User login
Nonsurgical Alternatives for Skin Cancer Treatment: Report From the AAD Meeting
An important topic at the 2014 Summer AAD Meeting in Chicago, Illinois, was the nonsurgical treatment of skin cancers, including adjuvant therapy, topical creams, photodynamic therapy, and radiation for melanoma and nonmelanoma skin cancers. Dr. Anthony M. Rossi discusses the benefits of some of these nonsurgical treatment options for skin cancers and describes how he uses them in his practice. He also discusses how to determine which treatment option is best for each patient and emphasizes the importance of patient compliance and close follow-up.
An important topic at the 2014 Summer AAD Meeting in Chicago, Illinois, was the nonsurgical treatment of skin cancers, including adjuvant therapy, topical creams, photodynamic therapy, and radiation for melanoma and nonmelanoma skin cancers. Dr. Anthony M. Rossi discusses the benefits of some of these nonsurgical treatment options for skin cancers and describes how he uses them in his practice. He also discusses how to determine which treatment option is best for each patient and emphasizes the importance of patient compliance and close follow-up.
An important topic at the 2014 Summer AAD Meeting in Chicago, Illinois, was the nonsurgical treatment of skin cancers, including adjuvant therapy, topical creams, photodynamic therapy, and radiation for melanoma and nonmelanoma skin cancers. Dr. Anthony M. Rossi discusses the benefits of some of these nonsurgical treatment options for skin cancers and describes how he uses them in his practice. He also discusses how to determine which treatment option is best for each patient and emphasizes the importance of patient compliance and close follow-up.
AUDIO: Unusual approaches to unusual tumors
CHICAGO – Doctors who don’t abide by accepted norms can sometimes achieve better outcomes when treating patients who have unusual and tenacious tumors. This was the theme of a session covering cases of confounding tumors at the American Academy of Dermatology summer meeting.
In an interview after the session, panel moderator and case presenter Dr. John A. Carucci, chief of Mohs and dermatologic surgery at New York (N.Y.) University, shared his thoughts on when certain kinds of imaging are more appropriate than others, even if it’s not the "usual way."
Dr. Carucci also addressed the use of postsurgical negative pressure wound therapy, radiation therapy in conjunction with Mohs surgery, and potentially controversial topics such as the use of a protein kinase inhibitor as a neoadjuvant therapy.
And what new information on staging squamous cell carcinomas has Dr. Carucci and his colleagues excited? Listen and find out. Dr. Carucci said that he had no financial conflicts to disclose.
On Twitter @whitneymcknight
CHICAGO – Doctors who don’t abide by accepted norms can sometimes achieve better outcomes when treating patients who have unusual and tenacious tumors. This was the theme of a session covering cases of confounding tumors at the American Academy of Dermatology summer meeting.
In an interview after the session, panel moderator and case presenter Dr. John A. Carucci, chief of Mohs and dermatologic surgery at New York (N.Y.) University, shared his thoughts on when certain kinds of imaging are more appropriate than others, even if it’s not the "usual way."
Dr. Carucci also addressed the use of postsurgical negative pressure wound therapy, radiation therapy in conjunction with Mohs surgery, and potentially controversial topics such as the use of a protein kinase inhibitor as a neoadjuvant therapy.
And what new information on staging squamous cell carcinomas has Dr. Carucci and his colleagues excited? Listen and find out. Dr. Carucci said that he had no financial conflicts to disclose.
On Twitter @whitneymcknight
CHICAGO – Doctors who don’t abide by accepted norms can sometimes achieve better outcomes when treating patients who have unusual and tenacious tumors. This was the theme of a session covering cases of confounding tumors at the American Academy of Dermatology summer meeting.
In an interview after the session, panel moderator and case presenter Dr. John A. Carucci, chief of Mohs and dermatologic surgery at New York (N.Y.) University, shared his thoughts on when certain kinds of imaging are more appropriate than others, even if it’s not the "usual way."
Dr. Carucci also addressed the use of postsurgical negative pressure wound therapy, radiation therapy in conjunction with Mohs surgery, and potentially controversial topics such as the use of a protein kinase inhibitor as a neoadjuvant therapy.
And what new information on staging squamous cell carcinomas has Dr. Carucci and his colleagues excited? Listen and find out. Dr. Carucci said that he had no financial conflicts to disclose.
On Twitter @whitneymcknight
EXPERT ANALYSIS FROM THE AAD SUMMER ACADEMY 2014
What Is Your Diagnosis? Granulocytic Sarcoma
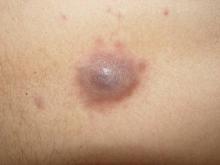
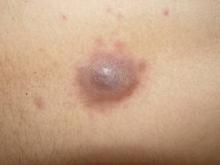
A 24-year-old man presented with a 5-cm asymptomatic, indurated, well-defined nodule with a violaceous center surrounded by a greenish halo on the lower back of 3 months’ duration, along with lymphadenopathy. There was no history of trauma, weight loss, bleeding, or systemic illness. A biopsy revealed sheets of pleomorphic cells with lobulated nuclei and detectable granular cytoplasm. Immunohistochemistry showed strong myeloperoxidase activity in neoplastic cells that were CD13- and CD56-.
The Diagnosis: Granulocytic Sarcoma
Granulocytic sarcoma (GS), sometimes known as leukemia cutis, myeloid sarcoma, or an extramedullary myeloid tumor, is a rare localized collection of immature cells of granulocytic series or the cells of each maturation step in extramedullary sites. Originally described in 1811, the condition was noted after identifying an extramedullary collection of immature granulocytes involving the orbit. Then in 1853 the term chloroma was coined because of the greenish appearance of the tumor when exposed to light due to myeloperoxidase activity in the tumor cells. Finally, in 1967 the term granulocytic sarcoma was introduced because the greenish appearance was not observed in all cases, providing a more correct term.1
Granulocytic sarcoma often manifests in patients with acute myeloid leukemia, chronic myelogenous leukemia, chronic idiopathic myelofibrosis, eosinophilia, refractory anemia, or polycythemia vera, and as an isolated form in patients without apparent hematologic disorder.2
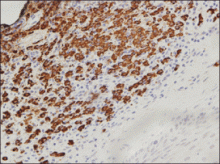
Cutaneous manifestations of leukemia can be specific (eg, GS) or nonspecific (eg, panniculitis, generalized pruritus).2,3 The tumor often appears as a solitary mass (Figure 1); however, in rare instances it can be multifocal. It usually presents as a rapidly growing nodular mass with solid consistence, and it is seen in patients of any age but most often those aged 45 to 55 years as well as in patients younger than 15 years.4
Granulocytic sarcoma is most commonly associated with acute myeloid leukemia. However, it is interesting that the chloroma appears before the onset of leukemia. Occasionally GS is associated with acute myeloid leukemia as a presenting symptom or as a manifestation of recurrence of the disease that was usually related to poor prognosis.5 In patients with GS that is associated with myeloproliferative neoplasms, the occurrence of GS frequently is associated with acute transformation of the disease and therefore a poor prognosis.6
The most common sites of involvement are bone, soft tissue, lymph nodes, and skin. The prognosis does not vary according to location.
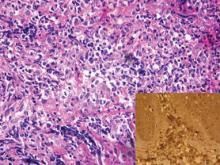
Cutaneous B-cell lymphoma should be considered in the differential diagnosis, as it may be clinically observed as single or multiple violaceous nodules involving the head, neck, and/or trunk. Histologically, there generally is a bottom-heavy infiltrate of lymphocytes sparing the epidermis (Figure 2). Immunohistochemistry, flow cytometry, or cytochemical stains often are needed to exclude lymphoma. Pseudochloroma is a rare disease characterized by a collection of normal hematopoietic tissue in locations other than GS. It can be distinguished from GS by its constituent of mature cells.7
Granulocytic sarcoma implies a therapeutic challenge for the clinician. It should always be considered as part of a systemic disease rather than an isolated skin disorder. In patients with evidence of leukemia and GS, systemic chemotherapy is the first line of treatment. Other effective treatment regimens include radiotherapy (electron beam radiation), either alone or in combination with systemic chemotherapy (eg, hydroxyurea, methotrexate).8 If GS persists after chemotherapy, local treatment (eg, radiotherapy, surgery) should be considered with variable results.2
1. Meis JM, Butler JJ, Osborne BM, et al. Granulocytic sarcoma in nonleukemic patients. Cancer. 1986;58:2697-2709.
2. Sun NCJ, Ellis R. Granulocytic sarcoma of the skin. Arch Dermatol. 1980;116:800-802.
3. Campidelli C, Agostinelli C, Stitson R, et al. Myeloid sarcoma. Extramedullary manifestation of myeloid disorders. Am J Clin Pathol. 2009;132:426-437.
4. Pulsoni A, Falcucci P, Anghel G, et al. Isolated granulocytic sarcoma of the skin in an elderly patient: good response to treatment with local radiotherapy and low dose methotrexate. J Eur Acad Dermatol Venereol. 2000;14:216-218.
5. Neiman RS, Barcos M, Berard C, et al. Granulocytic sarcoma: a clinicopathologic study of 61 biopsied cases. Cancer. 1981;48:1426-1437.
6. Byrd JC, Edenfield WJ, Dow NS, et al. Extramedullary myeloid cell tumors in myelodysplastic syndromes: not a true indication of impending acute myeloid leukemia. Leukemia. 1996;21:153-159.
7. Beswick SJ, Jones EL, Mahendra P, et al. Chloroma (aleukaemic leukaemia cutis) initially diagnosed as a cutaneous lymphoma. Clin Exp Dermatol. 2002;27:272-274.
8. Chen WY, Wang CW, Chang CH, et al. Clinicopathologic features and responses to radiotherapy of myeloid sarcoma. Radiat Oncol. 2013;8:245.
A 24-year-old man presented with a 5-cm asymptomatic, indurated, well-defined nodule with a violaceous center surrounded by a greenish halo on the lower back of 3 months’ duration, along with lymphadenopathy. There was no history of trauma, weight loss, bleeding, or systemic illness. A biopsy revealed sheets of pleomorphic cells with lobulated nuclei and detectable granular cytoplasm. Immunohistochemistry showed strong myeloperoxidase activity in neoplastic cells that were CD13- and CD56-.
The Diagnosis: Granulocytic Sarcoma
Granulocytic sarcoma (GS), sometimes known as leukemia cutis, myeloid sarcoma, or an extramedullary myeloid tumor, is a rare localized collection of immature cells of granulocytic series or the cells of each maturation step in extramedullary sites. Originally described in 1811, the condition was noted after identifying an extramedullary collection of immature granulocytes involving the orbit. Then in 1853 the term chloroma was coined because of the greenish appearance of the tumor when exposed to light due to myeloperoxidase activity in the tumor cells. Finally, in 1967 the term granulocytic sarcoma was introduced because the greenish appearance was not observed in all cases, providing a more correct term.1
Granulocytic sarcoma often manifests in patients with acute myeloid leukemia, chronic myelogenous leukemia, chronic idiopathic myelofibrosis, eosinophilia, refractory anemia, or polycythemia vera, and as an isolated form in patients without apparent hematologic disorder.2
Cutaneous manifestations of leukemia can be specific (eg, GS) or nonspecific (eg, panniculitis, generalized pruritus).2,3 The tumor often appears as a solitary mass (Figure 1); however, in rare instances it can be multifocal. It usually presents as a rapidly growing nodular mass with solid consistence, and it is seen in patients of any age but most often those aged 45 to 55 years as well as in patients younger than 15 years.4
Granulocytic sarcoma is most commonly associated with acute myeloid leukemia. However, it is interesting that the chloroma appears before the onset of leukemia. Occasionally GS is associated with acute myeloid leukemia as a presenting symptom or as a manifestation of recurrence of the disease that was usually related to poor prognosis.5 In patients with GS that is associated with myeloproliferative neoplasms, the occurrence of GS frequently is associated with acute transformation of the disease and therefore a poor prognosis.6
The most common sites of involvement are bone, soft tissue, lymph nodes, and skin. The prognosis does not vary according to location.
Cutaneous B-cell lymphoma should be considered in the differential diagnosis, as it may be clinically observed as single or multiple violaceous nodules involving the head, neck, and/or trunk. Histologically, there generally is a bottom-heavy infiltrate of lymphocytes sparing the epidermis (Figure 2). Immunohistochemistry, flow cytometry, or cytochemical stains often are needed to exclude lymphoma. Pseudochloroma is a rare disease characterized by a collection of normal hematopoietic tissue in locations other than GS. It can be distinguished from GS by its constituent of mature cells.7
Granulocytic sarcoma implies a therapeutic challenge for the clinician. It should always be considered as part of a systemic disease rather than an isolated skin disorder. In patients with evidence of leukemia and GS, systemic chemotherapy is the first line of treatment. Other effective treatment regimens include radiotherapy (electron beam radiation), either alone or in combination with systemic chemotherapy (eg, hydroxyurea, methotrexate).8 If GS persists after chemotherapy, local treatment (eg, radiotherapy, surgery) should be considered with variable results.2
A 24-year-old man presented with a 5-cm asymptomatic, indurated, well-defined nodule with a violaceous center surrounded by a greenish halo on the lower back of 3 months’ duration, along with lymphadenopathy. There was no history of trauma, weight loss, bleeding, or systemic illness. A biopsy revealed sheets of pleomorphic cells with lobulated nuclei and detectable granular cytoplasm. Immunohistochemistry showed strong myeloperoxidase activity in neoplastic cells that were CD13- and CD56-.
The Diagnosis: Granulocytic Sarcoma
Granulocytic sarcoma (GS), sometimes known as leukemia cutis, myeloid sarcoma, or an extramedullary myeloid tumor, is a rare localized collection of immature cells of granulocytic series or the cells of each maturation step in extramedullary sites. Originally described in 1811, the condition was noted after identifying an extramedullary collection of immature granulocytes involving the orbit. Then in 1853 the term chloroma was coined because of the greenish appearance of the tumor when exposed to light due to myeloperoxidase activity in the tumor cells. Finally, in 1967 the term granulocytic sarcoma was introduced because the greenish appearance was not observed in all cases, providing a more correct term.1
Granulocytic sarcoma often manifests in patients with acute myeloid leukemia, chronic myelogenous leukemia, chronic idiopathic myelofibrosis, eosinophilia, refractory anemia, or polycythemia vera, and as an isolated form in patients without apparent hematologic disorder.2
Cutaneous manifestations of leukemia can be specific (eg, GS) or nonspecific (eg, panniculitis, generalized pruritus).2,3 The tumor often appears as a solitary mass (Figure 1); however, in rare instances it can be multifocal. It usually presents as a rapidly growing nodular mass with solid consistence, and it is seen in patients of any age but most often those aged 45 to 55 years as well as in patients younger than 15 years.4
Granulocytic sarcoma is most commonly associated with acute myeloid leukemia. However, it is interesting that the chloroma appears before the onset of leukemia. Occasionally GS is associated with acute myeloid leukemia as a presenting symptom or as a manifestation of recurrence of the disease that was usually related to poor prognosis.5 In patients with GS that is associated with myeloproliferative neoplasms, the occurrence of GS frequently is associated with acute transformation of the disease and therefore a poor prognosis.6
The most common sites of involvement are bone, soft tissue, lymph nodes, and skin. The prognosis does not vary according to location.
Cutaneous B-cell lymphoma should be considered in the differential diagnosis, as it may be clinically observed as single or multiple violaceous nodules involving the head, neck, and/or trunk. Histologically, there generally is a bottom-heavy infiltrate of lymphocytes sparing the epidermis (Figure 2). Immunohistochemistry, flow cytometry, or cytochemical stains often are needed to exclude lymphoma. Pseudochloroma is a rare disease characterized by a collection of normal hematopoietic tissue in locations other than GS. It can be distinguished from GS by its constituent of mature cells.7
Granulocytic sarcoma implies a therapeutic challenge for the clinician. It should always be considered as part of a systemic disease rather than an isolated skin disorder. In patients with evidence of leukemia and GS, systemic chemotherapy is the first line of treatment. Other effective treatment regimens include radiotherapy (electron beam radiation), either alone or in combination with systemic chemotherapy (eg, hydroxyurea, methotrexate).8 If GS persists after chemotherapy, local treatment (eg, radiotherapy, surgery) should be considered with variable results.2
1. Meis JM, Butler JJ, Osborne BM, et al. Granulocytic sarcoma in nonleukemic patients. Cancer. 1986;58:2697-2709.
2. Sun NCJ, Ellis R. Granulocytic sarcoma of the skin. Arch Dermatol. 1980;116:800-802.
3. Campidelli C, Agostinelli C, Stitson R, et al. Myeloid sarcoma. Extramedullary manifestation of myeloid disorders. Am J Clin Pathol. 2009;132:426-437.
4. Pulsoni A, Falcucci P, Anghel G, et al. Isolated granulocytic sarcoma of the skin in an elderly patient: good response to treatment with local radiotherapy and low dose methotrexate. J Eur Acad Dermatol Venereol. 2000;14:216-218.
5. Neiman RS, Barcos M, Berard C, et al. Granulocytic sarcoma: a clinicopathologic study of 61 biopsied cases. Cancer. 1981;48:1426-1437.
6. Byrd JC, Edenfield WJ, Dow NS, et al. Extramedullary myeloid cell tumors in myelodysplastic syndromes: not a true indication of impending acute myeloid leukemia. Leukemia. 1996;21:153-159.
7. Beswick SJ, Jones EL, Mahendra P, et al. Chloroma (aleukaemic leukaemia cutis) initially diagnosed as a cutaneous lymphoma. Clin Exp Dermatol. 2002;27:272-274.
8. Chen WY, Wang CW, Chang CH, et al. Clinicopathologic features and responses to radiotherapy of myeloid sarcoma. Radiat Oncol. 2013;8:245.
1. Meis JM, Butler JJ, Osborne BM, et al. Granulocytic sarcoma in nonleukemic patients. Cancer. 1986;58:2697-2709.
2. Sun NCJ, Ellis R. Granulocytic sarcoma of the skin. Arch Dermatol. 1980;116:800-802.
3. Campidelli C, Agostinelli C, Stitson R, et al. Myeloid sarcoma. Extramedullary manifestation of myeloid disorders. Am J Clin Pathol. 2009;132:426-437.
4. Pulsoni A, Falcucci P, Anghel G, et al. Isolated granulocytic sarcoma of the skin in an elderly patient: good response to treatment with local radiotherapy and low dose methotrexate. J Eur Acad Dermatol Venereol. 2000;14:216-218.
5. Neiman RS, Barcos M, Berard C, et al. Granulocytic sarcoma: a clinicopathologic study of 61 biopsied cases. Cancer. 1981;48:1426-1437.
6. Byrd JC, Edenfield WJ, Dow NS, et al. Extramedullary myeloid cell tumors in myelodysplastic syndromes: not a true indication of impending acute myeloid leukemia. Leukemia. 1996;21:153-159.
7. Beswick SJ, Jones EL, Mahendra P, et al. Chloroma (aleukaemic leukaemia cutis) initially diagnosed as a cutaneous lymphoma. Clin Exp Dermatol. 2002;27:272-274.
8. Chen WY, Wang CW, Chang CH, et al. Clinicopathologic features and responses to radiotherapy of myeloid sarcoma. Radiat Oncol. 2013;8:245.
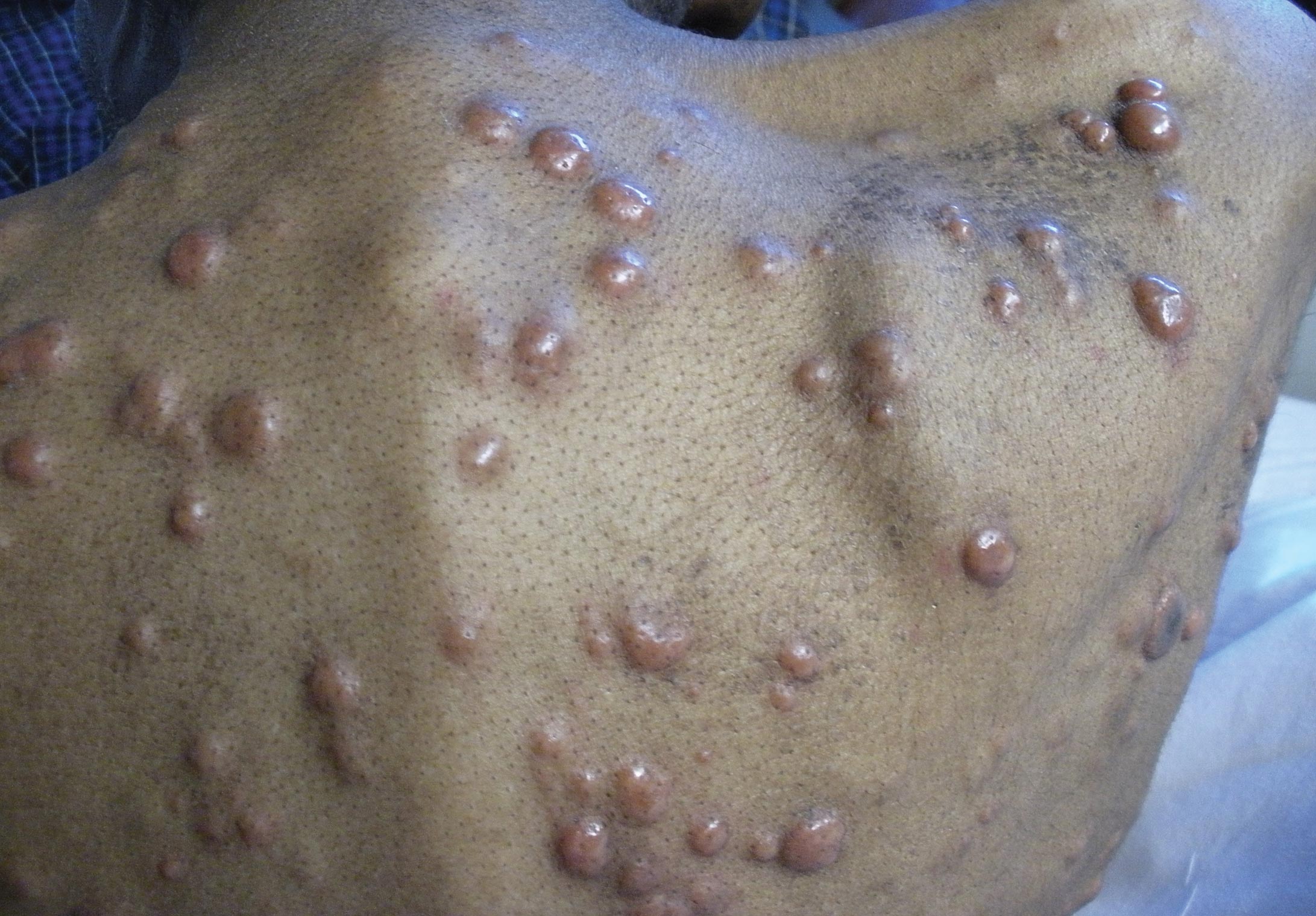
Tender Subcutaneous Nodules on the Back and Shoulders
The Diagnosis: Prostate Cancer Metastases
Cutaneous involvement of visceral tumors can occur indirectly, with the tumor causing changes in the skin without the presence of tumor cells (ie, paraneoplastic processes), or directly, with the presence of tumor cells in the skin (ie, metastasis).1 Cutaneous metastases from solid primary tumors are uncommon. The incidence of metastasis to the skin from visceral malignancies ranges from 0.3% to 9.0%.2 The most common primary internal tumors to metastasize to the skin are those arising from the breasts, lungs, colon, ovaries, and head and neck.3 Although prostate cancer is the most common cancer in males (making up 28% of new cancer diagnoses), excluding basal and squamous cell skin cancers, it rarely metastasizes to the skin, representing less than 1% of all cutaneous metastasis.4,5
There are 4 proposed mechanisms for metastatic dissemination to the skin: (1) direct invasion from underlying neoplasm; (2) implantation from a surgical scar; (3) spread through the lymphatics; and/or (4) hematogenous spread. The most common sites of prostate cancer cutaneous metastasis are the inguinal area, penis, and lower abdomen,2 which is likely due to spread via the lymphatic drainage. Cutaneous metastasis to distant sites such as the scalp or chest, as in our patient, may be secondary to hematogenous spread.
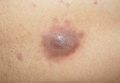
Generally, cutaneous metastases from visceral malignancies manifest as urticaria or a nonspecific macular rash.2 However, prostate cancer cutaneous metastases commonly present as papules or subcutaneous nodules but also can have inflammatory, cicatricial, sclerodermoid, telangiectatic, or zosteriform morphologies.2,5,6 The lesions of prostate cancer cutaneous metastases are typically well-circumscribed, flesh-colored or pink to red, oval plaques or nodules ranging in size from several millimeters to a few centimeters.2 The cutaneous lesions generally are multiple and can be localized or diffuse in distribution. The clinical differential diagnosis is broad in patients undergoing systemic treatment of prostate cancer and often includes drug reaction, cutaneous lymphoma, atypical mycobacterial or deep fungal infection, or paraneoplastic dermatosis.
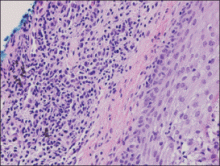
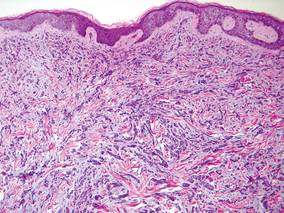
Laboratory studies often reveal an elevated serum prostate-specific antigen (PSA) level, a glycoprotein that functions in the liquefaction of seminal fluid made primarily by the prostate, often in benign hypertrophic and malignant prostatic processes.7 Definitive diagnosis of prostate cancer cutaneous metastasis can be established by histologic examination. Excisional or punch biopsy is preferred over superficial shave biopsy. Metastasis from prostate adenocarcinoma often reveals infiltrative growth of disorganized atypical epithelial cells along collagen bundles in the dermis and subcutis (Figure 1).2,8 The presence of lymphovascular invasion further increases the suspicion of metastasis. Metastatic lesions may resemble the primary lesion; however, they are often poorly differentiated, making histologic comparison difficult.
|
| Figure 1. Biopsy revealed cords of atypical epithelioid cells dissecting through dermal collagen bundles, accompanied by abundant mucin. Atypical cells displayed nuclear pleomorphism, and multiple mitotic figures were present (H&E, original magnification ×100). |
|
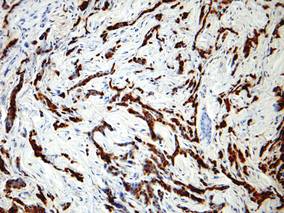
| Figure 2. Immunohistochemistry was strongly positive for prostate-specific antigen and focally positive for prostate-specific acid phosphatase (original magnification ×100). |
The diagnosis is confirmed immunohistochemically with PSA and/or prostate-specific acid phosphatase immunostaining (Figure 2), which together have nearly 100% specificity for prostate adenocarcinomas.9 Although rare, PSA and or prostate-specific acid phosphatase may be weakly positive in nephrogenic adenoma.10 Another biomarker, urinary prostate cancer antigen 3, is being evaluated to identify men with indolent prostate cancer.11 It is unknown how useful this marker will be in the immunohistochemical identification of prostate cancer cutaneous metastases.
A large review of reported cases of cutaneous metastases from internal malignancies revealed that the majority of patients had known systemic disease but enjoyed a good performance status at the time of diagnosis.2 As such, skin metastases may serve as the initial indicator of visceral recurrence. Prostate cancer is second only to lung cancer as the deadliest cancer in males,4 and cutaneous metastasis garners a particularly poor prognosis. The mean survival time after diagnosis of cutaneous metastasis is 7 months.8 Therefore, treatment of cutaneous metastases is largely palliative, including local excision and intralesional chemotherapy.
1. Thiers BH, Sahn RE, Callen JP. Cutaneous manifestations of internal malignancy. CA Cancer J Clin 2009;59:73-98.
2. Mueller TJ, Wu H, Greenberg RE, et al. Cutaneous metastases from genitourinary malignancies. Urology. 2004;63:1021-1026.
3. Brownstein MH, Helwig EB. Metastatic tumors of the skin. Cancer. 1972;29:1298-1307.
4. Jemal A, Siegel R, Xu J, et al. Cancer statistics, 2010. CA Cancer J Clin. 2010;60:277-300.
5. Schwartz RA. Cutaneous metastatic disease. J Am Acad Dermatol. 1995;33:161-182.
6. Reddy S, Bang RH, Contreras ME. Telangiectatic cutaneous metastasis from carcinoma of the prostate. Br J Dermatol. 2007;156:598-600.
7. Tosoian J, Loeb S. PSA and beyond: the past, present, and future of investigative biomarkers for prostate cancer. ScientificWorldJournal. 2010;10:1919-1931.
8. Wang SQ, Mecca PS, Myskowski PL, et al. Scrotal and penile papules and plaques as the initial manifestation of a cutaneous metastasis of adenocarcinoma of the prostate: case report and review of the literature. J Cutan Pathol. 2008;35:681-684.
9. Nadji M, Tabei SZ, Castro A, et al. Prostate-specific antigen: an immunohistochemical marker for prostatic neoplasms. Cancer. 1981;48:1229-1232.
10. Paner GP, Luthringer DJ, Amin MB. Best practice in diagnostic immunohistochemistry: prostate carcinoma and its mimics in needle core biopsies. Arch Pathol Lab Med. 2008;132:1388-1396.
11. Cooperberg MR, Carroll PR, Klotz L. Active surveillance for prostate cancer: progress and promise. J Clin Oncol. 2011;29:3669-3676.
The Diagnosis: Prostate Cancer Metastases
Cutaneous involvement of visceral tumors can occur indirectly, with the tumor causing changes in the skin without the presence of tumor cells (ie, paraneoplastic processes), or directly, with the presence of tumor cells in the skin (ie, metastasis).1 Cutaneous metastases from solid primary tumors are uncommon. The incidence of metastasis to the skin from visceral malignancies ranges from 0.3% to 9.0%.2 The most common primary internal tumors to metastasize to the skin are those arising from the breasts, lungs, colon, ovaries, and head and neck.3 Although prostate cancer is the most common cancer in males (making up 28% of new cancer diagnoses), excluding basal and squamous cell skin cancers, it rarely metastasizes to the skin, representing less than 1% of all cutaneous metastasis.4,5
There are 4 proposed mechanisms for metastatic dissemination to the skin: (1) direct invasion from underlying neoplasm; (2) implantation from a surgical scar; (3) spread through the lymphatics; and/or (4) hematogenous spread. The most common sites of prostate cancer cutaneous metastasis are the inguinal area, penis, and lower abdomen,2 which is likely due to spread via the lymphatic drainage. Cutaneous metastasis to distant sites such as the scalp or chest, as in our patient, may be secondary to hematogenous spread.
Generally, cutaneous metastases from visceral malignancies manifest as urticaria or a nonspecific macular rash.2 However, prostate cancer cutaneous metastases commonly present as papules or subcutaneous nodules but also can have inflammatory, cicatricial, sclerodermoid, telangiectatic, or zosteriform morphologies.2,5,6 The lesions of prostate cancer cutaneous metastases are typically well-circumscribed, flesh-colored or pink to red, oval plaques or nodules ranging in size from several millimeters to a few centimeters.2 The cutaneous lesions generally are multiple and can be localized or diffuse in distribution. The clinical differential diagnosis is broad in patients undergoing systemic treatment of prostate cancer and often includes drug reaction, cutaneous lymphoma, atypical mycobacterial or deep fungal infection, or paraneoplastic dermatosis.
Laboratory studies often reveal an elevated serum prostate-specific antigen (PSA) level, a glycoprotein that functions in the liquefaction of seminal fluid made primarily by the prostate, often in benign hypertrophic and malignant prostatic processes.7 Definitive diagnosis of prostate cancer cutaneous metastasis can be established by histologic examination. Excisional or punch biopsy is preferred over superficial shave biopsy. Metastasis from prostate adenocarcinoma often reveals infiltrative growth of disorganized atypical epithelial cells along collagen bundles in the dermis and subcutis (Figure 1).2,8 The presence of lymphovascular invasion further increases the suspicion of metastasis. Metastatic lesions may resemble the primary lesion; however, they are often poorly differentiated, making histologic comparison difficult.

|
| Figure 1. Biopsy revealed cords of atypical epithelioid cells dissecting through dermal collagen bundles, accompanied by abundant mucin. Atypical cells displayed nuclear pleomorphism, and multiple mitotic figures were present (H&E, original magnification ×100). |

|
| Figure 2. Immunohistochemistry was strongly positive for prostate-specific antigen and focally positive for prostate-specific acid phosphatase (original magnification ×100). |
The diagnosis is confirmed immunohistochemically with PSA and/or prostate-specific acid phosphatase immunostaining (Figure 2), which together have nearly 100% specificity for prostate adenocarcinomas.9 Although rare, PSA and or prostate-specific acid phosphatase may be weakly positive in nephrogenic adenoma.10 Another biomarker, urinary prostate cancer antigen 3, is being evaluated to identify men with indolent prostate cancer.11 It is unknown how useful this marker will be in the immunohistochemical identification of prostate cancer cutaneous metastases.
A large review of reported cases of cutaneous metastases from internal malignancies revealed that the majority of patients had known systemic disease but enjoyed a good performance status at the time of diagnosis.2 As such, skin metastases may serve as the initial indicator of visceral recurrence. Prostate cancer is second only to lung cancer as the deadliest cancer in males,4 and cutaneous metastasis garners a particularly poor prognosis. The mean survival time after diagnosis of cutaneous metastasis is 7 months.8 Therefore, treatment of cutaneous metastases is largely palliative, including local excision and intralesional chemotherapy.
The Diagnosis: Prostate Cancer Metastases
Cutaneous involvement of visceral tumors can occur indirectly, with the tumor causing changes in the skin without the presence of tumor cells (ie, paraneoplastic processes), or directly, with the presence of tumor cells in the skin (ie, metastasis).1 Cutaneous metastases from solid primary tumors are uncommon. The incidence of metastasis to the skin from visceral malignancies ranges from 0.3% to 9.0%.2 The most common primary internal tumors to metastasize to the skin are those arising from the breasts, lungs, colon, ovaries, and head and neck.3 Although prostate cancer is the most common cancer in males (making up 28% of new cancer diagnoses), excluding basal and squamous cell skin cancers, it rarely metastasizes to the skin, representing less than 1% of all cutaneous metastasis.4,5
There are 4 proposed mechanisms for metastatic dissemination to the skin: (1) direct invasion from underlying neoplasm; (2) implantation from a surgical scar; (3) spread through the lymphatics; and/or (4) hematogenous spread. The most common sites of prostate cancer cutaneous metastasis are the inguinal area, penis, and lower abdomen,2 which is likely due to spread via the lymphatic drainage. Cutaneous metastasis to distant sites such as the scalp or chest, as in our patient, may be secondary to hematogenous spread.
Generally, cutaneous metastases from visceral malignancies manifest as urticaria or a nonspecific macular rash.2 However, prostate cancer cutaneous metastases commonly present as papules or subcutaneous nodules but also can have inflammatory, cicatricial, sclerodermoid, telangiectatic, or zosteriform morphologies.2,5,6 The lesions of prostate cancer cutaneous metastases are typically well-circumscribed, flesh-colored or pink to red, oval plaques or nodules ranging in size from several millimeters to a few centimeters.2 The cutaneous lesions generally are multiple and can be localized or diffuse in distribution. The clinical differential diagnosis is broad in patients undergoing systemic treatment of prostate cancer and often includes drug reaction, cutaneous lymphoma, atypical mycobacterial or deep fungal infection, or paraneoplastic dermatosis.
Laboratory studies often reveal an elevated serum prostate-specific antigen (PSA) level, a glycoprotein that functions in the liquefaction of seminal fluid made primarily by the prostate, often in benign hypertrophic and malignant prostatic processes.7 Definitive diagnosis of prostate cancer cutaneous metastasis can be established by histologic examination. Excisional or punch biopsy is preferred over superficial shave biopsy. Metastasis from prostate adenocarcinoma often reveals infiltrative growth of disorganized atypical epithelial cells along collagen bundles in the dermis and subcutis (Figure 1).2,8 The presence of lymphovascular invasion further increases the suspicion of metastasis. Metastatic lesions may resemble the primary lesion; however, they are often poorly differentiated, making histologic comparison difficult.

|
| Figure 1. Biopsy revealed cords of atypical epithelioid cells dissecting through dermal collagen bundles, accompanied by abundant mucin. Atypical cells displayed nuclear pleomorphism, and multiple mitotic figures were present (H&E, original magnification ×100). |

|
| Figure 2. Immunohistochemistry was strongly positive for prostate-specific antigen and focally positive for prostate-specific acid phosphatase (original magnification ×100). |
The diagnosis is confirmed immunohistochemically with PSA and/or prostate-specific acid phosphatase immunostaining (Figure 2), which together have nearly 100% specificity for prostate adenocarcinomas.9 Although rare, PSA and or prostate-specific acid phosphatase may be weakly positive in nephrogenic adenoma.10 Another biomarker, urinary prostate cancer antigen 3, is being evaluated to identify men with indolent prostate cancer.11 It is unknown how useful this marker will be in the immunohistochemical identification of prostate cancer cutaneous metastases.
A large review of reported cases of cutaneous metastases from internal malignancies revealed that the majority of patients had known systemic disease but enjoyed a good performance status at the time of diagnosis.2 As such, skin metastases may serve as the initial indicator of visceral recurrence. Prostate cancer is second only to lung cancer as the deadliest cancer in males,4 and cutaneous metastasis garners a particularly poor prognosis. The mean survival time after diagnosis of cutaneous metastasis is 7 months.8 Therefore, treatment of cutaneous metastases is largely palliative, including local excision and intralesional chemotherapy.
1. Thiers BH, Sahn RE, Callen JP. Cutaneous manifestations of internal malignancy. CA Cancer J Clin 2009;59:73-98.
2. Mueller TJ, Wu H, Greenberg RE, et al. Cutaneous metastases from genitourinary malignancies. Urology. 2004;63:1021-1026.
3. Brownstein MH, Helwig EB. Metastatic tumors of the skin. Cancer. 1972;29:1298-1307.
4. Jemal A, Siegel R, Xu J, et al. Cancer statistics, 2010. CA Cancer J Clin. 2010;60:277-300.
5. Schwartz RA. Cutaneous metastatic disease. J Am Acad Dermatol. 1995;33:161-182.
6. Reddy S, Bang RH, Contreras ME. Telangiectatic cutaneous metastasis from carcinoma of the prostate. Br J Dermatol. 2007;156:598-600.
7. Tosoian J, Loeb S. PSA and beyond: the past, present, and future of investigative biomarkers for prostate cancer. ScientificWorldJournal. 2010;10:1919-1931.
8. Wang SQ, Mecca PS, Myskowski PL, et al. Scrotal and penile papules and plaques as the initial manifestation of a cutaneous metastasis of adenocarcinoma of the prostate: case report and review of the literature. J Cutan Pathol. 2008;35:681-684.
9. Nadji M, Tabei SZ, Castro A, et al. Prostate-specific antigen: an immunohistochemical marker for prostatic neoplasms. Cancer. 1981;48:1229-1232.
10. Paner GP, Luthringer DJ, Amin MB. Best practice in diagnostic immunohistochemistry: prostate carcinoma and its mimics in needle core biopsies. Arch Pathol Lab Med. 2008;132:1388-1396.
11. Cooperberg MR, Carroll PR, Klotz L. Active surveillance for prostate cancer: progress and promise. J Clin Oncol. 2011;29:3669-3676.
1. Thiers BH, Sahn RE, Callen JP. Cutaneous manifestations of internal malignancy. CA Cancer J Clin 2009;59:73-98.
2. Mueller TJ, Wu H, Greenberg RE, et al. Cutaneous metastases from genitourinary malignancies. Urology. 2004;63:1021-1026.
3. Brownstein MH, Helwig EB. Metastatic tumors of the skin. Cancer. 1972;29:1298-1307.
4. Jemal A, Siegel R, Xu J, et al. Cancer statistics, 2010. CA Cancer J Clin. 2010;60:277-300.
5. Schwartz RA. Cutaneous metastatic disease. J Am Acad Dermatol. 1995;33:161-182.
6. Reddy S, Bang RH, Contreras ME. Telangiectatic cutaneous metastasis from carcinoma of the prostate. Br J Dermatol. 2007;156:598-600.
7. Tosoian J, Loeb S. PSA and beyond: the past, present, and future of investigative biomarkers for prostate cancer. ScientificWorldJournal. 2010;10:1919-1931.
8. Wang SQ, Mecca PS, Myskowski PL, et al. Scrotal and penile papules and plaques as the initial manifestation of a cutaneous metastasis of adenocarcinoma of the prostate: case report and review of the literature. J Cutan Pathol. 2008;35:681-684.
9. Nadji M, Tabei SZ, Castro A, et al. Prostate-specific antigen: an immunohistochemical marker for prostatic neoplasms. Cancer. 1981;48:1229-1232.
10. Paner GP, Luthringer DJ, Amin MB. Best practice in diagnostic immunohistochemistry: prostate carcinoma and its mimics in needle core biopsies. Arch Pathol Lab Med. 2008;132:1388-1396.
11. Cooperberg MR, Carroll PR, Klotz L. Active surveillance for prostate cancer: progress and promise. J Clin Oncol. 2011;29:3669-3676.
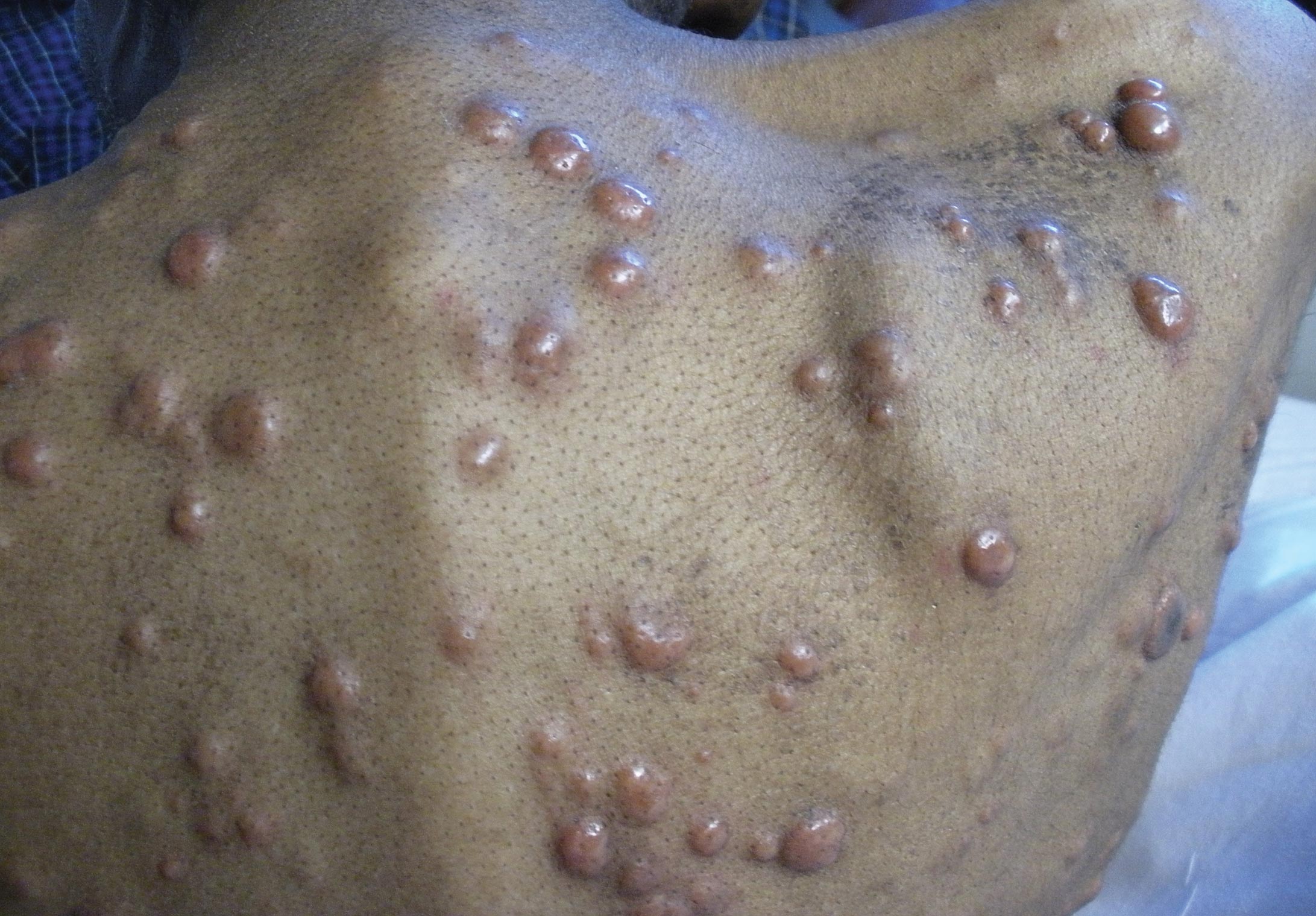
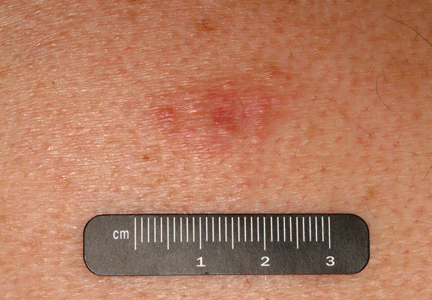
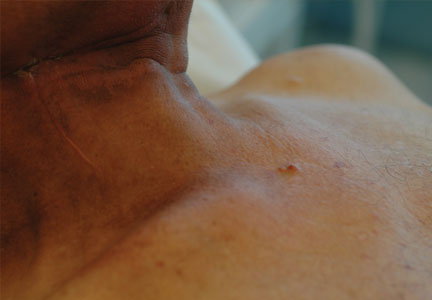
A 57-year-old cachectic man with a history of metastatic, hormone-refractory adenocarcinoma of the prostate presented with multiple tender subcutaneous nodules on the back and shoulders that developed over the course of 12 months. During that time, he was treated with cyclophosphamide, leuprolide acetate, bicalutamide, zoledronic acid, and filgrastim. A punch biopsy specimen obtained from the left shoulder revealed cords of atypical epithelioid cells dissecting through dermal collagen bundles, accompanied by abundant mucin. The atypical cells displayed nuclear pleomorphism, and multiple mitotic figures were observed.
Primary Mucinous Carcinoma of the Skin
To the Editor:
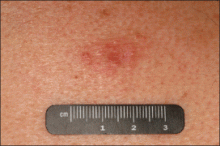
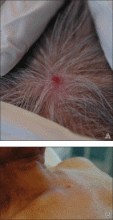
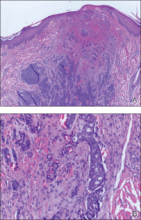
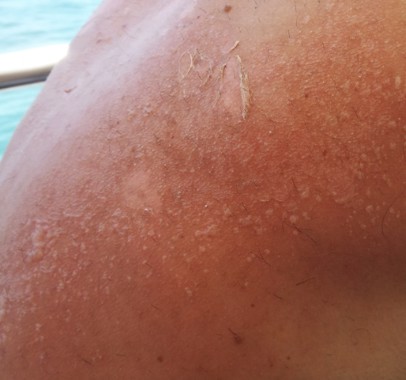
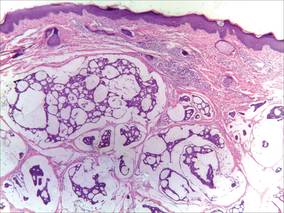
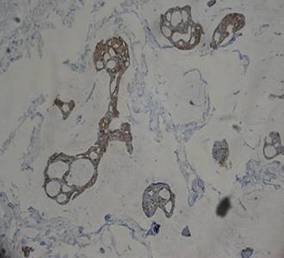
A 41-year-old man presented to our dermatology clinic with a recurrent asymptomatic nodule on the right cheek that had gradually increased in size over 1 year. The patient underwent laser excision at an outside facility 1 year after the first presentation. Pathology reports and tissue cultures from the excision were not available. The lesion recurred in the same location 6 months following excision. The patient reported no history of pain, fever, cough, weight loss, or loss of appetite, and he denied any trauma or radiation to the affected area. Dermatologic examination revealed a 24×18-mm, slightly elevated, dome-shaped, translucent, pink-colored tumor with telangiectasia on the right cheek (Figure 1). There was no tenderness or pruritus. Clinical examination and extensive radiographic studies revealed no primary disease. A complete blood cell count, biochemical tests, and serous tumor markers were within reference range. The lesion was resected with a 2-mm margin. The margins were free of tumor cells. Histopathology showed a circumscribed tumor with large amounts of mucin compartmentalized by fibrous septa and scattered floating islands of tumor cells in the dermis. Small-sized glands were organized in some areas of the tumor cells (Figure 2). Epithelial tumor cell islands contained uniform oval nuclei and focally vacuolated cytoplasm. The tumor cells were positive for cytokeratin 7 (CK7) (Figure 3) and negative for cytokeratin 20 (CK20), carcinoembryonic antigen, homeobox transcription factor (CDX2), villin, mucus-associated peptides of the thyroid transcription factor (TTF1), and prostate-specific antigen, all indicating a diagnosis of primary mucinous carcinoma of the skin (PMCS). No local recurrence, regional lymph node involvement, or distant metastasis was observed after resection.

|
| Figure 1. A 24×18-mm, slightly elevated, dome-shaped, translucent, pink-colored tumor with telangiectasia on the right cheek. |
|
| Figure 2. Histopathology showed a circumscribed tumor with large amounts of mucin compartmentalized by fibrous septa that contained scattered floating islands of tumor cells in the dermis (H&E, original magnification ×40). |
|
| Figure 3. The tumor cells were positive for cytokeratin 7 (original magnification ×400). |
Primary mucinous carcinoma of the skin is a rare malignant adnexal tumor. It was first described by Lennox et al1 in 1952 and was later given its name by Mendoza and Helwig2 through summarizing clinical and histochemical characteristics based on the observation of 14 cases. A PubMed search of articles indexed for MEDLINE using the search terms adenocarcinoma, mucinous (Medical Subject Headings); mucinous adenocarcinoma (title/abstract); colloid carcinoma* (title/abstract); skin neoplasms (Medical Subject Headings); skin neoplasm (title/abstract); skin cancer* (title/abstract), and primary (title/abstract) revealed more than 200 reported cases of PMCS. It usually is located in the head and neck region, with the eyelids being the most common site of presentation, but rare presentations in the gluteal region, axillae, hemithoraxes, vulva, arms, and legs also have been reported.3-6 Primary mucinous carcinoma of the skin mainly affects elderly patients (age range, 61–80 years), with men being affected more often than women. It often presents as a painless, solitary, nontender, sometimes ulcerated, translucent, white or reddish nodule. The tumors generally grow slowly with high rates of local recurrence and rare chances of distant metastasis.7 Most metastases involve the regional lymph nodes and lungs, but rare cases of bone marrow and parotid gland metastases also have been reported.8 Because PMCS is an indolent tumor, which may be mistaken for a benign tumor, it is not always histologically examined on initial presentation.
Primary mucinous carcinoma of the skin is characterized histopathologically by floating epithelial cell nests in mucinous lakes. It is unknown if this neoplasm shows eccrine- or apocrine-type differentiation. S-100 protein seems to indicate eccrine rather than apocrine differentiation.9 Misdiagnosis of PMCS is common, as it has an uncharacteristic gross appearance and may microscopically resemble cutaneous metastasis from a mucinous carcinoma of the breast, gastrointestinal tract, lungs, ovaries, or prostate. It is important to distinguish between metastatic tumors and PMCS because PMCS generally is more benign and has a better prognosis. A variety of stains have been reported as helpful in the diagnosis of PMCS, including CK7 and CK20, D2-40, p63, and others. In some reports, labeling for CK7 and CK20 was helpful in differentiating colon carcinoma from breast or skin malignancies but did not differentiate primary skin lesions from metastasis of breast malignancies to the skin.10-13 Paradela et al5 proposed D2-40 as a reliable immunostain to detect lymphatic invasion in node-negative primary tumors. Kalebi and Hale11 and Ivan et al12 highlighted the importance of p63 in excluding metastasis to the skin, particularly from the gastrointestinal tract, pancreaticobiliary tree, and lungs.
Even though these factors may help in establishing the primary site of the tumor, a final diagnosis only can be drawn when the patient has been subjected to a thorough investigation. In our patient, clinical workup did not reveal other possible primary sites in other organs or evidence of metastasis from the skin lesion to other sites. Based on the histologic findings, the diagnosis of PMCS was made.
The recommended treatment of PMCS varies from standard excision to wide local excision including dissection of the regional lymph nodes. Alam et al14 reported that Mohs micrographic surgery may be appropriate for first-line therapy because the procedure gives complete margin control and spares tissue. Immunohistochemistry should be used as an adjunct to routine hematoxylin and eosin staining to aid in ensuring negative margins.15 Considering a high potential for recurrence following surgical excision, it is important to detect hormone receptors in this tumor because patients may be treated using antiestrogenic drugs such as tamoxifen. Moreover, recurrent or metastatic PMCS is both resistant to radiotherapy and unresponsive to chemotherapy.3 Annual follow-up is recommended due to the potential for recurrence or metastasis.
1. Lennox B, Pearse AG, Richards HG. Mucin-secreting tumours of the skin, with special reference to the so-called mixed-salivary tumour of the skin and its relation to hidradenoma. J Pathol Bacteriol. 1952;64:865-890.
2. Mendoza S, Helwig EB. Mucinous (adenocystic) carcinoma of the skin. Arch Dermatol. 1971;103:68-78.
3. Breiting L, Dahlstrøm K, Christensen L, et al. Primary mucinous carcinoma of the skin. Am J Dermatopathol. 2007;29:595-596.
4. Krishnamurthy J, Saba F, Sunila. Primary mucinous carcinoma of the skin: a rare tumor in the gluteal region. Indian J Pathol Microbiol. 2009;52:225-227.
5. Paradela S, Castiñeiras I, Cuevas J, et al. Mucinous carcinoma of the skin: evaluation of lymphatic invasion with D2-40. Am J Dermatopathol. 2008;30:504-508.
6. Laco J, Simáková E, Svobodová J, et al. Recurrent mucinous carcinoma of skin mimicking primary mucinous carcinoma of parotid gland: a diagnostic pitfall. Cesk Patol. 2009;45:79-82.
7. Breiting L, Christensen L, Dahlstrøm K, et al. Primary mucinous carcinoma of the skin: a population-based study. Int J Dermatol. 2008;47:242-245.
8. Ajithkumar TV, Nileena N, Abraham EK, et al. Bone marrow relapse in primary mucinous carcinoma of skin. Am J Clin Oncol. 1999;22:303-304.
9. Bellezza G, Sidoni A, Bucciarelli E. Primary mucinous carcinoma of the skin. Am J Dermatopathol. 2000;22:166-170.
10. Levy G, Finkelstein A, McNiff JM. Immunohistochemical techniques to compare primary vs. metastatic mucinous carcinoma of the skin [published online ahead of print September 24, 2009]. J Cutan Pathol. 2010;37:411-415.
11. Kalebi A, Hale M. Primary mucinous carcinoma of the skin: usefulness of p63 in excluding metastasis and first report of psammoma bodies. Am J Dermatopathol. 2008;30:510.
12. Ivan D, Nash JW, Prieto VG, et al. Use of p63 expression in distinguishing primary and metastatic cutaneous adnexal neoplasms from metastatic adenocarcinoma to skin. J Cutan Pathol. 2007;34:474-480.
13. Papalas JA, Proia AD. Primary mucinous carcinoma of the eyelid: a clinicopathologic and immunohistochemical study of 4 cases and an update on recurrence rates. Arch Ophthalmol. 2010;128:1160-1165.
14. Alam M, Trela R, Kim N, et al. Treatment of primary mucinous carcinoma of the skin: meta-analysis of 189 cases. J Invest Dermatol. 2009;129:382.
15. Stranahan D, Cherpelis BS, Glass LF, et al. Immunohistochemical stains in Mohs surgery: a review [published online ahead of print April 16, 2009]. Dermatol Surg. 2009;35:1023-1034.
To the Editor:
A 41-year-old man presented to our dermatology clinic with a recurrent asymptomatic nodule on the right cheek that had gradually increased in size over 1 year. The patient underwent laser excision at an outside facility 1 year after the first presentation. Pathology reports and tissue cultures from the excision were not available. The lesion recurred in the same location 6 months following excision. The patient reported no history of pain, fever, cough, weight loss, or loss of appetite, and he denied any trauma or radiation to the affected area. Dermatologic examination revealed a 24×18-mm, slightly elevated, dome-shaped, translucent, pink-colored tumor with telangiectasia on the right cheek (Figure 1). There was no tenderness or pruritus. Clinical examination and extensive radiographic studies revealed no primary disease. A complete blood cell count, biochemical tests, and serous tumor markers were within reference range. The lesion was resected with a 2-mm margin. The margins were free of tumor cells. Histopathology showed a circumscribed tumor with large amounts of mucin compartmentalized by fibrous septa and scattered floating islands of tumor cells in the dermis. Small-sized glands were organized in some areas of the tumor cells (Figure 2). Epithelial tumor cell islands contained uniform oval nuclei and focally vacuolated cytoplasm. The tumor cells were positive for cytokeratin 7 (CK7) (Figure 3) and negative for cytokeratin 20 (CK20), carcinoembryonic antigen, homeobox transcription factor (CDX2), villin, mucus-associated peptides of the thyroid transcription factor (TTF1), and prostate-specific antigen, all indicating a diagnosis of primary mucinous carcinoma of the skin (PMCS). No local recurrence, regional lymph node involvement, or distant metastasis was observed after resection.

|
| Figure 1. A 24×18-mm, slightly elevated, dome-shaped, translucent, pink-colored tumor with telangiectasia on the right cheek. |

|
| Figure 2. Histopathology showed a circumscribed tumor with large amounts of mucin compartmentalized by fibrous septa that contained scattered floating islands of tumor cells in the dermis (H&E, original magnification ×40). |

|
| Figure 3. The tumor cells were positive for cytokeratin 7 (original magnification ×400). |
Primary mucinous carcinoma of the skin is a rare malignant adnexal tumor. It was first described by Lennox et al1 in 1952 and was later given its name by Mendoza and Helwig2 through summarizing clinical and histochemical characteristics based on the observation of 14 cases. A PubMed search of articles indexed for MEDLINE using the search terms adenocarcinoma, mucinous (Medical Subject Headings); mucinous adenocarcinoma (title/abstract); colloid carcinoma* (title/abstract); skin neoplasms (Medical Subject Headings); skin neoplasm (title/abstract); skin cancer* (title/abstract), and primary (title/abstract) revealed more than 200 reported cases of PMCS. It usually is located in the head and neck region, with the eyelids being the most common site of presentation, but rare presentations in the gluteal region, axillae, hemithoraxes, vulva, arms, and legs also have been reported.3-6 Primary mucinous carcinoma of the skin mainly affects elderly patients (age range, 61–80 years), with men being affected more often than women. It often presents as a painless, solitary, nontender, sometimes ulcerated, translucent, white or reddish nodule. The tumors generally grow slowly with high rates of local recurrence and rare chances of distant metastasis.7 Most metastases involve the regional lymph nodes and lungs, but rare cases of bone marrow and parotid gland metastases also have been reported.8 Because PMCS is an indolent tumor, which may be mistaken for a benign tumor, it is not always histologically examined on initial presentation.
Primary mucinous carcinoma of the skin is characterized histopathologically by floating epithelial cell nests in mucinous lakes. It is unknown if this neoplasm shows eccrine- or apocrine-type differentiation. S-100 protein seems to indicate eccrine rather than apocrine differentiation.9 Misdiagnosis of PMCS is common, as it has an uncharacteristic gross appearance and may microscopically resemble cutaneous metastasis from a mucinous carcinoma of the breast, gastrointestinal tract, lungs, ovaries, or prostate. It is important to distinguish between metastatic tumors and PMCS because PMCS generally is more benign and has a better prognosis. A variety of stains have been reported as helpful in the diagnosis of PMCS, including CK7 and CK20, D2-40, p63, and others. In some reports, labeling for CK7 and CK20 was helpful in differentiating colon carcinoma from breast or skin malignancies but did not differentiate primary skin lesions from metastasis of breast malignancies to the skin.10-13 Paradela et al5 proposed D2-40 as a reliable immunostain to detect lymphatic invasion in node-negative primary tumors. Kalebi and Hale11 and Ivan et al12 highlighted the importance of p63 in excluding metastasis to the skin, particularly from the gastrointestinal tract, pancreaticobiliary tree, and lungs.
Even though these factors may help in establishing the primary site of the tumor, a final diagnosis only can be drawn when the patient has been subjected to a thorough investigation. In our patient, clinical workup did not reveal other possible primary sites in other organs or evidence of metastasis from the skin lesion to other sites. Based on the histologic findings, the diagnosis of PMCS was made.
The recommended treatment of PMCS varies from standard excision to wide local excision including dissection of the regional lymph nodes. Alam et al14 reported that Mohs micrographic surgery may be appropriate for first-line therapy because the procedure gives complete margin control and spares tissue. Immunohistochemistry should be used as an adjunct to routine hematoxylin and eosin staining to aid in ensuring negative margins.15 Considering a high potential for recurrence following surgical excision, it is important to detect hormone receptors in this tumor because patients may be treated using antiestrogenic drugs such as tamoxifen. Moreover, recurrent or metastatic PMCS is both resistant to radiotherapy and unresponsive to chemotherapy.3 Annual follow-up is recommended due to the potential for recurrence or metastasis.
To the Editor:
A 41-year-old man presented to our dermatology clinic with a recurrent asymptomatic nodule on the right cheek that had gradually increased in size over 1 year. The patient underwent laser excision at an outside facility 1 year after the first presentation. Pathology reports and tissue cultures from the excision were not available. The lesion recurred in the same location 6 months following excision. The patient reported no history of pain, fever, cough, weight loss, or loss of appetite, and he denied any trauma or radiation to the affected area. Dermatologic examination revealed a 24×18-mm, slightly elevated, dome-shaped, translucent, pink-colored tumor with telangiectasia on the right cheek (Figure 1). There was no tenderness or pruritus. Clinical examination and extensive radiographic studies revealed no primary disease. A complete blood cell count, biochemical tests, and serous tumor markers were within reference range. The lesion was resected with a 2-mm margin. The margins were free of tumor cells. Histopathology showed a circumscribed tumor with large amounts of mucin compartmentalized by fibrous septa and scattered floating islands of tumor cells in the dermis. Small-sized glands were organized in some areas of the tumor cells (Figure 2). Epithelial tumor cell islands contained uniform oval nuclei and focally vacuolated cytoplasm. The tumor cells were positive for cytokeratin 7 (CK7) (Figure 3) and negative for cytokeratin 20 (CK20), carcinoembryonic antigen, homeobox transcription factor (CDX2), villin, mucus-associated peptides of the thyroid transcription factor (TTF1), and prostate-specific antigen, all indicating a diagnosis of primary mucinous carcinoma of the skin (PMCS). No local recurrence, regional lymph node involvement, or distant metastasis was observed after resection.

|
| Figure 1. A 24×18-mm, slightly elevated, dome-shaped, translucent, pink-colored tumor with telangiectasia on the right cheek. |

|
| Figure 2. Histopathology showed a circumscribed tumor with large amounts of mucin compartmentalized by fibrous septa that contained scattered floating islands of tumor cells in the dermis (H&E, original magnification ×40). |

|
| Figure 3. The tumor cells were positive for cytokeratin 7 (original magnification ×400). |
Primary mucinous carcinoma of the skin is a rare malignant adnexal tumor. It was first described by Lennox et al1 in 1952 and was later given its name by Mendoza and Helwig2 through summarizing clinical and histochemical characteristics based on the observation of 14 cases. A PubMed search of articles indexed for MEDLINE using the search terms adenocarcinoma, mucinous (Medical Subject Headings); mucinous adenocarcinoma (title/abstract); colloid carcinoma* (title/abstract); skin neoplasms (Medical Subject Headings); skin neoplasm (title/abstract); skin cancer* (title/abstract), and primary (title/abstract) revealed more than 200 reported cases of PMCS. It usually is located in the head and neck region, with the eyelids being the most common site of presentation, but rare presentations in the gluteal region, axillae, hemithoraxes, vulva, arms, and legs also have been reported.3-6 Primary mucinous carcinoma of the skin mainly affects elderly patients (age range, 61–80 years), with men being affected more often than women. It often presents as a painless, solitary, nontender, sometimes ulcerated, translucent, white or reddish nodule. The tumors generally grow slowly with high rates of local recurrence and rare chances of distant metastasis.7 Most metastases involve the regional lymph nodes and lungs, but rare cases of bone marrow and parotid gland metastases also have been reported.8 Because PMCS is an indolent tumor, which may be mistaken for a benign tumor, it is not always histologically examined on initial presentation.
Primary mucinous carcinoma of the skin is characterized histopathologically by floating epithelial cell nests in mucinous lakes. It is unknown if this neoplasm shows eccrine- or apocrine-type differentiation. S-100 protein seems to indicate eccrine rather than apocrine differentiation.9 Misdiagnosis of PMCS is common, as it has an uncharacteristic gross appearance and may microscopically resemble cutaneous metastasis from a mucinous carcinoma of the breast, gastrointestinal tract, lungs, ovaries, or prostate. It is important to distinguish between metastatic tumors and PMCS because PMCS generally is more benign and has a better prognosis. A variety of stains have been reported as helpful in the diagnosis of PMCS, including CK7 and CK20, D2-40, p63, and others. In some reports, labeling for CK7 and CK20 was helpful in differentiating colon carcinoma from breast or skin malignancies but did not differentiate primary skin lesions from metastasis of breast malignancies to the skin.10-13 Paradela et al5 proposed D2-40 as a reliable immunostain to detect lymphatic invasion in node-negative primary tumors. Kalebi and Hale11 and Ivan et al12 highlighted the importance of p63 in excluding metastasis to the skin, particularly from the gastrointestinal tract, pancreaticobiliary tree, and lungs.
Even though these factors may help in establishing the primary site of the tumor, a final diagnosis only can be drawn when the patient has been subjected to a thorough investigation. In our patient, clinical workup did not reveal other possible primary sites in other organs or evidence of metastasis from the skin lesion to other sites. Based on the histologic findings, the diagnosis of PMCS was made.
The recommended treatment of PMCS varies from standard excision to wide local excision including dissection of the regional lymph nodes. Alam et al14 reported that Mohs micrographic surgery may be appropriate for first-line therapy because the procedure gives complete margin control and spares tissue. Immunohistochemistry should be used as an adjunct to routine hematoxylin and eosin staining to aid in ensuring negative margins.15 Considering a high potential for recurrence following surgical excision, it is important to detect hormone receptors in this tumor because patients may be treated using antiestrogenic drugs such as tamoxifen. Moreover, recurrent or metastatic PMCS is both resistant to radiotherapy and unresponsive to chemotherapy.3 Annual follow-up is recommended due to the potential for recurrence or metastasis.
1. Lennox B, Pearse AG, Richards HG. Mucin-secreting tumours of the skin, with special reference to the so-called mixed-salivary tumour of the skin and its relation to hidradenoma. J Pathol Bacteriol. 1952;64:865-890.
2. Mendoza S, Helwig EB. Mucinous (adenocystic) carcinoma of the skin. Arch Dermatol. 1971;103:68-78.
3. Breiting L, Dahlstrøm K, Christensen L, et al. Primary mucinous carcinoma of the skin. Am J Dermatopathol. 2007;29:595-596.
4. Krishnamurthy J, Saba F, Sunila. Primary mucinous carcinoma of the skin: a rare tumor in the gluteal region. Indian J Pathol Microbiol. 2009;52:225-227.
5. Paradela S, Castiñeiras I, Cuevas J, et al. Mucinous carcinoma of the skin: evaluation of lymphatic invasion with D2-40. Am J Dermatopathol. 2008;30:504-508.
6. Laco J, Simáková E, Svobodová J, et al. Recurrent mucinous carcinoma of skin mimicking primary mucinous carcinoma of parotid gland: a diagnostic pitfall. Cesk Patol. 2009;45:79-82.
7. Breiting L, Christensen L, Dahlstrøm K, et al. Primary mucinous carcinoma of the skin: a population-based study. Int J Dermatol. 2008;47:242-245.
8. Ajithkumar TV, Nileena N, Abraham EK, et al. Bone marrow relapse in primary mucinous carcinoma of skin. Am J Clin Oncol. 1999;22:303-304.
9. Bellezza G, Sidoni A, Bucciarelli E. Primary mucinous carcinoma of the skin. Am J Dermatopathol. 2000;22:166-170.
10. Levy G, Finkelstein A, McNiff JM. Immunohistochemical techniques to compare primary vs. metastatic mucinous carcinoma of the skin [published online ahead of print September 24, 2009]. J Cutan Pathol. 2010;37:411-415.
11. Kalebi A, Hale M. Primary mucinous carcinoma of the skin: usefulness of p63 in excluding metastasis and first report of psammoma bodies. Am J Dermatopathol. 2008;30:510.
12. Ivan D, Nash JW, Prieto VG, et al. Use of p63 expression in distinguishing primary and metastatic cutaneous adnexal neoplasms from metastatic adenocarcinoma to skin. J Cutan Pathol. 2007;34:474-480.
13. Papalas JA, Proia AD. Primary mucinous carcinoma of the eyelid: a clinicopathologic and immunohistochemical study of 4 cases and an update on recurrence rates. Arch Ophthalmol. 2010;128:1160-1165.
14. Alam M, Trela R, Kim N, et al. Treatment of primary mucinous carcinoma of the skin: meta-analysis of 189 cases. J Invest Dermatol. 2009;129:382.
15. Stranahan D, Cherpelis BS, Glass LF, et al. Immunohistochemical stains in Mohs surgery: a review [published online ahead of print April 16, 2009]. Dermatol Surg. 2009;35:1023-1034.
1. Lennox B, Pearse AG, Richards HG. Mucin-secreting tumours of the skin, with special reference to the so-called mixed-salivary tumour of the skin and its relation to hidradenoma. J Pathol Bacteriol. 1952;64:865-890.
2. Mendoza S, Helwig EB. Mucinous (adenocystic) carcinoma of the skin. Arch Dermatol. 1971;103:68-78.
3. Breiting L, Dahlstrøm K, Christensen L, et al. Primary mucinous carcinoma of the skin. Am J Dermatopathol. 2007;29:595-596.
4. Krishnamurthy J, Saba F, Sunila. Primary mucinous carcinoma of the skin: a rare tumor in the gluteal region. Indian J Pathol Microbiol. 2009;52:225-227.
5. Paradela S, Castiñeiras I, Cuevas J, et al. Mucinous carcinoma of the skin: evaluation of lymphatic invasion with D2-40. Am J Dermatopathol. 2008;30:504-508.
6. Laco J, Simáková E, Svobodová J, et al. Recurrent mucinous carcinoma of skin mimicking primary mucinous carcinoma of parotid gland: a diagnostic pitfall. Cesk Patol. 2009;45:79-82.
7. Breiting L, Christensen L, Dahlstrøm K, et al. Primary mucinous carcinoma of the skin: a population-based study. Int J Dermatol. 2008;47:242-245.
8. Ajithkumar TV, Nileena N, Abraham EK, et al. Bone marrow relapse in primary mucinous carcinoma of skin. Am J Clin Oncol. 1999;22:303-304.
9. Bellezza G, Sidoni A, Bucciarelli E. Primary mucinous carcinoma of the skin. Am J Dermatopathol. 2000;22:166-170.
10. Levy G, Finkelstein A, McNiff JM. Immunohistochemical techniques to compare primary vs. metastatic mucinous carcinoma of the skin [published online ahead of print September 24, 2009]. J Cutan Pathol. 2010;37:411-415.
11. Kalebi A, Hale M. Primary mucinous carcinoma of the skin: usefulness of p63 in excluding metastasis and first report of psammoma bodies. Am J Dermatopathol. 2008;30:510.
12. Ivan D, Nash JW, Prieto VG, et al. Use of p63 expression in distinguishing primary and metastatic cutaneous adnexal neoplasms from metastatic adenocarcinoma to skin. J Cutan Pathol. 2007;34:474-480.
13. Papalas JA, Proia AD. Primary mucinous carcinoma of the eyelid: a clinicopathologic and immunohistochemical study of 4 cases and an update on recurrence rates. Arch Ophthalmol. 2010;128:1160-1165.
14. Alam M, Trela R, Kim N, et al. Treatment of primary mucinous carcinoma of the skin: meta-analysis of 189 cases. J Invest Dermatol. 2009;129:382.
15. Stranahan D, Cherpelis BS, Glass LF, et al. Immunohistochemical stains in Mohs surgery: a review [published online ahead of print April 16, 2009]. Dermatol Surg. 2009;35:1023-1034.
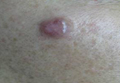
Pedunculated Scrotal Nodule in an Elderly Male: A Rare Presentation of Trichoblastoma
To the Editor:
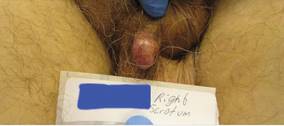
A 78-year-old man with no relevant dermatologic history presented for a routine skin examination. He had a large pedunculated nodule on the right scrotum that had been stable in size for more than 40 years. He reported the lesion had been diagnosed as a skin tag by his urologist 20 years prior and had remained asymptomatic in nature. On examination, a 2.5×1-cm, pedunculated, firm, rubbery nodule protruded from the anterior aspect of the right scrotal skin (Figure 1). Prominent telangiectases were noted on the surface of the lesion along with an irregular pigmented macule on the tip. There was no inguinal lymphadenopathy. Given the pigmentation at the tip of the lesion, he was advised to have it completely excised to rule out melanocytic atypia. The lesion subsequently was excised under local anesthesia and the resulting defect was primarily closed. A group of basaloid lobules within the dermis with scant stroma was noted (Figure 2). There was no epidermal connection and retraction artifact was absent. The epidermis appeared normal, except for basilar pigmentation corresponding to the pigmented macule at the tip of the lesion. Higher magnification of one of the basaloid islands demonstrated a papillary mesenchymal body (Figure 3). Immunohistochemistry with cytokeratin 20 showed positive staining of Merkel cells colonizing the basaloid islands (Figure 4). A diagnosis of trichoblastoma was rendered. Two nodular basal cell carcinomas (BCCs) were seen on initial presentation, but he remained free of recurrence of the scrotal tumor at 2-year follow-up.
|
Figure 1. Firm, pedunculated, flesh-colored nodule on the scrotum. |
|
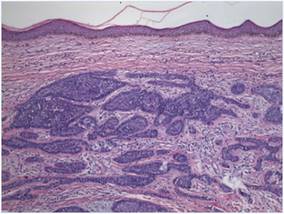
Figure 2. Basaloid tumor islands within the dermis with no epidermal connection (H&E, original magnification ×20). |
|
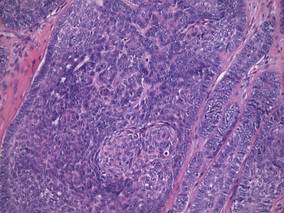
Figure 3. Papillary mesenchymal body adjacent to several dermal basaloid islands (H&E, original magnification ×40). |
|
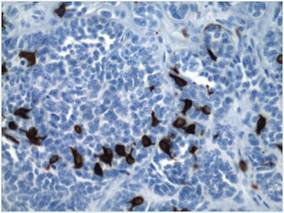
Figure 4. Cytokeratin 20 immunohistochemical staining indicated Merkel cells within the basaloid islands (original magnification ×40). |
Trichoblastoma is a rare benign tumor with differentiation toward the primitive hair follicle. The lesion typically presents as a slow-growing, solitary, well-circumscribed nodule that typically measures up to 3 cm and is predominantly located on the head and neck with a predilection for the scalp. Rarely the lesion will appear on the trunk, proximal extremities, or perianal and genital regions.1 Trichoblastomas show no gender predilection and predominantly affect adults between the fifth and seventh decades of life. In children it is the most common neoplasm arising in preexisting nevus sebaceous (Jadassohn nevus).2 Several clinical variants of trichoblastoma have been described, including giant trichoblastoma, pigmented trichoblastoma, and subcutaneous trichoblastoma, among others.3
Histologically, these tumors are characterized by well-circumscribed basaloid islands in the dermis with peripheral palisading closely resembling BCCs and trichoepitheliomas. Unlike BCCs, retraction artifact in trichoblastoma is a rare finding, while papillary mesenchymal bodies and an increased number of Merkel cells characterize both trichoblastomas and trichoepitheliomas, as in our patient.4 Other histologic features include lack of epidermal connection and location in the deeper dermis and/or subcutis. There also are reported cases with sebaceous, pigmented, rippled patterns and clear cell differentiation on histology.3,5
The genetic basis for trichoblastoma remains unclear. Although these primitive hair follicle tumors share histologic features with BCC and trichoepithelioma, which have both demonstrated PTCH (patched) mutations, no classic mutations in the PTCH gene were seen in 11 cases of trichoblastoma.6 In contrast, there have been reports of trichoblastoma occurring in Brooke-Spiegler syndrome and a case of a somatic CYLD (cylindromatosis) frameshift mutation in a trichoblastoma in a patient with Brooke-Spiegler syndrome7; however, the significance of CYLD mutation in thepathogenesis of trichoblastoma has not been fully elucidated.8
Trichoblastomas generally behave in an indolent asymptomatic manner, with malignant transformation being a rare occurrence.9 However, complete excision of these tumors is recommended given the potential for continued slow growth if incompletely removed. In the case of our patient, the lesion was completely excised at the outset. We plan to clinically monitor for evidence of disease recurrence as part of an annual full-body examination and have noted no evidence of recurrence in more than 2 years.
1. Brenn T, McKee PH. Tumors of the hair follicle. In: McKee PH, Calonje E, Granter SR, eds. Pathology of the Skin With Clinical Correlations. 3rd ed. London, England: Elsevier Mosby; 2005:1552-1557.
2. Jaqueti G, Requena L, Sánchez Yus E. Trichoblastoma is the most common neoplasm developed in nevus sebaceous of Jadassohn: a clinicopathologic study of series of 155 cases. Am J Dermatopathol. 2000;22:108-118.
3. Kim DW, Lee JH, Kim I. Giant melanotrichoblastoma. Am J Dermatopathol. 2011;33:e37-e40.
4. Schulz T, Hartschuh W. Merkel cells are absent in basal cell carcinomas but frequently found in trichoblastomas. an immunohistochemical study. J Cutan Pathol. 1997;24:14-24.
5. Swick BL, Baum CL, Walling HW. Rippled-patterntrichoblastoma with apocrine differentiation arising in a nevus sebaceus: report of a case and review of the literature. J Cutan Pathol. 2009;36:1200-1205.
6. Hafner C, Schmiemann V, Ruetten A, et al. PTCH mutations are not mainly involved in the pathogenesis of sporadic trichoblastomas [published online ahead of print June 26, 2007]. Hum Pathol. 2007;38:1496-1500.
7. Kazakov DV, Soukup R, Mukensnabl P, et al. Brooke-Spiegler syndrome: report of a case with combined lesions containing cylindromatous, spiradenomatous, trichoblastomatous, and sebaceous differentiation. Am J Dermatopathol. 2005;27:27-33.
8. Kazakov DV, Schaller J, Vanecek T, et al. Brooke-Spiegler syndrome: report of a case with a novel mutation in the CYLD gene and different types of somatic mutations in benign and malignant tumors [published online ahead of print February 4, 2010]. J Cutan Pathol. 2010;37:886-890.
9. Schulz T, Proske S, Hartschuh W, et al. High-grade trichoblastic carcinoma arising in trichoblastoma: a rare adnexal neoplasm often showing metastatic spread. Am J Dermatopathol. 2005;27:9-16
To the Editor:
A 78-year-old man with no relevant dermatologic history presented for a routine skin examination. He had a large pedunculated nodule on the right scrotum that had been stable in size for more than 40 years. He reported the lesion had been diagnosed as a skin tag by his urologist 20 years prior and had remained asymptomatic in nature. On examination, a 2.5×1-cm, pedunculated, firm, rubbery nodule protruded from the anterior aspect of the right scrotal skin (Figure 1). Prominent telangiectases were noted on the surface of the lesion along with an irregular pigmented macule on the tip. There was no inguinal lymphadenopathy. Given the pigmentation at the tip of the lesion, he was advised to have it completely excised to rule out melanocytic atypia. The lesion subsequently was excised under local anesthesia and the resulting defect was primarily closed. A group of basaloid lobules within the dermis with scant stroma was noted (Figure 2). There was no epidermal connection and retraction artifact was absent. The epidermis appeared normal, except for basilar pigmentation corresponding to the pigmented macule at the tip of the lesion. Higher magnification of one of the basaloid islands demonstrated a papillary mesenchymal body (Figure 3). Immunohistochemistry with cytokeratin 20 showed positive staining of Merkel cells colonizing the basaloid islands (Figure 4). A diagnosis of trichoblastoma was rendered. Two nodular basal cell carcinomas (BCCs) were seen on initial presentation, but he remained free of recurrence of the scrotal tumor at 2-year follow-up.

|
Figure 1. Firm, pedunculated, flesh-colored nodule on the scrotum. |

|
Figure 2. Basaloid tumor islands within the dermis with no epidermal connection (H&E, original magnification ×20). |

|
Figure 3. Papillary mesenchymal body adjacent to several dermal basaloid islands (H&E, original magnification ×40). |

|
Figure 4. Cytokeratin 20 immunohistochemical staining indicated Merkel cells within the basaloid islands (original magnification ×40). |
Trichoblastoma is a rare benign tumor with differentiation toward the primitive hair follicle. The lesion typically presents as a slow-growing, solitary, well-circumscribed nodule that typically measures up to 3 cm and is predominantly located on the head and neck with a predilection for the scalp. Rarely the lesion will appear on the trunk, proximal extremities, or perianal and genital regions.1 Trichoblastomas show no gender predilection and predominantly affect adults between the fifth and seventh decades of life. In children it is the most common neoplasm arising in preexisting nevus sebaceous (Jadassohn nevus).2 Several clinical variants of trichoblastoma have been described, including giant trichoblastoma, pigmented trichoblastoma, and subcutaneous trichoblastoma, among others.3
Histologically, these tumors are characterized by well-circumscribed basaloid islands in the dermis with peripheral palisading closely resembling BCCs and trichoepitheliomas. Unlike BCCs, retraction artifact in trichoblastoma is a rare finding, while papillary mesenchymal bodies and an increased number of Merkel cells characterize both trichoblastomas and trichoepitheliomas, as in our patient.4 Other histologic features include lack of epidermal connection and location in the deeper dermis and/or subcutis. There also are reported cases with sebaceous, pigmented, rippled patterns and clear cell differentiation on histology.3,5
The genetic basis for trichoblastoma remains unclear. Although these primitive hair follicle tumors share histologic features with BCC and trichoepithelioma, which have both demonstrated PTCH (patched) mutations, no classic mutations in the PTCH gene were seen in 11 cases of trichoblastoma.6 In contrast, there have been reports of trichoblastoma occurring in Brooke-Spiegler syndrome and a case of a somatic CYLD (cylindromatosis) frameshift mutation in a trichoblastoma in a patient with Brooke-Spiegler syndrome7; however, the significance of CYLD mutation in thepathogenesis of trichoblastoma has not been fully elucidated.8
Trichoblastomas generally behave in an indolent asymptomatic manner, with malignant transformation being a rare occurrence.9 However, complete excision of these tumors is recommended given the potential for continued slow growth if incompletely removed. In the case of our patient, the lesion was completely excised at the outset. We plan to clinically monitor for evidence of disease recurrence as part of an annual full-body examination and have noted no evidence of recurrence in more than 2 years.
To the Editor:
A 78-year-old man with no relevant dermatologic history presented for a routine skin examination. He had a large pedunculated nodule on the right scrotum that had been stable in size for more than 40 years. He reported the lesion had been diagnosed as a skin tag by his urologist 20 years prior and had remained asymptomatic in nature. On examination, a 2.5×1-cm, pedunculated, firm, rubbery nodule protruded from the anterior aspect of the right scrotal skin (Figure 1). Prominent telangiectases were noted on the surface of the lesion along with an irregular pigmented macule on the tip. There was no inguinal lymphadenopathy. Given the pigmentation at the tip of the lesion, he was advised to have it completely excised to rule out melanocytic atypia. The lesion subsequently was excised under local anesthesia and the resulting defect was primarily closed. A group of basaloid lobules within the dermis with scant stroma was noted (Figure 2). There was no epidermal connection and retraction artifact was absent. The epidermis appeared normal, except for basilar pigmentation corresponding to the pigmented macule at the tip of the lesion. Higher magnification of one of the basaloid islands demonstrated a papillary mesenchymal body (Figure 3). Immunohistochemistry with cytokeratin 20 showed positive staining of Merkel cells colonizing the basaloid islands (Figure 4). A diagnosis of trichoblastoma was rendered. Two nodular basal cell carcinomas (BCCs) were seen on initial presentation, but he remained free of recurrence of the scrotal tumor at 2-year follow-up.

|
Figure 1. Firm, pedunculated, flesh-colored nodule on the scrotum. |

|
Figure 2. Basaloid tumor islands within the dermis with no epidermal connection (H&E, original magnification ×20). |

|
Figure 3. Papillary mesenchymal body adjacent to several dermal basaloid islands (H&E, original magnification ×40). |

|
Figure 4. Cytokeratin 20 immunohistochemical staining indicated Merkel cells within the basaloid islands (original magnification ×40). |
Trichoblastoma is a rare benign tumor with differentiation toward the primitive hair follicle. The lesion typically presents as a slow-growing, solitary, well-circumscribed nodule that typically measures up to 3 cm and is predominantly located on the head and neck with a predilection for the scalp. Rarely the lesion will appear on the trunk, proximal extremities, or perianal and genital regions.1 Trichoblastomas show no gender predilection and predominantly affect adults between the fifth and seventh decades of life. In children it is the most common neoplasm arising in preexisting nevus sebaceous (Jadassohn nevus).2 Several clinical variants of trichoblastoma have been described, including giant trichoblastoma, pigmented trichoblastoma, and subcutaneous trichoblastoma, among others.3
Histologically, these tumors are characterized by well-circumscribed basaloid islands in the dermis with peripheral palisading closely resembling BCCs and trichoepitheliomas. Unlike BCCs, retraction artifact in trichoblastoma is a rare finding, while papillary mesenchymal bodies and an increased number of Merkel cells characterize both trichoblastomas and trichoepitheliomas, as in our patient.4 Other histologic features include lack of epidermal connection and location in the deeper dermis and/or subcutis. There also are reported cases with sebaceous, pigmented, rippled patterns and clear cell differentiation on histology.3,5
The genetic basis for trichoblastoma remains unclear. Although these primitive hair follicle tumors share histologic features with BCC and trichoepithelioma, which have both demonstrated PTCH (patched) mutations, no classic mutations in the PTCH gene were seen in 11 cases of trichoblastoma.6 In contrast, there have been reports of trichoblastoma occurring in Brooke-Spiegler syndrome and a case of a somatic CYLD (cylindromatosis) frameshift mutation in a trichoblastoma in a patient with Brooke-Spiegler syndrome7; however, the significance of CYLD mutation in thepathogenesis of trichoblastoma has not been fully elucidated.8
Trichoblastomas generally behave in an indolent asymptomatic manner, with malignant transformation being a rare occurrence.9 However, complete excision of these tumors is recommended given the potential for continued slow growth if incompletely removed. In the case of our patient, the lesion was completely excised at the outset. We plan to clinically monitor for evidence of disease recurrence as part of an annual full-body examination and have noted no evidence of recurrence in more than 2 years.
1. Brenn T, McKee PH. Tumors of the hair follicle. In: McKee PH, Calonje E, Granter SR, eds. Pathology of the Skin With Clinical Correlations. 3rd ed. London, England: Elsevier Mosby; 2005:1552-1557.
2. Jaqueti G, Requena L, Sánchez Yus E. Trichoblastoma is the most common neoplasm developed in nevus sebaceous of Jadassohn: a clinicopathologic study of series of 155 cases. Am J Dermatopathol. 2000;22:108-118.
3. Kim DW, Lee JH, Kim I. Giant melanotrichoblastoma. Am J Dermatopathol. 2011;33:e37-e40.
4. Schulz T, Hartschuh W. Merkel cells are absent in basal cell carcinomas but frequently found in trichoblastomas. an immunohistochemical study. J Cutan Pathol. 1997;24:14-24.
5. Swick BL, Baum CL, Walling HW. Rippled-patterntrichoblastoma with apocrine differentiation arising in a nevus sebaceus: report of a case and review of the literature. J Cutan Pathol. 2009;36:1200-1205.
6. Hafner C, Schmiemann V, Ruetten A, et al. PTCH mutations are not mainly involved in the pathogenesis of sporadic trichoblastomas [published online ahead of print June 26, 2007]. Hum Pathol. 2007;38:1496-1500.
7. Kazakov DV, Soukup R, Mukensnabl P, et al. Brooke-Spiegler syndrome: report of a case with combined lesions containing cylindromatous, spiradenomatous, trichoblastomatous, and sebaceous differentiation. Am J Dermatopathol. 2005;27:27-33.
8. Kazakov DV, Schaller J, Vanecek T, et al. Brooke-Spiegler syndrome: report of a case with a novel mutation in the CYLD gene and different types of somatic mutations in benign and malignant tumors [published online ahead of print February 4, 2010]. J Cutan Pathol. 2010;37:886-890.
9. Schulz T, Proske S, Hartschuh W, et al. High-grade trichoblastic carcinoma arising in trichoblastoma: a rare adnexal neoplasm often showing metastatic spread. Am J Dermatopathol. 2005;27:9-16
1. Brenn T, McKee PH. Tumors of the hair follicle. In: McKee PH, Calonje E, Granter SR, eds. Pathology of the Skin With Clinical Correlations. 3rd ed. London, England: Elsevier Mosby; 2005:1552-1557.
2. Jaqueti G, Requena L, Sánchez Yus E. Trichoblastoma is the most common neoplasm developed in nevus sebaceous of Jadassohn: a clinicopathologic study of series of 155 cases. Am J Dermatopathol. 2000;22:108-118.
3. Kim DW, Lee JH, Kim I. Giant melanotrichoblastoma. Am J Dermatopathol. 2011;33:e37-e40.
4. Schulz T, Hartschuh W. Merkel cells are absent in basal cell carcinomas but frequently found in trichoblastomas. an immunohistochemical study. J Cutan Pathol. 1997;24:14-24.
5. Swick BL, Baum CL, Walling HW. Rippled-patterntrichoblastoma with apocrine differentiation arising in a nevus sebaceus: report of a case and review of the literature. J Cutan Pathol. 2009;36:1200-1205.
6. Hafner C, Schmiemann V, Ruetten A, et al. PTCH mutations are not mainly involved in the pathogenesis of sporadic trichoblastomas [published online ahead of print June 26, 2007]. Hum Pathol. 2007;38:1496-1500.
7. Kazakov DV, Soukup R, Mukensnabl P, et al. Brooke-Spiegler syndrome: report of a case with combined lesions containing cylindromatous, spiradenomatous, trichoblastomatous, and sebaceous differentiation. Am J Dermatopathol. 2005;27:27-33.
8. Kazakov DV, Schaller J, Vanecek T, et al. Brooke-Spiegler syndrome: report of a case with a novel mutation in the CYLD gene and different types of somatic mutations in benign and malignant tumors [published online ahead of print February 4, 2010]. J Cutan Pathol. 2010;37:886-890.
9. Schulz T, Proske S, Hartschuh W, et al. High-grade trichoblastic carcinoma arising in trichoblastoma: a rare adnexal neoplasm often showing metastatic spread. Am J Dermatopathol. 2005;27:9-16
Primary Cutaneous Plasmacytoma
To the Editor:
Primary extramedullary plasmacytoma is a rare plasma cell neoplasm that occurs as an isolated finding without detectable underlying multiple myeloma. Uncommonly, it has been reported to occur in the skin as a primary cutaneous plasmacytoma (PCP). We describe a case of an asymptomatic plaque on the back that was found to contain a monoclonal proliferation of plasma cells without evidence of systemic disease despite thorough evaluation. We also include a brief discussion of PCPs, including diagnosis and treatment.
An indurated plaque was discovered on the upper back of a 57-year-old man during a routine follow-up visit for acne treatment. The patient reported the lesion had been present for approximately 2 years; it was asymptomatic and had not increased in size. A review of systems did not reveal any abnormalities. His medical history included hypercholesterolemia, hypertension, osteoarthritis, acne, type 2 diabetes mellitus, well-controlled herpes labialis, and gastroesophageal reflux disease. Physical examination revealed a nontender 2-cm erythematous plaque on the right side of the upper back (Figure 1). No other lesions or lymphadenopathy were noted. A biopsy was performed to rule out a primary neoplasm or metastatic lesion. Histopathologic examination of 3 separate punch biopsies revealed a perivascular infiltrate of lymphocytes and plasma cells in the papillary and superficial reticular dermis (Figure 2). On immunohistochemical staining, the plasma cells essentially were all κ positive, with rare λ-positive plasma cells (Figure 3).
The patient underwent complete evaluation for myeloma. A bone marrow biopsy was negative with no evidence of plasma cell dyscrasia. A skeletal survey showed no lytic lesions, and serum and urine protein electrophoreses were negative for M proteins. Urine immunofixation was negative for Bence Jones proteins. Computed tomography of the chest, abdomen, and pelvis showed no evidence of disease. Serum free k light chains were elevated at 32 mg/L (reference range, <19.4 mg/L), and IgA and b2-microglobulin also were elevated at 435 mg/dL (reference range, <400 mg/dL) and 2.8 mg/L (reference range, <1.8 mg/L), respectively. The k:λ ratio was slightly elevated at 2.2 (reference range, 0.26–1.65).
Local radiotherapy was considered but was rejected by the patient. A single 5-mg/cc intralesional injection of triamcinolone acetonide (1 cc total) was administered, which resulted in modest improvement but did not yield complete resolution. A repeat biopsy showed a monoclonal proliferation of plasma cells with areas of follicular mucinosis. Immunohistochemical studies also demonstrated CD3+ lymphocytes with more CD4+ cells than CD8+ cells and scattered CD20+ cells. Repeat serum and urine protein electrophoreses as well as bone marrow biopsy were negative or myeloma.
Because of the localized and recalcitrant nature of the neoplasm, the patient underwent wide local excision. Positron emission tomography performed 1 year following surgery demonstrated no residual disease. The patient has had no clinical recurrence but has demonstrated persistently elevated free k light chains. Seven years following the excision, there was no evidence of systemic disease.
Primary extramedullary plasmacytoma is a rare plasma cell neoplasm that occurs without detectable underlying multiple myeloma.1 Originally classified as a discrete entity, PCP currently is considered to be a variant of primary cutaneous marginal zone B-cell lymphoma.2 Cases of localization to the skin are thought to represent 2% to 4% of all primary extramedullary plasmacytomas.3 Very few cases of PCPs have been reported in the literature thus far.1 Primary cutaneous plasmacytomas are most commonly reported in Asian men, with an age range of 22 to 88 years; lesions typically present as slow-growing, reddish brown macules or plaques on the face, trunk, or extremities.4 They may be single or multiple, though solitary lesions are thought to portend a better prognosis. Associated features include lymphadenopathy, hepatosplenomegaly, hypergammaglobulinemia, and an elevated erythrocyte sedimentation rate.5 Multiple myeloma has been estimated to develop in one-third of patients.6 Primary cutaneous plasmacytoma also has been linked to organ transplantation and Epstein-Barr virus–associated proteins7 as well as recurrent herpes simplex virus infections.8
Diagnosis is based on a proliferation of a clonal k or λ light chain with the exclusion of multiple myeloma.9 Typical histologic findings include a diffuse or nodular dermal and subcuticular infiltrate of plasma cells, often with a grenz zone. Less commonly, an interstitial pattern predominates, mimicking a benign inflammatory process.4 The plasma cells may display varying degrees of maturation and atypia but no epidermotropism.10 On immunohistochemical staining, plasma cells stain positive for CD79a, CD34, and CD138, and also may stain positively for epithelial membrane antigen and vimentin.4
The differential diagnosis includes cutaneous plasmacytoma occurring in the setting of systemic disease, and reactive polyclonal processes occurring in the setting of chronic infection or autoimmune disease. Possible treatment options include intralesional steroids, psoralen plus UVA, topical immunomodulators,10 local radiotherapy,11 and excision. Prognosis is varied, with reported cases showing complete remission, local recurrence, and progression to systemic disease, with the development of multiple myeloma in one-third of cases.4 More aggressive cases are associated with multiple lesions, larger size, and IgA secretion by neoplastic cells.
Our patient with a solitary dermal infiltrate of monoclonal plasma cells did not have bone marrow involvement and had an unexplained elevated level of free k light chains in the serum. This unexplained relationship underscores the importance of continued surveillance in any cutaneous lymphoma.
1. Rongioletti F, Patterson JW, Rebora A. The histological and pathogenetic spectrum of cutaneous disease in monoclonal gammopathies. J Cutan Pathol. 2008;35:705-721.
2. Willemze R, Jaffe ES, Burg G, et al. WHO-EORTC classification for cutaneous lymphomas. Blood. 2005;105:3768-3785.
3. Muscardin LM, Pulsoni A, Cerroni L. Primary cutaneous plasmacytoma: report of a case with review of the literature. J Am Acad Dermatol. 2000;43(5, pt 2):962-965.
4. Kazakov DV, Belousova IE, Müller B, et al. Primary cutaneous plasmacytoma: a clinicopathological study of two cases with a long-term follow-up and review of the literature. J Cutan Pathol. 2002;29:244-248.
5. Aricò M, Bongiorno MR. Primary cutaneous plasmacytosis in a child. is this a new entity? J Eur Acad Dermatol. 2002;16:164-167.
6. Willoughby V, Werlang-Perurena A, Kelly A, et al. Primary cutaneous plasmacytoma (posttransplant lymphoproliferative disorder, plasmacytoma-like) in a heart transplant patient. Am J Dermatopathol. 2006;28:442-445.
7. Buffet M, Dupin N, Carlotti A, et al. Epstein-Barr virus–associated cutaneous plasmacytoma post–solid organ transplantation [in French]. Ann Dermatol Venereol. 2004;131:1085-1091.
8. Zendri E, Venturi C, Ricci R, et al. Primary cutaneous plasmacytoma: a role for a triggering stimulus? Clin Exp Dermatol. 2005;30:229-231.
9. Tüting T, Bork K. Primary plasmacytoma of the skin. J Am Acad Dermatol. 1996;34(2, pt 2):386-390.
10. Miura H, Itami S, Yoshikawa K. Treatment of facial lesion of cutaneous plasmacytosis with tacrolimus ointment. J Am Acad Dermatol. 2003;49:1195-1196.
11. Tessari G, Fabbian F, Colato C, et al. Primary cutaneous plasmacytoma after rejection of a transplanted kidney: case report and review of the literature. Int J Hematol. 2004;80:361-364.
To the Editor:
Primary extramedullary plasmacytoma is a rare plasma cell neoplasm that occurs as an isolated finding without detectable underlying multiple myeloma. Uncommonly, it has been reported to occur in the skin as a primary cutaneous plasmacytoma (PCP). We describe a case of an asymptomatic plaque on the back that was found to contain a monoclonal proliferation of plasma cells without evidence of systemic disease despite thorough evaluation. We also include a brief discussion of PCPs, including diagnosis and treatment.
An indurated plaque was discovered on the upper back of a 57-year-old man during a routine follow-up visit for acne treatment. The patient reported the lesion had been present for approximately 2 years; it was asymptomatic and had not increased in size. A review of systems did not reveal any abnormalities. His medical history included hypercholesterolemia, hypertension, osteoarthritis, acne, type 2 diabetes mellitus, well-controlled herpes labialis, and gastroesophageal reflux disease. Physical examination revealed a nontender 2-cm erythematous plaque on the right side of the upper back (Figure 1). No other lesions or lymphadenopathy were noted. A biopsy was performed to rule out a primary neoplasm or metastatic lesion. Histopathologic examination of 3 separate punch biopsies revealed a perivascular infiltrate of lymphocytes and plasma cells in the papillary and superficial reticular dermis (Figure 2). On immunohistochemical staining, the plasma cells essentially were all κ positive, with rare λ-positive plasma cells (Figure 3).
The patient underwent complete evaluation for myeloma. A bone marrow biopsy was negative with no evidence of plasma cell dyscrasia. A skeletal survey showed no lytic lesions, and serum and urine protein electrophoreses were negative for M proteins. Urine immunofixation was negative for Bence Jones proteins. Computed tomography of the chest, abdomen, and pelvis showed no evidence of disease. Serum free k light chains were elevated at 32 mg/L (reference range, <19.4 mg/L), and IgA and b2-microglobulin also were elevated at 435 mg/dL (reference range, <400 mg/dL) and 2.8 mg/L (reference range, <1.8 mg/L), respectively. The k:λ ratio was slightly elevated at 2.2 (reference range, 0.26–1.65).
Local radiotherapy was considered but was rejected by the patient. A single 5-mg/cc intralesional injection of triamcinolone acetonide (1 cc total) was administered, which resulted in modest improvement but did not yield complete resolution. A repeat biopsy showed a monoclonal proliferation of plasma cells with areas of follicular mucinosis. Immunohistochemical studies also demonstrated CD3+ lymphocytes with more CD4+ cells than CD8+ cells and scattered CD20+ cells. Repeat serum and urine protein electrophoreses as well as bone marrow biopsy were negative or myeloma.
Because of the localized and recalcitrant nature of the neoplasm, the patient underwent wide local excision. Positron emission tomography performed 1 year following surgery demonstrated no residual disease. The patient has had no clinical recurrence but has demonstrated persistently elevated free k light chains. Seven years following the excision, there was no evidence of systemic disease.
Primary extramedullary plasmacytoma is a rare plasma cell neoplasm that occurs without detectable underlying multiple myeloma.1 Originally classified as a discrete entity, PCP currently is considered to be a variant of primary cutaneous marginal zone B-cell lymphoma.2 Cases of localization to the skin are thought to represent 2% to 4% of all primary extramedullary plasmacytomas.3 Very few cases of PCPs have been reported in the literature thus far.1 Primary cutaneous plasmacytomas are most commonly reported in Asian men, with an age range of 22 to 88 years; lesions typically present as slow-growing, reddish brown macules or plaques on the face, trunk, or extremities.4 They may be single or multiple, though solitary lesions are thought to portend a better prognosis. Associated features include lymphadenopathy, hepatosplenomegaly, hypergammaglobulinemia, and an elevated erythrocyte sedimentation rate.5 Multiple myeloma has been estimated to develop in one-third of patients.6 Primary cutaneous plasmacytoma also has been linked to organ transplantation and Epstein-Barr virus–associated proteins7 as well as recurrent herpes simplex virus infections.8
Diagnosis is based on a proliferation of a clonal k or λ light chain with the exclusion of multiple myeloma.9 Typical histologic findings include a diffuse or nodular dermal and subcuticular infiltrate of plasma cells, often with a grenz zone. Less commonly, an interstitial pattern predominates, mimicking a benign inflammatory process.4 The plasma cells may display varying degrees of maturation and atypia but no epidermotropism.10 On immunohistochemical staining, plasma cells stain positive for CD79a, CD34, and CD138, and also may stain positively for epithelial membrane antigen and vimentin.4
The differential diagnosis includes cutaneous plasmacytoma occurring in the setting of systemic disease, and reactive polyclonal processes occurring in the setting of chronic infection or autoimmune disease. Possible treatment options include intralesional steroids, psoralen plus UVA, topical immunomodulators,10 local radiotherapy,11 and excision. Prognosis is varied, with reported cases showing complete remission, local recurrence, and progression to systemic disease, with the development of multiple myeloma in one-third of cases.4 More aggressive cases are associated with multiple lesions, larger size, and IgA secretion by neoplastic cells.
Our patient with a solitary dermal infiltrate of monoclonal plasma cells did not have bone marrow involvement and had an unexplained elevated level of free k light chains in the serum. This unexplained relationship underscores the importance of continued surveillance in any cutaneous lymphoma.
To the Editor:
Primary extramedullary plasmacytoma is a rare plasma cell neoplasm that occurs as an isolated finding without detectable underlying multiple myeloma. Uncommonly, it has been reported to occur in the skin as a primary cutaneous plasmacytoma (PCP). We describe a case of an asymptomatic plaque on the back that was found to contain a monoclonal proliferation of plasma cells without evidence of systemic disease despite thorough evaluation. We also include a brief discussion of PCPs, including diagnosis and treatment.
An indurated plaque was discovered on the upper back of a 57-year-old man during a routine follow-up visit for acne treatment. The patient reported the lesion had been present for approximately 2 years; it was asymptomatic and had not increased in size. A review of systems did not reveal any abnormalities. His medical history included hypercholesterolemia, hypertension, osteoarthritis, acne, type 2 diabetes mellitus, well-controlled herpes labialis, and gastroesophageal reflux disease. Physical examination revealed a nontender 2-cm erythematous plaque on the right side of the upper back (Figure 1). No other lesions or lymphadenopathy were noted. A biopsy was performed to rule out a primary neoplasm or metastatic lesion. Histopathologic examination of 3 separate punch biopsies revealed a perivascular infiltrate of lymphocytes and plasma cells in the papillary and superficial reticular dermis (Figure 2). On immunohistochemical staining, the plasma cells essentially were all κ positive, with rare λ-positive plasma cells (Figure 3).
The patient underwent complete evaluation for myeloma. A bone marrow biopsy was negative with no evidence of plasma cell dyscrasia. A skeletal survey showed no lytic lesions, and serum and urine protein electrophoreses were negative for M proteins. Urine immunofixation was negative for Bence Jones proteins. Computed tomography of the chest, abdomen, and pelvis showed no evidence of disease. Serum free k light chains were elevated at 32 mg/L (reference range, <19.4 mg/L), and IgA and b2-microglobulin also were elevated at 435 mg/dL (reference range, <400 mg/dL) and 2.8 mg/L (reference range, <1.8 mg/L), respectively. The k:λ ratio was slightly elevated at 2.2 (reference range, 0.26–1.65).
Local radiotherapy was considered but was rejected by the patient. A single 5-mg/cc intralesional injection of triamcinolone acetonide (1 cc total) was administered, which resulted in modest improvement but did not yield complete resolution. A repeat biopsy showed a monoclonal proliferation of plasma cells with areas of follicular mucinosis. Immunohistochemical studies also demonstrated CD3+ lymphocytes with more CD4+ cells than CD8+ cells and scattered CD20+ cells. Repeat serum and urine protein electrophoreses as well as bone marrow biopsy were negative or myeloma.
Because of the localized and recalcitrant nature of the neoplasm, the patient underwent wide local excision. Positron emission tomography performed 1 year following surgery demonstrated no residual disease. The patient has had no clinical recurrence but has demonstrated persistently elevated free k light chains. Seven years following the excision, there was no evidence of systemic disease.
Primary extramedullary plasmacytoma is a rare plasma cell neoplasm that occurs without detectable underlying multiple myeloma.1 Originally classified as a discrete entity, PCP currently is considered to be a variant of primary cutaneous marginal zone B-cell lymphoma.2 Cases of localization to the skin are thought to represent 2% to 4% of all primary extramedullary plasmacytomas.3 Very few cases of PCPs have been reported in the literature thus far.1 Primary cutaneous plasmacytomas are most commonly reported in Asian men, with an age range of 22 to 88 years; lesions typically present as slow-growing, reddish brown macules or plaques on the face, trunk, or extremities.4 They may be single or multiple, though solitary lesions are thought to portend a better prognosis. Associated features include lymphadenopathy, hepatosplenomegaly, hypergammaglobulinemia, and an elevated erythrocyte sedimentation rate.5 Multiple myeloma has been estimated to develop in one-third of patients.6 Primary cutaneous plasmacytoma also has been linked to organ transplantation and Epstein-Barr virus–associated proteins7 as well as recurrent herpes simplex virus infections.8
Diagnosis is based on a proliferation of a clonal k or λ light chain with the exclusion of multiple myeloma.9 Typical histologic findings include a diffuse or nodular dermal and subcuticular infiltrate of plasma cells, often with a grenz zone. Less commonly, an interstitial pattern predominates, mimicking a benign inflammatory process.4 The plasma cells may display varying degrees of maturation and atypia but no epidermotropism.10 On immunohistochemical staining, plasma cells stain positive for CD79a, CD34, and CD138, and also may stain positively for epithelial membrane antigen and vimentin.4
The differential diagnosis includes cutaneous plasmacytoma occurring in the setting of systemic disease, and reactive polyclonal processes occurring in the setting of chronic infection or autoimmune disease. Possible treatment options include intralesional steroids, psoralen plus UVA, topical immunomodulators,10 local radiotherapy,11 and excision. Prognosis is varied, with reported cases showing complete remission, local recurrence, and progression to systemic disease, with the development of multiple myeloma in one-third of cases.4 More aggressive cases are associated with multiple lesions, larger size, and IgA secretion by neoplastic cells.
Our patient with a solitary dermal infiltrate of monoclonal plasma cells did not have bone marrow involvement and had an unexplained elevated level of free k light chains in the serum. This unexplained relationship underscores the importance of continued surveillance in any cutaneous lymphoma.
1. Rongioletti F, Patterson JW, Rebora A. The histological and pathogenetic spectrum of cutaneous disease in monoclonal gammopathies. J Cutan Pathol. 2008;35:705-721.
2. Willemze R, Jaffe ES, Burg G, et al. WHO-EORTC classification for cutaneous lymphomas. Blood. 2005;105:3768-3785.
3. Muscardin LM, Pulsoni A, Cerroni L. Primary cutaneous plasmacytoma: report of a case with review of the literature. J Am Acad Dermatol. 2000;43(5, pt 2):962-965.
4. Kazakov DV, Belousova IE, Müller B, et al. Primary cutaneous plasmacytoma: a clinicopathological study of two cases with a long-term follow-up and review of the literature. J Cutan Pathol. 2002;29:244-248.
5. Aricò M, Bongiorno MR. Primary cutaneous plasmacytosis in a child. is this a new entity? J Eur Acad Dermatol. 2002;16:164-167.
6. Willoughby V, Werlang-Perurena A, Kelly A, et al. Primary cutaneous plasmacytoma (posttransplant lymphoproliferative disorder, plasmacytoma-like) in a heart transplant patient. Am J Dermatopathol. 2006;28:442-445.
7. Buffet M, Dupin N, Carlotti A, et al. Epstein-Barr virus–associated cutaneous plasmacytoma post–solid organ transplantation [in French]. Ann Dermatol Venereol. 2004;131:1085-1091.
8. Zendri E, Venturi C, Ricci R, et al. Primary cutaneous plasmacytoma: a role for a triggering stimulus? Clin Exp Dermatol. 2005;30:229-231.
9. Tüting T, Bork K. Primary plasmacytoma of the skin. J Am Acad Dermatol. 1996;34(2, pt 2):386-390.
10. Miura H, Itami S, Yoshikawa K. Treatment of facial lesion of cutaneous plasmacytosis with tacrolimus ointment. J Am Acad Dermatol. 2003;49:1195-1196.
11. Tessari G, Fabbian F, Colato C, et al. Primary cutaneous plasmacytoma after rejection of a transplanted kidney: case report and review of the literature. Int J Hematol. 2004;80:361-364.
1. Rongioletti F, Patterson JW, Rebora A. The histological and pathogenetic spectrum of cutaneous disease in monoclonal gammopathies. J Cutan Pathol. 2008;35:705-721.
2. Willemze R, Jaffe ES, Burg G, et al. WHO-EORTC classification for cutaneous lymphomas. Blood. 2005;105:3768-3785.
3. Muscardin LM, Pulsoni A, Cerroni L. Primary cutaneous plasmacytoma: report of a case with review of the literature. J Am Acad Dermatol. 2000;43(5, pt 2):962-965.
4. Kazakov DV, Belousova IE, Müller B, et al. Primary cutaneous plasmacytoma: a clinicopathological study of two cases with a long-term follow-up and review of the literature. J Cutan Pathol. 2002;29:244-248.
5. Aricò M, Bongiorno MR. Primary cutaneous plasmacytosis in a child. is this a new entity? J Eur Acad Dermatol. 2002;16:164-167.
6. Willoughby V, Werlang-Perurena A, Kelly A, et al. Primary cutaneous plasmacytoma (posttransplant lymphoproliferative disorder, plasmacytoma-like) in a heart transplant patient. Am J Dermatopathol. 2006;28:442-445.
7. Buffet M, Dupin N, Carlotti A, et al. Epstein-Barr virus–associated cutaneous plasmacytoma post–solid organ transplantation [in French]. Ann Dermatol Venereol. 2004;131:1085-1091.
8. Zendri E, Venturi C, Ricci R, et al. Primary cutaneous plasmacytoma: a role for a triggering stimulus? Clin Exp Dermatol. 2005;30:229-231.
9. Tüting T, Bork K. Primary plasmacytoma of the skin. J Am Acad Dermatol. 1996;34(2, pt 2):386-390.
10. Miura H, Itami S, Yoshikawa K. Treatment of facial lesion of cutaneous plasmacytosis with tacrolimus ointment. J Am Acad Dermatol. 2003;49:1195-1196.
11. Tessari G, Fabbian F, Colato C, et al. Primary cutaneous plasmacytoma after rejection of a transplanted kidney: case report and review of the literature. Int J Hematol. 2004;80:361-364.
Early Cutaneous Metastasis of Adenoid Cystic Carcinoma of the Salivary Gland
Adenoid cystic carcinoma (ACC) is the second most common malignant neoplasm of the major and minor salivary glands.1 The disease course generally is characterized by slow yet relentless progression with a poor long-term prognosis. The presence of distant metastasis heralds a poor prognosis in patients with ACC of the head and neck, with a median survival of 4.3 to 7.3 months.2 Twenty-five percent to 50% of ACCs of the salivary glands result in distant metastasis, primarily involving the lungs, bones,3-5 liver, and brain.4,5 Cutaneous metastasis of ACCs of the salivary glands is rare and usually is limited to the head and neck regions. We describe a case of early cutaneous metastasis of ACC on the head and neck, driving the identification of systemic diffusion of the neoplasm. Our case underscores the importance of conducting a complete skin examination in oncologic patients, especially focusing on cutaneous and subcutaneous nodules and papules that are eruptive or show rapid development.
Case Report
A 61-year-old man presented to the dermatology unit with a rapidly progressive mass on the right submandibular region. Five years prior he underwent wide excision of the lymph nodes of the right axilla because of the presence of a subcutaneous nodule, which revealed the absence of atypical histologic features; annual computed tomography (CT) scans were subsequently performed with no remarkable results. One year later, the presence of a small and stable submandibular nodule was noted; all biopsies performed were negative for cancer.
Wide excision of the submandibular mass was performed after clinical evaluation. Histopathologic examination revealed an ACC of the submandibular salivary gland. One week later, a dissection of the right side of the neck was performed with total sialectomy, right laterocervical lymphadenectomy (level 1-2 ab-3, Robbins), revealing nodal diffusion of the tumor cells (3 of 28 nodes were positive). Surgical treatment was immediately followed by radiotherapy (4800–6000 Gy) of the tumor region and lymphatic neck regions. At 6 months’ follow-up, clinical examination and a CT scan were negative for metastasis.
The patient was referred to our dermatology clinic 2 months later for evaluation of a nonspecific dermatitis that had developed at the surgical site on the right side of the neck. Physical examination revealed radiodermatitis on the scar. Additionally, slightly erythematous monomorphic and asymptomatic eruptive papules were noted on the scalp (Figure 1A) and on the left side of the clavicle (Figure 1B). The lesions had developed gradually over 10 days to a diameter of 5 mm. No lymphadenopathy was noted. Due to the characteristics of the lesions (morphology and rapid eruption) and the patient’s medical history, a skin biopsy was performed with a suspected diagnosis of cutaneous metastasis of ACC. The diagnosis was confirmed on histopathologic evaluation, which showed the characteristic features of metastatic ACC with solid areas as shown in Figure 2. Subsequent CT scans of the head, neck, and chest revealed lung and bone metastasis, which had not been present on prior investigation. A few days earlier the patient had developed a productive cough, hemoptysis, and mild dyspnea, which confirmed clinical progression of the disease. The patient was started on palliative chemotherapy and radiotherapy treatment.
Comment
Adenoid cystic carcinoma is a malignant salivary gland tumor characterized by slow yet relentless progression that is plagued by local recurrence, late metastases, and ultimately fatal outcomes.6 It represents approximately 7.5% of salivary gland malignancies.7 Few cases of cutaneous metastases of ACC have been reported on the head and neck as well as the abdomen,8-10 accounting for 1% to 2% of all cutaneous metastases,11 with typical distribution on the head and neck. The presence of distant metastases heralds a poor prognosis in head and neck cancers, with a median survival of 4.3 to 7.3 months.2 Treatment of this patient population usually is palliative. Patients with malignant salivary gland tumors should have a chest radiograph or CT scan at their initial assessment to exclude the possibility of lung metastasis. A higher risk for developing distant metastasis is associated with high-grade tumors such as ACC; salivary duct carcinomas; high-grade mucoepidermoid carcinomas; and tumors located in the submandibular gland, posterior tongue, and pharynx.12 The 3 major growth patterns are tubular, cribriform, and solid. The quantity of solid components is considered to be the most notable indication of a poor prognosis.13-15 In our patient, the site of the tumor—the submandibular region—as well as the presence of histopathologic solid structures suggested a poor prognosis. Our patient presented with eruptive papules on the scalp and on the left side of the clavicle, which was on the opposite side of the primary tumor. In many patients, cutaneous diffusion of the tumor precedes systemic diffusion and therefore can be considered as an early prognostic sign of metastasis.
Conclusion
Cutaneous metastasis represents an important sign of disease progression, allowing for the early discovery of systemic diffusion of the tumor as well as adjustment of the therapeutic approach. Our patient not only highlights a rare case of cutaneous metastases in this type of tumor but also the importance of conducting complete skin examinations in oncologic patients. Even minor findings should not be ignored in this patient population. In fact, if correctly interpreted the early discovery of cutaneous metastasis in patients with salivary gland tumors may determine a dramatic change in diagnosis, prognosis, and treatment.
1. Boukheris H, Curtis RE, Land CE, et al. Incidence of carcinoma of the major salivary glands according to WHO classification, 1992 to 2006: a population-based study in the United States [published online ahead of print October 27, 2009]. Cancer Epidemiol Biomarkers Prev. 2009;18:2899-2906.
2. Elnayal A, Moran CA, Fox PS, et al. Primary salivary gland-type lung cancer: imaging and clinical predictors of outcome. AJR Am J Roentgenol. 2013;201:W57-W63.
3. Perez DE, Magrin J, Almeida OP, et al. Multiple cutaneous metastases from a parotid adenoid cystic carcinoma. Pathol Oncol Res. 2007;13:167-169.
4. Khan AJ, DiGiovanna MP, Ross DA, et al. Adenoid cystic carcinoma: a retrospective clinical review. Int J Cancer. 2001;96:149-158.
5. Spiro RH. Distant metastasis in adenoid cystic carcinoma of salivary origin. Am J Surg. 1997;174:495-498.
6. El-Naggar AK, Huvos AG. Adenoid cystic carcinoma. In: Barnes EL, Kleihues P, Sobin LH, et al, eds. Pathology and Genetics of Head and Neck Tumours. World Health Organization Classification of Tumours. Lyon, France: IARC Press; 2005:221-222.
7. Ellis GL, Auclair PL. Tumors of the Salivary Glands. 4th ed. Washington, DC: Armed Forces Institute of Pathology; 1996.
8. Chang CH, Liao YL, Hong HS. Cutaneous metastasis from adenoid cystic carcinoma of the parotid gland. Dermatol Surg. 2003;29:775-779.
9. Nakamura M, Miyachi Y. Cutaneous metastasis from an adenoid cystic carcinoma of the lacrimal gland. Br J Dermatol. 1999;141:373-374.
10. Nascimento AG, Amaral AL, Prado LA, et al. Adenoid cystic carcinoma of salivary glands: a study of 61 cases with clinicopathologic correlation. Cancer. 1986;57:312-319.
11. Schwartz RA. Cutaneous metastatic disease. J Am Acad Dermatol. 1995;33 (2, pt 1):161-182; quiz 183-186.
12. Schwentner I, Obrist P, Thumfart W, et al. Distant metastasis of parotid gland tumors. Acta Otolaryngol. 2006;126:340-345.
13. Da Cruz Perez DE, de Abreu Alves F, Nobuko Nishimoto I, et al. Prognostic factors in head and neck adenoid cystic carcinoma. Oral Oncol. 2006;42:139-146.
14. Perzin KH, Gullane P, Clairmont AC. Adenoid cystic carcinomas arising in salivary glands: a correlation of histologic features and clinical course. Cancer. 1978;42:265-282.
15. Szanto PA, Luna MA, Tortoledo ME, et al. Histologic grading of adenoid cystic carcinoma of the salivary glands. Cancer. 1984;54:1062-1069.
Adenoid cystic carcinoma (ACC) is the second most common malignant neoplasm of the major and minor salivary glands.1 The disease course generally is characterized by slow yet relentless progression with a poor long-term prognosis. The presence of distant metastasis heralds a poor prognosis in patients with ACC of the head and neck, with a median survival of 4.3 to 7.3 months.2 Twenty-five percent to 50% of ACCs of the salivary glands result in distant metastasis, primarily involving the lungs, bones,3-5 liver, and brain.4,5 Cutaneous metastasis of ACCs of the salivary glands is rare and usually is limited to the head and neck regions. We describe a case of early cutaneous metastasis of ACC on the head and neck, driving the identification of systemic diffusion of the neoplasm. Our case underscores the importance of conducting a complete skin examination in oncologic patients, especially focusing on cutaneous and subcutaneous nodules and papules that are eruptive or show rapid development.
Case Report
A 61-year-old man presented to the dermatology unit with a rapidly progressive mass on the right submandibular region. Five years prior he underwent wide excision of the lymph nodes of the right axilla because of the presence of a subcutaneous nodule, which revealed the absence of atypical histologic features; annual computed tomography (CT) scans were subsequently performed with no remarkable results. One year later, the presence of a small and stable submandibular nodule was noted; all biopsies performed were negative for cancer.
Wide excision of the submandibular mass was performed after clinical evaluation. Histopathologic examination revealed an ACC of the submandibular salivary gland. One week later, a dissection of the right side of the neck was performed with total sialectomy, right laterocervical lymphadenectomy (level 1-2 ab-3, Robbins), revealing nodal diffusion of the tumor cells (3 of 28 nodes were positive). Surgical treatment was immediately followed by radiotherapy (4800–6000 Gy) of the tumor region and lymphatic neck regions. At 6 months’ follow-up, clinical examination and a CT scan were negative for metastasis.
The patient was referred to our dermatology clinic 2 months later for evaluation of a nonspecific dermatitis that had developed at the surgical site on the right side of the neck. Physical examination revealed radiodermatitis on the scar. Additionally, slightly erythematous monomorphic and asymptomatic eruptive papules were noted on the scalp (Figure 1A) and on the left side of the clavicle (Figure 1B). The lesions had developed gradually over 10 days to a diameter of 5 mm. No lymphadenopathy was noted. Due to the characteristics of the lesions (morphology and rapid eruption) and the patient’s medical history, a skin biopsy was performed with a suspected diagnosis of cutaneous metastasis of ACC. The diagnosis was confirmed on histopathologic evaluation, which showed the characteristic features of metastatic ACC with solid areas as shown in Figure 2. Subsequent CT scans of the head, neck, and chest revealed lung and bone metastasis, which had not been present on prior investigation. A few days earlier the patient had developed a productive cough, hemoptysis, and mild dyspnea, which confirmed clinical progression of the disease. The patient was started on palliative chemotherapy and radiotherapy treatment.
Comment
Adenoid cystic carcinoma is a malignant salivary gland tumor characterized by slow yet relentless progression that is plagued by local recurrence, late metastases, and ultimately fatal outcomes.6 It represents approximately 7.5% of salivary gland malignancies.7 Few cases of cutaneous metastases of ACC have been reported on the head and neck as well as the abdomen,8-10 accounting for 1% to 2% of all cutaneous metastases,11 with typical distribution on the head and neck. The presence of distant metastases heralds a poor prognosis in head and neck cancers, with a median survival of 4.3 to 7.3 months.2 Treatment of this patient population usually is palliative. Patients with malignant salivary gland tumors should have a chest radiograph or CT scan at their initial assessment to exclude the possibility of lung metastasis. A higher risk for developing distant metastasis is associated with high-grade tumors such as ACC; salivary duct carcinomas; high-grade mucoepidermoid carcinomas; and tumors located in the submandibular gland, posterior tongue, and pharynx.12 The 3 major growth patterns are tubular, cribriform, and solid. The quantity of solid components is considered to be the most notable indication of a poor prognosis.13-15 In our patient, the site of the tumor—the submandibular region—as well as the presence of histopathologic solid structures suggested a poor prognosis. Our patient presented with eruptive papules on the scalp and on the left side of the clavicle, which was on the opposite side of the primary tumor. In many patients, cutaneous diffusion of the tumor precedes systemic diffusion and therefore can be considered as an early prognostic sign of metastasis.
Conclusion
Cutaneous metastasis represents an important sign of disease progression, allowing for the early discovery of systemic diffusion of the tumor as well as adjustment of the therapeutic approach. Our patient not only highlights a rare case of cutaneous metastases in this type of tumor but also the importance of conducting complete skin examinations in oncologic patients. Even minor findings should not be ignored in this patient population. In fact, if correctly interpreted the early discovery of cutaneous metastasis in patients with salivary gland tumors may determine a dramatic change in diagnosis, prognosis, and treatment.
Adenoid cystic carcinoma (ACC) is the second most common malignant neoplasm of the major and minor salivary glands.1 The disease course generally is characterized by slow yet relentless progression with a poor long-term prognosis. The presence of distant metastasis heralds a poor prognosis in patients with ACC of the head and neck, with a median survival of 4.3 to 7.3 months.2 Twenty-five percent to 50% of ACCs of the salivary glands result in distant metastasis, primarily involving the lungs, bones,3-5 liver, and brain.4,5 Cutaneous metastasis of ACCs of the salivary glands is rare and usually is limited to the head and neck regions. We describe a case of early cutaneous metastasis of ACC on the head and neck, driving the identification of systemic diffusion of the neoplasm. Our case underscores the importance of conducting a complete skin examination in oncologic patients, especially focusing on cutaneous and subcutaneous nodules and papules that are eruptive or show rapid development.
Case Report
A 61-year-old man presented to the dermatology unit with a rapidly progressive mass on the right submandibular region. Five years prior he underwent wide excision of the lymph nodes of the right axilla because of the presence of a subcutaneous nodule, which revealed the absence of atypical histologic features; annual computed tomography (CT) scans were subsequently performed with no remarkable results. One year later, the presence of a small and stable submandibular nodule was noted; all biopsies performed were negative for cancer.
Wide excision of the submandibular mass was performed after clinical evaluation. Histopathologic examination revealed an ACC of the submandibular salivary gland. One week later, a dissection of the right side of the neck was performed with total sialectomy, right laterocervical lymphadenectomy (level 1-2 ab-3, Robbins), revealing nodal diffusion of the tumor cells (3 of 28 nodes were positive). Surgical treatment was immediately followed by radiotherapy (4800–6000 Gy) of the tumor region and lymphatic neck regions. At 6 months’ follow-up, clinical examination and a CT scan were negative for metastasis.
The patient was referred to our dermatology clinic 2 months later for evaluation of a nonspecific dermatitis that had developed at the surgical site on the right side of the neck. Physical examination revealed radiodermatitis on the scar. Additionally, slightly erythematous monomorphic and asymptomatic eruptive papules were noted on the scalp (Figure 1A) and on the left side of the clavicle (Figure 1B). The lesions had developed gradually over 10 days to a diameter of 5 mm. No lymphadenopathy was noted. Due to the characteristics of the lesions (morphology and rapid eruption) and the patient’s medical history, a skin biopsy was performed with a suspected diagnosis of cutaneous metastasis of ACC. The diagnosis was confirmed on histopathologic evaluation, which showed the characteristic features of metastatic ACC with solid areas as shown in Figure 2. Subsequent CT scans of the head, neck, and chest revealed lung and bone metastasis, which had not been present on prior investigation. A few days earlier the patient had developed a productive cough, hemoptysis, and mild dyspnea, which confirmed clinical progression of the disease. The patient was started on palliative chemotherapy and radiotherapy treatment.
Comment
Adenoid cystic carcinoma is a malignant salivary gland tumor characterized by slow yet relentless progression that is plagued by local recurrence, late metastases, and ultimately fatal outcomes.6 It represents approximately 7.5% of salivary gland malignancies.7 Few cases of cutaneous metastases of ACC have been reported on the head and neck as well as the abdomen,8-10 accounting for 1% to 2% of all cutaneous metastases,11 with typical distribution on the head and neck. The presence of distant metastases heralds a poor prognosis in head and neck cancers, with a median survival of 4.3 to 7.3 months.2 Treatment of this patient population usually is palliative. Patients with malignant salivary gland tumors should have a chest radiograph or CT scan at their initial assessment to exclude the possibility of lung metastasis. A higher risk for developing distant metastasis is associated with high-grade tumors such as ACC; salivary duct carcinomas; high-grade mucoepidermoid carcinomas; and tumors located in the submandibular gland, posterior tongue, and pharynx.12 The 3 major growth patterns are tubular, cribriform, and solid. The quantity of solid components is considered to be the most notable indication of a poor prognosis.13-15 In our patient, the site of the tumor—the submandibular region—as well as the presence of histopathologic solid structures suggested a poor prognosis. Our patient presented with eruptive papules on the scalp and on the left side of the clavicle, which was on the opposite side of the primary tumor. In many patients, cutaneous diffusion of the tumor precedes systemic diffusion and therefore can be considered as an early prognostic sign of metastasis.
Conclusion
Cutaneous metastasis represents an important sign of disease progression, allowing for the early discovery of systemic diffusion of the tumor as well as adjustment of the therapeutic approach. Our patient not only highlights a rare case of cutaneous metastases in this type of tumor but also the importance of conducting complete skin examinations in oncologic patients. Even minor findings should not be ignored in this patient population. In fact, if correctly interpreted the early discovery of cutaneous metastasis in patients with salivary gland tumors may determine a dramatic change in diagnosis, prognosis, and treatment.
1. Boukheris H, Curtis RE, Land CE, et al. Incidence of carcinoma of the major salivary glands according to WHO classification, 1992 to 2006: a population-based study in the United States [published online ahead of print October 27, 2009]. Cancer Epidemiol Biomarkers Prev. 2009;18:2899-2906.
2. Elnayal A, Moran CA, Fox PS, et al. Primary salivary gland-type lung cancer: imaging and clinical predictors of outcome. AJR Am J Roentgenol. 2013;201:W57-W63.
3. Perez DE, Magrin J, Almeida OP, et al. Multiple cutaneous metastases from a parotid adenoid cystic carcinoma. Pathol Oncol Res. 2007;13:167-169.
4. Khan AJ, DiGiovanna MP, Ross DA, et al. Adenoid cystic carcinoma: a retrospective clinical review. Int J Cancer. 2001;96:149-158.
5. Spiro RH. Distant metastasis in adenoid cystic carcinoma of salivary origin. Am J Surg. 1997;174:495-498.
6. El-Naggar AK, Huvos AG. Adenoid cystic carcinoma. In: Barnes EL, Kleihues P, Sobin LH, et al, eds. Pathology and Genetics of Head and Neck Tumours. World Health Organization Classification of Tumours. Lyon, France: IARC Press; 2005:221-222.
7. Ellis GL, Auclair PL. Tumors of the Salivary Glands. 4th ed. Washington, DC: Armed Forces Institute of Pathology; 1996.
8. Chang CH, Liao YL, Hong HS. Cutaneous metastasis from adenoid cystic carcinoma of the parotid gland. Dermatol Surg. 2003;29:775-779.
9. Nakamura M, Miyachi Y. Cutaneous metastasis from an adenoid cystic carcinoma of the lacrimal gland. Br J Dermatol. 1999;141:373-374.
10. Nascimento AG, Amaral AL, Prado LA, et al. Adenoid cystic carcinoma of salivary glands: a study of 61 cases with clinicopathologic correlation. Cancer. 1986;57:312-319.
11. Schwartz RA. Cutaneous metastatic disease. J Am Acad Dermatol. 1995;33 (2, pt 1):161-182; quiz 183-186.
12. Schwentner I, Obrist P, Thumfart W, et al. Distant metastasis of parotid gland tumors. Acta Otolaryngol. 2006;126:340-345.
13. Da Cruz Perez DE, de Abreu Alves F, Nobuko Nishimoto I, et al. Prognostic factors in head and neck adenoid cystic carcinoma. Oral Oncol. 2006;42:139-146.
14. Perzin KH, Gullane P, Clairmont AC. Adenoid cystic carcinomas arising in salivary glands: a correlation of histologic features and clinical course. Cancer. 1978;42:265-282.
15. Szanto PA, Luna MA, Tortoledo ME, et al. Histologic grading of adenoid cystic carcinoma of the salivary glands. Cancer. 1984;54:1062-1069.
1. Boukheris H, Curtis RE, Land CE, et al. Incidence of carcinoma of the major salivary glands according to WHO classification, 1992 to 2006: a population-based study in the United States [published online ahead of print October 27, 2009]. Cancer Epidemiol Biomarkers Prev. 2009;18:2899-2906.
2. Elnayal A, Moran CA, Fox PS, et al. Primary salivary gland-type lung cancer: imaging and clinical predictors of outcome. AJR Am J Roentgenol. 2013;201:W57-W63.
3. Perez DE, Magrin J, Almeida OP, et al. Multiple cutaneous metastases from a parotid adenoid cystic carcinoma. Pathol Oncol Res. 2007;13:167-169.
4. Khan AJ, DiGiovanna MP, Ross DA, et al. Adenoid cystic carcinoma: a retrospective clinical review. Int J Cancer. 2001;96:149-158.
5. Spiro RH. Distant metastasis in adenoid cystic carcinoma of salivary origin. Am J Surg. 1997;174:495-498.
6. El-Naggar AK, Huvos AG. Adenoid cystic carcinoma. In: Barnes EL, Kleihues P, Sobin LH, et al, eds. Pathology and Genetics of Head and Neck Tumours. World Health Organization Classification of Tumours. Lyon, France: IARC Press; 2005:221-222.
7. Ellis GL, Auclair PL. Tumors of the Salivary Glands. 4th ed. Washington, DC: Armed Forces Institute of Pathology; 1996.
8. Chang CH, Liao YL, Hong HS. Cutaneous metastasis from adenoid cystic carcinoma of the parotid gland. Dermatol Surg. 2003;29:775-779.
9. Nakamura M, Miyachi Y. Cutaneous metastasis from an adenoid cystic carcinoma of the lacrimal gland. Br J Dermatol. 1999;141:373-374.
10. Nascimento AG, Amaral AL, Prado LA, et al. Adenoid cystic carcinoma of salivary glands: a study of 61 cases with clinicopathologic correlation. Cancer. 1986;57:312-319.
11. Schwartz RA. Cutaneous metastatic disease. J Am Acad Dermatol. 1995;33 (2, pt 1):161-182; quiz 183-186.
12. Schwentner I, Obrist P, Thumfart W, et al. Distant metastasis of parotid gland tumors. Acta Otolaryngol. 2006;126:340-345.
13. Da Cruz Perez DE, de Abreu Alves F, Nobuko Nishimoto I, et al. Prognostic factors in head and neck adenoid cystic carcinoma. Oral Oncol. 2006;42:139-146.
14. Perzin KH, Gullane P, Clairmont AC. Adenoid cystic carcinomas arising in salivary glands: a correlation of histologic features and clinical course. Cancer. 1978;42:265-282.
15. Szanto PA, Luna MA, Tortoledo ME, et al. Histologic grading of adenoid cystic carcinoma of the salivary glands. Cancer. 1984;54:1062-1069.
Teenage blistering sunburns increased melanoma risk by 80% in white women
Melanoma risk increased by 80% among women who had five or more severe sunburns as teenagers, based on a review of data from more than 100,000 white women.
The findings were published online May 29 in Cancer Epidemiology, Biomarkers and Prevention, a journal of the American Association for Cancer Research.
"We investigated the relationship between a number of potential risk factors, including chronic sun exposure over long durations in adulthood and sun exposure in early life, and risk of different types of skin cancer," wrote Dr. Shaowei Wu of Brigham and Women’s Hospital, Boston, and colleagues.
Diagnoses of 6,955 basal cell carcinomas (BCCs), 880 squamous cell carcinomas (SCCs), and 779 melanomas were identified during 2.05 million person-years of follow-up. The relative risk of skin cancer was 68% higher for both BCC and SCC, and 80% higher for melanoma in women who reported five or more blistering sunburns between ages 15 and 25 years, compared with those who reported no such sunburns (Cancer Epidemiol. Biomarkers Prev. 2014;23:1080-9).
The study included 101,916 nurses participating in the Nurses’ Health Study II. The women enrolled between the ages of 25 and 42 years, and were followed for 20 years between 1989 and 2009. The eligible participants had no baseline history of any cancer and were diagnosed with incident BCC, SCC, or melanoma between baseline and the last follow-up.
Cumulative ultraviolet radiation exposure in adulthood, measured over long durations using a composite estimate called UV flux, also was associated with an increased risk of BCC and SCC, but not with melanoma. This suggests that "melanoma may have a more complicated relationship with sun exposure than SCC and BCC," the researchers said.
The study was limited by the approximate estimates of UV radiation exposure, incomplete information about personal behaviors such as sunscreen use, and the fact that BCC cases were not independently validated as SCC and melanoma. However, the findings "support heterogenous associations between sun exposure, other potential risk factors, and risk of different types of skin cancer, and thus may have potential implications for the prevention of skin cancers," the researchers noted.
Also, "our participants consisted entirely of white women, and thus the generalizability of the results to men and other ethnicities may be limited," they said.
The study was supported by the department of dermatology at Brigham and Women’s Hospital, and by a grant from the National Institutes of Health. Corresponding author Dr. Abrar A. Qureshi has served as a consultant and/or advisory board member for multiple pharmaceutical companies. The other authors had no financial conflicts to disclose.
Melanoma risk increased by 80% among women who had five or more severe sunburns as teenagers, based on a review of data from more than 100,000 white women.
The findings were published online May 29 in Cancer Epidemiology, Biomarkers and Prevention, a journal of the American Association for Cancer Research.
"We investigated the relationship between a number of potential risk factors, including chronic sun exposure over long durations in adulthood and sun exposure in early life, and risk of different types of skin cancer," wrote Dr. Shaowei Wu of Brigham and Women’s Hospital, Boston, and colleagues.
Diagnoses of 6,955 basal cell carcinomas (BCCs), 880 squamous cell carcinomas (SCCs), and 779 melanomas were identified during 2.05 million person-years of follow-up. The relative risk of skin cancer was 68% higher for both BCC and SCC, and 80% higher for melanoma in women who reported five or more blistering sunburns between ages 15 and 25 years, compared with those who reported no such sunburns (Cancer Epidemiol. Biomarkers Prev. 2014;23:1080-9).
The study included 101,916 nurses participating in the Nurses’ Health Study II. The women enrolled between the ages of 25 and 42 years, and were followed for 20 years between 1989 and 2009. The eligible participants had no baseline history of any cancer and were diagnosed with incident BCC, SCC, or melanoma between baseline and the last follow-up.
Cumulative ultraviolet radiation exposure in adulthood, measured over long durations using a composite estimate called UV flux, also was associated with an increased risk of BCC and SCC, but not with melanoma. This suggests that "melanoma may have a more complicated relationship with sun exposure than SCC and BCC," the researchers said.
The study was limited by the approximate estimates of UV radiation exposure, incomplete information about personal behaviors such as sunscreen use, and the fact that BCC cases were not independently validated as SCC and melanoma. However, the findings "support heterogenous associations between sun exposure, other potential risk factors, and risk of different types of skin cancer, and thus may have potential implications for the prevention of skin cancers," the researchers noted.
Also, "our participants consisted entirely of white women, and thus the generalizability of the results to men and other ethnicities may be limited," they said.
The study was supported by the department of dermatology at Brigham and Women’s Hospital, and by a grant from the National Institutes of Health. Corresponding author Dr. Abrar A. Qureshi has served as a consultant and/or advisory board member for multiple pharmaceutical companies. The other authors had no financial conflicts to disclose.
Melanoma risk increased by 80% among women who had five or more severe sunburns as teenagers, based on a review of data from more than 100,000 white women.
The findings were published online May 29 in Cancer Epidemiology, Biomarkers and Prevention, a journal of the American Association for Cancer Research.
"We investigated the relationship between a number of potential risk factors, including chronic sun exposure over long durations in adulthood and sun exposure in early life, and risk of different types of skin cancer," wrote Dr. Shaowei Wu of Brigham and Women’s Hospital, Boston, and colleagues.
Diagnoses of 6,955 basal cell carcinomas (BCCs), 880 squamous cell carcinomas (SCCs), and 779 melanomas were identified during 2.05 million person-years of follow-up. The relative risk of skin cancer was 68% higher for both BCC and SCC, and 80% higher for melanoma in women who reported five or more blistering sunburns between ages 15 and 25 years, compared with those who reported no such sunburns (Cancer Epidemiol. Biomarkers Prev. 2014;23:1080-9).
The study included 101,916 nurses participating in the Nurses’ Health Study II. The women enrolled between the ages of 25 and 42 years, and were followed for 20 years between 1989 and 2009. The eligible participants had no baseline history of any cancer and were diagnosed with incident BCC, SCC, or melanoma between baseline and the last follow-up.
Cumulative ultraviolet radiation exposure in adulthood, measured over long durations using a composite estimate called UV flux, also was associated with an increased risk of BCC and SCC, but not with melanoma. This suggests that "melanoma may have a more complicated relationship with sun exposure than SCC and BCC," the researchers said.
The study was limited by the approximate estimates of UV radiation exposure, incomplete information about personal behaviors such as sunscreen use, and the fact that BCC cases were not independently validated as SCC and melanoma. However, the findings "support heterogenous associations between sun exposure, other potential risk factors, and risk of different types of skin cancer, and thus may have potential implications for the prevention of skin cancers," the researchers noted.
Also, "our participants consisted entirely of white women, and thus the generalizability of the results to men and other ethnicities may be limited," they said.
The study was supported by the department of dermatology at Brigham and Women’s Hospital, and by a grant from the National Institutes of Health. Corresponding author Dr. Abrar A. Qureshi has served as a consultant and/or advisory board member for multiple pharmaceutical companies. The other authors had no financial conflicts to disclose.
FROM CANCER EPIDEMIOLOGY, BIOMARKERS AND PREVENTION
Key clinical point: Increased melanoma risk was mainly associated with greater sun exposure in early life, while greater sun exposure in both early life and adulthood were predictive of increased BCC and SCC risk.
Major finding: Skin cancer risk was 68% higher for BCC and SCC, and 80% higher for melanoma in women who reported five or more blistering sunburns between ages 15 and 25 years.
Data source: 101,916 nurses enrolled in the Nurses’ Health Study II (1989-2009).
Disclosures: The study was supported by the department of dermatology at Brigham and Women’s Hospital, and by a grant from the National Institutes of Health. Corresponding author Dr. Qureshi has served as a consultant and/or advisory board member for multiple pharmaceutical companies. The other authors had no financial conflicts to disclose.
Is There a Potential Benefit of Expressive Writing for Dermatology Patients?

A diagnosis of a chronic and/or serious medical condition can be a traumatic experience. Patients may experience not only intrusive thoughts but also avoidance behaviors. The affected individuals can express these trauma-associated symptoms as depression, fatigue, and sleep disturbance.
Milbury et al (J Clin Oncol. 2014;32:663-670) included 277 patients who had been diagnosed with renal cell carcinoma in a study to evaluate if writing about their deepest thoughts and feelings regarding their cancer (expressive writing) offered a quality-of-life benefit compared to writing about neutral topics (neutral writing). They found that expressive writing not only reduced the patients’ cancer-related symptoms but also improved their physical functioning.
What’s the issue?
Many of the conditions that affect dermatology patients are chronic, serious, or both. For example, psoriasis is a chronic skin condition with the potential for serious associated rheumatologic or metabolic sequelae. In addition, skin malignancies (eg, basal cell carcinoma, squamous cell carcinoma, malignant melanoma) and cutaneous lymphoma (eg, mycosis fungoides) are conditions that require chronic surveillance and possibly may have serious consequences.
Expressive writing for dermatology patients might result in remarkable improvement in emotional and physical health. Perhaps dermatologists should recommend this intervention for their patients.

A diagnosis of a chronic and/or serious medical condition can be a traumatic experience. Patients may experience not only intrusive thoughts but also avoidance behaviors. The affected individuals can express these trauma-associated symptoms as depression, fatigue, and sleep disturbance.
Milbury et al (J Clin Oncol. 2014;32:663-670) included 277 patients who had been diagnosed with renal cell carcinoma in a study to evaluate if writing about their deepest thoughts and feelings regarding their cancer (expressive writing) offered a quality-of-life benefit compared to writing about neutral topics (neutral writing). They found that expressive writing not only reduced the patients’ cancer-related symptoms but also improved their physical functioning.
What’s the issue?
Many of the conditions that affect dermatology patients are chronic, serious, or both. For example, psoriasis is a chronic skin condition with the potential for serious associated rheumatologic or metabolic sequelae. In addition, skin malignancies (eg, basal cell carcinoma, squamous cell carcinoma, malignant melanoma) and cutaneous lymphoma (eg, mycosis fungoides) are conditions that require chronic surveillance and possibly may have serious consequences.
Expressive writing for dermatology patients might result in remarkable improvement in emotional and physical health. Perhaps dermatologists should recommend this intervention for their patients.

A diagnosis of a chronic and/or serious medical condition can be a traumatic experience. Patients may experience not only intrusive thoughts but also avoidance behaviors. The affected individuals can express these trauma-associated symptoms as depression, fatigue, and sleep disturbance.
Milbury et al (J Clin Oncol. 2014;32:663-670) included 277 patients who had been diagnosed with renal cell carcinoma in a study to evaluate if writing about their deepest thoughts and feelings regarding their cancer (expressive writing) offered a quality-of-life benefit compared to writing about neutral topics (neutral writing). They found that expressive writing not only reduced the patients’ cancer-related symptoms but also improved their physical functioning.
What’s the issue?
Many of the conditions that affect dermatology patients are chronic, serious, or both. For example, psoriasis is a chronic skin condition with the potential for serious associated rheumatologic or metabolic sequelae. In addition, skin malignancies (eg, basal cell carcinoma, squamous cell carcinoma, malignant melanoma) and cutaneous lymphoma (eg, mycosis fungoides) are conditions that require chronic surveillance and possibly may have serious consequences.
Expressive writing for dermatology patients might result in remarkable improvement in emotional and physical health. Perhaps dermatologists should recommend this intervention for their patients.