User login
Tobacco plants used to manufacture malaria drug

Tobacco plants can be engineered to manufacture artemisinin at therapeutic levels, according research published in Molecular Plant.
The researchers noted that the majority of people who live in malaria-endemic areas cannot afford to buy artemisinin.
The drug’s high cost is due to the extraction process and the fact that it’s difficult to grow Artemisia annua, the original source of the drug, in climates where malaria is common.
Advances in synthetic biology have made it possible to produce artemisinin in yeast, but the manufacturing process is difficult to scale up.
Earlier studies showed that artemisinin can be grown in tobacco—a plant that’s relatively easy to genetically manipulate and that grows well in areas where malaria is endemic. But yields of artemisinin from those plants were low.
Now, Shashi Kumar, PhD, of the International Centre for Genetic
Engineering and Biotechnology in New Delhi, India, and his colleagues say they have overcome this problem.
In the Molecular Plant paper, Dr Kumar and his colleagues reported using a dual-transformation approach to boost the production of artemisinin in the tobacco plants.
The team first generated plants that contained transgenic chloroplasts, and the same plants were then manipulated again to insert genes into the nuclear genome as well.
Extract from the plants was shown to stop the growth of Plasmodium falciparum in vitro. Whole cells from the plant were also fed to mice infected with Plasmodium berghei, which greatly reduced levels of the parasite in the blood.
In fact, the researchers found the whole plant material was more effective in attacking the parasite than pure artemisinin, likely because encapsulation inside the plant cells protected the compound from degradation by digestive enzymes.
The researchers acknowledged that convincing people to eat tobacco plants is likely to be a hard sell. For that reason, they are now aiming to genetically engineer lettuce plants to produce artemisinin at therapeutic levels.
They said the lettuce containing the drug could be freeze dried, ground into a powder, and put into capsules for cost-effective delivery.
“Plant and animal science are increasingly coming together,” Dr Kumar said. “In the near future, you will see more drugs produced inside plants will be commercialized to reduce the drug cost.” ![]()

Tobacco plants can be engineered to manufacture artemisinin at therapeutic levels, according research published in Molecular Plant.
The researchers noted that the majority of people who live in malaria-endemic areas cannot afford to buy artemisinin.
The drug’s high cost is due to the extraction process and the fact that it’s difficult to grow Artemisia annua, the original source of the drug, in climates where malaria is common.
Advances in synthetic biology have made it possible to produce artemisinin in yeast, but the manufacturing process is difficult to scale up.
Earlier studies showed that artemisinin can be grown in tobacco—a plant that’s relatively easy to genetically manipulate and that grows well in areas where malaria is endemic. But yields of artemisinin from those plants were low.
Now, Shashi Kumar, PhD, of the International Centre for Genetic
Engineering and Biotechnology in New Delhi, India, and his colleagues say they have overcome this problem.
In the Molecular Plant paper, Dr Kumar and his colleagues reported using a dual-transformation approach to boost the production of artemisinin in the tobacco plants.
The team first generated plants that contained transgenic chloroplasts, and the same plants were then manipulated again to insert genes into the nuclear genome as well.
Extract from the plants was shown to stop the growth of Plasmodium falciparum in vitro. Whole cells from the plant were also fed to mice infected with Plasmodium berghei, which greatly reduced levels of the parasite in the blood.
In fact, the researchers found the whole plant material was more effective in attacking the parasite than pure artemisinin, likely because encapsulation inside the plant cells protected the compound from degradation by digestive enzymes.
The researchers acknowledged that convincing people to eat tobacco plants is likely to be a hard sell. For that reason, they are now aiming to genetically engineer lettuce plants to produce artemisinin at therapeutic levels.
They said the lettuce containing the drug could be freeze dried, ground into a powder, and put into capsules for cost-effective delivery.
“Plant and animal science are increasingly coming together,” Dr Kumar said. “In the near future, you will see more drugs produced inside plants will be commercialized to reduce the drug cost.” ![]()

Tobacco plants can be engineered to manufacture artemisinin at therapeutic levels, according research published in Molecular Plant.
The researchers noted that the majority of people who live in malaria-endemic areas cannot afford to buy artemisinin.
The drug’s high cost is due to the extraction process and the fact that it’s difficult to grow Artemisia annua, the original source of the drug, in climates where malaria is common.
Advances in synthetic biology have made it possible to produce artemisinin in yeast, but the manufacturing process is difficult to scale up.
Earlier studies showed that artemisinin can be grown in tobacco—a plant that’s relatively easy to genetically manipulate and that grows well in areas where malaria is endemic. But yields of artemisinin from those plants were low.
Now, Shashi Kumar, PhD, of the International Centre for Genetic
Engineering and Biotechnology in New Delhi, India, and his colleagues say they have overcome this problem.
In the Molecular Plant paper, Dr Kumar and his colleagues reported using a dual-transformation approach to boost the production of artemisinin in the tobacco plants.
The team first generated plants that contained transgenic chloroplasts, and the same plants were then manipulated again to insert genes into the nuclear genome as well.
Extract from the plants was shown to stop the growth of Plasmodium falciparum in vitro. Whole cells from the plant were also fed to mice infected with Plasmodium berghei, which greatly reduced levels of the parasite in the blood.
In fact, the researchers found the whole plant material was more effective in attacking the parasite than pure artemisinin, likely because encapsulation inside the plant cells protected the compound from degradation by digestive enzymes.
The researchers acknowledged that convincing people to eat tobacco plants is likely to be a hard sell. For that reason, they are now aiming to genetically engineer lettuce plants to produce artemisinin at therapeutic levels.
They said the lettuce containing the drug could be freeze dried, ground into a powder, and put into capsules for cost-effective delivery.
“Plant and animal science are increasingly coming together,” Dr Kumar said. “In the near future, you will see more drugs produced inside plants will be commercialized to reduce the drug cost.” ![]()
Improving cognitive function in cancer survivors

chemotherapy
Photo by Rhoda Baer
A new study suggests a computer program known as InsightTM may help improve cognitive function and overall well-being in cancer survivors.
The study included subjects who reported persistent problems with concentration and/or memory after receiving chemotherapy.
Using the Insight program significantly improved the subjects’ self-reported cognitive function and lowered their levels of anxiety, depression, fatigue, and stress.
However, there was no significant difference in the results of objective neuropsychological function tests between subjects who used the Insight program and subjects who received standard care.
These results were published in the Journal of Clinical Oncology.
“To the best of our knowledge, this is the largest cognitive intervention study that has shown a benefit for patients who are reporting persistent cognitive symptoms following chemotherapy,” said study author Victoria J. Bray, MD, of the University of Sydney in Australia.
About the study
Dr Bray and her colleagues enrolled 242 adult cancer survivors in Australia who had completed at least 3 cycles of chemotherapy in the prior 6 months to 60 months and reported persistent cognitive symptoms.
The subjects’ median age was 53 (range, 23 to 74). Nearly all were women (95%), and 89% had survived breast cancer.
At the beginning of the study, all subjects received a personalized, 30-minute telephone consultation that provided tips and strategies for coping with cognitive problems in daily life.
Subjects were then randomized to Insight (used at home) or standard oncology care per their treating physician.
The primary outcome of the study was self-reported cognitive function, which was assessed using a validated questionnaire known as FACT-COG. It evaluates perceived cognitive impairments, perceived cognitive abilities, and the impact of perceived cognitive impairment on quality of life.
Separate measures were used to evaluate objective neuropsychological function, anxiety/depression, fatigue, and stress.
About the intervention
Dr Bray and her colleagues described Insight as a neurocognitive learning program that uses exercises intended to improve cognition through speed and accuracy of information processing.
The program targets visual precision, divided attention, working memory, field of view, and visual processing speed, which are frequently affected in patients with cancer.
Subjects received the Insight program in disc form and were advised to use the program for 40 minutes 4 times a week for 15 weeks (40 hours total). The program had a built-in measure that could determine subjects’ compliance.
Key findings
Self-reported cognitive function was significantly better in the Insight group than the standard care group, both at the end of the 15-week program and 6 months later.
In addition, Insight participants had significantly lower levels of anxiety, depression, and fatigue immediately after the intervention, significant improvements in quality of life at 6 months, and significant improvements in stress at both time points.
Results of objective neuropsychological function tests were not significantly different between the Insight group and the standard care group, either immediately after the intervention or at 6 months.
Next steps
The researchers said longer follow-up is needed to determine if the effects of the Insight program are long-lasting. And there are still a number of other unanswered questions to be addressed in future research.
For one, it is unclear which method of delivering cognitive rehabilitation is better. A self-directed program such as this one may be suitable for some cancer survivors, while a group-based program may work better for others. It’s also unclear what the ideal duration and “dose” of cognitive training should be.
“If we could identify patients who are at risk of cognitive impairment, we could intervene earlier and possibly achieve even better results,” Dr Bray said. “We would also like to explore whether there is added benefit from combining cognitive training with physical exercise.” ![]()

chemotherapy
Photo by Rhoda Baer
A new study suggests a computer program known as InsightTM may help improve cognitive function and overall well-being in cancer survivors.
The study included subjects who reported persistent problems with concentration and/or memory after receiving chemotherapy.
Using the Insight program significantly improved the subjects’ self-reported cognitive function and lowered their levels of anxiety, depression, fatigue, and stress.
However, there was no significant difference in the results of objective neuropsychological function tests between subjects who used the Insight program and subjects who received standard care.
These results were published in the Journal of Clinical Oncology.
“To the best of our knowledge, this is the largest cognitive intervention study that has shown a benefit for patients who are reporting persistent cognitive symptoms following chemotherapy,” said study author Victoria J. Bray, MD, of the University of Sydney in Australia.
About the study
Dr Bray and her colleagues enrolled 242 adult cancer survivors in Australia who had completed at least 3 cycles of chemotherapy in the prior 6 months to 60 months and reported persistent cognitive symptoms.
The subjects’ median age was 53 (range, 23 to 74). Nearly all were women (95%), and 89% had survived breast cancer.
At the beginning of the study, all subjects received a personalized, 30-minute telephone consultation that provided tips and strategies for coping with cognitive problems in daily life.
Subjects were then randomized to Insight (used at home) or standard oncology care per their treating physician.
The primary outcome of the study was self-reported cognitive function, which was assessed using a validated questionnaire known as FACT-COG. It evaluates perceived cognitive impairments, perceived cognitive abilities, and the impact of perceived cognitive impairment on quality of life.
Separate measures were used to evaluate objective neuropsychological function, anxiety/depression, fatigue, and stress.
About the intervention
Dr Bray and her colleagues described Insight as a neurocognitive learning program that uses exercises intended to improve cognition through speed and accuracy of information processing.
The program targets visual precision, divided attention, working memory, field of view, and visual processing speed, which are frequently affected in patients with cancer.
Subjects received the Insight program in disc form and were advised to use the program for 40 minutes 4 times a week for 15 weeks (40 hours total). The program had a built-in measure that could determine subjects’ compliance.
Key findings
Self-reported cognitive function was significantly better in the Insight group than the standard care group, both at the end of the 15-week program and 6 months later.
In addition, Insight participants had significantly lower levels of anxiety, depression, and fatigue immediately after the intervention, significant improvements in quality of life at 6 months, and significant improvements in stress at both time points.
Results of objective neuropsychological function tests were not significantly different between the Insight group and the standard care group, either immediately after the intervention or at 6 months.
Next steps
The researchers said longer follow-up is needed to determine if the effects of the Insight program are long-lasting. And there are still a number of other unanswered questions to be addressed in future research.
For one, it is unclear which method of delivering cognitive rehabilitation is better. A self-directed program such as this one may be suitable for some cancer survivors, while a group-based program may work better for others. It’s also unclear what the ideal duration and “dose” of cognitive training should be.
“If we could identify patients who are at risk of cognitive impairment, we could intervene earlier and possibly achieve even better results,” Dr Bray said. “We would also like to explore whether there is added benefit from combining cognitive training with physical exercise.” ![]()

chemotherapy
Photo by Rhoda Baer
A new study suggests a computer program known as InsightTM may help improve cognitive function and overall well-being in cancer survivors.
The study included subjects who reported persistent problems with concentration and/or memory after receiving chemotherapy.
Using the Insight program significantly improved the subjects’ self-reported cognitive function and lowered their levels of anxiety, depression, fatigue, and stress.
However, there was no significant difference in the results of objective neuropsychological function tests between subjects who used the Insight program and subjects who received standard care.
These results were published in the Journal of Clinical Oncology.
“To the best of our knowledge, this is the largest cognitive intervention study that has shown a benefit for patients who are reporting persistent cognitive symptoms following chemotherapy,” said study author Victoria J. Bray, MD, of the University of Sydney in Australia.
About the study
Dr Bray and her colleagues enrolled 242 adult cancer survivors in Australia who had completed at least 3 cycles of chemotherapy in the prior 6 months to 60 months and reported persistent cognitive symptoms.
The subjects’ median age was 53 (range, 23 to 74). Nearly all were women (95%), and 89% had survived breast cancer.
At the beginning of the study, all subjects received a personalized, 30-minute telephone consultation that provided tips and strategies for coping with cognitive problems in daily life.
Subjects were then randomized to Insight (used at home) or standard oncology care per their treating physician.
The primary outcome of the study was self-reported cognitive function, which was assessed using a validated questionnaire known as FACT-COG. It evaluates perceived cognitive impairments, perceived cognitive abilities, and the impact of perceived cognitive impairment on quality of life.
Separate measures were used to evaluate objective neuropsychological function, anxiety/depression, fatigue, and stress.
About the intervention
Dr Bray and her colleagues described Insight as a neurocognitive learning program that uses exercises intended to improve cognition through speed and accuracy of information processing.
The program targets visual precision, divided attention, working memory, field of view, and visual processing speed, which are frequently affected in patients with cancer.
Subjects received the Insight program in disc form and were advised to use the program for 40 minutes 4 times a week for 15 weeks (40 hours total). The program had a built-in measure that could determine subjects’ compliance.
Key findings
Self-reported cognitive function was significantly better in the Insight group than the standard care group, both at the end of the 15-week program and 6 months later.
In addition, Insight participants had significantly lower levels of anxiety, depression, and fatigue immediately after the intervention, significant improvements in quality of life at 6 months, and significant improvements in stress at both time points.
Results of objective neuropsychological function tests were not significantly different between the Insight group and the standard care group, either immediately after the intervention or at 6 months.
Next steps
The researchers said longer follow-up is needed to determine if the effects of the Insight program are long-lasting. And there are still a number of other unanswered questions to be addressed in future research.
For one, it is unclear which method of delivering cognitive rehabilitation is better. A self-directed program such as this one may be suitable for some cancer survivors, while a group-based program may work better for others. It’s also unclear what the ideal duration and “dose” of cognitive training should be.
“If we could identify patients who are at risk of cognitive impairment, we could intervene earlier and possibly achieve even better results,” Dr Bray said. “We would also like to explore whether there is added benefit from combining cognitive training with physical exercise.” ![]()
Team maps genomic landscape of CBF-AML

whole-genome sequencing
Photo courtesy of the US
Food and Drug Administration
Whole-genome and whole-exome sequencing has provided new insight into the pathogenesis and development of core-binding factor acute myeloid leukemia (CBF-AML), according to researchers.
The team said their work has revealed “dramatic” differences in the genomic landscape of CBF-AMLs that contribute to the diversity of this disease.
The researchers reported their findings in Nature Genetics.
“We set out to understand the genetic variations that contribute to the development of CBF-AML using whole-exome and whole-genome sequencing,” said study author Jeffery Klco, MD, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
“Our goal was to define a detailed mutational landscape to understand better the genetic changes that contribute to disease.”
Dr Klco and his colleagues sequenced samples from 87 children and 78 adults with CBF-AML. Eighty-five of the patients had the RUNX1-RUNX1T1 subtype, and 80 had the CBFB-MYH11 subtype.
Development and relapse
The researchers identified several genes with mutations that may contribute to CBF-AML development, including CCND2, DHX15, ASXL2, ZBTB7A, and MGA.
“Many of the mutations we identified interfered with molecular signaling or epigenetic factors,” said study author Jinghui Zhang, PhD, of St. Jude Children’s Research Hospital.
“Some of the mutations, like ASXL2, are epigenetic regulators that modify the local state of chromatin,” Dr Klco noted. “Others, like ZBTB7A, appear to act like tumor suppressors.”
The researchers also compared mutations present at diagnosis and relapse in an attempt to understand how CBF-AML changes over time.
Their results suggested that KMT2C mutations are associated with relapse. Of the 4 patients in this study who had KMT2C mutations, 3 relapsed in less than 12 months, and the fourth had residual disease after a course of remission-induction therapy.
Similarities and differences
The researchers found a similar mutational landscape in adults and children with CBF-AML but differences between patients with the RUNX1-RUNX1T1 and CBFB-MYH11 subtypes.
NRAS was the most frequently mutated gene in CBF-AMLs, but NRAS mutations were more common in CBFB-MYH11 AML than RUNX1-RUNX1T1 AML. The same was true for mutations in NF1 and WT1.
Patients with both subtypes of CBF-AML had mutations in NRAS, KIT, NF1, WT1, FLT3, KRAS, MGA, TTN, CCND2, KDM6A, PHIP, TET2, HCN1, KMT2C, and SETD2.
But only patients with CBFB-MYH11 AML had mutations in PTPN11.
Only patients with RUNX1-RUNX1T1 AML had mutations in ASXL2, ZBTB7A, EZH2, SMC1A, DHX15, RAD21, CBL, DNM2, CSF3R, GIGYF2, SMC3, and ZNF687.
The researchers said their findings suggest a range of mutations may play roles in CBF-AML, but additional research is needed to confirm their precise function in the disease.
Further studies are already underway to fully evaluate the contributions of the different genes as well as the roles of the newly identified genetic alterations in CBF-AML. ![]()

whole-genome sequencing
Photo courtesy of the US
Food and Drug Administration
Whole-genome and whole-exome sequencing has provided new insight into the pathogenesis and development of core-binding factor acute myeloid leukemia (CBF-AML), according to researchers.
The team said their work has revealed “dramatic” differences in the genomic landscape of CBF-AMLs that contribute to the diversity of this disease.
The researchers reported their findings in Nature Genetics.
“We set out to understand the genetic variations that contribute to the development of CBF-AML using whole-exome and whole-genome sequencing,” said study author Jeffery Klco, MD, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
“Our goal was to define a detailed mutational landscape to understand better the genetic changes that contribute to disease.”
Dr Klco and his colleagues sequenced samples from 87 children and 78 adults with CBF-AML. Eighty-five of the patients had the RUNX1-RUNX1T1 subtype, and 80 had the CBFB-MYH11 subtype.
Development and relapse
The researchers identified several genes with mutations that may contribute to CBF-AML development, including CCND2, DHX15, ASXL2, ZBTB7A, and MGA.
“Many of the mutations we identified interfered with molecular signaling or epigenetic factors,” said study author Jinghui Zhang, PhD, of St. Jude Children’s Research Hospital.
“Some of the mutations, like ASXL2, are epigenetic regulators that modify the local state of chromatin,” Dr Klco noted. “Others, like ZBTB7A, appear to act like tumor suppressors.”
The researchers also compared mutations present at diagnosis and relapse in an attempt to understand how CBF-AML changes over time.
Their results suggested that KMT2C mutations are associated with relapse. Of the 4 patients in this study who had KMT2C mutations, 3 relapsed in less than 12 months, and the fourth had residual disease after a course of remission-induction therapy.
Similarities and differences
The researchers found a similar mutational landscape in adults and children with CBF-AML but differences between patients with the RUNX1-RUNX1T1 and CBFB-MYH11 subtypes.
NRAS was the most frequently mutated gene in CBF-AMLs, but NRAS mutations were more common in CBFB-MYH11 AML than RUNX1-RUNX1T1 AML. The same was true for mutations in NF1 and WT1.
Patients with both subtypes of CBF-AML had mutations in NRAS, KIT, NF1, WT1, FLT3, KRAS, MGA, TTN, CCND2, KDM6A, PHIP, TET2, HCN1, KMT2C, and SETD2.
But only patients with CBFB-MYH11 AML had mutations in PTPN11.
Only patients with RUNX1-RUNX1T1 AML had mutations in ASXL2, ZBTB7A, EZH2, SMC1A, DHX15, RAD21, CBL, DNM2, CSF3R, GIGYF2, SMC3, and ZNF687.
The researchers said their findings suggest a range of mutations may play roles in CBF-AML, but additional research is needed to confirm their precise function in the disease.
Further studies are already underway to fully evaluate the contributions of the different genes as well as the roles of the newly identified genetic alterations in CBF-AML. ![]()

whole-genome sequencing
Photo courtesy of the US
Food and Drug Administration
Whole-genome and whole-exome sequencing has provided new insight into the pathogenesis and development of core-binding factor acute myeloid leukemia (CBF-AML), according to researchers.
The team said their work has revealed “dramatic” differences in the genomic landscape of CBF-AMLs that contribute to the diversity of this disease.
The researchers reported their findings in Nature Genetics.
“We set out to understand the genetic variations that contribute to the development of CBF-AML using whole-exome and whole-genome sequencing,” said study author Jeffery Klco, MD, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
“Our goal was to define a detailed mutational landscape to understand better the genetic changes that contribute to disease.”
Dr Klco and his colleagues sequenced samples from 87 children and 78 adults with CBF-AML. Eighty-five of the patients had the RUNX1-RUNX1T1 subtype, and 80 had the CBFB-MYH11 subtype.
Development and relapse
The researchers identified several genes with mutations that may contribute to CBF-AML development, including CCND2, DHX15, ASXL2, ZBTB7A, and MGA.
“Many of the mutations we identified interfered with molecular signaling or epigenetic factors,” said study author Jinghui Zhang, PhD, of St. Jude Children’s Research Hospital.
“Some of the mutations, like ASXL2, are epigenetic regulators that modify the local state of chromatin,” Dr Klco noted. “Others, like ZBTB7A, appear to act like tumor suppressors.”
The researchers also compared mutations present at diagnosis and relapse in an attempt to understand how CBF-AML changes over time.
Their results suggested that KMT2C mutations are associated with relapse. Of the 4 patients in this study who had KMT2C mutations, 3 relapsed in less than 12 months, and the fourth had residual disease after a course of remission-induction therapy.
Similarities and differences
The researchers found a similar mutational landscape in adults and children with CBF-AML but differences between patients with the RUNX1-RUNX1T1 and CBFB-MYH11 subtypes.
NRAS was the most frequently mutated gene in CBF-AMLs, but NRAS mutations were more common in CBFB-MYH11 AML than RUNX1-RUNX1T1 AML. The same was true for mutations in NF1 and WT1.
Patients with both subtypes of CBF-AML had mutations in NRAS, KIT, NF1, WT1, FLT3, KRAS, MGA, TTN, CCND2, KDM6A, PHIP, TET2, HCN1, KMT2C, and SETD2.
But only patients with CBFB-MYH11 AML had mutations in PTPN11.
Only patients with RUNX1-RUNX1T1 AML had mutations in ASXL2, ZBTB7A, EZH2, SMC1A, DHX15, RAD21, CBL, DNM2, CSF3R, GIGYF2, SMC3, and ZNF687.
The researchers said their findings suggest a range of mutations may play roles in CBF-AML, but additional research is needed to confirm their precise function in the disease.
Further studies are already underway to fully evaluate the contributions of the different genes as well as the roles of the newly identified genetic alterations in CBF-AML. ![]()
Continuous Rd should be standard of care, group says

Photo courtesy of Celgene
Updated trial results support continuous treatment with lenalidomide and low-dose dexamethasone (Rd) as a standard of care for patients of all ages who have newly diagnosed multiple myeloma (MM) and are ineligible for stem cell transplant, according to researchers.
In this phase 3 trial, patients who received continuous Rd (until disease progression) had better progression-free survival (PFS) and overall survival (OS) than patients who received 18 cycles of Rd (Rd18) or a combination of melphalan, prednisone, and thalidomide (MPT).
Updated results from this study, known as the FIRST trial, were published in the Journal of Clinical Oncology.
Results were previously published in NEJM in 2014. The study was supported by Intergroupe Francophone du Myélome and Celgene Corporation, the makers of lenalidomide.
Thierry Facon, MD, of Centre Hospitalier Regional Universitaire de Lille in France, and his colleagues enrolled 1623 patients on this trial. They were newly diagnosed with MM and not eligible for stem cell transplant.
Patients were randomized to receive Rd in 28-day cycles until disease progression (n=535), Rd18 for 72 weeks (n=541), or MPT for 72 weeks (n=547).
Response
In the intent-to-treat population, the overall response rate was 81% for the continuous Rd group, 79% for the Rd18 group, and 67% in the MPT group. The complete response rates were 21%, 20%, and 12%, respectively.
The median duration of response was 32 months (range, 26-37) in the continuous Rd group, 22 months (range, 19-23) in the Rd18 group, and 22 months (range, 20-25) in the MPT group.
PFS and OS
The median PFS was 26.0 months in the continuous Rd group, 21.0 months in the Rd18 group, and 21.9 months in the MPT group. At 4 years, the PFS rate was 33%, 14%, and 13%, respectively.
The hazard ratio (HR) for continuous Rd vs MPT was 0.69. The HR for continuous Rd vs Rd18 was 0.71. And the HR for Rd18 vs MPT was 0.99.
The median OS was 58.9 months in the continuous Rd group, 56.7 months in the Rd18 group, and 48.5 months in the MPT group. At 4 years, the OS rate was 60%, 57%, and 51%, respectively.
The HR for continuous Rd vs MPT was 0.75. The HR for continuous Rd vs Rd18 was 0.91. And the HR for Rd18 vs MPT was 0.83.
Safety
The most frequent grade 3/4 hematologic treatment-emergent adverse events were neutropenia and anemia. The rate of grade 3/4 neutropenia was higher in the MPT group than the continuous Rd or Rd18 groups.
Infections were the most common grade 3/4 non-hematologic treatment-emergent adverse events. The rate of grade 3/4 infections was higher in the Rd groups than the MPT group.
Grade 3/4 peripheral sensory neuropathy was less frequent in the continuous Rd and Rd18 groups than the MPT group.
The incidence of invasive second primary malignancy was 4% in the continuous Rd group, 6% in the Rd18 group, and 6% in the MPT group. ![]()

Photo courtesy of Celgene
Updated trial results support continuous treatment with lenalidomide and low-dose dexamethasone (Rd) as a standard of care for patients of all ages who have newly diagnosed multiple myeloma (MM) and are ineligible for stem cell transplant, according to researchers.
In this phase 3 trial, patients who received continuous Rd (until disease progression) had better progression-free survival (PFS) and overall survival (OS) than patients who received 18 cycles of Rd (Rd18) or a combination of melphalan, prednisone, and thalidomide (MPT).
Updated results from this study, known as the FIRST trial, were published in the Journal of Clinical Oncology.
Results were previously published in NEJM in 2014. The study was supported by Intergroupe Francophone du Myélome and Celgene Corporation, the makers of lenalidomide.
Thierry Facon, MD, of Centre Hospitalier Regional Universitaire de Lille in France, and his colleagues enrolled 1623 patients on this trial. They were newly diagnosed with MM and not eligible for stem cell transplant.
Patients were randomized to receive Rd in 28-day cycles until disease progression (n=535), Rd18 for 72 weeks (n=541), or MPT for 72 weeks (n=547).
Response
In the intent-to-treat population, the overall response rate was 81% for the continuous Rd group, 79% for the Rd18 group, and 67% in the MPT group. The complete response rates were 21%, 20%, and 12%, respectively.
The median duration of response was 32 months (range, 26-37) in the continuous Rd group, 22 months (range, 19-23) in the Rd18 group, and 22 months (range, 20-25) in the MPT group.
PFS and OS
The median PFS was 26.0 months in the continuous Rd group, 21.0 months in the Rd18 group, and 21.9 months in the MPT group. At 4 years, the PFS rate was 33%, 14%, and 13%, respectively.
The hazard ratio (HR) for continuous Rd vs MPT was 0.69. The HR for continuous Rd vs Rd18 was 0.71. And the HR for Rd18 vs MPT was 0.99.
The median OS was 58.9 months in the continuous Rd group, 56.7 months in the Rd18 group, and 48.5 months in the MPT group. At 4 years, the OS rate was 60%, 57%, and 51%, respectively.
The HR for continuous Rd vs MPT was 0.75. The HR for continuous Rd vs Rd18 was 0.91. And the HR for Rd18 vs MPT was 0.83.
Safety
The most frequent grade 3/4 hematologic treatment-emergent adverse events were neutropenia and anemia. The rate of grade 3/4 neutropenia was higher in the MPT group than the continuous Rd or Rd18 groups.
Infections were the most common grade 3/4 non-hematologic treatment-emergent adverse events. The rate of grade 3/4 infections was higher in the Rd groups than the MPT group.
Grade 3/4 peripheral sensory neuropathy was less frequent in the continuous Rd and Rd18 groups than the MPT group.
The incidence of invasive second primary malignancy was 4% in the continuous Rd group, 6% in the Rd18 group, and 6% in the MPT group. ![]()

Photo courtesy of Celgene
Updated trial results support continuous treatment with lenalidomide and low-dose dexamethasone (Rd) as a standard of care for patients of all ages who have newly diagnosed multiple myeloma (MM) and are ineligible for stem cell transplant, according to researchers.
In this phase 3 trial, patients who received continuous Rd (until disease progression) had better progression-free survival (PFS) and overall survival (OS) than patients who received 18 cycles of Rd (Rd18) or a combination of melphalan, prednisone, and thalidomide (MPT).
Updated results from this study, known as the FIRST trial, were published in the Journal of Clinical Oncology.
Results were previously published in NEJM in 2014. The study was supported by Intergroupe Francophone du Myélome and Celgene Corporation, the makers of lenalidomide.
Thierry Facon, MD, of Centre Hospitalier Regional Universitaire de Lille in France, and his colleagues enrolled 1623 patients on this trial. They were newly diagnosed with MM and not eligible for stem cell transplant.
Patients were randomized to receive Rd in 28-day cycles until disease progression (n=535), Rd18 for 72 weeks (n=541), or MPT for 72 weeks (n=547).
Response
In the intent-to-treat population, the overall response rate was 81% for the continuous Rd group, 79% for the Rd18 group, and 67% in the MPT group. The complete response rates were 21%, 20%, and 12%, respectively.
The median duration of response was 32 months (range, 26-37) in the continuous Rd group, 22 months (range, 19-23) in the Rd18 group, and 22 months (range, 20-25) in the MPT group.
PFS and OS
The median PFS was 26.0 months in the continuous Rd group, 21.0 months in the Rd18 group, and 21.9 months in the MPT group. At 4 years, the PFS rate was 33%, 14%, and 13%, respectively.
The hazard ratio (HR) for continuous Rd vs MPT was 0.69. The HR for continuous Rd vs Rd18 was 0.71. And the HR for Rd18 vs MPT was 0.99.
The median OS was 58.9 months in the continuous Rd group, 56.7 months in the Rd18 group, and 48.5 months in the MPT group. At 4 years, the OS rate was 60%, 57%, and 51%, respectively.
The HR for continuous Rd vs MPT was 0.75. The HR for continuous Rd vs Rd18 was 0.91. And the HR for Rd18 vs MPT was 0.83.
Safety
The most frequent grade 3/4 hematologic treatment-emergent adverse events were neutropenia and anemia. The rate of grade 3/4 neutropenia was higher in the MPT group than the continuous Rd or Rd18 groups.
Infections were the most common grade 3/4 non-hematologic treatment-emergent adverse events. The rate of grade 3/4 infections was higher in the Rd groups than the MPT group.
Grade 3/4 peripheral sensory neuropathy was less frequent in the continuous Rd and Rd18 groups than the MPT group.
The incidence of invasive second primary malignancy was 4% in the continuous Rd group, 6% in the Rd18 group, and 6% in the MPT group. ![]()
Results support continued study of CMV vaccine
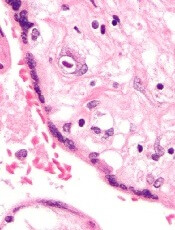
CMV infection
A vaccine designed to control cytomegalovirus (CMV) has produced favorable results in a phase 1 trial of healthy volunteers.
Investigators said the vaccine, known as Triplex, was well-tolerated at multiple dose levels.
The vaccine also generated “robust” and “durable” virus-specific immunity in subjects who were previously infected with CMV and those who were not.
The results of this study were published in Blood.
Triplex is a universal (non-HLA-restricted), recombinant modified vaccinia ankara viral vector vaccine engineered to induce a virus-specific T-cell response to 3 immuno-dominant proteins (UL83 [pp65], UL123 [IE1], and UL122 [IE2]) linked to CMV complications in the post-transplant setting.
Helocyte Inc. is developing the vaccine for control of CMV in recipients of allogeneic hematopoietic stem cell transplant (HSCT) and solid organ transplant.
This study was not funded by Helocyte. However, investigator Don J. Diamond, PhD, of City of Hope Comprehensive Cancer Center in Duarte, California is chair of Helocyte’s scientific advisory board and receives personal service fees from the company.
In this trial, Dr Diamond and his colleagues studied the response to Triplex in 24 healthy volunteers.
Subjects were divided into 3 groups of 8 receiving 3 different doses of the vaccine. The first dose level was 10xE7 plaque-forming units (pfu), the second was 5x10E7 pfu, and the third was 5x10E8 pfu.
The subjects received the vaccine in a volume of 1 mL by intramuscular injection in the upper arm and an identical booster injection 28 days later.
Safety
The investigators said Triplex was well tolerated in most subjects at all dose levels. There were no dose-limiting toxicities and no serious adverse events attributed to the vaccine.
One subject experienced a grade 3 injection site adverse event (erythema), which resolved in 1 day without treatment. In addition, there were 3 mild-to-moderate cutaneous reactions.
The investigators said the most common systemic reaction was mild-to-moderate flu-like symptoms. Most subjects in the highest dose group experienced these symptoms, as did a few subjects from the lower dose groups. All of these events were transient, self-limiting, and resolved.
Immunogenicity
The investigators reported “robust, functional, and durable” expansion of CMV-specific T cells after Triplex vaccination, in subjects with and without prior CMV infection.
At day 42, subjects had experienced a significant increase in pp65-specific T cells from baseline. The P values were 0.0003 for pp65-specific CD137+ CD8+ T cells and 0.001 for CD137+ CD4+ T cells.
Expansion remained above baseline levels until at least day 360 for pp65-specific CD137+ CD8+ T cells and at least until day 482 for pp65-specific CD137+ CD4+ T cells.
IE1-exon4- and IE2-exon5-specific T-cell expansions occurred as well, although the increase from baseline was not significant for IE1-exon4-specific T cells.
The median concentrations of IE2-exon5-specific T cells had increased significantly from baseline at day 42—for both CD137+ CD8+ T cells (P=0.014) and CD137+ CD4+ T cells (P=0.003).
The investigators noted that there was no significant difference in the responses to all 3 CMV libraries according to Triplex dose level, previous smallpox vaccination, or CMV-serostatus.
Next, the team found that Triplex vaccination induced significant expansion of pp65-specific IFN-γ+ CD8+ T cells. They said this suggests the vaccine was able to expand a functional subset of CMV-specific T cells, even in the absence of CMV viremia.
The median concentration of pp65-specific IFN-γ + CD8+ T cells increased significantly from baseline to day 28 (P=0.024), day 56 (P=0.003), day 100 (P=0.011), and day 360 (P=0.085).
There was no significant increase for pp65-specific IFN-γ + CD4+ T cells, IE1-exon4-specific IFN-γ+ T cells, or IE2-exon5-specific IFN-γ + T cells.
The investigators also said the Triplex vaccine induced significant vaccinia-specific T-cell increases by day 42 (P=0.0005), with an estimated decline to pre-vaccination levels at day 274.
These data supported the initiation of an ongoing phase 2 trial in which investigators are evaluating Triplex in patients undergoing allogeneic HSCT (NCT02506933).
“After years of work, it is very gratifying that we are making advancements in helping people worldwide achieve better health outcomes after a transplant procedure,” said Dr Diamond, who led the team that developed Triplex.
“Furthermore, Triplex’s favorable safety and immunogenicity may make the vaccine an ideal therapeutic platform to combat significant complications in many disease areas, like solid organ transplant and glioblastoma.” ![]()

CMV infection
A vaccine designed to control cytomegalovirus (CMV) has produced favorable results in a phase 1 trial of healthy volunteers.
Investigators said the vaccine, known as Triplex, was well-tolerated at multiple dose levels.
The vaccine also generated “robust” and “durable” virus-specific immunity in subjects who were previously infected with CMV and those who were not.
The results of this study were published in Blood.
Triplex is a universal (non-HLA-restricted), recombinant modified vaccinia ankara viral vector vaccine engineered to induce a virus-specific T-cell response to 3 immuno-dominant proteins (UL83 [pp65], UL123 [IE1], and UL122 [IE2]) linked to CMV complications in the post-transplant setting.
Helocyte Inc. is developing the vaccine for control of CMV in recipients of allogeneic hematopoietic stem cell transplant (HSCT) and solid organ transplant.
This study was not funded by Helocyte. However, investigator Don J. Diamond, PhD, of City of Hope Comprehensive Cancer Center in Duarte, California is chair of Helocyte’s scientific advisory board and receives personal service fees from the company.
In this trial, Dr Diamond and his colleagues studied the response to Triplex in 24 healthy volunteers.
Subjects were divided into 3 groups of 8 receiving 3 different doses of the vaccine. The first dose level was 10xE7 plaque-forming units (pfu), the second was 5x10E7 pfu, and the third was 5x10E8 pfu.
The subjects received the vaccine in a volume of 1 mL by intramuscular injection in the upper arm and an identical booster injection 28 days later.
Safety
The investigators said Triplex was well tolerated in most subjects at all dose levels. There were no dose-limiting toxicities and no serious adverse events attributed to the vaccine.
One subject experienced a grade 3 injection site adverse event (erythema), which resolved in 1 day without treatment. In addition, there were 3 mild-to-moderate cutaneous reactions.
The investigators said the most common systemic reaction was mild-to-moderate flu-like symptoms. Most subjects in the highest dose group experienced these symptoms, as did a few subjects from the lower dose groups. All of these events were transient, self-limiting, and resolved.
Immunogenicity
The investigators reported “robust, functional, and durable” expansion of CMV-specific T cells after Triplex vaccination, in subjects with and without prior CMV infection.
At day 42, subjects had experienced a significant increase in pp65-specific T cells from baseline. The P values were 0.0003 for pp65-specific CD137+ CD8+ T cells and 0.001 for CD137+ CD4+ T cells.
Expansion remained above baseline levels until at least day 360 for pp65-specific CD137+ CD8+ T cells and at least until day 482 for pp65-specific CD137+ CD4+ T cells.
IE1-exon4- and IE2-exon5-specific T-cell expansions occurred as well, although the increase from baseline was not significant for IE1-exon4-specific T cells.
The median concentrations of IE2-exon5-specific T cells had increased significantly from baseline at day 42—for both CD137+ CD8+ T cells (P=0.014) and CD137+ CD4+ T cells (P=0.003).
The investigators noted that there was no significant difference in the responses to all 3 CMV libraries according to Triplex dose level, previous smallpox vaccination, or CMV-serostatus.
Next, the team found that Triplex vaccination induced significant expansion of pp65-specific IFN-γ+ CD8+ T cells. They said this suggests the vaccine was able to expand a functional subset of CMV-specific T cells, even in the absence of CMV viremia.
The median concentration of pp65-specific IFN-γ + CD8+ T cells increased significantly from baseline to day 28 (P=0.024), day 56 (P=0.003), day 100 (P=0.011), and day 360 (P=0.085).
There was no significant increase for pp65-specific IFN-γ + CD4+ T cells, IE1-exon4-specific IFN-γ+ T cells, or IE2-exon5-specific IFN-γ + T cells.
The investigators also said the Triplex vaccine induced significant vaccinia-specific T-cell increases by day 42 (P=0.0005), with an estimated decline to pre-vaccination levels at day 274.
These data supported the initiation of an ongoing phase 2 trial in which investigators are evaluating Triplex in patients undergoing allogeneic HSCT (NCT02506933).
“After years of work, it is very gratifying that we are making advancements in helping people worldwide achieve better health outcomes after a transplant procedure,” said Dr Diamond, who led the team that developed Triplex.
“Furthermore, Triplex’s favorable safety and immunogenicity may make the vaccine an ideal therapeutic platform to combat significant complications in many disease areas, like solid organ transplant and glioblastoma.” ![]()

CMV infection
A vaccine designed to control cytomegalovirus (CMV) has produced favorable results in a phase 1 trial of healthy volunteers.
Investigators said the vaccine, known as Triplex, was well-tolerated at multiple dose levels.
The vaccine also generated “robust” and “durable” virus-specific immunity in subjects who were previously infected with CMV and those who were not.
The results of this study were published in Blood.
Triplex is a universal (non-HLA-restricted), recombinant modified vaccinia ankara viral vector vaccine engineered to induce a virus-specific T-cell response to 3 immuno-dominant proteins (UL83 [pp65], UL123 [IE1], and UL122 [IE2]) linked to CMV complications in the post-transplant setting.
Helocyte Inc. is developing the vaccine for control of CMV in recipients of allogeneic hematopoietic stem cell transplant (HSCT) and solid organ transplant.
This study was not funded by Helocyte. However, investigator Don J. Diamond, PhD, of City of Hope Comprehensive Cancer Center in Duarte, California is chair of Helocyte’s scientific advisory board and receives personal service fees from the company.
In this trial, Dr Diamond and his colleagues studied the response to Triplex in 24 healthy volunteers.
Subjects were divided into 3 groups of 8 receiving 3 different doses of the vaccine. The first dose level was 10xE7 plaque-forming units (pfu), the second was 5x10E7 pfu, and the third was 5x10E8 pfu.
The subjects received the vaccine in a volume of 1 mL by intramuscular injection in the upper arm and an identical booster injection 28 days later.
Safety
The investigators said Triplex was well tolerated in most subjects at all dose levels. There were no dose-limiting toxicities and no serious adverse events attributed to the vaccine.
One subject experienced a grade 3 injection site adverse event (erythema), which resolved in 1 day without treatment. In addition, there were 3 mild-to-moderate cutaneous reactions.
The investigators said the most common systemic reaction was mild-to-moderate flu-like symptoms. Most subjects in the highest dose group experienced these symptoms, as did a few subjects from the lower dose groups. All of these events were transient, self-limiting, and resolved.
Immunogenicity
The investigators reported “robust, functional, and durable” expansion of CMV-specific T cells after Triplex vaccination, in subjects with and without prior CMV infection.
At day 42, subjects had experienced a significant increase in pp65-specific T cells from baseline. The P values were 0.0003 for pp65-specific CD137+ CD8+ T cells and 0.001 for CD137+ CD4+ T cells.
Expansion remained above baseline levels until at least day 360 for pp65-specific CD137+ CD8+ T cells and at least until day 482 for pp65-specific CD137+ CD4+ T cells.
IE1-exon4- and IE2-exon5-specific T-cell expansions occurred as well, although the increase from baseline was not significant for IE1-exon4-specific T cells.
The median concentrations of IE2-exon5-specific T cells had increased significantly from baseline at day 42—for both CD137+ CD8+ T cells (P=0.014) and CD137+ CD4+ T cells (P=0.003).
The investigators noted that there was no significant difference in the responses to all 3 CMV libraries according to Triplex dose level, previous smallpox vaccination, or CMV-serostatus.
Next, the team found that Triplex vaccination induced significant expansion of pp65-specific IFN-γ+ CD8+ T cells. They said this suggests the vaccine was able to expand a functional subset of CMV-specific T cells, even in the absence of CMV viremia.
The median concentration of pp65-specific IFN-γ + CD8+ T cells increased significantly from baseline to day 28 (P=0.024), day 56 (P=0.003), day 100 (P=0.011), and day 360 (P=0.085).
There was no significant increase for pp65-specific IFN-γ + CD4+ T cells, IE1-exon4-specific IFN-γ+ T cells, or IE2-exon5-specific IFN-γ + T cells.
The investigators also said the Triplex vaccine induced significant vaccinia-specific T-cell increases by day 42 (P=0.0005), with an estimated decline to pre-vaccination levels at day 274.
These data supported the initiation of an ongoing phase 2 trial in which investigators are evaluating Triplex in patients undergoing allogeneic HSCT (NCT02506933).
“After years of work, it is very gratifying that we are making advancements in helping people worldwide achieve better health outcomes after a transplant procedure,” said Dr Diamond, who led the team that developed Triplex.
“Furthermore, Triplex’s favorable safety and immunogenicity may make the vaccine an ideal therapeutic platform to combat significant complications in many disease areas, like solid organ transplant and glioblastoma.” ![]()
Combo shows promise for treating T-ALL
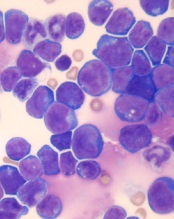
acute lymphoblastic leukemia
Image by Hind Medyou
Preclinical research suggests that 2 investigational drugs synergize to kill T-cell acute lymphoblastic leukemia (T-ALL) cells while having a minimal impact on normal blood cells.
Both drugs—the CK2 inhibitor CX-4945 and the BET inhibitor JQ1—have already been tested as single agents in clinical trials of hematologic malignancies and solid tumors.
However, the effects of the drugs in combination were not known until now.
“Previous studies provided us a rationale to test the combination of CX-4945 and JQ1 on refractory/relapsed T-cell leukemia,” said Hui Feng, MD, PhD, of Boston University School of Medicine in Massachusetts.
“Our findings suggest that the combination treatment of CX-4945 and JQ1 could be an effective strategy to target refractory/relapsed T-cell leukemia.”
Dr Feng and her colleagues reported these findings in Haematologica.
The researchers noted that targeting MYC-mediated transcriptional programs using JQ1 produces anti-leukemic activity in vitro and in vivo. However, global repression of transcription is likely to cause toxicities.
Therefore, the team theorized that finding drugs that synergize with JQ1 might allow them to reduce the dose of JQ1 and therefore decrease the risk of toxicity while enhancing the efficacy of treatment.
For this, the researchers looked to CX-4945, which is currently being investigated in clinical trials of breast cancer and multiple myeloma.
The team said CX-4945 has been shown to significantly reduce the growth and survival of human T-ALL cells on its own.
In a series of experiments, Dr Feng and her colleagues were able to show that CX-4945 destabilizes NOTCH1 and synergizes with JQ1 to induce apoptosis in human T-ALL cells.
The researchers also assessed the effects of JQ1 and CX-4946, alone and in combination, on normal peripheral blood monocytes (PBMs).
PBMs proved less sensitive than ALL-SIL T-ALL cells to each drug alone and to the drugs in combination. In fact, the combination had an antagonistic effect in PBMs.
Dr Feng and her colleagues said this research suggests JQ1 and CX-4946 in combination may be a feasible treatment option for relapsed/refractory T-ALL and other cancers involving CK2 and NOTCH1/MYC. ![]()

acute lymphoblastic leukemia
Image by Hind Medyou
Preclinical research suggests that 2 investigational drugs synergize to kill T-cell acute lymphoblastic leukemia (T-ALL) cells while having a minimal impact on normal blood cells.
Both drugs—the CK2 inhibitor CX-4945 and the BET inhibitor JQ1—have already been tested as single agents in clinical trials of hematologic malignancies and solid tumors.
However, the effects of the drugs in combination were not known until now.
“Previous studies provided us a rationale to test the combination of CX-4945 and JQ1 on refractory/relapsed T-cell leukemia,” said Hui Feng, MD, PhD, of Boston University School of Medicine in Massachusetts.
“Our findings suggest that the combination treatment of CX-4945 and JQ1 could be an effective strategy to target refractory/relapsed T-cell leukemia.”
Dr Feng and her colleagues reported these findings in Haematologica.
The researchers noted that targeting MYC-mediated transcriptional programs using JQ1 produces anti-leukemic activity in vitro and in vivo. However, global repression of transcription is likely to cause toxicities.
Therefore, the team theorized that finding drugs that synergize with JQ1 might allow them to reduce the dose of JQ1 and therefore decrease the risk of toxicity while enhancing the efficacy of treatment.
For this, the researchers looked to CX-4945, which is currently being investigated in clinical trials of breast cancer and multiple myeloma.
The team said CX-4945 has been shown to significantly reduce the growth and survival of human T-ALL cells on its own.
In a series of experiments, Dr Feng and her colleagues were able to show that CX-4945 destabilizes NOTCH1 and synergizes with JQ1 to induce apoptosis in human T-ALL cells.
The researchers also assessed the effects of JQ1 and CX-4946, alone and in combination, on normal peripheral blood monocytes (PBMs).
PBMs proved less sensitive than ALL-SIL T-ALL cells to each drug alone and to the drugs in combination. In fact, the combination had an antagonistic effect in PBMs.
Dr Feng and her colleagues said this research suggests JQ1 and CX-4946 in combination may be a feasible treatment option for relapsed/refractory T-ALL and other cancers involving CK2 and NOTCH1/MYC. ![]()

acute lymphoblastic leukemia
Image by Hind Medyou
Preclinical research suggests that 2 investigational drugs synergize to kill T-cell acute lymphoblastic leukemia (T-ALL) cells while having a minimal impact on normal blood cells.
Both drugs—the CK2 inhibitor CX-4945 and the BET inhibitor JQ1—have already been tested as single agents in clinical trials of hematologic malignancies and solid tumors.
However, the effects of the drugs in combination were not known until now.
“Previous studies provided us a rationale to test the combination of CX-4945 and JQ1 on refractory/relapsed T-cell leukemia,” said Hui Feng, MD, PhD, of Boston University School of Medicine in Massachusetts.
“Our findings suggest that the combination treatment of CX-4945 and JQ1 could be an effective strategy to target refractory/relapsed T-cell leukemia.”
Dr Feng and her colleagues reported these findings in Haematologica.
The researchers noted that targeting MYC-mediated transcriptional programs using JQ1 produces anti-leukemic activity in vitro and in vivo. However, global repression of transcription is likely to cause toxicities.
Therefore, the team theorized that finding drugs that synergize with JQ1 might allow them to reduce the dose of JQ1 and therefore decrease the risk of toxicity while enhancing the efficacy of treatment.
For this, the researchers looked to CX-4945, which is currently being investigated in clinical trials of breast cancer and multiple myeloma.
The team said CX-4945 has been shown to significantly reduce the growth and survival of human T-ALL cells on its own.
In a series of experiments, Dr Feng and her colleagues were able to show that CX-4945 destabilizes NOTCH1 and synergizes with JQ1 to induce apoptosis in human T-ALL cells.
The researchers also assessed the effects of JQ1 and CX-4946, alone and in combination, on normal peripheral blood monocytes (PBMs).
PBMs proved less sensitive than ALL-SIL T-ALL cells to each drug alone and to the drugs in combination. In fact, the combination had an antagonistic effect in PBMs.
Dr Feng and her colleagues said this research suggests JQ1 and CX-4946 in combination may be a feasible treatment option for relapsed/refractory T-ALL and other cancers involving CK2 and NOTCH1/MYC. ![]()
Drug approved to treat hemophilia B in Switzerland

Photo courtesy of Biogen
The Swiss Agency for Therapeutic Products, Swissmedic, has approved eftrenonacog alfa (Alprolix®) for the treatment of hemophilia B.
Eftrenonacog alfa is a recombinant factor IX Fc fusion protein indicated for both on-demand treatment and prophylaxis in previously treated patients with hemophilia B.
For prophylaxis, 1 dose of eftrenonacog alfa can be given every 7 days or every 10 days, with the ability to adjust the dosing interval based on individual response.
Eftrenonacog alfa is the only recombinant factor IX Fc fusion protein therapy approved in Switzerland for the treatment of hemophilia B.
“The Swiss approval of Alprolix is an important milestone for the hemophilia community, offering the opportunity for people with hemophilia B to experience prolonged protection from bleeds,” said Krassimir Mitchev, MD, PhD, vice president and medical therapeutic area head of hemophilia at Sobi, the company developing eftrenonacog alfa in collaboration with Biogen.
“We will now focus on ensuring timely and sustainable access to Alprolix in Switzerland.”
About eftrenonacog alfa
Eftrenonacog alfa is a recombinant clotting factor therapy developed by fusing factor IX to the Fc portion of immunoglobulin G subclass 1 (IgG1). This enables eftrenonacog alfa to use a naturally occurring pathway to prolong the time the therapy remains in the body.
Eftrenonacog alfa is currently approved for the treatment of hemophilia B in the European Economic Area, the US, Canada, Japan, Australia, New Zealand, and other countries.
Sobi and Biogen collaborate on the development and commercialization of eftrenonacog alfa.
The product has been evaluated in two phase 3 trials of patients with hemophilia B: the B-LONG study and the Kids B-LONG study.
B-LONG study
The B-LONG study included 123 male subjects with severe hemophilia B who were 12 years of age or older. They had no current or previous factor IX inhibitors and a history of 100 or more documented prior exposure days to factor IX products.
Patients received eftrenonacog alfa in 1 of 4 treatment arms:
- Weekly prophylaxis starting at 50 IU/kg, with pharmacokinetic (PK)-driven dose adjustments (n=63)
- Individualized interval prophylaxis starting at 100 IU/kg every 10 days, with PK-driven interval adjustments (n=29)
- On-demand treatment at 20 IU/kg to 100 IU/kg (n=27)
- Perioperative management (n=12, including 8 from arms 1-3).
Researchers assessed control of bleeding in all patients who experienced
a bleeding episode while on study. In total, 90.4% of bleeding episodes
were controlled by a single injection of eftrenonacog alfa.
The overall median annualized bleeding rates (ABRs)—including spontaneous and traumatic bleeds—were 2.95 in the weekly prophylaxis arm, 1.38 in the individualized interval prophylaxis arm, and 17.69 in the episodic treatment arm.
The perioperative management arm consisted of 12 patients undergoing 14 major surgical procedures. The treating physicians rated the hemostatic efficacy of eftrenonacog alfa as “excellent” or “good” in all surgeries.
Eftrenonacog alfa was considered generally well-tolerated. None of the patients developed inhibitors, and none reported anaphylaxis.
The most common adverse events—with an incidence of 5% or greater—occurring outside of the perioperative management arm were nasopharyngitis, influenza, arthralgia, upper respiratory infection, hypertension, and headache.
One serious adverse event may have been drug-related. The patient experienced obstructive uropathy in the setting of hematuria. However, he continued to receive eftrenonacog alfa, and the event resolved with medical management.
Kids B-LONG
In Kids B-LONG, researchers tested eftrenonacog alfa in 30 previously treated children younger than 12 who had severe hemophilia B. Patients had at least 50 prior exposure days to factor IX therapies.
Children who received eftrenonacog alfa prophylactically had an overall median ABR of 1.97. The median ABR for spontaneous joint bleeds was 0.
Approximately 33% of patients did not experience any bleeding episodes. About 92% of bleeding episodes were controlled by 1 or 2 injections of eftrenonacog alfa.
None of the patients developed inhibitors. Researchers said there were no treatment-related serious adverse events and no cases of serious allergic reactions or vascular thrombotic events.
None of the patients discontinued the study due to an adverse event. One adverse event—decreased appetite occurring in 1 patient—was considered related to eftrenonacog alfa.
The pattern of treatment-emergent adverse events in this study was generally consistent with results seen in adolescents and adults in the B-LONG study. ![]()

Photo courtesy of Biogen
The Swiss Agency for Therapeutic Products, Swissmedic, has approved eftrenonacog alfa (Alprolix®) for the treatment of hemophilia B.
Eftrenonacog alfa is a recombinant factor IX Fc fusion protein indicated for both on-demand treatment and prophylaxis in previously treated patients with hemophilia B.
For prophylaxis, 1 dose of eftrenonacog alfa can be given every 7 days or every 10 days, with the ability to adjust the dosing interval based on individual response.
Eftrenonacog alfa is the only recombinant factor IX Fc fusion protein therapy approved in Switzerland for the treatment of hemophilia B.
“The Swiss approval of Alprolix is an important milestone for the hemophilia community, offering the opportunity for people with hemophilia B to experience prolonged protection from bleeds,” said Krassimir Mitchev, MD, PhD, vice president and medical therapeutic area head of hemophilia at Sobi, the company developing eftrenonacog alfa in collaboration with Biogen.
“We will now focus on ensuring timely and sustainable access to Alprolix in Switzerland.”
About eftrenonacog alfa
Eftrenonacog alfa is a recombinant clotting factor therapy developed by fusing factor IX to the Fc portion of immunoglobulin G subclass 1 (IgG1). This enables eftrenonacog alfa to use a naturally occurring pathway to prolong the time the therapy remains in the body.
Eftrenonacog alfa is currently approved for the treatment of hemophilia B in the European Economic Area, the US, Canada, Japan, Australia, New Zealand, and other countries.
Sobi and Biogen collaborate on the development and commercialization of eftrenonacog alfa.
The product has been evaluated in two phase 3 trials of patients with hemophilia B: the B-LONG study and the Kids B-LONG study.
B-LONG study
The B-LONG study included 123 male subjects with severe hemophilia B who were 12 years of age or older. They had no current or previous factor IX inhibitors and a history of 100 or more documented prior exposure days to factor IX products.
Patients received eftrenonacog alfa in 1 of 4 treatment arms:
- Weekly prophylaxis starting at 50 IU/kg, with pharmacokinetic (PK)-driven dose adjustments (n=63)
- Individualized interval prophylaxis starting at 100 IU/kg every 10 days, with PK-driven interval adjustments (n=29)
- On-demand treatment at 20 IU/kg to 100 IU/kg (n=27)
- Perioperative management (n=12, including 8 from arms 1-3).
Researchers assessed control of bleeding in all patients who experienced
a bleeding episode while on study. In total, 90.4% of bleeding episodes
were controlled by a single injection of eftrenonacog alfa.
The overall median annualized bleeding rates (ABRs)—including spontaneous and traumatic bleeds—were 2.95 in the weekly prophylaxis arm, 1.38 in the individualized interval prophylaxis arm, and 17.69 in the episodic treatment arm.
The perioperative management arm consisted of 12 patients undergoing 14 major surgical procedures. The treating physicians rated the hemostatic efficacy of eftrenonacog alfa as “excellent” or “good” in all surgeries.
Eftrenonacog alfa was considered generally well-tolerated. None of the patients developed inhibitors, and none reported anaphylaxis.
The most common adverse events—with an incidence of 5% or greater—occurring outside of the perioperative management arm were nasopharyngitis, influenza, arthralgia, upper respiratory infection, hypertension, and headache.
One serious adverse event may have been drug-related. The patient experienced obstructive uropathy in the setting of hematuria. However, he continued to receive eftrenonacog alfa, and the event resolved with medical management.
Kids B-LONG
In Kids B-LONG, researchers tested eftrenonacog alfa in 30 previously treated children younger than 12 who had severe hemophilia B. Patients had at least 50 prior exposure days to factor IX therapies.
Children who received eftrenonacog alfa prophylactically had an overall median ABR of 1.97. The median ABR for spontaneous joint bleeds was 0.
Approximately 33% of patients did not experience any bleeding episodes. About 92% of bleeding episodes were controlled by 1 or 2 injections of eftrenonacog alfa.
None of the patients developed inhibitors. Researchers said there were no treatment-related serious adverse events and no cases of serious allergic reactions or vascular thrombotic events.
None of the patients discontinued the study due to an adverse event. One adverse event—decreased appetite occurring in 1 patient—was considered related to eftrenonacog alfa.
The pattern of treatment-emergent adverse events in this study was generally consistent with results seen in adolescents and adults in the B-LONG study. ![]()

Photo courtesy of Biogen
The Swiss Agency for Therapeutic Products, Swissmedic, has approved eftrenonacog alfa (Alprolix®) for the treatment of hemophilia B.
Eftrenonacog alfa is a recombinant factor IX Fc fusion protein indicated for both on-demand treatment and prophylaxis in previously treated patients with hemophilia B.
For prophylaxis, 1 dose of eftrenonacog alfa can be given every 7 days or every 10 days, with the ability to adjust the dosing interval based on individual response.
Eftrenonacog alfa is the only recombinant factor IX Fc fusion protein therapy approved in Switzerland for the treatment of hemophilia B.
“The Swiss approval of Alprolix is an important milestone for the hemophilia community, offering the opportunity for people with hemophilia B to experience prolonged protection from bleeds,” said Krassimir Mitchev, MD, PhD, vice president and medical therapeutic area head of hemophilia at Sobi, the company developing eftrenonacog alfa in collaboration with Biogen.
“We will now focus on ensuring timely and sustainable access to Alprolix in Switzerland.”
About eftrenonacog alfa
Eftrenonacog alfa is a recombinant clotting factor therapy developed by fusing factor IX to the Fc portion of immunoglobulin G subclass 1 (IgG1). This enables eftrenonacog alfa to use a naturally occurring pathway to prolong the time the therapy remains in the body.
Eftrenonacog alfa is currently approved for the treatment of hemophilia B in the European Economic Area, the US, Canada, Japan, Australia, New Zealand, and other countries.
Sobi and Biogen collaborate on the development and commercialization of eftrenonacog alfa.
The product has been evaluated in two phase 3 trials of patients with hemophilia B: the B-LONG study and the Kids B-LONG study.
B-LONG study
The B-LONG study included 123 male subjects with severe hemophilia B who were 12 years of age or older. They had no current or previous factor IX inhibitors and a history of 100 or more documented prior exposure days to factor IX products.
Patients received eftrenonacog alfa in 1 of 4 treatment arms:
- Weekly prophylaxis starting at 50 IU/kg, with pharmacokinetic (PK)-driven dose adjustments (n=63)
- Individualized interval prophylaxis starting at 100 IU/kg every 10 days, with PK-driven interval adjustments (n=29)
- On-demand treatment at 20 IU/kg to 100 IU/kg (n=27)
- Perioperative management (n=12, including 8 from arms 1-3).
Researchers assessed control of bleeding in all patients who experienced
a bleeding episode while on study. In total, 90.4% of bleeding episodes
were controlled by a single injection of eftrenonacog alfa.
The overall median annualized bleeding rates (ABRs)—including spontaneous and traumatic bleeds—were 2.95 in the weekly prophylaxis arm, 1.38 in the individualized interval prophylaxis arm, and 17.69 in the episodic treatment arm.
The perioperative management arm consisted of 12 patients undergoing 14 major surgical procedures. The treating physicians rated the hemostatic efficacy of eftrenonacog alfa as “excellent” or “good” in all surgeries.
Eftrenonacog alfa was considered generally well-tolerated. None of the patients developed inhibitors, and none reported anaphylaxis.
The most common adverse events—with an incidence of 5% or greater—occurring outside of the perioperative management arm were nasopharyngitis, influenza, arthralgia, upper respiratory infection, hypertension, and headache.
One serious adverse event may have been drug-related. The patient experienced obstructive uropathy in the setting of hematuria. However, he continued to receive eftrenonacog alfa, and the event resolved with medical management.
Kids B-LONG
In Kids B-LONG, researchers tested eftrenonacog alfa in 30 previously treated children younger than 12 who had severe hemophilia B. Patients had at least 50 prior exposure days to factor IX therapies.
Children who received eftrenonacog alfa prophylactically had an overall median ABR of 1.97. The median ABR for spontaneous joint bleeds was 0.
Approximately 33% of patients did not experience any bleeding episodes. About 92% of bleeding episodes were controlled by 1 or 2 injections of eftrenonacog alfa.
None of the patients developed inhibitors. Researchers said there were no treatment-related serious adverse events and no cases of serious allergic reactions or vascular thrombotic events.
None of the patients discontinued the study due to an adverse event. One adverse event—decreased appetite occurring in 1 patient—was considered related to eftrenonacog alfa.
The pattern of treatment-emergent adverse events in this study was generally consistent with results seen in adolescents and adults in the B-LONG study.
Treatment facility volume linked to survival in MM

Photo courtesy of the CDC
Patients with multiple myeloma (MM) are more likely to live longer if they are treated at a medical center where the staff has more experience with the disease, according to research published in the Journal of Clinical Oncology.
The study showed that patients treated at medical centers seeing 10 new MM patients per year had a 20% higher risk of death than patients treated at centers seeing 40 new MM patients per year.
Most cancer treatment centers in the US see fewer than 10 new MM patients per year.
“It is very difficult to be proficient when doctors are seeing only 1 or 2 new cases of multiple myeloma per year,” said study author Ronald Go, MD, of the Mayo Clinic in Rochester, Minnesota.
“Studies on cancer surgery have shown the more experience the center or practitioner has, the better the outcome. We wanted to see if volume matters when it comes to nonsurgical treatment of rare cancers such as multiple myeloma.”
To investigate, Dr Go and his colleagues used the National Cancer Database, examining outcomes for 94,722 newly diagnosed MM patients treated at 1333 facilities between 2003 and 2011.
The researchers grouped the facilities into quartiles according to the volume of MM patients treated there each year.
The mean number of MM patients treated per year was:
- Less than 3.6 for quartile 1 (Q1)
- 3.6 to 6.1 for Q2
- 6.1 to 10.3 for Q3
- More than 10.3 for Q4.
The majority of patients (60.3%) were treated in Q4 facilities. For all facilities, the median number of new MM patients per year was 6.1 (range, 3.6 to 10.3). The mean was 8.8 ± 9.9.
The researchers calculated the relationship between MM patient volume at these facilities and patient mortality, adjusting for demographic characteristics, socioeconomic factors, geographic factors, comorbidities, and year of diagnosis.
Outcomes
The unadjusted median overall survival was 26.9 months for patients treated at Q1 facilities, 29.1 months for Q2, 31.9 months for Q3, and 49.1 months for Q4 (P<0.001).
The 1-year mortality rate was 33.5% for patients treated at Q1 facilities, 32.3% for Q2, 30.7% for Q3, and 21.9% for Q4.
The researchers’ multivariable analysis showed that facility volume was independently associated with all-cause mortality.
Patients treated at the lower-quartile facilities had a higher risk of death than patients treated at Q4 facilities. The hazard ratios were 1.12 for patients at Q3 facilities, 1.12 for Q2, and 1.22 for Q1.
The researchers performed another analysis in which volume was treated as a continuous variable, and they compared various volume sizes to a reference volume of 10 patients per year.
Compared with facilities treating 10 new MM patients per year, facilities treating 20 MM patients per year had roughly 10% lower overall mortality rates.
Facilities treating 30 MM patients per year had about 15% lower mortality rates. And facilities treating 40 MM patients per year had 20% lower overall mortality rates.

Photo courtesy of the CDC
Patients with multiple myeloma (MM) are more likely to live longer if they are treated at a medical center where the staff has more experience with the disease, according to research published in the Journal of Clinical Oncology.
The study showed that patients treated at medical centers seeing 10 new MM patients per year had a 20% higher risk of death than patients treated at centers seeing 40 new MM patients per year.
Most cancer treatment centers in the US see fewer than 10 new MM patients per year.
“It is very difficult to be proficient when doctors are seeing only 1 or 2 new cases of multiple myeloma per year,” said study author Ronald Go, MD, of the Mayo Clinic in Rochester, Minnesota.
“Studies on cancer surgery have shown the more experience the center or practitioner has, the better the outcome. We wanted to see if volume matters when it comes to nonsurgical treatment of rare cancers such as multiple myeloma.”
To investigate, Dr Go and his colleagues used the National Cancer Database, examining outcomes for 94,722 newly diagnosed MM patients treated at 1333 facilities between 2003 and 2011.
The researchers grouped the facilities into quartiles according to the volume of MM patients treated there each year.
The mean number of MM patients treated per year was:
- Less than 3.6 for quartile 1 (Q1)
- 3.6 to 6.1 for Q2
- 6.1 to 10.3 for Q3
- More than 10.3 for Q4.
The majority of patients (60.3%) were treated in Q4 facilities. For all facilities, the median number of new MM patients per year was 6.1 (range, 3.6 to 10.3). The mean was 8.8 ± 9.9.
The researchers calculated the relationship between MM patient volume at these facilities and patient mortality, adjusting for demographic characteristics, socioeconomic factors, geographic factors, comorbidities, and year of diagnosis.
Outcomes
The unadjusted median overall survival was 26.9 months for patients treated at Q1 facilities, 29.1 months for Q2, 31.9 months for Q3, and 49.1 months for Q4 (P<0.001).
The 1-year mortality rate was 33.5% for patients treated at Q1 facilities, 32.3% for Q2, 30.7% for Q3, and 21.9% for Q4.
The researchers’ multivariable analysis showed that facility volume was independently associated with all-cause mortality.
Patients treated at the lower-quartile facilities had a higher risk of death than patients treated at Q4 facilities. The hazard ratios were 1.12 for patients at Q3 facilities, 1.12 for Q2, and 1.22 for Q1.
The researchers performed another analysis in which volume was treated as a continuous variable, and they compared various volume sizes to a reference volume of 10 patients per year.
Compared with facilities treating 10 new MM patients per year, facilities treating 20 MM patients per year had roughly 10% lower overall mortality rates.
Facilities treating 30 MM patients per year had about 15% lower mortality rates. And facilities treating 40 MM patients per year had 20% lower overall mortality rates.

Photo courtesy of the CDC
Patients with multiple myeloma (MM) are more likely to live longer if they are treated at a medical center where the staff has more experience with the disease, according to research published in the Journal of Clinical Oncology.
The study showed that patients treated at medical centers seeing 10 new MM patients per year had a 20% higher risk of death than patients treated at centers seeing 40 new MM patients per year.
Most cancer treatment centers in the US see fewer than 10 new MM patients per year.
“It is very difficult to be proficient when doctors are seeing only 1 or 2 new cases of multiple myeloma per year,” said study author Ronald Go, MD, of the Mayo Clinic in Rochester, Minnesota.
“Studies on cancer surgery have shown the more experience the center or practitioner has, the better the outcome. We wanted to see if volume matters when it comes to nonsurgical treatment of rare cancers such as multiple myeloma.”
To investigate, Dr Go and his colleagues used the National Cancer Database, examining outcomes for 94,722 newly diagnosed MM patients treated at 1333 facilities between 2003 and 2011.
The researchers grouped the facilities into quartiles according to the volume of MM patients treated there each year.
The mean number of MM patients treated per year was:
- Less than 3.6 for quartile 1 (Q1)
- 3.6 to 6.1 for Q2
- 6.1 to 10.3 for Q3
- More than 10.3 for Q4.
The majority of patients (60.3%) were treated in Q4 facilities. For all facilities, the median number of new MM patients per year was 6.1 (range, 3.6 to 10.3). The mean was 8.8 ± 9.9.
The researchers calculated the relationship between MM patient volume at these facilities and patient mortality, adjusting for demographic characteristics, socioeconomic factors, geographic factors, comorbidities, and year of diagnosis.
Outcomes
The unadjusted median overall survival was 26.9 months for patients treated at Q1 facilities, 29.1 months for Q2, 31.9 months for Q3, and 49.1 months for Q4 (P<0.001).
The 1-year mortality rate was 33.5% for patients treated at Q1 facilities, 32.3% for Q2, 30.7% for Q3, and 21.9% for Q4.
The researchers’ multivariable analysis showed that facility volume was independently associated with all-cause mortality.
Patients treated at the lower-quartile facilities had a higher risk of death than patients treated at Q4 facilities. The hazard ratios were 1.12 for patients at Q3 facilities, 1.12 for Q2, and 1.22 for Q1.
The researchers performed another analysis in which volume was treated as a continuous variable, and they compared various volume sizes to a reference volume of 10 patients per year.
Compared with facilities treating 10 new MM patients per year, facilities treating 20 MM patients per year had roughly 10% lower overall mortality rates.
Facilities treating 30 MM patients per year had about 15% lower mortality rates. And facilities treating 40 MM patients per year had 20% lower overall mortality rates.
Agent could treat hemophilia A and B, team says

A new bypassing agent mimics the pro-clotting activity of factor V Leiden and might prove effective for treating hemophilia A and B, according to preclinical research published in Blood.
“We know that patients who have severe hemophilia and also have mutations that increase clotting, such as factor V Leiden, experience less severe bleeding,” said study author Trevor Baglin, MD, of Addenbrooke’s Hospital in Cambridge, UK.
In patients with factor V Leiden, defects in the anticoagulant activated protein C (APC) mechanism lead to an overactive production of thrombin.
The researchers set out to determine if they could exploit this phenomenon to treat hemophilia by developing a direct inhibitor of APC. They modified serine protease inhibitors, known as serpins, to make them specific and efficient inhibitors of APC.
“We hypothesized that if we targeted the protein C pathway we could prolong thrombin production,” said study author James Huntington, PhD, of the University of Cambridge in the UK.
“We engineered a serpin so that it could selectively prevent APC from shutting down thrombin production before the formation of a stable clot.”
The researchers administered the serpin to mice with hemophilia B and clipped their tails. In this model, the blood loss decreased as the dose increased, with the highest dose reducing bleeding to the level of healthy mice.
Further injury models underscored that the serpin helped the majority of mice form stable clots, with higher doses resulting in quicker clot formation.
The serpin was also able to accelerate clot formation when added to blood samples from patients with hemophilia A.
“It is our understanding that because we are targeting a general anticlotting process, our serpin could effectively treat patients with either hemophilia A or B, including those who develop inhibitors to more traditional therapy,” Dr Huntington said.
“Additionally, we have focused on engineering the serpin to be both subcutaneously delivered and long-acting. This will free patients from the cumbersome thrice-weekly infusions that are necessary under many contemporary therapy regimens.”
“Within 3 years, we hope to be conducting our first-in-man trials of a subcutaneously administered form of our serpin,” Dr Baglin added.
“It is important to remember that the majority of people in the world with hemophilia have no access to therapy. A stable, subcutaneous, long-acting, effective hemostatic agent could bring treatment to a great deal many more hemophilia sufferers.”
This study forms part of a patent application by the authors, and the serpin is being developed into a therapeutic by a start-up company known as ApcinteX, with funding from Medicxi.

A new bypassing agent mimics the pro-clotting activity of factor V Leiden and might prove effective for treating hemophilia A and B, according to preclinical research published in Blood.
“We know that patients who have severe hemophilia and also have mutations that increase clotting, such as factor V Leiden, experience less severe bleeding,” said study author Trevor Baglin, MD, of Addenbrooke’s Hospital in Cambridge, UK.
In patients with factor V Leiden, defects in the anticoagulant activated protein C (APC) mechanism lead to an overactive production of thrombin.
The researchers set out to determine if they could exploit this phenomenon to treat hemophilia by developing a direct inhibitor of APC. They modified serine protease inhibitors, known as serpins, to make them specific and efficient inhibitors of APC.
“We hypothesized that if we targeted the protein C pathway we could prolong thrombin production,” said study author James Huntington, PhD, of the University of Cambridge in the UK.
“We engineered a serpin so that it could selectively prevent APC from shutting down thrombin production before the formation of a stable clot.”
The researchers administered the serpin to mice with hemophilia B and clipped their tails. In this model, the blood loss decreased as the dose increased, with the highest dose reducing bleeding to the level of healthy mice.
Further injury models underscored that the serpin helped the majority of mice form stable clots, with higher doses resulting in quicker clot formation.
The serpin was also able to accelerate clot formation when added to blood samples from patients with hemophilia A.
“It is our understanding that because we are targeting a general anticlotting process, our serpin could effectively treat patients with either hemophilia A or B, including those who develop inhibitors to more traditional therapy,” Dr Huntington said.
“Additionally, we have focused on engineering the serpin to be both subcutaneously delivered and long-acting. This will free patients from the cumbersome thrice-weekly infusions that are necessary under many contemporary therapy regimens.”
“Within 3 years, we hope to be conducting our first-in-man trials of a subcutaneously administered form of our serpin,” Dr Baglin added.
“It is important to remember that the majority of people in the world with hemophilia have no access to therapy. A stable, subcutaneous, long-acting, effective hemostatic agent could bring treatment to a great deal many more hemophilia sufferers.”
This study forms part of a patent application by the authors, and the serpin is being developed into a therapeutic by a start-up company known as ApcinteX, with funding from Medicxi.

A new bypassing agent mimics the pro-clotting activity of factor V Leiden and might prove effective for treating hemophilia A and B, according to preclinical research published in Blood.
“We know that patients who have severe hemophilia and also have mutations that increase clotting, such as factor V Leiden, experience less severe bleeding,” said study author Trevor Baglin, MD, of Addenbrooke’s Hospital in Cambridge, UK.
In patients with factor V Leiden, defects in the anticoagulant activated protein C (APC) mechanism lead to an overactive production of thrombin.
The researchers set out to determine if they could exploit this phenomenon to treat hemophilia by developing a direct inhibitor of APC. They modified serine protease inhibitors, known as serpins, to make them specific and efficient inhibitors of APC.
“We hypothesized that if we targeted the protein C pathway we could prolong thrombin production,” said study author James Huntington, PhD, of the University of Cambridge in the UK.
“We engineered a serpin so that it could selectively prevent APC from shutting down thrombin production before the formation of a stable clot.”
The researchers administered the serpin to mice with hemophilia B and clipped their tails. In this model, the blood loss decreased as the dose increased, with the highest dose reducing bleeding to the level of healthy mice.
Further injury models underscored that the serpin helped the majority of mice form stable clots, with higher doses resulting in quicker clot formation.
The serpin was also able to accelerate clot formation when added to blood samples from patients with hemophilia A.
“It is our understanding that because we are targeting a general anticlotting process, our serpin could effectively treat patients with either hemophilia A or B, including those who develop inhibitors to more traditional therapy,” Dr Huntington said.
“Additionally, we have focused on engineering the serpin to be both subcutaneously delivered and long-acting. This will free patients from the cumbersome thrice-weekly infusions that are necessary under many contemporary therapy regimens.”
“Within 3 years, we hope to be conducting our first-in-man trials of a subcutaneously administered form of our serpin,” Dr Baglin added.
“It is important to remember that the majority of people in the world with hemophilia have no access to therapy. A stable, subcutaneous, long-acting, effective hemostatic agent could bring treatment to a great deal many more hemophilia sufferers.”
This study forms part of a patent application by the authors, and the serpin is being developed into a therapeutic by a start-up company known as ApcinteX, with funding from Medicxi.
NORD publishes physician guide to CTCL

mycosis fungoides
The National Organization for Rare Disorders (NORD) has published a guide for physicians treating patients with cutaneous T-cell lymphoma (CTCL).
The guide contains information about disease classification, signs and symptoms of CTCL, methods of diagnosing the disease, standard therapies, and investigational therapies for CTCL.
The guide also includes a list of resources for physicians and patients.
“The NORD Physician Guide to Cutaneous T-Cell Lymphoma (CTCL)” is available for free on the NORD Physician Guides website.
The guide was made possible by an educational grant from Therakos, now a part of Mallinckrodt Pharmaceuticals.
The guide was developed in collaboration with Oleg E. Akilov, MD, PhD, of the University of Pittsburgh School of Medicine in Pennsylvania.
“Eczema and even some cases of psoriasis may look very similar to mycosis fungoides, the most common type of cutaneous T-cell lymphomas,” Dr Akilov noted.
“It is important to be aware of these similarities and to be ready to think about cutaneous lymphoma when a patient with ‘common dermatosis’ does not respond to regular treatments.”
About NORD guides
NORD established its physician guide series as part of a broader strategic initiative to promote earlier diagnosis and state-of-the-art care for people with rare diseases. Each online guide is written or reviewed by a medical professional with expertise on the topic.
Other recent guides in the series include:
- The NORD Physician Guide to Mitochondrial Myopathies
- The NORD Physician Guide to Paroxysmal Nocturnal Hemoglobinuria (PNH)
- The NORD Physician Guide to Atypical Hemolytic Uremic Syndrome (aHUS)
- The NORD Physician Guide to Nontuberculous Mycobacterial Lung Disease.
“People who have rare diseases often go for many years without a diagnosis,” said Marsha Lanes, a genetic counselor in NORD’s Educational Initiatives Department.
“The purpose of NORD’s free online physician guides is to reduce the time to diagnosis and encourage optimal treatment for patients with little-known and little-understood rare diseases.”

mycosis fungoides
The National Organization for Rare Disorders (NORD) has published a guide for physicians treating patients with cutaneous T-cell lymphoma (CTCL).
The guide contains information about disease classification, signs and symptoms of CTCL, methods of diagnosing the disease, standard therapies, and investigational therapies for CTCL.
The guide also includes a list of resources for physicians and patients.
“The NORD Physician Guide to Cutaneous T-Cell Lymphoma (CTCL)” is available for free on the NORD Physician Guides website.
The guide was made possible by an educational grant from Therakos, now a part of Mallinckrodt Pharmaceuticals.
The guide was developed in collaboration with Oleg E. Akilov, MD, PhD, of the University of Pittsburgh School of Medicine in Pennsylvania.
“Eczema and even some cases of psoriasis may look very similar to mycosis fungoides, the most common type of cutaneous T-cell lymphomas,” Dr Akilov noted.
“It is important to be aware of these similarities and to be ready to think about cutaneous lymphoma when a patient with ‘common dermatosis’ does not respond to regular treatments.”
About NORD guides
NORD established its physician guide series as part of a broader strategic initiative to promote earlier diagnosis and state-of-the-art care for people with rare diseases. Each online guide is written or reviewed by a medical professional with expertise on the topic.
Other recent guides in the series include:
- The NORD Physician Guide to Mitochondrial Myopathies
- The NORD Physician Guide to Paroxysmal Nocturnal Hemoglobinuria (PNH)
- The NORD Physician Guide to Atypical Hemolytic Uremic Syndrome (aHUS)
- The NORD Physician Guide to Nontuberculous Mycobacterial Lung Disease.
“People who have rare diseases often go for many years without a diagnosis,” said Marsha Lanes, a genetic counselor in NORD’s Educational Initiatives Department.
“The purpose of NORD’s free online physician guides is to reduce the time to diagnosis and encourage optimal treatment for patients with little-known and little-understood rare diseases.”

mycosis fungoides
The National Organization for Rare Disorders (NORD) has published a guide for physicians treating patients with cutaneous T-cell lymphoma (CTCL).
The guide contains information about disease classification, signs and symptoms of CTCL, methods of diagnosing the disease, standard therapies, and investigational therapies for CTCL.
The guide also includes a list of resources for physicians and patients.
“The NORD Physician Guide to Cutaneous T-Cell Lymphoma (CTCL)” is available for free on the NORD Physician Guides website.
The guide was made possible by an educational grant from Therakos, now a part of Mallinckrodt Pharmaceuticals.
The guide was developed in collaboration with Oleg E. Akilov, MD, PhD, of the University of Pittsburgh School of Medicine in Pennsylvania.
“Eczema and even some cases of psoriasis may look very similar to mycosis fungoides, the most common type of cutaneous T-cell lymphomas,” Dr Akilov noted.
“It is important to be aware of these similarities and to be ready to think about cutaneous lymphoma when a patient with ‘common dermatosis’ does not respond to regular treatments.”
About NORD guides
NORD established its physician guide series as part of a broader strategic initiative to promote earlier diagnosis and state-of-the-art care for people with rare diseases. Each online guide is written or reviewed by a medical professional with expertise on the topic.
Other recent guides in the series include:
- The NORD Physician Guide to Mitochondrial Myopathies
- The NORD Physician Guide to Paroxysmal Nocturnal Hemoglobinuria (PNH)
- The NORD Physician Guide to Atypical Hemolytic Uremic Syndrome (aHUS)
- The NORD Physician Guide to Nontuberculous Mycobacterial Lung Disease.
“People who have rare diseases often go for many years without a diagnosis,” said Marsha Lanes, a genetic counselor in NORD’s Educational Initiatives Department.
“The purpose of NORD’s free online physician guides is to reduce the time to diagnosis and encourage optimal treatment for patients with little-known and little-understood rare diseases.”