User login
IL-2 variant proves active against ATL, GVHD

Photo by Rhoda Baer
Researchers say they have created interleukin-2 (IL-2) variants that function as IL-2-receptor signaling “clamps” and allow for “fine tuning” of the signaling amplitude.
One variant, known as H9-RETR, was able to inhibit the actions of endogenous IL-2 and IL-15, prolong survival in a mouse model of graft-vs-host disease (GVHD), and inhibit the proliferation of cells derived from a patient with smoldering adult T-cell
leukemia (ATL).
The researchers reported these results in Immunity.
Warren J. Leonard, MD, of the National Heart, Lung, and Blood Institute in Bethesda, Maryland, and his colleagues developed IL-2 variants in which activity can be tuned to either boost or block immune responses, depending on the desired therapeutic application.
The researchers said these variants had high affinity for IL-2Rβ and inhibited binding of endogenous IL-2, but their interaction with γc was weakened, thereby weakening IL-2Rβ-γc heterodimerization.
The team found that IL-2 signaling strength was inversely correlated with the degree of mutation at the γc interface. And differential effects on cell proliferation were dependent upon the cells’ state of activation.
One of the IL-2 variants, H9-RETR, inhibited IL-2- and IL-15-mediated proliferation and cytotoxicity. H9-RETR inhibited cytokine signaling and natural killer cell activity as well or better than blocking antibodies to IL-2Rα and IL-2Rβ.
In experiments with cells isolated from a patient with smoldering ATL, H9-RETR blocked IL-2 signaling and inhibited the spontaneous proliferation of ATL cells. In this regard, H9-RETR was at least as effective as the anti-IL-2Rα antibody daclizumab and much more effective than the anti-IL-2Rβ antibody Mikβ1.
In a mouse model of GVHD, animals that received a stabilized, Fc-fusion version of H9-RETR (H9-RETR-Fc4) had significantly longer survival than control mice (which received only Fc4 protein).
All of the control mice had died by 40 days post-injection, but it took 60 days for all of the H9-RETR-Fc4-treated mice to die (P=0.0001).
The researchers believe their receptor-clamping approach could potentially be used to engineer other immune-system cytokines with therapeutic potential. ![]()

Photo by Rhoda Baer
Researchers say they have created interleukin-2 (IL-2) variants that function as IL-2-receptor signaling “clamps” and allow for “fine tuning” of the signaling amplitude.
One variant, known as H9-RETR, was able to inhibit the actions of endogenous IL-2 and IL-15, prolong survival in a mouse model of graft-vs-host disease (GVHD), and inhibit the proliferation of cells derived from a patient with smoldering adult T-cell
leukemia (ATL).
The researchers reported these results in Immunity.
Warren J. Leonard, MD, of the National Heart, Lung, and Blood Institute in Bethesda, Maryland, and his colleagues developed IL-2 variants in which activity can be tuned to either boost or block immune responses, depending on the desired therapeutic application.
The researchers said these variants had high affinity for IL-2Rβ and inhibited binding of endogenous IL-2, but their interaction with γc was weakened, thereby weakening IL-2Rβ-γc heterodimerization.
The team found that IL-2 signaling strength was inversely correlated with the degree of mutation at the γc interface. And differential effects on cell proliferation were dependent upon the cells’ state of activation.
One of the IL-2 variants, H9-RETR, inhibited IL-2- and IL-15-mediated proliferation and cytotoxicity. H9-RETR inhibited cytokine signaling and natural killer cell activity as well or better than blocking antibodies to IL-2Rα and IL-2Rβ.
In experiments with cells isolated from a patient with smoldering ATL, H9-RETR blocked IL-2 signaling and inhibited the spontaneous proliferation of ATL cells. In this regard, H9-RETR was at least as effective as the anti-IL-2Rα antibody daclizumab and much more effective than the anti-IL-2Rβ antibody Mikβ1.
In a mouse model of GVHD, animals that received a stabilized, Fc-fusion version of H9-RETR (H9-RETR-Fc4) had significantly longer survival than control mice (which received only Fc4 protein).
All of the control mice had died by 40 days post-injection, but it took 60 days for all of the H9-RETR-Fc4-treated mice to die (P=0.0001).
The researchers believe their receptor-clamping approach could potentially be used to engineer other immune-system cytokines with therapeutic potential. ![]()

Photo by Rhoda Baer
Researchers say they have created interleukin-2 (IL-2) variants that function as IL-2-receptor signaling “clamps” and allow for “fine tuning” of the signaling amplitude.
One variant, known as H9-RETR, was able to inhibit the actions of endogenous IL-2 and IL-15, prolong survival in a mouse model of graft-vs-host disease (GVHD), and inhibit the proliferation of cells derived from a patient with smoldering adult T-cell
leukemia (ATL).
The researchers reported these results in Immunity.
Warren J. Leonard, MD, of the National Heart, Lung, and Blood Institute in Bethesda, Maryland, and his colleagues developed IL-2 variants in which activity can be tuned to either boost or block immune responses, depending on the desired therapeutic application.
The researchers said these variants had high affinity for IL-2Rβ and inhibited binding of endogenous IL-2, but their interaction with γc was weakened, thereby weakening IL-2Rβ-γc heterodimerization.
The team found that IL-2 signaling strength was inversely correlated with the degree of mutation at the γc interface. And differential effects on cell proliferation were dependent upon the cells’ state of activation.
One of the IL-2 variants, H9-RETR, inhibited IL-2- and IL-15-mediated proliferation and cytotoxicity. H9-RETR inhibited cytokine signaling and natural killer cell activity as well or better than blocking antibodies to IL-2Rα and IL-2Rβ.
In experiments with cells isolated from a patient with smoldering ATL, H9-RETR blocked IL-2 signaling and inhibited the spontaneous proliferation of ATL cells. In this regard, H9-RETR was at least as effective as the anti-IL-2Rα antibody daclizumab and much more effective than the anti-IL-2Rβ antibody Mikβ1.
In a mouse model of GVHD, animals that received a stabilized, Fc-fusion version of H9-RETR (H9-RETR-Fc4) had significantly longer survival than control mice (which received only Fc4 protein).
All of the control mice had died by 40 days post-injection, but it took 60 days for all of the H9-RETR-Fc4-treated mice to die (P=0.0001).
The researchers believe their receptor-clamping approach could potentially be used to engineer other immune-system cytokines with therapeutic potential. ![]()
New insecticide targets malaria-carrying mosquito

Photo courtesy of the CDC
Recent progress in halting the spread of malaria has hinged, in part, on the use of insecticide-treated bed nets and spraying programs that target Anopheles gambiae mosquitoes.
Unfortunately, the mosquitoes are developing resistance to insecticides such as pyrethroid.
Wondering if they could defeat the mosquitoes by developing a new insecticide,a group of researchers set out to make blood meals toxic for Anopheles gambiae.
The team described their method in The Journal of Experimental Biology.
The researchers decided to target the mosquito glutamate gated chloride channel (AgGluCl), which is an essential component of the insect’s nervous system.
They generated antibodies that specifically targeted a portion of the protein exposed on the surface of nerves, a strategy they acknowledged was somewhat risky.
“Antibodies against a single mosquito antigen have never been shown to have mosquitocidal properties before, and the majority of previous research had focused on midgut antigens, while we were targeting a neuronal antigen expressed only in tissues found outside of the midgut,” said study author Jacob Meyers, a graduate student at Colorado State University in Fort Collins.
After injecting rabbits with a tiny portion of the surface of the AgGluCl protein channel, the researchers waited for the rabbits’ immune systems to begin producing antibodies tailored to the channel.
Then, the team collected the antibodies, mixed them with fresh blood, and fed the mixture to malaria-carrying mosquitoes (Anopheles gambiae), yellow fever-carrying mosquitoes (Aedes aegypti), and mosquitoes that carry the West Nile virus (Culex tarsalis).
Neither the yellow fever nor West Nile virus mosquitoes responded to the spiked blood. However, significant numbers of Anopheles gambiae mosquitoes died after consuming the blood/antibody cocktail. The highest antibody doses killed more than 90% of the insects within a day.
The researchers looked into why the yellow fever and West Nile virus mosquitoes had been immune to the antibodies and found the antibodies could not pass across the mosquitoes’ guts into the hemolymph. But the antibodies passed into the hemolymph of Anopheles gambiae mosquitoes with ease.
Intrigued by the antibodies’ mode of action, the researchers fed the insects a blood meal laced with the antibodies and a lethal dose of ivermectin, an insecticide that also targets the AgGluCl protein channel.
Then, the team monitored the mosquitoes’ survival to find out more about how the antibodies may destroy the insects. Mosquitoes that received ivermectin with the antibodies were more likely to survive than insects that received ivermectin alone.
“We believe that ivermectin is able to bind to AgGluCl,” Meyers said, “but the antibody keeps the channel from opening and becoming active.”
Having shown that antibodies targeted to the glutamate gated chloride channel in blood meals can be effective insecticides, the researchers are interested to find out if antibody-laced blood meals are equally deadly outside the lab.
First, though, the team plans to immunize cattle against the AgGluCl antigen and feed Anopheles gambiae on the immunized cattle in the lab. If the strategy proves successful, Meyers envisages a large-scale cattle immunization program as part of a combined attack on the malaria parasite. ![]()

Photo courtesy of the CDC
Recent progress in halting the spread of malaria has hinged, in part, on the use of insecticide-treated bed nets and spraying programs that target Anopheles gambiae mosquitoes.
Unfortunately, the mosquitoes are developing resistance to insecticides such as pyrethroid.
Wondering if they could defeat the mosquitoes by developing a new insecticide,a group of researchers set out to make blood meals toxic for Anopheles gambiae.
The team described their method in The Journal of Experimental Biology.
The researchers decided to target the mosquito glutamate gated chloride channel (AgGluCl), which is an essential component of the insect’s nervous system.
They generated antibodies that specifically targeted a portion of the protein exposed on the surface of nerves, a strategy they acknowledged was somewhat risky.
“Antibodies against a single mosquito antigen have never been shown to have mosquitocidal properties before, and the majority of previous research had focused on midgut antigens, while we were targeting a neuronal antigen expressed only in tissues found outside of the midgut,” said study author Jacob Meyers, a graduate student at Colorado State University in Fort Collins.
After injecting rabbits with a tiny portion of the surface of the AgGluCl protein channel, the researchers waited for the rabbits’ immune systems to begin producing antibodies tailored to the channel.
Then, the team collected the antibodies, mixed them with fresh blood, and fed the mixture to malaria-carrying mosquitoes (Anopheles gambiae), yellow fever-carrying mosquitoes (Aedes aegypti), and mosquitoes that carry the West Nile virus (Culex tarsalis).
Neither the yellow fever nor West Nile virus mosquitoes responded to the spiked blood. However, significant numbers of Anopheles gambiae mosquitoes died after consuming the blood/antibody cocktail. The highest antibody doses killed more than 90% of the insects within a day.
The researchers looked into why the yellow fever and West Nile virus mosquitoes had been immune to the antibodies and found the antibodies could not pass across the mosquitoes’ guts into the hemolymph. But the antibodies passed into the hemolymph of Anopheles gambiae mosquitoes with ease.
Intrigued by the antibodies’ mode of action, the researchers fed the insects a blood meal laced with the antibodies and a lethal dose of ivermectin, an insecticide that also targets the AgGluCl protein channel.
Then, the team monitored the mosquitoes’ survival to find out more about how the antibodies may destroy the insects. Mosquitoes that received ivermectin with the antibodies were more likely to survive than insects that received ivermectin alone.
“We believe that ivermectin is able to bind to AgGluCl,” Meyers said, “but the antibody keeps the channel from opening and becoming active.”
Having shown that antibodies targeted to the glutamate gated chloride channel in blood meals can be effective insecticides, the researchers are interested to find out if antibody-laced blood meals are equally deadly outside the lab.
First, though, the team plans to immunize cattle against the AgGluCl antigen and feed Anopheles gambiae on the immunized cattle in the lab. If the strategy proves successful, Meyers envisages a large-scale cattle immunization program as part of a combined attack on the malaria parasite. ![]()

Photo courtesy of the CDC
Recent progress in halting the spread of malaria has hinged, in part, on the use of insecticide-treated bed nets and spraying programs that target Anopheles gambiae mosquitoes.
Unfortunately, the mosquitoes are developing resistance to insecticides such as pyrethroid.
Wondering if they could defeat the mosquitoes by developing a new insecticide,a group of researchers set out to make blood meals toxic for Anopheles gambiae.
The team described their method in The Journal of Experimental Biology.
The researchers decided to target the mosquito glutamate gated chloride channel (AgGluCl), which is an essential component of the insect’s nervous system.
They generated antibodies that specifically targeted a portion of the protein exposed on the surface of nerves, a strategy they acknowledged was somewhat risky.
“Antibodies against a single mosquito antigen have never been shown to have mosquitocidal properties before, and the majority of previous research had focused on midgut antigens, while we were targeting a neuronal antigen expressed only in tissues found outside of the midgut,” said study author Jacob Meyers, a graduate student at Colorado State University in Fort Collins.
After injecting rabbits with a tiny portion of the surface of the AgGluCl protein channel, the researchers waited for the rabbits’ immune systems to begin producing antibodies tailored to the channel.
Then, the team collected the antibodies, mixed them with fresh blood, and fed the mixture to malaria-carrying mosquitoes (Anopheles gambiae), yellow fever-carrying mosquitoes (Aedes aegypti), and mosquitoes that carry the West Nile virus (Culex tarsalis).
Neither the yellow fever nor West Nile virus mosquitoes responded to the spiked blood. However, significant numbers of Anopheles gambiae mosquitoes died after consuming the blood/antibody cocktail. The highest antibody doses killed more than 90% of the insects within a day.
The researchers looked into why the yellow fever and West Nile virus mosquitoes had been immune to the antibodies and found the antibodies could not pass across the mosquitoes’ guts into the hemolymph. But the antibodies passed into the hemolymph of Anopheles gambiae mosquitoes with ease.
Intrigued by the antibodies’ mode of action, the researchers fed the insects a blood meal laced with the antibodies and a lethal dose of ivermectin, an insecticide that also targets the AgGluCl protein channel.
Then, the team monitored the mosquitoes’ survival to find out more about how the antibodies may destroy the insects. Mosquitoes that received ivermectin with the antibodies were more likely to survive than insects that received ivermectin alone.
“We believe that ivermectin is able to bind to AgGluCl,” Meyers said, “but the antibody keeps the channel from opening and becoming active.”
Having shown that antibodies targeted to the glutamate gated chloride channel in blood meals can be effective insecticides, the researchers are interested to find out if antibody-laced blood meals are equally deadly outside the lab.
First, though, the team plans to immunize cattle against the AgGluCl antigen and feed Anopheles gambiae on the immunized cattle in the lab. If the strategy proves successful, Meyers envisages a large-scale cattle immunization program as part of a combined attack on the malaria parasite. ![]()
System can identify life-threatening bleeding

Photo courtesy of UAB Hospital
A computerized system that analyzes vital signs can help healthcare professionals more accurately diagnose trauma patients with life-threatening bleeding, according to research published in Shock.
The system, known as APPRAISE, simultaneously analyzes blood pressure, heart rate, and breathing patterns during emergency transport.
And investigators found that APPRAISE accurately detected most cases of life-threatening bleeding.
“Providing faster care to patients who are bleeding to death saves lives,” said study author Andrew Reisner, MD, of Massachusetts General Hospital in Boston.
“While the clinical information that ambulance crews call in to trauma centers was sufficient to determine the presence of a life-threatening hemorrhage in about half the patients we studied, many other patients were in a ‘gray area’ and may or may not have been at risk of bleeding to death.”
“Our study demonstrated that automated analysis of patients’ vital signs during prehospital transport was significantly better at discriminating between patients who did and did not have life-threatening hemorrhage.”
The APPRAISE system incorporates software based on statistical techniques currently used in stock market trading and manufacturing to determine whether particular data points represent real problems and not random fluctuations.
The system uses an ultracompact personal computer to analyze data gathered by a standard patient monitor used in emergency transport vehicles.
For this study, the system was installed in 2 MedFlight helicopters and collected data on more than 200 trauma patients transported to participating Boston hospitals from February 2010 to December 2012. So that patients’ care was not affected by a still-unproven system, the APPRAISE system’s analysis was not provided to MedFlight crews.
The researchers also analyzed information from a 2005 study of vital sign data gathered manually by a Houston-based air ambulance system.
The team found the APPRAISE system could identify, with 76% sensitivity, patients who needed 9 or more units of packed red blood cells within 24 hours.
This was significantly more sensitive (P<0.05) than any prehospital Shock Index of 1.4 or higher (59%), initial systolic blood pressure less than 110 mmHg (50%), and any prehospital systolic blood pressure less than 90 mmHg (50%).
However, there was no signficant difference between the different measures with regard to specificity for identifying patients who did not need a blood transfusion within 24 hours.
Specificity was 87% for APPRAISE, 88% for any Shock Index of 1.4 or higher, 88% for initial systolic blood pressure less than 110 mmHg, and 90% for any prehospital systolic blood pressure less than 90 mmHg.
Notifications provided by APPRAISE would have been available within 10 minutes of initial monitoring and a median of 20 minutes before patients arrived at the trauma centers.
“The fact that decisions to proceed with surgery or to replenish lost blood often occur only after patients’ arrival means there are delays—sometimes brief but sometimes prolonged—in initiating such life-saving interventions,” Dr Reisner said.
“We are now working on a follow-up study to use this system in actual trauma care and will be measuring whether it truly leads to faster treatment of life-threatening hemorrhage and better patient outcomes. This approach could also be helpful for patients transported by ground ambulance and for hospitalized patients at risk of unexpected hemorrhage, such as during recovery from major surgery.” ![]()

Photo courtesy of UAB Hospital
A computerized system that analyzes vital signs can help healthcare professionals more accurately diagnose trauma patients with life-threatening bleeding, according to research published in Shock.
The system, known as APPRAISE, simultaneously analyzes blood pressure, heart rate, and breathing patterns during emergency transport.
And investigators found that APPRAISE accurately detected most cases of life-threatening bleeding.
“Providing faster care to patients who are bleeding to death saves lives,” said study author Andrew Reisner, MD, of Massachusetts General Hospital in Boston.
“While the clinical information that ambulance crews call in to trauma centers was sufficient to determine the presence of a life-threatening hemorrhage in about half the patients we studied, many other patients were in a ‘gray area’ and may or may not have been at risk of bleeding to death.”
“Our study demonstrated that automated analysis of patients’ vital signs during prehospital transport was significantly better at discriminating between patients who did and did not have life-threatening hemorrhage.”
The APPRAISE system incorporates software based on statistical techniques currently used in stock market trading and manufacturing to determine whether particular data points represent real problems and not random fluctuations.
The system uses an ultracompact personal computer to analyze data gathered by a standard patient monitor used in emergency transport vehicles.
For this study, the system was installed in 2 MedFlight helicopters and collected data on more than 200 trauma patients transported to participating Boston hospitals from February 2010 to December 2012. So that patients’ care was not affected by a still-unproven system, the APPRAISE system’s analysis was not provided to MedFlight crews.
The researchers also analyzed information from a 2005 study of vital sign data gathered manually by a Houston-based air ambulance system.
The team found the APPRAISE system could identify, with 76% sensitivity, patients who needed 9 or more units of packed red blood cells within 24 hours.
This was significantly more sensitive (P<0.05) than any prehospital Shock Index of 1.4 or higher (59%), initial systolic blood pressure less than 110 mmHg (50%), and any prehospital systolic blood pressure less than 90 mmHg (50%).
However, there was no signficant difference between the different measures with regard to specificity for identifying patients who did not need a blood transfusion within 24 hours.
Specificity was 87% for APPRAISE, 88% for any Shock Index of 1.4 or higher, 88% for initial systolic blood pressure less than 110 mmHg, and 90% for any prehospital systolic blood pressure less than 90 mmHg.
Notifications provided by APPRAISE would have been available within 10 minutes of initial monitoring and a median of 20 minutes before patients arrived at the trauma centers.
“The fact that decisions to proceed with surgery or to replenish lost blood often occur only after patients’ arrival means there are delays—sometimes brief but sometimes prolonged—in initiating such life-saving interventions,” Dr Reisner said.
“We are now working on a follow-up study to use this system in actual trauma care and will be measuring whether it truly leads to faster treatment of life-threatening hemorrhage and better patient outcomes. This approach could also be helpful for patients transported by ground ambulance and for hospitalized patients at risk of unexpected hemorrhage, such as during recovery from major surgery.” ![]()

Photo courtesy of UAB Hospital
A computerized system that analyzes vital signs can help healthcare professionals more accurately diagnose trauma patients with life-threatening bleeding, according to research published in Shock.
The system, known as APPRAISE, simultaneously analyzes blood pressure, heart rate, and breathing patterns during emergency transport.
And investigators found that APPRAISE accurately detected most cases of life-threatening bleeding.
“Providing faster care to patients who are bleeding to death saves lives,” said study author Andrew Reisner, MD, of Massachusetts General Hospital in Boston.
“While the clinical information that ambulance crews call in to trauma centers was sufficient to determine the presence of a life-threatening hemorrhage in about half the patients we studied, many other patients were in a ‘gray area’ and may or may not have been at risk of bleeding to death.”
“Our study demonstrated that automated analysis of patients’ vital signs during prehospital transport was significantly better at discriminating between patients who did and did not have life-threatening hemorrhage.”
The APPRAISE system incorporates software based on statistical techniques currently used in stock market trading and manufacturing to determine whether particular data points represent real problems and not random fluctuations.
The system uses an ultracompact personal computer to analyze data gathered by a standard patient monitor used in emergency transport vehicles.
For this study, the system was installed in 2 MedFlight helicopters and collected data on more than 200 trauma patients transported to participating Boston hospitals from February 2010 to December 2012. So that patients’ care was not affected by a still-unproven system, the APPRAISE system’s analysis was not provided to MedFlight crews.
The researchers also analyzed information from a 2005 study of vital sign data gathered manually by a Houston-based air ambulance system.
The team found the APPRAISE system could identify, with 76% sensitivity, patients who needed 9 or more units of packed red blood cells within 24 hours.
This was significantly more sensitive (P<0.05) than any prehospital Shock Index of 1.4 or higher (59%), initial systolic blood pressure less than 110 mmHg (50%), and any prehospital systolic blood pressure less than 90 mmHg (50%).
However, there was no signficant difference between the different measures with regard to specificity for identifying patients who did not need a blood transfusion within 24 hours.
Specificity was 87% for APPRAISE, 88% for any Shock Index of 1.4 or higher, 88% for initial systolic blood pressure less than 110 mmHg, and 90% for any prehospital systolic blood pressure less than 90 mmHg.
Notifications provided by APPRAISE would have been available within 10 minutes of initial monitoring and a median of 20 minutes before patients arrived at the trauma centers.
“The fact that decisions to proceed with surgery or to replenish lost blood often occur only after patients’ arrival means there are delays—sometimes brief but sometimes prolonged—in initiating such life-saving interventions,” Dr Reisner said.
“We are now working on a follow-up study to use this system in actual trauma care and will be measuring whether it truly leads to faster treatment of life-threatening hemorrhage and better patient outcomes. This approach could also be helpful for patients transported by ground ambulance and for hospitalized patients at risk of unexpected hemorrhage, such as during recovery from major surgery.” ![]()
Modified T cells may enhance MM treatment
Image by Daniel E. Sabath
Modified T cells derived from patients’ bone marrow can help treat multiple myeloma (MM), according to researchers.
They modified marrow-infiltrating lymphocytes (MILs) to target MM cells and administered them to patients following chemotherapy and hematopoietic stem cell transplant (HSCT).
Seven of the 22 patients treated achieved at least a 90% reduction in disease burden, which translated to an improvement in progression-free survival.
The researchers reported these results in Science Translational Medicine.
“What we learned in this small trial is that large numbers of activated MILs can selectively target and kill myeloma cells,” said Ivan Borrello, MD, of the Johns Hopkins University School of Medicine in Baltimore, Maryland.
Dr Borrello and his colleagues set out to test the MILs in 25 patients with newly diagnosed (n=14) or relapsed (n=11) MM. The patients’ median age was 56 years (range, 30-71), 64% were male, and 82% were white.
The median number of prior treatments was 2.13 (range, 1-6). For this trial, the patients received high-dose melphalan, autologous HSCT, and MIL therapy.
Patient outcomes
Prior to HSCT, 14 patients had a partial response (PR, 64%), 3 had a very good PR (14%), 2 had a minor response (8%), and 3 had stable disease (SD, 14%). After HSCT, 7 patients were in complete response (CR, 32%), 6 had a PR (27%), 6 had SD (27%), and 3 had progressive disease (PD, 14%).
Patients received MILs when they obtained the maximal response to the last therapy. The researchers retrieved MILs from each patient’s bone marrow and expanded them with anti-CD3/CD28 beads plus interleukin-2.
Twenty-two patients received a median of 9.5×108 MILs. Three patients relapsed before they could receive the MILs.
After MIL treatment, the overall response rate was 54%. Twenty-seven percent of patients achieved a CR, 27% had a PR, 23% had SD, and 14% had PD.
Patients who achieved at least a 90% reduction in disease burden (n=7) had superior progression-free survival compared to patients who did not respond as well to treatment (n=15).
The median progression-free survival was 25.1 months and 11.8 months, respectively (P=0.01). There was no significant difference between the groups with regard to overall survival.
The researchers said none of the patients experienced serious adverse events related to the MIL therapy.
Future directions
Dr Borrello said several US cancer centers have tested similar experimental treatments, but his team is believed to be the only one testing MILs. Other types of tumor-infiltrating cells can be used, but they are usually less plentiful in patients’ tumors and may not grow as well outside the body, he said.
“Typically, immune cells from solid tumors, called tumor-infiltrating lymphocytes, can be harvested and grown in only about 25% of patients who could potentially be eligible for the therapy,” said Kimberly Noonan, PhD, also of the Johns Hopkins University School of Medicine. “But in our clinical trial, we were able to harvest and grow MILs from all 22 patients.”
Dr Noonan said this small trial helped the researchers learn more about which patients may benefit from MIL therapy. For example, the team was able to determine how many of the MILs grown in the lab were specifically targeted to a patient’s tumor and whether they continued to target the tumor after being infused.
Additionally, the researchers found that patients with a high number of central memory cells before treatment had a better response to MIL therapy. Patients who began treatment with signs of an overactive immune response did not respond as well.
Dr Noonan said the team has used these data to guide 2 other ongoing trials of MILs. Those studies, she said, are trying to extend antitumor response and tumor specificity by combining the MILs with a cancer vaccine called GVAX and the drug lenalidomide, which stimulates T-cell responses.
All of the trials have also shed light on new ways to grow the MILs.
“In most of these trials, you see that the more cells you get, the better response you get in patients,” Dr Noonan said. “Learning how to improve cell growth may therefore improve the therapy.”
Researchers are also developing MILs to treat solid tumors such as lung, esophageal, and gastric cancers, as well as the pediatric cancers neuroblastoma and Ewing’s sarcoma. ![]()

Image by Daniel E. Sabath
Modified T cells derived from patients’ bone marrow can help treat multiple myeloma (MM), according to researchers.
They modified marrow-infiltrating lymphocytes (MILs) to target MM cells and administered them to patients following chemotherapy and hematopoietic stem cell transplant (HSCT).
Seven of the 22 patients treated achieved at least a 90% reduction in disease burden, which translated to an improvement in progression-free survival.
The researchers reported these results in Science Translational Medicine.
“What we learned in this small trial is that large numbers of activated MILs can selectively target and kill myeloma cells,” said Ivan Borrello, MD, of the Johns Hopkins University School of Medicine in Baltimore, Maryland.
Dr Borrello and his colleagues set out to test the MILs in 25 patients with newly diagnosed (n=14) or relapsed (n=11) MM. The patients’ median age was 56 years (range, 30-71), 64% were male, and 82% were white.
The median number of prior treatments was 2.13 (range, 1-6). For this trial, the patients received high-dose melphalan, autologous HSCT, and MIL therapy.
Patient outcomes
Prior to HSCT, 14 patients had a partial response (PR, 64%), 3 had a very good PR (14%), 2 had a minor response (8%), and 3 had stable disease (SD, 14%). After HSCT, 7 patients were in complete response (CR, 32%), 6 had a PR (27%), 6 had SD (27%), and 3 had progressive disease (PD, 14%).
Patients received MILs when they obtained the maximal response to the last therapy. The researchers retrieved MILs from each patient’s bone marrow and expanded them with anti-CD3/CD28 beads plus interleukin-2.
Twenty-two patients received a median of 9.5×108 MILs. Three patients relapsed before they could receive the MILs.
After MIL treatment, the overall response rate was 54%. Twenty-seven percent of patients achieved a CR, 27% had a PR, 23% had SD, and 14% had PD.
Patients who achieved at least a 90% reduction in disease burden (n=7) had superior progression-free survival compared to patients who did not respond as well to treatment (n=15).
The median progression-free survival was 25.1 months and 11.8 months, respectively (P=0.01). There was no significant difference between the groups with regard to overall survival.
The researchers said none of the patients experienced serious adverse events related to the MIL therapy.
Future directions
Dr Borrello said several US cancer centers have tested similar experimental treatments, but his team is believed to be the only one testing MILs. Other types of tumor-infiltrating cells can be used, but they are usually less plentiful in patients’ tumors and may not grow as well outside the body, he said.
“Typically, immune cells from solid tumors, called tumor-infiltrating lymphocytes, can be harvested and grown in only about 25% of patients who could potentially be eligible for the therapy,” said Kimberly Noonan, PhD, also of the Johns Hopkins University School of Medicine. “But in our clinical trial, we were able to harvest and grow MILs from all 22 patients.”
Dr Noonan said this small trial helped the researchers learn more about which patients may benefit from MIL therapy. For example, the team was able to determine how many of the MILs grown in the lab were specifically targeted to a patient’s tumor and whether they continued to target the tumor after being infused.
Additionally, the researchers found that patients with a high number of central memory cells before treatment had a better response to MIL therapy. Patients who began treatment with signs of an overactive immune response did not respond as well.
Dr Noonan said the team has used these data to guide 2 other ongoing trials of MILs. Those studies, she said, are trying to extend antitumor response and tumor specificity by combining the MILs with a cancer vaccine called GVAX and the drug lenalidomide, which stimulates T-cell responses.
All of the trials have also shed light on new ways to grow the MILs.
“In most of these trials, you see that the more cells you get, the better response you get in patients,” Dr Noonan said. “Learning how to improve cell growth may therefore improve the therapy.”
Researchers are also developing MILs to treat solid tumors such as lung, esophageal, and gastric cancers, as well as the pediatric cancers neuroblastoma and Ewing’s sarcoma. ![]()

Image by Daniel E. Sabath
Modified T cells derived from patients’ bone marrow can help treat multiple myeloma (MM), according to researchers.
They modified marrow-infiltrating lymphocytes (MILs) to target MM cells and administered them to patients following chemotherapy and hematopoietic stem cell transplant (HSCT).
Seven of the 22 patients treated achieved at least a 90% reduction in disease burden, which translated to an improvement in progression-free survival.
The researchers reported these results in Science Translational Medicine.
“What we learned in this small trial is that large numbers of activated MILs can selectively target and kill myeloma cells,” said Ivan Borrello, MD, of the Johns Hopkins University School of Medicine in Baltimore, Maryland.
Dr Borrello and his colleagues set out to test the MILs in 25 patients with newly diagnosed (n=14) or relapsed (n=11) MM. The patients’ median age was 56 years (range, 30-71), 64% were male, and 82% were white.
The median number of prior treatments was 2.13 (range, 1-6). For this trial, the patients received high-dose melphalan, autologous HSCT, and MIL therapy.
Patient outcomes
Prior to HSCT, 14 patients had a partial response (PR, 64%), 3 had a very good PR (14%), 2 had a minor response (8%), and 3 had stable disease (SD, 14%). After HSCT, 7 patients were in complete response (CR, 32%), 6 had a PR (27%), 6 had SD (27%), and 3 had progressive disease (PD, 14%).
Patients received MILs when they obtained the maximal response to the last therapy. The researchers retrieved MILs from each patient’s bone marrow and expanded them with anti-CD3/CD28 beads plus interleukin-2.
Twenty-two patients received a median of 9.5×108 MILs. Three patients relapsed before they could receive the MILs.
After MIL treatment, the overall response rate was 54%. Twenty-seven percent of patients achieved a CR, 27% had a PR, 23% had SD, and 14% had PD.
Patients who achieved at least a 90% reduction in disease burden (n=7) had superior progression-free survival compared to patients who did not respond as well to treatment (n=15).
The median progression-free survival was 25.1 months and 11.8 months, respectively (P=0.01). There was no significant difference between the groups with regard to overall survival.
The researchers said none of the patients experienced serious adverse events related to the MIL therapy.
Future directions
Dr Borrello said several US cancer centers have tested similar experimental treatments, but his team is believed to be the only one testing MILs. Other types of tumor-infiltrating cells can be used, but they are usually less plentiful in patients’ tumors and may not grow as well outside the body, he said.
“Typically, immune cells from solid tumors, called tumor-infiltrating lymphocytes, can be harvested and grown in only about 25% of patients who could potentially be eligible for the therapy,” said Kimberly Noonan, PhD, also of the Johns Hopkins University School of Medicine. “But in our clinical trial, we were able to harvest and grow MILs from all 22 patients.”
Dr Noonan said this small trial helped the researchers learn more about which patients may benefit from MIL therapy. For example, the team was able to determine how many of the MILs grown in the lab were specifically targeted to a patient’s tumor and whether they continued to target the tumor after being infused.
Additionally, the researchers found that patients with a high number of central memory cells before treatment had a better response to MIL therapy. Patients who began treatment with signs of an overactive immune response did not respond as well.
Dr Noonan said the team has used these data to guide 2 other ongoing trials of MILs. Those studies, she said, are trying to extend antitumor response and tumor specificity by combining the MILs with a cancer vaccine called GVAX and the drug lenalidomide, which stimulates T-cell responses.
All of the trials have also shed light on new ways to grow the MILs.
“In most of these trials, you see that the more cells you get, the better response you get in patients,” Dr Noonan said. “Learning how to improve cell growth may therefore improve the therapy.”
Researchers are also developing MILs to treat solid tumors such as lung, esophageal, and gastric cancers, as well as the pediatric cancers neuroblastoma and Ewing’s sarcoma. ![]()
Nanotherapy appears effective against MM

Photo by Aaron Logan
A nanoparticle-based therapy can effectively fight multiple myeloma (MM), according to preclinical research published in Molecular Cancer Therapy.
The nanoparticles protected their therapeutic cargo from degradation in the bloodstream and enhanced drug delivery into MM cells, thereby improving survival in mouse models of MM.
The nanoparticles carried an Sn 2 lipase-labile prodrug inhibitor of MYC-MAX dimerization (MI1-PD).
Researchers designed this prodrug for use in MM because MM pathogenesis is driven by the MYC oncoprotein, its dimerization with MAX, and the binding of this heterodimer to E-Boxes in the vicinity of target genes.
Previous research has shown that MYC inhibitors are extremely potent in vitro, but, when injected into the blood, they degrade immediately. The current study is the first to show that a MYC inhibitor can be effective in animals with cancer, as long as the drug has a vehicle to protect and deliver it into cancer cells.
“The nanoparticles serve as vehicles that protect the drug from the harsh environment of the blood,” said study author Gregory M. Lanza, MD, PhD, of the Washington University School of Medicine in St Louis, Missouri.
“In this case, the drug is modified into a prodrug and actually locked into the outer membrane of the particle.”
The nanoparticles are designed to carry targeting molecules that home in on and bind to MM cells that carry the complementary receptor. When a nanoparticle binds to an MM cell, their membranes fuse together, transferring MI1-PD to the malignant cell. When safely inside, MI1-PD is enzymatically freed to do its job, blocking MYC from activation.
In theory, noncancerous cells are unlikely to be adversely affected by MYC inhibition because healthy cells shouldn’t have highly activated MYC proteins to begin with, according to the researchers.
The team injected 2 sizes of MI1-PD-containing nanoparticles into mice with MM and found that the nanoparticles increased the animals’ median survival.
The median survival was 46 days for mice that received MI1-PD-containing, 200-nm nanoparticles and 28 days for mice that received control, 200-nm nanoparticles (P<0.05). For mice that received 20 nm nanoparticles, the median survival was 52 days and 29 days, respectively (P=0.001).
The researchers also pointed out that neither MI1 nor MI1-PD conferred a survival benefit for the mice when injected without nanoparticle protection.
“We’re excited about our results because there was no guarantee the nanotherapy would increase survival,” said study author Michael H. Tomasson, MD, of the Washington University School of Medicine.
“We injected the nanoparticles intravenously, and they found the tumors throughout the body, whether they were in the bone marrow, the spleen, or elsewhere.”
Drs Tomasson and Lanza said this technology is still years away from clinical trials, but they are optimistic about its future potential and are eager to begin that work. ![]()

Photo by Aaron Logan
A nanoparticle-based therapy can effectively fight multiple myeloma (MM), according to preclinical research published in Molecular Cancer Therapy.
The nanoparticles protected their therapeutic cargo from degradation in the bloodstream and enhanced drug delivery into MM cells, thereby improving survival in mouse models of MM.
The nanoparticles carried an Sn 2 lipase-labile prodrug inhibitor of MYC-MAX dimerization (MI1-PD).
Researchers designed this prodrug for use in MM because MM pathogenesis is driven by the MYC oncoprotein, its dimerization with MAX, and the binding of this heterodimer to E-Boxes in the vicinity of target genes.
Previous research has shown that MYC inhibitors are extremely potent in vitro, but, when injected into the blood, they degrade immediately. The current study is the first to show that a MYC inhibitor can be effective in animals with cancer, as long as the drug has a vehicle to protect and deliver it into cancer cells.
“The nanoparticles serve as vehicles that protect the drug from the harsh environment of the blood,” said study author Gregory M. Lanza, MD, PhD, of the Washington University School of Medicine in St Louis, Missouri.
“In this case, the drug is modified into a prodrug and actually locked into the outer membrane of the particle.”
The nanoparticles are designed to carry targeting molecules that home in on and bind to MM cells that carry the complementary receptor. When a nanoparticle binds to an MM cell, their membranes fuse together, transferring MI1-PD to the malignant cell. When safely inside, MI1-PD is enzymatically freed to do its job, blocking MYC from activation.
In theory, noncancerous cells are unlikely to be adversely affected by MYC inhibition because healthy cells shouldn’t have highly activated MYC proteins to begin with, according to the researchers.
The team injected 2 sizes of MI1-PD-containing nanoparticles into mice with MM and found that the nanoparticles increased the animals’ median survival.
The median survival was 46 days for mice that received MI1-PD-containing, 200-nm nanoparticles and 28 days for mice that received control, 200-nm nanoparticles (P<0.05). For mice that received 20 nm nanoparticles, the median survival was 52 days and 29 days, respectively (P=0.001).
The researchers also pointed out that neither MI1 nor MI1-PD conferred a survival benefit for the mice when injected without nanoparticle protection.
“We’re excited about our results because there was no guarantee the nanotherapy would increase survival,” said study author Michael H. Tomasson, MD, of the Washington University School of Medicine.
“We injected the nanoparticles intravenously, and they found the tumors throughout the body, whether they were in the bone marrow, the spleen, or elsewhere.”
Drs Tomasson and Lanza said this technology is still years away from clinical trials, but they are optimistic about its future potential and are eager to begin that work. ![]()

Photo by Aaron Logan
A nanoparticle-based therapy can effectively fight multiple myeloma (MM), according to preclinical research published in Molecular Cancer Therapy.
The nanoparticles protected their therapeutic cargo from degradation in the bloodstream and enhanced drug delivery into MM cells, thereby improving survival in mouse models of MM.
The nanoparticles carried an Sn 2 lipase-labile prodrug inhibitor of MYC-MAX dimerization (MI1-PD).
Researchers designed this prodrug for use in MM because MM pathogenesis is driven by the MYC oncoprotein, its dimerization with MAX, and the binding of this heterodimer to E-Boxes in the vicinity of target genes.
Previous research has shown that MYC inhibitors are extremely potent in vitro, but, when injected into the blood, they degrade immediately. The current study is the first to show that a MYC inhibitor can be effective in animals with cancer, as long as the drug has a vehicle to protect and deliver it into cancer cells.
“The nanoparticles serve as vehicles that protect the drug from the harsh environment of the blood,” said study author Gregory M. Lanza, MD, PhD, of the Washington University School of Medicine in St Louis, Missouri.
“In this case, the drug is modified into a prodrug and actually locked into the outer membrane of the particle.”
The nanoparticles are designed to carry targeting molecules that home in on and bind to MM cells that carry the complementary receptor. When a nanoparticle binds to an MM cell, their membranes fuse together, transferring MI1-PD to the malignant cell. When safely inside, MI1-PD is enzymatically freed to do its job, blocking MYC from activation.
In theory, noncancerous cells are unlikely to be adversely affected by MYC inhibition because healthy cells shouldn’t have highly activated MYC proteins to begin with, according to the researchers.
The team injected 2 sizes of MI1-PD-containing nanoparticles into mice with MM and found that the nanoparticles increased the animals’ median survival.
The median survival was 46 days for mice that received MI1-PD-containing, 200-nm nanoparticles and 28 days for mice that received control, 200-nm nanoparticles (P<0.05). For mice that received 20 nm nanoparticles, the median survival was 52 days and 29 days, respectively (P=0.001).
The researchers also pointed out that neither MI1 nor MI1-PD conferred a survival benefit for the mice when injected without nanoparticle protection.
“We’re excited about our results because there was no guarantee the nanotherapy would increase survival,” said study author Michael H. Tomasson, MD, of the Washington University School of Medicine.
“We injected the nanoparticles intravenously, and they found the tumors throughout the body, whether they were in the bone marrow, the spleen, or elsewhere.”
Drs Tomasson and Lanza said this technology is still years away from clinical trials, but they are optimistic about its future potential and are eager to begin that work. ![]()
CTLs captured on video destroying cancer cells
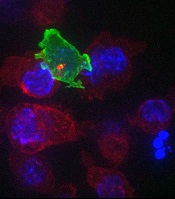
cancer cells (blue)
Image courtesy of Gillian
Griffiths and Jonny Settle
New research has illuminated the behavior of cytotoxic T cells (CTLs) as they hunt down and eliminate cancer cells.
Investigators used novel imaging techniques to capture the process on film and described their findings in an article published in Immunity.
The team captured the footage through high-resolution, 3D, time-lapse, multi-color imaging, making use of both spinning disk confocal microscopy and lattice light sheet microscopy.
These techniques involve capturing “slices” of an object and “stitching” them together to provide the final 3D images across the whole cell.
These approaches allowed the investigators to determine the order of events leading to the lethal “hit” CTLs deliver to cancer cells.
“Inside all of us lurks an army of ‘serial killers’ whose primary function is to kill again and again,” said study author Gillian Griffiths, PhD, of the Cambridge Institute for Medical Research in the UK.
“These [CTLs] patrol our bodies, identifying and destroying virally infected and cancer cells, and they do so with remarkable precision and efficiency.”
The CTLs, seen in the video as orange or green amorphous “blobs,” move around rapidly, investigating their environment as they travel.
When a CTL finds a cancer cell (blue), membrane protrusions rapidly explore the surface of the cell, checking for tell-tale signs that this is an uninvited guest.
The CTL binds to the cancer cell and injects cytotoxins (red) down microtubules to the interface between the T cell and the cancer cell, before puncturing the surface of the cancer cell and delivering its deadly cargo.
“In our bodies, where cells are packed together, it’s essential that the T cell focuses the lethal hit on its target,” Dr Griffiths explained. “Otherwise, it will cause collateral damage to neighboring, healthy cells.”
“Once the cytotoxins are injected into the cancer cell, its fate is sealed, and we can watch as it withers and dies. The T cell then moves on, hungry to find another victim.”
The investigators’ video is available on YouTube. ![]()

cancer cells (blue)
Image courtesy of Gillian
Griffiths and Jonny Settle
New research has illuminated the behavior of cytotoxic T cells (CTLs) as they hunt down and eliminate cancer cells.
Investigators used novel imaging techniques to capture the process on film and described their findings in an article published in Immunity.
The team captured the footage through high-resolution, 3D, time-lapse, multi-color imaging, making use of both spinning disk confocal microscopy and lattice light sheet microscopy.
These techniques involve capturing “slices” of an object and “stitching” them together to provide the final 3D images across the whole cell.
These approaches allowed the investigators to determine the order of events leading to the lethal “hit” CTLs deliver to cancer cells.
“Inside all of us lurks an army of ‘serial killers’ whose primary function is to kill again and again,” said study author Gillian Griffiths, PhD, of the Cambridge Institute for Medical Research in the UK.
“These [CTLs] patrol our bodies, identifying and destroying virally infected and cancer cells, and they do so with remarkable precision and efficiency.”
The CTLs, seen in the video as orange or green amorphous “blobs,” move around rapidly, investigating their environment as they travel.
When a CTL finds a cancer cell (blue), membrane protrusions rapidly explore the surface of the cell, checking for tell-tale signs that this is an uninvited guest.
The CTL binds to the cancer cell and injects cytotoxins (red) down microtubules to the interface between the T cell and the cancer cell, before puncturing the surface of the cancer cell and delivering its deadly cargo.
“In our bodies, where cells are packed together, it’s essential that the T cell focuses the lethal hit on its target,” Dr Griffiths explained. “Otherwise, it will cause collateral damage to neighboring, healthy cells.”
“Once the cytotoxins are injected into the cancer cell, its fate is sealed, and we can watch as it withers and dies. The T cell then moves on, hungry to find another victim.”
The investigators’ video is available on YouTube. ![]()

cancer cells (blue)
Image courtesy of Gillian
Griffiths and Jonny Settle
New research has illuminated the behavior of cytotoxic T cells (CTLs) as they hunt down and eliminate cancer cells.
Investigators used novel imaging techniques to capture the process on film and described their findings in an article published in Immunity.
The team captured the footage through high-resolution, 3D, time-lapse, multi-color imaging, making use of both spinning disk confocal microscopy and lattice light sheet microscopy.
These techniques involve capturing “slices” of an object and “stitching” them together to provide the final 3D images across the whole cell.
These approaches allowed the investigators to determine the order of events leading to the lethal “hit” CTLs deliver to cancer cells.
“Inside all of us lurks an army of ‘serial killers’ whose primary function is to kill again and again,” said study author Gillian Griffiths, PhD, of the Cambridge Institute for Medical Research in the UK.
“These [CTLs] patrol our bodies, identifying and destroying virally infected and cancer cells, and they do so with remarkable precision and efficiency.”
The CTLs, seen in the video as orange or green amorphous “blobs,” move around rapidly, investigating their environment as they travel.
When a CTL finds a cancer cell (blue), membrane protrusions rapidly explore the surface of the cell, checking for tell-tale signs that this is an uninvited guest.
The CTL binds to the cancer cell and injects cytotoxins (red) down microtubules to the interface between the T cell and the cancer cell, before puncturing the surface of the cancer cell and delivering its deadly cargo.
“In our bodies, where cells are packed together, it’s essential that the T cell focuses the lethal hit on its target,” Dr Griffiths explained. “Otherwise, it will cause collateral damage to neighboring, healthy cells.”
“Once the cytotoxins are injected into the cancer cell, its fate is sealed, and we can watch as it withers and dies. The T cell then moves on, hungry to find another victim.”
The investigators’ video is available on YouTube. ![]()
Approach could overcome drug resistance

Photo courtesy of the CDC
When treating patients with malaria and other infectious diseases, doctors should consider using drug combinations that reach similar parts of the body, according to researchers.
The group found that imperfect drug penetration—when drugs don’t reach all parts of the body—accelerates treatment resistance.
When there is a “pocket” of the body where only one drug is present, such as the brain or the digestive system, a pathogen can quickly develop resistance to one drug at a time.
“If there is a space where there is only one drug, that’s the place where the pathogen can start its escape,” said Pleuni Pennings, PhD, of San Francisco State University in California.
“Once it no longer has the first drug to deal with, it’s very easy for it to quickly become resistant to a second drug.”
Dr Pennings and her colleagues reported these findings in PNAS.
The team believes this research could have major implications for how treatment plans are designed and prescribed to patients with malaria, HIV, tuberculosis, and other ailments.
Because pathogens can quickly develop resistance to a single drug, providers often prescribe multiple drugs to increase their effectiveness.
The results of the study suggest that, when doing so, doctors should carefully consider which parts of the body each drug will reach and whether selecting medications with imperfect but similar penetrations might be the best treatment option.
“It may be better, in some cases, to leave a pocket of the body without any drugs instead of leaving a pocket with just one drug,” Dr Pennings said.
The study is the first to look at the connection between drug penetration and multidrug resistance. Dr Pennings and her colleagues ran computer simulations to observe the behavior of pathogens in response to changes in drugs and their levels of penetration.
The team found that, in instances where even small parts of the body could only be reached by one drug, the pathogen’s ability to build resistance to both drugs was accelerated compared to situations where no such pockets existed.
“This requires a new way of thinking about drug combinations that is a bit counterintuitive,” Dr Pennings said. “Suppose that drug A does not reach the brain, but drug B does. You’ll see the pathogen evolving resistance to drug B and assume that’s where the problem lies. But, in fact, it is drug A that is not doing its job because it’s not reaching the brain, and that’s the drug you may have to actually fix.”
With future research, Dr Pennings and her colleagues hope to outline the most effective drug combinations by exploring which parts of the body cannot be reached by specific drugs and where and how quickly specific pathogens are able to develop resistance. ![]()

Photo courtesy of the CDC
When treating patients with malaria and other infectious diseases, doctors should consider using drug combinations that reach similar parts of the body, according to researchers.
The group found that imperfect drug penetration—when drugs don’t reach all parts of the body—accelerates treatment resistance.
When there is a “pocket” of the body where only one drug is present, such as the brain or the digestive system, a pathogen can quickly develop resistance to one drug at a time.
“If there is a space where there is only one drug, that’s the place where the pathogen can start its escape,” said Pleuni Pennings, PhD, of San Francisco State University in California.
“Once it no longer has the first drug to deal with, it’s very easy for it to quickly become resistant to a second drug.”
Dr Pennings and her colleagues reported these findings in PNAS.
The team believes this research could have major implications for how treatment plans are designed and prescribed to patients with malaria, HIV, tuberculosis, and other ailments.
Because pathogens can quickly develop resistance to a single drug, providers often prescribe multiple drugs to increase their effectiveness.
The results of the study suggest that, when doing so, doctors should carefully consider which parts of the body each drug will reach and whether selecting medications with imperfect but similar penetrations might be the best treatment option.
“It may be better, in some cases, to leave a pocket of the body without any drugs instead of leaving a pocket with just one drug,” Dr Pennings said.
The study is the first to look at the connection between drug penetration and multidrug resistance. Dr Pennings and her colleagues ran computer simulations to observe the behavior of pathogens in response to changes in drugs and their levels of penetration.
The team found that, in instances where even small parts of the body could only be reached by one drug, the pathogen’s ability to build resistance to both drugs was accelerated compared to situations where no such pockets existed.
“This requires a new way of thinking about drug combinations that is a bit counterintuitive,” Dr Pennings said. “Suppose that drug A does not reach the brain, but drug B does. You’ll see the pathogen evolving resistance to drug B and assume that’s where the problem lies. But, in fact, it is drug A that is not doing its job because it’s not reaching the brain, and that’s the drug you may have to actually fix.”
With future research, Dr Pennings and her colleagues hope to outline the most effective drug combinations by exploring which parts of the body cannot be reached by specific drugs and where and how quickly specific pathogens are able to develop resistance. ![]()

Photo courtesy of the CDC
When treating patients with malaria and other infectious diseases, doctors should consider using drug combinations that reach similar parts of the body, according to researchers.
The group found that imperfect drug penetration—when drugs don’t reach all parts of the body—accelerates treatment resistance.
When there is a “pocket” of the body where only one drug is present, such as the brain or the digestive system, a pathogen can quickly develop resistance to one drug at a time.
“If there is a space where there is only one drug, that’s the place where the pathogen can start its escape,” said Pleuni Pennings, PhD, of San Francisco State University in California.
“Once it no longer has the first drug to deal with, it’s very easy for it to quickly become resistant to a second drug.”
Dr Pennings and her colleagues reported these findings in PNAS.
The team believes this research could have major implications for how treatment plans are designed and prescribed to patients with malaria, HIV, tuberculosis, and other ailments.
Because pathogens can quickly develop resistance to a single drug, providers often prescribe multiple drugs to increase their effectiveness.
The results of the study suggest that, when doing so, doctors should carefully consider which parts of the body each drug will reach and whether selecting medications with imperfect but similar penetrations might be the best treatment option.
“It may be better, in some cases, to leave a pocket of the body without any drugs instead of leaving a pocket with just one drug,” Dr Pennings said.
The study is the first to look at the connection between drug penetration and multidrug resistance. Dr Pennings and her colleagues ran computer simulations to observe the behavior of pathogens in response to changes in drugs and their levels of penetration.
The team found that, in instances where even small parts of the body could only be reached by one drug, the pathogen’s ability to build resistance to both drugs was accelerated compared to situations where no such pockets existed.
“This requires a new way of thinking about drug combinations that is a bit counterintuitive,” Dr Pennings said. “Suppose that drug A does not reach the brain, but drug B does. You’ll see the pathogen evolving resistance to drug B and assume that’s where the problem lies. But, in fact, it is drug A that is not doing its job because it’s not reaching the brain, and that’s the drug you may have to actually fix.”
With future research, Dr Pennings and her colleagues hope to outline the most effective drug combinations by exploring which parts of the body cannot be reached by specific drugs and where and how quickly specific pathogens are able to develop resistance.
Readmissions for severe sepsis fairly common

Photo courtesy of the CDC
DENVER—Severe sepsis is a significant cause of rehospitalization, along the lines of more commonly discussed conditions such as heart failure and pneumonia, according to research presented at the 2015 American Thoracic Society International Conference.
“Severe sepsis continues to be a common cause of hospitalization and has the associated high costs,” said Darya Rudym, MD, of New York University School of Medicine in New York.
She presented this finding at the meeting as publication number A3699.
Some quality measures associated with sepsis, such as length of hospital stay, have been studied previously. But Dr Rudym and her colleagues wanted to look at the rate of readmission within 30 days after discharge among patients with a diagnosis of severe sepsis.
The researchers also wanted to compare this rate to the readmission rates for outcomes reported by the Centers for Medicare and Medicaid Services, such as acute myocardial infarction, heart failure, and pneumonia.
The team examined inpatient discharges from Bellevue Hospital in New York City between July 2011 and July 2014 and identified subsequent readmissions to the hospital within 30 days.
During the 3-year study period, there were 22,712 discharges and an overall readmission rate of 15.31%, or 3477 patients.
The researchers used 3 different methods to identify patients with severe sepsis: Angus implementation, Martin implementation, and explicit ICD-9-CM sepsis codes.
There were 1801 cases of severe sepsis according to Angus implementation, 798 cases according to Martin implementation, and 579 cases according to ICD-9-CM sepsis codes.
In all, 266, 119, and 71 of these patients, respectively, were readmitted to the hospital within 30 days. This accounted for 14.77%, 14.91%, and 12.26% of cases, respectively.
The readmission rate for myocardial infarction was 8.67%. It was 14.46% for pneumonia, and 15% for heart failure.
“Readmission rates in severe sepsis are shown not to be significantly different from readmission rates in heart failure and pneumonia,” Dr Rudym noted.

Photo courtesy of the CDC
DENVER—Severe sepsis is a significant cause of rehospitalization, along the lines of more commonly discussed conditions such as heart failure and pneumonia, according to research presented at the 2015 American Thoracic Society International Conference.
“Severe sepsis continues to be a common cause of hospitalization and has the associated high costs,” said Darya Rudym, MD, of New York University School of Medicine in New York.
She presented this finding at the meeting as publication number A3699.
Some quality measures associated with sepsis, such as length of hospital stay, have been studied previously. But Dr Rudym and her colleagues wanted to look at the rate of readmission within 30 days after discharge among patients with a diagnosis of severe sepsis.
The researchers also wanted to compare this rate to the readmission rates for outcomes reported by the Centers for Medicare and Medicaid Services, such as acute myocardial infarction, heart failure, and pneumonia.
The team examined inpatient discharges from Bellevue Hospital in New York City between July 2011 and July 2014 and identified subsequent readmissions to the hospital within 30 days.
During the 3-year study period, there were 22,712 discharges and an overall readmission rate of 15.31%, or 3477 patients.
The researchers used 3 different methods to identify patients with severe sepsis: Angus implementation, Martin implementation, and explicit ICD-9-CM sepsis codes.
There were 1801 cases of severe sepsis according to Angus implementation, 798 cases according to Martin implementation, and 579 cases according to ICD-9-CM sepsis codes.
In all, 266, 119, and 71 of these patients, respectively, were readmitted to the hospital within 30 days. This accounted for 14.77%, 14.91%, and 12.26% of cases, respectively.
The readmission rate for myocardial infarction was 8.67%. It was 14.46% for pneumonia, and 15% for heart failure.
“Readmission rates in severe sepsis are shown not to be significantly different from readmission rates in heart failure and pneumonia,” Dr Rudym noted.

Photo courtesy of the CDC
DENVER—Severe sepsis is a significant cause of rehospitalization, along the lines of more commonly discussed conditions such as heart failure and pneumonia, according to research presented at the 2015 American Thoracic Society International Conference.
“Severe sepsis continues to be a common cause of hospitalization and has the associated high costs,” said Darya Rudym, MD, of New York University School of Medicine in New York.
She presented this finding at the meeting as publication number A3699.
Some quality measures associated with sepsis, such as length of hospital stay, have been studied previously. But Dr Rudym and her colleagues wanted to look at the rate of readmission within 30 days after discharge among patients with a diagnosis of severe sepsis.
The researchers also wanted to compare this rate to the readmission rates for outcomes reported by the Centers for Medicare and Medicaid Services, such as acute myocardial infarction, heart failure, and pneumonia.
The team examined inpatient discharges from Bellevue Hospital in New York City between July 2011 and July 2014 and identified subsequent readmissions to the hospital within 30 days.
During the 3-year study period, there were 22,712 discharges and an overall readmission rate of 15.31%, or 3477 patients.
The researchers used 3 different methods to identify patients with severe sepsis: Angus implementation, Martin implementation, and explicit ICD-9-CM sepsis codes.
There were 1801 cases of severe sepsis according to Angus implementation, 798 cases according to Martin implementation, and 579 cases according to ICD-9-CM sepsis codes.
In all, 266, 119, and 71 of these patients, respectively, were readmitted to the hospital within 30 days. This accounted for 14.77%, 14.91%, and 12.26% of cases, respectively.
The readmission rate for myocardial infarction was 8.67%. It was 14.46% for pneumonia, and 15% for heart failure.
“Readmission rates in severe sepsis are shown not to be significantly different from readmission rates in heart failure and pneumonia,” Dr Rudym noted.
How a vaccine may reduce the risk of ALL

Photo by Petr Kratochvil
Researchers believe they have discovered how a commonly administered vaccine protects children from developing acute lymphoblastic leukemia (ALL).
The Haemophilus influenzae Type b (Hib) vaccine is part of the standard vaccination schedule recommended for children by the US Centers for Disease Control and Prevention. The vaccine is routinely given in 4 doses before children reach 15 months of age.
The Hib vaccine prevents ear infections and meningitis caused by the Hib bacterium, but it may also protect against ALL.
This protection has been suggested in previous studies, but it is not well-known among the public at large, and the mechanism underlying this effect has been poorly understood.
Now, researchers have shown that recurrent Hib infections can shift certain genes into overdrive, converting pre-leukemic cells into full-blown cancer. The team described this work in Nature Immunology.
“These experiments help explain why the incidence of leukemia has been dramatically reduced since the advent of regular vaccinations during infancy,” said study author Markus Müschen, MD, PhD, of the University of California San Francisco.
“Hib and other childhood infections can cause recurrent and vehement immune responses, which we have found could lead to leukemia, but infants that have received vaccines are largely protected and acquire long-term immunity through very mild immune reactions.”
For this study, Dr Müschen and his colleagues tested the idea that chronic inflammation caused by recurrent infections might cause additional genetic lesions in blood cells already carrying an oncogene, promoting their transformation to overt disease.
The team conducted experiments in mice and discovered that the enzymes AID and RAG1-RAG2 drive this process. AID and RAG1-RAG2 introduce mutations in DNA that allow immune cells to adapt to infectious challenges, and these enzymes are necessary for a normal and efficient immune response.
The researchers found that, in the presence of chronic infection, AID and RAG1-RAG2 were hyperactivated, randomly cutting and mutating genes.
Additional experiments revealed that AID and RAG1-RAG2 working together is critical to introduce the additional lesions that result in ALL.
Though the researchers focused on a bacterial infection in this study, they believe the same mechanisms may be at work in viral infections.
The team is currently conducting experiments to determine if protection against leukemia is provided by vaccines against viral infections, such as the measles-mumps-rubella vaccine.

Photo by Petr Kratochvil
Researchers believe they have discovered how a commonly administered vaccine protects children from developing acute lymphoblastic leukemia (ALL).
The Haemophilus influenzae Type b (Hib) vaccine is part of the standard vaccination schedule recommended for children by the US Centers for Disease Control and Prevention. The vaccine is routinely given in 4 doses before children reach 15 months of age.
The Hib vaccine prevents ear infections and meningitis caused by the Hib bacterium, but it may also protect against ALL.
This protection has been suggested in previous studies, but it is not well-known among the public at large, and the mechanism underlying this effect has been poorly understood.
Now, researchers have shown that recurrent Hib infections can shift certain genes into overdrive, converting pre-leukemic cells into full-blown cancer. The team described this work in Nature Immunology.
“These experiments help explain why the incidence of leukemia has been dramatically reduced since the advent of regular vaccinations during infancy,” said study author Markus Müschen, MD, PhD, of the University of California San Francisco.
“Hib and other childhood infections can cause recurrent and vehement immune responses, which we have found could lead to leukemia, but infants that have received vaccines are largely protected and acquire long-term immunity through very mild immune reactions.”
For this study, Dr Müschen and his colleagues tested the idea that chronic inflammation caused by recurrent infections might cause additional genetic lesions in blood cells already carrying an oncogene, promoting their transformation to overt disease.
The team conducted experiments in mice and discovered that the enzymes AID and RAG1-RAG2 drive this process. AID and RAG1-RAG2 introduce mutations in DNA that allow immune cells to adapt to infectious challenges, and these enzymes are necessary for a normal and efficient immune response.
The researchers found that, in the presence of chronic infection, AID and RAG1-RAG2 were hyperactivated, randomly cutting and mutating genes.
Additional experiments revealed that AID and RAG1-RAG2 working together is critical to introduce the additional lesions that result in ALL.
Though the researchers focused on a bacterial infection in this study, they believe the same mechanisms may be at work in viral infections.
The team is currently conducting experiments to determine if protection against leukemia is provided by vaccines against viral infections, such as the measles-mumps-rubella vaccine.

Photo by Petr Kratochvil
Researchers believe they have discovered how a commonly administered vaccine protects children from developing acute lymphoblastic leukemia (ALL).
The Haemophilus influenzae Type b (Hib) vaccine is part of the standard vaccination schedule recommended for children by the US Centers for Disease Control and Prevention. The vaccine is routinely given in 4 doses before children reach 15 months of age.
The Hib vaccine prevents ear infections and meningitis caused by the Hib bacterium, but it may also protect against ALL.
This protection has been suggested in previous studies, but it is not well-known among the public at large, and the mechanism underlying this effect has been poorly understood.
Now, researchers have shown that recurrent Hib infections can shift certain genes into overdrive, converting pre-leukemic cells into full-blown cancer. The team described this work in Nature Immunology.
“These experiments help explain why the incidence of leukemia has been dramatically reduced since the advent of regular vaccinations during infancy,” said study author Markus Müschen, MD, PhD, of the University of California San Francisco.
“Hib and other childhood infections can cause recurrent and vehement immune responses, which we have found could lead to leukemia, but infants that have received vaccines are largely protected and acquire long-term immunity through very mild immune reactions.”
For this study, Dr Müschen and his colleagues tested the idea that chronic inflammation caused by recurrent infections might cause additional genetic lesions in blood cells already carrying an oncogene, promoting their transformation to overt disease.
The team conducted experiments in mice and discovered that the enzymes AID and RAG1-RAG2 drive this process. AID and RAG1-RAG2 introduce mutations in DNA that allow immune cells to adapt to infectious challenges, and these enzymes are necessary for a normal and efficient immune response.
The researchers found that, in the presence of chronic infection, AID and RAG1-RAG2 were hyperactivated, randomly cutting and mutating genes.
Additional experiments revealed that AID and RAG1-RAG2 working together is critical to introduce the additional lesions that result in ALL.
Though the researchers focused on a bacterial infection in this study, they believe the same mechanisms may be at work in viral infections.
The team is currently conducting experiments to determine if protection against leukemia is provided by vaccines against viral infections, such as the measles-mumps-rubella vaccine.
Drug on fast track to treat β-thalassemia

Image courtesy of NHLBI
The US Food and Drug Administration (FDA) has granted fast track designation to luspatercept for the treatment of patients with transfusion-dependent (TD) or non-transfusion-dependent (NTD) β-thalassemia.
Luspatercept is a modified activin receptor type IIB fusion protein that acts as a ligand trap for members of the transforming growth factor-β (TGF-β) superfamily involved in the late stages of erythropoiesis.
The drug regulates late-stage erythrocyte precursor differentiation and maturation. This mechanism of action is distinct from that of erythropoietin, which stimulates the proliferation of early stage erythrocyte precursor cells.
Luspatercept is currently in phase 2 trials in patients with β-thalassemia and individuals with myelodysplastic syndromes (MDS). Data from the trial in β-thalassemia were presented at the 2014 ASH Annual Meeting, and results from the MDS trial were recently presented at the 13th International Symposium on Myelodysplastic Syndromes.
Acceleron Pharma Inc. and Celgene Corporation are jointly developing luspatercept.
About fast track designation
The FDA’s fast track program is designed to facilitate the development of new drugs that are intended to treat serious or life-threatening conditions and that demonstrate the potential to address unmet medical needs.
The designation provides pharmaceutical companies with the opportunity for more frequent interaction with the FDA while developing a drug. It also allows a sponsor to submit sections of a biologics license application on a rolling basis, as they are finalized.
“The FDA’s fast track designation for the luspatercept development program recognizes the serious unmet medical needs of patients with β-thalassemia and the potential for luspatercept in this area,” said Jacqualyn A. Fouse, president of hematology/oncology for Celgene.
“Celgene and Acceleron are working diligently to initiate a phase 3 clinical program in 2015 to treat patients with β-thalassemia, and we look forward to continuing to work closely with health authorities and other important stakeholders to advance this program.”
Phase 2 trial in β-thalassemia
At ASH 2014, researchers presented results of a phase 2 trial in which they investigated whether luspatercept could increase hemoglobin levels and decrease transfusion dependence.
The goal was to increase hemoglobin levels 1.5 g/dL or more for at least 2 weeks in NTD patients and decrease the transfusion burden by 20% or more over 12 weeks in TD patients.
Thirty patients, 7 TD and 23 NTD, received an injection of luspatercept every 3 weeks for 3 months at doses ranging from 0.2 mg/kg to 1.0 mg/kg.
Three-quarters of patients who received 0.8 mg/kg to 1.0 mg/kg of luspatercept experienced an increase in their hemoglobin levels or a reduction in their transfusion burden.
Of the NTD patients, 8 of 12 with iron overload at baseline experienced a reduction in liver iron concentration of 1 mg or more at 16 weeks.
TD patients had a more than 60% reduction in transfusion burden over 12 weeks. This included 2 patients with β0 β0 genotype, who experienced a 79% and 75% reduction.
Luspatercept did not cause any treatment-related serious or severe adverse events. The most common adverse events were bone pain (20%), headache (17%), myalgia (13%), and asthenia (10%).
There was 1 grade 3 dose-limiting toxicity of worsening lumbar spine bone pain. And 3 patients discontinued treatment early, 1 each with occipital headache, ankle pain, and back pain.

Image courtesy of NHLBI
The US Food and Drug Administration (FDA) has granted fast track designation to luspatercept for the treatment of patients with transfusion-dependent (TD) or non-transfusion-dependent (NTD) β-thalassemia.
Luspatercept is a modified activin receptor type IIB fusion protein that acts as a ligand trap for members of the transforming growth factor-β (TGF-β) superfamily involved in the late stages of erythropoiesis.
The drug regulates late-stage erythrocyte precursor differentiation and maturation. This mechanism of action is distinct from that of erythropoietin, which stimulates the proliferation of early stage erythrocyte precursor cells.
Luspatercept is currently in phase 2 trials in patients with β-thalassemia and individuals with myelodysplastic syndromes (MDS). Data from the trial in β-thalassemia were presented at the 2014 ASH Annual Meeting, and results from the MDS trial were recently presented at the 13th International Symposium on Myelodysplastic Syndromes.
Acceleron Pharma Inc. and Celgene Corporation are jointly developing luspatercept.
About fast track designation
The FDA’s fast track program is designed to facilitate the development of new drugs that are intended to treat serious or life-threatening conditions and that demonstrate the potential to address unmet medical needs.
The designation provides pharmaceutical companies with the opportunity for more frequent interaction with the FDA while developing a drug. It also allows a sponsor to submit sections of a biologics license application on a rolling basis, as they are finalized.
“The FDA’s fast track designation for the luspatercept development program recognizes the serious unmet medical needs of patients with β-thalassemia and the potential for luspatercept in this area,” said Jacqualyn A. Fouse, president of hematology/oncology for Celgene.
“Celgene and Acceleron are working diligently to initiate a phase 3 clinical program in 2015 to treat patients with β-thalassemia, and we look forward to continuing to work closely with health authorities and other important stakeholders to advance this program.”
Phase 2 trial in β-thalassemia
At ASH 2014, researchers presented results of a phase 2 trial in which they investigated whether luspatercept could increase hemoglobin levels and decrease transfusion dependence.
The goal was to increase hemoglobin levels 1.5 g/dL or more for at least 2 weeks in NTD patients and decrease the transfusion burden by 20% or more over 12 weeks in TD patients.
Thirty patients, 7 TD and 23 NTD, received an injection of luspatercept every 3 weeks for 3 months at doses ranging from 0.2 mg/kg to 1.0 mg/kg.
Three-quarters of patients who received 0.8 mg/kg to 1.0 mg/kg of luspatercept experienced an increase in their hemoglobin levels or a reduction in their transfusion burden.
Of the NTD patients, 8 of 12 with iron overload at baseline experienced a reduction in liver iron concentration of 1 mg or more at 16 weeks.
TD patients had a more than 60% reduction in transfusion burden over 12 weeks. This included 2 patients with β0 β0 genotype, who experienced a 79% and 75% reduction.
Luspatercept did not cause any treatment-related serious or severe adverse events. The most common adverse events were bone pain (20%), headache (17%), myalgia (13%), and asthenia (10%).
There was 1 grade 3 dose-limiting toxicity of worsening lumbar spine bone pain. And 3 patients discontinued treatment early, 1 each with occipital headache, ankle pain, and back pain.

Image courtesy of NHLBI
The US Food and Drug Administration (FDA) has granted fast track designation to luspatercept for the treatment of patients with transfusion-dependent (TD) or non-transfusion-dependent (NTD) β-thalassemia.
Luspatercept is a modified activin receptor type IIB fusion protein that acts as a ligand trap for members of the transforming growth factor-β (TGF-β) superfamily involved in the late stages of erythropoiesis.
The drug regulates late-stage erythrocyte precursor differentiation and maturation. This mechanism of action is distinct from that of erythropoietin, which stimulates the proliferation of early stage erythrocyte precursor cells.
Luspatercept is currently in phase 2 trials in patients with β-thalassemia and individuals with myelodysplastic syndromes (MDS). Data from the trial in β-thalassemia were presented at the 2014 ASH Annual Meeting, and results from the MDS trial were recently presented at the 13th International Symposium on Myelodysplastic Syndromes.
Acceleron Pharma Inc. and Celgene Corporation are jointly developing luspatercept.
About fast track designation
The FDA’s fast track program is designed to facilitate the development of new drugs that are intended to treat serious or life-threatening conditions and that demonstrate the potential to address unmet medical needs.
The designation provides pharmaceutical companies with the opportunity for more frequent interaction with the FDA while developing a drug. It also allows a sponsor to submit sections of a biologics license application on a rolling basis, as they are finalized.
“The FDA’s fast track designation for the luspatercept development program recognizes the serious unmet medical needs of patients with β-thalassemia and the potential for luspatercept in this area,” said Jacqualyn A. Fouse, president of hematology/oncology for Celgene.
“Celgene and Acceleron are working diligently to initiate a phase 3 clinical program in 2015 to treat patients with β-thalassemia, and we look forward to continuing to work closely with health authorities and other important stakeholders to advance this program.”
Phase 2 trial in β-thalassemia
At ASH 2014, researchers presented results of a phase 2 trial in which they investigated whether luspatercept could increase hemoglobin levels and decrease transfusion dependence.
The goal was to increase hemoglobin levels 1.5 g/dL or more for at least 2 weeks in NTD patients and decrease the transfusion burden by 20% or more over 12 weeks in TD patients.
Thirty patients, 7 TD and 23 NTD, received an injection of luspatercept every 3 weeks for 3 months at doses ranging from 0.2 mg/kg to 1.0 mg/kg.
Three-quarters of patients who received 0.8 mg/kg to 1.0 mg/kg of luspatercept experienced an increase in their hemoglobin levels or a reduction in their transfusion burden.
Of the NTD patients, 8 of 12 with iron overload at baseline experienced a reduction in liver iron concentration of 1 mg or more at 16 weeks.
TD patients had a more than 60% reduction in transfusion burden over 12 weeks. This included 2 patients with β0 β0 genotype, who experienced a 79% and 75% reduction.
Luspatercept did not cause any treatment-related serious or severe adverse events. The most common adverse events were bone pain (20%), headache (17%), myalgia (13%), and asthenia (10%).
There was 1 grade 3 dose-limiting toxicity of worsening lumbar spine bone pain. And 3 patients discontinued treatment early, 1 each with occipital headache, ankle pain, and back pain.