User login
SILVER SPRING, MD. – A four-rod subdermal implant that slowly releases buprenorphine over 6 months should be approved for maintenance treatment of opioid dependence, although more work is needed to determine optimal dosing strategies and how to address the potential risks of the treatment, the majority of a Food and Drug Administration advisory panel recommended.
At a meeting on March 21, the FDA’s Psychopharmacologic Drugs Advisory Committee voted 10-4, with 1 abstention, to recommend approval, based on the efficacy, safety, and risk-benefit profile in opioid-addicted adults treated with the implants, in two phase III placebo-controlled studies. In those studies, the mean proportion of negative urine tests over 24 weeks, the primary endpoint, was significantly higher among those who received the buprenorphine implant, compared with those who had a placebo implant.
The panel agreed the treatment had a beneficial effect on reducing illicit opioid use, but those voting on both sides pointed out that more information on dosing was needed. The product is indicated for patients who are maintained on 12 mg-16 mg of oral buprenorphine a day. But as addiction treatment specialists on the panel pointed out, some patients are maintained on lower oral doses, and there were no data on how to use the product in such patients. The manufacturer, Titan Pharmaceuticals, plans to study that issue further.
The panel also voted 12-2 with 1 abstention, that the company had adequately characterized the safety profile of treatment with the buprenorphine implants, which was associated with the known side effects of the oral formulation in the trials. But there are also the potential complications associated with surgical implantation and removal of the rods every 6 months, as well as the potential for abuse, misuse and diversion, and accidental exposure, which is addressed in a complex risk management program proposed by the manufacturer.
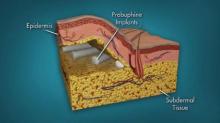
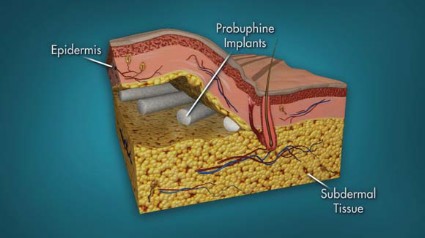
Using similar technology as contraceptive implants, the product consists of four rods, each measuring 26 mm by 2.5 mm and containing 80 mg of the buprenorphine, a partial mu-opioid receptor agonist that has been available in oral formulations since 2002 for the treatment of opioid dependence. The rods are subdermally implanted in the upper arm with a single-use application by a trained health care provider in an office-based surgical procedure with local anesthesia after the patient has been titrated with an oral dose. The rods stay in place for up to 6 months, at which time they are removed and replaced at another site.
"Clearly, the data need to be further assessed and the safety and dosing improved upon," said Dr. Christopher J. Kratochvil, professor of psychiatry and pediatrics, University of Nebraska, Omaha. But he voted in favor of approval, commenting, "This is will at least be an incremental step forward for a disorder that has very tragic consequences."
"Overall, I thought that benefit was shown and there might be a particular subset of patients who respond best to this intervention," said one of the two panelists with expertise on contraceptive implants, Dr. Geri D. Hewitt, an obstetrician-gynecologist at Ohio State University, Columbus, who voted for approval. "I did not see any evidence of significant harm; I do have questions about dosing and which physicians will be willing to place this and ... it’s going to be important to keep track of adverse event reporting," she added.
Voting against approval, Dr.. Laura F. McNicholas of the Center for Studies of Addiction at the University of Pennsylvania, Philadelphia, said she thought the product was approvable, but that approval before a dose-ranging study was conducted would be premature. A major concern she had was running out of sites to implant the rods for patients on long-term treatment, since many of her patients have been taking buprenorphine continuously for years. Many of her patients also are on doses under 12 mg a day, and there was no information on how to dose such patients with the implant, for which there is currently only one dose, she added.
So far, the company has identified four sites on the upper arms as appropriate for implantation and is studying this further.
The two studies enrolled 450 adults (mean age, mid-30s) addicted to an opioid, randomized to treatment with the buprenorphine implant or a placebo implant. The most common drug of abuse was heroin in 52% to 67%, followed by prescription analgesics in 33% to 48%.Over 24 weeks, the mean percentage of opioid-negative urine samples was statistically significantly greater among those who were treated with the buprenorphine implant (about 40% and about 35% among those who received the active drug vs. about 30% and 15%, respectively, among those on the placebo implant.)
About 80% of the patients were adequately treated with four implants, but in some patients, a fifth rod or supplemental treatment with sublingual buprenorphine was needed. Completion rates were lower among those on placebo (31% and 26%, vs. 64% and 66%, respectively). In the two studies, the most common implant site–related adverse events included pruritus, pain, erythema, edema, and hematoma.
The company’s Risk Evaluation and Mitigation Strategy (REMS) includes plans for restricted distribution, a specialty pharmacy only, and a training-certification program for health care providers who implant the rods. (The FDA requires a REMS for a drug when it determines that a risk management strategies beyond the label are needed to ensure the benefits of a drug outweigh its risks.) Both the panel and the FDA reviewers said the REMS needs to be improved.
When asked about the panel’s recommendation, Dr. Robert L DuPont said in an interview that it is a major development in medication-assisted treatment of opiate dependence in light of problems with diversion and nonmedical use of buprenorphine. "This novel dosing strategy removes the risk of diversion in a large population of drug abusing patients that poses a high risk of diversion and misuse," said Dr. DuPont, the first director of the National Institute on Drug Abuse.
"The development of abuse-resistant formulations of controlled substances is one of the best new ideas to turn back the major epidemic of prescription drug abuse."
If approved, the panel recommended that other potential problems with the device should be monitored closely, including removal of implants by nonmedical personnel for diversion (which was not seen in the studies) and the risk of long-term exposure to the components if they are not removed. The panel urged study of whether implants can be inserted in previous implant sites.
Buprenorphine can be administered in an office setting as opposed to a methadone clinic. The currently marketed buprenorphine products are Subutex and Suboxone (buprenorphine with naloxone), both available in generic formulations; and a sublingual formulation approved in 2010. In 2012, 10.7 million prescriptions were dispensed for buprenorphine products for an estimated 1 million patients, mostly the buprenorphine/naloxone combination, according to the FDA. Between January 2003 and December 2012, an estimated 40 million buprenorphine prescriptions were written. The top three categories of prescribers were general practitioners/family physicians/doctors of osteopathy (one category), psychiatrists, and internists.
If approved, the manufacturer plans to market it as Probuphine. The FDA usually follows the recommendations of its advisory panels. Panelists have been cleared of potential conflicts of interest related to the topic of the meeting. Occasionally, a panelist might be given a waiver, but not at this meeting.
SILVER SPRING, MD. – A four-rod subdermal implant that slowly releases buprenorphine over 6 months should be approved for maintenance treatment of opioid dependence, although more work is needed to determine optimal dosing strategies and how to address the potential risks of the treatment, the majority of a Food and Drug Administration advisory panel recommended.
At a meeting on March 21, the FDA’s Psychopharmacologic Drugs Advisory Committee voted 10-4, with 1 abstention, to recommend approval, based on the efficacy, safety, and risk-benefit profile in opioid-addicted adults treated with the implants, in two phase III placebo-controlled studies. In those studies, the mean proportion of negative urine tests over 24 weeks, the primary endpoint, was significantly higher among those who received the buprenorphine implant, compared with those who had a placebo implant.
The panel agreed the treatment had a beneficial effect on reducing illicit opioid use, but those voting on both sides pointed out that more information on dosing was needed. The product is indicated for patients who are maintained on 12 mg-16 mg of oral buprenorphine a day. But as addiction treatment specialists on the panel pointed out, some patients are maintained on lower oral doses, and there were no data on how to use the product in such patients. The manufacturer, Titan Pharmaceuticals, plans to study that issue further.
The panel also voted 12-2 with 1 abstention, that the company had adequately characterized the safety profile of treatment with the buprenorphine implants, which was associated with the known side effects of the oral formulation in the trials. But there are also the potential complications associated with surgical implantation and removal of the rods every 6 months, as well as the potential for abuse, misuse and diversion, and accidental exposure, which is addressed in a complex risk management program proposed by the manufacturer.
Using similar technology as contraceptive implants, the product consists of four rods, each measuring 26 mm by 2.5 mm and containing 80 mg of the buprenorphine, a partial mu-opioid receptor agonist that has been available in oral formulations since 2002 for the treatment of opioid dependence. The rods are subdermally implanted in the upper arm with a single-use application by a trained health care provider in an office-based surgical procedure with local anesthesia after the patient has been titrated with an oral dose. The rods stay in place for up to 6 months, at which time they are removed and replaced at another site.
"Clearly, the data need to be further assessed and the safety and dosing improved upon," said Dr. Christopher J. Kratochvil, professor of psychiatry and pediatrics, University of Nebraska, Omaha. But he voted in favor of approval, commenting, "This is will at least be an incremental step forward for a disorder that has very tragic consequences."
"Overall, I thought that benefit was shown and there might be a particular subset of patients who respond best to this intervention," said one of the two panelists with expertise on contraceptive implants, Dr. Geri D. Hewitt, an obstetrician-gynecologist at Ohio State University, Columbus, who voted for approval. "I did not see any evidence of significant harm; I do have questions about dosing and which physicians will be willing to place this and ... it’s going to be important to keep track of adverse event reporting," she added.
Voting against approval, Dr.. Laura F. McNicholas of the Center for Studies of Addiction at the University of Pennsylvania, Philadelphia, said she thought the product was approvable, but that approval before a dose-ranging study was conducted would be premature. A major concern she had was running out of sites to implant the rods for patients on long-term treatment, since many of her patients have been taking buprenorphine continuously for years. Many of her patients also are on doses under 12 mg a day, and there was no information on how to dose such patients with the implant, for which there is currently only one dose, she added.
So far, the company has identified four sites on the upper arms as appropriate for implantation and is studying this further.
The two studies enrolled 450 adults (mean age, mid-30s) addicted to an opioid, randomized to treatment with the buprenorphine implant or a placebo implant. The most common drug of abuse was heroin in 52% to 67%, followed by prescription analgesics in 33% to 48%.Over 24 weeks, the mean percentage of opioid-negative urine samples was statistically significantly greater among those who were treated with the buprenorphine implant (about 40% and about 35% among those who received the active drug vs. about 30% and 15%, respectively, among those on the placebo implant.)
About 80% of the patients were adequately treated with four implants, but in some patients, a fifth rod or supplemental treatment with sublingual buprenorphine was needed. Completion rates were lower among those on placebo (31% and 26%, vs. 64% and 66%, respectively). In the two studies, the most common implant site–related adverse events included pruritus, pain, erythema, edema, and hematoma.
The company’s Risk Evaluation and Mitigation Strategy (REMS) includes plans for restricted distribution, a specialty pharmacy only, and a training-certification program for health care providers who implant the rods. (The FDA requires a REMS for a drug when it determines that a risk management strategies beyond the label are needed to ensure the benefits of a drug outweigh its risks.) Both the panel and the FDA reviewers said the REMS needs to be improved.
When asked about the panel’s recommendation, Dr. Robert L DuPont said in an interview that it is a major development in medication-assisted treatment of opiate dependence in light of problems with diversion and nonmedical use of buprenorphine. "This novel dosing strategy removes the risk of diversion in a large population of drug abusing patients that poses a high risk of diversion and misuse," said Dr. DuPont, the first director of the National Institute on Drug Abuse.
"The development of abuse-resistant formulations of controlled substances is one of the best new ideas to turn back the major epidemic of prescription drug abuse."
If approved, the panel recommended that other potential problems with the device should be monitored closely, including removal of implants by nonmedical personnel for diversion (which was not seen in the studies) and the risk of long-term exposure to the components if they are not removed. The panel urged study of whether implants can be inserted in previous implant sites.
Buprenorphine can be administered in an office setting as opposed to a methadone clinic. The currently marketed buprenorphine products are Subutex and Suboxone (buprenorphine with naloxone), both available in generic formulations; and a sublingual formulation approved in 2010. In 2012, 10.7 million prescriptions were dispensed for buprenorphine products for an estimated 1 million patients, mostly the buprenorphine/naloxone combination, according to the FDA. Between January 2003 and December 2012, an estimated 40 million buprenorphine prescriptions were written. The top three categories of prescribers were general practitioners/family physicians/doctors of osteopathy (one category), psychiatrists, and internists.
If approved, the manufacturer plans to market it as Probuphine. The FDA usually follows the recommendations of its advisory panels. Panelists have been cleared of potential conflicts of interest related to the topic of the meeting. Occasionally, a panelist might be given a waiver, but not at this meeting.
SILVER SPRING, MD. – A four-rod subdermal implant that slowly releases buprenorphine over 6 months should be approved for maintenance treatment of opioid dependence, although more work is needed to determine optimal dosing strategies and how to address the potential risks of the treatment, the majority of a Food and Drug Administration advisory panel recommended.
At a meeting on March 21, the FDA’s Psychopharmacologic Drugs Advisory Committee voted 10-4, with 1 abstention, to recommend approval, based on the efficacy, safety, and risk-benefit profile in opioid-addicted adults treated with the implants, in two phase III placebo-controlled studies. In those studies, the mean proportion of negative urine tests over 24 weeks, the primary endpoint, was significantly higher among those who received the buprenorphine implant, compared with those who had a placebo implant.
The panel agreed the treatment had a beneficial effect on reducing illicit opioid use, but those voting on both sides pointed out that more information on dosing was needed. The product is indicated for patients who are maintained on 12 mg-16 mg of oral buprenorphine a day. But as addiction treatment specialists on the panel pointed out, some patients are maintained on lower oral doses, and there were no data on how to use the product in such patients. The manufacturer, Titan Pharmaceuticals, plans to study that issue further.
The panel also voted 12-2 with 1 abstention, that the company had adequately characterized the safety profile of treatment with the buprenorphine implants, which was associated with the known side effects of the oral formulation in the trials. But there are also the potential complications associated with surgical implantation and removal of the rods every 6 months, as well as the potential for abuse, misuse and diversion, and accidental exposure, which is addressed in a complex risk management program proposed by the manufacturer.
Using similar technology as contraceptive implants, the product consists of four rods, each measuring 26 mm by 2.5 mm and containing 80 mg of the buprenorphine, a partial mu-opioid receptor agonist that has been available in oral formulations since 2002 for the treatment of opioid dependence. The rods are subdermally implanted in the upper arm with a single-use application by a trained health care provider in an office-based surgical procedure with local anesthesia after the patient has been titrated with an oral dose. The rods stay in place for up to 6 months, at which time they are removed and replaced at another site.
"Clearly, the data need to be further assessed and the safety and dosing improved upon," said Dr. Christopher J. Kratochvil, professor of psychiatry and pediatrics, University of Nebraska, Omaha. But he voted in favor of approval, commenting, "This is will at least be an incremental step forward for a disorder that has very tragic consequences."
"Overall, I thought that benefit was shown and there might be a particular subset of patients who respond best to this intervention," said one of the two panelists with expertise on contraceptive implants, Dr. Geri D. Hewitt, an obstetrician-gynecologist at Ohio State University, Columbus, who voted for approval. "I did not see any evidence of significant harm; I do have questions about dosing and which physicians will be willing to place this and ... it’s going to be important to keep track of adverse event reporting," she added.
Voting against approval, Dr.. Laura F. McNicholas of the Center for Studies of Addiction at the University of Pennsylvania, Philadelphia, said she thought the product was approvable, but that approval before a dose-ranging study was conducted would be premature. A major concern she had was running out of sites to implant the rods for patients on long-term treatment, since many of her patients have been taking buprenorphine continuously for years. Many of her patients also are on doses under 12 mg a day, and there was no information on how to dose such patients with the implant, for which there is currently only one dose, she added.
So far, the company has identified four sites on the upper arms as appropriate for implantation and is studying this further.
The two studies enrolled 450 adults (mean age, mid-30s) addicted to an opioid, randomized to treatment with the buprenorphine implant or a placebo implant. The most common drug of abuse was heroin in 52% to 67%, followed by prescription analgesics in 33% to 48%.Over 24 weeks, the mean percentage of opioid-negative urine samples was statistically significantly greater among those who were treated with the buprenorphine implant (about 40% and about 35% among those who received the active drug vs. about 30% and 15%, respectively, among those on the placebo implant.)
About 80% of the patients were adequately treated with four implants, but in some patients, a fifth rod or supplemental treatment with sublingual buprenorphine was needed. Completion rates were lower among those on placebo (31% and 26%, vs. 64% and 66%, respectively). In the two studies, the most common implant site–related adverse events included pruritus, pain, erythema, edema, and hematoma.
The company’s Risk Evaluation and Mitigation Strategy (REMS) includes plans for restricted distribution, a specialty pharmacy only, and a training-certification program for health care providers who implant the rods. (The FDA requires a REMS for a drug when it determines that a risk management strategies beyond the label are needed to ensure the benefits of a drug outweigh its risks.) Both the panel and the FDA reviewers said the REMS needs to be improved.
When asked about the panel’s recommendation, Dr. Robert L DuPont said in an interview that it is a major development in medication-assisted treatment of opiate dependence in light of problems with diversion and nonmedical use of buprenorphine. "This novel dosing strategy removes the risk of diversion in a large population of drug abusing patients that poses a high risk of diversion and misuse," said Dr. DuPont, the first director of the National Institute on Drug Abuse.
"The development of abuse-resistant formulations of controlled substances is one of the best new ideas to turn back the major epidemic of prescription drug abuse."
If approved, the panel recommended that other potential problems with the device should be monitored closely, including removal of implants by nonmedical personnel for diversion (which was not seen in the studies) and the risk of long-term exposure to the components if they are not removed. The panel urged study of whether implants can be inserted in previous implant sites.
Buprenorphine can be administered in an office setting as opposed to a methadone clinic. The currently marketed buprenorphine products are Subutex and Suboxone (buprenorphine with naloxone), both available in generic formulations; and a sublingual formulation approved in 2010. In 2012, 10.7 million prescriptions were dispensed for buprenorphine products for an estimated 1 million patients, mostly the buprenorphine/naloxone combination, according to the FDA. Between January 2003 and December 2012, an estimated 40 million buprenorphine prescriptions were written. The top three categories of prescribers were general practitioners/family physicians/doctors of osteopathy (one category), psychiatrists, and internists.
If approved, the manufacturer plans to market it as Probuphine. The FDA usually follows the recommendations of its advisory panels. Panelists have been cleared of potential conflicts of interest related to the topic of the meeting. Occasionally, a panelist might be given a waiver, but not at this meeting.
FROM AN FDA ADVISORY PANEL MEETING