User login
FDA advises testing all donated blood for Zika virus

The US Food and Drug Administration (FDA) has issued a revised guidance intended to reduce the risk of Zika virus transmission via blood.
The guidance recommends that all states and US territories screen donated whole blood and blood components for the Zika virus with a test authorized under an investigational new drug application or a licensed test when available.
For plasma and certain platelet products, an FDA-approved pathogen-reduction device can be used instead of a screening test.
“There is still much uncertainty regarding the nature and extent of Zika virus transmission,” said Peter Marks, MD, PhD, director of the FDA’s Center for Biologics Evaluation and Research.
“At this time, the recommendation for testing the entire blood supply will help ensure that safe blood is available for all individuals who might need transfusion.”
In February, the FDA issued a guidance recommending that areas with active Zika virus transmission screen donated whole blood and blood components for the Zika virus, use pathogen-reduction devices, or halt blood collection and obtain blood from areas of the US without active virus transmission.
The FDA decided to update its guidance after considering the available scientific evidence, consulting with other public health agencies, and taking into consideration the potential serious health consequences of Zika virus infection to pregnant women and children born to women exposed to Zika virus during pregnancy.
“As new scientific and epidemiological information regarding Zika virus has become available, it’s clear that additional precautionary measures are necessary,” said Luciana Borio, MD, the FDA’s acting chief scientist.
“We are issuing revised guidance for immediate implementation in order to help maintain the safety of the US blood supply.”
Testing of donated blood is already underway in Florida, Puerto Rico, and other areas, and it has proven effective in identifying donations infected with Zika virus.
The FDA said nationwide testing will be in effect until the risk of Zika virus transmission via transfusion is reduced. ![]()

The US Food and Drug Administration (FDA) has issued a revised guidance intended to reduce the risk of Zika virus transmission via blood.
The guidance recommends that all states and US territories screen donated whole blood and blood components for the Zika virus with a test authorized under an investigational new drug application or a licensed test when available.
For plasma and certain platelet products, an FDA-approved pathogen-reduction device can be used instead of a screening test.
“There is still much uncertainty regarding the nature and extent of Zika virus transmission,” said Peter Marks, MD, PhD, director of the FDA’s Center for Biologics Evaluation and Research.
“At this time, the recommendation for testing the entire blood supply will help ensure that safe blood is available for all individuals who might need transfusion.”
In February, the FDA issued a guidance recommending that areas with active Zika virus transmission screen donated whole blood and blood components for the Zika virus, use pathogen-reduction devices, or halt blood collection and obtain blood from areas of the US without active virus transmission.
The FDA decided to update its guidance after considering the available scientific evidence, consulting with other public health agencies, and taking into consideration the potential serious health consequences of Zika virus infection to pregnant women and children born to women exposed to Zika virus during pregnancy.
“As new scientific and epidemiological information regarding Zika virus has become available, it’s clear that additional precautionary measures are necessary,” said Luciana Borio, MD, the FDA’s acting chief scientist.
“We are issuing revised guidance for immediate implementation in order to help maintain the safety of the US blood supply.”
Testing of donated blood is already underway in Florida, Puerto Rico, and other areas, and it has proven effective in identifying donations infected with Zika virus.
The FDA said nationwide testing will be in effect until the risk of Zika virus transmission via transfusion is reduced. ![]()

The US Food and Drug Administration (FDA) has issued a revised guidance intended to reduce the risk of Zika virus transmission via blood.
The guidance recommends that all states and US territories screen donated whole blood and blood components for the Zika virus with a test authorized under an investigational new drug application or a licensed test when available.
For plasma and certain platelet products, an FDA-approved pathogen-reduction device can be used instead of a screening test.
“There is still much uncertainty regarding the nature and extent of Zika virus transmission,” said Peter Marks, MD, PhD, director of the FDA’s Center for Biologics Evaluation and Research.
“At this time, the recommendation for testing the entire blood supply will help ensure that safe blood is available for all individuals who might need transfusion.”
In February, the FDA issued a guidance recommending that areas with active Zika virus transmission screen donated whole blood and blood components for the Zika virus, use pathogen-reduction devices, or halt blood collection and obtain blood from areas of the US without active virus transmission.
The FDA decided to update its guidance after considering the available scientific evidence, consulting with other public health agencies, and taking into consideration the potential serious health consequences of Zika virus infection to pregnant women and children born to women exposed to Zika virus during pregnancy.
“As new scientific and epidemiological information regarding Zika virus has become available, it’s clear that additional precautionary measures are necessary,” said Luciana Borio, MD, the FDA’s acting chief scientist.
“We are issuing revised guidance for immediate implementation in order to help maintain the safety of the US blood supply.”
Testing of donated blood is already underway in Florida, Puerto Rico, and other areas, and it has proven effective in identifying donations infected with Zika virus.
The FDA said nationwide testing will be in effect until the risk of Zika virus transmission via transfusion is reduced. ![]()
Native Israelis have higher risk of Hodgkin lymphoma

Photo from Hebrew
University of Jerusalem
A study of 2.3 million Jewish subjects suggests that being born in Israel increases a person’s risk of developing Hodgkin lymphoma (HL).
Native Israelis had a higher risk of HL than subjects who immigrated to Israel from Europe, Asia, or North Africa.
Researchers said this finding, published in Leukemia & Lymphoma, suggests that exposure to as-yet-unidentified elements of Israel’s environment increases the risk of HL.
“While we still need further studies to identify the specific causes of the high rates of Hodgkin lymphoma among native Israelis, our findings direct us to search for possible environmental causes in Israel and the neighboring countries,” said study author Hagai Levine, MD, of Hebrew University of Jerusalem in Israel.
“These causes could be not only environmental exposures but also diet, climate, social environment, and stress that may be related to chronic regional conflict.”
Dr Levine and his colleagues noted that the incidence of HL in Israel is among the highest in the world. Based on GLOBOCAN estimates for 2012, Israeli females have the highest age-standardized incidence rate of HL worldwide, and Israeli males have the second highest. From 1960 through 2005, Israel experienced a sharp rise in HL incidence among Israeli-born individuals of both sexes.
Despite evidence for an environmental etiology, very few risk factors have been identified. Studying immigrant populations provides a means of investigating the relative importance of genetic factors as compared to environmental factors in disease development.
With all that in mind, Dr Levine and his colleagues conducted the current study. They included Jewish men and women, ages 16 to 19 at examination, with no history of a cancer diagnosis.
Nationwide data on 2,285,009 adolescents, collected from 1967 through 2011, were linked to Israel’s Cancer Registry to obtain the incidence of HL until 2012.
During 47 million person-years of follow-up, there were 2093 cases of HL detected.
Multivariate analysis suggested native Israelis had a significantly higher risk of developing HL than individuals who immigrated to the country (hazard ratio=1.29, P<0.001). This increased risk was driven largely by an elevated risk for the nodular sclerosis subtype of HL (hazard ratio=1.59, P<0.001).
The researchers noted that the elevated risk appeared within one generation. The low incidence of HL observed for immigrants from Western Asia was no longer evident among Israeli-born individuals of Asian origin. Among immigrants, there was no difference by age at migration.
Therefore, the researchers suggested that immigration from a low-risk to a high-risk location, mostly to locales with a modern lifestyle and environment, is associated with an increase in HL incidence (mainly the nodular sclerosis subtype) within short periods, making genetic drift unlikely as a causal explanation.
On the other hand, the Israeli lifestyle and environment, either independently or by gene-environment interaction, may pose exceptional risks for HL.
“There is increasing evidence for developmental origins of health and disease, with different possible mechanisms, including epigenetic changes or endocrine disruption,” Dr Levine said.
“There is also increasing evidence on the role of prenatal stress in offspring development, including cancer development, especially for hematological malignancies. These data suggest that risk of HL (the nodular sclerosis subtype) is possibly increased due to preconception, prenatal, or early life exposures to a changing lifestyle and environment and its interaction with susceptibility genes.”
Dr Levine and his colleagues also found a higher risk of HL for women, subjects born in more recent years, those with a higher body mass index, and subjects of taller stature. ![]()

Photo from Hebrew
University of Jerusalem
A study of 2.3 million Jewish subjects suggests that being born in Israel increases a person’s risk of developing Hodgkin lymphoma (HL).
Native Israelis had a higher risk of HL than subjects who immigrated to Israel from Europe, Asia, or North Africa.
Researchers said this finding, published in Leukemia & Lymphoma, suggests that exposure to as-yet-unidentified elements of Israel’s environment increases the risk of HL.
“While we still need further studies to identify the specific causes of the high rates of Hodgkin lymphoma among native Israelis, our findings direct us to search for possible environmental causes in Israel and the neighboring countries,” said study author Hagai Levine, MD, of Hebrew University of Jerusalem in Israel.
“These causes could be not only environmental exposures but also diet, climate, social environment, and stress that may be related to chronic regional conflict.”
Dr Levine and his colleagues noted that the incidence of HL in Israel is among the highest in the world. Based on GLOBOCAN estimates for 2012, Israeli females have the highest age-standardized incidence rate of HL worldwide, and Israeli males have the second highest. From 1960 through 2005, Israel experienced a sharp rise in HL incidence among Israeli-born individuals of both sexes.
Despite evidence for an environmental etiology, very few risk factors have been identified. Studying immigrant populations provides a means of investigating the relative importance of genetic factors as compared to environmental factors in disease development.
With all that in mind, Dr Levine and his colleagues conducted the current study. They included Jewish men and women, ages 16 to 19 at examination, with no history of a cancer diagnosis.
Nationwide data on 2,285,009 adolescents, collected from 1967 through 2011, were linked to Israel’s Cancer Registry to obtain the incidence of HL until 2012.
During 47 million person-years of follow-up, there were 2093 cases of HL detected.
Multivariate analysis suggested native Israelis had a significantly higher risk of developing HL than individuals who immigrated to the country (hazard ratio=1.29, P<0.001). This increased risk was driven largely by an elevated risk for the nodular sclerosis subtype of HL (hazard ratio=1.59, P<0.001).
The researchers noted that the elevated risk appeared within one generation. The low incidence of HL observed for immigrants from Western Asia was no longer evident among Israeli-born individuals of Asian origin. Among immigrants, there was no difference by age at migration.
Therefore, the researchers suggested that immigration from a low-risk to a high-risk location, mostly to locales with a modern lifestyle and environment, is associated with an increase in HL incidence (mainly the nodular sclerosis subtype) within short periods, making genetic drift unlikely as a causal explanation.
On the other hand, the Israeli lifestyle and environment, either independently or by gene-environment interaction, may pose exceptional risks for HL.
“There is increasing evidence for developmental origins of health and disease, with different possible mechanisms, including epigenetic changes or endocrine disruption,” Dr Levine said.
“There is also increasing evidence on the role of prenatal stress in offspring development, including cancer development, especially for hematological malignancies. These data suggest that risk of HL (the nodular sclerosis subtype) is possibly increased due to preconception, prenatal, or early life exposures to a changing lifestyle and environment and its interaction with susceptibility genes.”
Dr Levine and his colleagues also found a higher risk of HL for women, subjects born in more recent years, those with a higher body mass index, and subjects of taller stature. ![]()

Photo from Hebrew
University of Jerusalem
A study of 2.3 million Jewish subjects suggests that being born in Israel increases a person’s risk of developing Hodgkin lymphoma (HL).
Native Israelis had a higher risk of HL than subjects who immigrated to Israel from Europe, Asia, or North Africa.
Researchers said this finding, published in Leukemia & Lymphoma, suggests that exposure to as-yet-unidentified elements of Israel’s environment increases the risk of HL.
“While we still need further studies to identify the specific causes of the high rates of Hodgkin lymphoma among native Israelis, our findings direct us to search for possible environmental causes in Israel and the neighboring countries,” said study author Hagai Levine, MD, of Hebrew University of Jerusalem in Israel.
“These causes could be not only environmental exposures but also diet, climate, social environment, and stress that may be related to chronic regional conflict.”
Dr Levine and his colleagues noted that the incidence of HL in Israel is among the highest in the world. Based on GLOBOCAN estimates for 2012, Israeli females have the highest age-standardized incidence rate of HL worldwide, and Israeli males have the second highest. From 1960 through 2005, Israel experienced a sharp rise in HL incidence among Israeli-born individuals of both sexes.
Despite evidence for an environmental etiology, very few risk factors have been identified. Studying immigrant populations provides a means of investigating the relative importance of genetic factors as compared to environmental factors in disease development.
With all that in mind, Dr Levine and his colleagues conducted the current study. They included Jewish men and women, ages 16 to 19 at examination, with no history of a cancer diagnosis.
Nationwide data on 2,285,009 adolescents, collected from 1967 through 2011, were linked to Israel’s Cancer Registry to obtain the incidence of HL until 2012.
During 47 million person-years of follow-up, there were 2093 cases of HL detected.
Multivariate analysis suggested native Israelis had a significantly higher risk of developing HL than individuals who immigrated to the country (hazard ratio=1.29, P<0.001). This increased risk was driven largely by an elevated risk for the nodular sclerosis subtype of HL (hazard ratio=1.59, P<0.001).
The researchers noted that the elevated risk appeared within one generation. The low incidence of HL observed for immigrants from Western Asia was no longer evident among Israeli-born individuals of Asian origin. Among immigrants, there was no difference by age at migration.
Therefore, the researchers suggested that immigration from a low-risk to a high-risk location, mostly to locales with a modern lifestyle and environment, is associated with an increase in HL incidence (mainly the nodular sclerosis subtype) within short periods, making genetic drift unlikely as a causal explanation.
On the other hand, the Israeli lifestyle and environment, either independently or by gene-environment interaction, may pose exceptional risks for HL.
“There is increasing evidence for developmental origins of health and disease, with different possible mechanisms, including epigenetic changes or endocrine disruption,” Dr Levine said.
“There is also increasing evidence on the role of prenatal stress in offspring development, including cancer development, especially for hematological malignancies. These data suggest that risk of HL (the nodular sclerosis subtype) is possibly increased due to preconception, prenatal, or early life exposures to a changing lifestyle and environment and its interaction with susceptibility genes.”
Dr Levine and his colleagues also found a higher risk of HL for women, subjects born in more recent years, those with a higher body mass index, and subjects of taller stature. ![]()
Ties to industry among NCCN guideline authors
A study published in JAMA Oncology has quantified financial ties to the pharmaceutical industry among authors of National Comprehensive Cancer Network (NCCN) guidelines.
In 2014, the authors studied received more money in research payments than “general” payments (for things like consulting, meals, and lodging)—$29 million vs $1.25 million.
But more of the authors received general payments than research payments—84% vs 47%.
Study investigators said this finding may mean that some of the guideline authors are receiving general payments unconnected to research. However, because the study only included 1 year of data, these results may not tell the full story.
“Understanding the extent to which guideline authors have financial relationships with the pharmaceutical industry—and the types of financial arrangements that they have—is useful for the NCCN and for the public,” said investigator Stacie Dusetzina, PhD, of the University of North Carolina at Chapel Hill.
“As we learn more about the role of industry payments in shaping prescribing and practice, it is best to proceed with caution and continue to encourage transparency.”
For this study, Dr Dusetzina and her colleagues analyzed financial conflicts of interest (FCOIs) for 125 panelists who worked on setting the NCCN guidelines for lung, breast, prostate, and colorectal cancer (the cancers with the highest incidence in the US).
Eighty-six percent (n=108) of the guideline authors reported at least 1 FCOI in 2014. The total value of FCOIs was $30,287,549, which included $29,036,127 in research payments and $1,251,422 in general payments.
Eighty-four percent of the authors (n=105) received general payments, and 47% (n=59) received research payments.
The authors received an average of $10,011 in general payments and an average of $236,066 in research payments.
The majority of the payments received were within the limits set by the NCCN, but 8 authors (6%) exceeded them. The NCCN says guideline authors cannot receive $20,000 or more from a single company or $50,000 or more in total.
Dr Dusetzina and her colleagues noted that this study was not designed to explore whether the payments influenced the guideline authors’ clinical practice or the recommendations they made in the guidelines.
However, finding a high prevalence of financial relationships with industry among guideline authors lays the foundation for future studies to investigate the impact of such relationships.
“It is not a given that industry funding leads to undue influence,” Dr Dusetzina said, “but it is important to analyze these relationships and the potential impact they have on care guidelines because they do influence patient care decisions and the cost of providing patient care.” ![]()
A study published in JAMA Oncology has quantified financial ties to the pharmaceutical industry among authors of National Comprehensive Cancer Network (NCCN) guidelines.
In 2014, the authors studied received more money in research payments than “general” payments (for things like consulting, meals, and lodging)—$29 million vs $1.25 million.
But more of the authors received general payments than research payments—84% vs 47%.
Study investigators said this finding may mean that some of the guideline authors are receiving general payments unconnected to research. However, because the study only included 1 year of data, these results may not tell the full story.
“Understanding the extent to which guideline authors have financial relationships with the pharmaceutical industry—and the types of financial arrangements that they have—is useful for the NCCN and for the public,” said investigator Stacie Dusetzina, PhD, of the University of North Carolina at Chapel Hill.
“As we learn more about the role of industry payments in shaping prescribing and practice, it is best to proceed with caution and continue to encourage transparency.”
For this study, Dr Dusetzina and her colleagues analyzed financial conflicts of interest (FCOIs) for 125 panelists who worked on setting the NCCN guidelines for lung, breast, prostate, and colorectal cancer (the cancers with the highest incidence in the US).
Eighty-six percent (n=108) of the guideline authors reported at least 1 FCOI in 2014. The total value of FCOIs was $30,287,549, which included $29,036,127 in research payments and $1,251,422 in general payments.
Eighty-four percent of the authors (n=105) received general payments, and 47% (n=59) received research payments.
The authors received an average of $10,011 in general payments and an average of $236,066 in research payments.
The majority of the payments received were within the limits set by the NCCN, but 8 authors (6%) exceeded them. The NCCN says guideline authors cannot receive $20,000 or more from a single company or $50,000 or more in total.
Dr Dusetzina and her colleagues noted that this study was not designed to explore whether the payments influenced the guideline authors’ clinical practice or the recommendations they made in the guidelines.
However, finding a high prevalence of financial relationships with industry among guideline authors lays the foundation for future studies to investigate the impact of such relationships.
“It is not a given that industry funding leads to undue influence,” Dr Dusetzina said, “but it is important to analyze these relationships and the potential impact they have on care guidelines because they do influence patient care decisions and the cost of providing patient care.” ![]()
A study published in JAMA Oncology has quantified financial ties to the pharmaceutical industry among authors of National Comprehensive Cancer Network (NCCN) guidelines.
In 2014, the authors studied received more money in research payments than “general” payments (for things like consulting, meals, and lodging)—$29 million vs $1.25 million.
But more of the authors received general payments than research payments—84% vs 47%.
Study investigators said this finding may mean that some of the guideline authors are receiving general payments unconnected to research. However, because the study only included 1 year of data, these results may not tell the full story.
“Understanding the extent to which guideline authors have financial relationships with the pharmaceutical industry—and the types of financial arrangements that they have—is useful for the NCCN and for the public,” said investigator Stacie Dusetzina, PhD, of the University of North Carolina at Chapel Hill.
“As we learn more about the role of industry payments in shaping prescribing and practice, it is best to proceed with caution and continue to encourage transparency.”
For this study, Dr Dusetzina and her colleagues analyzed financial conflicts of interest (FCOIs) for 125 panelists who worked on setting the NCCN guidelines for lung, breast, prostate, and colorectal cancer (the cancers with the highest incidence in the US).
Eighty-six percent (n=108) of the guideline authors reported at least 1 FCOI in 2014. The total value of FCOIs was $30,287,549, which included $29,036,127 in research payments and $1,251,422 in general payments.
Eighty-four percent of the authors (n=105) received general payments, and 47% (n=59) received research payments.
The authors received an average of $10,011 in general payments and an average of $236,066 in research payments.
The majority of the payments received were within the limits set by the NCCN, but 8 authors (6%) exceeded them. The NCCN says guideline authors cannot receive $20,000 or more from a single company or $50,000 or more in total.
Dr Dusetzina and her colleagues noted that this study was not designed to explore whether the payments influenced the guideline authors’ clinical practice or the recommendations they made in the guidelines.
However, finding a high prevalence of financial relationships with industry among guideline authors lays the foundation for future studies to investigate the impact of such relationships.
“It is not a given that industry funding leads to undue influence,” Dr Dusetzina said, “but it is important to analyze these relationships and the potential impact they have on care guidelines because they do influence patient care decisions and the cost of providing patient care.” ![]()
Potential therapeutic target for leukemia, other cancers

Photo by Thomas Semkow
Preclinical research indicates that a member of the Mediator protein complex plays a key role in hematopoiesis.
Investigators found that MED12 was required for the survival of hematopoietic stem and progenitor cells (HSPCs).
The team said this finding, along with the fact that MED12 mutations have been linked to leukemia and solid tumor malignancies, suggests that targeting MED12 hyperactivity might be a useful strategy for treating cancers.
“Because MED12 appears to be so essential to hematopoiesis, our study points to it as a possible target for future anticancer therapies for both chronic and acute forms of leukemia,” said Iannis Aifantis, PhD, of NYU Langone Medical Center in New York.
“Our study also suggests that MED12 hyperactivation or loss of control is a possible explanation for what factors may trigger these cancers and other solid tumors.”
Dr Aifantis and his colleagues described their study in Cell Stem Cell.
The investigators first analyzed the effects of MED12 deletion in mice. Mice bred to lack MED12 died within 2 weeks of birth and showed evidence of aberrant hematopoiesis—namely, a “severe reduction of bone marrow and thymus cellularity.”
Adult mice that were engineered to lose expression of MED12 after the injection of an activating molecule experienced a “rapid” reduction in bone marrow cellularity, as well as reductions in spleen and thymus size. These mice also had low white blood cell and platelet counts and died within 3 weeks of MED12 deletion.
Subsequent analyses of the animals’ bone marrow showed that estimates of HSPCs in each mouse fell from nearly 150,000 to 15,000 within 4 days of injection. Within 10 days, there were no HSPCs left.
Deleting MED12 was also lethal for human HSPCs. Colonies of CD34+ cells dropped from an average of 25 per plate to 5 per plate within 10 days of MED12 deletion.
On the other hand, MED12 did not affect the survival of other cell types. For example, MED12 deletion did not impact mouse embryonic fibroblasts, embryonic stem cells, or hair follicle stem cells.
In addition, deleting members of the Mediator kinase module besides MED12—MED13, CDK8, or CYCLIN C—did not have a significant effect on HSPCs and did not kill mice. The investigators said this provides further evidence that MED12—by loss of its function alone—is essential for hematopoiesis.
The team found that MED12 deletion destabilizes P300 binding at lineage-specific enhancers, which results in H3K27Ac depletion, enhancer de-activation, and the consequent loss of hematopoietic stem cell gene expression signatures.
As a next step, the investigators plan to screen blood samples from cancer patients for signs of MED12 mutations and uncontrolled HSPC development.
The team also hopes to determine the biological mechanisms involved in MED12 hyperactivation and identify drug molecules that could block MED12 hyperactivity and serve as potential MED12 inhibitors. ![]()

Photo by Thomas Semkow
Preclinical research indicates that a member of the Mediator protein complex plays a key role in hematopoiesis.
Investigators found that MED12 was required for the survival of hematopoietic stem and progenitor cells (HSPCs).
The team said this finding, along with the fact that MED12 mutations have been linked to leukemia and solid tumor malignancies, suggests that targeting MED12 hyperactivity might be a useful strategy for treating cancers.
“Because MED12 appears to be so essential to hematopoiesis, our study points to it as a possible target for future anticancer therapies for both chronic and acute forms of leukemia,” said Iannis Aifantis, PhD, of NYU Langone Medical Center in New York.
“Our study also suggests that MED12 hyperactivation or loss of control is a possible explanation for what factors may trigger these cancers and other solid tumors.”
Dr Aifantis and his colleagues described their study in Cell Stem Cell.
The investigators first analyzed the effects of MED12 deletion in mice. Mice bred to lack MED12 died within 2 weeks of birth and showed evidence of aberrant hematopoiesis—namely, a “severe reduction of bone marrow and thymus cellularity.”
Adult mice that were engineered to lose expression of MED12 after the injection of an activating molecule experienced a “rapid” reduction in bone marrow cellularity, as well as reductions in spleen and thymus size. These mice also had low white blood cell and platelet counts and died within 3 weeks of MED12 deletion.
Subsequent analyses of the animals’ bone marrow showed that estimates of HSPCs in each mouse fell from nearly 150,000 to 15,000 within 4 days of injection. Within 10 days, there were no HSPCs left.
Deleting MED12 was also lethal for human HSPCs. Colonies of CD34+ cells dropped from an average of 25 per plate to 5 per plate within 10 days of MED12 deletion.
On the other hand, MED12 did not affect the survival of other cell types. For example, MED12 deletion did not impact mouse embryonic fibroblasts, embryonic stem cells, or hair follicle stem cells.
In addition, deleting members of the Mediator kinase module besides MED12—MED13, CDK8, or CYCLIN C—did not have a significant effect on HSPCs and did not kill mice. The investigators said this provides further evidence that MED12—by loss of its function alone—is essential for hematopoiesis.
The team found that MED12 deletion destabilizes P300 binding at lineage-specific enhancers, which results in H3K27Ac depletion, enhancer de-activation, and the consequent loss of hematopoietic stem cell gene expression signatures.
As a next step, the investigators plan to screen blood samples from cancer patients for signs of MED12 mutations and uncontrolled HSPC development.
The team also hopes to determine the biological mechanisms involved in MED12 hyperactivation and identify drug molecules that could block MED12 hyperactivity and serve as potential MED12 inhibitors. ![]()

Photo by Thomas Semkow
Preclinical research indicates that a member of the Mediator protein complex plays a key role in hematopoiesis.
Investigators found that MED12 was required for the survival of hematopoietic stem and progenitor cells (HSPCs).
The team said this finding, along with the fact that MED12 mutations have been linked to leukemia and solid tumor malignancies, suggests that targeting MED12 hyperactivity might be a useful strategy for treating cancers.
“Because MED12 appears to be so essential to hematopoiesis, our study points to it as a possible target for future anticancer therapies for both chronic and acute forms of leukemia,” said Iannis Aifantis, PhD, of NYU Langone Medical Center in New York.
“Our study also suggests that MED12 hyperactivation or loss of control is a possible explanation for what factors may trigger these cancers and other solid tumors.”
Dr Aifantis and his colleagues described their study in Cell Stem Cell.
The investigators first analyzed the effects of MED12 deletion in mice. Mice bred to lack MED12 died within 2 weeks of birth and showed evidence of aberrant hematopoiesis—namely, a “severe reduction of bone marrow and thymus cellularity.”
Adult mice that were engineered to lose expression of MED12 after the injection of an activating molecule experienced a “rapid” reduction in bone marrow cellularity, as well as reductions in spleen and thymus size. These mice also had low white blood cell and platelet counts and died within 3 weeks of MED12 deletion.
Subsequent analyses of the animals’ bone marrow showed that estimates of HSPCs in each mouse fell from nearly 150,000 to 15,000 within 4 days of injection. Within 10 days, there were no HSPCs left.
Deleting MED12 was also lethal for human HSPCs. Colonies of CD34+ cells dropped from an average of 25 per plate to 5 per plate within 10 days of MED12 deletion.
On the other hand, MED12 did not affect the survival of other cell types. For example, MED12 deletion did not impact mouse embryonic fibroblasts, embryonic stem cells, or hair follicle stem cells.
In addition, deleting members of the Mediator kinase module besides MED12—MED13, CDK8, or CYCLIN C—did not have a significant effect on HSPCs and did not kill mice. The investigators said this provides further evidence that MED12—by loss of its function alone—is essential for hematopoiesis.
The team found that MED12 deletion destabilizes P300 binding at lineage-specific enhancers, which results in H3K27Ac depletion, enhancer de-activation, and the consequent loss of hematopoietic stem cell gene expression signatures.
As a next step, the investigators plan to screen blood samples from cancer patients for signs of MED12 mutations and uncontrolled HSPC development.
The team also hopes to determine the biological mechanisms involved in MED12 hyperactivation and identify drug molecules that could block MED12 hyperactivity and serve as potential MED12 inhibitors. ![]()
NICE approves bosutinib for routine NHS use
The National Institute for Health and Care Excellence (NICE) has issued a final guidance recommending that bosutinib (Bosulif), a tyrosine kinase inhibitor used to treat certain patients with chronic myeloid leukemia (CML), be made available through the National Health Service (NHS).
This means patients will no longer have to apply to the Cancer Drugs Fund (CDF) to obtain bosutinib.
The CDF is money the government sets aside to pay for cancer drugs that haven’t been approved by NICE and aren’t available within the NHS in England.
Following the decision to reform the CDF earlier this year, NICE began to reappraise all drugs currently in the CDF in April. Bosutinib is the first drug to be looked at through this reconsideration process.
NICE previously considered making bosutinib available through the NHS in 2013 but decided the drug was not cost-effective. So bosutinib was made available to patients via the CDF.
As part of the reappraisal process, Pfizer offered a discount for bosutinib. Taking this discount into consideration, as well as the limited treatment options for CML patients, NICE decided bosutinib is cost-effective.
Bosutinib has conditional approval from the European Commission to treat adults with Philadelphia-chromosome-positive CML in chronic phase, accelerated phase, or blast phase, but only if those patients have previously received one or more tyrosine kinase inhibitors and are not considered eligible for treatment with imatinib, nilotinib, or dasatinib. ![]()

The National Institute for Health and Care Excellence (NICE) has issued a final guidance recommending that bosutinib (Bosulif), a tyrosine kinase inhibitor used to treat certain patients with chronic myeloid leukemia (CML), be made available through the National Health Service (NHS).
This means patients will no longer have to apply to the Cancer Drugs Fund (CDF) to obtain bosutinib.
The CDF is money the government sets aside to pay for cancer drugs that haven’t been approved by NICE and aren’t available within the NHS in England.
Following the decision to reform the CDF earlier this year, NICE began to reappraise all drugs currently in the CDF in April. Bosutinib is the first drug to be looked at through this reconsideration process.
NICE previously considered making bosutinib available through the NHS in 2013 but decided the drug was not cost-effective. So bosutinib was made available to patients via the CDF.
As part of the reappraisal process, Pfizer offered a discount for bosutinib. Taking this discount into consideration, as well as the limited treatment options for CML patients, NICE decided bosutinib is cost-effective.
Bosutinib has conditional approval from the European Commission to treat adults with Philadelphia-chromosome-positive CML in chronic phase, accelerated phase, or blast phase, but only if those patients have previously received one or more tyrosine kinase inhibitors and are not considered eligible for treatment with imatinib, nilotinib, or dasatinib. ![]()

The National Institute for Health and Care Excellence (NICE) has issued a final guidance recommending that bosutinib (Bosulif), a tyrosine kinase inhibitor used to treat certain patients with chronic myeloid leukemia (CML), be made available through the National Health Service (NHS).
This means patients will no longer have to apply to the Cancer Drugs Fund (CDF) to obtain bosutinib.
The CDF is money the government sets aside to pay for cancer drugs that haven’t been approved by NICE and aren’t available within the NHS in England.
Following the decision to reform the CDF earlier this year, NICE began to reappraise all drugs currently in the CDF in April. Bosutinib is the first drug to be looked at through this reconsideration process.
NICE previously considered making bosutinib available through the NHS in 2013 but decided the drug was not cost-effective. So bosutinib was made available to patients via the CDF.
As part of the reappraisal process, Pfizer offered a discount for bosutinib. Taking this discount into consideration, as well as the limited treatment options for CML patients, NICE decided bosutinib is cost-effective.
Bosutinib has conditional approval from the European Commission to treat adults with Philadelphia-chromosome-positive CML in chronic phase, accelerated phase, or blast phase, but only if those patients have previously received one or more tyrosine kinase inhibitors and are not considered eligible for treatment with imatinib, nilotinib, or dasatinib. ![]()
FDA grants drug orphan designation for CLL

The US Food and Drug Administration (FDA) has granted orphan drug designation to the PI3K delta inhibitor TGR-1202 for the treatment of chronic lymphocytic leukemia (CLL).
The FDA grants orphan designation to drugs and biologics intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases.
This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the drug is approved.
About TGR-1202
TG Therapeutics, Inc. is developing TGR-1202 as a treatment for hematologic malignancies.
The drug is currently being evaluated in the phase 3 UNITY-CLL trial (NCT02612311), which includes patients with previously treated and untreated CLL. Patients are receiving TGR-1202 plus ublituximab, obinutuzumab plus chlorambucil, ublituximab alone, or TGR-1202 alone.
At EHA 2016, researchers reported preliminary results of a phase 1/1b study (NCT02268851) of TGR-1202 in combination with ibrutinib in patients with relapsed/refractory CLL or mantle cell lymphoma.
At ASCO 2016, researchers reported long-term follow-up of 2 studies of TGR-1202.
The first (TGR-1202-101, NCT01767766) is a phase 1 study of TGR-1202 in patients with relapsed or refractory hematologic malignancies.
The second (UTX-TGR-103, NCT02006485) is a phase 1/1b trial evaluating the combination of ublituximab and TGR-1202 in patients with relapsed or refractory non-Hodgkin lymphoma or CLL.
At ASH 2015, researchers reported results of a phase 1 trial (TGR-GA-106, NCT02100852) of TGR-1202 in combination with obinutuzumab and chlorambucil in patients with CLL. ![]()

The US Food and Drug Administration (FDA) has granted orphan drug designation to the PI3K delta inhibitor TGR-1202 for the treatment of chronic lymphocytic leukemia (CLL).
The FDA grants orphan designation to drugs and biologics intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases.
This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the drug is approved.
About TGR-1202
TG Therapeutics, Inc. is developing TGR-1202 as a treatment for hematologic malignancies.
The drug is currently being evaluated in the phase 3 UNITY-CLL trial (NCT02612311), which includes patients with previously treated and untreated CLL. Patients are receiving TGR-1202 plus ublituximab, obinutuzumab plus chlorambucil, ublituximab alone, or TGR-1202 alone.
At EHA 2016, researchers reported preliminary results of a phase 1/1b study (NCT02268851) of TGR-1202 in combination with ibrutinib in patients with relapsed/refractory CLL or mantle cell lymphoma.
At ASCO 2016, researchers reported long-term follow-up of 2 studies of TGR-1202.
The first (TGR-1202-101, NCT01767766) is a phase 1 study of TGR-1202 in patients with relapsed or refractory hematologic malignancies.
The second (UTX-TGR-103, NCT02006485) is a phase 1/1b trial evaluating the combination of ublituximab and TGR-1202 in patients with relapsed or refractory non-Hodgkin lymphoma or CLL.
At ASH 2015, researchers reported results of a phase 1 trial (TGR-GA-106, NCT02100852) of TGR-1202 in combination with obinutuzumab and chlorambucil in patients with CLL. ![]()

The US Food and Drug Administration (FDA) has granted orphan drug designation to the PI3K delta inhibitor TGR-1202 for the treatment of chronic lymphocytic leukemia (CLL).
The FDA grants orphan designation to drugs and biologics intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases.
This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the drug is approved.
About TGR-1202
TG Therapeutics, Inc. is developing TGR-1202 as a treatment for hematologic malignancies.
The drug is currently being evaluated in the phase 3 UNITY-CLL trial (NCT02612311), which includes patients with previously treated and untreated CLL. Patients are receiving TGR-1202 plus ublituximab, obinutuzumab plus chlorambucil, ublituximab alone, or TGR-1202 alone.
At EHA 2016, researchers reported preliminary results of a phase 1/1b study (NCT02268851) of TGR-1202 in combination with ibrutinib in patients with relapsed/refractory CLL or mantle cell lymphoma.
At ASCO 2016, researchers reported long-term follow-up of 2 studies of TGR-1202.
The first (TGR-1202-101, NCT01767766) is a phase 1 study of TGR-1202 in patients with relapsed or refractory hematologic malignancies.
The second (UTX-TGR-103, NCT02006485) is a phase 1/1b trial evaluating the combination of ublituximab and TGR-1202 in patients with relapsed or refractory non-Hodgkin lymphoma or CLL.
At ASH 2015, researchers reported results of a phase 1 trial (TGR-GA-106, NCT02100852) of TGR-1202 in combination with obinutuzumab and chlorambucil in patients with CLL. ![]()
Excess weight linked to myeloma, other cancers

An analysis of more than 1000 studies suggests multiple myeloma (MM) and 12 other cancers are associated with excess weight.
The data suggest that limiting weight gain over time could help reduce a person’s risk of developing these cancers.
A working group convened by the International Agency for Research on Cancer (IARC) conducted this analysis and reported the results in NEJM.
“The burden of cancer due to being overweight or obese is more extensive than what has been assumed,” said Graham Colditz, MD, DrPH, who chaired the IARC working group.
“Many of the newly identified cancers linked to excess weight haven’t been on people’s radar screens as having a weight component.”
In 2002, an IARC working group reported finding sufficient evidence linking excess weight to higher risks of colon, esophageal, kidney, breast, and uterine cancers.
Now, another IARC working group has found evidence linking excess weight and additional cancers.
The group reviewed more than 1000 epidemiologic studies. Most of the studies provided cancer risk estimates for adult body mass index (BMI), although some provided estimates for BMI or body shape in childhood/adolescence, changes in BMI or weight over time, or other indicators of adiposity.
The IARC working group reported the relative risk (RR) of developing various cancers for the highest BMI category evaluated, versus a normal BMI.
The group said there was sufficient evidence linking excess weight to the following cancers: adenocarcinoma (RR=4.8), gastric cardia (RR=1.8), colon and rectal cancer (RR=1.3), liver cancer (RR=1.8), gallbladder cancer (RR=1.3), pancreatic cancer (RR=1.5), postmenopausal breast cancer (RR=1.1), corpus uteri (RR=7.1), ovarian cancer (RR=1.1), renal cell cancer (RR=1.8), meningioma (RR=1.5), thyroid cancer (RR=1.1), and MM (RR=1.5).
Looking more closely at MM, the RR was 1.2 for adults who were overweight (BMI 25-29.9), 1.2 for those with class 1 obesity (BMI 30-34.9), and 1.5 for those with class 2 or 3 obesity (BMI 35-40+).
For most of the cancers, there was positive dose-response relationship; in other words, the higher the BMI, the greater the cancer risk.
In addition, the cancer risks associated with excess weight were similar for men and women and were consistent across geographic regions—North America, Europe, Asia, and the Middle East—where data were available.
“Significant numbers of the US and the world’s population are overweight,” Dr Colditz noted. “This is another wake-up call. It’s time to take our health and our diets seriously.”
Dr Colditz conceded that losing weight can be difficult. Therefore, he recommended that people who struggle with weight loss should focus on avoiding weight gain to reduce their risk of developing certain cancers. ![]()

An analysis of more than 1000 studies suggests multiple myeloma (MM) and 12 other cancers are associated with excess weight.
The data suggest that limiting weight gain over time could help reduce a person’s risk of developing these cancers.
A working group convened by the International Agency for Research on Cancer (IARC) conducted this analysis and reported the results in NEJM.
“The burden of cancer due to being overweight or obese is more extensive than what has been assumed,” said Graham Colditz, MD, DrPH, who chaired the IARC working group.
“Many of the newly identified cancers linked to excess weight haven’t been on people’s radar screens as having a weight component.”
In 2002, an IARC working group reported finding sufficient evidence linking excess weight to higher risks of colon, esophageal, kidney, breast, and uterine cancers.
Now, another IARC working group has found evidence linking excess weight and additional cancers.
The group reviewed more than 1000 epidemiologic studies. Most of the studies provided cancer risk estimates for adult body mass index (BMI), although some provided estimates for BMI or body shape in childhood/adolescence, changes in BMI or weight over time, or other indicators of adiposity.
The IARC working group reported the relative risk (RR) of developing various cancers for the highest BMI category evaluated, versus a normal BMI.
The group said there was sufficient evidence linking excess weight to the following cancers: adenocarcinoma (RR=4.8), gastric cardia (RR=1.8), colon and rectal cancer (RR=1.3), liver cancer (RR=1.8), gallbladder cancer (RR=1.3), pancreatic cancer (RR=1.5), postmenopausal breast cancer (RR=1.1), corpus uteri (RR=7.1), ovarian cancer (RR=1.1), renal cell cancer (RR=1.8), meningioma (RR=1.5), thyroid cancer (RR=1.1), and MM (RR=1.5).
Looking more closely at MM, the RR was 1.2 for adults who were overweight (BMI 25-29.9), 1.2 for those with class 1 obesity (BMI 30-34.9), and 1.5 for those with class 2 or 3 obesity (BMI 35-40+).
For most of the cancers, there was positive dose-response relationship; in other words, the higher the BMI, the greater the cancer risk.
In addition, the cancer risks associated with excess weight were similar for men and women and were consistent across geographic regions—North America, Europe, Asia, and the Middle East—where data were available.
“Significant numbers of the US and the world’s population are overweight,” Dr Colditz noted. “This is another wake-up call. It’s time to take our health and our diets seriously.”
Dr Colditz conceded that losing weight can be difficult. Therefore, he recommended that people who struggle with weight loss should focus on avoiding weight gain to reduce their risk of developing certain cancers. ![]()

An analysis of more than 1000 studies suggests multiple myeloma (MM) and 12 other cancers are associated with excess weight.
The data suggest that limiting weight gain over time could help reduce a person’s risk of developing these cancers.
A working group convened by the International Agency for Research on Cancer (IARC) conducted this analysis and reported the results in NEJM.
“The burden of cancer due to being overweight or obese is more extensive than what has been assumed,” said Graham Colditz, MD, DrPH, who chaired the IARC working group.
“Many of the newly identified cancers linked to excess weight haven’t been on people’s radar screens as having a weight component.”
In 2002, an IARC working group reported finding sufficient evidence linking excess weight to higher risks of colon, esophageal, kidney, breast, and uterine cancers.
Now, another IARC working group has found evidence linking excess weight and additional cancers.
The group reviewed more than 1000 epidemiologic studies. Most of the studies provided cancer risk estimates for adult body mass index (BMI), although some provided estimates for BMI or body shape in childhood/adolescence, changes in BMI or weight over time, or other indicators of adiposity.
The IARC working group reported the relative risk (RR) of developing various cancers for the highest BMI category evaluated, versus a normal BMI.
The group said there was sufficient evidence linking excess weight to the following cancers: adenocarcinoma (RR=4.8), gastric cardia (RR=1.8), colon and rectal cancer (RR=1.3), liver cancer (RR=1.8), gallbladder cancer (RR=1.3), pancreatic cancer (RR=1.5), postmenopausal breast cancer (RR=1.1), corpus uteri (RR=7.1), ovarian cancer (RR=1.1), renal cell cancer (RR=1.8), meningioma (RR=1.5), thyroid cancer (RR=1.1), and MM (RR=1.5).
Looking more closely at MM, the RR was 1.2 for adults who were overweight (BMI 25-29.9), 1.2 for those with class 1 obesity (BMI 30-34.9), and 1.5 for those with class 2 or 3 obesity (BMI 35-40+).
For most of the cancers, there was positive dose-response relationship; in other words, the higher the BMI, the greater the cancer risk.
In addition, the cancer risks associated with excess weight were similar for men and women and were consistent across geographic regions—North America, Europe, Asia, and the Middle East—where data were available.
“Significant numbers of the US and the world’s population are overweight,” Dr Colditz noted. “This is another wake-up call. It’s time to take our health and our diets seriously.”
Dr Colditz conceded that losing weight can be difficult. Therefore, he recommended that people who struggle with weight loss should focus on avoiding weight gain to reduce their risk of developing certain cancers.
Reversible FXIa inhibitor may reduce bleeding risk

Photo by Sakurai Midori
Researchers say they have developed an antibody that inhibits factor XIa (FXIa) without increasing the risk of bleeding, as well as an antibody that can reverse the inhibitor’s effects.
The inhibitor—known as DEF—staved off clotting in human blood and animal models.
Even when it was given at extremely high doses, DEF did not induce bleeding in the animals.
Still, the researchers developed an antibody—revC4—that can reverse DEF’s activity.
Tovo David, PhD, of University of California, San Francisco, and his colleagues described this work in Science Translational Medicine.
Initially, the researchers generated a series of human immunoglobulin Gs (IgGs) that blocked FXIa active-site function but did not bind FXI zymogen or other coagulation proteases.
The most potent of these was C24. This IgG inhibited FXIIa-induced thrombin generation and intrinsic pathway–triggered clot formation in human plasma and whole blood. C24 also inhibited FeCl3-induced arterial thrombosis in an FXI-humanized mouse.
Dr David and his colleagues then set out to improve upon C24, rendering it unable to activate complement, engage Fc receptors, or activate platelets and other cells. The resulting molecule was DEF.
The researchers tested DEF in rabbits and cynomolgus macaques. At doses much higher than those required to inhibit thrombus formation, DEF did not increase cuticle bleeding in the rabbits or cause spontaneous bleeding in the monkeys.
Despite this lack of bleeding, Dr David and his colleagues generated a human IgG that can reverse DEF activity because FXI deficiency can be associated with bleeding in humans.
This reversal agent—revC4—proved effective in human plasma (ex vivo) and in rabbits. revC4 reversed the anticoagulant effect of DEF within 30 minutes of dosing.
Based on these results, the researchers concluded that, with further development, their reversible FXIa-specific antibody might provide a new—and potentially safer—type of anticoagulant.

Photo by Sakurai Midori
Researchers say they have developed an antibody that inhibits factor XIa (FXIa) without increasing the risk of bleeding, as well as an antibody that can reverse the inhibitor’s effects.
The inhibitor—known as DEF—staved off clotting in human blood and animal models.
Even when it was given at extremely high doses, DEF did not induce bleeding in the animals.
Still, the researchers developed an antibody—revC4—that can reverse DEF’s activity.
Tovo David, PhD, of University of California, San Francisco, and his colleagues described this work in Science Translational Medicine.
Initially, the researchers generated a series of human immunoglobulin Gs (IgGs) that blocked FXIa active-site function but did not bind FXI zymogen or other coagulation proteases.
The most potent of these was C24. This IgG inhibited FXIIa-induced thrombin generation and intrinsic pathway–triggered clot formation in human plasma and whole blood. C24 also inhibited FeCl3-induced arterial thrombosis in an FXI-humanized mouse.
Dr David and his colleagues then set out to improve upon C24, rendering it unable to activate complement, engage Fc receptors, or activate platelets and other cells. The resulting molecule was DEF.
The researchers tested DEF in rabbits and cynomolgus macaques. At doses much higher than those required to inhibit thrombus formation, DEF did not increase cuticle bleeding in the rabbits or cause spontaneous bleeding in the monkeys.
Despite this lack of bleeding, Dr David and his colleagues generated a human IgG that can reverse DEF activity because FXI deficiency can be associated with bleeding in humans.
This reversal agent—revC4—proved effective in human plasma (ex vivo) and in rabbits. revC4 reversed the anticoagulant effect of DEF within 30 minutes of dosing.
Based on these results, the researchers concluded that, with further development, their reversible FXIa-specific antibody might provide a new—and potentially safer—type of anticoagulant.

Photo by Sakurai Midori
Researchers say they have developed an antibody that inhibits factor XIa (FXIa) without increasing the risk of bleeding, as well as an antibody that can reverse the inhibitor’s effects.
The inhibitor—known as DEF—staved off clotting in human blood and animal models.
Even when it was given at extremely high doses, DEF did not induce bleeding in the animals.
Still, the researchers developed an antibody—revC4—that can reverse DEF’s activity.
Tovo David, PhD, of University of California, San Francisco, and his colleagues described this work in Science Translational Medicine.
Initially, the researchers generated a series of human immunoglobulin Gs (IgGs) that blocked FXIa active-site function but did not bind FXI zymogen or other coagulation proteases.
The most potent of these was C24. This IgG inhibited FXIIa-induced thrombin generation and intrinsic pathway–triggered clot formation in human plasma and whole blood. C24 also inhibited FeCl3-induced arterial thrombosis in an FXI-humanized mouse.
Dr David and his colleagues then set out to improve upon C24, rendering it unable to activate complement, engage Fc receptors, or activate platelets and other cells. The resulting molecule was DEF.
The researchers tested DEF in rabbits and cynomolgus macaques. At doses much higher than those required to inhibit thrombus formation, DEF did not increase cuticle bleeding in the rabbits or cause spontaneous bleeding in the monkeys.
Despite this lack of bleeding, Dr David and his colleagues generated a human IgG that can reverse DEF activity because FXI deficiency can be associated with bleeding in humans.
This reversal agent—revC4—proved effective in human plasma (ex vivo) and in rabbits. revC4 reversed the anticoagulant effect of DEF within 30 minutes of dosing.
Based on these results, the researchers concluded that, with further development, their reversible FXIa-specific antibody might provide a new—and potentially safer—type of anticoagulant.
‘Barcoding’ reveals insights regarding HSCs

in the bone marrow
By assigning a “barcode” to hematopoietic stem cells (HSCs), researchers have found they can monitor the cells and study changes that occur over
time.
In tracking the barcoded HSCs, the team discovered why B-1a cells develop primarily during fetal and neonatal life, while adult bone marrow HSCs preferentially give rise to B-2 cells.
Joan Yuan, PhD, of Lund University in Sweden, and her colleagues described this discovery in Immunity.
“By assigning a barcode to the stem cells, we were able to track their performance over long periods of time and see which cells in the blood and the immune system they can induce,” Dr Yuan explained.
“Without the barcode, we only see a bunch of red and white blood cells, without knowing how they are related. This allows us to track which stem cell has given rise to which subsidiary cells and thereby distinguish the ‘family tree’ in the blood.”
In this way, the researchers found that B-1a cells and B-2 cells have a shared precursor in the fetal liver. And definitive fetal liver HSCs gave rise to both B-1a and B-2 cells. However, over time, the HSCs were not able to maintain B-1a output.
“The same stem cells exist within adults [and fetuses], but they have lost their ability to regenerate the entire immune system [in adulthood],” said study author Trine Kristiansen, a doctoral student at Lund University.
“By adding a protein normally only found in the stem cells of a fetus, we were able to reconstruct [the HSCs’] capacity to produce white blood cells.”
The researchers restored the HSC’s ability to produce B-1a cells by inducing expression of the RNA binding protein LIN28B, which regulates fetal hematopoiesis.
The team said these results suggest the decline in regenerative potential is a reversible state for HSCs. The researchers believe this finding could have implications for the treatment of blood disorders and particularly for HSC transplant.
“In this treatment, the patient’s blood system is replaced with that of an adult donor, which could mean losing the B cells that are only produced in fetuses,” Kristiansen said.
Without these cells, a person is at risk of developing immune system disorders that can lead to severe infections and autoimmune diseases.
“Every day, millions of blood cells die, and they can emit DNA and other debris that cause inflammation if not taken care of by the white blood cells,” said study author Elin Jaensson Gyllenbäck, PhD, of Lund University.
“The discovery is a step towards understanding which processes create a proper immune system for those who suffer from blood diseases.”

in the bone marrow
By assigning a “barcode” to hematopoietic stem cells (HSCs), researchers have found they can monitor the cells and study changes that occur over
time.
In tracking the barcoded HSCs, the team discovered why B-1a cells develop primarily during fetal and neonatal life, while adult bone marrow HSCs preferentially give rise to B-2 cells.
Joan Yuan, PhD, of Lund University in Sweden, and her colleagues described this discovery in Immunity.
“By assigning a barcode to the stem cells, we were able to track their performance over long periods of time and see which cells in the blood and the immune system they can induce,” Dr Yuan explained.
“Without the barcode, we only see a bunch of red and white blood cells, without knowing how they are related. This allows us to track which stem cell has given rise to which subsidiary cells and thereby distinguish the ‘family tree’ in the blood.”
In this way, the researchers found that B-1a cells and B-2 cells have a shared precursor in the fetal liver. And definitive fetal liver HSCs gave rise to both B-1a and B-2 cells. However, over time, the HSCs were not able to maintain B-1a output.
“The same stem cells exist within adults [and fetuses], but they have lost their ability to regenerate the entire immune system [in adulthood],” said study author Trine Kristiansen, a doctoral student at Lund University.
“By adding a protein normally only found in the stem cells of a fetus, we were able to reconstruct [the HSCs’] capacity to produce white blood cells.”
The researchers restored the HSC’s ability to produce B-1a cells by inducing expression of the RNA binding protein LIN28B, which regulates fetal hematopoiesis.
The team said these results suggest the decline in regenerative potential is a reversible state for HSCs. The researchers believe this finding could have implications for the treatment of blood disorders and particularly for HSC transplant.
“In this treatment, the patient’s blood system is replaced with that of an adult donor, which could mean losing the B cells that are only produced in fetuses,” Kristiansen said.
Without these cells, a person is at risk of developing immune system disorders that can lead to severe infections and autoimmune diseases.
“Every day, millions of blood cells die, and they can emit DNA and other debris that cause inflammation if not taken care of by the white blood cells,” said study author Elin Jaensson Gyllenbäck, PhD, of Lund University.
“The discovery is a step towards understanding which processes create a proper immune system for those who suffer from blood diseases.”

in the bone marrow
By assigning a “barcode” to hematopoietic stem cells (HSCs), researchers have found they can monitor the cells and study changes that occur over
time.
In tracking the barcoded HSCs, the team discovered why B-1a cells develop primarily during fetal and neonatal life, while adult bone marrow HSCs preferentially give rise to B-2 cells.
Joan Yuan, PhD, of Lund University in Sweden, and her colleagues described this discovery in Immunity.
“By assigning a barcode to the stem cells, we were able to track their performance over long periods of time and see which cells in the blood and the immune system they can induce,” Dr Yuan explained.
“Without the barcode, we only see a bunch of red and white blood cells, without knowing how they are related. This allows us to track which stem cell has given rise to which subsidiary cells and thereby distinguish the ‘family tree’ in the blood.”
In this way, the researchers found that B-1a cells and B-2 cells have a shared precursor in the fetal liver. And definitive fetal liver HSCs gave rise to both B-1a and B-2 cells. However, over time, the HSCs were not able to maintain B-1a output.
“The same stem cells exist within adults [and fetuses], but they have lost their ability to regenerate the entire immune system [in adulthood],” said study author Trine Kristiansen, a doctoral student at Lund University.
“By adding a protein normally only found in the stem cells of a fetus, we were able to reconstruct [the HSCs’] capacity to produce white blood cells.”
The researchers restored the HSC’s ability to produce B-1a cells by inducing expression of the RNA binding protein LIN28B, which regulates fetal hematopoiesis.
The team said these results suggest the decline in regenerative potential is a reversible state for HSCs. The researchers believe this finding could have implications for the treatment of blood disorders and particularly for HSC transplant.
“In this treatment, the patient’s blood system is replaced with that of an adult donor, which could mean losing the B cells that are only produced in fetuses,” Kristiansen said.
Without these cells, a person is at risk of developing immune system disorders that can lead to severe infections and autoimmune diseases.
“Every day, millions of blood cells die, and they can emit DNA and other debris that cause inflammation if not taken care of by the white blood cells,” said study author Elin Jaensson Gyllenbäck, PhD, of Lund University.
“The discovery is a step towards understanding which processes create a proper immune system for those who suffer from blood diseases.”
Nanoparticles speed blood clotting in multiple models
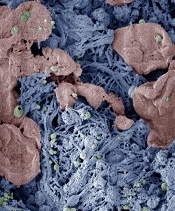
form clots in injured liver
Image courtesy of Erin Lavik
PHILADELPHIA—Researchers say they have developed nanoparticles that can congregate at sites of injury and speed blood clotting in vitro and in vivo.
The nanoparticles are designed to bind with activated platelets and help them join together to form clots faster.
The nanoparticles have helped stop bleeding and improved animals’ survival in different models of injury, without producing off-target clotting.
“When you have uncontrolled internal bleeding, that’s when these particles could really make a difference,” said Erin B. Lavik, ScD, of the University of Maryland, Baltimore County (UMBC) in Baltimore, Maryland.
“Compared to injuries that aren’t treated with the nanoparticles, we can cut bleeding time in half and reduce total blood loss.”
Dr Lavik discussed this work at the 252nd National Meeting & Exposition of the American Chemical Society.
She and her colleagues have been testing different versions of these nanoparticles for years. They previously reported results with the particles in 2012 and 2014.
The current nanoparticles are made from polylactic acid or polylactic co-glycolic acid. Dr Lavik said that by using the 2 different materials, the researchers can change the temperature at which the nanoparticles melt. Thus far, the team has developed nanoparticles that are stable up to 50° C (122° F).
The nanoparticles have an outer coating of polyethylene glycol and an attached peptide—RGD—that enables them to bind to natural platelets. Dr Lavik said the researchers chose RGD because platelets have receptors for that peptide, it’s ubiquitous in the body, and it wouldn’t alter the chemistry of the nanoparticles.
The researchers have tested the nanoparticles in a femoral injury model, a liver injury model, and blast injury model. The team has also started testing in models of brain and spinal cord injury, but the results are “very preliminary,” according to Dr Lavik.
In the femoral injury model, the nanoparticles halved bleeding time when compared to no treatment. In both the liver injury model and the blast injury model, the nanoparticles stopped bleeding and improved survival.
Dr Lavik said these experiments have shown that the nanoparticles successfully speed clotting, and the clots formed are mechanically robust.
The nanoparticles also clear the body easily. When the particles are bound into a clot, they stay as long as the clot remains. They begin to degrade once the clot does and clear out of the bloodstream quickly.
Dr Lavik said the researchers haven’t seen non-specific clotting with the nanoparticles yet. However, this side effect remains a possibility because the particles are designed to bind to activated platelets, which may not be confined to the site of injury.
One unfortunate side effect occurred with an earlier version of the nanoparticles. They triggered an immune response, activating complement in pig’s blood. However, the researchers were able to modify the particles to reduce complement activation and the accompanying complications.
In future studies, Dr Lavik and her colleagues plan to test whether the new nanoparticles activate complement in human blood. The team also aims to verify that the nanoparticles don’t cause non-specific clotting.

form clots in injured liver
Image courtesy of Erin Lavik
PHILADELPHIA—Researchers say they have developed nanoparticles that can congregate at sites of injury and speed blood clotting in vitro and in vivo.
The nanoparticles are designed to bind with activated platelets and help them join together to form clots faster.
The nanoparticles have helped stop bleeding and improved animals’ survival in different models of injury, without producing off-target clotting.
“When you have uncontrolled internal bleeding, that’s when these particles could really make a difference,” said Erin B. Lavik, ScD, of the University of Maryland, Baltimore County (UMBC) in Baltimore, Maryland.
“Compared to injuries that aren’t treated with the nanoparticles, we can cut bleeding time in half and reduce total blood loss.”
Dr Lavik discussed this work at the 252nd National Meeting & Exposition of the American Chemical Society.
She and her colleagues have been testing different versions of these nanoparticles for years. They previously reported results with the particles in 2012 and 2014.
The current nanoparticles are made from polylactic acid or polylactic co-glycolic acid. Dr Lavik said that by using the 2 different materials, the researchers can change the temperature at which the nanoparticles melt. Thus far, the team has developed nanoparticles that are stable up to 50° C (122° F).
The nanoparticles have an outer coating of polyethylene glycol and an attached peptide—RGD—that enables them to bind to natural platelets. Dr Lavik said the researchers chose RGD because platelets have receptors for that peptide, it’s ubiquitous in the body, and it wouldn’t alter the chemistry of the nanoparticles.
The researchers have tested the nanoparticles in a femoral injury model, a liver injury model, and blast injury model. The team has also started testing in models of brain and spinal cord injury, but the results are “very preliminary,” according to Dr Lavik.
In the femoral injury model, the nanoparticles halved bleeding time when compared to no treatment. In both the liver injury model and the blast injury model, the nanoparticles stopped bleeding and improved survival.
Dr Lavik said these experiments have shown that the nanoparticles successfully speed clotting, and the clots formed are mechanically robust.
The nanoparticles also clear the body easily. When the particles are bound into a clot, they stay as long as the clot remains. They begin to degrade once the clot does and clear out of the bloodstream quickly.
Dr Lavik said the researchers haven’t seen non-specific clotting with the nanoparticles yet. However, this side effect remains a possibility because the particles are designed to bind to activated platelets, which may not be confined to the site of injury.
One unfortunate side effect occurred with an earlier version of the nanoparticles. They triggered an immune response, activating complement in pig’s blood. However, the researchers were able to modify the particles to reduce complement activation and the accompanying complications.
In future studies, Dr Lavik and her colleagues plan to test whether the new nanoparticles activate complement in human blood. The team also aims to verify that the nanoparticles don’t cause non-specific clotting.

form clots in injured liver
Image courtesy of Erin Lavik
PHILADELPHIA—Researchers say they have developed nanoparticles that can congregate at sites of injury and speed blood clotting in vitro and in vivo.
The nanoparticles are designed to bind with activated platelets and help them join together to form clots faster.
The nanoparticles have helped stop bleeding and improved animals’ survival in different models of injury, without producing off-target clotting.
“When you have uncontrolled internal bleeding, that’s when these particles could really make a difference,” said Erin B. Lavik, ScD, of the University of Maryland, Baltimore County (UMBC) in Baltimore, Maryland.
“Compared to injuries that aren’t treated with the nanoparticles, we can cut bleeding time in half and reduce total blood loss.”
Dr Lavik discussed this work at the 252nd National Meeting & Exposition of the American Chemical Society.
She and her colleagues have been testing different versions of these nanoparticles for years. They previously reported results with the particles in 2012 and 2014.
The current nanoparticles are made from polylactic acid or polylactic co-glycolic acid. Dr Lavik said that by using the 2 different materials, the researchers can change the temperature at which the nanoparticles melt. Thus far, the team has developed nanoparticles that are stable up to 50° C (122° F).
The nanoparticles have an outer coating of polyethylene glycol and an attached peptide—RGD—that enables them to bind to natural platelets. Dr Lavik said the researchers chose RGD because platelets have receptors for that peptide, it’s ubiquitous in the body, and it wouldn’t alter the chemistry of the nanoparticles.
The researchers have tested the nanoparticles in a femoral injury model, a liver injury model, and blast injury model. The team has also started testing in models of brain and spinal cord injury, but the results are “very preliminary,” according to Dr Lavik.
In the femoral injury model, the nanoparticles halved bleeding time when compared to no treatment. In both the liver injury model and the blast injury model, the nanoparticles stopped bleeding and improved survival.
Dr Lavik said these experiments have shown that the nanoparticles successfully speed clotting, and the clots formed are mechanically robust.
The nanoparticles also clear the body easily. When the particles are bound into a clot, they stay as long as the clot remains. They begin to degrade once the clot does and clear out of the bloodstream quickly.
Dr Lavik said the researchers haven’t seen non-specific clotting with the nanoparticles yet. However, this side effect remains a possibility because the particles are designed to bind to activated platelets, which may not be confined to the site of injury.
One unfortunate side effect occurred with an earlier version of the nanoparticles. They triggered an immune response, activating complement in pig’s blood. However, the researchers were able to modify the particles to reduce complement activation and the accompanying complications.
In future studies, Dr Lavik and her colleagues plan to test whether the new nanoparticles activate complement in human blood. The team also aims to verify that the nanoparticles don’t cause non-specific clotting.