User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
New guideline for managing MCL
Rituximab should be included in first-line chemotherapy when treating mantle cell lymphoma, according to a new management guideline from the British Society for Haematology.
The best outcome data is for the R-CHOP regimen (rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisolone) followed by maintenance treatment with rituximab, wrote Pamela McKay, MD, of Beatson West of Scotland Cancer Centre in Glasgow, and her colleagues. The report was published in the British Journal of Haematology. But the combination of rituximab and bendamustine is also effective and a more favorable safety profile, according to the guideline. Single agent rituximab is not recommended.
At relapse, the guideline calls on physicians to take an individualized approach based on age, comorbidities, performance status, and response to prior therapy. Some options to consider include ibrutinib as a single agent or rituximab plus chemotherapy. The authors cautioned that there is little evidence to support maintenance rituximab after relapse treatment.
The guideline also explores the role of autologous stem cell transplantation (ASCT) and allogeneic SCT (alloSCT). The authors recommend that ASCT be considered as consolidation of first-line therapy for patients who are fit for intensive therapy. AlloSCT is a viable option in second remission among fit patients who have an appropriate donor and it may also be effective as a rescue therapy for patients who relapse after ASCT. But alloSCT is appropriate only as a first-line therapy for high-risk patients and is best used as part of a clinical trial, according to the recommendations.
The British Society of Haematology previously issued guidance on mantle cell lymphoma in 2012, but the updated document includes new drug therapeutic options and transplant data. The guideline includes a therapeutic algorithm to assist physicians in choosing first-line therapy, options after first relapse, and management in the case of higher relapse.
The guideline authors reported having no conflicts of interest.
SOURCE: McKay P et al. Br J Haematol. 2018 Jul;182(1):46-62.
Rituximab should be included in first-line chemotherapy when treating mantle cell lymphoma, according to a new management guideline from the British Society for Haematology.
The best outcome data is for the R-CHOP regimen (rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisolone) followed by maintenance treatment with rituximab, wrote Pamela McKay, MD, of Beatson West of Scotland Cancer Centre in Glasgow, and her colleagues. The report was published in the British Journal of Haematology. But the combination of rituximab and bendamustine is also effective and a more favorable safety profile, according to the guideline. Single agent rituximab is not recommended.
At relapse, the guideline calls on physicians to take an individualized approach based on age, comorbidities, performance status, and response to prior therapy. Some options to consider include ibrutinib as a single agent or rituximab plus chemotherapy. The authors cautioned that there is little evidence to support maintenance rituximab after relapse treatment.
The guideline also explores the role of autologous stem cell transplantation (ASCT) and allogeneic SCT (alloSCT). The authors recommend that ASCT be considered as consolidation of first-line therapy for patients who are fit for intensive therapy. AlloSCT is a viable option in second remission among fit patients who have an appropriate donor and it may also be effective as a rescue therapy for patients who relapse after ASCT. But alloSCT is appropriate only as a first-line therapy for high-risk patients and is best used as part of a clinical trial, according to the recommendations.
The British Society of Haematology previously issued guidance on mantle cell lymphoma in 2012, but the updated document includes new drug therapeutic options and transplant data. The guideline includes a therapeutic algorithm to assist physicians in choosing first-line therapy, options after first relapse, and management in the case of higher relapse.
The guideline authors reported having no conflicts of interest.
SOURCE: McKay P et al. Br J Haematol. 2018 Jul;182(1):46-62.
Rituximab should be included in first-line chemotherapy when treating mantle cell lymphoma, according to a new management guideline from the British Society for Haematology.
The best outcome data is for the R-CHOP regimen (rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisolone) followed by maintenance treatment with rituximab, wrote Pamela McKay, MD, of Beatson West of Scotland Cancer Centre in Glasgow, and her colleagues. The report was published in the British Journal of Haematology. But the combination of rituximab and bendamustine is also effective and a more favorable safety profile, according to the guideline. Single agent rituximab is not recommended.
At relapse, the guideline calls on physicians to take an individualized approach based on age, comorbidities, performance status, and response to prior therapy. Some options to consider include ibrutinib as a single agent or rituximab plus chemotherapy. The authors cautioned that there is little evidence to support maintenance rituximab after relapse treatment.
The guideline also explores the role of autologous stem cell transplantation (ASCT) and allogeneic SCT (alloSCT). The authors recommend that ASCT be considered as consolidation of first-line therapy for patients who are fit for intensive therapy. AlloSCT is a viable option in second remission among fit patients who have an appropriate donor and it may also be effective as a rescue therapy for patients who relapse after ASCT. But alloSCT is appropriate only as a first-line therapy for high-risk patients and is best used as part of a clinical trial, according to the recommendations.
The British Society of Haematology previously issued guidance on mantle cell lymphoma in 2012, but the updated document includes new drug therapeutic options and transplant data. The guideline includes a therapeutic algorithm to assist physicians in choosing first-line therapy, options after first relapse, and management in the case of higher relapse.
The guideline authors reported having no conflicts of interest.
SOURCE: McKay P et al. Br J Haematol. 2018 Jul;182(1):46-62.
FROM THE BRITISH JOURNAL OF HAEMATOLOGY
Ibrutinib/venetoclax shows early promise in relapsed CLL
STOCKHOLM – A chemotherapy-free regimen of ibrutinib plus venetoclax was generally safe and showed promising early efficacy in patients with relapsed or refractory chronic lymphocytic leukemia, investigators reported.
A planned interim analysis performed after the first 15 patients who had received two cycles of ibrutinib plus one of ibrutinib and venetoclax showed no treatment-related deaths or treatment interruptions, and all patients had clinical responses, including 8 with complete clinical remission (CR), reported Carsten U. Niemann, MD, PhD, from Rigshospitalet in Copenhagen, and his colleagues.
The goal of the ongoing VISION/HOVEN 141 study is to evaluate whether minimal residual disease (MRD)–guided therapy with the Bruton tyrosine kinase inhibitor ibrutinib and the BCL2 inhibitor venetoclax could lead to MRD negativity and allow select patients to stop treatment, Dr. Niemann said in an interview at the annual congress of the European Hematology Association.
“It’s a 100% clinical response rate and 53% CR. Obviously these are clinical responses, so we don’t have the CT scans, we don’t have the bone marrow biopsies, but we’re very happy to see even in the relapsed/refractory setting such good response rates,” he said.
The investigators are enrolling patients with relapsed/refractory chronic lymphocytic leukemia or small lymphocytic leukemia requiring treatment and starting all patients on ibrutinib 420 mg daily for the first 2 cycles, with venetoclax added in a 5-week ramp-up from 20 mg beginning with cycle 3 to a final dose of 400 mg daily for 15 total treatment cycles.
At the end of the induction phase, patients who are determined to be MRD-negative by flow cytometry at cycles 12 and 15, and by bone marrow at cycle 15, are randomized on a 1:2 basis to ibrutinib maintenance until disease progression or intolerable toxicity, or to observation until progression or loss of MRD negativity, at which time they start maintenance with ibrutinib until progression or toxicity, plus 12 months of venetoclax.
All 15 patients who were followed for 3 months had clinical responses, including 8 CRs (53%), 6 partial remissions (40%), and 1 partial remission with lymphocytosis (7%).
Three patients had ibrutinib dose reductions and two had venetoclax dose reductions, but no patients stopped treatment. Three patients had grade 2 adverse events (AEs), three had grade 3 AEs, and two had grade 4 AEs. There were no grade 5 AEs.
Two patients had serious AEs during the first two cycles with ibrutinib alone, one of which was a case of febrile neutropenia and one which was an adenocarcinoma of the lung. There were no serious AEs reported during venetoclax ramp-up. To date, there have been no cases of tumor lysis syndrome, atrial fibrillation, or bleeding events reported.
The results suggest that treatment with ibrutinib and venetoclax ramp-up is manageable in this patient population, and the study is ongoing, with further results expected to be reported at either the 2018 annual meeting of the American Society of Hematology or the 2019 annual meeting of the American Society of Clinical Oncology, Dr. Niemann said.
The study is supported by AbbVie and Janssen, which supplied the drugs and had the right to comment on the presentation. Dr. Niemann has previously disclosed consultancy fees from those companies and others.
SOURCE: Niemann CU et al. EHA Congress, Abstract PF346.
STOCKHOLM – A chemotherapy-free regimen of ibrutinib plus venetoclax was generally safe and showed promising early efficacy in patients with relapsed or refractory chronic lymphocytic leukemia, investigators reported.
A planned interim analysis performed after the first 15 patients who had received two cycles of ibrutinib plus one of ibrutinib and venetoclax showed no treatment-related deaths or treatment interruptions, and all patients had clinical responses, including 8 with complete clinical remission (CR), reported Carsten U. Niemann, MD, PhD, from Rigshospitalet in Copenhagen, and his colleagues.
The goal of the ongoing VISION/HOVEN 141 study is to evaluate whether minimal residual disease (MRD)–guided therapy with the Bruton tyrosine kinase inhibitor ibrutinib and the BCL2 inhibitor venetoclax could lead to MRD negativity and allow select patients to stop treatment, Dr. Niemann said in an interview at the annual congress of the European Hematology Association.
“It’s a 100% clinical response rate and 53% CR. Obviously these are clinical responses, so we don’t have the CT scans, we don’t have the bone marrow biopsies, but we’re very happy to see even in the relapsed/refractory setting such good response rates,” he said.
The investigators are enrolling patients with relapsed/refractory chronic lymphocytic leukemia or small lymphocytic leukemia requiring treatment and starting all patients on ibrutinib 420 mg daily for the first 2 cycles, with venetoclax added in a 5-week ramp-up from 20 mg beginning with cycle 3 to a final dose of 400 mg daily for 15 total treatment cycles.
At the end of the induction phase, patients who are determined to be MRD-negative by flow cytometry at cycles 12 and 15, and by bone marrow at cycle 15, are randomized on a 1:2 basis to ibrutinib maintenance until disease progression or intolerable toxicity, or to observation until progression or loss of MRD negativity, at which time they start maintenance with ibrutinib until progression or toxicity, plus 12 months of venetoclax.
All 15 patients who were followed for 3 months had clinical responses, including 8 CRs (53%), 6 partial remissions (40%), and 1 partial remission with lymphocytosis (7%).
Three patients had ibrutinib dose reductions and two had venetoclax dose reductions, but no patients stopped treatment. Three patients had grade 2 adverse events (AEs), three had grade 3 AEs, and two had grade 4 AEs. There were no grade 5 AEs.
Two patients had serious AEs during the first two cycles with ibrutinib alone, one of which was a case of febrile neutropenia and one which was an adenocarcinoma of the lung. There were no serious AEs reported during venetoclax ramp-up. To date, there have been no cases of tumor lysis syndrome, atrial fibrillation, or bleeding events reported.
The results suggest that treatment with ibrutinib and venetoclax ramp-up is manageable in this patient population, and the study is ongoing, with further results expected to be reported at either the 2018 annual meeting of the American Society of Hematology or the 2019 annual meeting of the American Society of Clinical Oncology, Dr. Niemann said.
The study is supported by AbbVie and Janssen, which supplied the drugs and had the right to comment on the presentation. Dr. Niemann has previously disclosed consultancy fees from those companies and others.
SOURCE: Niemann CU et al. EHA Congress, Abstract PF346.
STOCKHOLM – A chemotherapy-free regimen of ibrutinib plus venetoclax was generally safe and showed promising early efficacy in patients with relapsed or refractory chronic lymphocytic leukemia, investigators reported.
A planned interim analysis performed after the first 15 patients who had received two cycles of ibrutinib plus one of ibrutinib and venetoclax showed no treatment-related deaths or treatment interruptions, and all patients had clinical responses, including 8 with complete clinical remission (CR), reported Carsten U. Niemann, MD, PhD, from Rigshospitalet in Copenhagen, and his colleagues.
The goal of the ongoing VISION/HOVEN 141 study is to evaluate whether minimal residual disease (MRD)–guided therapy with the Bruton tyrosine kinase inhibitor ibrutinib and the BCL2 inhibitor venetoclax could lead to MRD negativity and allow select patients to stop treatment, Dr. Niemann said in an interview at the annual congress of the European Hematology Association.
“It’s a 100% clinical response rate and 53% CR. Obviously these are clinical responses, so we don’t have the CT scans, we don’t have the bone marrow biopsies, but we’re very happy to see even in the relapsed/refractory setting such good response rates,” he said.
The investigators are enrolling patients with relapsed/refractory chronic lymphocytic leukemia or small lymphocytic leukemia requiring treatment and starting all patients on ibrutinib 420 mg daily for the first 2 cycles, with venetoclax added in a 5-week ramp-up from 20 mg beginning with cycle 3 to a final dose of 400 mg daily for 15 total treatment cycles.
At the end of the induction phase, patients who are determined to be MRD-negative by flow cytometry at cycles 12 and 15, and by bone marrow at cycle 15, are randomized on a 1:2 basis to ibrutinib maintenance until disease progression or intolerable toxicity, or to observation until progression or loss of MRD negativity, at which time they start maintenance with ibrutinib until progression or toxicity, plus 12 months of venetoclax.
All 15 patients who were followed for 3 months had clinical responses, including 8 CRs (53%), 6 partial remissions (40%), and 1 partial remission with lymphocytosis (7%).
Three patients had ibrutinib dose reductions and two had venetoclax dose reductions, but no patients stopped treatment. Three patients had grade 2 adverse events (AEs), three had grade 3 AEs, and two had grade 4 AEs. There were no grade 5 AEs.
Two patients had serious AEs during the first two cycles with ibrutinib alone, one of which was a case of febrile neutropenia and one which was an adenocarcinoma of the lung. There were no serious AEs reported during venetoclax ramp-up. To date, there have been no cases of tumor lysis syndrome, atrial fibrillation, or bleeding events reported.
The results suggest that treatment with ibrutinib and venetoclax ramp-up is manageable in this patient population, and the study is ongoing, with further results expected to be reported at either the 2018 annual meeting of the American Society of Hematology or the 2019 annual meeting of the American Society of Clinical Oncology, Dr. Niemann said.
The study is supported by AbbVie and Janssen, which supplied the drugs and had the right to comment on the presentation. Dr. Niemann has previously disclosed consultancy fees from those companies and others.
SOURCE: Niemann CU et al. EHA Congress, Abstract PF346.
REPORTING FROM THE EHA CONGRESS
Key clinical point:
Major finding: All of the 15 patients analyzed to date had clinical responses to the combination, including 8 complete clinical remissions.
Study details: An ongoing, open-label, phase 2, randomized trial in patients with relapsed/refractory chronic lymphocytic leukemia/small lymphocytic leukemia.
Disclosures: The study is supported by AbbVie and Janssen, which supplied the drugs and had the right to comment on the presentation. Dr. Niemann has previously disclosed consultancy fees from those companies and others.
Source: Niemann CU et al. EHA Congress, Abstract PF346
Rapid venetoclax dose escalation aids relapsed CLL
STOCKHOLM – Patients with chronic lymphocytic leukemia (CLL) who experience relapse after therapy with a B-cell receptor signaling inhibitor tend to have a swiftly progressive disease course that requires immediate intervention. For these patients, a rapid venetoclax dose-escalation protocol may be a safe way to quickly regain disease control, and possibly bridge to salvage therapies, investigators reported.
Of 15 patients with CLL who relapsed after treatment with a B-cell receptor inhibitor (BCRi), all were able to get to their target dose of venetoclax under close inpatient monitoring at a median of 12 days, compared with the 35 days usually required for venetoclax dose escalation, reported Farrukh T. Awan, MD, of Ohio State University Comprehensive Cancer Center in Columbus, and his colleagues.
Only two patients developed clinical tumor lysis syndrome (TLS), a common occurrence with venetoclax therapy, and this adverse event was manageable, Dr. Awan said at the annual congress of the European Hematology Association.
“The reason why we have been doing a slow ramp up on venetoclax is the original toxicity issues that we saw early on,” he said in an interview. “But unfortunately, a lot of patients are progressing on these new agents and have very rapid disease progression, and what we have seen is that if you stop the ibrutinib, the disease progresses very quickly, and by the time they can get up to the effective dose of venetoclax, they’re too sick to continue, or they might even die from disease progression.”
To combat this problem, Dr. Awan and his colleagues developed a rapid dose escalation protocol that would ramp up from 20 mg to 400 mg, with increases every 1 or 2 days depending on tolerability and incident TLS. Lab tests for TLS were evaluated every 4-8 hours.
All patients were closely monitored in the hospital, and all were started on allopurinol or other uric acid–lowering agents before starting on venetoclax.
The investigators reported safety and efficacy outcomes for the patients in a retrospective analysis.
The median age of the patients, 12 men and 3 women, was 65 years (range, 58-86 years). Seven patients had Eastern Cooperative Oncology Group Performance Status of 0, seven had an ECOG score of 1, and one had a score of 2-4.
Ten patients had most recently been treated with a BCRi, either a Bruton’s tyrosine kinase inhibitor (ibrutinib or acalabrutinib), idelalisib, or entospletinib. Three patients received ibrutinib plus chemotherapy, and two received rituximab and dexamethasone followed by rituximab maintenance.
The median time to full venetoclax dose was 12 days (range, 5-21 days) and all 15 patients reached the target dose. The mean length of stay during the ramp-up period was 9.5 days (range, 6-22 days).
The incidence of clinical TLS was 13.2%, occurring in two patients, one at the initial 20-mg dose, and one at the 200-mg dose level. Another five patients had asymptomatic TLS. Other treatment-related adverse events were anemia in seven patients, neutropenia in six patients, thrombocytopenia in five patients, and lung infection in one patient.
Twelve patients had a partial response, one had stable disease, and two had progressive disease. The mean time to best response was 71 days.
One-year progression-free survival was 49%, and 1-year overall survival was 68%.
The investigators found that for patients who still have some disease control with a BCRi, it may be possible to keep them on that drug while transitioning to venetoclax. The rapid dose escalation protocol should only be attempted in highly experience comprehensive cancer centers, Dr. Awan said.
“Under very close monitoring in an experienced inpatient setting, where the nurses are very used to doing this on a weekly basis in a very high volume center, I think that our data show that we could do this without affecting toxicity significantly or mortality,” he said.
Venetoclax therapy could buy enough time for patients to bridge to other options, such as chimeric antigen receptor (CAR) T-cell therapy or allogeneic stem cell transplant, he noted.
“But if we had waited 4 weeks, most of these patients would not have made it,” he said.
The study was internally funded. Dr. Awan reported research funding from Gilead, Pharmacyclics, AbbVie, and Janssen.
SOURCE: Koenig K et al. EHA Congress, Abstract PF357.
STOCKHOLM – Patients with chronic lymphocytic leukemia (CLL) who experience relapse after therapy with a B-cell receptor signaling inhibitor tend to have a swiftly progressive disease course that requires immediate intervention. For these patients, a rapid venetoclax dose-escalation protocol may be a safe way to quickly regain disease control, and possibly bridge to salvage therapies, investigators reported.
Of 15 patients with CLL who relapsed after treatment with a B-cell receptor inhibitor (BCRi), all were able to get to their target dose of venetoclax under close inpatient monitoring at a median of 12 days, compared with the 35 days usually required for venetoclax dose escalation, reported Farrukh T. Awan, MD, of Ohio State University Comprehensive Cancer Center in Columbus, and his colleagues.
Only two patients developed clinical tumor lysis syndrome (TLS), a common occurrence with venetoclax therapy, and this adverse event was manageable, Dr. Awan said at the annual congress of the European Hematology Association.
“The reason why we have been doing a slow ramp up on venetoclax is the original toxicity issues that we saw early on,” he said in an interview. “But unfortunately, a lot of patients are progressing on these new agents and have very rapid disease progression, and what we have seen is that if you stop the ibrutinib, the disease progresses very quickly, and by the time they can get up to the effective dose of venetoclax, they’re too sick to continue, or they might even die from disease progression.”
To combat this problem, Dr. Awan and his colleagues developed a rapid dose escalation protocol that would ramp up from 20 mg to 400 mg, with increases every 1 or 2 days depending on tolerability and incident TLS. Lab tests for TLS were evaluated every 4-8 hours.
All patients were closely monitored in the hospital, and all were started on allopurinol or other uric acid–lowering agents before starting on venetoclax.
The investigators reported safety and efficacy outcomes for the patients in a retrospective analysis.
The median age of the patients, 12 men and 3 women, was 65 years (range, 58-86 years). Seven patients had Eastern Cooperative Oncology Group Performance Status of 0, seven had an ECOG score of 1, and one had a score of 2-4.
Ten patients had most recently been treated with a BCRi, either a Bruton’s tyrosine kinase inhibitor (ibrutinib or acalabrutinib), idelalisib, or entospletinib. Three patients received ibrutinib plus chemotherapy, and two received rituximab and dexamethasone followed by rituximab maintenance.
The median time to full venetoclax dose was 12 days (range, 5-21 days) and all 15 patients reached the target dose. The mean length of stay during the ramp-up period was 9.5 days (range, 6-22 days).
The incidence of clinical TLS was 13.2%, occurring in two patients, one at the initial 20-mg dose, and one at the 200-mg dose level. Another five patients had asymptomatic TLS. Other treatment-related adverse events were anemia in seven patients, neutropenia in six patients, thrombocytopenia in five patients, and lung infection in one patient.
Twelve patients had a partial response, one had stable disease, and two had progressive disease. The mean time to best response was 71 days.
One-year progression-free survival was 49%, and 1-year overall survival was 68%.
The investigators found that for patients who still have some disease control with a BCRi, it may be possible to keep them on that drug while transitioning to venetoclax. The rapid dose escalation protocol should only be attempted in highly experience comprehensive cancer centers, Dr. Awan said.
“Under very close monitoring in an experienced inpatient setting, where the nurses are very used to doing this on a weekly basis in a very high volume center, I think that our data show that we could do this without affecting toxicity significantly or mortality,” he said.
Venetoclax therapy could buy enough time for patients to bridge to other options, such as chimeric antigen receptor (CAR) T-cell therapy or allogeneic stem cell transplant, he noted.
“But if we had waited 4 weeks, most of these patients would not have made it,” he said.
The study was internally funded. Dr. Awan reported research funding from Gilead, Pharmacyclics, AbbVie, and Janssen.
SOURCE: Koenig K et al. EHA Congress, Abstract PF357.
STOCKHOLM – Patients with chronic lymphocytic leukemia (CLL) who experience relapse after therapy with a B-cell receptor signaling inhibitor tend to have a swiftly progressive disease course that requires immediate intervention. For these patients, a rapid venetoclax dose-escalation protocol may be a safe way to quickly regain disease control, and possibly bridge to salvage therapies, investigators reported.
Of 15 patients with CLL who relapsed after treatment with a B-cell receptor inhibitor (BCRi), all were able to get to their target dose of venetoclax under close inpatient monitoring at a median of 12 days, compared with the 35 days usually required for venetoclax dose escalation, reported Farrukh T. Awan, MD, of Ohio State University Comprehensive Cancer Center in Columbus, and his colleagues.
Only two patients developed clinical tumor lysis syndrome (TLS), a common occurrence with venetoclax therapy, and this adverse event was manageable, Dr. Awan said at the annual congress of the European Hematology Association.
“The reason why we have been doing a slow ramp up on venetoclax is the original toxicity issues that we saw early on,” he said in an interview. “But unfortunately, a lot of patients are progressing on these new agents and have very rapid disease progression, and what we have seen is that if you stop the ibrutinib, the disease progresses very quickly, and by the time they can get up to the effective dose of venetoclax, they’re too sick to continue, or they might even die from disease progression.”
To combat this problem, Dr. Awan and his colleagues developed a rapid dose escalation protocol that would ramp up from 20 mg to 400 mg, with increases every 1 or 2 days depending on tolerability and incident TLS. Lab tests for TLS were evaluated every 4-8 hours.
All patients were closely monitored in the hospital, and all were started on allopurinol or other uric acid–lowering agents before starting on venetoclax.
The investigators reported safety and efficacy outcomes for the patients in a retrospective analysis.
The median age of the patients, 12 men and 3 women, was 65 years (range, 58-86 years). Seven patients had Eastern Cooperative Oncology Group Performance Status of 0, seven had an ECOG score of 1, and one had a score of 2-4.
Ten patients had most recently been treated with a BCRi, either a Bruton’s tyrosine kinase inhibitor (ibrutinib or acalabrutinib), idelalisib, or entospletinib. Three patients received ibrutinib plus chemotherapy, and two received rituximab and dexamethasone followed by rituximab maintenance.
The median time to full venetoclax dose was 12 days (range, 5-21 days) and all 15 patients reached the target dose. The mean length of stay during the ramp-up period was 9.5 days (range, 6-22 days).
The incidence of clinical TLS was 13.2%, occurring in two patients, one at the initial 20-mg dose, and one at the 200-mg dose level. Another five patients had asymptomatic TLS. Other treatment-related adverse events were anemia in seven patients, neutropenia in six patients, thrombocytopenia in five patients, and lung infection in one patient.
Twelve patients had a partial response, one had stable disease, and two had progressive disease. The mean time to best response was 71 days.
One-year progression-free survival was 49%, and 1-year overall survival was 68%.
The investigators found that for patients who still have some disease control with a BCRi, it may be possible to keep them on that drug while transitioning to venetoclax. The rapid dose escalation protocol should only be attempted in highly experience comprehensive cancer centers, Dr. Awan said.
“Under very close monitoring in an experienced inpatient setting, where the nurses are very used to doing this on a weekly basis in a very high volume center, I think that our data show that we could do this without affecting toxicity significantly or mortality,” he said.
Venetoclax therapy could buy enough time for patients to bridge to other options, such as chimeric antigen receptor (CAR) T-cell therapy or allogeneic stem cell transplant, he noted.
“But if we had waited 4 weeks, most of these patients would not have made it,” he said.
The study was internally funded. Dr. Awan reported research funding from Gilead, Pharmacyclics, AbbVie, and Janssen.
SOURCE: Koenig K et al. EHA Congress, Abstract PF357.
REPORTING FROM THE EHA CONGRESS
Key clinical point:
Major finding: All patients reached the target dose of venetoclax, with only two cases of manageable clinical tumor lysis syndrome.
Study details: Retrospective analysis of outcomes for 15 patients with CLL who relapsed after treatment with a B-cell receptor signaling inhibitor.
Disclosures: The study was internally funded. Dr. Awan reported research funding from Gilead, Pharmacyclics, AbbVie, and Janssen.
Source: Koenig K et al. EHA Congress, Abstract PF357.
European Medicines Agency recommends CAR T-cell approvals
The European Medicines Agency (EMA) has recommended a handful of hematology medications for approval, including two chimeric antigen receptor (CAR) T-cell therapies.
All of the drugs must next be approved by the European Commission in order to be marketed to patients throughout Europe.
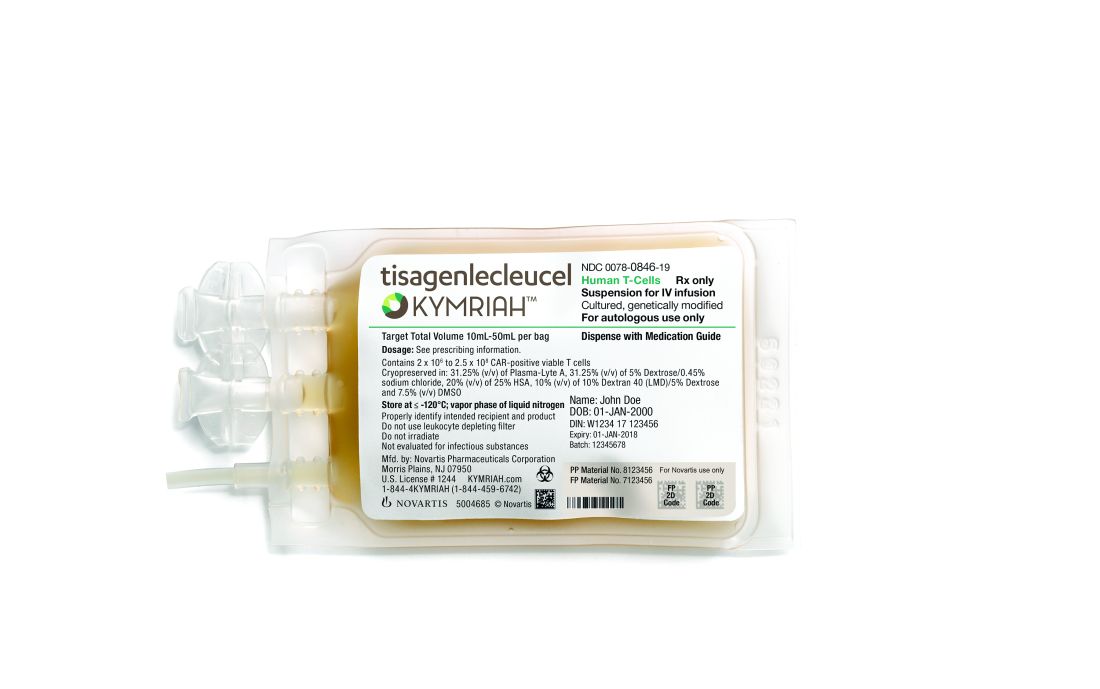
At the end of June, the EMA’s Committee for Medicinal Products for Human Use tisagenlecleucel (Kymriah) and axicabtagene ciloleucel (Yescarta).
The EMA approval recommendations come with risk management measures to address the potential for cytokine release syndrome with both of these treatments. Drug makers must use a patient registry to monitor the long-term safety and efficacy of the therapies.
The EMA is also recommending approval of caplacizumab for acquired thrombotic thrombocytopenic purpura, vonicog alfa for the treatment of von Willebrand disease, and daunorubicin/cytarabine for the treatment of acute myeloid leukemia.
The European Medicines Agency (EMA) has recommended a handful of hematology medications for approval, including two chimeric antigen receptor (CAR) T-cell therapies.
All of the drugs must next be approved by the European Commission in order to be marketed to patients throughout Europe.
At the end of June, the EMA’s Committee for Medicinal Products for Human Use tisagenlecleucel (Kymriah) and axicabtagene ciloleucel (Yescarta).
The EMA approval recommendations come with risk management measures to address the potential for cytokine release syndrome with both of these treatments. Drug makers must use a patient registry to monitor the long-term safety and efficacy of the therapies.
The EMA is also recommending approval of caplacizumab for acquired thrombotic thrombocytopenic purpura, vonicog alfa for the treatment of von Willebrand disease, and daunorubicin/cytarabine for the treatment of acute myeloid leukemia.
The European Medicines Agency (EMA) has recommended a handful of hematology medications for approval, including two chimeric antigen receptor (CAR) T-cell therapies.
All of the drugs must next be approved by the European Commission in order to be marketed to patients throughout Europe.
At the end of June, the EMA’s Committee for Medicinal Products for Human Use tisagenlecleucel (Kymriah) and axicabtagene ciloleucel (Yescarta).
The EMA approval recommendations come with risk management measures to address the potential for cytokine release syndrome with both of these treatments. Drug makers must use a patient registry to monitor the long-term safety and efficacy of the therapies.
The EMA is also recommending approval of caplacizumab for acquired thrombotic thrombocytopenic purpura, vonicog alfa for the treatment of von Willebrand disease, and daunorubicin/cytarabine for the treatment of acute myeloid leukemia.
Bortezomib plus vorinostat shows modest response in MCL
but was less impressive among patients with diffuse large B-cell lymphoma (DLBCL).
Victor Yazbeck, MD, of the Massey Cancer Center at Virginia Commonwealth University in Richmond, and his colleagues reported the findings from the multicenter, nonrandomized, phase 2 trial with 65 treated patients. The trial included three cohorts: 22 patients with MCL and no prior treatment with bortezomib; 4 patients with MCL and prior treatment with bortezomib; and 39 patients with relapsed or refractory DLBCL and no prior bortezomib.
The best results were seen among MCL patients with no prior bortezomib treatment, with an overall response rate of 31.8% and a median progression-free survival (PFS) of 7.6 months. Responses were limited among the DLBCL cohort, which had an overall response rate of 7.7% and a median PFS of just 1.8 months. Among MCL patients who had received prior bortezomib treatment, there were no responses.
From a safety perspective, the combination treatment was well tolerated. The most common grade 3 and 4 hematologic toxicities were thrombocytopenia, lymphopenia, and neutropenia. There was one death among the DLBCL patients and it was unclear if it was related to treatment or progression of disease.
“Patients with MCL had a higher [overall response rate] compared to those with DLBCL, most likely due to the single-agent activity of bortezomib in MCL,” the researchers wrote. “Overall, the synergism previously demonstrated in preclinical models could not be confirmed.”
The study was supported by the Southeast Phase 2 Consortium and by a grant from the National Cancer Institute. Dr. Yazbeck reported having no financial disclosures. One of his coauthors is an employee of Amgen and owns Amgen stock. Another coauthor receives research support from Takeda, Celgene, Karyopharm Therapeutics, Bristol-Myers Squibb, Merck, and Signal Genetics.
SOURCE: Yazbeck V et al. Clin Lymphoma Myeloma Leuk. 2018 Jun 6. doi: 10.1016/j.clml.2018.05.023.
but was less impressive among patients with diffuse large B-cell lymphoma (DLBCL).
Victor Yazbeck, MD, of the Massey Cancer Center at Virginia Commonwealth University in Richmond, and his colleagues reported the findings from the multicenter, nonrandomized, phase 2 trial with 65 treated patients. The trial included three cohorts: 22 patients with MCL and no prior treatment with bortezomib; 4 patients with MCL and prior treatment with bortezomib; and 39 patients with relapsed or refractory DLBCL and no prior bortezomib.
The best results were seen among MCL patients with no prior bortezomib treatment, with an overall response rate of 31.8% and a median progression-free survival (PFS) of 7.6 months. Responses were limited among the DLBCL cohort, which had an overall response rate of 7.7% and a median PFS of just 1.8 months. Among MCL patients who had received prior bortezomib treatment, there were no responses.
From a safety perspective, the combination treatment was well tolerated. The most common grade 3 and 4 hematologic toxicities were thrombocytopenia, lymphopenia, and neutropenia. There was one death among the DLBCL patients and it was unclear if it was related to treatment or progression of disease.
“Patients with MCL had a higher [overall response rate] compared to those with DLBCL, most likely due to the single-agent activity of bortezomib in MCL,” the researchers wrote. “Overall, the synergism previously demonstrated in preclinical models could not be confirmed.”
The study was supported by the Southeast Phase 2 Consortium and by a grant from the National Cancer Institute. Dr. Yazbeck reported having no financial disclosures. One of his coauthors is an employee of Amgen and owns Amgen stock. Another coauthor receives research support from Takeda, Celgene, Karyopharm Therapeutics, Bristol-Myers Squibb, Merck, and Signal Genetics.
SOURCE: Yazbeck V et al. Clin Lymphoma Myeloma Leuk. 2018 Jun 6. doi: 10.1016/j.clml.2018.05.023.
but was less impressive among patients with diffuse large B-cell lymphoma (DLBCL).
Victor Yazbeck, MD, of the Massey Cancer Center at Virginia Commonwealth University in Richmond, and his colleagues reported the findings from the multicenter, nonrandomized, phase 2 trial with 65 treated patients. The trial included three cohorts: 22 patients with MCL and no prior treatment with bortezomib; 4 patients with MCL and prior treatment with bortezomib; and 39 patients with relapsed or refractory DLBCL and no prior bortezomib.
The best results were seen among MCL patients with no prior bortezomib treatment, with an overall response rate of 31.8% and a median progression-free survival (PFS) of 7.6 months. Responses were limited among the DLBCL cohort, which had an overall response rate of 7.7% and a median PFS of just 1.8 months. Among MCL patients who had received prior bortezomib treatment, there were no responses.
From a safety perspective, the combination treatment was well tolerated. The most common grade 3 and 4 hematologic toxicities were thrombocytopenia, lymphopenia, and neutropenia. There was one death among the DLBCL patients and it was unclear if it was related to treatment or progression of disease.
“Patients with MCL had a higher [overall response rate] compared to those with DLBCL, most likely due to the single-agent activity of bortezomib in MCL,” the researchers wrote. “Overall, the synergism previously demonstrated in preclinical models could not be confirmed.”
The study was supported by the Southeast Phase 2 Consortium and by a grant from the National Cancer Institute. Dr. Yazbeck reported having no financial disclosures. One of his coauthors is an employee of Amgen and owns Amgen stock. Another coauthor receives research support from Takeda, Celgene, Karyopharm Therapeutics, Bristol-Myers Squibb, Merck, and Signal Genetics.
SOURCE: Yazbeck V et al. Clin Lymphoma Myeloma Leuk. 2018 Jun 6. doi: 10.1016/j.clml.2018.05.023.
FROM CLINICAL LYMPHOMA, MYELOMA AND LEUKEMIA
CAR T in DLBCL: Liso-cel has ‘remarkable’ efficacy in cohort
CHICAGO – The CD19–directed chimeric antigen receptor (CAR) T-cell product lisocabtagene maraleucel (liso-cel, JCAR017) produced durable responses in poor-prognosis patients with relapsed/refractory diffuse large B-cell lymphoma (DLBCL), follow-up results of a phase 1 trial show.
Nearly 90% of DLBCL patients who achieved complete response as their best response on liso-cel were alive at 1 year in the study, according to investigator Jeremy S. Abramson, MD, of Massachusetts General Hospital Cancer Center in Boston.
That result is “far superior to what we would have anticipated with conventional therapies in a largely chemorefractory DLBCL population,” Dr. Abramson said at the annual meeting of the American Society of Clinical Oncology.
The new data on liso-cel come on the heels of a second approval of a CAR T-cell therapy for DLBCL, noted Caron Jacobson, MD, of Dana-Farber Cancer Institute, Boston.
Axicabtagene ciloleucel (Yescarta) was approved in October 2017 by the U.S. Food and Drug Administration for relapsed or refractory large B-cell lymphomas, including DLBCL. In May 2018, tisagenlecleucel (Kymriah) received its second FDA approval to treat relapsed or refractory large B-cell lymphomas, including DLBCL.
CAR T-cell therapy “really has transformed outcomes for a group of patients who previously had no other standard of care and who… have a relatively short overall survival,” Dr. Jacobson said.
At the meeting, Dr. Abramson presented findings on DLBCL patients in TRANSCEND NHL 001, a phase 1, multicenter, open-label study of the CD-19 targeted CAR T-cell therapy in relapsed and refractory B-cell non-Hodgkin lymphoma.
About 90% of treated DLBCL patients had one or more poor-risk disease features, such as ECOG performance status 2 and primary refractory disease, which predict poor overall survival, according to Dr. Abramson.
Dr. Abramson’s presentation focused on 102 evaluable DLBCL patients in the dose-finding and dose-expansion cohorts of the TRANSCEND study, including a subset analysis of a core group of 73 patients who met the criteria for pivotal dose cohort of the study (1 x 108 cells given as a single dose).
For the full set of 102 DLBCL patients, the best overall response rate was 75%, including a best complete remission rate of 55%, according to presented data. In the core group of 73 DLBCL patients, best overall response and complete remission rates were 80% and 59%, respectively.
Investigators saw “encouraging” durable response rates at 6 months and beyond in the core DLBCL population, according to Dr. Abramson. Of patients with a complete remission at 3 months, 88% remained in complete remission at the 6-month follow-up, and 93% of those in remission at the 6-month time point were in ongoing response at a median follow-up of 8 months.
Median overall survival had not been reached in either the full or core DLBCL cohorts with a median of 12 months follow-up, he added, noting that 90% of patients who achieved complete remission as their best response remained alive at 1 year.
In terms of adverse effects, liso-cel is showing a low and manageable toxicity profile, with very low rates of severe cytokine release syndrome (CRS) and neurotoxicity at 1% and 13%, respectively, Dr. Abramson reported.
“This anti-CD19 CAR T cell has remarkable efficacy in a group of highly refractory aggressive B-cell non-Hodgkin lymphoma patients,” said Dr. Jacobson, commenting on results of the DLBCL subset.
Based on the data presented, liso-cel is “clearly competitive” with the approved CAR T-cell therapies, though she advised caution in comparing across studies. “I don’t think that there will be a randomized study of all three agents, but I do think that we’ll start to get comparative data from single institution experiences that are using all three products,” she said.
The pivotal DLBCL cohort of TRANSCEND NHL 001 has completed accrual and results will be presented at a future meeting, Dr. Abramson said.
Dr. Abramson reported disclosures related to Celgene, Genentech/Roche, Gilead Sciences, Novartis, Seattle Genetics, and Millennium.
SOURCE: Abramson JS et al. ASCO 2018. Abstract 7505.
CHICAGO – The CD19–directed chimeric antigen receptor (CAR) T-cell product lisocabtagene maraleucel (liso-cel, JCAR017) produced durable responses in poor-prognosis patients with relapsed/refractory diffuse large B-cell lymphoma (DLBCL), follow-up results of a phase 1 trial show.
Nearly 90% of DLBCL patients who achieved complete response as their best response on liso-cel were alive at 1 year in the study, according to investigator Jeremy S. Abramson, MD, of Massachusetts General Hospital Cancer Center in Boston.
That result is “far superior to what we would have anticipated with conventional therapies in a largely chemorefractory DLBCL population,” Dr. Abramson said at the annual meeting of the American Society of Clinical Oncology.
The new data on liso-cel come on the heels of a second approval of a CAR T-cell therapy for DLBCL, noted Caron Jacobson, MD, of Dana-Farber Cancer Institute, Boston.
Axicabtagene ciloleucel (Yescarta) was approved in October 2017 by the U.S. Food and Drug Administration for relapsed or refractory large B-cell lymphomas, including DLBCL. In May 2018, tisagenlecleucel (Kymriah) received its second FDA approval to treat relapsed or refractory large B-cell lymphomas, including DLBCL.
CAR T-cell therapy “really has transformed outcomes for a group of patients who previously had no other standard of care and who… have a relatively short overall survival,” Dr. Jacobson said.
At the meeting, Dr. Abramson presented findings on DLBCL patients in TRANSCEND NHL 001, a phase 1, multicenter, open-label study of the CD-19 targeted CAR T-cell therapy in relapsed and refractory B-cell non-Hodgkin lymphoma.
About 90% of treated DLBCL patients had one or more poor-risk disease features, such as ECOG performance status 2 and primary refractory disease, which predict poor overall survival, according to Dr. Abramson.
Dr. Abramson’s presentation focused on 102 evaluable DLBCL patients in the dose-finding and dose-expansion cohorts of the TRANSCEND study, including a subset analysis of a core group of 73 patients who met the criteria for pivotal dose cohort of the study (1 x 108 cells given as a single dose).
For the full set of 102 DLBCL patients, the best overall response rate was 75%, including a best complete remission rate of 55%, according to presented data. In the core group of 73 DLBCL patients, best overall response and complete remission rates were 80% and 59%, respectively.
Investigators saw “encouraging” durable response rates at 6 months and beyond in the core DLBCL population, according to Dr. Abramson. Of patients with a complete remission at 3 months, 88% remained in complete remission at the 6-month follow-up, and 93% of those in remission at the 6-month time point were in ongoing response at a median follow-up of 8 months.
Median overall survival had not been reached in either the full or core DLBCL cohorts with a median of 12 months follow-up, he added, noting that 90% of patients who achieved complete remission as their best response remained alive at 1 year.
In terms of adverse effects, liso-cel is showing a low and manageable toxicity profile, with very low rates of severe cytokine release syndrome (CRS) and neurotoxicity at 1% and 13%, respectively, Dr. Abramson reported.
“This anti-CD19 CAR T cell has remarkable efficacy in a group of highly refractory aggressive B-cell non-Hodgkin lymphoma patients,” said Dr. Jacobson, commenting on results of the DLBCL subset.
Based on the data presented, liso-cel is “clearly competitive” with the approved CAR T-cell therapies, though she advised caution in comparing across studies. “I don’t think that there will be a randomized study of all three agents, but I do think that we’ll start to get comparative data from single institution experiences that are using all three products,” she said.
The pivotal DLBCL cohort of TRANSCEND NHL 001 has completed accrual and results will be presented at a future meeting, Dr. Abramson said.
Dr. Abramson reported disclosures related to Celgene, Genentech/Roche, Gilead Sciences, Novartis, Seattle Genetics, and Millennium.
SOURCE: Abramson JS et al. ASCO 2018. Abstract 7505.
CHICAGO – The CD19–directed chimeric antigen receptor (CAR) T-cell product lisocabtagene maraleucel (liso-cel, JCAR017) produced durable responses in poor-prognosis patients with relapsed/refractory diffuse large B-cell lymphoma (DLBCL), follow-up results of a phase 1 trial show.
Nearly 90% of DLBCL patients who achieved complete response as their best response on liso-cel were alive at 1 year in the study, according to investigator Jeremy S. Abramson, MD, of Massachusetts General Hospital Cancer Center in Boston.
That result is “far superior to what we would have anticipated with conventional therapies in a largely chemorefractory DLBCL population,” Dr. Abramson said at the annual meeting of the American Society of Clinical Oncology.
The new data on liso-cel come on the heels of a second approval of a CAR T-cell therapy for DLBCL, noted Caron Jacobson, MD, of Dana-Farber Cancer Institute, Boston.
Axicabtagene ciloleucel (Yescarta) was approved in October 2017 by the U.S. Food and Drug Administration for relapsed or refractory large B-cell lymphomas, including DLBCL. In May 2018, tisagenlecleucel (Kymriah) received its second FDA approval to treat relapsed or refractory large B-cell lymphomas, including DLBCL.
CAR T-cell therapy “really has transformed outcomes for a group of patients who previously had no other standard of care and who… have a relatively short overall survival,” Dr. Jacobson said.
At the meeting, Dr. Abramson presented findings on DLBCL patients in TRANSCEND NHL 001, a phase 1, multicenter, open-label study of the CD-19 targeted CAR T-cell therapy in relapsed and refractory B-cell non-Hodgkin lymphoma.
About 90% of treated DLBCL patients had one or more poor-risk disease features, such as ECOG performance status 2 and primary refractory disease, which predict poor overall survival, according to Dr. Abramson.
Dr. Abramson’s presentation focused on 102 evaluable DLBCL patients in the dose-finding and dose-expansion cohorts of the TRANSCEND study, including a subset analysis of a core group of 73 patients who met the criteria for pivotal dose cohort of the study (1 x 108 cells given as a single dose).
For the full set of 102 DLBCL patients, the best overall response rate was 75%, including a best complete remission rate of 55%, according to presented data. In the core group of 73 DLBCL patients, best overall response and complete remission rates were 80% and 59%, respectively.
Investigators saw “encouraging” durable response rates at 6 months and beyond in the core DLBCL population, according to Dr. Abramson. Of patients with a complete remission at 3 months, 88% remained in complete remission at the 6-month follow-up, and 93% of those in remission at the 6-month time point were in ongoing response at a median follow-up of 8 months.
Median overall survival had not been reached in either the full or core DLBCL cohorts with a median of 12 months follow-up, he added, noting that 90% of patients who achieved complete remission as their best response remained alive at 1 year.
In terms of adverse effects, liso-cel is showing a low and manageable toxicity profile, with very low rates of severe cytokine release syndrome (CRS) and neurotoxicity at 1% and 13%, respectively, Dr. Abramson reported.
“This anti-CD19 CAR T cell has remarkable efficacy in a group of highly refractory aggressive B-cell non-Hodgkin lymphoma patients,” said Dr. Jacobson, commenting on results of the DLBCL subset.
Based on the data presented, liso-cel is “clearly competitive” with the approved CAR T-cell therapies, though she advised caution in comparing across studies. “I don’t think that there will be a randomized study of all three agents, but I do think that we’ll start to get comparative data from single institution experiences that are using all three products,” she said.
The pivotal DLBCL cohort of TRANSCEND NHL 001 has completed accrual and results will be presented at a future meeting, Dr. Abramson said.
Dr. Abramson reported disclosures related to Celgene, Genentech/Roche, Gilead Sciences, Novartis, Seattle Genetics, and Millennium.
SOURCE: Abramson JS et al. ASCO 2018. Abstract 7505.
REPORTING FROM ASCO 2018
Key clinical point: (DLBCL).
Major finding: Among DLBCL patients treated with the pivotal dose of liso-cel, 88% who were in complete remission at 3 months remained in complete remission at the 6 month follow-up.
Study details: Follow-up report on a cohort of DLBCL patients from TRANSCEND NHL 001, a phase 1 trial of liso-cel in relapsed and refractory B-cell NHL.
Disclosures: Dr. Abramson reported disclosures related to Celgene, Genentech/Roche, Gilead Sciences, Novartis, Seattle Genetics, and Millennium.
Source: Abramson JS et al. ASCO 2018. Abstract 7505.
Combo treatment under review for Waldenstrom macroglobulinemia
by the Food and Drug Administration.
Ibrutinib, a Bruton’s tyrosine kinase inhibitor, is already approved as a single agent for WM. The addition of rituximab to the indication is based on positive results from the phase 3 INNOVATE study. In particular, the trial showed a superior progression-free survival rate at 30 months for the ibrutinib-rituximab combination at 82%, compared with placebo plus rituximab at 28% (N Engl J Med. 2018;378:2399-410).
The study’s lead investigator, Meletios A. Dimopoulos, MD, called the combination a “new standard of care” for WM at the recent annual meeting of the American Society of Clinical Oncology.
Ibrutinib, marketed as Imbruvica, is jointly developed and commercialized by Pharmacyclics and Janssen Biotech.
by the Food and Drug Administration.
Ibrutinib, a Bruton’s tyrosine kinase inhibitor, is already approved as a single agent for WM. The addition of rituximab to the indication is based on positive results from the phase 3 INNOVATE study. In particular, the trial showed a superior progression-free survival rate at 30 months for the ibrutinib-rituximab combination at 82%, compared with placebo plus rituximab at 28% (N Engl J Med. 2018;378:2399-410).
The study’s lead investigator, Meletios A. Dimopoulos, MD, called the combination a “new standard of care” for WM at the recent annual meeting of the American Society of Clinical Oncology.
Ibrutinib, marketed as Imbruvica, is jointly developed and commercialized by Pharmacyclics and Janssen Biotech.
by the Food and Drug Administration.
Ibrutinib, a Bruton’s tyrosine kinase inhibitor, is already approved as a single agent for WM. The addition of rituximab to the indication is based on positive results from the phase 3 INNOVATE study. In particular, the trial showed a superior progression-free survival rate at 30 months for the ibrutinib-rituximab combination at 82%, compared with placebo plus rituximab at 28% (N Engl J Med. 2018;378:2399-410).
The study’s lead investigator, Meletios A. Dimopoulos, MD, called the combination a “new standard of care” for WM at the recent annual meeting of the American Society of Clinical Oncology.
Ibrutinib, marketed as Imbruvica, is jointly developed and commercialized by Pharmacyclics and Janssen Biotech.
Is CLL chemoimmunotherapy dead? Not yet
CHICAGO – Chemoimmunotherapy for chronic lymphocytic leukemia is on the way out, but there’s one scenario where it still plays a key role, according to one leukemia expert.
That scenario is not in relapsed or refractory chronic lymphocytic leukemia (CLL), where the use of fludarabine, cyclophosphamide, and rituximab (FCR) may be hard to justify today. Data supporting use of FCR in relapsed CLL show a median progression-free survival (PFS) of about 21 months, Susan M. O’Brien, MD, of the University of California, Irvine, said at the annual meeting of the American Society of Clinical Oncology. There is also data for bendamustine-rituximab retreatment showing a median event-free survival of about 15 months, she added.
By contrast, the 5-year follow-up data for the Bruton tyrosine kinase inhibitor ibrutinib in the relapsed/refractory setting shows a median PFS of 52 months, which is “extraordinary,” given that the patients had a median of four prior regimens, Dr. O’Brien said.
Similarly, recently published results from the randomized, phase 3 MURANO study of venetoclax plus rituximab in relapsed/refractory CLL showed that median PFS was not reached at a median follow-up of 23.8 months, versus a median of 17 months for the bendamustine-rituximab comparison arm (N Engl J Med. 2018;378[12]:1107-20).
“Thanks to the MURANO study, we likely will have an expanded label for venetoclax that includes the combination of venetoclax and rituximab,” Dr. O’Brien said. “I think it’s quite clear that either of these is dramatically better than what you get with retreatment with chemotherapy, so I personally don’t think there is a role for chemoimmunotherapy in the relapsed patient.”
On June 8, 2018, the Food and Drug Administration granted regular approval for venetoclax for patients with CLL or small lymphocytic lymphoma, with or without 17p deletion, who have received at least one prior therapy. The FDA also approved its use in combination with rituximab.*
But frontline CLL treatment is currently a little bit more complicated, Dr. O’Brien said.
Recent studies show favorable long-term outcomes with FCR frontline therapy in the immunoglobulin heavy chain variable gene (IgHV) –mutated subgroup of patients, she noted.
The longest follow-up comes from a study from investigators at the University of Texas MD Anderson Cancer Center, Houston, published in 2016. In that study, the 12.8-year PFS was 53.9% for IgHV-mutated patients, versus just 8.7% for patients with unmutated IgHV. Of the IgHV-mutated group, more than half achieved minimal residual disease (MRD) negativity after treatment (Blood. 2016 Jan 21; 127[3]: 303-9).
“I’m going to go out on a limb and I’m going to suggest that I think there is a cure fraction here,” Dr. O’Brien said. “On the other hand, if there’s not a cure fraction and they’re going to relapse after 17 years, that’s a pretty attractive endpoint, even if it’s not a cure fraction.”
Clinical practice guidelines now recognize IgHV mutation status as an important marker that should be obtained when deciding on treatment, Dr. O’Brien noted.
For unmutated patients, the RESONATE-2 trial showed that ibrutinib was superior to chlorambucil in older patients, many of whom had comorbid conditions. In the 3-year update, median PFS was approximately 15 months for chlorambucil, while for ibrutinib the median PFS was “nowhere near” being reached, Dr. O’Brien said.
Those data may not be so relevant for fit, unmutated patients, and two randomized trials comparing FCR with bendamustine and rituximab have yet to report data. However, one recent cross-trial comparison found fairly overlapping survival curves for the two chemoimmunotherapy approaches.
Dr. O’Brien said she would put older patients with comorbidities on ibrutinib if a clinical trial was not available, and for fit, unmutated patients, while more data are needed, she would also use ibrutinib. However, patient preference sometimes tips the scale in favor of FCR.
“The discussions sometimes are quite long about whether the patient should opt to take ibrutinib or FCR,” Dr. O’Brien said. “The last patient I had that discussion with elected to take FCR. When I asked him why, he said because he liked the idea of being finished in six cycles, off all therapy, and hopefully in remission.”
While Dr. O’Brien said she views chemoimmunotherapy as still relevant in IgHV-mutated patients, eventually it will go away, she concluded. Toward that end, there is considerable interest in venetoclax plus ibrutinib, a combination that, in early reports, has yielded very encouraging MRD results in first-line CLL.
“We have no long-term data, but very, very exciting MRD negativity data,” Dr. O’Brien said.
Dr. O’Brien reported relationships with Abbvie, Amgen, Celgene, Gilead Sciences, Janssen, Pfizer, Pharmacyclics, Sunesis Pharmaceuticals, and others.
*This story was updated 6/25/2018.
CHICAGO – Chemoimmunotherapy for chronic lymphocytic leukemia is on the way out, but there’s one scenario where it still plays a key role, according to one leukemia expert.
That scenario is not in relapsed or refractory chronic lymphocytic leukemia (CLL), where the use of fludarabine, cyclophosphamide, and rituximab (FCR) may be hard to justify today. Data supporting use of FCR in relapsed CLL show a median progression-free survival (PFS) of about 21 months, Susan M. O’Brien, MD, of the University of California, Irvine, said at the annual meeting of the American Society of Clinical Oncology. There is also data for bendamustine-rituximab retreatment showing a median event-free survival of about 15 months, she added.
By contrast, the 5-year follow-up data for the Bruton tyrosine kinase inhibitor ibrutinib in the relapsed/refractory setting shows a median PFS of 52 months, which is “extraordinary,” given that the patients had a median of four prior regimens, Dr. O’Brien said.
Similarly, recently published results from the randomized, phase 3 MURANO study of venetoclax plus rituximab in relapsed/refractory CLL showed that median PFS was not reached at a median follow-up of 23.8 months, versus a median of 17 months for the bendamustine-rituximab comparison arm (N Engl J Med. 2018;378[12]:1107-20).
“Thanks to the MURANO study, we likely will have an expanded label for venetoclax that includes the combination of venetoclax and rituximab,” Dr. O’Brien said. “I think it’s quite clear that either of these is dramatically better than what you get with retreatment with chemotherapy, so I personally don’t think there is a role for chemoimmunotherapy in the relapsed patient.”
On June 8, 2018, the Food and Drug Administration granted regular approval for venetoclax for patients with CLL or small lymphocytic lymphoma, with or without 17p deletion, who have received at least one prior therapy. The FDA also approved its use in combination with rituximab.*
But frontline CLL treatment is currently a little bit more complicated, Dr. O’Brien said.
Recent studies show favorable long-term outcomes with FCR frontline therapy in the immunoglobulin heavy chain variable gene (IgHV) –mutated subgroup of patients, she noted.
The longest follow-up comes from a study from investigators at the University of Texas MD Anderson Cancer Center, Houston, published in 2016. In that study, the 12.8-year PFS was 53.9% for IgHV-mutated patients, versus just 8.7% for patients with unmutated IgHV. Of the IgHV-mutated group, more than half achieved minimal residual disease (MRD) negativity after treatment (Blood. 2016 Jan 21; 127[3]: 303-9).
“I’m going to go out on a limb and I’m going to suggest that I think there is a cure fraction here,” Dr. O’Brien said. “On the other hand, if there’s not a cure fraction and they’re going to relapse after 17 years, that’s a pretty attractive endpoint, even if it’s not a cure fraction.”
Clinical practice guidelines now recognize IgHV mutation status as an important marker that should be obtained when deciding on treatment, Dr. O’Brien noted.
For unmutated patients, the RESONATE-2 trial showed that ibrutinib was superior to chlorambucil in older patients, many of whom had comorbid conditions. In the 3-year update, median PFS was approximately 15 months for chlorambucil, while for ibrutinib the median PFS was “nowhere near” being reached, Dr. O’Brien said.
Those data may not be so relevant for fit, unmutated patients, and two randomized trials comparing FCR with bendamustine and rituximab have yet to report data. However, one recent cross-trial comparison found fairly overlapping survival curves for the two chemoimmunotherapy approaches.
Dr. O’Brien said she would put older patients with comorbidities on ibrutinib if a clinical trial was not available, and for fit, unmutated patients, while more data are needed, she would also use ibrutinib. However, patient preference sometimes tips the scale in favor of FCR.
“The discussions sometimes are quite long about whether the patient should opt to take ibrutinib or FCR,” Dr. O’Brien said. “The last patient I had that discussion with elected to take FCR. When I asked him why, he said because he liked the idea of being finished in six cycles, off all therapy, and hopefully in remission.”
While Dr. O’Brien said she views chemoimmunotherapy as still relevant in IgHV-mutated patients, eventually it will go away, she concluded. Toward that end, there is considerable interest in venetoclax plus ibrutinib, a combination that, in early reports, has yielded very encouraging MRD results in first-line CLL.
“We have no long-term data, but very, very exciting MRD negativity data,” Dr. O’Brien said.
Dr. O’Brien reported relationships with Abbvie, Amgen, Celgene, Gilead Sciences, Janssen, Pfizer, Pharmacyclics, Sunesis Pharmaceuticals, and others.
*This story was updated 6/25/2018.
CHICAGO – Chemoimmunotherapy for chronic lymphocytic leukemia is on the way out, but there’s one scenario where it still plays a key role, according to one leukemia expert.
That scenario is not in relapsed or refractory chronic lymphocytic leukemia (CLL), where the use of fludarabine, cyclophosphamide, and rituximab (FCR) may be hard to justify today. Data supporting use of FCR in relapsed CLL show a median progression-free survival (PFS) of about 21 months, Susan M. O’Brien, MD, of the University of California, Irvine, said at the annual meeting of the American Society of Clinical Oncology. There is also data for bendamustine-rituximab retreatment showing a median event-free survival of about 15 months, she added.
By contrast, the 5-year follow-up data for the Bruton tyrosine kinase inhibitor ibrutinib in the relapsed/refractory setting shows a median PFS of 52 months, which is “extraordinary,” given that the patients had a median of four prior regimens, Dr. O’Brien said.
Similarly, recently published results from the randomized, phase 3 MURANO study of venetoclax plus rituximab in relapsed/refractory CLL showed that median PFS was not reached at a median follow-up of 23.8 months, versus a median of 17 months for the bendamustine-rituximab comparison arm (N Engl J Med. 2018;378[12]:1107-20).
“Thanks to the MURANO study, we likely will have an expanded label for venetoclax that includes the combination of venetoclax and rituximab,” Dr. O’Brien said. “I think it’s quite clear that either of these is dramatically better than what you get with retreatment with chemotherapy, so I personally don’t think there is a role for chemoimmunotherapy in the relapsed patient.”
On June 8, 2018, the Food and Drug Administration granted regular approval for venetoclax for patients with CLL or small lymphocytic lymphoma, with or without 17p deletion, who have received at least one prior therapy. The FDA also approved its use in combination with rituximab.*
But frontline CLL treatment is currently a little bit more complicated, Dr. O’Brien said.
Recent studies show favorable long-term outcomes with FCR frontline therapy in the immunoglobulin heavy chain variable gene (IgHV) –mutated subgroup of patients, she noted.
The longest follow-up comes from a study from investigators at the University of Texas MD Anderson Cancer Center, Houston, published in 2016. In that study, the 12.8-year PFS was 53.9% for IgHV-mutated patients, versus just 8.7% for patients with unmutated IgHV. Of the IgHV-mutated group, more than half achieved minimal residual disease (MRD) negativity after treatment (Blood. 2016 Jan 21; 127[3]: 303-9).
“I’m going to go out on a limb and I’m going to suggest that I think there is a cure fraction here,” Dr. O’Brien said. “On the other hand, if there’s not a cure fraction and they’re going to relapse after 17 years, that’s a pretty attractive endpoint, even if it’s not a cure fraction.”
Clinical practice guidelines now recognize IgHV mutation status as an important marker that should be obtained when deciding on treatment, Dr. O’Brien noted.
For unmutated patients, the RESONATE-2 trial showed that ibrutinib was superior to chlorambucil in older patients, many of whom had comorbid conditions. In the 3-year update, median PFS was approximately 15 months for chlorambucil, while for ibrutinib the median PFS was “nowhere near” being reached, Dr. O’Brien said.
Those data may not be so relevant for fit, unmutated patients, and two randomized trials comparing FCR with bendamustine and rituximab have yet to report data. However, one recent cross-trial comparison found fairly overlapping survival curves for the two chemoimmunotherapy approaches.
Dr. O’Brien said she would put older patients with comorbidities on ibrutinib if a clinical trial was not available, and for fit, unmutated patients, while more data are needed, she would also use ibrutinib. However, patient preference sometimes tips the scale in favor of FCR.
“The discussions sometimes are quite long about whether the patient should opt to take ibrutinib or FCR,” Dr. O’Brien said. “The last patient I had that discussion with elected to take FCR. When I asked him why, he said because he liked the idea of being finished in six cycles, off all therapy, and hopefully in remission.”
While Dr. O’Brien said she views chemoimmunotherapy as still relevant in IgHV-mutated patients, eventually it will go away, she concluded. Toward that end, there is considerable interest in venetoclax plus ibrutinib, a combination that, in early reports, has yielded very encouraging MRD results in first-line CLL.
“We have no long-term data, but very, very exciting MRD negativity data,” Dr. O’Brien said.
Dr. O’Brien reported relationships with Abbvie, Amgen, Celgene, Gilead Sciences, Janssen, Pfizer, Pharmacyclics, Sunesis Pharmaceuticals, and others.
*This story was updated 6/25/2018.
EXPERT ANALYSIS FROM ASCO 2018
DLBCL survival improved with novel antibody-drug conjugate
STOCKHOLM, SWEDEN – Adding an experimental antibody-drug conjugate to bendamustine and rituximab more than doubled overall survival over bendamustine/rituximab alone in patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL), investigators reported.
Among 80 transplant-ineligible patients with relapsed or refractory DLBCL in a phase 2 trial, the combination of the antibody-drug conjugate (ADC) polatuzumab vedotin plus bendamustine/rituximab (BR) was associated with a 40% complete response rate, compared with 15% for BR alone.
More importantly, the ADC was associated with 6.7 months median progression-free survival (PFS) versus 2 months for BR, and 11.8 months median overall survival (OS), versus 4.7 months for BR alone, reported Laurie Sehn, MD, from BC Cancer in Vancouver, British Columbia, Canada.
“This randomized phase 2 trial really is, so far, the only head-to-head comparison of a novel targeted agent against a standard therapy in this patient population that’s ineligible for transplant, and it demonstrated that the combination polatuzumab vedotin with BR significantly improved the response rates and progression-free survival, as well as overall survival,” she said at the annual congress of the European Hematology Association.
However, in a separate cohort of patients with follicular lymphoma in the same trial, there was no difference in either PFS or OS during follow-up to date, Dr. Sehn reported.
Polatuzumab vedotin consists of an antibody targeted against CD79b, an antigenic protein expressed on the surface of normal B cells, as well as DLBCL and follicular lymphoma cells.
Dr. Sehn and her colleagues enrolled 80 patients with DLBCL for whom first-line chemoimmunotherapy had failed and who were ineligible for stem cell transplant due to age and/or comorbidities.
A second cohort included 80 patients with follicular lymphoma. In this group, median PFS with polatuzumab vedotin/BR was 17 months versus 17.3 months for BR alone, and median overall survival had not been reached in either arm at the time of the data cutoff.
In the DLBCL cohort, patients were randomized to receive polatuzumab vedotin 1.8 mg/kg plus bendamustine 90mg/m2 for 2 days and rituximab 375mg/m2) or BR alone for six 21-day cycles.
The complete response rate by PET scan – the primary endpoint – was significantly higher with polatuzmab/BR at 40% versus 15% for BR alone (P = .012). Respective overall response rates were 45% versus 18% (P = .008). Also, median PFS with the polatuzmab/BR therapy was 6.7 months versus 2.0 months for BR alone, translating into a hazard ratio of 0.31 (P less than .0001).
Respective median overall survival was 11.8 versus 4.7 months, translating into a hazard ratio for the polatuzmab/BR combination of 0.35 (P = .0008).
The PET complete response rates were higher with polatuzmab/BR regardless of prior lines of therapy or refractory status, Dr. Sehn noted.
“In terms of the safety, I think importantly in the combination there were no unexpected toxicities, so typical to what we would expect with what’s known with this drug alone,” Dr. Sehn said.
Grade 3 or greater toxicities that were higher with the polatuzmab/BR combination included cytopenias, febrile neutropenia, and infections. The single serious adverse event that had a higher incidence in the polatuzumab/BR arm was febrile neutropenia (DLBCL). In total, 12% of patients in the polatuzumab-containing arm and 11% of patients in the BR-only arm died on study. Many of the deaths were due to disease progression.
Anton Hagenbeek, MD, PhD, from the Academic Medical Center at the University of Amsterdam, the Netherlands, who moderated the briefing but was not involved in the study, said that about 20%-30% of patients with relapsed/refractory DLBCL are positive for the CD33 antigen, the target of brentuximab vedotin (Adcetris), and noted that this agent is also being tested in a phase 2 trial.
Martin Hutchings, MD, PhD, from Rigshospitalet in Copenhagen, the Netherlands, who co-moderated the oral abstract session, commented that “it’s not so often that we see significant overall survival differences in a phase 2 study with 80 patients.”
Based on the results of this trial, polatuzumab has been granted breakthrough therapy designation by the U.S. Food and Drug Administration and a PRIME (priority medicine) designation from the European Medicines Agency.
SOURCE: Sehn LH et al. EHA Congress, Abstract S802.
STOCKHOLM, SWEDEN – Adding an experimental antibody-drug conjugate to bendamustine and rituximab more than doubled overall survival over bendamustine/rituximab alone in patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL), investigators reported.
Among 80 transplant-ineligible patients with relapsed or refractory DLBCL in a phase 2 trial, the combination of the antibody-drug conjugate (ADC) polatuzumab vedotin plus bendamustine/rituximab (BR) was associated with a 40% complete response rate, compared with 15% for BR alone.
More importantly, the ADC was associated with 6.7 months median progression-free survival (PFS) versus 2 months for BR, and 11.8 months median overall survival (OS), versus 4.7 months for BR alone, reported Laurie Sehn, MD, from BC Cancer in Vancouver, British Columbia, Canada.
“This randomized phase 2 trial really is, so far, the only head-to-head comparison of a novel targeted agent against a standard therapy in this patient population that’s ineligible for transplant, and it demonstrated that the combination polatuzumab vedotin with BR significantly improved the response rates and progression-free survival, as well as overall survival,” she said at the annual congress of the European Hematology Association.
However, in a separate cohort of patients with follicular lymphoma in the same trial, there was no difference in either PFS or OS during follow-up to date, Dr. Sehn reported.
Polatuzumab vedotin consists of an antibody targeted against CD79b, an antigenic protein expressed on the surface of normal B cells, as well as DLBCL and follicular lymphoma cells.
Dr. Sehn and her colleagues enrolled 80 patients with DLBCL for whom first-line chemoimmunotherapy had failed and who were ineligible for stem cell transplant due to age and/or comorbidities.
A second cohort included 80 patients with follicular lymphoma. In this group, median PFS with polatuzumab vedotin/BR was 17 months versus 17.3 months for BR alone, and median overall survival had not been reached in either arm at the time of the data cutoff.
In the DLBCL cohort, patients were randomized to receive polatuzumab vedotin 1.8 mg/kg plus bendamustine 90mg/m2 for 2 days and rituximab 375mg/m2) or BR alone for six 21-day cycles.
The complete response rate by PET scan – the primary endpoint – was significantly higher with polatuzmab/BR at 40% versus 15% for BR alone (P = .012). Respective overall response rates were 45% versus 18% (P = .008). Also, median PFS with the polatuzmab/BR therapy was 6.7 months versus 2.0 months for BR alone, translating into a hazard ratio of 0.31 (P less than .0001).
Respective median overall survival was 11.8 versus 4.7 months, translating into a hazard ratio for the polatuzmab/BR combination of 0.35 (P = .0008).
The PET complete response rates were higher with polatuzmab/BR regardless of prior lines of therapy or refractory status, Dr. Sehn noted.
“In terms of the safety, I think importantly in the combination there were no unexpected toxicities, so typical to what we would expect with what’s known with this drug alone,” Dr. Sehn said.
Grade 3 or greater toxicities that were higher with the polatuzmab/BR combination included cytopenias, febrile neutropenia, and infections. The single serious adverse event that had a higher incidence in the polatuzumab/BR arm was febrile neutropenia (DLBCL). In total, 12% of patients in the polatuzumab-containing arm and 11% of patients in the BR-only arm died on study. Many of the deaths were due to disease progression.
Anton Hagenbeek, MD, PhD, from the Academic Medical Center at the University of Amsterdam, the Netherlands, who moderated the briefing but was not involved in the study, said that about 20%-30% of patients with relapsed/refractory DLBCL are positive for the CD33 antigen, the target of brentuximab vedotin (Adcetris), and noted that this agent is also being tested in a phase 2 trial.
Martin Hutchings, MD, PhD, from Rigshospitalet in Copenhagen, the Netherlands, who co-moderated the oral abstract session, commented that “it’s not so often that we see significant overall survival differences in a phase 2 study with 80 patients.”
Based on the results of this trial, polatuzumab has been granted breakthrough therapy designation by the U.S. Food and Drug Administration and a PRIME (priority medicine) designation from the European Medicines Agency.
SOURCE: Sehn LH et al. EHA Congress, Abstract S802.
STOCKHOLM, SWEDEN – Adding an experimental antibody-drug conjugate to bendamustine and rituximab more than doubled overall survival over bendamustine/rituximab alone in patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL), investigators reported.
Among 80 transplant-ineligible patients with relapsed or refractory DLBCL in a phase 2 trial, the combination of the antibody-drug conjugate (ADC) polatuzumab vedotin plus bendamustine/rituximab (BR) was associated with a 40% complete response rate, compared with 15% for BR alone.
More importantly, the ADC was associated with 6.7 months median progression-free survival (PFS) versus 2 months for BR, and 11.8 months median overall survival (OS), versus 4.7 months for BR alone, reported Laurie Sehn, MD, from BC Cancer in Vancouver, British Columbia, Canada.
“This randomized phase 2 trial really is, so far, the only head-to-head comparison of a novel targeted agent against a standard therapy in this patient population that’s ineligible for transplant, and it demonstrated that the combination polatuzumab vedotin with BR significantly improved the response rates and progression-free survival, as well as overall survival,” she said at the annual congress of the European Hematology Association.
However, in a separate cohort of patients with follicular lymphoma in the same trial, there was no difference in either PFS or OS during follow-up to date, Dr. Sehn reported.
Polatuzumab vedotin consists of an antibody targeted against CD79b, an antigenic protein expressed on the surface of normal B cells, as well as DLBCL and follicular lymphoma cells.
Dr. Sehn and her colleagues enrolled 80 patients with DLBCL for whom first-line chemoimmunotherapy had failed and who were ineligible for stem cell transplant due to age and/or comorbidities.
A second cohort included 80 patients with follicular lymphoma. In this group, median PFS with polatuzumab vedotin/BR was 17 months versus 17.3 months for BR alone, and median overall survival had not been reached in either arm at the time of the data cutoff.
In the DLBCL cohort, patients were randomized to receive polatuzumab vedotin 1.8 mg/kg plus bendamustine 90mg/m2 for 2 days and rituximab 375mg/m2) or BR alone for six 21-day cycles.
The complete response rate by PET scan – the primary endpoint – was significantly higher with polatuzmab/BR at 40% versus 15% for BR alone (P = .012). Respective overall response rates were 45% versus 18% (P = .008). Also, median PFS with the polatuzmab/BR therapy was 6.7 months versus 2.0 months for BR alone, translating into a hazard ratio of 0.31 (P less than .0001).
Respective median overall survival was 11.8 versus 4.7 months, translating into a hazard ratio for the polatuzmab/BR combination of 0.35 (P = .0008).
The PET complete response rates were higher with polatuzmab/BR regardless of prior lines of therapy or refractory status, Dr. Sehn noted.
“In terms of the safety, I think importantly in the combination there were no unexpected toxicities, so typical to what we would expect with what’s known with this drug alone,” Dr. Sehn said.
Grade 3 or greater toxicities that were higher with the polatuzmab/BR combination included cytopenias, febrile neutropenia, and infections. The single serious adverse event that had a higher incidence in the polatuzumab/BR arm was febrile neutropenia (DLBCL). In total, 12% of patients in the polatuzumab-containing arm and 11% of patients in the BR-only arm died on study. Many of the deaths were due to disease progression.
Anton Hagenbeek, MD, PhD, from the Academic Medical Center at the University of Amsterdam, the Netherlands, who moderated the briefing but was not involved in the study, said that about 20%-30% of patients with relapsed/refractory DLBCL are positive for the CD33 antigen, the target of brentuximab vedotin (Adcetris), and noted that this agent is also being tested in a phase 2 trial.
Martin Hutchings, MD, PhD, from Rigshospitalet in Copenhagen, the Netherlands, who co-moderated the oral abstract session, commented that “it’s not so often that we see significant overall survival differences in a phase 2 study with 80 patients.”
Based on the results of this trial, polatuzumab has been granted breakthrough therapy designation by the U.S. Food and Drug Administration and a PRIME (priority medicine) designation from the European Medicines Agency.
SOURCE: Sehn LH et al. EHA Congress, Abstract S802.
REPORTING FROM THE EHA CONGRESS
Key clinical point:
Major finding: The complete response rate with polatuzumab vedotin plus bendamustine/rituximab (BR) was 40%, compared with 15% for BR alone.
Study details: Randomized controlled phase 2 trial in 80 patients with relapsed/refractory DLBCL.
Disclosures: The study was funded by Hoffman-La Roche. Dr. Sehn reported ties to Roche/Genentech and others.
Source: Sehn LH et al. EHA Congress, Abstract S802.
British good practice paper offers MCL diagnosis pearls
Immunohistochemical panels used in the diagnosis of mantle cell lymphoma should include cyclin D1 and SOX11 immunostaining, according to a good practice paper from the British Society of Haematology.
Pamela McKay, MD, of the Beatson West of Scotland Cancer Centre, Glasgow, and her colleagues provided based on a review of literature from 1980 to 2017. The good practice paper aims to offer best practice advice based on consensus where the evidence is limited. Specifically, the paper incorporates new information on molecular pathology and the use of positron emission tomography/computed tomography (PET/CT) scanning in staging of disease.
The top recommendations related to MCL diagnosis include performing lymph node excision or adequate core biopsy for diagnosis of nodal MCL. For non-nodal presentation, a tissue biopsy or peripheral blood can be used. Additionally, immunohistochemical panels should include cyclin D1 and SOX11 immunostaining.
In cases of atypical morphology, aberrant immunophenotype, equivocal cyclin D1 positivity, or unusual clinical presentation, the authors recommended fluorescence in situ hybridization (FISH) to demonstrate the presence of the t(11;14) translocation. They also recommended recording the Ki67 Proliferation Index at baseline, with an index of greater than 30% being indicative of a poorer outcome.
In terms of staging disease, Dr. McKay and her associates recommended that patients undergo staging with CT of the neck, chest, abdomen, and pelvis. They recommended against routine use of fluorodeoxyglucose PET for MCL staging, but said it could be considered if radical radiotherapy is being proposed for early-stage disease.
For cases with suspicion of central nervous system involvement, lumbar puncture with cytospin and immunophenotyping is recommended.
They recommended that all MCL patients have either their simplified or combined MCL international prognostic index score recorded at baseline.
All the authors made a declaration of interest to the British Society of Haematology and task force chairs, which may be viewed on request.
SOURCE: McKay P et al. Br J Haematol. 2018 Jun 8. doi: 10.1111/bjh.15281.
Immunohistochemical panels used in the diagnosis of mantle cell lymphoma should include cyclin D1 and SOX11 immunostaining, according to a good practice paper from the British Society of Haematology.
Pamela McKay, MD, of the Beatson West of Scotland Cancer Centre, Glasgow, and her colleagues provided based on a review of literature from 1980 to 2017. The good practice paper aims to offer best practice advice based on consensus where the evidence is limited. Specifically, the paper incorporates new information on molecular pathology and the use of positron emission tomography/computed tomography (PET/CT) scanning in staging of disease.
The top recommendations related to MCL diagnosis include performing lymph node excision or adequate core biopsy for diagnosis of nodal MCL. For non-nodal presentation, a tissue biopsy or peripheral blood can be used. Additionally, immunohistochemical panels should include cyclin D1 and SOX11 immunostaining.
In cases of atypical morphology, aberrant immunophenotype, equivocal cyclin D1 positivity, or unusual clinical presentation, the authors recommended fluorescence in situ hybridization (FISH) to demonstrate the presence of the t(11;14) translocation. They also recommended recording the Ki67 Proliferation Index at baseline, with an index of greater than 30% being indicative of a poorer outcome.
In terms of staging disease, Dr. McKay and her associates recommended that patients undergo staging with CT of the neck, chest, abdomen, and pelvis. They recommended against routine use of fluorodeoxyglucose PET for MCL staging, but said it could be considered if radical radiotherapy is being proposed for early-stage disease.
For cases with suspicion of central nervous system involvement, lumbar puncture with cytospin and immunophenotyping is recommended.
They recommended that all MCL patients have either their simplified or combined MCL international prognostic index score recorded at baseline.
All the authors made a declaration of interest to the British Society of Haematology and task force chairs, which may be viewed on request.
SOURCE: McKay P et al. Br J Haematol. 2018 Jun 8. doi: 10.1111/bjh.15281.
Immunohistochemical panels used in the diagnosis of mantle cell lymphoma should include cyclin D1 and SOX11 immunostaining, according to a good practice paper from the British Society of Haematology.
Pamela McKay, MD, of the Beatson West of Scotland Cancer Centre, Glasgow, and her colleagues provided based on a review of literature from 1980 to 2017. The good practice paper aims to offer best practice advice based on consensus where the evidence is limited. Specifically, the paper incorporates new information on molecular pathology and the use of positron emission tomography/computed tomography (PET/CT) scanning in staging of disease.
The top recommendations related to MCL diagnosis include performing lymph node excision or adequate core biopsy for diagnosis of nodal MCL. For non-nodal presentation, a tissue biopsy or peripheral blood can be used. Additionally, immunohistochemical panels should include cyclin D1 and SOX11 immunostaining.
In cases of atypical morphology, aberrant immunophenotype, equivocal cyclin D1 positivity, or unusual clinical presentation, the authors recommended fluorescence in situ hybridization (FISH) to demonstrate the presence of the t(11;14) translocation. They also recommended recording the Ki67 Proliferation Index at baseline, with an index of greater than 30% being indicative of a poorer outcome.
In terms of staging disease, Dr. McKay and her associates recommended that patients undergo staging with CT of the neck, chest, abdomen, and pelvis. They recommended against routine use of fluorodeoxyglucose PET for MCL staging, but said it could be considered if radical radiotherapy is being proposed for early-stage disease.
For cases with suspicion of central nervous system involvement, lumbar puncture with cytospin and immunophenotyping is recommended.
They recommended that all MCL patients have either their simplified or combined MCL international prognostic index score recorded at baseline.
All the authors made a declaration of interest to the British Society of Haematology and task force chairs, which may be viewed on request.
SOURCE: McKay P et al. Br J Haematol. 2018 Jun 8. doi: 10.1111/bjh.15281.
FROM THE BRITISH JOURNAL OF HAEMATOLOGY