User login
Family history impacts risk of second cancer after HL
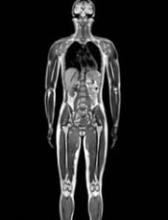
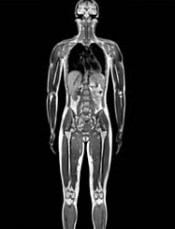
A new study suggests Hodgkin lymphoma (HL) survivors have a high risk of developing a second malignancy, particularly if they have a family history of that malignancy.
The research showed that HL survivors in Sweden were roughly 2.4 times more likely than individuals in the country’s general population to develop a second cancer.
The risk for HL survivors remained high 30 years after treatment, and the risk was even greater in HL survivors who had a family history of specific cancers.
“The vast majority of patients with Hodgkin lymphoma are cured with a combination of chemotherapy and radiotherapy,” said study author Amit Sud, MBChB, of The Institute of Cancer Research, London in the UK.
“Our research has shown that these patients are at substantially increased risk of a second cancer later in life and particularly if they have a family history of cancer.”
Dr Sud and his colleagues described this research in the Journal of Clinical Oncology.
The team analyzed data from the Swedish Family-Cancer Project Database. They identified 9522 HL patients diagnosed between 1965 and 2013. During a median follow-up of 12.6 years, there were 1215 second cancers in 1121 HL patients (12%).
Compared to the general population, the HL patients had a significantly higher risk of all second malignancies, with a standardized incident ratio (SIR) of 2.39 and an absolute excess risk of 71.2 cases per 10,000 person-years.
Cancer types
HL patients had a significantly increased risk of several malignancies. The overall SIRs were as follows:
- NHL—7.99
- Leukemia—6.46
- Connective tissue cancer—5.73
- Thyroid cancer—5.13
- Squamous cell carcinoma—4.44
- Lung cancer—3.61
- Pharyngeal cancer—3.52
- Esophageal cancer—2.62
- Brain cancer—2.58
- Breast cancer—2.52
- Colon cancer—2.21
- Pancreatic cancer—2.09
- Melanoma—2.08
- Colorectal cancer—1.85
- Stomach cancer—1.78
- Bladder cancer—1.57
- Prostate cancer—1.21.
The researchers calculated SIRs over time and found the risk for many of the cancers remained high over 30 years following HL treatment.
Family history
The researchers identified 28,277 first-degree relatives of the HL survivors. Thirty percent of HL survivors (n=2785) had 1 or more first-degree relatives with a family history of cancer.
The SIR for cancers was 1.02 in the relatives. The SIR for second cancers was 2.83 for HL survivors who had first-degree relatives with cancer and 2.16 for HL survivors who did not have any first-degree relatives with cancer.
The researchers said the increased risk of second malignancy was correlated with the number of first-degree relatives with cancer.
The SIR was 2.67 for HL patients who had a single first-degree relative with cancer and 3.40 for HL patients who had 2 or more first-degree relatives with cancer.
The SIRs for different cancer types (for HL patients with at least 1 first-degree relative with cancer and no first-degree relatives with cancer, respectively) were as follows:
- NHL—14.43 vs 7.83
- Leukemia—14.31 vs 6.37
- Squamous cell carcinoma—10.85 vs 4.30
- Lung cancer—11.24 vs 3.39
- Breast cancer—4.36 vs 2.36
- Colorectal cancer—3.71 vs 1.76.
Sex and age
The researchers found significant differences in the SIRs for second cancers between HL patients diagnosed before the age of 35 and those diagnosed after age 35.
For men, the SIRs were:
- All cancers—4.26 for <35, 2.08 for ≥ 35
- Colorectal cancer—4.07 for < 35, 1.73 for ≥35
- Lung cancer—6.16 for < 35, 3.20 for ≥35
- Breast cancer—12.60 for < 35, 4.58 for ≥35
- Squamous cell carcinoma—5.89 for < 35, 3.96 for ≥35
- NHL—15.9 for < 35, 6.93 for ≥35
- Leukemia—12.15 for < 35, 5.57 for ≥35.
For women, the SIRs were:
- All cancers—4.61 for <35, 1.73 for ≥ 35
- Colorectal cancer—1.31 for < 35, 1.65 for ≥35
- Lung cancer—8.84 for < 35, 2.50 for ≥35
- Breast cancer—6.00 for < 35, 1.14 for ≥35
- Squamous cell carcinoma—6.37 for < 35, 4.87 for ≥35
- NHL—6.23 for < 35, 6.55 for ≥35
- Leukemia—10.36 for < 35, 4.51 for ≥35.
“Younger women who have been treated with radiotherapy to the chest for Hodgkin lymphoma are already screened for breast cancer, but our study suggests that we should be looking at ways of monitoring survivors for other forms of cancer too, and potentially offering preventative interventions,” Dr Sud said.
“After patients are cured, they no longer encounter oncologists, so it’s important that other healthcare providers are aware of the increased risk to Hodgkin lymphoma survivors to improve early diagnosis of second cancers.” ![]()
A new study suggests Hodgkin lymphoma (HL) survivors have a high risk of developing a second malignancy, particularly if they have a family history of that malignancy.
The research showed that HL survivors in Sweden were roughly 2.4 times more likely than individuals in the country’s general population to develop a second cancer.
The risk for HL survivors remained high 30 years after treatment, and the risk was even greater in HL survivors who had a family history of specific cancers.
“The vast majority of patients with Hodgkin lymphoma are cured with a combination of chemotherapy and radiotherapy,” said study author Amit Sud, MBChB, of The Institute of Cancer Research, London in the UK.
“Our research has shown that these patients are at substantially increased risk of a second cancer later in life and particularly if they have a family history of cancer.”
Dr Sud and his colleagues described this research in the Journal of Clinical Oncology.
The team analyzed data from the Swedish Family-Cancer Project Database. They identified 9522 HL patients diagnosed between 1965 and 2013. During a median follow-up of 12.6 years, there were 1215 second cancers in 1121 HL patients (12%).
Compared to the general population, the HL patients had a significantly higher risk of all second malignancies, with a standardized incident ratio (SIR) of 2.39 and an absolute excess risk of 71.2 cases per 10,000 person-years.
Cancer types
HL patients had a significantly increased risk of several malignancies. The overall SIRs were as follows:
- NHL—7.99
- Leukemia—6.46
- Connective tissue cancer—5.73
- Thyroid cancer—5.13
- Squamous cell carcinoma—4.44
- Lung cancer—3.61
- Pharyngeal cancer—3.52
- Esophageal cancer—2.62
- Brain cancer—2.58
- Breast cancer—2.52
- Colon cancer—2.21
- Pancreatic cancer—2.09
- Melanoma—2.08
- Colorectal cancer—1.85
- Stomach cancer—1.78
- Bladder cancer—1.57
- Prostate cancer—1.21.
The researchers calculated SIRs over time and found the risk for many of the cancers remained high over 30 years following HL treatment.
Family history
The researchers identified 28,277 first-degree relatives of the HL survivors. Thirty percent of HL survivors (n=2785) had 1 or more first-degree relatives with a family history of cancer.
The SIR for cancers was 1.02 in the relatives. The SIR for second cancers was 2.83 for HL survivors who had first-degree relatives with cancer and 2.16 for HL survivors who did not have any first-degree relatives with cancer.
The researchers said the increased risk of second malignancy was correlated with the number of first-degree relatives with cancer.
The SIR was 2.67 for HL patients who had a single first-degree relative with cancer and 3.40 for HL patients who had 2 or more first-degree relatives with cancer.
The SIRs for different cancer types (for HL patients with at least 1 first-degree relative with cancer and no first-degree relatives with cancer, respectively) were as follows:
- NHL—14.43 vs 7.83
- Leukemia—14.31 vs 6.37
- Squamous cell carcinoma—10.85 vs 4.30
- Lung cancer—11.24 vs 3.39
- Breast cancer—4.36 vs 2.36
- Colorectal cancer—3.71 vs 1.76.
Sex and age
The researchers found significant differences in the SIRs for second cancers between HL patients diagnosed before the age of 35 and those diagnosed after age 35.
For men, the SIRs were:
- All cancers—4.26 for <35, 2.08 for ≥ 35
- Colorectal cancer—4.07 for < 35, 1.73 for ≥35
- Lung cancer—6.16 for < 35, 3.20 for ≥35
- Breast cancer—12.60 for < 35, 4.58 for ≥35
- Squamous cell carcinoma—5.89 for < 35, 3.96 for ≥35
- NHL—15.9 for < 35, 6.93 for ≥35
- Leukemia—12.15 for < 35, 5.57 for ≥35.
For women, the SIRs were:
- All cancers—4.61 for <35, 1.73 for ≥ 35
- Colorectal cancer—1.31 for < 35, 1.65 for ≥35
- Lung cancer—8.84 for < 35, 2.50 for ≥35
- Breast cancer—6.00 for < 35, 1.14 for ≥35
- Squamous cell carcinoma—6.37 for < 35, 4.87 for ≥35
- NHL—6.23 for < 35, 6.55 for ≥35
- Leukemia—10.36 for < 35, 4.51 for ≥35.
“Younger women who have been treated with radiotherapy to the chest for Hodgkin lymphoma are already screened for breast cancer, but our study suggests that we should be looking at ways of monitoring survivors for other forms of cancer too, and potentially offering preventative interventions,” Dr Sud said.
“After patients are cured, they no longer encounter oncologists, so it’s important that other healthcare providers are aware of the increased risk to Hodgkin lymphoma survivors to improve early diagnosis of second cancers.” ![]()
A new study suggests Hodgkin lymphoma (HL) survivors have a high risk of developing a second malignancy, particularly if they have a family history of that malignancy.
The research showed that HL survivors in Sweden were roughly 2.4 times more likely than individuals in the country’s general population to develop a second cancer.
The risk for HL survivors remained high 30 years after treatment, and the risk was even greater in HL survivors who had a family history of specific cancers.
“The vast majority of patients with Hodgkin lymphoma are cured with a combination of chemotherapy and radiotherapy,” said study author Amit Sud, MBChB, of The Institute of Cancer Research, London in the UK.
“Our research has shown that these patients are at substantially increased risk of a second cancer later in life and particularly if they have a family history of cancer.”
Dr Sud and his colleagues described this research in the Journal of Clinical Oncology.
The team analyzed data from the Swedish Family-Cancer Project Database. They identified 9522 HL patients diagnosed between 1965 and 2013. During a median follow-up of 12.6 years, there were 1215 second cancers in 1121 HL patients (12%).
Compared to the general population, the HL patients had a significantly higher risk of all second malignancies, with a standardized incident ratio (SIR) of 2.39 and an absolute excess risk of 71.2 cases per 10,000 person-years.
Cancer types
HL patients had a significantly increased risk of several malignancies. The overall SIRs were as follows:
- NHL—7.99
- Leukemia—6.46
- Connective tissue cancer—5.73
- Thyroid cancer—5.13
- Squamous cell carcinoma—4.44
- Lung cancer—3.61
- Pharyngeal cancer—3.52
- Esophageal cancer—2.62
- Brain cancer—2.58
- Breast cancer—2.52
- Colon cancer—2.21
- Pancreatic cancer—2.09
- Melanoma—2.08
- Colorectal cancer—1.85
- Stomach cancer—1.78
- Bladder cancer—1.57
- Prostate cancer—1.21.
The researchers calculated SIRs over time and found the risk for many of the cancers remained high over 30 years following HL treatment.
Family history
The researchers identified 28,277 first-degree relatives of the HL survivors. Thirty percent of HL survivors (n=2785) had 1 or more first-degree relatives with a family history of cancer.
The SIR for cancers was 1.02 in the relatives. The SIR for second cancers was 2.83 for HL survivors who had first-degree relatives with cancer and 2.16 for HL survivors who did not have any first-degree relatives with cancer.
The researchers said the increased risk of second malignancy was correlated with the number of first-degree relatives with cancer.
The SIR was 2.67 for HL patients who had a single first-degree relative with cancer and 3.40 for HL patients who had 2 or more first-degree relatives with cancer.
The SIRs for different cancer types (for HL patients with at least 1 first-degree relative with cancer and no first-degree relatives with cancer, respectively) were as follows:
- NHL—14.43 vs 7.83
- Leukemia—14.31 vs 6.37
- Squamous cell carcinoma—10.85 vs 4.30
- Lung cancer—11.24 vs 3.39
- Breast cancer—4.36 vs 2.36
- Colorectal cancer—3.71 vs 1.76.
Sex and age
The researchers found significant differences in the SIRs for second cancers between HL patients diagnosed before the age of 35 and those diagnosed after age 35.
For men, the SIRs were:
- All cancers—4.26 for <35, 2.08 for ≥ 35
- Colorectal cancer—4.07 for < 35, 1.73 for ≥35
- Lung cancer—6.16 for < 35, 3.20 for ≥35
- Breast cancer—12.60 for < 35, 4.58 for ≥35
- Squamous cell carcinoma—5.89 for < 35, 3.96 for ≥35
- NHL—15.9 for < 35, 6.93 for ≥35
- Leukemia—12.15 for < 35, 5.57 for ≥35.
For women, the SIRs were:
- All cancers—4.61 for <35, 1.73 for ≥ 35
- Colorectal cancer—1.31 for < 35, 1.65 for ≥35
- Lung cancer—8.84 for < 35, 2.50 for ≥35
- Breast cancer—6.00 for < 35, 1.14 for ≥35
- Squamous cell carcinoma—6.37 for < 35, 4.87 for ≥35
- NHL—6.23 for < 35, 6.55 for ≥35
- Leukemia—10.36 for < 35, 4.51 for ≥35.
“Younger women who have been treated with radiotherapy to the chest for Hodgkin lymphoma are already screened for breast cancer, but our study suggests that we should be looking at ways of monitoring survivors for other forms of cancer too, and potentially offering preventative interventions,” Dr Sud said.
“After patients are cured, they no longer encounter oncologists, so it’s important that other healthcare providers are aware of the increased risk to Hodgkin lymphoma survivors to improve early diagnosis of second cancers.” ![]()
Selinexor trials placed on partial hold
The US Food and Drug Administration (FDA) has placed a partial clinical hold on all trials of selinexor (KPT-330).
Selinexor is an inhibitor being evaluated in multiple trials of patients with relapsed and/or refractory hematologic and solid tumor malignancies.
While the partial clinical hold remains in effect, patients with stable disease or better may remain on selinexor.
However, no new patients may be enrolled in selinexor trials until the hold is lifted.
The FDA has indicated that the partial clinical hold is due to incomplete information in the existing version of the investigator’s brochure, including an incomplete list of serious adverse events associated with selinexor.
Karyopharm Therapeutics Inc., the company developing selinexor, said it has amended the brochure, updated the informed consent documents accordingly, and submitted the documents to the FDA as requested.
As of March 10, Karyopharm had provided all requested materials to the FDA believed to be required to lift the partial clinical hold. By regulation, the FDA has 30 days from the receipt of Karyopharm’s submission to notify the company whether the partial clinical hold is lifted.
Karyopharm said it is working with the FDA to seek the release of the hold and resume enrollment in its selinexor trials as expeditiously as possible. The company believes its previously disclosed enrollment rates and timelines for its ongoing trials will remain materially unchanged.
About selinexor
Selinexor is a selective inhibitor of nuclear export (SINE) XPO1 antagonist. The drug binds with and inhibits XPO1, leading to the accumulation of tumor suppressor proteins in the cell nucleus. This reinitiates and amplifies their tumor suppressor function and is believed to induce apoptosis in cancer cells while largely sparing normal cells.
To date, more than 1900 patients have been treated with selinexor. The drug is currently being evaluated in several trials across multiple cancer indications.
One of these is the phase 2 SOPRA trial, in which selinexor is being compared to investigator’s choice of therapy (1 of 3 potential salvage therapies). The trial is enrolling patients 60 years of age or older with relapsed or refractory acute myeloid leukemia who are ineligible for standard intensive chemotherapy and/or transplant.
The SADAL study is a phase 2b trial comparing high and low doses of selinexor in patients with relapsed and/or refractory de novo diffuse large B-cell lymphoma who have no therapeutic options of demonstrated clinical benefit.
STORM is a phase 2b trial evaluating selinexor and low-dose dexamethasone in patients with heavily pretreated multiple myeloma (MM). And STOMP is a phase 1b/2 study evaluating selinexor in combination with existing therapies across the broader population in MM.
Karyopharm is also planning a randomized, phase 3 study known as BOSTON. In this trial, researchers will compare selinexor plus bortezomib and low-dose dexamethasone to bortezomib and low-dose dexamethasone in MM patients who have had 1 to 3 prior lines of therapy.
Additional phase 1, 2, and 3 studies are ongoing or currently planned.
The US Food and Drug Administration (FDA) has placed a partial clinical hold on all trials of selinexor (KPT-330).
Selinexor is an inhibitor being evaluated in multiple trials of patients with relapsed and/or refractory hematologic and solid tumor malignancies.
While the partial clinical hold remains in effect, patients with stable disease or better may remain on selinexor.
However, no new patients may be enrolled in selinexor trials until the hold is lifted.
The FDA has indicated that the partial clinical hold is due to incomplete information in the existing version of the investigator’s brochure, including an incomplete list of serious adverse events associated with selinexor.
Karyopharm Therapeutics Inc., the company developing selinexor, said it has amended the brochure, updated the informed consent documents accordingly, and submitted the documents to the FDA as requested.
As of March 10, Karyopharm had provided all requested materials to the FDA believed to be required to lift the partial clinical hold. By regulation, the FDA has 30 days from the receipt of Karyopharm’s submission to notify the company whether the partial clinical hold is lifted.
Karyopharm said it is working with the FDA to seek the release of the hold and resume enrollment in its selinexor trials as expeditiously as possible. The company believes its previously disclosed enrollment rates and timelines for its ongoing trials will remain materially unchanged.
About selinexor
Selinexor is a selective inhibitor of nuclear export (SINE) XPO1 antagonist. The drug binds with and inhibits XPO1, leading to the accumulation of tumor suppressor proteins in the cell nucleus. This reinitiates and amplifies their tumor suppressor function and is believed to induce apoptosis in cancer cells while largely sparing normal cells.
To date, more than 1900 patients have been treated with selinexor. The drug is currently being evaluated in several trials across multiple cancer indications.
One of these is the phase 2 SOPRA trial, in which selinexor is being compared to investigator’s choice of therapy (1 of 3 potential salvage therapies). The trial is enrolling patients 60 years of age or older with relapsed or refractory acute myeloid leukemia who are ineligible for standard intensive chemotherapy and/or transplant.
The SADAL study is a phase 2b trial comparing high and low doses of selinexor in patients with relapsed and/or refractory de novo diffuse large B-cell lymphoma who have no therapeutic options of demonstrated clinical benefit.
STORM is a phase 2b trial evaluating selinexor and low-dose dexamethasone in patients with heavily pretreated multiple myeloma (MM). And STOMP is a phase 1b/2 study evaluating selinexor in combination with existing therapies across the broader population in MM.
Karyopharm is also planning a randomized, phase 3 study known as BOSTON. In this trial, researchers will compare selinexor plus bortezomib and low-dose dexamethasone to bortezomib and low-dose dexamethasone in MM patients who have had 1 to 3 prior lines of therapy.
Additional phase 1, 2, and 3 studies are ongoing or currently planned.
The US Food and Drug Administration (FDA) has placed a partial clinical hold on all trials of selinexor (KPT-330).
Selinexor is an inhibitor being evaluated in multiple trials of patients with relapsed and/or refractory hematologic and solid tumor malignancies.
While the partial clinical hold remains in effect, patients with stable disease or better may remain on selinexor.
However, no new patients may be enrolled in selinexor trials until the hold is lifted.
The FDA has indicated that the partial clinical hold is due to incomplete information in the existing version of the investigator’s brochure, including an incomplete list of serious adverse events associated with selinexor.
Karyopharm Therapeutics Inc., the company developing selinexor, said it has amended the brochure, updated the informed consent documents accordingly, and submitted the documents to the FDA as requested.
As of March 10, Karyopharm had provided all requested materials to the FDA believed to be required to lift the partial clinical hold. By regulation, the FDA has 30 days from the receipt of Karyopharm’s submission to notify the company whether the partial clinical hold is lifted.
Karyopharm said it is working with the FDA to seek the release of the hold and resume enrollment in its selinexor trials as expeditiously as possible. The company believes its previously disclosed enrollment rates and timelines for its ongoing trials will remain materially unchanged.
About selinexor
Selinexor is a selective inhibitor of nuclear export (SINE) XPO1 antagonist. The drug binds with and inhibits XPO1, leading to the accumulation of tumor suppressor proteins in the cell nucleus. This reinitiates and amplifies their tumor suppressor function and is believed to induce apoptosis in cancer cells while largely sparing normal cells.
To date, more than 1900 patients have been treated with selinexor. The drug is currently being evaluated in several trials across multiple cancer indications.
One of these is the phase 2 SOPRA trial, in which selinexor is being compared to investigator’s choice of therapy (1 of 3 potential salvage therapies). The trial is enrolling patients 60 years of age or older with relapsed or refractory acute myeloid leukemia who are ineligible for standard intensive chemotherapy and/or transplant.
The SADAL study is a phase 2b trial comparing high and low doses of selinexor in patients with relapsed and/or refractory de novo diffuse large B-cell lymphoma who have no therapeutic options of demonstrated clinical benefit.
STORM is a phase 2b trial evaluating selinexor and low-dose dexamethasone in patients with heavily pretreated multiple myeloma (MM). And STOMP is a phase 1b/2 study evaluating selinexor in combination with existing therapies across the broader population in MM.
Karyopharm is also planning a randomized, phase 3 study known as BOSTON. In this trial, researchers will compare selinexor plus bortezomib and low-dose dexamethasone to bortezomib and low-dose dexamethasone in MM patients who have had 1 to 3 prior lines of therapy.
Additional phase 1, 2, and 3 studies are ongoing or currently planned.
Sickle cell trait linked to end-stage renal disease
Black individuals with sickle cell trait (SCT) have an increased risk of developing end-stage renal disease (ESRD), according to new research.
The study indicates that having SCT actually doubles the risk of ESRD.
And the trait confers a similar degree of risk as APOL1 gene variants, which are currently the most widely recognized genetic contributors to kidney disease in blacks.
Researchers believe this finding may have important public policy implications for genetic counseling for individuals with SCT.
Rakhi P. Naik, MD, of Johns Hopkins University School of Medicine in Baltimore, Maryland, and her colleagues reported this finding in the Journal of the American Society of Nephrology.
Previous research suggested there is an association between SCT and chronic kidney disease, but it hasn’t been clear if that extends to ESRD. Studies have also suggested a possible association between kidney disease and hemoglobin C trait, but the link has not been confirmed.
So Dr Naik and her colleagues decided to investigate these potential links. To do so, the researchers analyzed data from a large, population-based study, the REasons for Geographic and Racial Differences in Stroke (REGARDS) study.
The team evaluated information on 9909 black individuals, 739 of whom had SCT and 243 of whom had hemoglobin C trait.
The data indicate that individuals with SCT have a 2-fold higher risk of developing ESRD when compared to those without SCT. But there is no association between hemoglobin C trait and ESRD.
At a median follow-up of 6.5 years, the incidence of ESRD was 5.4% (40/739) in participants with SCT, 2.5% (6/243) in subjects with hemoglobin C trait, and 2.6% (234/8927) in individuals without either trait.
The incidence rate for ESRD was 8.5 per 1000 person-years for participants with SCT, 3.9 per 1000 person-years for subjects with hemoglobin C trait, and 4.0 per 1000 person-years for individuals without either trait.
The researchers noted that SCT conferred a similar degree of ESRD risk as APOL1 gene variants. The hazard ratio for subjects with SCT was 2.03, and the hazard ratio for those with APOL1 high-risk genotypes was 1.77.
“Although you cannot change the genes you are born with, doctors can use this information to start screening for kidney disease earlier and to aggressively treat any other risk factors you may have, such as diabetes or high blood pressure,” Dr Naik said.
“We still need more studies to determine if there are other treatments that can be used to slow the progression of kidney disease, specifically in individuals with sickle cell trait.” ![]()
Black individuals with sickle cell trait (SCT) have an increased risk of developing end-stage renal disease (ESRD), according to new research.
The study indicates that having SCT actually doubles the risk of ESRD.
And the trait confers a similar degree of risk as APOL1 gene variants, which are currently the most widely recognized genetic contributors to kidney disease in blacks.
Researchers believe this finding may have important public policy implications for genetic counseling for individuals with SCT.
Rakhi P. Naik, MD, of Johns Hopkins University School of Medicine in Baltimore, Maryland, and her colleagues reported this finding in the Journal of the American Society of Nephrology.
Previous research suggested there is an association between SCT and chronic kidney disease, but it hasn’t been clear if that extends to ESRD. Studies have also suggested a possible association between kidney disease and hemoglobin C trait, but the link has not been confirmed.
So Dr Naik and her colleagues decided to investigate these potential links. To do so, the researchers analyzed data from a large, population-based study, the REasons for Geographic and Racial Differences in Stroke (REGARDS) study.
The team evaluated information on 9909 black individuals, 739 of whom had SCT and 243 of whom had hemoglobin C trait.
The data indicate that individuals with SCT have a 2-fold higher risk of developing ESRD when compared to those without SCT. But there is no association between hemoglobin C trait and ESRD.
At a median follow-up of 6.5 years, the incidence of ESRD was 5.4% (40/739) in participants with SCT, 2.5% (6/243) in subjects with hemoglobin C trait, and 2.6% (234/8927) in individuals without either trait.
The incidence rate for ESRD was 8.5 per 1000 person-years for participants with SCT, 3.9 per 1000 person-years for subjects with hemoglobin C trait, and 4.0 per 1000 person-years for individuals without either trait.
The researchers noted that SCT conferred a similar degree of ESRD risk as APOL1 gene variants. The hazard ratio for subjects with SCT was 2.03, and the hazard ratio for those with APOL1 high-risk genotypes was 1.77.
“Although you cannot change the genes you are born with, doctors can use this information to start screening for kidney disease earlier and to aggressively treat any other risk factors you may have, such as diabetes or high blood pressure,” Dr Naik said.
“We still need more studies to determine if there are other treatments that can be used to slow the progression of kidney disease, specifically in individuals with sickle cell trait.” ![]()
Black individuals with sickle cell trait (SCT) have an increased risk of developing end-stage renal disease (ESRD), according to new research.
The study indicates that having SCT actually doubles the risk of ESRD.
And the trait confers a similar degree of risk as APOL1 gene variants, which are currently the most widely recognized genetic contributors to kidney disease in blacks.
Researchers believe this finding may have important public policy implications for genetic counseling for individuals with SCT.
Rakhi P. Naik, MD, of Johns Hopkins University School of Medicine in Baltimore, Maryland, and her colleagues reported this finding in the Journal of the American Society of Nephrology.
Previous research suggested there is an association between SCT and chronic kidney disease, but it hasn’t been clear if that extends to ESRD. Studies have also suggested a possible association between kidney disease and hemoglobin C trait, but the link has not been confirmed.
So Dr Naik and her colleagues decided to investigate these potential links. To do so, the researchers analyzed data from a large, population-based study, the REasons for Geographic and Racial Differences in Stroke (REGARDS) study.
The team evaluated information on 9909 black individuals, 739 of whom had SCT and 243 of whom had hemoglobin C trait.
The data indicate that individuals with SCT have a 2-fold higher risk of developing ESRD when compared to those without SCT. But there is no association between hemoglobin C trait and ESRD.
At a median follow-up of 6.5 years, the incidence of ESRD was 5.4% (40/739) in participants with SCT, 2.5% (6/243) in subjects with hemoglobin C trait, and 2.6% (234/8927) in individuals without either trait.
The incidence rate for ESRD was 8.5 per 1000 person-years for participants with SCT, 3.9 per 1000 person-years for subjects with hemoglobin C trait, and 4.0 per 1000 person-years for individuals without either trait.
The researchers noted that SCT conferred a similar degree of ESRD risk as APOL1 gene variants. The hazard ratio for subjects with SCT was 2.03, and the hazard ratio for those with APOL1 high-risk genotypes was 1.77.
“Although you cannot change the genes you are born with, doctors can use this information to start screening for kidney disease earlier and to aggressively treat any other risk factors you may have, such as diabetes or high blood pressure,” Dr Naik said.
“We still need more studies to determine if there are other treatments that can be used to slow the progression of kidney disease, specifically in individuals with sickle cell trait.” ![]()
PRAC recommends suspending gadolinium agents
The European Medicines Agency’s (EMA) Pharmacovigilance and Risk Assessment Committee (PRAC) has recommended suspending marketing authorizations for 4 linear gadolinium contrast agents because of evidence that small amounts of the gadolinium they contain are deposited in the brain.
The agents concerned are intravenous injections of gadobenic acid, gadodiamide, gadopentetic acid, and gadoversetamide, which are given to patients to enhance images from magnetic resonance imaging (MRI) body scans.
The PRAC’s review of gadolinium agents found “convincing evidence” of accumulation of gadolinium in the brain from studies directly measuring gadolinium in brain tissues and areas of increased signal intensity seen on MRI scan images many months after the last injection of a gadolinium contrast agent.
Although no symptoms or diseases linked to gadolinium in the brain have been reported, the PRAC took a precautionary approach, noting that data on the long-term effects in the brain are limited.
Companies concerned by this review have the right to request that the PRAC re-examine its recommendations.
The PRAC’s recommendations will be sent to the Committee for Medicinal Products for Human Use (CHMP) for its opinion. Further details will be published at the time of the CHMP opinion.
The final stage of the review procedure is the adoption by the European Commission of a legally binding decision applicable in all European Union member states.
Details of the review, recommendations
The PRAC’s review was initiated on March 17, 2016, at the request of the European Commission, under Article 31 of Directive 2001/83/EC.
The review covers agents containing the following active substances: gadobenic acid, gadobutrol, gadodiamide, gadopentetic acid, gadoteric acid, gadoteridol, gadoversetamide, and gadoxetic acid.
The 4 agents recommended for suspension (gadobenic acid, gadodiamide, gadopentetic acid, and gadoversetamide) are linear agents. They have a structure more likely to release gadolinium, which can build up in body tissues.
Other agents, known as macrocyclic agents, are more stable and have a much lower propensity to release gadolinium.
The PRAC recommends that macrocyclic agents (gadobutrol, gadoteric acid, and gadoteridol) be used at the lowest dose that enhances images sufficiently to make diagnoses and only when unenhanced body scans are not suitable.
The PRAC has recommended that some linear agents remain available. The committee said that gadoxetic acid, a linear agent used at a low dose for liver scans, can remain on the market as it meets an important diagnostic need in patients with few alternatives.
In addition, a formulation of gadopentetic acid injected directly into joints should remain available because its gadolinium concentration is very low—around 200 times lower than those of intravenous products.
Both agents should be used at the lowest dose that enhances images sufficiently to make diagnoses and only if unenhanced scans are not suitable.
For those marketing authorizations recommended for suspension, the suspensions can be lifted if the respective companies provide evidence of new benefits in an identified patient group that outweigh its risks or show that their product (modified or not) does not release gadolinium significantly or lead to its retention in tissues. ![]()
The European Medicines Agency’s (EMA) Pharmacovigilance and Risk Assessment Committee (PRAC) has recommended suspending marketing authorizations for 4 linear gadolinium contrast agents because of evidence that small amounts of the gadolinium they contain are deposited in the brain.
The agents concerned are intravenous injections of gadobenic acid, gadodiamide, gadopentetic acid, and gadoversetamide, which are given to patients to enhance images from magnetic resonance imaging (MRI) body scans.
The PRAC’s review of gadolinium agents found “convincing evidence” of accumulation of gadolinium in the brain from studies directly measuring gadolinium in brain tissues and areas of increased signal intensity seen on MRI scan images many months after the last injection of a gadolinium contrast agent.
Although no symptoms or diseases linked to gadolinium in the brain have been reported, the PRAC took a precautionary approach, noting that data on the long-term effects in the brain are limited.
Companies concerned by this review have the right to request that the PRAC re-examine its recommendations.
The PRAC’s recommendations will be sent to the Committee for Medicinal Products for Human Use (CHMP) for its opinion. Further details will be published at the time of the CHMP opinion.
The final stage of the review procedure is the adoption by the European Commission of a legally binding decision applicable in all European Union member states.
Details of the review, recommendations
The PRAC’s review was initiated on March 17, 2016, at the request of the European Commission, under Article 31 of Directive 2001/83/EC.
The review covers agents containing the following active substances: gadobenic acid, gadobutrol, gadodiamide, gadopentetic acid, gadoteric acid, gadoteridol, gadoversetamide, and gadoxetic acid.
The 4 agents recommended for suspension (gadobenic acid, gadodiamide, gadopentetic acid, and gadoversetamide) are linear agents. They have a structure more likely to release gadolinium, which can build up in body tissues.
Other agents, known as macrocyclic agents, are more stable and have a much lower propensity to release gadolinium.
The PRAC recommends that macrocyclic agents (gadobutrol, gadoteric acid, and gadoteridol) be used at the lowest dose that enhances images sufficiently to make diagnoses and only when unenhanced body scans are not suitable.
The PRAC has recommended that some linear agents remain available. The committee said that gadoxetic acid, a linear agent used at a low dose for liver scans, can remain on the market as it meets an important diagnostic need in patients with few alternatives.
In addition, a formulation of gadopentetic acid injected directly into joints should remain available because its gadolinium concentration is very low—around 200 times lower than those of intravenous products.
Both agents should be used at the lowest dose that enhances images sufficiently to make diagnoses and only if unenhanced scans are not suitable.
For those marketing authorizations recommended for suspension, the suspensions can be lifted if the respective companies provide evidence of new benefits in an identified patient group that outweigh its risks or show that their product (modified or not) does not release gadolinium significantly or lead to its retention in tissues. ![]()
The European Medicines Agency’s (EMA) Pharmacovigilance and Risk Assessment Committee (PRAC) has recommended suspending marketing authorizations for 4 linear gadolinium contrast agents because of evidence that small amounts of the gadolinium they contain are deposited in the brain.
The agents concerned are intravenous injections of gadobenic acid, gadodiamide, gadopentetic acid, and gadoversetamide, which are given to patients to enhance images from magnetic resonance imaging (MRI) body scans.
The PRAC’s review of gadolinium agents found “convincing evidence” of accumulation of gadolinium in the brain from studies directly measuring gadolinium in brain tissues and areas of increased signal intensity seen on MRI scan images many months after the last injection of a gadolinium contrast agent.
Although no symptoms or diseases linked to gadolinium in the brain have been reported, the PRAC took a precautionary approach, noting that data on the long-term effects in the brain are limited.
Companies concerned by this review have the right to request that the PRAC re-examine its recommendations.
The PRAC’s recommendations will be sent to the Committee for Medicinal Products for Human Use (CHMP) for its opinion. Further details will be published at the time of the CHMP opinion.
The final stage of the review procedure is the adoption by the European Commission of a legally binding decision applicable in all European Union member states.
Details of the review, recommendations
The PRAC’s review was initiated on March 17, 2016, at the request of the European Commission, under Article 31 of Directive 2001/83/EC.
The review covers agents containing the following active substances: gadobenic acid, gadobutrol, gadodiamide, gadopentetic acid, gadoteric acid, gadoteridol, gadoversetamide, and gadoxetic acid.
The 4 agents recommended for suspension (gadobenic acid, gadodiamide, gadopentetic acid, and gadoversetamide) are linear agents. They have a structure more likely to release gadolinium, which can build up in body tissues.
Other agents, known as macrocyclic agents, are more stable and have a much lower propensity to release gadolinium.
The PRAC recommends that macrocyclic agents (gadobutrol, gadoteric acid, and gadoteridol) be used at the lowest dose that enhances images sufficiently to make diagnoses and only when unenhanced body scans are not suitable.
The PRAC has recommended that some linear agents remain available. The committee said that gadoxetic acid, a linear agent used at a low dose for liver scans, can remain on the market as it meets an important diagnostic need in patients with few alternatives.
In addition, a formulation of gadopentetic acid injected directly into joints should remain available because its gadolinium concentration is very low—around 200 times lower than those of intravenous products.
Both agents should be used at the lowest dose that enhances images sufficiently to make diagnoses and only if unenhanced scans are not suitable.
For those marketing authorizations recommended for suspension, the suspensions can be lifted if the respective companies provide evidence of new benefits in an identified patient group that outweigh its risks or show that their product (modified or not) does not release gadolinium significantly or lead to its retention in tissues. ![]()
Drug receives orphan designation for DLBCL
The US Food and Drug Administration (FDA) has granted orphan drug designation for eFT508 to treat diffuse large B-cell lymphoma (DLBCL).
eFT508 is a highly selective inhibitor of MNK1 and MNK2, enzymes that integrate signals from several oncogenic and immune signaling pathways.
The FDA grants orphan designation to drugs or biologics intended to treat a disease or condition affecting fewer than 200,000 patients in the US.
The orphan designation for eFT508 provides several incentives for eFFECTOR Therapeutics, the company developing eFT508.
These incentives include increased access to FDA reviewers to discuss clinical trial designs, the ability to qualify for tax credits for certain clinical research costs, the ability to apply for annual grant funding, a waiver of Prescription Drug User Fee Act filing fees, and the potential for 7 years of US marketing exclusivity if eFT508 is approved.
eFFECTOR has dosed the first subject in a phase 1/2 trial of eFT508 in patients with B-cell hematologic malignancies. The study is designed to evaluate the safety, pharmacokinetics, pharmacodynamics, and antitumor activity of eFT508.
eFFECTOR presented preclinical research of eFT508 in DLBCL at the 2015 ASH Annual Meeting. The poster is available for download from the eFFECTOR website.
The researchers reported that eFT508 demonstrated anti-proliferative activity against multiple DLBCL cell lines, including the TMD8, OCI-Ly3, and HBL1 cell lines.
eFT508 also exhibited “significant anti-tumor activity” in mouse models of TMD8 and HBL-1 ABC-DLBCL.
Finally, the researchers found that eFT508 synergized with everolimus, ibrutinib, and venetoclax both in vitro and in vivo. ![]()
The US Food and Drug Administration (FDA) has granted orphan drug designation for eFT508 to treat diffuse large B-cell lymphoma (DLBCL).
eFT508 is a highly selective inhibitor of MNK1 and MNK2, enzymes that integrate signals from several oncogenic and immune signaling pathways.
The FDA grants orphan designation to drugs or biologics intended to treat a disease or condition affecting fewer than 200,000 patients in the US.
The orphan designation for eFT508 provides several incentives for eFFECTOR Therapeutics, the company developing eFT508.
These incentives include increased access to FDA reviewers to discuss clinical trial designs, the ability to qualify for tax credits for certain clinical research costs, the ability to apply for annual grant funding, a waiver of Prescription Drug User Fee Act filing fees, and the potential for 7 years of US marketing exclusivity if eFT508 is approved.
eFFECTOR has dosed the first subject in a phase 1/2 trial of eFT508 in patients with B-cell hematologic malignancies. The study is designed to evaluate the safety, pharmacokinetics, pharmacodynamics, and antitumor activity of eFT508.
eFFECTOR presented preclinical research of eFT508 in DLBCL at the 2015 ASH Annual Meeting. The poster is available for download from the eFFECTOR website.
The researchers reported that eFT508 demonstrated anti-proliferative activity against multiple DLBCL cell lines, including the TMD8, OCI-Ly3, and HBL1 cell lines.
eFT508 also exhibited “significant anti-tumor activity” in mouse models of TMD8 and HBL-1 ABC-DLBCL.
Finally, the researchers found that eFT508 synergized with everolimus, ibrutinib, and venetoclax both in vitro and in vivo. ![]()
The US Food and Drug Administration (FDA) has granted orphan drug designation for eFT508 to treat diffuse large B-cell lymphoma (DLBCL).
eFT508 is a highly selective inhibitor of MNK1 and MNK2, enzymes that integrate signals from several oncogenic and immune signaling pathways.
The FDA grants orphan designation to drugs or biologics intended to treat a disease or condition affecting fewer than 200,000 patients in the US.
The orphan designation for eFT508 provides several incentives for eFFECTOR Therapeutics, the company developing eFT508.
These incentives include increased access to FDA reviewers to discuss clinical trial designs, the ability to qualify for tax credits for certain clinical research costs, the ability to apply for annual grant funding, a waiver of Prescription Drug User Fee Act filing fees, and the potential for 7 years of US marketing exclusivity if eFT508 is approved.
eFFECTOR has dosed the first subject in a phase 1/2 trial of eFT508 in patients with B-cell hematologic malignancies. The study is designed to evaluate the safety, pharmacokinetics, pharmacodynamics, and antitumor activity of eFT508.
eFFECTOR presented preclinical research of eFT508 in DLBCL at the 2015 ASH Annual Meeting. The poster is available for download from the eFFECTOR website.
The researchers reported that eFT508 demonstrated anti-proliferative activity against multiple DLBCL cell lines, including the TMD8, OCI-Ly3, and HBL1 cell lines.
eFT508 also exhibited “significant anti-tumor activity” in mouse models of TMD8 and HBL-1 ABC-DLBCL.
Finally, the researchers found that eFT508 synergized with everolimus, ibrutinib, and venetoclax both in vitro and in vivo. ![]()
FDA grants mAb orphan designation for MDS
The US Food and Drug Administration (FDA) has granted orphan drug designation for BI 836858, an anti-CD33 monoclonal antibody (mAb), in the treatment of myelodysplastic syndromes (MDS).
BI 836858 previously received orphan designation for the treatment of acute myeloid leukemia (AML).
The FDA grants orphan designation to drugs and biologics intended to treat, diagnose, or prevent rare diseases/disorders affecting fewer than 200,000 people in the US.
Orphan designation provides companies with certain incentives to develop products for rare diseases.
This includes a 50% tax break on research and development, a fee waiver, access to federal grants, and 7 years of market exclusivity if the product is approved.
About BI 836858
BI 836858 is a fully human, immunoglobulin G1 anti-CD33 mAb. It has been engineered for improved binding to FcgRIIIa to mediate natural killer cell antibody-dependent cellular cytotoxicity against CD33-expressing tumor cells.
BI 836858 is being developed by Boehringer Ingelheim.
A phase 1/2 trial (NCT02240706) of BI 836858 in patients with MDS is ongoing. The phase 1 portion was designed to evaluate various doses of the mAb in patients with low or intermediate-1 risk MDS with symptomatic anemia.
The phase 2 portion was designed to compare BI 836858 plus best supportive care to best supportive care alone in patients with low- or intermediate-1-risk MDS who have symptomatic anemia but do not have a 5q deletion.
BI 836858 is also being tested in combination with decitabine in a phase 1/2 study (NCT02632721) of patients with AML.
The goals of the phase 1 portion and the phase 1 extension are to determine the maximum-tolerated dose/recommended dose, safety, pharmacokinetics, and efficacy of BI 836858 in combination with decitabine.
The goals of the phase 2 portion of the study are to investigate the efficacy, safety, and pharmacokinetics of BI 836858 in combination with decitabine compared to decitabine monotherapy.
BI 836858 was previously evaluated in combination with decitabine in a preclinical study. The combination exhibited activity against AML in vitro. The research was published in Blood last year.
BI 836858 is also being evaluated as part of the Leukemia & Lymphoma Society’s Beat AML Master Trial program to advance treatment for patients with AML.
In this trial, investigators are using genomic technology to identify AML mutations in newly diagnosed patients over the age of 60 and match the patients with an investigational drug or drugs best suited to attack the mutations found. ![]()
The US Food and Drug Administration (FDA) has granted orphan drug designation for BI 836858, an anti-CD33 monoclonal antibody (mAb), in the treatment of myelodysplastic syndromes (MDS).
BI 836858 previously received orphan designation for the treatment of acute myeloid leukemia (AML).
The FDA grants orphan designation to drugs and biologics intended to treat, diagnose, or prevent rare diseases/disorders affecting fewer than 200,000 people in the US.
Orphan designation provides companies with certain incentives to develop products for rare diseases.
This includes a 50% tax break on research and development, a fee waiver, access to federal grants, and 7 years of market exclusivity if the product is approved.
About BI 836858
BI 836858 is a fully human, immunoglobulin G1 anti-CD33 mAb. It has been engineered for improved binding to FcgRIIIa to mediate natural killer cell antibody-dependent cellular cytotoxicity against CD33-expressing tumor cells.
BI 836858 is being developed by Boehringer Ingelheim.
A phase 1/2 trial (NCT02240706) of BI 836858 in patients with MDS is ongoing. The phase 1 portion was designed to evaluate various doses of the mAb in patients with low or intermediate-1 risk MDS with symptomatic anemia.
The phase 2 portion was designed to compare BI 836858 plus best supportive care to best supportive care alone in patients with low- or intermediate-1-risk MDS who have symptomatic anemia but do not have a 5q deletion.
BI 836858 is also being tested in combination with decitabine in a phase 1/2 study (NCT02632721) of patients with AML.
The goals of the phase 1 portion and the phase 1 extension are to determine the maximum-tolerated dose/recommended dose, safety, pharmacokinetics, and efficacy of BI 836858 in combination with decitabine.
The goals of the phase 2 portion of the study are to investigate the efficacy, safety, and pharmacokinetics of BI 836858 in combination with decitabine compared to decitabine monotherapy.
BI 836858 was previously evaluated in combination with decitabine in a preclinical study. The combination exhibited activity against AML in vitro. The research was published in Blood last year.
BI 836858 is also being evaluated as part of the Leukemia & Lymphoma Society’s Beat AML Master Trial program to advance treatment for patients with AML.
In this trial, investigators are using genomic technology to identify AML mutations in newly diagnosed patients over the age of 60 and match the patients with an investigational drug or drugs best suited to attack the mutations found. ![]()
The US Food and Drug Administration (FDA) has granted orphan drug designation for BI 836858, an anti-CD33 monoclonal antibody (mAb), in the treatment of myelodysplastic syndromes (MDS).
BI 836858 previously received orphan designation for the treatment of acute myeloid leukemia (AML).
The FDA grants orphan designation to drugs and biologics intended to treat, diagnose, or prevent rare diseases/disorders affecting fewer than 200,000 people in the US.
Orphan designation provides companies with certain incentives to develop products for rare diseases.
This includes a 50% tax break on research and development, a fee waiver, access to federal grants, and 7 years of market exclusivity if the product is approved.
About BI 836858
BI 836858 is a fully human, immunoglobulin G1 anti-CD33 mAb. It has been engineered for improved binding to FcgRIIIa to mediate natural killer cell antibody-dependent cellular cytotoxicity against CD33-expressing tumor cells.
BI 836858 is being developed by Boehringer Ingelheim.
A phase 1/2 trial (NCT02240706) of BI 836858 in patients with MDS is ongoing. The phase 1 portion was designed to evaluate various doses of the mAb in patients with low or intermediate-1 risk MDS with symptomatic anemia.
The phase 2 portion was designed to compare BI 836858 plus best supportive care to best supportive care alone in patients with low- or intermediate-1-risk MDS who have symptomatic anemia but do not have a 5q deletion.
BI 836858 is also being tested in combination with decitabine in a phase 1/2 study (NCT02632721) of patients with AML.
The goals of the phase 1 portion and the phase 1 extension are to determine the maximum-tolerated dose/recommended dose, safety, pharmacokinetics, and efficacy of BI 836858 in combination with decitabine.
The goals of the phase 2 portion of the study are to investigate the efficacy, safety, and pharmacokinetics of BI 836858 in combination with decitabine compared to decitabine monotherapy.
BI 836858 was previously evaluated in combination with decitabine in a preclinical study. The combination exhibited activity against AML in vitro. The research was published in Blood last year.
BI 836858 is also being evaluated as part of the Leukemia & Lymphoma Society’s Beat AML Master Trial program to advance treatment for patients with AML.
In this trial, investigators are using genomic technology to identify AML mutations in newly diagnosed patients over the age of 60 and match the patients with an investigational drug or drugs best suited to attack the mutations found. ![]()
Imatinib is safe, effective long-term, team says
Long-term follow-up of patients treated with imatinib suggests the drug can remain effective beyond 10 years and does not confer “unacceptable” cumulative toxicity, according to researchers.
The group evaluated data on patients who had newly diagnosed, chronic-phase chronic myeloid leukemia (CML) when they began treatment with imatinib.
The median treatment duration was 8.9 years, and the estimated 10-year survival rate ranged from 64.4% to 84.4%.
The researchers said serious adverse events (AEs) thought to be related to imatinib were uncommon and typically occurred early, within the first year of treatment.
These results were reported in NEJM. The research was funded by Novartis Pharmaceuticals, which markets imatinib as Gleevec.
“The long-term success of this treatment confirms the remarkable success we’ve seen since the very first Gleevec trials,” said study author Brian Druker, MD, a physician-scientist at Oregon Health & Science University in Portland, Oregon, who led the original clinical development of Gleevec.
“This study reinforces the notion that we can create effective and non-toxic therapies.”
The study enrolled 1106 newly diagnosed, chronic-phase CML patients at 177 cancer centers in more than 16 countries. Half were assigned to treatment with imatinib (n=533) and the other half to interferon alfa plus cytarabine.
This study allowed for cross-over between the treatment arms, and 65.6% of patients in the cytarabine/interferon alfa arm ultimately crossed over to the imatinib arm.
However, when assessing the effects of imatinib, the researchers focused only on the patients who were first randomized to receive imatinib.
The median follow-up was 10.9 years (range, 0 to 11.7, which included follow-up after patients discontinued study treatment).
Of the patients randomized to imatinib, 48.3% (n=267) completed treatment with the drug. The median duration of first-line imatinib was 8.9 years (range, <0.1 to 11.7).
For patients who did not complete imatinib treatment, reasons for discontinuation included a lack of efficacy (15.9%), withdrawn consent (10.3%), AEs (6.9%), because they proceeded to transplant (3.8%), death (3.4%), protocol violation (3.1%), loss to follow-up (2.7%), cross over to the interferon arm (2.5%), administrative problems (2.2%), abnormal laboratory values (0.5%), or abnormal procedure (0.4%).
Safety
The incidence of serious AEs considered related to imatinib was 9.3% (51/551).
Drug-related serious AEs occurring in at least 2 patients included abdominal pain (n=4), anemia (n=3), congestive cardiac failure (n=3), gastrointestinal hemorrhage (n=3), vomiting (n=3), alanine aminotransferase increase (n=2), cardiac arrest (n=2), conjunctival hemorrhage (n=2), and melana (n=2).
Six patients had a second neoplasm (benign, malignant, or unspecified).
Response
The cumulative rate of complete cytogenetic response (CCR) at the end of the trial was 82.8%.
In the intent-to-treat population, the rate of CCR went from 52.8% in the first year to 22.2% at year 10.
Among evaluable patients, the rate of CCR went from 70.9% (292/412) in the first year to 91.8% (123/134) in year 10.
In the intent-to-treat population, the rate of major molecular response went from 27.7% in the first year to 34.4% at year 10.
Among evaluable patients, the rate of major molecular response went from 50.2% (153/305) in the first year to 93.1% (190/204) in year 10.
Progression and survival
The rate of progression was 6.9% (38/553) in the intent-to-treat population. Most of these patients (n=34) progressed during the first 4 years.
There were 260 patients who were still alive and receiving imatinib at 10 years and 96 patients who were alive but not receiving imatinib.
The researchers did not know the survival status of 111 patients, and there were 86 known deaths at 10 years (89 by the end of the study).
The estimated 10-year survival rate ranged from 64.4% (assuming all 111 patients with unknown status had died) to 84.4% (assuming all 111 were alive).
The cause of death was CML in 50 patients, a secondary malignant condition in 11, a cardiac disorder/cardiovascular disease in 7, infectious disease in 5, and “other” causes in 16 patients. ![]()
Long-term follow-up of patients treated with imatinib suggests the drug can remain effective beyond 10 years and does not confer “unacceptable” cumulative toxicity, according to researchers.
The group evaluated data on patients who had newly diagnosed, chronic-phase chronic myeloid leukemia (CML) when they began treatment with imatinib.
The median treatment duration was 8.9 years, and the estimated 10-year survival rate ranged from 64.4% to 84.4%.
The researchers said serious adverse events (AEs) thought to be related to imatinib were uncommon and typically occurred early, within the first year of treatment.
These results were reported in NEJM. The research was funded by Novartis Pharmaceuticals, which markets imatinib as Gleevec.
“The long-term success of this treatment confirms the remarkable success we’ve seen since the very first Gleevec trials,” said study author Brian Druker, MD, a physician-scientist at Oregon Health & Science University in Portland, Oregon, who led the original clinical development of Gleevec.
“This study reinforces the notion that we can create effective and non-toxic therapies.”
The study enrolled 1106 newly diagnosed, chronic-phase CML patients at 177 cancer centers in more than 16 countries. Half were assigned to treatment with imatinib (n=533) and the other half to interferon alfa plus cytarabine.
This study allowed for cross-over between the treatment arms, and 65.6% of patients in the cytarabine/interferon alfa arm ultimately crossed over to the imatinib arm.
However, when assessing the effects of imatinib, the researchers focused only on the patients who were first randomized to receive imatinib.
The median follow-up was 10.9 years (range, 0 to 11.7, which included follow-up after patients discontinued study treatment).
Of the patients randomized to imatinib, 48.3% (n=267) completed treatment with the drug. The median duration of first-line imatinib was 8.9 years (range, <0.1 to 11.7).
For patients who did not complete imatinib treatment, reasons for discontinuation included a lack of efficacy (15.9%), withdrawn consent (10.3%), AEs (6.9%), because they proceeded to transplant (3.8%), death (3.4%), protocol violation (3.1%), loss to follow-up (2.7%), cross over to the interferon arm (2.5%), administrative problems (2.2%), abnormal laboratory values (0.5%), or abnormal procedure (0.4%).
Safety
The incidence of serious AEs considered related to imatinib was 9.3% (51/551).
Drug-related serious AEs occurring in at least 2 patients included abdominal pain (n=4), anemia (n=3), congestive cardiac failure (n=3), gastrointestinal hemorrhage (n=3), vomiting (n=3), alanine aminotransferase increase (n=2), cardiac arrest (n=2), conjunctival hemorrhage (n=2), and melana (n=2).
Six patients had a second neoplasm (benign, malignant, or unspecified).
Response
The cumulative rate of complete cytogenetic response (CCR) at the end of the trial was 82.8%.
In the intent-to-treat population, the rate of CCR went from 52.8% in the first year to 22.2% at year 10.
Among evaluable patients, the rate of CCR went from 70.9% (292/412) in the first year to 91.8% (123/134) in year 10.
In the intent-to-treat population, the rate of major molecular response went from 27.7% in the first year to 34.4% at year 10.
Among evaluable patients, the rate of major molecular response went from 50.2% (153/305) in the first year to 93.1% (190/204) in year 10.
Progression and survival
The rate of progression was 6.9% (38/553) in the intent-to-treat population. Most of these patients (n=34) progressed during the first 4 years.
There were 260 patients who were still alive and receiving imatinib at 10 years and 96 patients who were alive but not receiving imatinib.
The researchers did not know the survival status of 111 patients, and there were 86 known deaths at 10 years (89 by the end of the study).
The estimated 10-year survival rate ranged from 64.4% (assuming all 111 patients with unknown status had died) to 84.4% (assuming all 111 were alive).
The cause of death was CML in 50 patients, a secondary malignant condition in 11, a cardiac disorder/cardiovascular disease in 7, infectious disease in 5, and “other” causes in 16 patients. ![]()
Long-term follow-up of patients treated with imatinib suggests the drug can remain effective beyond 10 years and does not confer “unacceptable” cumulative toxicity, according to researchers.
The group evaluated data on patients who had newly diagnosed, chronic-phase chronic myeloid leukemia (CML) when they began treatment with imatinib.
The median treatment duration was 8.9 years, and the estimated 10-year survival rate ranged from 64.4% to 84.4%.
The researchers said serious adverse events (AEs) thought to be related to imatinib were uncommon and typically occurred early, within the first year of treatment.
These results were reported in NEJM. The research was funded by Novartis Pharmaceuticals, which markets imatinib as Gleevec.
“The long-term success of this treatment confirms the remarkable success we’ve seen since the very first Gleevec trials,” said study author Brian Druker, MD, a physician-scientist at Oregon Health & Science University in Portland, Oregon, who led the original clinical development of Gleevec.
“This study reinforces the notion that we can create effective and non-toxic therapies.”
The study enrolled 1106 newly diagnosed, chronic-phase CML patients at 177 cancer centers in more than 16 countries. Half were assigned to treatment with imatinib (n=533) and the other half to interferon alfa plus cytarabine.
This study allowed for cross-over between the treatment arms, and 65.6% of patients in the cytarabine/interferon alfa arm ultimately crossed over to the imatinib arm.
However, when assessing the effects of imatinib, the researchers focused only on the patients who were first randomized to receive imatinib.
The median follow-up was 10.9 years (range, 0 to 11.7, which included follow-up after patients discontinued study treatment).
Of the patients randomized to imatinib, 48.3% (n=267) completed treatment with the drug. The median duration of first-line imatinib was 8.9 years (range, <0.1 to 11.7).
For patients who did not complete imatinib treatment, reasons for discontinuation included a lack of efficacy (15.9%), withdrawn consent (10.3%), AEs (6.9%), because they proceeded to transplant (3.8%), death (3.4%), protocol violation (3.1%), loss to follow-up (2.7%), cross over to the interferon arm (2.5%), administrative problems (2.2%), abnormal laboratory values (0.5%), or abnormal procedure (0.4%).
Safety
The incidence of serious AEs considered related to imatinib was 9.3% (51/551).
Drug-related serious AEs occurring in at least 2 patients included abdominal pain (n=4), anemia (n=3), congestive cardiac failure (n=3), gastrointestinal hemorrhage (n=3), vomiting (n=3), alanine aminotransferase increase (n=2), cardiac arrest (n=2), conjunctival hemorrhage (n=2), and melana (n=2).
Six patients had a second neoplasm (benign, malignant, or unspecified).
Response
The cumulative rate of complete cytogenetic response (CCR) at the end of the trial was 82.8%.
In the intent-to-treat population, the rate of CCR went from 52.8% in the first year to 22.2% at year 10.
Among evaluable patients, the rate of CCR went from 70.9% (292/412) in the first year to 91.8% (123/134) in year 10.
In the intent-to-treat population, the rate of major molecular response went from 27.7% in the first year to 34.4% at year 10.
Among evaluable patients, the rate of major molecular response went from 50.2% (153/305) in the first year to 93.1% (190/204) in year 10.
Progression and survival
The rate of progression was 6.9% (38/553) in the intent-to-treat population. Most of these patients (n=34) progressed during the first 4 years.
There were 260 patients who were still alive and receiving imatinib at 10 years and 96 patients who were alive but not receiving imatinib.
The researchers did not know the survival status of 111 patients, and there were 86 known deaths at 10 years (89 by the end of the study).
The estimated 10-year survival rate ranged from 64.4% (assuming all 111 patients with unknown status had died) to 84.4% (assuming all 111 were alive).
The cause of death was CML in 50 patients, a secondary malignant condition in 11, a cardiac disorder/cardiovascular disease in 7, infectious disease in 5, and “other” causes in 16 patients. ![]()
Drug exhibits anti-myeloma activity in mice, humans
An experimental drug called LCL161 stimulates the immune system to fight multiple myeloma (MM), according to research published in Nature Medicine.
Investigators said LCL161 exhibited “robust” activity in a transgenic myeloma mouse model and in patients with relapsed/refractory MM.
Single-agent LCL161 did not produce responses in MM patients, but patients did respond to treatment with LCL161 and cyclophosphamide.
The investigators also found that single-agent LCL161 provided “long-term anti-tumor protection” in mice, and combining LCL161 with an antibody against PD-1 could cure mice of MM.
“The drug, LCL161, was initially developed to promote tumor death,” said study author Marta Chesi, PhD, of Mayo Clinic Arizona in Scottsdale.
“However, we found that the drug does not kill tumor cells directly. Rather, it makes them more visible to the immune system that recognizes them as foreign invaders and eliminates them.”
Dr Chesi and her colleagues explained that the cellular inhibitors of apoptosis (cIAP) 1 and 2 have been identified as potential therapeutic targets in some cancers.
And LCL161 is a small-molecule IAP antagonist that induces tumor necrosis factor-mediated apoptosis in cancer cells. However, the investigators found that LCL161 was not directly cytotoxic to MM cells.
Instead, the drug upregulated tumor-cell-autonomous type I interferon signaling and induced an acute inflammatory response. This led to the activation of macrophages and dendritic cells, which prompted phagocytosis in MM cells.
Results in mice
The investigators first tested LCL161 alone (at a dose previously shown to be well-tolerated) in Vk*MYC transgenic mice with established MM.
The team said they observed a reduction in tumor burden that was comparable to that observed in response to drugs currently used to treat MM—carfilzomib, bortezomib, melphalan, cyclophosphamide, panobinostat, dexamethasone, and pomalidomide.
The investigators then tested the combination of LCL161 and a PD1 antibody in Vk12598-tumor-bearing mice.
The team said the combination was curative in all mice that completed 2 weeks of treatment. In fact, it was more effective than combination treatment with LCL161 and cyclophosphamide.
Results in patients
Dr Chesi and her colleagues conducted a phase 2 trial of LCL161 in 25 patients with relapsed/refractory MM. Patients could receive cyclophosphamide if they failed to respond or progressed after 8 weeks of treatment with LCL161 alone.
The patients’ median age was 68 (range, 47-90), and they had a median of 3 prior therapies (range, 1-6). Forty-four percent of patients had high-risk features, 28% had relapsed disease, and 72% had relapsed and refractory disease.
Four patients experienced grade 2 cytokine release syndrome when they received LCL161 at a dose of 1800 mg weekly, so the dose was lowered to 1200 mg.
None of the patients responded to single-agent LCL161. So 23 of the patients received 500 mg of weekly cyclophosphamide as well.
There was 1 complete response to the combination therapy, 1 very good partial response, 2 partial responses, and 1 minimal response. The median progression-free survival in these patients was 10 months.
Grade 3 adverse events included decrease in neutrophil count (28%), decrease in lymphocyte count (28%), anemia (24%), fatigue (16%), hyperglycemia (12%), syncope (12%), decrease in white blood cell count (12%), decrease in platelet count (8%), increase in lymphocyte count (8%), nausea (4%), vomiting (4%), diarrhea (4%), maculo-papular rash (4%), hypotension (4%), lung infection (4%), pain in extremity (4%), and urticaria (4%).
Grade 4 events included decrease in lymphocyte count (24%), decrease in neutrophil count (8%), decrease in white blood cell count (8%), hyperuricemia (4%), decrease in platelet count (4%), and sepsis (4%).
Based on these results, the investigators said the combination of LCL161 and cyclophosphamide is “an attractive platform for future trials,” and the same is true for LCL161 in combination with anti-PD1 therapy.
The phase 2 trial was sponsored by Mayo Clinic and the National Cancer Institute. Novartis provided LCL161 for this research and supported the trial. ![]()
An experimental drug called LCL161 stimulates the immune system to fight multiple myeloma (MM), according to research published in Nature Medicine.
Investigators said LCL161 exhibited “robust” activity in a transgenic myeloma mouse model and in patients with relapsed/refractory MM.
Single-agent LCL161 did not produce responses in MM patients, but patients did respond to treatment with LCL161 and cyclophosphamide.
The investigators also found that single-agent LCL161 provided “long-term anti-tumor protection” in mice, and combining LCL161 with an antibody against PD-1 could cure mice of MM.
“The drug, LCL161, was initially developed to promote tumor death,” said study author Marta Chesi, PhD, of Mayo Clinic Arizona in Scottsdale.
“However, we found that the drug does not kill tumor cells directly. Rather, it makes them more visible to the immune system that recognizes them as foreign invaders and eliminates them.”
Dr Chesi and her colleagues explained that the cellular inhibitors of apoptosis (cIAP) 1 and 2 have been identified as potential therapeutic targets in some cancers.
And LCL161 is a small-molecule IAP antagonist that induces tumor necrosis factor-mediated apoptosis in cancer cells. However, the investigators found that LCL161 was not directly cytotoxic to MM cells.
Instead, the drug upregulated tumor-cell-autonomous type I interferon signaling and induced an acute inflammatory response. This led to the activation of macrophages and dendritic cells, which prompted phagocytosis in MM cells.
Results in mice
The investigators first tested LCL161 alone (at a dose previously shown to be well-tolerated) in Vk*MYC transgenic mice with established MM.
The team said they observed a reduction in tumor burden that was comparable to that observed in response to drugs currently used to treat MM—carfilzomib, bortezomib, melphalan, cyclophosphamide, panobinostat, dexamethasone, and pomalidomide.
The investigators then tested the combination of LCL161 and a PD1 antibody in Vk12598-tumor-bearing mice.
The team said the combination was curative in all mice that completed 2 weeks of treatment. In fact, it was more effective than combination treatment with LCL161 and cyclophosphamide.
Results in patients
Dr Chesi and her colleagues conducted a phase 2 trial of LCL161 in 25 patients with relapsed/refractory MM. Patients could receive cyclophosphamide if they failed to respond or progressed after 8 weeks of treatment with LCL161 alone.
The patients’ median age was 68 (range, 47-90), and they had a median of 3 prior therapies (range, 1-6). Forty-four percent of patients had high-risk features, 28% had relapsed disease, and 72% had relapsed and refractory disease.
Four patients experienced grade 2 cytokine release syndrome when they received LCL161 at a dose of 1800 mg weekly, so the dose was lowered to 1200 mg.
None of the patients responded to single-agent LCL161. So 23 of the patients received 500 mg of weekly cyclophosphamide as well.
There was 1 complete response to the combination therapy, 1 very good partial response, 2 partial responses, and 1 minimal response. The median progression-free survival in these patients was 10 months.
Grade 3 adverse events included decrease in neutrophil count (28%), decrease in lymphocyte count (28%), anemia (24%), fatigue (16%), hyperglycemia (12%), syncope (12%), decrease in white blood cell count (12%), decrease in platelet count (8%), increase in lymphocyte count (8%), nausea (4%), vomiting (4%), diarrhea (4%), maculo-papular rash (4%), hypotension (4%), lung infection (4%), pain in extremity (4%), and urticaria (4%).
Grade 4 events included decrease in lymphocyte count (24%), decrease in neutrophil count (8%), decrease in white blood cell count (8%), hyperuricemia (4%), decrease in platelet count (4%), and sepsis (4%).
Based on these results, the investigators said the combination of LCL161 and cyclophosphamide is “an attractive platform for future trials,” and the same is true for LCL161 in combination with anti-PD1 therapy.
The phase 2 trial was sponsored by Mayo Clinic and the National Cancer Institute. Novartis provided LCL161 for this research and supported the trial. ![]()
An experimental drug called LCL161 stimulates the immune system to fight multiple myeloma (MM), according to research published in Nature Medicine.
Investigators said LCL161 exhibited “robust” activity in a transgenic myeloma mouse model and in patients with relapsed/refractory MM.
Single-agent LCL161 did not produce responses in MM patients, but patients did respond to treatment with LCL161 and cyclophosphamide.
The investigators also found that single-agent LCL161 provided “long-term anti-tumor protection” in mice, and combining LCL161 with an antibody against PD-1 could cure mice of MM.
“The drug, LCL161, was initially developed to promote tumor death,” said study author Marta Chesi, PhD, of Mayo Clinic Arizona in Scottsdale.
“However, we found that the drug does not kill tumor cells directly. Rather, it makes them more visible to the immune system that recognizes them as foreign invaders and eliminates them.”
Dr Chesi and her colleagues explained that the cellular inhibitors of apoptosis (cIAP) 1 and 2 have been identified as potential therapeutic targets in some cancers.
And LCL161 is a small-molecule IAP antagonist that induces tumor necrosis factor-mediated apoptosis in cancer cells. However, the investigators found that LCL161 was not directly cytotoxic to MM cells.
Instead, the drug upregulated tumor-cell-autonomous type I interferon signaling and induced an acute inflammatory response. This led to the activation of macrophages and dendritic cells, which prompted phagocytosis in MM cells.
Results in mice
The investigators first tested LCL161 alone (at a dose previously shown to be well-tolerated) in Vk*MYC transgenic mice with established MM.
The team said they observed a reduction in tumor burden that was comparable to that observed in response to drugs currently used to treat MM—carfilzomib, bortezomib, melphalan, cyclophosphamide, panobinostat, dexamethasone, and pomalidomide.
The investigators then tested the combination of LCL161 and a PD1 antibody in Vk12598-tumor-bearing mice.
The team said the combination was curative in all mice that completed 2 weeks of treatment. In fact, it was more effective than combination treatment with LCL161 and cyclophosphamide.
Results in patients
Dr Chesi and her colleagues conducted a phase 2 trial of LCL161 in 25 patients with relapsed/refractory MM. Patients could receive cyclophosphamide if they failed to respond or progressed after 8 weeks of treatment with LCL161 alone.
The patients’ median age was 68 (range, 47-90), and they had a median of 3 prior therapies (range, 1-6). Forty-four percent of patients had high-risk features, 28% had relapsed disease, and 72% had relapsed and refractory disease.
Four patients experienced grade 2 cytokine release syndrome when they received LCL161 at a dose of 1800 mg weekly, so the dose was lowered to 1200 mg.
None of the patients responded to single-agent LCL161. So 23 of the patients received 500 mg of weekly cyclophosphamide as well.
There was 1 complete response to the combination therapy, 1 very good partial response, 2 partial responses, and 1 minimal response. The median progression-free survival in these patients was 10 months.
Grade 3 adverse events included decrease in neutrophil count (28%), decrease in lymphocyte count (28%), anemia (24%), fatigue (16%), hyperglycemia (12%), syncope (12%), decrease in white blood cell count (12%), decrease in platelet count (8%), increase in lymphocyte count (8%), nausea (4%), vomiting (4%), diarrhea (4%), maculo-papular rash (4%), hypotension (4%), lung infection (4%), pain in extremity (4%), and urticaria (4%).
Grade 4 events included decrease in lymphocyte count (24%), decrease in neutrophil count (8%), decrease in white blood cell count (8%), hyperuricemia (4%), decrease in platelet count (4%), and sepsis (4%).
Based on these results, the investigators said the combination of LCL161 and cyclophosphamide is “an attractive platform for future trials,” and the same is true for LCL161 in combination with anti-PD1 therapy.
The phase 2 trial was sponsored by Mayo Clinic and the National Cancer Institute. Novartis provided LCL161 for this research and supported the trial.
Transfusion practice may harm certain patients
A retrospective study indicates that a high transfusion ratio of fresh frozen plasma (FFP) to red blood cells (RBCs) may not be beneficial for surgical patients who do not have traumatic injuries.
In fact, the data suggest a high FFP:RBC ratio may be harmful for some of these patients.
“The strategy of giving patients requiring massive transfusion greater amounts of fresh frozen plasma, relative to the amount of red blood cells, has spilled over from trauma patients into unstudied patient populations,” said Daniel Dante Yeh, MD, of Massachusetts General Hospital in Boston.
“This may have important consequences, since our results suggest that certain populations may be harmed by this practice.”
Dr Yeh and his colleagues reported these results in JAMA Surgery.
The researchers reviewed all massive transfusions performed at Massachusetts General Hospital from January 1, 2009, through December 31, 2012.
A transfusion qualified as “massive” if at least 10 units of RBCs were given in the first 24 hours after a patient’s admission to the operating room, emergency department, or intensive care unit.
The researchers included all patients who received massive transfusions during the study period and survived more than 30 minutes after hospital arrival.
According to these criteria, there were 865 massive transfusion events. The total number of units transfused was 16,569 for RBCs, 13,933 for FFP, 5228 for cryoprecipitate, and 22,635 for platelets.
A majority of the massive transfusion recipients were not trauma patients (88.7%). Most of the transfusions were performed for intraoperative bleeding (62.9%).
The researchers compared patients who survived at least 30 days from hospital arrival to those who did not. Patients who died were older and received more RBCs (P<0.001), FFP (P<0.001), and cryoprecipitate (P=0.008).
However, the FFP:RBC ratios of survivors and non-survivors were similar. The median was 1:1.5 for survivors and 1:1.4 for non-survivors (P=0.43).
Patients without trauma
Among all non-trauma patients (n=767), there was no significant difference in the adjusted odds ratio (aOR) for 30-day mortality between patients who received a transfusion with a high FFP:RBC ratio and those who received one with a low FFP:RBC ratio (aOR=1.10, P=0.65). (The analysis was adjusted for patient age and total units of RBCs transfused.)
Among patients undergoing vascular surgery, those who received transfusions with a high FFP:RBC ratio were less likely to die within 30 days than those who received transfusions with a low FFP:RBC ratio (aOR=0.16, P=0.02).
However, among general surgery and medical service patients, those receiving transfusions with a high FFP:RBC ratio were more likely to die within 30 days than those receiving low-ratio transfusions. The aOR was 4.27 (P=0.02) for general surgery and 8.48 (P=0.02) for medicine.
“Finding evidence of increased mortality in some patients was surprising because that is directly contradictory to what is expected and intended,” Dr Yeh said. “Avoiding unnecessary FFP transfusion is important because there have been reports that associated the use of excess FFP with worse outcomes among patients that required less-than-massive transfusions.”
“Because our study is retrospective, it can only point to the need for further research. Ratio-based transfusion has been studied in trauma patients, most recently in a landmark, multicenter, randomized study called the PROPRR trial. Similar studies now need to be performed in non-trauma patients before the approach can be accepted as standard practice here at MGH [Massachusetts General Hospital] and elsewhere.”
A retrospective study indicates that a high transfusion ratio of fresh frozen plasma (FFP) to red blood cells (RBCs) may not be beneficial for surgical patients who do not have traumatic injuries.
In fact, the data suggest a high FFP:RBC ratio may be harmful for some of these patients.
“The strategy of giving patients requiring massive transfusion greater amounts of fresh frozen plasma, relative to the amount of red blood cells, has spilled over from trauma patients into unstudied patient populations,” said Daniel Dante Yeh, MD, of Massachusetts General Hospital in Boston.
“This may have important consequences, since our results suggest that certain populations may be harmed by this practice.”
Dr Yeh and his colleagues reported these results in JAMA Surgery.
The researchers reviewed all massive transfusions performed at Massachusetts General Hospital from January 1, 2009, through December 31, 2012.
A transfusion qualified as “massive” if at least 10 units of RBCs were given in the first 24 hours after a patient’s admission to the operating room, emergency department, or intensive care unit.
The researchers included all patients who received massive transfusions during the study period and survived more than 30 minutes after hospital arrival.
According to these criteria, there were 865 massive transfusion events. The total number of units transfused was 16,569 for RBCs, 13,933 for FFP, 5228 for cryoprecipitate, and 22,635 for platelets.
A majority of the massive transfusion recipients were not trauma patients (88.7%). Most of the transfusions were performed for intraoperative bleeding (62.9%).
The researchers compared patients who survived at least 30 days from hospital arrival to those who did not. Patients who died were older and received more RBCs (P<0.001), FFP (P<0.001), and cryoprecipitate (P=0.008).
However, the FFP:RBC ratios of survivors and non-survivors were similar. The median was 1:1.5 for survivors and 1:1.4 for non-survivors (P=0.43).
Patients without trauma
Among all non-trauma patients (n=767), there was no significant difference in the adjusted odds ratio (aOR) for 30-day mortality between patients who received a transfusion with a high FFP:RBC ratio and those who received one with a low FFP:RBC ratio (aOR=1.10, P=0.65). (The analysis was adjusted for patient age and total units of RBCs transfused.)
Among patients undergoing vascular surgery, those who received transfusions with a high FFP:RBC ratio were less likely to die within 30 days than those who received transfusions with a low FFP:RBC ratio (aOR=0.16, P=0.02).
However, among general surgery and medical service patients, those receiving transfusions with a high FFP:RBC ratio were more likely to die within 30 days than those receiving low-ratio transfusions. The aOR was 4.27 (P=0.02) for general surgery and 8.48 (P=0.02) for medicine.
“Finding evidence of increased mortality in some patients was surprising because that is directly contradictory to what is expected and intended,” Dr Yeh said. “Avoiding unnecessary FFP transfusion is important because there have been reports that associated the use of excess FFP with worse outcomes among patients that required less-than-massive transfusions.”
“Because our study is retrospective, it can only point to the need for further research. Ratio-based transfusion has been studied in trauma patients, most recently in a landmark, multicenter, randomized study called the PROPRR trial. Similar studies now need to be performed in non-trauma patients before the approach can be accepted as standard practice here at MGH [Massachusetts General Hospital] and elsewhere.”
A retrospective study indicates that a high transfusion ratio of fresh frozen plasma (FFP) to red blood cells (RBCs) may not be beneficial for surgical patients who do not have traumatic injuries.
In fact, the data suggest a high FFP:RBC ratio may be harmful for some of these patients.
“The strategy of giving patients requiring massive transfusion greater amounts of fresh frozen plasma, relative to the amount of red blood cells, has spilled over from trauma patients into unstudied patient populations,” said Daniel Dante Yeh, MD, of Massachusetts General Hospital in Boston.
“This may have important consequences, since our results suggest that certain populations may be harmed by this practice.”
Dr Yeh and his colleagues reported these results in JAMA Surgery.
The researchers reviewed all massive transfusions performed at Massachusetts General Hospital from January 1, 2009, through December 31, 2012.
A transfusion qualified as “massive” if at least 10 units of RBCs were given in the first 24 hours after a patient’s admission to the operating room, emergency department, or intensive care unit.
The researchers included all patients who received massive transfusions during the study period and survived more than 30 minutes after hospital arrival.
According to these criteria, there were 865 massive transfusion events. The total number of units transfused was 16,569 for RBCs, 13,933 for FFP, 5228 for cryoprecipitate, and 22,635 for platelets.
A majority of the massive transfusion recipients were not trauma patients (88.7%). Most of the transfusions were performed for intraoperative bleeding (62.9%).
The researchers compared patients who survived at least 30 days from hospital arrival to those who did not. Patients who died were older and received more RBCs (P<0.001), FFP (P<0.001), and cryoprecipitate (P=0.008).
However, the FFP:RBC ratios of survivors and non-survivors were similar. The median was 1:1.5 for survivors and 1:1.4 for non-survivors (P=0.43).
Patients without trauma
Among all non-trauma patients (n=767), there was no significant difference in the adjusted odds ratio (aOR) for 30-day mortality between patients who received a transfusion with a high FFP:RBC ratio and those who received one with a low FFP:RBC ratio (aOR=1.10, P=0.65). (The analysis was adjusted for patient age and total units of RBCs transfused.)
Among patients undergoing vascular surgery, those who received transfusions with a high FFP:RBC ratio were less likely to die within 30 days than those who received transfusions with a low FFP:RBC ratio (aOR=0.16, P=0.02).
However, among general surgery and medical service patients, those receiving transfusions with a high FFP:RBC ratio were more likely to die within 30 days than those receiving low-ratio transfusions. The aOR was 4.27 (P=0.02) for general surgery and 8.48 (P=0.02) for medicine.
“Finding evidence of increased mortality in some patients was surprising because that is directly contradictory to what is expected and intended,” Dr Yeh said. “Avoiding unnecessary FFP transfusion is important because there have been reports that associated the use of excess FFP with worse outcomes among patients that required less-than-massive transfusions.”
“Because our study is retrospective, it can only point to the need for further research. Ratio-based transfusion has been studied in trauma patients, most recently in a landmark, multicenter, randomized study called the PROPRR trial. Similar studies now need to be performed in non-trauma patients before the approach can be accepted as standard practice here at MGH [Massachusetts General Hospital] and elsewhere.”
FDA lifts clinical hold on AML trials
The US Food and Drug Administration (FDA) has lifted the clinical hold placed on 3 trials of vadastuximab talirine (SGN-CD33A), an antibody-drug conjugate targeting CD33, in acute myeloid leukemia (AML).
Last December, 1 trial was placed on full clinical hold (enrollment was halted and no further dosing of subjects was allowed), and 2 were placed on partial hold (enrollment was halted, but existing patients could continue treatment with re-consent).
All 3 of the holds were due to the potential risk of hepatotoxicity in patients who underwent allogeneic hematopoietic stem cell transplant (HSCT) before or after treatment with vadastuximab talirine.
In particular, the holds were in response to 6 cases of hepatotoxicity, including several cases of veno-occlusive disease (VOD), with 4 fatal events.
At the time the holds were announced, Seattle Genetics, Inc., the company developing vadastuximab talirine, said it was working with the FDA to determine whether there is an association between hepatotoxicity and treatment with the drug.
The company analyzed data from more than 350 patients treated with vadastuximab talirine and found no such association.
The rate of VOD they observed was “within the background rate of VOD in AML patients receiving allo-transplant,” according to Clay B. Siegall, PhD, president, chief executive officer, and chairman of the board at Seattle Genetics.
Dr Siegall said the company would not disclose the exact rate of VOD in these trials.
Resuming trials
As Seattle Genetics found no evidence to suggest that vadastuximab talirine increased the risk of hepatotoxicity, the FDA lifted the clinical holds on all 3 trials. The 2 trials placed on partial hold will continue, but the trial placed on full hold will not.
One of the trials that will continue is a phase 1 study of vadastuximab talirine alone and in combination with hypomethylating agents in both newly diagnosed and relapsed AML patients.
The other trial is a phase 1 study of vadastuximab talirine in combination with 7+3 chemotherapy in newly diagnosed, younger AML patients. (Results from this trial were presented at the 2016 ASH Annual Meeting.)
Cancelled trial
The trial that will not resume is a phase 1/2 study of vadastuximab talirine monotherapy pre- and post-allogeneic HSCT in patients with relapsed, chemo-resistant AML.
Seattle Genetics said it will not continue with this trial because of the challenges of developing therapies in this specific setting.
“It’s a very small group of patients, and we’re going to focus on the 3 biggest groups of patients [older and younger patients newly diagnosed with AML and patients with myelodysplastic syndromes] so we can really impact AML in the biggest way,” Dr Siegall said.
He noted that this decision does not prevent patients from undergoing HSCT after receiving vadastuximab talirine.
In the phase 1/2 trial, patients received vadastuximab talirine directly before HSCT, a practice that will not continue. However, patients can undergo HSCT as long as the transplant doesn’t occur immediately after treatment with vadastuximab talirine.
Moving forward
Two other trials of vadastuximab talirine were not affected by the clinical holds and have continued to enroll patients.
One is CASCADE, a randomized, phase 3 trial of vadastuximab talirine as front-line therapy in older AML patients. The other is a phase 1/2 trial of vadastuximab talirine as front-line therapy in patients with myelodysplastic syndromes.
Seattle Genetics is also planning to begin a randomized, phase 2 trial comparing 7+3 chemotherapy alone to 7+3 in combination with vadastuximab talirine in younger patients with previously untreated AML. The company plans to start the trial later this year.
Going forward, additional risk mitigation measures will be implemented in all vadastuximab talirine studies, including revised eligibility criteria and stopping rules for VOD.
Specifically, trials will not be stopped if the incidence of VOD is considered within the normal range, and an adjudication committee consisting of 2 experts will be tasked with verifying reports of VOD.
In addition, patients with liver cirrhosis due to alcohol abuse are no longer eligible for trials of vadastuximab talirine.
The US Food and Drug Administration (FDA) has lifted the clinical hold placed on 3 trials of vadastuximab talirine (SGN-CD33A), an antibody-drug conjugate targeting CD33, in acute myeloid leukemia (AML).
Last December, 1 trial was placed on full clinical hold (enrollment was halted and no further dosing of subjects was allowed), and 2 were placed on partial hold (enrollment was halted, but existing patients could continue treatment with re-consent).
All 3 of the holds were due to the potential risk of hepatotoxicity in patients who underwent allogeneic hematopoietic stem cell transplant (HSCT) before or after treatment with vadastuximab talirine.
In particular, the holds were in response to 6 cases of hepatotoxicity, including several cases of veno-occlusive disease (VOD), with 4 fatal events.
At the time the holds were announced, Seattle Genetics, Inc., the company developing vadastuximab talirine, said it was working with the FDA to determine whether there is an association between hepatotoxicity and treatment with the drug.
The company analyzed data from more than 350 patients treated with vadastuximab talirine and found no such association.
The rate of VOD they observed was “within the background rate of VOD in AML patients receiving allo-transplant,” according to Clay B. Siegall, PhD, president, chief executive officer, and chairman of the board at Seattle Genetics.
Dr Siegall said the company would not disclose the exact rate of VOD in these trials.
Resuming trials
As Seattle Genetics found no evidence to suggest that vadastuximab talirine increased the risk of hepatotoxicity, the FDA lifted the clinical holds on all 3 trials. The 2 trials placed on partial hold will continue, but the trial placed on full hold will not.
One of the trials that will continue is a phase 1 study of vadastuximab talirine alone and in combination with hypomethylating agents in both newly diagnosed and relapsed AML patients.
The other trial is a phase 1 study of vadastuximab talirine in combination with 7+3 chemotherapy in newly diagnosed, younger AML patients. (Results from this trial were presented at the 2016 ASH Annual Meeting.)
Cancelled trial
The trial that will not resume is a phase 1/2 study of vadastuximab talirine monotherapy pre- and post-allogeneic HSCT in patients with relapsed, chemo-resistant AML.
Seattle Genetics said it will not continue with this trial because of the challenges of developing therapies in this specific setting.
“It’s a very small group of patients, and we’re going to focus on the 3 biggest groups of patients [older and younger patients newly diagnosed with AML and patients with myelodysplastic syndromes] so we can really impact AML in the biggest way,” Dr Siegall said.
He noted that this decision does not prevent patients from undergoing HSCT after receiving vadastuximab talirine.
In the phase 1/2 trial, patients received vadastuximab talirine directly before HSCT, a practice that will not continue. However, patients can undergo HSCT as long as the transplant doesn’t occur immediately after treatment with vadastuximab talirine.
Moving forward
Two other trials of vadastuximab talirine were not affected by the clinical holds and have continued to enroll patients.
One is CASCADE, a randomized, phase 3 trial of vadastuximab talirine as front-line therapy in older AML patients. The other is a phase 1/2 trial of vadastuximab talirine as front-line therapy in patients with myelodysplastic syndromes.
Seattle Genetics is also planning to begin a randomized, phase 2 trial comparing 7+3 chemotherapy alone to 7+3 in combination with vadastuximab talirine in younger patients with previously untreated AML. The company plans to start the trial later this year.
Going forward, additional risk mitigation measures will be implemented in all vadastuximab talirine studies, including revised eligibility criteria and stopping rules for VOD.
Specifically, trials will not be stopped if the incidence of VOD is considered within the normal range, and an adjudication committee consisting of 2 experts will be tasked with verifying reports of VOD.
In addition, patients with liver cirrhosis due to alcohol abuse are no longer eligible for trials of vadastuximab talirine.
The US Food and Drug Administration (FDA) has lifted the clinical hold placed on 3 trials of vadastuximab talirine (SGN-CD33A), an antibody-drug conjugate targeting CD33, in acute myeloid leukemia (AML).
Last December, 1 trial was placed on full clinical hold (enrollment was halted and no further dosing of subjects was allowed), and 2 were placed on partial hold (enrollment was halted, but existing patients could continue treatment with re-consent).
All 3 of the holds were due to the potential risk of hepatotoxicity in patients who underwent allogeneic hematopoietic stem cell transplant (HSCT) before or after treatment with vadastuximab talirine.
In particular, the holds were in response to 6 cases of hepatotoxicity, including several cases of veno-occlusive disease (VOD), with 4 fatal events.
At the time the holds were announced, Seattle Genetics, Inc., the company developing vadastuximab talirine, said it was working with the FDA to determine whether there is an association between hepatotoxicity and treatment with the drug.
The company analyzed data from more than 350 patients treated with vadastuximab talirine and found no such association.
The rate of VOD they observed was “within the background rate of VOD in AML patients receiving allo-transplant,” according to Clay B. Siegall, PhD, president, chief executive officer, and chairman of the board at Seattle Genetics.
Dr Siegall said the company would not disclose the exact rate of VOD in these trials.
Resuming trials
As Seattle Genetics found no evidence to suggest that vadastuximab talirine increased the risk of hepatotoxicity, the FDA lifted the clinical holds on all 3 trials. The 2 trials placed on partial hold will continue, but the trial placed on full hold will not.
One of the trials that will continue is a phase 1 study of vadastuximab talirine alone and in combination with hypomethylating agents in both newly diagnosed and relapsed AML patients.
The other trial is a phase 1 study of vadastuximab talirine in combination with 7+3 chemotherapy in newly diagnosed, younger AML patients. (Results from this trial were presented at the 2016 ASH Annual Meeting.)
Cancelled trial
The trial that will not resume is a phase 1/2 study of vadastuximab talirine monotherapy pre- and post-allogeneic HSCT in patients with relapsed, chemo-resistant AML.
Seattle Genetics said it will not continue with this trial because of the challenges of developing therapies in this specific setting.
“It’s a very small group of patients, and we’re going to focus on the 3 biggest groups of patients [older and younger patients newly diagnosed with AML and patients with myelodysplastic syndromes] so we can really impact AML in the biggest way,” Dr Siegall said.
He noted that this decision does not prevent patients from undergoing HSCT after receiving vadastuximab talirine.
In the phase 1/2 trial, patients received vadastuximab talirine directly before HSCT, a practice that will not continue. However, patients can undergo HSCT as long as the transplant doesn’t occur immediately after treatment with vadastuximab talirine.
Moving forward
Two other trials of vadastuximab talirine were not affected by the clinical holds and have continued to enroll patients.
One is CASCADE, a randomized, phase 3 trial of vadastuximab talirine as front-line therapy in older AML patients. The other is a phase 1/2 trial of vadastuximab talirine as front-line therapy in patients with myelodysplastic syndromes.
Seattle Genetics is also planning to begin a randomized, phase 2 trial comparing 7+3 chemotherapy alone to 7+3 in combination with vadastuximab talirine in younger patients with previously untreated AML. The company plans to start the trial later this year.
Going forward, additional risk mitigation measures will be implemented in all vadastuximab talirine studies, including revised eligibility criteria and stopping rules for VOD.
Specifically, trials will not be stopped if the incidence of VOD is considered within the normal range, and an adjudication committee consisting of 2 experts will be tasked with verifying reports of VOD.
In addition, patients with liver cirrhosis due to alcohol abuse are no longer eligible for trials of vadastuximab talirine.