User login
Telehealth Program Helps Rural Women Veterans With Chronic Pain
Women veterans who live in rural areas face multifaceted challenges in managing chronic pain. Some barriers are logistical, including distance from health care facilities and the time required to get there, while others are financial, such as the cost of gas. Researchers from the Veterans Rural Health Resource Center and the Iowa City Veterans Affairs (VA) Health Care System designed a telehealth intervention specifically for rural women. What the study revealed was that social interactions and camaraderie may be just as important in reducing pain as gentle exercise and behavioral changes.
The participants, recruited from a Midwestern VA health care system, were dealing with chronic pain. In baseline measurements, average scores on the Pain, Enjoyment of Life, and General Activity three-item scale (PEG-3) indicated severe pain and functional interference. Notably, the researchers point out, rural women veterans with chronic pain are less likely than urban veterans to receive specialty pain care.
The researchers designed a program of pain self-management options, allowing the participants to sample from a range of empirically supported approaches in one easily accessible format. The program was primarily delivered over video, to support group processes as well as access to video-based components of the intervention (such as yoga lessons) or to display pages from the participant manual for in-session review. The researchers planned it as a women-only space, to provide a “psychologically safe, gender-sensitive, empowering environment.”
Eight weekly 90-minute sessions featured mindful movement (gentle yoga, graduated walking), peer connection and support, and an introduction to an evidence-based pain or lifestyle self-management topic such as nutrition. The program also included content based on previous work in chronic pain including acceptance and commitment therapy, cognitive behavioral therapy, and dialectical behavior therapy.
Of the 44 participants, 84% completed the intervention. About half of treatment completers (47%) were deemed responders, reporting a ≥ 30% reduction on their PEG-3 total scores. On the Global Impression of Change scale, 87% reported improvement.
Of the 30 participants who provided follow-up data, 94% were satisfied or very satisfied; no one reported being dissatisfied with the intervention.
In qualitative interviews, though, the researchers say a clear theme emerged, reflecting the impact and benefit of the balance of three social and psychological components: rapport with facilitators, connection with other women veterans, and maintenance of individuality.
In fact, the social support elements of the current intervention may have directly contributed to the observed improvements in pain severity and interference, the researchers suggest. They also cite another potential mechanism: impacting loneliness. Other studies have found that loneliness is prevalent among rural-dwelling women with chronic illnesses; research in rural settings also suggests that interventions focused on shared interests and common experiences might reduce loneliness. Moreover, reducing loneliness may have an independent benefit: In that research, loneliness was a risk factor for developing the pain, depression, and fatigue symptom cluster.
The women in the telehealth study often spoke about the emotional benefits and camaraderie of the group sessions. They reported feeling understood. “The most memorable thing was that I just felt good,” one participant said. “…They helped me approach what was going on in my life. …[T]hey never grouped us together. We were all individuals with similar problems.”
Social support might help to buffer stress, which in turn may improve the experience of pain, via its effect on stressor appraisals and coping resources. Thus, their findings pair well, the researchers say, with the creation of the Women Veterans Network (WoVeN), a national peer-facilitated social support intervention aimed at ameliorating loneliness and increasing support among women veterans. Ultimately, they suggest, their findings may lead to help for women veterans living with chronic pain—whether they live in the country or the city.
Women veterans who live in rural areas face multifaceted challenges in managing chronic pain. Some barriers are logistical, including distance from health care facilities and the time required to get there, while others are financial, such as the cost of gas. Researchers from the Veterans Rural Health Resource Center and the Iowa City Veterans Affairs (VA) Health Care System designed a telehealth intervention specifically for rural women. What the study revealed was that social interactions and camaraderie may be just as important in reducing pain as gentle exercise and behavioral changes.
The participants, recruited from a Midwestern VA health care system, were dealing with chronic pain. In baseline measurements, average scores on the Pain, Enjoyment of Life, and General Activity three-item scale (PEG-3) indicated severe pain and functional interference. Notably, the researchers point out, rural women veterans with chronic pain are less likely than urban veterans to receive specialty pain care.
The researchers designed a program of pain self-management options, allowing the participants to sample from a range of empirically supported approaches in one easily accessible format. The program was primarily delivered over video, to support group processes as well as access to video-based components of the intervention (such as yoga lessons) or to display pages from the participant manual for in-session review. The researchers planned it as a women-only space, to provide a “psychologically safe, gender-sensitive, empowering environment.”
Eight weekly 90-minute sessions featured mindful movement (gentle yoga, graduated walking), peer connection and support, and an introduction to an evidence-based pain or lifestyle self-management topic such as nutrition. The program also included content based on previous work in chronic pain including acceptance and commitment therapy, cognitive behavioral therapy, and dialectical behavior therapy.
Of the 44 participants, 84% completed the intervention. About half of treatment completers (47%) were deemed responders, reporting a ≥ 30% reduction on their PEG-3 total scores. On the Global Impression of Change scale, 87% reported improvement.
Of the 30 participants who provided follow-up data, 94% were satisfied or very satisfied; no one reported being dissatisfied with the intervention.
In qualitative interviews, though, the researchers say a clear theme emerged, reflecting the impact and benefit of the balance of three social and psychological components: rapport with facilitators, connection with other women veterans, and maintenance of individuality.
In fact, the social support elements of the current intervention may have directly contributed to the observed improvements in pain severity and interference, the researchers suggest. They also cite another potential mechanism: impacting loneliness. Other studies have found that loneliness is prevalent among rural-dwelling women with chronic illnesses; research in rural settings also suggests that interventions focused on shared interests and common experiences might reduce loneliness. Moreover, reducing loneliness may have an independent benefit: In that research, loneliness was a risk factor for developing the pain, depression, and fatigue symptom cluster.
The women in the telehealth study often spoke about the emotional benefits and camaraderie of the group sessions. They reported feeling understood. “The most memorable thing was that I just felt good,” one participant said. “…They helped me approach what was going on in my life. …[T]hey never grouped us together. We were all individuals with similar problems.”
Social support might help to buffer stress, which in turn may improve the experience of pain, via its effect on stressor appraisals and coping resources. Thus, their findings pair well, the researchers say, with the creation of the Women Veterans Network (WoVeN), a national peer-facilitated social support intervention aimed at ameliorating loneliness and increasing support among women veterans. Ultimately, they suggest, their findings may lead to help for women veterans living with chronic pain—whether they live in the country or the city.
Women veterans who live in rural areas face multifaceted challenges in managing chronic pain. Some barriers are logistical, including distance from health care facilities and the time required to get there, while others are financial, such as the cost of gas. Researchers from the Veterans Rural Health Resource Center and the Iowa City Veterans Affairs (VA) Health Care System designed a telehealth intervention specifically for rural women. What the study revealed was that social interactions and camaraderie may be just as important in reducing pain as gentle exercise and behavioral changes.
The participants, recruited from a Midwestern VA health care system, were dealing with chronic pain. In baseline measurements, average scores on the Pain, Enjoyment of Life, and General Activity three-item scale (PEG-3) indicated severe pain and functional interference. Notably, the researchers point out, rural women veterans with chronic pain are less likely than urban veterans to receive specialty pain care.
The researchers designed a program of pain self-management options, allowing the participants to sample from a range of empirically supported approaches in one easily accessible format. The program was primarily delivered over video, to support group processes as well as access to video-based components of the intervention (such as yoga lessons) or to display pages from the participant manual for in-session review. The researchers planned it as a women-only space, to provide a “psychologically safe, gender-sensitive, empowering environment.”
Eight weekly 90-minute sessions featured mindful movement (gentle yoga, graduated walking), peer connection and support, and an introduction to an evidence-based pain or lifestyle self-management topic such as nutrition. The program also included content based on previous work in chronic pain including acceptance and commitment therapy, cognitive behavioral therapy, and dialectical behavior therapy.
Of the 44 participants, 84% completed the intervention. About half of treatment completers (47%) were deemed responders, reporting a ≥ 30% reduction on their PEG-3 total scores. On the Global Impression of Change scale, 87% reported improvement.
Of the 30 participants who provided follow-up data, 94% were satisfied or very satisfied; no one reported being dissatisfied with the intervention.
In qualitative interviews, though, the researchers say a clear theme emerged, reflecting the impact and benefit of the balance of three social and psychological components: rapport with facilitators, connection with other women veterans, and maintenance of individuality.
In fact, the social support elements of the current intervention may have directly contributed to the observed improvements in pain severity and interference, the researchers suggest. They also cite another potential mechanism: impacting loneliness. Other studies have found that loneliness is prevalent among rural-dwelling women with chronic illnesses; research in rural settings also suggests that interventions focused on shared interests and common experiences might reduce loneliness. Moreover, reducing loneliness may have an independent benefit: In that research, loneliness was a risk factor for developing the pain, depression, and fatigue symptom cluster.
The women in the telehealth study often spoke about the emotional benefits and camaraderie of the group sessions. They reported feeling understood. “The most memorable thing was that I just felt good,” one participant said. “…They helped me approach what was going on in my life. …[T]hey never grouped us together. We were all individuals with similar problems.”
Social support might help to buffer stress, which in turn may improve the experience of pain, via its effect on stressor appraisals and coping resources. Thus, their findings pair well, the researchers say, with the creation of the Women Veterans Network (WoVeN), a national peer-facilitated social support intervention aimed at ameliorating loneliness and increasing support among women veterans. Ultimately, they suggest, their findings may lead to help for women veterans living with chronic pain—whether they live in the country or the city.
Thermal Therapy is Associated With Decreased Pain Treatment Use: A Retrospective Cohort Study
Thermal Therapy is Associated With Decreased Pain Treatment Use: A Retrospective Cohort Study
Growing recognition of the limitations of long-term opioid therapy for chronic noncancer pain has highlighted the importance of nonpharmacologic approaches to pain treatment.1,2 These treatments are varied and may include psychological and behavioral therapies (eg, cognitive behavioral therapy for chronic pain), exercise and movement therapies (eg, yoga), and manual therapies (eg, chiropractic). Body surface cold therapy, while predominantly used to reduce postoperative pain and inflammation,3,4 pain subsequent to acute musculoskeletal injury,5 and pain related to muscle soreness,6 is a nonpharmacologic treatment that has shown pain-reducing benefits for chronic low back pain and knee osteoarthritis, as has its counterpart, superficial heat therapy.7-9 Heat therapy has also been shown to improve strength, flexibility, and activities of daily living in patients with chronic low back pain.10,11 Cold and heat therapies are commonly used complementarily. Cold therapies aim to reduce blood flow and inflammation and are often used immediately following trauma to an affected area, whereas heat therapies increase blood flow and metabolic activity and are commonly used to promote healing.5
Heat and cold therapies (also known as thermal therapies) benefit resource-limited health care systems, as most devices require a single expenditure and can be self-administered by patients at home as part of their pain self-management plan. In addition, these pain self-management tools may attenuate the need for more expensive specialty pain care and ongoing analgesic pharmacotherapy. Despite their potential, few studies have characterized the benefits of thermal therapies for patients with heterogeneous chronic pain syndromes.
The purpose of this retrospective cohort study was to examine the potential clinical and health system benefits of patient-administered thermal therapy. Our primary hypothesis was that patients with chronic musculoskeletal pain who received a thermal therapy self-management device would have fewer days of opioid prescriptions compared with a sample of matched control patients. Secondarily, we hypothesized that patients who received a thermal therapy device would have lower utilization of specialty pain care, fewer potentially hazardous opioid prescriptions (eg, high-dose opioid therapy and concurrent opioid and benzodiazepine prescriptions), fewer prescriptions for nonopioid analgesic medications, and decreased pain intensity when compared with matched controls.
Methods
This retrospective cohort study compared pain pharmacotherapy, pain treatment utilization, and pain intensity outcomes between patients who received a thermal therapy device and matched patients who did not. The study was approved by the US Department of Veterans Affairs (VA) Portland Health Care System Institutional Review Board and was granted a waiver of informed consent to access patient electronic health records (EHRs).
Pain Care
The VA uses ThermaZone thermal therapy devices (Innovative Medical Equipment) for chronic pain treatment. The device uses thermoelectric technology to provide point-of-contact cooling and heating therapy through site-specific pads (eg, ankle, knee, hip, back, elbow, shoulder). Patients place pads on sites where they experience pain, and temperature regulated water circulates through the device and to the pad, providing consistent, localized thermal therapy. The pads range in temperature from 1 °C to 52 °C, and temperatures are self-monitored and controlled by the patient.
Standard pain care in this study followed the VA stepped model of pain care, which builds on a foundation of patient education for pain self-management approaches (eg, exercise, mindfulness, relaxation, social support).12 According to the VA stepped model of pain care, all patients with chronic pain should engage in these foundational self-management approaches. However, some patients require more intensive care. The VA stepped-up treatment engages primary and specialty care services—such as physical therapy, pharmacy, complementary and integrative health approaches, mental health, and substance use services—and, when indicated, may escalate treatment to interdisciplinary pain teams or tertiary pain centers. In this retrospective cohort study, treatment patients received a thermal therapy device and standard of care, whereas control patients received standard care only.
Sample Selection
Eligible patients were aged ≥ 18 years, had a musculoskeletal pain diagnosis documented in the EHR in the year prior to thermal therapy device receipt (or during the same period for a treatment patient’s matched control), and were enrolled in VA health care during the study period. Patients who died during the study period were excluded. Treatment patients received a thermal therapy device from the VA between January 1, 2017, and December 31, 2018, when complete data on thermal therapy devices were available. For control patients, the VA Corporate Data Warehouse (CDW) was used to identify VA patients with characteristics similar to those of treated patients.
We modeled the probability that a patient would receive a thermal therapy device using logistic regression. Predictor variables were measured in the year prior to device receipt and included variables associated with pain treatment utilization and analgesic pharmacotherapy receipt, as recommended by Brookhart et al.13 These included age, sex, race, ethnicity, VA service-connected disability status, comorbidities, receipt of medications for opioid use disorder, pain diagnoses, mental health diagnoses, and substance use disorder diagnoses.14-19
The resulting propensity scores (eg, predicted probabilities) were used to match treatment patients 1:1 with control patients using a nearest-neighbor matching algorithm.20 This procedure matches a control patient with the closest propensity score to that of the corresponding treatment patient. An exact match on VA facility was required (eg, treatment patients and matched controls received care at the same VA facility). Standardized differences were used to assess covariate balance between the matched groups, and kernel density plots of propensity scores tested for sufficient overlap.21 Control patients were selected from a pool of 1,150,149 patients.
Study Variables
The index date was the date the thermal therapy device was released to treatment patients or the same date for the matched controls. Data were extracted from the CDW over a 24-month period: 12 months prior to the index date through 12 months afterwards. Collecting data in the 12 months prior to treatment initiation allowed us to adjust for covariates and provided greater precision, as recommended for observational study designs.22
Treatment conditions were defined dichotomously as receipt vs nonreceipt of a thermal therapy device. The primary outcome was the number of days of opioid use in the 12 months following the index date. Additional outcomes included days of high-dose opioid therapy (≥ 50 mg morphine equivalent [MME] daily), concurrent opioid and benzodiazepine prescriptions, and nonopioid analgesic pharmacotherapy (eg, nonsteroidal anti-inflammatory drugs, acetaminophen, muscle relaxants). All prescription data were based on medication fills obtained from VA pharmacy records. Additional outcomes included the number of visits to physical therapy, occupational therapy, physical medicine and rehabilitation, and interdisciplinary pain clinics, including interventional pain medicine.
Pain intensity ratings were collected as part of routine VA care using a numeric scale from 0 (no pain) to 10 (worst possible pain). Pain intensity trajectories were computed using all available EHR-derived pain intensity score data for each patient in the 12 months prior to and following the index date.
Covariates were extracted from the EHR and evaluated in the year prior to the index date, unless otherwise noted. They included age at the index date; self-reported sex, and race and ethnicity; service-connected disability status (disability awarded as a result of military service-related trauma or injury); Charlson Comorbidity Index; and diagnoses of opioid use disorder, alcohol use disorder, other substance use disorder, mood disorder, posttraumatic stress disorder, other anxiety disorder, psychotic disorder, neuropathic pain, and headache pain.23 All diagnoses were coded as yes if identified in the EHR as a focus of treatment during any clinical encounter in the year prior to the index date or no if not identified as a focus of treatment.
The number of days patients had been living with pain was calculated and defined as the number of days from the first pain diagnosis available in a patient’s EHR to the index date. Finally, the number of pain-related surgeries (eg, surgeries with ≥ 1 pain diagnoses associated with the clinical encounter) and average pain intensity were computed for the pre- and postindex date evaluation periods and included as model covariates.
Statistical Analyses
For the 4 pharmacotherapy and 4 nonpharmacologic treatment usage outcomes, we tested the fit of linear models and several models with count distributions using the Bayesian information criterion.24 Count distributions included Poisson, zero-inflated Poisson, negative binomial, and zero-inflated negative binomial. With 1 exception (days of high-dose opioid use), a negative binomial distribution best fit the data. For days of high-dose opioid use, a Gaussian distribution best fit the data.
Eight separate mixed-effects regression analyses compared changes in each outcome from the 12-month preindex period through the 12-month postindex period between treatment and control patients by testing the Time × Treatment interaction. This approach statistically accounts for observed pretreatment differences in outcome variables. Statistics for the main effects of time and treatment are also presented. To reduce bias, models controlled for covariates specified previously.
For pain intensity, we used random-effects growth modeling to quantify both fixed and random effects of pain intensity at the index date (eg, the model intercept), which estimates pain at the time of treatment initiation, and change in pain during the 12 months following the index date (eg, the model slope), which characterizes the trajectory of pain intensity ratings.25 The model included piecewise components of pain score trajectories in the 12 months prior to the index date and in the 12 months following the index date. Several types of change for the 12-month postindex observation period were explored—including quadratic and cubic curvilinear change. A linear model for change in pain over time provided the best fit based on the Bayesian information criterion and parsimony of model parameters.26 We report estimates of change in pain over time in monthly intervals for ease of interpretation. However, models used all individual pain scores rather than computing monthly averages when > 1 pain score was available within a month, as recommended in previous research.27 This approach makes optimal use of all available data. Both random effects (intercept and slope) were regressed onto the set of covariates described previously.
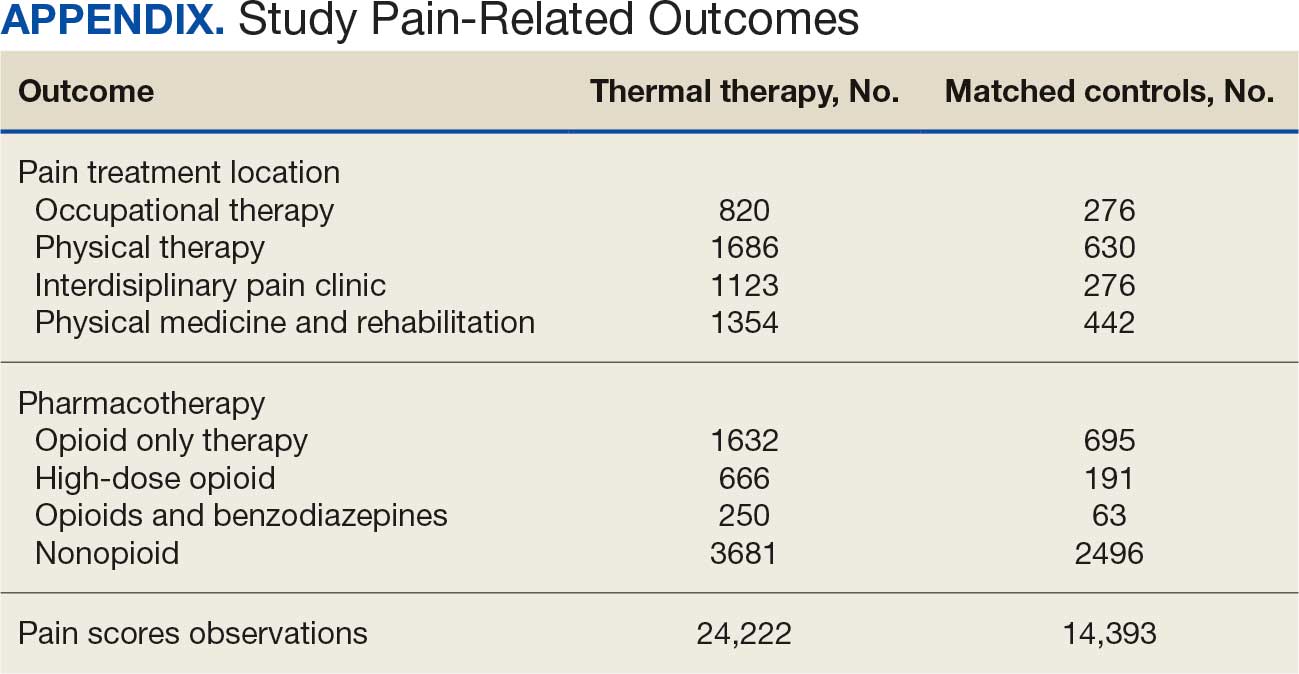
This study used data available in the EHR over the 24-month observation period. We characterize the density (eg, frequency) of all outcome variables by treatment condition in the Appendix. Because the hypotheses were directional, the authors used a 2-sided α = 0.10 and applied a Bonferroni correction for the 9 statistical tests performed, resulting in an adjusted α of 0.01. Treatment utilization and pharmacotherapy outcome analyses were performed in Stata, version 16.1. Random-effects growth modeling of pain score trajectories was performed using Mplus, version 8.8.
Results
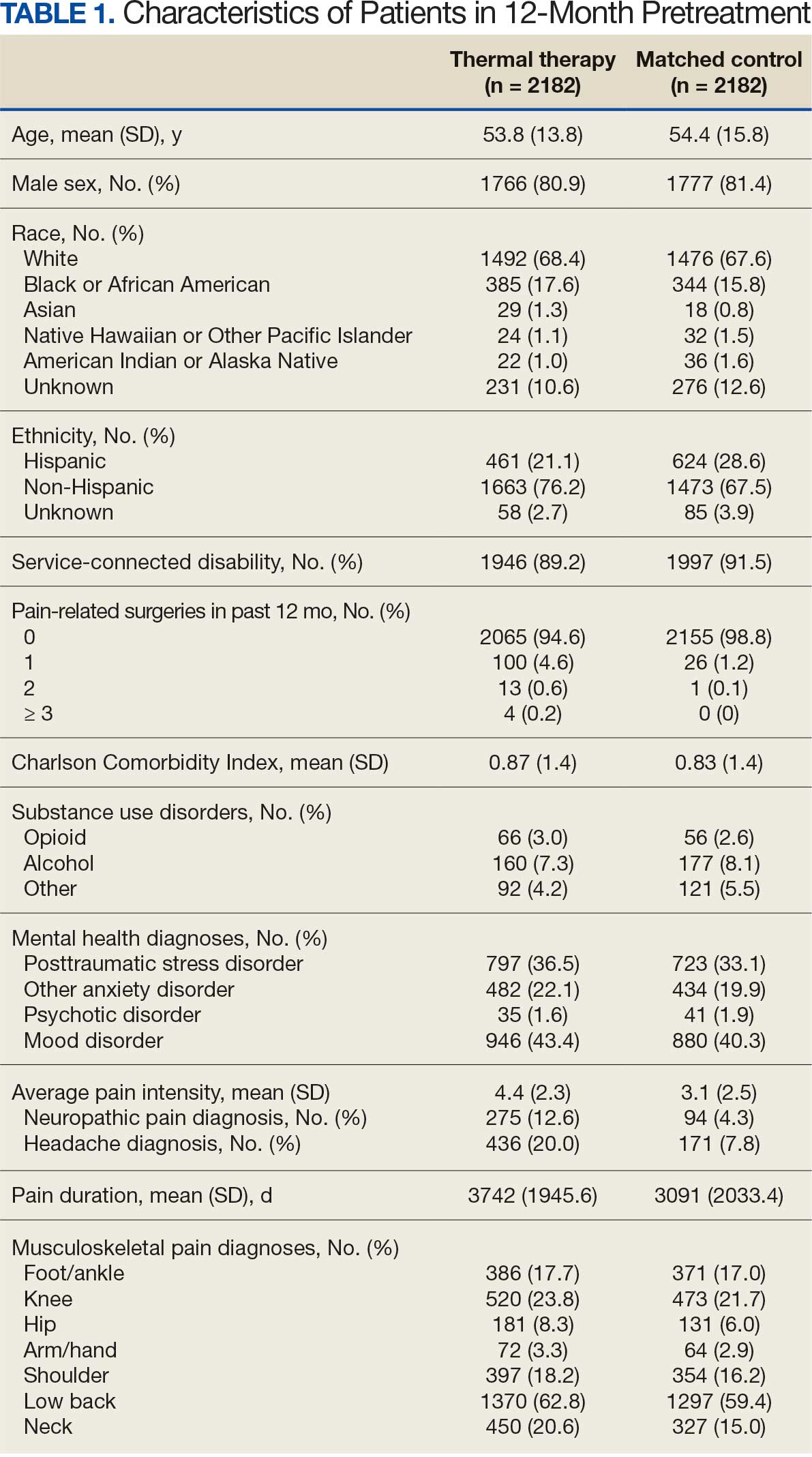
There were 2182 patients in the treatment group and 2182 matched controls. The mean (SD) age was 54 (15) years; 81% were male, and about two-thirds (68%) identified as White and non-Hispanic. Mental health comorbidities were common, with > 40% of the sample having diagnoses of a mood disorder and/or posttraumatic stress disorder. Nearly all patients (90%) had VA service-connected disability ratings. Among patients with chronic musculoskeletal pain, 8% had comorbid neuropathic pain and 14% had headache. The mean (SD) duration of chronic pain across both patient groups was 3416 (2016) days, or about 9.4 years (Table 1).
Pharmacotherapy
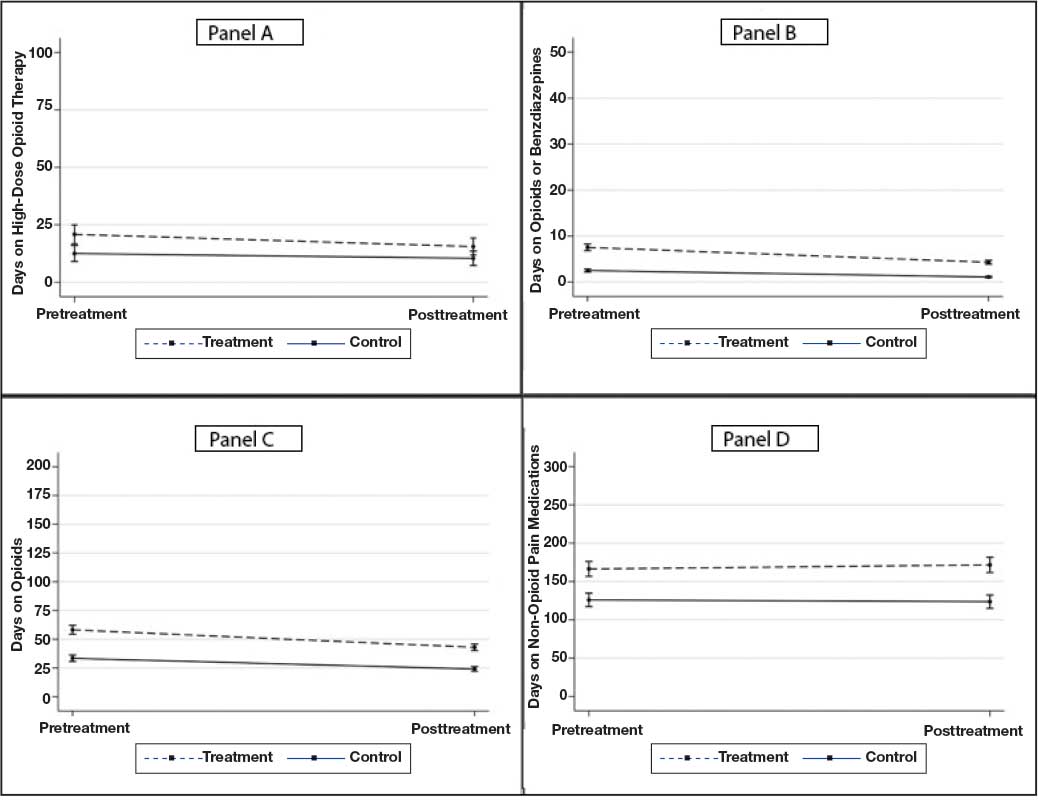
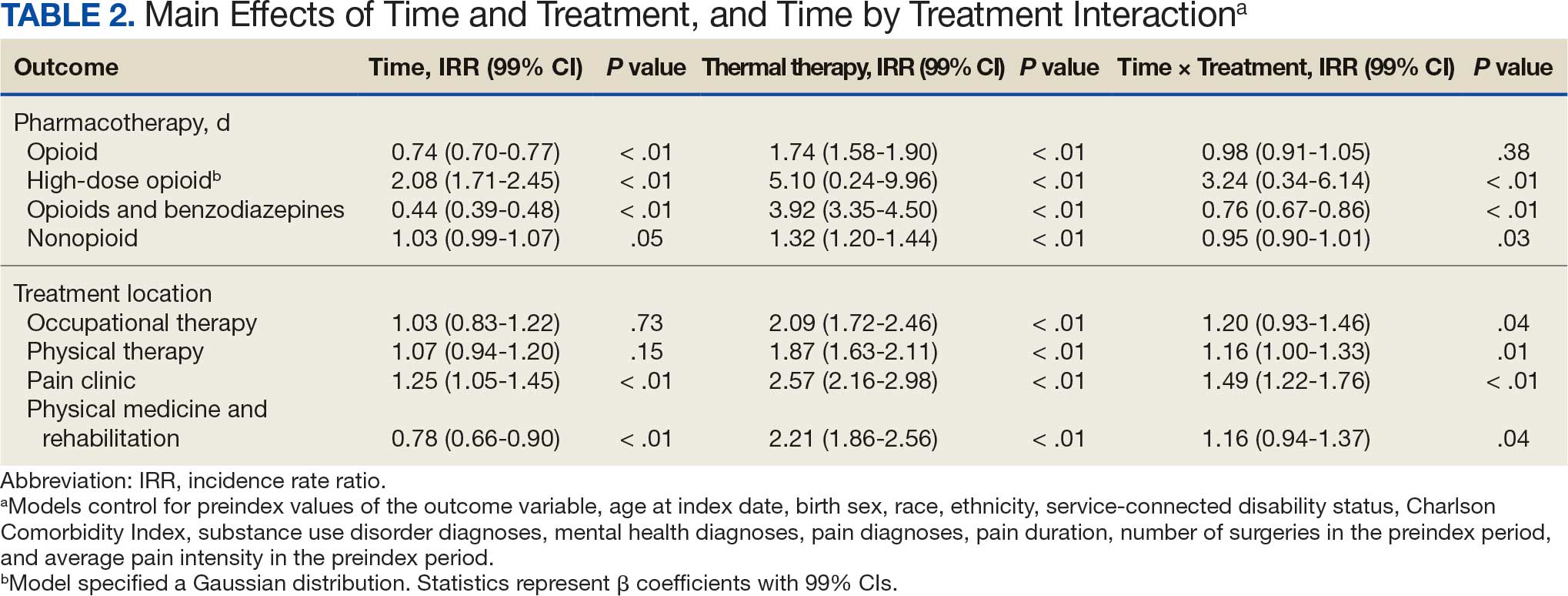
High-dose opioid use (> 50 MME daily dose), days of opioid use, and concurrent opioid and benzodiazepine use decreased for all patients from the pre- to posttreatment period (Table 2). However, high-dose opioid use (Time × Treatment interaction, 3.24; 99% CI, 0.34 to 6.14) (Figure 1A) and concurrent opioid and benzodiazepine use (Time × Treatment interaction, 0.76; 99% CI, 0.67 to 0.86) (Figure 1B) had a larger decrease for the treatment vs matched control group. Treatment and matched control patients had comparable reductions in days of opioid use (Time × Treatment interaction, 0.98; 99% CI, 0.91 to 1.05) (Figure 1C). Neither group showed changes in nonopioid analgesic pharmacotherapy over time (main effect of time, incidence rate ratio [IRR], 1.03; 99% CI, 0.99 to 1.07; Time × Treatment interaction, 0.95; 99% CI, 0.90 to 1.01) (Figure 1D).
Nonpharmacologic Pain Treatment
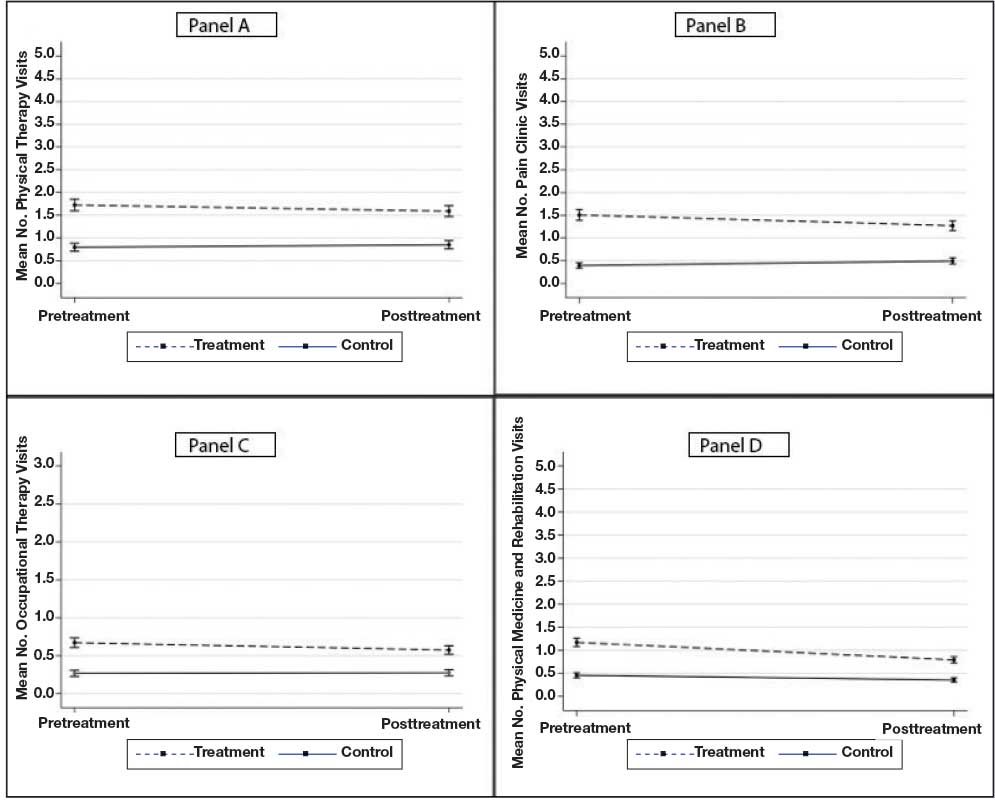
The number of physical therapy and pain clinic visits declined for treatment patients and increased slightly for matched control patients (Figure 2A and 2B). For occupational therapy visits, neither group showed changes over time (main effect of time IRR, 1.03; 99% CI, 0.83 to 1.22; Time × Treatment interaction, 1.20; 99% CI, 0.93 to 1.46) (Figure 2C). For physical medicine and rehabilitation visits, both groups decreased use over time (main effect of time IRR, 0.78; 99% CI, 0.66 to 0.90), but this decrease did not differ between treatment and control patients (Time × Treatment interaction, 1.16; 99% CI, 0.94 to 1.37) (Figure 2D).
Pain Intensity
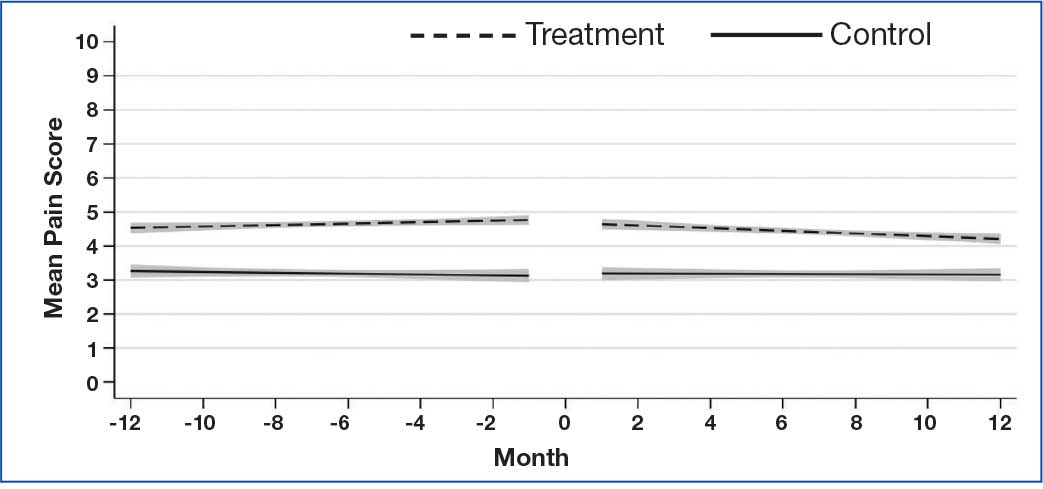
Pain intensity decreased for both groups by an estimated 0.02 points per month (99% CI, -0.04 to -0.01; P < .01), or 0.24 points over the 12-month postindex follow-up period (Figure 3). There were no statistically significant differences between treatment and control patients in pain intensity changes over the follow-up period (B = -0.02; 99% CI, -0.06 to 0.02; P = .15).
Discussion
Patients with musculoskeletal pain who received a thermal therapy device showed a larger decline in use of some specialty pain treatments, including physical therapy and specialty pain clinic services, when compared with matched control patients. One possible explanation is that patients who used the device may have had their pain adequately managed and thus required less specialty pain care. However, the absence of clinically significant changes in pain intensity over time suggests that pain intensity alone did not account for the observed changes in pain treatment use. We were unable to measure constructs of pain interference or functioning, which may be better predictors of functional restoration, as these data are not routinely collected within the VA and documented in the EHR. Future studies could clarify these findings by including measures of pain interference and functioning.
Although the overall declines in physical therapy and specialty pain clinic use associated with thermal therapy were modest (< 1 visit per patient), the impact of these reductions can be profound. In resource-limited health care settings, even small reductions in high-cost care utilization could be of great value in that health systems could offset costs associated with treating chronic pain without compromising quality of care or key clinical outcomes, such as pain intensity. This study, however, did not include a cost analysis. Future studies should incorporate formal cost analyses to quantify cost offsets that may result from decreased specialty pain treatment use.
Patients who received thermal therapy devices did not show clinically meaningful reductions in pain intensity over time, defined as reductions in pain intensity of 1.7 to 2.0 on a 0 to 10 scale.28,29 This finding is consistent with prior research that demonstrates relatively stable pain intensity self-ratings longitudinally by patients with chronic pain diagnoses, when assessed in the context of usual clinical care.30 This finding, however, is inconsistent with prior literature that demonstrates pain-reducing benefits of thermal therapy for low back pain and knee osteoarthritis.7-9
In this study, pain intensity ratings were derived from the EHR during routine outpatient clinical encounters and not at the time thermal therapy was self-administered, as has been done in prior clinical trials.7-9 Pain location was not specified at the time of pain ratings, and it is possible that patients may have been endorsing pain in areas of the body that had not been treated by thermal therapy. Patient-level variability in pain intensity ratings (eg, within-patient range over time) was not examined, although prior research indicates substantial variability.30 While average pain intensity ratings in the current study did not change, an examination of patient variability warrants further study, as a narrowing of pain intensity ratings can be perceived, by patients, as demonstrable improvement and has been associated with improved physical and psychological outcomes.31 Furthermore, pain intensity does not characterize physical or emotional functioning that can be captured with more comprehensive validated measures, some of which are recommended outcomes in pain clinical trials.32
Our findings point to reductions in all forms of opioid use across both treatment and control patients. Data from the VA and in the US more generally point to downward trends in opioid prescribing during the study period.33 This decline is likely due to increased use of risk mitigation approaches, such as routine urine drug screens and review of prescription drug monitoring databases.34 These state-level databases track prescribing of controlled substances, including opioids and benzodiazepines, within a state. Implementation of these practices has been associated with declines in higher risk opioid prescribing.34 Findings from this study further point to associations of reduced higher risk opioid use among patients who received thermal therapy devices. In the full sample of patients, reductions in days of opioid use, high-dose opioid use, and co-use of opioids and benzodiazepines were observed across all patients, with greater reductions observed in high-dose opioid use and concurrent opioid and benzodiazepine use among patients who received a thermal therapy device. Experimental studies indicate that the endogenous opioid system is implicated in pain relief and activated by thermal therapies. 35 Differential reductions in higher-risk opioid use among patients who received thermal therapy devices in our study may be associated with endogenous opioid activation, though this was not specifically measured. It is also unclear whether thermal therapy was provided by clinicians in the context of opioid tapering or other risk mitigation efforts, or patients reduced higher risk opioid use of their own volition. Prior research has identified both patient- and clinician-initiated opioid tapering and discontinuation.36 While a thorough explication of opioid dose reduction was beyond the scope of this study, future qualitative work could help explain potential benefits of thermal therapy in the context of analgesic pharmacotherapy use, including opioid medications.
Limitations
The extent to which patients used the thermal therapy device could not be measured; therefore, device receipt was used as a proxy for use. However, it was not possible to determine whether the frequency and duration of device use was associated with study outcomes. Treatment and control groups demonstrated some differences in outcome variables at the index date. Potential known biases were addressed using propensity score matching procedures and statistical procedures that controlled for patient demographic and clinical characteristics, as well as pretreatment values of all outcome variables. Nevertheless, as an observational study, this analysis cannot account for all known and unknown confounders, and a randomized controlled trial is needed to make claims of causality. The study population consisted of US veterans and included a low proportion of women. As a result, the findings may not be generalizable to other patient populations. Finally, prescription dispensing data, used as a proxy for pharmacotherapy use, do not necessarily reflect actual medication use.
Conclusions
This study is among the first to examine associations between thermal therapy and specialty pain treatment and analgesic pharmacotherapy use among US veterans. Although the retrospective cohort study design does not allow causal inferences regarding the efficacy of thermal therapy for veterans with chronic musculoskeletal pain, confidence in the findings is strengthened by methodological and statistical control of known confounders. Future trials employing experimental designs are needed to further clarify the clinical and health systems benefits of thermal therapy for musculoskeletal pain syndromes.
- Krebs EE, Gravely A, Nugent S, et al. Effect of opioid vs nonopioid medications on pain-related function in patients with chronic back pain or hip or knee osteoarthritis pain: the SPACE randomized clinical trial. JAMA. 2018;2018:872-882. doi:10.1001/jama.2018.0899
- Becker WC, DeBar LL, Heapy AA, et al. A research agenda for advancing non-pharmacological management of chronic musculoskeletal pain: findings from a VHA state-of-the-art conference. J Gen Intern Med. 2018;33:1-15. doi:10.1007/s11606-018-4345-6
- Fernandes IA, Armond ACV, Falci SGM. The effectiveness of the cold therapy (cryotherapy) in the management of inflammatory parameters after removal of mandibular third molars: a meta-analysis. Int Arch Otorhinolaryngol. 2019;23:221-228. doi:10.1055/s-0039-1677755
- Quinlan P, Davis J, Fields K, et al. Effects of localized cold therapy on pain in postoperative spinal fusion patients: a randomized controlled trial. Orthop Nurs. 2017;36:344-349. doi:10.1097/NOR.0000000000000382
- Malanga GA, Yan N, Stark J. Mechanisms and efficacy of heat and cold therapies for musculoskeletal injury. Postgrad Med. 2015;127:57-65. doi:10.1080/00325481.2015.992719
- Wang Y, Li S, Zhang Y, et al. Heat and cold therapy reduce pain in patients with delayed onset muscle soreness: a systematic review and meta-analysis of 32 randomized controlled trials. Phys Ther Sport. 2021;48:177-187. doi:10.1016/j.ptsp.2021.01.004
- Ariana M, Afrasiabifar A, Doulatabad SN, et al. The effect of local heat therapy versus cold rub gel on pain and joint functions in patients with knee osteoarthritis. Clin Nurs Res. 2022;31:1014-1022. doi:10.1177/10547738211035502
- French SD, Cameron M, Walker BF, et al. Superficial heat or cold for low back pain. Cochrane Database Syst Rev. 2006;1:CD004750. doi:10.1002/14651858.CD004750.pub2
- Tao XG, Bernacki EJ. A randomized clinical trial of continuous low-level heat therapy for acute muscular low back pain in the workplace. J Occup Environ Med. 2005;47:1298- 1306. doi:10.1097/01.jom.0000184877.01691.a3
- Freiwald J, Hoppe MW, Beermann W, et al. Effects of supplemental heat therapy in multimodal treated chronic low back pain patients on strength and flexibility. Clin Biomech. 2018;57:107-113. doi:10.1016/j.clinbiomech.2018.06.008
- Freiwald J, Magni A, Fanlo-Mazas P, et al. A role for superficial heat therapy in the management of nonspecific, mild-to-moderate low back pain in current clinical practice: a narrative review. Life. 2021;11:780. doi:10.3390/life11080780
- Kerns RD, Philip EJ, Lee AW, et al. Implementation of the Veterans Health Administration National Pain Management Strategy. Transl Behav Med. 2011;1:635-643. doi:10.1007/s13142-011-0094-3
- Brookhart MA, Schneeweiss S, Rothman KJ, et al. Variable selection for propensity score models. Am J Epidemiol. 2006;163:1149-1156. doi:10.1093/aje/kwj149
- Edlund MJ, Martin BC, Devries A, et al. Trends in use of opioids for chronic non-cancer pain among individuals with mental health and substance use disorders: the TROUP study. Clin J Pain. 2010;26:1-8. doi:10.1097/AJP.0b013e3181b99f35
- Kaur S, Stechuchak KM, Coffman CJ, et al. Gender differences in health care utilization among veterans with chronic pain. J Gen Intern Med. 2007;22:228-233. doi:10.1007/s11606-006-0048-5
- Meghani SH, Cho E. Self-reported pain and utilization of pain treatment between minorities and non-minorities in the United States. Public Health Nurs. 2009;26:307-316. doi:10.1111/j.1525-1446.2009.00785.x
- Morasco BJ, Duckart JP, Carr TP, et al. Clinical characteristics of veterans prescribed high doses of opioid medications for chronic non-cancer pain. Pain. 2010;151:625-632. doi:10.1016/j.pain.2010.08.002
- Nielsen S, Lintzeris N, Bruno R, et al. Benzodiazepine use among chronic pain patients prescribed opioids: associations with pain, physical and mental health, and health service utilization. Pain Med. 2015;16:356-366. doi:10.1111/pme.12594
- Quinn PD, Hur K, Chang Z, et al. Incident and long-term opioid therapy among patients with psychiatric conditions and medications: a national study of commercial health care claims. Pain. 2017;158:140-148. doi:10.1097/j.pain.0000000000000730
- Rubin DB. Matching to remove bias in observational studies. Biometrics. 1973;29:159-183.
- Rosenbaum PR, Rubin DB. Constructing a control group using multivariate matched sampling methods that incorporate the propensity score. Am Stat. 1985;39:33-38. doi:10.1007/s11596-025-00052-0
- Steiner PM, Cook TD, Shadish WR, et al. The importance of covariate selection in controlling for selection bias in observational studies. Psychol Methods. 2010;15:250-267. doi:10.1037/a0018719
- Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373- 383. doi:10.1016/0021-9681(87)90171-8
- Long JS, Freese J. Regression Models for Categorical Dependent Variables Using Stata. 3rd ed. Stata Press; 2014.
- Muthén LK, Muthén BO. Mplus User’s Guide. 8th ed. Muthén & Muthén; 1998-2017.
- Hedeker D, Gibbons RD. Longitudinal Data Analysis. Wiley; 2006.
- Dobscha SK, Morasco BJ, Kovas AE, et al. Short-term variability in outpatient pain intensity scores in a national sample of older veterans with chronic pain. Pain Med. 2015;16:855-865. doi:10.1111/pme.12643
- Bahreini M, Safaie A, Mirfazaelian H, et al. How much change in pain score does really matter to patients? Am J Emerg Med. 2020;38:1641-1646. doi:10.1016/j.ajem.2019.158489
- Suzuki H, Aono S, Inoue S, et al. Clinically significant changes in pain along the pain intensity numerical rating scale in patients with chronic low back pain. PLoS One. 2020;15:e0229228. doi:10.1371/journal.pone.0229228
- McPherson SM, Smith CL, Dobscha SK, et al. Changes in pain intensity after discontinuation of long-term opioid therapy for chronic noncancer pain. Pain. 2018;159:2097- 2104. doi:10.1097/j.pain.0000000000001315
- Andrews NE, Strong J, Meredith PJ. Activity pacing, avoidance, endurance, and associations with patient functioning in chronic pain: a systematic and meta-analysis. Arch Phys Med Rehabil. 2012;93:2109-121. doi:10.1016/j.apmr.2012.05.029
- Dworkin RH, Turk DC, Farrar JT, et al. Core outcome measures for chronic pain clinical trials: IMMPACT recommendations. Pain. 2005;113:9-19. doi:10.1016/j.pain.2004.09.012
- Gellad WF, Good CB, Shulkin DJ. Addressing the opioid epidemic in the United States: Lessons from the Department of Veterans Affairs. JAMA Intern Med. 2017;177:611- 612. doi:10.1001/jamainternmed.2017.0147
- Lin LA, Bohnert ASB, Kerns RD, et al. Impact of the Opioid Safety Initiative on opioid-related prescribing in veterans. Pain. 2017;158:833-839. doi:10.1097/j.pain.0000000000000837
- Sirucek L, Price RC, Gandhi W, et al. Endogenous opioids contribute to the feeling of pain relief in humans. Pain. 2021;162:2821-2831. doi:10.1097/j.pain.0000000000002285
- Lovejoy TI, Morasco BJ, Demidenko MI, et al. Reasons for discontinuation of long-term opioid therapy in patients with and without substance use disorders. Pain. 2017;158:526- 534. doi:10.1097/j.pain.0000000000000796
Growing recognition of the limitations of long-term opioid therapy for chronic noncancer pain has highlighted the importance of nonpharmacologic approaches to pain treatment.1,2 These treatments are varied and may include psychological and behavioral therapies (eg, cognitive behavioral therapy for chronic pain), exercise and movement therapies (eg, yoga), and manual therapies (eg, chiropractic). Body surface cold therapy, while predominantly used to reduce postoperative pain and inflammation,3,4 pain subsequent to acute musculoskeletal injury,5 and pain related to muscle soreness,6 is a nonpharmacologic treatment that has shown pain-reducing benefits for chronic low back pain and knee osteoarthritis, as has its counterpart, superficial heat therapy.7-9 Heat therapy has also been shown to improve strength, flexibility, and activities of daily living in patients with chronic low back pain.10,11 Cold and heat therapies are commonly used complementarily. Cold therapies aim to reduce blood flow and inflammation and are often used immediately following trauma to an affected area, whereas heat therapies increase blood flow and metabolic activity and are commonly used to promote healing.5
Heat and cold therapies (also known as thermal therapies) benefit resource-limited health care systems, as most devices require a single expenditure and can be self-administered by patients at home as part of their pain self-management plan. In addition, these pain self-management tools may attenuate the need for more expensive specialty pain care and ongoing analgesic pharmacotherapy. Despite their potential, few studies have characterized the benefits of thermal therapies for patients with heterogeneous chronic pain syndromes.
The purpose of this retrospective cohort study was to examine the potential clinical and health system benefits of patient-administered thermal therapy. Our primary hypothesis was that patients with chronic musculoskeletal pain who received a thermal therapy self-management device would have fewer days of opioid prescriptions compared with a sample of matched control patients. Secondarily, we hypothesized that patients who received a thermal therapy device would have lower utilization of specialty pain care, fewer potentially hazardous opioid prescriptions (eg, high-dose opioid therapy and concurrent opioid and benzodiazepine prescriptions), fewer prescriptions for nonopioid analgesic medications, and decreased pain intensity when compared with matched controls.
Methods
This retrospective cohort study compared pain pharmacotherapy, pain treatment utilization, and pain intensity outcomes between patients who received a thermal therapy device and matched patients who did not. The study was approved by the US Department of Veterans Affairs (VA) Portland Health Care System Institutional Review Board and was granted a waiver of informed consent to access patient electronic health records (EHRs).
Pain Care
The VA uses ThermaZone thermal therapy devices (Innovative Medical Equipment) for chronic pain treatment. The device uses thermoelectric technology to provide point-of-contact cooling and heating therapy through site-specific pads (eg, ankle, knee, hip, back, elbow, shoulder). Patients place pads on sites where they experience pain, and temperature regulated water circulates through the device and to the pad, providing consistent, localized thermal therapy. The pads range in temperature from 1 °C to 52 °C, and temperatures are self-monitored and controlled by the patient.
Standard pain care in this study followed the VA stepped model of pain care, which builds on a foundation of patient education for pain self-management approaches (eg, exercise, mindfulness, relaxation, social support).12 According to the VA stepped model of pain care, all patients with chronic pain should engage in these foundational self-management approaches. However, some patients require more intensive care. The VA stepped-up treatment engages primary and specialty care services—such as physical therapy, pharmacy, complementary and integrative health approaches, mental health, and substance use services—and, when indicated, may escalate treatment to interdisciplinary pain teams or tertiary pain centers. In this retrospective cohort study, treatment patients received a thermal therapy device and standard of care, whereas control patients received standard care only.
Sample Selection
Eligible patients were aged ≥ 18 years, had a musculoskeletal pain diagnosis documented in the EHR in the year prior to thermal therapy device receipt (or during the same period for a treatment patient’s matched control), and were enrolled in VA health care during the study period. Patients who died during the study period were excluded. Treatment patients received a thermal therapy device from the VA between January 1, 2017, and December 31, 2018, when complete data on thermal therapy devices were available. For control patients, the VA Corporate Data Warehouse (CDW) was used to identify VA patients with characteristics similar to those of treated patients.
We modeled the probability that a patient would receive a thermal therapy device using logistic regression. Predictor variables were measured in the year prior to device receipt and included variables associated with pain treatment utilization and analgesic pharmacotherapy receipt, as recommended by Brookhart et al.13 These included age, sex, race, ethnicity, VA service-connected disability status, comorbidities, receipt of medications for opioid use disorder, pain diagnoses, mental health diagnoses, and substance use disorder diagnoses.14-19
The resulting propensity scores (eg, predicted probabilities) were used to match treatment patients 1:1 with control patients using a nearest-neighbor matching algorithm.20 This procedure matches a control patient with the closest propensity score to that of the corresponding treatment patient. An exact match on VA facility was required (eg, treatment patients and matched controls received care at the same VA facility). Standardized differences were used to assess covariate balance between the matched groups, and kernel density plots of propensity scores tested for sufficient overlap.21 Control patients were selected from a pool of 1,150,149 patients.
Study Variables
The index date was the date the thermal therapy device was released to treatment patients or the same date for the matched controls. Data were extracted from the CDW over a 24-month period: 12 months prior to the index date through 12 months afterwards. Collecting data in the 12 months prior to treatment initiation allowed us to adjust for covariates and provided greater precision, as recommended for observational study designs.22
Treatment conditions were defined dichotomously as receipt vs nonreceipt of a thermal therapy device. The primary outcome was the number of days of opioid use in the 12 months following the index date. Additional outcomes included days of high-dose opioid therapy (≥ 50 mg morphine equivalent [MME] daily), concurrent opioid and benzodiazepine prescriptions, and nonopioid analgesic pharmacotherapy (eg, nonsteroidal anti-inflammatory drugs, acetaminophen, muscle relaxants). All prescription data were based on medication fills obtained from VA pharmacy records. Additional outcomes included the number of visits to physical therapy, occupational therapy, physical medicine and rehabilitation, and interdisciplinary pain clinics, including interventional pain medicine.
Pain intensity ratings were collected as part of routine VA care using a numeric scale from 0 (no pain) to 10 (worst possible pain). Pain intensity trajectories were computed using all available EHR-derived pain intensity score data for each patient in the 12 months prior to and following the index date.
Covariates were extracted from the EHR and evaluated in the year prior to the index date, unless otherwise noted. They included age at the index date; self-reported sex, and race and ethnicity; service-connected disability status (disability awarded as a result of military service-related trauma or injury); Charlson Comorbidity Index; and diagnoses of opioid use disorder, alcohol use disorder, other substance use disorder, mood disorder, posttraumatic stress disorder, other anxiety disorder, psychotic disorder, neuropathic pain, and headache pain.23 All diagnoses were coded as yes if identified in the EHR as a focus of treatment during any clinical encounter in the year prior to the index date or no if not identified as a focus of treatment.
The number of days patients had been living with pain was calculated and defined as the number of days from the first pain diagnosis available in a patient’s EHR to the index date. Finally, the number of pain-related surgeries (eg, surgeries with ≥ 1 pain diagnoses associated with the clinical encounter) and average pain intensity were computed for the pre- and postindex date evaluation periods and included as model covariates.
Statistical Analyses
For the 4 pharmacotherapy and 4 nonpharmacologic treatment usage outcomes, we tested the fit of linear models and several models with count distributions using the Bayesian information criterion.24 Count distributions included Poisson, zero-inflated Poisson, negative binomial, and zero-inflated negative binomial. With 1 exception (days of high-dose opioid use), a negative binomial distribution best fit the data. For days of high-dose opioid use, a Gaussian distribution best fit the data.
Eight separate mixed-effects regression analyses compared changes in each outcome from the 12-month preindex period through the 12-month postindex period between treatment and control patients by testing the Time × Treatment interaction. This approach statistically accounts for observed pretreatment differences in outcome variables. Statistics for the main effects of time and treatment are also presented. To reduce bias, models controlled for covariates specified previously.
For pain intensity, we used random-effects growth modeling to quantify both fixed and random effects of pain intensity at the index date (eg, the model intercept), which estimates pain at the time of treatment initiation, and change in pain during the 12 months following the index date (eg, the model slope), which characterizes the trajectory of pain intensity ratings.25 The model included piecewise components of pain score trajectories in the 12 months prior to the index date and in the 12 months following the index date. Several types of change for the 12-month postindex observation period were explored—including quadratic and cubic curvilinear change. A linear model for change in pain over time provided the best fit based on the Bayesian information criterion and parsimony of model parameters.26 We report estimates of change in pain over time in monthly intervals for ease of interpretation. However, models used all individual pain scores rather than computing monthly averages when > 1 pain score was available within a month, as recommended in previous research.27 This approach makes optimal use of all available data. Both random effects (intercept and slope) were regressed onto the set of covariates described previously.
This study used data available in the EHR over the 24-month observation period. We characterize the density (eg, frequency) of all outcome variables by treatment condition in the Appendix. Because the hypotheses were directional, the authors used a 2-sided α = 0.10 and applied a Bonferroni correction for the 9 statistical tests performed, resulting in an adjusted α of 0.01. Treatment utilization and pharmacotherapy outcome analyses were performed in Stata, version 16.1. Random-effects growth modeling of pain score trajectories was performed using Mplus, version 8.8.

Results
There were 2182 patients in the treatment group and 2182 matched controls. The mean (SD) age was 54 (15) years; 81% were male, and about two-thirds (68%) identified as White and non-Hispanic. Mental health comorbidities were common, with > 40% of the sample having diagnoses of a mood disorder and/or posttraumatic stress disorder. Nearly all patients (90%) had VA service-connected disability ratings. Among patients with chronic musculoskeletal pain, 8% had comorbid neuropathic pain and 14% had headache. The mean (SD) duration of chronic pain across both patient groups was 3416 (2016) days, or about 9.4 years (Table 1).

Pharmacotherapy
High-dose opioid use (> 50 MME daily dose), days of opioid use, and concurrent opioid and benzodiazepine use decreased for all patients from the pre- to posttreatment period (Table 2). However, high-dose opioid use (Time × Treatment interaction, 3.24; 99% CI, 0.34 to 6.14) (Figure 1A) and concurrent opioid and benzodiazepine use (Time × Treatment interaction, 0.76; 99% CI, 0.67 to 0.86) (Figure 1B) had a larger decrease for the treatment vs matched control group. Treatment and matched control patients had comparable reductions in days of opioid use (Time × Treatment interaction, 0.98; 99% CI, 0.91 to 1.05) (Figure 1C). Neither group showed changes in nonopioid analgesic pharmacotherapy over time (main effect of time, incidence rate ratio [IRR], 1.03; 99% CI, 0.99 to 1.07; Time × Treatment interaction, 0.95; 99% CI, 0.90 to 1.01) (Figure 1D).


Nonpharmacologic Pain Treatment
The number of physical therapy and pain clinic visits declined for treatment patients and increased slightly for matched control patients (Figure 2A and 2B). For occupational therapy visits, neither group showed changes over time (main effect of time IRR, 1.03; 99% CI, 0.83 to 1.22; Time × Treatment interaction, 1.20; 99% CI, 0.93 to 1.46) (Figure 2C). For physical medicine and rehabilitation visits, both groups decreased use over time (main effect of time IRR, 0.78; 99% CI, 0.66 to 0.90), but this decrease did not differ between treatment and control patients (Time × Treatment interaction, 1.16; 99% CI, 0.94 to 1.37) (Figure 2D).

Pain Intensity
Pain intensity decreased for both groups by an estimated 0.02 points per month (99% CI, -0.04 to -0.01; P < .01), or 0.24 points over the 12-month postindex follow-up period (Figure 3). There were no statistically significant differences between treatment and control patients in pain intensity changes over the follow-up period (B = -0.02; 99% CI, -0.06 to 0.02; P = .15).

Discussion
Patients with musculoskeletal pain who received a thermal therapy device showed a larger decline in use of some specialty pain treatments, including physical therapy and specialty pain clinic services, when compared with matched control patients. One possible explanation is that patients who used the device may have had their pain adequately managed and thus required less specialty pain care. However, the absence of clinically significant changes in pain intensity over time suggests that pain intensity alone did not account for the observed changes in pain treatment use. We were unable to measure constructs of pain interference or functioning, which may be better predictors of functional restoration, as these data are not routinely collected within the VA and documented in the EHR. Future studies could clarify these findings by including measures of pain interference and functioning.
Although the overall declines in physical therapy and specialty pain clinic use associated with thermal therapy were modest (< 1 visit per patient), the impact of these reductions can be profound. In resource-limited health care settings, even small reductions in high-cost care utilization could be of great value in that health systems could offset costs associated with treating chronic pain without compromising quality of care or key clinical outcomes, such as pain intensity. This study, however, did not include a cost analysis. Future studies should incorporate formal cost analyses to quantify cost offsets that may result from decreased specialty pain treatment use.
Patients who received thermal therapy devices did not show clinically meaningful reductions in pain intensity over time, defined as reductions in pain intensity of 1.7 to 2.0 on a 0 to 10 scale.28,29 This finding is consistent with prior research that demonstrates relatively stable pain intensity self-ratings longitudinally by patients with chronic pain diagnoses, when assessed in the context of usual clinical care.30 This finding, however, is inconsistent with prior literature that demonstrates pain-reducing benefits of thermal therapy for low back pain and knee osteoarthritis.7-9
In this study, pain intensity ratings were derived from the EHR during routine outpatient clinical encounters and not at the time thermal therapy was self-administered, as has been done in prior clinical trials.7-9 Pain location was not specified at the time of pain ratings, and it is possible that patients may have been endorsing pain in areas of the body that had not been treated by thermal therapy. Patient-level variability in pain intensity ratings (eg, within-patient range over time) was not examined, although prior research indicates substantial variability.30 While average pain intensity ratings in the current study did not change, an examination of patient variability warrants further study, as a narrowing of pain intensity ratings can be perceived, by patients, as demonstrable improvement and has been associated with improved physical and psychological outcomes.31 Furthermore, pain intensity does not characterize physical or emotional functioning that can be captured with more comprehensive validated measures, some of which are recommended outcomes in pain clinical trials.32
Our findings point to reductions in all forms of opioid use across both treatment and control patients. Data from the VA and in the US more generally point to downward trends in opioid prescribing during the study period.33 This decline is likely due to increased use of risk mitigation approaches, such as routine urine drug screens and review of prescription drug monitoring databases.34 These state-level databases track prescribing of controlled substances, including opioids and benzodiazepines, within a state. Implementation of these practices has been associated with declines in higher risk opioid prescribing.34 Findings from this study further point to associations of reduced higher risk opioid use among patients who received thermal therapy devices. In the full sample of patients, reductions in days of opioid use, high-dose opioid use, and co-use of opioids and benzodiazepines were observed across all patients, with greater reductions observed in high-dose opioid use and concurrent opioid and benzodiazepine use among patients who received a thermal therapy device. Experimental studies indicate that the endogenous opioid system is implicated in pain relief and activated by thermal therapies. 35 Differential reductions in higher-risk opioid use among patients who received thermal therapy devices in our study may be associated with endogenous opioid activation, though this was not specifically measured. It is also unclear whether thermal therapy was provided by clinicians in the context of opioid tapering or other risk mitigation efforts, or patients reduced higher risk opioid use of their own volition. Prior research has identified both patient- and clinician-initiated opioid tapering and discontinuation.36 While a thorough explication of opioid dose reduction was beyond the scope of this study, future qualitative work could help explain potential benefits of thermal therapy in the context of analgesic pharmacotherapy use, including opioid medications.
Limitations
The extent to which patients used the thermal therapy device could not be measured; therefore, device receipt was used as a proxy for use. However, it was not possible to determine whether the frequency and duration of device use was associated with study outcomes. Treatment and control groups demonstrated some differences in outcome variables at the index date. Potential known biases were addressed using propensity score matching procedures and statistical procedures that controlled for patient demographic and clinical characteristics, as well as pretreatment values of all outcome variables. Nevertheless, as an observational study, this analysis cannot account for all known and unknown confounders, and a randomized controlled trial is needed to make claims of causality. The study population consisted of US veterans and included a low proportion of women. As a result, the findings may not be generalizable to other patient populations. Finally, prescription dispensing data, used as a proxy for pharmacotherapy use, do not necessarily reflect actual medication use.
Conclusions
This study is among the first to examine associations between thermal therapy and specialty pain treatment and analgesic pharmacotherapy use among US veterans. Although the retrospective cohort study design does not allow causal inferences regarding the efficacy of thermal therapy for veterans with chronic musculoskeletal pain, confidence in the findings is strengthened by methodological and statistical control of known confounders. Future trials employing experimental designs are needed to further clarify the clinical and health systems benefits of thermal therapy for musculoskeletal pain syndromes.
Growing recognition of the limitations of long-term opioid therapy for chronic noncancer pain has highlighted the importance of nonpharmacologic approaches to pain treatment.1,2 These treatments are varied and may include psychological and behavioral therapies (eg, cognitive behavioral therapy for chronic pain), exercise and movement therapies (eg, yoga), and manual therapies (eg, chiropractic). Body surface cold therapy, while predominantly used to reduce postoperative pain and inflammation,3,4 pain subsequent to acute musculoskeletal injury,5 and pain related to muscle soreness,6 is a nonpharmacologic treatment that has shown pain-reducing benefits for chronic low back pain and knee osteoarthritis, as has its counterpart, superficial heat therapy.7-9 Heat therapy has also been shown to improve strength, flexibility, and activities of daily living in patients with chronic low back pain.10,11 Cold and heat therapies are commonly used complementarily. Cold therapies aim to reduce blood flow and inflammation and are often used immediately following trauma to an affected area, whereas heat therapies increase blood flow and metabolic activity and are commonly used to promote healing.5
Heat and cold therapies (also known as thermal therapies) benefit resource-limited health care systems, as most devices require a single expenditure and can be self-administered by patients at home as part of their pain self-management plan. In addition, these pain self-management tools may attenuate the need for more expensive specialty pain care and ongoing analgesic pharmacotherapy. Despite their potential, few studies have characterized the benefits of thermal therapies for patients with heterogeneous chronic pain syndromes.
The purpose of this retrospective cohort study was to examine the potential clinical and health system benefits of patient-administered thermal therapy. Our primary hypothesis was that patients with chronic musculoskeletal pain who received a thermal therapy self-management device would have fewer days of opioid prescriptions compared with a sample of matched control patients. Secondarily, we hypothesized that patients who received a thermal therapy device would have lower utilization of specialty pain care, fewer potentially hazardous opioid prescriptions (eg, high-dose opioid therapy and concurrent opioid and benzodiazepine prescriptions), fewer prescriptions for nonopioid analgesic medications, and decreased pain intensity when compared with matched controls.
Methods
This retrospective cohort study compared pain pharmacotherapy, pain treatment utilization, and pain intensity outcomes between patients who received a thermal therapy device and matched patients who did not. The study was approved by the US Department of Veterans Affairs (VA) Portland Health Care System Institutional Review Board and was granted a waiver of informed consent to access patient electronic health records (EHRs).
Pain Care
The VA uses ThermaZone thermal therapy devices (Innovative Medical Equipment) for chronic pain treatment. The device uses thermoelectric technology to provide point-of-contact cooling and heating therapy through site-specific pads (eg, ankle, knee, hip, back, elbow, shoulder). Patients place pads on sites where they experience pain, and temperature regulated water circulates through the device and to the pad, providing consistent, localized thermal therapy. The pads range in temperature from 1 °C to 52 °C, and temperatures are self-monitored and controlled by the patient.
Standard pain care in this study followed the VA stepped model of pain care, which builds on a foundation of patient education for pain self-management approaches (eg, exercise, mindfulness, relaxation, social support).12 According to the VA stepped model of pain care, all patients with chronic pain should engage in these foundational self-management approaches. However, some patients require more intensive care. The VA stepped-up treatment engages primary and specialty care services—such as physical therapy, pharmacy, complementary and integrative health approaches, mental health, and substance use services—and, when indicated, may escalate treatment to interdisciplinary pain teams or tertiary pain centers. In this retrospective cohort study, treatment patients received a thermal therapy device and standard of care, whereas control patients received standard care only.
Sample Selection
Eligible patients were aged ≥ 18 years, had a musculoskeletal pain diagnosis documented in the EHR in the year prior to thermal therapy device receipt (or during the same period for a treatment patient’s matched control), and were enrolled in VA health care during the study period. Patients who died during the study period were excluded. Treatment patients received a thermal therapy device from the VA between January 1, 2017, and December 31, 2018, when complete data on thermal therapy devices were available. For control patients, the VA Corporate Data Warehouse (CDW) was used to identify VA patients with characteristics similar to those of treated patients.
We modeled the probability that a patient would receive a thermal therapy device using logistic regression. Predictor variables were measured in the year prior to device receipt and included variables associated with pain treatment utilization and analgesic pharmacotherapy receipt, as recommended by Brookhart et al.13 These included age, sex, race, ethnicity, VA service-connected disability status, comorbidities, receipt of medications for opioid use disorder, pain diagnoses, mental health diagnoses, and substance use disorder diagnoses.14-19
The resulting propensity scores (eg, predicted probabilities) were used to match treatment patients 1:1 with control patients using a nearest-neighbor matching algorithm.20 This procedure matches a control patient with the closest propensity score to that of the corresponding treatment patient. An exact match on VA facility was required (eg, treatment patients and matched controls received care at the same VA facility). Standardized differences were used to assess covariate balance between the matched groups, and kernel density plots of propensity scores tested for sufficient overlap.21 Control patients were selected from a pool of 1,150,149 patients.
Study Variables
The index date was the date the thermal therapy device was released to treatment patients or the same date for the matched controls. Data were extracted from the CDW over a 24-month period: 12 months prior to the index date through 12 months afterwards. Collecting data in the 12 months prior to treatment initiation allowed us to adjust for covariates and provided greater precision, as recommended for observational study designs.22
Treatment conditions were defined dichotomously as receipt vs nonreceipt of a thermal therapy device. The primary outcome was the number of days of opioid use in the 12 months following the index date. Additional outcomes included days of high-dose opioid therapy (≥ 50 mg morphine equivalent [MME] daily), concurrent opioid and benzodiazepine prescriptions, and nonopioid analgesic pharmacotherapy (eg, nonsteroidal anti-inflammatory drugs, acetaminophen, muscle relaxants). All prescription data were based on medication fills obtained from VA pharmacy records. Additional outcomes included the number of visits to physical therapy, occupational therapy, physical medicine and rehabilitation, and interdisciplinary pain clinics, including interventional pain medicine.
Pain intensity ratings were collected as part of routine VA care using a numeric scale from 0 (no pain) to 10 (worst possible pain). Pain intensity trajectories were computed using all available EHR-derived pain intensity score data for each patient in the 12 months prior to and following the index date.
Covariates were extracted from the EHR and evaluated in the year prior to the index date, unless otherwise noted. They included age at the index date; self-reported sex, and race and ethnicity; service-connected disability status (disability awarded as a result of military service-related trauma or injury); Charlson Comorbidity Index; and diagnoses of opioid use disorder, alcohol use disorder, other substance use disorder, mood disorder, posttraumatic stress disorder, other anxiety disorder, psychotic disorder, neuropathic pain, and headache pain.23 All diagnoses were coded as yes if identified in the EHR as a focus of treatment during any clinical encounter in the year prior to the index date or no if not identified as a focus of treatment.
The number of days patients had been living with pain was calculated and defined as the number of days from the first pain diagnosis available in a patient’s EHR to the index date. Finally, the number of pain-related surgeries (eg, surgeries with ≥ 1 pain diagnoses associated with the clinical encounter) and average pain intensity were computed for the pre- and postindex date evaluation periods and included as model covariates.
Statistical Analyses
For the 4 pharmacotherapy and 4 nonpharmacologic treatment usage outcomes, we tested the fit of linear models and several models with count distributions using the Bayesian information criterion.24 Count distributions included Poisson, zero-inflated Poisson, negative binomial, and zero-inflated negative binomial. With 1 exception (days of high-dose opioid use), a negative binomial distribution best fit the data. For days of high-dose opioid use, a Gaussian distribution best fit the data.
Eight separate mixed-effects regression analyses compared changes in each outcome from the 12-month preindex period through the 12-month postindex period between treatment and control patients by testing the Time × Treatment interaction. This approach statistically accounts for observed pretreatment differences in outcome variables. Statistics for the main effects of time and treatment are also presented. To reduce bias, models controlled for covariates specified previously.
For pain intensity, we used random-effects growth modeling to quantify both fixed and random effects of pain intensity at the index date (eg, the model intercept), which estimates pain at the time of treatment initiation, and change in pain during the 12 months following the index date (eg, the model slope), which characterizes the trajectory of pain intensity ratings.25 The model included piecewise components of pain score trajectories in the 12 months prior to the index date and in the 12 months following the index date. Several types of change for the 12-month postindex observation period were explored—including quadratic and cubic curvilinear change. A linear model for change in pain over time provided the best fit based on the Bayesian information criterion and parsimony of model parameters.26 We report estimates of change in pain over time in monthly intervals for ease of interpretation. However, models used all individual pain scores rather than computing monthly averages when > 1 pain score was available within a month, as recommended in previous research.27 This approach makes optimal use of all available data. Both random effects (intercept and slope) were regressed onto the set of covariates described previously.
This study used data available in the EHR over the 24-month observation period. We characterize the density (eg, frequency) of all outcome variables by treatment condition in the Appendix. Because the hypotheses were directional, the authors used a 2-sided α = 0.10 and applied a Bonferroni correction for the 9 statistical tests performed, resulting in an adjusted α of 0.01. Treatment utilization and pharmacotherapy outcome analyses were performed in Stata, version 16.1. Random-effects growth modeling of pain score trajectories was performed using Mplus, version 8.8.

Results
There were 2182 patients in the treatment group and 2182 matched controls. The mean (SD) age was 54 (15) years; 81% were male, and about two-thirds (68%) identified as White and non-Hispanic. Mental health comorbidities were common, with > 40% of the sample having diagnoses of a mood disorder and/or posttraumatic stress disorder. Nearly all patients (90%) had VA service-connected disability ratings. Among patients with chronic musculoskeletal pain, 8% had comorbid neuropathic pain and 14% had headache. The mean (SD) duration of chronic pain across both patient groups was 3416 (2016) days, or about 9.4 years (Table 1).

Pharmacotherapy
High-dose opioid use (> 50 MME daily dose), days of opioid use, and concurrent opioid and benzodiazepine use decreased for all patients from the pre- to posttreatment period (Table 2). However, high-dose opioid use (Time × Treatment interaction, 3.24; 99% CI, 0.34 to 6.14) (Figure 1A) and concurrent opioid and benzodiazepine use (Time × Treatment interaction, 0.76; 99% CI, 0.67 to 0.86) (Figure 1B) had a larger decrease for the treatment vs matched control group. Treatment and matched control patients had comparable reductions in days of opioid use (Time × Treatment interaction, 0.98; 99% CI, 0.91 to 1.05) (Figure 1C). Neither group showed changes in nonopioid analgesic pharmacotherapy over time (main effect of time, incidence rate ratio [IRR], 1.03; 99% CI, 0.99 to 1.07; Time × Treatment interaction, 0.95; 99% CI, 0.90 to 1.01) (Figure 1D).


Nonpharmacologic Pain Treatment
The number of physical therapy and pain clinic visits declined for treatment patients and increased slightly for matched control patients (Figure 2A and 2B). For occupational therapy visits, neither group showed changes over time (main effect of time IRR, 1.03; 99% CI, 0.83 to 1.22; Time × Treatment interaction, 1.20; 99% CI, 0.93 to 1.46) (Figure 2C). For physical medicine and rehabilitation visits, both groups decreased use over time (main effect of time IRR, 0.78; 99% CI, 0.66 to 0.90), but this decrease did not differ between treatment and control patients (Time × Treatment interaction, 1.16; 99% CI, 0.94 to 1.37) (Figure 2D).

Pain Intensity
Pain intensity decreased for both groups by an estimated 0.02 points per month (99% CI, -0.04 to -0.01; P < .01), or 0.24 points over the 12-month postindex follow-up period (Figure 3). There were no statistically significant differences between treatment and control patients in pain intensity changes over the follow-up period (B = -0.02; 99% CI, -0.06 to 0.02; P = .15).

Discussion
Patients with musculoskeletal pain who received a thermal therapy device showed a larger decline in use of some specialty pain treatments, including physical therapy and specialty pain clinic services, when compared with matched control patients. One possible explanation is that patients who used the device may have had their pain adequately managed and thus required less specialty pain care. However, the absence of clinically significant changes in pain intensity over time suggests that pain intensity alone did not account for the observed changes in pain treatment use. We were unable to measure constructs of pain interference or functioning, which may be better predictors of functional restoration, as these data are not routinely collected within the VA and documented in the EHR. Future studies could clarify these findings by including measures of pain interference and functioning.
Although the overall declines in physical therapy and specialty pain clinic use associated with thermal therapy were modest (< 1 visit per patient), the impact of these reductions can be profound. In resource-limited health care settings, even small reductions in high-cost care utilization could be of great value in that health systems could offset costs associated with treating chronic pain without compromising quality of care or key clinical outcomes, such as pain intensity. This study, however, did not include a cost analysis. Future studies should incorporate formal cost analyses to quantify cost offsets that may result from decreased specialty pain treatment use.
Patients who received thermal therapy devices did not show clinically meaningful reductions in pain intensity over time, defined as reductions in pain intensity of 1.7 to 2.0 on a 0 to 10 scale.28,29 This finding is consistent with prior research that demonstrates relatively stable pain intensity self-ratings longitudinally by patients with chronic pain diagnoses, when assessed in the context of usual clinical care.30 This finding, however, is inconsistent with prior literature that demonstrates pain-reducing benefits of thermal therapy for low back pain and knee osteoarthritis.7-9
In this study, pain intensity ratings were derived from the EHR during routine outpatient clinical encounters and not at the time thermal therapy was self-administered, as has been done in prior clinical trials.7-9 Pain location was not specified at the time of pain ratings, and it is possible that patients may have been endorsing pain in areas of the body that had not been treated by thermal therapy. Patient-level variability in pain intensity ratings (eg, within-patient range over time) was not examined, although prior research indicates substantial variability.30 While average pain intensity ratings in the current study did not change, an examination of patient variability warrants further study, as a narrowing of pain intensity ratings can be perceived, by patients, as demonstrable improvement and has been associated with improved physical and psychological outcomes.31 Furthermore, pain intensity does not characterize physical or emotional functioning that can be captured with more comprehensive validated measures, some of which are recommended outcomes in pain clinical trials.32
Our findings point to reductions in all forms of opioid use across both treatment and control patients. Data from the VA and in the US more generally point to downward trends in opioid prescribing during the study period.33 This decline is likely due to increased use of risk mitigation approaches, such as routine urine drug screens and review of prescription drug monitoring databases.34 These state-level databases track prescribing of controlled substances, including opioids and benzodiazepines, within a state. Implementation of these practices has been associated with declines in higher risk opioid prescribing.34 Findings from this study further point to associations of reduced higher risk opioid use among patients who received thermal therapy devices. In the full sample of patients, reductions in days of opioid use, high-dose opioid use, and co-use of opioids and benzodiazepines were observed across all patients, with greater reductions observed in high-dose opioid use and concurrent opioid and benzodiazepine use among patients who received a thermal therapy device. Experimental studies indicate that the endogenous opioid system is implicated in pain relief and activated by thermal therapies. 35 Differential reductions in higher-risk opioid use among patients who received thermal therapy devices in our study may be associated with endogenous opioid activation, though this was not specifically measured. It is also unclear whether thermal therapy was provided by clinicians in the context of opioid tapering or other risk mitigation efforts, or patients reduced higher risk opioid use of their own volition. Prior research has identified both patient- and clinician-initiated opioid tapering and discontinuation.36 While a thorough explication of opioid dose reduction was beyond the scope of this study, future qualitative work could help explain potential benefits of thermal therapy in the context of analgesic pharmacotherapy use, including opioid medications.
Limitations
The extent to which patients used the thermal therapy device could not be measured; therefore, device receipt was used as a proxy for use. However, it was not possible to determine whether the frequency and duration of device use was associated with study outcomes. Treatment and control groups demonstrated some differences in outcome variables at the index date. Potential known biases were addressed using propensity score matching procedures and statistical procedures that controlled for patient demographic and clinical characteristics, as well as pretreatment values of all outcome variables. Nevertheless, as an observational study, this analysis cannot account for all known and unknown confounders, and a randomized controlled trial is needed to make claims of causality. The study population consisted of US veterans and included a low proportion of women. As a result, the findings may not be generalizable to other patient populations. Finally, prescription dispensing data, used as a proxy for pharmacotherapy use, do not necessarily reflect actual medication use.
Conclusions
This study is among the first to examine associations between thermal therapy and specialty pain treatment and analgesic pharmacotherapy use among US veterans. Although the retrospective cohort study design does not allow causal inferences regarding the efficacy of thermal therapy for veterans with chronic musculoskeletal pain, confidence in the findings is strengthened by methodological and statistical control of known confounders. Future trials employing experimental designs are needed to further clarify the clinical and health systems benefits of thermal therapy for musculoskeletal pain syndromes.
- Krebs EE, Gravely A, Nugent S, et al. Effect of opioid vs nonopioid medications on pain-related function in patients with chronic back pain or hip or knee osteoarthritis pain: the SPACE randomized clinical trial. JAMA. 2018;2018:872-882. doi:10.1001/jama.2018.0899
- Becker WC, DeBar LL, Heapy AA, et al. A research agenda for advancing non-pharmacological management of chronic musculoskeletal pain: findings from a VHA state-of-the-art conference. J Gen Intern Med. 2018;33:1-15. doi:10.1007/s11606-018-4345-6
- Fernandes IA, Armond ACV, Falci SGM. The effectiveness of the cold therapy (cryotherapy) in the management of inflammatory parameters after removal of mandibular third molars: a meta-analysis. Int Arch Otorhinolaryngol. 2019;23:221-228. doi:10.1055/s-0039-1677755
- Quinlan P, Davis J, Fields K, et al. Effects of localized cold therapy on pain in postoperative spinal fusion patients: a randomized controlled trial. Orthop Nurs. 2017;36:344-349. doi:10.1097/NOR.0000000000000382
- Malanga GA, Yan N, Stark J. Mechanisms and efficacy of heat and cold therapies for musculoskeletal injury. Postgrad Med. 2015;127:57-65. doi:10.1080/00325481.2015.992719
- Wang Y, Li S, Zhang Y, et al. Heat and cold therapy reduce pain in patients with delayed onset muscle soreness: a systematic review and meta-analysis of 32 randomized controlled trials. Phys Ther Sport. 2021;48:177-187. doi:10.1016/j.ptsp.2021.01.004
- Ariana M, Afrasiabifar A, Doulatabad SN, et al. The effect of local heat therapy versus cold rub gel on pain and joint functions in patients with knee osteoarthritis. Clin Nurs Res. 2022;31:1014-1022. doi:10.1177/10547738211035502
- French SD, Cameron M, Walker BF, et al. Superficial heat or cold for low back pain. Cochrane Database Syst Rev. 2006;1:CD004750. doi:10.1002/14651858.CD004750.pub2
- Tao XG, Bernacki EJ. A randomized clinical trial of continuous low-level heat therapy for acute muscular low back pain in the workplace. J Occup Environ Med. 2005;47:1298- 1306. doi:10.1097/01.jom.0000184877.01691.a3
- Freiwald J, Hoppe MW, Beermann W, et al. Effects of supplemental heat therapy in multimodal treated chronic low back pain patients on strength and flexibility. Clin Biomech. 2018;57:107-113. doi:10.1016/j.clinbiomech.2018.06.008
- Freiwald J, Magni A, Fanlo-Mazas P, et al. A role for superficial heat therapy in the management of nonspecific, mild-to-moderate low back pain in current clinical practice: a narrative review. Life. 2021;11:780. doi:10.3390/life11080780
- Kerns RD, Philip EJ, Lee AW, et al. Implementation of the Veterans Health Administration National Pain Management Strategy. Transl Behav Med. 2011;1:635-643. doi:10.1007/s13142-011-0094-3
- Brookhart MA, Schneeweiss S, Rothman KJ, et al. Variable selection for propensity score models. Am J Epidemiol. 2006;163:1149-1156. doi:10.1093/aje/kwj149
- Edlund MJ, Martin BC, Devries A, et al. Trends in use of opioids for chronic non-cancer pain among individuals with mental health and substance use disorders: the TROUP study. Clin J Pain. 2010;26:1-8. doi:10.1097/AJP.0b013e3181b99f35
- Kaur S, Stechuchak KM, Coffman CJ, et al. Gender differences in health care utilization among veterans with chronic pain. J Gen Intern Med. 2007;22:228-233. doi:10.1007/s11606-006-0048-5
- Meghani SH, Cho E. Self-reported pain and utilization of pain treatment between minorities and non-minorities in the United States. Public Health Nurs. 2009;26:307-316. doi:10.1111/j.1525-1446.2009.00785.x
- Morasco BJ, Duckart JP, Carr TP, et al. Clinical characteristics of veterans prescribed high doses of opioid medications for chronic non-cancer pain. Pain. 2010;151:625-632. doi:10.1016/j.pain.2010.08.002
- Nielsen S, Lintzeris N, Bruno R, et al. Benzodiazepine use among chronic pain patients prescribed opioids: associations with pain, physical and mental health, and health service utilization. Pain Med. 2015;16:356-366. doi:10.1111/pme.12594
- Quinn PD, Hur K, Chang Z, et al. Incident and long-term opioid therapy among patients with psychiatric conditions and medications: a national study of commercial health care claims. Pain. 2017;158:140-148. doi:10.1097/j.pain.0000000000000730
- Rubin DB. Matching to remove bias in observational studies. Biometrics. 1973;29:159-183.
- Rosenbaum PR, Rubin DB. Constructing a control group using multivariate matched sampling methods that incorporate the propensity score. Am Stat. 1985;39:33-38. doi:10.1007/s11596-025-00052-0
- Steiner PM, Cook TD, Shadish WR, et al. The importance of covariate selection in controlling for selection bias in observational studies. Psychol Methods. 2010;15:250-267. doi:10.1037/a0018719
- Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373- 383. doi:10.1016/0021-9681(87)90171-8
- Long JS, Freese J. Regression Models for Categorical Dependent Variables Using Stata. 3rd ed. Stata Press; 2014.
- Muthén LK, Muthén BO. Mplus User’s Guide. 8th ed. Muthén & Muthén; 1998-2017.
- Hedeker D, Gibbons RD. Longitudinal Data Analysis. Wiley; 2006.
- Dobscha SK, Morasco BJ, Kovas AE, et al. Short-term variability in outpatient pain intensity scores in a national sample of older veterans with chronic pain. Pain Med. 2015;16:855-865. doi:10.1111/pme.12643
- Bahreini M, Safaie A, Mirfazaelian H, et al. How much change in pain score does really matter to patients? Am J Emerg Med. 2020;38:1641-1646. doi:10.1016/j.ajem.2019.158489
- Suzuki H, Aono S, Inoue S, et al. Clinically significant changes in pain along the pain intensity numerical rating scale in patients with chronic low back pain. PLoS One. 2020;15:e0229228. doi:10.1371/journal.pone.0229228
- McPherson SM, Smith CL, Dobscha SK, et al. Changes in pain intensity after discontinuation of long-term opioid therapy for chronic noncancer pain. Pain. 2018;159:2097- 2104. doi:10.1097/j.pain.0000000000001315
- Andrews NE, Strong J, Meredith PJ. Activity pacing, avoidance, endurance, and associations with patient functioning in chronic pain: a systematic and meta-analysis. Arch Phys Med Rehabil. 2012;93:2109-121. doi:10.1016/j.apmr.2012.05.029
- Dworkin RH, Turk DC, Farrar JT, et al. Core outcome measures for chronic pain clinical trials: IMMPACT recommendations. Pain. 2005;113:9-19. doi:10.1016/j.pain.2004.09.012
- Gellad WF, Good CB, Shulkin DJ. Addressing the opioid epidemic in the United States: Lessons from the Department of Veterans Affairs. JAMA Intern Med. 2017;177:611- 612. doi:10.1001/jamainternmed.2017.0147
- Lin LA, Bohnert ASB, Kerns RD, et al. Impact of the Opioid Safety Initiative on opioid-related prescribing in veterans. Pain. 2017;158:833-839. doi:10.1097/j.pain.0000000000000837
- Sirucek L, Price RC, Gandhi W, et al. Endogenous opioids contribute to the feeling of pain relief in humans. Pain. 2021;162:2821-2831. doi:10.1097/j.pain.0000000000002285
- Lovejoy TI, Morasco BJ, Demidenko MI, et al. Reasons for discontinuation of long-term opioid therapy in patients with and without substance use disorders. Pain. 2017;158:526- 534. doi:10.1097/j.pain.0000000000000796
- Krebs EE, Gravely A, Nugent S, et al. Effect of opioid vs nonopioid medications on pain-related function in patients with chronic back pain or hip or knee osteoarthritis pain: the SPACE randomized clinical trial. JAMA. 2018;2018:872-882. doi:10.1001/jama.2018.0899
- Becker WC, DeBar LL, Heapy AA, et al. A research agenda for advancing non-pharmacological management of chronic musculoskeletal pain: findings from a VHA state-of-the-art conference. J Gen Intern Med. 2018;33:1-15. doi:10.1007/s11606-018-4345-6
- Fernandes IA, Armond ACV, Falci SGM. The effectiveness of the cold therapy (cryotherapy) in the management of inflammatory parameters after removal of mandibular third molars: a meta-analysis. Int Arch Otorhinolaryngol. 2019;23:221-228. doi:10.1055/s-0039-1677755
- Quinlan P, Davis J, Fields K, et al. Effects of localized cold therapy on pain in postoperative spinal fusion patients: a randomized controlled trial. Orthop Nurs. 2017;36:344-349. doi:10.1097/NOR.0000000000000382
- Malanga GA, Yan N, Stark J. Mechanisms and efficacy of heat and cold therapies for musculoskeletal injury. Postgrad Med. 2015;127:57-65. doi:10.1080/00325481.2015.992719
- Wang Y, Li S, Zhang Y, et al. Heat and cold therapy reduce pain in patients with delayed onset muscle soreness: a systematic review and meta-analysis of 32 randomized controlled trials. Phys Ther Sport. 2021;48:177-187. doi:10.1016/j.ptsp.2021.01.004
- Ariana M, Afrasiabifar A, Doulatabad SN, et al. The effect of local heat therapy versus cold rub gel on pain and joint functions in patients with knee osteoarthritis. Clin Nurs Res. 2022;31:1014-1022. doi:10.1177/10547738211035502
- French SD, Cameron M, Walker BF, et al. Superficial heat or cold for low back pain. Cochrane Database Syst Rev. 2006;1:CD004750. doi:10.1002/14651858.CD004750.pub2
- Tao XG, Bernacki EJ. A randomized clinical trial of continuous low-level heat therapy for acute muscular low back pain in the workplace. J Occup Environ Med. 2005;47:1298- 1306. doi:10.1097/01.jom.0000184877.01691.a3
- Freiwald J, Hoppe MW, Beermann W, et al. Effects of supplemental heat therapy in multimodal treated chronic low back pain patients on strength and flexibility. Clin Biomech. 2018;57:107-113. doi:10.1016/j.clinbiomech.2018.06.008
- Freiwald J, Magni A, Fanlo-Mazas P, et al. A role for superficial heat therapy in the management of nonspecific, mild-to-moderate low back pain in current clinical practice: a narrative review. Life. 2021;11:780. doi:10.3390/life11080780
- Kerns RD, Philip EJ, Lee AW, et al. Implementation of the Veterans Health Administration National Pain Management Strategy. Transl Behav Med. 2011;1:635-643. doi:10.1007/s13142-011-0094-3
- Brookhart MA, Schneeweiss S, Rothman KJ, et al. Variable selection for propensity score models. Am J Epidemiol. 2006;163:1149-1156. doi:10.1093/aje/kwj149
- Edlund MJ, Martin BC, Devries A, et al. Trends in use of opioids for chronic non-cancer pain among individuals with mental health and substance use disorders: the TROUP study. Clin J Pain. 2010;26:1-8. doi:10.1097/AJP.0b013e3181b99f35
- Kaur S, Stechuchak KM, Coffman CJ, et al. Gender differences in health care utilization among veterans with chronic pain. J Gen Intern Med. 2007;22:228-233. doi:10.1007/s11606-006-0048-5
- Meghani SH, Cho E. Self-reported pain and utilization of pain treatment between minorities and non-minorities in the United States. Public Health Nurs. 2009;26:307-316. doi:10.1111/j.1525-1446.2009.00785.x
- Morasco BJ, Duckart JP, Carr TP, et al. Clinical characteristics of veterans prescribed high doses of opioid medications for chronic non-cancer pain. Pain. 2010;151:625-632. doi:10.1016/j.pain.2010.08.002
- Nielsen S, Lintzeris N, Bruno R, et al. Benzodiazepine use among chronic pain patients prescribed opioids: associations with pain, physical and mental health, and health service utilization. Pain Med. 2015;16:356-366. doi:10.1111/pme.12594
- Quinn PD, Hur K, Chang Z, et al. Incident and long-term opioid therapy among patients with psychiatric conditions and medications: a national study of commercial health care claims. Pain. 2017;158:140-148. doi:10.1097/j.pain.0000000000000730
- Rubin DB. Matching to remove bias in observational studies. Biometrics. 1973;29:159-183.
- Rosenbaum PR, Rubin DB. Constructing a control group using multivariate matched sampling methods that incorporate the propensity score. Am Stat. 1985;39:33-38. doi:10.1007/s11596-025-00052-0
- Steiner PM, Cook TD, Shadish WR, et al. The importance of covariate selection in controlling for selection bias in observational studies. Psychol Methods. 2010;15:250-267. doi:10.1037/a0018719
- Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373- 383. doi:10.1016/0021-9681(87)90171-8
- Long JS, Freese J. Regression Models for Categorical Dependent Variables Using Stata. 3rd ed. Stata Press; 2014.
- Muthén LK, Muthén BO. Mplus User’s Guide. 8th ed. Muthén & Muthén; 1998-2017.
- Hedeker D, Gibbons RD. Longitudinal Data Analysis. Wiley; 2006.
- Dobscha SK, Morasco BJ, Kovas AE, et al. Short-term variability in outpatient pain intensity scores in a national sample of older veterans with chronic pain. Pain Med. 2015;16:855-865. doi:10.1111/pme.12643
- Bahreini M, Safaie A, Mirfazaelian H, et al. How much change in pain score does really matter to patients? Am J Emerg Med. 2020;38:1641-1646. doi:10.1016/j.ajem.2019.158489
- Suzuki H, Aono S, Inoue S, et al. Clinically significant changes in pain along the pain intensity numerical rating scale in patients with chronic low back pain. PLoS One. 2020;15:e0229228. doi:10.1371/journal.pone.0229228
- McPherson SM, Smith CL, Dobscha SK, et al. Changes in pain intensity after discontinuation of long-term opioid therapy for chronic noncancer pain. Pain. 2018;159:2097- 2104. doi:10.1097/j.pain.0000000000001315
- Andrews NE, Strong J, Meredith PJ. Activity pacing, avoidance, endurance, and associations with patient functioning in chronic pain: a systematic and meta-analysis. Arch Phys Med Rehabil. 2012;93:2109-121. doi:10.1016/j.apmr.2012.05.029
- Dworkin RH, Turk DC, Farrar JT, et al. Core outcome measures for chronic pain clinical trials: IMMPACT recommendations. Pain. 2005;113:9-19. doi:10.1016/j.pain.2004.09.012
- Gellad WF, Good CB, Shulkin DJ. Addressing the opioid epidemic in the United States: Lessons from the Department of Veterans Affairs. JAMA Intern Med. 2017;177:611- 612. doi:10.1001/jamainternmed.2017.0147
- Lin LA, Bohnert ASB, Kerns RD, et al. Impact of the Opioid Safety Initiative on opioid-related prescribing in veterans. Pain. 2017;158:833-839. doi:10.1097/j.pain.0000000000000837
- Sirucek L, Price RC, Gandhi W, et al. Endogenous opioids contribute to the feeling of pain relief in humans. Pain. 2021;162:2821-2831. doi:10.1097/j.pain.0000000000002285
- Lovejoy TI, Morasco BJ, Demidenko MI, et al. Reasons for discontinuation of long-term opioid therapy in patients with and without substance use disorders. Pain. 2017;158:526- 534. doi:10.1097/j.pain.0000000000000796
Thermal Therapy is Associated With Decreased Pain Treatment Use: A Retrospective Cohort Study
Thermal Therapy is Associated With Decreased Pain Treatment Use: A Retrospective Cohort Study
Primary Care Clinician and Patient Knowledge, Interest, and Use of Integrative Treatment Options for Chronic Low Back Pain Management
Primary Care Clinician and Patient Knowledge, Interest, and Use of Integrative Treatment Options for Chronic Low Back Pain Management
More than 50 million US adults report experiencing chronic pain, with nearly 7% experiencing high-impact chronic pain.1-3 Chronic pain negatively affects daily function, results in lost productivity, is a leading cause of disability, and is more prevalent among veterans compared with the general population.1,2,4-6 Estimates from 2021 suggest the prevalence of chronic pain among veterans exceeds 30%; > 11% experienced high-impact chronic pain.1
Primary care practitioners (PCPs) have a prominent role in chronic pain management. Pharmacologic options for treating pain, once a mainstay of therapy, present several challenges for patients and PCPs, including drug-drug interactions and adverse effects.7 The US opioid epidemic and shift to a biopsychosocial model of chronic pain care have increased emphasis on nonpharmacologic treatment options.8,9 These include integrative modalities, which incorporate conventional approaches with an array of complementary health approaches.10-12
Integrative therapy is a prominent feature in whole person care, which may be best exemplified by the US Department of Veterans Affairs (VA) Whole Health System of care.13-14 Whole health empowers an individual to take charge of their health and well-being so they can “live their life to the fullest.”14 As implemented in the Veterans Health Administration (VHA), whole health includes the use of evidence-based
METHODS
Using a cross-sectional survey design, PCPs and patients with chronic back pain affiliated with the VA Ann Arbor Healthcare System were invited to participate in separate but similar surveys to assess knowledge, interest, and use of nonpharmacologic integrative modalities for the treatment of chronic pain. In May, June, and July 2023, 78 PCPs received 3 email
Both survey instruments are available upon request, were developed by the study team, and included a mix of yes/no questions, “select all that apply” items, Likert scale response items, and open-ended questions. For one question about which modalities they would like available, the respondent was instructed to select up to 5 modalities. The instruments were extensively pretested by members of the study team, which included 2 PCPs and a nonveteran with chronic back pain.
The list of integrative modalities included in the survey was derived from the tier 1 and tier 2 complementary and integrative health modalities identified in a VHA Directive on complementary and integrative health.15,16 Tier 1 approaches are considered to have sufficient evidence and must be made available to veterans either within a VA medical facility or in the community. Tier 2 approaches are generally considered safe and may be made available but do not have sufficient evidence to mandate their provision. For participant ease, the integrative modalities were divided into 5 subgroups: manual therapies, energy/biofield therapies, mental health therapies, nutrition counseling, and movement therapies. The clinician survey assessed clinicians’ training and interest, clinical and personal use, and perceived barriers to providing integrative modalities for chronic pain. Professional and personal demographic data were also collected. Similarly, the patient survey assessed use of integrative therapies, perceptions of and interest in integrative modalities, and potential barriers to use. Demographic and health-related information was also collected.
Data analysis included descriptive statistics (eg, frequency counts, means, medians) and visual graphic displays. Separate analyses were conducted for clinicians and patients in addition to a comparative analysis of the use and potential interest in integrative modalities. Analysis were conducted using R software. This study was deemed nonresearch quality improvement by the VA Ann Arbor Healthcare System facility research oversight board and institutional review board approval was not solicited.
RESULTS
Twenty-eight clinicians completed the survey, yielding a participation rate of 36%. Participating clinicians had a median (IQR) age of 48 years (9.5), 15 self-identified as White (54%), 8 as Asian (29%), 15 as female (54%), 26 as non-Hispanic (93%), and 25 were medical doctors or doctors of osteopathy (89%). Nineteen (68%) worked at the main hospital outpatient clinic, and 9 practiced at community-based outpatient clinics (CBOCs). Thirteen respondents (46%) reported having no formal education or training in integrative approaches. Among those with prior training, 8 clinicians had nutrition counseling (29%) and 7 had psychologic therapy training (25%). Thirteen respondents (46%) also reported using integrative modalities for personal health needs: 8 used psychological therapies, 8 used movement therapies, 10 used integrative modalities for stress management or relaxation, and 8 used them for physical symptoms (Table 1).
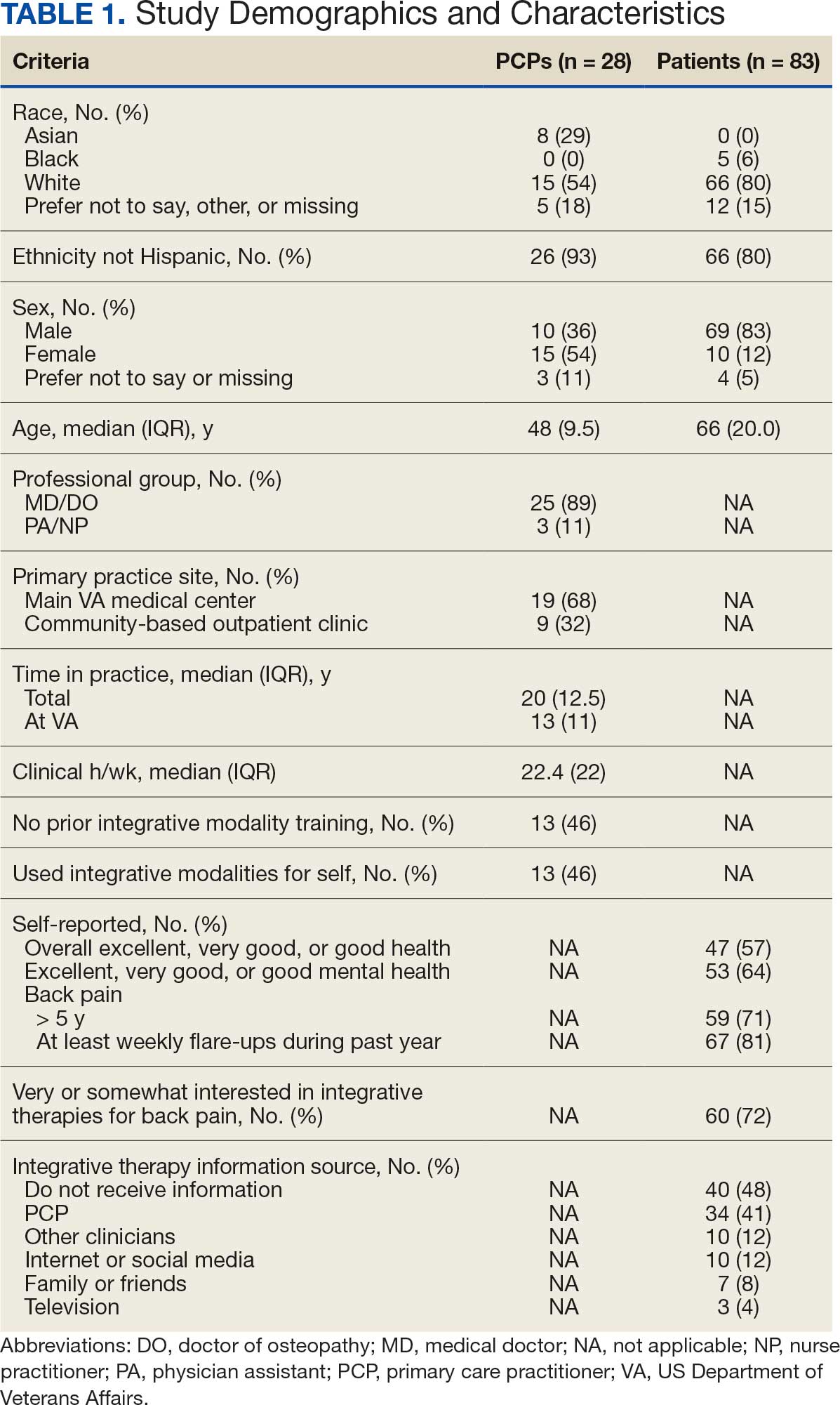
Overall, 85 of 200 patients (43%) responded to the study survey. Two patients indicated they did not have chronic back pain and were excluded. Patients had a median (IQR) age of 66 (20) years, with 66 self-identifying as White (80%), 69 as male (83%), and 66 as non-Hispanic (80%). Forty-four patients (53%) received care at CBOCs. Forty-seven patients reported excellent, very good, or good overall health (57%), while 53 reported excellent, very good, or good mental health (64%). Fifty-nine patients reported back pain duration > 5 years (71%), and 67 (81%) indicated experiencing back pain flare-ups at least once per week over the previous 12 months. Sixty patients (72%) indicated they were somewhat or very interested in using integrative therapies as a back pain treatment; however, 40 patients (48%) indicated they had not received information about these therapies. Among those who indicated they had received information, the most frequently reported source was their PCP (41%). Most patients (72%) also reported feeling somewhat to very comfortable discussing integrative medicine therapies with their PCP.
Integrative Therapy Recommendations and Use
PCPs reported recommending multiple integrative modalities: 23 (82%) recommended cognitive-behavioral therapy, 22 (79%) recommended acupuncture, 21 (75%) recommended chiropractic, 19 (68%) recommended battlefield acupuncture, recommended massage 18 (64%), 17 (61%) recommended meditation or mindfulness, and 15 (54%) recommended movement therapies such as yoga or tai chi/qigong (Figure 1). The only therapies used by at least half of the patients were chiropractic used by 59 patients (71%) and acupuncture by 42 patients (51%). Thirty-eight patients (46%) reported massage use and 21 patients (25%) used cognitive-behavioral therapy (Table 2).
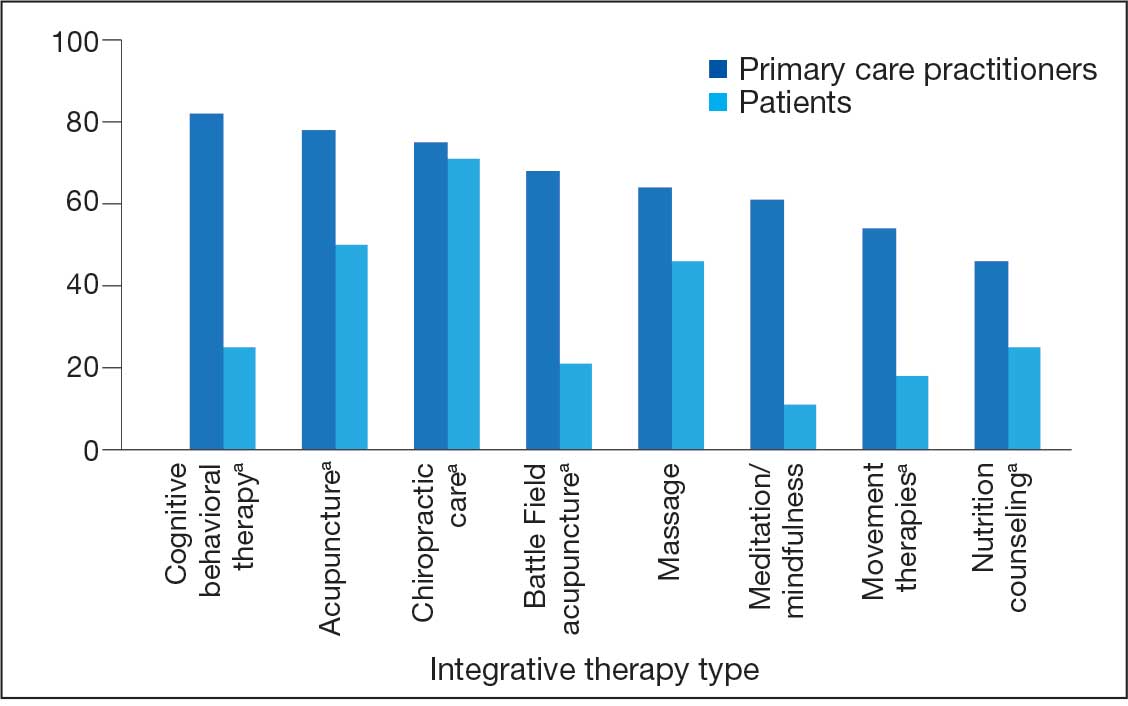
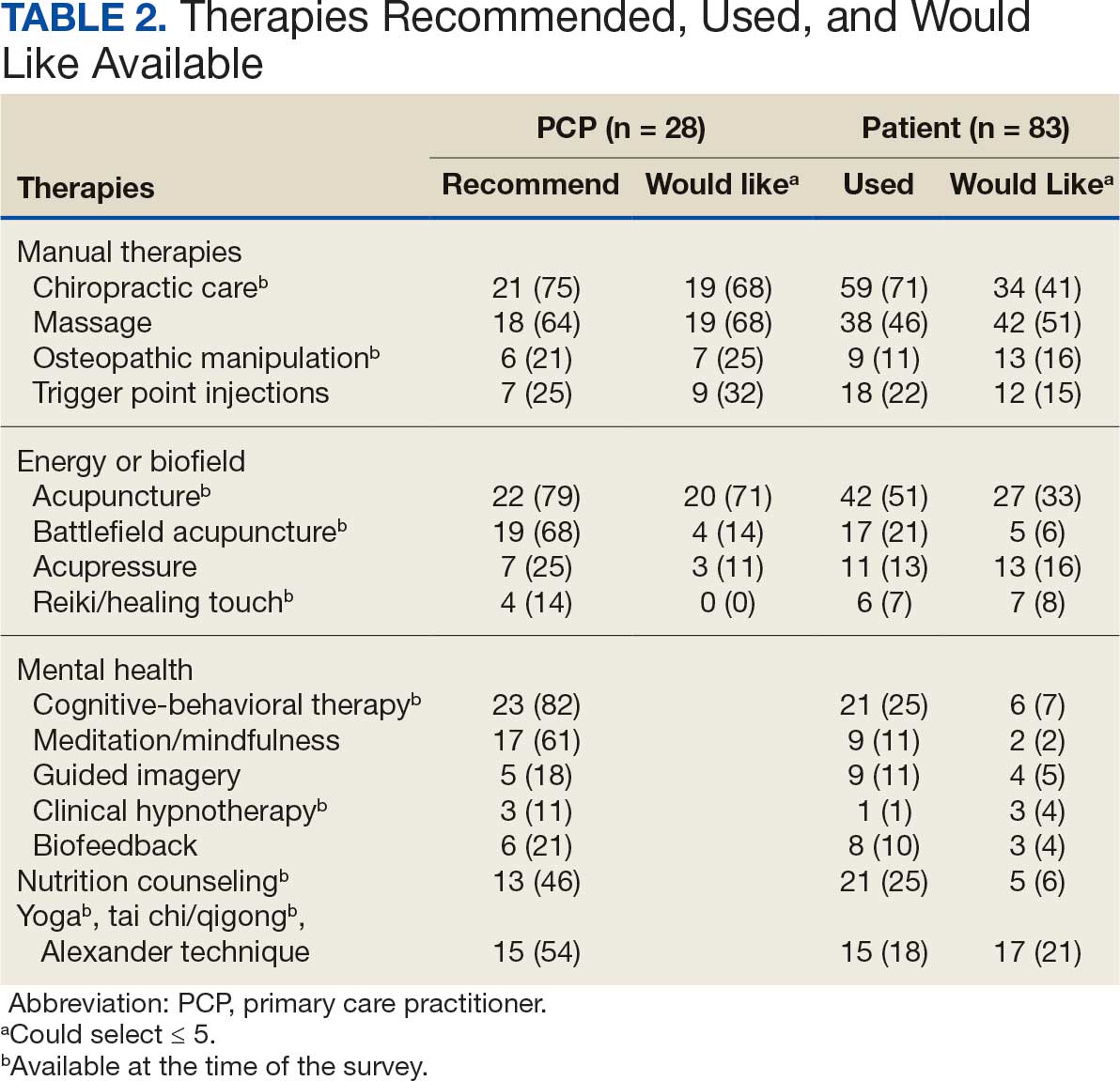
Integrative Therapies Desired
A majority of PCPs identified acupuncture (n = 20, 71%), chiropractic (n = 19, 68%), and massage (n = 19, 68%) as therapies they would most like to have available for patients with chronic pain (Figure 2). Similarly, patients identified massage (n = 42, 51%), chiropractic (n = 34, 41%), and acupuncture (n = 27, 33%) as most desired. Seventeen patients (21%) expressed interest in movement therapies.
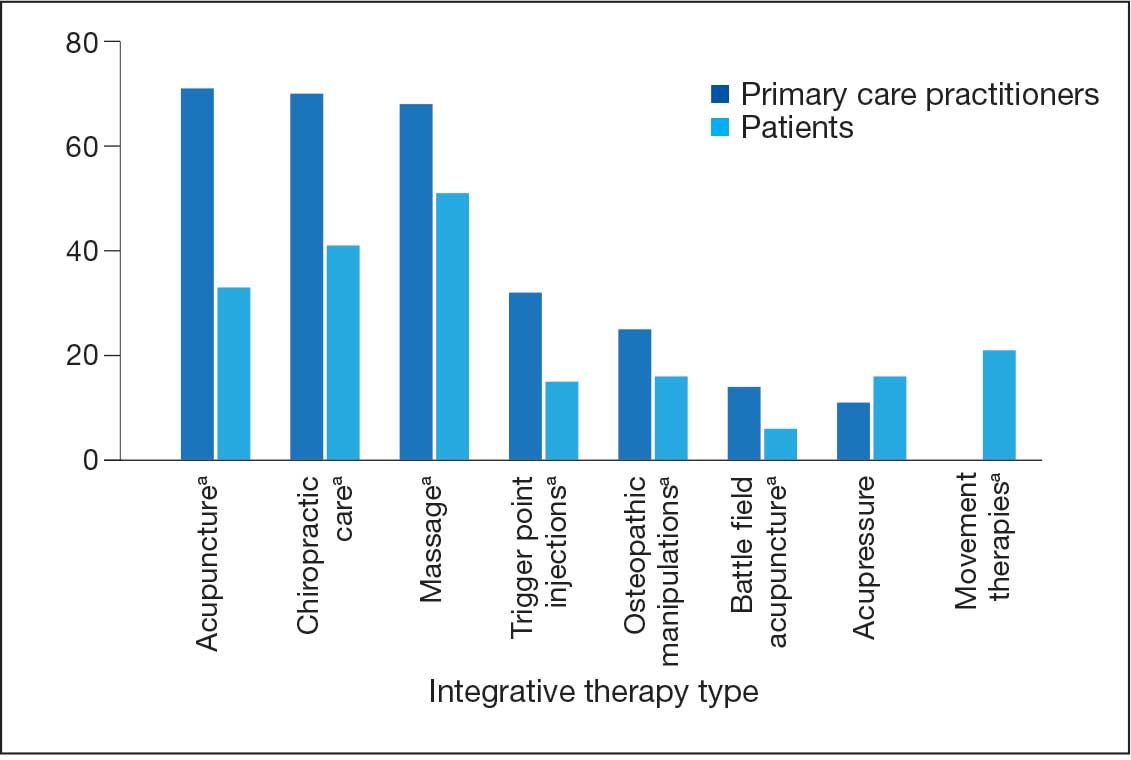
Barriers to Integrative Therapies Use
When asked about barriers to use, 26 PCPs (93%) identified access to services as a somewhat or extremely likely barrier, and 22 identified time constraints (79%) (Table 3). However, 17 PCPs (61%) noted lack of familiarity, and 18 (64%) noted a lack of scientific evidence as barriers to recommending integrative modalities. Among patients, 33 (40%) indicated not knowing what services were available at their facility as a barrier, 32 (39%) were not familiar with specific therapies, and 21 (25%) indicated a lack of clarity about the benefits of a specific therapy. Only 14 patients (17%) indicated that there were no obstacles to use.

DISCUSSION
Use of integrative therapies, including complementary treatments, is an increasingly important part of chronic pain management. This survey study suggests VA PCPs are willing to recommend integrative therapies and patients with chronic back pain both desire and use several therapies. Moreover, both groups expressed interest in greater availability of similar therapies. The results also highlight key barriers, such as knowledge gaps, that should be addressed to increase the uptake of integrative modalities for managing chronic pain.
An increasing number of US adults are using complementary health approaches, an important component of integrative therapy.12 This trend includes an increase in use for pain management, from 42.3% in 2002 to 49.2% in 2022; chiropractic care, acupuncture, and massage were most frequently used.12 Similarly, chiropractic, acupuncture and massage were most often used by this sample of veterans with chronic back pain and were identified by the highest percentages of PCPs and patients as the therapies they would most like available.
There were areas where the opinions of patients and clinicians differed. As has been seen previously reported, clinicians largely recommended cognitive-behavioral therapy while patients showed less interest.17 Additionally, while patients expressed interest in the availability of movement therapies, such as yoga, PCPs expressed more interest in other strategies, such as trigger point injections. These differences may reflect true preference or a tendency for clinicians and patients to select therapies with which they are more familiar. Additional research is needed to better understand the acceptability and potential use of integrative health treatments across a broad array of therapeutic options.
Despite VHA policy requiring facilities to provide certain complementary and integrative health modalities, almost all PCPs identified access to services as a major obstacle.15 Based on evidence and a rigorous vetting process, services currently required on-site, via telehealth, or through community partners include acupuncture and battlefield acupuncture (battlefield auricular acupuncture), biofeedback, clinical hypnosis, guided imagery, medical massage therapy, medication, tai chi/qigong, and yoga. Optional approaches, which may be made available to veterans, include chiropractic and healing touch. Outside the VHA, some states have introduced or enacted legislation mandating insurance coverage of nonpharmacological pain treatments.18 However, these requirements and mandates do not help address challenges such as the availability of trained/qualified practitioners.19,20 Ensuring access to complementary and integrative health treatments requires a more concerted effort to ensure that supply meets demand. It is also important to acknowledge the budgetary and physical space constraints that further limit access to services. Although expansion and integration of integrative medicine services remain a priority within the VA Whole Health program, implementation is contingent on available financial and infrastructure resources.
Time was also identified by PCPs as a barrier to recommending integrative therapies to patients. Developing and implementing time-efficient communication strategies for patient education such as concise talking points and informational handouts could help address this barrier. Furthermore, leveraging existing programs and engaging the entire health care team in patient education and referral could help increase integrative and complementary therapy uptake and use.
Although access and time were identified as major barriers, these findings also suggest that PCP and patient knowledge are another target area for enhancing the use of complementary and integrative therapies. Like prior research, most clinicians identified a lack of familiarity with certain services and a lack of scientific evidence as extremely or somewhat likely to affect their ability to offer integrative services to patients with chronic pain.21 Likewise, about 40% of patients identified being unfamiliar with a specific therapy as one of the major obstacles to receiving integrative therapies, with a similar number identifying PCPs as a source of information. The lack of familiarity may be due in part to the evolving nomenclature, with terms such as alternative, complementary, and integrative used to describe approaches outside what is often considered conventional medicine.10 On the other hand, there has also been considerable expansion in the number of therapies within this domain, along with an expanding evidence base. This suggests a need for targeted educational strategies for clinicians and patients, which can be rapidly deployed and continuously adapted as new therapies and evidence emerge.
Limitations
There are some inherent limitations with a survey-based approach, including sampling, non-response, and social desirability biases. In addition, this study only included PCPs and patients affiliated with a single VA medical center. Steps to mitigate these limitations included maintaining survey anonymity and reporting information about respondent characteristics to enhance transparency about the representativeness of the study findings.
CONCLUSIONS
Expanding the use of nonpharmacological pain treatments, including integrative modalities, is essential for safe and effective chronic pain management and reducing opioid use. Our findings show that VA PCPs and patients with chronic back pain are interested in and have some experience with certain integrative therapies. However, even within the context of a health care system that supports the use of integrative therapies for chronic pain as part of whole person care, increasing uptake will require addressing access and time-related constraints as well as ongoing clinician and patient education.
- Rikard SM, Strahan AE, Schmit KM, et al. Chronic pain among adults — United States, 2018-2021. MMWR Morb Mortal Wkly Rep. 2023;72:379-385. doi:10.15585/mmwr.mm7215a1
- Yong RJ, Mullins PM, Bhattacharyya N. Prevalence of chronic pain among adults in the United States. Pain. 2022;163:E328-E332. doi:10.1097/j.pain.0000000000002291
- Nahin RL, Feinberg T, Kapos FP, Terman GW. Estimated rates of incident and persistent chronic pain among US adults, 2019-2020. JAMA Netw Open. 2023;6:e2313563. doi:10.1001/jamanetworkopen.2023.13563
- Ferrari AJ, Santomauro DF, Aali A, et al. Global incidence, prevalence, years lived with disability (YLDs), disability-adjusted life-years (DALYs), and healthy life expectancy (HALE) for 371 diseases and injuries in 204 countries and territories and 811 subnational locations, 1990–2021: a systematic analysis for the Global Burden of Disease Study 2021. The Lancet. 2024;403:2133-2161. doi:10.1016/S0140-6736(24)00757-8 5.
- Qureshi AR, Patel M, Neumark S, et al. Prevalence of chronic non-cancer pain among military veterans: a systematic review and meta-analysis of observational studies. BMJ Mil Health. 2025;171:310-314. doi:10.1136/military-2023-002554
- Feldman DE, Nahin RL. Disability among persons with chronic severe back pain: results from a nationally representative population-based sample. J Pain. 2022;23:2144-2154. doi:10.1016/j.jpain.2022.07.016
- Qaseem A, Wilt TJ, McLean RM, Forciea MA. Noninvasive treatments for acute, subacute, and chronic low back pain: a clinical practice guideline from the American College of Physicians. Ann Intern Med. 2017;166:514-530. doi:10.7326/M16-2367
- van Erp RMA, Huijnen IPJ, Jakobs MLG, Kleijnen J, Smeets RJEM. Effectiveness of primary care interventions using a biopsychosocial approach in chronic low back pain: a systematic review. Pain Practice. 2019;19:224-241. doi:10.1111/papr.12735
- Chou R, Deyo R, Friedly J, et al. Nonpharmacologic therapies for low back pain: a systematic review for an American College of physicians clinical practice guideline. Ann Intern Med. 2017;166:493-505. doi:10.7326/M16-2459
- Complementary, alternative, or integrative health: what’s in a name? National Institutes of Health, National Center for Complementary and Integrative Health. Updated April 2021. Accessed December 15, 2025. https://www.nccih.nih.gov/health/complementary-alternative-or-integrative-health-whats-in-a-name.
- Taylor SL, Elwy AR. Complementary and alternative medicine for US veterans and active duty military personnel promising steps to improve their health. Med Care. 2014;52:S1-S4. doi:10.1097/MLR.0000000000000270.
- Nahin RL, Rhee A, Stussman B. Use of complementary health approaches overall and for pain management by US adults. JAMA. 2024;331:613-615. doi:10.1001/jama.2023.26775
- Gantt CJ, Donovan N, Khung M. Veterans Affairs’ Whole Health System of Care for transitioning service members and veterans. Mil Med. 2023;188:28-32. doi:10.1093/milmed/usad047
- Bokhour BG, Hyde J, Kligler B, et al. From patient outcomes to system change: evaluating the impact of VHA’s implementation of the Whole Health System of Care. Health Serv Res. 2022;57:53-65. doi:10.1111/1475-6773.13938
- Department of Veterans Affairs VHA. VHA Policy Directive 1137: Provision of Complementary and Integrative Health. December 2022. Accessed December 15, 2025. https://www.va.gov/VHApublications/ViewPublication.asp?pub_ID=10072
- Giannitrapani KF, Holliday JR, Miake-Lye IM, Hempel S, Taylor SL. Synthesizing the strength of the evidence of complementary and integrative health therapies for pain. Pain Med. 2019;20:1831-1840. doi:10.1093/pm/pnz068
- Belitskaya-Levy I, David Clark J, Shih MC, Bair MJ. Treatment preferences for chronic low back pain: views of veterans and their providers. J Pain Res. 2021;14:161-171. doi:10.2147/JPR.S290400
- Onstott TN, Hurst S, Kronick R, Tsou AC, Groessl E, McMenamin SB. Health insurance mandates for nonpharmacological pain treatments in 7 US states. JAMA Netw Open. 2024;7:E245737. doi:10.1001/jamanetworkopen.2024.5737
- Sullivan M, Leach M, Snow J, Moonaz S. The North American yoga therapy workforce survey. Complement Ther Med. 2017;31:39-48. doi:10.1016/j.ctim.2017.01.006
- Bolton R, Ritter G, Highland K, Larson MJ. The relationship between capacity and utilization of nonpharmacologic therapies in the US Military Health System. BMC Health Serv Res. 2022;22. doi:10.1186/s12913-022-07700-4
- Stussman BJ, Nahin RL, Barnes PM, Scott R, Feinberg T, Ward BW. Reasons office-based physicians in the United States recommend common complementary health approaches to patients: an exploratory study using a national survey. J Integr Complement Med. 2022;28:651-663. doi:10.1089/jicm.2022.0493
More than 50 million US adults report experiencing chronic pain, with nearly 7% experiencing high-impact chronic pain.1-3 Chronic pain negatively affects daily function, results in lost productivity, is a leading cause of disability, and is more prevalent among veterans compared with the general population.1,2,4-6 Estimates from 2021 suggest the prevalence of chronic pain among veterans exceeds 30%; > 11% experienced high-impact chronic pain.1
Primary care practitioners (PCPs) have a prominent role in chronic pain management. Pharmacologic options for treating pain, once a mainstay of therapy, present several challenges for patients and PCPs, including drug-drug interactions and adverse effects.7 The US opioid epidemic and shift to a biopsychosocial model of chronic pain care have increased emphasis on nonpharmacologic treatment options.8,9 These include integrative modalities, which incorporate conventional approaches with an array of complementary health approaches.10-12
Integrative therapy is a prominent feature in whole person care, which may be best exemplified by the US Department of Veterans Affairs (VA) Whole Health System of care.13-14 Whole health empowers an individual to take charge of their health and well-being so they can “live their life to the fullest.”14 As implemented in the Veterans Health Administration (VHA), whole health includes the use of evidence-based
METHODS
Using a cross-sectional survey design, PCPs and patients with chronic back pain affiliated with the VA Ann Arbor Healthcare System were invited to participate in separate but similar surveys to assess knowledge, interest, and use of nonpharmacologic integrative modalities for the treatment of chronic pain. In May, June, and July 2023, 78 PCPs received 3 email
Both survey instruments are available upon request, were developed by the study team, and included a mix of yes/no questions, “select all that apply” items, Likert scale response items, and open-ended questions. For one question about which modalities they would like available, the respondent was instructed to select up to 5 modalities. The instruments were extensively pretested by members of the study team, which included 2 PCPs and a nonveteran with chronic back pain.
The list of integrative modalities included in the survey was derived from the tier 1 and tier 2 complementary and integrative health modalities identified in a VHA Directive on complementary and integrative health.15,16 Tier 1 approaches are considered to have sufficient evidence and must be made available to veterans either within a VA medical facility or in the community. Tier 2 approaches are generally considered safe and may be made available but do not have sufficient evidence to mandate their provision. For participant ease, the integrative modalities were divided into 5 subgroups: manual therapies, energy/biofield therapies, mental health therapies, nutrition counseling, and movement therapies. The clinician survey assessed clinicians’ training and interest, clinical and personal use, and perceived barriers to providing integrative modalities for chronic pain. Professional and personal demographic data were also collected. Similarly, the patient survey assessed use of integrative therapies, perceptions of and interest in integrative modalities, and potential barriers to use. Demographic and health-related information was also collected.
Data analysis included descriptive statistics (eg, frequency counts, means, medians) and visual graphic displays. Separate analyses were conducted for clinicians and patients in addition to a comparative analysis of the use and potential interest in integrative modalities. Analysis were conducted using R software. This study was deemed nonresearch quality improvement by the VA Ann Arbor Healthcare System facility research oversight board and institutional review board approval was not solicited.
RESULTS
Twenty-eight clinicians completed the survey, yielding a participation rate of 36%. Participating clinicians had a median (IQR) age of 48 years (9.5), 15 self-identified as White (54%), 8 as Asian (29%), 15 as female (54%), 26 as non-Hispanic (93%), and 25 were medical doctors or doctors of osteopathy (89%). Nineteen (68%) worked at the main hospital outpatient clinic, and 9 practiced at community-based outpatient clinics (CBOCs). Thirteen respondents (46%) reported having no formal education or training in integrative approaches. Among those with prior training, 8 clinicians had nutrition counseling (29%) and 7 had psychologic therapy training (25%). Thirteen respondents (46%) also reported using integrative modalities for personal health needs: 8 used psychological therapies, 8 used movement therapies, 10 used integrative modalities for stress management or relaxation, and 8 used them for physical symptoms (Table 1).

Overall, 85 of 200 patients (43%) responded to the study survey. Two patients indicated they did not have chronic back pain and were excluded. Patients had a median (IQR) age of 66 (20) years, with 66 self-identifying as White (80%), 69 as male (83%), and 66 as non-Hispanic (80%). Forty-four patients (53%) received care at CBOCs. Forty-seven patients reported excellent, very good, or good overall health (57%), while 53 reported excellent, very good, or good mental health (64%). Fifty-nine patients reported back pain duration > 5 years (71%), and 67 (81%) indicated experiencing back pain flare-ups at least once per week over the previous 12 months. Sixty patients (72%) indicated they were somewhat or very interested in using integrative therapies as a back pain treatment; however, 40 patients (48%) indicated they had not received information about these therapies. Among those who indicated they had received information, the most frequently reported source was their PCP (41%). Most patients (72%) also reported feeling somewhat to very comfortable discussing integrative medicine therapies with their PCP.
Integrative Therapy Recommendations and Use
PCPs reported recommending multiple integrative modalities: 23 (82%) recommended cognitive-behavioral therapy, 22 (79%) recommended acupuncture, 21 (75%) recommended chiropractic, 19 (68%) recommended battlefield acupuncture, recommended massage 18 (64%), 17 (61%) recommended meditation or mindfulness, and 15 (54%) recommended movement therapies such as yoga or tai chi/qigong (Figure 1). The only therapies used by at least half of the patients were chiropractic used by 59 patients (71%) and acupuncture by 42 patients (51%). Thirty-eight patients (46%) reported massage use and 21 patients (25%) used cognitive-behavioral therapy (Table 2).


Integrative Therapies Desired
A majority of PCPs identified acupuncture (n = 20, 71%), chiropractic (n = 19, 68%), and massage (n = 19, 68%) as therapies they would most like to have available for patients with chronic pain (Figure 2). Similarly, patients identified massage (n = 42, 51%), chiropractic (n = 34, 41%), and acupuncture (n = 27, 33%) as most desired. Seventeen patients (21%) expressed interest in movement therapies.

Barriers to Integrative Therapies Use
When asked about barriers to use, 26 PCPs (93%) identified access to services as a somewhat or extremely likely barrier, and 22 identified time constraints (79%) (Table 3). However, 17 PCPs (61%) noted lack of familiarity, and 18 (64%) noted a lack of scientific evidence as barriers to recommending integrative modalities. Among patients, 33 (40%) indicated not knowing what services were available at their facility as a barrier, 32 (39%) were not familiar with specific therapies, and 21 (25%) indicated a lack of clarity about the benefits of a specific therapy. Only 14 patients (17%) indicated that there were no obstacles to use.

DISCUSSION
Use of integrative therapies, including complementary treatments, is an increasingly important part of chronic pain management. This survey study suggests VA PCPs are willing to recommend integrative therapies and patients with chronic back pain both desire and use several therapies. Moreover, both groups expressed interest in greater availability of similar therapies. The results also highlight key barriers, such as knowledge gaps, that should be addressed to increase the uptake of integrative modalities for managing chronic pain.
An increasing number of US adults are using complementary health approaches, an important component of integrative therapy.12 This trend includes an increase in use for pain management, from 42.3% in 2002 to 49.2% in 2022; chiropractic care, acupuncture, and massage were most frequently used.12 Similarly, chiropractic, acupuncture and massage were most often used by this sample of veterans with chronic back pain and were identified by the highest percentages of PCPs and patients as the therapies they would most like available.
There were areas where the opinions of patients and clinicians differed. As has been seen previously reported, clinicians largely recommended cognitive-behavioral therapy while patients showed less interest.17 Additionally, while patients expressed interest in the availability of movement therapies, such as yoga, PCPs expressed more interest in other strategies, such as trigger point injections. These differences may reflect true preference or a tendency for clinicians and patients to select therapies with which they are more familiar. Additional research is needed to better understand the acceptability and potential use of integrative health treatments across a broad array of therapeutic options.
Despite VHA policy requiring facilities to provide certain complementary and integrative health modalities, almost all PCPs identified access to services as a major obstacle.15 Based on evidence and a rigorous vetting process, services currently required on-site, via telehealth, or through community partners include acupuncture and battlefield acupuncture (battlefield auricular acupuncture), biofeedback, clinical hypnosis, guided imagery, medical massage therapy, medication, tai chi/qigong, and yoga. Optional approaches, which may be made available to veterans, include chiropractic and healing touch. Outside the VHA, some states have introduced or enacted legislation mandating insurance coverage of nonpharmacological pain treatments.18 However, these requirements and mandates do not help address challenges such as the availability of trained/qualified practitioners.19,20 Ensuring access to complementary and integrative health treatments requires a more concerted effort to ensure that supply meets demand. It is also important to acknowledge the budgetary and physical space constraints that further limit access to services. Although expansion and integration of integrative medicine services remain a priority within the VA Whole Health program, implementation is contingent on available financial and infrastructure resources.
Time was also identified by PCPs as a barrier to recommending integrative therapies to patients. Developing and implementing time-efficient communication strategies for patient education such as concise talking points and informational handouts could help address this barrier. Furthermore, leveraging existing programs and engaging the entire health care team in patient education and referral could help increase integrative and complementary therapy uptake and use.
Although access and time were identified as major barriers, these findings also suggest that PCP and patient knowledge are another target area for enhancing the use of complementary and integrative therapies. Like prior research, most clinicians identified a lack of familiarity with certain services and a lack of scientific evidence as extremely or somewhat likely to affect their ability to offer integrative services to patients with chronic pain.21 Likewise, about 40% of patients identified being unfamiliar with a specific therapy as one of the major obstacles to receiving integrative therapies, with a similar number identifying PCPs as a source of information. The lack of familiarity may be due in part to the evolving nomenclature, with terms such as alternative, complementary, and integrative used to describe approaches outside what is often considered conventional medicine.10 On the other hand, there has also been considerable expansion in the number of therapies within this domain, along with an expanding evidence base. This suggests a need for targeted educational strategies for clinicians and patients, which can be rapidly deployed and continuously adapted as new therapies and evidence emerge.
Limitations
There are some inherent limitations with a survey-based approach, including sampling, non-response, and social desirability biases. In addition, this study only included PCPs and patients affiliated with a single VA medical center. Steps to mitigate these limitations included maintaining survey anonymity and reporting information about respondent characteristics to enhance transparency about the representativeness of the study findings.
CONCLUSIONS
Expanding the use of nonpharmacological pain treatments, including integrative modalities, is essential for safe and effective chronic pain management and reducing opioid use. Our findings show that VA PCPs and patients with chronic back pain are interested in and have some experience with certain integrative therapies. However, even within the context of a health care system that supports the use of integrative therapies for chronic pain as part of whole person care, increasing uptake will require addressing access and time-related constraints as well as ongoing clinician and patient education.
More than 50 million US adults report experiencing chronic pain, with nearly 7% experiencing high-impact chronic pain.1-3 Chronic pain negatively affects daily function, results in lost productivity, is a leading cause of disability, and is more prevalent among veterans compared with the general population.1,2,4-6 Estimates from 2021 suggest the prevalence of chronic pain among veterans exceeds 30%; > 11% experienced high-impact chronic pain.1
Primary care practitioners (PCPs) have a prominent role in chronic pain management. Pharmacologic options for treating pain, once a mainstay of therapy, present several challenges for patients and PCPs, including drug-drug interactions and adverse effects.7 The US opioid epidemic and shift to a biopsychosocial model of chronic pain care have increased emphasis on nonpharmacologic treatment options.8,9 These include integrative modalities, which incorporate conventional approaches with an array of complementary health approaches.10-12
Integrative therapy is a prominent feature in whole person care, which may be best exemplified by the US Department of Veterans Affairs (VA) Whole Health System of care.13-14 Whole health empowers an individual to take charge of their health and well-being so they can “live their life to the fullest.”14 As implemented in the Veterans Health Administration (VHA), whole health includes the use of evidence-based
METHODS
Using a cross-sectional survey design, PCPs and patients with chronic back pain affiliated with the VA Ann Arbor Healthcare System were invited to participate in separate but similar surveys to assess knowledge, interest, and use of nonpharmacologic integrative modalities for the treatment of chronic pain. In May, June, and July 2023, 78 PCPs received 3 email
Both survey instruments are available upon request, were developed by the study team, and included a mix of yes/no questions, “select all that apply” items, Likert scale response items, and open-ended questions. For one question about which modalities they would like available, the respondent was instructed to select up to 5 modalities. The instruments were extensively pretested by members of the study team, which included 2 PCPs and a nonveteran with chronic back pain.
The list of integrative modalities included in the survey was derived from the tier 1 and tier 2 complementary and integrative health modalities identified in a VHA Directive on complementary and integrative health.15,16 Tier 1 approaches are considered to have sufficient evidence and must be made available to veterans either within a VA medical facility or in the community. Tier 2 approaches are generally considered safe and may be made available but do not have sufficient evidence to mandate their provision. For participant ease, the integrative modalities were divided into 5 subgroups: manual therapies, energy/biofield therapies, mental health therapies, nutrition counseling, and movement therapies. The clinician survey assessed clinicians’ training and interest, clinical and personal use, and perceived barriers to providing integrative modalities for chronic pain. Professional and personal demographic data were also collected. Similarly, the patient survey assessed use of integrative therapies, perceptions of and interest in integrative modalities, and potential barriers to use. Demographic and health-related information was also collected.
Data analysis included descriptive statistics (eg, frequency counts, means, medians) and visual graphic displays. Separate analyses were conducted for clinicians and patients in addition to a comparative analysis of the use and potential interest in integrative modalities. Analysis were conducted using R software. This study was deemed nonresearch quality improvement by the VA Ann Arbor Healthcare System facility research oversight board and institutional review board approval was not solicited.
RESULTS
Twenty-eight clinicians completed the survey, yielding a participation rate of 36%. Participating clinicians had a median (IQR) age of 48 years (9.5), 15 self-identified as White (54%), 8 as Asian (29%), 15 as female (54%), 26 as non-Hispanic (93%), and 25 were medical doctors or doctors of osteopathy (89%). Nineteen (68%) worked at the main hospital outpatient clinic, and 9 practiced at community-based outpatient clinics (CBOCs). Thirteen respondents (46%) reported having no formal education or training in integrative approaches. Among those with prior training, 8 clinicians had nutrition counseling (29%) and 7 had psychologic therapy training (25%). Thirteen respondents (46%) also reported using integrative modalities for personal health needs: 8 used psychological therapies, 8 used movement therapies, 10 used integrative modalities for stress management or relaxation, and 8 used them for physical symptoms (Table 1).

Overall, 85 of 200 patients (43%) responded to the study survey. Two patients indicated they did not have chronic back pain and were excluded. Patients had a median (IQR) age of 66 (20) years, with 66 self-identifying as White (80%), 69 as male (83%), and 66 as non-Hispanic (80%). Forty-four patients (53%) received care at CBOCs. Forty-seven patients reported excellent, very good, or good overall health (57%), while 53 reported excellent, very good, or good mental health (64%). Fifty-nine patients reported back pain duration > 5 years (71%), and 67 (81%) indicated experiencing back pain flare-ups at least once per week over the previous 12 months. Sixty patients (72%) indicated they were somewhat or very interested in using integrative therapies as a back pain treatment; however, 40 patients (48%) indicated they had not received information about these therapies. Among those who indicated they had received information, the most frequently reported source was their PCP (41%). Most patients (72%) also reported feeling somewhat to very comfortable discussing integrative medicine therapies with their PCP.
Integrative Therapy Recommendations and Use
PCPs reported recommending multiple integrative modalities: 23 (82%) recommended cognitive-behavioral therapy, 22 (79%) recommended acupuncture, 21 (75%) recommended chiropractic, 19 (68%) recommended battlefield acupuncture, recommended massage 18 (64%), 17 (61%) recommended meditation or mindfulness, and 15 (54%) recommended movement therapies such as yoga or tai chi/qigong (Figure 1). The only therapies used by at least half of the patients were chiropractic used by 59 patients (71%) and acupuncture by 42 patients (51%). Thirty-eight patients (46%) reported massage use and 21 patients (25%) used cognitive-behavioral therapy (Table 2).


Integrative Therapies Desired
A majority of PCPs identified acupuncture (n = 20, 71%), chiropractic (n = 19, 68%), and massage (n = 19, 68%) as therapies they would most like to have available for patients with chronic pain (Figure 2). Similarly, patients identified massage (n = 42, 51%), chiropractic (n = 34, 41%), and acupuncture (n = 27, 33%) as most desired. Seventeen patients (21%) expressed interest in movement therapies.

Barriers to Integrative Therapies Use
When asked about barriers to use, 26 PCPs (93%) identified access to services as a somewhat or extremely likely barrier, and 22 identified time constraints (79%) (Table 3). However, 17 PCPs (61%) noted lack of familiarity, and 18 (64%) noted a lack of scientific evidence as barriers to recommending integrative modalities. Among patients, 33 (40%) indicated not knowing what services were available at their facility as a barrier, 32 (39%) were not familiar with specific therapies, and 21 (25%) indicated a lack of clarity about the benefits of a specific therapy. Only 14 patients (17%) indicated that there were no obstacles to use.

DISCUSSION
Use of integrative therapies, including complementary treatments, is an increasingly important part of chronic pain management. This survey study suggests VA PCPs are willing to recommend integrative therapies and patients with chronic back pain both desire and use several therapies. Moreover, both groups expressed interest in greater availability of similar therapies. The results also highlight key barriers, such as knowledge gaps, that should be addressed to increase the uptake of integrative modalities for managing chronic pain.
An increasing number of US adults are using complementary health approaches, an important component of integrative therapy.12 This trend includes an increase in use for pain management, from 42.3% in 2002 to 49.2% in 2022; chiropractic care, acupuncture, and massage were most frequently used.12 Similarly, chiropractic, acupuncture and massage were most often used by this sample of veterans with chronic back pain and were identified by the highest percentages of PCPs and patients as the therapies they would most like available.
There were areas where the opinions of patients and clinicians differed. As has been seen previously reported, clinicians largely recommended cognitive-behavioral therapy while patients showed less interest.17 Additionally, while patients expressed interest in the availability of movement therapies, such as yoga, PCPs expressed more interest in other strategies, such as trigger point injections. These differences may reflect true preference or a tendency for clinicians and patients to select therapies with which they are more familiar. Additional research is needed to better understand the acceptability and potential use of integrative health treatments across a broad array of therapeutic options.
Despite VHA policy requiring facilities to provide certain complementary and integrative health modalities, almost all PCPs identified access to services as a major obstacle.15 Based on evidence and a rigorous vetting process, services currently required on-site, via telehealth, or through community partners include acupuncture and battlefield acupuncture (battlefield auricular acupuncture), biofeedback, clinical hypnosis, guided imagery, medical massage therapy, medication, tai chi/qigong, and yoga. Optional approaches, which may be made available to veterans, include chiropractic and healing touch. Outside the VHA, some states have introduced or enacted legislation mandating insurance coverage of nonpharmacological pain treatments.18 However, these requirements and mandates do not help address challenges such as the availability of trained/qualified practitioners.19,20 Ensuring access to complementary and integrative health treatments requires a more concerted effort to ensure that supply meets demand. It is also important to acknowledge the budgetary and physical space constraints that further limit access to services. Although expansion and integration of integrative medicine services remain a priority within the VA Whole Health program, implementation is contingent on available financial and infrastructure resources.
Time was also identified by PCPs as a barrier to recommending integrative therapies to patients. Developing and implementing time-efficient communication strategies for patient education such as concise talking points and informational handouts could help address this barrier. Furthermore, leveraging existing programs and engaging the entire health care team in patient education and referral could help increase integrative and complementary therapy uptake and use.
Although access and time were identified as major barriers, these findings also suggest that PCP and patient knowledge are another target area for enhancing the use of complementary and integrative therapies. Like prior research, most clinicians identified a lack of familiarity with certain services and a lack of scientific evidence as extremely or somewhat likely to affect their ability to offer integrative services to patients with chronic pain.21 Likewise, about 40% of patients identified being unfamiliar with a specific therapy as one of the major obstacles to receiving integrative therapies, with a similar number identifying PCPs as a source of information. The lack of familiarity may be due in part to the evolving nomenclature, with terms such as alternative, complementary, and integrative used to describe approaches outside what is often considered conventional medicine.10 On the other hand, there has also been considerable expansion in the number of therapies within this domain, along with an expanding evidence base. This suggests a need for targeted educational strategies for clinicians and patients, which can be rapidly deployed and continuously adapted as new therapies and evidence emerge.
Limitations
There are some inherent limitations with a survey-based approach, including sampling, non-response, and social desirability biases. In addition, this study only included PCPs and patients affiliated with a single VA medical center. Steps to mitigate these limitations included maintaining survey anonymity and reporting information about respondent characteristics to enhance transparency about the representativeness of the study findings.
CONCLUSIONS
Expanding the use of nonpharmacological pain treatments, including integrative modalities, is essential for safe and effective chronic pain management and reducing opioid use. Our findings show that VA PCPs and patients with chronic back pain are interested in and have some experience with certain integrative therapies. However, even within the context of a health care system that supports the use of integrative therapies for chronic pain as part of whole person care, increasing uptake will require addressing access and time-related constraints as well as ongoing clinician and patient education.
- Rikard SM, Strahan AE, Schmit KM, et al. Chronic pain among adults — United States, 2018-2021. MMWR Morb Mortal Wkly Rep. 2023;72:379-385. doi:10.15585/mmwr.mm7215a1
- Yong RJ, Mullins PM, Bhattacharyya N. Prevalence of chronic pain among adults in the United States. Pain. 2022;163:E328-E332. doi:10.1097/j.pain.0000000000002291
- Nahin RL, Feinberg T, Kapos FP, Terman GW. Estimated rates of incident and persistent chronic pain among US adults, 2019-2020. JAMA Netw Open. 2023;6:e2313563. doi:10.1001/jamanetworkopen.2023.13563
- Ferrari AJ, Santomauro DF, Aali A, et al. Global incidence, prevalence, years lived with disability (YLDs), disability-adjusted life-years (DALYs), and healthy life expectancy (HALE) for 371 diseases and injuries in 204 countries and territories and 811 subnational locations, 1990–2021: a systematic analysis for the Global Burden of Disease Study 2021. The Lancet. 2024;403:2133-2161. doi:10.1016/S0140-6736(24)00757-8 5.
- Qureshi AR, Patel M, Neumark S, et al. Prevalence of chronic non-cancer pain among military veterans: a systematic review and meta-analysis of observational studies. BMJ Mil Health. 2025;171:310-314. doi:10.1136/military-2023-002554
- Feldman DE, Nahin RL. Disability among persons with chronic severe back pain: results from a nationally representative population-based sample. J Pain. 2022;23:2144-2154. doi:10.1016/j.jpain.2022.07.016
- Qaseem A, Wilt TJ, McLean RM, Forciea MA. Noninvasive treatments for acute, subacute, and chronic low back pain: a clinical practice guideline from the American College of Physicians. Ann Intern Med. 2017;166:514-530. doi:10.7326/M16-2367
- van Erp RMA, Huijnen IPJ, Jakobs MLG, Kleijnen J, Smeets RJEM. Effectiveness of primary care interventions using a biopsychosocial approach in chronic low back pain: a systematic review. Pain Practice. 2019;19:224-241. doi:10.1111/papr.12735
- Chou R, Deyo R, Friedly J, et al. Nonpharmacologic therapies for low back pain: a systematic review for an American College of physicians clinical practice guideline. Ann Intern Med. 2017;166:493-505. doi:10.7326/M16-2459
- Complementary, alternative, or integrative health: what’s in a name? National Institutes of Health, National Center for Complementary and Integrative Health. Updated April 2021. Accessed December 15, 2025. https://www.nccih.nih.gov/health/complementary-alternative-or-integrative-health-whats-in-a-name.
- Taylor SL, Elwy AR. Complementary and alternative medicine for US veterans and active duty military personnel promising steps to improve their health. Med Care. 2014;52:S1-S4. doi:10.1097/MLR.0000000000000270.
- Nahin RL, Rhee A, Stussman B. Use of complementary health approaches overall and for pain management by US adults. JAMA. 2024;331:613-615. doi:10.1001/jama.2023.26775
- Gantt CJ, Donovan N, Khung M. Veterans Affairs’ Whole Health System of Care for transitioning service members and veterans. Mil Med. 2023;188:28-32. doi:10.1093/milmed/usad047
- Bokhour BG, Hyde J, Kligler B, et al. From patient outcomes to system change: evaluating the impact of VHA’s implementation of the Whole Health System of Care. Health Serv Res. 2022;57:53-65. doi:10.1111/1475-6773.13938
- Department of Veterans Affairs VHA. VHA Policy Directive 1137: Provision of Complementary and Integrative Health. December 2022. Accessed December 15, 2025. https://www.va.gov/VHApublications/ViewPublication.asp?pub_ID=10072
- Giannitrapani KF, Holliday JR, Miake-Lye IM, Hempel S, Taylor SL. Synthesizing the strength of the evidence of complementary and integrative health therapies for pain. Pain Med. 2019;20:1831-1840. doi:10.1093/pm/pnz068
- Belitskaya-Levy I, David Clark J, Shih MC, Bair MJ. Treatment preferences for chronic low back pain: views of veterans and their providers. J Pain Res. 2021;14:161-171. doi:10.2147/JPR.S290400
- Onstott TN, Hurst S, Kronick R, Tsou AC, Groessl E, McMenamin SB. Health insurance mandates for nonpharmacological pain treatments in 7 US states. JAMA Netw Open. 2024;7:E245737. doi:10.1001/jamanetworkopen.2024.5737
- Sullivan M, Leach M, Snow J, Moonaz S. The North American yoga therapy workforce survey. Complement Ther Med. 2017;31:39-48. doi:10.1016/j.ctim.2017.01.006
- Bolton R, Ritter G, Highland K, Larson MJ. The relationship between capacity and utilization of nonpharmacologic therapies in the US Military Health System. BMC Health Serv Res. 2022;22. doi:10.1186/s12913-022-07700-4
- Stussman BJ, Nahin RL, Barnes PM, Scott R, Feinberg T, Ward BW. Reasons office-based physicians in the United States recommend common complementary health approaches to patients: an exploratory study using a national survey. J Integr Complement Med. 2022;28:651-663. doi:10.1089/jicm.2022.0493
- Rikard SM, Strahan AE, Schmit KM, et al. Chronic pain among adults — United States, 2018-2021. MMWR Morb Mortal Wkly Rep. 2023;72:379-385. doi:10.15585/mmwr.mm7215a1
- Yong RJ, Mullins PM, Bhattacharyya N. Prevalence of chronic pain among adults in the United States. Pain. 2022;163:E328-E332. doi:10.1097/j.pain.0000000000002291
- Nahin RL, Feinberg T, Kapos FP, Terman GW. Estimated rates of incident and persistent chronic pain among US adults, 2019-2020. JAMA Netw Open. 2023;6:e2313563. doi:10.1001/jamanetworkopen.2023.13563
- Ferrari AJ, Santomauro DF, Aali A, et al. Global incidence, prevalence, years lived with disability (YLDs), disability-adjusted life-years (DALYs), and healthy life expectancy (HALE) for 371 diseases and injuries in 204 countries and territories and 811 subnational locations, 1990–2021: a systematic analysis for the Global Burden of Disease Study 2021. The Lancet. 2024;403:2133-2161. doi:10.1016/S0140-6736(24)00757-8 5.
- Qureshi AR, Patel M, Neumark S, et al. Prevalence of chronic non-cancer pain among military veterans: a systematic review and meta-analysis of observational studies. BMJ Mil Health. 2025;171:310-314. doi:10.1136/military-2023-002554
- Feldman DE, Nahin RL. Disability among persons with chronic severe back pain: results from a nationally representative population-based sample. J Pain. 2022;23:2144-2154. doi:10.1016/j.jpain.2022.07.016
- Qaseem A, Wilt TJ, McLean RM, Forciea MA. Noninvasive treatments for acute, subacute, and chronic low back pain: a clinical practice guideline from the American College of Physicians. Ann Intern Med. 2017;166:514-530. doi:10.7326/M16-2367
- van Erp RMA, Huijnen IPJ, Jakobs MLG, Kleijnen J, Smeets RJEM. Effectiveness of primary care interventions using a biopsychosocial approach in chronic low back pain: a systematic review. Pain Practice. 2019;19:224-241. doi:10.1111/papr.12735
- Chou R, Deyo R, Friedly J, et al. Nonpharmacologic therapies for low back pain: a systematic review for an American College of physicians clinical practice guideline. Ann Intern Med. 2017;166:493-505. doi:10.7326/M16-2459
- Complementary, alternative, or integrative health: what’s in a name? National Institutes of Health, National Center for Complementary and Integrative Health. Updated April 2021. Accessed December 15, 2025. https://www.nccih.nih.gov/health/complementary-alternative-or-integrative-health-whats-in-a-name.
- Taylor SL, Elwy AR. Complementary and alternative medicine for US veterans and active duty military personnel promising steps to improve their health. Med Care. 2014;52:S1-S4. doi:10.1097/MLR.0000000000000270.
- Nahin RL, Rhee A, Stussman B. Use of complementary health approaches overall and for pain management by US adults. JAMA. 2024;331:613-615. doi:10.1001/jama.2023.26775
- Gantt CJ, Donovan N, Khung M. Veterans Affairs’ Whole Health System of Care for transitioning service members and veterans. Mil Med. 2023;188:28-32. doi:10.1093/milmed/usad047
- Bokhour BG, Hyde J, Kligler B, et al. From patient outcomes to system change: evaluating the impact of VHA’s implementation of the Whole Health System of Care. Health Serv Res. 2022;57:53-65. doi:10.1111/1475-6773.13938
- Department of Veterans Affairs VHA. VHA Policy Directive 1137: Provision of Complementary and Integrative Health. December 2022. Accessed December 15, 2025. https://www.va.gov/VHApublications/ViewPublication.asp?pub_ID=10072
- Giannitrapani KF, Holliday JR, Miake-Lye IM, Hempel S, Taylor SL. Synthesizing the strength of the evidence of complementary and integrative health therapies for pain. Pain Med. 2019;20:1831-1840. doi:10.1093/pm/pnz068
- Belitskaya-Levy I, David Clark J, Shih MC, Bair MJ. Treatment preferences for chronic low back pain: views of veterans and their providers. J Pain Res. 2021;14:161-171. doi:10.2147/JPR.S290400
- Onstott TN, Hurst S, Kronick R, Tsou AC, Groessl E, McMenamin SB. Health insurance mandates for nonpharmacological pain treatments in 7 US states. JAMA Netw Open. 2024;7:E245737. doi:10.1001/jamanetworkopen.2024.5737
- Sullivan M, Leach M, Snow J, Moonaz S. The North American yoga therapy workforce survey. Complement Ther Med. 2017;31:39-48. doi:10.1016/j.ctim.2017.01.006
- Bolton R, Ritter G, Highland K, Larson MJ. The relationship between capacity and utilization of nonpharmacologic therapies in the US Military Health System. BMC Health Serv Res. 2022;22. doi:10.1186/s12913-022-07700-4
- Stussman BJ, Nahin RL, Barnes PM, Scott R, Feinberg T, Ward BW. Reasons office-based physicians in the United States recommend common complementary health approaches to patients: an exploratory study using a national survey. J Integr Complement Med. 2022;28:651-663. doi:10.1089/jicm.2022.0493
Primary Care Clinician and Patient Knowledge, Interest, and Use of Integrative Treatment Options for Chronic Low Back Pain Management
Primary Care Clinician and Patient Knowledge, Interest, and Use of Integrative Treatment Options for Chronic Low Back Pain Management
Rural Cancer Survivors Are More Likely to Have Chronic Pain
TOPLINE:
Rural cancer survivors experience significantly higher rates of chronic pain at 43.0% than those among urban survivors at 33.5%. Even after controlling for demographics and health conditions, rural residents showed 21% higher odds of experiencing chronic pain.
METHODOLOGY:
- Chronic pain prevalence among cancer survivors is twice that of the general US population and is associated with numerous negative outcomes. Rural residence is frequently linked to debilitating long-term survivorship effects, and current data lack information on whether chronic pain disparity exists specifically for rural cancer survivors.
- Researchers pooled data from the 2019–2021 and 2023 National Health Interview Survey, a cross–sectional survey conducted by the National Center for Health Statistics.
- Analysis included 5542 adult cancer survivors diagnosed within the previous 5 years, with 51.6% female participants and 48.4% male participants.
- Chronic pain was defined as pain experienced on most or all days over the past 3 months, following National Center for Health Statistics conventions.
- Rural residence classification was based on noncore or nonmetropolitan counties using the modified National Center for Health Statistics Urban–Rural Classification Scheme for Counties.
TAKEAWAY:
- Rural cancer survivors showed significantly higher odds of experiencing chronic pain compared with urban survivors (odds ratio [OR], 1.21; 95% CI, 1.01-1.45).
- Rural survivors were more likely to be non–Hispanic White, have less than a 4-year college degree, have an income below 200% of the federal poverty level, and have slightly more chronic health conditions.
- Having an income below 100% of the federal poverty level was associated with doubled odds of chronic pain (OR, 2.07; 95% CI, 1.54-2.77) compared with having an income at least four times the federal poverty level.
- Each additional health condition increased the odds of experiencing chronic pain by 32% (OR, 1.32; 95% CI, 1.26-1.39).
IN PRACTICE:
“Policymakers and health systems should work to close this gap by increasing the availability of pain management resources for rural cancer survivors. Approaches could include innovative payment models for integrative medicine in rural areas or supporting rural clinician access to pain specialists,” the authors of the study wrote.
SOURCE:
This study was led by Hyojin Choi, PhD, Department of Family Medicine, The Robert Larner MD College of Medicine, University of Vermont in Burlington, Vermont. It was published online in JAMA Network Open.
LIMITATIONS:
The authors note that the cross–sectional design of the study and limited information on individual respondents’ use of multimodal pain treatment options constrain the interpretation of findings.
DISCLOSURES:
The authors did not report any relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
Rural cancer survivors experience significantly higher rates of chronic pain at 43.0% than those among urban survivors at 33.5%. Even after controlling for demographics and health conditions, rural residents showed 21% higher odds of experiencing chronic pain.
METHODOLOGY:
- Chronic pain prevalence among cancer survivors is twice that of the general US population and is associated with numerous negative outcomes. Rural residence is frequently linked to debilitating long-term survivorship effects, and current data lack information on whether chronic pain disparity exists specifically for rural cancer survivors.
- Researchers pooled data from the 2019–2021 and 2023 National Health Interview Survey, a cross–sectional survey conducted by the National Center for Health Statistics.
- Analysis included 5542 adult cancer survivors diagnosed within the previous 5 years, with 51.6% female participants and 48.4% male participants.
- Chronic pain was defined as pain experienced on most or all days over the past 3 months, following National Center for Health Statistics conventions.
- Rural residence classification was based on noncore or nonmetropolitan counties using the modified National Center for Health Statistics Urban–Rural Classification Scheme for Counties.
TAKEAWAY:
- Rural cancer survivors showed significantly higher odds of experiencing chronic pain compared with urban survivors (odds ratio [OR], 1.21; 95% CI, 1.01-1.45).
- Rural survivors were more likely to be non–Hispanic White, have less than a 4-year college degree, have an income below 200% of the federal poverty level, and have slightly more chronic health conditions.
- Having an income below 100% of the federal poverty level was associated with doubled odds of chronic pain (OR, 2.07; 95% CI, 1.54-2.77) compared with having an income at least four times the federal poverty level.
- Each additional health condition increased the odds of experiencing chronic pain by 32% (OR, 1.32; 95% CI, 1.26-1.39).
IN PRACTICE:
“Policymakers and health systems should work to close this gap by increasing the availability of pain management resources for rural cancer survivors. Approaches could include innovative payment models for integrative medicine in rural areas or supporting rural clinician access to pain specialists,” the authors of the study wrote.
SOURCE:
This study was led by Hyojin Choi, PhD, Department of Family Medicine, The Robert Larner MD College of Medicine, University of Vermont in Burlington, Vermont. It was published online in JAMA Network Open.
LIMITATIONS:
The authors note that the cross–sectional design of the study and limited information on individual respondents’ use of multimodal pain treatment options constrain the interpretation of findings.
DISCLOSURES:
The authors did not report any relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
Rural cancer survivors experience significantly higher rates of chronic pain at 43.0% than those among urban survivors at 33.5%. Even after controlling for demographics and health conditions, rural residents showed 21% higher odds of experiencing chronic pain.
METHODOLOGY:
- Chronic pain prevalence among cancer survivors is twice that of the general US population and is associated with numerous negative outcomes. Rural residence is frequently linked to debilitating long-term survivorship effects, and current data lack information on whether chronic pain disparity exists specifically for rural cancer survivors.
- Researchers pooled data from the 2019–2021 and 2023 National Health Interview Survey, a cross–sectional survey conducted by the National Center for Health Statistics.
- Analysis included 5542 adult cancer survivors diagnosed within the previous 5 years, with 51.6% female participants and 48.4% male participants.
- Chronic pain was defined as pain experienced on most or all days over the past 3 months, following National Center for Health Statistics conventions.
- Rural residence classification was based on noncore or nonmetropolitan counties using the modified National Center for Health Statistics Urban–Rural Classification Scheme for Counties.
TAKEAWAY:
- Rural cancer survivors showed significantly higher odds of experiencing chronic pain compared with urban survivors (odds ratio [OR], 1.21; 95% CI, 1.01-1.45).
- Rural survivors were more likely to be non–Hispanic White, have less than a 4-year college degree, have an income below 200% of the federal poverty level, and have slightly more chronic health conditions.
- Having an income below 100% of the federal poverty level was associated with doubled odds of chronic pain (OR, 2.07; 95% CI, 1.54-2.77) compared with having an income at least four times the federal poverty level.
- Each additional health condition increased the odds of experiencing chronic pain by 32% (OR, 1.32; 95% CI, 1.26-1.39).
IN PRACTICE:
“Policymakers and health systems should work to close this gap by increasing the availability of pain management resources for rural cancer survivors. Approaches could include innovative payment models for integrative medicine in rural areas or supporting rural clinician access to pain specialists,” the authors of the study wrote.
SOURCE:
This study was led by Hyojin Choi, PhD, Department of Family Medicine, The Robert Larner MD College of Medicine, University of Vermont in Burlington, Vermont. It was published online in JAMA Network Open.
LIMITATIONS:
The authors note that the cross–sectional design of the study and limited information on individual respondents’ use of multimodal pain treatment options constrain the interpretation of findings.
DISCLOSURES:
The authors did not report any relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
A True Community: The Vet-to-Vet Program for Chronic Pain
A True Community: The Vet-to-Vet Program for Chronic Pain
The Veterans Health Administration (VHA) has continued to advance its understanding and treatment of chronic pain. The VHA National Pain Management Strategy emphasizes the significance of the social context of pain while underscoring the importance of self-management.1 This established strategy ensures that all veterans have access to the appropriate pain care in the proper setting.2 VHA has instituted a stepped care model of pain management, delineating the domains of primary care, secondary consultative services, and tertiary care.3 This directive emphasized a biopsychosocial approach to pain management to prioritize the relationship between biological, psychological, and social factors that influence how veterans experience pain and should commensurately influence how it is managed.
The VHA Office of Patient-Centered Care and Cultural Transformation implemented the Whole Health System of Care as part of the Comprehensive Addiction and Recovery Act, which included a VHA directive to expand pain management.4,5 Reorientation within this system shifts from defining veterans as passive care recipients to viewing them as active partners in their own care and health. This partnership places additional emphasis on peer-led explorations of mission, aspiration, and purpose.6
Peer-led groups, also known as mutual aid, mutual support, and mutual help groups, have historically been successful for patients undergoing treatment for substance use disorders (eg, Alcoholics Anonymous).7 Mutual help groups have 3 defining characteristics. First, they are run by participants, not professionals, though the latter may have been integral in the founding of the groups. Second, participants share a similar problem (eg, disease state, experience, disposition). Finally, there is a reciprocal exchange of information and psychological support among participants.8,9 Mutual help groups that address chronic pain are rare but becoming more common.10-12 Emerging evidence suggests a positive relationship between peer support and improved well-being, self-efficacy, pain management, and pain self-management skills (eg, activity pacing).13-15
Storytelling as a tool for healing has a long history in indigenous and Western medical traditions.16-19 This includes the treatment of chronic disease, including pain.20,21 The use of storytelling in health care overlaps with the role it plays within many mutual help groups focused on chronic disease treatment.22 Storytelling allows an individual to share their experience with a disease, and take a more active role in their health, and facilitate stronger bonds with others.22 In effect, storytelling is not only important to group cohesion—it also plays a role in an individual’s healing.
Vet-to-Vet
The VHA Office of Rural Health funds Vet-to-Vet, a peer-to-peer program to address limited access to care for rural veterans with chronic pain. Similar to the VHA National Pain Management Strategy, Vet-to-Vet is grounded in the significance of the social context of pain and underscores the importance of self-management.1 The program combines pain care, mutual help, and storytelling to support veterans living with chronic pain. While the primary focus of Vet-to-Vet is rural veterans, the program serves any veteran experiencing chronic pain who is isolated from services, including home-bound urban veterans.
Following mutual help principles, Vet-to-Vet peer facilitators lead weekly online drop-in meetings. Meetings follow the general structure of reiterating group ground rules and sharing an individual pain story, followed by open discussions centered on well-being, chronic pain management, or any topic the group wishes to discuss. Meetings typically end with a mindfulness exercise. The organizational structure that supports Vet-to-Vet includes the implementation support team, site leads, Vet-to-Vet peer facilitators, and national partners (Figure 1).
Implementation Support Team
The implementation support team consists of a principal investigator, coinvestigator, program manager, and program support specialist. The team provides facilitator training, monthly community practice sessions for Vet-to-Vet peer facilitators and site leads, and weekly office hours for site leads. The implementation support team also recruits new Vet-to-Vet sites; potential new locations ideally have an existing whole health program, leadership support, committed site and cosite leads, and ≥ 3 peer facilitator volunteers.
Site Leads
Most site and cosite leads are based in whole health or pain management teams and are whole health coaches or peer support specialists. The site lead is responsible for standing up the program and documenting encounters, recruiting and supporting peer facilitators and participants, and overseeing the meeting. During meetings, site leads generally leave their cameras off and only speak when called into the group; the peer facilitators lead the meetings. The implementation support team recommends that site leads dedicate ≥ 4 hours per week to Vet-to-Vet; 2 hours for weekly group meetings and 2 hours for documentation (ie, entering notes into the participants’ electronic health records) and supporting peer facilitators and participants. Cosite lead responsibilities vary by location, with some sites having 2 leads that equally share duties and others having a primary lead and a colead available if the site lead is unable to attend a meeting.
Vet-to-Vet Peer Facilitators
Peer facilitators are the core of the program. They lead meetings from start to finish. Like participants, they also experience chronic pain and are volunteers. The implementation support team encourages sites to establish volunteer peer facilitators, rather than assigning peer support specialists to facilitate meetings. Veterans are eager to connect and give back to their communities, and the Vet-to-Vet peer facilitator role is an opportunity for those unable to work to connect with peers and add meaning to their lives. Even if a VHA employee is a veteran who has chronic pain, they are not eligible to serve as this could create a service provider/service recipient dynamic that is not in the spirit of mutual help.
Vet-to-Vet peer facilitators attend a virtual 3-day training held by the implementation support team prior to starting. These training sessions are available on a quarterly basis and facilitated by the Vet-to-Vet program manager and 2 current peer facilitators. Training content includes established whole health facilitator training materials and program-specific storytelling training materials. Once trained, peer facilitators attend storytelling practice sessions and collaborate with their site leads during weekly meetings.
Participants
Vet-to-Vet participants find the program through direct outreach from site leads, word of mouth, and referrals. The only criteria to join are that the individual is a veteran who experiences chronic pain and is enrolled in the VHA (site leads can assist with enrollment if needed). Participants are not required to have a diagnosis or engage in any other health care. There is no commitment and no end date. Some participants only come once; others have attended for > 3 years. This approach is intended to embrace the idea that the need for support ebbs and flows.
National Partners
The VHA Office of Rural Health provides technical support. The Center for Development and Civic Engagement onboards peer facilitators as VHA volunteers. The Office of Patient-Centered Care and Cultural Transformation provides national guidance and site-level collaboration. The VHA Pain Management, Opioid Safety, and Prescription Drug Monitoring Program supports site recruitment. In addition to the VHA partners, 4 veteran evaluation consultants who have experience with chronic pain but do not participate in Vet-to-Vet meetings provide advice on evaluation activities, such as question development and communication strategies.
Evaluation
This evaluation shares preliminary results from a pilot evaluation of the Rocky Mountain Regional VA Medical Center (RMRVAMC) Vet-to-Vet group. It is intended for program improvement, was deemed nonresearch by the Colorado Multiple Institutional Review Board, and was structured using the RE-AIM (Reach, Effectiveness, Adoption, Implementation, and Maintenance) framework.23 This evaluation focused on capturing measures related to reach and effectiveness, while a forthcoming evaluation includes elements of adoption, implementation, and maintenance.
In 2022, 16 Vet-to-Vet peer facilitators and participants completed surveys and interviews to share their experience. Interviews were recorded, transcribed, and coded in ATLAS.ti. A priori codes were based on interview guide questions and emergent descriptive codes were used to identify specific topics which were categorized into RE-AIM domains, barriers, facilitators, what participants learned, how participants applied what they learned to their lives, and participant reported outcomes. This article contains high-level findings from the evaluation; more detailed results will be included in the ongoing evaluation.
Results
The RMRVAMC Vet-to-Vet group has met weekly since April 2022. Four Vet-to-Vet peer facilitators and 12 individuals participated in the pilot Vet-to-Vet group and evaluation. The mean age was 62 years, most were men, and half were married. Most participants lived in rural areas with a mean distance of 125 miles to the nearest VAMC. Many experienced multiple kinds of pain, with a mean 4.5 on a 10-point scale (bothered “a lot”). All participants reported that they experienced pain daily.
Participation in Vet-to-Vet meetings was high; 3 of 4 peer facilitators and 7 of 12 participants completed the first 6 months of the program. In interviews, participants described the positive impact of the program. They emphasized the importance of connecting with other veterans and helping one another, with one noting that opportunities to connect with other veterans “just drops off a lot” (peer facilitator 3) after leaving active duty.
Some participants and Vet-to-Vet peer facilitators outlined the content of the sessions (eg, learning about how pain impacts the body and one’s family relationships) and shared the skills they learned (eg, goal setting, self-advocacy) (Table). Most spoke about learning from one another and the power of sharing stories with one peer facilitator sharing how they felt that witnessing another participant’s story “really shifted how I was thinking about things and how I perceived people” (peer facilitator 1).

Participants reported several ways the program impacted their lives, such as learning that they could get help, how to get help, and how to overcome the mental aspects of chronic pain. One veteran shared profound health impacts and attributed the Vet-to-Vet program to having one of the best years of their life. Even those who did not attend many meetings spoke of it positively and stated that it should continue so others could try (Table).
From January 2022 to September 2025, > 80 veterans attended ≥ 1 meeting at RMRVAMC; 29 attended ≥ 1 meeting in the last quarter. There were > 1400 Vet-to-Vet encounters at RMRVAMC, with a mean (SD) of 14.2 (19.2) and a median of 4.5 encounters per participant. Half of the veterans attend ≥ 5 meetings, and one-third attended ≥ 10 meetings.
Since June 2023, 15 additional VHA facilities launched Vet-to-Vet programs. As of October 2025, > 350 veterans have participated in ≥ 1 Vet-to-Vet meeting, totaling > 4500 Vet-to-Vet encounters since the program’s inception (Figure 2).
Challenges
The RMRVAMC site and cosite leads are part of the national implementation team and dedicate substantial time to developing the program: 40 and 10 hours per week, respectively. Site leads at new locations do not receive funding for Vet-to-Vet activities and are recommended to dedicate only 4 hours per week to the program. Formally embedding Vet-to-Vet into the site leads’ roles is critical for sustainment.
The Vet-to-Vet model has changed. The initial Vet-to-Vet cohort included the 6-week Taking Charge of My Life and Health curriculum prior to moving to the mutual help format.24 While this curriculum still informs peer facilitator training, it is not used in new groups. It has anecdotally been reported that this change was positive, but the impact of this adaptation is unknown.
This evaluation cohort was small (16 participants) and initial patient reported and administrative outcomes were inconclusive. However, most veterans who stopped participating in Vet-to-Vet spoke fondly of their experiences with the program.
CONCLUSIONS
Vet-to-Vet is a promising new initiative to support self-management and social connection in chronic pain care. The program employs a mutual help approach and storytelling to empower veterans living with chronic pain. The effectiveness of these strategies will be evaluated, which will inform its continued growth. The program's current goals focus on sustainment at existing sites and expansion to new sites to reach more rural veterans across the VA enterprise. While Vet-to-Vet is designed to serve those who experience chronic pain, a partnership with the Office of Whole Health has established goals to begin expanding this model to other chronic conditions in 2026.
- Kerns RD, Philip EJ, Lee AW, Rosenberger PH. Implementation of the Veterans Health Administration national pain management strategy. Transl Behav Med. 2011;1:635-643. doi:10.1007/s13142-011-0094-3
- Pain Management, Opioid Safety, and PDMP (PMOP). US Department of Veterans Affairs. Updated August 21, 2025. Accessed September 25, 2025. https://www.va.gov/PAINMANAGEMENT/Providers/IntegratedTeambasedPainCare.asp
- US Department of Veterans Affairs. VHA Directive 2009-053. October 28, 2009. Accessed September 25, 2025. https://www.va.gov/PAINMANAGEMENT/docs/VHA09PainDirective.pdf
- Comprehensive Addiction and Recovery Act of 2016, S524, 114th Cong (2015-2016). Pub L No. 114-198. July 22, 2016. Accessed September 25, 2025. https://www.congress.gov/bill/114th-congress/senate-bill/524
- Bokhour B, Hyde J, Zeliadt, Mohr D. Whole Health System of Care Evaluation. US Department of Veterans Affairs. February 18, 2020. Accessed September 25, 2025. https://www.va.gov/WHOLEHEALTH/docs/EPCC_WHSevaluation_FinalReport_508.pdf
- Gaudet T, Kligler B. Whole health in the whole system of the veterans administration: how will we know we have reached this future state? J Altern Complement Med. 2019;25:S7-S11. doi:10.1089/acm.2018.29061.gau
- Kelly JF, Yeterian JD. The role of mutual-help groups in extending the framework of treatment. Alcohol Res Health. 2011;33:350-355.
- Humphreys K. Self-help/mutual aid organizations: the view from Mars. Subst Use Misuse. 1997;32:2105-2109. doi:10.3109/10826089709035622
- Chinman M, Kloos B, O’Connell M, Davidson L. Service providers’ views of psychiatric mutual support groups. J Community Psychol. 2002;30:349-366. doi:10.1002/jcop.10010
- Shue SA, McGuire AB, Matthias MS. Facilitators and barriers to implementation of a peer support intervention for patients with chronic pain: a qualitative study. Pain Med. 2019;20:1311-1320. doi:10.1093/pm/pny229
- Pester BD, Tankha H, Caño A, et al. Facing pain together: a randomized controlled trial of the effects of Facebook support groups on adults with chronic pain. J Pain. 2022;23:2121-2134. doi:10.1016/j.jpain.2022.07.013
- Matthias MS, McGuire AB, Kukla M, Daggy J, Myers LJ, Bair MJ. A brief peer support intervention for veterans with chronic musculoskeletal pain: a pilot study of feasibility and effectiveness. Pain Med. 2015;16:81-87. doi:10.1111/pme.12571
- Finlay KA, Elander J. Reflecting the transition from pain management services to chronic pain support group attendance: an interpretative phenomenological analysis. Br J Health Psychol. 2016;21:660-676. doi:10.1111/bjhp.12194
- Finlay KA, Peacock S, Elander J. Developing successful social support: an interpretative phenomenological analysis of mechanisms and processes in a chronic pain support group. Psychol Health. 2018;33:846-871. doi:10.1080/08870446.2017.1421188
- Farr M, Brant H, Patel R, et al. Experiences of patient-led chronic pain peer support groups after pain management programs: a qualitative study. Pain Med. 2021;22:2884-2895. doi:10.1093/pm/pnab189
- Mehl-Madrona L. Narrative Medicine: The Use of History and Story in the Healing Process. Bear & Company; 2007.
- Fioretti C, Mazzocco K, Riva S, Oliveri S, Masiero M, Pravettoni G. Research studies on patients’ illness experience using the Narrative Medicine approach: a systematic review. BMJ Open. 2016;6:e011220. doi:10.1136/bmjopen-2016-011220
- Hall JM, Powell J. Understanding the person through narrative. Nurs Res Pract. 2011;2011:293837. doi:10.1155/2011/293837
- Ricks L, Kitchens S, Goodrich T, Hancock E. My story: the use of narrative therapy in individual and group counseling. J Creat Ment Health. 2014;9:99-110. doi:10.1080/15401383.2013.870947
- Hydén L-C. Illness and narrative. Sociol Health Illn. 1997;19:48-69. doi:10.1111/j.1467-9566.1997.tb00015.x
- Georgiadis E, Johnson MI. Incorporating personal narratives in positive psychology interventions to manage chronic pain. Front Pain Res (Lausanne). 2023;4:1253310. doi:10.3389/fpain.2023.1253310
- Gucciardi E, Jean-Pierre N, Karam G, Sidani S. Designing and delivering facilitated storytelling interventions for chronic disease self-management: a scoping review. BMC Health Serv Res. 2016;16:249. doi:10.1186/s12913-016-1474-7
- Glasgow RE, Vogt TM, Boles SM. Evaluating the public health impact of health promotion interventions: the RE-AIM framework. Am J Public Health. 1999;89:1322-1327. doi:10.2105/ajph.89.9.1322
- Abadi M, Richard B, Shamblen S, et al. Achieving whole health: a preliminary study of TCMLH, a group-based program promoting self-care and empowerment among veterans. Health Educ Behav. 2022;49:347-357. doi:10.1177/10901981211011043
The Veterans Health Administration (VHA) has continued to advance its understanding and treatment of chronic pain. The VHA National Pain Management Strategy emphasizes the significance of the social context of pain while underscoring the importance of self-management.1 This established strategy ensures that all veterans have access to the appropriate pain care in the proper setting.2 VHA has instituted a stepped care model of pain management, delineating the domains of primary care, secondary consultative services, and tertiary care.3 This directive emphasized a biopsychosocial approach to pain management to prioritize the relationship between biological, psychological, and social factors that influence how veterans experience pain and should commensurately influence how it is managed.
The VHA Office of Patient-Centered Care and Cultural Transformation implemented the Whole Health System of Care as part of the Comprehensive Addiction and Recovery Act, which included a VHA directive to expand pain management.4,5 Reorientation within this system shifts from defining veterans as passive care recipients to viewing them as active partners in their own care and health. This partnership places additional emphasis on peer-led explorations of mission, aspiration, and purpose.6
Peer-led groups, also known as mutual aid, mutual support, and mutual help groups, have historically been successful for patients undergoing treatment for substance use disorders (eg, Alcoholics Anonymous).7 Mutual help groups have 3 defining characteristics. First, they are run by participants, not professionals, though the latter may have been integral in the founding of the groups. Second, participants share a similar problem (eg, disease state, experience, disposition). Finally, there is a reciprocal exchange of information and psychological support among participants.8,9 Mutual help groups that address chronic pain are rare but becoming more common.10-12 Emerging evidence suggests a positive relationship between peer support and improved well-being, self-efficacy, pain management, and pain self-management skills (eg, activity pacing).13-15
Storytelling as a tool for healing has a long history in indigenous and Western medical traditions.16-19 This includes the treatment of chronic disease, including pain.20,21 The use of storytelling in health care overlaps with the role it plays within many mutual help groups focused on chronic disease treatment.22 Storytelling allows an individual to share their experience with a disease, and take a more active role in their health, and facilitate stronger bonds with others.22 In effect, storytelling is not only important to group cohesion—it also plays a role in an individual’s healing.
Vet-to-Vet
The VHA Office of Rural Health funds Vet-to-Vet, a peer-to-peer program to address limited access to care for rural veterans with chronic pain. Similar to the VHA National Pain Management Strategy, Vet-to-Vet is grounded in the significance of the social context of pain and underscores the importance of self-management.1 The program combines pain care, mutual help, and storytelling to support veterans living with chronic pain. While the primary focus of Vet-to-Vet is rural veterans, the program serves any veteran experiencing chronic pain who is isolated from services, including home-bound urban veterans.
Following mutual help principles, Vet-to-Vet peer facilitators lead weekly online drop-in meetings. Meetings follow the general structure of reiterating group ground rules and sharing an individual pain story, followed by open discussions centered on well-being, chronic pain management, or any topic the group wishes to discuss. Meetings typically end with a mindfulness exercise. The organizational structure that supports Vet-to-Vet includes the implementation support team, site leads, Vet-to-Vet peer facilitators, and national partners (Figure 1).

Implementation Support Team
The implementation support team consists of a principal investigator, coinvestigator, program manager, and program support specialist. The team provides facilitator training, monthly community practice sessions for Vet-to-Vet peer facilitators and site leads, and weekly office hours for site leads. The implementation support team also recruits new Vet-to-Vet sites; potential new locations ideally have an existing whole health program, leadership support, committed site and cosite leads, and ≥ 3 peer facilitator volunteers.
Site Leads
Most site and cosite leads are based in whole health or pain management teams and are whole health coaches or peer support specialists. The site lead is responsible for standing up the program and documenting encounters, recruiting and supporting peer facilitators and participants, and overseeing the meeting. During meetings, site leads generally leave their cameras off and only speak when called into the group; the peer facilitators lead the meetings. The implementation support team recommends that site leads dedicate ≥ 4 hours per week to Vet-to-Vet; 2 hours for weekly group meetings and 2 hours for documentation (ie, entering notes into the participants’ electronic health records) and supporting peer facilitators and participants. Cosite lead responsibilities vary by location, with some sites having 2 leads that equally share duties and others having a primary lead and a colead available if the site lead is unable to attend a meeting.
Vet-to-Vet Peer Facilitators
Peer facilitators are the core of the program. They lead meetings from start to finish. Like participants, they also experience chronic pain and are volunteers. The implementation support team encourages sites to establish volunteer peer facilitators, rather than assigning peer support specialists to facilitate meetings. Veterans are eager to connect and give back to their communities, and the Vet-to-Vet peer facilitator role is an opportunity for those unable to work to connect with peers and add meaning to their lives. Even if a VHA employee is a veteran who has chronic pain, they are not eligible to serve as this could create a service provider/service recipient dynamic that is not in the spirit of mutual help.
Vet-to-Vet peer facilitators attend a virtual 3-day training held by the implementation support team prior to starting. These training sessions are available on a quarterly basis and facilitated by the Vet-to-Vet program manager and 2 current peer facilitators. Training content includes established whole health facilitator training materials and program-specific storytelling training materials. Once trained, peer facilitators attend storytelling practice sessions and collaborate with their site leads during weekly meetings.
Participants
Vet-to-Vet participants find the program through direct outreach from site leads, word of mouth, and referrals. The only criteria to join are that the individual is a veteran who experiences chronic pain and is enrolled in the VHA (site leads can assist with enrollment if needed). Participants are not required to have a diagnosis or engage in any other health care. There is no commitment and no end date. Some participants only come once; others have attended for > 3 years. This approach is intended to embrace the idea that the need for support ebbs and flows.
National Partners
The VHA Office of Rural Health provides technical support. The Center for Development and Civic Engagement onboards peer facilitators as VHA volunteers. The Office of Patient-Centered Care and Cultural Transformation provides national guidance and site-level collaboration. The VHA Pain Management, Opioid Safety, and Prescription Drug Monitoring Program supports site recruitment. In addition to the VHA partners, 4 veteran evaluation consultants who have experience with chronic pain but do not participate in Vet-to-Vet meetings provide advice on evaluation activities, such as question development and communication strategies.
Evaluation
This evaluation shares preliminary results from a pilot evaluation of the Rocky Mountain Regional VA Medical Center (RMRVAMC) Vet-to-Vet group. It is intended for program improvement, was deemed nonresearch by the Colorado Multiple Institutional Review Board, and was structured using the RE-AIM (Reach, Effectiveness, Adoption, Implementation, and Maintenance) framework.23 This evaluation focused on capturing measures related to reach and effectiveness, while a forthcoming evaluation includes elements of adoption, implementation, and maintenance.
In 2022, 16 Vet-to-Vet peer facilitators and participants completed surveys and interviews to share their experience. Interviews were recorded, transcribed, and coded in ATLAS.ti. A priori codes were based on interview guide questions and emergent descriptive codes were used to identify specific topics which were categorized into RE-AIM domains, barriers, facilitators, what participants learned, how participants applied what they learned to their lives, and participant reported outcomes. This article contains high-level findings from the evaluation; more detailed results will be included in the ongoing evaluation.
Results
The RMRVAMC Vet-to-Vet group has met weekly since April 2022. Four Vet-to-Vet peer facilitators and 12 individuals participated in the pilot Vet-to-Vet group and evaluation. The mean age was 62 years, most were men, and half were married. Most participants lived in rural areas with a mean distance of 125 miles to the nearest VAMC. Many experienced multiple kinds of pain, with a mean 4.5 on a 10-point scale (bothered “a lot”). All participants reported that they experienced pain daily.
Participation in Vet-to-Vet meetings was high; 3 of 4 peer facilitators and 7 of 12 participants completed the first 6 months of the program. In interviews, participants described the positive impact of the program. They emphasized the importance of connecting with other veterans and helping one another, with one noting that opportunities to connect with other veterans “just drops off a lot” (peer facilitator 3) after leaving active duty.
Some participants and Vet-to-Vet peer facilitators outlined the content of the sessions (eg, learning about how pain impacts the body and one’s family relationships) and shared the skills they learned (eg, goal setting, self-advocacy) (Table). Most spoke about learning from one another and the power of sharing stories with one peer facilitator sharing how they felt that witnessing another participant’s story “really shifted how I was thinking about things and how I perceived people” (peer facilitator 1).

Participants reported several ways the program impacted their lives, such as learning that they could get help, how to get help, and how to overcome the mental aspects of chronic pain. One veteran shared profound health impacts and attributed the Vet-to-Vet program to having one of the best years of their life. Even those who did not attend many meetings spoke of it positively and stated that it should continue so others could try (Table).
From January 2022 to September 2025, > 80 veterans attended ≥ 1 meeting at RMRVAMC; 29 attended ≥ 1 meeting in the last quarter. There were > 1400 Vet-to-Vet encounters at RMRVAMC, with a mean (SD) of 14.2 (19.2) and a median of 4.5 encounters per participant. Half of the veterans attend ≥ 5 meetings, and one-third attended ≥ 10 meetings.
Since June 2023, 15 additional VHA facilities launched Vet-to-Vet programs. As of October 2025, > 350 veterans have participated in ≥ 1 Vet-to-Vet meeting, totaling > 4500 Vet-to-Vet encounters since the program’s inception (Figure 2).

Challenges
The RMRVAMC site and cosite leads are part of the national implementation team and dedicate substantial time to developing the program: 40 and 10 hours per week, respectively. Site leads at new locations do not receive funding for Vet-to-Vet activities and are recommended to dedicate only 4 hours per week to the program. Formally embedding Vet-to-Vet into the site leads’ roles is critical for sustainment.
The Vet-to-Vet model has changed. The initial Vet-to-Vet cohort included the 6-week Taking Charge of My Life and Health curriculum prior to moving to the mutual help format.24 While this curriculum still informs peer facilitator training, it is not used in new groups. It has anecdotally been reported that this change was positive, but the impact of this adaptation is unknown.
This evaluation cohort was small (16 participants) and initial patient reported and administrative outcomes were inconclusive. However, most veterans who stopped participating in Vet-to-Vet spoke fondly of their experiences with the program.
CONCLUSIONS
Vet-to-Vet is a promising new initiative to support self-management and social connection in chronic pain care. The program employs a mutual help approach and storytelling to empower veterans living with chronic pain. The effectiveness of these strategies will be evaluated, which will inform its continued growth. The program's current goals focus on sustainment at existing sites and expansion to new sites to reach more rural veterans across the VA enterprise. While Vet-to-Vet is designed to serve those who experience chronic pain, a partnership with the Office of Whole Health has established goals to begin expanding this model to other chronic conditions in 2026.
The Veterans Health Administration (VHA) has continued to advance its understanding and treatment of chronic pain. The VHA National Pain Management Strategy emphasizes the significance of the social context of pain while underscoring the importance of self-management.1 This established strategy ensures that all veterans have access to the appropriate pain care in the proper setting.2 VHA has instituted a stepped care model of pain management, delineating the domains of primary care, secondary consultative services, and tertiary care.3 This directive emphasized a biopsychosocial approach to pain management to prioritize the relationship between biological, psychological, and social factors that influence how veterans experience pain and should commensurately influence how it is managed.
The VHA Office of Patient-Centered Care and Cultural Transformation implemented the Whole Health System of Care as part of the Comprehensive Addiction and Recovery Act, which included a VHA directive to expand pain management.4,5 Reorientation within this system shifts from defining veterans as passive care recipients to viewing them as active partners in their own care and health. This partnership places additional emphasis on peer-led explorations of mission, aspiration, and purpose.6
Peer-led groups, also known as mutual aid, mutual support, and mutual help groups, have historically been successful for patients undergoing treatment for substance use disorders (eg, Alcoholics Anonymous).7 Mutual help groups have 3 defining characteristics. First, they are run by participants, not professionals, though the latter may have been integral in the founding of the groups. Second, participants share a similar problem (eg, disease state, experience, disposition). Finally, there is a reciprocal exchange of information and psychological support among participants.8,9 Mutual help groups that address chronic pain are rare but becoming more common.10-12 Emerging evidence suggests a positive relationship between peer support and improved well-being, self-efficacy, pain management, and pain self-management skills (eg, activity pacing).13-15
Storytelling as a tool for healing has a long history in indigenous and Western medical traditions.16-19 This includes the treatment of chronic disease, including pain.20,21 The use of storytelling in health care overlaps with the role it plays within many mutual help groups focused on chronic disease treatment.22 Storytelling allows an individual to share their experience with a disease, and take a more active role in their health, and facilitate stronger bonds with others.22 In effect, storytelling is not only important to group cohesion—it also plays a role in an individual’s healing.
Vet-to-Vet
The VHA Office of Rural Health funds Vet-to-Vet, a peer-to-peer program to address limited access to care for rural veterans with chronic pain. Similar to the VHA National Pain Management Strategy, Vet-to-Vet is grounded in the significance of the social context of pain and underscores the importance of self-management.1 The program combines pain care, mutual help, and storytelling to support veterans living with chronic pain. While the primary focus of Vet-to-Vet is rural veterans, the program serves any veteran experiencing chronic pain who is isolated from services, including home-bound urban veterans.
Following mutual help principles, Vet-to-Vet peer facilitators lead weekly online drop-in meetings. Meetings follow the general structure of reiterating group ground rules and sharing an individual pain story, followed by open discussions centered on well-being, chronic pain management, or any topic the group wishes to discuss. Meetings typically end with a mindfulness exercise. The organizational structure that supports Vet-to-Vet includes the implementation support team, site leads, Vet-to-Vet peer facilitators, and national partners (Figure 1).

Implementation Support Team
The implementation support team consists of a principal investigator, coinvestigator, program manager, and program support specialist. The team provides facilitator training, monthly community practice sessions for Vet-to-Vet peer facilitators and site leads, and weekly office hours for site leads. The implementation support team also recruits new Vet-to-Vet sites; potential new locations ideally have an existing whole health program, leadership support, committed site and cosite leads, and ≥ 3 peer facilitator volunteers.
Site Leads
Most site and cosite leads are based in whole health or pain management teams and are whole health coaches or peer support specialists. The site lead is responsible for standing up the program and documenting encounters, recruiting and supporting peer facilitators and participants, and overseeing the meeting. During meetings, site leads generally leave their cameras off and only speak when called into the group; the peer facilitators lead the meetings. The implementation support team recommends that site leads dedicate ≥ 4 hours per week to Vet-to-Vet; 2 hours for weekly group meetings and 2 hours for documentation (ie, entering notes into the participants’ electronic health records) and supporting peer facilitators and participants. Cosite lead responsibilities vary by location, with some sites having 2 leads that equally share duties and others having a primary lead and a colead available if the site lead is unable to attend a meeting.
Vet-to-Vet Peer Facilitators
Peer facilitators are the core of the program. They lead meetings from start to finish. Like participants, they also experience chronic pain and are volunteers. The implementation support team encourages sites to establish volunteer peer facilitators, rather than assigning peer support specialists to facilitate meetings. Veterans are eager to connect and give back to their communities, and the Vet-to-Vet peer facilitator role is an opportunity for those unable to work to connect with peers and add meaning to their lives. Even if a VHA employee is a veteran who has chronic pain, they are not eligible to serve as this could create a service provider/service recipient dynamic that is not in the spirit of mutual help.
Vet-to-Vet peer facilitators attend a virtual 3-day training held by the implementation support team prior to starting. These training sessions are available on a quarterly basis and facilitated by the Vet-to-Vet program manager and 2 current peer facilitators. Training content includes established whole health facilitator training materials and program-specific storytelling training materials. Once trained, peer facilitators attend storytelling practice sessions and collaborate with their site leads during weekly meetings.
Participants
Vet-to-Vet participants find the program through direct outreach from site leads, word of mouth, and referrals. The only criteria to join are that the individual is a veteran who experiences chronic pain and is enrolled in the VHA (site leads can assist with enrollment if needed). Participants are not required to have a diagnosis or engage in any other health care. There is no commitment and no end date. Some participants only come once; others have attended for > 3 years. This approach is intended to embrace the idea that the need for support ebbs and flows.
National Partners
The VHA Office of Rural Health provides technical support. The Center for Development and Civic Engagement onboards peer facilitators as VHA volunteers. The Office of Patient-Centered Care and Cultural Transformation provides national guidance and site-level collaboration. The VHA Pain Management, Opioid Safety, and Prescription Drug Monitoring Program supports site recruitment. In addition to the VHA partners, 4 veteran evaluation consultants who have experience with chronic pain but do not participate in Vet-to-Vet meetings provide advice on evaluation activities, such as question development and communication strategies.
Evaluation
This evaluation shares preliminary results from a pilot evaluation of the Rocky Mountain Regional VA Medical Center (RMRVAMC) Vet-to-Vet group. It is intended for program improvement, was deemed nonresearch by the Colorado Multiple Institutional Review Board, and was structured using the RE-AIM (Reach, Effectiveness, Adoption, Implementation, and Maintenance) framework.23 This evaluation focused on capturing measures related to reach and effectiveness, while a forthcoming evaluation includes elements of adoption, implementation, and maintenance.
In 2022, 16 Vet-to-Vet peer facilitators and participants completed surveys and interviews to share their experience. Interviews were recorded, transcribed, and coded in ATLAS.ti. A priori codes were based on interview guide questions and emergent descriptive codes were used to identify specific topics which were categorized into RE-AIM domains, barriers, facilitators, what participants learned, how participants applied what they learned to their lives, and participant reported outcomes. This article contains high-level findings from the evaluation; more detailed results will be included in the ongoing evaluation.
Results
The RMRVAMC Vet-to-Vet group has met weekly since April 2022. Four Vet-to-Vet peer facilitators and 12 individuals participated in the pilot Vet-to-Vet group and evaluation. The mean age was 62 years, most were men, and half were married. Most participants lived in rural areas with a mean distance of 125 miles to the nearest VAMC. Many experienced multiple kinds of pain, with a mean 4.5 on a 10-point scale (bothered “a lot”). All participants reported that they experienced pain daily.
Participation in Vet-to-Vet meetings was high; 3 of 4 peer facilitators and 7 of 12 participants completed the first 6 months of the program. In interviews, participants described the positive impact of the program. They emphasized the importance of connecting with other veterans and helping one another, with one noting that opportunities to connect with other veterans “just drops off a lot” (peer facilitator 3) after leaving active duty.
Some participants and Vet-to-Vet peer facilitators outlined the content of the sessions (eg, learning about how pain impacts the body and one’s family relationships) and shared the skills they learned (eg, goal setting, self-advocacy) (Table). Most spoke about learning from one another and the power of sharing stories with one peer facilitator sharing how they felt that witnessing another participant’s story “really shifted how I was thinking about things and how I perceived people” (peer facilitator 1).

Participants reported several ways the program impacted their lives, such as learning that they could get help, how to get help, and how to overcome the mental aspects of chronic pain. One veteran shared profound health impacts and attributed the Vet-to-Vet program to having one of the best years of their life. Even those who did not attend many meetings spoke of it positively and stated that it should continue so others could try (Table).
From January 2022 to September 2025, > 80 veterans attended ≥ 1 meeting at RMRVAMC; 29 attended ≥ 1 meeting in the last quarter. There were > 1400 Vet-to-Vet encounters at RMRVAMC, with a mean (SD) of 14.2 (19.2) and a median of 4.5 encounters per participant. Half of the veterans attend ≥ 5 meetings, and one-third attended ≥ 10 meetings.
Since June 2023, 15 additional VHA facilities launched Vet-to-Vet programs. As of October 2025, > 350 veterans have participated in ≥ 1 Vet-to-Vet meeting, totaling > 4500 Vet-to-Vet encounters since the program’s inception (Figure 2).

Challenges
The RMRVAMC site and cosite leads are part of the national implementation team and dedicate substantial time to developing the program: 40 and 10 hours per week, respectively. Site leads at new locations do not receive funding for Vet-to-Vet activities and are recommended to dedicate only 4 hours per week to the program. Formally embedding Vet-to-Vet into the site leads’ roles is critical for sustainment.
The Vet-to-Vet model has changed. The initial Vet-to-Vet cohort included the 6-week Taking Charge of My Life and Health curriculum prior to moving to the mutual help format.24 While this curriculum still informs peer facilitator training, it is not used in new groups. It has anecdotally been reported that this change was positive, but the impact of this adaptation is unknown.
This evaluation cohort was small (16 participants) and initial patient reported and administrative outcomes were inconclusive. However, most veterans who stopped participating in Vet-to-Vet spoke fondly of their experiences with the program.
CONCLUSIONS
Vet-to-Vet is a promising new initiative to support self-management and social connection in chronic pain care. The program employs a mutual help approach and storytelling to empower veterans living with chronic pain. The effectiveness of these strategies will be evaluated, which will inform its continued growth. The program's current goals focus on sustainment at existing sites and expansion to new sites to reach more rural veterans across the VA enterprise. While Vet-to-Vet is designed to serve those who experience chronic pain, a partnership with the Office of Whole Health has established goals to begin expanding this model to other chronic conditions in 2026.
- Kerns RD, Philip EJ, Lee AW, Rosenberger PH. Implementation of the Veterans Health Administration national pain management strategy. Transl Behav Med. 2011;1:635-643. doi:10.1007/s13142-011-0094-3
- Pain Management, Opioid Safety, and PDMP (PMOP). US Department of Veterans Affairs. Updated August 21, 2025. Accessed September 25, 2025. https://www.va.gov/PAINMANAGEMENT/Providers/IntegratedTeambasedPainCare.asp
- US Department of Veterans Affairs. VHA Directive 2009-053. October 28, 2009. Accessed September 25, 2025. https://www.va.gov/PAINMANAGEMENT/docs/VHA09PainDirective.pdf
- Comprehensive Addiction and Recovery Act of 2016, S524, 114th Cong (2015-2016). Pub L No. 114-198. July 22, 2016. Accessed September 25, 2025. https://www.congress.gov/bill/114th-congress/senate-bill/524
- Bokhour B, Hyde J, Zeliadt, Mohr D. Whole Health System of Care Evaluation. US Department of Veterans Affairs. February 18, 2020. Accessed September 25, 2025. https://www.va.gov/WHOLEHEALTH/docs/EPCC_WHSevaluation_FinalReport_508.pdf
- Gaudet T, Kligler B. Whole health in the whole system of the veterans administration: how will we know we have reached this future state? J Altern Complement Med. 2019;25:S7-S11. doi:10.1089/acm.2018.29061.gau
- Kelly JF, Yeterian JD. The role of mutual-help groups in extending the framework of treatment. Alcohol Res Health. 2011;33:350-355.
- Humphreys K. Self-help/mutual aid organizations: the view from Mars. Subst Use Misuse. 1997;32:2105-2109. doi:10.3109/10826089709035622
- Chinman M, Kloos B, O’Connell M, Davidson L. Service providers’ views of psychiatric mutual support groups. J Community Psychol. 2002;30:349-366. doi:10.1002/jcop.10010
- Shue SA, McGuire AB, Matthias MS. Facilitators and barriers to implementation of a peer support intervention for patients with chronic pain: a qualitative study. Pain Med. 2019;20:1311-1320. doi:10.1093/pm/pny229
- Pester BD, Tankha H, Caño A, et al. Facing pain together: a randomized controlled trial of the effects of Facebook support groups on adults with chronic pain. J Pain. 2022;23:2121-2134. doi:10.1016/j.jpain.2022.07.013
- Matthias MS, McGuire AB, Kukla M, Daggy J, Myers LJ, Bair MJ. A brief peer support intervention for veterans with chronic musculoskeletal pain: a pilot study of feasibility and effectiveness. Pain Med. 2015;16:81-87. doi:10.1111/pme.12571
- Finlay KA, Elander J. Reflecting the transition from pain management services to chronic pain support group attendance: an interpretative phenomenological analysis. Br J Health Psychol. 2016;21:660-676. doi:10.1111/bjhp.12194
- Finlay KA, Peacock S, Elander J. Developing successful social support: an interpretative phenomenological analysis of mechanisms and processes in a chronic pain support group. Psychol Health. 2018;33:846-871. doi:10.1080/08870446.2017.1421188
- Farr M, Brant H, Patel R, et al. Experiences of patient-led chronic pain peer support groups after pain management programs: a qualitative study. Pain Med. 2021;22:2884-2895. doi:10.1093/pm/pnab189
- Mehl-Madrona L. Narrative Medicine: The Use of History and Story in the Healing Process. Bear & Company; 2007.
- Fioretti C, Mazzocco K, Riva S, Oliveri S, Masiero M, Pravettoni G. Research studies on patients’ illness experience using the Narrative Medicine approach: a systematic review. BMJ Open. 2016;6:e011220. doi:10.1136/bmjopen-2016-011220
- Hall JM, Powell J. Understanding the person through narrative. Nurs Res Pract. 2011;2011:293837. doi:10.1155/2011/293837
- Ricks L, Kitchens S, Goodrich T, Hancock E. My story: the use of narrative therapy in individual and group counseling. J Creat Ment Health. 2014;9:99-110. doi:10.1080/15401383.2013.870947
- Hydén L-C. Illness and narrative. Sociol Health Illn. 1997;19:48-69. doi:10.1111/j.1467-9566.1997.tb00015.x
- Georgiadis E, Johnson MI. Incorporating personal narratives in positive psychology interventions to manage chronic pain. Front Pain Res (Lausanne). 2023;4:1253310. doi:10.3389/fpain.2023.1253310
- Gucciardi E, Jean-Pierre N, Karam G, Sidani S. Designing and delivering facilitated storytelling interventions for chronic disease self-management: a scoping review. BMC Health Serv Res. 2016;16:249. doi:10.1186/s12913-016-1474-7
- Glasgow RE, Vogt TM, Boles SM. Evaluating the public health impact of health promotion interventions: the RE-AIM framework. Am J Public Health. 1999;89:1322-1327. doi:10.2105/ajph.89.9.1322
- Abadi M, Richard B, Shamblen S, et al. Achieving whole health: a preliminary study of TCMLH, a group-based program promoting self-care and empowerment among veterans. Health Educ Behav. 2022;49:347-357. doi:10.1177/10901981211011043
- Kerns RD, Philip EJ, Lee AW, Rosenberger PH. Implementation of the Veterans Health Administration national pain management strategy. Transl Behav Med. 2011;1:635-643. doi:10.1007/s13142-011-0094-3
- Pain Management, Opioid Safety, and PDMP (PMOP). US Department of Veterans Affairs. Updated August 21, 2025. Accessed September 25, 2025. https://www.va.gov/PAINMANAGEMENT/Providers/IntegratedTeambasedPainCare.asp
- US Department of Veterans Affairs. VHA Directive 2009-053. October 28, 2009. Accessed September 25, 2025. https://www.va.gov/PAINMANAGEMENT/docs/VHA09PainDirective.pdf
- Comprehensive Addiction and Recovery Act of 2016, S524, 114th Cong (2015-2016). Pub L No. 114-198. July 22, 2016. Accessed September 25, 2025. https://www.congress.gov/bill/114th-congress/senate-bill/524
- Bokhour B, Hyde J, Zeliadt, Mohr D. Whole Health System of Care Evaluation. US Department of Veterans Affairs. February 18, 2020. Accessed September 25, 2025. https://www.va.gov/WHOLEHEALTH/docs/EPCC_WHSevaluation_FinalReport_508.pdf
- Gaudet T, Kligler B. Whole health in the whole system of the veterans administration: how will we know we have reached this future state? J Altern Complement Med. 2019;25:S7-S11. doi:10.1089/acm.2018.29061.gau
- Kelly JF, Yeterian JD. The role of mutual-help groups in extending the framework of treatment. Alcohol Res Health. 2011;33:350-355.
- Humphreys K. Self-help/mutual aid organizations: the view from Mars. Subst Use Misuse. 1997;32:2105-2109. doi:10.3109/10826089709035622
- Chinman M, Kloos B, O’Connell M, Davidson L. Service providers’ views of psychiatric mutual support groups. J Community Psychol. 2002;30:349-366. doi:10.1002/jcop.10010
- Shue SA, McGuire AB, Matthias MS. Facilitators and barriers to implementation of a peer support intervention for patients with chronic pain: a qualitative study. Pain Med. 2019;20:1311-1320. doi:10.1093/pm/pny229
- Pester BD, Tankha H, Caño A, et al. Facing pain together: a randomized controlled trial of the effects of Facebook support groups on adults with chronic pain. J Pain. 2022;23:2121-2134. doi:10.1016/j.jpain.2022.07.013
- Matthias MS, McGuire AB, Kukla M, Daggy J, Myers LJ, Bair MJ. A brief peer support intervention for veterans with chronic musculoskeletal pain: a pilot study of feasibility and effectiveness. Pain Med. 2015;16:81-87. doi:10.1111/pme.12571
- Finlay KA, Elander J. Reflecting the transition from pain management services to chronic pain support group attendance: an interpretative phenomenological analysis. Br J Health Psychol. 2016;21:660-676. doi:10.1111/bjhp.12194
- Finlay KA, Peacock S, Elander J. Developing successful social support: an interpretative phenomenological analysis of mechanisms and processes in a chronic pain support group. Psychol Health. 2018;33:846-871. doi:10.1080/08870446.2017.1421188
- Farr M, Brant H, Patel R, et al. Experiences of patient-led chronic pain peer support groups after pain management programs: a qualitative study. Pain Med. 2021;22:2884-2895. doi:10.1093/pm/pnab189
- Mehl-Madrona L. Narrative Medicine: The Use of History and Story in the Healing Process. Bear & Company; 2007.
- Fioretti C, Mazzocco K, Riva S, Oliveri S, Masiero M, Pravettoni G. Research studies on patients’ illness experience using the Narrative Medicine approach: a systematic review. BMJ Open. 2016;6:e011220. doi:10.1136/bmjopen-2016-011220
- Hall JM, Powell J. Understanding the person through narrative. Nurs Res Pract. 2011;2011:293837. doi:10.1155/2011/293837
- Ricks L, Kitchens S, Goodrich T, Hancock E. My story: the use of narrative therapy in individual and group counseling. J Creat Ment Health. 2014;9:99-110. doi:10.1080/15401383.2013.870947
- Hydén L-C. Illness and narrative. Sociol Health Illn. 1997;19:48-69. doi:10.1111/j.1467-9566.1997.tb00015.x
- Georgiadis E, Johnson MI. Incorporating personal narratives in positive psychology interventions to manage chronic pain. Front Pain Res (Lausanne). 2023;4:1253310. doi:10.3389/fpain.2023.1253310
- Gucciardi E, Jean-Pierre N, Karam G, Sidani S. Designing and delivering facilitated storytelling interventions for chronic disease self-management: a scoping review. BMC Health Serv Res. 2016;16:249. doi:10.1186/s12913-016-1474-7
- Glasgow RE, Vogt TM, Boles SM. Evaluating the public health impact of health promotion interventions: the RE-AIM framework. Am J Public Health. 1999;89:1322-1327. doi:10.2105/ajph.89.9.1322
- Abadi M, Richard B, Shamblen S, et al. Achieving whole health: a preliminary study of TCMLH, a group-based program promoting self-care and empowerment among veterans. Health Educ Behav. 2022;49:347-357. doi:10.1177/10901981211011043
A True Community: The Vet-to-Vet Program for Chronic Pain
A True Community: The Vet-to-Vet Program for Chronic Pain
Time to Reconsider Tramadol for Chronic Pain?
Time to Reconsider Tramadol for Chronic Pain?
Tramadol, a commonly prescribed opioid often viewed as a safer option for chronic pain, provided limited pain relief while increasing the risk for serious adverse effects, results of a new analysis showed.
In a systematic review and meta-analysis, investigators found that tramadol offered clinically insignificant pain relief, while doubling the likelihood of serious adverse events, most commonly cardiac complications.
"Given the limited analgesic benefits and increased risk of harm, tramadol use for chronic pain should be reconsidered," Jehad Ahmad Barakji, MD, of the Centre for Clinical Intervention Research at Rigshospitalet in Copenhagen, Denmark, told Medscape Medical News.
"Across different chronic pain conditions, tramadol's pain-relieving effect appears modest, and while some patients may experience relief, most will not gain substantial benefit," he added.
However, the researchers cautioned that the certainty of the evidence was low-to-moderate and that the quality of the underlying trials varied substantially.
The study was published on October 7 in BMJ Evidence-Based Medicine.
Popularity Outpacing Proof
Tramadol, a dual-action opioid that modulates serotonin and norepinephrine pathways, has long been promoted as a middle-ground analgesic - less addictive than morphine but stronger than nonopioid medications. It is approved for moderate-to-severe pain, including postoperative and chronic conditions.
Prescriptions have risen sharply worldwide, fueled by perceptions of safety and a belief that tramadol carries a lower risk for dependence. A recent global analysis estimated that nearly 18% of adults have used tramadol in their lifetime, and > 80% of those users combined it with at least 1 other substance.
Despite its widespread use, evidence supporting tramadol's long-term effectiveness and safety in chronic pain has been limited and inconsistent. Previous research has largely focused on short-term or condition-specific outcomes, such as osteoarthritis or neuropathic pain, leaving uncertainty about the drug's overall risk-benefit profile.
Small Benefit, Big Risk
The current study is the first comprehensive systematic review to evaluate tramadol alone for chronic pain using both meta-analysis and trail sequential analysis, provided a broader view of efficacy and safety across pain types.
"We sought to fill this gap by evaluating the totality of evidence to guide clinical practice," Barakji said.
The analysis included 19 randomized, placebo-controlled trials with 6569 adults. The average participant age was 56 years, and study durations ranged from 4 to 16 weeks. Pain intensity was typically assessed with the 0-10 numeric rating scale (NRS), while function and quality of life were measured with validated patient-reported tools.
Across the pooled analysis, participants receiving tramadol experienced an average pain reduction of 0.9 points on the NRS compared with placebo - a difference below the 1-point threshold considered clinically meaningful. About 7% more patients in the tramadol groups achieved noticeable pain relief, but investigators said the benefit was modest and uncertain.
Serious adverse events were twice as common among tramadol users, most often involving cardiac complications such as chest pain, coronary artery disease, or heart failure. Nonserious adverse effects, including nausea, dizziness, constipation, and drowsiness, were frequent and contributed to higher discontinuation rates among tramadol recipients.
The researchers acknowledged that most included trials were at a high risk for bias due to incomplete outcome reporting, small sample sizes, and inconsistent assessment methods, factors that may have exaggerated benefits and underestimated harms.
Reevaluating Tramadol's Role
Commenting on the research, Jessica Otte, MD, clinical associate professor in the Department of Family Medicine at The University of British Columbia, Vancouver, British Columbia, Canada, said the new review stands out for examining tramadol's use across a range of chronic pain conditions, an area where clinicians often struggle to help patients achieve meaningful and sustained relief.
Otte, who has studied the drug's safety and effectiveness through The University of British Columbia's Therapeutics Initiative, said the Danish team's analysis expands on earlier research that largely focused on single pain conditions. The findings, she said, add weight to growing evidence that challenges tramadol's perceived advantages over other analgesics.
"This review doesn't change the narrative but strengthens it: Tramdol's reputation as a safer or uniquely effective opioid is increasingly difficult to defend," she told Medscape Medical News.
While she believes the study makes an important contribution, Otte said the results should be interpreted with caution because of gaps in the underlying evidence. The Danish authors noted similar concerns, and Otte agreed that many of the included trials had methodological shortcomings.
"A lot of the studies had biases that made use less certain about what was reported," she said. "Many outcomes that matter to patients - like quality of life, functional improvement, or withdrawal - were either inconsistently measured or not reported at all."
She added that patients assigned to tramadol were more likely to discontinue early, both overall and because of adverse effects, raising concern about tolerability and attrition bias.
Also commenting, Houman Danesh, MD, professor and medical director of pain management at the Icahn School of Medicine at Mount Sinai in New York City, said the review provides moderate-certainty evidence that tramadol increases the risk for serious adverse events; but its broad approach - evaluating efficacy and safety across multiple chronic pain conditions - could be a confounding factor.
"Tramadol may benefit some conditions more than others," he noted, which could alter the overall risk-benefit profile.
In his clinical experience, Danesh said severe complications such as cardiac events are uncommon. He explained that heart rhythm disturbances occasionally associated with tramadol generally arise when patients are taking other medications that affect cardiac conduction.
Danesh emphasized that clinicians should weigh the study alongside other research and their own experience when deciding whether to prescribe the drug.
"It's important to take this study into consideration," he said, "but there are multiple studies that support the use of tramadol, and we have to look at the totality of the evidence."
Written by Carla Cantor.
The authors declared no competing interests and received no specific grant from any funding agency. Otte and Danesh reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Tramadol, a commonly prescribed opioid often viewed as a safer option for chronic pain, provided limited pain relief while increasing the risk for serious adverse effects, results of a new analysis showed.
In a systematic review and meta-analysis, investigators found that tramadol offered clinically insignificant pain relief, while doubling the likelihood of serious adverse events, most commonly cardiac complications.
"Given the limited analgesic benefits and increased risk of harm, tramadol use for chronic pain should be reconsidered," Jehad Ahmad Barakji, MD, of the Centre for Clinical Intervention Research at Rigshospitalet in Copenhagen, Denmark, told Medscape Medical News.
"Across different chronic pain conditions, tramadol's pain-relieving effect appears modest, and while some patients may experience relief, most will not gain substantial benefit," he added.
However, the researchers cautioned that the certainty of the evidence was low-to-moderate and that the quality of the underlying trials varied substantially.
The study was published on October 7 in BMJ Evidence-Based Medicine.
Popularity Outpacing Proof
Tramadol, a dual-action opioid that modulates serotonin and norepinephrine pathways, has long been promoted as a middle-ground analgesic - less addictive than morphine but stronger than nonopioid medications. It is approved for moderate-to-severe pain, including postoperative and chronic conditions.
Prescriptions have risen sharply worldwide, fueled by perceptions of safety and a belief that tramadol carries a lower risk for dependence. A recent global analysis estimated that nearly 18% of adults have used tramadol in their lifetime, and > 80% of those users combined it with at least 1 other substance.
Despite its widespread use, evidence supporting tramadol's long-term effectiveness and safety in chronic pain has been limited and inconsistent. Previous research has largely focused on short-term or condition-specific outcomes, such as osteoarthritis or neuropathic pain, leaving uncertainty about the drug's overall risk-benefit profile.
Small Benefit, Big Risk
The current study is the first comprehensive systematic review to evaluate tramadol alone for chronic pain using both meta-analysis and trail sequential analysis, provided a broader view of efficacy and safety across pain types.
"We sought to fill this gap by evaluating the totality of evidence to guide clinical practice," Barakji said.
The analysis included 19 randomized, placebo-controlled trials with 6569 adults. The average participant age was 56 years, and study durations ranged from 4 to 16 weeks. Pain intensity was typically assessed with the 0-10 numeric rating scale (NRS), while function and quality of life were measured with validated patient-reported tools.
Across the pooled analysis, participants receiving tramadol experienced an average pain reduction of 0.9 points on the NRS compared with placebo - a difference below the 1-point threshold considered clinically meaningful. About 7% more patients in the tramadol groups achieved noticeable pain relief, but investigators said the benefit was modest and uncertain.
Serious adverse events were twice as common among tramadol users, most often involving cardiac complications such as chest pain, coronary artery disease, or heart failure. Nonserious adverse effects, including nausea, dizziness, constipation, and drowsiness, were frequent and contributed to higher discontinuation rates among tramadol recipients.
The researchers acknowledged that most included trials were at a high risk for bias due to incomplete outcome reporting, small sample sizes, and inconsistent assessment methods, factors that may have exaggerated benefits and underestimated harms.
Reevaluating Tramadol's Role
Commenting on the research, Jessica Otte, MD, clinical associate professor in the Department of Family Medicine at The University of British Columbia, Vancouver, British Columbia, Canada, said the new review stands out for examining tramadol's use across a range of chronic pain conditions, an area where clinicians often struggle to help patients achieve meaningful and sustained relief.
Otte, who has studied the drug's safety and effectiveness through The University of British Columbia's Therapeutics Initiative, said the Danish team's analysis expands on earlier research that largely focused on single pain conditions. The findings, she said, add weight to growing evidence that challenges tramadol's perceived advantages over other analgesics.
"This review doesn't change the narrative but strengthens it: Tramdol's reputation as a safer or uniquely effective opioid is increasingly difficult to defend," she told Medscape Medical News.
While she believes the study makes an important contribution, Otte said the results should be interpreted with caution because of gaps in the underlying evidence. The Danish authors noted similar concerns, and Otte agreed that many of the included trials had methodological shortcomings.
"A lot of the studies had biases that made use less certain about what was reported," she said. "Many outcomes that matter to patients - like quality of life, functional improvement, or withdrawal - were either inconsistently measured or not reported at all."
She added that patients assigned to tramadol were more likely to discontinue early, both overall and because of adverse effects, raising concern about tolerability and attrition bias.
Also commenting, Houman Danesh, MD, professor and medical director of pain management at the Icahn School of Medicine at Mount Sinai in New York City, said the review provides moderate-certainty evidence that tramadol increases the risk for serious adverse events; but its broad approach - evaluating efficacy and safety across multiple chronic pain conditions - could be a confounding factor.
"Tramadol may benefit some conditions more than others," he noted, which could alter the overall risk-benefit profile.
In his clinical experience, Danesh said severe complications such as cardiac events are uncommon. He explained that heart rhythm disturbances occasionally associated with tramadol generally arise when patients are taking other medications that affect cardiac conduction.
Danesh emphasized that clinicians should weigh the study alongside other research and their own experience when deciding whether to prescribe the drug.
"It's important to take this study into consideration," he said, "but there are multiple studies that support the use of tramadol, and we have to look at the totality of the evidence."
Written by Carla Cantor.
The authors declared no competing interests and received no specific grant from any funding agency. Otte and Danesh reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Tramadol, a commonly prescribed opioid often viewed as a safer option for chronic pain, provided limited pain relief while increasing the risk for serious adverse effects, results of a new analysis showed.
In a systematic review and meta-analysis, investigators found that tramadol offered clinically insignificant pain relief, while doubling the likelihood of serious adverse events, most commonly cardiac complications.
"Given the limited analgesic benefits and increased risk of harm, tramadol use for chronic pain should be reconsidered," Jehad Ahmad Barakji, MD, of the Centre for Clinical Intervention Research at Rigshospitalet in Copenhagen, Denmark, told Medscape Medical News.
"Across different chronic pain conditions, tramadol's pain-relieving effect appears modest, and while some patients may experience relief, most will not gain substantial benefit," he added.
However, the researchers cautioned that the certainty of the evidence was low-to-moderate and that the quality of the underlying trials varied substantially.
The study was published on October 7 in BMJ Evidence-Based Medicine.
Popularity Outpacing Proof
Tramadol, a dual-action opioid that modulates serotonin and norepinephrine pathways, has long been promoted as a middle-ground analgesic - less addictive than morphine but stronger than nonopioid medications. It is approved for moderate-to-severe pain, including postoperative and chronic conditions.
Prescriptions have risen sharply worldwide, fueled by perceptions of safety and a belief that tramadol carries a lower risk for dependence. A recent global analysis estimated that nearly 18% of adults have used tramadol in their lifetime, and > 80% of those users combined it with at least 1 other substance.
Despite its widespread use, evidence supporting tramadol's long-term effectiveness and safety in chronic pain has been limited and inconsistent. Previous research has largely focused on short-term or condition-specific outcomes, such as osteoarthritis or neuropathic pain, leaving uncertainty about the drug's overall risk-benefit profile.
Small Benefit, Big Risk
The current study is the first comprehensive systematic review to evaluate tramadol alone for chronic pain using both meta-analysis and trail sequential analysis, provided a broader view of efficacy and safety across pain types.
"We sought to fill this gap by evaluating the totality of evidence to guide clinical practice," Barakji said.
The analysis included 19 randomized, placebo-controlled trials with 6569 adults. The average participant age was 56 years, and study durations ranged from 4 to 16 weeks. Pain intensity was typically assessed with the 0-10 numeric rating scale (NRS), while function and quality of life were measured with validated patient-reported tools.
Across the pooled analysis, participants receiving tramadol experienced an average pain reduction of 0.9 points on the NRS compared with placebo - a difference below the 1-point threshold considered clinically meaningful. About 7% more patients in the tramadol groups achieved noticeable pain relief, but investigators said the benefit was modest and uncertain.
Serious adverse events were twice as common among tramadol users, most often involving cardiac complications such as chest pain, coronary artery disease, or heart failure. Nonserious adverse effects, including nausea, dizziness, constipation, and drowsiness, were frequent and contributed to higher discontinuation rates among tramadol recipients.
The researchers acknowledged that most included trials were at a high risk for bias due to incomplete outcome reporting, small sample sizes, and inconsistent assessment methods, factors that may have exaggerated benefits and underestimated harms.
Reevaluating Tramadol's Role
Commenting on the research, Jessica Otte, MD, clinical associate professor in the Department of Family Medicine at The University of British Columbia, Vancouver, British Columbia, Canada, said the new review stands out for examining tramadol's use across a range of chronic pain conditions, an area where clinicians often struggle to help patients achieve meaningful and sustained relief.
Otte, who has studied the drug's safety and effectiveness through The University of British Columbia's Therapeutics Initiative, said the Danish team's analysis expands on earlier research that largely focused on single pain conditions. The findings, she said, add weight to growing evidence that challenges tramadol's perceived advantages over other analgesics.
"This review doesn't change the narrative but strengthens it: Tramdol's reputation as a safer or uniquely effective opioid is increasingly difficult to defend," she told Medscape Medical News.
While she believes the study makes an important contribution, Otte said the results should be interpreted with caution because of gaps in the underlying evidence. The Danish authors noted similar concerns, and Otte agreed that many of the included trials had methodological shortcomings.
"A lot of the studies had biases that made use less certain about what was reported," she said. "Many outcomes that matter to patients - like quality of life, functional improvement, or withdrawal - were either inconsistently measured or not reported at all."
She added that patients assigned to tramadol were more likely to discontinue early, both overall and because of adverse effects, raising concern about tolerability and attrition bias.
Also commenting, Houman Danesh, MD, professor and medical director of pain management at the Icahn School of Medicine at Mount Sinai in New York City, said the review provides moderate-certainty evidence that tramadol increases the risk for serious adverse events; but its broad approach - evaluating efficacy and safety across multiple chronic pain conditions - could be a confounding factor.
"Tramadol may benefit some conditions more than others," he noted, which could alter the overall risk-benefit profile.
In his clinical experience, Danesh said severe complications such as cardiac events are uncommon. He explained that heart rhythm disturbances occasionally associated with tramadol generally arise when patients are taking other medications that affect cardiac conduction.
Danesh emphasized that clinicians should weigh the study alongside other research and their own experience when deciding whether to prescribe the drug.
"It's important to take this study into consideration," he said, "but there are multiple studies that support the use of tramadol, and we have to look at the totality of the evidence."
Written by Carla Cantor.
The authors declared no competing interests and received no specific grant from any funding agency. Otte and Danesh reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Time to Reconsider Tramadol for Chronic Pain?
Time to Reconsider Tramadol for Chronic Pain?
Data Trends 2025: Acute Pain
Data Trends 2025: Acute Pain
Click to view more from Federal Health Care Data Trends 2025.
- Baumann L, et al. Curr Pain Headache Rep. 2023;27(9):437-444. doi:10.1007/s11916-023-01127-0
- Reif S, et al. Mil Med. 2018;183(9-10):e330-e337. doi:10.1093/milmed/usx200
- Sharp LK, e t a l . Pain. 2023;164( 4 ) : 749-757. doi:10.1097/j .pain.0000000000002759
- Dalton MK, et al. J Trauma Acute Care Surg. 2021;91(2S Suppl 2):S213-S220. doi:10.1097/TA.0000000000003133
- Mahyar L, et al. Reg Anesth Pain Med. 2024;49(2):117-121. doi:10.1136/rapm-2023-104610
- Gupta K, et al. Eur J Trauma Emerg Surg. 2025;51(1):103. doi:10.1007/s00068-025-02778-x
- Mariano ER, et al. Reg Anesth Pain Med. 2022;47(2):118-127. doi:10.1136/rapm-2021-103083
Click to view more from Federal Health Care Data Trends 2025.
Click to view more from Federal Health Care Data Trends 2025.
- Baumann L, et al. Curr Pain Headache Rep. 2023;27(9):437-444. doi:10.1007/s11916-023-01127-0
- Reif S, et al. Mil Med. 2018;183(9-10):e330-e337. doi:10.1093/milmed/usx200
- Sharp LK, e t a l . Pain. 2023;164( 4 ) : 749-757. doi:10.1097/j .pain.0000000000002759
- Dalton MK, et al. J Trauma Acute Care Surg. 2021;91(2S Suppl 2):S213-S220. doi:10.1097/TA.0000000000003133
- Mahyar L, et al. Reg Anesth Pain Med. 2024;49(2):117-121. doi:10.1136/rapm-2023-104610
- Gupta K, et al. Eur J Trauma Emerg Surg. 2025;51(1):103. doi:10.1007/s00068-025-02778-x
- Mariano ER, et al. Reg Anesth Pain Med. 2022;47(2):118-127. doi:10.1136/rapm-2021-103083
- Baumann L, et al. Curr Pain Headache Rep. 2023;27(9):437-444. doi:10.1007/s11916-023-01127-0
- Reif S, et al. Mil Med. 2018;183(9-10):e330-e337. doi:10.1093/milmed/usx200
- Sharp LK, e t a l . Pain. 2023;164( 4 ) : 749-757. doi:10.1097/j .pain.0000000000002759
- Dalton MK, et al. J Trauma Acute Care Surg. 2021;91(2S Suppl 2):S213-S220. doi:10.1097/TA.0000000000003133
- Mahyar L, et al. Reg Anesth Pain Med. 2024;49(2):117-121. doi:10.1136/rapm-2023-104610
- Gupta K, et al. Eur J Trauma Emerg Surg. 2025;51(1):103. doi:10.1007/s00068-025-02778-x
- Mariano ER, et al. Reg Anesth Pain Med. 2022;47(2):118-127. doi:10.1136/rapm-2021-103083
Data Trends 2025: Acute Pain
Data Trends 2025: Acute Pain
Sports Injuries of the Hip in Primary Care
This transcript has been edited for clarity.
Matthew F. Watto, MD: Welcome back to The Curbsiders. I’m Dr Matthew Frank Watto, here with my great friend and America’s primary care physician, Dr Paul Nelson Williams. Paul, how are you feeling about sports injuries?
Paul N. Williams, MD: I’m feeling great, Matt.
Watto: You had a sports injury of the hip. Maybe that’s an overshare, Paul, but we talked about it on a podcast with Dr Carlin Senter (part 1 and part 2).
Williams: I think I’ve shared more than my hip injury, for sure.
Watto: Whenever a patient presented with hip pain, I used to pray it was trochanteric bursitis, which now I know is not really the right thing to think about. Intra-articular hip pain presents as anterior hip pain, usually in the crease of the hip. Depending on the patient’s age and history, the differential for that type of pain includes iliopsoas tendonitis, FAI syndrome, a labral tear, a bone stress injury of the femoral neck, or osteoarthritis.
So, what exactly is FAI and how might we diagnose it?
Williams: FAI is what the cool kids call femoral acetabular impingement, and it’s exactly what it sounds like.
Something is pinching or impinging upon the joint itself and preventing full range of motion. This is a ball-and-socket joint, so it should have tremendous range of motion, able to move in all planes. If it’s impinged, then pain will occur with certain movements. There’s a cam type, which is characterized by enlargement of the femoral head neck junction, or a pincer type, which has more to do with overhang of the acetabulum, and it can also be mixed. In any case, impingement upon the patient’s full range of motion results in pain.
You evaluate this with a couple of tests — the FABER and the FADIR.
The FABER is flexion, abduction, and external rotation, and the FADIR is flexion, adduction, and internal rotation. If you elicit anterior pain with either of those tests, it’s probably one of the intra-articular pathologies, although it is hard to know for sure which one it is because these tests are fairly sensitive but not very specific.
Watto: You can get x-rays to help with the diagnosis. You would order two views of the hip: an AP of the pelvis, which is just a straight-on shot to look for arthritis or fracture. Is there a healthy joint line there? The second is the Dunn view, in which the hip is flexed 90 degrees and abducted about 20 degrees. You are looking for fracture or impingement. You can diagnose FAI based on that view, and you might be able to diagnose a hip stress injury or osteoarthritis.
Unfortunately, you’re not going to see a labral tear, but Dr Senter said that both FAI and labral tears are treated the same way, with physical therapy. Patients with FAI who aren’t getting better might end up going for surgery, so at some point I would refer them to orthopedic surgery. But I feel much more comfortable now diagnosing these conditions with these tests.
Let’s talk a little bit about trochanteric pain syndrome. I used to think it was all bursitis. Why is that not correct?
Williams: It’s nice of you to feign ignorance for the purpose of education. It used to be thought of as bursitis, but these days we know it is probably more likely a tendinopathy.

Trochanteric pain syndrome was formerly known as trochanteric bursitis, but the bursa is not typically involved. Trochanteric pain syndrome is a tendinopathy of the surrounding structures: the gluteus medius, the iliotibial band, and the tensor fascia latae. The way these structures relate looks a bit like the face of a clock, as you can see on the infographic. In general, you manage this condition the same way you do with bursitis — physical therapy. You can also give corticosteroid injections. Physical therapy is probably more durable in terms of pain relief and functionality, but in the short term, corticosteroids might provide some degree of analgesia as well.
Watto: The last thing we wanted to mention is bone stress injury, which can occur in high-mileage runners (20 miles or more per week). Patients with bone stress injury need to rest, usually non‒weight bearing, for a period of time.

Treatment of a bone stress fracture depends on which side it’s on (top or bottom). If it’s on the top of the femoral neck (the tension side), it has to be fixed. If it’s on the compression side (the bottom side of the femoral neck), it might be able to be managed conservatively, but many patients are going to need surgery. This is a big deal. But it’s a spectrum; in some cases the bone is merely irritated and unhappy, without a break in the cortex. Those patients might not need surgery.
In patients with a fracture of the femoral neck — especially younger, healthier patients — you should think about getting a bone density test and screening for relative energy deficiency in sport. This used to be called the female athlete triad, which includes disrupted menstrual cycles, being underweight, and fracture. We should be screening patients, asking them in a nonjudgmental way about their relationship with food, to make sure they are getting an appropriate number of calories.
They are actually in an energy deficit. They’re not eating enough to maintain a healthy body with so much activity.
Williams: If you’re interested in this topic, you should refer to the full podcast with Dr Senter which is chock-full of helpful information.
Dr Watto, Clinical Assistant Professor, Department of Medicine, Perelman School of Medicine at University of Pennsylvania; Internist, Department of Medicine, Hospital Medicine Section, Pennsylvania Hospital, Philadelphia, Pennsylvania, has disclosed no relevant financial relationships. Dr Williams, Associate Professor of Clinical Medicine, Department of General Internal Medicine, Lewis Katz School of Medicine; Staff Physician, Department of General Internal Medicine, Temple Internal Medicine Associates, Philadelphia, Pennsylvania, disclosed ties with The Curbsiders.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Matthew F. Watto, MD: Welcome back to The Curbsiders. I’m Dr Matthew Frank Watto, here with my great friend and America’s primary care physician, Dr Paul Nelson Williams. Paul, how are you feeling about sports injuries?
Paul N. Williams, MD: I’m feeling great, Matt.
Watto: You had a sports injury of the hip. Maybe that’s an overshare, Paul, but we talked about it on a podcast with Dr Carlin Senter (part 1 and part 2).
Williams: I think I’ve shared more than my hip injury, for sure.
Watto: Whenever a patient presented with hip pain, I used to pray it was trochanteric bursitis, which now I know is not really the right thing to think about. Intra-articular hip pain presents as anterior hip pain, usually in the crease of the hip. Depending on the patient’s age and history, the differential for that type of pain includes iliopsoas tendonitis, FAI syndrome, a labral tear, a bone stress injury of the femoral neck, or osteoarthritis.
So, what exactly is FAI and how might we diagnose it?
Williams: FAI is what the cool kids call femoral acetabular impingement, and it’s exactly what it sounds like.

Something is pinching or impinging upon the joint itself and preventing full range of motion. This is a ball-and-socket joint, so it should have tremendous range of motion, able to move in all planes. If it’s impinged, then pain will occur with certain movements. There’s a cam type, which is characterized by enlargement of the femoral head neck junction, or a pincer type, which has more to do with overhang of the acetabulum, and it can also be mixed. In any case, impingement upon the patient’s full range of motion results in pain.
You evaluate this with a couple of tests — the FABER and the FADIR.
The FABER is flexion, abduction, and external rotation, and the FADIR is flexion, adduction, and internal rotation. If you elicit anterior pain with either of those tests, it’s probably one of the intra-articular pathologies, although it is hard to know for sure which one it is because these tests are fairly sensitive but not very specific.
Watto: You can get x-rays to help with the diagnosis. You would order two views of the hip: an AP of the pelvis, which is just a straight-on shot to look for arthritis or fracture. Is there a healthy joint line there? The second is the Dunn view, in which the hip is flexed 90 degrees and abducted about 20 degrees. You are looking for fracture or impingement. You can diagnose FAI based on that view, and you might be able to diagnose a hip stress injury or osteoarthritis.
Unfortunately, you’re not going to see a labral tear, but Dr Senter said that both FAI and labral tears are treated the same way, with physical therapy. Patients with FAI who aren’t getting better might end up going for surgery, so at some point I would refer them to orthopedic surgery. But I feel much more comfortable now diagnosing these conditions with these tests.
Let’s talk a little bit about trochanteric pain syndrome. I used to think it was all bursitis. Why is that not correct?
Williams: It’s nice of you to feign ignorance for the purpose of education. It used to be thought of as bursitis, but these days we know it is probably more likely a tendinopathy.

Trochanteric pain syndrome was formerly known as trochanteric bursitis, but the bursa is not typically involved. Trochanteric pain syndrome is a tendinopathy of the surrounding structures: the gluteus medius, the iliotibial band, and the tensor fascia latae. The way these structures relate looks a bit like the face of a clock, as you can see on the infographic. In general, you manage this condition the same way you do with bursitis — physical therapy. You can also give corticosteroid injections. Physical therapy is probably more durable in terms of pain relief and functionality, but in the short term, corticosteroids might provide some degree of analgesia as well.
Watto: The last thing we wanted to mention is bone stress injury, which can occur in high-mileage runners (20 miles or more per week). Patients with bone stress injury need to rest, usually non‒weight bearing, for a period of time.

Treatment of a bone stress fracture depends on which side it’s on (top or bottom). If it’s on the top of the femoral neck (the tension side), it has to be fixed. If it’s on the compression side (the bottom side of the femoral neck), it might be able to be managed conservatively, but many patients are going to need surgery. This is a big deal. But it’s a spectrum; in some cases the bone is merely irritated and unhappy, without a break in the cortex. Those patients might not need surgery.
In patients with a fracture of the femoral neck — especially younger, healthier patients — you should think about getting a bone density test and screening for relative energy deficiency in sport. This used to be called the female athlete triad, which includes disrupted menstrual cycles, being underweight, and fracture. We should be screening patients, asking them in a nonjudgmental way about their relationship with food, to make sure they are getting an appropriate number of calories.
They are actually in an energy deficit. They’re not eating enough to maintain a healthy body with so much activity.
Williams: If you’re interested in this topic, you should refer to the full podcast with Dr Senter which is chock-full of helpful information.
Dr Watto, Clinical Assistant Professor, Department of Medicine, Perelman School of Medicine at University of Pennsylvania; Internist, Department of Medicine, Hospital Medicine Section, Pennsylvania Hospital, Philadelphia, Pennsylvania, has disclosed no relevant financial relationships. Dr Williams, Associate Professor of Clinical Medicine, Department of General Internal Medicine, Lewis Katz School of Medicine; Staff Physician, Department of General Internal Medicine, Temple Internal Medicine Associates, Philadelphia, Pennsylvania, disclosed ties with The Curbsiders.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Matthew F. Watto, MD: Welcome back to The Curbsiders. I’m Dr Matthew Frank Watto, here with my great friend and America’s primary care physician, Dr Paul Nelson Williams. Paul, how are you feeling about sports injuries?
Paul N. Williams, MD: I’m feeling great, Matt.
Watto: You had a sports injury of the hip. Maybe that’s an overshare, Paul, but we talked about it on a podcast with Dr Carlin Senter (part 1 and part 2).
Williams: I think I’ve shared more than my hip injury, for sure.
Watto: Whenever a patient presented with hip pain, I used to pray it was trochanteric bursitis, which now I know is not really the right thing to think about. Intra-articular hip pain presents as anterior hip pain, usually in the crease of the hip. Depending on the patient’s age and history, the differential for that type of pain includes iliopsoas tendonitis, FAI syndrome, a labral tear, a bone stress injury of the femoral neck, or osteoarthritis.
So, what exactly is FAI and how might we diagnose it?
Williams: FAI is what the cool kids call femoral acetabular impingement, and it’s exactly what it sounds like.

Something is pinching or impinging upon the joint itself and preventing full range of motion. This is a ball-and-socket joint, so it should have tremendous range of motion, able to move in all planes. If it’s impinged, then pain will occur with certain movements. There’s a cam type, which is characterized by enlargement of the femoral head neck junction, or a pincer type, which has more to do with overhang of the acetabulum, and it can also be mixed. In any case, impingement upon the patient’s full range of motion results in pain.
You evaluate this with a couple of tests — the FABER and the FADIR.
The FABER is flexion, abduction, and external rotation, and the FADIR is flexion, adduction, and internal rotation. If you elicit anterior pain with either of those tests, it’s probably one of the intra-articular pathologies, although it is hard to know for sure which one it is because these tests are fairly sensitive but not very specific.
Watto: You can get x-rays to help with the diagnosis. You would order two views of the hip: an AP of the pelvis, which is just a straight-on shot to look for arthritis or fracture. Is there a healthy joint line there? The second is the Dunn view, in which the hip is flexed 90 degrees and abducted about 20 degrees. You are looking for fracture or impingement. You can diagnose FAI based on that view, and you might be able to diagnose a hip stress injury or osteoarthritis.
Unfortunately, you’re not going to see a labral tear, but Dr Senter said that both FAI and labral tears are treated the same way, with physical therapy. Patients with FAI who aren’t getting better might end up going for surgery, so at some point I would refer them to orthopedic surgery. But I feel much more comfortable now diagnosing these conditions with these tests.
Let’s talk a little bit about trochanteric pain syndrome. I used to think it was all bursitis. Why is that not correct?
Williams: It’s nice of you to feign ignorance for the purpose of education. It used to be thought of as bursitis, but these days we know it is probably more likely a tendinopathy.

Trochanteric pain syndrome was formerly known as trochanteric bursitis, but the bursa is not typically involved. Trochanteric pain syndrome is a tendinopathy of the surrounding structures: the gluteus medius, the iliotibial band, and the tensor fascia latae. The way these structures relate looks a bit like the face of a clock, as you can see on the infographic. In general, you manage this condition the same way you do with bursitis — physical therapy. You can also give corticosteroid injections. Physical therapy is probably more durable in terms of pain relief and functionality, but in the short term, corticosteroids might provide some degree of analgesia as well.
Watto: The last thing we wanted to mention is bone stress injury, which can occur in high-mileage runners (20 miles or more per week). Patients with bone stress injury need to rest, usually non‒weight bearing, for a period of time.

Treatment of a bone stress fracture depends on which side it’s on (top or bottom). If it’s on the top of the femoral neck (the tension side), it has to be fixed. If it’s on the compression side (the bottom side of the femoral neck), it might be able to be managed conservatively, but many patients are going to need surgery. This is a big deal. But it’s a spectrum; in some cases the bone is merely irritated and unhappy, without a break in the cortex. Those patients might not need surgery.
In patients with a fracture of the femoral neck — especially younger, healthier patients — you should think about getting a bone density test and screening for relative energy deficiency in sport. This used to be called the female athlete triad, which includes disrupted menstrual cycles, being underweight, and fracture. We should be screening patients, asking them in a nonjudgmental way about their relationship with food, to make sure they are getting an appropriate number of calories.
They are actually in an energy deficit. They’re not eating enough to maintain a healthy body with so much activity.
Williams: If you’re interested in this topic, you should refer to the full podcast with Dr Senter which is chock-full of helpful information.
Dr Watto, Clinical Assistant Professor, Department of Medicine, Perelman School of Medicine at University of Pennsylvania; Internist, Department of Medicine, Hospital Medicine Section, Pennsylvania Hospital, Philadelphia, Pennsylvania, has disclosed no relevant financial relationships. Dr Williams, Associate Professor of Clinical Medicine, Department of General Internal Medicine, Lewis Katz School of Medicine; Staff Physician, Department of General Internal Medicine, Temple Internal Medicine Associates, Philadelphia, Pennsylvania, disclosed ties with The Curbsiders.
A version of this article appeared on Medscape.com.
More Americans Than Ever Suffer From Chronic Pain
More Americans than ever are hurting with enduring, life-restricting pain. Like obesity, this condition is on the rise, according to figures in a new National Center for Health Statistics (NCHS) Data Brief from the Centers for Disease Control and Prevention (CDC).
Both types increased with age and with decreasing urbanization level. Women were more likely than men to have HICP (23.2% vs 7.3%).
Like obesity, chronic pain is multifactorial and is best managed with multidisciplinary intervention, said Jianguo Cheng, MD, PhD, a professor of anesthesiology and medical director of the Cleveland Clinic Consortium for Pain in Ohio. “It’s a complex mix of genetic, biological, and psychosocial dimensions that can cause ongoing pain out of proportion to the original limited injury that triggered it.”
While today’s longer lifespans are the primary driver of the increase, noted Martin Cheatle, PhD, an associate professor of psychiatry, anesthesiology, and critical care and director of behavioral medicine at the Penn Pain Medicine Center at the University of Pennsylvania’s Perelman School of Medicine, Philadelphia, another important factor is the more than 100 million Americans who suffer from obesity. “Obesity is a major risk factor for chronic pain conditions including advancing joint disease, low back pain, and diabetic neuropathies,” he said.
Age is an amplifier, agreed Beth Darnall, PhD, a professor of anesthesiology and perioperative and pain medicine and director of the Pain Relief Innovations Lab at Stanford University in Palo Alto, California, but the increases in chronic pain and HICP cut across age strata.
“Across the board we see striking increases in chronic pain, such as a 5% increase for those 65 and older, and a nearly 2% increase in HICP in that same age group,” Darnall said, referencing the changes from 2019 data in the new NCHS Data Brief. “And an almost 4% increase was observed for the youngest adult age category,18 to 29. Some of our research is now focusing on how to best treat chronic pain in young adults.”
The rise in chronic pain is broadly linked to the overall decline in the health of the US population, as indicated by the CDC 2024’s Chronic Disease Prevalence in the US: Sociodemographic and Geographic Variations by Zip Code Tabulation Area.
The Opioid Crisis and COVID
Beginning in 2016, in response to the opioid crisis and CDC guidelines, opioid prescribing for chronic pain rapidly dropped, both in terms of new prescriptions and tapering of doses of long-term users. “Reduced opioid prescribing yielded benefits for some patients but created new problems and harms for other patients,” said Darnall. Cheng added that the CDC’s recommendations on opioid prescribing were widely misinterpreted and were applied to patients with painful conditions such as cancer and sickle cell disease who were not intended to be affected by the guidelines. “In addition, although medical opioid prescribing dropped by 50%, overdose deaths from non-medical opioid sources increased by more 50%.”
Currently, most opioid overdoses are related to heroin, fentanyl, and newer drugs of abuse such as xylazine. “Most pain clinicians would agree that opioids are not first-line therapy for chronic non-cancer pain, but in a select number of well-vetted patients, opioids can be very effective in improving functionality and quality of life as part of a multimodal approach to pain care,” Cheatle said.
The impact of the opioid crisis is complex, said Cheatle, noting that only 8%-10% of pain patients on long-term opioid therapy develop a use disorder. “However, opioids were overly prescribed due to clinicians’ lack of training in core competencies of pain management and the insurance companies’ refusal to adequately cover non-opioid therapies such as acupuncture, cognitive behavioral therapy, extended physical therapy, and medical massage,” he said.
He pointed out that in the late 1990s there were more than 1000 multidisciplinary pain centers, whereas currently there are many fewer owing to lack of insurance reimbursement. “This results in more possibly avoidable invasive surgeries, which can further contribute to the increase in chronic pain.”
The COVID pandemic further exacerbated the pain problem and delayed access to timely medical interventions for many people. Some adopted a more sedentary lifestyle, already entrenched in today’s technology-driven society, leading in turn to weight gain and more chronic pain. “The isolation and lack of normal human connections during the pandemic could exacerbate pain and loss of autonomy,” Cheatle said. And some individuals developed painful neurologic conditions related to long-haul COVID, for which there is no effective treatment.
Best Approach
“Historically, pain has been treated as a purely biomedical issue. Bringing a biopsychosocial perspective to pain care can support pain relief,” said Darnall. Multiple national clinical guidance documents have called for a comprehensive approach that considers the whole person: their circumstances, their needs, their stressors, and their environment. “And we must provide patients meaningful access to the lowest-risk, non-pharmacologic treatments first – and ideally early on,” she said.
Even effective medications rarely make a person pain free, so other approaches are needed in tandem, Darnall said. Support for stronger patient competency in self-management of chronic pain is mounting.
“It’s vitally important that we help people know how to help themselves have less pain – how to steer their mind and body toward relief by using pain relief skills,” she said. “By so doing they can cultivate a critical level of control over their pain and are less at its mercy, which supports good mood and is shown to help people be more active as the impacts of pain diminish.”
Darnall outlined a recent development in the primary care setting that involves offering patients a brief program in pain-relief skills training. Within Veterans Affairs primary care, for example, patients receive several 30-minute sessions in pain reduction techniques. Outside of the VA, primary care clinics are incorporating an evidence-based, one-session 2-hour pain relief skills class called Empowered Relief, as standard care.
The class teaches participants three pain management skills and creates a personalized plan for each that includes a free app for ongoing daily use.
Since pain causes agitation in the central nervous system, manifested as fast heart rate, rapid breathing, muscle tension, and distress, people learn various ways to calm the central nervous system – with, for example, a sound technology known as binaural audio to deepen the relaxation response. “They also learn to identify and target worry about pain and develop self-soothing actions to interrupt unhelpful patterns,” Darnall said.
Data from randomized chronic pain studies, including one by her group using a virtual reality training program for lower back pain, show that 3 months after the training program people report clinically meaningful reductions in pain intensity, pain interference with daily activities, and sleep disturbance, as well as pain-related distress, anxiety, and fatigue
While psychological and complementary approaches have been effective in improving function and mood, there are barriers to accessing them, said Cheatle, such as lack of insurance coverage and the stigma associated with nontraditional, especially psychological, care.
Prevention
Good lifestyle behaviors promote better health as people age. “Maintaining a healthy weight, staying active, prioritizing good sleep, and avoiding smoking and alcohol use can support better health and buffer against chronic diseases and pain,” Darnall said.
Cheatle noted the importance of maintaining a safe work environment and avoiding injury risks by, for example, wearing a seatbelt or a cycling helmet.
The Future
“We need to ensure all individuals have access to effective, low-burden pain treatments, including evidence-based treatments they can receive from home so as to minimize treatment disparities.” Darnall said. Also needed is better comprehensive treatment for acute and chronic pain alike. “If we treat acute pain better, we will have fewer people transitioning to the chronic pain state.”
To that end, added Cheng, healthcare professionals in every specialty from doctors and nurses to psychologists and chiropractors need to develop co-competencies in pain management.
For Cheatle, the near future looks bleak. “There are some pioneering bioengineering approaches to reduce chronic pain and novel pharmacologic agents such as calcitonin gene-related peptide inhibitors for intractable migraines, but just changing insurance reimbursement for a comprehensive approach to chronic pain care and bolstering healthcare provider education on core pain competencies will benefit the over 50 million adults who suffer from chronic pain.”
Cheng, however, is more sanguine. “I don’t expect miracles in 10 years’ time, but we’re making rapid progress in understanding the genetics of chronic pain and the mechanisms of disease and therapy. We’re developing biomarkers to help in prognosis and monitor disease progress.” In the meantime, he pointed to an expanding array of non-pharmaceutical options, including neuromodulatory approaches such as nerve blocks and spinal cord stimulation.
Cheng, Cheatle, and Darnall disclosed no relevant competing interests.
A version of this article appeared on Medscape.com.
More Americans than ever are hurting with enduring, life-restricting pain. Like obesity, this condition is on the rise, according to figures in a new National Center for Health Statistics (NCHS) Data Brief from the Centers for Disease Control and Prevention (CDC).
Both types increased with age and with decreasing urbanization level. Women were more likely than men to have HICP (23.2% vs 7.3%).
Like obesity, chronic pain is multifactorial and is best managed with multidisciplinary intervention, said Jianguo Cheng, MD, PhD, a professor of anesthesiology and medical director of the Cleveland Clinic Consortium for Pain in Ohio. “It’s a complex mix of genetic, biological, and psychosocial dimensions that can cause ongoing pain out of proportion to the original limited injury that triggered it.”
While today’s longer lifespans are the primary driver of the increase, noted Martin Cheatle, PhD, an associate professor of psychiatry, anesthesiology, and critical care and director of behavioral medicine at the Penn Pain Medicine Center at the University of Pennsylvania’s Perelman School of Medicine, Philadelphia, another important factor is the more than 100 million Americans who suffer from obesity. “Obesity is a major risk factor for chronic pain conditions including advancing joint disease, low back pain, and diabetic neuropathies,” he said.
Age is an amplifier, agreed Beth Darnall, PhD, a professor of anesthesiology and perioperative and pain medicine and director of the Pain Relief Innovations Lab at Stanford University in Palo Alto, California, but the increases in chronic pain and HICP cut across age strata.
“Across the board we see striking increases in chronic pain, such as a 5% increase for those 65 and older, and a nearly 2% increase in HICP in that same age group,” Darnall said, referencing the changes from 2019 data in the new NCHS Data Brief. “And an almost 4% increase was observed for the youngest adult age category,18 to 29. Some of our research is now focusing on how to best treat chronic pain in young adults.”
The rise in chronic pain is broadly linked to the overall decline in the health of the US population, as indicated by the CDC 2024’s Chronic Disease Prevalence in the US: Sociodemographic and Geographic Variations by Zip Code Tabulation Area.
The Opioid Crisis and COVID
Beginning in 2016, in response to the opioid crisis and CDC guidelines, opioid prescribing for chronic pain rapidly dropped, both in terms of new prescriptions and tapering of doses of long-term users. “Reduced opioid prescribing yielded benefits for some patients but created new problems and harms for other patients,” said Darnall. Cheng added that the CDC’s recommendations on opioid prescribing were widely misinterpreted and were applied to patients with painful conditions such as cancer and sickle cell disease who were not intended to be affected by the guidelines. “In addition, although medical opioid prescribing dropped by 50%, overdose deaths from non-medical opioid sources increased by more 50%.”
Currently, most opioid overdoses are related to heroin, fentanyl, and newer drugs of abuse such as xylazine. “Most pain clinicians would agree that opioids are not first-line therapy for chronic non-cancer pain, but in a select number of well-vetted patients, opioids can be very effective in improving functionality and quality of life as part of a multimodal approach to pain care,” Cheatle said.
The impact of the opioid crisis is complex, said Cheatle, noting that only 8%-10% of pain patients on long-term opioid therapy develop a use disorder. “However, opioids were overly prescribed due to clinicians’ lack of training in core competencies of pain management and the insurance companies’ refusal to adequately cover non-opioid therapies such as acupuncture, cognitive behavioral therapy, extended physical therapy, and medical massage,” he said.
He pointed out that in the late 1990s there were more than 1000 multidisciplinary pain centers, whereas currently there are many fewer owing to lack of insurance reimbursement. “This results in more possibly avoidable invasive surgeries, which can further contribute to the increase in chronic pain.”
The COVID pandemic further exacerbated the pain problem and delayed access to timely medical interventions for many people. Some adopted a more sedentary lifestyle, already entrenched in today’s technology-driven society, leading in turn to weight gain and more chronic pain. “The isolation and lack of normal human connections during the pandemic could exacerbate pain and loss of autonomy,” Cheatle said. And some individuals developed painful neurologic conditions related to long-haul COVID, for which there is no effective treatment.
Best Approach
“Historically, pain has been treated as a purely biomedical issue. Bringing a biopsychosocial perspective to pain care can support pain relief,” said Darnall. Multiple national clinical guidance documents have called for a comprehensive approach that considers the whole person: their circumstances, their needs, their stressors, and their environment. “And we must provide patients meaningful access to the lowest-risk, non-pharmacologic treatments first – and ideally early on,” she said.
Even effective medications rarely make a person pain free, so other approaches are needed in tandem, Darnall said. Support for stronger patient competency in self-management of chronic pain is mounting.
“It’s vitally important that we help people know how to help themselves have less pain – how to steer their mind and body toward relief by using pain relief skills,” she said. “By so doing they can cultivate a critical level of control over their pain and are less at its mercy, which supports good mood and is shown to help people be more active as the impacts of pain diminish.”
Darnall outlined a recent development in the primary care setting that involves offering patients a brief program in pain-relief skills training. Within Veterans Affairs primary care, for example, patients receive several 30-minute sessions in pain reduction techniques. Outside of the VA, primary care clinics are incorporating an evidence-based, one-session 2-hour pain relief skills class called Empowered Relief, as standard care.
The class teaches participants three pain management skills and creates a personalized plan for each that includes a free app for ongoing daily use.
Since pain causes agitation in the central nervous system, manifested as fast heart rate, rapid breathing, muscle tension, and distress, people learn various ways to calm the central nervous system – with, for example, a sound technology known as binaural audio to deepen the relaxation response. “They also learn to identify and target worry about pain and develop self-soothing actions to interrupt unhelpful patterns,” Darnall said.
Data from randomized chronic pain studies, including one by her group using a virtual reality training program for lower back pain, show that 3 months after the training program people report clinically meaningful reductions in pain intensity, pain interference with daily activities, and sleep disturbance, as well as pain-related distress, anxiety, and fatigue
While psychological and complementary approaches have been effective in improving function and mood, there are barriers to accessing them, said Cheatle, such as lack of insurance coverage and the stigma associated with nontraditional, especially psychological, care.
Prevention
Good lifestyle behaviors promote better health as people age. “Maintaining a healthy weight, staying active, prioritizing good sleep, and avoiding smoking and alcohol use can support better health and buffer against chronic diseases and pain,” Darnall said.
Cheatle noted the importance of maintaining a safe work environment and avoiding injury risks by, for example, wearing a seatbelt or a cycling helmet.
The Future
“We need to ensure all individuals have access to effective, low-burden pain treatments, including evidence-based treatments they can receive from home so as to minimize treatment disparities.” Darnall said. Also needed is better comprehensive treatment for acute and chronic pain alike. “If we treat acute pain better, we will have fewer people transitioning to the chronic pain state.”
To that end, added Cheng, healthcare professionals in every specialty from doctors and nurses to psychologists and chiropractors need to develop co-competencies in pain management.
For Cheatle, the near future looks bleak. “There are some pioneering bioengineering approaches to reduce chronic pain and novel pharmacologic agents such as calcitonin gene-related peptide inhibitors for intractable migraines, but just changing insurance reimbursement for a comprehensive approach to chronic pain care and bolstering healthcare provider education on core pain competencies will benefit the over 50 million adults who suffer from chronic pain.”
Cheng, however, is more sanguine. “I don’t expect miracles in 10 years’ time, but we’re making rapid progress in understanding the genetics of chronic pain and the mechanisms of disease and therapy. We’re developing biomarkers to help in prognosis and monitor disease progress.” In the meantime, he pointed to an expanding array of non-pharmaceutical options, including neuromodulatory approaches such as nerve blocks and spinal cord stimulation.
Cheng, Cheatle, and Darnall disclosed no relevant competing interests.
A version of this article appeared on Medscape.com.
More Americans than ever are hurting with enduring, life-restricting pain. Like obesity, this condition is on the rise, according to figures in a new National Center for Health Statistics (NCHS) Data Brief from the Centers for Disease Control and Prevention (CDC).
Both types increased with age and with decreasing urbanization level. Women were more likely than men to have HICP (23.2% vs 7.3%).
Like obesity, chronic pain is multifactorial and is best managed with multidisciplinary intervention, said Jianguo Cheng, MD, PhD, a professor of anesthesiology and medical director of the Cleveland Clinic Consortium for Pain in Ohio. “It’s a complex mix of genetic, biological, and psychosocial dimensions that can cause ongoing pain out of proportion to the original limited injury that triggered it.”
While today’s longer lifespans are the primary driver of the increase, noted Martin Cheatle, PhD, an associate professor of psychiatry, anesthesiology, and critical care and director of behavioral medicine at the Penn Pain Medicine Center at the University of Pennsylvania’s Perelman School of Medicine, Philadelphia, another important factor is the more than 100 million Americans who suffer from obesity. “Obesity is a major risk factor for chronic pain conditions including advancing joint disease, low back pain, and diabetic neuropathies,” he said.
Age is an amplifier, agreed Beth Darnall, PhD, a professor of anesthesiology and perioperative and pain medicine and director of the Pain Relief Innovations Lab at Stanford University in Palo Alto, California, but the increases in chronic pain and HICP cut across age strata.
“Across the board we see striking increases in chronic pain, such as a 5% increase for those 65 and older, and a nearly 2% increase in HICP in that same age group,” Darnall said, referencing the changes from 2019 data in the new NCHS Data Brief. “And an almost 4% increase was observed for the youngest adult age category,18 to 29. Some of our research is now focusing on how to best treat chronic pain in young adults.”
The rise in chronic pain is broadly linked to the overall decline in the health of the US population, as indicated by the CDC 2024’s Chronic Disease Prevalence in the US: Sociodemographic and Geographic Variations by Zip Code Tabulation Area.
The Opioid Crisis and COVID
Beginning in 2016, in response to the opioid crisis and CDC guidelines, opioid prescribing for chronic pain rapidly dropped, both in terms of new prescriptions and tapering of doses of long-term users. “Reduced opioid prescribing yielded benefits for some patients but created new problems and harms for other patients,” said Darnall. Cheng added that the CDC’s recommendations on opioid prescribing were widely misinterpreted and were applied to patients with painful conditions such as cancer and sickle cell disease who were not intended to be affected by the guidelines. “In addition, although medical opioid prescribing dropped by 50%, overdose deaths from non-medical opioid sources increased by more 50%.”
Currently, most opioid overdoses are related to heroin, fentanyl, and newer drugs of abuse such as xylazine. “Most pain clinicians would agree that opioids are not first-line therapy for chronic non-cancer pain, but in a select number of well-vetted patients, opioids can be very effective in improving functionality and quality of life as part of a multimodal approach to pain care,” Cheatle said.
The impact of the opioid crisis is complex, said Cheatle, noting that only 8%-10% of pain patients on long-term opioid therapy develop a use disorder. “However, opioids were overly prescribed due to clinicians’ lack of training in core competencies of pain management and the insurance companies’ refusal to adequately cover non-opioid therapies such as acupuncture, cognitive behavioral therapy, extended physical therapy, and medical massage,” he said.
He pointed out that in the late 1990s there were more than 1000 multidisciplinary pain centers, whereas currently there are many fewer owing to lack of insurance reimbursement. “This results in more possibly avoidable invasive surgeries, which can further contribute to the increase in chronic pain.”
The COVID pandemic further exacerbated the pain problem and delayed access to timely medical interventions for many people. Some adopted a more sedentary lifestyle, already entrenched in today’s technology-driven society, leading in turn to weight gain and more chronic pain. “The isolation and lack of normal human connections during the pandemic could exacerbate pain and loss of autonomy,” Cheatle said. And some individuals developed painful neurologic conditions related to long-haul COVID, for which there is no effective treatment.
Best Approach
“Historically, pain has been treated as a purely biomedical issue. Bringing a biopsychosocial perspective to pain care can support pain relief,” said Darnall. Multiple national clinical guidance documents have called for a comprehensive approach that considers the whole person: their circumstances, their needs, their stressors, and their environment. “And we must provide patients meaningful access to the lowest-risk, non-pharmacologic treatments first – and ideally early on,” she said.
Even effective medications rarely make a person pain free, so other approaches are needed in tandem, Darnall said. Support for stronger patient competency in self-management of chronic pain is mounting.
“It’s vitally important that we help people know how to help themselves have less pain – how to steer their mind and body toward relief by using pain relief skills,” she said. “By so doing they can cultivate a critical level of control over their pain and are less at its mercy, which supports good mood and is shown to help people be more active as the impacts of pain diminish.”
Darnall outlined a recent development in the primary care setting that involves offering patients a brief program in pain-relief skills training. Within Veterans Affairs primary care, for example, patients receive several 30-minute sessions in pain reduction techniques. Outside of the VA, primary care clinics are incorporating an evidence-based, one-session 2-hour pain relief skills class called Empowered Relief, as standard care.
The class teaches participants three pain management skills and creates a personalized plan for each that includes a free app for ongoing daily use.
Since pain causes agitation in the central nervous system, manifested as fast heart rate, rapid breathing, muscle tension, and distress, people learn various ways to calm the central nervous system – with, for example, a sound technology known as binaural audio to deepen the relaxation response. “They also learn to identify and target worry about pain and develop self-soothing actions to interrupt unhelpful patterns,” Darnall said.
Data from randomized chronic pain studies, including one by her group using a virtual reality training program for lower back pain, show that 3 months after the training program people report clinically meaningful reductions in pain intensity, pain interference with daily activities, and sleep disturbance, as well as pain-related distress, anxiety, and fatigue
While psychological and complementary approaches have been effective in improving function and mood, there are barriers to accessing them, said Cheatle, such as lack of insurance coverage and the stigma associated with nontraditional, especially psychological, care.
Prevention
Good lifestyle behaviors promote better health as people age. “Maintaining a healthy weight, staying active, prioritizing good sleep, and avoiding smoking and alcohol use can support better health and buffer against chronic diseases and pain,” Darnall said.
Cheatle noted the importance of maintaining a safe work environment and avoiding injury risks by, for example, wearing a seatbelt or a cycling helmet.
The Future
“We need to ensure all individuals have access to effective, low-burden pain treatments, including evidence-based treatments they can receive from home so as to minimize treatment disparities.” Darnall said. Also needed is better comprehensive treatment for acute and chronic pain alike. “If we treat acute pain better, we will have fewer people transitioning to the chronic pain state.”
To that end, added Cheng, healthcare professionals in every specialty from doctors and nurses to psychologists and chiropractors need to develop co-competencies in pain management.
For Cheatle, the near future looks bleak. “There are some pioneering bioengineering approaches to reduce chronic pain and novel pharmacologic agents such as calcitonin gene-related peptide inhibitors for intractable migraines, but just changing insurance reimbursement for a comprehensive approach to chronic pain care and bolstering healthcare provider education on core pain competencies will benefit the over 50 million adults who suffer from chronic pain.”
Cheng, however, is more sanguine. “I don’t expect miracles in 10 years’ time, but we’re making rapid progress in understanding the genetics of chronic pain and the mechanisms of disease and therapy. We’re developing biomarkers to help in prognosis and monitor disease progress.” In the meantime, he pointed to an expanding array of non-pharmaceutical options, including neuromodulatory approaches such as nerve blocks and spinal cord stimulation.
Cheng, Cheatle, and Darnall disclosed no relevant competing interests.
A version of this article appeared on Medscape.com.
Total Intravenous Anesthesia Enables Earlier Facial Nerve Monitoring Than Sevoflurane in Ear Surgery
TOPLINE:
Total intravenous anesthesia (TIVA) enables earlier intraoperative monitoring of facial nerve activity than sevoflurane anesthesia during ear surgery, with reduced patient-ventilator dyssynchrony and fewer requirements for postoperative antiemetics.
METHODOLOGY:
- Researchers evaluated the difference in the timeliness of intraoperative monitoring of facial nerve activity during ear surgery with TIVA vs sevoflurane anesthesia.
- They included 98 patients aged 18-74 years undergoing ear surgery between November 2021 and November 2022; patients were randomly assigned to receive either TIVA or sevoflurane during the procedure. Of these, 92 were included in the final analysis.
- Neuromuscular function was monitored quantitatively throughout anesthesia with train-of-four counts and train-of-four ratios.
- The time from the administration of rocuronium to the start of facial nerve monitoring was recorded.
- The primary outcome measure focused on the recovery index, defined as the time interval between a train-of-four ratio of 0.25 and 0.75; the key secondary outcome was the time to reach a train-of-four ratio of 0.25 from rocuronium administration.
TAKEAWAY:
- The time to reach a train-of-four ratio of 0.25 was achieved earlier with TIVA than with sevoflurane (34 minutes vs 51 minutes; P < .001).
- Patient-ventilator dyssynchrony occurred less frequently in the TIVA group than in the sevoflurane group (15% vs 39%; P = .01).
- Postoperative requests for antiemetics were less frequent in the TIVA group than in the sevoflurane group (2% vs 17%; P = .03).
IN PRACTICE:
“We suggest that TIVA may be a better choice than sevoflurane anesthesia to meet an earlier request” for intraoperative facial nerve monitoring by surgeons, the study authors wrote.
SOURCE:
The study was led by Yu Jeong Bang, MD, of the Department of Anesthesiology and Pain Medicine at Sungkyunkwan University School of Medicine, in Seoul, Republic of Korea. It was published online on November 27, 2024, in The Canadian Journal of Anesthesia.
LIMITATIONS:
A careful interpretation of results may be necessary when clinicians use balanced anesthesia, such as sevoflurane with adjuvants like opioids or nonopioids. The feasibility of intraoperative facial nerve monitoring was decided by the surgeon during surgery, and the lowest stimulation intensity threshold for electromyography amplitude was not detected, as it was not the focus of this study. Although patients requiring intraoperative facial nerve monitoring during ear surgery were enrolled, some did not undergo the procedure based on the surgeon’s judgment.
DISCLOSURES:
This study did not receive any funding. The authors disclosed no relevant conflicts of interest.
This article was created using several editorial tools, including artificial intelligence, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Total intravenous anesthesia (TIVA) enables earlier intraoperative monitoring of facial nerve activity than sevoflurane anesthesia during ear surgery, with reduced patient-ventilator dyssynchrony and fewer requirements for postoperative antiemetics.
METHODOLOGY:
- Researchers evaluated the difference in the timeliness of intraoperative monitoring of facial nerve activity during ear surgery with TIVA vs sevoflurane anesthesia.
- They included 98 patients aged 18-74 years undergoing ear surgery between November 2021 and November 2022; patients were randomly assigned to receive either TIVA or sevoflurane during the procedure. Of these, 92 were included in the final analysis.
- Neuromuscular function was monitored quantitatively throughout anesthesia with train-of-four counts and train-of-four ratios.
- The time from the administration of rocuronium to the start of facial nerve monitoring was recorded.
- The primary outcome measure focused on the recovery index, defined as the time interval between a train-of-four ratio of 0.25 and 0.75; the key secondary outcome was the time to reach a train-of-four ratio of 0.25 from rocuronium administration.
TAKEAWAY:
- The time to reach a train-of-four ratio of 0.25 was achieved earlier with TIVA than with sevoflurane (34 minutes vs 51 minutes; P < .001).
- Patient-ventilator dyssynchrony occurred less frequently in the TIVA group than in the sevoflurane group (15% vs 39%; P = .01).
- Postoperative requests for antiemetics were less frequent in the TIVA group than in the sevoflurane group (2% vs 17%; P = .03).
IN PRACTICE:
“We suggest that TIVA may be a better choice than sevoflurane anesthesia to meet an earlier request” for intraoperative facial nerve monitoring by surgeons, the study authors wrote.
SOURCE:
The study was led by Yu Jeong Bang, MD, of the Department of Anesthesiology and Pain Medicine at Sungkyunkwan University School of Medicine, in Seoul, Republic of Korea. It was published online on November 27, 2024, in The Canadian Journal of Anesthesia.
LIMITATIONS:
A careful interpretation of results may be necessary when clinicians use balanced anesthesia, such as sevoflurane with adjuvants like opioids or nonopioids. The feasibility of intraoperative facial nerve monitoring was decided by the surgeon during surgery, and the lowest stimulation intensity threshold for electromyography amplitude was not detected, as it was not the focus of this study. Although patients requiring intraoperative facial nerve monitoring during ear surgery were enrolled, some did not undergo the procedure based on the surgeon’s judgment.
DISCLOSURES:
This study did not receive any funding. The authors disclosed no relevant conflicts of interest.
This article was created using several editorial tools, including artificial intelligence, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Total intravenous anesthesia (TIVA) enables earlier intraoperative monitoring of facial nerve activity than sevoflurane anesthesia during ear surgery, with reduced patient-ventilator dyssynchrony and fewer requirements for postoperative antiemetics.
METHODOLOGY:
- Researchers evaluated the difference in the timeliness of intraoperative monitoring of facial nerve activity during ear surgery with TIVA vs sevoflurane anesthesia.
- They included 98 patients aged 18-74 years undergoing ear surgery between November 2021 and November 2022; patients were randomly assigned to receive either TIVA or sevoflurane during the procedure. Of these, 92 were included in the final analysis.
- Neuromuscular function was monitored quantitatively throughout anesthesia with train-of-four counts and train-of-four ratios.
- The time from the administration of rocuronium to the start of facial nerve monitoring was recorded.
- The primary outcome measure focused on the recovery index, defined as the time interval between a train-of-four ratio of 0.25 and 0.75; the key secondary outcome was the time to reach a train-of-four ratio of 0.25 from rocuronium administration.
TAKEAWAY:
- The time to reach a train-of-four ratio of 0.25 was achieved earlier with TIVA than with sevoflurane (34 minutes vs 51 minutes; P < .001).
- Patient-ventilator dyssynchrony occurred less frequently in the TIVA group than in the sevoflurane group (15% vs 39%; P = .01).
- Postoperative requests for antiemetics were less frequent in the TIVA group than in the sevoflurane group (2% vs 17%; P = .03).
IN PRACTICE:
“We suggest that TIVA may be a better choice than sevoflurane anesthesia to meet an earlier request” for intraoperative facial nerve monitoring by surgeons, the study authors wrote.
SOURCE:
The study was led by Yu Jeong Bang, MD, of the Department of Anesthesiology and Pain Medicine at Sungkyunkwan University School of Medicine, in Seoul, Republic of Korea. It was published online on November 27, 2024, in The Canadian Journal of Anesthesia.
LIMITATIONS:
A careful interpretation of results may be necessary when clinicians use balanced anesthesia, such as sevoflurane with adjuvants like opioids or nonopioids. The feasibility of intraoperative facial nerve monitoring was decided by the surgeon during surgery, and the lowest stimulation intensity threshold for electromyography amplitude was not detected, as it was not the focus of this study. Although patients requiring intraoperative facial nerve monitoring during ear surgery were enrolled, some did not undergo the procedure based on the surgeon’s judgment.
DISCLOSURES:
This study did not receive any funding. The authors disclosed no relevant conflicts of interest.
This article was created using several editorial tools, including artificial intelligence, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.