User login
Mogamulizumab achieves objective responses in relapsed/refractory adult T-cell leukemia-lymphoma
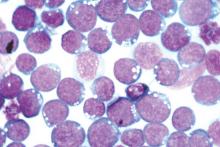
CHICAGO – The anti-CCR4 monoclonal antibody mogamulizumab was superior to other investigator-selected therapies for the treatment of patients with relapsed/refractory adult T-cell leukemia-lymphoma (ATL), based on results from 71 patients in a prospective, multicenter, randomized study reported at the annual meeting of the American Society of Clinical Oncology.
Commonly used cytotoxic regimens provided limited therapeutic benefit for these patients, but mogamulizumab resulted in an objective response rate that supports its therapeutic potential in this setting, reported Dr. Adrienne Alise Phillips of New York Presbyterian/Weill Cornell Medical College, New York.
A malignancy of T-cells infected with HTLV-1, ATL has a poor prognosis with a median overall survival of less than 3 months in patients with relapsed/refractory disease. CCR4 is expressed in over 90% of ATL patients, and mogamulizumab is approved in Japan for ATL as well as for peripheral T-cell lymphoma and cutaneous T-cell lymphoma.
The 71 patients in the study were from the United States, the European Union and Latin America. The study is the largest randomized clinical trial of relapsed/refractory adult T-cell leukemia-lymphoma thus far conducted. The patients were randomized in 2:1 fashion 47:24 patients) to mogamulizumab, 1.0 mg/kg, given weekly for the first 4-week cycle and then biweekly, or to one of three investigator choice regimens [gemcitabine and oxaliplatin, DHAP (dexamethasone, high-dose cytarabine, and cisplatin), or pralatrexate]. Patients who were in the investigator-choice arm and whose disease progressed were permitted to cross over to mogamulizumab.
The primary endpoint was objective response rate based on modified Tsukasaki criteria and assessed by the treating investigator and in blinded fashion by independent review.
The objective response rate in the mogamulizumab-treated group was 23.4% (11 of 47) by independent review and 34% (16 of 47) by the treating investigator. In the investigator choice group, the overall response rate was 2 of 24 by independent review and 0 of 24 by the treating investigator.
The confirmed objective response rate after 1 month in the mogamulizumab-treated group was 10.6% by independent review and 14.9% by the treating investigator; there were no confirmed responses in the investigator-choice arm. Of 18 patients who crossed over to mogamulizumab, 3 responded. The median duration of response for mogamulizumab was 5 months; one patient had a complete response that lasted over 9 months and the survival data are not yet mature.
Mogamulizumab had few drug-related adverse events, primarily infusion reactions (46.8%), rash/drug eruption (25.5%) and infections (14.9%).
Dr. Phillips disclosed ties to Celgene, Genentech, and Takeda, as well as research funding from Kyowa Hakko Kirin, the sponsor of the study.
On Twitter @maryjodales

|
| Mary Jo Dales/Frontline Medical News Dr. Sonali M. Smith |
Mogamulizumab was superior to investigator’s choice therapy in the largest prospective randomized trial of this very rare disease. Approximately one-third of patients responded, while the response to investigator’s choice therapies was zero. The potential impact of mogamulizumab on T-cell regulation is intriguing. Could it have applications in other T-cell non-Hodgkin’s lymphomas and cutaneous T-cell lymphomas?
Dr. Sonali M. Smith is with the University of Chicago and was the invited discussant of the study.

|
| Mary Jo Dales/Frontline Medical News Dr. Sonali M. Smith |
Mogamulizumab was superior to investigator’s choice therapy in the largest prospective randomized trial of this very rare disease. Approximately one-third of patients responded, while the response to investigator’s choice therapies was zero. The potential impact of mogamulizumab on T-cell regulation is intriguing. Could it have applications in other T-cell non-Hodgkin’s lymphomas and cutaneous T-cell lymphomas?
Dr. Sonali M. Smith is with the University of Chicago and was the invited discussant of the study.

|
| Mary Jo Dales/Frontline Medical News Dr. Sonali M. Smith |
Mogamulizumab was superior to investigator’s choice therapy in the largest prospective randomized trial of this very rare disease. Approximately one-third of patients responded, while the response to investigator’s choice therapies was zero. The potential impact of mogamulizumab on T-cell regulation is intriguing. Could it have applications in other T-cell non-Hodgkin’s lymphomas and cutaneous T-cell lymphomas?
Dr. Sonali M. Smith is with the University of Chicago and was the invited discussant of the study.
CHICAGO – The anti-CCR4 monoclonal antibody mogamulizumab was superior to other investigator-selected therapies for the treatment of patients with relapsed/refractory adult T-cell leukemia-lymphoma (ATL), based on results from 71 patients in a prospective, multicenter, randomized study reported at the annual meeting of the American Society of Clinical Oncology.
Commonly used cytotoxic regimens provided limited therapeutic benefit for these patients, but mogamulizumab resulted in an objective response rate that supports its therapeutic potential in this setting, reported Dr. Adrienne Alise Phillips of New York Presbyterian/Weill Cornell Medical College, New York.
A malignancy of T-cells infected with HTLV-1, ATL has a poor prognosis with a median overall survival of less than 3 months in patients with relapsed/refractory disease. CCR4 is expressed in over 90% of ATL patients, and mogamulizumab is approved in Japan for ATL as well as for peripheral T-cell lymphoma and cutaneous T-cell lymphoma.
The 71 patients in the study were from the United States, the European Union and Latin America. The study is the largest randomized clinical trial of relapsed/refractory adult T-cell leukemia-lymphoma thus far conducted. The patients were randomized in 2:1 fashion 47:24 patients) to mogamulizumab, 1.0 mg/kg, given weekly for the first 4-week cycle and then biweekly, or to one of three investigator choice regimens [gemcitabine and oxaliplatin, DHAP (dexamethasone, high-dose cytarabine, and cisplatin), or pralatrexate]. Patients who were in the investigator-choice arm and whose disease progressed were permitted to cross over to mogamulizumab.
The primary endpoint was objective response rate based on modified Tsukasaki criteria and assessed by the treating investigator and in blinded fashion by independent review.
The objective response rate in the mogamulizumab-treated group was 23.4% (11 of 47) by independent review and 34% (16 of 47) by the treating investigator. In the investigator choice group, the overall response rate was 2 of 24 by independent review and 0 of 24 by the treating investigator.
The confirmed objective response rate after 1 month in the mogamulizumab-treated group was 10.6% by independent review and 14.9% by the treating investigator; there were no confirmed responses in the investigator-choice arm. Of 18 patients who crossed over to mogamulizumab, 3 responded. The median duration of response for mogamulizumab was 5 months; one patient had a complete response that lasted over 9 months and the survival data are not yet mature.
Mogamulizumab had few drug-related adverse events, primarily infusion reactions (46.8%), rash/drug eruption (25.5%) and infections (14.9%).
Dr. Phillips disclosed ties to Celgene, Genentech, and Takeda, as well as research funding from Kyowa Hakko Kirin, the sponsor of the study.
On Twitter @maryjodales
CHICAGO – The anti-CCR4 monoclonal antibody mogamulizumab was superior to other investigator-selected therapies for the treatment of patients with relapsed/refractory adult T-cell leukemia-lymphoma (ATL), based on results from 71 patients in a prospective, multicenter, randomized study reported at the annual meeting of the American Society of Clinical Oncology.
Commonly used cytotoxic regimens provided limited therapeutic benefit for these patients, but mogamulizumab resulted in an objective response rate that supports its therapeutic potential in this setting, reported Dr. Adrienne Alise Phillips of New York Presbyterian/Weill Cornell Medical College, New York.
A malignancy of T-cells infected with HTLV-1, ATL has a poor prognosis with a median overall survival of less than 3 months in patients with relapsed/refractory disease. CCR4 is expressed in over 90% of ATL patients, and mogamulizumab is approved in Japan for ATL as well as for peripheral T-cell lymphoma and cutaneous T-cell lymphoma.
The 71 patients in the study were from the United States, the European Union and Latin America. The study is the largest randomized clinical trial of relapsed/refractory adult T-cell leukemia-lymphoma thus far conducted. The patients were randomized in 2:1 fashion 47:24 patients) to mogamulizumab, 1.0 mg/kg, given weekly for the first 4-week cycle and then biweekly, or to one of three investigator choice regimens [gemcitabine and oxaliplatin, DHAP (dexamethasone, high-dose cytarabine, and cisplatin), or pralatrexate]. Patients who were in the investigator-choice arm and whose disease progressed were permitted to cross over to mogamulizumab.
The primary endpoint was objective response rate based on modified Tsukasaki criteria and assessed by the treating investigator and in blinded fashion by independent review.
The objective response rate in the mogamulizumab-treated group was 23.4% (11 of 47) by independent review and 34% (16 of 47) by the treating investigator. In the investigator choice group, the overall response rate was 2 of 24 by independent review and 0 of 24 by the treating investigator.
The confirmed objective response rate after 1 month in the mogamulizumab-treated group was 10.6% by independent review and 14.9% by the treating investigator; there were no confirmed responses in the investigator-choice arm. Of 18 patients who crossed over to mogamulizumab, 3 responded. The median duration of response for mogamulizumab was 5 months; one patient had a complete response that lasted over 9 months and the survival data are not yet mature.
Mogamulizumab had few drug-related adverse events, primarily infusion reactions (46.8%), rash/drug eruption (25.5%) and infections (14.9%).
Dr. Phillips disclosed ties to Celgene, Genentech, and Takeda, as well as research funding from Kyowa Hakko Kirin, the sponsor of the study.
On Twitter @maryjodales
AT THE 2016 ASCO ANNUAL MEETING
Key clinical point: The anti-CCR4 monoclonal antibody mogamulizumab was superior to other investigator-selected therapies for the treatment of patients with relapsed/refractory adult T-cell leukemia-lymphoma.
Major finding: The confirmed objective response rate after 1 month in the mogamulizumab-treated group was 10.6% by independent review and 14.9% by the treating investigator; there were no confirmed responses in the investigator-choice arm.
Data source: Prospective, multicenter, randomized study of 71 patients from the United States, the European Union, and Latin America.
Disclosures: Dr. Phillips disclosed ties to Celgene, Genentech, and Takeda, as well as research funding from Kyowa Hakko Kirin, the sponsor of the study.
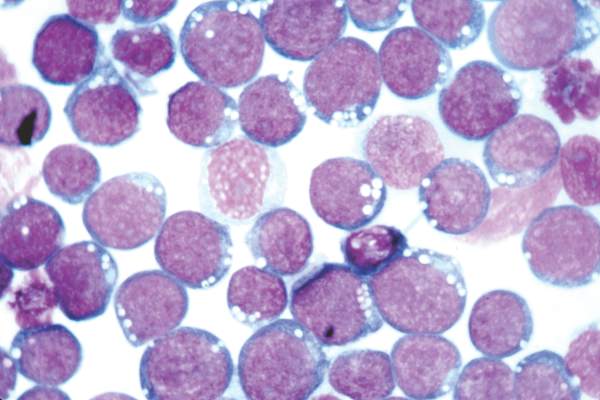
Alemtuzumab plus CHOP didn’t boost survival in elderly patients with peripheral T-cell lymphomas
CHICAGO – Adding the monoclonal anti-CD52 antibody alemtuzumab to CHOP (A-CHOP) increased response rates in elderly patients with peripheral T-cell lymphomas but did not improve their survival, based on the final results from 116 patients treated in the international ACT-2 phase III trial.
Complete responses were seen in 43% of 58 patients given CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisolone) and in 60% of 58 patients given A-CHOP in the trial. However, trial participants did not significantly differ in event-free survival and progression-free survival at 3 years.
Further, overall survival at 3 years was 38% for the patients given A-CHOP and 56% for the patients given CHOP. The poorer overall survival was mainly the result of treatment-related toxicity, Dr. Lorenz H. Trümper reported at the annual meeting of the American Society of Clinical Oncology.
The estimated 3-year disease-free survival is 25% for elderly patients with peripheral T-cell lymphomas. Previous phase II trials had indicated that alemtuzumab was active in primary and relapsed T-cell lymphoma, prompting the study of adjuvant alemtuzumab in combination with dose-dense CHOP-14 in patients with previously untreated peripheral T-cell lymphoma, he said.
Although the treatment protocol demanded stringent monitoring for cytomegalovirus and Epstein-Barr virus and anti-infective prophylaxis, there were more grade 3 or higher infections in the A-CHOP group (40%) than the CHOP group (21%). The higher infection rates were attributed to higher rates of grade 3/4 hematotoxicity in patients given A-CHOP. Grade 4 leukocytopenia was seen in 70% with A-CHOP and 54% with CHOP; grade 3/4 thrombocytopenia was seen in 19% given A-CHOP and 13% given CHOP, according to Dr. Trümper of the University of Göttingen, Germany.
For the study, 116 patients from 52 centers were randomized to receive either six cycles of CHOP or A-CHOP given at 14-day intervals with granulocyte-colony stimulating factor (G-CSF) support. Initially, patients received a total of 360 mg of alemtuzumab (90 mg given at each of the first four cycles of CHOP). After patient 39 was enrolled, the dose was reduced to 120 mg (30 mg given at each of the first four cycles of CHOP). Median patient age was 69 years, and 58% of the trial participants were men.
Treatment was completed as planned in 79% of the CHOP patients and in 57% of the A-CHOP patients.
The study was sponsored by the University of Göttingen. Dr. Trümper is a consultant or adviser to Hexal and Janssen-Ortho, and receives research funding from Genzyme, the maker of alemtuzumab (Lemtrada), as well as other drug companies.
On Twitter @maryjodales
CHICAGO – Adding the monoclonal anti-CD52 antibody alemtuzumab to CHOP (A-CHOP) increased response rates in elderly patients with peripheral T-cell lymphomas but did not improve their survival, based on the final results from 116 patients treated in the international ACT-2 phase III trial.
Complete responses were seen in 43% of 58 patients given CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisolone) and in 60% of 58 patients given A-CHOP in the trial. However, trial participants did not significantly differ in event-free survival and progression-free survival at 3 years.
Further, overall survival at 3 years was 38% for the patients given A-CHOP and 56% for the patients given CHOP. The poorer overall survival was mainly the result of treatment-related toxicity, Dr. Lorenz H. Trümper reported at the annual meeting of the American Society of Clinical Oncology.
The estimated 3-year disease-free survival is 25% for elderly patients with peripheral T-cell lymphomas. Previous phase II trials had indicated that alemtuzumab was active in primary and relapsed T-cell lymphoma, prompting the study of adjuvant alemtuzumab in combination with dose-dense CHOP-14 in patients with previously untreated peripheral T-cell lymphoma, he said.
Although the treatment protocol demanded stringent monitoring for cytomegalovirus and Epstein-Barr virus and anti-infective prophylaxis, there were more grade 3 or higher infections in the A-CHOP group (40%) than the CHOP group (21%). The higher infection rates were attributed to higher rates of grade 3/4 hematotoxicity in patients given A-CHOP. Grade 4 leukocytopenia was seen in 70% with A-CHOP and 54% with CHOP; grade 3/4 thrombocytopenia was seen in 19% given A-CHOP and 13% given CHOP, according to Dr. Trümper of the University of Göttingen, Germany.
For the study, 116 patients from 52 centers were randomized to receive either six cycles of CHOP or A-CHOP given at 14-day intervals with granulocyte-colony stimulating factor (G-CSF) support. Initially, patients received a total of 360 mg of alemtuzumab (90 mg given at each of the first four cycles of CHOP). After patient 39 was enrolled, the dose was reduced to 120 mg (30 mg given at each of the first four cycles of CHOP). Median patient age was 69 years, and 58% of the trial participants were men.
Treatment was completed as planned in 79% of the CHOP patients and in 57% of the A-CHOP patients.
The study was sponsored by the University of Göttingen. Dr. Trümper is a consultant or adviser to Hexal and Janssen-Ortho, and receives research funding from Genzyme, the maker of alemtuzumab (Lemtrada), as well as other drug companies.
On Twitter @maryjodales
CHICAGO – Adding the monoclonal anti-CD52 antibody alemtuzumab to CHOP (A-CHOP) increased response rates in elderly patients with peripheral T-cell lymphomas but did not improve their survival, based on the final results from 116 patients treated in the international ACT-2 phase III trial.
Complete responses were seen in 43% of 58 patients given CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisolone) and in 60% of 58 patients given A-CHOP in the trial. However, trial participants did not significantly differ in event-free survival and progression-free survival at 3 years.
Further, overall survival at 3 years was 38% for the patients given A-CHOP and 56% for the patients given CHOP. The poorer overall survival was mainly the result of treatment-related toxicity, Dr. Lorenz H. Trümper reported at the annual meeting of the American Society of Clinical Oncology.
The estimated 3-year disease-free survival is 25% for elderly patients with peripheral T-cell lymphomas. Previous phase II trials had indicated that alemtuzumab was active in primary and relapsed T-cell lymphoma, prompting the study of adjuvant alemtuzumab in combination with dose-dense CHOP-14 in patients with previously untreated peripheral T-cell lymphoma, he said.
Although the treatment protocol demanded stringent monitoring for cytomegalovirus and Epstein-Barr virus and anti-infective prophylaxis, there were more grade 3 or higher infections in the A-CHOP group (40%) than the CHOP group (21%). The higher infection rates were attributed to higher rates of grade 3/4 hematotoxicity in patients given A-CHOP. Grade 4 leukocytopenia was seen in 70% with A-CHOP and 54% with CHOP; grade 3/4 thrombocytopenia was seen in 19% given A-CHOP and 13% given CHOP, according to Dr. Trümper of the University of Göttingen, Germany.
For the study, 116 patients from 52 centers were randomized to receive either six cycles of CHOP or A-CHOP given at 14-day intervals with granulocyte-colony stimulating factor (G-CSF) support. Initially, patients received a total of 360 mg of alemtuzumab (90 mg given at each of the first four cycles of CHOP). After patient 39 was enrolled, the dose was reduced to 120 mg (30 mg given at each of the first four cycles of CHOP). Median patient age was 69 years, and 58% of the trial participants were men.
Treatment was completed as planned in 79% of the CHOP patients and in 57% of the A-CHOP patients.
The study was sponsored by the University of Göttingen. Dr. Trümper is a consultant or adviser to Hexal and Janssen-Ortho, and receives research funding from Genzyme, the maker of alemtuzumab (Lemtrada), as well as other drug companies.
On Twitter @maryjodales
AT ASCO 16
Key clinical point: Adding the monoclonal anti-CD52 antibody alemtuzumab to CHOP (A-CHOP) increased response rates in elderly patients with peripheral T-cell lymphomas but did not improve their survival.
Major finding: Overall survival at 3 years was 38% for the patients given A-CHOP and 56% for the patients given CHOP.
Data source: Results from 116 patients treated in the international ACT-2 phase III trial.
Disclosures: The study was sponsored by the University of Göttingen, Germany. Dr. Trümper is a consultant or adviser to Hexal and Janssen-Ortho, and receives research funding from Genzyme, the maker of alemtuzumab, as well as other drug companies.
FDA grants accelerated approval to nivolumab for Hodgkin lymphoma
The Food and Drug Administration has granted accelerated approval to nivolumab for the treatment of patients with classical Hodgkin lymphoma (cHL) that has relapsed or progressed after autologous hematopoietic stem cell transplantation (HSCT) and posttransplantation brentuximab vedotin.
Approval was based on a 65% objective response rate in 95 patients treated with nivolumab following autologous HSCT and posttransplantation brentuximab vedotin. All patients in the single-arm, multicenter trial had relapsed or refractory cHL and were enrolled regardless of PD-L1 expression status. Patients received a median of 17 doses of nivolumab, the FDA said in a written statement.
The median time to response was 2.1 months (range, 0.7-5.7 months). The estimated median duration of response was 8.7 months.
The FDA also issued a warning for complications of allogeneic HSCT after nivolumab, reporting that transplant-related deaths have occurred. Health care professionals should follow patients closely for early evidence of transplant-related complications, such as hyperacute graft-versus-host disease (GVHD), severe acute GVHD, steroid-requiring febrile syndrome, hepatic veno-occlusive disease, and other immune-mediated adverse reactions, they said.
The most common adverse reactions in a second single-arm study used to evaluate safety (n = 263) were upper respiratory tract infection, cough, pyrexia, and diarrhea. Other immune-mediated adverse reactions, occurring in 1%-5% of patients, included rash, pneumonitis, hepatitis, hyperthyroidism, and colitis. The most common serious adverse reactions, which were reported in 1%-3% of patients, were pneumonia, pleural effusion, pneumonitis, pyrexia, infusion-related reaction, and rash.
Nivolumab is marketed as Opdivo by Bristol-Myers Squibb and has been previously approved to treat advanced renal cell carcinoma, lung cancer, and melanoma.
On Twitter @NikolaidesLaura
The Food and Drug Administration has granted accelerated approval to nivolumab for the treatment of patients with classical Hodgkin lymphoma (cHL) that has relapsed or progressed after autologous hematopoietic stem cell transplantation (HSCT) and posttransplantation brentuximab vedotin.
Approval was based on a 65% objective response rate in 95 patients treated with nivolumab following autologous HSCT and posttransplantation brentuximab vedotin. All patients in the single-arm, multicenter trial had relapsed or refractory cHL and were enrolled regardless of PD-L1 expression status. Patients received a median of 17 doses of nivolumab, the FDA said in a written statement.
The median time to response was 2.1 months (range, 0.7-5.7 months). The estimated median duration of response was 8.7 months.
The FDA also issued a warning for complications of allogeneic HSCT after nivolumab, reporting that transplant-related deaths have occurred. Health care professionals should follow patients closely for early evidence of transplant-related complications, such as hyperacute graft-versus-host disease (GVHD), severe acute GVHD, steroid-requiring febrile syndrome, hepatic veno-occlusive disease, and other immune-mediated adverse reactions, they said.
The most common adverse reactions in a second single-arm study used to evaluate safety (n = 263) were upper respiratory tract infection, cough, pyrexia, and diarrhea. Other immune-mediated adverse reactions, occurring in 1%-5% of patients, included rash, pneumonitis, hepatitis, hyperthyroidism, and colitis. The most common serious adverse reactions, which were reported in 1%-3% of patients, were pneumonia, pleural effusion, pneumonitis, pyrexia, infusion-related reaction, and rash.
Nivolumab is marketed as Opdivo by Bristol-Myers Squibb and has been previously approved to treat advanced renal cell carcinoma, lung cancer, and melanoma.
On Twitter @NikolaidesLaura
The Food and Drug Administration has granted accelerated approval to nivolumab for the treatment of patients with classical Hodgkin lymphoma (cHL) that has relapsed or progressed after autologous hematopoietic stem cell transplantation (HSCT) and posttransplantation brentuximab vedotin.
Approval was based on a 65% objective response rate in 95 patients treated with nivolumab following autologous HSCT and posttransplantation brentuximab vedotin. All patients in the single-arm, multicenter trial had relapsed or refractory cHL and were enrolled regardless of PD-L1 expression status. Patients received a median of 17 doses of nivolumab, the FDA said in a written statement.
The median time to response was 2.1 months (range, 0.7-5.7 months). The estimated median duration of response was 8.7 months.
The FDA also issued a warning for complications of allogeneic HSCT after nivolumab, reporting that transplant-related deaths have occurred. Health care professionals should follow patients closely for early evidence of transplant-related complications, such as hyperacute graft-versus-host disease (GVHD), severe acute GVHD, steroid-requiring febrile syndrome, hepatic veno-occlusive disease, and other immune-mediated adverse reactions, they said.
The most common adverse reactions in a second single-arm study used to evaluate safety (n = 263) were upper respiratory tract infection, cough, pyrexia, and diarrhea. Other immune-mediated adverse reactions, occurring in 1%-5% of patients, included rash, pneumonitis, hepatitis, hyperthyroidism, and colitis. The most common serious adverse reactions, which were reported in 1%-3% of patients, were pneumonia, pleural effusion, pneumonitis, pyrexia, infusion-related reaction, and rash.
Nivolumab is marketed as Opdivo by Bristol-Myers Squibb and has been previously approved to treat advanced renal cell carcinoma, lung cancer, and melanoma.
On Twitter @NikolaidesLaura
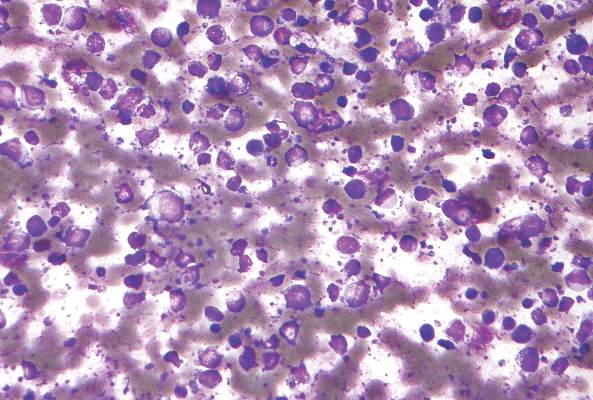
Epstein-Barr virus DNA in plasma reliably detects EBV-positive lymphoproliferative disorders
Detection of Epstein-Barr virus DNA in plasma reliably signaled “a broad range” of EBV+ diseases, according to investigators.
In contrast, the presence of EBV DNA in peripheral blood mononuclear cells did not reliably predict EBV diseases, said Dr. Jennifer A. Kanakry and her associates at Johns Hopkins University, Baltimore. Patients without EBV diseases can have EBV DNA in their PBMCs, particularly if they are immunocompromised, the researchers observed.
Latent EBV infection is associated with lymphomas, lymphoproliferative disorders, hemophagocytic lymphohistiocytosis, solid tumors, and other diseases. To characterize the relationship between these diseases and EBV DNA, the researchers studied viral quantitative real-time polymerase chain reaction assays of plasma and PBMCs from 2,146 patients tested at Johns Hopkins over 5 years. Patients were usually immunocompromised and hospitalized, the investigators noted (Blood 2016;127:2007-17).
A total of 535 patients (25%) had EBV detected in plasma or PBMCs. Notably, 69% of patients who did not have EBV diseases had EBV in PBMCs, but not in plasma. Among 105 patients with active systemic EBV+ diseases, 99% had EBV DNA in plasma, but only 54% had EBV in PBMCs. Furthermore, the number of copies of EBV DNA distinguished untreated EBV+ lymphoma, remitted EBV+ lymphoma, and EBV- lymphoma, and also distinguished untreated, EBV+ post-transplantation lymphoproliferative disorder (PTLD), EBV+ PTLD in remission, and EBV– PTLD.
“Cell-free (plasma) EBV DNA performs better than cellular EBV DNA as a marker of a broad range of EBV+ diseases,” the investigators concluded. “Within a largely immunocompromised and hospitalized cohort, detection of EBV DNA in plasma is uncommon in the absence of EBV+ disease.”
The National Cancer Institute, National Institutes of Health, and Center for AIDS Research funded the study. The researchers had no disclosures.
Detection of Epstein-Barr virus DNA in plasma reliably signaled “a broad range” of EBV+ diseases, according to investigators.
In contrast, the presence of EBV DNA in peripheral blood mononuclear cells did not reliably predict EBV diseases, said Dr. Jennifer A. Kanakry and her associates at Johns Hopkins University, Baltimore. Patients without EBV diseases can have EBV DNA in their PBMCs, particularly if they are immunocompromised, the researchers observed.
Latent EBV infection is associated with lymphomas, lymphoproliferative disorders, hemophagocytic lymphohistiocytosis, solid tumors, and other diseases. To characterize the relationship between these diseases and EBV DNA, the researchers studied viral quantitative real-time polymerase chain reaction assays of plasma and PBMCs from 2,146 patients tested at Johns Hopkins over 5 years. Patients were usually immunocompromised and hospitalized, the investigators noted (Blood 2016;127:2007-17).
A total of 535 patients (25%) had EBV detected in plasma or PBMCs. Notably, 69% of patients who did not have EBV diseases had EBV in PBMCs, but not in plasma. Among 105 patients with active systemic EBV+ diseases, 99% had EBV DNA in plasma, but only 54% had EBV in PBMCs. Furthermore, the number of copies of EBV DNA distinguished untreated EBV+ lymphoma, remitted EBV+ lymphoma, and EBV- lymphoma, and also distinguished untreated, EBV+ post-transplantation lymphoproliferative disorder (PTLD), EBV+ PTLD in remission, and EBV– PTLD.
“Cell-free (plasma) EBV DNA performs better than cellular EBV DNA as a marker of a broad range of EBV+ diseases,” the investigators concluded. “Within a largely immunocompromised and hospitalized cohort, detection of EBV DNA in plasma is uncommon in the absence of EBV+ disease.”
The National Cancer Institute, National Institutes of Health, and Center for AIDS Research funded the study. The researchers had no disclosures.
Detection of Epstein-Barr virus DNA in plasma reliably signaled “a broad range” of EBV+ diseases, according to investigators.
In contrast, the presence of EBV DNA in peripheral blood mononuclear cells did not reliably predict EBV diseases, said Dr. Jennifer A. Kanakry and her associates at Johns Hopkins University, Baltimore. Patients without EBV diseases can have EBV DNA in their PBMCs, particularly if they are immunocompromised, the researchers observed.
Latent EBV infection is associated with lymphomas, lymphoproliferative disorders, hemophagocytic lymphohistiocytosis, solid tumors, and other diseases. To characterize the relationship between these diseases and EBV DNA, the researchers studied viral quantitative real-time polymerase chain reaction assays of plasma and PBMCs from 2,146 patients tested at Johns Hopkins over 5 years. Patients were usually immunocompromised and hospitalized, the investigators noted (Blood 2016;127:2007-17).
A total of 535 patients (25%) had EBV detected in plasma or PBMCs. Notably, 69% of patients who did not have EBV diseases had EBV in PBMCs, but not in plasma. Among 105 patients with active systemic EBV+ diseases, 99% had EBV DNA in plasma, but only 54% had EBV in PBMCs. Furthermore, the number of copies of EBV DNA distinguished untreated EBV+ lymphoma, remitted EBV+ lymphoma, and EBV- lymphoma, and also distinguished untreated, EBV+ post-transplantation lymphoproliferative disorder (PTLD), EBV+ PTLD in remission, and EBV– PTLD.
“Cell-free (plasma) EBV DNA performs better than cellular EBV DNA as a marker of a broad range of EBV+ diseases,” the investigators concluded. “Within a largely immunocompromised and hospitalized cohort, detection of EBV DNA in plasma is uncommon in the absence of EBV+ disease.”
The National Cancer Institute, National Institutes of Health, and Center for AIDS Research funded the study. The researchers had no disclosures.
FROM BLOOD
Key clinical point: Epstein-Barr virus DNA was a more specific indicator of EBV+ diseases when detected in plasma, as opposed to peripheral blood mononuclear cells.
Major finding: Among 105 patients with active systemic EBV+ diseases, 99% had EBV DNA in plasma, but 54% had EBV in PBMCs.
Data source: Viral quantitative real-time polymerase chain reaction assays of plasma and PBMCs from 2,146 patients.
Disclosures: The National Cancer Institute, National Institutes of Health, and Center for AIDS Research funded the study. The researchers had no disclosures.
Dose-escalated blinatumomab plus dexamethasone induced responses in relapsed/refractory DLBCL
Blinatumomab, administered in a stepwise, dose-escalated regimen with prophylactic dexamethasone, induced responses in patients with relapsed/refractory diffuse large B-cell lymphoma (DLBCL), according to a report published in Blood.
The phase II study included 25 patients who were refractory to their last treatment, had relapsed after autologous hematopoietic stem cell transplant (HSCT), or had relapsed disease and were ineligible for autologous HSCT.
In a previous phase I study that did not use a dose-escalated regimen, neurological adverse events were dose limiting at a maximum tolerated blinatumomab dose of 60 mcg/d. The current study used a 14-day stepwise dose escalation and corticosteroid premedication to minimize adverse events: blinatumomab was given at 9 mcg/d in the first week, 28 mcg/d in the second week, and 112 mcg/d thereafter.
Patients had received a median of three prior therapies. In total, 17 of 25 patients ended in cycle 1 (induction) because of disease progression, adverse events, or physician decision; 7 ended in cycle 2 (consolidation), and 1 ended in retreatment.
Among the 21 evaluable patients who received at least 1 week of blinatumomab at the target dose of 112 mcg/day or who discontinued treatment earlier because of progressive disease, the overall response rate was 43% (9/21) and the complete response rate was 19% (4/21). “At least 1 week of treatment at the target dose of 112 mcg/d appears to be required for efficacy,” wrote Dr. Andreas Viardot of the department of internal medicine III, University Hospital Ulm (Germany) and colleagues (Blood. 2016;127(11):1410-16).
The most common adverse events of grade 3 or higher were thrombocytopenia (17%) and leukopenia (17%). Grade 3 neurologic events included encephalopathy (9%) and aphasia (9%); no grade 4 or 5 neurologic events were reported. Cytokine release syndrome or cytokine storm were not reported.
Blinatumomab (Blincyto) is a bispecific T-cell engager antibody approved for the treatment of Philadelphia chromosome-negative relapsed or refractory B-cell precursor acute lymphoblastic leukemia. Blinatumomab transiently links CD3-positive T cells to CD19-positive B cells, which activates T cells and leads to T cell–mediated lysis of tumor cells and concomitant T-cell proliferation.
The study was supported by Amgen, the maker of Blincyto. Dr. Viardot reported having financial ties with Amgen and several other drug companies.
Blinatumomab, administered in a stepwise, dose-escalated regimen with prophylactic dexamethasone, induced responses in patients with relapsed/refractory diffuse large B-cell lymphoma (DLBCL), according to a report published in Blood.
The phase II study included 25 patients who were refractory to their last treatment, had relapsed after autologous hematopoietic stem cell transplant (HSCT), or had relapsed disease and were ineligible for autologous HSCT.
In a previous phase I study that did not use a dose-escalated regimen, neurological adverse events were dose limiting at a maximum tolerated blinatumomab dose of 60 mcg/d. The current study used a 14-day stepwise dose escalation and corticosteroid premedication to minimize adverse events: blinatumomab was given at 9 mcg/d in the first week, 28 mcg/d in the second week, and 112 mcg/d thereafter.
Patients had received a median of three prior therapies. In total, 17 of 25 patients ended in cycle 1 (induction) because of disease progression, adverse events, or physician decision; 7 ended in cycle 2 (consolidation), and 1 ended in retreatment.
Among the 21 evaluable patients who received at least 1 week of blinatumomab at the target dose of 112 mcg/day or who discontinued treatment earlier because of progressive disease, the overall response rate was 43% (9/21) and the complete response rate was 19% (4/21). “At least 1 week of treatment at the target dose of 112 mcg/d appears to be required for efficacy,” wrote Dr. Andreas Viardot of the department of internal medicine III, University Hospital Ulm (Germany) and colleagues (Blood. 2016;127(11):1410-16).
The most common adverse events of grade 3 or higher were thrombocytopenia (17%) and leukopenia (17%). Grade 3 neurologic events included encephalopathy (9%) and aphasia (9%); no grade 4 or 5 neurologic events were reported. Cytokine release syndrome or cytokine storm were not reported.
Blinatumomab (Blincyto) is a bispecific T-cell engager antibody approved for the treatment of Philadelphia chromosome-negative relapsed or refractory B-cell precursor acute lymphoblastic leukemia. Blinatumomab transiently links CD3-positive T cells to CD19-positive B cells, which activates T cells and leads to T cell–mediated lysis of tumor cells and concomitant T-cell proliferation.
The study was supported by Amgen, the maker of Blincyto. Dr. Viardot reported having financial ties with Amgen and several other drug companies.
Blinatumomab, administered in a stepwise, dose-escalated regimen with prophylactic dexamethasone, induced responses in patients with relapsed/refractory diffuse large B-cell lymphoma (DLBCL), according to a report published in Blood.
The phase II study included 25 patients who were refractory to their last treatment, had relapsed after autologous hematopoietic stem cell transplant (HSCT), or had relapsed disease and were ineligible for autologous HSCT.
In a previous phase I study that did not use a dose-escalated regimen, neurological adverse events were dose limiting at a maximum tolerated blinatumomab dose of 60 mcg/d. The current study used a 14-day stepwise dose escalation and corticosteroid premedication to minimize adverse events: blinatumomab was given at 9 mcg/d in the first week, 28 mcg/d in the second week, and 112 mcg/d thereafter.
Patients had received a median of three prior therapies. In total, 17 of 25 patients ended in cycle 1 (induction) because of disease progression, adverse events, or physician decision; 7 ended in cycle 2 (consolidation), and 1 ended in retreatment.
Among the 21 evaluable patients who received at least 1 week of blinatumomab at the target dose of 112 mcg/day or who discontinued treatment earlier because of progressive disease, the overall response rate was 43% (9/21) and the complete response rate was 19% (4/21). “At least 1 week of treatment at the target dose of 112 mcg/d appears to be required for efficacy,” wrote Dr. Andreas Viardot of the department of internal medicine III, University Hospital Ulm (Germany) and colleagues (Blood. 2016;127(11):1410-16).
The most common adverse events of grade 3 or higher were thrombocytopenia (17%) and leukopenia (17%). Grade 3 neurologic events included encephalopathy (9%) and aphasia (9%); no grade 4 or 5 neurologic events were reported. Cytokine release syndrome or cytokine storm were not reported.
Blinatumomab (Blincyto) is a bispecific T-cell engager antibody approved for the treatment of Philadelphia chromosome-negative relapsed or refractory B-cell precursor acute lymphoblastic leukemia. Blinatumomab transiently links CD3-positive T cells to CD19-positive B cells, which activates T cells and leads to T cell–mediated lysis of tumor cells and concomitant T-cell proliferation.
The study was supported by Amgen, the maker of Blincyto. Dr. Viardot reported having financial ties with Amgen and several other drug companies.
FROM BLOOD
Key clinical point: Stepwise dose-escalated administration of blinatumomab plus dexamethasone may be effective in patients with relapsed/refractory diffuse large B-cell lymphoma (DLBCL).
Major finding: The overall response rate in 21 evaluable patients was 43% and the complete response rate was 19%.
Data sources: Phase II study of 25 patients who had received a median of three prior therapies.
Disclosures: The study was supported by Amgen, the maker of blinatumomab. Dr. Viardot reported having financial ties with Amgen and several other drug makers.
FDG-PET guides need for eBEACOPP in advanced Hodgkin’s
Using FDG-PET (fluorodeoxyglucose positron emission tomography) imaging to gauge treatment response after the first two rounds of ABVD therapy helps to determine which patients with advanced Hodgkin’s lymphoma should be switched to a more aggressive eBEACOPP regimen, according to the results of the Southwest Oncology Group (SWOG) S0816 study.
In this large U.S. trial of PET scanning to guide treatment approach in people with high-risk stage II or stage III-IV Hodgkin’s lymphoma, progression-free survival at 2 years for those with early interim positive PET scans was 64%, which is much higher than the expected progression-free survival of 15%-30%, according to Dr. Oliver Press, a SWOG member at Fred Hutchinson Cancer Research Center and the lead author of study, which was published ahead of print in the Journal of Clinical Oncology (2016 April 11. doi: 10.1200/JCO.2015.63.1119).
In addition, just 20% of patients in the trial were exposed to eBEACOPP, which usually results in infertility, can cause sustained heart or lung damage, and increases the risk of secondary cancers.
Researchers recruited 358 Hodgkin’s patients to the trial and were able to evaluate 331 of them. All trial volunteers were given two rounds of standard ABVD (doxorubicin, bleomycin, vinblastine, and dacarbazine) chemotherapy, followed by a PET scan. If the scan was negative, patients received four more cycles of ABVD. If the scan was positive, with a Deauville score of 4-5, they were advised to switch to eBEACOPP (bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine, and prednisone), a seven-drug combination used in Europe. Of 60 patients with positive interim PET scans, 11 patients declined to switch, and 49 switched as planned to six cycles of eBEACOPP.
With a median follow-up of nearly 40 months, the Kaplan-Meier estimate for 2-year overall survival was 98%, and the 2-year estimate for progression-free survival was 79%. In the subset of patients who had positive PET scans after two cycles of ABVD, the 2-year estimate for progression-free survival was 64%, more than double the expected remission rate.
At least seven phase II and III cooperative group studies are underway testing this approach in advanced-stage Hodgkin’s lymphoma, the researchers wrote. “We hope that in the future, molecular biomarker studies at initial diagnosis, or the combination of biomarkers and molecular imaging, may define patients who require more intense therapy with eBEACOPP or other novel targeted drugs with greater accuracy than is achievable with current technology.”
The study was funded by the National Cancer Institute, the David and Patricia Giuliani Family Foundation, the Lymphoma Foundation, the Adam Spector Fund for Hodgkin Research, and the Ernest & Jeanette Dicker Charitable Foundation.
On Twitter @maryjodales
Using FDG-PET (fluorodeoxyglucose positron emission tomography) imaging to gauge treatment response after the first two rounds of ABVD therapy helps to determine which patients with advanced Hodgkin’s lymphoma should be switched to a more aggressive eBEACOPP regimen, according to the results of the Southwest Oncology Group (SWOG) S0816 study.
In this large U.S. trial of PET scanning to guide treatment approach in people with high-risk stage II or stage III-IV Hodgkin’s lymphoma, progression-free survival at 2 years for those with early interim positive PET scans was 64%, which is much higher than the expected progression-free survival of 15%-30%, according to Dr. Oliver Press, a SWOG member at Fred Hutchinson Cancer Research Center and the lead author of study, which was published ahead of print in the Journal of Clinical Oncology (2016 April 11. doi: 10.1200/JCO.2015.63.1119).
In addition, just 20% of patients in the trial were exposed to eBEACOPP, which usually results in infertility, can cause sustained heart or lung damage, and increases the risk of secondary cancers.
Researchers recruited 358 Hodgkin’s patients to the trial and were able to evaluate 331 of them. All trial volunteers were given two rounds of standard ABVD (doxorubicin, bleomycin, vinblastine, and dacarbazine) chemotherapy, followed by a PET scan. If the scan was negative, patients received four more cycles of ABVD. If the scan was positive, with a Deauville score of 4-5, they were advised to switch to eBEACOPP (bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine, and prednisone), a seven-drug combination used in Europe. Of 60 patients with positive interim PET scans, 11 patients declined to switch, and 49 switched as planned to six cycles of eBEACOPP.
With a median follow-up of nearly 40 months, the Kaplan-Meier estimate for 2-year overall survival was 98%, and the 2-year estimate for progression-free survival was 79%. In the subset of patients who had positive PET scans after two cycles of ABVD, the 2-year estimate for progression-free survival was 64%, more than double the expected remission rate.
At least seven phase II and III cooperative group studies are underway testing this approach in advanced-stage Hodgkin’s lymphoma, the researchers wrote. “We hope that in the future, molecular biomarker studies at initial diagnosis, or the combination of biomarkers and molecular imaging, may define patients who require more intense therapy with eBEACOPP or other novel targeted drugs with greater accuracy than is achievable with current technology.”
The study was funded by the National Cancer Institute, the David and Patricia Giuliani Family Foundation, the Lymphoma Foundation, the Adam Spector Fund for Hodgkin Research, and the Ernest & Jeanette Dicker Charitable Foundation.
On Twitter @maryjodales
Using FDG-PET (fluorodeoxyglucose positron emission tomography) imaging to gauge treatment response after the first two rounds of ABVD therapy helps to determine which patients with advanced Hodgkin’s lymphoma should be switched to a more aggressive eBEACOPP regimen, according to the results of the Southwest Oncology Group (SWOG) S0816 study.
In this large U.S. trial of PET scanning to guide treatment approach in people with high-risk stage II or stage III-IV Hodgkin’s lymphoma, progression-free survival at 2 years for those with early interim positive PET scans was 64%, which is much higher than the expected progression-free survival of 15%-30%, according to Dr. Oliver Press, a SWOG member at Fred Hutchinson Cancer Research Center and the lead author of study, which was published ahead of print in the Journal of Clinical Oncology (2016 April 11. doi: 10.1200/JCO.2015.63.1119).
In addition, just 20% of patients in the trial were exposed to eBEACOPP, which usually results in infertility, can cause sustained heart or lung damage, and increases the risk of secondary cancers.
Researchers recruited 358 Hodgkin’s patients to the trial and were able to evaluate 331 of them. All trial volunteers were given two rounds of standard ABVD (doxorubicin, bleomycin, vinblastine, and dacarbazine) chemotherapy, followed by a PET scan. If the scan was negative, patients received four more cycles of ABVD. If the scan was positive, with a Deauville score of 4-5, they were advised to switch to eBEACOPP (bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine, and prednisone), a seven-drug combination used in Europe. Of 60 patients with positive interim PET scans, 11 patients declined to switch, and 49 switched as planned to six cycles of eBEACOPP.
With a median follow-up of nearly 40 months, the Kaplan-Meier estimate for 2-year overall survival was 98%, and the 2-year estimate for progression-free survival was 79%. In the subset of patients who had positive PET scans after two cycles of ABVD, the 2-year estimate for progression-free survival was 64%, more than double the expected remission rate.
At least seven phase II and III cooperative group studies are underway testing this approach in advanced-stage Hodgkin’s lymphoma, the researchers wrote. “We hope that in the future, molecular biomarker studies at initial diagnosis, or the combination of biomarkers and molecular imaging, may define patients who require more intense therapy with eBEACOPP or other novel targeted drugs with greater accuracy than is achievable with current technology.”
The study was funded by the National Cancer Institute, the David and Patricia Giuliani Family Foundation, the Lymphoma Foundation, the Adam Spector Fund for Hodgkin Research, and the Ernest & Jeanette Dicker Charitable Foundation.
On Twitter @maryjodales
FROM JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Using FDG-PET imaging to gauge treatment response after the first two rounds of ABVD therapy helps to determine which patients with advanced Hodgkin’s lymphoma should be switched to the eBEACOPP regimen.
Major finding: Progression-free survival at 2 years for those with early interim positive PET scans was 64%; the historical progression-free survival for this group is 15%-30%.
Data source: Evaluations of 331 patients in the Southwest Oncology Group S0816 study.
Disclosures: The study was funded by the National Cancer Institute, the David and Patricia Giuliani Family Foundation, the Lymphoma Foundation, the Adam Spector Fund for Hodgkin Research, and the Ernest & Jeanette Dicker Charitable Foundation.
OTX015 dose for lymphoma narrowed in phase 1 study
As a single agent for use in patients with lymphoma, an acceptable once-daily dose of OTX015 appears to be 80 mg on a 14 days on, 7 days off schedule, the results of a phase 1 study indicate.
The small-molecule inhibitor, which inhibits binding of bromodomain and exterminal proteins to acetylated histones, was associated with acceptable toxicity and efficacy in this regimen. The investigational drug is now being tested in expansion cohorts on a schedule of 14 days every 3 weeks, a regimen projected to allow for recovery from the drug’s toxic effects, Dr. Sandy Amorin of Hôpital Saint Louis, Paris, and associates reported.
The drug also is being evaluated in patients with acute leukemias.
Adults with nonleukemia hematologic malignancies that progressed on standard therapies participated in the open-label study, which was conducted at seven university hospital centers in Europe. Oral OTX015 was given once a day at one of five doses (10 mg, 20 mg, 40 mg, 80 mg, and 120 mg). The 3 + 3 study design permitted evaluation of alternative administration schedules. The primary endpoint was dose-limiting toxicity in the first treatment cycle (21 days). Secondary objectives were to evaluate safety, pharmacokinetics, and preliminary clinical activity of OTX015. The study is ongoing and is registered with ClinicalTrials.gov, number NCT01713582.
The study included 33 patients with lymphoma and 12 with myeloma; patients’ median age was 66 years, and they had received a median of four lines of prior therapy. No dose-limiting toxicities were seen in three patients given doses as high as 80 mg once a day. However, grade 4 thrombocytopenia occurred in five of six patients on a 21-day schedule of 40 mg twice a day. No patient tolerated various schedules of 120 mg once a day (Lancet Haematol. 2016;3[4]:e196-204).
The researchers then examined the 80 mg once a day dose on a continuous basis in four patients, two of whom developed grade 4 thrombocytopenia. In light of these and other toxicities, a regimen was proposed of 80 mg once a day on a schedule of 14 days on, 7 days off.
Thrombocytopenia affected 43 of 45 patients, and 26 of them had grade 3-4 events. Other grade 3-4 events were infrequent. Anemia was seen in 41, and neutropenia in 23.
Of three patients with diffuse large B-cell lymphoma, two had complete responses at 120 mg once a day, and one had a partial response at 80 mg once a day. Six additional patients, two with diffuse large B-cell lymphoma and four with indolent lymphomas, had evidence of clinical activity, but did not meet the criteria for an objective response.
The study was funded by the developers of OTX015, Oncoethix GmbH, a wholly owned subsidiary of Merck Sharp & Dohme.
On Twitter @maryjodales
As a single agent for use in patients with lymphoma, an acceptable once-daily dose of OTX015 appears to be 80 mg on a 14 days on, 7 days off schedule, the results of a phase 1 study indicate.
The small-molecule inhibitor, which inhibits binding of bromodomain and exterminal proteins to acetylated histones, was associated with acceptable toxicity and efficacy in this regimen. The investigational drug is now being tested in expansion cohorts on a schedule of 14 days every 3 weeks, a regimen projected to allow for recovery from the drug’s toxic effects, Dr. Sandy Amorin of Hôpital Saint Louis, Paris, and associates reported.
The drug also is being evaluated in patients with acute leukemias.
Adults with nonleukemia hematologic malignancies that progressed on standard therapies participated in the open-label study, which was conducted at seven university hospital centers in Europe. Oral OTX015 was given once a day at one of five doses (10 mg, 20 mg, 40 mg, 80 mg, and 120 mg). The 3 + 3 study design permitted evaluation of alternative administration schedules. The primary endpoint was dose-limiting toxicity in the first treatment cycle (21 days). Secondary objectives were to evaluate safety, pharmacokinetics, and preliminary clinical activity of OTX015. The study is ongoing and is registered with ClinicalTrials.gov, number NCT01713582.
The study included 33 patients with lymphoma and 12 with myeloma; patients’ median age was 66 years, and they had received a median of four lines of prior therapy. No dose-limiting toxicities were seen in three patients given doses as high as 80 mg once a day. However, grade 4 thrombocytopenia occurred in five of six patients on a 21-day schedule of 40 mg twice a day. No patient tolerated various schedules of 120 mg once a day (Lancet Haematol. 2016;3[4]:e196-204).
The researchers then examined the 80 mg once a day dose on a continuous basis in four patients, two of whom developed grade 4 thrombocytopenia. In light of these and other toxicities, a regimen was proposed of 80 mg once a day on a schedule of 14 days on, 7 days off.
Thrombocytopenia affected 43 of 45 patients, and 26 of them had grade 3-4 events. Other grade 3-4 events were infrequent. Anemia was seen in 41, and neutropenia in 23.
Of three patients with diffuse large B-cell lymphoma, two had complete responses at 120 mg once a day, and one had a partial response at 80 mg once a day. Six additional patients, two with diffuse large B-cell lymphoma and four with indolent lymphomas, had evidence of clinical activity, but did not meet the criteria for an objective response.
The study was funded by the developers of OTX015, Oncoethix GmbH, a wholly owned subsidiary of Merck Sharp & Dohme.
On Twitter @maryjodales
As a single agent for use in patients with lymphoma, an acceptable once-daily dose of OTX015 appears to be 80 mg on a 14 days on, 7 days off schedule, the results of a phase 1 study indicate.
The small-molecule inhibitor, which inhibits binding of bromodomain and exterminal proteins to acetylated histones, was associated with acceptable toxicity and efficacy in this regimen. The investigational drug is now being tested in expansion cohorts on a schedule of 14 days every 3 weeks, a regimen projected to allow for recovery from the drug’s toxic effects, Dr. Sandy Amorin of Hôpital Saint Louis, Paris, and associates reported.
The drug also is being evaluated in patients with acute leukemias.
Adults with nonleukemia hematologic malignancies that progressed on standard therapies participated in the open-label study, which was conducted at seven university hospital centers in Europe. Oral OTX015 was given once a day at one of five doses (10 mg, 20 mg, 40 mg, 80 mg, and 120 mg). The 3 + 3 study design permitted evaluation of alternative administration schedules. The primary endpoint was dose-limiting toxicity in the first treatment cycle (21 days). Secondary objectives were to evaluate safety, pharmacokinetics, and preliminary clinical activity of OTX015. The study is ongoing and is registered with ClinicalTrials.gov, number NCT01713582.
The study included 33 patients with lymphoma and 12 with myeloma; patients’ median age was 66 years, and they had received a median of four lines of prior therapy. No dose-limiting toxicities were seen in three patients given doses as high as 80 mg once a day. However, grade 4 thrombocytopenia occurred in five of six patients on a 21-day schedule of 40 mg twice a day. No patient tolerated various schedules of 120 mg once a day (Lancet Haematol. 2016;3[4]:e196-204).
The researchers then examined the 80 mg once a day dose on a continuous basis in four patients, two of whom developed grade 4 thrombocytopenia. In light of these and other toxicities, a regimen was proposed of 80 mg once a day on a schedule of 14 days on, 7 days off.
Thrombocytopenia affected 43 of 45 patients, and 26 of them had grade 3-4 events. Other grade 3-4 events were infrequent. Anemia was seen in 41, and neutropenia in 23.
Of three patients with diffuse large B-cell lymphoma, two had complete responses at 120 mg once a day, and one had a partial response at 80 mg once a day. Six additional patients, two with diffuse large B-cell lymphoma and four with indolent lymphomas, had evidence of clinical activity, but did not meet the criteria for an objective response.
The study was funded by the developers of OTX015, Oncoethix GmbH, a wholly owned subsidiary of Merck Sharp & Dohme.
On Twitter @maryjodales
FROM THE LANCET HAEMATOLOGY
Key clinical point: For lymphoma patients, a regimen has been determined for the small-molecule inhibitor OTX015 that was associated with acceptable toxicity and efficacy.
Major finding: On a regimen of 80 mg once a day on a schedule of 14 days on, 7 days off, thrombocytopenia affected 43 of 45 patients, and 26 of them had grade 3-4 events. However, other grade 3-4 events were infrequent.
Data source: The open-label study NCT01713582 was conducted at seven university hospital centers in Europe.
Disclosures: The study was funded by the developers of OTX015, Oncoethix GmbH, a wholly owned subsidiary of Merck Sharp & Dohme.
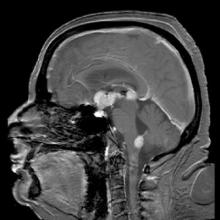
Temsirolimus results in good but short-duration responses in primary CNS lymphoma
Single-agent therapy with temsirolimus was active in patients with relapsed/refractory primary central nervous system lymphoma, but most of the responses were short lived, results of a phase II trial show.
Among 37 patients with primary CNS lymphoma (PCNSL) for whom firstline therapy had failed, there were five complete responses (CR), three CR unconfirmed, and 12 partial responses (PR), for an overall response rate (ORR) of 54%, reported Dr. Agnieszka Korfel from Charité University Medicine Berlin (Germany) and colleagues.
The median progression-free survival (PFS), however, was just 2.1 months, although 1 patient had PFS of 15.8 months duration, and another had a response lasting for more than 44 months, the investigators noted in a study published online in the Journal of Clinical Oncology (doi: 10.1200/JCO.2015.64.9897).
The rationale for trying temsirolimus (Torisel), an inhibitor of the mammalian target of rapamycin (mTOR), came from studies showing the drug’s efficacy against relapsed/refractory mantle-cell lymphoma and against other, more aggressive forms of non-Hodgkin lymphoma. Patients with relapsed/refractory aggressive lymphomas tolerate temsirolimus relatively well, and the drug has the ability to penetrate brain tumor tissue, the authors noted.
They enrolled 37 patients with a median age of 70 years and a median time since their last treatment of 3.9 months into an open-label trial. The patients were all immuncompetent with histologically confirmed primary central nervous system lymphoma for whom high-dose methotrexate-based chemotherapy had failed and for whom high-dose chemotherapy with autologous stem cell transplant had either failed or was not an option.
The first six patients were treated with temsirolimus 25 mg intravenously once weekly, and the remaining 31 were treated with 75 mg IV once weekly until disease progression, intolerable toxicity, patient or physician decision to terminate, or death.
As noted before, ORR, the primary endpoint, was 54%. Median overall survival (OS), a secondary endpoint, was 3.7 months, and 1-year and 2-year OS were 19% and 16.2%, respectively.
The most frequently occurring toxicities include hyperglycemia, myelosuppression, pneumonias and other infections, and fatigue. A total of 28 severe adverse events occurred in 21 patients, including infectious episodes, hospitalizations because of disease progression, deep-vein thromboses, hyperglycemia, and one case each of seizures, grade 4 thrombocytopenia, drug fever, hyponatremia, renal insufficiency, and atrial fibrillation.
“Although most responses were short lived, some patients achieved long-term control. Thus, further evaluation in combination with other drugs seems reasonable. However, one has to be aware of the risk of hematotoxicity and infections necessitating primary antibiotic prophylaxis. Definition of biomarkers allowing identification of potential responders and those who are at particular risk for toxicity would be highly desirable,” the investigators concluded.
Single-agent therapy with temsirolimus was active in patients with relapsed/refractory primary central nervous system lymphoma, but most of the responses were short lived, results of a phase II trial show.
Among 37 patients with primary CNS lymphoma (PCNSL) for whom firstline therapy had failed, there were five complete responses (CR), three CR unconfirmed, and 12 partial responses (PR), for an overall response rate (ORR) of 54%, reported Dr. Agnieszka Korfel from Charité University Medicine Berlin (Germany) and colleagues.
The median progression-free survival (PFS), however, was just 2.1 months, although 1 patient had PFS of 15.8 months duration, and another had a response lasting for more than 44 months, the investigators noted in a study published online in the Journal of Clinical Oncology (doi: 10.1200/JCO.2015.64.9897).
The rationale for trying temsirolimus (Torisel), an inhibitor of the mammalian target of rapamycin (mTOR), came from studies showing the drug’s efficacy against relapsed/refractory mantle-cell lymphoma and against other, more aggressive forms of non-Hodgkin lymphoma. Patients with relapsed/refractory aggressive lymphomas tolerate temsirolimus relatively well, and the drug has the ability to penetrate brain tumor tissue, the authors noted.
They enrolled 37 patients with a median age of 70 years and a median time since their last treatment of 3.9 months into an open-label trial. The patients were all immuncompetent with histologically confirmed primary central nervous system lymphoma for whom high-dose methotrexate-based chemotherapy had failed and for whom high-dose chemotherapy with autologous stem cell transplant had either failed or was not an option.
The first six patients were treated with temsirolimus 25 mg intravenously once weekly, and the remaining 31 were treated with 75 mg IV once weekly until disease progression, intolerable toxicity, patient or physician decision to terminate, or death.
As noted before, ORR, the primary endpoint, was 54%. Median overall survival (OS), a secondary endpoint, was 3.7 months, and 1-year and 2-year OS were 19% and 16.2%, respectively.
The most frequently occurring toxicities include hyperglycemia, myelosuppression, pneumonias and other infections, and fatigue. A total of 28 severe adverse events occurred in 21 patients, including infectious episodes, hospitalizations because of disease progression, deep-vein thromboses, hyperglycemia, and one case each of seizures, grade 4 thrombocytopenia, drug fever, hyponatremia, renal insufficiency, and atrial fibrillation.
“Although most responses were short lived, some patients achieved long-term control. Thus, further evaluation in combination with other drugs seems reasonable. However, one has to be aware of the risk of hematotoxicity and infections necessitating primary antibiotic prophylaxis. Definition of biomarkers allowing identification of potential responders and those who are at particular risk for toxicity would be highly desirable,” the investigators concluded.
Single-agent therapy with temsirolimus was active in patients with relapsed/refractory primary central nervous system lymphoma, but most of the responses were short lived, results of a phase II trial show.
Among 37 patients with primary CNS lymphoma (PCNSL) for whom firstline therapy had failed, there were five complete responses (CR), three CR unconfirmed, and 12 partial responses (PR), for an overall response rate (ORR) of 54%, reported Dr. Agnieszka Korfel from Charité University Medicine Berlin (Germany) and colleagues.
The median progression-free survival (PFS), however, was just 2.1 months, although 1 patient had PFS of 15.8 months duration, and another had a response lasting for more than 44 months, the investigators noted in a study published online in the Journal of Clinical Oncology (doi: 10.1200/JCO.2015.64.9897).
The rationale for trying temsirolimus (Torisel), an inhibitor of the mammalian target of rapamycin (mTOR), came from studies showing the drug’s efficacy against relapsed/refractory mantle-cell lymphoma and against other, more aggressive forms of non-Hodgkin lymphoma. Patients with relapsed/refractory aggressive lymphomas tolerate temsirolimus relatively well, and the drug has the ability to penetrate brain tumor tissue, the authors noted.
They enrolled 37 patients with a median age of 70 years and a median time since their last treatment of 3.9 months into an open-label trial. The patients were all immuncompetent with histologically confirmed primary central nervous system lymphoma for whom high-dose methotrexate-based chemotherapy had failed and for whom high-dose chemotherapy with autologous stem cell transplant had either failed or was not an option.
The first six patients were treated with temsirolimus 25 mg intravenously once weekly, and the remaining 31 were treated with 75 mg IV once weekly until disease progression, intolerable toxicity, patient or physician decision to terminate, or death.
As noted before, ORR, the primary endpoint, was 54%. Median overall survival (OS), a secondary endpoint, was 3.7 months, and 1-year and 2-year OS were 19% and 16.2%, respectively.
The most frequently occurring toxicities include hyperglycemia, myelosuppression, pneumonias and other infections, and fatigue. A total of 28 severe adverse events occurred in 21 patients, including infectious episodes, hospitalizations because of disease progression, deep-vein thromboses, hyperglycemia, and one case each of seizures, grade 4 thrombocytopenia, drug fever, hyponatremia, renal insufficiency, and atrial fibrillation.
“Although most responses were short lived, some patients achieved long-term control. Thus, further evaluation in combination with other drugs seems reasonable. However, one has to be aware of the risk of hematotoxicity and infections necessitating primary antibiotic prophylaxis. Definition of biomarkers allowing identification of potential responders and those who are at particular risk for toxicity would be highly desirable,” the investigators concluded.
FROM JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Relapsed/refractory primary CNS lymphoma has a poor prognosis and no standard treatment option.
Major finding: The overall response rate to once-weekly temsirolimus was 54%; most responses were short lived.
Data source: Open-label phase 2 study in 37 adults with relapsed/refractory primary CNS lymphoma.
Disclosures: The study was supported by Pfizer Germany. Dr. Korfel and several colleagues disclosed research support from or consulting/advising for the company.
Pregnancy did not increase Hodgkin lymphoma relapse rate
Women who become pregnant while in remission from Hodgkin lymphoma were not at increased risk for cancer relapse, according to an analysis of data from Swedish health care registries combined with medical records.
Of 449 women who were diagnosed with Hodgkin lymphoma between 1992 and 2009, 144 (32%) became pregnant during follow-up, which started 6 months after diagnosis, when the disease was assumed to be in remission. Only one of these women experienced a pregnancy-associated relapse, which was defined as a relapse occurring during pregnancy or within 5 years of delivery. Of the women who did not become pregnant, 46 had a relapse.
The effect of pregnancy on relapse has been a concern of patients and clinicians, but “our findings suggest that the risk of pregnancy-associated relapse does not need to be taken into account in family planning for women whose Hodgkin lymphoma is in remission,” said Caroline E. Weibull of Karolinska Institutet in Stockholm, and her associates.
The researchers used the nationwide “Swedish Cancer Register” to identify all cases of Hodgkin lymphoma (reporting is mandatory) and merged this data with clinical information from other registries and medical records.
The pregnancy rates were similar among women who had limited- and advanced-stage disease and among women with and without B symptoms at diagnosis – a finding that negates consideration of a so-called “healthy mother effect” in protecting against relapse, they wrote (J Clin Onc. 2015 Dec. 14 [doi:10.1200/JCO.2015.63.3446]).
The researchers also found that the absolute risk for relapse was highest in the first 2-3 years after diagnosis, which suggests that women should be advised, “if possible, to wait 2 years after cessation of treatment before becoming pregnant.” Additionally, the relapse rate more than doubled in women aged 30 years or older at diagnosis, compared with women aged 18-24 years at diagnosis – a finding consistent with previous research, they noted.
Women in the study were aged 18-40 at diagnosis. Follow-up ended on the date of relapse, the date of death, or at the end of 2010, whichever came first.
Women who become pregnant while in remission from Hodgkin lymphoma were not at increased risk for cancer relapse, according to an analysis of data from Swedish health care registries combined with medical records.
Of 449 women who were diagnosed with Hodgkin lymphoma between 1992 and 2009, 144 (32%) became pregnant during follow-up, which started 6 months after diagnosis, when the disease was assumed to be in remission. Only one of these women experienced a pregnancy-associated relapse, which was defined as a relapse occurring during pregnancy or within 5 years of delivery. Of the women who did not become pregnant, 46 had a relapse.
The effect of pregnancy on relapse has been a concern of patients and clinicians, but “our findings suggest that the risk of pregnancy-associated relapse does not need to be taken into account in family planning for women whose Hodgkin lymphoma is in remission,” said Caroline E. Weibull of Karolinska Institutet in Stockholm, and her associates.
The researchers used the nationwide “Swedish Cancer Register” to identify all cases of Hodgkin lymphoma (reporting is mandatory) and merged this data with clinical information from other registries and medical records.
The pregnancy rates were similar among women who had limited- and advanced-stage disease and among women with and without B symptoms at diagnosis – a finding that negates consideration of a so-called “healthy mother effect” in protecting against relapse, they wrote (J Clin Onc. 2015 Dec. 14 [doi:10.1200/JCO.2015.63.3446]).
The researchers also found that the absolute risk for relapse was highest in the first 2-3 years after diagnosis, which suggests that women should be advised, “if possible, to wait 2 years after cessation of treatment before becoming pregnant.” Additionally, the relapse rate more than doubled in women aged 30 years or older at diagnosis, compared with women aged 18-24 years at diagnosis – a finding consistent with previous research, they noted.
Women in the study were aged 18-40 at diagnosis. Follow-up ended on the date of relapse, the date of death, or at the end of 2010, whichever came first.
Women who become pregnant while in remission from Hodgkin lymphoma were not at increased risk for cancer relapse, according to an analysis of data from Swedish health care registries combined with medical records.
Of 449 women who were diagnosed with Hodgkin lymphoma between 1992 and 2009, 144 (32%) became pregnant during follow-up, which started 6 months after diagnosis, when the disease was assumed to be in remission. Only one of these women experienced a pregnancy-associated relapse, which was defined as a relapse occurring during pregnancy or within 5 years of delivery. Of the women who did not become pregnant, 46 had a relapse.
The effect of pregnancy on relapse has been a concern of patients and clinicians, but “our findings suggest that the risk of pregnancy-associated relapse does not need to be taken into account in family planning for women whose Hodgkin lymphoma is in remission,” said Caroline E. Weibull of Karolinska Institutet in Stockholm, and her associates.
The researchers used the nationwide “Swedish Cancer Register” to identify all cases of Hodgkin lymphoma (reporting is mandatory) and merged this data with clinical information from other registries and medical records.
The pregnancy rates were similar among women who had limited- and advanced-stage disease and among women with and without B symptoms at diagnosis – a finding that negates consideration of a so-called “healthy mother effect” in protecting against relapse, they wrote (J Clin Onc. 2015 Dec. 14 [doi:10.1200/JCO.2015.63.3446]).
The researchers also found that the absolute risk for relapse was highest in the first 2-3 years after diagnosis, which suggests that women should be advised, “if possible, to wait 2 years after cessation of treatment before becoming pregnant.” Additionally, the relapse rate more than doubled in women aged 30 years or older at diagnosis, compared with women aged 18-24 years at diagnosis – a finding consistent with previous research, they noted.
Women in the study were aged 18-40 at diagnosis. Follow-up ended on the date of relapse, the date of death, or at the end of 2010, whichever came first.
FROM JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Pregnancy did not increase the risk of relapse of Hodgkin lymphoma in a population-based study.
Major finding: Of 144 women who became pregnant 6 months or longer after diagnosis of Hodgkin lymphoma, 1 experienced a pregnancy-associated relapse.
Data source: Population-based study utilizing Swedish health care registries and medical records, in which 449 women with Hodgkin lymphoma diagnoses, and 47 relapses, were identified.
Disclosures: The study was supported by the Swedish Cancer Society, the Strategic Research Program in Epidemiology at Karolinska Institutet, the Swedish Society for Medicine, and the Swedish Society for Medical Research.
Expert panel issues guidelines for treatment of hematologic cancers in pregnancy
Consensus guidelines for the perinatal management of hematologic malignancies detected during pregnancy have been issued by a panel of international experts.
The guidelines, published online in the Journal of Clinical Oncology, aim to ensure that timely treatment of the cancers is not delayed in pregnant women (doi: 10.1200/JCO.2015.62.4445).
While rare, hematologic malignancies in pregnancy introduce clinical, social, ethical, and moral dilemmas. Evidence-based data are scarce, according to the researchers, who note the International Network on Cancer, Infertility and Pregnancy registers all cancers occurring during gestation.
“Patient accrual is ongoing and essential, because registration of new cases and long-term follow-up will improve clinical knowledge and increase the level of evidence,” Dr. Michael Lishner of Tel Aviv University and Meir Medical Center, Kfar Saba, Israel, and his fellow panelists wrote.
Hodgkin lymphoma
The researchers note that Hodgkin lymphoma is the most common hematologic cancer in pregnancy, and the prognosis for these patients is excellent. When diagnosed during the first trimester, a regimen based on vinblastine monotherapy has been used. ABVD (doxorubicin, bleomycin, vinblastine, dacarbazine) therapy can be used postpartum and has been used in cases of progression during pregnancy, the panelists wrote.
“The limited data available suggest that ABVD may be administered safely and effectively during the latter phases of pregnancy,” the panel wrote. “Although it may be associated with prematurity and lower birth weights, studies have not reported significant disadvantages.”
Non-Hodgkin lymphoma
The second most common cancer in pregnancy is non-Hodgkin lymphoma. In the case of indolent disease, watchful waiting is possible, with the intent to treat with monoclonal antibodies – with or without chemotherapy – if symptoms or evidence of disease progression are noted. Steroids can be administered during the first trimester as a bridge to the second trimester, when chemotherapy can be used with relatively greater safety, the panelists noted.
Aggressive lymphomas diagnosed before 20 weeks’ gestation warrant pregnancy termination and treatment, they recommend. When diagnosed after 20 weeks, therapy should be comparable to that given a nonpregnant woman, including monoclonal antibodies (R-CHOP).
Chronic myeloid leukemia
Chronic myeloid leukemia occurs in approximately 1 in 100,000 pregnancies and is typically diagnosed during routine blood testing in an asymptomatic patient. As a result, treatment may not be needed until the patient’s white count or platelet count have risen to levels associated with the onset of symptoms. An approximate guideline is a white cell count greater than 100 X 109/L and a platelet count greater than 500 X 109/L.
Therapeutic approaches in pregnancy include interferon-a (INF-a), which does not inhibit DNA synthesis or readily cross the placenta, and leukapheresis, which is frequently required two to three times per week during the first and second trimesters. Counts tend to drop during the third trimester, allowing less frequent intervention.
Consideration should be given to adding aspirin or low-molecular-weight heparin (LMWH) when the platelet count exceeds 1,000 X 109/L.
Myeloproliferative neoplasms
The most common myeloproliferative neoplasm seen in women of childbearing age is essential thrombocytosis.
“A large meta-analysis of pregnant women with essential thrombocytosis reported a live birth rate of 50%-70%, first trimester loss in 25%-40%, late pregnancy loss in 10%, placental abruption in 3.6%, and intrauterine growth restriction in 4.5%. Maternal morbidity is rare, but stroke has been reported,” according to the panelists.
Limited literature suggests similar outcomes for pregnant women with polycythemia vera and primary myelofibrosis.
In low-risk pregnancies, aspirin (75 mg/day) should be offered unless clearly contraindicated. For women with polycythemia vera, venesection may be continued when tolerated to maintain the hematocrit within the gestation-appropriate range.
Fetal ultrasound scans should be performed at 20, 26, and 34 weeks of gestation and uterine artery Doppler should be performed at 20 weeks’ gestation. If the mean pulsatility index exceeds 1.4, the pregnancy may be considered high risk, and treatment and monitoring should be increased.
In high-risk pregnancies, additional treatment includes cytoreductive therapy with or without LMWH. If cytoreductive therapy is required, INF-a should be titrated to maintain a platelet count of less than 400 X 109/L and hematocrit within appropriate range.
Local protocols regarding interruption of LMWH should be adhered to during labor, and dehydration should be avoided. Platelet count and hematocrit may increase postpartum, requiring cytoreductive therapy. Thromboprophylaxis should be considered at 6 weeks’ postpartum because of the increased risk of thrombosis, the guidelines note.
Acute leukemia
“The remarkable anemia, thrombocytopenia, and neutropenia that characterize acute myeloid and lymphoblastic leukemia” require prompt treatment. Leukapheresis in the presence of clinically significant evidence of leukostasis can be considered, regardless of gestational stage. When patients are diagnosed with acute myeloid leukemia during the first trimester, pregnancy termination followed by conventional induction therapy (cytarabine/anthracycline) is recommended.
Those diagnosed later in pregnancy can receive conventional induction therapy, although this seems to be associated with increased risk of fetal growth restriction and even fetal loss. “Notably, neonates rarely experience neutropenia and cardiac impairment unless exposed to lipophilic idarubicin, which should not be used,” the panelists wrote.
When acute promyelocytic leukemia is diagnosed in the first trimester, pregnancy termination is recommended before initiating conventional ATRA-anthracycline therapy. Later in pregnancy, the regimen demonstrates low teratogenicity and can be used in women diagnosed after that stage. Arsenic treatment is highly teratogenic and is prohibited throughout gestation.
Acute lymphocytic leukemia (ALL) requires prophylactic CNS therapy, including methotrexate and L-asparaginase, which are fetotoxic. Methotrexate interferes with organogenesis and is prohibited before week 20 of gestation. L-asparaginase may increase the high risk for thromboembolic events attributed to the combination of pregnancy and malignancy.
Notably, tyrosine kinase inhibitors, essential for patients with Philadelphia chromosome–positive ALL, are teratogenic. Given these limitations, women diagnosed with ALL before 20 weeks’ gestation should undergo termination of the pregnancy and start conventional treatment. After week 20, conventional chemotherapy can be administered during pregnancy. Tyrosine kinase inhibitors can be initiated postpartum.
The guidelines also contain recommendations on diagnostic testing and radiotherapy, maternal supportive care, and perinatal and pediatric aspects of hematologic malignancies in pregnancy. An online appendix offers recommendations on the treatment of rare hematologic malignancies, including hairy cell leukemia, multiple myeloma, and myelodysplastic syndromes.
Dr. Lishner and nine of his coauthors had no financial relationships to disclose. Three coauthors received honoraria and research funding or are consultants to a wide variety of drug makers.
On Twitter @maryjodales
Consensus guidelines for the perinatal management of hematologic malignancies detected during pregnancy have been issued by a panel of international experts.
The guidelines, published online in the Journal of Clinical Oncology, aim to ensure that timely treatment of the cancers is not delayed in pregnant women (doi: 10.1200/JCO.2015.62.4445).
While rare, hematologic malignancies in pregnancy introduce clinical, social, ethical, and moral dilemmas. Evidence-based data are scarce, according to the researchers, who note the International Network on Cancer, Infertility and Pregnancy registers all cancers occurring during gestation.
“Patient accrual is ongoing and essential, because registration of new cases and long-term follow-up will improve clinical knowledge and increase the level of evidence,” Dr. Michael Lishner of Tel Aviv University and Meir Medical Center, Kfar Saba, Israel, and his fellow panelists wrote.
Hodgkin lymphoma
The researchers note that Hodgkin lymphoma is the most common hematologic cancer in pregnancy, and the prognosis for these patients is excellent. When diagnosed during the first trimester, a regimen based on vinblastine monotherapy has been used. ABVD (doxorubicin, bleomycin, vinblastine, dacarbazine) therapy can be used postpartum and has been used in cases of progression during pregnancy, the panelists wrote.
“The limited data available suggest that ABVD may be administered safely and effectively during the latter phases of pregnancy,” the panel wrote. “Although it may be associated with prematurity and lower birth weights, studies have not reported significant disadvantages.”
Non-Hodgkin lymphoma
The second most common cancer in pregnancy is non-Hodgkin lymphoma. In the case of indolent disease, watchful waiting is possible, with the intent to treat with monoclonal antibodies – with or without chemotherapy – if symptoms or evidence of disease progression are noted. Steroids can be administered during the first trimester as a bridge to the second trimester, when chemotherapy can be used with relatively greater safety, the panelists noted.
Aggressive lymphomas diagnosed before 20 weeks’ gestation warrant pregnancy termination and treatment, they recommend. When diagnosed after 20 weeks, therapy should be comparable to that given a nonpregnant woman, including monoclonal antibodies (R-CHOP).
Chronic myeloid leukemia
Chronic myeloid leukemia occurs in approximately 1 in 100,000 pregnancies and is typically diagnosed during routine blood testing in an asymptomatic patient. As a result, treatment may not be needed until the patient’s white count or platelet count have risen to levels associated with the onset of symptoms. An approximate guideline is a white cell count greater than 100 X 109/L and a platelet count greater than 500 X 109/L.
Therapeutic approaches in pregnancy include interferon-a (INF-a), which does not inhibit DNA synthesis or readily cross the placenta, and leukapheresis, which is frequently required two to three times per week during the first and second trimesters. Counts tend to drop during the third trimester, allowing less frequent intervention.
Consideration should be given to adding aspirin or low-molecular-weight heparin (LMWH) when the platelet count exceeds 1,000 X 109/L.
Myeloproliferative neoplasms
The most common myeloproliferative neoplasm seen in women of childbearing age is essential thrombocytosis.
“A large meta-analysis of pregnant women with essential thrombocytosis reported a live birth rate of 50%-70%, first trimester loss in 25%-40%, late pregnancy loss in 10%, placental abruption in 3.6%, and intrauterine growth restriction in 4.5%. Maternal morbidity is rare, but stroke has been reported,” according to the panelists.
Limited literature suggests similar outcomes for pregnant women with polycythemia vera and primary myelofibrosis.
In low-risk pregnancies, aspirin (75 mg/day) should be offered unless clearly contraindicated. For women with polycythemia vera, venesection may be continued when tolerated to maintain the hematocrit within the gestation-appropriate range.
Fetal ultrasound scans should be performed at 20, 26, and 34 weeks of gestation and uterine artery Doppler should be performed at 20 weeks’ gestation. If the mean pulsatility index exceeds 1.4, the pregnancy may be considered high risk, and treatment and monitoring should be increased.
In high-risk pregnancies, additional treatment includes cytoreductive therapy with or without LMWH. If cytoreductive therapy is required, INF-a should be titrated to maintain a platelet count of less than 400 X 109/L and hematocrit within appropriate range.
Local protocols regarding interruption of LMWH should be adhered to during labor, and dehydration should be avoided. Platelet count and hematocrit may increase postpartum, requiring cytoreductive therapy. Thromboprophylaxis should be considered at 6 weeks’ postpartum because of the increased risk of thrombosis, the guidelines note.
Acute leukemia
“The remarkable anemia, thrombocytopenia, and neutropenia that characterize acute myeloid and lymphoblastic leukemia” require prompt treatment. Leukapheresis in the presence of clinically significant evidence of leukostasis can be considered, regardless of gestational stage. When patients are diagnosed with acute myeloid leukemia during the first trimester, pregnancy termination followed by conventional induction therapy (cytarabine/anthracycline) is recommended.
Those diagnosed later in pregnancy can receive conventional induction therapy, although this seems to be associated with increased risk of fetal growth restriction and even fetal loss. “Notably, neonates rarely experience neutropenia and cardiac impairment unless exposed to lipophilic idarubicin, which should not be used,” the panelists wrote.
When acute promyelocytic leukemia is diagnosed in the first trimester, pregnancy termination is recommended before initiating conventional ATRA-anthracycline therapy. Later in pregnancy, the regimen demonstrates low teratogenicity and can be used in women diagnosed after that stage. Arsenic treatment is highly teratogenic and is prohibited throughout gestation.
Acute lymphocytic leukemia (ALL) requires prophylactic CNS therapy, including methotrexate and L-asparaginase, which are fetotoxic. Methotrexate interferes with organogenesis and is prohibited before week 20 of gestation. L-asparaginase may increase the high risk for thromboembolic events attributed to the combination of pregnancy and malignancy.
Notably, tyrosine kinase inhibitors, essential for patients with Philadelphia chromosome–positive ALL, are teratogenic. Given these limitations, women diagnosed with ALL before 20 weeks’ gestation should undergo termination of the pregnancy and start conventional treatment. After week 20, conventional chemotherapy can be administered during pregnancy. Tyrosine kinase inhibitors can be initiated postpartum.
The guidelines also contain recommendations on diagnostic testing and radiotherapy, maternal supportive care, and perinatal and pediatric aspects of hematologic malignancies in pregnancy. An online appendix offers recommendations on the treatment of rare hematologic malignancies, including hairy cell leukemia, multiple myeloma, and myelodysplastic syndromes.
Dr. Lishner and nine of his coauthors had no financial relationships to disclose. Three coauthors received honoraria and research funding or are consultants to a wide variety of drug makers.
On Twitter @maryjodales
Consensus guidelines for the perinatal management of hematologic malignancies detected during pregnancy have been issued by a panel of international experts.
The guidelines, published online in the Journal of Clinical Oncology, aim to ensure that timely treatment of the cancers is not delayed in pregnant women (doi: 10.1200/JCO.2015.62.4445).
While rare, hematologic malignancies in pregnancy introduce clinical, social, ethical, and moral dilemmas. Evidence-based data are scarce, according to the researchers, who note the International Network on Cancer, Infertility and Pregnancy registers all cancers occurring during gestation.
“Patient accrual is ongoing and essential, because registration of new cases and long-term follow-up will improve clinical knowledge and increase the level of evidence,” Dr. Michael Lishner of Tel Aviv University and Meir Medical Center, Kfar Saba, Israel, and his fellow panelists wrote.
Hodgkin lymphoma
The researchers note that Hodgkin lymphoma is the most common hematologic cancer in pregnancy, and the prognosis for these patients is excellent. When diagnosed during the first trimester, a regimen based on vinblastine monotherapy has been used. ABVD (doxorubicin, bleomycin, vinblastine, dacarbazine) therapy can be used postpartum and has been used in cases of progression during pregnancy, the panelists wrote.
“The limited data available suggest that ABVD may be administered safely and effectively during the latter phases of pregnancy,” the panel wrote. “Although it may be associated with prematurity and lower birth weights, studies have not reported significant disadvantages.”
Non-Hodgkin lymphoma
The second most common cancer in pregnancy is non-Hodgkin lymphoma. In the case of indolent disease, watchful waiting is possible, with the intent to treat with monoclonal antibodies – with or without chemotherapy – if symptoms or evidence of disease progression are noted. Steroids can be administered during the first trimester as a bridge to the second trimester, when chemotherapy can be used with relatively greater safety, the panelists noted.
Aggressive lymphomas diagnosed before 20 weeks’ gestation warrant pregnancy termination and treatment, they recommend. When diagnosed after 20 weeks, therapy should be comparable to that given a nonpregnant woman, including monoclonal antibodies (R-CHOP).
Chronic myeloid leukemia
Chronic myeloid leukemia occurs in approximately 1 in 100,000 pregnancies and is typically diagnosed during routine blood testing in an asymptomatic patient. As a result, treatment may not be needed until the patient’s white count or platelet count have risen to levels associated with the onset of symptoms. An approximate guideline is a white cell count greater than 100 X 109/L and a platelet count greater than 500 X 109/L.
Therapeutic approaches in pregnancy include interferon-a (INF-a), which does not inhibit DNA synthesis or readily cross the placenta, and leukapheresis, which is frequently required two to three times per week during the first and second trimesters. Counts tend to drop during the third trimester, allowing less frequent intervention.
Consideration should be given to adding aspirin or low-molecular-weight heparin (LMWH) when the platelet count exceeds 1,000 X 109/L.
Myeloproliferative neoplasms
The most common myeloproliferative neoplasm seen in women of childbearing age is essential thrombocytosis.
“A large meta-analysis of pregnant women with essential thrombocytosis reported a live birth rate of 50%-70%, first trimester loss in 25%-40%, late pregnancy loss in 10%, placental abruption in 3.6%, and intrauterine growth restriction in 4.5%. Maternal morbidity is rare, but stroke has been reported,” according to the panelists.
Limited literature suggests similar outcomes for pregnant women with polycythemia vera and primary myelofibrosis.
In low-risk pregnancies, aspirin (75 mg/day) should be offered unless clearly contraindicated. For women with polycythemia vera, venesection may be continued when tolerated to maintain the hematocrit within the gestation-appropriate range.
Fetal ultrasound scans should be performed at 20, 26, and 34 weeks of gestation and uterine artery Doppler should be performed at 20 weeks’ gestation. If the mean pulsatility index exceeds 1.4, the pregnancy may be considered high risk, and treatment and monitoring should be increased.
In high-risk pregnancies, additional treatment includes cytoreductive therapy with or without LMWH. If cytoreductive therapy is required, INF-a should be titrated to maintain a platelet count of less than 400 X 109/L and hematocrit within appropriate range.
Local protocols regarding interruption of LMWH should be adhered to during labor, and dehydration should be avoided. Platelet count and hematocrit may increase postpartum, requiring cytoreductive therapy. Thromboprophylaxis should be considered at 6 weeks’ postpartum because of the increased risk of thrombosis, the guidelines note.
Acute leukemia
“The remarkable anemia, thrombocytopenia, and neutropenia that characterize acute myeloid and lymphoblastic leukemia” require prompt treatment. Leukapheresis in the presence of clinically significant evidence of leukostasis can be considered, regardless of gestational stage. When patients are diagnosed with acute myeloid leukemia during the first trimester, pregnancy termination followed by conventional induction therapy (cytarabine/anthracycline) is recommended.
Those diagnosed later in pregnancy can receive conventional induction therapy, although this seems to be associated with increased risk of fetal growth restriction and even fetal loss. “Notably, neonates rarely experience neutropenia and cardiac impairment unless exposed to lipophilic idarubicin, which should not be used,” the panelists wrote.
When acute promyelocytic leukemia is diagnosed in the first trimester, pregnancy termination is recommended before initiating conventional ATRA-anthracycline therapy. Later in pregnancy, the regimen demonstrates low teratogenicity and can be used in women diagnosed after that stage. Arsenic treatment is highly teratogenic and is prohibited throughout gestation.
Acute lymphocytic leukemia (ALL) requires prophylactic CNS therapy, including methotrexate and L-asparaginase, which are fetotoxic. Methotrexate interferes with organogenesis and is prohibited before week 20 of gestation. L-asparaginase may increase the high risk for thromboembolic events attributed to the combination of pregnancy and malignancy.
Notably, tyrosine kinase inhibitors, essential for patients with Philadelphia chromosome–positive ALL, are teratogenic. Given these limitations, women diagnosed with ALL before 20 weeks’ gestation should undergo termination of the pregnancy and start conventional treatment. After week 20, conventional chemotherapy can be administered during pregnancy. Tyrosine kinase inhibitors can be initiated postpartum.
The guidelines also contain recommendations on diagnostic testing and radiotherapy, maternal supportive care, and perinatal and pediatric aspects of hematologic malignancies in pregnancy. An online appendix offers recommendations on the treatment of rare hematologic malignancies, including hairy cell leukemia, multiple myeloma, and myelodysplastic syndromes.
Dr. Lishner and nine of his coauthors had no financial relationships to disclose. Three coauthors received honoraria and research funding or are consultants to a wide variety of drug makers.
On Twitter @maryjodales
FROM JOURNAL OF CLINICAL ONCOLOGY