User login
AVAHO
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]


Optimizing Care for Veterans at Risk of Cancer From Camp Lejeune Water Exposure
Optimizing Care for Veterans at Risk of Cancer From Camp Lejeune Water Exposure
Clinical awareness of cancers associated with Camp Lejeune water contamination exposure remains limited despite legal and policy advances. Gaps persist in early symptom recognition and timely diagnostic evaluation before a definitive cancer diagnosis among exposed personnel. This may represent missed opportunities for earlier identification of volatile organic compounds (VOCs)-related cancers and for less invasive treatment options for veterans in this high-risk population.
Federal health care practitioners (HCPs), especially those in primary care and internal medicine, are uniquely positioned to bridge this gap. By improving the recognition of symptoms, pertinent physical examination findings, and implementing a diagnostic screening panel, HCPs can support accurate diagnoses and facilitate earlier treatment to improve health and quality of life for this population.
From 1953 to 1985, as many as 1 million military personnel, civilian workers, and their families stationed at US Marine Corps Base Camp Lejeune were unknowingly exposed to toxic and carcinogenic chemicals in drinking and bathing water.1 Three of the 8 main water sources on base were contaminated with VOCs, which are associated with multiple cancers.1-3
The US Department of Veterans Affairs (VA) recognizes 15 conditions associated with Camp Lejeune contaminated water exposure for VA benefits, including 10 cancers: adult leukemia; aplastic anemia and other myelodysplastic syndromes (MDS); bladder, esophageal, kidney, liver, breast (male and female), and lung cancers; multiple myeloma; and non-Hodgkin lymphoma (NHL).4
BACKGROUND
Established in 1942, Camp Lejeune is an important Marine Corps training installation. Between 1953 and 1985, multiple on-base water systems were contaminated with VOCs, including trichloroethylene (TCE), perchloroethylene (PCE), benzene, and vinyl chloride, due to improper waste disposal and industrial runoff from on- and off-base sources.5 Tarawa Terrace water treatment plant (WTP) was contaminated primarily with PCE from November 1957 to February 1987. Hadnot Point WTP was contaminated with TCE from August 1953 to December 1984, along with PCE, and benzene, toluene, ethylbenzene, and xylene (BTEX). Holcomb Boulevard WTP, established in 1972, was contaminated with TCE from June 1972 to February 1985.2 These contaminants entered the drinking and bathing water supply over decades, and exposure often occurred concurrently across = 1 VOC, compounding health risks.2,3 This prolonged 32-year VOC exposure window underlies current concerns regarding long-term cancer risk among affected service members, civilian employees, and family members. Epidemiologic research has found statistically significant associations between VOC exposure and multiple cancers, neurologic conditions, and reproductive issues.6 Specifically, TCE is associated with higher risks of hematologic cancers, multiple myeloma, NHL, and kidney cancer.3 PCE is linked with kidney cancer, benzene with multiple myeloma and NHL, and vinyl chloride with hepatobiliary cancers.3 A cohort mortality study compared Camp Lejeune personnel with a control group at Camp Pendleton from 1972 to 1985 and found a 3-fold higher incidence or mortality rate for kidney, esophageal, and female breast cancers, leukemia, and lymphoma among exposed Camp Lejeune personnel.6 Notably, personnel assigned to Camp Lejeune for as little as 6 months faced up to a 6-fold increase in cancer risk; the average military assignment between 1975 and 1985 was 18 months.3,6
Honoring America's Veterans and Caring for Camp Lejeune Families Act of 2012, the Sergeant First Class Heath Robinson Honoring Our Promise to Address Comprehensive Toxics (PACT) Act of 2022, the Camp Lejeune Justice Act of 2022, and the pending Ensuring Justice for Camp Lejeune Victims Act of 2025 provide health care and legal resources for personnel and families affected by Camp Lejeune’s contaminated water.6-8 These laws acknowledge associations between exposure and specific health conditions and expanded health care, benefits, and legal recourse for affected veterans, survivors, and their families.8,9
CANCERS LINKED TO CAMP LEJEUNE
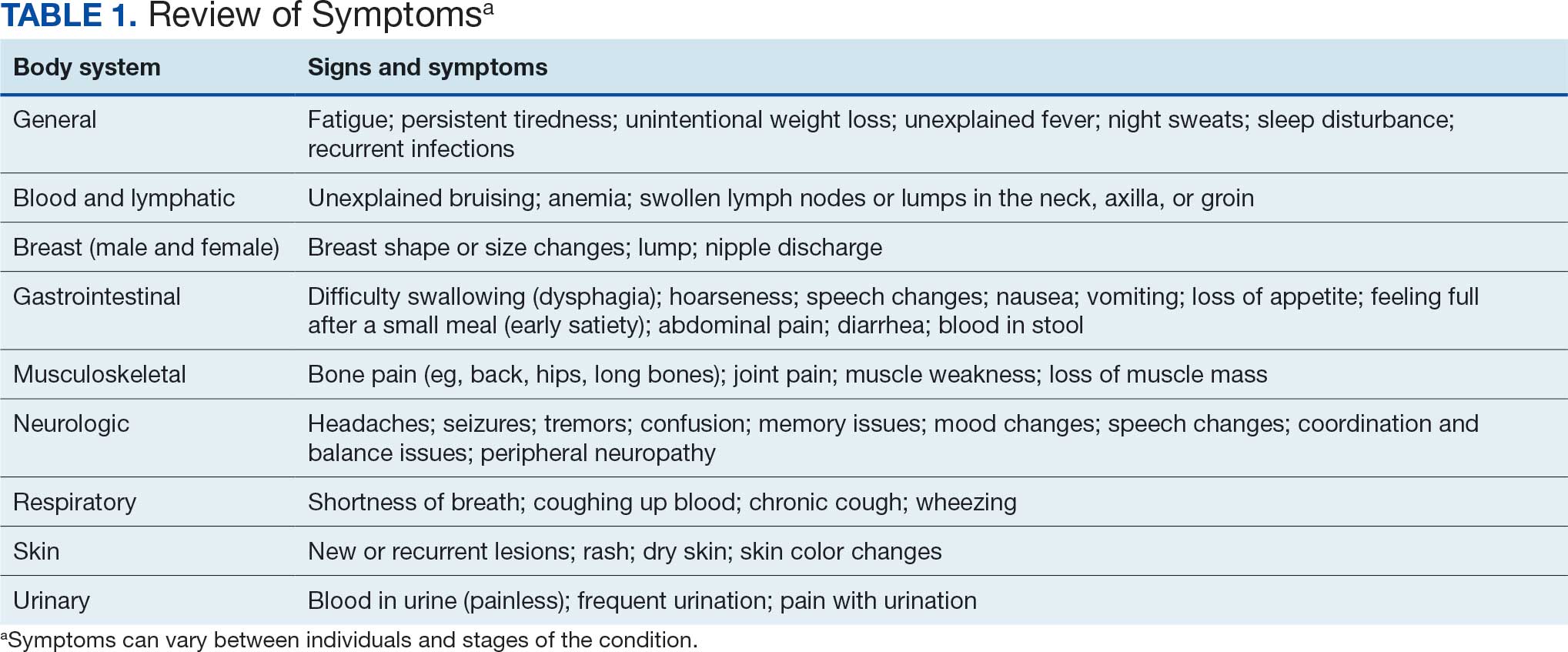
Camp Lejeune VOC-contaminated water exposure is associated with solid tumor and hematologic cancers. Symptoms, physical examination findings, and diagnostic considerations vary by cancer type (Table 1).
Bladder Cancer
The US incidence rate of bladder cancer for both males and females is 18 per 100,000 individuals per year, with a death rate of 4.1 per 100,000 individuals per year, and a 2.1% lifetime diagnosis risk.10 Personnel exposed to VOCs at Camp Lejeune had a 9% higher risk of developing bladder cancer and a 2% increased mortality compared with an unexposed control group at Camp Pendleton.1,7 Other bladder cancer subtypes at increased risk are papillary transitional cell carcinoma, nonpapillary transition cell carcinoma, and urothelial carcinoma.7 This is consistent with prior research that found PCE exposure is associated with an increased risk for bladder cancer.3,7,11 Smoking and tobacco use remain significant risk factors for bladder cancer.12
Symptomatology. The most common symptom associated with bladder cancer is painless hematuria (gross or microscopic). Other often delayed symptoms include urinary frequency, urgency, or nocturia.13,14
Diagnostics. Screening tests include urinalysis for hematuria, urine cytology, and cystoscopy with biopsy as the gold standard for diagnosis and staging.15,16
Kidney Cancer
The US incidence rate of kidney cancer and renal pelvis cancer for both males and females is 17.5 per 100,000 individuals per year, with a death rate of 3.4 per 100,000, and a 1.8% lifetime diagnosis risk.17 Camp Lejeune personnel exposed to VOCs had a 6% increased risk of developing kidney cancer and renal pelvis cancer and a 21% higher mortality risk compared with Camp Pendleton controls.1,7 Subtypes at risk include renal cell carcinoma and papillary carcinoma.7 This is consistent with prior research that found exposures to TCE and PCE are associated with a 3-fold increased risk of kidney cancer.3,7
Symptomatology. Hematuria, flank pain, and a palpable abdominal mass are common symptoms associated with kidney cancer. In advanced stages, other symptoms may include left-sided varicocele, anemia, weight loss, fatigue, fever, and night sweats.18
Diagnostics. Screening tests include urinalysis to assess the presence of blood, complete blood count (CBC) to assess anemia, calcium (elevated), and lactate dehydrogenase (LDH), which may be elevated. Imaging strategies include abdominal computed tomography (CT), magnetic resonance imaging (MRI), or ultrasound.19
Esophageal Cancer
The US incidence rate of esophageal cancer for both males and females is 4.2 per 100,000 individuals per year, the death rate is 3.7 per 100,000 individuals per year, and a 0.5% lifetime diagnosis risk.20 VOC-exposed Camp Lejeune personnel had a 27% increased incidence and 25% increased mortality compared with the control group.1,7 Esophageal cancer subtypes at elevated risk include squamous cell carcinoma and adenocarcinoma. This is consistent with prior research that found Camp Lejeune water exposure is associated with a 3-fold increased risk for esophageal cancer.7 Additional risk factors include history of smoking and alcohol use.21
Symptomatology. Esophageal cancer is often asymptomatic with potential symptoms that include dysphagia, hoarseness, and weight loss in advanced disease.22
Diagnostics. Endoscopy with biopsy is the definitive method for diagnosis.23
Liver Cancer
The US incidence rate of liver cancer and intrahepatic bile duct cancer for both males and females is 9.4 per 100,000 individuals per year, with a death rate of 6.6 per 100,000 individuals per year, and a 1.1% lifetime diagnosis risk.24 VOC-exposed personnel had a 1% higher mortality than controls.1
Symptomatology. Liver cancer is often asymptomatic and appears in late stages.25 Common symptoms include right upper quadrant pain, early satiety, nausea, vomiting, loss of appetite, weight loss, ascites, jaundice, and abnormal bleeding or bruising.25,26
Diagnostics. Diagnostic tests may include an ultrasound, CT, or MRI. Additional laboratory testing may include liver function, a-fetoprotein blood, CBC, renal function, calcium, and hepatitis panel screening for hepatitis B and C.27,28
Lung Cancer
The US incidence rate of lung cancer for both males and females is 47.8 per 100,000 individuals per year, with a death rate of 31.5 per 100,000 individuals per year, and a 5.4% lifetime diagnosis risk.29 VOC-exposed personnel had a 16% increased risk and 19% higher mortality.1,7 Subtypes include large cell, small cell, non-small cell, squamous cell, and adenocarcinoma.7 Smoking is an additional risk factor.30
Symptomatology. Symptoms of lung cancer include cough, shortness of breath, chest pain worse with deep breathing, unexplained weight loss, fatigue, night sweats, and recurrent fevers. Advanced stages may metastasize or spread to the liver, bones, and brain.31
Diagnostics. Low-dose CT and chest X-ray are used for screening.32
Breast Cancer
The US incidence rate of female breast cancer is 130.8 per 100,000 individuals per year, with a death rate of 19.2 per 100,000 individuals per year, and a 13.0% lifetime risk of diagnosis.33 For female VOC-exposed personnel, there was an equal risk of developing breast cancer as the control group.1 However, exposed females at Camp Lejeune had a 23% higher mortality risk compared to the control group.7 Breast cancer subtypes among females include ductal carcinoma, lobular carcinoma, and ductal-lobular carcinoma.1
The US incidence rate of male breast cancer is 1.3 per 100,000 individuals per year, with a death rate of 0.3 per 100,000 individuals per year.34,35 The lifetime risk for males developing breast cancer is 137.7 per 100,000 and about 70 to 100 times less common in men than women.36
Male personnel exposed at Camp Lejeune had a 4% increased risk for developing breast cancer compared to Camp Pendleton.7 However, mortality was lower in the Camp Lejeune group.1 Although male breast cancer is rare, males at Camp Lejeune had a higher incidence, indicating a link between TCE, PCE, vinyl chloride exposures and male breast cancer.37 Male breast cancer is more often diagnosed in advanced stages than female breast cancer due to the lack of awareness or absence of routine screenings.38 The most common breast cancer type in males is invasive ductal carcinoma, accounting for 85% to 90% of cases; lobular carcinoma is the second most common type.39
Symptomatology. In both females and males, breast cancer symptoms include painless, firm mass or lump in the breast (left breast slightly more common than right), skin changes or dimpling, nipple retraction or turning inward, and nipple discharge. Breast cancer can spread to the lymph nodes and can be appreciated in axilla or clavicular regions.40
Diagnostics. The diagnostic evaluation for breast cancer is similar for females and males. It includes a clinical breast examination, diagnostic mammogram, and ultrasound.41 Mammograms can distinguish between gynecomastia and cancer, especially in males.42 A core or fine needle biopsy is needed to confirm diagnosis.41
Adult Leukemia
The US incidence rate of leukemia for both male and female was 14.4 per 100,000 individuals per year, with a death rate of 5.8 per 100,000 individuals per year, and a 1.5% lifetime diagnosis risk.43
VOC-exposed personnel had a 7% higher risk of developing leukemia and a 13% increased mortality risk compared with the control group.1,7 Subtypes of leukemia at risk included a 38% increased incidence of acute myeloid/monocytic leukemia (AML) and a 2% increased incidence of chronic lymphocytic leukemia (CLL).1 Benzene and TCE exposures are known risk factors for AML and other leukemias.7 Personnel at Camp Lejeune had 3 times the incidence or mortality for leukemia, specifically AML mortality at 20%.7 Smoking is an additional risk factor for certain leukemias, especially AML.30
Symptomatology. Symptoms associated with leukemia are often nonspecific and may include fatigue, pallor, easy bruising or bleeding (skin or gums), recurrent infections secondary to neutropenia, fever, night sweats, pain or feeling full after a small meal due to enlarged spleen or liver, and weight loss.44,45
Diagnostics. An initial screening includes a CBC with differential, a peripheral smear to detect the presence of blast cells, as well as Auer rods in myeloid blast cells in AML or smudge cells in CLL. Confirmatory tests may include bone marrow biopsy or flow cytometry. A referral to a hematologist is recommended for any suspected leukemia.46,47
Myelodysplastic Syndromes
Aplastic anemia and MDS are considered rare disorders.48 Aplastic anemia is a nonmalignant bone marrow failure disorder with pancytopenia and hypocellular bone marrow due to the loss of hematopoietic stem cells.48 MDS is a type of hematopoietic cancer where the bone marrow produces abnormal blood cells or does not make enough healthy cells.49 This can lead to an increased risk for infection, cytopenias, neutropenia, refractory anemia, and thrombocytopenia, and progression to AML in some patients.49
The reported US incidence of MDS from 1975 to 2013 was 6.7 per 100,000 for males and 3.7 per 100,000 for females.50 Benzene exposure is linked to MDS and a known cause of AML.1 VOC-exposed personnel had a 68% increased risk of developing MDS and a 2.3-fold increased mortality risk compared to controls.1,7
Symptomatology. Some patients are asymptomatic at diagnosis.51 Symptoms related to cytopenia include fatigue, pallor, purpura, petechiae, bleeding of skin, gum, or nose, recurrent infections, fever, bone pain, loss of appetite, and weight loss.50,51
Diagnostics. Initial workup includes a CBC with differential to assess for anemia, white blood cell and absolute neutrophil counts (low), and thrombocytopenia.52 A peripheral blood smear may show myeloid blast cells. A bone marrow aspiration and biopsy, flow cytometry, and cytogenetic or molecular testing may be performed. If MDS is suspected, a referral to a hematologist should be considered.52
Multiple Myeloma
The US incidence rate of multiple myeloma for both males and females is 7.3 per 100,000 individuals per year, with a mortality rate of 2.9 per 100,000 individuals per year, and a 0.8% lifetime diagnosis risk.53 VOC-exposed personnel had a 13% increased risk of developing multiple myeloma and an 8% increased mortality risk compared to unexposed personnel.1,7
Symptomatology. Multiple myeloma may be asymptomatic in early stages. The most common presenting symptom is bone pain, especially in the back, hips, and long bones, due to hypercalcemia from increased reabsorption, plasma cell tumor overgrowth in the bone marrow, and lytic lesions.54 Additional symptoms include fatigue and pallor related to anemia, leukopenia, thrombocytopenia, recurrent infections, extreme thirst, frequent urination, dehydration, confusion associated with hypercalcemia, peripheral neuropathy, loss of appetite, weight loss, and renal impairment or failure.54
Diagnostics. Testing considerations include a CBC with a peripheral blood smear to evaluate anemia and rouleaux formation of red blood cells (seen in > 50% of patients with multiple myeloma), comprehensive metabolic panel (CMP) to assess kidney function, calcium levels (elevated), serum and urine protein electrophoresis with immunofixation to detect monoclonal protein (detected in > 80% of patients with multiple myeloma) and Bence-Jones proteins, serum free light chain assay, and a bone marrow biopsy for diagnosis.55,56
MRI of the spine and pelvis is the most sensitive to detecting bone marrow involvement and focal lesions before lytic lesion progression occurs and for assessing spinal cord compression.57 PET/CT is more sensitive at detecting extramedullary disease, outside of the spine, and for patients that cannot undergo MRI.57 A whole-body low-dose CT, either alone or with PET, is more sensitive than an X-ray at detecting lytic lesions, fractures, or osteoporosis associated with multiple myeloma.57
Non-Hodgkin Lymphoma
The US incidence rate of NHL for both males and females are 18.7 per 100,000 individuals per year, the death rate is 4.9 per 100,000 individuals per year, and a 2% lifetime diagnosis risk.58 VOC-exposed personnel had a 1% higher risk of developing NHL and a decreased mortality risk compared to the control group.1,7 Specific NHL subtypes with increased risk in the exposed cohort are mantle cell (26%), follicular (7%), Burkitt (53%), and marginal zone B-cell (45%).7
Symptomatology. NHL often presents with painless lymphadenopathy or enlarged lymph nodes involving the cervical, axillary, inguinal regions.59,60 Other symptoms include frequent infections, unexplained bruising, weight loss, and “B symptoms,” such as fever and night sweats.59,60 Some patients develop a mediastinal mass in the thorax, which if large may lead to cough or shortness of breath.59
Diagnostics. The initial diagnostic workup includes CBC with differential and LDH, which may be elevated.60,61 Imaging may begin with a chest X-ray to assess for a mediastinal mass; however, CTs of the chest, abdomen, and pelvis provide more detail to better assess for NHL. Whole body PET/CT is considered the gold standard for assessing and staging systemic involvement. If enlarged lymph nodes are present, a biopsy can confirm the subtype of NHL.60,61
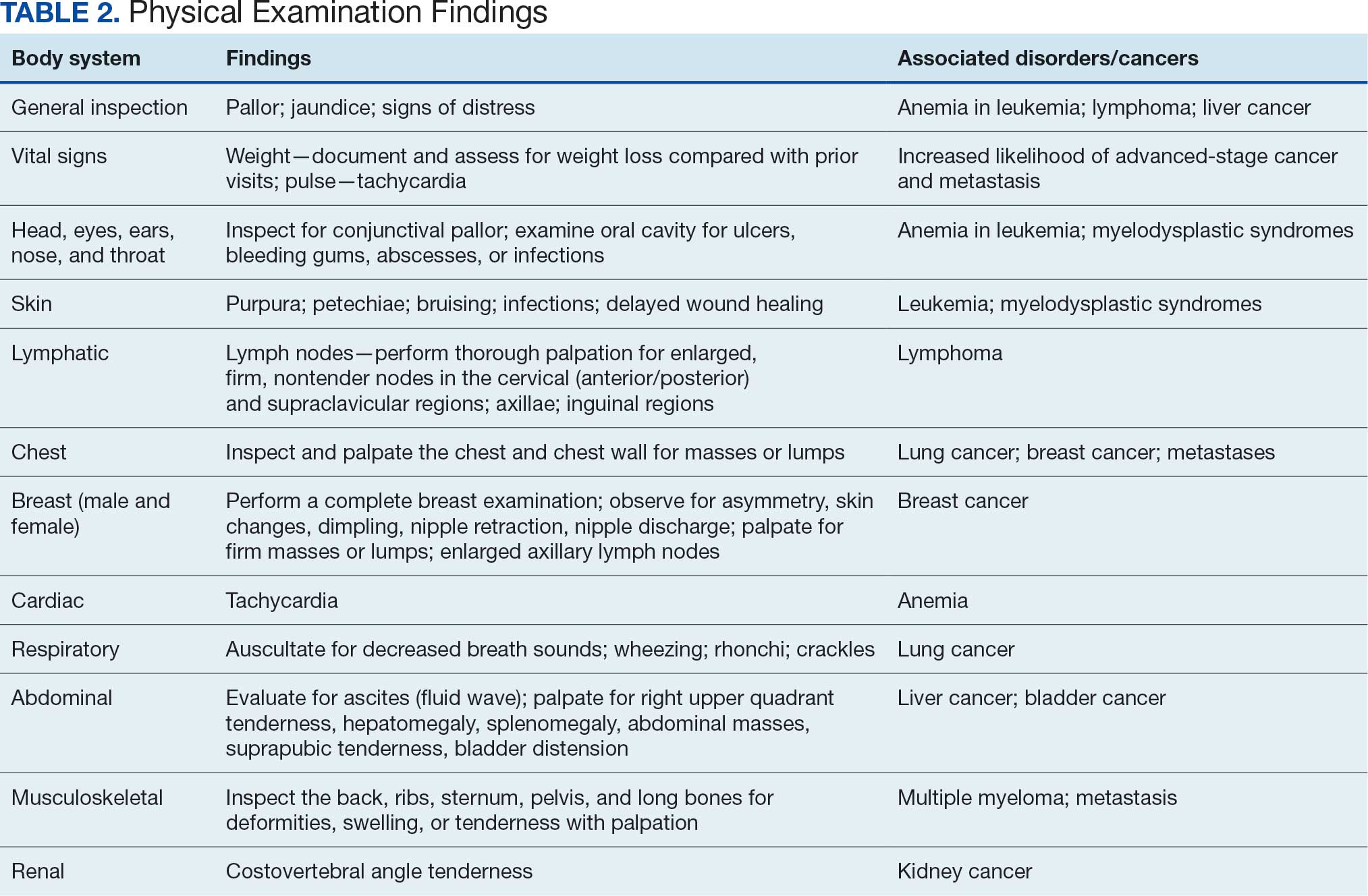
PHYSICAL EXAMINATION
A focused physical examination may aid HCPs in early detection of the cancers associated with Camp Lejeune (Table 2). The physical examination can guide diagnostic testing and imaging for further assessment and workup for VOC-related cancers.
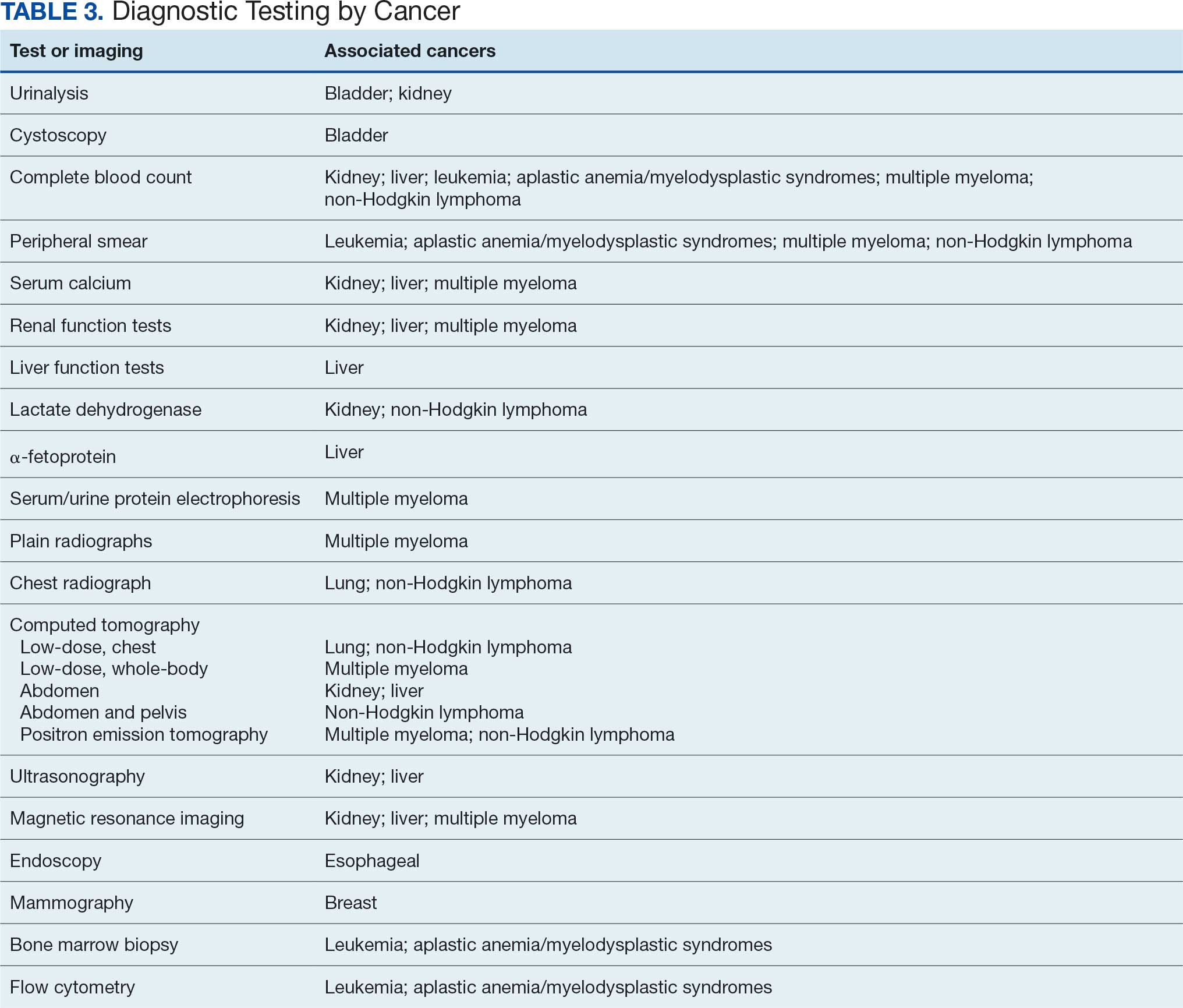
Proposed Diagnostic Screening Panel
Primary care and internal medicine HCPs have the opportunity to improve patient health outcomes by implementing a targeted diagnostic screening panel for identified veterans previously stationed at Camp Lejeune. Early identification of cancers associated with VOCs exposure can facilitate earlier treatment interventions and improve health and quality of life outcomes. The following diagnostic screening panel outlines a potential cost-effective strategy for evaluating and detecting the 10 cancers associated with VOC exposure in Camp Lejeune water.
Baseline Screening
Implementing a diagnostic screening panel in this high-risk cohort can lead to earlier diagnosis, reduce mortality, and improve patient outcomes through early intervention, which in turn may result in less invasive treatment. This approach may also reduce health care costs by avoiding costs associated with delayed diagnosis and advanced-stage cancer care (Tables 3 and 4).
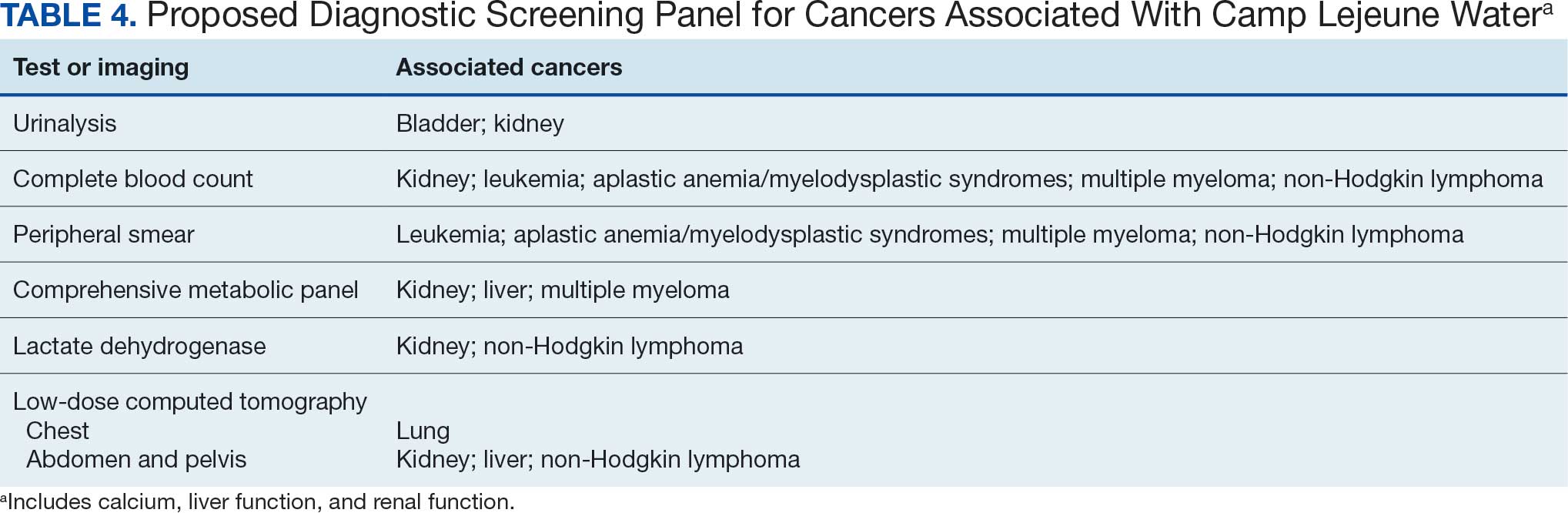
A baseline panel of tests for exposed veterans could include:
- A CBC with differential and peripheral smear to assess for anemia, leukemia, thrombocytopenia, and blast cells associated with leukemias, MDS, multiple myeloma, and NHL.19,46,47,52,55,56,60,61
- CMP evaluates calcium, total protein, renal and liver renal function. Elevated test results may indicate kidney or liver cancer or multiple myeloma.19,27,28,55,56
- LDH testing may reveal levels that are elevated from tissue damage or high cell turnover in kidney cancer, multiple myeloma, and NHL.19,55,56,60,61
- Urinalysis with microscopy may detect hematuria, proteinuria and cellular casts in bladder and kidney cancers.13,24,19
- Low-dose CTs of the chest, abdomen, and pelvis are recommended for early identification of any masses or lymphadenopathy in lung, kidney, liver cancers, and NHL.19,27,28,32,60,61
COST EFFICIENCY
Screening Panel Cost
According to the Medicare Clinical Laboratory Fee Schedule payment cap for 2018, the mean cost for the proposed blood workup was $35 (CBC, $10; CMP, $13; LDH, $8; urinalysis, $4).62 Medicare procedure price schedule for 2025 includes $351 for a CT of the abdomen and pelvis with and without contrast (Current Procedural Terminology [CPT] code 74177) and $187 for a CT of the chest with and without contrast (CPT code 71270).63,64 The total proposed diagnostic screening panel payment cost about $572.
Cancer Care Cost
The average cost for initial cancer care across all cancer sites from 2007 to 2013 was $43,516 per patient; Camp Lejeune-associated cancers ranged from $26,443 for bladder cancer to $89,947 for esophageal cancer care.64 Further, the last year of life cost across all cancer sites averaged $109,727, and Camp Lejeune-associated cancer types ranged from $76,101 for breast cancer to $169,588 for leukemia.65
CONCLUSIONS
From 1953 to 1985, up to 1 million military personnel, civilian workers, and their families stationed at Camp Lejeune were unknowingly exposed to toxic and carcinogenic VOCs, which are associated with = 10 cancers, including bladder, kidney, esophageal, liver, lung, breast, and hematologic malignancies.1-4 Some veterans may be asymptomatic, whereas others present with subtle or specific symptoms that can vary by individual and the type and stage of cancer. HCPs have an opportunity to improve patient outcomes through awareness in identifying symptoms associated with Camp Lejeune water exposure and performing a thorough baseline physical examination, especially noting lymphadenopathy, unexplained weight loss, or masses, which can guide further diagnostic evaluation. Timely screening can identify cancers earlier, reducing delays in care, mitigating the cost burden associated with advanced-stage cancer treatment, improving survival outcomes, and enhancing quality of life. Primary care and internal medicine HCPs specifically play a crucial role in early recognition, physical assessment, and appropriate screening tools. A proposed panel includes CBC with differential and peripheral smear, CMP, LDH, urinalysis, and low-dose CTs of the chest, abdomen and pelvis. Implementation should be guided by clinical judgment and patient-specific risk factors. The proposed diagnostic screening panel is a small price to pay for those who served in any capacity at Camp Lejeune.
- Bove FJ, Greek A, Gatiba R, et al. Cancer incidence among Marines and Navy personnel and civilian workers exposed to industrial solvents in drinking water at US Marine Corps Base Camp Lejeune: a cohort study. Environ Health Perspect. 2024;132:107008. doi:10.1289/EHP14966
- Maslia ML, Aral MM, Ruckart PZ, Bove FJ. Reconstructing historical VOC concentrations in drinking water for epidemiological studies at a US military base: summary of results. Water (Basel). 2016;8:449. doi:10.3390/w8100449
- Rosenfeld PE, Spaeth KR, McCarthy SJ, et al. Camp Lejeune Marine cancer risk assessment for exposure to contaminated drinking water from 1955 to 1987. Water Air Soil Pollut. 2024;235(2). doi:10.1007/s11270-023-06863-y
- US Department of Veterans Affairs, Veterans Health Administration. Camp Lejeune: past water contamination. Updated April 15, 2025. Accessed March 3, 2026. https://www.publichealth.va.gov/exposures/camp-lejeune/
- Jung K, Khan A, Mocharnuk R, et al. Clinical encounter with three cancer patients affected by groundwater contamination at Camp Lejeune: a case series and review of the literature. J Med Case Rep. 2022;16(1):272. doi:10.1186/s13256-022-03501-9
- Honoring America's Veterans and Caring for Camp Lejeune Familes Act of 2012, Pub L No. 112-154. Janey Ensminger Act. Congress.gov. Accessed April 15, 2026. https://ww.congress.gov/bill/112th-congress/house-bill/1627
- Bove FJ, Greek A, Gatiba R, et al. Evaluation of mortality among Marines, Navy personnel, and civilian workers exposed to contaminated drinking water at USMC Base Camp Lejeune: a cohort study. Environ Health. 2024;23(1):61. doi:10.1186/s12940-024-01099-7
- Honoring our PACT Act of 2022 (Pub L No. 117-168): expansion of health care eligibility and toxic exposure screenings. Congress.gov. Accessed March 3, 2026. https://www.congress.gov/bill/117th-congress/house-bill/3967
- Ensuring Justice for Camp Lejeune Victims Act of 2025. Congress.gov. Accessed March 24, 2026. https://www.congress.gov/bill/119th-congress/house-bill/4145
- SEER. Cancer stat facts: bladder cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/urinb.html
- Agency for Toxic Substances and Disease Registry. ATSDR assessment of the evidence for the drinking water contaminants at Camp Lejeune and specific cancers and other diseases. Published January 13, 2017. Accessed March 3, 2026. https://www.atsdr.cdc.gov/camp-lejeune/media/pdfs/2024/10/ATSDR_summary_of_the_evidence_for_causality_TCE_PCE_508.pdf
- National Cancer Institute. What is bladder cancer? Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder
- National Cancer Institute. Bladder cancer symptoms. Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/symptoms
- American Cancer Society. Bladder cancer signs and symptoms. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- National Cancer Institute. Bladder cancer screening. Updated April 27, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/screening
- American Cancer Society. Tests for bladder cancer. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: kidney and renal pelvis cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/kidrp.html
- American Cancer Society. Kidney cancer signs and symptoms. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for kidney cancer. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: esophageal cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/esoph.html
- Engel LS, Chow WH, Vaughan TL, et al. Population attributable risks of esophageal and gastric cancers.
J Natl Cancer Inst. 2003;95(18):1404-1413. doi:10.1093/jnci/djg047 - American Cancer Society. Signs and symptoms of esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: liver and intrahepatic bile duct cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/livibd.html
- American Cancer Society. Signs and symptoms of liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. What is liver cancer? Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer
- American Cancer Society. Tests for liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/how-diagnosed.html
- National Cancer Institute. Liver cancer screening. Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer/screening
- SEER. Cancer stat facts: lung cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/lungb.html
- US Department of Health and Human Services. The Health Consequences of Smoking: A Report of the Surgeon General. Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2004. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK44695/pdf/Bookshelf_NBK44695.pdf
- American Cancer Society. Lung cancer signs and symptoms. Updated February 27, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for lung cancer. Updated January 29, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: female breast cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/breast.html
- SEER. SEER*Explorer breast incidence and mortality comparison. Updated July 2, 2025. Accessed March 3, 2026. https://seer.cancer.gov/statistics-network/explorer/application.html
- Susan G. Komen. Male breast cancer. Updated June 3, 2025. Accessed March 3, 2026. https://www.komen.org/breast-cancer/facts-statistics/male-breast-cancer/
- American Cancer Society. Key statistics for breast cancer in men. Updated January 16, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/about/key-statistics.html
- Ruckart PZ, Bove FJ, Shanley E 3rd, et al. Evaluation of contaminated drinking water and male breast cancer at Marine Corps Base Camp Lejeune, North Carolina: a case-control study. Environ Health. 2015;14:74. doi:10.1186/s12940-015-0061-4
- Culell P, Solernou L, Tarazona J, et al. Male breast cancer: a multicentric study. Breast J. 2007;13:213-215. doi:10.1111/j.1524-4741.2007.00412.x
- Giordano SH, Cohen DS, Buzdar AU, et al. Breast carcinoma in men: a population-based study. Cancer. 2004;101:51-57. doi:10.1002/cncr.20312
- American Cancer Society. Signs and symptoms of breast cancer in men. Updated April 27, 2018. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests to help diagnose breast cancer in men. Updated December 20, 2021. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/how-diagnosed.html
- Evans GF, Anthony T, Turnage RH, et al. The diagnostic accuracy of mammography in the evaluation of male breast disease. Am J Surg. 2001;181:96-100. doi:10.1016/S0002-9610(00)00571-7
- SEER. Cancer stat facts: leukemia. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/leuks.html
- American Cancer Society. Signs and symptoms of acute myeloid leukemia (AML). Updated March 20, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Signs and symptoms of chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for acute myeloid leukemia (AML). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/how-diagnosed.html
- American Cancer Society. Tests for chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/how-diagnosed.html
- Giudice V, Selleri C. Aplastic anemia: pathophysiology. Semin Hematol. 2022;59:13-20. doi:10.1053/j.seminhematol.2021.12.002
- National Cancer Institute. Myelodysplastic syndromes treatment (PDQ®)–patient version. Updated October 4, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/myeloproliferative/patient/myelodysplastic-treatment-pdq
- SEER. Cancer statistics review (CSR) 1975-2013: myelodysplastic syndromes. Accessed March 3, 2026. https://seer.cancer.gov/archive/csr/1975_2013/browse_csr.php?sectionSEL=30&pageSEL=sect_30_table.01
- American Cancer Society. Signs and symptoms of myelodysplastic syndrome (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for myelodysplastic syndromes (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: myeloma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/mulmy.html
- American Cancer Society. Signs and symptoms of multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/testing.html
- Laubach JP. Multiple myeloma: clinical features, laboratory manifestations, and diagnosis. UpToDate. Updated April 28, 2025. Accessed March 3, 2026. https://www.uptodate.com/contents/multiple-myeloma-clinical-features-laboratory-manifestations-and-diagnosis
- Hillengass J, Usmani S, Rajkumar SV, et al. International Myeloma Working Group consensus recommendations on imaging in monoclonal plasma cell disorders. Lancet Oncol. 2019;20:e302-e312. doi:10.1016/S1470-2045(19)30309-2
- SEER. Cancer stat facts: non-Hodgkin lymphoma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/nhl.html
- American Cancer Society. Signs and symptoms of non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. Non-Hodgkin lymphoma treatment (PDQ®)–patient version. Updated August 22, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/lymphoma/patient/adult-nhl-treatment-pdq
- American Cancer Society. Tests for non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/how-diagnosed.html
- College of American Pathologists. Medicare clinical laboratory fee schedule. Published November 2017. Accessed March 3, 2026. https://documents.cap.org/documents/2018-final-medicare-clfs-rates.pdf
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/71270/
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/74177/
- National Cancer Institute. Cancer trends progress report: financial burden of cancer care. Updated April 2025. Accessed March 3, 2026. https://progressreport.cancer.gov/after/economic_burden
Clinical awareness of cancers associated with Camp Lejeune water contamination exposure remains limited despite legal and policy advances. Gaps persist in early symptom recognition and timely diagnostic evaluation before a definitive cancer diagnosis among exposed personnel. This may represent missed opportunities for earlier identification of volatile organic compounds (VOCs)-related cancers and for less invasive treatment options for veterans in this high-risk population.
Federal health care practitioners (HCPs), especially those in primary care and internal medicine, are uniquely positioned to bridge this gap. By improving the recognition of symptoms, pertinent physical examination findings, and implementing a diagnostic screening panel, HCPs can support accurate diagnoses and facilitate earlier treatment to improve health and quality of life for this population.
From 1953 to 1985, as many as 1 million military personnel, civilian workers, and their families stationed at US Marine Corps Base Camp Lejeune were unknowingly exposed to toxic and carcinogenic chemicals in drinking and bathing water.1 Three of the 8 main water sources on base were contaminated with VOCs, which are associated with multiple cancers.1-3
The US Department of Veterans Affairs (VA) recognizes 15 conditions associated with Camp Lejeune contaminated water exposure for VA benefits, including 10 cancers: adult leukemia; aplastic anemia and other myelodysplastic syndromes (MDS); bladder, esophageal, kidney, liver, breast (male and female), and lung cancers; multiple myeloma; and non-Hodgkin lymphoma (NHL).4
BACKGROUND
Established in 1942, Camp Lejeune is an important Marine Corps training installation. Between 1953 and 1985, multiple on-base water systems were contaminated with VOCs, including trichloroethylene (TCE), perchloroethylene (PCE), benzene, and vinyl chloride, due to improper waste disposal and industrial runoff from on- and off-base sources.5 Tarawa Terrace water treatment plant (WTP) was contaminated primarily with PCE from November 1957 to February 1987. Hadnot Point WTP was contaminated with TCE from August 1953 to December 1984, along with PCE, and benzene, toluene, ethylbenzene, and xylene (BTEX). Holcomb Boulevard WTP, established in 1972, was contaminated with TCE from June 1972 to February 1985.2 These contaminants entered the drinking and bathing water supply over decades, and exposure often occurred concurrently across = 1 VOC, compounding health risks.2,3 This prolonged 32-year VOC exposure window underlies current concerns regarding long-term cancer risk among affected service members, civilian employees, and family members. Epidemiologic research has found statistically significant associations between VOC exposure and multiple cancers, neurologic conditions, and reproductive issues.6 Specifically, TCE is associated with higher risks of hematologic cancers, multiple myeloma, NHL, and kidney cancer.3 PCE is linked with kidney cancer, benzene with multiple myeloma and NHL, and vinyl chloride with hepatobiliary cancers.3 A cohort mortality study compared Camp Lejeune personnel with a control group at Camp Pendleton from 1972 to 1985 and found a 3-fold higher incidence or mortality rate for kidney, esophageal, and female breast cancers, leukemia, and lymphoma among exposed Camp Lejeune personnel.6 Notably, personnel assigned to Camp Lejeune for as little as 6 months faced up to a 6-fold increase in cancer risk; the average military assignment between 1975 and 1985 was 18 months.3,6
Honoring America's Veterans and Caring for Camp Lejeune Families Act of 2012, the Sergeant First Class Heath Robinson Honoring Our Promise to Address Comprehensive Toxics (PACT) Act of 2022, the Camp Lejeune Justice Act of 2022, and the pending Ensuring Justice for Camp Lejeune Victims Act of 2025 provide health care and legal resources for personnel and families affected by Camp Lejeune’s contaminated water.6-8 These laws acknowledge associations between exposure and specific health conditions and expanded health care, benefits, and legal recourse for affected veterans, survivors, and their families.8,9
CANCERS LINKED TO CAMP LEJEUNE
Camp Lejeune VOC-contaminated water exposure is associated with solid tumor and hematologic cancers. Symptoms, physical examination findings, and diagnostic considerations vary by cancer type (Table 1).

Bladder Cancer
The US incidence rate of bladder cancer for both males and females is 18 per 100,000 individuals per year, with a death rate of 4.1 per 100,000 individuals per year, and a 2.1% lifetime diagnosis risk.10 Personnel exposed to VOCs at Camp Lejeune had a 9% higher risk of developing bladder cancer and a 2% increased mortality compared with an unexposed control group at Camp Pendleton.1,7 Other bladder cancer subtypes at increased risk are papillary transitional cell carcinoma, nonpapillary transition cell carcinoma, and urothelial carcinoma.7 This is consistent with prior research that found PCE exposure is associated with an increased risk for bladder cancer.3,7,11 Smoking and tobacco use remain significant risk factors for bladder cancer.12
Symptomatology. The most common symptom associated with bladder cancer is painless hematuria (gross or microscopic). Other often delayed symptoms include urinary frequency, urgency, or nocturia.13,14
Diagnostics. Screening tests include urinalysis for hematuria, urine cytology, and cystoscopy with biopsy as the gold standard for diagnosis and staging.15,16
Kidney Cancer
The US incidence rate of kidney cancer and renal pelvis cancer for both males and females is 17.5 per 100,000 individuals per year, with a death rate of 3.4 per 100,000, and a 1.8% lifetime diagnosis risk.17 Camp Lejeune personnel exposed to VOCs had a 6% increased risk of developing kidney cancer and renal pelvis cancer and a 21% higher mortality risk compared with Camp Pendleton controls.1,7 Subtypes at risk include renal cell carcinoma and papillary carcinoma.7 This is consistent with prior research that found exposures to TCE and PCE are associated with a 3-fold increased risk of kidney cancer.3,7
Symptomatology. Hematuria, flank pain, and a palpable abdominal mass are common symptoms associated with kidney cancer. In advanced stages, other symptoms may include left-sided varicocele, anemia, weight loss, fatigue, fever, and night sweats.18
Diagnostics. Screening tests include urinalysis to assess the presence of blood, complete blood count (CBC) to assess anemia, calcium (elevated), and lactate dehydrogenase (LDH), which may be elevated. Imaging strategies include abdominal computed tomography (CT), magnetic resonance imaging (MRI), or ultrasound.19
Esophageal Cancer
The US incidence rate of esophageal cancer for both males and females is 4.2 per 100,000 individuals per year, the death rate is 3.7 per 100,000 individuals per year, and a 0.5% lifetime diagnosis risk.20 VOC-exposed Camp Lejeune personnel had a 27% increased incidence and 25% increased mortality compared with the control group.1,7 Esophageal cancer subtypes at elevated risk include squamous cell carcinoma and adenocarcinoma. This is consistent with prior research that found Camp Lejeune water exposure is associated with a 3-fold increased risk for esophageal cancer.7 Additional risk factors include history of smoking and alcohol use.21
Symptomatology. Esophageal cancer is often asymptomatic with potential symptoms that include dysphagia, hoarseness, and weight loss in advanced disease.22
Diagnostics. Endoscopy with biopsy is the definitive method for diagnosis.23
Liver Cancer
The US incidence rate of liver cancer and intrahepatic bile duct cancer for both males and females is 9.4 per 100,000 individuals per year, with a death rate of 6.6 per 100,000 individuals per year, and a 1.1% lifetime diagnosis risk.24 VOC-exposed personnel had a 1% higher mortality than controls.1
Symptomatology. Liver cancer is often asymptomatic and appears in late stages.25 Common symptoms include right upper quadrant pain, early satiety, nausea, vomiting, loss of appetite, weight loss, ascites, jaundice, and abnormal bleeding or bruising.25,26
Diagnostics. Diagnostic tests may include an ultrasound, CT, or MRI. Additional laboratory testing may include liver function, a-fetoprotein blood, CBC, renal function, calcium, and hepatitis panel screening for hepatitis B and C.27,28
Lung Cancer
The US incidence rate of lung cancer for both males and females is 47.8 per 100,000 individuals per year, with a death rate of 31.5 per 100,000 individuals per year, and a 5.4% lifetime diagnosis risk.29 VOC-exposed personnel had a 16% increased risk and 19% higher mortality.1,7 Subtypes include large cell, small cell, non-small cell, squamous cell, and adenocarcinoma.7 Smoking is an additional risk factor.30
Symptomatology. Symptoms of lung cancer include cough, shortness of breath, chest pain worse with deep breathing, unexplained weight loss, fatigue, night sweats, and recurrent fevers. Advanced stages may metastasize or spread to the liver, bones, and brain.31
Diagnostics. Low-dose CT and chest X-ray are used for screening.32
Breast Cancer
The US incidence rate of female breast cancer is 130.8 per 100,000 individuals per year, with a death rate of 19.2 per 100,000 individuals per year, and a 13.0% lifetime risk of diagnosis.33 For female VOC-exposed personnel, there was an equal risk of developing breast cancer as the control group.1 However, exposed females at Camp Lejeune had a 23% higher mortality risk compared to the control group.7 Breast cancer subtypes among females include ductal carcinoma, lobular carcinoma, and ductal-lobular carcinoma.1
The US incidence rate of male breast cancer is 1.3 per 100,000 individuals per year, with a death rate of 0.3 per 100,000 individuals per year.34,35 The lifetime risk for males developing breast cancer is 137.7 per 100,000 and about 70 to 100 times less common in men than women.36
Male personnel exposed at Camp Lejeune had a 4% increased risk for developing breast cancer compared to Camp Pendleton.7 However, mortality was lower in the Camp Lejeune group.1 Although male breast cancer is rare, males at Camp Lejeune had a higher incidence, indicating a link between TCE, PCE, vinyl chloride exposures and male breast cancer.37 Male breast cancer is more often diagnosed in advanced stages than female breast cancer due to the lack of awareness or absence of routine screenings.38 The most common breast cancer type in males is invasive ductal carcinoma, accounting for 85% to 90% of cases; lobular carcinoma is the second most common type.39
Symptomatology. In both females and males, breast cancer symptoms include painless, firm mass or lump in the breast (left breast slightly more common than right), skin changes or dimpling, nipple retraction or turning inward, and nipple discharge. Breast cancer can spread to the lymph nodes and can be appreciated in axilla or clavicular regions.40
Diagnostics. The diagnostic evaluation for breast cancer is similar for females and males. It includes a clinical breast examination, diagnostic mammogram, and ultrasound.41 Mammograms can distinguish between gynecomastia and cancer, especially in males.42 A core or fine needle biopsy is needed to confirm diagnosis.41
Adult Leukemia
The US incidence rate of leukemia for both male and female was 14.4 per 100,000 individuals per year, with a death rate of 5.8 per 100,000 individuals per year, and a 1.5% lifetime diagnosis risk.43
VOC-exposed personnel had a 7% higher risk of developing leukemia and a 13% increased mortality risk compared with the control group.1,7 Subtypes of leukemia at risk included a 38% increased incidence of acute myeloid/monocytic leukemia (AML) and a 2% increased incidence of chronic lymphocytic leukemia (CLL).1 Benzene and TCE exposures are known risk factors for AML and other leukemias.7 Personnel at Camp Lejeune had 3 times the incidence or mortality for leukemia, specifically AML mortality at 20%.7 Smoking is an additional risk factor for certain leukemias, especially AML.30
Symptomatology. Symptoms associated with leukemia are often nonspecific and may include fatigue, pallor, easy bruising or bleeding (skin or gums), recurrent infections secondary to neutropenia, fever, night sweats, pain or feeling full after a small meal due to enlarged spleen or liver, and weight loss.44,45
Diagnostics. An initial screening includes a CBC with differential, a peripheral smear to detect the presence of blast cells, as well as Auer rods in myeloid blast cells in AML or smudge cells in CLL. Confirmatory tests may include bone marrow biopsy or flow cytometry. A referral to a hematologist is recommended for any suspected leukemia.46,47
Myelodysplastic Syndromes
Aplastic anemia and MDS are considered rare disorders.48 Aplastic anemia is a nonmalignant bone marrow failure disorder with pancytopenia and hypocellular bone marrow due to the loss of hematopoietic stem cells.48 MDS is a type of hematopoietic cancer where the bone marrow produces abnormal blood cells or does not make enough healthy cells.49 This can lead to an increased risk for infection, cytopenias, neutropenia, refractory anemia, and thrombocytopenia, and progression to AML in some patients.49
The reported US incidence of MDS from 1975 to 2013 was 6.7 per 100,000 for males and 3.7 per 100,000 for females.50 Benzene exposure is linked to MDS and a known cause of AML.1 VOC-exposed personnel had a 68% increased risk of developing MDS and a 2.3-fold increased mortality risk compared to controls.1,7
Symptomatology. Some patients are asymptomatic at diagnosis.51 Symptoms related to cytopenia include fatigue, pallor, purpura, petechiae, bleeding of skin, gum, or nose, recurrent infections, fever, bone pain, loss of appetite, and weight loss.50,51
Diagnostics. Initial workup includes a CBC with differential to assess for anemia, white blood cell and absolute neutrophil counts (low), and thrombocytopenia.52 A peripheral blood smear may show myeloid blast cells. A bone marrow aspiration and biopsy, flow cytometry, and cytogenetic or molecular testing may be performed. If MDS is suspected, a referral to a hematologist should be considered.52
Multiple Myeloma
The US incidence rate of multiple myeloma for both males and females is 7.3 per 100,000 individuals per year, with a mortality rate of 2.9 per 100,000 individuals per year, and a 0.8% lifetime diagnosis risk.53 VOC-exposed personnel had a 13% increased risk of developing multiple myeloma and an 8% increased mortality risk compared to unexposed personnel.1,7
Symptomatology. Multiple myeloma may be asymptomatic in early stages. The most common presenting symptom is bone pain, especially in the back, hips, and long bones, due to hypercalcemia from increased reabsorption, plasma cell tumor overgrowth in the bone marrow, and lytic lesions.54 Additional symptoms include fatigue and pallor related to anemia, leukopenia, thrombocytopenia, recurrent infections, extreme thirst, frequent urination, dehydration, confusion associated with hypercalcemia, peripheral neuropathy, loss of appetite, weight loss, and renal impairment or failure.54
Diagnostics. Testing considerations include a CBC with a peripheral blood smear to evaluate anemia and rouleaux formation of red blood cells (seen in > 50% of patients with multiple myeloma), comprehensive metabolic panel (CMP) to assess kidney function, calcium levels (elevated), serum and urine protein electrophoresis with immunofixation to detect monoclonal protein (detected in > 80% of patients with multiple myeloma) and Bence-Jones proteins, serum free light chain assay, and a bone marrow biopsy for diagnosis.55,56
MRI of the spine and pelvis is the most sensitive to detecting bone marrow involvement and focal lesions before lytic lesion progression occurs and for assessing spinal cord compression.57 PET/CT is more sensitive at detecting extramedullary disease, outside of the spine, and for patients that cannot undergo MRI.57 A whole-body low-dose CT, either alone or with PET, is more sensitive than an X-ray at detecting lytic lesions, fractures, or osteoporosis associated with multiple myeloma.57
Non-Hodgkin Lymphoma
The US incidence rate of NHL for both males and females are 18.7 per 100,000 individuals per year, the death rate is 4.9 per 100,000 individuals per year, and a 2% lifetime diagnosis risk.58 VOC-exposed personnel had a 1% higher risk of developing NHL and a decreased mortality risk compared to the control group.1,7 Specific NHL subtypes with increased risk in the exposed cohort are mantle cell (26%), follicular (7%), Burkitt (53%), and marginal zone B-cell (45%).7
Symptomatology. NHL often presents with painless lymphadenopathy or enlarged lymph nodes involving the cervical, axillary, inguinal regions.59,60 Other symptoms include frequent infections, unexplained bruising, weight loss, and “B symptoms,” such as fever and night sweats.59,60 Some patients develop a mediastinal mass in the thorax, which if large may lead to cough or shortness of breath.59
Diagnostics. The initial diagnostic workup includes CBC with differential and LDH, which may be elevated.60,61 Imaging may begin with a chest X-ray to assess for a mediastinal mass; however, CTs of the chest, abdomen, and pelvis provide more detail to better assess for NHL. Whole body PET/CT is considered the gold standard for assessing and staging systemic involvement. If enlarged lymph nodes are present, a biopsy can confirm the subtype of NHL.60,61
PHYSICAL EXAMINATION
A focused physical examination may aid HCPs in early detection of the cancers associated with Camp Lejeune (Table 2). The physical examination can guide diagnostic testing and imaging for further assessment and workup for VOC-related cancers.

Proposed Diagnostic Screening Panel
Primary care and internal medicine HCPs have the opportunity to improve patient health outcomes by implementing a targeted diagnostic screening panel for identified veterans previously stationed at Camp Lejeune. Early identification of cancers associated with VOCs exposure can facilitate earlier treatment interventions and improve health and quality of life outcomes. The following diagnostic screening panel outlines a potential cost-effective strategy for evaluating and detecting the 10 cancers associated with VOC exposure in Camp Lejeune water.
Baseline Screening
Implementing a diagnostic screening panel in this high-risk cohort can lead to earlier diagnosis, reduce mortality, and improve patient outcomes through early intervention, which in turn may result in less invasive treatment. This approach may also reduce health care costs by avoiding costs associated with delayed diagnosis and advanced-stage cancer care (Tables 3 and 4).


A baseline panel of tests for exposed veterans could include:
- A CBC with differential and peripheral smear to assess for anemia, leukemia, thrombocytopenia, and blast cells associated with leukemias, MDS, multiple myeloma, and NHL.19,46,47,52,55,56,60,61
- CMP evaluates calcium, total protein, renal and liver renal function. Elevated test results may indicate kidney or liver cancer or multiple myeloma.19,27,28,55,56
- LDH testing may reveal levels that are elevated from tissue damage or high cell turnover in kidney cancer, multiple myeloma, and NHL.19,55,56,60,61
- Urinalysis with microscopy may detect hematuria, proteinuria and cellular casts in bladder and kidney cancers.13,24,19
- Low-dose CTs of the chest, abdomen, and pelvis are recommended for early identification of any masses or lymphadenopathy in lung, kidney, liver cancers, and NHL.19,27,28,32,60,61
COST EFFICIENCY
Screening Panel Cost
According to the Medicare Clinical Laboratory Fee Schedule payment cap for 2018, the mean cost for the proposed blood workup was $35 (CBC, $10; CMP, $13; LDH, $8; urinalysis, $4).62 Medicare procedure price schedule for 2025 includes $351 for a CT of the abdomen and pelvis with and without contrast (Current Procedural Terminology [CPT] code 74177) and $187 for a CT of the chest with and without contrast (CPT code 71270).63,64 The total proposed diagnostic screening panel payment cost about $572.
Cancer Care Cost
The average cost for initial cancer care across all cancer sites from 2007 to 2013 was $43,516 per patient; Camp Lejeune-associated cancers ranged from $26,443 for bladder cancer to $89,947 for esophageal cancer care.64 Further, the last year of life cost across all cancer sites averaged $109,727, and Camp Lejeune-associated cancer types ranged from $76,101 for breast cancer to $169,588 for leukemia.65
CONCLUSIONS
From 1953 to 1985, up to 1 million military personnel, civilian workers, and their families stationed at Camp Lejeune were unknowingly exposed to toxic and carcinogenic VOCs, which are associated with = 10 cancers, including bladder, kidney, esophageal, liver, lung, breast, and hematologic malignancies.1-4 Some veterans may be asymptomatic, whereas others present with subtle or specific symptoms that can vary by individual and the type and stage of cancer. HCPs have an opportunity to improve patient outcomes through awareness in identifying symptoms associated with Camp Lejeune water exposure and performing a thorough baseline physical examination, especially noting lymphadenopathy, unexplained weight loss, or masses, which can guide further diagnostic evaluation. Timely screening can identify cancers earlier, reducing delays in care, mitigating the cost burden associated with advanced-stage cancer treatment, improving survival outcomes, and enhancing quality of life. Primary care and internal medicine HCPs specifically play a crucial role in early recognition, physical assessment, and appropriate screening tools. A proposed panel includes CBC with differential and peripheral smear, CMP, LDH, urinalysis, and low-dose CTs of the chest, abdomen and pelvis. Implementation should be guided by clinical judgment and patient-specific risk factors. The proposed diagnostic screening panel is a small price to pay for those who served in any capacity at Camp Lejeune.
Clinical awareness of cancers associated with Camp Lejeune water contamination exposure remains limited despite legal and policy advances. Gaps persist in early symptom recognition and timely diagnostic evaluation before a definitive cancer diagnosis among exposed personnel. This may represent missed opportunities for earlier identification of volatile organic compounds (VOCs)-related cancers and for less invasive treatment options for veterans in this high-risk population.
Federal health care practitioners (HCPs), especially those in primary care and internal medicine, are uniquely positioned to bridge this gap. By improving the recognition of symptoms, pertinent physical examination findings, and implementing a diagnostic screening panel, HCPs can support accurate diagnoses and facilitate earlier treatment to improve health and quality of life for this population.
From 1953 to 1985, as many as 1 million military personnel, civilian workers, and their families stationed at US Marine Corps Base Camp Lejeune were unknowingly exposed to toxic and carcinogenic chemicals in drinking and bathing water.1 Three of the 8 main water sources on base were contaminated with VOCs, which are associated with multiple cancers.1-3
The US Department of Veterans Affairs (VA) recognizes 15 conditions associated with Camp Lejeune contaminated water exposure for VA benefits, including 10 cancers: adult leukemia; aplastic anemia and other myelodysplastic syndromes (MDS); bladder, esophageal, kidney, liver, breast (male and female), and lung cancers; multiple myeloma; and non-Hodgkin lymphoma (NHL).4
BACKGROUND
Established in 1942, Camp Lejeune is an important Marine Corps training installation. Between 1953 and 1985, multiple on-base water systems were contaminated with VOCs, including trichloroethylene (TCE), perchloroethylene (PCE), benzene, and vinyl chloride, due to improper waste disposal and industrial runoff from on- and off-base sources.5 Tarawa Terrace water treatment plant (WTP) was contaminated primarily with PCE from November 1957 to February 1987. Hadnot Point WTP was contaminated with TCE from August 1953 to December 1984, along with PCE, and benzene, toluene, ethylbenzene, and xylene (BTEX). Holcomb Boulevard WTP, established in 1972, was contaminated with TCE from June 1972 to February 1985.2 These contaminants entered the drinking and bathing water supply over decades, and exposure often occurred concurrently across = 1 VOC, compounding health risks.2,3 This prolonged 32-year VOC exposure window underlies current concerns regarding long-term cancer risk among affected service members, civilian employees, and family members. Epidemiologic research has found statistically significant associations between VOC exposure and multiple cancers, neurologic conditions, and reproductive issues.6 Specifically, TCE is associated with higher risks of hematologic cancers, multiple myeloma, NHL, and kidney cancer.3 PCE is linked with kidney cancer, benzene with multiple myeloma and NHL, and vinyl chloride with hepatobiliary cancers.3 A cohort mortality study compared Camp Lejeune personnel with a control group at Camp Pendleton from 1972 to 1985 and found a 3-fold higher incidence or mortality rate for kidney, esophageal, and female breast cancers, leukemia, and lymphoma among exposed Camp Lejeune personnel.6 Notably, personnel assigned to Camp Lejeune for as little as 6 months faced up to a 6-fold increase in cancer risk; the average military assignment between 1975 and 1985 was 18 months.3,6
Honoring America's Veterans and Caring for Camp Lejeune Families Act of 2012, the Sergeant First Class Heath Robinson Honoring Our Promise to Address Comprehensive Toxics (PACT) Act of 2022, the Camp Lejeune Justice Act of 2022, and the pending Ensuring Justice for Camp Lejeune Victims Act of 2025 provide health care and legal resources for personnel and families affected by Camp Lejeune’s contaminated water.6-8 These laws acknowledge associations between exposure and specific health conditions and expanded health care, benefits, and legal recourse for affected veterans, survivors, and their families.8,9
CANCERS LINKED TO CAMP LEJEUNE
Camp Lejeune VOC-contaminated water exposure is associated with solid tumor and hematologic cancers. Symptoms, physical examination findings, and diagnostic considerations vary by cancer type (Table 1).

Bladder Cancer
The US incidence rate of bladder cancer for both males and females is 18 per 100,000 individuals per year, with a death rate of 4.1 per 100,000 individuals per year, and a 2.1% lifetime diagnosis risk.10 Personnel exposed to VOCs at Camp Lejeune had a 9% higher risk of developing bladder cancer and a 2% increased mortality compared with an unexposed control group at Camp Pendleton.1,7 Other bladder cancer subtypes at increased risk are papillary transitional cell carcinoma, nonpapillary transition cell carcinoma, and urothelial carcinoma.7 This is consistent with prior research that found PCE exposure is associated with an increased risk for bladder cancer.3,7,11 Smoking and tobacco use remain significant risk factors for bladder cancer.12
Symptomatology. The most common symptom associated with bladder cancer is painless hematuria (gross or microscopic). Other often delayed symptoms include urinary frequency, urgency, or nocturia.13,14
Diagnostics. Screening tests include urinalysis for hematuria, urine cytology, and cystoscopy with biopsy as the gold standard for diagnosis and staging.15,16
Kidney Cancer
The US incidence rate of kidney cancer and renal pelvis cancer for both males and females is 17.5 per 100,000 individuals per year, with a death rate of 3.4 per 100,000, and a 1.8% lifetime diagnosis risk.17 Camp Lejeune personnel exposed to VOCs had a 6% increased risk of developing kidney cancer and renal pelvis cancer and a 21% higher mortality risk compared with Camp Pendleton controls.1,7 Subtypes at risk include renal cell carcinoma and papillary carcinoma.7 This is consistent with prior research that found exposures to TCE and PCE are associated with a 3-fold increased risk of kidney cancer.3,7
Symptomatology. Hematuria, flank pain, and a palpable abdominal mass are common symptoms associated with kidney cancer. In advanced stages, other symptoms may include left-sided varicocele, anemia, weight loss, fatigue, fever, and night sweats.18
Diagnostics. Screening tests include urinalysis to assess the presence of blood, complete blood count (CBC) to assess anemia, calcium (elevated), and lactate dehydrogenase (LDH), which may be elevated. Imaging strategies include abdominal computed tomography (CT), magnetic resonance imaging (MRI), or ultrasound.19
Esophageal Cancer
The US incidence rate of esophageal cancer for both males and females is 4.2 per 100,000 individuals per year, the death rate is 3.7 per 100,000 individuals per year, and a 0.5% lifetime diagnosis risk.20 VOC-exposed Camp Lejeune personnel had a 27% increased incidence and 25% increased mortality compared with the control group.1,7 Esophageal cancer subtypes at elevated risk include squamous cell carcinoma and adenocarcinoma. This is consistent with prior research that found Camp Lejeune water exposure is associated with a 3-fold increased risk for esophageal cancer.7 Additional risk factors include history of smoking and alcohol use.21
Symptomatology. Esophageal cancer is often asymptomatic with potential symptoms that include dysphagia, hoarseness, and weight loss in advanced disease.22
Diagnostics. Endoscopy with biopsy is the definitive method for diagnosis.23
Liver Cancer
The US incidence rate of liver cancer and intrahepatic bile duct cancer for both males and females is 9.4 per 100,000 individuals per year, with a death rate of 6.6 per 100,000 individuals per year, and a 1.1% lifetime diagnosis risk.24 VOC-exposed personnel had a 1% higher mortality than controls.1
Symptomatology. Liver cancer is often asymptomatic and appears in late stages.25 Common symptoms include right upper quadrant pain, early satiety, nausea, vomiting, loss of appetite, weight loss, ascites, jaundice, and abnormal bleeding or bruising.25,26
Diagnostics. Diagnostic tests may include an ultrasound, CT, or MRI. Additional laboratory testing may include liver function, a-fetoprotein blood, CBC, renal function, calcium, and hepatitis panel screening for hepatitis B and C.27,28
Lung Cancer
The US incidence rate of lung cancer for both males and females is 47.8 per 100,000 individuals per year, with a death rate of 31.5 per 100,000 individuals per year, and a 5.4% lifetime diagnosis risk.29 VOC-exposed personnel had a 16% increased risk and 19% higher mortality.1,7 Subtypes include large cell, small cell, non-small cell, squamous cell, and adenocarcinoma.7 Smoking is an additional risk factor.30
Symptomatology. Symptoms of lung cancer include cough, shortness of breath, chest pain worse with deep breathing, unexplained weight loss, fatigue, night sweats, and recurrent fevers. Advanced stages may metastasize or spread to the liver, bones, and brain.31
Diagnostics. Low-dose CT and chest X-ray are used for screening.32
Breast Cancer
The US incidence rate of female breast cancer is 130.8 per 100,000 individuals per year, with a death rate of 19.2 per 100,000 individuals per year, and a 13.0% lifetime risk of diagnosis.33 For female VOC-exposed personnel, there was an equal risk of developing breast cancer as the control group.1 However, exposed females at Camp Lejeune had a 23% higher mortality risk compared to the control group.7 Breast cancer subtypes among females include ductal carcinoma, lobular carcinoma, and ductal-lobular carcinoma.1
The US incidence rate of male breast cancer is 1.3 per 100,000 individuals per year, with a death rate of 0.3 per 100,000 individuals per year.34,35 The lifetime risk for males developing breast cancer is 137.7 per 100,000 and about 70 to 100 times less common in men than women.36
Male personnel exposed at Camp Lejeune had a 4% increased risk for developing breast cancer compared to Camp Pendleton.7 However, mortality was lower in the Camp Lejeune group.1 Although male breast cancer is rare, males at Camp Lejeune had a higher incidence, indicating a link between TCE, PCE, vinyl chloride exposures and male breast cancer.37 Male breast cancer is more often diagnosed in advanced stages than female breast cancer due to the lack of awareness or absence of routine screenings.38 The most common breast cancer type in males is invasive ductal carcinoma, accounting for 85% to 90% of cases; lobular carcinoma is the second most common type.39
Symptomatology. In both females and males, breast cancer symptoms include painless, firm mass or lump in the breast (left breast slightly more common than right), skin changes or dimpling, nipple retraction or turning inward, and nipple discharge. Breast cancer can spread to the lymph nodes and can be appreciated in axilla or clavicular regions.40
Diagnostics. The diagnostic evaluation for breast cancer is similar for females and males. It includes a clinical breast examination, diagnostic mammogram, and ultrasound.41 Mammograms can distinguish between gynecomastia and cancer, especially in males.42 A core or fine needle biopsy is needed to confirm diagnosis.41
Adult Leukemia
The US incidence rate of leukemia for both male and female was 14.4 per 100,000 individuals per year, with a death rate of 5.8 per 100,000 individuals per year, and a 1.5% lifetime diagnosis risk.43
VOC-exposed personnel had a 7% higher risk of developing leukemia and a 13% increased mortality risk compared with the control group.1,7 Subtypes of leukemia at risk included a 38% increased incidence of acute myeloid/monocytic leukemia (AML) and a 2% increased incidence of chronic lymphocytic leukemia (CLL).1 Benzene and TCE exposures are known risk factors for AML and other leukemias.7 Personnel at Camp Lejeune had 3 times the incidence or mortality for leukemia, specifically AML mortality at 20%.7 Smoking is an additional risk factor for certain leukemias, especially AML.30
Symptomatology. Symptoms associated with leukemia are often nonspecific and may include fatigue, pallor, easy bruising or bleeding (skin or gums), recurrent infections secondary to neutropenia, fever, night sweats, pain or feeling full after a small meal due to enlarged spleen or liver, and weight loss.44,45
Diagnostics. An initial screening includes a CBC with differential, a peripheral smear to detect the presence of blast cells, as well as Auer rods in myeloid blast cells in AML or smudge cells in CLL. Confirmatory tests may include bone marrow biopsy or flow cytometry. A referral to a hematologist is recommended for any suspected leukemia.46,47
Myelodysplastic Syndromes
Aplastic anemia and MDS are considered rare disorders.48 Aplastic anemia is a nonmalignant bone marrow failure disorder with pancytopenia and hypocellular bone marrow due to the loss of hematopoietic stem cells.48 MDS is a type of hematopoietic cancer where the bone marrow produces abnormal blood cells or does not make enough healthy cells.49 This can lead to an increased risk for infection, cytopenias, neutropenia, refractory anemia, and thrombocytopenia, and progression to AML in some patients.49
The reported US incidence of MDS from 1975 to 2013 was 6.7 per 100,000 for males and 3.7 per 100,000 for females.50 Benzene exposure is linked to MDS and a known cause of AML.1 VOC-exposed personnel had a 68% increased risk of developing MDS and a 2.3-fold increased mortality risk compared to controls.1,7
Symptomatology. Some patients are asymptomatic at diagnosis.51 Symptoms related to cytopenia include fatigue, pallor, purpura, petechiae, bleeding of skin, gum, or nose, recurrent infections, fever, bone pain, loss of appetite, and weight loss.50,51
Diagnostics. Initial workup includes a CBC with differential to assess for anemia, white blood cell and absolute neutrophil counts (low), and thrombocytopenia.52 A peripheral blood smear may show myeloid blast cells. A bone marrow aspiration and biopsy, flow cytometry, and cytogenetic or molecular testing may be performed. If MDS is suspected, a referral to a hematologist should be considered.52
Multiple Myeloma
The US incidence rate of multiple myeloma for both males and females is 7.3 per 100,000 individuals per year, with a mortality rate of 2.9 per 100,000 individuals per year, and a 0.8% lifetime diagnosis risk.53 VOC-exposed personnel had a 13% increased risk of developing multiple myeloma and an 8% increased mortality risk compared to unexposed personnel.1,7
Symptomatology. Multiple myeloma may be asymptomatic in early stages. The most common presenting symptom is bone pain, especially in the back, hips, and long bones, due to hypercalcemia from increased reabsorption, plasma cell tumor overgrowth in the bone marrow, and lytic lesions.54 Additional symptoms include fatigue and pallor related to anemia, leukopenia, thrombocytopenia, recurrent infections, extreme thirst, frequent urination, dehydration, confusion associated with hypercalcemia, peripheral neuropathy, loss of appetite, weight loss, and renal impairment or failure.54
Diagnostics. Testing considerations include a CBC with a peripheral blood smear to evaluate anemia and rouleaux formation of red blood cells (seen in > 50% of patients with multiple myeloma), comprehensive metabolic panel (CMP) to assess kidney function, calcium levels (elevated), serum and urine protein electrophoresis with immunofixation to detect monoclonal protein (detected in > 80% of patients with multiple myeloma) and Bence-Jones proteins, serum free light chain assay, and a bone marrow biopsy for diagnosis.55,56
MRI of the spine and pelvis is the most sensitive to detecting bone marrow involvement and focal lesions before lytic lesion progression occurs and for assessing spinal cord compression.57 PET/CT is more sensitive at detecting extramedullary disease, outside of the spine, and for patients that cannot undergo MRI.57 A whole-body low-dose CT, either alone or with PET, is more sensitive than an X-ray at detecting lytic lesions, fractures, or osteoporosis associated with multiple myeloma.57
Non-Hodgkin Lymphoma
The US incidence rate of NHL for both males and females are 18.7 per 100,000 individuals per year, the death rate is 4.9 per 100,000 individuals per year, and a 2% lifetime diagnosis risk.58 VOC-exposed personnel had a 1% higher risk of developing NHL and a decreased mortality risk compared to the control group.1,7 Specific NHL subtypes with increased risk in the exposed cohort are mantle cell (26%), follicular (7%), Burkitt (53%), and marginal zone B-cell (45%).7
Symptomatology. NHL often presents with painless lymphadenopathy or enlarged lymph nodes involving the cervical, axillary, inguinal regions.59,60 Other symptoms include frequent infections, unexplained bruising, weight loss, and “B symptoms,” such as fever and night sweats.59,60 Some patients develop a mediastinal mass in the thorax, which if large may lead to cough or shortness of breath.59
Diagnostics. The initial diagnostic workup includes CBC with differential and LDH, which may be elevated.60,61 Imaging may begin with a chest X-ray to assess for a mediastinal mass; however, CTs of the chest, abdomen, and pelvis provide more detail to better assess for NHL. Whole body PET/CT is considered the gold standard for assessing and staging systemic involvement. If enlarged lymph nodes are present, a biopsy can confirm the subtype of NHL.60,61
PHYSICAL EXAMINATION
A focused physical examination may aid HCPs in early detection of the cancers associated with Camp Lejeune (Table 2). The physical examination can guide diagnostic testing and imaging for further assessment and workup for VOC-related cancers.

Proposed Diagnostic Screening Panel
Primary care and internal medicine HCPs have the opportunity to improve patient health outcomes by implementing a targeted diagnostic screening panel for identified veterans previously stationed at Camp Lejeune. Early identification of cancers associated with VOCs exposure can facilitate earlier treatment interventions and improve health and quality of life outcomes. The following diagnostic screening panel outlines a potential cost-effective strategy for evaluating and detecting the 10 cancers associated with VOC exposure in Camp Lejeune water.
Baseline Screening
Implementing a diagnostic screening panel in this high-risk cohort can lead to earlier diagnosis, reduce mortality, and improve patient outcomes through early intervention, which in turn may result in less invasive treatment. This approach may also reduce health care costs by avoiding costs associated with delayed diagnosis and advanced-stage cancer care (Tables 3 and 4).


A baseline panel of tests for exposed veterans could include:
- A CBC with differential and peripheral smear to assess for anemia, leukemia, thrombocytopenia, and blast cells associated with leukemias, MDS, multiple myeloma, and NHL.19,46,47,52,55,56,60,61
- CMP evaluates calcium, total protein, renal and liver renal function. Elevated test results may indicate kidney or liver cancer or multiple myeloma.19,27,28,55,56
- LDH testing may reveal levels that are elevated from tissue damage or high cell turnover in kidney cancer, multiple myeloma, and NHL.19,55,56,60,61
- Urinalysis with microscopy may detect hematuria, proteinuria and cellular casts in bladder and kidney cancers.13,24,19
- Low-dose CTs of the chest, abdomen, and pelvis are recommended for early identification of any masses or lymphadenopathy in lung, kidney, liver cancers, and NHL.19,27,28,32,60,61
COST EFFICIENCY
Screening Panel Cost
According to the Medicare Clinical Laboratory Fee Schedule payment cap for 2018, the mean cost for the proposed blood workup was $35 (CBC, $10; CMP, $13; LDH, $8; urinalysis, $4).62 Medicare procedure price schedule for 2025 includes $351 for a CT of the abdomen and pelvis with and without contrast (Current Procedural Terminology [CPT] code 74177) and $187 for a CT of the chest with and without contrast (CPT code 71270).63,64 The total proposed diagnostic screening panel payment cost about $572.
Cancer Care Cost
The average cost for initial cancer care across all cancer sites from 2007 to 2013 was $43,516 per patient; Camp Lejeune-associated cancers ranged from $26,443 for bladder cancer to $89,947 for esophageal cancer care.64 Further, the last year of life cost across all cancer sites averaged $109,727, and Camp Lejeune-associated cancer types ranged from $76,101 for breast cancer to $169,588 for leukemia.65
CONCLUSIONS
From 1953 to 1985, up to 1 million military personnel, civilian workers, and their families stationed at Camp Lejeune were unknowingly exposed to toxic and carcinogenic VOCs, which are associated with = 10 cancers, including bladder, kidney, esophageal, liver, lung, breast, and hematologic malignancies.1-4 Some veterans may be asymptomatic, whereas others present with subtle or specific symptoms that can vary by individual and the type and stage of cancer. HCPs have an opportunity to improve patient outcomes through awareness in identifying symptoms associated with Camp Lejeune water exposure and performing a thorough baseline physical examination, especially noting lymphadenopathy, unexplained weight loss, or masses, which can guide further diagnostic evaluation. Timely screening can identify cancers earlier, reducing delays in care, mitigating the cost burden associated with advanced-stage cancer treatment, improving survival outcomes, and enhancing quality of life. Primary care and internal medicine HCPs specifically play a crucial role in early recognition, physical assessment, and appropriate screening tools. A proposed panel includes CBC with differential and peripheral smear, CMP, LDH, urinalysis, and low-dose CTs of the chest, abdomen and pelvis. Implementation should be guided by clinical judgment and patient-specific risk factors. The proposed diagnostic screening panel is a small price to pay for those who served in any capacity at Camp Lejeune.
- Bove FJ, Greek A, Gatiba R, et al. Cancer incidence among Marines and Navy personnel and civilian workers exposed to industrial solvents in drinking water at US Marine Corps Base Camp Lejeune: a cohort study. Environ Health Perspect. 2024;132:107008. doi:10.1289/EHP14966
- Maslia ML, Aral MM, Ruckart PZ, Bove FJ. Reconstructing historical VOC concentrations in drinking water for epidemiological studies at a US military base: summary of results. Water (Basel). 2016;8:449. doi:10.3390/w8100449
- Rosenfeld PE, Spaeth KR, McCarthy SJ, et al. Camp Lejeune Marine cancer risk assessment for exposure to contaminated drinking water from 1955 to 1987. Water Air Soil Pollut. 2024;235(2). doi:10.1007/s11270-023-06863-y
- US Department of Veterans Affairs, Veterans Health Administration. Camp Lejeune: past water contamination. Updated April 15, 2025. Accessed March 3, 2026. https://www.publichealth.va.gov/exposures/camp-lejeune/
- Jung K, Khan A, Mocharnuk R, et al. Clinical encounter with three cancer patients affected by groundwater contamination at Camp Lejeune: a case series and review of the literature. J Med Case Rep. 2022;16(1):272. doi:10.1186/s13256-022-03501-9
- Honoring America's Veterans and Caring for Camp Lejeune Familes Act of 2012, Pub L No. 112-154. Janey Ensminger Act. Congress.gov. Accessed April 15, 2026. https://ww.congress.gov/bill/112th-congress/house-bill/1627
- Bove FJ, Greek A, Gatiba R, et al. Evaluation of mortality among Marines, Navy personnel, and civilian workers exposed to contaminated drinking water at USMC Base Camp Lejeune: a cohort study. Environ Health. 2024;23(1):61. doi:10.1186/s12940-024-01099-7
- Honoring our PACT Act of 2022 (Pub L No. 117-168): expansion of health care eligibility and toxic exposure screenings. Congress.gov. Accessed March 3, 2026. https://www.congress.gov/bill/117th-congress/house-bill/3967
- Ensuring Justice for Camp Lejeune Victims Act of 2025. Congress.gov. Accessed March 24, 2026. https://www.congress.gov/bill/119th-congress/house-bill/4145
- SEER. Cancer stat facts: bladder cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/urinb.html
- Agency for Toxic Substances and Disease Registry. ATSDR assessment of the evidence for the drinking water contaminants at Camp Lejeune and specific cancers and other diseases. Published January 13, 2017. Accessed March 3, 2026. https://www.atsdr.cdc.gov/camp-lejeune/media/pdfs/2024/10/ATSDR_summary_of_the_evidence_for_causality_TCE_PCE_508.pdf
- National Cancer Institute. What is bladder cancer? Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder
- National Cancer Institute. Bladder cancer symptoms. Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/symptoms
- American Cancer Society. Bladder cancer signs and symptoms. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- National Cancer Institute. Bladder cancer screening. Updated April 27, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/screening
- American Cancer Society. Tests for bladder cancer. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: kidney and renal pelvis cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/kidrp.html
- American Cancer Society. Kidney cancer signs and symptoms. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for kidney cancer. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: esophageal cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/esoph.html
- Engel LS, Chow WH, Vaughan TL, et al. Population attributable risks of esophageal and gastric cancers.
J Natl Cancer Inst. 2003;95(18):1404-1413. doi:10.1093/jnci/djg047 - American Cancer Society. Signs and symptoms of esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: liver and intrahepatic bile duct cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/livibd.html
- American Cancer Society. Signs and symptoms of liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. What is liver cancer? Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer
- American Cancer Society. Tests for liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/how-diagnosed.html
- National Cancer Institute. Liver cancer screening. Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer/screening
- SEER. Cancer stat facts: lung cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/lungb.html
- US Department of Health and Human Services. The Health Consequences of Smoking: A Report of the Surgeon General. Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2004. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK44695/pdf/Bookshelf_NBK44695.pdf
- American Cancer Society. Lung cancer signs and symptoms. Updated February 27, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for lung cancer. Updated January 29, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: female breast cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/breast.html
- SEER. SEER*Explorer breast incidence and mortality comparison. Updated July 2, 2025. Accessed March 3, 2026. https://seer.cancer.gov/statistics-network/explorer/application.html
- Susan G. Komen. Male breast cancer. Updated June 3, 2025. Accessed March 3, 2026. https://www.komen.org/breast-cancer/facts-statistics/male-breast-cancer/
- American Cancer Society. Key statistics for breast cancer in men. Updated January 16, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/about/key-statistics.html
- Ruckart PZ, Bove FJ, Shanley E 3rd, et al. Evaluation of contaminated drinking water and male breast cancer at Marine Corps Base Camp Lejeune, North Carolina: a case-control study. Environ Health. 2015;14:74. doi:10.1186/s12940-015-0061-4
- Culell P, Solernou L, Tarazona J, et al. Male breast cancer: a multicentric study. Breast J. 2007;13:213-215. doi:10.1111/j.1524-4741.2007.00412.x
- Giordano SH, Cohen DS, Buzdar AU, et al. Breast carcinoma in men: a population-based study. Cancer. 2004;101:51-57. doi:10.1002/cncr.20312
- American Cancer Society. Signs and symptoms of breast cancer in men. Updated April 27, 2018. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests to help diagnose breast cancer in men. Updated December 20, 2021. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/how-diagnosed.html
- Evans GF, Anthony T, Turnage RH, et al. The diagnostic accuracy of mammography in the evaluation of male breast disease. Am J Surg. 2001;181:96-100. doi:10.1016/S0002-9610(00)00571-7
- SEER. Cancer stat facts: leukemia. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/leuks.html
- American Cancer Society. Signs and symptoms of acute myeloid leukemia (AML). Updated March 20, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Signs and symptoms of chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for acute myeloid leukemia (AML). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/how-diagnosed.html
- American Cancer Society. Tests for chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/how-diagnosed.html
- Giudice V, Selleri C. Aplastic anemia: pathophysiology. Semin Hematol. 2022;59:13-20. doi:10.1053/j.seminhematol.2021.12.002
- National Cancer Institute. Myelodysplastic syndromes treatment (PDQ®)–patient version. Updated October 4, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/myeloproliferative/patient/myelodysplastic-treatment-pdq
- SEER. Cancer statistics review (CSR) 1975-2013: myelodysplastic syndromes. Accessed March 3, 2026. https://seer.cancer.gov/archive/csr/1975_2013/browse_csr.php?sectionSEL=30&pageSEL=sect_30_table.01
- American Cancer Society. Signs and symptoms of myelodysplastic syndrome (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for myelodysplastic syndromes (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: myeloma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/mulmy.html
- American Cancer Society. Signs and symptoms of multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/testing.html
- Laubach JP. Multiple myeloma: clinical features, laboratory manifestations, and diagnosis. UpToDate. Updated April 28, 2025. Accessed March 3, 2026. https://www.uptodate.com/contents/multiple-myeloma-clinical-features-laboratory-manifestations-and-diagnosis
- Hillengass J, Usmani S, Rajkumar SV, et al. International Myeloma Working Group consensus recommendations on imaging in monoclonal plasma cell disorders. Lancet Oncol. 2019;20:e302-e312. doi:10.1016/S1470-2045(19)30309-2
- SEER. Cancer stat facts: non-Hodgkin lymphoma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/nhl.html
- American Cancer Society. Signs and symptoms of non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. Non-Hodgkin lymphoma treatment (PDQ®)–patient version. Updated August 22, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/lymphoma/patient/adult-nhl-treatment-pdq
- American Cancer Society. Tests for non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/how-diagnosed.html
- College of American Pathologists. Medicare clinical laboratory fee schedule. Published November 2017. Accessed March 3, 2026. https://documents.cap.org/documents/2018-final-medicare-clfs-rates.pdf
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/71270/
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/74177/
- National Cancer Institute. Cancer trends progress report: financial burden of cancer care. Updated April 2025. Accessed March 3, 2026. https://progressreport.cancer.gov/after/economic_burden
- Bove FJ, Greek A, Gatiba R, et al. Cancer incidence among Marines and Navy personnel and civilian workers exposed to industrial solvents in drinking water at US Marine Corps Base Camp Lejeune: a cohort study. Environ Health Perspect. 2024;132:107008. doi:10.1289/EHP14966
- Maslia ML, Aral MM, Ruckart PZ, Bove FJ. Reconstructing historical VOC concentrations in drinking water for epidemiological studies at a US military base: summary of results. Water (Basel). 2016;8:449. doi:10.3390/w8100449
- Rosenfeld PE, Spaeth KR, McCarthy SJ, et al. Camp Lejeune Marine cancer risk assessment for exposure to contaminated drinking water from 1955 to 1987. Water Air Soil Pollut. 2024;235(2). doi:10.1007/s11270-023-06863-y
- US Department of Veterans Affairs, Veterans Health Administration. Camp Lejeune: past water contamination. Updated April 15, 2025. Accessed March 3, 2026. https://www.publichealth.va.gov/exposures/camp-lejeune/
- Jung K, Khan A, Mocharnuk R, et al. Clinical encounter with three cancer patients affected by groundwater contamination at Camp Lejeune: a case series and review of the literature. J Med Case Rep. 2022;16(1):272. doi:10.1186/s13256-022-03501-9
- Honoring America's Veterans and Caring for Camp Lejeune Familes Act of 2012, Pub L No. 112-154. Janey Ensminger Act. Congress.gov. Accessed April 15, 2026. https://ww.congress.gov/bill/112th-congress/house-bill/1627
- Bove FJ, Greek A, Gatiba R, et al. Evaluation of mortality among Marines, Navy personnel, and civilian workers exposed to contaminated drinking water at USMC Base Camp Lejeune: a cohort study. Environ Health. 2024;23(1):61. doi:10.1186/s12940-024-01099-7
- Honoring our PACT Act of 2022 (Pub L No. 117-168): expansion of health care eligibility and toxic exposure screenings. Congress.gov. Accessed March 3, 2026. https://www.congress.gov/bill/117th-congress/house-bill/3967
- Ensuring Justice for Camp Lejeune Victims Act of 2025. Congress.gov. Accessed March 24, 2026. https://www.congress.gov/bill/119th-congress/house-bill/4145
- SEER. Cancer stat facts: bladder cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/urinb.html
- Agency for Toxic Substances and Disease Registry. ATSDR assessment of the evidence for the drinking water contaminants at Camp Lejeune and specific cancers and other diseases. Published January 13, 2017. Accessed March 3, 2026. https://www.atsdr.cdc.gov/camp-lejeune/media/pdfs/2024/10/ATSDR_summary_of_the_evidence_for_causality_TCE_PCE_508.pdf
- National Cancer Institute. What is bladder cancer? Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder
- National Cancer Institute. Bladder cancer symptoms. Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/symptoms
- American Cancer Society. Bladder cancer signs and symptoms. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- National Cancer Institute. Bladder cancer screening. Updated April 27, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/screening
- American Cancer Society. Tests for bladder cancer. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: kidney and renal pelvis cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/kidrp.html
- American Cancer Society. Kidney cancer signs and symptoms. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for kidney cancer. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: esophageal cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/esoph.html
- Engel LS, Chow WH, Vaughan TL, et al. Population attributable risks of esophageal and gastric cancers.
J Natl Cancer Inst. 2003;95(18):1404-1413. doi:10.1093/jnci/djg047 - American Cancer Society. Signs and symptoms of esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: liver and intrahepatic bile duct cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/livibd.html
- American Cancer Society. Signs and symptoms of liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. What is liver cancer? Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer
- American Cancer Society. Tests for liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/how-diagnosed.html
- National Cancer Institute. Liver cancer screening. Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer/screening
- SEER. Cancer stat facts: lung cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/lungb.html
- US Department of Health and Human Services. The Health Consequences of Smoking: A Report of the Surgeon General. Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2004. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK44695/pdf/Bookshelf_NBK44695.pdf
- American Cancer Society. Lung cancer signs and symptoms. Updated February 27, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for lung cancer. Updated January 29, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: female breast cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/breast.html
- SEER. SEER*Explorer breast incidence and mortality comparison. Updated July 2, 2025. Accessed March 3, 2026. https://seer.cancer.gov/statistics-network/explorer/application.html
- Susan G. Komen. Male breast cancer. Updated June 3, 2025. Accessed March 3, 2026. https://www.komen.org/breast-cancer/facts-statistics/male-breast-cancer/
- American Cancer Society. Key statistics for breast cancer in men. Updated January 16, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/about/key-statistics.html
- Ruckart PZ, Bove FJ, Shanley E 3rd, et al. Evaluation of contaminated drinking water and male breast cancer at Marine Corps Base Camp Lejeune, North Carolina: a case-control study. Environ Health. 2015;14:74. doi:10.1186/s12940-015-0061-4
- Culell P, Solernou L, Tarazona J, et al. Male breast cancer: a multicentric study. Breast J. 2007;13:213-215. doi:10.1111/j.1524-4741.2007.00412.x
- Giordano SH, Cohen DS, Buzdar AU, et al. Breast carcinoma in men: a population-based study. Cancer. 2004;101:51-57. doi:10.1002/cncr.20312
- American Cancer Society. Signs and symptoms of breast cancer in men. Updated April 27, 2018. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests to help diagnose breast cancer in men. Updated December 20, 2021. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/how-diagnosed.html
- Evans GF, Anthony T, Turnage RH, et al. The diagnostic accuracy of mammography in the evaluation of male breast disease. Am J Surg. 2001;181:96-100. doi:10.1016/S0002-9610(00)00571-7
- SEER. Cancer stat facts: leukemia. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/leuks.html
- American Cancer Society. Signs and symptoms of acute myeloid leukemia (AML). Updated March 20, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Signs and symptoms of chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for acute myeloid leukemia (AML). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/how-diagnosed.html
- American Cancer Society. Tests for chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/how-diagnosed.html
- Giudice V, Selleri C. Aplastic anemia: pathophysiology. Semin Hematol. 2022;59:13-20. doi:10.1053/j.seminhematol.2021.12.002
- National Cancer Institute. Myelodysplastic syndromes treatment (PDQ®)–patient version. Updated October 4, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/myeloproliferative/patient/myelodysplastic-treatment-pdq
- SEER. Cancer statistics review (CSR) 1975-2013: myelodysplastic syndromes. Accessed March 3, 2026. https://seer.cancer.gov/archive/csr/1975_2013/browse_csr.php?sectionSEL=30&pageSEL=sect_30_table.01
- American Cancer Society. Signs and symptoms of myelodysplastic syndrome (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for myelodysplastic syndromes (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: myeloma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/mulmy.html
- American Cancer Society. Signs and symptoms of multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/testing.html
- Laubach JP. Multiple myeloma: clinical features, laboratory manifestations, and diagnosis. UpToDate. Updated April 28, 2025. Accessed March 3, 2026. https://www.uptodate.com/contents/multiple-myeloma-clinical-features-laboratory-manifestations-and-diagnosis
- Hillengass J, Usmani S, Rajkumar SV, et al. International Myeloma Working Group consensus recommendations on imaging in monoclonal plasma cell disorders. Lancet Oncol. 2019;20:e302-e312. doi:10.1016/S1470-2045(19)30309-2
- SEER. Cancer stat facts: non-Hodgkin lymphoma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/nhl.html
- American Cancer Society. Signs and symptoms of non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. Non-Hodgkin lymphoma treatment (PDQ®)–patient version. Updated August 22, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/lymphoma/patient/adult-nhl-treatment-pdq
- American Cancer Society. Tests for non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/how-diagnosed.html
- College of American Pathologists. Medicare clinical laboratory fee schedule. Published November 2017. Accessed March 3, 2026. https://documents.cap.org/documents/2018-final-medicare-clfs-rates.pdf
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/71270/
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/74177/
- National Cancer Institute. Cancer trends progress report: financial burden of cancer care. Updated April 2025. Accessed March 3, 2026. https://progressreport.cancer.gov/after/economic_burden
Optimizing Care for Veterans at Risk of Cancer From Camp Lejeune Water Exposure
Optimizing Care for Veterans at Risk of Cancer From Camp Lejeune Water Exposure
'An Organization in Turmoil': Ken Kizer on the Challenges Facing the VA
'An Organization in Turmoil': Ken Kizer on the Challenges Facing the VA
Kenneth W. Kizer, MD, MPH, knows a thing or two about transition at the US Department of Veterans Affairs (VA). He served as VA Under Secretary of Health from 1994 to 1999, stepping in during an era of crisis with a mandate for transformation.
Kizer, a Distinguished Professor Emeritus at the University of California, Davis School of Medicine, is among the top thinkers about the VA and its future. He recently spoke with Federal Practitioner about community care, the electronic health record transition, and other challenges facing the Veterans Health Administration (VHA).
At stake, Kizer explained, is an invaluable service for veterans—and much more. “VA is the largest provider of training for... multiple types of health professionals that people use every day,” he said. “There’s also the research, the direct care provided to veterans, and the contingency support the VA provides, which was very well demonstrated during the COVID pandemic. These are things that benefit all Americans, not just veterans.”
When you look at the VA, what do you see?
I see an organization in turmoil, a great health care system struggling with multiple major challenges simultaneously. The VHA is becoming a very large health insurance program without the necessary infrastructure, and costs are rising rapidly. And it is trying to roll out a new EHR and implement new third-party administrator contracts while suffering from significant staffing reductions and very depressed morale.
There are a host of other high-visibility and high-impact issues, including a major reorganization. There’s been a paucity of details about exactly what is going to change, who is going to be doing what, and how the changes will affect staffing and workflow.
How will the loss of 35,000 health care positions affect veterans' care?
If you don’t have enough people, then you’re not going to be able to provide the care that is needed. Years ago, I led a project assessing the Roseburg VA Medical Center in Central Oregon. Among other things, there were a lot of problems with cardiology care. The biggest complaint the cardiologists had, and why the hospital couldn’t keep cardiologists on staff, was that there weren’t enough support staff to do the electrocardiograms. The cardiologists had to do the electrocardiograms themselves, which meant they weren’t doing other things they should be doing. You can amplify that example in a hundred different ways in VA today. If physicians don’t have adequate support, they get frustrated and disenfranchised. And they leave.
One of the fallacies I’ve heard mentioned in some congressional hearings is that it’s mainly a matter of lower pay in the VA. Pay may be an issue somewhere on the list of recruiting challenges, but more important issues higher up are things like the support clinicians receive, the work environment, whether they feel valued, and how easy it is for them to do their work. Case in point: If you put in a new EHR that doesn’t work as well as the existing one, then some doctors are going to leave.
Is VA being pushed toward privatization?
At some point it becomes a self-fulfilling prophecy. If you don’t have the staff to provide the services, then you refer more veterans to the community, and you get in a downward spiral. Patients are going to the community, you lose more staff, you continue to be unable to provide services, and more care goes to the community.
A part of this equation that hasn’t been given adequate attention is VA’s teaching mission. If care is increasingly going to the community, those patients won’t be available for the trainees in teaching programs. That in turn impacts the pipeline of clinicians who will be available to serve the population at large. The negative effects will be seen far beyond the VA.
Why have you expressed concern over VA care fragmentation?1
Greater than 80% of VA ICU [intensive care unit] care is now being provided in community hospitals. When patients are discharged from those hospitals, they often continue getting follow-up care in the community because VA doesn’t have good mechanisms to reconnect those patients back to VA care.2
[Other researchers] found that the majority of emergency department care for enrolled veterans in New York State was being paid for by entities other than the VA, most commonly Medicare but also Medicaid and private insurance. Where follow-up care occurred often depended on who paid for the emergency department visit, not necessarily what was best for the patient.3
The core problem is that the VA has very little insight into what’s happening when its enrollees get care that is paid for by another payer. VA doesn’t know when their patients are in a private hospital emergency department, so they can’t reach out in real time, and they can’t reconnect with them afterward.
That is very different than for commercial health plans. They know when one of their enrollees is admitted to an out-of-network hospital, and when they are discharged, and they follow up immediately. VA doesn’t have the infrastructure in place to do that.
Why did the VA spend $44 billion on Medicare Advantage double-payments from 2018 to 2021?4
That number is much larger now—$87 billion from 2019 to 2023. Here’s the problem: When VA enrollees are also enrolled in a Medicare Advantage plan, the Medicare plan gets paid to provide the care for those veterans. But when those enrollees come to the VA, the VA provides and pays for the care but cannot bill Medicare for the costs. So the federal government ends up paying twice for care of the same person.
In a paper I coauthored last December we showed that in 2023 alone VA spent $23 billion for care of veterans enrolled in Medicare Advantage plan. Those duplicative payments accounted for almost 20% of VA's entire medical care budget.5
How can fragmented care be reduced?
Two things really stand out. First, real-time health insurance data sharing across payers is foundational. VA has to know when its patients get care by non-VA providers if it is going to coordinate and provide follow-up care. As a first step, VA and the Centers for Medicare & Medicaid Services need to create a data sharing platform for veterans dually enrolled in VA and Medicare or Medicaid.
This is not a new idea. I tried to do it when I was Under Secretary for Health in the late 1990s, but it never happened for various political reasons. Others have tried since. Maybe now, given how much money is at stake, it will finally get done.
Second, the VA needs to implement rigorous case management for high utilizers. The costs are not evenly distributed across enrollees. Approximately 10% of community care users account for almost 90% of community care expenditures. Common sense says you should intensely manage the care of those high-need patients who account for so much of the costs, try to avoid out-of-network ICU and emergency department care as much as possible, and build relationships with other providers so there are clear mechanisms to reconnect those patients back to VA care after an acute episode is treated outside the VA health system.
Is community care itself the problem?
No. Community care is a good thing for many veterans. It has increased access and made it easier for enrolled veterans to get care in some situations. The problem is that the VA hasn’t built in the mechanisms and processes to share information, manage complex patients, provide follow-up care, or oversee quality in community care.
Historically, VA has been an integrated delivery system that provided the overwhelming majority of care within its own facilities. However, over the last decade it has become a hybrid purchaser-provider system. It has become a very large purchaser of non-VA care, going from about $7 billion to $50 billion in community care spending over the past decade. But the VA hasn’t built the infrastructure—information exchange, case management, utilization review, quality oversight—that a hybrid purchaser-provider system needs to be a prudent purchaser.
What is your perspective on VHA's EHR transition?
The many problems with the rollout of the Oracle/Cerner EHR have been well-documented by the Inspector General, frontline clinicians, and others. The problems have been so bad that implementation has been halted a couple times. They’re now moving forward again, but it remains to be seen whether the problems truly have been fixed.
Still unaddressed is the more fundamental question of whether VistA could have been upgraded and modernized at far less cost and disruption of care. No thorough, deliberative analysis of that was ever done. And some of the ostensible problems with upgrading VistA in years past are no longer an issue.
Given the challenges VA faces, are you optimistic about its future?
While there definitely are problems, they are all solvable. Every challenge the VA is facing can be addressed. The question is when and how, and whether the VA is going to be given a fair chance to work through its challenges.
As for those who look to the private sector and think that’s the solution: They haven’t looked closely enough. The private sector is also struggling with staffing and financing issues, many of the same issues VA is dealing with, just in a somewhat different way. The problems in the private sector will be an increasing challenge for community care going forward.
Overall, my life experience is that dark times are always followed by daylight, so I am confident there are brighter days ahead for VA.
1. Kizer KW. Curbing the growing fragmentation of veterans’ health care. JAMA Health Forum. 2025;6:e254148. doi:10.1001/jamahealthforum.2025.4148
2. Hahn Z, Naiditch H, Talisa V, et al. Intensive care unit admissions purchased or delivered by veterans in the VA health care system. JAMA Health Forum. 2025;6:e255605. doi:10.1001/jamahealthforum.2025.5605
3. Vashi AA, Urech T, Wu S, Asch S. Fragmented financing in emergency department use among US veterans. JAMA Health Forum. 2025;6:e255635. doi:10.1001/jamahealthforum.2025.5635
4. Maremont M, Weaver C, McGinty T. Insurers collected billions from medicare for veterans who cost them almost nothing. The Wall Street Journal. December 2, 2024. Accessed March 17, 2026. https://www.wsj.com/health/healthcare /veterans-medicare-insurers-collect-billions-bfd47d27
5. Trivedi AN, Jiang L, Meyers DJ, et al. Spending by the Veterans Affairs health care system for Medicare Advantage Enrollees. JAMA Health Forum. 2025;6:e255653. doi:10.1001/jamahealthforum.2025.5653
Kenneth W. Kizer, MD, MPH, knows a thing or two about transition at the US Department of Veterans Affairs (VA). He served as VA Under Secretary of Health from 1994 to 1999, stepping in during an era of crisis with a mandate for transformation.
Kizer, a Distinguished Professor Emeritus at the University of California, Davis School of Medicine, is among the top thinkers about the VA and its future. He recently spoke with Federal Practitioner about community care, the electronic health record transition, and other challenges facing the Veterans Health Administration (VHA).
At stake, Kizer explained, is an invaluable service for veterans—and much more. “VA is the largest provider of training for... multiple types of health professionals that people use every day,” he said. “There’s also the research, the direct care provided to veterans, and the contingency support the VA provides, which was very well demonstrated during the COVID pandemic. These are things that benefit all Americans, not just veterans.”
When you look at the VA, what do you see?
I see an organization in turmoil, a great health care system struggling with multiple major challenges simultaneously. The VHA is becoming a very large health insurance program without the necessary infrastructure, and costs are rising rapidly. And it is trying to roll out a new EHR and implement new third-party administrator contracts while suffering from significant staffing reductions and very depressed morale.
There are a host of other high-visibility and high-impact issues, including a major reorganization. There’s been a paucity of details about exactly what is going to change, who is going to be doing what, and how the changes will affect staffing and workflow.
How will the loss of 35,000 health care positions affect veterans' care?
If you don’t have enough people, then you’re not going to be able to provide the care that is needed. Years ago, I led a project assessing the Roseburg VA Medical Center in Central Oregon. Among other things, there were a lot of problems with cardiology care. The biggest complaint the cardiologists had, and why the hospital couldn’t keep cardiologists on staff, was that there weren’t enough support staff to do the electrocardiograms. The cardiologists had to do the electrocardiograms themselves, which meant they weren’t doing other things they should be doing. You can amplify that example in a hundred different ways in VA today. If physicians don’t have adequate support, they get frustrated and disenfranchised. And they leave.
One of the fallacies I’ve heard mentioned in some congressional hearings is that it’s mainly a matter of lower pay in the VA. Pay may be an issue somewhere on the list of recruiting challenges, but more important issues higher up are things like the support clinicians receive, the work environment, whether they feel valued, and how easy it is for them to do their work. Case in point: If you put in a new EHR that doesn’t work as well as the existing one, then some doctors are going to leave.
Is VA being pushed toward privatization?
At some point it becomes a self-fulfilling prophecy. If you don’t have the staff to provide the services, then you refer more veterans to the community, and you get in a downward spiral. Patients are going to the community, you lose more staff, you continue to be unable to provide services, and more care goes to the community.
A part of this equation that hasn’t been given adequate attention is VA’s teaching mission. If care is increasingly going to the community, those patients won’t be available for the trainees in teaching programs. That in turn impacts the pipeline of clinicians who will be available to serve the population at large. The negative effects will be seen far beyond the VA.
Why have you expressed concern over VA care fragmentation?1
Greater than 80% of VA ICU [intensive care unit] care is now being provided in community hospitals. When patients are discharged from those hospitals, they often continue getting follow-up care in the community because VA doesn’t have good mechanisms to reconnect those patients back to VA care.2
[Other researchers] found that the majority of emergency department care for enrolled veterans in New York State was being paid for by entities other than the VA, most commonly Medicare but also Medicaid and private insurance. Where follow-up care occurred often depended on who paid for the emergency department visit, not necessarily what was best for the patient.3
The core problem is that the VA has very little insight into what’s happening when its enrollees get care that is paid for by another payer. VA doesn’t know when their patients are in a private hospital emergency department, so they can’t reach out in real time, and they can’t reconnect with them afterward.
That is very different than for commercial health plans. They know when one of their enrollees is admitted to an out-of-network hospital, and when they are discharged, and they follow up immediately. VA doesn’t have the infrastructure in place to do that.
Why did the VA spend $44 billion on Medicare Advantage double-payments from 2018 to 2021?4
That number is much larger now—$87 billion from 2019 to 2023. Here’s the problem: When VA enrollees are also enrolled in a Medicare Advantage plan, the Medicare plan gets paid to provide the care for those veterans. But when those enrollees come to the VA, the VA provides and pays for the care but cannot bill Medicare for the costs. So the federal government ends up paying twice for care of the same person.
In a paper I coauthored last December we showed that in 2023 alone VA spent $23 billion for care of veterans enrolled in Medicare Advantage plan. Those duplicative payments accounted for almost 20% of VA's entire medical care budget.5
How can fragmented care be reduced?
Two things really stand out. First, real-time health insurance data sharing across payers is foundational. VA has to know when its patients get care by non-VA providers if it is going to coordinate and provide follow-up care. As a first step, VA and the Centers for Medicare & Medicaid Services need to create a data sharing platform for veterans dually enrolled in VA and Medicare or Medicaid.
This is not a new idea. I tried to do it when I was Under Secretary for Health in the late 1990s, but it never happened for various political reasons. Others have tried since. Maybe now, given how much money is at stake, it will finally get done.
Second, the VA needs to implement rigorous case management for high utilizers. The costs are not evenly distributed across enrollees. Approximately 10% of community care users account for almost 90% of community care expenditures. Common sense says you should intensely manage the care of those high-need patients who account for so much of the costs, try to avoid out-of-network ICU and emergency department care as much as possible, and build relationships with other providers so there are clear mechanisms to reconnect those patients back to VA care after an acute episode is treated outside the VA health system.
Is community care itself the problem?
No. Community care is a good thing for many veterans. It has increased access and made it easier for enrolled veterans to get care in some situations. The problem is that the VA hasn’t built in the mechanisms and processes to share information, manage complex patients, provide follow-up care, or oversee quality in community care.
Historically, VA has been an integrated delivery system that provided the overwhelming majority of care within its own facilities. However, over the last decade it has become a hybrid purchaser-provider system. It has become a very large purchaser of non-VA care, going from about $7 billion to $50 billion in community care spending over the past decade. But the VA hasn’t built the infrastructure—information exchange, case management, utilization review, quality oversight—that a hybrid purchaser-provider system needs to be a prudent purchaser.
What is your perspective on VHA's EHR transition?
The many problems with the rollout of the Oracle/Cerner EHR have been well-documented by the Inspector General, frontline clinicians, and others. The problems have been so bad that implementation has been halted a couple times. They’re now moving forward again, but it remains to be seen whether the problems truly have been fixed.
Still unaddressed is the more fundamental question of whether VistA could have been upgraded and modernized at far less cost and disruption of care. No thorough, deliberative analysis of that was ever done. And some of the ostensible problems with upgrading VistA in years past are no longer an issue.
Given the challenges VA faces, are you optimistic about its future?
While there definitely are problems, they are all solvable. Every challenge the VA is facing can be addressed. The question is when and how, and whether the VA is going to be given a fair chance to work through its challenges.
As for those who look to the private sector and think that’s the solution: They haven’t looked closely enough. The private sector is also struggling with staffing and financing issues, many of the same issues VA is dealing with, just in a somewhat different way. The problems in the private sector will be an increasing challenge for community care going forward.
Overall, my life experience is that dark times are always followed by daylight, so I am confident there are brighter days ahead for VA.
Kenneth W. Kizer, MD, MPH, knows a thing or two about transition at the US Department of Veterans Affairs (VA). He served as VA Under Secretary of Health from 1994 to 1999, stepping in during an era of crisis with a mandate for transformation.
Kizer, a Distinguished Professor Emeritus at the University of California, Davis School of Medicine, is among the top thinkers about the VA and its future. He recently spoke with Federal Practitioner about community care, the electronic health record transition, and other challenges facing the Veterans Health Administration (VHA).
At stake, Kizer explained, is an invaluable service for veterans—and much more. “VA is the largest provider of training for... multiple types of health professionals that people use every day,” he said. “There’s also the research, the direct care provided to veterans, and the contingency support the VA provides, which was very well demonstrated during the COVID pandemic. These are things that benefit all Americans, not just veterans.”
When you look at the VA, what do you see?
I see an organization in turmoil, a great health care system struggling with multiple major challenges simultaneously. The VHA is becoming a very large health insurance program without the necessary infrastructure, and costs are rising rapidly. And it is trying to roll out a new EHR and implement new third-party administrator contracts while suffering from significant staffing reductions and very depressed morale.
There are a host of other high-visibility and high-impact issues, including a major reorganization. There’s been a paucity of details about exactly what is going to change, who is going to be doing what, and how the changes will affect staffing and workflow.
How will the loss of 35,000 health care positions affect veterans' care?
If you don’t have enough people, then you’re not going to be able to provide the care that is needed. Years ago, I led a project assessing the Roseburg VA Medical Center in Central Oregon. Among other things, there were a lot of problems with cardiology care. The biggest complaint the cardiologists had, and why the hospital couldn’t keep cardiologists on staff, was that there weren’t enough support staff to do the electrocardiograms. The cardiologists had to do the electrocardiograms themselves, which meant they weren’t doing other things they should be doing. You can amplify that example in a hundred different ways in VA today. If physicians don’t have adequate support, they get frustrated and disenfranchised. And they leave.
One of the fallacies I’ve heard mentioned in some congressional hearings is that it’s mainly a matter of lower pay in the VA. Pay may be an issue somewhere on the list of recruiting challenges, but more important issues higher up are things like the support clinicians receive, the work environment, whether they feel valued, and how easy it is for them to do their work. Case in point: If you put in a new EHR that doesn’t work as well as the existing one, then some doctors are going to leave.
Is VA being pushed toward privatization?
At some point it becomes a self-fulfilling prophecy. If you don’t have the staff to provide the services, then you refer more veterans to the community, and you get in a downward spiral. Patients are going to the community, you lose more staff, you continue to be unable to provide services, and more care goes to the community.
A part of this equation that hasn’t been given adequate attention is VA’s teaching mission. If care is increasingly going to the community, those patients won’t be available for the trainees in teaching programs. That in turn impacts the pipeline of clinicians who will be available to serve the population at large. The negative effects will be seen far beyond the VA.
Why have you expressed concern over VA care fragmentation?1
Greater than 80% of VA ICU [intensive care unit] care is now being provided in community hospitals. When patients are discharged from those hospitals, they often continue getting follow-up care in the community because VA doesn’t have good mechanisms to reconnect those patients back to VA care.2
[Other researchers] found that the majority of emergency department care for enrolled veterans in New York State was being paid for by entities other than the VA, most commonly Medicare but also Medicaid and private insurance. Where follow-up care occurred often depended on who paid for the emergency department visit, not necessarily what was best for the patient.3
The core problem is that the VA has very little insight into what’s happening when its enrollees get care that is paid for by another payer. VA doesn’t know when their patients are in a private hospital emergency department, so they can’t reach out in real time, and they can’t reconnect with them afterward.
That is very different than for commercial health plans. They know when one of their enrollees is admitted to an out-of-network hospital, and when they are discharged, and they follow up immediately. VA doesn’t have the infrastructure in place to do that.
Why did the VA spend $44 billion on Medicare Advantage double-payments from 2018 to 2021?4
That number is much larger now—$87 billion from 2019 to 2023. Here’s the problem: When VA enrollees are also enrolled in a Medicare Advantage plan, the Medicare plan gets paid to provide the care for those veterans. But when those enrollees come to the VA, the VA provides and pays for the care but cannot bill Medicare for the costs. So the federal government ends up paying twice for care of the same person.
In a paper I coauthored last December we showed that in 2023 alone VA spent $23 billion for care of veterans enrolled in Medicare Advantage plan. Those duplicative payments accounted for almost 20% of VA's entire medical care budget.5
How can fragmented care be reduced?
Two things really stand out. First, real-time health insurance data sharing across payers is foundational. VA has to know when its patients get care by non-VA providers if it is going to coordinate and provide follow-up care. As a first step, VA and the Centers for Medicare & Medicaid Services need to create a data sharing platform for veterans dually enrolled in VA and Medicare or Medicaid.
This is not a new idea. I tried to do it when I was Under Secretary for Health in the late 1990s, but it never happened for various political reasons. Others have tried since. Maybe now, given how much money is at stake, it will finally get done.
Second, the VA needs to implement rigorous case management for high utilizers. The costs are not evenly distributed across enrollees. Approximately 10% of community care users account for almost 90% of community care expenditures. Common sense says you should intensely manage the care of those high-need patients who account for so much of the costs, try to avoid out-of-network ICU and emergency department care as much as possible, and build relationships with other providers so there are clear mechanisms to reconnect those patients back to VA care after an acute episode is treated outside the VA health system.
Is community care itself the problem?
No. Community care is a good thing for many veterans. It has increased access and made it easier for enrolled veterans to get care in some situations. The problem is that the VA hasn’t built in the mechanisms and processes to share information, manage complex patients, provide follow-up care, or oversee quality in community care.
Historically, VA has been an integrated delivery system that provided the overwhelming majority of care within its own facilities. However, over the last decade it has become a hybrid purchaser-provider system. It has become a very large purchaser of non-VA care, going from about $7 billion to $50 billion in community care spending over the past decade. But the VA hasn’t built the infrastructure—information exchange, case management, utilization review, quality oversight—that a hybrid purchaser-provider system needs to be a prudent purchaser.
What is your perspective on VHA's EHR transition?
The many problems with the rollout of the Oracle/Cerner EHR have been well-documented by the Inspector General, frontline clinicians, and others. The problems have been so bad that implementation has been halted a couple times. They’re now moving forward again, but it remains to be seen whether the problems truly have been fixed.
Still unaddressed is the more fundamental question of whether VistA could have been upgraded and modernized at far less cost and disruption of care. No thorough, deliberative analysis of that was ever done. And some of the ostensible problems with upgrading VistA in years past are no longer an issue.
Given the challenges VA faces, are you optimistic about its future?
While there definitely are problems, they are all solvable. Every challenge the VA is facing can be addressed. The question is when and how, and whether the VA is going to be given a fair chance to work through its challenges.
As for those who look to the private sector and think that’s the solution: They haven’t looked closely enough. The private sector is also struggling with staffing and financing issues, many of the same issues VA is dealing with, just in a somewhat different way. The problems in the private sector will be an increasing challenge for community care going forward.
Overall, my life experience is that dark times are always followed by daylight, so I am confident there are brighter days ahead for VA.
1. Kizer KW. Curbing the growing fragmentation of veterans’ health care. JAMA Health Forum. 2025;6:e254148. doi:10.1001/jamahealthforum.2025.4148
2. Hahn Z, Naiditch H, Talisa V, et al. Intensive care unit admissions purchased or delivered by veterans in the VA health care system. JAMA Health Forum. 2025;6:e255605. doi:10.1001/jamahealthforum.2025.5605
3. Vashi AA, Urech T, Wu S, Asch S. Fragmented financing in emergency department use among US veterans. JAMA Health Forum. 2025;6:e255635. doi:10.1001/jamahealthforum.2025.5635
4. Maremont M, Weaver C, McGinty T. Insurers collected billions from medicare for veterans who cost them almost nothing. The Wall Street Journal. December 2, 2024. Accessed March 17, 2026. https://www.wsj.com/health/healthcare /veterans-medicare-insurers-collect-billions-bfd47d27
5. Trivedi AN, Jiang L, Meyers DJ, et al. Spending by the Veterans Affairs health care system for Medicare Advantage Enrollees. JAMA Health Forum. 2025;6:e255653. doi:10.1001/jamahealthforum.2025.5653
1. Kizer KW. Curbing the growing fragmentation of veterans’ health care. JAMA Health Forum. 2025;6:e254148. doi:10.1001/jamahealthforum.2025.4148
2. Hahn Z, Naiditch H, Talisa V, et al. Intensive care unit admissions purchased or delivered by veterans in the VA health care system. JAMA Health Forum. 2025;6:e255605. doi:10.1001/jamahealthforum.2025.5605
3. Vashi AA, Urech T, Wu S, Asch S. Fragmented financing in emergency department use among US veterans. JAMA Health Forum. 2025;6:e255635. doi:10.1001/jamahealthforum.2025.5635
4. Maremont M, Weaver C, McGinty T. Insurers collected billions from medicare for veterans who cost them almost nothing. The Wall Street Journal. December 2, 2024. Accessed March 17, 2026. https://www.wsj.com/health/healthcare /veterans-medicare-insurers-collect-billions-bfd47d27
5. Trivedi AN, Jiang L, Meyers DJ, et al. Spending by the Veterans Affairs health care system for Medicare Advantage Enrollees. JAMA Health Forum. 2025;6:e255653. doi:10.1001/jamahealthforum.2025.5653
'An Organization in Turmoil': Ken Kizer on the Challenges Facing the VA
'An Organization in Turmoil': Ken Kizer on the Challenges Facing the VA
Cell-Free DNA Blood Test Shows Strong Performance in Detecting Early-Stage CRC
Cell-Free DNA Blood Test Shows Strong Performance in Detecting Early-Stage CRC
TOPLINE:
A novel, blood-based test developed using fragmentomic features of cell-free DNA (cfDNA) detects colorectal cancer (CRC) with a 90.4% sensitivity and shows consistent performance across stages and tumor locations.
METHODOLOGY:
- Researchers conducted a prospective case-control study to develop and validate a noninvasive cfDNA-based screening test for CRC.
- Adults aged 40-89 years with CRC or advanced adenomas were enrolled at a tertiary center in South Korea between 2021 and 2024.
- Blood samples were drawn after colonoscopy, but prior to treatment, in patients with CRC, advanced adenomas, and asymptomatic controls with normal colonoscopy results.
- A model was trained on fragmentonic features derived from whole genome sequencing of cfDNA from 1250 participants and validated for its diagnostic performance in the remaining 427 participants, including all with advanced adenomas.
- The primary endpoint was the sensitivity of the cfDNA test for detecting CRC. The area under the receiver operating characteristic curve (AUROC) was also calculated.
TAKEAWAY:
- The cfDNA test detected CRC with 90.4% sensitivity and an AUROC of 0.978.
- Sensitivity by CRC stage was 84.2% for stage I, 85.0% for stage II, 94.4% for stage III, 100% for stage IV.
- Advanced adenomas were detected with 58.3% sensitivity and an AUROC of 0.862.
- Among individuals with normal colonoscopy findings, the test was correctly negative 94.7% of the time.
- Diagnostic sensitivities were consistent between left- and right-sided CRC tumors, among participants aged < 60 years and ≥ 60 years, and across left- and right-sided advanced adenomas.
IN PRACTICE:
"This highlights the potential clinical utility of the test in identifying candidates for minimally invasive therapeutic approaches tool for CRC," the authors wrote. "Notably, the high sensitivity observed for early-stage CRC and the favorable sensitivity for [advanced adenoma] suggest that this cfDNA test may offer benefits not only in diagnosis but also in prognosis and ultimately in CRC prevention."
SOURCE:
This study was led by Seung Wook Hong, MD, Asan Medical Center in Seoul, South Korea. It was published online on November 19, 2025, in the American Journal of Gastroenterology.
LIMITATIONS:
The case-control design introduced spectrum bias by comparing clearly defined CRC and advanced adenomas cases with individuals who had normal colonoscopy results. The CRC prevalence of 17% to 18% was higher than that observed in true screening populations, limiting generalizability. The exclusively Korean cohort limited extrapolation to non-Asian populations.
DISCLOSURES:
The study received support from GC Genome, Yongin, South Korea. The authors reported no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
A novel, blood-based test developed using fragmentomic features of cell-free DNA (cfDNA) detects colorectal cancer (CRC) with a 90.4% sensitivity and shows consistent performance across stages and tumor locations.
METHODOLOGY:
- Researchers conducted a prospective case-control study to develop and validate a noninvasive cfDNA-based screening test for CRC.
- Adults aged 40-89 years with CRC or advanced adenomas were enrolled at a tertiary center in South Korea between 2021 and 2024.
- Blood samples were drawn after colonoscopy, but prior to treatment, in patients with CRC, advanced adenomas, and asymptomatic controls with normal colonoscopy results.
- A model was trained on fragmentonic features derived from whole genome sequencing of cfDNA from 1250 participants and validated for its diagnostic performance in the remaining 427 participants, including all with advanced adenomas.
- The primary endpoint was the sensitivity of the cfDNA test for detecting CRC. The area under the receiver operating characteristic curve (AUROC) was also calculated.
TAKEAWAY:
- The cfDNA test detected CRC with 90.4% sensitivity and an AUROC of 0.978.
- Sensitivity by CRC stage was 84.2% for stage I, 85.0% for stage II, 94.4% for stage III, 100% for stage IV.
- Advanced adenomas were detected with 58.3% sensitivity and an AUROC of 0.862.
- Among individuals with normal colonoscopy findings, the test was correctly negative 94.7% of the time.
- Diagnostic sensitivities were consistent between left- and right-sided CRC tumors, among participants aged < 60 years and ≥ 60 years, and across left- and right-sided advanced adenomas.
IN PRACTICE:
"This highlights the potential clinical utility of the test in identifying candidates for minimally invasive therapeutic approaches tool for CRC," the authors wrote. "Notably, the high sensitivity observed for early-stage CRC and the favorable sensitivity for [advanced adenoma] suggest that this cfDNA test may offer benefits not only in diagnosis but also in prognosis and ultimately in CRC prevention."
SOURCE:
This study was led by Seung Wook Hong, MD, Asan Medical Center in Seoul, South Korea. It was published online on November 19, 2025, in the American Journal of Gastroenterology.
LIMITATIONS:
The case-control design introduced spectrum bias by comparing clearly defined CRC and advanced adenomas cases with individuals who had normal colonoscopy results. The CRC prevalence of 17% to 18% was higher than that observed in true screening populations, limiting generalizability. The exclusively Korean cohort limited extrapolation to non-Asian populations.
DISCLOSURES:
The study received support from GC Genome, Yongin, South Korea. The authors reported no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
A novel, blood-based test developed using fragmentomic features of cell-free DNA (cfDNA) detects colorectal cancer (CRC) with a 90.4% sensitivity and shows consistent performance across stages and tumor locations.
METHODOLOGY:
- Researchers conducted a prospective case-control study to develop and validate a noninvasive cfDNA-based screening test for CRC.
- Adults aged 40-89 years with CRC or advanced adenomas were enrolled at a tertiary center in South Korea between 2021 and 2024.
- Blood samples were drawn after colonoscopy, but prior to treatment, in patients with CRC, advanced adenomas, and asymptomatic controls with normal colonoscopy results.
- A model was trained on fragmentonic features derived from whole genome sequencing of cfDNA from 1250 participants and validated for its diagnostic performance in the remaining 427 participants, including all with advanced adenomas.
- The primary endpoint was the sensitivity of the cfDNA test for detecting CRC. The area under the receiver operating characteristic curve (AUROC) was also calculated.
TAKEAWAY:
- The cfDNA test detected CRC with 90.4% sensitivity and an AUROC of 0.978.
- Sensitivity by CRC stage was 84.2% for stage I, 85.0% for stage II, 94.4% for stage III, 100% for stage IV.
- Advanced adenomas were detected with 58.3% sensitivity and an AUROC of 0.862.
- Among individuals with normal colonoscopy findings, the test was correctly negative 94.7% of the time.
- Diagnostic sensitivities were consistent between left- and right-sided CRC tumors, among participants aged < 60 years and ≥ 60 years, and across left- and right-sided advanced adenomas.
IN PRACTICE:
"This highlights the potential clinical utility of the test in identifying candidates for minimally invasive therapeutic approaches tool for CRC," the authors wrote. "Notably, the high sensitivity observed for early-stage CRC and the favorable sensitivity for [advanced adenoma] suggest that this cfDNA test may offer benefits not only in diagnosis but also in prognosis and ultimately in CRC prevention."
SOURCE:
This study was led by Seung Wook Hong, MD, Asan Medical Center in Seoul, South Korea. It was published online on November 19, 2025, in the American Journal of Gastroenterology.
LIMITATIONS:
The case-control design introduced spectrum bias by comparing clearly defined CRC and advanced adenomas cases with individuals who had normal colonoscopy results. The CRC prevalence of 17% to 18% was higher than that observed in true screening populations, limiting generalizability. The exclusively Korean cohort limited extrapolation to non-Asian populations.
DISCLOSURES:
The study received support from GC Genome, Yongin, South Korea. The authors reported no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
Cell-Free DNA Blood Test Shows Strong Performance in Detecting Early-Stage CRC
Cell-Free DNA Blood Test Shows Strong Performance in Detecting Early-Stage CRC
Anticoagulation Stewardship Efforts Via Indication Reviews at a Veterans Affairs Health Care System
Anticoagulation Stewardship Efforts Via Indication Reviews at a Veterans Affairs Health Care System
Due to the underlying mechanism of atrial fibrillation (Afib), clots can form within the left atrial appendage. Clots that become dislodged may lead to ischemic stroke and possibly death. The 2023 guidelines for atrial fibrillation from the American College of Cardiology and American Heart Association recommend anticoagulation therapy for patients with an Afib diagnosis and a CHA2DS2-VASc (congestive heart failure, hypertension, age ≥ 75 years, diabetes, stroke/vascular disease, age 65 to 74 years, and female sex) score pertinent for ≥ 1 non–sex-related factor (score ≥ 2 for women; ≥ 1 for men) to prevent stroke-related complications. The CHA2DS2-VASc score is a 9-point scoring tool based on comorbidities and conditions that increase risk of stroke in patients with Afib. Each value correlates to an annualized stroke risk percentage that increases as the score increases.
In clinical practice, patients meeting these thresholds are indicated for anticoagulation and are considered for indefinite use unless ≥ 1 of the following conditions are present: bleeding risk outweighs the stroke prevention benefit, Afib is episodic (< 48 hours) or a nonpharmacologic intervention, such as a left atrial appendage occlusion (LAAO) device is present.1
In patients with a diagnosed venous thromboembolism (VTE), such as deep vein thrombosis or pulmonary embolism, anticoagulation is used to treat the current thrombosis and prevent embolization that can ultimately lead to death. The 2021 guideline for VTE from the American College of Chest Physicians identifies certain risk factors that increase risk for VTE and categorizes them as transient or persistent. Transient risk factors include hospitalization > 3 days, major trauma, surgery, cast immobilization, hormone therapy, pregnancy, or prolonged travel > 8 hours. Persistent risk factors include malignancy, thrombophilia, and certain medications.
The guideline recommends therapy durations based on event frequency, the presence and classification of provoking risk factors, and bleeding risk. As the risk of recurrent thrombosis and other potential complications is greatest in the first 3 to 6 months after a diagnosed event, at least 3 months anticoagulation therapy is recommended following VTE diagnosis. At the 3-month mark, all regimens are suggested to be re-evaluated and considered for extended treatment duration if the event was unprovoked, recurrent, secondary to a persistent risk factor, or low bleed risk.2Anticoagulation is an important guideline-recommended pharmacologic intervention for various disease states, although its use is not without risks. The Institute for Safe Medication Practices has classified oral anticoagulants as high-alert medications. This designation was made because anticoagulant medications have the potential to cause harm when used or omitted in error and lead to life-threatening bleed or thrombotic complications.3Anticoagulation stewardship ensures that anticoagulation therapy is appropriately initiated, maintained, and discontinued when indicated. Because of the potential for harm, anticoagulation stewardship is an important part of Afib and VTE management. Pharmacists can help verify and evaluate anticoagulation therapies. Research suggests that pharmacist-led anticoagulation stewardship efforts may play a role in ensuring safer patient outcomes.4The purpose of this quality improvement (QI) study was to implement pharmacist-led anticoagulation stewardship practices at Veterans Affairs Phoenix Health Care System (VAPHCS) to identify veterans with Afib not currently on anticoagulation, as well as to identify veterans with a history of VTE events who have completed a sufficient treatment duration.
Methods
Anticoagulation stewardship efforts were implemented in 2 cohorts of patients: those with Afib who may be indicated to initiate anticoagulation, and those with a history of VTE events who may be indicated to consider anticoagulation discontinuation. Patient records were reviewed using a standardized note template, and recommendations to either initiate or discontinue anticoagulation therapy were documented. The VAPHCS Research Service reviewed this study and determined that it was not research and was exempt from institutional review board review.
Atrial Fibrillation Cohort
A population health dashboard created by the Stroke Prevention in Atrial Fibrillation/Flutter Targeting the uNTreated: a focus on health care disparities (SPAFF-TNT-D) national VA study team was used to identify veterans at VAPHCS with a diagnosis of Afib without an active VA prescription for an anticoagulant. The dashboard filtered and produced data points from the medical record that correlated to the components of the CHA2DS2-VASc score. All veterans identified by the dashboard with scores of 7 or 8 were included. No patients had a score of 9. Comprehensive chart reviews of available VA and non–VA-provided care records were conducted by the investigators, and a standardized note template designed by the SPAFF-TNT-D team (eAppendix 1) was used to document findings within the electronic health record (EHR). If anticoagulation was deemed to be indicated, the assigned primary care practitioner (PCP) as listed in the EHR was alerted to the note by the investigators for further evaluation and consideration of prescribing anticoagulation.
Venous Thromboembolism Cohort
VAPHCS pharmacy informatics pulled data that included veterans with documented VTE and an active VA anticoagulant prescription between November 2022 and November 2023. Veterans were reviewed in chronological order based on when the anticoagulant prescription was written. All veterans were included until an equal number of charts were reviewed in both the Afib and VTE cohorts. Comprehensive chart review of available VA- and non–VA-provided care records was conducted by the investigators, and a standardized note template as designed by the investigators (eAppendix 2) was used to document findings within the EHR. If the duration of anticoagulation therapy was deemed sufficient, the assigned anticoagulation clinical pharmacist practitioner (CPP) was alerted to the note by the investigators for further evaluation and consideration of discontinuing anticoagulation.
EHR reviews were conducted in October and November 2023 and lasted about 10 to 20 minutes per patient. To evaluate completeness and accuracy of the documented findings within the EHR, both investigators reviewed and cosigned the completed note template and verified the correct PCP was alerted to the recommendation for appropriate continuity of care. Results were reviewed in March 2024.
Outcomes
Atrial fibrillation cohort. The primary outcome was the number of veterans with Afib who were recommended to start anticoagulation therapy. Additional outcomes evaluated included the number of interventions completed, action taken by PCPs in response to the provided recommendation, and reasons provided by the investigators for not recommending initiation of anticoagulation therapy in specific veteran cases.
Venous thromboembolism cohort. The primary outcome was the number of veterans with a history of VTE events recommended to discontinue anticoagulation therapy. Additional outcomes included number of interventions completed, action taken by the anticoagulation CPP in response to the provided recommendation, and reasons provided by the investigators for not recommending discontinuation of anticoagulation therapy in specific veteran cases.
Analysis
Sample size was determined by the inclusion criteria and was not designed to attain statistical power. Data embedded in the Afib cohort standardized note template, also known as health factors, were later used for data analysis. Recommendations in the VTE cohort were manually tracked and recorded by the investigators. Results for this study were analyzed using descriptive statistics.
Results
A total of 114 veterans were reviewed and included in this study: 57 in each cohort. Seven recommendations were made regarding anticoagulation initiation for patients with Afib and 7 were made for anticoagulation discontinuation for patients with VTE (Table 1).
In the Afib cohort, 1 veteran was successfully initiated on anticoagulation therapy and 1 veteran was deemed appropriate for initiation of anticoagulation but was not reachable. Of the 5 recommendations with no action taken, 4 PCPs acknowledged the alert with no further documentation, and 1 PCP deferred the decision to cardiology with no further documentation. In the VTE cohort, 3 veterans successfully discontinued anticoagulation therapy and 2 veterans were further evaluated by the anticoagulation CPP and deemed appropriate to continue therapy based on potential for malignancy. Of the 2 recommendations with no action taken, 1 anticoagulation CPP acknowledged the alert with no further documentation and 1 anticoagulation CPP suggested further evaluation by PCP with no further documentation.
In the Afib cohort, a nonpharmacologic approach was defined as documentation of a LAAO device. An inaccurate diagnosis was defined as an Afib diagnosis being used in a previous visit, although there was no further confirmation of diagnosis via chart review. Veterans classified as already being on anticoagulation had documentation of non–VA-written anticoagulant prescriptions or receiving a supply of anticoagulants from a facility such as a nursing home. Anticoagulation was defined as unfavorable if a documented risk/benefit conversation was found via EHR review. Anticoagulation was defined as not indicated if the Afib was documented as transient, episodic, or historical (Table 2).
In the VTE cohort, no recommendations for discontinuation were made for veterans indicated to continue anticoagulation due to a concurrent Afib diagnosis. Chronic or recurrent events were defined as documentation of multiple VTE events and associated dates in the EHR. Persistent risk factors included malignancy or medications contributing to hypercoagulable states. Thrombophilia was defined as having documentation of a diagnosis in the EHR. An unprovoked event was defined as VTE without any documented transient risk factors (eg, hospitalization, trauma, surgery, cast immobilization, hormone therapy, pregnancy, or prolonged travel). Anticoagulation had already been discontinued in 1 veteran after the data were collected but before chart review occurred (Table 3).

Discussion
Pharmacy-led indication reviews resulted in appropriate recommendations for anticoagulation use in veterans with Afib and a history of VTE events. Overall, 12.3% of chart reviews in each cohort resulted in a recommendation being made, which was similar to the rate found by Koolian et al.5 In that study, 10% of recommendations were related to initiation or interruption of anticoagulation. This recommendation category consisted of several subcategories, including “suggesting therapeutic anticoagulation when none is currently ordered” and “suggesting anticoagulation cessation if no longer indicated,” but specific numerical prevalence was not provided.5
Online dashboard use allowed for greater population health management and identification of veterans with Afib who were not on active anticoagulation, providing opportunities to prevent stroke-related complications. Wang et al completed a similarly designed study that included a population health tool to identify patients with Afib who were not on anticoagulation and implemented pharmacist-led chart review and facilitation of recommendations to the responsible clinician. This study reviewed 1727 patients and recommended initiation of anticoagulation therapy for 75 (4.3%).6 The current study had a higher percentage of patients with recommendations for changes despite its smaller size.
Evaluating the duration of therapy for anticoagulation in veterans with a history of VTE events provided an opportunity to reduce unnecessary exposure to anticoagulation and minimize bleeding risks. Using a chart review process and standardized note template enabled the documentation of pertinent information that could be readily reviewed by the PCP. This process is a step toward ensuring VAPHCS PCPs provide guideline-recommended care and actively prevent stroke and bleeding complications. Adoption of this process into the current VAPHCS Anticoagulation Clinic workflow for review of veterans with either Afib or VTE could lead to more EHRs being reviewed and recommendations made, ultimately improving patient outcomes.
Therapeutic interventions based on the recommendations were completed for 1 of 7 veterans (14%) and 3 of 7 veterans (43%) in the Afib and VTE cohorts, respectively. The prevalence of completed interventions in this anticoagulation stewardship study was higher than those in Wang et al, who found only 9% of their recommendations resulted in PCPs considering action related to anticoagulation, and only 4% were successfully initiated.6
In the Afib cohort, veterans identified by the dashboard with a CHA2DS2-VASc of 7 or 8 were prioritized for review. Reviewing these veterans ensured that patients with the highest stroke risk were sufficiently evaluated and started on anticoagulation as needed to reduce stroke-related complications. In contrast, because these veterans had higher CHA2DS2-VASc scores, they may have already been evaluated for anticoagulation in the past and had a documented rationale for not being placed on anticoagulation (LAAO device placement was the most common rationale). Focusing on veterans with a lower CHA2DS2-VASc score such as 1 for men or 2 for women could potentially include more opportunities for recommendations. Although stroke risk may be lower in this population compared with those with higher CHA2DS2-VASc scores, guideline-recommended anticoagulation use may be missed for these patients.
In the VTE cohort, veterans with an anticoagulant prescription written 12 months before data collection were prioritized for review. Reviewing these veterans ensured that anticoagulation therapy met guideline recommendations of at least 3 months, with potential for extended duration upon further evaluation by a provider at that time. Based on collected results, most veterans were already reevaluated and had documented reasons why anticoagulation was still indicated; concurrent Afib was most common followed by chronic or recurrent VTE. Reviewing veterans with more recent prescriptions just over the recommended 3-month duration could potentially include more opportunities for recommendations to be made. It is more likely that by 3 months another PCP had not already weighed in on the duration of therapy, and the anticoagulation CPP could ensure a thorough review is conducted with guideline-based recommendations.
Most published literature on anticoagulation stewardship efforts is focused on inpatient management and policy changes, or concentrate on attributes of therapy such as appropriate dosing and drug interactions. This study highlighted that gaps in care related to anticoagulation use and discontinuation are present in the VAPHCS population and can be appropriately addressed via pharmacist-led indication reviews. Future studies designed to focus on initiating anticoagulation where appropriate, and discontinuing where a sufficient treatment period has been completed, are warranted to minimize this gap in care and allow health systems to work toward process changes to ensure safe and optimized care is provided for the patients they serve.
Limitations
In the Afib cohort, 5 of 7 recommendations (71%) had no further action taken by the PCP, which may represent a barrier to care. In contrast, 2 of 7 recommendations (29%) had no further action in the VTE cohort. It is possible that the difference can be attributed to the anticoagulation CPP receiving VTE alerts and PCPs receiving Afib alerts. The anticoagulation CPP was familiar with this QI study and may have better understood the purpose of the chart review and the need to provide a timely response. PCPs may have been less likely to take action because they were unfamiliar with the anticoagulation stewardship initiative and standardized note template or overwhelmed by too many EHR alerts.
The lack of PCP response to a virtual alert or message also was observed by Wang et al, whereas Koolian et al reported higher intervention completion rates, with verbal recommendations being made to the responsible clinicians. To further ensure these pertinent recommendations for anticoagulation initiation in veterans with Afib are properly reviewed and evaluated, future research could include intentional follow-up with the PCP regarding the alert, PCP-specific education about the anticoagulation stewardship initiative and the role of the standardized note template, and collaboration with PCPs to identify alternative ways to relay recommendations in a way that would ensure the completion of appropriate and timely review.
Conclusions
This study identified gaps in care related to anticoagulation needs in the VAPHCS veteran population. Utilizing a standardized indication review process allows pharmacists to evaluate anticoagulant use for both appropriate indication and duration of therapy. Providing recommendations via chart review notes and alerting respective PCPs and CPPs results in veterans receiving safe and optimized care regarding their anticoagulation needs.
- Joglar JA, Chung MK, Armbruster AL, et al. 2023 ACC/AHA/ACCP/HRS guideline for the diagnosis and management of atrial fibrillation: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2024;149:e1-e156. doi:10.1161/CIR.0000000000001193
- Stevens SM, Woller SC, Kreuziger LB, et al. Antithrombotic therapy for VTE disease: second update of the CHEST guideline and expert panel report. Chest. 2021;160:e545-e608. doi:10.1016/j.chest.2021.07.055
- Institute for Safe Medication Practices (ISMP). List of high-alert medications in community/ambulatory care settings. ISMP. September 30, 2021. Accessed September 11, 2025. https://home.ecri.org/blogs/ismp-resources/high-alert-medications-in-community-ambulatory-care-settings
- Burnett AE, Barnes GD. A call to action for anticoagulation stewardship. Res Pract Thromb Haemost. 2022;6:e12757. doi:10.1002/rth2.12757
- Koolian M, Wiseman D, Mantzanis H, et al. Anticoagulation stewardship: descriptive analysis of a novel approach to appropriate anticoagulant prescription. Res Pract Thromb Haemost. 2022;6:e12758. doi:10.1002/rth2.12758
- Wang SV, Rogers JR, Jin Y, et al. Stepped-wedge randomised trial to evaluate population health intervention designed to increase appropriate anticoagulation in patients with atrial fibrillation. BMJ Qual Saf. 2019;28:835-842. doi:10.1136/bmjqs-2019-009367
Due to the underlying mechanism of atrial fibrillation (Afib), clots can form within the left atrial appendage. Clots that become dislodged may lead to ischemic stroke and possibly death. The 2023 guidelines for atrial fibrillation from the American College of Cardiology and American Heart Association recommend anticoagulation therapy for patients with an Afib diagnosis and a CHA2DS2-VASc (congestive heart failure, hypertension, age ≥ 75 years, diabetes, stroke/vascular disease, age 65 to 74 years, and female sex) score pertinent for ≥ 1 non–sex-related factor (score ≥ 2 for women; ≥ 1 for men) to prevent stroke-related complications. The CHA2DS2-VASc score is a 9-point scoring tool based on comorbidities and conditions that increase risk of stroke in patients with Afib. Each value correlates to an annualized stroke risk percentage that increases as the score increases.
In clinical practice, patients meeting these thresholds are indicated for anticoagulation and are considered for indefinite use unless ≥ 1 of the following conditions are present: bleeding risk outweighs the stroke prevention benefit, Afib is episodic (< 48 hours) or a nonpharmacologic intervention, such as a left atrial appendage occlusion (LAAO) device is present.1
In patients with a diagnosed venous thromboembolism (VTE), such as deep vein thrombosis or pulmonary embolism, anticoagulation is used to treat the current thrombosis and prevent embolization that can ultimately lead to death. The 2021 guideline for VTE from the American College of Chest Physicians identifies certain risk factors that increase risk for VTE and categorizes them as transient or persistent. Transient risk factors include hospitalization > 3 days, major trauma, surgery, cast immobilization, hormone therapy, pregnancy, or prolonged travel > 8 hours. Persistent risk factors include malignancy, thrombophilia, and certain medications.
The guideline recommends therapy durations based on event frequency, the presence and classification of provoking risk factors, and bleeding risk. As the risk of recurrent thrombosis and other potential complications is greatest in the first 3 to 6 months after a diagnosed event, at least 3 months anticoagulation therapy is recommended following VTE diagnosis. At the 3-month mark, all regimens are suggested to be re-evaluated and considered for extended treatment duration if the event was unprovoked, recurrent, secondary to a persistent risk factor, or low bleed risk.2Anticoagulation is an important guideline-recommended pharmacologic intervention for various disease states, although its use is not without risks. The Institute for Safe Medication Practices has classified oral anticoagulants as high-alert medications. This designation was made because anticoagulant medications have the potential to cause harm when used or omitted in error and lead to life-threatening bleed or thrombotic complications.3Anticoagulation stewardship ensures that anticoagulation therapy is appropriately initiated, maintained, and discontinued when indicated. Because of the potential for harm, anticoagulation stewardship is an important part of Afib and VTE management. Pharmacists can help verify and evaluate anticoagulation therapies. Research suggests that pharmacist-led anticoagulation stewardship efforts may play a role in ensuring safer patient outcomes.4The purpose of this quality improvement (QI) study was to implement pharmacist-led anticoagulation stewardship practices at Veterans Affairs Phoenix Health Care System (VAPHCS) to identify veterans with Afib not currently on anticoagulation, as well as to identify veterans with a history of VTE events who have completed a sufficient treatment duration.
Methods
Anticoagulation stewardship efforts were implemented in 2 cohorts of patients: those with Afib who may be indicated to initiate anticoagulation, and those with a history of VTE events who may be indicated to consider anticoagulation discontinuation. Patient records were reviewed using a standardized note template, and recommendations to either initiate or discontinue anticoagulation therapy were documented. The VAPHCS Research Service reviewed this study and determined that it was not research and was exempt from institutional review board review.
Atrial Fibrillation Cohort
A population health dashboard created by the Stroke Prevention in Atrial Fibrillation/Flutter Targeting the uNTreated: a focus on health care disparities (SPAFF-TNT-D) national VA study team was used to identify veterans at VAPHCS with a diagnosis of Afib without an active VA prescription for an anticoagulant. The dashboard filtered and produced data points from the medical record that correlated to the components of the CHA2DS2-VASc score. All veterans identified by the dashboard with scores of 7 or 8 were included. No patients had a score of 9. Comprehensive chart reviews of available VA and non–VA-provided care records were conducted by the investigators, and a standardized note template designed by the SPAFF-TNT-D team (eAppendix 1) was used to document findings within the electronic health record (EHR). If anticoagulation was deemed to be indicated, the assigned primary care practitioner (PCP) as listed in the EHR was alerted to the note by the investigators for further evaluation and consideration of prescribing anticoagulation.
Venous Thromboembolism Cohort
VAPHCS pharmacy informatics pulled data that included veterans with documented VTE and an active VA anticoagulant prescription between November 2022 and November 2023. Veterans were reviewed in chronological order based on when the anticoagulant prescription was written. All veterans were included until an equal number of charts were reviewed in both the Afib and VTE cohorts. Comprehensive chart review of available VA- and non–VA-provided care records was conducted by the investigators, and a standardized note template as designed by the investigators (eAppendix 2) was used to document findings within the EHR. If the duration of anticoagulation therapy was deemed sufficient, the assigned anticoagulation clinical pharmacist practitioner (CPP) was alerted to the note by the investigators for further evaluation and consideration of discontinuing anticoagulation.
EHR reviews were conducted in October and November 2023 and lasted about 10 to 20 minutes per patient. To evaluate completeness and accuracy of the documented findings within the EHR, both investigators reviewed and cosigned the completed note template and verified the correct PCP was alerted to the recommendation for appropriate continuity of care. Results were reviewed in March 2024.
Outcomes
Atrial fibrillation cohort. The primary outcome was the number of veterans with Afib who were recommended to start anticoagulation therapy. Additional outcomes evaluated included the number of interventions completed, action taken by PCPs in response to the provided recommendation, and reasons provided by the investigators for not recommending initiation of anticoagulation therapy in specific veteran cases.
Venous thromboembolism cohort. The primary outcome was the number of veterans with a history of VTE events recommended to discontinue anticoagulation therapy. Additional outcomes included number of interventions completed, action taken by the anticoagulation CPP in response to the provided recommendation, and reasons provided by the investigators for not recommending discontinuation of anticoagulation therapy in specific veteran cases.
Analysis
Sample size was determined by the inclusion criteria and was not designed to attain statistical power. Data embedded in the Afib cohort standardized note template, also known as health factors, were later used for data analysis. Recommendations in the VTE cohort were manually tracked and recorded by the investigators. Results for this study were analyzed using descriptive statistics.
Results
A total of 114 veterans were reviewed and included in this study: 57 in each cohort. Seven recommendations were made regarding anticoagulation initiation for patients with Afib and 7 were made for anticoagulation discontinuation for patients with VTE (Table 1).

In the Afib cohort, 1 veteran was successfully initiated on anticoagulation therapy and 1 veteran was deemed appropriate for initiation of anticoagulation but was not reachable. Of the 5 recommendations with no action taken, 4 PCPs acknowledged the alert with no further documentation, and 1 PCP deferred the decision to cardiology with no further documentation. In the VTE cohort, 3 veterans successfully discontinued anticoagulation therapy and 2 veterans were further evaluated by the anticoagulation CPP and deemed appropriate to continue therapy based on potential for malignancy. Of the 2 recommendations with no action taken, 1 anticoagulation CPP acknowledged the alert with no further documentation and 1 anticoagulation CPP suggested further evaluation by PCP with no further documentation.
In the Afib cohort, a nonpharmacologic approach was defined as documentation of a LAAO device. An inaccurate diagnosis was defined as an Afib diagnosis being used in a previous visit, although there was no further confirmation of diagnosis via chart review. Veterans classified as already being on anticoagulation had documentation of non–VA-written anticoagulant prescriptions or receiving a supply of anticoagulants from a facility such as a nursing home. Anticoagulation was defined as unfavorable if a documented risk/benefit conversation was found via EHR review. Anticoagulation was defined as not indicated if the Afib was documented as transient, episodic, or historical (Table 2).

In the VTE cohort, no recommendations for discontinuation were made for veterans indicated to continue anticoagulation due to a concurrent Afib diagnosis. Chronic or recurrent events were defined as documentation of multiple VTE events and associated dates in the EHR. Persistent risk factors included malignancy or medications contributing to hypercoagulable states. Thrombophilia was defined as having documentation of a diagnosis in the EHR. An unprovoked event was defined as VTE without any documented transient risk factors (eg, hospitalization, trauma, surgery, cast immobilization, hormone therapy, pregnancy, or prolonged travel). Anticoagulation had already been discontinued in 1 veteran after the data were collected but before chart review occurred (Table 3).

Discussion
Pharmacy-led indication reviews resulted in appropriate recommendations for anticoagulation use in veterans with Afib and a history of VTE events. Overall, 12.3% of chart reviews in each cohort resulted in a recommendation being made, which was similar to the rate found by Koolian et al.5 In that study, 10% of recommendations were related to initiation or interruption of anticoagulation. This recommendation category consisted of several subcategories, including “suggesting therapeutic anticoagulation when none is currently ordered” and “suggesting anticoagulation cessation if no longer indicated,” but specific numerical prevalence was not provided.5
Online dashboard use allowed for greater population health management and identification of veterans with Afib who were not on active anticoagulation, providing opportunities to prevent stroke-related complications. Wang et al completed a similarly designed study that included a population health tool to identify patients with Afib who were not on anticoagulation and implemented pharmacist-led chart review and facilitation of recommendations to the responsible clinician. This study reviewed 1727 patients and recommended initiation of anticoagulation therapy for 75 (4.3%).6 The current study had a higher percentage of patients with recommendations for changes despite its smaller size.
Evaluating the duration of therapy for anticoagulation in veterans with a history of VTE events provided an opportunity to reduce unnecessary exposure to anticoagulation and minimize bleeding risks. Using a chart review process and standardized note template enabled the documentation of pertinent information that could be readily reviewed by the PCP. This process is a step toward ensuring VAPHCS PCPs provide guideline-recommended care and actively prevent stroke and bleeding complications. Adoption of this process into the current VAPHCS Anticoagulation Clinic workflow for review of veterans with either Afib or VTE could lead to more EHRs being reviewed and recommendations made, ultimately improving patient outcomes.
Therapeutic interventions based on the recommendations were completed for 1 of 7 veterans (14%) and 3 of 7 veterans (43%) in the Afib and VTE cohorts, respectively. The prevalence of completed interventions in this anticoagulation stewardship study was higher than those in Wang et al, who found only 9% of their recommendations resulted in PCPs considering action related to anticoagulation, and only 4% were successfully initiated.6
In the Afib cohort, veterans identified by the dashboard with a CHA2DS2-VASc of 7 or 8 were prioritized for review. Reviewing these veterans ensured that patients with the highest stroke risk were sufficiently evaluated and started on anticoagulation as needed to reduce stroke-related complications. In contrast, because these veterans had higher CHA2DS2-VASc scores, they may have already been evaluated for anticoagulation in the past and had a documented rationale for not being placed on anticoagulation (LAAO device placement was the most common rationale). Focusing on veterans with a lower CHA2DS2-VASc score such as 1 for men or 2 for women could potentially include more opportunities for recommendations. Although stroke risk may be lower in this population compared with those with higher CHA2DS2-VASc scores, guideline-recommended anticoagulation use may be missed for these patients.
In the VTE cohort, veterans with an anticoagulant prescription written 12 months before data collection were prioritized for review. Reviewing these veterans ensured that anticoagulation therapy met guideline recommendations of at least 3 months, with potential for extended duration upon further evaluation by a provider at that time. Based on collected results, most veterans were already reevaluated and had documented reasons why anticoagulation was still indicated; concurrent Afib was most common followed by chronic or recurrent VTE. Reviewing veterans with more recent prescriptions just over the recommended 3-month duration could potentially include more opportunities for recommendations to be made. It is more likely that by 3 months another PCP had not already weighed in on the duration of therapy, and the anticoagulation CPP could ensure a thorough review is conducted with guideline-based recommendations.
Most published literature on anticoagulation stewardship efforts is focused on inpatient management and policy changes, or concentrate on attributes of therapy such as appropriate dosing and drug interactions. This study highlighted that gaps in care related to anticoagulation use and discontinuation are present in the VAPHCS population and can be appropriately addressed via pharmacist-led indication reviews. Future studies designed to focus on initiating anticoagulation where appropriate, and discontinuing where a sufficient treatment period has been completed, are warranted to minimize this gap in care and allow health systems to work toward process changes to ensure safe and optimized care is provided for the patients they serve.
Limitations
In the Afib cohort, 5 of 7 recommendations (71%) had no further action taken by the PCP, which may represent a barrier to care. In contrast, 2 of 7 recommendations (29%) had no further action in the VTE cohort. It is possible that the difference can be attributed to the anticoagulation CPP receiving VTE alerts and PCPs receiving Afib alerts. The anticoagulation CPP was familiar with this QI study and may have better understood the purpose of the chart review and the need to provide a timely response. PCPs may have been less likely to take action because they were unfamiliar with the anticoagulation stewardship initiative and standardized note template or overwhelmed by too many EHR alerts.
The lack of PCP response to a virtual alert or message also was observed by Wang et al, whereas Koolian et al reported higher intervention completion rates, with verbal recommendations being made to the responsible clinicians. To further ensure these pertinent recommendations for anticoagulation initiation in veterans with Afib are properly reviewed and evaluated, future research could include intentional follow-up with the PCP regarding the alert, PCP-specific education about the anticoagulation stewardship initiative and the role of the standardized note template, and collaboration with PCPs to identify alternative ways to relay recommendations in a way that would ensure the completion of appropriate and timely review.
Conclusions
This study identified gaps in care related to anticoagulation needs in the VAPHCS veteran population. Utilizing a standardized indication review process allows pharmacists to evaluate anticoagulant use for both appropriate indication and duration of therapy. Providing recommendations via chart review notes and alerting respective PCPs and CPPs results in veterans receiving safe and optimized care regarding their anticoagulation needs.
Due to the underlying mechanism of atrial fibrillation (Afib), clots can form within the left atrial appendage. Clots that become dislodged may lead to ischemic stroke and possibly death. The 2023 guidelines for atrial fibrillation from the American College of Cardiology and American Heart Association recommend anticoagulation therapy for patients with an Afib diagnosis and a CHA2DS2-VASc (congestive heart failure, hypertension, age ≥ 75 years, diabetes, stroke/vascular disease, age 65 to 74 years, and female sex) score pertinent for ≥ 1 non–sex-related factor (score ≥ 2 for women; ≥ 1 for men) to prevent stroke-related complications. The CHA2DS2-VASc score is a 9-point scoring tool based on comorbidities and conditions that increase risk of stroke in patients with Afib. Each value correlates to an annualized stroke risk percentage that increases as the score increases.
In clinical practice, patients meeting these thresholds are indicated for anticoagulation and are considered for indefinite use unless ≥ 1 of the following conditions are present: bleeding risk outweighs the stroke prevention benefit, Afib is episodic (< 48 hours) or a nonpharmacologic intervention, such as a left atrial appendage occlusion (LAAO) device is present.1
In patients with a diagnosed venous thromboembolism (VTE), such as deep vein thrombosis or pulmonary embolism, anticoagulation is used to treat the current thrombosis and prevent embolization that can ultimately lead to death. The 2021 guideline for VTE from the American College of Chest Physicians identifies certain risk factors that increase risk for VTE and categorizes them as transient or persistent. Transient risk factors include hospitalization > 3 days, major trauma, surgery, cast immobilization, hormone therapy, pregnancy, or prolonged travel > 8 hours. Persistent risk factors include malignancy, thrombophilia, and certain medications.
The guideline recommends therapy durations based on event frequency, the presence and classification of provoking risk factors, and bleeding risk. As the risk of recurrent thrombosis and other potential complications is greatest in the first 3 to 6 months after a diagnosed event, at least 3 months anticoagulation therapy is recommended following VTE diagnosis. At the 3-month mark, all regimens are suggested to be re-evaluated and considered for extended treatment duration if the event was unprovoked, recurrent, secondary to a persistent risk factor, or low bleed risk.2Anticoagulation is an important guideline-recommended pharmacologic intervention for various disease states, although its use is not without risks. The Institute for Safe Medication Practices has classified oral anticoagulants as high-alert medications. This designation was made because anticoagulant medications have the potential to cause harm when used or omitted in error and lead to life-threatening bleed or thrombotic complications.3Anticoagulation stewardship ensures that anticoagulation therapy is appropriately initiated, maintained, and discontinued when indicated. Because of the potential for harm, anticoagulation stewardship is an important part of Afib and VTE management. Pharmacists can help verify and evaluate anticoagulation therapies. Research suggests that pharmacist-led anticoagulation stewardship efforts may play a role in ensuring safer patient outcomes.4The purpose of this quality improvement (QI) study was to implement pharmacist-led anticoagulation stewardship practices at Veterans Affairs Phoenix Health Care System (VAPHCS) to identify veterans with Afib not currently on anticoagulation, as well as to identify veterans with a history of VTE events who have completed a sufficient treatment duration.
Methods
Anticoagulation stewardship efforts were implemented in 2 cohorts of patients: those with Afib who may be indicated to initiate anticoagulation, and those with a history of VTE events who may be indicated to consider anticoagulation discontinuation. Patient records were reviewed using a standardized note template, and recommendations to either initiate or discontinue anticoagulation therapy were documented. The VAPHCS Research Service reviewed this study and determined that it was not research and was exempt from institutional review board review.
Atrial Fibrillation Cohort
A population health dashboard created by the Stroke Prevention in Atrial Fibrillation/Flutter Targeting the uNTreated: a focus on health care disparities (SPAFF-TNT-D) national VA study team was used to identify veterans at VAPHCS with a diagnosis of Afib without an active VA prescription for an anticoagulant. The dashboard filtered and produced data points from the medical record that correlated to the components of the CHA2DS2-VASc score. All veterans identified by the dashboard with scores of 7 or 8 were included. No patients had a score of 9. Comprehensive chart reviews of available VA and non–VA-provided care records were conducted by the investigators, and a standardized note template designed by the SPAFF-TNT-D team (eAppendix 1) was used to document findings within the electronic health record (EHR). If anticoagulation was deemed to be indicated, the assigned primary care practitioner (PCP) as listed in the EHR was alerted to the note by the investigators for further evaluation and consideration of prescribing anticoagulation.
Venous Thromboembolism Cohort
VAPHCS pharmacy informatics pulled data that included veterans with documented VTE and an active VA anticoagulant prescription between November 2022 and November 2023. Veterans were reviewed in chronological order based on when the anticoagulant prescription was written. All veterans were included until an equal number of charts were reviewed in both the Afib and VTE cohorts. Comprehensive chart review of available VA- and non–VA-provided care records was conducted by the investigators, and a standardized note template as designed by the investigators (eAppendix 2) was used to document findings within the EHR. If the duration of anticoagulation therapy was deemed sufficient, the assigned anticoagulation clinical pharmacist practitioner (CPP) was alerted to the note by the investigators for further evaluation and consideration of discontinuing anticoagulation.
EHR reviews were conducted in October and November 2023 and lasted about 10 to 20 minutes per patient. To evaluate completeness and accuracy of the documented findings within the EHR, both investigators reviewed and cosigned the completed note template and verified the correct PCP was alerted to the recommendation for appropriate continuity of care. Results were reviewed in March 2024.
Outcomes
Atrial fibrillation cohort. The primary outcome was the number of veterans with Afib who were recommended to start anticoagulation therapy. Additional outcomes evaluated included the number of interventions completed, action taken by PCPs in response to the provided recommendation, and reasons provided by the investigators for not recommending initiation of anticoagulation therapy in specific veteran cases.
Venous thromboembolism cohort. The primary outcome was the number of veterans with a history of VTE events recommended to discontinue anticoagulation therapy. Additional outcomes included number of interventions completed, action taken by the anticoagulation CPP in response to the provided recommendation, and reasons provided by the investigators for not recommending discontinuation of anticoagulation therapy in specific veteran cases.
Analysis
Sample size was determined by the inclusion criteria and was not designed to attain statistical power. Data embedded in the Afib cohort standardized note template, also known as health factors, were later used for data analysis. Recommendations in the VTE cohort were manually tracked and recorded by the investigators. Results for this study were analyzed using descriptive statistics.
Results
A total of 114 veterans were reviewed and included in this study: 57 in each cohort. Seven recommendations were made regarding anticoagulation initiation for patients with Afib and 7 were made for anticoagulation discontinuation for patients with VTE (Table 1).

In the Afib cohort, 1 veteran was successfully initiated on anticoagulation therapy and 1 veteran was deemed appropriate for initiation of anticoagulation but was not reachable. Of the 5 recommendations with no action taken, 4 PCPs acknowledged the alert with no further documentation, and 1 PCP deferred the decision to cardiology with no further documentation. In the VTE cohort, 3 veterans successfully discontinued anticoagulation therapy and 2 veterans were further evaluated by the anticoagulation CPP and deemed appropriate to continue therapy based on potential for malignancy. Of the 2 recommendations with no action taken, 1 anticoagulation CPP acknowledged the alert with no further documentation and 1 anticoagulation CPP suggested further evaluation by PCP with no further documentation.
In the Afib cohort, a nonpharmacologic approach was defined as documentation of a LAAO device. An inaccurate diagnosis was defined as an Afib diagnosis being used in a previous visit, although there was no further confirmation of diagnosis via chart review. Veterans classified as already being on anticoagulation had documentation of non–VA-written anticoagulant prescriptions or receiving a supply of anticoagulants from a facility such as a nursing home. Anticoagulation was defined as unfavorable if a documented risk/benefit conversation was found via EHR review. Anticoagulation was defined as not indicated if the Afib was documented as transient, episodic, or historical (Table 2).

In the VTE cohort, no recommendations for discontinuation were made for veterans indicated to continue anticoagulation due to a concurrent Afib diagnosis. Chronic or recurrent events were defined as documentation of multiple VTE events and associated dates in the EHR. Persistent risk factors included malignancy or medications contributing to hypercoagulable states. Thrombophilia was defined as having documentation of a diagnosis in the EHR. An unprovoked event was defined as VTE without any documented transient risk factors (eg, hospitalization, trauma, surgery, cast immobilization, hormone therapy, pregnancy, or prolonged travel). Anticoagulation had already been discontinued in 1 veteran after the data were collected but before chart review occurred (Table 3).

Discussion
Pharmacy-led indication reviews resulted in appropriate recommendations for anticoagulation use in veterans with Afib and a history of VTE events. Overall, 12.3% of chart reviews in each cohort resulted in a recommendation being made, which was similar to the rate found by Koolian et al.5 In that study, 10% of recommendations were related to initiation or interruption of anticoagulation. This recommendation category consisted of several subcategories, including “suggesting therapeutic anticoagulation when none is currently ordered” and “suggesting anticoagulation cessation if no longer indicated,” but specific numerical prevalence was not provided.5
Online dashboard use allowed for greater population health management and identification of veterans with Afib who were not on active anticoagulation, providing opportunities to prevent stroke-related complications. Wang et al completed a similarly designed study that included a population health tool to identify patients with Afib who were not on anticoagulation and implemented pharmacist-led chart review and facilitation of recommendations to the responsible clinician. This study reviewed 1727 patients and recommended initiation of anticoagulation therapy for 75 (4.3%).6 The current study had a higher percentage of patients with recommendations for changes despite its smaller size.
Evaluating the duration of therapy for anticoagulation in veterans with a history of VTE events provided an opportunity to reduce unnecessary exposure to anticoagulation and minimize bleeding risks. Using a chart review process and standardized note template enabled the documentation of pertinent information that could be readily reviewed by the PCP. This process is a step toward ensuring VAPHCS PCPs provide guideline-recommended care and actively prevent stroke and bleeding complications. Adoption of this process into the current VAPHCS Anticoagulation Clinic workflow for review of veterans with either Afib or VTE could lead to more EHRs being reviewed and recommendations made, ultimately improving patient outcomes.
Therapeutic interventions based on the recommendations were completed for 1 of 7 veterans (14%) and 3 of 7 veterans (43%) in the Afib and VTE cohorts, respectively. The prevalence of completed interventions in this anticoagulation stewardship study was higher than those in Wang et al, who found only 9% of their recommendations resulted in PCPs considering action related to anticoagulation, and only 4% were successfully initiated.6
In the Afib cohort, veterans identified by the dashboard with a CHA2DS2-VASc of 7 or 8 were prioritized for review. Reviewing these veterans ensured that patients with the highest stroke risk were sufficiently evaluated and started on anticoagulation as needed to reduce stroke-related complications. In contrast, because these veterans had higher CHA2DS2-VASc scores, they may have already been evaluated for anticoagulation in the past and had a documented rationale for not being placed on anticoagulation (LAAO device placement was the most common rationale). Focusing on veterans with a lower CHA2DS2-VASc score such as 1 for men or 2 for women could potentially include more opportunities for recommendations. Although stroke risk may be lower in this population compared with those with higher CHA2DS2-VASc scores, guideline-recommended anticoagulation use may be missed for these patients.
In the VTE cohort, veterans with an anticoagulant prescription written 12 months before data collection were prioritized for review. Reviewing these veterans ensured that anticoagulation therapy met guideline recommendations of at least 3 months, with potential for extended duration upon further evaluation by a provider at that time. Based on collected results, most veterans were already reevaluated and had documented reasons why anticoagulation was still indicated; concurrent Afib was most common followed by chronic or recurrent VTE. Reviewing veterans with more recent prescriptions just over the recommended 3-month duration could potentially include more opportunities for recommendations to be made. It is more likely that by 3 months another PCP had not already weighed in on the duration of therapy, and the anticoagulation CPP could ensure a thorough review is conducted with guideline-based recommendations.
Most published literature on anticoagulation stewardship efforts is focused on inpatient management and policy changes, or concentrate on attributes of therapy such as appropriate dosing and drug interactions. This study highlighted that gaps in care related to anticoagulation use and discontinuation are present in the VAPHCS population and can be appropriately addressed via pharmacist-led indication reviews. Future studies designed to focus on initiating anticoagulation where appropriate, and discontinuing where a sufficient treatment period has been completed, are warranted to minimize this gap in care and allow health systems to work toward process changes to ensure safe and optimized care is provided for the patients they serve.
Limitations
In the Afib cohort, 5 of 7 recommendations (71%) had no further action taken by the PCP, which may represent a barrier to care. In contrast, 2 of 7 recommendations (29%) had no further action in the VTE cohort. It is possible that the difference can be attributed to the anticoagulation CPP receiving VTE alerts and PCPs receiving Afib alerts. The anticoagulation CPP was familiar with this QI study and may have better understood the purpose of the chart review and the need to provide a timely response. PCPs may have been less likely to take action because they were unfamiliar with the anticoagulation stewardship initiative and standardized note template or overwhelmed by too many EHR alerts.
The lack of PCP response to a virtual alert or message also was observed by Wang et al, whereas Koolian et al reported higher intervention completion rates, with verbal recommendations being made to the responsible clinicians. To further ensure these pertinent recommendations for anticoagulation initiation in veterans with Afib are properly reviewed and evaluated, future research could include intentional follow-up with the PCP regarding the alert, PCP-specific education about the anticoagulation stewardship initiative and the role of the standardized note template, and collaboration with PCPs to identify alternative ways to relay recommendations in a way that would ensure the completion of appropriate and timely review.
Conclusions
This study identified gaps in care related to anticoagulation needs in the VAPHCS veteran population. Utilizing a standardized indication review process allows pharmacists to evaluate anticoagulant use for both appropriate indication and duration of therapy. Providing recommendations via chart review notes and alerting respective PCPs and CPPs results in veterans receiving safe and optimized care regarding their anticoagulation needs.
- Joglar JA, Chung MK, Armbruster AL, et al. 2023 ACC/AHA/ACCP/HRS guideline for the diagnosis and management of atrial fibrillation: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2024;149:e1-e156. doi:10.1161/CIR.0000000000001193
- Stevens SM, Woller SC, Kreuziger LB, et al. Antithrombotic therapy for VTE disease: second update of the CHEST guideline and expert panel report. Chest. 2021;160:e545-e608. doi:10.1016/j.chest.2021.07.055
- Institute for Safe Medication Practices (ISMP). List of high-alert medications in community/ambulatory care settings. ISMP. September 30, 2021. Accessed September 11, 2025. https://home.ecri.org/blogs/ismp-resources/high-alert-medications-in-community-ambulatory-care-settings
- Burnett AE, Barnes GD. A call to action for anticoagulation stewardship. Res Pract Thromb Haemost. 2022;6:e12757. doi:10.1002/rth2.12757
- Koolian M, Wiseman D, Mantzanis H, et al. Anticoagulation stewardship: descriptive analysis of a novel approach to appropriate anticoagulant prescription. Res Pract Thromb Haemost. 2022;6:e12758. doi:10.1002/rth2.12758
- Wang SV, Rogers JR, Jin Y, et al. Stepped-wedge randomised trial to evaluate population health intervention designed to increase appropriate anticoagulation in patients with atrial fibrillation. BMJ Qual Saf. 2019;28:835-842. doi:10.1136/bmjqs-2019-009367
- Joglar JA, Chung MK, Armbruster AL, et al. 2023 ACC/AHA/ACCP/HRS guideline for the diagnosis and management of atrial fibrillation: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2024;149:e1-e156. doi:10.1161/CIR.0000000000001193
- Stevens SM, Woller SC, Kreuziger LB, et al. Antithrombotic therapy for VTE disease: second update of the CHEST guideline and expert panel report. Chest. 2021;160:e545-e608. doi:10.1016/j.chest.2021.07.055
- Institute for Safe Medication Practices (ISMP). List of high-alert medications in community/ambulatory care settings. ISMP. September 30, 2021. Accessed September 11, 2025. https://home.ecri.org/blogs/ismp-resources/high-alert-medications-in-community-ambulatory-care-settings
- Burnett AE, Barnes GD. A call to action for anticoagulation stewardship. Res Pract Thromb Haemost. 2022;6:e12757. doi:10.1002/rth2.12757
- Koolian M, Wiseman D, Mantzanis H, et al. Anticoagulation stewardship: descriptive analysis of a novel approach to appropriate anticoagulant prescription. Res Pract Thromb Haemost. 2022;6:e12758. doi:10.1002/rth2.12758
- Wang SV, Rogers JR, Jin Y, et al. Stepped-wedge randomised trial to evaluate population health intervention designed to increase appropriate anticoagulation in patients with atrial fibrillation. BMJ Qual Saf. 2019;28:835-842. doi:10.1136/bmjqs-2019-009367
Anticoagulation Stewardship Efforts Via Indication Reviews at a Veterans Affairs Health Care System
Anticoagulation Stewardship Efforts Via Indication Reviews at a Veterans Affairs Health Care System
Geographic Clusters Show Uneven Cancer Screening in the US
Geographic Clusters Show Uneven Cancer Screening in the US
TOPLINE:
An analysis of 3142 US counties revealed that county-level screening for breast, cervical, and colorectal cancer increased overall between 1997 and 2019; however, despite the reduced geographic variation, persistently high-screening clusters remained in the Northeast, whereas persistently low-screening clusters remained in the Southwest.
METHODOLOGY:
- Cancer screening reduces mortality. Despite guideline recommendation, the uptake of breast, cervical, and colorectal cancer screening in the US falls short of national goals and varies across sociodemographic groups. To date, only a few studies have examined geographic and temporal patterns of screening.
- To address this gap, researchers conducted a cross-sectional study using an ecological panel design to analyze county-level screening prevalence across 3142 US mainland counties from 1997 to 2019, deriving prevalence estimates from Behavioral Risk Factor Surveillance System (BRFSS) and National Health Interview Survey (NHIS) data over 3- to 5-year periods.
- Spatial autocorrelation analyses, including Global Moran I and the bivariate local indicator of spatial autocorrelation, were performed to assess geographic clusters of cancer screening within each period. Four types of local geographic clusters of county-level cancer screening were identified: counties with persistently high screening rates, counties with persistently low screening rates, counties in which screening rates decreased from high to low, and counties in which screening rates increased from low to high.
- Screening prevalence was compared across multiple time windows for different modalities (mammography, a Papanicolaou test, colonoscopy, colorectal cancer test, endoscopy, and a fecal occult blood test [FOBT]). Overall, 3101 counties were analyzed for mammography and the Papanicolaou test, 3107 counties for colonoscopy, 3100 counties for colorectal cancer test, 3089 counties for endoscopy, and 3090 counties for the FOBT.
TAKEAWAY:
- Overall screening prevalence increased from 1997 to 2019, and global spatial autocorrelation declined over time. For instance, the distribution of mammography screening became 83% more uniform in more recent years (Moran I, 0.57 in 1997-1999 vs 0.10 in 2017-2019). Similarly, Papanicolaou test screening became more uniform in more recent years (Moran I, 0.44 vs. 0.07). These changes indicate reduced geographic heterogeneity.
- Colonoscopy and endoscopy use increased, surpassing a 50% prevalence in many counties for 2010; however, FOBT use declined. Spatial clustering also attenuated, with a 23.4% declined in Moran I for colonoscopy from 2011-2016 to 2017-2019, a 12.3% decline in the colorectal cancer test from 2004-2007 to 2008-2010, and a 14.0% decline for endoscopy from 2004-2007 to 2008-2010.
- Persistently high-/high-screening clusters were concentrated in the Northeast for mammography and colorectal cancer screening and in the East for Papanicolaou test screening, whereas persistently low-/low-screening clusters were concentrated in the Southwest for the same modalities.
- Clusters of low- and high-screening counties were more disadvantaged -- with lower socioeconomic status and a higher proportion of non-White residents -- than other cluster types, suggesting some improvement in screening uptake in more disadvantaged areas. Counties with persistently low screening exhibited greater socioeconomic disadvantages -- lower media household income, higher poverty, lower home values, and lower educational attainment -- than those with persistently high screening.
IN PRACTICE:
"This cross-sectional study found that despite secular increases that reduced geographic variation in screening, local clusters of high and low screening persisted in the Northeast and Southwest US, respectively. Future studies could incorporate health care access characteristics to explain why areas of low screening did not catch up to optimize cancer screening practice," the authors wrote.
SOURCE:
The study, led by Pranoti Pradhan, PhD, Harvard T.H. Chan School of Public Health, Boston, was published online in JAMA Network Open.
LIMITATIONS:
The county-level estimates were modeled using BRFSS, NHIS, and US Census data, which might be susceptible to sampling biases despite corrections for nonresponse and noncoverage. Researchers lacked data on specific health systems characteristics that may have directly driven changes in prevalence and were restricted to using screening time intervals available from the Small Area Estimates for Cancer-Relates Measures from the National Cancer Institute, rather than those according to US Preventive Services Task Force guidelines. Additionally, the spatial cluster method was sensitive to county size and arrangement, which may have influenced local cluster detection.
DISCLOSURES:
This research was supported by the T32 Cancer Prevention and Control Funding Fellowship and T32 Cancer Epidemiology Fellowship at the Harvard T.H. Chan School of Public Health. The authors declared having no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
An analysis of 3142 US counties revealed that county-level screening for breast, cervical, and colorectal cancer increased overall between 1997 and 2019; however, despite the reduced geographic variation, persistently high-screening clusters remained in the Northeast, whereas persistently low-screening clusters remained in the Southwest.
METHODOLOGY:
- Cancer screening reduces mortality. Despite guideline recommendation, the uptake of breast, cervical, and colorectal cancer screening in the US falls short of national goals and varies across sociodemographic groups. To date, only a few studies have examined geographic and temporal patterns of screening.
- To address this gap, researchers conducted a cross-sectional study using an ecological panel design to analyze county-level screening prevalence across 3142 US mainland counties from 1997 to 2019, deriving prevalence estimates from Behavioral Risk Factor Surveillance System (BRFSS) and National Health Interview Survey (NHIS) data over 3- to 5-year periods.
- Spatial autocorrelation analyses, including Global Moran I and the bivariate local indicator of spatial autocorrelation, were performed to assess geographic clusters of cancer screening within each period. Four types of local geographic clusters of county-level cancer screening were identified: counties with persistently high screening rates, counties with persistently low screening rates, counties in which screening rates decreased from high to low, and counties in which screening rates increased from low to high.
- Screening prevalence was compared across multiple time windows for different modalities (mammography, a Papanicolaou test, colonoscopy, colorectal cancer test, endoscopy, and a fecal occult blood test [FOBT]). Overall, 3101 counties were analyzed for mammography and the Papanicolaou test, 3107 counties for colonoscopy, 3100 counties for colorectal cancer test, 3089 counties for endoscopy, and 3090 counties for the FOBT.
TAKEAWAY:
- Overall screening prevalence increased from 1997 to 2019, and global spatial autocorrelation declined over time. For instance, the distribution of mammography screening became 83% more uniform in more recent years (Moran I, 0.57 in 1997-1999 vs 0.10 in 2017-2019). Similarly, Papanicolaou test screening became more uniform in more recent years (Moran I, 0.44 vs. 0.07). These changes indicate reduced geographic heterogeneity.
- Colonoscopy and endoscopy use increased, surpassing a 50% prevalence in many counties for 2010; however, FOBT use declined. Spatial clustering also attenuated, with a 23.4% declined in Moran I for colonoscopy from 2011-2016 to 2017-2019, a 12.3% decline in the colorectal cancer test from 2004-2007 to 2008-2010, and a 14.0% decline for endoscopy from 2004-2007 to 2008-2010.
- Persistently high-/high-screening clusters were concentrated in the Northeast for mammography and colorectal cancer screening and in the East for Papanicolaou test screening, whereas persistently low-/low-screening clusters were concentrated in the Southwest for the same modalities.
- Clusters of low- and high-screening counties were more disadvantaged -- with lower socioeconomic status and a higher proportion of non-White residents -- than other cluster types, suggesting some improvement in screening uptake in more disadvantaged areas. Counties with persistently low screening exhibited greater socioeconomic disadvantages -- lower media household income, higher poverty, lower home values, and lower educational attainment -- than those with persistently high screening.
IN PRACTICE:
"This cross-sectional study found that despite secular increases that reduced geographic variation in screening, local clusters of high and low screening persisted in the Northeast and Southwest US, respectively. Future studies could incorporate health care access characteristics to explain why areas of low screening did not catch up to optimize cancer screening practice," the authors wrote.
SOURCE:
The study, led by Pranoti Pradhan, PhD, Harvard T.H. Chan School of Public Health, Boston, was published online in JAMA Network Open.
LIMITATIONS:
The county-level estimates were modeled using BRFSS, NHIS, and US Census data, which might be susceptible to sampling biases despite corrections for nonresponse and noncoverage. Researchers lacked data on specific health systems characteristics that may have directly driven changes in prevalence and were restricted to using screening time intervals available from the Small Area Estimates for Cancer-Relates Measures from the National Cancer Institute, rather than those according to US Preventive Services Task Force guidelines. Additionally, the spatial cluster method was sensitive to county size and arrangement, which may have influenced local cluster detection.
DISCLOSURES:
This research was supported by the T32 Cancer Prevention and Control Funding Fellowship and T32 Cancer Epidemiology Fellowship at the Harvard T.H. Chan School of Public Health. The authors declared having no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
An analysis of 3142 US counties revealed that county-level screening for breast, cervical, and colorectal cancer increased overall between 1997 and 2019; however, despite the reduced geographic variation, persistently high-screening clusters remained in the Northeast, whereas persistently low-screening clusters remained in the Southwest.
METHODOLOGY:
- Cancer screening reduces mortality. Despite guideline recommendation, the uptake of breast, cervical, and colorectal cancer screening in the US falls short of national goals and varies across sociodemographic groups. To date, only a few studies have examined geographic and temporal patterns of screening.
- To address this gap, researchers conducted a cross-sectional study using an ecological panel design to analyze county-level screening prevalence across 3142 US mainland counties from 1997 to 2019, deriving prevalence estimates from Behavioral Risk Factor Surveillance System (BRFSS) and National Health Interview Survey (NHIS) data over 3- to 5-year periods.
- Spatial autocorrelation analyses, including Global Moran I and the bivariate local indicator of spatial autocorrelation, were performed to assess geographic clusters of cancer screening within each period. Four types of local geographic clusters of county-level cancer screening were identified: counties with persistently high screening rates, counties with persistently low screening rates, counties in which screening rates decreased from high to low, and counties in which screening rates increased from low to high.
- Screening prevalence was compared across multiple time windows for different modalities (mammography, a Papanicolaou test, colonoscopy, colorectal cancer test, endoscopy, and a fecal occult blood test [FOBT]). Overall, 3101 counties were analyzed for mammography and the Papanicolaou test, 3107 counties for colonoscopy, 3100 counties for colorectal cancer test, 3089 counties for endoscopy, and 3090 counties for the FOBT.
TAKEAWAY:
- Overall screening prevalence increased from 1997 to 2019, and global spatial autocorrelation declined over time. For instance, the distribution of mammography screening became 83% more uniform in more recent years (Moran I, 0.57 in 1997-1999 vs 0.10 in 2017-2019). Similarly, Papanicolaou test screening became more uniform in more recent years (Moran I, 0.44 vs. 0.07). These changes indicate reduced geographic heterogeneity.
- Colonoscopy and endoscopy use increased, surpassing a 50% prevalence in many counties for 2010; however, FOBT use declined. Spatial clustering also attenuated, with a 23.4% declined in Moran I for colonoscopy from 2011-2016 to 2017-2019, a 12.3% decline in the colorectal cancer test from 2004-2007 to 2008-2010, and a 14.0% decline for endoscopy from 2004-2007 to 2008-2010.
- Persistently high-/high-screening clusters were concentrated in the Northeast for mammography and colorectal cancer screening and in the East for Papanicolaou test screening, whereas persistently low-/low-screening clusters were concentrated in the Southwest for the same modalities.
- Clusters of low- and high-screening counties were more disadvantaged -- with lower socioeconomic status and a higher proportion of non-White residents -- than other cluster types, suggesting some improvement in screening uptake in more disadvantaged areas. Counties with persistently low screening exhibited greater socioeconomic disadvantages -- lower media household income, higher poverty, lower home values, and lower educational attainment -- than those with persistently high screening.
IN PRACTICE:
"This cross-sectional study found that despite secular increases that reduced geographic variation in screening, local clusters of high and low screening persisted in the Northeast and Southwest US, respectively. Future studies could incorporate health care access characteristics to explain why areas of low screening did not catch up to optimize cancer screening practice," the authors wrote.
SOURCE:
The study, led by Pranoti Pradhan, PhD, Harvard T.H. Chan School of Public Health, Boston, was published online in JAMA Network Open.
LIMITATIONS:
The county-level estimates were modeled using BRFSS, NHIS, and US Census data, which might be susceptible to sampling biases despite corrections for nonresponse and noncoverage. Researchers lacked data on specific health systems characteristics that may have directly driven changes in prevalence and were restricted to using screening time intervals available from the Small Area Estimates for Cancer-Relates Measures from the National Cancer Institute, rather than those according to US Preventive Services Task Force guidelines. Additionally, the spatial cluster method was sensitive to county size and arrangement, which may have influenced local cluster detection.
DISCLOSURES:
This research was supported by the T32 Cancer Prevention and Control Funding Fellowship and T32 Cancer Epidemiology Fellowship at the Harvard T.H. Chan School of Public Health. The authors declared having no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
Geographic Clusters Show Uneven Cancer Screening in the US
Geographic Clusters Show Uneven Cancer Screening in the US
Team-Based Care is Crucial for Head-and-Neck Cancer Cases
Team-Based Care is Crucial for Head-and-Neck Cancer Cases
PHOENIX – A 70-year-old Vietnam veteran with oropharyngeal cancer presented challenges beyond his disease.
He couldn’t afford transportation for daily radiation treatments and had lost > 10% of his body weight due to pain and eating difficulties, recalled radiation oncologist Vinita Takiar, MD, PhD, in a presentation at the annual meeting of the Association of VA Hematology/Oncology.
To make matters more difficult, his wife held medical power of attorney despite his apparent competence to make decisions, said Takiar, who formerly worked with the US Department of Veterans Affairs (VA) Cincinnati Healthcare System and is now chair of radiation oncology at Penn State University.
All these factors would likely have derailed his treatment if not for a coordinated team intervention, Takiar said. Fortunately, the clinic launched a multifaceted effort involving representatives from the social work, dentistry, ethics, nutrition, and chaplaincy departments.
When surgery became impossible because the patient couldn’t lie on the operating table for adequate tumor exposure, she said, the existing team framework enabled a seamless and rapid transition to radiation with concurrent chemotherapy.
The patient completed treatment with an excellent response, offering a lesson in the importance of multidisciplinary care in head-and-neck cancers, she said.
In fact, when it comes to these forms of cancer, coordinated care “is probably more impactful than any treatment that we’re going to come up with,” she said. “The data show that when we do multidisciplinary care and we do it well, it actually improves the patient experience and outcomes.”
As Takiar noted, teamwork matters in many ways. It leads to better logistics and can address disparities, reduce financial burden and stigma, and even increase clinical trial involvement.
She pointed to studies linking teamwork to better outcomes, support for patients, and overall survival.
Takiar highlighted different parts of teams headed by radiation oncologists who act as “a node to improve multimodal care delivery.”
Speech and swallowing specialists, for example, are helpful in head-and-neck cancer because “there’s an impact on speech, swallowing, and appearance. Our patients don’t want to go out to dinner with friends because they can’t do it.”
Dentists and prosthodontists are key team members too: “I have dentists who have my cell phone number. They just call me: ‘Can I do this extraction? Was this in your radiation field? What was the dose?’”
Other team members include ear, nose, and throat specialists, palliative and supportive care specialists, medical oncologists, nurses, pathologists, transportation workers, and service connection specialists. She noted that previous military experience can affect radiation therapy. For example, the physical restraints required during treatment present particular challenges for veterans who’ve had wartime trauma. These patients may require therapy adjustments.
What’s next on the horizon? Takiar highlighted precision oncology and molecular profiling, artificial intelligence in care decisions and in radiation planning, telemedicine and virtual tumor boards, and expanded survivorship programs.
As for now, she urged colleagues to not be afraid to chat with radiation oncologists. “Please talk to us. We prioritize open communication and shared decision-making with the entire team,” she said. “If you see something and think your radiation oncologist should know about it, you think it was caused by the radiation, you should reach out to us.”
Takiar reported no disclosures.
PHOENIX – A 70-year-old Vietnam veteran with oropharyngeal cancer presented challenges beyond his disease.
He couldn’t afford transportation for daily radiation treatments and had lost > 10% of his body weight due to pain and eating difficulties, recalled radiation oncologist Vinita Takiar, MD, PhD, in a presentation at the annual meeting of the Association of VA Hematology/Oncology.
To make matters more difficult, his wife held medical power of attorney despite his apparent competence to make decisions, said Takiar, who formerly worked with the US Department of Veterans Affairs (VA) Cincinnati Healthcare System and is now chair of radiation oncology at Penn State University.
All these factors would likely have derailed his treatment if not for a coordinated team intervention, Takiar said. Fortunately, the clinic launched a multifaceted effort involving representatives from the social work, dentistry, ethics, nutrition, and chaplaincy departments.
When surgery became impossible because the patient couldn’t lie on the operating table for adequate tumor exposure, she said, the existing team framework enabled a seamless and rapid transition to radiation with concurrent chemotherapy.
The patient completed treatment with an excellent response, offering a lesson in the importance of multidisciplinary care in head-and-neck cancers, she said.
In fact, when it comes to these forms of cancer, coordinated care “is probably more impactful than any treatment that we’re going to come up with,” she said. “The data show that when we do multidisciplinary care and we do it well, it actually improves the patient experience and outcomes.”
As Takiar noted, teamwork matters in many ways. It leads to better logistics and can address disparities, reduce financial burden and stigma, and even increase clinical trial involvement.
She pointed to studies linking teamwork to better outcomes, support for patients, and overall survival.
Takiar highlighted different parts of teams headed by radiation oncologists who act as “a node to improve multimodal care delivery.”
Speech and swallowing specialists, for example, are helpful in head-and-neck cancer because “there’s an impact on speech, swallowing, and appearance. Our patients don’t want to go out to dinner with friends because they can’t do it.”
Dentists and prosthodontists are key team members too: “I have dentists who have my cell phone number. They just call me: ‘Can I do this extraction? Was this in your radiation field? What was the dose?’”
Other team members include ear, nose, and throat specialists, palliative and supportive care specialists, medical oncologists, nurses, pathologists, transportation workers, and service connection specialists. She noted that previous military experience can affect radiation therapy. For example, the physical restraints required during treatment present particular challenges for veterans who’ve had wartime trauma. These patients may require therapy adjustments.
What’s next on the horizon? Takiar highlighted precision oncology and molecular profiling, artificial intelligence in care decisions and in radiation planning, telemedicine and virtual tumor boards, and expanded survivorship programs.
As for now, she urged colleagues to not be afraid to chat with radiation oncologists. “Please talk to us. We prioritize open communication and shared decision-making with the entire team,” she said. “If you see something and think your radiation oncologist should know about it, you think it was caused by the radiation, you should reach out to us.”
Takiar reported no disclosures.
PHOENIX – A 70-year-old Vietnam veteran with oropharyngeal cancer presented challenges beyond his disease.
He couldn’t afford transportation for daily radiation treatments and had lost > 10% of his body weight due to pain and eating difficulties, recalled radiation oncologist Vinita Takiar, MD, PhD, in a presentation at the annual meeting of the Association of VA Hematology/Oncology.
To make matters more difficult, his wife held medical power of attorney despite his apparent competence to make decisions, said Takiar, who formerly worked with the US Department of Veterans Affairs (VA) Cincinnati Healthcare System and is now chair of radiation oncology at Penn State University.
All these factors would likely have derailed his treatment if not for a coordinated team intervention, Takiar said. Fortunately, the clinic launched a multifaceted effort involving representatives from the social work, dentistry, ethics, nutrition, and chaplaincy departments.
When surgery became impossible because the patient couldn’t lie on the operating table for adequate tumor exposure, she said, the existing team framework enabled a seamless and rapid transition to radiation with concurrent chemotherapy.
The patient completed treatment with an excellent response, offering a lesson in the importance of multidisciplinary care in head-and-neck cancers, she said.
In fact, when it comes to these forms of cancer, coordinated care “is probably more impactful than any treatment that we’re going to come up with,” she said. “The data show that when we do multidisciplinary care and we do it well, it actually improves the patient experience and outcomes.”
As Takiar noted, teamwork matters in many ways. It leads to better logistics and can address disparities, reduce financial burden and stigma, and even increase clinical trial involvement.
She pointed to studies linking teamwork to better outcomes, support for patients, and overall survival.
Takiar highlighted different parts of teams headed by radiation oncologists who act as “a node to improve multimodal care delivery.”
Speech and swallowing specialists, for example, are helpful in head-and-neck cancer because “there’s an impact on speech, swallowing, and appearance. Our patients don’t want to go out to dinner with friends because they can’t do it.”
Dentists and prosthodontists are key team members too: “I have dentists who have my cell phone number. They just call me: ‘Can I do this extraction? Was this in your radiation field? What was the dose?’”
Other team members include ear, nose, and throat specialists, palliative and supportive care specialists, medical oncologists, nurses, pathologists, transportation workers, and service connection specialists. She noted that previous military experience can affect radiation therapy. For example, the physical restraints required during treatment present particular challenges for veterans who’ve had wartime trauma. These patients may require therapy adjustments.
What’s next on the horizon? Takiar highlighted precision oncology and molecular profiling, artificial intelligence in care decisions and in radiation planning, telemedicine and virtual tumor boards, and expanded survivorship programs.
As for now, she urged colleagues to not be afraid to chat with radiation oncologists. “Please talk to us. We prioritize open communication and shared decision-making with the entire team,” she said. “If you see something and think your radiation oncologist should know about it, you think it was caused by the radiation, you should reach out to us.”
Takiar reported no disclosures.
Team-Based Care is Crucial for Head-and-Neck Cancer Cases
Team-Based Care is Crucial for Head-and-Neck Cancer Cases
Artificial Intelligence Shows Promise in Detecting Missed Interval Breast Cancer on Screening Mammograms
TOPLINE:
An artificial intelligence (AI) system flagged high-risk areas on mammograms for potentially missed interval breast cancers (IBCs), which radiologists had also retrospectively identified as abnormal. Moreover, the AI detected a substantial number of IBCs that manual review had overlooked.
METHODOLOGY:
- Researchers conducted a retrospective analysis of 119 IBC screening mammograms of women (mean age, 57.3 years) with a high breast density (Breast Imaging Reporting and Data System [BI-RADS] c/d, 63.0%) using data retrieved from Cancer Registries of Eastern Switzerland and Grisons-Glarus databases.
- A recorded tumour was classified as IBC when an invasive or in situ BC was diagnosed within 24 months after a normal screening mammogram.
- Three radiologists retrospectively assessed the mammograms for visible signs of BC, which were then classified as either potentially missed IBCs or IBCs without retrospective abnormalities on the basis of consensus conference recommendations of radiologists.
- An AI system generated two scores (a scale of 0 to 100): a case score reflecting the likelihood that the mammogram currently harbours cancer and a risk score estimating the probability of a BC diagnosis within 2 years.
TAKEAWAY:
- Radiologists classified 68.9% of IBCs as those having no retrospective abnormalities and assigned significantly higher BI-RADS scores to the remaining 31.1% of potentially missed IBCs (P < .05).
- Potentially missed IBCs received significantly higher AI case scores (mean, 54.1 vs 23.1; P < .05) and were assigned to a higher risk category (48.7% vs 14.6%; P < .05) than IBCs without retrospective abnormalities.
- Of all IBC cases, 46.2% received an AI case score > 25, 25.2% scored > 50, and 13.4% scored > 75.
- Potentially missed IBCs scored widely between low and high risk and case scores, whereas IBCs without retrospective abnormalities scored low case and risk scores. Specifically, 73.0% of potentially missed IBCs vs 34.1% of IBCs without retrospective abnormalities had case scores > 25, 51.4% vs 13.4% had case scores > 50, and 29.7% vs 6.1% had case scores > 75.
IN PRACTICE:
“Our research highlights that an AI system can identify BC signs in relevant portions of IBC screening mammograms and thus potentially reduce the number of IBCs in an MSP [mammography screening program] that currently does not utilize an AI system,” the authors of the study concluded, adding that “it can identify some IBCs that are not visible to humans (IBCs without retrospective abnormalities).”
SOURCE:
This study was led by Jonas Subelack, Chair of Health Economics, Policy and Management, School of Medicine, University of St. Gallen, St. Gallen, Switzerland. It was published online in European Radiology.
LIMITATIONS:
The retrospective study design inherently limited causal conclusions. Without access to diagnostic mammograms or the detailed position of BC, researchers could not evaluate whether AI-marked lesions corresponded to later detected BCs.
DISCLOSURES:
This research was funded by the Cancer League of Eastern Switzerland. One author reported receiving consulting and speaker fees from iCAD.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
An artificial intelligence (AI) system flagged high-risk areas on mammograms for potentially missed interval breast cancers (IBCs), which radiologists had also retrospectively identified as abnormal. Moreover, the AI detected a substantial number of IBCs that manual review had overlooked.
METHODOLOGY:
- Researchers conducted a retrospective analysis of 119 IBC screening mammograms of women (mean age, 57.3 years) with a high breast density (Breast Imaging Reporting and Data System [BI-RADS] c/d, 63.0%) using data retrieved from Cancer Registries of Eastern Switzerland and Grisons-Glarus databases.
- A recorded tumour was classified as IBC when an invasive or in situ BC was diagnosed within 24 months after a normal screening mammogram.
- Three radiologists retrospectively assessed the mammograms for visible signs of BC, which were then classified as either potentially missed IBCs or IBCs without retrospective abnormalities on the basis of consensus conference recommendations of radiologists.
- An AI system generated two scores (a scale of 0 to 100): a case score reflecting the likelihood that the mammogram currently harbours cancer and a risk score estimating the probability of a BC diagnosis within 2 years.
TAKEAWAY:
- Radiologists classified 68.9% of IBCs as those having no retrospective abnormalities and assigned significantly higher BI-RADS scores to the remaining 31.1% of potentially missed IBCs (P < .05).
- Potentially missed IBCs received significantly higher AI case scores (mean, 54.1 vs 23.1; P < .05) and were assigned to a higher risk category (48.7% vs 14.6%; P < .05) than IBCs without retrospective abnormalities.
- Of all IBC cases, 46.2% received an AI case score > 25, 25.2% scored > 50, and 13.4% scored > 75.
- Potentially missed IBCs scored widely between low and high risk and case scores, whereas IBCs without retrospective abnormalities scored low case and risk scores. Specifically, 73.0% of potentially missed IBCs vs 34.1% of IBCs without retrospective abnormalities had case scores > 25, 51.4% vs 13.4% had case scores > 50, and 29.7% vs 6.1% had case scores > 75.
IN PRACTICE:
“Our research highlights that an AI system can identify BC signs in relevant portions of IBC screening mammograms and thus potentially reduce the number of IBCs in an MSP [mammography screening program] that currently does not utilize an AI system,” the authors of the study concluded, adding that “it can identify some IBCs that are not visible to humans (IBCs without retrospective abnormalities).”
SOURCE:
This study was led by Jonas Subelack, Chair of Health Economics, Policy and Management, School of Medicine, University of St. Gallen, St. Gallen, Switzerland. It was published online in European Radiology.
LIMITATIONS:
The retrospective study design inherently limited causal conclusions. Without access to diagnostic mammograms or the detailed position of BC, researchers could not evaluate whether AI-marked lesions corresponded to later detected BCs.
DISCLOSURES:
This research was funded by the Cancer League of Eastern Switzerland. One author reported receiving consulting and speaker fees from iCAD.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
An artificial intelligence (AI) system flagged high-risk areas on mammograms for potentially missed interval breast cancers (IBCs), which radiologists had also retrospectively identified as abnormal. Moreover, the AI detected a substantial number of IBCs that manual review had overlooked.
METHODOLOGY:
- Researchers conducted a retrospective analysis of 119 IBC screening mammograms of women (mean age, 57.3 years) with a high breast density (Breast Imaging Reporting and Data System [BI-RADS] c/d, 63.0%) using data retrieved from Cancer Registries of Eastern Switzerland and Grisons-Glarus databases.
- A recorded tumour was classified as IBC when an invasive or in situ BC was diagnosed within 24 months after a normal screening mammogram.
- Three radiologists retrospectively assessed the mammograms for visible signs of BC, which were then classified as either potentially missed IBCs or IBCs without retrospective abnormalities on the basis of consensus conference recommendations of radiologists.
- An AI system generated two scores (a scale of 0 to 100): a case score reflecting the likelihood that the mammogram currently harbours cancer and a risk score estimating the probability of a BC diagnosis within 2 years.
TAKEAWAY:
- Radiologists classified 68.9% of IBCs as those having no retrospective abnormalities and assigned significantly higher BI-RADS scores to the remaining 31.1% of potentially missed IBCs (P < .05).
- Potentially missed IBCs received significantly higher AI case scores (mean, 54.1 vs 23.1; P < .05) and were assigned to a higher risk category (48.7% vs 14.6%; P < .05) than IBCs without retrospective abnormalities.
- Of all IBC cases, 46.2% received an AI case score > 25, 25.2% scored > 50, and 13.4% scored > 75.
- Potentially missed IBCs scored widely between low and high risk and case scores, whereas IBCs without retrospective abnormalities scored low case and risk scores. Specifically, 73.0% of potentially missed IBCs vs 34.1% of IBCs without retrospective abnormalities had case scores > 25, 51.4% vs 13.4% had case scores > 50, and 29.7% vs 6.1% had case scores > 75.
IN PRACTICE:
“Our research highlights that an AI system can identify BC signs in relevant portions of IBC screening mammograms and thus potentially reduce the number of IBCs in an MSP [mammography screening program] that currently does not utilize an AI system,” the authors of the study concluded, adding that “it can identify some IBCs that are not visible to humans (IBCs without retrospective abnormalities).”
SOURCE:
This study was led by Jonas Subelack, Chair of Health Economics, Policy and Management, School of Medicine, University of St. Gallen, St. Gallen, Switzerland. It was published online in European Radiology.
LIMITATIONS:
The retrospective study design inherently limited causal conclusions. Without access to diagnostic mammograms or the detailed position of BC, researchers could not evaluate whether AI-marked lesions corresponded to later detected BCs.
DISCLOSURES:
This research was funded by the Cancer League of Eastern Switzerland. One author reported receiving consulting and speaker fees from iCAD.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
Atypical Intrathoracic Manifestations of Metastatic Prostate Cancer: A Case Series
Atypical Intrathoracic Manifestations of Metastatic Prostate Cancer: A Case Series
Prostate cancer is the most common noncutaneous cancer in men, accounting for 29% of all incident cancer cases.1 Typically, prostate cancer metastasizes to bone and regional lymph nodes.2 However, intrathoracic manifestation may occur. This report presents 3 cases of rare intrathoracic manifestations of metastatic prostate cancer with a review of the current literature.
CASE PRESENTATIONS
Case 1
A 71-year-old male who was an active smoker and a long-standing employment as a plumber was diagnosed with rectal cancer in 2022. He completed neoadjuvant capecitabine and radiation therapy followed by a rectosigmoidectomy. Several weeks after surgery, the patient presented to the emergency department (ED) with a dry cough and worsening shortness of breath. Point-of-care ultrasound of the lungs revealed a moderate right pleural effusion with several nodular pleural masses. A chest computed tomography (CT) confirmed these findings (Figure 1). A CT of the abdomen and pelvis revealed prostatomegaly with the medial lobe of the prostate protruding into the bladder; however, no enlarged retroperitoneal, mesenteric or pelvic lymph nodes were noted. The patient underwent a right pleural fluid drainage and pleural mass biopsy. Pleural mass histomorphology as well as immunohistochemical (IHC) stains were consistent with metastatic prostate adenocarcinoma. The pleural fluid cytology also was consistent with metastatic prostate adenocarcinoma.
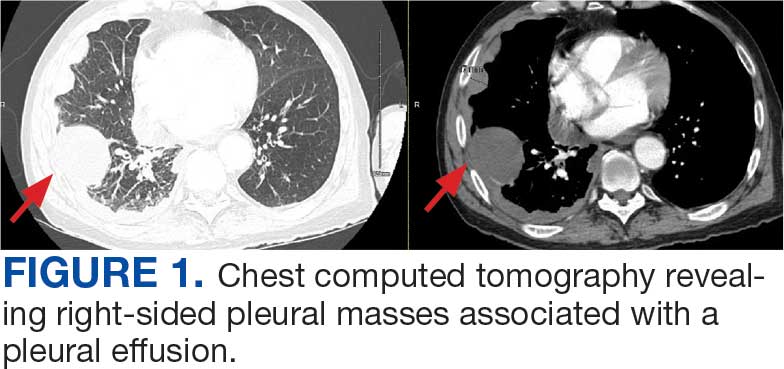
Immunohistochemistry showed weak positive staining for prostate-specific NK3 homeobox 1 gene (NKX3.1), alpha-methylacyl-CoA racemase gene (AMACR), and prosaposin, and negative transcription termination factor (TTF-1), keratin-7 (CK7), and prosaposin, and negative transcription termination factor (TTF-1), keratin-7 (CK7), keratin-20, and caudal type homeobox 2 gene (CDX2) (Figure 2) 2). The patient's prostate-specific antigen (PSA) was found to be elevated at 33.9 ng/mL (reference range, < 4 ng/mL).
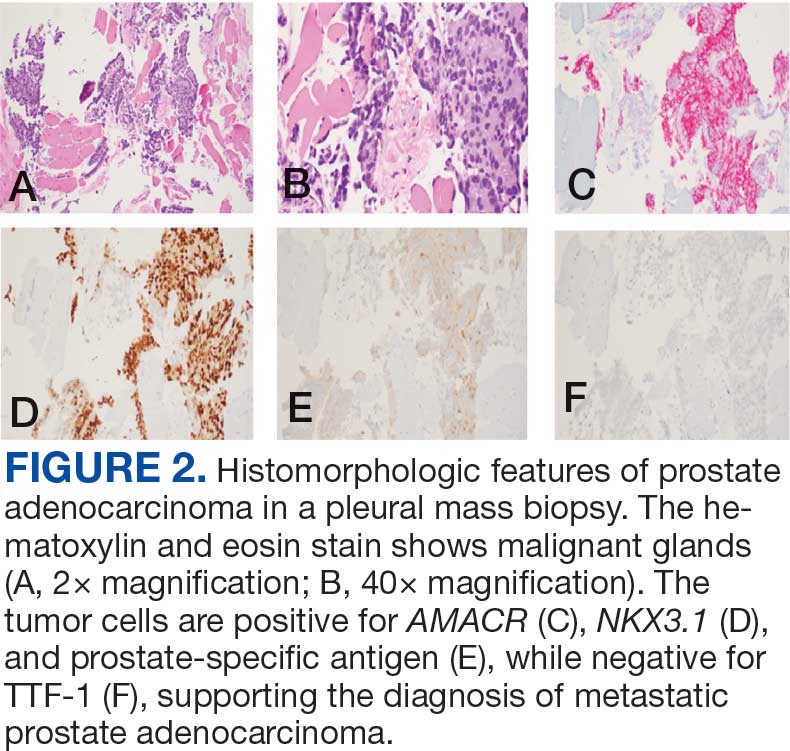
Case 2
A 71-year-old male with a history of alcohol use disorder and a 30-year smoking history presented to the ED with worsening dyspnea on exertion. The patient’s baseline exercise tolerance decreased to walking for only 1 block. He reported unintentional weight loss of about 30 pounds over the prior year, no recent respiratory infections, no prior breathing problems, and no personal or family history of cancer. Chest CT revealed findings of bilateral peribronchial opacities as well as mediastinal and hilar lymphadenopathy (Figure 3). The patient developed hypoxic respiratory failure necessitating intubation, mechanical ventilation, and management in the medical intensive care unit, where he was treated for postobstructive pneumonia. Fiberoptic bronchoscopy revealed endobronchial lesions in the right and left upper lobe that were partially obstructing the airway (Figure 4).
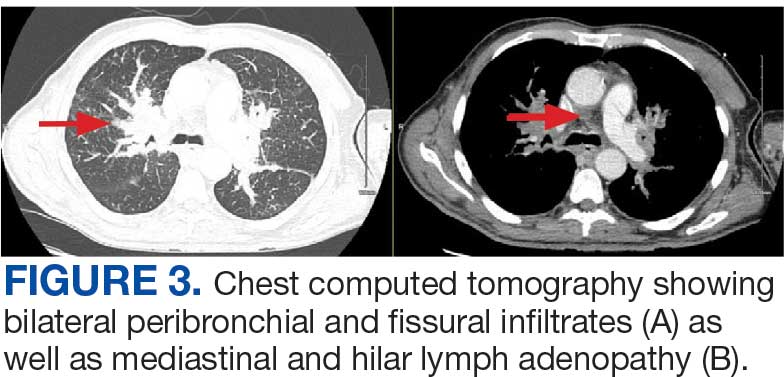
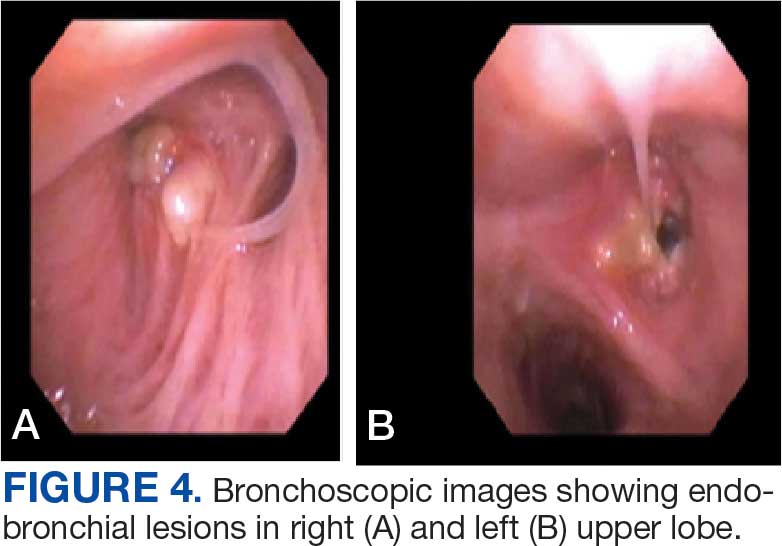
The endobronchial masses were debulked using forceps, and samples were sent for surgical pathology evaluation. Staging was completed using linear endobronchial ultrasound, which revealed an enlarged subcarinal lymph node (S7). The surgical pathology of the endobronchial mass and the subcarinal lymph node cytology were consistent with metastatic adenocarcinoma of the prostate. The tumor cells were positive for AE1/AE3, PSA, and NKX3.1, but were negative for CK7 and TTF-1 (Figure 5). Further imaging revealed an enlarged heterogeneous prostate gland, prominent pelvic nodes, and left retroperitoneal lymphadenopathy, as well as sclerotic foci within the T10 vertebral body and right inferior pubic ramus. PSA was also found to be significantly elevated at 700 ng/mL.
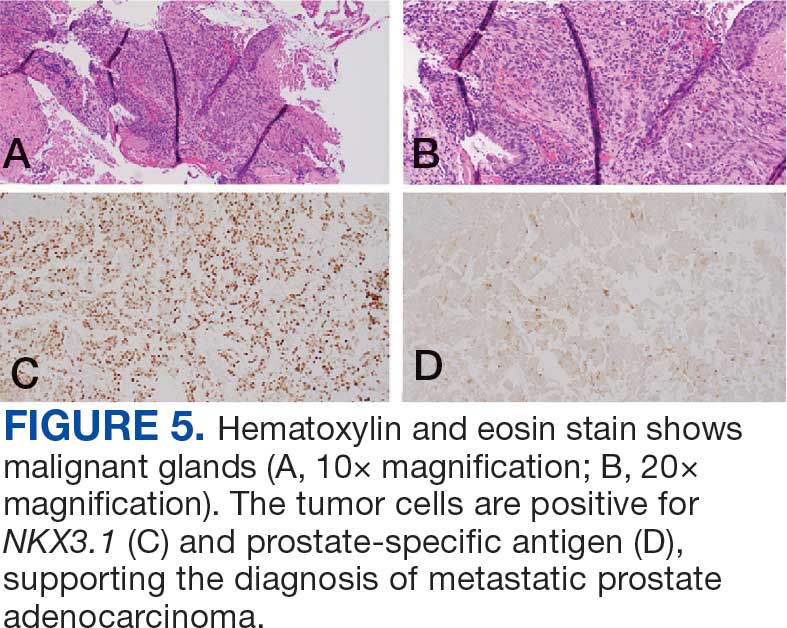
Case 3
An 80-year-old male veteran with a history of prostate cancer and recently diagnosed T2N1M0 head and neck squamous cell carcinoma was referred to the Pulmonary service for evaluation of a pulmonary nodule. His medical history was notable for prostate cancer diagnosed 12 years earlier, with an unknown Gleason score. Initial treatment included prostatectomy followed by whole pelvic radiation therapy a year after, due to elevated PSA in surveillance monitoring. This treatment led to remission. After establishing remission for > 10 years, the patient was started on low-dose testosterone replacement therapy to address complications of radiation therapy, namely hypogonadism.
On evaluation, a chest CT was significant for a large 2-cm right middle lobe nodule (Figure 6). At that time, PSA was noted to be borderline elevated at 4.2 ng/mL, and whole-body imaging did not reveal any lesions elsewhere, specifically no bone metastasis. Biopsies of the right middle lobe lung nodule revealed adenocarcinoma consistent with metastatic prostate cancer. Testosterone therapy was promptly discontinued.
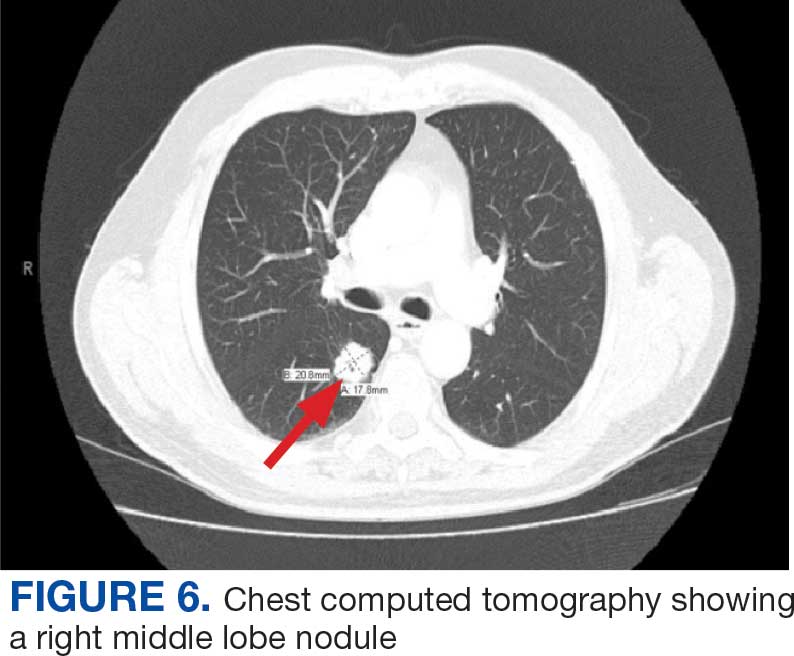
The patient initially refused androgen deprivation therapy owing to the antiandrogenic adverse effects. However, subsequent chest CTs revealed growing lung nodules, which convinced him to proceed with androgen deprivation therapy followed by palliative radiation, and chemotherapy and management of malignant pleural effusion with indwelling small bore pleural catheter for about 10 years. He died from COVID-19 during the pandemic.
DISCUSSION
These cases highlight the importance of including prostate cancer in the differential diagnoses of male patients with intrathoracic abnormalities, even in the absence of metastasis to the more common sites. In a large cohort study of 74,826 patients with metastatic prostate cancer, Gandaglia et al found that the most frequent sites of metastasis were bone (84.0%) and distant lymph nodes (10.6%).2 However, thoracic involvement was observed in 9.1% of cases, with isolated thoracic metastasis being rare. The cases described in this report exemplify exceptionally uncommon occurrences within that 9.1%.
Pleural metastases, as observed in Case 1, are a particularly rare manifestation. In a 10-year retrospective assessment, Vinjamoori et al discovered pleural nodules or masses in only 6 of 82 patients (7.3%) with atypical metastases.3 Adrenal and liver metastases accounted for 15% and 37% of cases with atypical distribution. As such, isolated pleural disease is rare even in atypical presentations.3
As seen in Case 2, endobronchial metastases producing airway obstruction are also rare, with the most common primary cancers associated with endobronchial metastasis being breast, colon, and renal cancer.4 The available literature on this presentation is confined to case reports. Hameed et al reported a case of synchronous biopsy-proven endobronchial metastasis from prostate cancer.5 These cases highlight the importance of maintaining a high level of clinical awareness when encountering endobronchial lesions in patients with prostate cancer.
Case 3 presents a unique situation of lung metastases without any involvement of the bones. It is well known—and was confirmed by Heidenreich et al—that lung metastases in prostate adenocarcinoma usually coincide with extensive osseous disease.6 This instance highlights the importance of watchful monitoring for unusual patterns of cancer recurrence.
Immunohistochemistry stains that are specific to prostate cancer include antibodies against PSA. Prostate-specific membrane antigen is another marker that is far more present in malignant than in benign prostate tissue.
The NKX3.1 gene encodes a homeobox protein, which is a transcription factor and tumor suppressor. In prostate cancer, there is loss of heterozygosity of the gene and stains for the IHC antibody to NKX3.1.7
On the other hand, lung cells stain positive for TTF-1, which is produced by surfactant-producing type 2 pneumocytes and club cells in the lung. Antibodies to TTF-1, a common IHC stain, are used to identify adenocarcinoma of lung origin and may carry a prognostic value.7
The immunohistochemistry profiles, specifically the presence of prostate-specific markers such as PSA and NKX3.1, played a vital role in making the diagnosis.
In Case 1, weak TTF-1 positivity was noted, an unusual finding in metastatic prostate adenocarcinoma. Marak et al documented a rare case of TTF-1–positive metastatic prostate cancer, illustrating the potential for diagnostic confusion with primary lung malignancies.8
The 3 cases described in this report demonstrate the importance of clinical consideration, serial follow-up of PSA levels, using more prostate-specific positron emission tomography tracers (eg, Pylarify) alongside traditional imaging, and tissue biopsy to detect unusual metastases.
CONCLUSIONS
Although thoracic metastases from prostate cancer are rare, these presentations highlight the importance of clinical awareness regarding atypical cases. Pleural disease, endobronchial lesions, and isolated pulmonary nodules might be the first clinical manifestation of metastatic prostate cancer. A high index of suspicion, appropriate imaging, and judicious use of immunohistochemistry are important to ensure accurate diagnosis and optimal patient management.
- Siegel RL, Giaquinto AN, Jemal A. Cancer statistics, 2024. CA Cancer J Clin. 2024;74(1):12-49. doi:10.3322/caac.21820
- Gandaglia G, Abdollah F, Schiffmann J, et al. Distribution of metastatic sites in patients with prostate cancer: a population-based analysis. Prostate. 2014;74(2):210-216. doi:10.1002/pros.22742
- Vinjamoori AH, Jagannathan JP, Shinagare AB, et al. Atypical metastases from prostate cancer: 10-year experience at a single institution. AJR Am J Roentgenol. 2012;199(2):367-372. doi:10.2214/AJR.11.7533
- Salud A, Porcel JM, Rovirosa A, Bellmunt J. Endobronchial metastatic disease: analysis of 32 cases. J Surg Oncol. 1996;62(4):249-252. doi:10.1002/(SICI)1096- 9098(199608)62:4<249::AID-JSO4>3.0.CO;2-6
- Hameed M, Haq IU, Yousaf M, Hussein M, Rashid U, Al-Bozom I. Endobronchial metastases secondary to prostate cancer: a case report and literature review. Respir Med Case Rep. 2020;32:101326. doi:10.1016/j.rmcr.2020.101326
- Heidenreich A, Bastian PJ, Bellmunt J, et al; for the European Association of Urology. EAU guidelines on prostate cancer. Part II: treatment of advanced, relapsing, and castration- resistant prostate cancer. Eur Urol. 2014;65(2):467- 479. doi:10.1016/j.eururo.2013.11.002
- Schallenberg S, Dernbach G, Dragomir MP, et al. TTF-1 status in early-stage lung adenocarcinoma is an independent predictor of relapse and survival superior to tumor grading. Eur J Cancer. 2024;197:113474. doi:10.1016/j.ejca.2023.113474
- Marak C, Guddati AK, Ashraf A, Smith J, Kaushik P. Prostate adenocarcinoma with atypical immunohistochemistry presenting with a Cheerio sign. AIM Clinical Cases. 2023;1:e220508. doi:10.7326/aimcc.2022.0508
Prostate cancer is the most common noncutaneous cancer in men, accounting for 29% of all incident cancer cases.1 Typically, prostate cancer metastasizes to bone and regional lymph nodes.2 However, intrathoracic manifestation may occur. This report presents 3 cases of rare intrathoracic manifestations of metastatic prostate cancer with a review of the current literature.
CASE PRESENTATIONS
Case 1
A 71-year-old male who was an active smoker and a long-standing employment as a plumber was diagnosed with rectal cancer in 2022. He completed neoadjuvant capecitabine and radiation therapy followed by a rectosigmoidectomy. Several weeks after surgery, the patient presented to the emergency department (ED) with a dry cough and worsening shortness of breath. Point-of-care ultrasound of the lungs revealed a moderate right pleural effusion with several nodular pleural masses. A chest computed tomography (CT) confirmed these findings (Figure 1). A CT of the abdomen and pelvis revealed prostatomegaly with the medial lobe of the prostate protruding into the bladder; however, no enlarged retroperitoneal, mesenteric or pelvic lymph nodes were noted. The patient underwent a right pleural fluid drainage and pleural mass biopsy. Pleural mass histomorphology as well as immunohistochemical (IHC) stains were consistent with metastatic prostate adenocarcinoma. The pleural fluid cytology also was consistent with metastatic prostate adenocarcinoma.

Immunohistochemistry showed weak positive staining for prostate-specific NK3 homeobox 1 gene (NKX3.1), alpha-methylacyl-CoA racemase gene (AMACR), and prosaposin, and negative transcription termination factor (TTF-1), keratin-7 (CK7), and prosaposin, and negative transcription termination factor (TTF-1), keratin-7 (CK7), keratin-20, and caudal type homeobox 2 gene (CDX2) (Figure 2) 2). The patient's prostate-specific antigen (PSA) was found to be elevated at 33.9 ng/mL (reference range, < 4 ng/mL).

Case 2
A 71-year-old male with a history of alcohol use disorder and a 30-year smoking history presented to the ED with worsening dyspnea on exertion. The patient’s baseline exercise tolerance decreased to walking for only 1 block. He reported unintentional weight loss of about 30 pounds over the prior year, no recent respiratory infections, no prior breathing problems, and no personal or family history of cancer. Chest CT revealed findings of bilateral peribronchial opacities as well as mediastinal and hilar lymphadenopathy (Figure 3). The patient developed hypoxic respiratory failure necessitating intubation, mechanical ventilation, and management in the medical intensive care unit, where he was treated for postobstructive pneumonia. Fiberoptic bronchoscopy revealed endobronchial lesions in the right and left upper lobe that were partially obstructing the airway (Figure 4).


The endobronchial masses were debulked using forceps, and samples were sent for surgical pathology evaluation. Staging was completed using linear endobronchial ultrasound, which revealed an enlarged subcarinal lymph node (S7). The surgical pathology of the endobronchial mass and the subcarinal lymph node cytology were consistent with metastatic adenocarcinoma of the prostate. The tumor cells were positive for AE1/AE3, PSA, and NKX3.1, but were negative for CK7 and TTF-1 (Figure 5). Further imaging revealed an enlarged heterogeneous prostate gland, prominent pelvic nodes, and left retroperitoneal lymphadenopathy, as well as sclerotic foci within the T10 vertebral body and right inferior pubic ramus. PSA was also found to be significantly elevated at 700 ng/mL.

Case 3
An 80-year-old male veteran with a history of prostate cancer and recently diagnosed T2N1M0 head and neck squamous cell carcinoma was referred to the Pulmonary service for evaluation of a pulmonary nodule. His medical history was notable for prostate cancer diagnosed 12 years earlier, with an unknown Gleason score. Initial treatment included prostatectomy followed by whole pelvic radiation therapy a year after, due to elevated PSA in surveillance monitoring. This treatment led to remission. After establishing remission for > 10 years, the patient was started on low-dose testosterone replacement therapy to address complications of radiation therapy, namely hypogonadism.
On evaluation, a chest CT was significant for a large 2-cm right middle lobe nodule (Figure 6). At that time, PSA was noted to be borderline elevated at 4.2 ng/mL, and whole-body imaging did not reveal any lesions elsewhere, specifically no bone metastasis. Biopsies of the right middle lobe lung nodule revealed adenocarcinoma consistent with metastatic prostate cancer. Testosterone therapy was promptly discontinued.

The patient initially refused androgen deprivation therapy owing to the antiandrogenic adverse effects. However, subsequent chest CTs revealed growing lung nodules, which convinced him to proceed with androgen deprivation therapy followed by palliative radiation, and chemotherapy and management of malignant pleural effusion with indwelling small bore pleural catheter for about 10 years. He died from COVID-19 during the pandemic.
DISCUSSION
These cases highlight the importance of including prostate cancer in the differential diagnoses of male patients with intrathoracic abnormalities, even in the absence of metastasis to the more common sites. In a large cohort study of 74,826 patients with metastatic prostate cancer, Gandaglia et al found that the most frequent sites of metastasis were bone (84.0%) and distant lymph nodes (10.6%).2 However, thoracic involvement was observed in 9.1% of cases, with isolated thoracic metastasis being rare. The cases described in this report exemplify exceptionally uncommon occurrences within that 9.1%.
Pleural metastases, as observed in Case 1, are a particularly rare manifestation. In a 10-year retrospective assessment, Vinjamoori et al discovered pleural nodules or masses in only 6 of 82 patients (7.3%) with atypical metastases.3 Adrenal and liver metastases accounted for 15% and 37% of cases with atypical distribution. As such, isolated pleural disease is rare even in atypical presentations.3
As seen in Case 2, endobronchial metastases producing airway obstruction are also rare, with the most common primary cancers associated with endobronchial metastasis being breast, colon, and renal cancer.4 The available literature on this presentation is confined to case reports. Hameed et al reported a case of synchronous biopsy-proven endobronchial metastasis from prostate cancer.5 These cases highlight the importance of maintaining a high level of clinical awareness when encountering endobronchial lesions in patients with prostate cancer.
Case 3 presents a unique situation of lung metastases without any involvement of the bones. It is well known—and was confirmed by Heidenreich et al—that lung metastases in prostate adenocarcinoma usually coincide with extensive osseous disease.6 This instance highlights the importance of watchful monitoring for unusual patterns of cancer recurrence.
Immunohistochemistry stains that are specific to prostate cancer include antibodies against PSA. Prostate-specific membrane antigen is another marker that is far more present in malignant than in benign prostate tissue.
The NKX3.1 gene encodes a homeobox protein, which is a transcription factor and tumor suppressor. In prostate cancer, there is loss of heterozygosity of the gene and stains for the IHC antibody to NKX3.1.7
On the other hand, lung cells stain positive for TTF-1, which is produced by surfactant-producing type 2 pneumocytes and club cells in the lung. Antibodies to TTF-1, a common IHC stain, are used to identify adenocarcinoma of lung origin and may carry a prognostic value.7
The immunohistochemistry profiles, specifically the presence of prostate-specific markers such as PSA and NKX3.1, played a vital role in making the diagnosis.
In Case 1, weak TTF-1 positivity was noted, an unusual finding in metastatic prostate adenocarcinoma. Marak et al documented a rare case of TTF-1–positive metastatic prostate cancer, illustrating the potential for diagnostic confusion with primary lung malignancies.8
The 3 cases described in this report demonstrate the importance of clinical consideration, serial follow-up of PSA levels, using more prostate-specific positron emission tomography tracers (eg, Pylarify) alongside traditional imaging, and tissue biopsy to detect unusual metastases.
CONCLUSIONS
Although thoracic metastases from prostate cancer are rare, these presentations highlight the importance of clinical awareness regarding atypical cases. Pleural disease, endobronchial lesions, and isolated pulmonary nodules might be the first clinical manifestation of metastatic prostate cancer. A high index of suspicion, appropriate imaging, and judicious use of immunohistochemistry are important to ensure accurate diagnosis and optimal patient management.
Prostate cancer is the most common noncutaneous cancer in men, accounting for 29% of all incident cancer cases.1 Typically, prostate cancer metastasizes to bone and regional lymph nodes.2 However, intrathoracic manifestation may occur. This report presents 3 cases of rare intrathoracic manifestations of metastatic prostate cancer with a review of the current literature.
CASE PRESENTATIONS
Case 1
A 71-year-old male who was an active smoker and a long-standing employment as a plumber was diagnosed with rectal cancer in 2022. He completed neoadjuvant capecitabine and radiation therapy followed by a rectosigmoidectomy. Several weeks after surgery, the patient presented to the emergency department (ED) with a dry cough and worsening shortness of breath. Point-of-care ultrasound of the lungs revealed a moderate right pleural effusion with several nodular pleural masses. A chest computed tomography (CT) confirmed these findings (Figure 1). A CT of the abdomen and pelvis revealed prostatomegaly with the medial lobe of the prostate protruding into the bladder; however, no enlarged retroperitoneal, mesenteric or pelvic lymph nodes were noted. The patient underwent a right pleural fluid drainage and pleural mass biopsy. Pleural mass histomorphology as well as immunohistochemical (IHC) stains were consistent with metastatic prostate adenocarcinoma. The pleural fluid cytology also was consistent with metastatic prostate adenocarcinoma.

Immunohistochemistry showed weak positive staining for prostate-specific NK3 homeobox 1 gene (NKX3.1), alpha-methylacyl-CoA racemase gene (AMACR), and prosaposin, and negative transcription termination factor (TTF-1), keratin-7 (CK7), and prosaposin, and negative transcription termination factor (TTF-1), keratin-7 (CK7), keratin-20, and caudal type homeobox 2 gene (CDX2) (Figure 2) 2). The patient's prostate-specific antigen (PSA) was found to be elevated at 33.9 ng/mL (reference range, < 4 ng/mL).

Case 2
A 71-year-old male with a history of alcohol use disorder and a 30-year smoking history presented to the ED with worsening dyspnea on exertion. The patient’s baseline exercise tolerance decreased to walking for only 1 block. He reported unintentional weight loss of about 30 pounds over the prior year, no recent respiratory infections, no prior breathing problems, and no personal or family history of cancer. Chest CT revealed findings of bilateral peribronchial opacities as well as mediastinal and hilar lymphadenopathy (Figure 3). The patient developed hypoxic respiratory failure necessitating intubation, mechanical ventilation, and management in the medical intensive care unit, where he was treated for postobstructive pneumonia. Fiberoptic bronchoscopy revealed endobronchial lesions in the right and left upper lobe that were partially obstructing the airway (Figure 4).


The endobronchial masses were debulked using forceps, and samples were sent for surgical pathology evaluation. Staging was completed using linear endobronchial ultrasound, which revealed an enlarged subcarinal lymph node (S7). The surgical pathology of the endobronchial mass and the subcarinal lymph node cytology were consistent with metastatic adenocarcinoma of the prostate. The tumor cells were positive for AE1/AE3, PSA, and NKX3.1, but were negative for CK7 and TTF-1 (Figure 5). Further imaging revealed an enlarged heterogeneous prostate gland, prominent pelvic nodes, and left retroperitoneal lymphadenopathy, as well as sclerotic foci within the T10 vertebral body and right inferior pubic ramus. PSA was also found to be significantly elevated at 700 ng/mL.

Case 3
An 80-year-old male veteran with a history of prostate cancer and recently diagnosed T2N1M0 head and neck squamous cell carcinoma was referred to the Pulmonary service for evaluation of a pulmonary nodule. His medical history was notable for prostate cancer diagnosed 12 years earlier, with an unknown Gleason score. Initial treatment included prostatectomy followed by whole pelvic radiation therapy a year after, due to elevated PSA in surveillance monitoring. This treatment led to remission. After establishing remission for > 10 years, the patient was started on low-dose testosterone replacement therapy to address complications of radiation therapy, namely hypogonadism.
On evaluation, a chest CT was significant for a large 2-cm right middle lobe nodule (Figure 6). At that time, PSA was noted to be borderline elevated at 4.2 ng/mL, and whole-body imaging did not reveal any lesions elsewhere, specifically no bone metastasis. Biopsies of the right middle lobe lung nodule revealed adenocarcinoma consistent with metastatic prostate cancer. Testosterone therapy was promptly discontinued.

The patient initially refused androgen deprivation therapy owing to the antiandrogenic adverse effects. However, subsequent chest CTs revealed growing lung nodules, which convinced him to proceed with androgen deprivation therapy followed by palliative radiation, and chemotherapy and management of malignant pleural effusion with indwelling small bore pleural catheter for about 10 years. He died from COVID-19 during the pandemic.
DISCUSSION
These cases highlight the importance of including prostate cancer in the differential diagnoses of male patients with intrathoracic abnormalities, even in the absence of metastasis to the more common sites. In a large cohort study of 74,826 patients with metastatic prostate cancer, Gandaglia et al found that the most frequent sites of metastasis were bone (84.0%) and distant lymph nodes (10.6%).2 However, thoracic involvement was observed in 9.1% of cases, with isolated thoracic metastasis being rare. The cases described in this report exemplify exceptionally uncommon occurrences within that 9.1%.
Pleural metastases, as observed in Case 1, are a particularly rare manifestation. In a 10-year retrospective assessment, Vinjamoori et al discovered pleural nodules or masses in only 6 of 82 patients (7.3%) with atypical metastases.3 Adrenal and liver metastases accounted for 15% and 37% of cases with atypical distribution. As such, isolated pleural disease is rare even in atypical presentations.3
As seen in Case 2, endobronchial metastases producing airway obstruction are also rare, with the most common primary cancers associated with endobronchial metastasis being breast, colon, and renal cancer.4 The available literature on this presentation is confined to case reports. Hameed et al reported a case of synchronous biopsy-proven endobronchial metastasis from prostate cancer.5 These cases highlight the importance of maintaining a high level of clinical awareness when encountering endobronchial lesions in patients with prostate cancer.
Case 3 presents a unique situation of lung metastases without any involvement of the bones. It is well known—and was confirmed by Heidenreich et al—that lung metastases in prostate adenocarcinoma usually coincide with extensive osseous disease.6 This instance highlights the importance of watchful monitoring for unusual patterns of cancer recurrence.
Immunohistochemistry stains that are specific to prostate cancer include antibodies against PSA. Prostate-specific membrane antigen is another marker that is far more present in malignant than in benign prostate tissue.
The NKX3.1 gene encodes a homeobox protein, which is a transcription factor and tumor suppressor. In prostate cancer, there is loss of heterozygosity of the gene and stains for the IHC antibody to NKX3.1.7
On the other hand, lung cells stain positive for TTF-1, which is produced by surfactant-producing type 2 pneumocytes and club cells in the lung. Antibodies to TTF-1, a common IHC stain, are used to identify adenocarcinoma of lung origin and may carry a prognostic value.7
The immunohistochemistry profiles, specifically the presence of prostate-specific markers such as PSA and NKX3.1, played a vital role in making the diagnosis.
In Case 1, weak TTF-1 positivity was noted, an unusual finding in metastatic prostate adenocarcinoma. Marak et al documented a rare case of TTF-1–positive metastatic prostate cancer, illustrating the potential for diagnostic confusion with primary lung malignancies.8
The 3 cases described in this report demonstrate the importance of clinical consideration, serial follow-up of PSA levels, using more prostate-specific positron emission tomography tracers (eg, Pylarify) alongside traditional imaging, and tissue biopsy to detect unusual metastases.
CONCLUSIONS
Although thoracic metastases from prostate cancer are rare, these presentations highlight the importance of clinical awareness regarding atypical cases. Pleural disease, endobronchial lesions, and isolated pulmonary nodules might be the first clinical manifestation of metastatic prostate cancer. A high index of suspicion, appropriate imaging, and judicious use of immunohistochemistry are important to ensure accurate diagnosis and optimal patient management.
- Siegel RL, Giaquinto AN, Jemal A. Cancer statistics, 2024. CA Cancer J Clin. 2024;74(1):12-49. doi:10.3322/caac.21820
- Gandaglia G, Abdollah F, Schiffmann J, et al. Distribution of metastatic sites in patients with prostate cancer: a population-based analysis. Prostate. 2014;74(2):210-216. doi:10.1002/pros.22742
- Vinjamoori AH, Jagannathan JP, Shinagare AB, et al. Atypical metastases from prostate cancer: 10-year experience at a single institution. AJR Am J Roentgenol. 2012;199(2):367-372. doi:10.2214/AJR.11.7533
- Salud A, Porcel JM, Rovirosa A, Bellmunt J. Endobronchial metastatic disease: analysis of 32 cases. J Surg Oncol. 1996;62(4):249-252. doi:10.1002/(SICI)1096- 9098(199608)62:4<249::AID-JSO4>3.0.CO;2-6
- Hameed M, Haq IU, Yousaf M, Hussein M, Rashid U, Al-Bozom I. Endobronchial metastases secondary to prostate cancer: a case report and literature review. Respir Med Case Rep. 2020;32:101326. doi:10.1016/j.rmcr.2020.101326
- Heidenreich A, Bastian PJ, Bellmunt J, et al; for the European Association of Urology. EAU guidelines on prostate cancer. Part II: treatment of advanced, relapsing, and castration- resistant prostate cancer. Eur Urol. 2014;65(2):467- 479. doi:10.1016/j.eururo.2013.11.002
- Schallenberg S, Dernbach G, Dragomir MP, et al. TTF-1 status in early-stage lung adenocarcinoma is an independent predictor of relapse and survival superior to tumor grading. Eur J Cancer. 2024;197:113474. doi:10.1016/j.ejca.2023.113474
- Marak C, Guddati AK, Ashraf A, Smith J, Kaushik P. Prostate adenocarcinoma with atypical immunohistochemistry presenting with a Cheerio sign. AIM Clinical Cases. 2023;1:e220508. doi:10.7326/aimcc.2022.0508
- Siegel RL, Giaquinto AN, Jemal A. Cancer statistics, 2024. CA Cancer J Clin. 2024;74(1):12-49. doi:10.3322/caac.21820
- Gandaglia G, Abdollah F, Schiffmann J, et al. Distribution of metastatic sites in patients with prostate cancer: a population-based analysis. Prostate. 2014;74(2):210-216. doi:10.1002/pros.22742
- Vinjamoori AH, Jagannathan JP, Shinagare AB, et al. Atypical metastases from prostate cancer: 10-year experience at a single institution. AJR Am J Roentgenol. 2012;199(2):367-372. doi:10.2214/AJR.11.7533
- Salud A, Porcel JM, Rovirosa A, Bellmunt J. Endobronchial metastatic disease: analysis of 32 cases. J Surg Oncol. 1996;62(4):249-252. doi:10.1002/(SICI)1096- 9098(199608)62:4<249::AID-JSO4>3.0.CO;2-6
- Hameed M, Haq IU, Yousaf M, Hussein M, Rashid U, Al-Bozom I. Endobronchial metastases secondary to prostate cancer: a case report and literature review. Respir Med Case Rep. 2020;32:101326. doi:10.1016/j.rmcr.2020.101326
- Heidenreich A, Bastian PJ, Bellmunt J, et al; for the European Association of Urology. EAU guidelines on prostate cancer. Part II: treatment of advanced, relapsing, and castration- resistant prostate cancer. Eur Urol. 2014;65(2):467- 479. doi:10.1016/j.eururo.2013.11.002
- Schallenberg S, Dernbach G, Dragomir MP, et al. TTF-1 status in early-stage lung adenocarcinoma is an independent predictor of relapse and survival superior to tumor grading. Eur J Cancer. 2024;197:113474. doi:10.1016/j.ejca.2023.113474
- Marak C, Guddati AK, Ashraf A, Smith J, Kaushik P. Prostate adenocarcinoma with atypical immunohistochemistry presenting with a Cheerio sign. AIM Clinical Cases. 2023;1:e220508. doi:10.7326/aimcc.2022.0508
Atypical Intrathoracic Manifestations of Metastatic Prostate Cancer: A Case Series
Atypical Intrathoracic Manifestations of Metastatic Prostate Cancer: A Case Series
Comprehensive Genomic Profiles of Melanoma in Veterans Compared to Reference Databases
Comprehensive Genomic Profiles of Melanoma in Veterans Compared to Reference Databases
The veteran population, with its unique and diverse types of exposure and military service experiences, faces distinct health factors compared with the general population. These factors can be categorized into exposures during military service and those occurring postservice. While the latter phase incorporates psychological issues that may arise while transitioning to civilian life, the service period is associated with major physical, chemical, and psychological exposures that can impact veterans’ health. Carcinogenesis related to military exposures is concerning, and different types of malignancies have been associated with military exposures.1 The 2022 introduction of the Cancer Moonshot initiative served as a breeding ground for multiple projects aimed at investigation of exposure-related carcinogenesis, prompting increased attention and efforts to linking specific exposures to specific malignancies.2
Melanoma is the deadliest skin cancer, accounting for 1.3% of all cancer deaths.3 Although it may only account for 1% to 5% of skin cancer diagnoses, its incidence in the United States’ population has been increasing.4,5 There were 97,610 estimated new cases of melanoma in 2023, according to the National Cancer Institute.6
The incidence of melanoma may be higher in the military population compared with the general population.7 Melanoma is the fourth-most common cancer diagnosed in veterans.8
Several demographic characteristics of the US military population are associated with higher melanoma incidence and poorer prognosis, including male sex, older age, and White race. Apart from sun exposure—a known risk factor for melanoma development—other factors, such as service branch, seem to contribute to risk, with the highest melanoma rates noted in the Air Force.9 According to a study by Chang et al, veterans have a higher risk of stage III (18%) or stage IV (13%) melanoma at initial diagnosis.8
Molecular testing of metastatic melanoma is currently the standard of care for guiding the use of US Food and Drug Administration-approved targeted therapies such as BRAF, MEK, and KIT inhibitors. This comparative analysis details the melanoma comprehensive genomic profiles observed at a large US Department of Veterans Affairs (VA) medical center (VAMC) and those reported in reference databases.
Methods
A query to select all metastatic melanomas sent for comprehensive genomic profiling from the Kansas City VAMC (KCVAMC), identified 35 cases from 2019 through 2023 as the study population. The health records of these patients were reviewed to collect demographic information, military service history, melanoma history, other medical, social, and family histories. The comprehensive genomic profiling reports were reviewed to collect the reported pathogenic variants, microsatellite instability (MSI) status, and tumor mutational burden (TMB) for each case.
The Catalogue of Somatic Mutations in Cancer (COSMIC) was used to identify the most commonly mutated genes in melanomas from The Cancer Genome Atlas for the general population.4,5 The literature was consulted to determine the MSI status and TMB in melanomas from The Cancer Genome Atlas for separate reference populations.6,7 The frequency of MSI-high (MSI-H) status, TMB ≥ 10 mutations/megabase (mut/Mb), and mutations in each of the 20 most commonly mutated genes was determined and compared between melanomas from The Cancer Genome Atlas and KCVAMC cases. Corresponding P values were calculated to identify significant differences. Values were calculated for the entire sample as well as a subgroup with Agent Orange (AO) exposure. The study was approved by the KCVAMC Institutional Review Board.
Results
The mean (SD) age of study participants was 72.9 (9.4) years (range, 39-90 years). The mean (SD) duration of military service was 1654 (1421) days (about 4 years, 6 months, and 10 days). Of the 35 patients included, 22 (63%) served during the Vietnam era (November 1, 1965, to April 30, 1975) and 2 (6%) served during the Persian Gulf War era (August 2, 1990, to February 28, 1991). Seventeen veterans (49%) served in the Army, 9 in the Navy (26%), 5 in the Air Force (14%), and 4 in the Marine Corps (11%). Definitive AO exposure was noted in 13 patients (37%) (Table 1).
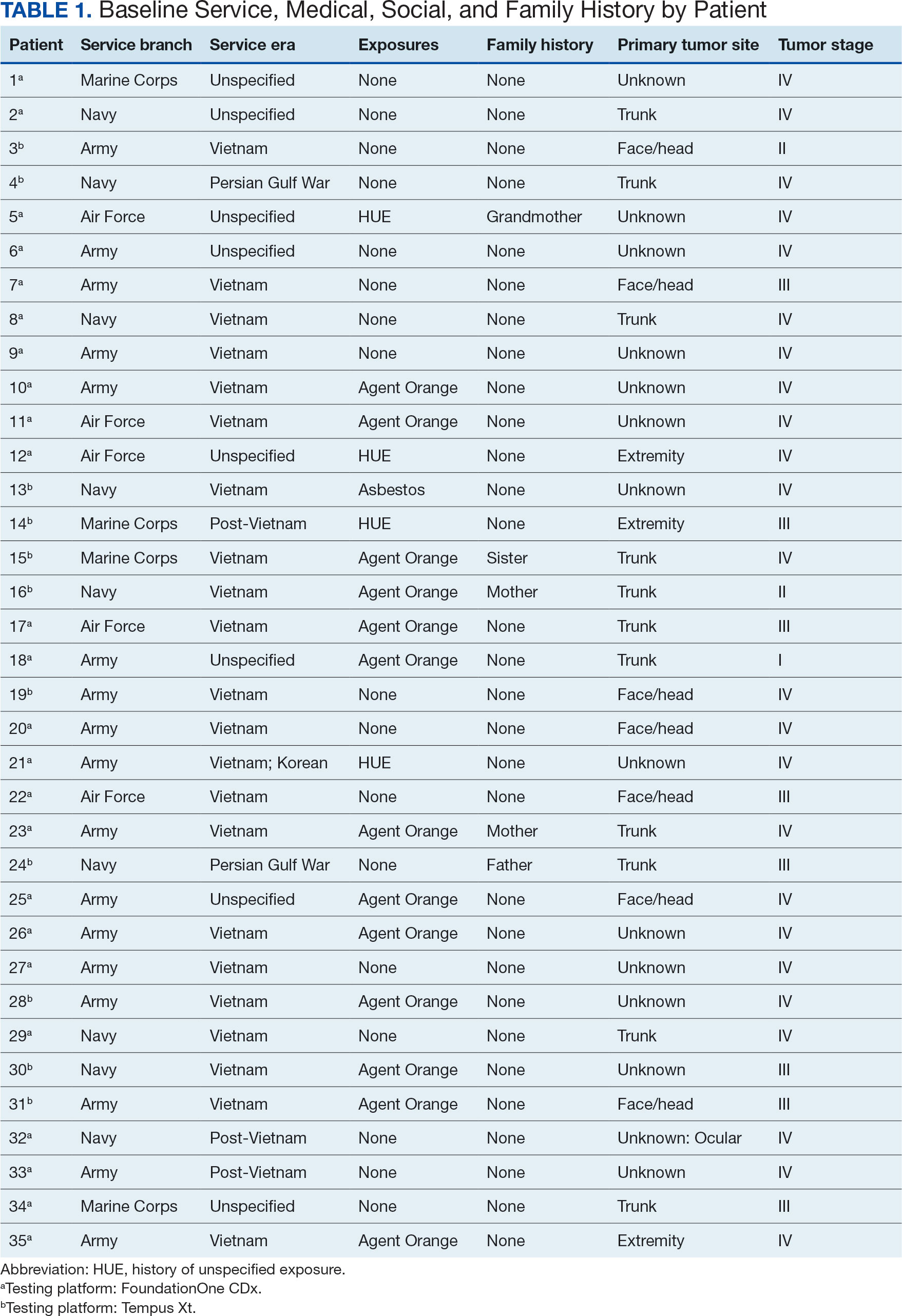
Of the 35 patients, 24 (69%) had metastatic disease and the primary site of melanoma was unknown in 14 patients (40%). One patient (Patient 32) had an intraocular melanoma. The primary site was the trunk for 11 patients (31%), the face/head for 7 patients (20%) and extremities for 3 patients (9%). Eight patients (23%) were pT3 stage (thickness > 2 mm but < 4 mm), 7 patients (20%) were pT4 stage (thickness > 4 mm), and 5 patients (14%) were pT1 (thickness ≥ 1 mm). One patient had a primary lesion at pT2 stage, and 1 had a Tis stage lesion. Three patients (9%) had a family history of melanoma in a first-degree relative.
The list of genes mutated in melanoma cells in the study population is provided in the eAppendix.10,11 Twenty-seven patients (77%) had mutations in TERT promoter, 15 (43%) in CDKN2A/B, 13 (37%) in BRAF, 11 (31%) in NF1, 9 (26%) in TP53, and 8 (23%) in NRAS (Table 2). The majority of mutations in TERT promoter were c.- 146C>T (18 of 27 patients [67%]), whereas c.-124C>T was the second-most common (8 of 27 patients [30%]). The 2 observed mutations in the 13 patients with BRAF mutations were V600E and V600K, with almost equal distribution (54% and 46%, respectively). The mean (SD) TMB was 33.2 (39) mut/Mb (range, 1-203 mut/Mb). Ten patients (29%) had a TMB < 10 mut/Mb, whereas 24 (69%) had a TMB > 10 mut/Mb. The TMB could not be determined in 1 case. The frequency of TMB-high tumors in the study population compared with frequency in the reference population is shown in Table 3.12 Only 3 patients (0.64%) in the reference population had MSI-H tumors, and the microsatellite status could not be determined in those tumors (Table 4).13 Table 5 outlines statistically significant findings.
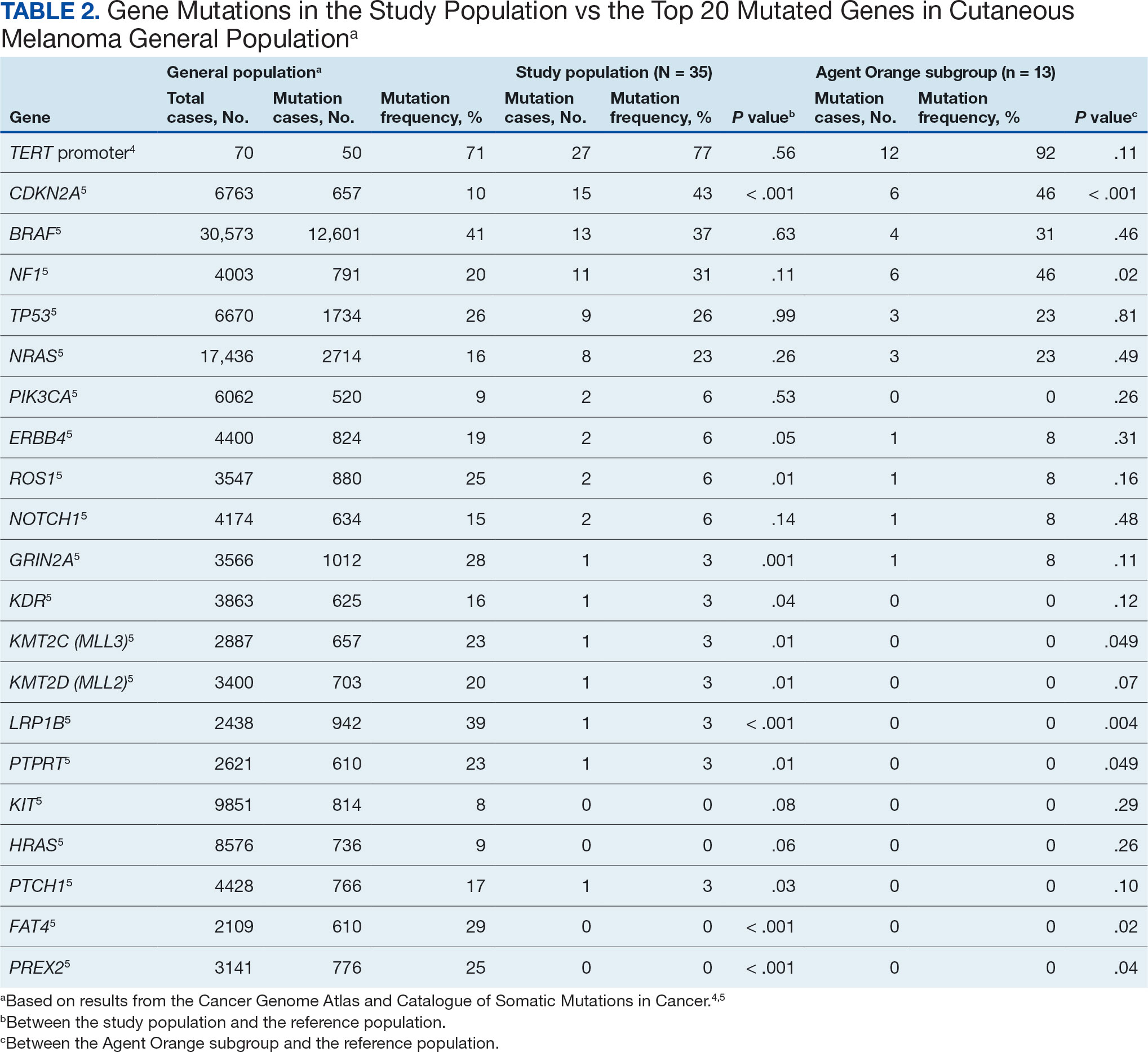
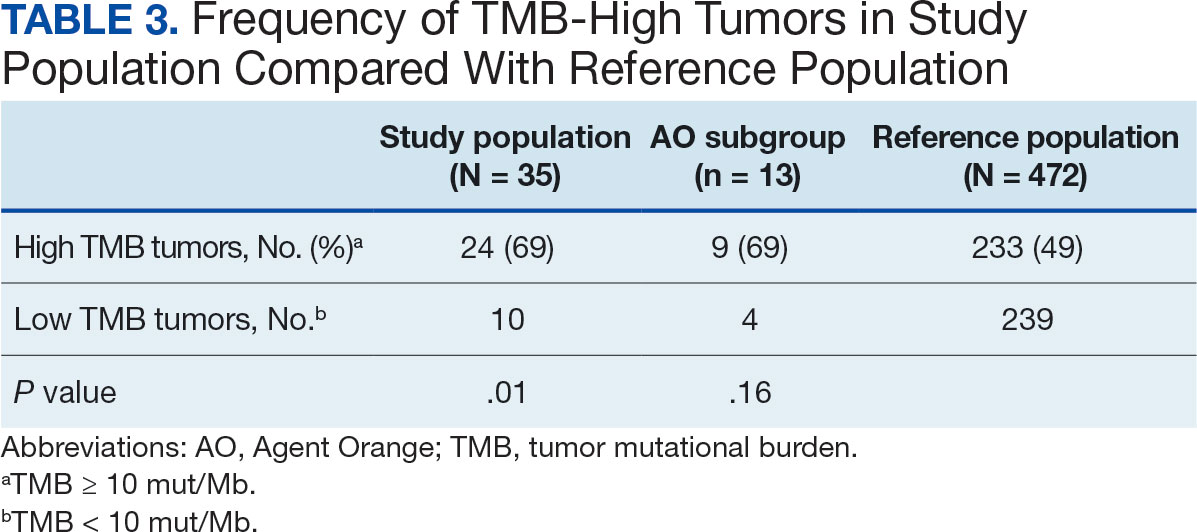

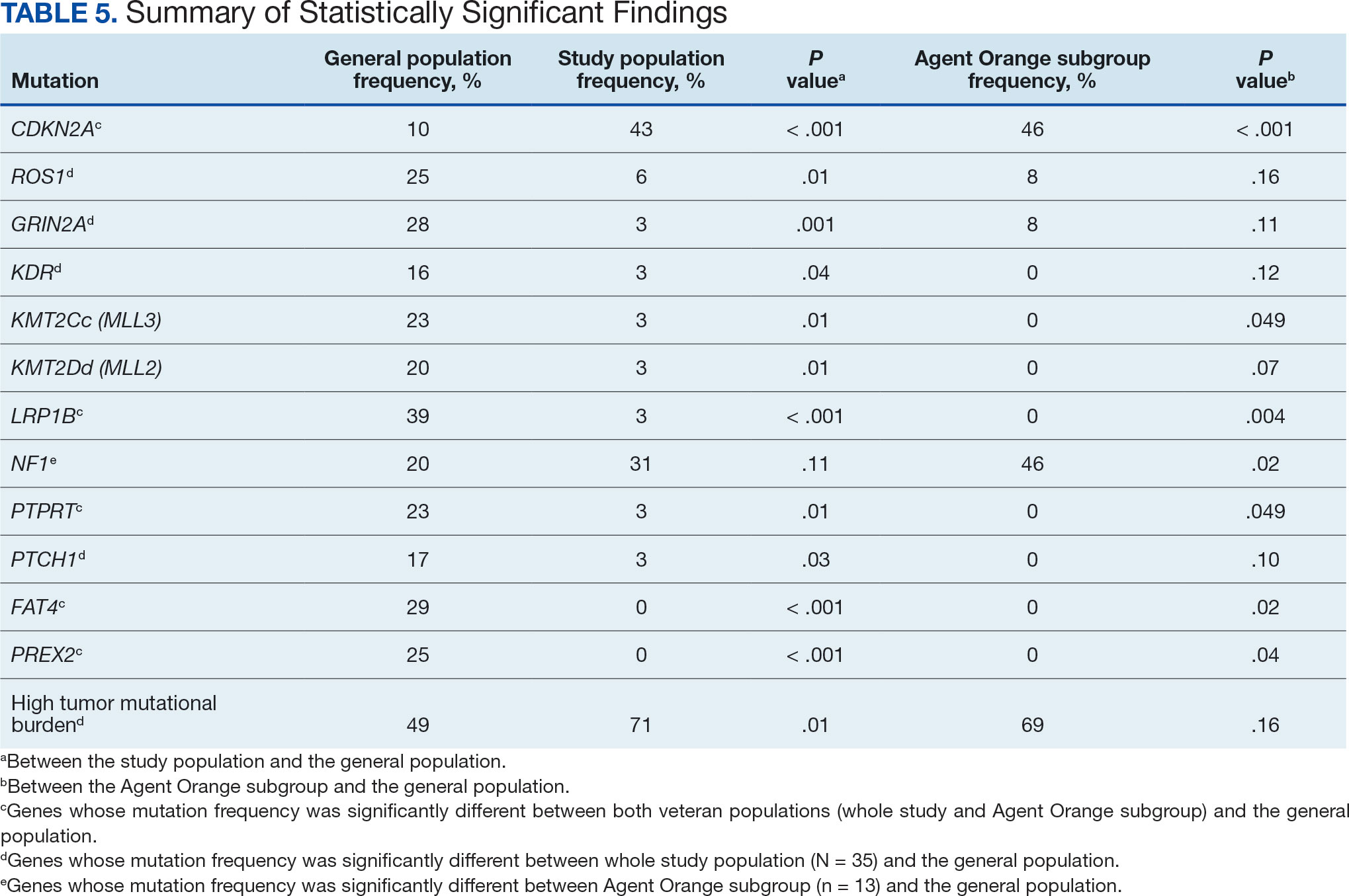
Agent Orange Subgroup
AO was a tactical herbicide used by the US military, named for the orange band around the storage barrels. Possible mutagenic properties of AO have been attributed to its byproduct, dioxin. Among the most common cancers known to be associated with AO exposure are bladder and prostate carcinoma and hematopoietic neoplasms. The association between genetic alterations and AO exposure was studied in veterans with prostate cancer.14 However, to our knowledge, insufficient information is available to determine whether an association exists between exposure to herbicides used in Vietnam or the contaminant dioxin and melanoma. Because a significant proportion of this study population had a well-documented history of AO exposure (37.1%), we were able to analyze them as a subgroup and to separately compare their mutation frequency with the general population.
Results were notable for different distributions of the most frequently mutated genes in the AO subgroup compared with the whole study population. As such, TERT promoter remained the most frequently mutated gene (92%), followed by CDKN2A/B (46%); however, frequency of mutations in NF1 (46%) outnumbered those of BRAF (31%), the fourth-most common mutation. Moreover, when compared with the general melanoma population, a significantly higher frequency of mutations in the NF1 gene was observed in the AO subgroup—not the entire study population.
Discussion
Given that veterans constitute a distinct population, there is reasonable interest in investigating characteristic health issues related to military service. Skin cancer—melanoma in particular—has been researched recently in a veteran population. The differences in demographics, tumor characteristics, and melanoma- specific survival in veterans compared with the general population have already been assessed. According to Chang et al, compared with the general population, veterans are more likely to present with metastatic disease and have lower 5-year survival rates.8
Melanoma is one of the most highly mutated malignancies.15 Fortunately, the most common mutation in melanoma, BRAF V600E, is now considered therapeutically targetable. However, there are still many mutations that are less often discussed and not well understood. Regardless of therapeutic implications, all mutations observed in melanoma are worth investigating because a tumor’s genomic profile also can provide prognostic and etiologic information. Developing comprehensive descriptions of melanoma mutational profiles in specific populations is critical to advancing etiologic understanding and informing prevention strategies.
Our results demonstrate the high prevalence of TERT promoter mutations with characteristic ultraviolet signature (C>T) in the study population. This aligns with general evidence that TERT promoter mutations are common in cutaneous melanomas: 77% of this study sample and up to 86% of all mutations are TERT promoter mutations, according to Davis et al.15 TERT promoter mutations are positively associated with the initiation, invasion, and metastasis of melanoma. In certain subtypes, there is evidence that the presence of TERT promoter mutations is significantly associated with risk for extranodal metastasis and death.16 The second-most common mutated gene in the veteran study population was CDKN2A/B (43%), and the third-most mutated gene was BRAF (37%).
In chronically sun-exposed skin NF1, NRAS, and occasionally BRAF V600K mutations tend to predominate. BRAF V600E mutations, on the other hand, are rare in these melanomas.15 In our study population, the most prevalent melanoma site was the trunk (31%), which is considered a location with an intermittent pattern of sun exposure.17
This study population also had a higher frequency of CDKN2A/B mutations. High frequencies of CDKN2A/B mutations have been reported in familial melanomas, but only 1 patient with CDKN2A/B mutations had a known family history of melanoma.15 Tumors in the study population showed significantly lower frequency of mutations in ROS1, GRIN2A, KDR, KMT2C (MLL3), KMT2D (MLL2), LRP1B, PTPRT, PTCH1, FAT4, and PREX2 (P < .05).
In this study the subgroup of veterans with AO exposure differed from the whole study population. As such, CDKN2A/B mutations were observed with the same frequency as NF1 mutations (46% each); however, BRAF mutations constituted only 31% of the mutations. In addition, the frequency of NF1 mutations was significantly higher in the AO subgroup compared with the general population, but not in the whole study population.
Our sample also differed from the reference population by showing a significantly higher frequency of TMB-high (ie, ≥ 10 mut/Mb) tumors (71% vs 49%; P = .01).12 Interestingly, no significant difference in the frequency of TMB-high tumors was observed between the AO subgroup and the reference population (69% vs 49%; P = .16). There also was no statistically significant difference between the frequency of MSI-H tumors in our study population and the reference population (P = .64).13
One patient in the study population had uveal melanoma. Mutations encountered in this patient’s tumor differed from the general mutational profile of tumors. None of the 21 mutations depicted in Table 2 were present in this sample.10,11 On the other hand, those mutations frequently observed in intraocular melanomas, BAP1 and GNA11, were present in this patient.18 Additionally, this particular melanoma possessed mutations in genes RICTOR, RAD21, and PIK3R1.
Limitations
This study population consisted exclusively of male patients, introducing sex as a potential confounder in analyzing differences between the study population and the general population. As noted in a 2020 systematic review, there were no sex-based differences in the frequency of mutations in BRAF, NRAS, and KIT genes.19
Regarding NF1 mutations, only NF1-mutated acral and mucosal melanomas were more frequently observed in female patients, whereas nonacral NF1-mutated melanomas were more frequently observed in male patients.20 However, there is currently no clear evidence of whether the mutational landscapes of cutaneous melanoma differ by sex.21 Among the 11 cases with NF1-mutatation, site of origin was known in 6, 5 of which originated at nonacral sites. Although the AO subgroup also consisted entirely of male patients, this does not explain the observed increased frequency of NF1 mutations relative to the general population. No such difference was observed between the whole study population, which also consisted exclusively of male patients, and the general population. The similar frequencies of nonacral location in the whole study population (3 acral, 18 nonacral, 14 unknown site of origin) and AO subgroup (1 acral, 7 nonacral, 5 unknown site of origin) preclude location as an explanation.
The Cancer Genome Atlas Network proposed a framework for genomic classification of melanoma into 4 subtypes based on the pattern of the most prevalent significantly mutated genes: mutant BRAF, mutant RAS, mutant NF1, and triple–wild-type. According to that study, BRAF mutations were indeed associated with younger age, in contrast to the NF1-mutant genomic subtype, which was more prevalent in older individuals with higher TMB.22 This emphasizes the need to interpret the potential association of AO exposure and NF1 mutation in melanoma with caution, although additional studies are required to observe the difference between the veteran population and age-matched general population.
On the other hand, Yu et al reported no significant differences of TMB values between patients aged < 60 and ≥ 60 years with melanoma.23 In short, the observed differences we report in our limited study warrant additional investigation with larger sample sizes, sex-matched controlling, and age-matched controlling. The study was limited by its small sample size and the single location.
Conclusion
The genomic profile of melanomas in the veteran population appears to be similar to that of the general population with a few possible differences. Melanomas in the veteran study population showed a higher frequency of CDKN2A/B mutations; lower frequency of ROS1, GRIN2A, KDR, KMT2C (MLL3), KMT2D (MLL2), LRP1B, PTPRT, PTCH1, FAT4, and PREX2 mutations; and higher TMB. In addition, melanomas in the AO subgroup showed higher frequencies of NF1 mutations. The significance of such findings remains to be determined by further investigation.
- Bytnar JA, McGlynn KA, et al. Cancer incidence in the US military: An updated analysis. Cancer. 2024;130(1):96-106. doi:10.1002/cncr.34978
- Singer DS. A new phase of the Cancer Moonshot to end cancer as we know it. Nat Med. 2022;28(7):1345-1347. doi:10.1038/s41591-022-01881-5
- Koczkodaj P, Sulkowska U, Didkowska J, et al. Melanoma mortality trends in 28 European countries: a retrospective analysis for the years 1960-2020. Cancers (Basel). 2023;15(5):1514. Published 2023 Feb 28. doi:10.3390/cancers15051514
- Okobi OE, Abreo E, Sams NP, et al. Trends in melanoma incidence, prevalence, stage at diagnosis, and survival: an analysis of the United States Cancer Statistics (USCS) database. Cureus. 2024;16(10):e70697. doi:10.7759/cureus.70697
- Bartling SJ, Rivard SC, Meyerle JH. Melanoma in an active duty marine. Mil Med. 2017;182:e2034-e2039. doi:10.7205/MILMED-D-17-00127
- American Cancer Society. Cancer facts & figures 2023. American Cancer Society; 2023. Accessed June 20, 2025. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2023/2023-cancer-facts-and-figures.pdf
- Rezaei SJ, Kim J, Onyeka S, et al. Skin cancer and other dermatologic conditions among US veterans. JAMA Dermatol. 2024;160(10):1107-1111. doi:10.1001/jamadermatol.2024.3043
- Chang MS, La J, Trepanowski N, et al. Increased relative proportions of advanced melanoma among veterans: a comparative analysis with the Surveillance, Epidemiology, and End Results registry. J Am Acad Dermatol. 2022;87:72-79. doi:10.1016/j.jaad.2022.02.063
- Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78:1185-1192. doi:10.1016/j.jaad.2017.11.062
- Huang FW, Hodis E, Xu MJ, et al. Highly recurrent TERT promoter mutations in human melanoma. Science. 2013;339:957-959. doi:10.1126/science.1229259
- Tate JG, Bamford S, Jubb HC, et al. COSMIC: the Catalogue of Somatic Mutations in Cancer. Nucleic Acids Res. 2019;47:D941-D947. doi:10.1093/nar/gky1015
- Li M, Gao X, Wang X. Identification of tumor mutation burden-associated molecular and clinical features in cancer by analyzing multi-omics data. Front Immunol. 2023;14:1090838. doi:10.3389/fimmu.2023.1090838
- Bonneville R, Krook MA, Kautto EA, et al. Landscape of microsatellite instability across 39 cancer types. JCO Precis Oncol. 2017;2017:PO.17.00073. doi:10.1200/PO.17.00073
- Lui AJ, Pagadala MS, Zhong AY, et al. Agent Orange exposure and prostate cancer risk in the Million Veteran Program. medRxiv [Preprint]. 2023:2023.06.14.23291413. doi:10.1101/2023.06.14.23291413
- Davis EJ, Johnson DB, Sosman JA, et al. Melanoma: what do all the mutations mean? Cancer. 2018;124:3490-3499. doi:10.1002/cncr.31345
- Guo Y, Chen Y, Zhang L, et al. TERT promoter mutations and telomerase in melanoma. J Oncol. 2022;2022:6300329. doi:10.1155/2022/6300329
- Whiteman DC, Stickley M, Watt P, et al. Anatomic site, sun exposure, and risk of cutaneous melanoma. J Clin Oncol. 2006;24:3172-3177. doi:10.1200/JCO.2006.06.1325
- Decatur CL, Ong E, Garg N, et al. Driver mutations in uveal melanoma: associations with gene expression profile and patient outcomes. JAMA Ophthalmol. 2016;134:728-733. doi:10.1001/jamaophthalmol.2016.0903
- Gutiérrez-Castañeda LD, Nova JA, Tovar-Parra JD. Frequency of mutations in BRAF, NRAS, and KIT in different populations and histological subtypes of melanoma: a systemic review. Melanoma Res. 2020;30:62- 70. doi:10.1097/CMR.0000000000000628
- Thielmann CM, Chorti E, Matull J, et al. NF1-mutated melanomas reveal distinct clinical characteristics depending on tumour origin and respond favourably to immune checkpoint inhibitors. Eur J Cancer. 2021;159:113-124. doi:10.1016/j.ejca.2021.09.035
- D’Ecclesiis O, Caini S, Martinoli C, et al. Gender-dependent specificities in cutaneous melanoma predisposition, risk factors, somatic mutations, prognostic and predictive factors: a systematic review. Int J Environ Res Public Health. 2021;18:7945. doi:10.3390/ijerph18157945
- Cancer Genome Atlas Network. Genomic classification of cutaneous melanoma. Cell. 2015;161:1681-1696. doi:10.1016/j.cell.2015.05.044
- Yu Z, Wang J, Feng L, et al. Association of tumor mutational burden with age in solid tumors. J Clin Oncol. 2020;38:e13590-e13590. doi:10.1200/JCO.2020.38.15_suppl.e13590
The veteran population, with its unique and diverse types of exposure and military service experiences, faces distinct health factors compared with the general population. These factors can be categorized into exposures during military service and those occurring postservice. While the latter phase incorporates psychological issues that may arise while transitioning to civilian life, the service period is associated with major physical, chemical, and psychological exposures that can impact veterans’ health. Carcinogenesis related to military exposures is concerning, and different types of malignancies have been associated with military exposures.1 The 2022 introduction of the Cancer Moonshot initiative served as a breeding ground for multiple projects aimed at investigation of exposure-related carcinogenesis, prompting increased attention and efforts to linking specific exposures to specific malignancies.2
Melanoma is the deadliest skin cancer, accounting for 1.3% of all cancer deaths.3 Although it may only account for 1% to 5% of skin cancer diagnoses, its incidence in the United States’ population has been increasing.4,5 There were 97,610 estimated new cases of melanoma in 2023, according to the National Cancer Institute.6
The incidence of melanoma may be higher in the military population compared with the general population.7 Melanoma is the fourth-most common cancer diagnosed in veterans.8
Several demographic characteristics of the US military population are associated with higher melanoma incidence and poorer prognosis, including male sex, older age, and White race. Apart from sun exposure—a known risk factor for melanoma development—other factors, such as service branch, seem to contribute to risk, with the highest melanoma rates noted in the Air Force.9 According to a study by Chang et al, veterans have a higher risk of stage III (18%) or stage IV (13%) melanoma at initial diagnosis.8
Molecular testing of metastatic melanoma is currently the standard of care for guiding the use of US Food and Drug Administration-approved targeted therapies such as BRAF, MEK, and KIT inhibitors. This comparative analysis details the melanoma comprehensive genomic profiles observed at a large US Department of Veterans Affairs (VA) medical center (VAMC) and those reported in reference databases.
Methods
A query to select all metastatic melanomas sent for comprehensive genomic profiling from the Kansas City VAMC (KCVAMC), identified 35 cases from 2019 through 2023 as the study population. The health records of these patients were reviewed to collect demographic information, military service history, melanoma history, other medical, social, and family histories. The comprehensive genomic profiling reports were reviewed to collect the reported pathogenic variants, microsatellite instability (MSI) status, and tumor mutational burden (TMB) for each case.
The Catalogue of Somatic Mutations in Cancer (COSMIC) was used to identify the most commonly mutated genes in melanomas from The Cancer Genome Atlas for the general population.4,5 The literature was consulted to determine the MSI status and TMB in melanomas from The Cancer Genome Atlas for separate reference populations.6,7 The frequency of MSI-high (MSI-H) status, TMB ≥ 10 mutations/megabase (mut/Mb), and mutations in each of the 20 most commonly mutated genes was determined and compared between melanomas from The Cancer Genome Atlas and KCVAMC cases. Corresponding P values were calculated to identify significant differences. Values were calculated for the entire sample as well as a subgroup with Agent Orange (AO) exposure. The study was approved by the KCVAMC Institutional Review Board.
Results
The mean (SD) age of study participants was 72.9 (9.4) years (range, 39-90 years). The mean (SD) duration of military service was 1654 (1421) days (about 4 years, 6 months, and 10 days). Of the 35 patients included, 22 (63%) served during the Vietnam era (November 1, 1965, to April 30, 1975) and 2 (6%) served during the Persian Gulf War era (August 2, 1990, to February 28, 1991). Seventeen veterans (49%) served in the Army, 9 in the Navy (26%), 5 in the Air Force (14%), and 4 in the Marine Corps (11%). Definitive AO exposure was noted in 13 patients (37%) (Table 1).

Of the 35 patients, 24 (69%) had metastatic disease and the primary site of melanoma was unknown in 14 patients (40%). One patient (Patient 32) had an intraocular melanoma. The primary site was the trunk for 11 patients (31%), the face/head for 7 patients (20%) and extremities for 3 patients (9%). Eight patients (23%) were pT3 stage (thickness > 2 mm but < 4 mm), 7 patients (20%) were pT4 stage (thickness > 4 mm), and 5 patients (14%) were pT1 (thickness ≥ 1 mm). One patient had a primary lesion at pT2 stage, and 1 had a Tis stage lesion. Three patients (9%) had a family history of melanoma in a first-degree relative.
The list of genes mutated in melanoma cells in the study population is provided in the eAppendix.10,11 Twenty-seven patients (77%) had mutations in TERT promoter, 15 (43%) in CDKN2A/B, 13 (37%) in BRAF, 11 (31%) in NF1, 9 (26%) in TP53, and 8 (23%) in NRAS (Table 2). The majority of mutations in TERT promoter were c.- 146C>T (18 of 27 patients [67%]), whereas c.-124C>T was the second-most common (8 of 27 patients [30%]). The 2 observed mutations in the 13 patients with BRAF mutations were V600E and V600K, with almost equal distribution (54% and 46%, respectively). The mean (SD) TMB was 33.2 (39) mut/Mb (range, 1-203 mut/Mb). Ten patients (29%) had a TMB < 10 mut/Mb, whereas 24 (69%) had a TMB > 10 mut/Mb. The TMB could not be determined in 1 case. The frequency of TMB-high tumors in the study population compared with frequency in the reference population is shown in Table 3.12 Only 3 patients (0.64%) in the reference population had MSI-H tumors, and the microsatellite status could not be determined in those tumors (Table 4).13 Table 5 outlines statistically significant findings.




Agent Orange Subgroup
AO was a tactical herbicide used by the US military, named for the orange band around the storage barrels. Possible mutagenic properties of AO have been attributed to its byproduct, dioxin. Among the most common cancers known to be associated with AO exposure are bladder and prostate carcinoma and hematopoietic neoplasms. The association between genetic alterations and AO exposure was studied in veterans with prostate cancer.14 However, to our knowledge, insufficient information is available to determine whether an association exists between exposure to herbicides used in Vietnam or the contaminant dioxin and melanoma. Because a significant proportion of this study population had a well-documented history of AO exposure (37.1%), we were able to analyze them as a subgroup and to separately compare their mutation frequency with the general population.
Results were notable for different distributions of the most frequently mutated genes in the AO subgroup compared with the whole study population. As such, TERT promoter remained the most frequently mutated gene (92%), followed by CDKN2A/B (46%); however, frequency of mutations in NF1 (46%) outnumbered those of BRAF (31%), the fourth-most common mutation. Moreover, when compared with the general melanoma population, a significantly higher frequency of mutations in the NF1 gene was observed in the AO subgroup—not the entire study population.
Discussion
Given that veterans constitute a distinct population, there is reasonable interest in investigating characteristic health issues related to military service. Skin cancer—melanoma in particular—has been researched recently in a veteran population. The differences in demographics, tumor characteristics, and melanoma- specific survival in veterans compared with the general population have already been assessed. According to Chang et al, compared with the general population, veterans are more likely to present with metastatic disease and have lower 5-year survival rates.8
Melanoma is one of the most highly mutated malignancies.15 Fortunately, the most common mutation in melanoma, BRAF V600E, is now considered therapeutically targetable. However, there are still many mutations that are less often discussed and not well understood. Regardless of therapeutic implications, all mutations observed in melanoma are worth investigating because a tumor’s genomic profile also can provide prognostic and etiologic information. Developing comprehensive descriptions of melanoma mutational profiles in specific populations is critical to advancing etiologic understanding and informing prevention strategies.
Our results demonstrate the high prevalence of TERT promoter mutations with characteristic ultraviolet signature (C>T) in the study population. This aligns with general evidence that TERT promoter mutations are common in cutaneous melanomas: 77% of this study sample and up to 86% of all mutations are TERT promoter mutations, according to Davis et al.15 TERT promoter mutations are positively associated with the initiation, invasion, and metastasis of melanoma. In certain subtypes, there is evidence that the presence of TERT promoter mutations is significantly associated with risk for extranodal metastasis and death.16 The second-most common mutated gene in the veteran study population was CDKN2A/B (43%), and the third-most mutated gene was BRAF (37%).
In chronically sun-exposed skin NF1, NRAS, and occasionally BRAF V600K mutations tend to predominate. BRAF V600E mutations, on the other hand, are rare in these melanomas.15 In our study population, the most prevalent melanoma site was the trunk (31%), which is considered a location with an intermittent pattern of sun exposure.17
This study population also had a higher frequency of CDKN2A/B mutations. High frequencies of CDKN2A/B mutations have been reported in familial melanomas, but only 1 patient with CDKN2A/B mutations had a known family history of melanoma.15 Tumors in the study population showed significantly lower frequency of mutations in ROS1, GRIN2A, KDR, KMT2C (MLL3), KMT2D (MLL2), LRP1B, PTPRT, PTCH1, FAT4, and PREX2 (P < .05).
In this study the subgroup of veterans with AO exposure differed from the whole study population. As such, CDKN2A/B mutations were observed with the same frequency as NF1 mutations (46% each); however, BRAF mutations constituted only 31% of the mutations. In addition, the frequency of NF1 mutations was significantly higher in the AO subgroup compared with the general population, but not in the whole study population.
Our sample also differed from the reference population by showing a significantly higher frequency of TMB-high (ie, ≥ 10 mut/Mb) tumors (71% vs 49%; P = .01).12 Interestingly, no significant difference in the frequency of TMB-high tumors was observed between the AO subgroup and the reference population (69% vs 49%; P = .16). There also was no statistically significant difference between the frequency of MSI-H tumors in our study population and the reference population (P = .64).13
One patient in the study population had uveal melanoma. Mutations encountered in this patient’s tumor differed from the general mutational profile of tumors. None of the 21 mutations depicted in Table 2 were present in this sample.10,11 On the other hand, those mutations frequently observed in intraocular melanomas, BAP1 and GNA11, were present in this patient.18 Additionally, this particular melanoma possessed mutations in genes RICTOR, RAD21, and PIK3R1.
Limitations
This study population consisted exclusively of male patients, introducing sex as a potential confounder in analyzing differences between the study population and the general population. As noted in a 2020 systematic review, there were no sex-based differences in the frequency of mutations in BRAF, NRAS, and KIT genes.19
Regarding NF1 mutations, only NF1-mutated acral and mucosal melanomas were more frequently observed in female patients, whereas nonacral NF1-mutated melanomas were more frequently observed in male patients.20 However, there is currently no clear evidence of whether the mutational landscapes of cutaneous melanoma differ by sex.21 Among the 11 cases with NF1-mutatation, site of origin was known in 6, 5 of which originated at nonacral sites. Although the AO subgroup also consisted entirely of male patients, this does not explain the observed increased frequency of NF1 mutations relative to the general population. No such difference was observed between the whole study population, which also consisted exclusively of male patients, and the general population. The similar frequencies of nonacral location in the whole study population (3 acral, 18 nonacral, 14 unknown site of origin) and AO subgroup (1 acral, 7 nonacral, 5 unknown site of origin) preclude location as an explanation.
The Cancer Genome Atlas Network proposed a framework for genomic classification of melanoma into 4 subtypes based on the pattern of the most prevalent significantly mutated genes: mutant BRAF, mutant RAS, mutant NF1, and triple–wild-type. According to that study, BRAF mutations were indeed associated with younger age, in contrast to the NF1-mutant genomic subtype, which was more prevalent in older individuals with higher TMB.22 This emphasizes the need to interpret the potential association of AO exposure and NF1 mutation in melanoma with caution, although additional studies are required to observe the difference between the veteran population and age-matched general population.
On the other hand, Yu et al reported no significant differences of TMB values between patients aged < 60 and ≥ 60 years with melanoma.23 In short, the observed differences we report in our limited study warrant additional investigation with larger sample sizes, sex-matched controlling, and age-matched controlling. The study was limited by its small sample size and the single location.
Conclusion
The genomic profile of melanomas in the veteran population appears to be similar to that of the general population with a few possible differences. Melanomas in the veteran study population showed a higher frequency of CDKN2A/B mutations; lower frequency of ROS1, GRIN2A, KDR, KMT2C (MLL3), KMT2D (MLL2), LRP1B, PTPRT, PTCH1, FAT4, and PREX2 mutations; and higher TMB. In addition, melanomas in the AO subgroup showed higher frequencies of NF1 mutations. The significance of such findings remains to be determined by further investigation.
The veteran population, with its unique and diverse types of exposure and military service experiences, faces distinct health factors compared with the general population. These factors can be categorized into exposures during military service and those occurring postservice. While the latter phase incorporates psychological issues that may arise while transitioning to civilian life, the service period is associated with major physical, chemical, and psychological exposures that can impact veterans’ health. Carcinogenesis related to military exposures is concerning, and different types of malignancies have been associated with military exposures.1 The 2022 introduction of the Cancer Moonshot initiative served as a breeding ground for multiple projects aimed at investigation of exposure-related carcinogenesis, prompting increased attention and efforts to linking specific exposures to specific malignancies.2
Melanoma is the deadliest skin cancer, accounting for 1.3% of all cancer deaths.3 Although it may only account for 1% to 5% of skin cancer diagnoses, its incidence in the United States’ population has been increasing.4,5 There were 97,610 estimated new cases of melanoma in 2023, according to the National Cancer Institute.6
The incidence of melanoma may be higher in the military population compared with the general population.7 Melanoma is the fourth-most common cancer diagnosed in veterans.8
Several demographic characteristics of the US military population are associated with higher melanoma incidence and poorer prognosis, including male sex, older age, and White race. Apart from sun exposure—a known risk factor for melanoma development—other factors, such as service branch, seem to contribute to risk, with the highest melanoma rates noted in the Air Force.9 According to a study by Chang et al, veterans have a higher risk of stage III (18%) or stage IV (13%) melanoma at initial diagnosis.8
Molecular testing of metastatic melanoma is currently the standard of care for guiding the use of US Food and Drug Administration-approved targeted therapies such as BRAF, MEK, and KIT inhibitors. This comparative analysis details the melanoma comprehensive genomic profiles observed at a large US Department of Veterans Affairs (VA) medical center (VAMC) and those reported in reference databases.
Methods
A query to select all metastatic melanomas sent for comprehensive genomic profiling from the Kansas City VAMC (KCVAMC), identified 35 cases from 2019 through 2023 as the study population. The health records of these patients were reviewed to collect demographic information, military service history, melanoma history, other medical, social, and family histories. The comprehensive genomic profiling reports were reviewed to collect the reported pathogenic variants, microsatellite instability (MSI) status, and tumor mutational burden (TMB) for each case.
The Catalogue of Somatic Mutations in Cancer (COSMIC) was used to identify the most commonly mutated genes in melanomas from The Cancer Genome Atlas for the general population.4,5 The literature was consulted to determine the MSI status and TMB in melanomas from The Cancer Genome Atlas for separate reference populations.6,7 The frequency of MSI-high (MSI-H) status, TMB ≥ 10 mutations/megabase (mut/Mb), and mutations in each of the 20 most commonly mutated genes was determined and compared between melanomas from The Cancer Genome Atlas and KCVAMC cases. Corresponding P values were calculated to identify significant differences. Values were calculated for the entire sample as well as a subgroup with Agent Orange (AO) exposure. The study was approved by the KCVAMC Institutional Review Board.
Results
The mean (SD) age of study participants was 72.9 (9.4) years (range, 39-90 years). The mean (SD) duration of military service was 1654 (1421) days (about 4 years, 6 months, and 10 days). Of the 35 patients included, 22 (63%) served during the Vietnam era (November 1, 1965, to April 30, 1975) and 2 (6%) served during the Persian Gulf War era (August 2, 1990, to February 28, 1991). Seventeen veterans (49%) served in the Army, 9 in the Navy (26%), 5 in the Air Force (14%), and 4 in the Marine Corps (11%). Definitive AO exposure was noted in 13 patients (37%) (Table 1).

Of the 35 patients, 24 (69%) had metastatic disease and the primary site of melanoma was unknown in 14 patients (40%). One patient (Patient 32) had an intraocular melanoma. The primary site was the trunk for 11 patients (31%), the face/head for 7 patients (20%) and extremities for 3 patients (9%). Eight patients (23%) were pT3 stage (thickness > 2 mm but < 4 mm), 7 patients (20%) were pT4 stage (thickness > 4 mm), and 5 patients (14%) were pT1 (thickness ≥ 1 mm). One patient had a primary lesion at pT2 stage, and 1 had a Tis stage lesion. Three patients (9%) had a family history of melanoma in a first-degree relative.
The list of genes mutated in melanoma cells in the study population is provided in the eAppendix.10,11 Twenty-seven patients (77%) had mutations in TERT promoter, 15 (43%) in CDKN2A/B, 13 (37%) in BRAF, 11 (31%) in NF1, 9 (26%) in TP53, and 8 (23%) in NRAS (Table 2). The majority of mutations in TERT promoter were c.- 146C>T (18 of 27 patients [67%]), whereas c.-124C>T was the second-most common (8 of 27 patients [30%]). The 2 observed mutations in the 13 patients with BRAF mutations were V600E and V600K, with almost equal distribution (54% and 46%, respectively). The mean (SD) TMB was 33.2 (39) mut/Mb (range, 1-203 mut/Mb). Ten patients (29%) had a TMB < 10 mut/Mb, whereas 24 (69%) had a TMB > 10 mut/Mb. The TMB could not be determined in 1 case. The frequency of TMB-high tumors in the study population compared with frequency in the reference population is shown in Table 3.12 Only 3 patients (0.64%) in the reference population had MSI-H tumors, and the microsatellite status could not be determined in those tumors (Table 4).13 Table 5 outlines statistically significant findings.




Agent Orange Subgroup
AO was a tactical herbicide used by the US military, named for the orange band around the storage barrels. Possible mutagenic properties of AO have been attributed to its byproduct, dioxin. Among the most common cancers known to be associated with AO exposure are bladder and prostate carcinoma and hematopoietic neoplasms. The association between genetic alterations and AO exposure was studied in veterans with prostate cancer.14 However, to our knowledge, insufficient information is available to determine whether an association exists between exposure to herbicides used in Vietnam or the contaminant dioxin and melanoma. Because a significant proportion of this study population had a well-documented history of AO exposure (37.1%), we were able to analyze them as a subgroup and to separately compare their mutation frequency with the general population.
Results were notable for different distributions of the most frequently mutated genes in the AO subgroup compared with the whole study population. As such, TERT promoter remained the most frequently mutated gene (92%), followed by CDKN2A/B (46%); however, frequency of mutations in NF1 (46%) outnumbered those of BRAF (31%), the fourth-most common mutation. Moreover, when compared with the general melanoma population, a significantly higher frequency of mutations in the NF1 gene was observed in the AO subgroup—not the entire study population.
Discussion
Given that veterans constitute a distinct population, there is reasonable interest in investigating characteristic health issues related to military service. Skin cancer—melanoma in particular—has been researched recently in a veteran population. The differences in demographics, tumor characteristics, and melanoma- specific survival in veterans compared with the general population have already been assessed. According to Chang et al, compared with the general population, veterans are more likely to present with metastatic disease and have lower 5-year survival rates.8
Melanoma is one of the most highly mutated malignancies.15 Fortunately, the most common mutation in melanoma, BRAF V600E, is now considered therapeutically targetable. However, there are still many mutations that are less often discussed and not well understood. Regardless of therapeutic implications, all mutations observed in melanoma are worth investigating because a tumor’s genomic profile also can provide prognostic and etiologic information. Developing comprehensive descriptions of melanoma mutational profiles in specific populations is critical to advancing etiologic understanding and informing prevention strategies.
Our results demonstrate the high prevalence of TERT promoter mutations with characteristic ultraviolet signature (C>T) in the study population. This aligns with general evidence that TERT promoter mutations are common in cutaneous melanomas: 77% of this study sample and up to 86% of all mutations are TERT promoter mutations, according to Davis et al.15 TERT promoter mutations are positively associated with the initiation, invasion, and metastasis of melanoma. In certain subtypes, there is evidence that the presence of TERT promoter mutations is significantly associated with risk for extranodal metastasis and death.16 The second-most common mutated gene in the veteran study population was CDKN2A/B (43%), and the third-most mutated gene was BRAF (37%).
In chronically sun-exposed skin NF1, NRAS, and occasionally BRAF V600K mutations tend to predominate. BRAF V600E mutations, on the other hand, are rare in these melanomas.15 In our study population, the most prevalent melanoma site was the trunk (31%), which is considered a location with an intermittent pattern of sun exposure.17
This study population also had a higher frequency of CDKN2A/B mutations. High frequencies of CDKN2A/B mutations have been reported in familial melanomas, but only 1 patient with CDKN2A/B mutations had a known family history of melanoma.15 Tumors in the study population showed significantly lower frequency of mutations in ROS1, GRIN2A, KDR, KMT2C (MLL3), KMT2D (MLL2), LRP1B, PTPRT, PTCH1, FAT4, and PREX2 (P < .05).
In this study the subgroup of veterans with AO exposure differed from the whole study population. As such, CDKN2A/B mutations were observed with the same frequency as NF1 mutations (46% each); however, BRAF mutations constituted only 31% of the mutations. In addition, the frequency of NF1 mutations was significantly higher in the AO subgroup compared with the general population, but not in the whole study population.
Our sample also differed from the reference population by showing a significantly higher frequency of TMB-high (ie, ≥ 10 mut/Mb) tumors (71% vs 49%; P = .01).12 Interestingly, no significant difference in the frequency of TMB-high tumors was observed between the AO subgroup and the reference population (69% vs 49%; P = .16). There also was no statistically significant difference between the frequency of MSI-H tumors in our study population and the reference population (P = .64).13
One patient in the study population had uveal melanoma. Mutations encountered in this patient’s tumor differed from the general mutational profile of tumors. None of the 21 mutations depicted in Table 2 were present in this sample.10,11 On the other hand, those mutations frequently observed in intraocular melanomas, BAP1 and GNA11, were present in this patient.18 Additionally, this particular melanoma possessed mutations in genes RICTOR, RAD21, and PIK3R1.
Limitations
This study population consisted exclusively of male patients, introducing sex as a potential confounder in analyzing differences between the study population and the general population. As noted in a 2020 systematic review, there were no sex-based differences in the frequency of mutations in BRAF, NRAS, and KIT genes.19
Regarding NF1 mutations, only NF1-mutated acral and mucosal melanomas were more frequently observed in female patients, whereas nonacral NF1-mutated melanomas were more frequently observed in male patients.20 However, there is currently no clear evidence of whether the mutational landscapes of cutaneous melanoma differ by sex.21 Among the 11 cases with NF1-mutatation, site of origin was known in 6, 5 of which originated at nonacral sites. Although the AO subgroup also consisted entirely of male patients, this does not explain the observed increased frequency of NF1 mutations relative to the general population. No such difference was observed between the whole study population, which also consisted exclusively of male patients, and the general population. The similar frequencies of nonacral location in the whole study population (3 acral, 18 nonacral, 14 unknown site of origin) and AO subgroup (1 acral, 7 nonacral, 5 unknown site of origin) preclude location as an explanation.
The Cancer Genome Atlas Network proposed a framework for genomic classification of melanoma into 4 subtypes based on the pattern of the most prevalent significantly mutated genes: mutant BRAF, mutant RAS, mutant NF1, and triple–wild-type. According to that study, BRAF mutations were indeed associated with younger age, in contrast to the NF1-mutant genomic subtype, which was more prevalent in older individuals with higher TMB.22 This emphasizes the need to interpret the potential association of AO exposure and NF1 mutation in melanoma with caution, although additional studies are required to observe the difference between the veteran population and age-matched general population.
On the other hand, Yu et al reported no significant differences of TMB values between patients aged < 60 and ≥ 60 years with melanoma.23 In short, the observed differences we report in our limited study warrant additional investigation with larger sample sizes, sex-matched controlling, and age-matched controlling. The study was limited by its small sample size and the single location.
Conclusion
The genomic profile of melanomas in the veteran population appears to be similar to that of the general population with a few possible differences. Melanomas in the veteran study population showed a higher frequency of CDKN2A/B mutations; lower frequency of ROS1, GRIN2A, KDR, KMT2C (MLL3), KMT2D (MLL2), LRP1B, PTPRT, PTCH1, FAT4, and PREX2 mutations; and higher TMB. In addition, melanomas in the AO subgroup showed higher frequencies of NF1 mutations. The significance of such findings remains to be determined by further investigation.
- Bytnar JA, McGlynn KA, et al. Cancer incidence in the US military: An updated analysis. Cancer. 2024;130(1):96-106. doi:10.1002/cncr.34978
- Singer DS. A new phase of the Cancer Moonshot to end cancer as we know it. Nat Med. 2022;28(7):1345-1347. doi:10.1038/s41591-022-01881-5
- Koczkodaj P, Sulkowska U, Didkowska J, et al. Melanoma mortality trends in 28 European countries: a retrospective analysis for the years 1960-2020. Cancers (Basel). 2023;15(5):1514. Published 2023 Feb 28. doi:10.3390/cancers15051514
- Okobi OE, Abreo E, Sams NP, et al. Trends in melanoma incidence, prevalence, stage at diagnosis, and survival: an analysis of the United States Cancer Statistics (USCS) database. Cureus. 2024;16(10):e70697. doi:10.7759/cureus.70697
- Bartling SJ, Rivard SC, Meyerle JH. Melanoma in an active duty marine. Mil Med. 2017;182:e2034-e2039. doi:10.7205/MILMED-D-17-00127
- American Cancer Society. Cancer facts & figures 2023. American Cancer Society; 2023. Accessed June 20, 2025. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2023/2023-cancer-facts-and-figures.pdf
- Rezaei SJ, Kim J, Onyeka S, et al. Skin cancer and other dermatologic conditions among US veterans. JAMA Dermatol. 2024;160(10):1107-1111. doi:10.1001/jamadermatol.2024.3043
- Chang MS, La J, Trepanowski N, et al. Increased relative proportions of advanced melanoma among veterans: a comparative analysis with the Surveillance, Epidemiology, and End Results registry. J Am Acad Dermatol. 2022;87:72-79. doi:10.1016/j.jaad.2022.02.063
- Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78:1185-1192. doi:10.1016/j.jaad.2017.11.062
- Huang FW, Hodis E, Xu MJ, et al. Highly recurrent TERT promoter mutations in human melanoma. Science. 2013;339:957-959. doi:10.1126/science.1229259
- Tate JG, Bamford S, Jubb HC, et al. COSMIC: the Catalogue of Somatic Mutations in Cancer. Nucleic Acids Res. 2019;47:D941-D947. doi:10.1093/nar/gky1015
- Li M, Gao X, Wang X. Identification of tumor mutation burden-associated molecular and clinical features in cancer by analyzing multi-omics data. Front Immunol. 2023;14:1090838. doi:10.3389/fimmu.2023.1090838
- Bonneville R, Krook MA, Kautto EA, et al. Landscape of microsatellite instability across 39 cancer types. JCO Precis Oncol. 2017;2017:PO.17.00073. doi:10.1200/PO.17.00073
- Lui AJ, Pagadala MS, Zhong AY, et al. Agent Orange exposure and prostate cancer risk in the Million Veteran Program. medRxiv [Preprint]. 2023:2023.06.14.23291413. doi:10.1101/2023.06.14.23291413
- Davis EJ, Johnson DB, Sosman JA, et al. Melanoma: what do all the mutations mean? Cancer. 2018;124:3490-3499. doi:10.1002/cncr.31345
- Guo Y, Chen Y, Zhang L, et al. TERT promoter mutations and telomerase in melanoma. J Oncol. 2022;2022:6300329. doi:10.1155/2022/6300329
- Whiteman DC, Stickley M, Watt P, et al. Anatomic site, sun exposure, and risk of cutaneous melanoma. J Clin Oncol. 2006;24:3172-3177. doi:10.1200/JCO.2006.06.1325
- Decatur CL, Ong E, Garg N, et al. Driver mutations in uveal melanoma: associations with gene expression profile and patient outcomes. JAMA Ophthalmol. 2016;134:728-733. doi:10.1001/jamaophthalmol.2016.0903
- Gutiérrez-Castañeda LD, Nova JA, Tovar-Parra JD. Frequency of mutations in BRAF, NRAS, and KIT in different populations and histological subtypes of melanoma: a systemic review. Melanoma Res. 2020;30:62- 70. doi:10.1097/CMR.0000000000000628
- Thielmann CM, Chorti E, Matull J, et al. NF1-mutated melanomas reveal distinct clinical characteristics depending on tumour origin and respond favourably to immune checkpoint inhibitors. Eur J Cancer. 2021;159:113-124. doi:10.1016/j.ejca.2021.09.035
- D’Ecclesiis O, Caini S, Martinoli C, et al. Gender-dependent specificities in cutaneous melanoma predisposition, risk factors, somatic mutations, prognostic and predictive factors: a systematic review. Int J Environ Res Public Health. 2021;18:7945. doi:10.3390/ijerph18157945
- Cancer Genome Atlas Network. Genomic classification of cutaneous melanoma. Cell. 2015;161:1681-1696. doi:10.1016/j.cell.2015.05.044
- Yu Z, Wang J, Feng L, et al. Association of tumor mutational burden with age in solid tumors. J Clin Oncol. 2020;38:e13590-e13590. doi:10.1200/JCO.2020.38.15_suppl.e13590
- Bytnar JA, McGlynn KA, et al. Cancer incidence in the US military: An updated analysis. Cancer. 2024;130(1):96-106. doi:10.1002/cncr.34978
- Singer DS. A new phase of the Cancer Moonshot to end cancer as we know it. Nat Med. 2022;28(7):1345-1347. doi:10.1038/s41591-022-01881-5
- Koczkodaj P, Sulkowska U, Didkowska J, et al. Melanoma mortality trends in 28 European countries: a retrospective analysis for the years 1960-2020. Cancers (Basel). 2023;15(5):1514. Published 2023 Feb 28. doi:10.3390/cancers15051514
- Okobi OE, Abreo E, Sams NP, et al. Trends in melanoma incidence, prevalence, stage at diagnosis, and survival: an analysis of the United States Cancer Statistics (USCS) database. Cureus. 2024;16(10):e70697. doi:10.7759/cureus.70697
- Bartling SJ, Rivard SC, Meyerle JH. Melanoma in an active duty marine. Mil Med. 2017;182:e2034-e2039. doi:10.7205/MILMED-D-17-00127
- American Cancer Society. Cancer facts & figures 2023. American Cancer Society; 2023. Accessed June 20, 2025. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2023/2023-cancer-facts-and-figures.pdf
- Rezaei SJ, Kim J, Onyeka S, et al. Skin cancer and other dermatologic conditions among US veterans. JAMA Dermatol. 2024;160(10):1107-1111. doi:10.1001/jamadermatol.2024.3043
- Chang MS, La J, Trepanowski N, et al. Increased relative proportions of advanced melanoma among veterans: a comparative analysis with the Surveillance, Epidemiology, and End Results registry. J Am Acad Dermatol. 2022;87:72-79. doi:10.1016/j.jaad.2022.02.063
- Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78:1185-1192. doi:10.1016/j.jaad.2017.11.062
- Huang FW, Hodis E, Xu MJ, et al. Highly recurrent TERT promoter mutations in human melanoma. Science. 2013;339:957-959. doi:10.1126/science.1229259
- Tate JG, Bamford S, Jubb HC, et al. COSMIC: the Catalogue of Somatic Mutations in Cancer. Nucleic Acids Res. 2019;47:D941-D947. doi:10.1093/nar/gky1015
- Li M, Gao X, Wang X. Identification of tumor mutation burden-associated molecular and clinical features in cancer by analyzing multi-omics data. Front Immunol. 2023;14:1090838. doi:10.3389/fimmu.2023.1090838
- Bonneville R, Krook MA, Kautto EA, et al. Landscape of microsatellite instability across 39 cancer types. JCO Precis Oncol. 2017;2017:PO.17.00073. doi:10.1200/PO.17.00073
- Lui AJ, Pagadala MS, Zhong AY, et al. Agent Orange exposure and prostate cancer risk in the Million Veteran Program. medRxiv [Preprint]. 2023:2023.06.14.23291413. doi:10.1101/2023.06.14.23291413
- Davis EJ, Johnson DB, Sosman JA, et al. Melanoma: what do all the mutations mean? Cancer. 2018;124:3490-3499. doi:10.1002/cncr.31345
- Guo Y, Chen Y, Zhang L, et al. TERT promoter mutations and telomerase in melanoma. J Oncol. 2022;2022:6300329. doi:10.1155/2022/6300329
- Whiteman DC, Stickley M, Watt P, et al. Anatomic site, sun exposure, and risk of cutaneous melanoma. J Clin Oncol. 2006;24:3172-3177. doi:10.1200/JCO.2006.06.1325
- Decatur CL, Ong E, Garg N, et al. Driver mutations in uveal melanoma: associations with gene expression profile and patient outcomes. JAMA Ophthalmol. 2016;134:728-733. doi:10.1001/jamaophthalmol.2016.0903
- Gutiérrez-Castañeda LD, Nova JA, Tovar-Parra JD. Frequency of mutations in BRAF, NRAS, and KIT in different populations and histological subtypes of melanoma: a systemic review. Melanoma Res. 2020;30:62- 70. doi:10.1097/CMR.0000000000000628
- Thielmann CM, Chorti E, Matull J, et al. NF1-mutated melanomas reveal distinct clinical characteristics depending on tumour origin and respond favourably to immune checkpoint inhibitors. Eur J Cancer. 2021;159:113-124. doi:10.1016/j.ejca.2021.09.035
- D’Ecclesiis O, Caini S, Martinoli C, et al. Gender-dependent specificities in cutaneous melanoma predisposition, risk factors, somatic mutations, prognostic and predictive factors: a systematic review. Int J Environ Res Public Health. 2021;18:7945. doi:10.3390/ijerph18157945
- Cancer Genome Atlas Network. Genomic classification of cutaneous melanoma. Cell. 2015;161:1681-1696. doi:10.1016/j.cell.2015.05.044
- Yu Z, Wang J, Feng L, et al. Association of tumor mutational burden with age in solid tumors. J Clin Oncol. 2020;38:e13590-e13590. doi:10.1200/JCO.2020.38.15_suppl.e13590
Comprehensive Genomic Profiles of Melanoma in Veterans Compared to Reference Databases
Comprehensive Genomic Profiles of Melanoma in Veterans Compared to Reference Databases
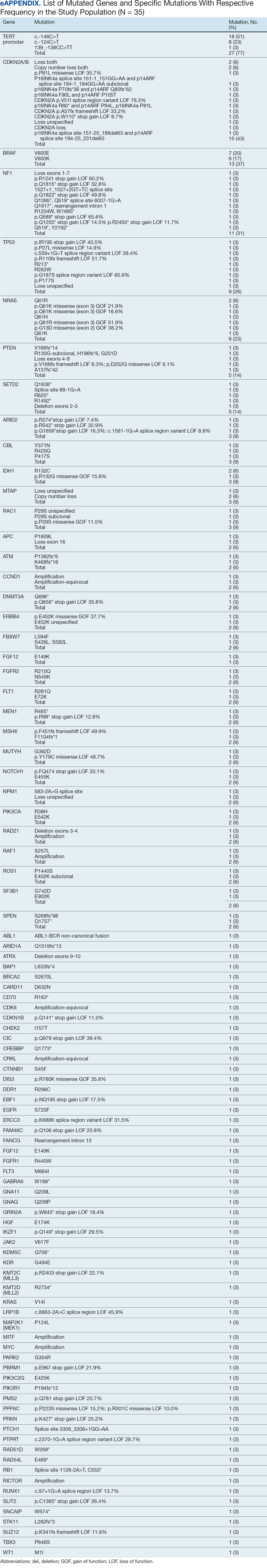
'Distress is the Norm': How Oncologists Can Open the Door to Patient Mental Health
'Distress is the Norm': How Oncologists Can Open the Door to Patient Mental Health
For patients with cancer, the determining factor in whether they pursue mental health services is often whether their oncologist explicitly says it is a good idea, a psychologist said during the July Association of VA Hematology and Oncology (AVAHO) seminar in Long Beach, California, on treating veterans with renal cell carcinoma (RCC).
Kysa Christie, PhD, of the West Los Angeles Veterans Affairs Medical Center, presented findings from a 2018 study in which researchers asked Swiss patients with cancer whether their oncologist discussed their emotional health with them.
In terms of boosting intake, it did not matter if oncologists acknowledged distress or pointed out that psychosocial services existed. Instead, a direct recommendation made a difference, increasing the likelihood of using the services over a 4-month period after initial assessment (odds ratio, 6.27).
“What it took was, ‘I really recommend this. This is something that I would want you to try,’” Christie said.
Oncologists are crucial links between patients and mental health services, Christie said: “If people don’t ask about [distress], you’re not going to see it, but it’s there. Distress is the norm, right? It is not a weakness. It is something that we expect to see.”
Christie noted that an estimated 20% of cancer patients have major depressive disorder, and 35% to 40% have a diagnosable psychiatric condition. RCC shows disproportionately high rates of mental strain. According to Christie, research suggests that about three-fourths of the population report elevated levels of distress as evidenced by patients who scored ≥ 5 on the NCCN Distress Thermometer. Patients with cancer have an estimated 20% higher risk of suicide, especially during the first 12 months after diagnosis and at end of life, she added.
“Early during a diagnosis phase, where you’re having a lot of tests being done, you know something is happening. But you don’t know what,” Christie said. “It could be very serious. That’s just a lot of stress to hold and not know how to plan for.”
After diagnosis, routine could set in and lower distress, she said. Then terminal illness may spike it back up again. Does mental health treatment work in patients with cancer?
“There’s a really strong body of evidence-based treatments for depression, anxiety, adjustment disorders, and coping with different cancers,” Christie said. But it is a step too far to expect patients to ask for help while they are juggling appointments, tests, infusions, and more. “It’s a big ask, right? It’s setting people up for failure.”
To help, Christie said she is embedded with a medical oncology team and routinely talks with the staff about which patients may need help. “One thing I like to do is try to have brief visits with veterans and introduce myself when they come to clinic. I treat it like an opt-out rather than an opt-in program: I’ll just pop into the exam room. They don’t have to ask to see me.”
Christie focuses on open-ended questions and talks about resources ranging from support groups and brief appointments to extensive individual therapy.
Another approach is a strategy known as the “warm handoff,” when an oncologist directly introduces a patient to a mental health professional. “It’s a transfer of care in front of the veteran: It’s much more time-efficient than putting in a referral.”
Christie explained how this can work. A clinician will ask her to meet with a patient during an appointment, perhaps in a couple minutes.
“Then I pop into the room, and the oncologist says, ‘Thanks for joining us. This is Mr. Jones. He has been experiencing feelings of anxiety and sadness, and we’d appreciate your help in exploring some options that might help.’ I turn to the patient and ask, ‘What more would you add?’ Then I either take Mr. Jones back to my office or stay in clinic, and we’re off to the races.”
Christie reported no disclosures.
For patients with cancer, the determining factor in whether they pursue mental health services is often whether their oncologist explicitly says it is a good idea, a psychologist said during the July Association of VA Hematology and Oncology (AVAHO) seminar in Long Beach, California, on treating veterans with renal cell carcinoma (RCC).
Kysa Christie, PhD, of the West Los Angeles Veterans Affairs Medical Center, presented findings from a 2018 study in which researchers asked Swiss patients with cancer whether their oncologist discussed their emotional health with them.
In terms of boosting intake, it did not matter if oncologists acknowledged distress or pointed out that psychosocial services existed. Instead, a direct recommendation made a difference, increasing the likelihood of using the services over a 4-month period after initial assessment (odds ratio, 6.27).
“What it took was, ‘I really recommend this. This is something that I would want you to try,’” Christie said.
Oncologists are crucial links between patients and mental health services, Christie said: “If people don’t ask about [distress], you’re not going to see it, but it’s there. Distress is the norm, right? It is not a weakness. It is something that we expect to see.”
Christie noted that an estimated 20% of cancer patients have major depressive disorder, and 35% to 40% have a diagnosable psychiatric condition. RCC shows disproportionately high rates of mental strain. According to Christie, research suggests that about three-fourths of the population report elevated levels of distress as evidenced by patients who scored ≥ 5 on the NCCN Distress Thermometer. Patients with cancer have an estimated 20% higher risk of suicide, especially during the first 12 months after diagnosis and at end of life, she added.
“Early during a diagnosis phase, where you’re having a lot of tests being done, you know something is happening. But you don’t know what,” Christie said. “It could be very serious. That’s just a lot of stress to hold and not know how to plan for.”
After diagnosis, routine could set in and lower distress, she said. Then terminal illness may spike it back up again. Does mental health treatment work in patients with cancer?
“There’s a really strong body of evidence-based treatments for depression, anxiety, adjustment disorders, and coping with different cancers,” Christie said. But it is a step too far to expect patients to ask for help while they are juggling appointments, tests, infusions, and more. “It’s a big ask, right? It’s setting people up for failure.”
To help, Christie said she is embedded with a medical oncology team and routinely talks with the staff about which patients may need help. “One thing I like to do is try to have brief visits with veterans and introduce myself when they come to clinic. I treat it like an opt-out rather than an opt-in program: I’ll just pop into the exam room. They don’t have to ask to see me.”
Christie focuses on open-ended questions and talks about resources ranging from support groups and brief appointments to extensive individual therapy.
Another approach is a strategy known as the “warm handoff,” when an oncologist directly introduces a patient to a mental health professional. “It’s a transfer of care in front of the veteran: It’s much more time-efficient than putting in a referral.”
Christie explained how this can work. A clinician will ask her to meet with a patient during an appointment, perhaps in a couple minutes.
“Then I pop into the room, and the oncologist says, ‘Thanks for joining us. This is Mr. Jones. He has been experiencing feelings of anxiety and sadness, and we’d appreciate your help in exploring some options that might help.’ I turn to the patient and ask, ‘What more would you add?’ Then I either take Mr. Jones back to my office or stay in clinic, and we’re off to the races.”
Christie reported no disclosures.
For patients with cancer, the determining factor in whether they pursue mental health services is often whether their oncologist explicitly says it is a good idea, a psychologist said during the July Association of VA Hematology and Oncology (AVAHO) seminar in Long Beach, California, on treating veterans with renal cell carcinoma (RCC).
Kysa Christie, PhD, of the West Los Angeles Veterans Affairs Medical Center, presented findings from a 2018 study in which researchers asked Swiss patients with cancer whether their oncologist discussed their emotional health with them.
In terms of boosting intake, it did not matter if oncologists acknowledged distress or pointed out that psychosocial services existed. Instead, a direct recommendation made a difference, increasing the likelihood of using the services over a 4-month period after initial assessment (odds ratio, 6.27).
“What it took was, ‘I really recommend this. This is something that I would want you to try,’” Christie said.
Oncologists are crucial links between patients and mental health services, Christie said: “If people don’t ask about [distress], you’re not going to see it, but it’s there. Distress is the norm, right? It is not a weakness. It is something that we expect to see.”
Christie noted that an estimated 20% of cancer patients have major depressive disorder, and 35% to 40% have a diagnosable psychiatric condition. RCC shows disproportionately high rates of mental strain. According to Christie, research suggests that about three-fourths of the population report elevated levels of distress as evidenced by patients who scored ≥ 5 on the NCCN Distress Thermometer. Patients with cancer have an estimated 20% higher risk of suicide, especially during the first 12 months after diagnosis and at end of life, she added.
“Early during a diagnosis phase, where you’re having a lot of tests being done, you know something is happening. But you don’t know what,” Christie said. “It could be very serious. That’s just a lot of stress to hold and not know how to plan for.”
After diagnosis, routine could set in and lower distress, she said. Then terminal illness may spike it back up again. Does mental health treatment work in patients with cancer?
“There’s a really strong body of evidence-based treatments for depression, anxiety, adjustment disorders, and coping with different cancers,” Christie said. But it is a step too far to expect patients to ask for help while they are juggling appointments, tests, infusions, and more. “It’s a big ask, right? It’s setting people up for failure.”
To help, Christie said she is embedded with a medical oncology team and routinely talks with the staff about which patients may need help. “One thing I like to do is try to have brief visits with veterans and introduce myself when they come to clinic. I treat it like an opt-out rather than an opt-in program: I’ll just pop into the exam room. They don’t have to ask to see me.”
Christie focuses on open-ended questions and talks about resources ranging from support groups and brief appointments to extensive individual therapy.
Another approach is a strategy known as the “warm handoff,” when an oncologist directly introduces a patient to a mental health professional. “It’s a transfer of care in front of the veteran: It’s much more time-efficient than putting in a referral.”
Christie explained how this can work. A clinician will ask her to meet with a patient during an appointment, perhaps in a couple minutes.
“Then I pop into the room, and the oncologist says, ‘Thanks for joining us. This is Mr. Jones. He has been experiencing feelings of anxiety and sadness, and we’d appreciate your help in exploring some options that might help.’ I turn to the patient and ask, ‘What more would you add?’ Then I either take Mr. Jones back to my office or stay in clinic, and we’re off to the races.”
Christie reported no disclosures.
'Distress is the Norm': How Oncologists Can Open the Door to Patient Mental Health
'Distress is the Norm': How Oncologists Can Open the Door to Patient Mental Health