User login
How B-cell lymphoma evades NK cells

Image by Joshua Strokes
Researchers say they have determined how lymphoma cells evade natural killer (NK) cells, and this discovery has revealed potential solutions to the problem.
The team found that NK-cell activation and a second, “triggering” event are both necessary for NK cells to exhibit cytotoxicity in the presence of B-cell lymphoma.
Previous research demonstrated this 2-step process in vitro. Now, researchers have shown that it occurs in vivo.
Dr Ralph Mocikat, of Helmholtz Zentrum München in Munich, Germany, and his colleagues described this research in the European Journal of Immunology.
The team conducted experiments using transplantable tumors, a λ-myc-transgenic model of endogenously arising lymphoma that mimics human Burkitt lymphoma, and mice deficient in the NK group 2 D (NKG2D) receptor.
The experiments showed that NK cells could eliminate lymphoma cells after receiving 2 signals. The first was NK-cell activation, which gave rise to IFN-γ expression.
The researchers found that NK cells could be activated in the presence of MHC class Ilow tumor cells or by injecting bone marrow-derived dendritic cells. Previous research had shown that interleukin 2 (IL-2) and IL-15 could activate NK cells.
The second step involved the NKG2D receptor and its ligands. NKG2D ligands are located on the surface of tumor cells and bind to NK cells. The researchers found that, if these ligands are down-regulated, the NK cells cannot carry out cytotoxic activity.
However, the team found they could increase NKG2D ligand levels. They introduced bortezomib to the tumor cell line 291 and saw a roughly 4-fold increase in NKG2D ligand levels.
“Our results show that the transfer of NK cells is a possible strategic option to treat B-cell lymphoma,” Dr Mocikat said. “According to our findings, this therapeutic approach can be optimized when transferred NK cells are already activated in vitro prior to their injection, thus bypassing the missing activation potential in the tumor microenvironment. An additional injection of IFN-γ or of antibodies against IL-10 could further support the immune activity.” ![]()

Image by Joshua Strokes
Researchers say they have determined how lymphoma cells evade natural killer (NK) cells, and this discovery has revealed potential solutions to the problem.
The team found that NK-cell activation and a second, “triggering” event are both necessary for NK cells to exhibit cytotoxicity in the presence of B-cell lymphoma.
Previous research demonstrated this 2-step process in vitro. Now, researchers have shown that it occurs in vivo.
Dr Ralph Mocikat, of Helmholtz Zentrum München in Munich, Germany, and his colleagues described this research in the European Journal of Immunology.
The team conducted experiments using transplantable tumors, a λ-myc-transgenic model of endogenously arising lymphoma that mimics human Burkitt lymphoma, and mice deficient in the NK group 2 D (NKG2D) receptor.
The experiments showed that NK cells could eliminate lymphoma cells after receiving 2 signals. The first was NK-cell activation, which gave rise to IFN-γ expression.
The researchers found that NK cells could be activated in the presence of MHC class Ilow tumor cells or by injecting bone marrow-derived dendritic cells. Previous research had shown that interleukin 2 (IL-2) and IL-15 could activate NK cells.
The second step involved the NKG2D receptor and its ligands. NKG2D ligands are located on the surface of tumor cells and bind to NK cells. The researchers found that, if these ligands are down-regulated, the NK cells cannot carry out cytotoxic activity.
However, the team found they could increase NKG2D ligand levels. They introduced bortezomib to the tumor cell line 291 and saw a roughly 4-fold increase in NKG2D ligand levels.
“Our results show that the transfer of NK cells is a possible strategic option to treat B-cell lymphoma,” Dr Mocikat said. “According to our findings, this therapeutic approach can be optimized when transferred NK cells are already activated in vitro prior to their injection, thus bypassing the missing activation potential in the tumor microenvironment. An additional injection of IFN-γ or of antibodies against IL-10 could further support the immune activity.” ![]()

Image by Joshua Strokes
Researchers say they have determined how lymphoma cells evade natural killer (NK) cells, and this discovery has revealed potential solutions to the problem.
The team found that NK-cell activation and a second, “triggering” event are both necessary for NK cells to exhibit cytotoxicity in the presence of B-cell lymphoma.
Previous research demonstrated this 2-step process in vitro. Now, researchers have shown that it occurs in vivo.
Dr Ralph Mocikat, of Helmholtz Zentrum München in Munich, Germany, and his colleagues described this research in the European Journal of Immunology.
The team conducted experiments using transplantable tumors, a λ-myc-transgenic model of endogenously arising lymphoma that mimics human Burkitt lymphoma, and mice deficient in the NK group 2 D (NKG2D) receptor.
The experiments showed that NK cells could eliminate lymphoma cells after receiving 2 signals. The first was NK-cell activation, which gave rise to IFN-γ expression.
The researchers found that NK cells could be activated in the presence of MHC class Ilow tumor cells or by injecting bone marrow-derived dendritic cells. Previous research had shown that interleukin 2 (IL-2) and IL-15 could activate NK cells.
The second step involved the NKG2D receptor and its ligands. NKG2D ligands are located on the surface of tumor cells and bind to NK cells. The researchers found that, if these ligands are down-regulated, the NK cells cannot carry out cytotoxic activity.
However, the team found they could increase NKG2D ligand levels. They introduced bortezomib to the tumor cell line 291 and saw a roughly 4-fold increase in NKG2D ligand levels.
“Our results show that the transfer of NK cells is a possible strategic option to treat B-cell lymphoma,” Dr Mocikat said. “According to our findings, this therapeutic approach can be optimized when transferred NK cells are already activated in vitro prior to their injection, thus bypassing the missing activation potential in the tumor microenvironment. An additional injection of IFN-γ or of antibodies against IL-10 could further support the immune activity.” ![]()
AYAs with cancer receive aggressive EOL care

patient and her father
Photo by Rhoda Baer
In a retrospective study, a majority of adolescents and young adults (AYAs) with terminal cancer received aggressive end-of-life (EOL) care.
Investigators looked at the use of intensive care, emergency room visits, chemotherapy use, and hospitalization among more than 600 AYAs with cancer who were treated at Kaiser Permanente in California.
Nearly 70% of patients made use of at least one of these measures in the last month of their life.
The investigators noted that their findings, which were published in JAMA Oncology, may not reflect care for the wider US population. But the study does suggest a need for more research into whether this
pattern reflects AYA cancer patients’ preferences for EOL care.
“A young person facing the end of life is a particularly difficult issue,” said study author Jennifer Mack, MD, MPH, of the Dana-Farber Cancer Institute in Boston, Massachusetts.
“While use of aggressive measures might be an informed decision by young people who would do anything they could to live longer, some interventions come with a cost, which is a poorer quality of life. This study raises questions about what kind of care they’re getting and how we can get them to the best quality of life at the end of their lives.”
The study included 633 patients, ages 15 to 39, who died of cancer between 2001 and 2010. The patients, who received care at Kaiser Permanente Southern California, had either been diagnosed with stage IV cancer or had a recurrence of stage I-III cancer. An initial review of a subset of 111 patients showed that death had been anticipated in 98% of cases.
The most common cancer diagnosis was gastrointestinal cancer (17%), while other common diagnoses were breast cancer (15%), genitourinary cancers (11%), leukemia (14%), and lymphoma (10%).
The investigators measured the use of 4 aggressive treatment measures—intensive care, emergency room visits, chemotherapy, and hospitalization—in patients’ last month of life.
Overall, 68% of patients (449/663) received at least one of these medically intensive EOL care measures. Eleven percent of patients (72/663) received chemotherapy, 22% (144/663) were admitted to the intensive care unit, 22% (147/663) had more than one emergency department visit, and 62% (413/663) were hospitalized.
Rates of hospitalization were higher among patients diagnosed with stage IV disease (66%) than among patients with stage I to III disease—66% and 58%, respectively (P=0.04).
The percentage of patients who received at least one medically intensive EOL care measure was higher in the stage IV cohort as well—71% and 63%, respectively (P=0.03). But there were no significant differences between the cohorts with regard to the other measures.
The investigators said these findings suggest the need to better understand EOL care preferences and decision-making in AYAs with cancer.
“We should think about talking with younger patients earlier about their prognoses, identifying their preferences, and working with them to deliver care that reflects those preferences,” Dr Mack said. “It may be that aggressive care is what they want, but they may end up on this pathway without thoughtful conversation and may be without recognition that they are dying.” ![]()

patient and her father
Photo by Rhoda Baer
In a retrospective study, a majority of adolescents and young adults (AYAs) with terminal cancer received aggressive end-of-life (EOL) care.
Investigators looked at the use of intensive care, emergency room visits, chemotherapy use, and hospitalization among more than 600 AYAs with cancer who were treated at Kaiser Permanente in California.
Nearly 70% of patients made use of at least one of these measures in the last month of their life.
The investigators noted that their findings, which were published in JAMA Oncology, may not reflect care for the wider US population. But the study does suggest a need for more research into whether this
pattern reflects AYA cancer patients’ preferences for EOL care.
“A young person facing the end of life is a particularly difficult issue,” said study author Jennifer Mack, MD, MPH, of the Dana-Farber Cancer Institute in Boston, Massachusetts.
“While use of aggressive measures might be an informed decision by young people who would do anything they could to live longer, some interventions come with a cost, which is a poorer quality of life. This study raises questions about what kind of care they’re getting and how we can get them to the best quality of life at the end of their lives.”
The study included 633 patients, ages 15 to 39, who died of cancer between 2001 and 2010. The patients, who received care at Kaiser Permanente Southern California, had either been diagnosed with stage IV cancer or had a recurrence of stage I-III cancer. An initial review of a subset of 111 patients showed that death had been anticipated in 98% of cases.
The most common cancer diagnosis was gastrointestinal cancer (17%), while other common diagnoses were breast cancer (15%), genitourinary cancers (11%), leukemia (14%), and lymphoma (10%).
The investigators measured the use of 4 aggressive treatment measures—intensive care, emergency room visits, chemotherapy, and hospitalization—in patients’ last month of life.
Overall, 68% of patients (449/663) received at least one of these medically intensive EOL care measures. Eleven percent of patients (72/663) received chemotherapy, 22% (144/663) were admitted to the intensive care unit, 22% (147/663) had more than one emergency department visit, and 62% (413/663) were hospitalized.
Rates of hospitalization were higher among patients diagnosed with stage IV disease (66%) than among patients with stage I to III disease—66% and 58%, respectively (P=0.04).
The percentage of patients who received at least one medically intensive EOL care measure was higher in the stage IV cohort as well—71% and 63%, respectively (P=0.03). But there were no significant differences between the cohorts with regard to the other measures.
The investigators said these findings suggest the need to better understand EOL care preferences and decision-making in AYAs with cancer.
“We should think about talking with younger patients earlier about their prognoses, identifying their preferences, and working with them to deliver care that reflects those preferences,” Dr Mack said. “It may be that aggressive care is what they want, but they may end up on this pathway without thoughtful conversation and may be without recognition that they are dying.” ![]()

patient and her father
Photo by Rhoda Baer
In a retrospective study, a majority of adolescents and young adults (AYAs) with terminal cancer received aggressive end-of-life (EOL) care.
Investigators looked at the use of intensive care, emergency room visits, chemotherapy use, and hospitalization among more than 600 AYAs with cancer who were treated at Kaiser Permanente in California.
Nearly 70% of patients made use of at least one of these measures in the last month of their life.
The investigators noted that their findings, which were published in JAMA Oncology, may not reflect care for the wider US population. But the study does suggest a need for more research into whether this
pattern reflects AYA cancer patients’ preferences for EOL care.
“A young person facing the end of life is a particularly difficult issue,” said study author Jennifer Mack, MD, MPH, of the Dana-Farber Cancer Institute in Boston, Massachusetts.
“While use of aggressive measures might be an informed decision by young people who would do anything they could to live longer, some interventions come with a cost, which is a poorer quality of life. This study raises questions about what kind of care they’re getting and how we can get them to the best quality of life at the end of their lives.”
The study included 633 patients, ages 15 to 39, who died of cancer between 2001 and 2010. The patients, who received care at Kaiser Permanente Southern California, had either been diagnosed with stage IV cancer or had a recurrence of stage I-III cancer. An initial review of a subset of 111 patients showed that death had been anticipated in 98% of cases.
The most common cancer diagnosis was gastrointestinal cancer (17%), while other common diagnoses were breast cancer (15%), genitourinary cancers (11%), leukemia (14%), and lymphoma (10%).
The investigators measured the use of 4 aggressive treatment measures—intensive care, emergency room visits, chemotherapy, and hospitalization—in patients’ last month of life.
Overall, 68% of patients (449/663) received at least one of these medically intensive EOL care measures. Eleven percent of patients (72/663) received chemotherapy, 22% (144/663) were admitted to the intensive care unit, 22% (147/663) had more than one emergency department visit, and 62% (413/663) were hospitalized.
Rates of hospitalization were higher among patients diagnosed with stage IV disease (66%) than among patients with stage I to III disease—66% and 58%, respectively (P=0.04).
The percentage of patients who received at least one medically intensive EOL care measure was higher in the stage IV cohort as well—71% and 63%, respectively (P=0.03). But there were no significant differences between the cohorts with regard to the other measures.
The investigators said these findings suggest the need to better understand EOL care preferences and decision-making in AYAs with cancer.
“We should think about talking with younger patients earlier about their prognoses, identifying their preferences, and working with them to deliver care that reflects those preferences,” Dr Mack said. “It may be that aggressive care is what they want, but they may end up on this pathway without thoughtful conversation and may be without recognition that they are dying.” ![]()
Combo shows potential for treating PEL

reviewing an image
Photo from the University
of Southern California
Immunomodulatory drugs (IMiDs) and BRD4 inhibitors hold promise for treating primary effusion lymphoma (PEL), according to preclinical research.
The IMiDs lenalidomide and pomalidomide had antiproliferative effects in PEL cell lines, and lenalidomide exhibited synergistic cytotoxicity with the BRD4 inhibitors JQ-1, IBET151, and PFI-1.
In mouse models of PEL, treatment with lenalidomide and JQ-1 significantly prolonged survival.
Preet Chaudhary, MD, PhD, of the University of Southern California, Los Angeles, and his colleagues reported these results in Oncogene.
The researchers first tested lenalidomide and pomalidomide in 9 PEL cell lines. Both drugs exhibited antiproliferative effects, at clinically achievable doses, in 6 of the cell lines (BC-3, BCBL-1, JSC-1, VG-1, UMPEL-1, and UMPEL-3).
The IMiDs displayed activity in the remaining 3 cells lines (BC-1, BCP-1, and APK-1) as well, but only at high doses (ranging from 2.6 μM to 10 μM for lenalidomide and from 226 nM to 744 nM for pomalidomide).
Looking into the mechanism of IMiDs in PEL, the researchers found these drugs induce G1 cell-cycle arrest without inducing Kaposi’s sarcoma-associated herpes virus lytic cycle reactivation.
Further investigation revealed that interferon α, β, and γ are cytotoxic to PEL, but they are not essential for the antiproliferative effects of IMiDs. On the other hand, cereblon-dependent suppression of IRF4 and rapid degradation of IKZF1 are essential.
The researchers also found that knocking down MYC enhanced IMiD activity. This led them to BRD4 inhibitors, which are known to target MYC transcription.
The team tested lenalidomide in combination with JQ-1, IBET151, and PFI-1. The IMiD was “highly synergistic” with all 3 BRD4 inhibitors in BC-3 and BCBL-1 cell lines.
In both cell lines, treatment with lenalidomide and JQ-1 significantly decreased the expression of MYC and IRF4 at protein and mRNA levels. The treatment also resulted in G1 cell cycle arrest, cleavage of poly (ADP-ribose) polymerase, and apoptosis.
So the researchers tested this combination in a mouse model of PEL (NOD-SCID mice injected with BC-3 cells). And they observed a significant improvement in median survival for mice that received combination treatment, compared to lenalidomide or JQ-1 alone—51 days, 35 days, and 42 days, respectively (P≤0.01).![]()

reviewing an image
Photo from the University
of Southern California
Immunomodulatory drugs (IMiDs) and BRD4 inhibitors hold promise for treating primary effusion lymphoma (PEL), according to preclinical research.
The IMiDs lenalidomide and pomalidomide had antiproliferative effects in PEL cell lines, and lenalidomide exhibited synergistic cytotoxicity with the BRD4 inhibitors JQ-1, IBET151, and PFI-1.
In mouse models of PEL, treatment with lenalidomide and JQ-1 significantly prolonged survival.
Preet Chaudhary, MD, PhD, of the University of Southern California, Los Angeles, and his colleagues reported these results in Oncogene.
The researchers first tested lenalidomide and pomalidomide in 9 PEL cell lines. Both drugs exhibited antiproliferative effects, at clinically achievable doses, in 6 of the cell lines (BC-3, BCBL-1, JSC-1, VG-1, UMPEL-1, and UMPEL-3).
The IMiDs displayed activity in the remaining 3 cells lines (BC-1, BCP-1, and APK-1) as well, but only at high doses (ranging from 2.6 μM to 10 μM for lenalidomide and from 226 nM to 744 nM for pomalidomide).
Looking into the mechanism of IMiDs in PEL, the researchers found these drugs induce G1 cell-cycle arrest without inducing Kaposi’s sarcoma-associated herpes virus lytic cycle reactivation.
Further investigation revealed that interferon α, β, and γ are cytotoxic to PEL, but they are not essential for the antiproliferative effects of IMiDs. On the other hand, cereblon-dependent suppression of IRF4 and rapid degradation of IKZF1 are essential.
The researchers also found that knocking down MYC enhanced IMiD activity. This led them to BRD4 inhibitors, which are known to target MYC transcription.
The team tested lenalidomide in combination with JQ-1, IBET151, and PFI-1. The IMiD was “highly synergistic” with all 3 BRD4 inhibitors in BC-3 and BCBL-1 cell lines.
In both cell lines, treatment with lenalidomide and JQ-1 significantly decreased the expression of MYC and IRF4 at protein and mRNA levels. The treatment also resulted in G1 cell cycle arrest, cleavage of poly (ADP-ribose) polymerase, and apoptosis.
So the researchers tested this combination in a mouse model of PEL (NOD-SCID mice injected with BC-3 cells). And they observed a significant improvement in median survival for mice that received combination treatment, compared to lenalidomide or JQ-1 alone—51 days, 35 days, and 42 days, respectively (P≤0.01).![]()

reviewing an image
Photo from the University
of Southern California
Immunomodulatory drugs (IMiDs) and BRD4 inhibitors hold promise for treating primary effusion lymphoma (PEL), according to preclinical research.
The IMiDs lenalidomide and pomalidomide had antiproliferative effects in PEL cell lines, and lenalidomide exhibited synergistic cytotoxicity with the BRD4 inhibitors JQ-1, IBET151, and PFI-1.
In mouse models of PEL, treatment with lenalidomide and JQ-1 significantly prolonged survival.
Preet Chaudhary, MD, PhD, of the University of Southern California, Los Angeles, and his colleagues reported these results in Oncogene.
The researchers first tested lenalidomide and pomalidomide in 9 PEL cell lines. Both drugs exhibited antiproliferative effects, at clinically achievable doses, in 6 of the cell lines (BC-3, BCBL-1, JSC-1, VG-1, UMPEL-1, and UMPEL-3).
The IMiDs displayed activity in the remaining 3 cells lines (BC-1, BCP-1, and APK-1) as well, but only at high doses (ranging from 2.6 μM to 10 μM for lenalidomide and from 226 nM to 744 nM for pomalidomide).
Looking into the mechanism of IMiDs in PEL, the researchers found these drugs induce G1 cell-cycle arrest without inducing Kaposi’s sarcoma-associated herpes virus lytic cycle reactivation.
Further investigation revealed that interferon α, β, and γ are cytotoxic to PEL, but they are not essential for the antiproliferative effects of IMiDs. On the other hand, cereblon-dependent suppression of IRF4 and rapid degradation of IKZF1 are essential.
The researchers also found that knocking down MYC enhanced IMiD activity. This led them to BRD4 inhibitors, which are known to target MYC transcription.
The team tested lenalidomide in combination with JQ-1, IBET151, and PFI-1. The IMiD was “highly synergistic” with all 3 BRD4 inhibitors in BC-3 and BCBL-1 cell lines.
In both cell lines, treatment with lenalidomide and JQ-1 significantly decreased the expression of MYC and IRF4 at protein and mRNA levels. The treatment also resulted in G1 cell cycle arrest, cleavage of poly (ADP-ribose) polymerase, and apoptosis.
So the researchers tested this combination in a mouse model of PEL (NOD-SCID mice injected with BC-3 cells). And they observed a significant improvement in median survival for mice that received combination treatment, compared to lenalidomide or JQ-1 alone—51 days, 35 days, and 42 days, respectively (P≤0.01).![]()
Model helps explain chaotic angiogenesis
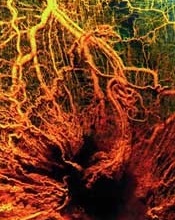
Image by Louis Heiser
and Robert Ackland
Scientists say they have developed a model that provides new insight into chaotic angiogenesis, a hallmark of cancer.
The model shows how proteins involved in angiogenesis communicate with each other and how tumors take charge of the protein signaling chain that controls blood vessel growth.
The work, published in PNAS, suggests that Jagged ligands play a major role in the chaotic vessel growth observed around tumors.
In normal growth, an endothelial cell sprouts from an existing blood vessel as a tip. Others that follow the tip cell become the stalk cells that ultimately form vessel walls. The Notch signaling pathway directs the endothelial cell’s decision to become a tip or stalk.
Notch receptors bind with Delta ligand or Jagged ligand molecules produced by cells. How they interact determines the cell’s fate.
When Notch and Delta bind, they prompt a few cells to be tips and adjacent ones to be stalks. The Notch-Delta pathway has been studied extensively and is the target of many angiogenesis inhibitors now in use, according to the scientists.
“We wondered exactly what Notch-Jagged signaling does that is not done in Notch-Delta signaling,” said study author Marcelo Boareto, PhD, of the Swiss Federal Institute of Technology in Zurich.
“We find that when the cells communicate mostly via Jagged, we see a new kind of cell that is not exactly tip and not exactly stalk, but somewhere in between. This compromised cell is the major difference between normal and tumor angiogenesis and suggests that if Notch-Jagged signaling can be somehow suppressed without affecting Notch-Delta, we can probably disrupt tumor angiogenesis.”
The scientists found the tip/stalk hybrid cells do form new blood vessels, but these vessels rarely mature.
“High levels of Jagged in the environment can trigger the formation of blood vessels that are useful to the tumor: fast-developing, leaky, and spread chaotically all over the tumor mass,” Dr Boareto said.
“Tumors don’t have to wait for the vessels to develop,” added study author José Onuchic, PhD, of Rice University in Houston, Texas. “They take advantage of the leakiness of the structure.”
The scientists’ model also takes into account the effect of vascular endothelial growth factor, which triggers angiogenesis and is overexpressed by tumor cells.
“It is very interesting how the tumor hijacks this important mechanism, which is needed for the development of a functional vessel, and amplifies it to generate pathological angiogenesis that leads to uncontrolled growth,” Dr Onuchic said. ![]()

Image by Louis Heiser
and Robert Ackland
Scientists say they have developed a model that provides new insight into chaotic angiogenesis, a hallmark of cancer.
The model shows how proteins involved in angiogenesis communicate with each other and how tumors take charge of the protein signaling chain that controls blood vessel growth.
The work, published in PNAS, suggests that Jagged ligands play a major role in the chaotic vessel growth observed around tumors.
In normal growth, an endothelial cell sprouts from an existing blood vessel as a tip. Others that follow the tip cell become the stalk cells that ultimately form vessel walls. The Notch signaling pathway directs the endothelial cell’s decision to become a tip or stalk.
Notch receptors bind with Delta ligand or Jagged ligand molecules produced by cells. How they interact determines the cell’s fate.
When Notch and Delta bind, they prompt a few cells to be tips and adjacent ones to be stalks. The Notch-Delta pathway has been studied extensively and is the target of many angiogenesis inhibitors now in use, according to the scientists.
“We wondered exactly what Notch-Jagged signaling does that is not done in Notch-Delta signaling,” said study author Marcelo Boareto, PhD, of the Swiss Federal Institute of Technology in Zurich.
“We find that when the cells communicate mostly via Jagged, we see a new kind of cell that is not exactly tip and not exactly stalk, but somewhere in between. This compromised cell is the major difference between normal and tumor angiogenesis and suggests that if Notch-Jagged signaling can be somehow suppressed without affecting Notch-Delta, we can probably disrupt tumor angiogenesis.”
The scientists found the tip/stalk hybrid cells do form new blood vessels, but these vessels rarely mature.
“High levels of Jagged in the environment can trigger the formation of blood vessels that are useful to the tumor: fast-developing, leaky, and spread chaotically all over the tumor mass,” Dr Boareto said.
“Tumors don’t have to wait for the vessels to develop,” added study author José Onuchic, PhD, of Rice University in Houston, Texas. “They take advantage of the leakiness of the structure.”
The scientists’ model also takes into account the effect of vascular endothelial growth factor, which triggers angiogenesis and is overexpressed by tumor cells.
“It is very interesting how the tumor hijacks this important mechanism, which is needed for the development of a functional vessel, and amplifies it to generate pathological angiogenesis that leads to uncontrolled growth,” Dr Onuchic said. ![]()

Image by Louis Heiser
and Robert Ackland
Scientists say they have developed a model that provides new insight into chaotic angiogenesis, a hallmark of cancer.
The model shows how proteins involved in angiogenesis communicate with each other and how tumors take charge of the protein signaling chain that controls blood vessel growth.
The work, published in PNAS, suggests that Jagged ligands play a major role in the chaotic vessel growth observed around tumors.
In normal growth, an endothelial cell sprouts from an existing blood vessel as a tip. Others that follow the tip cell become the stalk cells that ultimately form vessel walls. The Notch signaling pathway directs the endothelial cell’s decision to become a tip or stalk.
Notch receptors bind with Delta ligand or Jagged ligand molecules produced by cells. How they interact determines the cell’s fate.
When Notch and Delta bind, they prompt a few cells to be tips and adjacent ones to be stalks. The Notch-Delta pathway has been studied extensively and is the target of many angiogenesis inhibitors now in use, according to the scientists.
“We wondered exactly what Notch-Jagged signaling does that is not done in Notch-Delta signaling,” said study author Marcelo Boareto, PhD, of the Swiss Federal Institute of Technology in Zurich.
“We find that when the cells communicate mostly via Jagged, we see a new kind of cell that is not exactly tip and not exactly stalk, but somewhere in between. This compromised cell is the major difference between normal and tumor angiogenesis and suggests that if Notch-Jagged signaling can be somehow suppressed without affecting Notch-Delta, we can probably disrupt tumor angiogenesis.”
The scientists found the tip/stalk hybrid cells do form new blood vessels, but these vessels rarely mature.
“High levels of Jagged in the environment can trigger the formation of blood vessels that are useful to the tumor: fast-developing, leaky, and spread chaotically all over the tumor mass,” Dr Boareto said.
“Tumors don’t have to wait for the vessels to develop,” added study author José Onuchic, PhD, of Rice University in Houston, Texas. “They take advantage of the leakiness of the structure.”
The scientists’ model also takes into account the effect of vascular endothelial growth factor, which triggers angiogenesis and is overexpressed by tumor cells.
“It is very interesting how the tumor hijacks this important mechanism, which is needed for the development of a functional vessel, and amplifies it to generate pathological angiogenesis that leads to uncontrolled growth,” Dr Onuchic said. ![]()
Sepsis readmissions common and costly, group says

Photo courtesy of the CDC
Hospital readmissions for sepsis are under-recognized, and additional measures are needed to prevent these readmissions, researchers say.
The group conducted a study of California hospitals that showed sepsis accounts for roughly the same percentage of readmissions as heart attacks and congestive heart failure.
However, sepsis readmissions cost the healthcare system more than heart attack and congestive heart failure readmissions combined.
The research was published in Critical Care Medicine.
“Our study shows how common sepsis readmissions are and [elucidates] some of the factors that are associated with higher risk of readmission after these severe infections,” said study author Dong Chang, MD, of Harbor-UCLA Medical Center in Los Angeles, California.
“In addition, we show that sepsis readmissions have a significant impact on healthcare expenditures relative to other high-risk conditions that are receiving active attention and interventions. Based on these results, we believe that sepsis readmissions are under-recognized and should be among the conditions that are targeted for intervention by policymakers.”
The researchers analyzed data on sepsis admissions for adults 18 years of age and older at all California hospitals from 2009 through 2011. The team also analyzed data for congestive heart failure and heart attack admissions during the same period.
There were 240,198 admissions for sepsis, 193,153 for congestive heart failure, and 105,684 for heart attacks. The all-cause, 30-day readmission rate was 20.4% for sepsis, 23.6% for congestive heart failure, and 17.7% for heart attacks.
Patients with sepsis were readmitted because of respiratory failure; pneumonia; urinary tract infections; renal infections; renal failure; intestinal infections; complications with devices, implants, or grafts; and other causes.
Sepsis readmission rates were higher among young adults than older adults, among men than women, among black and Native American patients than other racial groups, and among lower-income patients than those with higher incomes. In addition, patients with other concurrent health problems were more likely to be readmitted than those with sepsis alone.
The estimated annual cost of sepsis-related readmissions in California during the study period was $500 million, compared with $229 million for congestive heart failure and $142 million for heart attacks.
“These findings suggest that efforts to reduce hospital readmissions need to include sepsis prominently, at least on par with heart failure and myocardial infarction,” said study author Martin Shapiro, MD, PhD, of the David Geffen School of Medicine at UCLA.
Next, the researchers plan to examine why patients are readmitted after sepsis and the percentage of those readmissions that are due to processes that can be improved upon, such as discharge practices, follow-up care, and teaching patients how to take their medications. ![]()

Photo courtesy of the CDC
Hospital readmissions for sepsis are under-recognized, and additional measures are needed to prevent these readmissions, researchers say.
The group conducted a study of California hospitals that showed sepsis accounts for roughly the same percentage of readmissions as heart attacks and congestive heart failure.
However, sepsis readmissions cost the healthcare system more than heart attack and congestive heart failure readmissions combined.
The research was published in Critical Care Medicine.
“Our study shows how common sepsis readmissions are and [elucidates] some of the factors that are associated with higher risk of readmission after these severe infections,” said study author Dong Chang, MD, of Harbor-UCLA Medical Center in Los Angeles, California.
“In addition, we show that sepsis readmissions have a significant impact on healthcare expenditures relative to other high-risk conditions that are receiving active attention and interventions. Based on these results, we believe that sepsis readmissions are under-recognized and should be among the conditions that are targeted for intervention by policymakers.”
The researchers analyzed data on sepsis admissions for adults 18 years of age and older at all California hospitals from 2009 through 2011. The team also analyzed data for congestive heart failure and heart attack admissions during the same period.
There were 240,198 admissions for sepsis, 193,153 for congestive heart failure, and 105,684 for heart attacks. The all-cause, 30-day readmission rate was 20.4% for sepsis, 23.6% for congestive heart failure, and 17.7% for heart attacks.
Patients with sepsis were readmitted because of respiratory failure; pneumonia; urinary tract infections; renal infections; renal failure; intestinal infections; complications with devices, implants, or grafts; and other causes.
Sepsis readmission rates were higher among young adults than older adults, among men than women, among black and Native American patients than other racial groups, and among lower-income patients than those with higher incomes. In addition, patients with other concurrent health problems were more likely to be readmitted than those with sepsis alone.
The estimated annual cost of sepsis-related readmissions in California during the study period was $500 million, compared with $229 million for congestive heart failure and $142 million for heart attacks.
“These findings suggest that efforts to reduce hospital readmissions need to include sepsis prominently, at least on par with heart failure and myocardial infarction,” said study author Martin Shapiro, MD, PhD, of the David Geffen School of Medicine at UCLA.
Next, the researchers plan to examine why patients are readmitted after sepsis and the percentage of those readmissions that are due to processes that can be improved upon, such as discharge practices, follow-up care, and teaching patients how to take their medications. ![]()

Photo courtesy of the CDC
Hospital readmissions for sepsis are under-recognized, and additional measures are needed to prevent these readmissions, researchers say.
The group conducted a study of California hospitals that showed sepsis accounts for roughly the same percentage of readmissions as heart attacks and congestive heart failure.
However, sepsis readmissions cost the healthcare system more than heart attack and congestive heart failure readmissions combined.
The research was published in Critical Care Medicine.
“Our study shows how common sepsis readmissions are and [elucidates] some of the factors that are associated with higher risk of readmission after these severe infections,” said study author Dong Chang, MD, of Harbor-UCLA Medical Center in Los Angeles, California.
“In addition, we show that sepsis readmissions have a significant impact on healthcare expenditures relative to other high-risk conditions that are receiving active attention and interventions. Based on these results, we believe that sepsis readmissions are under-recognized and should be among the conditions that are targeted for intervention by policymakers.”
The researchers analyzed data on sepsis admissions for adults 18 years of age and older at all California hospitals from 2009 through 2011. The team also analyzed data for congestive heart failure and heart attack admissions during the same period.
There were 240,198 admissions for sepsis, 193,153 for congestive heart failure, and 105,684 for heart attacks. The all-cause, 30-day readmission rate was 20.4% for sepsis, 23.6% for congestive heart failure, and 17.7% for heart attacks.
Patients with sepsis were readmitted because of respiratory failure; pneumonia; urinary tract infections; renal infections; renal failure; intestinal infections; complications with devices, implants, or grafts; and other causes.
Sepsis readmission rates were higher among young adults than older adults, among men than women, among black and Native American patients than other racial groups, and among lower-income patients than those with higher incomes. In addition, patients with other concurrent health problems were more likely to be readmitted than those with sepsis alone.
The estimated annual cost of sepsis-related readmissions in California during the study period was $500 million, compared with $229 million for congestive heart failure and $142 million for heart attacks.
“These findings suggest that efforts to reduce hospital readmissions need to include sepsis prominently, at least on par with heart failure and myocardial infarction,” said study author Martin Shapiro, MD, PhD, of the David Geffen School of Medicine at UCLA.
Next, the researchers plan to examine why patients are readmitted after sepsis and the percentage of those readmissions that are due to processes that can be improved upon, such as discharge practices, follow-up care, and teaching patients how to take their medications. ![]()
Early progression predicts overall survival in FL

Photo by Rhoda Baer
The goal for many cancer patients is to reach the 5-year mark without progression, but a new study suggests 2 years might be a more appropriate goal for patients with follicular lymphoma (FL).
Previous research indicated that about 20% of FL patients relapse within 2 years of treatment.
Now, researchers have found these patients have a significantly worse 5-year survival rate than patients who make it past the 2-year mark without progressing.
Carla Casulo, MD, of the University of Rochester in New York, and her colleagues recounted these findings in the Journal of Clinical Oncology.
The team analyzed data from 588 patients with stage 2-4 FL who received first-line therapy with rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP).
These patients could be separated into 2 groups: those with disease progression within 2 years of diagnosis (19%, n=110) and those who did not progress within the 2-year period (71%, n=420).
Eight percent of patients (n=46) were lost to follow-up, and 2% (n=12) died of causes other than progression within the 2-year period.
At 2 years, the overall survival (OS) rate was 68% in the early progression group and 97% in the group that did not progress. At 5 years, OS rates were 50% and 90%, respectively.
In unadjusted Cox regression analysis, early progression was associated with lower OS (hazard ratio[HR]=7.17). The same was true after the researchers adjusted for FLIPI score (HR=6.44).
The team observed similar results in a validation cohort of 147 FL patients who received first-line R-CHOP. At 2 years, the OS rate was 64% in the early progression group and 98% in the group that did not progress. At 5 years, OS rates were 34% and 94%, respectively.
Again, in an unadjusted analysis, early progression was associated with lower OS (HR=20.0). And this trend was maintained after the researchers adjusted for FLIPI score (HR=19.8).
The researchers also found that, for patients in the early progression group, clinical factors that were predictive of inferior OS were age, ECOG performance score, nodal sites, and disease stage. For the group that did not progress within 2 years, clinical factors that were predictive of OS were age and extranodal sites.
In a Cox regression analysis that encompassed these factors and early progression, only early progression, age, and ECOG performance scores remained significantly predictive of OS.
“[W]e have confirmed that all relapsed patients are not equal and therefore should not be approached the same at diagnosis, nor at the time of relapse, in terms of therapies,” Dr Casulo said.
“It will be critical to predict who is most likely to relapse early. We believe that targeted sequencing or gene-expression profiling will be important to understanding how to improve the outcomes of this group.” ![]()

Photo by Rhoda Baer
The goal for many cancer patients is to reach the 5-year mark without progression, but a new study suggests 2 years might be a more appropriate goal for patients with follicular lymphoma (FL).
Previous research indicated that about 20% of FL patients relapse within 2 years of treatment.
Now, researchers have found these patients have a significantly worse 5-year survival rate than patients who make it past the 2-year mark without progressing.
Carla Casulo, MD, of the University of Rochester in New York, and her colleagues recounted these findings in the Journal of Clinical Oncology.
The team analyzed data from 588 patients with stage 2-4 FL who received first-line therapy with rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP).
These patients could be separated into 2 groups: those with disease progression within 2 years of diagnosis (19%, n=110) and those who did not progress within the 2-year period (71%, n=420).
Eight percent of patients (n=46) were lost to follow-up, and 2% (n=12) died of causes other than progression within the 2-year period.
At 2 years, the overall survival (OS) rate was 68% in the early progression group and 97% in the group that did not progress. At 5 years, OS rates were 50% and 90%, respectively.
In unadjusted Cox regression analysis, early progression was associated with lower OS (hazard ratio[HR]=7.17). The same was true after the researchers adjusted for FLIPI score (HR=6.44).
The team observed similar results in a validation cohort of 147 FL patients who received first-line R-CHOP. At 2 years, the OS rate was 64% in the early progression group and 98% in the group that did not progress. At 5 years, OS rates were 34% and 94%, respectively.
Again, in an unadjusted analysis, early progression was associated with lower OS (HR=20.0). And this trend was maintained after the researchers adjusted for FLIPI score (HR=19.8).
The researchers also found that, for patients in the early progression group, clinical factors that were predictive of inferior OS were age, ECOG performance score, nodal sites, and disease stage. For the group that did not progress within 2 years, clinical factors that were predictive of OS were age and extranodal sites.
In a Cox regression analysis that encompassed these factors and early progression, only early progression, age, and ECOG performance scores remained significantly predictive of OS.
“[W]e have confirmed that all relapsed patients are not equal and therefore should not be approached the same at diagnosis, nor at the time of relapse, in terms of therapies,” Dr Casulo said.
“It will be critical to predict who is most likely to relapse early. We believe that targeted sequencing or gene-expression profiling will be important to understanding how to improve the outcomes of this group.” ![]()

Photo by Rhoda Baer
The goal for many cancer patients is to reach the 5-year mark without progression, but a new study suggests 2 years might be a more appropriate goal for patients with follicular lymphoma (FL).
Previous research indicated that about 20% of FL patients relapse within 2 years of treatment.
Now, researchers have found these patients have a significantly worse 5-year survival rate than patients who make it past the 2-year mark without progressing.
Carla Casulo, MD, of the University of Rochester in New York, and her colleagues recounted these findings in the Journal of Clinical Oncology.
The team analyzed data from 588 patients with stage 2-4 FL who received first-line therapy with rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP).
These patients could be separated into 2 groups: those with disease progression within 2 years of diagnosis (19%, n=110) and those who did not progress within the 2-year period (71%, n=420).
Eight percent of patients (n=46) were lost to follow-up, and 2% (n=12) died of causes other than progression within the 2-year period.
At 2 years, the overall survival (OS) rate was 68% in the early progression group and 97% in the group that did not progress. At 5 years, OS rates were 50% and 90%, respectively.
In unadjusted Cox regression analysis, early progression was associated with lower OS (hazard ratio[HR]=7.17). The same was true after the researchers adjusted for FLIPI score (HR=6.44).
The team observed similar results in a validation cohort of 147 FL patients who received first-line R-CHOP. At 2 years, the OS rate was 64% in the early progression group and 98% in the group that did not progress. At 5 years, OS rates were 34% and 94%, respectively.
Again, in an unadjusted analysis, early progression was associated with lower OS (HR=20.0). And this trend was maintained after the researchers adjusted for FLIPI score (HR=19.8).
The researchers also found that, for patients in the early progression group, clinical factors that were predictive of inferior OS were age, ECOG performance score, nodal sites, and disease stage. For the group that did not progress within 2 years, clinical factors that were predictive of OS were age and extranodal sites.
In a Cox regression analysis that encompassed these factors and early progression, only early progression, age, and ECOG performance scores remained significantly predictive of OS.
“[W]e have confirmed that all relapsed patients are not equal and therefore should not be approached the same at diagnosis, nor at the time of relapse, in terms of therapies,” Dr Casulo said.
“It will be critical to predict who is most likely to relapse early. We believe that targeted sequencing or gene-expression profiling will be important to understanding how to improve the outcomes of this group.” ![]()
Benefit of extended anticoagulation is temporary, study shows

Results of the PADIS-PE trial suggest that patients with a first episode of pulmonary embolism (PE) benefit from extended anticoagulation therapy, but that benefit is lost after the treatment is stopped.
When patients received warfarin for 18 months after an initial 6 months of anticoagulation therapy, they had a lower risk of venous thromboembolism (VTE)
recurrence than patients who did not receive additional anticoagulation.
However, this benefit was lost once the patients stopped taking warfarin.
Francis Couturaud, MD, PhD, of the Universite de Bretagne Occidentale in Brest, France, and his colleagues reported these findings in JAMA.
The researchers analyzed 371 adult patients who had experienced a first episode of symptomatic, unprovoked PE and were initially treated for 6 months with a vitamin K antagonist.
The patients were then randomized to warfarin or placebo for 18 months. After randomization, 4 patients were lost to follow-up, all after month 18, and 1 withdrew due to an adverse event.
The median follow-up was 24 months. The primary outcome was a composite of recurrent VTE and major bleeding at 18 months after randomization.
During the 18-month treatment period, the primary outcome occurred in 3% (6/184) of patients in the warfarin arm and 13.5% (25/187) in the placebo arm (hazard ratio [HR]=0.22, P=0.001).
This was driven by a reduction in the risk of recurrent VTE, as the risk of major bleeding was higher in the warfarin arm. Recurrent VTE occurred in 3 patients in the warfarin arm and 25 in the placebo arm (HR=0.15, P<0.001). And major bleeding occurred in 4 patients in the warfarin arm and 1 in the placebo arm (HR=3.96, P=0.22).
Once patients stopped receiving warfarin, the reduction in risk of VTE recurrence was lost. During the entire study period (a median of 41 months), the primary outcome occurred in 21% (n=33) of patients in the warfarin arm and 24% (n=42) in the placebo arm (HR=0.75, P=0.22).
There was no significant difference between the treatment arms with regard to recurrent VTE (HR=0.69, P=0.14) or major bleeding (HR=1.12, P=0.85).
The researchers said these results suggest patients with an unprovoked PE require long-term VTE prevention measures. But further investigation is needed to determine whether these should include systematic treatment with vitamin K antagonists, new anticoagulants, or aspirin, or whether the measures should be tailored according to patient risk factors. ![]()

Results of the PADIS-PE trial suggest that patients with a first episode of pulmonary embolism (PE) benefit from extended anticoagulation therapy, but that benefit is lost after the treatment is stopped.
When patients received warfarin for 18 months after an initial 6 months of anticoagulation therapy, they had a lower risk of venous thromboembolism (VTE)
recurrence than patients who did not receive additional anticoagulation.
However, this benefit was lost once the patients stopped taking warfarin.
Francis Couturaud, MD, PhD, of the Universite de Bretagne Occidentale in Brest, France, and his colleagues reported these findings in JAMA.
The researchers analyzed 371 adult patients who had experienced a first episode of symptomatic, unprovoked PE and were initially treated for 6 months with a vitamin K antagonist.
The patients were then randomized to warfarin or placebo for 18 months. After randomization, 4 patients were lost to follow-up, all after month 18, and 1 withdrew due to an adverse event.
The median follow-up was 24 months. The primary outcome was a composite of recurrent VTE and major bleeding at 18 months after randomization.
During the 18-month treatment period, the primary outcome occurred in 3% (6/184) of patients in the warfarin arm and 13.5% (25/187) in the placebo arm (hazard ratio [HR]=0.22, P=0.001).
This was driven by a reduction in the risk of recurrent VTE, as the risk of major bleeding was higher in the warfarin arm. Recurrent VTE occurred in 3 patients in the warfarin arm and 25 in the placebo arm (HR=0.15, P<0.001). And major bleeding occurred in 4 patients in the warfarin arm and 1 in the placebo arm (HR=3.96, P=0.22).
Once patients stopped receiving warfarin, the reduction in risk of VTE recurrence was lost. During the entire study period (a median of 41 months), the primary outcome occurred in 21% (n=33) of patients in the warfarin arm and 24% (n=42) in the placebo arm (HR=0.75, P=0.22).
There was no significant difference between the treatment arms with regard to recurrent VTE (HR=0.69, P=0.14) or major bleeding (HR=1.12, P=0.85).
The researchers said these results suggest patients with an unprovoked PE require long-term VTE prevention measures. But further investigation is needed to determine whether these should include systematic treatment with vitamin K antagonists, new anticoagulants, or aspirin, or whether the measures should be tailored according to patient risk factors. ![]()

Results of the PADIS-PE trial suggest that patients with a first episode of pulmonary embolism (PE) benefit from extended anticoagulation therapy, but that benefit is lost after the treatment is stopped.
When patients received warfarin for 18 months after an initial 6 months of anticoagulation therapy, they had a lower risk of venous thromboembolism (VTE)
recurrence than patients who did not receive additional anticoagulation.
However, this benefit was lost once the patients stopped taking warfarin.
Francis Couturaud, MD, PhD, of the Universite de Bretagne Occidentale in Brest, France, and his colleagues reported these findings in JAMA.
The researchers analyzed 371 adult patients who had experienced a first episode of symptomatic, unprovoked PE and were initially treated for 6 months with a vitamin K antagonist.
The patients were then randomized to warfarin or placebo for 18 months. After randomization, 4 patients were lost to follow-up, all after month 18, and 1 withdrew due to an adverse event.
The median follow-up was 24 months. The primary outcome was a composite of recurrent VTE and major bleeding at 18 months after randomization.
During the 18-month treatment period, the primary outcome occurred in 3% (6/184) of patients in the warfarin arm and 13.5% (25/187) in the placebo arm (hazard ratio [HR]=0.22, P=0.001).
This was driven by a reduction in the risk of recurrent VTE, as the risk of major bleeding was higher in the warfarin arm. Recurrent VTE occurred in 3 patients in the warfarin arm and 25 in the placebo arm (HR=0.15, P<0.001). And major bleeding occurred in 4 patients in the warfarin arm and 1 in the placebo arm (HR=3.96, P=0.22).
Once patients stopped receiving warfarin, the reduction in risk of VTE recurrence was lost. During the entire study period (a median of 41 months), the primary outcome occurred in 21% (n=33) of patients in the warfarin arm and 24% (n=42) in the placebo arm (HR=0.75, P=0.22).
There was no significant difference between the treatment arms with regard to recurrent VTE (HR=0.69, P=0.14) or major bleeding (HR=1.12, P=0.85).
The researchers said these results suggest patients with an unprovoked PE require long-term VTE prevention measures. But further investigation is needed to determine whether these should include systematic treatment with vitamin K antagonists, new anticoagulants, or aspirin, or whether the measures should be tailored according to patient risk factors.
NICE wants more info on PI3Kδ inhibitor

The UK’s National Institute for Health and Care Excellence (NICE) has opened a consultation on a preliminary draft guidance for the PI3Kδ inhibitor idelalisib (Zydelig).
The agency has requested additional information from Gilead Sciences, the company developing idelalisib, to inform a decision on the use of this drug
in combination with rituximab for adults with chronic lymphocytic
leukemia (CLL).
NICE said it has questions about the cost-effectiveness of this treatment that have not been answered. Idelalisib costs £3114.75 for sixty 150-mg tablets, and the mean cost of a 1-year treatment course is £37,922.
Until NICE receives the requested information, the agency said it cannot recommend idelalisib plus rituximab for adults with untreated CLL who have 17p deletion or TP53 mutation, adults with relapsed CLL, or adults whose CLL is refractory and retreatment with previous regimens is not considered appropriate.
“The independent appraisal committee, which is developing the guidance on behalf of NICE, considered evidence from the company, clinical experts, and patient representatives,” said Carole Longson, NICE Health Technology Evaluation Centre Director.
“It concluded there were still questions to be answered about the cost-effectiveness of the treatment. We have requested further analysis from the company. We want to ensure we have as much information as possible to make an informed recommendation.”
Specifically, NICE has requested:
- A revised cost-effectiveness analysis for the comparison of idelalisib plus rituximab with rituximab alone, best supportive care, and ofatumumab
- A sensitivity analysis exploring the length of treatment benefit of idelalisib plus rituximab from treatment discontinuation up to 5 years
- A sensitivity analysis exploring the effects of reducing the proportion of non-responders having intravenous immunoglobulin from 45% to 20% or less and increasing the number of responders having intravenous immunoglobulin from 0% to 20%
- A sensitivity analysis exploring the effect of using clinical effectiveness data from the subgroup of people in Study 116 whose disease is refractory.
The independent appraisal committee will review this information and develop further draft guidance.
Until final guidance is issued, National Health Service bodies should make decisions locally on the funding of specific treatments. Once NICE issues its final guidance on a technology, it replaces local recommendations.

The UK’s National Institute for Health and Care Excellence (NICE) has opened a consultation on a preliminary draft guidance for the PI3Kδ inhibitor idelalisib (Zydelig).
The agency has requested additional information from Gilead Sciences, the company developing idelalisib, to inform a decision on the use of this drug
in combination with rituximab for adults with chronic lymphocytic
leukemia (CLL).
NICE said it has questions about the cost-effectiveness of this treatment that have not been answered. Idelalisib costs £3114.75 for sixty 150-mg tablets, and the mean cost of a 1-year treatment course is £37,922.
Until NICE receives the requested information, the agency said it cannot recommend idelalisib plus rituximab for adults with untreated CLL who have 17p deletion or TP53 mutation, adults with relapsed CLL, or adults whose CLL is refractory and retreatment with previous regimens is not considered appropriate.
“The independent appraisal committee, which is developing the guidance on behalf of NICE, considered evidence from the company, clinical experts, and patient representatives,” said Carole Longson, NICE Health Technology Evaluation Centre Director.
“It concluded there were still questions to be answered about the cost-effectiveness of the treatment. We have requested further analysis from the company. We want to ensure we have as much information as possible to make an informed recommendation.”
Specifically, NICE has requested:
- A revised cost-effectiveness analysis for the comparison of idelalisib plus rituximab with rituximab alone, best supportive care, and ofatumumab
- A sensitivity analysis exploring the length of treatment benefit of idelalisib plus rituximab from treatment discontinuation up to 5 years
- A sensitivity analysis exploring the effects of reducing the proportion of non-responders having intravenous immunoglobulin from 45% to 20% or less and increasing the number of responders having intravenous immunoglobulin from 0% to 20%
- A sensitivity analysis exploring the effect of using clinical effectiveness data from the subgroup of people in Study 116 whose disease is refractory.
The independent appraisal committee will review this information and develop further draft guidance.
Until final guidance is issued, National Health Service bodies should make decisions locally on the funding of specific treatments. Once NICE issues its final guidance on a technology, it replaces local recommendations.

The UK’s National Institute for Health and Care Excellence (NICE) has opened a consultation on a preliminary draft guidance for the PI3Kδ inhibitor idelalisib (Zydelig).
The agency has requested additional information from Gilead Sciences, the company developing idelalisib, to inform a decision on the use of this drug
in combination with rituximab for adults with chronic lymphocytic
leukemia (CLL).
NICE said it has questions about the cost-effectiveness of this treatment that have not been answered. Idelalisib costs £3114.75 for sixty 150-mg tablets, and the mean cost of a 1-year treatment course is £37,922.
Until NICE receives the requested information, the agency said it cannot recommend idelalisib plus rituximab for adults with untreated CLL who have 17p deletion or TP53 mutation, adults with relapsed CLL, or adults whose CLL is refractory and retreatment with previous regimens is not considered appropriate.
“The independent appraisal committee, which is developing the guidance on behalf of NICE, considered evidence from the company, clinical experts, and patient representatives,” said Carole Longson, NICE Health Technology Evaluation Centre Director.
“It concluded there were still questions to be answered about the cost-effectiveness of the treatment. We have requested further analysis from the company. We want to ensure we have as much information as possible to make an informed recommendation.”
Specifically, NICE has requested:
- A revised cost-effectiveness analysis for the comparison of idelalisib plus rituximab with rituximab alone, best supportive care, and ofatumumab
- A sensitivity analysis exploring the length of treatment benefit of idelalisib plus rituximab from treatment discontinuation up to 5 years
- A sensitivity analysis exploring the effects of reducing the proportion of non-responders having intravenous immunoglobulin from 45% to 20% or less and increasing the number of responders having intravenous immunoglobulin from 0% to 20%
- A sensitivity analysis exploring the effect of using clinical effectiveness data from the subgroup of people in Study 116 whose disease is refractory.
The independent appraisal committee will review this information and develop further draft guidance.
Until final guidance is issued, National Health Service bodies should make decisions locally on the funding of specific treatments. Once NICE issues its final guidance on a technology, it replaces local recommendations.
Drug may fight cancer by suppressing inflammation

Intermittent dosing with the immunosuppressant rapamycin can break an inflammatory loop that fuels cancer relapse, according to researchers.
The team found that rapamycin selectively breaks the cascade of inflammatory events that follow cellular senescence.
Once disrupted, it takes time for the inflammatory loop to re-establish itself. This suggests intermittent dosing could provide a way to reap the benefits of rapamycin while reducing side effects.
The researchers described these findings in Nature Cell Biology.
“We think this could provide a paradigm shift in the treatment of age-related disease, including cancer,” said study author Judith Campisi, PhD, of the Buck Institute for Research on Aging in Novato, California.
“Imagine the possibility of taking a pill for a few days or weeks every few years, as opposed to taking something with side effects every day for the rest of your life. It’s a new way of looking at how we could deal with age-related maladies.”
For this study, Dr Campisi and her colleagues administered rapamycin to mice with prostate cancer after they had received treatment with DNA-damaging chemotherapy.
The rapamycin reduced the secretion of inflammatory cytokines from senescent cells by suppressing the mTOR pathway. This suppressed the cells’ ability to stimulate tumor growth.
“DNA-damaging chemotherapy causes senescence, both to the tumor and its microenvironment,” Dr Campisi explained. “The tumor shrinks, but the immediate tissue environment is inflamed. We think signals from those inflamed cells trigger residual cancer cells to grow again. In the mice, rapamycin suppressed the ability of the tumor cells to relapse.”
The researchers said the paradigm-shifting potential of intermittent rapamycin dosing is based on the fact that it takes time for the inflammatory loop—fueled by the senescence-associated secretory phenotype (SASP)—to form and time for it to re-establish itself after a brief treatment period with rapamycin.
“Rapamycin blocks the production of a protein called IL-1α,” explained study author Remi-Martin Laberge, PhD, also of the Buck Institute.
“This in turn, suppresses IL6, a well-known inflammatory cytokine, at the level of transcription, which prevents the production of the IL6 protein. Because it acts at a deeper level within the cellular process, it takes longer for it to get started again.”
Dr Laberge also pointed out that treatment with rapamycin selectivity impacts the SASP, preserving the function of factors essential for wound healing.
“It’s an elegant solution,” Dr Laberge said. “Imagine using a small hammer to delicately knock out one thing that is causing problems. We knocked it out, and it stayed out long enough to benefit the health of the animal.”
“We have yet to fully understand why suppressing the mTOR pathway via rapamycin increases lifespan and health span in mice,” Dr Campisi added. “This work helps illuminate the puzzle. Perhaps the mice are living longer because they have less overall inflammation, and maybe intermittent dosing will make it possible for us to use [rapamycin] more widely in humans.”

Intermittent dosing with the immunosuppressant rapamycin can break an inflammatory loop that fuels cancer relapse, according to researchers.
The team found that rapamycin selectively breaks the cascade of inflammatory events that follow cellular senescence.
Once disrupted, it takes time for the inflammatory loop to re-establish itself. This suggests intermittent dosing could provide a way to reap the benefits of rapamycin while reducing side effects.
The researchers described these findings in Nature Cell Biology.
“We think this could provide a paradigm shift in the treatment of age-related disease, including cancer,” said study author Judith Campisi, PhD, of the Buck Institute for Research on Aging in Novato, California.
“Imagine the possibility of taking a pill for a few days or weeks every few years, as opposed to taking something with side effects every day for the rest of your life. It’s a new way of looking at how we could deal with age-related maladies.”
For this study, Dr Campisi and her colleagues administered rapamycin to mice with prostate cancer after they had received treatment with DNA-damaging chemotherapy.
The rapamycin reduced the secretion of inflammatory cytokines from senescent cells by suppressing the mTOR pathway. This suppressed the cells’ ability to stimulate tumor growth.
“DNA-damaging chemotherapy causes senescence, both to the tumor and its microenvironment,” Dr Campisi explained. “The tumor shrinks, but the immediate tissue environment is inflamed. We think signals from those inflamed cells trigger residual cancer cells to grow again. In the mice, rapamycin suppressed the ability of the tumor cells to relapse.”
The researchers said the paradigm-shifting potential of intermittent rapamycin dosing is based on the fact that it takes time for the inflammatory loop—fueled by the senescence-associated secretory phenotype (SASP)—to form and time for it to re-establish itself after a brief treatment period with rapamycin.
“Rapamycin blocks the production of a protein called IL-1α,” explained study author Remi-Martin Laberge, PhD, also of the Buck Institute.
“This in turn, suppresses IL6, a well-known inflammatory cytokine, at the level of transcription, which prevents the production of the IL6 protein. Because it acts at a deeper level within the cellular process, it takes longer for it to get started again.”
Dr Laberge also pointed out that treatment with rapamycin selectivity impacts the SASP, preserving the function of factors essential for wound healing.
“It’s an elegant solution,” Dr Laberge said. “Imagine using a small hammer to delicately knock out one thing that is causing problems. We knocked it out, and it stayed out long enough to benefit the health of the animal.”
“We have yet to fully understand why suppressing the mTOR pathway via rapamycin increases lifespan and health span in mice,” Dr Campisi added. “This work helps illuminate the puzzle. Perhaps the mice are living longer because they have less overall inflammation, and maybe intermittent dosing will make it possible for us to use [rapamycin] more widely in humans.”

Intermittent dosing with the immunosuppressant rapamycin can break an inflammatory loop that fuels cancer relapse, according to researchers.
The team found that rapamycin selectively breaks the cascade of inflammatory events that follow cellular senescence.
Once disrupted, it takes time for the inflammatory loop to re-establish itself. This suggests intermittent dosing could provide a way to reap the benefits of rapamycin while reducing side effects.
The researchers described these findings in Nature Cell Biology.
“We think this could provide a paradigm shift in the treatment of age-related disease, including cancer,” said study author Judith Campisi, PhD, of the Buck Institute for Research on Aging in Novato, California.
“Imagine the possibility of taking a pill for a few days or weeks every few years, as opposed to taking something with side effects every day for the rest of your life. It’s a new way of looking at how we could deal with age-related maladies.”
For this study, Dr Campisi and her colleagues administered rapamycin to mice with prostate cancer after they had received treatment with DNA-damaging chemotherapy.
The rapamycin reduced the secretion of inflammatory cytokines from senescent cells by suppressing the mTOR pathway. This suppressed the cells’ ability to stimulate tumor growth.
“DNA-damaging chemotherapy causes senescence, both to the tumor and its microenvironment,” Dr Campisi explained. “The tumor shrinks, but the immediate tissue environment is inflamed. We think signals from those inflamed cells trigger residual cancer cells to grow again. In the mice, rapamycin suppressed the ability of the tumor cells to relapse.”
The researchers said the paradigm-shifting potential of intermittent rapamycin dosing is based on the fact that it takes time for the inflammatory loop—fueled by the senescence-associated secretory phenotype (SASP)—to form and time for it to re-establish itself after a brief treatment period with rapamycin.
“Rapamycin blocks the production of a protein called IL-1α,” explained study author Remi-Martin Laberge, PhD, also of the Buck Institute.
“This in turn, suppresses IL6, a well-known inflammatory cytokine, at the level of transcription, which prevents the production of the IL6 protein. Because it acts at a deeper level within the cellular process, it takes longer for it to get started again.”
Dr Laberge also pointed out that treatment with rapamycin selectivity impacts the SASP, preserving the function of factors essential for wound healing.
“It’s an elegant solution,” Dr Laberge said. “Imagine using a small hammer to delicately knock out one thing that is causing problems. We knocked it out, and it stayed out long enough to benefit the health of the animal.”
“We have yet to fully understand why suppressing the mTOR pathway via rapamycin increases lifespan and health span in mice,” Dr Campisi added. “This work helps illuminate the puzzle. Perhaps the mice are living longer because they have less overall inflammation, and maybe intermittent dosing will make it possible for us to use [rapamycin] more widely in humans.”
Methotrexate could treat MPNs cheaply, team says
Photo courtesy of the
National Cancer Institute
Preclinical research suggests the antineoplastic agent methotrexate (MTX) could be used to treat patients with myeloproliferative neoplasms (MPNs).
Experiments in Drosophila and human cell lines showed that MTX reduces JAK/STAT pathway activity and MPN-associated pathway signaling.
Researchers therefore speculated that low doses of MTX might treat MPNs as effectively as the JAK1/2 inhibitor ruxolitinib, but for a lower cost.
“Given that a year’s course of low-dose MTX costs around £30, the potential to repurpose MTX could provide thousands of patients with a much-needed treatment option and also generate substantial savings for healthcare systems,” said study author Martin Zeidler, DPhil, of The University of Sheffield in the UK.
He and his colleagues noted that, in comparison, ruxolitinib costs more than £40,000 per year per patient.
The researchers made this comparison and described their work with MTX in PLOS ONE.
The team used cells from the fruit fly Drosophila to screen for small molecules that suppress the JAK/STAT signaling pathway, which is central to the development of MPNs in humans. The screen suggested that MTX and a related molecule, aminopterin, suppress STAT activation.
So the researchers conducted experiments in human cell lines and found that MTX suppresses human JAK/STAT signaling without affecting other phosphorylation-dependent pathways.
The team also found that MTX significantly reduces STAT5 phosphorylation in cells expressing JAK2 V617F. However, MTX-treated cells can still respond to physiological levels of erythropoietin.
The researchers are now looking to undertake clinical trials to examine the possibility of repurposing low-dose MTX for the treatment of MPNs.
“We have the potential to revolutionize the treatment of this group of chronic diseases—a breakthrough that may ultimately represent a new treatment option able to bring relief to both patients and health funders,” Dr Zeidler said.

Photo courtesy of the
National Cancer Institute
Preclinical research suggests the antineoplastic agent methotrexate (MTX) could be used to treat patients with myeloproliferative neoplasms (MPNs).
Experiments in Drosophila and human cell lines showed that MTX reduces JAK/STAT pathway activity and MPN-associated pathway signaling.
Researchers therefore speculated that low doses of MTX might treat MPNs as effectively as the JAK1/2 inhibitor ruxolitinib, but for a lower cost.
“Given that a year’s course of low-dose MTX costs around £30, the potential to repurpose MTX could provide thousands of patients with a much-needed treatment option and also generate substantial savings for healthcare systems,” said study author Martin Zeidler, DPhil, of The University of Sheffield in the UK.
He and his colleagues noted that, in comparison, ruxolitinib costs more than £40,000 per year per patient.
The researchers made this comparison and described their work with MTX in PLOS ONE.
The team used cells from the fruit fly Drosophila to screen for small molecules that suppress the JAK/STAT signaling pathway, which is central to the development of MPNs in humans. The screen suggested that MTX and a related molecule, aminopterin, suppress STAT activation.
So the researchers conducted experiments in human cell lines and found that MTX suppresses human JAK/STAT signaling without affecting other phosphorylation-dependent pathways.
The team also found that MTX significantly reduces STAT5 phosphorylation in cells expressing JAK2 V617F. However, MTX-treated cells can still respond to physiological levels of erythropoietin.
The researchers are now looking to undertake clinical trials to examine the possibility of repurposing low-dose MTX for the treatment of MPNs.
“We have the potential to revolutionize the treatment of this group of chronic diseases—a breakthrough that may ultimately represent a new treatment option able to bring relief to both patients and health funders,” Dr Zeidler said.

Photo courtesy of the
National Cancer Institute
Preclinical research suggests the antineoplastic agent methotrexate (MTX) could be used to treat patients with myeloproliferative neoplasms (MPNs).
Experiments in Drosophila and human cell lines showed that MTX reduces JAK/STAT pathway activity and MPN-associated pathway signaling.
Researchers therefore speculated that low doses of MTX might treat MPNs as effectively as the JAK1/2 inhibitor ruxolitinib, but for a lower cost.
“Given that a year’s course of low-dose MTX costs around £30, the potential to repurpose MTX could provide thousands of patients with a much-needed treatment option and also generate substantial savings for healthcare systems,” said study author Martin Zeidler, DPhil, of The University of Sheffield in the UK.
He and his colleagues noted that, in comparison, ruxolitinib costs more than £40,000 per year per patient.
The researchers made this comparison and described their work with MTX in PLOS ONE.
The team used cells from the fruit fly Drosophila to screen for small molecules that suppress the JAK/STAT signaling pathway, which is central to the development of MPNs in humans. The screen suggested that MTX and a related molecule, aminopterin, suppress STAT activation.
So the researchers conducted experiments in human cell lines and found that MTX suppresses human JAK/STAT signaling without affecting other phosphorylation-dependent pathways.
The team also found that MTX significantly reduces STAT5 phosphorylation in cells expressing JAK2 V617F. However, MTX-treated cells can still respond to physiological levels of erythropoietin.
The researchers are now looking to undertake clinical trials to examine the possibility of repurposing low-dose MTX for the treatment of MPNs.
“We have the potential to revolutionize the treatment of this group of chronic diseases—a breakthrough that may ultimately represent a new treatment option able to bring relief to both patients and health funders,” Dr Zeidler said.