User login
Therapy granted fast track designation to treat GVHD

Photo by Chad McNeeley
The US Food and Drug Administration (FDA) has granted fast track designation for ProTmune™, a programmed cellular immunotherapy, to reduce the incidence and severity of acute graft-versus-host disease (GVHD) in patients undergoing allogeneic hematopoietic stem cell transplant (HSCT).
ProTmune is produced by modulating a donor-sourced, mobilized peripheral blood (mPB) graft ex vivo with 2 small molecules (FT1050 and FT4145) to enhance the biological properties and therapeutic function of the graft’s immune cells.
The programmed mPB graft is adoptively transferred and administered to a patient as a one-time intravenous infusion.
ProTmune is being developed by Fate Therapeutics, Inc.
The company is conducting a phase 1/2 trial testing ProTmune for the prevention of acute GVHD and cytomegalovirus infection in adults with hematologic malignancies who are undergoing HSCT.
The trial design consists of an initial 10-subject, phase 1 stage, during which all subjects undergoing allogeneic mPB HSCT will receive ProTmune.
Following an independent data monitoring committee safety review, a 60-subject, randomized, controlled phase 2 stage is expected to begin. In this stage, subjects undergoing allogeneic mPB HSCT will be assigned to receive either ProTmune or a conventional mPB cell graft in a 1:1 ratio.
About fast track designation
The FDA’s fast track program is designed to facilitate and expedite the development and review of new drugs intended to treat serious or life-threatening conditions and address unmet medical need.
Through the fast track program, a product may be eligible for priority review. In addition, the company developing the drug may be allowed to submit sections of the biologic license application or new drug application on a rolling basis as data become available.
Fast track designation also provides the company with opportunities for more frequent meetings with the FDA to discuss the drug’s development plan and ensure collection of the appropriate data needed to support drug approval. And the designation allows for more frequent written communication from the FDA about things such as the design of proposed clinical trials and the use of biomarkers. ![]()

Photo by Chad McNeeley
The US Food and Drug Administration (FDA) has granted fast track designation for ProTmune™, a programmed cellular immunotherapy, to reduce the incidence and severity of acute graft-versus-host disease (GVHD) in patients undergoing allogeneic hematopoietic stem cell transplant (HSCT).
ProTmune is produced by modulating a donor-sourced, mobilized peripheral blood (mPB) graft ex vivo with 2 small molecules (FT1050 and FT4145) to enhance the biological properties and therapeutic function of the graft’s immune cells.
The programmed mPB graft is adoptively transferred and administered to a patient as a one-time intravenous infusion.
ProTmune is being developed by Fate Therapeutics, Inc.
The company is conducting a phase 1/2 trial testing ProTmune for the prevention of acute GVHD and cytomegalovirus infection in adults with hematologic malignancies who are undergoing HSCT.
The trial design consists of an initial 10-subject, phase 1 stage, during which all subjects undergoing allogeneic mPB HSCT will receive ProTmune.
Following an independent data monitoring committee safety review, a 60-subject, randomized, controlled phase 2 stage is expected to begin. In this stage, subjects undergoing allogeneic mPB HSCT will be assigned to receive either ProTmune or a conventional mPB cell graft in a 1:1 ratio.
About fast track designation
The FDA’s fast track program is designed to facilitate and expedite the development and review of new drugs intended to treat serious or life-threatening conditions and address unmet medical need.
Through the fast track program, a product may be eligible for priority review. In addition, the company developing the drug may be allowed to submit sections of the biologic license application or new drug application on a rolling basis as data become available.
Fast track designation also provides the company with opportunities for more frequent meetings with the FDA to discuss the drug’s development plan and ensure collection of the appropriate data needed to support drug approval. And the designation allows for more frequent written communication from the FDA about things such as the design of proposed clinical trials and the use of biomarkers. ![]()

Photo by Chad McNeeley
The US Food and Drug Administration (FDA) has granted fast track designation for ProTmune™, a programmed cellular immunotherapy, to reduce the incidence and severity of acute graft-versus-host disease (GVHD) in patients undergoing allogeneic hematopoietic stem cell transplant (HSCT).
ProTmune is produced by modulating a donor-sourced, mobilized peripheral blood (mPB) graft ex vivo with 2 small molecules (FT1050 and FT4145) to enhance the biological properties and therapeutic function of the graft’s immune cells.
The programmed mPB graft is adoptively transferred and administered to a patient as a one-time intravenous infusion.
ProTmune is being developed by Fate Therapeutics, Inc.
The company is conducting a phase 1/2 trial testing ProTmune for the prevention of acute GVHD and cytomegalovirus infection in adults with hematologic malignancies who are undergoing HSCT.
The trial design consists of an initial 10-subject, phase 1 stage, during which all subjects undergoing allogeneic mPB HSCT will receive ProTmune.
Following an independent data monitoring committee safety review, a 60-subject, randomized, controlled phase 2 stage is expected to begin. In this stage, subjects undergoing allogeneic mPB HSCT will be assigned to receive either ProTmune or a conventional mPB cell graft in a 1:1 ratio.
About fast track designation
The FDA’s fast track program is designed to facilitate and expedite the development and review of new drugs intended to treat serious or life-threatening conditions and address unmet medical need.
Through the fast track program, a product may be eligible for priority review. In addition, the company developing the drug may be allowed to submit sections of the biologic license application or new drug application on a rolling basis as data become available.
Fast track designation also provides the company with opportunities for more frequent meetings with the FDA to discuss the drug’s development plan and ensure collection of the appropriate data needed to support drug approval. And the designation allows for more frequent written communication from the FDA about things such as the design of proposed clinical trials and the use of biomarkers. ![]()
A new approach to treat AML?

Preclinical research suggests that activating the STING pathway may be a feasible approach for treating acute myeloid leukemia (AML).
The STING protein has been shown to play a crucial role in the immune system’s ability to “sense” cancer by recognizing and responding to DNA from tumor cells.
In past studies, researchers injected compounds that activate the STING pathway directly into solid tumors in mice, and this produced potent anti-tumor immune responses.
In a new study, researchers injected substances that mimic tumor-cell DNA into the bloodstream and found they could stimulate STING to provoke a life-extending immune response in mice with AML.
“Delivery of these substances into the blood led to massive immune responses,” said study author Justin Kline, MD, of the University of Chicago in Illinois.
“I’ve worked extensively with animal models of this disease, and have never seen responses like this.”
This research, published in Cell Reports, is the first demonstration that activating the STING pathway could be effective in hematologic malignancies.
STING (short for STimulator of INterferon Genes) plays a role in detecting threats, such as viral infections or cancer. STING is activated when DNA turns up in the wrong place, inside the cell but outside the nucleus.
When it encounters such misplaced DNA, STING induces the production of interferon-beta and other chemical signals that recruit certain components of the immune system to manage the threat, such as leukemia-specific killer T cells.
In this study, the researchers found that mice with established AML were rarely able to launch an effective immune response against the disease.
But when the team exposed the mice to DMXAA (5,6-dimethylxanthenone-4-acetic acid), a molecule that activates STING, the immune system responded aggressively, culminating in the activation of highly potent, cancer-cell killing T cells.
This response prolonged survival and, in some cases, cured the mice of their leukemia. About 60% of DMXAA-treated mice survived long-term. They were even able to protect themselves when “re-challenged” with AML cells.
Because of significant differences between mice and humans, DMXAA does not activate the human STING pathway, but researchers have found that several cyclic dinucleotides—signaling molecules produced by bacteria—have a comparable effect in stimulating the STING pathway.
This leads to an immune response that begins with the production of type I interferons and proceeds to later, more powerful stages, ultimately including leukemia-specific T cells.
“Our results provide strong rationale for the clinical translation of STING agonists as immune therapy for leukemia and possibly other hematologic
malignancies,” said study author Emily Curran, MD, of the University of Chicago.
However, Dr Kline noted that this approach is “not without risk.” He said it can induce “a lot of inflammation, fever, even shock.” Such a stimulated immune system can be “too effective,” especially when the therapy is given through the blood stream, rather than injected into a solid tumor.
“I think drug makers will want to focus on intra-tumoral injection studies before they are ready to bet on systemic infusion,” Dr Kline said. “But this is an important first step.” ![]()

Preclinical research suggests that activating the STING pathway may be a feasible approach for treating acute myeloid leukemia (AML).
The STING protein has been shown to play a crucial role in the immune system’s ability to “sense” cancer by recognizing and responding to DNA from tumor cells.
In past studies, researchers injected compounds that activate the STING pathway directly into solid tumors in mice, and this produced potent anti-tumor immune responses.
In a new study, researchers injected substances that mimic tumor-cell DNA into the bloodstream and found they could stimulate STING to provoke a life-extending immune response in mice with AML.
“Delivery of these substances into the blood led to massive immune responses,” said study author Justin Kline, MD, of the University of Chicago in Illinois.
“I’ve worked extensively with animal models of this disease, and have never seen responses like this.”
This research, published in Cell Reports, is the first demonstration that activating the STING pathway could be effective in hematologic malignancies.
STING (short for STimulator of INterferon Genes) plays a role in detecting threats, such as viral infections or cancer. STING is activated when DNA turns up in the wrong place, inside the cell but outside the nucleus.
When it encounters such misplaced DNA, STING induces the production of interferon-beta and other chemical signals that recruit certain components of the immune system to manage the threat, such as leukemia-specific killer T cells.
In this study, the researchers found that mice with established AML were rarely able to launch an effective immune response against the disease.
But when the team exposed the mice to DMXAA (5,6-dimethylxanthenone-4-acetic acid), a molecule that activates STING, the immune system responded aggressively, culminating in the activation of highly potent, cancer-cell killing T cells.
This response prolonged survival and, in some cases, cured the mice of their leukemia. About 60% of DMXAA-treated mice survived long-term. They were even able to protect themselves when “re-challenged” with AML cells.
Because of significant differences between mice and humans, DMXAA does not activate the human STING pathway, but researchers have found that several cyclic dinucleotides—signaling molecules produced by bacteria—have a comparable effect in stimulating the STING pathway.
This leads to an immune response that begins with the production of type I interferons and proceeds to later, more powerful stages, ultimately including leukemia-specific T cells.
“Our results provide strong rationale for the clinical translation of STING agonists as immune therapy for leukemia and possibly other hematologic
malignancies,” said study author Emily Curran, MD, of the University of Chicago.
However, Dr Kline noted that this approach is “not without risk.” He said it can induce “a lot of inflammation, fever, even shock.” Such a stimulated immune system can be “too effective,” especially when the therapy is given through the blood stream, rather than injected into a solid tumor.
“I think drug makers will want to focus on intra-tumoral injection studies before they are ready to bet on systemic infusion,” Dr Kline said. “But this is an important first step.” ![]()

Preclinical research suggests that activating the STING pathway may be a feasible approach for treating acute myeloid leukemia (AML).
The STING protein has been shown to play a crucial role in the immune system’s ability to “sense” cancer by recognizing and responding to DNA from tumor cells.
In past studies, researchers injected compounds that activate the STING pathway directly into solid tumors in mice, and this produced potent anti-tumor immune responses.
In a new study, researchers injected substances that mimic tumor-cell DNA into the bloodstream and found they could stimulate STING to provoke a life-extending immune response in mice with AML.
“Delivery of these substances into the blood led to massive immune responses,” said study author Justin Kline, MD, of the University of Chicago in Illinois.
“I’ve worked extensively with animal models of this disease, and have never seen responses like this.”
This research, published in Cell Reports, is the first demonstration that activating the STING pathway could be effective in hematologic malignancies.
STING (short for STimulator of INterferon Genes) plays a role in detecting threats, such as viral infections or cancer. STING is activated when DNA turns up in the wrong place, inside the cell but outside the nucleus.
When it encounters such misplaced DNA, STING induces the production of interferon-beta and other chemical signals that recruit certain components of the immune system to manage the threat, such as leukemia-specific killer T cells.
In this study, the researchers found that mice with established AML were rarely able to launch an effective immune response against the disease.
But when the team exposed the mice to DMXAA (5,6-dimethylxanthenone-4-acetic acid), a molecule that activates STING, the immune system responded aggressively, culminating in the activation of highly potent, cancer-cell killing T cells.
This response prolonged survival and, in some cases, cured the mice of their leukemia. About 60% of DMXAA-treated mice survived long-term. They were even able to protect themselves when “re-challenged” with AML cells.
Because of significant differences between mice and humans, DMXAA does not activate the human STING pathway, but researchers have found that several cyclic dinucleotides—signaling molecules produced by bacteria—have a comparable effect in stimulating the STING pathway.
This leads to an immune response that begins with the production of type I interferons and proceeds to later, more powerful stages, ultimately including leukemia-specific T cells.
“Our results provide strong rationale for the clinical translation of STING agonists as immune therapy for leukemia and possibly other hematologic
malignancies,” said study author Emily Curran, MD, of the University of Chicago.
However, Dr Kline noted that this approach is “not without risk.” He said it can induce “a lot of inflammation, fever, even shock.” Such a stimulated immune system can be “too effective,” especially when the therapy is given through the blood stream, rather than injected into a solid tumor.
“I think drug makers will want to focus on intra-tumoral injection studies before they are ready to bet on systemic infusion,” Dr Kline said. “But this is an important first step.” ![]()
FDA approves use of assay to screen blood for Zika virus

Photo courtesy of UAB Hospital
The US Food and Drug Administration (FDA) has approved the use of a new assay to screen donated blood for the Zika virus.
The Procleix Zika Virus Assay is approved for use under an investigational new drug study protocol.
Blood banks can use the test to screen donated blood for the Zika virus in potentially endemic areas of the southern US. Testing may be extended to other areas of the US if the Zika virus continues to spread.
The Procleix Zika Virus Assay, which was co-developed by Hologic, Inc. and Grifols, is designed to run on the Procleix Panther System, an automated, nucleic acid technology blood screening platform. The system has received regulatory approvals in countries around the world and is currently in development for the US market.
“The American Red Cross is pleased to participate in the Procleix Zika Virus Assay investigational study, which will allow us to begin blood donor testing for Zika virus early this summer in areas most likely to have local mosquito transmission of the virus,” said Susan Stramer, PhD, vice-president of scientific affairs at the American Red Cross.
The FDA previously authorized use of another test, the cobas® Zika test (developed by Roche), to screen blood donations for Zika virus. The cobas Zika test can be used under an investigational new drug study protocol for screening donated blood in areas with active, mosquito-borne transmission of the Zika virus. ![]()

Photo courtesy of UAB Hospital
The US Food and Drug Administration (FDA) has approved the use of a new assay to screen donated blood for the Zika virus.
The Procleix Zika Virus Assay is approved for use under an investigational new drug study protocol.
Blood banks can use the test to screen donated blood for the Zika virus in potentially endemic areas of the southern US. Testing may be extended to other areas of the US if the Zika virus continues to spread.
The Procleix Zika Virus Assay, which was co-developed by Hologic, Inc. and Grifols, is designed to run on the Procleix Panther System, an automated, nucleic acid technology blood screening platform. The system has received regulatory approvals in countries around the world and is currently in development for the US market.
“The American Red Cross is pleased to participate in the Procleix Zika Virus Assay investigational study, which will allow us to begin blood donor testing for Zika virus early this summer in areas most likely to have local mosquito transmission of the virus,” said Susan Stramer, PhD, vice-president of scientific affairs at the American Red Cross.
The FDA previously authorized use of another test, the cobas® Zika test (developed by Roche), to screen blood donations for Zika virus. The cobas Zika test can be used under an investigational new drug study protocol for screening donated blood in areas with active, mosquito-borne transmission of the Zika virus. ![]()

Photo courtesy of UAB Hospital
The US Food and Drug Administration (FDA) has approved the use of a new assay to screen donated blood for the Zika virus.
The Procleix Zika Virus Assay is approved for use under an investigational new drug study protocol.
Blood banks can use the test to screen donated blood for the Zika virus in potentially endemic areas of the southern US. Testing may be extended to other areas of the US if the Zika virus continues to spread.
The Procleix Zika Virus Assay, which was co-developed by Hologic, Inc. and Grifols, is designed to run on the Procleix Panther System, an automated, nucleic acid technology blood screening platform. The system has received regulatory approvals in countries around the world and is currently in development for the US market.
“The American Red Cross is pleased to participate in the Procleix Zika Virus Assay investigational study, which will allow us to begin blood donor testing for Zika virus early this summer in areas most likely to have local mosquito transmission of the virus,” said Susan Stramer, PhD, vice-president of scientific affairs at the American Red Cross.
The FDA previously authorized use of another test, the cobas® Zika test (developed by Roche), to screen blood donations for Zika virus. The cobas Zika test can be used under an investigational new drug study protocol for screening donated blood in areas with active, mosquito-borne transmission of the Zika virus. ![]()
Study provides clues to AML survival after chemotherapy

Preclinical research suggests that some leukemia cells harvest energy resources from normal cells during chemotherapy, and this helps the leukemia cells survive and thrive after treatment.
Investigators found that acute myeloid leukemia (AML) cells are capable of stealing mitochondria from stromal cells, and these stolen mitochondria give an energy boost to surviving AML cells, which helps fuel the leukemia’s resurgence after chemotherapy.
“There are multiple mechanisms for resistance to chemotherapy, and it will be important to target them all in order to eliminate all leukemic cells,” said Jean-François Peyron, PhD, of the Centre Méditerranéen de Médecine Moléculaire (C3M) in Nice, France.
“Targeting this protective mitochondrial transfer could represent a new strategy to improve the efficacy of the current treatments for acute myeloid leukemia.”
Dr Peyron and his colleagues described their discovery of the mitochondrial transfer in Blood.
The team conducted their experiments using cell cultures and mouse models of AML. They found that nearly all AML cells died when exposed to chemotherapy drugs, but some survived. And these cells issued a “mayday” signal that “tricked” nearby non-cancerous cells into yielding their mitochondria to the AML cells, thus strengthening the leukemia cells.
“Mitochondria produce the energy that is vital for cell functions,” explained study author Emmanuel Griessinger, PhD, also of C3M.
“Through the uptake of mitochondria, chemotherapy-injured acute myeloid leukemia cells recover new energy to survive.”
The AML cells were found to increase their mitochondria mass by an average of 14%. This increase led to a 1.5-fold increase in energy production and significantly better survival rates. That is, the leukemia cells that had a high level of mitochondria were also more resistant to the chemotherapy.
The investigators observed the phenomenon in several types of leukemia cells, most notably leukemia-initiating cells. The team said this finding may explain why it can be difficult to treat AML and other cancers.
They also believe these findings offer new hope for developing better treatments for AML. If researchers can find a way to interfere with the transfer of mitochondria, that could reduce the risk of relapse.
The study may also shed light on other cancer types. Similar mechanisms may be at play in other hematologic malignancies and even solid tumors, according to the investigators.
For now, the team’s next step for this research is to identify the mechanism underlying the transfer of mitochondria in AML. ![]()

Preclinical research suggests that some leukemia cells harvest energy resources from normal cells during chemotherapy, and this helps the leukemia cells survive and thrive after treatment.
Investigators found that acute myeloid leukemia (AML) cells are capable of stealing mitochondria from stromal cells, and these stolen mitochondria give an energy boost to surviving AML cells, which helps fuel the leukemia’s resurgence after chemotherapy.
“There are multiple mechanisms for resistance to chemotherapy, and it will be important to target them all in order to eliminate all leukemic cells,” said Jean-François Peyron, PhD, of the Centre Méditerranéen de Médecine Moléculaire (C3M) in Nice, France.
“Targeting this protective mitochondrial transfer could represent a new strategy to improve the efficacy of the current treatments for acute myeloid leukemia.”
Dr Peyron and his colleagues described their discovery of the mitochondrial transfer in Blood.
The team conducted their experiments using cell cultures and mouse models of AML. They found that nearly all AML cells died when exposed to chemotherapy drugs, but some survived. And these cells issued a “mayday” signal that “tricked” nearby non-cancerous cells into yielding their mitochondria to the AML cells, thus strengthening the leukemia cells.
“Mitochondria produce the energy that is vital for cell functions,” explained study author Emmanuel Griessinger, PhD, also of C3M.
“Through the uptake of mitochondria, chemotherapy-injured acute myeloid leukemia cells recover new energy to survive.”
The AML cells were found to increase their mitochondria mass by an average of 14%. This increase led to a 1.5-fold increase in energy production and significantly better survival rates. That is, the leukemia cells that had a high level of mitochondria were also more resistant to the chemotherapy.
The investigators observed the phenomenon in several types of leukemia cells, most notably leukemia-initiating cells. The team said this finding may explain why it can be difficult to treat AML and other cancers.
They also believe these findings offer new hope for developing better treatments for AML. If researchers can find a way to interfere with the transfer of mitochondria, that could reduce the risk of relapse.
The study may also shed light on other cancer types. Similar mechanisms may be at play in other hematologic malignancies and even solid tumors, according to the investigators.
For now, the team’s next step for this research is to identify the mechanism underlying the transfer of mitochondria in AML. ![]()

Preclinical research suggests that some leukemia cells harvest energy resources from normal cells during chemotherapy, and this helps the leukemia cells survive and thrive after treatment.
Investigators found that acute myeloid leukemia (AML) cells are capable of stealing mitochondria from stromal cells, and these stolen mitochondria give an energy boost to surviving AML cells, which helps fuel the leukemia’s resurgence after chemotherapy.
“There are multiple mechanisms for resistance to chemotherapy, and it will be important to target them all in order to eliminate all leukemic cells,” said Jean-François Peyron, PhD, of the Centre Méditerranéen de Médecine Moléculaire (C3M) in Nice, France.
“Targeting this protective mitochondrial transfer could represent a new strategy to improve the efficacy of the current treatments for acute myeloid leukemia.”
Dr Peyron and his colleagues described their discovery of the mitochondrial transfer in Blood.
The team conducted their experiments using cell cultures and mouse models of AML. They found that nearly all AML cells died when exposed to chemotherapy drugs, but some survived. And these cells issued a “mayday” signal that “tricked” nearby non-cancerous cells into yielding their mitochondria to the AML cells, thus strengthening the leukemia cells.
“Mitochondria produce the energy that is vital for cell functions,” explained study author Emmanuel Griessinger, PhD, also of C3M.
“Through the uptake of mitochondria, chemotherapy-injured acute myeloid leukemia cells recover new energy to survive.”
The AML cells were found to increase their mitochondria mass by an average of 14%. This increase led to a 1.5-fold increase in energy production and significantly better survival rates. That is, the leukemia cells that had a high level of mitochondria were also more resistant to the chemotherapy.
The investigators observed the phenomenon in several types of leukemia cells, most notably leukemia-initiating cells. The team said this finding may explain why it can be difficult to treat AML and other cancers.
They also believe these findings offer new hope for developing better treatments for AML. If researchers can find a way to interfere with the transfer of mitochondria, that could reduce the risk of relapse.
The study may also shed light on other cancer types. Similar mechanisms may be at play in other hematologic malignancies and even solid tumors, according to the investigators.
For now, the team’s next step for this research is to identify the mechanism underlying the transfer of mitochondria in AML. ![]()
Long-term opioid use may not benefit SCD patients

and a normal one
Image by Betty Pace
Results of a small study suggest that long-term opioid treatment may not be the best option for pain management in adults with sickle cell disease (SCD).
The study showed that patients who received opioids long-term often fared worse in measures of pain, fatigue, and curtailed daily activities than patients who were not on long-term opioids.
Researchers reported these findings in the American Journal of Preventive Medicine.
“We need to be careful and skeptical about giving increasing doses of opioids to patients with sickle cell disease who are in chronic pain if it isn’t effective,” said study author C. Patrick Carroll, MD, of the Johns Hopkins University School of Medicine in Baltimore, Maryland.
“Too little is known about the effects of long-term opioid management of chronic pain.”
For this study, Dr Carroll and his colleagues enrolled 83 SCD patients—57 women and 26 men. All were 18 and older, and their average age was 39.
Twenty-nine of the patients had been prescribed daily, long-acting opioids to manage their pain. The remaining 54 patients weren’t on long-term opioids.
The patients filled out daily electronic pain diaries for 90 days. Self-reported levels of pain, physical activity, fatigue, and pain-related daily activity interference were recorded, along with self-reported levels of pain relief and medication satisfaction on a scale of 0 to 100.
Crises, pain, and fatigue
The proportion of days in vaso-occlusive crisis was significantly higher for patients who were on long-term opioid therapy than for those who were not—29% and 11.9%, respectively (P<0.01).
Patients on long-term opioids also reported significantly higher levels of crisis pain—60.6 and 41.0, respectively (P<0.001)—and non-crisis pain—34.5 and 10.3, respectively (P<0.001).
Patients on long-term opioid therapy had higher levels of pain-related activity interference, both on non-crisis days—24.9 and 7.4, respectively (P<0.001)—and crisis days—56.7 and 37.7, respectively (P<0.01).
And patients on long-term opioid therapy had higher levels of fatigue on non-crisis days—49.7 and 27.0, respectively (P<0.001)—and crisis days—66.1 and 53.0, respectively (P<0.05).
Central sensitization
The researchers also performed some standard measures of pain processing on the test subjects, which measured and averaged variables such as how intensely participants experienced unpleasant heat and pressure.
The team was particularly interested in the phenomenon of central sensitization, in which the central nervous system amplifies painful sensations.
Central sensitization may be one way that opioids can increase pain sensitivity, and it also may play a role in how SCD causes chronic pain, Dr Carroll said.
For example, one such measure of central sensitization uses repeated pokes from a mildly painful stimulus in succession. In people who have this hypersensitization, each poke is perceived as more intense than the last because the nervous system becomes progressively more sensitive to the pain.
Combining the data from several measures of central sensitization, the researchers used a scoring system that sets a normal measurement at 0 and rates how abnormal something is by how far the values move away from 0. They calculated a central sensitization index for patients on long-term opioids and those not taking them.
Overall, patients on long-term opioid therapy showed higher levels of central sensitization, with an index of 0.34, than those who were not on opioids, with an index of -0.10.
In patients who were not on long-term opioid therapy, the level of central sensitization correlated with the level of non-crisis pain. However, in patients who were taking long-term opioid therapy and also had higher levels of central sensitization and clinical pain, the correlation essentially vanished.
Dr Carroll said this was surprising and suggests the mechanisms of pain in SCD patients on long-term opioid therapy may be different from patients who don’t take daily opioids for pain.
Dr Carroll cautioned that this work is preliminary and should not lead to the discontinuation of long-term opioid therapy in SCD patients.
“We need to better understand how long-term opioid use affects pain sensitization and determine if certain people are more sensitive to these effects so we can prescribe the best treatment option for each individual patient,” Dr Carroll said. “We also need to learn more about how sickle cell disease may sensitize the nervous system.” ![]()

and a normal one
Image by Betty Pace
Results of a small study suggest that long-term opioid treatment may not be the best option for pain management in adults with sickle cell disease (SCD).
The study showed that patients who received opioids long-term often fared worse in measures of pain, fatigue, and curtailed daily activities than patients who were not on long-term opioids.
Researchers reported these findings in the American Journal of Preventive Medicine.
“We need to be careful and skeptical about giving increasing doses of opioids to patients with sickle cell disease who are in chronic pain if it isn’t effective,” said study author C. Patrick Carroll, MD, of the Johns Hopkins University School of Medicine in Baltimore, Maryland.
“Too little is known about the effects of long-term opioid management of chronic pain.”
For this study, Dr Carroll and his colleagues enrolled 83 SCD patients—57 women and 26 men. All were 18 and older, and their average age was 39.
Twenty-nine of the patients had been prescribed daily, long-acting opioids to manage their pain. The remaining 54 patients weren’t on long-term opioids.
The patients filled out daily electronic pain diaries for 90 days. Self-reported levels of pain, physical activity, fatigue, and pain-related daily activity interference were recorded, along with self-reported levels of pain relief and medication satisfaction on a scale of 0 to 100.
Crises, pain, and fatigue
The proportion of days in vaso-occlusive crisis was significantly higher for patients who were on long-term opioid therapy than for those who were not—29% and 11.9%, respectively (P<0.01).
Patients on long-term opioids also reported significantly higher levels of crisis pain—60.6 and 41.0, respectively (P<0.001)—and non-crisis pain—34.5 and 10.3, respectively (P<0.001).
Patients on long-term opioid therapy had higher levels of pain-related activity interference, both on non-crisis days—24.9 and 7.4, respectively (P<0.001)—and crisis days—56.7 and 37.7, respectively (P<0.01).
And patients on long-term opioid therapy had higher levels of fatigue on non-crisis days—49.7 and 27.0, respectively (P<0.001)—and crisis days—66.1 and 53.0, respectively (P<0.05).
Central sensitization
The researchers also performed some standard measures of pain processing on the test subjects, which measured and averaged variables such as how intensely participants experienced unpleasant heat and pressure.
The team was particularly interested in the phenomenon of central sensitization, in which the central nervous system amplifies painful sensations.
Central sensitization may be one way that opioids can increase pain sensitivity, and it also may play a role in how SCD causes chronic pain, Dr Carroll said.
For example, one such measure of central sensitization uses repeated pokes from a mildly painful stimulus in succession. In people who have this hypersensitization, each poke is perceived as more intense than the last because the nervous system becomes progressively more sensitive to the pain.
Combining the data from several measures of central sensitization, the researchers used a scoring system that sets a normal measurement at 0 and rates how abnormal something is by how far the values move away from 0. They calculated a central sensitization index for patients on long-term opioids and those not taking them.
Overall, patients on long-term opioid therapy showed higher levels of central sensitization, with an index of 0.34, than those who were not on opioids, with an index of -0.10.
In patients who were not on long-term opioid therapy, the level of central sensitization correlated with the level of non-crisis pain. However, in patients who were taking long-term opioid therapy and also had higher levels of central sensitization and clinical pain, the correlation essentially vanished.
Dr Carroll said this was surprising and suggests the mechanisms of pain in SCD patients on long-term opioid therapy may be different from patients who don’t take daily opioids for pain.
Dr Carroll cautioned that this work is preliminary and should not lead to the discontinuation of long-term opioid therapy in SCD patients.
“We need to better understand how long-term opioid use affects pain sensitization and determine if certain people are more sensitive to these effects so we can prescribe the best treatment option for each individual patient,” Dr Carroll said. “We also need to learn more about how sickle cell disease may sensitize the nervous system.” ![]()

and a normal one
Image by Betty Pace
Results of a small study suggest that long-term opioid treatment may not be the best option for pain management in adults with sickle cell disease (SCD).
The study showed that patients who received opioids long-term often fared worse in measures of pain, fatigue, and curtailed daily activities than patients who were not on long-term opioids.
Researchers reported these findings in the American Journal of Preventive Medicine.
“We need to be careful and skeptical about giving increasing doses of opioids to patients with sickle cell disease who are in chronic pain if it isn’t effective,” said study author C. Patrick Carroll, MD, of the Johns Hopkins University School of Medicine in Baltimore, Maryland.
“Too little is known about the effects of long-term opioid management of chronic pain.”
For this study, Dr Carroll and his colleagues enrolled 83 SCD patients—57 women and 26 men. All were 18 and older, and their average age was 39.
Twenty-nine of the patients had been prescribed daily, long-acting opioids to manage their pain. The remaining 54 patients weren’t on long-term opioids.
The patients filled out daily electronic pain diaries for 90 days. Self-reported levels of pain, physical activity, fatigue, and pain-related daily activity interference were recorded, along with self-reported levels of pain relief and medication satisfaction on a scale of 0 to 100.
Crises, pain, and fatigue
The proportion of days in vaso-occlusive crisis was significantly higher for patients who were on long-term opioid therapy than for those who were not—29% and 11.9%, respectively (P<0.01).
Patients on long-term opioids also reported significantly higher levels of crisis pain—60.6 and 41.0, respectively (P<0.001)—and non-crisis pain—34.5 and 10.3, respectively (P<0.001).
Patients on long-term opioid therapy had higher levels of pain-related activity interference, both on non-crisis days—24.9 and 7.4, respectively (P<0.001)—and crisis days—56.7 and 37.7, respectively (P<0.01).
And patients on long-term opioid therapy had higher levels of fatigue on non-crisis days—49.7 and 27.0, respectively (P<0.001)—and crisis days—66.1 and 53.0, respectively (P<0.05).
Central sensitization
The researchers also performed some standard measures of pain processing on the test subjects, which measured and averaged variables such as how intensely participants experienced unpleasant heat and pressure.
The team was particularly interested in the phenomenon of central sensitization, in which the central nervous system amplifies painful sensations.
Central sensitization may be one way that opioids can increase pain sensitivity, and it also may play a role in how SCD causes chronic pain, Dr Carroll said.
For example, one such measure of central sensitization uses repeated pokes from a mildly painful stimulus in succession. In people who have this hypersensitization, each poke is perceived as more intense than the last because the nervous system becomes progressively more sensitive to the pain.
Combining the data from several measures of central sensitization, the researchers used a scoring system that sets a normal measurement at 0 and rates how abnormal something is by how far the values move away from 0. They calculated a central sensitization index for patients on long-term opioids and those not taking them.
Overall, patients on long-term opioid therapy showed higher levels of central sensitization, with an index of 0.34, than those who were not on opioids, with an index of -0.10.
In patients who were not on long-term opioid therapy, the level of central sensitization correlated with the level of non-crisis pain. However, in patients who were taking long-term opioid therapy and also had higher levels of central sensitization and clinical pain, the correlation essentially vanished.
Dr Carroll said this was surprising and suggests the mechanisms of pain in SCD patients on long-term opioid therapy may be different from patients who don’t take daily opioids for pain.
Dr Carroll cautioned that this work is preliminary and should not lead to the discontinuation of long-term opioid therapy in SCD patients.
“We need to better understand how long-term opioid use affects pain sensitization and determine if certain people are more sensitive to these effects so we can prescribe the best treatment option for each individual patient,” Dr Carroll said. “We also need to learn more about how sickle cell disease may sensitize the nervous system.” ![]()
Health Canada shortens deferral for MSM blood donors
Photo by Marja Helander
Health Canada has decided to change its policy regarding blood donations from men who have sex with men (MSM).
The policy has been that MSMs can only donate blood if they have abstained from sexual contact with another man for a period of 5 years. As of August 15, that period will be shortened to 1 year.
The change is a result of proposals from Canada’s blood operators, Canadian Blood Services and Héma-Québec.
These proposals included scientific data indicating that decreasing the deferral period for MSMs would not affect the safety of the blood supply.
The change brings Canada in line with several other countries that have implemented a 1-year deferral period for MSM blood donors, including the US, Australia, New Zealand, England, Scotland, and France.
Health Canada said the country’s blood system continues to have rigorous scientific and screening processes in place to protect the safety of Canadians.
Canadian Blood Services and Héma-Québec will continue to screen all donations for HIV and other infections. As an extra precaution, Health Canada and the blood operators will monitor donations from new donors to see if there is an increase in HIV or other infection rates.
According to Health Canada, there has not been a single HIV infection from blood transfusion in Canada in 25 years.
However, the frequency of HIV infection remains higher among MSMs (10%) than among heterosexuals or lesbians (less than 1% for both). And it was because of this that Canada implemented the 5-year deferral period for MSM blood donors. Prior to the implementation of that policy, MSMs were completely barred from donating blood in Canada.
“It has been demonstrated that implementing a 5-year temporary exclusion in 2013 had no impact on the safety of the transfusion system,” said Marc Germain, vice-president of medical affairs at Héma-Québec.
“As a result of recent data concerning transfusion safety, the exclusion policy applied to men who have had sex with another man could be reviewed. [The new 1-year deferral period] is scientifically justified and will not endanger the very high level of safety of blood products.”
“This is an exciting, incremental step forward in updating our blood donation criteria based on the latest scientific evidence,” said Graham Sher, chief executive officer of Canadian Blood Services.
“Canadian Blood Services is dedicated to being as minimally restrictive as possible while also maintaining the safety of the blood supply.”
Canadian Blood Services said it is exploring the possibility of moving toward behavior-based screening of blood donors. The organization is working with researchers, the LGBTQ community, patient groups, and other stakeholders to determine how best to gather the scientific evidence required to determine future changes to donor eligibility criteria. ![]()

Photo by Marja Helander
Health Canada has decided to change its policy regarding blood donations from men who have sex with men (MSM).
The policy has been that MSMs can only donate blood if they have abstained from sexual contact with another man for a period of 5 years. As of August 15, that period will be shortened to 1 year.
The change is a result of proposals from Canada’s blood operators, Canadian Blood Services and Héma-Québec.
These proposals included scientific data indicating that decreasing the deferral period for MSMs would not affect the safety of the blood supply.
The change brings Canada in line with several other countries that have implemented a 1-year deferral period for MSM blood donors, including the US, Australia, New Zealand, England, Scotland, and France.
Health Canada said the country’s blood system continues to have rigorous scientific and screening processes in place to protect the safety of Canadians.
Canadian Blood Services and Héma-Québec will continue to screen all donations for HIV and other infections. As an extra precaution, Health Canada and the blood operators will monitor donations from new donors to see if there is an increase in HIV or other infection rates.
According to Health Canada, there has not been a single HIV infection from blood transfusion in Canada in 25 years.
However, the frequency of HIV infection remains higher among MSMs (10%) than among heterosexuals or lesbians (less than 1% for both). And it was because of this that Canada implemented the 5-year deferral period for MSM blood donors. Prior to the implementation of that policy, MSMs were completely barred from donating blood in Canada.
“It has been demonstrated that implementing a 5-year temporary exclusion in 2013 had no impact on the safety of the transfusion system,” said Marc Germain, vice-president of medical affairs at Héma-Québec.
“As a result of recent data concerning transfusion safety, the exclusion policy applied to men who have had sex with another man could be reviewed. [The new 1-year deferral period] is scientifically justified and will not endanger the very high level of safety of blood products.”
“This is an exciting, incremental step forward in updating our blood donation criteria based on the latest scientific evidence,” said Graham Sher, chief executive officer of Canadian Blood Services.
“Canadian Blood Services is dedicated to being as minimally restrictive as possible while also maintaining the safety of the blood supply.”
Canadian Blood Services said it is exploring the possibility of moving toward behavior-based screening of blood donors. The organization is working with researchers, the LGBTQ community, patient groups, and other stakeholders to determine how best to gather the scientific evidence required to determine future changes to donor eligibility criteria. ![]()

Photo by Marja Helander
Health Canada has decided to change its policy regarding blood donations from men who have sex with men (MSM).
The policy has been that MSMs can only donate blood if they have abstained from sexual contact with another man for a period of 5 years. As of August 15, that period will be shortened to 1 year.
The change is a result of proposals from Canada’s blood operators, Canadian Blood Services and Héma-Québec.
These proposals included scientific data indicating that decreasing the deferral period for MSMs would not affect the safety of the blood supply.
The change brings Canada in line with several other countries that have implemented a 1-year deferral period for MSM blood donors, including the US, Australia, New Zealand, England, Scotland, and France.
Health Canada said the country’s blood system continues to have rigorous scientific and screening processes in place to protect the safety of Canadians.
Canadian Blood Services and Héma-Québec will continue to screen all donations for HIV and other infections. As an extra precaution, Health Canada and the blood operators will monitor donations from new donors to see if there is an increase in HIV or other infection rates.
According to Health Canada, there has not been a single HIV infection from blood transfusion in Canada in 25 years.
However, the frequency of HIV infection remains higher among MSMs (10%) than among heterosexuals or lesbians (less than 1% for both). And it was because of this that Canada implemented the 5-year deferral period for MSM blood donors. Prior to the implementation of that policy, MSMs were completely barred from donating blood in Canada.
“It has been demonstrated that implementing a 5-year temporary exclusion in 2013 had no impact on the safety of the transfusion system,” said Marc Germain, vice-president of medical affairs at Héma-Québec.
“As a result of recent data concerning transfusion safety, the exclusion policy applied to men who have had sex with another man could be reviewed. [The new 1-year deferral period] is scientifically justified and will not endanger the very high level of safety of blood products.”
“This is an exciting, incremental step forward in updating our blood donation criteria based on the latest scientific evidence,” said Graham Sher, chief executive officer of Canadian Blood Services.
“Canadian Blood Services is dedicated to being as minimally restrictive as possible while also maintaining the safety of the blood supply.”
Canadian Blood Services said it is exploring the possibility of moving toward behavior-based screening of blood donors. The organization is working with researchers, the LGBTQ community, patient groups, and other stakeholders to determine how best to gather the scientific evidence required to determine future changes to donor eligibility criteria. ![]()
How CML cells respond to stress

Image by Difu Wu
Researchers have used a tiny force probe to compare how healthy hematopoietic cells and cancerous ones respond to stress.
They found that cells harvested from the bone marrow of 5 patients with chronic myeloid leukemia (CML) appeared much stiffer than comparable samples taken from 5 healthy volunteers.
In addition, the researchers were able to identify areas of localized brittle failure events in the CML cells.
“What makes this work so exciting to us is not simply seeing a difference between the stiffness of healthy and cancerous cells but observing that the cancerous cells also lost their dynamic ductility and behaved as more breakable objects,” said study author Françoise Argoul, PhD, of the French National Centre for Research (CNRS) in Lyon, France.
Dr Argoul and her colleagues described this work in Physical Biology.
The researchers believe the mechanical signatures obtained by squeezing or deforming cells could potentially assist physicians in determining the presence of CML and other hematologic malignancies.
The mechanical data might also provide clues as to how long the cells have been affected by the cancer.
“We would like to construct a hematopoietic cancer cell chart where the loss of cell mechanical functions could be graded, depending on the leukemia and its stage of evolution,” Dr Argoul said.
Thinking about how the technique might be applied in a hospital setting, she added that biopsy needles could, in principle, be adapted to allow local sensing of internal soft tissue structures.
However, before the researchers can even progress to testing cells inside the body and preparing for clinical trials, they must first build up sufficient information from their measurements on isolated cells under a range of conditions in the lab. ![]()

Image by Difu Wu
Researchers have used a tiny force probe to compare how healthy hematopoietic cells and cancerous ones respond to stress.
They found that cells harvested from the bone marrow of 5 patients with chronic myeloid leukemia (CML) appeared much stiffer than comparable samples taken from 5 healthy volunteers.
In addition, the researchers were able to identify areas of localized brittle failure events in the CML cells.
“What makes this work so exciting to us is not simply seeing a difference between the stiffness of healthy and cancerous cells but observing that the cancerous cells also lost their dynamic ductility and behaved as more breakable objects,” said study author Françoise Argoul, PhD, of the French National Centre for Research (CNRS) in Lyon, France.
Dr Argoul and her colleagues described this work in Physical Biology.
The researchers believe the mechanical signatures obtained by squeezing or deforming cells could potentially assist physicians in determining the presence of CML and other hematologic malignancies.
The mechanical data might also provide clues as to how long the cells have been affected by the cancer.
“We would like to construct a hematopoietic cancer cell chart where the loss of cell mechanical functions could be graded, depending on the leukemia and its stage of evolution,” Dr Argoul said.
Thinking about how the technique might be applied in a hospital setting, she added that biopsy needles could, in principle, be adapted to allow local sensing of internal soft tissue structures.
However, before the researchers can even progress to testing cells inside the body and preparing for clinical trials, they must first build up sufficient information from their measurements on isolated cells under a range of conditions in the lab. ![]()

Image by Difu Wu
Researchers have used a tiny force probe to compare how healthy hematopoietic cells and cancerous ones respond to stress.
They found that cells harvested from the bone marrow of 5 patients with chronic myeloid leukemia (CML) appeared much stiffer than comparable samples taken from 5 healthy volunteers.
In addition, the researchers were able to identify areas of localized brittle failure events in the CML cells.
“What makes this work so exciting to us is not simply seeing a difference between the stiffness of healthy and cancerous cells but observing that the cancerous cells also lost their dynamic ductility and behaved as more breakable objects,” said study author Françoise Argoul, PhD, of the French National Centre for Research (CNRS) in Lyon, France.
Dr Argoul and her colleagues described this work in Physical Biology.
The researchers believe the mechanical signatures obtained by squeezing or deforming cells could potentially assist physicians in determining the presence of CML and other hematologic malignancies.
The mechanical data might also provide clues as to how long the cells have been affected by the cancer.
“We would like to construct a hematopoietic cancer cell chart where the loss of cell mechanical functions could be graded, depending on the leukemia and its stage of evolution,” Dr Argoul said.
Thinking about how the technique might be applied in a hospital setting, she added that biopsy needles could, in principle, be adapted to allow local sensing of internal soft tissue structures.
However, before the researchers can even progress to testing cells inside the body and preparing for clinical trials, they must first build up sufficient information from their measurements on isolated cells under a range of conditions in the lab.
Protein may be biomarker of cGVHD
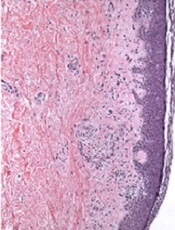
Image courtesy of PLOS ONE
A study published in Blood has revealed a protein that could be used to diagnose chronic graft-versus-host disease (cGVHD).
Investigators found elevated levels of the protein, CXCL10, in the blood of transplant recipients around the time they developed cGVHD.
The team therefore believes that testing a transplant patient for this protein could potentially enable early diagnosis of cGVHD.
“Diagnostic tests are desperately needed to make blood and marrow transplants safer,” said study author Kirk Schultz, of BC Children’s Hospital in Vancouver, British Columbia, Canada.
“At this time, there are no good tests to diagnose cGVHD, and the disease can only be identified too late—when it is already established. If we can diagnose it earlier and better, then treatments can be used to stop it before it becomes a chronic, disabling disease.”
For this study, Dr Schultz and his colleagues searched for cGVHD biomarkers in blood samples from 2 groups of adult patients—36 cGVHD patients who were at least 1 month from diagnosis and 31 time-matched control subjects without cGVHD.
This revealed 11 potential biomarkers. The investigators went on to test the validity of these biomarkers in 2 replication cohorts, which included a total of 134 patients with cGVHD and 154 control subjects.
Results showed that the inflammatory protein CXCL10 was consistently elevated in patients with cGVHD. This protein appears to impact a patient’s normal immune cells, preventing the body from fighting cGVHD.
The investigators also found evidence to suggest that another protein, sBAFF, is a biomarker of cGVHD.
Although these results may bring us one step closer to a diagnostic test for cGVHD, the team said further study is needed in larger patient groups.

Image courtesy of PLOS ONE
A study published in Blood has revealed a protein that could be used to diagnose chronic graft-versus-host disease (cGVHD).
Investigators found elevated levels of the protein, CXCL10, in the blood of transplant recipients around the time they developed cGVHD.
The team therefore believes that testing a transplant patient for this protein could potentially enable early diagnosis of cGVHD.
“Diagnostic tests are desperately needed to make blood and marrow transplants safer,” said study author Kirk Schultz, of BC Children’s Hospital in Vancouver, British Columbia, Canada.
“At this time, there are no good tests to diagnose cGVHD, and the disease can only be identified too late—when it is already established. If we can diagnose it earlier and better, then treatments can be used to stop it before it becomes a chronic, disabling disease.”
For this study, Dr Schultz and his colleagues searched for cGVHD biomarkers in blood samples from 2 groups of adult patients—36 cGVHD patients who were at least 1 month from diagnosis and 31 time-matched control subjects without cGVHD.
This revealed 11 potential biomarkers. The investigators went on to test the validity of these biomarkers in 2 replication cohorts, which included a total of 134 patients with cGVHD and 154 control subjects.
Results showed that the inflammatory protein CXCL10 was consistently elevated in patients with cGVHD. This protein appears to impact a patient’s normal immune cells, preventing the body from fighting cGVHD.
The investigators also found evidence to suggest that another protein, sBAFF, is a biomarker of cGVHD.
Although these results may bring us one step closer to a diagnostic test for cGVHD, the team said further study is needed in larger patient groups.

Image courtesy of PLOS ONE
A study published in Blood has revealed a protein that could be used to diagnose chronic graft-versus-host disease (cGVHD).
Investigators found elevated levels of the protein, CXCL10, in the blood of transplant recipients around the time they developed cGVHD.
The team therefore believes that testing a transplant patient for this protein could potentially enable early diagnosis of cGVHD.
“Diagnostic tests are desperately needed to make blood and marrow transplants safer,” said study author Kirk Schultz, of BC Children’s Hospital in Vancouver, British Columbia, Canada.
“At this time, there are no good tests to diagnose cGVHD, and the disease can only be identified too late—when it is already established. If we can diagnose it earlier and better, then treatments can be used to stop it before it becomes a chronic, disabling disease.”
For this study, Dr Schultz and his colleagues searched for cGVHD biomarkers in blood samples from 2 groups of adult patients—36 cGVHD patients who were at least 1 month from diagnosis and 31 time-matched control subjects without cGVHD.
This revealed 11 potential biomarkers. The investigators went on to test the validity of these biomarkers in 2 replication cohorts, which included a total of 134 patients with cGVHD and 154 control subjects.
Results showed that the inflammatory protein CXCL10 was consistently elevated in patients with cGVHD. This protein appears to impact a patient’s normal immune cells, preventing the body from fighting cGVHD.
The investigators also found evidence to suggest that another protein, sBAFF, is a biomarker of cGVHD.
Although these results may bring us one step closer to a diagnostic test for cGVHD, the team said further study is needed in larger patient groups.
Scientists describe mechanism of IMiDs

Researchers say they have identified the molecular mechanism of thalidomide and other immunomodulatory drugs (IMiDs), a finding that is relevant to the treatment of multiple myeloma and other hematologic malignancies.
Previous research showed that cereblon, a cellular protein, plays an important role in the function of IMiDs.
However, the exact details of how cereblon mediates the effects of IMiDs were not clear.
The new study, published in Nature Medicine, provides an explanation.
Researchers found that, inside cells, cereblon usually binds to the proteins CD147 and MCT1. Among other things, these 2 proteins promote proliferation, metabolism, and the formation of new blood vessels. In cancers such as multiple myeloma, tumor cells contain particularly high levels of CD147 and MCT1.
As a protein complex, CD147 and MCT1 always occur as a pair. However, to find their other half and become activated, the proteins require the help of cereblon.
Binding to cereblon promotes development and stability of the complex, which, in return, stimulates cell growth and facilitates the excretion of metabolic products like lactate.
In diseases such as multiple myeloma, an increased abundance of this protein complex enables tumor cells to multiply and spread rapidly.
If such a cancer is treated with IMiDs, the drug virtually displaces the complex from its binding to cereblon. As a result, CD147 and MCT1 can no longer be activated, and they vanish.
“Ultimately, this causes the tumor cells to die,” said study author Ruth Eichner, MD, of Technische Universität München in Munich, Germany.
But the disruption of the protein complex is also responsible for the severe birth defects that can occur in the children of women who take thalidomide and other IMiDs when pregnant.
“The mechanisms are identical,” said study author Florian Bassermann, MD, PhD, of Technische Universität München.
“A specific inactivation of the protein complex resulted in the same developmental defects observed after thalidomide treatment.”
The researchers said this confirms the prevailing hypothesis that the typical IMiD-induced birth defects are related to the reduced and abnormal formation of new blood vessels. That’s because, without CD147 and MCT1, blood vessels cannot develop properly.
The team also said these findings could be used to assess the efficacy of IMiD treatment before actually giving an IMiD to the patient.
“The disappearance of the protein complex could only be observed in patients that had responded well to this type of treatment,” Dr Bassermann explained.
Therefore, he and his colleagues believe this information could be used to assess a patient’s response before starting an IMiD. A sample of the patient’s tumor cells could be cultured and treated with IMiDs. If the cells showed a disruption of the complex, the patient would most likely benefit from IMiD treatment.
The researchers also think the results of this study could lead to the development of new anticancer therapies.
The team said the CD147-MCT1 protein complex is a particularly attractive target for tumor treatment, as it is mainly found on the surface of cells and virtually links the inside to the outside of the cell.
Therefore, inactivation of the complex might be achieved using specifically produced antibodies and other distinctive drugs—a possibility Dr Bassermann and his team are now exploring.

Researchers say they have identified the molecular mechanism of thalidomide and other immunomodulatory drugs (IMiDs), a finding that is relevant to the treatment of multiple myeloma and other hematologic malignancies.
Previous research showed that cereblon, a cellular protein, plays an important role in the function of IMiDs.
However, the exact details of how cereblon mediates the effects of IMiDs were not clear.
The new study, published in Nature Medicine, provides an explanation.
Researchers found that, inside cells, cereblon usually binds to the proteins CD147 and MCT1. Among other things, these 2 proteins promote proliferation, metabolism, and the formation of new blood vessels. In cancers such as multiple myeloma, tumor cells contain particularly high levels of CD147 and MCT1.
As a protein complex, CD147 and MCT1 always occur as a pair. However, to find their other half and become activated, the proteins require the help of cereblon.
Binding to cereblon promotes development and stability of the complex, which, in return, stimulates cell growth and facilitates the excretion of metabolic products like lactate.
In diseases such as multiple myeloma, an increased abundance of this protein complex enables tumor cells to multiply and spread rapidly.
If such a cancer is treated with IMiDs, the drug virtually displaces the complex from its binding to cereblon. As a result, CD147 and MCT1 can no longer be activated, and they vanish.
“Ultimately, this causes the tumor cells to die,” said study author Ruth Eichner, MD, of Technische Universität München in Munich, Germany.
But the disruption of the protein complex is also responsible for the severe birth defects that can occur in the children of women who take thalidomide and other IMiDs when pregnant.
“The mechanisms are identical,” said study author Florian Bassermann, MD, PhD, of Technische Universität München.
“A specific inactivation of the protein complex resulted in the same developmental defects observed after thalidomide treatment.”
The researchers said this confirms the prevailing hypothesis that the typical IMiD-induced birth defects are related to the reduced and abnormal formation of new blood vessels. That’s because, without CD147 and MCT1, blood vessels cannot develop properly.
The team also said these findings could be used to assess the efficacy of IMiD treatment before actually giving an IMiD to the patient.
“The disappearance of the protein complex could only be observed in patients that had responded well to this type of treatment,” Dr Bassermann explained.
Therefore, he and his colleagues believe this information could be used to assess a patient’s response before starting an IMiD. A sample of the patient’s tumor cells could be cultured and treated with IMiDs. If the cells showed a disruption of the complex, the patient would most likely benefit from IMiD treatment.
The researchers also think the results of this study could lead to the development of new anticancer therapies.
The team said the CD147-MCT1 protein complex is a particularly attractive target for tumor treatment, as it is mainly found on the surface of cells and virtually links the inside to the outside of the cell.
Therefore, inactivation of the complex might be achieved using specifically produced antibodies and other distinctive drugs—a possibility Dr Bassermann and his team are now exploring.

Researchers say they have identified the molecular mechanism of thalidomide and other immunomodulatory drugs (IMiDs), a finding that is relevant to the treatment of multiple myeloma and other hematologic malignancies.
Previous research showed that cereblon, a cellular protein, plays an important role in the function of IMiDs.
However, the exact details of how cereblon mediates the effects of IMiDs were not clear.
The new study, published in Nature Medicine, provides an explanation.
Researchers found that, inside cells, cereblon usually binds to the proteins CD147 and MCT1. Among other things, these 2 proteins promote proliferation, metabolism, and the formation of new blood vessels. In cancers such as multiple myeloma, tumor cells contain particularly high levels of CD147 and MCT1.
As a protein complex, CD147 and MCT1 always occur as a pair. However, to find their other half and become activated, the proteins require the help of cereblon.
Binding to cereblon promotes development and stability of the complex, which, in return, stimulates cell growth and facilitates the excretion of metabolic products like lactate.
In diseases such as multiple myeloma, an increased abundance of this protein complex enables tumor cells to multiply and spread rapidly.
If such a cancer is treated with IMiDs, the drug virtually displaces the complex from its binding to cereblon. As a result, CD147 and MCT1 can no longer be activated, and they vanish.
“Ultimately, this causes the tumor cells to die,” said study author Ruth Eichner, MD, of Technische Universität München in Munich, Germany.
But the disruption of the protein complex is also responsible for the severe birth defects that can occur in the children of women who take thalidomide and other IMiDs when pregnant.
“The mechanisms are identical,” said study author Florian Bassermann, MD, PhD, of Technische Universität München.
“A specific inactivation of the protein complex resulted in the same developmental defects observed after thalidomide treatment.”
The researchers said this confirms the prevailing hypothesis that the typical IMiD-induced birth defects are related to the reduced and abnormal formation of new blood vessels. That’s because, without CD147 and MCT1, blood vessels cannot develop properly.
The team also said these findings could be used to assess the efficacy of IMiD treatment before actually giving an IMiD to the patient.
“The disappearance of the protein complex could only be observed in patients that had responded well to this type of treatment,” Dr Bassermann explained.
Therefore, he and his colleagues believe this information could be used to assess a patient’s response before starting an IMiD. A sample of the patient’s tumor cells could be cultured and treated with IMiDs. If the cells showed a disruption of the complex, the patient would most likely benefit from IMiD treatment.
The researchers also think the results of this study could lead to the development of new anticancer therapies.
The team said the CD147-MCT1 protein complex is a particularly attractive target for tumor treatment, as it is mainly found on the surface of cells and virtually links the inside to the outside of the cell.
Therefore, inactivation of the complex might be achieved using specifically produced antibodies and other distinctive drugs—a possibility Dr Bassermann and his team are now exploring.
FDA authorizes use of new Zika assay
The US Food and Drug Administration (FDA) has granted emergency use authorization (EUA) for a new assay designed to detect Zika virus infection.
The Aptima® Zika Virus assay is a molecular diagnostic tool that can be used to detect RNA from the Zika virus in human serum and plasma specimens.
The assay has not been FDA-cleared or approved. An EUA allows for the use of unapproved medical products or unapproved uses of approved medical products in an emergency.
The products must be used to diagnose, treat, or prevent serious or life-threatening conditions caused by chemical, biological, radiological, or nuclear threat agents, when there are no adequate alternatives.
This means the Aptima Zika Virus assay is only authorized as long as circumstances exist to justify the authorization of the emergency use of in vitro diagnostics for the detection of Zika virus under section 564(b)(1) of the Federal Food, Drug & Cosmetic Act, 21 U.S.C.§360bbb-3(b)(1), unless the authorization is terminated or revoked sooner.
About the assay
The Aptima Zika Virus assay was developed by Hologic, Inc. The assay runs on the Hologic Panther® system, an integrated platform that fully automates all aspects of nucleic acid amplification testing.
The Aptima Zika Virus assay will be available for use in all 50 states, Puerto Rico, and US territories.
The assay is designed to be used in individuals meeting Centers for Disease Control and Prevention (CDC) Zika virus clinical criteria (eg, clinical signs and symptoms associated with Zika virus infection) and/or CDC Zika virus epidemiological criteria (eg, history of residence in or travel to a geographic region with active Zika transmission at the time of travel, or other epidemiologic criteria for which Zika virus testing may be indicated), by laboratories in the US that are certified under the Clinical Laboratory Improvement Amendments of 1988, 42 U.S.C. § 263a, to perform high complexity tests, or by similarly qualified non-US laboratories.
For more information about the Aptima Zika Virus assay, visit www.hologic.com/zika.
About Zika testing
The FDA has granted EUAs for 4 other tests designed to detect Zika virus:
- RealStar® Zika Virus RT-PCR Kit U.S. (altona Diagnostics GmbH)
- Zika Virus RNA Qualitative Real-Time RT-PCR test (Focus Diagnostics)
- Trioplex Real-time RT-PCR Assay (CDC)
- Zika IgM Antibody Capture Enzyme-Linked Immunosorbent Assay (Zika MAC-ELISA, CDC).
The FDA has also authorized use of the cobas® Zika test (Roche) to screen blood donations for Zika virus. The test may be used under an investigational new drug application for screening donated blood in areas with active mosquito-borne transmission of the virus.
The US Food and Drug Administration (FDA) has granted emergency use authorization (EUA) for a new assay designed to detect Zika virus infection.
The Aptima® Zika Virus assay is a molecular diagnostic tool that can be used to detect RNA from the Zika virus in human serum and plasma specimens.
The assay has not been FDA-cleared or approved. An EUA allows for the use of unapproved medical products or unapproved uses of approved medical products in an emergency.
The products must be used to diagnose, treat, or prevent serious or life-threatening conditions caused by chemical, biological, radiological, or nuclear threat agents, when there are no adequate alternatives.
This means the Aptima Zika Virus assay is only authorized as long as circumstances exist to justify the authorization of the emergency use of in vitro diagnostics for the detection of Zika virus under section 564(b)(1) of the Federal Food, Drug & Cosmetic Act, 21 U.S.C.§360bbb-3(b)(1), unless the authorization is terminated or revoked sooner.
About the assay
The Aptima Zika Virus assay was developed by Hologic, Inc. The assay runs on the Hologic Panther® system, an integrated platform that fully automates all aspects of nucleic acid amplification testing.
The Aptima Zika Virus assay will be available for use in all 50 states, Puerto Rico, and US territories.
The assay is designed to be used in individuals meeting Centers for Disease Control and Prevention (CDC) Zika virus clinical criteria (eg, clinical signs and symptoms associated with Zika virus infection) and/or CDC Zika virus epidemiological criteria (eg, history of residence in or travel to a geographic region with active Zika transmission at the time of travel, or other epidemiologic criteria for which Zika virus testing may be indicated), by laboratories in the US that are certified under the Clinical Laboratory Improvement Amendments of 1988, 42 U.S.C. § 263a, to perform high complexity tests, or by similarly qualified non-US laboratories.
For more information about the Aptima Zika Virus assay, visit www.hologic.com/zika.
About Zika testing
The FDA has granted EUAs for 4 other tests designed to detect Zika virus:
- RealStar® Zika Virus RT-PCR Kit U.S. (altona Diagnostics GmbH)
- Zika Virus RNA Qualitative Real-Time RT-PCR test (Focus Diagnostics)
- Trioplex Real-time RT-PCR Assay (CDC)
- Zika IgM Antibody Capture Enzyme-Linked Immunosorbent Assay (Zika MAC-ELISA, CDC).
The FDA has also authorized use of the cobas® Zika test (Roche) to screen blood donations for Zika virus. The test may be used under an investigational new drug application for screening donated blood in areas with active mosquito-borne transmission of the virus.
The US Food and Drug Administration (FDA) has granted emergency use authorization (EUA) for a new assay designed to detect Zika virus infection.
The Aptima® Zika Virus assay is a molecular diagnostic tool that can be used to detect RNA from the Zika virus in human serum and plasma specimens.
The assay has not been FDA-cleared or approved. An EUA allows for the use of unapproved medical products or unapproved uses of approved medical products in an emergency.
The products must be used to diagnose, treat, or prevent serious or life-threatening conditions caused by chemical, biological, radiological, or nuclear threat agents, when there are no adequate alternatives.
This means the Aptima Zika Virus assay is only authorized as long as circumstances exist to justify the authorization of the emergency use of in vitro diagnostics for the detection of Zika virus under section 564(b)(1) of the Federal Food, Drug & Cosmetic Act, 21 U.S.C.§360bbb-3(b)(1), unless the authorization is terminated or revoked sooner.
About the assay
The Aptima Zika Virus assay was developed by Hologic, Inc. The assay runs on the Hologic Panther® system, an integrated platform that fully automates all aspects of nucleic acid amplification testing.
The Aptima Zika Virus assay will be available for use in all 50 states, Puerto Rico, and US territories.
The assay is designed to be used in individuals meeting Centers for Disease Control and Prevention (CDC) Zika virus clinical criteria (eg, clinical signs and symptoms associated with Zika virus infection) and/or CDC Zika virus epidemiological criteria (eg, history of residence in or travel to a geographic region with active Zika transmission at the time of travel, or other epidemiologic criteria for which Zika virus testing may be indicated), by laboratories in the US that are certified under the Clinical Laboratory Improvement Amendments of 1988, 42 U.S.C. § 263a, to perform high complexity tests, or by similarly qualified non-US laboratories.
For more information about the Aptima Zika Virus assay, visit www.hologic.com/zika.
About Zika testing
The FDA has granted EUAs for 4 other tests designed to detect Zika virus:
- RealStar® Zika Virus RT-PCR Kit U.S. (altona Diagnostics GmbH)
- Zika Virus RNA Qualitative Real-Time RT-PCR test (Focus Diagnostics)
- Trioplex Real-time RT-PCR Assay (CDC)
- Zika IgM Antibody Capture Enzyme-Linked Immunosorbent Assay (Zika MAC-ELISA, CDC).
The FDA has also authorized use of the cobas® Zika test (Roche) to screen blood donations for Zika virus. The test may be used under an investigational new drug application for screening donated blood in areas with active mosquito-borne transmission of the virus.