User login
Drug receives priority review for FL
The US Food and Drug Administration (FDA) has granted priority review to a supplemental biologics license application (sBLA) for obinutuzumab (Gazyva®).
With this sBLA, Genentech is seeking approval for obinutuzumab to be used, first in combination with chemotherapy and then alone as maintenance, in patients with previously untreated follicular lymphoma (FL).
The FDA grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
The agency’s goal is to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The FDA plans to make a decision on the sBLA for obinutuzumab by December 23, 2017.
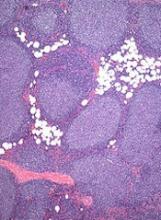
The sBLA is supported by results of the GALLIUM study, which were presented at the 2016 ASH Annual Meeting.
About obinutuzumab
Obinutuzumab is a glycoengineered, humanized, monoclonal antibody that selectively binds to the extracellular domain of the CD20 antigen on B cells.
The drug is FDA-approved for use in combination with chlorambucil to treat patients with previously untreated chronic lymphocytic leukemia.
Obinutuzumab is also approved to treat FL patients who relapse after, or are refractory to, a rituximab-containing regimen. In these patients, obinutuzumab is given first in combination with bendamustine and then alone as maintenance.
The full prescribing information for obinutuzumab is available at http://www.Gazyva.com.
About the GALLIUM study
GALLIUM enrolled 1401 patients with previously untreated, indolent non-Hodgkin lymphoma, including 1202 with FL.
Half of the FL patients (n=601) were randomized to receive obinutuzumab plus chemotherapy (followed by obinutuzumab maintenance for up to 2 years), and half were randomized to rituximab plus chemotherapy (followed by rituximab maintenance for up to 2 years).
The different chemotherapies used were CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisolone), CVP (cyclophosphamide, vincristine, and prednisolone), and bendamustine.
Patients who received obinutuzumab had significantly better progression-free survival than patients who received rituximab. The 3-year progression-free survival rate was 73.3% in the rituximab arm and 80% in the obinutuzumab arm (hazard ratio [HR]=0.66, P=0.0012).
There was no significant difference between the treatment arms with regard to overall survival. The 3-year overall survival was 92.1% in the rituximab arm and 94% in the obinutuzumab arm (HR=0.75, P=0.21).
The overall incidence of adverse events (AEs) was 98.3% in the rituximab arm and 99.5% in the obinutuzumab arm. The incidence of serious AEs was 39.9% and 46.1%, respectively.
The incidence of grade 3 or higher AEs was higher among patients who received obinutuzumab.
Grade 3 or higher AEs occurring in at least 5% of patients in either arm (rituximab and obinutuzumab, respectively) included neutropenia (67.8% and 74.6%), leukopenia (37.9% and 43.9%), febrile neutropenia (4.9% and 6.9%), infections and infestations (3.7% and 6.7%), and thrombocytopenia (2.7% and 6.1%). ![]()
The US Food and Drug Administration (FDA) has granted priority review to a supplemental biologics license application (sBLA) for obinutuzumab (Gazyva®).
With this sBLA, Genentech is seeking approval for obinutuzumab to be used, first in combination with chemotherapy and then alone as maintenance, in patients with previously untreated follicular lymphoma (FL).
The FDA grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
The agency’s goal is to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The FDA plans to make a decision on the sBLA for obinutuzumab by December 23, 2017.
The sBLA is supported by results of the GALLIUM study, which were presented at the 2016 ASH Annual Meeting.
About obinutuzumab
Obinutuzumab is a glycoengineered, humanized, monoclonal antibody that selectively binds to the extracellular domain of the CD20 antigen on B cells.
The drug is FDA-approved for use in combination with chlorambucil to treat patients with previously untreated chronic lymphocytic leukemia.
Obinutuzumab is also approved to treat FL patients who relapse after, or are refractory to, a rituximab-containing regimen. In these patients, obinutuzumab is given first in combination with bendamustine and then alone as maintenance.
The full prescribing information for obinutuzumab is available at http://www.Gazyva.com.
About the GALLIUM study
GALLIUM enrolled 1401 patients with previously untreated, indolent non-Hodgkin lymphoma, including 1202 with FL.
Half of the FL patients (n=601) were randomized to receive obinutuzumab plus chemotherapy (followed by obinutuzumab maintenance for up to 2 years), and half were randomized to rituximab plus chemotherapy (followed by rituximab maintenance for up to 2 years).
The different chemotherapies used were CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisolone), CVP (cyclophosphamide, vincristine, and prednisolone), and bendamustine.
Patients who received obinutuzumab had significantly better progression-free survival than patients who received rituximab. The 3-year progression-free survival rate was 73.3% in the rituximab arm and 80% in the obinutuzumab arm (hazard ratio [HR]=0.66, P=0.0012).
There was no significant difference between the treatment arms with regard to overall survival. The 3-year overall survival was 92.1% in the rituximab arm and 94% in the obinutuzumab arm (HR=0.75, P=0.21).
The overall incidence of adverse events (AEs) was 98.3% in the rituximab arm and 99.5% in the obinutuzumab arm. The incidence of serious AEs was 39.9% and 46.1%, respectively.
The incidence of grade 3 or higher AEs was higher among patients who received obinutuzumab.
Grade 3 or higher AEs occurring in at least 5% of patients in either arm (rituximab and obinutuzumab, respectively) included neutropenia (67.8% and 74.6%), leukopenia (37.9% and 43.9%), febrile neutropenia (4.9% and 6.9%), infections and infestations (3.7% and 6.7%), and thrombocytopenia (2.7% and 6.1%). ![]()
The US Food and Drug Administration (FDA) has granted priority review to a supplemental biologics license application (sBLA) for obinutuzumab (Gazyva®).
With this sBLA, Genentech is seeking approval for obinutuzumab to be used, first in combination with chemotherapy and then alone as maintenance, in patients with previously untreated follicular lymphoma (FL).
The FDA grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
The agency’s goal is to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The FDA plans to make a decision on the sBLA for obinutuzumab by December 23, 2017.
The sBLA is supported by results of the GALLIUM study, which were presented at the 2016 ASH Annual Meeting.
About obinutuzumab
Obinutuzumab is a glycoengineered, humanized, monoclonal antibody that selectively binds to the extracellular domain of the CD20 antigen on B cells.
The drug is FDA-approved for use in combination with chlorambucil to treat patients with previously untreated chronic lymphocytic leukemia.
Obinutuzumab is also approved to treat FL patients who relapse after, or are refractory to, a rituximab-containing regimen. In these patients, obinutuzumab is given first in combination with bendamustine and then alone as maintenance.
The full prescribing information for obinutuzumab is available at http://www.Gazyva.com.
About the GALLIUM study
GALLIUM enrolled 1401 patients with previously untreated, indolent non-Hodgkin lymphoma, including 1202 with FL.
Half of the FL patients (n=601) were randomized to receive obinutuzumab plus chemotherapy (followed by obinutuzumab maintenance for up to 2 years), and half were randomized to rituximab plus chemotherapy (followed by rituximab maintenance for up to 2 years).
The different chemotherapies used were CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisolone), CVP (cyclophosphamide, vincristine, and prednisolone), and bendamustine.
Patients who received obinutuzumab had significantly better progression-free survival than patients who received rituximab. The 3-year progression-free survival rate was 73.3% in the rituximab arm and 80% in the obinutuzumab arm (hazard ratio [HR]=0.66, P=0.0012).
There was no significant difference between the treatment arms with regard to overall survival. The 3-year overall survival was 92.1% in the rituximab arm and 94% in the obinutuzumab arm (HR=0.75, P=0.21).
The overall incidence of adverse events (AEs) was 98.3% in the rituximab arm and 99.5% in the obinutuzumab arm. The incidence of serious AEs was 39.9% and 46.1%, respectively.
The incidence of grade 3 or higher AEs was higher among patients who received obinutuzumab.
Grade 3 or higher AEs occurring in at least 5% of patients in either arm (rituximab and obinutuzumab, respectively) included neutropenia (67.8% and 74.6%), leukopenia (37.9% and 43.9%), febrile neutropenia (4.9% and 6.9%), infections and infestations (3.7% and 6.7%), and thrombocytopenia (2.7% and 6.1%). ![]()
Drug granted breakthrough designation for CTCL
The US Food and Drug Administration (FDA) has granted breakthrough therapy designation to mogamulizumab for the treatment of adults with cutaneous T-cell lymphoma (CTCL) who have received at least 1 prior systemic therapy.
Mogamulizumab is a humanized monoclonal antibody directed against CCR4. It is being developed by Kyowa Hakko Kirin Co., Ltd.
The breakthrough designation for mogamulizumab in CTCL is based on data from the phase 3 MAVORIC study, the largest randomized trial in CTCL.
In MAVORIC, researchers are comparing mogamulizumab and vorinostat in patients with CTCL (both mycosis fungoides and Sézary syndrome) who have failed at least 1 prior systemic treatment.
In April, Kyowa Hakko Kirin announced results from this trial, which showed that patients treated with mogamulizumab have significantly better progression-free survival than patients treated with vorinostat. The company also said mogamulizumab has a tolerable safety profile.
Kyowa Hakko Kirin has not provided any data from MAVORIC but is working with investigators on the future presentation and publication of results from this trial.
Results of a phase 1/2 study of mogamulizumab in previously treated CTCL patients were published in Blood in March 2015.
About breakthrough designation
The FDA’s breakthrough designation is intended to expedite the development and review of new treatments for serious or life-threatening conditions.
The designation entitles the company developing a therapy to more intensive FDA guidance on an efficient and accelerated development program, as well as eligibility for other actions to expedite FDA review, such as rolling submission and priority review.
To earn breakthrough designation, a treatment must show encouraging early clinical results demonstrating substantial improvement over available therapies with regard to a clinically significant endpoint, or it must fulfill an unmet need. ![]()
The US Food and Drug Administration (FDA) has granted breakthrough therapy designation to mogamulizumab for the treatment of adults with cutaneous T-cell lymphoma (CTCL) who have received at least 1 prior systemic therapy.
Mogamulizumab is a humanized monoclonal antibody directed against CCR4. It is being developed by Kyowa Hakko Kirin Co., Ltd.
The breakthrough designation for mogamulizumab in CTCL is based on data from the phase 3 MAVORIC study, the largest randomized trial in CTCL.
In MAVORIC, researchers are comparing mogamulizumab and vorinostat in patients with CTCL (both mycosis fungoides and Sézary syndrome) who have failed at least 1 prior systemic treatment.
In April, Kyowa Hakko Kirin announced results from this trial, which showed that patients treated with mogamulizumab have significantly better progression-free survival than patients treated with vorinostat. The company also said mogamulizumab has a tolerable safety profile.
Kyowa Hakko Kirin has not provided any data from MAVORIC but is working with investigators on the future presentation and publication of results from this trial.
Results of a phase 1/2 study of mogamulizumab in previously treated CTCL patients were published in Blood in March 2015.
About breakthrough designation
The FDA’s breakthrough designation is intended to expedite the development and review of new treatments for serious or life-threatening conditions.
The designation entitles the company developing a therapy to more intensive FDA guidance on an efficient and accelerated development program, as well as eligibility for other actions to expedite FDA review, such as rolling submission and priority review.
To earn breakthrough designation, a treatment must show encouraging early clinical results demonstrating substantial improvement over available therapies with regard to a clinically significant endpoint, or it must fulfill an unmet need. ![]()
The US Food and Drug Administration (FDA) has granted breakthrough therapy designation to mogamulizumab for the treatment of adults with cutaneous T-cell lymphoma (CTCL) who have received at least 1 prior systemic therapy.
Mogamulizumab is a humanized monoclonal antibody directed against CCR4. It is being developed by Kyowa Hakko Kirin Co., Ltd.
The breakthrough designation for mogamulizumab in CTCL is based on data from the phase 3 MAVORIC study, the largest randomized trial in CTCL.
In MAVORIC, researchers are comparing mogamulizumab and vorinostat in patients with CTCL (both mycosis fungoides and Sézary syndrome) who have failed at least 1 prior systemic treatment.
In April, Kyowa Hakko Kirin announced results from this trial, which showed that patients treated with mogamulizumab have significantly better progression-free survival than patients treated with vorinostat. The company also said mogamulizumab has a tolerable safety profile.
Kyowa Hakko Kirin has not provided any data from MAVORIC but is working with investigators on the future presentation and publication of results from this trial.
Results of a phase 1/2 study of mogamulizumab in previously treated CTCL patients were published in Blood in March 2015.
About breakthrough designation
The FDA’s breakthrough designation is intended to expedite the development and review of new treatments for serious or life-threatening conditions.
The designation entitles the company developing a therapy to more intensive FDA guidance on an efficient and accelerated development program, as well as eligibility for other actions to expedite FDA review, such as rolling submission and priority review.
To earn breakthrough designation, a treatment must show encouraging early clinical results demonstrating substantial improvement over available therapies with regard to a clinically significant endpoint, or it must fulfill an unmet need. ![]()
Researchers estimate risk of death from BIA-ALCL
The risk of death from breast implant-associated anaplastic large-cell lymphoma (BIA-ALCL) is less than 1 in a million, according to a study published in Aesthetic Surgery Journal.
Researchers analyzed data on breast implants and estimated the risk of death from BIA-ALCL to be 0.4 micromorts for a woman with bilateral, textured implants.
One micromort means a person’s risk of dying is 1 in a million. For context, a person who drives a car for 1 hour per day is said to have a micromort of 2, which is 5 times the BIA-ALCL micromort.
The researchers noted that there are no documented cases of BIA-ALCL in patients who have only received smooth-surface breast implants. Therefore, the risk of death from BIA-ALCL in a woman with smooth breast implants is “essentially 0.”
“We conducted this micromort study to bring real-life perspective for all existing and potential breast augmentation patients who might have reservations about implants based on the recent media coverage indicating that breast implants can be fatal—a sensationalized take on a very rare and very treatable condition,” said study author William P. Adams, Jr, MD, a professor in the Department of Plastic Surgery at the University of Texas Southwestern in Dallas.
“This analysis resonates with patients. They get it when you explain to a patient that their micromort risk from skiing for 1 day is 2 times higher than the micromort risk of having a textured breast implant for their lifetime—or that traveling 8 hours by car carries a 40-times higher micromort risk than having 2 textured breast implants for their lifetime.”
Dr Adams and his co-author analyzed data from the International Society of Aesthetic Plastic Surgery, the American Society of Plastic Surgeons, the American Society for Aesthetic Plastic Surgery, and the Austrian Breast Implant Register, as well as studies by Allergan and Sientra.
This led to a “conservative estimate” that approximately 30 million patients have textured breast implants worldwide (not including breast reconstructions).
This figure and the report of 12 deaths from BIA-ALCL worldwide suggest the risk of death from BIA-ALCL is 0.4 micromorts per patient (with 2 textured implants) or 0.2 micromorts per textured implant.
“The findings of this study are very important for patient education,” said study author David A. Sieber, MD, a plastic surgeon in private practice in San Francisco, California.
“The clear lymphoproliferative nature of BIA-ALCL, along with the calculated risks associated with its diagnosis, should be used for discussion during new consultations or at the time of presentation for evaluation of delayed-onset seromas.” ![]()
The risk of death from breast implant-associated anaplastic large-cell lymphoma (BIA-ALCL) is less than 1 in a million, according to a study published in Aesthetic Surgery Journal.
Researchers analyzed data on breast implants and estimated the risk of death from BIA-ALCL to be 0.4 micromorts for a woman with bilateral, textured implants.
One micromort means a person’s risk of dying is 1 in a million. For context, a person who drives a car for 1 hour per day is said to have a micromort of 2, which is 5 times the BIA-ALCL micromort.
The researchers noted that there are no documented cases of BIA-ALCL in patients who have only received smooth-surface breast implants. Therefore, the risk of death from BIA-ALCL in a woman with smooth breast implants is “essentially 0.”
“We conducted this micromort study to bring real-life perspective for all existing and potential breast augmentation patients who might have reservations about implants based on the recent media coverage indicating that breast implants can be fatal—a sensationalized take on a very rare and very treatable condition,” said study author William P. Adams, Jr, MD, a professor in the Department of Plastic Surgery at the University of Texas Southwestern in Dallas.
“This analysis resonates with patients. They get it when you explain to a patient that their micromort risk from skiing for 1 day is 2 times higher than the micromort risk of having a textured breast implant for their lifetime—or that traveling 8 hours by car carries a 40-times higher micromort risk than having 2 textured breast implants for their lifetime.”
Dr Adams and his co-author analyzed data from the International Society of Aesthetic Plastic Surgery, the American Society of Plastic Surgeons, the American Society for Aesthetic Plastic Surgery, and the Austrian Breast Implant Register, as well as studies by Allergan and Sientra.
This led to a “conservative estimate” that approximately 30 million patients have textured breast implants worldwide (not including breast reconstructions).
This figure and the report of 12 deaths from BIA-ALCL worldwide suggest the risk of death from BIA-ALCL is 0.4 micromorts per patient (with 2 textured implants) or 0.2 micromorts per textured implant.
“The findings of this study are very important for patient education,” said study author David A. Sieber, MD, a plastic surgeon in private practice in San Francisco, California.
“The clear lymphoproliferative nature of BIA-ALCL, along with the calculated risks associated with its diagnosis, should be used for discussion during new consultations or at the time of presentation for evaluation of delayed-onset seromas.” ![]()
The risk of death from breast implant-associated anaplastic large-cell lymphoma (BIA-ALCL) is less than 1 in a million, according to a study published in Aesthetic Surgery Journal.
Researchers analyzed data on breast implants and estimated the risk of death from BIA-ALCL to be 0.4 micromorts for a woman with bilateral, textured implants.
One micromort means a person’s risk of dying is 1 in a million. For context, a person who drives a car for 1 hour per day is said to have a micromort of 2, which is 5 times the BIA-ALCL micromort.
The researchers noted that there are no documented cases of BIA-ALCL in patients who have only received smooth-surface breast implants. Therefore, the risk of death from BIA-ALCL in a woman with smooth breast implants is “essentially 0.”
“We conducted this micromort study to bring real-life perspective for all existing and potential breast augmentation patients who might have reservations about implants based on the recent media coverage indicating that breast implants can be fatal—a sensationalized take on a very rare and very treatable condition,” said study author William P. Adams, Jr, MD, a professor in the Department of Plastic Surgery at the University of Texas Southwestern in Dallas.
“This analysis resonates with patients. They get it when you explain to a patient that their micromort risk from skiing for 1 day is 2 times higher than the micromort risk of having a textured breast implant for their lifetime—or that traveling 8 hours by car carries a 40-times higher micromort risk than having 2 textured breast implants for their lifetime.”
Dr Adams and his co-author analyzed data from the International Society of Aesthetic Plastic Surgery, the American Society of Plastic Surgeons, the American Society for Aesthetic Plastic Surgery, and the Austrian Breast Implant Register, as well as studies by Allergan and Sientra.
This led to a “conservative estimate” that approximately 30 million patients have textured breast implants worldwide (not including breast reconstructions).
This figure and the report of 12 deaths from BIA-ALCL worldwide suggest the risk of death from BIA-ALCL is 0.4 micromorts per patient (with 2 textured implants) or 0.2 micromorts per textured implant.
“The findings of this study are very important for patient education,” said study author David A. Sieber, MD, a plastic surgeon in private practice in San Francisco, California.
“The clear lymphoproliferative nature of BIA-ALCL, along with the calculated risks associated with its diagnosis, should be used for discussion during new consultations or at the time of presentation for evaluation of delayed-onset seromas.” ![]()
High healthcare costs follow CCSs into adulthood
New research suggests survivors of childhood cancer can incur high out-of-pocket medical costs into adulthood and may forgo healthcare to lessen this financial burden.
The study showed that childhood cancer survivors (CCSs) were more likely than non-CCSs to have out-of-pocket medical costs that were at least 10% of their annual income.
And these high costs were associated with delaying care or skipping it altogether.
These findings were published in the Journal of Clinical Oncology.
“Survivors who reported spending a higher percentage of their income on out-of-pocket medical costs were not only more likely to report financial burden, they also were at risk for undertaking behaviors potentially detrimental to their health in order to save money,” said study author Ryan Nipp, MD, of Massachusetts General Hospital Cancer Center in Boston.
“While studies have identified associations between financial burden and patients’ treatment outcomes, quality of life, and even survival among adults with cancer, as far as we know, this is the first to report these associations in survivors of childhood cancer.”
For this research, Dr Nipp and his colleagues surveyed participants in the Childhood Cancer Survivor Study. This included adults who had been treated for childhood cancers between 1970 and 1986 along with a control group of siblings not affected by cancer.
In 2011 and 2012, participants were asked to provide information about their health insurance, the out-of-pocket healthcare costs they paid during the previous year, and sociodemographic information such as annual income and employment status.
The researchers also asked participants whether medical costs posed a financial burden and, if so, what measures they had taken to deal with that burden.
Study population
The researchers received complete responses from 580 CCSs and 173 of their siblings without a history of cancer. The most common cancer diagnosis was leukemia (33%), followed by Hodgkin lymphoma (14%), while non-Hodgkin lymphoma was less common (7%).
CCSs were a mean of 30.2 years from diagnosis. Use of chemotherapy (77%), radiation (66%), and surgery (81%) were common. Few patients had cancer recurrence (13%) or second cancers (5%).
There was no significant difference between CCSs and their siblings with regard to age at the time of the survey (P=0.071), household income (P=0.053), education (P=0.345), health insurance status (P=0.317), or having at least 1 hospitalization in the past year (P=0.270).
However, CCSs were significantly more likely than siblings to have chronic health conditions (P<0.001). Forty percent of CCSs had severe or life-threatening chronic conditions, compared to 17% of siblings.
Seventy-six percent of CCSs and 80% of siblings were employed. Twenty-nine percent of CCSs and 39% of siblings had household incomes exceeding $100,000. Twelve percent of CCSs and 5% of siblings had household incomes below $20,000.
Ninety-one percent of CCSs and 93% of siblings were insured. Most subjects in both groups (81% and 87%, respectively) had employer-sponsored insurance.
Results
CCSs were significantly more likely than their siblings to have out-of-pocket medical costs that were at least 10% of their annual income—10% and 3%, respectively (P<0.001).
Among CCSs, those with higher out-of-pocket costs (≥10% vs <10% of income) were more likely to have household incomes below $50,000 (odds ratio [OR]=5.5) and to report being hospitalized in the past year (OR=2.3).
CCSs with a higher percentage of their income spent on out-of-pocket costs were also more likely to:
- Have problems paying their medical bills (OR=8.8)
- Report inability to pay for basic costs of living such as food, heat, or rent (OR=6.1)
- Defer healthcare for a medical problem (OR=3.1)
- Skip a test, treatment, or follow-up (OR=2.1)
- Consider filing for bankruptcy (OR=6.4).
“A more comprehensive understanding of the relationship between high out-of-pocket medical costs and the adverse effects of increased financial burden on cancer survivors could be instrumental in helping us identify those at risk for higher costs to help us address their financial challenges and improve health outcomes,” Dr Nipp said.
“It could also help inform policy changes to help meet the unique needs of cancer survivors and improve our understanding of how both higher costs and resulting financial burden influence patients’ approach to their medical care and decision-making.” ![]()
New research suggests survivors of childhood cancer can incur high out-of-pocket medical costs into adulthood and may forgo healthcare to lessen this financial burden.
The study showed that childhood cancer survivors (CCSs) were more likely than non-CCSs to have out-of-pocket medical costs that were at least 10% of their annual income.
And these high costs were associated with delaying care or skipping it altogether.
These findings were published in the Journal of Clinical Oncology.
“Survivors who reported spending a higher percentage of their income on out-of-pocket medical costs were not only more likely to report financial burden, they also were at risk for undertaking behaviors potentially detrimental to their health in order to save money,” said study author Ryan Nipp, MD, of Massachusetts General Hospital Cancer Center in Boston.
“While studies have identified associations between financial burden and patients’ treatment outcomes, quality of life, and even survival among adults with cancer, as far as we know, this is the first to report these associations in survivors of childhood cancer.”
For this research, Dr Nipp and his colleagues surveyed participants in the Childhood Cancer Survivor Study. This included adults who had been treated for childhood cancers between 1970 and 1986 along with a control group of siblings not affected by cancer.
In 2011 and 2012, participants were asked to provide information about their health insurance, the out-of-pocket healthcare costs they paid during the previous year, and sociodemographic information such as annual income and employment status.
The researchers also asked participants whether medical costs posed a financial burden and, if so, what measures they had taken to deal with that burden.
Study population
The researchers received complete responses from 580 CCSs and 173 of their siblings without a history of cancer. The most common cancer diagnosis was leukemia (33%), followed by Hodgkin lymphoma (14%), while non-Hodgkin lymphoma was less common (7%).
CCSs were a mean of 30.2 years from diagnosis. Use of chemotherapy (77%), radiation (66%), and surgery (81%) were common. Few patients had cancer recurrence (13%) or second cancers (5%).
There was no significant difference between CCSs and their siblings with regard to age at the time of the survey (P=0.071), household income (P=0.053), education (P=0.345), health insurance status (P=0.317), or having at least 1 hospitalization in the past year (P=0.270).
However, CCSs were significantly more likely than siblings to have chronic health conditions (P<0.001). Forty percent of CCSs had severe or life-threatening chronic conditions, compared to 17% of siblings.
Seventy-six percent of CCSs and 80% of siblings were employed. Twenty-nine percent of CCSs and 39% of siblings had household incomes exceeding $100,000. Twelve percent of CCSs and 5% of siblings had household incomes below $20,000.
Ninety-one percent of CCSs and 93% of siblings were insured. Most subjects in both groups (81% and 87%, respectively) had employer-sponsored insurance.
Results
CCSs were significantly more likely than their siblings to have out-of-pocket medical costs that were at least 10% of their annual income—10% and 3%, respectively (P<0.001).
Among CCSs, those with higher out-of-pocket costs (≥10% vs <10% of income) were more likely to have household incomes below $50,000 (odds ratio [OR]=5.5) and to report being hospitalized in the past year (OR=2.3).
CCSs with a higher percentage of their income spent on out-of-pocket costs were also more likely to:
- Have problems paying their medical bills (OR=8.8)
- Report inability to pay for basic costs of living such as food, heat, or rent (OR=6.1)
- Defer healthcare for a medical problem (OR=3.1)
- Skip a test, treatment, or follow-up (OR=2.1)
- Consider filing for bankruptcy (OR=6.4).
“A more comprehensive understanding of the relationship between high out-of-pocket medical costs and the adverse effects of increased financial burden on cancer survivors could be instrumental in helping us identify those at risk for higher costs to help us address their financial challenges and improve health outcomes,” Dr Nipp said.
“It could also help inform policy changes to help meet the unique needs of cancer survivors and improve our understanding of how both higher costs and resulting financial burden influence patients’ approach to their medical care and decision-making.” ![]()
New research suggests survivors of childhood cancer can incur high out-of-pocket medical costs into adulthood and may forgo healthcare to lessen this financial burden.
The study showed that childhood cancer survivors (CCSs) were more likely than non-CCSs to have out-of-pocket medical costs that were at least 10% of their annual income.
And these high costs were associated with delaying care or skipping it altogether.
These findings were published in the Journal of Clinical Oncology.
“Survivors who reported spending a higher percentage of their income on out-of-pocket medical costs were not only more likely to report financial burden, they also were at risk for undertaking behaviors potentially detrimental to their health in order to save money,” said study author Ryan Nipp, MD, of Massachusetts General Hospital Cancer Center in Boston.
“While studies have identified associations between financial burden and patients’ treatment outcomes, quality of life, and even survival among adults with cancer, as far as we know, this is the first to report these associations in survivors of childhood cancer.”
For this research, Dr Nipp and his colleagues surveyed participants in the Childhood Cancer Survivor Study. This included adults who had been treated for childhood cancers between 1970 and 1986 along with a control group of siblings not affected by cancer.
In 2011 and 2012, participants were asked to provide information about their health insurance, the out-of-pocket healthcare costs they paid during the previous year, and sociodemographic information such as annual income and employment status.
The researchers also asked participants whether medical costs posed a financial burden and, if so, what measures they had taken to deal with that burden.
Study population
The researchers received complete responses from 580 CCSs and 173 of their siblings without a history of cancer. The most common cancer diagnosis was leukemia (33%), followed by Hodgkin lymphoma (14%), while non-Hodgkin lymphoma was less common (7%).
CCSs were a mean of 30.2 years from diagnosis. Use of chemotherapy (77%), radiation (66%), and surgery (81%) were common. Few patients had cancer recurrence (13%) or second cancers (5%).
There was no significant difference between CCSs and their siblings with regard to age at the time of the survey (P=0.071), household income (P=0.053), education (P=0.345), health insurance status (P=0.317), or having at least 1 hospitalization in the past year (P=0.270).
However, CCSs were significantly more likely than siblings to have chronic health conditions (P<0.001). Forty percent of CCSs had severe or life-threatening chronic conditions, compared to 17% of siblings.
Seventy-six percent of CCSs and 80% of siblings were employed. Twenty-nine percent of CCSs and 39% of siblings had household incomes exceeding $100,000. Twelve percent of CCSs and 5% of siblings had household incomes below $20,000.
Ninety-one percent of CCSs and 93% of siblings were insured. Most subjects in both groups (81% and 87%, respectively) had employer-sponsored insurance.
Results
CCSs were significantly more likely than their siblings to have out-of-pocket medical costs that were at least 10% of their annual income—10% and 3%, respectively (P<0.001).
Among CCSs, those with higher out-of-pocket costs (≥10% vs <10% of income) were more likely to have household incomes below $50,000 (odds ratio [OR]=5.5) and to report being hospitalized in the past year (OR=2.3).
CCSs with a higher percentage of their income spent on out-of-pocket costs were also more likely to:
- Have problems paying their medical bills (OR=8.8)
- Report inability to pay for basic costs of living such as food, heat, or rent (OR=6.1)
- Defer healthcare for a medical problem (OR=3.1)
- Skip a test, treatment, or follow-up (OR=2.1)
- Consider filing for bankruptcy (OR=6.4).
“A more comprehensive understanding of the relationship between high out-of-pocket medical costs and the adverse effects of increased financial burden on cancer survivors could be instrumental in helping us identify those at risk for higher costs to help us address their financial challenges and improve health outcomes,” Dr Nipp said.
“It could also help inform policy changes to help meet the unique needs of cancer survivors and improve our understanding of how both higher costs and resulting financial burden influence patients’ approach to their medical care and decision-making.” ![]()
Drug granted orphan designation for chemo-induced ototoxicity
The US Food and Drug Administration (FDA) has granted orphan drug designation to SENS-401 to be used for the prevention of platinum-induced ototoxicity in pediatric patients.
Platinum-based chemotherapies, particularly cisplatin, can induce severe hearing loss in cancer patients, but there is no pharmaceutical agent approved to treat this side effect.
“Hearing loss in pediatric oncology patients is one of the most frequent side effects of cisplatin treatment and may disable them for the rest of their lives,” said Nawal Ouzren, CEO of Sensorion, the company developing SENS-401.
“Based on its unique profile and the data generated to date, we believe SENS-401 has the potential to be a safe and effective treatment for this serious medical condition where a significant unmet need exists. As such, we look forward to working with the FDA and EMA [European Medicines Agency] to set up an IND [investigational new drug application] and design a phase 2 clinical trial in order to evaluate SENS-401 in this indication.”
About SENS-401
SENS-401 (R-azasetron besylate) is a small molecule intended to protect and preserve inner ear tissue when lesions cause progressive or sequelar hearing impairments. The drug can be taken orally or via an injection.
SENS-401 is one of the two enantiomer forms of SENS-218 (azasetron), a racemic molecule belonging to the family of setrons marketed in Asia under the name Serotone. Pharmacological and pharmacokinetic tests have shown a superior profile for SENS-401 compared with the other enantiomer or the racemic form.
Healthy subjects demonstrated a “very good clinical tolerance” to SENS-401 in a phase 1 study, according to Sensorion. The company is planning to launch a phase 2 trial of the drug for platinum-induced ototoxicity in 2018.
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved. ![]()
The US Food and Drug Administration (FDA) has granted orphan drug designation to SENS-401 to be used for the prevention of platinum-induced ototoxicity in pediatric patients.
Platinum-based chemotherapies, particularly cisplatin, can induce severe hearing loss in cancer patients, but there is no pharmaceutical agent approved to treat this side effect.
“Hearing loss in pediatric oncology patients is one of the most frequent side effects of cisplatin treatment and may disable them for the rest of their lives,” said Nawal Ouzren, CEO of Sensorion, the company developing SENS-401.
“Based on its unique profile and the data generated to date, we believe SENS-401 has the potential to be a safe and effective treatment for this serious medical condition where a significant unmet need exists. As such, we look forward to working with the FDA and EMA [European Medicines Agency] to set up an IND [investigational new drug application] and design a phase 2 clinical trial in order to evaluate SENS-401 in this indication.”
About SENS-401
SENS-401 (R-azasetron besylate) is a small molecule intended to protect and preserve inner ear tissue when lesions cause progressive or sequelar hearing impairments. The drug can be taken orally or via an injection.
SENS-401 is one of the two enantiomer forms of SENS-218 (azasetron), a racemic molecule belonging to the family of setrons marketed in Asia under the name Serotone. Pharmacological and pharmacokinetic tests have shown a superior profile for SENS-401 compared with the other enantiomer or the racemic form.
Healthy subjects demonstrated a “very good clinical tolerance” to SENS-401 in a phase 1 study, according to Sensorion. The company is planning to launch a phase 2 trial of the drug for platinum-induced ototoxicity in 2018.
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved. ![]()
The US Food and Drug Administration (FDA) has granted orphan drug designation to SENS-401 to be used for the prevention of platinum-induced ototoxicity in pediatric patients.
Platinum-based chemotherapies, particularly cisplatin, can induce severe hearing loss in cancer patients, but there is no pharmaceutical agent approved to treat this side effect.
“Hearing loss in pediatric oncology patients is one of the most frequent side effects of cisplatin treatment and may disable them for the rest of their lives,” said Nawal Ouzren, CEO of Sensorion, the company developing SENS-401.
“Based on its unique profile and the data generated to date, we believe SENS-401 has the potential to be a safe and effective treatment for this serious medical condition where a significant unmet need exists. As such, we look forward to working with the FDA and EMA [European Medicines Agency] to set up an IND [investigational new drug application] and design a phase 2 clinical trial in order to evaluate SENS-401 in this indication.”
About SENS-401
SENS-401 (R-azasetron besylate) is a small molecule intended to protect and preserve inner ear tissue when lesions cause progressive or sequelar hearing impairments. The drug can be taken orally or via an injection.
SENS-401 is one of the two enantiomer forms of SENS-218 (azasetron), a racemic molecule belonging to the family of setrons marketed in Asia under the name Serotone. Pharmacological and pharmacokinetic tests have shown a superior profile for SENS-401 compared with the other enantiomer or the racemic form.
Healthy subjects demonstrated a “very good clinical tolerance” to SENS-401 in a phase 1 study, according to Sensorion. The company is planning to launch a phase 2 trial of the drug for platinum-induced ototoxicity in 2018.
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved. ![]()
Antibody could treat AML, MM, and NHL
An investigational antibody has demonstrated activity against acute myeloid leukemia (AML), multiple myeloma (MM), and non-Hodgkin lymphoma (NHL), according to researchers.
PF-06747143 is a humanized CXCR4 immunoglobulin G1 (IgG1) antibody that binds to CXCR4 and inhibits both CXCL12-mediated signaling pathways and cell migration.
Whether given alone or in combination with chemotherapy, PF-06747143 demonstrated efficacy in mouse models of NHL, MM, and AML.
Treatment involving PF-06747143—alone or in combination—eradicated more cancer cells than did standard treatment options.
These results were published in Blood Advances. The research was sponsored by Pfizer, Inc., the company developing PF-06747143.
“One of the major limitations we see in treating blood cancers is the failure to clear cancer cells from the bone marrow,” said study author Flavia Pernasetti, PhD, of Pfizer Oncology Research and Development.
“Because the bone marrow allows the cancer cells to flourish, removing these cells is an essential step in treating these malignancies effectively.”
With this goal in mind, Dr Pernasetti and her colleagues looked to the mechanisms that control the movement of cells into the bone marrow (BM) in the first place—the chemokine receptor CXCR4 and its ligand CXCL12.
The researchers created PF-06747143, which attacks and kills cancer cells directly but also removes cancer cells from the BM so they can be killed by other treatments.
Results in NHL
The researchers first tested PF-06747143 in an NHL Ramos xenograft model. Mice received PF-06747143 or a control IgG1 antibody at 10 mg/kg on days 1 and 8.
PF-06747143 significantly inhibited tumor growth compared to the control antibody (P<0.0001). Seventy percent of PF-06747143-treated mice had tumor volumes below their initial size at the end of the study.
PF-06747143 produced a dose-dependent response that was sustained until the end of the study, even after treatment was stopped.
Results in MM
The researchers tested PF-06747143 in a disseminated MM model, in which the OPM2-Luc tumor cells were implanted intravenously and migrated spontaneously to the BM.
Mice received PF-06747143 or IgG1 control at 10 mg/kg weekly for 5 doses. Other mice received melphalan at 1 mg/kg twice a week for a total of 4 cycles.
On day 30, PF-06747143 had significantly inhibited BM tumor growth compared to the control antibody or melphalan (P<0.0001).
PF-06747143-treated mice also had a significant survival benefit. The median survival was 33.5 days for mice that received the control antibody and 36 days for mice treated with melphalan. However, there were no deaths in the PF-06747143-treated mice by day 50, which marked the end of the study (P<0.0001).
The researchers also tested PF-06747143 at a lower dose (1 mg/kg weekly for a total of 7 doses), both alone and in combination with bortezomib (0.5 mg/kg twice a week for a total of 4 cycles).
The median survival was 34 days in the control mice, 44 days in mice that received bortezomib alone, and 47 days in mice that received PF-06747143 alone. However, there were no deaths in the combination arm at day 51, which was the end of the study (P<0.0003).
Results in AML
The researchers tested PF-06747143 in an AML disseminated tumor model using MV4-11 cells.
They compared PF-06747143 (given at 0.1, 1, or 10 mg/kg weekly for 4 doses) to the chemotherapeutic agent daunorubicin (2 mg/kg on days 1, 3, and 5), the FLT3 inhibitor crenolanib (7.5 mg/kg twice a day, on days 11-15 and 25-29), and a control IgG1 antibody (10 mg/kg weekly for 4 doses).
PF-06747143 (at 10 mg/kg), daunorubicin, and crenolanib all significantly reduced the number of tumor cells in the peripheral blood and BM when compared with the control antibody (P<0.05).
PF-06746143 treatment (at 10 mg/kg) reduced the number of AML cells in the BM by 95.9%, while daunorubicin reduced them by 84.5% and crenolanib by 80.5%.
The median survival was 36 days for mice that received PF-06747143 at 0.1 mg/kg, 41 days for mice that received the control antibody, 47 days for mice that received PF-06747143 at 1 mg/kg, and 63 days for mice that received PF-06747143 at 10 mg/kg.
The researchers also found that PF-06747143 had a “strong combinatorial effect” with daunorubicin and cytarabine in a chemotherapy-resistant model of AML. The team noted that only 36% of BM cells are positive for CXCR4 in this model.
Treatment with PF-06747143 alone reduced the percentage of AML cells in the BM to 80%, combination daunorubicin and cytarabine reduced it to 27%, and combination PF-06747143, daunorubicin, and cytarabine reduced the percentage of AML cells in the BM to 0.3%.
“Our preliminary preclinical results are encouraging,” Dr Pernasetti said, “and we are very excited to see how our antibody fares in clinical testing.”
PF-06747143 is currently being evaluated in a phase 1 trial of AML patients. ![]()
An investigational antibody has demonstrated activity against acute myeloid leukemia (AML), multiple myeloma (MM), and non-Hodgkin lymphoma (NHL), according to researchers.
PF-06747143 is a humanized CXCR4 immunoglobulin G1 (IgG1) antibody that binds to CXCR4 and inhibits both CXCL12-mediated signaling pathways and cell migration.
Whether given alone or in combination with chemotherapy, PF-06747143 demonstrated efficacy in mouse models of NHL, MM, and AML.
Treatment involving PF-06747143—alone or in combination—eradicated more cancer cells than did standard treatment options.
These results were published in Blood Advances. The research was sponsored by Pfizer, Inc., the company developing PF-06747143.
“One of the major limitations we see in treating blood cancers is the failure to clear cancer cells from the bone marrow,” said study author Flavia Pernasetti, PhD, of Pfizer Oncology Research and Development.
“Because the bone marrow allows the cancer cells to flourish, removing these cells is an essential step in treating these malignancies effectively.”
With this goal in mind, Dr Pernasetti and her colleagues looked to the mechanisms that control the movement of cells into the bone marrow (BM) in the first place—the chemokine receptor CXCR4 and its ligand CXCL12.
The researchers created PF-06747143, which attacks and kills cancer cells directly but also removes cancer cells from the BM so they can be killed by other treatments.
Results in NHL
The researchers first tested PF-06747143 in an NHL Ramos xenograft model. Mice received PF-06747143 or a control IgG1 antibody at 10 mg/kg on days 1 and 8.
PF-06747143 significantly inhibited tumor growth compared to the control antibody (P<0.0001). Seventy percent of PF-06747143-treated mice had tumor volumes below their initial size at the end of the study.
PF-06747143 produced a dose-dependent response that was sustained until the end of the study, even after treatment was stopped.
Results in MM
The researchers tested PF-06747143 in a disseminated MM model, in which the OPM2-Luc tumor cells were implanted intravenously and migrated spontaneously to the BM.
Mice received PF-06747143 or IgG1 control at 10 mg/kg weekly for 5 doses. Other mice received melphalan at 1 mg/kg twice a week for a total of 4 cycles.
On day 30, PF-06747143 had significantly inhibited BM tumor growth compared to the control antibody or melphalan (P<0.0001).
PF-06747143-treated mice also had a significant survival benefit. The median survival was 33.5 days for mice that received the control antibody and 36 days for mice treated with melphalan. However, there were no deaths in the PF-06747143-treated mice by day 50, which marked the end of the study (P<0.0001).
The researchers also tested PF-06747143 at a lower dose (1 mg/kg weekly for a total of 7 doses), both alone and in combination with bortezomib (0.5 mg/kg twice a week for a total of 4 cycles).
The median survival was 34 days in the control mice, 44 days in mice that received bortezomib alone, and 47 days in mice that received PF-06747143 alone. However, there were no deaths in the combination arm at day 51, which was the end of the study (P<0.0003).
Results in AML
The researchers tested PF-06747143 in an AML disseminated tumor model using MV4-11 cells.
They compared PF-06747143 (given at 0.1, 1, or 10 mg/kg weekly for 4 doses) to the chemotherapeutic agent daunorubicin (2 mg/kg on days 1, 3, and 5), the FLT3 inhibitor crenolanib (7.5 mg/kg twice a day, on days 11-15 and 25-29), and a control IgG1 antibody (10 mg/kg weekly for 4 doses).
PF-06747143 (at 10 mg/kg), daunorubicin, and crenolanib all significantly reduced the number of tumor cells in the peripheral blood and BM when compared with the control antibody (P<0.05).
PF-06746143 treatment (at 10 mg/kg) reduced the number of AML cells in the BM by 95.9%, while daunorubicin reduced them by 84.5% and crenolanib by 80.5%.
The median survival was 36 days for mice that received PF-06747143 at 0.1 mg/kg, 41 days for mice that received the control antibody, 47 days for mice that received PF-06747143 at 1 mg/kg, and 63 days for mice that received PF-06747143 at 10 mg/kg.
The researchers also found that PF-06747143 had a “strong combinatorial effect” with daunorubicin and cytarabine in a chemotherapy-resistant model of AML. The team noted that only 36% of BM cells are positive for CXCR4 in this model.
Treatment with PF-06747143 alone reduced the percentage of AML cells in the BM to 80%, combination daunorubicin and cytarabine reduced it to 27%, and combination PF-06747143, daunorubicin, and cytarabine reduced the percentage of AML cells in the BM to 0.3%.
“Our preliminary preclinical results are encouraging,” Dr Pernasetti said, “and we are very excited to see how our antibody fares in clinical testing.”
PF-06747143 is currently being evaluated in a phase 1 trial of AML patients. ![]()
An investigational antibody has demonstrated activity against acute myeloid leukemia (AML), multiple myeloma (MM), and non-Hodgkin lymphoma (NHL), according to researchers.
PF-06747143 is a humanized CXCR4 immunoglobulin G1 (IgG1) antibody that binds to CXCR4 and inhibits both CXCL12-mediated signaling pathways and cell migration.
Whether given alone or in combination with chemotherapy, PF-06747143 demonstrated efficacy in mouse models of NHL, MM, and AML.
Treatment involving PF-06747143—alone or in combination—eradicated more cancer cells than did standard treatment options.
These results were published in Blood Advances. The research was sponsored by Pfizer, Inc., the company developing PF-06747143.
“One of the major limitations we see in treating blood cancers is the failure to clear cancer cells from the bone marrow,” said study author Flavia Pernasetti, PhD, of Pfizer Oncology Research and Development.
“Because the bone marrow allows the cancer cells to flourish, removing these cells is an essential step in treating these malignancies effectively.”
With this goal in mind, Dr Pernasetti and her colleagues looked to the mechanisms that control the movement of cells into the bone marrow (BM) in the first place—the chemokine receptor CXCR4 and its ligand CXCL12.
The researchers created PF-06747143, which attacks and kills cancer cells directly but also removes cancer cells from the BM so they can be killed by other treatments.
Results in NHL
The researchers first tested PF-06747143 in an NHL Ramos xenograft model. Mice received PF-06747143 or a control IgG1 antibody at 10 mg/kg on days 1 and 8.
PF-06747143 significantly inhibited tumor growth compared to the control antibody (P<0.0001). Seventy percent of PF-06747143-treated mice had tumor volumes below their initial size at the end of the study.
PF-06747143 produced a dose-dependent response that was sustained until the end of the study, even after treatment was stopped.
Results in MM
The researchers tested PF-06747143 in a disseminated MM model, in which the OPM2-Luc tumor cells were implanted intravenously and migrated spontaneously to the BM.
Mice received PF-06747143 or IgG1 control at 10 mg/kg weekly for 5 doses. Other mice received melphalan at 1 mg/kg twice a week for a total of 4 cycles.
On day 30, PF-06747143 had significantly inhibited BM tumor growth compared to the control antibody or melphalan (P<0.0001).
PF-06747143-treated mice also had a significant survival benefit. The median survival was 33.5 days for mice that received the control antibody and 36 days for mice treated with melphalan. However, there were no deaths in the PF-06747143-treated mice by day 50, which marked the end of the study (P<0.0001).
The researchers also tested PF-06747143 at a lower dose (1 mg/kg weekly for a total of 7 doses), both alone and in combination with bortezomib (0.5 mg/kg twice a week for a total of 4 cycles).
The median survival was 34 days in the control mice, 44 days in mice that received bortezomib alone, and 47 days in mice that received PF-06747143 alone. However, there were no deaths in the combination arm at day 51, which was the end of the study (P<0.0003).
Results in AML
The researchers tested PF-06747143 in an AML disseminated tumor model using MV4-11 cells.
They compared PF-06747143 (given at 0.1, 1, or 10 mg/kg weekly for 4 doses) to the chemotherapeutic agent daunorubicin (2 mg/kg on days 1, 3, and 5), the FLT3 inhibitor crenolanib (7.5 mg/kg twice a day, on days 11-15 and 25-29), and a control IgG1 antibody (10 mg/kg weekly for 4 doses).
PF-06747143 (at 10 mg/kg), daunorubicin, and crenolanib all significantly reduced the number of tumor cells in the peripheral blood and BM when compared with the control antibody (P<0.05).
PF-06746143 treatment (at 10 mg/kg) reduced the number of AML cells in the BM by 95.9%, while daunorubicin reduced them by 84.5% and crenolanib by 80.5%.
The median survival was 36 days for mice that received PF-06747143 at 0.1 mg/kg, 41 days for mice that received the control antibody, 47 days for mice that received PF-06747143 at 1 mg/kg, and 63 days for mice that received PF-06747143 at 10 mg/kg.
The researchers also found that PF-06747143 had a “strong combinatorial effect” with daunorubicin and cytarabine in a chemotherapy-resistant model of AML. The team noted that only 36% of BM cells are positive for CXCR4 in this model.
Treatment with PF-06747143 alone reduced the percentage of AML cells in the BM to 80%, combination daunorubicin and cytarabine reduced it to 27%, and combination PF-06747143, daunorubicin, and cytarabine reduced the percentage of AML cells in the BM to 0.3%.
“Our preliminary preclinical results are encouraging,” Dr Pernasetti said, “and we are very excited to see how our antibody fares in clinical testing.”
PF-06747143 is currently being evaluated in a phase 1 trial of AML patients. ![]()
Drug granted priority review for CTCL
The US Food and Drug Administration (FDA) has granted priority review to a supplemental biologics license application (sBLA) seeking approval for brentuximab vedotin (Adcetris) as a treatment for cutaneous T-cell lymphoma (CTCL).
The FDA grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
The agency’s goal is to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The FDA plans to make a decision on the sBLA for brentuximab vedotin by December 16, 2017.
The sBLA is supported by data from the phase 3 ALCANZA trial and a pair of phase 2 investigator-sponsored trials.
About brentuximab vedotin
Brentuximab vedotin is an antibody-drug conjugate directed to CD30, which is expressed on skin lesions in approximately 50% of patients with CTCL. The drug is being developed by Seattle Genetics and Takeda Pharmaceutical Company Limited.
Brentuximab vedotin is currently FDA-approved for 3 indications.
The drug is approved to treat patients with classical Hodgkin lymphoma after failure of autologous hematopoietic stem cell transplant (auto-HSCT) or after failure of at least 2 prior multi-agent chemotherapy regimens in patients who are not auto-HSCT candidates. Brentuximab vedotin initially had accelerated approval for this indication, but it was later converted to full approval.
Brentuximab vedotin also has full approval as consolidation for patients with classical Hodgkin lymphoma who have a high risk of relapse or progression after auto-HSCT.
And the drug has accelerated approval for the treatment of patients with systemic anaplastic large-cell lymphoma (sALCL) after failure of at least 1 prior multi-agent chemotherapy regimen. This accelerated approval is based on overall response rate. Continued approval for the sALCL indication may be contingent upon verification and description of clinical benefit in confirmatory trials.
Brentuximab vedotin previously received breakthrough therapy designation from the FDA for the treatment of patients with CD30-expressing mycosis fungoides (MF) and patients with primary cutaneous ALCL who require systemic therapy and have received 1 prior systemic therapy.
Brentuximab vedotin also has orphan drug designation from the FDA for the treatment of MF. ![]()
The US Food and Drug Administration (FDA) has granted priority review to a supplemental biologics license application (sBLA) seeking approval for brentuximab vedotin (Adcetris) as a treatment for cutaneous T-cell lymphoma (CTCL).
The FDA grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
The agency’s goal is to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The FDA plans to make a decision on the sBLA for brentuximab vedotin by December 16, 2017.
The sBLA is supported by data from the phase 3 ALCANZA trial and a pair of phase 2 investigator-sponsored trials.
About brentuximab vedotin
Brentuximab vedotin is an antibody-drug conjugate directed to CD30, which is expressed on skin lesions in approximately 50% of patients with CTCL. The drug is being developed by Seattle Genetics and Takeda Pharmaceutical Company Limited.
Brentuximab vedotin is currently FDA-approved for 3 indications.
The drug is approved to treat patients with classical Hodgkin lymphoma after failure of autologous hematopoietic stem cell transplant (auto-HSCT) or after failure of at least 2 prior multi-agent chemotherapy regimens in patients who are not auto-HSCT candidates. Brentuximab vedotin initially had accelerated approval for this indication, but it was later converted to full approval.
Brentuximab vedotin also has full approval as consolidation for patients with classical Hodgkin lymphoma who have a high risk of relapse or progression after auto-HSCT.
And the drug has accelerated approval for the treatment of patients with systemic anaplastic large-cell lymphoma (sALCL) after failure of at least 1 prior multi-agent chemotherapy regimen. This accelerated approval is based on overall response rate. Continued approval for the sALCL indication may be contingent upon verification and description of clinical benefit in confirmatory trials.
Brentuximab vedotin previously received breakthrough therapy designation from the FDA for the treatment of patients with CD30-expressing mycosis fungoides (MF) and patients with primary cutaneous ALCL who require systemic therapy and have received 1 prior systemic therapy.
Brentuximab vedotin also has orphan drug designation from the FDA for the treatment of MF. ![]()
The US Food and Drug Administration (FDA) has granted priority review to a supplemental biologics license application (sBLA) seeking approval for brentuximab vedotin (Adcetris) as a treatment for cutaneous T-cell lymphoma (CTCL).
The FDA grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
The agency’s goal is to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The FDA plans to make a decision on the sBLA for brentuximab vedotin by December 16, 2017.
The sBLA is supported by data from the phase 3 ALCANZA trial and a pair of phase 2 investigator-sponsored trials.
About brentuximab vedotin
Brentuximab vedotin is an antibody-drug conjugate directed to CD30, which is expressed on skin lesions in approximately 50% of patients with CTCL. The drug is being developed by Seattle Genetics and Takeda Pharmaceutical Company Limited.
Brentuximab vedotin is currently FDA-approved for 3 indications.
The drug is approved to treat patients with classical Hodgkin lymphoma after failure of autologous hematopoietic stem cell transplant (auto-HSCT) or after failure of at least 2 prior multi-agent chemotherapy regimens in patients who are not auto-HSCT candidates. Brentuximab vedotin initially had accelerated approval for this indication, but it was later converted to full approval.
Brentuximab vedotin also has full approval as consolidation for patients with classical Hodgkin lymphoma who have a high risk of relapse or progression after auto-HSCT.
And the drug has accelerated approval for the treatment of patients with systemic anaplastic large-cell lymphoma (sALCL) after failure of at least 1 prior multi-agent chemotherapy regimen. This accelerated approval is based on overall response rate. Continued approval for the sALCL indication may be contingent upon verification and description of clinical benefit in confirmatory trials.
Brentuximab vedotin previously received breakthrough therapy designation from the FDA for the treatment of patients with CD30-expressing mycosis fungoides (MF) and patients with primary cutaneous ALCL who require systemic therapy and have received 1 prior systemic therapy.
Brentuximab vedotin also has orphan drug designation from the FDA for the treatment of MF.
New cancer diagnosis linked to arterial thromboembolism
Patients newly diagnosed with cancer may have a short-term increased risk of arterial thromboembolism, according to a new study.
The research showed that, within 6 months of their diagnosis, cancer patients had a rate of arterial thromboembolism that was more than double the rate in matched control patients without cancer.
However, the risk of arterial thromboembolism varied by cancer type.
Babak B. Navi, MD, of Weill Cornell Medicine in New York, New York, and his colleagues reported these findings in the Journal of the American College of Cardiology.
The researchers used the Surveillance Epidemiology and End Results–Medicare linked database to identify patients with a new primary diagnosis of breast, lung, prostate, colorectal, bladder, pancreatic, or gastric cancer or non-Hodgkin lymphoma from 2002 to 2011.
The team matched these patients (by demographics and comorbidities) to Medicare enrollees without cancer, collecting data on 279,719 pairs of subjects. The subjects were followed through 2012.
Arterial thromboembolism
The study’s primary outcome was the cumulative incidence of arterial thromboembolism, defined as any inpatient or outpatient diagnosis of myocardial infarction or ischemic stroke.
The incidence of arterial thromboembolism at 3 months was 3.4% in cancer patients and 1.1% in controls. At 6 months, it was 4.7% and 2.2%, respectively. At 1 year, it was 6.5% and 4.2%, respectively. And at 2 years, it was 9.1% and 8.1%, respectively.
The hazard ratios (HRs) for arterial thromboembolism among cancer patients were 5.2 at 0 to 1 month, 2.1 at 1 to 3 months, 1.4 at 3 to 6 months, 1.1 at 6 to 9 months, and 1.1 at 9 to 12 months.
The risk of arterial thromboembolism varied by cancer type, with the greatest excess risk observed in lung cancer. The 6-month cumulative incidence was 8.3% in lung cancer patients and 2.4% in matched controls (P<0.001).
In patients with non-Hodgkin lymphoma, the 6-month cumulative incidence of arterial thromboembolism was 5.4%, compared to 2.2% in matched controls (P<0.001).
Myocardial infarction
The cumulative incidence of myocardial infarction at 3 months was 1.4% in cancer patients and 0.3% in controls.
At 6 months, it was 2.0% and 0.7%, respectively. At 1 year, it was 2.6% and 1.4%, respectively. And at 2 years, it was 3.7% and 2.8%, respectively.
The HRs for myocardial infarction among cancer patients were 7.3 at 0 to 1 month, 3.0 at 1 to 3 months, 1.8 at 3 to 6 months, 1.3 at 6 to 9 months, and 1.0 at 9 to 12 months.
Ischemic stroke
The cumulative incidence of ischemic stroke at 3 months was 2.1% in cancer patients and 0.8% in controls.
At 6 months, it was 3.0% and 1.6%, respectively. At 1 year, it was 4.3% and 3.1%, respectively. And at 2 years, it was 6.1% and 5.8%, respectively.
The HRs for ischemic stroke among cancer patients were 4.5 at 0 to 1 month, 1.7 at 1 to 3 months, 1.3 at 3 to 6 months, 1.0 at 6 to 9 months, and 1.1 at 9 to 12 months.
The researchers said these findings raise the question of whether patients with newly diagnosed cancer should be considered for antithrombotic and statin medicines for primary prevention of cardiovascular disease.
The team stressed that because patients with cancer are also prone to bleeding due to frequent coagulopathy and invasive procedures, carefully designed clinical trials are needed to answer these questions.
Patients newly diagnosed with cancer may have a short-term increased risk of arterial thromboembolism, according to a new study.
The research showed that, within 6 months of their diagnosis, cancer patients had a rate of arterial thromboembolism that was more than double the rate in matched control patients without cancer.
However, the risk of arterial thromboembolism varied by cancer type.
Babak B. Navi, MD, of Weill Cornell Medicine in New York, New York, and his colleagues reported these findings in the Journal of the American College of Cardiology.
The researchers used the Surveillance Epidemiology and End Results–Medicare linked database to identify patients with a new primary diagnosis of breast, lung, prostate, colorectal, bladder, pancreatic, or gastric cancer or non-Hodgkin lymphoma from 2002 to 2011.
The team matched these patients (by demographics and comorbidities) to Medicare enrollees without cancer, collecting data on 279,719 pairs of subjects. The subjects were followed through 2012.
Arterial thromboembolism
The study’s primary outcome was the cumulative incidence of arterial thromboembolism, defined as any inpatient or outpatient diagnosis of myocardial infarction or ischemic stroke.
The incidence of arterial thromboembolism at 3 months was 3.4% in cancer patients and 1.1% in controls. At 6 months, it was 4.7% and 2.2%, respectively. At 1 year, it was 6.5% and 4.2%, respectively. And at 2 years, it was 9.1% and 8.1%, respectively.
The hazard ratios (HRs) for arterial thromboembolism among cancer patients were 5.2 at 0 to 1 month, 2.1 at 1 to 3 months, 1.4 at 3 to 6 months, 1.1 at 6 to 9 months, and 1.1 at 9 to 12 months.
The risk of arterial thromboembolism varied by cancer type, with the greatest excess risk observed in lung cancer. The 6-month cumulative incidence was 8.3% in lung cancer patients and 2.4% in matched controls (P<0.001).
In patients with non-Hodgkin lymphoma, the 6-month cumulative incidence of arterial thromboembolism was 5.4%, compared to 2.2% in matched controls (P<0.001).
Myocardial infarction
The cumulative incidence of myocardial infarction at 3 months was 1.4% in cancer patients and 0.3% in controls.
At 6 months, it was 2.0% and 0.7%, respectively. At 1 year, it was 2.6% and 1.4%, respectively. And at 2 years, it was 3.7% and 2.8%, respectively.
The HRs for myocardial infarction among cancer patients were 7.3 at 0 to 1 month, 3.0 at 1 to 3 months, 1.8 at 3 to 6 months, 1.3 at 6 to 9 months, and 1.0 at 9 to 12 months.
Ischemic stroke
The cumulative incidence of ischemic stroke at 3 months was 2.1% in cancer patients and 0.8% in controls.
At 6 months, it was 3.0% and 1.6%, respectively. At 1 year, it was 4.3% and 3.1%, respectively. And at 2 years, it was 6.1% and 5.8%, respectively.
The HRs for ischemic stroke among cancer patients were 4.5 at 0 to 1 month, 1.7 at 1 to 3 months, 1.3 at 3 to 6 months, 1.0 at 6 to 9 months, and 1.1 at 9 to 12 months.
The researchers said these findings raise the question of whether patients with newly diagnosed cancer should be considered for antithrombotic and statin medicines for primary prevention of cardiovascular disease.
The team stressed that because patients with cancer are also prone to bleeding due to frequent coagulopathy and invasive procedures, carefully designed clinical trials are needed to answer these questions.
Patients newly diagnosed with cancer may have a short-term increased risk of arterial thromboembolism, according to a new study.
The research showed that, within 6 months of their diagnosis, cancer patients had a rate of arterial thromboembolism that was more than double the rate in matched control patients without cancer.
However, the risk of arterial thromboembolism varied by cancer type.
Babak B. Navi, MD, of Weill Cornell Medicine in New York, New York, and his colleagues reported these findings in the Journal of the American College of Cardiology.
The researchers used the Surveillance Epidemiology and End Results–Medicare linked database to identify patients with a new primary diagnosis of breast, lung, prostate, colorectal, bladder, pancreatic, or gastric cancer or non-Hodgkin lymphoma from 2002 to 2011.
The team matched these patients (by demographics and comorbidities) to Medicare enrollees without cancer, collecting data on 279,719 pairs of subjects. The subjects were followed through 2012.
Arterial thromboembolism
The study’s primary outcome was the cumulative incidence of arterial thromboembolism, defined as any inpatient or outpatient diagnosis of myocardial infarction or ischemic stroke.
The incidence of arterial thromboembolism at 3 months was 3.4% in cancer patients and 1.1% in controls. At 6 months, it was 4.7% and 2.2%, respectively. At 1 year, it was 6.5% and 4.2%, respectively. And at 2 years, it was 9.1% and 8.1%, respectively.
The hazard ratios (HRs) for arterial thromboembolism among cancer patients were 5.2 at 0 to 1 month, 2.1 at 1 to 3 months, 1.4 at 3 to 6 months, 1.1 at 6 to 9 months, and 1.1 at 9 to 12 months.
The risk of arterial thromboembolism varied by cancer type, with the greatest excess risk observed in lung cancer. The 6-month cumulative incidence was 8.3% in lung cancer patients and 2.4% in matched controls (P<0.001).
In patients with non-Hodgkin lymphoma, the 6-month cumulative incidence of arterial thromboembolism was 5.4%, compared to 2.2% in matched controls (P<0.001).
Myocardial infarction
The cumulative incidence of myocardial infarction at 3 months was 1.4% in cancer patients and 0.3% in controls.
At 6 months, it was 2.0% and 0.7%, respectively. At 1 year, it was 2.6% and 1.4%, respectively. And at 2 years, it was 3.7% and 2.8%, respectively.
The HRs for myocardial infarction among cancer patients were 7.3 at 0 to 1 month, 3.0 at 1 to 3 months, 1.8 at 3 to 6 months, 1.3 at 6 to 9 months, and 1.0 at 9 to 12 months.
Ischemic stroke
The cumulative incidence of ischemic stroke at 3 months was 2.1% in cancer patients and 0.8% in controls.
At 6 months, it was 3.0% and 1.6%, respectively. At 1 year, it was 4.3% and 3.1%, respectively. And at 2 years, it was 6.1% and 5.8%, respectively.
The HRs for ischemic stroke among cancer patients were 4.5 at 0 to 1 month, 1.7 at 1 to 3 months, 1.3 at 3 to 6 months, 1.0 at 6 to 9 months, and 1.1 at 9 to 12 months.
The researchers said these findings raise the question of whether patients with newly diagnosed cancer should be considered for antithrombotic and statin medicines for primary prevention of cardiovascular disease.
The team stressed that because patients with cancer are also prone to bleeding due to frequent coagulopathy and invasive procedures, carefully designed clinical trials are needed to answer these questions.
Analysis reveals poor outcomes in refractory DLBCL
Results from the SCHOLAR-1 study revealed poor outcomes of salvage therapy in patients with refractory diffuse large B-cell lymphoma (DLBCL).
This retrospective study included data on patients enrolled in 2 randomized trials and 2 academic databases.
The patients had primary refractory disease, were refractory to second-line or later therapy, or had relapsed within 12 months of autologous stem cell transplant (ASCT).
Twenty-six percent of patients responded to salvage therapy, with 7% achieving a complete response (CR).
The median overall survival (OS) was 6.3 months, and 20% of patients were still alive at 2 years’ follow-up.
Christian Gisselbrecht, MD, of Saint Louis Hospital in Paris, France, and his colleagues reported these findings in Blood. SCHOLAR-1 was funded through an unrestricted grant from Kite Pharma.
“SCHOLAR-1 demonstrates the uniformly poor treatment outcomes for patients with aggressive non-Hodgkin lymphoma and emphasizes the need for breakthrough therapies for these refractory patients,” Dr Gisselbrecht said.
Patient characteristics
The study included pooled, patient-level data from 2 phase 3 trials and 2 databases:
- The Canadian Cancer Trials Group study LY.12 (n=219)
- The Lymphoma Academic Research Organization’s CORAL study (n=170)
- A cohort from MD Anderson Cancer Center (n=165)
- A cohort from the Molecular Epidemiology Resource of the University of Iowa/Mayo Clinic Lymphoma Specialized Program of Research Excellence (n=82).
There were a total of 636 patients who met criteria for refractory DLBCL, which included primary mediastinal B-cell lymphoma and transformed follicular lymphoma.
Twenty-eight percent of patients were primary refractory, 50% were refractory to second-line or later therapy, and 22% had relapsed within 12 months of transplant.
The patients’ median age was 55 (range, 19-81), and 64% were male. Seventy-three percent had an ECOG performance status of 0-1, 14% had a status of 2-4, and 13% were missing this data. Seventy-two percent of patients had stage III-IV disease, 27% had stage I-II disease, and less than 1% were missing this data.
Treatments
The MD Anderson cohort included patients who were relapsed/refractory to initial rituximab-containing chemotherapy, had failed salvage platinum-containing chemotherapy, and received a second salvage therapy at MD Anderson.
The University of Iowa/Mayo Clinic cohort included unselected, newly diagnosed patients with lymphoma who entered prospective documentation of primary and subsequent treatments and outcomes.
In the LY.12 study, patients were enrolled upon relapse after anthracycline-containing therapy and randomized to 1 of 2 salvage regimens, with a goal of consolidative ASCT.
The CORAL study enrolled patients in their first relapse or whose lymphoma was refractory to first-line therapy. They were randomized to 1 of 2 salvage regimens, with a goal of consolidative ASCT.
In the LY.12 and CORAL studies, eligible patients with CD20+ lymphoma were randomized to rituximab maintenance or observation post-ASCT.
Response
In all, 523 patients were evaluated for response. The overall response rate (ORR) was 26%, with a 7% CR rate and an 18% partial response rate.
Among patients with primary refractory disease, the ORR was 20%, and the CR rate was 3%.
Among patients who were refractory to second-line or later therapy, the ORR was 26%, and the CR rate was 10%.
Among patients who relapsed after transplant, the ORR was 34%, and the CR rate was 15%.
Survival
A total of 603 patients were evaluated for survival.
The median OS from the start of salvage therapy was 6.3 months (range, 5.9-7.0). The 1-year OS rate was 28%, and the 2-year OS was 20%.
Among primary refractory patients, the median OS was 7.1 months (range, 6.0-8.1), 1-year OS was 29%, and 2-year OS was 24%.
Among patient who were refractory to second-line or later therapy, the median OS was 6.1 months (range, 5.2-7.0), 1-year OS was 26%, and 2-year OS was 17%.
Among patients who relapsed after transplant, the median OS was 6.2 months (range, 5.2-7.6), 1-year OS was 32%, and 2-year OS was 19%.
“Although 60% to 70% of non-Hodgkin lymphoma patients survive 5 years after rituximab-based chemotherapy and autologous stem cell transplant, nearly half of them either do not respond or relapse shortly after transplant,” Dr Gisselbrecht noted.
“SCHOLAR-1 provides a rigorous measure of outcomes for these patients who do not benefit from currently available therapies, and this landmark study will serve as an important historical control for evaluating new therapeutic candidates in the field of non-Hodgkin lymphoma.”
Results from the SCHOLAR-1 study revealed poor outcomes of salvage therapy in patients with refractory diffuse large B-cell lymphoma (DLBCL).
This retrospective study included data on patients enrolled in 2 randomized trials and 2 academic databases.
The patients had primary refractory disease, were refractory to second-line or later therapy, or had relapsed within 12 months of autologous stem cell transplant (ASCT).
Twenty-six percent of patients responded to salvage therapy, with 7% achieving a complete response (CR).
The median overall survival (OS) was 6.3 months, and 20% of patients were still alive at 2 years’ follow-up.
Christian Gisselbrecht, MD, of Saint Louis Hospital in Paris, France, and his colleagues reported these findings in Blood. SCHOLAR-1 was funded through an unrestricted grant from Kite Pharma.
“SCHOLAR-1 demonstrates the uniformly poor treatment outcomes for patients with aggressive non-Hodgkin lymphoma and emphasizes the need for breakthrough therapies for these refractory patients,” Dr Gisselbrecht said.
Patient characteristics
The study included pooled, patient-level data from 2 phase 3 trials and 2 databases:
- The Canadian Cancer Trials Group study LY.12 (n=219)
- The Lymphoma Academic Research Organization’s CORAL study (n=170)
- A cohort from MD Anderson Cancer Center (n=165)
- A cohort from the Molecular Epidemiology Resource of the University of Iowa/Mayo Clinic Lymphoma Specialized Program of Research Excellence (n=82).
There were a total of 636 patients who met criteria for refractory DLBCL, which included primary mediastinal B-cell lymphoma and transformed follicular lymphoma.
Twenty-eight percent of patients were primary refractory, 50% were refractory to second-line or later therapy, and 22% had relapsed within 12 months of transplant.
The patients’ median age was 55 (range, 19-81), and 64% were male. Seventy-three percent had an ECOG performance status of 0-1, 14% had a status of 2-4, and 13% were missing this data. Seventy-two percent of patients had stage III-IV disease, 27% had stage I-II disease, and less than 1% were missing this data.
Treatments
The MD Anderson cohort included patients who were relapsed/refractory to initial rituximab-containing chemotherapy, had failed salvage platinum-containing chemotherapy, and received a second salvage therapy at MD Anderson.
The University of Iowa/Mayo Clinic cohort included unselected, newly diagnosed patients with lymphoma who entered prospective documentation of primary and subsequent treatments and outcomes.
In the LY.12 study, patients were enrolled upon relapse after anthracycline-containing therapy and randomized to 1 of 2 salvage regimens, with a goal of consolidative ASCT.
The CORAL study enrolled patients in their first relapse or whose lymphoma was refractory to first-line therapy. They were randomized to 1 of 2 salvage regimens, with a goal of consolidative ASCT.
In the LY.12 and CORAL studies, eligible patients with CD20+ lymphoma were randomized to rituximab maintenance or observation post-ASCT.
Response
In all, 523 patients were evaluated for response. The overall response rate (ORR) was 26%, with a 7% CR rate and an 18% partial response rate.
Among patients with primary refractory disease, the ORR was 20%, and the CR rate was 3%.
Among patients who were refractory to second-line or later therapy, the ORR was 26%, and the CR rate was 10%.
Among patients who relapsed after transplant, the ORR was 34%, and the CR rate was 15%.
Survival
A total of 603 patients were evaluated for survival.
The median OS from the start of salvage therapy was 6.3 months (range, 5.9-7.0). The 1-year OS rate was 28%, and the 2-year OS was 20%.
Among primary refractory patients, the median OS was 7.1 months (range, 6.0-8.1), 1-year OS was 29%, and 2-year OS was 24%.
Among patient who were refractory to second-line or later therapy, the median OS was 6.1 months (range, 5.2-7.0), 1-year OS was 26%, and 2-year OS was 17%.
Among patients who relapsed after transplant, the median OS was 6.2 months (range, 5.2-7.6), 1-year OS was 32%, and 2-year OS was 19%.
“Although 60% to 70% of non-Hodgkin lymphoma patients survive 5 years after rituximab-based chemotherapy and autologous stem cell transplant, nearly half of them either do not respond or relapse shortly after transplant,” Dr Gisselbrecht noted.
“SCHOLAR-1 provides a rigorous measure of outcomes for these patients who do not benefit from currently available therapies, and this landmark study will serve as an important historical control for evaluating new therapeutic candidates in the field of non-Hodgkin lymphoma.”
Results from the SCHOLAR-1 study revealed poor outcomes of salvage therapy in patients with refractory diffuse large B-cell lymphoma (DLBCL).
This retrospective study included data on patients enrolled in 2 randomized trials and 2 academic databases.
The patients had primary refractory disease, were refractory to second-line or later therapy, or had relapsed within 12 months of autologous stem cell transplant (ASCT).
Twenty-six percent of patients responded to salvage therapy, with 7% achieving a complete response (CR).
The median overall survival (OS) was 6.3 months, and 20% of patients were still alive at 2 years’ follow-up.
Christian Gisselbrecht, MD, of Saint Louis Hospital in Paris, France, and his colleagues reported these findings in Blood. SCHOLAR-1 was funded through an unrestricted grant from Kite Pharma.
“SCHOLAR-1 demonstrates the uniformly poor treatment outcomes for patients with aggressive non-Hodgkin lymphoma and emphasizes the need for breakthrough therapies for these refractory patients,” Dr Gisselbrecht said.
Patient characteristics
The study included pooled, patient-level data from 2 phase 3 trials and 2 databases:
- The Canadian Cancer Trials Group study LY.12 (n=219)
- The Lymphoma Academic Research Organization’s CORAL study (n=170)
- A cohort from MD Anderson Cancer Center (n=165)
- A cohort from the Molecular Epidemiology Resource of the University of Iowa/Mayo Clinic Lymphoma Specialized Program of Research Excellence (n=82).
There were a total of 636 patients who met criteria for refractory DLBCL, which included primary mediastinal B-cell lymphoma and transformed follicular lymphoma.
Twenty-eight percent of patients were primary refractory, 50% were refractory to second-line or later therapy, and 22% had relapsed within 12 months of transplant.
The patients’ median age was 55 (range, 19-81), and 64% were male. Seventy-three percent had an ECOG performance status of 0-1, 14% had a status of 2-4, and 13% were missing this data. Seventy-two percent of patients had stage III-IV disease, 27% had stage I-II disease, and less than 1% were missing this data.
Treatments
The MD Anderson cohort included patients who were relapsed/refractory to initial rituximab-containing chemotherapy, had failed salvage platinum-containing chemotherapy, and received a second salvage therapy at MD Anderson.
The University of Iowa/Mayo Clinic cohort included unselected, newly diagnosed patients with lymphoma who entered prospective documentation of primary and subsequent treatments and outcomes.
In the LY.12 study, patients were enrolled upon relapse after anthracycline-containing therapy and randomized to 1 of 2 salvage regimens, with a goal of consolidative ASCT.
The CORAL study enrolled patients in their first relapse or whose lymphoma was refractory to first-line therapy. They were randomized to 1 of 2 salvage regimens, with a goal of consolidative ASCT.
In the LY.12 and CORAL studies, eligible patients with CD20+ lymphoma were randomized to rituximab maintenance or observation post-ASCT.
Response
In all, 523 patients were evaluated for response. The overall response rate (ORR) was 26%, with a 7% CR rate and an 18% partial response rate.
Among patients with primary refractory disease, the ORR was 20%, and the CR rate was 3%.
Among patients who were refractory to second-line or later therapy, the ORR was 26%, and the CR rate was 10%.
Among patients who relapsed after transplant, the ORR was 34%, and the CR rate was 15%.
Survival
A total of 603 patients were evaluated for survival.
The median OS from the start of salvage therapy was 6.3 months (range, 5.9-7.0). The 1-year OS rate was 28%, and the 2-year OS was 20%.
Among primary refractory patients, the median OS was 7.1 months (range, 6.0-8.1), 1-year OS was 29%, and 2-year OS was 24%.
Among patient who were refractory to second-line or later therapy, the median OS was 6.1 months (range, 5.2-7.0), 1-year OS was 26%, and 2-year OS was 17%.
Among patients who relapsed after transplant, the median OS was 6.2 months (range, 5.2-7.6), 1-year OS was 32%, and 2-year OS was 19%.
“Although 60% to 70% of non-Hodgkin lymphoma patients survive 5 years after rituximab-based chemotherapy and autologous stem cell transplant, nearly half of them either do not respond or relapse shortly after transplant,” Dr Gisselbrecht noted.
“SCHOLAR-1 provides a rigorous measure of outcomes for these patients who do not benefit from currently available therapies, and this landmark study will serve as an important historical control for evaluating new therapeutic candidates in the field of non-Hodgkin lymphoma.”
FDA grants acalabrutinib breakthrough designation
The US Food and Drug Administration (FDA) has granted breakthrough therapy designation to acalabrutinib, a BTK inhibitor being developed to treat multiple B-cell malignancies.
The breakthrough designation applies to acalabrutinib as a treatment for patients with mantle cell lymphoma (MCL) who have received at least 1 prior therapy.
The FDA’s breakthrough designation is intended to expedite the development and review of new treatments for serious or life-threatening conditions.
The designation entitles the company developing a therapy to more intensive FDA guidance on an efficient and accelerated development program, as well as eligibility for other actions to expedite FDA review, such as a rolling submission and priority review.
To earn breakthrough designation, a treatment must show encouraging early clinical results demonstrating substantial improvement over available therapies with regard to a clinically significant endpoint, or it must fulfill an unmet need.
The FDA granted acalabrutinib breakthrough designation based on data from the drug’s development program, which includes data from the phase 2 ACE-LY-004 trial in patients with relapsed or refractory MCL.
Data from this trial have not yet been released to the public but are expected to be presented at an upcoming medical meeting.
The US Food and Drug Administration (FDA) has granted breakthrough therapy designation to acalabrutinib, a BTK inhibitor being developed to treat multiple B-cell malignancies.
The breakthrough designation applies to acalabrutinib as a treatment for patients with mantle cell lymphoma (MCL) who have received at least 1 prior therapy.
The FDA’s breakthrough designation is intended to expedite the development and review of new treatments for serious or life-threatening conditions.
The designation entitles the company developing a therapy to more intensive FDA guidance on an efficient and accelerated development program, as well as eligibility for other actions to expedite FDA review, such as a rolling submission and priority review.
To earn breakthrough designation, a treatment must show encouraging early clinical results demonstrating substantial improvement over available therapies with regard to a clinically significant endpoint, or it must fulfill an unmet need.
The FDA granted acalabrutinib breakthrough designation based on data from the drug’s development program, which includes data from the phase 2 ACE-LY-004 trial in patients with relapsed or refractory MCL.
Data from this trial have not yet been released to the public but are expected to be presented at an upcoming medical meeting.
The US Food and Drug Administration (FDA) has granted breakthrough therapy designation to acalabrutinib, a BTK inhibitor being developed to treat multiple B-cell malignancies.
The breakthrough designation applies to acalabrutinib as a treatment for patients with mantle cell lymphoma (MCL) who have received at least 1 prior therapy.
The FDA’s breakthrough designation is intended to expedite the development and review of new treatments for serious or life-threatening conditions.
The designation entitles the company developing a therapy to more intensive FDA guidance on an efficient and accelerated development program, as well as eligibility for other actions to expedite FDA review, such as a rolling submission and priority review.
To earn breakthrough designation, a treatment must show encouraging early clinical results demonstrating substantial improvement over available therapies with regard to a clinically significant endpoint, or it must fulfill an unmet need.
The FDA granted acalabrutinib breakthrough designation based on data from the drug’s development program, which includes data from the phase 2 ACE-LY-004 trial in patients with relapsed or refractory MCL.
Data from this trial have not yet been released to the public but are expected to be presented at an upcoming medical meeting.