User login
Survey reveals need to evaluate EOL discussions

Photo courtesy of the
National Cancer Institute
and Mathews Media Group
End-of-life (EOL) discussions often occur “too late” for patients with hematologic malignancies, according to a survey of US hematologists.
The researchers who conducted the survey speculate that physicians may delay EOL discussions with these patients because, unlike most solid tumors,
which are incurable when they reach an advanced stage, many advanced hematologic malignancies remain curable.
So it may not be clear that a patient has entered the EOL phase.
Oreofe O. Odejide, MD, of the Dana-Farber Cancer Institute in Boston, Massachusetts, and colleagues conducted the survey and reported the results in a letter to JAMA Internal Medicine.
The researchers mailed their survey on EOL discussions to US hematologists found in the clinical directory of the American Society of Hematology. The individuals surveyed provide direct care for adults with hematologic malignancies.
Three hundred and forty-nine hematologists completed the survey. Most were men (75.4%), and they had a median age of 52. More than half (55.4%) practiced in community centers, and 42.9% practiced primarily in tertiary centers.
Three hundred and forty-five individuals answered the question about typical timing of EOL discussions, and 55.9% said these discussions occur too late.
Hematologists practicing in tertiary centers were more likely to report late EOL discussions than those practicing in community centers—64.9% and 48.7%, respectively (P=0.003). This difference was still significant in multivariable analysis, with an odds ratio of 1.92 (P=0.004).
When it comes to specific aspects of EOL care, 42.5% of the hematologists reported conducting their first conversation about resuscitation status at less than optimal times; 23.2% reported waiting until death was clearly imminent before having an initial conversation about hospice care; and 39.9% reported waiting until death was clearly imminent before having an initial conversation about the preferred site of death.
The researchers said the lack of a clear distinction between the curative and EOL phases of hematologic malignancies may explain these findings. Additionally, physicians may hesitate to have EOL discussions because they don’t want to affect a patient’s mentality or because they themselves find it difficult to “give up” on patients who might still be cured. ![]()

Photo courtesy of the
National Cancer Institute
and Mathews Media Group
End-of-life (EOL) discussions often occur “too late” for patients with hematologic malignancies, according to a survey of US hematologists.
The researchers who conducted the survey speculate that physicians may delay EOL discussions with these patients because, unlike most solid tumors,
which are incurable when they reach an advanced stage, many advanced hematologic malignancies remain curable.
So it may not be clear that a patient has entered the EOL phase.
Oreofe O. Odejide, MD, of the Dana-Farber Cancer Institute in Boston, Massachusetts, and colleagues conducted the survey and reported the results in a letter to JAMA Internal Medicine.
The researchers mailed their survey on EOL discussions to US hematologists found in the clinical directory of the American Society of Hematology. The individuals surveyed provide direct care for adults with hematologic malignancies.
Three hundred and forty-nine hematologists completed the survey. Most were men (75.4%), and they had a median age of 52. More than half (55.4%) practiced in community centers, and 42.9% practiced primarily in tertiary centers.
Three hundred and forty-five individuals answered the question about typical timing of EOL discussions, and 55.9% said these discussions occur too late.
Hematologists practicing in tertiary centers were more likely to report late EOL discussions than those practicing in community centers—64.9% and 48.7%, respectively (P=0.003). This difference was still significant in multivariable analysis, with an odds ratio of 1.92 (P=0.004).
When it comes to specific aspects of EOL care, 42.5% of the hematologists reported conducting their first conversation about resuscitation status at less than optimal times; 23.2% reported waiting until death was clearly imminent before having an initial conversation about hospice care; and 39.9% reported waiting until death was clearly imminent before having an initial conversation about the preferred site of death.
The researchers said the lack of a clear distinction between the curative and EOL phases of hematologic malignancies may explain these findings. Additionally, physicians may hesitate to have EOL discussions because they don’t want to affect a patient’s mentality or because they themselves find it difficult to “give up” on patients who might still be cured. ![]()

Photo courtesy of the
National Cancer Institute
and Mathews Media Group
End-of-life (EOL) discussions often occur “too late” for patients with hematologic malignancies, according to a survey of US hematologists.
The researchers who conducted the survey speculate that physicians may delay EOL discussions with these patients because, unlike most solid tumors,
which are incurable when they reach an advanced stage, many advanced hematologic malignancies remain curable.
So it may not be clear that a patient has entered the EOL phase.
Oreofe O. Odejide, MD, of the Dana-Farber Cancer Institute in Boston, Massachusetts, and colleagues conducted the survey and reported the results in a letter to JAMA Internal Medicine.
The researchers mailed their survey on EOL discussions to US hematologists found in the clinical directory of the American Society of Hematology. The individuals surveyed provide direct care for adults with hematologic malignancies.
Three hundred and forty-nine hematologists completed the survey. Most were men (75.4%), and they had a median age of 52. More than half (55.4%) practiced in community centers, and 42.9% practiced primarily in tertiary centers.
Three hundred and forty-five individuals answered the question about typical timing of EOL discussions, and 55.9% said these discussions occur too late.
Hematologists practicing in tertiary centers were more likely to report late EOL discussions than those practicing in community centers—64.9% and 48.7%, respectively (P=0.003). This difference was still significant in multivariable analysis, with an odds ratio of 1.92 (P=0.004).
When it comes to specific aspects of EOL care, 42.5% of the hematologists reported conducting their first conversation about resuscitation status at less than optimal times; 23.2% reported waiting until death was clearly imminent before having an initial conversation about hospice care; and 39.9% reported waiting until death was clearly imminent before having an initial conversation about the preferred site of death.
The researchers said the lack of a clear distinction between the curative and EOL phases of hematologic malignancies may explain these findings. Additionally, physicians may hesitate to have EOL discussions because they don’t want to affect a patient’s mentality or because they themselves find it difficult to “give up” on patients who might still be cured. ![]()
FDA changes deferral policy for MSM blood donors
Photo by Михаило Јовановић
The US Food and Drug Administration (FDA) has issued a final guidance outlining updated blood donor deferral recommendations.
As part of the guidance, the FDA is changing its recommendation that men who have sex with men (MSM) be indefinitely deferred from donating blood—a policy that has been in place for approximately 30 years.
Now, the agency is recommending that MSMs be deferred for 12 months since their last sexual contact with another man.
The FDA’s guidance also reflects a change in the rationale for deferring potential blood donors with hemophilia or related clotting disorders who have received clotting factor concentrates.
The FDA recommends that blood establishments make corresponding revisions to donor educational materials, donor history questionnaires, and accompanying materials, as well as donor requalification and product management procedures.
MSM deferral
The FDA said its recommendation regarding MSM blood donors reflects the most current scientific evidence and will help ensure continued safety of the blood supply by reducing the risk of human immunodeficiency virus (HIV) transmission by blood and blood products.
The agency also said this recommendation better aligns the deferral period for MSMs with the deferral period for other men and women at increased risk for HIV infection, such as those who had a recent blood transfusion or those who have been accidentally exposed to the blood of another individual.
Before issuing this guidance, the FDA reviewed its policies regarding HIV transmission through blood products to determine appropriate changes based on the most recent scientific evidence. The agency examined a variety of studies, epidemiologic data, and shared experiences from other countries that have made recent MSM deferral policy changes.
“In reviewing our policies to help reduce the risk of HIV transmission through blood products, we rigorously examined several alternative options, including individual risk assessment,” said Peter Marks, MD, PhD, deputy director of the FDA’s Center for Biologics Evaluation and Research.
“Ultimately, the 12-month deferral window is supported by the best available scientific evidence, at this point in time, relevant to the US population. We will continue to actively conduct research in this area and further revise our policies as new data emerge.”
Several countries, including the UK and Australia, currently have 12-month deferral policies for MSM blood donors.
During the change in Australia from an indefinite blood donor deferral policy for MSMs to a 12-month deferral, studies evaluating over 8 million units of donated blood were performed using a national blood surveillance system. These studies (CR Seed et al, Transfusion 2010; TTA Lucky et al, Transfusion 2014) show no change in risk to the blood supply with use of the 12-month deferral.
A study conducted in the UK produced similar results, although it also suggested that 3 in 10 MSMs don’t comply with the 12-month deferral policy.
And a study conducted in Canada, which recently shortened its MSM deferral period to 5 years, showed no change in risk to the blood supply with the 5-year deferral as compared to indefinite deferral. Based on these results, Canadian regulators are considering changing to a 12-month deferral period as well.
Patients with clotting disorders
The FDA’s new guidance also reflects a change in the rationale for deferring patients with hemophilia or related clotting disorders who have received clotting factor concentrates. Previously, potential donors with hemophilia or related clotting disorders were deferred due to the increased risk of HIV transmission to potential recipients.
Based on new scientific evidence, these potential donors are still deferred, but not due to the risk of HIV transmission—instead, for their own protection due to potential harm from large needles used during the donation process.
FDA policies and actions
Throughout the process of updating blood donor deferral policies over the past several years, the FDA has worked with other government agencies, considered input from external advisory committees, reviewed comments from stakeholders to its May 2015 draft guidance, and examined the most recent available scientific evidence to support the current policy revision.
The FDA has also implemented a nationally representative safety monitoring system for the blood supply with assistance from the National Heart, Lung and Blood Institute at the National Institutes of Health. This system will provide information to help inform future actions the FDA may take on blood donor policies.
The FDA said it will continue to reevaluate and update its blood donor deferral policies as new scientific information becomes available. ![]()

Photo by Михаило Јовановић
The US Food and Drug Administration (FDA) has issued a final guidance outlining updated blood donor deferral recommendations.
As part of the guidance, the FDA is changing its recommendation that men who have sex with men (MSM) be indefinitely deferred from donating blood—a policy that has been in place for approximately 30 years.
Now, the agency is recommending that MSMs be deferred for 12 months since their last sexual contact with another man.
The FDA’s guidance also reflects a change in the rationale for deferring potential blood donors with hemophilia or related clotting disorders who have received clotting factor concentrates.
The FDA recommends that blood establishments make corresponding revisions to donor educational materials, donor history questionnaires, and accompanying materials, as well as donor requalification and product management procedures.
MSM deferral
The FDA said its recommendation regarding MSM blood donors reflects the most current scientific evidence and will help ensure continued safety of the blood supply by reducing the risk of human immunodeficiency virus (HIV) transmission by blood and blood products.
The agency also said this recommendation better aligns the deferral period for MSMs with the deferral period for other men and women at increased risk for HIV infection, such as those who had a recent blood transfusion or those who have been accidentally exposed to the blood of another individual.
Before issuing this guidance, the FDA reviewed its policies regarding HIV transmission through blood products to determine appropriate changes based on the most recent scientific evidence. The agency examined a variety of studies, epidemiologic data, and shared experiences from other countries that have made recent MSM deferral policy changes.
“In reviewing our policies to help reduce the risk of HIV transmission through blood products, we rigorously examined several alternative options, including individual risk assessment,” said Peter Marks, MD, PhD, deputy director of the FDA’s Center for Biologics Evaluation and Research.
“Ultimately, the 12-month deferral window is supported by the best available scientific evidence, at this point in time, relevant to the US population. We will continue to actively conduct research in this area and further revise our policies as new data emerge.”
Several countries, including the UK and Australia, currently have 12-month deferral policies for MSM blood donors.
During the change in Australia from an indefinite blood donor deferral policy for MSMs to a 12-month deferral, studies evaluating over 8 million units of donated blood were performed using a national blood surveillance system. These studies (CR Seed et al, Transfusion 2010; TTA Lucky et al, Transfusion 2014) show no change in risk to the blood supply with use of the 12-month deferral.
A study conducted in the UK produced similar results, although it also suggested that 3 in 10 MSMs don’t comply with the 12-month deferral policy.
And a study conducted in Canada, which recently shortened its MSM deferral period to 5 years, showed no change in risk to the blood supply with the 5-year deferral as compared to indefinite deferral. Based on these results, Canadian regulators are considering changing to a 12-month deferral period as well.
Patients with clotting disorders
The FDA’s new guidance also reflects a change in the rationale for deferring patients with hemophilia or related clotting disorders who have received clotting factor concentrates. Previously, potential donors with hemophilia or related clotting disorders were deferred due to the increased risk of HIV transmission to potential recipients.
Based on new scientific evidence, these potential donors are still deferred, but not due to the risk of HIV transmission—instead, for their own protection due to potential harm from large needles used during the donation process.
FDA policies and actions
Throughout the process of updating blood donor deferral policies over the past several years, the FDA has worked with other government agencies, considered input from external advisory committees, reviewed comments from stakeholders to its May 2015 draft guidance, and examined the most recent available scientific evidence to support the current policy revision.
The FDA has also implemented a nationally representative safety monitoring system for the blood supply with assistance from the National Heart, Lung and Blood Institute at the National Institutes of Health. This system will provide information to help inform future actions the FDA may take on blood donor policies.
The FDA said it will continue to reevaluate and update its blood donor deferral policies as new scientific information becomes available. ![]()

Photo by Михаило Јовановић
The US Food and Drug Administration (FDA) has issued a final guidance outlining updated blood donor deferral recommendations.
As part of the guidance, the FDA is changing its recommendation that men who have sex with men (MSM) be indefinitely deferred from donating blood—a policy that has been in place for approximately 30 years.
Now, the agency is recommending that MSMs be deferred for 12 months since their last sexual contact with another man.
The FDA’s guidance also reflects a change in the rationale for deferring potential blood donors with hemophilia or related clotting disorders who have received clotting factor concentrates.
The FDA recommends that blood establishments make corresponding revisions to donor educational materials, donor history questionnaires, and accompanying materials, as well as donor requalification and product management procedures.
MSM deferral
The FDA said its recommendation regarding MSM blood donors reflects the most current scientific evidence and will help ensure continued safety of the blood supply by reducing the risk of human immunodeficiency virus (HIV) transmission by blood and blood products.
The agency also said this recommendation better aligns the deferral period for MSMs with the deferral period for other men and women at increased risk for HIV infection, such as those who had a recent blood transfusion or those who have been accidentally exposed to the blood of another individual.
Before issuing this guidance, the FDA reviewed its policies regarding HIV transmission through blood products to determine appropriate changes based on the most recent scientific evidence. The agency examined a variety of studies, epidemiologic data, and shared experiences from other countries that have made recent MSM deferral policy changes.
“In reviewing our policies to help reduce the risk of HIV transmission through blood products, we rigorously examined several alternative options, including individual risk assessment,” said Peter Marks, MD, PhD, deputy director of the FDA’s Center for Biologics Evaluation and Research.
“Ultimately, the 12-month deferral window is supported by the best available scientific evidence, at this point in time, relevant to the US population. We will continue to actively conduct research in this area and further revise our policies as new data emerge.”
Several countries, including the UK and Australia, currently have 12-month deferral policies for MSM blood donors.
During the change in Australia from an indefinite blood donor deferral policy for MSMs to a 12-month deferral, studies evaluating over 8 million units of donated blood were performed using a national blood surveillance system. These studies (CR Seed et al, Transfusion 2010; TTA Lucky et al, Transfusion 2014) show no change in risk to the blood supply with use of the 12-month deferral.
A study conducted in the UK produced similar results, although it also suggested that 3 in 10 MSMs don’t comply with the 12-month deferral policy.
And a study conducted in Canada, which recently shortened its MSM deferral period to 5 years, showed no change in risk to the blood supply with the 5-year deferral as compared to indefinite deferral. Based on these results, Canadian regulators are considering changing to a 12-month deferral period as well.
Patients with clotting disorders
The FDA’s new guidance also reflects a change in the rationale for deferring patients with hemophilia or related clotting disorders who have received clotting factor concentrates. Previously, potential donors with hemophilia or related clotting disorders were deferred due to the increased risk of HIV transmission to potential recipients.
Based on new scientific evidence, these potential donors are still deferred, but not due to the risk of HIV transmission—instead, for their own protection due to potential harm from large needles used during the donation process.
FDA policies and actions
Throughout the process of updating blood donor deferral policies over the past several years, the FDA has worked with other government agencies, considered input from external advisory committees, reviewed comments from stakeholders to its May 2015 draft guidance, and examined the most recent available scientific evidence to support the current policy revision.
The FDA has also implemented a nationally representative safety monitoring system for the blood supply with assistance from the National Heart, Lung and Blood Institute at the National Institutes of Health. This system will provide information to help inform future actions the FDA may take on blood donor policies.
The FDA said it will continue to reevaluate and update its blood donor deferral policies as new scientific information becomes available. ![]()
Antibody shows promise for treating CLL

Preclinical research suggests a humanized monoclonal antibody called cirmtuzumab (UC-961) might be an effective treatment for chronic lymphocytic leukemia (CLL).
Experiments revealed that the Wnt5a protein acts on the tumor-surface proteins ROR1 and ROR2 to accelerate the proliferation and spread of CLL cells.
But cirmtuzumab, which is specific for ROR1, can block the effects of Wnt5a and inhibit the growth and spread of CLL cells both in vitro and in vivo.
Investigators reported these results in The Journal of Clinical Investigation.
They noted that ROR1 and ROR2 are considered orphan receptors, which are expressed primarily during embryonic development. The expression of these proteins, particularly ROR1, becomes suppressed during fetal development and is negligible on normal adult tissues. However, CLL and many solid tissue cancers re-express these orphan receptors.
“Our findings show that ROR1 and ROR2 team up to stimulate tumor cell growth and metastasis in response to Wnt5a, which appears overexpressed in patients with CLL and can act as a survival/growth factor for leukemia cells,” said study author Thomas J. Kipps, MD, PhD, of Moores Cancer Center at the University of California, San Diego.
“By blocking the capacity of Wnt5a to stimulate tumor cells, cirmtuzumab can inhibit the growth and spread of cancer cells. We now have better insight into how cirmtuzumab works against leukemia cells. This should help find better ways to treat patients who have other cancers with cirmtuzumab, which currently is being evaluated in a phase 1 clinical trial for patients with CLL.”
The JCI paper follows a series of related findings by Dr Kipps and his colleagues in recent years.
In 2008, they reported that patients vaccinated with their own CLL cells could make antibodies against ROR1, some of which had the ability to reduce CLL cell survival. They found ROR1 on CLL cells but not on all normal adult tissues examined.
In 2012, the investigators reported finding ROR1 on many different types of cancer, particularly cancers that appear less differentiated and more likely to spread to other parts in the body. Because this protein was not found on normal adult tissues, these findings made ROR1 a new target for anticancer drug research.
In June 2013, the team linked ROR1 to a process used in early development, suggesting cancer cells hijack an embryological process called epithelial-mesenchymal transition to spread or metastasize more quickly.
In January 2014, the investigators reported expression of ROR1 resulted in a faster-developing, more aggressive form of CLL in mice.
In September 2014, the team launched a phase 1 trial of cirmtuzumab in patients with CLL. The trial is ongoing.
In November 2014, the investigators described cellular experiments indicating that cirmtuzumab might be effective against cancer stem cells, which appear responsible for the relapse and spread of cancer after conventional therapy.
The latest research more precisely defines the roles of ROR1 and ROR2 in CLL development.
Both are evolutionarily conserved proteins that are found in many species and are most active in the early stages of embryogenesis when cells are migrating to form organs and parts of the body. The lack of either during this process results in severe developmental abnormalities.
Low levels of ROR2 remain in some adult tissues, but ROR1 is found only in cancer cells. The investigators found that, in response to signaling by Wnt5a, ROR1 and ROR2 come together to signal the growth and migration of CLL cells.
But treating mice with cirmtuzumab disrupted the process, inhibiting the engraftment of CLL cells and slowing or stopping the disease from spreading. ![]()

Preclinical research suggests a humanized monoclonal antibody called cirmtuzumab (UC-961) might be an effective treatment for chronic lymphocytic leukemia (CLL).
Experiments revealed that the Wnt5a protein acts on the tumor-surface proteins ROR1 and ROR2 to accelerate the proliferation and spread of CLL cells.
But cirmtuzumab, which is specific for ROR1, can block the effects of Wnt5a and inhibit the growth and spread of CLL cells both in vitro and in vivo.
Investigators reported these results in The Journal of Clinical Investigation.
They noted that ROR1 and ROR2 are considered orphan receptors, which are expressed primarily during embryonic development. The expression of these proteins, particularly ROR1, becomes suppressed during fetal development and is negligible on normal adult tissues. However, CLL and many solid tissue cancers re-express these orphan receptors.
“Our findings show that ROR1 and ROR2 team up to stimulate tumor cell growth and metastasis in response to Wnt5a, which appears overexpressed in patients with CLL and can act as a survival/growth factor for leukemia cells,” said study author Thomas J. Kipps, MD, PhD, of Moores Cancer Center at the University of California, San Diego.
“By blocking the capacity of Wnt5a to stimulate tumor cells, cirmtuzumab can inhibit the growth and spread of cancer cells. We now have better insight into how cirmtuzumab works against leukemia cells. This should help find better ways to treat patients who have other cancers with cirmtuzumab, which currently is being evaluated in a phase 1 clinical trial for patients with CLL.”
The JCI paper follows a series of related findings by Dr Kipps and his colleagues in recent years.
In 2008, they reported that patients vaccinated with their own CLL cells could make antibodies against ROR1, some of which had the ability to reduce CLL cell survival. They found ROR1 on CLL cells but not on all normal adult tissues examined.
In 2012, the investigators reported finding ROR1 on many different types of cancer, particularly cancers that appear less differentiated and more likely to spread to other parts in the body. Because this protein was not found on normal adult tissues, these findings made ROR1 a new target for anticancer drug research.
In June 2013, the team linked ROR1 to a process used in early development, suggesting cancer cells hijack an embryological process called epithelial-mesenchymal transition to spread or metastasize more quickly.
In January 2014, the investigators reported expression of ROR1 resulted in a faster-developing, more aggressive form of CLL in mice.
In September 2014, the team launched a phase 1 trial of cirmtuzumab in patients with CLL. The trial is ongoing.
In November 2014, the investigators described cellular experiments indicating that cirmtuzumab might be effective against cancer stem cells, which appear responsible for the relapse and spread of cancer after conventional therapy.
The latest research more precisely defines the roles of ROR1 and ROR2 in CLL development.
Both are evolutionarily conserved proteins that are found in many species and are most active in the early stages of embryogenesis when cells are migrating to form organs and parts of the body. The lack of either during this process results in severe developmental abnormalities.
Low levels of ROR2 remain in some adult tissues, but ROR1 is found only in cancer cells. The investigators found that, in response to signaling by Wnt5a, ROR1 and ROR2 come together to signal the growth and migration of CLL cells.
But treating mice with cirmtuzumab disrupted the process, inhibiting the engraftment of CLL cells and slowing or stopping the disease from spreading. ![]()

Preclinical research suggests a humanized monoclonal antibody called cirmtuzumab (UC-961) might be an effective treatment for chronic lymphocytic leukemia (CLL).
Experiments revealed that the Wnt5a protein acts on the tumor-surface proteins ROR1 and ROR2 to accelerate the proliferation and spread of CLL cells.
But cirmtuzumab, which is specific for ROR1, can block the effects of Wnt5a and inhibit the growth and spread of CLL cells both in vitro and in vivo.
Investigators reported these results in The Journal of Clinical Investigation.
They noted that ROR1 and ROR2 are considered orphan receptors, which are expressed primarily during embryonic development. The expression of these proteins, particularly ROR1, becomes suppressed during fetal development and is negligible on normal adult tissues. However, CLL and many solid tissue cancers re-express these orphan receptors.
“Our findings show that ROR1 and ROR2 team up to stimulate tumor cell growth and metastasis in response to Wnt5a, which appears overexpressed in patients with CLL and can act as a survival/growth factor for leukemia cells,” said study author Thomas J. Kipps, MD, PhD, of Moores Cancer Center at the University of California, San Diego.
“By blocking the capacity of Wnt5a to stimulate tumor cells, cirmtuzumab can inhibit the growth and spread of cancer cells. We now have better insight into how cirmtuzumab works against leukemia cells. This should help find better ways to treat patients who have other cancers with cirmtuzumab, which currently is being evaluated in a phase 1 clinical trial for patients with CLL.”
The JCI paper follows a series of related findings by Dr Kipps and his colleagues in recent years.
In 2008, they reported that patients vaccinated with their own CLL cells could make antibodies against ROR1, some of which had the ability to reduce CLL cell survival. They found ROR1 on CLL cells but not on all normal adult tissues examined.
In 2012, the investigators reported finding ROR1 on many different types of cancer, particularly cancers that appear less differentiated and more likely to spread to other parts in the body. Because this protein was not found on normal adult tissues, these findings made ROR1 a new target for anticancer drug research.
In June 2013, the team linked ROR1 to a process used in early development, suggesting cancer cells hijack an embryological process called epithelial-mesenchymal transition to spread or metastasize more quickly.
In January 2014, the investigators reported expression of ROR1 resulted in a faster-developing, more aggressive form of CLL in mice.
In September 2014, the team launched a phase 1 trial of cirmtuzumab in patients with CLL. The trial is ongoing.
In November 2014, the investigators described cellular experiments indicating that cirmtuzumab might be effective against cancer stem cells, which appear responsible for the relapse and spread of cancer after conventional therapy.
The latest research more precisely defines the roles of ROR1 and ROR2 in CLL development.
Both are evolutionarily conserved proteins that are found in many species and are most active in the early stages of embryogenesis when cells are migrating to form organs and parts of the body. The lack of either during this process results in severe developmental abnormalities.
Low levels of ROR2 remain in some adult tissues, but ROR1 is found only in cancer cells. The investigators found that, in response to signaling by Wnt5a, ROR1 and ROR2 come together to signal the growth and migration of CLL cells.
But treating mice with cirmtuzumab disrupted the process, inhibiting the engraftment of CLL cells and slowing or stopping the disease from spreading. ![]()
Anemia tied to cognitive impairment

A population-based study conducted in Germany has suggested a link between anemia and mild cognitive impairment (MCI).
Researchers found that subjects with anemia, defined as hemoglobin <13 g/dL in men and <12 g/dL in women, performed worse on cognitive tests than their nonanemic peers.
And MCI occurred almost twice as often in subjects with anemia than in subjects with normal hemoglobin levels.
This study was published in the Journal of Alzheimer’s Disease.
About MCI
MCI represents an intermediate and possibly modifiable stage between normal cognitive aging and dementia. Although individuals with MCI have an increased risk of developing dementia or Alzheimer’s disease, they can also remain stable for many years or even revert to a cognitively normal state over time. This modifiable characteristic makes the concept of MCI a promising target in the prevention of dementia.
The following 4 criteria are used to diagnose MCI. First, subjects must report a decline in cognitive performance over the past 2 years. Second, they must show a cognitive impairment in objective cognitive tasks that is greater than one would expect taking their age and education into consideration.
Third, the impairment must not be as pronounced as in demented individuals since people with MCI can perform normal daily living activities or are only slightly impaired in carrying out complex instrumental functions. Fourth, the cognitive impairment has to be insufficient to fulfil criteria for dementia.
The concept of MCI distinguishes between amnestic MCI (aMCI) and non-amnestic MCI (naMCI). In the former, impairment in the memory domain is evident, most likely reflecting Alzheimer’s disease pathology. In the latter, impairment in non-memory domains is present, mainly reflecting vascular pathology but also frontotemporal dementia or dementia with Lewy bodies.
Study details
The Heinz Nixdorf Recall study is an observational, population-based, prospective study in which researchers examined 4814 subjects between 2000 and 2003. Subjects were 50 to 80 years of age and lived in the metropolitan Ruhr Area. Both genders were equally represented.
After 5 years, the researchers conducted a second examination with 92% of the subjects taking part. The publication includes cross-sectional results of the second examination.
First, 163 subjects with anemia and 3870 without anemia were included to compare their performance in all cognitive subtests.
The subjects took verbal memory tests, which were used to gauge immediate recall and delayed recall. They were also tested on executive functioning, which included problem-solving/speed of processing, verbal fluency, visual spatial organization, and the clock drawing test.
In the initial analysis, anemic subjects showed more pronounced cardiovascular risk profiles and lower cognitive performance in all administered cognitive subtests. After adjusting for age, anemic subjects showed a significantly lower performance in the immediate recall task (P=0.009) and the verbal fluency task (P=0.004).
Next, the researchers compared 579 subjects diagnosed with MCI—299 with aMCI and 280 with naMCI—to 1438 cognitively normal subjects to determine the association between anemia at follow-up and MCI.
The team found that MCI occurred more often in anemic than non-anemic subjects. The unadjusted odds ratio (OR) was 2.59 (P<0.001). The OR after adjustment for age, gender, and years of education was 2.15 (P=0.002).
In a third analysis, the researchers adjusted for the aforementioned variables as well as body mass index, high-sensitivity C-reactive protein, glomerular filtration rate, cholesterol, serum iron, hypertension, diabetes mellitus, history of coronary heart disease, history of stroke, history of cancer, APOE4, smoking, severe depressive symptoms, and use of antidepressants. The OR after adjustment for these factors was 1.92 (P=0.04).
Similar results were found for aMCI and naMCI. The researchers said this suggests that having a low hemoglobin level may contribute to cognitive impairment via different pathways.
The team believes that, overall, their study results indicate that anemia is associated with an increased risk of MCI independent of traditional cardiovascular risk factors. They said the association between anemia and MCI has important clinical relevance because—depending on etiology—anemia can be treated effectively, and this might provide means to prevent or delay cognitive decline. ![]()

A population-based study conducted in Germany has suggested a link between anemia and mild cognitive impairment (MCI).
Researchers found that subjects with anemia, defined as hemoglobin <13 g/dL in men and <12 g/dL in women, performed worse on cognitive tests than their nonanemic peers.
And MCI occurred almost twice as often in subjects with anemia than in subjects with normal hemoglobin levels.
This study was published in the Journal of Alzheimer’s Disease.
About MCI
MCI represents an intermediate and possibly modifiable stage between normal cognitive aging and dementia. Although individuals with MCI have an increased risk of developing dementia or Alzheimer’s disease, they can also remain stable for many years or even revert to a cognitively normal state over time. This modifiable characteristic makes the concept of MCI a promising target in the prevention of dementia.
The following 4 criteria are used to diagnose MCI. First, subjects must report a decline in cognitive performance over the past 2 years. Second, they must show a cognitive impairment in objective cognitive tasks that is greater than one would expect taking their age and education into consideration.
Third, the impairment must not be as pronounced as in demented individuals since people with MCI can perform normal daily living activities or are only slightly impaired in carrying out complex instrumental functions. Fourth, the cognitive impairment has to be insufficient to fulfil criteria for dementia.
The concept of MCI distinguishes between amnestic MCI (aMCI) and non-amnestic MCI (naMCI). In the former, impairment in the memory domain is evident, most likely reflecting Alzheimer’s disease pathology. In the latter, impairment in non-memory domains is present, mainly reflecting vascular pathology but also frontotemporal dementia or dementia with Lewy bodies.
Study details
The Heinz Nixdorf Recall study is an observational, population-based, prospective study in which researchers examined 4814 subjects between 2000 and 2003. Subjects were 50 to 80 years of age and lived in the metropolitan Ruhr Area. Both genders were equally represented.
After 5 years, the researchers conducted a second examination with 92% of the subjects taking part. The publication includes cross-sectional results of the second examination.
First, 163 subjects with anemia and 3870 without anemia were included to compare their performance in all cognitive subtests.
The subjects took verbal memory tests, which were used to gauge immediate recall and delayed recall. They were also tested on executive functioning, which included problem-solving/speed of processing, verbal fluency, visual spatial organization, and the clock drawing test.
In the initial analysis, anemic subjects showed more pronounced cardiovascular risk profiles and lower cognitive performance in all administered cognitive subtests. After adjusting for age, anemic subjects showed a significantly lower performance in the immediate recall task (P=0.009) and the verbal fluency task (P=0.004).
Next, the researchers compared 579 subjects diagnosed with MCI—299 with aMCI and 280 with naMCI—to 1438 cognitively normal subjects to determine the association between anemia at follow-up and MCI.
The team found that MCI occurred more often in anemic than non-anemic subjects. The unadjusted odds ratio (OR) was 2.59 (P<0.001). The OR after adjustment for age, gender, and years of education was 2.15 (P=0.002).
In a third analysis, the researchers adjusted for the aforementioned variables as well as body mass index, high-sensitivity C-reactive protein, glomerular filtration rate, cholesterol, serum iron, hypertension, diabetes mellitus, history of coronary heart disease, history of stroke, history of cancer, APOE4, smoking, severe depressive symptoms, and use of antidepressants. The OR after adjustment for these factors was 1.92 (P=0.04).
Similar results were found for aMCI and naMCI. The researchers said this suggests that having a low hemoglobin level may contribute to cognitive impairment via different pathways.
The team believes that, overall, their study results indicate that anemia is associated with an increased risk of MCI independent of traditional cardiovascular risk factors. They said the association between anemia and MCI has important clinical relevance because—depending on etiology—anemia can be treated effectively, and this might provide means to prevent or delay cognitive decline. ![]()

A population-based study conducted in Germany has suggested a link between anemia and mild cognitive impairment (MCI).
Researchers found that subjects with anemia, defined as hemoglobin <13 g/dL in men and <12 g/dL in women, performed worse on cognitive tests than their nonanemic peers.
And MCI occurred almost twice as often in subjects with anemia than in subjects with normal hemoglobin levels.
This study was published in the Journal of Alzheimer’s Disease.
About MCI
MCI represents an intermediate and possibly modifiable stage between normal cognitive aging and dementia. Although individuals with MCI have an increased risk of developing dementia or Alzheimer’s disease, they can also remain stable for many years or even revert to a cognitively normal state over time. This modifiable characteristic makes the concept of MCI a promising target in the prevention of dementia.
The following 4 criteria are used to diagnose MCI. First, subjects must report a decline in cognitive performance over the past 2 years. Second, they must show a cognitive impairment in objective cognitive tasks that is greater than one would expect taking their age and education into consideration.
Third, the impairment must not be as pronounced as in demented individuals since people with MCI can perform normal daily living activities or are only slightly impaired in carrying out complex instrumental functions. Fourth, the cognitive impairment has to be insufficient to fulfil criteria for dementia.
The concept of MCI distinguishes between amnestic MCI (aMCI) and non-amnestic MCI (naMCI). In the former, impairment in the memory domain is evident, most likely reflecting Alzheimer’s disease pathology. In the latter, impairment in non-memory domains is present, mainly reflecting vascular pathology but also frontotemporal dementia or dementia with Lewy bodies.
Study details
The Heinz Nixdorf Recall study is an observational, population-based, prospective study in which researchers examined 4814 subjects between 2000 and 2003. Subjects were 50 to 80 years of age and lived in the metropolitan Ruhr Area. Both genders were equally represented.
After 5 years, the researchers conducted a second examination with 92% of the subjects taking part. The publication includes cross-sectional results of the second examination.
First, 163 subjects with anemia and 3870 without anemia were included to compare their performance in all cognitive subtests.
The subjects took verbal memory tests, which were used to gauge immediate recall and delayed recall. They were also tested on executive functioning, which included problem-solving/speed of processing, verbal fluency, visual spatial organization, and the clock drawing test.
In the initial analysis, anemic subjects showed more pronounced cardiovascular risk profiles and lower cognitive performance in all administered cognitive subtests. After adjusting for age, anemic subjects showed a significantly lower performance in the immediate recall task (P=0.009) and the verbal fluency task (P=0.004).
Next, the researchers compared 579 subjects diagnosed with MCI—299 with aMCI and 280 with naMCI—to 1438 cognitively normal subjects to determine the association between anemia at follow-up and MCI.
The team found that MCI occurred more often in anemic than non-anemic subjects. The unadjusted odds ratio (OR) was 2.59 (P<0.001). The OR after adjustment for age, gender, and years of education was 2.15 (P=0.002).
In a third analysis, the researchers adjusted for the aforementioned variables as well as body mass index, high-sensitivity C-reactive protein, glomerular filtration rate, cholesterol, serum iron, hypertension, diabetes mellitus, history of coronary heart disease, history of stroke, history of cancer, APOE4, smoking, severe depressive symptoms, and use of antidepressants. The OR after adjustment for these factors was 1.92 (P=0.04).
Similar results were found for aMCI and naMCI. The researchers said this suggests that having a low hemoglobin level may contribute to cognitive impairment via different pathways.
The team believes that, overall, their study results indicate that anemia is associated with an increased risk of MCI independent of traditional cardiovascular risk factors. They said the association between anemia and MCI has important clinical relevance because—depending on etiology—anemia can be treated effectively, and this might provide means to prevent or delay cognitive decline. ![]()
CHMP recommends FVIII product for hemophilia A

The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended BAY 81-8973, a recombinant factor VIII (FVIII) compound, for approval in the European Union.
BAY 81-8973 is an unmodified, full-length, recombinant FVIII compound intended for the treatment and prophylaxis of bleeding in hemophilia A patients of all ages.
The CHMP’s recommendation has been relayed to the European Commission, which is expected to make a decision regarding BAY 81-8973 in the coming weeks.
If approved, BAY 81-8973 will be marketed for the aforementioned indication in the 28 member countries of the European Union, as well as Iceland, Liechtenstein, and Norway.
The CHMP’s recommendation is based on results from the LEOPOLD trials, which were presented at the National Hemophilia Foundation’s 67th Annual Meeting last August.
LEOPOLD trials
The LEOPOLD Clinical Development Program consists of 3 multinational clinical trials designed to evaluate the pharmacokinetics, efficacy, and safety of BAY 81-8973 in subjects with severe hemophilia A (<1% FVIII:C).
LEOPOLD I is an open-label, cross-over, phase 3 study of males ages 12 to 65. The objectives were to demonstrate the efficacy and safety of BAY 81-8973 when used as prophylaxis, for the treatment of bleeding episodes, and for maintaining hemostasis during surgery. In LEOPOLD I, investigators assigned subjects to either the 2- or 3-times-weekly dosing regimens based on each patient’s phenotype, prior bleeding history and other factors.
LEOPOLD II is a randomized, cross-over, open-label trial conducted in male subjects ages 12 to 65. In this phase 3 study, 80 subjects were randomized to receive BAY 81-8973 either as a low-dose prophylaxis regimen (20-30 IU/kg; n=28) twice per week, high-dose prophylaxis (30-40 IU/kg; n=31) 3 times a week, or on-demand (n=21).
The primary objective was to demonstrate the superiority of prophylaxis over on-demand therapy, with the primary endpoint being bleeding frequency at 12 months.
LEOPOLD Kids is an open-label, non-randomized, phase 3 study. Part A is designed to evaluate the efficacy and safety of BAY 81-8973 for prophylaxis, treatment of bleeds, and surgical management in previously treated children at least 12 years of age with twice or 3 times per week or every other day prophylaxis regimens. Part B of the study, which involves previously untreated patients, is ongoing.
Bayer HealthCare AG has submitted marketing applications for BAY 81-8973 in the US and several other countries and is pursuing regulatory approvals worldwide. ![]()

The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended BAY 81-8973, a recombinant factor VIII (FVIII) compound, for approval in the European Union.
BAY 81-8973 is an unmodified, full-length, recombinant FVIII compound intended for the treatment and prophylaxis of bleeding in hemophilia A patients of all ages.
The CHMP’s recommendation has been relayed to the European Commission, which is expected to make a decision regarding BAY 81-8973 in the coming weeks.
If approved, BAY 81-8973 will be marketed for the aforementioned indication in the 28 member countries of the European Union, as well as Iceland, Liechtenstein, and Norway.
The CHMP’s recommendation is based on results from the LEOPOLD trials, which were presented at the National Hemophilia Foundation’s 67th Annual Meeting last August.
LEOPOLD trials
The LEOPOLD Clinical Development Program consists of 3 multinational clinical trials designed to evaluate the pharmacokinetics, efficacy, and safety of BAY 81-8973 in subjects with severe hemophilia A (<1% FVIII:C).
LEOPOLD I is an open-label, cross-over, phase 3 study of males ages 12 to 65. The objectives were to demonstrate the efficacy and safety of BAY 81-8973 when used as prophylaxis, for the treatment of bleeding episodes, and for maintaining hemostasis during surgery. In LEOPOLD I, investigators assigned subjects to either the 2- or 3-times-weekly dosing regimens based on each patient’s phenotype, prior bleeding history and other factors.
LEOPOLD II is a randomized, cross-over, open-label trial conducted in male subjects ages 12 to 65. In this phase 3 study, 80 subjects were randomized to receive BAY 81-8973 either as a low-dose prophylaxis regimen (20-30 IU/kg; n=28) twice per week, high-dose prophylaxis (30-40 IU/kg; n=31) 3 times a week, or on-demand (n=21).
The primary objective was to demonstrate the superiority of prophylaxis over on-demand therapy, with the primary endpoint being bleeding frequency at 12 months.
LEOPOLD Kids is an open-label, non-randomized, phase 3 study. Part A is designed to evaluate the efficacy and safety of BAY 81-8973 for prophylaxis, treatment of bleeds, and surgical management in previously treated children at least 12 years of age with twice or 3 times per week or every other day prophylaxis regimens. Part B of the study, which involves previously untreated patients, is ongoing.
Bayer HealthCare AG has submitted marketing applications for BAY 81-8973 in the US and several other countries and is pursuing regulatory approvals worldwide. ![]()

The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended BAY 81-8973, a recombinant factor VIII (FVIII) compound, for approval in the European Union.
BAY 81-8973 is an unmodified, full-length, recombinant FVIII compound intended for the treatment and prophylaxis of bleeding in hemophilia A patients of all ages.
The CHMP’s recommendation has been relayed to the European Commission, which is expected to make a decision regarding BAY 81-8973 in the coming weeks.
If approved, BAY 81-8973 will be marketed for the aforementioned indication in the 28 member countries of the European Union, as well as Iceland, Liechtenstein, and Norway.
The CHMP’s recommendation is based on results from the LEOPOLD trials, which were presented at the National Hemophilia Foundation’s 67th Annual Meeting last August.
LEOPOLD trials
The LEOPOLD Clinical Development Program consists of 3 multinational clinical trials designed to evaluate the pharmacokinetics, efficacy, and safety of BAY 81-8973 in subjects with severe hemophilia A (<1% FVIII:C).
LEOPOLD I is an open-label, cross-over, phase 3 study of males ages 12 to 65. The objectives were to demonstrate the efficacy and safety of BAY 81-8973 when used as prophylaxis, for the treatment of bleeding episodes, and for maintaining hemostasis during surgery. In LEOPOLD I, investigators assigned subjects to either the 2- or 3-times-weekly dosing regimens based on each patient’s phenotype, prior bleeding history and other factors.
LEOPOLD II is a randomized, cross-over, open-label trial conducted in male subjects ages 12 to 65. In this phase 3 study, 80 subjects were randomized to receive BAY 81-8973 either as a low-dose prophylaxis regimen (20-30 IU/kg; n=28) twice per week, high-dose prophylaxis (30-40 IU/kg; n=31) 3 times a week, or on-demand (n=21).
The primary objective was to demonstrate the superiority of prophylaxis over on-demand therapy, with the primary endpoint being bleeding frequency at 12 months.
LEOPOLD Kids is an open-label, non-randomized, phase 3 study. Part A is designed to evaluate the efficacy and safety of BAY 81-8973 for prophylaxis, treatment of bleeds, and surgical management in previously treated children at least 12 years of age with twice or 3 times per week or every other day prophylaxis regimens. Part B of the study, which involves previously untreated patients, is ongoing.
Bayer HealthCare AG has submitted marketing applications for BAY 81-8973 in the US and several other countries and is pursuing regulatory approvals worldwide. ![]()
A microfluidic biochip for blood cell counts

Researchers say they have created a biosensor capable of counting blood cells electrically using only a drop of blood.
The microfluidic device can measure red blood cell, platelet, and white blood cell counts using as little as 11 µL of blood.
The device electrically counts the different types of blood cells based on their size and membrane properties.
To count leukocyte and its differentials, red blood cells are selectively lysed, and the remaining white blood cells are individually counted. Specific cells like neutrophils can be counted using multi-frequency analysis, which probes the membrane properties of the cells.
For red blood cells and platelets, 1 µL of whole blood is diluted with PBS on-chip, and the cells are counted electrically. The total time for measurement is under 20 minutes.
The researchers described this device in TECHNOLOGY.
“Our biosensor exhibits the potential to improve patient care in a spectrum of settings,” said Rashid Bashir, PhD, of the University of Illinois at Urbana-Champaign.
He noted that the device could be particularly useful in resource-limited settings where laboratory tests are often inaccessible due to costs, poor prevalence of laboratory facilities, and the difficulty of follow-up upon receiving results that take days to process.
“There exists a huge potential to translate our biosensor commercially for blood cell count applications,” added Umer Hassan, PhD, of the University of Illinois at Urbana-Champaign.
“The translation of our technology will result in minimal to no experience requirement for device operation. Even patients can perform the test at the comfort of their home and share the results with their primary care physicians via electronic means too.”
“The technology is scalable, and, in future, we plan to apply it to many other potential applications in the areas of animal diagnostics, blood transfusion analysis, ER/ICU applications, and blood cell counting for chemotherapy management,” Dr Bashir said.
The researchers are now working to further develop a portable prototype of the cell counter.
“The cartridges will be disposable and the size of a credit card,” Dr Umer said. “The base unit or the reader will be portable and possibly hand-held. Our technology has the potential to reduce the cost of the test to less than $10, as compared to $100 or more currently charged.” ![]()

Researchers say they have created a biosensor capable of counting blood cells electrically using only a drop of blood.
The microfluidic device can measure red blood cell, platelet, and white blood cell counts using as little as 11 µL of blood.
The device electrically counts the different types of blood cells based on their size and membrane properties.
To count leukocyte and its differentials, red blood cells are selectively lysed, and the remaining white blood cells are individually counted. Specific cells like neutrophils can be counted using multi-frequency analysis, which probes the membrane properties of the cells.
For red blood cells and platelets, 1 µL of whole blood is diluted with PBS on-chip, and the cells are counted electrically. The total time for measurement is under 20 minutes.
The researchers described this device in TECHNOLOGY.
“Our biosensor exhibits the potential to improve patient care in a spectrum of settings,” said Rashid Bashir, PhD, of the University of Illinois at Urbana-Champaign.
He noted that the device could be particularly useful in resource-limited settings where laboratory tests are often inaccessible due to costs, poor prevalence of laboratory facilities, and the difficulty of follow-up upon receiving results that take days to process.
“There exists a huge potential to translate our biosensor commercially for blood cell count applications,” added Umer Hassan, PhD, of the University of Illinois at Urbana-Champaign.
“The translation of our technology will result in minimal to no experience requirement for device operation. Even patients can perform the test at the comfort of their home and share the results with their primary care physicians via electronic means too.”
“The technology is scalable, and, in future, we plan to apply it to many other potential applications in the areas of animal diagnostics, blood transfusion analysis, ER/ICU applications, and blood cell counting for chemotherapy management,” Dr Bashir said.
The researchers are now working to further develop a portable prototype of the cell counter.
“The cartridges will be disposable and the size of a credit card,” Dr Umer said. “The base unit or the reader will be portable and possibly hand-held. Our technology has the potential to reduce the cost of the test to less than $10, as compared to $100 or more currently charged.” ![]()

Researchers say they have created a biosensor capable of counting blood cells electrically using only a drop of blood.
The microfluidic device can measure red blood cell, platelet, and white blood cell counts using as little as 11 µL of blood.
The device electrically counts the different types of blood cells based on their size and membrane properties.
To count leukocyte and its differentials, red blood cells are selectively lysed, and the remaining white blood cells are individually counted. Specific cells like neutrophils can be counted using multi-frequency analysis, which probes the membrane properties of the cells.
For red blood cells and platelets, 1 µL of whole blood is diluted with PBS on-chip, and the cells are counted electrically. The total time for measurement is under 20 minutes.
The researchers described this device in TECHNOLOGY.
“Our biosensor exhibits the potential to improve patient care in a spectrum of settings,” said Rashid Bashir, PhD, of the University of Illinois at Urbana-Champaign.
He noted that the device could be particularly useful in resource-limited settings where laboratory tests are often inaccessible due to costs, poor prevalence of laboratory facilities, and the difficulty of follow-up upon receiving results that take days to process.
“There exists a huge potential to translate our biosensor commercially for blood cell count applications,” added Umer Hassan, PhD, of the University of Illinois at Urbana-Champaign.
“The translation of our technology will result in minimal to no experience requirement for device operation. Even patients can perform the test at the comfort of their home and share the results with their primary care physicians via electronic means too.”
“The technology is scalable, and, in future, we plan to apply it to many other potential applications in the areas of animal diagnostics, blood transfusion analysis, ER/ICU applications, and blood cell counting for chemotherapy management,” Dr Bashir said.
The researchers are now working to further develop a portable prototype of the cell counter.
“The cartridges will be disposable and the size of a credit card,” Dr Umer said. “The base unit or the reader will be portable and possibly hand-held. Our technology has the potential to reduce the cost of the test to less than $10, as compared to $100 or more currently charged.” ![]()
Myeloma drug could treat sickle cell disease, team says

and a normal one
Image by Betty Pace
Preclinical research suggests a drug used to treat multiple myeloma (MM) might also prove effective in the treatment of sickle cell disease (SCD).
Researchers say this study is the first to reveal how the drug, pomalidomide, increases the production of fetal hemoglobin, which is known to interfere with the sickling of red blood cells.
“We knew the drug would make fetal hemoglobin, but we didn’t know to what extent or how,” said Lionel Blanc, PhD, of the Feinstein Institute for Medical Research in Manhasset, New York.
He and his colleagues reported their findings in Blood.
The researchers’ in vitro experiments revealed that pomalidomide reverses γ-globin silencing during adult erythropoiesis, and the drug delays the maturation of early erythroid precursors without impairing terminal differentiation.
The team also found that pomalidomide selectively targets BCL11A and SOX6 to induce γ-globin synthesis, the drug’s mechanism of action during erythropoiesis is independent of IKZF1 degradation, and pomalidomide partially reprograms adult erythroid progenitors to a fetal-like state.
Finally, the researchers discovered that pomalidomide’s mechanism of action is conserved in cells from SCD patients, and treatment with pomalidomide leads to γ-globin production in MM patients.
“We can also say something else—that hydroxyurea, the only FDA-approved drug for sickle cell anemia, was less effective than pomalidomide and appeared to act through a different mechanism of action,” Dr Blanc said.
“The current therapy is good, but not everyone responds equally to hydroxyurea, and what we hope with pomalidomide is to improve this.”
Dr Blanc and his colleagues plan to launch a clinical trial in the near future to test pomalidomide in young adults with SCD. ![]()

and a normal one
Image by Betty Pace
Preclinical research suggests a drug used to treat multiple myeloma (MM) might also prove effective in the treatment of sickle cell disease (SCD).
Researchers say this study is the first to reveal how the drug, pomalidomide, increases the production of fetal hemoglobin, which is known to interfere with the sickling of red blood cells.
“We knew the drug would make fetal hemoglobin, but we didn’t know to what extent or how,” said Lionel Blanc, PhD, of the Feinstein Institute for Medical Research in Manhasset, New York.
He and his colleagues reported their findings in Blood.
The researchers’ in vitro experiments revealed that pomalidomide reverses γ-globin silencing during adult erythropoiesis, and the drug delays the maturation of early erythroid precursors without impairing terminal differentiation.
The team also found that pomalidomide selectively targets BCL11A and SOX6 to induce γ-globin synthesis, the drug’s mechanism of action during erythropoiesis is independent of IKZF1 degradation, and pomalidomide partially reprograms adult erythroid progenitors to a fetal-like state.
Finally, the researchers discovered that pomalidomide’s mechanism of action is conserved in cells from SCD patients, and treatment with pomalidomide leads to γ-globin production in MM patients.
“We can also say something else—that hydroxyurea, the only FDA-approved drug for sickle cell anemia, was less effective than pomalidomide and appeared to act through a different mechanism of action,” Dr Blanc said.
“The current therapy is good, but not everyone responds equally to hydroxyurea, and what we hope with pomalidomide is to improve this.”
Dr Blanc and his colleagues plan to launch a clinical trial in the near future to test pomalidomide in young adults with SCD. ![]()

and a normal one
Image by Betty Pace
Preclinical research suggests a drug used to treat multiple myeloma (MM) might also prove effective in the treatment of sickle cell disease (SCD).
Researchers say this study is the first to reveal how the drug, pomalidomide, increases the production of fetal hemoglobin, which is known to interfere with the sickling of red blood cells.
“We knew the drug would make fetal hemoglobin, but we didn’t know to what extent or how,” said Lionel Blanc, PhD, of the Feinstein Institute for Medical Research in Manhasset, New York.
He and his colleagues reported their findings in Blood.
The researchers’ in vitro experiments revealed that pomalidomide reverses γ-globin silencing during adult erythropoiesis, and the drug delays the maturation of early erythroid precursors without impairing terminal differentiation.
The team also found that pomalidomide selectively targets BCL11A and SOX6 to induce γ-globin synthesis, the drug’s mechanism of action during erythropoiesis is independent of IKZF1 degradation, and pomalidomide partially reprograms adult erythroid progenitors to a fetal-like state.
Finally, the researchers discovered that pomalidomide’s mechanism of action is conserved in cells from SCD patients, and treatment with pomalidomide leads to γ-globin production in MM patients.
“We can also say something else—that hydroxyurea, the only FDA-approved drug for sickle cell anemia, was less effective than pomalidomide and appeared to act through a different mechanism of action,” Dr Blanc said.
“The current therapy is good, but not everyone responds equally to hydroxyurea, and what we hope with pomalidomide is to improve this.”
Dr Blanc and his colleagues plan to launch a clinical trial in the near future to test pomalidomide in young adults with SCD.
Fear really does curdle blood, study suggests

Photo by Graham Colm
Horror movies may actually be “blood-curdling,” according to a study published in The BMJ.
The research showed that watching a scary movie was associated with an increase in the clotting protein factor VIII (FVIII).
Study subjects were more likely to experience an increase in FVIII while watching a horror film than an educational film.
So using the term “blood-curdling” to describe feeling extreme fear may be justified, researchers said.
The term dates back to medieval times and is based on the concept that fear would “run the blood cold” or “curdle” the blood, but the validity of this theory has never been studied.
So Banne Nemeth, MD, of Leiden University Medical Centre in The Netherlands, and his colleagues set out to assess whether acute fear can curdle blood.
Their study involved 24 healthy volunteers age 30 or younger. Fourteen subjects were assigned to watch a frightening movie followed by a non-threatening educational movie, and 10 were assigned to watch the movies in reverse order.
The movies were viewed more than a week apart at the same time of day and in a comfortable and relaxed environment. Both lasted approximately 90 minutes.
Before and after each movie (within 15 minutes), blood samples were taken and analyzed for markers of clotting activity.
After each movie, participants rated the fear they experienced using a visual analogue fear scale ranging from 0 (no fear at all) to 10 (worst fear imaginable). They also reported whether they had already seen the movie and completed a general questionnaire on lifestyle and favorite movie genre.
The horror movie was perceived to be more frightening than the educational movie, with a 5.4 mean difference in fear rating scores.
The difference in FVIII levels before and after watching the movies was higher for the horror movie than for the educational movie. The mean difference of differences was 11.1 IU/dL (95% confidence interval: 1.2 to 21.0 IU/dL).
FVIII levels increased in 12 (57%) subjects during the horror movie and in 3 (14%) during the educational movie. FVIII levels decreased in 18 (86%) participants during the educational movie and in 9 (43%) during the horror movie.
However, the researchers found no effect of either movie on thrombin-antithrombin complexes, D-dimer, or prothrombin fragments 1+2. They said this suggests that although acute fear may trigger coagulation, it does not lead to actual clot formation.
The team pointed out some study limitations but concluded that, in young and healthy adults, “watching blood-curdling movies is associated with an increase in blood coagulant factor VIII without actual thrombin formation.”
The researchers believe this phenomenon may confer an important evolutionary benefit by preparing the body for blood loss during life-threatening situations.

Photo by Graham Colm
Horror movies may actually be “blood-curdling,” according to a study published in The BMJ.
The research showed that watching a scary movie was associated with an increase in the clotting protein factor VIII (FVIII).
Study subjects were more likely to experience an increase in FVIII while watching a horror film than an educational film.
So using the term “blood-curdling” to describe feeling extreme fear may be justified, researchers said.
The term dates back to medieval times and is based on the concept that fear would “run the blood cold” or “curdle” the blood, but the validity of this theory has never been studied.
So Banne Nemeth, MD, of Leiden University Medical Centre in The Netherlands, and his colleagues set out to assess whether acute fear can curdle blood.
Their study involved 24 healthy volunteers age 30 or younger. Fourteen subjects were assigned to watch a frightening movie followed by a non-threatening educational movie, and 10 were assigned to watch the movies in reverse order.
The movies were viewed more than a week apart at the same time of day and in a comfortable and relaxed environment. Both lasted approximately 90 minutes.
Before and after each movie (within 15 minutes), blood samples were taken and analyzed for markers of clotting activity.
After each movie, participants rated the fear they experienced using a visual analogue fear scale ranging from 0 (no fear at all) to 10 (worst fear imaginable). They also reported whether they had already seen the movie and completed a general questionnaire on lifestyle and favorite movie genre.
The horror movie was perceived to be more frightening than the educational movie, with a 5.4 mean difference in fear rating scores.
The difference in FVIII levels before and after watching the movies was higher for the horror movie than for the educational movie. The mean difference of differences was 11.1 IU/dL (95% confidence interval: 1.2 to 21.0 IU/dL).
FVIII levels increased in 12 (57%) subjects during the horror movie and in 3 (14%) during the educational movie. FVIII levels decreased in 18 (86%) participants during the educational movie and in 9 (43%) during the horror movie.
However, the researchers found no effect of either movie on thrombin-antithrombin complexes, D-dimer, or prothrombin fragments 1+2. They said this suggests that although acute fear may trigger coagulation, it does not lead to actual clot formation.
The team pointed out some study limitations but concluded that, in young and healthy adults, “watching blood-curdling movies is associated with an increase in blood coagulant factor VIII without actual thrombin formation.”
The researchers believe this phenomenon may confer an important evolutionary benefit by preparing the body for blood loss during life-threatening situations.

Photo by Graham Colm
Horror movies may actually be “blood-curdling,” according to a study published in The BMJ.
The research showed that watching a scary movie was associated with an increase in the clotting protein factor VIII (FVIII).
Study subjects were more likely to experience an increase in FVIII while watching a horror film than an educational film.
So using the term “blood-curdling” to describe feeling extreme fear may be justified, researchers said.
The term dates back to medieval times and is based on the concept that fear would “run the blood cold” or “curdle” the blood, but the validity of this theory has never been studied.
So Banne Nemeth, MD, of Leiden University Medical Centre in The Netherlands, and his colleagues set out to assess whether acute fear can curdle blood.
Their study involved 24 healthy volunteers age 30 or younger. Fourteen subjects were assigned to watch a frightening movie followed by a non-threatening educational movie, and 10 were assigned to watch the movies in reverse order.
The movies were viewed more than a week apart at the same time of day and in a comfortable and relaxed environment. Both lasted approximately 90 minutes.
Before and after each movie (within 15 minutes), blood samples were taken and analyzed for markers of clotting activity.
After each movie, participants rated the fear they experienced using a visual analogue fear scale ranging from 0 (no fear at all) to 10 (worst fear imaginable). They also reported whether they had already seen the movie and completed a general questionnaire on lifestyle and favorite movie genre.
The horror movie was perceived to be more frightening than the educational movie, with a 5.4 mean difference in fear rating scores.
The difference in FVIII levels before and after watching the movies was higher for the horror movie than for the educational movie. The mean difference of differences was 11.1 IU/dL (95% confidence interval: 1.2 to 21.0 IU/dL).
FVIII levels increased in 12 (57%) subjects during the horror movie and in 3 (14%) during the educational movie. FVIII levels decreased in 18 (86%) participants during the educational movie and in 9 (43%) during the horror movie.
However, the researchers found no effect of either movie on thrombin-antithrombin complexes, D-dimer, or prothrombin fragments 1+2. They said this suggests that although acute fear may trigger coagulation, it does not lead to actual clot formation.
The team pointed out some study limitations but concluded that, in young and healthy adults, “watching blood-curdling movies is associated with an increase in blood coagulant factor VIII without actual thrombin formation.”
The researchers believe this phenomenon may confer an important evolutionary benefit by preparing the body for blood loss during life-threatening situations.
A potential target for IDH1-mutant cancers
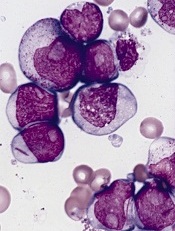
acute myeloid leukemia
Researchers say they have identified a potential therapeutic target for IDH1-mutant malignancies.
Preclinical experiments showed that tumors characterized by IDH1 mutations are “extremely vulnerable” to depletion of NAD+, a metabolic cofactor.
“Our finding . . . supports the proposal that medications that can decrease levels of this metabolite, which are in development, have the potential to specifically treat these cancers,” said Daniel Cahill, MD, PhD, of Massachusetts General Hospital in Boston.
Dr Cahill and his colleagues described their research in Cancer Cell.
IDH1 and IDH2 are known to play essential roles in cellular metabolism, including the processes by which cells convert glucose and other nutrients into ATP, providing the energy needed for cellular survival.
In 2008, researchers discovered that IDH1 mutations are involved in several cancers. The gene is mutated in around 20% of adult gliomas, in more than 10% of acute myeloid leukemias, and in a smaller percentage of other cancers.
One clear result of IDH1 mutation is a 100-fold elevation in levels of the metabolite 2-HG, which is known to mediate several properties that lead to tumor development. Drugs that decrease levels of 2-HG are currently under development.
Dr Cahill and his colleagues wanted to find additional ways of blocking the mutation’s effects, so they inhibited mutant IDH1 in tumor cells. They were surprised to find that this reduced 2-HG levels, but, in many cases, that reduction did not halt cell growth.
Detailed metabolic profiling of IDH1-mutant cells revealed that inhibiting the mutated enzyme greatly increased levels of NAD+, a cofactor that plays a role in several cellular energy processes.
Additional experiments showed that depletion of NAD+ induced the death of IDH1-mutant tumor cells and inhibited tumor growth in an animal model of glioma.
“Accumulation of excess 2-HG is known to drive several changes leading to tumor development, but our results indicate that simply depleting 2-HG levels was not sufficient to halt the growth of several types of later-stage IDH1-mutant tumors,” said Hiroaki Wakimoto, MD, PhD, of Massachusetts General Hospital.
“In addition, we found that mutant IDH1 reduces expression of an enzyme that maintains NAD+ levels, rendering IDH1-mutant tumor cells highly sensitive to direct NAD+ depletion. Several drugs that inhibit the synthesis of NAD+ are already in clinical trials, and these agents may prove useful for patients with IDH-mutant cancers. While we primarily focused on IDH1-mutant gliomas, we also found evidence that NAD+ inhibition could slow the growth of other types of cancer with this mutation.”

acute myeloid leukemia
Researchers say they have identified a potential therapeutic target for IDH1-mutant malignancies.
Preclinical experiments showed that tumors characterized by IDH1 mutations are “extremely vulnerable” to depletion of NAD+, a metabolic cofactor.
“Our finding . . . supports the proposal that medications that can decrease levels of this metabolite, which are in development, have the potential to specifically treat these cancers,” said Daniel Cahill, MD, PhD, of Massachusetts General Hospital in Boston.
Dr Cahill and his colleagues described their research in Cancer Cell.
IDH1 and IDH2 are known to play essential roles in cellular metabolism, including the processes by which cells convert glucose and other nutrients into ATP, providing the energy needed for cellular survival.
In 2008, researchers discovered that IDH1 mutations are involved in several cancers. The gene is mutated in around 20% of adult gliomas, in more than 10% of acute myeloid leukemias, and in a smaller percentage of other cancers.
One clear result of IDH1 mutation is a 100-fold elevation in levels of the metabolite 2-HG, which is known to mediate several properties that lead to tumor development. Drugs that decrease levels of 2-HG are currently under development.
Dr Cahill and his colleagues wanted to find additional ways of blocking the mutation’s effects, so they inhibited mutant IDH1 in tumor cells. They were surprised to find that this reduced 2-HG levels, but, in many cases, that reduction did not halt cell growth.
Detailed metabolic profiling of IDH1-mutant cells revealed that inhibiting the mutated enzyme greatly increased levels of NAD+, a cofactor that plays a role in several cellular energy processes.
Additional experiments showed that depletion of NAD+ induced the death of IDH1-mutant tumor cells and inhibited tumor growth in an animal model of glioma.
“Accumulation of excess 2-HG is known to drive several changes leading to tumor development, but our results indicate that simply depleting 2-HG levels was not sufficient to halt the growth of several types of later-stage IDH1-mutant tumors,” said Hiroaki Wakimoto, MD, PhD, of Massachusetts General Hospital.
“In addition, we found that mutant IDH1 reduces expression of an enzyme that maintains NAD+ levels, rendering IDH1-mutant tumor cells highly sensitive to direct NAD+ depletion. Several drugs that inhibit the synthesis of NAD+ are already in clinical trials, and these agents may prove useful for patients with IDH-mutant cancers. While we primarily focused on IDH1-mutant gliomas, we also found evidence that NAD+ inhibition could slow the growth of other types of cancer with this mutation.”

acute myeloid leukemia
Researchers say they have identified a potential therapeutic target for IDH1-mutant malignancies.
Preclinical experiments showed that tumors characterized by IDH1 mutations are “extremely vulnerable” to depletion of NAD+, a metabolic cofactor.
“Our finding . . . supports the proposal that medications that can decrease levels of this metabolite, which are in development, have the potential to specifically treat these cancers,” said Daniel Cahill, MD, PhD, of Massachusetts General Hospital in Boston.
Dr Cahill and his colleagues described their research in Cancer Cell.
IDH1 and IDH2 are known to play essential roles in cellular metabolism, including the processes by which cells convert glucose and other nutrients into ATP, providing the energy needed for cellular survival.
In 2008, researchers discovered that IDH1 mutations are involved in several cancers. The gene is mutated in around 20% of adult gliomas, in more than 10% of acute myeloid leukemias, and in a smaller percentage of other cancers.
One clear result of IDH1 mutation is a 100-fold elevation in levels of the metabolite 2-HG, which is known to mediate several properties that lead to tumor development. Drugs that decrease levels of 2-HG are currently under development.
Dr Cahill and his colleagues wanted to find additional ways of blocking the mutation’s effects, so they inhibited mutant IDH1 in tumor cells. They were surprised to find that this reduced 2-HG levels, but, in many cases, that reduction did not halt cell growth.
Detailed metabolic profiling of IDH1-mutant cells revealed that inhibiting the mutated enzyme greatly increased levels of NAD+, a cofactor that plays a role in several cellular energy processes.
Additional experiments showed that depletion of NAD+ induced the death of IDH1-mutant tumor cells and inhibited tumor growth in an animal model of glioma.
“Accumulation of excess 2-HG is known to drive several changes leading to tumor development, but our results indicate that simply depleting 2-HG levels was not sufficient to halt the growth of several types of later-stage IDH1-mutant tumors,” said Hiroaki Wakimoto, MD, PhD, of Massachusetts General Hospital.
“In addition, we found that mutant IDH1 reduces expression of an enzyme that maintains NAD+ levels, rendering IDH1-mutant tumor cells highly sensitive to direct NAD+ depletion. Several drugs that inhibit the synthesis of NAD+ are already in clinical trials, and these agents may prove useful for patients with IDH-mutant cancers. While we primarily focused on IDH1-mutant gliomas, we also found evidence that NAD+ inhibition could slow the growth of other types of cancer with this mutation.”
Antiplatelet agent shows promise in phase 1 trial

Image by Andre E.X. Brown
Results of a phase 1 study suggest the experimental agent PZ-128 provides rapid and reversible inhibition of platelet aggregation.
PZ-128 inhibited platelet aggregation in a cohort of patients who had vascular disease or multiple risk factors for coronary artery disease.
This effect was dose-dependent and diminished over time. The drug did not affect bleeding, coagulation, clinical chemistry, or ECG parameters.
Investigators reported these results in Arteriosclerosis, Thrombosis and Vascular Biology.
The team explained that PZ-128 is a protease-activated receptor-1 (PAR1)-based pepducin intended as an antiplatelet agent. Pepducins are membrane-tethered, cell-penetrating lipopeptides that target the cytoplasmic surface of their cognate receptor.
The current study is the first demonstration of pepducins’ potential benefits in humans, said study author Athan Kuliopulos, MD, PhD, of Tufts Medical Center in Boston, Massachusetts.
He and his colleagues tested PZ-128 in 31 patients, ages 43 to 74. The patients had vascular disease (22%)—coronary artery disease, previous myocardial infarction, etc.—or multiple coronary artery disease risk factors—dyslipidemia (81%), hypertension (69%), diabetes (34%), etc.
Many of the patients were taking other medications at baseline, including blood pressure medications (66%), aspirin (63%), lipid-lowering medications (56%), and diabetes medications (22%).
The patients received PZ-128 by 1- to 2-hour-long intravenous infusion at doses ranging from 0.01 mg/kg to 2 mg/kg. The investigators evaluated patients at baseline and 0.5, 1, 2, 6, and 24 hours after dosing, as well as 7 to 10 days after dosing.
PZ-128 had a dose-dependent inhibitory effect on platelet aggregation stimulated by the PAR1 agonist SFLLRN (8 µmol/L). At 30 minutes to 6 hours after dosing, the investigators observed 20% to 40% inhibition with PZ-128 at 0.3 mg/kg, 40% to 60% inhibition at 0.5 mg/kg, and 80% to 100% inhibition at 1 to 2 mg/kg.
Patients who were receiving aspirin in the 0.5 mg/kg and 1 mg/kg dose cohorts had 65% to 100% inhibition of final aggregation to SFLLRN at 30 minutes to 2 hours after dosing and 95% to 100% inhibition by 6 hours.
The impact of PZ-128 was reversible, with 50% recovery of aggregation to SFLLRN by 24 hours and near-complete recovery by 192 hours.
The investigators said PZ-128 did not have any significant effects on aggregation induced by AYPGKF, ADP, or collagen, which suggests the observed effects were specific to PAR1.
Likewise, the team said PZ-128 did not have any significant effects on bleeding, coagulation, clinical chemistry, or ECG parameters.
The investigators noted that there are currently no drugs on the market that target PAR1 and can be used during procedures when the risk of serious complications is high.
The PAR1 inhibitor vorapaxar is available for non-acute use in patients with a prior myocardial infarction or current peripheral artery disease. But the drug, whose effects build slowly and are long-lasting, was not approved for use during cardiac procedures due to a risk of excessive bleeding, Dr Kuliopulos said.
By contrast, PZ-128 appears able to block PAR1 fast enough to be used in an urgent procedure and for a time short enough to limit bleeding risk afterward, he said. Of course, more research is needed to confirm this.
To that end, the investigators are planning a phase 2 study of PZ-128 in up to 600 patients who have acute coronary syndromes or are undergoing percutaneous coronary intervention.

Image by Andre E.X. Brown
Results of a phase 1 study suggest the experimental agent PZ-128 provides rapid and reversible inhibition of platelet aggregation.
PZ-128 inhibited platelet aggregation in a cohort of patients who had vascular disease or multiple risk factors for coronary artery disease.
This effect was dose-dependent and diminished over time. The drug did not affect bleeding, coagulation, clinical chemistry, or ECG parameters.
Investigators reported these results in Arteriosclerosis, Thrombosis and Vascular Biology.
The team explained that PZ-128 is a protease-activated receptor-1 (PAR1)-based pepducin intended as an antiplatelet agent. Pepducins are membrane-tethered, cell-penetrating lipopeptides that target the cytoplasmic surface of their cognate receptor.
The current study is the first demonstration of pepducins’ potential benefits in humans, said study author Athan Kuliopulos, MD, PhD, of Tufts Medical Center in Boston, Massachusetts.
He and his colleagues tested PZ-128 in 31 patients, ages 43 to 74. The patients had vascular disease (22%)—coronary artery disease, previous myocardial infarction, etc.—or multiple coronary artery disease risk factors—dyslipidemia (81%), hypertension (69%), diabetes (34%), etc.
Many of the patients were taking other medications at baseline, including blood pressure medications (66%), aspirin (63%), lipid-lowering medications (56%), and diabetes medications (22%).
The patients received PZ-128 by 1- to 2-hour-long intravenous infusion at doses ranging from 0.01 mg/kg to 2 mg/kg. The investigators evaluated patients at baseline and 0.5, 1, 2, 6, and 24 hours after dosing, as well as 7 to 10 days after dosing.
PZ-128 had a dose-dependent inhibitory effect on platelet aggregation stimulated by the PAR1 agonist SFLLRN (8 µmol/L). At 30 minutes to 6 hours after dosing, the investigators observed 20% to 40% inhibition with PZ-128 at 0.3 mg/kg, 40% to 60% inhibition at 0.5 mg/kg, and 80% to 100% inhibition at 1 to 2 mg/kg.
Patients who were receiving aspirin in the 0.5 mg/kg and 1 mg/kg dose cohorts had 65% to 100% inhibition of final aggregation to SFLLRN at 30 minutes to 2 hours after dosing and 95% to 100% inhibition by 6 hours.
The impact of PZ-128 was reversible, with 50% recovery of aggregation to SFLLRN by 24 hours and near-complete recovery by 192 hours.
The investigators said PZ-128 did not have any significant effects on aggregation induced by AYPGKF, ADP, or collagen, which suggests the observed effects were specific to PAR1.
Likewise, the team said PZ-128 did not have any significant effects on bleeding, coagulation, clinical chemistry, or ECG parameters.
The investigators noted that there are currently no drugs on the market that target PAR1 and can be used during procedures when the risk of serious complications is high.
The PAR1 inhibitor vorapaxar is available for non-acute use in patients with a prior myocardial infarction or current peripheral artery disease. But the drug, whose effects build slowly and are long-lasting, was not approved for use during cardiac procedures due to a risk of excessive bleeding, Dr Kuliopulos said.
By contrast, PZ-128 appears able to block PAR1 fast enough to be used in an urgent procedure and for a time short enough to limit bleeding risk afterward, he said. Of course, more research is needed to confirm this.
To that end, the investigators are planning a phase 2 study of PZ-128 in up to 600 patients who have acute coronary syndromes or are undergoing percutaneous coronary intervention.

Image by Andre E.X. Brown
Results of a phase 1 study suggest the experimental agent PZ-128 provides rapid and reversible inhibition of platelet aggregation.
PZ-128 inhibited platelet aggregation in a cohort of patients who had vascular disease or multiple risk factors for coronary artery disease.
This effect was dose-dependent and diminished over time. The drug did not affect bleeding, coagulation, clinical chemistry, or ECG parameters.
Investigators reported these results in Arteriosclerosis, Thrombosis and Vascular Biology.
The team explained that PZ-128 is a protease-activated receptor-1 (PAR1)-based pepducin intended as an antiplatelet agent. Pepducins are membrane-tethered, cell-penetrating lipopeptides that target the cytoplasmic surface of their cognate receptor.
The current study is the first demonstration of pepducins’ potential benefits in humans, said study author Athan Kuliopulos, MD, PhD, of Tufts Medical Center in Boston, Massachusetts.
He and his colleagues tested PZ-128 in 31 patients, ages 43 to 74. The patients had vascular disease (22%)—coronary artery disease, previous myocardial infarction, etc.—or multiple coronary artery disease risk factors—dyslipidemia (81%), hypertension (69%), diabetes (34%), etc.
Many of the patients were taking other medications at baseline, including blood pressure medications (66%), aspirin (63%), lipid-lowering medications (56%), and diabetes medications (22%).
The patients received PZ-128 by 1- to 2-hour-long intravenous infusion at doses ranging from 0.01 mg/kg to 2 mg/kg. The investigators evaluated patients at baseline and 0.5, 1, 2, 6, and 24 hours after dosing, as well as 7 to 10 days after dosing.
PZ-128 had a dose-dependent inhibitory effect on platelet aggregation stimulated by the PAR1 agonist SFLLRN (8 µmol/L). At 30 minutes to 6 hours after dosing, the investigators observed 20% to 40% inhibition with PZ-128 at 0.3 mg/kg, 40% to 60% inhibition at 0.5 mg/kg, and 80% to 100% inhibition at 1 to 2 mg/kg.
Patients who were receiving aspirin in the 0.5 mg/kg and 1 mg/kg dose cohorts had 65% to 100% inhibition of final aggregation to SFLLRN at 30 minutes to 2 hours after dosing and 95% to 100% inhibition by 6 hours.
The impact of PZ-128 was reversible, with 50% recovery of aggregation to SFLLRN by 24 hours and near-complete recovery by 192 hours.
The investigators said PZ-128 did not have any significant effects on aggregation induced by AYPGKF, ADP, or collagen, which suggests the observed effects were specific to PAR1.
Likewise, the team said PZ-128 did not have any significant effects on bleeding, coagulation, clinical chemistry, or ECG parameters.
The investigators noted that there are currently no drugs on the market that target PAR1 and can be used during procedures when the risk of serious complications is high.
The PAR1 inhibitor vorapaxar is available for non-acute use in patients with a prior myocardial infarction or current peripheral artery disease. But the drug, whose effects build slowly and are long-lasting, was not approved for use during cardiac procedures due to a risk of excessive bleeding, Dr Kuliopulos said.
By contrast, PZ-128 appears able to block PAR1 fast enough to be used in an urgent procedure and for a time short enough to limit bleeding risk afterward, he said. Of course, more research is needed to confirm this.
To that end, the investigators are planning a phase 2 study of PZ-128 in up to 600 patients who have acute coronary syndromes or are undergoing percutaneous coronary intervention.