User login
Is your patient’s cannabis use problematic?
CASE
Jessica F is a new 23-year-old patient at your clinic who is seeing you to discuss her severe anxiety. She also has asthma and reports during your exploration of her family history that her father has been diagnosed with schizophrenia. She has been using 3 cartridges of cannabis vape daily to help “calm her mind” but has never tried other psychotropic medications and has never been referred to a psychiatrist.
How would you proceed with this patient?
Despite emerging evidence of the harmful effects of cannabis consumption, public perception of harm has steadily declined over the past 10 years.1,2 More adults are using cannabis than before and using it more frequently. Among primary care patients who consume cannabis recreationally, about half report less than monthly consumption; 15% use it weekly, and 20% daily.3 The potency of cannabis products has also increased. In the past 2 decades, the average tetrahydrocannabinol (THC) content of recreational cannabis rose from 3% to 19%, and high-THC content delivery modalities such as vaporizer pens (“vapes”) were introduced.4,5
Health hazards of cannabis use include gastrointestinal dysfunction (eg, cannabinoid hyperemesis syndrome), acute psychosis or exacerbation of an existing mood, anxiety, or psychotic disorder, and cardiovascular sequelae such as myocardial infarction or dysrhythmia.6 Potential long-term effects include neurocognitive impairment among adolescents who use cannabis,7-9 worse outcomes in anxiety and mood disorders,10 schizophrenia,11 cardiovascular sequelae,12 chronic bronchitis,13 negative impact on reproductive function,14 and poor birth outcomes.15-17
Hidden in plain sight. Many patients who use cannabis report that their primary care physicians are unaware of their cannabis consumption.18 Inadequate screening for cannabis can be attributed to time constraints, inconsistent definitions for problematic or risky cannabis use, and lack of guidance.19,20 This article offers a more inclusive definition of “problematic cannabis use,” presents an up-to-date framework for evaluating it in the outpatient setting, and outlines potential interventions.

Your patient doesn’t meetthe DSM criteria, but …
Although it is important to identify cannabis use disorder (CUD) as defined in the Diagnostic and Statistical Manual of Mental Disorders, 5th edition (DSM-5; TABLE 121,22), consider also the immediate and long-term consequences of cannabis use for individuals who do not meet criteria for CUD. “Problematic cannabis use,” as we define it, may also involve (a) high-risk behaviors or (b) contraindicating medical or psychiatric comorbidities (TABLE 26-9).

CASE
The patient in our case exhibited
Continue to: Guidelines for screening and evaluation
Guidelines for screening and evaluation
All primary care patients should be screened for problematic cannabis use, but especially teenagers, young adults, pregnant women, and patients with a mental health or substance use history. A variation of the single question used to screen for alcohol use disorder can be applied to cannabis use.23 We recommend asking the initial question, “Over the past month, how many days a week on average have you used cannabis and products that contain THC?” Although some guidelines emphasize frequency of cannabis use when identifying problematic consumption,24,25 duration of behavior and content of THC are also important indicators.19 Inquire about cannabis consumption over 1 month to differentiate sporadic use from longstanding persistent use.
Explore what types of cannabis the patient is ingesting and whether the patient uses cannabis heavily (4 or more times a week on average). Also determine the method of ingestion (eg, eating, vaping, smoking), THC-content (%, if known), and estimated weight of daily cannabis use in grams (TABLE 326). Although patients may not always be able to provide accurate answers, you can gain a sense of the quantity and forms of cannabis a patient is ingesting to inform future conversations on risk and harm reduction.27

Assess a patient’s risk for harm
Cannabis use has the potential to cause immediate harm (linked to a single event of problematic cannabis use) and long-term harm (linked to a recurring pattern of problematic consumption). Cannabis can be especially harmful for patients with the following medical comorbidities or psychosocial factors, and should be avoided.
Cardiovascular disease. Cannabis is associated with an elevated risk for acute coronary syndrome and cardiovascular disease.28 Long-term cannabis use is linked to increased frequency of anginal events, development of cardiac arrhythmias, peripheral arteritis, coronary vasospasms, and problems with platelet aggregation.29,30 Strongly caution against cannabis use with patients who have a history of cardiovascular disease, orthostatic hypotension, tachyarrhythmia, or hypertension.
Pulmonary disease. Patients with pulmonary disease such as asthma may find cannabis helpful as a short-term bronchodilator.31 However, for patients with underlying pulmonary disease who also smoke cigarettes, strongly discourage the smoking of cannabis or hashish, as that may worsen asthma symptoms,32 increase risk of chronic bronchitis,33 and increase cough, sputum production, and wheezing.31 There is currently insufficient evidence to suggest a positive association between cannabis use and the development of chronic obstructive pulmonary disease.34
Continue to: Family history of psychotic disorders
Family history of psychotic disorders. Cannabis is associated with a dose-dependent risk of schizophrenia, which is especially pronounced in patients with a family history of schizophrenia.35 Among patients with a history of psychosis, heavy cannabis use has been associated with increased hospitalizations, increased positive symptoms, and more frequent relapses.36-38
Pregnancy, current or planned. Some women turn to cannabis during pregnancy due to its antiemetic properties. However, perinatal exposure to cannabis is associated with significant risk to the offspring. Maternal cannabis use during the first and second trimesters of pregnancy is associated with decreased performance of the child on measures of function at 3 years of age.39 In addition, cannabis consumption during pregnancy is linked to increased frequency of childhood behavioral issues, inattention, hyperactivity, and impulsivity.40 Peripartum cannabis exposure can affect birth outcomes and is correlated with lower birth weight, incidence of preterm labor, and neonatal intensive care unit admission.15-17,41 Of note, the THC concentration in breast milk peaks at 1 hour after the nursing mother inhales cannabis and typically dissipates after 4 hours.42
Age < 25 years. Chronic heavy use of cannabis in those younger than 25 is associated with higher likelihood of developing CUD, lower IQ,9 lower level of educational attainment, lower income,43 and decreased executive function.8
Substance use disorder history. Recreational cannabis use can hinder recovery from other substance use disorders.44
Consider these 5 interventions
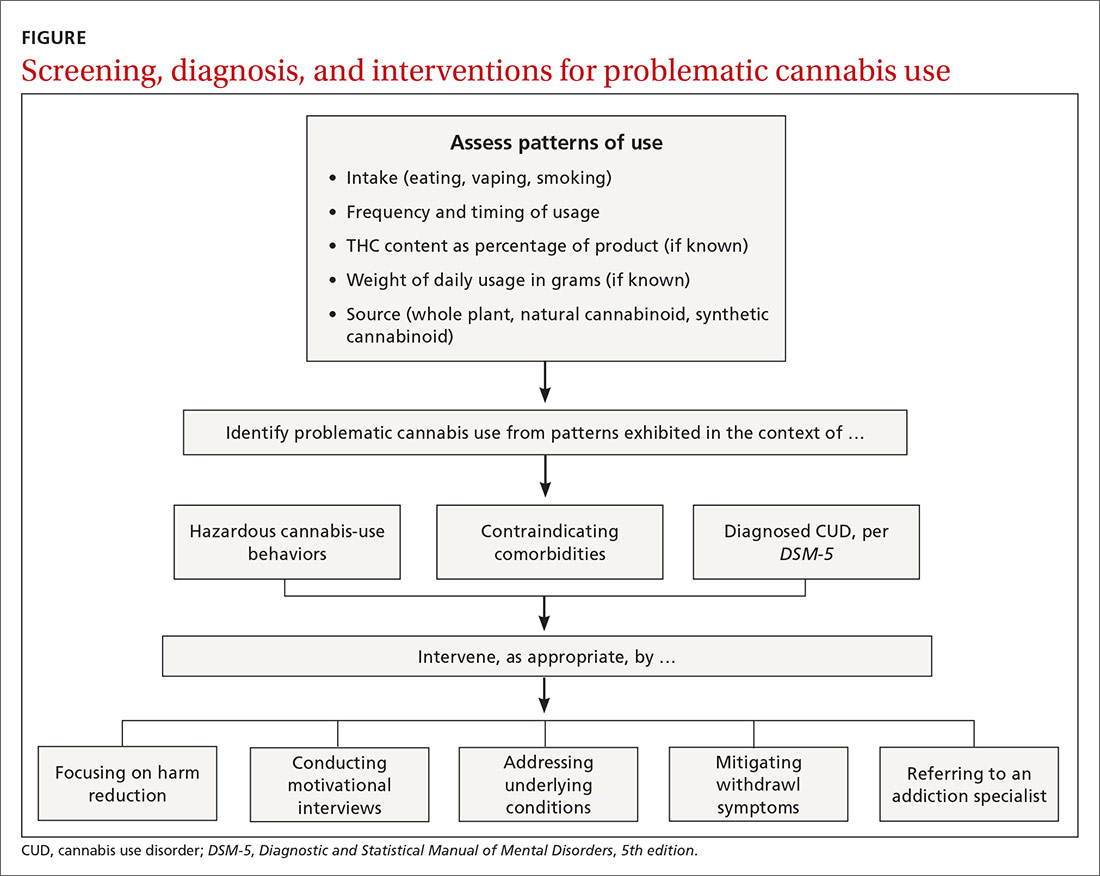
Physicians can address problematic cannabis use with a 5-pronged approach: (1) harm reduction, (2) motivational interviewing, (3) addressing underlying conditions, (4) mitigating withdrawal symptoms, and (5) referring to an addiction specialist (FIGURE).
Continue to: Harm reduction
Harm reduction
Harm reduction applies to all individuals who use cannabis but especially to problematic cannabis users. Ask users to abstain from cannabis for limited periods of time to see how such abstinence affects other areas of their life. While abstinence is a goal, be prepared to perform non-abstinence-based interventions. The goal of harm reduction is to encourage behaviors that minimize health risks to which cannabis users are exposed. Encourage patients to:
Abstain from driving while intoxicated. Cannabis use while driving slows reaction time,45 impairs road tracking (driving with correct road position),46 increases weaving,47 and causes a loss of anticipatory reactions learned in driving practice.48 Risk of crashing is significantly increased with elevated levels of THC, and driving within 1 hour of cannabis ingestion nearly doubles the risk of a crash.49-51
Abstain from vaping THC-containing products. The Centers for Disease Control and Prevention recommends that patients minimize the use of THC-containing e-cigarette or vaping products in light of the thousands of reports in the United States of product-associated lung injury, which in some cases have led to death.52
Clarify serving sizes and recognize delayed effects. Inexperienced cannabis users often are confused by recommended serving sizes for edible cannabis products. A typical cannabis-infused brownie may contain 100 mg of THC when the recommended serving size typically is 10 mg. THC content is included on the label of cannabis edibles purchased in state-regulated stores; these products are tested regularly in laboratories designated by the state.
Due to the delayed onset of THC’s effect, there have been numerous cases of patients taking a higher-than-intended dose of edible cannabis that caused acute intoxication and psychomedical sequelae leading to emergency hospital visits and, in some cases, death.6,53 Individuals should start at a low dose and gradually work up to a higher dose as tolerated. Patients naïve to cannabis should be especially cautious when ingesting edible products.
Continue to: Abstain from cannabis with high THC content
Abstain from cannabis with high THC content. High-potency cannabis (> 10% THC) is associated with earlier onset of first-episode psychosis.54,55
Motivational interviewing
Motivational interviewing (MI) is a psychosocial approach that emphasizes a patient’s self-efficacy and an interviewer’s positive feedback to collaboratively address substance use.56 MI can be performed in short, discrete sessions. Such interventions can reduce the average number of days of cannabis use. One large-scale Cochrane review found that cognitive behavioral therapy (CBT), motivational enhancement therapy, or the 2 therapies combined most consistently reduced the frequency of cannabis use reported by patients at early follow-up.57
Address underlying conditions
Some patients use cannabis to self-medicate for pain, insomnia, nausea, and anxiety. Identify these conditions and address them with first-line pharmacologic or psychotherapeutic interventions when possible. This is especially important for conditions in which long-term cannabis use may adversely impact outcomes, such as in posttraumatic stress disorder, anxiety, and mood disorders.58-60 Little evidence exists for the use of cannabis as treatment of any primary psychiatric disorder.61,62 Family physicians who are uncomfortable treating a specific underlying condition can consult specialists in pain management, sleep medicine, psychiatry, and neurology.
Mitigate withdrawal symptoms
Discontinuation of cannabis use may lead to withdrawal symptoms such as waxing and waning irritability, restlessness, sweating, aggression, anxiety, depressed mood, sleep disturbance, or changes in appetite.63,64 These symptoms typically emerge within the first couple days of abstinence and can last up to 28 days.63,64 Although the US Food and Drug Administration has not approved any medications for CUD treatment, and there are no established protocols for detoxification, there is evidence that CBT or medications such as gabapentin or zolpidem can reduce the intensity of withdrawal symptoms.65,66
Refer to an addiction specialist
Consider referring patients with problematic cannabis use to an addiction specialist with expertise in psychopharmacologic and psychotherapeutic approaches to managing substance use.
Continue to: CASE
CASE
You renew Ms. F’s asthma medications, discuss her cannabis use, start her on a selective serotonin reuptake inhibitor, and refer her to an outpatient psychiatrist. Over the next few weeks, you and the outpatient psychiatrist employ brief motivational interviewing around cannabis use, and you provide psychoeducation around potential harms of use when driving and in light of the patient’s asthma.
The patient’s anxiety symptoms decrease with up-titration of the SSRI by the outpatient psychiatrist and with enrollment in individual CBT. She is slowly able to taper off cannabis vaping with continued motivational interviewing and encouragement, despite withdrawal-induced anxiety and sleep disturbance.
CORRESPONDENCE
Michael Hsu, MD, Brigham & Women’s Hospital, 75 Francis Street, Boston, MA 02215; [email protected].
1. Sarvet AL, Wall MM, Keyes KM, et al. Recent rapid decrease in adolescents’ perception that marijuana is harmful, but no concurrent increase in use. Drug Alcohol Depend. 2018;186:68-74.
2. Compton WM, Han B, Jones CM, Blanco C, Hughes A. Marijuana use and use disorders in adults in the USA, 2002-14: analysis of annual cross-sectional surveys. Lancet Psychiatry. 2016;3:954-964.
3. Lapham GT, Lee AK, Caldeiro RM, et al. Frequency of cannabis use among primary care patients in Washington state. J Am Board Fam Med. 2017;30:795‐805.
4. Chandra S, Radwan MM, Majumdar CG, et al. New trends in cannabis potency in USA and Europe during the last decade (2008-2017). Eur Arch Psychiatry Clin Neurosci. 2019;269:5-15.
5. Sevigny EL, Pacula RL, Heaton P. The effects of medical marijuana laws on potency. Int J Drug Policy. 2014;25:308-319.
6. Monte AA, Shelton SK, Mills E, et al. Acute illness associated with cannabis use, by route of exposure: an observational study. Ann Intern Med. 2019;170:531-537.
7. Scott JC, Slomiak ST, Jones JD, et al. Association of cannabis with cognitive functioning in adolescents and young adults: a systematic review and meta-analysis. JAMA Psychiatry. 2018;75:585-595.
8. Gruber SA, Sagar KA, Dahlgren MK, et al. Age of onset of marijuana use and executive function. Psychol Addict Behav. 2012;26:496-506.
9. Meier MH, Caspi A, Ambler A, et al. Persistent cannabis users show neuropsychological decline from childhood to midlife. Proc Natl Acad Sci U S A. 2012;109:E2657-E2664.
10. Mammen G, Rueda S, Roerecke M, et al. Association of cannabis with long-term clinical symptoms in anxiety and mood disorders: a systematic review of prospective studies. J Clin Psychiatry. 2018;79:17r11839.
11. Gage SH, Hickman M, Zammit S. Association between cannabis and psychosis: epidemiologic evidence. Biol Psychiatry. 2016;79:549-556.
12. Singh A, Saluja S, Kumar A, et al. Cardiovascular complications of marijuana and related substances: a review. Cardiol Ther. 2018;7:45-59.
13. Volkow ND, Compton WM, Weiss SR. Adverse health effects of marijuana use. N Engl J Med. 2014;370:2219-2227.
14. Bari M, Battista N, Pirazzi V, et al. The manifold actions of endocannabinoids on female and male reproductive events. Front Biosci (Landmark Ed). 2011;16:498-516.
15. Hayatbakhsh MR, Flenady VJ, Gibbons KS, et al. Birth outcomes associated with cannabis use before and during pregnancy. Pediatr Res. 2012;71:215-219.
16. Saurel-Cubizolles M-J, Prunet C, Blondel B. Cannabis use during pregnancy in France in 2010. BJOG. 2014;121:971-977.
17. Prunet C, Delnord M, Saurel-Cubizolles M-J, et al. Risk factors of preterm birth in France in 2010 and changes since 1995: results from the French national perinatal surveys. J Gynecol Obstet Hum Reprod. 2017;46:19-28.
18. Kondrad EC, Reed AJ, Simpson MJ, et al. Lack of communication about medical marijuana use between doctors and their patients. J Am Board Fam Med. 2018;31:805-808.
19. Casajuana C, López-Pelayo H, Balcells MM, et al. Definitions of risky and problematic cannabis use: a systematic review. Subst Use Misuse. 2016;51:1760-1770.
20. Norberg MM, Gates P, Dillon P, et al. Screening and managing cannabis use: comparing GP’s and nurses’ knowledge, beliefs, and behavior. Subst Abuse Treat Prev Policy. 2012;7:31.
21. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Washington DC: APA Publishing; 2013:509-516.
22. Hasin DS, Saha TD, Kerridge BT, et al. Prevalence of marijuana use disorders in the United States between 2001-2002 and 2012-2013. JAMA Psychiatry. 2015;72:1235-1242.
23. Smith PC, Schmidt SM, Allensworth-Davies D, et al. A single-question screening test for drug use in primary care. Arch Intern Med. 2010;170:1155-1160.
24. Fischer B, Jones W, Shuper P, et al. 12-month follow-up of an exploratory ‘brief intervention’ for high-frequency cannabis users among Canadian university students. Subst Abuse Treat Prev Policy. 2012;7:15.
25. Turner SD, Spithoff S, Kahan M. Approach to cannabis use disorder in primary care: focus on youth and other high-risk users. Can Fam Physician. 2014;60:801-808.
26. Smart R, Caulkins JP, Kilmer B, et al. Variation in cannabis potency & prices in a newly-legal market: evidence from 30 million cannabis sales in Washington State. Addiction. 2017;112:2167-2177.
27. Bonn-Miller MO, Loflin MJE, Thomas BF, et al. Labeling accuracy of cannabidiol extracts sold online. JAMA. 2017;318:1708-1709.
28. Richards JR, Bing ML, Moulin AK, et al. Cannabis use and acute coronary syndrome. Clin Toxicol (Phila). 2019;57:831-841.
29. Subramaniam VN, Menezes AR, DeSchutter A, et al. The cardiovascular effects of marijuana: are the potential adverse effects worth the high? Mo Med. 2019;116:146-153.
30. Jones RT. Cardiovascular system effects of marijuana. J Clin Pharmacol. 2002;42:58S-63S.
31. Tetrault JM, Crothers K, Moore BA, et al. Effects of marijuana smoking on pulmonary function and respiratory complications: a systematic review. Arch Intern Med. 2007;167:221-228.
32. Bramness JG, von Soest T. A longitudinal study of cannabis use increasing the use of asthma medication in young Norwegian adults. BMC Pulm Med. 2019;19:52.
33. Moore BA, Augustson EM, Moser RP, et al. Respiratory effects of marijuana and tobacco use in a U.S. sample. J Gen Intern Med. 2005;20:33-37.
34. Tashkin DP. Does marijuana pose risks for chronic airflow obstruction? Ann Am Thorac Soc. 2015;12:235-236.
35. McGuire PK, Jones P, Harvey I, et al. Morbid risk of schizophrenia for relatives of patients with cannabis-associated psychosis. Schizophr Res. 1995;15:277-281.
36. Hall W, Degenhardt L. Cannabis use and the risk of developing a psychotic disorder. World Psychiatry. 2008;7:68-71.
37. Gerlach J, Koret B, Gereš N, et al. Clinical challenges in patients with first episode psychosis and cannabis use: mini-review and a case study. Psychiatr Danub. 2019;31(suppl 2):162-170.
38. Patel R, Wilson R, Jackson R, et al. Association of cannabis use with hospital admission and antipsychotic treatment failure in first episode psychosis: an observational study. BMJ Open. 2016;6:e009888.
39. Day NL, Richardson GA, Goldschmidt L, et al. Effect of prenatal marijuana exposure on the cognitive development of offspring at age three. Neurotoxicol Teratol. 1994;16:169-175.
40. Goldschmidt L, Day NL, Richardson GA. Effects of prenatal marijuana exposure on child behavior problems at age 10. Neurotoxicol Teratol. 2000;22:325-336.
41. Corsi DJ, Walsh L, Weiss D, et al. Association between self-reported prenatal cannabis use and maternal, perinatal, and neonatal outcomes. JAMA. 2019;322:145-152.
42. Baker T, Datta P, Rewers-Felkins K, et al. Transfer of inhaled cannabis into human breast milk. Obstet Gynecol. 2018;131:783-788.
43. Thompson K, Leadbeater B, Ames M, et al. Associations between marijuana use trajectories and educational and occupational success in young adulthood. Prev Sci. 2019;20:257-269.
44. Yuan M, Kanellopoulos T, Kotbi N. Cannabis use and psychiatric illness in the context of medical marijuana legalization: a clinical perspective. Gen Hosp Psychiatry. 2019;61:82-83.
45. Ronen A, Gershon P, Drobiner H, et al. Effects of THC on driving performance, physiological state and subjective feelings relative to alcohol. Accid Anal Prev. 2008;40:926-934.
46. Robbe H. Marijuana’s impairing effects on driving are moderate when taken alone but severe when combined with alcohol. Hum Psychopharmacol Clin Exp. 1998;13(suppl 2):S70-S78.
47. Lenné MG, Dietze PM, Triggs TJ, et al. The effects of cannabis and alcohol on simulated arterial driving: influences of driving experience and task demand. Accid Anal Prev. 2010;42:859-866.
48. Anderson BM, Rizzo M, Block RI, et al. Sex differences in the effects of marijuana on simulated driving performance. J Psychoactive Drugs. 2010;42:19-30.
49. Laumon B, Gadegbeku B, Martin J-L, Biecheler M-B. Cannabis intoxication and fatal road crashes in France: population based case-control study. BMJ. 2005;331:1371.
50. Asbridge M, Poulin C, Donato A. Motor vehicle collision risk and driving under the influence of cannabis: evidence from adolescents in Atlantic Canada. Accid Anal Prev. 2005;37:1025-1034.
51. Mann RE, Adlaf E, Zhao J, et al. Cannabis use and self-reported collisions in a representative sample of adult drivers. J Safety Res. 2007;38:669-674.
52. Taylor J, Wiens T, Peterson J, et al. Characteristics of e-cigarette, or vaping, products used by patients with associated lung injury and products seized by law enforcement—Minnesota, 2018 and 2019. MMWR Morb Mortal Wkly Rep. 2019;68:1096-1100.
53. Hancock-Allen JB, Barker L, VanDyke M, et al. Notes from the field: death following ingestion of an edible marijuana product—Colorado, March 2014. MMWR Morb Mortal Wkly Rep. 2015;64:771-772.
54. Murray RM, Quigley H, Quattrone D, et al. Traditional marijuana, high-potency cannabis and synthetic cannabinoids: increasing risk for psychosis. World Psychiatry. 2016;15:195-204.
55. Di Forti MD, Sallis H, Allegri F, et al. Daily use, especially of high-potency cannabis, drives the earlier onset of psychosis in cannabis users. Schizophr Bull. 2014;40:1509-1517.
56. Miller WR. Motivational interviewing: research, practice, and puzzles. Addict Behav. 1996;21:835-842.
57. Gates PJ, Sabioni P, Copeland J, et al. Psychosocial interventions for cannabis use disorder. Cochrane Database Syst Rev. 2016;(5):CD005336.
58. Wilkinson ST, Stefanovics E, Rosenheck RA. Marijuana use is associated with worse outcomes in symptom severity and violent behavior in patients with posttraumatic stress disorder. J Clin Psychiatry. 2015;76:1174-1180.
59. Cougle JR, Bonn-Miller MO, Vujanovic AA, et al. Posttraumatic stress disorder and cannabis use in a nationally representative sample. Psychol Addict Behav. 2011;25:554-558.
60. Johnson MJ, Pierce JD, Mavandadi S, et al. Mental health symptom severity in cannabis using and non-using veterans with probable PTSD. J Affect Disord. 2016;190:439-442.
61. Wilkinson ST, Radhakrishnan R, D’Souza DC. A systematic review of the evidence for medical marijuana in psychiatric indications. J Clin Psychiatry. 2016;77:1050-1064.
62. Black N, Stockings E, Campbell G, et al. Cannabinoids for the treatment of mental disorders and symptoms of mental disorders: a systematic review and meta-analysis. Lancet Psychiatry. 2019;6:995-1010.
63. Bonnet U, Preuss U. The cannabis withdrawal syndrome: current insights. Subst Abuse Rehabil. 2017;8:9-37.
64. Vandrey R, Smith MT, McCann UD, et al. Sleep disturbance and the effects of extended-release zolpidem during cannabis withdrawal. Drug Alcohol Depend. 2011;117:38-44.
65. Mason BJ, Crean R, Goodell V, et al. A proof-of-concept randomized controlled study of gabapentin: effects on cannabis use, withdrawal and executive function deficits in cannabis-dependent adults. Neuropsychopharmacology. 2012;37:1689-1698.
66. Weinstein A, Miller H, Tal E, et al. Treatment of cannabis withdrawal syndrome using cognitive-behavioral therapy and relapse prevention for cannabis dependence. J Groups Addict Recover. 2010;5:240-263.
CASE
Jessica F is a new 23-year-old patient at your clinic who is seeing you to discuss her severe anxiety. She also has asthma and reports during your exploration of her family history that her father has been diagnosed with schizophrenia. She has been using 3 cartridges of cannabis vape daily to help “calm her mind” but has never tried other psychotropic medications and has never been referred to a psychiatrist.
How would you proceed with this patient?
Despite emerging evidence of the harmful effects of cannabis consumption, public perception of harm has steadily declined over the past 10 years.1,2 More adults are using cannabis than before and using it more frequently. Among primary care patients who consume cannabis recreationally, about half report less than monthly consumption; 15% use it weekly, and 20% daily.3 The potency of cannabis products has also increased. In the past 2 decades, the average tetrahydrocannabinol (THC) content of recreational cannabis rose from 3% to 19%, and high-THC content delivery modalities such as vaporizer pens (“vapes”) were introduced.4,5
Health hazards of cannabis use include gastrointestinal dysfunction (eg, cannabinoid hyperemesis syndrome), acute psychosis or exacerbation of an existing mood, anxiety, or psychotic disorder, and cardiovascular sequelae such as myocardial infarction or dysrhythmia.6 Potential long-term effects include neurocognitive impairment among adolescents who use cannabis,7-9 worse outcomes in anxiety and mood disorders,10 schizophrenia,11 cardiovascular sequelae,12 chronic bronchitis,13 negative impact on reproductive function,14 and poor birth outcomes.15-17
Hidden in plain sight. Many patients who use cannabis report that their primary care physicians are unaware of their cannabis consumption.18 Inadequate screening for cannabis can be attributed to time constraints, inconsistent definitions for problematic or risky cannabis use, and lack of guidance.19,20 This article offers a more inclusive definition of “problematic cannabis use,” presents an up-to-date framework for evaluating it in the outpatient setting, and outlines potential interventions.

Your patient doesn’t meetthe DSM criteria, but …
Although it is important to identify cannabis use disorder (CUD) as defined in the Diagnostic and Statistical Manual of Mental Disorders, 5th edition (DSM-5; TABLE 121,22), consider also the immediate and long-term consequences of cannabis use for individuals who do not meet criteria for CUD. “Problematic cannabis use,” as we define it, may also involve (a) high-risk behaviors or (b) contraindicating medical or psychiatric comorbidities (TABLE 26-9).

CASE
The patient in our case exhibited
Continue to: Guidelines for screening and evaluation
Guidelines for screening and evaluation
All primary care patients should be screened for problematic cannabis use, but especially teenagers, young adults, pregnant women, and patients with a mental health or substance use history. A variation of the single question used to screen for alcohol use disorder can be applied to cannabis use.23 We recommend asking the initial question, “Over the past month, how many days a week on average have you used cannabis and products that contain THC?” Although some guidelines emphasize frequency of cannabis use when identifying problematic consumption,24,25 duration of behavior and content of THC are also important indicators.19 Inquire about cannabis consumption over 1 month to differentiate sporadic use from longstanding persistent use.
Explore what types of cannabis the patient is ingesting and whether the patient uses cannabis heavily (4 or more times a week on average). Also determine the method of ingestion (eg, eating, vaping, smoking), THC-content (%, if known), and estimated weight of daily cannabis use in grams (TABLE 326). Although patients may not always be able to provide accurate answers, you can gain a sense of the quantity and forms of cannabis a patient is ingesting to inform future conversations on risk and harm reduction.27

Assess a patient’s risk for harm
Cannabis use has the potential to cause immediate harm (linked to a single event of problematic cannabis use) and long-term harm (linked to a recurring pattern of problematic consumption). Cannabis can be especially harmful for patients with the following medical comorbidities or psychosocial factors, and should be avoided.
Cardiovascular disease. Cannabis is associated with an elevated risk for acute coronary syndrome and cardiovascular disease.28 Long-term cannabis use is linked to increased frequency of anginal events, development of cardiac arrhythmias, peripheral arteritis, coronary vasospasms, and problems with platelet aggregation.29,30 Strongly caution against cannabis use with patients who have a history of cardiovascular disease, orthostatic hypotension, tachyarrhythmia, or hypertension.
Pulmonary disease. Patients with pulmonary disease such as asthma may find cannabis helpful as a short-term bronchodilator.31 However, for patients with underlying pulmonary disease who also smoke cigarettes, strongly discourage the smoking of cannabis or hashish, as that may worsen asthma symptoms,32 increase risk of chronic bronchitis,33 and increase cough, sputum production, and wheezing.31 There is currently insufficient evidence to suggest a positive association between cannabis use and the development of chronic obstructive pulmonary disease.34
Continue to: Family history of psychotic disorders
Family history of psychotic disorders. Cannabis is associated with a dose-dependent risk of schizophrenia, which is especially pronounced in patients with a family history of schizophrenia.35 Among patients with a history of psychosis, heavy cannabis use has been associated with increased hospitalizations, increased positive symptoms, and more frequent relapses.36-38
Pregnancy, current or planned. Some women turn to cannabis during pregnancy due to its antiemetic properties. However, perinatal exposure to cannabis is associated with significant risk to the offspring. Maternal cannabis use during the first and second trimesters of pregnancy is associated with decreased performance of the child on measures of function at 3 years of age.39 In addition, cannabis consumption during pregnancy is linked to increased frequency of childhood behavioral issues, inattention, hyperactivity, and impulsivity.40 Peripartum cannabis exposure can affect birth outcomes and is correlated with lower birth weight, incidence of preterm labor, and neonatal intensive care unit admission.15-17,41 Of note, the THC concentration in breast milk peaks at 1 hour after the nursing mother inhales cannabis and typically dissipates after 4 hours.42
Age < 25 years. Chronic heavy use of cannabis in those younger than 25 is associated with higher likelihood of developing CUD, lower IQ,9 lower level of educational attainment, lower income,43 and decreased executive function.8
Substance use disorder history. Recreational cannabis use can hinder recovery from other substance use disorders.44
Consider these 5 interventions
Physicians can address problematic cannabis use with a 5-pronged approach: (1) harm reduction, (2) motivational interviewing, (3) addressing underlying conditions, (4) mitigating withdrawal symptoms, and (5) referring to an addiction specialist (FIGURE).

Continue to: Harm reduction
Harm reduction
Harm reduction applies to all individuals who use cannabis but especially to problematic cannabis users. Ask users to abstain from cannabis for limited periods of time to see how such abstinence affects other areas of their life. While abstinence is a goal, be prepared to perform non-abstinence-based interventions. The goal of harm reduction is to encourage behaviors that minimize health risks to which cannabis users are exposed. Encourage patients to:
Abstain from driving while intoxicated. Cannabis use while driving slows reaction time,45 impairs road tracking (driving with correct road position),46 increases weaving,47 and causes a loss of anticipatory reactions learned in driving practice.48 Risk of crashing is significantly increased with elevated levels of THC, and driving within 1 hour of cannabis ingestion nearly doubles the risk of a crash.49-51
Abstain from vaping THC-containing products. The Centers for Disease Control and Prevention recommends that patients minimize the use of THC-containing e-cigarette or vaping products in light of the thousands of reports in the United States of product-associated lung injury, which in some cases have led to death.52
Clarify serving sizes and recognize delayed effects. Inexperienced cannabis users often are confused by recommended serving sizes for edible cannabis products. A typical cannabis-infused brownie may contain 100 mg of THC when the recommended serving size typically is 10 mg. THC content is included on the label of cannabis edibles purchased in state-regulated stores; these products are tested regularly in laboratories designated by the state.
Due to the delayed onset of THC’s effect, there have been numerous cases of patients taking a higher-than-intended dose of edible cannabis that caused acute intoxication and psychomedical sequelae leading to emergency hospital visits and, in some cases, death.6,53 Individuals should start at a low dose and gradually work up to a higher dose as tolerated. Patients naïve to cannabis should be especially cautious when ingesting edible products.
Continue to: Abstain from cannabis with high THC content
Abstain from cannabis with high THC content. High-potency cannabis (> 10% THC) is associated with earlier onset of first-episode psychosis.54,55
Motivational interviewing
Motivational interviewing (MI) is a psychosocial approach that emphasizes a patient’s self-efficacy and an interviewer’s positive feedback to collaboratively address substance use.56 MI can be performed in short, discrete sessions. Such interventions can reduce the average number of days of cannabis use. One large-scale Cochrane review found that cognitive behavioral therapy (CBT), motivational enhancement therapy, or the 2 therapies combined most consistently reduced the frequency of cannabis use reported by patients at early follow-up.57
Address underlying conditions
Some patients use cannabis to self-medicate for pain, insomnia, nausea, and anxiety. Identify these conditions and address them with first-line pharmacologic or psychotherapeutic interventions when possible. This is especially important for conditions in which long-term cannabis use may adversely impact outcomes, such as in posttraumatic stress disorder, anxiety, and mood disorders.58-60 Little evidence exists for the use of cannabis as treatment of any primary psychiatric disorder.61,62 Family physicians who are uncomfortable treating a specific underlying condition can consult specialists in pain management, sleep medicine, psychiatry, and neurology.
Mitigate withdrawal symptoms
Discontinuation of cannabis use may lead to withdrawal symptoms such as waxing and waning irritability, restlessness, sweating, aggression, anxiety, depressed mood, sleep disturbance, or changes in appetite.63,64 These symptoms typically emerge within the first couple days of abstinence and can last up to 28 days.63,64 Although the US Food and Drug Administration has not approved any medications for CUD treatment, and there are no established protocols for detoxification, there is evidence that CBT or medications such as gabapentin or zolpidem can reduce the intensity of withdrawal symptoms.65,66
Refer to an addiction specialist
Consider referring patients with problematic cannabis use to an addiction specialist with expertise in psychopharmacologic and psychotherapeutic approaches to managing substance use.
Continue to: CASE
CASE
You renew Ms. F’s asthma medications, discuss her cannabis use, start her on a selective serotonin reuptake inhibitor, and refer her to an outpatient psychiatrist. Over the next few weeks, you and the outpatient psychiatrist employ brief motivational interviewing around cannabis use, and you provide psychoeducation around potential harms of use when driving and in light of the patient’s asthma.
The patient’s anxiety symptoms decrease with up-titration of the SSRI by the outpatient psychiatrist and with enrollment in individual CBT. She is slowly able to taper off cannabis vaping with continued motivational interviewing and encouragement, despite withdrawal-induced anxiety and sleep disturbance.
CORRESPONDENCE
Michael Hsu, MD, Brigham & Women’s Hospital, 75 Francis Street, Boston, MA 02215; [email protected].
CASE
Jessica F is a new 23-year-old patient at your clinic who is seeing you to discuss her severe anxiety. She also has asthma and reports during your exploration of her family history that her father has been diagnosed with schizophrenia. She has been using 3 cartridges of cannabis vape daily to help “calm her mind” but has never tried other psychotropic medications and has never been referred to a psychiatrist.
How would you proceed with this patient?
Despite emerging evidence of the harmful effects of cannabis consumption, public perception of harm has steadily declined over the past 10 years.1,2 More adults are using cannabis than before and using it more frequently. Among primary care patients who consume cannabis recreationally, about half report less than monthly consumption; 15% use it weekly, and 20% daily.3 The potency of cannabis products has also increased. In the past 2 decades, the average tetrahydrocannabinol (THC) content of recreational cannabis rose from 3% to 19%, and high-THC content delivery modalities such as vaporizer pens (“vapes”) were introduced.4,5
Health hazards of cannabis use include gastrointestinal dysfunction (eg, cannabinoid hyperemesis syndrome), acute psychosis or exacerbation of an existing mood, anxiety, or psychotic disorder, and cardiovascular sequelae such as myocardial infarction or dysrhythmia.6 Potential long-term effects include neurocognitive impairment among adolescents who use cannabis,7-9 worse outcomes in anxiety and mood disorders,10 schizophrenia,11 cardiovascular sequelae,12 chronic bronchitis,13 negative impact on reproductive function,14 and poor birth outcomes.15-17
Hidden in plain sight. Many patients who use cannabis report that their primary care physicians are unaware of their cannabis consumption.18 Inadequate screening for cannabis can be attributed to time constraints, inconsistent definitions for problematic or risky cannabis use, and lack of guidance.19,20 This article offers a more inclusive definition of “problematic cannabis use,” presents an up-to-date framework for evaluating it in the outpatient setting, and outlines potential interventions.

Your patient doesn’t meetthe DSM criteria, but …
Although it is important to identify cannabis use disorder (CUD) as defined in the Diagnostic and Statistical Manual of Mental Disorders, 5th edition (DSM-5; TABLE 121,22), consider also the immediate and long-term consequences of cannabis use for individuals who do not meet criteria for CUD. “Problematic cannabis use,” as we define it, may also involve (a) high-risk behaviors or (b) contraindicating medical or psychiatric comorbidities (TABLE 26-9).

CASE
The patient in our case exhibited
Continue to: Guidelines for screening and evaluation
Guidelines for screening and evaluation
All primary care patients should be screened for problematic cannabis use, but especially teenagers, young adults, pregnant women, and patients with a mental health or substance use history. A variation of the single question used to screen for alcohol use disorder can be applied to cannabis use.23 We recommend asking the initial question, “Over the past month, how many days a week on average have you used cannabis and products that contain THC?” Although some guidelines emphasize frequency of cannabis use when identifying problematic consumption,24,25 duration of behavior and content of THC are also important indicators.19 Inquire about cannabis consumption over 1 month to differentiate sporadic use from longstanding persistent use.
Explore what types of cannabis the patient is ingesting and whether the patient uses cannabis heavily (4 or more times a week on average). Also determine the method of ingestion (eg, eating, vaping, smoking), THC-content (%, if known), and estimated weight of daily cannabis use in grams (TABLE 326). Although patients may not always be able to provide accurate answers, you can gain a sense of the quantity and forms of cannabis a patient is ingesting to inform future conversations on risk and harm reduction.27

Assess a patient’s risk for harm
Cannabis use has the potential to cause immediate harm (linked to a single event of problematic cannabis use) and long-term harm (linked to a recurring pattern of problematic consumption). Cannabis can be especially harmful for patients with the following medical comorbidities or psychosocial factors, and should be avoided.
Cardiovascular disease. Cannabis is associated with an elevated risk for acute coronary syndrome and cardiovascular disease.28 Long-term cannabis use is linked to increased frequency of anginal events, development of cardiac arrhythmias, peripheral arteritis, coronary vasospasms, and problems with platelet aggregation.29,30 Strongly caution against cannabis use with patients who have a history of cardiovascular disease, orthostatic hypotension, tachyarrhythmia, or hypertension.
Pulmonary disease. Patients with pulmonary disease such as asthma may find cannabis helpful as a short-term bronchodilator.31 However, for patients with underlying pulmonary disease who also smoke cigarettes, strongly discourage the smoking of cannabis or hashish, as that may worsen asthma symptoms,32 increase risk of chronic bronchitis,33 and increase cough, sputum production, and wheezing.31 There is currently insufficient evidence to suggest a positive association between cannabis use and the development of chronic obstructive pulmonary disease.34
Continue to: Family history of psychotic disorders
Family history of psychotic disorders. Cannabis is associated with a dose-dependent risk of schizophrenia, which is especially pronounced in patients with a family history of schizophrenia.35 Among patients with a history of psychosis, heavy cannabis use has been associated with increased hospitalizations, increased positive symptoms, and more frequent relapses.36-38
Pregnancy, current or planned. Some women turn to cannabis during pregnancy due to its antiemetic properties. However, perinatal exposure to cannabis is associated with significant risk to the offspring. Maternal cannabis use during the first and second trimesters of pregnancy is associated with decreased performance of the child on measures of function at 3 years of age.39 In addition, cannabis consumption during pregnancy is linked to increased frequency of childhood behavioral issues, inattention, hyperactivity, and impulsivity.40 Peripartum cannabis exposure can affect birth outcomes and is correlated with lower birth weight, incidence of preterm labor, and neonatal intensive care unit admission.15-17,41 Of note, the THC concentration in breast milk peaks at 1 hour after the nursing mother inhales cannabis and typically dissipates after 4 hours.42
Age < 25 years. Chronic heavy use of cannabis in those younger than 25 is associated with higher likelihood of developing CUD, lower IQ,9 lower level of educational attainment, lower income,43 and decreased executive function.8
Substance use disorder history. Recreational cannabis use can hinder recovery from other substance use disorders.44
Consider these 5 interventions
Physicians can address problematic cannabis use with a 5-pronged approach: (1) harm reduction, (2) motivational interviewing, (3) addressing underlying conditions, (4) mitigating withdrawal symptoms, and (5) referring to an addiction specialist (FIGURE).

Continue to: Harm reduction
Harm reduction
Harm reduction applies to all individuals who use cannabis but especially to problematic cannabis users. Ask users to abstain from cannabis for limited periods of time to see how such abstinence affects other areas of their life. While abstinence is a goal, be prepared to perform non-abstinence-based interventions. The goal of harm reduction is to encourage behaviors that minimize health risks to which cannabis users are exposed. Encourage patients to:
Abstain from driving while intoxicated. Cannabis use while driving slows reaction time,45 impairs road tracking (driving with correct road position),46 increases weaving,47 and causes a loss of anticipatory reactions learned in driving practice.48 Risk of crashing is significantly increased with elevated levels of THC, and driving within 1 hour of cannabis ingestion nearly doubles the risk of a crash.49-51
Abstain from vaping THC-containing products. The Centers for Disease Control and Prevention recommends that patients minimize the use of THC-containing e-cigarette or vaping products in light of the thousands of reports in the United States of product-associated lung injury, which in some cases have led to death.52
Clarify serving sizes and recognize delayed effects. Inexperienced cannabis users often are confused by recommended serving sizes for edible cannabis products. A typical cannabis-infused brownie may contain 100 mg of THC when the recommended serving size typically is 10 mg. THC content is included on the label of cannabis edibles purchased in state-regulated stores; these products are tested regularly in laboratories designated by the state.
Due to the delayed onset of THC’s effect, there have been numerous cases of patients taking a higher-than-intended dose of edible cannabis that caused acute intoxication and psychomedical sequelae leading to emergency hospital visits and, in some cases, death.6,53 Individuals should start at a low dose and gradually work up to a higher dose as tolerated. Patients naïve to cannabis should be especially cautious when ingesting edible products.
Continue to: Abstain from cannabis with high THC content
Abstain from cannabis with high THC content. High-potency cannabis (> 10% THC) is associated with earlier onset of first-episode psychosis.54,55
Motivational interviewing
Motivational interviewing (MI) is a psychosocial approach that emphasizes a patient’s self-efficacy and an interviewer’s positive feedback to collaboratively address substance use.56 MI can be performed in short, discrete sessions. Such interventions can reduce the average number of days of cannabis use. One large-scale Cochrane review found that cognitive behavioral therapy (CBT), motivational enhancement therapy, or the 2 therapies combined most consistently reduced the frequency of cannabis use reported by patients at early follow-up.57
Address underlying conditions
Some patients use cannabis to self-medicate for pain, insomnia, nausea, and anxiety. Identify these conditions and address them with first-line pharmacologic or psychotherapeutic interventions when possible. This is especially important for conditions in which long-term cannabis use may adversely impact outcomes, such as in posttraumatic stress disorder, anxiety, and mood disorders.58-60 Little evidence exists for the use of cannabis as treatment of any primary psychiatric disorder.61,62 Family physicians who are uncomfortable treating a specific underlying condition can consult specialists in pain management, sleep medicine, psychiatry, and neurology.
Mitigate withdrawal symptoms
Discontinuation of cannabis use may lead to withdrawal symptoms such as waxing and waning irritability, restlessness, sweating, aggression, anxiety, depressed mood, sleep disturbance, or changes in appetite.63,64 These symptoms typically emerge within the first couple days of abstinence and can last up to 28 days.63,64 Although the US Food and Drug Administration has not approved any medications for CUD treatment, and there are no established protocols for detoxification, there is evidence that CBT or medications such as gabapentin or zolpidem can reduce the intensity of withdrawal symptoms.65,66
Refer to an addiction specialist
Consider referring patients with problematic cannabis use to an addiction specialist with expertise in psychopharmacologic and psychotherapeutic approaches to managing substance use.
Continue to: CASE
CASE
You renew Ms. F’s asthma medications, discuss her cannabis use, start her on a selective serotonin reuptake inhibitor, and refer her to an outpatient psychiatrist. Over the next few weeks, you and the outpatient psychiatrist employ brief motivational interviewing around cannabis use, and you provide psychoeducation around potential harms of use when driving and in light of the patient’s asthma.
The patient’s anxiety symptoms decrease with up-titration of the SSRI by the outpatient psychiatrist and with enrollment in individual CBT. She is slowly able to taper off cannabis vaping with continued motivational interviewing and encouragement, despite withdrawal-induced anxiety and sleep disturbance.
CORRESPONDENCE
Michael Hsu, MD, Brigham & Women’s Hospital, 75 Francis Street, Boston, MA 02215; [email protected].
1. Sarvet AL, Wall MM, Keyes KM, et al. Recent rapid decrease in adolescents’ perception that marijuana is harmful, but no concurrent increase in use. Drug Alcohol Depend. 2018;186:68-74.
2. Compton WM, Han B, Jones CM, Blanco C, Hughes A. Marijuana use and use disorders in adults in the USA, 2002-14: analysis of annual cross-sectional surveys. Lancet Psychiatry. 2016;3:954-964.
3. Lapham GT, Lee AK, Caldeiro RM, et al. Frequency of cannabis use among primary care patients in Washington state. J Am Board Fam Med. 2017;30:795‐805.
4. Chandra S, Radwan MM, Majumdar CG, et al. New trends in cannabis potency in USA and Europe during the last decade (2008-2017). Eur Arch Psychiatry Clin Neurosci. 2019;269:5-15.
5. Sevigny EL, Pacula RL, Heaton P. The effects of medical marijuana laws on potency. Int J Drug Policy. 2014;25:308-319.
6. Monte AA, Shelton SK, Mills E, et al. Acute illness associated with cannabis use, by route of exposure: an observational study. Ann Intern Med. 2019;170:531-537.
7. Scott JC, Slomiak ST, Jones JD, et al. Association of cannabis with cognitive functioning in adolescents and young adults: a systematic review and meta-analysis. JAMA Psychiatry. 2018;75:585-595.
8. Gruber SA, Sagar KA, Dahlgren MK, et al. Age of onset of marijuana use and executive function. Psychol Addict Behav. 2012;26:496-506.
9. Meier MH, Caspi A, Ambler A, et al. Persistent cannabis users show neuropsychological decline from childhood to midlife. Proc Natl Acad Sci U S A. 2012;109:E2657-E2664.
10. Mammen G, Rueda S, Roerecke M, et al. Association of cannabis with long-term clinical symptoms in anxiety and mood disorders: a systematic review of prospective studies. J Clin Psychiatry. 2018;79:17r11839.
11. Gage SH, Hickman M, Zammit S. Association between cannabis and psychosis: epidemiologic evidence. Biol Psychiatry. 2016;79:549-556.
12. Singh A, Saluja S, Kumar A, et al. Cardiovascular complications of marijuana and related substances: a review. Cardiol Ther. 2018;7:45-59.
13. Volkow ND, Compton WM, Weiss SR. Adverse health effects of marijuana use. N Engl J Med. 2014;370:2219-2227.
14. Bari M, Battista N, Pirazzi V, et al. The manifold actions of endocannabinoids on female and male reproductive events. Front Biosci (Landmark Ed). 2011;16:498-516.
15. Hayatbakhsh MR, Flenady VJ, Gibbons KS, et al. Birth outcomes associated with cannabis use before and during pregnancy. Pediatr Res. 2012;71:215-219.
16. Saurel-Cubizolles M-J, Prunet C, Blondel B. Cannabis use during pregnancy in France in 2010. BJOG. 2014;121:971-977.
17. Prunet C, Delnord M, Saurel-Cubizolles M-J, et al. Risk factors of preterm birth in France in 2010 and changes since 1995: results from the French national perinatal surveys. J Gynecol Obstet Hum Reprod. 2017;46:19-28.
18. Kondrad EC, Reed AJ, Simpson MJ, et al. Lack of communication about medical marijuana use between doctors and their patients. J Am Board Fam Med. 2018;31:805-808.
19. Casajuana C, López-Pelayo H, Balcells MM, et al. Definitions of risky and problematic cannabis use: a systematic review. Subst Use Misuse. 2016;51:1760-1770.
20. Norberg MM, Gates P, Dillon P, et al. Screening and managing cannabis use: comparing GP’s and nurses’ knowledge, beliefs, and behavior. Subst Abuse Treat Prev Policy. 2012;7:31.
21. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Washington DC: APA Publishing; 2013:509-516.
22. Hasin DS, Saha TD, Kerridge BT, et al. Prevalence of marijuana use disorders in the United States between 2001-2002 and 2012-2013. JAMA Psychiatry. 2015;72:1235-1242.
23. Smith PC, Schmidt SM, Allensworth-Davies D, et al. A single-question screening test for drug use in primary care. Arch Intern Med. 2010;170:1155-1160.
24. Fischer B, Jones W, Shuper P, et al. 12-month follow-up of an exploratory ‘brief intervention’ for high-frequency cannabis users among Canadian university students. Subst Abuse Treat Prev Policy. 2012;7:15.
25. Turner SD, Spithoff S, Kahan M. Approach to cannabis use disorder in primary care: focus on youth and other high-risk users. Can Fam Physician. 2014;60:801-808.
26. Smart R, Caulkins JP, Kilmer B, et al. Variation in cannabis potency & prices in a newly-legal market: evidence from 30 million cannabis sales in Washington State. Addiction. 2017;112:2167-2177.
27. Bonn-Miller MO, Loflin MJE, Thomas BF, et al. Labeling accuracy of cannabidiol extracts sold online. JAMA. 2017;318:1708-1709.
28. Richards JR, Bing ML, Moulin AK, et al. Cannabis use and acute coronary syndrome. Clin Toxicol (Phila). 2019;57:831-841.
29. Subramaniam VN, Menezes AR, DeSchutter A, et al. The cardiovascular effects of marijuana: are the potential adverse effects worth the high? Mo Med. 2019;116:146-153.
30. Jones RT. Cardiovascular system effects of marijuana. J Clin Pharmacol. 2002;42:58S-63S.
31. Tetrault JM, Crothers K, Moore BA, et al. Effects of marijuana smoking on pulmonary function and respiratory complications: a systematic review. Arch Intern Med. 2007;167:221-228.
32. Bramness JG, von Soest T. A longitudinal study of cannabis use increasing the use of asthma medication in young Norwegian adults. BMC Pulm Med. 2019;19:52.
33. Moore BA, Augustson EM, Moser RP, et al. Respiratory effects of marijuana and tobacco use in a U.S. sample. J Gen Intern Med. 2005;20:33-37.
34. Tashkin DP. Does marijuana pose risks for chronic airflow obstruction? Ann Am Thorac Soc. 2015;12:235-236.
35. McGuire PK, Jones P, Harvey I, et al. Morbid risk of schizophrenia for relatives of patients with cannabis-associated psychosis. Schizophr Res. 1995;15:277-281.
36. Hall W, Degenhardt L. Cannabis use and the risk of developing a psychotic disorder. World Psychiatry. 2008;7:68-71.
37. Gerlach J, Koret B, Gereš N, et al. Clinical challenges in patients with first episode psychosis and cannabis use: mini-review and a case study. Psychiatr Danub. 2019;31(suppl 2):162-170.
38. Patel R, Wilson R, Jackson R, et al. Association of cannabis use with hospital admission and antipsychotic treatment failure in first episode psychosis: an observational study. BMJ Open. 2016;6:e009888.
39. Day NL, Richardson GA, Goldschmidt L, et al. Effect of prenatal marijuana exposure on the cognitive development of offspring at age three. Neurotoxicol Teratol. 1994;16:169-175.
40. Goldschmidt L, Day NL, Richardson GA. Effects of prenatal marijuana exposure on child behavior problems at age 10. Neurotoxicol Teratol. 2000;22:325-336.
41. Corsi DJ, Walsh L, Weiss D, et al. Association between self-reported prenatal cannabis use and maternal, perinatal, and neonatal outcomes. JAMA. 2019;322:145-152.
42. Baker T, Datta P, Rewers-Felkins K, et al. Transfer of inhaled cannabis into human breast milk. Obstet Gynecol. 2018;131:783-788.
43. Thompson K, Leadbeater B, Ames M, et al. Associations between marijuana use trajectories and educational and occupational success in young adulthood. Prev Sci. 2019;20:257-269.
44. Yuan M, Kanellopoulos T, Kotbi N. Cannabis use and psychiatric illness in the context of medical marijuana legalization: a clinical perspective. Gen Hosp Psychiatry. 2019;61:82-83.
45. Ronen A, Gershon P, Drobiner H, et al. Effects of THC on driving performance, physiological state and subjective feelings relative to alcohol. Accid Anal Prev. 2008;40:926-934.
46. Robbe H. Marijuana’s impairing effects on driving are moderate when taken alone but severe when combined with alcohol. Hum Psychopharmacol Clin Exp. 1998;13(suppl 2):S70-S78.
47. Lenné MG, Dietze PM, Triggs TJ, et al. The effects of cannabis and alcohol on simulated arterial driving: influences of driving experience and task demand. Accid Anal Prev. 2010;42:859-866.
48. Anderson BM, Rizzo M, Block RI, et al. Sex differences in the effects of marijuana on simulated driving performance. J Psychoactive Drugs. 2010;42:19-30.
49. Laumon B, Gadegbeku B, Martin J-L, Biecheler M-B. Cannabis intoxication and fatal road crashes in France: population based case-control study. BMJ. 2005;331:1371.
50. Asbridge M, Poulin C, Donato A. Motor vehicle collision risk and driving under the influence of cannabis: evidence from adolescents in Atlantic Canada. Accid Anal Prev. 2005;37:1025-1034.
51. Mann RE, Adlaf E, Zhao J, et al. Cannabis use and self-reported collisions in a representative sample of adult drivers. J Safety Res. 2007;38:669-674.
52. Taylor J, Wiens T, Peterson J, et al. Characteristics of e-cigarette, or vaping, products used by patients with associated lung injury and products seized by law enforcement—Minnesota, 2018 and 2019. MMWR Morb Mortal Wkly Rep. 2019;68:1096-1100.
53. Hancock-Allen JB, Barker L, VanDyke M, et al. Notes from the field: death following ingestion of an edible marijuana product—Colorado, March 2014. MMWR Morb Mortal Wkly Rep. 2015;64:771-772.
54. Murray RM, Quigley H, Quattrone D, et al. Traditional marijuana, high-potency cannabis and synthetic cannabinoids: increasing risk for psychosis. World Psychiatry. 2016;15:195-204.
55. Di Forti MD, Sallis H, Allegri F, et al. Daily use, especially of high-potency cannabis, drives the earlier onset of psychosis in cannabis users. Schizophr Bull. 2014;40:1509-1517.
56. Miller WR. Motivational interviewing: research, practice, and puzzles. Addict Behav. 1996;21:835-842.
57. Gates PJ, Sabioni P, Copeland J, et al. Psychosocial interventions for cannabis use disorder. Cochrane Database Syst Rev. 2016;(5):CD005336.
58. Wilkinson ST, Stefanovics E, Rosenheck RA. Marijuana use is associated with worse outcomes in symptom severity and violent behavior in patients with posttraumatic stress disorder. J Clin Psychiatry. 2015;76:1174-1180.
59. Cougle JR, Bonn-Miller MO, Vujanovic AA, et al. Posttraumatic stress disorder and cannabis use in a nationally representative sample. Psychol Addict Behav. 2011;25:554-558.
60. Johnson MJ, Pierce JD, Mavandadi S, et al. Mental health symptom severity in cannabis using and non-using veterans with probable PTSD. J Affect Disord. 2016;190:439-442.
61. Wilkinson ST, Radhakrishnan R, D’Souza DC. A systematic review of the evidence for medical marijuana in psychiatric indications. J Clin Psychiatry. 2016;77:1050-1064.
62. Black N, Stockings E, Campbell G, et al. Cannabinoids for the treatment of mental disorders and symptoms of mental disorders: a systematic review and meta-analysis. Lancet Psychiatry. 2019;6:995-1010.
63. Bonnet U, Preuss U. The cannabis withdrawal syndrome: current insights. Subst Abuse Rehabil. 2017;8:9-37.
64. Vandrey R, Smith MT, McCann UD, et al. Sleep disturbance and the effects of extended-release zolpidem during cannabis withdrawal. Drug Alcohol Depend. 2011;117:38-44.
65. Mason BJ, Crean R, Goodell V, et al. A proof-of-concept randomized controlled study of gabapentin: effects on cannabis use, withdrawal and executive function deficits in cannabis-dependent adults. Neuropsychopharmacology. 2012;37:1689-1698.
66. Weinstein A, Miller H, Tal E, et al. Treatment of cannabis withdrawal syndrome using cognitive-behavioral therapy and relapse prevention for cannabis dependence. J Groups Addict Recover. 2010;5:240-263.
1. Sarvet AL, Wall MM, Keyes KM, et al. Recent rapid decrease in adolescents’ perception that marijuana is harmful, but no concurrent increase in use. Drug Alcohol Depend. 2018;186:68-74.
2. Compton WM, Han B, Jones CM, Blanco C, Hughes A. Marijuana use and use disorders in adults in the USA, 2002-14: analysis of annual cross-sectional surveys. Lancet Psychiatry. 2016;3:954-964.
3. Lapham GT, Lee AK, Caldeiro RM, et al. Frequency of cannabis use among primary care patients in Washington state. J Am Board Fam Med. 2017;30:795‐805.
4. Chandra S, Radwan MM, Majumdar CG, et al. New trends in cannabis potency in USA and Europe during the last decade (2008-2017). Eur Arch Psychiatry Clin Neurosci. 2019;269:5-15.
5. Sevigny EL, Pacula RL, Heaton P. The effects of medical marijuana laws on potency. Int J Drug Policy. 2014;25:308-319.
6. Monte AA, Shelton SK, Mills E, et al. Acute illness associated with cannabis use, by route of exposure: an observational study. Ann Intern Med. 2019;170:531-537.
7. Scott JC, Slomiak ST, Jones JD, et al. Association of cannabis with cognitive functioning in adolescents and young adults: a systematic review and meta-analysis. JAMA Psychiatry. 2018;75:585-595.
8. Gruber SA, Sagar KA, Dahlgren MK, et al. Age of onset of marijuana use and executive function. Psychol Addict Behav. 2012;26:496-506.
9. Meier MH, Caspi A, Ambler A, et al. Persistent cannabis users show neuropsychological decline from childhood to midlife. Proc Natl Acad Sci U S A. 2012;109:E2657-E2664.
10. Mammen G, Rueda S, Roerecke M, et al. Association of cannabis with long-term clinical symptoms in anxiety and mood disorders: a systematic review of prospective studies. J Clin Psychiatry. 2018;79:17r11839.
11. Gage SH, Hickman M, Zammit S. Association between cannabis and psychosis: epidemiologic evidence. Biol Psychiatry. 2016;79:549-556.
12. Singh A, Saluja S, Kumar A, et al. Cardiovascular complications of marijuana and related substances: a review. Cardiol Ther. 2018;7:45-59.
13. Volkow ND, Compton WM, Weiss SR. Adverse health effects of marijuana use. N Engl J Med. 2014;370:2219-2227.
14. Bari M, Battista N, Pirazzi V, et al. The manifold actions of endocannabinoids on female and male reproductive events. Front Biosci (Landmark Ed). 2011;16:498-516.
15. Hayatbakhsh MR, Flenady VJ, Gibbons KS, et al. Birth outcomes associated with cannabis use before and during pregnancy. Pediatr Res. 2012;71:215-219.
16. Saurel-Cubizolles M-J, Prunet C, Blondel B. Cannabis use during pregnancy in France in 2010. BJOG. 2014;121:971-977.
17. Prunet C, Delnord M, Saurel-Cubizolles M-J, et al. Risk factors of preterm birth in France in 2010 and changes since 1995: results from the French national perinatal surveys. J Gynecol Obstet Hum Reprod. 2017;46:19-28.
18. Kondrad EC, Reed AJ, Simpson MJ, et al. Lack of communication about medical marijuana use between doctors and their patients. J Am Board Fam Med. 2018;31:805-808.
19. Casajuana C, López-Pelayo H, Balcells MM, et al. Definitions of risky and problematic cannabis use: a systematic review. Subst Use Misuse. 2016;51:1760-1770.
20. Norberg MM, Gates P, Dillon P, et al. Screening and managing cannabis use: comparing GP’s and nurses’ knowledge, beliefs, and behavior. Subst Abuse Treat Prev Policy. 2012;7:31.
21. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Washington DC: APA Publishing; 2013:509-516.
22. Hasin DS, Saha TD, Kerridge BT, et al. Prevalence of marijuana use disorders in the United States between 2001-2002 and 2012-2013. JAMA Psychiatry. 2015;72:1235-1242.
23. Smith PC, Schmidt SM, Allensworth-Davies D, et al. A single-question screening test for drug use in primary care. Arch Intern Med. 2010;170:1155-1160.
24. Fischer B, Jones W, Shuper P, et al. 12-month follow-up of an exploratory ‘brief intervention’ for high-frequency cannabis users among Canadian university students. Subst Abuse Treat Prev Policy. 2012;7:15.
25. Turner SD, Spithoff S, Kahan M. Approach to cannabis use disorder in primary care: focus on youth and other high-risk users. Can Fam Physician. 2014;60:801-808.
26. Smart R, Caulkins JP, Kilmer B, et al. Variation in cannabis potency & prices in a newly-legal market: evidence from 30 million cannabis sales in Washington State. Addiction. 2017;112:2167-2177.
27. Bonn-Miller MO, Loflin MJE, Thomas BF, et al. Labeling accuracy of cannabidiol extracts sold online. JAMA. 2017;318:1708-1709.
28. Richards JR, Bing ML, Moulin AK, et al. Cannabis use and acute coronary syndrome. Clin Toxicol (Phila). 2019;57:831-841.
29. Subramaniam VN, Menezes AR, DeSchutter A, et al. The cardiovascular effects of marijuana: are the potential adverse effects worth the high? Mo Med. 2019;116:146-153.
30. Jones RT. Cardiovascular system effects of marijuana. J Clin Pharmacol. 2002;42:58S-63S.
31. Tetrault JM, Crothers K, Moore BA, et al. Effects of marijuana smoking on pulmonary function and respiratory complications: a systematic review. Arch Intern Med. 2007;167:221-228.
32. Bramness JG, von Soest T. A longitudinal study of cannabis use increasing the use of asthma medication in young Norwegian adults. BMC Pulm Med. 2019;19:52.
33. Moore BA, Augustson EM, Moser RP, et al. Respiratory effects of marijuana and tobacco use in a U.S. sample. J Gen Intern Med. 2005;20:33-37.
34. Tashkin DP. Does marijuana pose risks for chronic airflow obstruction? Ann Am Thorac Soc. 2015;12:235-236.
35. McGuire PK, Jones P, Harvey I, et al. Morbid risk of schizophrenia for relatives of patients with cannabis-associated psychosis. Schizophr Res. 1995;15:277-281.
36. Hall W, Degenhardt L. Cannabis use and the risk of developing a psychotic disorder. World Psychiatry. 2008;7:68-71.
37. Gerlach J, Koret B, Gereš N, et al. Clinical challenges in patients with first episode psychosis and cannabis use: mini-review and a case study. Psychiatr Danub. 2019;31(suppl 2):162-170.
38. Patel R, Wilson R, Jackson R, et al. Association of cannabis use with hospital admission and antipsychotic treatment failure in first episode psychosis: an observational study. BMJ Open. 2016;6:e009888.
39. Day NL, Richardson GA, Goldschmidt L, et al. Effect of prenatal marijuana exposure on the cognitive development of offspring at age three. Neurotoxicol Teratol. 1994;16:169-175.
40. Goldschmidt L, Day NL, Richardson GA. Effects of prenatal marijuana exposure on child behavior problems at age 10. Neurotoxicol Teratol. 2000;22:325-336.
41. Corsi DJ, Walsh L, Weiss D, et al. Association between self-reported prenatal cannabis use and maternal, perinatal, and neonatal outcomes. JAMA. 2019;322:145-152.
42. Baker T, Datta P, Rewers-Felkins K, et al. Transfer of inhaled cannabis into human breast milk. Obstet Gynecol. 2018;131:783-788.
43. Thompson K, Leadbeater B, Ames M, et al. Associations between marijuana use trajectories and educational and occupational success in young adulthood. Prev Sci. 2019;20:257-269.
44. Yuan M, Kanellopoulos T, Kotbi N. Cannabis use and psychiatric illness in the context of medical marijuana legalization: a clinical perspective. Gen Hosp Psychiatry. 2019;61:82-83.
45. Ronen A, Gershon P, Drobiner H, et al. Effects of THC on driving performance, physiological state and subjective feelings relative to alcohol. Accid Anal Prev. 2008;40:926-934.
46. Robbe H. Marijuana’s impairing effects on driving are moderate when taken alone but severe when combined with alcohol. Hum Psychopharmacol Clin Exp. 1998;13(suppl 2):S70-S78.
47. Lenné MG, Dietze PM, Triggs TJ, et al. The effects of cannabis and alcohol on simulated arterial driving: influences of driving experience and task demand. Accid Anal Prev. 2010;42:859-866.
48. Anderson BM, Rizzo M, Block RI, et al. Sex differences in the effects of marijuana on simulated driving performance. J Psychoactive Drugs. 2010;42:19-30.
49. Laumon B, Gadegbeku B, Martin J-L, Biecheler M-B. Cannabis intoxication and fatal road crashes in France: population based case-control study. BMJ. 2005;331:1371.
50. Asbridge M, Poulin C, Donato A. Motor vehicle collision risk and driving under the influence of cannabis: evidence from adolescents in Atlantic Canada. Accid Anal Prev. 2005;37:1025-1034.
51. Mann RE, Adlaf E, Zhao J, et al. Cannabis use and self-reported collisions in a representative sample of adult drivers. J Safety Res. 2007;38:669-674.
52. Taylor J, Wiens T, Peterson J, et al. Characteristics of e-cigarette, or vaping, products used by patients with associated lung injury and products seized by law enforcement—Minnesota, 2018 and 2019. MMWR Morb Mortal Wkly Rep. 2019;68:1096-1100.
53. Hancock-Allen JB, Barker L, VanDyke M, et al. Notes from the field: death following ingestion of an edible marijuana product—Colorado, March 2014. MMWR Morb Mortal Wkly Rep. 2015;64:771-772.
54. Murray RM, Quigley H, Quattrone D, et al. Traditional marijuana, high-potency cannabis and synthetic cannabinoids: increasing risk for psychosis. World Psychiatry. 2016;15:195-204.
55. Di Forti MD, Sallis H, Allegri F, et al. Daily use, especially of high-potency cannabis, drives the earlier onset of psychosis in cannabis users. Schizophr Bull. 2014;40:1509-1517.
56. Miller WR. Motivational interviewing: research, practice, and puzzles. Addict Behav. 1996;21:835-842.
57. Gates PJ, Sabioni P, Copeland J, et al. Psychosocial interventions for cannabis use disorder. Cochrane Database Syst Rev. 2016;(5):CD005336.
58. Wilkinson ST, Stefanovics E, Rosenheck RA. Marijuana use is associated with worse outcomes in symptom severity and violent behavior in patients with posttraumatic stress disorder. J Clin Psychiatry. 2015;76:1174-1180.
59. Cougle JR, Bonn-Miller MO, Vujanovic AA, et al. Posttraumatic stress disorder and cannabis use in a nationally representative sample. Psychol Addict Behav. 2011;25:554-558.
60. Johnson MJ, Pierce JD, Mavandadi S, et al. Mental health symptom severity in cannabis using and non-using veterans with probable PTSD. J Affect Disord. 2016;190:439-442.
61. Wilkinson ST, Radhakrishnan R, D’Souza DC. A systematic review of the evidence for medical marijuana in psychiatric indications. J Clin Psychiatry. 2016;77:1050-1064.
62. Black N, Stockings E, Campbell G, et al. Cannabinoids for the treatment of mental disorders and symptoms of mental disorders: a systematic review and meta-analysis. Lancet Psychiatry. 2019;6:995-1010.
63. Bonnet U, Preuss U. The cannabis withdrawal syndrome: current insights. Subst Abuse Rehabil. 2017;8:9-37.
64. Vandrey R, Smith MT, McCann UD, et al. Sleep disturbance and the effects of extended-release zolpidem during cannabis withdrawal. Drug Alcohol Depend. 2011;117:38-44.
65. Mason BJ, Crean R, Goodell V, et al. A proof-of-concept randomized controlled study of gabapentin: effects on cannabis use, withdrawal and executive function deficits in cannabis-dependent adults. Neuropsychopharmacology. 2012;37:1689-1698.
66. Weinstein A, Miller H, Tal E, et al. Treatment of cannabis withdrawal syndrome using cognitive-behavioral therapy and relapse prevention for cannabis dependence. J Groups Addict Recover. 2010;5:240-263.
PRACTICE RECOMMENDATIONS
› Address underlying conditions for which patients use recreational cannabis to manage symptoms. B
› Consider discrete, in-office sessions of motivational interviewing and referral for cognitive behavioral therapy for patients with problematic cannabis use. B
› Provide counseling around harm reduction for all patients—especially those with problematic cannabis use. C
› Consider referral to an addiction specialist for patients with cannabis use disorder or other problematic cannabis use. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Cannabis use disorder
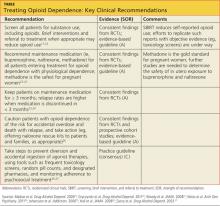
Diagnosing and Treating Opioid Dependence
CASE Sam M., age 48, is in your office for the first time in more than two years. He has gained a considerable amount of weight and appears a bit sluggish, and you wonder whether he's depressed. As you take a history, Sam reminds you that he was laid off 16 months ago and had been caring for his wife, who sustained a debilitating back injury. When you saw her recently, she told you she's back to work and pain-free. So you're taken aback when Sam asks you to refill his wife's oxycodone prescription for lingering pain that often keeps her up at night.
If Sam were your patient, would you suspect opioid dependence?
Dependence on opioid analgesics and the adverse consequences associated with it have steadily increased during the past decade. Consider the following:
• Between 2004 and 2008, the number of emergency department visits related to nonmedical prescription opioid use more than doubled, rising by 111%.1
• The increasing prevalence of opioid abuse has led to a recent spike in unintentional deaths,2 with the number of lives lost to opioid analgesic overdose now exceeding that of heroin or cocaine.3
• More than 75% of opioids used for nonmedical purposes were prescribed for someone else.4
The course of opioid use is highly variable. Some people start with a legitimate medical prescription for an opioid analgesic, then continue taking it after the pain subsides. Others experiment briefly with nonmedical prescription opioids or use them intermittently without adverse effect. Some progress from prescription opioids to heroin, despite its dangers.5 Still others experience a catastrophic outcome, such as an overdose or severe accident, the first time they use opioids.6 Rapid progression from misuse of opioids to dependence is most likely to occur in vulnerable populations, such as those with concurrent mental illness, other substance use disorders, or increased sensitivity to pain.7
Understanding the terms. Before we continue, a word about terminology is in order. Misuse generally refers to the use of a medication in a manner (ie, purpose, dose, or frequency) other than its intended use, whereas drug addiction is the repeated use of a drug despite resulting harm. Here we will use opioid dependence to mean a pattern of increasing use characterized by significant impairment and distress and an inability to stop, and opioid withdrawal to reflect a constellation of symptoms, such as insomnia, nausea, diarrhea, and muscle aches, that can follow physiological dependence (though not necessarily opioid dependence). Our definitions of these terms are consistent with those of the American Psychiatric Association (APA).8 Worth noting, however, is the fact that as the APA prepares for the publication of the 5th edition of its Diagnostic and Statistical Manual of Mental Disorders, its Substance Disorder Work Group has proposed replacing the term opioid dependence with opioid use disorder to reduce the confusion associated with these definitions.9
Assessing Illicit Opioid Use: Start With a Targeted Question
Most patients who are opioid dependent do not seek treatment for it10 and are typically free of medical sequelae associated with drug addiction when they see family practitioners. The absence of self-reporting and obvious physical signs and symptoms, coupled with the increase in illicit use of prescription opioids, underscores the need for clinicians to identify patients who are abusing opioids and ensure that they get the help they need.
Screening tools. There are a number of screening tools you can use for this purpose—eg, CAGE-Adapted to Include Drugs (CAGE-AID) and Drug Abuse Screening Test (DAST)11,12—but they have not been found to be significantly better than a careful substance abuse history.13
Straightforward questions. You can start by asking, "Do you take any medications for pain?" If the answer is Yes, get the name of the drug and inquire about the frequency of use and the route, the amount typically taken, and the duration of the current use pattern. Ask specifically about opioids when taking a substance abuse history. After a question about alcohol use, you can say, "Do you use any other drugs in a serious way? Marijuana? Opioids, like Percocet, Vicodin, or Oxycontin?" Although it can be very difficult to detect opioid dependence if the patient is not forthcoming, other likely indicators of drug-seeking behavior should trigger additional questions. (See "Opioid dependence: Red Flags to Keep in Mind."14-16)
"Brief" protocols. Recent studies of Screening, Brief Intervention, and Referral to Treatment (SBIRT) programs have found that the simple, time-limited interventions they offer (visit www.samhsa.gov/prevention/sbirt/SBIRTwhitepaper.pdf to learn more) lead to a reduction in self-reported illicit opioid use.17,18 Clinicians can readily incorporate SBIRT protocols into routine practice, as an evidence-based and often reimbursable approach to substance abuse.17
Additional Steps Before Initiating Treatment
After screening and diagnostic evaluation provide evidence that a patient is opioid dependent, you can take several steps to guide him or her to the appropriate treatment.
A thorough biopsychosocial assessment covering co-occurring psychiatric illnesses, pain, psychosocial stressors contributing to opioid use, and infectious disease screening is required to gain a clear picture of the patient's situation. In every case, acute emergencies such as suicidal ideation require immediate intervention, which may include hospitalization.19
Assess the patient's desire for help. After the initial assessment, it is often helpful to categorize the patient's "stage of change" (precontemplation, contemplation, preparation, action, or maintenance)20 and to tailor your next step accordingly. A patient who denies that opioid use is a problem or who is clearly ambivalent about seeking treatment may require a conversation that uses principles of motivational interviewing—a collaborative approach that aims to evoke and strengthen personal motivation for change.21 Consider a question that encourages him or her to express reasons for change, such as: "How would you like your current situation to be different?" As almost everyone abusing opioids has thoughts about stopping, such a question may help the patient focus on specific changes.
CASE When you question Sam about his interest in oxycodone, he breaks down. He's been unable to find work or to lose the excess weight he gained during the many months he cared for his wife. He tells you that soon after his wife stopped taking the pain pills, he started taking them. At first, he took one occasionally. Then he started taking the opioids every day, and finally, whenever he awakened at night. Now, Sam says, he has no more pills, and he's nauseous, depressed, and unable to sleep—and looking to you for help.
Sam fits the criteria for opioid withdrawal as a result of physiological dependence; further questioning reveals that he also suffers from opioid dependence, and that he is receptive to treatment.
Recommending Treatment and Following Up
Several options are available for patients who, like Sam, have signs and symptoms of opioid withdrawal as a result of physiological dependence. You can provide a referral to a clinician who specializes in addiction, recommend detoxification and/or treatment in an inpatient facility, or initiate pharmacologic treatment and provide a referral to a behavioral therapist. Whatever the initial approach, most patients will ultimately be treated as outpatients, with a combination of pharmacotherapy and behavioral therapy—often, with monitoring and oversight by a primary care provider. Which approach to pursue should be guided by evidence-based recommendations (see table17,22-27) and jointly decided by provider and patient.
Medication Plays a Key Role in Recovery
Recommend medication-assisted treatment, either with an agonist (buprenorphine or methadone) or an antagonist (naltrexone), for every patient with physiological opioid dependence. The goals of pharmacotherapy are to prevent or reduce withdrawal symptoms and craving, avoid relapse, and restore to a normal state any physiological functions (eg, sleep, bowel habits) that have been disrupted by opioid use.28 When continued for at least three months, medication has been shown to improve outcomes.23,24,29 In one recent study, 49% of opioid-dependent participants who were still taking buprenorphine-naloxone at 12 weeks had successful outcomes (minimal or no opioid use), compared with 7% of those undergoing a brief buprenorphine-naloxone taper.24
There are risks associated with medication-assisted therapy, however. Those of greatest concern are a potential increase in drug interactions, the risk of diversion (a concern with both buprenorphine and methadone), and the potential for accidental overdose.2,30
Buprenorphine, a partial m-opioid receptor agonist, is a Schedule III controlled substance and can be dispensed by a pharmacist, making inpatient opioid detoxification unnecessary for many opioid-dependent patients. Clinicians who wish to prescribe buprenorphine for the treatment of opioid dependence must complete an 8-hour course, offered by the American Medical Association and the APA, among other medical groups, and obtain a Drug Enforcement Administration (code "X") license.31
Buprenorphine has a high affinity for, and a slow dissociation from, m-opioid receptors, resulting in the displacement of other opioids from the m receptor and less severe withdrawal.32 As a partial agonist, buprenorphine attenuates opioid withdrawal symptoms with a ceiling, or near-maximal, effect at 16 mg, thereby lowering the risk for overdose.33 A sublingual formulation that combines buprenorphine with naloxone, an opioid antagonist that exerts its full effect when injected but is minimally absorbed sublingually, reduces the potential for abuse of buprenorphine without interfering with its effectiveness.34
Compared with methadone, buprenorphine is less likely to interact with antiretroviral medications or to cause QTc prolongation, erectile dysfunction, or cognitive or psychomotor impairment.31,35-37 Limitations include the ceiling effect, which can be a problem for cases in which more agonist is needed; cost (approximately $12/d); and the lack of approval by the FDA for use during pregnancy.
Buprenorphine maintenance involves three phases: induction, stabilization, and maintenance.38 Induction takes place in a clinician's office at the time the patient experiences opioid withdrawal symptoms, typically 6 to 48 hours after taking the last opioid. Extended treatment improves clinical outcomes,23,24 and longer-term maintenance (of indefinite duration) is frequently required.
Naltrexone is a m-receptor antagonist, and therefore does not cause physical dependence or have agonist effects such as euphoria and sedation. As a result, it has no diversion value and may appeal to those who view opioid-agonist pharmacotherapy as simply trading one drug for another.39 Naltrexone is not a controlled substance and is not subject to the regulatory requirements that buprenorphine and methadone face.
Although agonists can be started in the first day or two after a patient decides to stop using opioids, patients must be opioid-free for at least seven days before starting naltrexone. That's because its antagonist properties will precipitate withdrawal if another opioid is present on the opioid receptors. During the seven-day "washout" period, opioid withdrawal symptoms can be treated with medications such as clonidine and dicyclomine, but such symptoms make patients especially vulnerable to relapse while waiting to start naltrexone.
Oral naltrexone's effectiveness as a treatment for opioid dependence has been limited by poor patient adherence. But a long-acting intramuscular form of the drug, approved by the FDA in 2010 and requiring once-a-month injection, mitigates this concern.40,41
Methadone is a full m-opioid agonist, administered daily at specialized clinics, as a maintenance therapy for opioid dependence. Although office-based physicians can prescribe methadone for pain, the drug can only be used for opioid dependence under the auspices of state- and federally regulated opioid treatment programs (http://findtreatment.samhsa.gov/TreatmentLocator/faces/quickSearch.jspx; a mobile phone application is also available at www.samhsa.gov/mobile/treat mentlocator.aspx).
Methadone, a Schedule III controlled substance with a half-life averaging 24 to 36 hours, requires daily dosing.42 Its slow metabolism and long half-life increase the risk for overdose.
Methadone is best for patients who are highly dependent on opioids and likely to benefit from a structured treatment environment with daily supervision (although patients who are doing well may earn take-home privileges so they don't have to come to the clinic every day).43 New patients should receive an initial dose of 30 mg or less, and a maximum first-day dose of 40 mg.44
Methadone use remains the standard of care for pregnant women being treated for opioid dependence, while studies of the effects of buprenorphine and naltrexone on a developing fetus continue. Although methadone's efficacy, particularly in lower doses, is similar to that of buprenorphine,45 its adverse effect profile is worse. Adverse effects include drug interactions, the potential for respiratory depression (especially in combination with alcohol or sedatives), QTc prolongation (which requires monitoring by ECG), sedation, and weight gain, and should be considered before methadone is selected as a maintenance pharmacotherapy.30,37,46 And, because relapse rates within 12 months of tapering off methadone have been reported to exceed 80%,47 both the clinician and the patient need to consider the likelihood of long-term, even lifelong, maintenance before initiating treatment.
Behavioral Interventions Are a Vital Part of the Picture
Studies evaluating the extent to which various types and amounts of counseling improve outcomes compared with pharmacotherapy alone have had conflicting results.24,48 Nonetheless, most clinicians consider counseling a critical component of treatment for opioid dependence and recommend, at a minimum, either individual or group counseling (various modalities have been shown to be effective) and regular attendance at a self-help group like Narcotics Anonymous. Contingency management, a type of therapy that uses prizes as incentives for desired behaviors; and family therapy, individual counseling, and community-based programs have all been found to improve outcomes.6,49
CASE You refer Sam to an addiction psychiatrist, who stabilizes him on 16 mg/d buprenorphine/naloxone as part of an outpatient treatment program. Sam is enrolled in a weekly buprenorphine stabilization group, where he gives a urine sample each week. He also begins seeing a social worker weekly for counseling and attends Narcotics Anonymous meetings two to three times a week. At a follow-up appointment with you six months later, he reports that he has been abstinent from oxycodone for six months, his sleep is improved, and he feels better about his chances of finding another job.
Your Role in Safeguarding the Patient
With the rising prevalence of opioid overdose, patient education aimed at crisis prevention is crucial as well. Warn patients of the risk for accidental overdose, often associated with relapse, stressing the importance of continuing treatment and taking their maintenance medication exactly as prescribed.
There are other steps that can be taken to safeguard patients—for example, providing naloxone rescue kits to patients and their families when appropriate. The clinician can also institute diversion and overdose prevention measures for patients taking buprenorphine or methadone—providing a lock box for take-home medication, implementing treatment contracts, and using a designated pharmacy to dispense buprenorphine, for example.26,27,50
Regular monitoring, urine drug screens, and random pill counts, in which patients are typically given 24 hours to bring in their prescribed medication so it can be counted, can also help keep patients on track. Treatment for concurrent psychiatric disorders—depression, anxiety, and personality disorders are common among patients with opioid dependence—is also likely to improve treatment outcomes.
References
1. CDC. Emergency department visits involving nonmedical use of selected prescription drugs - United States, 2004-2008. MMWR Morb Mortal Wkly Rep. 2010;59:705-709.
2. Bohnert AS, Valenstein M, Bair MJ, et al. Association between opioid prescribing patterns and opioid overdose-related deaths. JAMA. 2011;305:1315-1321.
3. Warner M, Chen LH, Makuc DM. Increase in fatal poisonings involving opioid analgesics in the United States, 1999-2006. NCHS Data Brief. 2009;22:1-8.
4. Substance Abuse and Mental Health Services Administration. Results From the 2009 National Survey on Drug Use and Health: Volume I. Summary of National Findings. Rockville, Md: SAMHSA, Office of Applied Studies; 2010. NSDUH Series H-38A, HHS publication SMA 10-4856. www.samhsa.gov/dataNSDUH/2k9NSDUH/2k9Results.htm. Accessed August 22, 2012.
5. Hser YI, Huang D, Brecht ML, et al. Contrasting trajectories of heroin, cocaine, and methamphetamine use. J Addict Dis. 2008;27:13-21.
6. Veilleux JC, Colvin PJ, Anderson J, et al. A review of opioid dependence treatment: pharmacological and psychosocial interventions to treat opioid addiction. Clin Psychol Rev. 2011;30:155-166.
7. George O, Koob GF. Individual differences in prefrontal cortex function and the transition from drug use to drug dependence. Neurosci Biobehav Rev. 2011;35:232-247.
8. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 4th ed, text rev (DSM-IV-TR). Arlington, VA: American Psychiatric Association; 2000.
9. American Psychiatric Association. R 19 opioid use disorder (2012). www.dsm5.org/ProposedRevisions/Pages/proposedrevision.aspx?rid=460. Accessed June 20, 2012.
10. Substance Abuse and Mental Health Services Administration. Results From the 2008 National Survey on Drug Use and Health: National Findings. Rockville, MD: SAMHSA, Office of Applied Studies; 2009. NSDUH Series H-36, HHS publication SMA 09-4434. www.samhsa.gov/data/nsduh/2k8nsduh/2k8Results.htm. Accessed August 22, 2012.
11. Brown RL, Rounds LA. Conjoint screening questionnaires for alcohol and other drug abuse: criterion validity in a primary care practice. Wis Med J. 1995;94:135-140.
12. Skinner HA. The drug abuse screening test. Addict Behav. 1982;7:363-371.
13. US Preventive Services Task Force. Screening for Illicit Drug Use: US Preventive Services Task Force Recommendation Statement. January 2008. www.uspreventiveservicestaskforce.org/uspstf08/druguse/drugrs.htm. Accessed May 7, 2012.
14. Gourlay D, Caplan Y, Heit H. Urine Drug Testing in Clinical Practice: Dispelling the Myths and Designing Strategies. San Francisco, Calif: California Academy of Family Physicians; 2006.
15. Jackman R, Purvis J, Mallett B. Chronic nonmalignant pain in primary care. Am Fam Physician. 2008;78:1155-1162.
16. McBane S, Weigle N. Is it time to drug test your chronic pain patient? J Fam Pract. 2010;59:628-633.
17. Madras BK, Compton WM, Avula D, et al. Screening, brief interventions, referral to treatment (SBIRT) for illicit drug and alcohol use at multiple healthcare sites: comparison at intake and 6 months later. Drug Alcohol Depend. 2009;99:280-295.
18. The InSight Project Research Group. SBIRT outcomes in Houston: final report on InSight, a hospital district-based program for patients at risk for alcohol or drug use problems. Alcohol Clin Exp Res. 2009;33:1374-1381.
19. Borges G, Walters EE, Kessler RC. Associations of substance use, abuse, and dependence with subsequent suicidal behavior. Am J Epidemiol. 2000;151:781-789.
20. Prochaska JO, DiClemente CC. Stages and processes of self-change of smoking: toward an integrative model of change. J Consult Clin Psychol. 1983;51:390-395.
21. Smedslund G, Berg RC, Hammerstrom KT, et al. Motivational interviewing for substance abuse. Cochrane Database Syst Rev. 2011;(5):CD008063.
22. Gryczynski J, Mitchell SG, Peterson TR, et al. The relationship between services delivered and substance use outcomes in New Mexico's Screening, Brief Intervention, Referral and Treatment (SBIRT) Initiative. Drug Alcohol Depend. 2011;118:152-157.
23. Woody GE, Poole SA, Subramaniam G, et al. Extended vs short-term buprenorphine-naloxone for treatment of opioid-addicted youth: a randomized trial. JAMA. 2008;300:2003-2011.
24. Weiss RD, Potter JS, Fiellin DA, et al. Adjunctive counseling during brief and extended buprenorphine-naloxone treatment for prescription opioid dependence: a 2-phase randomized controlled trial. Arch Gen Psychiatry. 2011;68:1238-1246.
25. Johansson BA, Berglund M, Lindgren A. Efficacy of maintenance treatment with naltrexone for opioid dependence: a meta-analytical review. Addiction. 2006;101:491-503.
26. Hall AJ, Logan JE, Toblin RL, et al. Patterns of abuse among unintentional pharmaceutical overdose fatalities. JAMA. 2008;300:2613-2620.
27. Zacny J, Bigelow G, Compton P, et al. College on Problems of Drug Dependence taskforce on prescription opioid non-medical use and abuse: position statement. Drug Alcohol Depend. 2003;69:215-232.
28. Kreek MJ. Rationale for maintenance pharmacotherapy of opiate dependence. Res Publ Assoc Res Nerv Ment Dis. 1992;70:205-230.
29. Mattick RP, Breen C, Kimber J, et al. Methadone maintenance therapy versus no opioid replacement therapy for opioid dependence. Cochrane Database Syst Rev. 2009;(3):CD002209.
30. McCance-Katz EF, Sullivan LE, Nallani S. Drug interactions of clinical importance among the opioids, methadone and buprenorphine, and other frequently prescribed medications: a review. Am J Addict. 2010;19:4-16.
31. Office of National Drug Control Policy Reauthorization Act of 2006 (ONDCPRA), HR 6344, 109th Cong, 2nd Sess (2006).
32. Lewis JW, Walter D. Buprenorphine—background to its development as a treatment for opiate dependence. In: Blaine JD, ed. Buprenorphine: An Alternative Treatment for Opioid Dependence. Rockville, MD: National Institute on Drug Abuse; 1992:5-11. NIDA Research Monograph, No. 121. http://archives.drugabuse.gov/pdf/monographs/121.pdf. Accessed August 22, 2012.
33. Walsh SL, Preston KL, Stitzer ML, et al. Clinical pharmacology of buprenorphine: ceiling effects at high doses. Clin Pharmacol Ther. 1994;55:569-580.
34. Alho H, Sinclair D, Vuori E, et al. Abuse liability of buprenorphine-naloxone tablets in untreated IV drug users. Drug Alcohol Depend. 2007;88:75-78.
35. Hallinan R, Byrne A, Agho K, et al. Erectile dysfunction in men receiving methadone and buprenorphine maintenance treatment. J Sex Med. 2008;5:684-692.
36. Rapeli P, Fabritius C, Alho H, et al. Methadone vs. buprenorphine/naloxone during early opioid substitution treatment: a naturalistic comparison of cognitive performance relative to healthy controls. BMC Clin Pharmacol. 2007;7:5.
37. Wedam EF, Bigelow GE, Johnson RE, et al. QT-interval effects of methadone, levomethadyl, and buprenorphine in a randomized trial. Arch Intern Med. 2007;167:2469-2475.
38. Center for Substance Abuse Treatment. Clinical Guidelines for the Use of Buprenorphine in the Treatment of Opioid Addiction. Rockville, MD: Substance Abuse and Mental Health Services Administration; 2004. Treatment Improvement Protocol (TIP) Series 40. DHHS publication SMA 04‐3939.
39. Kleber HD. Methadone maintenance 4 decades later: thousands of lives saved but still controversial. JAMA. 2008;300:2303-2305.
40. Hulse GK, Morris N, Arnold-Reed D, et al. Improving clinical outcomes in treating heroin dependence: randomized, controlled trial of oral or implant naltrexone. Arch Gen Psychiatry. 2009;66:1108-1115.
41. FDA. FDA approves injectable drug to treat opioid-dependent patients. October 12, 2010. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/2010/ucm229109.htm. Accessed September 11, 2012.
42. Inturrisi CE, Verebely K. The levels of methadone in the plasma in methadone maintenance. Clin Pharmacol Ther. 1972;13(5 pt 1):633-637.
43. Stitzer M, Bigelow G, Lawrence C, et al. Medication take-home as a reinforcer in a methadone maintenance program. Addict Behav. 1977;2:9-14.
44. Code of Federal Regulations. Title 42.8.12. Federal Opioid Treatment Standards. October 2010.
45. Johnson RE. Chutuape MA, Strain EC, et al. A comparison of levomethadyl acetate, buprenorphine, and methadone for opioid dependence. N Engl J Med. 2000;343:1290-1297.
46. Krantz MJ, Martin J, Stimmel B, et al. QTc interval screening in methadone treatment. Ann Intern Med. 2009;150:387-395.
47. Ball JC, Lange WR, Myers CP, et al. Reducing the risk of AIDS through methadone maintenance treatment. J Health Soc Behav. 1988;29:214-226.
48. Fiellin DA, Pantalon MV, Chawarski MC, et al. Counseling plus buprenorphine-naloxone maintenance therapy for opioid dependence. N Engl J Med. 2006;355:365-374.
49. Defulio A, Everly JJ, Leoutsakos JM, et al. Employment-based reinforcement of adherence to an FDA approved extended release formulation of naltrexone in opioid-dependent adults: a randomized controlled trial. Drug Alcohol Depend. 2012;120:48-54.
50. Savage SR. Management of opioid medications in patients with chronic pain and risk of substance misuse. Curr Psychiatry Rep. 2009;11:377-384.
This article was originally published in the Journal of Family Practice. 2012;61:588-597.
CASE Sam M., age 48, is in your office for the first time in more than two years. He has gained a considerable amount of weight and appears a bit sluggish, and you wonder whether he's depressed. As you take a history, Sam reminds you that he was laid off 16 months ago and had been caring for his wife, who sustained a debilitating back injury. When you saw her recently, she told you she's back to work and pain-free. So you're taken aback when Sam asks you to refill his wife's oxycodone prescription for lingering pain that often keeps her up at night.
If Sam were your patient, would you suspect opioid dependence?
Dependence on opioid analgesics and the adverse consequences associated with it have steadily increased during the past decade. Consider the following:
• Between 2004 and 2008, the number of emergency department visits related to nonmedical prescription opioid use more than doubled, rising by 111%.1
• The increasing prevalence of opioid abuse has led to a recent spike in unintentional deaths,2 with the number of lives lost to opioid analgesic overdose now exceeding that of heroin or cocaine.3
• More than 75% of opioids used for nonmedical purposes were prescribed for someone else.4
The course of opioid use is highly variable. Some people start with a legitimate medical prescription for an opioid analgesic, then continue taking it after the pain subsides. Others experiment briefly with nonmedical prescription opioids or use them intermittently without adverse effect. Some progress from prescription opioids to heroin, despite its dangers.5 Still others experience a catastrophic outcome, such as an overdose or severe accident, the first time they use opioids.6 Rapid progression from misuse of opioids to dependence is most likely to occur in vulnerable populations, such as those with concurrent mental illness, other substance use disorders, or increased sensitivity to pain.7
Understanding the terms. Before we continue, a word about terminology is in order. Misuse generally refers to the use of a medication in a manner (ie, purpose, dose, or frequency) other than its intended use, whereas drug addiction is the repeated use of a drug despite resulting harm. Here we will use opioid dependence to mean a pattern of increasing use characterized by significant impairment and distress and an inability to stop, and opioid withdrawal to reflect a constellation of symptoms, such as insomnia, nausea, diarrhea, and muscle aches, that can follow physiological dependence (though not necessarily opioid dependence). Our definitions of these terms are consistent with those of the American Psychiatric Association (APA).8 Worth noting, however, is the fact that as the APA prepares for the publication of the 5th edition of its Diagnostic and Statistical Manual of Mental Disorders, its Substance Disorder Work Group has proposed replacing the term opioid dependence with opioid use disorder to reduce the confusion associated with these definitions.9
Assessing Illicit Opioid Use: Start With a Targeted Question
Most patients who are opioid dependent do not seek treatment for it10 and are typically free of medical sequelae associated with drug addiction when they see family practitioners. The absence of self-reporting and obvious physical signs and symptoms, coupled with the increase in illicit use of prescription opioids, underscores the need for clinicians to identify patients who are abusing opioids and ensure that they get the help they need.
Screening tools. There are a number of screening tools you can use for this purpose—eg, CAGE-Adapted to Include Drugs (CAGE-AID) and Drug Abuse Screening Test (DAST)11,12—but they have not been found to be significantly better than a careful substance abuse history.13
Straightforward questions. You can start by asking, "Do you take any medications for pain?" If the answer is Yes, get the name of the drug and inquire about the frequency of use and the route, the amount typically taken, and the duration of the current use pattern. Ask specifically about opioids when taking a substance abuse history. After a question about alcohol use, you can say, "Do you use any other drugs in a serious way? Marijuana? Opioids, like Percocet, Vicodin, or Oxycontin?" Although it can be very difficult to detect opioid dependence if the patient is not forthcoming, other likely indicators of drug-seeking behavior should trigger additional questions. (See "Opioid dependence: Red Flags to Keep in Mind."14-16)
"Brief" protocols. Recent studies of Screening, Brief Intervention, and Referral to Treatment (SBIRT) programs have found that the simple, time-limited interventions they offer (visit www.samhsa.gov/prevention/sbirt/SBIRTwhitepaper.pdf to learn more) lead to a reduction in self-reported illicit opioid use.17,18 Clinicians can readily incorporate SBIRT protocols into routine practice, as an evidence-based and often reimbursable approach to substance abuse.17
Additional Steps Before Initiating Treatment
After screening and diagnostic evaluation provide evidence that a patient is opioid dependent, you can take several steps to guide him or her to the appropriate treatment.
A thorough biopsychosocial assessment covering co-occurring psychiatric illnesses, pain, psychosocial stressors contributing to opioid use, and infectious disease screening is required to gain a clear picture of the patient's situation. In every case, acute emergencies such as suicidal ideation require immediate intervention, which may include hospitalization.19
Assess the patient's desire for help. After the initial assessment, it is often helpful to categorize the patient's "stage of change" (precontemplation, contemplation, preparation, action, or maintenance)20 and to tailor your next step accordingly. A patient who denies that opioid use is a problem or who is clearly ambivalent about seeking treatment may require a conversation that uses principles of motivational interviewing—a collaborative approach that aims to evoke and strengthen personal motivation for change.21 Consider a question that encourages him or her to express reasons for change, such as: "How would you like your current situation to be different?" As almost everyone abusing opioids has thoughts about stopping, such a question may help the patient focus on specific changes.
CASE When you question Sam about his interest in oxycodone, he breaks down. He's been unable to find work or to lose the excess weight he gained during the many months he cared for his wife. He tells you that soon after his wife stopped taking the pain pills, he started taking them. At first, he took one occasionally. Then he started taking the opioids every day, and finally, whenever he awakened at night. Now, Sam says, he has no more pills, and he's nauseous, depressed, and unable to sleep—and looking to you for help.
Sam fits the criteria for opioid withdrawal as a result of physiological dependence; further questioning reveals that he also suffers from opioid dependence, and that he is receptive to treatment.
Recommending Treatment and Following Up
Several options are available for patients who, like Sam, have signs and symptoms of opioid withdrawal as a result of physiological dependence. You can provide a referral to a clinician who specializes in addiction, recommend detoxification and/or treatment in an inpatient facility, or initiate pharmacologic treatment and provide a referral to a behavioral therapist. Whatever the initial approach, most patients will ultimately be treated as outpatients, with a combination of pharmacotherapy and behavioral therapy—often, with monitoring and oversight by a primary care provider. Which approach to pursue should be guided by evidence-based recommendations (see table17,22-27) and jointly decided by provider and patient.
Medication Plays a Key Role in Recovery
Recommend medication-assisted treatment, either with an agonist (buprenorphine or methadone) or an antagonist (naltrexone), for every patient with physiological opioid dependence. The goals of pharmacotherapy are to prevent or reduce withdrawal symptoms and craving, avoid relapse, and restore to a normal state any physiological functions (eg, sleep, bowel habits) that have been disrupted by opioid use.28 When continued for at least three months, medication has been shown to improve outcomes.23,24,29 In one recent study, 49% of opioid-dependent participants who were still taking buprenorphine-naloxone at 12 weeks had successful outcomes (minimal or no opioid use), compared with 7% of those undergoing a brief buprenorphine-naloxone taper.24
There are risks associated with medication-assisted therapy, however. Those of greatest concern are a potential increase in drug interactions, the risk of diversion (a concern with both buprenorphine and methadone), and the potential for accidental overdose.2,30
Buprenorphine, a partial m-opioid receptor agonist, is a Schedule III controlled substance and can be dispensed by a pharmacist, making inpatient opioid detoxification unnecessary for many opioid-dependent patients. Clinicians who wish to prescribe buprenorphine for the treatment of opioid dependence must complete an 8-hour course, offered by the American Medical Association and the APA, among other medical groups, and obtain a Drug Enforcement Administration (code "X") license.31
Buprenorphine has a high affinity for, and a slow dissociation from, m-opioid receptors, resulting in the displacement of other opioids from the m receptor and less severe withdrawal.32 As a partial agonist, buprenorphine attenuates opioid withdrawal symptoms with a ceiling, or near-maximal, effect at 16 mg, thereby lowering the risk for overdose.33 A sublingual formulation that combines buprenorphine with naloxone, an opioid antagonist that exerts its full effect when injected but is minimally absorbed sublingually, reduces the potential for abuse of buprenorphine without interfering with its effectiveness.34
Compared with methadone, buprenorphine is less likely to interact with antiretroviral medications or to cause QTc prolongation, erectile dysfunction, or cognitive or psychomotor impairment.31,35-37 Limitations include the ceiling effect, which can be a problem for cases in which more agonist is needed; cost (approximately $12/d); and the lack of approval by the FDA for use during pregnancy.
Buprenorphine maintenance involves three phases: induction, stabilization, and maintenance.38 Induction takes place in a clinician's office at the time the patient experiences opioid withdrawal symptoms, typically 6 to 48 hours after taking the last opioid. Extended treatment improves clinical outcomes,23,24 and longer-term maintenance (of indefinite duration) is frequently required.
Naltrexone is a m-receptor antagonist, and therefore does not cause physical dependence or have agonist effects such as euphoria and sedation. As a result, it has no diversion value and may appeal to those who view opioid-agonist pharmacotherapy as simply trading one drug for another.39 Naltrexone is not a controlled substance and is not subject to the regulatory requirements that buprenorphine and methadone face.
Although agonists can be started in the first day or two after a patient decides to stop using opioids, patients must be opioid-free for at least seven days before starting naltrexone. That's because its antagonist properties will precipitate withdrawal if another opioid is present on the opioid receptors. During the seven-day "washout" period, opioid withdrawal symptoms can be treated with medications such as clonidine and dicyclomine, but such symptoms make patients especially vulnerable to relapse while waiting to start naltrexone.
Oral naltrexone's effectiveness as a treatment for opioid dependence has been limited by poor patient adherence. But a long-acting intramuscular form of the drug, approved by the FDA in 2010 and requiring once-a-month injection, mitigates this concern.40,41
Methadone is a full m-opioid agonist, administered daily at specialized clinics, as a maintenance therapy for opioid dependence. Although office-based physicians can prescribe methadone for pain, the drug can only be used for opioid dependence under the auspices of state- and federally regulated opioid treatment programs (http://findtreatment.samhsa.gov/TreatmentLocator/faces/quickSearch.jspx; a mobile phone application is also available at www.samhsa.gov/mobile/treat mentlocator.aspx).
Methadone, a Schedule III controlled substance with a half-life averaging 24 to 36 hours, requires daily dosing.42 Its slow metabolism and long half-life increase the risk for overdose.
Methadone is best for patients who are highly dependent on opioids and likely to benefit from a structured treatment environment with daily supervision (although patients who are doing well may earn take-home privileges so they don't have to come to the clinic every day).43 New patients should receive an initial dose of 30 mg or less, and a maximum first-day dose of 40 mg.44
Methadone use remains the standard of care for pregnant women being treated for opioid dependence, while studies of the effects of buprenorphine and naltrexone on a developing fetus continue. Although methadone's efficacy, particularly in lower doses, is similar to that of buprenorphine,45 its adverse effect profile is worse. Adverse effects include drug interactions, the potential for respiratory depression (especially in combination with alcohol or sedatives), QTc prolongation (which requires monitoring by ECG), sedation, and weight gain, and should be considered before methadone is selected as a maintenance pharmacotherapy.30,37,46 And, because relapse rates within 12 months of tapering off methadone have been reported to exceed 80%,47 both the clinician and the patient need to consider the likelihood of long-term, even lifelong, maintenance before initiating treatment.
Behavioral Interventions Are a Vital Part of the Picture
Studies evaluating the extent to which various types and amounts of counseling improve outcomes compared with pharmacotherapy alone have had conflicting results.24,48 Nonetheless, most clinicians consider counseling a critical component of treatment for opioid dependence and recommend, at a minimum, either individual or group counseling (various modalities have been shown to be effective) and regular attendance at a self-help group like Narcotics Anonymous. Contingency management, a type of therapy that uses prizes as incentives for desired behaviors; and family therapy, individual counseling, and community-based programs have all been found to improve outcomes.6,49
CASE You refer Sam to an addiction psychiatrist, who stabilizes him on 16 mg/d buprenorphine/naloxone as part of an outpatient treatment program. Sam is enrolled in a weekly buprenorphine stabilization group, where he gives a urine sample each week. He also begins seeing a social worker weekly for counseling and attends Narcotics Anonymous meetings two to three times a week. At a follow-up appointment with you six months later, he reports that he has been abstinent from oxycodone for six months, his sleep is improved, and he feels better about his chances of finding another job.
Your Role in Safeguarding the Patient
With the rising prevalence of opioid overdose, patient education aimed at crisis prevention is crucial as well. Warn patients of the risk for accidental overdose, often associated with relapse, stressing the importance of continuing treatment and taking their maintenance medication exactly as prescribed.
There are other steps that can be taken to safeguard patients—for example, providing naloxone rescue kits to patients and their families when appropriate. The clinician can also institute diversion and overdose prevention measures for patients taking buprenorphine or methadone—providing a lock box for take-home medication, implementing treatment contracts, and using a designated pharmacy to dispense buprenorphine, for example.26,27,50
Regular monitoring, urine drug screens, and random pill counts, in which patients are typically given 24 hours to bring in their prescribed medication so it can be counted, can also help keep patients on track. Treatment for concurrent psychiatric disorders—depression, anxiety, and personality disorders are common among patients with opioid dependence—is also likely to improve treatment outcomes.
References
1. CDC. Emergency department visits involving nonmedical use of selected prescription drugs - United States, 2004-2008. MMWR Morb Mortal Wkly Rep. 2010;59:705-709.
2. Bohnert AS, Valenstein M, Bair MJ, et al. Association between opioid prescribing patterns and opioid overdose-related deaths. JAMA. 2011;305:1315-1321.
3. Warner M, Chen LH, Makuc DM. Increase in fatal poisonings involving opioid analgesics in the United States, 1999-2006. NCHS Data Brief. 2009;22:1-8.
4. Substance Abuse and Mental Health Services Administration. Results From the 2009 National Survey on Drug Use and Health: Volume I. Summary of National Findings. Rockville, Md: SAMHSA, Office of Applied Studies; 2010. NSDUH Series H-38A, HHS publication SMA 10-4856. www.samhsa.gov/dataNSDUH/2k9NSDUH/2k9Results.htm. Accessed August 22, 2012.
5. Hser YI, Huang D, Brecht ML, et al. Contrasting trajectories of heroin, cocaine, and methamphetamine use. J Addict Dis. 2008;27:13-21.
6. Veilleux JC, Colvin PJ, Anderson J, et al. A review of opioid dependence treatment: pharmacological and psychosocial interventions to treat opioid addiction. Clin Psychol Rev. 2011;30:155-166.
7. George O, Koob GF. Individual differences in prefrontal cortex function and the transition from drug use to drug dependence. Neurosci Biobehav Rev. 2011;35:232-247.
8. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 4th ed, text rev (DSM-IV-TR). Arlington, VA: American Psychiatric Association; 2000.
9. American Psychiatric Association. R 19 opioid use disorder (2012). www.dsm5.org/ProposedRevisions/Pages/proposedrevision.aspx?rid=460. Accessed June 20, 2012.
10. Substance Abuse and Mental Health Services Administration. Results From the 2008 National Survey on Drug Use and Health: National Findings. Rockville, MD: SAMHSA, Office of Applied Studies; 2009. NSDUH Series H-36, HHS publication SMA 09-4434. www.samhsa.gov/data/nsduh/2k8nsduh/2k8Results.htm. Accessed August 22, 2012.
11. Brown RL, Rounds LA. Conjoint screening questionnaires for alcohol and other drug abuse: criterion validity in a primary care practice. Wis Med J. 1995;94:135-140.
12. Skinner HA. The drug abuse screening test. Addict Behav. 1982;7:363-371.
13. US Preventive Services Task Force. Screening for Illicit Drug Use: US Preventive Services Task Force Recommendation Statement. January 2008. www.uspreventiveservicestaskforce.org/uspstf08/druguse/drugrs.htm. Accessed May 7, 2012.
14. Gourlay D, Caplan Y, Heit H. Urine Drug Testing in Clinical Practice: Dispelling the Myths and Designing Strategies. San Francisco, Calif: California Academy of Family Physicians; 2006.
15. Jackman R, Purvis J, Mallett B. Chronic nonmalignant pain in primary care. Am Fam Physician. 2008;78:1155-1162.
16. McBane S, Weigle N. Is it time to drug test your chronic pain patient? J Fam Pract. 2010;59:628-633.
17. Madras BK, Compton WM, Avula D, et al. Screening, brief interventions, referral to treatment (SBIRT) for illicit drug and alcohol use at multiple healthcare sites: comparison at intake and 6 months later. Drug Alcohol Depend. 2009;99:280-295.
18. The InSight Project Research Group. SBIRT outcomes in Houston: final report on InSight, a hospital district-based program for patients at risk for alcohol or drug use problems. Alcohol Clin Exp Res. 2009;33:1374-1381.
19. Borges G, Walters EE, Kessler RC. Associations of substance use, abuse, and dependence with subsequent suicidal behavior. Am J Epidemiol. 2000;151:781-789.
20. Prochaska JO, DiClemente CC. Stages and processes of self-change of smoking: toward an integrative model of change. J Consult Clin Psychol. 1983;51:390-395.
21. Smedslund G, Berg RC, Hammerstrom KT, et al. Motivational interviewing for substance abuse. Cochrane Database Syst Rev. 2011;(5):CD008063.
22. Gryczynski J, Mitchell SG, Peterson TR, et al. The relationship between services delivered and substance use outcomes in New Mexico's Screening, Brief Intervention, Referral and Treatment (SBIRT) Initiative. Drug Alcohol Depend. 2011;118:152-157.
23. Woody GE, Poole SA, Subramaniam G, et al. Extended vs short-term buprenorphine-naloxone for treatment of opioid-addicted youth: a randomized trial. JAMA. 2008;300:2003-2011.
24. Weiss RD, Potter JS, Fiellin DA, et al. Adjunctive counseling during brief and extended buprenorphine-naloxone treatment for prescription opioid dependence: a 2-phase randomized controlled trial. Arch Gen Psychiatry. 2011;68:1238-1246.
25. Johansson BA, Berglund M, Lindgren A. Efficacy of maintenance treatment with naltrexone for opioid dependence: a meta-analytical review. Addiction. 2006;101:491-503.
26. Hall AJ, Logan JE, Toblin RL, et al. Patterns of abuse among unintentional pharmaceutical overdose fatalities. JAMA. 2008;300:2613-2620.
27. Zacny J, Bigelow G, Compton P, et al. College on Problems of Drug Dependence taskforce on prescription opioid non-medical use and abuse: position statement. Drug Alcohol Depend. 2003;69:215-232.
28. Kreek MJ. Rationale for maintenance pharmacotherapy of opiate dependence. Res Publ Assoc Res Nerv Ment Dis. 1992;70:205-230.
29. Mattick RP, Breen C, Kimber J, et al. Methadone maintenance therapy versus no opioid replacement therapy for opioid dependence. Cochrane Database Syst Rev. 2009;(3):CD002209.
30. McCance-Katz EF, Sullivan LE, Nallani S. Drug interactions of clinical importance among the opioids, methadone and buprenorphine, and other frequently prescribed medications: a review. Am J Addict. 2010;19:4-16.
31. Office of National Drug Control Policy Reauthorization Act of 2006 (ONDCPRA), HR 6344, 109th Cong, 2nd Sess (2006).
32. Lewis JW, Walter D. Buprenorphine—background to its development as a treatment for opiate dependence. In: Blaine JD, ed. Buprenorphine: An Alternative Treatment for Opioid Dependence. Rockville, MD: National Institute on Drug Abuse; 1992:5-11. NIDA Research Monograph, No. 121. http://archives.drugabuse.gov/pdf/monographs/121.pdf. Accessed August 22, 2012.
33. Walsh SL, Preston KL, Stitzer ML, et al. Clinical pharmacology of buprenorphine: ceiling effects at high doses. Clin Pharmacol Ther. 1994;55:569-580.
34. Alho H, Sinclair D, Vuori E, et al. Abuse liability of buprenorphine-naloxone tablets in untreated IV drug users. Drug Alcohol Depend. 2007;88:75-78.
35. Hallinan R, Byrne A, Agho K, et al. Erectile dysfunction in men receiving methadone and buprenorphine maintenance treatment. J Sex Med. 2008;5:684-692.
36. Rapeli P, Fabritius C, Alho H, et al. Methadone vs. buprenorphine/naloxone during early opioid substitution treatment: a naturalistic comparison of cognitive performance relative to healthy controls. BMC Clin Pharmacol. 2007;7:5.
37. Wedam EF, Bigelow GE, Johnson RE, et al. QT-interval effects of methadone, levomethadyl, and buprenorphine in a randomized trial. Arch Intern Med. 2007;167:2469-2475.
38. Center for Substance Abuse Treatment. Clinical Guidelines for the Use of Buprenorphine in the Treatment of Opioid Addiction. Rockville, MD: Substance Abuse and Mental Health Services Administration; 2004. Treatment Improvement Protocol (TIP) Series 40. DHHS publication SMA 04‐3939.
39. Kleber HD. Methadone maintenance 4 decades later: thousands of lives saved but still controversial. JAMA. 2008;300:2303-2305.
40. Hulse GK, Morris N, Arnold-Reed D, et al. Improving clinical outcomes in treating heroin dependence: randomized, controlled trial of oral or implant naltrexone. Arch Gen Psychiatry. 2009;66:1108-1115.
41. FDA. FDA approves injectable drug to treat opioid-dependent patients. October 12, 2010. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/2010/ucm229109.htm. Accessed September 11, 2012.
42. Inturrisi CE, Verebely K. The levels of methadone in the plasma in methadone maintenance. Clin Pharmacol Ther. 1972;13(5 pt 1):633-637.
43. Stitzer M, Bigelow G, Lawrence C, et al. Medication take-home as a reinforcer in a methadone maintenance program. Addict Behav. 1977;2:9-14.
44. Code of Federal Regulations. Title 42.8.12. Federal Opioid Treatment Standards. October 2010.
45. Johnson RE. Chutuape MA, Strain EC, et al. A comparison of levomethadyl acetate, buprenorphine, and methadone for opioid dependence. N Engl J Med. 2000;343:1290-1297.
46. Krantz MJ, Martin J, Stimmel B, et al. QTc interval screening in methadone treatment. Ann Intern Med. 2009;150:387-395.
47. Ball JC, Lange WR, Myers CP, et al. Reducing the risk of AIDS through methadone maintenance treatment. J Health Soc Behav. 1988;29:214-226.
48. Fiellin DA, Pantalon MV, Chawarski MC, et al. Counseling plus buprenorphine-naloxone maintenance therapy for opioid dependence. N Engl J Med. 2006;355:365-374.
49. Defulio A, Everly JJ, Leoutsakos JM, et al. Employment-based reinforcement of adherence to an FDA approved extended release formulation of naltrexone in opioid-dependent adults: a randomized controlled trial. Drug Alcohol Depend. 2012;120:48-54.
50. Savage SR. Management of opioid medications in patients with chronic pain and risk of substance misuse. Curr Psychiatry Rep. 2009;11:377-384.
This article was originally published in the Journal of Family Practice. 2012;61:588-597.
CASE Sam M., age 48, is in your office for the first time in more than two years. He has gained a considerable amount of weight and appears a bit sluggish, and you wonder whether he's depressed. As you take a history, Sam reminds you that he was laid off 16 months ago and had been caring for his wife, who sustained a debilitating back injury. When you saw her recently, she told you she's back to work and pain-free. So you're taken aback when Sam asks you to refill his wife's oxycodone prescription for lingering pain that often keeps her up at night.
If Sam were your patient, would you suspect opioid dependence?
Dependence on opioid analgesics and the adverse consequences associated with it have steadily increased during the past decade. Consider the following:
• Between 2004 and 2008, the number of emergency department visits related to nonmedical prescription opioid use more than doubled, rising by 111%.1
• The increasing prevalence of opioid abuse has led to a recent spike in unintentional deaths,2 with the number of lives lost to opioid analgesic overdose now exceeding that of heroin or cocaine.3
• More than 75% of opioids used for nonmedical purposes were prescribed for someone else.4
The course of opioid use is highly variable. Some people start with a legitimate medical prescription for an opioid analgesic, then continue taking it after the pain subsides. Others experiment briefly with nonmedical prescription opioids or use them intermittently without adverse effect. Some progress from prescription opioids to heroin, despite its dangers.5 Still others experience a catastrophic outcome, such as an overdose or severe accident, the first time they use opioids.6 Rapid progression from misuse of opioids to dependence is most likely to occur in vulnerable populations, such as those with concurrent mental illness, other substance use disorders, or increased sensitivity to pain.7
Understanding the terms. Before we continue, a word about terminology is in order. Misuse generally refers to the use of a medication in a manner (ie, purpose, dose, or frequency) other than its intended use, whereas drug addiction is the repeated use of a drug despite resulting harm. Here we will use opioid dependence to mean a pattern of increasing use characterized by significant impairment and distress and an inability to stop, and opioid withdrawal to reflect a constellation of symptoms, such as insomnia, nausea, diarrhea, and muscle aches, that can follow physiological dependence (though not necessarily opioid dependence). Our definitions of these terms are consistent with those of the American Psychiatric Association (APA).8 Worth noting, however, is the fact that as the APA prepares for the publication of the 5th edition of its Diagnostic and Statistical Manual of Mental Disorders, its Substance Disorder Work Group has proposed replacing the term opioid dependence with opioid use disorder to reduce the confusion associated with these definitions.9
Assessing Illicit Opioid Use: Start With a Targeted Question
Most patients who are opioid dependent do not seek treatment for it10 and are typically free of medical sequelae associated with drug addiction when they see family practitioners. The absence of self-reporting and obvious physical signs and symptoms, coupled with the increase in illicit use of prescription opioids, underscores the need for clinicians to identify patients who are abusing opioids and ensure that they get the help they need.
Screening tools. There are a number of screening tools you can use for this purpose—eg, CAGE-Adapted to Include Drugs (CAGE-AID) and Drug Abuse Screening Test (DAST)11,12—but they have not been found to be significantly better than a careful substance abuse history.13
Straightforward questions. You can start by asking, "Do you take any medications for pain?" If the answer is Yes, get the name of the drug and inquire about the frequency of use and the route, the amount typically taken, and the duration of the current use pattern. Ask specifically about opioids when taking a substance abuse history. After a question about alcohol use, you can say, "Do you use any other drugs in a serious way? Marijuana? Opioids, like Percocet, Vicodin, or Oxycontin?" Although it can be very difficult to detect opioid dependence if the patient is not forthcoming, other likely indicators of drug-seeking behavior should trigger additional questions. (See "Opioid dependence: Red Flags to Keep in Mind."14-16)
"Brief" protocols. Recent studies of Screening, Brief Intervention, and Referral to Treatment (SBIRT) programs have found that the simple, time-limited interventions they offer (visit www.samhsa.gov/prevention/sbirt/SBIRTwhitepaper.pdf to learn more) lead to a reduction in self-reported illicit opioid use.17,18 Clinicians can readily incorporate SBIRT protocols into routine practice, as an evidence-based and often reimbursable approach to substance abuse.17
Additional Steps Before Initiating Treatment
After screening and diagnostic evaluation provide evidence that a patient is opioid dependent, you can take several steps to guide him or her to the appropriate treatment.
A thorough biopsychosocial assessment covering co-occurring psychiatric illnesses, pain, psychosocial stressors contributing to opioid use, and infectious disease screening is required to gain a clear picture of the patient's situation. In every case, acute emergencies such as suicidal ideation require immediate intervention, which may include hospitalization.19
Assess the patient's desire for help. After the initial assessment, it is often helpful to categorize the patient's "stage of change" (precontemplation, contemplation, preparation, action, or maintenance)20 and to tailor your next step accordingly. A patient who denies that opioid use is a problem or who is clearly ambivalent about seeking treatment may require a conversation that uses principles of motivational interviewing—a collaborative approach that aims to evoke and strengthen personal motivation for change.21 Consider a question that encourages him or her to express reasons for change, such as: "How would you like your current situation to be different?" As almost everyone abusing opioids has thoughts about stopping, such a question may help the patient focus on specific changes.
CASE When you question Sam about his interest in oxycodone, he breaks down. He's been unable to find work or to lose the excess weight he gained during the many months he cared for his wife. He tells you that soon after his wife stopped taking the pain pills, he started taking them. At first, he took one occasionally. Then he started taking the opioids every day, and finally, whenever he awakened at night. Now, Sam says, he has no more pills, and he's nauseous, depressed, and unable to sleep—and looking to you for help.
Sam fits the criteria for opioid withdrawal as a result of physiological dependence; further questioning reveals that he also suffers from opioid dependence, and that he is receptive to treatment.
Recommending Treatment and Following Up
Several options are available for patients who, like Sam, have signs and symptoms of opioid withdrawal as a result of physiological dependence. You can provide a referral to a clinician who specializes in addiction, recommend detoxification and/or treatment in an inpatient facility, or initiate pharmacologic treatment and provide a referral to a behavioral therapist. Whatever the initial approach, most patients will ultimately be treated as outpatients, with a combination of pharmacotherapy and behavioral therapy—often, with monitoring and oversight by a primary care provider. Which approach to pursue should be guided by evidence-based recommendations (see table17,22-27) and jointly decided by provider and patient.
Medication Plays a Key Role in Recovery
Recommend medication-assisted treatment, either with an agonist (buprenorphine or methadone) or an antagonist (naltrexone), for every patient with physiological opioid dependence. The goals of pharmacotherapy are to prevent or reduce withdrawal symptoms and craving, avoid relapse, and restore to a normal state any physiological functions (eg, sleep, bowel habits) that have been disrupted by opioid use.28 When continued for at least three months, medication has been shown to improve outcomes.23,24,29 In one recent study, 49% of opioid-dependent participants who were still taking buprenorphine-naloxone at 12 weeks had successful outcomes (minimal or no opioid use), compared with 7% of those undergoing a brief buprenorphine-naloxone taper.24
There are risks associated with medication-assisted therapy, however. Those of greatest concern are a potential increase in drug interactions, the risk of diversion (a concern with both buprenorphine and methadone), and the potential for accidental overdose.2,30
Buprenorphine, a partial m-opioid receptor agonist, is a Schedule III controlled substance and can be dispensed by a pharmacist, making inpatient opioid detoxification unnecessary for many opioid-dependent patients. Clinicians who wish to prescribe buprenorphine for the treatment of opioid dependence must complete an 8-hour course, offered by the American Medical Association and the APA, among other medical groups, and obtain a Drug Enforcement Administration (code "X") license.31
Buprenorphine has a high affinity for, and a slow dissociation from, m-opioid receptors, resulting in the displacement of other opioids from the m receptor and less severe withdrawal.32 As a partial agonist, buprenorphine attenuates opioid withdrawal symptoms with a ceiling, or near-maximal, effect at 16 mg, thereby lowering the risk for overdose.33 A sublingual formulation that combines buprenorphine with naloxone, an opioid antagonist that exerts its full effect when injected but is minimally absorbed sublingually, reduces the potential for abuse of buprenorphine without interfering with its effectiveness.34
Compared with methadone, buprenorphine is less likely to interact with antiretroviral medications or to cause QTc prolongation, erectile dysfunction, or cognitive or psychomotor impairment.31,35-37 Limitations include the ceiling effect, which can be a problem for cases in which more agonist is needed; cost (approximately $12/d); and the lack of approval by the FDA for use during pregnancy.
Buprenorphine maintenance involves three phases: induction, stabilization, and maintenance.38 Induction takes place in a clinician's office at the time the patient experiences opioid withdrawal symptoms, typically 6 to 48 hours after taking the last opioid. Extended treatment improves clinical outcomes,23,24 and longer-term maintenance (of indefinite duration) is frequently required.
Naltrexone is a m-receptor antagonist, and therefore does not cause physical dependence or have agonist effects such as euphoria and sedation. As a result, it has no diversion value and may appeal to those who view opioid-agonist pharmacotherapy as simply trading one drug for another.39 Naltrexone is not a controlled substance and is not subject to the regulatory requirements that buprenorphine and methadone face.
Although agonists can be started in the first day or two after a patient decides to stop using opioids, patients must be opioid-free for at least seven days before starting naltrexone. That's because its antagonist properties will precipitate withdrawal if another opioid is present on the opioid receptors. During the seven-day "washout" period, opioid withdrawal symptoms can be treated with medications such as clonidine and dicyclomine, but such symptoms make patients especially vulnerable to relapse while waiting to start naltrexone.
Oral naltrexone's effectiveness as a treatment for opioid dependence has been limited by poor patient adherence. But a long-acting intramuscular form of the drug, approved by the FDA in 2010 and requiring once-a-month injection, mitigates this concern.40,41
Methadone is a full m-opioid agonist, administered daily at specialized clinics, as a maintenance therapy for opioid dependence. Although office-based physicians can prescribe methadone for pain, the drug can only be used for opioid dependence under the auspices of state- and federally regulated opioid treatment programs (http://findtreatment.samhsa.gov/TreatmentLocator/faces/quickSearch.jspx; a mobile phone application is also available at www.samhsa.gov/mobile/treat mentlocator.aspx).
Methadone, a Schedule III controlled substance with a half-life averaging 24 to 36 hours, requires daily dosing.42 Its slow metabolism and long half-life increase the risk for overdose.
Methadone is best for patients who are highly dependent on opioids and likely to benefit from a structured treatment environment with daily supervision (although patients who are doing well may earn take-home privileges so they don't have to come to the clinic every day).43 New patients should receive an initial dose of 30 mg or less, and a maximum first-day dose of 40 mg.44
Methadone use remains the standard of care for pregnant women being treated for opioid dependence, while studies of the effects of buprenorphine and naltrexone on a developing fetus continue. Although methadone's efficacy, particularly in lower doses, is similar to that of buprenorphine,45 its adverse effect profile is worse. Adverse effects include drug interactions, the potential for respiratory depression (especially in combination with alcohol or sedatives), QTc prolongation (which requires monitoring by ECG), sedation, and weight gain, and should be considered before methadone is selected as a maintenance pharmacotherapy.30,37,46 And, because relapse rates within 12 months of tapering off methadone have been reported to exceed 80%,47 both the clinician and the patient need to consider the likelihood of long-term, even lifelong, maintenance before initiating treatment.
Behavioral Interventions Are a Vital Part of the Picture
Studies evaluating the extent to which various types and amounts of counseling improve outcomes compared with pharmacotherapy alone have had conflicting results.24,48 Nonetheless, most clinicians consider counseling a critical component of treatment for opioid dependence and recommend, at a minimum, either individual or group counseling (various modalities have been shown to be effective) and regular attendance at a self-help group like Narcotics Anonymous. Contingency management, a type of therapy that uses prizes as incentives for desired behaviors; and family therapy, individual counseling, and community-based programs have all been found to improve outcomes.6,49
CASE You refer Sam to an addiction psychiatrist, who stabilizes him on 16 mg/d buprenorphine/naloxone as part of an outpatient treatment program. Sam is enrolled in a weekly buprenorphine stabilization group, where he gives a urine sample each week. He also begins seeing a social worker weekly for counseling and attends Narcotics Anonymous meetings two to three times a week. At a follow-up appointment with you six months later, he reports that he has been abstinent from oxycodone for six months, his sleep is improved, and he feels better about his chances of finding another job.
Your Role in Safeguarding the Patient
With the rising prevalence of opioid overdose, patient education aimed at crisis prevention is crucial as well. Warn patients of the risk for accidental overdose, often associated with relapse, stressing the importance of continuing treatment and taking their maintenance medication exactly as prescribed.
There are other steps that can be taken to safeguard patients—for example, providing naloxone rescue kits to patients and their families when appropriate. The clinician can also institute diversion and overdose prevention measures for patients taking buprenorphine or methadone—providing a lock box for take-home medication, implementing treatment contracts, and using a designated pharmacy to dispense buprenorphine, for example.26,27,50
Regular monitoring, urine drug screens, and random pill counts, in which patients are typically given 24 hours to bring in their prescribed medication so it can be counted, can also help keep patients on track. Treatment for concurrent psychiatric disorders—depression, anxiety, and personality disorders are common among patients with opioid dependence—is also likely to improve treatment outcomes.
References
1. CDC. Emergency department visits involving nonmedical use of selected prescription drugs - United States, 2004-2008. MMWR Morb Mortal Wkly Rep. 2010;59:705-709.
2. Bohnert AS, Valenstein M, Bair MJ, et al. Association between opioid prescribing patterns and opioid overdose-related deaths. JAMA. 2011;305:1315-1321.
3. Warner M, Chen LH, Makuc DM. Increase in fatal poisonings involving opioid analgesics in the United States, 1999-2006. NCHS Data Brief. 2009;22:1-8.
4. Substance Abuse and Mental Health Services Administration. Results From the 2009 National Survey on Drug Use and Health: Volume I. Summary of National Findings. Rockville, Md: SAMHSA, Office of Applied Studies; 2010. NSDUH Series H-38A, HHS publication SMA 10-4856. www.samhsa.gov/dataNSDUH/2k9NSDUH/2k9Results.htm. Accessed August 22, 2012.
5. Hser YI, Huang D, Brecht ML, et al. Contrasting trajectories of heroin, cocaine, and methamphetamine use. J Addict Dis. 2008;27:13-21.
6. Veilleux JC, Colvin PJ, Anderson J, et al. A review of opioid dependence treatment: pharmacological and psychosocial interventions to treat opioid addiction. Clin Psychol Rev. 2011;30:155-166.
7. George O, Koob GF. Individual differences in prefrontal cortex function and the transition from drug use to drug dependence. Neurosci Biobehav Rev. 2011;35:232-247.
8. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 4th ed, text rev (DSM-IV-TR). Arlington, VA: American Psychiatric Association; 2000.
9. American Psychiatric Association. R 19 opioid use disorder (2012). www.dsm5.org/ProposedRevisions/Pages/proposedrevision.aspx?rid=460. Accessed June 20, 2012.
10. Substance Abuse and Mental Health Services Administration. Results From the 2008 National Survey on Drug Use and Health: National Findings. Rockville, MD: SAMHSA, Office of Applied Studies; 2009. NSDUH Series H-36, HHS publication SMA 09-4434. www.samhsa.gov/data/nsduh/2k8nsduh/2k8Results.htm. Accessed August 22, 2012.
11. Brown RL, Rounds LA. Conjoint screening questionnaires for alcohol and other drug abuse: criterion validity in a primary care practice. Wis Med J. 1995;94:135-140.
12. Skinner HA. The drug abuse screening test. Addict Behav. 1982;7:363-371.
13. US Preventive Services Task Force. Screening for Illicit Drug Use: US Preventive Services Task Force Recommendation Statement. January 2008. www.uspreventiveservicestaskforce.org/uspstf08/druguse/drugrs.htm. Accessed May 7, 2012.
14. Gourlay D, Caplan Y, Heit H. Urine Drug Testing in Clinical Practice: Dispelling the Myths and Designing Strategies. San Francisco, Calif: California Academy of Family Physicians; 2006.
15. Jackman R, Purvis J, Mallett B. Chronic nonmalignant pain in primary care. Am Fam Physician. 2008;78:1155-1162.
16. McBane S, Weigle N. Is it time to drug test your chronic pain patient? J Fam Pract. 2010;59:628-633.
17. Madras BK, Compton WM, Avula D, et al. Screening, brief interventions, referral to treatment (SBIRT) for illicit drug and alcohol use at multiple healthcare sites: comparison at intake and 6 months later. Drug Alcohol Depend. 2009;99:280-295.
18. The InSight Project Research Group. SBIRT outcomes in Houston: final report on InSight, a hospital district-based program for patients at risk for alcohol or drug use problems. Alcohol Clin Exp Res. 2009;33:1374-1381.
19. Borges G, Walters EE, Kessler RC. Associations of substance use, abuse, and dependence with subsequent suicidal behavior. Am J Epidemiol. 2000;151:781-789.
20. Prochaska JO, DiClemente CC. Stages and processes of self-change of smoking: toward an integrative model of change. J Consult Clin Psychol. 1983;51:390-395.
21. Smedslund G, Berg RC, Hammerstrom KT, et al. Motivational interviewing for substance abuse. Cochrane Database Syst Rev. 2011;(5):CD008063.
22. Gryczynski J, Mitchell SG, Peterson TR, et al. The relationship between services delivered and substance use outcomes in New Mexico's Screening, Brief Intervention, Referral and Treatment (SBIRT) Initiative. Drug Alcohol Depend. 2011;118:152-157.
23. Woody GE, Poole SA, Subramaniam G, et al. Extended vs short-term buprenorphine-naloxone for treatment of opioid-addicted youth: a randomized trial. JAMA. 2008;300:2003-2011.
24. Weiss RD, Potter JS, Fiellin DA, et al. Adjunctive counseling during brief and extended buprenorphine-naloxone treatment for prescription opioid dependence: a 2-phase randomized controlled trial. Arch Gen Psychiatry. 2011;68:1238-1246.
25. Johansson BA, Berglund M, Lindgren A. Efficacy of maintenance treatment with naltrexone for opioid dependence: a meta-analytical review. Addiction. 2006;101:491-503.
26. Hall AJ, Logan JE, Toblin RL, et al. Patterns of abuse among unintentional pharmaceutical overdose fatalities. JAMA. 2008;300:2613-2620.
27. Zacny J, Bigelow G, Compton P, et al. College on Problems of Drug Dependence taskforce on prescription opioid non-medical use and abuse: position statement. Drug Alcohol Depend. 2003;69:215-232.
28. Kreek MJ. Rationale for maintenance pharmacotherapy of opiate dependence. Res Publ Assoc Res Nerv Ment Dis. 1992;70:205-230.
29. Mattick RP, Breen C, Kimber J, et al. Methadone maintenance therapy versus no opioid replacement therapy for opioid dependence. Cochrane Database Syst Rev. 2009;(3):CD002209.
30. McCance-Katz EF, Sullivan LE, Nallani S. Drug interactions of clinical importance among the opioids, methadone and buprenorphine, and other frequently prescribed medications: a review. Am J Addict. 2010;19:4-16.
31. Office of National Drug Control Policy Reauthorization Act of 2006 (ONDCPRA), HR 6344, 109th Cong, 2nd Sess (2006).
32. Lewis JW, Walter D. Buprenorphine—background to its development as a treatment for opiate dependence. In: Blaine JD, ed. Buprenorphine: An Alternative Treatment for Opioid Dependence. Rockville, MD: National Institute on Drug Abuse; 1992:5-11. NIDA Research Monograph, No. 121. http://archives.drugabuse.gov/pdf/monographs/121.pdf. Accessed August 22, 2012.
33. Walsh SL, Preston KL, Stitzer ML, et al. Clinical pharmacology of buprenorphine: ceiling effects at high doses. Clin Pharmacol Ther. 1994;55:569-580.
34. Alho H, Sinclair D, Vuori E, et al. Abuse liability of buprenorphine-naloxone tablets in untreated IV drug users. Drug Alcohol Depend. 2007;88:75-78.
35. Hallinan R, Byrne A, Agho K, et al. Erectile dysfunction in men receiving methadone and buprenorphine maintenance treatment. J Sex Med. 2008;5:684-692.
36. Rapeli P, Fabritius C, Alho H, et al. Methadone vs. buprenorphine/naloxone during early opioid substitution treatment: a naturalistic comparison of cognitive performance relative to healthy controls. BMC Clin Pharmacol. 2007;7:5.
37. Wedam EF, Bigelow GE, Johnson RE, et al. QT-interval effects of methadone, levomethadyl, and buprenorphine in a randomized trial. Arch Intern Med. 2007;167:2469-2475.
38. Center for Substance Abuse Treatment. Clinical Guidelines for the Use of Buprenorphine in the Treatment of Opioid Addiction. Rockville, MD: Substance Abuse and Mental Health Services Administration; 2004. Treatment Improvement Protocol (TIP) Series 40. DHHS publication SMA 04‐3939.
39. Kleber HD. Methadone maintenance 4 decades later: thousands of lives saved but still controversial. JAMA. 2008;300:2303-2305.
40. Hulse GK, Morris N, Arnold-Reed D, et al. Improving clinical outcomes in treating heroin dependence: randomized, controlled trial of oral or implant naltrexone. Arch Gen Psychiatry. 2009;66:1108-1115.
41. FDA. FDA approves injectable drug to treat opioid-dependent patients. October 12, 2010. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/2010/ucm229109.htm. Accessed September 11, 2012.
42. Inturrisi CE, Verebely K. The levels of methadone in the plasma in methadone maintenance. Clin Pharmacol Ther. 1972;13(5 pt 1):633-637.
43. Stitzer M, Bigelow G, Lawrence C, et al. Medication take-home as a reinforcer in a methadone maintenance program. Addict Behav. 1977;2:9-14.
44. Code of Federal Regulations. Title 42.8.12. Federal Opioid Treatment Standards. October 2010.
45. Johnson RE. Chutuape MA, Strain EC, et al. A comparison of levomethadyl acetate, buprenorphine, and methadone for opioid dependence. N Engl J Med. 2000;343:1290-1297.
46. Krantz MJ, Martin J, Stimmel B, et al. QTc interval screening in methadone treatment. Ann Intern Med. 2009;150:387-395.
47. Ball JC, Lange WR, Myers CP, et al. Reducing the risk of AIDS through methadone maintenance treatment. J Health Soc Behav. 1988;29:214-226.
48. Fiellin DA, Pantalon MV, Chawarski MC, et al. Counseling plus buprenorphine-naloxone maintenance therapy for opioid dependence. N Engl J Med. 2006;355:365-374.
49. Defulio A, Everly JJ, Leoutsakos JM, et al. Employment-based reinforcement of adherence to an FDA approved extended release formulation of naltrexone in opioid-dependent adults: a randomized controlled trial. Drug Alcohol Depend. 2012;120:48-54.
50. Savage SR. Management of opioid medications in patients with chronic pain and risk of substance misuse. Curr Psychiatry Rep. 2009;11:377-384.
This article was originally published in the Journal of Family Practice. 2012;61:588-597.
Diagnosing and treating opioid dependence
• Ask all patients about the inappropriate use of substances, including prescription opioids. A
• Recommend pharmacotherapy for patients entering treatment for opioid dependence. A
• Warn patients who are opioid dependent about the risk of accidental fatal overdose, particularly with relapse. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE Sam M, age 48, is in your office for the first time in more than 2 years. He has gained a considerable amount of weight and appears a bit sluggish, and you wonder whether he’s depressed. While taking a history, Sam reminds you that he was laid off 16 months ago and had been caring for his wife, who sustained a debilitating back injury. When you saw her recently, she told you she’s back to work and pain-free. So you’re taken aback when Sam asks you to refill his wife’s oxycodone prescription for lingering pain that often keeps her up at night.
If Sam were your patient, would you suspect opioid dependence?
Dependence on opioid analgesics and the adverse consequences associated with it have steadily increased during the past decade. Consider the following:
- Between 2004 and 2008, the number of emergency department visits related to nonmedical prescription opioid use more than doubled, rising by 111%.1
- The increasing prevalence of opioid abuse has led to a recent spike in unintentional deaths,2 with the number of lives lost to opioid analgesic overdose now exceeding that of heroin or cocaine.3
- More than 75% of opioids used for nonmedical purposes were prescribed for someone else.4
The course of opioid use is highly variable. Some people start with a legitimate medical prescription for an opioid analgesic, then continue taking it after the pain subsides. Others experiment briefly with nonmedical prescription opioids or use them intermittently without adverse effect. Some progress from prescription opioids to heroin, despite its dangers.5 Still others have a catastrophic outcome, such as an overdose or severe accident, the first time they use opioids.6 Rapid progression from misuse of opioids to dependence is most likely in vulnerable populations, such as those with concurrent mental illness, other substance use disorders, or increased sensitivity to pain.7
Understanding the terms. Before we continue, a word about terminology is in order. “Misuse” generally refers to the use of a medication in a manner (ie, purpose, dose, or frequency) other than its intended use, while “drug addiction” is the repeated use of a drug despite resulting harm. Here we will use “opioid dependence” to mean a pattern of increasing use characterized by significant impairment and distress and an inability to stop, and “opioid withdrawal” to reflect a constellation of symptoms, such as insomnia, nausea, diarrhea, and muscle aches, that can follow physiological dependence (though not necessarily opioid dependence). Our definitions of these terms are consistent with those of the American Psychiatric Association (APA).8 Worth noting, however, is the fact that as the APA prepares for the publication of the 5th edition of its Diagnostic and Statistical Manual of Mental Disorders, its Substance Disorder Work Group has proposed replacing the term “opioid dependence” with “opioid use disorder” to reduce the confusion associated with these definitions.9
Assessing illicit opioid use: Start with a targeted question
Most patients who are opioid dependent do not seek treatment for it,10 and are typically free of medical sequelae associated with drug addiction when they see family practitioners. The absence of self-reporting and obvious physical signs and symptoms, coupled with the increase in illicit use of prescription opioids, underscores the need for family physicians to identify patients who are abusing opioids and ensure that they get the help they need.
Screening tools. There are a number of screening tools you can use for this purpose—eg, CAGE-Adapted to Include Drugs (CAGE-AID) and Drug Abuse Screening Test (DAST)11,12—but they have not been found to be significantly better than a careful substance abuse history.13
Straightforward questions. You can start by asking, “Do you take any medications for pain?” If the answer is Yes, get the name of the drug and inquire about the frequency of use and the route, the amount typically taken, and the duration of the current use pattern. Ask specifically about opioids when taking a substance abuse history. After a question about alcohol use, you can say, “Do you use any other drugs in a serious way? Marijuana? Opioids like Percocet, Vicodin, or Oxycontin?” Although it can be very difficult to detect opioid dependence if the patient is not forthcoming, other likely indicators of drug-seeking behavior should trigger additional questions. (See “Opioid dependence: Red flags to keep in mind”.14-16)
“Brief” protocols. Recent studies of Screening, Brief Intervention, and Referral to Treatment (SBIRT) programs have found that the simple, time-limited interventions they offer (visit http://www.samhsa.gov/prevention/sbirt/SBIRTwhitepaper.pdf to learn more) lead to a reduction in self-reported illicit opioid use.17,18 Family physicians can readily incorporate SBIRT protocols into routine practice, as an evidence-based and often reimbursable approach to substance abuse.17
Suspect opioid dependence in a patient who:
- describes pain resulting from back or orthopedic injuries without corresponding documentation or imaging
- requests a specific opioid for pain management
- shows little interest in a physical exam, diagnostic testing, or nonpharmacological remedies
- talks about changes in work or relationship status
- ceases to participate in activities or hobbies that previously occupied a considerable amount of his or her time. This may signal social isolation or indicate that the patient is spending a great deal of time in pursuit of opioids.
Additional steps before initiating treatment
After screening and diagnostic evaluation provide evidence that a patient is opioid dependent, you can take several steps to guide him or her to the appropriate treatment.
A thorough biopsychosocial assessment covering co-occurring psychiatric illnesses, pain, psychosocial stressors contributing to opioid use, and infectious disease screening is required to gain a clear picture of the patient’s situation. In every case, acute emergencies such as suicidal ideation require immediate intervention, which may involve hospitalization.19
Assess the patient’s desire for help. After the initial assessment, it is often helpful to categorize the patient’s “stage of change” (precontemplation, contemplation, preparation, action, or maintenance),20 and to tailor your next step accordingly. A patient who denies that opioid use is a problem or is clearly ambivalent about seeking treatment may require a conversation that uses principles of motivational interviewing—a collaborative approach that aims to evoke and strengthen personal motivation for change.21 Consider a question that encourages him or her to express reasons for change, such as: “How would you like your current situation to be different?” As almost everyone abusing opioids has thoughts about stopping, such a question may help the patient focus on specific changes.
CASE When you question Sam about his interest in oxycodone, he breaks down. He’s been unable to find work or to lose the excess weight he gained during the many months he cared for his wife. He tells you that soon after his wife stopped taking the pain pills, he started taking them. At first, he took one occasionally. Then he started taking the opioids every day, and finally, whenever he awakened at night. Now, Sam says, he has no more pills, and he’s nauseous, depressed, and unable to sleep—and looking to you for help.
Sam fits the criteria for opioid withdrawal as a result of physiological dependence; further questioning reveals that he also suffers from opioid dependence, and that he is receptive to treatment.
Recommending treatment and following up
Several options are available for patients who, like Sam, have signs and symptoms of opioid withdrawal as a result of physiological dependence. You can provide a referral to a physician specializing in addiction, recommend detoxification and/or treatment in an inpatient facility, or initiate pharmacological treatment and provide a referral to a behavioral therapist. Whatever the initial approach, most patients will ultimately be treated as outpatients, with a combination of pharmacotherapy and behavioral therapy—often, with monitoring and oversight by a primary care physician. Which approach to pursue should be guided by evidence-based recommendations (TABLE)17,22-27 and jointly decided by physician and patient.
TABLE
Treating opioid dependence: Key clinical recommendations
| Recommendation | Evidence (SOR) | Comments |
|---|---|---|
| Screen all patients for substance use, including opioids. Brief interventions and referral to treatment when appropriate may reduce opioid use17,22 | Consistent findings from RCTs; evidence-based guideline (A) | SBIRT reduces self-reported opioid use; efforts to replicate such reports with objective evidence (eg, toxicology screens) are underway |
| Recommend maintenance medication (ie, buprenorphine, naltrexone, methadone) for all patients entering treatment for opioid dependence with physiological dependence; methadone is the safest for pregnant women23-25 | Consistent findings from RCTs; evidence-based guideline (A) | Methadone is the gold standard for pregnant women; further studies are needed to determine the safety of in utero exposure to buprenorphine and naltrexone |
| Keep patients on maintenance medication for ≥3 months; higher relapse rates are noted when medication is discontinued in <3 months23,24 | Consistent findings from RCTs (A) | Relapse rates are higher when maintenance medication is discontinued in <3 months |
| Caution patients with opioid dependence of the risk for accidental overdose and death with relapse and take action—eg, offering naloxone rescue kits to patients and families, as appropriate26 | Consistent findings from RCTs and prospective cohort studies; evidence-based guideline (A) | |
| Take steps to prevent diversion and accidental ingestion of agonist therapies, using tools such as frequent toxicology screens, random pill counts, and designated pharmacies, and monitoring adherence to psychosocial treatment26,27 | Practice guideline (consensus) (C) | |
| RCTs, randomized clinical trials; SBIRT, Screening, Brief Intervention, and Referral to Treatment; SOR, strength of recommendation. | ||
Medication plays a key role in recovery
Recommend medication-assisted treatment, either with an agonist (buprenorphine or methadone) or an antagonist (naltrexone), for every patient with physiological opioid dependence. The goals of pharmacotherapy are to prevent or reduce withdrawal symptoms and craving, avoid relapse, and restore to a normal state any physiological functions (eg, sleep, bowel movements) that have been disrupted by opioid use.28 When continued for ≥3 months, medication has been shown to improve outcomes.23,24,29 In one recent study, 49% of opioid-dependent participants who were still taking buprenorphine-naloxone at 12 weeks had successful outcomes (minimal or no opioid use), vs 7% of those undergoing a brief buprenorphine-naloxone taper.24
There are risks associated with medication-assisted therapy, however. The ones of greatest concern are a potential increase in drug-drug interactions, the risk of diversion (a concern with both buprenorphine and methadone), and the potential for accidental overdose.2,30
Buprenorphine, a partial mu-opioid receptor agonist, is a Schedule III controlled substance and can be dispensed by a pharmacy, making inpatient opioid detoxification unnecessary for many opioid-dependent patients. Physicians who wish to prescribe buprenorphine for the treatment of opioid dependence must complete an 8-hour course, offered by the American Medical Association and the APA, among other medical groups, and obtain a Drug Enforcement Administration code (“X”) license. 31
Buprenorphine has a high affinity for, and a slow dissociation from, mu-opioid receptors, resulting in the displacement of other opioids from the mu receptor and less severe withdrawal.32 As a partial agonist, buprenorphine attenuates opioid withdrawal symptoms with a ceiling, or near maximal, effect at 16 mg, thereby lowering the risk for overdose.33 A sublingual formulation that combines buprenorphine with naloxone, an opioid antagonist that exerts its full effect when injected but is minimally absorbed sublingually, reduces the potential for abuse of buprenorphine without interfering with its effectiveness.34
Compared with methadone, buprenorphine is less likely to interact with antiretroviral medications or to cause QTc prolongation, erectile dysfunction, or cognitive or psychomotor impairment.31,35-37 Limitations include the ceiling effect, which can be a problem for cases in which more agonist is needed; cost (approximately $12/d), and the lack of approval by the US Food and Drug Administration (FDA) for use during pregnancy.
Buprenorphine maintenance involves 3 phases: induction, stabilization, and maintenance.38 Induction takes place in a clinician’s office at the time the patient experiences opioid withdrawal symptoms, typically 6 to 48 hours after taking the last opioid. Extended treatment improves clinical outcomes,23,24 and longer-term maintenance (of indefinite duration) is frequently required.
Naltrexone is a mu-receptor antagonist, and therefore does not cause physical dependence or have agonist effects such as euphoria and sedation. As a result, it has no diversion value and may appeal to those who view opioid-agonist pharmacotherapy as simply trading one drug for another.39 Naltrexone is not a controlled substance and is not subject to the regulatory requirements that buprenorphine and methadone face.
Although agonists can be started in the first day or 2 after a patient decides to stop using opioids, patients must be opioid-free for ≥7 days before starting naltrexone. That’s because its antagonist properties will precipitate withdrawal if another opioid is present on the opioid receptors. During the 7-day “washout” period, you can treat opioid withdrawal symptoms with medications such as clonidine and dicyclomine, but such symptoms make patients especially vulnerable to relapse while waiting to start naltrexone.
Oral naltrexone’s effectiveness as a treatment for opioid dependence has been limited by poor adherence. But a long-acting intramuscular form of the drug, approved by the FDA in 2010 and requiring once-a-month injection, mitigates this concern.40,41
Methadone is a full mu-opioid agonist, administered daily at specialized clinics, as a maintenance therapy for opioid dependence. Although office-based physicians can prescribe methadone for pain, the drug can only be used for opioid dependence under the auspices of state- and federally regulated opioid treatment programs (http://findtreatment.samhsa.gov/TreatmentLocator/faces/quickSearch.jspx; a mobile phone application is also available at http://www.samhsa.gov/mobile/treatmentlocator.aspx).
Methadone, a Schedule III controlled substance with a half-life averaging 24 to 36 hours, requires daily dosing.42 Its slow metabolism and long half-life increase the risk for overdose.
Methadone is best for patients who are highly dependent on opioids and likely to benefit from a structured treatment environment with daily supervision (although patients who are doing well may earn take-home privileges so they don’t have to come to the clinic every day).43 New patients should receive an initial dose of 30 mg or less, and a maximum first-day dose of 40 mg.44
Methadone remains the standard of care for pregnant women being treated for opioid dependence, while studies of the effects of buprenorphine and naltrexone on a developing fetus continue. Although methadone’s efficacy, particularly in lower doses, is similar to that of buprenorphine,45 its adverse effect profile is worse. Adverse effects include drug-drug interactions, the potential for respiratory depression (especially when combined with alcohol or sedatives), QTc prolongation (which requires monitoring by electrocardiogram), sedation, and weight gain, and should be considered before selecting methadone as a maintenance pharmacotherapy.30,37,46 And, because relapse rates within 12 months of tapering off methadone have been reported to exceed 80%,47 both the clinician and the patient need to consider the likelihood of long-term, even lifelong, maintenance before initiating treatment.
Behavioral interventions are a vital part of the picture
Studies evaluating the extent to which various types and amounts of counseling improve outcomes compared with pharmacotherapy alone have had conflicting results.24,48 Nonetheless, most clinicians consider counseling to be a critical component of treatment for opioid dependence and recommend, at a minimum, either individual or group counseling (various modalities have been shown to be effective) and regular attendance at a self-help group like Narcotics Anonymous. Contingency management, a type of therapy that uses prizes as incentives for desired behaviors; and family therapy, individual counseling, and community-based programs have all been found to improve outcomes.6,49
CASE You refer Sam to an addiction psychiatrist, who stabilizes him on 16 mg buprenorphine/naloxone daily as part of an outpatient treatment program. Sam is enrolled in a weekly buprenorphine stabilization group, where he gives a urine sample each week. He also begins seeing a social worker weekly for counseling and attends Narcotics Anonymous meetings 2 to 3 times a week. At a follow-up appointment with you 6 months later, he reports that he has been abstinent from oxycodone for 6 months, his sleep is improved, and he feels better about his chances of finding another job.
Your role in safeguarding the patient
With the rising prevalence of opioid overdose, patient education aimed at crisis prevention is crucial, as well. Warn patients of the risk of accidental overdose, often associated with relapse, stressing the importance of continuing treatment and taking their maintenance medication exactly as prescribed.
There are other steps you can take to safeguard patients—eg, providing naloxone rescue kits to patients and their families when appropriate. You can also institute diversion and overdose prevention measures for patients taking buprenorphine or methadone—providing a lock box for take-home medication, implementing treatment contracts, and using a designated pharmacy to dispense buprenorphine, for example.26,27,50
Regular monitoring, urine drug screens (see TABLE W1), and random pill counts, in which patients are typically given 24 hours to bring in their prescribed medication so it can be counted, can also help keep patients on track. Treatment for concurrent psychiatric disorders—depression, anxiety, and personality disorders are common among patients with opioid dependence—is likely to improve the outcome of treatment, as well.
TABLE W1
Pharmacokinetics of common opioids: Time detectable in urine*
| Drug (half-life) | Time detectable in urine | Comment |
|---|---|---|
| Codeine (2.5-3 h) | 48 h | Pharmacogenetic-dependent effects may affect detection |
| Fentanyl Transdermal (17 h) Submucosal (7 h) | Not usually detected in urine (lack of metabolites) | Excretion of transdermal fentanyl can last days |
| Hydromorphone IR (2.3 h) ER (18.6 h) | 2-4 d | Significant interpatient variability |
| Methadone (8-59 h) | 3 d | |
| Morphine (1.5-2 h) | 48-72 h | 90% eliminated within 24 h |
| Oxycodone IR (3.2 h) ER (4.5 h) | Often not detected in urine | High-fat meals may increase serum concentrations of ER formulation |
| Propoxyphene Parent drug (6-12 h) Metabolite (30-36 h) | 6-48 h | |
| ER, extended release; IR, immediate release. *Previously appeared in: McBane S, Weige N. Is it time to drug test your chronic pain patient? J Fam Pract. 2010;59:628-633. Sources: Clinical Pharmacology [online]. Tampa, FL: Gold Standard Inc; 2010. Available at: http://cp.gsm.com. Accessed March 5, 2010; Drug Facts and Comparisons [online]. 2010. Available at: http://www.factsandcomparisons.com/. Accessed March 5, 2010. | ||
CORRESPONDENCE Kevin P. Hill, MD, MHS, McLean Hospital, 115 Mill Street, Belmont, MA 02478; [email protected]
1. Centers for Disease Control and Prevention (CDC). Emergency department visits involving nonmedical use of selected prescription drugs - United States, 2004-2008. MMWR Morb Mortal Wkly Rep. 2010;59:705-709.
2. Bohnert AS, Valenstein M, Bair MJ, et al. Association between opioid prescribing patterns and opioid overdose-related deaths. JAMA. 2011;305:1315-1321.
3. Warner M, Chen LH, Makuc DM. Increase in fatal poisonings involving opioid analgesics in the United States, 1999-2006. NCHS Data Brief. 2009;(22):1-8.4.
4. Substance Abuse and Mental Health Services Administration. Results From the 2009 National Survey on Drug Use and Health: Volume I. Summary of National Findings. Rockville, Md: SAMHSA, Office of Applied Studies; 2010. NSDUH Series H-38A, HHS publication SMA 10-4856. Available at: http://www.samhsa.gov/data/NSDUH/2k9NSDUH/2k9Results.htm. Accessed August 22, 2012.
5. Hser YI, Huang D, Brecht ML, et al. Contrasting trajectories of heroin, cocaine, and methamphetamine use. J Addict Dis. 2008;27:13-21.
6. Veilleux JC, Colvin PJ, Anderson J, et al. A review of opioid dependence treatment: pharmacological and psychosocial interventions to treat opioid addiction. Clin Psychol Rev. 2011;30:155-166.
7. George O, Koob GF. Individual differences in prefrontal cortex function and the transition from drug use to drug dependence. Neurosci Biobehav Rev. 2011;35:232-247.
8. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 4th ed, text rev (DSM-IV-TR). Arlington, Va: American Psychiatric Association; 2000.
9. American Psychiatric Association. R 19 opioid use disorder. http://www.dsm5.org/ProposedRevisions/Pages/proposedrevision.aspx?rid=460. Updated April 30, 2012. Accessed June 20, 2012.
10. Substance Abuse and Mental Health Services Administration. Results From the 2008 National Survey on Drug Use and Health: National Findings. Rockville, Md: SAMHSA, Office of Applied Studies; 2009. NSDUH Series H-36, HHS publication SMA 09-4434. Available at: http://www.samhsa.gov/data/nsduh/2k8nsduh/2k8Results.htm. Accessed August 22, 2012.
11. Brown RL, Rounds LA. Conjoint screening questionnaires for alcohol and other drug abuse: criterion validity in a primary care practice. Wis Med J. 1995;94:135-140.
12. Skinner HA. The drug abuse screening test. Addict Behav. 1982;7:363-371.
13. US Preventive Services Task Force. Screening for Illicit Drug Use: U.S. Preventive Services Task Force Recommendation Statement. January 2008. Available at: http://www.uspreventiveservicestaskforce.org/uspstf08/druguse/drugrs.htm. Accessed May 7, 2012.
14. Gourlay D, Caplan Y, Heit H. Urine Drug Testing in Clinical Practice: Dispelling the Myths and Designing Strategies. San Francisco, Calif: California Academy of Family Physicians; 2006.
15. Jackman R, Purvis J, Mallett B. Chronic nonmalignant pain in primary care. Am Fam Physician. 2008;78:1155-1162.
16. McBane S, Weigle N. Is it time to drug test your chronic pain patient? J Fam Pract. 2010;59:628-633.
17. Madras BK, Compton WM, Avula D, et al. Screening, brief interventions, referral to treatment (SBIRT) for illicit drug and alcohol use at multiple healthcare sites: comparison at intake and 6 months later. Drug Alcohol Depend. 2009;99:280-295.
18. The InSight Project Research Group. SBIRT outcomes in Houston: final report on InSight, a hospital district-based program for patients at risk for alcohol or drug use problems. Alcohol Clin Exp Res. 2009;33:1374-1381.
19. Borges G, Walters EE, Kessler RC. Associations of substance use, abuse, and dependence with subsequent suicidal behavior. Am J Epidemiol. 2000;151:781-789.
20. Prochaska JO, DiClemente CC. Stages and processes of self-change of smoking: toward an integrative model of change. J Consult Clin Psychol. 1983;51:390-395.
21. Smedslund G, Berg RC, Hammerstrom KT, et al. Motivational interviewing for substance abuse. Cochrane Database Syst Rev. 2011;(5):CD008063.-
22. Gryczynski J, Mitchell SG, Peterson TR, et al. The relationship between services delivered and substance use outcomes in New Mexico’s Screening, Brief Intervention, Referral and Treatment (SBIRT) Initiative. Drug Alcohol Depend. 2011;118:152-157.
23. Woody GE, Poole SA, Subramaniam G, et al. Extended vs short-term buprenorphine-naloxone for treatment of opioid-addicted youth: a randomized trial. JAMA. 2008;300:2003-2011.
24. Weiss RD, Potter JS, Fiellin DA, et al. Adjunctive counseling during brief and extended buprenorphine-naloxone treatment for prescription opioid dependence: a 2-phase randomized controlled trial. Arch Gen Psychiatry. 2011;68:1238-1246.
25. Johansson BA, Berglund M, Lindgren A. Efficacy of maintenance treatment with naltrexone for opioid dependence: a meta-analytical review. Addiction. 2006;101:491-503.
26. Hall AJ, Logan JE, Toblin RL, et al. Patterns of abuse among unintentional pharmaceutical overdose fatalities. JAMA. 2008;300:2613-2620.
27. Zacny J, Bigelow G, Compton P, et al. College on Problems of Drug Dependence taskforce on prescription opioid non-medical use and abuse: position statement. Drug Alcohol Depend. 2003;69:215-232.
28. Kreek MJ. Rationale for maintenance pharmacotherapy of opiate dependence. Res Publ Assoc Res Nerv Ment Dis. 1992;70:205-230.
29. Mattick RP, Breen C, Kimber J, et al. Methadone maintenance therapy versus no opioid replacement therapy for opioid dependence. Cochrane Database Syst Rev. 2009;(3):CD002209.-
30. McCance-Katz EF, Sullivan LE, Nallani S. Drug interactions of clinical importance among the opioids, methadone and buprenorphine, and other frequently prescribed medications: a review. Am J Addict. 2010;19:4-16.
31. Office of National Drug Control Policy Reauthorization Act of 2006 (ONDCPRA), HR 6344, 109th Cong, 2nd Sess (2006).
32. Lewis JW, Walter D. Buprenorphine—background to its development as a treatment for opiate dependence. In: Blaine JD, ed. Buprenorphine: An Alternative Treatment for Opioid Dependence. Rockville, Md: National Institute on Drug Abuse; 1992:5-11. NIDA Research Monograph, No. 121. Available at: http://archives.drugabuse.gov/pdf/monographs/121.pdf. Accessed August 22, 2012.
33. Walsh SL, Preston KL, Stitzer ML, et al. Clinical pharmacology of buprenorphine: ceiling effects at high doses. Clin Pharmacol Ther. 1994;55:569-580.
34. Alho H, Sinclair D, Vuori E, et al. Abuse liability of buprenorphine-naloxone tablets in untreated IV drug users. Drug Alcohol Depend. 2007;88:75-78.
35. Hallinan R, Byrne A, Agho K, et al. Erectile dysfunction in men receiving methadone and buprenorphine maintenance treatment. J Sex Med. 2008;5:684-692.
36. Rapeli P, Fabritius C, Alho H, et al. Methadone vs. buprenorphine/naloxone during early opioid substitution treatment: a naturalistic comparison of cognitive performance relative to healthy controls. BMC Clin Pharmacol. 2007;7:5.-
37. Wedam EF, Bigelow GE, Johnson RE, et al. QT-interval effects of methadone, levomethadyl, and buprenorphine in a randomized trial. Arch Intern Med. 2007;167:2469-2475.
38. Center for Substance Abuse Treatment. Clinical Guidelines for the Use of Buprenorphine in the Treatment of Opioid Addiction. Rockville, Md: Substance Abuse and Mental Health Services Administration; 2004. Treatment Improvement Protocol (TIP) Series 40. DHHS publication SMA 04-3939.
39. Kleber HD. Methadone maintenance 4 decades later: thousands of lives saved but still controversial. JAMA. 2008;300:2303-2305.
40. Hulse GK, Morris N, Arnold-Reed D, et al. Improving clinical outcomes in treating heroin dependence: randomized, controlled trial of oral or implant naltrexone. Arch Gen Psychiatry. 2009;66:1108-1115.
41. US Food and Drug Administration. FDA approves injectable drug to treat opioid-dependent patients. October 12, 2010. Available at: http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/2010/ucm229109.htm. Accessed September 11, 2012.
42. Inturrisi CE, Verebely K. The levels of methadone in the plasma in methadone maintenance. Clin Pharmacol Ther. 1972;13 (5 pt 1):633-637.
43. Stitzer M, Bigelow G, Lawrence C, et al. Medication take-home as a reinforcer in a methadone maintenance program. Addict Behav. 1977;2:9-14.
44. Code of Federal Regulations. Title 42.8.12. Federal Opioid Treatment Standards. October 2010.
45. Johnson RE, Chutuape MA, Strain EC, et al. A comparison of levomethadyl acetate, buprenorphine, and methadone for opioid dependence. N Engl J Med. 2000;343:1290-1297.
46. Krantz MJ, Martin J, Stimmel B, et al. QTc interval screening in methadone treatment. Ann Intern Med. 2009;150:387-395.
47. Ball JC, Lange WR, Myers CP, et al. Reducing the risk of AIDS through methadone maintenance treatment. J Health Soc Behav. 1988;29:214-226.
48. Fiellin DA, Pantalon MV, Chawarski MC, et al. Counseling plus buprenorphine-naloxone maintenance therapy for opioid dependence. N Engl J Med. 2006;355:365-374.
49. Defulio A, Everly JJ, Leoutsakos JM, et al. Employment-based reinforcement of adherence to an FDA approved extended release formulation of naltrexone in opioid-dependent adults: a randomized controlled trial. Drug Alcohol Depend. 2012;120:48-54.
50. Savage SR. Management of opioid medications in patients with chronic pain and risk of substance misuse. Curr Psychiatry Rep. 2009;11:377-384.
• Ask all patients about the inappropriate use of substances, including prescription opioids. A
• Recommend pharmacotherapy for patients entering treatment for opioid dependence. A
• Warn patients who are opioid dependent about the risk of accidental fatal overdose, particularly with relapse. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE Sam M, age 48, is in your office for the first time in more than 2 years. He has gained a considerable amount of weight and appears a bit sluggish, and you wonder whether he’s depressed. While taking a history, Sam reminds you that he was laid off 16 months ago and had been caring for his wife, who sustained a debilitating back injury. When you saw her recently, she told you she’s back to work and pain-free. So you’re taken aback when Sam asks you to refill his wife’s oxycodone prescription for lingering pain that often keeps her up at night.
If Sam were your patient, would you suspect opioid dependence?
Dependence on opioid analgesics and the adverse consequences associated with it have steadily increased during the past decade. Consider the following:
- Between 2004 and 2008, the number of emergency department visits related to nonmedical prescription opioid use more than doubled, rising by 111%.1
- The increasing prevalence of opioid abuse has led to a recent spike in unintentional deaths,2 with the number of lives lost to opioid analgesic overdose now exceeding that of heroin or cocaine.3
- More than 75% of opioids used for nonmedical purposes were prescribed for someone else.4
The course of opioid use is highly variable. Some people start with a legitimate medical prescription for an opioid analgesic, then continue taking it after the pain subsides. Others experiment briefly with nonmedical prescription opioids or use them intermittently without adverse effect. Some progress from prescription opioids to heroin, despite its dangers.5 Still others have a catastrophic outcome, such as an overdose or severe accident, the first time they use opioids.6 Rapid progression from misuse of opioids to dependence is most likely in vulnerable populations, such as those with concurrent mental illness, other substance use disorders, or increased sensitivity to pain.7
Understanding the terms. Before we continue, a word about terminology is in order. “Misuse” generally refers to the use of a medication in a manner (ie, purpose, dose, or frequency) other than its intended use, while “drug addiction” is the repeated use of a drug despite resulting harm. Here we will use “opioid dependence” to mean a pattern of increasing use characterized by significant impairment and distress and an inability to stop, and “opioid withdrawal” to reflect a constellation of symptoms, such as insomnia, nausea, diarrhea, and muscle aches, that can follow physiological dependence (though not necessarily opioid dependence). Our definitions of these terms are consistent with those of the American Psychiatric Association (APA).8 Worth noting, however, is the fact that as the APA prepares for the publication of the 5th edition of its Diagnostic and Statistical Manual of Mental Disorders, its Substance Disorder Work Group has proposed replacing the term “opioid dependence” with “opioid use disorder” to reduce the confusion associated with these definitions.9
Assessing illicit opioid use: Start with a targeted question
Most patients who are opioid dependent do not seek treatment for it,10 and are typically free of medical sequelae associated with drug addiction when they see family practitioners. The absence of self-reporting and obvious physical signs and symptoms, coupled with the increase in illicit use of prescription opioids, underscores the need for family physicians to identify patients who are abusing opioids and ensure that they get the help they need.
Screening tools. There are a number of screening tools you can use for this purpose—eg, CAGE-Adapted to Include Drugs (CAGE-AID) and Drug Abuse Screening Test (DAST)11,12—but they have not been found to be significantly better than a careful substance abuse history.13
Straightforward questions. You can start by asking, “Do you take any medications for pain?” If the answer is Yes, get the name of the drug and inquire about the frequency of use and the route, the amount typically taken, and the duration of the current use pattern. Ask specifically about opioids when taking a substance abuse history. After a question about alcohol use, you can say, “Do you use any other drugs in a serious way? Marijuana? Opioids like Percocet, Vicodin, or Oxycontin?” Although it can be very difficult to detect opioid dependence if the patient is not forthcoming, other likely indicators of drug-seeking behavior should trigger additional questions. (See “Opioid dependence: Red flags to keep in mind”.14-16)
“Brief” protocols. Recent studies of Screening, Brief Intervention, and Referral to Treatment (SBIRT) programs have found that the simple, time-limited interventions they offer (visit http://www.samhsa.gov/prevention/sbirt/SBIRTwhitepaper.pdf to learn more) lead to a reduction in self-reported illicit opioid use.17,18 Family physicians can readily incorporate SBIRT protocols into routine practice, as an evidence-based and often reimbursable approach to substance abuse.17
Suspect opioid dependence in a patient who:
- describes pain resulting from back or orthopedic injuries without corresponding documentation or imaging
- requests a specific opioid for pain management
- shows little interest in a physical exam, diagnostic testing, or nonpharmacological remedies
- talks about changes in work or relationship status
- ceases to participate in activities or hobbies that previously occupied a considerable amount of his or her time. This may signal social isolation or indicate that the patient is spending a great deal of time in pursuit of opioids.
Additional steps before initiating treatment
After screening and diagnostic evaluation provide evidence that a patient is opioid dependent, you can take several steps to guide him or her to the appropriate treatment.
A thorough biopsychosocial assessment covering co-occurring psychiatric illnesses, pain, psychosocial stressors contributing to opioid use, and infectious disease screening is required to gain a clear picture of the patient’s situation. In every case, acute emergencies such as suicidal ideation require immediate intervention, which may involve hospitalization.19
Assess the patient’s desire for help. After the initial assessment, it is often helpful to categorize the patient’s “stage of change” (precontemplation, contemplation, preparation, action, or maintenance),20 and to tailor your next step accordingly. A patient who denies that opioid use is a problem or is clearly ambivalent about seeking treatment may require a conversation that uses principles of motivational interviewing—a collaborative approach that aims to evoke and strengthen personal motivation for change.21 Consider a question that encourages him or her to express reasons for change, such as: “How would you like your current situation to be different?” As almost everyone abusing opioids has thoughts about stopping, such a question may help the patient focus on specific changes.
CASE When you question Sam about his interest in oxycodone, he breaks down. He’s been unable to find work or to lose the excess weight he gained during the many months he cared for his wife. He tells you that soon after his wife stopped taking the pain pills, he started taking them. At first, he took one occasionally. Then he started taking the opioids every day, and finally, whenever he awakened at night. Now, Sam says, he has no more pills, and he’s nauseous, depressed, and unable to sleep—and looking to you for help.
Sam fits the criteria for opioid withdrawal as a result of physiological dependence; further questioning reveals that he also suffers from opioid dependence, and that he is receptive to treatment.
Recommending treatment and following up
Several options are available for patients who, like Sam, have signs and symptoms of opioid withdrawal as a result of physiological dependence. You can provide a referral to a physician specializing in addiction, recommend detoxification and/or treatment in an inpatient facility, or initiate pharmacological treatment and provide a referral to a behavioral therapist. Whatever the initial approach, most patients will ultimately be treated as outpatients, with a combination of pharmacotherapy and behavioral therapy—often, with monitoring and oversight by a primary care physician. Which approach to pursue should be guided by evidence-based recommendations (TABLE)17,22-27 and jointly decided by physician and patient.
TABLE
Treating opioid dependence: Key clinical recommendations
| Recommendation | Evidence (SOR) | Comments |
|---|---|---|
| Screen all patients for substance use, including opioids. Brief interventions and referral to treatment when appropriate may reduce opioid use17,22 | Consistent findings from RCTs; evidence-based guideline (A) | SBIRT reduces self-reported opioid use; efforts to replicate such reports with objective evidence (eg, toxicology screens) are underway |
| Recommend maintenance medication (ie, buprenorphine, naltrexone, methadone) for all patients entering treatment for opioid dependence with physiological dependence; methadone is the safest for pregnant women23-25 | Consistent findings from RCTs; evidence-based guideline (A) | Methadone is the gold standard for pregnant women; further studies are needed to determine the safety of in utero exposure to buprenorphine and naltrexone |
| Keep patients on maintenance medication for ≥3 months; higher relapse rates are noted when medication is discontinued in <3 months23,24 | Consistent findings from RCTs (A) | Relapse rates are higher when maintenance medication is discontinued in <3 months |
| Caution patients with opioid dependence of the risk for accidental overdose and death with relapse and take action—eg, offering naloxone rescue kits to patients and families, as appropriate26 | Consistent findings from RCTs and prospective cohort studies; evidence-based guideline (A) | |
| Take steps to prevent diversion and accidental ingestion of agonist therapies, using tools such as frequent toxicology screens, random pill counts, and designated pharmacies, and monitoring adherence to psychosocial treatment26,27 | Practice guideline (consensus) (C) | |
| RCTs, randomized clinical trials; SBIRT, Screening, Brief Intervention, and Referral to Treatment; SOR, strength of recommendation. | ||
Medication plays a key role in recovery
Recommend medication-assisted treatment, either with an agonist (buprenorphine or methadone) or an antagonist (naltrexone), for every patient with physiological opioid dependence. The goals of pharmacotherapy are to prevent or reduce withdrawal symptoms and craving, avoid relapse, and restore to a normal state any physiological functions (eg, sleep, bowel movements) that have been disrupted by opioid use.28 When continued for ≥3 months, medication has been shown to improve outcomes.23,24,29 In one recent study, 49% of opioid-dependent participants who were still taking buprenorphine-naloxone at 12 weeks had successful outcomes (minimal or no opioid use), vs 7% of those undergoing a brief buprenorphine-naloxone taper.24
There are risks associated with medication-assisted therapy, however. The ones of greatest concern are a potential increase in drug-drug interactions, the risk of diversion (a concern with both buprenorphine and methadone), and the potential for accidental overdose.2,30
Buprenorphine, a partial mu-opioid receptor agonist, is a Schedule III controlled substance and can be dispensed by a pharmacy, making inpatient opioid detoxification unnecessary for many opioid-dependent patients. Physicians who wish to prescribe buprenorphine for the treatment of opioid dependence must complete an 8-hour course, offered by the American Medical Association and the APA, among other medical groups, and obtain a Drug Enforcement Administration code (“X”) license. 31
Buprenorphine has a high affinity for, and a slow dissociation from, mu-opioid receptors, resulting in the displacement of other opioids from the mu receptor and less severe withdrawal.32 As a partial agonist, buprenorphine attenuates opioid withdrawal symptoms with a ceiling, or near maximal, effect at 16 mg, thereby lowering the risk for overdose.33 A sublingual formulation that combines buprenorphine with naloxone, an opioid antagonist that exerts its full effect when injected but is minimally absorbed sublingually, reduces the potential for abuse of buprenorphine without interfering with its effectiveness.34
Compared with methadone, buprenorphine is less likely to interact with antiretroviral medications or to cause QTc prolongation, erectile dysfunction, or cognitive or psychomotor impairment.31,35-37 Limitations include the ceiling effect, which can be a problem for cases in which more agonist is needed; cost (approximately $12/d), and the lack of approval by the US Food and Drug Administration (FDA) for use during pregnancy.
Buprenorphine maintenance involves 3 phases: induction, stabilization, and maintenance.38 Induction takes place in a clinician’s office at the time the patient experiences opioid withdrawal symptoms, typically 6 to 48 hours after taking the last opioid. Extended treatment improves clinical outcomes,23,24 and longer-term maintenance (of indefinite duration) is frequently required.
Naltrexone is a mu-receptor antagonist, and therefore does not cause physical dependence or have agonist effects such as euphoria and sedation. As a result, it has no diversion value and may appeal to those who view opioid-agonist pharmacotherapy as simply trading one drug for another.39 Naltrexone is not a controlled substance and is not subject to the regulatory requirements that buprenorphine and methadone face.
Although agonists can be started in the first day or 2 after a patient decides to stop using opioids, patients must be opioid-free for ≥7 days before starting naltrexone. That’s because its antagonist properties will precipitate withdrawal if another opioid is present on the opioid receptors. During the 7-day “washout” period, you can treat opioid withdrawal symptoms with medications such as clonidine and dicyclomine, but such symptoms make patients especially vulnerable to relapse while waiting to start naltrexone.
Oral naltrexone’s effectiveness as a treatment for opioid dependence has been limited by poor adherence. But a long-acting intramuscular form of the drug, approved by the FDA in 2010 and requiring once-a-month injection, mitigates this concern.40,41
Methadone is a full mu-opioid agonist, administered daily at specialized clinics, as a maintenance therapy for opioid dependence. Although office-based physicians can prescribe methadone for pain, the drug can only be used for opioid dependence under the auspices of state- and federally regulated opioid treatment programs (http://findtreatment.samhsa.gov/TreatmentLocator/faces/quickSearch.jspx; a mobile phone application is also available at http://www.samhsa.gov/mobile/treatmentlocator.aspx).
Methadone, a Schedule III controlled substance with a half-life averaging 24 to 36 hours, requires daily dosing.42 Its slow metabolism and long half-life increase the risk for overdose.
Methadone is best for patients who are highly dependent on opioids and likely to benefit from a structured treatment environment with daily supervision (although patients who are doing well may earn take-home privileges so they don’t have to come to the clinic every day).43 New patients should receive an initial dose of 30 mg or less, and a maximum first-day dose of 40 mg.44
Methadone remains the standard of care for pregnant women being treated for opioid dependence, while studies of the effects of buprenorphine and naltrexone on a developing fetus continue. Although methadone’s efficacy, particularly in lower doses, is similar to that of buprenorphine,45 its adverse effect profile is worse. Adverse effects include drug-drug interactions, the potential for respiratory depression (especially when combined with alcohol or sedatives), QTc prolongation (which requires monitoring by electrocardiogram), sedation, and weight gain, and should be considered before selecting methadone as a maintenance pharmacotherapy.30,37,46 And, because relapse rates within 12 months of tapering off methadone have been reported to exceed 80%,47 both the clinician and the patient need to consider the likelihood of long-term, even lifelong, maintenance before initiating treatment.
Behavioral interventions are a vital part of the picture
Studies evaluating the extent to which various types and amounts of counseling improve outcomes compared with pharmacotherapy alone have had conflicting results.24,48 Nonetheless, most clinicians consider counseling to be a critical component of treatment for opioid dependence and recommend, at a minimum, either individual or group counseling (various modalities have been shown to be effective) and regular attendance at a self-help group like Narcotics Anonymous. Contingency management, a type of therapy that uses prizes as incentives for desired behaviors; and family therapy, individual counseling, and community-based programs have all been found to improve outcomes.6,49
CASE You refer Sam to an addiction psychiatrist, who stabilizes him on 16 mg buprenorphine/naloxone daily as part of an outpatient treatment program. Sam is enrolled in a weekly buprenorphine stabilization group, where he gives a urine sample each week. He also begins seeing a social worker weekly for counseling and attends Narcotics Anonymous meetings 2 to 3 times a week. At a follow-up appointment with you 6 months later, he reports that he has been abstinent from oxycodone for 6 months, his sleep is improved, and he feels better about his chances of finding another job.
Your role in safeguarding the patient
With the rising prevalence of opioid overdose, patient education aimed at crisis prevention is crucial, as well. Warn patients of the risk of accidental overdose, often associated with relapse, stressing the importance of continuing treatment and taking their maintenance medication exactly as prescribed.
There are other steps you can take to safeguard patients—eg, providing naloxone rescue kits to patients and their families when appropriate. You can also institute diversion and overdose prevention measures for patients taking buprenorphine or methadone—providing a lock box for take-home medication, implementing treatment contracts, and using a designated pharmacy to dispense buprenorphine, for example.26,27,50
Regular monitoring, urine drug screens (see TABLE W1), and random pill counts, in which patients are typically given 24 hours to bring in their prescribed medication so it can be counted, can also help keep patients on track. Treatment for concurrent psychiatric disorders—depression, anxiety, and personality disorders are common among patients with opioid dependence—is likely to improve the outcome of treatment, as well.
TABLE W1
Pharmacokinetics of common opioids: Time detectable in urine*
| Drug (half-life) | Time detectable in urine | Comment |
|---|---|---|
| Codeine (2.5-3 h) | 48 h | Pharmacogenetic-dependent effects may affect detection |
| Fentanyl Transdermal (17 h) Submucosal (7 h) | Not usually detected in urine (lack of metabolites) | Excretion of transdermal fentanyl can last days |
| Hydromorphone IR (2.3 h) ER (18.6 h) | 2-4 d | Significant interpatient variability |
| Methadone (8-59 h) | 3 d | |
| Morphine (1.5-2 h) | 48-72 h | 90% eliminated within 24 h |
| Oxycodone IR (3.2 h) ER (4.5 h) | Often not detected in urine | High-fat meals may increase serum concentrations of ER formulation |
| Propoxyphene Parent drug (6-12 h) Metabolite (30-36 h) | 6-48 h | |
| ER, extended release; IR, immediate release. *Previously appeared in: McBane S, Weige N. Is it time to drug test your chronic pain patient? J Fam Pract. 2010;59:628-633. Sources: Clinical Pharmacology [online]. Tampa, FL: Gold Standard Inc; 2010. Available at: http://cp.gsm.com. Accessed March 5, 2010; Drug Facts and Comparisons [online]. 2010. Available at: http://www.factsandcomparisons.com/. Accessed March 5, 2010. | ||
CORRESPONDENCE Kevin P. Hill, MD, MHS, McLean Hospital, 115 Mill Street, Belmont, MA 02478; [email protected]
• Ask all patients about the inappropriate use of substances, including prescription opioids. A
• Recommend pharmacotherapy for patients entering treatment for opioid dependence. A
• Warn patients who are opioid dependent about the risk of accidental fatal overdose, particularly with relapse. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE Sam M, age 48, is in your office for the first time in more than 2 years. He has gained a considerable amount of weight and appears a bit sluggish, and you wonder whether he’s depressed. While taking a history, Sam reminds you that he was laid off 16 months ago and had been caring for his wife, who sustained a debilitating back injury. When you saw her recently, she told you she’s back to work and pain-free. So you’re taken aback when Sam asks you to refill his wife’s oxycodone prescription for lingering pain that often keeps her up at night.
If Sam were your patient, would you suspect opioid dependence?
Dependence on opioid analgesics and the adverse consequences associated with it have steadily increased during the past decade. Consider the following:
- Between 2004 and 2008, the number of emergency department visits related to nonmedical prescription opioid use more than doubled, rising by 111%.1
- The increasing prevalence of opioid abuse has led to a recent spike in unintentional deaths,2 with the number of lives lost to opioid analgesic overdose now exceeding that of heroin or cocaine.3
- More than 75% of opioids used for nonmedical purposes were prescribed for someone else.4
The course of opioid use is highly variable. Some people start with a legitimate medical prescription for an opioid analgesic, then continue taking it after the pain subsides. Others experiment briefly with nonmedical prescription opioids or use them intermittently without adverse effect. Some progress from prescription opioids to heroin, despite its dangers.5 Still others have a catastrophic outcome, such as an overdose or severe accident, the first time they use opioids.6 Rapid progression from misuse of opioids to dependence is most likely in vulnerable populations, such as those with concurrent mental illness, other substance use disorders, or increased sensitivity to pain.7
Understanding the terms. Before we continue, a word about terminology is in order. “Misuse” generally refers to the use of a medication in a manner (ie, purpose, dose, or frequency) other than its intended use, while “drug addiction” is the repeated use of a drug despite resulting harm. Here we will use “opioid dependence” to mean a pattern of increasing use characterized by significant impairment and distress and an inability to stop, and “opioid withdrawal” to reflect a constellation of symptoms, such as insomnia, nausea, diarrhea, and muscle aches, that can follow physiological dependence (though not necessarily opioid dependence). Our definitions of these terms are consistent with those of the American Psychiatric Association (APA).8 Worth noting, however, is the fact that as the APA prepares for the publication of the 5th edition of its Diagnostic and Statistical Manual of Mental Disorders, its Substance Disorder Work Group has proposed replacing the term “opioid dependence” with “opioid use disorder” to reduce the confusion associated with these definitions.9
Assessing illicit opioid use: Start with a targeted question
Most patients who are opioid dependent do not seek treatment for it,10 and are typically free of medical sequelae associated with drug addiction when they see family practitioners. The absence of self-reporting and obvious physical signs and symptoms, coupled with the increase in illicit use of prescription opioids, underscores the need for family physicians to identify patients who are abusing opioids and ensure that they get the help they need.
Screening tools. There are a number of screening tools you can use for this purpose—eg, CAGE-Adapted to Include Drugs (CAGE-AID) and Drug Abuse Screening Test (DAST)11,12—but they have not been found to be significantly better than a careful substance abuse history.13
Straightforward questions. You can start by asking, “Do you take any medications for pain?” If the answer is Yes, get the name of the drug and inquire about the frequency of use and the route, the amount typically taken, and the duration of the current use pattern. Ask specifically about opioids when taking a substance abuse history. After a question about alcohol use, you can say, “Do you use any other drugs in a serious way? Marijuana? Opioids like Percocet, Vicodin, or Oxycontin?” Although it can be very difficult to detect opioid dependence if the patient is not forthcoming, other likely indicators of drug-seeking behavior should trigger additional questions. (See “Opioid dependence: Red flags to keep in mind”.14-16)
“Brief” protocols. Recent studies of Screening, Brief Intervention, and Referral to Treatment (SBIRT) programs have found that the simple, time-limited interventions they offer (visit http://www.samhsa.gov/prevention/sbirt/SBIRTwhitepaper.pdf to learn more) lead to a reduction in self-reported illicit opioid use.17,18 Family physicians can readily incorporate SBIRT protocols into routine practice, as an evidence-based and often reimbursable approach to substance abuse.17
Suspect opioid dependence in a patient who:
- describes pain resulting from back or orthopedic injuries without corresponding documentation or imaging
- requests a specific opioid for pain management
- shows little interest in a physical exam, diagnostic testing, or nonpharmacological remedies
- talks about changes in work or relationship status
- ceases to participate in activities or hobbies that previously occupied a considerable amount of his or her time. This may signal social isolation or indicate that the patient is spending a great deal of time in pursuit of opioids.
Additional steps before initiating treatment
After screening and diagnostic evaluation provide evidence that a patient is opioid dependent, you can take several steps to guide him or her to the appropriate treatment.
A thorough biopsychosocial assessment covering co-occurring psychiatric illnesses, pain, psychosocial stressors contributing to opioid use, and infectious disease screening is required to gain a clear picture of the patient’s situation. In every case, acute emergencies such as suicidal ideation require immediate intervention, which may involve hospitalization.19
Assess the patient’s desire for help. After the initial assessment, it is often helpful to categorize the patient’s “stage of change” (precontemplation, contemplation, preparation, action, or maintenance),20 and to tailor your next step accordingly. A patient who denies that opioid use is a problem or is clearly ambivalent about seeking treatment may require a conversation that uses principles of motivational interviewing—a collaborative approach that aims to evoke and strengthen personal motivation for change.21 Consider a question that encourages him or her to express reasons for change, such as: “How would you like your current situation to be different?” As almost everyone abusing opioids has thoughts about stopping, such a question may help the patient focus on specific changes.
CASE When you question Sam about his interest in oxycodone, he breaks down. He’s been unable to find work or to lose the excess weight he gained during the many months he cared for his wife. He tells you that soon after his wife stopped taking the pain pills, he started taking them. At first, he took one occasionally. Then he started taking the opioids every day, and finally, whenever he awakened at night. Now, Sam says, he has no more pills, and he’s nauseous, depressed, and unable to sleep—and looking to you for help.
Sam fits the criteria for opioid withdrawal as a result of physiological dependence; further questioning reveals that he also suffers from opioid dependence, and that he is receptive to treatment.
Recommending treatment and following up
Several options are available for patients who, like Sam, have signs and symptoms of opioid withdrawal as a result of physiological dependence. You can provide a referral to a physician specializing in addiction, recommend detoxification and/or treatment in an inpatient facility, or initiate pharmacological treatment and provide a referral to a behavioral therapist. Whatever the initial approach, most patients will ultimately be treated as outpatients, with a combination of pharmacotherapy and behavioral therapy—often, with monitoring and oversight by a primary care physician. Which approach to pursue should be guided by evidence-based recommendations (TABLE)17,22-27 and jointly decided by physician and patient.
TABLE
Treating opioid dependence: Key clinical recommendations
| Recommendation | Evidence (SOR) | Comments |
|---|---|---|
| Screen all patients for substance use, including opioids. Brief interventions and referral to treatment when appropriate may reduce opioid use17,22 | Consistent findings from RCTs; evidence-based guideline (A) | SBIRT reduces self-reported opioid use; efforts to replicate such reports with objective evidence (eg, toxicology screens) are underway |
| Recommend maintenance medication (ie, buprenorphine, naltrexone, methadone) for all patients entering treatment for opioid dependence with physiological dependence; methadone is the safest for pregnant women23-25 | Consistent findings from RCTs; evidence-based guideline (A) | Methadone is the gold standard for pregnant women; further studies are needed to determine the safety of in utero exposure to buprenorphine and naltrexone |
| Keep patients on maintenance medication for ≥3 months; higher relapse rates are noted when medication is discontinued in <3 months23,24 | Consistent findings from RCTs (A) | Relapse rates are higher when maintenance medication is discontinued in <3 months |
| Caution patients with opioid dependence of the risk for accidental overdose and death with relapse and take action—eg, offering naloxone rescue kits to patients and families, as appropriate26 | Consistent findings from RCTs and prospective cohort studies; evidence-based guideline (A) | |
| Take steps to prevent diversion and accidental ingestion of agonist therapies, using tools such as frequent toxicology screens, random pill counts, and designated pharmacies, and monitoring adherence to psychosocial treatment26,27 | Practice guideline (consensus) (C) | |
| RCTs, randomized clinical trials; SBIRT, Screening, Brief Intervention, and Referral to Treatment; SOR, strength of recommendation. | ||
Medication plays a key role in recovery
Recommend medication-assisted treatment, either with an agonist (buprenorphine or methadone) or an antagonist (naltrexone), for every patient with physiological opioid dependence. The goals of pharmacotherapy are to prevent or reduce withdrawal symptoms and craving, avoid relapse, and restore to a normal state any physiological functions (eg, sleep, bowel movements) that have been disrupted by opioid use.28 When continued for ≥3 months, medication has been shown to improve outcomes.23,24,29 In one recent study, 49% of opioid-dependent participants who were still taking buprenorphine-naloxone at 12 weeks had successful outcomes (minimal or no opioid use), vs 7% of those undergoing a brief buprenorphine-naloxone taper.24
There are risks associated with medication-assisted therapy, however. The ones of greatest concern are a potential increase in drug-drug interactions, the risk of diversion (a concern with both buprenorphine and methadone), and the potential for accidental overdose.2,30
Buprenorphine, a partial mu-opioid receptor agonist, is a Schedule III controlled substance and can be dispensed by a pharmacy, making inpatient opioid detoxification unnecessary for many opioid-dependent patients. Physicians who wish to prescribe buprenorphine for the treatment of opioid dependence must complete an 8-hour course, offered by the American Medical Association and the APA, among other medical groups, and obtain a Drug Enforcement Administration code (“X”) license. 31
Buprenorphine has a high affinity for, and a slow dissociation from, mu-opioid receptors, resulting in the displacement of other opioids from the mu receptor and less severe withdrawal.32 As a partial agonist, buprenorphine attenuates opioid withdrawal symptoms with a ceiling, or near maximal, effect at 16 mg, thereby lowering the risk for overdose.33 A sublingual formulation that combines buprenorphine with naloxone, an opioid antagonist that exerts its full effect when injected but is minimally absorbed sublingually, reduces the potential for abuse of buprenorphine without interfering with its effectiveness.34
Compared with methadone, buprenorphine is less likely to interact with antiretroviral medications or to cause QTc prolongation, erectile dysfunction, or cognitive or psychomotor impairment.31,35-37 Limitations include the ceiling effect, which can be a problem for cases in which more agonist is needed; cost (approximately $12/d), and the lack of approval by the US Food and Drug Administration (FDA) for use during pregnancy.
Buprenorphine maintenance involves 3 phases: induction, stabilization, and maintenance.38 Induction takes place in a clinician’s office at the time the patient experiences opioid withdrawal symptoms, typically 6 to 48 hours after taking the last opioid. Extended treatment improves clinical outcomes,23,24 and longer-term maintenance (of indefinite duration) is frequently required.
Naltrexone is a mu-receptor antagonist, and therefore does not cause physical dependence or have agonist effects such as euphoria and sedation. As a result, it has no diversion value and may appeal to those who view opioid-agonist pharmacotherapy as simply trading one drug for another.39 Naltrexone is not a controlled substance and is not subject to the regulatory requirements that buprenorphine and methadone face.
Although agonists can be started in the first day or 2 after a patient decides to stop using opioids, patients must be opioid-free for ≥7 days before starting naltrexone. That’s because its antagonist properties will precipitate withdrawal if another opioid is present on the opioid receptors. During the 7-day “washout” period, you can treat opioid withdrawal symptoms with medications such as clonidine and dicyclomine, but such symptoms make patients especially vulnerable to relapse while waiting to start naltrexone.
Oral naltrexone’s effectiveness as a treatment for opioid dependence has been limited by poor adherence. But a long-acting intramuscular form of the drug, approved by the FDA in 2010 and requiring once-a-month injection, mitigates this concern.40,41
Methadone is a full mu-opioid agonist, administered daily at specialized clinics, as a maintenance therapy for opioid dependence. Although office-based physicians can prescribe methadone for pain, the drug can only be used for opioid dependence under the auspices of state- and federally regulated opioid treatment programs (http://findtreatment.samhsa.gov/TreatmentLocator/faces/quickSearch.jspx; a mobile phone application is also available at http://www.samhsa.gov/mobile/treatmentlocator.aspx).
Methadone, a Schedule III controlled substance with a half-life averaging 24 to 36 hours, requires daily dosing.42 Its slow metabolism and long half-life increase the risk for overdose.
Methadone is best for patients who are highly dependent on opioids and likely to benefit from a structured treatment environment with daily supervision (although patients who are doing well may earn take-home privileges so they don’t have to come to the clinic every day).43 New patients should receive an initial dose of 30 mg or less, and a maximum first-day dose of 40 mg.44
Methadone remains the standard of care for pregnant women being treated for opioid dependence, while studies of the effects of buprenorphine and naltrexone on a developing fetus continue. Although methadone’s efficacy, particularly in lower doses, is similar to that of buprenorphine,45 its adverse effect profile is worse. Adverse effects include drug-drug interactions, the potential for respiratory depression (especially when combined with alcohol or sedatives), QTc prolongation (which requires monitoring by electrocardiogram), sedation, and weight gain, and should be considered before selecting methadone as a maintenance pharmacotherapy.30,37,46 And, because relapse rates within 12 months of tapering off methadone have been reported to exceed 80%,47 both the clinician and the patient need to consider the likelihood of long-term, even lifelong, maintenance before initiating treatment.
Behavioral interventions are a vital part of the picture
Studies evaluating the extent to which various types and amounts of counseling improve outcomes compared with pharmacotherapy alone have had conflicting results.24,48 Nonetheless, most clinicians consider counseling to be a critical component of treatment for opioid dependence and recommend, at a minimum, either individual or group counseling (various modalities have been shown to be effective) and regular attendance at a self-help group like Narcotics Anonymous. Contingency management, a type of therapy that uses prizes as incentives for desired behaviors; and family therapy, individual counseling, and community-based programs have all been found to improve outcomes.6,49
CASE You refer Sam to an addiction psychiatrist, who stabilizes him on 16 mg buprenorphine/naloxone daily as part of an outpatient treatment program. Sam is enrolled in a weekly buprenorphine stabilization group, where he gives a urine sample each week. He also begins seeing a social worker weekly for counseling and attends Narcotics Anonymous meetings 2 to 3 times a week. At a follow-up appointment with you 6 months later, he reports that he has been abstinent from oxycodone for 6 months, his sleep is improved, and he feels better about his chances of finding another job.
Your role in safeguarding the patient
With the rising prevalence of opioid overdose, patient education aimed at crisis prevention is crucial, as well. Warn patients of the risk of accidental overdose, often associated with relapse, stressing the importance of continuing treatment and taking their maintenance medication exactly as prescribed.
There are other steps you can take to safeguard patients—eg, providing naloxone rescue kits to patients and their families when appropriate. You can also institute diversion and overdose prevention measures for patients taking buprenorphine or methadone—providing a lock box for take-home medication, implementing treatment contracts, and using a designated pharmacy to dispense buprenorphine, for example.26,27,50
Regular monitoring, urine drug screens (see TABLE W1), and random pill counts, in which patients are typically given 24 hours to bring in their prescribed medication so it can be counted, can also help keep patients on track. Treatment for concurrent psychiatric disorders—depression, anxiety, and personality disorders are common among patients with opioid dependence—is likely to improve the outcome of treatment, as well.
TABLE W1
Pharmacokinetics of common opioids: Time detectable in urine*
| Drug (half-life) | Time detectable in urine | Comment |
|---|---|---|
| Codeine (2.5-3 h) | 48 h | Pharmacogenetic-dependent effects may affect detection |
| Fentanyl Transdermal (17 h) Submucosal (7 h) | Not usually detected in urine (lack of metabolites) | Excretion of transdermal fentanyl can last days |
| Hydromorphone IR (2.3 h) ER (18.6 h) | 2-4 d | Significant interpatient variability |
| Methadone (8-59 h) | 3 d | |
| Morphine (1.5-2 h) | 48-72 h | 90% eliminated within 24 h |
| Oxycodone IR (3.2 h) ER (4.5 h) | Often not detected in urine | High-fat meals may increase serum concentrations of ER formulation |
| Propoxyphene Parent drug (6-12 h) Metabolite (30-36 h) | 6-48 h | |
| ER, extended release; IR, immediate release. *Previously appeared in: McBane S, Weige N. Is it time to drug test your chronic pain patient? J Fam Pract. 2010;59:628-633. Sources: Clinical Pharmacology [online]. Tampa, FL: Gold Standard Inc; 2010. Available at: http://cp.gsm.com. Accessed March 5, 2010; Drug Facts and Comparisons [online]. 2010. Available at: http://www.factsandcomparisons.com/. Accessed March 5, 2010. | ||
CORRESPONDENCE Kevin P. Hill, MD, MHS, McLean Hospital, 115 Mill Street, Belmont, MA 02478; [email protected]
1. Centers for Disease Control and Prevention (CDC). Emergency department visits involving nonmedical use of selected prescription drugs - United States, 2004-2008. MMWR Morb Mortal Wkly Rep. 2010;59:705-709.
2. Bohnert AS, Valenstein M, Bair MJ, et al. Association between opioid prescribing patterns and opioid overdose-related deaths. JAMA. 2011;305:1315-1321.
3. Warner M, Chen LH, Makuc DM. Increase in fatal poisonings involving opioid analgesics in the United States, 1999-2006. NCHS Data Brief. 2009;(22):1-8.4.
4. Substance Abuse and Mental Health Services Administration. Results From the 2009 National Survey on Drug Use and Health: Volume I. Summary of National Findings. Rockville, Md: SAMHSA, Office of Applied Studies; 2010. NSDUH Series H-38A, HHS publication SMA 10-4856. Available at: http://www.samhsa.gov/data/NSDUH/2k9NSDUH/2k9Results.htm. Accessed August 22, 2012.
5. Hser YI, Huang D, Brecht ML, et al. Contrasting trajectories of heroin, cocaine, and methamphetamine use. J Addict Dis. 2008;27:13-21.
6. Veilleux JC, Colvin PJ, Anderson J, et al. A review of opioid dependence treatment: pharmacological and psychosocial interventions to treat opioid addiction. Clin Psychol Rev. 2011;30:155-166.
7. George O, Koob GF. Individual differences in prefrontal cortex function and the transition from drug use to drug dependence. Neurosci Biobehav Rev. 2011;35:232-247.
8. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 4th ed, text rev (DSM-IV-TR). Arlington, Va: American Psychiatric Association; 2000.
9. American Psychiatric Association. R 19 opioid use disorder. http://www.dsm5.org/ProposedRevisions/Pages/proposedrevision.aspx?rid=460. Updated April 30, 2012. Accessed June 20, 2012.
10. Substance Abuse and Mental Health Services Administration. Results From the 2008 National Survey on Drug Use and Health: National Findings. Rockville, Md: SAMHSA, Office of Applied Studies; 2009. NSDUH Series H-36, HHS publication SMA 09-4434. Available at: http://www.samhsa.gov/data/nsduh/2k8nsduh/2k8Results.htm. Accessed August 22, 2012.
11. Brown RL, Rounds LA. Conjoint screening questionnaires for alcohol and other drug abuse: criterion validity in a primary care practice. Wis Med J. 1995;94:135-140.
12. Skinner HA. The drug abuse screening test. Addict Behav. 1982;7:363-371.
13. US Preventive Services Task Force. Screening for Illicit Drug Use: U.S. Preventive Services Task Force Recommendation Statement. January 2008. Available at: http://www.uspreventiveservicestaskforce.org/uspstf08/druguse/drugrs.htm. Accessed May 7, 2012.
14. Gourlay D, Caplan Y, Heit H. Urine Drug Testing in Clinical Practice: Dispelling the Myths and Designing Strategies. San Francisco, Calif: California Academy of Family Physicians; 2006.
15. Jackman R, Purvis J, Mallett B. Chronic nonmalignant pain in primary care. Am Fam Physician. 2008;78:1155-1162.
16. McBane S, Weigle N. Is it time to drug test your chronic pain patient? J Fam Pract. 2010;59:628-633.
17. Madras BK, Compton WM, Avula D, et al. Screening, brief interventions, referral to treatment (SBIRT) for illicit drug and alcohol use at multiple healthcare sites: comparison at intake and 6 months later. Drug Alcohol Depend. 2009;99:280-295.
18. The InSight Project Research Group. SBIRT outcomes in Houston: final report on InSight, a hospital district-based program for patients at risk for alcohol or drug use problems. Alcohol Clin Exp Res. 2009;33:1374-1381.
19. Borges G, Walters EE, Kessler RC. Associations of substance use, abuse, and dependence with subsequent suicidal behavior. Am J Epidemiol. 2000;151:781-789.
20. Prochaska JO, DiClemente CC. Stages and processes of self-change of smoking: toward an integrative model of change. J Consult Clin Psychol. 1983;51:390-395.
21. Smedslund G, Berg RC, Hammerstrom KT, et al. Motivational interviewing for substance abuse. Cochrane Database Syst Rev. 2011;(5):CD008063.-
22. Gryczynski J, Mitchell SG, Peterson TR, et al. The relationship between services delivered and substance use outcomes in New Mexico’s Screening, Brief Intervention, Referral and Treatment (SBIRT) Initiative. Drug Alcohol Depend. 2011;118:152-157.
23. Woody GE, Poole SA, Subramaniam G, et al. Extended vs short-term buprenorphine-naloxone for treatment of opioid-addicted youth: a randomized trial. JAMA. 2008;300:2003-2011.
24. Weiss RD, Potter JS, Fiellin DA, et al. Adjunctive counseling during brief and extended buprenorphine-naloxone treatment for prescription opioid dependence: a 2-phase randomized controlled trial. Arch Gen Psychiatry. 2011;68:1238-1246.
25. Johansson BA, Berglund M, Lindgren A. Efficacy of maintenance treatment with naltrexone for opioid dependence: a meta-analytical review. Addiction. 2006;101:491-503.
26. Hall AJ, Logan JE, Toblin RL, et al. Patterns of abuse among unintentional pharmaceutical overdose fatalities. JAMA. 2008;300:2613-2620.
27. Zacny J, Bigelow G, Compton P, et al. College on Problems of Drug Dependence taskforce on prescription opioid non-medical use and abuse: position statement. Drug Alcohol Depend. 2003;69:215-232.
28. Kreek MJ. Rationale for maintenance pharmacotherapy of opiate dependence. Res Publ Assoc Res Nerv Ment Dis. 1992;70:205-230.
29. Mattick RP, Breen C, Kimber J, et al. Methadone maintenance therapy versus no opioid replacement therapy for opioid dependence. Cochrane Database Syst Rev. 2009;(3):CD002209.-
30. McCance-Katz EF, Sullivan LE, Nallani S. Drug interactions of clinical importance among the opioids, methadone and buprenorphine, and other frequently prescribed medications: a review. Am J Addict. 2010;19:4-16.
31. Office of National Drug Control Policy Reauthorization Act of 2006 (ONDCPRA), HR 6344, 109th Cong, 2nd Sess (2006).
32. Lewis JW, Walter D. Buprenorphine—background to its development as a treatment for opiate dependence. In: Blaine JD, ed. Buprenorphine: An Alternative Treatment for Opioid Dependence. Rockville, Md: National Institute on Drug Abuse; 1992:5-11. NIDA Research Monograph, No. 121. Available at: http://archives.drugabuse.gov/pdf/monographs/121.pdf. Accessed August 22, 2012.
33. Walsh SL, Preston KL, Stitzer ML, et al. Clinical pharmacology of buprenorphine: ceiling effects at high doses. Clin Pharmacol Ther. 1994;55:569-580.
34. Alho H, Sinclair D, Vuori E, et al. Abuse liability of buprenorphine-naloxone tablets in untreated IV drug users. Drug Alcohol Depend. 2007;88:75-78.
35. Hallinan R, Byrne A, Agho K, et al. Erectile dysfunction in men receiving methadone and buprenorphine maintenance treatment. J Sex Med. 2008;5:684-692.
36. Rapeli P, Fabritius C, Alho H, et al. Methadone vs. buprenorphine/naloxone during early opioid substitution treatment: a naturalistic comparison of cognitive performance relative to healthy controls. BMC Clin Pharmacol. 2007;7:5.-
37. Wedam EF, Bigelow GE, Johnson RE, et al. QT-interval effects of methadone, levomethadyl, and buprenorphine in a randomized trial. Arch Intern Med. 2007;167:2469-2475.
38. Center for Substance Abuse Treatment. Clinical Guidelines for the Use of Buprenorphine in the Treatment of Opioid Addiction. Rockville, Md: Substance Abuse and Mental Health Services Administration; 2004. Treatment Improvement Protocol (TIP) Series 40. DHHS publication SMA 04-3939.
39. Kleber HD. Methadone maintenance 4 decades later: thousands of lives saved but still controversial. JAMA. 2008;300:2303-2305.
40. Hulse GK, Morris N, Arnold-Reed D, et al. Improving clinical outcomes in treating heroin dependence: randomized, controlled trial of oral or implant naltrexone. Arch Gen Psychiatry. 2009;66:1108-1115.
41. US Food and Drug Administration. FDA approves injectable drug to treat opioid-dependent patients. October 12, 2010. Available at: http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/2010/ucm229109.htm. Accessed September 11, 2012.
42. Inturrisi CE, Verebely K. The levels of methadone in the plasma in methadone maintenance. Clin Pharmacol Ther. 1972;13 (5 pt 1):633-637.
43. Stitzer M, Bigelow G, Lawrence C, et al. Medication take-home as a reinforcer in a methadone maintenance program. Addict Behav. 1977;2:9-14.
44. Code of Federal Regulations. Title 42.8.12. Federal Opioid Treatment Standards. October 2010.
45. Johnson RE, Chutuape MA, Strain EC, et al. A comparison of levomethadyl acetate, buprenorphine, and methadone for opioid dependence. N Engl J Med. 2000;343:1290-1297.
46. Krantz MJ, Martin J, Stimmel B, et al. QTc interval screening in methadone treatment. Ann Intern Med. 2009;150:387-395.
47. Ball JC, Lange WR, Myers CP, et al. Reducing the risk of AIDS through methadone maintenance treatment. J Health Soc Behav. 1988;29:214-226.
48. Fiellin DA, Pantalon MV, Chawarski MC, et al. Counseling plus buprenorphine-naloxone maintenance therapy for opioid dependence. N Engl J Med. 2006;355:365-374.
49. Defulio A, Everly JJ, Leoutsakos JM, et al. Employment-based reinforcement of adherence to an FDA approved extended release formulation of naltrexone in opioid-dependent adults: a randomized controlled trial. Drug Alcohol Depend. 2012;120:48-54.
50. Savage SR. Management of opioid medications in patients with chronic pain and risk of substance misuse. Curr Psychiatry Rep. 2009;11:377-384.
1. Centers for Disease Control and Prevention (CDC). Emergency department visits involving nonmedical use of selected prescription drugs - United States, 2004-2008. MMWR Morb Mortal Wkly Rep. 2010;59:705-709.
2. Bohnert AS, Valenstein M, Bair MJ, et al. Association between opioid prescribing patterns and opioid overdose-related deaths. JAMA. 2011;305:1315-1321.
3. Warner M, Chen LH, Makuc DM. Increase in fatal poisonings involving opioid analgesics in the United States, 1999-2006. NCHS Data Brief. 2009;(22):1-8.4.
4. Substance Abuse and Mental Health Services Administration. Results From the 2009 National Survey on Drug Use and Health: Volume I. Summary of National Findings. Rockville, Md: SAMHSA, Office of Applied Studies; 2010. NSDUH Series H-38A, HHS publication SMA 10-4856. Available at: http://www.samhsa.gov/data/NSDUH/2k9NSDUH/2k9Results.htm. Accessed August 22, 2012.
5. Hser YI, Huang D, Brecht ML, et al. Contrasting trajectories of heroin, cocaine, and methamphetamine use. J Addict Dis. 2008;27:13-21.
6. Veilleux JC, Colvin PJ, Anderson J, et al. A review of opioid dependence treatment: pharmacological and psychosocial interventions to treat opioid addiction. Clin Psychol Rev. 2011;30:155-166.
7. George O, Koob GF. Individual differences in prefrontal cortex function and the transition from drug use to drug dependence. Neurosci Biobehav Rev. 2011;35:232-247.
8. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 4th ed, text rev (DSM-IV-TR). Arlington, Va: American Psychiatric Association; 2000.
9. American Psychiatric Association. R 19 opioid use disorder. http://www.dsm5.org/ProposedRevisions/Pages/proposedrevision.aspx?rid=460. Updated April 30, 2012. Accessed June 20, 2012.
10. Substance Abuse and Mental Health Services Administration. Results From the 2008 National Survey on Drug Use and Health: National Findings. Rockville, Md: SAMHSA, Office of Applied Studies; 2009. NSDUH Series H-36, HHS publication SMA 09-4434. Available at: http://www.samhsa.gov/data/nsduh/2k8nsduh/2k8Results.htm. Accessed August 22, 2012.
11. Brown RL, Rounds LA. Conjoint screening questionnaires for alcohol and other drug abuse: criterion validity in a primary care practice. Wis Med J. 1995;94:135-140.
12. Skinner HA. The drug abuse screening test. Addict Behav. 1982;7:363-371.
13. US Preventive Services Task Force. Screening for Illicit Drug Use: U.S. Preventive Services Task Force Recommendation Statement. January 2008. Available at: http://www.uspreventiveservicestaskforce.org/uspstf08/druguse/drugrs.htm. Accessed May 7, 2012.
14. Gourlay D, Caplan Y, Heit H. Urine Drug Testing in Clinical Practice: Dispelling the Myths and Designing Strategies. San Francisco, Calif: California Academy of Family Physicians; 2006.
15. Jackman R, Purvis J, Mallett B. Chronic nonmalignant pain in primary care. Am Fam Physician. 2008;78:1155-1162.
16. McBane S, Weigle N. Is it time to drug test your chronic pain patient? J Fam Pract. 2010;59:628-633.
17. Madras BK, Compton WM, Avula D, et al. Screening, brief interventions, referral to treatment (SBIRT) for illicit drug and alcohol use at multiple healthcare sites: comparison at intake and 6 months later. Drug Alcohol Depend. 2009;99:280-295.
18. The InSight Project Research Group. SBIRT outcomes in Houston: final report on InSight, a hospital district-based program for patients at risk for alcohol or drug use problems. Alcohol Clin Exp Res. 2009;33:1374-1381.
19. Borges G, Walters EE, Kessler RC. Associations of substance use, abuse, and dependence with subsequent suicidal behavior. Am J Epidemiol. 2000;151:781-789.
20. Prochaska JO, DiClemente CC. Stages and processes of self-change of smoking: toward an integrative model of change. J Consult Clin Psychol. 1983;51:390-395.
21. Smedslund G, Berg RC, Hammerstrom KT, et al. Motivational interviewing for substance abuse. Cochrane Database Syst Rev. 2011;(5):CD008063.-
22. Gryczynski J, Mitchell SG, Peterson TR, et al. The relationship between services delivered and substance use outcomes in New Mexico’s Screening, Brief Intervention, Referral and Treatment (SBIRT) Initiative. Drug Alcohol Depend. 2011;118:152-157.
23. Woody GE, Poole SA, Subramaniam G, et al. Extended vs short-term buprenorphine-naloxone for treatment of opioid-addicted youth: a randomized trial. JAMA. 2008;300:2003-2011.
24. Weiss RD, Potter JS, Fiellin DA, et al. Adjunctive counseling during brief and extended buprenorphine-naloxone treatment for prescription opioid dependence: a 2-phase randomized controlled trial. Arch Gen Psychiatry. 2011;68:1238-1246.
25. Johansson BA, Berglund M, Lindgren A. Efficacy of maintenance treatment with naltrexone for opioid dependence: a meta-analytical review. Addiction. 2006;101:491-503.
26. Hall AJ, Logan JE, Toblin RL, et al. Patterns of abuse among unintentional pharmaceutical overdose fatalities. JAMA. 2008;300:2613-2620.
27. Zacny J, Bigelow G, Compton P, et al. College on Problems of Drug Dependence taskforce on prescription opioid non-medical use and abuse: position statement. Drug Alcohol Depend. 2003;69:215-232.
28. Kreek MJ. Rationale for maintenance pharmacotherapy of opiate dependence. Res Publ Assoc Res Nerv Ment Dis. 1992;70:205-230.
29. Mattick RP, Breen C, Kimber J, et al. Methadone maintenance therapy versus no opioid replacement therapy for opioid dependence. Cochrane Database Syst Rev. 2009;(3):CD002209.-
30. McCance-Katz EF, Sullivan LE, Nallani S. Drug interactions of clinical importance among the opioids, methadone and buprenorphine, and other frequently prescribed medications: a review. Am J Addict. 2010;19:4-16.
31. Office of National Drug Control Policy Reauthorization Act of 2006 (ONDCPRA), HR 6344, 109th Cong, 2nd Sess (2006).
32. Lewis JW, Walter D. Buprenorphine—background to its development as a treatment for opiate dependence. In: Blaine JD, ed. Buprenorphine: An Alternative Treatment for Opioid Dependence. Rockville, Md: National Institute on Drug Abuse; 1992:5-11. NIDA Research Monograph, No. 121. Available at: http://archives.drugabuse.gov/pdf/monographs/121.pdf. Accessed August 22, 2012.
33. Walsh SL, Preston KL, Stitzer ML, et al. Clinical pharmacology of buprenorphine: ceiling effects at high doses. Clin Pharmacol Ther. 1994;55:569-580.
34. Alho H, Sinclair D, Vuori E, et al. Abuse liability of buprenorphine-naloxone tablets in untreated IV drug users. Drug Alcohol Depend. 2007;88:75-78.
35. Hallinan R, Byrne A, Agho K, et al. Erectile dysfunction in men receiving methadone and buprenorphine maintenance treatment. J Sex Med. 2008;5:684-692.
36. Rapeli P, Fabritius C, Alho H, et al. Methadone vs. buprenorphine/naloxone during early opioid substitution treatment: a naturalistic comparison of cognitive performance relative to healthy controls. BMC Clin Pharmacol. 2007;7:5.-
37. Wedam EF, Bigelow GE, Johnson RE, et al. QT-interval effects of methadone, levomethadyl, and buprenorphine in a randomized trial. Arch Intern Med. 2007;167:2469-2475.
38. Center for Substance Abuse Treatment. Clinical Guidelines for the Use of Buprenorphine in the Treatment of Opioid Addiction. Rockville, Md: Substance Abuse and Mental Health Services Administration; 2004. Treatment Improvement Protocol (TIP) Series 40. DHHS publication SMA 04-3939.
39. Kleber HD. Methadone maintenance 4 decades later: thousands of lives saved but still controversial. JAMA. 2008;300:2303-2305.
40. Hulse GK, Morris N, Arnold-Reed D, et al. Improving clinical outcomes in treating heroin dependence: randomized, controlled trial of oral or implant naltrexone. Arch Gen Psychiatry. 2009;66:1108-1115.
41. US Food and Drug Administration. FDA approves injectable drug to treat opioid-dependent patients. October 12, 2010. Available at: http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/2010/ucm229109.htm. Accessed September 11, 2012.
42. Inturrisi CE, Verebely K. The levels of methadone in the plasma in methadone maintenance. Clin Pharmacol Ther. 1972;13 (5 pt 1):633-637.
43. Stitzer M, Bigelow G, Lawrence C, et al. Medication take-home as a reinforcer in a methadone maintenance program. Addict Behav. 1977;2:9-14.
44. Code of Federal Regulations. Title 42.8.12. Federal Opioid Treatment Standards. October 2010.
45. Johnson RE, Chutuape MA, Strain EC, et al. A comparison of levomethadyl acetate, buprenorphine, and methadone for opioid dependence. N Engl J Med. 2000;343:1290-1297.
46. Krantz MJ, Martin J, Stimmel B, et al. QTc interval screening in methadone treatment. Ann Intern Med. 2009;150:387-395.
47. Ball JC, Lange WR, Myers CP, et al. Reducing the risk of AIDS through methadone maintenance treatment. J Health Soc Behav. 1988;29:214-226.
48. Fiellin DA, Pantalon MV, Chawarski MC, et al. Counseling plus buprenorphine-naloxone maintenance therapy for opioid dependence. N Engl J Med. 2006;355:365-374.
49. Defulio A, Everly JJ, Leoutsakos JM, et al. Employment-based reinforcement of adherence to an FDA approved extended release formulation of naltrexone in opioid-dependent adults: a randomized controlled trial. Drug Alcohol Depend. 2012;120:48-54.
50. Savage SR. Management of opioid medications in patients with chronic pain and risk of substance misuse. Curr Psychiatry Rep. 2009;11:377-384.