User login
Case Q: How can I best remove my patient’s difficult-to-find implant?
Individuals spend close to half of their lives preventing, or planning for, pregnancy. As such, contraception plays a major role in patient-provider interactions. Contraception counseling and management is a common scenario encountered in the general gynecologist’s practice. Luckily, we have 2 evidence-based guidelines developed by the US Centers for Disease Control and Prevention (CDC) that support the provision of contraceptive care:
- US Medical Eligibility for Contraceptive Use (US-MEC),1 which provides guidance on which patients can safely use a method
- US Selected Practice Recommendations for Contraceptive Use (US-SPR),2 which provides method-specific guidance on how to use a method (including how to: initiate or start a method; manage adherence issues, such as a missed pill, etc; and manage common issues like breakthrough bleeding).
Both of these guidelines are updated routinely and are publicly available online or for free, through smartphone applications.
While most contraceptive care is straightforward, there are circumstances that require additional consideration. In the concluding part of this series on contraceptive conundrums, we review 2 clinical cases, existing evidence to guide management decisions, and our recommendations.
CASE 1 Patient presents with hard-to-remove implant
A 44-year-old patient (G2P2) with a new diagnosis of estrogen and progesterone-receptor–positive breast cancer is undergoing her evaluation with her oncologist who recommends removal of her contraceptive implant, which has been in place for 2 years. She presents to your office for removal; however, the device is no longer palpable.
What are your next steps?
Conundrum 1. Should you attempt to remove it?
No, never attempt implant removal if you cannot palpate or localize it. Localization of the implant needs to occur prior to any attempt. However, we recommend checking the contra-lateral arm before sending the patient to obtain imaging, especially if you have no formal documentation regarding in which arm the implant was placed. The next step is identifying what type of implant the patient likely has so you can correctly interpret imaging studies.
Conundrum 2. What type of subdermal contraceptive device is it likely to be?
Currently, the only subdermal contraceptive device available for placement in the United States is the 68-mg etonogestrel implant, marketed with the brand name Nexplanon. This device was initially approved by the US Food and Drug Administration in 2001 and measures 4 cm in length by 2 mm in diameter. It is placed in the medial upper arm, about 8 cm proximal to the medial epicondyle and 3 cm posterior to the sulcus between the biceps and triceps muscles. (The implant should no longer be placed over the bicipital groove.) The implant is impregnated with 15 mg of barium sulfate, making it radiopaque and able to be seen on imaging modalities such as ultrasonography (10–18 mHz high frequency transducer) and x-ray (arm anteroposterior and lateral) for localization in cases in which the device becomes nonpalpable.3
Clinicians also may encounter devices which are no longer marketed in the United States, or which are only available in other countries, and thus should be aware of the appearance and imaging characteristics. It is important to let your imaging team know these characteristics as well:
- From 2006–2010, a 68-mg etonogestrel implant marketed under the name Implanon was available in the United States.4 It has the same dimensions and general placement recommendations as the Nexplanon etonogestrel device but is not able to be seen via imaging.
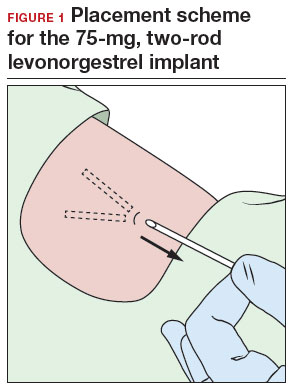
- A 2-arm, 75-mg levonorgestrel (LNG) device known as Jadelle (or, Norplant II; FIGURE 1) received FDA approval in 1996 and is currently only available overseas.5 It is also placed in the upper, inner arm in a V-shape using a single incision, and has dimensions similar to the etonogestrel implants.
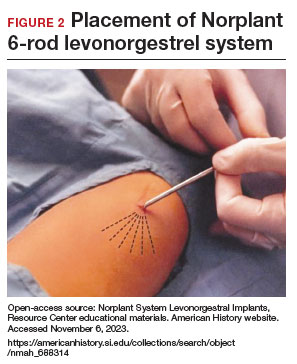
- From 1990– 2002, the 6-rod device known as Norplant was available in the United States. Each rod measured 3.4 cm in length and contained 36 mg of LNG (FIGURE 2).
Continue to: How do you approach removal of a deep contraceptive implant?...
How do you approach removal of a deep contraceptive implant?
Clinicians who are not trained in deep or difficult implant removal should refer patients to a trained provider (eg, a complex family planning subspecialist), or if not available, partner with a health care practitioner that has expertise in the anatomy of the upper arm (eg, vascular surgery, orthopedics, or interventional radiology). A resource for finding a nearby trained provider is the Organon Information Center (1-877-467-5266). However, when these services are not readily available, consider the following 3-step approach to complex implant removal.
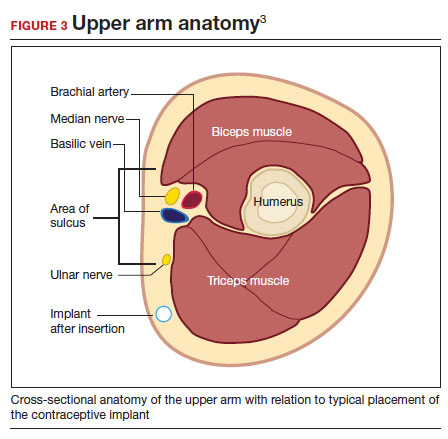
- Be familiar with the anatomy of the upper arm (FIGURE 3). Nonpalpable implants may be close to or under the biceps or triceps fascia or be near critically important and fragile structures like the neurovascular bundle of the upper arm. Prior to attempting a difficult implant removal, ensure that you are well acquainted with critical structures in the upper arm.
- Locate the device. Prior to attempting removal, localize the device using either x-ray or ultrasonography, depending on local availability. Ultrasound offers the advantage of mapping the location in 3 dimensions, with the ability to map the device with skin markings immediately prior to removal. Typically, a highfrequency transducer (15- or 18-MHz) is used, such as for breast imaging, either in a clinician’s office or in coordination with radiology. If device removal is attempted the same day, the proximal, midportion, and distal aspects of the device should be marked with a skin pen, and it should be noted what position the arm is in when the device is marked (eg, arm flexed at elbow and externally rotated so that the wrist is parallel to the ear).
Rarely, if a device is not seen in the expected extremity, imaging of the contralateral arm or a chest x-ray can be undertaken to rule out mis-documented laterality or a migrated device. Lastly, if no device is seen, and the patient has no memory of device removal, you can obtain the patient’s etonogestrel levels. (Resource: Merck National Service Center, 1-877-888-4231.)
Removal procedure. For nonpalpable implants, strong consideration should be given to performing the procedure with ultrasonography guidance. Rarely, fluoroscopic guidance may be useful for orientation in challenging cases, which may require coordination with other services, such as interventional radiology.
Cleaning and anesthetizing the site is similar to routine removal of a palpable implant. A 2- to 3-mm skin incision is made, either at the distal end of the implant (if one end is amenable to traditional pop-out technique) or over the midportion of the device (if a clinician has experience using the “U” technique).6 The incision should be parallel to the long axis of the implant and not perpendicular, to facilitate extension of the incision if needed during the procedure. Straight or curved hemostat clamps can then be used for blunt dissection of the subcutaneous tissues and to grasp the end of the device. Experienced clinicians may have access to a modified vasectomy clamp (with a
Indications for referral. Typically, referral to a complex family planning specialist or vascular surgeon is required for cases that involve dissection of the muscular fascia or where dissection would be in close proximity to critical neurologic or vascular structures.
CASE 1 Conclusion
Ultrasonography of the patient’s extremity demonstrated a
CASE 2 Patient enquires about immediate IUD insertion
A 28-year-old patient (G1P0) arrives at your clinic for a contraceptive consultation. They report a condom break during intercourse 4 days ago. Prior to that they used condoms consistently with each act of intercourse. They have used combined hormonal contraceptive pills in the past but had difficulty remembering to take them consistently. The patient and their partner have been mutually monogamous for 6 months and have no plans for pregnancy. Last menstrual period was 12 days ago. Their cycles are regular but heavy and painful. They are interested in using a hormonal IUD for contraception and would love to get it today.
- Do not attempt removal of a nonpalpable implant without prior localization via imaging
- Ultrasound-guided removal procedures using a “U” technique are successful for many deep implant removals but require specialized equipment and training
- Referral to a complex family planning specialist or other specialist is highly recommended for implants located below the triceps fascia or close to the nerves and vessels of the upper arm
- Never attempt to remove a nonpalpable implant prior to determining its location via imaging
Continue to: Is same-day IUD an option?...
Is same-day IUD an option?
Yes. This patient needs EC given the recent condom break, but they are still eligible for having an IUD placed today if their pregnancy test is negative and after counseling of the potential risks and benefits. According to the US-SPR it is reasonable to insert an IUD at any time during the cycle as long as you are reasonably certain the patient is not pregnant.7
Options for EC are:
- 1.5-mg oral LNG pill
- 30-mg oral UPA pill
- copper IUD (cu-IUD).
If they are interested in the cu-IUD for long-term contraception, by having a cu-IUD placed they can get both their needs met—EC and an ongoing method of contraception. Any patient receiving EC, whether a pill or an IUD, should be counseled to repeat a home urine pregnancy test in 2 to 4 weeks.
Given the favorable non–contraceptive benefits associated with 52-mg LNG-IUDs, many clinicians and patients have advocated for additional evidence regarding the use of hormonal IUDs alone for EC.
What is the evidence concerning LNG-IUD placement as EC?
The 52-mg LNG-IUD has not been mechanistically proven to work as an EC, but growing evidence exists showing that it is safe for same-day or “quick start” placement even in a population seeking EC—if their pregnancy test result is negative at the time of presentation.
Turok and colleagues performed a noninferiority trial comparing 1-month pregnancy rates after placement of either an LNG-IUD or a cu-IUD for EC.8 This study concluded that the LNG-IUD (which resulted in 1 pregnancy in 317 users; pregnancy rate, 0.3%; 95% confidence interval [CI], 0.01–1.70) is noninferior to cu-IUD (0 pregnancies in 321 users; pregnancy rate, 0%; 95% CI, 0.0–1.1) for EC. Although encouraging, only a small percentage of the study population seeking EC who received an IUD were actually at high risk of pregnancy (eg, they were not mid-cycle or were recently using contraception), which is why it is difficult to determine if the LNG-IUD actually works mechanistically as an EC. More likely, the LNG-IUD helps prevent pregnancy due to its ongoing contraceptive effect.9 Ongoing acts of intercourse post–oral EC initiation without starting a method of contraception is one of the main reasons for EC failure, which is why starting a method immediately is so effective at preventing pregnancy.10
A systematic review conducted by Ramanadhan and colleagues concluded that Turok’s 2021 trial is the only relevant study specific to 52-mg LNG-IUD use as EC, but they also mention that its results are limited in the strength of its conclusions due to biases in randomization, including11:
- the study groups were not balanced in that there was a 10% difference in reported use of contraception at last intercourse, which means that the LNG-IUD group had a lower baseline risk of pregnancy
- and a rare primary outcome (ie, pregnancy, which requires a larger sample size to know if the method works as an EC).
The review authors concluded that more studies are needed to further validate the effectiveness of using the 52-mg LNG-IUD as EC. Thus, for those at highest risk of pregnancy from recent unprotected sex and desiring a 52-mg IUD, it is probably best to continue combining oral EC with a 52-mg LNG-IUD and utilizing the LNG-IUD only as EC on a limited, case-by-case basis.
What we recommend
For anyone with a negative pregnancy test on the day of presentation, the studies mentioned further support the practice of same-day placement of a 52-mg LNG-IUD. However, those seeking EC who are at highest risk for an unplanned pregnancy (ie, the unprotected sex was mid-cycle), we recommend co-administering the LNG-IUD with oral LNG for EC.
CASE 2 Conclusion
After a conversation with the patient about all contraceptive options, through shared decision making the patient decided to take 1.5 mg of oral LNG and have a 52-mg LNG-IUD placed in the office today. They do not wish to be pregnant at this time and would choose termination if they became pregnant. They understood their pregnancy risk and opted to plan a urine pregnancy test at home in 2 weeks with a clear understanding that they should return to clinic immediately if the test is positive. ●
- A copper IUD is the most effective method of emergency contraception (EC).
- 52-mg LNG-IUDs are an emerging consideration for EC, but evidence is still lacking that they work as EC (or whether they just prevent pregnancy after placement for subsequent acts of intercourse). Clinicians should utilize shared decision making and advise patients to repeat a pregnancy test at home in 2 to 4 weeks
- Any patient receiving EC, whether a pill or an IUD, should be counseled to repeat a home urine pregnancy test in 2 to 4 weeks
- Any type of IUD can be placed same day if the clinician is reasonably sure the patient is not pregnant
- It appears safe to co-administer the 52-mg LNG-IUD with oral EC for those seeking emergency contraception but also want to use an LNG-IUD for contraception going forward
- Curtis KM, Jatlaoui TC, Tepper NK, et al. U.S. Selected Practice Recommendations for Contraceptive Use, 2016. Morb Mortal Wkly Rep. 2016;65:1-66. https://doi .org/10.15585/mmwr .rr6504a1
- Centers for Disease Control and Prevention. National Center for Chronic Disease Prevention and Health Promotion, Division of Reproductive Health. US Selected Practice Recommendations for Contraceptive Use (US-SPR). Accessed October 11, 2023. https://www.cdc.gov/reproductivehealth /contraception/mmwr/spr/summary.html
- Nexplanon [package insert]. Whitehouse Station, NJ: Merck; 2018.
- US Food and Drug Administration. Implanon (etonogestrel implant) 2006. Accessed November 6, 2023. https://www .accessdata.fda.gov/drugsatfda_docs/nda/2006 /021529s000_Lbl.pdf
- US Food and Drug Administration. Jadelle (levonorgestrel implant) 2016. Accessed November 6, 2023. https://www. accessdata.fda.gov/drugsatfda_docs/label/2016/020544s 010lbl.pdf
- Chen MJ, Creinin MD. Removal of a nonpalpable etonogestrel implant with preprocedure ultrasonography and modified vasectomy clamp. Obstet Gynecol. 2015;126:935-938.
- Curtis KM, Jatlaoui TC, Tepper NK, et al. U.S. Selected Practice Recommendations for Contraceptive Use, 2016. MMWR Recomm Rep Morb Mortal Wkly. 2016;65:1-66. https://doi .org/10.15585/mmwr.rr6504a1
- Turok DK, Gero A, Simmons RG, et al. Levonorgestrel vs. copper intrauterine devices for emergency contraception. N Engl J Med. 2021;384:335-344. https://pubmed.ncbi.nlm .nih.gov/33503342/
- Kaiser JE, Turok DK, Gero A, et al. One-year pregnancy and continuation rates after placement of levonorgestrel or copper intrauterine devices for emergency contraception: a randomized controlled trial. Am J Obstet Gynecol. 2023;228:438.e1-438.e10. https://doi.org/10.1016/j.ajog.2022 .11.1296
- Sander PM, Raymond EG, Weaver MA. Emergency contraceptive use as a marker of future risky sex, pregnancy, and sexually transmitted infection. Am J Obstet Gynecol. 2009;201:146.e1-e6.
- Ramanadhan S, Goldstuck N, Henderson JT, et al. Progestin intrauterine devices versus copper intrauterine devices for emergency contraception. Cochrane Database Syst Rev. 2023;2:CD013744. https://doi.org/10.1002/14651858 .CD013744.pub2
Individuals spend close to half of their lives preventing, or planning for, pregnancy. As such, contraception plays a major role in patient-provider interactions. Contraception counseling and management is a common scenario encountered in the general gynecologist’s practice. Luckily, we have 2 evidence-based guidelines developed by the US Centers for Disease Control and Prevention (CDC) that support the provision of contraceptive care:
- US Medical Eligibility for Contraceptive Use (US-MEC),1 which provides guidance on which patients can safely use a method
- US Selected Practice Recommendations for Contraceptive Use (US-SPR),2 which provides method-specific guidance on how to use a method (including how to: initiate or start a method; manage adherence issues, such as a missed pill, etc; and manage common issues like breakthrough bleeding).
Both of these guidelines are updated routinely and are publicly available online or for free, through smartphone applications.
While most contraceptive care is straightforward, there are circumstances that require additional consideration. In the concluding part of this series on contraceptive conundrums, we review 2 clinical cases, existing evidence to guide management decisions, and our recommendations.
CASE 1 Patient presents with hard-to-remove implant
A 44-year-old patient (G2P2) with a new diagnosis of estrogen and progesterone-receptor–positive breast cancer is undergoing her evaluation with her oncologist who recommends removal of her contraceptive implant, which has been in place for 2 years. She presents to your office for removal; however, the device is no longer palpable.
What are your next steps?
Conundrum 1. Should you attempt to remove it?
No, never attempt implant removal if you cannot palpate or localize it. Localization of the implant needs to occur prior to any attempt. However, we recommend checking the contra-lateral arm before sending the patient to obtain imaging, especially if you have no formal documentation regarding in which arm the implant was placed. The next step is identifying what type of implant the patient likely has so you can correctly interpret imaging studies.
Conundrum 2. What type of subdermal contraceptive device is it likely to be?
Currently, the only subdermal contraceptive device available for placement in the United States is the 68-mg etonogestrel implant, marketed with the brand name Nexplanon. This device was initially approved by the US Food and Drug Administration in 2001 and measures 4 cm in length by 2 mm in diameter. It is placed in the medial upper arm, about 8 cm proximal to the medial epicondyle and 3 cm posterior to the sulcus between the biceps and triceps muscles. (The implant should no longer be placed over the bicipital groove.) The implant is impregnated with 15 mg of barium sulfate, making it radiopaque and able to be seen on imaging modalities such as ultrasonography (10–18 mHz high frequency transducer) and x-ray (arm anteroposterior and lateral) for localization in cases in which the device becomes nonpalpable.3
Clinicians also may encounter devices which are no longer marketed in the United States, or which are only available in other countries, and thus should be aware of the appearance and imaging characteristics. It is important to let your imaging team know these characteristics as well:
- From 2006–2010, a 68-mg etonogestrel implant marketed under the name Implanon was available in the United States.4 It has the same dimensions and general placement recommendations as the Nexplanon etonogestrel device but is not able to be seen via imaging.
- A 2-arm, 75-mg levonorgestrel (LNG) device known as Jadelle (or, Norplant II; FIGURE 1) received FDA approval in 1996 and is currently only available overseas.5 It is also placed in the upper, inner arm in a V-shape using a single incision, and has dimensions similar to the etonogestrel implants.
- From 1990– 2002, the 6-rod device known as Norplant was available in the United States. Each rod measured 3.4 cm in length and contained 36 mg of LNG (FIGURE 2).


Continue to: How do you approach removal of a deep contraceptive implant?...
How do you approach removal of a deep contraceptive implant?
Clinicians who are not trained in deep or difficult implant removal should refer patients to a trained provider (eg, a complex family planning subspecialist), or if not available, partner with a health care practitioner that has expertise in the anatomy of the upper arm (eg, vascular surgery, orthopedics, or interventional radiology). A resource for finding a nearby trained provider is the Organon Information Center (1-877-467-5266). However, when these services are not readily available, consider the following 3-step approach to complex implant removal.
- Be familiar with the anatomy of the upper arm (FIGURE 3). Nonpalpable implants may be close to or under the biceps or triceps fascia or be near critically important and fragile structures like the neurovascular bundle of the upper arm. Prior to attempting a difficult implant removal, ensure that you are well acquainted with critical structures in the upper arm.
- Locate the device. Prior to attempting removal, localize the device using either x-ray or ultrasonography, depending on local availability. Ultrasound offers the advantage of mapping the location in 3 dimensions, with the ability to map the device with skin markings immediately prior to removal. Typically, a highfrequency transducer (15- or 18-MHz) is used, such as for breast imaging, either in a clinician’s office or in coordination with radiology. If device removal is attempted the same day, the proximal, midportion, and distal aspects of the device should be marked with a skin pen, and it should be noted what position the arm is in when the device is marked (eg, arm flexed at elbow and externally rotated so that the wrist is parallel to the ear).

Rarely, if a device is not seen in the expected extremity, imaging of the contralateral arm or a chest x-ray can be undertaken to rule out mis-documented laterality or a migrated device. Lastly, if no device is seen, and the patient has no memory of device removal, you can obtain the patient’s etonogestrel levels. (Resource: Merck National Service Center, 1-877-888-4231.)
Removal procedure. For nonpalpable implants, strong consideration should be given to performing the procedure with ultrasonography guidance. Rarely, fluoroscopic guidance may be useful for orientation in challenging cases, which may require coordination with other services, such as interventional radiology.
Cleaning and anesthetizing the site is similar to routine removal of a palpable implant. A 2- to 3-mm skin incision is made, either at the distal end of the implant (if one end is amenable to traditional pop-out technique) or over the midportion of the device (if a clinician has experience using the “U” technique).6 The incision should be parallel to the long axis of the implant and not perpendicular, to facilitate extension of the incision if needed during the procedure. Straight or curved hemostat clamps can then be used for blunt dissection of the subcutaneous tissues and to grasp the end of the device. Experienced clinicians may have access to a modified vasectomy clamp (with a
Indications for referral. Typically, referral to a complex family planning specialist or vascular surgeon is required for cases that involve dissection of the muscular fascia or where dissection would be in close proximity to critical neurologic or vascular structures.
CASE 1 Conclusion
Ultrasonography of the patient’s extremity demonstrated a
CASE 2 Patient enquires about immediate IUD insertion
A 28-year-old patient (G1P0) arrives at your clinic for a contraceptive consultation. They report a condom break during intercourse 4 days ago. Prior to that they used condoms consistently with each act of intercourse. They have used combined hormonal contraceptive pills in the past but had difficulty remembering to take them consistently. The patient and their partner have been mutually monogamous for 6 months and have no plans for pregnancy. Last menstrual period was 12 days ago. Their cycles are regular but heavy and painful. They are interested in using a hormonal IUD for contraception and would love to get it today.
- Do not attempt removal of a nonpalpable implant without prior localization via imaging
- Ultrasound-guided removal procedures using a “U” technique are successful for many deep implant removals but require specialized equipment and training
- Referral to a complex family planning specialist or other specialist is highly recommended for implants located below the triceps fascia or close to the nerves and vessels of the upper arm
- Never attempt to remove a nonpalpable implant prior to determining its location via imaging
Continue to: Is same-day IUD an option?...
Is same-day IUD an option?
Yes. This patient needs EC given the recent condom break, but they are still eligible for having an IUD placed today if their pregnancy test is negative and after counseling of the potential risks and benefits. According to the US-SPR it is reasonable to insert an IUD at any time during the cycle as long as you are reasonably certain the patient is not pregnant.7
Options for EC are:
- 1.5-mg oral LNG pill
- 30-mg oral UPA pill
- copper IUD (cu-IUD).
If they are interested in the cu-IUD for long-term contraception, by having a cu-IUD placed they can get both their needs met—EC and an ongoing method of contraception. Any patient receiving EC, whether a pill or an IUD, should be counseled to repeat a home urine pregnancy test in 2 to 4 weeks.
Given the favorable non–contraceptive benefits associated with 52-mg LNG-IUDs, many clinicians and patients have advocated for additional evidence regarding the use of hormonal IUDs alone for EC.
What is the evidence concerning LNG-IUD placement as EC?
The 52-mg LNG-IUD has not been mechanistically proven to work as an EC, but growing evidence exists showing that it is safe for same-day or “quick start” placement even in a population seeking EC—if their pregnancy test result is negative at the time of presentation.
Turok and colleagues performed a noninferiority trial comparing 1-month pregnancy rates after placement of either an LNG-IUD or a cu-IUD for EC.8 This study concluded that the LNG-IUD (which resulted in 1 pregnancy in 317 users; pregnancy rate, 0.3%; 95% confidence interval [CI], 0.01–1.70) is noninferior to cu-IUD (0 pregnancies in 321 users; pregnancy rate, 0%; 95% CI, 0.0–1.1) for EC. Although encouraging, only a small percentage of the study population seeking EC who received an IUD were actually at high risk of pregnancy (eg, they were not mid-cycle or were recently using contraception), which is why it is difficult to determine if the LNG-IUD actually works mechanistically as an EC. More likely, the LNG-IUD helps prevent pregnancy due to its ongoing contraceptive effect.9 Ongoing acts of intercourse post–oral EC initiation without starting a method of contraception is one of the main reasons for EC failure, which is why starting a method immediately is so effective at preventing pregnancy.10
A systematic review conducted by Ramanadhan and colleagues concluded that Turok’s 2021 trial is the only relevant study specific to 52-mg LNG-IUD use as EC, but they also mention that its results are limited in the strength of its conclusions due to biases in randomization, including11:
- the study groups were not balanced in that there was a 10% difference in reported use of contraception at last intercourse, which means that the LNG-IUD group had a lower baseline risk of pregnancy
- and a rare primary outcome (ie, pregnancy, which requires a larger sample size to know if the method works as an EC).
The review authors concluded that more studies are needed to further validate the effectiveness of using the 52-mg LNG-IUD as EC. Thus, for those at highest risk of pregnancy from recent unprotected sex and desiring a 52-mg IUD, it is probably best to continue combining oral EC with a 52-mg LNG-IUD and utilizing the LNG-IUD only as EC on a limited, case-by-case basis.
What we recommend
For anyone with a negative pregnancy test on the day of presentation, the studies mentioned further support the practice of same-day placement of a 52-mg LNG-IUD. However, those seeking EC who are at highest risk for an unplanned pregnancy (ie, the unprotected sex was mid-cycle), we recommend co-administering the LNG-IUD with oral LNG for EC.
CASE 2 Conclusion
After a conversation with the patient about all contraceptive options, through shared decision making the patient decided to take 1.5 mg of oral LNG and have a 52-mg LNG-IUD placed in the office today. They do not wish to be pregnant at this time and would choose termination if they became pregnant. They understood their pregnancy risk and opted to plan a urine pregnancy test at home in 2 weeks with a clear understanding that they should return to clinic immediately if the test is positive. ●
- A copper IUD is the most effective method of emergency contraception (EC).
- 52-mg LNG-IUDs are an emerging consideration for EC, but evidence is still lacking that they work as EC (or whether they just prevent pregnancy after placement for subsequent acts of intercourse). Clinicians should utilize shared decision making and advise patients to repeat a pregnancy test at home in 2 to 4 weeks
- Any patient receiving EC, whether a pill or an IUD, should be counseled to repeat a home urine pregnancy test in 2 to 4 weeks
- Any type of IUD can be placed same day if the clinician is reasonably sure the patient is not pregnant
- It appears safe to co-administer the 52-mg LNG-IUD with oral EC for those seeking emergency contraception but also want to use an LNG-IUD for contraception going forward
Individuals spend close to half of their lives preventing, or planning for, pregnancy. As such, contraception plays a major role in patient-provider interactions. Contraception counseling and management is a common scenario encountered in the general gynecologist’s practice. Luckily, we have 2 evidence-based guidelines developed by the US Centers for Disease Control and Prevention (CDC) that support the provision of contraceptive care:
- US Medical Eligibility for Contraceptive Use (US-MEC),1 which provides guidance on which patients can safely use a method
- US Selected Practice Recommendations for Contraceptive Use (US-SPR),2 which provides method-specific guidance on how to use a method (including how to: initiate or start a method; manage adherence issues, such as a missed pill, etc; and manage common issues like breakthrough bleeding).
Both of these guidelines are updated routinely and are publicly available online or for free, through smartphone applications.
While most contraceptive care is straightforward, there are circumstances that require additional consideration. In the concluding part of this series on contraceptive conundrums, we review 2 clinical cases, existing evidence to guide management decisions, and our recommendations.
CASE 1 Patient presents with hard-to-remove implant
A 44-year-old patient (G2P2) with a new diagnosis of estrogen and progesterone-receptor–positive breast cancer is undergoing her evaluation with her oncologist who recommends removal of her contraceptive implant, which has been in place for 2 years. She presents to your office for removal; however, the device is no longer palpable.
What are your next steps?
Conundrum 1. Should you attempt to remove it?
No, never attempt implant removal if you cannot palpate or localize it. Localization of the implant needs to occur prior to any attempt. However, we recommend checking the contra-lateral arm before sending the patient to obtain imaging, especially if you have no formal documentation regarding in which arm the implant was placed. The next step is identifying what type of implant the patient likely has so you can correctly interpret imaging studies.
Conundrum 2. What type of subdermal contraceptive device is it likely to be?
Currently, the only subdermal contraceptive device available for placement in the United States is the 68-mg etonogestrel implant, marketed with the brand name Nexplanon. This device was initially approved by the US Food and Drug Administration in 2001 and measures 4 cm in length by 2 mm in diameter. It is placed in the medial upper arm, about 8 cm proximal to the medial epicondyle and 3 cm posterior to the sulcus between the biceps and triceps muscles. (The implant should no longer be placed over the bicipital groove.) The implant is impregnated with 15 mg of barium sulfate, making it radiopaque and able to be seen on imaging modalities such as ultrasonography (10–18 mHz high frequency transducer) and x-ray (arm anteroposterior and lateral) for localization in cases in which the device becomes nonpalpable.3
Clinicians also may encounter devices which are no longer marketed in the United States, or which are only available in other countries, and thus should be aware of the appearance and imaging characteristics. It is important to let your imaging team know these characteristics as well:
- From 2006–2010, a 68-mg etonogestrel implant marketed under the name Implanon was available in the United States.4 It has the same dimensions and general placement recommendations as the Nexplanon etonogestrel device but is not able to be seen via imaging.
- A 2-arm, 75-mg levonorgestrel (LNG) device known as Jadelle (or, Norplant II; FIGURE 1) received FDA approval in 1996 and is currently only available overseas.5 It is also placed in the upper, inner arm in a V-shape using a single incision, and has dimensions similar to the etonogestrel implants.
- From 1990– 2002, the 6-rod device known as Norplant was available in the United States. Each rod measured 3.4 cm in length and contained 36 mg of LNG (FIGURE 2).


Continue to: How do you approach removal of a deep contraceptive implant?...
How do you approach removal of a deep contraceptive implant?
Clinicians who are not trained in deep or difficult implant removal should refer patients to a trained provider (eg, a complex family planning subspecialist), or if not available, partner with a health care practitioner that has expertise in the anatomy of the upper arm (eg, vascular surgery, orthopedics, or interventional radiology). A resource for finding a nearby trained provider is the Organon Information Center (1-877-467-5266). However, when these services are not readily available, consider the following 3-step approach to complex implant removal.
- Be familiar with the anatomy of the upper arm (FIGURE 3). Nonpalpable implants may be close to or under the biceps or triceps fascia or be near critically important and fragile structures like the neurovascular bundle of the upper arm. Prior to attempting a difficult implant removal, ensure that you are well acquainted with critical structures in the upper arm.
- Locate the device. Prior to attempting removal, localize the device using either x-ray or ultrasonography, depending on local availability. Ultrasound offers the advantage of mapping the location in 3 dimensions, with the ability to map the device with skin markings immediately prior to removal. Typically, a highfrequency transducer (15- or 18-MHz) is used, such as for breast imaging, either in a clinician’s office or in coordination with radiology. If device removal is attempted the same day, the proximal, midportion, and distal aspects of the device should be marked with a skin pen, and it should be noted what position the arm is in when the device is marked (eg, arm flexed at elbow and externally rotated so that the wrist is parallel to the ear).

Rarely, if a device is not seen in the expected extremity, imaging of the contralateral arm or a chest x-ray can be undertaken to rule out mis-documented laterality or a migrated device. Lastly, if no device is seen, and the patient has no memory of device removal, you can obtain the patient’s etonogestrel levels. (Resource: Merck National Service Center, 1-877-888-4231.)
Removal procedure. For nonpalpable implants, strong consideration should be given to performing the procedure with ultrasonography guidance. Rarely, fluoroscopic guidance may be useful for orientation in challenging cases, which may require coordination with other services, such as interventional radiology.
Cleaning and anesthetizing the site is similar to routine removal of a palpable implant. A 2- to 3-mm skin incision is made, either at the distal end of the implant (if one end is amenable to traditional pop-out technique) or over the midportion of the device (if a clinician has experience using the “U” technique).6 The incision should be parallel to the long axis of the implant and not perpendicular, to facilitate extension of the incision if needed during the procedure. Straight or curved hemostat clamps can then be used for blunt dissection of the subcutaneous tissues and to grasp the end of the device. Experienced clinicians may have access to a modified vasectomy clamp (with a
Indications for referral. Typically, referral to a complex family planning specialist or vascular surgeon is required for cases that involve dissection of the muscular fascia or where dissection would be in close proximity to critical neurologic or vascular structures.
CASE 1 Conclusion
Ultrasonography of the patient’s extremity demonstrated a
CASE 2 Patient enquires about immediate IUD insertion
A 28-year-old patient (G1P0) arrives at your clinic for a contraceptive consultation. They report a condom break during intercourse 4 days ago. Prior to that they used condoms consistently with each act of intercourse. They have used combined hormonal contraceptive pills in the past but had difficulty remembering to take them consistently. The patient and their partner have been mutually monogamous for 6 months and have no plans for pregnancy. Last menstrual period was 12 days ago. Their cycles are regular but heavy and painful. They are interested in using a hormonal IUD for contraception and would love to get it today.
- Do not attempt removal of a nonpalpable implant without prior localization via imaging
- Ultrasound-guided removal procedures using a “U” technique are successful for many deep implant removals but require specialized equipment and training
- Referral to a complex family planning specialist or other specialist is highly recommended for implants located below the triceps fascia or close to the nerves and vessels of the upper arm
- Never attempt to remove a nonpalpable implant prior to determining its location via imaging
Continue to: Is same-day IUD an option?...
Is same-day IUD an option?
Yes. This patient needs EC given the recent condom break, but they are still eligible for having an IUD placed today if their pregnancy test is negative and after counseling of the potential risks and benefits. According to the US-SPR it is reasonable to insert an IUD at any time during the cycle as long as you are reasonably certain the patient is not pregnant.7
Options for EC are:
- 1.5-mg oral LNG pill
- 30-mg oral UPA pill
- copper IUD (cu-IUD).
If they are interested in the cu-IUD for long-term contraception, by having a cu-IUD placed they can get both their needs met—EC and an ongoing method of contraception. Any patient receiving EC, whether a pill or an IUD, should be counseled to repeat a home urine pregnancy test in 2 to 4 weeks.
Given the favorable non–contraceptive benefits associated with 52-mg LNG-IUDs, many clinicians and patients have advocated for additional evidence regarding the use of hormonal IUDs alone for EC.
What is the evidence concerning LNG-IUD placement as EC?
The 52-mg LNG-IUD has not been mechanistically proven to work as an EC, but growing evidence exists showing that it is safe for same-day or “quick start” placement even in a population seeking EC—if their pregnancy test result is negative at the time of presentation.
Turok and colleagues performed a noninferiority trial comparing 1-month pregnancy rates after placement of either an LNG-IUD or a cu-IUD for EC.8 This study concluded that the LNG-IUD (which resulted in 1 pregnancy in 317 users; pregnancy rate, 0.3%; 95% confidence interval [CI], 0.01–1.70) is noninferior to cu-IUD (0 pregnancies in 321 users; pregnancy rate, 0%; 95% CI, 0.0–1.1) for EC. Although encouraging, only a small percentage of the study population seeking EC who received an IUD were actually at high risk of pregnancy (eg, they were not mid-cycle or were recently using contraception), which is why it is difficult to determine if the LNG-IUD actually works mechanistically as an EC. More likely, the LNG-IUD helps prevent pregnancy due to its ongoing contraceptive effect.9 Ongoing acts of intercourse post–oral EC initiation without starting a method of contraception is one of the main reasons for EC failure, which is why starting a method immediately is so effective at preventing pregnancy.10
A systematic review conducted by Ramanadhan and colleagues concluded that Turok’s 2021 trial is the only relevant study specific to 52-mg LNG-IUD use as EC, but they also mention that its results are limited in the strength of its conclusions due to biases in randomization, including11:
- the study groups were not balanced in that there was a 10% difference in reported use of contraception at last intercourse, which means that the LNG-IUD group had a lower baseline risk of pregnancy
- and a rare primary outcome (ie, pregnancy, which requires a larger sample size to know if the method works as an EC).
The review authors concluded that more studies are needed to further validate the effectiveness of using the 52-mg LNG-IUD as EC. Thus, for those at highest risk of pregnancy from recent unprotected sex and desiring a 52-mg IUD, it is probably best to continue combining oral EC with a 52-mg LNG-IUD and utilizing the LNG-IUD only as EC on a limited, case-by-case basis.
What we recommend
For anyone with a negative pregnancy test on the day of presentation, the studies mentioned further support the practice of same-day placement of a 52-mg LNG-IUD. However, those seeking EC who are at highest risk for an unplanned pregnancy (ie, the unprotected sex was mid-cycle), we recommend co-administering the LNG-IUD with oral LNG for EC.
CASE 2 Conclusion
After a conversation with the patient about all contraceptive options, through shared decision making the patient decided to take 1.5 mg of oral LNG and have a 52-mg LNG-IUD placed in the office today. They do not wish to be pregnant at this time and would choose termination if they became pregnant. They understood their pregnancy risk and opted to plan a urine pregnancy test at home in 2 weeks with a clear understanding that they should return to clinic immediately if the test is positive. ●
- A copper IUD is the most effective method of emergency contraception (EC).
- 52-mg LNG-IUDs are an emerging consideration for EC, but evidence is still lacking that they work as EC (or whether they just prevent pregnancy after placement for subsequent acts of intercourse). Clinicians should utilize shared decision making and advise patients to repeat a pregnancy test at home in 2 to 4 weeks
- Any patient receiving EC, whether a pill or an IUD, should be counseled to repeat a home urine pregnancy test in 2 to 4 weeks
- Any type of IUD can be placed same day if the clinician is reasonably sure the patient is not pregnant
- It appears safe to co-administer the 52-mg LNG-IUD with oral EC for those seeking emergency contraception but also want to use an LNG-IUD for contraception going forward
- Curtis KM, Jatlaoui TC, Tepper NK, et al. U.S. Selected Practice Recommendations for Contraceptive Use, 2016. Morb Mortal Wkly Rep. 2016;65:1-66. https://doi .org/10.15585/mmwr .rr6504a1
- Centers for Disease Control and Prevention. National Center for Chronic Disease Prevention and Health Promotion, Division of Reproductive Health. US Selected Practice Recommendations for Contraceptive Use (US-SPR). Accessed October 11, 2023. https://www.cdc.gov/reproductivehealth /contraception/mmwr/spr/summary.html
- Nexplanon [package insert]. Whitehouse Station, NJ: Merck; 2018.
- US Food and Drug Administration. Implanon (etonogestrel implant) 2006. Accessed November 6, 2023. https://www .accessdata.fda.gov/drugsatfda_docs/nda/2006 /021529s000_Lbl.pdf
- US Food and Drug Administration. Jadelle (levonorgestrel implant) 2016. Accessed November 6, 2023. https://www. accessdata.fda.gov/drugsatfda_docs/label/2016/020544s 010lbl.pdf
- Chen MJ, Creinin MD. Removal of a nonpalpable etonogestrel implant with preprocedure ultrasonography and modified vasectomy clamp. Obstet Gynecol. 2015;126:935-938.
- Curtis KM, Jatlaoui TC, Tepper NK, et al. U.S. Selected Practice Recommendations for Contraceptive Use, 2016. MMWR Recomm Rep Morb Mortal Wkly. 2016;65:1-66. https://doi .org/10.15585/mmwr.rr6504a1
- Turok DK, Gero A, Simmons RG, et al. Levonorgestrel vs. copper intrauterine devices for emergency contraception. N Engl J Med. 2021;384:335-344. https://pubmed.ncbi.nlm .nih.gov/33503342/
- Kaiser JE, Turok DK, Gero A, et al. One-year pregnancy and continuation rates after placement of levonorgestrel or copper intrauterine devices for emergency contraception: a randomized controlled trial. Am J Obstet Gynecol. 2023;228:438.e1-438.e10. https://doi.org/10.1016/j.ajog.2022 .11.1296
- Sander PM, Raymond EG, Weaver MA. Emergency contraceptive use as a marker of future risky sex, pregnancy, and sexually transmitted infection. Am J Obstet Gynecol. 2009;201:146.e1-e6.
- Ramanadhan S, Goldstuck N, Henderson JT, et al. Progestin intrauterine devices versus copper intrauterine devices for emergency contraception. Cochrane Database Syst Rev. 2023;2:CD013744. https://doi.org/10.1002/14651858 .CD013744.pub2
- Curtis KM, Jatlaoui TC, Tepper NK, et al. U.S. Selected Practice Recommendations for Contraceptive Use, 2016. Morb Mortal Wkly Rep. 2016;65:1-66. https://doi .org/10.15585/mmwr .rr6504a1
- Centers for Disease Control and Prevention. National Center for Chronic Disease Prevention and Health Promotion, Division of Reproductive Health. US Selected Practice Recommendations for Contraceptive Use (US-SPR). Accessed October 11, 2023. https://www.cdc.gov/reproductivehealth /contraception/mmwr/spr/summary.html
- Nexplanon [package insert]. Whitehouse Station, NJ: Merck; 2018.
- US Food and Drug Administration. Implanon (etonogestrel implant) 2006. Accessed November 6, 2023. https://www .accessdata.fda.gov/drugsatfda_docs/nda/2006 /021529s000_Lbl.pdf
- US Food and Drug Administration. Jadelle (levonorgestrel implant) 2016. Accessed November 6, 2023. https://www. accessdata.fda.gov/drugsatfda_docs/label/2016/020544s 010lbl.pdf
- Chen MJ, Creinin MD. Removal of a nonpalpable etonogestrel implant with preprocedure ultrasonography and modified vasectomy clamp. Obstet Gynecol. 2015;126:935-938.
- Curtis KM, Jatlaoui TC, Tepper NK, et al. U.S. Selected Practice Recommendations for Contraceptive Use, 2016. MMWR Recomm Rep Morb Mortal Wkly. 2016;65:1-66. https://doi .org/10.15585/mmwr.rr6504a1
- Turok DK, Gero A, Simmons RG, et al. Levonorgestrel vs. copper intrauterine devices for emergency contraception. N Engl J Med. 2021;384:335-344. https://pubmed.ncbi.nlm .nih.gov/33503342/
- Kaiser JE, Turok DK, Gero A, et al. One-year pregnancy and continuation rates after placement of levonorgestrel or copper intrauterine devices for emergency contraception: a randomized controlled trial. Am J Obstet Gynecol. 2023;228:438.e1-438.e10. https://doi.org/10.1016/j.ajog.2022 .11.1296
- Sander PM, Raymond EG, Weaver MA. Emergency contraceptive use as a marker of future risky sex, pregnancy, and sexually transmitted infection. Am J Obstet Gynecol. 2009;201:146.e1-e6.
- Ramanadhan S, Goldstuck N, Henderson JT, et al. Progestin intrauterine devices versus copper intrauterine devices for emergency contraception. Cochrane Database Syst Rev. 2023;2:CD013744. https://doi.org/10.1002/14651858 .CD013744.pub2
Case Q: How soon after taking emergency contraception can a patient begin hormonal contraception?
Individuals spend close to half of their lives preventing, or planning for, pregnancy. As such, contraception plays a major role in patient-provider interactions. Contraception counseling and management is a common scenario encountered in the general gynecologist’s practice. Luckily, we have two evidence-based guidelines developed by the US Centers for Disease Control and Prevention (CDC) that support the provision of contraceptive care:
- US Medical Eligibility for Contraceptive Use (US-MEC),1 which provides guidance on which patients can safely use a method
- US Selected Practice Recommendations for Contraceptive Use (US-SPR),2 which provides method-specific guidance on how to use a method (including how to: initiate or start a method; manage adherence issues, such as a missed pill, etc; and manage common issues like breakthrough bleeding). Both of these guidelines are updated routinely and are publicly available online or for free, through smartphone applications.
While most contraceptive care is straightforward, there are circumstances that require additional consideration. In this 3-part series we review 3 clinical cases, existing evidence to guide management decisions, and our recommendations. In part 1, we focus on restarting hormonal contraception after ulipristal acetate administration. In parts 2 and 3, we will discuss removal of a nonpalpable contraceptive implant and the consideration of a levonorgestrel-releasing intrauterine device (LNG-IUD) for emergency contraception.
- After using ulipristal acetate for emergency contraception, advise patients to wait at least 5 days to initiate hormonal contraception and about the importance of abstaining or using a back-up method for another 7 days with the start of their hormonal contraceptive method
CASE Meeting emergency and follow-up contraception needs
A 27-year-old woman (G0) presents to you after having unprotected intercourse 4 days ago. She does not formally track her menstrual cycles and is unsure when her last menstrual period was. She is not using contraception but is interested in starting a method. After counseling, she elects to take a dose of oral ulipristal acetate (UPA; Ella) now for emergency contraception and would like to start a combined oral contraceptive (COC) pill moving forward.
How soon after taking UPA should you tell her to start the combined hormonal pill?
Effectiveness of hormonal contraception following UPA
UPA does not appear to decrease the efficacy of COCs when started around the same time. However, immediately starting a hormonal contraceptive can decrease the effectiveness of UPA, and as such, it is recommended to take UPA and then abstain or use a backup method for 7 days before initiating a hormonal contraceptive method.1 By obtaining some additional information from your patient and with the use of shared decision making, though, your patient may be able to start their contraceptive method earlier than 5 days after UPA.
What is UPA
UPA is a progesterone receptor modulator used for emergency contraception intenhded to prevent pregnancy after unprotected intercourse or contraceptive failure.3 It works by delaying follicular rupture at least 5 days, if taken before the peak of the luteinizing hormone (LH) surge. If taken after that timeframe, it does not work. Since UPA competes for the progesterone receptor, there is a concern that the effectiveness of UPA may be decreased if a progestin-containing form of contraception is started immediately after taking UPA, or vice versa.4 Several studies have now specifically looked at the interaction between UPA and progestin-containing contraceptives, including at how UPA is impacted by the contraceptive method, and conversely, how the contraceptive method is impacted by UPA.5-8
Data on types of hormonal contraception. Brache and colleagues demonstrated that UPA users who started a desogestrel progestin-only pill (DSG POP) the next day had higher rates of ovulation within 5 days of taking UPA (45%), compared with those who the next day started a placebo pill (3%).6 This type of progestin-only pill is not available in the United States.
A study by Edelman and colleagues demonstrated similar findings in those starting a COC pill containing estrogen and progestin. When taking a COC two days after UPA use, more participants had evidence of follicular rupture in less than 5 days.5 It should be noted that these studies focused on ovulation, which—while necessary for conception to occur—is a surrogate biomarker for pregnancy risk. Additional studies have looked at the impact of UPA on the COC and have not found that UPA impacts ovulation suppression of the COC with its initiation or use.8
Considering unprotected intercourse and UPA timing. Of course, the risk of pregnancy is reliant on cycle timing plus the presence of viable sperm in the reproductive tract. Sperm have been shown to only be viable in the reproductive tract for 5 days, which could result in fertilization and subsequent pregnancy. Longevity of an egg is much shorter, at 12 to 24 hours after ovulation. For this patient, her exposure was 4 days ago, but sperm are only viable for approximately 5 days—she could consider taking the UPA now and then starting a COC earlier than 5 days since she only needs an extra day or two of protection from the UPA from the sperm in her reproductive tract. Your patient’s involvement in this decision making is paramount, as only they can prioritize their desire to avoid pregnancy from their recent act of unprotected intercourse versus their immediate needs for starting their method of contraception. It is important that individuals abstain from sexual activity or use an additional back-up method during the first 7 days of starting their method of contraception.
Continue to: Counseling considerations for the case patient...
Counseling considerations for the case patient
For a patient planning to start or resume a hormonal contraceptive method after taking UPA, the waiting period recommended by the CDC (5 days) is most beneficial for patients who are uncertain about their menstrual cycle timing in relation to the act of unprotected intercourse that already occurred and need to prioritize maximum effectiveness of emergency contraception.
Patients with unsure cycle-sex timing planning to self-start or resume a short-term hormonal contraceptive method (eg, pills, patches, or rings), should be counseled to wait 5 days after the most recent act of unprotected sex, before taking their hormonal contraceptive method.7 Patients with unsure cycle-sex timing planning to use provider-dependent hormonal contraceptive methods (eg, those requiring a prescription, including a progestin-contraceptive implant or depot medroxyprogesterone acetate) should also be counseled to wait. Timing of levonorgestrel and copper intrauterine devices are addressed in part 3 of this series.
However, if your patient has a good understanding of their menstrual cycle, and the primary concern is exposure from subsequent sexual encounters and not the recent unprotected intercourse, it is advisable to provide UPA and immediately initiate a contraceptive method. One of the primary reasons for emergency contraception failure is that its effectiveness is limited to the most recent act of unprotected sexual intercourse and does not extend to subsequent acts throughout the month.
For these patients with sure cycle-sex timing who are planning to start or resume short-or long-term contraceptive methods, and whose primary concern is to prevent pregnancy risk from subsequent sexual encounters, immediately initiating a contraceptive method is advisable. For provider-dependent methods, we must weigh the risk of unintended pregnancy from the act of intercourse that already occurred (and the potential to increase that risk by initiating a method that could compromise UPA efficacy) versus the future risk of pregnancy if the patient cannot return for a contraception visit.7
In short, starting the contraceptive method at the time of UPA use can be considered after shared decision making with the patient and understanding what their primary concerns are.
Important point
Counsel on using backup barrier contraception after UPA
Oral emergency contraception only covers that one act of unprotected intercourse and does not continue to protect a patient from pregnancy for the rest of their cycle. When taken before ovulation, UPA works by delaying follicular development and rupture for at least 5 days. Patients who continue to have unprotected intercourse after taking UPA are at a high risk of an unintended pregnancy from this ‘stalled’ follicle that will eventually ovulate. Follicular maturation resumes after UPA’s effects wane, and the patient is primed for ovulation (and therefore unintended pregnancy) if ongoing unprotected intercourse occurs for the rest of their cycle.
Therefore, it is important to counsel patients on the need, if they do not desire a pregnancy, to abstain or start a method of contraception.
Final question
What about starting or resuming non–hormonal contraceptive methods?
Non-hormonal contraceptive methods can be started immediately with UPA use.1
CASE Resolved
After shared decision making, the patient decides to start using the COC pill. You prescribe her both UPA for emergency contraception and a combined hormonal contraceptive pill. Given her unsure cycle-sex timing, she expresses to you that her most important priority is preventing unintended pregnancy. You counsel her to set a reminder on her phone to start taking the pill 5 days from her most recent act of unprotected intercourse. You also counsel her to use a back-up barrier method of contraception for 7 days after starting her COC pill. ●

- Curtis KM, Jatlaoui TC, Tepper NK, et al. U.S. Selected Practice Recommendations for Contraceptive Use, 2016. Morb Mortal Wkly Rep. 2016;65:1-66. https://doi .org/10.15585/mmwr.rr6504a1
- Centers for Disease Control and Prevention. National Center for Chronic Disease Prevention and Health Promotion, Division of Reproductive Health. US Selected Practice Recommendations for Contraceptive Use (US-SPR). Accessed October 11, 2023. https://www.cdc.gov/reproductivehealth /contraception/mmwr/spr/summary.html
- Ella [package insert]. Charleston, SC; Afaxys, Inc. 2014.
- Salcedo J, Rodriguez MI, Curtis KM, et al. When can a woman resume or initiate contraception after taking emergency contraceptive pills? A systematic review. Contraception. 2013;87:602-604. https://doi.org/10.1016 /j.contraception.2012.08.013
- Edelman AB, Jensen JT, McCrimmon S, et al. Combined oral contraceptive interference with the ability of ulipristal acetate to delay ovulation: a prospective cohort study. Contraception. 2018;98:463-466. doi: 10.1016/j.contraception.2018.08.003
- Brache V, Cochon L, Duijkers IJM, et al. A prospective, randomized, pharmacodynamic study of quick-starting a desogestrel progestin-only pill following ulipristal acetate for emergency contraception. Hum Reprod Oxf Engl. 2015;30:2785-2793. https://doi.org/10.1093/humrep /dev241
- Cameron ST, Berger C, Michie L, et al. The effects on ovarian activity of ulipristal acetate when ‘quickstarting’ a combined oral contraceptive pill: a prospective, randomized, doubleblind parallel-arm, placebo-controlled study. Hum Reprod. 2015;30:1566-1572. doi: 10.1093/humrep/dev115
- Banh C, Rautenberg T, Diujkers I, et al. The effects on ovarian activity of delaying versus immediately restarting combined oral contraception after missing three pills and taking ulipristal acetate 30 mg. Contraception. 2020;102:145-151. doi: 10.1016/j.contraception.2020.05.013
- American Society for Emergency Contraception. Providing ongoing hormonal contraception after use of emergency contraceptive pills. September 2016. Accessed October 11, 2023. chrome-extension://efaidnbmnnnibpcajpcglclefindmkaj /https://www.americansocietyforec.org/_files/ugd/7f2e0b _ff1bc90bea204644ba28d1b0e6a6a6a8.pdf
Individuals spend close to half of their lives preventing, or planning for, pregnancy. As such, contraception plays a major role in patient-provider interactions. Contraception counseling and management is a common scenario encountered in the general gynecologist’s practice. Luckily, we have two evidence-based guidelines developed by the US Centers for Disease Control and Prevention (CDC) that support the provision of contraceptive care:
- US Medical Eligibility for Contraceptive Use (US-MEC),1 which provides guidance on which patients can safely use a method
- US Selected Practice Recommendations for Contraceptive Use (US-SPR),2 which provides method-specific guidance on how to use a method (including how to: initiate or start a method; manage adherence issues, such as a missed pill, etc; and manage common issues like breakthrough bleeding). Both of these guidelines are updated routinely and are publicly available online or for free, through smartphone applications.
While most contraceptive care is straightforward, there are circumstances that require additional consideration. In this 3-part series we review 3 clinical cases, existing evidence to guide management decisions, and our recommendations. In part 1, we focus on restarting hormonal contraception after ulipristal acetate administration. In parts 2 and 3, we will discuss removal of a nonpalpable contraceptive implant and the consideration of a levonorgestrel-releasing intrauterine device (LNG-IUD) for emergency contraception.
- After using ulipristal acetate for emergency contraception, advise patients to wait at least 5 days to initiate hormonal contraception and about the importance of abstaining or using a back-up method for another 7 days with the start of their hormonal contraceptive method
CASE Meeting emergency and follow-up contraception needs
A 27-year-old woman (G0) presents to you after having unprotected intercourse 4 days ago. She does not formally track her menstrual cycles and is unsure when her last menstrual period was. She is not using contraception but is interested in starting a method. After counseling, she elects to take a dose of oral ulipristal acetate (UPA; Ella) now for emergency contraception and would like to start a combined oral contraceptive (COC) pill moving forward.
How soon after taking UPA should you tell her to start the combined hormonal pill?
Effectiveness of hormonal contraception following UPA
UPA does not appear to decrease the efficacy of COCs when started around the same time. However, immediately starting a hormonal contraceptive can decrease the effectiveness of UPA, and as such, it is recommended to take UPA and then abstain or use a backup method for 7 days before initiating a hormonal contraceptive method.1 By obtaining some additional information from your patient and with the use of shared decision making, though, your patient may be able to start their contraceptive method earlier than 5 days after UPA.
What is UPA
UPA is a progesterone receptor modulator used for emergency contraception intenhded to prevent pregnancy after unprotected intercourse or contraceptive failure.3 It works by delaying follicular rupture at least 5 days, if taken before the peak of the luteinizing hormone (LH) surge. If taken after that timeframe, it does not work. Since UPA competes for the progesterone receptor, there is a concern that the effectiveness of UPA may be decreased if a progestin-containing form of contraception is started immediately after taking UPA, or vice versa.4 Several studies have now specifically looked at the interaction between UPA and progestin-containing contraceptives, including at how UPA is impacted by the contraceptive method, and conversely, how the contraceptive method is impacted by UPA.5-8
Data on types of hormonal contraception. Brache and colleagues demonstrated that UPA users who started a desogestrel progestin-only pill (DSG POP) the next day had higher rates of ovulation within 5 days of taking UPA (45%), compared with those who the next day started a placebo pill (3%).6 This type of progestin-only pill is not available in the United States.
A study by Edelman and colleagues demonstrated similar findings in those starting a COC pill containing estrogen and progestin. When taking a COC two days after UPA use, more participants had evidence of follicular rupture in less than 5 days.5 It should be noted that these studies focused on ovulation, which—while necessary for conception to occur—is a surrogate biomarker for pregnancy risk. Additional studies have looked at the impact of UPA on the COC and have not found that UPA impacts ovulation suppression of the COC with its initiation or use.8
Considering unprotected intercourse and UPA timing. Of course, the risk of pregnancy is reliant on cycle timing plus the presence of viable sperm in the reproductive tract. Sperm have been shown to only be viable in the reproductive tract for 5 days, which could result in fertilization and subsequent pregnancy. Longevity of an egg is much shorter, at 12 to 24 hours after ovulation. For this patient, her exposure was 4 days ago, but sperm are only viable for approximately 5 days—she could consider taking the UPA now and then starting a COC earlier than 5 days since she only needs an extra day or two of protection from the UPA from the sperm in her reproductive tract. Your patient’s involvement in this decision making is paramount, as only they can prioritize their desire to avoid pregnancy from their recent act of unprotected intercourse versus their immediate needs for starting their method of contraception. It is important that individuals abstain from sexual activity or use an additional back-up method during the first 7 days of starting their method of contraception.
Continue to: Counseling considerations for the case patient...
Counseling considerations for the case patient
For a patient planning to start or resume a hormonal contraceptive method after taking UPA, the waiting period recommended by the CDC (5 days) is most beneficial for patients who are uncertain about their menstrual cycle timing in relation to the act of unprotected intercourse that already occurred and need to prioritize maximum effectiveness of emergency contraception.
Patients with unsure cycle-sex timing planning to self-start or resume a short-term hormonal contraceptive method (eg, pills, patches, or rings), should be counseled to wait 5 days after the most recent act of unprotected sex, before taking their hormonal contraceptive method.7 Patients with unsure cycle-sex timing planning to use provider-dependent hormonal contraceptive methods (eg, those requiring a prescription, including a progestin-contraceptive implant or depot medroxyprogesterone acetate) should also be counseled to wait. Timing of levonorgestrel and copper intrauterine devices are addressed in part 3 of this series.
However, if your patient has a good understanding of their menstrual cycle, and the primary concern is exposure from subsequent sexual encounters and not the recent unprotected intercourse, it is advisable to provide UPA and immediately initiate a contraceptive method. One of the primary reasons for emergency contraception failure is that its effectiveness is limited to the most recent act of unprotected sexual intercourse and does not extend to subsequent acts throughout the month.
For these patients with sure cycle-sex timing who are planning to start or resume short-or long-term contraceptive methods, and whose primary concern is to prevent pregnancy risk from subsequent sexual encounters, immediately initiating a contraceptive method is advisable. For provider-dependent methods, we must weigh the risk of unintended pregnancy from the act of intercourse that already occurred (and the potential to increase that risk by initiating a method that could compromise UPA efficacy) versus the future risk of pregnancy if the patient cannot return for a contraception visit.7
In short, starting the contraceptive method at the time of UPA use can be considered after shared decision making with the patient and understanding what their primary concerns are.
Important point
Counsel on using backup barrier contraception after UPA
Oral emergency contraception only covers that one act of unprotected intercourse and does not continue to protect a patient from pregnancy for the rest of their cycle. When taken before ovulation, UPA works by delaying follicular development and rupture for at least 5 days. Patients who continue to have unprotected intercourse after taking UPA are at a high risk of an unintended pregnancy from this ‘stalled’ follicle that will eventually ovulate. Follicular maturation resumes after UPA’s effects wane, and the patient is primed for ovulation (and therefore unintended pregnancy) if ongoing unprotected intercourse occurs for the rest of their cycle.
Therefore, it is important to counsel patients on the need, if they do not desire a pregnancy, to abstain or start a method of contraception.
Final question
What about starting or resuming non–hormonal contraceptive methods?
Non-hormonal contraceptive methods can be started immediately with UPA use.1
CASE Resolved
After shared decision making, the patient decides to start using the COC pill. You prescribe her both UPA for emergency contraception and a combined hormonal contraceptive pill. Given her unsure cycle-sex timing, she expresses to you that her most important priority is preventing unintended pregnancy. You counsel her to set a reminder on her phone to start taking the pill 5 days from her most recent act of unprotected intercourse. You also counsel her to use a back-up barrier method of contraception for 7 days after starting her COC pill. ●

Individuals spend close to half of their lives preventing, or planning for, pregnancy. As such, contraception plays a major role in patient-provider interactions. Contraception counseling and management is a common scenario encountered in the general gynecologist’s practice. Luckily, we have two evidence-based guidelines developed by the US Centers for Disease Control and Prevention (CDC) that support the provision of contraceptive care:
- US Medical Eligibility for Contraceptive Use (US-MEC),1 which provides guidance on which patients can safely use a method
- US Selected Practice Recommendations for Contraceptive Use (US-SPR),2 which provides method-specific guidance on how to use a method (including how to: initiate or start a method; manage adherence issues, such as a missed pill, etc; and manage common issues like breakthrough bleeding). Both of these guidelines are updated routinely and are publicly available online or for free, through smartphone applications.
While most contraceptive care is straightforward, there are circumstances that require additional consideration. In this 3-part series we review 3 clinical cases, existing evidence to guide management decisions, and our recommendations. In part 1, we focus on restarting hormonal contraception after ulipristal acetate administration. In parts 2 and 3, we will discuss removal of a nonpalpable contraceptive implant and the consideration of a levonorgestrel-releasing intrauterine device (LNG-IUD) for emergency contraception.
- After using ulipristal acetate for emergency contraception, advise patients to wait at least 5 days to initiate hormonal contraception and about the importance of abstaining or using a back-up method for another 7 days with the start of their hormonal contraceptive method
CASE Meeting emergency and follow-up contraception needs
A 27-year-old woman (G0) presents to you after having unprotected intercourse 4 days ago. She does not formally track her menstrual cycles and is unsure when her last menstrual period was. She is not using contraception but is interested in starting a method. After counseling, she elects to take a dose of oral ulipristal acetate (UPA; Ella) now for emergency contraception and would like to start a combined oral contraceptive (COC) pill moving forward.
How soon after taking UPA should you tell her to start the combined hormonal pill?
Effectiveness of hormonal contraception following UPA
UPA does not appear to decrease the efficacy of COCs when started around the same time. However, immediately starting a hormonal contraceptive can decrease the effectiveness of UPA, and as such, it is recommended to take UPA and then abstain or use a backup method for 7 days before initiating a hormonal contraceptive method.1 By obtaining some additional information from your patient and with the use of shared decision making, though, your patient may be able to start their contraceptive method earlier than 5 days after UPA.
What is UPA
UPA is a progesterone receptor modulator used for emergency contraception intenhded to prevent pregnancy after unprotected intercourse or contraceptive failure.3 It works by delaying follicular rupture at least 5 days, if taken before the peak of the luteinizing hormone (LH) surge. If taken after that timeframe, it does not work. Since UPA competes for the progesterone receptor, there is a concern that the effectiveness of UPA may be decreased if a progestin-containing form of contraception is started immediately after taking UPA, or vice versa.4 Several studies have now specifically looked at the interaction between UPA and progestin-containing contraceptives, including at how UPA is impacted by the contraceptive method, and conversely, how the contraceptive method is impacted by UPA.5-8
Data on types of hormonal contraception. Brache and colleagues demonstrated that UPA users who started a desogestrel progestin-only pill (DSG POP) the next day had higher rates of ovulation within 5 days of taking UPA (45%), compared with those who the next day started a placebo pill (3%).6 This type of progestin-only pill is not available in the United States.
A study by Edelman and colleagues demonstrated similar findings in those starting a COC pill containing estrogen and progestin. When taking a COC two days after UPA use, more participants had evidence of follicular rupture in less than 5 days.5 It should be noted that these studies focused on ovulation, which—while necessary for conception to occur—is a surrogate biomarker for pregnancy risk. Additional studies have looked at the impact of UPA on the COC and have not found that UPA impacts ovulation suppression of the COC with its initiation or use.8
Considering unprotected intercourse and UPA timing. Of course, the risk of pregnancy is reliant on cycle timing plus the presence of viable sperm in the reproductive tract. Sperm have been shown to only be viable in the reproductive tract for 5 days, which could result in fertilization and subsequent pregnancy. Longevity of an egg is much shorter, at 12 to 24 hours after ovulation. For this patient, her exposure was 4 days ago, but sperm are only viable for approximately 5 days—she could consider taking the UPA now and then starting a COC earlier than 5 days since she only needs an extra day or two of protection from the UPA from the sperm in her reproductive tract. Your patient’s involvement in this decision making is paramount, as only they can prioritize their desire to avoid pregnancy from their recent act of unprotected intercourse versus their immediate needs for starting their method of contraception. It is important that individuals abstain from sexual activity or use an additional back-up method during the first 7 days of starting their method of contraception.
Continue to: Counseling considerations for the case patient...
Counseling considerations for the case patient
For a patient planning to start or resume a hormonal contraceptive method after taking UPA, the waiting period recommended by the CDC (5 days) is most beneficial for patients who are uncertain about their menstrual cycle timing in relation to the act of unprotected intercourse that already occurred and need to prioritize maximum effectiveness of emergency contraception.
Patients with unsure cycle-sex timing planning to self-start or resume a short-term hormonal contraceptive method (eg, pills, patches, or rings), should be counseled to wait 5 days after the most recent act of unprotected sex, before taking their hormonal contraceptive method.7 Patients with unsure cycle-sex timing planning to use provider-dependent hormonal contraceptive methods (eg, those requiring a prescription, including a progestin-contraceptive implant or depot medroxyprogesterone acetate) should also be counseled to wait. Timing of levonorgestrel and copper intrauterine devices are addressed in part 3 of this series.
However, if your patient has a good understanding of their menstrual cycle, and the primary concern is exposure from subsequent sexual encounters and not the recent unprotected intercourse, it is advisable to provide UPA and immediately initiate a contraceptive method. One of the primary reasons for emergency contraception failure is that its effectiveness is limited to the most recent act of unprotected sexual intercourse and does not extend to subsequent acts throughout the month.
For these patients with sure cycle-sex timing who are planning to start or resume short-or long-term contraceptive methods, and whose primary concern is to prevent pregnancy risk from subsequent sexual encounters, immediately initiating a contraceptive method is advisable. For provider-dependent methods, we must weigh the risk of unintended pregnancy from the act of intercourse that already occurred (and the potential to increase that risk by initiating a method that could compromise UPA efficacy) versus the future risk of pregnancy if the patient cannot return for a contraception visit.7
In short, starting the contraceptive method at the time of UPA use can be considered after shared decision making with the patient and understanding what their primary concerns are.
Important point
Counsel on using backup barrier contraception after UPA
Oral emergency contraception only covers that one act of unprotected intercourse and does not continue to protect a patient from pregnancy for the rest of their cycle. When taken before ovulation, UPA works by delaying follicular development and rupture for at least 5 days. Patients who continue to have unprotected intercourse after taking UPA are at a high risk of an unintended pregnancy from this ‘stalled’ follicle that will eventually ovulate. Follicular maturation resumes after UPA’s effects wane, and the patient is primed for ovulation (and therefore unintended pregnancy) if ongoing unprotected intercourse occurs for the rest of their cycle.
Therefore, it is important to counsel patients on the need, if they do not desire a pregnancy, to abstain or start a method of contraception.
Final question
What about starting or resuming non–hormonal contraceptive methods?
Non-hormonal contraceptive methods can be started immediately with UPA use.1
CASE Resolved
After shared decision making, the patient decides to start using the COC pill. You prescribe her both UPA for emergency contraception and a combined hormonal contraceptive pill. Given her unsure cycle-sex timing, she expresses to you that her most important priority is preventing unintended pregnancy. You counsel her to set a reminder on her phone to start taking the pill 5 days from her most recent act of unprotected intercourse. You also counsel her to use a back-up barrier method of contraception for 7 days after starting her COC pill. ●

- Curtis KM, Jatlaoui TC, Tepper NK, et al. U.S. Selected Practice Recommendations for Contraceptive Use, 2016. Morb Mortal Wkly Rep. 2016;65:1-66. https://doi .org/10.15585/mmwr.rr6504a1
- Centers for Disease Control and Prevention. National Center for Chronic Disease Prevention and Health Promotion, Division of Reproductive Health. US Selected Practice Recommendations for Contraceptive Use (US-SPR). Accessed October 11, 2023. https://www.cdc.gov/reproductivehealth /contraception/mmwr/spr/summary.html
- Ella [package insert]. Charleston, SC; Afaxys, Inc. 2014.
- Salcedo J, Rodriguez MI, Curtis KM, et al. When can a woman resume or initiate contraception after taking emergency contraceptive pills? A systematic review. Contraception. 2013;87:602-604. https://doi.org/10.1016 /j.contraception.2012.08.013
- Edelman AB, Jensen JT, McCrimmon S, et al. Combined oral contraceptive interference with the ability of ulipristal acetate to delay ovulation: a prospective cohort study. Contraception. 2018;98:463-466. doi: 10.1016/j.contraception.2018.08.003
- Brache V, Cochon L, Duijkers IJM, et al. A prospective, randomized, pharmacodynamic study of quick-starting a desogestrel progestin-only pill following ulipristal acetate for emergency contraception. Hum Reprod Oxf Engl. 2015;30:2785-2793. https://doi.org/10.1093/humrep /dev241
- Cameron ST, Berger C, Michie L, et al. The effects on ovarian activity of ulipristal acetate when ‘quickstarting’ a combined oral contraceptive pill: a prospective, randomized, doubleblind parallel-arm, placebo-controlled study. Hum Reprod. 2015;30:1566-1572. doi: 10.1093/humrep/dev115
- Banh C, Rautenberg T, Diujkers I, et al. The effects on ovarian activity of delaying versus immediately restarting combined oral contraception after missing three pills and taking ulipristal acetate 30 mg. Contraception. 2020;102:145-151. doi: 10.1016/j.contraception.2020.05.013
- American Society for Emergency Contraception. Providing ongoing hormonal contraception after use of emergency contraceptive pills. September 2016. Accessed October 11, 2023. chrome-extension://efaidnbmnnnibpcajpcglclefindmkaj /https://www.americansocietyforec.org/_files/ugd/7f2e0b _ff1bc90bea204644ba28d1b0e6a6a6a8.pdf
- Curtis KM, Jatlaoui TC, Tepper NK, et al. U.S. Selected Practice Recommendations for Contraceptive Use, 2016. Morb Mortal Wkly Rep. 2016;65:1-66. https://doi .org/10.15585/mmwr.rr6504a1
- Centers for Disease Control and Prevention. National Center for Chronic Disease Prevention and Health Promotion, Division of Reproductive Health. US Selected Practice Recommendations for Contraceptive Use (US-SPR). Accessed October 11, 2023. https://www.cdc.gov/reproductivehealth /contraception/mmwr/spr/summary.html
- Ella [package insert]. Charleston, SC; Afaxys, Inc. 2014.
- Salcedo J, Rodriguez MI, Curtis KM, et al. When can a woman resume or initiate contraception after taking emergency contraceptive pills? A systematic review. Contraception. 2013;87:602-604. https://doi.org/10.1016 /j.contraception.2012.08.013
- Edelman AB, Jensen JT, McCrimmon S, et al. Combined oral contraceptive interference with the ability of ulipristal acetate to delay ovulation: a prospective cohort study. Contraception. 2018;98:463-466. doi: 10.1016/j.contraception.2018.08.003
- Brache V, Cochon L, Duijkers IJM, et al. A prospective, randomized, pharmacodynamic study of quick-starting a desogestrel progestin-only pill following ulipristal acetate for emergency contraception. Hum Reprod Oxf Engl. 2015;30:2785-2793. https://doi.org/10.1093/humrep /dev241
- Cameron ST, Berger C, Michie L, et al. The effects on ovarian activity of ulipristal acetate when ‘quickstarting’ a combined oral contraceptive pill: a prospective, randomized, doubleblind parallel-arm, placebo-controlled study. Hum Reprod. 2015;30:1566-1572. doi: 10.1093/humrep/dev115
- Banh C, Rautenberg T, Diujkers I, et al. The effects on ovarian activity of delaying versus immediately restarting combined oral contraception after missing three pills and taking ulipristal acetate 30 mg. Contraception. 2020;102:145-151. doi: 10.1016/j.contraception.2020.05.013
- American Society for Emergency Contraception. Providing ongoing hormonal contraception after use of emergency contraceptive pills. September 2016. Accessed October 11, 2023. chrome-extension://efaidnbmnnnibpcajpcglclefindmkaj /https://www.americansocietyforec.org/_files/ugd/7f2e0b _ff1bc90bea204644ba28d1b0e6a6a6a8.pdf
