User login
Drug disappoints in phase 3 HSCT trial
The antiviral drug brincidofovir did not meet the primary endpoint of the phase 3 SUPPRESS trial, according to the drug’s developer, Chimerix.
Brincidofovir did not prevent clinically significant cytomegalovirus (CMV) infection through week 24 after hematopoietic stem cell transplant (HSCT).
However, the president and CEO of Chimerix said the company still believes there is a “viable path forward” for the drug.
Brincidofovir is an oral nucleotide analog that has shown in vitro antiviral activity against all 5 families of DNA viruses that affect humans, including herpes viruses and adenovirus.
The SUPPRESS trial, which was initiated in August 2013 and fully enrolled in June 2015, was informed by a successful phase 2 trial conducted in HSCT recipients.
The SUPPRESS trial enrolled and treated 452 adults who received allogeneic HSCTs from more than 40 transplant centers in the US, Canada, and Europe.
Subjects received twice-weekly brincidofovir or placebo (2:1 ratio) from the early post-transplant period through week 14 post-transplant, the period of highest risk for viral infections.
All patients in the trial were CMV-seropositive, placing them at high risk of CMV infection. The most common indications leading to HSCT were acute myelogenous leukemia (43% of patients), myelodysplasia (17%), non-Hodgkin lymphoma (10%), and acute lymphocytic leukemia (9%).
During the on-treatment period through week 14 after HSCT, fewer patients in the brincidofovir arm had a CMV infection, which was consistent with results from the phase 2 study of the drug.
However, during the 10 weeks off treatment from week 14 to week 24, there was an increase in CMV infections in the brincidofovir arm compared to the control arm. And there was a non-statistically significant increase in mortality in the brincidofovir arm compared to the control arm.
Preliminary analysis suggests the failure in preventing CMV infections and the increased mortality in the brincidofovir arm were driven by confirmed cases of graft-versus-host-disease (GVHD), which resulted in a significantly higher use of corticosteroids than in the control arm.
Both GVHD and the use of corticosteroids are risk factors for “late” CMV infection that occurs after the discontinuation of the antiviral in HSCT recipients.
The rate of study drug discontinuation for gastrointestinal events was less than 10% in this study, which is comparable to that observed in the phase 2 trial of brincidofovir in a similar HSCT population.
“While we are clearly disappointed in the top-line results from SUPPRESS, we remain committed to better understanding the full data set as we consider potential paths forward for brincidofovir,” said M. Michelle Berrey, MD, president and CEO of Chimerix.
“We will be evaluating the subgroups of patients within SUPPRESS, such as T-cell-depleted transplant recipients who have a lower risk of GVHD, to better understand these results and inform our next steps,” said W. Garrett Nichols, MD, Chimerix’s chief medical officer.
“We are reaching out to investigators and other experts to help us assess the complete data set to understand what may have caused the results of the SUPPRESS trial to differ substantially from those seen in the phase 2 study. Additionally, we are in communication with the US Food and Drug Administration and other regulatory bodies and will share any updates on the brincidofovir clinical program when we can.”
“With data currently in hand, we believe that brincidofovir may ultimately demonstrate a positive risk-benefit profile for the treatment of adenovirus and smallpox, as well as use in other populations in need of a novel compound for DNA viral infections.”
Chimerix plans to continue the programs testing brincidofovir in serious adenovirus infections and in smallpox. Pending the availability of complete data from SUPPRESS, including secondary endpoints in other dsDNA viral infections, Chimerix has elected to pause further enrollment in the phase 3 SUSTAIN and SURPASS trials in kidney transplant recipients.
A full analysis of the SUPPRESS trial results is ongoing. The data are scheduled to be presented at the BMT Tandem Meetings in Honolulu, Hawaii, in February. ![]()
The antiviral drug brincidofovir did not meet the primary endpoint of the phase 3 SUPPRESS trial, according to the drug’s developer, Chimerix.
Brincidofovir did not prevent clinically significant cytomegalovirus (CMV) infection through week 24 after hematopoietic stem cell transplant (HSCT).
However, the president and CEO of Chimerix said the company still believes there is a “viable path forward” for the drug.
Brincidofovir is an oral nucleotide analog that has shown in vitro antiviral activity against all 5 families of DNA viruses that affect humans, including herpes viruses and adenovirus.
The SUPPRESS trial, which was initiated in August 2013 and fully enrolled in June 2015, was informed by a successful phase 2 trial conducted in HSCT recipients.
The SUPPRESS trial enrolled and treated 452 adults who received allogeneic HSCTs from more than 40 transplant centers in the US, Canada, and Europe.
Subjects received twice-weekly brincidofovir or placebo (2:1 ratio) from the early post-transplant period through week 14 post-transplant, the period of highest risk for viral infections.
All patients in the trial were CMV-seropositive, placing them at high risk of CMV infection. The most common indications leading to HSCT were acute myelogenous leukemia (43% of patients), myelodysplasia (17%), non-Hodgkin lymphoma (10%), and acute lymphocytic leukemia (9%).
During the on-treatment period through week 14 after HSCT, fewer patients in the brincidofovir arm had a CMV infection, which was consistent with results from the phase 2 study of the drug.
However, during the 10 weeks off treatment from week 14 to week 24, there was an increase in CMV infections in the brincidofovir arm compared to the control arm. And there was a non-statistically significant increase in mortality in the brincidofovir arm compared to the control arm.
Preliminary analysis suggests the failure in preventing CMV infections and the increased mortality in the brincidofovir arm were driven by confirmed cases of graft-versus-host-disease (GVHD), which resulted in a significantly higher use of corticosteroids than in the control arm.
Both GVHD and the use of corticosteroids are risk factors for “late” CMV infection that occurs after the discontinuation of the antiviral in HSCT recipients.
The rate of study drug discontinuation for gastrointestinal events was less than 10% in this study, which is comparable to that observed in the phase 2 trial of brincidofovir in a similar HSCT population.
“While we are clearly disappointed in the top-line results from SUPPRESS, we remain committed to better understanding the full data set as we consider potential paths forward for brincidofovir,” said M. Michelle Berrey, MD, president and CEO of Chimerix.
“We will be evaluating the subgroups of patients within SUPPRESS, such as T-cell-depleted transplant recipients who have a lower risk of GVHD, to better understand these results and inform our next steps,” said W. Garrett Nichols, MD, Chimerix’s chief medical officer.
“We are reaching out to investigators and other experts to help us assess the complete data set to understand what may have caused the results of the SUPPRESS trial to differ substantially from those seen in the phase 2 study. Additionally, we are in communication with the US Food and Drug Administration and other regulatory bodies and will share any updates on the brincidofovir clinical program when we can.”
“With data currently in hand, we believe that brincidofovir may ultimately demonstrate a positive risk-benefit profile for the treatment of adenovirus and smallpox, as well as use in other populations in need of a novel compound for DNA viral infections.”
Chimerix plans to continue the programs testing brincidofovir in serious adenovirus infections and in smallpox. Pending the availability of complete data from SUPPRESS, including secondary endpoints in other dsDNA viral infections, Chimerix has elected to pause further enrollment in the phase 3 SUSTAIN and SURPASS trials in kidney transplant recipients.
A full analysis of the SUPPRESS trial results is ongoing. The data are scheduled to be presented at the BMT Tandem Meetings in Honolulu, Hawaii, in February. ![]()
The antiviral drug brincidofovir did not meet the primary endpoint of the phase 3 SUPPRESS trial, according to the drug’s developer, Chimerix.
Brincidofovir did not prevent clinically significant cytomegalovirus (CMV) infection through week 24 after hematopoietic stem cell transplant (HSCT).
However, the president and CEO of Chimerix said the company still believes there is a “viable path forward” for the drug.
Brincidofovir is an oral nucleotide analog that has shown in vitro antiviral activity against all 5 families of DNA viruses that affect humans, including herpes viruses and adenovirus.
The SUPPRESS trial, which was initiated in August 2013 and fully enrolled in June 2015, was informed by a successful phase 2 trial conducted in HSCT recipients.
The SUPPRESS trial enrolled and treated 452 adults who received allogeneic HSCTs from more than 40 transplant centers in the US, Canada, and Europe.
Subjects received twice-weekly brincidofovir or placebo (2:1 ratio) from the early post-transplant period through week 14 post-transplant, the period of highest risk for viral infections.
All patients in the trial were CMV-seropositive, placing them at high risk of CMV infection. The most common indications leading to HSCT were acute myelogenous leukemia (43% of patients), myelodysplasia (17%), non-Hodgkin lymphoma (10%), and acute lymphocytic leukemia (9%).
During the on-treatment period through week 14 after HSCT, fewer patients in the brincidofovir arm had a CMV infection, which was consistent with results from the phase 2 study of the drug.
However, during the 10 weeks off treatment from week 14 to week 24, there was an increase in CMV infections in the brincidofovir arm compared to the control arm. And there was a non-statistically significant increase in mortality in the brincidofovir arm compared to the control arm.
Preliminary analysis suggests the failure in preventing CMV infections and the increased mortality in the brincidofovir arm were driven by confirmed cases of graft-versus-host-disease (GVHD), which resulted in a significantly higher use of corticosteroids than in the control arm.
Both GVHD and the use of corticosteroids are risk factors for “late” CMV infection that occurs after the discontinuation of the antiviral in HSCT recipients.
The rate of study drug discontinuation for gastrointestinal events was less than 10% in this study, which is comparable to that observed in the phase 2 trial of brincidofovir in a similar HSCT population.
“While we are clearly disappointed in the top-line results from SUPPRESS, we remain committed to better understanding the full data set as we consider potential paths forward for brincidofovir,” said M. Michelle Berrey, MD, president and CEO of Chimerix.
“We will be evaluating the subgroups of patients within SUPPRESS, such as T-cell-depleted transplant recipients who have a lower risk of GVHD, to better understand these results and inform our next steps,” said W. Garrett Nichols, MD, Chimerix’s chief medical officer.
“We are reaching out to investigators and other experts to help us assess the complete data set to understand what may have caused the results of the SUPPRESS trial to differ substantially from those seen in the phase 2 study. Additionally, we are in communication with the US Food and Drug Administration and other regulatory bodies and will share any updates on the brincidofovir clinical program when we can.”
“With data currently in hand, we believe that brincidofovir may ultimately demonstrate a positive risk-benefit profile for the treatment of adenovirus and smallpox, as well as use in other populations in need of a novel compound for DNA viral infections.”
Chimerix plans to continue the programs testing brincidofovir in serious adenovirus infections and in smallpox. Pending the availability of complete data from SUPPRESS, including secondary endpoints in other dsDNA viral infections, Chimerix has elected to pause further enrollment in the phase 3 SUSTAIN and SURPASS trials in kidney transplant recipients.
A full analysis of the SUPPRESS trial results is ongoing. The data are scheduled to be presented at the BMT Tandem Meetings in Honolulu, Hawaii, in February. ![]()
Study reveals germline variants in AML, other cancers

A study published in Nature Communications has shed light on the hereditary elements of 12 cancer types.
Investigators looked for rare germline mutations in genes known to be associated with cancer and found the frequency of these mutations varied widely, from 4% in the acute myeloid leukemia (AML) cases studied to 19% in cases of ovarian cancer.
The team’s analysis also revealed an unexpected inherited component to stomach cancer and provided some clarity on the consequences of certain mutations in the BRCA1 and BRCA2 genes.
Li Ding, PhD, of Washington University School of Medicine in St Louis, Missouri, and her colleagues conducted this study, analyzing genetic information from more than 4000 cancer cases included in The Cancer Genome Atlas project.
“In general, we have known that ovarian and breast cancers have a significant inherited component, and others, such as acute myeloid leukemia and lung cancer, have a much smaller inherited genetic contribution,” Dr Ding said. “But this is the first time, on a large scale, that we’ve been able to pinpoint gene culprits or even the actual mutations responsible for cancer susceptibility.”
To help tease out cancer’s inherited components, Dr Ding and her colleagues looked for germline truncations in 114 genes known to be associated with cancer.
“We looked for germline mutations in the tumor, but it was not enough for the mutations simply to be present,” Dr Ding said. “They needed to be enriched in the tumor—present at higher frequency. If a mutation is present in the germline and amplified in the tumor, there is a high likelihood it is playing a role in the cancer.”
The investigators found germline truncations in all 12 cancer types analyzed, but the mutations occurred in varying frequencies depending on the cancer.
The percentage of tumors with truncations in the germline was 4% for AML and glioblastoma; 5% for kidney cancer; 7% for lung adenocarcinoma and endometrial cancer; 8% for head and neck cancer, glioma, lung squamous cell carcinoma, and prostate cancer; 9% for breast cancer; 11% for stomach cancer; and 19% for ovarian cancer.
“We also found a significant number of germline truncations in the BRCA1 and BRCA2 genes present in tumor types other than breast cancer, including stomach and prostate cancers, for example,” Dr Ding said. “This suggests we should pay attention to the potential involvement of these 2 genes in other cancer types.”
The investigators said they identified 13 cancer genes with significant enrichment of rare truncations. Some of these were associated with specific cancers—for example, RAD51C in AML, PALB2 in stomach cancer, and MSH6 in endometrial cancer.
And the team observed significant, tumor-specific loss of heterozygosity in 9 genes—ATM, BAP1, BRCA1/2, BRIP1, FANCM, PALB2, and RAD51C/D.
Dr Ding said more research is needed to confirm these results before they can be used to advise patients making healthcare decisions.
“Our strategy of investigating germline-tumor interactions provides a good way to prioritize important mutations that we should focus on,” she said. “For the information to eventually be used in the clinic, we will need to perform this type of analysis on even larger numbers of patients.” ![]()

A study published in Nature Communications has shed light on the hereditary elements of 12 cancer types.
Investigators looked for rare germline mutations in genes known to be associated with cancer and found the frequency of these mutations varied widely, from 4% in the acute myeloid leukemia (AML) cases studied to 19% in cases of ovarian cancer.
The team’s analysis also revealed an unexpected inherited component to stomach cancer and provided some clarity on the consequences of certain mutations in the BRCA1 and BRCA2 genes.
Li Ding, PhD, of Washington University School of Medicine in St Louis, Missouri, and her colleagues conducted this study, analyzing genetic information from more than 4000 cancer cases included in The Cancer Genome Atlas project.
“In general, we have known that ovarian and breast cancers have a significant inherited component, and others, such as acute myeloid leukemia and lung cancer, have a much smaller inherited genetic contribution,” Dr Ding said. “But this is the first time, on a large scale, that we’ve been able to pinpoint gene culprits or even the actual mutations responsible for cancer susceptibility.”
To help tease out cancer’s inherited components, Dr Ding and her colleagues looked for germline truncations in 114 genes known to be associated with cancer.
“We looked for germline mutations in the tumor, but it was not enough for the mutations simply to be present,” Dr Ding said. “They needed to be enriched in the tumor—present at higher frequency. If a mutation is present in the germline and amplified in the tumor, there is a high likelihood it is playing a role in the cancer.”
The investigators found germline truncations in all 12 cancer types analyzed, but the mutations occurred in varying frequencies depending on the cancer.
The percentage of tumors with truncations in the germline was 4% for AML and glioblastoma; 5% for kidney cancer; 7% for lung adenocarcinoma and endometrial cancer; 8% for head and neck cancer, glioma, lung squamous cell carcinoma, and prostate cancer; 9% for breast cancer; 11% for stomach cancer; and 19% for ovarian cancer.
“We also found a significant number of germline truncations in the BRCA1 and BRCA2 genes present in tumor types other than breast cancer, including stomach and prostate cancers, for example,” Dr Ding said. “This suggests we should pay attention to the potential involvement of these 2 genes in other cancer types.”
The investigators said they identified 13 cancer genes with significant enrichment of rare truncations. Some of these were associated with specific cancers—for example, RAD51C in AML, PALB2 in stomach cancer, and MSH6 in endometrial cancer.
And the team observed significant, tumor-specific loss of heterozygosity in 9 genes—ATM, BAP1, BRCA1/2, BRIP1, FANCM, PALB2, and RAD51C/D.
Dr Ding said more research is needed to confirm these results before they can be used to advise patients making healthcare decisions.
“Our strategy of investigating germline-tumor interactions provides a good way to prioritize important mutations that we should focus on,” she said. “For the information to eventually be used in the clinic, we will need to perform this type of analysis on even larger numbers of patients.” ![]()

A study published in Nature Communications has shed light on the hereditary elements of 12 cancer types.
Investigators looked for rare germline mutations in genes known to be associated with cancer and found the frequency of these mutations varied widely, from 4% in the acute myeloid leukemia (AML) cases studied to 19% in cases of ovarian cancer.
The team’s analysis also revealed an unexpected inherited component to stomach cancer and provided some clarity on the consequences of certain mutations in the BRCA1 and BRCA2 genes.
Li Ding, PhD, of Washington University School of Medicine in St Louis, Missouri, and her colleagues conducted this study, analyzing genetic information from more than 4000 cancer cases included in The Cancer Genome Atlas project.
“In general, we have known that ovarian and breast cancers have a significant inherited component, and others, such as acute myeloid leukemia and lung cancer, have a much smaller inherited genetic contribution,” Dr Ding said. “But this is the first time, on a large scale, that we’ve been able to pinpoint gene culprits or even the actual mutations responsible for cancer susceptibility.”
To help tease out cancer’s inherited components, Dr Ding and her colleagues looked for germline truncations in 114 genes known to be associated with cancer.
“We looked for germline mutations in the tumor, but it was not enough for the mutations simply to be present,” Dr Ding said. “They needed to be enriched in the tumor—present at higher frequency. If a mutation is present in the germline and amplified in the tumor, there is a high likelihood it is playing a role in the cancer.”
The investigators found germline truncations in all 12 cancer types analyzed, but the mutations occurred in varying frequencies depending on the cancer.
The percentage of tumors with truncations in the germline was 4% for AML and glioblastoma; 5% for kidney cancer; 7% for lung adenocarcinoma and endometrial cancer; 8% for head and neck cancer, glioma, lung squamous cell carcinoma, and prostate cancer; 9% for breast cancer; 11% for stomach cancer; and 19% for ovarian cancer.
“We also found a significant number of germline truncations in the BRCA1 and BRCA2 genes present in tumor types other than breast cancer, including stomach and prostate cancers, for example,” Dr Ding said. “This suggests we should pay attention to the potential involvement of these 2 genes in other cancer types.”
The investigators said they identified 13 cancer genes with significant enrichment of rare truncations. Some of these were associated with specific cancers—for example, RAD51C in AML, PALB2 in stomach cancer, and MSH6 in endometrial cancer.
And the team observed significant, tumor-specific loss of heterozygosity in 9 genes—ATM, BAP1, BRCA1/2, BRIP1, FANCM, PALB2, and RAD51C/D.
Dr Ding said more research is needed to confirm these results before they can be used to advise patients making healthcare decisions.
“Our strategy of investigating germline-tumor interactions provides a good way to prioritize important mutations that we should focus on,” she said. “For the information to eventually be used in the clinic, we will need to perform this type of analysis on even larger numbers of patients.” ![]()
Education may increase clinical trial participation

Photo courtesy of NCI
and Matthews Media Group
A new study suggests that educating cancer patients about clinical trials—with either a general or patient-specific program—increases the likelihood that patients will enroll in such trials.
After completing either type of educational program, 21% of the cancer patients studied chose to enroll in clinical trials.
Traditionally, less than 5% of cancer patients decide to participate in trials, according to the American Cancer Society.
“Unfortunately, although clinical trials are critical for advancing cancer treatment and ultimately serve as the basis for new standards of care, very few patients participate,” said Neal J. Meropol, MD, of Case Western Reserve University School of Medicine in Cleveland, Ohio.
“We want to close the patient knowledge gap and positively affect their attitudes toward clinical trials.”
Dr Meropol and his colleagues described their effort to do just that in the Journal of Clinical Oncology.
The researchers compared a tailored video education program on clinical trials, PRE-ACT (Preparatory Education about Clinical Trials), to educational information delivered as written text.
PRE-ACT is an intervention in which patients access a website to take an online survey. The survey gauges the individual patient’s knowledge and attitudes about clinical trials, and then, based on that patient’s answers, video clips are presented addressing his or her specific concerns.
For example, patients may worry they will receive a placebo rather than active treatment, so one video clip explains how placebos are used ethically in cancer studies, and the fact that very few studies will include a placebo without any active treatment. The videos also help patients clarify their preferences in terms of quality of life or length of life.
Results
Dr Meropol and his colleagues enrolled 1255 cancer patients in the study. Half of them participated in the PRE-ACT program, and the other half received written information about clinical trials that was not specifically chosen based on their survey responses.
Both interventions improved patients’ knowledge and attitudes regarding clinical trials when compared with baseline (all P<0.001).
Patients in both arms said they felt more prepared to consider enrolling in a clinical trial after completing their assigned educational program (P<0.001), but there was a trend favoring the PRE-ACT arm (P<0.09).
Patients in the PRE-ACT arm also showed a significantly greater increase in knowledge (P<0.001) and a significantly greater decrease in attitudinal barriers (P<0.001) than patients in the text-only arm. And PRE-ACT was associated with greater patient satisfaction.
Financial concerns
During the course of this research, Dr Meropol and his colleagues made a surprising discovery. Video clips meant to address concerns about the costs of clinical trial treatment actually caused a spike in worries about the out-of-pocket costs of clinical trials.
These financial concerns generated yet another paper that appeared in the same edition of the Journal of Clinical Oncology.
“What was a surprise is that giving people information about costs in general terms made them more anxious,” Dr Meropol said. “It was not surprising to us that these concerns actually affect distress, add to decisional conflict, and interfere with decision-making.”
“This finding highlighted for us that communication about costs is both necessary and challenging. It indicates that we need to be sensitive to patients’ cost concerns as they navigate decisions about cancer care.”
The researchers are now planning to develop tools to assist patients with financial navigation. The team is also developing a web-based educational program for oncology nurses to help them discuss clinical trial participation with patients. ![]()

Photo courtesy of NCI
and Matthews Media Group
A new study suggests that educating cancer patients about clinical trials—with either a general or patient-specific program—increases the likelihood that patients will enroll in such trials.
After completing either type of educational program, 21% of the cancer patients studied chose to enroll in clinical trials.
Traditionally, less than 5% of cancer patients decide to participate in trials, according to the American Cancer Society.
“Unfortunately, although clinical trials are critical for advancing cancer treatment and ultimately serve as the basis for new standards of care, very few patients participate,” said Neal J. Meropol, MD, of Case Western Reserve University School of Medicine in Cleveland, Ohio.
“We want to close the patient knowledge gap and positively affect their attitudes toward clinical trials.”
Dr Meropol and his colleagues described their effort to do just that in the Journal of Clinical Oncology.
The researchers compared a tailored video education program on clinical trials, PRE-ACT (Preparatory Education about Clinical Trials), to educational information delivered as written text.
PRE-ACT is an intervention in which patients access a website to take an online survey. The survey gauges the individual patient’s knowledge and attitudes about clinical trials, and then, based on that patient’s answers, video clips are presented addressing his or her specific concerns.
For example, patients may worry they will receive a placebo rather than active treatment, so one video clip explains how placebos are used ethically in cancer studies, and the fact that very few studies will include a placebo without any active treatment. The videos also help patients clarify their preferences in terms of quality of life or length of life.
Results
Dr Meropol and his colleagues enrolled 1255 cancer patients in the study. Half of them participated in the PRE-ACT program, and the other half received written information about clinical trials that was not specifically chosen based on their survey responses.
Both interventions improved patients’ knowledge and attitudes regarding clinical trials when compared with baseline (all P<0.001).
Patients in both arms said they felt more prepared to consider enrolling in a clinical trial after completing their assigned educational program (P<0.001), but there was a trend favoring the PRE-ACT arm (P<0.09).
Patients in the PRE-ACT arm also showed a significantly greater increase in knowledge (P<0.001) and a significantly greater decrease in attitudinal barriers (P<0.001) than patients in the text-only arm. And PRE-ACT was associated with greater patient satisfaction.
Financial concerns
During the course of this research, Dr Meropol and his colleagues made a surprising discovery. Video clips meant to address concerns about the costs of clinical trial treatment actually caused a spike in worries about the out-of-pocket costs of clinical trials.
These financial concerns generated yet another paper that appeared in the same edition of the Journal of Clinical Oncology.
“What was a surprise is that giving people information about costs in general terms made them more anxious,” Dr Meropol said. “It was not surprising to us that these concerns actually affect distress, add to decisional conflict, and interfere with decision-making.”
“This finding highlighted for us that communication about costs is both necessary and challenging. It indicates that we need to be sensitive to patients’ cost concerns as they navigate decisions about cancer care.”
The researchers are now planning to develop tools to assist patients with financial navigation. The team is also developing a web-based educational program for oncology nurses to help them discuss clinical trial participation with patients. ![]()

Photo courtesy of NCI
and Matthews Media Group
A new study suggests that educating cancer patients about clinical trials—with either a general or patient-specific program—increases the likelihood that patients will enroll in such trials.
After completing either type of educational program, 21% of the cancer patients studied chose to enroll in clinical trials.
Traditionally, less than 5% of cancer patients decide to participate in trials, according to the American Cancer Society.
“Unfortunately, although clinical trials are critical for advancing cancer treatment and ultimately serve as the basis for new standards of care, very few patients participate,” said Neal J. Meropol, MD, of Case Western Reserve University School of Medicine in Cleveland, Ohio.
“We want to close the patient knowledge gap and positively affect their attitudes toward clinical trials.”
Dr Meropol and his colleagues described their effort to do just that in the Journal of Clinical Oncology.
The researchers compared a tailored video education program on clinical trials, PRE-ACT (Preparatory Education about Clinical Trials), to educational information delivered as written text.
PRE-ACT is an intervention in which patients access a website to take an online survey. The survey gauges the individual patient’s knowledge and attitudes about clinical trials, and then, based on that patient’s answers, video clips are presented addressing his or her specific concerns.
For example, patients may worry they will receive a placebo rather than active treatment, so one video clip explains how placebos are used ethically in cancer studies, and the fact that very few studies will include a placebo without any active treatment. The videos also help patients clarify their preferences in terms of quality of life or length of life.
Results
Dr Meropol and his colleagues enrolled 1255 cancer patients in the study. Half of them participated in the PRE-ACT program, and the other half received written information about clinical trials that was not specifically chosen based on their survey responses.
Both interventions improved patients’ knowledge and attitudes regarding clinical trials when compared with baseline (all P<0.001).
Patients in both arms said they felt more prepared to consider enrolling in a clinical trial after completing their assigned educational program (P<0.001), but there was a trend favoring the PRE-ACT arm (P<0.09).
Patients in the PRE-ACT arm also showed a significantly greater increase in knowledge (P<0.001) and a significantly greater decrease in attitudinal barriers (P<0.001) than patients in the text-only arm. And PRE-ACT was associated with greater patient satisfaction.
Financial concerns
During the course of this research, Dr Meropol and his colleagues made a surprising discovery. Video clips meant to address concerns about the costs of clinical trial treatment actually caused a spike in worries about the out-of-pocket costs of clinical trials.
These financial concerns generated yet another paper that appeared in the same edition of the Journal of Clinical Oncology.
“What was a surprise is that giving people information about costs in general terms made them more anxious,” Dr Meropol said. “It was not surprising to us that these concerns actually affect distress, add to decisional conflict, and interfere with decision-making.”
“This finding highlighted for us that communication about costs is both necessary and challenging. It indicates that we need to be sensitive to patients’ cost concerns as they navigate decisions about cancer care.”
The researchers are now planning to develop tools to assist patients with financial navigation. The team is also developing a web-based educational program for oncology nurses to help them discuss clinical trial participation with patients. ![]()
Team explains how artemisinin kills malaria parasite

infecting a red blood cell
Photo courtesy of St. Jude
Children’s Research Hospital
Researchers say they have gained a better understanding of how the antimalarial drug artemisinin kills the Plasmodium falciparum parasite.
A chemical proteomics analysis revealed more than 120 protein targets of artemisinin and the mechanism that activates its killing effect.
Given the emergence of artemisinin resistance, the team believes their findings could aid the design of new treatments against drug-resistant parasites.
They reported the findings in Nature Communications.
Previously, only 2 targets of artemisinin had been identified, and their correlation with the parasite-killing effect of the drug had been questioned.
Lin Qingsong, PhD, of the National University of Singapore, and his colleagues identified 124 protein targets of artemisinin in P falciparum. Many of these newly identified protein targets are involved in essential biological processes in the parasite, thus explaining artemisinin’s potent killing effect.
The research suggests that, through its promiscuous targeting mechanism, artemisinin targets the blood-eating nature of the malaria parasite, binding to a broad spectrum of targets simultaneously and fatally disrupting the biochemistry of the parasite.
The study also showed that the main activator of artemisinin is heme, an iron-containing compound that is either biosynthesized by the parasite at its early developmental ring stage or derived from hemoglobin digestion in the later stages.
The drug activation level was found to be much lower in ring-stage parasites, given that artemisinin activation requires heme, which is of much lower abundance and is biosynthesized by the parasite.
In comparison, during the late stages of its life cycle, the parasite actively digests the hemoglobin in infected blood cells as its primary energy source. This releases large amounts of heme, and the drug is able to specifically respond to parasite-infected cells and effectively attack the disease-causing parasites.
“The current findings not only provide a more complete picture of how artemisinin and its derivatives work but also suggest new ways of using the drug,” Dr Lin said. “For instance, to improve drug activation at ring stage, we can explore enhancing the level of heme biosynthesis in the parasite.”
“By understanding that hemoglobin digestion releases huge amounts of heme, which brings about the effective killing mechanism in the later stages, we can also consider prolonging the treatment time to ensure that the drug can effectively kill the parasite through multiple cycles.”
In addition, the researchers are planning to develop novel artemisinin analogues with more specific targeting properties. ![]()

infecting a red blood cell
Photo courtesy of St. Jude
Children’s Research Hospital
Researchers say they have gained a better understanding of how the antimalarial drug artemisinin kills the Plasmodium falciparum parasite.
A chemical proteomics analysis revealed more than 120 protein targets of artemisinin and the mechanism that activates its killing effect.
Given the emergence of artemisinin resistance, the team believes their findings could aid the design of new treatments against drug-resistant parasites.
They reported the findings in Nature Communications.
Previously, only 2 targets of artemisinin had been identified, and their correlation with the parasite-killing effect of the drug had been questioned.
Lin Qingsong, PhD, of the National University of Singapore, and his colleagues identified 124 protein targets of artemisinin in P falciparum. Many of these newly identified protein targets are involved in essential biological processes in the parasite, thus explaining artemisinin’s potent killing effect.
The research suggests that, through its promiscuous targeting mechanism, artemisinin targets the blood-eating nature of the malaria parasite, binding to a broad spectrum of targets simultaneously and fatally disrupting the biochemistry of the parasite.
The study also showed that the main activator of artemisinin is heme, an iron-containing compound that is either biosynthesized by the parasite at its early developmental ring stage or derived from hemoglobin digestion in the later stages.
The drug activation level was found to be much lower in ring-stage parasites, given that artemisinin activation requires heme, which is of much lower abundance and is biosynthesized by the parasite.
In comparison, during the late stages of its life cycle, the parasite actively digests the hemoglobin in infected blood cells as its primary energy source. This releases large amounts of heme, and the drug is able to specifically respond to parasite-infected cells and effectively attack the disease-causing parasites.
“The current findings not only provide a more complete picture of how artemisinin and its derivatives work but also suggest new ways of using the drug,” Dr Lin said. “For instance, to improve drug activation at ring stage, we can explore enhancing the level of heme biosynthesis in the parasite.”
“By understanding that hemoglobin digestion releases huge amounts of heme, which brings about the effective killing mechanism in the later stages, we can also consider prolonging the treatment time to ensure that the drug can effectively kill the parasite through multiple cycles.”
In addition, the researchers are planning to develop novel artemisinin analogues with more specific targeting properties. ![]()

infecting a red blood cell
Photo courtesy of St. Jude
Children’s Research Hospital
Researchers say they have gained a better understanding of how the antimalarial drug artemisinin kills the Plasmodium falciparum parasite.
A chemical proteomics analysis revealed more than 120 protein targets of artemisinin and the mechanism that activates its killing effect.
Given the emergence of artemisinin resistance, the team believes their findings could aid the design of new treatments against drug-resistant parasites.
They reported the findings in Nature Communications.
Previously, only 2 targets of artemisinin had been identified, and their correlation with the parasite-killing effect of the drug had been questioned.
Lin Qingsong, PhD, of the National University of Singapore, and his colleagues identified 124 protein targets of artemisinin in P falciparum. Many of these newly identified protein targets are involved in essential biological processes in the parasite, thus explaining artemisinin’s potent killing effect.
The research suggests that, through its promiscuous targeting mechanism, artemisinin targets the blood-eating nature of the malaria parasite, binding to a broad spectrum of targets simultaneously and fatally disrupting the biochemistry of the parasite.
The study also showed that the main activator of artemisinin is heme, an iron-containing compound that is either biosynthesized by the parasite at its early developmental ring stage or derived from hemoglobin digestion in the later stages.
The drug activation level was found to be much lower in ring-stage parasites, given that artemisinin activation requires heme, which is of much lower abundance and is biosynthesized by the parasite.
In comparison, during the late stages of its life cycle, the parasite actively digests the hemoglobin in infected blood cells as its primary energy source. This releases large amounts of heme, and the drug is able to specifically respond to parasite-infected cells and effectively attack the disease-causing parasites.
“The current findings not only provide a more complete picture of how artemisinin and its derivatives work but also suggest new ways of using the drug,” Dr Lin said. “For instance, to improve drug activation at ring stage, we can explore enhancing the level of heme biosynthesis in the parasite.”
“By understanding that hemoglobin digestion releases huge amounts of heme, which brings about the effective killing mechanism in the later stages, we can also consider prolonging the treatment time to ensure that the drug can effectively kill the parasite through multiple cycles.”
In addition, the researchers are planning to develop novel artemisinin analogues with more specific targeting properties. ![]()
De novo mutation linked to FA subtype
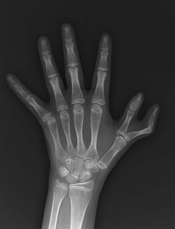
with Fanconi anemia
Researchers say they have established the cause of a subtype of Fanconi anemia (FA)—a de novo mutation in 1 allele of RAD51, a gene responsible for repairing DNA damage.
The team made this discovery in a child with an FA-like syndrome who has healthy parents and a healthy sister.
“The particular mutation in this patient was a surprise to us,” said Patrick May, PhD, of the University of Luxembourg.
“It occurred only in 1 of the 2 RAD51 gene copies, which every person carries in the genome, but every RAD51 gene copy was normal in the child’s parents.”
Dr May and his colleagues described the mutation in Nature Communications.
Specifically, the researchers found a de novo g.41022153G>A; p.Ala293Thr (NM_002875) missense mutation in 1 allele of RAD51.
They said this heterozygous mutation causes a novel FA subtype, dubbed “FA-R,” which appears to be the first subtype of FA caused by a dominant-negative mutation.
The patient the researchers analyzed has microcephaly and mental retardation but has reached adulthood without the bone marrow failure and pediatric cancers typically observed in patients with FA.
Until this case, researchers believed that mutations leading to FA showed recessive inheritance and therefore had to be derived from both parents to lead to FA. Spontaneous mutations of the RAD51 gene like in this case had not been observed.
Dr May and his colleagues said their finding has implications for genetic counseling of families with a high risk for FA. Previously, people who wanted to have children but had relatives suffering from FA were screened for mutations in 1 of the 17 genes connected with the disease. Now, the risk of having a child with FA has to be recalculated.
“Furthermore, understanding this mutation teaches us more about how the RAD51 gene product protects the DNA and how disruptions of DNA repair may lead to leukemia and solid tumors,” Dr May said. “Of course, understanding the origins of human cancer will help us diagnose it with more confidence earlier and devise new therapies to prevent or mitigate it.” ![]()

with Fanconi anemia
Researchers say they have established the cause of a subtype of Fanconi anemia (FA)—a de novo mutation in 1 allele of RAD51, a gene responsible for repairing DNA damage.
The team made this discovery in a child with an FA-like syndrome who has healthy parents and a healthy sister.
“The particular mutation in this patient was a surprise to us,” said Patrick May, PhD, of the University of Luxembourg.
“It occurred only in 1 of the 2 RAD51 gene copies, which every person carries in the genome, but every RAD51 gene copy was normal in the child’s parents.”
Dr May and his colleagues described the mutation in Nature Communications.
Specifically, the researchers found a de novo g.41022153G>A; p.Ala293Thr (NM_002875) missense mutation in 1 allele of RAD51.
They said this heterozygous mutation causes a novel FA subtype, dubbed “FA-R,” which appears to be the first subtype of FA caused by a dominant-negative mutation.
The patient the researchers analyzed has microcephaly and mental retardation but has reached adulthood without the bone marrow failure and pediatric cancers typically observed in patients with FA.
Until this case, researchers believed that mutations leading to FA showed recessive inheritance and therefore had to be derived from both parents to lead to FA. Spontaneous mutations of the RAD51 gene like in this case had not been observed.
Dr May and his colleagues said their finding has implications for genetic counseling of families with a high risk for FA. Previously, people who wanted to have children but had relatives suffering from FA were screened for mutations in 1 of the 17 genes connected with the disease. Now, the risk of having a child with FA has to be recalculated.
“Furthermore, understanding this mutation teaches us more about how the RAD51 gene product protects the DNA and how disruptions of DNA repair may lead to leukemia and solid tumors,” Dr May said. “Of course, understanding the origins of human cancer will help us diagnose it with more confidence earlier and devise new therapies to prevent or mitigate it.” ![]()

with Fanconi anemia
Researchers say they have established the cause of a subtype of Fanconi anemia (FA)—a de novo mutation in 1 allele of RAD51, a gene responsible for repairing DNA damage.
The team made this discovery in a child with an FA-like syndrome who has healthy parents and a healthy sister.
“The particular mutation in this patient was a surprise to us,” said Patrick May, PhD, of the University of Luxembourg.
“It occurred only in 1 of the 2 RAD51 gene copies, which every person carries in the genome, but every RAD51 gene copy was normal in the child’s parents.”
Dr May and his colleagues described the mutation in Nature Communications.
Specifically, the researchers found a de novo g.41022153G>A; p.Ala293Thr (NM_002875) missense mutation in 1 allele of RAD51.
They said this heterozygous mutation causes a novel FA subtype, dubbed “FA-R,” which appears to be the first subtype of FA caused by a dominant-negative mutation.
The patient the researchers analyzed has microcephaly and mental retardation but has reached adulthood without the bone marrow failure and pediatric cancers typically observed in patients with FA.
Until this case, researchers believed that mutations leading to FA showed recessive inheritance and therefore had to be derived from both parents to lead to FA. Spontaneous mutations of the RAD51 gene like in this case had not been observed.
Dr May and his colleagues said their finding has implications for genetic counseling of families with a high risk for FA. Previously, people who wanted to have children but had relatives suffering from FA were screened for mutations in 1 of the 17 genes connected with the disease. Now, the risk of having a child with FA has to be recalculated.
“Furthermore, understanding this mutation teaches us more about how the RAD51 gene product protects the DNA and how disruptions of DNA repair may lead to leukemia and solid tumors,” Dr May said. “Of course, understanding the origins of human cancer will help us diagnose it with more confidence earlier and devise new therapies to prevent or mitigate it.” ![]()
Women can take anticoagulants and hormones safely, study suggests

A study published in Blood indicates that women on anticoagulants can take estrogen-containing contraception or hormone replacement therapy
without an increased risk of venous thromboembolism (VTE) or uterine bleeding.
If a woman is diagnosed with VTE, she is often advised to stop hormone therapy, even while receiving anticoagulant therapy, because she is thought to have an increased risk of VTE recurrence.
However, this practice is based on the known association between hormone therapy and increased clotting risk in the absence of anticoagulants. The safety of the concurrent use of these medications had not been previously explored.
“While it has been common practice among healthcare providers to avoid prescribing hormone therapy and anticoagulants at the same time, there has been no evidence to support this decision,” said study author Ida Martinelli, MD, of the A. Bianchi Bonomi Hemophilia and Thrombosis Center in Milan, Italy.
“We conducted this study to address the fear felt by both the physician and patient when making the decision to stop or continue hormone therapy in this setting.”
The researchers compared the incidences of recurrent VTE and abnormal uterine bleeding in 1888 women who received anticoagulants with or without concurrent hormone therapy.
The team analyzed data from the EINSTEIN DVT and PE study, which was performed to evaluate the safety and efficacy of 2 anticoagulants—the direct oral anticoagulant rivaroxaban and the current standard of care, a low-molecular-weight heparin (enoxaparin) followed by a vitamin K antagonist (VKA).
Women of child-bearing potential were advised to use adequate methods of contraception to avoid birth defects.
Of all the women in the study, 475 used hormone therapy during the analysis period. Medications used included estrogen-only pills, combined estrogen-progestogen contraceptives, and progestin-only contraceptives.
Participants were questioned about symptoms or signs of recurrent VTE and bleeding, including uterine bleeding, during each follow-up visit.
Seven recurrent VTEs occurred while patients were using hormone therapy, while 38 events occurred during a period when patients were not using hormone therapy.
Based on this analysis, the researchers concluded that women on anticoagulants and hormone therapy experienced recurrent VTE at a rate of 3.7% per year. In contrast, those not on hormone therapy had a recurrence rate of 4.7% per year.
The incidence of abnormal uterine bleeding in patients on hormonal therapy was 22.5%, compared to 21.4% for women not on hormone therapy.
According to the study authors, the similar incidence of VTE and abnormal uterine bleeding in women who did and did not receive hormone therapy suggest that combined use of these therapies is safe.
The study also showed that abnormal uterine bleeding occurred more frequently with rivaroxaban than with enoxaparin/VKA. The bleeding rate was estimated at 29.8% per year for patients on rivaroxaban and 15.5% per year in the enoxaparin/VKA group.
The researchers said this outcome suggests the need for further studies on rivaroxaban, which may be preferred for its convenience over enoxaparin/VKA.
“For the first time, we demonstrate that women suffering from blood clots can safely take hormone-containing contraceptives or hormone replacement therapy with anticoagulants, providing women the freedom to choose the method of birth control and other hormone-containing medications they prefer,” Dr Martinelli said.
“While further investigation is needed to evaluate the inconvenience of abnormal uterine bleeding with rivaroxaban and the other direct oral anticoagulants, these results dispel former misconceptions and should allow clinicians to confidently treat their patients who take blood thinners and hormones concurrently.” ![]()

A study published in Blood indicates that women on anticoagulants can take estrogen-containing contraception or hormone replacement therapy
without an increased risk of venous thromboembolism (VTE) or uterine bleeding.
If a woman is diagnosed with VTE, she is often advised to stop hormone therapy, even while receiving anticoagulant therapy, because she is thought to have an increased risk of VTE recurrence.
However, this practice is based on the known association between hormone therapy and increased clotting risk in the absence of anticoagulants. The safety of the concurrent use of these medications had not been previously explored.
“While it has been common practice among healthcare providers to avoid prescribing hormone therapy and anticoagulants at the same time, there has been no evidence to support this decision,” said study author Ida Martinelli, MD, of the A. Bianchi Bonomi Hemophilia and Thrombosis Center in Milan, Italy.
“We conducted this study to address the fear felt by both the physician and patient when making the decision to stop or continue hormone therapy in this setting.”
The researchers compared the incidences of recurrent VTE and abnormal uterine bleeding in 1888 women who received anticoagulants with or without concurrent hormone therapy.
The team analyzed data from the EINSTEIN DVT and PE study, which was performed to evaluate the safety and efficacy of 2 anticoagulants—the direct oral anticoagulant rivaroxaban and the current standard of care, a low-molecular-weight heparin (enoxaparin) followed by a vitamin K antagonist (VKA).
Women of child-bearing potential were advised to use adequate methods of contraception to avoid birth defects.
Of all the women in the study, 475 used hormone therapy during the analysis period. Medications used included estrogen-only pills, combined estrogen-progestogen contraceptives, and progestin-only contraceptives.
Participants were questioned about symptoms or signs of recurrent VTE and bleeding, including uterine bleeding, during each follow-up visit.
Seven recurrent VTEs occurred while patients were using hormone therapy, while 38 events occurred during a period when patients were not using hormone therapy.
Based on this analysis, the researchers concluded that women on anticoagulants and hormone therapy experienced recurrent VTE at a rate of 3.7% per year. In contrast, those not on hormone therapy had a recurrence rate of 4.7% per year.
The incidence of abnormal uterine bleeding in patients on hormonal therapy was 22.5%, compared to 21.4% for women not on hormone therapy.
According to the study authors, the similar incidence of VTE and abnormal uterine bleeding in women who did and did not receive hormone therapy suggest that combined use of these therapies is safe.
The study also showed that abnormal uterine bleeding occurred more frequently with rivaroxaban than with enoxaparin/VKA. The bleeding rate was estimated at 29.8% per year for patients on rivaroxaban and 15.5% per year in the enoxaparin/VKA group.
The researchers said this outcome suggests the need for further studies on rivaroxaban, which may be preferred for its convenience over enoxaparin/VKA.
“For the first time, we demonstrate that women suffering from blood clots can safely take hormone-containing contraceptives or hormone replacement therapy with anticoagulants, providing women the freedom to choose the method of birth control and other hormone-containing medications they prefer,” Dr Martinelli said.
“While further investigation is needed to evaluate the inconvenience of abnormal uterine bleeding with rivaroxaban and the other direct oral anticoagulants, these results dispel former misconceptions and should allow clinicians to confidently treat their patients who take blood thinners and hormones concurrently.” ![]()

A study published in Blood indicates that women on anticoagulants can take estrogen-containing contraception or hormone replacement therapy
without an increased risk of venous thromboembolism (VTE) or uterine bleeding.
If a woman is diagnosed with VTE, she is often advised to stop hormone therapy, even while receiving anticoagulant therapy, because she is thought to have an increased risk of VTE recurrence.
However, this practice is based on the known association between hormone therapy and increased clotting risk in the absence of anticoagulants. The safety of the concurrent use of these medications had not been previously explored.
“While it has been common practice among healthcare providers to avoid prescribing hormone therapy and anticoagulants at the same time, there has been no evidence to support this decision,” said study author Ida Martinelli, MD, of the A. Bianchi Bonomi Hemophilia and Thrombosis Center in Milan, Italy.
“We conducted this study to address the fear felt by both the physician and patient when making the decision to stop or continue hormone therapy in this setting.”
The researchers compared the incidences of recurrent VTE and abnormal uterine bleeding in 1888 women who received anticoagulants with or without concurrent hormone therapy.
The team analyzed data from the EINSTEIN DVT and PE study, which was performed to evaluate the safety and efficacy of 2 anticoagulants—the direct oral anticoagulant rivaroxaban and the current standard of care, a low-molecular-weight heparin (enoxaparin) followed by a vitamin K antagonist (VKA).
Women of child-bearing potential were advised to use adequate methods of contraception to avoid birth defects.
Of all the women in the study, 475 used hormone therapy during the analysis period. Medications used included estrogen-only pills, combined estrogen-progestogen contraceptives, and progestin-only contraceptives.
Participants were questioned about symptoms or signs of recurrent VTE and bleeding, including uterine bleeding, during each follow-up visit.
Seven recurrent VTEs occurred while patients were using hormone therapy, while 38 events occurred during a period when patients were not using hormone therapy.
Based on this analysis, the researchers concluded that women on anticoagulants and hormone therapy experienced recurrent VTE at a rate of 3.7% per year. In contrast, those not on hormone therapy had a recurrence rate of 4.7% per year.
The incidence of abnormal uterine bleeding in patients on hormonal therapy was 22.5%, compared to 21.4% for women not on hormone therapy.
According to the study authors, the similar incidence of VTE and abnormal uterine bleeding in women who did and did not receive hormone therapy suggest that combined use of these therapies is safe.
The study also showed that abnormal uterine bleeding occurred more frequently with rivaroxaban than with enoxaparin/VKA. The bleeding rate was estimated at 29.8% per year for patients on rivaroxaban and 15.5% per year in the enoxaparin/VKA group.
The researchers said this outcome suggests the need for further studies on rivaroxaban, which may be preferred for its convenience over enoxaparin/VKA.
“For the first time, we demonstrate that women suffering from blood clots can safely take hormone-containing contraceptives or hormone replacement therapy with anticoagulants, providing women the freedom to choose the method of birth control and other hormone-containing medications they prefer,” Dr Martinelli said.
“While further investigation is needed to evaluate the inconvenience of abnormal uterine bleeding with rivaroxaban and the other direct oral anticoagulants, these results dispel former misconceptions and should allow clinicians to confidently treat their patients who take blood thinners and hormones concurrently.” ![]()
Combo prolongs PFS in phase 3 MM trial

Photo by Bill Branson
Results of the phase 3 ENDEAVOR trial suggest combination carfilzomib and dexamethasone prolongs progression-free survival (PFS) in patients with relapsed or refractory multiple myeloma (MM), when compared to bortezomib plus dexamethasone.
The median PFS was 18.7 months in the carfilzomib arm and 9.4 months in the bortezomib arm.
It is not clear whether this translates to an improvement in overall survival, as those data are not yet mature.
Treatment discontinuation due to adverse events (AEs) and on-study deaths were comparable between the treatment arms, although there were several AEs that occurred more frequently in the carfilzomib arm than the bortezomib arm.
These results were published in The Lancet Oncology. The trial was funded by Onyx Pharmaceuticals, Inc., a subsidiary of Amgen.
Patient treatment and characteristics
The ENDEAVOR trial included 929 patients with relapsed/refractory MM who had received 1 to 3 prior therapeutic regimens. They were randomized to receive carfilzomib in combination with low-dose dexamethasone (n=464) or bortezomib with low-dose dexamethasone (n=465) until progression.
Patients received carfilzomib as a 30-minute infusion on days 1, 2, 8, 9, 15, and 16 of 28-day treatment cycles, along with 20 mg of dexamethasone. For cycle 1 only, carfilzomib was administered at 20 mg/m2 on days 1 and 2, followed by escalation to 56 mg/m2 from day 8. Patients who tolerated 56 mg/m2 in cycle 1 were kept at this dose for subsequent cycles.
Patients who received bortezomib (1.3 mg/m2) with low-dose dexamethasone (20 mg) were given bortezomib subcutaneously or intravenously at the discretion of the investigator. More than 75% of the patients received bortezomib subcutaneously.
Baseline characteristics were generally balanced between the treatment arms. Both arms had a median age of 65 (overall range, 30-89), and about half were male. Three-quarters of patients in both arms were white, a little over 10% were Asian, 2% were black, and about 10% did not report race/ethnicity.
A majority of patients in both arms (more than 90%) had an ECOG score of 0 or 1. Most patients (more than 60%) had standard-risk cytogenetics.
The median number of prior treatment regimens was 2 in both arms. Patients in both arms had received prior lenalidomide (38% in both arms), thalidomide (45% in the carfilzomib arm and 53% in the bortezomib arm), bortezomib (54% in both arms), and carfilzomib (<1% in both arms).
Results
The median PFS in the carfilzomib arm was roughly double that of the bortezomib arm—18.7 months and 9.4 months, respectively. The hazard ratio was 0.53 (P<0.0001).
Overall survival data are not mature and are still being monitored.
The overall response rate was 77% in the carfilzomib arm and 63% in the bortezomib arm (P<0.0001). The duration of response was 21.3 months and 10.4 months, respectively.
The proportion of patients achieving a very good partial response or better was 54.3% in the carfilzomib arm and 29% in the bortezomib arm (P<0.0001). The complete response rates were 13% and 6%, respectively (P<0.001).
Treatment discontinuation due to AEs and on-study deaths were comparable between the arms. There were 75 deaths in the carfilzomib arm and 88 deaths in the bortezomib arm.
Of the 263 patients in the carfilzomib arm who discontinued treatment, 65 did so because of AEs. Of the 351 patients in the bortezomib arm who discontinued treatment, 73 did so because of AEs.
A number of known AEs were reported at a higher rate in the carfilzomib arm than the bortezomib arm, including any-grade dyspnea (28% vs 13%), hypertension (25% vs 3%), pyrexia (27% vs 14%), cough (25% vs 15%), cardiac failure (8% vs 3%), and acute renal failure (8% vs 5%).
Rates of grade 3 or higher AEs were 73% in the carfilzomib arm and 67% in the bortezomib arm. Grade 3 or higher AEs of interest in the carfilzomib and bortezomib arms, respectively, were hypertension (9% vs 3%), dyspnea (5% vs 2%), cardiac failure (5% vs 2%), acute renal failure (4% vs 3%), ischemic heart disease (2% vs 2%), and pulmonary hypertension (0.6% vs 0.2%). ![]()

Photo by Bill Branson
Results of the phase 3 ENDEAVOR trial suggest combination carfilzomib and dexamethasone prolongs progression-free survival (PFS) in patients with relapsed or refractory multiple myeloma (MM), when compared to bortezomib plus dexamethasone.
The median PFS was 18.7 months in the carfilzomib arm and 9.4 months in the bortezomib arm.
It is not clear whether this translates to an improvement in overall survival, as those data are not yet mature.
Treatment discontinuation due to adverse events (AEs) and on-study deaths were comparable between the treatment arms, although there were several AEs that occurred more frequently in the carfilzomib arm than the bortezomib arm.
These results were published in The Lancet Oncology. The trial was funded by Onyx Pharmaceuticals, Inc., a subsidiary of Amgen.
Patient treatment and characteristics
The ENDEAVOR trial included 929 patients with relapsed/refractory MM who had received 1 to 3 prior therapeutic regimens. They were randomized to receive carfilzomib in combination with low-dose dexamethasone (n=464) or bortezomib with low-dose dexamethasone (n=465) until progression.
Patients received carfilzomib as a 30-minute infusion on days 1, 2, 8, 9, 15, and 16 of 28-day treatment cycles, along with 20 mg of dexamethasone. For cycle 1 only, carfilzomib was administered at 20 mg/m2 on days 1 and 2, followed by escalation to 56 mg/m2 from day 8. Patients who tolerated 56 mg/m2 in cycle 1 were kept at this dose for subsequent cycles.
Patients who received bortezomib (1.3 mg/m2) with low-dose dexamethasone (20 mg) were given bortezomib subcutaneously or intravenously at the discretion of the investigator. More than 75% of the patients received bortezomib subcutaneously.
Baseline characteristics were generally balanced between the treatment arms. Both arms had a median age of 65 (overall range, 30-89), and about half were male. Three-quarters of patients in both arms were white, a little over 10% were Asian, 2% were black, and about 10% did not report race/ethnicity.
A majority of patients in both arms (more than 90%) had an ECOG score of 0 or 1. Most patients (more than 60%) had standard-risk cytogenetics.
The median number of prior treatment regimens was 2 in both arms. Patients in both arms had received prior lenalidomide (38% in both arms), thalidomide (45% in the carfilzomib arm and 53% in the bortezomib arm), bortezomib (54% in both arms), and carfilzomib (<1% in both arms).
Results
The median PFS in the carfilzomib arm was roughly double that of the bortezomib arm—18.7 months and 9.4 months, respectively. The hazard ratio was 0.53 (P<0.0001).
Overall survival data are not mature and are still being monitored.
The overall response rate was 77% in the carfilzomib arm and 63% in the bortezomib arm (P<0.0001). The duration of response was 21.3 months and 10.4 months, respectively.
The proportion of patients achieving a very good partial response or better was 54.3% in the carfilzomib arm and 29% in the bortezomib arm (P<0.0001). The complete response rates were 13% and 6%, respectively (P<0.001).
Treatment discontinuation due to AEs and on-study deaths were comparable between the arms. There were 75 deaths in the carfilzomib arm and 88 deaths in the bortezomib arm.
Of the 263 patients in the carfilzomib arm who discontinued treatment, 65 did so because of AEs. Of the 351 patients in the bortezomib arm who discontinued treatment, 73 did so because of AEs.
A number of known AEs were reported at a higher rate in the carfilzomib arm than the bortezomib arm, including any-grade dyspnea (28% vs 13%), hypertension (25% vs 3%), pyrexia (27% vs 14%), cough (25% vs 15%), cardiac failure (8% vs 3%), and acute renal failure (8% vs 5%).
Rates of grade 3 or higher AEs were 73% in the carfilzomib arm and 67% in the bortezomib arm. Grade 3 or higher AEs of interest in the carfilzomib and bortezomib arms, respectively, were hypertension (9% vs 3%), dyspnea (5% vs 2%), cardiac failure (5% vs 2%), acute renal failure (4% vs 3%), ischemic heart disease (2% vs 2%), and pulmonary hypertension (0.6% vs 0.2%). ![]()

Photo by Bill Branson
Results of the phase 3 ENDEAVOR trial suggest combination carfilzomib and dexamethasone prolongs progression-free survival (PFS) in patients with relapsed or refractory multiple myeloma (MM), when compared to bortezomib plus dexamethasone.
The median PFS was 18.7 months in the carfilzomib arm and 9.4 months in the bortezomib arm.
It is not clear whether this translates to an improvement in overall survival, as those data are not yet mature.
Treatment discontinuation due to adverse events (AEs) and on-study deaths were comparable between the treatment arms, although there were several AEs that occurred more frequently in the carfilzomib arm than the bortezomib arm.
These results were published in The Lancet Oncology. The trial was funded by Onyx Pharmaceuticals, Inc., a subsidiary of Amgen.
Patient treatment and characteristics
The ENDEAVOR trial included 929 patients with relapsed/refractory MM who had received 1 to 3 prior therapeutic regimens. They were randomized to receive carfilzomib in combination with low-dose dexamethasone (n=464) or bortezomib with low-dose dexamethasone (n=465) until progression.
Patients received carfilzomib as a 30-minute infusion on days 1, 2, 8, 9, 15, and 16 of 28-day treatment cycles, along with 20 mg of dexamethasone. For cycle 1 only, carfilzomib was administered at 20 mg/m2 on days 1 and 2, followed by escalation to 56 mg/m2 from day 8. Patients who tolerated 56 mg/m2 in cycle 1 were kept at this dose for subsequent cycles.
Patients who received bortezomib (1.3 mg/m2) with low-dose dexamethasone (20 mg) were given bortezomib subcutaneously or intravenously at the discretion of the investigator. More than 75% of the patients received bortezomib subcutaneously.
Baseline characteristics were generally balanced between the treatment arms. Both arms had a median age of 65 (overall range, 30-89), and about half were male. Three-quarters of patients in both arms were white, a little over 10% were Asian, 2% were black, and about 10% did not report race/ethnicity.
A majority of patients in both arms (more than 90%) had an ECOG score of 0 or 1. Most patients (more than 60%) had standard-risk cytogenetics.
The median number of prior treatment regimens was 2 in both arms. Patients in both arms had received prior lenalidomide (38% in both arms), thalidomide (45% in the carfilzomib arm and 53% in the bortezomib arm), bortezomib (54% in both arms), and carfilzomib (<1% in both arms).
Results
The median PFS in the carfilzomib arm was roughly double that of the bortezomib arm—18.7 months and 9.4 months, respectively. The hazard ratio was 0.53 (P<0.0001).
Overall survival data are not mature and are still being monitored.
The overall response rate was 77% in the carfilzomib arm and 63% in the bortezomib arm (P<0.0001). The duration of response was 21.3 months and 10.4 months, respectively.
The proportion of patients achieving a very good partial response or better was 54.3% in the carfilzomib arm and 29% in the bortezomib arm (P<0.0001). The complete response rates were 13% and 6%, respectively (P<0.001).
Treatment discontinuation due to AEs and on-study deaths were comparable between the arms. There were 75 deaths in the carfilzomib arm and 88 deaths in the bortezomib arm.
Of the 263 patients in the carfilzomib arm who discontinued treatment, 65 did so because of AEs. Of the 351 patients in the bortezomib arm who discontinued treatment, 73 did so because of AEs.
A number of known AEs were reported at a higher rate in the carfilzomib arm than the bortezomib arm, including any-grade dyspnea (28% vs 13%), hypertension (25% vs 3%), pyrexia (27% vs 14%), cough (25% vs 15%), cardiac failure (8% vs 3%), and acute renal failure (8% vs 5%).
Rates of grade 3 or higher AEs were 73% in the carfilzomib arm and 67% in the bortezomib arm. Grade 3 or higher AEs of interest in the carfilzomib and bortezomib arms, respectively, were hypertension (9% vs 3%), dyspnea (5% vs 2%), cardiac failure (5% vs 2%), acute renal failure (4% vs 3%), ischemic heart disease (2% vs 2%), and pulmonary hypertension (0.6% vs 0.2%).
Protein discovery could have therapeutic implications

Photo courtesy of The
Scripps Research Institute
New research shows how NPM1—a protein implicated in non-Hodgkin lymphoma, acute myelogenous leukemia, and other cancers—twists and morphs into different structures.
This protein has many functions and, when mutated, has been shown to interfere with cells’ normal tumor suppressing ability.
Previous research showed that a section of NPM1, called the N-terminal domain, doesn’t have a defined, folded structure.
Instead, the protein morphs between 2 forms: a 1-subunit disordered monomer and a 5-subunit folded pentamer.
Until now, the mechanism behind this transformation was unknown, but researchers believed this monomer-pentamer equilibrium could be important for the protein’s location and functioning in the cell.
Ashok Deniz, PhD, of The Scripps Research Institute in La Jolla, California, and his colleagues conducted the current study to shed light on how this transformation occurs. They reported their findings in Angewandte Chemie.
The researchers used a combination of 3 techniques to analyze NPM1—single-molecule biophysics, fluorescence resonance energy transfer, and circular dichroism.
These techniques revealed that NPM1’s transformation can proceed through more than one pathway. In one pathway, the transformation begins when the cell sends signals to attach phosphoryl groups to NPM1.
This phosphorylation prompts the ordered pentamer to become disordered and likely causes NPM1 to shuttle outside the cell’s nucleus. A meeting with a binding partner can mediate the reverse transformation to a pentamer.
However, when NPM1 does become a pentamer again under these conditions, which likely causes it to move back to the nucleolus, it takes a different path instead of just retracing its earlier steps.
The study also revealed many intermediate states between monomer and pentamer structures. And it showed that these states can be manipulated or “tuned” by changing conditions such as salt levels, phosphorylation, and partner binding, which may explain how cells regulate NPM1’s multiple functions.
The researchers said future studies could shed more light on the biological functions of these different structures and how they might be used in future cancer therapies.
“We’re studying basic biophysics, but we believe the complexity and rules we uncover for the physics of protein disorder and folding could one day also be used for better designs of therapeutics,” Dr Deniz said.
He and his colleagues also believe that combining the 3 techniques used in this study, plus a novel protein-labeling technique for single-molecule fluorescence, could be a useful strategy for studying other unstructured, intrinsically disordered proteins.

Photo courtesy of The
Scripps Research Institute
New research shows how NPM1—a protein implicated in non-Hodgkin lymphoma, acute myelogenous leukemia, and other cancers—twists and morphs into different structures.
This protein has many functions and, when mutated, has been shown to interfere with cells’ normal tumor suppressing ability.
Previous research showed that a section of NPM1, called the N-terminal domain, doesn’t have a defined, folded structure.
Instead, the protein morphs between 2 forms: a 1-subunit disordered monomer and a 5-subunit folded pentamer.
Until now, the mechanism behind this transformation was unknown, but researchers believed this monomer-pentamer equilibrium could be important for the protein’s location and functioning in the cell.
Ashok Deniz, PhD, of The Scripps Research Institute in La Jolla, California, and his colleagues conducted the current study to shed light on how this transformation occurs. They reported their findings in Angewandte Chemie.
The researchers used a combination of 3 techniques to analyze NPM1—single-molecule biophysics, fluorescence resonance energy transfer, and circular dichroism.
These techniques revealed that NPM1’s transformation can proceed through more than one pathway. In one pathway, the transformation begins when the cell sends signals to attach phosphoryl groups to NPM1.
This phosphorylation prompts the ordered pentamer to become disordered and likely causes NPM1 to shuttle outside the cell’s nucleus. A meeting with a binding partner can mediate the reverse transformation to a pentamer.
However, when NPM1 does become a pentamer again under these conditions, which likely causes it to move back to the nucleolus, it takes a different path instead of just retracing its earlier steps.
The study also revealed many intermediate states between monomer and pentamer structures. And it showed that these states can be manipulated or “tuned” by changing conditions such as salt levels, phosphorylation, and partner binding, which may explain how cells regulate NPM1’s multiple functions.
The researchers said future studies could shed more light on the biological functions of these different structures and how they might be used in future cancer therapies.
“We’re studying basic biophysics, but we believe the complexity and rules we uncover for the physics of protein disorder and folding could one day also be used for better designs of therapeutics,” Dr Deniz said.
He and his colleagues also believe that combining the 3 techniques used in this study, plus a novel protein-labeling technique for single-molecule fluorescence, could be a useful strategy for studying other unstructured, intrinsically disordered proteins.

Photo courtesy of The
Scripps Research Institute
New research shows how NPM1—a protein implicated in non-Hodgkin lymphoma, acute myelogenous leukemia, and other cancers—twists and morphs into different structures.
This protein has many functions and, when mutated, has been shown to interfere with cells’ normal tumor suppressing ability.
Previous research showed that a section of NPM1, called the N-terminal domain, doesn’t have a defined, folded structure.
Instead, the protein morphs between 2 forms: a 1-subunit disordered monomer and a 5-subunit folded pentamer.
Until now, the mechanism behind this transformation was unknown, but researchers believed this monomer-pentamer equilibrium could be important for the protein’s location and functioning in the cell.
Ashok Deniz, PhD, of The Scripps Research Institute in La Jolla, California, and his colleagues conducted the current study to shed light on how this transformation occurs. They reported their findings in Angewandte Chemie.
The researchers used a combination of 3 techniques to analyze NPM1—single-molecule biophysics, fluorescence resonance energy transfer, and circular dichroism.
These techniques revealed that NPM1’s transformation can proceed through more than one pathway. In one pathway, the transformation begins when the cell sends signals to attach phosphoryl groups to NPM1.
This phosphorylation prompts the ordered pentamer to become disordered and likely causes NPM1 to shuttle outside the cell’s nucleus. A meeting with a binding partner can mediate the reverse transformation to a pentamer.
However, when NPM1 does become a pentamer again under these conditions, which likely causes it to move back to the nucleolus, it takes a different path instead of just retracing its earlier steps.
The study also revealed many intermediate states between monomer and pentamer structures. And it showed that these states can be manipulated or “tuned” by changing conditions such as salt levels, phosphorylation, and partner binding, which may explain how cells regulate NPM1’s multiple functions.
The researchers said future studies could shed more light on the biological functions of these different structures and how they might be used in future cancer therapies.
“We’re studying basic biophysics, but we believe the complexity and rules we uncover for the physics of protein disorder and folding could one day also be used for better designs of therapeutics,” Dr Deniz said.
He and his colleagues also believe that combining the 3 techniques used in this study, plus a novel protein-labeling technique for single-molecule fluorescence, could be a useful strategy for studying other unstructured, intrinsically disordered proteins.
Antihemophilic factor meets phase 3 endpoint

An antihemophilic factor that was recently approved in the US has met the primary endpoint of a phase 3 study, according to the product’s maker, Baxalta.
The product is Adynovate, a recombinant pegylated factor VIII (FVIII) treatment based on the full-length Advate molecule.
The study enrolled patients younger than 12 years of age who had severe hemophilia A. And, thus far, none of these patients has developed inhibitors to Adynovate, which was the study’s primary endpoint.
Adynovate also proved effective for preventing bleeds in some patients and did not appear to produce any serious adverse events.
Adynovate was approved in the US last month for use as routine prophylaxis and on-demand treatment and control of bleeding episodes in hemophilia A patients age 12 and older.
Baxalta said the phase 3 data will form the basis of a US filing for a pediatric indication in early 2016.
The study was designed to assess the safety and immunogenicity of Adynovate in young, previously treated patients with severe hemophilia A. The study enrolled 73 such patients.
Researchers assessed the hemostatic efficacy of Adynovate as prophylaxis and for treatment of bleeding episodes. All patients received prophylactic Adynovate treatment (a median of 1.9 infusions per week) and were followed for 6 months.
The study’s primary endpoint was met, as no patients developed inhibitory antibodies to the drug. In addition, no treatment-related serious adverse events were reported.
About 73% of patients had no joint bleeds while on treatment, and about 38% had no bleeds of any kind.
The median annualized bleeding rate among patients treated with Adynovate was 2.0 (range, 0-49.8), and the mean was 3.0, which is comparable to the rates seen in adults treated with the product.
“These initial efficacy and safety findings indicate a potentially valuable role for Adynovate to treat pediatric patients with hemophilia A, with data consistent with what was reported in clinical studies among adult patients,” said John Orloff, MD, head of research & development and chief scientific officer at Baxalta.
With these results, the company plans to file for marketing authorization in Europe and aims to file for a pediatric indication in the US in early 2016.
Adynovate is currently under regulatory review in Japan, Canada, and Switzerland. Baxalta plans to present the complete data from this study at a congress in 2016.

An antihemophilic factor that was recently approved in the US has met the primary endpoint of a phase 3 study, according to the product’s maker, Baxalta.
The product is Adynovate, a recombinant pegylated factor VIII (FVIII) treatment based on the full-length Advate molecule.
The study enrolled patients younger than 12 years of age who had severe hemophilia A. And, thus far, none of these patients has developed inhibitors to Adynovate, which was the study’s primary endpoint.
Adynovate also proved effective for preventing bleeds in some patients and did not appear to produce any serious adverse events.
Adynovate was approved in the US last month for use as routine prophylaxis and on-demand treatment and control of bleeding episodes in hemophilia A patients age 12 and older.
Baxalta said the phase 3 data will form the basis of a US filing for a pediatric indication in early 2016.
The study was designed to assess the safety and immunogenicity of Adynovate in young, previously treated patients with severe hemophilia A. The study enrolled 73 such patients.
Researchers assessed the hemostatic efficacy of Adynovate as prophylaxis and for treatment of bleeding episodes. All patients received prophylactic Adynovate treatment (a median of 1.9 infusions per week) and were followed for 6 months.
The study’s primary endpoint was met, as no patients developed inhibitory antibodies to the drug. In addition, no treatment-related serious adverse events were reported.
About 73% of patients had no joint bleeds while on treatment, and about 38% had no bleeds of any kind.
The median annualized bleeding rate among patients treated with Adynovate was 2.0 (range, 0-49.8), and the mean was 3.0, which is comparable to the rates seen in adults treated with the product.
“These initial efficacy and safety findings indicate a potentially valuable role for Adynovate to treat pediatric patients with hemophilia A, with data consistent with what was reported in clinical studies among adult patients,” said John Orloff, MD, head of research & development and chief scientific officer at Baxalta.
With these results, the company plans to file for marketing authorization in Europe and aims to file for a pediatric indication in the US in early 2016.
Adynovate is currently under regulatory review in Japan, Canada, and Switzerland. Baxalta plans to present the complete data from this study at a congress in 2016.

An antihemophilic factor that was recently approved in the US has met the primary endpoint of a phase 3 study, according to the product’s maker, Baxalta.
The product is Adynovate, a recombinant pegylated factor VIII (FVIII) treatment based on the full-length Advate molecule.
The study enrolled patients younger than 12 years of age who had severe hemophilia A. And, thus far, none of these patients has developed inhibitors to Adynovate, which was the study’s primary endpoint.
Adynovate also proved effective for preventing bleeds in some patients and did not appear to produce any serious adverse events.
Adynovate was approved in the US last month for use as routine prophylaxis and on-demand treatment and control of bleeding episodes in hemophilia A patients age 12 and older.
Baxalta said the phase 3 data will form the basis of a US filing for a pediatric indication in early 2016.
The study was designed to assess the safety and immunogenicity of Adynovate in young, previously treated patients with severe hemophilia A. The study enrolled 73 such patients.
Researchers assessed the hemostatic efficacy of Adynovate as prophylaxis and for treatment of bleeding episodes. All patients received prophylactic Adynovate treatment (a median of 1.9 infusions per week) and were followed for 6 months.
The study’s primary endpoint was met, as no patients developed inhibitory antibodies to the drug. In addition, no treatment-related serious adverse events were reported.
About 73% of patients had no joint bleeds while on treatment, and about 38% had no bleeds of any kind.
The median annualized bleeding rate among patients treated with Adynovate was 2.0 (range, 0-49.8), and the mean was 3.0, which is comparable to the rates seen in adults treated with the product.
“These initial efficacy and safety findings indicate a potentially valuable role for Adynovate to treat pediatric patients with hemophilia A, with data consistent with what was reported in clinical studies among adult patients,” said John Orloff, MD, head of research & development and chief scientific officer at Baxalta.
With these results, the company plans to file for marketing authorization in Europe and aims to file for a pediatric indication in the US in early 2016.
Adynovate is currently under regulatory review in Japan, Canada, and Switzerland. Baxalta plans to present the complete data from this study at a congress in 2016.
LAAC a better long-term value than anticoagulants, team says

Photo courtesy of
Boston Scientific
Researchers have compared the cost-effectiveness of left atrial appendage closure (LAAC) with the Watchman device to treatment with warfarin or novel oral anticoagulants (NOACs) as stroke prophylaxis in patients with non-valvular atrial fibrillation.
The results showed that, in the long-term, both NOACs and LAAC were cost-effective relative to warfarin.
LAAC was cost-saving relative to warfarin by year 10 and relative to NOACs by year 5. NOACs were not cost-saving relative to warfarin over the 20-year period studied.
Vivek Reddy, MD, of the Mount Sinai Medical Center in New York, New York, and his colleagues reported these findings in the Journal of American College of Cardiology.
The researchers used a Markov model to evaluate the cost-effectiveness of warfarin, NOACs, and LAAC with the Watchman device from the perspective of the Centers for Medicare & Medicaid Services over a lifetime horizon, which was defined as 20 years.
The patients were 70 years of age and at moderate risk for stroke and bleeding. Clinical event rates, stroke outcomes, and quality of life information were drawn predominantly from 4-year data from the PROTECT AF trial and meta-analyses of warfarin and NOACs.
Costs were presented in 2015 US dollars. The researchers assessed cost-effectiveness annually to determine if there was an observable time horizon over which treatment options reached accepted levels of cost-effectiveness.
They found that LAAC was more expensive than warfarin in the first post-procedure year, and patients had fewer quality-adjusted life years (QALYs) with LAAC.
But by year 3, LAAC-treated patients had gained more QALYs than warfarin-treated patients. And LAAC became cost-effective relative to warfarin by year 7, with a cost per QALY gained of $42,994.
By year 10, LAAC was more effective and less costly than warfarin. This was true for the remainder of the 20-year time horizon. Over a lifetime, LAAC provided an additional 0.506 life-years and 0.638 QALYs relative to warfarin.
NOACs were more effective than warfarin in year 1 and achieved cost-effectiveness relative to warfarin at year 16, with a cost per QALY gained of $48,446.
NOACs were not cost-saving relative to warfarin over 20 years. However, the incremental cost-effectiveness ratio continued to decrease over time so the cost per QALY gained at 20 years was $40,602.
Compared to NOACs, LAAC was more expensive in the first year post-procedure. However, by year 5, LAAC was less expensive—$20,892 vs $20,924—and more effective—3.455 QALYs vs 3.448 QALYs. This remained true for the rest of the time horizon.
Over a patient’s lifetime, LAAC was estimated to provide an additional 0.298 life-years and 0.349 QALYs relative to NOACs.
Dr Reddy and other authors of this paper are paid consultants to Boston Scientific, manufacturer of the Watchman device. Another author is a full-time employee of Boston Scientific, and yet another author has a financial interest in the Watchman device. Dr Reddy is also a paid consultant for and has received grant support from Coherex and St. Jude Medical.

Photo courtesy of
Boston Scientific
Researchers have compared the cost-effectiveness of left atrial appendage closure (LAAC) with the Watchman device to treatment with warfarin or novel oral anticoagulants (NOACs) as stroke prophylaxis in patients with non-valvular atrial fibrillation.
The results showed that, in the long-term, both NOACs and LAAC were cost-effective relative to warfarin.
LAAC was cost-saving relative to warfarin by year 10 and relative to NOACs by year 5. NOACs were not cost-saving relative to warfarin over the 20-year period studied.
Vivek Reddy, MD, of the Mount Sinai Medical Center in New York, New York, and his colleagues reported these findings in the Journal of American College of Cardiology.
The researchers used a Markov model to evaluate the cost-effectiveness of warfarin, NOACs, and LAAC with the Watchman device from the perspective of the Centers for Medicare & Medicaid Services over a lifetime horizon, which was defined as 20 years.
The patients were 70 years of age and at moderate risk for stroke and bleeding. Clinical event rates, stroke outcomes, and quality of life information were drawn predominantly from 4-year data from the PROTECT AF trial and meta-analyses of warfarin and NOACs.
Costs were presented in 2015 US dollars. The researchers assessed cost-effectiveness annually to determine if there was an observable time horizon over which treatment options reached accepted levels of cost-effectiveness.
They found that LAAC was more expensive than warfarin in the first post-procedure year, and patients had fewer quality-adjusted life years (QALYs) with LAAC.
But by year 3, LAAC-treated patients had gained more QALYs than warfarin-treated patients. And LAAC became cost-effective relative to warfarin by year 7, with a cost per QALY gained of $42,994.
By year 10, LAAC was more effective and less costly than warfarin. This was true for the remainder of the 20-year time horizon. Over a lifetime, LAAC provided an additional 0.506 life-years and 0.638 QALYs relative to warfarin.
NOACs were more effective than warfarin in year 1 and achieved cost-effectiveness relative to warfarin at year 16, with a cost per QALY gained of $48,446.
NOACs were not cost-saving relative to warfarin over 20 years. However, the incremental cost-effectiveness ratio continued to decrease over time so the cost per QALY gained at 20 years was $40,602.
Compared to NOACs, LAAC was more expensive in the first year post-procedure. However, by year 5, LAAC was less expensive—$20,892 vs $20,924—and more effective—3.455 QALYs vs 3.448 QALYs. This remained true for the rest of the time horizon.
Over a patient’s lifetime, LAAC was estimated to provide an additional 0.298 life-years and 0.349 QALYs relative to NOACs.
Dr Reddy and other authors of this paper are paid consultants to Boston Scientific, manufacturer of the Watchman device. Another author is a full-time employee of Boston Scientific, and yet another author has a financial interest in the Watchman device. Dr Reddy is also a paid consultant for and has received grant support from Coherex and St. Jude Medical.

Photo courtesy of
Boston Scientific
Researchers have compared the cost-effectiveness of left atrial appendage closure (LAAC) with the Watchman device to treatment with warfarin or novel oral anticoagulants (NOACs) as stroke prophylaxis in patients with non-valvular atrial fibrillation.
The results showed that, in the long-term, both NOACs and LAAC were cost-effective relative to warfarin.
LAAC was cost-saving relative to warfarin by year 10 and relative to NOACs by year 5. NOACs were not cost-saving relative to warfarin over the 20-year period studied.
Vivek Reddy, MD, of the Mount Sinai Medical Center in New York, New York, and his colleagues reported these findings in the Journal of American College of Cardiology.
The researchers used a Markov model to evaluate the cost-effectiveness of warfarin, NOACs, and LAAC with the Watchman device from the perspective of the Centers for Medicare & Medicaid Services over a lifetime horizon, which was defined as 20 years.
The patients were 70 years of age and at moderate risk for stroke and bleeding. Clinical event rates, stroke outcomes, and quality of life information were drawn predominantly from 4-year data from the PROTECT AF trial and meta-analyses of warfarin and NOACs.
Costs were presented in 2015 US dollars. The researchers assessed cost-effectiveness annually to determine if there was an observable time horizon over which treatment options reached accepted levels of cost-effectiveness.
They found that LAAC was more expensive than warfarin in the first post-procedure year, and patients had fewer quality-adjusted life years (QALYs) with LAAC.
But by year 3, LAAC-treated patients had gained more QALYs than warfarin-treated patients. And LAAC became cost-effective relative to warfarin by year 7, with a cost per QALY gained of $42,994.
By year 10, LAAC was more effective and less costly than warfarin. This was true for the remainder of the 20-year time horizon. Over a lifetime, LAAC provided an additional 0.506 life-years and 0.638 QALYs relative to warfarin.
NOACs were more effective than warfarin in year 1 and achieved cost-effectiveness relative to warfarin at year 16, with a cost per QALY gained of $48,446.
NOACs were not cost-saving relative to warfarin over 20 years. However, the incremental cost-effectiveness ratio continued to decrease over time so the cost per QALY gained at 20 years was $40,602.
Compared to NOACs, LAAC was more expensive in the first year post-procedure. However, by year 5, LAAC was less expensive—$20,892 vs $20,924—and more effective—3.455 QALYs vs 3.448 QALYs. This remained true for the rest of the time horizon.
Over a patient’s lifetime, LAAC was estimated to provide an additional 0.298 life-years and 0.349 QALYs relative to NOACs.
Dr Reddy and other authors of this paper are paid consultants to Boston Scientific, manufacturer of the Watchman device. Another author is a full-time employee of Boston Scientific, and yet another author has a financial interest in the Watchman device. Dr Reddy is also a paid consultant for and has received grant support from Coherex and St. Jude Medical.