User login
Physical activity can benefit kids with cancer

Photo by Bill Branson
Children with cancer can benefit from “adapted” physical activities, according to a pilot study published in ecancermedicalscience.
Investigators followed 11 cancer patients, ages 10 to 18, on a dog sledding expedition, which involved adapted training activities.
All of the patients completed the training and the expedition, and they exhibited significant improvements in physical and psychological health after completing the program.
“What I learned from this study is that we doctors have the false belief that kids with cancer cannot practice sport because they are too tired or weak from their treatments,” said study author Nicolas André, MD, PhD, of Hôpital d'Enfants de la Timone in Marseille, France.
“These perceptions are at least partly wrong. Adapted physical activities can be performed by most children with cancer, even during their treatment, and can bring a lot to children.”
To arrive at these conclusions, Dr André and his colleagues measured the effects of a 6-week long adapted physical activity program on children and adolescents with cancer.
The study included 11 patients—4 girls and 7 boys—with a mean age of 14.3 ± 2.9 years. Seven of the patients were still receiving treatment.
About the program
The patients first completed a physical preparation program consisting of general conditioning to improve their strength, speed, endurance, flexibility, skill, and ability to handle greater workloads.
Typically, these activities lasted from 60 to 120 minutes and took place 1 to 5 times a week. The intensity of physical activity was adjusted to each subject.
After completing the preparation, the patients began a 5-day long expedition in Quebec, Canada. They completed a session of physical training in the snow and had their first contact with the pack of dogs and the sleds the day before departure.
Overall, the subjects engaged in physical activity 4 to 5 hours per day during the expedition.
Results
The patients performed a series of physical tests and completed psychological questionnaires before and after the program. The results showed improvements in all physical and psychological parameters.
After completing the program, the subjects reported significant improvements in global self-esteem (P=0.02), perceived sport competence (P=0.02), and perceived physical strength (P=0.001).
They also demonstrated significant improvements in their ability to do sit-ups (P=0.01), their muscle tone (P=0.01), and their resting heart rate (P=0.03).
“Based on our work over the last 8 years, we all are convinced that practicing adapted physical activity is very positive for children with cancer,” said study author Laurent Grélot, PhD, of Aix Marseille University in France.
“It avoids cardiovascular and muscular deconditioning, can decrease treatment-induced fatigue, and can help maintaining social integration.”
Based on the success of this pilot study, the investigators are conducting a randomized trial to evaluate the benefits of adapted physical activities for children with cancer. ![]()

Photo by Bill Branson
Children with cancer can benefit from “adapted” physical activities, according to a pilot study published in ecancermedicalscience.
Investigators followed 11 cancer patients, ages 10 to 18, on a dog sledding expedition, which involved adapted training activities.
All of the patients completed the training and the expedition, and they exhibited significant improvements in physical and psychological health after completing the program.
“What I learned from this study is that we doctors have the false belief that kids with cancer cannot practice sport because they are too tired or weak from their treatments,” said study author Nicolas André, MD, PhD, of Hôpital d'Enfants de la Timone in Marseille, France.
“These perceptions are at least partly wrong. Adapted physical activities can be performed by most children with cancer, even during their treatment, and can bring a lot to children.”
To arrive at these conclusions, Dr André and his colleagues measured the effects of a 6-week long adapted physical activity program on children and adolescents with cancer.
The study included 11 patients—4 girls and 7 boys—with a mean age of 14.3 ± 2.9 years. Seven of the patients were still receiving treatment.
About the program
The patients first completed a physical preparation program consisting of general conditioning to improve their strength, speed, endurance, flexibility, skill, and ability to handle greater workloads.
Typically, these activities lasted from 60 to 120 minutes and took place 1 to 5 times a week. The intensity of physical activity was adjusted to each subject.
After completing the preparation, the patients began a 5-day long expedition in Quebec, Canada. They completed a session of physical training in the snow and had their first contact with the pack of dogs and the sleds the day before departure.
Overall, the subjects engaged in physical activity 4 to 5 hours per day during the expedition.
Results
The patients performed a series of physical tests and completed psychological questionnaires before and after the program. The results showed improvements in all physical and psychological parameters.
After completing the program, the subjects reported significant improvements in global self-esteem (P=0.02), perceived sport competence (P=0.02), and perceived physical strength (P=0.001).
They also demonstrated significant improvements in their ability to do sit-ups (P=0.01), their muscle tone (P=0.01), and their resting heart rate (P=0.03).
“Based on our work over the last 8 years, we all are convinced that practicing adapted physical activity is very positive for children with cancer,” said study author Laurent Grélot, PhD, of Aix Marseille University in France.
“It avoids cardiovascular and muscular deconditioning, can decrease treatment-induced fatigue, and can help maintaining social integration.”
Based on the success of this pilot study, the investigators are conducting a randomized trial to evaluate the benefits of adapted physical activities for children with cancer. ![]()

Photo by Bill Branson
Children with cancer can benefit from “adapted” physical activities, according to a pilot study published in ecancermedicalscience.
Investigators followed 11 cancer patients, ages 10 to 18, on a dog sledding expedition, which involved adapted training activities.
All of the patients completed the training and the expedition, and they exhibited significant improvements in physical and psychological health after completing the program.
“What I learned from this study is that we doctors have the false belief that kids with cancer cannot practice sport because they are too tired or weak from their treatments,” said study author Nicolas André, MD, PhD, of Hôpital d'Enfants de la Timone in Marseille, France.
“These perceptions are at least partly wrong. Adapted physical activities can be performed by most children with cancer, even during their treatment, and can bring a lot to children.”
To arrive at these conclusions, Dr André and his colleagues measured the effects of a 6-week long adapted physical activity program on children and adolescents with cancer.
The study included 11 patients—4 girls and 7 boys—with a mean age of 14.3 ± 2.9 years. Seven of the patients were still receiving treatment.
About the program
The patients first completed a physical preparation program consisting of general conditioning to improve their strength, speed, endurance, flexibility, skill, and ability to handle greater workloads.
Typically, these activities lasted from 60 to 120 minutes and took place 1 to 5 times a week. The intensity of physical activity was adjusted to each subject.
After completing the preparation, the patients began a 5-day long expedition in Quebec, Canada. They completed a session of physical training in the snow and had their first contact with the pack of dogs and the sleds the day before departure.
Overall, the subjects engaged in physical activity 4 to 5 hours per day during the expedition.
Results
The patients performed a series of physical tests and completed psychological questionnaires before and after the program. The results showed improvements in all physical and psychological parameters.
After completing the program, the subjects reported significant improvements in global self-esteem (P=0.02), perceived sport competence (P=0.02), and perceived physical strength (P=0.001).
They also demonstrated significant improvements in their ability to do sit-ups (P=0.01), their muscle tone (P=0.01), and their resting heart rate (P=0.03).
“Based on our work over the last 8 years, we all are convinced that practicing adapted physical activity is very positive for children with cancer,” said study author Laurent Grélot, PhD, of Aix Marseille University in France.
“It avoids cardiovascular and muscular deconditioning, can decrease treatment-induced fatigue, and can help maintaining social integration.”
Based on the success of this pilot study, the investigators are conducting a randomized trial to evaluate the benefits of adapted physical activities for children with cancer. ![]()
Recombinant vWF can safely halt bleeding in vWD
Results of a phase 3 trial suggest the recombinant von Willebrand factor (rvWF) product BAX 111 can safely control bleeding in patients with severe von Willebrand disease (vWD).
BAX 111, given with or without recombinant factor VIII (rFVIII), controlled bleeding episodes in 100% of patients treated.
None of the patients developed vWF or FVIII inhibitors, anti-VWF binding antibodies, or antibodies against host cell proteins.
There were 8 adverse events (AEs) associated with BAX 111, 2 of which were serious.
Investigators reported these results in Blood. The trial was sponsored by Baxalta, Inc., the company developing BAX 111.
Compared to other marketed vWF concentrates, BAX 111 contains a more consistent concentration of ultra-large-molecular-weight multimers. And the product is developed using a plasma- and albumin-free manufacturing method.
For the phase 3 trial, investigators set out to test BAX 111 in 49 patients who had received vWF concentrate treatment for at least 1 severe vWD-related bleed within the last 12 months. The patients’ median age was 37 (range, 18 to 64), and there were slightly more females (54.1%) than males (45.9%).
To observe BAX 111’s activity at varying doses and in different treatment scenarios, the investigators administered BAX 111 at 50 IU/kg or 80 IU/kg either alone or with rFVIII.
The majority of patients received as-needed treatment of BAX 111 at 40-60 IU/kg for regular bleeding episodes and up to 80 IU/kg for major bleeds. About 32% of treated subjects (7/22) also received an antifibrinolytic agent, such as tranexamic acid, during bleeding episodes.
In all, 22 patients experienced at least 1 bleeding episode that was treated with BAX 111. Seventeen of these patients had type 3 vWD, 4 had type 2A, and 1 had type 2N.
The investigators assessed 192 bleeding episodes treated with BAX 111 for hemostatic efficacy. Most bleeds (n=122) were minor, 61 were moderate, 7 were major/severe, and 2 were of unknown severity.
Efficacy
All 192 bleeds were treated successfully. When vWD specialists rated BAX 111’s ability to control bleeds on a scale of “excellent” to “no response,” they rated BAX 111 as “excellent” (96.9%) or “good” (3.1%).
One infusion of BAX 111 was sufficient in treating most bleeds (81.8%, n=157). One mucosal bleed (occurring in the genital tract and oral cavity simultaneously) required 4 infusions, which was the maximum number of infusions administered during the study.
The median doses of BAX 111 and rFVIII administered per bleed were 46.5 IU/kg and 33.6 IU/kg, respectively.
The activity of BAX 111 remained the same whether it was given with or without rFVIII. Patients who received BAX 111 alone experienced a rapid increase in their naturally produced FVIII.
In fact, within 6 hours of an infusion, patients had produced enough of the protein for proper clotting. This response was sustained through 72 hours post-infusion, suggesting that patients who receive BAX 111 are not likely to require additional rFVIII infusions.
Safety
There were 125 AEs in this trial, but only 8 were considered related to BAX 111. Six AEs (occurring in 4 patients) were not serious. These included mild infusion site paresthesia, moderate dysgeusia, moderate tachycardia, mild ECG T-wave inversion, mild generalized pruritus, and mild hot flush.
One patient experienced 2 simultaneous serious AEs—chest discomfort and increased heart rate. The symptoms improved after 10 minutes of oxygen treatment, and the subject fully recovered within 3 hours. The subject had a history of allergic responses to cryoprecipitate and a plasma-derived vWF concentrate.
None of the patients developed anti-VWF neutralizing or binding antibodies, FVIII neutralizing antibodies, or antibodies against recombinant furin, Chinese hamster ovary host cell proteins, or murine immunoglobulin G.
There were no clinical signs of thrombotic events and no clinically significant abnormal D-dimer values post-treatment.
“These efficacy and safety data . . . represent a major advance in our quest to develop an optimal treatment for people living with vWD,” said study investigator Bruce Ewenstein, MD, PhD, of Baxalta, Inc.
“As this product is specifically designed to be administered without FVIII, it will allow physicians to dose vWF and FVIII separately and precisely based on the needs of each individual patient. This treatment strategy has the potential to become the standard of care for patients with severe von Willebrand disease.” ![]()

Results of a phase 3 trial suggest the recombinant von Willebrand factor (rvWF) product BAX 111 can safely control bleeding in patients with severe von Willebrand disease (vWD).
BAX 111, given with or without recombinant factor VIII (rFVIII), controlled bleeding episodes in 100% of patients treated.
None of the patients developed vWF or FVIII inhibitors, anti-VWF binding antibodies, or antibodies against host cell proteins.
There were 8 adverse events (AEs) associated with BAX 111, 2 of which were serious.
Investigators reported these results in Blood. The trial was sponsored by Baxalta, Inc., the company developing BAX 111.
Compared to other marketed vWF concentrates, BAX 111 contains a more consistent concentration of ultra-large-molecular-weight multimers. And the product is developed using a plasma- and albumin-free manufacturing method.
For the phase 3 trial, investigators set out to test BAX 111 in 49 patients who had received vWF concentrate treatment for at least 1 severe vWD-related bleed within the last 12 months. The patients’ median age was 37 (range, 18 to 64), and there were slightly more females (54.1%) than males (45.9%).
To observe BAX 111’s activity at varying doses and in different treatment scenarios, the investigators administered BAX 111 at 50 IU/kg or 80 IU/kg either alone or with rFVIII.
The majority of patients received as-needed treatment of BAX 111 at 40-60 IU/kg for regular bleeding episodes and up to 80 IU/kg for major bleeds. About 32% of treated subjects (7/22) also received an antifibrinolytic agent, such as tranexamic acid, during bleeding episodes.
In all, 22 patients experienced at least 1 bleeding episode that was treated with BAX 111. Seventeen of these patients had type 3 vWD, 4 had type 2A, and 1 had type 2N.
The investigators assessed 192 bleeding episodes treated with BAX 111 for hemostatic efficacy. Most bleeds (n=122) were minor, 61 were moderate, 7 were major/severe, and 2 were of unknown severity.
Efficacy
All 192 bleeds were treated successfully. When vWD specialists rated BAX 111’s ability to control bleeds on a scale of “excellent” to “no response,” they rated BAX 111 as “excellent” (96.9%) or “good” (3.1%).
One infusion of BAX 111 was sufficient in treating most bleeds (81.8%, n=157). One mucosal bleed (occurring in the genital tract and oral cavity simultaneously) required 4 infusions, which was the maximum number of infusions administered during the study.
The median doses of BAX 111 and rFVIII administered per bleed were 46.5 IU/kg and 33.6 IU/kg, respectively.
The activity of BAX 111 remained the same whether it was given with or without rFVIII. Patients who received BAX 111 alone experienced a rapid increase in their naturally produced FVIII.
In fact, within 6 hours of an infusion, patients had produced enough of the protein for proper clotting. This response was sustained through 72 hours post-infusion, suggesting that patients who receive BAX 111 are not likely to require additional rFVIII infusions.
Safety
There were 125 AEs in this trial, but only 8 were considered related to BAX 111. Six AEs (occurring in 4 patients) were not serious. These included mild infusion site paresthesia, moderate dysgeusia, moderate tachycardia, mild ECG T-wave inversion, mild generalized pruritus, and mild hot flush.
One patient experienced 2 simultaneous serious AEs—chest discomfort and increased heart rate. The symptoms improved after 10 minutes of oxygen treatment, and the subject fully recovered within 3 hours. The subject had a history of allergic responses to cryoprecipitate and a plasma-derived vWF concentrate.
None of the patients developed anti-VWF neutralizing or binding antibodies, FVIII neutralizing antibodies, or antibodies against recombinant furin, Chinese hamster ovary host cell proteins, or murine immunoglobulin G.
There were no clinical signs of thrombotic events and no clinically significant abnormal D-dimer values post-treatment.
“These efficacy and safety data . . . represent a major advance in our quest to develop an optimal treatment for people living with vWD,” said study investigator Bruce Ewenstein, MD, PhD, of Baxalta, Inc.
“As this product is specifically designed to be administered without FVIII, it will allow physicians to dose vWF and FVIII separately and precisely based on the needs of each individual patient. This treatment strategy has the potential to become the standard of care for patients with severe von Willebrand disease.” ![]()

Results of a phase 3 trial suggest the recombinant von Willebrand factor (rvWF) product BAX 111 can safely control bleeding in patients with severe von Willebrand disease (vWD).
BAX 111, given with or without recombinant factor VIII (rFVIII), controlled bleeding episodes in 100% of patients treated.
None of the patients developed vWF or FVIII inhibitors, anti-VWF binding antibodies, or antibodies against host cell proteins.
There were 8 adverse events (AEs) associated with BAX 111, 2 of which were serious.
Investigators reported these results in Blood. The trial was sponsored by Baxalta, Inc., the company developing BAX 111.
Compared to other marketed vWF concentrates, BAX 111 contains a more consistent concentration of ultra-large-molecular-weight multimers. And the product is developed using a plasma- and albumin-free manufacturing method.
For the phase 3 trial, investigators set out to test BAX 111 in 49 patients who had received vWF concentrate treatment for at least 1 severe vWD-related bleed within the last 12 months. The patients’ median age was 37 (range, 18 to 64), and there were slightly more females (54.1%) than males (45.9%).
To observe BAX 111’s activity at varying doses and in different treatment scenarios, the investigators administered BAX 111 at 50 IU/kg or 80 IU/kg either alone or with rFVIII.
The majority of patients received as-needed treatment of BAX 111 at 40-60 IU/kg for regular bleeding episodes and up to 80 IU/kg for major bleeds. About 32% of treated subjects (7/22) also received an antifibrinolytic agent, such as tranexamic acid, during bleeding episodes.
In all, 22 patients experienced at least 1 bleeding episode that was treated with BAX 111. Seventeen of these patients had type 3 vWD, 4 had type 2A, and 1 had type 2N.
The investigators assessed 192 bleeding episodes treated with BAX 111 for hemostatic efficacy. Most bleeds (n=122) were minor, 61 were moderate, 7 were major/severe, and 2 were of unknown severity.
Efficacy
All 192 bleeds were treated successfully. When vWD specialists rated BAX 111’s ability to control bleeds on a scale of “excellent” to “no response,” they rated BAX 111 as “excellent” (96.9%) or “good” (3.1%).
One infusion of BAX 111 was sufficient in treating most bleeds (81.8%, n=157). One mucosal bleed (occurring in the genital tract and oral cavity simultaneously) required 4 infusions, which was the maximum number of infusions administered during the study.
The median doses of BAX 111 and rFVIII administered per bleed were 46.5 IU/kg and 33.6 IU/kg, respectively.
The activity of BAX 111 remained the same whether it was given with or without rFVIII. Patients who received BAX 111 alone experienced a rapid increase in their naturally produced FVIII.
In fact, within 6 hours of an infusion, patients had produced enough of the protein for proper clotting. This response was sustained through 72 hours post-infusion, suggesting that patients who receive BAX 111 are not likely to require additional rFVIII infusions.
Safety
There were 125 AEs in this trial, but only 8 were considered related to BAX 111. Six AEs (occurring in 4 patients) were not serious. These included mild infusion site paresthesia, moderate dysgeusia, moderate tachycardia, mild ECG T-wave inversion, mild generalized pruritus, and mild hot flush.
One patient experienced 2 simultaneous serious AEs—chest discomfort and increased heart rate. The symptoms improved after 10 minutes of oxygen treatment, and the subject fully recovered within 3 hours. The subject had a history of allergic responses to cryoprecipitate and a plasma-derived vWF concentrate.
None of the patients developed anti-VWF neutralizing or binding antibodies, FVIII neutralizing antibodies, or antibodies against recombinant furin, Chinese hamster ovary host cell proteins, or murine immunoglobulin G.
There were no clinical signs of thrombotic events and no clinically significant abnormal D-dimer values post-treatment.
“These efficacy and safety data . . . represent a major advance in our quest to develop an optimal treatment for people living with vWD,” said study investigator Bruce Ewenstein, MD, PhD, of Baxalta, Inc.
“As this product is specifically designed to be administered without FVIII, it will allow physicians to dose vWF and FVIII separately and precisely based on the needs of each individual patient. This treatment strategy has the potential to become the standard of care for patients with severe von Willebrand disease.” ![]()
FDA clears 7-day storage of platelets
The US Food and Drug Administration (FDA) has cleared a label change for the Fenwal Amicus system, a cell-separation device, to allow for 7-day storage of platelets collected via this system.
The Fenwal Amicus system is used to collect platelets, platelets with concurrent plasma and red cells, and mononuclear cells.
The label change means platelets collected via the Fenwal Amicus system can now be stored for 7 days instead of 5, but conditions apply.
“Our customers now have the ability to store and use 7-day platelets processed by Amicus to help meet an important need in transfusion medicine,” said Dean Gregory, of Fresenius Kabi North America, the company that manufactures the Fenwal Amicus system.
“We designed Amicus to help optimize apheresis programs for blood-collection professionals. With this new clearance, the Amicus system becomes an even more versatile choice.”
Previously, platelets were limited to a shelf life of 5 days due to the absence of appropriate bacterial testing methods and appropriate instructions for use.
The label change for the Fenwal Amicus system means blood centers that want to store apheresis-derived platelets for 7 days must label every product with a statement that the product must be tested with a bacterial detection device cleared by the FDA and labeled as a “safety measure.”
To support this requirement, Fresenius Kabi distributes the Verax Platelet PGD test, a rapid platelet PGD test that is cleared by the FDA as a “safety measure” for use in leukoreduced apheresis platelets 24 hours prior to transfusion.
For more information on the Verax Platelet PGD test, visit the Verax Biomedical website. For more information on the Fenwal Amicus system, visit the Fenwal website. ![]()

The US Food and Drug Administration (FDA) has cleared a label change for the Fenwal Amicus system, a cell-separation device, to allow for 7-day storage of platelets collected via this system.
The Fenwal Amicus system is used to collect platelets, platelets with concurrent plasma and red cells, and mononuclear cells.
The label change means platelets collected via the Fenwal Amicus system can now be stored for 7 days instead of 5, but conditions apply.
“Our customers now have the ability to store and use 7-day platelets processed by Amicus to help meet an important need in transfusion medicine,” said Dean Gregory, of Fresenius Kabi North America, the company that manufactures the Fenwal Amicus system.
“We designed Amicus to help optimize apheresis programs for blood-collection professionals. With this new clearance, the Amicus system becomes an even more versatile choice.”
Previously, platelets were limited to a shelf life of 5 days due to the absence of appropriate bacterial testing methods and appropriate instructions for use.
The label change for the Fenwal Amicus system means blood centers that want to store apheresis-derived platelets for 7 days must label every product with a statement that the product must be tested with a bacterial detection device cleared by the FDA and labeled as a “safety measure.”
To support this requirement, Fresenius Kabi distributes the Verax Platelet PGD test, a rapid platelet PGD test that is cleared by the FDA as a “safety measure” for use in leukoreduced apheresis platelets 24 hours prior to transfusion.
For more information on the Verax Platelet PGD test, visit the Verax Biomedical website. For more information on the Fenwal Amicus system, visit the Fenwal website. ![]()

The US Food and Drug Administration (FDA) has cleared a label change for the Fenwal Amicus system, a cell-separation device, to allow for 7-day storage of platelets collected via this system.
The Fenwal Amicus system is used to collect platelets, platelets with concurrent plasma and red cells, and mononuclear cells.
The label change means platelets collected via the Fenwal Amicus system can now be stored for 7 days instead of 5, but conditions apply.
“Our customers now have the ability to store and use 7-day platelets processed by Amicus to help meet an important need in transfusion medicine,” said Dean Gregory, of Fresenius Kabi North America, the company that manufactures the Fenwal Amicus system.
“We designed Amicus to help optimize apheresis programs for blood-collection professionals. With this new clearance, the Amicus system becomes an even more versatile choice.”
Previously, platelets were limited to a shelf life of 5 days due to the absence of appropriate bacterial testing methods and appropriate instructions for use.
The label change for the Fenwal Amicus system means blood centers that want to store apheresis-derived platelets for 7 days must label every product with a statement that the product must be tested with a bacterial detection device cleared by the FDA and labeled as a “safety measure.”
To support this requirement, Fresenius Kabi distributes the Verax Platelet PGD test, a rapid platelet PGD test that is cleared by the FDA as a “safety measure” for use in leukoreduced apheresis platelets 24 hours prior to transfusion.
For more information on the Verax Platelet PGD test, visit the Verax Biomedical website. For more information on the Fenwal Amicus system, visit the Fenwal website. ![]()
Group creates vascular graft that resists thrombosis
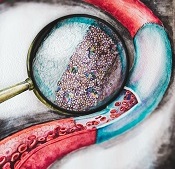
The enlarged section shows
the drug-loaded film that
coats the inside of the graft.
Image courtesy of ITMO
University/Yulia Chapurina
Researchers say they have developed a vascular graft that may be able to resist thrombosis indefinitely.
The team found they could trap urokinase-type plasminogen activator inside a porous, alumina sol–gel film, and this film retained its thrombolytic activity when applied to a polymer vascular graft.
Vladimir Vinogradov, PhD, of ITMO University in St. Petersburg, Russia, and his colleagues described this work in the Journal of Medicinal Chemistry.
The team synthesized a thin film made of densely packed aluminum oxide nanorods blended with molecules of the thrombolytic enzyme urokinase-type plasminogen activator.
When adhered to the inner surface of a polymer vascular graft, the film causes the parietal area of the graft to fill with a stable concentration of plasmin, which is capable of dissolving any clots that appear.
The researchers said the unique properties of the film arise from its structure, which represents a porous matrix, accommodating the plasminogen activator.
The matrix protects the plasminogen activator from the aggressive environment of the organism while also preserving the enzyme’s ability to interact with certain external agents through a system of pores.
In particular, the matrix lets in plasminogen. When plasminogen meets the plasminogen activator inside the matrix, clot-dissolving plasmin forms.
“In order to test how our improved vascular graft worked, we grew an artificial clot made of blood plasma mixed with thrombin and placed it inside the graft,” said Yulia Chapurina, also of ITMO University.
“The results of the experiment amazed us. Very soon, the clot started to dissolve and leak through the graft. In reality, our coating would destroy clots at the stage of formation, constantly ensuring an unobstructed blood flow in the graft.”
The researchers noted that the lifetime of drug-eluting grafts currently in use is often determined by the amount of drug stored within them. In essence, this means they can only postpone clot formation.
But the graft the researchers developed traps the drug inside a porous, protective shell, which, the team said, makes the lifetime of such a graft practically unlimited.
“Our approach is concept-based and can be applied not only to artificial blood vessels, but to any kind of implants,” Dr Vinogradov said. “You just need to take the right kind of drug.”
“For example, after the implantation of an artificial ureter, urease crystals often start to grow inside, and doctors do not know how to deal with this problem. It is possible to apply a similar drug-containing coating that dissolves urease. The same approach may be used for kidney or liver surgery, but these are plans for the future.” ![]()

The enlarged section shows
the drug-loaded film that
coats the inside of the graft.
Image courtesy of ITMO
University/Yulia Chapurina
Researchers say they have developed a vascular graft that may be able to resist thrombosis indefinitely.
The team found they could trap urokinase-type plasminogen activator inside a porous, alumina sol–gel film, and this film retained its thrombolytic activity when applied to a polymer vascular graft.
Vladimir Vinogradov, PhD, of ITMO University in St. Petersburg, Russia, and his colleagues described this work in the Journal of Medicinal Chemistry.
The team synthesized a thin film made of densely packed aluminum oxide nanorods blended with molecules of the thrombolytic enzyme urokinase-type plasminogen activator.
When adhered to the inner surface of a polymer vascular graft, the film causes the parietal area of the graft to fill with a stable concentration of plasmin, which is capable of dissolving any clots that appear.
The researchers said the unique properties of the film arise from its structure, which represents a porous matrix, accommodating the plasminogen activator.
The matrix protects the plasminogen activator from the aggressive environment of the organism while also preserving the enzyme’s ability to interact with certain external agents through a system of pores.
In particular, the matrix lets in plasminogen. When plasminogen meets the plasminogen activator inside the matrix, clot-dissolving plasmin forms.
“In order to test how our improved vascular graft worked, we grew an artificial clot made of blood plasma mixed with thrombin and placed it inside the graft,” said Yulia Chapurina, also of ITMO University.
“The results of the experiment amazed us. Very soon, the clot started to dissolve and leak through the graft. In reality, our coating would destroy clots at the stage of formation, constantly ensuring an unobstructed blood flow in the graft.”
The researchers noted that the lifetime of drug-eluting grafts currently in use is often determined by the amount of drug stored within them. In essence, this means they can only postpone clot formation.
But the graft the researchers developed traps the drug inside a porous, protective shell, which, the team said, makes the lifetime of such a graft practically unlimited.
“Our approach is concept-based and can be applied not only to artificial blood vessels, but to any kind of implants,” Dr Vinogradov said. “You just need to take the right kind of drug.”
“For example, after the implantation of an artificial ureter, urease crystals often start to grow inside, and doctors do not know how to deal with this problem. It is possible to apply a similar drug-containing coating that dissolves urease. The same approach may be used for kidney or liver surgery, but these are plans for the future.” ![]()

The enlarged section shows
the drug-loaded film that
coats the inside of the graft.
Image courtesy of ITMO
University/Yulia Chapurina
Researchers say they have developed a vascular graft that may be able to resist thrombosis indefinitely.
The team found they could trap urokinase-type plasminogen activator inside a porous, alumina sol–gel film, and this film retained its thrombolytic activity when applied to a polymer vascular graft.
Vladimir Vinogradov, PhD, of ITMO University in St. Petersburg, Russia, and his colleagues described this work in the Journal of Medicinal Chemistry.
The team synthesized a thin film made of densely packed aluminum oxide nanorods blended with molecules of the thrombolytic enzyme urokinase-type plasminogen activator.
When adhered to the inner surface of a polymer vascular graft, the film causes the parietal area of the graft to fill with a stable concentration of plasmin, which is capable of dissolving any clots that appear.
The researchers said the unique properties of the film arise from its structure, which represents a porous matrix, accommodating the plasminogen activator.
The matrix protects the plasminogen activator from the aggressive environment of the organism while also preserving the enzyme’s ability to interact with certain external agents through a system of pores.
In particular, the matrix lets in plasminogen. When plasminogen meets the plasminogen activator inside the matrix, clot-dissolving plasmin forms.
“In order to test how our improved vascular graft worked, we grew an artificial clot made of blood plasma mixed with thrombin and placed it inside the graft,” said Yulia Chapurina, also of ITMO University.
“The results of the experiment amazed us. Very soon, the clot started to dissolve and leak through the graft. In reality, our coating would destroy clots at the stage of formation, constantly ensuring an unobstructed blood flow in the graft.”
The researchers noted that the lifetime of drug-eluting grafts currently in use is often determined by the amount of drug stored within them. In essence, this means they can only postpone clot formation.
But the graft the researchers developed traps the drug inside a porous, protective shell, which, the team said, makes the lifetime of such a graft practically unlimited.
“Our approach is concept-based and can be applied not only to artificial blood vessels, but to any kind of implants,” Dr Vinogradov said. “You just need to take the right kind of drug.”
“For example, after the implantation of an artificial ureter, urease crystals often start to grow inside, and doctors do not know how to deal with this problem. It is possible to apply a similar drug-containing coating that dissolves urease. The same approach may be used for kidney or liver surgery, but these are plans for the future.” ![]()
T-cell activation is a dynamic process, study shows
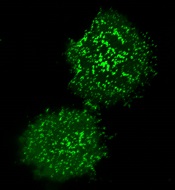
plasma membrane upon T-cell
activation. Image courtesy
of the Salk Institute
New research suggests that, contrary to previous assumptions, T-cell activation is a fluid process that relies on a dynamic protein network at the cell surface.
Previously, scientists thought T-cell activation was a static process in which molecules assemble within a T cell.
Instead, investigators discovered that molecules rapidly come and go, and the times at which these molecules arrive and depart affect the immune response.
“This is a completely new principle for how T-cell activity is controlled—whether it ignores or responds to a threat,” said Björn Lillemeier, PhD, of the Salk Institute in La Jolla, California.
He and his colleagues described this discovery in Nature Immunology.
They noted that the protein kinase ZAP-70 plays a central role in T-cell activation. Until now, scientists assumed that a silent form of ZAP-70 floats around inside the T cell until a threat is detected. The threat recruits ZAP-70 to the cell surface and activates it.
By analyzing mutant forms of ZAP-70, Dr Lillemeier’s group discovered that, instead of ZAP-70 binding the T-cell receptor firmly, it comes in contact with the receptor sporadically. Each time this happens, ZAP-70 has to adopt an unfavorable shape that forces it back inside the cell.
This cycle continues until a second molecule, Lck, helps ZAP-70 remain with the T-cell receptor. The prolonged stay at the cell surface activates ZAP-70 and prompts the T cell to attack invaders and diseased cells.
The investigators said this study suggests the steps underlying T-cell activation are more dynamic than the less mobile modes scientists had suspected before. And the research highlights how ZAP-70 and other molecules communicate in space and time, which is crucial for controlling the ultimate activity of a T cell.
By understanding this process, Dr Lillemeier said, “We might be able to encourage the immune system to be a little more sensitive in order to recognize and eliminate diseases.”
His team is now working to identify new principles that determine if T cells respond to a threat or stay quiet. They are also testing whether their findings could be applied across additional processes in T cells and other immune cells.
Because proteins have many of the same modular building blocks, Dr Lillemeier said that, in principle, any protein with structural characteristics comparable to those of ZAP-70 could be controlled by similar mechanisms. ![]()

plasma membrane upon T-cell
activation. Image courtesy
of the Salk Institute
New research suggests that, contrary to previous assumptions, T-cell activation is a fluid process that relies on a dynamic protein network at the cell surface.
Previously, scientists thought T-cell activation was a static process in which molecules assemble within a T cell.
Instead, investigators discovered that molecules rapidly come and go, and the times at which these molecules arrive and depart affect the immune response.
“This is a completely new principle for how T-cell activity is controlled—whether it ignores or responds to a threat,” said Björn Lillemeier, PhD, of the Salk Institute in La Jolla, California.
He and his colleagues described this discovery in Nature Immunology.
They noted that the protein kinase ZAP-70 plays a central role in T-cell activation. Until now, scientists assumed that a silent form of ZAP-70 floats around inside the T cell until a threat is detected. The threat recruits ZAP-70 to the cell surface and activates it.
By analyzing mutant forms of ZAP-70, Dr Lillemeier’s group discovered that, instead of ZAP-70 binding the T-cell receptor firmly, it comes in contact with the receptor sporadically. Each time this happens, ZAP-70 has to adopt an unfavorable shape that forces it back inside the cell.
This cycle continues until a second molecule, Lck, helps ZAP-70 remain with the T-cell receptor. The prolonged stay at the cell surface activates ZAP-70 and prompts the T cell to attack invaders and diseased cells.
The investigators said this study suggests the steps underlying T-cell activation are more dynamic than the less mobile modes scientists had suspected before. And the research highlights how ZAP-70 and other molecules communicate in space and time, which is crucial for controlling the ultimate activity of a T cell.
By understanding this process, Dr Lillemeier said, “We might be able to encourage the immune system to be a little more sensitive in order to recognize and eliminate diseases.”
His team is now working to identify new principles that determine if T cells respond to a threat or stay quiet. They are also testing whether their findings could be applied across additional processes in T cells and other immune cells.
Because proteins have many of the same modular building blocks, Dr Lillemeier said that, in principle, any protein with structural characteristics comparable to those of ZAP-70 could be controlled by similar mechanisms. ![]()

plasma membrane upon T-cell
activation. Image courtesy
of the Salk Institute
New research suggests that, contrary to previous assumptions, T-cell activation is a fluid process that relies on a dynamic protein network at the cell surface.
Previously, scientists thought T-cell activation was a static process in which molecules assemble within a T cell.
Instead, investigators discovered that molecules rapidly come and go, and the times at which these molecules arrive and depart affect the immune response.
“This is a completely new principle for how T-cell activity is controlled—whether it ignores or responds to a threat,” said Björn Lillemeier, PhD, of the Salk Institute in La Jolla, California.
He and his colleagues described this discovery in Nature Immunology.
They noted that the protein kinase ZAP-70 plays a central role in T-cell activation. Until now, scientists assumed that a silent form of ZAP-70 floats around inside the T cell until a threat is detected. The threat recruits ZAP-70 to the cell surface and activates it.
By analyzing mutant forms of ZAP-70, Dr Lillemeier’s group discovered that, instead of ZAP-70 binding the T-cell receptor firmly, it comes in contact with the receptor sporadically. Each time this happens, ZAP-70 has to adopt an unfavorable shape that forces it back inside the cell.
This cycle continues until a second molecule, Lck, helps ZAP-70 remain with the T-cell receptor. The prolonged stay at the cell surface activates ZAP-70 and prompts the T cell to attack invaders and diseased cells.
The investigators said this study suggests the steps underlying T-cell activation are more dynamic than the less mobile modes scientists had suspected before. And the research highlights how ZAP-70 and other molecules communicate in space and time, which is crucial for controlling the ultimate activity of a T cell.
By understanding this process, Dr Lillemeier said, “We might be able to encourage the immune system to be a little more sensitive in order to recognize and eliminate diseases.”
His team is now working to identify new principles that determine if T cells respond to a threat or stay quiet. They are also testing whether their findings could be applied across additional processes in T cells and other immune cells.
Because proteins have many of the same modular building blocks, Dr Lillemeier said that, in principle, any protein with structural characteristics comparable to those of ZAP-70 could be controlled by similar mechanisms. ![]()
How a molecule turns B cells into macrophages

pseudopodia to engulf particles
The transcription factor C/EBPα reprograms B cells into macrophages by “short-circuiting” the cells so they re-express genes reserved for embryonic development, according to research published in Stem Cell Reports.
Over the past 28 years, researchers have shown that a number of specialized cell types can be forcibly converted into other cell types, but the science of how this change takes place is still emerging.
“For a long time, it was unclear whether forcing cell fate decisions by expressing transcription factors in the wrong cell type could teach us something about what happens normally during physiological differentiation,” said Thomas Graf, PhD, of the Center for Genomic Regulation in Barcelona, Spain.
“What we have now found is that the two processes are actually surprisingly similar.”
The researchers found that B-cell transdifferentiation takes place when C/EBPα binds to two regions of DNA that act as gene expression enhancers. One of these regions is normally active in immune cells, and the other is only turned on when macrophage precursors are ready to differentiate.
This indicates that the convergence of these two enhancer pathways can cause the B cell to act like a macrophage precursor, thus triggering the unnatural transdifferentiation.
“This has taught us a great deal about how a transcription factor can activate a new gene expression program (in our case, that of macrophages) but has left us in the dark about the other part of the equation; namely, how the factor silences the B-cell program, something that must happen if transdifferentiation is to work,” Dr Graf said. “This is one of the questions we are focusing on now.”
Dr Graf is interested in this pathway because C/EBPα-induced, B cell-to-macrophage transdifferentiation can convert both human B-cell lymphoma and leukemia cells into functional, non-cancerous macrophages.
He believes that induced transdifferentiation could become therapeutically relevant if a drug could be found that can replace the transcription factor. And understanding the mechanisms of the process could help labs that use this transdifferentiation approach to generate cells for regenerative purposes. ![]()

pseudopodia to engulf particles
The transcription factor C/EBPα reprograms B cells into macrophages by “short-circuiting” the cells so they re-express genes reserved for embryonic development, according to research published in Stem Cell Reports.
Over the past 28 years, researchers have shown that a number of specialized cell types can be forcibly converted into other cell types, but the science of how this change takes place is still emerging.
“For a long time, it was unclear whether forcing cell fate decisions by expressing transcription factors in the wrong cell type could teach us something about what happens normally during physiological differentiation,” said Thomas Graf, PhD, of the Center for Genomic Regulation in Barcelona, Spain.
“What we have now found is that the two processes are actually surprisingly similar.”
The researchers found that B-cell transdifferentiation takes place when C/EBPα binds to two regions of DNA that act as gene expression enhancers. One of these regions is normally active in immune cells, and the other is only turned on when macrophage precursors are ready to differentiate.
This indicates that the convergence of these two enhancer pathways can cause the B cell to act like a macrophage precursor, thus triggering the unnatural transdifferentiation.
“This has taught us a great deal about how a transcription factor can activate a new gene expression program (in our case, that of macrophages) but has left us in the dark about the other part of the equation; namely, how the factor silences the B-cell program, something that must happen if transdifferentiation is to work,” Dr Graf said. “This is one of the questions we are focusing on now.”
Dr Graf is interested in this pathway because C/EBPα-induced, B cell-to-macrophage transdifferentiation can convert both human B-cell lymphoma and leukemia cells into functional, non-cancerous macrophages.
He believes that induced transdifferentiation could become therapeutically relevant if a drug could be found that can replace the transcription factor. And understanding the mechanisms of the process could help labs that use this transdifferentiation approach to generate cells for regenerative purposes. ![]()

pseudopodia to engulf particles
The transcription factor C/EBPα reprograms B cells into macrophages by “short-circuiting” the cells so they re-express genes reserved for embryonic development, according to research published in Stem Cell Reports.
Over the past 28 years, researchers have shown that a number of specialized cell types can be forcibly converted into other cell types, but the science of how this change takes place is still emerging.
“For a long time, it was unclear whether forcing cell fate decisions by expressing transcription factors in the wrong cell type could teach us something about what happens normally during physiological differentiation,” said Thomas Graf, PhD, of the Center for Genomic Regulation in Barcelona, Spain.
“What we have now found is that the two processes are actually surprisingly similar.”
The researchers found that B-cell transdifferentiation takes place when C/EBPα binds to two regions of DNA that act as gene expression enhancers. One of these regions is normally active in immune cells, and the other is only turned on when macrophage precursors are ready to differentiate.
This indicates that the convergence of these two enhancer pathways can cause the B cell to act like a macrophage precursor, thus triggering the unnatural transdifferentiation.
“This has taught us a great deal about how a transcription factor can activate a new gene expression program (in our case, that of macrophages) but has left us in the dark about the other part of the equation; namely, how the factor silences the B-cell program, something that must happen if transdifferentiation is to work,” Dr Graf said. “This is one of the questions we are focusing on now.”
Dr Graf is interested in this pathway because C/EBPα-induced, B cell-to-macrophage transdifferentiation can convert both human B-cell lymphoma and leukemia cells into functional, non-cancerous macrophages.
He believes that induced transdifferentiation could become therapeutically relevant if a drug could be found that can replace the transcription factor. And understanding the mechanisms of the process could help labs that use this transdifferentiation approach to generate cells for regenerative purposes. ![]()
Microfluidics can improve epigenomic analysis, team says

his student, Zhenning Cao.
Dr Lu is holding the chip
used in the study.
Photo from Virginia Tech
A new technique enables epigenomic analysis using fewer cells than other methods involving chromatin immunoprecipitation and deep sequencing (ChIP-seq), according to researchers.
The technique, microfluidic oscillatory washing-based ChIP-seq (MOWChIP-seq), allows for genome-wide analysis of histone modifications using as few as 100
cells.
Using MOWChIP-seq, researchers uncovered new information regarding early hematopoiesis.
The team described this work in Nature Methods.
They used multilayer soft lithography to create a poly (dimethylsiloxane) device with a microfluidic chamber for ChIP. The researchers flowed magnetic beads coated with a ChIP antibody into the chamber, which formed a packed bed.
They then flowed sonicated chromatin fragments through the chamber, which were adsorbed onto the bead surface. The team said the gaps between the immunoprecipitation beads are smaller than 2 μm and facilitate rapid, high-efficiency adsorption of target chromatin fragments under the small diffusion length.
The researchers washed the beads by oscillatory washing in two different buffers to remove nonspecifically adsorbed chromatin fragments. Then, they flowed the beads out of the chamber and collected them for off-chip processing.
“The use of a packed bed of beads for ChIP allowed us to collect the chromatin fragments with a very high efficiency,” said study author Chang Lu, PhD, of Virginia Tech in Blacksburg.
“At the same time, effective washing for removing undesired molecules and debris guarantees the purity of the collected molecules. These two factors constitute a successful strategy for epigenomic analysis with extremely high sensitivity.”
In addition, the entire MOWChIP-seq process takes about 90 minutes.
To test MOWChIP-seq, Dr Lu and his colleagues used the technique to study the epigenomes of hematopoietic stem and progenitor cells (HSPCs) isolated from the fetal liver of a mouse.
“Little is known about the dynamics of the epigenome during embryonic hematopoiesis, largely due to the difficulty in isolating sufficient quantities of these cells from developing embryos,” said study author Kai Tan, PhD, of the University of Iowa in Iowa City. “This technology is the perfect tool for tackling this problem.”
MOWChIP-seq revealed new enhancers and super enhancers in the HSPCs.
By comparing all of the enhancers they identified to an enhancer catalog covering 16 blood cell types, the researchers found that 2561 (58%) of the enhancers they found were unique to fetal liver HSPCs. They said this suggests enhancer activity is highly dynamic during early hematopoiesis.
Now, the researchers plan to use MOWChIP-seq to study other epigenomic changes involved in inflammation and cancer.
“Our technology paves the way for studies of epigenomes with extremely low numbers of cells from animals and from patients,” Dr Lu said.
Virginia Tech Intellectual Properties has filed a utility patent on MOWChIP-seq on behalf of Dr Lu. ![]()

his student, Zhenning Cao.
Dr Lu is holding the chip
used in the study.
Photo from Virginia Tech
A new technique enables epigenomic analysis using fewer cells than other methods involving chromatin immunoprecipitation and deep sequencing (ChIP-seq), according to researchers.
The technique, microfluidic oscillatory washing-based ChIP-seq (MOWChIP-seq), allows for genome-wide analysis of histone modifications using as few as 100
cells.
Using MOWChIP-seq, researchers uncovered new information regarding early hematopoiesis.
The team described this work in Nature Methods.
They used multilayer soft lithography to create a poly (dimethylsiloxane) device with a microfluidic chamber for ChIP. The researchers flowed magnetic beads coated with a ChIP antibody into the chamber, which formed a packed bed.
They then flowed sonicated chromatin fragments through the chamber, which were adsorbed onto the bead surface. The team said the gaps between the immunoprecipitation beads are smaller than 2 μm and facilitate rapid, high-efficiency adsorption of target chromatin fragments under the small diffusion length.
The researchers washed the beads by oscillatory washing in two different buffers to remove nonspecifically adsorbed chromatin fragments. Then, they flowed the beads out of the chamber and collected them for off-chip processing.
“The use of a packed bed of beads for ChIP allowed us to collect the chromatin fragments with a very high efficiency,” said study author Chang Lu, PhD, of Virginia Tech in Blacksburg.
“At the same time, effective washing for removing undesired molecules and debris guarantees the purity of the collected molecules. These two factors constitute a successful strategy for epigenomic analysis with extremely high sensitivity.”
In addition, the entire MOWChIP-seq process takes about 90 minutes.
To test MOWChIP-seq, Dr Lu and his colleagues used the technique to study the epigenomes of hematopoietic stem and progenitor cells (HSPCs) isolated from the fetal liver of a mouse.
“Little is known about the dynamics of the epigenome during embryonic hematopoiesis, largely due to the difficulty in isolating sufficient quantities of these cells from developing embryos,” said study author Kai Tan, PhD, of the University of Iowa in Iowa City. “This technology is the perfect tool for tackling this problem.”
MOWChIP-seq revealed new enhancers and super enhancers in the HSPCs.
By comparing all of the enhancers they identified to an enhancer catalog covering 16 blood cell types, the researchers found that 2561 (58%) of the enhancers they found were unique to fetal liver HSPCs. They said this suggests enhancer activity is highly dynamic during early hematopoiesis.
Now, the researchers plan to use MOWChIP-seq to study other epigenomic changes involved in inflammation and cancer.
“Our technology paves the way for studies of epigenomes with extremely low numbers of cells from animals and from patients,” Dr Lu said.
Virginia Tech Intellectual Properties has filed a utility patent on MOWChIP-seq on behalf of Dr Lu. ![]()

his student, Zhenning Cao.
Dr Lu is holding the chip
used in the study.
Photo from Virginia Tech
A new technique enables epigenomic analysis using fewer cells than other methods involving chromatin immunoprecipitation and deep sequencing (ChIP-seq), according to researchers.
The technique, microfluidic oscillatory washing-based ChIP-seq (MOWChIP-seq), allows for genome-wide analysis of histone modifications using as few as 100
cells.
Using MOWChIP-seq, researchers uncovered new information regarding early hematopoiesis.
The team described this work in Nature Methods.
They used multilayer soft lithography to create a poly (dimethylsiloxane) device with a microfluidic chamber for ChIP. The researchers flowed magnetic beads coated with a ChIP antibody into the chamber, which formed a packed bed.
They then flowed sonicated chromatin fragments through the chamber, which were adsorbed onto the bead surface. The team said the gaps between the immunoprecipitation beads are smaller than 2 μm and facilitate rapid, high-efficiency adsorption of target chromatin fragments under the small diffusion length.
The researchers washed the beads by oscillatory washing in two different buffers to remove nonspecifically adsorbed chromatin fragments. Then, they flowed the beads out of the chamber and collected them for off-chip processing.
“The use of a packed bed of beads for ChIP allowed us to collect the chromatin fragments with a very high efficiency,” said study author Chang Lu, PhD, of Virginia Tech in Blacksburg.
“At the same time, effective washing for removing undesired molecules and debris guarantees the purity of the collected molecules. These two factors constitute a successful strategy for epigenomic analysis with extremely high sensitivity.”
In addition, the entire MOWChIP-seq process takes about 90 minutes.
To test MOWChIP-seq, Dr Lu and his colleagues used the technique to study the epigenomes of hematopoietic stem and progenitor cells (HSPCs) isolated from the fetal liver of a mouse.
“Little is known about the dynamics of the epigenome during embryonic hematopoiesis, largely due to the difficulty in isolating sufficient quantities of these cells from developing embryos,” said study author Kai Tan, PhD, of the University of Iowa in Iowa City. “This technology is the perfect tool for tackling this problem.”
MOWChIP-seq revealed new enhancers and super enhancers in the HSPCs.
By comparing all of the enhancers they identified to an enhancer catalog covering 16 blood cell types, the researchers found that 2561 (58%) of the enhancers they found were unique to fetal liver HSPCs. They said this suggests enhancer activity is highly dynamic during early hematopoiesis.
Now, the researchers plan to use MOWChIP-seq to study other epigenomic changes involved in inflammation and cancer.
“Our technology paves the way for studies of epigenomes with extremely low numbers of cells from animals and from patients,” Dr Lu said.
Virginia Tech Intellectual Properties has filed a utility patent on MOWChIP-seq on behalf of Dr Lu.
Newfound mechanism could be used to fight cancers

apoptosis in cancer cells
Researchers say they have identified a new mechanism by which the tumor suppressor protein p53 triggers apoptosis, and they believe this process could be harnessed to kill cancer cells.
The team discovered how p53 acts in the cytoplasm to trigger cell death by binding to and activating the BAX protein.
The process involves a shape change in one of p53’s amino acids that serves as the “switch” to activate BAX and trigger the apoptotic pathway.
The team also identified the enzyme in the cytoplasm that promotes the change that controls the “switch.”
Richard Kriwacki, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee, and his colleagues described these findings in Molecular Cell.
Like up to half of all proteins, p53 includes both structured and disordered regions. A disordered region is a segment that does not adopt a single shape but
remains flexible and constantly switches between different shapes until
it encounters a partner.
Dr Kriwacki and his colleagues showed that both structured and disordered regions of p53 play a role in BAX activation in the cytoplasm.
The process starts when a structured region of p53 known as the DNA-binding domain binds to BAX. That sets the stage for the unstructured region of p53 to form a second bond, which activates BAX and triggers apoptosis.
“There were no previous reports of this disordered region of p53 binding to BAX, so the finding that this region was the key to BAX activation was a total surprise,” Dr Kriwacki said.
The disordered p53 segment included the amino acid proline, which can change between two shapes, particularly in the presence of the enzyme Pin1.
Using NMR spectroscopy, the researchers showed that the proline shape change promotes p53 binding and activation of BAX.
“These results expand our understanding of the different ways p53 modulates cell behavior,” Dr Kriwacki said. “The findings also raise the possibility of killing tumor cells using small molecules to trigger BAX-dependent apoptosis.”

apoptosis in cancer cells
Researchers say they have identified a new mechanism by which the tumor suppressor protein p53 triggers apoptosis, and they believe this process could be harnessed to kill cancer cells.
The team discovered how p53 acts in the cytoplasm to trigger cell death by binding to and activating the BAX protein.
The process involves a shape change in one of p53’s amino acids that serves as the “switch” to activate BAX and trigger the apoptotic pathway.
The team also identified the enzyme in the cytoplasm that promotes the change that controls the “switch.”
Richard Kriwacki, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee, and his colleagues described these findings in Molecular Cell.
Like up to half of all proteins, p53 includes both structured and disordered regions. A disordered region is a segment that does not adopt a single shape but
remains flexible and constantly switches between different shapes until
it encounters a partner.
Dr Kriwacki and his colleagues showed that both structured and disordered regions of p53 play a role in BAX activation in the cytoplasm.
The process starts when a structured region of p53 known as the DNA-binding domain binds to BAX. That sets the stage for the unstructured region of p53 to form a second bond, which activates BAX and triggers apoptosis.
“There were no previous reports of this disordered region of p53 binding to BAX, so the finding that this region was the key to BAX activation was a total surprise,” Dr Kriwacki said.
The disordered p53 segment included the amino acid proline, which can change between two shapes, particularly in the presence of the enzyme Pin1.
Using NMR spectroscopy, the researchers showed that the proline shape change promotes p53 binding and activation of BAX.
“These results expand our understanding of the different ways p53 modulates cell behavior,” Dr Kriwacki said. “The findings also raise the possibility of killing tumor cells using small molecules to trigger BAX-dependent apoptosis.”

apoptosis in cancer cells
Researchers say they have identified a new mechanism by which the tumor suppressor protein p53 triggers apoptosis, and they believe this process could be harnessed to kill cancer cells.
The team discovered how p53 acts in the cytoplasm to trigger cell death by binding to and activating the BAX protein.
The process involves a shape change in one of p53’s amino acids that serves as the “switch” to activate BAX and trigger the apoptotic pathway.
The team also identified the enzyme in the cytoplasm that promotes the change that controls the “switch.”
Richard Kriwacki, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee, and his colleagues described these findings in Molecular Cell.
Like up to half of all proteins, p53 includes both structured and disordered regions. A disordered region is a segment that does not adopt a single shape but
remains flexible and constantly switches between different shapes until
it encounters a partner.
Dr Kriwacki and his colleagues showed that both structured and disordered regions of p53 play a role in BAX activation in the cytoplasm.
The process starts when a structured region of p53 known as the DNA-binding domain binds to BAX. That sets the stage for the unstructured region of p53 to form a second bond, which activates BAX and triggers apoptosis.
“There were no previous reports of this disordered region of p53 binding to BAX, so the finding that this region was the key to BAX activation was a total surprise,” Dr Kriwacki said.
The disordered p53 segment included the amino acid proline, which can change between two shapes, particularly in the presence of the enzyme Pin1.
Using NMR spectroscopy, the researchers showed that the proline shape change promotes p53 binding and activation of BAX.
“These results expand our understanding of the different ways p53 modulates cell behavior,” Dr Kriwacki said. “The findings also raise the possibility of killing tumor cells using small molecules to trigger BAX-dependent apoptosis.”
Delayed cancer diagnosis tied to care dissatisfaction

Photo courtesy of NIH
Patients who make 3 or more trips to the general practitioner (GP) before they are referred for cancer tests are more likely to be dissatisfied with subsequent care, according to research published in the European Journal of Cancer Care.
Researchers analyzed survey responses from nearly 60,000 cancer patients and found that about 23% had visited their GP 3 or more times before they were referred for cancer tests.
These patients were more likely than patients with 1 or 2 GP visits to report negative experiences with regard to many different aspects of their care.
“This research shows that first impressions go a long way in determining how cancer patients view their experience of cancer treatment,” said study author Georgios Lyratzopoulos, MD, of University College London in the UK.
“A negative experience of diagnosis can trigger loss of confidence in their care throughout the cancer journey. When they occur, diagnostic delays are largely due to cancer symptoms being extremely hard to distinguish from other diseases, combined with a lack of accurate and easy-to-use tests. New diagnostic tools to help doctors decide which patients need referring are vital to improve the care experience for even more cancer patients.”
Dr Lyratzopoulos and his colleagues initially analyzed survey data from 73,462 cancer patients, but 15,355 of those patients (21%) had not seen a GP prior to their cancer diagnosis.
Of the 58,107 patients who had at least 1 primary care consultation, 44,827 (77%) had seen their GP once or twice before referral for cancer tests, and 13,280 (23%) had seen the GP 3 or more times.
The researchers analyzed patients’ responses to 12 survey questions that assessed satisfaction with various aspects of their cancer care.
And results showed that patients who had seen their GP 3 or more times before referral were significantly more likely than patients with 1 to 2 GP visits to report negative experiences across 10 of the 12 aspects of care.
The 12 aspects of care and patient responses (for 3+ GP visits and 1-2 visits, respectively) were as follows:
- Dissatisfied with overall care: 16% vs 9% (adjusted* odds ratio [aOR]=1.44, P<0.001)
- Dissatisfied with the way they were informed of their cancer: 22% vs 14% (aOR=1.38, P<0.001)
- Dissatisfied with their involvement in decision-making: 32% vs 25% (aOR=1.13, P<0.001)
- Lack of confidence/trust in their doctors: 19% vs 12% (aOR=1.22, P<0.001)
- Lack of confidence/trust in their nurses: 36% vs 27% (aOR=1.22, P<0.001)
- Thought doctors/nurses deliberately withheld information: 16% vs 10% (aOR=1.27, P<0.001)
- Dissatisfied with staff support: 41% vs 27% (aOR=1.68, P<0.001)
- Dissatisfied with integration of various caregivers (GP, hospital doctors/nurses, specialist nurses, etc): 45% vs 32% (aOR=1.48, P<0.001)
- Dissatisfied with the amount of time they had to wait at appointments with their hospital doctor: 31% vs 28% (aOR=1.10, P=0.007)
- Said their GP did not receive enough information about their condition or hospital treatment: 8% vs 5% (aOR=1.36, P<0.001)
- Said they did not receive clear written information about what they should or should not do after leaving the hospital: 20% vs 14% (aOR=1.06, P=0.100)
- Said they did not receive the name of a Clinical Nurse Specialist who would be in charge of their care: 11% vs 9% (aOR=1.00, P=0.894).
“This is the first time we’ve had direct feedback from patients on such a large scale to show how the timeliness of their diagnosis colors their experience of the care they later receive,” said Sara Hiom, of Cancer Research UK, which supported this study.
“It’s another good reason to highlight the importance of diagnosing cancer as quickly as possible, not just to give patients the best chances of survival, but also to improve their experience of the care they receive throughout their cancer journey.”
*The odds ratio was adjusted for patient age, sex, ethnicity, cancer diagnosis, and response tendency (likelihood of giving a positive or negative response to questions).

Photo courtesy of NIH
Patients who make 3 or more trips to the general practitioner (GP) before they are referred for cancer tests are more likely to be dissatisfied with subsequent care, according to research published in the European Journal of Cancer Care.
Researchers analyzed survey responses from nearly 60,000 cancer patients and found that about 23% had visited their GP 3 or more times before they were referred for cancer tests.
These patients were more likely than patients with 1 or 2 GP visits to report negative experiences with regard to many different aspects of their care.
“This research shows that first impressions go a long way in determining how cancer patients view their experience of cancer treatment,” said study author Georgios Lyratzopoulos, MD, of University College London in the UK.
“A negative experience of diagnosis can trigger loss of confidence in their care throughout the cancer journey. When they occur, diagnostic delays are largely due to cancer symptoms being extremely hard to distinguish from other diseases, combined with a lack of accurate and easy-to-use tests. New diagnostic tools to help doctors decide which patients need referring are vital to improve the care experience for even more cancer patients.”
Dr Lyratzopoulos and his colleagues initially analyzed survey data from 73,462 cancer patients, but 15,355 of those patients (21%) had not seen a GP prior to their cancer diagnosis.
Of the 58,107 patients who had at least 1 primary care consultation, 44,827 (77%) had seen their GP once or twice before referral for cancer tests, and 13,280 (23%) had seen the GP 3 or more times.
The researchers analyzed patients’ responses to 12 survey questions that assessed satisfaction with various aspects of their cancer care.
And results showed that patients who had seen their GP 3 or more times before referral were significantly more likely than patients with 1 to 2 GP visits to report negative experiences across 10 of the 12 aspects of care.
The 12 aspects of care and patient responses (for 3+ GP visits and 1-2 visits, respectively) were as follows:
- Dissatisfied with overall care: 16% vs 9% (adjusted* odds ratio [aOR]=1.44, P<0.001)
- Dissatisfied with the way they were informed of their cancer: 22% vs 14% (aOR=1.38, P<0.001)
- Dissatisfied with their involvement in decision-making: 32% vs 25% (aOR=1.13, P<0.001)
- Lack of confidence/trust in their doctors: 19% vs 12% (aOR=1.22, P<0.001)
- Lack of confidence/trust in their nurses: 36% vs 27% (aOR=1.22, P<0.001)
- Thought doctors/nurses deliberately withheld information: 16% vs 10% (aOR=1.27, P<0.001)
- Dissatisfied with staff support: 41% vs 27% (aOR=1.68, P<0.001)
- Dissatisfied with integration of various caregivers (GP, hospital doctors/nurses, specialist nurses, etc): 45% vs 32% (aOR=1.48, P<0.001)
- Dissatisfied with the amount of time they had to wait at appointments with their hospital doctor: 31% vs 28% (aOR=1.10, P=0.007)
- Said their GP did not receive enough information about their condition or hospital treatment: 8% vs 5% (aOR=1.36, P<0.001)
- Said they did not receive clear written information about what they should or should not do after leaving the hospital: 20% vs 14% (aOR=1.06, P=0.100)
- Said they did not receive the name of a Clinical Nurse Specialist who would be in charge of their care: 11% vs 9% (aOR=1.00, P=0.894).
“This is the first time we’ve had direct feedback from patients on such a large scale to show how the timeliness of their diagnosis colors their experience of the care they later receive,” said Sara Hiom, of Cancer Research UK, which supported this study.
“It’s another good reason to highlight the importance of diagnosing cancer as quickly as possible, not just to give patients the best chances of survival, but also to improve their experience of the care they receive throughout their cancer journey.”
*The odds ratio was adjusted for patient age, sex, ethnicity, cancer diagnosis, and response tendency (likelihood of giving a positive or negative response to questions).

Photo courtesy of NIH
Patients who make 3 or more trips to the general practitioner (GP) before they are referred for cancer tests are more likely to be dissatisfied with subsequent care, according to research published in the European Journal of Cancer Care.
Researchers analyzed survey responses from nearly 60,000 cancer patients and found that about 23% had visited their GP 3 or more times before they were referred for cancer tests.
These patients were more likely than patients with 1 or 2 GP visits to report negative experiences with regard to many different aspects of their care.
“This research shows that first impressions go a long way in determining how cancer patients view their experience of cancer treatment,” said study author Georgios Lyratzopoulos, MD, of University College London in the UK.
“A negative experience of diagnosis can trigger loss of confidence in their care throughout the cancer journey. When they occur, diagnostic delays are largely due to cancer symptoms being extremely hard to distinguish from other diseases, combined with a lack of accurate and easy-to-use tests. New diagnostic tools to help doctors decide which patients need referring are vital to improve the care experience for even more cancer patients.”
Dr Lyratzopoulos and his colleagues initially analyzed survey data from 73,462 cancer patients, but 15,355 of those patients (21%) had not seen a GP prior to their cancer diagnosis.
Of the 58,107 patients who had at least 1 primary care consultation, 44,827 (77%) had seen their GP once or twice before referral for cancer tests, and 13,280 (23%) had seen the GP 3 or more times.
The researchers analyzed patients’ responses to 12 survey questions that assessed satisfaction with various aspects of their cancer care.
And results showed that patients who had seen their GP 3 or more times before referral were significantly more likely than patients with 1 to 2 GP visits to report negative experiences across 10 of the 12 aspects of care.
The 12 aspects of care and patient responses (for 3+ GP visits and 1-2 visits, respectively) were as follows:
- Dissatisfied with overall care: 16% vs 9% (adjusted* odds ratio [aOR]=1.44, P<0.001)
- Dissatisfied with the way they were informed of their cancer: 22% vs 14% (aOR=1.38, P<0.001)
- Dissatisfied with their involvement in decision-making: 32% vs 25% (aOR=1.13, P<0.001)
- Lack of confidence/trust in their doctors: 19% vs 12% (aOR=1.22, P<0.001)
- Lack of confidence/trust in their nurses: 36% vs 27% (aOR=1.22, P<0.001)
- Thought doctors/nurses deliberately withheld information: 16% vs 10% (aOR=1.27, P<0.001)
- Dissatisfied with staff support: 41% vs 27% (aOR=1.68, P<0.001)
- Dissatisfied with integration of various caregivers (GP, hospital doctors/nurses, specialist nurses, etc): 45% vs 32% (aOR=1.48, P<0.001)
- Dissatisfied with the amount of time they had to wait at appointments with their hospital doctor: 31% vs 28% (aOR=1.10, P=0.007)
- Said their GP did not receive enough information about their condition or hospital treatment: 8% vs 5% (aOR=1.36, P<0.001)
- Said they did not receive clear written information about what they should or should not do after leaving the hospital: 20% vs 14% (aOR=1.06, P=0.100)
- Said they did not receive the name of a Clinical Nurse Specialist who would be in charge of their care: 11% vs 9% (aOR=1.00, P=0.894).
“This is the first time we’ve had direct feedback from patients on such a large scale to show how the timeliness of their diagnosis colors their experience of the care they later receive,” said Sara Hiom, of Cancer Research UK, which supported this study.
“It’s another good reason to highlight the importance of diagnosing cancer as quickly as possible, not just to give patients the best chances of survival, but also to improve their experience of the care they receive throughout their cancer journey.”
*The odds ratio was adjusted for patient age, sex, ethnicity, cancer diagnosis, and response tendency (likelihood of giving a positive or negative response to questions).
Combo may overcome TKI resistance in CML

Schürch, MD, PhD, (left)
and Adrian Ochsenbein, MD
Photo by Susi Bürki
Combining a tyrosine kinase inhibitor (TKI) with a monoclonal antibody (mAb) may circumvent TKI resistance in chronic myeloid leukemia (CML), according to preclinical research published in Science Translational Medicine.
To understand how TKI resistance develops, researchers analyzed the effect of these drugs on BCR-ABL1+ leukemia cell lines, cells from patients with newly diagnosed CML, and mouse models of CML.
The team found that TKIs induce CD70 expression in leukemic stem cells (LSCs) by downregulating microRNA-29. This results in reduced CD70 promoter DNA methylation and upregulation of the transcription factor specificity protein 1 (SP1).
The increase in CD70 triggers CD27 signaling and compensatory Wnt pathway activation. The researchers said this suggests LSCs evade TKIs by activating Wnt signaling through this route.
So the team hypothesized that combination treatment with a TKI and a mAb blocking the CD70/CD27 interaction would eradicate LSCs.
First, they tested an αCD27 mAb alone or in combination with a TKI in leukemia cell lines. Compared to either agent alone, αCD27/imatinib cotreatment significantly (P<0.001) reduced cell growth by inhibiting proliferation and enhancing apoptosis in SD-1 cells.
The researchers observed similar results when they tested the αCD27 mAb and nilotinib in SD-1 cells, as well as when they tested the αCD27 mAb with imatinib or ponatinib in KBM5 and KBM5r cells.
The team noted that αCD27/imatinib cotreatment inhibited Wnt pathway activation significantly stronger than either compound alone (P<0.001) but had little to no effect on Notch, Hedgehog, and MAP kinase pathways.
The researchers also conducted in vitro tests with an αCD70 mAb (clone 41D12-D) that was specifically designed to block the CD70/CD27 interaction without inducing effector functions such as antibody-dependent cell- or complement-mediated cytotoxicity and antibody-dependent cell-mediated phagocytosis.
They found that αCD70/imatinib cotreatment “potently reduced” CD34+ CML stem/progenitor cells in liquid cultures by inhibiting proliferation and increasing apoptosis. The combination also significantly impaired colony formation in semisolid cultures when compared to either agent alone (P<0.05).
In addition, αCD70/imatinib cotreatment eliminated human CD34+ CML stem/progenitor cells in murine xenografts. The LSCs were completely eradicated in 9 of 12 mice treated.
In a murine CML model, combination treatment with imatinib and an αCD70 mAb (clone FR70) significantly improved survival (P<0.001) compared to either agent alone. And 60% of mice (9 of 15) that received the combination were alive 90 days after transplantation.
The researchers said this suggests the LSCs were completely eradicated or at least effectively controlled long-term.

Schürch, MD, PhD, (left)
and Adrian Ochsenbein, MD
Photo by Susi Bürki
Combining a tyrosine kinase inhibitor (TKI) with a monoclonal antibody (mAb) may circumvent TKI resistance in chronic myeloid leukemia (CML), according to preclinical research published in Science Translational Medicine.
To understand how TKI resistance develops, researchers analyzed the effect of these drugs on BCR-ABL1+ leukemia cell lines, cells from patients with newly diagnosed CML, and mouse models of CML.
The team found that TKIs induce CD70 expression in leukemic stem cells (LSCs) by downregulating microRNA-29. This results in reduced CD70 promoter DNA methylation and upregulation of the transcription factor specificity protein 1 (SP1).
The increase in CD70 triggers CD27 signaling and compensatory Wnt pathway activation. The researchers said this suggests LSCs evade TKIs by activating Wnt signaling through this route.
So the team hypothesized that combination treatment with a TKI and a mAb blocking the CD70/CD27 interaction would eradicate LSCs.
First, they tested an αCD27 mAb alone or in combination with a TKI in leukemia cell lines. Compared to either agent alone, αCD27/imatinib cotreatment significantly (P<0.001) reduced cell growth by inhibiting proliferation and enhancing apoptosis in SD-1 cells.
The researchers observed similar results when they tested the αCD27 mAb and nilotinib in SD-1 cells, as well as when they tested the αCD27 mAb with imatinib or ponatinib in KBM5 and KBM5r cells.
The team noted that αCD27/imatinib cotreatment inhibited Wnt pathway activation significantly stronger than either compound alone (P<0.001) but had little to no effect on Notch, Hedgehog, and MAP kinase pathways.
The researchers also conducted in vitro tests with an αCD70 mAb (clone 41D12-D) that was specifically designed to block the CD70/CD27 interaction without inducing effector functions such as antibody-dependent cell- or complement-mediated cytotoxicity and antibody-dependent cell-mediated phagocytosis.
They found that αCD70/imatinib cotreatment “potently reduced” CD34+ CML stem/progenitor cells in liquid cultures by inhibiting proliferation and increasing apoptosis. The combination also significantly impaired colony formation in semisolid cultures when compared to either agent alone (P<0.05).
In addition, αCD70/imatinib cotreatment eliminated human CD34+ CML stem/progenitor cells in murine xenografts. The LSCs were completely eradicated in 9 of 12 mice treated.
In a murine CML model, combination treatment with imatinib and an αCD70 mAb (clone FR70) significantly improved survival (P<0.001) compared to either agent alone. And 60% of mice (9 of 15) that received the combination were alive 90 days after transplantation.
The researchers said this suggests the LSCs were completely eradicated or at least effectively controlled long-term.

Schürch, MD, PhD, (left)
and Adrian Ochsenbein, MD
Photo by Susi Bürki
Combining a tyrosine kinase inhibitor (TKI) with a monoclonal antibody (mAb) may circumvent TKI resistance in chronic myeloid leukemia (CML), according to preclinical research published in Science Translational Medicine.
To understand how TKI resistance develops, researchers analyzed the effect of these drugs on BCR-ABL1+ leukemia cell lines, cells from patients with newly diagnosed CML, and mouse models of CML.
The team found that TKIs induce CD70 expression in leukemic stem cells (LSCs) by downregulating microRNA-29. This results in reduced CD70 promoter DNA methylation and upregulation of the transcription factor specificity protein 1 (SP1).
The increase in CD70 triggers CD27 signaling and compensatory Wnt pathway activation. The researchers said this suggests LSCs evade TKIs by activating Wnt signaling through this route.
So the team hypothesized that combination treatment with a TKI and a mAb blocking the CD70/CD27 interaction would eradicate LSCs.
First, they tested an αCD27 mAb alone or in combination with a TKI in leukemia cell lines. Compared to either agent alone, αCD27/imatinib cotreatment significantly (P<0.001) reduced cell growth by inhibiting proliferation and enhancing apoptosis in SD-1 cells.
The researchers observed similar results when they tested the αCD27 mAb and nilotinib in SD-1 cells, as well as when they tested the αCD27 mAb with imatinib or ponatinib in KBM5 and KBM5r cells.
The team noted that αCD27/imatinib cotreatment inhibited Wnt pathway activation significantly stronger than either compound alone (P<0.001) but had little to no effect on Notch, Hedgehog, and MAP kinase pathways.
The researchers also conducted in vitro tests with an αCD70 mAb (clone 41D12-D) that was specifically designed to block the CD70/CD27 interaction without inducing effector functions such as antibody-dependent cell- or complement-mediated cytotoxicity and antibody-dependent cell-mediated phagocytosis.
They found that αCD70/imatinib cotreatment “potently reduced” CD34+ CML stem/progenitor cells in liquid cultures by inhibiting proliferation and increasing apoptosis. The combination also significantly impaired colony formation in semisolid cultures when compared to either agent alone (P<0.05).
In addition, αCD70/imatinib cotreatment eliminated human CD34+ CML stem/progenitor cells in murine xenografts. The LSCs were completely eradicated in 9 of 12 mice treated.
In a murine CML model, combination treatment with imatinib and an αCD70 mAb (clone FR70) significantly improved survival (P<0.001) compared to either agent alone. And 60% of mice (9 of 15) that received the combination were alive 90 days after transplantation.
The researchers said this suggests the LSCs were completely eradicated or at least effectively controlled long-term.