User login
Study reveals delays in emergency blood transfusions

Photo courtesy of UAB Hospital
A new study suggests that as few as 2% of patients with life-threatening bleeding after serious injury receive optimal blood transfusion therapy in England and Wales.
Researchers estimate that nearly 5000 trauma patients sustain a major hemorrhage in England and Wales each year, and one-third of those patients die.
The current study, published in the British Journal of Surgery, highlights how delays in transfusions may contribute to this death rate.
“The rapid and consistent delivery of blood, plasma, platelets, and other clotting products to trauma patients is essential to maintain clotting during hemorrhage and has been shown to halve mortality,” said study author Karim Brohi, MBBS, of Queen Mary University of London in the UK.
“However, we found that only 2% of patients with massive hemorrhage received the optimal type of blood transfusion for their resuscitation. There is a clear opportunity for clinicians to improve the delivery of blood and clotting products during resuscitation for major hemorrhage.”
Dr Brohi and his colleagues analyzed 442 patients treated at 22 hospitals in England and Wales. The patients had experienced major hemorrhage as a result of injuries and received at least 4 units of packed red blood cells (PRBCs) in the first 24 hours of admission.
The patients’ median age was 38 (range, 24-54), and 74% were men. Thirty-three percent of patients (n=146) had massive hemorrhage.
Mortality from bleeding tended to occur early in these patients. Twenty-seven percent of patients (n=117) died in hospital—18% (n=79) within the first 24 hours. The 30-day mortality rate was about 27% (n=119), and 33% of evaluable patients had died at 1 year (127/383).
All 442 patients received PRBCs. The median number of PRBC units transfused within 24 hours was 7 (range, 5-11), and the median number of PRBC units given in 30 days was 9 (range, 6-15).
The average time to transfusion of PRBCs was longer than expected, at 41 minutes (range, 1-122).
Similarly, the researchers found the administration of blood components such as plasma and platelets to be significantly delayed, occurring, on average, 2 to 3 hours after admission.
Three-quarters of patients (n=330) received fresh-frozen plasma (FFP). The median number of FFP units given within 24 hours was 4 (range, 0-7), and the time to first FFP transfusion was 87 minutes (range, 42.5-229).
About 45% of patients (n=197) received platelets. The median dose was 0 (range, 0-1), and the time to first platelet transfusion was 146 minutes (range, 72.5-364).
About 28% of patients (n=122) received cryoprecipitate. The median dose was 0 (range, 0-1), and the time to first cryoprecipitate infusion was 179.5 minutes (range, 84.5-333.5).
“The rapid delivery of the right mix of blood components in an emergency environment is extremely challenging,” Dr Brohi said.
“Some transfusion components have to be thawed and, at present, aren’t always available for the patient quickly enough. More research is also needed into techniques and devices to control bleeding earlier, even at the scene of injury.”
The researchers noted that this study had its limitations, such as incomplete data for some patients. ![]()

Photo courtesy of UAB Hospital
A new study suggests that as few as 2% of patients with life-threatening bleeding after serious injury receive optimal blood transfusion therapy in England and Wales.
Researchers estimate that nearly 5000 trauma patients sustain a major hemorrhage in England and Wales each year, and one-third of those patients die.
The current study, published in the British Journal of Surgery, highlights how delays in transfusions may contribute to this death rate.
“The rapid and consistent delivery of blood, plasma, platelets, and other clotting products to trauma patients is essential to maintain clotting during hemorrhage and has been shown to halve mortality,” said study author Karim Brohi, MBBS, of Queen Mary University of London in the UK.
“However, we found that only 2% of patients with massive hemorrhage received the optimal type of blood transfusion for their resuscitation. There is a clear opportunity for clinicians to improve the delivery of blood and clotting products during resuscitation for major hemorrhage.”
Dr Brohi and his colleagues analyzed 442 patients treated at 22 hospitals in England and Wales. The patients had experienced major hemorrhage as a result of injuries and received at least 4 units of packed red blood cells (PRBCs) in the first 24 hours of admission.
The patients’ median age was 38 (range, 24-54), and 74% were men. Thirty-three percent of patients (n=146) had massive hemorrhage.
Mortality from bleeding tended to occur early in these patients. Twenty-seven percent of patients (n=117) died in hospital—18% (n=79) within the first 24 hours. The 30-day mortality rate was about 27% (n=119), and 33% of evaluable patients had died at 1 year (127/383).
All 442 patients received PRBCs. The median number of PRBC units transfused within 24 hours was 7 (range, 5-11), and the median number of PRBC units given in 30 days was 9 (range, 6-15).
The average time to transfusion of PRBCs was longer than expected, at 41 minutes (range, 1-122).
Similarly, the researchers found the administration of blood components such as plasma and platelets to be significantly delayed, occurring, on average, 2 to 3 hours after admission.
Three-quarters of patients (n=330) received fresh-frozen plasma (FFP). The median number of FFP units given within 24 hours was 4 (range, 0-7), and the time to first FFP transfusion was 87 minutes (range, 42.5-229).
About 45% of patients (n=197) received platelets. The median dose was 0 (range, 0-1), and the time to first platelet transfusion was 146 minutes (range, 72.5-364).
About 28% of patients (n=122) received cryoprecipitate. The median dose was 0 (range, 0-1), and the time to first cryoprecipitate infusion was 179.5 minutes (range, 84.5-333.5).
“The rapid delivery of the right mix of blood components in an emergency environment is extremely challenging,” Dr Brohi said.
“Some transfusion components have to be thawed and, at present, aren’t always available for the patient quickly enough. More research is also needed into techniques and devices to control bleeding earlier, even at the scene of injury.”
The researchers noted that this study had its limitations, such as incomplete data for some patients. ![]()

Photo courtesy of UAB Hospital
A new study suggests that as few as 2% of patients with life-threatening bleeding after serious injury receive optimal blood transfusion therapy in England and Wales.
Researchers estimate that nearly 5000 trauma patients sustain a major hemorrhage in England and Wales each year, and one-third of those patients die.
The current study, published in the British Journal of Surgery, highlights how delays in transfusions may contribute to this death rate.
“The rapid and consistent delivery of blood, plasma, platelets, and other clotting products to trauma patients is essential to maintain clotting during hemorrhage and has been shown to halve mortality,” said study author Karim Brohi, MBBS, of Queen Mary University of London in the UK.
“However, we found that only 2% of patients with massive hemorrhage received the optimal type of blood transfusion for their resuscitation. There is a clear opportunity for clinicians to improve the delivery of blood and clotting products during resuscitation for major hemorrhage.”
Dr Brohi and his colleagues analyzed 442 patients treated at 22 hospitals in England and Wales. The patients had experienced major hemorrhage as a result of injuries and received at least 4 units of packed red blood cells (PRBCs) in the first 24 hours of admission.
The patients’ median age was 38 (range, 24-54), and 74% were men. Thirty-three percent of patients (n=146) had massive hemorrhage.
Mortality from bleeding tended to occur early in these patients. Twenty-seven percent of patients (n=117) died in hospital—18% (n=79) within the first 24 hours. The 30-day mortality rate was about 27% (n=119), and 33% of evaluable patients had died at 1 year (127/383).
All 442 patients received PRBCs. The median number of PRBC units transfused within 24 hours was 7 (range, 5-11), and the median number of PRBC units given in 30 days was 9 (range, 6-15).
The average time to transfusion of PRBCs was longer than expected, at 41 minutes (range, 1-122).
Similarly, the researchers found the administration of blood components such as plasma and platelets to be significantly delayed, occurring, on average, 2 to 3 hours after admission.
Three-quarters of patients (n=330) received fresh-frozen plasma (FFP). The median number of FFP units given within 24 hours was 4 (range, 0-7), and the time to first FFP transfusion was 87 minutes (range, 42.5-229).
About 45% of patients (n=197) received platelets. The median dose was 0 (range, 0-1), and the time to first platelet transfusion was 146 minutes (range, 72.5-364).
About 28% of patients (n=122) received cryoprecipitate. The median dose was 0 (range, 0-1), and the time to first cryoprecipitate infusion was 179.5 minutes (range, 84.5-333.5).
“The rapid delivery of the right mix of blood components in an emergency environment is extremely challenging,” Dr Brohi said.
“Some transfusion components have to be thawed and, at present, aren’t always available for the patient quickly enough. More research is also needed into techniques and devices to control bleeding earlier, even at the scene of injury.”
The researchers noted that this study had its limitations, such as incomplete data for some patients. ![]()
MF drug trials placed on partial clinical hold
The US Food and Drug Administration (FDA) has placed a partial clinical hold on trials conducted under the investigational new drug
(IND) application for pacritinib.
Pacritinib is a JAK2/FLT3 inhibitor being developed by CTI BioPharma for the treatment of myelofibrosis (MF).
The partial clinical hold impacts part of the clinical work currently being conducted under the pacritinib IND and will also affect planned clinical trials.
The FDA said the reasons for the partial clinical hold are excess mortality and other adverse events in pacritinib-treated patients (compared to the control arm) in the PERSIST-1 trial.
The excess mortality was most evident during the non-randomized crossover period following the initial 24 weeks of randomized treatment, during which patients in the control arm could switch to pacritinib treatment.
Under the partial clinical hold, investigators may not enroll new patients or start pacritinib as initial or crossover treatment, and patients not deriving benefit after 30 weeks of pacritinib treatment must stop using pacritinib.
In addition, the FDA has recommended that CTI BioPharma make certain modifications to protocols, provide certain notifications, revise relevant statements in the related investigator’s brochure and informed consent documents, and take certain other actions.
CTI BioPharma said it intends to implement the FDA’s recommendations, and all clinical investigators worldwide have been notified of the partial clinical hold.
Just before the FDA notified CTI BioPharma of the partial clinical hold, the company completed enrollment in the phase 3 PERSIST-2 trial.
In PERSIST-2, researchers are comparing the efficacy and safety of pacritinib and best available therapy in patients with thrombocytopenia and primary MF, post-polycythemia vera MF, or post-essential thrombocythemia MF.
Under the partial clinical hold, patients on this trial who are currently receiving pacritinib may continue to do so unless they are not deriving benefit after 30 weeks of pacritinib treatment, and crossover of patients from the control arm to the pacritinib arm will not be allowed. ![]()

The US Food and Drug Administration (FDA) has placed a partial clinical hold on trials conducted under the investigational new drug
(IND) application for pacritinib.
Pacritinib is a JAK2/FLT3 inhibitor being developed by CTI BioPharma for the treatment of myelofibrosis (MF).
The partial clinical hold impacts part of the clinical work currently being conducted under the pacritinib IND and will also affect planned clinical trials.
The FDA said the reasons for the partial clinical hold are excess mortality and other adverse events in pacritinib-treated patients (compared to the control arm) in the PERSIST-1 trial.
The excess mortality was most evident during the non-randomized crossover period following the initial 24 weeks of randomized treatment, during which patients in the control arm could switch to pacritinib treatment.
Under the partial clinical hold, investigators may not enroll new patients or start pacritinib as initial or crossover treatment, and patients not deriving benefit after 30 weeks of pacritinib treatment must stop using pacritinib.
In addition, the FDA has recommended that CTI BioPharma make certain modifications to protocols, provide certain notifications, revise relevant statements in the related investigator’s brochure and informed consent documents, and take certain other actions.
CTI BioPharma said it intends to implement the FDA’s recommendations, and all clinical investigators worldwide have been notified of the partial clinical hold.
Just before the FDA notified CTI BioPharma of the partial clinical hold, the company completed enrollment in the phase 3 PERSIST-2 trial.
In PERSIST-2, researchers are comparing the efficacy and safety of pacritinib and best available therapy in patients with thrombocytopenia and primary MF, post-polycythemia vera MF, or post-essential thrombocythemia MF.
Under the partial clinical hold, patients on this trial who are currently receiving pacritinib may continue to do so unless they are not deriving benefit after 30 weeks of pacritinib treatment, and crossover of patients from the control arm to the pacritinib arm will not be allowed. ![]()

The US Food and Drug Administration (FDA) has placed a partial clinical hold on trials conducted under the investigational new drug
(IND) application for pacritinib.
Pacritinib is a JAK2/FLT3 inhibitor being developed by CTI BioPharma for the treatment of myelofibrosis (MF).
The partial clinical hold impacts part of the clinical work currently being conducted under the pacritinib IND and will also affect planned clinical trials.
The FDA said the reasons for the partial clinical hold are excess mortality and other adverse events in pacritinib-treated patients (compared to the control arm) in the PERSIST-1 trial.
The excess mortality was most evident during the non-randomized crossover period following the initial 24 weeks of randomized treatment, during which patients in the control arm could switch to pacritinib treatment.
Under the partial clinical hold, investigators may not enroll new patients or start pacritinib as initial or crossover treatment, and patients not deriving benefit after 30 weeks of pacritinib treatment must stop using pacritinib.
In addition, the FDA has recommended that CTI BioPharma make certain modifications to protocols, provide certain notifications, revise relevant statements in the related investigator’s brochure and informed consent documents, and take certain other actions.
CTI BioPharma said it intends to implement the FDA’s recommendations, and all clinical investigators worldwide have been notified of the partial clinical hold.
Just before the FDA notified CTI BioPharma of the partial clinical hold, the company completed enrollment in the phase 3 PERSIST-2 trial.
In PERSIST-2, researchers are comparing the efficacy and safety of pacritinib and best available therapy in patients with thrombocytopenia and primary MF, post-polycythemia vera MF, or post-essential thrombocythemia MF.
Under the partial clinical hold, patients on this trial who are currently receiving pacritinib may continue to do so unless they are not deriving benefit after 30 weeks of pacritinib treatment, and crossover of patients from the control arm to the pacritinib arm will not be allowed. ![]()
Tandem HSCT regimen may cure MM

Photo by Chad McNeeley
Tandem autologous/allogeneic hematopoietic stem cell transplant (HSCT) may cure multiple myeloma (MM), according to a phase 2 study.
The study included newly diagnosed patients who received induction therapy followed by an autograft and a non-myeloablative allograft from a matched sibling donor.
The patients have been followed for a median of 8.8 years.
The probability of progression-free survival at 10 years is 41%, and the overall survival is 62%.
The rate of chronic graft-vs-host disease (GVHD) is high, but the rate of non-relapse mortality is low.
“In many hospitals, doctors have abandoned the use of allografts for multiple myeloma due to the risk of toxicity and relapse,” said study author Jean Roy, MD, of the Maisonneuve-Rosemont Hospital and University of Montreal in Quebec, Canada.
“Our results, on the other hand, have led us to offer the treatment to more patients, especially younger patients and those with poorer prognoses.”
Dr Roy and his colleagues reported these results in Bone Marrow Transplantation.
The researchers assessed 92 patients newly diagnosed with MM between 2001 and 2010. Their median age was 52 (range, 39-64), and 97% had Durie–Salmon stage II or III disease.
Patients received an induction regimen consisting of vincristine, doxorubicin, and dexamethasone (2001–2007, n=75) or a bortezomib-based regimen (2008–2010, n=17).
After induction, patients underwent autologous HSCT using melphalan 200 mg/m2.
A median of 4 months later (range, 2-13), after complete clinical recovery, the patients received an allogeneic transplant from a 6/6 HLA-matched sibling donor. Fifty-seven percent of patients had achieved at least a very good partial response before the second transplant.
The allogeneic transplant was performed on an outpatient basis. The graft consisted of G-CSF-mobilized peripheral blood stem cells (target dose ≥ 4×106 CD34+cells/kg).
The conditioning regimen consisted of fludarabine at 30 mg/m2 and cyclophosphamide at 300 mg/m2 for 5 days. GVHD prophylaxis was oral tacrolimus and mycophenolate mofetil.
Results
At a median follow-up of 8.8 years, 56 patients were still alive (and 36 had died).
Forty patients had no evidence of progression, including 11 patients who were still taking systemic immunosuppressive drugs for GVHD. The remaining 16 patients who were still alive had relapsed after allogeneic HSCT.
A total of 45 patients relapsed. Thirty-nine went on to receive treatment with at least 1 new drug (thalidomide, lenalidomide, bortezomib, or pomalidomide).
They received a median of 2 lines of therapy (range, 1–6), and their 5-year overall survival from first relapse was 49%. Ten of these patients were in very good partial response or greater at last follow-up.
Ten patients died from causes other than refractory MM—6 from refractory GVHD, 3 from lung adenocarcinoma (1 smoker), and 1 from invasive aspergillosis.
The cumulative incidence of extensive chronic GVHD was 79%. The cumulative incidence of grade 2-4 acute GVHD at 6 months was 9%. And 3 patients developed grade 3-4 acute GVHD.
Among the 56 patients who were still alive at last follow-up, the probability of being on any systemic immunosuppressive treatment for GVHD is 38% at 5 years and 22% at 10 years.
The probability of overall survival at 10 years is 62%, the probability of progression-free survival is 41%, and the cumulative incidence of non-relapse mortality is 10%. ![]()

Photo by Chad McNeeley
Tandem autologous/allogeneic hematopoietic stem cell transplant (HSCT) may cure multiple myeloma (MM), according to a phase 2 study.
The study included newly diagnosed patients who received induction therapy followed by an autograft and a non-myeloablative allograft from a matched sibling donor.
The patients have been followed for a median of 8.8 years.
The probability of progression-free survival at 10 years is 41%, and the overall survival is 62%.
The rate of chronic graft-vs-host disease (GVHD) is high, but the rate of non-relapse mortality is low.
“In many hospitals, doctors have abandoned the use of allografts for multiple myeloma due to the risk of toxicity and relapse,” said study author Jean Roy, MD, of the Maisonneuve-Rosemont Hospital and University of Montreal in Quebec, Canada.
“Our results, on the other hand, have led us to offer the treatment to more patients, especially younger patients and those with poorer prognoses.”
Dr Roy and his colleagues reported these results in Bone Marrow Transplantation.
The researchers assessed 92 patients newly diagnosed with MM between 2001 and 2010. Their median age was 52 (range, 39-64), and 97% had Durie–Salmon stage II or III disease.
Patients received an induction regimen consisting of vincristine, doxorubicin, and dexamethasone (2001–2007, n=75) or a bortezomib-based regimen (2008–2010, n=17).
After induction, patients underwent autologous HSCT using melphalan 200 mg/m2.
A median of 4 months later (range, 2-13), after complete clinical recovery, the patients received an allogeneic transplant from a 6/6 HLA-matched sibling donor. Fifty-seven percent of patients had achieved at least a very good partial response before the second transplant.
The allogeneic transplant was performed on an outpatient basis. The graft consisted of G-CSF-mobilized peripheral blood stem cells (target dose ≥ 4×106 CD34+cells/kg).
The conditioning regimen consisted of fludarabine at 30 mg/m2 and cyclophosphamide at 300 mg/m2 for 5 days. GVHD prophylaxis was oral tacrolimus and mycophenolate mofetil.
Results
At a median follow-up of 8.8 years, 56 patients were still alive (and 36 had died).
Forty patients had no evidence of progression, including 11 patients who were still taking systemic immunosuppressive drugs for GVHD. The remaining 16 patients who were still alive had relapsed after allogeneic HSCT.
A total of 45 patients relapsed. Thirty-nine went on to receive treatment with at least 1 new drug (thalidomide, lenalidomide, bortezomib, or pomalidomide).
They received a median of 2 lines of therapy (range, 1–6), and their 5-year overall survival from first relapse was 49%. Ten of these patients were in very good partial response or greater at last follow-up.
Ten patients died from causes other than refractory MM—6 from refractory GVHD, 3 from lung adenocarcinoma (1 smoker), and 1 from invasive aspergillosis.
The cumulative incidence of extensive chronic GVHD was 79%. The cumulative incidence of grade 2-4 acute GVHD at 6 months was 9%. And 3 patients developed grade 3-4 acute GVHD.
Among the 56 patients who were still alive at last follow-up, the probability of being on any systemic immunosuppressive treatment for GVHD is 38% at 5 years and 22% at 10 years.
The probability of overall survival at 10 years is 62%, the probability of progression-free survival is 41%, and the cumulative incidence of non-relapse mortality is 10%. ![]()

Photo by Chad McNeeley
Tandem autologous/allogeneic hematopoietic stem cell transplant (HSCT) may cure multiple myeloma (MM), according to a phase 2 study.
The study included newly diagnosed patients who received induction therapy followed by an autograft and a non-myeloablative allograft from a matched sibling donor.
The patients have been followed for a median of 8.8 years.
The probability of progression-free survival at 10 years is 41%, and the overall survival is 62%.
The rate of chronic graft-vs-host disease (GVHD) is high, but the rate of non-relapse mortality is low.
“In many hospitals, doctors have abandoned the use of allografts for multiple myeloma due to the risk of toxicity and relapse,” said study author Jean Roy, MD, of the Maisonneuve-Rosemont Hospital and University of Montreal in Quebec, Canada.
“Our results, on the other hand, have led us to offer the treatment to more patients, especially younger patients and those with poorer prognoses.”
Dr Roy and his colleagues reported these results in Bone Marrow Transplantation.
The researchers assessed 92 patients newly diagnosed with MM between 2001 and 2010. Their median age was 52 (range, 39-64), and 97% had Durie–Salmon stage II or III disease.
Patients received an induction regimen consisting of vincristine, doxorubicin, and dexamethasone (2001–2007, n=75) or a bortezomib-based regimen (2008–2010, n=17).
After induction, patients underwent autologous HSCT using melphalan 200 mg/m2.
A median of 4 months later (range, 2-13), after complete clinical recovery, the patients received an allogeneic transplant from a 6/6 HLA-matched sibling donor. Fifty-seven percent of patients had achieved at least a very good partial response before the second transplant.
The allogeneic transplant was performed on an outpatient basis. The graft consisted of G-CSF-mobilized peripheral blood stem cells (target dose ≥ 4×106 CD34+cells/kg).
The conditioning regimen consisted of fludarabine at 30 mg/m2 and cyclophosphamide at 300 mg/m2 for 5 days. GVHD prophylaxis was oral tacrolimus and mycophenolate mofetil.
Results
At a median follow-up of 8.8 years, 56 patients were still alive (and 36 had died).
Forty patients had no evidence of progression, including 11 patients who were still taking systemic immunosuppressive drugs for GVHD. The remaining 16 patients who were still alive had relapsed after allogeneic HSCT.
A total of 45 patients relapsed. Thirty-nine went on to receive treatment with at least 1 new drug (thalidomide, lenalidomide, bortezomib, or pomalidomide).
They received a median of 2 lines of therapy (range, 1–6), and their 5-year overall survival from first relapse was 49%. Ten of these patients were in very good partial response or greater at last follow-up.
Ten patients died from causes other than refractory MM—6 from refractory GVHD, 3 from lung adenocarcinoma (1 smoker), and 1 from invasive aspergillosis.
The cumulative incidence of extensive chronic GVHD was 79%. The cumulative incidence of grade 2-4 acute GVHD at 6 months was 9%. And 3 patients developed grade 3-4 acute GVHD.
Among the 56 patients who were still alive at last follow-up, the probability of being on any systemic immunosuppressive treatment for GVHD is 38% at 5 years and 22% at 10 years.
The probability of overall survival at 10 years is 62%, the probability of progression-free survival is 41%, and the cumulative incidence of non-relapse mortality is 10%. ![]()
EMA recommends therapy for bleeding disorder

The European Medicines Agency (EMA) has recommended marketing authorization for Coagadex to treat hereditary factor X deficiency, a rare bleeding disorder.
The active substance in Coagadex is coagulation factor X, a protein derived from human plasma.
Coagadex is intended to treat and prevent bleeding episodes and control bleeding during surgical procedures in patients with hereditary factor X deficiency.
If the European Commission follows the EMA’s recommendation, Coagadex will be the first specific replacement therapy approved in the European Union (EU) to treat patients with this disorder.
Current treatment for factor X deficiency in the EU includes replacement therapies that contain a mix of coagulation factors. These are associated with dosing problems and the risk of elevating other clotting factors, which may result in complications.
Due to the lack of specific treatment options for factor X deficiency, the EMA’s Committee for Medicinal Products for Human Use (CHMP) decided to speed up the evaluation of Coagadex and recommended marketing authorization following an accelerated assessment. This is one of the agency’s tools to speed up patient access to new medicines if they address an unmet medical need.
The CHMP based its recommendation for authorization of Coagadex on the results of 2 non-randomized studies.
The first trial included 16 patients who received Coagadex for pharmacokinetic evaluation, on-demand treatment and control of bleeding episodes, and/or perioperative management of minor surgical or dental procedures.
Coagadex was used to treat 208 bleeding episodes, and 187 of these episodes (in 15 patients) were evaluated for efficacy. Ninety-eight episodes were major bleeds, 88 were minor bleeds, and 1 was not assessed.
One hundred and fifty-five bleeds (83%) were treated with a single infusion of Coagadex, 28 (15%) were treated with 2 infusions, 3 bleeds (2%) required 3 infusions, and 1 bleed (0.5%) required 4 infusions. Four bleeding episodes in 2 patients were considered treatment failures.
The mean dose of Coagadex per infusion was 25.4 IU/kg, and the mean total dose was 30.4 IU/kg. The recommended dose of 25 IU/kg to treat a bleed was maintained for 14 of the 16 patients. The other 2 patients used doses of up to 30 IU/kg and 33 IU/kg.
There were 176 adverse events in this trial, but only 6 events in 2 patients were considered possibly related to Coagadex. This included 2 reports of infusion site erythema in 1 patient, 2 reports of fatigue in 1 patient, 1 report of back pain, and 1 report of infusion site pain.
The second trial included patients who received Coagadex for perioperative management. Five patients received Coagadex for 7 surgical procedures.
For major surgeries, a median of 13 infusions (range, 2-15) and a median cumulative dose of 181 IU/kg (range, 45-210 IU/kg) were required to maintain hemostasis.
For minor surgeries, a median of 2.5 infusions (range, 1-4) and a median cumulative dose of 89 IU/kg (range, 51-127 IU/kg) were required to maintain hemostasis.
There were no adverse events related to Coagadex in this trial.
The company developing Coagadex, Bio Products Laboratory, received scientific advice on the design of the trials from the CHMP. Scientific advice is one of the agency’s main tools to facilitate and stimulate research and development within the EU.
Because factor X deficiency is rare, Coagadex was designated as an orphan medicine by the EMA’s Committee for Orphan Medicinal Products. Orphan designation gives pharmaceutical companies access to incentives that encourage the development of medicines for patients with rare diseases.
The CHMP’s recommendation to approve Coagadex for use in the EU is an intermediary step on Coagadex’s path to patient access. The CHMP’s opinion will now be sent to the European Commission for the adoption of a decision on an EU-wide marketing authorization.
Once a marketing authorization has been granted, decisions about price and reimbursement will take place at the level of each member state, taking into account the potential role/use of this medicine in the context of the national health system of that country. ![]()

The European Medicines Agency (EMA) has recommended marketing authorization for Coagadex to treat hereditary factor X deficiency, a rare bleeding disorder.
The active substance in Coagadex is coagulation factor X, a protein derived from human plasma.
Coagadex is intended to treat and prevent bleeding episodes and control bleeding during surgical procedures in patients with hereditary factor X deficiency.
If the European Commission follows the EMA’s recommendation, Coagadex will be the first specific replacement therapy approved in the European Union (EU) to treat patients with this disorder.
Current treatment for factor X deficiency in the EU includes replacement therapies that contain a mix of coagulation factors. These are associated with dosing problems and the risk of elevating other clotting factors, which may result in complications.
Due to the lack of specific treatment options for factor X deficiency, the EMA’s Committee for Medicinal Products for Human Use (CHMP) decided to speed up the evaluation of Coagadex and recommended marketing authorization following an accelerated assessment. This is one of the agency’s tools to speed up patient access to new medicines if they address an unmet medical need.
The CHMP based its recommendation for authorization of Coagadex on the results of 2 non-randomized studies.
The first trial included 16 patients who received Coagadex for pharmacokinetic evaluation, on-demand treatment and control of bleeding episodes, and/or perioperative management of minor surgical or dental procedures.
Coagadex was used to treat 208 bleeding episodes, and 187 of these episodes (in 15 patients) were evaluated for efficacy. Ninety-eight episodes were major bleeds, 88 were minor bleeds, and 1 was not assessed.
One hundred and fifty-five bleeds (83%) were treated with a single infusion of Coagadex, 28 (15%) were treated with 2 infusions, 3 bleeds (2%) required 3 infusions, and 1 bleed (0.5%) required 4 infusions. Four bleeding episodes in 2 patients were considered treatment failures.
The mean dose of Coagadex per infusion was 25.4 IU/kg, and the mean total dose was 30.4 IU/kg. The recommended dose of 25 IU/kg to treat a bleed was maintained for 14 of the 16 patients. The other 2 patients used doses of up to 30 IU/kg and 33 IU/kg.
There were 176 adverse events in this trial, but only 6 events in 2 patients were considered possibly related to Coagadex. This included 2 reports of infusion site erythema in 1 patient, 2 reports of fatigue in 1 patient, 1 report of back pain, and 1 report of infusion site pain.
The second trial included patients who received Coagadex for perioperative management. Five patients received Coagadex for 7 surgical procedures.
For major surgeries, a median of 13 infusions (range, 2-15) and a median cumulative dose of 181 IU/kg (range, 45-210 IU/kg) were required to maintain hemostasis.
For minor surgeries, a median of 2.5 infusions (range, 1-4) and a median cumulative dose of 89 IU/kg (range, 51-127 IU/kg) were required to maintain hemostasis.
There were no adverse events related to Coagadex in this trial.
The company developing Coagadex, Bio Products Laboratory, received scientific advice on the design of the trials from the CHMP. Scientific advice is one of the agency’s main tools to facilitate and stimulate research and development within the EU.
Because factor X deficiency is rare, Coagadex was designated as an orphan medicine by the EMA’s Committee for Orphan Medicinal Products. Orphan designation gives pharmaceutical companies access to incentives that encourage the development of medicines for patients with rare diseases.
The CHMP’s recommendation to approve Coagadex for use in the EU is an intermediary step on Coagadex’s path to patient access. The CHMP’s opinion will now be sent to the European Commission for the adoption of a decision on an EU-wide marketing authorization.
Once a marketing authorization has been granted, decisions about price and reimbursement will take place at the level of each member state, taking into account the potential role/use of this medicine in the context of the national health system of that country. ![]()

The European Medicines Agency (EMA) has recommended marketing authorization for Coagadex to treat hereditary factor X deficiency, a rare bleeding disorder.
The active substance in Coagadex is coagulation factor X, a protein derived from human plasma.
Coagadex is intended to treat and prevent bleeding episodes and control bleeding during surgical procedures in patients with hereditary factor X deficiency.
If the European Commission follows the EMA’s recommendation, Coagadex will be the first specific replacement therapy approved in the European Union (EU) to treat patients with this disorder.
Current treatment for factor X deficiency in the EU includes replacement therapies that contain a mix of coagulation factors. These are associated with dosing problems and the risk of elevating other clotting factors, which may result in complications.
Due to the lack of specific treatment options for factor X deficiency, the EMA’s Committee for Medicinal Products for Human Use (CHMP) decided to speed up the evaluation of Coagadex and recommended marketing authorization following an accelerated assessment. This is one of the agency’s tools to speed up patient access to new medicines if they address an unmet medical need.
The CHMP based its recommendation for authorization of Coagadex on the results of 2 non-randomized studies.
The first trial included 16 patients who received Coagadex for pharmacokinetic evaluation, on-demand treatment and control of bleeding episodes, and/or perioperative management of minor surgical or dental procedures.
Coagadex was used to treat 208 bleeding episodes, and 187 of these episodes (in 15 patients) were evaluated for efficacy. Ninety-eight episodes were major bleeds, 88 were minor bleeds, and 1 was not assessed.
One hundred and fifty-five bleeds (83%) were treated with a single infusion of Coagadex, 28 (15%) were treated with 2 infusions, 3 bleeds (2%) required 3 infusions, and 1 bleed (0.5%) required 4 infusions. Four bleeding episodes in 2 patients were considered treatment failures.
The mean dose of Coagadex per infusion was 25.4 IU/kg, and the mean total dose was 30.4 IU/kg. The recommended dose of 25 IU/kg to treat a bleed was maintained for 14 of the 16 patients. The other 2 patients used doses of up to 30 IU/kg and 33 IU/kg.
There were 176 adverse events in this trial, but only 6 events in 2 patients were considered possibly related to Coagadex. This included 2 reports of infusion site erythema in 1 patient, 2 reports of fatigue in 1 patient, 1 report of back pain, and 1 report of infusion site pain.
The second trial included patients who received Coagadex for perioperative management. Five patients received Coagadex for 7 surgical procedures.
For major surgeries, a median of 13 infusions (range, 2-15) and a median cumulative dose of 181 IU/kg (range, 45-210 IU/kg) were required to maintain hemostasis.
For minor surgeries, a median of 2.5 infusions (range, 1-4) and a median cumulative dose of 89 IU/kg (range, 51-127 IU/kg) were required to maintain hemostasis.
There were no adverse events related to Coagadex in this trial.
The company developing Coagadex, Bio Products Laboratory, received scientific advice on the design of the trials from the CHMP. Scientific advice is one of the agency’s main tools to facilitate and stimulate research and development within the EU.
Because factor X deficiency is rare, Coagadex was designated as an orphan medicine by the EMA’s Committee for Orphan Medicinal Products. Orphan designation gives pharmaceutical companies access to incentives that encourage the development of medicines for patients with rare diseases.
The CHMP’s recommendation to approve Coagadex for use in the EU is an intermediary step on Coagadex’s path to patient access. The CHMP’s opinion will now be sent to the European Commission for the adoption of a decision on an EU-wide marketing authorization.
Once a marketing authorization has been granted, decisions about price and reimbursement will take place at the level of each member state, taking into account the potential role/use of this medicine in the context of the national health system of that country. ![]()
Induction ALL treatment can cause bone loss
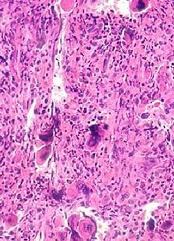
of cancellous bone
Patients with acute lymphoblastic leukemia (ALL) may experience significant bone loss much earlier than previously assumed, according to a study published in the journal Bone.
Investigators analyzed a cohort of adolescent and young adult ALL patients before and after their first month of chemotherapy and observed “significant alterations” to cancellous and cortical bone in this short period of time.
Previous studies to determine the changes to bone density during ALL therapy had focused on the cumulative effects of chemotherapy after months or even years of treatment.
“In clinic, we would see patients with fractures and vertebral compression during the very first few weeks of treatment,” said study author Etan Orgel, MD, of Children’s Hospital Los Angeles in California.
“But we were unaware of any study that specifically examined bone before chemotherapy and immediately after the first 30 days of treatment, which would allow us to understand the impact of this early treatment phase.”
So Dr Orgel and his colleagues conducted a prospective study of 38 patients, ages 10 to 21, who were newly diagnosed with ALL.
The team used quantitative computerized tomography (QCT) to assess leukemia-related changes to bone at diagnosis and then the subsequent effects of the induction phase of chemotherapy.
All of the patients received a 28-day induction regimen consisting of vincristine, pegylated L-asparaginase, anthracycline (daunorubicin or doxorubicin), and a glucocorticoid (either prednisone at 60 mg/m2/day for 28 days or dexamethasone at 10 mg/m2/day for 14 days).
The investigators compared the patients to age- and sex-matched controls and found that leukemia did not dramatically alter the properties of bone before chemotherapy.
However, QCT revealed significant changes during the 30-day induction phase in the 35 patients who were well enough to undergo imaging after treatment.
The patients experienced a significant decrease in cancellous volumetric bone mineral density, which was measured in the spine. The median decrease was 27% (P<0.001).
There was no significant change in cortical volumetric bone mineral density, which was measured in the tibia (−0.0%, P=0.860) or femur (−0.7%, P=0.290).
But there was significant cortical thinning in the tibia. The average cortical thickness decreased 1.2% (P<0.001), and the cortical area decreased 0.4% (P=0.014).
The femur was less affected, the investigators said. There was a decrease in average cortical thickness, but this was not significant (-0.3%, P=0.740).
To help clinicians relate to these findings, the investigators also measured bone mineral density using the older but more widely available technique of dual-energy x-ray absorptiometry. They found that it underestimated these changes as compared to QCT measurements.
“Now that we know how soon bone toxicity occurs, we need to re-evaluate our approaches to managing these changes and focus research efforts on new ways to mitigate this common yet significant adverse effect,” said study author Steven Mittelman, MD, PhD, of Children’s Hospital Los Angeles. ![]()

of cancellous bone
Patients with acute lymphoblastic leukemia (ALL) may experience significant bone loss much earlier than previously assumed, according to a study published in the journal Bone.
Investigators analyzed a cohort of adolescent and young adult ALL patients before and after their first month of chemotherapy and observed “significant alterations” to cancellous and cortical bone in this short period of time.
Previous studies to determine the changes to bone density during ALL therapy had focused on the cumulative effects of chemotherapy after months or even years of treatment.
“In clinic, we would see patients with fractures and vertebral compression during the very first few weeks of treatment,” said study author Etan Orgel, MD, of Children’s Hospital Los Angeles in California.
“But we were unaware of any study that specifically examined bone before chemotherapy and immediately after the first 30 days of treatment, which would allow us to understand the impact of this early treatment phase.”
So Dr Orgel and his colleagues conducted a prospective study of 38 patients, ages 10 to 21, who were newly diagnosed with ALL.
The team used quantitative computerized tomography (QCT) to assess leukemia-related changes to bone at diagnosis and then the subsequent effects of the induction phase of chemotherapy.
All of the patients received a 28-day induction regimen consisting of vincristine, pegylated L-asparaginase, anthracycline (daunorubicin or doxorubicin), and a glucocorticoid (either prednisone at 60 mg/m2/day for 28 days or dexamethasone at 10 mg/m2/day for 14 days).
The investigators compared the patients to age- and sex-matched controls and found that leukemia did not dramatically alter the properties of bone before chemotherapy.
However, QCT revealed significant changes during the 30-day induction phase in the 35 patients who were well enough to undergo imaging after treatment.
The patients experienced a significant decrease in cancellous volumetric bone mineral density, which was measured in the spine. The median decrease was 27% (P<0.001).
There was no significant change in cortical volumetric bone mineral density, which was measured in the tibia (−0.0%, P=0.860) or femur (−0.7%, P=0.290).
But there was significant cortical thinning in the tibia. The average cortical thickness decreased 1.2% (P<0.001), and the cortical area decreased 0.4% (P=0.014).
The femur was less affected, the investigators said. There was a decrease in average cortical thickness, but this was not significant (-0.3%, P=0.740).
To help clinicians relate to these findings, the investigators also measured bone mineral density using the older but more widely available technique of dual-energy x-ray absorptiometry. They found that it underestimated these changes as compared to QCT measurements.
“Now that we know how soon bone toxicity occurs, we need to re-evaluate our approaches to managing these changes and focus research efforts on new ways to mitigate this common yet significant adverse effect,” said study author Steven Mittelman, MD, PhD, of Children’s Hospital Los Angeles. ![]()

of cancellous bone
Patients with acute lymphoblastic leukemia (ALL) may experience significant bone loss much earlier than previously assumed, according to a study published in the journal Bone.
Investigators analyzed a cohort of adolescent and young adult ALL patients before and after their first month of chemotherapy and observed “significant alterations” to cancellous and cortical bone in this short period of time.
Previous studies to determine the changes to bone density during ALL therapy had focused on the cumulative effects of chemotherapy after months or even years of treatment.
“In clinic, we would see patients with fractures and vertebral compression during the very first few weeks of treatment,” said study author Etan Orgel, MD, of Children’s Hospital Los Angeles in California.
“But we were unaware of any study that specifically examined bone before chemotherapy and immediately after the first 30 days of treatment, which would allow us to understand the impact of this early treatment phase.”
So Dr Orgel and his colleagues conducted a prospective study of 38 patients, ages 10 to 21, who were newly diagnosed with ALL.
The team used quantitative computerized tomography (QCT) to assess leukemia-related changes to bone at diagnosis and then the subsequent effects of the induction phase of chemotherapy.
All of the patients received a 28-day induction regimen consisting of vincristine, pegylated L-asparaginase, anthracycline (daunorubicin or doxorubicin), and a glucocorticoid (either prednisone at 60 mg/m2/day for 28 days or dexamethasone at 10 mg/m2/day for 14 days).
The investigators compared the patients to age- and sex-matched controls and found that leukemia did not dramatically alter the properties of bone before chemotherapy.
However, QCT revealed significant changes during the 30-day induction phase in the 35 patients who were well enough to undergo imaging after treatment.
The patients experienced a significant decrease in cancellous volumetric bone mineral density, which was measured in the spine. The median decrease was 27% (P<0.001).
There was no significant change in cortical volumetric bone mineral density, which was measured in the tibia (−0.0%, P=0.860) or femur (−0.7%, P=0.290).
But there was significant cortical thinning in the tibia. The average cortical thickness decreased 1.2% (P<0.001), and the cortical area decreased 0.4% (P=0.014).
The femur was less affected, the investigators said. There was a decrease in average cortical thickness, but this was not significant (-0.3%, P=0.740).
To help clinicians relate to these findings, the investigators also measured bone mineral density using the older but more widely available technique of dual-energy x-ray absorptiometry. They found that it underestimated these changes as compared to QCT measurements.
“Now that we know how soon bone toxicity occurs, we need to re-evaluate our approaches to managing these changes and focus research efforts on new ways to mitigate this common yet significant adverse effect,” said study author Steven Mittelman, MD, PhD, of Children’s Hospital Los Angeles. ![]()
Rivaroxaban trial results hold up, EMA says

The European Medicines Agency (EMA) has concluded that a defect discovered in a system used to measure international normalized ratios (INRs) in the ROCKET AF study does not change the study’s overall conclusions.
The Alere INRatio Monitor System (INRatio Monitor or INRatio2 Monitor and INRatio Test Strips) was recalled in December 2014 after it was found to produce falsely low test results.
A recent investigation by The BMJ suggested this defect may have impacted the results of ROCKET AF, in which researchers compared warfarin to rivaroxaban (Xarelto) in patients with non-valvular atrial fibrillation (NVAF).
However, the EMA said further analyses of the study suggest the issue with the INRatio system did not affect the overall safety or benefit-risk balance of rivaroxaban. So rivaroxaban can continue to be used as before, in line with the current prescribing information.
The ROCKET AF study was the main clinical trial underpinning the use of rivaroxaban in patients with NVAF.
The results suggested rivaroxaban was noninferior to warfarin for preventing stroke or systemic embolism in these patients. And there was no significant difference between the treatment arms with regard to major or nonmajor clinically relevant bleeding.
The INRatio system was used to measure INRs in study subjects taking warfarin. Because of the defect, there were concerns that the system could have provided lower INR values in some patients in the warfarin group.
The lower values could, in turn, have led investigators to give too high a dose in the warfarin group, increasing their risk of bleeding and therefore giving a false impression of the comparative safety of rivaroxaban.
So the EMA’s Committee for Medicinal Products for Human Use (CHMP) assessed further analyses of the ROCKET AF study data, taking into account the defect of the INRatio system.
The CHMP concluded that any incorrect measurements obtained with the defective system would have had a marginal effect on the study results, and the safety of rivaroxaban remains unchanged.
In addition, the CHMP said data from other large studies confirmed the comparative safety of rivaroxaban and showed similar rates of bleeding in their warfarin groups.
The CHMP therefore concluded that the benefit-risk balance of rivaroxaban in patients with NVAF remains unchanged.
The CHMP’s assessment report, which includes detailed information on the analyses performed, will be published on the EMA’s website soon.
The EMA started investigating this issue as soon as it was informed of the defect of the INRatio system by the marketing authorization holder of rivaroxaban, Bayer Pharma AG, in September 2015.
Bayer said that, although the INRatio system was recalled in December 2014, the company and its development partner, Janssen, did not become aware of the defect until September 2015. ![]()

The European Medicines Agency (EMA) has concluded that a defect discovered in a system used to measure international normalized ratios (INRs) in the ROCKET AF study does not change the study’s overall conclusions.
The Alere INRatio Monitor System (INRatio Monitor or INRatio2 Monitor and INRatio Test Strips) was recalled in December 2014 after it was found to produce falsely low test results.
A recent investigation by The BMJ suggested this defect may have impacted the results of ROCKET AF, in which researchers compared warfarin to rivaroxaban (Xarelto) in patients with non-valvular atrial fibrillation (NVAF).
However, the EMA said further analyses of the study suggest the issue with the INRatio system did not affect the overall safety or benefit-risk balance of rivaroxaban. So rivaroxaban can continue to be used as before, in line with the current prescribing information.
The ROCKET AF study was the main clinical trial underpinning the use of rivaroxaban in patients with NVAF.
The results suggested rivaroxaban was noninferior to warfarin for preventing stroke or systemic embolism in these patients. And there was no significant difference between the treatment arms with regard to major or nonmajor clinically relevant bleeding.
The INRatio system was used to measure INRs in study subjects taking warfarin. Because of the defect, there were concerns that the system could have provided lower INR values in some patients in the warfarin group.
The lower values could, in turn, have led investigators to give too high a dose in the warfarin group, increasing their risk of bleeding and therefore giving a false impression of the comparative safety of rivaroxaban.
So the EMA’s Committee for Medicinal Products for Human Use (CHMP) assessed further analyses of the ROCKET AF study data, taking into account the defect of the INRatio system.
The CHMP concluded that any incorrect measurements obtained with the defective system would have had a marginal effect on the study results, and the safety of rivaroxaban remains unchanged.
In addition, the CHMP said data from other large studies confirmed the comparative safety of rivaroxaban and showed similar rates of bleeding in their warfarin groups.
The CHMP therefore concluded that the benefit-risk balance of rivaroxaban in patients with NVAF remains unchanged.
The CHMP’s assessment report, which includes detailed information on the analyses performed, will be published on the EMA’s website soon.
The EMA started investigating this issue as soon as it was informed of the defect of the INRatio system by the marketing authorization holder of rivaroxaban, Bayer Pharma AG, in September 2015.
Bayer said that, although the INRatio system was recalled in December 2014, the company and its development partner, Janssen, did not become aware of the defect until September 2015. ![]()

The European Medicines Agency (EMA) has concluded that a defect discovered in a system used to measure international normalized ratios (INRs) in the ROCKET AF study does not change the study’s overall conclusions.
The Alere INRatio Monitor System (INRatio Monitor or INRatio2 Monitor and INRatio Test Strips) was recalled in December 2014 after it was found to produce falsely low test results.
A recent investigation by The BMJ suggested this defect may have impacted the results of ROCKET AF, in which researchers compared warfarin to rivaroxaban (Xarelto) in patients with non-valvular atrial fibrillation (NVAF).
However, the EMA said further analyses of the study suggest the issue with the INRatio system did not affect the overall safety or benefit-risk balance of rivaroxaban. So rivaroxaban can continue to be used as before, in line with the current prescribing information.
The ROCKET AF study was the main clinical trial underpinning the use of rivaroxaban in patients with NVAF.
The results suggested rivaroxaban was noninferior to warfarin for preventing stroke or systemic embolism in these patients. And there was no significant difference between the treatment arms with regard to major or nonmajor clinically relevant bleeding.
The INRatio system was used to measure INRs in study subjects taking warfarin. Because of the defect, there were concerns that the system could have provided lower INR values in some patients in the warfarin group.
The lower values could, in turn, have led investigators to give too high a dose in the warfarin group, increasing their risk of bleeding and therefore giving a false impression of the comparative safety of rivaroxaban.
So the EMA’s Committee for Medicinal Products for Human Use (CHMP) assessed further analyses of the ROCKET AF study data, taking into account the defect of the INRatio system.
The CHMP concluded that any incorrect measurements obtained with the defective system would have had a marginal effect on the study results, and the safety of rivaroxaban remains unchanged.
In addition, the CHMP said data from other large studies confirmed the comparative safety of rivaroxaban and showed similar rates of bleeding in their warfarin groups.
The CHMP therefore concluded that the benefit-risk balance of rivaroxaban in patients with NVAF remains unchanged.
The CHMP’s assessment report, which includes detailed information on the analyses performed, will be published on the EMA’s website soon.
The EMA started investigating this issue as soon as it was informed of the defect of the INRatio system by the marketing authorization holder of rivaroxaban, Bayer Pharma AG, in September 2015.
Bayer said that, although the INRatio system was recalled in December 2014, the company and its development partner, Janssen, did not become aware of the defect until September 2015. ![]()
New assay detects persistent CML better, team says
A new assay is more accurate than the current gold standard for detecting residual disease in patients with chronic myeloid leukemia (CML), according to a study published in The Journal of Molecular Diagnostics.
Investigators found this test, a DNA-based digital PCR (dPCR) assay, could detect persistent disease in 81% of samples taken from CML patients who were in remission according to reverse transcriptase-quantitative PCR (RT-qPCR).
RT-qPCR is currently the most widely used method for monitoring residual disease in CML patients.
“If validated in clinical trials of stopping TKIs [tyrosine kinase inhibitors], this technique [the dPCR assay] will permit a more personalized approach to recommendations for dose reduction or drug cessation in individual patients, ensuring that therapy is withdrawn only from patients with the highest chance of long-term remission,” said investigator Jane F. Apperley, MD, PhD, of Imperial College London in the UK.
For this study, Dr Apperley and her colleagues compared the sensitivity of the dPCR assay to 3 other quantitative PCR methods currently used to measure residual CML—RT-qPCR, quantitative PCR (qPCR), and reverse transcriptase-digital PCR (RT-dPCR).
Thirty-six samples were taken from 6 patients with early CML who were thought to be in deep molecular remission, as indicated by RT-qPCR results.
Repeat analysis using dPCR with preamplification detected persistent disease in 81% of the samples. In comparison, the detection rate was 25% using RT-dPCR and 19% for qPCR.
“We conclude that dPCR for BCR-ABL1 DNA is the most sensitive available method of residual disease detection in CML and may prove useful in the management of TKI withdrawal,” Dr Apperley said.
She and her colleagues believe the new assay has the potential to dramatically impact CML management. They foresee that, immediately after CML diagnosis, the patient’s genomic breakpoints would be identified, enabling the design of a patient-specific assay.
The patient’s response to therapy would be monitored using standard RT-qPCR until reaching molecular remission. At that point, routine monitoring would be augmented with dPCR, allowing better-informed treatment decisions and improved patient management.
According to Dr Apperley, the new method improves on previous methodologies in 2 key areas. First, the dPCR platform provides greater sensitivity.
And second, dPCR is a DNA-based method that allows identification of BCR-ABL1 fusion junctions by targeted next-generation sequencing. This enables the rapid generation of high-performing DNA-based hydrolysis probe assays that are specific to the individual molecular footprint of each patient’s CML clone, although the number and location of fusion junctions may vary among patients.
“The technique we describe, with which we successfully mapped a disease-specific junction in all patients tested, is relatively simple, cost-effective, and suited to a high-throughput laboratory,” Dr Apperley concluded. ![]()

A new assay is more accurate than the current gold standard for detecting residual disease in patients with chronic myeloid leukemia (CML), according to a study published in The Journal of Molecular Diagnostics.
Investigators found this test, a DNA-based digital PCR (dPCR) assay, could detect persistent disease in 81% of samples taken from CML patients who were in remission according to reverse transcriptase-quantitative PCR (RT-qPCR).
RT-qPCR is currently the most widely used method for monitoring residual disease in CML patients.
“If validated in clinical trials of stopping TKIs [tyrosine kinase inhibitors], this technique [the dPCR assay] will permit a more personalized approach to recommendations for dose reduction or drug cessation in individual patients, ensuring that therapy is withdrawn only from patients with the highest chance of long-term remission,” said investigator Jane F. Apperley, MD, PhD, of Imperial College London in the UK.
For this study, Dr Apperley and her colleagues compared the sensitivity of the dPCR assay to 3 other quantitative PCR methods currently used to measure residual CML—RT-qPCR, quantitative PCR (qPCR), and reverse transcriptase-digital PCR (RT-dPCR).
Thirty-six samples were taken from 6 patients with early CML who were thought to be in deep molecular remission, as indicated by RT-qPCR results.
Repeat analysis using dPCR with preamplification detected persistent disease in 81% of the samples. In comparison, the detection rate was 25% using RT-dPCR and 19% for qPCR.
“We conclude that dPCR for BCR-ABL1 DNA is the most sensitive available method of residual disease detection in CML and may prove useful in the management of TKI withdrawal,” Dr Apperley said.
She and her colleagues believe the new assay has the potential to dramatically impact CML management. They foresee that, immediately after CML diagnosis, the patient’s genomic breakpoints would be identified, enabling the design of a patient-specific assay.
The patient’s response to therapy would be monitored using standard RT-qPCR until reaching molecular remission. At that point, routine monitoring would be augmented with dPCR, allowing better-informed treatment decisions and improved patient management.
According to Dr Apperley, the new method improves on previous methodologies in 2 key areas. First, the dPCR platform provides greater sensitivity.
And second, dPCR is a DNA-based method that allows identification of BCR-ABL1 fusion junctions by targeted next-generation sequencing. This enables the rapid generation of high-performing DNA-based hydrolysis probe assays that are specific to the individual molecular footprint of each patient’s CML clone, although the number and location of fusion junctions may vary among patients.
“The technique we describe, with which we successfully mapped a disease-specific junction in all patients tested, is relatively simple, cost-effective, and suited to a high-throughput laboratory,” Dr Apperley concluded. ![]()

A new assay is more accurate than the current gold standard for detecting residual disease in patients with chronic myeloid leukemia (CML), according to a study published in The Journal of Molecular Diagnostics.
Investigators found this test, a DNA-based digital PCR (dPCR) assay, could detect persistent disease in 81% of samples taken from CML patients who were in remission according to reverse transcriptase-quantitative PCR (RT-qPCR).
RT-qPCR is currently the most widely used method for monitoring residual disease in CML patients.
“If validated in clinical trials of stopping TKIs [tyrosine kinase inhibitors], this technique [the dPCR assay] will permit a more personalized approach to recommendations for dose reduction or drug cessation in individual patients, ensuring that therapy is withdrawn only from patients with the highest chance of long-term remission,” said investigator Jane F. Apperley, MD, PhD, of Imperial College London in the UK.
For this study, Dr Apperley and her colleagues compared the sensitivity of the dPCR assay to 3 other quantitative PCR methods currently used to measure residual CML—RT-qPCR, quantitative PCR (qPCR), and reverse transcriptase-digital PCR (RT-dPCR).
Thirty-six samples were taken from 6 patients with early CML who were thought to be in deep molecular remission, as indicated by RT-qPCR results.
Repeat analysis using dPCR with preamplification detected persistent disease in 81% of the samples. In comparison, the detection rate was 25% using RT-dPCR and 19% for qPCR.
“We conclude that dPCR for BCR-ABL1 DNA is the most sensitive available method of residual disease detection in CML and may prove useful in the management of TKI withdrawal,” Dr Apperley said.
She and her colleagues believe the new assay has the potential to dramatically impact CML management. They foresee that, immediately after CML diagnosis, the patient’s genomic breakpoints would be identified, enabling the design of a patient-specific assay.
The patient’s response to therapy would be monitored using standard RT-qPCR until reaching molecular remission. At that point, routine monitoring would be augmented with dPCR, allowing better-informed treatment decisions and improved patient management.
According to Dr Apperley, the new method improves on previous methodologies in 2 key areas. First, the dPCR platform provides greater sensitivity.
And second, dPCR is a DNA-based method that allows identification of BCR-ABL1 fusion junctions by targeted next-generation sequencing. This enables the rapid generation of high-performing DNA-based hydrolysis probe assays that are specific to the individual molecular footprint of each patient’s CML clone, although the number and location of fusion junctions may vary among patients.
“The technique we describe, with which we successfully mapped a disease-specific junction in all patients tested, is relatively simple, cost-effective, and suited to a high-throughput laboratory,” Dr Apperley concluded.
FDA approves drug for patients receiving MEC

Photo by Rhoda Baer
The US Food and Drug Administration (FDA) has approved a supplemental new drug application for single-dose fosaprepitant dimeglumine (Emend) for injection.
The agency approved the substance P/neurokinin-1 (NK1) receptor antagonist for use in combination with other anti-emetic medicines to prevent delayed nausea and vomiting in adults receiving initial and repeat courses of moderately emetogenic chemotherapy (MEC).
This makes fosaprepitant dimeglumine the first intravenous NK1 receptor antagonist approved in the US for patients receiving either highly emetogenic chemotherapy or MEC.
Fosaprepitant dimeglumine has not been studied for the treatment of established nausea and vomiting.
The FDA’s latest approval of fosaprepitant dimeglumine is supported by data from a phase 3 study published in the Annals of Oncology.
Patients receiving MEC were given ondansetron and dexamethasone (n=498) or ondansetron and dexamethasone plus a single intravenous infusion of fosaprepitant dimeglumine (n=502).
The primary endpoint was complete response (CR)—defined as no vomiting and no use of rescue therapy—in the delayed phase of chemotherapy-induced nausea and vomiting, which is 25 to 120 hours after the initiation of chemotherapy.
Secondary endpoints included CR in the overall and acute phases—0 to 120 and 0 to 24 hours after MEC initiation, respectively—and no vomiting in the overall phase.
The fosaprepitant regimen improved CR significantly in the delayed and overall phases but not in the acute phase.
In the delayed phase, the CR rate was 78.9% with the fosaprepitant regimen and 68.5% with the control regimen (P<0.001). In the acute phase, the CR rate was 93.2% and 91.0%, respectively (P=0.184). Overall, the CR rate was 77.1% and 66.9%, respectively (P<0.001).
In the overall phase, the proportion of subjects with no vomiting was 82.7% with the fosaprepitant regimen and 72.9% with the control regimen (P<0.001). The proportion of patients with no significant nausea was 83.2% and 77.9%, respectively (P=0.030).
The most common adverse events reported in the fosaprepitant and control arms, respectively, were fatigue (15% vs 13%), diarrhea (13% vs 11%), neutropenia (8% vs 7%), asthenia (4% vs 3%), anemia (3% vs 2%), peripheral neuropathy (3% vs 2%), leukopenia (2% vs 1%), dyspepsia (2% vs 1%), urinary tract infection (2% vs 1%), and pain in extremity (2% vs 1%).
Fosaprepitant dimeglumine is a product of Merck. For more details on the drug, see the prescribing information.

Photo by Rhoda Baer
The US Food and Drug Administration (FDA) has approved a supplemental new drug application for single-dose fosaprepitant dimeglumine (Emend) for injection.
The agency approved the substance P/neurokinin-1 (NK1) receptor antagonist for use in combination with other anti-emetic medicines to prevent delayed nausea and vomiting in adults receiving initial and repeat courses of moderately emetogenic chemotherapy (MEC).
This makes fosaprepitant dimeglumine the first intravenous NK1 receptor antagonist approved in the US for patients receiving either highly emetogenic chemotherapy or MEC.
Fosaprepitant dimeglumine has not been studied for the treatment of established nausea and vomiting.
The FDA’s latest approval of fosaprepitant dimeglumine is supported by data from a phase 3 study published in the Annals of Oncology.
Patients receiving MEC were given ondansetron and dexamethasone (n=498) or ondansetron and dexamethasone plus a single intravenous infusion of fosaprepitant dimeglumine (n=502).
The primary endpoint was complete response (CR)—defined as no vomiting and no use of rescue therapy—in the delayed phase of chemotherapy-induced nausea and vomiting, which is 25 to 120 hours after the initiation of chemotherapy.
Secondary endpoints included CR in the overall and acute phases—0 to 120 and 0 to 24 hours after MEC initiation, respectively—and no vomiting in the overall phase.
The fosaprepitant regimen improved CR significantly in the delayed and overall phases but not in the acute phase.
In the delayed phase, the CR rate was 78.9% with the fosaprepitant regimen and 68.5% with the control regimen (P<0.001). In the acute phase, the CR rate was 93.2% and 91.0%, respectively (P=0.184). Overall, the CR rate was 77.1% and 66.9%, respectively (P<0.001).
In the overall phase, the proportion of subjects with no vomiting was 82.7% with the fosaprepitant regimen and 72.9% with the control regimen (P<0.001). The proportion of patients with no significant nausea was 83.2% and 77.9%, respectively (P=0.030).
The most common adverse events reported in the fosaprepitant and control arms, respectively, were fatigue (15% vs 13%), diarrhea (13% vs 11%), neutropenia (8% vs 7%), asthenia (4% vs 3%), anemia (3% vs 2%), peripheral neuropathy (3% vs 2%), leukopenia (2% vs 1%), dyspepsia (2% vs 1%), urinary tract infection (2% vs 1%), and pain in extremity (2% vs 1%).
Fosaprepitant dimeglumine is a product of Merck. For more details on the drug, see the prescribing information.

Photo by Rhoda Baer
The US Food and Drug Administration (FDA) has approved a supplemental new drug application for single-dose fosaprepitant dimeglumine (Emend) for injection.
The agency approved the substance P/neurokinin-1 (NK1) receptor antagonist for use in combination with other anti-emetic medicines to prevent delayed nausea and vomiting in adults receiving initial and repeat courses of moderately emetogenic chemotherapy (MEC).
This makes fosaprepitant dimeglumine the first intravenous NK1 receptor antagonist approved in the US for patients receiving either highly emetogenic chemotherapy or MEC.
Fosaprepitant dimeglumine has not been studied for the treatment of established nausea and vomiting.
The FDA’s latest approval of fosaprepitant dimeglumine is supported by data from a phase 3 study published in the Annals of Oncology.
Patients receiving MEC were given ondansetron and dexamethasone (n=498) or ondansetron and dexamethasone plus a single intravenous infusion of fosaprepitant dimeglumine (n=502).
The primary endpoint was complete response (CR)—defined as no vomiting and no use of rescue therapy—in the delayed phase of chemotherapy-induced nausea and vomiting, which is 25 to 120 hours after the initiation of chemotherapy.
Secondary endpoints included CR in the overall and acute phases—0 to 120 and 0 to 24 hours after MEC initiation, respectively—and no vomiting in the overall phase.
The fosaprepitant regimen improved CR significantly in the delayed and overall phases but not in the acute phase.
In the delayed phase, the CR rate was 78.9% with the fosaprepitant regimen and 68.5% with the control regimen (P<0.001). In the acute phase, the CR rate was 93.2% and 91.0%, respectively (P=0.184). Overall, the CR rate was 77.1% and 66.9%, respectively (P<0.001).
In the overall phase, the proportion of subjects with no vomiting was 82.7% with the fosaprepitant regimen and 72.9% with the control regimen (P<0.001). The proportion of patients with no significant nausea was 83.2% and 77.9%, respectively (P=0.030).
The most common adverse events reported in the fosaprepitant and control arms, respectively, were fatigue (15% vs 13%), diarrhea (13% vs 11%), neutropenia (8% vs 7%), asthenia (4% vs 3%), anemia (3% vs 2%), peripheral neuropathy (3% vs 2%), leukopenia (2% vs 1%), dyspepsia (2% vs 1%), urinary tract infection (2% vs 1%), and pain in extremity (2% vs 1%).
Fosaprepitant dimeglumine is a product of Merck. For more details on the drug, see the prescribing information.
Brazil reports Zika transmission via transfusion
Photo by Marja Helander
Health officials in Campinas, Brazil, have reported 2 cases of the Zika virus that were likely transmitted through blood transfusions.
The officials said both transfusions occurred last year, but transmission of the virus wasn’t confirmed until recently.
A liver transplant recipient appeared to have contracted Zika through a transfusion in March 2015, and a gunshot victim appeared to have contracted the virus after multiple transfusions in April 2015.
Doctors originally thought the gunshot victim had dengue fever, so his blood was not tested for the Zika virus until January 28. The man later died of his wounds.
The liver transplant recipient did not show any symptoms of Zika infection, but testing confirmed that both he and his blood donor had the virus.
Protecting the world’s blood supply
Even before these cases of Zika transmission were reported, countries around the world were implementing blood donor deferral policies in attempts to protect the blood supply.
The US Food and Drug Administration is still reviewing its blood donation policy with regard to the virus, but the American Red Cross and AABB have recommended donor self-deferral.
Both organizations said people should refrain from donating blood for 28 days if they have visited Mexico, the Caribbean, Central America, or South America in the past 4 weeks.
NHS Blood and Transplant has also implemented a 28-day deferral period for potential blood donors in England and North Wales who have travelled to countries where the Zika virus is endemic.
A spokesperson for NHS Blood and Transplant said travel to most Zika-endemic countries already brings a blood donation deferral period of at least 28 days. So the agency doesn’t expect the new deferral policy to have a significant impact on the number of people who can donate following travel abroad.
Canadian Blood Services has said that anyone who has travelled outside of Canada, the continental US, and Europe will be temporarily ineligible to give blood for 3 weeks (21 days). This policy has been implemented across the country.
The 21-day waiting period also applies to cord blood and stem cell donors who have travelled to affected areas. Héma-Québec (Quebec’s blood operator) is implementing the same change.
Canadian Blood Services said the new deferral policy will reduce the number of people available to donate in the coming months and therefore urged Canadians to donate before they travel. The agency also urged new and current donors who have not recently travelled outside of Canada, the continental US, and Europe to book an appointment to donate.
Hong Kong’s Red Cross Blood Transfusion Service has begun screening potential blood donors. Anyone who has resided in or visited any countries affected by the Zika virus is screened and deferred for blood donation for at least 28 days from the date he or she departed from the affected country.
South Korea’s health authorities have implemented a 30-day deferral period for potential donors who have visited Zika-endemic areas.
The Australian Red Cross Blood Service said it already defers potential blood donors who have travelled to countries with mosquito-borne viruses that are a transfusion-transmission risk, such as dengue and malaria. So all countries affected by Zika outbreaks are already covered by temporary travel deferrals in Australia.
However, the organization said it will continue to monitor the countries impacted by the virus and will make any adjustments to deferrals as required.

Photo by Marja Helander
Health officials in Campinas, Brazil, have reported 2 cases of the Zika virus that were likely transmitted through blood transfusions.
The officials said both transfusions occurred last year, but transmission of the virus wasn’t confirmed until recently.
A liver transplant recipient appeared to have contracted Zika through a transfusion in March 2015, and a gunshot victim appeared to have contracted the virus after multiple transfusions in April 2015.
Doctors originally thought the gunshot victim had dengue fever, so his blood was not tested for the Zika virus until January 28. The man later died of his wounds.
The liver transplant recipient did not show any symptoms of Zika infection, but testing confirmed that both he and his blood donor had the virus.
Protecting the world’s blood supply
Even before these cases of Zika transmission were reported, countries around the world were implementing blood donor deferral policies in attempts to protect the blood supply.
The US Food and Drug Administration is still reviewing its blood donation policy with regard to the virus, but the American Red Cross and AABB have recommended donor self-deferral.
Both organizations said people should refrain from donating blood for 28 days if they have visited Mexico, the Caribbean, Central America, or South America in the past 4 weeks.
NHS Blood and Transplant has also implemented a 28-day deferral period for potential blood donors in England and North Wales who have travelled to countries where the Zika virus is endemic.
A spokesperson for NHS Blood and Transplant said travel to most Zika-endemic countries already brings a blood donation deferral period of at least 28 days. So the agency doesn’t expect the new deferral policy to have a significant impact on the number of people who can donate following travel abroad.
Canadian Blood Services has said that anyone who has travelled outside of Canada, the continental US, and Europe will be temporarily ineligible to give blood for 3 weeks (21 days). This policy has been implemented across the country.
The 21-day waiting period also applies to cord blood and stem cell donors who have travelled to affected areas. Héma-Québec (Quebec’s blood operator) is implementing the same change.
Canadian Blood Services said the new deferral policy will reduce the number of people available to donate in the coming months and therefore urged Canadians to donate before they travel. The agency also urged new and current donors who have not recently travelled outside of Canada, the continental US, and Europe to book an appointment to donate.
Hong Kong’s Red Cross Blood Transfusion Service has begun screening potential blood donors. Anyone who has resided in or visited any countries affected by the Zika virus is screened and deferred for blood donation for at least 28 days from the date he or she departed from the affected country.
South Korea’s health authorities have implemented a 30-day deferral period for potential donors who have visited Zika-endemic areas.
The Australian Red Cross Blood Service said it already defers potential blood donors who have travelled to countries with mosquito-borne viruses that are a transfusion-transmission risk, such as dengue and malaria. So all countries affected by Zika outbreaks are already covered by temporary travel deferrals in Australia.
However, the organization said it will continue to monitor the countries impacted by the virus and will make any adjustments to deferrals as required.

Photo by Marja Helander
Health officials in Campinas, Brazil, have reported 2 cases of the Zika virus that were likely transmitted through blood transfusions.
The officials said both transfusions occurred last year, but transmission of the virus wasn’t confirmed until recently.
A liver transplant recipient appeared to have contracted Zika through a transfusion in March 2015, and a gunshot victim appeared to have contracted the virus after multiple transfusions in April 2015.
Doctors originally thought the gunshot victim had dengue fever, so his blood was not tested for the Zika virus until January 28. The man later died of his wounds.
The liver transplant recipient did not show any symptoms of Zika infection, but testing confirmed that both he and his blood donor had the virus.
Protecting the world’s blood supply
Even before these cases of Zika transmission were reported, countries around the world were implementing blood donor deferral policies in attempts to protect the blood supply.
The US Food and Drug Administration is still reviewing its blood donation policy with regard to the virus, but the American Red Cross and AABB have recommended donor self-deferral.
Both organizations said people should refrain from donating blood for 28 days if they have visited Mexico, the Caribbean, Central America, or South America in the past 4 weeks.
NHS Blood and Transplant has also implemented a 28-day deferral period for potential blood donors in England and North Wales who have travelled to countries where the Zika virus is endemic.
A spokesperson for NHS Blood and Transplant said travel to most Zika-endemic countries already brings a blood donation deferral period of at least 28 days. So the agency doesn’t expect the new deferral policy to have a significant impact on the number of people who can donate following travel abroad.
Canadian Blood Services has said that anyone who has travelled outside of Canada, the continental US, and Europe will be temporarily ineligible to give blood for 3 weeks (21 days). This policy has been implemented across the country.
The 21-day waiting period also applies to cord blood and stem cell donors who have travelled to affected areas. Héma-Québec (Quebec’s blood operator) is implementing the same change.
Canadian Blood Services said the new deferral policy will reduce the number of people available to donate in the coming months and therefore urged Canadians to donate before they travel. The agency also urged new and current donors who have not recently travelled outside of Canada, the continental US, and Europe to book an appointment to donate.
Hong Kong’s Red Cross Blood Transfusion Service has begun screening potential blood donors. Anyone who has resided in or visited any countries affected by the Zika virus is screened and deferred for blood donation for at least 28 days from the date he or she departed from the affected country.
South Korea’s health authorities have implemented a 30-day deferral period for potential donors who have visited Zika-endemic areas.
The Australian Red Cross Blood Service said it already defers potential blood donors who have travelled to countries with mosquito-borne viruses that are a transfusion-transmission risk, such as dengue and malaria. So all countries affected by Zika outbreaks are already covered by temporary travel deferrals in Australia.
However, the organization said it will continue to monitor the countries impacted by the virus and will make any adjustments to deferrals as required.
Generic imatinib launched with savings program

Photo by Rhoda Baer
Sun Pharma has announced the US launch of imatinib mesylate tablets, which are a generic version of Novartis’s Gleevec, for indications approved by the US Food and Drug Administration (FDA).
As part of this launch, Sun Pharma has rolled out a savings card program. The goal is to provide greater access to imatinib mesylate tablets for patients who have commercial insurance, but their out-of-pocket cost may exceed an affordable amount.
Sun Pharma’s Imatinib Mesylate Savings Card will reduce patient’s co-payment to $10. The card will also offer patients an additional savings benefit of up to $700 for a 30-day fill to offset any additional out-of-pocket cost should they be required to meet their deductible or co-insurance.
Participating pharmacies across the US can use the patient’s card as part of this program.
Eligible patients can participate in Sun Pharma’s Imatinib Mesylate Savings Card program by registering at www.imatinibrx.com or by requesting a savings card from their oncologist. Sun Pharma will be supplying its Imatinib Mesylate Savings Cards to more than 4500 oncologists.
Sun Pharma has established a Hub service so patients can call and speak with a trained healthcare professional about imatinib mesylate. The number is 1-844-502-5950.
In addition, qualifying patients can receive Sun Pharma’s imatinib mesylate at no cost. Based on qualifications for applying and including a doctor’s prescription, the Hub service will determine if a patient is qualified to receive imatinib mesylate for free. Upon acceptance, the prescription will be processed and delivered to the qualifying patient at no cost.
Sun Pharma’s imatinib mesylate was approved by the FDA in December 2015 and was granted 180 days of marketing exclusivity from the time of its launch. The drug is available in 100 mg and 400 mg tablets.
It is approved to treat:
- Newly diagnosed adult and pediatric patients with Philadelphia-chromosome-positive chronic myeloid leukemia (Ph+ CML) in chronic phase
- Patients with Ph+ CML in blast crisis, accelerated phase, or in chronic phase after failure of interferon-alpha therapy
- Adults with relapsed or refractory Ph+ acute lymphoblastic leukemia
- Adults with myelodysplastic/myeloproliferative diseases associated with PDGFR gene re-arrangements
- Adults with aggressive systemic mastocytosis without the D816V c-Kit mutation or with c-Kit mutational status unknown
- Adults with hypereosinophilic syndrome and/or chronic eosinophilic leukemia, including those who have the FIP1L1-PDGFRα fusion kinase
- Adult patients with unresectable, recurrent, and/or metastatic dermatofibrosarcoma protuberans.
Sun Pharma’s imatinib mesylate is not approved to treat patients with KIT (CD117)-positive unresectable and/or metastatic malignant gastrointestinal stromal tumors.

Photo by Rhoda Baer
Sun Pharma has announced the US launch of imatinib mesylate tablets, which are a generic version of Novartis’s Gleevec, for indications approved by the US Food and Drug Administration (FDA).
As part of this launch, Sun Pharma has rolled out a savings card program. The goal is to provide greater access to imatinib mesylate tablets for patients who have commercial insurance, but their out-of-pocket cost may exceed an affordable amount.
Sun Pharma’s Imatinib Mesylate Savings Card will reduce patient’s co-payment to $10. The card will also offer patients an additional savings benefit of up to $700 for a 30-day fill to offset any additional out-of-pocket cost should they be required to meet their deductible or co-insurance.
Participating pharmacies across the US can use the patient’s card as part of this program.
Eligible patients can participate in Sun Pharma’s Imatinib Mesylate Savings Card program by registering at www.imatinibrx.com or by requesting a savings card from their oncologist. Sun Pharma will be supplying its Imatinib Mesylate Savings Cards to more than 4500 oncologists.
Sun Pharma has established a Hub service so patients can call and speak with a trained healthcare professional about imatinib mesylate. The number is 1-844-502-5950.
In addition, qualifying patients can receive Sun Pharma’s imatinib mesylate at no cost. Based on qualifications for applying and including a doctor’s prescription, the Hub service will determine if a patient is qualified to receive imatinib mesylate for free. Upon acceptance, the prescription will be processed and delivered to the qualifying patient at no cost.
Sun Pharma’s imatinib mesylate was approved by the FDA in December 2015 and was granted 180 days of marketing exclusivity from the time of its launch. The drug is available in 100 mg and 400 mg tablets.
It is approved to treat:
- Newly diagnosed adult and pediatric patients with Philadelphia-chromosome-positive chronic myeloid leukemia (Ph+ CML) in chronic phase
- Patients with Ph+ CML in blast crisis, accelerated phase, or in chronic phase after failure of interferon-alpha therapy
- Adults with relapsed or refractory Ph+ acute lymphoblastic leukemia
- Adults with myelodysplastic/myeloproliferative diseases associated with PDGFR gene re-arrangements
- Adults with aggressive systemic mastocytosis without the D816V c-Kit mutation or with c-Kit mutational status unknown
- Adults with hypereosinophilic syndrome and/or chronic eosinophilic leukemia, including those who have the FIP1L1-PDGFRα fusion kinase
- Adult patients with unresectable, recurrent, and/or metastatic dermatofibrosarcoma protuberans.
Sun Pharma’s imatinib mesylate is not approved to treat patients with KIT (CD117)-positive unresectable and/or metastatic malignant gastrointestinal stromal tumors.

Photo by Rhoda Baer
Sun Pharma has announced the US launch of imatinib mesylate tablets, which are a generic version of Novartis’s Gleevec, for indications approved by the US Food and Drug Administration (FDA).
As part of this launch, Sun Pharma has rolled out a savings card program. The goal is to provide greater access to imatinib mesylate tablets for patients who have commercial insurance, but their out-of-pocket cost may exceed an affordable amount.
Sun Pharma’s Imatinib Mesylate Savings Card will reduce patient’s co-payment to $10. The card will also offer patients an additional savings benefit of up to $700 for a 30-day fill to offset any additional out-of-pocket cost should they be required to meet their deductible or co-insurance.
Participating pharmacies across the US can use the patient’s card as part of this program.
Eligible patients can participate in Sun Pharma’s Imatinib Mesylate Savings Card program by registering at www.imatinibrx.com or by requesting a savings card from their oncologist. Sun Pharma will be supplying its Imatinib Mesylate Savings Cards to more than 4500 oncologists.
Sun Pharma has established a Hub service so patients can call and speak with a trained healthcare professional about imatinib mesylate. The number is 1-844-502-5950.
In addition, qualifying patients can receive Sun Pharma’s imatinib mesylate at no cost. Based on qualifications for applying and including a doctor’s prescription, the Hub service will determine if a patient is qualified to receive imatinib mesylate for free. Upon acceptance, the prescription will be processed and delivered to the qualifying patient at no cost.
Sun Pharma’s imatinib mesylate was approved by the FDA in December 2015 and was granted 180 days of marketing exclusivity from the time of its launch. The drug is available in 100 mg and 400 mg tablets.
It is approved to treat:
- Newly diagnosed adult and pediatric patients with Philadelphia-chromosome-positive chronic myeloid leukemia (Ph+ CML) in chronic phase
- Patients with Ph+ CML in blast crisis, accelerated phase, or in chronic phase after failure of interferon-alpha therapy
- Adults with relapsed or refractory Ph+ acute lymphoblastic leukemia
- Adults with myelodysplastic/myeloproliferative diseases associated with PDGFR gene re-arrangements
- Adults with aggressive systemic mastocytosis without the D816V c-Kit mutation or with c-Kit mutational status unknown
- Adults with hypereosinophilic syndrome and/or chronic eosinophilic leukemia, including those who have the FIP1L1-PDGFRα fusion kinase
- Adult patients with unresectable, recurrent, and/or metastatic dermatofibrosarcoma protuberans.
Sun Pharma’s imatinib mesylate is not approved to treat patients with KIT (CD117)-positive unresectable and/or metastatic malignant gastrointestinal stromal tumors.