User login
In children with moderate to severe acute asthma, albuterol delivered by a conventional jet nebulizer led to more improvement in forced expiratory volume in 1 second (FEV1) than delivery via a breath-enhanced nebulizer.
Only one previous study has compared the two types of nebulizers in children with acute asthma. It showed that the new technology is noninferior to the older device, but it had a small sample size and did not examine spirometry data.
Mike Gardiner, MD, of the department of pediatrics, University of California, San Diego, and Matthew H. Wilkinson, MD, of the department of pediatrics, University of Texas at Austin, conducted a randomized, observer-blind study to look at effectiveness of these two nebulizers in a larger population of pediatric users. The results were published in the Journal of Pediatrics.
At a large, urban pediatric emergency department, researchers randomized 107 children (aged 6-18 years) presenting with a moderate to severe asthma exacerbation to receive one or the other nebulizer.
Children treated with the conventional jet nebulizer had a greater improvement in FEV1 (+13.8% vs. +9.1% of predicted; P = .04). The improvements were similar in a subgroup analysis of 57 subjects who met ATS/ERS (American Thoracic Society/ European Respiratory Society) spirometry guidelines (+14.5% vs. +8.5% of predicted; P = .03).
The researchers found no significant differences in changes in Pediatric Asthma Score, Pediatric Asthma Severity Score, ED length of stay, or admission rate. There was no significant difference in side effects between the two groups.
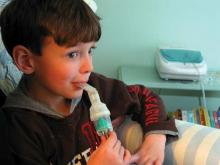
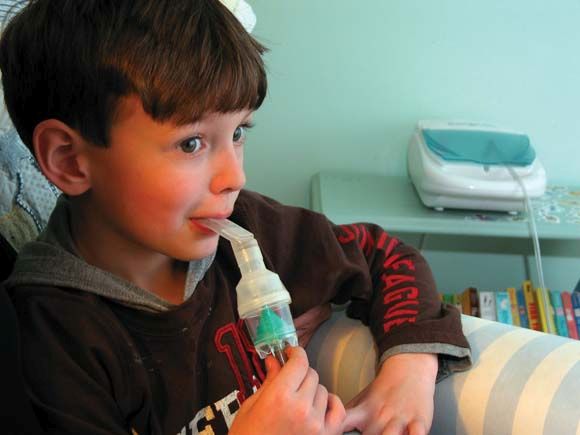
Breath-enhanced nebulizers use a holding chamber to store continuously nebulized medication, and one-way valves that direct exhaled air away from the holding chamber. The system reduces medication loss during exhalation and delivers a bolus of medication.
In lung models and healthy adult controls, breath-enhanced nebulizers achieved more effective lung deposition of aerosol. The authors speculate that the reduced clinical effect of the breath-enhanced nebulizer could be because the design of the mouthpiece, which allows a nonaerosolized “dead space” volume to be inhaled first. This volume may have a greater clinical impact in children than in adults.
Children experiencing asthma also have a rapid and shallow breathing pattern, which could also lead to a larger contribution of “dead space” to the overall dose, thus reducing drug exposure.
The study, Comparison of Breath-Enhanced and T-Piece Nebulizers in Children With Acute Asthma (NCT02566902) was funded by the University of Texas Southwestern, Austin. The authors declared no conflicts of interest.
SOURCE: Gardiner M, Wilkinson M. J Pediatr. 2019;204:245-9.
In children with moderate to severe acute asthma, albuterol delivered by a conventional jet nebulizer led to more improvement in forced expiratory volume in 1 second (FEV1) than delivery via a breath-enhanced nebulizer.
Only one previous study has compared the two types of nebulizers in children with acute asthma. It showed that the new technology is noninferior to the older device, but it had a small sample size and did not examine spirometry data.
Mike Gardiner, MD, of the department of pediatrics, University of California, San Diego, and Matthew H. Wilkinson, MD, of the department of pediatrics, University of Texas at Austin, conducted a randomized, observer-blind study to look at effectiveness of these two nebulizers in a larger population of pediatric users. The results were published in the Journal of Pediatrics.
At a large, urban pediatric emergency department, researchers randomized 107 children (aged 6-18 years) presenting with a moderate to severe asthma exacerbation to receive one or the other nebulizer.
Children treated with the conventional jet nebulizer had a greater improvement in FEV1 (+13.8% vs. +9.1% of predicted; P = .04). The improvements were similar in a subgroup analysis of 57 subjects who met ATS/ERS (American Thoracic Society/ European Respiratory Society) spirometry guidelines (+14.5% vs. +8.5% of predicted; P = .03).
The researchers found no significant differences in changes in Pediatric Asthma Score, Pediatric Asthma Severity Score, ED length of stay, or admission rate. There was no significant difference in side effects between the two groups.
Breath-enhanced nebulizers use a holding chamber to store continuously nebulized medication, and one-way valves that direct exhaled air away from the holding chamber. The system reduces medication loss during exhalation and delivers a bolus of medication.
In lung models and healthy adult controls, breath-enhanced nebulizers achieved more effective lung deposition of aerosol. The authors speculate that the reduced clinical effect of the breath-enhanced nebulizer could be because the design of the mouthpiece, which allows a nonaerosolized “dead space” volume to be inhaled first. This volume may have a greater clinical impact in children than in adults.
Children experiencing asthma also have a rapid and shallow breathing pattern, which could also lead to a larger contribution of “dead space” to the overall dose, thus reducing drug exposure.
The study, Comparison of Breath-Enhanced and T-Piece Nebulizers in Children With Acute Asthma (NCT02566902) was funded by the University of Texas Southwestern, Austin. The authors declared no conflicts of interest.
SOURCE: Gardiner M, Wilkinson M. J Pediatr. 2019;204:245-9.
In children with moderate to severe acute asthma, albuterol delivered by a conventional jet nebulizer led to more improvement in forced expiratory volume in 1 second (FEV1) than delivery via a breath-enhanced nebulizer.
Only one previous study has compared the two types of nebulizers in children with acute asthma. It showed that the new technology is noninferior to the older device, but it had a small sample size and did not examine spirometry data.
Mike Gardiner, MD, of the department of pediatrics, University of California, San Diego, and Matthew H. Wilkinson, MD, of the department of pediatrics, University of Texas at Austin, conducted a randomized, observer-blind study to look at effectiveness of these two nebulizers in a larger population of pediatric users. The results were published in the Journal of Pediatrics.
At a large, urban pediatric emergency department, researchers randomized 107 children (aged 6-18 years) presenting with a moderate to severe asthma exacerbation to receive one or the other nebulizer.
Children treated with the conventional jet nebulizer had a greater improvement in FEV1 (+13.8% vs. +9.1% of predicted; P = .04). The improvements were similar in a subgroup analysis of 57 subjects who met ATS/ERS (American Thoracic Society/ European Respiratory Society) spirometry guidelines (+14.5% vs. +8.5% of predicted; P = .03).
The researchers found no significant differences in changes in Pediatric Asthma Score, Pediatric Asthma Severity Score, ED length of stay, or admission rate. There was no significant difference in side effects between the two groups.
Breath-enhanced nebulizers use a holding chamber to store continuously nebulized medication, and one-way valves that direct exhaled air away from the holding chamber. The system reduces medication loss during exhalation and delivers a bolus of medication.
In lung models and healthy adult controls, breath-enhanced nebulizers achieved more effective lung deposition of aerosol. The authors speculate that the reduced clinical effect of the breath-enhanced nebulizer could be because the design of the mouthpiece, which allows a nonaerosolized “dead space” volume to be inhaled first. This volume may have a greater clinical impact in children than in adults.
Children experiencing asthma also have a rapid and shallow breathing pattern, which could also lead to a larger contribution of “dead space” to the overall dose, thus reducing drug exposure.
The study, Comparison of Breath-Enhanced and T-Piece Nebulizers in Children With Acute Asthma (NCT02566902) was funded by the University of Texas Southwestern, Austin. The authors declared no conflicts of interest.
SOURCE: Gardiner M, Wilkinson M. J Pediatr. 2019;204:245-9.
FROM THE JOURNAL OF PEDIATRICS
Key clinical point: New nebulizers worked better in adults, but pediatric populations may pose a challenge.
Major finding: FEV1 improved +13.8% in standard inhalers vs. +9.1% in breath-enhanced analyzers.
Study details: Randomized, controlled trial (n = 107).
Disclosures: The study was funded by the University of Texas Southwestern, Austin. The authors declared no conflicts of interest.
Source: Gardiner M, Wilkinson M. J Pediatr. 2019;204:245-9.