User login
Nicotinamide Prevents Actinic Keratoses, Basal Cell Carcinomas, and Squamous Cell Carcinomas

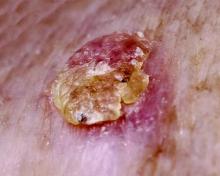
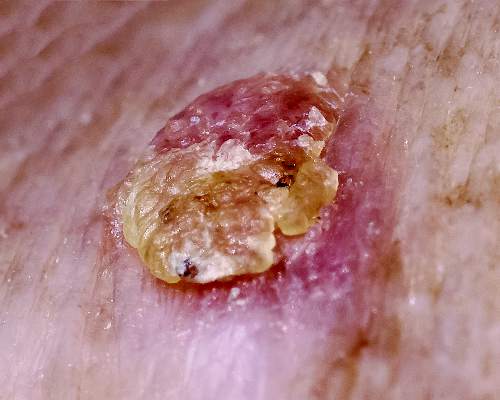
Chen et al (N Engl J Med. 2015;373:1618-1626) performed a multicenter, phase 3, double-blind, randomized, placebo-controlled trial. Results demonstrated that nicotinamide effectively decreased the rates of new nonmelanoma skin cancers (NMSCs) and actinic keratoses (AKs) in high-risk patients who had at least 2 histologically confirmed NMSCs in the last 5 years. In comparison to participants who received placebo, individuals who received nicotinamide 500 mg twice daily during the 12-month study (branded with a predictive acronym ONTRAC [oral nicotinamide to reduce actinic cancer]) had reduced rates of AKs of up to 20%, basal cell carcinomas of 20%, squamous cell carcinomas of 30%, and NMSCs of 23%. However, the effect of nicotinamide on NMSCs was not maintained at evaluation 6 months after discontinuation; the number of basal cell carcinomas was similar, and the number of squamous cell carcinomas was greater in participants who received nicotinamide in comparison to individuals who received placebo.
What’s the issue?
The risk for skin cancer is increased by UV radiation that damages DNA, suppresses cutaneous immunity, and inhibits DNA repair by depleting cellular adenosine triphosphate. Nicotinamide, an amide form of vitamin B3, has been demonstrated to not only reduce UV radiation–induced immunosuppression but also to prevent UV radiation–induced adenosine triphosphate depletion and glycolic blockade. Nicotinamide, which is classified as a food additive, also has neuroprotective and antioxidant functions and reduces pigmentation, wrinkles, and sebum production. Although oral nicotinamide has been demonstrated to reduce NMSCs and AKs, topical application has been shown to improve many skin conditions such as acne, atopic dermatitis, isoniazid-induced pellagra, and rosacea.
In contrast to nicotinic acid (niacin), nicotinamide is not associated with headaches, hypotension, flushing, itching, or vasodilatation. At high oral doses, side effects of nicotinamide that have been hypothesized or observed in animals, humans, or both have included the development of Parkinson disease, insulin sensitivity and diabetes mellitus, and liver toxicity. Although there are no reports in humans of growth retardation, teratogenicity, or oncogenicity, Rolfe (J Cosmet Dermatol. 2014;13:324-328) discussed that fetal blood levels of nicotinamide are greater than corresponding maternal blood levels because it is able to cross the placenta. However, according to Chen et al, no clinically significant between-group differences were found with respect to the number or types of adverse events that occurred in the placebo participants and the individuals who received 1000 mg daily of nicotinamide. Chen et al implied that there were additional benefits in the recipients of nicotinamide with regards to cognitive function and transepidermal water loss.
Perhaps all patients with a history of AKs, basal cell carcinomas, or squamous cell carcinomas should receive lifelong nicotinamide. Also, it might be reasonable to consider that all individuals older than 18 years who are not pregnant or breastfeeding with increased sun exposure but no history of AKs or NMSC add nicotinamide to their daily diets as a proactive measure for chemoprevention. Would you suggest nicotinamide to your patients?

Chen et al (N Engl J Med. 2015;373:1618-1626) performed a multicenter, phase 3, double-blind, randomized, placebo-controlled trial. Results demonstrated that nicotinamide effectively decreased the rates of new nonmelanoma skin cancers (NMSCs) and actinic keratoses (AKs) in high-risk patients who had at least 2 histologically confirmed NMSCs in the last 5 years. In comparison to participants who received placebo, individuals who received nicotinamide 500 mg twice daily during the 12-month study (branded with a predictive acronym ONTRAC [oral nicotinamide to reduce actinic cancer]) had reduced rates of AKs of up to 20%, basal cell carcinomas of 20%, squamous cell carcinomas of 30%, and NMSCs of 23%. However, the effect of nicotinamide on NMSCs was not maintained at evaluation 6 months after discontinuation; the number of basal cell carcinomas was similar, and the number of squamous cell carcinomas was greater in participants who received nicotinamide in comparison to individuals who received placebo.
What’s the issue?
The risk for skin cancer is increased by UV radiation that damages DNA, suppresses cutaneous immunity, and inhibits DNA repair by depleting cellular adenosine triphosphate. Nicotinamide, an amide form of vitamin B3, has been demonstrated to not only reduce UV radiation–induced immunosuppression but also to prevent UV radiation–induced adenosine triphosphate depletion and glycolic blockade. Nicotinamide, which is classified as a food additive, also has neuroprotective and antioxidant functions and reduces pigmentation, wrinkles, and sebum production. Although oral nicotinamide has been demonstrated to reduce NMSCs and AKs, topical application has been shown to improve many skin conditions such as acne, atopic dermatitis, isoniazid-induced pellagra, and rosacea.
In contrast to nicotinic acid (niacin), nicotinamide is not associated with headaches, hypotension, flushing, itching, or vasodilatation. At high oral doses, side effects of nicotinamide that have been hypothesized or observed in animals, humans, or both have included the development of Parkinson disease, insulin sensitivity and diabetes mellitus, and liver toxicity. Although there are no reports in humans of growth retardation, teratogenicity, or oncogenicity, Rolfe (J Cosmet Dermatol. 2014;13:324-328) discussed that fetal blood levels of nicotinamide are greater than corresponding maternal blood levels because it is able to cross the placenta. However, according to Chen et al, no clinically significant between-group differences were found with respect to the number or types of adverse events that occurred in the placebo participants and the individuals who received 1000 mg daily of nicotinamide. Chen et al implied that there were additional benefits in the recipients of nicotinamide with regards to cognitive function and transepidermal water loss.
Perhaps all patients with a history of AKs, basal cell carcinomas, or squamous cell carcinomas should receive lifelong nicotinamide. Also, it might be reasonable to consider that all individuals older than 18 years who are not pregnant or breastfeeding with increased sun exposure but no history of AKs or NMSC add nicotinamide to their daily diets as a proactive measure for chemoprevention. Would you suggest nicotinamide to your patients?

Chen et al (N Engl J Med. 2015;373:1618-1626) performed a multicenter, phase 3, double-blind, randomized, placebo-controlled trial. Results demonstrated that nicotinamide effectively decreased the rates of new nonmelanoma skin cancers (NMSCs) and actinic keratoses (AKs) in high-risk patients who had at least 2 histologically confirmed NMSCs in the last 5 years. In comparison to participants who received placebo, individuals who received nicotinamide 500 mg twice daily during the 12-month study (branded with a predictive acronym ONTRAC [oral nicotinamide to reduce actinic cancer]) had reduced rates of AKs of up to 20%, basal cell carcinomas of 20%, squamous cell carcinomas of 30%, and NMSCs of 23%. However, the effect of nicotinamide on NMSCs was not maintained at evaluation 6 months after discontinuation; the number of basal cell carcinomas was similar, and the number of squamous cell carcinomas was greater in participants who received nicotinamide in comparison to individuals who received placebo.
What’s the issue?
The risk for skin cancer is increased by UV radiation that damages DNA, suppresses cutaneous immunity, and inhibits DNA repair by depleting cellular adenosine triphosphate. Nicotinamide, an amide form of vitamin B3, has been demonstrated to not only reduce UV radiation–induced immunosuppression but also to prevent UV radiation–induced adenosine triphosphate depletion and glycolic blockade. Nicotinamide, which is classified as a food additive, also has neuroprotective and antioxidant functions and reduces pigmentation, wrinkles, and sebum production. Although oral nicotinamide has been demonstrated to reduce NMSCs and AKs, topical application has been shown to improve many skin conditions such as acne, atopic dermatitis, isoniazid-induced pellagra, and rosacea.
In contrast to nicotinic acid (niacin), nicotinamide is not associated with headaches, hypotension, flushing, itching, or vasodilatation. At high oral doses, side effects of nicotinamide that have been hypothesized or observed in animals, humans, or both have included the development of Parkinson disease, insulin sensitivity and diabetes mellitus, and liver toxicity. Although there are no reports in humans of growth retardation, teratogenicity, or oncogenicity, Rolfe (J Cosmet Dermatol. 2014;13:324-328) discussed that fetal blood levels of nicotinamide are greater than corresponding maternal blood levels because it is able to cross the placenta. However, according to Chen et al, no clinically significant between-group differences were found with respect to the number or types of adverse events that occurred in the placebo participants and the individuals who received 1000 mg daily of nicotinamide. Chen et al implied that there were additional benefits in the recipients of nicotinamide with regards to cognitive function and transepidermal water loss.
Perhaps all patients with a history of AKs, basal cell carcinomas, or squamous cell carcinomas should receive lifelong nicotinamide. Also, it might be reasonable to consider that all individuals older than 18 years who are not pregnant or breastfeeding with increased sun exposure but no history of AKs or NMSC add nicotinamide to their daily diets as a proactive measure for chemoprevention. Would you suggest nicotinamide to your patients?
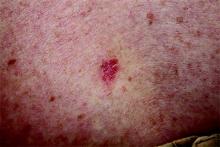
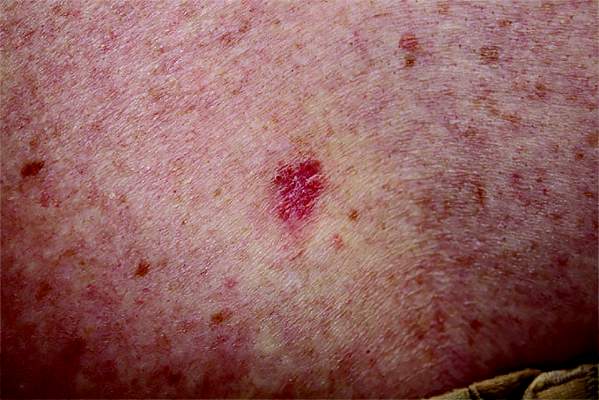
Poor Sun Protection Practices in Hispanic Patients Increases Risk for Skin Cancer
The most common type of skin cancer in Hispanic patients is basal cell carcinoma, according to Maritza Perez, MD, Associate Clinical Professor of Dermatology at the Icahn School of Medicine at Mount Sinai, New York, New York. Dr. Perez reviewed skin cancer in Hispanics as part of the “Skin Issues in Latino Patients” forum at the 74th Annual Meeting of the American Academy of Dermatology (AAD)(March 4-8, 2016) in Washington, DC. The head and neck region is the most common location of BCC in Hispanics, and it is more common among Hispanic males. Squamous cell carcinomas are the second most common, and melanomas are third most common.
The mortality rate from squamous cell carcinoma is higher in Hispanic patients compared to white patients. Dr. Perez indicated that UV light exposure is a predisposing factor for these types of skin cancer, and poor sun protection practices in this population are to blame.
“Patients [in this population] do not perceive that they are prone to skin cancer,” said Dr. Perez. This erroneous perception has led to delayed diagnosis and treatment of patients in this group, which has caused increased mortality and decreased survival.
Marta Rendon, MD, director of the session at the AAD and Medical Director of The Rendon Center for Dermatology & Aesthetic Medicine, Boca Raton, Florida, noted that Latinos are the fastest growing minority population in the United States and it is estimated that they will be approximately 30% of the population by the year 2050. As a result, dermatologists must emphasize sun protection, avoidance of midday sun, and avoidance of UV radiation.
In his Cutis practical pearls, “Patient Compliance With Photoprotection,” Vincent A. DeLeo, MD, provided tips on sunscreen use. “Patients need a realistic approach to photoprotection based on their genetics, including Fitzpatrick skin type and family history . . . and lifestyle history, which should include location of residence as well as occupation and recreational pursuits,” he said. “These inquiries should lead to a frank discussion of the patient’s risk for developing photodamage and skin cancer.”
Encourage routine use of a sunscreen with a sun protection factor of 30 or higher that carries a “broad spectrum” label. “There is evidence that sunscreens prevent squamous cell carcinoma, actinic keratosis, and photoaging. Early evidence, less strong but positive, also suggests protection against basal cell carcinoma and melanoma,” Dr. DeLeo reported.
The most common type of skin cancer in Hispanic patients is basal cell carcinoma, according to Maritza Perez, MD, Associate Clinical Professor of Dermatology at the Icahn School of Medicine at Mount Sinai, New York, New York. Dr. Perez reviewed skin cancer in Hispanics as part of the “Skin Issues in Latino Patients” forum at the 74th Annual Meeting of the American Academy of Dermatology (AAD)(March 4-8, 2016) in Washington, DC. The head and neck region is the most common location of BCC in Hispanics, and it is more common among Hispanic males. Squamous cell carcinomas are the second most common, and melanomas are third most common.
The mortality rate from squamous cell carcinoma is higher in Hispanic patients compared to white patients. Dr. Perez indicated that UV light exposure is a predisposing factor for these types of skin cancer, and poor sun protection practices in this population are to blame.
“Patients [in this population] do not perceive that they are prone to skin cancer,” said Dr. Perez. This erroneous perception has led to delayed diagnosis and treatment of patients in this group, which has caused increased mortality and decreased survival.
Marta Rendon, MD, director of the session at the AAD and Medical Director of The Rendon Center for Dermatology & Aesthetic Medicine, Boca Raton, Florida, noted that Latinos are the fastest growing minority population in the United States and it is estimated that they will be approximately 30% of the population by the year 2050. As a result, dermatologists must emphasize sun protection, avoidance of midday sun, and avoidance of UV radiation.
In his Cutis practical pearls, “Patient Compliance With Photoprotection,” Vincent A. DeLeo, MD, provided tips on sunscreen use. “Patients need a realistic approach to photoprotection based on their genetics, including Fitzpatrick skin type and family history . . . and lifestyle history, which should include location of residence as well as occupation and recreational pursuits,” he said. “These inquiries should lead to a frank discussion of the patient’s risk for developing photodamage and skin cancer.”
Encourage routine use of a sunscreen with a sun protection factor of 30 or higher that carries a “broad spectrum” label. “There is evidence that sunscreens prevent squamous cell carcinoma, actinic keratosis, and photoaging. Early evidence, less strong but positive, also suggests protection against basal cell carcinoma and melanoma,” Dr. DeLeo reported.
The most common type of skin cancer in Hispanic patients is basal cell carcinoma, according to Maritza Perez, MD, Associate Clinical Professor of Dermatology at the Icahn School of Medicine at Mount Sinai, New York, New York. Dr. Perez reviewed skin cancer in Hispanics as part of the “Skin Issues in Latino Patients” forum at the 74th Annual Meeting of the American Academy of Dermatology (AAD)(March 4-8, 2016) in Washington, DC. The head and neck region is the most common location of BCC in Hispanics, and it is more common among Hispanic males. Squamous cell carcinomas are the second most common, and melanomas are third most common.
The mortality rate from squamous cell carcinoma is higher in Hispanic patients compared to white patients. Dr. Perez indicated that UV light exposure is a predisposing factor for these types of skin cancer, and poor sun protection practices in this population are to blame.
“Patients [in this population] do not perceive that they are prone to skin cancer,” said Dr. Perez. This erroneous perception has led to delayed diagnosis and treatment of patients in this group, which has caused increased mortality and decreased survival.
Marta Rendon, MD, director of the session at the AAD and Medical Director of The Rendon Center for Dermatology & Aesthetic Medicine, Boca Raton, Florida, noted that Latinos are the fastest growing minority population in the United States and it is estimated that they will be approximately 30% of the population by the year 2050. As a result, dermatologists must emphasize sun protection, avoidance of midday sun, and avoidance of UV radiation.
In his Cutis practical pearls, “Patient Compliance With Photoprotection,” Vincent A. DeLeo, MD, provided tips on sunscreen use. “Patients need a realistic approach to photoprotection based on their genetics, including Fitzpatrick skin type and family history . . . and lifestyle history, which should include location of residence as well as occupation and recreational pursuits,” he said. “These inquiries should lead to a frank discussion of the patient’s risk for developing photodamage and skin cancer.”
Encourage routine use of a sunscreen with a sun protection factor of 30 or higher that carries a “broad spectrum” label. “There is evidence that sunscreens prevent squamous cell carcinoma, actinic keratosis, and photoaging. Early evidence, less strong but positive, also suggests protection against basal cell carcinoma and melanoma,” Dr. DeLeo reported.
Itraconazole targets basal cell carcinoma
WAIKOLOA, HAWAII – The familiar oral triazole antifungal agent itraconazole (Sporanox) is under active investigation for an unexpected use: as adjunctive therapy in patients with locally advanced or metastatic basal cell carcinoma.
“The promise of this drug is that the use of itraconazole with vismodegib or sonidegib may actually enhance the effectiveness of those drugs and also reduce the frequency of grade 2 toxicities by perhaps allowing a lower dose of vismodegib or sonidegib,” Dr. David L. Swanson said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
“Be looking for this drug that we use to treat toenail fungus as a potential drug for locally advanced or metastatic basal cell carcinoma,” advised Dr. Swanson, a dermatologist at the Mayo Clinic in Scottsdale, Ariz.
Vismodegib (Erivedge) has been a game changer for patients with inoperable locally advanced or metastatic breast cancer. “The response to this drug was amazing,” Dr. Swanson said of the landmark study which led to its approval in 2012 (N Engl J Med. 2012; 366[23]:2171-9).
Sonidegib (Odomzo), which like vismodegib inhibits the essential Hedgehog signaling pathway component known as Smoothened, was approved by the Food and Drug Administration in 2015 as the second oral drug in this novel class.
While the clinical benefits of these two drugs in patients with the most horrific basal cell carcinomas are extremely impressive, vismodegib and sonidegib have two major drawbacks: The tumors eventually develop resistance and commence growing again, and onerous grade 2 side effects requiring dose reduction are extremely common. The most frequent of these limiting side effects are a disturbed sense of taste, muscle spasms, alopecia, and weight loss.
The hope is that itraconazole may be of help with both issues, according to Dr. Swanson. It turns out that the antifungal agent is also an inhibitor of the Hedgehog pathway, and via a different mechanism than that of vismodegib and sonidegib.
He pointed to an international open-label exploratory phase II study led by Dr. Jean Y. Tang of Stanford (Calif.) University. The investigators treated 19 patients with a total of 90 basal cell carcinomas with oral itraconazole at 200 mg twice a day for 1 month or 100 mg twice a day for an average of 2.3 months.
The treatment reduced Hedgehog signaling pathway activity by 65%, Ki67 tumor cell proliferation by 45%, and tumor area by 24% (J Clin Oncol. 2014 Mar 10;32[8]:745-51).
These results aren’t as dramatic as what’s achieved using vismodegib or sonidegib. As stand-alone therapy, itraconazole doesn’t compare with those agents. However, the hope is that when itraconazole is prescribed in conjunction with vismodegib or sonidegib it will permit the latter drugs to be used at lower doses with no drop-off in efficacy, which would mean less grade 2 toxicity. Moreover, since itraconazole inhibits Hedgehog signaling through a mechanism that is different from that of the more potent agents, combination therapy might delay onset of tumor resistance, Dr. Swanson explained.
He reported having no financial conflicts of interest regarding his presentation.
SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – The familiar oral triazole antifungal agent itraconazole (Sporanox) is under active investigation for an unexpected use: as adjunctive therapy in patients with locally advanced or metastatic basal cell carcinoma.
“The promise of this drug is that the use of itraconazole with vismodegib or sonidegib may actually enhance the effectiveness of those drugs and also reduce the frequency of grade 2 toxicities by perhaps allowing a lower dose of vismodegib or sonidegib,” Dr. David L. Swanson said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
“Be looking for this drug that we use to treat toenail fungus as a potential drug for locally advanced or metastatic basal cell carcinoma,” advised Dr. Swanson, a dermatologist at the Mayo Clinic in Scottsdale, Ariz.
Vismodegib (Erivedge) has been a game changer for patients with inoperable locally advanced or metastatic breast cancer. “The response to this drug was amazing,” Dr. Swanson said of the landmark study which led to its approval in 2012 (N Engl J Med. 2012; 366[23]:2171-9).
Sonidegib (Odomzo), which like vismodegib inhibits the essential Hedgehog signaling pathway component known as Smoothened, was approved by the Food and Drug Administration in 2015 as the second oral drug in this novel class.
While the clinical benefits of these two drugs in patients with the most horrific basal cell carcinomas are extremely impressive, vismodegib and sonidegib have two major drawbacks: The tumors eventually develop resistance and commence growing again, and onerous grade 2 side effects requiring dose reduction are extremely common. The most frequent of these limiting side effects are a disturbed sense of taste, muscle spasms, alopecia, and weight loss.
The hope is that itraconazole may be of help with both issues, according to Dr. Swanson. It turns out that the antifungal agent is also an inhibitor of the Hedgehog pathway, and via a different mechanism than that of vismodegib and sonidegib.
He pointed to an international open-label exploratory phase II study led by Dr. Jean Y. Tang of Stanford (Calif.) University. The investigators treated 19 patients with a total of 90 basal cell carcinomas with oral itraconazole at 200 mg twice a day for 1 month or 100 mg twice a day for an average of 2.3 months.
The treatment reduced Hedgehog signaling pathway activity by 65%, Ki67 tumor cell proliferation by 45%, and tumor area by 24% (J Clin Oncol. 2014 Mar 10;32[8]:745-51).
These results aren’t as dramatic as what’s achieved using vismodegib or sonidegib. As stand-alone therapy, itraconazole doesn’t compare with those agents. However, the hope is that when itraconazole is prescribed in conjunction with vismodegib or sonidegib it will permit the latter drugs to be used at lower doses with no drop-off in efficacy, which would mean less grade 2 toxicity. Moreover, since itraconazole inhibits Hedgehog signaling through a mechanism that is different from that of the more potent agents, combination therapy might delay onset of tumor resistance, Dr. Swanson explained.
He reported having no financial conflicts of interest regarding his presentation.
SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – The familiar oral triazole antifungal agent itraconazole (Sporanox) is under active investigation for an unexpected use: as adjunctive therapy in patients with locally advanced or metastatic basal cell carcinoma.
“The promise of this drug is that the use of itraconazole with vismodegib or sonidegib may actually enhance the effectiveness of those drugs and also reduce the frequency of grade 2 toxicities by perhaps allowing a lower dose of vismodegib or sonidegib,” Dr. David L. Swanson said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
“Be looking for this drug that we use to treat toenail fungus as a potential drug for locally advanced or metastatic basal cell carcinoma,” advised Dr. Swanson, a dermatologist at the Mayo Clinic in Scottsdale, Ariz.
Vismodegib (Erivedge) has been a game changer for patients with inoperable locally advanced or metastatic breast cancer. “The response to this drug was amazing,” Dr. Swanson said of the landmark study which led to its approval in 2012 (N Engl J Med. 2012; 366[23]:2171-9).
Sonidegib (Odomzo), which like vismodegib inhibits the essential Hedgehog signaling pathway component known as Smoothened, was approved by the Food and Drug Administration in 2015 as the second oral drug in this novel class.
While the clinical benefits of these two drugs in patients with the most horrific basal cell carcinomas are extremely impressive, vismodegib and sonidegib have two major drawbacks: The tumors eventually develop resistance and commence growing again, and onerous grade 2 side effects requiring dose reduction are extremely common. The most frequent of these limiting side effects are a disturbed sense of taste, muscle spasms, alopecia, and weight loss.
The hope is that itraconazole may be of help with both issues, according to Dr. Swanson. It turns out that the antifungal agent is also an inhibitor of the Hedgehog pathway, and via a different mechanism than that of vismodegib and sonidegib.
He pointed to an international open-label exploratory phase II study led by Dr. Jean Y. Tang of Stanford (Calif.) University. The investigators treated 19 patients with a total of 90 basal cell carcinomas with oral itraconazole at 200 mg twice a day for 1 month or 100 mg twice a day for an average of 2.3 months.
The treatment reduced Hedgehog signaling pathway activity by 65%, Ki67 tumor cell proliferation by 45%, and tumor area by 24% (J Clin Oncol. 2014 Mar 10;32[8]:745-51).
These results aren’t as dramatic as what’s achieved using vismodegib or sonidegib. As stand-alone therapy, itraconazole doesn’t compare with those agents. However, the hope is that when itraconazole is prescribed in conjunction with vismodegib or sonidegib it will permit the latter drugs to be used at lower doses with no drop-off in efficacy, which would mean less grade 2 toxicity. Moreover, since itraconazole inhibits Hedgehog signaling through a mechanism that is different from that of the more potent agents, combination therapy might delay onset of tumor resistance, Dr. Swanson explained.
He reported having no financial conflicts of interest regarding his presentation.
SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
When the Doctor Is Not a Doctor

It is now common for patients to arrive in a physician office and never see the physician. Instead, patients are seen by so-called physician extenders. As our population ages, the need for medical care continues to grow beyond the capacity of the 900,000 US physicians that provide required services, particularly in the first level (primary care). The response to the physician shortage has entailed a variety of strategies. There has been a major immigration of foreign physicians, particularly from India; US medical schools have been encouraged to increase enrollment; and new medical schools have been inaugurated. Physicians have been pushed to adopt electronic medical records to permit increased throughput of patients in office practices. These multiple approaches have had an effect, though sometimes the results are undesirable. For example, complicated computer programs often detract from the physician-patient relationship.
One of the early solutions offered to deal with the doctor shortage in primary care was the concept of physician extenders (PEs), also called mid-level practitioners, who are professionals trained to take on a number of the simpler tasks performed by physicians. There are 2 basic classes of PEs: nurse practitioners and physician assistants. Nurse practitioners are originally trained to perform nursing but then undertake a course of study including scientific courses and clinical exposure to various parts of medicine. Physician assistants receive similar training. The duration of training for PEs usually is 18 to 24 months, whereas physicians attend medical school for 4 years. Unlike physicians, mid-level practitioners do not enter physician postgraduate residency training programs, which last many years.
The original concept was that PEs would work side by side with physicians who would supervise the care provided by the PEs. This team concept was designed to free physicians from the more mundane aspects of medical care and allow them to focus on the more challenging diagnostic and therapeutic issues presented by individual patients. In an era in which the burden of documentation has become increasingly onerous, the assistance of paraprofessionals can spare physicians the entry of redundant details in electronic databases that do not contribute to patient welfare.
However, research suggests that the concept of mid-level providers undertaking first-level care side by side with physicians has diverged from the original goal. An article by Coldiron and Ratnarathorn (JAMA Dermatol. 2014;150:1153-1159) studied Medicare billing data. The authors discovered that a variety of activities, many with higher reimbursement than primary care, were billed directly by PEs without apparent physician involvement, including a large number of complex invasive procedures, more than half in dermatology. Their article focused on dermatologic procedures, such as the destruction of skin cancers and advanced surgical repairs, but they listed many other procedures that are typically in the domain of highly trained physicians, including radiologic interpretations such as mammography and joint injections such as spinal injections. The data they presented were substantiated by publications in the medical literature suggesting that mid-level providers at certain hospitals even perform heart catheterizations and gastrointestinal endoscopies.
There have been no apologies for the unsupervised conduct of physician activities by nonphysicians. On the contrary, many PEs claim to be as well trained and proficient as medical doctors. Coldiron and Ratnarathorn argued otherwise. They pointed out that physicians receive an average of 10,000 hours of training compared to 2000 hours for mid-level practitioners, and they raised concerns about misdiagnoses, complications, and unnecessary procedures performed by PEs without supervision. In an editorial, Jalian and Avram (JAMA Dermatol. 2014;150:1149-1151) pointed out that a disproportionate number of cases of lawsuits for laser-induced injuries are related to performance by nonphysicians.
The pressures to allow nonphysicians to practice medicine independently are increasing. There is a shortage of physicians, especially in states such as Massachusetts that have substantial governmental limitation of physician reimbursement. In Massachusetts, regulations encourage mid-level practitioners to practice without physician supervision and even call themselves “doctors.” Furthermore, hospitals have faced residency funding cuts by Medicare and have had regulatory limitation of work hours by medical doctors in residency training. As a result, many institutions have turned to PEs to perform procedures that are typically performed by medical doctors.
Perhaps the greatest pressure favoring use of nonphysicians is financial. Mid-level practitioners receive lower salaries, typically 45% less, than medical doctors. In an era in which lowering costs has supplanted the goal of offering the best medical care possible, the attraction of replacement of a physician by a professional with less training becomes irresistible. It also is of concern that many physicians ignore the requirement to supervise the work of mid-level practitioners to maximize profit. Physicians often hire a mid-level provider rather than finding another physician to partner in their practice. Patients referred to a dermatologist often are seen by a PE and never even see the physician.
The concept of PEs working in a team with physicians remains an excellent approach to remedying the shortage of medical doctors, but we need to return to the original plan. Physician extenders should perform primary care rather than complex and lucrative subspecialties. There must be adequate supervision and definitely participation by physicians in rendering care.
All of the authors in the articles cited argue for greater regulation of unsupervised PEs to prevent performance of procedures where they lack expertise. Although the regulatory approach is sensible, it is more important to ensure that patients choose who gives them their medical care. They should not be obligated to see mid-level practitioners if they want to see a medical doctor. Above all, patients must be informed of the qualifications of those who provide their medical care. They should not be blindsided when they arrive for an appointment with their physician and find themselves shunted to a PE. We must not allow financial considerations to override the integrity of the medical care process.
What do you think is the optimal and safest role for PEs in a dermatology practice?
We want to know your views! Tell us what you think.

It is now common for patients to arrive in a physician office and never see the physician. Instead, patients are seen by so-called physician extenders. As our population ages, the need for medical care continues to grow beyond the capacity of the 900,000 US physicians that provide required services, particularly in the first level (primary care). The response to the physician shortage has entailed a variety of strategies. There has been a major immigration of foreign physicians, particularly from India; US medical schools have been encouraged to increase enrollment; and new medical schools have been inaugurated. Physicians have been pushed to adopt electronic medical records to permit increased throughput of patients in office practices. These multiple approaches have had an effect, though sometimes the results are undesirable. For example, complicated computer programs often detract from the physician-patient relationship.
One of the early solutions offered to deal with the doctor shortage in primary care was the concept of physician extenders (PEs), also called mid-level practitioners, who are professionals trained to take on a number of the simpler tasks performed by physicians. There are 2 basic classes of PEs: nurse practitioners and physician assistants. Nurse practitioners are originally trained to perform nursing but then undertake a course of study including scientific courses and clinical exposure to various parts of medicine. Physician assistants receive similar training. The duration of training for PEs usually is 18 to 24 months, whereas physicians attend medical school for 4 years. Unlike physicians, mid-level practitioners do not enter physician postgraduate residency training programs, which last many years.
The original concept was that PEs would work side by side with physicians who would supervise the care provided by the PEs. This team concept was designed to free physicians from the more mundane aspects of medical care and allow them to focus on the more challenging diagnostic and therapeutic issues presented by individual patients. In an era in which the burden of documentation has become increasingly onerous, the assistance of paraprofessionals can spare physicians the entry of redundant details in electronic databases that do not contribute to patient welfare.
However, research suggests that the concept of mid-level providers undertaking first-level care side by side with physicians has diverged from the original goal. An article by Coldiron and Ratnarathorn (JAMA Dermatol. 2014;150:1153-1159) studied Medicare billing data. The authors discovered that a variety of activities, many with higher reimbursement than primary care, were billed directly by PEs without apparent physician involvement, including a large number of complex invasive procedures, more than half in dermatology. Their article focused on dermatologic procedures, such as the destruction of skin cancers and advanced surgical repairs, but they listed many other procedures that are typically in the domain of highly trained physicians, including radiologic interpretations such as mammography and joint injections such as spinal injections. The data they presented were substantiated by publications in the medical literature suggesting that mid-level providers at certain hospitals even perform heart catheterizations and gastrointestinal endoscopies.
There have been no apologies for the unsupervised conduct of physician activities by nonphysicians. On the contrary, many PEs claim to be as well trained and proficient as medical doctors. Coldiron and Ratnarathorn argued otherwise. They pointed out that physicians receive an average of 10,000 hours of training compared to 2000 hours for mid-level practitioners, and they raised concerns about misdiagnoses, complications, and unnecessary procedures performed by PEs without supervision. In an editorial, Jalian and Avram (JAMA Dermatol. 2014;150:1149-1151) pointed out that a disproportionate number of cases of lawsuits for laser-induced injuries are related to performance by nonphysicians.
The pressures to allow nonphysicians to practice medicine independently are increasing. There is a shortage of physicians, especially in states such as Massachusetts that have substantial governmental limitation of physician reimbursement. In Massachusetts, regulations encourage mid-level practitioners to practice without physician supervision and even call themselves “doctors.” Furthermore, hospitals have faced residency funding cuts by Medicare and have had regulatory limitation of work hours by medical doctors in residency training. As a result, many institutions have turned to PEs to perform procedures that are typically performed by medical doctors.
Perhaps the greatest pressure favoring use of nonphysicians is financial. Mid-level practitioners receive lower salaries, typically 45% less, than medical doctors. In an era in which lowering costs has supplanted the goal of offering the best medical care possible, the attraction of replacement of a physician by a professional with less training becomes irresistible. It also is of concern that many physicians ignore the requirement to supervise the work of mid-level practitioners to maximize profit. Physicians often hire a mid-level provider rather than finding another physician to partner in their practice. Patients referred to a dermatologist often are seen by a PE and never even see the physician.
The concept of PEs working in a team with physicians remains an excellent approach to remedying the shortage of medical doctors, but we need to return to the original plan. Physician extenders should perform primary care rather than complex and lucrative subspecialties. There must be adequate supervision and definitely participation by physicians in rendering care.
All of the authors in the articles cited argue for greater regulation of unsupervised PEs to prevent performance of procedures where they lack expertise. Although the regulatory approach is sensible, it is more important to ensure that patients choose who gives them their medical care. They should not be obligated to see mid-level practitioners if they want to see a medical doctor. Above all, patients must be informed of the qualifications of those who provide their medical care. They should not be blindsided when they arrive for an appointment with their physician and find themselves shunted to a PE. We must not allow financial considerations to override the integrity of the medical care process.
What do you think is the optimal and safest role for PEs in a dermatology practice?
We want to know your views! Tell us what you think.

It is now common for patients to arrive in a physician office and never see the physician. Instead, patients are seen by so-called physician extenders. As our population ages, the need for medical care continues to grow beyond the capacity of the 900,000 US physicians that provide required services, particularly in the first level (primary care). The response to the physician shortage has entailed a variety of strategies. There has been a major immigration of foreign physicians, particularly from India; US medical schools have been encouraged to increase enrollment; and new medical schools have been inaugurated. Physicians have been pushed to adopt electronic medical records to permit increased throughput of patients in office practices. These multiple approaches have had an effect, though sometimes the results are undesirable. For example, complicated computer programs often detract from the physician-patient relationship.
One of the early solutions offered to deal with the doctor shortage in primary care was the concept of physician extenders (PEs), also called mid-level practitioners, who are professionals trained to take on a number of the simpler tasks performed by physicians. There are 2 basic classes of PEs: nurse practitioners and physician assistants. Nurse practitioners are originally trained to perform nursing but then undertake a course of study including scientific courses and clinical exposure to various parts of medicine. Physician assistants receive similar training. The duration of training for PEs usually is 18 to 24 months, whereas physicians attend medical school for 4 years. Unlike physicians, mid-level practitioners do not enter physician postgraduate residency training programs, which last many years.
The original concept was that PEs would work side by side with physicians who would supervise the care provided by the PEs. This team concept was designed to free physicians from the more mundane aspects of medical care and allow them to focus on the more challenging diagnostic and therapeutic issues presented by individual patients. In an era in which the burden of documentation has become increasingly onerous, the assistance of paraprofessionals can spare physicians the entry of redundant details in electronic databases that do not contribute to patient welfare.
However, research suggests that the concept of mid-level providers undertaking first-level care side by side with physicians has diverged from the original goal. An article by Coldiron and Ratnarathorn (JAMA Dermatol. 2014;150:1153-1159) studied Medicare billing data. The authors discovered that a variety of activities, many with higher reimbursement than primary care, were billed directly by PEs without apparent physician involvement, including a large number of complex invasive procedures, more than half in dermatology. Their article focused on dermatologic procedures, such as the destruction of skin cancers and advanced surgical repairs, but they listed many other procedures that are typically in the domain of highly trained physicians, including radiologic interpretations such as mammography and joint injections such as spinal injections. The data they presented were substantiated by publications in the medical literature suggesting that mid-level providers at certain hospitals even perform heart catheterizations and gastrointestinal endoscopies.
There have been no apologies for the unsupervised conduct of physician activities by nonphysicians. On the contrary, many PEs claim to be as well trained and proficient as medical doctors. Coldiron and Ratnarathorn argued otherwise. They pointed out that physicians receive an average of 10,000 hours of training compared to 2000 hours for mid-level practitioners, and they raised concerns about misdiagnoses, complications, and unnecessary procedures performed by PEs without supervision. In an editorial, Jalian and Avram (JAMA Dermatol. 2014;150:1149-1151) pointed out that a disproportionate number of cases of lawsuits for laser-induced injuries are related to performance by nonphysicians.
The pressures to allow nonphysicians to practice medicine independently are increasing. There is a shortage of physicians, especially in states such as Massachusetts that have substantial governmental limitation of physician reimbursement. In Massachusetts, regulations encourage mid-level practitioners to practice without physician supervision and even call themselves “doctors.” Furthermore, hospitals have faced residency funding cuts by Medicare and have had regulatory limitation of work hours by medical doctors in residency training. As a result, many institutions have turned to PEs to perform procedures that are typically performed by medical doctors.
Perhaps the greatest pressure favoring use of nonphysicians is financial. Mid-level practitioners receive lower salaries, typically 45% less, than medical doctors. In an era in which lowering costs has supplanted the goal of offering the best medical care possible, the attraction of replacement of a physician by a professional with less training becomes irresistible. It also is of concern that many physicians ignore the requirement to supervise the work of mid-level practitioners to maximize profit. Physicians often hire a mid-level provider rather than finding another physician to partner in their practice. Patients referred to a dermatologist often are seen by a PE and never even see the physician.
The concept of PEs working in a team with physicians remains an excellent approach to remedying the shortage of medical doctors, but we need to return to the original plan. Physician extenders should perform primary care rather than complex and lucrative subspecialties. There must be adequate supervision and definitely participation by physicians in rendering care.
All of the authors in the articles cited argue for greater regulation of unsupervised PEs to prevent performance of procedures where they lack expertise. Although the regulatory approach is sensible, it is more important to ensure that patients choose who gives them their medical care. They should not be obligated to see mid-level practitioners if they want to see a medical doctor. Above all, patients must be informed of the qualifications of those who provide their medical care. They should not be blindsided when they arrive for an appointment with their physician and find themselves shunted to a PE. We must not allow financial considerations to override the integrity of the medical care process.
What do you think is the optimal and safest role for PEs in a dermatology practice?
We want to know your views! Tell us what you think.
Average person with atopic dermatitis has no increased risk of actinic keratosis or nonmelanoma skin cancer
People with atopic dermatitis do not appear to be at greater risk for actinic keratosis or basal cell and squamous cell cancer, according to a recent population-based, cross-sectional study.
“This is the first study to examine the association between atopic dermatitis and actinic keratosis [AK]. Our findings suggest that within a population-based sample, atopic dermatitis patients do not have more AKs than the rest of the population. Patients with atopic dermatitis were not found to have more AKs or keratotic cancers [basal or squamous cell cancers]. Moreover, individuals with atopic dermatitis seem to be less likely to develop multiple AKs,” said Dr. Enes Hajdarbegovic and his associates of the Erasmus Medical Centre, Rotterdam, the Netherlands.
The study is part of an ongoing, prospective, Dutch population-based cohort study that follows people in a district of Rotterdam since 1990. There are now 14,926 participants in the database. The current study included 4,375 participants who had undergone full body skin examinations; 56% of patients were female, and the mean age was 68 years (Br J Dermatol. 2016 Jan 29. doi: 10.1111/bjd.14423).
Twenty-four percent had 1 or more AKs; 57% had 1-3 of these lesions; 23% had 4-9, and 20% had more than 10. The mean age of participants with AK was significantly higher, compared with those without AK (73 years vs. 66 years; P less than .01).
Of the 4,375 participants screened, 6.3% met the diagnostic criteria for atopic dermatitis. A lower proportion of those with atopic dermatitis had AK: 16% vs. 24%, respectively (P = .002). In a multinomial model, atopic dermatitis patients were 78% less likely to have 10 or more AKs than were those without atopic dermatitis. No effect of atopic dermatitis was found on basal cell cancer (adjusted odds ratio, 0.71) and squamous cell cancer (adjusted OR, 1.54).
The authors explained that it is already known that patients with severe atopic dermatitis exposed to ultraviolet light and immunosuppressants are at increased risk of keratinocyte malignancies. This study shows that a community-dwelling person with moderate atopic dermatitis does not develop more AKs or keratinocyte cancers.
The investigators said they had no relevant financial disclosures.
People with atopic dermatitis do not appear to be at greater risk for actinic keratosis or basal cell and squamous cell cancer, according to a recent population-based, cross-sectional study.
“This is the first study to examine the association between atopic dermatitis and actinic keratosis [AK]. Our findings suggest that within a population-based sample, atopic dermatitis patients do not have more AKs than the rest of the population. Patients with atopic dermatitis were not found to have more AKs or keratotic cancers [basal or squamous cell cancers]. Moreover, individuals with atopic dermatitis seem to be less likely to develop multiple AKs,” said Dr. Enes Hajdarbegovic and his associates of the Erasmus Medical Centre, Rotterdam, the Netherlands.
The study is part of an ongoing, prospective, Dutch population-based cohort study that follows people in a district of Rotterdam since 1990. There are now 14,926 participants in the database. The current study included 4,375 participants who had undergone full body skin examinations; 56% of patients were female, and the mean age was 68 years (Br J Dermatol. 2016 Jan 29. doi: 10.1111/bjd.14423).
Twenty-four percent had 1 or more AKs; 57% had 1-3 of these lesions; 23% had 4-9, and 20% had more than 10. The mean age of participants with AK was significantly higher, compared with those without AK (73 years vs. 66 years; P less than .01).
Of the 4,375 participants screened, 6.3% met the diagnostic criteria for atopic dermatitis. A lower proportion of those with atopic dermatitis had AK: 16% vs. 24%, respectively (P = .002). In a multinomial model, atopic dermatitis patients were 78% less likely to have 10 or more AKs than were those without atopic dermatitis. No effect of atopic dermatitis was found on basal cell cancer (adjusted odds ratio, 0.71) and squamous cell cancer (adjusted OR, 1.54).
The authors explained that it is already known that patients with severe atopic dermatitis exposed to ultraviolet light and immunosuppressants are at increased risk of keratinocyte malignancies. This study shows that a community-dwelling person with moderate atopic dermatitis does not develop more AKs or keratinocyte cancers.
The investigators said they had no relevant financial disclosures.
People with atopic dermatitis do not appear to be at greater risk for actinic keratosis or basal cell and squamous cell cancer, according to a recent population-based, cross-sectional study.
“This is the first study to examine the association between atopic dermatitis and actinic keratosis [AK]. Our findings suggest that within a population-based sample, atopic dermatitis patients do not have more AKs than the rest of the population. Patients with atopic dermatitis were not found to have more AKs or keratotic cancers [basal or squamous cell cancers]. Moreover, individuals with atopic dermatitis seem to be less likely to develop multiple AKs,” said Dr. Enes Hajdarbegovic and his associates of the Erasmus Medical Centre, Rotterdam, the Netherlands.
The study is part of an ongoing, prospective, Dutch population-based cohort study that follows people in a district of Rotterdam since 1990. There are now 14,926 participants in the database. The current study included 4,375 participants who had undergone full body skin examinations; 56% of patients were female, and the mean age was 68 years (Br J Dermatol. 2016 Jan 29. doi: 10.1111/bjd.14423).
Twenty-four percent had 1 or more AKs; 57% had 1-3 of these lesions; 23% had 4-9, and 20% had more than 10. The mean age of participants with AK was significantly higher, compared with those without AK (73 years vs. 66 years; P less than .01).
Of the 4,375 participants screened, 6.3% met the diagnostic criteria for atopic dermatitis. A lower proportion of those with atopic dermatitis had AK: 16% vs. 24%, respectively (P = .002). In a multinomial model, atopic dermatitis patients were 78% less likely to have 10 or more AKs than were those without atopic dermatitis. No effect of atopic dermatitis was found on basal cell cancer (adjusted odds ratio, 0.71) and squamous cell cancer (adjusted OR, 1.54).
The authors explained that it is already known that patients with severe atopic dermatitis exposed to ultraviolet light and immunosuppressants are at increased risk of keratinocyte malignancies. This study shows that a community-dwelling person with moderate atopic dermatitis does not develop more AKs or keratinocyte cancers.
The investigators said they had no relevant financial disclosures.
FROM THE BRITISH JOURNAL OF DERMATOLOGY
Key clinical point: People with atopic dermatitis do not appear to be at greater risk for actinic keratosis or basal cell and squamous cell cancer.
Major finding: In a multinomial model, atopic dermatitis patients were 78% less likely to have 10 or more actinic keratoses than were those without atopic dermatitis. No effect of atopic dermatitis was found on basal cell cancer (adjusted OR, 0.71) and squamous cell cancer (adjusted OR, 1.54).
Data source: A prospective, Dutch population-based cohort study of 4,375 participants who had undergone full body skin examinations.
Disclosures: The investigators said they had no relevant financial disclosures.
Organ transplant recipients face increased risk of BCC
Recipients of a solid organ transplant face up to a sixfold increase in the risk of developing a basal cell carcinoma – a risk that seems to increase as time passes.
A pretransplant history of squamous cell carcinoma (SCC) increased this risk to 55 times that seen in the general population, Dr. Britta Krynitz and her colleagues reported (Br J Dermatol. 2015. doi: 10.1111/bjd.14153).
But even when the pretransplant SCC group was removed from the final analysis, the risk of basal cell carcinoma after transplant was five times that of the general population, “indicating that a pretransplant SCC has limited effect on BCC risk overall and that organ transplantation per se is a strong driver of posttransplant BCC risk,” wrote Dr. Krynitz of Karolinska Institute, Stockholm, and her coauthors.
“Our results strongly suggest tumor promoter effects of the immunosuppressive drugs in the pathogenesis of post-transplantation BCC,” the team said. “We speculate that calcineurin inhibitors and also antiproliferative drugs, often used in combination with corticosteroids, play a role.”
The researchers investigated the incidence of both BCC and SCC in a cohort of 4,023 patients who underwent solid organ transplant from 2004 to 2011. Their median age at the time of transplant was 53 years; most (59%) received a kidney. Other organs transplanted were liver (22%), heart and/or lung (15%), and other organs (4%). The median follow-up time was 3.4 years; the longest follow-up was 5.5 years.
Only 17 of patients had a history of melanoma, and 19 patients a history of SCC – less than 1% for each skin cancer. Seven percent (301) of patients had experienced some form of nonskin cancer.
By the end of follow-up, 341 BCCs had developed among 175 patients – an incidence of 6.7%. About half developed more than one BCC.
The researchers compared these patients to a group of almost 200,000 nontransplant patients who had developed BCC. Among these, the median age at BCC appearance was significantly older (71 years); 39% had more than one lesion.
The overall relative risk of BCC was increased sixfold in transplant recipients and was similar between the genders. However, the risk varied according to the type of organ received. Kidney recipients were at the highest risk (relative risk, 7.2), and those who received other organs had a lower risk (heart/lung: RR, 5.8; liver: RR, 2.6).
The risk also appeared to increase over time, the authors noted. From 0 to 2 years, it was 5.8; from 3 to 5 years, it increased to 7.0.
Among men, 54% of lesions appeared in the head/neck area and 35% on the trunk – a similar distribution to that seen in the nontransplant control group. Among women, there were differences between transplant patients and controls: 44% of lesions appeared on patients’ head/neck, compared with 60% in the control group, and 34% appeared on the truck, compared with 24% in the control group.
Histology was similar, as were the proportions of aggressive type II and highly aggressive type III lesions.
A total of 199 SCCs developed among 87 patients during follow-up, a ratio to BCC of 1:1.7. “The low ratio was probably due to the short follow-up in our study,” the authors noted.
The Welander Foundation, the Westerberg Foundation, and the Strategic Research Program in Epidemiology at Karolinska Institute sponsored the study. None of the authors had any financial declarations.
Recipients of a solid organ transplant face up to a sixfold increase in the risk of developing a basal cell carcinoma – a risk that seems to increase as time passes.
A pretransplant history of squamous cell carcinoma (SCC) increased this risk to 55 times that seen in the general population, Dr. Britta Krynitz and her colleagues reported (Br J Dermatol. 2015. doi: 10.1111/bjd.14153).
But even when the pretransplant SCC group was removed from the final analysis, the risk of basal cell carcinoma after transplant was five times that of the general population, “indicating that a pretransplant SCC has limited effect on BCC risk overall and that organ transplantation per se is a strong driver of posttransplant BCC risk,” wrote Dr. Krynitz of Karolinska Institute, Stockholm, and her coauthors.
“Our results strongly suggest tumor promoter effects of the immunosuppressive drugs in the pathogenesis of post-transplantation BCC,” the team said. “We speculate that calcineurin inhibitors and also antiproliferative drugs, often used in combination with corticosteroids, play a role.”
The researchers investigated the incidence of both BCC and SCC in a cohort of 4,023 patients who underwent solid organ transplant from 2004 to 2011. Their median age at the time of transplant was 53 years; most (59%) received a kidney. Other organs transplanted were liver (22%), heart and/or lung (15%), and other organs (4%). The median follow-up time was 3.4 years; the longest follow-up was 5.5 years.
Only 17 of patients had a history of melanoma, and 19 patients a history of SCC – less than 1% for each skin cancer. Seven percent (301) of patients had experienced some form of nonskin cancer.
By the end of follow-up, 341 BCCs had developed among 175 patients – an incidence of 6.7%. About half developed more than one BCC.
The researchers compared these patients to a group of almost 200,000 nontransplant patients who had developed BCC. Among these, the median age at BCC appearance was significantly older (71 years); 39% had more than one lesion.
The overall relative risk of BCC was increased sixfold in transplant recipients and was similar between the genders. However, the risk varied according to the type of organ received. Kidney recipients were at the highest risk (relative risk, 7.2), and those who received other organs had a lower risk (heart/lung: RR, 5.8; liver: RR, 2.6).
The risk also appeared to increase over time, the authors noted. From 0 to 2 years, it was 5.8; from 3 to 5 years, it increased to 7.0.
Among men, 54% of lesions appeared in the head/neck area and 35% on the trunk – a similar distribution to that seen in the nontransplant control group. Among women, there were differences between transplant patients and controls: 44% of lesions appeared on patients’ head/neck, compared with 60% in the control group, and 34% appeared on the truck, compared with 24% in the control group.
Histology was similar, as were the proportions of aggressive type II and highly aggressive type III lesions.
A total of 199 SCCs developed among 87 patients during follow-up, a ratio to BCC of 1:1.7. “The low ratio was probably due to the short follow-up in our study,” the authors noted.
The Welander Foundation, the Westerberg Foundation, and the Strategic Research Program in Epidemiology at Karolinska Institute sponsored the study. None of the authors had any financial declarations.
Recipients of a solid organ transplant face up to a sixfold increase in the risk of developing a basal cell carcinoma – a risk that seems to increase as time passes.
A pretransplant history of squamous cell carcinoma (SCC) increased this risk to 55 times that seen in the general population, Dr. Britta Krynitz and her colleagues reported (Br J Dermatol. 2015. doi: 10.1111/bjd.14153).
But even when the pretransplant SCC group was removed from the final analysis, the risk of basal cell carcinoma after transplant was five times that of the general population, “indicating that a pretransplant SCC has limited effect on BCC risk overall and that organ transplantation per se is a strong driver of posttransplant BCC risk,” wrote Dr. Krynitz of Karolinska Institute, Stockholm, and her coauthors.
“Our results strongly suggest tumor promoter effects of the immunosuppressive drugs in the pathogenesis of post-transplantation BCC,” the team said. “We speculate that calcineurin inhibitors and also antiproliferative drugs, often used in combination with corticosteroids, play a role.”
The researchers investigated the incidence of both BCC and SCC in a cohort of 4,023 patients who underwent solid organ transplant from 2004 to 2011. Their median age at the time of transplant was 53 years; most (59%) received a kidney. Other organs transplanted were liver (22%), heart and/or lung (15%), and other organs (4%). The median follow-up time was 3.4 years; the longest follow-up was 5.5 years.
Only 17 of patients had a history of melanoma, and 19 patients a history of SCC – less than 1% for each skin cancer. Seven percent (301) of patients had experienced some form of nonskin cancer.
By the end of follow-up, 341 BCCs had developed among 175 patients – an incidence of 6.7%. About half developed more than one BCC.
The researchers compared these patients to a group of almost 200,000 nontransplant patients who had developed BCC. Among these, the median age at BCC appearance was significantly older (71 years); 39% had more than one lesion.
The overall relative risk of BCC was increased sixfold in transplant recipients and was similar between the genders. However, the risk varied according to the type of organ received. Kidney recipients were at the highest risk (relative risk, 7.2), and those who received other organs had a lower risk (heart/lung: RR, 5.8; liver: RR, 2.6).
The risk also appeared to increase over time, the authors noted. From 0 to 2 years, it was 5.8; from 3 to 5 years, it increased to 7.0.
Among men, 54% of lesions appeared in the head/neck area and 35% on the trunk – a similar distribution to that seen in the nontransplant control group. Among women, there were differences between transplant patients and controls: 44% of lesions appeared on patients’ head/neck, compared with 60% in the control group, and 34% appeared on the truck, compared with 24% in the control group.
Histology was similar, as were the proportions of aggressive type II and highly aggressive type III lesions.
A total of 199 SCCs developed among 87 patients during follow-up, a ratio to BCC of 1:1.7. “The low ratio was probably due to the short follow-up in our study,” the authors noted.
The Welander Foundation, the Westerberg Foundation, and the Strategic Research Program in Epidemiology at Karolinska Institute sponsored the study. None of the authors had any financial declarations.
FROM BRITISH JOURNAL OF DERMATOLOGY
Key clinical point: The risk of a basal cell carcinoma increases after solid organ transplant.
Major finding: Transplant patients have a sixfold increased risk of BCC.
Data source: A retrospective database study of 4,000 transplant patients and almost 200,000 controls.
Disclosures: The Welander Foundation, the Westerberg Foundation, and the Strategic Research Program in Epidemiology at Karolinska Institute sponsored the study. None of the authors had any financial declarations.
Sirolimus reduced posttransplant skin cancer risk
Sirolimus protects organ-transplant recipients against developing skin cancer, reducing their risk by 40%, according to a retrospective cohort study published in JAMA Dermatology on Jan. 20.
Recipients of solid organs are at three- to fourfold higher risk of developing cancer, compared with the general population, and the most common type they get is nonmelanoma skin cancer. The risk of developing cutaneous squamous cell carcinoma is 65-250 times higher in organ-transplant recipients. Drugs that reduce the growth and proliferation of tumor cells by inhibiting mTOR (mammalian target of rapamycin), including sirolimus, are believed to reduce this cancer risk, said Pritesh S. Karia of the department of dermatology, Brigham and Women’s Hospital and Harvard University, Boston, and his associates (JAMA Dermatol. 2016 Jan 20. doi: 10.1001/jamadermatol.2015.5548).
The investigators reviewed the electronic medical records of 329 patients (mean age, 56 years) who underwent organ transplantation at one of the two medical centers during a 9-year period and who then developed a cancer of any type. The study participants received renal (53.8%), heart (17.6%), lung (16.4%), liver (10.3%), or mixed-organ (1.8%) transplants. The most common index cancers they developed post transplant included cutaneous squamous cell carcinoma (31.9%), basal cell carcinoma (22.5%), and melanoma (2.7%).
Of the 329 patients, 97 (29.5%) then received sirolimus, while 232 (70.5%) did not. During a median follow-up of 38 months, 130 of these patients (39.5%) developed a second posttransplant cancer. The sirolimus-treated group showed a reduction in risk for cancer of any type, compared with the group that did not receive sirolimus (30.9% of 97 vs. 43.1% of 232).
Nearly all (88.5%) of the second posttransplant cancers that developed were skin cancers, and sirolimus reduced the risk of skin cancers by 40%. The 1-year, 3-year, and 5-year rates of skin cancer after an index posttransplant cancer were 9.3%, 20.6%, and 24.7% in the sirolimus group, compared with 17.7%, 31.0%, and 35.8%, respectively, in the untreated group, “thus demonstrating a lower risk for skin cancer with sirolimus treatment,” they said.
“Even for patients who have already had difficulty with skin cancer formation, mTOR inhibition appears to be of benefit. No difference in cancer outcomes was observable between sirolimus-treated and [untreated] groups because poor outcomes were rare,” Mr. Karia and his associates wrote.
These findings suggest that sirolimus chemoprevention should be considered for the subset of organ-transplant recipients who develop post-transplant cancer, they noted. The results also highlight the need for dermatologists and transplant physicians “to be aware of skin cancer history, coordinate regular posttransplant surveillance of skin cancers” in patients with organ transplant recipients, especially those with a history of skin cancer, and to communicate closely “as skin cancers form to consider reduction in immunosuppressive therapy or conversion to an mTOR-based regimen if skin cancer formation is of concern,” they added.
This study was supported by sirolimus manufacturer Novartis Pharmaceuticals. Mr. Karia and his associates reported having no relevant financial disclosures.
Sirolimus protects organ-transplant recipients against developing skin cancer, reducing their risk by 40%, according to a retrospective cohort study published in JAMA Dermatology on Jan. 20.
Recipients of solid organs are at three- to fourfold higher risk of developing cancer, compared with the general population, and the most common type they get is nonmelanoma skin cancer. The risk of developing cutaneous squamous cell carcinoma is 65-250 times higher in organ-transplant recipients. Drugs that reduce the growth and proliferation of tumor cells by inhibiting mTOR (mammalian target of rapamycin), including sirolimus, are believed to reduce this cancer risk, said Pritesh S. Karia of the department of dermatology, Brigham and Women’s Hospital and Harvard University, Boston, and his associates (JAMA Dermatol. 2016 Jan 20. doi: 10.1001/jamadermatol.2015.5548).
The investigators reviewed the electronic medical records of 329 patients (mean age, 56 years) who underwent organ transplantation at one of the two medical centers during a 9-year period and who then developed a cancer of any type. The study participants received renal (53.8%), heart (17.6%), lung (16.4%), liver (10.3%), or mixed-organ (1.8%) transplants. The most common index cancers they developed post transplant included cutaneous squamous cell carcinoma (31.9%), basal cell carcinoma (22.5%), and melanoma (2.7%).
Of the 329 patients, 97 (29.5%) then received sirolimus, while 232 (70.5%) did not. During a median follow-up of 38 months, 130 of these patients (39.5%) developed a second posttransplant cancer. The sirolimus-treated group showed a reduction in risk for cancer of any type, compared with the group that did not receive sirolimus (30.9% of 97 vs. 43.1% of 232).
Nearly all (88.5%) of the second posttransplant cancers that developed were skin cancers, and sirolimus reduced the risk of skin cancers by 40%. The 1-year, 3-year, and 5-year rates of skin cancer after an index posttransplant cancer were 9.3%, 20.6%, and 24.7% in the sirolimus group, compared with 17.7%, 31.0%, and 35.8%, respectively, in the untreated group, “thus demonstrating a lower risk for skin cancer with sirolimus treatment,” they said.
“Even for patients who have already had difficulty with skin cancer formation, mTOR inhibition appears to be of benefit. No difference in cancer outcomes was observable between sirolimus-treated and [untreated] groups because poor outcomes were rare,” Mr. Karia and his associates wrote.
These findings suggest that sirolimus chemoprevention should be considered for the subset of organ-transplant recipients who develop post-transplant cancer, they noted. The results also highlight the need for dermatologists and transplant physicians “to be aware of skin cancer history, coordinate regular posttransplant surveillance of skin cancers” in patients with organ transplant recipients, especially those with a history of skin cancer, and to communicate closely “as skin cancers form to consider reduction in immunosuppressive therapy or conversion to an mTOR-based regimen if skin cancer formation is of concern,” they added.
This study was supported by sirolimus manufacturer Novartis Pharmaceuticals. Mr. Karia and his associates reported having no relevant financial disclosures.
Sirolimus protects organ-transplant recipients against developing skin cancer, reducing their risk by 40%, according to a retrospective cohort study published in JAMA Dermatology on Jan. 20.
Recipients of solid organs are at three- to fourfold higher risk of developing cancer, compared with the general population, and the most common type they get is nonmelanoma skin cancer. The risk of developing cutaneous squamous cell carcinoma is 65-250 times higher in organ-transplant recipients. Drugs that reduce the growth and proliferation of tumor cells by inhibiting mTOR (mammalian target of rapamycin), including sirolimus, are believed to reduce this cancer risk, said Pritesh S. Karia of the department of dermatology, Brigham and Women’s Hospital and Harvard University, Boston, and his associates (JAMA Dermatol. 2016 Jan 20. doi: 10.1001/jamadermatol.2015.5548).
The investigators reviewed the electronic medical records of 329 patients (mean age, 56 years) who underwent organ transplantation at one of the two medical centers during a 9-year period and who then developed a cancer of any type. The study participants received renal (53.8%), heart (17.6%), lung (16.4%), liver (10.3%), or mixed-organ (1.8%) transplants. The most common index cancers they developed post transplant included cutaneous squamous cell carcinoma (31.9%), basal cell carcinoma (22.5%), and melanoma (2.7%).
Of the 329 patients, 97 (29.5%) then received sirolimus, while 232 (70.5%) did not. During a median follow-up of 38 months, 130 of these patients (39.5%) developed a second posttransplant cancer. The sirolimus-treated group showed a reduction in risk for cancer of any type, compared with the group that did not receive sirolimus (30.9% of 97 vs. 43.1% of 232).
Nearly all (88.5%) of the second posttransplant cancers that developed were skin cancers, and sirolimus reduced the risk of skin cancers by 40%. The 1-year, 3-year, and 5-year rates of skin cancer after an index posttransplant cancer were 9.3%, 20.6%, and 24.7% in the sirolimus group, compared with 17.7%, 31.0%, and 35.8%, respectively, in the untreated group, “thus demonstrating a lower risk for skin cancer with sirolimus treatment,” they said.
“Even for patients who have already had difficulty with skin cancer formation, mTOR inhibition appears to be of benefit. No difference in cancer outcomes was observable between sirolimus-treated and [untreated] groups because poor outcomes were rare,” Mr. Karia and his associates wrote.
These findings suggest that sirolimus chemoprevention should be considered for the subset of organ-transplant recipients who develop post-transplant cancer, they noted. The results also highlight the need for dermatologists and transplant physicians “to be aware of skin cancer history, coordinate regular posttransplant surveillance of skin cancers” in patients with organ transplant recipients, especially those with a history of skin cancer, and to communicate closely “as skin cancers form to consider reduction in immunosuppressive therapy or conversion to an mTOR-based regimen if skin cancer formation is of concern,” they added.
This study was supported by sirolimus manufacturer Novartis Pharmaceuticals. Mr. Karia and his associates reported having no relevant financial disclosures.
FROM JAMA DERMATOLOGY
Key clinical point: Sirolimus protects organ-transplant recipients against skin cancer.
Major finding: The 1-year, 3-year, and 5-year rates of skin cancer after an index posttransplant cancer were 9.3%, 20.6%, and 24.7% in the sirolimus group, compared with 17.7%, 31.0%, and 35.8% in the untreated group.
Data source: A retrospective cohort study of 329 organ-transplant recipients who had already developed one cancer likely related to their immunosuppressive therapy.
Disclosures: This study was supported by sirolimus manufacturer Novartis Pharmaceuticals. Mr. Karia and his associates reported having no relevant financial disclosures.
EADV: Vismodegib treatment breaks don’t hurt efficacy
COPENHAGEN – Treatment breaks due to adverse events in patients taking vismodegib for advanced basal cell carcinoma don’t appear to compromise the oral hedgehog pathway inhibitor’s efficacy; in fact, they might even enhance it, according to a prespecified interim analysis of the STEVIE trial.
STEVIE is an ongoing phase II, long-term, open-label international study designed primarily to assess the safety of vismodegib (Erivedge) in a situation similar to routine clinical practice. Efficacy and impact on quality of life are secondary endpoints. Although STEVIE has enrolled 1,227 patients, a prespecified interim analysis was conducted in the first 499 followed for at least 12 months, of whom 468 had locally advanced basal cell carcinoma (BCC) and 31 had metastatic BCC, explained Dr. Johan Hansson, an oncologist at the Karolinska Institute in Stockholm.
The drug was dosed at 150 mg once daily continuously in 28-day cycles until disease progression, intolerable toxicity, or study withdrawal. Safety follow-up was conducted at 1, 3, 5, 9, and 12 months. In an earlier report, the complete and partial response rates were 34% and 33%, respectively, in patients with locally advanced BCC, and 7% and 31% in those with metastatic disease (Lancet Oncol. 2015 Jun;16[6]:729-36).
Dr. Hansson presented new data on efficacy outcomes broken down according to treatment breaks, as well as quality of life results, at the annual congress of the European Academy of Dermatology and Venereology.
Twenty-six percent of patients had one or more treatment breaks. Seventy-six patients had one, 41 had two, and 14 had three or more. The median duration of the breaks was 22 days. The two most frequent reasons for treatment breaks were intolerable adverse events in 53% of cases, and lesser adverse events in 23%.
Close to 100% of STEVIE participants had treatment-emergent adverse events. The most common were muscle spasms, alopecia, altered sense of smell, and weight loss.
Although the number of patients with treatment breaks was relatively small, the response rates were higher in patients with more treatment breaks. So was median treatment duration as well as the median number of capsules taken.
Median progression-free survival was 19.8 months in patients with no treatment breaks, was 19.0 months in those with one, and hasn’t yet been reached in patients with two or more breaks.
In interpreting these findings, Dr. Hansson said, “We have to remember that although intriguing, these are tentative results from an exploratory analysis of subgroups in an ongoing study and should be interpreted with caution.”
The oncologist added, however, based upon these promising results he and his coinvestigators plan to look further into the concept of deliberate intermittent dosing of vismodegib.
Quality of life was assessed using the Skindex-16 questionnaire at baseline, again after two and seven 28-day cycles of vismodegib, and at 12 months. Three domains were examined: emotion, function, and symptoms.
A clinically meaningful improvement – defined as a 10-point or greater reduction from baseline – was seen in the emotion domain at all time points in patients with locally advanced BCC, with median improvements of 14.3 points after two cycles and 23.8 points after seven cycles and at the 12-month mark. Clinically meaningful improvement in symptom scores on the Skindex-16 were noted in patients aged 65 and older, in women, and in those with BCCs in locations other than the head or neck. However, no clinically meaningful improvement in the domain of function was seen at any time in patients with locally advanced BCC.
Patients with metastatic BCC didn’t show significant improvement in any of the three quality of life domains at any time point, added Dr. Hansson.
The STEVIE trial is sponsored by F. Hoffmann–La Roche/Genentech. Dr. Hansson reported receiving research grants from and serving as a consultant to Bristol-Myers Squibb, GlaxoSmithKline, Merck, Novartis, and Roche.
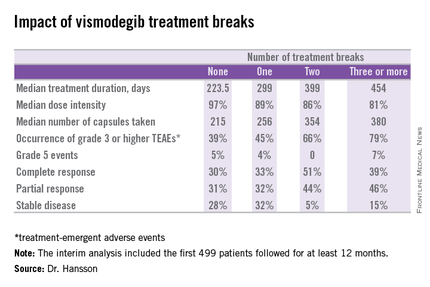
COPENHAGEN – Treatment breaks due to adverse events in patients taking vismodegib for advanced basal cell carcinoma don’t appear to compromise the oral hedgehog pathway inhibitor’s efficacy; in fact, they might even enhance it, according to a prespecified interim analysis of the STEVIE trial.
STEVIE is an ongoing phase II, long-term, open-label international study designed primarily to assess the safety of vismodegib (Erivedge) in a situation similar to routine clinical practice. Efficacy and impact on quality of life are secondary endpoints. Although STEVIE has enrolled 1,227 patients, a prespecified interim analysis was conducted in the first 499 followed for at least 12 months, of whom 468 had locally advanced basal cell carcinoma (BCC) and 31 had metastatic BCC, explained Dr. Johan Hansson, an oncologist at the Karolinska Institute in Stockholm.
The drug was dosed at 150 mg once daily continuously in 28-day cycles until disease progression, intolerable toxicity, or study withdrawal. Safety follow-up was conducted at 1, 3, 5, 9, and 12 months. In an earlier report, the complete and partial response rates were 34% and 33%, respectively, in patients with locally advanced BCC, and 7% and 31% in those with metastatic disease (Lancet Oncol. 2015 Jun;16[6]:729-36).
Dr. Hansson presented new data on efficacy outcomes broken down according to treatment breaks, as well as quality of life results, at the annual congress of the European Academy of Dermatology and Venereology.
Twenty-six percent of patients had one or more treatment breaks. Seventy-six patients had one, 41 had two, and 14 had three or more. The median duration of the breaks was 22 days. The two most frequent reasons for treatment breaks were intolerable adverse events in 53% of cases, and lesser adverse events in 23%.
Close to 100% of STEVIE participants had treatment-emergent adverse events. The most common were muscle spasms, alopecia, altered sense of smell, and weight loss.
Although the number of patients with treatment breaks was relatively small, the response rates were higher in patients with more treatment breaks. So was median treatment duration as well as the median number of capsules taken.
Median progression-free survival was 19.8 months in patients with no treatment breaks, was 19.0 months in those with one, and hasn’t yet been reached in patients with two or more breaks.
In interpreting these findings, Dr. Hansson said, “We have to remember that although intriguing, these are tentative results from an exploratory analysis of subgroups in an ongoing study and should be interpreted with caution.”
The oncologist added, however, based upon these promising results he and his coinvestigators plan to look further into the concept of deliberate intermittent dosing of vismodegib.
Quality of life was assessed using the Skindex-16 questionnaire at baseline, again after two and seven 28-day cycles of vismodegib, and at 12 months. Three domains were examined: emotion, function, and symptoms.
A clinically meaningful improvement – defined as a 10-point or greater reduction from baseline – was seen in the emotion domain at all time points in patients with locally advanced BCC, with median improvements of 14.3 points after two cycles and 23.8 points after seven cycles and at the 12-month mark. Clinically meaningful improvement in symptom scores on the Skindex-16 were noted in patients aged 65 and older, in women, and in those with BCCs in locations other than the head or neck. However, no clinically meaningful improvement in the domain of function was seen at any time in patients with locally advanced BCC.
Patients with metastatic BCC didn’t show significant improvement in any of the three quality of life domains at any time point, added Dr. Hansson.
The STEVIE trial is sponsored by F. Hoffmann–La Roche/Genentech. Dr. Hansson reported receiving research grants from and serving as a consultant to Bristol-Myers Squibb, GlaxoSmithKline, Merck, Novartis, and Roche.

COPENHAGEN – Treatment breaks due to adverse events in patients taking vismodegib for advanced basal cell carcinoma don’t appear to compromise the oral hedgehog pathway inhibitor’s efficacy; in fact, they might even enhance it, according to a prespecified interim analysis of the STEVIE trial.
STEVIE is an ongoing phase II, long-term, open-label international study designed primarily to assess the safety of vismodegib (Erivedge) in a situation similar to routine clinical practice. Efficacy and impact on quality of life are secondary endpoints. Although STEVIE has enrolled 1,227 patients, a prespecified interim analysis was conducted in the first 499 followed for at least 12 months, of whom 468 had locally advanced basal cell carcinoma (BCC) and 31 had metastatic BCC, explained Dr. Johan Hansson, an oncologist at the Karolinska Institute in Stockholm.
The drug was dosed at 150 mg once daily continuously in 28-day cycles until disease progression, intolerable toxicity, or study withdrawal. Safety follow-up was conducted at 1, 3, 5, 9, and 12 months. In an earlier report, the complete and partial response rates were 34% and 33%, respectively, in patients with locally advanced BCC, and 7% and 31% in those with metastatic disease (Lancet Oncol. 2015 Jun;16[6]:729-36).
Dr. Hansson presented new data on efficacy outcomes broken down according to treatment breaks, as well as quality of life results, at the annual congress of the European Academy of Dermatology and Venereology.
Twenty-six percent of patients had one or more treatment breaks. Seventy-six patients had one, 41 had two, and 14 had three or more. The median duration of the breaks was 22 days. The two most frequent reasons for treatment breaks were intolerable adverse events in 53% of cases, and lesser adverse events in 23%.
Close to 100% of STEVIE participants had treatment-emergent adverse events. The most common were muscle spasms, alopecia, altered sense of smell, and weight loss.
Although the number of patients with treatment breaks was relatively small, the response rates were higher in patients with more treatment breaks. So was median treatment duration as well as the median number of capsules taken.
Median progression-free survival was 19.8 months in patients with no treatment breaks, was 19.0 months in those with one, and hasn’t yet been reached in patients with two or more breaks.
In interpreting these findings, Dr. Hansson said, “We have to remember that although intriguing, these are tentative results from an exploratory analysis of subgroups in an ongoing study and should be interpreted with caution.”
The oncologist added, however, based upon these promising results he and his coinvestigators plan to look further into the concept of deliberate intermittent dosing of vismodegib.
Quality of life was assessed using the Skindex-16 questionnaire at baseline, again after two and seven 28-day cycles of vismodegib, and at 12 months. Three domains were examined: emotion, function, and symptoms.
A clinically meaningful improvement – defined as a 10-point or greater reduction from baseline – was seen in the emotion domain at all time points in patients with locally advanced BCC, with median improvements of 14.3 points after two cycles and 23.8 points after seven cycles and at the 12-month mark. Clinically meaningful improvement in symptom scores on the Skindex-16 were noted in patients aged 65 and older, in women, and in those with BCCs in locations other than the head or neck. However, no clinically meaningful improvement in the domain of function was seen at any time in patients with locally advanced BCC.
Patients with metastatic BCC didn’t show significant improvement in any of the three quality of life domains at any time point, added Dr. Hansson.
The STEVIE trial is sponsored by F. Hoffmann–La Roche/Genentech. Dr. Hansson reported receiving research grants from and serving as a consultant to Bristol-Myers Squibb, GlaxoSmithKline, Merck, Novartis, and Roche.

AT THE EADV CONGRESS
Key clinical point: Treatment breaks due to adverse events in patients taking vismodegib for advanced basal cell carcinoma don’t compromise efficacy.
Major finding: The complete response rate to vismodegib in patients with advanced BCC was intriguingly higher in those with more treatment breaks due to adverse events.
Data source: A prespecified interim analysis of the first 499 patients with advanced BCC enrolled in STEVIE, a large ongoing phase II, long-term, open-label international safety study of vismodegib.
Disclosures: The STEVIE trial is sponsored by F. Hoffmann–La Roche/Genentech. The presenter reported receiving research grants from and serving as a consultant to Bristol-Myers Squibb, GlaxoSmithKline, Merck, Novartis, and Roche.
Nonmelanoma skin cancer initially misdiagnosed in 36% of small cohort
In a small sample study, 10 (36%) of 28 children and young adults diagnosed with nonmelanoma skin cancer were given a misdiagnosis initially, suggesting that young patients with NMSC risk factors may require heightened monitoring from health care providers, according to a study published in Journal of the American Academy of Dermatology (2015 doi: 10.1016/j.jaad.2015.08.007).
In their efforts to identify potential risk factors and gaps in care associated with NMSC in pediatric populations, lead author Hasan Khosravi of Harvard Medical School, Boston, and his associates examined records from 28 patients and 182 occurrences of NMSC, collected from Boston Children’s Hospital between 1993 and 2014.
Thirteen (46%) of the 28 pediatric NMSC patients had a history of prolonged immunosuppression, radiation therapy, chemotherapy, voriconazole use, or a combination of these. Among these 28 patients, 19 were diagnosed with basal cell carcinoma (BCC), 7 were diagnosed with squamous cell carcinoma (SCC), and 2 were diagnosed with both BCC and SCC.
The authors noted significant delays in the initial diagnosis in both types of cancer; the mean number of days from time of lesion onset to diagnosis was 667 for SCC and 1,176 for BCC. When misdiagnosed, carcinomas were incorrectly identified as viral wart and graft-versus-host disease for SCC, and psoriasis, acrochordon, wart, nevus, and atypical nevus for BCC.
Even so, interventions were effective in the pediatric population, the authors noted.
“Although most of our patients developed subsequent NMSC after their initial diagnosis, the majority of cases were treated successfully with surgical excision, without recurrence or spread of disease. This suggests that interventions in children and young adults that involve prevention of subsequent disease may be most impactful,” they wrote.
The researchers had no conflicts to declare.
In a small sample study, 10 (36%) of 28 children and young adults diagnosed with nonmelanoma skin cancer were given a misdiagnosis initially, suggesting that young patients with NMSC risk factors may require heightened monitoring from health care providers, according to a study published in Journal of the American Academy of Dermatology (2015 doi: 10.1016/j.jaad.2015.08.007).
In their efforts to identify potential risk factors and gaps in care associated with NMSC in pediatric populations, lead author Hasan Khosravi of Harvard Medical School, Boston, and his associates examined records from 28 patients and 182 occurrences of NMSC, collected from Boston Children’s Hospital between 1993 and 2014.
Thirteen (46%) of the 28 pediatric NMSC patients had a history of prolonged immunosuppression, radiation therapy, chemotherapy, voriconazole use, or a combination of these. Among these 28 patients, 19 were diagnosed with basal cell carcinoma (BCC), 7 were diagnosed with squamous cell carcinoma (SCC), and 2 were diagnosed with both BCC and SCC.
The authors noted significant delays in the initial diagnosis in both types of cancer; the mean number of days from time of lesion onset to diagnosis was 667 for SCC and 1,176 for BCC. When misdiagnosed, carcinomas were incorrectly identified as viral wart and graft-versus-host disease for SCC, and psoriasis, acrochordon, wart, nevus, and atypical nevus for BCC.
Even so, interventions were effective in the pediatric population, the authors noted.
“Although most of our patients developed subsequent NMSC after their initial diagnosis, the majority of cases were treated successfully with surgical excision, without recurrence or spread of disease. This suggests that interventions in children and young adults that involve prevention of subsequent disease may be most impactful,” they wrote.
The researchers had no conflicts to declare.
In a small sample study, 10 (36%) of 28 children and young adults diagnosed with nonmelanoma skin cancer were given a misdiagnosis initially, suggesting that young patients with NMSC risk factors may require heightened monitoring from health care providers, according to a study published in Journal of the American Academy of Dermatology (2015 doi: 10.1016/j.jaad.2015.08.007).
In their efforts to identify potential risk factors and gaps in care associated with NMSC in pediatric populations, lead author Hasan Khosravi of Harvard Medical School, Boston, and his associates examined records from 28 patients and 182 occurrences of NMSC, collected from Boston Children’s Hospital between 1993 and 2014.
Thirteen (46%) of the 28 pediatric NMSC patients had a history of prolonged immunosuppression, radiation therapy, chemotherapy, voriconazole use, or a combination of these. Among these 28 patients, 19 were diagnosed with basal cell carcinoma (BCC), 7 were diagnosed with squamous cell carcinoma (SCC), and 2 were diagnosed with both BCC and SCC.
The authors noted significant delays in the initial diagnosis in both types of cancer; the mean number of days from time of lesion onset to diagnosis was 667 for SCC and 1,176 for BCC. When misdiagnosed, carcinomas were incorrectly identified as viral wart and graft-versus-host disease for SCC, and psoriasis, acrochordon, wart, nevus, and atypical nevus for BCC.
Even so, interventions were effective in the pediatric population, the authors noted.
“Although most of our patients developed subsequent NMSC after their initial diagnosis, the majority of cases were treated successfully with surgical excision, without recurrence or spread of disease. This suggests that interventions in children and young adults that involve prevention of subsequent disease may be most impactful,” they wrote.
The researchers had no conflicts to declare.
FROM JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
EADV: Prophylactic photodynamic therapy benefits transplant recipients
COPENHAGEN – Twice-yearly prophylactic photodynamic therapy for primary prevention of actinic keratoses and squamous cell carcinomas is a novel and effective strategy that addresses the problem of accelerated photocarcinogenesis in organ transplant recipients, according to an interim analysis of a multinational, randomized, controlled trial.
“The overall aim is to prevent squamous cell carcinoma development. Photodynamic therapy is well established for secondary prevention of further AKs, and these very early data show that it can also be used for primary prevention in very high-risk patients,” Dr. Katrine Togsverd-Bo said at the annual congress of the European Academy of Dermatology and Venereology.
Accelerated carcinogenesis on sun-exposed skin is a major concern in organ transplant recipients (OTRs). They experience early onset of multiple AKs, with field cancerization and up to a 100-fold increased risk of squamous cell carcinomas (SCCs). Moreover, their SCCs are at substantially greater risk of metastasis than SCCs occurring in the general population, noted Dr. Togsverd-Bo of Bispebjerg Hospital and the University of Copenhagen.
She presented an interim analysis of an ongoing 5-year prospective randomized trial in 50 renal transplant recipients at academic dermatology centers in Copenhagen, Oslo, and Gothenburg, Sweden. All participants had clinically normal-appearing skin at baseline, with no history of AKs or SCCs. They are undergoing twice-yearly, split-side photodynamic therapy (PDT) on the face, forearm, and hand, with the opposite side serving as the untreated control.
To date, 25 patients have completed 3 years of the study. At 3 years of prospective follow-up by blinded evaluators, 50% of patients had AKs on their untreated side, compared with 26% on the prophylactic PDT side. The collective number of AKs on untreated skin was 43, compared with just 8 AKs on PDT-treated skin. Seven patients had AKs only on their untreated side, six had AKs on both sides, and none had any AKs only on their PDT-treated side.
The twice-yearly prophylactic PDT regimen consists of a 3-hour application of 20% methyl aminolevulinate as a photosensitizer followed by applications of a conventional LED light at 37 J/cm2.
Dr. Togsverd-Bo reported having no financial conflicts regarding her study.
COPENHAGEN – Twice-yearly prophylactic photodynamic therapy for primary prevention of actinic keratoses and squamous cell carcinomas is a novel and effective strategy that addresses the problem of accelerated photocarcinogenesis in organ transplant recipients, according to an interim analysis of a multinational, randomized, controlled trial.
“The overall aim is to prevent squamous cell carcinoma development. Photodynamic therapy is well established for secondary prevention of further AKs, and these very early data show that it can also be used for primary prevention in very high-risk patients,” Dr. Katrine Togsverd-Bo said at the annual congress of the European Academy of Dermatology and Venereology.
Accelerated carcinogenesis on sun-exposed skin is a major concern in organ transplant recipients (OTRs). They experience early onset of multiple AKs, with field cancerization and up to a 100-fold increased risk of squamous cell carcinomas (SCCs). Moreover, their SCCs are at substantially greater risk of metastasis than SCCs occurring in the general population, noted Dr. Togsverd-Bo of Bispebjerg Hospital and the University of Copenhagen.
She presented an interim analysis of an ongoing 5-year prospective randomized trial in 50 renal transplant recipients at academic dermatology centers in Copenhagen, Oslo, and Gothenburg, Sweden. All participants had clinically normal-appearing skin at baseline, with no history of AKs or SCCs. They are undergoing twice-yearly, split-side photodynamic therapy (PDT) on the face, forearm, and hand, with the opposite side serving as the untreated control.
To date, 25 patients have completed 3 years of the study. At 3 years of prospective follow-up by blinded evaluators, 50% of patients had AKs on their untreated side, compared with 26% on the prophylactic PDT side. The collective number of AKs on untreated skin was 43, compared with just 8 AKs on PDT-treated skin. Seven patients had AKs only on their untreated side, six had AKs on both sides, and none had any AKs only on their PDT-treated side.
The twice-yearly prophylactic PDT regimen consists of a 3-hour application of 20% methyl aminolevulinate as a photosensitizer followed by applications of a conventional LED light at 37 J/cm2.
Dr. Togsverd-Bo reported having no financial conflicts regarding her study.
COPENHAGEN – Twice-yearly prophylactic photodynamic therapy for primary prevention of actinic keratoses and squamous cell carcinomas is a novel and effective strategy that addresses the problem of accelerated photocarcinogenesis in organ transplant recipients, according to an interim analysis of a multinational, randomized, controlled trial.
“The overall aim is to prevent squamous cell carcinoma development. Photodynamic therapy is well established for secondary prevention of further AKs, and these very early data show that it can also be used for primary prevention in very high-risk patients,” Dr. Katrine Togsverd-Bo said at the annual congress of the European Academy of Dermatology and Venereology.
Accelerated carcinogenesis on sun-exposed skin is a major concern in organ transplant recipients (OTRs). They experience early onset of multiple AKs, with field cancerization and up to a 100-fold increased risk of squamous cell carcinomas (SCCs). Moreover, their SCCs are at substantially greater risk of metastasis than SCCs occurring in the general population, noted Dr. Togsverd-Bo of Bispebjerg Hospital and the University of Copenhagen.
She presented an interim analysis of an ongoing 5-year prospective randomized trial in 50 renal transplant recipients at academic dermatology centers in Copenhagen, Oslo, and Gothenburg, Sweden. All participants had clinically normal-appearing skin at baseline, with no history of AKs or SCCs. They are undergoing twice-yearly, split-side photodynamic therapy (PDT) on the face, forearm, and hand, with the opposite side serving as the untreated control.
To date, 25 patients have completed 3 years of the study. At 3 years of prospective follow-up by blinded evaluators, 50% of patients had AKs on their untreated side, compared with 26% on the prophylactic PDT side. The collective number of AKs on untreated skin was 43, compared with just 8 AKs on PDT-treated skin. Seven patients had AKs only on their untreated side, six had AKs on both sides, and none had any AKs only on their PDT-treated side.
The twice-yearly prophylactic PDT regimen consists of a 3-hour application of 20% methyl aminolevulinate as a photosensitizer followed by applications of a conventional LED light at 37 J/cm2.
Dr. Togsverd-Bo reported having no financial conflicts regarding her study.
AT THE EADV CONGRESS
Key clinical point: Prophylactic photodynamic therapy is a new and effective strategy for primary prevention of actinic keratoses and squamous cell carcinomas in organ transplant recipients.
Major finding: At 3 years of follow-up, 25 renal transplant recipients collectively had 8 actinic keratoses on the side of their face, forearms, and hands treated with twice-yearly prophylactic photodynamic therapy, compared with 43 AKs on the untreated control side.
Data source: This is an interim 3-year analysis from an ongoing 5-year prospective multinational, randomized, controlled trial involving 50 renal transplant recipients.
Disclosures: The presenter reported having no financial conflicts regarding this ongoing study.