User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
Powered by CHEST Physician, Clinician Reviews, MDedge Family Medicine, Internal Medicine News, and The Journal of Clinical Outcomes Management.
E-cigarettes: Prices down, sales up
Any economist could have predicted it: As the .
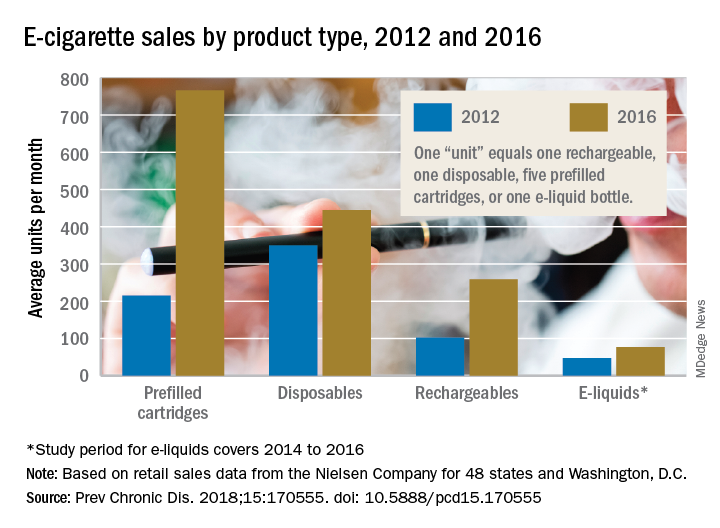
The average prices of three mutually exclusive e-cigarette products – rechargeable devices, disposable devices, and disposable cartridges filled with e-liquid – all dropped from 2012 to 2016, as did that of a fourth product – e-liquid bottles for filling reusable cartridges – not available nationwide until 2014. At the same time, average monthly sales for e-cigarette products overall rose by a statistically significant 132%, Teresa W. Wang, PhD, of the Centers for Disease Control and Prevention, Atlanta, and her associates reported in Preventing Chronic Disease.
Sales of prefilled cartridges, the most popular product by the end of the study period, increased 256%, going from 215 units per 100,000 people each month in 2012 to 766 units. [For the study, a unit was defined as one rechargeable, one disposable, one pack of five prefilled cartridges, or one bottle of e-liquid.] Disposables were the most popular product at the start of the study period but had the smallest relative increase (27%), while monthly sales of rechargeables jumped by 154% and e-liquids saw a 64% rise, the investigators said.
Price decreases for the three products available in 2012 were all significant: The average price per unit was down 48% for rechargeables by 2016, 14% for disposables, and 12% for prefilled cartridges. E-liquids were 9% cheaper by 2016, but that change did not reach significance, they noted.
“Overall, the increase in e-cigarette sales and decrease in price is consistent with previous studies demonstrating that e-cigarette sales are responsive to their own price changes. These trends suggest that, if e-cigarette prices continue to decrease, their sales may also continue to rise,” Dr. Wang and her associates wrote.
The data for the study came from the Nielsen Company and were based on retail sales at convenience stores; supermarkets; drug, dollar, and club stores; and military commissaries in the 48 contiguous states and Washington, D.C. One study limitation was the lack of data from tobacco/vape shops and the Internet.
SOURCE: Wang TW et al. Prev Chronic Dis. 2018;15:170555. doi: 10.5888/pcd15.170555.
Any economist could have predicted it: As the .

The average prices of three mutually exclusive e-cigarette products – rechargeable devices, disposable devices, and disposable cartridges filled with e-liquid – all dropped from 2012 to 2016, as did that of a fourth product – e-liquid bottles for filling reusable cartridges – not available nationwide until 2014. At the same time, average monthly sales for e-cigarette products overall rose by a statistically significant 132%, Teresa W. Wang, PhD, of the Centers for Disease Control and Prevention, Atlanta, and her associates reported in Preventing Chronic Disease.
Sales of prefilled cartridges, the most popular product by the end of the study period, increased 256%, going from 215 units per 100,000 people each month in 2012 to 766 units. [For the study, a unit was defined as one rechargeable, one disposable, one pack of five prefilled cartridges, or one bottle of e-liquid.] Disposables were the most popular product at the start of the study period but had the smallest relative increase (27%), while monthly sales of rechargeables jumped by 154% and e-liquids saw a 64% rise, the investigators said.
Price decreases for the three products available in 2012 were all significant: The average price per unit was down 48% for rechargeables by 2016, 14% for disposables, and 12% for prefilled cartridges. E-liquids were 9% cheaper by 2016, but that change did not reach significance, they noted.
“Overall, the increase in e-cigarette sales and decrease in price is consistent with previous studies demonstrating that e-cigarette sales are responsive to their own price changes. These trends suggest that, if e-cigarette prices continue to decrease, their sales may also continue to rise,” Dr. Wang and her associates wrote.
The data for the study came from the Nielsen Company and were based on retail sales at convenience stores; supermarkets; drug, dollar, and club stores; and military commissaries in the 48 contiguous states and Washington, D.C. One study limitation was the lack of data from tobacco/vape shops and the Internet.
SOURCE: Wang TW et al. Prev Chronic Dis. 2018;15:170555. doi: 10.5888/pcd15.170555.
Any economist could have predicted it: As the .

The average prices of three mutually exclusive e-cigarette products – rechargeable devices, disposable devices, and disposable cartridges filled with e-liquid – all dropped from 2012 to 2016, as did that of a fourth product – e-liquid bottles for filling reusable cartridges – not available nationwide until 2014. At the same time, average monthly sales for e-cigarette products overall rose by a statistically significant 132%, Teresa W. Wang, PhD, of the Centers for Disease Control and Prevention, Atlanta, and her associates reported in Preventing Chronic Disease.
Sales of prefilled cartridges, the most popular product by the end of the study period, increased 256%, going from 215 units per 100,000 people each month in 2012 to 766 units. [For the study, a unit was defined as one rechargeable, one disposable, one pack of five prefilled cartridges, or one bottle of e-liquid.] Disposables were the most popular product at the start of the study period but had the smallest relative increase (27%), while monthly sales of rechargeables jumped by 154% and e-liquids saw a 64% rise, the investigators said.
Price decreases for the three products available in 2012 were all significant: The average price per unit was down 48% for rechargeables by 2016, 14% for disposables, and 12% for prefilled cartridges. E-liquids were 9% cheaper by 2016, but that change did not reach significance, they noted.
“Overall, the increase in e-cigarette sales and decrease in price is consistent with previous studies demonstrating that e-cigarette sales are responsive to their own price changes. These trends suggest that, if e-cigarette prices continue to decrease, their sales may also continue to rise,” Dr. Wang and her associates wrote.
The data for the study came from the Nielsen Company and were based on retail sales at convenience stores; supermarkets; drug, dollar, and club stores; and military commissaries in the 48 contiguous states and Washington, D.C. One study limitation was the lack of data from tobacco/vape shops and the Internet.
SOURCE: Wang TW et al. Prev Chronic Dis. 2018;15:170555. doi: 10.5888/pcd15.170555.
FROM PREVENTING CHRONIC DISEASE
Four syndromes suggest life-threatening PVL-positive S. aureus infection
MALMO, SWEDEN – Methicillin-resistant Staphylococcus aureus gets the blame in the Americas as the main cause of a great wave of community-acquired severe invasive staphylococcal infections in children and adolescents during the past nearly 2 decades, but many European pediatric infectious disease specialists believe that Panton-Valentine leukocidin (PVL), a frequent co-traveler with MRSA, is the true bad actor.
“The American literature focused first on MRSA, but we’ve seen very similar, very severe cases with MSSA [methicillin-susceptible S. aureus] PVL-positive and MRSA PVL-positive infections,” Pablo Rojo, MD, PhD, said at the annual meeting of the European Society for Paediatric Infectious Diseases.
“It is only because at the beginning there were so many MRSA cases in the States that they thought that was the driver of the disease. It is still unclear. There is still a discussion. But I wanted to bring you my opinion and that of many other authors that it’s mostly PVL-associated,” added Dr. Rojo of Complutense University in Madrid.
He was senior author of a multinational European and Israeli prospective study of risk factors associated with the severity of invasive community-acquired S. aureus infections in children, with invasive infection being defined as hospitalization for an infection with S. aureus isolated from a normally sterile body site such as blood, bone, or cerebrospinal fluid, or S. aureus pneumonia. They identified 152 affected children, 17% of whom had severe community-acquired invasive S. aureus, defined by death or admission to a pediatric intensive care unit due to respiratory failure or hemodynamic instability.
The prevalence of PVL-positive S. aureus infection in the overall invasive infection group was 19%, while 8% of the isolates were MRSA. while MRSA was not associated with a significantly increased risk. The other independent risk factors for severe outcome were pneumonia, with an adjusted 13-fold increased risk, and leukopenia at admission, with an associated 18-fold risk (Clin Microbiol Infect. 2016 Jul;22[7]:643.e1-6.).
Of note, the virulence of PVL stems from the pore-forming toxin’s ability to lyse white blood cells. Because a leukocyte count is always available once a patient reaches the ED, severe leukopenia as defined by a count of less than 3,000 cells/mm3 at admission becomes a useful early marker of the likely severity of any case of S. aureus invasive disease, according to Dr. Rojo.
He highlighted four key syndromes involving severe invasive S. aureus infection in previously healthy children and adolescents that entail a high likelihood of being PVL positive and should cause physicians to run – not walk – to start appropriate empiric therapy. He also described the treatment regimen that he and other European thought leaders recommend for severe PVL-positive S. aureus invasive infections.
The microbiologic diagnosis of PVL can be made by ELISA (enzyme-linked immunoassay) to detect the toxin in an S. aureus isolate, by a rapid monoclonal antibody test, or by polymerase chain reaction to detect PVL genes in an S. aureus isolate. But don’t wait for test results to initiate treatment because these are high-mortality syndromes, he advised.
“Many people tell me, ‘My lab doesn’t have a way to diagnose PVL.’ And it’s true, it’s not available in real life at many hospitals. My message to you is that you don’t need to wait for a microbiological diagnosis or the results to come back from a sample you have sent to the reference lab in the main referral center. We can base our diagnosis and decision to treat on clinical grounds if we focus on these four very uncommon syndromes involving invasive S. aureus infection. I think if you have any child with these symptoms you have to manage them on the assumption that PVL is present,” said Dr. Rojo, principal investigator of the European Project on Invasive S. aureus Pediatric Infections.
The four key syndromes
The four syndromes are severe S. aureus pneumonia, S. aureus bone and joint infections with multiple foci, S. aureus osteomyelitis complicated by deep vein thrombosis, and invasive S. aureus infection plus shock.
- Severe S. aureus pneumonia. Investigators at Claude Bernard University in Lyon, France, have done extensive pioneering work on severe PVL-positive S. aureus invasive infections in children. In an early paper, they highlighted the characteristics that distinguish severe PVL-positive pneumonia: it typically occurs in previously healthy children and adolescents without underlying comorbid conditions, and it is often preceded by a influenza-like syndrome followed by an acute severe pneumonia with hemoptysis. Mortality was very high in this early series, with nearly half of the patients being dead within the first several days after admission (Lancet. 2002 Mar 2;359[9308]:753-9).
- Severe osteomyelitis. Investigators at Baylor College of Medicine, Houston, were among the first to observe that osteomyelitis caused by PVL-positive strains of S. aureus are associated with more severe local disease, with multiple affected areas, bigger abscesses, a greater systemic inflammatory response, and more surgeries required compared with osteomyelitis caused by PVL-negative S. aureus (Pediatrics. 2006 Feb;117[2]:433-40).
- Osteomyelitis with deep vein thrombosis. When a child hospitalized for acute hematogenous osteomyelitis due to S. aureus develops difficulty breathing, that’s a red flag for a severe PVL-positive infection involving deep vein thrombosis. Indeed, investigators at the Leeds (England) General Infirmary have reported that deep vein thrombosis in the setting of S. aureus osteomyelitis is associated with a greater than eightfold increased likelihood of a PVL-positive infection (Br J Hosp Med [Lond]. 2015 Jan;76[1]:18-24). Also, patients with PVL-positive osteomyelitis and deep vein thrombosis are prone to formation of septic emboli.
- Osteomyelitis with septic shock. The Lyon group compared outcomes in 14 pediatric patients with PVL-positive S. aureus osteomyelitis and a control group of 17 patients with PVL-negative disease. All 14 PVL-positive patients had severe sepsis and 6 of them had septic shock. In contrast, none of the controls did. Median duration of hospitalization was 46 days in the PVL-positive group, compared with 13 days in controls (Pediatr Infect Dis J. 2007 Nov;26[11]:1042-8).
Treatment
No randomized trials exist to guide treatment, but Dr. Rojo recommends the protocol utilized by the Lyon group: a bactericidal antibiotic – vancomycin or a beta-lactam – to take on the S. aureus, coupled with a ribosomally active antibiotic – clindamycin or linezolid – to suppress the PVL toxin’s virulence expression. The French group cites both in vitro and in vivo evidence that clindamycin and linezolid in their standard dosing have such an antitoxin effect (Clin Microbiol Rev. 2017 Oct;30[4]:887-917).
In addition, Dr. Rojo recommends utilizing any of the commercially available intravenous immunoglobulin (IVIG) products on the basis of work by investigators at Vanderbilt University in Nashville, Tenn., who have demonstrated that these products contain functional neutralizing antibodies against S. aureus leukocidins. This observation provides a likely explanation for anecdotal reports of improved outcomes in IVIG-treated patients with toxin-associated staphylococcal disease (Antimicrob Agents Chemother. 2017 Oct 24;61[11]. pii: e00968-17).
Challenged as to when specifically he would use IVIG in light of the global shortage of immunoglobulins, Dr. Rojo replied: “Not in every invasive S. aureus infection, but in serious infections that are PVL positive. I think if you have a child with one of these four syndromes who is in a pediatric ICU, you should use it. I mean, the mortality is around 30% in healthy children, so you would not stop from giving it. The risk of giving IVIG is very low, no side effects, so I highly recommend it for these severe cases.”
He reported having no financial conflicts.
MALMO, SWEDEN – Methicillin-resistant Staphylococcus aureus gets the blame in the Americas as the main cause of a great wave of community-acquired severe invasive staphylococcal infections in children and adolescents during the past nearly 2 decades, but many European pediatric infectious disease specialists believe that Panton-Valentine leukocidin (PVL), a frequent co-traveler with MRSA, is the true bad actor.
“The American literature focused first on MRSA, but we’ve seen very similar, very severe cases with MSSA [methicillin-susceptible S. aureus] PVL-positive and MRSA PVL-positive infections,” Pablo Rojo, MD, PhD, said at the annual meeting of the European Society for Paediatric Infectious Diseases.
“It is only because at the beginning there were so many MRSA cases in the States that they thought that was the driver of the disease. It is still unclear. There is still a discussion. But I wanted to bring you my opinion and that of many other authors that it’s mostly PVL-associated,” added Dr. Rojo of Complutense University in Madrid.
He was senior author of a multinational European and Israeli prospective study of risk factors associated with the severity of invasive community-acquired S. aureus infections in children, with invasive infection being defined as hospitalization for an infection with S. aureus isolated from a normally sterile body site such as blood, bone, or cerebrospinal fluid, or S. aureus pneumonia. They identified 152 affected children, 17% of whom had severe community-acquired invasive S. aureus, defined by death or admission to a pediatric intensive care unit due to respiratory failure or hemodynamic instability.
The prevalence of PVL-positive S. aureus infection in the overall invasive infection group was 19%, while 8% of the isolates were MRSA. while MRSA was not associated with a significantly increased risk. The other independent risk factors for severe outcome were pneumonia, with an adjusted 13-fold increased risk, and leukopenia at admission, with an associated 18-fold risk (Clin Microbiol Infect. 2016 Jul;22[7]:643.e1-6.).
Of note, the virulence of PVL stems from the pore-forming toxin’s ability to lyse white blood cells. Because a leukocyte count is always available once a patient reaches the ED, severe leukopenia as defined by a count of less than 3,000 cells/mm3 at admission becomes a useful early marker of the likely severity of any case of S. aureus invasive disease, according to Dr. Rojo.
He highlighted four key syndromes involving severe invasive S. aureus infection in previously healthy children and adolescents that entail a high likelihood of being PVL positive and should cause physicians to run – not walk – to start appropriate empiric therapy. He also described the treatment regimen that he and other European thought leaders recommend for severe PVL-positive S. aureus invasive infections.
The microbiologic diagnosis of PVL can be made by ELISA (enzyme-linked immunoassay) to detect the toxin in an S. aureus isolate, by a rapid monoclonal antibody test, or by polymerase chain reaction to detect PVL genes in an S. aureus isolate. But don’t wait for test results to initiate treatment because these are high-mortality syndromes, he advised.
“Many people tell me, ‘My lab doesn’t have a way to diagnose PVL.’ And it’s true, it’s not available in real life at many hospitals. My message to you is that you don’t need to wait for a microbiological diagnosis or the results to come back from a sample you have sent to the reference lab in the main referral center. We can base our diagnosis and decision to treat on clinical grounds if we focus on these four very uncommon syndromes involving invasive S. aureus infection. I think if you have any child with these symptoms you have to manage them on the assumption that PVL is present,” said Dr. Rojo, principal investigator of the European Project on Invasive S. aureus Pediatric Infections.
The four key syndromes
The four syndromes are severe S. aureus pneumonia, S. aureus bone and joint infections with multiple foci, S. aureus osteomyelitis complicated by deep vein thrombosis, and invasive S. aureus infection plus shock.
- Severe S. aureus pneumonia. Investigators at Claude Bernard University in Lyon, France, have done extensive pioneering work on severe PVL-positive S. aureus invasive infections in children. In an early paper, they highlighted the characteristics that distinguish severe PVL-positive pneumonia: it typically occurs in previously healthy children and adolescents without underlying comorbid conditions, and it is often preceded by a influenza-like syndrome followed by an acute severe pneumonia with hemoptysis. Mortality was very high in this early series, with nearly half of the patients being dead within the first several days after admission (Lancet. 2002 Mar 2;359[9308]:753-9).
- Severe osteomyelitis. Investigators at Baylor College of Medicine, Houston, were among the first to observe that osteomyelitis caused by PVL-positive strains of S. aureus are associated with more severe local disease, with multiple affected areas, bigger abscesses, a greater systemic inflammatory response, and more surgeries required compared with osteomyelitis caused by PVL-negative S. aureus (Pediatrics. 2006 Feb;117[2]:433-40).
- Osteomyelitis with deep vein thrombosis. When a child hospitalized for acute hematogenous osteomyelitis due to S. aureus develops difficulty breathing, that’s a red flag for a severe PVL-positive infection involving deep vein thrombosis. Indeed, investigators at the Leeds (England) General Infirmary have reported that deep vein thrombosis in the setting of S. aureus osteomyelitis is associated with a greater than eightfold increased likelihood of a PVL-positive infection (Br J Hosp Med [Lond]. 2015 Jan;76[1]:18-24). Also, patients with PVL-positive osteomyelitis and deep vein thrombosis are prone to formation of septic emboli.
- Osteomyelitis with septic shock. The Lyon group compared outcomes in 14 pediatric patients with PVL-positive S. aureus osteomyelitis and a control group of 17 patients with PVL-negative disease. All 14 PVL-positive patients had severe sepsis and 6 of them had septic shock. In contrast, none of the controls did. Median duration of hospitalization was 46 days in the PVL-positive group, compared with 13 days in controls (Pediatr Infect Dis J. 2007 Nov;26[11]:1042-8).
Treatment
No randomized trials exist to guide treatment, but Dr. Rojo recommends the protocol utilized by the Lyon group: a bactericidal antibiotic – vancomycin or a beta-lactam – to take on the S. aureus, coupled with a ribosomally active antibiotic – clindamycin or linezolid – to suppress the PVL toxin’s virulence expression. The French group cites both in vitro and in vivo evidence that clindamycin and linezolid in their standard dosing have such an antitoxin effect (Clin Microbiol Rev. 2017 Oct;30[4]:887-917).
In addition, Dr. Rojo recommends utilizing any of the commercially available intravenous immunoglobulin (IVIG) products on the basis of work by investigators at Vanderbilt University in Nashville, Tenn., who have demonstrated that these products contain functional neutralizing antibodies against S. aureus leukocidins. This observation provides a likely explanation for anecdotal reports of improved outcomes in IVIG-treated patients with toxin-associated staphylococcal disease (Antimicrob Agents Chemother. 2017 Oct 24;61[11]. pii: e00968-17).
Challenged as to when specifically he would use IVIG in light of the global shortage of immunoglobulins, Dr. Rojo replied: “Not in every invasive S. aureus infection, but in serious infections that are PVL positive. I think if you have a child with one of these four syndromes who is in a pediatric ICU, you should use it. I mean, the mortality is around 30% in healthy children, so you would not stop from giving it. The risk of giving IVIG is very low, no side effects, so I highly recommend it for these severe cases.”
He reported having no financial conflicts.
MALMO, SWEDEN – Methicillin-resistant Staphylococcus aureus gets the blame in the Americas as the main cause of a great wave of community-acquired severe invasive staphylococcal infections in children and adolescents during the past nearly 2 decades, but many European pediatric infectious disease specialists believe that Panton-Valentine leukocidin (PVL), a frequent co-traveler with MRSA, is the true bad actor.
“The American literature focused first on MRSA, but we’ve seen very similar, very severe cases with MSSA [methicillin-susceptible S. aureus] PVL-positive and MRSA PVL-positive infections,” Pablo Rojo, MD, PhD, said at the annual meeting of the European Society for Paediatric Infectious Diseases.
“It is only because at the beginning there were so many MRSA cases in the States that they thought that was the driver of the disease. It is still unclear. There is still a discussion. But I wanted to bring you my opinion and that of many other authors that it’s mostly PVL-associated,” added Dr. Rojo of Complutense University in Madrid.
He was senior author of a multinational European and Israeli prospective study of risk factors associated with the severity of invasive community-acquired S. aureus infections in children, with invasive infection being defined as hospitalization for an infection with S. aureus isolated from a normally sterile body site such as blood, bone, or cerebrospinal fluid, or S. aureus pneumonia. They identified 152 affected children, 17% of whom had severe community-acquired invasive S. aureus, defined by death or admission to a pediatric intensive care unit due to respiratory failure or hemodynamic instability.
The prevalence of PVL-positive S. aureus infection in the overall invasive infection group was 19%, while 8% of the isolates were MRSA. while MRSA was not associated with a significantly increased risk. The other independent risk factors for severe outcome were pneumonia, with an adjusted 13-fold increased risk, and leukopenia at admission, with an associated 18-fold risk (Clin Microbiol Infect. 2016 Jul;22[7]:643.e1-6.).
Of note, the virulence of PVL stems from the pore-forming toxin’s ability to lyse white blood cells. Because a leukocyte count is always available once a patient reaches the ED, severe leukopenia as defined by a count of less than 3,000 cells/mm3 at admission becomes a useful early marker of the likely severity of any case of S. aureus invasive disease, according to Dr. Rojo.
He highlighted four key syndromes involving severe invasive S. aureus infection in previously healthy children and adolescents that entail a high likelihood of being PVL positive and should cause physicians to run – not walk – to start appropriate empiric therapy. He also described the treatment regimen that he and other European thought leaders recommend for severe PVL-positive S. aureus invasive infections.
The microbiologic diagnosis of PVL can be made by ELISA (enzyme-linked immunoassay) to detect the toxin in an S. aureus isolate, by a rapid monoclonal antibody test, or by polymerase chain reaction to detect PVL genes in an S. aureus isolate. But don’t wait for test results to initiate treatment because these are high-mortality syndromes, he advised.
“Many people tell me, ‘My lab doesn’t have a way to diagnose PVL.’ And it’s true, it’s not available in real life at many hospitals. My message to you is that you don’t need to wait for a microbiological diagnosis or the results to come back from a sample you have sent to the reference lab in the main referral center. We can base our diagnosis and decision to treat on clinical grounds if we focus on these four very uncommon syndromes involving invasive S. aureus infection. I think if you have any child with these symptoms you have to manage them on the assumption that PVL is present,” said Dr. Rojo, principal investigator of the European Project on Invasive S. aureus Pediatric Infections.
The four key syndromes
The four syndromes are severe S. aureus pneumonia, S. aureus bone and joint infections with multiple foci, S. aureus osteomyelitis complicated by deep vein thrombosis, and invasive S. aureus infection plus shock.
- Severe S. aureus pneumonia. Investigators at Claude Bernard University in Lyon, France, have done extensive pioneering work on severe PVL-positive S. aureus invasive infections in children. In an early paper, they highlighted the characteristics that distinguish severe PVL-positive pneumonia: it typically occurs in previously healthy children and adolescents without underlying comorbid conditions, and it is often preceded by a influenza-like syndrome followed by an acute severe pneumonia with hemoptysis. Mortality was very high in this early series, with nearly half of the patients being dead within the first several days after admission (Lancet. 2002 Mar 2;359[9308]:753-9).
- Severe osteomyelitis. Investigators at Baylor College of Medicine, Houston, were among the first to observe that osteomyelitis caused by PVL-positive strains of S. aureus are associated with more severe local disease, with multiple affected areas, bigger abscesses, a greater systemic inflammatory response, and more surgeries required compared with osteomyelitis caused by PVL-negative S. aureus (Pediatrics. 2006 Feb;117[2]:433-40).
- Osteomyelitis with deep vein thrombosis. When a child hospitalized for acute hematogenous osteomyelitis due to S. aureus develops difficulty breathing, that’s a red flag for a severe PVL-positive infection involving deep vein thrombosis. Indeed, investigators at the Leeds (England) General Infirmary have reported that deep vein thrombosis in the setting of S. aureus osteomyelitis is associated with a greater than eightfold increased likelihood of a PVL-positive infection (Br J Hosp Med [Lond]. 2015 Jan;76[1]:18-24). Also, patients with PVL-positive osteomyelitis and deep vein thrombosis are prone to formation of septic emboli.
- Osteomyelitis with septic shock. The Lyon group compared outcomes in 14 pediatric patients with PVL-positive S. aureus osteomyelitis and a control group of 17 patients with PVL-negative disease. All 14 PVL-positive patients had severe sepsis and 6 of them had septic shock. In contrast, none of the controls did. Median duration of hospitalization was 46 days in the PVL-positive group, compared with 13 days in controls (Pediatr Infect Dis J. 2007 Nov;26[11]:1042-8).
Treatment
No randomized trials exist to guide treatment, but Dr. Rojo recommends the protocol utilized by the Lyon group: a bactericidal antibiotic – vancomycin or a beta-lactam – to take on the S. aureus, coupled with a ribosomally active antibiotic – clindamycin or linezolid – to suppress the PVL toxin’s virulence expression. The French group cites both in vitro and in vivo evidence that clindamycin and linezolid in their standard dosing have such an antitoxin effect (Clin Microbiol Rev. 2017 Oct;30[4]:887-917).
In addition, Dr. Rojo recommends utilizing any of the commercially available intravenous immunoglobulin (IVIG) products on the basis of work by investigators at Vanderbilt University in Nashville, Tenn., who have demonstrated that these products contain functional neutralizing antibodies against S. aureus leukocidins. This observation provides a likely explanation for anecdotal reports of improved outcomes in IVIG-treated patients with toxin-associated staphylococcal disease (Antimicrob Agents Chemother. 2017 Oct 24;61[11]. pii: e00968-17).
Challenged as to when specifically he would use IVIG in light of the global shortage of immunoglobulins, Dr. Rojo replied: “Not in every invasive S. aureus infection, but in serious infections that are PVL positive. I think if you have a child with one of these four syndromes who is in a pediatric ICU, you should use it. I mean, the mortality is around 30% in healthy children, so you would not stop from giving it. The risk of giving IVIG is very low, no side effects, so I highly recommend it for these severe cases.”
He reported having no financial conflicts.
EXPERT ANALYSIS FROM ESPID 2018
FDA rejects mepolizumab on efficacy, but supports safety for COPD
Asthma drug mepolizumab could be added safely to inhaled corticosteroids for maintenance therapy to help reduce exacerbations in chronic obstructive pulmonary disease (COPD) patients who meet criteria for eosinophil counts, but the current data do not support its efficacy strongly enough for approval, according to a majority of members of the Food and Drug Administration’s Pulmonary-Allergy Drugs Advisory Committee.
The committee voted 16-3 that there was insufficient evidence of efficacy to support guided by eosinophil levels; they also voted 16-3 that the risk-benefit profile was not adequate to support approval.
However, on a voting question of safety, the committee voted 17-2 that the safety data on mepolizumab were sufficient to support approval.
Mepolizumab, a humanized monoclonal antibody, is currently approved for the treatment of asthma with eosinophilic phenotype for patients aged 12 years and older and for adults with eosinophilic granulomatosis with polyangiitis. Manufacturer GlaxoSmithKline is seeking approval for its use as an add-on therapy in COPD patients at a subcutaneous dose of 100 mg every 4 weeks. Mepolizumab works by binding to interleukin-5 (IL-5) and reducing eosinophil maturation and survival, which prompted GlaxoSmithKline to pursue an indication for COPD patients in a high-eosinophil stratum.
The application was supported in part by two concurrent randomized trials of 52 weeks’ duration.
Banu A. Karimi-Shah, MD, clinical team leader of the FDA’s Division of Pulmonary, Allergy, and Rheumatology Products, presented data from the two studies, referred to as Study 106 and Study 113.
In Study 106, researchers found statistically significant reductions in exacerbations for patients in the highest eosinophil group. However, challenges of the studies included a lack of consensus over the definition and possible relevance of an eosinophilic COPD phenotype, Dr. Karimi-Shah said in a presentation at the meeting.
In Study 113, mepolizumab had no significant impact on reducing moderate to severe exacerbations at either a 100-mg or 300-mg dose, Dr. Karimi-Shah said. In addition, most secondary endpoints, with the exception of reducing time to the first exacerbation among patients in the highest eosinophil group, did not consistently support the primary endpoint of exacerbation reduction in either study, she said.
Robert Busch, MD, also of the FDA’s Division of Pulmonary, Allergy, and Rheumatology products, served as a clinical reviewer and presented data on safety, efficacy, and risk-benefit profile of mepolizumab.
Dr. Busch noted that the variability in blood eosinophils make it challenging to use as a potential marker to identify patients who would benefit from mepolizumab as an add-on therapy.
Overall, most of the committee agreed on the existence of an eosinophilic COPD phenotype, but expressed concern about the threshold being used.
“The studies were not particularly well controlled regarding the characterization of patients,” said William J. Calhoun, MD, of the University of Texas Medical Branch, Galveston, who cast one of the ‘no’ votes on the question of efficacy.
By contrast, Jeffrey S. Wagener, MD, of the University of Colorado at Denver, Aurora, referenced his background in cystic fibrosis, and voted “yes” on the question of efficacy. “For patients that have no other option, this is a step forward,” he said.
Committee members on both sides of the vote emphasized the need for more research with larger numbers, better patient characterization, and more female patients. The committee members reported no relevant conflicts of interest.
Asthma drug mepolizumab could be added safely to inhaled corticosteroids for maintenance therapy to help reduce exacerbations in chronic obstructive pulmonary disease (COPD) patients who meet criteria for eosinophil counts, but the current data do not support its efficacy strongly enough for approval, according to a majority of members of the Food and Drug Administration’s Pulmonary-Allergy Drugs Advisory Committee.
The committee voted 16-3 that there was insufficient evidence of efficacy to support guided by eosinophil levels; they also voted 16-3 that the risk-benefit profile was not adequate to support approval.
However, on a voting question of safety, the committee voted 17-2 that the safety data on mepolizumab were sufficient to support approval.
Mepolizumab, a humanized monoclonal antibody, is currently approved for the treatment of asthma with eosinophilic phenotype for patients aged 12 years and older and for adults with eosinophilic granulomatosis with polyangiitis. Manufacturer GlaxoSmithKline is seeking approval for its use as an add-on therapy in COPD patients at a subcutaneous dose of 100 mg every 4 weeks. Mepolizumab works by binding to interleukin-5 (IL-5) and reducing eosinophil maturation and survival, which prompted GlaxoSmithKline to pursue an indication for COPD patients in a high-eosinophil stratum.
The application was supported in part by two concurrent randomized trials of 52 weeks’ duration.
Banu A. Karimi-Shah, MD, clinical team leader of the FDA’s Division of Pulmonary, Allergy, and Rheumatology Products, presented data from the two studies, referred to as Study 106 and Study 113.
In Study 106, researchers found statistically significant reductions in exacerbations for patients in the highest eosinophil group. However, challenges of the studies included a lack of consensus over the definition and possible relevance of an eosinophilic COPD phenotype, Dr. Karimi-Shah said in a presentation at the meeting.
In Study 113, mepolizumab had no significant impact on reducing moderate to severe exacerbations at either a 100-mg or 300-mg dose, Dr. Karimi-Shah said. In addition, most secondary endpoints, with the exception of reducing time to the first exacerbation among patients in the highest eosinophil group, did not consistently support the primary endpoint of exacerbation reduction in either study, she said.
Robert Busch, MD, also of the FDA’s Division of Pulmonary, Allergy, and Rheumatology products, served as a clinical reviewer and presented data on safety, efficacy, and risk-benefit profile of mepolizumab.
Dr. Busch noted that the variability in blood eosinophils make it challenging to use as a potential marker to identify patients who would benefit from mepolizumab as an add-on therapy.
Overall, most of the committee agreed on the existence of an eosinophilic COPD phenotype, but expressed concern about the threshold being used.
“The studies were not particularly well controlled regarding the characterization of patients,” said William J. Calhoun, MD, of the University of Texas Medical Branch, Galveston, who cast one of the ‘no’ votes on the question of efficacy.
By contrast, Jeffrey S. Wagener, MD, of the University of Colorado at Denver, Aurora, referenced his background in cystic fibrosis, and voted “yes” on the question of efficacy. “For patients that have no other option, this is a step forward,” he said.
Committee members on both sides of the vote emphasized the need for more research with larger numbers, better patient characterization, and more female patients. The committee members reported no relevant conflicts of interest.
Asthma drug mepolizumab could be added safely to inhaled corticosteroids for maintenance therapy to help reduce exacerbations in chronic obstructive pulmonary disease (COPD) patients who meet criteria for eosinophil counts, but the current data do not support its efficacy strongly enough for approval, according to a majority of members of the Food and Drug Administration’s Pulmonary-Allergy Drugs Advisory Committee.
The committee voted 16-3 that there was insufficient evidence of efficacy to support guided by eosinophil levels; they also voted 16-3 that the risk-benefit profile was not adequate to support approval.
However, on a voting question of safety, the committee voted 17-2 that the safety data on mepolizumab were sufficient to support approval.
Mepolizumab, a humanized monoclonal antibody, is currently approved for the treatment of asthma with eosinophilic phenotype for patients aged 12 years and older and for adults with eosinophilic granulomatosis with polyangiitis. Manufacturer GlaxoSmithKline is seeking approval for its use as an add-on therapy in COPD patients at a subcutaneous dose of 100 mg every 4 weeks. Mepolizumab works by binding to interleukin-5 (IL-5) and reducing eosinophil maturation and survival, which prompted GlaxoSmithKline to pursue an indication for COPD patients in a high-eosinophil stratum.
The application was supported in part by two concurrent randomized trials of 52 weeks’ duration.
Banu A. Karimi-Shah, MD, clinical team leader of the FDA’s Division of Pulmonary, Allergy, and Rheumatology Products, presented data from the two studies, referred to as Study 106 and Study 113.
In Study 106, researchers found statistically significant reductions in exacerbations for patients in the highest eosinophil group. However, challenges of the studies included a lack of consensus over the definition and possible relevance of an eosinophilic COPD phenotype, Dr. Karimi-Shah said in a presentation at the meeting.
In Study 113, mepolizumab had no significant impact on reducing moderate to severe exacerbations at either a 100-mg or 300-mg dose, Dr. Karimi-Shah said. In addition, most secondary endpoints, with the exception of reducing time to the first exacerbation among patients in the highest eosinophil group, did not consistently support the primary endpoint of exacerbation reduction in either study, she said.
Robert Busch, MD, also of the FDA’s Division of Pulmonary, Allergy, and Rheumatology products, served as a clinical reviewer and presented data on safety, efficacy, and risk-benefit profile of mepolizumab.
Dr. Busch noted that the variability in blood eosinophils make it challenging to use as a potential marker to identify patients who would benefit from mepolizumab as an add-on therapy.
Overall, most of the committee agreed on the existence of an eosinophilic COPD phenotype, but expressed concern about the threshold being used.
“The studies were not particularly well controlled regarding the characterization of patients,” said William J. Calhoun, MD, of the University of Texas Medical Branch, Galveston, who cast one of the ‘no’ votes on the question of efficacy.
By contrast, Jeffrey S. Wagener, MD, of the University of Colorado at Denver, Aurora, referenced his background in cystic fibrosis, and voted “yes” on the question of efficacy. “For patients that have no other option, this is a step forward,” he said.
Committee members on both sides of the vote emphasized the need for more research with larger numbers, better patient characterization, and more female patients. The committee members reported no relevant conflicts of interest.
FROM AN FDA ADVISORY COMMITTEE MEETING
ED key to reducing pediatric asthma x-rays
ATLANTA – but accomplishing this goal takes more than a new clinical practice guideline, according to a quality improvement team at the Monroe Carell Jr. Children’s Hospital at Vanderbilt University, Nashville, Tenn.
The team eventually reduced the chest x-ray rate for pediatric asthma exacerbations from 30% to 15% without increasing 3-day all-cause readmissions, but it took some sleuthing in the ED and good relations with staff. “We were way out in left field when we started this. Working in silos is never ideal,” said senior project member David Johnson, MD, a pediatric hospitalist and assistant professor of pediatrics at Vanderbilt.
It’s been known for a while that chest x-rays are almost always a waste of time and money for asthma exacerbations, and national guidelines recommend against them. X-rays don’t improve outcomes and needlessly expose children to radiation.
In 2014, some of the providers at Vanderbilt, which has about 1,700 asthma encounters a year, realized that the institution’s 30% x-ray rate was a problem. The quality improvement team hoped a new guideline would address the issue, but that didn’t happen. “We roll out clinical practice guidelines” from on high, “and think people will magically change their behavior,” but they don’t, Dr. Johnson said at the annual Pediatric Hospital Medicine meeting.
The guideline was not being fully implemented. So the team asked the ED what was the standard procedure for a child presenting with asthma exacerbation. It turned out that the ED had a dyspnea order set that the team ”had no idea existed.” Chest x-rays were at the top of the list; next came blood gases, ventilation-perfusion scans, and leg Dopplers, he said.
The investigators tried to get rid of the whole order set but were unsuccessful. The ED department did, however, let the team eliminate chest x-rays in the default order set in July 2015. That helped, but more changes were needed.
The next conversation was to figure out why x-rays were being ordered in the first place. ED staff said they were worried about missing something, especially pneumonia. They also thought they were helping hospitalists by getting x-rays before sending kids to the ward even though, in reality, it didn’t matter whether x-rays were done a few hours later on the floor. ED providers also said that ill-appearing children often got better after a few hours but were kept back from discharge because x-ray results were still pending and that sometimes these results revealed problems at 3 a.m. that had nothing to do with why the patients were in the ED but still required a work-up.
This discussion opened a door. The ED staff didn’t want to order unnecessary x-rays, either. That led to talks about letting kids declare themselves a bit before x-rays were ordered. ED staff liked the idea, so the guidelines were updated in early 2016 to say that chest x-rays should only be ordered if there is persistent severe respiratory distress with hypoxia, there are focal findings that don’t improve after 12 hours of treatment, or there were concerns for pneumomediastinum or collapsed lung. The updated guidelines were posted in work areas and brought home by resident education. A reminder was added to the electronic medical record system that popped up when someone tried to order a chest x-ray for an child with asthma.
It worked. Chest x-ray rates in asthma fell to 15%, and have remained there since.
“We gave them permission to take their foot off the throttle and wait a little bit, and we don’t have more kids bouncing back from reduced x-rays.” The approach is “probably generalizable everywhere,” Dr. Johnson said.
It was essential that an ED fellow, Caroline Watnick, MD, led the effort and eventually bridged the gap between hospitalists and ED providers. In the end, “the change wasn’t something from the outside,” Dr. Johnson said.
There was no industry funding, and Dr. Johnson didn’t have any disclosures. The Pediatric Hospital Medicine meeting is sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
ATLANTA – but accomplishing this goal takes more than a new clinical practice guideline, according to a quality improvement team at the Monroe Carell Jr. Children’s Hospital at Vanderbilt University, Nashville, Tenn.
The team eventually reduced the chest x-ray rate for pediatric asthma exacerbations from 30% to 15% without increasing 3-day all-cause readmissions, but it took some sleuthing in the ED and good relations with staff. “We were way out in left field when we started this. Working in silos is never ideal,” said senior project member David Johnson, MD, a pediatric hospitalist and assistant professor of pediatrics at Vanderbilt.
It’s been known for a while that chest x-rays are almost always a waste of time and money for asthma exacerbations, and national guidelines recommend against them. X-rays don’t improve outcomes and needlessly expose children to radiation.
In 2014, some of the providers at Vanderbilt, which has about 1,700 asthma encounters a year, realized that the institution’s 30% x-ray rate was a problem. The quality improvement team hoped a new guideline would address the issue, but that didn’t happen. “We roll out clinical practice guidelines” from on high, “and think people will magically change their behavior,” but they don’t, Dr. Johnson said at the annual Pediatric Hospital Medicine meeting.
The guideline was not being fully implemented. So the team asked the ED what was the standard procedure for a child presenting with asthma exacerbation. It turned out that the ED had a dyspnea order set that the team ”had no idea existed.” Chest x-rays were at the top of the list; next came blood gases, ventilation-perfusion scans, and leg Dopplers, he said.
The investigators tried to get rid of the whole order set but were unsuccessful. The ED department did, however, let the team eliminate chest x-rays in the default order set in July 2015. That helped, but more changes were needed.
The next conversation was to figure out why x-rays were being ordered in the first place. ED staff said they were worried about missing something, especially pneumonia. They also thought they were helping hospitalists by getting x-rays before sending kids to the ward even though, in reality, it didn’t matter whether x-rays were done a few hours later on the floor. ED providers also said that ill-appearing children often got better after a few hours but were kept back from discharge because x-ray results were still pending and that sometimes these results revealed problems at 3 a.m. that had nothing to do with why the patients were in the ED but still required a work-up.
This discussion opened a door. The ED staff didn’t want to order unnecessary x-rays, either. That led to talks about letting kids declare themselves a bit before x-rays were ordered. ED staff liked the idea, so the guidelines were updated in early 2016 to say that chest x-rays should only be ordered if there is persistent severe respiratory distress with hypoxia, there are focal findings that don’t improve after 12 hours of treatment, or there were concerns for pneumomediastinum or collapsed lung. The updated guidelines were posted in work areas and brought home by resident education. A reminder was added to the electronic medical record system that popped up when someone tried to order a chest x-ray for an child with asthma.
It worked. Chest x-ray rates in asthma fell to 15%, and have remained there since.
“We gave them permission to take their foot off the throttle and wait a little bit, and we don’t have more kids bouncing back from reduced x-rays.” The approach is “probably generalizable everywhere,” Dr. Johnson said.
It was essential that an ED fellow, Caroline Watnick, MD, led the effort and eventually bridged the gap between hospitalists and ED providers. In the end, “the change wasn’t something from the outside,” Dr. Johnson said.
There was no industry funding, and Dr. Johnson didn’t have any disclosures. The Pediatric Hospital Medicine meeting is sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
ATLANTA – but accomplishing this goal takes more than a new clinical practice guideline, according to a quality improvement team at the Monroe Carell Jr. Children’s Hospital at Vanderbilt University, Nashville, Tenn.
The team eventually reduced the chest x-ray rate for pediatric asthma exacerbations from 30% to 15% without increasing 3-day all-cause readmissions, but it took some sleuthing in the ED and good relations with staff. “We were way out in left field when we started this. Working in silos is never ideal,” said senior project member David Johnson, MD, a pediatric hospitalist and assistant professor of pediatrics at Vanderbilt.
It’s been known for a while that chest x-rays are almost always a waste of time and money for asthma exacerbations, and national guidelines recommend against them. X-rays don’t improve outcomes and needlessly expose children to radiation.
In 2014, some of the providers at Vanderbilt, which has about 1,700 asthma encounters a year, realized that the institution’s 30% x-ray rate was a problem. The quality improvement team hoped a new guideline would address the issue, but that didn’t happen. “We roll out clinical practice guidelines” from on high, “and think people will magically change their behavior,” but they don’t, Dr. Johnson said at the annual Pediatric Hospital Medicine meeting.
The guideline was not being fully implemented. So the team asked the ED what was the standard procedure for a child presenting with asthma exacerbation. It turned out that the ED had a dyspnea order set that the team ”had no idea existed.” Chest x-rays were at the top of the list; next came blood gases, ventilation-perfusion scans, and leg Dopplers, he said.
The investigators tried to get rid of the whole order set but were unsuccessful. The ED department did, however, let the team eliminate chest x-rays in the default order set in July 2015. That helped, but more changes were needed.
The next conversation was to figure out why x-rays were being ordered in the first place. ED staff said they were worried about missing something, especially pneumonia. They also thought they were helping hospitalists by getting x-rays before sending kids to the ward even though, in reality, it didn’t matter whether x-rays were done a few hours later on the floor. ED providers also said that ill-appearing children often got better after a few hours but were kept back from discharge because x-ray results were still pending and that sometimes these results revealed problems at 3 a.m. that had nothing to do with why the patients were in the ED but still required a work-up.
This discussion opened a door. The ED staff didn’t want to order unnecessary x-rays, either. That led to talks about letting kids declare themselves a bit before x-rays were ordered. ED staff liked the idea, so the guidelines were updated in early 2016 to say that chest x-rays should only be ordered if there is persistent severe respiratory distress with hypoxia, there are focal findings that don’t improve after 12 hours of treatment, or there were concerns for pneumomediastinum or collapsed lung. The updated guidelines were posted in work areas and brought home by resident education. A reminder was added to the electronic medical record system that popped up when someone tried to order a chest x-ray for an child with asthma.
It worked. Chest x-ray rates in asthma fell to 15%, and have remained there since.
“We gave them permission to take their foot off the throttle and wait a little bit, and we don’t have more kids bouncing back from reduced x-rays.” The approach is “probably generalizable everywhere,” Dr. Johnson said.
It was essential that an ED fellow, Caroline Watnick, MD, led the effort and eventually bridged the gap between hospitalists and ED providers. In the end, “the change wasn’t something from the outside,” Dr. Johnson said.
There was no industry funding, and Dr. Johnson didn’t have any disclosures. The Pediatric Hospital Medicine meeting is sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
REPORTING FROM PHM 2018
Key clinical point: Reduction of chest x-rays for routine pediatric asthma exacerbations in the ED can be accomplished with a team effort.
Major finding: A team project reduced x-rays for pediatric asthma exacerbations from 30% to 15% without increasing 3-day, all-cause readmissions.
Study details: Pre/post quality improvement analysis of asthma encounters in the Monroe Carell Jr. Children’s Hospital, Nashville, Tenn., starting in 2014.
Disclosures: There was no industry funding, and the presenter didn’t have any disclosures.
DTPa-HBV-IPV/Hib in infancy maintains lasting immune memory against HBV in teens
MALMO, SWEDEN – Four doses of hexavalent diphtheria-tetanus-acellular pertussis-hepatitis B-inactivated poliovirus/Haemophilus influenza type b vaccine given in infancy provides reassuringly long-lasting immune memory against hepatitis B among 14- to 15-year-olds, Tino F. Schwarz, MD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
He presented the fourth and final study in a series evaluating the antibody persistence and immune memory against hepatitis B (HBV) in recipients of the complete four-dose series of hexavalent DTPa-HBV-IPV/Hib vaccine in infancy. Because exposure to HBV can increase during adolescence, it was essential to determine whether antibody persistence is maintained, explained Dr. Schwarz of Juliusspital Hospital in Wurzburg, Germany.
“As expected, we saw a decrease in anti-HBs [hepatitis B surface antigen] antibody levels over the years, with persistent seroprotection in 85% of children at age 4-5 years, 72% at 7-8 years, 61% at 12-13 years, and now 54% of adolescents at 14-15 years. But we could demonstrate a very strong anamnestic response in the trial. This is good information. It clearly shows that, in patients who are exposed to hepatitis B, we can certainly guarantee that they are protected. It’s a good result for public health. The vaccine is a very robust vaccine which induces a very strong response over the years. It can be boosted, but from an immunologic point of view it is not required,” he said.
The multicenter study included 268 adolescents aged 14-15 years who had received the four-dose hexavalent vaccine series in infancy. Their antibody persistence against anti-HBs was measured, then measured once again 1 month after receiving a challenge dose of monovalent HBV vaccine.
Prechallenge, 105 of the teens were seronegative, 144 were seroprotected as defined by an anti-HBs concentration of at least 10 mIU/mL, and 19 had low seropositivity marked by an antibody level of 6 to less than 10 mIU/mL. Yet 1 month after the booster, which was intended to mimic the impact of real-world exposure to HBV, 83% of the initially seronegative subjects had an anti-HBs concentration of 10 mIU/mL or more, and 67% of them had a level of at least 100 mIU/mL.
“We saw a clear fantastic anamnestic response,” Dr. Schwarz declared.
Overall, 93% of study participants seroconverted, and 87% of them had anti-HBs titers of 100 mIU/mL, “which is the level we’d like to achieve in vaccinees,” he observed.
The booster monovalent HBV vaccine was well tolerated, with one-third of subjects complaining of mild local injection site pain and 30% noting fatigue. But in response to a question posed by session chair Ronald de Groot, MD, emeritus professor of pediatrics at Radboud University in Nijmegen, the Netherlands, Dr. Schwarz said these study results indicate there’s no need for routine boosting in healthy adolescents such as those in the trial. Immunocompromised individuals might be a different story, but they weren’t investigated.
But what about in physicians and surgeons, where protection against HBV infection is essential? Dr. de Groot asked.
“In Germany, we require a titer of 100 mIU/mL or more in medical staff, but we’re quite alone in Europe. Other countries do not require booster vaccination for medical staff. The data we’ve shown here is quite reassuring: If you get exposed, you in effect get a booster. It’s complicated to test surgeons in their offices; better to just rely on the anamnestic response that we’ve demonstrated,” Dr. Schwarz replied.
He reported serving as a consultant to GlaxoSmithKline, which funded the study, as well as to Pfizer and Sanofi Pasteur.
MALMO, SWEDEN – Four doses of hexavalent diphtheria-tetanus-acellular pertussis-hepatitis B-inactivated poliovirus/Haemophilus influenza type b vaccine given in infancy provides reassuringly long-lasting immune memory against hepatitis B among 14- to 15-year-olds, Tino F. Schwarz, MD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
He presented the fourth and final study in a series evaluating the antibody persistence and immune memory against hepatitis B (HBV) in recipients of the complete four-dose series of hexavalent DTPa-HBV-IPV/Hib vaccine in infancy. Because exposure to HBV can increase during adolescence, it was essential to determine whether antibody persistence is maintained, explained Dr. Schwarz of Juliusspital Hospital in Wurzburg, Germany.
“As expected, we saw a decrease in anti-HBs [hepatitis B surface antigen] antibody levels over the years, with persistent seroprotection in 85% of children at age 4-5 years, 72% at 7-8 years, 61% at 12-13 years, and now 54% of adolescents at 14-15 years. But we could demonstrate a very strong anamnestic response in the trial. This is good information. It clearly shows that, in patients who are exposed to hepatitis B, we can certainly guarantee that they are protected. It’s a good result for public health. The vaccine is a very robust vaccine which induces a very strong response over the years. It can be boosted, but from an immunologic point of view it is not required,” he said.
The multicenter study included 268 adolescents aged 14-15 years who had received the four-dose hexavalent vaccine series in infancy. Their antibody persistence against anti-HBs was measured, then measured once again 1 month after receiving a challenge dose of monovalent HBV vaccine.
Prechallenge, 105 of the teens were seronegative, 144 were seroprotected as defined by an anti-HBs concentration of at least 10 mIU/mL, and 19 had low seropositivity marked by an antibody level of 6 to less than 10 mIU/mL. Yet 1 month after the booster, which was intended to mimic the impact of real-world exposure to HBV, 83% of the initially seronegative subjects had an anti-HBs concentration of 10 mIU/mL or more, and 67% of them had a level of at least 100 mIU/mL.
“We saw a clear fantastic anamnestic response,” Dr. Schwarz declared.
Overall, 93% of study participants seroconverted, and 87% of them had anti-HBs titers of 100 mIU/mL, “which is the level we’d like to achieve in vaccinees,” he observed.
The booster monovalent HBV vaccine was well tolerated, with one-third of subjects complaining of mild local injection site pain and 30% noting fatigue. But in response to a question posed by session chair Ronald de Groot, MD, emeritus professor of pediatrics at Radboud University in Nijmegen, the Netherlands, Dr. Schwarz said these study results indicate there’s no need for routine boosting in healthy adolescents such as those in the trial. Immunocompromised individuals might be a different story, but they weren’t investigated.
But what about in physicians and surgeons, where protection against HBV infection is essential? Dr. de Groot asked.
“In Germany, we require a titer of 100 mIU/mL or more in medical staff, but we’re quite alone in Europe. Other countries do not require booster vaccination for medical staff. The data we’ve shown here is quite reassuring: If you get exposed, you in effect get a booster. It’s complicated to test surgeons in their offices; better to just rely on the anamnestic response that we’ve demonstrated,” Dr. Schwarz replied.
He reported serving as a consultant to GlaxoSmithKline, which funded the study, as well as to Pfizer and Sanofi Pasteur.
MALMO, SWEDEN – Four doses of hexavalent diphtheria-tetanus-acellular pertussis-hepatitis B-inactivated poliovirus/Haemophilus influenza type b vaccine given in infancy provides reassuringly long-lasting immune memory against hepatitis B among 14- to 15-year-olds, Tino F. Schwarz, MD, reported at the annual meeting of the European Society for Paediatric Infectious Diseases.
He presented the fourth and final study in a series evaluating the antibody persistence and immune memory against hepatitis B (HBV) in recipients of the complete four-dose series of hexavalent DTPa-HBV-IPV/Hib vaccine in infancy. Because exposure to HBV can increase during adolescence, it was essential to determine whether antibody persistence is maintained, explained Dr. Schwarz of Juliusspital Hospital in Wurzburg, Germany.
“As expected, we saw a decrease in anti-HBs [hepatitis B surface antigen] antibody levels over the years, with persistent seroprotection in 85% of children at age 4-5 years, 72% at 7-8 years, 61% at 12-13 years, and now 54% of adolescents at 14-15 years. But we could demonstrate a very strong anamnestic response in the trial. This is good information. It clearly shows that, in patients who are exposed to hepatitis B, we can certainly guarantee that they are protected. It’s a good result for public health. The vaccine is a very robust vaccine which induces a very strong response over the years. It can be boosted, but from an immunologic point of view it is not required,” he said.
The multicenter study included 268 adolescents aged 14-15 years who had received the four-dose hexavalent vaccine series in infancy. Their antibody persistence against anti-HBs was measured, then measured once again 1 month after receiving a challenge dose of monovalent HBV vaccine.
Prechallenge, 105 of the teens were seronegative, 144 were seroprotected as defined by an anti-HBs concentration of at least 10 mIU/mL, and 19 had low seropositivity marked by an antibody level of 6 to less than 10 mIU/mL. Yet 1 month after the booster, which was intended to mimic the impact of real-world exposure to HBV, 83% of the initially seronegative subjects had an anti-HBs concentration of 10 mIU/mL or more, and 67% of them had a level of at least 100 mIU/mL.
“We saw a clear fantastic anamnestic response,” Dr. Schwarz declared.
Overall, 93% of study participants seroconverted, and 87% of them had anti-HBs titers of 100 mIU/mL, “which is the level we’d like to achieve in vaccinees,” he observed.
The booster monovalent HBV vaccine was well tolerated, with one-third of subjects complaining of mild local injection site pain and 30% noting fatigue. But in response to a question posed by session chair Ronald de Groot, MD, emeritus professor of pediatrics at Radboud University in Nijmegen, the Netherlands, Dr. Schwarz said these study results indicate there’s no need for routine boosting in healthy adolescents such as those in the trial. Immunocompromised individuals might be a different story, but they weren’t investigated.
But what about in physicians and surgeons, where protection against HBV infection is essential? Dr. de Groot asked.
“In Germany, we require a titer of 100 mIU/mL or more in medical staff, but we’re quite alone in Europe. Other countries do not require booster vaccination for medical staff. The data we’ve shown here is quite reassuring: If you get exposed, you in effect get a booster. It’s complicated to test surgeons in their offices; better to just rely on the anamnestic response that we’ve demonstrated,” Dr. Schwarz replied.
He reported serving as a consultant to GlaxoSmithKline, which funded the study, as well as to Pfizer and Sanofi Pasteur.
REPORTING FROM ESPID 2018
Key clinical point:
Major finding: Ninety-three percent of recipients of four doses of hexavalent DTPa-HBV-IPV/Hib vaccine in infancy were seroprotected against HBV at age 14-15 years.
Study details: This was a prospective study of antibody persistence and immune memory in 268 teens aged 14-15 before and 1 month after receiving a booster challenge HBV monovalent vaccine.
Disclosures: The presenter reported serving as a consultant to GlaxoSmithKline, which funded the study, as well as to Pfizer and Sanofi Pasteur.
Asthma medication ratio identifies high-risk pediatric patients
ATLANTA – An according to researchers from the Medical University of South Carolina (MUSC), Charleston.
The asthma medication ratio (AMR) – the number of prescriptions for controller medications divided by the number of prescriptions for both controller and rescue medications – has been around for a while, but it’s mostly been used as a quality metric. The new study shows that it’s also useful in the clinic to identify children who could benefit from extra attention.
A perfect ratio of 1 means that control is good without rescue inhalers. The ratio falls as the number of rescue inhalers goes up, signaling poorer control. Children with a ratio below 0.5 are considered high risk; they’d hit that mark if, for instance, they were prescribed one control medication such as fluticasone propionate (Flovent) and two albuterol rescue inhalers in a month.
If control is good, “you should only need a rescue inhaler very, very sporadically;” high-risk children probably need a higher dose of their controller, or help with compliance, explained lead investigator Annie L. Andrews, MD, associate professor of pediatrics at MUSC.
The university uses the EPIC records system, which incorporates prescription data from Surescripts, so the number of asthma medication fills is already available. The system just needs to be adjusted to calculate and report AMRs monthly, something Dr. Andrews and her team are working on. “The information is right there, but it’s an untapped resource,” she said. “We just need to crunch the numbers, and operationalize it. Why are we waiting until kids are in the hospital” to intervene?
Dr. Andrews presented a proof-of-concept study at the Pediatric Hospital Medicine meeting. Her team identified 214,452 asthma patients aged 2-17 years with at least one claim for an inhaled corticosteroid in the Truven MarketScan Medicaid database from 2013-14.
They calculated AMRs for each child every 3 months over a 15-month period. About 9% of children at any given time had AMRs below 0.5.
The first AMR was at or above 0.5 in 93,512 children; 18.1% had a subsequent asthma-related event, meaning an ED visit or hospitalization, during the course of the study. Among the 17,635 children with an initial AMR below 0.5, 25% had asthma-related events. The initial AMR couldn’t be calculated in 103,305 children, which likely meant they had less-active disease. Those children had the lowest proportion of asthma events, at 13.9%.
An AMR below 0.5 nearly doubled the risk of an asthma-related hospitalization or ED visit in the subsequent 3 months, with an odds ratios ranging from 1.7 to 1.9, compared with other children. The findings were statistically significant.
In short, serial AMRs helped predict exacerbations among Medicaid children. The team showed the same trend among commercially insured children in a recently published study. The only difference was that Medicaid children had a higher proportion of high-risk AMRs, and a higher number of asthma events (Am J Manag Care. 2018 Jun;24[6]:294-300). Together, the studies validate “the rolling 3-month AMR as an appropriate method for identifying children at high risk for imminent exacerbation,” the investigators concluded.
With automatic AMR reporting already in the works at MUSC, “we are now trying to figure out how to intervene. Do we just tell providers who their high-risk kids are and let them figure out how to contact families, or do we use this information to contact families directly? That’s kind of what I favor: ‘Hey, your kid just popped up as high risk, so let’s figure out what you need. Do you need a new prescription or a reminder to see your doctor?’ ” Dr. Andrews said.
Her team is developing a mobile app to communicate with families.
The mean age in the study was 7.9 years; 59% of the children were boys, and 41% were black.
The work was funded by the National Institutes of Health, among others. Dr. Andrews had no disclosures. The meeting was sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
ATLANTA – An according to researchers from the Medical University of South Carolina (MUSC), Charleston.
The asthma medication ratio (AMR) – the number of prescriptions for controller medications divided by the number of prescriptions for both controller and rescue medications – has been around for a while, but it’s mostly been used as a quality metric. The new study shows that it’s also useful in the clinic to identify children who could benefit from extra attention.
A perfect ratio of 1 means that control is good without rescue inhalers. The ratio falls as the number of rescue inhalers goes up, signaling poorer control. Children with a ratio below 0.5 are considered high risk; they’d hit that mark if, for instance, they were prescribed one control medication such as fluticasone propionate (Flovent) and two albuterol rescue inhalers in a month.
If control is good, “you should only need a rescue inhaler very, very sporadically;” high-risk children probably need a higher dose of their controller, or help with compliance, explained lead investigator Annie L. Andrews, MD, associate professor of pediatrics at MUSC.
The university uses the EPIC records system, which incorporates prescription data from Surescripts, so the number of asthma medication fills is already available. The system just needs to be adjusted to calculate and report AMRs monthly, something Dr. Andrews and her team are working on. “The information is right there, but it’s an untapped resource,” she said. “We just need to crunch the numbers, and operationalize it. Why are we waiting until kids are in the hospital” to intervene?
Dr. Andrews presented a proof-of-concept study at the Pediatric Hospital Medicine meeting. Her team identified 214,452 asthma patients aged 2-17 years with at least one claim for an inhaled corticosteroid in the Truven MarketScan Medicaid database from 2013-14.
They calculated AMRs for each child every 3 months over a 15-month period. About 9% of children at any given time had AMRs below 0.5.
The first AMR was at or above 0.5 in 93,512 children; 18.1% had a subsequent asthma-related event, meaning an ED visit or hospitalization, during the course of the study. Among the 17,635 children with an initial AMR below 0.5, 25% had asthma-related events. The initial AMR couldn’t be calculated in 103,305 children, which likely meant they had less-active disease. Those children had the lowest proportion of asthma events, at 13.9%.
An AMR below 0.5 nearly doubled the risk of an asthma-related hospitalization or ED visit in the subsequent 3 months, with an odds ratios ranging from 1.7 to 1.9, compared with other children. The findings were statistically significant.
In short, serial AMRs helped predict exacerbations among Medicaid children. The team showed the same trend among commercially insured children in a recently published study. The only difference was that Medicaid children had a higher proportion of high-risk AMRs, and a higher number of asthma events (Am J Manag Care. 2018 Jun;24[6]:294-300). Together, the studies validate “the rolling 3-month AMR as an appropriate method for identifying children at high risk for imminent exacerbation,” the investigators concluded.
With automatic AMR reporting already in the works at MUSC, “we are now trying to figure out how to intervene. Do we just tell providers who their high-risk kids are and let them figure out how to contact families, or do we use this information to contact families directly? That’s kind of what I favor: ‘Hey, your kid just popped up as high risk, so let’s figure out what you need. Do you need a new prescription or a reminder to see your doctor?’ ” Dr. Andrews said.
Her team is developing a mobile app to communicate with families.
The mean age in the study was 7.9 years; 59% of the children were boys, and 41% were black.
The work was funded by the National Institutes of Health, among others. Dr. Andrews had no disclosures. The meeting was sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
ATLANTA – An according to researchers from the Medical University of South Carolina (MUSC), Charleston.
The asthma medication ratio (AMR) – the number of prescriptions for controller medications divided by the number of prescriptions for both controller and rescue medications – has been around for a while, but it’s mostly been used as a quality metric. The new study shows that it’s also useful in the clinic to identify children who could benefit from extra attention.
A perfect ratio of 1 means that control is good without rescue inhalers. The ratio falls as the number of rescue inhalers goes up, signaling poorer control. Children with a ratio below 0.5 are considered high risk; they’d hit that mark if, for instance, they were prescribed one control medication such as fluticasone propionate (Flovent) and two albuterol rescue inhalers in a month.
If control is good, “you should only need a rescue inhaler very, very sporadically;” high-risk children probably need a higher dose of their controller, or help with compliance, explained lead investigator Annie L. Andrews, MD, associate professor of pediatrics at MUSC.
The university uses the EPIC records system, which incorporates prescription data from Surescripts, so the number of asthma medication fills is already available. The system just needs to be adjusted to calculate and report AMRs monthly, something Dr. Andrews and her team are working on. “The information is right there, but it’s an untapped resource,” she said. “We just need to crunch the numbers, and operationalize it. Why are we waiting until kids are in the hospital” to intervene?
Dr. Andrews presented a proof-of-concept study at the Pediatric Hospital Medicine meeting. Her team identified 214,452 asthma patients aged 2-17 years with at least one claim for an inhaled corticosteroid in the Truven MarketScan Medicaid database from 2013-14.
They calculated AMRs for each child every 3 months over a 15-month period. About 9% of children at any given time had AMRs below 0.5.
The first AMR was at or above 0.5 in 93,512 children; 18.1% had a subsequent asthma-related event, meaning an ED visit or hospitalization, during the course of the study. Among the 17,635 children with an initial AMR below 0.5, 25% had asthma-related events. The initial AMR couldn’t be calculated in 103,305 children, which likely meant they had less-active disease. Those children had the lowest proportion of asthma events, at 13.9%.
An AMR below 0.5 nearly doubled the risk of an asthma-related hospitalization or ED visit in the subsequent 3 months, with an odds ratios ranging from 1.7 to 1.9, compared with other children. The findings were statistically significant.
In short, serial AMRs helped predict exacerbations among Medicaid children. The team showed the same trend among commercially insured children in a recently published study. The only difference was that Medicaid children had a higher proportion of high-risk AMRs, and a higher number of asthma events (Am J Manag Care. 2018 Jun;24[6]:294-300). Together, the studies validate “the rolling 3-month AMR as an appropriate method for identifying children at high risk for imminent exacerbation,” the investigators concluded.
With automatic AMR reporting already in the works at MUSC, “we are now trying to figure out how to intervene. Do we just tell providers who their high-risk kids are and let them figure out how to contact families, or do we use this information to contact families directly? That’s kind of what I favor: ‘Hey, your kid just popped up as high risk, so let’s figure out what you need. Do you need a new prescription or a reminder to see your doctor?’ ” Dr. Andrews said.
Her team is developing a mobile app to communicate with families.
The mean age in the study was 7.9 years; 59% of the children were boys, and 41% were black.
The work was funded by the National Institutes of Health, among others. Dr. Andrews had no disclosures. The meeting was sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
REPORTING FROM PHM 2018
Key clinical point: The asthma medication ratio is useful in the clinic to identify children who could benefit from extra attention.
Major finding: About 9% of children at any given time had AMRs below 0.5, meaning they were at high risk for acute exacerbations.
Study details: Review of more than 200,000 pediatric asthma patients on Medicaid
Disclosures: The work was funded by the National Institutes of Health, among others. The study lead had no disclosures.
Ten tips for managing patients with both heart failure and COPD
Patients with both are prone to hospital readmissions that detract from quality of life and dramatically drive up care costs.
Because the two chronic diseases spring from the same root cause and share overlapping symptoms, strategies that improve clinical outcomes in one can also benefit the other, Ravi Kalhan, MD, and R. Kannan Mutharasan, MD, wrote in CHEST Journal (doi: 10.1016/j.chest.2018.06.001).
“Both conditions are characterized by periods of clinical stability punctuated by episodes of exacerbation and are typified by gradual functional decline,” wrote the colleagues, both of Northwestern University, Chicago. “From a patient perspective, both conditions lead to highly overlapping patterns of symptoms, involve complicated medication regimens, and have courses highly sensitive to adherence and lifestyle modification. Therefore, disease management strategies for both conditions can be synergistic.”
The team came up with a “Top 10 list” of practical tips for reducing readmissions in patients with this challenging combination.
Diagnose accurately
An acute hospitalization is often the first time these patients pop up on the radar. This is a great time to employ spirometry to accurately diagnose COPD. It’s also appropriate to conduct a chest CT and check right heart size, diameter of the pulmonary artery, and the presence of coronary calcification. The authors noted that relatively little is known about the course of patients with combined asthma and HF in contrast to COPD and HF.
Detect admissions for exacerbations early
Check soon to find out if this is a readmission, get an acute plan going, and don’t wait to implement multidisciplinary interventions. “First, specialist involvement can occur more rapidly, allowing for faster identification of any root causes driving the HF or COPD syndromes, and allowing for more rapid institution of treatment plans to control the acute exacerbation. Second, early identification during hospitalization allows time to deploy multidisciplinary interventions, such as disease management education, social work evaluation, follow-up appointment scheduling, and coordination of home services. These interventions are less effective, and are often not implemented, if initiated toward the end of hospitalization.”
Use specialist management in the hospital
Get experts on board fast. An integrated team means a coordinated treatment plan that’s easier to follow and more effective therapeutically. Specialist care may impact rates of readmission: weight loss with diuretics; discharge doses of guideline-directed medical therapy for heart failure; and higher rates of discharge on long-acting beta-agonists, long-acting muscarinic antagonists, inhaled corticosteroids, and home supplemental oxygen.
Modify the underlying disease substrate
Heart failure is more likely to arise from a correctable pathophysiology, so find it early and treat it thoroughly – especially in younger patients. Ischemic heart disease, valvular heart disease, systemic hypertension, and pulmonary hypertension all have potential to make the HF syndrome more tractable.
Apply and intensify evidence-based therapies
Start in the hospital if possible; if not, begin upon discharge. “The order of application of these therapies can be bewildering, as many strategies for initiation and up-titration of these medications are reasonable. Not only are there long-term outcome benefits for these therapies, evidence suggests early initiation of HF therapies can reduce 30-day readmissions.”
Activate the patient and develop critical health behaviors
Medical regimens for these diseases can be complex, and they must be supported by patient engagement. “Many strategies for engaging patients in care have been tested, including teaching to goal, motivational interviewing, and teach-back methods of activation and engagement. Often these methods are time intensive. Because physician time is increasingly constrained, a team approach is particularly useful.”
Set up feedback loops
“Course correction should the patient decompensate is critically important to maintaining outpatient success. Feedback loops can allow for clinical stabilization before rehospitalization is necessary.” Self-monitoring with individually set benchmarks is critical.
Arrange an early follow-up appointment prior to discharge
About half of Medicare patients with these conditions are readmitted before they’ve even had a postdischarge follow-up appointment. Ideally this should occur within 7 days. The purpose of early follow-up is to identify and address gaps in the discharge plan of care, revise the discharge plan of care to adapt to the outpatient environment, and reinforce critical health behaviors.
Consider and address other comorbidities
Comorbidities are the rule rather than the exception and contribute to many readmissions. Get primary care on the team and enlist their help in managing these issues before they lead to an exacerbation. “Meticulous control – even perfect control were it possible – of cardiopulmonary disease would still leave patients vulnerable to significant risk of readmission from other causes.”
Consider ancillary supportive services at home
Patients may be overwhelmed by the complexity of postdischarge care. Home health assistance can help in getting patients to physical therapy, continuing patient education, and providing a home clinical assessment.
Neither of the authors had any financial disclosures.
Patients with both are prone to hospital readmissions that detract from quality of life and dramatically drive up care costs.
Because the two chronic diseases spring from the same root cause and share overlapping symptoms, strategies that improve clinical outcomes in one can also benefit the other, Ravi Kalhan, MD, and R. Kannan Mutharasan, MD, wrote in CHEST Journal (doi: 10.1016/j.chest.2018.06.001).
“Both conditions are characterized by periods of clinical stability punctuated by episodes of exacerbation and are typified by gradual functional decline,” wrote the colleagues, both of Northwestern University, Chicago. “From a patient perspective, both conditions lead to highly overlapping patterns of symptoms, involve complicated medication regimens, and have courses highly sensitive to adherence and lifestyle modification. Therefore, disease management strategies for both conditions can be synergistic.”
The team came up with a “Top 10 list” of practical tips for reducing readmissions in patients with this challenging combination.
Diagnose accurately
An acute hospitalization is often the first time these patients pop up on the radar. This is a great time to employ spirometry to accurately diagnose COPD. It’s also appropriate to conduct a chest CT and check right heart size, diameter of the pulmonary artery, and the presence of coronary calcification. The authors noted that relatively little is known about the course of patients with combined asthma and HF in contrast to COPD and HF.
Detect admissions for exacerbations early
Check soon to find out if this is a readmission, get an acute plan going, and don’t wait to implement multidisciplinary interventions. “First, specialist involvement can occur more rapidly, allowing for faster identification of any root causes driving the HF or COPD syndromes, and allowing for more rapid institution of treatment plans to control the acute exacerbation. Second, early identification during hospitalization allows time to deploy multidisciplinary interventions, such as disease management education, social work evaluation, follow-up appointment scheduling, and coordination of home services. These interventions are less effective, and are often not implemented, if initiated toward the end of hospitalization.”
Use specialist management in the hospital
Get experts on board fast. An integrated team means a coordinated treatment plan that’s easier to follow and more effective therapeutically. Specialist care may impact rates of readmission: weight loss with diuretics; discharge doses of guideline-directed medical therapy for heart failure; and higher rates of discharge on long-acting beta-agonists, long-acting muscarinic antagonists, inhaled corticosteroids, and home supplemental oxygen.
Modify the underlying disease substrate
Heart failure is more likely to arise from a correctable pathophysiology, so find it early and treat it thoroughly – especially in younger patients. Ischemic heart disease, valvular heart disease, systemic hypertension, and pulmonary hypertension all have potential to make the HF syndrome more tractable.
Apply and intensify evidence-based therapies
Start in the hospital if possible; if not, begin upon discharge. “The order of application of these therapies can be bewildering, as many strategies for initiation and up-titration of these medications are reasonable. Not only are there long-term outcome benefits for these therapies, evidence suggests early initiation of HF therapies can reduce 30-day readmissions.”
Activate the patient and develop critical health behaviors
Medical regimens for these diseases can be complex, and they must be supported by patient engagement. “Many strategies for engaging patients in care have been tested, including teaching to goal, motivational interviewing, and teach-back methods of activation and engagement. Often these methods are time intensive. Because physician time is increasingly constrained, a team approach is particularly useful.”
Set up feedback loops
“Course correction should the patient decompensate is critically important to maintaining outpatient success. Feedback loops can allow for clinical stabilization before rehospitalization is necessary.” Self-monitoring with individually set benchmarks is critical.
Arrange an early follow-up appointment prior to discharge
About half of Medicare patients with these conditions are readmitted before they’ve even had a postdischarge follow-up appointment. Ideally this should occur within 7 days. The purpose of early follow-up is to identify and address gaps in the discharge plan of care, revise the discharge plan of care to adapt to the outpatient environment, and reinforce critical health behaviors.
Consider and address other comorbidities
Comorbidities are the rule rather than the exception and contribute to many readmissions. Get primary care on the team and enlist their help in managing these issues before they lead to an exacerbation. “Meticulous control – even perfect control were it possible – of cardiopulmonary disease would still leave patients vulnerable to significant risk of readmission from other causes.”
Consider ancillary supportive services at home
Patients may be overwhelmed by the complexity of postdischarge care. Home health assistance can help in getting patients to physical therapy, continuing patient education, and providing a home clinical assessment.
Neither of the authors had any financial disclosures.
Patients with both are prone to hospital readmissions that detract from quality of life and dramatically drive up care costs.
Because the two chronic diseases spring from the same root cause and share overlapping symptoms, strategies that improve clinical outcomes in one can also benefit the other, Ravi Kalhan, MD, and R. Kannan Mutharasan, MD, wrote in CHEST Journal (doi: 10.1016/j.chest.2018.06.001).
“Both conditions are characterized by periods of clinical stability punctuated by episodes of exacerbation and are typified by gradual functional decline,” wrote the colleagues, both of Northwestern University, Chicago. “From a patient perspective, both conditions lead to highly overlapping patterns of symptoms, involve complicated medication regimens, and have courses highly sensitive to adherence and lifestyle modification. Therefore, disease management strategies for both conditions can be synergistic.”
The team came up with a “Top 10 list” of practical tips for reducing readmissions in patients with this challenging combination.
Diagnose accurately
An acute hospitalization is often the first time these patients pop up on the radar. This is a great time to employ spirometry to accurately diagnose COPD. It’s also appropriate to conduct a chest CT and check right heart size, diameter of the pulmonary artery, and the presence of coronary calcification. The authors noted that relatively little is known about the course of patients with combined asthma and HF in contrast to COPD and HF.
Detect admissions for exacerbations early
Check soon to find out if this is a readmission, get an acute plan going, and don’t wait to implement multidisciplinary interventions. “First, specialist involvement can occur more rapidly, allowing for faster identification of any root causes driving the HF or COPD syndromes, and allowing for more rapid institution of treatment plans to control the acute exacerbation. Second, early identification during hospitalization allows time to deploy multidisciplinary interventions, such as disease management education, social work evaluation, follow-up appointment scheduling, and coordination of home services. These interventions are less effective, and are often not implemented, if initiated toward the end of hospitalization.”
Use specialist management in the hospital
Get experts on board fast. An integrated team means a coordinated treatment plan that’s easier to follow and more effective therapeutically. Specialist care may impact rates of readmission: weight loss with diuretics; discharge doses of guideline-directed medical therapy for heart failure; and higher rates of discharge on long-acting beta-agonists, long-acting muscarinic antagonists, inhaled corticosteroids, and home supplemental oxygen.
Modify the underlying disease substrate
Heart failure is more likely to arise from a correctable pathophysiology, so find it early and treat it thoroughly – especially in younger patients. Ischemic heart disease, valvular heart disease, systemic hypertension, and pulmonary hypertension all have potential to make the HF syndrome more tractable.
Apply and intensify evidence-based therapies
Start in the hospital if possible; if not, begin upon discharge. “The order of application of these therapies can be bewildering, as many strategies for initiation and up-titration of these medications are reasonable. Not only are there long-term outcome benefits for these therapies, evidence suggests early initiation of HF therapies can reduce 30-day readmissions.”
Activate the patient and develop critical health behaviors
Medical regimens for these diseases can be complex, and they must be supported by patient engagement. “Many strategies for engaging patients in care have been tested, including teaching to goal, motivational interviewing, and teach-back methods of activation and engagement. Often these methods are time intensive. Because physician time is increasingly constrained, a team approach is particularly useful.”
Set up feedback loops
“Course correction should the patient decompensate is critically important to maintaining outpatient success. Feedback loops can allow for clinical stabilization before rehospitalization is necessary.” Self-monitoring with individually set benchmarks is critical.
Arrange an early follow-up appointment prior to discharge
About half of Medicare patients with these conditions are readmitted before they’ve even had a postdischarge follow-up appointment. Ideally this should occur within 7 days. The purpose of early follow-up is to identify and address gaps in the discharge plan of care, revise the discharge plan of care to adapt to the outpatient environment, and reinforce critical health behaviors.
Consider and address other comorbidities
Comorbidities are the rule rather than the exception and contribute to many readmissions. Get primary care on the team and enlist their help in managing these issues before they lead to an exacerbation. “Meticulous control – even perfect control were it possible – of cardiopulmonary disease would still leave patients vulnerable to significant risk of readmission from other causes.”
Consider ancillary supportive services at home
Patients may be overwhelmed by the complexity of postdischarge care. Home health assistance can help in getting patients to physical therapy, continuing patient education, and providing a home clinical assessment.
Neither of the authors had any financial disclosures.
EXPERT ANALYSIS FROM CHEST JOURNAL
New guidance offered for managing poorly controlled asthma in children
published in the Annals of Allergy, Asthma & Immunology.
“Although many children with asthma achieve symptom control with appropriate management, a substantial subset does not,” Bradley E. Chipps, MD, from the Capital Allergy & Respiratory Disease Center in Sacramento, Calif., and his colleagues wrote in the recommendations sponsored by the American College of Allergy, Asthma, and Immunology. “These children should undergo a step-up in care, but when and how to do that is not always straightforward. The Pediatric Asthma Yardstick is a practical resource for starting or adjusting controller therapy based on the options that are currently available for children, from infants to 18 years of age.”
In their recommendations, the authors grouped patients into age ranges of adolescent (12-18 years), school aged (6-11 years), and young children (5 years and under) as well as severity classifications.
Adolescents and school-aged children
For adolescents and school-aged children, step 1 was classified as intermittent asthma that can be controlled with low-dose inhaled corticosteroids (ICS) with short-acting beta2-agonist (SABA) for as-needed relief. Children considered for stepping up to the next therapy should show symptoms of mild persistent asthma that the authors recommended controlling with low-dose ICS, leukotriene receptor antagonist (LTRA), or low-dose theophylline with as-needed SABA.
In children 12-18 years with moderate persistent asthma (step 3), the authors recommended a combination of low-dose ICA and a long-acting beta2-agonist (LABA), while children 6-11 years should receive a medium dose of ICS; other considerations for school-aged children include a medium-high dose of ICS, a low-dose combination of ICS and LTRA, or low-dose ICS together with theophylline.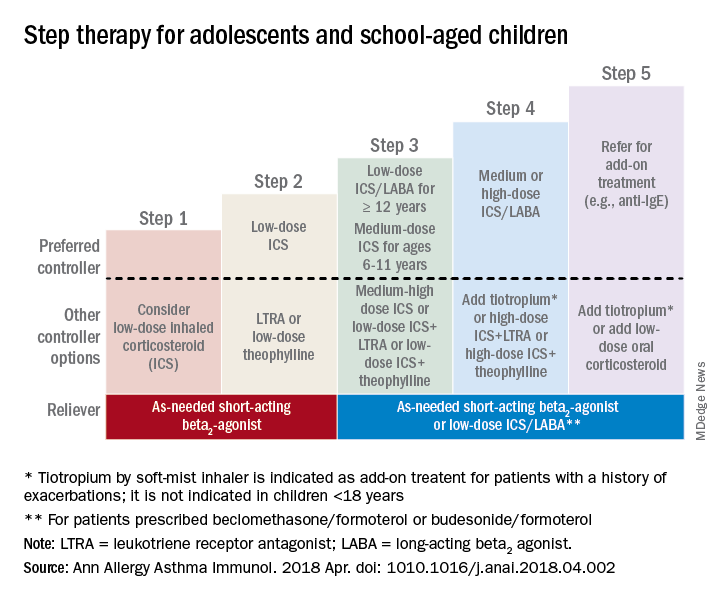
Adolescent or school-aged children with severe persistent asthma (step 4) should take a medium or high dose of ICS together with LABA, with the authors recommending adding tiotropium to a soft mist inhaler, combination high-dose ICS and LTRA, or a combination high-dose ICS and theophylline.
Dr. Chipps and his coauthors recommended children stepping up therapy beyond severe persistent asthma (step 5) should add on treatment such as low-dose oral corticosteroids, anti-immunoglobulin E therapy, and adding tiotropium to a soft-mist inhaler.
For adolescent and school-aged children going to steps 3-5, Dr. Chipps and his coauthors recommended prescribing as needed a short-acting beta2 agonist or low-dose ICS/LABA.
Children 5 years and younger
In children 5 years and younger, intermittent asthma (step 1) should be considered if the child has infrequent or viral wheezing but few or no symptoms in the interim that can be controlled with as-needed SABA. These young children who show symptoms of mild persistent asthma (step 2) can be treated with daily low-dose ICS, with other controller options of LTRA or intermittent ICS.
Stepping up therapy from mild to moderate persistent asthma (step 3), young children should receive double the daily dose of low-dose ICS from the previous step or use the low-dose ICS together with LTRA; if children show symptoms of severe persistent asthma (step 4), they should continue their daily controller and be referred to a specialist; other considerations for controllers at this step included adding LTRA, adding intermittent ICS, or increasing ICS frequency.
Other factors to consider

Inconsistencies in response to medication can occur because of comorbid conditions such as obesity, rhinosinusitis, respiratory infection or gastroesophageal reflux; suboptimal inhaled drug delivery; or failure to comply with treatment because of not wanting to take medication (common in adolescents), belief that even controller medicine can be taken intermittently, family stress, cost including lack of insurance or medication not covered by insurance. “Before adjusting therapy, it is important to ensure that the child’s change in symptoms is due to asthma and not to any of these factors that need to be addressed,” Dr. Chipps and his colleagues wrote.
Collaboration among children, their parents, and clinicians is needed to achieve good asthma control because of the “variable presentation within individuals and within the population of children affected” with asthma, they wrote.
The article summarizing the guidelines was sponsored by the American College of Allergy, Asthma, and Immunology. Most of the authors report various financial relationships with companies including AstraZeneca, Aerocrine, Aviragen, Boehringer Ingelheim, Cephalon, Circassia, Commense, Genentech, GlaxoSmithKline, Greer, Meda, Merck, Mylan, Novartis, Patara, Regeneron, Sanofi, TEVA, Theravance, and Vectura Group. Dr. Farrar and Dr. Szefler had no financial interests to disclose.
SOURCE: Chipps BE et al. Ann Allergy Asthma Immunol. 2018 Apr. doi: 1010.1016/j.anai.2018.04.002.
published in the Annals of Allergy, Asthma & Immunology.
“Although many children with asthma achieve symptom control with appropriate management, a substantial subset does not,” Bradley E. Chipps, MD, from the Capital Allergy & Respiratory Disease Center in Sacramento, Calif., and his colleagues wrote in the recommendations sponsored by the American College of Allergy, Asthma, and Immunology. “These children should undergo a step-up in care, but when and how to do that is not always straightforward. The Pediatric Asthma Yardstick is a practical resource for starting or adjusting controller therapy based on the options that are currently available for children, from infants to 18 years of age.”
In their recommendations, the authors grouped patients into age ranges of adolescent (12-18 years), school aged (6-11 years), and young children (5 years and under) as well as severity classifications.
Adolescents and school-aged children
For adolescents and school-aged children, step 1 was classified as intermittent asthma that can be controlled with low-dose inhaled corticosteroids (ICS) with short-acting beta2-agonist (SABA) for as-needed relief. Children considered for stepping up to the next therapy should show symptoms of mild persistent asthma that the authors recommended controlling with low-dose ICS, leukotriene receptor antagonist (LTRA), or low-dose theophylline with as-needed SABA.
In children 12-18 years with moderate persistent asthma (step 3), the authors recommended a combination of low-dose ICA and a long-acting beta2-agonist (LABA), while children 6-11 years should receive a medium dose of ICS; other considerations for school-aged children include a medium-high dose of ICS, a low-dose combination of ICS and LTRA, or low-dose ICS together with theophylline.
Adolescent or school-aged children with severe persistent asthma (step 4) should take a medium or high dose of ICS together with LABA, with the authors recommending adding tiotropium to a soft mist inhaler, combination high-dose ICS and LTRA, or a combination high-dose ICS and theophylline.
Dr. Chipps and his coauthors recommended children stepping up therapy beyond severe persistent asthma (step 5) should add on treatment such as low-dose oral corticosteroids, anti-immunoglobulin E therapy, and adding tiotropium to a soft-mist inhaler.
For adolescent and school-aged children going to steps 3-5, Dr. Chipps and his coauthors recommended prescribing as needed a short-acting beta2 agonist or low-dose ICS/LABA.
Children 5 years and younger
In children 5 years and younger, intermittent asthma (step 1) should be considered if the child has infrequent or viral wheezing but few or no symptoms in the interim that can be controlled with as-needed SABA. These young children who show symptoms of mild persistent asthma (step 2) can be treated with daily low-dose ICS, with other controller options of LTRA or intermittent ICS.
Stepping up therapy from mild to moderate persistent asthma (step 3), young children should receive double the daily dose of low-dose ICS from the previous step or use the low-dose ICS together with LTRA; if children show symptoms of severe persistent asthma (step 4), they should continue their daily controller and be referred to a specialist; other considerations for controllers at this step included adding LTRA, adding intermittent ICS, or increasing ICS frequency.
Other factors to consider

Inconsistencies in response to medication can occur because of comorbid conditions such as obesity, rhinosinusitis, respiratory infection or gastroesophageal reflux; suboptimal inhaled drug delivery; or failure to comply with treatment because of not wanting to take medication (common in adolescents), belief that even controller medicine can be taken intermittently, family stress, cost including lack of insurance or medication not covered by insurance. “Before adjusting therapy, it is important to ensure that the child’s change in symptoms is due to asthma and not to any of these factors that need to be addressed,” Dr. Chipps and his colleagues wrote.
Collaboration among children, their parents, and clinicians is needed to achieve good asthma control because of the “variable presentation within individuals and within the population of children affected” with asthma, they wrote.
The article summarizing the guidelines was sponsored by the American College of Allergy, Asthma, and Immunology. Most of the authors report various financial relationships with companies including AstraZeneca, Aerocrine, Aviragen, Boehringer Ingelheim, Cephalon, Circassia, Commense, Genentech, GlaxoSmithKline, Greer, Meda, Merck, Mylan, Novartis, Patara, Regeneron, Sanofi, TEVA, Theravance, and Vectura Group. Dr. Farrar and Dr. Szefler had no financial interests to disclose.
SOURCE: Chipps BE et al. Ann Allergy Asthma Immunol. 2018 Apr. doi: 1010.1016/j.anai.2018.04.002.
published in the Annals of Allergy, Asthma & Immunology.
“Although many children with asthma achieve symptom control with appropriate management, a substantial subset does not,” Bradley E. Chipps, MD, from the Capital Allergy & Respiratory Disease Center in Sacramento, Calif., and his colleagues wrote in the recommendations sponsored by the American College of Allergy, Asthma, and Immunology. “These children should undergo a step-up in care, but when and how to do that is not always straightforward. The Pediatric Asthma Yardstick is a practical resource for starting or adjusting controller therapy based on the options that are currently available for children, from infants to 18 years of age.”
In their recommendations, the authors grouped patients into age ranges of adolescent (12-18 years), school aged (6-11 years), and young children (5 years and under) as well as severity classifications.
Adolescents and school-aged children
For adolescents and school-aged children, step 1 was classified as intermittent asthma that can be controlled with low-dose inhaled corticosteroids (ICS) with short-acting beta2-agonist (SABA) for as-needed relief. Children considered for stepping up to the next therapy should show symptoms of mild persistent asthma that the authors recommended controlling with low-dose ICS, leukotriene receptor antagonist (LTRA), or low-dose theophylline with as-needed SABA.
In children 12-18 years with moderate persistent asthma (step 3), the authors recommended a combination of low-dose ICA and a long-acting beta2-agonist (LABA), while children 6-11 years should receive a medium dose of ICS; other considerations for school-aged children include a medium-high dose of ICS, a low-dose combination of ICS and LTRA, or low-dose ICS together with theophylline.
Adolescent or school-aged children with severe persistent asthma (step 4) should take a medium or high dose of ICS together with LABA, with the authors recommending adding tiotropium to a soft mist inhaler, combination high-dose ICS and LTRA, or a combination high-dose ICS and theophylline.
Dr. Chipps and his coauthors recommended children stepping up therapy beyond severe persistent asthma (step 5) should add on treatment such as low-dose oral corticosteroids, anti-immunoglobulin E therapy, and adding tiotropium to a soft-mist inhaler.
For adolescent and school-aged children going to steps 3-5, Dr. Chipps and his coauthors recommended prescribing as needed a short-acting beta2 agonist or low-dose ICS/LABA.
Children 5 years and younger
In children 5 years and younger, intermittent asthma (step 1) should be considered if the child has infrequent or viral wheezing but few or no symptoms in the interim that can be controlled with as-needed SABA. These young children who show symptoms of mild persistent asthma (step 2) can be treated with daily low-dose ICS, with other controller options of LTRA or intermittent ICS.
Stepping up therapy from mild to moderate persistent asthma (step 3), young children should receive double the daily dose of low-dose ICS from the previous step or use the low-dose ICS together with LTRA; if children show symptoms of severe persistent asthma (step 4), they should continue their daily controller and be referred to a specialist; other considerations for controllers at this step included adding LTRA, adding intermittent ICS, or increasing ICS frequency.
Other factors to consider

Inconsistencies in response to medication can occur because of comorbid conditions such as obesity, rhinosinusitis, respiratory infection or gastroesophageal reflux; suboptimal inhaled drug delivery; or failure to comply with treatment because of not wanting to take medication (common in adolescents), belief that even controller medicine can be taken intermittently, family stress, cost including lack of insurance or medication not covered by insurance. “Before adjusting therapy, it is important to ensure that the child’s change in symptoms is due to asthma and not to any of these factors that need to be addressed,” Dr. Chipps and his colleagues wrote.
Collaboration among children, their parents, and clinicians is needed to achieve good asthma control because of the “variable presentation within individuals and within the population of children affected” with asthma, they wrote.
The article summarizing the guidelines was sponsored by the American College of Allergy, Asthma, and Immunology. Most of the authors report various financial relationships with companies including AstraZeneca, Aerocrine, Aviragen, Boehringer Ingelheim, Cephalon, Circassia, Commense, Genentech, GlaxoSmithKline, Greer, Meda, Merck, Mylan, Novartis, Patara, Regeneron, Sanofi, TEVA, Theravance, and Vectura Group. Dr. Farrar and Dr. Szefler had no financial interests to disclose.
SOURCE: Chipps BE et al. Ann Allergy Asthma Immunol. 2018 Apr. doi: 1010.1016/j.anai.2018.04.002.
FROM ANNALS OF ALLERGY, ASTHMA & IMMUNOLOGY
Respiratory illness is the most common pediatric emergency in ambulatory settings
followed by psychiatric and behavioral illness, seizures, and syncope, according to results published July 20 in Pediatrics.
Investigators conducted a retrospective observational study of data from the Indianapolis emergency medical services (EMS) system between Jan. 1, 2012, and Dec. 31, 2014. All patients younger than 18 years were eligible.
Of 38,841 pediatric EMS transports in the Indianapolis metropolitan area during the 3-year period, fewer than 1% (322) were verified as originating from an ambulatory practice, reported Matthew L. Yuknis, MD, and his coauthors at Indiana University, Indianapolis. Respiratory distress was the most common emergency (58%), followed by psychiatric and behavioral illness (6%), seizure (6%), and syncope (5%).
The most common interventions were use of supplemental oxygen (27%), albuterol (26%), and intravascular access (11%). The most common critical care interventions were administration of fluid bolus (2%), benzodiazepine (2%), or racemic or intramuscular epinephrine (1%). None required use of an artificial airway, cardiopulmonary resuscitation, intraosseous access, or bag mask ventilation, Dr. Yuknis and his colleagues said.
The average time from call to on-scene arrival was 6 minutes (ranging from less than 1 to 15 minutes). The average patient transport time was 13 minutes (ranging from less than 1 to 38 minutes). The average annual frequency of pediatric outpatient emergencies was 42 emergencies per 100,000 people under 18 years of age. Lower socioeconomic status was correlated with increased frequency of emergencies in ambulatory settings, the authors reported.
“These findings update and clarify existing literature with regard to the frequency of pediatric emergencies in the ambulatory setting, the conditions these patients present with, and the use of EMS data to define these events,” the authors wrote. Additionally, the findings can be used to “inform future decisions regarding necessary equipment and procedures.”
No relevant financial disclosures were reported. There was no external funding.
SOURCE: Yuknis M et al. Pediatrics. 2018. doi: 10.1542/peds.2017-3082.
followed by psychiatric and behavioral illness, seizures, and syncope, according to results published July 20 in Pediatrics.
Investigators conducted a retrospective observational study of data from the Indianapolis emergency medical services (EMS) system between Jan. 1, 2012, and Dec. 31, 2014. All patients younger than 18 years were eligible.
Of 38,841 pediatric EMS transports in the Indianapolis metropolitan area during the 3-year period, fewer than 1% (322) were verified as originating from an ambulatory practice, reported Matthew L. Yuknis, MD, and his coauthors at Indiana University, Indianapolis. Respiratory distress was the most common emergency (58%), followed by psychiatric and behavioral illness (6%), seizure (6%), and syncope (5%).
The most common interventions were use of supplemental oxygen (27%), albuterol (26%), and intravascular access (11%). The most common critical care interventions were administration of fluid bolus (2%), benzodiazepine (2%), or racemic or intramuscular epinephrine (1%). None required use of an artificial airway, cardiopulmonary resuscitation, intraosseous access, or bag mask ventilation, Dr. Yuknis and his colleagues said.
The average time from call to on-scene arrival was 6 minutes (ranging from less than 1 to 15 minutes). The average patient transport time was 13 minutes (ranging from less than 1 to 38 minutes). The average annual frequency of pediatric outpatient emergencies was 42 emergencies per 100,000 people under 18 years of age. Lower socioeconomic status was correlated with increased frequency of emergencies in ambulatory settings, the authors reported.
“These findings update and clarify existing literature with regard to the frequency of pediatric emergencies in the ambulatory setting, the conditions these patients present with, and the use of EMS data to define these events,” the authors wrote. Additionally, the findings can be used to “inform future decisions regarding necessary equipment and procedures.”
No relevant financial disclosures were reported. There was no external funding.
SOURCE: Yuknis M et al. Pediatrics. 2018. doi: 10.1542/peds.2017-3082.
followed by psychiatric and behavioral illness, seizures, and syncope, according to results published July 20 in Pediatrics.
Investigators conducted a retrospective observational study of data from the Indianapolis emergency medical services (EMS) system between Jan. 1, 2012, and Dec. 31, 2014. All patients younger than 18 years were eligible.
Of 38,841 pediatric EMS transports in the Indianapolis metropolitan area during the 3-year period, fewer than 1% (322) were verified as originating from an ambulatory practice, reported Matthew L. Yuknis, MD, and his coauthors at Indiana University, Indianapolis. Respiratory distress was the most common emergency (58%), followed by psychiatric and behavioral illness (6%), seizure (6%), and syncope (5%).
The most common interventions were use of supplemental oxygen (27%), albuterol (26%), and intravascular access (11%). The most common critical care interventions were administration of fluid bolus (2%), benzodiazepine (2%), or racemic or intramuscular epinephrine (1%). None required use of an artificial airway, cardiopulmonary resuscitation, intraosseous access, or bag mask ventilation, Dr. Yuknis and his colleagues said.
The average time from call to on-scene arrival was 6 minutes (ranging from less than 1 to 15 minutes). The average patient transport time was 13 minutes (ranging from less than 1 to 38 minutes). The average annual frequency of pediatric outpatient emergencies was 42 emergencies per 100,000 people under 18 years of age. Lower socioeconomic status was correlated with increased frequency of emergencies in ambulatory settings, the authors reported.
“These findings update and clarify existing literature with regard to the frequency of pediatric emergencies in the ambulatory setting, the conditions these patients present with, and the use of EMS data to define these events,” the authors wrote. Additionally, the findings can be used to “inform future decisions regarding necessary equipment and procedures.”
No relevant financial disclosures were reported. There was no external funding.
SOURCE: Yuknis M et al. Pediatrics. 2018. doi: 10.1542/peds.2017-3082.
FROM PEDIATRICS
Key clinical point: Respiratory illness was the most common pediatric emergency in ambulatory settings.
Major finding: Among pediatric emergency medical services from ambulatory settings, 58% were caused by respiratory illness.
Study details: A retrospective observational study of 38,841 EMS transports in the Indianapolis metropolitan area over 3 years.
Disclosures: No relevant financial disclosures were reported. There was no external funding.
Source: Yuknis M et al. Pediatrics. 2018. doi: 10.1542/peds.2017-3082.
Later diagnosis predicts poorer outcomes in adult-diagnosed cystic fibrosis
Older age at diagnosis, diabetes, and poorer lung function are all , new research suggests.
A growing number of people with cystic fibrosis are diagnosed in adulthood, partly because of increased awareness among physicians of variations in disease presentation, more accessible genotyping, and easier diagnostic criteria.
Adult-diagnosed cystic fibrosis patients generally have a milder form of the disease than that of those diagnosed in childhood; however, less is known about their prognosis and life expectancy.
Researchers reported the outcomes of a retrospective cohort study of 362 adults diagnosed with cystic fibrosis at age 18 years or older. The median age at diagnosis was 34.3 years, and 71% of patients presented with pulmonary and/or gastrointestinal symptoms. The study was published in Annals of the American Thoracic Society.
The patients were followed for a median of 7.7 years, during which time there were 15 lung transplants and 33 deaths without transplant. Overall, 10-year lung transplant–free survival was 87.7%, and 15-year survival was 86.1%.
Those who were diagnosed young and who had higher lung function had the best median survival times. For each 5-year increase in age at diagnosis, the risk of death or transplant increased by 24%, and for each 5% decrease in forced expiratory volume in one second (FEV1), the risk was 35% higher.
Individuals who had diabetes at baseline had a more than fourfold higher risk of death or transplant than did those without diabetes.
“While newborn screening programs will reduce the rate of missed diagnoses in the future, clinicians still need to consider CF as a possible diagnosis if individuals are presenting with suspicious CF symptoms (e.g. GI or pulmonary symptoms) during adulthood, particularly if born prior to the introduction of newborn screening in their jurisdiction,” wrote Sameer Desai, of the University of British Columbia, Vancouver, and his coauthors.
Commenting on the association with diabetes, the authors noted that this finding had some uncertainty but suggested the additional inflammatory burden could increase the risk of death in individuals with cystic fibrosis.
The authors highlighted that fewer than 5% of people with adult-diagnosed cystic fibrosis had two copies of the F508del mutation, which is associated with severe, early-onset disease. However, those who were homozygous for that mutation tended to be diagnosed at a younger adult age, had worse nutritional status and a lower FEV1 percent predicted, compared with the overall adult-diagnosed population.
“This finding suggests potential delays in CF diagnosis for these people leading to worse outcomes,” the authors wrote.
The researchers also identified 25 individuals who had a possible unconfirmed diagnosis based on the most recent cystic fibrosis diagnostic guidelines. These individuals were either asymptomatic or had unknown symptoms, had sweat chlorides at or below 60 mmol/L (where available), and either unknown or two non–cystic fibrosis–causing mutations. They were also more likely to be male, to be nonwhite, to have increased unknown mutations, and to be pancreatic sufficient, compared with individuals with a confirmed diagnosis.
The study looked at whether Pseudomonas aeruginosa and Burkholderia cepacia complex increased the risk of transplant or death, but found these did not significantly predict survival.
“Adult CF clinicians can use this information to educate newly diagnosed adults with CF about their prognosis and to guide treatment decisions, specifically those at high-risk for a worse prognosis,” the authors wrote.
The study was partly funded by the Rare Disease Foundation. Two authors declared support from Cystic Fibrosis Canada, but no other conflicts of interest were declared.
SOURCE: Desai A et al. Ann Am Thorac Soc. 2018 Jun 26. doi: 10.1513/AnnalsATS.201801-037OC.
Older age at diagnosis, diabetes, and poorer lung function are all , new research suggests.
A growing number of people with cystic fibrosis are diagnosed in adulthood, partly because of increased awareness among physicians of variations in disease presentation, more accessible genotyping, and easier diagnostic criteria.
Adult-diagnosed cystic fibrosis patients generally have a milder form of the disease than that of those diagnosed in childhood; however, less is known about their prognosis and life expectancy.
Researchers reported the outcomes of a retrospective cohort study of 362 adults diagnosed with cystic fibrosis at age 18 years or older. The median age at diagnosis was 34.3 years, and 71% of patients presented with pulmonary and/or gastrointestinal symptoms. The study was published in Annals of the American Thoracic Society.
The patients were followed for a median of 7.7 years, during which time there were 15 lung transplants and 33 deaths without transplant. Overall, 10-year lung transplant–free survival was 87.7%, and 15-year survival was 86.1%.
Those who were diagnosed young and who had higher lung function had the best median survival times. For each 5-year increase in age at diagnosis, the risk of death or transplant increased by 24%, and for each 5% decrease in forced expiratory volume in one second (FEV1), the risk was 35% higher.
Individuals who had diabetes at baseline had a more than fourfold higher risk of death or transplant than did those without diabetes.
“While newborn screening programs will reduce the rate of missed diagnoses in the future, clinicians still need to consider CF as a possible diagnosis if individuals are presenting with suspicious CF symptoms (e.g. GI or pulmonary symptoms) during adulthood, particularly if born prior to the introduction of newborn screening in their jurisdiction,” wrote Sameer Desai, of the University of British Columbia, Vancouver, and his coauthors.
Commenting on the association with diabetes, the authors noted that this finding had some uncertainty but suggested the additional inflammatory burden could increase the risk of death in individuals with cystic fibrosis.
The authors highlighted that fewer than 5% of people with adult-diagnosed cystic fibrosis had two copies of the F508del mutation, which is associated with severe, early-onset disease. However, those who were homozygous for that mutation tended to be diagnosed at a younger adult age, had worse nutritional status and a lower FEV1 percent predicted, compared with the overall adult-diagnosed population.
“This finding suggests potential delays in CF diagnosis for these people leading to worse outcomes,” the authors wrote.
The researchers also identified 25 individuals who had a possible unconfirmed diagnosis based on the most recent cystic fibrosis diagnostic guidelines. These individuals were either asymptomatic or had unknown symptoms, had sweat chlorides at or below 60 mmol/L (where available), and either unknown or two non–cystic fibrosis–causing mutations. They were also more likely to be male, to be nonwhite, to have increased unknown mutations, and to be pancreatic sufficient, compared with individuals with a confirmed diagnosis.
The study looked at whether Pseudomonas aeruginosa and Burkholderia cepacia complex increased the risk of transplant or death, but found these did not significantly predict survival.
“Adult CF clinicians can use this information to educate newly diagnosed adults with CF about their prognosis and to guide treatment decisions, specifically those at high-risk for a worse prognosis,” the authors wrote.
The study was partly funded by the Rare Disease Foundation. Two authors declared support from Cystic Fibrosis Canada, but no other conflicts of interest were declared.
SOURCE: Desai A et al. Ann Am Thorac Soc. 2018 Jun 26. doi: 10.1513/AnnalsATS.201801-037OC.
Older age at diagnosis, diabetes, and poorer lung function are all , new research suggests.
A growing number of people with cystic fibrosis are diagnosed in adulthood, partly because of increased awareness among physicians of variations in disease presentation, more accessible genotyping, and easier diagnostic criteria.
Adult-diagnosed cystic fibrosis patients generally have a milder form of the disease than that of those diagnosed in childhood; however, less is known about their prognosis and life expectancy.
Researchers reported the outcomes of a retrospective cohort study of 362 adults diagnosed with cystic fibrosis at age 18 years or older. The median age at diagnosis was 34.3 years, and 71% of patients presented with pulmonary and/or gastrointestinal symptoms. The study was published in Annals of the American Thoracic Society.
The patients were followed for a median of 7.7 years, during which time there were 15 lung transplants and 33 deaths without transplant. Overall, 10-year lung transplant–free survival was 87.7%, and 15-year survival was 86.1%.
Those who were diagnosed young and who had higher lung function had the best median survival times. For each 5-year increase in age at diagnosis, the risk of death or transplant increased by 24%, and for each 5% decrease in forced expiratory volume in one second (FEV1), the risk was 35% higher.
Individuals who had diabetes at baseline had a more than fourfold higher risk of death or transplant than did those without diabetes.
“While newborn screening programs will reduce the rate of missed diagnoses in the future, clinicians still need to consider CF as a possible diagnosis if individuals are presenting with suspicious CF symptoms (e.g. GI or pulmonary symptoms) during adulthood, particularly if born prior to the introduction of newborn screening in their jurisdiction,” wrote Sameer Desai, of the University of British Columbia, Vancouver, and his coauthors.
Commenting on the association with diabetes, the authors noted that this finding had some uncertainty but suggested the additional inflammatory burden could increase the risk of death in individuals with cystic fibrosis.
The authors highlighted that fewer than 5% of people with adult-diagnosed cystic fibrosis had two copies of the F508del mutation, which is associated with severe, early-onset disease. However, those who were homozygous for that mutation tended to be diagnosed at a younger adult age, had worse nutritional status and a lower FEV1 percent predicted, compared with the overall adult-diagnosed population.
“This finding suggests potential delays in CF diagnosis for these people leading to worse outcomes,” the authors wrote.
The researchers also identified 25 individuals who had a possible unconfirmed diagnosis based on the most recent cystic fibrosis diagnostic guidelines. These individuals were either asymptomatic or had unknown symptoms, had sweat chlorides at or below 60 mmol/L (where available), and either unknown or two non–cystic fibrosis–causing mutations. They were also more likely to be male, to be nonwhite, to have increased unknown mutations, and to be pancreatic sufficient, compared with individuals with a confirmed diagnosis.
The study looked at whether Pseudomonas aeruginosa and Burkholderia cepacia complex increased the risk of transplant or death, but found these did not significantly predict survival.
“Adult CF clinicians can use this information to educate newly diagnosed adults with CF about their prognosis and to guide treatment decisions, specifically those at high-risk for a worse prognosis,” the authors wrote.
The study was partly funded by the Rare Disease Foundation. Two authors declared support from Cystic Fibrosis Canada, but no other conflicts of interest were declared.
SOURCE: Desai A et al. Ann Am Thorac Soc. 2018 Jun 26. doi: 10.1513/AnnalsATS.201801-037OC.
FROM ANNALS OF THE AMERICAN THORACIC SOCIETY
Key clinical point: Older age at diagnosis is a risk factor for death or transplant in adult-diagnosed cystic fibrosis.
Major finding: The risk of death or transplant increases by 24% per 5-year increase in age at diagnosis in adult-diagnosed cystic fibrosis.
Study details: A retrospective cohort study of 362 adults diagnosed with cystic fibrosis.
Disclosures: The study was partly funded by The Rare Disease Foundation. Two authors declared support from Cystic Fibrosis Canada, but no other conflicts of interest were declared.
Source: Desai A et al. Ann Am Thorac Soc. 2018 Jun 26. doi: 10.1513/AnnalsATS.201801-037OC.