User login
Fruit fly findings may have implications for leukemia, other cancers
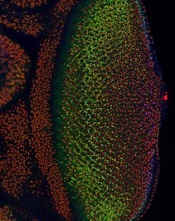
Image courtesy of
Northwestern University
In studying the fruit fly equivalent of an oncogene implicated in human leukemias, researchers have gained insight into how developing cells switch to a specialized state and how that process might go awry in cancers.
The team found that levels of the protein Yan start fluctuating wildly when a cell is switching from a stem-like state to a more specialized state. If the levels of Yan don’t or can’t fluctuate, the cell doesn’t differentiate.
The Yan protein is called Tel-1 in humans, and the gene that produces the Tel-1 protein, the Tel-1 oncogene, is frequently mutated in human leukemias.
Richard W. Carthew, PhD, of Northwestern University in Evanston, Illinois, and his colleagues conducted this research and reported the results in eLife.
The researchers studied cell behavior in the eye of Drosophila melanogaster, the common fruit fly, which has many of the same oncogenes as humans.
The team was surprised to discover that fluctuating levels of Yan were needed for cell differentiation.
“This mad fluctuation, or noise, happens at the time of cell transition,” Dr Carthew explained. “For the first time, we see there is a brief time period as the developing cell goes from point A to point B. The noise is a state of ‘in between’ and is important for cells to switch to a more specialized state. This limbo might be where normal cells take a cancerous path.”
He noted that it takes 15 to 20 hours for a fruit fly cell to transition from an unspecialized to a specialized state. The researchers found the Yan protein is “noisy,” or fluctuating, for 6 to 8 of those hours.
The team also found that a molecular signal received by the cell receptor EGFR is important for turning the noise off. If that signal is not received, the cell remains in an uncontrolled state.
The EGFR protein that turns off the noise in flies is called Her-2 in humans, and the Her-2 oncogene is known to play an important role in breast cancer.
“On the surface, flies and humans are very different, but we share a remarkable amount of infrastructure,” Dr Carthew noted. “We can use fruit fly genetics to understand how humans work and how things go wrong in cancer and other diseases.” ![]()

Image courtesy of
Northwestern University
In studying the fruit fly equivalent of an oncogene implicated in human leukemias, researchers have gained insight into how developing cells switch to a specialized state and how that process might go awry in cancers.
The team found that levels of the protein Yan start fluctuating wildly when a cell is switching from a stem-like state to a more specialized state. If the levels of Yan don’t or can’t fluctuate, the cell doesn’t differentiate.
The Yan protein is called Tel-1 in humans, and the gene that produces the Tel-1 protein, the Tel-1 oncogene, is frequently mutated in human leukemias.
Richard W. Carthew, PhD, of Northwestern University in Evanston, Illinois, and his colleagues conducted this research and reported the results in eLife.
The researchers studied cell behavior in the eye of Drosophila melanogaster, the common fruit fly, which has many of the same oncogenes as humans.
The team was surprised to discover that fluctuating levels of Yan were needed for cell differentiation.
“This mad fluctuation, or noise, happens at the time of cell transition,” Dr Carthew explained. “For the first time, we see there is a brief time period as the developing cell goes from point A to point B. The noise is a state of ‘in between’ and is important for cells to switch to a more specialized state. This limbo might be where normal cells take a cancerous path.”
He noted that it takes 15 to 20 hours for a fruit fly cell to transition from an unspecialized to a specialized state. The researchers found the Yan protein is “noisy,” or fluctuating, for 6 to 8 of those hours.
The team also found that a molecular signal received by the cell receptor EGFR is important for turning the noise off. If that signal is not received, the cell remains in an uncontrolled state.
The EGFR protein that turns off the noise in flies is called Her-2 in humans, and the Her-2 oncogene is known to play an important role in breast cancer.
“On the surface, flies and humans are very different, but we share a remarkable amount of infrastructure,” Dr Carthew noted. “We can use fruit fly genetics to understand how humans work and how things go wrong in cancer and other diseases.” ![]()

Image courtesy of
Northwestern University
In studying the fruit fly equivalent of an oncogene implicated in human leukemias, researchers have gained insight into how developing cells switch to a specialized state and how that process might go awry in cancers.
The team found that levels of the protein Yan start fluctuating wildly when a cell is switching from a stem-like state to a more specialized state. If the levels of Yan don’t or can’t fluctuate, the cell doesn’t differentiate.
The Yan protein is called Tel-1 in humans, and the gene that produces the Tel-1 protein, the Tel-1 oncogene, is frequently mutated in human leukemias.
Richard W. Carthew, PhD, of Northwestern University in Evanston, Illinois, and his colleagues conducted this research and reported the results in eLife.
The researchers studied cell behavior in the eye of Drosophila melanogaster, the common fruit fly, which has many of the same oncogenes as humans.
The team was surprised to discover that fluctuating levels of Yan were needed for cell differentiation.
“This mad fluctuation, or noise, happens at the time of cell transition,” Dr Carthew explained. “For the first time, we see there is a brief time period as the developing cell goes from point A to point B. The noise is a state of ‘in between’ and is important for cells to switch to a more specialized state. This limbo might be where normal cells take a cancerous path.”
He noted that it takes 15 to 20 hours for a fruit fly cell to transition from an unspecialized to a specialized state. The researchers found the Yan protein is “noisy,” or fluctuating, for 6 to 8 of those hours.
The team also found that a molecular signal received by the cell receptor EGFR is important for turning the noise off. If that signal is not received, the cell remains in an uncontrolled state.
The EGFR protein that turns off the noise in flies is called Her-2 in humans, and the Her-2 oncogene is known to play an important role in breast cancer.
“On the surface, flies and humans are very different, but we share a remarkable amount of infrastructure,” Dr Carthew noted. “We can use fruit fly genetics to understand how humans work and how things go wrong in cancer and other diseases.” ![]()
How p53 and telomeres stave off cancer

telomeres in green
Image by Claus Azzalin
Research published in The EMBO Journal suggests that p53 has tumor suppressor functions related to telomeres.
The study showed, for the first time, that p53 can suppress accumulated DNA damage at telomeres.
P53 is known to regulate the genome’s integrity. When DNA is damaged, p53 helps activate the transcription of genes that regulate the cell cycle and induce apoptosis.
However, prior studies have shown that p53 can bind at many locations across the genome, including sites that are not responsible for activating these regulatory genes.
Paul Lieberman, PhD, of The Wistar Institute in Philadelphia, Pennsylvania, and his colleagues decided to study these binding sites to see if p53 and telomeres might be more closely related than previous research suggested.
“We believed that p53 may be responsible for a more direct protective effect in telomeres,” Dr Lieberman said.
Using ChIP-sequencing, he and his colleagues identified p53 binding sites in subtelomeres.
They found that when p53 was bound to subtelomeres, the protein was able to suppress the formation of a histone modification called γ-H2AX.
This histone is modified in greater amounts when there is a double-strand break on DNA. If it persists, the break is not repaired, so suppressing its expression means the DNA is being preserved.
Additionally, p53 was able to prevent DNA degradation in telomeres, thereby keeping them intact and allowing them to more properly protect the tips of chromosomes.
“Based on our findings, we propose that the modifications to chromatin made by p53 enhance local DNA repair or protection,” Dr Lieberman said. “This would be yet another tumor suppressor function of p53, thus providing additional framework for just how important this gene is in protecting us from cancer.” ![]()

telomeres in green
Image by Claus Azzalin
Research published in The EMBO Journal suggests that p53 has tumor suppressor functions related to telomeres.
The study showed, for the first time, that p53 can suppress accumulated DNA damage at telomeres.
P53 is known to regulate the genome’s integrity. When DNA is damaged, p53 helps activate the transcription of genes that regulate the cell cycle and induce apoptosis.
However, prior studies have shown that p53 can bind at many locations across the genome, including sites that are not responsible for activating these regulatory genes.
Paul Lieberman, PhD, of The Wistar Institute in Philadelphia, Pennsylvania, and his colleagues decided to study these binding sites to see if p53 and telomeres might be more closely related than previous research suggested.
“We believed that p53 may be responsible for a more direct protective effect in telomeres,” Dr Lieberman said.
Using ChIP-sequencing, he and his colleagues identified p53 binding sites in subtelomeres.
They found that when p53 was bound to subtelomeres, the protein was able to suppress the formation of a histone modification called γ-H2AX.
This histone is modified in greater amounts when there is a double-strand break on DNA. If it persists, the break is not repaired, so suppressing its expression means the DNA is being preserved.
Additionally, p53 was able to prevent DNA degradation in telomeres, thereby keeping them intact and allowing them to more properly protect the tips of chromosomes.
“Based on our findings, we propose that the modifications to chromatin made by p53 enhance local DNA repair or protection,” Dr Lieberman said. “This would be yet another tumor suppressor function of p53, thus providing additional framework for just how important this gene is in protecting us from cancer.” ![]()

telomeres in green
Image by Claus Azzalin
Research published in The EMBO Journal suggests that p53 has tumor suppressor functions related to telomeres.
The study showed, for the first time, that p53 can suppress accumulated DNA damage at telomeres.
P53 is known to regulate the genome’s integrity. When DNA is damaged, p53 helps activate the transcription of genes that regulate the cell cycle and induce apoptosis.
However, prior studies have shown that p53 can bind at many locations across the genome, including sites that are not responsible for activating these regulatory genes.
Paul Lieberman, PhD, of The Wistar Institute in Philadelphia, Pennsylvania, and his colleagues decided to study these binding sites to see if p53 and telomeres might be more closely related than previous research suggested.
“We believed that p53 may be responsible for a more direct protective effect in telomeres,” Dr Lieberman said.
Using ChIP-sequencing, he and his colleagues identified p53 binding sites in subtelomeres.
They found that when p53 was bound to subtelomeres, the protein was able to suppress the formation of a histone modification called γ-H2AX.
This histone is modified in greater amounts when there is a double-strand break on DNA. If it persists, the break is not repaired, so suppressing its expression means the DNA is being preserved.
Additionally, p53 was able to prevent DNA degradation in telomeres, thereby keeping them intact and allowing them to more properly protect the tips of chromosomes.
“Based on our findings, we propose that the modifications to chromatin made by p53 enhance local DNA repair or protection,” Dr Lieberman said. “This would be yet another tumor suppressor function of p53, thus providing additional framework for just how important this gene is in protecting us from cancer.” ![]()
MRD test can predict AML relapse
Photo by William Weinert
A test measuring minimal residual disease (MRD) can help predict relapse in patients with acute myeloid leukemia (AML), according to a study published in NEJM.
Investigators used the test to detect MRD in samples from patients with NPM1-mutated AML who were deemed standard-risk by conventional methods.
The team said the presence of MRD after treatment provided powerful prognostic information independent of other risk factors.
“What we have been able to identify is a group of patients who otherwise would be thought to do quite well, who, in fact, have a very poor prognosis, and who are not well served currently,” said study author Robert Hills, DPhil, of Cardiff University School of Medicine in the UK.
“This opens up the exciting prospect that we can do the same for other groups of patients as well.”
For this study, Dr Hills and his colleagues used a reverse-transcriptase quantitative polymerase-chain-reaction assay to detect MRD in 2569 samples (902 bone marrow samples and 1667 peripheral blood samples) from 346 patients with NPM1-mutated AML.
The patients had received 2 cycles of chemotherapy as part of the National Cancer Research Institute AML17 trial. They were treated at centers in the UK, Denmark, and New Zealand. All patients were shown to be at standard risk of relapse using conventional tests.
The investigators found that NPM1-mutated transcripts persisted in the blood of 15% of patients after the second cycle of chemotherapy.
This finding was associated with a greater risk of relapse after 3 years of follow-up.
Eighty-two percent of patients with NPM1-mutated transcripts had relapsed within 3 years, compared to 30% of patients who had no detectable NPM1. The hazard ratio was 4.80 (P<0.001).
Patients with traces of NPM1 also had a lower rate of survival—24%, compared to 75% for patients without detectable NPM1. The hazard ratio was 4.38 (P<0.001).
In multivariate analysis, the presence of MRD was the only significant prognostic factor for relapse or death. The hazard ratios were 5.09 and 4.84, respectively (P<0.001 for both).
The investigators validated these findings in a cohort of 91 patients with NPM1-mutated AML.
The presence of MRD in the blood of these patients was associated with a higher cumulative incidence of relapse—70% vs 31% (P=0.001)—and a lower rate of survival—40% vs 87% (P=0.001)—at 2 years.
The investigators also found that, with sequential monitoring of MRD, a rising level of NPM1-mutated transcripts could predict relapse.
“Conventional methods for guiding treatment for this aggressive type of leukemia are inadequate,” said study author David Grimwade, PhD, of King’s College London in the UK.
“The MRD test is an invaluable tool to assess treatment response and identify those patients for whom chemotherapy is not sufficient and require stem cell transplantation or new treatments.” ![]()

Photo by William Weinert
A test measuring minimal residual disease (MRD) can help predict relapse in patients with acute myeloid leukemia (AML), according to a study published in NEJM.
Investigators used the test to detect MRD in samples from patients with NPM1-mutated AML who were deemed standard-risk by conventional methods.
The team said the presence of MRD after treatment provided powerful prognostic information independent of other risk factors.
“What we have been able to identify is a group of patients who otherwise would be thought to do quite well, who, in fact, have a very poor prognosis, and who are not well served currently,” said study author Robert Hills, DPhil, of Cardiff University School of Medicine in the UK.
“This opens up the exciting prospect that we can do the same for other groups of patients as well.”
For this study, Dr Hills and his colleagues used a reverse-transcriptase quantitative polymerase-chain-reaction assay to detect MRD in 2569 samples (902 bone marrow samples and 1667 peripheral blood samples) from 346 patients with NPM1-mutated AML.
The patients had received 2 cycles of chemotherapy as part of the National Cancer Research Institute AML17 trial. They were treated at centers in the UK, Denmark, and New Zealand. All patients were shown to be at standard risk of relapse using conventional tests.
The investigators found that NPM1-mutated transcripts persisted in the blood of 15% of patients after the second cycle of chemotherapy.
This finding was associated with a greater risk of relapse after 3 years of follow-up.
Eighty-two percent of patients with NPM1-mutated transcripts had relapsed within 3 years, compared to 30% of patients who had no detectable NPM1. The hazard ratio was 4.80 (P<0.001).
Patients with traces of NPM1 also had a lower rate of survival—24%, compared to 75% for patients without detectable NPM1. The hazard ratio was 4.38 (P<0.001).
In multivariate analysis, the presence of MRD was the only significant prognostic factor for relapse or death. The hazard ratios were 5.09 and 4.84, respectively (P<0.001 for both).
The investigators validated these findings in a cohort of 91 patients with NPM1-mutated AML.
The presence of MRD in the blood of these patients was associated with a higher cumulative incidence of relapse—70% vs 31% (P=0.001)—and a lower rate of survival—40% vs 87% (P=0.001)—at 2 years.
The investigators also found that, with sequential monitoring of MRD, a rising level of NPM1-mutated transcripts could predict relapse.
“Conventional methods for guiding treatment for this aggressive type of leukemia are inadequate,” said study author David Grimwade, PhD, of King’s College London in the UK.
“The MRD test is an invaluable tool to assess treatment response and identify those patients for whom chemotherapy is not sufficient and require stem cell transplantation or new treatments.” ![]()

Photo by William Weinert
A test measuring minimal residual disease (MRD) can help predict relapse in patients with acute myeloid leukemia (AML), according to a study published in NEJM.
Investigators used the test to detect MRD in samples from patients with NPM1-mutated AML who were deemed standard-risk by conventional methods.
The team said the presence of MRD after treatment provided powerful prognostic information independent of other risk factors.
“What we have been able to identify is a group of patients who otherwise would be thought to do quite well, who, in fact, have a very poor prognosis, and who are not well served currently,” said study author Robert Hills, DPhil, of Cardiff University School of Medicine in the UK.
“This opens up the exciting prospect that we can do the same for other groups of patients as well.”
For this study, Dr Hills and his colleagues used a reverse-transcriptase quantitative polymerase-chain-reaction assay to detect MRD in 2569 samples (902 bone marrow samples and 1667 peripheral blood samples) from 346 patients with NPM1-mutated AML.
The patients had received 2 cycles of chemotherapy as part of the National Cancer Research Institute AML17 trial. They were treated at centers in the UK, Denmark, and New Zealand. All patients were shown to be at standard risk of relapse using conventional tests.
The investigators found that NPM1-mutated transcripts persisted in the blood of 15% of patients after the second cycle of chemotherapy.
This finding was associated with a greater risk of relapse after 3 years of follow-up.
Eighty-two percent of patients with NPM1-mutated transcripts had relapsed within 3 years, compared to 30% of patients who had no detectable NPM1. The hazard ratio was 4.80 (P<0.001).
Patients with traces of NPM1 also had a lower rate of survival—24%, compared to 75% for patients without detectable NPM1. The hazard ratio was 4.38 (P<0.001).
In multivariate analysis, the presence of MRD was the only significant prognostic factor for relapse or death. The hazard ratios were 5.09 and 4.84, respectively (P<0.001 for both).
The investigators validated these findings in a cohort of 91 patients with NPM1-mutated AML.
The presence of MRD in the blood of these patients was associated with a higher cumulative incidence of relapse—70% vs 31% (P=0.001)—and a lower rate of survival—40% vs 87% (P=0.001)—at 2 years.
The investigators also found that, with sequential monitoring of MRD, a rising level of NPM1-mutated transcripts could predict relapse.
“Conventional methods for guiding treatment for this aggressive type of leukemia are inadequate,” said study author David Grimwade, PhD, of King’s College London in the UK.
“The MRD test is an invaluable tool to assess treatment response and identify those patients for whom chemotherapy is not sufficient and require stem cell transplantation or new treatments.” ![]()
Fertility concerns of AYA cancer survivors

patient and her father
Photo by Rhoda Baer
Results of a small study suggest that adolescent and young adult (AYA) cancer survivors have a range of concerns regarding their fertility.
The female cancer survivors studied were more likely than males to describe feeling distressed and overwhelmed about their fertility.
Females also tended to worry about pregnancy-related health risks and cancer recurrence.
However, AYA cancer survivors of both sexes expressed concerns about genetic risk factors and how infertility might impact their future lives.
Catherine Benedict, PhD, of Memorial Sloan Kettering Cancer Center in New York, New York, and her colleagues conducted this research and reported the results in the Journal of Adolescent and Young Adult Oncology.
The researchers assessed fertility concerns in 43 AYA cancer survivors. They were 16 to 24 at the time of the study, had been diagnosed with cancer between ages 14 and 18, and were at least 6 months post-treatment.
The subjects either completed an individual interview (n=26) or participated in 1 of 4 focus groups (n=17).
Before treatment, 5 of the males had banked sperm, but none of the females took steps to preserve their fertility. More males (50%) than females (39%) reported uncertainty about their fertility.
The researchers identified 3 themes when discussing fertility with the study subjects: fertility concerns, emotions raised when discussing fertility, and strategies used to manage fertility concerns.
Concerns
Some subjects expressed concerns about how potential infertility could affect dating and relationships with partners (8% of males and 20% of females).
Some subjects were concerned about the health risks associated with having children—both risks to the subjects themselves and to any potential children (39% of males and 30% of females).
And some subjects were concerned about how potential infertility would affect their lives going forward (31% of males and 20% of females).
Emotions
When it came to emotions associated with fertility discussions, some subjects said they felt distressed and overwhelmed (23% of males and 30% of females).
However, some subjects said they weren’t concerned about their fertility (31% of males and 15% of females) or they felt hopeful despite the risk of infertility (8% of males and 20% of females).
Managing concerns
The subjects also mentioned a few strategies for managing fertility concerns. Some said they had accepted infertility (23% of males and 15% of females).
Some subjects said they weren’t going to worry about their fertility until they were older and actually wanted to have children (23% of males and 20% of females).
And some subjects said they would rely on assisted reproductive technology if necessary (31% of males and 20% of females).
Dr Benedict and her colleagues said this study suggests AYA cancer survivors may have a number of reproductive concerns and fertility-related distress, which may affect other areas of psychosocial functioning.
So future research should explore how to best incorporate fertility-related informational and support services more fully into survivorship care. ![]()

patient and her father
Photo by Rhoda Baer
Results of a small study suggest that adolescent and young adult (AYA) cancer survivors have a range of concerns regarding their fertility.
The female cancer survivors studied were more likely than males to describe feeling distressed and overwhelmed about their fertility.
Females also tended to worry about pregnancy-related health risks and cancer recurrence.
However, AYA cancer survivors of both sexes expressed concerns about genetic risk factors and how infertility might impact their future lives.
Catherine Benedict, PhD, of Memorial Sloan Kettering Cancer Center in New York, New York, and her colleagues conducted this research and reported the results in the Journal of Adolescent and Young Adult Oncology.
The researchers assessed fertility concerns in 43 AYA cancer survivors. They were 16 to 24 at the time of the study, had been diagnosed with cancer between ages 14 and 18, and were at least 6 months post-treatment.
The subjects either completed an individual interview (n=26) or participated in 1 of 4 focus groups (n=17).
Before treatment, 5 of the males had banked sperm, but none of the females took steps to preserve their fertility. More males (50%) than females (39%) reported uncertainty about their fertility.
The researchers identified 3 themes when discussing fertility with the study subjects: fertility concerns, emotions raised when discussing fertility, and strategies used to manage fertility concerns.
Concerns
Some subjects expressed concerns about how potential infertility could affect dating and relationships with partners (8% of males and 20% of females).
Some subjects were concerned about the health risks associated with having children—both risks to the subjects themselves and to any potential children (39% of males and 30% of females).
And some subjects were concerned about how potential infertility would affect their lives going forward (31% of males and 20% of females).
Emotions
When it came to emotions associated with fertility discussions, some subjects said they felt distressed and overwhelmed (23% of males and 30% of females).
However, some subjects said they weren’t concerned about their fertility (31% of males and 15% of females) or they felt hopeful despite the risk of infertility (8% of males and 20% of females).
Managing concerns
The subjects also mentioned a few strategies for managing fertility concerns. Some said they had accepted infertility (23% of males and 15% of females).
Some subjects said they weren’t going to worry about their fertility until they were older and actually wanted to have children (23% of males and 20% of females).
And some subjects said they would rely on assisted reproductive technology if necessary (31% of males and 20% of females).
Dr Benedict and her colleagues said this study suggests AYA cancer survivors may have a number of reproductive concerns and fertility-related distress, which may affect other areas of psychosocial functioning.
So future research should explore how to best incorporate fertility-related informational and support services more fully into survivorship care. ![]()

patient and her father
Photo by Rhoda Baer
Results of a small study suggest that adolescent and young adult (AYA) cancer survivors have a range of concerns regarding their fertility.
The female cancer survivors studied were more likely than males to describe feeling distressed and overwhelmed about their fertility.
Females also tended to worry about pregnancy-related health risks and cancer recurrence.
However, AYA cancer survivors of both sexes expressed concerns about genetic risk factors and how infertility might impact their future lives.
Catherine Benedict, PhD, of Memorial Sloan Kettering Cancer Center in New York, New York, and her colleagues conducted this research and reported the results in the Journal of Adolescent and Young Adult Oncology.
The researchers assessed fertility concerns in 43 AYA cancer survivors. They were 16 to 24 at the time of the study, had been diagnosed with cancer between ages 14 and 18, and were at least 6 months post-treatment.
The subjects either completed an individual interview (n=26) or participated in 1 of 4 focus groups (n=17).
Before treatment, 5 of the males had banked sperm, but none of the females took steps to preserve their fertility. More males (50%) than females (39%) reported uncertainty about their fertility.
The researchers identified 3 themes when discussing fertility with the study subjects: fertility concerns, emotions raised when discussing fertility, and strategies used to manage fertility concerns.
Concerns
Some subjects expressed concerns about how potential infertility could affect dating and relationships with partners (8% of males and 20% of females).
Some subjects were concerned about the health risks associated with having children—both risks to the subjects themselves and to any potential children (39% of males and 30% of females).
And some subjects were concerned about how potential infertility would affect their lives going forward (31% of males and 20% of females).
Emotions
When it came to emotions associated with fertility discussions, some subjects said they felt distressed and overwhelmed (23% of males and 30% of females).
However, some subjects said they weren’t concerned about their fertility (31% of males and 15% of females) or they felt hopeful despite the risk of infertility (8% of males and 20% of females).
Managing concerns
The subjects also mentioned a few strategies for managing fertility concerns. Some said they had accepted infertility (23% of males and 15% of females).
Some subjects said they weren’t going to worry about their fertility until they were older and actually wanted to have children (23% of males and 20% of females).
And some subjects said they would rely on assisted reproductive technology if necessary (31% of males and 20% of females).
Dr Benedict and her colleagues said this study suggests AYA cancer survivors may have a number of reproductive concerns and fertility-related distress, which may affect other areas of psychosocial functioning.
So future research should explore how to best incorporate fertility-related informational and support services more fully into survivorship care. ![]()
Gene therapy could treat FVII deficiency

Photo by Julien Valroff
Gene therapy can safely and effectively treat factor VII (FVII) deficiency in dogs, according to research published in Blood.
Three of the 4 dogs treated expressed levels of FVII that would be therapeutic in humans, with long-term stability.
In one dog, the effects have persisted for more than 2.5 years and are ongoing.
In addition, researchers said the therapy appears safe, based on kidney function, liver function, and blood measurements.
The team believes this success in large animals holds considerable potential for a safe, effective, and long-lasting new treatment in humans with FVII deficiency.
“Our finding has great clinical relevance for patients with FVII deficiency,” said Paris Margaritis, DPhil, of The Children’s Hospital of Philadelphia in Pennsylvania.
“These dogs have the type of mutation found in the majority of patients with this disorder, so this approach could lead to a sustained gene therapy in people.”
Dr Margaritis and his colleagues studied dogs with a G96E missense FVII mutation (FVII-G96E), which have less than 1% FVII activity. The researchers said these dogs had undetectable plasmatic antigen by Western blot and therefore represent the most prevalent type of human FVII deficiency.
“We developed a unique animal model of this disease after identifying dogs with naturally occurring FVII deficiency,” Dr Margaritis said. “Our investigations enabled us to design the corrective gene to insert into our virus vector in the current study.”
The researchers tested liver-directed, adeno-associated viral (AAV) serotype 8 vector delivery of a canine FVII zymogen transgene (cFVII) in 4 FVII-G96E dogs, known as Otis, N22, N24, and N25.
The animals received escalating doses of AAV8-cFVII, ranging from 2E11 vg/kg to 4.95E13 vg/kg.
Efficacy
At baseline, all dogs had less than 1% cFVII activity levels. After receiving AAV8-cFVII, they experienced a reduction in prothrombin time that reached a plateau:
- From 121.1± 1.0s to 22.1 ± 3.2s in N24 (4.95E13 vg/kg)
- From 83.5 ± 18.4s to 25.0 ± 3.6s in N22 (2E12 vg/kg)
- From 96.0 ± 5.8s to 28.7 ± 3.7s in N25 (6E11 vg/kg)
- From 124.8 ± 4.6s to 50.4 ± 4.0s in Otis (2E11 vg/kg).
The researchers also observed an increase in cFVII expression for all of the dogs but Otis. Levels of cFVII antigen reached plateaus of:
- 8320 ± 1800 ng/ml in N24, which is 770 ± 167 percent of normal
- 307 ± 56 ng/ml in N22, which is 29.7 ± 6.2 percent of normal
- 170 ± 62 ng/ml in N25, which is 15.7 ± 5.7 percent of normal.
These clinically therapeutic cFVII antigen levels have, thus far, persisted for more than 1 year in N22 and N25 and for 2.6 years in N24.
Safety
The researchers found that measures of liver and kidney function were within the normal range in all of the dogs. Likewise, D-dimer and fibrinogen levels were within the normal range.
The team said these results suggest that continuous cFVII expression—even in N24, which averaged 8.3 μg/ml for 2.6 years—did not result in evident activation of the endogenous coagulation system or induce a chronic pathological state.
N24 did exhibit a transient elevation of anti-cFVII IgG2. However, this response was not inhibitory and did not affect the transgene expression plateau.
The researchers did not detect anti-cFVII IgG2 in the other 3 dogs, nor did the team detect anti-cFVII IgG1 in any of the dogs studied.
“This work is very exciting and promising,” said study author Timothy Nichols, MD, of The University of North Carolina at Chapel Hill.
“The FVII-deficient dogs tolerated the initial gene therapy infusions very well and have had no adverse side effects over several years of follow-up. In other related studies in dogs with hemophilia B, similar positive findings have translated to people with hemophilia B.” ![]()

Photo by Julien Valroff
Gene therapy can safely and effectively treat factor VII (FVII) deficiency in dogs, according to research published in Blood.
Three of the 4 dogs treated expressed levels of FVII that would be therapeutic in humans, with long-term stability.
In one dog, the effects have persisted for more than 2.5 years and are ongoing.
In addition, researchers said the therapy appears safe, based on kidney function, liver function, and blood measurements.
The team believes this success in large animals holds considerable potential for a safe, effective, and long-lasting new treatment in humans with FVII deficiency.
“Our finding has great clinical relevance for patients with FVII deficiency,” said Paris Margaritis, DPhil, of The Children’s Hospital of Philadelphia in Pennsylvania.
“These dogs have the type of mutation found in the majority of patients with this disorder, so this approach could lead to a sustained gene therapy in people.”
Dr Margaritis and his colleagues studied dogs with a G96E missense FVII mutation (FVII-G96E), which have less than 1% FVII activity. The researchers said these dogs had undetectable plasmatic antigen by Western blot and therefore represent the most prevalent type of human FVII deficiency.
“We developed a unique animal model of this disease after identifying dogs with naturally occurring FVII deficiency,” Dr Margaritis said. “Our investigations enabled us to design the corrective gene to insert into our virus vector in the current study.”
The researchers tested liver-directed, adeno-associated viral (AAV) serotype 8 vector delivery of a canine FVII zymogen transgene (cFVII) in 4 FVII-G96E dogs, known as Otis, N22, N24, and N25.
The animals received escalating doses of AAV8-cFVII, ranging from 2E11 vg/kg to 4.95E13 vg/kg.
Efficacy
At baseline, all dogs had less than 1% cFVII activity levels. After receiving AAV8-cFVII, they experienced a reduction in prothrombin time that reached a plateau:
- From 121.1± 1.0s to 22.1 ± 3.2s in N24 (4.95E13 vg/kg)
- From 83.5 ± 18.4s to 25.0 ± 3.6s in N22 (2E12 vg/kg)
- From 96.0 ± 5.8s to 28.7 ± 3.7s in N25 (6E11 vg/kg)
- From 124.8 ± 4.6s to 50.4 ± 4.0s in Otis (2E11 vg/kg).
The researchers also observed an increase in cFVII expression for all of the dogs but Otis. Levels of cFVII antigen reached plateaus of:
- 8320 ± 1800 ng/ml in N24, which is 770 ± 167 percent of normal
- 307 ± 56 ng/ml in N22, which is 29.7 ± 6.2 percent of normal
- 170 ± 62 ng/ml in N25, which is 15.7 ± 5.7 percent of normal.
These clinically therapeutic cFVII antigen levels have, thus far, persisted for more than 1 year in N22 and N25 and for 2.6 years in N24.
Safety
The researchers found that measures of liver and kidney function were within the normal range in all of the dogs. Likewise, D-dimer and fibrinogen levels were within the normal range.
The team said these results suggest that continuous cFVII expression—even in N24, which averaged 8.3 μg/ml for 2.6 years—did not result in evident activation of the endogenous coagulation system or induce a chronic pathological state.
N24 did exhibit a transient elevation of anti-cFVII IgG2. However, this response was not inhibitory and did not affect the transgene expression plateau.
The researchers did not detect anti-cFVII IgG2 in the other 3 dogs, nor did the team detect anti-cFVII IgG1 in any of the dogs studied.
“This work is very exciting and promising,” said study author Timothy Nichols, MD, of The University of North Carolina at Chapel Hill.
“The FVII-deficient dogs tolerated the initial gene therapy infusions very well and have had no adverse side effects over several years of follow-up. In other related studies in dogs with hemophilia B, similar positive findings have translated to people with hemophilia B.” ![]()

Photo by Julien Valroff
Gene therapy can safely and effectively treat factor VII (FVII) deficiency in dogs, according to research published in Blood.
Three of the 4 dogs treated expressed levels of FVII that would be therapeutic in humans, with long-term stability.
In one dog, the effects have persisted for more than 2.5 years and are ongoing.
In addition, researchers said the therapy appears safe, based on kidney function, liver function, and blood measurements.
The team believes this success in large animals holds considerable potential for a safe, effective, and long-lasting new treatment in humans with FVII deficiency.
“Our finding has great clinical relevance for patients with FVII deficiency,” said Paris Margaritis, DPhil, of The Children’s Hospital of Philadelphia in Pennsylvania.
“These dogs have the type of mutation found in the majority of patients with this disorder, so this approach could lead to a sustained gene therapy in people.”
Dr Margaritis and his colleagues studied dogs with a G96E missense FVII mutation (FVII-G96E), which have less than 1% FVII activity. The researchers said these dogs had undetectable plasmatic antigen by Western blot and therefore represent the most prevalent type of human FVII deficiency.
“We developed a unique animal model of this disease after identifying dogs with naturally occurring FVII deficiency,” Dr Margaritis said. “Our investigations enabled us to design the corrective gene to insert into our virus vector in the current study.”
The researchers tested liver-directed, adeno-associated viral (AAV) serotype 8 vector delivery of a canine FVII zymogen transgene (cFVII) in 4 FVII-G96E dogs, known as Otis, N22, N24, and N25.
The animals received escalating doses of AAV8-cFVII, ranging from 2E11 vg/kg to 4.95E13 vg/kg.
Efficacy
At baseline, all dogs had less than 1% cFVII activity levels. After receiving AAV8-cFVII, they experienced a reduction in prothrombin time that reached a plateau:
- From 121.1± 1.0s to 22.1 ± 3.2s in N24 (4.95E13 vg/kg)
- From 83.5 ± 18.4s to 25.0 ± 3.6s in N22 (2E12 vg/kg)
- From 96.0 ± 5.8s to 28.7 ± 3.7s in N25 (6E11 vg/kg)
- From 124.8 ± 4.6s to 50.4 ± 4.0s in Otis (2E11 vg/kg).
The researchers also observed an increase in cFVII expression for all of the dogs but Otis. Levels of cFVII antigen reached plateaus of:
- 8320 ± 1800 ng/ml in N24, which is 770 ± 167 percent of normal
- 307 ± 56 ng/ml in N22, which is 29.7 ± 6.2 percent of normal
- 170 ± 62 ng/ml in N25, which is 15.7 ± 5.7 percent of normal.
These clinically therapeutic cFVII antigen levels have, thus far, persisted for more than 1 year in N22 and N25 and for 2.6 years in N24.
Safety
The researchers found that measures of liver and kidney function were within the normal range in all of the dogs. Likewise, D-dimer and fibrinogen levels were within the normal range.
The team said these results suggest that continuous cFVII expression—even in N24, which averaged 8.3 μg/ml for 2.6 years—did not result in evident activation of the endogenous coagulation system or induce a chronic pathological state.
N24 did exhibit a transient elevation of anti-cFVII IgG2. However, this response was not inhibitory and did not affect the transgene expression plateau.
The researchers did not detect anti-cFVII IgG2 in the other 3 dogs, nor did the team detect anti-cFVII IgG1 in any of the dogs studied.
“This work is very exciting and promising,” said study author Timothy Nichols, MD, of The University of North Carolina at Chapel Hill.
“The FVII-deficient dogs tolerated the initial gene therapy infusions very well and have had no adverse side effects over several years of follow-up. In other related studies in dogs with hemophilia B, similar positive findings have translated to people with hemophilia B.” ![]()
FDA expands approved use of MM drug

Photo courtesy of Amgen
The US Food and Drug Administration (FDA) has approved the use of carfilzomib (Kyprolis) in combination with dexamethasone or lenalidomide plus dexamethasone to treat patients with relapsed or refractory multiple myeloma (MM) who have received 1 to 3 prior lines of therapy.
The FDA also approved use of carfilzomib as a single agent to treat patients with relapsed or refractory MM who have received 1 or more lines of therapy.
This decision converts to full approval the initial accelerated approval granted for single-agent carfilzomib in July 2012.
In July 2015, the FDA approved the use of carfilzomib in combination with lenalidomide and dexamethasone for the treatment of patients with relapsed MM who have received 1 to 3 prior lines of therapy.
The latest approval of carfilzomib is based on results from the phase 3 ENDEAVOR study.
Trial results
The ENDEAVOR trial included 929 MM patients whose disease had relapsed after 1 to 3 prior therapeutic regimens. The patients received either carfilzomib plus dexamethasone (n=464) or bortezomib plus dexamethasone (n=465) until disease progression.
The primary endpoint was progression-free survival. The median was 18.7 months in the carfilzomib arm and 9.4 months in the bortezomib arm. The hazard ratio was 0.53 (P<0.0001).
Overall survival data are not yet mature and continue to be monitored.
Treatment discontinuation due to adverse events and on-study deaths were comparable between the 2 treatment arms.
However, a number of known adverse events were reported at a higher rate in the carfilzomib arm than the bortezomib arm, including any-grade dyspnea (28% vs 13%), hypertension (25% vs 3%), pyrexia (27% vs 14%), cough (25% vs 15%), cardiac failure (8% vs 3%), and acute renal failure (8% vs 5%).
Carfilzomib is marketed as Kyprolis by Onyx Pharmaceuticals, Inc., a subsidiary of Amgen that holds development and commercialization rights to the drug globally, with the exception of Japan. For more information on the drug, visit www.kyprolis.com. ![]()

Photo courtesy of Amgen
The US Food and Drug Administration (FDA) has approved the use of carfilzomib (Kyprolis) in combination with dexamethasone or lenalidomide plus dexamethasone to treat patients with relapsed or refractory multiple myeloma (MM) who have received 1 to 3 prior lines of therapy.
The FDA also approved use of carfilzomib as a single agent to treat patients with relapsed or refractory MM who have received 1 or more lines of therapy.
This decision converts to full approval the initial accelerated approval granted for single-agent carfilzomib in July 2012.
In July 2015, the FDA approved the use of carfilzomib in combination with lenalidomide and dexamethasone for the treatment of patients with relapsed MM who have received 1 to 3 prior lines of therapy.
The latest approval of carfilzomib is based on results from the phase 3 ENDEAVOR study.
Trial results
The ENDEAVOR trial included 929 MM patients whose disease had relapsed after 1 to 3 prior therapeutic regimens. The patients received either carfilzomib plus dexamethasone (n=464) or bortezomib plus dexamethasone (n=465) until disease progression.
The primary endpoint was progression-free survival. The median was 18.7 months in the carfilzomib arm and 9.4 months in the bortezomib arm. The hazard ratio was 0.53 (P<0.0001).
Overall survival data are not yet mature and continue to be monitored.
Treatment discontinuation due to adverse events and on-study deaths were comparable between the 2 treatment arms.
However, a number of known adverse events were reported at a higher rate in the carfilzomib arm than the bortezomib arm, including any-grade dyspnea (28% vs 13%), hypertension (25% vs 3%), pyrexia (27% vs 14%), cough (25% vs 15%), cardiac failure (8% vs 3%), and acute renal failure (8% vs 5%).
Carfilzomib is marketed as Kyprolis by Onyx Pharmaceuticals, Inc., a subsidiary of Amgen that holds development and commercialization rights to the drug globally, with the exception of Japan. For more information on the drug, visit www.kyprolis.com. ![]()

Photo courtesy of Amgen
The US Food and Drug Administration (FDA) has approved the use of carfilzomib (Kyprolis) in combination with dexamethasone or lenalidomide plus dexamethasone to treat patients with relapsed or refractory multiple myeloma (MM) who have received 1 to 3 prior lines of therapy.
The FDA also approved use of carfilzomib as a single agent to treat patients with relapsed or refractory MM who have received 1 or more lines of therapy.
This decision converts to full approval the initial accelerated approval granted for single-agent carfilzomib in July 2012.
In July 2015, the FDA approved the use of carfilzomib in combination with lenalidomide and dexamethasone for the treatment of patients with relapsed MM who have received 1 to 3 prior lines of therapy.
The latest approval of carfilzomib is based on results from the phase 3 ENDEAVOR study.
Trial results
The ENDEAVOR trial included 929 MM patients whose disease had relapsed after 1 to 3 prior therapeutic regimens. The patients received either carfilzomib plus dexamethasone (n=464) or bortezomib plus dexamethasone (n=465) until disease progression.
The primary endpoint was progression-free survival. The median was 18.7 months in the carfilzomib arm and 9.4 months in the bortezomib arm. The hazard ratio was 0.53 (P<0.0001).
Overall survival data are not yet mature and continue to be monitored.
Treatment discontinuation due to adverse events and on-study deaths were comparable between the 2 treatment arms.
However, a number of known adverse events were reported at a higher rate in the carfilzomib arm than the bortezomib arm, including any-grade dyspnea (28% vs 13%), hypertension (25% vs 3%), pyrexia (27% vs 14%), cough (25% vs 15%), cardiac failure (8% vs 3%), and acute renal failure (8% vs 5%).
Carfilzomib is marketed as Kyprolis by Onyx Pharmaceuticals, Inc., a subsidiary of Amgen that holds development and commercialization rights to the drug globally, with the exception of Japan. For more information on the drug, visit www.kyprolis.com. ![]()
End-of-life cancer care by country
in the intensive care unit
A study of end-of-life cancer care practices in 7 countries suggests the US has the lowest proportion of deaths in the hospital and the lowest number of days in the hospital for patients in their last 6 months of life.
However, the US performed poorly in other aspects of care, particularly intensive care unit admissions and hospital expenditures.
The other countries included in the study were Belgium, Canada, England, Germany, the Netherlands, and Norway.
The research was published in JAMA.
Ezekiel J. Emanuel, MD, PhD, of the University of Pennsylvania in Philadelphia, and his colleagues examined patterns of care, healthcare utilization, and expenditures for dying cancer patients in the 7 aforementioned countries.
The researchers first analyzed data from 2010 that included subjects older than 65 years of age who died with cancer.
The proportion of patients who died in the hospital was 22.2% in the US, 29.4% in the Netherlands, 38.3% in Germany, 41.7% in England, 44.7% in Norway, 51.2% in Belgium, and 52.1% in Canada.
In the last 180 days of life, the mean number of days in the hospital per capita was 27.7 in Belgium, 24.8 in Norway, 21.7 in Germany, 19 in Canada, 18.3 in England, 17.8 in the Netherlands, and 10.7 in the US.
The proportion of patients admitted to the intensive care unit in their last 180 days of life was 40.3% in the US, 18.5% in Belgium, 15.2% in Canada, 10.2% in the Netherlands, and 8.2% in Germany. Data were not available for England and Norway.
In the last 180 days of life, average per capita hospital expenditures (in USD) were higher in Canada ($21,840), Norway ($19,783), and the US ($18,500), intermediate in Germany ($16,221) and Belgium ($15,699), and lowest in the Netherlands ($10,936) and England ($9342).
Analyses that included decedents of any age, decedents older than 65 years of age with lung cancer, and decedents older than 65 years in the US and Germany from 2012 showed similar results.
The researchers said this suggests the differences observed were driven more by end-of-life care practices and organization rather than differences in cohort identification. ![]()

in the intensive care unit
A study of end-of-life cancer care practices in 7 countries suggests the US has the lowest proportion of deaths in the hospital and the lowest number of days in the hospital for patients in their last 6 months of life.
However, the US performed poorly in other aspects of care, particularly intensive care unit admissions and hospital expenditures.
The other countries included in the study were Belgium, Canada, England, Germany, the Netherlands, and Norway.
The research was published in JAMA.
Ezekiel J. Emanuel, MD, PhD, of the University of Pennsylvania in Philadelphia, and his colleagues examined patterns of care, healthcare utilization, and expenditures for dying cancer patients in the 7 aforementioned countries.
The researchers first analyzed data from 2010 that included subjects older than 65 years of age who died with cancer.
The proportion of patients who died in the hospital was 22.2% in the US, 29.4% in the Netherlands, 38.3% in Germany, 41.7% in England, 44.7% in Norway, 51.2% in Belgium, and 52.1% in Canada.
In the last 180 days of life, the mean number of days in the hospital per capita was 27.7 in Belgium, 24.8 in Norway, 21.7 in Germany, 19 in Canada, 18.3 in England, 17.8 in the Netherlands, and 10.7 in the US.
The proportion of patients admitted to the intensive care unit in their last 180 days of life was 40.3% in the US, 18.5% in Belgium, 15.2% in Canada, 10.2% in the Netherlands, and 8.2% in Germany. Data were not available for England and Norway.
In the last 180 days of life, average per capita hospital expenditures (in USD) were higher in Canada ($21,840), Norway ($19,783), and the US ($18,500), intermediate in Germany ($16,221) and Belgium ($15,699), and lowest in the Netherlands ($10,936) and England ($9342).
Analyses that included decedents of any age, decedents older than 65 years of age with lung cancer, and decedents older than 65 years in the US and Germany from 2012 showed similar results.
The researchers said this suggests the differences observed were driven more by end-of-life care practices and organization rather than differences in cohort identification. ![]()

in the intensive care unit
A study of end-of-life cancer care practices in 7 countries suggests the US has the lowest proportion of deaths in the hospital and the lowest number of days in the hospital for patients in their last 6 months of life.
However, the US performed poorly in other aspects of care, particularly intensive care unit admissions and hospital expenditures.
The other countries included in the study were Belgium, Canada, England, Germany, the Netherlands, and Norway.
The research was published in JAMA.
Ezekiel J. Emanuel, MD, PhD, of the University of Pennsylvania in Philadelphia, and his colleagues examined patterns of care, healthcare utilization, and expenditures for dying cancer patients in the 7 aforementioned countries.
The researchers first analyzed data from 2010 that included subjects older than 65 years of age who died with cancer.
The proportion of patients who died in the hospital was 22.2% in the US, 29.4% in the Netherlands, 38.3% in Germany, 41.7% in England, 44.7% in Norway, 51.2% in Belgium, and 52.1% in Canada.
In the last 180 days of life, the mean number of days in the hospital per capita was 27.7 in Belgium, 24.8 in Norway, 21.7 in Germany, 19 in Canada, 18.3 in England, 17.8 in the Netherlands, and 10.7 in the US.
The proportion of patients admitted to the intensive care unit in their last 180 days of life was 40.3% in the US, 18.5% in Belgium, 15.2% in Canada, 10.2% in the Netherlands, and 8.2% in Germany. Data were not available for England and Norway.
In the last 180 days of life, average per capita hospital expenditures (in USD) were higher in Canada ($21,840), Norway ($19,783), and the US ($18,500), intermediate in Germany ($16,221) and Belgium ($15,699), and lowest in the Netherlands ($10,936) and England ($9342).
Analyses that included decedents of any age, decedents older than 65 years of age with lung cancer, and decedents older than 65 years in the US and Germany from 2012 showed similar results.
The researchers said this suggests the differences observed were driven more by end-of-life care practices and organization rather than differences in cohort identification.
Drug granted another breakthrough designation for CLL

The US Food and Drug Administration (FDA) has granted breakthrough therapy designation to the BCL-2 inhibitor venetoclax when given with rituximab to treat patients with relapsed or refractory chronic lymphocytic leukemia (CLL).
Venetoclax already had breakthrough designation from the FDA as single-agent treatment for patients with relapsed or refractory CLL and 17p deletion.
The drug was granted priority review for this indication as well.
Breakthrough therapy designation is designed to accelerate the development and review of medicines that demonstrate early clinical evidence of a substantial improvement over current treatment options for serious diseases.
The latest breakthrough designation for venetoclax is supported by a phase 2 study of the drug in combination with rituximab in patients with relapsed/refractory CLL. Results from this trial were presented at the 2015 ASH Annual Meeting (abstract 325).
Another phase 2 trial presented at that meeting (abstract LBA-6) showed that single-agent venetoclax is effective against CLL as well.
The drug has also proven active against other hematologic malignancies, including acute myeloid lekemia and multiple myeloma.
However, venetoclax has been shown to pose a risk of tumor lysis syndrome (TLS). In fact, TLS-related deaths temporarily halted enrollment in trials of venetoclax. But researchers discovered ways to reduce the risk of TLS, and the trials continued.
Venetoclax is being developed by AbbVie in partnership with Genentech and Roche.

The US Food and Drug Administration (FDA) has granted breakthrough therapy designation to the BCL-2 inhibitor venetoclax when given with rituximab to treat patients with relapsed or refractory chronic lymphocytic leukemia (CLL).
Venetoclax already had breakthrough designation from the FDA as single-agent treatment for patients with relapsed or refractory CLL and 17p deletion.
The drug was granted priority review for this indication as well.
Breakthrough therapy designation is designed to accelerate the development and review of medicines that demonstrate early clinical evidence of a substantial improvement over current treatment options for serious diseases.
The latest breakthrough designation for venetoclax is supported by a phase 2 study of the drug in combination with rituximab in patients with relapsed/refractory CLL. Results from this trial were presented at the 2015 ASH Annual Meeting (abstract 325).
Another phase 2 trial presented at that meeting (abstract LBA-6) showed that single-agent venetoclax is effective against CLL as well.
The drug has also proven active against other hematologic malignancies, including acute myeloid lekemia and multiple myeloma.
However, venetoclax has been shown to pose a risk of tumor lysis syndrome (TLS). In fact, TLS-related deaths temporarily halted enrollment in trials of venetoclax. But researchers discovered ways to reduce the risk of TLS, and the trials continued.
Venetoclax is being developed by AbbVie in partnership with Genentech and Roche.

The US Food and Drug Administration (FDA) has granted breakthrough therapy designation to the BCL-2 inhibitor venetoclax when given with rituximab to treat patients with relapsed or refractory chronic lymphocytic leukemia (CLL).
Venetoclax already had breakthrough designation from the FDA as single-agent treatment for patients with relapsed or refractory CLL and 17p deletion.
The drug was granted priority review for this indication as well.
Breakthrough therapy designation is designed to accelerate the development and review of medicines that demonstrate early clinical evidence of a substantial improvement over current treatment options for serious diseases.
The latest breakthrough designation for venetoclax is supported by a phase 2 study of the drug in combination with rituximab in patients with relapsed/refractory CLL. Results from this trial were presented at the 2015 ASH Annual Meeting (abstract 325).
Another phase 2 trial presented at that meeting (abstract LBA-6) showed that single-agent venetoclax is effective against CLL as well.
The drug has also proven active against other hematologic malignancies, including acute myeloid lekemia and multiple myeloma.
However, venetoclax has been shown to pose a risk of tumor lysis syndrome (TLS). In fact, TLS-related deaths temporarily halted enrollment in trials of venetoclax. But researchers discovered ways to reduce the risk of TLS, and the trials continued.
Venetoclax is being developed by AbbVie in partnership with Genentech and Roche.
Obesity linked to VTE in kids

Photo by Matthew Lester
A single-center, retrospective study has revealed an association between obesity and venous thromboembolism (VTE) in children and adolescents.
While obesity is a well-established risk factor for VTE in adults, previous studies in pediatric populations have yielded mixed results.
The new study, however, showed that obesity, as determined by body mass index (BMI), was a statistically significant predictor of VTE in juveniles.
The research was published in Hospital Pediatrics.
“This is important because the incidence of pediatric VTE has increased dramatically over the last 20 years, and childhood obesity remains highly prevalent in the United States,” said study author Elizabeth Halvorson, MD, of Wake Forest Baptist Medical Center in Winston-Salem, North Carolina.
For this study, she and her colleagues conducted a retrospective chart review of inpatients at Wake Forest Baptist’s Brenner Children’s Hospital between January 2000 and September 2012.
The researchers identified 88 patients, ages 2 to 18, who had confirmed cases of VTE. The team compared these patients to control subjects (2 controls per case) matched by age, gender, and the presence of a central venous catheter.
Of the 88 patients with VTE, 33 (37.5%) were obese, although most of them had known risk factors for VTE in addition to obesity.
In univariate analysis, the researchers found a statistically significant association between VTE and obesity, or increased BMI z score (P=0.002).
In a multivariate analysis, obesity remained a significant predictor of VTE. The odds ratio (OR) was 3.1 (P=0.007).
Other factors were significant predictors of VTE as well, including bacteremia (OR: 4.9; P=0.02), a stay in the intensive care unit (OR: 2.5; P=0.02), and the use of oral contraceptives (OR: 17.4; P<0.001).
“Our study presents data from a single institution with a relatively small sample size,” Dr Halvorson noted. “But it does demonstrate an association between obesity and VTE in children, which should be explored further in larger future studies.”

Photo by Matthew Lester
A single-center, retrospective study has revealed an association between obesity and venous thromboembolism (VTE) in children and adolescents.
While obesity is a well-established risk factor for VTE in adults, previous studies in pediatric populations have yielded mixed results.
The new study, however, showed that obesity, as determined by body mass index (BMI), was a statistically significant predictor of VTE in juveniles.
The research was published in Hospital Pediatrics.
“This is important because the incidence of pediatric VTE has increased dramatically over the last 20 years, and childhood obesity remains highly prevalent in the United States,” said study author Elizabeth Halvorson, MD, of Wake Forest Baptist Medical Center in Winston-Salem, North Carolina.
For this study, she and her colleagues conducted a retrospective chart review of inpatients at Wake Forest Baptist’s Brenner Children’s Hospital between January 2000 and September 2012.
The researchers identified 88 patients, ages 2 to 18, who had confirmed cases of VTE. The team compared these patients to control subjects (2 controls per case) matched by age, gender, and the presence of a central venous catheter.
Of the 88 patients with VTE, 33 (37.5%) were obese, although most of them had known risk factors for VTE in addition to obesity.
In univariate analysis, the researchers found a statistically significant association between VTE and obesity, or increased BMI z score (P=0.002).
In a multivariate analysis, obesity remained a significant predictor of VTE. The odds ratio (OR) was 3.1 (P=0.007).
Other factors were significant predictors of VTE as well, including bacteremia (OR: 4.9; P=0.02), a stay in the intensive care unit (OR: 2.5; P=0.02), and the use of oral contraceptives (OR: 17.4; P<0.001).
“Our study presents data from a single institution with a relatively small sample size,” Dr Halvorson noted. “But it does demonstrate an association between obesity and VTE in children, which should be explored further in larger future studies.”

Photo by Matthew Lester
A single-center, retrospective study has revealed an association between obesity and venous thromboembolism (VTE) in children and adolescents.
While obesity is a well-established risk factor for VTE in adults, previous studies in pediatric populations have yielded mixed results.
The new study, however, showed that obesity, as determined by body mass index (BMI), was a statistically significant predictor of VTE in juveniles.
The research was published in Hospital Pediatrics.
“This is important because the incidence of pediatric VTE has increased dramatically over the last 20 years, and childhood obesity remains highly prevalent in the United States,” said study author Elizabeth Halvorson, MD, of Wake Forest Baptist Medical Center in Winston-Salem, North Carolina.
For this study, she and her colleagues conducted a retrospective chart review of inpatients at Wake Forest Baptist’s Brenner Children’s Hospital between January 2000 and September 2012.
The researchers identified 88 patients, ages 2 to 18, who had confirmed cases of VTE. The team compared these patients to control subjects (2 controls per case) matched by age, gender, and the presence of a central venous catheter.
Of the 88 patients with VTE, 33 (37.5%) were obese, although most of them had known risk factors for VTE in addition to obesity.
In univariate analysis, the researchers found a statistically significant association between VTE and obesity, or increased BMI z score (P=0.002).
In a multivariate analysis, obesity remained a significant predictor of VTE. The odds ratio (OR) was 3.1 (P=0.007).
Other factors were significant predictors of VTE as well, including bacteremia (OR: 4.9; P=0.02), a stay in the intensive care unit (OR: 2.5; P=0.02), and the use of oral contraceptives (OR: 17.4; P<0.001).
“Our study presents data from a single institution with a relatively small sample size,” Dr Halvorson noted. “But it does demonstrate an association between obesity and VTE in children, which should be explored further in larger future studies.”
Protein may be therapeutic target for AML

and Matt McCormack, PhD
Photo courtesy of the
Walter and Eliza Hall
Institute of Medical Research
Preclinical research suggests the Hhex protein could be a cancer-specific therapeutic target for acute myeloid leukemia (AML).
Investigators discovered that loss of the Hhex protein halted leukemia cell growth and division in vitro and in vivo, but normal cells were unaffected by the loss of Hhex.
Matt McCormack, PhD, of the Walter and Eliza Hall Institute of Medical Research in Parkville, Victoria, Australia, and his colleagues relayed these findings in Genes and Development.
“There is an urgent need for new therapies to treat AML,” Dr McCormack said. “We showed blocking the Hhex protein could put the brakes on leukemia growth and completely eliminate AML in preclinical models. This could be targeted by new drugs to treat AML in humans.”
Specifically, the investigators found that Hhex was overexpressed in human AML, and the protein was essential for the maintenance of AML driven by the oncogenic fusion protein MLL-ENL and its downstream effectors, HoxA9 and Meis1.
However, Hhex was not required for normal myelopoiesis.
“Hhex is only essential for the leukemic cells, meaning we could target and treat leukemia without toxic effects on normal cells, avoiding many of the serious side effects that come with standard cancer treatments,” Dr McCormack said.
“We also know that most people with AML have increased levels of Hhex, often associated with adverse outcomes, further indicating it is an important target for new AML drugs.”
Dr McCormack and his colleagues also attempted to determine the mechanism by which Hhex promotes AML.
They found the protein represses the tumor suppressors p16INK4a and p19ARF in leukemic stem cells by regulating the Polycomb-repressive complex 2 (PRC2). They said that Hhex binds to the Cdkn2a locus and directly interacts with PRC2 to enable H3K27me3-mediated epigenetic repression.
“Hhex works by recruiting epigenetic factors to growth-control genes, effectively silencing them,” said author Ben Shields, PhD, also of the Walter and Eliza Hall Institute.
“This allows the leukemia cells to reproduce and accumulate more damage, contributing to the speed of AML progression.”
Dr McCormack said that although drugs inhibiting epigenetic modification have been tested against AML in the past, they have caused significant toxicity because their targets are also required for normal blood cell function.
“Unlike the epigenetic factors targeted previously, Hhex only regulates a small number of genes and is dispensable for normal blood cells,” Dr McCormack reiterated.
“This gives us a rare opportunity to kill AML cells without causing many side effects. We now hope to identify the critical regions of the Hhex protein that enable it to function, which will allow us to design much-needed new drugs to treat AML.”

and Matt McCormack, PhD
Photo courtesy of the
Walter and Eliza Hall
Institute of Medical Research
Preclinical research suggests the Hhex protein could be a cancer-specific therapeutic target for acute myeloid leukemia (AML).
Investigators discovered that loss of the Hhex protein halted leukemia cell growth and division in vitro and in vivo, but normal cells were unaffected by the loss of Hhex.
Matt McCormack, PhD, of the Walter and Eliza Hall Institute of Medical Research in Parkville, Victoria, Australia, and his colleagues relayed these findings in Genes and Development.
“There is an urgent need for new therapies to treat AML,” Dr McCormack said. “We showed blocking the Hhex protein could put the brakes on leukemia growth and completely eliminate AML in preclinical models. This could be targeted by new drugs to treat AML in humans.”
Specifically, the investigators found that Hhex was overexpressed in human AML, and the protein was essential for the maintenance of AML driven by the oncogenic fusion protein MLL-ENL and its downstream effectors, HoxA9 and Meis1.
However, Hhex was not required for normal myelopoiesis.
“Hhex is only essential for the leukemic cells, meaning we could target and treat leukemia without toxic effects on normal cells, avoiding many of the serious side effects that come with standard cancer treatments,” Dr McCormack said.
“We also know that most people with AML have increased levels of Hhex, often associated with adverse outcomes, further indicating it is an important target for new AML drugs.”
Dr McCormack and his colleagues also attempted to determine the mechanism by which Hhex promotes AML.
They found the protein represses the tumor suppressors p16INK4a and p19ARF in leukemic stem cells by regulating the Polycomb-repressive complex 2 (PRC2). They said that Hhex binds to the Cdkn2a locus and directly interacts with PRC2 to enable H3K27me3-mediated epigenetic repression.
“Hhex works by recruiting epigenetic factors to growth-control genes, effectively silencing them,” said author Ben Shields, PhD, also of the Walter and Eliza Hall Institute.
“This allows the leukemia cells to reproduce and accumulate more damage, contributing to the speed of AML progression.”
Dr McCormack said that although drugs inhibiting epigenetic modification have been tested against AML in the past, they have caused significant toxicity because their targets are also required for normal blood cell function.
“Unlike the epigenetic factors targeted previously, Hhex only regulates a small number of genes and is dispensable for normal blood cells,” Dr McCormack reiterated.
“This gives us a rare opportunity to kill AML cells without causing many side effects. We now hope to identify the critical regions of the Hhex protein that enable it to function, which will allow us to design much-needed new drugs to treat AML.”

and Matt McCormack, PhD
Photo courtesy of the
Walter and Eliza Hall
Institute of Medical Research
Preclinical research suggests the Hhex protein could be a cancer-specific therapeutic target for acute myeloid leukemia (AML).
Investigators discovered that loss of the Hhex protein halted leukemia cell growth and division in vitro and in vivo, but normal cells were unaffected by the loss of Hhex.
Matt McCormack, PhD, of the Walter and Eliza Hall Institute of Medical Research in Parkville, Victoria, Australia, and his colleagues relayed these findings in Genes and Development.
“There is an urgent need for new therapies to treat AML,” Dr McCormack said. “We showed blocking the Hhex protein could put the brakes on leukemia growth and completely eliminate AML in preclinical models. This could be targeted by new drugs to treat AML in humans.”
Specifically, the investigators found that Hhex was overexpressed in human AML, and the protein was essential for the maintenance of AML driven by the oncogenic fusion protein MLL-ENL and its downstream effectors, HoxA9 and Meis1.
However, Hhex was not required for normal myelopoiesis.
“Hhex is only essential for the leukemic cells, meaning we could target and treat leukemia without toxic effects on normal cells, avoiding many of the serious side effects that come with standard cancer treatments,” Dr McCormack said.
“We also know that most people with AML have increased levels of Hhex, often associated with adverse outcomes, further indicating it is an important target for new AML drugs.”
Dr McCormack and his colleagues also attempted to determine the mechanism by which Hhex promotes AML.
They found the protein represses the tumor suppressors p16INK4a and p19ARF in leukemic stem cells by regulating the Polycomb-repressive complex 2 (PRC2). They said that Hhex binds to the Cdkn2a locus and directly interacts with PRC2 to enable H3K27me3-mediated epigenetic repression.
“Hhex works by recruiting epigenetic factors to growth-control genes, effectively silencing them,” said author Ben Shields, PhD, also of the Walter and Eliza Hall Institute.
“This allows the leukemia cells to reproduce and accumulate more damage, contributing to the speed of AML progression.”
Dr McCormack said that although drugs inhibiting epigenetic modification have been tested against AML in the past, they have caused significant toxicity because their targets are also required for normal blood cell function.
“Unlike the epigenetic factors targeted previously, Hhex only regulates a small number of genes and is dispensable for normal blood cells,” Dr McCormack reiterated.
“This gives us a rare opportunity to kill AML cells without causing many side effects. We now hope to identify the critical regions of the Hhex protein that enable it to function, which will allow us to design much-needed new drugs to treat AML.”