User login
VULVAR PAIN SYNDROMES A bounty of treatments—but not all of them are proven
- Part 1: Making the correct diagnosis
(September 2011) - Part 3: Vestibulodynia
(November 2011)
As we discussed in the first installment of this three-part series in the September issue of OBG Management, the causes of vulvar pain are many, and the diagnosis of this common complaint can be difficult. Once the diagnosis of vulvodynia has been made, however, the challenge shifts to finding an effective treatment. Here, our expert panel discusses the many options available, the data (or lack of it) behind each therapy, and what to do in refractory cases.
In Part 3 of this series, in the November issue, the focus will be vestibulodynia.

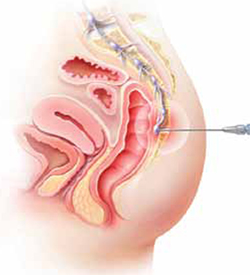
The lower vagina and vulva are richly supplied with peripheral nerves and are, therefore, sensitive to pain, particularly the region of the hymeneal ring. Although the pudendal nerve (arrow) courses through the area, it is an uncommon source of vulvar pain.
Management of vulvar pain begins with simple measures
Dr. Lonky: How do you approach treatment of vulvar pain syndromes?
Dr. Haefner: I often advise the patient to begin with simple measures. For example, I recommend that she wear cotton underwear during the day, but no underwear at night. If she perspires with exercise, wicking underwear may be helpful. I also counsel the patient to avoid vulvar irritants, douches, and the application of soap of any kind to the vulva. Cool gel packs are sometimes helpful.
When it comes to intercourse, I recommend adequate lubrication using any of a number of effective products, such as olive oil, vitamin E oil, Replens, Slippery Stuff, Astroglide, KY Liquid, and others.
There is an extensive list of lubricants at http://www.med.umich.edu/sexualhealth/resources/guide.htm
Topical agents might offer relief—but so might placebo
Dr. Lonky: What is the role of topical medications, including anesthetics, for treating vulvar pain syndromes?
Dr. Edwards: I don’t find topical medications to be particularly useful in the treatment of vulvodynia, except for lidocaine 2% jelly, or lidocaine 5% ointment, which tends to burn with application—but I never start a patient on only one medication, so judging the effectiveness of a topical therapy is difficult in that context. Good studies of topical medications in the treatment of vulvar pain syndromes are lacking, other than the recent report on amitriptyline and baclofen.1
Dr. Haefner: For minor degrees of pain, consider lidocaine 5% ointment.
Lidocaine/prilocaine (eutectic mixture of local anesthesia or LMX) may be used but can be irritating.
Doxepin 5% cream can be applied to skin daily, gradually increasing the number of daily applications to as many as four.
Topical amitriptyline 2% with baclofen 2% in a water washable base has also been used for point tenderness (squirt 0.5 cc from a syringe onto the finger and apply it to the affected area three times a day).1
Dr. Gunter: Topical estrogen is prescribed by many providers, but we lack studies supporting its efficacy, except for reversing hypoestrogenic changes in postmenopausal women. Some providers use a high-dose, compounded topical estrogen with lidocaine for vestibulodynia. Certainly, local hypoestrogenic changes should be reversed in postmenopausal women before a diagnosis of vulvodynia or vestibulodynia is given.
As for other topical therapies, they are widely used. Some women report improvement with application of plain petrolatum.2 Response rates of 33% to 46% after use of a topical placebo for vestibulodynia are well described in the literature.3,4
Topical analgesics are used frequently, either sporadically (during pain flares) or regularly (daily application). One method of application for localized vestibulodynia involves liberally coating a cotton ball with lidocaine 5% and then applying it to the vestibule overnight (for at least 8 hours of exposure). In this study, after 7 weeks, 76% of women were able to be sexually active, compared with 36% before the start of treatment. However, a randomized, placebo-controlled trial that included lidocaine 5% cream in one arm identified only a 20% reduction in pain for women who had localized vestibulodynia—although, in this trial, the lidocaine was massaged into the vestibule four times daily.5 In this study, interestingly enough, topical lidocaine was less effective than topical placebo, which produced a 33% response rate.3
Lidocaine gel has also been used, although some women report more local irritation with gel than with ointment.
Dr. Lonky: Do we have any data on topical application of other drugs?
Dr. Gunter: Compounded adjuvant medications have been evaluated. In a retrospective study of topical gabapentin in a Lipoderm base, women who had generalized or localized vulvodynia applied a dose of 2%, 4%, or 6% three times daily. Of these women, 80% experienced a reduction of at least 50% in the pain score. In addition, 67% of patients who had localized vestibular pain were able to resume intercourse.6
A retrospective review of 38 women who used 2% amitriptyline and 2% baclofen in a Lipoderm cream for localized vestibular pain found that 53% experienced an improvement in symptoms of at least 60%, but there was no change in the frequency of sexual intercourse.1
Do tricyclic antidepressants ease chronic pain?
Dr. Lonky: Let’s talk, for a moment, about the use of oral tricyclic antidepressants in the treatment of vulvar pain syndromes. What do we know?
Dr. Haefner: Tricyclic antidepressants are a common treatment for vulvar pain. This group of drugs (including amitriptyline [Elavil], nortriptyline [Pamelor], and desipramine [Norpramin]) has been used to treat many idiopathic chronic pain conditions. Published and presented reports indicate that these drugs elicit about a 60% response rate for various pain conditions. A trial by the National Institutes of Health (NIH) is under way, analyzing the use of antidepressants in women who have vulvar pain.
Although treatment with tricyclic antidepressants has generally been reserved for women who have generalized vulvodynia, recent reports have found these medications to be helpful in the treatment of vestibular pain as well. The mechanism of action is thought to be related to inhibition of the reuptake of transmitters—specifically, norepinephrine and serotonin. However, the mechanism of action may be more closely related to anticholinergic effects. Tricyclics affect sodium channels and the N-methyl-d-aspartate (NMDA) receptor.
If you choose to prescribe one of these medications, consider emphasizing to the patient its effect on the sensation of pain rather than its effect on depression.
Dr. Lonky: Are there any types of patients who should not take a tricyclic?
Dr. Haefner: Yes. A patient should not take a tricyclic if she is pregnant, breastfeeding, or planning to conceive. These medicines also add to the effects of alcohol and other central nervous system depressants.
Dr. Lonky: What dosage is recommended?
Dr. Haefner: The dosage for pain control varies, depending on the age of the patient and the particular agent used. Amitriptyline is often used as a first-line medication. I start the patient on 10 to 25 mg nightly and increase that amount by 10 to 25 mg weekly, not to exceed 150 mg daily. A sample regimen might be 10 mg at bedtime for 1 week. If symptoms persist, increase the dose to 20 mg at bedtime for another week, and so on. Once a dose is established that provides relief, the patient should continue to take that amount nightly. Advise the patient not to discontinue the drug abruptly. Rather, it should be weaned.
In patients who are 60 years or older, I give a starting dose of 5 to 10 mg and increase it by 10 mg weekly.
In all age groups, it is important to advise patients to avoid consuming more than one alcoholic beverage daily while taking this medication. And in reproductive-age women, contraception is critical.
Dr. Edwards: I call these drugs tricyclic medications rather than antidepressants. They are extremely useful in managing the neuropathic component of vulvar pain. Despite a recent, apparently well-conducted study showing a lack of benefit, my 25 years of personal clinical experience with tricyclics convince me that I should wait for follow-up studies before abandoning this therapy.3
The pain literature reports that higher doses than previously reported of tricyclic medications are needed for optimal management of neuropathic pain. Doses from 100 to 150 mg are often required for substantial improvement, and a major design flaw in many studies of the effect of tricyclic medications on vulvodynia is the use of an insufficient dose.
Because of their low cost and their effectiveness, tricyclic medications are my first-line therapy for women who do not suffer severe constipation or dry eyes. The effect on depression is a useful side effect, I find.
Dr. Gunter: Although adjuvant medications, such as antidepressants and anticonvulsants, are considered by more than 80% of practitioners to be effective for vulvodynia, it is important to understand that only one randomized, double-blind, placebo-controlled prospective study has evaluated this approach, and that study found a placebo response rate of 33%.3,7
Randomized studies indicate that low-dose amitriptyline (10–20 mg) and desipramine (150 mg) are ineffective for provoked vestibulodynia.3,8 Cohort and retrospective studies with higher doses of amitriptyline (40 to 60 mg/day) indicate that improvement in pain scores of 50% or more can be achieved for 47% to 59% of women who have localized provoked vestibulodynia and generalized unprovoked vulvodynia.9-11
Tricyclic antidepressants and anticonvulsants should be prescribed with caution for patients 65 years and older because they increase the risk of falls.
I give nortriptyline as a first-line agent to women who have both provoked and unprovoked pain. In general, it has fewer anticholinergic side effects than amitriptyline and is generic—it also is taken once daily. For women who have unprovoked pain, I use gabapentin as a second-line agent.
Dr. Lonky: Are any other antidepressants useful in the treatment of vulvar pain?
Dr. Haefner: I sometimes give duloxetine [Cymbalta], starting with an oral daily dose of 30 mg for 1 week. If symptoms persist, I increase the daily dose to a total of 60 mg. (If the patient is depressed, I have her take 30 mg twice daily; if she isn’t depressed, I have her take the full dose of 60 mg in the morning.) I also occasionally utilize venlafaxine [Effexor XR] for pain control, starting with an oral morning dose of 37.5 mg. This dose can be increased to 75 mg/day.
Dr. Edwards: Literature on venlafaxine for neuropathic pain suggests maximal effects at doses of 150 to 225 mg of the extended release formulation, which is often well tolerated. I start patients on 37.5 mg and increase weekly until I reach the 150-mg threshold.12
Are anticonvulsants effective pain relievers?
Dr. Lonky: How effective are oral anticonvulsants such as gabapentin [Neurontin]?
Dr. Haefner: Gabapentin has been used to treat chronic pain conditions. The drug is available in 100-mg, 300-mg, 400-mg, 600-mg, and 800-mg tablets. It is typically initiated at an oral dose of 300 mg daily for 3 days. The dosage is then increased to 300 mg twice daily for 3 days and, finally, to 300 mg three times daily. If necessary, it can gradually be increased to a total of 3,600 mg daily (usually divided into three doses). No more than 1,200 mg should be administered in a single dose. Side effects include somnolence, mental changes, dizziness, and weight gain.
Dr. Edwards: After tricyclic medications, I find gabapentin to be the most beneficial and easily tolerated agent. I give it to patients who have contraindications to tricyclics and who lack a strong component of depression.
Dr. Gunter: Retrospective reviews have found gabapentin to produce improvement of 80% or more in pain scores for 64% to 82% of women who have generalized unprovoked vulvodynia. And a small open-label, prospective trial of lamotrigine [Lamictal] found that it produced statistically significant improvement for generalized vulvodynia.13-15
Dr. Lonky: What do you know about the use of the anticonvulsants pregabalin [Lyrica] and topiramate [Topamax] to treat vulvar pain syndromes?
Dr. Gunter: Pregabalin was reported to reduce symptoms by 80% for generalized, unprovoked vulvodynia in one case report.16
Dr. Edwards: I find that pregabalin is less well tolerated (and more expensive) than gabapentin, so it is one of the last agents I prescribe. I reported a small, uncontrolled series of patients who were treated with pregabalin. Of those who tolerated the drug, two thirds of the women improved by approximately 62%, as judged by a visual analog scale.
Dr. Haefner: Pregabalin is a relatively new addition to the armamentarium. I give 50 mg orally for 4 days to start. If symptoms persist, I increase the dose to 50 mg twice a day for 4 days. If symptoms still persist, I up the dose again to 50 mg three times daily and gradually increase it to 100 mg three times daily, if necessary. Some reports describe a dose as high as 300 mg twice daily (maximum).
As for topiramate, I have been using it much more frequently for vulvodynia and noticing many fewer side effects than with gabapentin.
you offer them?
A patient who has a short duration of pain often responds to topical medications. In contrast, someone who has experienced pain for years is unlikely to get adequate relief from topical medications alone. These patients often require oral tricyclic antidepressants or anticonvulsants, or both. I often start these medications before deciding whether physical therapy is necessary. If the drugs do not provide adequate relief, then I refer the patient to physical therapy.
In some cases, I begin with physical therapy and add other treatments, if necessary. A patient who has localized vulvodynia who tightens her bulbocavernosus and levator ani muscles upon gentle touch may benefit from starting with physical therapy.
I reserve surgery—vestibulectomy—for the patient who has localized pain that has not responded to numerous treatments.
—Hope K. Haefner, MD
Does capsaicin interrupt the pain circuit?
Dr. Lonky: Capsaicin has been mentioned in the literature as a therapy for vulvar pain. Is it effective? How does it work?
Dr. Haefner: Capsaicin activates A-delta sensory neurons and unmyelinated C fibers. It is a vanillyl amide that evokes the sensation of burning pain. It has been proposed as a means of desensitization, which occurs as an acute reaction mediated by neuropeptides (including substance P).17 Steinberg and colleagues found that topical capsaicin significantly decreased pain with intercourse.17 Patients applied capsaicin 0.025% cream for 20 minutes daily for 12 weeks.
In a study by Murina and colleagues, 33 women were treated with topical capsaicin 0.05%. The capsaicin cream was applied to the vulva twice daily for 30 days, then once daily for 30 days, then twice weekly for 4 months. In this study, however, the response to treatment was only partial.18
Dr. Edwards: I have never had a patient willing to try capsaicin after I describe the therapy to them.
Dr. Gunter: Two studies have evaluated daily applications of capsaicin in concentrations of 0.025% and 0.05%—one of them the study by Murina and colleagues that Dr. Haefner mentioned.2,18 The initial release of substance P causes significant burning on application, so pretreatment with local anesthetic to help the patient tolerate the capsaicin is recommended, which could potentially confound the results. In one study, daily pain scores, as well as pain with intercourse, improved significantly for 59% of participants, but no patient experienced complete resolution of symptoms—and within 2 weeks after capsaicin was discontinued, symptoms returned.2,18 I have had only one patient in 15 years of practice who was willing to try capsaicin and who could get past the initial burning.
Another application for botulinum toxin type A?
Dr. Lonky: Is botulinum toxin type A [Botox] at all effective?
Dr. Haefner: Botulinum toxin type A has been utilized to treat provoked vestibulodynia as well as vaginismus and was beneficial.19-23 It blocks the cholinergic innervation of the target tissue. The therapeutic dose ranges from 20 IU to 300 IU.24
A placebo-controlled trial found that injection of 20 IU of botulinum toxin into the vestibule of women with vestibulodynia did not reduce pain, improve sexual functioning, or impact the quality of life, compared with placebo.25 However, this study utilized a lower dose of botulinum toxin than was used in many of the other studies.
Dr. Edwards: I have only used botulinum toxin type A in a low dose. I injected 6 IU of botulinum toxin A into the periphery of the vestibule at 3, 6, 9, and 12 o’clock in six patients, and half improved modestly. I am not prepared to use electromyography (EMG) localization in my office, but from anecdotal reports, as well as several small series and placebo-controlled trials, I would conclude that some patients improve. Those who have hypertonic pelvic floor muscles are likely to be the best candidates for this treatment.
Because the agent relieves pain only modestly, and because it is not covered by insurance for this application, I refer the patient to a gynecologist in my area who administers the drug under EMG localization.
Dr. Gunter: Given the well-documented effect on muscle spasticity, as well as studies that suggest they are also anti-nociceptive agents, botulinum toxins are certainly an attractive concept for vulvodynia. A small case series and a case report indicated significant improvement with vestibular injections of 20 to 40 U of botulinum toxin. However, a randomized, placebo-controlled, double-blind study indicated no significant improvement for women with localized vestibular pain.25-27
I discuss botulinum toxin A with my patients. I explain that my clinical experience does differ from results published in the literature. I find that many women with vestibulodynia opt to try an injection before proceeding to vestibulectomy.
When combined with pelvic floor physical therapy, botulinum toxins are highly effective at treating muscle spasm and can be very useful for women who have a component to their pain of vaginismus or high-tone pelvic floor dysfunction.
How useful are steroids and nerve blocks?
Dr. Lonky: Is there a role for local injections of glucocorticoids or serial nerve blocks?
Dr. Edwards: The occasional patient with very localized pain (trigger point) responds fairly well in my office to intralesional corticosteroids. I have not used or seen reports describing administration of intralesional corticosteroids into a larger area, although two practitioners have told me informally that it is useful in their hands.
As a dermatologist, I cannot perform blocks. I have referred patients to gyn and pain clinics for this purpose, but neither venue has been willing to administer the blocks.
Dr. Haefner: Patients who present with small, localized areas of pain may benefit from local injections. In small areas—for example, a painful spot 1 cm in diameter—triamcinolone acetonide in combination with bupivacaine may be helpful. It is important to use a small dose of steroid in a small area, however, because tissue erosion or ulceration can occur with too high a dose of steroid in the skin. For large areas, as much as 40 mg of triamcinolone acetonide may be utilized in a single monthly dose. Generally, the dose is repeated monthly, if necessary, as many as three or four times.
For large areas, bupivacaine 0.25% is utilized, and for small areas, bupivacaine 0.5% is injected into the vulva along with the steroid. The steroid should be drawn into the syringe first (because the vial can be used at a later time), followed by bupivacaine, which is a single-dose vial.
Pudendal nerve blocks using bupivacaine have been helpful in some patients—particularly those who have unilateral pain.
Dr. Gunter: Local injections with a variety of agents for localized, provoked vestibulodynia have been described, including steroids, botulinum toxins, and interferon. It is important to interpret these studies with caution, however, as placebo response rates with injection therapy are significant.25
A retrospective review of submucous injections of methylprednisolone and lidocaine found that 68% of women had a complete or marked response, and two case reports describe success with betamethasone.26,28-30 I offer steroid injections to women who have vestibulodynia before proceeding to vestibulectomy; I find that about 50% get at least partial relief. As Dr. Haefner indicated from her own experience, my success seems best with steroid injections in the vestibule when the painful area is smaller.
Using injectable steroids and botulinum toxins, I estimate that I can prevent 33% to 40% of vestibulectomies. Although this may not be better than the placebo response rate and certainly represents biased patient selection (patients are not required to try local injection before vestibulectomy for vestibulodynia), those who are successful are uniformly happy for trying it, and those for whom it did not work are not unhappy that they “gave it a try,” as these women are all motivated to avoid surgery, if possible.
Pudendal nerve blocks with triamcinolone are also described for women who have generalized unprovoked vulvodynia due to suspected pudendal neuralgia. Ganglion impar blocks—steroid injection around the terminal branch of the sympathetic chain in the presacral space—have also been performed with good results for generalized vulvodynia. I have had good success with pudendal nerve blocks for unilateral pain that is suspected to be pudendal in origin and also with ganglion impar blocks for women with generalized vulvodynia, especially postmenopausal women. I perform all of my own nerve blocks (FIGURE).
Ganglion impar block
Ganglion impar blocks—steroid injection around the terminal branch of the sympathetic chain in the presacral space—may provide relief from generalized vulvodynia in some women.
Does physical therapy play a role in easing vulvar pain?
Dr. Lonky: What is the role of physical therapy and pelvic floor muscle rehabilitation?
Dr. Gunter: All women who have high-tone pelvic floor dysfunction should be referred to a physical therapist. Many women who lack muscle spasm but experience vulvar pain can still benefit from physical therapy, as gentle stretching and vibration therapy can sometimes be helpful. A physical therapist can also perform biofeedback.
Dr. Edwards: Physical therapy is crucial. It is my first-line therapy overall, with adjunctive oral medication for neuropathic pain. Besides addressing pelvic floor abnormalities, physical therapy can serve as desensitization therapy and psychological support.
Dr. Haefner: Physical therapy has been successful in the treatment of a number of disorders, including migraine and tension headaches, asthma, and anxiety disorders. It is also used in the treatment of vulvar pain. Physical therapists who have experience in vulvar pain may be extremely helpful, particularly if there is concomitant vaginismus—which isn’t uncommon in this population.
For vulvodynia, techniques include internal (vaginal and rectal) and external soft-tissue mobilization and myofascial release; trigger-point pressure; visceral, urogenital, and joint manipulation; electrical stimulation; therapeutic exercises; active pelvic floor retraining; biofeedback; bladder and bowel retraining; and therapeutic ultrasound.
Biofeedback may be used to assist in developing self-regulation strategies for confronting and reducing pain. Patients who have vestibular pain in general have an increased resting tone and a decreased contraction tone. With the aid of an electronic measurement and amplification system or biofeedback machine, an individual can view a display of numbers on a meter, or colored lights, to assess nerve and muscle tension. In this way, she may be able to develop voluntary control over the biological systems involved in pain, discomfort, and disease.
The duration of physical therapy overall and the frequency of visits varies from person to person. Success rates in the range of 60% to 80% have been reported.
How should we respond when medical treatment fails?
Dr. Lonky: What is the proper approach to the patient who has recalcitrant vulvar pain who fails all medical treatments and is not a candidate for vestibulectomy because her pain is outside the vestibule?
Dr. Edwards: The pain clinic. Actually, in a perfect world, the role of the gynecologist or dermatologist would be to give the patient a diagnosis, after which a pain clinic would offer treatment.
All patients should receive counseling. And clinicians who lack expertise should refer the patient to a vulvodynia specialist.
Dr. Haefner: This type of patient may benefit from physical therapy. Bupivacaine steroid injections could also be considered. A sacral nerve stimulator should be considered if the other measures fail to provide adequate relief.
I agree that counseling is extremely helpful in the patient who has vulvodynia. Sexual counseling, with tips on positions for intercourse, lubricants, and control of uncomfortable situations, is of utmost importance.
Dr. Gunter: I offer oral medications and nerve blocks (typically, ganglion impar blocks), and many patients do well.
I also highly recommend advanced programs for mind-body techniques.
Patients who fail all therapies may be candidates for a nerve stimulator, depending on psychiatric comorbidities and response to selective diagnostic nerve blocks.
What’s in the pipeline?
Dr. Lonky: What therapies for vulvar pain are on the horizon?
Dr. Edwards: I believe that cognitive behavioral therapy, sex therapy, and couple counseling will play a larger role in the management of vulvar pain.
Dr. Gunter: Any therapy used in other pain conditions will probably eventually find its way to the management of vulvodynia.
Some investigators believe that Tarlov cysts play a role in vulvar pain and recommend that all women undergo sacral spine and nerve-root magnetic resonance imaging. The problem is that many asymptomatic women have Tarlov cysts, and the surgery to remove them is not at all risk-free. I strongly believe that more research is needed before we can suggest that Tarlov cysts be removed.
Dr. Haefner: Transcutaneous electrical stimulation and biofeedback have been used successfully in the treatment of vulvodynia.31 Some patients benefit from spinal cord stimulators, such as the sacral nerve stimulator, for pain control. Sacral nerve modulation (SNM) works primarily by modulation of the nerve signals to and from the pelvic floor muscles, bladder, and rectum. It applies low-amplitude electrical stimulation to the third sacral nerve via electrodes in a tined lead passing through the S3 foramen, which contains afferent sensory, efferent autonomic motor, and voluntary somatic nerves. Other studies have utilized a different spinal cord level. More studies are needed to demonstrate the full effect of SNM on vulvodynia.
A comprehensive review of the various treatments for vulvodynia can be found in the Journal of Lower Genital Tract Disease.2
Dr. Lonky: Thanks again for your expertise. We’ll focus on provoked vestibulodynia in the final installment of this series on vulvar pain, in the November 2011 issue of OBG Management.
Vulvar pain therapies mentioned in this discussion
| Lifestyle changes Cotton and/or wicking underwear Avoidance of vulvar irritants, douches, soap Use of lubricants during intercourse |
| Physical therapy Internal (vaginal and rectal) and external soft-tissue mobilization and myofascial release Trigger-point pressure Visceral, urogenital, and joint manipulation Electrical stimulation Therapeutic exercises Active pelvic floor retraining Biofeedback Bladder and bowel retraining Therapeutic ultrasound |
| Topical agents Lidocaine 2% jelly Lidocaine 5% ointment Lidocaine/prilocaine Doxepin 5% cream Amitriptyline 2%/baclofen 2% Estrogen Petrolatum Gabapentin |
Oral agents Tricyclic antidepressants
Other antidepressants
Anticonvulsants
|
| Other agents Capsaicin Botulinum toxin type A Corticosteroids Nerve block |
We want to hear from you! Tell us what you think.
1. Nyirjesy P, Lev-Sagie A, Mathew L, Culhane JF. Topical amitriptyline-baclofen cream for the treatment of provoked vestibulodynia. J Lower Genital Tract Dis. 2009;13(4):230-236.
2. Haefner HK, Collins ME, Davis GD, et al. The vulvodynia guideline. J Lower Genital Tract Dis. 2005;9(1):40-51.
3. Foster DC, Kotok MB, Huang L, et al. Oral desipramine and topical lidocaine for vulvodynia: a randomized controlled trial. Obstet Gynecol. 2010;116(3):583-593.
4. Nyirjesy P, Sobel JD, Weitz MV, Leaman DJ, Small MJ, Gelone SP. Cromolyn cream for recalcitrant idiopathic vulvar vestibulitis: results of a placebo-controlled trial. Sex Transm Infect. 2001;77(1):53-57.
5. Zolnoun DA, Hartmann KE, Steege JF. Overnight 5% lidocaine ointment for treatment of vulvar vestibulitis. Obstet Gynecol. 2003;102(1):84-87.
6. Boardman LA, Cooper AS, Blais LR, Raker CA. Topical gabapentin in the treatment of localized and generalized vulvodynia. Obstet Gynecol. 2008;112(3):579-585.
7. Reed BD, Haefner HK, Edwards L. A survey on diagnosis and treatment of vulvodynia researchers and members of the International Society for the Study of Vulvovaginal Disease. J Reprod Med. 2008;53(12):921-929.
8. Brown CS, Wan J, Bachmann G, Rosen R. Self-management amitriptyline, and amitriptyline plus triamcinolone in the management of vulvodynia. J Women Health (Larchmt). 2009;18(2):163-169.
9. Reed BD, Caron AM, Gorenflo DW, Haefner HK. Treatment of vulvodynia with tricyclic antidepressants: efficacy and associated factors. J Low Genit Tract Dis. 2006;10(4):245-251.
10. Munday PE. Response to treatment in dysaesthetic vulvodynia. J Obstet Gynecol. 2001;21(6):610-613.
11. McKay M. Dysesthetic (“essential”) vulvodynia: treatment with amitriptyline. J Reprod Med. 1993;38(1):9-13.
12. Dworkin RH, O’Connor AB, Backonja M, et al. Pharmacologic management of neuropathic pain: evidence-based recommendations. Pain. 2007;132(3):237-251.
13. Harris G, Horowitz B, Borgida A. Evaluation of gabapentin in the treatment of generalized vulvodynia unprovoked. J Reprod Med. 2007;52(2):103-106.
14. Ben-David B, Friedman M. Gabapentin therapy for vulvodynia. Anesth Analg. 1999;89(6):1459-1460.
15. Meltzer-Brody SE, Zolnoun D, Steege JF, Rinaldi KL, Leserman J. Open-label trial of lamotrigine focusing on efficacy in vulvodynia. J Reprod Med. 2009;54(3):171-178.
16. Jerome L. Pregabalin-induced remission in a 62-year-old woman with a 20-year history of vulvodynia. Pain Res Manag. 2007;12(3):212-214.
17. Steinberg AC, Oyama IA, Rejba AE, Kellogg-Spadt S, Whitmore KE. Capsaicin for the treatment of vulvar vestibulitis. Am J Obstet Gynecol. 2005;192(5):1549-1553.
18. Murina F, Radici G, Bianco V. Capsaicin and the treatment of vulvar vestibulitis syndrome: a valuable alternative? Med General Med. 2004;6(4):48.-
19. Park AJ, Paraiso MFR. Successful use of botulinum toxin type A in the treatment of refractory postoperative dyspareunia. Obstet Gynecol. 2009;114(2 pt 2):484-487.
20. Brown CS, Glazer HI, Vogt V, Menkes D, Bachmann G. Subjective and objective outcomes of botulinum toxin type A treatment in vestibulodynia: pilot data. J Reprod Med. 2006;51(8):635-641.
21. Dykstra DD, Presthus J. Botulinum toxin type A for the treatment of provoked vestibulodynia: an open-label pilot study. J Reprod Med. 2006;51(6):467-470.
22. Romito S, Bottanelli M, Pellegrini M, Vicentini S, Rizzuto N, Bertolasi L. Botulinum toxin for the treatment of genital pain syndromes. Gynecol Obstet Invest. 2004;58(3):164-167.
23. Bertolasi L, Frasson E, Cappelletti JY, Vicentini S, Bordignon M, Graziottin A. Botulinum neurotoxin type A injections for vaginismus secondary to vulvar vestibulitis syndrome. Obstet Gynecol. 2009;114(5):1008-1016.
24. Presthus JB, Dykstra DD. Botulinum toxin therapy for vulvodynia. NVA News. National Vulvodynia Association. 2007;12(3).
25. Peterson CD, Giraldi A, Lundvall L, Kristensen E. Botulinum toxin type A—a novel treatment for provoked vestibulodynia? Results from a randomized placebo-controlled, double-blinded study. J Sex Med. 2009;6(9):2523-2537.
26. Yoon H, Chung WS, Shim BS. Botulinum toxin A for the management of vulvodynia. Int J Impot Res. 2007;19(1):84-87.
27. Gunter J, Brewer A, Tawfik O. Botulinum toxin A for vulvodynia: a case report. J Pain. 2004;5(4):238-240.
28. Segal D, Tifheret H, Lazr S. Submucous infiltration of betamethasone and lidocaine in the treatment of vulvar vestibulitis. Eur J Obstet Gynecol Reprod Biol. 2003;107(1):105-106.
29. Murina F, Tassan P, Roberti P, Bianco V. Treatment of vulvar vestibulitis with submucous infiltrations of methylprednisolone and lidocaine. An alternative approach. J Reprod Med. 2001;46(8):713-716.
30. Dede M, Yenen MC, Yilmaz A, Baser I. Successful treatment of persistent vulvodynia with submucous infiltration of betamethasone and lidocaine. Eur J Obstet Gynecol Reprod Biol. 2006;124(2):258-259.
31. Dionisi B, Anglana F, Inghirami P, Lippa P, Senatori R. Use of transcutaneous electrical stimulation and biofeedback for the treatment of vulvodynia (vulvar vestibular syndrome): results of 3 years of experience [in Italian]. Minerva Ginecol. 2008;60(6):485-491.
- Part 1: Making the correct diagnosis
(September 2011) - Part 3: Vestibulodynia
(November 2011)
As we discussed in the first installment of this three-part series in the September issue of OBG Management, the causes of vulvar pain are many, and the diagnosis of this common complaint can be difficult. Once the diagnosis of vulvodynia has been made, however, the challenge shifts to finding an effective treatment. Here, our expert panel discusses the many options available, the data (or lack of it) behind each therapy, and what to do in refractory cases.
In Part 3 of this series, in the November issue, the focus will be vestibulodynia.

The lower vagina and vulva are richly supplied with peripheral nerves and are, therefore, sensitive to pain, particularly the region of the hymeneal ring. Although the pudendal nerve (arrow) courses through the area, it is an uncommon source of vulvar pain.
Management of vulvar pain begins with simple measures
Dr. Lonky: How do you approach treatment of vulvar pain syndromes?
Dr. Haefner: I often advise the patient to begin with simple measures. For example, I recommend that she wear cotton underwear during the day, but no underwear at night. If she perspires with exercise, wicking underwear may be helpful. I also counsel the patient to avoid vulvar irritants, douches, and the application of soap of any kind to the vulva. Cool gel packs are sometimes helpful.
When it comes to intercourse, I recommend adequate lubrication using any of a number of effective products, such as olive oil, vitamin E oil, Replens, Slippery Stuff, Astroglide, KY Liquid, and others.
There is an extensive list of lubricants at http://www.med.umich.edu/sexualhealth/resources/guide.htm
Topical agents might offer relief—but so might placebo
Dr. Lonky: What is the role of topical medications, including anesthetics, for treating vulvar pain syndromes?
Dr. Edwards: I don’t find topical medications to be particularly useful in the treatment of vulvodynia, except for lidocaine 2% jelly, or lidocaine 5% ointment, which tends to burn with application—but I never start a patient on only one medication, so judging the effectiveness of a topical therapy is difficult in that context. Good studies of topical medications in the treatment of vulvar pain syndromes are lacking, other than the recent report on amitriptyline and baclofen.1
Dr. Haefner: For minor degrees of pain, consider lidocaine 5% ointment.
Lidocaine/prilocaine (eutectic mixture of local anesthesia or LMX) may be used but can be irritating.
Doxepin 5% cream can be applied to skin daily, gradually increasing the number of daily applications to as many as four.
Topical amitriptyline 2% with baclofen 2% in a water washable base has also been used for point tenderness (squirt 0.5 cc from a syringe onto the finger and apply it to the affected area three times a day).1
Dr. Gunter: Topical estrogen is prescribed by many providers, but we lack studies supporting its efficacy, except for reversing hypoestrogenic changes in postmenopausal women. Some providers use a high-dose, compounded topical estrogen with lidocaine for vestibulodynia. Certainly, local hypoestrogenic changes should be reversed in postmenopausal women before a diagnosis of vulvodynia or vestibulodynia is given.
As for other topical therapies, they are widely used. Some women report improvement with application of plain petrolatum.2 Response rates of 33% to 46% after use of a topical placebo for vestibulodynia are well described in the literature.3,4
Topical analgesics are used frequently, either sporadically (during pain flares) or regularly (daily application). One method of application for localized vestibulodynia involves liberally coating a cotton ball with lidocaine 5% and then applying it to the vestibule overnight (for at least 8 hours of exposure). In this study, after 7 weeks, 76% of women were able to be sexually active, compared with 36% before the start of treatment. However, a randomized, placebo-controlled trial that included lidocaine 5% cream in one arm identified only a 20% reduction in pain for women who had localized vestibulodynia—although, in this trial, the lidocaine was massaged into the vestibule four times daily.5 In this study, interestingly enough, topical lidocaine was less effective than topical placebo, which produced a 33% response rate.3
Lidocaine gel has also been used, although some women report more local irritation with gel than with ointment.
Dr. Lonky: Do we have any data on topical application of other drugs?
Dr. Gunter: Compounded adjuvant medications have been evaluated. In a retrospective study of topical gabapentin in a Lipoderm base, women who had generalized or localized vulvodynia applied a dose of 2%, 4%, or 6% three times daily. Of these women, 80% experienced a reduction of at least 50% in the pain score. In addition, 67% of patients who had localized vestibular pain were able to resume intercourse.6
A retrospective review of 38 women who used 2% amitriptyline and 2% baclofen in a Lipoderm cream for localized vestibular pain found that 53% experienced an improvement in symptoms of at least 60%, but there was no change in the frequency of sexual intercourse.1
Do tricyclic antidepressants ease chronic pain?
Dr. Lonky: Let’s talk, for a moment, about the use of oral tricyclic antidepressants in the treatment of vulvar pain syndromes. What do we know?
Dr. Haefner: Tricyclic antidepressants are a common treatment for vulvar pain. This group of drugs (including amitriptyline [Elavil], nortriptyline [Pamelor], and desipramine [Norpramin]) has been used to treat many idiopathic chronic pain conditions. Published and presented reports indicate that these drugs elicit about a 60% response rate for various pain conditions. A trial by the National Institutes of Health (NIH) is under way, analyzing the use of antidepressants in women who have vulvar pain.
Although treatment with tricyclic antidepressants has generally been reserved for women who have generalized vulvodynia, recent reports have found these medications to be helpful in the treatment of vestibular pain as well. The mechanism of action is thought to be related to inhibition of the reuptake of transmitters—specifically, norepinephrine and serotonin. However, the mechanism of action may be more closely related to anticholinergic effects. Tricyclics affect sodium channels and the N-methyl-d-aspartate (NMDA) receptor.
If you choose to prescribe one of these medications, consider emphasizing to the patient its effect on the sensation of pain rather than its effect on depression.
Dr. Lonky: Are there any types of patients who should not take a tricyclic?
Dr. Haefner: Yes. A patient should not take a tricyclic if she is pregnant, breastfeeding, or planning to conceive. These medicines also add to the effects of alcohol and other central nervous system depressants.
Dr. Lonky: What dosage is recommended?
Dr. Haefner: The dosage for pain control varies, depending on the age of the patient and the particular agent used. Amitriptyline is often used as a first-line medication. I start the patient on 10 to 25 mg nightly and increase that amount by 10 to 25 mg weekly, not to exceed 150 mg daily. A sample regimen might be 10 mg at bedtime for 1 week. If symptoms persist, increase the dose to 20 mg at bedtime for another week, and so on. Once a dose is established that provides relief, the patient should continue to take that amount nightly. Advise the patient not to discontinue the drug abruptly. Rather, it should be weaned.
In patients who are 60 years or older, I give a starting dose of 5 to 10 mg and increase it by 10 mg weekly.
In all age groups, it is important to advise patients to avoid consuming more than one alcoholic beverage daily while taking this medication. And in reproductive-age women, contraception is critical.
Dr. Edwards: I call these drugs tricyclic medications rather than antidepressants. They are extremely useful in managing the neuropathic component of vulvar pain. Despite a recent, apparently well-conducted study showing a lack of benefit, my 25 years of personal clinical experience with tricyclics convince me that I should wait for follow-up studies before abandoning this therapy.3
The pain literature reports that higher doses than previously reported of tricyclic medications are needed for optimal management of neuropathic pain. Doses from 100 to 150 mg are often required for substantial improvement, and a major design flaw in many studies of the effect of tricyclic medications on vulvodynia is the use of an insufficient dose.
Because of their low cost and their effectiveness, tricyclic medications are my first-line therapy for women who do not suffer severe constipation or dry eyes. The effect on depression is a useful side effect, I find.
Dr. Gunter: Although adjuvant medications, such as antidepressants and anticonvulsants, are considered by more than 80% of practitioners to be effective for vulvodynia, it is important to understand that only one randomized, double-blind, placebo-controlled prospective study has evaluated this approach, and that study found a placebo response rate of 33%.3,7
Randomized studies indicate that low-dose amitriptyline (10–20 mg) and desipramine (150 mg) are ineffective for provoked vestibulodynia.3,8 Cohort and retrospective studies with higher doses of amitriptyline (40 to 60 mg/day) indicate that improvement in pain scores of 50% or more can be achieved for 47% to 59% of women who have localized provoked vestibulodynia and generalized unprovoked vulvodynia.9-11
Tricyclic antidepressants and anticonvulsants should be prescribed with caution for patients 65 years and older because they increase the risk of falls.
I give nortriptyline as a first-line agent to women who have both provoked and unprovoked pain. In general, it has fewer anticholinergic side effects than amitriptyline and is generic—it also is taken once daily. For women who have unprovoked pain, I use gabapentin as a second-line agent.
Dr. Lonky: Are any other antidepressants useful in the treatment of vulvar pain?
Dr. Haefner: I sometimes give duloxetine [Cymbalta], starting with an oral daily dose of 30 mg for 1 week. If symptoms persist, I increase the daily dose to a total of 60 mg. (If the patient is depressed, I have her take 30 mg twice daily; if she isn’t depressed, I have her take the full dose of 60 mg in the morning.) I also occasionally utilize venlafaxine [Effexor XR] for pain control, starting with an oral morning dose of 37.5 mg. This dose can be increased to 75 mg/day.
Dr. Edwards: Literature on venlafaxine for neuropathic pain suggests maximal effects at doses of 150 to 225 mg of the extended release formulation, which is often well tolerated. I start patients on 37.5 mg and increase weekly until I reach the 150-mg threshold.12
Are anticonvulsants effective pain relievers?
Dr. Lonky: How effective are oral anticonvulsants such as gabapentin [Neurontin]?
Dr. Haefner: Gabapentin has been used to treat chronic pain conditions. The drug is available in 100-mg, 300-mg, 400-mg, 600-mg, and 800-mg tablets. It is typically initiated at an oral dose of 300 mg daily for 3 days. The dosage is then increased to 300 mg twice daily for 3 days and, finally, to 300 mg three times daily. If necessary, it can gradually be increased to a total of 3,600 mg daily (usually divided into three doses). No more than 1,200 mg should be administered in a single dose. Side effects include somnolence, mental changes, dizziness, and weight gain.
Dr. Edwards: After tricyclic medications, I find gabapentin to be the most beneficial and easily tolerated agent. I give it to patients who have contraindications to tricyclics and who lack a strong component of depression.
Dr. Gunter: Retrospective reviews have found gabapentin to produce improvement of 80% or more in pain scores for 64% to 82% of women who have generalized unprovoked vulvodynia. And a small open-label, prospective trial of lamotrigine [Lamictal] found that it produced statistically significant improvement for generalized vulvodynia.13-15
Dr. Lonky: What do you know about the use of the anticonvulsants pregabalin [Lyrica] and topiramate [Topamax] to treat vulvar pain syndromes?
Dr. Gunter: Pregabalin was reported to reduce symptoms by 80% for generalized, unprovoked vulvodynia in one case report.16
Dr. Edwards: I find that pregabalin is less well tolerated (and more expensive) than gabapentin, so it is one of the last agents I prescribe. I reported a small, uncontrolled series of patients who were treated with pregabalin. Of those who tolerated the drug, two thirds of the women improved by approximately 62%, as judged by a visual analog scale.
Dr. Haefner: Pregabalin is a relatively new addition to the armamentarium. I give 50 mg orally for 4 days to start. If symptoms persist, I increase the dose to 50 mg twice a day for 4 days. If symptoms still persist, I up the dose again to 50 mg three times daily and gradually increase it to 100 mg three times daily, if necessary. Some reports describe a dose as high as 300 mg twice daily (maximum).
As for topiramate, I have been using it much more frequently for vulvodynia and noticing many fewer side effects than with gabapentin.
you offer them?
A patient who has a short duration of pain often responds to topical medications. In contrast, someone who has experienced pain for years is unlikely to get adequate relief from topical medications alone. These patients often require oral tricyclic antidepressants or anticonvulsants, or both. I often start these medications before deciding whether physical therapy is necessary. If the drugs do not provide adequate relief, then I refer the patient to physical therapy.
In some cases, I begin with physical therapy and add other treatments, if necessary. A patient who has localized vulvodynia who tightens her bulbocavernosus and levator ani muscles upon gentle touch may benefit from starting with physical therapy.
I reserve surgery—vestibulectomy—for the patient who has localized pain that has not responded to numerous treatments.
—Hope K. Haefner, MD
Does capsaicin interrupt the pain circuit?
Dr. Lonky: Capsaicin has been mentioned in the literature as a therapy for vulvar pain. Is it effective? How does it work?
Dr. Haefner: Capsaicin activates A-delta sensory neurons and unmyelinated C fibers. It is a vanillyl amide that evokes the sensation of burning pain. It has been proposed as a means of desensitization, which occurs as an acute reaction mediated by neuropeptides (including substance P).17 Steinberg and colleagues found that topical capsaicin significantly decreased pain with intercourse.17 Patients applied capsaicin 0.025% cream for 20 minutes daily for 12 weeks.
In a study by Murina and colleagues, 33 women were treated with topical capsaicin 0.05%. The capsaicin cream was applied to the vulva twice daily for 30 days, then once daily for 30 days, then twice weekly for 4 months. In this study, however, the response to treatment was only partial.18
Dr. Edwards: I have never had a patient willing to try capsaicin after I describe the therapy to them.
Dr. Gunter: Two studies have evaluated daily applications of capsaicin in concentrations of 0.025% and 0.05%—one of them the study by Murina and colleagues that Dr. Haefner mentioned.2,18 The initial release of substance P causes significant burning on application, so pretreatment with local anesthetic to help the patient tolerate the capsaicin is recommended, which could potentially confound the results. In one study, daily pain scores, as well as pain with intercourse, improved significantly for 59% of participants, but no patient experienced complete resolution of symptoms—and within 2 weeks after capsaicin was discontinued, symptoms returned.2,18 I have had only one patient in 15 years of practice who was willing to try capsaicin and who could get past the initial burning.
Another application for botulinum toxin type A?
Dr. Lonky: Is botulinum toxin type A [Botox] at all effective?
Dr. Haefner: Botulinum toxin type A has been utilized to treat provoked vestibulodynia as well as vaginismus and was beneficial.19-23 It blocks the cholinergic innervation of the target tissue. The therapeutic dose ranges from 20 IU to 300 IU.24
A placebo-controlled trial found that injection of 20 IU of botulinum toxin into the vestibule of women with vestibulodynia did not reduce pain, improve sexual functioning, or impact the quality of life, compared with placebo.25 However, this study utilized a lower dose of botulinum toxin than was used in many of the other studies.
Dr. Edwards: I have only used botulinum toxin type A in a low dose. I injected 6 IU of botulinum toxin A into the periphery of the vestibule at 3, 6, 9, and 12 o’clock in six patients, and half improved modestly. I am not prepared to use electromyography (EMG) localization in my office, but from anecdotal reports, as well as several small series and placebo-controlled trials, I would conclude that some patients improve. Those who have hypertonic pelvic floor muscles are likely to be the best candidates for this treatment.
Because the agent relieves pain only modestly, and because it is not covered by insurance for this application, I refer the patient to a gynecologist in my area who administers the drug under EMG localization.
Dr. Gunter: Given the well-documented effect on muscle spasticity, as well as studies that suggest they are also anti-nociceptive agents, botulinum toxins are certainly an attractive concept for vulvodynia. A small case series and a case report indicated significant improvement with vestibular injections of 20 to 40 U of botulinum toxin. However, a randomized, placebo-controlled, double-blind study indicated no significant improvement for women with localized vestibular pain.25-27
I discuss botulinum toxin A with my patients. I explain that my clinical experience does differ from results published in the literature. I find that many women with vestibulodynia opt to try an injection before proceeding to vestibulectomy.
When combined with pelvic floor physical therapy, botulinum toxins are highly effective at treating muscle spasm and can be very useful for women who have a component to their pain of vaginismus or high-tone pelvic floor dysfunction.
How useful are steroids and nerve blocks?
Dr. Lonky: Is there a role for local injections of glucocorticoids or serial nerve blocks?
Dr. Edwards: The occasional patient with very localized pain (trigger point) responds fairly well in my office to intralesional corticosteroids. I have not used or seen reports describing administration of intralesional corticosteroids into a larger area, although two practitioners have told me informally that it is useful in their hands.
As a dermatologist, I cannot perform blocks. I have referred patients to gyn and pain clinics for this purpose, but neither venue has been willing to administer the blocks.
Dr. Haefner: Patients who present with small, localized areas of pain may benefit from local injections. In small areas—for example, a painful spot 1 cm in diameter—triamcinolone acetonide in combination with bupivacaine may be helpful. It is important to use a small dose of steroid in a small area, however, because tissue erosion or ulceration can occur with too high a dose of steroid in the skin. For large areas, as much as 40 mg of triamcinolone acetonide may be utilized in a single monthly dose. Generally, the dose is repeated monthly, if necessary, as many as three or four times.
For large areas, bupivacaine 0.25% is utilized, and for small areas, bupivacaine 0.5% is injected into the vulva along with the steroid. The steroid should be drawn into the syringe first (because the vial can be used at a later time), followed by bupivacaine, which is a single-dose vial.
Pudendal nerve blocks using bupivacaine have been helpful in some patients—particularly those who have unilateral pain.
Dr. Gunter: Local injections with a variety of agents for localized, provoked vestibulodynia have been described, including steroids, botulinum toxins, and interferon. It is important to interpret these studies with caution, however, as placebo response rates with injection therapy are significant.25
A retrospective review of submucous injections of methylprednisolone and lidocaine found that 68% of women had a complete or marked response, and two case reports describe success with betamethasone.26,28-30 I offer steroid injections to women who have vestibulodynia before proceeding to vestibulectomy; I find that about 50% get at least partial relief. As Dr. Haefner indicated from her own experience, my success seems best with steroid injections in the vestibule when the painful area is smaller.
Using injectable steroids and botulinum toxins, I estimate that I can prevent 33% to 40% of vestibulectomies. Although this may not be better than the placebo response rate and certainly represents biased patient selection (patients are not required to try local injection before vestibulectomy for vestibulodynia), those who are successful are uniformly happy for trying it, and those for whom it did not work are not unhappy that they “gave it a try,” as these women are all motivated to avoid surgery, if possible.
Pudendal nerve blocks with triamcinolone are also described for women who have generalized unprovoked vulvodynia due to suspected pudendal neuralgia. Ganglion impar blocks—steroid injection around the terminal branch of the sympathetic chain in the presacral space—have also been performed with good results for generalized vulvodynia. I have had good success with pudendal nerve blocks for unilateral pain that is suspected to be pudendal in origin and also with ganglion impar blocks for women with generalized vulvodynia, especially postmenopausal women. I perform all of my own nerve blocks (FIGURE).

Ganglion impar block
Ganglion impar blocks—steroid injection around the terminal branch of the sympathetic chain in the presacral space—may provide relief from generalized vulvodynia in some women.
Does physical therapy play a role in easing vulvar pain?
Dr. Lonky: What is the role of physical therapy and pelvic floor muscle rehabilitation?
Dr. Gunter: All women who have high-tone pelvic floor dysfunction should be referred to a physical therapist. Many women who lack muscle spasm but experience vulvar pain can still benefit from physical therapy, as gentle stretching and vibration therapy can sometimes be helpful. A physical therapist can also perform biofeedback.
Dr. Edwards: Physical therapy is crucial. It is my first-line therapy overall, with adjunctive oral medication for neuropathic pain. Besides addressing pelvic floor abnormalities, physical therapy can serve as desensitization therapy and psychological support.
Dr. Haefner: Physical therapy has been successful in the treatment of a number of disorders, including migraine and tension headaches, asthma, and anxiety disorders. It is also used in the treatment of vulvar pain. Physical therapists who have experience in vulvar pain may be extremely helpful, particularly if there is concomitant vaginismus—which isn’t uncommon in this population.
For vulvodynia, techniques include internal (vaginal and rectal) and external soft-tissue mobilization and myofascial release; trigger-point pressure; visceral, urogenital, and joint manipulation; electrical stimulation; therapeutic exercises; active pelvic floor retraining; biofeedback; bladder and bowel retraining; and therapeutic ultrasound.
Biofeedback may be used to assist in developing self-regulation strategies for confronting and reducing pain. Patients who have vestibular pain in general have an increased resting tone and a decreased contraction tone. With the aid of an electronic measurement and amplification system or biofeedback machine, an individual can view a display of numbers on a meter, or colored lights, to assess nerve and muscle tension. In this way, she may be able to develop voluntary control over the biological systems involved in pain, discomfort, and disease.
The duration of physical therapy overall and the frequency of visits varies from person to person. Success rates in the range of 60% to 80% have been reported.
How should we respond when medical treatment fails?
Dr. Lonky: What is the proper approach to the patient who has recalcitrant vulvar pain who fails all medical treatments and is not a candidate for vestibulectomy because her pain is outside the vestibule?
Dr. Edwards: The pain clinic. Actually, in a perfect world, the role of the gynecologist or dermatologist would be to give the patient a diagnosis, after which a pain clinic would offer treatment.
All patients should receive counseling. And clinicians who lack expertise should refer the patient to a vulvodynia specialist.
Dr. Haefner: This type of patient may benefit from physical therapy. Bupivacaine steroid injections could also be considered. A sacral nerve stimulator should be considered if the other measures fail to provide adequate relief.
I agree that counseling is extremely helpful in the patient who has vulvodynia. Sexual counseling, with tips on positions for intercourse, lubricants, and control of uncomfortable situations, is of utmost importance.
Dr. Gunter: I offer oral medications and nerve blocks (typically, ganglion impar blocks), and many patients do well.
I also highly recommend advanced programs for mind-body techniques.
Patients who fail all therapies may be candidates for a nerve stimulator, depending on psychiatric comorbidities and response to selective diagnostic nerve blocks.
What’s in the pipeline?
Dr. Lonky: What therapies for vulvar pain are on the horizon?
Dr. Edwards: I believe that cognitive behavioral therapy, sex therapy, and couple counseling will play a larger role in the management of vulvar pain.
Dr. Gunter: Any therapy used in other pain conditions will probably eventually find its way to the management of vulvodynia.
Some investigators believe that Tarlov cysts play a role in vulvar pain and recommend that all women undergo sacral spine and nerve-root magnetic resonance imaging. The problem is that many asymptomatic women have Tarlov cysts, and the surgery to remove them is not at all risk-free. I strongly believe that more research is needed before we can suggest that Tarlov cysts be removed.
Dr. Haefner: Transcutaneous electrical stimulation and biofeedback have been used successfully in the treatment of vulvodynia.31 Some patients benefit from spinal cord stimulators, such as the sacral nerve stimulator, for pain control. Sacral nerve modulation (SNM) works primarily by modulation of the nerve signals to and from the pelvic floor muscles, bladder, and rectum. It applies low-amplitude electrical stimulation to the third sacral nerve via electrodes in a tined lead passing through the S3 foramen, which contains afferent sensory, efferent autonomic motor, and voluntary somatic nerves. Other studies have utilized a different spinal cord level. More studies are needed to demonstrate the full effect of SNM on vulvodynia.
A comprehensive review of the various treatments for vulvodynia can be found in the Journal of Lower Genital Tract Disease.2
Dr. Lonky: Thanks again for your expertise. We’ll focus on provoked vestibulodynia in the final installment of this series on vulvar pain, in the November 2011 issue of OBG Management.
Vulvar pain therapies mentioned in this discussion
| Lifestyle changes Cotton and/or wicking underwear Avoidance of vulvar irritants, douches, soap Use of lubricants during intercourse |
| Physical therapy Internal (vaginal and rectal) and external soft-tissue mobilization and myofascial release Trigger-point pressure Visceral, urogenital, and joint manipulation Electrical stimulation Therapeutic exercises Active pelvic floor retraining Biofeedback Bladder and bowel retraining Therapeutic ultrasound |
| Topical agents Lidocaine 2% jelly Lidocaine 5% ointment Lidocaine/prilocaine Doxepin 5% cream Amitriptyline 2%/baclofen 2% Estrogen Petrolatum Gabapentin |
Oral agents Tricyclic antidepressants
Other antidepressants
Anticonvulsants
|
| Other agents Capsaicin Botulinum toxin type A Corticosteroids Nerve block |
We want to hear from you! Tell us what you think.
- Part 1: Making the correct diagnosis
(September 2011) - Part 3: Vestibulodynia
(November 2011)
As we discussed in the first installment of this three-part series in the September issue of OBG Management, the causes of vulvar pain are many, and the diagnosis of this common complaint can be difficult. Once the diagnosis of vulvodynia has been made, however, the challenge shifts to finding an effective treatment. Here, our expert panel discusses the many options available, the data (or lack of it) behind each therapy, and what to do in refractory cases.
In Part 3 of this series, in the November issue, the focus will be vestibulodynia.

The lower vagina and vulva are richly supplied with peripheral nerves and are, therefore, sensitive to pain, particularly the region of the hymeneal ring. Although the pudendal nerve (arrow) courses through the area, it is an uncommon source of vulvar pain.
Management of vulvar pain begins with simple measures
Dr. Lonky: How do you approach treatment of vulvar pain syndromes?
Dr. Haefner: I often advise the patient to begin with simple measures. For example, I recommend that she wear cotton underwear during the day, but no underwear at night. If she perspires with exercise, wicking underwear may be helpful. I also counsel the patient to avoid vulvar irritants, douches, and the application of soap of any kind to the vulva. Cool gel packs are sometimes helpful.
When it comes to intercourse, I recommend adequate lubrication using any of a number of effective products, such as olive oil, vitamin E oil, Replens, Slippery Stuff, Astroglide, KY Liquid, and others.
There is an extensive list of lubricants at http://www.med.umich.edu/sexualhealth/resources/guide.htm
Topical agents might offer relief—but so might placebo
Dr. Lonky: What is the role of topical medications, including anesthetics, for treating vulvar pain syndromes?
Dr. Edwards: I don’t find topical medications to be particularly useful in the treatment of vulvodynia, except for lidocaine 2% jelly, or lidocaine 5% ointment, which tends to burn with application—but I never start a patient on only one medication, so judging the effectiveness of a topical therapy is difficult in that context. Good studies of topical medications in the treatment of vulvar pain syndromes are lacking, other than the recent report on amitriptyline and baclofen.1
Dr. Haefner: For minor degrees of pain, consider lidocaine 5% ointment.
Lidocaine/prilocaine (eutectic mixture of local anesthesia or LMX) may be used but can be irritating.
Doxepin 5% cream can be applied to skin daily, gradually increasing the number of daily applications to as many as four.
Topical amitriptyline 2% with baclofen 2% in a water washable base has also been used for point tenderness (squirt 0.5 cc from a syringe onto the finger and apply it to the affected area three times a day).1
Dr. Gunter: Topical estrogen is prescribed by many providers, but we lack studies supporting its efficacy, except for reversing hypoestrogenic changes in postmenopausal women. Some providers use a high-dose, compounded topical estrogen with lidocaine for vestibulodynia. Certainly, local hypoestrogenic changes should be reversed in postmenopausal women before a diagnosis of vulvodynia or vestibulodynia is given.
As for other topical therapies, they are widely used. Some women report improvement with application of plain petrolatum.2 Response rates of 33% to 46% after use of a topical placebo for vestibulodynia are well described in the literature.3,4
Topical analgesics are used frequently, either sporadically (during pain flares) or regularly (daily application). One method of application for localized vestibulodynia involves liberally coating a cotton ball with lidocaine 5% and then applying it to the vestibule overnight (for at least 8 hours of exposure). In this study, after 7 weeks, 76% of women were able to be sexually active, compared with 36% before the start of treatment. However, a randomized, placebo-controlled trial that included lidocaine 5% cream in one arm identified only a 20% reduction in pain for women who had localized vestibulodynia—although, in this trial, the lidocaine was massaged into the vestibule four times daily.5 In this study, interestingly enough, topical lidocaine was less effective than topical placebo, which produced a 33% response rate.3
Lidocaine gel has also been used, although some women report more local irritation with gel than with ointment.
Dr. Lonky: Do we have any data on topical application of other drugs?
Dr. Gunter: Compounded adjuvant medications have been evaluated. In a retrospective study of topical gabapentin in a Lipoderm base, women who had generalized or localized vulvodynia applied a dose of 2%, 4%, or 6% three times daily. Of these women, 80% experienced a reduction of at least 50% in the pain score. In addition, 67% of patients who had localized vestibular pain were able to resume intercourse.6
A retrospective review of 38 women who used 2% amitriptyline and 2% baclofen in a Lipoderm cream for localized vestibular pain found that 53% experienced an improvement in symptoms of at least 60%, but there was no change in the frequency of sexual intercourse.1
Do tricyclic antidepressants ease chronic pain?
Dr. Lonky: Let’s talk, for a moment, about the use of oral tricyclic antidepressants in the treatment of vulvar pain syndromes. What do we know?
Dr. Haefner: Tricyclic antidepressants are a common treatment for vulvar pain. This group of drugs (including amitriptyline [Elavil], nortriptyline [Pamelor], and desipramine [Norpramin]) has been used to treat many idiopathic chronic pain conditions. Published and presented reports indicate that these drugs elicit about a 60% response rate for various pain conditions. A trial by the National Institutes of Health (NIH) is under way, analyzing the use of antidepressants in women who have vulvar pain.
Although treatment with tricyclic antidepressants has generally been reserved for women who have generalized vulvodynia, recent reports have found these medications to be helpful in the treatment of vestibular pain as well. The mechanism of action is thought to be related to inhibition of the reuptake of transmitters—specifically, norepinephrine and serotonin. However, the mechanism of action may be more closely related to anticholinergic effects. Tricyclics affect sodium channels and the N-methyl-d-aspartate (NMDA) receptor.
If you choose to prescribe one of these medications, consider emphasizing to the patient its effect on the sensation of pain rather than its effect on depression.
Dr. Lonky: Are there any types of patients who should not take a tricyclic?
Dr. Haefner: Yes. A patient should not take a tricyclic if she is pregnant, breastfeeding, or planning to conceive. These medicines also add to the effects of alcohol and other central nervous system depressants.
Dr. Lonky: What dosage is recommended?
Dr. Haefner: The dosage for pain control varies, depending on the age of the patient and the particular agent used. Amitriptyline is often used as a first-line medication. I start the patient on 10 to 25 mg nightly and increase that amount by 10 to 25 mg weekly, not to exceed 150 mg daily. A sample regimen might be 10 mg at bedtime for 1 week. If symptoms persist, increase the dose to 20 mg at bedtime for another week, and so on. Once a dose is established that provides relief, the patient should continue to take that amount nightly. Advise the patient not to discontinue the drug abruptly. Rather, it should be weaned.
In patients who are 60 years or older, I give a starting dose of 5 to 10 mg and increase it by 10 mg weekly.
In all age groups, it is important to advise patients to avoid consuming more than one alcoholic beverage daily while taking this medication. And in reproductive-age women, contraception is critical.
Dr. Edwards: I call these drugs tricyclic medications rather than antidepressants. They are extremely useful in managing the neuropathic component of vulvar pain. Despite a recent, apparently well-conducted study showing a lack of benefit, my 25 years of personal clinical experience with tricyclics convince me that I should wait for follow-up studies before abandoning this therapy.3
The pain literature reports that higher doses than previously reported of tricyclic medications are needed for optimal management of neuropathic pain. Doses from 100 to 150 mg are often required for substantial improvement, and a major design flaw in many studies of the effect of tricyclic medications on vulvodynia is the use of an insufficient dose.
Because of their low cost and their effectiveness, tricyclic medications are my first-line therapy for women who do not suffer severe constipation or dry eyes. The effect on depression is a useful side effect, I find.
Dr. Gunter: Although adjuvant medications, such as antidepressants and anticonvulsants, are considered by more than 80% of practitioners to be effective for vulvodynia, it is important to understand that only one randomized, double-blind, placebo-controlled prospective study has evaluated this approach, and that study found a placebo response rate of 33%.3,7
Randomized studies indicate that low-dose amitriptyline (10–20 mg) and desipramine (150 mg) are ineffective for provoked vestibulodynia.3,8 Cohort and retrospective studies with higher doses of amitriptyline (40 to 60 mg/day) indicate that improvement in pain scores of 50% or more can be achieved for 47% to 59% of women who have localized provoked vestibulodynia and generalized unprovoked vulvodynia.9-11
Tricyclic antidepressants and anticonvulsants should be prescribed with caution for patients 65 years and older because they increase the risk of falls.
I give nortriptyline as a first-line agent to women who have both provoked and unprovoked pain. In general, it has fewer anticholinergic side effects than amitriptyline and is generic—it also is taken once daily. For women who have unprovoked pain, I use gabapentin as a second-line agent.
Dr. Lonky: Are any other antidepressants useful in the treatment of vulvar pain?
Dr. Haefner: I sometimes give duloxetine [Cymbalta], starting with an oral daily dose of 30 mg for 1 week. If symptoms persist, I increase the daily dose to a total of 60 mg. (If the patient is depressed, I have her take 30 mg twice daily; if she isn’t depressed, I have her take the full dose of 60 mg in the morning.) I also occasionally utilize venlafaxine [Effexor XR] for pain control, starting with an oral morning dose of 37.5 mg. This dose can be increased to 75 mg/day.
Dr. Edwards: Literature on venlafaxine for neuropathic pain suggests maximal effects at doses of 150 to 225 mg of the extended release formulation, which is often well tolerated. I start patients on 37.5 mg and increase weekly until I reach the 150-mg threshold.12
Are anticonvulsants effective pain relievers?
Dr. Lonky: How effective are oral anticonvulsants such as gabapentin [Neurontin]?
Dr. Haefner: Gabapentin has been used to treat chronic pain conditions. The drug is available in 100-mg, 300-mg, 400-mg, 600-mg, and 800-mg tablets. It is typically initiated at an oral dose of 300 mg daily for 3 days. The dosage is then increased to 300 mg twice daily for 3 days and, finally, to 300 mg three times daily. If necessary, it can gradually be increased to a total of 3,600 mg daily (usually divided into three doses). No more than 1,200 mg should be administered in a single dose. Side effects include somnolence, mental changes, dizziness, and weight gain.
Dr. Edwards: After tricyclic medications, I find gabapentin to be the most beneficial and easily tolerated agent. I give it to patients who have contraindications to tricyclics and who lack a strong component of depression.
Dr. Gunter: Retrospective reviews have found gabapentin to produce improvement of 80% or more in pain scores for 64% to 82% of women who have generalized unprovoked vulvodynia. And a small open-label, prospective trial of lamotrigine [Lamictal] found that it produced statistically significant improvement for generalized vulvodynia.13-15
Dr. Lonky: What do you know about the use of the anticonvulsants pregabalin [Lyrica] and topiramate [Topamax] to treat vulvar pain syndromes?
Dr. Gunter: Pregabalin was reported to reduce symptoms by 80% for generalized, unprovoked vulvodynia in one case report.16
Dr. Edwards: I find that pregabalin is less well tolerated (and more expensive) than gabapentin, so it is one of the last agents I prescribe. I reported a small, uncontrolled series of patients who were treated with pregabalin. Of those who tolerated the drug, two thirds of the women improved by approximately 62%, as judged by a visual analog scale.
Dr. Haefner: Pregabalin is a relatively new addition to the armamentarium. I give 50 mg orally for 4 days to start. If symptoms persist, I increase the dose to 50 mg twice a day for 4 days. If symptoms still persist, I up the dose again to 50 mg three times daily and gradually increase it to 100 mg three times daily, if necessary. Some reports describe a dose as high as 300 mg twice daily (maximum).
As for topiramate, I have been using it much more frequently for vulvodynia and noticing many fewer side effects than with gabapentin.
you offer them?
A patient who has a short duration of pain often responds to topical medications. In contrast, someone who has experienced pain for years is unlikely to get adequate relief from topical medications alone. These patients often require oral tricyclic antidepressants or anticonvulsants, or both. I often start these medications before deciding whether physical therapy is necessary. If the drugs do not provide adequate relief, then I refer the patient to physical therapy.
In some cases, I begin with physical therapy and add other treatments, if necessary. A patient who has localized vulvodynia who tightens her bulbocavernosus and levator ani muscles upon gentle touch may benefit from starting with physical therapy.
I reserve surgery—vestibulectomy—for the patient who has localized pain that has not responded to numerous treatments.
—Hope K. Haefner, MD
Does capsaicin interrupt the pain circuit?
Dr. Lonky: Capsaicin has been mentioned in the literature as a therapy for vulvar pain. Is it effective? How does it work?
Dr. Haefner: Capsaicin activates A-delta sensory neurons and unmyelinated C fibers. It is a vanillyl amide that evokes the sensation of burning pain. It has been proposed as a means of desensitization, which occurs as an acute reaction mediated by neuropeptides (including substance P).17 Steinberg and colleagues found that topical capsaicin significantly decreased pain with intercourse.17 Patients applied capsaicin 0.025% cream for 20 minutes daily for 12 weeks.
In a study by Murina and colleagues, 33 women were treated with topical capsaicin 0.05%. The capsaicin cream was applied to the vulva twice daily for 30 days, then once daily for 30 days, then twice weekly for 4 months. In this study, however, the response to treatment was only partial.18
Dr. Edwards: I have never had a patient willing to try capsaicin after I describe the therapy to them.
Dr. Gunter: Two studies have evaluated daily applications of capsaicin in concentrations of 0.025% and 0.05%—one of them the study by Murina and colleagues that Dr. Haefner mentioned.2,18 The initial release of substance P causes significant burning on application, so pretreatment with local anesthetic to help the patient tolerate the capsaicin is recommended, which could potentially confound the results. In one study, daily pain scores, as well as pain with intercourse, improved significantly for 59% of participants, but no patient experienced complete resolution of symptoms—and within 2 weeks after capsaicin was discontinued, symptoms returned.2,18 I have had only one patient in 15 years of practice who was willing to try capsaicin and who could get past the initial burning.
Another application for botulinum toxin type A?
Dr. Lonky: Is botulinum toxin type A [Botox] at all effective?
Dr. Haefner: Botulinum toxin type A has been utilized to treat provoked vestibulodynia as well as vaginismus and was beneficial.19-23 It blocks the cholinergic innervation of the target tissue. The therapeutic dose ranges from 20 IU to 300 IU.24
A placebo-controlled trial found that injection of 20 IU of botulinum toxin into the vestibule of women with vestibulodynia did not reduce pain, improve sexual functioning, or impact the quality of life, compared with placebo.25 However, this study utilized a lower dose of botulinum toxin than was used in many of the other studies.
Dr. Edwards: I have only used botulinum toxin type A in a low dose. I injected 6 IU of botulinum toxin A into the periphery of the vestibule at 3, 6, 9, and 12 o’clock in six patients, and half improved modestly. I am not prepared to use electromyography (EMG) localization in my office, but from anecdotal reports, as well as several small series and placebo-controlled trials, I would conclude that some patients improve. Those who have hypertonic pelvic floor muscles are likely to be the best candidates for this treatment.
Because the agent relieves pain only modestly, and because it is not covered by insurance for this application, I refer the patient to a gynecologist in my area who administers the drug under EMG localization.
Dr. Gunter: Given the well-documented effect on muscle spasticity, as well as studies that suggest they are also anti-nociceptive agents, botulinum toxins are certainly an attractive concept for vulvodynia. A small case series and a case report indicated significant improvement with vestibular injections of 20 to 40 U of botulinum toxin. However, a randomized, placebo-controlled, double-blind study indicated no significant improvement for women with localized vestibular pain.25-27
I discuss botulinum toxin A with my patients. I explain that my clinical experience does differ from results published in the literature. I find that many women with vestibulodynia opt to try an injection before proceeding to vestibulectomy.
When combined with pelvic floor physical therapy, botulinum toxins are highly effective at treating muscle spasm and can be very useful for women who have a component to their pain of vaginismus or high-tone pelvic floor dysfunction.
How useful are steroids and nerve blocks?
Dr. Lonky: Is there a role for local injections of glucocorticoids or serial nerve blocks?
Dr. Edwards: The occasional patient with very localized pain (trigger point) responds fairly well in my office to intralesional corticosteroids. I have not used or seen reports describing administration of intralesional corticosteroids into a larger area, although two practitioners have told me informally that it is useful in their hands.
As a dermatologist, I cannot perform blocks. I have referred patients to gyn and pain clinics for this purpose, but neither venue has been willing to administer the blocks.
Dr. Haefner: Patients who present with small, localized areas of pain may benefit from local injections. In small areas—for example, a painful spot 1 cm in diameter—triamcinolone acetonide in combination with bupivacaine may be helpful. It is important to use a small dose of steroid in a small area, however, because tissue erosion or ulceration can occur with too high a dose of steroid in the skin. For large areas, as much as 40 mg of triamcinolone acetonide may be utilized in a single monthly dose. Generally, the dose is repeated monthly, if necessary, as many as three or four times.
For large areas, bupivacaine 0.25% is utilized, and for small areas, bupivacaine 0.5% is injected into the vulva along with the steroid. The steroid should be drawn into the syringe first (because the vial can be used at a later time), followed by bupivacaine, which is a single-dose vial.
Pudendal nerve blocks using bupivacaine have been helpful in some patients—particularly those who have unilateral pain.
Dr. Gunter: Local injections with a variety of agents for localized, provoked vestibulodynia have been described, including steroids, botulinum toxins, and interferon. It is important to interpret these studies with caution, however, as placebo response rates with injection therapy are significant.25
A retrospective review of submucous injections of methylprednisolone and lidocaine found that 68% of women had a complete or marked response, and two case reports describe success with betamethasone.26,28-30 I offer steroid injections to women who have vestibulodynia before proceeding to vestibulectomy; I find that about 50% get at least partial relief. As Dr. Haefner indicated from her own experience, my success seems best with steroid injections in the vestibule when the painful area is smaller.
Using injectable steroids and botulinum toxins, I estimate that I can prevent 33% to 40% of vestibulectomies. Although this may not be better than the placebo response rate and certainly represents biased patient selection (patients are not required to try local injection before vestibulectomy for vestibulodynia), those who are successful are uniformly happy for trying it, and those for whom it did not work are not unhappy that they “gave it a try,” as these women are all motivated to avoid surgery, if possible.
Pudendal nerve blocks with triamcinolone are also described for women who have generalized unprovoked vulvodynia due to suspected pudendal neuralgia. Ganglion impar blocks—steroid injection around the terminal branch of the sympathetic chain in the presacral space—have also been performed with good results for generalized vulvodynia. I have had good success with pudendal nerve blocks for unilateral pain that is suspected to be pudendal in origin and also with ganglion impar blocks for women with generalized vulvodynia, especially postmenopausal women. I perform all of my own nerve blocks (FIGURE).

Ganglion impar block
Ganglion impar blocks—steroid injection around the terminal branch of the sympathetic chain in the presacral space—may provide relief from generalized vulvodynia in some women.
Does physical therapy play a role in easing vulvar pain?
Dr. Lonky: What is the role of physical therapy and pelvic floor muscle rehabilitation?
Dr. Gunter: All women who have high-tone pelvic floor dysfunction should be referred to a physical therapist. Many women who lack muscle spasm but experience vulvar pain can still benefit from physical therapy, as gentle stretching and vibration therapy can sometimes be helpful. A physical therapist can also perform biofeedback.
Dr. Edwards: Physical therapy is crucial. It is my first-line therapy overall, with adjunctive oral medication for neuropathic pain. Besides addressing pelvic floor abnormalities, physical therapy can serve as desensitization therapy and psychological support.
Dr. Haefner: Physical therapy has been successful in the treatment of a number of disorders, including migraine and tension headaches, asthma, and anxiety disorders. It is also used in the treatment of vulvar pain. Physical therapists who have experience in vulvar pain may be extremely helpful, particularly if there is concomitant vaginismus—which isn’t uncommon in this population.
For vulvodynia, techniques include internal (vaginal and rectal) and external soft-tissue mobilization and myofascial release; trigger-point pressure; visceral, urogenital, and joint manipulation; electrical stimulation; therapeutic exercises; active pelvic floor retraining; biofeedback; bladder and bowel retraining; and therapeutic ultrasound.
Biofeedback may be used to assist in developing self-regulation strategies for confronting and reducing pain. Patients who have vestibular pain in general have an increased resting tone and a decreased contraction tone. With the aid of an electronic measurement and amplification system or biofeedback machine, an individual can view a display of numbers on a meter, or colored lights, to assess nerve and muscle tension. In this way, she may be able to develop voluntary control over the biological systems involved in pain, discomfort, and disease.
The duration of physical therapy overall and the frequency of visits varies from person to person. Success rates in the range of 60% to 80% have been reported.
How should we respond when medical treatment fails?
Dr. Lonky: What is the proper approach to the patient who has recalcitrant vulvar pain who fails all medical treatments and is not a candidate for vestibulectomy because her pain is outside the vestibule?
Dr. Edwards: The pain clinic. Actually, in a perfect world, the role of the gynecologist or dermatologist would be to give the patient a diagnosis, after which a pain clinic would offer treatment.
All patients should receive counseling. And clinicians who lack expertise should refer the patient to a vulvodynia specialist.
Dr. Haefner: This type of patient may benefit from physical therapy. Bupivacaine steroid injections could also be considered. A sacral nerve stimulator should be considered if the other measures fail to provide adequate relief.
I agree that counseling is extremely helpful in the patient who has vulvodynia. Sexual counseling, with tips on positions for intercourse, lubricants, and control of uncomfortable situations, is of utmost importance.
Dr. Gunter: I offer oral medications and nerve blocks (typically, ganglion impar blocks), and many patients do well.
I also highly recommend advanced programs for mind-body techniques.
Patients who fail all therapies may be candidates for a nerve stimulator, depending on psychiatric comorbidities and response to selective diagnostic nerve blocks.
What’s in the pipeline?
Dr. Lonky: What therapies for vulvar pain are on the horizon?
Dr. Edwards: I believe that cognitive behavioral therapy, sex therapy, and couple counseling will play a larger role in the management of vulvar pain.
Dr. Gunter: Any therapy used in other pain conditions will probably eventually find its way to the management of vulvodynia.
Some investigators believe that Tarlov cysts play a role in vulvar pain and recommend that all women undergo sacral spine and nerve-root magnetic resonance imaging. The problem is that many asymptomatic women have Tarlov cysts, and the surgery to remove them is not at all risk-free. I strongly believe that more research is needed before we can suggest that Tarlov cysts be removed.
Dr. Haefner: Transcutaneous electrical stimulation and biofeedback have been used successfully in the treatment of vulvodynia.31 Some patients benefit from spinal cord stimulators, such as the sacral nerve stimulator, for pain control. Sacral nerve modulation (SNM) works primarily by modulation of the nerve signals to and from the pelvic floor muscles, bladder, and rectum. It applies low-amplitude electrical stimulation to the third sacral nerve via electrodes in a tined lead passing through the S3 foramen, which contains afferent sensory, efferent autonomic motor, and voluntary somatic nerves. Other studies have utilized a different spinal cord level. More studies are needed to demonstrate the full effect of SNM on vulvodynia.
A comprehensive review of the various treatments for vulvodynia can be found in the Journal of Lower Genital Tract Disease.2
Dr. Lonky: Thanks again for your expertise. We’ll focus on provoked vestibulodynia in the final installment of this series on vulvar pain, in the November 2011 issue of OBG Management.
Vulvar pain therapies mentioned in this discussion
| Lifestyle changes Cotton and/or wicking underwear Avoidance of vulvar irritants, douches, soap Use of lubricants during intercourse |
| Physical therapy Internal (vaginal and rectal) and external soft-tissue mobilization and myofascial release Trigger-point pressure Visceral, urogenital, and joint manipulation Electrical stimulation Therapeutic exercises Active pelvic floor retraining Biofeedback Bladder and bowel retraining Therapeutic ultrasound |
| Topical agents Lidocaine 2% jelly Lidocaine 5% ointment Lidocaine/prilocaine Doxepin 5% cream Amitriptyline 2%/baclofen 2% Estrogen Petrolatum Gabapentin |
Oral agents Tricyclic antidepressants
Other antidepressants
Anticonvulsants
|
| Other agents Capsaicin Botulinum toxin type A Corticosteroids Nerve block |
We want to hear from you! Tell us what you think.
1. Nyirjesy P, Lev-Sagie A, Mathew L, Culhane JF. Topical amitriptyline-baclofen cream for the treatment of provoked vestibulodynia. J Lower Genital Tract Dis. 2009;13(4):230-236.
2. Haefner HK, Collins ME, Davis GD, et al. The vulvodynia guideline. J Lower Genital Tract Dis. 2005;9(1):40-51.
3. Foster DC, Kotok MB, Huang L, et al. Oral desipramine and topical lidocaine for vulvodynia: a randomized controlled trial. Obstet Gynecol. 2010;116(3):583-593.
4. Nyirjesy P, Sobel JD, Weitz MV, Leaman DJ, Small MJ, Gelone SP. Cromolyn cream for recalcitrant idiopathic vulvar vestibulitis: results of a placebo-controlled trial. Sex Transm Infect. 2001;77(1):53-57.
5. Zolnoun DA, Hartmann KE, Steege JF. Overnight 5% lidocaine ointment for treatment of vulvar vestibulitis. Obstet Gynecol. 2003;102(1):84-87.
6. Boardman LA, Cooper AS, Blais LR, Raker CA. Topical gabapentin in the treatment of localized and generalized vulvodynia. Obstet Gynecol. 2008;112(3):579-585.
7. Reed BD, Haefner HK, Edwards L. A survey on diagnosis and treatment of vulvodynia researchers and members of the International Society for the Study of Vulvovaginal Disease. J Reprod Med. 2008;53(12):921-929.
8. Brown CS, Wan J, Bachmann G, Rosen R. Self-management amitriptyline, and amitriptyline plus triamcinolone in the management of vulvodynia. J Women Health (Larchmt). 2009;18(2):163-169.
9. Reed BD, Caron AM, Gorenflo DW, Haefner HK. Treatment of vulvodynia with tricyclic antidepressants: efficacy and associated factors. J Low Genit Tract Dis. 2006;10(4):245-251.
10. Munday PE. Response to treatment in dysaesthetic vulvodynia. J Obstet Gynecol. 2001;21(6):610-613.
11. McKay M. Dysesthetic (“essential”) vulvodynia: treatment with amitriptyline. J Reprod Med. 1993;38(1):9-13.
12. Dworkin RH, O’Connor AB, Backonja M, et al. Pharmacologic management of neuropathic pain: evidence-based recommendations. Pain. 2007;132(3):237-251.
13. Harris G, Horowitz B, Borgida A. Evaluation of gabapentin in the treatment of generalized vulvodynia unprovoked. J Reprod Med. 2007;52(2):103-106.
14. Ben-David B, Friedman M. Gabapentin therapy for vulvodynia. Anesth Analg. 1999;89(6):1459-1460.
15. Meltzer-Brody SE, Zolnoun D, Steege JF, Rinaldi KL, Leserman J. Open-label trial of lamotrigine focusing on efficacy in vulvodynia. J Reprod Med. 2009;54(3):171-178.
16. Jerome L. Pregabalin-induced remission in a 62-year-old woman with a 20-year history of vulvodynia. Pain Res Manag. 2007;12(3):212-214.
17. Steinberg AC, Oyama IA, Rejba AE, Kellogg-Spadt S, Whitmore KE. Capsaicin for the treatment of vulvar vestibulitis. Am J Obstet Gynecol. 2005;192(5):1549-1553.
18. Murina F, Radici G, Bianco V. Capsaicin and the treatment of vulvar vestibulitis syndrome: a valuable alternative? Med General Med. 2004;6(4):48.-
19. Park AJ, Paraiso MFR. Successful use of botulinum toxin type A in the treatment of refractory postoperative dyspareunia. Obstet Gynecol. 2009;114(2 pt 2):484-487.
20. Brown CS, Glazer HI, Vogt V, Menkes D, Bachmann G. Subjective and objective outcomes of botulinum toxin type A treatment in vestibulodynia: pilot data. J Reprod Med. 2006;51(8):635-641.
21. Dykstra DD, Presthus J. Botulinum toxin type A for the treatment of provoked vestibulodynia: an open-label pilot study. J Reprod Med. 2006;51(6):467-470.
22. Romito S, Bottanelli M, Pellegrini M, Vicentini S, Rizzuto N, Bertolasi L. Botulinum toxin for the treatment of genital pain syndromes. Gynecol Obstet Invest. 2004;58(3):164-167.
23. Bertolasi L, Frasson E, Cappelletti JY, Vicentini S, Bordignon M, Graziottin A. Botulinum neurotoxin type A injections for vaginismus secondary to vulvar vestibulitis syndrome. Obstet Gynecol. 2009;114(5):1008-1016.
24. Presthus JB, Dykstra DD. Botulinum toxin therapy for vulvodynia. NVA News. National Vulvodynia Association. 2007;12(3).
25. Peterson CD, Giraldi A, Lundvall L, Kristensen E. Botulinum toxin type A—a novel treatment for provoked vestibulodynia? Results from a randomized placebo-controlled, double-blinded study. J Sex Med. 2009;6(9):2523-2537.
26. Yoon H, Chung WS, Shim BS. Botulinum toxin A for the management of vulvodynia. Int J Impot Res. 2007;19(1):84-87.
27. Gunter J, Brewer A, Tawfik O. Botulinum toxin A for vulvodynia: a case report. J Pain. 2004;5(4):238-240.
28. Segal D, Tifheret H, Lazr S. Submucous infiltration of betamethasone and lidocaine in the treatment of vulvar vestibulitis. Eur J Obstet Gynecol Reprod Biol. 2003;107(1):105-106.
29. Murina F, Tassan P, Roberti P, Bianco V. Treatment of vulvar vestibulitis with submucous infiltrations of methylprednisolone and lidocaine. An alternative approach. J Reprod Med. 2001;46(8):713-716.
30. Dede M, Yenen MC, Yilmaz A, Baser I. Successful treatment of persistent vulvodynia with submucous infiltration of betamethasone and lidocaine. Eur J Obstet Gynecol Reprod Biol. 2006;124(2):258-259.
31. Dionisi B, Anglana F, Inghirami P, Lippa P, Senatori R. Use of transcutaneous electrical stimulation and biofeedback for the treatment of vulvodynia (vulvar vestibular syndrome): results of 3 years of experience [in Italian]. Minerva Ginecol. 2008;60(6):485-491.
1. Nyirjesy P, Lev-Sagie A, Mathew L, Culhane JF. Topical amitriptyline-baclofen cream for the treatment of provoked vestibulodynia. J Lower Genital Tract Dis. 2009;13(4):230-236.
2. Haefner HK, Collins ME, Davis GD, et al. The vulvodynia guideline. J Lower Genital Tract Dis. 2005;9(1):40-51.
3. Foster DC, Kotok MB, Huang L, et al. Oral desipramine and topical lidocaine for vulvodynia: a randomized controlled trial. Obstet Gynecol. 2010;116(3):583-593.
4. Nyirjesy P, Sobel JD, Weitz MV, Leaman DJ, Small MJ, Gelone SP. Cromolyn cream for recalcitrant idiopathic vulvar vestibulitis: results of a placebo-controlled trial. Sex Transm Infect. 2001;77(1):53-57.
5. Zolnoun DA, Hartmann KE, Steege JF. Overnight 5% lidocaine ointment for treatment of vulvar vestibulitis. Obstet Gynecol. 2003;102(1):84-87.
6. Boardman LA, Cooper AS, Blais LR, Raker CA. Topical gabapentin in the treatment of localized and generalized vulvodynia. Obstet Gynecol. 2008;112(3):579-585.
7. Reed BD, Haefner HK, Edwards L. A survey on diagnosis and treatment of vulvodynia researchers and members of the International Society for the Study of Vulvovaginal Disease. J Reprod Med. 2008;53(12):921-929.
8. Brown CS, Wan J, Bachmann G, Rosen R. Self-management amitriptyline, and amitriptyline plus triamcinolone in the management of vulvodynia. J Women Health (Larchmt). 2009;18(2):163-169.
9. Reed BD, Caron AM, Gorenflo DW, Haefner HK. Treatment of vulvodynia with tricyclic antidepressants: efficacy and associated factors. J Low Genit Tract Dis. 2006;10(4):245-251.
10. Munday PE. Response to treatment in dysaesthetic vulvodynia. J Obstet Gynecol. 2001;21(6):610-613.
11. McKay M. Dysesthetic (“essential”) vulvodynia: treatment with amitriptyline. J Reprod Med. 1993;38(1):9-13.
12. Dworkin RH, O’Connor AB, Backonja M, et al. Pharmacologic management of neuropathic pain: evidence-based recommendations. Pain. 2007;132(3):237-251.
13. Harris G, Horowitz B, Borgida A. Evaluation of gabapentin in the treatment of generalized vulvodynia unprovoked. J Reprod Med. 2007;52(2):103-106.
14. Ben-David B, Friedman M. Gabapentin therapy for vulvodynia. Anesth Analg. 1999;89(6):1459-1460.
15. Meltzer-Brody SE, Zolnoun D, Steege JF, Rinaldi KL, Leserman J. Open-label trial of lamotrigine focusing on efficacy in vulvodynia. J Reprod Med. 2009;54(3):171-178.
16. Jerome L. Pregabalin-induced remission in a 62-year-old woman with a 20-year history of vulvodynia. Pain Res Manag. 2007;12(3):212-214.
17. Steinberg AC, Oyama IA, Rejba AE, Kellogg-Spadt S, Whitmore KE. Capsaicin for the treatment of vulvar vestibulitis. Am J Obstet Gynecol. 2005;192(5):1549-1553.
18. Murina F, Radici G, Bianco V. Capsaicin and the treatment of vulvar vestibulitis syndrome: a valuable alternative? Med General Med. 2004;6(4):48.-
19. Park AJ, Paraiso MFR. Successful use of botulinum toxin type A in the treatment of refractory postoperative dyspareunia. Obstet Gynecol. 2009;114(2 pt 2):484-487.
20. Brown CS, Glazer HI, Vogt V, Menkes D, Bachmann G. Subjective and objective outcomes of botulinum toxin type A treatment in vestibulodynia: pilot data. J Reprod Med. 2006;51(8):635-641.
21. Dykstra DD, Presthus J. Botulinum toxin type A for the treatment of provoked vestibulodynia: an open-label pilot study. J Reprod Med. 2006;51(6):467-470.
22. Romito S, Bottanelli M, Pellegrini M, Vicentini S, Rizzuto N, Bertolasi L. Botulinum toxin for the treatment of genital pain syndromes. Gynecol Obstet Invest. 2004;58(3):164-167.
23. Bertolasi L, Frasson E, Cappelletti JY, Vicentini S, Bordignon M, Graziottin A. Botulinum neurotoxin type A injections for vaginismus secondary to vulvar vestibulitis syndrome. Obstet Gynecol. 2009;114(5):1008-1016.
24. Presthus JB, Dykstra DD. Botulinum toxin therapy for vulvodynia. NVA News. National Vulvodynia Association. 2007;12(3).
25. Peterson CD, Giraldi A, Lundvall L, Kristensen E. Botulinum toxin type A—a novel treatment for provoked vestibulodynia? Results from a randomized placebo-controlled, double-blinded study. J Sex Med. 2009;6(9):2523-2537.
26. Yoon H, Chung WS, Shim BS. Botulinum toxin A for the management of vulvodynia. Int J Impot Res. 2007;19(1):84-87.
27. Gunter J, Brewer A, Tawfik O. Botulinum toxin A for vulvodynia: a case report. J Pain. 2004;5(4):238-240.
28. Segal D, Tifheret H, Lazr S. Submucous infiltration of betamethasone and lidocaine in the treatment of vulvar vestibulitis. Eur J Obstet Gynecol Reprod Biol. 2003;107(1):105-106.
29. Murina F, Tassan P, Roberti P, Bianco V. Treatment of vulvar vestibulitis with submucous infiltrations of methylprednisolone and lidocaine. An alternative approach. J Reprod Med. 2001;46(8):713-716.
30. Dede M, Yenen MC, Yilmaz A, Baser I. Successful treatment of persistent vulvodynia with submucous infiltration of betamethasone and lidocaine. Eur J Obstet Gynecol Reprod Biol. 2006;124(2):258-259.
31. Dionisi B, Anglana F, Inghirami P, Lippa P, Senatori R. Use of transcutaneous electrical stimulation and biofeedback for the treatment of vulvodynia (vulvar vestibular syndrome): results of 3 years of experience [in Italian]. Minerva Ginecol. 2008;60(6):485-491.
Does trocar-guided vaginal mesh improve the durability of repair of recurrent pelvic organ prolapse?
- Are new tools for correcting prolapse and incontinence better just because they’re new?
Anne M. Weber, MD, MS (Web Exclusive Commentary, February 2009)
A woman’s lifetime risk of surgery for pelvic organ prolapse is approximately 7%; more than 300,000 prolapse surgeries are performed annually in the United States alone.1 Of the women who undergo surgery, an estimated 13% will require re-operation within 5 years, and as many as 29% will undergo another surgery for genital prolapse or a related condition at some point during their life.2
In the hope of improving the effectiveness and durability of vaginal prolapse repairs, many pelvic reconstructive surgeons have turned to reinforcement with synthetic mesh. However, because of concern about a higher risk of complications relative to other approaches, the use of transvaginal mesh for prolapse repair has come under increased scrutiny by the FDA. At this time, almost one quarter of all prolapse repairs involve placement of transvaginal mesh.3
Details of the trial
Withagen and colleagues enrolled 194 women from 13 Dutch medical centers. All women had recurrent pelvic organ prolapse and were randomized to either trocar-guided transvaginal mesh repair (Prolift) or conventional vaginal prolapse repair without mesh (i.e., native-tissue repair).
As in several other trials evaluating transvaginal mesh for anterior vaginal prolapse, the investigators found that it led to improved anatomic outcomes, compared with native-tissue repair, but at the expense of a higher complication rate. In this study, women in the mesh group experienced a cumulative rate of vaginal mesh exposure of 16.9% by 12 months and had a higher rate of hematoma and greater voiding dysfunction in the immediate postoperative period.
The most clinically relevant outcome to patients who have prolapse is resolution of their symptom of vaginal bulging; this symptom improved similarly in both treatment groups.4
Strengths and weakness of the study
This study has a number of strengths:
- an excellent rate of follow-up (98%)
- a large number of study sites
- use of multiple validated outcome measures.
The principal weakness is the use of strict anatomic criteria as the primary outcome measure. In the general population, 40% of asymptomatic women who present for annual gynecologic examination have vaginal support of Stage 2 or higher and would have been considered failures by this definition.5 Prolapse beyond the hymen appears to be a more clinically relevant threshold for defining anatomic success.
FDA gets involved
In July 2011, the FDA issued a safety update on the use of transvaginal mesh for prolapse repair. Its principal findings:
- Serious adverse events are not rare
- Transvaginally placed mesh in prolapse repair does not conclusively improve clinical outcomes over traditional non-mesh repair.3
The FDA noted that patients who undergo prolapse repair with mesh are subject to a unique set of complications, including erosion and mesh contraction, which can be life-altering and, in some women, may require multiple surgeries to correct.
Given the safety concerns regarding transvaginal mesh, the FDA is considering changing the regulatory process for the introduction of new transvaginal mesh devices.
No single surgical approach for the correction of pelvic organ prolapse is superior for all women, be it traditional vaginal non-mesh repair, mesh-augmented repair, or the open or laparoscopic approach. There may be circumstances when transvaginal mesh is the best choice. However, given the potential risks, it seems clear that 1) its use should be judicious and 2) mesh-augmented repair should be performed only by surgeons who have appropriate training and only on patients who have been fully informed of the risks and benefits of all treatment options.—Matthew D. Barber, MD, MHS
We want to hear from you! Tell us what you think.
1. Jelovsek JE, Maher C, Barber MD. Pelvic organ prolapse. Lancet. 2007;369(9566):1027-1038.
2. Clark AL, Gregory T, Smith VJ, Edwards R. Epidemiologic evaluation of reoperation for surgically treated pelvic organ prolapse and urinary incontinence. Am J Obstet Gynecol. 2003;189(5):1261-1267.
3. FDA Safety Communication: UPDATE on Serious Complications Associated with Transvaginal Placement of Surgical Mesh for Pelvic Organ Prolapse. US Food and Drug Administration Web site. http://www.fda.gov/MedicalDevices/Safety/AlertsandNotices/ucm262435.htm. Published July 13 2011. Accessed September 19, 2011.
4. Barber MD, Brubaker L, Nygaard I, et al. Pelvic Floor Disorders Network. Defining success after surgery for pelvic organ prolapse. Obstet Gynecol. 2009;114(3):600-609.
5. Swift S, Woodman P, O’Boyle A, et al. Pelvic Organ Support Study (POSST): the distribution, clinical definition, and epidemiologic condition of pelvic organ support defects. Am J Obstet Gynecol. 2005;192(3):795-806.
- Are new tools for correcting prolapse and incontinence better just because they’re new?
Anne M. Weber, MD, MS (Web Exclusive Commentary, February 2009)
A woman’s lifetime risk of surgery for pelvic organ prolapse is approximately 7%; more than 300,000 prolapse surgeries are performed annually in the United States alone.1 Of the women who undergo surgery, an estimated 13% will require re-operation within 5 years, and as many as 29% will undergo another surgery for genital prolapse or a related condition at some point during their life.2
In the hope of improving the effectiveness and durability of vaginal prolapse repairs, many pelvic reconstructive surgeons have turned to reinforcement with synthetic mesh. However, because of concern about a higher risk of complications relative to other approaches, the use of transvaginal mesh for prolapse repair has come under increased scrutiny by the FDA. At this time, almost one quarter of all prolapse repairs involve placement of transvaginal mesh.3
Details of the trial
Withagen and colleagues enrolled 194 women from 13 Dutch medical centers. All women had recurrent pelvic organ prolapse and were randomized to either trocar-guided transvaginal mesh repair (Prolift) or conventional vaginal prolapse repair without mesh (i.e., native-tissue repair).
As in several other trials evaluating transvaginal mesh for anterior vaginal prolapse, the investigators found that it led to improved anatomic outcomes, compared with native-tissue repair, but at the expense of a higher complication rate. In this study, women in the mesh group experienced a cumulative rate of vaginal mesh exposure of 16.9% by 12 months and had a higher rate of hematoma and greater voiding dysfunction in the immediate postoperative period.
The most clinically relevant outcome to patients who have prolapse is resolution of their symptom of vaginal bulging; this symptom improved similarly in both treatment groups.4
Strengths and weakness of the study
This study has a number of strengths:
- an excellent rate of follow-up (98%)
- a large number of study sites
- use of multiple validated outcome measures.
The principal weakness is the use of strict anatomic criteria as the primary outcome measure. In the general population, 40% of asymptomatic women who present for annual gynecologic examination have vaginal support of Stage 2 or higher and would have been considered failures by this definition.5 Prolapse beyond the hymen appears to be a more clinically relevant threshold for defining anatomic success.
FDA gets involved
In July 2011, the FDA issued a safety update on the use of transvaginal mesh for prolapse repair. Its principal findings:
- Serious adverse events are not rare
- Transvaginally placed mesh in prolapse repair does not conclusively improve clinical outcomes over traditional non-mesh repair.3
The FDA noted that patients who undergo prolapse repair with mesh are subject to a unique set of complications, including erosion and mesh contraction, which can be life-altering and, in some women, may require multiple surgeries to correct.
Given the safety concerns regarding transvaginal mesh, the FDA is considering changing the regulatory process for the introduction of new transvaginal mesh devices.
No single surgical approach for the correction of pelvic organ prolapse is superior for all women, be it traditional vaginal non-mesh repair, mesh-augmented repair, or the open or laparoscopic approach. There may be circumstances when transvaginal mesh is the best choice. However, given the potential risks, it seems clear that 1) its use should be judicious and 2) mesh-augmented repair should be performed only by surgeons who have appropriate training and only on patients who have been fully informed of the risks and benefits of all treatment options.—Matthew D. Barber, MD, MHS
We want to hear from you! Tell us what you think.
- Are new tools for correcting prolapse and incontinence better just because they’re new?
Anne M. Weber, MD, MS (Web Exclusive Commentary, February 2009)
A woman’s lifetime risk of surgery for pelvic organ prolapse is approximately 7%; more than 300,000 prolapse surgeries are performed annually in the United States alone.1 Of the women who undergo surgery, an estimated 13% will require re-operation within 5 years, and as many as 29% will undergo another surgery for genital prolapse or a related condition at some point during their life.2
In the hope of improving the effectiveness and durability of vaginal prolapse repairs, many pelvic reconstructive surgeons have turned to reinforcement with synthetic mesh. However, because of concern about a higher risk of complications relative to other approaches, the use of transvaginal mesh for prolapse repair has come under increased scrutiny by the FDA. At this time, almost one quarter of all prolapse repairs involve placement of transvaginal mesh.3
Details of the trial
Withagen and colleagues enrolled 194 women from 13 Dutch medical centers. All women had recurrent pelvic organ prolapse and were randomized to either trocar-guided transvaginal mesh repair (Prolift) or conventional vaginal prolapse repair without mesh (i.e., native-tissue repair).
As in several other trials evaluating transvaginal mesh for anterior vaginal prolapse, the investigators found that it led to improved anatomic outcomes, compared with native-tissue repair, but at the expense of a higher complication rate. In this study, women in the mesh group experienced a cumulative rate of vaginal mesh exposure of 16.9% by 12 months and had a higher rate of hematoma and greater voiding dysfunction in the immediate postoperative period.
The most clinically relevant outcome to patients who have prolapse is resolution of their symptom of vaginal bulging; this symptom improved similarly in both treatment groups.4
Strengths and weakness of the study
This study has a number of strengths:
- an excellent rate of follow-up (98%)
- a large number of study sites
- use of multiple validated outcome measures.
The principal weakness is the use of strict anatomic criteria as the primary outcome measure. In the general population, 40% of asymptomatic women who present for annual gynecologic examination have vaginal support of Stage 2 or higher and would have been considered failures by this definition.5 Prolapse beyond the hymen appears to be a more clinically relevant threshold for defining anatomic success.
FDA gets involved
In July 2011, the FDA issued a safety update on the use of transvaginal mesh for prolapse repair. Its principal findings:
- Serious adverse events are not rare
- Transvaginally placed mesh in prolapse repair does not conclusively improve clinical outcomes over traditional non-mesh repair.3
The FDA noted that patients who undergo prolapse repair with mesh are subject to a unique set of complications, including erosion and mesh contraction, which can be life-altering and, in some women, may require multiple surgeries to correct.
Given the safety concerns regarding transvaginal mesh, the FDA is considering changing the regulatory process for the introduction of new transvaginal mesh devices.
No single surgical approach for the correction of pelvic organ prolapse is superior for all women, be it traditional vaginal non-mesh repair, mesh-augmented repair, or the open or laparoscopic approach. There may be circumstances when transvaginal mesh is the best choice. However, given the potential risks, it seems clear that 1) its use should be judicious and 2) mesh-augmented repair should be performed only by surgeons who have appropriate training and only on patients who have been fully informed of the risks and benefits of all treatment options.—Matthew D. Barber, MD, MHS
We want to hear from you! Tell us what you think.
1. Jelovsek JE, Maher C, Barber MD. Pelvic organ prolapse. Lancet. 2007;369(9566):1027-1038.
2. Clark AL, Gregory T, Smith VJ, Edwards R. Epidemiologic evaluation of reoperation for surgically treated pelvic organ prolapse and urinary incontinence. Am J Obstet Gynecol. 2003;189(5):1261-1267.
3. FDA Safety Communication: UPDATE on Serious Complications Associated with Transvaginal Placement of Surgical Mesh for Pelvic Organ Prolapse. US Food and Drug Administration Web site. http://www.fda.gov/MedicalDevices/Safety/AlertsandNotices/ucm262435.htm. Published July 13 2011. Accessed September 19, 2011.
4. Barber MD, Brubaker L, Nygaard I, et al. Pelvic Floor Disorders Network. Defining success after surgery for pelvic organ prolapse. Obstet Gynecol. 2009;114(3):600-609.
5. Swift S, Woodman P, O’Boyle A, et al. Pelvic Organ Support Study (POSST): the distribution, clinical definition, and epidemiologic condition of pelvic organ support defects. Am J Obstet Gynecol. 2005;192(3):795-806.
1. Jelovsek JE, Maher C, Barber MD. Pelvic organ prolapse. Lancet. 2007;369(9566):1027-1038.
2. Clark AL, Gregory T, Smith VJ, Edwards R. Epidemiologic evaluation of reoperation for surgically treated pelvic organ prolapse and urinary incontinence. Am J Obstet Gynecol. 2003;189(5):1261-1267.
3. FDA Safety Communication: UPDATE on Serious Complications Associated with Transvaginal Placement of Surgical Mesh for Pelvic Organ Prolapse. US Food and Drug Administration Web site. http://www.fda.gov/MedicalDevices/Safety/AlertsandNotices/ucm262435.htm. Published July 13 2011. Accessed September 19, 2011.
4. Barber MD, Brubaker L, Nygaard I, et al. Pelvic Floor Disorders Network. Defining success after surgery for pelvic organ prolapse. Obstet Gynecol. 2009;114(3):600-609.
5. Swift S, Woodman P, O’Boyle A, et al. Pelvic Organ Support Study (POSST): the distribution, clinical definition, and epidemiologic condition of pelvic organ support defects. Am J Obstet Gynecol. 2005;192(3):795-806.
Hospital-Focused Practice
As regular readers of The Hospitalist are aware, essentially every specialty in medicine is adopting the hospitalist model to some degree. After the “legacy” specialties of medicine and pediatrics, the model has more recently been embraced enthusiastically by neurologists, obstetricians, and general surgeons. But even fields like dermatology and ENT have put a hospitalist version of their specialties in place in at least a few places.
Did you know there is a Society for Dermatology Hospitalists? Did you know that the Neurohospitalist Society has its own journal? Did you know OB hospitalists have a really neat website, and the Society of OB/GYN Hospitalists is scheduled to have its first annual meeting in Boulder, Colo., Sept. 23-25?
It’ll make your head spin if you think about it too long. All of this raises a number of issues, including the need for more precise terminology to describe these fields and their practitioners.
The Need for Better Terminology
For example, now that we have neurohospitalists and psychiatric hospitalists, is it time to start attaching a modifier or prefix every time we use the word “hospitalist,” including when referring to “medical” hospitalists? I don’t think so. For the time being, I propose that when used alone, the word “hospitalist” still refers to a doctor who provides general medical care for adult inpatients. But I think any other use of the word does require a modifier, as in “peds hospitalist” or “GI hospitalist.”
(I think my view makes sense, but then, I’ve tried for years to ensure nocternist, with an E—NOCTernal intERNIST)—is the preferred spelling over nocternist, with a U. But Google returns nine hits for the former and 365,000 for the latter. Looks like I lost that one.)
Terminology for general and trauma surgeons is tricky. There is an emerging field of acute-care surgery, distinct from general surgery, which some argue passionately is nothing like a hospitalist model, and they tend to be offended if one uses the latter term. So, for now, we’ll need to use both “acute-care surgeon” and “surgical hospitalist” carefully. Although there are meaningful distinctions between acute-care surgery and a “standard” general surgery practice devoted to the hospital, there is an awful lot of overlap in the Venn diagrams of their expertise and what they do. But for now, it looks like we should expect both “acute-care surgeon” and “surgical hospitalist” to appear commonly, and the context will determine whether the terms could be used interchangeably.
While “obstetric hospitalist,” or “OB hospitalist,” is a perfectly useful term, I think it is great when laborist is substituted, at least in informal communication.
We still need a way to speak of all of these clinical roles (I don’t think we can properly call them specialties yet). I propose that we refer to all of them as specialties within the realm of “hospital-focused practice.” I’ve borrowed this term from the American Board of Internal Medicine’s Recognition of Focused Practice in Hospital Medicine, the new pathway to Maintenance of Certification.
And what about those doctors in each specialty who continue to practice in the traditional inpatient and outpatient model? Let’s call them “traditionalists.”
Hospital-Focused Practice
A rational vocabulary is only one of many significant issues raised by the growth of hospital-focused disciplines. In January, I participated in an SHM-convened, and AHA-supported, meeting of 11 practitioners who were hospitalists in neurology, obstetrics, general surgery, medicine, pediatrics, and ENT. (Sadly, the invited dermatology hospitalist couldn’t make it.) The meeting was filled with interest and sharing of lessons learned in each field. We discussed questions, and I have provided a very brief answer to each based on the conversation during the meeting and my own work with practices across many different specialties that have adopted the hospitalist model:
What are the reasons each specialty is turning to this model, and what is its prevalence? Hospitalists have appeared in a specialty largely to fill the void left by the traditionalists who no longer want to care for unattached patients admitted through the ED, or who want to leave the hospital altogether for a solely outpatient practice.
What are typical staffing models, night coverage arrangements, and provider career sustainability? These vary a lot by specialty, but laborists typically work 24-hour, in-house shifts. Surgical hospitalists usually work 12-hour shifts if they are in-house all the time, or 24-hour shifts if they take call from home. Neurohospitalists essentially always take call from home (did you even have to ask?).
Career longevity is still a matter of speculation, but the majority of those who have transitioned from traditional to hospitalist practice in their field are convinced they will have a longer career than if they hadn’t made the switch.
What are the effects of this practice model on clinical quality, patient outcomes, healthcare economics, and liability? It will be really difficult to get convincing research data on the quality effects of the hospitalist model in many fields. After more than 15 years in operation, research about the quality effects of the medical hospitalist model is not robust enough to satisfy some. But OB hospitalists may be the exception here. There is hope that their continuous, on-site presence will reduce complications from emergencies, and in doing so might reduce malpractice risk.
What is the prevalent financial model? The experience across a lot of healthcare settings to this point is that professional fee revenue alone usually is not enough to support a hospitalist practice model in any specialty. Just like medical and pediatric hospitalist models, the hospital in which the doctors practice usually provides additional financial support.
Hospitals usually are willing to do this because they are able to reallocate dollars spent paying for numerous specialty doctors to take ED call with poor performance, and instead use those dollars to support a hospitalist practice in that specialty that promises a better return on the investment.
Join us in November for a meeting to understand the implications of hospital-focused practice. Those of us at the January meeting of specialty hospitalists thought that it would be valuable to convene a much larger meeting to think about issues like those above and others. At the Nov. 4 meeting in Las Vegas, we plan to hear from such national figures as CMS’ chief medical officer, physicians practicing in a hospitalist model, and hospital and healthcare executives. The meeting will be structured to promote interaction and communication from attendees.
I hope to see you in Las Vegas. We have a lot to learn from one another.
Dr. Nelson has been a practicing hospitalist since 1988 and is cofounder and past president of SHM. He is a principal in Nelson Flores Hospital Medicine Consultants, a national hospitalist practice management consulting firm (www.nelsonflores.com). He is course codirector and faculty for SHM’s “Best Practices in Managing a Hospital Medicine Program” course. This column represents his views and is not intended to reflect an official position of SHM.
As regular readers of The Hospitalist are aware, essentially every specialty in medicine is adopting the hospitalist model to some degree. After the “legacy” specialties of medicine and pediatrics, the model has more recently been embraced enthusiastically by neurologists, obstetricians, and general surgeons. But even fields like dermatology and ENT have put a hospitalist version of their specialties in place in at least a few places.
Did you know there is a Society for Dermatology Hospitalists? Did you know that the Neurohospitalist Society has its own journal? Did you know OB hospitalists have a really neat website, and the Society of OB/GYN Hospitalists is scheduled to have its first annual meeting in Boulder, Colo., Sept. 23-25?
It’ll make your head spin if you think about it too long. All of this raises a number of issues, including the need for more precise terminology to describe these fields and their practitioners.
The Need for Better Terminology
For example, now that we have neurohospitalists and psychiatric hospitalists, is it time to start attaching a modifier or prefix every time we use the word “hospitalist,” including when referring to “medical” hospitalists? I don’t think so. For the time being, I propose that when used alone, the word “hospitalist” still refers to a doctor who provides general medical care for adult inpatients. But I think any other use of the word does require a modifier, as in “peds hospitalist” or “GI hospitalist.”
(I think my view makes sense, but then, I’ve tried for years to ensure nocternist, with an E—NOCTernal intERNIST)—is the preferred spelling over nocternist, with a U. But Google returns nine hits for the former and 365,000 for the latter. Looks like I lost that one.)
Terminology for general and trauma surgeons is tricky. There is an emerging field of acute-care surgery, distinct from general surgery, which some argue passionately is nothing like a hospitalist model, and they tend to be offended if one uses the latter term. So, for now, we’ll need to use both “acute-care surgeon” and “surgical hospitalist” carefully. Although there are meaningful distinctions between acute-care surgery and a “standard” general surgery practice devoted to the hospital, there is an awful lot of overlap in the Venn diagrams of their expertise and what they do. But for now, it looks like we should expect both “acute-care surgeon” and “surgical hospitalist” to appear commonly, and the context will determine whether the terms could be used interchangeably.
While “obstetric hospitalist,” or “OB hospitalist,” is a perfectly useful term, I think it is great when laborist is substituted, at least in informal communication.
We still need a way to speak of all of these clinical roles (I don’t think we can properly call them specialties yet). I propose that we refer to all of them as specialties within the realm of “hospital-focused practice.” I’ve borrowed this term from the American Board of Internal Medicine’s Recognition of Focused Practice in Hospital Medicine, the new pathway to Maintenance of Certification.
And what about those doctors in each specialty who continue to practice in the traditional inpatient and outpatient model? Let’s call them “traditionalists.”
Hospital-Focused Practice
A rational vocabulary is only one of many significant issues raised by the growth of hospital-focused disciplines. In January, I participated in an SHM-convened, and AHA-supported, meeting of 11 practitioners who were hospitalists in neurology, obstetrics, general surgery, medicine, pediatrics, and ENT. (Sadly, the invited dermatology hospitalist couldn’t make it.) The meeting was filled with interest and sharing of lessons learned in each field. We discussed questions, and I have provided a very brief answer to each based on the conversation during the meeting and my own work with practices across many different specialties that have adopted the hospitalist model:
What are the reasons each specialty is turning to this model, and what is its prevalence? Hospitalists have appeared in a specialty largely to fill the void left by the traditionalists who no longer want to care for unattached patients admitted through the ED, or who want to leave the hospital altogether for a solely outpatient practice.
What are typical staffing models, night coverage arrangements, and provider career sustainability? These vary a lot by specialty, but laborists typically work 24-hour, in-house shifts. Surgical hospitalists usually work 12-hour shifts if they are in-house all the time, or 24-hour shifts if they take call from home. Neurohospitalists essentially always take call from home (did you even have to ask?).
Career longevity is still a matter of speculation, but the majority of those who have transitioned from traditional to hospitalist practice in their field are convinced they will have a longer career than if they hadn’t made the switch.
What are the effects of this practice model on clinical quality, patient outcomes, healthcare economics, and liability? It will be really difficult to get convincing research data on the quality effects of the hospitalist model in many fields. After more than 15 years in operation, research about the quality effects of the medical hospitalist model is not robust enough to satisfy some. But OB hospitalists may be the exception here. There is hope that their continuous, on-site presence will reduce complications from emergencies, and in doing so might reduce malpractice risk.
What is the prevalent financial model? The experience across a lot of healthcare settings to this point is that professional fee revenue alone usually is not enough to support a hospitalist practice model in any specialty. Just like medical and pediatric hospitalist models, the hospital in which the doctors practice usually provides additional financial support.
Hospitals usually are willing to do this because they are able to reallocate dollars spent paying for numerous specialty doctors to take ED call with poor performance, and instead use those dollars to support a hospitalist practice in that specialty that promises a better return on the investment.
Join us in November for a meeting to understand the implications of hospital-focused practice. Those of us at the January meeting of specialty hospitalists thought that it would be valuable to convene a much larger meeting to think about issues like those above and others. At the Nov. 4 meeting in Las Vegas, we plan to hear from such national figures as CMS’ chief medical officer, physicians practicing in a hospitalist model, and hospital and healthcare executives. The meeting will be structured to promote interaction and communication from attendees.
I hope to see you in Las Vegas. We have a lot to learn from one another.
Dr. Nelson has been a practicing hospitalist since 1988 and is cofounder and past president of SHM. He is a principal in Nelson Flores Hospital Medicine Consultants, a national hospitalist practice management consulting firm (www.nelsonflores.com). He is course codirector and faculty for SHM’s “Best Practices in Managing a Hospital Medicine Program” course. This column represents his views and is not intended to reflect an official position of SHM.
As regular readers of The Hospitalist are aware, essentially every specialty in medicine is adopting the hospitalist model to some degree. After the “legacy” specialties of medicine and pediatrics, the model has more recently been embraced enthusiastically by neurologists, obstetricians, and general surgeons. But even fields like dermatology and ENT have put a hospitalist version of their specialties in place in at least a few places.
Did you know there is a Society for Dermatology Hospitalists? Did you know that the Neurohospitalist Society has its own journal? Did you know OB hospitalists have a really neat website, and the Society of OB/GYN Hospitalists is scheduled to have its first annual meeting in Boulder, Colo., Sept. 23-25?
It’ll make your head spin if you think about it too long. All of this raises a number of issues, including the need for more precise terminology to describe these fields and their practitioners.
The Need for Better Terminology
For example, now that we have neurohospitalists and psychiatric hospitalists, is it time to start attaching a modifier or prefix every time we use the word “hospitalist,” including when referring to “medical” hospitalists? I don’t think so. For the time being, I propose that when used alone, the word “hospitalist” still refers to a doctor who provides general medical care for adult inpatients. But I think any other use of the word does require a modifier, as in “peds hospitalist” or “GI hospitalist.”
(I think my view makes sense, but then, I’ve tried for years to ensure nocternist, with an E—NOCTernal intERNIST)—is the preferred spelling over nocternist, with a U. But Google returns nine hits for the former and 365,000 for the latter. Looks like I lost that one.)
Terminology for general and trauma surgeons is tricky. There is an emerging field of acute-care surgery, distinct from general surgery, which some argue passionately is nothing like a hospitalist model, and they tend to be offended if one uses the latter term. So, for now, we’ll need to use both “acute-care surgeon” and “surgical hospitalist” carefully. Although there are meaningful distinctions between acute-care surgery and a “standard” general surgery practice devoted to the hospital, there is an awful lot of overlap in the Venn diagrams of their expertise and what they do. But for now, it looks like we should expect both “acute-care surgeon” and “surgical hospitalist” to appear commonly, and the context will determine whether the terms could be used interchangeably.
While “obstetric hospitalist,” or “OB hospitalist,” is a perfectly useful term, I think it is great when laborist is substituted, at least in informal communication.
We still need a way to speak of all of these clinical roles (I don’t think we can properly call them specialties yet). I propose that we refer to all of them as specialties within the realm of “hospital-focused practice.” I’ve borrowed this term from the American Board of Internal Medicine’s Recognition of Focused Practice in Hospital Medicine, the new pathway to Maintenance of Certification.
And what about those doctors in each specialty who continue to practice in the traditional inpatient and outpatient model? Let’s call them “traditionalists.”
Hospital-Focused Practice
A rational vocabulary is only one of many significant issues raised by the growth of hospital-focused disciplines. In January, I participated in an SHM-convened, and AHA-supported, meeting of 11 practitioners who were hospitalists in neurology, obstetrics, general surgery, medicine, pediatrics, and ENT. (Sadly, the invited dermatology hospitalist couldn’t make it.) The meeting was filled with interest and sharing of lessons learned in each field. We discussed questions, and I have provided a very brief answer to each based on the conversation during the meeting and my own work with practices across many different specialties that have adopted the hospitalist model:
What are the reasons each specialty is turning to this model, and what is its prevalence? Hospitalists have appeared in a specialty largely to fill the void left by the traditionalists who no longer want to care for unattached patients admitted through the ED, or who want to leave the hospital altogether for a solely outpatient practice.
What are typical staffing models, night coverage arrangements, and provider career sustainability? These vary a lot by specialty, but laborists typically work 24-hour, in-house shifts. Surgical hospitalists usually work 12-hour shifts if they are in-house all the time, or 24-hour shifts if they take call from home. Neurohospitalists essentially always take call from home (did you even have to ask?).
Career longevity is still a matter of speculation, but the majority of those who have transitioned from traditional to hospitalist practice in their field are convinced they will have a longer career than if they hadn’t made the switch.
What are the effects of this practice model on clinical quality, patient outcomes, healthcare economics, and liability? It will be really difficult to get convincing research data on the quality effects of the hospitalist model in many fields. After more than 15 years in operation, research about the quality effects of the medical hospitalist model is not robust enough to satisfy some. But OB hospitalists may be the exception here. There is hope that their continuous, on-site presence will reduce complications from emergencies, and in doing so might reduce malpractice risk.
What is the prevalent financial model? The experience across a lot of healthcare settings to this point is that professional fee revenue alone usually is not enough to support a hospitalist practice model in any specialty. Just like medical and pediatric hospitalist models, the hospital in which the doctors practice usually provides additional financial support.
Hospitals usually are willing to do this because they are able to reallocate dollars spent paying for numerous specialty doctors to take ED call with poor performance, and instead use those dollars to support a hospitalist practice in that specialty that promises a better return on the investment.
Join us in November for a meeting to understand the implications of hospital-focused practice. Those of us at the January meeting of specialty hospitalists thought that it would be valuable to convene a much larger meeting to think about issues like those above and others. At the Nov. 4 meeting in Las Vegas, we plan to hear from such national figures as CMS’ chief medical officer, physicians practicing in a hospitalist model, and hospital and healthcare executives. The meeting will be structured to promote interaction and communication from attendees.
I hope to see you in Las Vegas. We have a lot to learn from one another.
Dr. Nelson has been a practicing hospitalist since 1988 and is cofounder and past president of SHM. He is a principal in Nelson Flores Hospital Medicine Consultants, a national hospitalist practice management consulting firm (www.nelsonflores.com). He is course codirector and faculty for SHM’s “Best Practices in Managing a Hospital Medicine Program” course. This column represents his views and is not intended to reflect an official position of SHM.
Vulvar pain syndromes: Making the correct diagnosis
Although the incidence of vulvar pain has increased over the past decade—thanks to both greater awareness and increasing numbers of affected women—the phenomenon is not a recent development. As early as 1874, T. Galliard Thomas wrote, “[T]his disorder, although fortunately not very frequent, is by no means very rare.”1 He went on to express “surprise” that it had not been “more generally and fully described.”
Despite the focus Thomas directed to the issue, vulvar pain did not get much attention until the 21st century, when a number of studies began to gauge its prevalence. For example, in a study in Boston of about 5,000 women, the lifetime prevalence of chronic vulvar pain was 16%.2 And in a study in Texas, the prevalence of vulvar pain in an urban, largely minority population was estimated to be 11%.3 The Boston study also reported that “nearly 40% of women chose not to seek treatment, and, of those who did, 60% saw three or more doctors, many of whom could not provide a diagnosis.”2
Clearly, there is a need for comprehensive information on vulvar pain and its causes, symptoms, diagnosis, and treatment. To address the lack of guidance, OBG Management Contributing Editor Neal M. Lonky, MD, assembled a panel of experts on vulvar pain syndromes and invited them to share their considerable knowledge. The ensuing discussion, presented in three parts, offers a gold mine of information.
In this opening article, the panel focuses on causes, symptomatology, and diagnosis of this common complaint. In Part 2, which will appear in the October issue of this journal, the focus is the bounty of treatment options. Part 3 follows in November, when the discussion shifts to vestibulodynia.
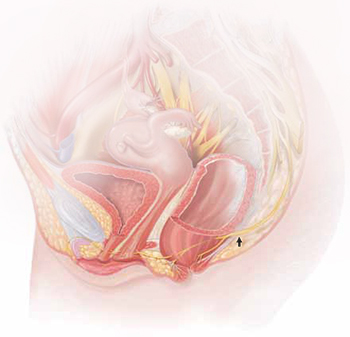
The lower vagina and vulva are richly supplied with peripheral nerves and are, therefore, sensitive to pain, particularly the region of the hymeneal ring. Although the pudendal nerve (arrow) courses through the area, it is an uncommon source of vulvar pain.
Common diagnoses—and misdiagnoses
Dr. Lonky: What are the most common diagnoses when vulvar pain is the complaint?
Dr. Gunter: The most common cause of chronic vulvar pain is vulvodynia, although lichen simplex chronicus, chronic yeast infections, and non-neoplastic epithelial disorders, such as lichen sclerosus and lichen planus, can also produce irritation and pain. In postmenopausal women, atrophic vaginitis can also cause a burning pain, although symptoms are typically more vaginal than vulvar. Yeast and lichen simplex chronicus typically produce itching, although sometimes they can present with irritation and pain, so they must be considered in the differential diagnosis. It is important to remember that many women with vulvodynia have used multiple topical agents and may have developed complex hygiene rituals in an attempt to treat their symptoms, which can result in a secondary lichen simplex chronicus.
That said, there is a high frequency of misdiagnosis with yeast. For example, in a study by Nyirjesy and colleagues, two thirds of women who were referred to a tertiary clinic for chronic vulvovaginal candidiasis were found to have a noninfectious entity instead—most commonly lichen simplex chronicus and vulvodynia.4
Dr. Edwards: The most common “diagnosis” for vulvar pain is vulvodynia. However, the definition of vulvodynia is pain—i.e., burning, rawness, irritation, soreness, aching, or stabbing or stinging sensations—in the absence of skin disease, infection, or specific neurologic disease. Therefore, even though the usual cause of vulvar pain is vulvodynia, it is a diagnosis of exclusion, and skin disease, infection, and neurologic disease must be ruled out.
In regard to infection, Candida albicans and bacterial vaginosis (BV) are usually the first conditions that are considered when a patient complains of vulvar pain, but they are not common causes of vulvar pain and are never causes of chronic vulvar pain. Very rarely they may cause recurrent pain that clears, at least briefly, with treatment.
Candida albicans is usually primarily pruritic, and BV produces discharge and odor, sometimes with minor symptoms. Non-albicans Candida (e.g., Candida glabrata) is nearly always asymptomatic, but it occasionally causes irritation and burning.
Group B streptococcus is another infectious entity that very, very occasionally causes irritation and dyspareunia but is usually only a colonizer.
Herpes simplex virus is a cause of recurrent but not chronic pain.
Chronic pain is more likely to be caused by skin disease than by infection. Lichen simplex chronicus causes itching; any pain is due to erosions from scratching.
Dr. Haefner: Several other infectious conditions or their treatments can cause vulvar pain. For example, herpes (particularly primary herpes infection) is classically associated with vulvar pain. The pain is so great that, at times, the patient requires admission for pain control. Surprisingly, despite the known pain of herpes, approximately 80% of patients who have it are unaware of their diagnosis.
Although condyloma is generally a painless condition, many patients complain of pain following treatment for it, whether treatment involves topical medications or laser surgery.
Chancroid is a painful vulvar ulcer. Trichomonas can sometimes be associated with vulvar pain.
Dr. Lonky: What terminology do we use when we discuss vulvar pain?
Dr. Haefner: The current terminology used to describe vulvar pain was published in 2004, after years of debate over nomenclature within the International Society for the Study of Vulvovaginal Disease.5 The terminology lists two major categories of vulvar pain:
- pain related to a specific disorder. This category encompasses numerous conditions that feature an abnormal appearance of the vulva (Table 1).
TABLE 1
Terminology and classification of vulvar pain from the International Society for the Study of Vulvovaginal Disease
|
| SOURCE: Moyal-Barracco and Lynch.5 Reproduced with permission from the Journal of Reproductive Medicine. |
- vulvodynia, in which the vulva appears normal, other than occasional erythema, which is most prominent at the duct openings (vestibular ducts—Bartholin’s and Skene’s).
As for vulvar pain, there are two major forms:
- hyperalgesia (a low threshold for pain)
- allodynia (pain in response to light touch).
Some diseases that are associated with vulvar pain do not qualify for the diagnosis of vulvodynia (Table 2) because they are associated with an abnormal appearance of the vulva.
TABLE 2
Conditions other than vulvodynia that are associated with vulvar pain
| Acute irritant contact dermatitis (e.g., erosion due to podofilox, imiquimod, cantharidin, fluorouracil, or podophyllin toxin) |
| Aphthous ulcer |
| Atrophy |
| Bartholin’s abscess |
| Candidiasis |
| Carcinoma |
| Chronic irritant contact dermatitis |
| Endometriosis |
| Herpes (simplex and zoster) |
| Immunobullous diseases (including cicatricial pemphigoid, pemphigus vulgaris, linear immunoglobulin A disease, etc.) |
| Lichen planus |
| Lichen sclerosus |
| Podophyllin overdose (see above) |
| Prolapsed urethra |
| Sjögren’s syndrome |
| Trauma |
| Trichomoniasis |
| Vulvar intraepithelial neoplasia |
What needs to be ruled out for a diagnosis of vulvodynia?
Dr. Lonky: What skin diseases need to be ruled out before vulvodynia can be diagnosed?
Dr. Edwards: Skin diseases that affect the vulva are usually pruritic—pain is a later sign. Lichen simplex chronicus (also known as eczema) is pruritus caused by any irritant; any pain that arises is produced by visible excoriations from scratching.
Lichen sclerosus manifests as white epithelium that has a crinkling, shiny, or waxy texture. It can produce pain, especially dyspareunia. The pain is caused by erosions that arise from fragility and introital narrowing and inelasticity.
Vulvovaginal lichen planus is usually erosive and preferentially affects mucous membranes, especially the vestibule; it sometimes affects the vagina and mouth, as well.
Desquamative inflammatory vaginitis is most likely a skin disease that affects only the vagina. It involves introital redness and a clinically and microscopically purulent vaginal discharge that also reveals parabasal cells and absent lactobacilli.
Dr. Lonky: You mentioned that neurologic diseases can sometimes cause vulvar pain. Which ones?
Dr. Edwards: Pudendal neuralgia, diabetic neuropathy, and post-herpetic neuralgia are the most common specific neurologic causes of vulvar pain. Multiple sclerosis can also produce pain syndromes. Post-herpetic neuralgia follows herpes zoster—not herpes simplex—virus infection.
Dr. Lonky: Any other conditions that can cause vulvar pain?
Dr. Haefner: Aphthous ulcers are common and are often flared by stress.
Non-neoplastic epithelial disorders are also seen frequently in health-care providers’ offices; many patients who experience them report pain on the vulva.
It is always important to consider cancer when a patient has an abnormal vulvar appearance and pain that has persisted despite treatment.
What are the most common vulvar pain syndromes?
Dr. Lonky: If you were to rank vulvar pain syndromes according to their prevalence, what would the most common syndromes be?
Dr. Gunter: Given the misdiagnosis of many women, who are told they have chronic yeast infection, as I mentioned, it’s hard to know which vulvar pain syndromes are most prevalent. I suspect that lichen simplex chronicus is most common, followed by vulvodynia, with chronic yeast infection a distant third.
My experience reflects what Nyirjesy and colleagues4 found: 65% to 75% of women referred to my clinic with chronic yeast actually have lichen simplex chronicus or vulvodynia. In postmenopausal women, atrophic vaginitis is also a consideration; it’s becoming more common now that the use of systemic hormone replacement therapy is decreasing.
Dr. Lonky: What about subsets of vulvodynia? Which ones are most common?
Dr. Edwards: There is good evidence of marked overlap among subsets of vulvodynia. The vast majority of women who have vulvodynia experience primarily provoked vestibular pain, regardless of age. However, I find that almost all patients also report pain that extends beyond the vestibule at times, as well as occasional unprovoked pain.
The diagnosis requires the exclusion of other causes of vulvar pain, and the subset is identified by the location of pain (that is, is it strictly localized or generalized or even migratory?) and its provoked or unprovoked nature.
Localized clitoral pain and vulvar pain localized to one side of the vulva are extremely uncommon, but they do occur. And although I rarely encounter teenagers and prepubertal children who have vulvodynia, I do have patients in both age groups who have vulvodynia.
Dr. Lonky: Are there racial differences in the prevalence of vulvodynia?
Dr. Edwards: Although several good studies show that women of African descent and white patients are equally likely to experience vulvodynia, the vast majority (99%) of my patients who have vulvodynia are white. My patients of African descent consult me primarily for itching or discharge.
My local demographics prevent me from judging the likelihood of Asians having vulvodynia, and our Hispanic population has limited access to health care.
In general, I don’t think that demographics are useful in making the diagnosis of vulvodynia.
Do women who have vulvar pain tend to have comorbidities?
Dr. Lonky: Do your patients who have vulvodynia or another vulvar pain syndrome tend to have comorbidities? If so, is this information helpful in establishing the diagnosis and planning therapy?
Dr. Haefner: Women who have vulvodynia often have other medical problems as well. In my practice, when new patients who have vulvodynia complete their intake survey, they often report a history of headache, irritable bowel syndrome, interstitial cystitis, fibromyalgia,6 chronic fatigue syndrome, back pain, and temporomandibular joint (TMJ) disorder. These comorbidities are not particularly helpful in establishing the diagnosis of vulvodynia, but they are an important consideration when choosing therapy for the patient. Often, the medications chosen to treat one condition will also benefit another condition. However, it’s important to check for potential interactions between drugs before prescribing a new treatment.
Dr. Gunter: A significant number of women who have vulvodynia also have other chronic pain syndromes. For example, the incidence of bladder pain syndrome–interstitial cystitis is 68% to 82% among women who have vulvodynia, compared with a baseline rate among all women of 6% to 11%.7-10 The rate of irritable bowel syndrome is more than doubled among women who have vulvodynia, compared with the general population (27% versus 12%).8 Another common comorbidity, hypertonic somatic dysfunction of the pelvic floor, is identified in 10% to 90% of women who have chronic vulvar pain.8,11,12 These women also have a higher incidence of nongenital pain syndromes, such as fibromyalgia, migraine, and TMJ dysfunction, than the general population, as Dr. Haefner noted.8,12,13
Many studies have evaluated psychological and emotional contributions to chronic vulvar pain. Pain and depression are intimately related—the incidence of depression among all people who experience chronic pain ranges from 27% to 54%, compared with 5% to 17% among the general population.14-16 The relationship is complex because chronic illness in general is associated with depression. Nevertheless, several studies have noted an increase in anxiety, stress, and depression among women who have vulvodynia.17-19
I screen every patient for depression using a Patient Health Questionnaire (PHQ-9); I also screen for anxiety. I find that a significant percentage of patients in my clinic are depressed or have an anxiety disorder. Failure to address these comorbidities makes treatment very difficult. I typically prescribe citalopram (Celexa), although there is some question whether it can safely be combined with a tricyclic antidepressant. We also offer stress-reduction classes, teach every patient the value of diaphragmatic breathing, offer mind-body classes for anxiety and stress, and provide intensive programs where the patient can learn important self-care skills, such as pacing (spacing activities throughout the day in a manner that avoids aggravating the pain), and address her anxiety and stress in a more guided manner. We also have a psychologist who specializes in pain for any patient who may need one-on-one counseling.
Dr. Edwards: The presence of comorbidities is somewhat useful in making the diagnosis of vulvodynia. I question my diagnosis, in fact, when a patient who has vulvodynia does not have headaches, low energy, depression, anxiety, irritable bowel syndrome, constipation, fibromyalgia, chronic fatigue, sensitivity to medications, TMJ dysfunction, or urinary symptoms.
How common is pudendal neuralgia?
Dr. Lonky: How prevalent is a finding of pudendal neuralgia?
Dr. Edwards: The prevalence and incidence of pudendal neuralgia are not known. Those who specialize in this condition think it is relatively common. I do not identify or suspect it very often. Its definitive diagnosis and management are outside the purview of the general gynecologist, but the general gynecologist should recognize the symptoms of pudendal neuralgia and refer the patient for evaluation and therapy.
Dr. Lonky: What are those symptoms?
Dr. Haefner: Pudendal neuralgia often occurs following trauma to the pudendal nerve. The pudendal nerve arises from sacral nerves, generally sacral nerves 2 to 4. Several tests can be utilized to diagnose this condition, including quantitative sensor tests, pudendal nerve motor latency tests, electromyography (EMG), and pudendal nerve blocks.20
Nantes Criteria allow for making a diagnosis of pudendal neuralgia (Table 3).21
TABLE 3
Nantes Criteria for pudendal neuralgia by pudendal nerve entrapment
Essential criteria
|
Complementary diagnostic criteria
|
Exclusion criteria
|
Associated signs not excluding the diagnosis
|
| SOURCE: Labat et al.21 Reproduced with permission from Neurology and Urodynamics. |
Initial treatments for pudendal neuralgia should be conservative. Treatments consist of lifestyle changes to prevent flare of disease. Physical therapy, medical management, nerve blocks, and alternative treatments may be beneficial.
Pudendal nerve entrapment is often exacerbated by sitting (not on a toilet seat, however) and is reduced in a standing position. It tends to increase in intensity throughout the day.22 The final treatment for pudendal nerve entrapment is surgery if the nerve is compressed. By this time, the generalist is not generally the provider who performs the surgery.
Dr. Gunter: I believe pudendal neuralgia is sometimes overdiagnosed. EMG studies of the pudendal nerve, often touted as a diagnostic tool, are unreliable (they can be abnormal after vaginal delivery or vaginal hysterectomy, for example). In my experience, bilateral pain is less likely to be pudendal neuralgia; spontaneous bilateral compression neuropathy at exactly the same level is not a common phenomenon in chronic pain.
I reserve the diagnosis of pudendal neuralgia for women who have allodynia in the distribution of the pudendal nerve with severe pain on sitting, and who have exquisite tenderness when pressure is applied over the pudendal nerve (at the level of the ischial spine on vaginal examination). Typically, the vaginal sidewall on the affected side is very sensitive to light touch. I do see pudendal nerve pain after vaginal surgery when there has been some compromise of the pudendal nerve or the sacral plexus. This is typically unilateral pain.
Dr. Lonky: Thank you all. We’ll continue our discussion, with a focus on treatment, in the October 2011 issue.
- Part 2: A bounty of treatment options
(October 2011) - Part 3: Vestibulodynia
(November 2011)
We want to hear from you! Tell us what you think.
1. Thomas TG. Practical Treatise on the Diseases of Women. Philadelphia Pa: Henry C. Lea; 1874.
2. Harlow BL, Stewart EG. A population-based assessment of chronic unexplained vulvar pain: have we underestimated the prevalence of vulvodynia? J Am Med Womens Assoc. 2003;58(2):82-88.
3. Lavy RJ, Hynan LS, Haley RW. Prevalence of vulvar pain in an urban minority population. J Reprod Med. 2007;52(1):59-62.
4. Nyirjesy P, Peyton C, Weitz MV, Mathew L, Culhane JF. Causes of chronic vaginitis: analysis of a prospective database of affected women. Obstet Gynecol. 2006;108(5):1185-1191.
5. Moyal-Barracco M, Lynch PJ. 2003 ISSVD terminology and classification of vulvodynia: a historical perspective. J Reprod Med. 2004;49(10):772-777.
6. Yunas MB. Fibromyalgia and overlapping disorders: the unifying concept of central sensitivity syndromes. Semin Arthritis Rheum. 2007;36(6):339-356.
7. Kahn BS, Tatro C, Parsons CL, Willems JJ. Prevalence of interstitial cystitis in vulvodynia patients detected by bladder potassium sensitivity. J Sex Med. 2010;7(2 Pt 2):996-1002.
8. Arnold JD, Bachman GS, Rosen R, Kelly S, Rhoads GG. Vulvodynia: characteristics and associations with comorbidities and quality of life. Obstet Gynecol. 2006;107(3):617-624.
9. Parsons CL, Dell J, Stanford EJ, et al. The prevalence of interstitial cystitis in gynecologic patients with pelvic pain, as detected by intravesical potassium sensitivity. Am J Obstet Gynecol. 2002;187(5):1395-1400.
10. Clemens JQ, Meenan RT, O’Keefe Rosetti MC, et al. Prevalence of interstitial cystitis symptoms in a managed care population. J Urol. 2005;174(2):576-580.
11. Engman M, Lindehammar H, Wijma B. Surface electromyography diagnostics in women with partial vaginismus with or without vulvar vestibulitis and in asymptomatic women. J Psychosom Obstet Gynecol. 2004;25(3-4):281-294.
12. Gunter J. Vulvodynia: new thoughts on a devastating condition. Obstet Gynecol Surv. 2007;62(12):812-819.
13. Gordon AS, Panahlan-Jand M, McComb F, Melegari C, Sharp S. Characteristics of women with vulvar pain disorders: a Web-based survey. J Sex Marital Ther. 2003;29(suppl 1):45.-
14. Whitten CE, Cristobal K. Chronic pain is a chronic condition not just a symptom. Permanente J. 2005;9(3):43.-
15. Manchikanti L, Fellows B, Pampati V, et al. Comparison of psychological status of chronic pain patients and the general population. Pain Physician. 2002;5(1):40-48.
16. Banks SM, Kerns RD. Explaining the high rates of depression in chronic pain: a diathesis-stress framework. Psychological Bulletin. 1996;119(1):95-110.
17. Sadownik LA. Clinical correlates of vulvodynia patients. A prospective study of 300 patients. J Reprod Med. 2000;5:40-48.Editor found in PubMed: Sadownik LA. Clinical profile of vulvodynia patients. A prospective study of 300 patients. J Reprod Med. 2000;45(8):679–684. Could not find the citation listed. Please confirm.
18. Reed BD, Haefner HK, Punch MR, Roth RS, Gorenflo DW, Gillespie BW. Psychosocial and sexual functioning in women with vulvodynia and chronic pelvic pain. A comparative evaluation. J Reprod Med. 2000;45(8):624-632.
19. Landry T, Bergeron S. Biopsychosocial factors associated with dyspareunia in a community sample of adolescent girls. Arch Sex Behav. 2011;June 22.
20. Goldstein A, Pukall C, Goldstein I. When Sex Hurts: A Woman’s Guide to Banishing Sexual Pain. Cambridge Mass: Da Capo Lifelong Books; 2011;117-126.
21. Labat JJ, Riant T, Robert R, Amarenco G, Lefaucheur JP, Rigaud J. Diagnostic criteria for pudendal neuralgia by pudendal nerve entrapment (Nantes criteria). Neurol Urodyn. 2008;27(4):306-310.
22. Popeney C, Answell V, Renney K. Pudendal entrapment as an etiology of chronic perineal pain: Diagnosis and treatment. Neurol Urodyn. 2007;26(6):820-827.
Although the incidence of vulvar pain has increased over the past decade—thanks to both greater awareness and increasing numbers of affected women—the phenomenon is not a recent development. As early as 1874, T. Galliard Thomas wrote, “[T]his disorder, although fortunately not very frequent, is by no means very rare.”1 He went on to express “surprise” that it had not been “more generally and fully described.”
Despite the focus Thomas directed to the issue, vulvar pain did not get much attention until the 21st century, when a number of studies began to gauge its prevalence. For example, in a study in Boston of about 5,000 women, the lifetime prevalence of chronic vulvar pain was 16%.2 And in a study in Texas, the prevalence of vulvar pain in an urban, largely minority population was estimated to be 11%.3 The Boston study also reported that “nearly 40% of women chose not to seek treatment, and, of those who did, 60% saw three or more doctors, many of whom could not provide a diagnosis.”2
Clearly, there is a need for comprehensive information on vulvar pain and its causes, symptoms, diagnosis, and treatment. To address the lack of guidance, OBG Management Contributing Editor Neal M. Lonky, MD, assembled a panel of experts on vulvar pain syndromes and invited them to share their considerable knowledge. The ensuing discussion, presented in three parts, offers a gold mine of information.
In this opening article, the panel focuses on causes, symptomatology, and diagnosis of this common complaint. In Part 2, which will appear in the October issue of this journal, the focus is the bounty of treatment options. Part 3 follows in November, when the discussion shifts to vestibulodynia.

The lower vagina and vulva are richly supplied with peripheral nerves and are, therefore, sensitive to pain, particularly the region of the hymeneal ring. Although the pudendal nerve (arrow) courses through the area, it is an uncommon source of vulvar pain.
Common diagnoses—and misdiagnoses
Dr. Lonky: What are the most common diagnoses when vulvar pain is the complaint?
Dr. Gunter: The most common cause of chronic vulvar pain is vulvodynia, although lichen simplex chronicus, chronic yeast infections, and non-neoplastic epithelial disorders, such as lichen sclerosus and lichen planus, can also produce irritation and pain. In postmenopausal women, atrophic vaginitis can also cause a burning pain, although symptoms are typically more vaginal than vulvar. Yeast and lichen simplex chronicus typically produce itching, although sometimes they can present with irritation and pain, so they must be considered in the differential diagnosis. It is important to remember that many women with vulvodynia have used multiple topical agents and may have developed complex hygiene rituals in an attempt to treat their symptoms, which can result in a secondary lichen simplex chronicus.
That said, there is a high frequency of misdiagnosis with yeast. For example, in a study by Nyirjesy and colleagues, two thirds of women who were referred to a tertiary clinic for chronic vulvovaginal candidiasis were found to have a noninfectious entity instead—most commonly lichen simplex chronicus and vulvodynia.4
Dr. Edwards: The most common “diagnosis” for vulvar pain is vulvodynia. However, the definition of vulvodynia is pain—i.e., burning, rawness, irritation, soreness, aching, or stabbing or stinging sensations—in the absence of skin disease, infection, or specific neurologic disease. Therefore, even though the usual cause of vulvar pain is vulvodynia, it is a diagnosis of exclusion, and skin disease, infection, and neurologic disease must be ruled out.
In regard to infection, Candida albicans and bacterial vaginosis (BV) are usually the first conditions that are considered when a patient complains of vulvar pain, but they are not common causes of vulvar pain and are never causes of chronic vulvar pain. Very rarely they may cause recurrent pain that clears, at least briefly, with treatment.
Candida albicans is usually primarily pruritic, and BV produces discharge and odor, sometimes with minor symptoms. Non-albicans Candida (e.g., Candida glabrata) is nearly always asymptomatic, but it occasionally causes irritation and burning.
Group B streptococcus is another infectious entity that very, very occasionally causes irritation and dyspareunia but is usually only a colonizer.
Herpes simplex virus is a cause of recurrent but not chronic pain.
Chronic pain is more likely to be caused by skin disease than by infection. Lichen simplex chronicus causes itching; any pain is due to erosions from scratching.
Dr. Haefner: Several other infectious conditions or their treatments can cause vulvar pain. For example, herpes (particularly primary herpes infection) is classically associated with vulvar pain. The pain is so great that, at times, the patient requires admission for pain control. Surprisingly, despite the known pain of herpes, approximately 80% of patients who have it are unaware of their diagnosis.
Although condyloma is generally a painless condition, many patients complain of pain following treatment for it, whether treatment involves topical medications or laser surgery.
Chancroid is a painful vulvar ulcer. Trichomonas can sometimes be associated with vulvar pain.
Dr. Lonky: What terminology do we use when we discuss vulvar pain?
Dr. Haefner: The current terminology used to describe vulvar pain was published in 2004, after years of debate over nomenclature within the International Society for the Study of Vulvovaginal Disease.5 The terminology lists two major categories of vulvar pain:
- pain related to a specific disorder. This category encompasses numerous conditions that feature an abnormal appearance of the vulva (Table 1).
TABLE 1
Terminology and classification of vulvar pain from the International Society for the Study of Vulvovaginal Disease
|
| SOURCE: Moyal-Barracco and Lynch.5 Reproduced with permission from the Journal of Reproductive Medicine. |
- vulvodynia, in which the vulva appears normal, other than occasional erythema, which is most prominent at the duct openings (vestibular ducts—Bartholin’s and Skene’s).
As for vulvar pain, there are two major forms:
- hyperalgesia (a low threshold for pain)
- allodynia (pain in response to light touch).
Some diseases that are associated with vulvar pain do not qualify for the diagnosis of vulvodynia (Table 2) because they are associated with an abnormal appearance of the vulva.
TABLE 2
Conditions other than vulvodynia that are associated with vulvar pain
| Acute irritant contact dermatitis (e.g., erosion due to podofilox, imiquimod, cantharidin, fluorouracil, or podophyllin toxin) |
| Aphthous ulcer |
| Atrophy |
| Bartholin’s abscess |
| Candidiasis |
| Carcinoma |
| Chronic irritant contact dermatitis |
| Endometriosis |
| Herpes (simplex and zoster) |
| Immunobullous diseases (including cicatricial pemphigoid, pemphigus vulgaris, linear immunoglobulin A disease, etc.) |
| Lichen planus |
| Lichen sclerosus |
| Podophyllin overdose (see above) |
| Prolapsed urethra |
| Sjögren’s syndrome |
| Trauma |
| Trichomoniasis |
| Vulvar intraepithelial neoplasia |
What needs to be ruled out for a diagnosis of vulvodynia?
Dr. Lonky: What skin diseases need to be ruled out before vulvodynia can be diagnosed?
Dr. Edwards: Skin diseases that affect the vulva are usually pruritic—pain is a later sign. Lichen simplex chronicus (also known as eczema) is pruritus caused by any irritant; any pain that arises is produced by visible excoriations from scratching.
Lichen sclerosus manifests as white epithelium that has a crinkling, shiny, or waxy texture. It can produce pain, especially dyspareunia. The pain is caused by erosions that arise from fragility and introital narrowing and inelasticity.
Vulvovaginal lichen planus is usually erosive and preferentially affects mucous membranes, especially the vestibule; it sometimes affects the vagina and mouth, as well.
Desquamative inflammatory vaginitis is most likely a skin disease that affects only the vagina. It involves introital redness and a clinically and microscopically purulent vaginal discharge that also reveals parabasal cells and absent lactobacilli.
Dr. Lonky: You mentioned that neurologic diseases can sometimes cause vulvar pain. Which ones?
Dr. Edwards: Pudendal neuralgia, diabetic neuropathy, and post-herpetic neuralgia are the most common specific neurologic causes of vulvar pain. Multiple sclerosis can also produce pain syndromes. Post-herpetic neuralgia follows herpes zoster—not herpes simplex—virus infection.
Dr. Lonky: Any other conditions that can cause vulvar pain?
Dr. Haefner: Aphthous ulcers are common and are often flared by stress.
Non-neoplastic epithelial disorders are also seen frequently in health-care providers’ offices; many patients who experience them report pain on the vulva.
It is always important to consider cancer when a patient has an abnormal vulvar appearance and pain that has persisted despite treatment.
What are the most common vulvar pain syndromes?
Dr. Lonky: If you were to rank vulvar pain syndromes according to their prevalence, what would the most common syndromes be?
Dr. Gunter: Given the misdiagnosis of many women, who are told they have chronic yeast infection, as I mentioned, it’s hard to know which vulvar pain syndromes are most prevalent. I suspect that lichen simplex chronicus is most common, followed by vulvodynia, with chronic yeast infection a distant third.
My experience reflects what Nyirjesy and colleagues4 found: 65% to 75% of women referred to my clinic with chronic yeast actually have lichen simplex chronicus or vulvodynia. In postmenopausal women, atrophic vaginitis is also a consideration; it’s becoming more common now that the use of systemic hormone replacement therapy is decreasing.
Dr. Lonky: What about subsets of vulvodynia? Which ones are most common?
Dr. Edwards: There is good evidence of marked overlap among subsets of vulvodynia. The vast majority of women who have vulvodynia experience primarily provoked vestibular pain, regardless of age. However, I find that almost all patients also report pain that extends beyond the vestibule at times, as well as occasional unprovoked pain.
The diagnosis requires the exclusion of other causes of vulvar pain, and the subset is identified by the location of pain (that is, is it strictly localized or generalized or even migratory?) and its provoked or unprovoked nature.
Localized clitoral pain and vulvar pain localized to one side of the vulva are extremely uncommon, but they do occur. And although I rarely encounter teenagers and prepubertal children who have vulvodynia, I do have patients in both age groups who have vulvodynia.
Dr. Lonky: Are there racial differences in the prevalence of vulvodynia?
Dr. Edwards: Although several good studies show that women of African descent and white patients are equally likely to experience vulvodynia, the vast majority (99%) of my patients who have vulvodynia are white. My patients of African descent consult me primarily for itching or discharge.
My local demographics prevent me from judging the likelihood of Asians having vulvodynia, and our Hispanic population has limited access to health care.
In general, I don’t think that demographics are useful in making the diagnosis of vulvodynia.
Do women who have vulvar pain tend to have comorbidities?
Dr. Lonky: Do your patients who have vulvodynia or another vulvar pain syndrome tend to have comorbidities? If so, is this information helpful in establishing the diagnosis and planning therapy?
Dr. Haefner: Women who have vulvodynia often have other medical problems as well. In my practice, when new patients who have vulvodynia complete their intake survey, they often report a history of headache, irritable bowel syndrome, interstitial cystitis, fibromyalgia,6 chronic fatigue syndrome, back pain, and temporomandibular joint (TMJ) disorder. These comorbidities are not particularly helpful in establishing the diagnosis of vulvodynia, but they are an important consideration when choosing therapy for the patient. Often, the medications chosen to treat one condition will also benefit another condition. However, it’s important to check for potential interactions between drugs before prescribing a new treatment.
Dr. Gunter: A significant number of women who have vulvodynia also have other chronic pain syndromes. For example, the incidence of bladder pain syndrome–interstitial cystitis is 68% to 82% among women who have vulvodynia, compared with a baseline rate among all women of 6% to 11%.7-10 The rate of irritable bowel syndrome is more than doubled among women who have vulvodynia, compared with the general population (27% versus 12%).8 Another common comorbidity, hypertonic somatic dysfunction of the pelvic floor, is identified in 10% to 90% of women who have chronic vulvar pain.8,11,12 These women also have a higher incidence of nongenital pain syndromes, such as fibromyalgia, migraine, and TMJ dysfunction, than the general population, as Dr. Haefner noted.8,12,13
Many studies have evaluated psychological and emotional contributions to chronic vulvar pain. Pain and depression are intimately related—the incidence of depression among all people who experience chronic pain ranges from 27% to 54%, compared with 5% to 17% among the general population.14-16 The relationship is complex because chronic illness in general is associated with depression. Nevertheless, several studies have noted an increase in anxiety, stress, and depression among women who have vulvodynia.17-19
I screen every patient for depression using a Patient Health Questionnaire (PHQ-9); I also screen for anxiety. I find that a significant percentage of patients in my clinic are depressed or have an anxiety disorder. Failure to address these comorbidities makes treatment very difficult. I typically prescribe citalopram (Celexa), although there is some question whether it can safely be combined with a tricyclic antidepressant. We also offer stress-reduction classes, teach every patient the value of diaphragmatic breathing, offer mind-body classes for anxiety and stress, and provide intensive programs where the patient can learn important self-care skills, such as pacing (spacing activities throughout the day in a manner that avoids aggravating the pain), and address her anxiety and stress in a more guided manner. We also have a psychologist who specializes in pain for any patient who may need one-on-one counseling.
Dr. Edwards: The presence of comorbidities is somewhat useful in making the diagnosis of vulvodynia. I question my diagnosis, in fact, when a patient who has vulvodynia does not have headaches, low energy, depression, anxiety, irritable bowel syndrome, constipation, fibromyalgia, chronic fatigue, sensitivity to medications, TMJ dysfunction, or urinary symptoms.
How common is pudendal neuralgia?
Dr. Lonky: How prevalent is a finding of pudendal neuralgia?
Dr. Edwards: The prevalence and incidence of pudendal neuralgia are not known. Those who specialize in this condition think it is relatively common. I do not identify or suspect it very often. Its definitive diagnosis and management are outside the purview of the general gynecologist, but the general gynecologist should recognize the symptoms of pudendal neuralgia and refer the patient for evaluation and therapy.
Dr. Lonky: What are those symptoms?
Dr. Haefner: Pudendal neuralgia often occurs following trauma to the pudendal nerve. The pudendal nerve arises from sacral nerves, generally sacral nerves 2 to 4. Several tests can be utilized to diagnose this condition, including quantitative sensor tests, pudendal nerve motor latency tests, electromyography (EMG), and pudendal nerve blocks.20
Nantes Criteria allow for making a diagnosis of pudendal neuralgia (Table 3).21
TABLE 3
Nantes Criteria for pudendal neuralgia by pudendal nerve entrapment
Essential criteria
|
Complementary diagnostic criteria
|
Exclusion criteria
|
Associated signs not excluding the diagnosis
|
| SOURCE: Labat et al.21 Reproduced with permission from Neurology and Urodynamics. |
Initial treatments for pudendal neuralgia should be conservative. Treatments consist of lifestyle changes to prevent flare of disease. Physical therapy, medical management, nerve blocks, and alternative treatments may be beneficial.
Pudendal nerve entrapment is often exacerbated by sitting (not on a toilet seat, however) and is reduced in a standing position. It tends to increase in intensity throughout the day.22 The final treatment for pudendal nerve entrapment is surgery if the nerve is compressed. By this time, the generalist is not generally the provider who performs the surgery.
Dr. Gunter: I believe pudendal neuralgia is sometimes overdiagnosed. EMG studies of the pudendal nerve, often touted as a diagnostic tool, are unreliable (they can be abnormal after vaginal delivery or vaginal hysterectomy, for example). In my experience, bilateral pain is less likely to be pudendal neuralgia; spontaneous bilateral compression neuropathy at exactly the same level is not a common phenomenon in chronic pain.
I reserve the diagnosis of pudendal neuralgia for women who have allodynia in the distribution of the pudendal nerve with severe pain on sitting, and who have exquisite tenderness when pressure is applied over the pudendal nerve (at the level of the ischial spine on vaginal examination). Typically, the vaginal sidewall on the affected side is very sensitive to light touch. I do see pudendal nerve pain after vaginal surgery when there has been some compromise of the pudendal nerve or the sacral plexus. This is typically unilateral pain.
Dr. Lonky: Thank you all. We’ll continue our discussion, with a focus on treatment, in the October 2011 issue.
- Part 2: A bounty of treatment options
(October 2011) - Part 3: Vestibulodynia
(November 2011)
We want to hear from you! Tell us what you think.
Although the incidence of vulvar pain has increased over the past decade—thanks to both greater awareness and increasing numbers of affected women—the phenomenon is not a recent development. As early as 1874, T. Galliard Thomas wrote, “[T]his disorder, although fortunately not very frequent, is by no means very rare.”1 He went on to express “surprise” that it had not been “more generally and fully described.”
Despite the focus Thomas directed to the issue, vulvar pain did not get much attention until the 21st century, when a number of studies began to gauge its prevalence. For example, in a study in Boston of about 5,000 women, the lifetime prevalence of chronic vulvar pain was 16%.2 And in a study in Texas, the prevalence of vulvar pain in an urban, largely minority population was estimated to be 11%.3 The Boston study also reported that “nearly 40% of women chose not to seek treatment, and, of those who did, 60% saw three or more doctors, many of whom could not provide a diagnosis.”2
Clearly, there is a need for comprehensive information on vulvar pain and its causes, symptoms, diagnosis, and treatment. To address the lack of guidance, OBG Management Contributing Editor Neal M. Lonky, MD, assembled a panel of experts on vulvar pain syndromes and invited them to share their considerable knowledge. The ensuing discussion, presented in three parts, offers a gold mine of information.
In this opening article, the panel focuses on causes, symptomatology, and diagnosis of this common complaint. In Part 2, which will appear in the October issue of this journal, the focus is the bounty of treatment options. Part 3 follows in November, when the discussion shifts to vestibulodynia.

The lower vagina and vulva are richly supplied with peripheral nerves and are, therefore, sensitive to pain, particularly the region of the hymeneal ring. Although the pudendal nerve (arrow) courses through the area, it is an uncommon source of vulvar pain.
Common diagnoses—and misdiagnoses
Dr. Lonky: What are the most common diagnoses when vulvar pain is the complaint?
Dr. Gunter: The most common cause of chronic vulvar pain is vulvodynia, although lichen simplex chronicus, chronic yeast infections, and non-neoplastic epithelial disorders, such as lichen sclerosus and lichen planus, can also produce irritation and pain. In postmenopausal women, atrophic vaginitis can also cause a burning pain, although symptoms are typically more vaginal than vulvar. Yeast and lichen simplex chronicus typically produce itching, although sometimes they can present with irritation and pain, so they must be considered in the differential diagnosis. It is important to remember that many women with vulvodynia have used multiple topical agents and may have developed complex hygiene rituals in an attempt to treat their symptoms, which can result in a secondary lichen simplex chronicus.
That said, there is a high frequency of misdiagnosis with yeast. For example, in a study by Nyirjesy and colleagues, two thirds of women who were referred to a tertiary clinic for chronic vulvovaginal candidiasis were found to have a noninfectious entity instead—most commonly lichen simplex chronicus and vulvodynia.4
Dr. Edwards: The most common “diagnosis” for vulvar pain is vulvodynia. However, the definition of vulvodynia is pain—i.e., burning, rawness, irritation, soreness, aching, or stabbing or stinging sensations—in the absence of skin disease, infection, or specific neurologic disease. Therefore, even though the usual cause of vulvar pain is vulvodynia, it is a diagnosis of exclusion, and skin disease, infection, and neurologic disease must be ruled out.
In regard to infection, Candida albicans and bacterial vaginosis (BV) are usually the first conditions that are considered when a patient complains of vulvar pain, but they are not common causes of vulvar pain and are never causes of chronic vulvar pain. Very rarely they may cause recurrent pain that clears, at least briefly, with treatment.
Candida albicans is usually primarily pruritic, and BV produces discharge and odor, sometimes with minor symptoms. Non-albicans Candida (e.g., Candida glabrata) is nearly always asymptomatic, but it occasionally causes irritation and burning.
Group B streptococcus is another infectious entity that very, very occasionally causes irritation and dyspareunia but is usually only a colonizer.
Herpes simplex virus is a cause of recurrent but not chronic pain.
Chronic pain is more likely to be caused by skin disease than by infection. Lichen simplex chronicus causes itching; any pain is due to erosions from scratching.
Dr. Haefner: Several other infectious conditions or their treatments can cause vulvar pain. For example, herpes (particularly primary herpes infection) is classically associated with vulvar pain. The pain is so great that, at times, the patient requires admission for pain control. Surprisingly, despite the known pain of herpes, approximately 80% of patients who have it are unaware of their diagnosis.
Although condyloma is generally a painless condition, many patients complain of pain following treatment for it, whether treatment involves topical medications or laser surgery.
Chancroid is a painful vulvar ulcer. Trichomonas can sometimes be associated with vulvar pain.
Dr. Lonky: What terminology do we use when we discuss vulvar pain?
Dr. Haefner: The current terminology used to describe vulvar pain was published in 2004, after years of debate over nomenclature within the International Society for the Study of Vulvovaginal Disease.5 The terminology lists two major categories of vulvar pain:
- pain related to a specific disorder. This category encompasses numerous conditions that feature an abnormal appearance of the vulva (Table 1).
TABLE 1
Terminology and classification of vulvar pain from the International Society for the Study of Vulvovaginal Disease
|
| SOURCE: Moyal-Barracco and Lynch.5 Reproduced with permission from the Journal of Reproductive Medicine. |
- vulvodynia, in which the vulva appears normal, other than occasional erythema, which is most prominent at the duct openings (vestibular ducts—Bartholin’s and Skene’s).
As for vulvar pain, there are two major forms:
- hyperalgesia (a low threshold for pain)
- allodynia (pain in response to light touch).
Some diseases that are associated with vulvar pain do not qualify for the diagnosis of vulvodynia (Table 2) because they are associated with an abnormal appearance of the vulva.
TABLE 2
Conditions other than vulvodynia that are associated with vulvar pain
| Acute irritant contact dermatitis (e.g., erosion due to podofilox, imiquimod, cantharidin, fluorouracil, or podophyllin toxin) |
| Aphthous ulcer |
| Atrophy |
| Bartholin’s abscess |
| Candidiasis |
| Carcinoma |
| Chronic irritant contact dermatitis |
| Endometriosis |
| Herpes (simplex and zoster) |
| Immunobullous diseases (including cicatricial pemphigoid, pemphigus vulgaris, linear immunoglobulin A disease, etc.) |
| Lichen planus |
| Lichen sclerosus |
| Podophyllin overdose (see above) |
| Prolapsed urethra |
| Sjögren’s syndrome |
| Trauma |
| Trichomoniasis |
| Vulvar intraepithelial neoplasia |
What needs to be ruled out for a diagnosis of vulvodynia?
Dr. Lonky: What skin diseases need to be ruled out before vulvodynia can be diagnosed?
Dr. Edwards: Skin diseases that affect the vulva are usually pruritic—pain is a later sign. Lichen simplex chronicus (also known as eczema) is pruritus caused by any irritant; any pain that arises is produced by visible excoriations from scratching.
Lichen sclerosus manifests as white epithelium that has a crinkling, shiny, or waxy texture. It can produce pain, especially dyspareunia. The pain is caused by erosions that arise from fragility and introital narrowing and inelasticity.
Vulvovaginal lichen planus is usually erosive and preferentially affects mucous membranes, especially the vestibule; it sometimes affects the vagina and mouth, as well.
Desquamative inflammatory vaginitis is most likely a skin disease that affects only the vagina. It involves introital redness and a clinically and microscopically purulent vaginal discharge that also reveals parabasal cells and absent lactobacilli.
Dr. Lonky: You mentioned that neurologic diseases can sometimes cause vulvar pain. Which ones?
Dr. Edwards: Pudendal neuralgia, diabetic neuropathy, and post-herpetic neuralgia are the most common specific neurologic causes of vulvar pain. Multiple sclerosis can also produce pain syndromes. Post-herpetic neuralgia follows herpes zoster—not herpes simplex—virus infection.
Dr. Lonky: Any other conditions that can cause vulvar pain?
Dr. Haefner: Aphthous ulcers are common and are often flared by stress.
Non-neoplastic epithelial disorders are also seen frequently in health-care providers’ offices; many patients who experience them report pain on the vulva.
It is always important to consider cancer when a patient has an abnormal vulvar appearance and pain that has persisted despite treatment.
What are the most common vulvar pain syndromes?
Dr. Lonky: If you were to rank vulvar pain syndromes according to their prevalence, what would the most common syndromes be?
Dr. Gunter: Given the misdiagnosis of many women, who are told they have chronic yeast infection, as I mentioned, it’s hard to know which vulvar pain syndromes are most prevalent. I suspect that lichen simplex chronicus is most common, followed by vulvodynia, with chronic yeast infection a distant third.
My experience reflects what Nyirjesy and colleagues4 found: 65% to 75% of women referred to my clinic with chronic yeast actually have lichen simplex chronicus or vulvodynia. In postmenopausal women, atrophic vaginitis is also a consideration; it’s becoming more common now that the use of systemic hormone replacement therapy is decreasing.
Dr. Lonky: What about subsets of vulvodynia? Which ones are most common?
Dr. Edwards: There is good evidence of marked overlap among subsets of vulvodynia. The vast majority of women who have vulvodynia experience primarily provoked vestibular pain, regardless of age. However, I find that almost all patients also report pain that extends beyond the vestibule at times, as well as occasional unprovoked pain.
The diagnosis requires the exclusion of other causes of vulvar pain, and the subset is identified by the location of pain (that is, is it strictly localized or generalized or even migratory?) and its provoked or unprovoked nature.
Localized clitoral pain and vulvar pain localized to one side of the vulva are extremely uncommon, but they do occur. And although I rarely encounter teenagers and prepubertal children who have vulvodynia, I do have patients in both age groups who have vulvodynia.
Dr. Lonky: Are there racial differences in the prevalence of vulvodynia?
Dr. Edwards: Although several good studies show that women of African descent and white patients are equally likely to experience vulvodynia, the vast majority (99%) of my patients who have vulvodynia are white. My patients of African descent consult me primarily for itching or discharge.
My local demographics prevent me from judging the likelihood of Asians having vulvodynia, and our Hispanic population has limited access to health care.
In general, I don’t think that demographics are useful in making the diagnosis of vulvodynia.
Do women who have vulvar pain tend to have comorbidities?
Dr. Lonky: Do your patients who have vulvodynia or another vulvar pain syndrome tend to have comorbidities? If so, is this information helpful in establishing the diagnosis and planning therapy?
Dr. Haefner: Women who have vulvodynia often have other medical problems as well. In my practice, when new patients who have vulvodynia complete their intake survey, they often report a history of headache, irritable bowel syndrome, interstitial cystitis, fibromyalgia,6 chronic fatigue syndrome, back pain, and temporomandibular joint (TMJ) disorder. These comorbidities are not particularly helpful in establishing the diagnosis of vulvodynia, but they are an important consideration when choosing therapy for the patient. Often, the medications chosen to treat one condition will also benefit another condition. However, it’s important to check for potential interactions between drugs before prescribing a new treatment.
Dr. Gunter: A significant number of women who have vulvodynia also have other chronic pain syndromes. For example, the incidence of bladder pain syndrome–interstitial cystitis is 68% to 82% among women who have vulvodynia, compared with a baseline rate among all women of 6% to 11%.7-10 The rate of irritable bowel syndrome is more than doubled among women who have vulvodynia, compared with the general population (27% versus 12%).8 Another common comorbidity, hypertonic somatic dysfunction of the pelvic floor, is identified in 10% to 90% of women who have chronic vulvar pain.8,11,12 These women also have a higher incidence of nongenital pain syndromes, such as fibromyalgia, migraine, and TMJ dysfunction, than the general population, as Dr. Haefner noted.8,12,13
Many studies have evaluated psychological and emotional contributions to chronic vulvar pain. Pain and depression are intimately related—the incidence of depression among all people who experience chronic pain ranges from 27% to 54%, compared with 5% to 17% among the general population.14-16 The relationship is complex because chronic illness in general is associated with depression. Nevertheless, several studies have noted an increase in anxiety, stress, and depression among women who have vulvodynia.17-19
I screen every patient for depression using a Patient Health Questionnaire (PHQ-9); I also screen for anxiety. I find that a significant percentage of patients in my clinic are depressed or have an anxiety disorder. Failure to address these comorbidities makes treatment very difficult. I typically prescribe citalopram (Celexa), although there is some question whether it can safely be combined with a tricyclic antidepressant. We also offer stress-reduction classes, teach every patient the value of diaphragmatic breathing, offer mind-body classes for anxiety and stress, and provide intensive programs where the patient can learn important self-care skills, such as pacing (spacing activities throughout the day in a manner that avoids aggravating the pain), and address her anxiety and stress in a more guided manner. We also have a psychologist who specializes in pain for any patient who may need one-on-one counseling.
Dr. Edwards: The presence of comorbidities is somewhat useful in making the diagnosis of vulvodynia. I question my diagnosis, in fact, when a patient who has vulvodynia does not have headaches, low energy, depression, anxiety, irritable bowel syndrome, constipation, fibromyalgia, chronic fatigue, sensitivity to medications, TMJ dysfunction, or urinary symptoms.
How common is pudendal neuralgia?
Dr. Lonky: How prevalent is a finding of pudendal neuralgia?
Dr. Edwards: The prevalence and incidence of pudendal neuralgia are not known. Those who specialize in this condition think it is relatively common. I do not identify or suspect it very often. Its definitive diagnosis and management are outside the purview of the general gynecologist, but the general gynecologist should recognize the symptoms of pudendal neuralgia and refer the patient for evaluation and therapy.
Dr. Lonky: What are those symptoms?
Dr. Haefner: Pudendal neuralgia often occurs following trauma to the pudendal nerve. The pudendal nerve arises from sacral nerves, generally sacral nerves 2 to 4. Several tests can be utilized to diagnose this condition, including quantitative sensor tests, pudendal nerve motor latency tests, electromyography (EMG), and pudendal nerve blocks.20
Nantes Criteria allow for making a diagnosis of pudendal neuralgia (Table 3).21
TABLE 3
Nantes Criteria for pudendal neuralgia by pudendal nerve entrapment
Essential criteria
|
Complementary diagnostic criteria
|
Exclusion criteria
|
Associated signs not excluding the diagnosis
|
| SOURCE: Labat et al.21 Reproduced with permission from Neurology and Urodynamics. |
Initial treatments for pudendal neuralgia should be conservative. Treatments consist of lifestyle changes to prevent flare of disease. Physical therapy, medical management, nerve blocks, and alternative treatments may be beneficial.
Pudendal nerve entrapment is often exacerbated by sitting (not on a toilet seat, however) and is reduced in a standing position. It tends to increase in intensity throughout the day.22 The final treatment for pudendal nerve entrapment is surgery if the nerve is compressed. By this time, the generalist is not generally the provider who performs the surgery.
Dr. Gunter: I believe pudendal neuralgia is sometimes overdiagnosed. EMG studies of the pudendal nerve, often touted as a diagnostic tool, are unreliable (they can be abnormal after vaginal delivery or vaginal hysterectomy, for example). In my experience, bilateral pain is less likely to be pudendal neuralgia; spontaneous bilateral compression neuropathy at exactly the same level is not a common phenomenon in chronic pain.
I reserve the diagnosis of pudendal neuralgia for women who have allodynia in the distribution of the pudendal nerve with severe pain on sitting, and who have exquisite tenderness when pressure is applied over the pudendal nerve (at the level of the ischial spine on vaginal examination). Typically, the vaginal sidewall on the affected side is very sensitive to light touch. I do see pudendal nerve pain after vaginal surgery when there has been some compromise of the pudendal nerve or the sacral plexus. This is typically unilateral pain.
Dr. Lonky: Thank you all. We’ll continue our discussion, with a focus on treatment, in the October 2011 issue.
- Part 2: A bounty of treatment options
(October 2011) - Part 3: Vestibulodynia
(November 2011)
We want to hear from you! Tell us what you think.
1. Thomas TG. Practical Treatise on the Diseases of Women. Philadelphia Pa: Henry C. Lea; 1874.
2. Harlow BL, Stewart EG. A population-based assessment of chronic unexplained vulvar pain: have we underestimated the prevalence of vulvodynia? J Am Med Womens Assoc. 2003;58(2):82-88.
3. Lavy RJ, Hynan LS, Haley RW. Prevalence of vulvar pain in an urban minority population. J Reprod Med. 2007;52(1):59-62.
4. Nyirjesy P, Peyton C, Weitz MV, Mathew L, Culhane JF. Causes of chronic vaginitis: analysis of a prospective database of affected women. Obstet Gynecol. 2006;108(5):1185-1191.
5. Moyal-Barracco M, Lynch PJ. 2003 ISSVD terminology and classification of vulvodynia: a historical perspective. J Reprod Med. 2004;49(10):772-777.
6. Yunas MB. Fibromyalgia and overlapping disorders: the unifying concept of central sensitivity syndromes. Semin Arthritis Rheum. 2007;36(6):339-356.
7. Kahn BS, Tatro C, Parsons CL, Willems JJ. Prevalence of interstitial cystitis in vulvodynia patients detected by bladder potassium sensitivity. J Sex Med. 2010;7(2 Pt 2):996-1002.
8. Arnold JD, Bachman GS, Rosen R, Kelly S, Rhoads GG. Vulvodynia: characteristics and associations with comorbidities and quality of life. Obstet Gynecol. 2006;107(3):617-624.
9. Parsons CL, Dell J, Stanford EJ, et al. The prevalence of interstitial cystitis in gynecologic patients with pelvic pain, as detected by intravesical potassium sensitivity. Am J Obstet Gynecol. 2002;187(5):1395-1400.
10. Clemens JQ, Meenan RT, O’Keefe Rosetti MC, et al. Prevalence of interstitial cystitis symptoms in a managed care population. J Urol. 2005;174(2):576-580.
11. Engman M, Lindehammar H, Wijma B. Surface electromyography diagnostics in women with partial vaginismus with or without vulvar vestibulitis and in asymptomatic women. J Psychosom Obstet Gynecol. 2004;25(3-4):281-294.
12. Gunter J. Vulvodynia: new thoughts on a devastating condition. Obstet Gynecol Surv. 2007;62(12):812-819.
13. Gordon AS, Panahlan-Jand M, McComb F, Melegari C, Sharp S. Characteristics of women with vulvar pain disorders: a Web-based survey. J Sex Marital Ther. 2003;29(suppl 1):45.-
14. Whitten CE, Cristobal K. Chronic pain is a chronic condition not just a symptom. Permanente J. 2005;9(3):43.-
15. Manchikanti L, Fellows B, Pampati V, et al. Comparison of psychological status of chronic pain patients and the general population. Pain Physician. 2002;5(1):40-48.
16. Banks SM, Kerns RD. Explaining the high rates of depression in chronic pain: a diathesis-stress framework. Psychological Bulletin. 1996;119(1):95-110.
17. Sadownik LA. Clinical correlates of vulvodynia patients. A prospective study of 300 patients. J Reprod Med. 2000;5:40-48.Editor found in PubMed: Sadownik LA. Clinical profile of vulvodynia patients. A prospective study of 300 patients. J Reprod Med. 2000;45(8):679–684. Could not find the citation listed. Please confirm.
18. Reed BD, Haefner HK, Punch MR, Roth RS, Gorenflo DW, Gillespie BW. Psychosocial and sexual functioning in women with vulvodynia and chronic pelvic pain. A comparative evaluation. J Reprod Med. 2000;45(8):624-632.
19. Landry T, Bergeron S. Biopsychosocial factors associated with dyspareunia in a community sample of adolescent girls. Arch Sex Behav. 2011;June 22.
20. Goldstein A, Pukall C, Goldstein I. When Sex Hurts: A Woman’s Guide to Banishing Sexual Pain. Cambridge Mass: Da Capo Lifelong Books; 2011;117-126.
21. Labat JJ, Riant T, Robert R, Amarenco G, Lefaucheur JP, Rigaud J. Diagnostic criteria for pudendal neuralgia by pudendal nerve entrapment (Nantes criteria). Neurol Urodyn. 2008;27(4):306-310.
22. Popeney C, Answell V, Renney K. Pudendal entrapment as an etiology of chronic perineal pain: Diagnosis and treatment. Neurol Urodyn. 2007;26(6):820-827.
1. Thomas TG. Practical Treatise on the Diseases of Women. Philadelphia Pa: Henry C. Lea; 1874.
2. Harlow BL, Stewart EG. A population-based assessment of chronic unexplained vulvar pain: have we underestimated the prevalence of vulvodynia? J Am Med Womens Assoc. 2003;58(2):82-88.
3. Lavy RJ, Hynan LS, Haley RW. Prevalence of vulvar pain in an urban minority population. J Reprod Med. 2007;52(1):59-62.
4. Nyirjesy P, Peyton C, Weitz MV, Mathew L, Culhane JF. Causes of chronic vaginitis: analysis of a prospective database of affected women. Obstet Gynecol. 2006;108(5):1185-1191.
5. Moyal-Barracco M, Lynch PJ. 2003 ISSVD terminology and classification of vulvodynia: a historical perspective. J Reprod Med. 2004;49(10):772-777.
6. Yunas MB. Fibromyalgia and overlapping disorders: the unifying concept of central sensitivity syndromes. Semin Arthritis Rheum. 2007;36(6):339-356.
7. Kahn BS, Tatro C, Parsons CL, Willems JJ. Prevalence of interstitial cystitis in vulvodynia patients detected by bladder potassium sensitivity. J Sex Med. 2010;7(2 Pt 2):996-1002.
8. Arnold JD, Bachman GS, Rosen R, Kelly S, Rhoads GG. Vulvodynia: characteristics and associations with comorbidities and quality of life. Obstet Gynecol. 2006;107(3):617-624.
9. Parsons CL, Dell J, Stanford EJ, et al. The prevalence of interstitial cystitis in gynecologic patients with pelvic pain, as detected by intravesical potassium sensitivity. Am J Obstet Gynecol. 2002;187(5):1395-1400.
10. Clemens JQ, Meenan RT, O’Keefe Rosetti MC, et al. Prevalence of interstitial cystitis symptoms in a managed care population. J Urol. 2005;174(2):576-580.
11. Engman M, Lindehammar H, Wijma B. Surface electromyography diagnostics in women with partial vaginismus with or without vulvar vestibulitis and in asymptomatic women. J Psychosom Obstet Gynecol. 2004;25(3-4):281-294.
12. Gunter J. Vulvodynia: new thoughts on a devastating condition. Obstet Gynecol Surv. 2007;62(12):812-819.
13. Gordon AS, Panahlan-Jand M, McComb F, Melegari C, Sharp S. Characteristics of women with vulvar pain disorders: a Web-based survey. J Sex Marital Ther. 2003;29(suppl 1):45.-
14. Whitten CE, Cristobal K. Chronic pain is a chronic condition not just a symptom. Permanente J. 2005;9(3):43.-
15. Manchikanti L, Fellows B, Pampati V, et al. Comparison of psychological status of chronic pain patients and the general population. Pain Physician. 2002;5(1):40-48.
16. Banks SM, Kerns RD. Explaining the high rates of depression in chronic pain: a diathesis-stress framework. Psychological Bulletin. 1996;119(1):95-110.
17. Sadownik LA. Clinical correlates of vulvodynia patients. A prospective study of 300 patients. J Reprod Med. 2000;5:40-48.Editor found in PubMed: Sadownik LA. Clinical profile of vulvodynia patients. A prospective study of 300 patients. J Reprod Med. 2000;45(8):679–684. Could not find the citation listed. Please confirm.
18. Reed BD, Haefner HK, Punch MR, Roth RS, Gorenflo DW, Gillespie BW. Psychosocial and sexual functioning in women with vulvodynia and chronic pelvic pain. A comparative evaluation. J Reprod Med. 2000;45(8):624-632.
19. Landry T, Bergeron S. Biopsychosocial factors associated with dyspareunia in a community sample of adolescent girls. Arch Sex Behav. 2011;June 22.
20. Goldstein A, Pukall C, Goldstein I. When Sex Hurts: A Woman’s Guide to Banishing Sexual Pain. Cambridge Mass: Da Capo Lifelong Books; 2011;117-126.
21. Labat JJ, Riant T, Robert R, Amarenco G, Lefaucheur JP, Rigaud J. Diagnostic criteria for pudendal neuralgia by pudendal nerve entrapment (Nantes criteria). Neurol Urodyn. 2008;27(4):306-310.
22. Popeney C, Answell V, Renney K. Pudendal entrapment as an etiology of chronic perineal pain: Diagnosis and treatment. Neurol Urodyn. 2007;26(6):820-827.
IN THIS ARTICLE
Does electronic fetal heart rate monitoring reduce the risk of neonatal death?
Obstetric care providers have two patients: the mother and the fetus. Although it is relatively easy to tell when the mother is unwell, determining the well-being of the fetus is far more difficult.
Several tests have been developed to confirm fetal well-being during labor. The most widely used is electronic fetal heart rate monitoring (EFM), also referred to as fetal cardiotocography. EFM was introduced by Hon and Lee in the 1960s, and is now the most common obstetric procedure in the United States.1,2 It is noninvasive, simple to perform, inexpensive, and readily available in almost all obstetric units. However, despite our best efforts, we have little objective evidence that EFM has improved perinatal outcomes.
A 2006 Cochrane review of 12 randomized, controlled trials (RCTs) involving more than 37,000 women concluded that, compared with intermittent auscultation—the only acceptable control because randomization to no intrapartum monitoring would be unethical—the only benefit of intrapartum EFM was a reduction in the incidence of seizures in the early neonatal period (the number needed to treat to prevent one event was 661). However, this finding did not translate into a diminished risk of seizures after the first week of life. There was otherwise no significant difference in perinatal outcomes, including no difference in the rates of cerebral palsy or death, although EFM was associated with an increased risk of obstetric intervention and operative delivery.3
It is this contradiction between the almost routine use of intrapartum EFM in the United States and the lack of evidence supporting its use that the authors hoped to address in their analysis.
What did Chen et al find?
In the lead-up to their analysis, the authors make a compelling argument that the existing data—including the 12 RCTs summarized in the Cochrane review3—are flawed. They raise specific concerns about such issues as “low-quality” study design, insufficient data in low-risk populations, and the use of pathologic antecedents (such as newborn encephalopathy) instead of cerebral palsy as a clinical endpoint. These are all reasonable and valid critiques.
So how did Chen and colleagues proceed? Did they design and execute a high-quality prospective study to address these issues? Did they reanalyze the existing RCTs using more sophisticated statistical methodology in an effort to correct for these deficiencies?
They did neither. They simply carried out another retrospective study using a large but poorly validated data set. In doing so, they transgressed and indeed aggravated all of the concerns they themselves raised about the existing literature. Specifically:
- Their retrospective analysis is a far inferior study design, compared with the RCTs they criticized
- Their efforts to distinguish between high-risk and low-risk pregnancies were rudimentary at best and relied on reported birth/death certificate data, which—as the authors themselves and the accompanying editorial concede4—is a notoriously unreliable source
- They made no effort to look at any medium- or long-term measures of neurologic injury.
The observation that EFM was associated with a decreased risk of neonatal seizures and 5-minute Apgar scores below 4 is not novel. Neither is the observation that EFM is associated with an increased risk of operative delivery—both cesarean and operative vaginal delivery. The only novel observation in this study is that, in a cohort of 1.7 million singleton pregnancies, EFM appeared to be associated with a decrease in the risk of early neonatal death (defined as death within the first 6 days of life), although no such association was noted for deaths in the late neonatal (7–27 days) or postneonatal (28–364 days) periods.
Limitations of the study design
RCTs remain the gold standard for clinical trials, and for good reason. The absence of randomization in the current study poses significant limitations. It prevents us from understanding why some women received intrapartum EFM while others did not. This makes it impossible to determine if we are comparing two equal groups, a limitation that cannot be overcome even with the most elegant of statistical analyses.
More concerning, however, is the lack of an adequate control group. The authors conclude that “the use of electronic fetal heart rate monitoring was associated with a substantial decrease in early neonatal mortality and morbidity.” This begs the question: compared with what? In the numerous RCTs on this topic, intrapartum EFM was compared head-to-head with a standardized protocol of intermittent auscultation, whereas the comparison group in the current study was women who did not receive EFM.3 Stated differently, the absence of EFM is not equivalent to intermittent auscultation. An alternative and, in my opinion, far more likely explanation for the observed difference in mortality is that the current study compares women who received intrapartum EFM with those who simply had inadequate fetal monitoring in labor. And I am not aware of any report or, for that matter, any obstetric care provider who believes that it is unnecessary to monitor fetal well-being in labor.
The conclusion of this study should have been that adequate monitoring of the fetus in labor can prevent early neonatal death, not that adequate monitoring of the fetus in labor with EFM can prevent early neonatal death. Moreover, the authors’ attempt to deflect this issue by referring to the current study as an example of “reality-based medicine” as opposed to “evidence-based medicine” undermines the very foundation of scientific investigation.
More questions than answers
The major conclusion of this study is that EFM protects against early neonatal death. So why is there no information about cause of death? These data should be readily available from a linked birth/death certificate data set. Such information might help to determine whether the excess early neonatal deaths were related to EFM or, more likely, to other variables surrounding or related to the delivery, such as the inability to perform an emergency cesarean, if indicated, or the lack of providers skilled in neonatal resuscitation.
Not only does this analysis add little to the literature—it muddies the waters even further. In my opinion, the authors missed the boat entirely when it came to interpreting these data—but plaintiff’s attorneys won’t make the same mistake. I anticipate that, in the years ahead, this study will be quoted more by lawyers than by physicians. The study effectively seals the fate of any legal case in which EFM was not used in labor, despite the fact that the best available evidence shows no significant difference in perinatal morbidity and mortality between EFM and other standardized methods of intrapartum fetal monitoring, such as intermittent auscultation.—ERROL R. NORWITZ, MD, PHD
We want to hear from you! Tell us what you think.
1. Hon EH, Lee ST. Electronic evaluation of the fetal heart rate. VIII. Patterns preceding fetal death further observations. Am J Obstet Gynecol. 1963;87:814-826.
2. American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 106: Intrapartum fetal heart rate monitoring: nomenclature interpretation, and general management principles. Obstet Gynecol. 2009;114(1):192-202.
3. Alfirevic Z, Devane D, Gyte GM. Continuous cardiotocography (CTG) as a form of electronic fetal monitoring (EFM) for fetal assessment during labour. Cochrane Database Syst Rev. 2006;3:CD006066.-
4. Devoe LD. Electronic fetal monitoring: Does it really lead to better outcomes? Am J Obstet Gynecol. 2011;204(6):455-456.
Obstetric care providers have two patients: the mother and the fetus. Although it is relatively easy to tell when the mother is unwell, determining the well-being of the fetus is far more difficult.
Several tests have been developed to confirm fetal well-being during labor. The most widely used is electronic fetal heart rate monitoring (EFM), also referred to as fetal cardiotocography. EFM was introduced by Hon and Lee in the 1960s, and is now the most common obstetric procedure in the United States.1,2 It is noninvasive, simple to perform, inexpensive, and readily available in almost all obstetric units. However, despite our best efforts, we have little objective evidence that EFM has improved perinatal outcomes.
A 2006 Cochrane review of 12 randomized, controlled trials (RCTs) involving more than 37,000 women concluded that, compared with intermittent auscultation—the only acceptable control because randomization to no intrapartum monitoring would be unethical—the only benefit of intrapartum EFM was a reduction in the incidence of seizures in the early neonatal period (the number needed to treat to prevent one event was 661). However, this finding did not translate into a diminished risk of seizures after the first week of life. There was otherwise no significant difference in perinatal outcomes, including no difference in the rates of cerebral palsy or death, although EFM was associated with an increased risk of obstetric intervention and operative delivery.3
It is this contradiction between the almost routine use of intrapartum EFM in the United States and the lack of evidence supporting its use that the authors hoped to address in their analysis.
What did Chen et al find?
In the lead-up to their analysis, the authors make a compelling argument that the existing data—including the 12 RCTs summarized in the Cochrane review3—are flawed. They raise specific concerns about such issues as “low-quality” study design, insufficient data in low-risk populations, and the use of pathologic antecedents (such as newborn encephalopathy) instead of cerebral palsy as a clinical endpoint. These are all reasonable and valid critiques.
So how did Chen and colleagues proceed? Did they design and execute a high-quality prospective study to address these issues? Did they reanalyze the existing RCTs using more sophisticated statistical methodology in an effort to correct for these deficiencies?
They did neither. They simply carried out another retrospective study using a large but poorly validated data set. In doing so, they transgressed and indeed aggravated all of the concerns they themselves raised about the existing literature. Specifically:
- Their retrospective analysis is a far inferior study design, compared with the RCTs they criticized
- Their efforts to distinguish between high-risk and low-risk pregnancies were rudimentary at best and relied on reported birth/death certificate data, which—as the authors themselves and the accompanying editorial concede4—is a notoriously unreliable source
- They made no effort to look at any medium- or long-term measures of neurologic injury.
The observation that EFM was associated with a decreased risk of neonatal seizures and 5-minute Apgar scores below 4 is not novel. Neither is the observation that EFM is associated with an increased risk of operative delivery—both cesarean and operative vaginal delivery. The only novel observation in this study is that, in a cohort of 1.7 million singleton pregnancies, EFM appeared to be associated with a decrease in the risk of early neonatal death (defined as death within the first 6 days of life), although no such association was noted for deaths in the late neonatal (7–27 days) or postneonatal (28–364 days) periods.
Limitations of the study design
RCTs remain the gold standard for clinical trials, and for good reason. The absence of randomization in the current study poses significant limitations. It prevents us from understanding why some women received intrapartum EFM while others did not. This makes it impossible to determine if we are comparing two equal groups, a limitation that cannot be overcome even with the most elegant of statistical analyses.
More concerning, however, is the lack of an adequate control group. The authors conclude that “the use of electronic fetal heart rate monitoring was associated with a substantial decrease in early neonatal mortality and morbidity.” This begs the question: compared with what? In the numerous RCTs on this topic, intrapartum EFM was compared head-to-head with a standardized protocol of intermittent auscultation, whereas the comparison group in the current study was women who did not receive EFM.3 Stated differently, the absence of EFM is not equivalent to intermittent auscultation. An alternative and, in my opinion, far more likely explanation for the observed difference in mortality is that the current study compares women who received intrapartum EFM with those who simply had inadequate fetal monitoring in labor. And I am not aware of any report or, for that matter, any obstetric care provider who believes that it is unnecessary to monitor fetal well-being in labor.
The conclusion of this study should have been that adequate monitoring of the fetus in labor can prevent early neonatal death, not that adequate monitoring of the fetus in labor with EFM can prevent early neonatal death. Moreover, the authors’ attempt to deflect this issue by referring to the current study as an example of “reality-based medicine” as opposed to “evidence-based medicine” undermines the very foundation of scientific investigation.
More questions than answers
The major conclusion of this study is that EFM protects against early neonatal death. So why is there no information about cause of death? These data should be readily available from a linked birth/death certificate data set. Such information might help to determine whether the excess early neonatal deaths were related to EFM or, more likely, to other variables surrounding or related to the delivery, such as the inability to perform an emergency cesarean, if indicated, or the lack of providers skilled in neonatal resuscitation.
Not only does this analysis add little to the literature—it muddies the waters even further. In my opinion, the authors missed the boat entirely when it came to interpreting these data—but plaintiff’s attorneys won’t make the same mistake. I anticipate that, in the years ahead, this study will be quoted more by lawyers than by physicians. The study effectively seals the fate of any legal case in which EFM was not used in labor, despite the fact that the best available evidence shows no significant difference in perinatal morbidity and mortality between EFM and other standardized methods of intrapartum fetal monitoring, such as intermittent auscultation.—ERROL R. NORWITZ, MD, PHD
We want to hear from you! Tell us what you think.
Obstetric care providers have two patients: the mother and the fetus. Although it is relatively easy to tell when the mother is unwell, determining the well-being of the fetus is far more difficult.
Several tests have been developed to confirm fetal well-being during labor. The most widely used is electronic fetal heart rate monitoring (EFM), also referred to as fetal cardiotocography. EFM was introduced by Hon and Lee in the 1960s, and is now the most common obstetric procedure in the United States.1,2 It is noninvasive, simple to perform, inexpensive, and readily available in almost all obstetric units. However, despite our best efforts, we have little objective evidence that EFM has improved perinatal outcomes.
A 2006 Cochrane review of 12 randomized, controlled trials (RCTs) involving more than 37,000 women concluded that, compared with intermittent auscultation—the only acceptable control because randomization to no intrapartum monitoring would be unethical—the only benefit of intrapartum EFM was a reduction in the incidence of seizures in the early neonatal period (the number needed to treat to prevent one event was 661). However, this finding did not translate into a diminished risk of seizures after the first week of life. There was otherwise no significant difference in perinatal outcomes, including no difference in the rates of cerebral palsy or death, although EFM was associated with an increased risk of obstetric intervention and operative delivery.3
It is this contradiction between the almost routine use of intrapartum EFM in the United States and the lack of evidence supporting its use that the authors hoped to address in their analysis.
What did Chen et al find?
In the lead-up to their analysis, the authors make a compelling argument that the existing data—including the 12 RCTs summarized in the Cochrane review3—are flawed. They raise specific concerns about such issues as “low-quality” study design, insufficient data in low-risk populations, and the use of pathologic antecedents (such as newborn encephalopathy) instead of cerebral palsy as a clinical endpoint. These are all reasonable and valid critiques.
So how did Chen and colleagues proceed? Did they design and execute a high-quality prospective study to address these issues? Did they reanalyze the existing RCTs using more sophisticated statistical methodology in an effort to correct for these deficiencies?
They did neither. They simply carried out another retrospective study using a large but poorly validated data set. In doing so, they transgressed and indeed aggravated all of the concerns they themselves raised about the existing literature. Specifically:
- Their retrospective analysis is a far inferior study design, compared with the RCTs they criticized
- Their efforts to distinguish between high-risk and low-risk pregnancies were rudimentary at best and relied on reported birth/death certificate data, which—as the authors themselves and the accompanying editorial concede4—is a notoriously unreliable source
- They made no effort to look at any medium- or long-term measures of neurologic injury.
The observation that EFM was associated with a decreased risk of neonatal seizures and 5-minute Apgar scores below 4 is not novel. Neither is the observation that EFM is associated with an increased risk of operative delivery—both cesarean and operative vaginal delivery. The only novel observation in this study is that, in a cohort of 1.7 million singleton pregnancies, EFM appeared to be associated with a decrease in the risk of early neonatal death (defined as death within the first 6 days of life), although no such association was noted for deaths in the late neonatal (7–27 days) or postneonatal (28–364 days) periods.
Limitations of the study design
RCTs remain the gold standard for clinical trials, and for good reason. The absence of randomization in the current study poses significant limitations. It prevents us from understanding why some women received intrapartum EFM while others did not. This makes it impossible to determine if we are comparing two equal groups, a limitation that cannot be overcome even with the most elegant of statistical analyses.
More concerning, however, is the lack of an adequate control group. The authors conclude that “the use of electronic fetal heart rate monitoring was associated with a substantial decrease in early neonatal mortality and morbidity.” This begs the question: compared with what? In the numerous RCTs on this topic, intrapartum EFM was compared head-to-head with a standardized protocol of intermittent auscultation, whereas the comparison group in the current study was women who did not receive EFM.3 Stated differently, the absence of EFM is not equivalent to intermittent auscultation. An alternative and, in my opinion, far more likely explanation for the observed difference in mortality is that the current study compares women who received intrapartum EFM with those who simply had inadequate fetal monitoring in labor. And I am not aware of any report or, for that matter, any obstetric care provider who believes that it is unnecessary to monitor fetal well-being in labor.
The conclusion of this study should have been that adequate monitoring of the fetus in labor can prevent early neonatal death, not that adequate monitoring of the fetus in labor with EFM can prevent early neonatal death. Moreover, the authors’ attempt to deflect this issue by referring to the current study as an example of “reality-based medicine” as opposed to “evidence-based medicine” undermines the very foundation of scientific investigation.
More questions than answers
The major conclusion of this study is that EFM protects against early neonatal death. So why is there no information about cause of death? These data should be readily available from a linked birth/death certificate data set. Such information might help to determine whether the excess early neonatal deaths were related to EFM or, more likely, to other variables surrounding or related to the delivery, such as the inability to perform an emergency cesarean, if indicated, or the lack of providers skilled in neonatal resuscitation.
Not only does this analysis add little to the literature—it muddies the waters even further. In my opinion, the authors missed the boat entirely when it came to interpreting these data—but plaintiff’s attorneys won’t make the same mistake. I anticipate that, in the years ahead, this study will be quoted more by lawyers than by physicians. The study effectively seals the fate of any legal case in which EFM was not used in labor, despite the fact that the best available evidence shows no significant difference in perinatal morbidity and mortality between EFM and other standardized methods of intrapartum fetal monitoring, such as intermittent auscultation.—ERROL R. NORWITZ, MD, PHD
We want to hear from you! Tell us what you think.
1. Hon EH, Lee ST. Electronic evaluation of the fetal heart rate. VIII. Patterns preceding fetal death further observations. Am J Obstet Gynecol. 1963;87:814-826.
2. American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 106: Intrapartum fetal heart rate monitoring: nomenclature interpretation, and general management principles. Obstet Gynecol. 2009;114(1):192-202.
3. Alfirevic Z, Devane D, Gyte GM. Continuous cardiotocography (CTG) as a form of electronic fetal monitoring (EFM) for fetal assessment during labour. Cochrane Database Syst Rev. 2006;3:CD006066.-
4. Devoe LD. Electronic fetal monitoring: Does it really lead to better outcomes? Am J Obstet Gynecol. 2011;204(6):455-456.
1. Hon EH, Lee ST. Electronic evaluation of the fetal heart rate. VIII. Patterns preceding fetal death further observations. Am J Obstet Gynecol. 1963;87:814-826.
2. American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 106: Intrapartum fetal heart rate monitoring: nomenclature interpretation, and general management principles. Obstet Gynecol. 2009;114(1):192-202.
3. Alfirevic Z, Devane D, Gyte GM. Continuous cardiotocography (CTG) as a form of electronic fetal monitoring (EFM) for fetal assessment during labour. Cochrane Database Syst Rev. 2006;3:CD006066.-
4. Devoe LD. Electronic fetal monitoring: Does it really lead to better outcomes? Am J Obstet Gynecol. 2011;204(6):455-456.
Does minimally invasive hysterectomy cost more than the abdominal approach?
The evolution of the laparoscope from diagnostic tool to operative instrument came as quite a shake-up to some. For example, when Professor Kurt Semm pioneered efforts in operative laparoscopy in Germany in the late 1960s and early 1970s, one of his peers suggested he undergo a scan to rule out brain damage!1 (Until then, the laparoscope had been used only to perform tubal sterilization.)
Fast forward to 1988, when Harry Reich, MD, performed the first laparoscopic hysterectomy.2 Within 5 years of that milestone, the literature was replete with information on laparoscopic-assisted vaginal hysterectomy and total laparoscopic hysterectomy. Surgeons such as Tom Lyons, MD, and Harry Hasson, MD, described the advantages, relative ease, and safety profile of laparoscopic supracervical hysterectomy. However, despite accumulating evidence of the benefits of minimally invasive hysterectomy, including speedier recovery and fewer complications, the president of ACOG felt compelled to question, in a 1992 editorial in Obstetrics & Gynecology, whether operative laparoscopy was a “surgical advance” or a “technical gimmick.”3
By 2007, a Cochrane review of hysterectomy for benign gynecologic disease had concluded that the laparoscopic approach is associated with less intraoperative blood loss, a smaller decline in hemoglobin levels, faster recovery, and fewer wound and abdominal infections and febrile episodes than the abdominal approach is.
Moreover, the laparoscopic approach to hysterectomy is more cost-effective than the abdominal route. In a 2009 study, unadjusted expenditures for laparoscopic hysterectomy averaged $10,868 per case, compared with $12,086 for abdominal hysterectomy and $9,544 for vaginal hysterectomy. Adjusted expenditures for outpatient minimally invasive hysterectomy (laparoscopic or vaginal) were markedly lower than for inpatient abdominal hysterectomy.4
Nevertheless, as I noted in my 2008 presidential address to the 37th Global Congress of Minimally Invasive Gynecology of the AAGL, we have not yet achieved widespread adoption of laparoscopic hysterectomy in the United States. Although more than 95% of cholecystectomy cases are performed laparoscopically—as well as 70% of hernia repairs and 80% of bariatric procedures—only 15% of hysterectomies are laparoscopic, and nearly two thirds are completed using an open abdominal technique.
Jonsdottir et al: Details of the study
Enter the current study: a retrospective analysis from the Division of Minimally Invasive Gynecologic Surgery at Brigham and Women’s Hospital in Boston. It may finally deliver the much-needed death knell for open abdominal hysterectomy. The study of 2,133 women who underwent hysterectomy (1,054 in 2006 and 1,079 in 2009) found that the total number of hysterectomies remained stable while the percentages of abdominal and laparoscopic cases changed markedly. The percentage of hysterectomies that were performed abdominally declined from 64.7% in 2006 to 35.8% in 2009, and the percentage of laparoscopic procedures rose from 17.7% to 46% over the same period.
Along with this change, the overall rate of intraoperative complications decreased significantly—from 7.2% to 4%—and so did the mean percentage of postoperative complications—from 18% to 5.7%. Although operative costs did increase significantly, there was no change in the mean total cost.
Jonsdottir and colleagues cite various reasons for the shift toward minimally invasive hysterectomy:
- a nationwide change in practice patterns
- increasing awareness among patients of the benefits of minimally invasive procedures
- establishment of a minimally invasive gynecologic surgery program at Brigham and Women’s Hospital in 2006
- a preponderance of hysterectomies performed by the gynecologic oncology service or the minimally invasive gynecologic surgery group, both of which are adept in minimally invasive hysterectomy.
Use of the robot increased dramatically
Although robotic hysterectomy was the least common approach identified in this study, the rate nearly tripled over 3 years. This trend toward increasing use of robotic assistance in hysterectomy is seen nationally; according to industry estimates, 26% of all hysterectomy procedures are performed via the robot, as opposed to 18% performed via laparoscopy, 15% performed vaginally, and 38% performed via the open abdominal approach. Although a study by Pasic and colleagues noted increased hospital costs per case with use of the robot for hysterectomy, Jonsdottir and coworkers found the calculated cost to society to be lowest with robotic hysterectomy.5
These findings in context
In another retrospective study that evaluated the use of minimally invasive hysterectomy over a 20-year period at my institution—a tertiary-care, community teaching hospital—Moen and colleagues noted a decrease in the percentage of abdominal hysterectomy from 77% to 35%, whereas minimally invasive hysterectomy (vaginal and laparoscopic approaches) increased from 23% to 64.8%.6 By 2009, 13.6% of hysterectomies at my institution were completed with robotic assistance.
Interestingly, the majority of abdominal, laparoscopic supracervical, and robotic-assisted hysterectomies were performed by generalists, whereas the majority of vaginal and total laparoscopic hysterectomies were performed by fellowship-trained specialists.6
I agree with a recent position statement from the AAGL, which concludes that most hysterectomies for benign indications should utilize the vaginal or laparoscopic approach.7 AAGL also recommends that efforts to facilitate these approaches continue. Surgeons who lack the requisite skills and training should seek assistance from expert colleagues or refer the patient to a surgeon with such expertise.—CHARLES E. MILLER, MD
1. Miller CE. In tribute to Professor Kurt Semm: a true innovator—1927–2003. NewsScope: Newsletter of American Association of Gynecologic Laparoscopists. 2003;17(3):7.-
2. Reich H, DeCaprio J, McGlynn F. Laparoscopic hysterectomy. J Gynecol Surg. 1989;5:213-216.
3. Pitkin RM. Operative laparoscopy: surgical advance or technical gimmick. Obstet Gynecol. 1992;79(3):441-442.
4. Warren L, Ladapo JA, Borah BJ, Gunnarsson CL. Open abdominal versus laparoscopic and vaginal hysterectomy: analysis of a large United States payer measuring quality and cost of care. J Minim Invasive Gynecol. 2009;16(5):581-588.
5. Pasic RP, Rizzo JA, Fang H, Ross S, Moore M, Gunnarsson C. Comparing robot-assisted with conventional laparoscopic hysterectomy: impact on cost and clinical outcomes. J Minim Invasive Gynecol. 2010;17(6):730-738.
6. Moen MD, Noone M, Pesch D, Vassallo B, Miller C. Minimally invasive surgery for hysterectomy: evolving trends over 20 years at a tertiary care community teaching hospital. Female Pelvic Med Reconstr Surg. 2011;17(2):S9.-
7. AAGL Position Statement: route of hysterectomy to treat benign uterine disease. J Minim Invasive Gynecol. 2011;18(1):1-3.
The evolution of the laparoscope from diagnostic tool to operative instrument came as quite a shake-up to some. For example, when Professor Kurt Semm pioneered efforts in operative laparoscopy in Germany in the late 1960s and early 1970s, one of his peers suggested he undergo a scan to rule out brain damage!1 (Until then, the laparoscope had been used only to perform tubal sterilization.)
Fast forward to 1988, when Harry Reich, MD, performed the first laparoscopic hysterectomy.2 Within 5 years of that milestone, the literature was replete with information on laparoscopic-assisted vaginal hysterectomy and total laparoscopic hysterectomy. Surgeons such as Tom Lyons, MD, and Harry Hasson, MD, described the advantages, relative ease, and safety profile of laparoscopic supracervical hysterectomy. However, despite accumulating evidence of the benefits of minimally invasive hysterectomy, including speedier recovery and fewer complications, the president of ACOG felt compelled to question, in a 1992 editorial in Obstetrics & Gynecology, whether operative laparoscopy was a “surgical advance” or a “technical gimmick.”3
By 2007, a Cochrane review of hysterectomy for benign gynecologic disease had concluded that the laparoscopic approach is associated with less intraoperative blood loss, a smaller decline in hemoglobin levels, faster recovery, and fewer wound and abdominal infections and febrile episodes than the abdominal approach is.
Moreover, the laparoscopic approach to hysterectomy is more cost-effective than the abdominal route. In a 2009 study, unadjusted expenditures for laparoscopic hysterectomy averaged $10,868 per case, compared with $12,086 for abdominal hysterectomy and $9,544 for vaginal hysterectomy. Adjusted expenditures for outpatient minimally invasive hysterectomy (laparoscopic or vaginal) were markedly lower than for inpatient abdominal hysterectomy.4
Nevertheless, as I noted in my 2008 presidential address to the 37th Global Congress of Minimally Invasive Gynecology of the AAGL, we have not yet achieved widespread adoption of laparoscopic hysterectomy in the United States. Although more than 95% of cholecystectomy cases are performed laparoscopically—as well as 70% of hernia repairs and 80% of bariatric procedures—only 15% of hysterectomies are laparoscopic, and nearly two thirds are completed using an open abdominal technique.
Jonsdottir et al: Details of the study
Enter the current study: a retrospective analysis from the Division of Minimally Invasive Gynecologic Surgery at Brigham and Women’s Hospital in Boston. It may finally deliver the much-needed death knell for open abdominal hysterectomy. The study of 2,133 women who underwent hysterectomy (1,054 in 2006 and 1,079 in 2009) found that the total number of hysterectomies remained stable while the percentages of abdominal and laparoscopic cases changed markedly. The percentage of hysterectomies that were performed abdominally declined from 64.7% in 2006 to 35.8% in 2009, and the percentage of laparoscopic procedures rose from 17.7% to 46% over the same period.
Along with this change, the overall rate of intraoperative complications decreased significantly—from 7.2% to 4%—and so did the mean percentage of postoperative complications—from 18% to 5.7%. Although operative costs did increase significantly, there was no change in the mean total cost.
Jonsdottir and colleagues cite various reasons for the shift toward minimally invasive hysterectomy:
- a nationwide change in practice patterns
- increasing awareness among patients of the benefits of minimally invasive procedures
- establishment of a minimally invasive gynecologic surgery program at Brigham and Women’s Hospital in 2006
- a preponderance of hysterectomies performed by the gynecologic oncology service or the minimally invasive gynecologic surgery group, both of which are adept in minimally invasive hysterectomy.
Use of the robot increased dramatically
Although robotic hysterectomy was the least common approach identified in this study, the rate nearly tripled over 3 years. This trend toward increasing use of robotic assistance in hysterectomy is seen nationally; according to industry estimates, 26% of all hysterectomy procedures are performed via the robot, as opposed to 18% performed via laparoscopy, 15% performed vaginally, and 38% performed via the open abdominal approach. Although a study by Pasic and colleagues noted increased hospital costs per case with use of the robot for hysterectomy, Jonsdottir and coworkers found the calculated cost to society to be lowest with robotic hysterectomy.5
These findings in context
In another retrospective study that evaluated the use of minimally invasive hysterectomy over a 20-year period at my institution—a tertiary-care, community teaching hospital—Moen and colleagues noted a decrease in the percentage of abdominal hysterectomy from 77% to 35%, whereas minimally invasive hysterectomy (vaginal and laparoscopic approaches) increased from 23% to 64.8%.6 By 2009, 13.6% of hysterectomies at my institution were completed with robotic assistance.
Interestingly, the majority of abdominal, laparoscopic supracervical, and robotic-assisted hysterectomies were performed by generalists, whereas the majority of vaginal and total laparoscopic hysterectomies were performed by fellowship-trained specialists.6
I agree with a recent position statement from the AAGL, which concludes that most hysterectomies for benign indications should utilize the vaginal or laparoscopic approach.7 AAGL also recommends that efforts to facilitate these approaches continue. Surgeons who lack the requisite skills and training should seek assistance from expert colleagues or refer the patient to a surgeon with such expertise.—CHARLES E. MILLER, MD
The evolution of the laparoscope from diagnostic tool to operative instrument came as quite a shake-up to some. For example, when Professor Kurt Semm pioneered efforts in operative laparoscopy in Germany in the late 1960s and early 1970s, one of his peers suggested he undergo a scan to rule out brain damage!1 (Until then, the laparoscope had been used only to perform tubal sterilization.)
Fast forward to 1988, when Harry Reich, MD, performed the first laparoscopic hysterectomy.2 Within 5 years of that milestone, the literature was replete with information on laparoscopic-assisted vaginal hysterectomy and total laparoscopic hysterectomy. Surgeons such as Tom Lyons, MD, and Harry Hasson, MD, described the advantages, relative ease, and safety profile of laparoscopic supracervical hysterectomy. However, despite accumulating evidence of the benefits of minimally invasive hysterectomy, including speedier recovery and fewer complications, the president of ACOG felt compelled to question, in a 1992 editorial in Obstetrics & Gynecology, whether operative laparoscopy was a “surgical advance” or a “technical gimmick.”3
By 2007, a Cochrane review of hysterectomy for benign gynecologic disease had concluded that the laparoscopic approach is associated with less intraoperative blood loss, a smaller decline in hemoglobin levels, faster recovery, and fewer wound and abdominal infections and febrile episodes than the abdominal approach is.
Moreover, the laparoscopic approach to hysterectomy is more cost-effective than the abdominal route. In a 2009 study, unadjusted expenditures for laparoscopic hysterectomy averaged $10,868 per case, compared with $12,086 for abdominal hysterectomy and $9,544 for vaginal hysterectomy. Adjusted expenditures for outpatient minimally invasive hysterectomy (laparoscopic or vaginal) were markedly lower than for inpatient abdominal hysterectomy.4
Nevertheless, as I noted in my 2008 presidential address to the 37th Global Congress of Minimally Invasive Gynecology of the AAGL, we have not yet achieved widespread adoption of laparoscopic hysterectomy in the United States. Although more than 95% of cholecystectomy cases are performed laparoscopically—as well as 70% of hernia repairs and 80% of bariatric procedures—only 15% of hysterectomies are laparoscopic, and nearly two thirds are completed using an open abdominal technique.
Jonsdottir et al: Details of the study
Enter the current study: a retrospective analysis from the Division of Minimally Invasive Gynecologic Surgery at Brigham and Women’s Hospital in Boston. It may finally deliver the much-needed death knell for open abdominal hysterectomy. The study of 2,133 women who underwent hysterectomy (1,054 in 2006 and 1,079 in 2009) found that the total number of hysterectomies remained stable while the percentages of abdominal and laparoscopic cases changed markedly. The percentage of hysterectomies that were performed abdominally declined from 64.7% in 2006 to 35.8% in 2009, and the percentage of laparoscopic procedures rose from 17.7% to 46% over the same period.
Along with this change, the overall rate of intraoperative complications decreased significantly—from 7.2% to 4%—and so did the mean percentage of postoperative complications—from 18% to 5.7%. Although operative costs did increase significantly, there was no change in the mean total cost.
Jonsdottir and colleagues cite various reasons for the shift toward minimally invasive hysterectomy:
- a nationwide change in practice patterns
- increasing awareness among patients of the benefits of minimally invasive procedures
- establishment of a minimally invasive gynecologic surgery program at Brigham and Women’s Hospital in 2006
- a preponderance of hysterectomies performed by the gynecologic oncology service or the minimally invasive gynecologic surgery group, both of which are adept in minimally invasive hysterectomy.
Use of the robot increased dramatically
Although robotic hysterectomy was the least common approach identified in this study, the rate nearly tripled over 3 years. This trend toward increasing use of robotic assistance in hysterectomy is seen nationally; according to industry estimates, 26% of all hysterectomy procedures are performed via the robot, as opposed to 18% performed via laparoscopy, 15% performed vaginally, and 38% performed via the open abdominal approach. Although a study by Pasic and colleagues noted increased hospital costs per case with use of the robot for hysterectomy, Jonsdottir and coworkers found the calculated cost to society to be lowest with robotic hysterectomy.5
These findings in context
In another retrospective study that evaluated the use of minimally invasive hysterectomy over a 20-year period at my institution—a tertiary-care, community teaching hospital—Moen and colleagues noted a decrease in the percentage of abdominal hysterectomy from 77% to 35%, whereas minimally invasive hysterectomy (vaginal and laparoscopic approaches) increased from 23% to 64.8%.6 By 2009, 13.6% of hysterectomies at my institution were completed with robotic assistance.
Interestingly, the majority of abdominal, laparoscopic supracervical, and robotic-assisted hysterectomies were performed by generalists, whereas the majority of vaginal and total laparoscopic hysterectomies were performed by fellowship-trained specialists.6
I agree with a recent position statement from the AAGL, which concludes that most hysterectomies for benign indications should utilize the vaginal or laparoscopic approach.7 AAGL also recommends that efforts to facilitate these approaches continue. Surgeons who lack the requisite skills and training should seek assistance from expert colleagues or refer the patient to a surgeon with such expertise.—CHARLES E. MILLER, MD
1. Miller CE. In tribute to Professor Kurt Semm: a true innovator—1927–2003. NewsScope: Newsletter of American Association of Gynecologic Laparoscopists. 2003;17(3):7.-
2. Reich H, DeCaprio J, McGlynn F. Laparoscopic hysterectomy. J Gynecol Surg. 1989;5:213-216.
3. Pitkin RM. Operative laparoscopy: surgical advance or technical gimmick. Obstet Gynecol. 1992;79(3):441-442.
4. Warren L, Ladapo JA, Borah BJ, Gunnarsson CL. Open abdominal versus laparoscopic and vaginal hysterectomy: analysis of a large United States payer measuring quality and cost of care. J Minim Invasive Gynecol. 2009;16(5):581-588.
5. Pasic RP, Rizzo JA, Fang H, Ross S, Moore M, Gunnarsson C. Comparing robot-assisted with conventional laparoscopic hysterectomy: impact on cost and clinical outcomes. J Minim Invasive Gynecol. 2010;17(6):730-738.
6. Moen MD, Noone M, Pesch D, Vassallo B, Miller C. Minimally invasive surgery for hysterectomy: evolving trends over 20 years at a tertiary care community teaching hospital. Female Pelvic Med Reconstr Surg. 2011;17(2):S9.-
7. AAGL Position Statement: route of hysterectomy to treat benign uterine disease. J Minim Invasive Gynecol. 2011;18(1):1-3.
1. Miller CE. In tribute to Professor Kurt Semm: a true innovator—1927–2003. NewsScope: Newsletter of American Association of Gynecologic Laparoscopists. 2003;17(3):7.-
2. Reich H, DeCaprio J, McGlynn F. Laparoscopic hysterectomy. J Gynecol Surg. 1989;5:213-216.
3. Pitkin RM. Operative laparoscopy: surgical advance or technical gimmick. Obstet Gynecol. 1992;79(3):441-442.
4. Warren L, Ladapo JA, Borah BJ, Gunnarsson CL. Open abdominal versus laparoscopic and vaginal hysterectomy: analysis of a large United States payer measuring quality and cost of care. J Minim Invasive Gynecol. 2009;16(5):581-588.
5. Pasic RP, Rizzo JA, Fang H, Ross S, Moore M, Gunnarsson C. Comparing robot-assisted with conventional laparoscopic hysterectomy: impact on cost and clinical outcomes. J Minim Invasive Gynecol. 2010;17(6):730-738.
6. Moen MD, Noone M, Pesch D, Vassallo B, Miller C. Minimally invasive surgery for hysterectomy: evolving trends over 20 years at a tertiary care community teaching hospital. Female Pelvic Med Reconstr Surg. 2011;17(2):S9.-
7. AAGL Position Statement: route of hysterectomy to treat benign uterine disease. J Minim Invasive Gynecol. 2011;18(1):1-3.
Power Struggles
Many hospitalist practices are started by “traditionalists”: primary-care physicians (PCPs) active in the outpatient and hospital settings. The practice typically grows due in large part to the leadership of the founders. Ultimately, the practice is made up of both the founders and a cadre of part- or full-time hospitalists who don’t work in the outpatient setting. And sometimes they have different incentives and ideas about how the practice should operate.
When these individuals disagree, which group should break the tie—the founding “hybrid” or “rotating” doctors who work part time on the hospitalist service or the doctors who work only as hospitalists?
This is a reasonably common issue for “medical” hospitalist groups, and in many cases is becoming an issue for groups in other specialties that adopt the hospitalist model, such as surgical hospitalists, laborists, etc.
A Common Scenario
Let me illustrate this issue with a composite of several former consulting clients. Let’s say this is a hospitalist practice that serves a 250-bed community hospital. One large private internal medicine group adopted a “rotating hospitalist” model there in the late 1990s. One of the internists provided the daytime hospital coverage for all the group’s patients one week out of every six. Their hospital volume grew quickly. They were asked to take on responsibility for admitting an increasing portion of the unassigned patients, provide care for patients referred by other PCPs who wanted to drop out of hospital work, and increasingly were asked to consult on patients admitted by surgeons.
When faced with this situation, many PCP groups decided to exit the hospital themselves and turn that work over to hospitalists. This group stuck it out. At first, the one doctor in the group covering the hospital each week kept up with the growing volume by simply working harder and longer every day. Eventually, the group sought financial help from the hospital to hire hospitalists who didn’t have outpatient responsibilities.
Years passed, and this PCP group transitioned to employment by the hospital, just like the full-time hospitalists. And by this time, the hospitalist practice was seen as distinct from the original PCP group. About 80% of the staffing was provided by hospitalists who didn’t work in the outpatient setting, the remainder by PCPs who essentially founded the practice. The PCPs chose to continue providing hospital care, both because they found it professionally satisfying and their compensation formula made it attractive for generating production in the hospital.
Tensions arose between the hospitalists and the “hybrids.” The hybrids refused to work night shifts and generally were unable to fill in for unplanned absences by the hospitalists. And because of the PCPs’ compensation formula, and possibly the work ethic of more senior doctors, they favored managing larger patient volumes and decreasing weekend staffing significantly to allow more weekends off in total for everyone. The hospitalists had other ideas about these things, and they were unhappy that the PCPs would have first say about when they could work hospital shifts, thereby decreasing the hospitalists’ scheduling flexibility.
The hospitalists were all within a few years of their residency training, and most of the PCPs were midcareer. This created a social divide, making it that much more difficult for the two groups to work through the issues. While the hybrid doctors saw the hospitalists as good clinicians, and vice versa, each group said: “The other guys are difficult to work with. They don’t understand what it is like for us.”
Need for Paradigm Shift
At many sites, the doctors and their administrative counterparts get stuck in a stalemate and have the same, unhappy conversations repeatedly. These conversations are really gripe sessions more than anything else.
I think the best solution is for everyone to acknowledge the valuable contribution of the hybrid doctors in founding and leading the hospitalist practice through years of growth, but also to begin seeing the hospitalist practice as being owned and governed primarily by the hospitalists who do most of the work. For most issues in which the two factions can’t agree, the hospitalists should have the tie-breaking vote.
While this approach reduces the autonomy of the hybrid doctors to make operational decisions, it doesn’t mean they have zero influence. In fact, the practice usually has a critical need for the hybrid doctors to continue providing some of the staffing. This usually means that the practice will need to ensure it puts together a package of compensation and available shifts on the schedule to ensure the hybrids want to remain active in the practice.
In most cases, all involved should ensure that those hybrid doctors who want to remain active in the hospital, and perform well in the hospitalist practice, should have the opportunity to do so indefinitely.
Compensation Methods for Hybrids
Even if the hybrids and hospitalists are able to harmoniously agree on things like work schedules, the hybrid doctors often have compensation schemes such that when working in the hospital, they have different financial incentives from the hospitalists. (I’m using “hybrid” to describe physicians who work in both inpatient and outpatient settings, usually more time in the office practice.) A common situation is that the production (i.e. wRVUs) generated in the hospital counts toward their office productivity. So the hybrids and the hospitalists will have different ideas about how hard they want to work.
The solution here is to divorce the office and hospital compensation schemes. There should be no connection between the compensation in the two settings, and both should be designed to ensure a competitive amount of money and performance incentives appropriate for that setting. Such methods usually mean that a day of work in the office will result in a different-sized paycheck than what comes with a day of work in the hospital.
Dr. Nelson has been a practicing hospitalist since 1988 and is cofounder and past president of SHM. He is a principal in Nelson Flores Hospital Medicine Consultants, a national hospitalist practice management consulting firm. He is course codirector and faculty for SHM’s “Best Practices in Managing a Hospital Medicine Program” course. This column represents his views and is not intended to reflect an official position of SHM.
Many hospitalist practices are started by “traditionalists”: primary-care physicians (PCPs) active in the outpatient and hospital settings. The practice typically grows due in large part to the leadership of the founders. Ultimately, the practice is made up of both the founders and a cadre of part- or full-time hospitalists who don’t work in the outpatient setting. And sometimes they have different incentives and ideas about how the practice should operate.
When these individuals disagree, which group should break the tie—the founding “hybrid” or “rotating” doctors who work part time on the hospitalist service or the doctors who work only as hospitalists?
This is a reasonably common issue for “medical” hospitalist groups, and in many cases is becoming an issue for groups in other specialties that adopt the hospitalist model, such as surgical hospitalists, laborists, etc.
A Common Scenario
Let me illustrate this issue with a composite of several former consulting clients. Let’s say this is a hospitalist practice that serves a 250-bed community hospital. One large private internal medicine group adopted a “rotating hospitalist” model there in the late 1990s. One of the internists provided the daytime hospital coverage for all the group’s patients one week out of every six. Their hospital volume grew quickly. They were asked to take on responsibility for admitting an increasing portion of the unassigned patients, provide care for patients referred by other PCPs who wanted to drop out of hospital work, and increasingly were asked to consult on patients admitted by surgeons.
When faced with this situation, many PCP groups decided to exit the hospital themselves and turn that work over to hospitalists. This group stuck it out. At first, the one doctor in the group covering the hospital each week kept up with the growing volume by simply working harder and longer every day. Eventually, the group sought financial help from the hospital to hire hospitalists who didn’t have outpatient responsibilities.
Years passed, and this PCP group transitioned to employment by the hospital, just like the full-time hospitalists. And by this time, the hospitalist practice was seen as distinct from the original PCP group. About 80% of the staffing was provided by hospitalists who didn’t work in the outpatient setting, the remainder by PCPs who essentially founded the practice. The PCPs chose to continue providing hospital care, both because they found it professionally satisfying and their compensation formula made it attractive for generating production in the hospital.
Tensions arose between the hospitalists and the “hybrids.” The hybrids refused to work night shifts and generally were unable to fill in for unplanned absences by the hospitalists. And because of the PCPs’ compensation formula, and possibly the work ethic of more senior doctors, they favored managing larger patient volumes and decreasing weekend staffing significantly to allow more weekends off in total for everyone. The hospitalists had other ideas about these things, and they were unhappy that the PCPs would have first say about when they could work hospital shifts, thereby decreasing the hospitalists’ scheduling flexibility.
The hospitalists were all within a few years of their residency training, and most of the PCPs were midcareer. This created a social divide, making it that much more difficult for the two groups to work through the issues. While the hybrid doctors saw the hospitalists as good clinicians, and vice versa, each group said: “The other guys are difficult to work with. They don’t understand what it is like for us.”
Need for Paradigm Shift
At many sites, the doctors and their administrative counterparts get stuck in a stalemate and have the same, unhappy conversations repeatedly. These conversations are really gripe sessions more than anything else.
I think the best solution is for everyone to acknowledge the valuable contribution of the hybrid doctors in founding and leading the hospitalist practice through years of growth, but also to begin seeing the hospitalist practice as being owned and governed primarily by the hospitalists who do most of the work. For most issues in which the two factions can’t agree, the hospitalists should have the tie-breaking vote.
While this approach reduces the autonomy of the hybrid doctors to make operational decisions, it doesn’t mean they have zero influence. In fact, the practice usually has a critical need for the hybrid doctors to continue providing some of the staffing. This usually means that the practice will need to ensure it puts together a package of compensation and available shifts on the schedule to ensure the hybrids want to remain active in the practice.
In most cases, all involved should ensure that those hybrid doctors who want to remain active in the hospital, and perform well in the hospitalist practice, should have the opportunity to do so indefinitely.
Compensation Methods for Hybrids
Even if the hybrids and hospitalists are able to harmoniously agree on things like work schedules, the hybrid doctors often have compensation schemes such that when working in the hospital, they have different financial incentives from the hospitalists. (I’m using “hybrid” to describe physicians who work in both inpatient and outpatient settings, usually more time in the office practice.) A common situation is that the production (i.e. wRVUs) generated in the hospital counts toward their office productivity. So the hybrids and the hospitalists will have different ideas about how hard they want to work.
The solution here is to divorce the office and hospital compensation schemes. There should be no connection between the compensation in the two settings, and both should be designed to ensure a competitive amount of money and performance incentives appropriate for that setting. Such methods usually mean that a day of work in the office will result in a different-sized paycheck than what comes with a day of work in the hospital.
Dr. Nelson has been a practicing hospitalist since 1988 and is cofounder and past president of SHM. He is a principal in Nelson Flores Hospital Medicine Consultants, a national hospitalist practice management consulting firm. He is course codirector and faculty for SHM’s “Best Practices in Managing a Hospital Medicine Program” course. This column represents his views and is not intended to reflect an official position of SHM.
Many hospitalist practices are started by “traditionalists”: primary-care physicians (PCPs) active in the outpatient and hospital settings. The practice typically grows due in large part to the leadership of the founders. Ultimately, the practice is made up of both the founders and a cadre of part- or full-time hospitalists who don’t work in the outpatient setting. And sometimes they have different incentives and ideas about how the practice should operate.
When these individuals disagree, which group should break the tie—the founding “hybrid” or “rotating” doctors who work part time on the hospitalist service or the doctors who work only as hospitalists?
This is a reasonably common issue for “medical” hospitalist groups, and in many cases is becoming an issue for groups in other specialties that adopt the hospitalist model, such as surgical hospitalists, laborists, etc.
A Common Scenario
Let me illustrate this issue with a composite of several former consulting clients. Let’s say this is a hospitalist practice that serves a 250-bed community hospital. One large private internal medicine group adopted a “rotating hospitalist” model there in the late 1990s. One of the internists provided the daytime hospital coverage for all the group’s patients one week out of every six. Their hospital volume grew quickly. They were asked to take on responsibility for admitting an increasing portion of the unassigned patients, provide care for patients referred by other PCPs who wanted to drop out of hospital work, and increasingly were asked to consult on patients admitted by surgeons.
When faced with this situation, many PCP groups decided to exit the hospital themselves and turn that work over to hospitalists. This group stuck it out. At first, the one doctor in the group covering the hospital each week kept up with the growing volume by simply working harder and longer every day. Eventually, the group sought financial help from the hospital to hire hospitalists who didn’t have outpatient responsibilities.
Years passed, and this PCP group transitioned to employment by the hospital, just like the full-time hospitalists. And by this time, the hospitalist practice was seen as distinct from the original PCP group. About 80% of the staffing was provided by hospitalists who didn’t work in the outpatient setting, the remainder by PCPs who essentially founded the practice. The PCPs chose to continue providing hospital care, both because they found it professionally satisfying and their compensation formula made it attractive for generating production in the hospital.
Tensions arose between the hospitalists and the “hybrids.” The hybrids refused to work night shifts and generally were unable to fill in for unplanned absences by the hospitalists. And because of the PCPs’ compensation formula, and possibly the work ethic of more senior doctors, they favored managing larger patient volumes and decreasing weekend staffing significantly to allow more weekends off in total for everyone. The hospitalists had other ideas about these things, and they were unhappy that the PCPs would have first say about when they could work hospital shifts, thereby decreasing the hospitalists’ scheduling flexibility.
The hospitalists were all within a few years of their residency training, and most of the PCPs were midcareer. This created a social divide, making it that much more difficult for the two groups to work through the issues. While the hybrid doctors saw the hospitalists as good clinicians, and vice versa, each group said: “The other guys are difficult to work with. They don’t understand what it is like for us.”
Need for Paradigm Shift
At many sites, the doctors and their administrative counterparts get stuck in a stalemate and have the same, unhappy conversations repeatedly. These conversations are really gripe sessions more than anything else.
I think the best solution is for everyone to acknowledge the valuable contribution of the hybrid doctors in founding and leading the hospitalist practice through years of growth, but also to begin seeing the hospitalist practice as being owned and governed primarily by the hospitalists who do most of the work. For most issues in which the two factions can’t agree, the hospitalists should have the tie-breaking vote.
While this approach reduces the autonomy of the hybrid doctors to make operational decisions, it doesn’t mean they have zero influence. In fact, the practice usually has a critical need for the hybrid doctors to continue providing some of the staffing. This usually means that the practice will need to ensure it puts together a package of compensation and available shifts on the schedule to ensure the hybrids want to remain active in the practice.
In most cases, all involved should ensure that those hybrid doctors who want to remain active in the hospital, and perform well in the hospitalist practice, should have the opportunity to do so indefinitely.
Compensation Methods for Hybrids
Even if the hybrids and hospitalists are able to harmoniously agree on things like work schedules, the hybrid doctors often have compensation schemes such that when working in the hospital, they have different financial incentives from the hospitalists. (I’m using “hybrid” to describe physicians who work in both inpatient and outpatient settings, usually more time in the office practice.) A common situation is that the production (i.e. wRVUs) generated in the hospital counts toward their office productivity. So the hybrids and the hospitalists will have different ideas about how hard they want to work.
The solution here is to divorce the office and hospital compensation schemes. There should be no connection between the compensation in the two settings, and both should be designed to ensure a competitive amount of money and performance incentives appropriate for that setting. Such methods usually mean that a day of work in the office will result in a different-sized paycheck than what comes with a day of work in the hospital.
Dr. Nelson has been a practicing hospitalist since 1988 and is cofounder and past president of SHM. He is a principal in Nelson Flores Hospital Medicine Consultants, a national hospitalist practice management consulting firm. He is course codirector and faculty for SHM’s “Best Practices in Managing a Hospital Medicine Program” course. This column represents his views and is not intended to reflect an official position of SHM.
Does the use of multiple maneuvers in the management of shoulder dystocia increase the risk of neonatal injury?
- Pelvic injury from the McRoberts maneuver?
(Medical Verdicts, August 2011)
Shoulder dystocia occurs in 0.6% to 1.4% of vaginal births and is highly unpredictable. It can cause maternal lacerations, increase the risk of postpartum hemorrhage, and lead to major neonatal injuries such as fractures of the clavicle or humerus, palsies of the brachial plexus, hypoxic-ischemic encephalopathy (HIE), and, even, death. It is also a leading cause of monetary awards in obstetric-related malpractice litigation.
Hoffman and colleagues analyzed 2,018 cases of shoulder dystocia, 101 of which (5.2%) incurred neonatal injury. A total of 3,751 maneuvers were performed, with an average number of maneuvers per case of 1.86. The total number of maneuvers performed in any given case of shoulder dystocia correlated significantly with the rate of neonatal injury.
When five or more maneuvers were used, the rate of neonatal injury exceeded 20%. The authors did not specify whether the neonatal injuries reflected in this figure included bone fracture as well as peripheral nerve injury, or only the latter. They also failed to specify how many brachial plexus injuries resolved spontaneously.
As for which maneuvers were most effective, delivery of the posterior arm and shoulder had a higher rate of success (84%), compared with the McRoberts maneuver, suprapubic pressure, and other maneuvers (24.3% to 72.0% success rate). However, the authors continue to recommend that the McRoberts maneuver and suprapubic pressure be the first maneuvers utilized when shoulder dystocia occurs.
Large study size was a strength
This study, emanating from 12 centers that make up the Consortium on Safe Labor, is probably the largest to date to examine issues related to shoulder dystocia. The sheer magnitude of this study lends great credence to the findings. Another significant strength: Trained obstetric abstractors reviewed the entire medical record of both the mother and newborn.
That said, as with most studies related to shoulder dystocia, there is the possibility of ascertainment bias. Moreover, the study was not randomized. Nor was there uniformity among cases in terms of the maneuvers used or the order in which they were performed, both of which were based largely on provider preference, theoretical models, and expert opinion.
This analysis encompassed the following maneuvers to manage shoulder dystocia:
- McRoberts maneuver
- suprapubic pressure
- Rubin maneuver
- delivery of the posterior arm
- Woods corkscrew maneuver
- Gaskin maneuver (delivery in the maternal knee-chest position)
- Zavanelli maneuver
- fundal pressure.
ACOG recommends the McRoberts maneuver as an initial intervention, followed by suprapubic pressure, when shoulder dystocia occurs.1
The rate of injury was low
At 5.2%, the rate of clinically relevant injury resulting from shoulder dystocia in this study was low. As the authors note, the reported rate of injury in shoulder dystocia cases in general ranges from 4% to as high as 40%.
In this study, if one excludes the 41 cases of bone fracture (either clavicular or humeral) that occurred, which will heal without long-term sequelae, the rate of injury (i.e., neonatal peripheral nerve injury) was just 3%—60 cases of Erb’s palsy and four cases of Klumpke’s palsy among 2,018 cases of shoulder dystocia.
In general, the rate of HIE related to intractable shoulder dystocia is also exceedingly low. In this study, the authors reported only six cases (0.29%). However, the mean head-to-body delivery time in the cases involving HIE was 10.75 minutes, which is troubling. One case of HIE occurred within a reported time frame of 3 minutes.
No real differences between providers were identified
No significant differences in the rate of injury were observed when shoulder dystocia was managed primarily by a resident physician (7.4%), compared with a midwife (2.9%) or attending physician (5.2%). This finding is somewhat confusing because the authors attributed “primary” management to the individual who delivered the newborn and “thus initiated the maneuvers.” I suspect that when a case was managed by a resident, an attending physician was actively involved.
No causal relationship was established
The authors have made a valiant argument that delivery of the posterior arm should be a priority when shoulder dystocia occurs. However, their findings do not confirm a cause-effect relationship between delivery of the posterior arm and resolution of dystocia, for the following reasons:
- Patients were not randomized to a uniform series of maneuvers.
- The order in which the maneuvers were employed could be determined in only 65.7% of cases.
- Management of shoulder dystocia at the study centers was heterogeneous in nature.
For now, the McRoberts maneuver and suprapubic pressure remain first-line maneuvers of choice. This study, along with several other recent investigations, does suggest that delivery of the posterior arm should be considered if these initial maneuvers are unsuccessful.
As simulation is utilized increasingly in resident learning, providers should become more comfortable facilitating delivery of the posterior arm. —Robert B. Gherman, MD
We want to hear from you! Tell us what you think.
Reference
1. American College of Obstetricians and Gynecologists Committee on Practice Bulletins—Gynecology. ACOG Practice Bulletin No. 40: Shoulder dystocia. Obstet Gynecol. 2002;;100(5 Pt 1)::1045-1050.
- Pelvic injury from the McRoberts maneuver?
(Medical Verdicts, August 2011)
Shoulder dystocia occurs in 0.6% to 1.4% of vaginal births and is highly unpredictable. It can cause maternal lacerations, increase the risk of postpartum hemorrhage, and lead to major neonatal injuries such as fractures of the clavicle or humerus, palsies of the brachial plexus, hypoxic-ischemic encephalopathy (HIE), and, even, death. It is also a leading cause of monetary awards in obstetric-related malpractice litigation.
Hoffman and colleagues analyzed 2,018 cases of shoulder dystocia, 101 of which (5.2%) incurred neonatal injury. A total of 3,751 maneuvers were performed, with an average number of maneuvers per case of 1.86. The total number of maneuvers performed in any given case of shoulder dystocia correlated significantly with the rate of neonatal injury.
When five or more maneuvers were used, the rate of neonatal injury exceeded 20%. The authors did not specify whether the neonatal injuries reflected in this figure included bone fracture as well as peripheral nerve injury, or only the latter. They also failed to specify how many brachial plexus injuries resolved spontaneously.
As for which maneuvers were most effective, delivery of the posterior arm and shoulder had a higher rate of success (84%), compared with the McRoberts maneuver, suprapubic pressure, and other maneuvers (24.3% to 72.0% success rate). However, the authors continue to recommend that the McRoberts maneuver and suprapubic pressure be the first maneuvers utilized when shoulder dystocia occurs.
Large study size was a strength
This study, emanating from 12 centers that make up the Consortium on Safe Labor, is probably the largest to date to examine issues related to shoulder dystocia. The sheer magnitude of this study lends great credence to the findings. Another significant strength: Trained obstetric abstractors reviewed the entire medical record of both the mother and newborn.
That said, as with most studies related to shoulder dystocia, there is the possibility of ascertainment bias. Moreover, the study was not randomized. Nor was there uniformity among cases in terms of the maneuvers used or the order in which they were performed, both of which were based largely on provider preference, theoretical models, and expert opinion.
This analysis encompassed the following maneuvers to manage shoulder dystocia:
- McRoberts maneuver
- suprapubic pressure
- Rubin maneuver
- delivery of the posterior arm
- Woods corkscrew maneuver
- Gaskin maneuver (delivery in the maternal knee-chest position)
- Zavanelli maneuver
- fundal pressure.
ACOG recommends the McRoberts maneuver as an initial intervention, followed by suprapubic pressure, when shoulder dystocia occurs.1
The rate of injury was low
At 5.2%, the rate of clinically relevant injury resulting from shoulder dystocia in this study was low. As the authors note, the reported rate of injury in shoulder dystocia cases in general ranges from 4% to as high as 40%.
In this study, if one excludes the 41 cases of bone fracture (either clavicular or humeral) that occurred, which will heal without long-term sequelae, the rate of injury (i.e., neonatal peripheral nerve injury) was just 3%—60 cases of Erb’s palsy and four cases of Klumpke’s palsy among 2,018 cases of shoulder dystocia.
In general, the rate of HIE related to intractable shoulder dystocia is also exceedingly low. In this study, the authors reported only six cases (0.29%). However, the mean head-to-body delivery time in the cases involving HIE was 10.75 minutes, which is troubling. One case of HIE occurred within a reported time frame of 3 minutes.
No real differences between providers were identified
No significant differences in the rate of injury were observed when shoulder dystocia was managed primarily by a resident physician (7.4%), compared with a midwife (2.9%) or attending physician (5.2%). This finding is somewhat confusing because the authors attributed “primary” management to the individual who delivered the newborn and “thus initiated the maneuvers.” I suspect that when a case was managed by a resident, an attending physician was actively involved.
No causal relationship was established
The authors have made a valiant argument that delivery of the posterior arm should be a priority when shoulder dystocia occurs. However, their findings do not confirm a cause-effect relationship between delivery of the posterior arm and resolution of dystocia, for the following reasons:
- Patients were not randomized to a uniform series of maneuvers.
- The order in which the maneuvers were employed could be determined in only 65.7% of cases.
- Management of shoulder dystocia at the study centers was heterogeneous in nature.
For now, the McRoberts maneuver and suprapubic pressure remain first-line maneuvers of choice. This study, along with several other recent investigations, does suggest that delivery of the posterior arm should be considered if these initial maneuvers are unsuccessful.
As simulation is utilized increasingly in resident learning, providers should become more comfortable facilitating delivery of the posterior arm. —Robert B. Gherman, MD
We want to hear from you! Tell us what you think.
- Pelvic injury from the McRoberts maneuver?
(Medical Verdicts, August 2011)
Shoulder dystocia occurs in 0.6% to 1.4% of vaginal births and is highly unpredictable. It can cause maternal lacerations, increase the risk of postpartum hemorrhage, and lead to major neonatal injuries such as fractures of the clavicle or humerus, palsies of the brachial plexus, hypoxic-ischemic encephalopathy (HIE), and, even, death. It is also a leading cause of monetary awards in obstetric-related malpractice litigation.
Hoffman and colleagues analyzed 2,018 cases of shoulder dystocia, 101 of which (5.2%) incurred neonatal injury. A total of 3,751 maneuvers were performed, with an average number of maneuvers per case of 1.86. The total number of maneuvers performed in any given case of shoulder dystocia correlated significantly with the rate of neonatal injury.
When five or more maneuvers were used, the rate of neonatal injury exceeded 20%. The authors did not specify whether the neonatal injuries reflected in this figure included bone fracture as well as peripheral nerve injury, or only the latter. They also failed to specify how many brachial plexus injuries resolved spontaneously.
As for which maneuvers were most effective, delivery of the posterior arm and shoulder had a higher rate of success (84%), compared with the McRoberts maneuver, suprapubic pressure, and other maneuvers (24.3% to 72.0% success rate). However, the authors continue to recommend that the McRoberts maneuver and suprapubic pressure be the first maneuvers utilized when shoulder dystocia occurs.
Large study size was a strength
This study, emanating from 12 centers that make up the Consortium on Safe Labor, is probably the largest to date to examine issues related to shoulder dystocia. The sheer magnitude of this study lends great credence to the findings. Another significant strength: Trained obstetric abstractors reviewed the entire medical record of both the mother and newborn.
That said, as with most studies related to shoulder dystocia, there is the possibility of ascertainment bias. Moreover, the study was not randomized. Nor was there uniformity among cases in terms of the maneuvers used or the order in which they were performed, both of which were based largely on provider preference, theoretical models, and expert opinion.
This analysis encompassed the following maneuvers to manage shoulder dystocia:
- McRoberts maneuver
- suprapubic pressure
- Rubin maneuver
- delivery of the posterior arm
- Woods corkscrew maneuver
- Gaskin maneuver (delivery in the maternal knee-chest position)
- Zavanelli maneuver
- fundal pressure.
ACOG recommends the McRoberts maneuver as an initial intervention, followed by suprapubic pressure, when shoulder dystocia occurs.1
The rate of injury was low
At 5.2%, the rate of clinically relevant injury resulting from shoulder dystocia in this study was low. As the authors note, the reported rate of injury in shoulder dystocia cases in general ranges from 4% to as high as 40%.
In this study, if one excludes the 41 cases of bone fracture (either clavicular or humeral) that occurred, which will heal without long-term sequelae, the rate of injury (i.e., neonatal peripheral nerve injury) was just 3%—60 cases of Erb’s palsy and four cases of Klumpke’s palsy among 2,018 cases of shoulder dystocia.
In general, the rate of HIE related to intractable shoulder dystocia is also exceedingly low. In this study, the authors reported only six cases (0.29%). However, the mean head-to-body delivery time in the cases involving HIE was 10.75 minutes, which is troubling. One case of HIE occurred within a reported time frame of 3 minutes.
No real differences between providers were identified
No significant differences in the rate of injury were observed when shoulder dystocia was managed primarily by a resident physician (7.4%), compared with a midwife (2.9%) or attending physician (5.2%). This finding is somewhat confusing because the authors attributed “primary” management to the individual who delivered the newborn and “thus initiated the maneuvers.” I suspect that when a case was managed by a resident, an attending physician was actively involved.
No causal relationship was established
The authors have made a valiant argument that delivery of the posterior arm should be a priority when shoulder dystocia occurs. However, their findings do not confirm a cause-effect relationship between delivery of the posterior arm and resolution of dystocia, for the following reasons:
- Patients were not randomized to a uniform series of maneuvers.
- The order in which the maneuvers were employed could be determined in only 65.7% of cases.
- Management of shoulder dystocia at the study centers was heterogeneous in nature.
For now, the McRoberts maneuver and suprapubic pressure remain first-line maneuvers of choice. This study, along with several other recent investigations, does suggest that delivery of the posterior arm should be considered if these initial maneuvers are unsuccessful.
As simulation is utilized increasingly in resident learning, providers should become more comfortable facilitating delivery of the posterior arm. —Robert B. Gherman, MD
We want to hear from you! Tell us what you think.
Reference
1. American College of Obstetricians and Gynecologists Committee on Practice Bulletins—Gynecology. ACOG Practice Bulletin No. 40: Shoulder dystocia. Obstet Gynecol. 2002;;100(5 Pt 1)::1045-1050.
Reference
1. American College of Obstetricians and Gynecologists Committee on Practice Bulletins—Gynecology. ACOG Practice Bulletin No. 40: Shoulder dystocia. Obstet Gynecol. 2002;;100(5 Pt 1)::1045-1050.
How should you evaluate a patient who has a cytologic diagnosis of atypical glandular cells (AGC)?
AGC is a relatively uncommon cytologic finding, with a mean reporting rate in 2003 of just 0.4% in the United States, according to guidelines from the ASCCP, the society for lower genital tract disease.1
“Although AGC is frequently caused by benign conditions, such as reactive changes and polyps, clinicians should be aware that it is not uncommon for AGC to be associated with a significant underlying neoplastic condition, including adenocarcinomas of the cervix, endometrium, ovary, and fallopian tube. Recent series have reported that 9%–38% of women with AGC have significant neoplasia…and 3%–17% have invasive cancer,” the guidelines state.1
In recent years, evidence-based guidelines such as this one have helped clinicians improve the care that they offer to their patients. Nevertheless, the cytologic diagnosis of AGC presents a dilemma. The significant rate of neoplasia associated with this finding, combined with the lack of sensitivity of methods of evaluation, is responsible for this quandary. Although clinicians try to avoid over-testing, there is a real concern about missing a critical diagnosis.
As ASCCP guidelines point out, all of the diagnostic testing done to investigate this cytologic finding lack sensitivity.1 This observation led to the recommendation that multiple modalities be combined in the evaluation of these patients.1 Previously published data suggest that women with AGC cytology are under-managed in both their initial and secondary evaluations.2
Authors explore management in women found to have cancer
Cheng and colleagues report on a large series of women who had a first-time diagnosis of AGC. These patients were drawn from an extensive, heterogeneous screening population in Taiwan. The report focuses only on patients who were ultimately found to have a diagnosis of invasive cancer—not those who had premalignant conditions such as cervical intraepithelial neoplasia (CIN) 2, CIN 3, adenocarcinoma in situ, or endometrial hyperplasia. The study confirms a high relative risk (RR) of gynecologic malignancy among women with a cytologic finding of AGC. The greatest risk is cervical cancer (RR, 17.85), followed by uterine cancer (RR, 5.68) and ovarian cancer (RR, 2.04).
Should we include US imaging in our assessment?
Current guidelines recommend colposcopy, endocervical curettage, cervical biopsy, and human papillomavirus (HPV) DNA testing (for high-risk types only) in women who have AGC. They also call for endometrial assessment among women at risk of uterine malignancy, including all women older than 35 years and those younger than 35 who have risk factors, such as unexplained vaginal bleeding, or a condition that suggests chronic anovulation.
Cheng and colleagues also include a recommendation for US imaging in women who have AGC, because of the risk of ovarian cancer. However, although the relative risk of ovarian cancer is roughly doubled among women who have AGC, compared with women with normal cytology, the absolute risk remains quite low. In this sample of 8,281 patients, for example, there were only 12 cases (0.14%).
Nor do the data presented by Cheng and colleagues make a compelling case for the addition of routine US in these patients. However, based on the results of this study, it may be prudent to consider US in certain risk groups when evaluation of the cervix and uterus is negative, such as women who have a family history of ovarian cancer, women who have breast cancer, or women who have a cytologic finding of adenocarcinoma.
HPV DNA testing was not included in this study
The authors suggest, instead, that this modality could be used for triage. However, both of the studies they cite to back this recommendation precede the 2006 ASCCP guidelines, which recommend testing for high-risk HPV types as part of the initial evaluation of women who have AGC but advise against using HPV DNA testing for triage.
More recent studies have confirmed that the sensitivity of HPV DNA testing is only approximately 80%, which is not sufficient for triage in this population.3 Such testing may reassure clinicians—but only after complete evaluation of the cervix and uterus. It can be used to guide subsequent surveillance after diagnostic studies, however.
We should adhere to ASCCP guidelines for the management of women who have AGC cytology, by performing:
- colposcopy with endocervical sampling in all women with this designation
- endometrial sampling in women 35 years and older and in women younger than 35 who have unexplained vaginal bleeding, chronic anovulation, or other clinical indications suggesting that they have a heightened risk of neoplastic endometrial lesions
- HPV DNA testing at the time of colposcopy.
As we mentioned, pelvic ultrasonography may be advisable if the patient has certain risk factors for ovarian cancer, such as a family history of ovarian malignancy, age older than 60 years, persistent AGC, or a cytologic finding of adenocarcinoma without uterine or cervical origin.
Compliance with these guidelines will reduce the risk of a missed diagnosis of neoplasia or cancer without excessive testing.
Women who have persistent AGC despite a negative comprehensive evaluation may be at increased risk of clinically significant disease. Clinicians should be aware of the guidelines for these women, which include the need for excisional biopsies and endometrial evaluation.4—Charles J. Dunton, MD, and Norman A. Brest, MD
We want to hear from you! Tell us what you think.
1. Wright TC, Jr, Massad LS, Dunton CJ, Spitzer M, Wilkinson EJ, Solomon D. 2006 Amercian Society for Colposcopy and Cervical Pathology-sponsored Consensus Conference 2006 consensus guidelines for the management of women with abnormal cervical cancer screening tests. Am J Obstet Gynecol. 2007;197(4):346-355.
2. Sharpless KE, Schnatz PF, Mandavilli S, Greene JF, Sorosky JI. Lack of adherence to practice guidelines for women with atypical glandular cells on cervical cytology. Obstet Gynecol. 2005;105(3):501-506.
3. Schnatz PF, Sharpless KE, O’Sullivan DM. Use of human papillomavirus testing in the management of atypical glandular cells. J Low Genit Tract Dis. 2009;13(2):94-101.
4. Dunton CJ. Management of atypical glandular cells and adenocarcinoma in situ. Obstet Gynecol Clin North Am. 2008;35(4):623-632.
AGC is a relatively uncommon cytologic finding, with a mean reporting rate in 2003 of just 0.4% in the United States, according to guidelines from the ASCCP, the society for lower genital tract disease.1
“Although AGC is frequently caused by benign conditions, such as reactive changes and polyps, clinicians should be aware that it is not uncommon for AGC to be associated with a significant underlying neoplastic condition, including adenocarcinomas of the cervix, endometrium, ovary, and fallopian tube. Recent series have reported that 9%–38% of women with AGC have significant neoplasia…and 3%–17% have invasive cancer,” the guidelines state.1
In recent years, evidence-based guidelines such as this one have helped clinicians improve the care that they offer to their patients. Nevertheless, the cytologic diagnosis of AGC presents a dilemma. The significant rate of neoplasia associated with this finding, combined with the lack of sensitivity of methods of evaluation, is responsible for this quandary. Although clinicians try to avoid over-testing, there is a real concern about missing a critical diagnosis.
As ASCCP guidelines point out, all of the diagnostic testing done to investigate this cytologic finding lack sensitivity.1 This observation led to the recommendation that multiple modalities be combined in the evaluation of these patients.1 Previously published data suggest that women with AGC cytology are under-managed in both their initial and secondary evaluations.2
Authors explore management in women found to have cancer
Cheng and colleagues report on a large series of women who had a first-time diagnosis of AGC. These patients were drawn from an extensive, heterogeneous screening population in Taiwan. The report focuses only on patients who were ultimately found to have a diagnosis of invasive cancer—not those who had premalignant conditions such as cervical intraepithelial neoplasia (CIN) 2, CIN 3, adenocarcinoma in situ, or endometrial hyperplasia. The study confirms a high relative risk (RR) of gynecologic malignancy among women with a cytologic finding of AGC. The greatest risk is cervical cancer (RR, 17.85), followed by uterine cancer (RR, 5.68) and ovarian cancer (RR, 2.04).
Should we include US imaging in our assessment?
Current guidelines recommend colposcopy, endocervical curettage, cervical biopsy, and human papillomavirus (HPV) DNA testing (for high-risk types only) in women who have AGC. They also call for endometrial assessment among women at risk of uterine malignancy, including all women older than 35 years and those younger than 35 who have risk factors, such as unexplained vaginal bleeding, or a condition that suggests chronic anovulation.
Cheng and colleagues also include a recommendation for US imaging in women who have AGC, because of the risk of ovarian cancer. However, although the relative risk of ovarian cancer is roughly doubled among women who have AGC, compared with women with normal cytology, the absolute risk remains quite low. In this sample of 8,281 patients, for example, there were only 12 cases (0.14%).
Nor do the data presented by Cheng and colleagues make a compelling case for the addition of routine US in these patients. However, based on the results of this study, it may be prudent to consider US in certain risk groups when evaluation of the cervix and uterus is negative, such as women who have a family history of ovarian cancer, women who have breast cancer, or women who have a cytologic finding of adenocarcinoma.
HPV DNA testing was not included in this study
The authors suggest, instead, that this modality could be used for triage. However, both of the studies they cite to back this recommendation precede the 2006 ASCCP guidelines, which recommend testing for high-risk HPV types as part of the initial evaluation of women who have AGC but advise against using HPV DNA testing for triage.
More recent studies have confirmed that the sensitivity of HPV DNA testing is only approximately 80%, which is not sufficient for triage in this population.3 Such testing may reassure clinicians—but only after complete evaluation of the cervix and uterus. It can be used to guide subsequent surveillance after diagnostic studies, however.
We should adhere to ASCCP guidelines for the management of women who have AGC cytology, by performing:
- colposcopy with endocervical sampling in all women with this designation
- endometrial sampling in women 35 years and older and in women younger than 35 who have unexplained vaginal bleeding, chronic anovulation, or other clinical indications suggesting that they have a heightened risk of neoplastic endometrial lesions
- HPV DNA testing at the time of colposcopy.
As we mentioned, pelvic ultrasonography may be advisable if the patient has certain risk factors for ovarian cancer, such as a family history of ovarian malignancy, age older than 60 years, persistent AGC, or a cytologic finding of adenocarcinoma without uterine or cervical origin.
Compliance with these guidelines will reduce the risk of a missed diagnosis of neoplasia or cancer without excessive testing.
Women who have persistent AGC despite a negative comprehensive evaluation may be at increased risk of clinically significant disease. Clinicians should be aware of the guidelines for these women, which include the need for excisional biopsies and endometrial evaluation.4—Charles J. Dunton, MD, and Norman A. Brest, MD
We want to hear from you! Tell us what you think.
AGC is a relatively uncommon cytologic finding, with a mean reporting rate in 2003 of just 0.4% in the United States, according to guidelines from the ASCCP, the society for lower genital tract disease.1
“Although AGC is frequently caused by benign conditions, such as reactive changes and polyps, clinicians should be aware that it is not uncommon for AGC to be associated with a significant underlying neoplastic condition, including adenocarcinomas of the cervix, endometrium, ovary, and fallopian tube. Recent series have reported that 9%–38% of women with AGC have significant neoplasia…and 3%–17% have invasive cancer,” the guidelines state.1
In recent years, evidence-based guidelines such as this one have helped clinicians improve the care that they offer to their patients. Nevertheless, the cytologic diagnosis of AGC presents a dilemma. The significant rate of neoplasia associated with this finding, combined with the lack of sensitivity of methods of evaluation, is responsible for this quandary. Although clinicians try to avoid over-testing, there is a real concern about missing a critical diagnosis.
As ASCCP guidelines point out, all of the diagnostic testing done to investigate this cytologic finding lack sensitivity.1 This observation led to the recommendation that multiple modalities be combined in the evaluation of these patients.1 Previously published data suggest that women with AGC cytology are under-managed in both their initial and secondary evaluations.2
Authors explore management in women found to have cancer
Cheng and colleagues report on a large series of women who had a first-time diagnosis of AGC. These patients were drawn from an extensive, heterogeneous screening population in Taiwan. The report focuses only on patients who were ultimately found to have a diagnosis of invasive cancer—not those who had premalignant conditions such as cervical intraepithelial neoplasia (CIN) 2, CIN 3, adenocarcinoma in situ, or endometrial hyperplasia. The study confirms a high relative risk (RR) of gynecologic malignancy among women with a cytologic finding of AGC. The greatest risk is cervical cancer (RR, 17.85), followed by uterine cancer (RR, 5.68) and ovarian cancer (RR, 2.04).
Should we include US imaging in our assessment?
Current guidelines recommend colposcopy, endocervical curettage, cervical biopsy, and human papillomavirus (HPV) DNA testing (for high-risk types only) in women who have AGC. They also call for endometrial assessment among women at risk of uterine malignancy, including all women older than 35 years and those younger than 35 who have risk factors, such as unexplained vaginal bleeding, or a condition that suggests chronic anovulation.
Cheng and colleagues also include a recommendation for US imaging in women who have AGC, because of the risk of ovarian cancer. However, although the relative risk of ovarian cancer is roughly doubled among women who have AGC, compared with women with normal cytology, the absolute risk remains quite low. In this sample of 8,281 patients, for example, there were only 12 cases (0.14%).
Nor do the data presented by Cheng and colleagues make a compelling case for the addition of routine US in these patients. However, based on the results of this study, it may be prudent to consider US in certain risk groups when evaluation of the cervix and uterus is negative, such as women who have a family history of ovarian cancer, women who have breast cancer, or women who have a cytologic finding of adenocarcinoma.
HPV DNA testing was not included in this study
The authors suggest, instead, that this modality could be used for triage. However, both of the studies they cite to back this recommendation precede the 2006 ASCCP guidelines, which recommend testing for high-risk HPV types as part of the initial evaluation of women who have AGC but advise against using HPV DNA testing for triage.
More recent studies have confirmed that the sensitivity of HPV DNA testing is only approximately 80%, which is not sufficient for triage in this population.3 Such testing may reassure clinicians—but only after complete evaluation of the cervix and uterus. It can be used to guide subsequent surveillance after diagnostic studies, however.
We should adhere to ASCCP guidelines for the management of women who have AGC cytology, by performing:
- colposcopy with endocervical sampling in all women with this designation
- endometrial sampling in women 35 years and older and in women younger than 35 who have unexplained vaginal bleeding, chronic anovulation, or other clinical indications suggesting that they have a heightened risk of neoplastic endometrial lesions
- HPV DNA testing at the time of colposcopy.
As we mentioned, pelvic ultrasonography may be advisable if the patient has certain risk factors for ovarian cancer, such as a family history of ovarian malignancy, age older than 60 years, persistent AGC, or a cytologic finding of adenocarcinoma without uterine or cervical origin.
Compliance with these guidelines will reduce the risk of a missed diagnosis of neoplasia or cancer without excessive testing.
Women who have persistent AGC despite a negative comprehensive evaluation may be at increased risk of clinically significant disease. Clinicians should be aware of the guidelines for these women, which include the need for excisional biopsies and endometrial evaluation.4—Charles J. Dunton, MD, and Norman A. Brest, MD
We want to hear from you! Tell us what you think.
1. Wright TC, Jr, Massad LS, Dunton CJ, Spitzer M, Wilkinson EJ, Solomon D. 2006 Amercian Society for Colposcopy and Cervical Pathology-sponsored Consensus Conference 2006 consensus guidelines for the management of women with abnormal cervical cancer screening tests. Am J Obstet Gynecol. 2007;197(4):346-355.
2. Sharpless KE, Schnatz PF, Mandavilli S, Greene JF, Sorosky JI. Lack of adherence to practice guidelines for women with atypical glandular cells on cervical cytology. Obstet Gynecol. 2005;105(3):501-506.
3. Schnatz PF, Sharpless KE, O’Sullivan DM. Use of human papillomavirus testing in the management of atypical glandular cells. J Low Genit Tract Dis. 2009;13(2):94-101.
4. Dunton CJ. Management of atypical glandular cells and adenocarcinoma in situ. Obstet Gynecol Clin North Am. 2008;35(4):623-632.
1. Wright TC, Jr, Massad LS, Dunton CJ, Spitzer M, Wilkinson EJ, Solomon D. 2006 Amercian Society for Colposcopy and Cervical Pathology-sponsored Consensus Conference 2006 consensus guidelines for the management of women with abnormal cervical cancer screening tests. Am J Obstet Gynecol. 2007;197(4):346-355.
2. Sharpless KE, Schnatz PF, Mandavilli S, Greene JF, Sorosky JI. Lack of adherence to practice guidelines for women with atypical glandular cells on cervical cytology. Obstet Gynecol. 2005;105(3):501-506.
3. Schnatz PF, Sharpless KE, O’Sullivan DM. Use of human papillomavirus testing in the management of atypical glandular cells. J Low Genit Tract Dis. 2009;13(2):94-101.
4. Dunton CJ. Management of atypical glandular cells and adenocarcinoma in situ. Obstet Gynecol Clin North Am. 2008;35(4):623-632.
Remove the black-box warning for depot medroxyprogesterone acetate!
- Update on contraception
Rachel B. Rapkin, MD; Mitchell D. Creinin, MD (August 2011) - 10 (+1) practical, evidence-based recommendations for you to improve contraceptive care now
Colleen Krajewski, MD; Mark D. Walters, MD (August 2011) - Levonorgestrel or ulipristal: Is one a better emergency contraceptive than the other?
Robert L. Barbieri, MD (Editorial; March 2011) - IUD use in nulliparous and adolescent women
Jennefer A. Russo, MD; Mitchell D. Creinin, MD (Update on Contraception, August 2010)
The Food and Drug Administration (FDA) should remove the black-box warning regarding skeletal health that the agency inserted into labeling for the injectable contraceptive depot medroxyprogesterone acetate (DMPA; Depo-Provera) in 2004. The black box 1) indicates that use of DMPA for longer than 2 years may reduce peak bone mass and place a woman at increased risk of osteoporotic fracture and 2) suggests that ObGyns order bone-density assessment in women who use DMPA long-term.
This warning is not based on scientific evidence. By depriving women of long-term use of a safe, effective contraceptive, the black-box warning damages women’s health and the public health.
The scientific argument for removing the black box
Although DMPA does cause reduced ovarian estradiol production, with a decline in bone mineral density (BMD), complete recovery of BMD occurs approximately 1 or 2 years after discontinuation in adolescent girls1,2 and 3 years after discontinuation in adult women.3,4 BMD trends observed with the use of DMPA parallel those associated with another normal but hypoestrogenic state: breastfeeding. Although BMD declines noted in nursing mothers are similar to those associated with DMPA, breastfeeding one or more infants has not been reported to increase the risk of subsequent osteoporotic fractures.5 The FDA does not warn women against breastfeeding for fear of a theoretical negative impact on skeletal health.
BMD can help predict a risk of osteoporotic fracture in menopausal women, but it is not a valid surrogate endpoint for fracture in women of reproductive age.6 Basing clinical recommendations on invalid surrogate endpoints can cause harm.7-9 Clinical recommendations, including those from the FDA, should be based on sound studies that address important clinical outcomes.
No convincing evidence links the use of DMPA for contraception to fracture. Studies of menopausal women have not suggested that prior use of DMPA increased their risk of osteoporosis.10-12 Two recently published case-control studies, one using a Danish National Patient Registry13 and one based on the United Kingdom Family Practice Research database,14 have demonstrated that DMPA is associated with an elevated risk of fracture in reproductive-age women. However, a cohort analysis using the same British Family Practice database clarifies that the elevated fracture risk observed in women using DMPA occurred before initiation of injectable contraception and was not the result of DMPA use.15

A clearly harmful effect on women’s health
The black-box warning for DMPA has caused clinicians and women to reduce use of this effective method of contraception. The 2008 National Survey of Family Growth shows that the overall percentage of US women, 15 to 44 years of age, who now use DMPA has declined—from 3.3% in 2002 to 2.0% in 2006 to 2008.16
In fact, a survey of ObGyns in Florida found that 1) almost one half of respondents place a time limit on duration of DMPA and 2) two thirds of those respondents base that restriction on the black-box warning.17 Restricting the use of DMPA can lead to more unintended pregnancies and induced abortions.
In the Florida survey, two thirds of respondents order BMD assessment in women who use DMPA, with 58% of those physicians who do so attributing their decision to, again, the black-box warning. Indeed, more than 5% of respondents reported that they selectively prescribe bisphosphonates to women of reproductive age who use DMPA—an expensive practice that is not evidence-based and is potentially dangerous.
FDA guidance is far out of step
The black-box warning for DMPA is not in accord with the guidance provided major medical and public health organizations. The World Health Organization,6 the American College of Obstetricians and Gynecologists,18 the Society for Adolescent Medicine,19 and the Society of Obstetrics and Gynecology of Canada,20 have all stated that skeletal health concerns shouldn’t restrict the use of DMPA, including the duration of use.
In the US Medical Eligibility Criteria for contraceptive use, the Centers for Disease Control and Prevention declared that use of DMPA is Category 1 (no restriction on use) in women 18 to 45 years of age and Category 2 (advantages of the method generally outweigh theoretical or proven risks) in younger women.21 The FDA’s black-box warning is therefore inconsistent with the recommendation of its sister agency.
An opportunity to improve the health of women
The FDA appropriately takes action when a pharmaceutical company mislabels a product or promotes it in a way that isn’t based on sound evidence. Shouldn’t the FDA apply the same scientific standards to itself? Guidance included in DMPA’s black box is not evidence-based—and that reduces use of DMPA in US women and increases the potential for harm. Removing the black box would encourage use of this convenient, effective, and safe contraceptive22 and, in turn, improve the health of women and their families.
Again: We urge the FDA to promptly remove the black-box warning from labeling for DMPA.
Do you agree? We want to hear your opinion: Write to us in care of The Editors, at [email protected]!
Long-acting reversible contraceptives are safe, effective for most women, study shows
But most use other methods because of lack of knowledge and cost concerns
Long-acting reversible contraceptives (LARC) are safe and effective for almost all women of reproductive age, according to an ACOG practice bulletin published in the July issue of Obstetrics & Gynecology.
Eve Espey, MD, MPH, and Rameet H. Singh, MD, MPH, from ACOG in Washington, DC, and colleagues compiled the latest recommendations on LARC use. The recommendations identify candidates for LARCs and help obstetricians and gynecologists manage LARC-related clinical issues.
The investigators reported that the LARC methods currently available have few contraindications, and that most women are eligible to use them. Intrauterine devices (IUDs) are suitable for women with a previous ectopic pregnancy and for women immediately after abortion or miscarriage. Routine antibiotic prophylaxis is not recommended before IUD insertion.
Copper IUD inserted up to 5 days after unprotected intercourse is the most effective method of postcoital contraception. The copper IUD can be used and remains effective for up to 10 continuous years. The levonorgestrel IUD may be effective for up to 7 years.
IUD complications are rare and include expulsion, method failure, and perforation. Implants can be safely inserted at any time after childbirth in non-breastfeeding women, or after 4 weeks for breastfeeding mothers. All LARC methods are safe for nulliparous women and adolescents.
The use of IUDs increased from 1.3% to 5.5% from 2002 to 2006-2008. Despite the benefits of LARCs, the majority of women choose other birth control methods, probably due to lack of knowledge about LARCs and cost concerns.
“LARC methods are the best tool we have to fight against unintended pregnancies, which currently account for 49% of US pregnancies each year,” Espey said in a statement.
Copyright © 2011 HealthDay. All rights reserved.
Has the FDA’s warning about DMPA had a chilling effect? Does the black-box warning prevent you from prescribing DMPA? Or does it alter your approach to using it? How?
To tell us, click here
1. Scholes D, LaCroix AZ, Ichikawa LE, Barlow WE, Ott SM. Change in bone mineral density among adolescent women using and discontinuing depot medroxyprogesterone acetate contraception. Arch Pediatric Adolesc Med. 2005;159(2):139-144.
2. Harel Z, Johnson CC, Gold MA, et al. Recovery of bone mineral density in adolescents following the use of depot medroxyprogesterone acetate contraceptive injections. Contraception. 2010;81(4):281-291.
3. Scholes D, LaCroix AZ, Ichikawa LE, Barlow WE, Ott SM. Injectable hormone contraception and bone density: Results from a prospective study. Epidemiology. 2002;13(5):581-7.
4. Kaunitz AM, Miller PD, Rice VM, Ross D, McClung MR. Bone mineral density in women aged 25-35 years receiving depot medroxyprogesterone acetate: recovery following discontinuation. Contraception. 2006;74(2):90-99.
5. Schnatz PF, Barker KG, Marakovits KA, O’Sullivan DM. Effects of age at first pregnancy and breast-feeding on the development of postmenopausal osteoporosis. Menopause. 2010;17(6):1161-1166.
6. World Health Organization WHO Epidemiological Record No. 35: WHO statement on hormonal contraception and bone health. Geneva Switzerland: World Health Organization. 2005;80(35):302-304.
7. Grimes DA, Schulz KF. Surrogate end points in clinical research: hazardous to your health. Obstet Gynecol. 2005;105(5 pt 1):1114-1118.
8. Grimes DA, Schulz KF, Raymond EG. Surrogate end points in women’s health research: science protoscience, and pseudoscience. Fertil Steril. 2010;93(6):1731-1734.
9. Shulman LP, Bateman LH, Creinin MD, et al. Surrogate markers, emboldened and boxed warnings, and an expanding culture of misinformation: evidence-based clinical science should guide FDA decision making about product labeling. Contraception. 2006;73(5):440-442.
10. Cundy T, Cornish J, Roberts H, Reid IR. Menopausal bone loss in long-term users of depot medroxyprogesterone acetate contraception. Am J Obstet Gynecol. 2002;186(5):978-983.
11. Orr-Walker BJ, Evans MC, Ames RW, Clearwater JM, Cundy T, Reid IR. The effect of past use of the injectable contraceptive depot medroxyprogesterone acetate on bone mineral density in normal post-menopausal women. Clin Endocrinol (Oxf). 1998;49(5):615-618.
12. Viola AS, Castro S, Marchi NM, Bahamondes MV, Viola CFM, Bahamondes L. Long-term assessment of forearm bone mineral density in postmenopausal former users of depot medroxyprogesterone acetate. Contraception. In press.
13. Vestergaard P, Rejnmark L, Mosekilde L. The effects of depot medroxyprogesterone acetate and intrauterine device use on fracture risk in Danish women. Contraception. 2008;78(6):459-464.
14. Meier C, Brauchli YB, Jick SS, Kraenzlin ME, Meier CR. Use of depot medroxyprogesterone acetate and fracture risk. J Clin Endocrin Metab. 2010;95(11):4909-4916.
15. Kaunitz AM, Harel Z, Bone H, et al. Retrospective cohort study of DMPA and fractures in reproductive age women. Paper presented at: Association of Reproductive Health Professional Annual Meeting; September 24, 2010; Atlanta, GA.
16. Mosher WD, Jones J. Use of contraception in the United States: 1982-2008. National Center for Health Statistics. Vital Health Stat 23. 2010;23(29):1-44.
17. Paschall S, Kaunitz AM. Depo-Provera and skeletal health: A survey of Florida obstetrics and gynecologist physicians. Contraception. 2008;78(5):370-376.
18. American College of Obstetricians and Gynecologists Committee on Gynecologic Practice. ACOG Committee Opinion No. 415: Depot medroxyprogesterone acetate and bone effects. Obstet Gynecol. 2008;112(3):727-730.
19. Cromer BA, Scholes D, Berenson A, Cundy T, Clark MK, Kaunitz AM; Society for Adolescent Medicine. Depot medroxyprogesterone acetate and bone mineral density in adolescent—the black box warning: A position paper of the Society for Adolescent Medicine. J Adolesc Health. 2006;39(2):296-301.
20. Black A; Ad Hoc DMPA Committee of the Society of Obstetricians and Gynaecologists of Canada. Canadian contraception consensus: update on depot medroxyprogesterone acetate (DMPA). J Obstet Gynaecol Can. 2006;28(4):305-313.
21. Centers for Disease Control and Prevention. US medical eligibility criteria contraceptive use 2010. MMWR. 2010;59 (RR04);1:6.
22. Kaunitz AM, Grimes DA. Removing the black box warning for depot medroxyprogesterone acetate [published online ahead of print February 21 2011]. Contraception. doi:10.1016/j.contraception.2011.01.009.
- Update on contraception
Rachel B. Rapkin, MD; Mitchell D. Creinin, MD (August 2011) - 10 (+1) practical, evidence-based recommendations for you to improve contraceptive care now
Colleen Krajewski, MD; Mark D. Walters, MD (August 2011) - Levonorgestrel or ulipristal: Is one a better emergency contraceptive than the other?
Robert L. Barbieri, MD (Editorial; March 2011) - IUD use in nulliparous and adolescent women
Jennefer A. Russo, MD; Mitchell D. Creinin, MD (Update on Contraception, August 2010)
The Food and Drug Administration (FDA) should remove the black-box warning regarding skeletal health that the agency inserted into labeling for the injectable contraceptive depot medroxyprogesterone acetate (DMPA; Depo-Provera) in 2004. The black box 1) indicates that use of DMPA for longer than 2 years may reduce peak bone mass and place a woman at increased risk of osteoporotic fracture and 2) suggests that ObGyns order bone-density assessment in women who use DMPA long-term.
This warning is not based on scientific evidence. By depriving women of long-term use of a safe, effective contraceptive, the black-box warning damages women’s health and the public health.
The scientific argument for removing the black box
Although DMPA does cause reduced ovarian estradiol production, with a decline in bone mineral density (BMD), complete recovery of BMD occurs approximately 1 or 2 years after discontinuation in adolescent girls1,2 and 3 years after discontinuation in adult women.3,4 BMD trends observed with the use of DMPA parallel those associated with another normal but hypoestrogenic state: breastfeeding. Although BMD declines noted in nursing mothers are similar to those associated with DMPA, breastfeeding one or more infants has not been reported to increase the risk of subsequent osteoporotic fractures.5 The FDA does not warn women against breastfeeding for fear of a theoretical negative impact on skeletal health.
BMD can help predict a risk of osteoporotic fracture in menopausal women, but it is not a valid surrogate endpoint for fracture in women of reproductive age.6 Basing clinical recommendations on invalid surrogate endpoints can cause harm.7-9 Clinical recommendations, including those from the FDA, should be based on sound studies that address important clinical outcomes.
No convincing evidence links the use of DMPA for contraception to fracture. Studies of menopausal women have not suggested that prior use of DMPA increased their risk of osteoporosis.10-12 Two recently published case-control studies, one using a Danish National Patient Registry13 and one based on the United Kingdom Family Practice Research database,14 have demonstrated that DMPA is associated with an elevated risk of fracture in reproductive-age women. However, a cohort analysis using the same British Family Practice database clarifies that the elevated fracture risk observed in women using DMPA occurred before initiation of injectable contraception and was not the result of DMPA use.15

A clearly harmful effect on women’s health
The black-box warning for DMPA has caused clinicians and women to reduce use of this effective method of contraception. The 2008 National Survey of Family Growth shows that the overall percentage of US women, 15 to 44 years of age, who now use DMPA has declined—from 3.3% in 2002 to 2.0% in 2006 to 2008.16
In fact, a survey of ObGyns in Florida found that 1) almost one half of respondents place a time limit on duration of DMPA and 2) two thirds of those respondents base that restriction on the black-box warning.17 Restricting the use of DMPA can lead to more unintended pregnancies and induced abortions.
In the Florida survey, two thirds of respondents order BMD assessment in women who use DMPA, with 58% of those physicians who do so attributing their decision to, again, the black-box warning. Indeed, more than 5% of respondents reported that they selectively prescribe bisphosphonates to women of reproductive age who use DMPA—an expensive practice that is not evidence-based and is potentially dangerous.
FDA guidance is far out of step
The black-box warning for DMPA is not in accord with the guidance provided major medical and public health organizations. The World Health Organization,6 the American College of Obstetricians and Gynecologists,18 the Society for Adolescent Medicine,19 and the Society of Obstetrics and Gynecology of Canada,20 have all stated that skeletal health concerns shouldn’t restrict the use of DMPA, including the duration of use.
In the US Medical Eligibility Criteria for contraceptive use, the Centers for Disease Control and Prevention declared that use of DMPA is Category 1 (no restriction on use) in women 18 to 45 years of age and Category 2 (advantages of the method generally outweigh theoretical or proven risks) in younger women.21 The FDA’s black-box warning is therefore inconsistent with the recommendation of its sister agency.
An opportunity to improve the health of women
The FDA appropriately takes action when a pharmaceutical company mislabels a product or promotes it in a way that isn’t based on sound evidence. Shouldn’t the FDA apply the same scientific standards to itself? Guidance included in DMPA’s black box is not evidence-based—and that reduces use of DMPA in US women and increases the potential for harm. Removing the black box would encourage use of this convenient, effective, and safe contraceptive22 and, in turn, improve the health of women and their families.
Again: We urge the FDA to promptly remove the black-box warning from labeling for DMPA.
Do you agree? We want to hear your opinion: Write to us in care of The Editors, at [email protected]!
Long-acting reversible contraceptives are safe, effective for most women, study shows
But most use other methods because of lack of knowledge and cost concerns
Long-acting reversible contraceptives (LARC) are safe and effective for almost all women of reproductive age, according to an ACOG practice bulletin published in the July issue of Obstetrics & Gynecology.
Eve Espey, MD, MPH, and Rameet H. Singh, MD, MPH, from ACOG in Washington, DC, and colleagues compiled the latest recommendations on LARC use. The recommendations identify candidates for LARCs and help obstetricians and gynecologists manage LARC-related clinical issues.
The investigators reported that the LARC methods currently available have few contraindications, and that most women are eligible to use them. Intrauterine devices (IUDs) are suitable for women with a previous ectopic pregnancy and for women immediately after abortion or miscarriage. Routine antibiotic prophylaxis is not recommended before IUD insertion.
Copper IUD inserted up to 5 days after unprotected intercourse is the most effective method of postcoital contraception. The copper IUD can be used and remains effective for up to 10 continuous years. The levonorgestrel IUD may be effective for up to 7 years.
IUD complications are rare and include expulsion, method failure, and perforation. Implants can be safely inserted at any time after childbirth in non-breastfeeding women, or after 4 weeks for breastfeeding mothers. All LARC methods are safe for nulliparous women and adolescents.
The use of IUDs increased from 1.3% to 5.5% from 2002 to 2006-2008. Despite the benefits of LARCs, the majority of women choose other birth control methods, probably due to lack of knowledge about LARCs and cost concerns.
“LARC methods are the best tool we have to fight against unintended pregnancies, which currently account for 49% of US pregnancies each year,” Espey said in a statement.
Copyright © 2011 HealthDay. All rights reserved.
Has the FDA’s warning about DMPA had a chilling effect? Does the black-box warning prevent you from prescribing DMPA? Or does it alter your approach to using it? How?
To tell us, click here
- Update on contraception
Rachel B. Rapkin, MD; Mitchell D. Creinin, MD (August 2011) - 10 (+1) practical, evidence-based recommendations for you to improve contraceptive care now
Colleen Krajewski, MD; Mark D. Walters, MD (August 2011) - Levonorgestrel or ulipristal: Is one a better emergency contraceptive than the other?
Robert L. Barbieri, MD (Editorial; March 2011) - IUD use in nulliparous and adolescent women
Jennefer A. Russo, MD; Mitchell D. Creinin, MD (Update on Contraception, August 2010)
The Food and Drug Administration (FDA) should remove the black-box warning regarding skeletal health that the agency inserted into labeling for the injectable contraceptive depot medroxyprogesterone acetate (DMPA; Depo-Provera) in 2004. The black box 1) indicates that use of DMPA for longer than 2 years may reduce peak bone mass and place a woman at increased risk of osteoporotic fracture and 2) suggests that ObGyns order bone-density assessment in women who use DMPA long-term.
This warning is not based on scientific evidence. By depriving women of long-term use of a safe, effective contraceptive, the black-box warning damages women’s health and the public health.
The scientific argument for removing the black box
Although DMPA does cause reduced ovarian estradiol production, with a decline in bone mineral density (BMD), complete recovery of BMD occurs approximately 1 or 2 years after discontinuation in adolescent girls1,2 and 3 years after discontinuation in adult women.3,4 BMD trends observed with the use of DMPA parallel those associated with another normal but hypoestrogenic state: breastfeeding. Although BMD declines noted in nursing mothers are similar to those associated with DMPA, breastfeeding one or more infants has not been reported to increase the risk of subsequent osteoporotic fractures.5 The FDA does not warn women against breastfeeding for fear of a theoretical negative impact on skeletal health.
BMD can help predict a risk of osteoporotic fracture in menopausal women, but it is not a valid surrogate endpoint for fracture in women of reproductive age.6 Basing clinical recommendations on invalid surrogate endpoints can cause harm.7-9 Clinical recommendations, including those from the FDA, should be based on sound studies that address important clinical outcomes.
No convincing evidence links the use of DMPA for contraception to fracture. Studies of menopausal women have not suggested that prior use of DMPA increased their risk of osteoporosis.10-12 Two recently published case-control studies, one using a Danish National Patient Registry13 and one based on the United Kingdom Family Practice Research database,14 have demonstrated that DMPA is associated with an elevated risk of fracture in reproductive-age women. However, a cohort analysis using the same British Family Practice database clarifies that the elevated fracture risk observed in women using DMPA occurred before initiation of injectable contraception and was not the result of DMPA use.15

A clearly harmful effect on women’s health
The black-box warning for DMPA has caused clinicians and women to reduce use of this effective method of contraception. The 2008 National Survey of Family Growth shows that the overall percentage of US women, 15 to 44 years of age, who now use DMPA has declined—from 3.3% in 2002 to 2.0% in 2006 to 2008.16
In fact, a survey of ObGyns in Florida found that 1) almost one half of respondents place a time limit on duration of DMPA and 2) two thirds of those respondents base that restriction on the black-box warning.17 Restricting the use of DMPA can lead to more unintended pregnancies and induced abortions.
In the Florida survey, two thirds of respondents order BMD assessment in women who use DMPA, with 58% of those physicians who do so attributing their decision to, again, the black-box warning. Indeed, more than 5% of respondents reported that they selectively prescribe bisphosphonates to women of reproductive age who use DMPA—an expensive practice that is not evidence-based and is potentially dangerous.
FDA guidance is far out of step
The black-box warning for DMPA is not in accord with the guidance provided major medical and public health organizations. The World Health Organization,6 the American College of Obstetricians and Gynecologists,18 the Society for Adolescent Medicine,19 and the Society of Obstetrics and Gynecology of Canada,20 have all stated that skeletal health concerns shouldn’t restrict the use of DMPA, including the duration of use.
In the US Medical Eligibility Criteria for contraceptive use, the Centers for Disease Control and Prevention declared that use of DMPA is Category 1 (no restriction on use) in women 18 to 45 years of age and Category 2 (advantages of the method generally outweigh theoretical or proven risks) in younger women.21 The FDA’s black-box warning is therefore inconsistent with the recommendation of its sister agency.
An opportunity to improve the health of women
The FDA appropriately takes action when a pharmaceutical company mislabels a product or promotes it in a way that isn’t based on sound evidence. Shouldn’t the FDA apply the same scientific standards to itself? Guidance included in DMPA’s black box is not evidence-based—and that reduces use of DMPA in US women and increases the potential for harm. Removing the black box would encourage use of this convenient, effective, and safe contraceptive22 and, in turn, improve the health of women and their families.
Again: We urge the FDA to promptly remove the black-box warning from labeling for DMPA.
Do you agree? We want to hear your opinion: Write to us in care of The Editors, at [email protected]!
Long-acting reversible contraceptives are safe, effective for most women, study shows
But most use other methods because of lack of knowledge and cost concerns
Long-acting reversible contraceptives (LARC) are safe and effective for almost all women of reproductive age, according to an ACOG practice bulletin published in the July issue of Obstetrics & Gynecology.
Eve Espey, MD, MPH, and Rameet H. Singh, MD, MPH, from ACOG in Washington, DC, and colleagues compiled the latest recommendations on LARC use. The recommendations identify candidates for LARCs and help obstetricians and gynecologists manage LARC-related clinical issues.
The investigators reported that the LARC methods currently available have few contraindications, and that most women are eligible to use them. Intrauterine devices (IUDs) are suitable for women with a previous ectopic pregnancy and for women immediately after abortion or miscarriage. Routine antibiotic prophylaxis is not recommended before IUD insertion.
Copper IUD inserted up to 5 days after unprotected intercourse is the most effective method of postcoital contraception. The copper IUD can be used and remains effective for up to 10 continuous years. The levonorgestrel IUD may be effective for up to 7 years.
IUD complications are rare and include expulsion, method failure, and perforation. Implants can be safely inserted at any time after childbirth in non-breastfeeding women, or after 4 weeks for breastfeeding mothers. All LARC methods are safe for nulliparous women and adolescents.
The use of IUDs increased from 1.3% to 5.5% from 2002 to 2006-2008. Despite the benefits of LARCs, the majority of women choose other birth control methods, probably due to lack of knowledge about LARCs and cost concerns.
“LARC methods are the best tool we have to fight against unintended pregnancies, which currently account for 49% of US pregnancies each year,” Espey said in a statement.
Copyright © 2011 HealthDay. All rights reserved.
Has the FDA’s warning about DMPA had a chilling effect? Does the black-box warning prevent you from prescribing DMPA? Or does it alter your approach to using it? How?
To tell us, click here
1. Scholes D, LaCroix AZ, Ichikawa LE, Barlow WE, Ott SM. Change in bone mineral density among adolescent women using and discontinuing depot medroxyprogesterone acetate contraception. Arch Pediatric Adolesc Med. 2005;159(2):139-144.
2. Harel Z, Johnson CC, Gold MA, et al. Recovery of bone mineral density in adolescents following the use of depot medroxyprogesterone acetate contraceptive injections. Contraception. 2010;81(4):281-291.
3. Scholes D, LaCroix AZ, Ichikawa LE, Barlow WE, Ott SM. Injectable hormone contraception and bone density: Results from a prospective study. Epidemiology. 2002;13(5):581-7.
4. Kaunitz AM, Miller PD, Rice VM, Ross D, McClung MR. Bone mineral density in women aged 25-35 years receiving depot medroxyprogesterone acetate: recovery following discontinuation. Contraception. 2006;74(2):90-99.
5. Schnatz PF, Barker KG, Marakovits KA, O’Sullivan DM. Effects of age at first pregnancy and breast-feeding on the development of postmenopausal osteoporosis. Menopause. 2010;17(6):1161-1166.
6. World Health Organization WHO Epidemiological Record No. 35: WHO statement on hormonal contraception and bone health. Geneva Switzerland: World Health Organization. 2005;80(35):302-304.
7. Grimes DA, Schulz KF. Surrogate end points in clinical research: hazardous to your health. Obstet Gynecol. 2005;105(5 pt 1):1114-1118.
8. Grimes DA, Schulz KF, Raymond EG. Surrogate end points in women’s health research: science protoscience, and pseudoscience. Fertil Steril. 2010;93(6):1731-1734.
9. Shulman LP, Bateman LH, Creinin MD, et al. Surrogate markers, emboldened and boxed warnings, and an expanding culture of misinformation: evidence-based clinical science should guide FDA decision making about product labeling. Contraception. 2006;73(5):440-442.
10. Cundy T, Cornish J, Roberts H, Reid IR. Menopausal bone loss in long-term users of depot medroxyprogesterone acetate contraception. Am J Obstet Gynecol. 2002;186(5):978-983.
11. Orr-Walker BJ, Evans MC, Ames RW, Clearwater JM, Cundy T, Reid IR. The effect of past use of the injectable contraceptive depot medroxyprogesterone acetate on bone mineral density in normal post-menopausal women. Clin Endocrinol (Oxf). 1998;49(5):615-618.
12. Viola AS, Castro S, Marchi NM, Bahamondes MV, Viola CFM, Bahamondes L. Long-term assessment of forearm bone mineral density in postmenopausal former users of depot medroxyprogesterone acetate. Contraception. In press.
13. Vestergaard P, Rejnmark L, Mosekilde L. The effects of depot medroxyprogesterone acetate and intrauterine device use on fracture risk in Danish women. Contraception. 2008;78(6):459-464.
14. Meier C, Brauchli YB, Jick SS, Kraenzlin ME, Meier CR. Use of depot medroxyprogesterone acetate and fracture risk. J Clin Endocrin Metab. 2010;95(11):4909-4916.
15. Kaunitz AM, Harel Z, Bone H, et al. Retrospective cohort study of DMPA and fractures in reproductive age women. Paper presented at: Association of Reproductive Health Professional Annual Meeting; September 24, 2010; Atlanta, GA.
16. Mosher WD, Jones J. Use of contraception in the United States: 1982-2008. National Center for Health Statistics. Vital Health Stat 23. 2010;23(29):1-44.
17. Paschall S, Kaunitz AM. Depo-Provera and skeletal health: A survey of Florida obstetrics and gynecologist physicians. Contraception. 2008;78(5):370-376.
18. American College of Obstetricians and Gynecologists Committee on Gynecologic Practice. ACOG Committee Opinion No. 415: Depot medroxyprogesterone acetate and bone effects. Obstet Gynecol. 2008;112(3):727-730.
19. Cromer BA, Scholes D, Berenson A, Cundy T, Clark MK, Kaunitz AM; Society for Adolescent Medicine. Depot medroxyprogesterone acetate and bone mineral density in adolescent—the black box warning: A position paper of the Society for Adolescent Medicine. J Adolesc Health. 2006;39(2):296-301.
20. Black A; Ad Hoc DMPA Committee of the Society of Obstetricians and Gynaecologists of Canada. Canadian contraception consensus: update on depot medroxyprogesterone acetate (DMPA). J Obstet Gynaecol Can. 2006;28(4):305-313.
21. Centers for Disease Control and Prevention. US medical eligibility criteria contraceptive use 2010. MMWR. 2010;59 (RR04);1:6.
22. Kaunitz AM, Grimes DA. Removing the black box warning for depot medroxyprogesterone acetate [published online ahead of print February 21 2011]. Contraception. doi:10.1016/j.contraception.2011.01.009.
1. Scholes D, LaCroix AZ, Ichikawa LE, Barlow WE, Ott SM. Change in bone mineral density among adolescent women using and discontinuing depot medroxyprogesterone acetate contraception. Arch Pediatric Adolesc Med. 2005;159(2):139-144.
2. Harel Z, Johnson CC, Gold MA, et al. Recovery of bone mineral density in adolescents following the use of depot medroxyprogesterone acetate contraceptive injections. Contraception. 2010;81(4):281-291.
3. Scholes D, LaCroix AZ, Ichikawa LE, Barlow WE, Ott SM. Injectable hormone contraception and bone density: Results from a prospective study. Epidemiology. 2002;13(5):581-7.
4. Kaunitz AM, Miller PD, Rice VM, Ross D, McClung MR. Bone mineral density in women aged 25-35 years receiving depot medroxyprogesterone acetate: recovery following discontinuation. Contraception. 2006;74(2):90-99.
5. Schnatz PF, Barker KG, Marakovits KA, O’Sullivan DM. Effects of age at first pregnancy and breast-feeding on the development of postmenopausal osteoporosis. Menopause. 2010;17(6):1161-1166.
6. World Health Organization WHO Epidemiological Record No. 35: WHO statement on hormonal contraception and bone health. Geneva Switzerland: World Health Organization. 2005;80(35):302-304.
7. Grimes DA, Schulz KF. Surrogate end points in clinical research: hazardous to your health. Obstet Gynecol. 2005;105(5 pt 1):1114-1118.
8. Grimes DA, Schulz KF, Raymond EG. Surrogate end points in women’s health research: science protoscience, and pseudoscience. Fertil Steril. 2010;93(6):1731-1734.
9. Shulman LP, Bateman LH, Creinin MD, et al. Surrogate markers, emboldened and boxed warnings, and an expanding culture of misinformation: evidence-based clinical science should guide FDA decision making about product labeling. Contraception. 2006;73(5):440-442.
10. Cundy T, Cornish J, Roberts H, Reid IR. Menopausal bone loss in long-term users of depot medroxyprogesterone acetate contraception. Am J Obstet Gynecol. 2002;186(5):978-983.
11. Orr-Walker BJ, Evans MC, Ames RW, Clearwater JM, Cundy T, Reid IR. The effect of past use of the injectable contraceptive depot medroxyprogesterone acetate on bone mineral density in normal post-menopausal women. Clin Endocrinol (Oxf). 1998;49(5):615-618.
12. Viola AS, Castro S, Marchi NM, Bahamondes MV, Viola CFM, Bahamondes L. Long-term assessment of forearm bone mineral density in postmenopausal former users of depot medroxyprogesterone acetate. Contraception. In press.
13. Vestergaard P, Rejnmark L, Mosekilde L. The effects of depot medroxyprogesterone acetate and intrauterine device use on fracture risk in Danish women. Contraception. 2008;78(6):459-464.
14. Meier C, Brauchli YB, Jick SS, Kraenzlin ME, Meier CR. Use of depot medroxyprogesterone acetate and fracture risk. J Clin Endocrin Metab. 2010;95(11):4909-4916.
15. Kaunitz AM, Harel Z, Bone H, et al. Retrospective cohort study of DMPA and fractures in reproductive age women. Paper presented at: Association of Reproductive Health Professional Annual Meeting; September 24, 2010; Atlanta, GA.
16. Mosher WD, Jones J. Use of contraception in the United States: 1982-2008. National Center for Health Statistics. Vital Health Stat 23. 2010;23(29):1-44.
17. Paschall S, Kaunitz AM. Depo-Provera and skeletal health: A survey of Florida obstetrics and gynecologist physicians. Contraception. 2008;78(5):370-376.
18. American College of Obstetricians and Gynecologists Committee on Gynecologic Practice. ACOG Committee Opinion No. 415: Depot medroxyprogesterone acetate and bone effects. Obstet Gynecol. 2008;112(3):727-730.
19. Cromer BA, Scholes D, Berenson A, Cundy T, Clark MK, Kaunitz AM; Society for Adolescent Medicine. Depot medroxyprogesterone acetate and bone mineral density in adolescent—the black box warning: A position paper of the Society for Adolescent Medicine. J Adolesc Health. 2006;39(2):296-301.
20. Black A; Ad Hoc DMPA Committee of the Society of Obstetricians and Gynaecologists of Canada. Canadian contraception consensus: update on depot medroxyprogesterone acetate (DMPA). J Obstet Gynaecol Can. 2006;28(4):305-313.
21. Centers for Disease Control and Prevention. US medical eligibility criteria contraceptive use 2010. MMWR. 2010;59 (RR04);1:6.
22. Kaunitz AM, Grimes DA. Removing the black box warning for depot medroxyprogesterone acetate [published online ahead of print February 21 2011]. Contraception. doi:10.1016/j.contraception.2011.01.009.
IN THIS ARTICLE











