified etiologic pathogens in 100% of a group of patients hospitalized for community-acquired pneumonia, Kathryn Hendrickson, MD, reported at the annual meeting of the American College of Physicians.
The investigational new diagnostic assay, the BioFire Pneumonia Panel, is now under Food and Drug Administration review for marketing clearance. (CAP), observed Dr. Hendrickson, an internal medicine resident at Providence Portland (Ore.) Medical Center. The new product is designed to complement the currently available respiratory panels from BioFire.
“Rapid-detection results in less empiric antibiotic use in hospitalized patients. When it’s FDA approved, this investigational sputum PCR panel will simplify the diagnostic bundle while improving antibiotic stewardship,” she observed.
She presented a prospective study of 63 patients with CAP hospitalized at the medical center, all of whom were evaluated by two laboratory methods: the hospital’s standard bundle of diagnostic tests and the new BioFire film array panel. The purpose was to determine if there was a difference between the two tests in the detection rate of viral and/or bacterial pathogens as well as the clinical significance of any such differences; that is, was there an impact on days of treatment and length of hospital stay?
Traditional diagnostic methods detect an etiologic pathogen in at best half of hospitalized CAP patients, and the results take too much time. So Providence Portland Medical Center adopted as its standard diagnostic bundle a nasopharyngeal swab and a BioFire film array PCR that’s currently on the market and can detect nine viruses and three bacteria, along with urine antigens for Legionella sp. and Streptococcus pneumoniae, nucleic acid amplification testing for S. pneumoniae and Staphylococcus aureus, and blood and sputum cultures. In contrast, the investigational panel probes for 17 viruses, 18 bacterial pathogens, and seven antibiotic-resistant genes; it also measures procalcitonin levels in order to distinguish between bacterial colonization and invasion.
The new BioFire Pneumonia Panel detected a mean of 1.4 species of pathogenic bacteria in 79% of patients, while the standard diagnostic bundle detected 0.7 species in 59% of patients. The investigational panel identified a mean of 1.0 species of viral pathogens in 86% of the CAP patients; the standard bundle detected a mean of 0.6 species in 56%.
All told, any CAP pathogen was detected in 100% of patients using the new panel, with a mean of 2.5 different pathogens identified. The standard bundle detected any pathogen in 84% of patients, with half as many different pathogens found, according to Dr. Hendrickson.
A peak procalcitonin level of 0.25 ng/mL or less, which was defined as bacterial colonization, was associated with a mean 7 days of treatment, while a level above that threshold was associated with 11.3 days of treatment. Patients with a peak procalcitonin of 0.25 ng/mL or less had an average hospital length of stay of 5.9 days, versus 7.8 days for those with a higher procalcitonin indicative of bacterial invasion.
The new biofilm assay reports information about the abundance of 15 of the 18 bacterial targets in the sample. However, in contrast to peak procalcitonin, Dr. Hendrickson and her coinvestigators didn’t find this bacterial quantitation feature to be substantially more useful than a coin flip in distinguishing bacterial colonization from invasion.
She reported having no financial conflicts regarding the head-to-head comparative study, which was supported by BioFire Diagnostics.
REPORTING FROM ACP INTERNAL MEDICINE
Key clinical point: A new CAP diagnostic panel represents a significant advance in clinical care.
Major finding: The investigational BioFire Pneumonia Panel identified specific pathogens in 100% of patients hospitalized for CAP, compared with 84% using the hospital’s standard test bundle.
Study details: This was a prospective head-to-head study comparing two approaches to identification of specific pathogens in 63 patients hospitalized for CAP.
Disclosures: The study was supported by BioFire Diagnostics. The presenter reported having no financial conflicts.
COPD opposites: Utah and West Virginia
New estimates of chronic obstructive pulmonary disease (COPD) may have Utah residents breathing a sigh of relief. West Virginians, not so much.
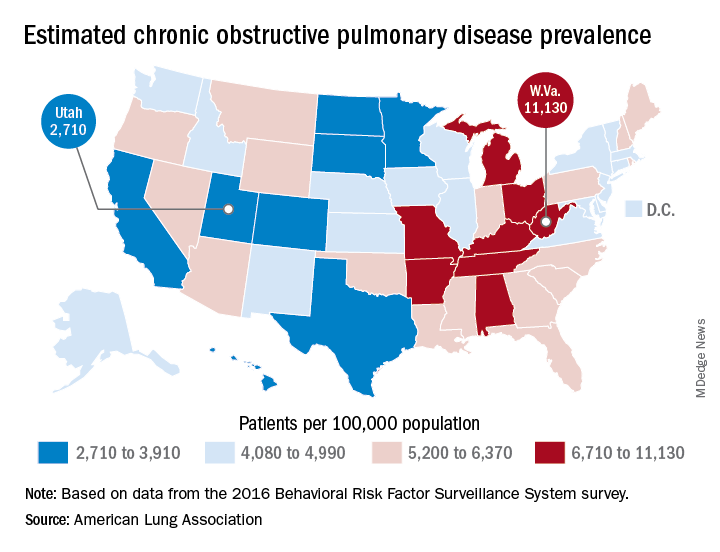
The Beehive State has the lowest prevalence of COPD in the country at 2,710 per 100,000 population, while the Mountain State tops the charts at 11,130 per 100,000, according to estimates from the American Lung Association. (Crude rates were calculated by MDedge News using the ALA’s estimates for total persons with COPD in each state and Census Bureau estimates for population.)
Other states with freer-breathing residents include Minnesota, which was just behind Utah with an estimated rate of 3,000 per 100,000 population, Hawaii (3,182), Colorado (3,334), and California (3,409). West Virginia’s rate, however, seems to be an outlier. The state with the next-highest rate, Kentucky, has a calculated prevalence of 8,890 per 100,000 population, followed by Tennessee at 7,880, Alabama at 7,400, and Arkansas at 7,330, using the ALA’s estimates, which were based on data from the 2016 Behavioral Risk Factor Surveillance System survey.
New estimates of chronic obstructive pulmonary disease (COPD) may have Utah residents breathing a sigh of relief. West Virginians, not so much.

The Beehive State has the lowest prevalence of COPD in the country at 2,710 per 100,000 population, while the Mountain State tops the charts at 11,130 per 100,000, according to estimates from the American Lung Association. (Crude rates were calculated by MDedge News using the ALA’s estimates for total persons with COPD in each state and Census Bureau estimates for population.)
Other states with freer-breathing residents include Minnesota, which was just behind Utah with an estimated rate of 3,000 per 100,000 population, Hawaii (3,182), Colorado (3,334), and California (3,409). West Virginia’s rate, however, seems to be an outlier. The state with the next-highest rate, Kentucky, has a calculated prevalence of 8,890 per 100,000 population, followed by Tennessee at 7,880, Alabama at 7,400, and Arkansas at 7,330, using the ALA’s estimates, which were based on data from the 2016 Behavioral Risk Factor Surveillance System survey.
New estimates of chronic obstructive pulmonary disease (COPD) may have Utah residents breathing a sigh of relief. West Virginians, not so much.

The Beehive State has the lowest prevalence of COPD in the country at 2,710 per 100,000 population, while the Mountain State tops the charts at 11,130 per 100,000, according to estimates from the American Lung Association. (Crude rates were calculated by MDedge News using the ALA’s estimates for total persons with COPD in each state and Census Bureau estimates for population.)
Other states with freer-breathing residents include Minnesota, which was just behind Utah with an estimated rate of 3,000 per 100,000 population, Hawaii (3,182), Colorado (3,334), and California (3,409). West Virginia’s rate, however, seems to be an outlier. The state with the next-highest rate, Kentucky, has a calculated prevalence of 8,890 per 100,000 population, followed by Tennessee at 7,880, Alabama at 7,400, and Arkansas at 7,330, using the ALA’s estimates, which were based on data from the 2016 Behavioral Risk Factor Surveillance System survey.
FDA gives Orkambi indication for younger patients
(CF), according to its manufacturer, Vertex Pharmaceuticals. Specifically, the drug is meant to treat the most common underlying cause of CF – having two copies of the F508del-CFTR mutation – and is the first drug to treat it.
The approval is based on a phase 3, two-part, open-label, multicenter study that assessed various doses in patents aged 2-5 years. The study demonstrated safety and tolerability in that age group equivalent to that seen in older patients. The drug is expected to be available for this age group within 2-4 weeks of this approval.
Available as oral granules in two doses for weight-based dosing (either lumacaftor 100 mg/ivacaftor 125 mg or lumacaftor 150 mg/ivacaftor 188 mg), the compound targets the defective chloride channels responsible for CF; the two halves work together to increase the number of chloride channels on cell surfaces and also improve their function.
Orkambi should be prescribed only for patients with CF who have the dual F508del-CFTR mutation; it is not indicated for other types of CF. Patients should not take this drug if they are taking drugs such as rifampin, phenytoin, triazolam, or cyclosporine because of possible drug interactions. It can also lead to worsening liver function and elevated blood liver enzymes, increased blood pressure, or cataracts. The most common side effects include breathing problems, nausea, fatigue, and rash. Full prescribing information is available on the FDA website.
(CF), according to its manufacturer, Vertex Pharmaceuticals. Specifically, the drug is meant to treat the most common underlying cause of CF – having two copies of the F508del-CFTR mutation – and is the first drug to treat it.
The approval is based on a phase 3, two-part, open-label, multicenter study that assessed various doses in patents aged 2-5 years. The study demonstrated safety and tolerability in that age group equivalent to that seen in older patients. The drug is expected to be available for this age group within 2-4 weeks of this approval.
Available as oral granules in two doses for weight-based dosing (either lumacaftor 100 mg/ivacaftor 125 mg or lumacaftor 150 mg/ivacaftor 188 mg), the compound targets the defective chloride channels responsible for CF; the two halves work together to increase the number of chloride channels on cell surfaces and also improve their function.
Orkambi should be prescribed only for patients with CF who have the dual F508del-CFTR mutation; it is not indicated for other types of CF. Patients should not take this drug if they are taking drugs such as rifampin, phenytoin, triazolam, or cyclosporine because of possible drug interactions. It can also lead to worsening liver function and elevated blood liver enzymes, increased blood pressure, or cataracts. The most common side effects include breathing problems, nausea, fatigue, and rash. Full prescribing information is available on the FDA website.
(CF), according to its manufacturer, Vertex Pharmaceuticals. Specifically, the drug is meant to treat the most common underlying cause of CF – having two copies of the F508del-CFTR mutation – and is the first drug to treat it.
The approval is based on a phase 3, two-part, open-label, multicenter study that assessed various doses in patents aged 2-5 years. The study demonstrated safety and tolerability in that age group equivalent to that seen in older patients. The drug is expected to be available for this age group within 2-4 weeks of this approval.
Available as oral granules in two doses for weight-based dosing (either lumacaftor 100 mg/ivacaftor 125 mg or lumacaftor 150 mg/ivacaftor 188 mg), the compound targets the defective chloride channels responsible for CF; the two halves work together to increase the number of chloride channels on cell surfaces and also improve their function.
Orkambi should be prescribed only for patients with CF who have the dual F508del-CFTR mutation; it is not indicated for other types of CF. Patients should not take this drug if they are taking drugs such as rifampin, phenytoin, triazolam, or cyclosporine because of possible drug interactions. It can also lead to worsening liver function and elevated blood liver enzymes, increased blood pressure, or cataracts. The most common side effects include breathing problems, nausea, fatigue, and rash. Full prescribing information is available on the FDA website.
Telomere length linked to COPD exacerbations, mortality
according to a study published in Chest.
The evidence suggests that chronic obstructive pulmonary disease (COPD) may be a disease of accelerated aging, partly because of its relation to other senescence-related disorders such as osteoporosis and dementia, but also because it shows an exponential increase in prevalence in older age.
Telomere lengths are a measure of cellular senescence, and previous research has found that the telomeres are shortened in the peripheral leukocytes of patients with COPD, compared with healthy controls.
In this study, researchers examined the absolute telomere length of 576 people with moderate to severe COPD who were participating in the MACRO (Macrolide Azithromycin for Prevention of Exacerbations of COPD) study.
They found that individuals in the lowest quartile of telomere lengths had significantly worse health status and a higher exacerbation rate after accounting for treatment, compared with individuals in the higher quartile.
Patients with shorter telomere length had worse health status, as defined by higher St. George’s Respiratory Questionnaire scores. In the placebo arm of the study, the exacerbation rate (rate ratio, 1.50; 95% confidence interval, 1.16-1.95; P = .002) and mortality risk (hazard ratio, 9.45; 95% CI, 2.85-31.36; P = .015) were significantly higher in the shorter telomere group than in the longer telomere group; these differences were not observed in the azithromycin arm.
Patients with shorter telomeres also had a 800% higher risk of total mortality, compared with individuals with longer telomeres, although this was only evident in the placebo arm of the study, not the azithromycin arm. However, the authors noted that these data should be interpreted with caution because of the small number of deaths during the study.
“Together, these data support the notion that COPD is a systemic disease of accelerated aging and that replicative senescence, denoted by peripheral blood telomeres, is associated with poor health outcomes in COPD,” wrote Minhee Jin, of the University of British Columbia, Vancouver, and coauthors.
“It is now well established that replicative senescence results in a change of cellular phenotype to a proinflammatory state, a process that has been referred to as senescence-associated secretory phenotype,” they added.
The study also found that the median value for telomere length across the study participants – who had a mean age of 66 years – was equivalent to the expected value for someone in their 80s, “suggesting that on average MACRO participants were biologically much older than their chronological age.”
Researchers also noted that patients in the lowest quartile of telomere length had significantly lower forced vital capacity values, which suggested shorter telomeres could be a biomarker of restrictive physiology.
MACRO was funded by the U.S. National Heart, Lung, and Blood Institute, and the biomarker component of the study was funded by the Canadian Respiratory Research Network, Genome Canada, and the St. Paul’s Hospital Foundation. One author was an employee of GenomeDx Biosciences, three declared funding from or consultancies with the pharmaceutical industry. No other conflicts of interest were reported.
SOURCE: Jin M et al. Chest. 2018 Jul 12. doi: 10.1016/j.chest.2018.05.022.
according to a study published in Chest.
The evidence suggests that chronic obstructive pulmonary disease (COPD) may be a disease of accelerated aging, partly because of its relation to other senescence-related disorders such as osteoporosis and dementia, but also because it shows an exponential increase in prevalence in older age.
Telomere lengths are a measure of cellular senescence, and previous research has found that the telomeres are shortened in the peripheral leukocytes of patients with COPD, compared with healthy controls.
In this study, researchers examined the absolute telomere length of 576 people with moderate to severe COPD who were participating in the MACRO (Macrolide Azithromycin for Prevention of Exacerbations of COPD) study.
They found that individuals in the lowest quartile of telomere lengths had significantly worse health status and a higher exacerbation rate after accounting for treatment, compared with individuals in the higher quartile.
Patients with shorter telomere length had worse health status, as defined by higher St. George’s Respiratory Questionnaire scores. In the placebo arm of the study, the exacerbation rate (rate ratio, 1.50; 95% confidence interval, 1.16-1.95; P = .002) and mortality risk (hazard ratio, 9.45; 95% CI, 2.85-31.36; P = .015) were significantly higher in the shorter telomere group than in the longer telomere group; these differences were not observed in the azithromycin arm.
Patients with shorter telomeres also had a 800% higher risk of total mortality, compared with individuals with longer telomeres, although this was only evident in the placebo arm of the study, not the azithromycin arm. However, the authors noted that these data should be interpreted with caution because of the small number of deaths during the study.
“Together, these data support the notion that COPD is a systemic disease of accelerated aging and that replicative senescence, denoted by peripheral blood telomeres, is associated with poor health outcomes in COPD,” wrote Minhee Jin, of the University of British Columbia, Vancouver, and coauthors.
“It is now well established that replicative senescence results in a change of cellular phenotype to a proinflammatory state, a process that has been referred to as senescence-associated secretory phenotype,” they added.
The study also found that the median value for telomere length across the study participants – who had a mean age of 66 years – was equivalent to the expected value for someone in their 80s, “suggesting that on average MACRO participants were biologically much older than their chronological age.”
Researchers also noted that patients in the lowest quartile of telomere length had significantly lower forced vital capacity values, which suggested shorter telomeres could be a biomarker of restrictive physiology.
MACRO was funded by the U.S. National Heart, Lung, and Blood Institute, and the biomarker component of the study was funded by the Canadian Respiratory Research Network, Genome Canada, and the St. Paul’s Hospital Foundation. One author was an employee of GenomeDx Biosciences, three declared funding from or consultancies with the pharmaceutical industry. No other conflicts of interest were reported.
SOURCE: Jin M et al. Chest. 2018 Jul 12. doi: 10.1016/j.chest.2018.05.022.
according to a study published in Chest.
The evidence suggests that chronic obstructive pulmonary disease (COPD) may be a disease of accelerated aging, partly because of its relation to other senescence-related disorders such as osteoporosis and dementia, but also because it shows an exponential increase in prevalence in older age.
Telomere lengths are a measure of cellular senescence, and previous research has found that the telomeres are shortened in the peripheral leukocytes of patients with COPD, compared with healthy controls.
In this study, researchers examined the absolute telomere length of 576 people with moderate to severe COPD who were participating in the MACRO (Macrolide Azithromycin for Prevention of Exacerbations of COPD) study.
They found that individuals in the lowest quartile of telomere lengths had significantly worse health status and a higher exacerbation rate after accounting for treatment, compared with individuals in the higher quartile.
Patients with shorter telomere length had worse health status, as defined by higher St. George’s Respiratory Questionnaire scores. In the placebo arm of the study, the exacerbation rate (rate ratio, 1.50; 95% confidence interval, 1.16-1.95; P = .002) and mortality risk (hazard ratio, 9.45; 95% CI, 2.85-31.36; P = .015) were significantly higher in the shorter telomere group than in the longer telomere group; these differences were not observed in the azithromycin arm.
Patients with shorter telomeres also had a 800% higher risk of total mortality, compared with individuals with longer telomeres, although this was only evident in the placebo arm of the study, not the azithromycin arm. However, the authors noted that these data should be interpreted with caution because of the small number of deaths during the study.
“Together, these data support the notion that COPD is a systemic disease of accelerated aging and that replicative senescence, denoted by peripheral blood telomeres, is associated with poor health outcomes in COPD,” wrote Minhee Jin, of the University of British Columbia, Vancouver, and coauthors.
“It is now well established that replicative senescence results in a change of cellular phenotype to a proinflammatory state, a process that has been referred to as senescence-associated secretory phenotype,” they added.
The study also found that the median value for telomere length across the study participants – who had a mean age of 66 years – was equivalent to the expected value for someone in their 80s, “suggesting that on average MACRO participants were biologically much older than their chronological age.”
Researchers also noted that patients in the lowest quartile of telomere length had significantly lower forced vital capacity values, which suggested shorter telomeres could be a biomarker of restrictive physiology.
MACRO was funded by the U.S. National Heart, Lung, and Blood Institute, and the biomarker component of the study was funded by the Canadian Respiratory Research Network, Genome Canada, and the St. Paul’s Hospital Foundation. One author was an employee of GenomeDx Biosciences, three declared funding from or consultancies with the pharmaceutical industry. No other conflicts of interest were reported.
SOURCE: Jin M et al. Chest. 2018 Jul 12. doi: 10.1016/j.chest.2018.05.022.
FROM CHEST
Key clinical point: Shorter telomeres are linked to an increased risk of chronic obstructive pulmonary disease exacerbations.
Major finding: Patients with shorter telomeres had a 800% higher risk of total mortality, compared with individuals with longer telomeres.
Study details: Data from 576 patients with chronic obstructive pulmonary disease who participated in the MACRO study.
Disclosures: MACRO was funded by the U.S. National Heart, Lung, and Blood Institute, and the biomarker component of the study was funded by the Canadian Respiratory Research Network and the Canadian Institutes of Health Research Genome Canada, and the St. Paul’s Hospital Foundation. One author was an employee of GenomeDx Biosciences, and three authors declared funding from or consultancies with the pharmaceutical industry. No other conflicts of interest were reported.
Source: Jin M et al. Chest. 2018 Jul 12. doi: 10.1016/j.chest.2018.05.022.