User login
Food allergy and eosinophilic esophagitis: Learning what to avoid
More children and even adults seem to be allergic to various foods these days than in the past. Also apparently on the rise is a linked condition, eosinophilic esophagitis.
The reason for these increases is not clear. This article confines itself to what we know about the mechanisms of food allergies and eosinophilic esophagitis, how to diagnose them, and how to treat them.
FOOD ALLERGIES ARE COMMON, AND MORE PREVALENT THAN EVER
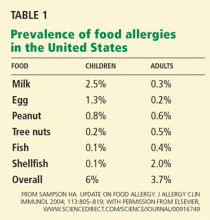
Food allergies—abnormal immune responses to food proteins1—affect an estimated 6% to 8% of young children and 3% to 4% of adults in the United States,2,3 and their prevalence appears to be rising in developed countries. Studies in US and British children indicate that peanut allergy has doubled in the past decade. 4
Any food can provoke a reaction, but only a few foods account for most of the significant allergic reactions: cow’s milk, soy, wheat, eggs, peanuts, tree nuts, fish, and shellfish.
Approximately 80% of allergies to milk, egg, wheat, and soy resolve by the time the patient reaches early adolescence.6 Fewer cases resolve in children with tree nut allergies (approximately 9%) or peanut allergy (20%),7,8 and allergies to fish and shellfish often develop or persist in adulthood.
A family history of an atopic disease such as asthma, allergic rhinitis, eczema, or food allergy is a risk factor for developing a food allergy. 3 Considering that the rate of peanut allergy has doubled in children over the past 10 years, environmental factors may also play a role.3
How we tolerate foods or become allergic to them
The gut, the largest mucosal organ in the body, is exposed to large quantities of foreign proteins daily. Most protein is broken down by stomach acid and digestive enzymes into lessantigenic peptides or is bound by secretory immunoglobulin A (IgA), which prevents it from being absorbed. Further, the epithelial cells lining the gut do not allow large molecules to pass easily, having tight intracellular junctions and being covered with mucus.
For these reasons, less than 2% of the protein in food is absorbed in an allergenic form.9 The reason food allergies are more prevalent in children is most likely that children have an immature gut barrier, lower IgA levels, a higher gastric pH, and lower proteolytic enzyme levels.
When dietary proteins do cross the gut barrier, the immune system normally suppresses the allergic response. Regulatory T cells, dendritic cells, and local immune responses play critical roles in the development of tolerance. Several types of regulatory T cells, such as Tr1 cells (which secrete interleukin 10), TH3 cells (which secrete transforming growth factor beta), CD4+CD25+ regulatory T cells, gamma-delta T cells, and CD8+ suppressor cells can all contribute to suppressing allergic responses.10 Dendritic cells also help induce tolerance by stimulating CD4+ T cells to secrete transforming growth factor beta, which leads to the production of interleukin 10 and additional transforming growth factor beta.11
Factors that contribute to food allergy
Many factors may contribute to whether a person becomes tolerant to or sensitized to a specific food protein.
The dose of antigen. Tolerance can develop after either high or low doses of antigens, but by different mechanisms.
The antigen structure. Soluble antigens are less sensitizing than particulate antigens.12,13
Processing of foods. Dry-roasted peanuts are more allergenic than raw or boiled peanuts, partly because they are less soluble.13
The route of initial exposure. Sensitization to food proteins can occur directly through the gut or the skin. Alternatively, it can occur indirectly via the respiratory tract. Skin exposure may be especially sensitizing in children with atopic dermatitis.14,15
The gut flora. When mice are raised in a germ-free environment, they fail to develop normal tolerance.16 They are also more likely to become sensitized if they are treated with antibiotics or if they lack toll-like receptors that recognize bacterial lipopolysaccharides.17 Furthermore, human studies suggest that probiotics promote tolerance, especially in preventing atopic dermatitis, although the studies have had conflicting results.18–21
The gastric pH. Murine and human studies reveal that antacid medications increase the risk of food allergy.22,23
Genetic susceptibility. A child with a sibling who is allergic to peanuts is approximately 10 times more likely to be allergic to peanuts than predicted by the rate in the general population. Although no risk-conferring gene has been identified, a study of twins showed concordance for peanut allergy in 64.3% of identical twins vs 6.8% of fraternal twins.24
Three types of immune responses to food
Immunologic reactions to foods can be divided into three categories: mediated by immunoglobulin E (IgE), non-IgE-mediated, and mixed. Therefore, these disorders can present as an acute, potentially life-threatening reaction or as a chronic disease such as eosinophilic gastoenteropathy.
IgE-mediated reactions are immediate hypersensitivity responses. In most patients, an IgE-mediated mechanism can be confirmed by a positive skin test or a test for food-specific IgE in the serum. In this article, the term “food allergy” refers to an IgE-mediated reaction to a food, unless otherwise indicated.
Non-IgE-mediated reactions have a delayed onset and chronic symptoms. Commonly, they are confined to the gastrointestinal tract; examples are food-protein-induced enterocolitis, proctitis, and proctocolitis and celiac disease.3,26,27 However, other diseases such as contact dermatitis, dermatitis herpetiformis, and food-induced pulmonary hemosiderosis (Heiner syndrome) are also considered non-IgE-mediated allergies.
Mixed-reaction disorders are chronic and include the eosinophilic gastroenteropathies, ie, eosinophilic proctocolitis, eosinophilic gastroenteritis, and eosinophilic esophagitis.28 The pathophysiology of these diseases is poorly understood. Many patients have evidence of allergic sensitivities to food or to environmental allergens, or both, but whether these sensitivities have a causal role in these disorders is not clear.
Atopic dermatitis, another complicated disease process, may be associated with mixedreaction food allergy, as approximately 35% of young children with moderate to severe atopic dermatitis have food allergies.29
Diagnosis of IgE-mediated food allergies
A thorough history and physical examination are key to diagnosing an IgE-mediated food allergy.
The history should include potential culprit foods, the quantity eaten, the timing of the onset of symptoms, and related factors such as exercise, alcohol intake, or medication use. Symptoms of an IgE-mediated reaction are generally rapid in onset but may be delayed up to a few hours, while non-IgE mediated symptoms may present several hours to days later.
Food challenge. A double-blind, placebocontrolled oral food challenge is the gold standard for the diagnosis of food allergies. (The food to be tested is hidden in other food or in capsules.) However, this test poses significant risks, and other diagnostic methods are more practical for screening.
Skin-prick tests with commercially available extracts are a rapid and sensitive method of screening for allergy to several foods.
Negative skin-prick tests have an estimated negative predictive value of more than 95% and can therefore exclude IgE-mediated food allergies.
A positive test indicates the presence of IgE against a specific food allergen and suggests a clinical food allergy, although the specificity of the test is only about 50%, making a positive result difficult to interpret. Although the size of the skin-test response does not necessarily correlate with the potential severity of a reaction, a response larger than 3 mm does indicate a greater likelihood of clinical reactivity. A positive test is most helpful in confirming the diagnosis of IgE-mediated food allergy when combined with a clear history of food-induced symptoms.
The proteins in commercially based extracts of most fruits and vegetables are often labile; therefore, skin testing with fresh fruits and vegetables may be indicated.30
Immunoassays. Radioallergosorbent tests (RASTs) and fluorescent enzyme immunoassays are used to identity food-specific IgE antibodies in the serum. The commercially available tests do not use radioactivity, but the term “RAST” is still commonly used.
Immunoassays are generally less sensitive and more costly than skin-prick tests, and their results are not immediately available, unlike those of skin-prick testing. However, these in vitro tests are not affected by antihistamine use and are useful in patients with severe dermatologic conditions or severe anaphylaxis, for whom skin-prick testing would not be appropriate.
However, unlike a negative skin-prick test, an undetectable serum food-specific IgE level has a low negative predictive value, and an undetectable level may be associated with symptoms of an allergic reaction for 10% to 25% of patients.29 Therefore, if one suspects an allergic reaction but no food-specific IgE can be detected in the serum, confirming the absence of a clinical allergy must be done with a skin-prick test or with a physician-supervised oral challenge, or both.
Managing food allergy by avoiding the allergen
Food allergies are managed by strictly avoiding food allergens and by taking medications such as self-injectable epinephrine for anaphylactic symptoms.
Patients and caregivers must be educated about reading food labels, avoiding high-risk situations such as eating at buffets and other restaurants with high risk of cross-contamination, wearing a medical-alert bracelet, recognizing and managing early symptoms of an allergic reaction, and calling for emergency services if they are having an allergic reaction. Since January 2006, the US Food and Drug Administration has required food manufacturers to list common food allergens on food labels (cow’s milk, soy, wheat, egg, peanut, tree nuts, fish, and shellfish), and the labeling must use simple, easily understood terms, such as “milk” instead of “whey.” However, it is still prudent to read all ingredients listed on the label.
Experimental treatments for food allergies
Humanized monoclonal anti-IgE antibodies such as talizumab (also known as TNX-901) and omalizumab (Xolair) have been developed, but their use in food allergy has been limited. In a study in patients with peanut allergy, injections of talizumab increased the threshold for sensitivity to peanuts in most patients, but 25% of the patients did not have any improvement.32 A study of omalizumab in patients with peanut allergy was stopped after adverse reactions developed during oral peanut challenges.33
Oral immunotherapy. Recent studies suggest it may be possible to induce oral tolerance in patients with IgE-mediated food allergy. Pilot studies have shown that frequent, increasing doses of food allergens (egg, milk, and peanut) may raise the threshold at which symptoms occur.34–36 Though these studies suggest that oral immunotherapy may protect some patients against a reaction if they accidentally ingest a food they are allergic to, some patients could not reach the goal doses because allergic symptoms were provoked.
At this early stage, these strategies must be considered investigational, and more randomized, placebo-controlled studies are needed. Further studies will also be needed to assess whether oral immunotherapy induces only short-term desensitization (in which case the allergen needs to be ingested daily to prevent reactions) or sustained tolerance (in which case the antigenic protein can be ingested without symptoms despite periods of abstinence).
THE ROLE OF FOOD ALLERGY IN EOSINOPHILIC ESOPHAGITIS
Eosinophilic esophagitis has been recognized with increasing frequency in both children and adults over the past several years. Symptoms can include difficulty feeding, failure to thrive, vomiting, epigastric or chest pain, dysphagia, and food impaction.
Diagnostic criteria for eosinophilic esophagitis are37:
- Clinical symptoms of esophageal dysfunction
- At least 15 eosinophils per high-power field in at least one esophageal biopsy specimen
- No response to a proton-pump inhibitor in high doses (up to 2 mg/kg/day) for 1 to 2 months, or normal results on pH probe monitoring of the esophagus (the reason for this criterion is that patients with gastroesophageal reflux disease can also have large numbers of eosinophils in the esophagus—more than 100 per highpower field38)
- Exclusion of other causes.
Though the cause of eosinophilic esophagitis is not completely understood, atopy has been strongly implicated as a factor. More than 50% of patients with eosinophilic esophagitis also have an atopic condition (eg, atopic dermatitis, allergic rhinitis, asthma), as well as positive results on skin-prick testing or measurement of antigen-specific IgE in the serum.39–41 Also, since most patients improve with either dietary restriction or elemental diets, food sensitization appears to play a considerable role.
As with atopic conditions such as asthma, atopic dermatitis, allergic rhinitis, and food allergy, eosinophilic esophagitis has been linked with immune responses involving helper T cell 2 (TH2). Adults and children with eosinophilic esophagitis have been found to have elevated eosinophil counts and total IgE levels in peripheral blood.37 In the esophagus, patients have elevated levels of the TH2 cytokines often seen in atopic patients (eg, interleukins 4, 5, and 13) and mast cells.42,43 In mice, eosinophilic esophagitis can be induced by allergen exposure and overexpression of TH2 cytokines.44,45 Expression of eotaxin-3, a potent eosinophil chemoattractant, was noted to be higher in children with eosinophilic esophagitis than in controls.46
Of interest, some patients with eosinophilic esophagitis say their symptoms vary with the seasons, correlating with seasonal changes in esophageal eosinophil levels.47,48
Studies linking eosinophilic esophagitis and food allergy in children
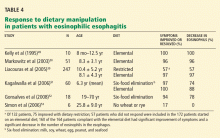
Kelly et al49 reported that 10 children with chronic symptomatic gastroesophageal reflux and eosinophilic esophagitis all had partial or complete resolution of symptoms on an elemental diet.
Markowitz et al50 found that symptoms of chronic reflux disease and eosinophilic esophagitis improved in 49 of 51 children on an elemental diet, and the number of eosinophils in the distal esophagus decreased significantly.
Liacouras et al39 reported similar findings in a 10-year experience. Of 132 children who had eosinophilic esophagitis, 75 improved with dietary restriction based on results of skin-prick and patch testing. The 57 patients who did not respond and 115 others were started on an elemental diet. Of the 164 patients who complied with the elemental diet, 160 had significant improvement of symptoms and a significant decrease in the number of eosinophils in the esophagus. Individual foods were reintroduced approximately every 5 days, and esophagogastroduodenoscopy with biopsies was performed 4 to 8 weeks after the last was reintroduced into the diet.
In a retrospective study, Kagalwalla et al51 reported that 60 children with eosinophilic esophagitis were treated with either an elemental diet or a six-food elimination diet (no milk, soy, wheat, egg, peanut, or seafood). The two groups showed similar clinical and histologic improvements.
Collectively, these studies in pediatric patients imply that food allergy is a significant factor in the pathogenesis of eosinophilic esophagitis.
Studies in adults
Fewer studies of the link between food allergy and eosinophilic esophagitis have been done in adults.
In a preliminary study, 18 adults followed the six-food elimination diet. Symptoms improved in 17 (94%), and histologic findings improved in 14 (78%).52
On the other hand, in six adult patients with eosinophilic esophagitis, Simon et al53 found that only one had improvement in symptoms after eliminating wheat and rye from the diet, and none had significant changes in the number of eosinophils in the esophagus.
In a 37-year-old man with eosinophilic esophagitis, symptoms improved after eliminating egg from his diet.54
Yamazaki et al55 measured expression of interleukin 5 and interleukin 13 in 15 adult patients with eosinophilic esophagitis. Food and aeroallergens that included milk, soy, dust mite, ragweed, and Aspergillus induced significantly more interleukin 5 production in these patients than in atopic controls, suggesting that both foods and aeroallergens may have a role in the pathogenesis of eosinophilic esophagitis in adults.
How to identify potential food triggers of eosinophilic esophagitis
Though elemental diets have been associated with a decrease in symptoms and esophageal eosinophilia, elemental formulas are expensive and unpalatable and pose a risk of nutritional deprivation. Identifying specific food allergens to eliminate from the diet in patients with eosinophilic esophagitis may be less expensive and more desirable than a very limited or elemental diet.
However, potential food triggers have been hard to identify in eosinophilic esophagitis. A recent consensus report did not recommend in vitro food allergy testing,37 owing to a lack of positive or negative predictive values for food-specific IgE level testing in eosinophilic esophagitis. Furthermore, the absence of IgE does not eliminate a food as a potential trigger, since non-IgE mechanisms may play a role.
Skin-prick testing is one of the currently validated diagnostic methods. Several studies have used skin-prick testing of foods in patients with eosinophilic esophagitis. In these studies, approximately two-thirds of patients had positive test reactions to at least one food, most often to common food allergens such as cow’s milk, egg, soy, wheat, and peanut, but also to rye, beef, and bean.37 In a recent article,56 81% of adult patients with eosinophilic esophagitis had one or more allergens identified by skin-prick testing, and 50% of the patients tested positive for one or more food allergens.
Atopy patch testing. The combination of skin-prick testing and atopy patch testing may be more effective than skin-prick testing alone in identifying potential food triggers. Atopy patch testing has been used in the diagnosis of non-IgE cell-mediated (delayed) immune responses, in which T cells may play a significant role.
Atopy patch testing is similar to patch testing for contact dermatitis. It involves placing a small quantity of food on the skin and evaluating for a local delayed reaction after a set time.
In two studies,50,57 146 children with biopsy-proven eosinophilic esophagitis had foods eliminated from the diet on the basis of positive skin-prick tests and atopy patch tests. Approximately 77% of the children had significant reduction of esophageal eosinophils in biopsy specimens (from 20 per high-power field to 1.1). The foods most commonly implicated by skin-prick testing were cow’s milk, egg, wheat, peanut, shellfish, peas, beef, fish, rye, and tomato; those identified by atopy patch testing were cow’s milk, egg, wheat, corn, beef, milk, soy, rye, chicken, oats, and potato. The combination of both types of testing had a negative predictive value of 88% to 100% for all foods except milk, while the positive predictive value was greater than 74% for the most common foods causing eosinophilic esophagitis.58
Though atopy patch testing shows some usefulness in identifying foods that may elicit non-IgE-mediated reactions, currently these tests are not validated and have been evaluated in only a small number of studies. Currently, no standardized testing materials, methods of application, or interpretation of results exist, and no studies have included a control population to validate atopy patch testing. More studies are needed to validate atopy patch testing as a reliable diagnostic tool before it can be recommended as a component of routine diagnostic evaluation in patients with eosinophilic esophagitis.
- Bruijnzeel-Koomen C, Ortolani C, Aas K, et al. Adverse reactions to food. European Academy of Allergology and Clinical Immunology Subcommittee. Allergy 1995; 50:623–635.
- Sampson HA. Update on food allergy. J Allergy Clin Immunol 2004; 113:805–819.
- Sicherer SH, Sampson HA. 9. Food allergy. J Allergy Clin Immunol 2006; 117 (suppl 2):S470–S475.
- Sicherer SH, Munoz-Furlong A, Sampson HA. Prevalence of peanut and tree nut allergy in the United States determined by means of a random digit dial telephone survey: a 5-year follow-up study. J Allergy Clin Immunol 2003; 112:1203–1207.
- American College of Allergy, Asthma, & Immunology. Food allergy: a practice parameter. Ann Allergy Asthma Immunol 2006; 96( suppl 2):S1–S68.
- Wood RA. The natural history of food allergy. Pediatrics 2003; 111:1631–1637.
- Hourihane JO, Roberts SA, Warner JO. Resolution of peanut allergy: case-control study. BMJ 1998; 316:1271–1275.
- Fleischer DM, Conover-Walker MK, Matsui EC, Wood RA. The natural history of tree nut allergy. J Allergy Clin Immunol 2005; 116:1087–1093.
- Husby S, Foged N, Host A, Svehag SE. Passage of dietary antigens into the blood of children with coeliac disease. Quantification and size distribution of absorbed antigens. Gut 1987; 28:1062–1072.
- Mowat AM. Anatomical basis of tolerance and immunity to intestinal antigens. Nat Rev Immunol 2003; 3:331–341.
- Frossard CP, Tropia L, Hauser C, Eigenmann PA. Lymphocytes in Peyer patches regulate clinical tolerance in a murine model of food allergy. J Allergy Clin Immunol 2004; 113:958–964.
- Jain SL, Barone KS, Flanagan MP, Michael JG. Activation patterns of murine B cells after oral administration of an encapsulated soluble antigen. Vaccine 1996; 14:1291–1297.
- Kopper RA, Odum NJ, Sen M, Helm RM, Stanley JS, Burks AW. Peanut protein allergens: the effect of roasting on solubility and allergenicity. Int Arch Allergy Immunol 2005; 136:16–22.
- Lack G. Epidemiologic risks for food allergy. J Allergy Clin Immunol 2008; 121:1331–1336.
- Lack G, Fox D, Northstone K, Golding J; Avon Longitudinal Study of Parents and Children Study Team. Factors associated with the development of peanut allergy in childhood. N Engl J Med 2003; 348:977–985.
- Sudo N, Sawamura S, Tanaka K, Aiba Y, Kubo C, Koga Y. The requirement of intestinal bacterial flora for the development of an IgE production system fully susceptible to oral tolerance induction. J Immunol 1997; 159:1739–1745.
- Bashir ME, Louie S, Shi HN, Nagler-Anderson C. Toll-like receptor 4 signaling by intestinal microbes influences susceptibility to food allergy. J Immunol 2004; 172:6978–6987.
- Kopp MV, Hennemuth I, Heinzmann A, Urbanek R. Randomized, double-blind, placebo-controlled trial of probiotics for primary prevention: no clinical effects of lactobacillus GG supplementation. Pediatrics 2008; 121:e850–e856.
- Kukkonen K, Savilahti E, Haahtela T, et al. Probiotics and prebiotic galacto-oligosaccharides in the prevention of allergic diseases: a randomized, double-blind, placebo-controlled trial. J Allergy Clin Immunol 2007; 119:192–198.
- Osborn DA, Sinn JK. Probiotics in infants for prevention of allergic disease and food hypersensitivity. Cochrane Database Syst Rev 2007;CD006475.
- Prescott SL, Bjorksten B. Probiotics for the prevention or treatment of allergic diseases. J Allergy Clin Immunol 2007; 120:255–262.
- Untersmayr E, Jensen-Jarolim E. The role of protein digestibility and antacids on food allergy outcomes. J Allergy Clin Immunol 2008; 121:1301–1308.
- Untersmayr E, Scholl I, Swoboda I, et al. Antacid medication inhibits digestion of dietary proteins and causes food allergy: a fish allergy model in BALB/c mice. J Allergy Clin Immunol 2003; 112:616–623.
- Sicherer SH, Furlong TJ, Maes HH, Desnick RJ, Sampson HA, Gelb BD. Genetics of peanut allergy: a twin study. J Allergy Clin Immunol 2000; 106:53–56.
- Sicherer SH, Sampson HA. Food allergy: recent advances in pathophysiology and treatment. Annu Rev Med 2009; 60:261–277.
- Sampson HA, Anderson JA. Summary and recommendations: classification of gastrointestinal manifestations due to immunologic reactions to foods in infants and young children. J Pediatr Gastroenterol Nutr 2000; 30( suppl 1):S87–S94.
- Sampson HA, Sicherer SH, Birnbaum AH. AGA technical review on the evaluation of food allergy in gastrointestinal disorders. American Gastroenterological Association. Gastroenterology 2001; 120:1026–1040.
- Spergel JM, Pawlowski NA. Food allergy. Mechanisms, diagnosis, and management in children. Pediatr Clin North Am 2002; 49:73–96.
- Sampson HA. Utility of food-specific IgE concentrations in predicting symptomatic food allergy. J Allergy Clin Immunol 2001; 107:891–896.
- Ortolani C, Ispano M, Pastorello EA, Ansaloni R, Magri GC. Comparison of results of skin prick tests (with fresh foods and commercial food extracts) and RAST in 100 patients with oral allergy syndrome. J Allergy Clin Immunol 1989; 83:683–690.
- Perry TT, Matsui EC, Kay Conover-Walker M, Wood RA. The relationship of allergen-specific IgE levels and oral food challenge outcome. J Allergy Clin Immunol 2004; 114:144–149.
- Leung DY, Sampson HA, Yunginger JW, et al; Avon Longitudinal Study of Parents and Children Study Team. Effect of anti-IgE therapy in patients with peanut allergy. N Engl J Med 2003; 348:986–993.
- Sampson HA. A phase II, randomized double-blind, parallel-group, placebo-controlled, oral food challenge trial of Xolair (omalizumab) in peanut allergy (TOPS). J Allergy Clin Immunol 2007; 119 (suppl 1):S117.
- Buchanan AD, Green TD, Jones SM, et al Egg oral immunotherapy in nonanaphylactic children with egg allergy. J Allergy Clin Immunol 2007; 119:199–205.
- Burks AW, Jones SM. Egg oral immunotherapy in non-anaphylactic children with egg allergy: follow-up. J Allergy Clin Immunol 2008; 121:270–271.
- Skripak JM, Nash SD, Rowley H, et al. A randomized, double-blind, placebo-controlled study of milk oral immunotherapy for cow's milk allergy. J Allergy Clin Immunol 2008; 122:1154–1160.
- Furuta GT, Liacouras CA, Collins MH, et al; First International Gastrointestinal Eosinophil Research Symposium (FIGERS) Subcommittees. Eosinophilic esophagitis in children and adults: a systematic review and consensus recommendations for diagnosis and treatment. Gastroenterology 2007; 133:1342–1363.
- Rodrigo S, Abboud G, Oh D, et al. High intraepithelial eosinophil counts in esophageal squamous epithelium are not specific for eosinophilic esophagitis in adults. Am J Gastroenterol 2008; 103:435–442.
- Liacouras CA, Spergel JM, Ruchelli E, et al. Eosinophilic esophagitis: a 10-year experience in 381 children. Clin Gastroenterol Hepatol 2005; 3:1198–1206.
- Simon D, Marti H, Heer P, Simon HU, Braathen LR, Straumann A. Eosinophilic esophagitis is frequently associated with IgE-mediated allergic airway diseases. J Allergy Clin Immunol 2005; 115:1090–1092.
- Rothenberg ME, Mishra A, Collins MH, Putnam PE. Pathogenesis and clinical features of eosinophilic esophagitis. J Allergy Clin Immunol 2001; 108:891–894.
- Gupta SK, Fitzgerald JF, Kondratyuk T, HogenEsch H. Cytokine expression in normal and inflamed esophageal mucosa: a study into the pathogenesis of allergic eosinophilic esophagitis. J Pediatr Gastroenterol Nutr 2006; 42:22–26.
- Straumann A, Bauer M, Fischer B, Blaser K, Simon HU. Idiopathic eosinophilic esophagitis is associated with a T(H)2-type allergic inflammatory response. J Allergy Clin Immunol 2001; 108:954–961.
- Mishra A, Rothenberg ME. Intratracheal IL-13 induces eosinophilic esophagitis by an IL-5, eotaxin-1, and STAT6-dependent mechanism. Gastroenterology 2003; 125:1419–1427.
- Akei HS, Mishra A, Blanchard C, Rothenberg ME. Epicutaneous antigen exposure primes for experimental eosinophilic esophagitis in mice. Gastroenterology 2005; 129:985–994.
- Blanchard C, Wang N, Stringer KF, et al. Eotaxin-3 and a uniquely conserved gene-expression profile in eosinophilic esophagitis. J Clin Invest 2006; 116:536–547.
- Fogg MI, Ruchelli E, Spergel JM. Pollen and eosinophilic esophagitis. J Allergy Clin Immunol 2003; 112:796–797.
- Almansa C, Krishna M, Buchner AM, et al. Seasonal distribution in newly diagnosed cases of eosinophilic esophagitis in adults. Am J Gastroenterol 2009; 104:828–833.
- Kelly KJ, Lazenby AJ, Rowe PC, Yardley JH, Perman JA, Sampson HA. Eosinophilic esophagitis attributed to gastroesophageal reflux: improvement with an amino acid-based formula. Gastroenterology 1995; 109:1503–1512.
- Markowitz JE, Spergel JM, Ruchelli E, Liacouras CA. Elemental diet is an effective treatment for eosinophilic esophagitis in children and adolescents. Am J Gastroenterol 2003; 98:777–782.
- Kagalwalla AF, Sentongo TA, Ritz S, et al. Effect of six-food elimination diet on clinical and histologic outcomes in eosinophilic esophagitis. Clin Gastroenterol Hepatol 2006; 4:1097–1102.
- Gonsalves N, Yang GY, Doerfler B, et al. A prospective clinical trial of six food elimination diet and reintroduction of causative agents in adults with eosinophilic esophagitis [abstract]. Gastroenterology 2008; 134( suppl 1):A104–A105.
- Simon D, Straumann A, Wenk A, Spichtin H, Simon HU, Braathen LR. Eosinophilic esophagitis in adults—no clinical relevance of wheat and rye sensitizations. Allergy 2006; 61:1480–1483.
- Antón Remirez J, Escudero R, Caceres O, Fernandez-Benitez M. Eosinophilic esophagitis. Allergol Immunopathol (Madr) 2006; 34:79–81.
- Yamazaki K, Murray JA, Arora AS, et al. Allergen-specific in vitro cytokine production in adult patients with eosinophilic esophagitis. Dig Dis Sci 2006; 51:1934–1941.
- Penfield JD, Lang DM, Goldblum JR, Lopez R, Falk GW. The role of allergy evaluation in adults with eosinophilic esophagitis. J Clin Gastroenterol 2009(Epub ahead of print)
- Spergel JM, Andrews T, Brown-Whitehorn TF, Beausoleil JL, Liacouras CA. Treatment of eosinophilic esophagitis with specific food elimination diet directed by a combination of skin prick and patch tests. Ann Allergy Asthma Immunol 2005; 95:336–343.
- Spergel JM, Brown-Whitehorn T, Beausoleil JL, Shuker M, Liacouras CA. Predictive values for skin prick test and atopy patch test for eosinophilic esophagitis. J Allergy Clin Immunol 2007; 119:509–511.
More children and even adults seem to be allergic to various foods these days than in the past. Also apparently on the rise is a linked condition, eosinophilic esophagitis.
The reason for these increases is not clear. This article confines itself to what we know about the mechanisms of food allergies and eosinophilic esophagitis, how to diagnose them, and how to treat them.
FOOD ALLERGIES ARE COMMON, AND MORE PREVALENT THAN EVER
Food allergies—abnormal immune responses to food proteins1—affect an estimated 6% to 8% of young children and 3% to 4% of adults in the United States,2,3 and their prevalence appears to be rising in developed countries. Studies in US and British children indicate that peanut allergy has doubled in the past decade. 4
Any food can provoke a reaction, but only a few foods account for most of the significant allergic reactions: cow’s milk, soy, wheat, eggs, peanuts, tree nuts, fish, and shellfish.
Approximately 80% of allergies to milk, egg, wheat, and soy resolve by the time the patient reaches early adolescence.6 Fewer cases resolve in children with tree nut allergies (approximately 9%) or peanut allergy (20%),7,8 and allergies to fish and shellfish often develop or persist in adulthood.
A family history of an atopic disease such as asthma, allergic rhinitis, eczema, or food allergy is a risk factor for developing a food allergy. 3 Considering that the rate of peanut allergy has doubled in children over the past 10 years, environmental factors may also play a role.3
How we tolerate foods or become allergic to them
The gut, the largest mucosal organ in the body, is exposed to large quantities of foreign proteins daily. Most protein is broken down by stomach acid and digestive enzymes into lessantigenic peptides or is bound by secretory immunoglobulin A (IgA), which prevents it from being absorbed. Further, the epithelial cells lining the gut do not allow large molecules to pass easily, having tight intracellular junctions and being covered with mucus.
For these reasons, less than 2% of the protein in food is absorbed in an allergenic form.9 The reason food allergies are more prevalent in children is most likely that children have an immature gut barrier, lower IgA levels, a higher gastric pH, and lower proteolytic enzyme levels.
When dietary proteins do cross the gut barrier, the immune system normally suppresses the allergic response. Regulatory T cells, dendritic cells, and local immune responses play critical roles in the development of tolerance. Several types of regulatory T cells, such as Tr1 cells (which secrete interleukin 10), TH3 cells (which secrete transforming growth factor beta), CD4+CD25+ regulatory T cells, gamma-delta T cells, and CD8+ suppressor cells can all contribute to suppressing allergic responses.10 Dendritic cells also help induce tolerance by stimulating CD4+ T cells to secrete transforming growth factor beta, which leads to the production of interleukin 10 and additional transforming growth factor beta.11
Factors that contribute to food allergy
Many factors may contribute to whether a person becomes tolerant to or sensitized to a specific food protein.
The dose of antigen. Tolerance can develop after either high or low doses of antigens, but by different mechanisms.
The antigen structure. Soluble antigens are less sensitizing than particulate antigens.12,13
Processing of foods. Dry-roasted peanuts are more allergenic than raw or boiled peanuts, partly because they are less soluble.13
The route of initial exposure. Sensitization to food proteins can occur directly through the gut or the skin. Alternatively, it can occur indirectly via the respiratory tract. Skin exposure may be especially sensitizing in children with atopic dermatitis.14,15
The gut flora. When mice are raised in a germ-free environment, they fail to develop normal tolerance.16 They are also more likely to become sensitized if they are treated with antibiotics or if they lack toll-like receptors that recognize bacterial lipopolysaccharides.17 Furthermore, human studies suggest that probiotics promote tolerance, especially in preventing atopic dermatitis, although the studies have had conflicting results.18–21
The gastric pH. Murine and human studies reveal that antacid medications increase the risk of food allergy.22,23
Genetic susceptibility. A child with a sibling who is allergic to peanuts is approximately 10 times more likely to be allergic to peanuts than predicted by the rate in the general population. Although no risk-conferring gene has been identified, a study of twins showed concordance for peanut allergy in 64.3% of identical twins vs 6.8% of fraternal twins.24
Three types of immune responses to food
Immunologic reactions to foods can be divided into three categories: mediated by immunoglobulin E (IgE), non-IgE-mediated, and mixed. Therefore, these disorders can present as an acute, potentially life-threatening reaction or as a chronic disease such as eosinophilic gastoenteropathy.
IgE-mediated reactions are immediate hypersensitivity responses. In most patients, an IgE-mediated mechanism can be confirmed by a positive skin test or a test for food-specific IgE in the serum. In this article, the term “food allergy” refers to an IgE-mediated reaction to a food, unless otherwise indicated.
Non-IgE-mediated reactions have a delayed onset and chronic symptoms. Commonly, they are confined to the gastrointestinal tract; examples are food-protein-induced enterocolitis, proctitis, and proctocolitis and celiac disease.3,26,27 However, other diseases such as contact dermatitis, dermatitis herpetiformis, and food-induced pulmonary hemosiderosis (Heiner syndrome) are also considered non-IgE-mediated allergies.
Mixed-reaction disorders are chronic and include the eosinophilic gastroenteropathies, ie, eosinophilic proctocolitis, eosinophilic gastroenteritis, and eosinophilic esophagitis.28 The pathophysiology of these diseases is poorly understood. Many patients have evidence of allergic sensitivities to food or to environmental allergens, or both, but whether these sensitivities have a causal role in these disorders is not clear.
Atopic dermatitis, another complicated disease process, may be associated with mixedreaction food allergy, as approximately 35% of young children with moderate to severe atopic dermatitis have food allergies.29
Diagnosis of IgE-mediated food allergies
A thorough history and physical examination are key to diagnosing an IgE-mediated food allergy.
The history should include potential culprit foods, the quantity eaten, the timing of the onset of symptoms, and related factors such as exercise, alcohol intake, or medication use. Symptoms of an IgE-mediated reaction are generally rapid in onset but may be delayed up to a few hours, while non-IgE mediated symptoms may present several hours to days later.
Food challenge. A double-blind, placebocontrolled oral food challenge is the gold standard for the diagnosis of food allergies. (The food to be tested is hidden in other food or in capsules.) However, this test poses significant risks, and other diagnostic methods are more practical for screening.
Skin-prick tests with commercially available extracts are a rapid and sensitive method of screening for allergy to several foods.
Negative skin-prick tests have an estimated negative predictive value of more than 95% and can therefore exclude IgE-mediated food allergies.
A positive test indicates the presence of IgE against a specific food allergen and suggests a clinical food allergy, although the specificity of the test is only about 50%, making a positive result difficult to interpret. Although the size of the skin-test response does not necessarily correlate with the potential severity of a reaction, a response larger than 3 mm does indicate a greater likelihood of clinical reactivity. A positive test is most helpful in confirming the diagnosis of IgE-mediated food allergy when combined with a clear history of food-induced symptoms.
The proteins in commercially based extracts of most fruits and vegetables are often labile; therefore, skin testing with fresh fruits and vegetables may be indicated.30
Immunoassays. Radioallergosorbent tests (RASTs) and fluorescent enzyme immunoassays are used to identity food-specific IgE antibodies in the serum. The commercially available tests do not use radioactivity, but the term “RAST” is still commonly used.
Immunoassays are generally less sensitive and more costly than skin-prick tests, and their results are not immediately available, unlike those of skin-prick testing. However, these in vitro tests are not affected by antihistamine use and are useful in patients with severe dermatologic conditions or severe anaphylaxis, for whom skin-prick testing would not be appropriate.
However, unlike a negative skin-prick test, an undetectable serum food-specific IgE level has a low negative predictive value, and an undetectable level may be associated with symptoms of an allergic reaction for 10% to 25% of patients.29 Therefore, if one suspects an allergic reaction but no food-specific IgE can be detected in the serum, confirming the absence of a clinical allergy must be done with a skin-prick test or with a physician-supervised oral challenge, or both.
Managing food allergy by avoiding the allergen
Food allergies are managed by strictly avoiding food allergens and by taking medications such as self-injectable epinephrine for anaphylactic symptoms.
Patients and caregivers must be educated about reading food labels, avoiding high-risk situations such as eating at buffets and other restaurants with high risk of cross-contamination, wearing a medical-alert bracelet, recognizing and managing early symptoms of an allergic reaction, and calling for emergency services if they are having an allergic reaction. Since January 2006, the US Food and Drug Administration has required food manufacturers to list common food allergens on food labels (cow’s milk, soy, wheat, egg, peanut, tree nuts, fish, and shellfish), and the labeling must use simple, easily understood terms, such as “milk” instead of “whey.” However, it is still prudent to read all ingredients listed on the label.
Experimental treatments for food allergies
Humanized monoclonal anti-IgE antibodies such as talizumab (also known as TNX-901) and omalizumab (Xolair) have been developed, but their use in food allergy has been limited. In a study in patients with peanut allergy, injections of talizumab increased the threshold for sensitivity to peanuts in most patients, but 25% of the patients did not have any improvement.32 A study of omalizumab in patients with peanut allergy was stopped after adverse reactions developed during oral peanut challenges.33
Oral immunotherapy. Recent studies suggest it may be possible to induce oral tolerance in patients with IgE-mediated food allergy. Pilot studies have shown that frequent, increasing doses of food allergens (egg, milk, and peanut) may raise the threshold at which symptoms occur.34–36 Though these studies suggest that oral immunotherapy may protect some patients against a reaction if they accidentally ingest a food they are allergic to, some patients could not reach the goal doses because allergic symptoms were provoked.
At this early stage, these strategies must be considered investigational, and more randomized, placebo-controlled studies are needed. Further studies will also be needed to assess whether oral immunotherapy induces only short-term desensitization (in which case the allergen needs to be ingested daily to prevent reactions) or sustained tolerance (in which case the antigenic protein can be ingested without symptoms despite periods of abstinence).
THE ROLE OF FOOD ALLERGY IN EOSINOPHILIC ESOPHAGITIS
Eosinophilic esophagitis has been recognized with increasing frequency in both children and adults over the past several years. Symptoms can include difficulty feeding, failure to thrive, vomiting, epigastric or chest pain, dysphagia, and food impaction.
Diagnostic criteria for eosinophilic esophagitis are37:
- Clinical symptoms of esophageal dysfunction
- At least 15 eosinophils per high-power field in at least one esophageal biopsy specimen
- No response to a proton-pump inhibitor in high doses (up to 2 mg/kg/day) for 1 to 2 months, or normal results on pH probe monitoring of the esophagus (the reason for this criterion is that patients with gastroesophageal reflux disease can also have large numbers of eosinophils in the esophagus—more than 100 per highpower field38)
- Exclusion of other causes.
Though the cause of eosinophilic esophagitis is not completely understood, atopy has been strongly implicated as a factor. More than 50% of patients with eosinophilic esophagitis also have an atopic condition (eg, atopic dermatitis, allergic rhinitis, asthma), as well as positive results on skin-prick testing or measurement of antigen-specific IgE in the serum.39–41 Also, since most patients improve with either dietary restriction or elemental diets, food sensitization appears to play a considerable role.
As with atopic conditions such as asthma, atopic dermatitis, allergic rhinitis, and food allergy, eosinophilic esophagitis has been linked with immune responses involving helper T cell 2 (TH2). Adults and children with eosinophilic esophagitis have been found to have elevated eosinophil counts and total IgE levels in peripheral blood.37 In the esophagus, patients have elevated levels of the TH2 cytokines often seen in atopic patients (eg, interleukins 4, 5, and 13) and mast cells.42,43 In mice, eosinophilic esophagitis can be induced by allergen exposure and overexpression of TH2 cytokines.44,45 Expression of eotaxin-3, a potent eosinophil chemoattractant, was noted to be higher in children with eosinophilic esophagitis than in controls.46
Of interest, some patients with eosinophilic esophagitis say their symptoms vary with the seasons, correlating with seasonal changes in esophageal eosinophil levels.47,48
Studies linking eosinophilic esophagitis and food allergy in children
Kelly et al49 reported that 10 children with chronic symptomatic gastroesophageal reflux and eosinophilic esophagitis all had partial or complete resolution of symptoms on an elemental diet.
Markowitz et al50 found that symptoms of chronic reflux disease and eosinophilic esophagitis improved in 49 of 51 children on an elemental diet, and the number of eosinophils in the distal esophagus decreased significantly.
Liacouras et al39 reported similar findings in a 10-year experience. Of 132 children who had eosinophilic esophagitis, 75 improved with dietary restriction based on results of skin-prick and patch testing. The 57 patients who did not respond and 115 others were started on an elemental diet. Of the 164 patients who complied with the elemental diet, 160 had significant improvement of symptoms and a significant decrease in the number of eosinophils in the esophagus. Individual foods were reintroduced approximately every 5 days, and esophagogastroduodenoscopy with biopsies was performed 4 to 8 weeks after the last was reintroduced into the diet.
In a retrospective study, Kagalwalla et al51 reported that 60 children with eosinophilic esophagitis were treated with either an elemental diet or a six-food elimination diet (no milk, soy, wheat, egg, peanut, or seafood). The two groups showed similar clinical and histologic improvements.
Collectively, these studies in pediatric patients imply that food allergy is a significant factor in the pathogenesis of eosinophilic esophagitis.
Studies in adults
Fewer studies of the link between food allergy and eosinophilic esophagitis have been done in adults.
In a preliminary study, 18 adults followed the six-food elimination diet. Symptoms improved in 17 (94%), and histologic findings improved in 14 (78%).52
On the other hand, in six adult patients with eosinophilic esophagitis, Simon et al53 found that only one had improvement in symptoms after eliminating wheat and rye from the diet, and none had significant changes in the number of eosinophils in the esophagus.
In a 37-year-old man with eosinophilic esophagitis, symptoms improved after eliminating egg from his diet.54
Yamazaki et al55 measured expression of interleukin 5 and interleukin 13 in 15 adult patients with eosinophilic esophagitis. Food and aeroallergens that included milk, soy, dust mite, ragweed, and Aspergillus induced significantly more interleukin 5 production in these patients than in atopic controls, suggesting that both foods and aeroallergens may have a role in the pathogenesis of eosinophilic esophagitis in adults.
How to identify potential food triggers of eosinophilic esophagitis
Though elemental diets have been associated with a decrease in symptoms and esophageal eosinophilia, elemental formulas are expensive and unpalatable and pose a risk of nutritional deprivation. Identifying specific food allergens to eliminate from the diet in patients with eosinophilic esophagitis may be less expensive and more desirable than a very limited or elemental diet.
However, potential food triggers have been hard to identify in eosinophilic esophagitis. A recent consensus report did not recommend in vitro food allergy testing,37 owing to a lack of positive or negative predictive values for food-specific IgE level testing in eosinophilic esophagitis. Furthermore, the absence of IgE does not eliminate a food as a potential trigger, since non-IgE mechanisms may play a role.
Skin-prick testing is one of the currently validated diagnostic methods. Several studies have used skin-prick testing of foods in patients with eosinophilic esophagitis. In these studies, approximately two-thirds of patients had positive test reactions to at least one food, most often to common food allergens such as cow’s milk, egg, soy, wheat, and peanut, but also to rye, beef, and bean.37 In a recent article,56 81% of adult patients with eosinophilic esophagitis had one or more allergens identified by skin-prick testing, and 50% of the patients tested positive for one or more food allergens.
Atopy patch testing. The combination of skin-prick testing and atopy patch testing may be more effective than skin-prick testing alone in identifying potential food triggers. Atopy patch testing has been used in the diagnosis of non-IgE cell-mediated (delayed) immune responses, in which T cells may play a significant role.
Atopy patch testing is similar to patch testing for contact dermatitis. It involves placing a small quantity of food on the skin and evaluating for a local delayed reaction after a set time.
In two studies,50,57 146 children with biopsy-proven eosinophilic esophagitis had foods eliminated from the diet on the basis of positive skin-prick tests and atopy patch tests. Approximately 77% of the children had significant reduction of esophageal eosinophils in biopsy specimens (from 20 per high-power field to 1.1). The foods most commonly implicated by skin-prick testing were cow’s milk, egg, wheat, peanut, shellfish, peas, beef, fish, rye, and tomato; those identified by atopy patch testing were cow’s milk, egg, wheat, corn, beef, milk, soy, rye, chicken, oats, and potato. The combination of both types of testing had a negative predictive value of 88% to 100% for all foods except milk, while the positive predictive value was greater than 74% for the most common foods causing eosinophilic esophagitis.58
Though atopy patch testing shows some usefulness in identifying foods that may elicit non-IgE-mediated reactions, currently these tests are not validated and have been evaluated in only a small number of studies. Currently, no standardized testing materials, methods of application, or interpretation of results exist, and no studies have included a control population to validate atopy patch testing. More studies are needed to validate atopy patch testing as a reliable diagnostic tool before it can be recommended as a component of routine diagnostic evaluation in patients with eosinophilic esophagitis.
More children and even adults seem to be allergic to various foods these days than in the past. Also apparently on the rise is a linked condition, eosinophilic esophagitis.
The reason for these increases is not clear. This article confines itself to what we know about the mechanisms of food allergies and eosinophilic esophagitis, how to diagnose them, and how to treat them.
FOOD ALLERGIES ARE COMMON, AND MORE PREVALENT THAN EVER
Food allergies—abnormal immune responses to food proteins1—affect an estimated 6% to 8% of young children and 3% to 4% of adults in the United States,2,3 and their prevalence appears to be rising in developed countries. Studies in US and British children indicate that peanut allergy has doubled in the past decade. 4
Any food can provoke a reaction, but only a few foods account for most of the significant allergic reactions: cow’s milk, soy, wheat, eggs, peanuts, tree nuts, fish, and shellfish.
Approximately 80% of allergies to milk, egg, wheat, and soy resolve by the time the patient reaches early adolescence.6 Fewer cases resolve in children with tree nut allergies (approximately 9%) or peanut allergy (20%),7,8 and allergies to fish and shellfish often develop or persist in adulthood.
A family history of an atopic disease such as asthma, allergic rhinitis, eczema, or food allergy is a risk factor for developing a food allergy. 3 Considering that the rate of peanut allergy has doubled in children over the past 10 years, environmental factors may also play a role.3
How we tolerate foods or become allergic to them
The gut, the largest mucosal organ in the body, is exposed to large quantities of foreign proteins daily. Most protein is broken down by stomach acid and digestive enzymes into lessantigenic peptides or is bound by secretory immunoglobulin A (IgA), which prevents it from being absorbed. Further, the epithelial cells lining the gut do not allow large molecules to pass easily, having tight intracellular junctions and being covered with mucus.
For these reasons, less than 2% of the protein in food is absorbed in an allergenic form.9 The reason food allergies are more prevalent in children is most likely that children have an immature gut barrier, lower IgA levels, a higher gastric pH, and lower proteolytic enzyme levels.
When dietary proteins do cross the gut barrier, the immune system normally suppresses the allergic response. Regulatory T cells, dendritic cells, and local immune responses play critical roles in the development of tolerance. Several types of regulatory T cells, such as Tr1 cells (which secrete interleukin 10), TH3 cells (which secrete transforming growth factor beta), CD4+CD25+ regulatory T cells, gamma-delta T cells, and CD8+ suppressor cells can all contribute to suppressing allergic responses.10 Dendritic cells also help induce tolerance by stimulating CD4+ T cells to secrete transforming growth factor beta, which leads to the production of interleukin 10 and additional transforming growth factor beta.11
Factors that contribute to food allergy
Many factors may contribute to whether a person becomes tolerant to or sensitized to a specific food protein.
The dose of antigen. Tolerance can develop after either high or low doses of antigens, but by different mechanisms.
The antigen structure. Soluble antigens are less sensitizing than particulate antigens.12,13
Processing of foods. Dry-roasted peanuts are more allergenic than raw or boiled peanuts, partly because they are less soluble.13
The route of initial exposure. Sensitization to food proteins can occur directly through the gut or the skin. Alternatively, it can occur indirectly via the respiratory tract. Skin exposure may be especially sensitizing in children with atopic dermatitis.14,15
The gut flora. When mice are raised in a germ-free environment, they fail to develop normal tolerance.16 They are also more likely to become sensitized if they are treated with antibiotics or if they lack toll-like receptors that recognize bacterial lipopolysaccharides.17 Furthermore, human studies suggest that probiotics promote tolerance, especially in preventing atopic dermatitis, although the studies have had conflicting results.18–21
The gastric pH. Murine and human studies reveal that antacid medications increase the risk of food allergy.22,23
Genetic susceptibility. A child with a sibling who is allergic to peanuts is approximately 10 times more likely to be allergic to peanuts than predicted by the rate in the general population. Although no risk-conferring gene has been identified, a study of twins showed concordance for peanut allergy in 64.3% of identical twins vs 6.8% of fraternal twins.24
Three types of immune responses to food
Immunologic reactions to foods can be divided into three categories: mediated by immunoglobulin E (IgE), non-IgE-mediated, and mixed. Therefore, these disorders can present as an acute, potentially life-threatening reaction or as a chronic disease such as eosinophilic gastoenteropathy.
IgE-mediated reactions are immediate hypersensitivity responses. In most patients, an IgE-mediated mechanism can be confirmed by a positive skin test or a test for food-specific IgE in the serum. In this article, the term “food allergy” refers to an IgE-mediated reaction to a food, unless otherwise indicated.
Non-IgE-mediated reactions have a delayed onset and chronic symptoms. Commonly, they are confined to the gastrointestinal tract; examples are food-protein-induced enterocolitis, proctitis, and proctocolitis and celiac disease.3,26,27 However, other diseases such as contact dermatitis, dermatitis herpetiformis, and food-induced pulmonary hemosiderosis (Heiner syndrome) are also considered non-IgE-mediated allergies.
Mixed-reaction disorders are chronic and include the eosinophilic gastroenteropathies, ie, eosinophilic proctocolitis, eosinophilic gastroenteritis, and eosinophilic esophagitis.28 The pathophysiology of these diseases is poorly understood. Many patients have evidence of allergic sensitivities to food or to environmental allergens, or both, but whether these sensitivities have a causal role in these disorders is not clear.
Atopic dermatitis, another complicated disease process, may be associated with mixedreaction food allergy, as approximately 35% of young children with moderate to severe atopic dermatitis have food allergies.29
Diagnosis of IgE-mediated food allergies
A thorough history and physical examination are key to diagnosing an IgE-mediated food allergy.
The history should include potential culprit foods, the quantity eaten, the timing of the onset of symptoms, and related factors such as exercise, alcohol intake, or medication use. Symptoms of an IgE-mediated reaction are generally rapid in onset but may be delayed up to a few hours, while non-IgE mediated symptoms may present several hours to days later.
Food challenge. A double-blind, placebocontrolled oral food challenge is the gold standard for the diagnosis of food allergies. (The food to be tested is hidden in other food or in capsules.) However, this test poses significant risks, and other diagnostic methods are more practical for screening.
Skin-prick tests with commercially available extracts are a rapid and sensitive method of screening for allergy to several foods.
Negative skin-prick tests have an estimated negative predictive value of more than 95% and can therefore exclude IgE-mediated food allergies.
A positive test indicates the presence of IgE against a specific food allergen and suggests a clinical food allergy, although the specificity of the test is only about 50%, making a positive result difficult to interpret. Although the size of the skin-test response does not necessarily correlate with the potential severity of a reaction, a response larger than 3 mm does indicate a greater likelihood of clinical reactivity. A positive test is most helpful in confirming the diagnosis of IgE-mediated food allergy when combined with a clear history of food-induced symptoms.
The proteins in commercially based extracts of most fruits and vegetables are often labile; therefore, skin testing with fresh fruits and vegetables may be indicated.30
Immunoassays. Radioallergosorbent tests (RASTs) and fluorescent enzyme immunoassays are used to identity food-specific IgE antibodies in the serum. The commercially available tests do not use radioactivity, but the term “RAST” is still commonly used.
Immunoassays are generally less sensitive and more costly than skin-prick tests, and their results are not immediately available, unlike those of skin-prick testing. However, these in vitro tests are not affected by antihistamine use and are useful in patients with severe dermatologic conditions or severe anaphylaxis, for whom skin-prick testing would not be appropriate.
However, unlike a negative skin-prick test, an undetectable serum food-specific IgE level has a low negative predictive value, and an undetectable level may be associated with symptoms of an allergic reaction for 10% to 25% of patients.29 Therefore, if one suspects an allergic reaction but no food-specific IgE can be detected in the serum, confirming the absence of a clinical allergy must be done with a skin-prick test or with a physician-supervised oral challenge, or both.
Managing food allergy by avoiding the allergen
Food allergies are managed by strictly avoiding food allergens and by taking medications such as self-injectable epinephrine for anaphylactic symptoms.
Patients and caregivers must be educated about reading food labels, avoiding high-risk situations such as eating at buffets and other restaurants with high risk of cross-contamination, wearing a medical-alert bracelet, recognizing and managing early symptoms of an allergic reaction, and calling for emergency services if they are having an allergic reaction. Since January 2006, the US Food and Drug Administration has required food manufacturers to list common food allergens on food labels (cow’s milk, soy, wheat, egg, peanut, tree nuts, fish, and shellfish), and the labeling must use simple, easily understood terms, such as “milk” instead of “whey.” However, it is still prudent to read all ingredients listed on the label.
Experimental treatments for food allergies
Humanized monoclonal anti-IgE antibodies such as talizumab (also known as TNX-901) and omalizumab (Xolair) have been developed, but their use in food allergy has been limited. In a study in patients with peanut allergy, injections of talizumab increased the threshold for sensitivity to peanuts in most patients, but 25% of the patients did not have any improvement.32 A study of omalizumab in patients with peanut allergy was stopped after adverse reactions developed during oral peanut challenges.33
Oral immunotherapy. Recent studies suggest it may be possible to induce oral tolerance in patients with IgE-mediated food allergy. Pilot studies have shown that frequent, increasing doses of food allergens (egg, milk, and peanut) may raise the threshold at which symptoms occur.34–36 Though these studies suggest that oral immunotherapy may protect some patients against a reaction if they accidentally ingest a food they are allergic to, some patients could not reach the goal doses because allergic symptoms were provoked.
At this early stage, these strategies must be considered investigational, and more randomized, placebo-controlled studies are needed. Further studies will also be needed to assess whether oral immunotherapy induces only short-term desensitization (in which case the allergen needs to be ingested daily to prevent reactions) or sustained tolerance (in which case the antigenic protein can be ingested without symptoms despite periods of abstinence).
THE ROLE OF FOOD ALLERGY IN EOSINOPHILIC ESOPHAGITIS
Eosinophilic esophagitis has been recognized with increasing frequency in both children and adults over the past several years. Symptoms can include difficulty feeding, failure to thrive, vomiting, epigastric or chest pain, dysphagia, and food impaction.
Diagnostic criteria for eosinophilic esophagitis are37:
- Clinical symptoms of esophageal dysfunction
- At least 15 eosinophils per high-power field in at least one esophageal biopsy specimen
- No response to a proton-pump inhibitor in high doses (up to 2 mg/kg/day) for 1 to 2 months, or normal results on pH probe monitoring of the esophagus (the reason for this criterion is that patients with gastroesophageal reflux disease can also have large numbers of eosinophils in the esophagus—more than 100 per highpower field38)
- Exclusion of other causes.
Though the cause of eosinophilic esophagitis is not completely understood, atopy has been strongly implicated as a factor. More than 50% of patients with eosinophilic esophagitis also have an atopic condition (eg, atopic dermatitis, allergic rhinitis, asthma), as well as positive results on skin-prick testing or measurement of antigen-specific IgE in the serum.39–41 Also, since most patients improve with either dietary restriction or elemental diets, food sensitization appears to play a considerable role.
As with atopic conditions such as asthma, atopic dermatitis, allergic rhinitis, and food allergy, eosinophilic esophagitis has been linked with immune responses involving helper T cell 2 (TH2). Adults and children with eosinophilic esophagitis have been found to have elevated eosinophil counts and total IgE levels in peripheral blood.37 In the esophagus, patients have elevated levels of the TH2 cytokines often seen in atopic patients (eg, interleukins 4, 5, and 13) and mast cells.42,43 In mice, eosinophilic esophagitis can be induced by allergen exposure and overexpression of TH2 cytokines.44,45 Expression of eotaxin-3, a potent eosinophil chemoattractant, was noted to be higher in children with eosinophilic esophagitis than in controls.46
Of interest, some patients with eosinophilic esophagitis say their symptoms vary with the seasons, correlating with seasonal changes in esophageal eosinophil levels.47,48
Studies linking eosinophilic esophagitis and food allergy in children
Kelly et al49 reported that 10 children with chronic symptomatic gastroesophageal reflux and eosinophilic esophagitis all had partial or complete resolution of symptoms on an elemental diet.
Markowitz et al50 found that symptoms of chronic reflux disease and eosinophilic esophagitis improved in 49 of 51 children on an elemental diet, and the number of eosinophils in the distal esophagus decreased significantly.
Liacouras et al39 reported similar findings in a 10-year experience. Of 132 children who had eosinophilic esophagitis, 75 improved with dietary restriction based on results of skin-prick and patch testing. The 57 patients who did not respond and 115 others were started on an elemental diet. Of the 164 patients who complied with the elemental diet, 160 had significant improvement of symptoms and a significant decrease in the number of eosinophils in the esophagus. Individual foods were reintroduced approximately every 5 days, and esophagogastroduodenoscopy with biopsies was performed 4 to 8 weeks after the last was reintroduced into the diet.
In a retrospective study, Kagalwalla et al51 reported that 60 children with eosinophilic esophagitis were treated with either an elemental diet or a six-food elimination diet (no milk, soy, wheat, egg, peanut, or seafood). The two groups showed similar clinical and histologic improvements.
Collectively, these studies in pediatric patients imply that food allergy is a significant factor in the pathogenesis of eosinophilic esophagitis.
Studies in adults
Fewer studies of the link between food allergy and eosinophilic esophagitis have been done in adults.
In a preliminary study, 18 adults followed the six-food elimination diet. Symptoms improved in 17 (94%), and histologic findings improved in 14 (78%).52
On the other hand, in six adult patients with eosinophilic esophagitis, Simon et al53 found that only one had improvement in symptoms after eliminating wheat and rye from the diet, and none had significant changes in the number of eosinophils in the esophagus.
In a 37-year-old man with eosinophilic esophagitis, symptoms improved after eliminating egg from his diet.54
Yamazaki et al55 measured expression of interleukin 5 and interleukin 13 in 15 adult patients with eosinophilic esophagitis. Food and aeroallergens that included milk, soy, dust mite, ragweed, and Aspergillus induced significantly more interleukin 5 production in these patients than in atopic controls, suggesting that both foods and aeroallergens may have a role in the pathogenesis of eosinophilic esophagitis in adults.
How to identify potential food triggers of eosinophilic esophagitis
Though elemental diets have been associated with a decrease in symptoms and esophageal eosinophilia, elemental formulas are expensive and unpalatable and pose a risk of nutritional deprivation. Identifying specific food allergens to eliminate from the diet in patients with eosinophilic esophagitis may be less expensive and more desirable than a very limited or elemental diet.
However, potential food triggers have been hard to identify in eosinophilic esophagitis. A recent consensus report did not recommend in vitro food allergy testing,37 owing to a lack of positive or negative predictive values for food-specific IgE level testing in eosinophilic esophagitis. Furthermore, the absence of IgE does not eliminate a food as a potential trigger, since non-IgE mechanisms may play a role.
Skin-prick testing is one of the currently validated diagnostic methods. Several studies have used skin-prick testing of foods in patients with eosinophilic esophagitis. In these studies, approximately two-thirds of patients had positive test reactions to at least one food, most often to common food allergens such as cow’s milk, egg, soy, wheat, and peanut, but also to rye, beef, and bean.37 In a recent article,56 81% of adult patients with eosinophilic esophagitis had one or more allergens identified by skin-prick testing, and 50% of the patients tested positive for one or more food allergens.
Atopy patch testing. The combination of skin-prick testing and atopy patch testing may be more effective than skin-prick testing alone in identifying potential food triggers. Atopy patch testing has been used in the diagnosis of non-IgE cell-mediated (delayed) immune responses, in which T cells may play a significant role.
Atopy patch testing is similar to patch testing for contact dermatitis. It involves placing a small quantity of food on the skin and evaluating for a local delayed reaction after a set time.
In two studies,50,57 146 children with biopsy-proven eosinophilic esophagitis had foods eliminated from the diet on the basis of positive skin-prick tests and atopy patch tests. Approximately 77% of the children had significant reduction of esophageal eosinophils in biopsy specimens (from 20 per high-power field to 1.1). The foods most commonly implicated by skin-prick testing were cow’s milk, egg, wheat, peanut, shellfish, peas, beef, fish, rye, and tomato; those identified by atopy patch testing were cow’s milk, egg, wheat, corn, beef, milk, soy, rye, chicken, oats, and potato. The combination of both types of testing had a negative predictive value of 88% to 100% for all foods except milk, while the positive predictive value was greater than 74% for the most common foods causing eosinophilic esophagitis.58
Though atopy patch testing shows some usefulness in identifying foods that may elicit non-IgE-mediated reactions, currently these tests are not validated and have been evaluated in only a small number of studies. Currently, no standardized testing materials, methods of application, or interpretation of results exist, and no studies have included a control population to validate atopy patch testing. More studies are needed to validate atopy patch testing as a reliable diagnostic tool before it can be recommended as a component of routine diagnostic evaluation in patients with eosinophilic esophagitis.
- Bruijnzeel-Koomen C, Ortolani C, Aas K, et al. Adverse reactions to food. European Academy of Allergology and Clinical Immunology Subcommittee. Allergy 1995; 50:623–635.
- Sampson HA. Update on food allergy. J Allergy Clin Immunol 2004; 113:805–819.
- Sicherer SH, Sampson HA. 9. Food allergy. J Allergy Clin Immunol 2006; 117 (suppl 2):S470–S475.
- Sicherer SH, Munoz-Furlong A, Sampson HA. Prevalence of peanut and tree nut allergy in the United States determined by means of a random digit dial telephone survey: a 5-year follow-up study. J Allergy Clin Immunol 2003; 112:1203–1207.
- American College of Allergy, Asthma, & Immunology. Food allergy: a practice parameter. Ann Allergy Asthma Immunol 2006; 96( suppl 2):S1–S68.
- Wood RA. The natural history of food allergy. Pediatrics 2003; 111:1631–1637.
- Hourihane JO, Roberts SA, Warner JO. Resolution of peanut allergy: case-control study. BMJ 1998; 316:1271–1275.
- Fleischer DM, Conover-Walker MK, Matsui EC, Wood RA. The natural history of tree nut allergy. J Allergy Clin Immunol 2005; 116:1087–1093.
- Husby S, Foged N, Host A, Svehag SE. Passage of dietary antigens into the blood of children with coeliac disease. Quantification and size distribution of absorbed antigens. Gut 1987; 28:1062–1072.
- Mowat AM. Anatomical basis of tolerance and immunity to intestinal antigens. Nat Rev Immunol 2003; 3:331–341.
- Frossard CP, Tropia L, Hauser C, Eigenmann PA. Lymphocytes in Peyer patches regulate clinical tolerance in a murine model of food allergy. J Allergy Clin Immunol 2004; 113:958–964.
- Jain SL, Barone KS, Flanagan MP, Michael JG. Activation patterns of murine B cells after oral administration of an encapsulated soluble antigen. Vaccine 1996; 14:1291–1297.
- Kopper RA, Odum NJ, Sen M, Helm RM, Stanley JS, Burks AW. Peanut protein allergens: the effect of roasting on solubility and allergenicity. Int Arch Allergy Immunol 2005; 136:16–22.
- Lack G. Epidemiologic risks for food allergy. J Allergy Clin Immunol 2008; 121:1331–1336.
- Lack G, Fox D, Northstone K, Golding J; Avon Longitudinal Study of Parents and Children Study Team. Factors associated with the development of peanut allergy in childhood. N Engl J Med 2003; 348:977–985.
- Sudo N, Sawamura S, Tanaka K, Aiba Y, Kubo C, Koga Y. The requirement of intestinal bacterial flora for the development of an IgE production system fully susceptible to oral tolerance induction. J Immunol 1997; 159:1739–1745.
- Bashir ME, Louie S, Shi HN, Nagler-Anderson C. Toll-like receptor 4 signaling by intestinal microbes influences susceptibility to food allergy. J Immunol 2004; 172:6978–6987.
- Kopp MV, Hennemuth I, Heinzmann A, Urbanek R. Randomized, double-blind, placebo-controlled trial of probiotics for primary prevention: no clinical effects of lactobacillus GG supplementation. Pediatrics 2008; 121:e850–e856.
- Kukkonen K, Savilahti E, Haahtela T, et al. Probiotics and prebiotic galacto-oligosaccharides in the prevention of allergic diseases: a randomized, double-blind, placebo-controlled trial. J Allergy Clin Immunol 2007; 119:192–198.
- Osborn DA, Sinn JK. Probiotics in infants for prevention of allergic disease and food hypersensitivity. Cochrane Database Syst Rev 2007;CD006475.
- Prescott SL, Bjorksten B. Probiotics for the prevention or treatment of allergic diseases. J Allergy Clin Immunol 2007; 120:255–262.
- Untersmayr E, Jensen-Jarolim E. The role of protein digestibility and antacids on food allergy outcomes. J Allergy Clin Immunol 2008; 121:1301–1308.
- Untersmayr E, Scholl I, Swoboda I, et al. Antacid medication inhibits digestion of dietary proteins and causes food allergy: a fish allergy model in BALB/c mice. J Allergy Clin Immunol 2003; 112:616–623.
- Sicherer SH, Furlong TJ, Maes HH, Desnick RJ, Sampson HA, Gelb BD. Genetics of peanut allergy: a twin study. J Allergy Clin Immunol 2000; 106:53–56.
- Sicherer SH, Sampson HA. Food allergy: recent advances in pathophysiology and treatment. Annu Rev Med 2009; 60:261–277.
- Sampson HA, Anderson JA. Summary and recommendations: classification of gastrointestinal manifestations due to immunologic reactions to foods in infants and young children. J Pediatr Gastroenterol Nutr 2000; 30( suppl 1):S87–S94.
- Sampson HA, Sicherer SH, Birnbaum AH. AGA technical review on the evaluation of food allergy in gastrointestinal disorders. American Gastroenterological Association. Gastroenterology 2001; 120:1026–1040.
- Spergel JM, Pawlowski NA. Food allergy. Mechanisms, diagnosis, and management in children. Pediatr Clin North Am 2002; 49:73–96.
- Sampson HA. Utility of food-specific IgE concentrations in predicting symptomatic food allergy. J Allergy Clin Immunol 2001; 107:891–896.
- Ortolani C, Ispano M, Pastorello EA, Ansaloni R, Magri GC. Comparison of results of skin prick tests (with fresh foods and commercial food extracts) and RAST in 100 patients with oral allergy syndrome. J Allergy Clin Immunol 1989; 83:683–690.
- Perry TT, Matsui EC, Kay Conover-Walker M, Wood RA. The relationship of allergen-specific IgE levels and oral food challenge outcome. J Allergy Clin Immunol 2004; 114:144–149.
- Leung DY, Sampson HA, Yunginger JW, et al; Avon Longitudinal Study of Parents and Children Study Team. Effect of anti-IgE therapy in patients with peanut allergy. N Engl J Med 2003; 348:986–993.
- Sampson HA. A phase II, randomized double-blind, parallel-group, placebo-controlled, oral food challenge trial of Xolair (omalizumab) in peanut allergy (TOPS). J Allergy Clin Immunol 2007; 119 (suppl 1):S117.
- Buchanan AD, Green TD, Jones SM, et al Egg oral immunotherapy in nonanaphylactic children with egg allergy. J Allergy Clin Immunol 2007; 119:199–205.
- Burks AW, Jones SM. Egg oral immunotherapy in non-anaphylactic children with egg allergy: follow-up. J Allergy Clin Immunol 2008; 121:270–271.
- Skripak JM, Nash SD, Rowley H, et al. A randomized, double-blind, placebo-controlled study of milk oral immunotherapy for cow's milk allergy. J Allergy Clin Immunol 2008; 122:1154–1160.
- Furuta GT, Liacouras CA, Collins MH, et al; First International Gastrointestinal Eosinophil Research Symposium (FIGERS) Subcommittees. Eosinophilic esophagitis in children and adults: a systematic review and consensus recommendations for diagnosis and treatment. Gastroenterology 2007; 133:1342–1363.
- Rodrigo S, Abboud G, Oh D, et al. High intraepithelial eosinophil counts in esophageal squamous epithelium are not specific for eosinophilic esophagitis in adults. Am J Gastroenterol 2008; 103:435–442.
- Liacouras CA, Spergel JM, Ruchelli E, et al. Eosinophilic esophagitis: a 10-year experience in 381 children. Clin Gastroenterol Hepatol 2005; 3:1198–1206.
- Simon D, Marti H, Heer P, Simon HU, Braathen LR, Straumann A. Eosinophilic esophagitis is frequently associated with IgE-mediated allergic airway diseases. J Allergy Clin Immunol 2005; 115:1090–1092.
- Rothenberg ME, Mishra A, Collins MH, Putnam PE. Pathogenesis and clinical features of eosinophilic esophagitis. J Allergy Clin Immunol 2001; 108:891–894.
- Gupta SK, Fitzgerald JF, Kondratyuk T, HogenEsch H. Cytokine expression in normal and inflamed esophageal mucosa: a study into the pathogenesis of allergic eosinophilic esophagitis. J Pediatr Gastroenterol Nutr 2006; 42:22–26.
- Straumann A, Bauer M, Fischer B, Blaser K, Simon HU. Idiopathic eosinophilic esophagitis is associated with a T(H)2-type allergic inflammatory response. J Allergy Clin Immunol 2001; 108:954–961.
- Mishra A, Rothenberg ME. Intratracheal IL-13 induces eosinophilic esophagitis by an IL-5, eotaxin-1, and STAT6-dependent mechanism. Gastroenterology 2003; 125:1419–1427.
- Akei HS, Mishra A, Blanchard C, Rothenberg ME. Epicutaneous antigen exposure primes for experimental eosinophilic esophagitis in mice. Gastroenterology 2005; 129:985–994.
- Blanchard C, Wang N, Stringer KF, et al. Eotaxin-3 and a uniquely conserved gene-expression profile in eosinophilic esophagitis. J Clin Invest 2006; 116:536–547.
- Fogg MI, Ruchelli E, Spergel JM. Pollen and eosinophilic esophagitis. J Allergy Clin Immunol 2003; 112:796–797.
- Almansa C, Krishna M, Buchner AM, et al. Seasonal distribution in newly diagnosed cases of eosinophilic esophagitis in adults. Am J Gastroenterol 2009; 104:828–833.
- Kelly KJ, Lazenby AJ, Rowe PC, Yardley JH, Perman JA, Sampson HA. Eosinophilic esophagitis attributed to gastroesophageal reflux: improvement with an amino acid-based formula. Gastroenterology 1995; 109:1503–1512.
- Markowitz JE, Spergel JM, Ruchelli E, Liacouras CA. Elemental diet is an effective treatment for eosinophilic esophagitis in children and adolescents. Am J Gastroenterol 2003; 98:777–782.
- Kagalwalla AF, Sentongo TA, Ritz S, et al. Effect of six-food elimination diet on clinical and histologic outcomes in eosinophilic esophagitis. Clin Gastroenterol Hepatol 2006; 4:1097–1102.
- Gonsalves N, Yang GY, Doerfler B, et al. A prospective clinical trial of six food elimination diet and reintroduction of causative agents in adults with eosinophilic esophagitis [abstract]. Gastroenterology 2008; 134( suppl 1):A104–A105.
- Simon D, Straumann A, Wenk A, Spichtin H, Simon HU, Braathen LR. Eosinophilic esophagitis in adults—no clinical relevance of wheat and rye sensitizations. Allergy 2006; 61:1480–1483.
- Antón Remirez J, Escudero R, Caceres O, Fernandez-Benitez M. Eosinophilic esophagitis. Allergol Immunopathol (Madr) 2006; 34:79–81.
- Yamazaki K, Murray JA, Arora AS, et al. Allergen-specific in vitro cytokine production in adult patients with eosinophilic esophagitis. Dig Dis Sci 2006; 51:1934–1941.
- Penfield JD, Lang DM, Goldblum JR, Lopez R, Falk GW. The role of allergy evaluation in adults with eosinophilic esophagitis. J Clin Gastroenterol 2009(Epub ahead of print)
- Spergel JM, Andrews T, Brown-Whitehorn TF, Beausoleil JL, Liacouras CA. Treatment of eosinophilic esophagitis with specific food elimination diet directed by a combination of skin prick and patch tests. Ann Allergy Asthma Immunol 2005; 95:336–343.
- Spergel JM, Brown-Whitehorn T, Beausoleil JL, Shuker M, Liacouras CA. Predictive values for skin prick test and atopy patch test for eosinophilic esophagitis. J Allergy Clin Immunol 2007; 119:509–511.
- Bruijnzeel-Koomen C, Ortolani C, Aas K, et al. Adverse reactions to food. European Academy of Allergology and Clinical Immunology Subcommittee. Allergy 1995; 50:623–635.
- Sampson HA. Update on food allergy. J Allergy Clin Immunol 2004; 113:805–819.
- Sicherer SH, Sampson HA. 9. Food allergy. J Allergy Clin Immunol 2006; 117 (suppl 2):S470–S475.
- Sicherer SH, Munoz-Furlong A, Sampson HA. Prevalence of peanut and tree nut allergy in the United States determined by means of a random digit dial telephone survey: a 5-year follow-up study. J Allergy Clin Immunol 2003; 112:1203–1207.
- American College of Allergy, Asthma, & Immunology. Food allergy: a practice parameter. Ann Allergy Asthma Immunol 2006; 96( suppl 2):S1–S68.
- Wood RA. The natural history of food allergy. Pediatrics 2003; 111:1631–1637.
- Hourihane JO, Roberts SA, Warner JO. Resolution of peanut allergy: case-control study. BMJ 1998; 316:1271–1275.
- Fleischer DM, Conover-Walker MK, Matsui EC, Wood RA. The natural history of tree nut allergy. J Allergy Clin Immunol 2005; 116:1087–1093.
- Husby S, Foged N, Host A, Svehag SE. Passage of dietary antigens into the blood of children with coeliac disease. Quantification and size distribution of absorbed antigens. Gut 1987; 28:1062–1072.
- Mowat AM. Anatomical basis of tolerance and immunity to intestinal antigens. Nat Rev Immunol 2003; 3:331–341.
- Frossard CP, Tropia L, Hauser C, Eigenmann PA. Lymphocytes in Peyer patches regulate clinical tolerance in a murine model of food allergy. J Allergy Clin Immunol 2004; 113:958–964.
- Jain SL, Barone KS, Flanagan MP, Michael JG. Activation patterns of murine B cells after oral administration of an encapsulated soluble antigen. Vaccine 1996; 14:1291–1297.
- Kopper RA, Odum NJ, Sen M, Helm RM, Stanley JS, Burks AW. Peanut protein allergens: the effect of roasting on solubility and allergenicity. Int Arch Allergy Immunol 2005; 136:16–22.
- Lack G. Epidemiologic risks for food allergy. J Allergy Clin Immunol 2008; 121:1331–1336.
- Lack G, Fox D, Northstone K, Golding J; Avon Longitudinal Study of Parents and Children Study Team. Factors associated with the development of peanut allergy in childhood. N Engl J Med 2003; 348:977–985.
- Sudo N, Sawamura S, Tanaka K, Aiba Y, Kubo C, Koga Y. The requirement of intestinal bacterial flora for the development of an IgE production system fully susceptible to oral tolerance induction. J Immunol 1997; 159:1739–1745.
- Bashir ME, Louie S, Shi HN, Nagler-Anderson C. Toll-like receptor 4 signaling by intestinal microbes influences susceptibility to food allergy. J Immunol 2004; 172:6978–6987.
- Kopp MV, Hennemuth I, Heinzmann A, Urbanek R. Randomized, double-blind, placebo-controlled trial of probiotics for primary prevention: no clinical effects of lactobacillus GG supplementation. Pediatrics 2008; 121:e850–e856.
- Kukkonen K, Savilahti E, Haahtela T, et al. Probiotics and prebiotic galacto-oligosaccharides in the prevention of allergic diseases: a randomized, double-blind, placebo-controlled trial. J Allergy Clin Immunol 2007; 119:192–198.
- Osborn DA, Sinn JK. Probiotics in infants for prevention of allergic disease and food hypersensitivity. Cochrane Database Syst Rev 2007;CD006475.
- Prescott SL, Bjorksten B. Probiotics for the prevention or treatment of allergic diseases. J Allergy Clin Immunol 2007; 120:255–262.
- Untersmayr E, Jensen-Jarolim E. The role of protein digestibility and antacids on food allergy outcomes. J Allergy Clin Immunol 2008; 121:1301–1308.
- Untersmayr E, Scholl I, Swoboda I, et al. Antacid medication inhibits digestion of dietary proteins and causes food allergy: a fish allergy model in BALB/c mice. J Allergy Clin Immunol 2003; 112:616–623.
- Sicherer SH, Furlong TJ, Maes HH, Desnick RJ, Sampson HA, Gelb BD. Genetics of peanut allergy: a twin study. J Allergy Clin Immunol 2000; 106:53–56.
- Sicherer SH, Sampson HA. Food allergy: recent advances in pathophysiology and treatment. Annu Rev Med 2009; 60:261–277.
- Sampson HA, Anderson JA. Summary and recommendations: classification of gastrointestinal manifestations due to immunologic reactions to foods in infants and young children. J Pediatr Gastroenterol Nutr 2000; 30( suppl 1):S87–S94.
- Sampson HA, Sicherer SH, Birnbaum AH. AGA technical review on the evaluation of food allergy in gastrointestinal disorders. American Gastroenterological Association. Gastroenterology 2001; 120:1026–1040.
- Spergel JM, Pawlowski NA. Food allergy. Mechanisms, diagnosis, and management in children. Pediatr Clin North Am 2002; 49:73–96.
- Sampson HA. Utility of food-specific IgE concentrations in predicting symptomatic food allergy. J Allergy Clin Immunol 2001; 107:891–896.
- Ortolani C, Ispano M, Pastorello EA, Ansaloni R, Magri GC. Comparison of results of skin prick tests (with fresh foods and commercial food extracts) and RAST in 100 patients with oral allergy syndrome. J Allergy Clin Immunol 1989; 83:683–690.
- Perry TT, Matsui EC, Kay Conover-Walker M, Wood RA. The relationship of allergen-specific IgE levels and oral food challenge outcome. J Allergy Clin Immunol 2004; 114:144–149.
- Leung DY, Sampson HA, Yunginger JW, et al; Avon Longitudinal Study of Parents and Children Study Team. Effect of anti-IgE therapy in patients with peanut allergy. N Engl J Med 2003; 348:986–993.
- Sampson HA. A phase II, randomized double-blind, parallel-group, placebo-controlled, oral food challenge trial of Xolair (omalizumab) in peanut allergy (TOPS). J Allergy Clin Immunol 2007; 119 (suppl 1):S117.
- Buchanan AD, Green TD, Jones SM, et al Egg oral immunotherapy in nonanaphylactic children with egg allergy. J Allergy Clin Immunol 2007; 119:199–205.
- Burks AW, Jones SM. Egg oral immunotherapy in non-anaphylactic children with egg allergy: follow-up. J Allergy Clin Immunol 2008; 121:270–271.
- Skripak JM, Nash SD, Rowley H, et al. A randomized, double-blind, placebo-controlled study of milk oral immunotherapy for cow's milk allergy. J Allergy Clin Immunol 2008; 122:1154–1160.
- Furuta GT, Liacouras CA, Collins MH, et al; First International Gastrointestinal Eosinophil Research Symposium (FIGERS) Subcommittees. Eosinophilic esophagitis in children and adults: a systematic review and consensus recommendations for diagnosis and treatment. Gastroenterology 2007; 133:1342–1363.
- Rodrigo S, Abboud G, Oh D, et al. High intraepithelial eosinophil counts in esophageal squamous epithelium are not specific for eosinophilic esophagitis in adults. Am J Gastroenterol 2008; 103:435–442.
- Liacouras CA, Spergel JM, Ruchelli E, et al. Eosinophilic esophagitis: a 10-year experience in 381 children. Clin Gastroenterol Hepatol 2005; 3:1198–1206.
- Simon D, Marti H, Heer P, Simon HU, Braathen LR, Straumann A. Eosinophilic esophagitis is frequently associated with IgE-mediated allergic airway diseases. J Allergy Clin Immunol 2005; 115:1090–1092.
- Rothenberg ME, Mishra A, Collins MH, Putnam PE. Pathogenesis and clinical features of eosinophilic esophagitis. J Allergy Clin Immunol 2001; 108:891–894.
- Gupta SK, Fitzgerald JF, Kondratyuk T, HogenEsch H. Cytokine expression in normal and inflamed esophageal mucosa: a study into the pathogenesis of allergic eosinophilic esophagitis. J Pediatr Gastroenterol Nutr 2006; 42:22–26.
- Straumann A, Bauer M, Fischer B, Blaser K, Simon HU. Idiopathic eosinophilic esophagitis is associated with a T(H)2-type allergic inflammatory response. J Allergy Clin Immunol 2001; 108:954–961.
- Mishra A, Rothenberg ME. Intratracheal IL-13 induces eosinophilic esophagitis by an IL-5, eotaxin-1, and STAT6-dependent mechanism. Gastroenterology 2003; 125:1419–1427.
- Akei HS, Mishra A, Blanchard C, Rothenberg ME. Epicutaneous antigen exposure primes for experimental eosinophilic esophagitis in mice. Gastroenterology 2005; 129:985–994.
- Blanchard C, Wang N, Stringer KF, et al. Eotaxin-3 and a uniquely conserved gene-expression profile in eosinophilic esophagitis. J Clin Invest 2006; 116:536–547.
- Fogg MI, Ruchelli E, Spergel JM. Pollen and eosinophilic esophagitis. J Allergy Clin Immunol 2003; 112:796–797.
- Almansa C, Krishna M, Buchner AM, et al. Seasonal distribution in newly diagnosed cases of eosinophilic esophagitis in adults. Am J Gastroenterol 2009; 104:828–833.
- Kelly KJ, Lazenby AJ, Rowe PC, Yardley JH, Perman JA, Sampson HA. Eosinophilic esophagitis attributed to gastroesophageal reflux: improvement with an amino acid-based formula. Gastroenterology 1995; 109:1503–1512.
- Markowitz JE, Spergel JM, Ruchelli E, Liacouras CA. Elemental diet is an effective treatment for eosinophilic esophagitis in children and adolescents. Am J Gastroenterol 2003; 98:777–782.
- Kagalwalla AF, Sentongo TA, Ritz S, et al. Effect of six-food elimination diet on clinical and histologic outcomes in eosinophilic esophagitis. Clin Gastroenterol Hepatol 2006; 4:1097–1102.
- Gonsalves N, Yang GY, Doerfler B, et al. A prospective clinical trial of six food elimination diet and reintroduction of causative agents in adults with eosinophilic esophagitis [abstract]. Gastroenterology 2008; 134( suppl 1):A104–A105.
- Simon D, Straumann A, Wenk A, Spichtin H, Simon HU, Braathen LR. Eosinophilic esophagitis in adults—no clinical relevance of wheat and rye sensitizations. Allergy 2006; 61:1480–1483.
- Antón Remirez J, Escudero R, Caceres O, Fernandez-Benitez M. Eosinophilic esophagitis. Allergol Immunopathol (Madr) 2006; 34:79–81.
- Yamazaki K, Murray JA, Arora AS, et al. Allergen-specific in vitro cytokine production in adult patients with eosinophilic esophagitis. Dig Dis Sci 2006; 51:1934–1941.
- Penfield JD, Lang DM, Goldblum JR, Lopez R, Falk GW. The role of allergy evaluation in adults with eosinophilic esophagitis. J Clin Gastroenterol 2009(Epub ahead of print)
- Spergel JM, Andrews T, Brown-Whitehorn TF, Beausoleil JL, Liacouras CA. Treatment of eosinophilic esophagitis with specific food elimination diet directed by a combination of skin prick and patch tests. Ann Allergy Asthma Immunol 2005; 95:336–343.
- Spergel JM, Brown-Whitehorn T, Beausoleil JL, Shuker M, Liacouras CA. Predictive values for skin prick test and atopy patch test for eosinophilic esophagitis. J Allergy Clin Immunol 2007; 119:509–511.
KEY POINTS
- Food allergies can be classified as mediated by immunoglobulin E (IgE-mediated), non-IgE-mediated, or mixed. Their clinical presentation can vary from life-threatening anaphylaxis in IgE-mediated reactions to chronic, delayed symptoms as seen in eosinophilic esophagitis (a mixed reaction).
- The diagnosis of an IgE-mediated food allergy is made by taking a complete history and performing directed testing—skin-prick testing or measurement of foodspecific IgE levels in the serum, or both.
- Despite promising developments, food allergies continue to be treated primarily by telling patients to avoid allergens and to initiate therapy if ingestion occurs.
- Because most patients with eosinophilic esophagitis have a strong history of atopic disease and respond to allergen-free diets, a complete evaluation by a specialist in allergy and immunology is recommended.