User login
Migraine headache: When to consider these newer agents
Migraine is a headache disorder that often causes unilateral pain, photophobia, phonophobia, nausea, and vomiting. More than 70% of office visits for migraine are made to primary care physicians.1 Recent data suggest migraine may be caused primarily by neuronal dysfunction and only secondarily by vasodilation.2 Although there are numerous classes of drugs used for migraine prevention and treatment, their success has been limited by inadequate efficacy, tolerability, and patient adherence.3 The discovery of pro-inflammatory markers such as calcitonin gene-related peptide (CGRP) has led to the development of new medications to prevent and treat migraine.4

Pathophysiology, Dx and triggers, indications for pharmacotherapy
Pathophysiology. A migraine is thought to be caused by cortical spreading depression (CSD), a depolarization of glial and neuronal cell membranes.5
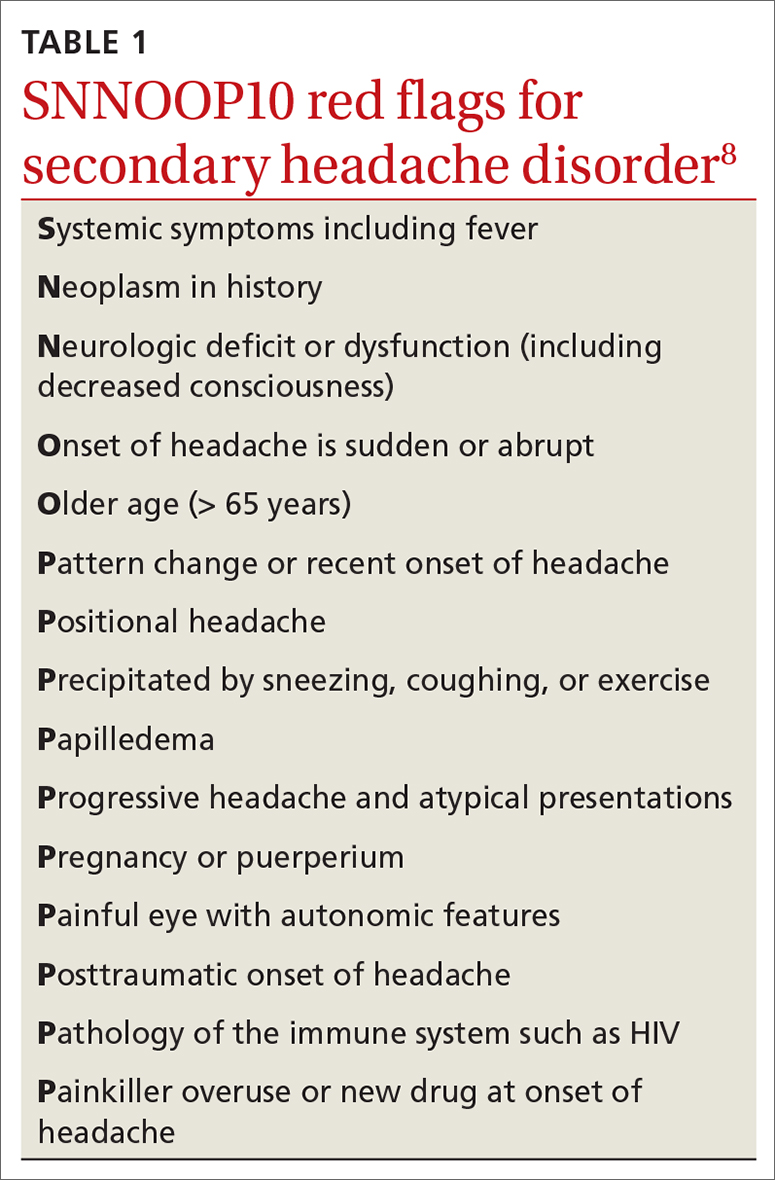
Dx and triggers. In 2018, the International Headache Society revised its guidelines for the diagnosis of migraine.7 According to the 3rd edition of The International Classification of Headache Disorders (ICHD-3), the diagnosis of migraine is made when a patient has at least 5 headache attacks that last 4 to 72 hours and have at least 2 of the following characteristics: (1) unilateral location, (2) pulsating quality, (3) moderate-to-severe pain intensity, and (4) aggravated by or causing avoidance of routine physical activity.7 The headache attacks also should have (1) associated nausea or vomiting or (2) photophobia and phonophobia.7 The presence of atypical signs or symptoms as indicated by the SNNOOP10 mnemonic raises concerns for secondary headaches and the need for further investigation into the cause of the headache (TABLE 1).8 It is not possible to detect every secondary headache with standard neuroimaging, but the SNNOOP10 red flags can help determine when imaging may be indicated.8 Potential triggers for migraine can be found in TABLE 2.9
Indications for pharmacotherapy. All patients receiving a diagnosis of migraine should be offered acute pharmacologic treatment. Consider preventive therapy anytime there are ≥ 4 headache days per month, debilitating attacks despite acute therapy, overuse of acute medication (> 2 d/wk), difficulty tolerating acute medication, patient preference, or presence of certain migraine subtypes.7,10

Acute treatments
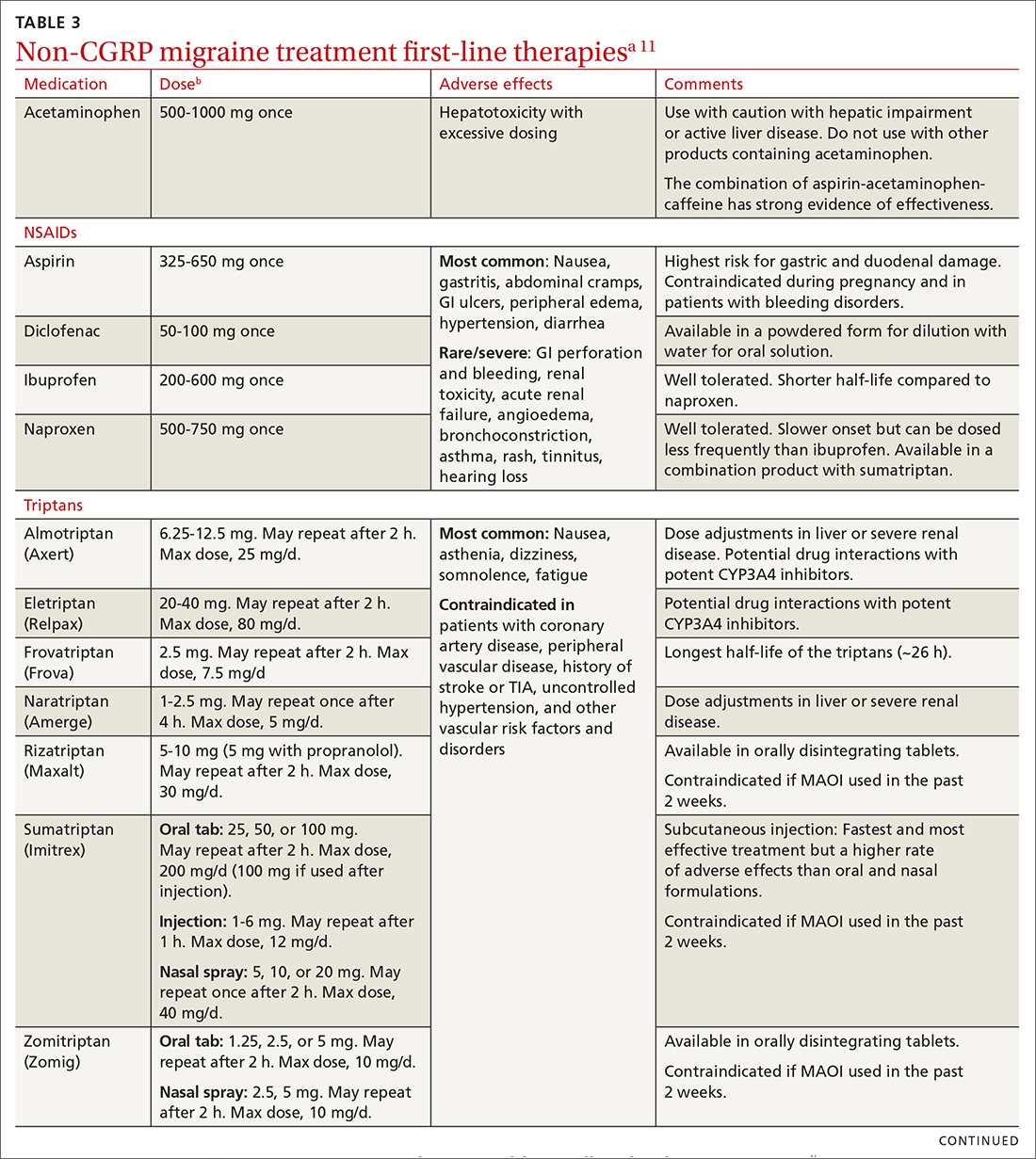
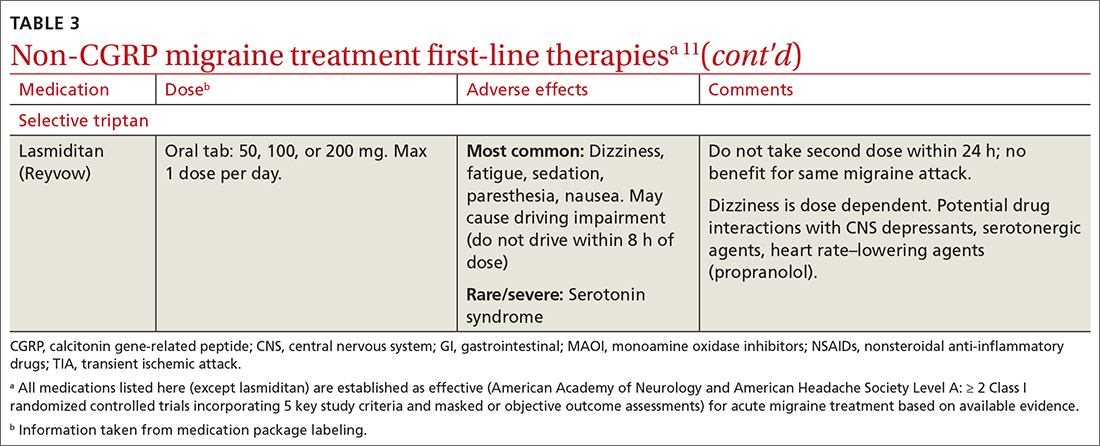
Abortive therapies for migraine include analgesics such as nonsteroidal anti-inflammatory drugs (NSAIDs) and acetaminophen, and ergot alkaloids, triptans, or small-molecule CGRP receptor antagonists (gepants). Prompt administration increases the chance of success with acute therapy. Medications with the highest levels of efficacy based on the 2015 guidelines from the American Headache Society (AHS) are given in TABLE 3.11 Lasmiditan (Reyvow) is not included in the 2015 guidelines, as it was approved after publication of the guidelines.
Non-CGRP first-line therapies
NSAIDs and acetaminophen. NSAIDs such as aspirin, diclofenac, ibuprofen, and naproxen have a high level of evidence to support their use as first-line treatments for mild-to-moderate migraine attacks. Trials consistently demonstrate their superiority to placebo in headache relief and complete pain relief at 2 hours. There is no recommendation for selecting one NSAID over another; however, consider their frequency of dosing and adverse effect profiles. The number needed to treat for complete pain relief at 2 hours ranges from 7 to 10 for most NSAIDs.11,12 In some placebo-controlled studies, acetaminophen was less effective than NSAIDs, but was safer because it did not cause gastric irritation or antiplatelet effects.12
Triptans inhibit 5-HT1B/1D receptors. Consider formulation, route of administration, cost, and pharmacokinetics when selecting a triptan. Patients who do not respond well to one triptan may respond favorably to another. A meta-analysis of the effectiveness of the 7 available agents found that triptans at standard doses provided pain relief within 2 hours in 42% to 76% of patients, and sustained freedom from pain for 2 hours in 18% to 50% of patients.13 Lasmiditan is a selective serotonin receptor (5-HT1F) agonist that lacks vasoconstrictor activity. This is an option for patients with relative contraindications to triptans due to cardiovascular risk factors.10
Continue to: Second-line therapies
Second-line therapies
Intranasal dihydroergotamine has a favorable adverse event profile and greater evidence for efficacy compared with ergotamine. Compared with triptans, intranasal dihydroergotamine has a high level of efficacy but causes more adverse effects.14 Severe nausea is common, and dihydroergotamine often is used in combination with an antiemetic drug. Dihydroergotamine should not be used within 24 hours of taking a triptan, and it is contraindicated for patients who have hypertension or ischemic heart disease or who are pregnant or breastfeeding. There is also the potential for adverse drug interactions.15
Antiemetics may be helpful for migraine associated with severe nausea or vomiting. The dopamine antagonists metoclopramide, prochlorperazine, and chlorpromazine have demonstrated benefit in randomized placebo-controlled trials.11 Ondansetron has not been studied extensively, but sometimes is used in clinical practice. Nonoral routes of administration may be useful in patients having trouble swallowing medications or in those experiencing significant nausea or vomiting early during migraine attacks.
Due to the high potential for abuse, opioids should not be used routinely for the treatment of migraine.12 There is no high-quality evidence supporting the efficacy of barbiturates (ie, butalbital-containing compounds) for acute migraine treatment.11 Moreover, use of these agents may increase the likelihood of progression from episodic to chronic migraine.16
Gepants for acute migraine treatment
Neuropeptide CGRP is released from trigeminal nerves and is a potent dilator of cerebral and dural vessels, playing a key role in regulating blood flow to the brain. Other roles of CGRP include the release of inflammatory agents from mast cells and the transmission of painful stimuli from intracranial vessels.17 The CGRP receptor or ligand can be targeted by small-molecule receptor antagonists for acute and preventive migraine treatment (and by monoclonal antibodies solely for prevention, discussed later). It has been theorized that gepants bind to CGRP receptors, resulting in decreased blood flow to the brain, inhibition of neurogenic inflammation, and reduced pain signaling.17 Unlike triptans and ergotamine derivatives, these novel treatments do not constrict blood vessels and may have a unique role in patients with contraindications to triptans.
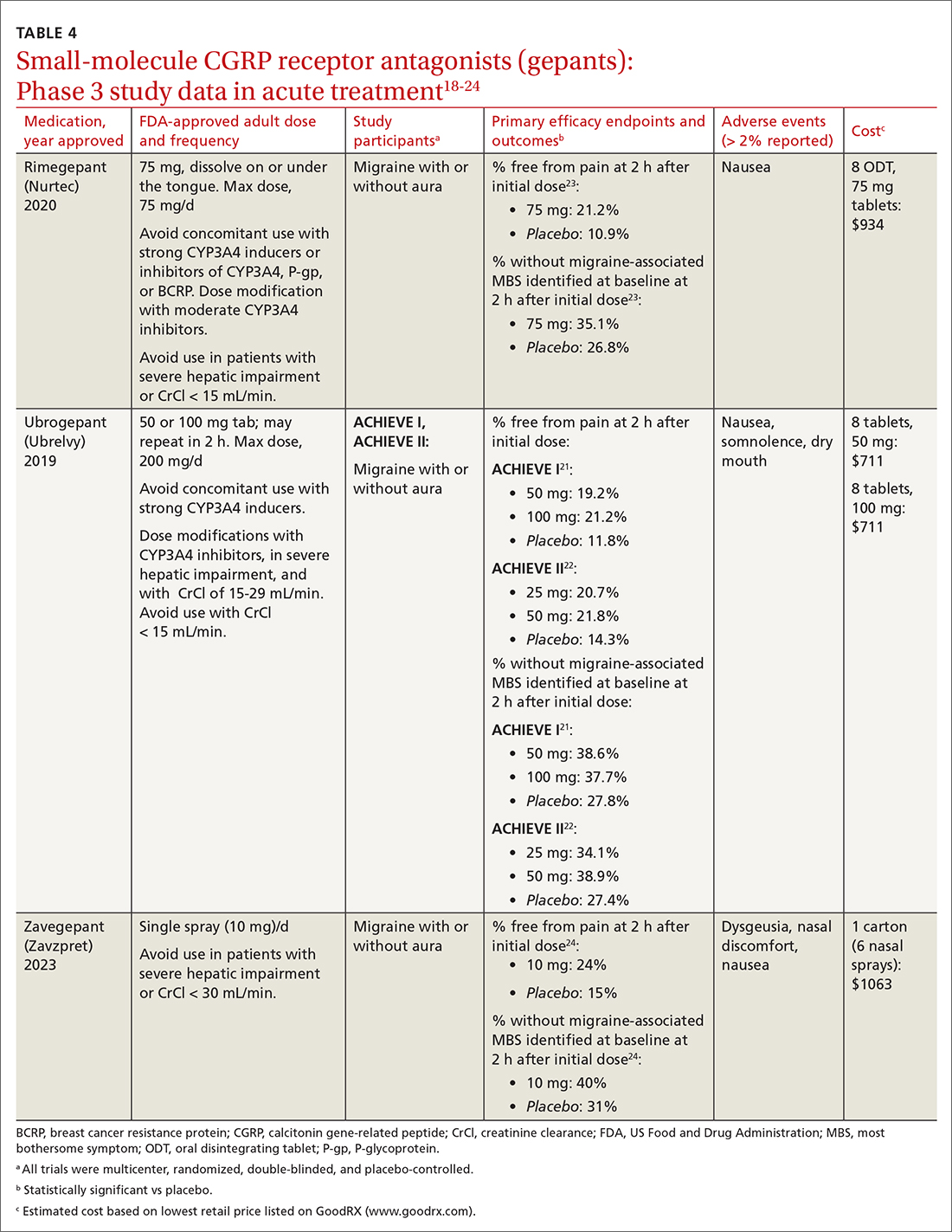
The 3 gepants approved for acute treatment—ubrogepant (Ubrelvy),18 rimegepant (Nurtec),19 and zavegepant (Zavzpret)20—were compared with placebo in clinical trials and were shown to increase the number of patients who were completely pain free at 2 hours, were free of the most bothersome associated symptom (photophobia, phonophobia, or nausea) at 2 hours, and remained pain free at 24 hours (TABLE 418-24).
Continue to: Ubrogrepant
Ubrogepant, in 2 Phase 3 trials (ACHIEVE I and ACHIEVE II) demonstrated effectiveness compared with placebo.21,22 The most common adverse effects reported were nausea and somnolence at very low rates. Pain-relief rates at 2 hours post dose (> 60% of participants) were higher than pain-free rates, and a significantly higher percentage (> 40%) of ubrogepant-treated participants reported ability to function normally on the Functional Disability Scale.25
Rimegepant was also superior to placebo (59% vs 43%) in pain relief at 2 hours post dose and other secondary endpoints.23 Rimegepant also has potential drug interactions
Zavegepant, approved in March 2023, is administered once daily as a 10-mg nasal spray. In its Phase 3 trial, zavegepant was significantly superior to placebo at 2 hours post dose in freedom from pain (24% v 15%), and in freedom from the most bothersome symptom (40% v 31%).24 Dosage modifications are not needed with mild-to-moderate renal or hepatic disease.20
Worth noting. The safety of using ubrogepant to treat more than 8 migraine episodes in a 30-day period has not been established. The safety of using more than 18 doses of zavegepant in a 30-day period also has not been established. With ubrogepant and rimegepant, there are dosing modifications for concomitant use with specific drugs (CYP3A4 inhibitors and inducers) due to potential interactions and in patients with hepatic or renal impairment.18,19
There are no trials comparing efficacy of CGRP antagonists to triptans. Recognizing that these newer medications would be costly, the AHS position statement released in 2019 recommends that gepants be considered for those with contraindications to triptans or for whom at least 2 oral triptans have failed (as determined by a validated patient outcome questionnaire).10 Step therapy with documentation of previous trials and therapy failures is often required by insurance companies prior to gepant coverage.
Continue to: Preventive therapies
Preventive therapies
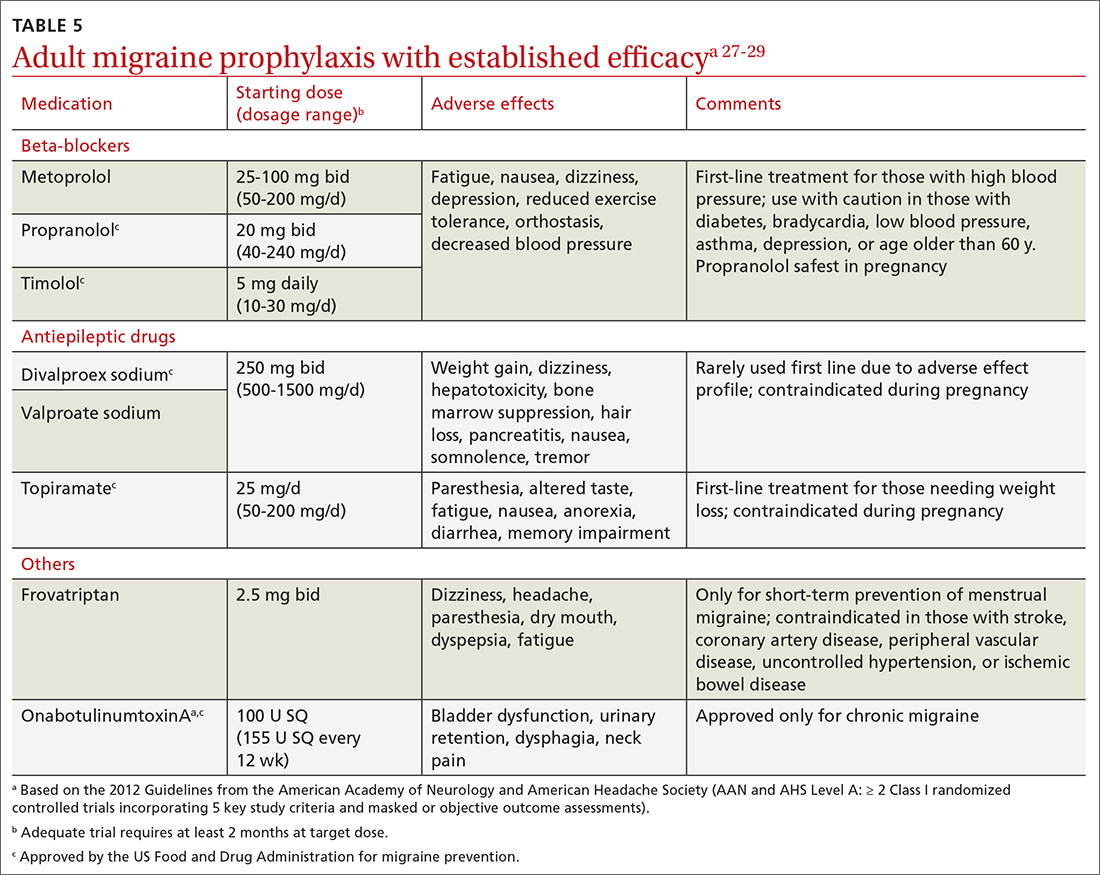
Preventive migraine therapies are used to reduce duration, frequency, and severity of attacks, the need for acute treatment, and overall headache disability.26 Medications typically are chosen based on efficacy, adverse effect profile, and patient comorbidities. Barriers to successful use include poor patient adherence and tolerability, the need for slow dose titration, and long-term use (minimum of 2 months) at maximum tolerated or minimum effective doses. Medications with established efficacy (Level Aa) based on the 2012 guidelines from the American Academy of Neurology (AAN) and the AHS are given in TABLE 5.27-29
Drugs having received the strongest level of evidence for migraine prevention are metoprolol, propranolol, timolol, topiramate, valproate sodium, divalproex sodium, and onabotulinumtoxinA (Botox), and frovatriptan for menstrual migraine prevention. Because these guidelines were last updated in 2012, they did not cover gepants (which will be discussed shortly). The AHS released a position statement in 2019 supporting the use of
CGRP-targeted prevention
Four anti-CGRP mAbs and 2 gepants have been approved for migraine prevention in the United States. Differences between products include targets (ligand vs receptor), antibody IgG subtype, bioavailability, route of administration, and frequency of administration.28 As noted in the Phase 3 studies (TABLE 619,30-47), these therapies are highly efficacious, safe, and tolerable.
Gepants. Rimegepant, discussed earlier for migraine treatment, is one of the CGRP receptor antagonists approved for prevention. The other is atogepant (Qulipta), approved only for prevention. Ubrogepant is not approved for prevention.
Anti-CGRP mAb is the only medication class specifically created for migraine prevention.10,26 As already noted, several efficacious non-CGRP treatment options are available for migraine prevention. However, higher doses of those agents, if needed,
Continue to: The targeted anti-CGRP approach...
The targeted anti-CGRP approach, which can be used by patients with liver or kidney disease, results in decreased toxicity and minimal drug interactions. Long half-lives allow for monthly or quarterly injections, possibly resulting in increased compliance.28 Dose titration is not needed, allowing for more rapid symptom management. The large molecular size of a mAb limits its transfer across the blood-brain barrier, making central nervous system adverse effects unlikely.28 Despite the compelling mAb pharmacologic properties, their use may be limited by a lack of long-term safety data and the need for parenteral administration. Although immunogenicity—the development of neutralizing antibodies—can limit long-term tolerability or efficacy of mAbs generally,26,28 anti-CGRP mAbs were engineered to minimally activate the immune system and have not been associated with immune suppression, opportunistic infections, malignancies, or decreased efficacy.28
A pooled meta-analysis including 4 trials (3166 patients) found that CGRP mAbs compared with placebo significantly improved patient response rates, defined as at least a 50% and 75% reduction in monthly headache/migraine days from baseline to Weeks 9 to 12.48 Another meta-analysis including 8 trials (2292 patients) found a significant reduction from baseline in monthly migraine days and monthly acute migraine medication consumption among patients taking CGRP mAbs compared with those taking placebo.49 Open-label extension studies have shown progressive and cumulative benefits in individuals who respond to anti-CGRP mAbs. Therefore, several treatment cycles may be necessary to determine overall efficacy of therapy.10,28
Cost initially can be a barrier. Insurance companies often require step therapy before agreeing to cover mAb therapy, which aligns with the 2019 AHS position statement.10
When combination treatment may be appropriate
Monotherapy is the usual approach to preventing migraine due to advantages of efficacy, simplified regimens, lower cost, and reduced adverse effects.51 However, if a patient does not benefit from monotherapy even after trying dose titrations as tolerated or switching therapies, trying complementary combination therapy is appropriate. Despite a shortage of clinical trials supporting the use of 2 or more preventive medications with different mechanisms of action, this strategy is used clinically.10 Consider combination therapy in those with refractory disease, partial responses, or intolerance to recommended doses.52 Articles reporting on case study reviews have rationalized the combined use of onabotulinumtoxinA and anti-CGRP mAbs, noting better migraine control.51,53 The 2019 AHS position statement recommends adding a mAb to an existing preventive treatment regimen with no other changes until mAb effectiveness is determined, as the risk for drug interactions on dual therapy is low.10 Safety and efficacy also have been demonstrated with the combination of preventive anti-CGRP mAbs and acute treatment with gepants as needed.54
CORRESPONDENCE
Emily Peterson, PharmD, BCACP, 3640 Middlebury Road, Iowa City, IA 52242; [email protected]
1. Lipton RB, Nicholson RA, Reed ML, et al. Diagnosis, consultation, treatment, and impact of migraine in the US: results of the OVERCOME (US) study. Headache. 2022;62:122-140. doi: 10.1111/head.14259
2. Burstein R, Noseda R, Borsook D. Migraine: multiple processes; complext pathophysiology. J Neurosci. 2015;35:6619-6629. doi: 10.1523/JNEUROSCI.0373-15.2015
3. Edvinsson L, Haanes KA, Warfvinge K, et al. CGRP as the target of new migraine therapies - successful translation from bench to clinic. Nat Rev Neurol. 2018;14:338-350. doi: 10.1038/s41582-018-0003-1
4. McGrath K, Rague A, Thesing C, et al. Migraine: expanding our Tx arsenal. J Fam Pract. 2019;68:10-14;16-24.
5. Dodick DW. Migraine. Lancet. 2018;391:1315-1330. doi: 10.1016/S0140-6736(18)30478-1
6. Agostoni EC, Barbanti P, Calabresi P, et al. Current and emerging evidence-based treatment options in chronic migraine: a narrative review. J Headache Pain. 2019;20:92. doi: 10.1186/s10194-019-1038-4
7. IHS. Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3rd edition. Cephalalgia. 2018;38:1-211. doi: 10.1177/0333102417738202
8. Do TP, Remmers A, Schytz HW, et al. Red and orange flags for secondary headaches in clinical practice: SNNOOP10 list. Neurology. 2019;92:134-144. doi: 10.1212/WNL.0000000000006697
9. NIH. Migraine. Accessed July 30, 2023.
10. AHS. The American Headache Society position statement on integrating new migraine treatments into clinical practice. Headache. 2019;59:1-18. doi: 10.1111/head.13456
11. Marmura MJ, Silberstein SD, Schwedt TJ. The acute treatment of migraine in adults: the American Headache Society evidence assessment of migraine pharmacotherapies. Headache. 2015;55:3-20. doi: 10.1111/head.12499
12. Mayans L, Walling A. Acute migraine headache: treatment strategies. Am Fam Physician. 2018;97:243-251.
13. Cameron C, Kelly S, Hsieh SC, et al. Triptans in the acute treatment of migraine: a systematic review and network meta-analysis. Headache. 2015;55(suppl 4):221-235. doi: 10.1111/head.12601
14. Becker WJ. Acute migraine treatment. Continuum (Minneap Minn). 2015;21:953-972. doi: 10.1212/CON.0000000000000192
15. Migranal (dihydroergotamine mesylate) Package insert. Valeant Pharmaceuticals North America; 2019. Accessed June 17, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2019/020148Orig1s025lbl.pdf
16. Minen MT, Tanev K, Friedman BW. Evaluation and treatment of migraine in the emergency department: a review. Headache. 2014;54:1131-45. doi: 10.1111/head.12399
17. Durham PL. CGRP-receptor antagonists--a fresh approach to migraine therapy? N Engl J Med. 2004;350:1073-1075. doi: 10.1056/NEJMp048016
18. Ubrelvy (ubrogepant). Package insert. Allergan, Inc.; 2019. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2019/211765s000lbl.pdf
19. Nurtec ODT (rimegepant sulfate). Package insert. Biohaven Pharmaceuticals, Inc.; 2021. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2021/212728s006lbl.pdf
20. Zavzpret (zavegepant). Package insert. Pfizer Labs.; 2023. Accessed July 15, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2023/216386s000lbl.pdf
21. Dodick DW, Lipton RB, Ailani J, et al. Ubrogepant for the treatment of migraine. N Engl J Med. 2019;381:2230-2241. doi: 10.1056/NEJMoa1813049
22. Lipton RB, Dodick DW, Ailani J, et al. Effect of ubrogepant vs placebo on pain and the most bothersome associated symptom in the acute treatment of migraine: the ACHIEVE II randomized clinical trial. JAMA. 2019;322:1887-1898. doi: 10.1001/jama.2019.16711
23. Croop R, Goadsby PJ, Stock DA, et al. Efficacy, safety, and tolerability of rimegepant orally disintegrating tablet for the acute treatment of migraine: a randomised, phase 3, double-blind, placebo-controlled trial. Lancet. 2019;394:737-745. doi: 10.1016/S0140-6736(19)31606-X
24. Lipton RB, Croop R, Stock DA, et al. Safety, tolerability, and efficacy of zavegepant 10 mg nasal spray for the acute treatment of migraine in the USA: a phase 3, double-blind, randomised, placebo-controlled multicentre trial. Lancet Neurol. 2023;22:209-217. doi: 10.1016/S1474-4422(22)00517-8
25. Dodick DW, Lipton RB, Ailani J, et al. Ubrogepant, an acute treatment for migraine, improved patient-reported functional disability and satisfaction in 2 single-attack phase 3 randomized trials, ACHIEVE I and II. Headache. 2020;60:686-700. doi: 10.1111/head.13766
26. Burch R. Migraine and tension-type headache: diagnosis and treatment. Med Clin North Am. 2019;103:215-233. doi:10.1016/j.mcna.2018.10.003
27. Silberstein SD, Holland S, Freitag F, et al. Evidence-based guideline update: pharmacologic treatment for episodic migraine prevention in adults: report of the Quality Standards Subcommittee of the American Academy of Neurology and the American Headache Society. Neurology. 2012;78:1337-1345. doi: 10.1212/WNL.0b013e3182535d20
28. Dodick DW. CGRP ligand and receptor monoclonal antibodies for migraine prevention: evidence review and clinical implications. Cephalalgia. 2019;39:445-458. doi: 10.1177/ 0333102418821662
29. Pringsheim T, Davenport WJ, Becker WJ. Prophylaxis of migraine headache. CMAJ. 2010;182:E269-276. doi: 10.1503/cmaj.081657
30. Vyepti (eptinezumab-jjmr). Package insert. Lundbeck Pharmaceuticals LLV; 2020. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2020/761119s000lbl.pdf
31. Aimovig (erenumab-aooe). Package insert. Amgen Inc.; 2021. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2021/761077s009lbl.pdf
32. Ajovy (fremanezumab-vfrm). Package insert. Teva Pharmaceuticals USA, Inc.; 2018. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2018/761089s000lbl.pdf
33. Emgality (galcanezumab-gnlm). Package insert. Eli Lilly and Company; 2018. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2018/761063s000lbl.pdf
34. Ashina M, Saper J, Cady R, et al. Eptinezumab in episodic migraine: a randomized, double-blind, placebo-controlled study (PROMISE-1). Cephalalgia. 2020;40:241-254. doi: 10.1177/0333102420905132
35. Lipton RB, Goadsby PJ, Smith J, et al. Efficacy and safety of eptinezumab in patients with chronic migraine: PROMISE-2. Neurology. 2020;94:e1365-e1377. doi: 10.1212/WNL.0000000000009169
36. Dodick DW, Ashina M, Brandes JL, et al. ARISE: a phase 3 randomized trial of erenumab for episodic migraine. Cephalalgia. 2018;38:1026-1037. doi: 10.1177/0333102418759786
37. Goadsby PJ, Reuter U, Hallström Y, et al. A controlled trial of erenumab for episodic migraine. N Engl J Med. 2017;377:2123-2132. doi: 10.1056/NEJMoa1705848
38. Reuter U, Goadsby PJ, Lanteri-Minet M, et al. Efficacy and tolerability of erenumab in patients with episodic migraine in whom two-to-four previous preventive treatments were unsuccessful: a randomised, double-blind, placebo-controlled, phase 3b study. Lancet. 2018;392:2280-2287. doi: 10.1016/S0140-6736(18)32534-0
39. Silberstein SD, Dodick DW, Bigal ME, et al. Fremanezumab for the preventive treatment of chronic migraine. N Engl J Med. 2017; 377:2113-2122. doi: 10.1056/NEJMoa1709038
40. Dodick DW, Silberstein SD, Bigal ME, et al. Effect of fremanezumab compared with placebo for prevention of episodic migraine: a randomized clinical trial. JAMA. 2018;319:1999-2008. doi: 10.1001/jama.2018.4853
41. Stauffer VL, Dodick DW, Zhang Q, et al. Evaluation of galcanezumab for the prevention of episodic migraine: the EVOLVE-1 randomized clinical trial. JAMA Neurol. 2018;75:1080-1088. doi: 10.1001/jamaneurol.2018.1212
42. Skljarevski V, Matharu M, Millen BA, et al. Efficacy and safety of galcanezumab for the prevention of episodic migraine: results of the EVOLVE-2 phase 3 randomized controlled clinical trial. Cephalalgia. 2018;38:1442-1454. doi: 10.1177/0333102418779543
43. Detke HC, Goadsby PJ, Wang S, et al. Galcanezumab in chronic migraine: the randomized, double-blind, placebo-controlled REGAIN study. Neurology. 2018;91:e2211-e2221. doi: 10.1212/WNL.0000000000006640
44. Goadsby PJ, Dodick DW, Leone M, at al. Trial of galcanezumab in prevention of episodic cluster headache. N Engl J Med. 2019; 381:132-141. doi: 10.1056/NEJMoa1813440
45. Croop R, Lipton RB, Kudrow D, et al. Oral rimegepant for preventive treatment of migraine: a phase 2/3, randomised, double-blind, placebo-controlled trial. Lancet. 2021;397:51-60. doi: 10.1016/S0140-6736(20)32544-7
46. Ailani J, Lipton RB, Goadsby PJ, et al. Atogepant for the preventive treatment of migraine. N Engl J Med. 2021;385:695-706. doi: 10.1056/NEJMoa2035908
47. Qulipta (atogepant). Package insert. AbbVie; 2021. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2021/215206Orig1s000lbl.pdf
48. Han L, Liu Y, Xiong H, et al. CGRP monoclonal antibody for preventive treatment of chronic migraine: an update of meta-analysis. Brain Behav. 2019;9:e01215. doi: 10.1002/brb3.1215
49. Zhu Y, Liu Y, Zhao J, et al. The efficacy and safety of calcitonin gene-related peptide monoclonal antibody for episodic migraine: a meta-analysis. Neurol Sci. 2018;39:2097-2106. doi: 10.1007/s10072-018-3547-3
50. Szperka CL, VanderPluym J, Orr SL, et al. Recommendations on the use of anti-CGRP monoclonal antibodies in children and adolescents. Headache. 2018;58:1658-1669. doi: 10.1111/head.13414
51. Pellesi L, Do TP, Ashina H, et al. Dual therapy with anti-CGRP monoclonal antibodies and botulinum toxin for migraine prevention: is there a rationale? Headache. 2020;60:1056-1065. doi: 10.1111/head.13843
52. D’Antona L, Matharu M. Identifying and managing refractory migraine: barriers and opportunities? J Headache Pain. 2019;20:89. doi: 10.1186/s10194-019-1040-x
53. Cohen F, Armand C, Lipton RB, et al. Efficacy and tolerability of calcitonin gene-related peptide targeted monoclonal antibody medications as add-on therapy to onabotulinumtoxinA in patients with chronic migraine. Pain Med. 2021;1857-1863. doi: 10.1093/pm/pnab093
54. Berman G, Croop R, Kudrow D, et al. Safety of rimegepant, an oral CGRP receptor antagonist, plus CGRP monoclonal antibodies for migraine. Headache. 2020;60:1734-1742. doi: 10.1111/head.13930
Migraine is a headache disorder that often causes unilateral pain, photophobia, phonophobia, nausea, and vomiting. More than 70% of office visits for migraine are made to primary care physicians.1 Recent data suggest migraine may be caused primarily by neuronal dysfunction and only secondarily by vasodilation.2 Although there are numerous classes of drugs used for migraine prevention and treatment, their success has been limited by inadequate efficacy, tolerability, and patient adherence.3 The discovery of pro-inflammatory markers such as calcitonin gene-related peptide (CGRP) has led to the development of new medications to prevent and treat migraine.4

Pathophysiology, Dx and triggers, indications for pharmacotherapy
Pathophysiology. A migraine is thought to be caused by cortical spreading depression (CSD), a depolarization of glial and neuronal cell membranes.5
Dx and triggers. In 2018, the International Headache Society revised its guidelines for the diagnosis of migraine.7 According to the 3rd edition of The International Classification of Headache Disorders (ICHD-3), the diagnosis of migraine is made when a patient has at least 5 headache attacks that last 4 to 72 hours and have at least 2 of the following characteristics: (1) unilateral location, (2) pulsating quality, (3) moderate-to-severe pain intensity, and (4) aggravated by or causing avoidance of routine physical activity.7 The headache attacks also should have (1) associated nausea or vomiting or (2) photophobia and phonophobia.7 The presence of atypical signs or symptoms as indicated by the SNNOOP10 mnemonic raises concerns for secondary headaches and the need for further investigation into the cause of the headache (TABLE 1).8 It is not possible to detect every secondary headache with standard neuroimaging, but the SNNOOP10 red flags can help determine when imaging may be indicated.8 Potential triggers for migraine can be found in TABLE 2.9

Indications for pharmacotherapy. All patients receiving a diagnosis of migraine should be offered acute pharmacologic treatment. Consider preventive therapy anytime there are ≥ 4 headache days per month, debilitating attacks despite acute therapy, overuse of acute medication (> 2 d/wk), difficulty tolerating acute medication, patient preference, or presence of certain migraine subtypes.7,10

Acute treatments
Abortive therapies for migraine include analgesics such as nonsteroidal anti-inflammatory drugs (NSAIDs) and acetaminophen, and ergot alkaloids, triptans, or small-molecule CGRP receptor antagonists (gepants). Prompt administration increases the chance of success with acute therapy. Medications with the highest levels of efficacy based on the 2015 guidelines from the American Headache Society (AHS) are given in TABLE 3.11 Lasmiditan (Reyvow) is not included in the 2015 guidelines, as it was approved after publication of the guidelines.


Non-CGRP first-line therapies
NSAIDs and acetaminophen. NSAIDs such as aspirin, diclofenac, ibuprofen, and naproxen have a high level of evidence to support their use as first-line treatments for mild-to-moderate migraine attacks. Trials consistently demonstrate their superiority to placebo in headache relief and complete pain relief at 2 hours. There is no recommendation for selecting one NSAID over another; however, consider their frequency of dosing and adverse effect profiles. The number needed to treat for complete pain relief at 2 hours ranges from 7 to 10 for most NSAIDs.11,12 In some placebo-controlled studies, acetaminophen was less effective than NSAIDs, but was safer because it did not cause gastric irritation or antiplatelet effects.12
Triptans inhibit 5-HT1B/1D receptors. Consider formulation, route of administration, cost, and pharmacokinetics when selecting a triptan. Patients who do not respond well to one triptan may respond favorably to another. A meta-analysis of the effectiveness of the 7 available agents found that triptans at standard doses provided pain relief within 2 hours in 42% to 76% of patients, and sustained freedom from pain for 2 hours in 18% to 50% of patients.13 Lasmiditan is a selective serotonin receptor (5-HT1F) agonist that lacks vasoconstrictor activity. This is an option for patients with relative contraindications to triptans due to cardiovascular risk factors.10
Continue to: Second-line therapies
Second-line therapies
Intranasal dihydroergotamine has a favorable adverse event profile and greater evidence for efficacy compared with ergotamine. Compared with triptans, intranasal dihydroergotamine has a high level of efficacy but causes more adverse effects.14 Severe nausea is common, and dihydroergotamine often is used in combination with an antiemetic drug. Dihydroergotamine should not be used within 24 hours of taking a triptan, and it is contraindicated for patients who have hypertension or ischemic heart disease or who are pregnant or breastfeeding. There is also the potential for adverse drug interactions.15
Antiemetics may be helpful for migraine associated with severe nausea or vomiting. The dopamine antagonists metoclopramide, prochlorperazine, and chlorpromazine have demonstrated benefit in randomized placebo-controlled trials.11 Ondansetron has not been studied extensively, but sometimes is used in clinical practice. Nonoral routes of administration may be useful in patients having trouble swallowing medications or in those experiencing significant nausea or vomiting early during migraine attacks.
Due to the high potential for abuse, opioids should not be used routinely for the treatment of migraine.12 There is no high-quality evidence supporting the efficacy of barbiturates (ie, butalbital-containing compounds) for acute migraine treatment.11 Moreover, use of these agents may increase the likelihood of progression from episodic to chronic migraine.16
Gepants for acute migraine treatment
Neuropeptide CGRP is released from trigeminal nerves and is a potent dilator of cerebral and dural vessels, playing a key role in regulating blood flow to the brain. Other roles of CGRP include the release of inflammatory agents from mast cells and the transmission of painful stimuli from intracranial vessels.17 The CGRP receptor or ligand can be targeted by small-molecule receptor antagonists for acute and preventive migraine treatment (and by monoclonal antibodies solely for prevention, discussed later). It has been theorized that gepants bind to CGRP receptors, resulting in decreased blood flow to the brain, inhibition of neurogenic inflammation, and reduced pain signaling.17 Unlike triptans and ergotamine derivatives, these novel treatments do not constrict blood vessels and may have a unique role in patients with contraindications to triptans.
The 3 gepants approved for acute treatment—ubrogepant (Ubrelvy),18 rimegepant (Nurtec),19 and zavegepant (Zavzpret)20—were compared with placebo in clinical trials and were shown to increase the number of patients who were completely pain free at 2 hours, were free of the most bothersome associated symptom (photophobia, phonophobia, or nausea) at 2 hours, and remained pain free at 24 hours (TABLE 418-24).

Continue to: Ubrogrepant
Ubrogepant, in 2 Phase 3 trials (ACHIEVE I and ACHIEVE II) demonstrated effectiveness compared with placebo.21,22 The most common adverse effects reported were nausea and somnolence at very low rates. Pain-relief rates at 2 hours post dose (> 60% of participants) were higher than pain-free rates, and a significantly higher percentage (> 40%) of ubrogepant-treated participants reported ability to function normally on the Functional Disability Scale.25
Rimegepant was also superior to placebo (59% vs 43%) in pain relief at 2 hours post dose and other secondary endpoints.23 Rimegepant also has potential drug interactions
Zavegepant, approved in March 2023, is administered once daily as a 10-mg nasal spray. In its Phase 3 trial, zavegepant was significantly superior to placebo at 2 hours post dose in freedom from pain (24% v 15%), and in freedom from the most bothersome symptom (40% v 31%).24 Dosage modifications are not needed with mild-to-moderate renal or hepatic disease.20
Worth noting. The safety of using ubrogepant to treat more than 8 migraine episodes in a 30-day period has not been established. The safety of using more than 18 doses of zavegepant in a 30-day period also has not been established. With ubrogepant and rimegepant, there are dosing modifications for concomitant use with specific drugs (CYP3A4 inhibitors and inducers) due to potential interactions and in patients with hepatic or renal impairment.18,19
There are no trials comparing efficacy of CGRP antagonists to triptans. Recognizing that these newer medications would be costly, the AHS position statement released in 2019 recommends that gepants be considered for those with contraindications to triptans or for whom at least 2 oral triptans have failed (as determined by a validated patient outcome questionnaire).10 Step therapy with documentation of previous trials and therapy failures is often required by insurance companies prior to gepant coverage.
Continue to: Preventive therapies
Preventive therapies
Preventive migraine therapies are used to reduce duration, frequency, and severity of attacks, the need for acute treatment, and overall headache disability.26 Medications typically are chosen based on efficacy, adverse effect profile, and patient comorbidities. Barriers to successful use include poor patient adherence and tolerability, the need for slow dose titration, and long-term use (minimum of 2 months) at maximum tolerated or minimum effective doses. Medications with established efficacy (Level Aa) based on the 2012 guidelines from the American Academy of Neurology (AAN) and the AHS are given in TABLE 5.27-29

Drugs having received the strongest level of evidence for migraine prevention are metoprolol, propranolol, timolol, topiramate, valproate sodium, divalproex sodium, and onabotulinumtoxinA (Botox), and frovatriptan for menstrual migraine prevention. Because these guidelines were last updated in 2012, they did not cover gepants (which will be discussed shortly). The AHS released a position statement in 2019 supporting the use of
CGRP-targeted prevention
Four anti-CGRP mAbs and 2 gepants have been approved for migraine prevention in the United States. Differences between products include targets (ligand vs receptor), antibody IgG subtype, bioavailability, route of administration, and frequency of administration.28 As noted in the Phase 3 studies (TABLE 619,30-47), these therapies are highly efficacious, safe, and tolerable.
Gepants. Rimegepant, discussed earlier for migraine treatment, is one of the CGRP receptor antagonists approved for prevention. The other is atogepant (Qulipta), approved only for prevention. Ubrogepant is not approved for prevention.
Anti-CGRP mAb is the only medication class specifically created for migraine prevention.10,26 As already noted, several efficacious non-CGRP treatment options are available for migraine prevention. However, higher doses of those agents, if needed,
Continue to: The targeted anti-CGRP approach...
The targeted anti-CGRP approach, which can be used by patients with liver or kidney disease, results in decreased toxicity and minimal drug interactions. Long half-lives allow for monthly or quarterly injections, possibly resulting in increased compliance.28 Dose titration is not needed, allowing for more rapid symptom management. The large molecular size of a mAb limits its transfer across the blood-brain barrier, making central nervous system adverse effects unlikely.28 Despite the compelling mAb pharmacologic properties, their use may be limited by a lack of long-term safety data and the need for parenteral administration. Although immunogenicity—the development of neutralizing antibodies—can limit long-term tolerability or efficacy of mAbs generally,26,28 anti-CGRP mAbs were engineered to minimally activate the immune system and have not been associated with immune suppression, opportunistic infections, malignancies, or decreased efficacy.28
A pooled meta-analysis including 4 trials (3166 patients) found that CGRP mAbs compared with placebo significantly improved patient response rates, defined as at least a 50% and 75% reduction in monthly headache/migraine days from baseline to Weeks 9 to 12.48 Another meta-analysis including 8 trials (2292 patients) found a significant reduction from baseline in monthly migraine days and monthly acute migraine medication consumption among patients taking CGRP mAbs compared with those taking placebo.49 Open-label extension studies have shown progressive and cumulative benefits in individuals who respond to anti-CGRP mAbs. Therefore, several treatment cycles may be necessary to determine overall efficacy of therapy.10,28
Cost initially can be a barrier. Insurance companies often require step therapy before agreeing to cover mAb therapy, which aligns with the 2019 AHS position statement.10
When combination treatment may be appropriate
Monotherapy is the usual approach to preventing migraine due to advantages of efficacy, simplified regimens, lower cost, and reduced adverse effects.51 However, if a patient does not benefit from monotherapy even after trying dose titrations as tolerated or switching therapies, trying complementary combination therapy is appropriate. Despite a shortage of clinical trials supporting the use of 2 or more preventive medications with different mechanisms of action, this strategy is used clinically.10 Consider combination therapy in those with refractory disease, partial responses, or intolerance to recommended doses.52 Articles reporting on case study reviews have rationalized the combined use of onabotulinumtoxinA and anti-CGRP mAbs, noting better migraine control.51,53 The 2019 AHS position statement recommends adding a mAb to an existing preventive treatment regimen with no other changes until mAb effectiveness is determined, as the risk for drug interactions on dual therapy is low.10 Safety and efficacy also have been demonstrated with the combination of preventive anti-CGRP mAbs and acute treatment with gepants as needed.54
CORRESPONDENCE
Emily Peterson, PharmD, BCACP, 3640 Middlebury Road, Iowa City, IA 52242; [email protected]
Migraine is a headache disorder that often causes unilateral pain, photophobia, phonophobia, nausea, and vomiting. More than 70% of office visits for migraine are made to primary care physicians.1 Recent data suggest migraine may be caused primarily by neuronal dysfunction and only secondarily by vasodilation.2 Although there are numerous classes of drugs used for migraine prevention and treatment, their success has been limited by inadequate efficacy, tolerability, and patient adherence.3 The discovery of pro-inflammatory markers such as calcitonin gene-related peptide (CGRP) has led to the development of new medications to prevent and treat migraine.4

Pathophysiology, Dx and triggers, indications for pharmacotherapy
Pathophysiology. A migraine is thought to be caused by cortical spreading depression (CSD), a depolarization of glial and neuronal cell membranes.5
Dx and triggers. In 2018, the International Headache Society revised its guidelines for the diagnosis of migraine.7 According to the 3rd edition of The International Classification of Headache Disorders (ICHD-3), the diagnosis of migraine is made when a patient has at least 5 headache attacks that last 4 to 72 hours and have at least 2 of the following characteristics: (1) unilateral location, (2) pulsating quality, (3) moderate-to-severe pain intensity, and (4) aggravated by or causing avoidance of routine physical activity.7 The headache attacks also should have (1) associated nausea or vomiting or (2) photophobia and phonophobia.7 The presence of atypical signs or symptoms as indicated by the SNNOOP10 mnemonic raises concerns for secondary headaches and the need for further investigation into the cause of the headache (TABLE 1).8 It is not possible to detect every secondary headache with standard neuroimaging, but the SNNOOP10 red flags can help determine when imaging may be indicated.8 Potential triggers for migraine can be found in TABLE 2.9

Indications for pharmacotherapy. All patients receiving a diagnosis of migraine should be offered acute pharmacologic treatment. Consider preventive therapy anytime there are ≥ 4 headache days per month, debilitating attacks despite acute therapy, overuse of acute medication (> 2 d/wk), difficulty tolerating acute medication, patient preference, or presence of certain migraine subtypes.7,10

Acute treatments
Abortive therapies for migraine include analgesics such as nonsteroidal anti-inflammatory drugs (NSAIDs) and acetaminophen, and ergot alkaloids, triptans, or small-molecule CGRP receptor antagonists (gepants). Prompt administration increases the chance of success with acute therapy. Medications with the highest levels of efficacy based on the 2015 guidelines from the American Headache Society (AHS) are given in TABLE 3.11 Lasmiditan (Reyvow) is not included in the 2015 guidelines, as it was approved after publication of the guidelines.


Non-CGRP first-line therapies
NSAIDs and acetaminophen. NSAIDs such as aspirin, diclofenac, ibuprofen, and naproxen have a high level of evidence to support their use as first-line treatments for mild-to-moderate migraine attacks. Trials consistently demonstrate their superiority to placebo in headache relief and complete pain relief at 2 hours. There is no recommendation for selecting one NSAID over another; however, consider their frequency of dosing and adverse effect profiles. The number needed to treat for complete pain relief at 2 hours ranges from 7 to 10 for most NSAIDs.11,12 In some placebo-controlled studies, acetaminophen was less effective than NSAIDs, but was safer because it did not cause gastric irritation or antiplatelet effects.12
Triptans inhibit 5-HT1B/1D receptors. Consider formulation, route of administration, cost, and pharmacokinetics when selecting a triptan. Patients who do not respond well to one triptan may respond favorably to another. A meta-analysis of the effectiveness of the 7 available agents found that triptans at standard doses provided pain relief within 2 hours in 42% to 76% of patients, and sustained freedom from pain for 2 hours in 18% to 50% of patients.13 Lasmiditan is a selective serotonin receptor (5-HT1F) agonist that lacks vasoconstrictor activity. This is an option for patients with relative contraindications to triptans due to cardiovascular risk factors.10
Continue to: Second-line therapies
Second-line therapies
Intranasal dihydroergotamine has a favorable adverse event profile and greater evidence for efficacy compared with ergotamine. Compared with triptans, intranasal dihydroergotamine has a high level of efficacy but causes more adverse effects.14 Severe nausea is common, and dihydroergotamine often is used in combination with an antiemetic drug. Dihydroergotamine should not be used within 24 hours of taking a triptan, and it is contraindicated for patients who have hypertension or ischemic heart disease or who are pregnant or breastfeeding. There is also the potential for adverse drug interactions.15
Antiemetics may be helpful for migraine associated with severe nausea or vomiting. The dopamine antagonists metoclopramide, prochlorperazine, and chlorpromazine have demonstrated benefit in randomized placebo-controlled trials.11 Ondansetron has not been studied extensively, but sometimes is used in clinical practice. Nonoral routes of administration may be useful in patients having trouble swallowing medications or in those experiencing significant nausea or vomiting early during migraine attacks.
Due to the high potential for abuse, opioids should not be used routinely for the treatment of migraine.12 There is no high-quality evidence supporting the efficacy of barbiturates (ie, butalbital-containing compounds) for acute migraine treatment.11 Moreover, use of these agents may increase the likelihood of progression from episodic to chronic migraine.16
Gepants for acute migraine treatment
Neuropeptide CGRP is released from trigeminal nerves and is a potent dilator of cerebral and dural vessels, playing a key role in regulating blood flow to the brain. Other roles of CGRP include the release of inflammatory agents from mast cells and the transmission of painful stimuli from intracranial vessels.17 The CGRP receptor or ligand can be targeted by small-molecule receptor antagonists for acute and preventive migraine treatment (and by monoclonal antibodies solely for prevention, discussed later). It has been theorized that gepants bind to CGRP receptors, resulting in decreased blood flow to the brain, inhibition of neurogenic inflammation, and reduced pain signaling.17 Unlike triptans and ergotamine derivatives, these novel treatments do not constrict blood vessels and may have a unique role in patients with contraindications to triptans.
The 3 gepants approved for acute treatment—ubrogepant (Ubrelvy),18 rimegepant (Nurtec),19 and zavegepant (Zavzpret)20—were compared with placebo in clinical trials and were shown to increase the number of patients who were completely pain free at 2 hours, were free of the most bothersome associated symptom (photophobia, phonophobia, or nausea) at 2 hours, and remained pain free at 24 hours (TABLE 418-24).

Continue to: Ubrogrepant
Ubrogepant, in 2 Phase 3 trials (ACHIEVE I and ACHIEVE II) demonstrated effectiveness compared with placebo.21,22 The most common adverse effects reported were nausea and somnolence at very low rates. Pain-relief rates at 2 hours post dose (> 60% of participants) were higher than pain-free rates, and a significantly higher percentage (> 40%) of ubrogepant-treated participants reported ability to function normally on the Functional Disability Scale.25
Rimegepant was also superior to placebo (59% vs 43%) in pain relief at 2 hours post dose and other secondary endpoints.23 Rimegepant also has potential drug interactions
Zavegepant, approved in March 2023, is administered once daily as a 10-mg nasal spray. In its Phase 3 trial, zavegepant was significantly superior to placebo at 2 hours post dose in freedom from pain (24% v 15%), and in freedom from the most bothersome symptom (40% v 31%).24 Dosage modifications are not needed with mild-to-moderate renal or hepatic disease.20
Worth noting. The safety of using ubrogepant to treat more than 8 migraine episodes in a 30-day period has not been established. The safety of using more than 18 doses of zavegepant in a 30-day period also has not been established. With ubrogepant and rimegepant, there are dosing modifications for concomitant use with specific drugs (CYP3A4 inhibitors and inducers) due to potential interactions and in patients with hepatic or renal impairment.18,19
There are no trials comparing efficacy of CGRP antagonists to triptans. Recognizing that these newer medications would be costly, the AHS position statement released in 2019 recommends that gepants be considered for those with contraindications to triptans or for whom at least 2 oral triptans have failed (as determined by a validated patient outcome questionnaire).10 Step therapy with documentation of previous trials and therapy failures is often required by insurance companies prior to gepant coverage.
Continue to: Preventive therapies
Preventive therapies
Preventive migraine therapies are used to reduce duration, frequency, and severity of attacks, the need for acute treatment, and overall headache disability.26 Medications typically are chosen based on efficacy, adverse effect profile, and patient comorbidities. Barriers to successful use include poor patient adherence and tolerability, the need for slow dose titration, and long-term use (minimum of 2 months) at maximum tolerated or minimum effective doses. Medications with established efficacy (Level Aa) based on the 2012 guidelines from the American Academy of Neurology (AAN) and the AHS are given in TABLE 5.27-29

Drugs having received the strongest level of evidence for migraine prevention are metoprolol, propranolol, timolol, topiramate, valproate sodium, divalproex sodium, and onabotulinumtoxinA (Botox), and frovatriptan for menstrual migraine prevention. Because these guidelines were last updated in 2012, they did not cover gepants (which will be discussed shortly). The AHS released a position statement in 2019 supporting the use of
CGRP-targeted prevention
Four anti-CGRP mAbs and 2 gepants have been approved for migraine prevention in the United States. Differences between products include targets (ligand vs receptor), antibody IgG subtype, bioavailability, route of administration, and frequency of administration.28 As noted in the Phase 3 studies (TABLE 619,30-47), these therapies are highly efficacious, safe, and tolerable.
Gepants. Rimegepant, discussed earlier for migraine treatment, is one of the CGRP receptor antagonists approved for prevention. The other is atogepant (Qulipta), approved only for prevention. Ubrogepant is not approved for prevention.
Anti-CGRP mAb is the only medication class specifically created for migraine prevention.10,26 As already noted, several efficacious non-CGRP treatment options are available for migraine prevention. However, higher doses of those agents, if needed,
Continue to: The targeted anti-CGRP approach...
The targeted anti-CGRP approach, which can be used by patients with liver or kidney disease, results in decreased toxicity and minimal drug interactions. Long half-lives allow for monthly or quarterly injections, possibly resulting in increased compliance.28 Dose titration is not needed, allowing for more rapid symptom management. The large molecular size of a mAb limits its transfer across the blood-brain barrier, making central nervous system adverse effects unlikely.28 Despite the compelling mAb pharmacologic properties, their use may be limited by a lack of long-term safety data and the need for parenteral administration. Although immunogenicity—the development of neutralizing antibodies—can limit long-term tolerability or efficacy of mAbs generally,26,28 anti-CGRP mAbs were engineered to minimally activate the immune system and have not been associated with immune suppression, opportunistic infections, malignancies, or decreased efficacy.28
A pooled meta-analysis including 4 trials (3166 patients) found that CGRP mAbs compared with placebo significantly improved patient response rates, defined as at least a 50% and 75% reduction in monthly headache/migraine days from baseline to Weeks 9 to 12.48 Another meta-analysis including 8 trials (2292 patients) found a significant reduction from baseline in monthly migraine days and monthly acute migraine medication consumption among patients taking CGRP mAbs compared with those taking placebo.49 Open-label extension studies have shown progressive and cumulative benefits in individuals who respond to anti-CGRP mAbs. Therefore, several treatment cycles may be necessary to determine overall efficacy of therapy.10,28
Cost initially can be a barrier. Insurance companies often require step therapy before agreeing to cover mAb therapy, which aligns with the 2019 AHS position statement.10
When combination treatment may be appropriate
Monotherapy is the usual approach to preventing migraine due to advantages of efficacy, simplified regimens, lower cost, and reduced adverse effects.51 However, if a patient does not benefit from monotherapy even after trying dose titrations as tolerated or switching therapies, trying complementary combination therapy is appropriate. Despite a shortage of clinical trials supporting the use of 2 or more preventive medications with different mechanisms of action, this strategy is used clinically.10 Consider combination therapy in those with refractory disease, partial responses, or intolerance to recommended doses.52 Articles reporting on case study reviews have rationalized the combined use of onabotulinumtoxinA and anti-CGRP mAbs, noting better migraine control.51,53 The 2019 AHS position statement recommends adding a mAb to an existing preventive treatment regimen with no other changes until mAb effectiveness is determined, as the risk for drug interactions on dual therapy is low.10 Safety and efficacy also have been demonstrated with the combination of preventive anti-CGRP mAbs and acute treatment with gepants as needed.54
CORRESPONDENCE
Emily Peterson, PharmD, BCACP, 3640 Middlebury Road, Iowa City, IA 52242; [email protected]
1. Lipton RB, Nicholson RA, Reed ML, et al. Diagnosis, consultation, treatment, and impact of migraine in the US: results of the OVERCOME (US) study. Headache. 2022;62:122-140. doi: 10.1111/head.14259
2. Burstein R, Noseda R, Borsook D. Migraine: multiple processes; complext pathophysiology. J Neurosci. 2015;35:6619-6629. doi: 10.1523/JNEUROSCI.0373-15.2015
3. Edvinsson L, Haanes KA, Warfvinge K, et al. CGRP as the target of new migraine therapies - successful translation from bench to clinic. Nat Rev Neurol. 2018;14:338-350. doi: 10.1038/s41582-018-0003-1
4. McGrath K, Rague A, Thesing C, et al. Migraine: expanding our Tx arsenal. J Fam Pract. 2019;68:10-14;16-24.
5. Dodick DW. Migraine. Lancet. 2018;391:1315-1330. doi: 10.1016/S0140-6736(18)30478-1
6. Agostoni EC, Barbanti P, Calabresi P, et al. Current and emerging evidence-based treatment options in chronic migraine: a narrative review. J Headache Pain. 2019;20:92. doi: 10.1186/s10194-019-1038-4
7. IHS. Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3rd edition. Cephalalgia. 2018;38:1-211. doi: 10.1177/0333102417738202
8. Do TP, Remmers A, Schytz HW, et al. Red and orange flags for secondary headaches in clinical practice: SNNOOP10 list. Neurology. 2019;92:134-144. doi: 10.1212/WNL.0000000000006697
9. NIH. Migraine. Accessed July 30, 2023.
10. AHS. The American Headache Society position statement on integrating new migraine treatments into clinical practice. Headache. 2019;59:1-18. doi: 10.1111/head.13456
11. Marmura MJ, Silberstein SD, Schwedt TJ. The acute treatment of migraine in adults: the American Headache Society evidence assessment of migraine pharmacotherapies. Headache. 2015;55:3-20. doi: 10.1111/head.12499
12. Mayans L, Walling A. Acute migraine headache: treatment strategies. Am Fam Physician. 2018;97:243-251.
13. Cameron C, Kelly S, Hsieh SC, et al. Triptans in the acute treatment of migraine: a systematic review and network meta-analysis. Headache. 2015;55(suppl 4):221-235. doi: 10.1111/head.12601
14. Becker WJ. Acute migraine treatment. Continuum (Minneap Minn). 2015;21:953-972. doi: 10.1212/CON.0000000000000192
15. Migranal (dihydroergotamine mesylate) Package insert. Valeant Pharmaceuticals North America; 2019. Accessed June 17, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2019/020148Orig1s025lbl.pdf
16. Minen MT, Tanev K, Friedman BW. Evaluation and treatment of migraine in the emergency department: a review. Headache. 2014;54:1131-45. doi: 10.1111/head.12399
17. Durham PL. CGRP-receptor antagonists--a fresh approach to migraine therapy? N Engl J Med. 2004;350:1073-1075. doi: 10.1056/NEJMp048016
18. Ubrelvy (ubrogepant). Package insert. Allergan, Inc.; 2019. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2019/211765s000lbl.pdf
19. Nurtec ODT (rimegepant sulfate). Package insert. Biohaven Pharmaceuticals, Inc.; 2021. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2021/212728s006lbl.pdf
20. Zavzpret (zavegepant). Package insert. Pfizer Labs.; 2023. Accessed July 15, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2023/216386s000lbl.pdf
21. Dodick DW, Lipton RB, Ailani J, et al. Ubrogepant for the treatment of migraine. N Engl J Med. 2019;381:2230-2241. doi: 10.1056/NEJMoa1813049
22. Lipton RB, Dodick DW, Ailani J, et al. Effect of ubrogepant vs placebo on pain and the most bothersome associated symptom in the acute treatment of migraine: the ACHIEVE II randomized clinical trial. JAMA. 2019;322:1887-1898. doi: 10.1001/jama.2019.16711
23. Croop R, Goadsby PJ, Stock DA, et al. Efficacy, safety, and tolerability of rimegepant orally disintegrating tablet for the acute treatment of migraine: a randomised, phase 3, double-blind, placebo-controlled trial. Lancet. 2019;394:737-745. doi: 10.1016/S0140-6736(19)31606-X
24. Lipton RB, Croop R, Stock DA, et al. Safety, tolerability, and efficacy of zavegepant 10 mg nasal spray for the acute treatment of migraine in the USA: a phase 3, double-blind, randomised, placebo-controlled multicentre trial. Lancet Neurol. 2023;22:209-217. doi: 10.1016/S1474-4422(22)00517-8
25. Dodick DW, Lipton RB, Ailani J, et al. Ubrogepant, an acute treatment for migraine, improved patient-reported functional disability and satisfaction in 2 single-attack phase 3 randomized trials, ACHIEVE I and II. Headache. 2020;60:686-700. doi: 10.1111/head.13766
26. Burch R. Migraine and tension-type headache: diagnosis and treatment. Med Clin North Am. 2019;103:215-233. doi:10.1016/j.mcna.2018.10.003
27. Silberstein SD, Holland S, Freitag F, et al. Evidence-based guideline update: pharmacologic treatment for episodic migraine prevention in adults: report of the Quality Standards Subcommittee of the American Academy of Neurology and the American Headache Society. Neurology. 2012;78:1337-1345. doi: 10.1212/WNL.0b013e3182535d20
28. Dodick DW. CGRP ligand and receptor monoclonal antibodies for migraine prevention: evidence review and clinical implications. Cephalalgia. 2019;39:445-458. doi: 10.1177/ 0333102418821662
29. Pringsheim T, Davenport WJ, Becker WJ. Prophylaxis of migraine headache. CMAJ. 2010;182:E269-276. doi: 10.1503/cmaj.081657
30. Vyepti (eptinezumab-jjmr). Package insert. Lundbeck Pharmaceuticals LLV; 2020. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2020/761119s000lbl.pdf
31. Aimovig (erenumab-aooe). Package insert. Amgen Inc.; 2021. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2021/761077s009lbl.pdf
32. Ajovy (fremanezumab-vfrm). Package insert. Teva Pharmaceuticals USA, Inc.; 2018. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2018/761089s000lbl.pdf
33. Emgality (galcanezumab-gnlm). Package insert. Eli Lilly and Company; 2018. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2018/761063s000lbl.pdf
34. Ashina M, Saper J, Cady R, et al. Eptinezumab in episodic migraine: a randomized, double-blind, placebo-controlled study (PROMISE-1). Cephalalgia. 2020;40:241-254. doi: 10.1177/0333102420905132
35. Lipton RB, Goadsby PJ, Smith J, et al. Efficacy and safety of eptinezumab in patients with chronic migraine: PROMISE-2. Neurology. 2020;94:e1365-e1377. doi: 10.1212/WNL.0000000000009169
36. Dodick DW, Ashina M, Brandes JL, et al. ARISE: a phase 3 randomized trial of erenumab for episodic migraine. Cephalalgia. 2018;38:1026-1037. doi: 10.1177/0333102418759786
37. Goadsby PJ, Reuter U, Hallström Y, et al. A controlled trial of erenumab for episodic migraine. N Engl J Med. 2017;377:2123-2132. doi: 10.1056/NEJMoa1705848
38. Reuter U, Goadsby PJ, Lanteri-Minet M, et al. Efficacy and tolerability of erenumab in patients with episodic migraine in whom two-to-four previous preventive treatments were unsuccessful: a randomised, double-blind, placebo-controlled, phase 3b study. Lancet. 2018;392:2280-2287. doi: 10.1016/S0140-6736(18)32534-0
39. Silberstein SD, Dodick DW, Bigal ME, et al. Fremanezumab for the preventive treatment of chronic migraine. N Engl J Med. 2017; 377:2113-2122. doi: 10.1056/NEJMoa1709038
40. Dodick DW, Silberstein SD, Bigal ME, et al. Effect of fremanezumab compared with placebo for prevention of episodic migraine: a randomized clinical trial. JAMA. 2018;319:1999-2008. doi: 10.1001/jama.2018.4853
41. Stauffer VL, Dodick DW, Zhang Q, et al. Evaluation of galcanezumab for the prevention of episodic migraine: the EVOLVE-1 randomized clinical trial. JAMA Neurol. 2018;75:1080-1088. doi: 10.1001/jamaneurol.2018.1212
42. Skljarevski V, Matharu M, Millen BA, et al. Efficacy and safety of galcanezumab for the prevention of episodic migraine: results of the EVOLVE-2 phase 3 randomized controlled clinical trial. Cephalalgia. 2018;38:1442-1454. doi: 10.1177/0333102418779543
43. Detke HC, Goadsby PJ, Wang S, et al. Galcanezumab in chronic migraine: the randomized, double-blind, placebo-controlled REGAIN study. Neurology. 2018;91:e2211-e2221. doi: 10.1212/WNL.0000000000006640
44. Goadsby PJ, Dodick DW, Leone M, at al. Trial of galcanezumab in prevention of episodic cluster headache. N Engl J Med. 2019; 381:132-141. doi: 10.1056/NEJMoa1813440
45. Croop R, Lipton RB, Kudrow D, et al. Oral rimegepant for preventive treatment of migraine: a phase 2/3, randomised, double-blind, placebo-controlled trial. Lancet. 2021;397:51-60. doi: 10.1016/S0140-6736(20)32544-7
46. Ailani J, Lipton RB, Goadsby PJ, et al. Atogepant for the preventive treatment of migraine. N Engl J Med. 2021;385:695-706. doi: 10.1056/NEJMoa2035908
47. Qulipta (atogepant). Package insert. AbbVie; 2021. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2021/215206Orig1s000lbl.pdf
48. Han L, Liu Y, Xiong H, et al. CGRP monoclonal antibody for preventive treatment of chronic migraine: an update of meta-analysis. Brain Behav. 2019;9:e01215. doi: 10.1002/brb3.1215
49. Zhu Y, Liu Y, Zhao J, et al. The efficacy and safety of calcitonin gene-related peptide monoclonal antibody for episodic migraine: a meta-analysis. Neurol Sci. 2018;39:2097-2106. doi: 10.1007/s10072-018-3547-3
50. Szperka CL, VanderPluym J, Orr SL, et al. Recommendations on the use of anti-CGRP monoclonal antibodies in children and adolescents. Headache. 2018;58:1658-1669. doi: 10.1111/head.13414
51. Pellesi L, Do TP, Ashina H, et al. Dual therapy with anti-CGRP monoclonal antibodies and botulinum toxin for migraine prevention: is there a rationale? Headache. 2020;60:1056-1065. doi: 10.1111/head.13843
52. D’Antona L, Matharu M. Identifying and managing refractory migraine: barriers and opportunities? J Headache Pain. 2019;20:89. doi: 10.1186/s10194-019-1040-x
53. Cohen F, Armand C, Lipton RB, et al. Efficacy and tolerability of calcitonin gene-related peptide targeted monoclonal antibody medications as add-on therapy to onabotulinumtoxinA in patients with chronic migraine. Pain Med. 2021;1857-1863. doi: 10.1093/pm/pnab093
54. Berman G, Croop R, Kudrow D, et al. Safety of rimegepant, an oral CGRP receptor antagonist, plus CGRP monoclonal antibodies for migraine. Headache. 2020;60:1734-1742. doi: 10.1111/head.13930
1. Lipton RB, Nicholson RA, Reed ML, et al. Diagnosis, consultation, treatment, and impact of migraine in the US: results of the OVERCOME (US) study. Headache. 2022;62:122-140. doi: 10.1111/head.14259
2. Burstein R, Noseda R, Borsook D. Migraine: multiple processes; complext pathophysiology. J Neurosci. 2015;35:6619-6629. doi: 10.1523/JNEUROSCI.0373-15.2015
3. Edvinsson L, Haanes KA, Warfvinge K, et al. CGRP as the target of new migraine therapies - successful translation from bench to clinic. Nat Rev Neurol. 2018;14:338-350. doi: 10.1038/s41582-018-0003-1
4. McGrath K, Rague A, Thesing C, et al. Migraine: expanding our Tx arsenal. J Fam Pract. 2019;68:10-14;16-24.
5. Dodick DW. Migraine. Lancet. 2018;391:1315-1330. doi: 10.1016/S0140-6736(18)30478-1
6. Agostoni EC, Barbanti P, Calabresi P, et al. Current and emerging evidence-based treatment options in chronic migraine: a narrative review. J Headache Pain. 2019;20:92. doi: 10.1186/s10194-019-1038-4
7. IHS. Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3rd edition. Cephalalgia. 2018;38:1-211. doi: 10.1177/0333102417738202
8. Do TP, Remmers A, Schytz HW, et al. Red and orange flags for secondary headaches in clinical practice: SNNOOP10 list. Neurology. 2019;92:134-144. doi: 10.1212/WNL.0000000000006697
9. NIH. Migraine. Accessed July 30, 2023.
10. AHS. The American Headache Society position statement on integrating new migraine treatments into clinical practice. Headache. 2019;59:1-18. doi: 10.1111/head.13456
11. Marmura MJ, Silberstein SD, Schwedt TJ. The acute treatment of migraine in adults: the American Headache Society evidence assessment of migraine pharmacotherapies. Headache. 2015;55:3-20. doi: 10.1111/head.12499
12. Mayans L, Walling A. Acute migraine headache: treatment strategies. Am Fam Physician. 2018;97:243-251.
13. Cameron C, Kelly S, Hsieh SC, et al. Triptans in the acute treatment of migraine: a systematic review and network meta-analysis. Headache. 2015;55(suppl 4):221-235. doi: 10.1111/head.12601
14. Becker WJ. Acute migraine treatment. Continuum (Minneap Minn). 2015;21:953-972. doi: 10.1212/CON.0000000000000192
15. Migranal (dihydroergotamine mesylate) Package insert. Valeant Pharmaceuticals North America; 2019. Accessed June 17, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2019/020148Orig1s025lbl.pdf
16. Minen MT, Tanev K, Friedman BW. Evaluation and treatment of migraine in the emergency department: a review. Headache. 2014;54:1131-45. doi: 10.1111/head.12399
17. Durham PL. CGRP-receptor antagonists--a fresh approach to migraine therapy? N Engl J Med. 2004;350:1073-1075. doi: 10.1056/NEJMp048016
18. Ubrelvy (ubrogepant). Package insert. Allergan, Inc.; 2019. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2019/211765s000lbl.pdf
19. Nurtec ODT (rimegepant sulfate). Package insert. Biohaven Pharmaceuticals, Inc.; 2021. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2021/212728s006lbl.pdf
20. Zavzpret (zavegepant). Package insert. Pfizer Labs.; 2023. Accessed July 15, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2023/216386s000lbl.pdf
21. Dodick DW, Lipton RB, Ailani J, et al. Ubrogepant for the treatment of migraine. N Engl J Med. 2019;381:2230-2241. doi: 10.1056/NEJMoa1813049
22. Lipton RB, Dodick DW, Ailani J, et al. Effect of ubrogepant vs placebo on pain and the most bothersome associated symptom in the acute treatment of migraine: the ACHIEVE II randomized clinical trial. JAMA. 2019;322:1887-1898. doi: 10.1001/jama.2019.16711
23. Croop R, Goadsby PJ, Stock DA, et al. Efficacy, safety, and tolerability of rimegepant orally disintegrating tablet for the acute treatment of migraine: a randomised, phase 3, double-blind, placebo-controlled trial. Lancet. 2019;394:737-745. doi: 10.1016/S0140-6736(19)31606-X
24. Lipton RB, Croop R, Stock DA, et al. Safety, tolerability, and efficacy of zavegepant 10 mg nasal spray for the acute treatment of migraine in the USA: a phase 3, double-blind, randomised, placebo-controlled multicentre trial. Lancet Neurol. 2023;22:209-217. doi: 10.1016/S1474-4422(22)00517-8
25. Dodick DW, Lipton RB, Ailani J, et al. Ubrogepant, an acute treatment for migraine, improved patient-reported functional disability and satisfaction in 2 single-attack phase 3 randomized trials, ACHIEVE I and II. Headache. 2020;60:686-700. doi: 10.1111/head.13766
26. Burch R. Migraine and tension-type headache: diagnosis and treatment. Med Clin North Am. 2019;103:215-233. doi:10.1016/j.mcna.2018.10.003
27. Silberstein SD, Holland S, Freitag F, et al. Evidence-based guideline update: pharmacologic treatment for episodic migraine prevention in adults: report of the Quality Standards Subcommittee of the American Academy of Neurology and the American Headache Society. Neurology. 2012;78:1337-1345. doi: 10.1212/WNL.0b013e3182535d20
28. Dodick DW. CGRP ligand and receptor monoclonal antibodies for migraine prevention: evidence review and clinical implications. Cephalalgia. 2019;39:445-458. doi: 10.1177/ 0333102418821662
29. Pringsheim T, Davenport WJ, Becker WJ. Prophylaxis of migraine headache. CMAJ. 2010;182:E269-276. doi: 10.1503/cmaj.081657
30. Vyepti (eptinezumab-jjmr). Package insert. Lundbeck Pharmaceuticals LLV; 2020. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2020/761119s000lbl.pdf
31. Aimovig (erenumab-aooe). Package insert. Amgen Inc.; 2021. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2021/761077s009lbl.pdf
32. Ajovy (fremanezumab-vfrm). Package insert. Teva Pharmaceuticals USA, Inc.; 2018. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2018/761089s000lbl.pdf
33. Emgality (galcanezumab-gnlm). Package insert. Eli Lilly and Company; 2018. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2018/761063s000lbl.pdf
34. Ashina M, Saper J, Cady R, et al. Eptinezumab in episodic migraine: a randomized, double-blind, placebo-controlled study (PROMISE-1). Cephalalgia. 2020;40:241-254. doi: 10.1177/0333102420905132
35. Lipton RB, Goadsby PJ, Smith J, et al. Efficacy and safety of eptinezumab in patients with chronic migraine: PROMISE-2. Neurology. 2020;94:e1365-e1377. doi: 10.1212/WNL.0000000000009169
36. Dodick DW, Ashina M, Brandes JL, et al. ARISE: a phase 3 randomized trial of erenumab for episodic migraine. Cephalalgia. 2018;38:1026-1037. doi: 10.1177/0333102418759786
37. Goadsby PJ, Reuter U, Hallström Y, et al. A controlled trial of erenumab for episodic migraine. N Engl J Med. 2017;377:2123-2132. doi: 10.1056/NEJMoa1705848
38. Reuter U, Goadsby PJ, Lanteri-Minet M, et al. Efficacy and tolerability of erenumab in patients with episodic migraine in whom two-to-four previous preventive treatments were unsuccessful: a randomised, double-blind, placebo-controlled, phase 3b study. Lancet. 2018;392:2280-2287. doi: 10.1016/S0140-6736(18)32534-0
39. Silberstein SD, Dodick DW, Bigal ME, et al. Fremanezumab for the preventive treatment of chronic migraine. N Engl J Med. 2017; 377:2113-2122. doi: 10.1056/NEJMoa1709038
40. Dodick DW, Silberstein SD, Bigal ME, et al. Effect of fremanezumab compared with placebo for prevention of episodic migraine: a randomized clinical trial. JAMA. 2018;319:1999-2008. doi: 10.1001/jama.2018.4853
41. Stauffer VL, Dodick DW, Zhang Q, et al. Evaluation of galcanezumab for the prevention of episodic migraine: the EVOLVE-1 randomized clinical trial. JAMA Neurol. 2018;75:1080-1088. doi: 10.1001/jamaneurol.2018.1212
42. Skljarevski V, Matharu M, Millen BA, et al. Efficacy and safety of galcanezumab for the prevention of episodic migraine: results of the EVOLVE-2 phase 3 randomized controlled clinical trial. Cephalalgia. 2018;38:1442-1454. doi: 10.1177/0333102418779543
43. Detke HC, Goadsby PJ, Wang S, et al. Galcanezumab in chronic migraine: the randomized, double-blind, placebo-controlled REGAIN study. Neurology. 2018;91:e2211-e2221. doi: 10.1212/WNL.0000000000006640
44. Goadsby PJ, Dodick DW, Leone M, at al. Trial of galcanezumab in prevention of episodic cluster headache. N Engl J Med. 2019; 381:132-141. doi: 10.1056/NEJMoa1813440
45. Croop R, Lipton RB, Kudrow D, et al. Oral rimegepant for preventive treatment of migraine: a phase 2/3, randomised, double-blind, placebo-controlled trial. Lancet. 2021;397:51-60. doi: 10.1016/S0140-6736(20)32544-7
46. Ailani J, Lipton RB, Goadsby PJ, et al. Atogepant for the preventive treatment of migraine. N Engl J Med. 2021;385:695-706. doi: 10.1056/NEJMoa2035908
47. Qulipta (atogepant). Package insert. AbbVie; 2021. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2021/215206Orig1s000lbl.pdf
48. Han L, Liu Y, Xiong H, et al. CGRP monoclonal antibody for preventive treatment of chronic migraine: an update of meta-analysis. Brain Behav. 2019;9:e01215. doi: 10.1002/brb3.1215
49. Zhu Y, Liu Y, Zhao J, et al. The efficacy and safety of calcitonin gene-related peptide monoclonal antibody for episodic migraine: a meta-analysis. Neurol Sci. 2018;39:2097-2106. doi: 10.1007/s10072-018-3547-3
50. Szperka CL, VanderPluym J, Orr SL, et al. Recommendations on the use of anti-CGRP monoclonal antibodies in children and adolescents. Headache. 2018;58:1658-1669. doi: 10.1111/head.13414
51. Pellesi L, Do TP, Ashina H, et al. Dual therapy with anti-CGRP monoclonal antibodies and botulinum toxin for migraine prevention: is there a rationale? Headache. 2020;60:1056-1065. doi: 10.1111/head.13843
52. D’Antona L, Matharu M. Identifying and managing refractory migraine: barriers and opportunities? J Headache Pain. 2019;20:89. doi: 10.1186/s10194-019-1040-x
53. Cohen F, Armand C, Lipton RB, et al. Efficacy and tolerability of calcitonin gene-related peptide targeted monoclonal antibody medications as add-on therapy to onabotulinumtoxinA in patients with chronic migraine. Pain Med. 2021;1857-1863. doi: 10.1093/pm/pnab093
54. Berman G, Croop R, Kudrow D, et al. Safety of rimegepant, an oral CGRP receptor antagonist, plus CGRP monoclonal antibodies for migraine. Headache. 2020;60:1734-1742. doi: 10.1111/head.13930
PRACTICE RECOMMENDATIONS
› Consider small-molecule calcitonin gene-related peptide (CGRP) receptor antagonists (gepants) for acute migraine treatment after treatment failure of at least 2 non-CGRP first-line therapies. A
› Consider anti-CGRP monoclonal antibodies or gepants for migraine prevention if traditional therapies have proven ineffective or are contraindicated or intolerable to the patient. A
› Add an anti-CGRP monoclonal antibody or gepant to existing preventive treatment if the patient continues to experience migraine. B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
FDA approves motixafortide for stem cell mobilization in myeloma
The success of autologous stem cell transplantation (ASCT) depends on adequate mobilization of stem cells during the treatment process. Collection of a sufficient number of stem cells to perform two transplantations is recommended. However, in up to 47% of patients, collecting target numbers of hematopoietic stem cells for ASCT after one apheresis session remains a challenge, BioLineRx explained in a press release today, announcing the approval.
The goal of combining motixafortide with filgrastim is to mobilize stem cells more reliably than filgrastim can alone, with fewer days of apheresis sessions and fewer doses of filgrastim.
“We believe [motixafortide] will play a critical role in addressing unmet needs and introduce a new treatment paradigm for” patients with multiple myeloma, CEO Philip Serlin said in the release.
The drug approval was based on the GENESIS trial, which randomized 122 patients to either motixafortide plus filgrastim or placebo plus filgrastim.
BioLineRx said the trial included patients considered representative of the typical multiple myeloma population undergoing ASCT, with a median age of 63 years and with about 70% of patients in both arms of the trial receiving lenalidomide-containing induction therapy.
Motixafortide plus filgrastim enabled 67.5% of patients to achieve the stem cell collection goal of 6 million or more CD34+ cells/kg within two apheresis sessions, versus 9.5% of patients receiving the placebo plus filgrastim regimen. Additionally, 92.5% of patients reached the stem cell collection goal in up to two apheresis sessions in the motixafortide arm and 21.4% in the placebo arm.
However, “the data are descriptive and were not statistically powered nor prespecified. The information should be cautiously interpreted,” the company said.
Serious adverse reactions occurred in 5.4% of patients in the motixafortide arm, including vomiting, injection-site reaction, hypersensitivity reaction, injection-site cellulitis, hypokalemia, and hypoxia. The most common adverse reactions, occurring in more than 20% of patients, were injection site reactions (pain, erythema, and pruritus), pruritus, flushing, and back pain.
Labeling for the subcutaneous injection is available online.
A version of this article first appeared on Medscape.com.
The success of autologous stem cell transplantation (ASCT) depends on adequate mobilization of stem cells during the treatment process. Collection of a sufficient number of stem cells to perform two transplantations is recommended. However, in up to 47% of patients, collecting target numbers of hematopoietic stem cells for ASCT after one apheresis session remains a challenge, BioLineRx explained in a press release today, announcing the approval.
The goal of combining motixafortide with filgrastim is to mobilize stem cells more reliably than filgrastim can alone, with fewer days of apheresis sessions and fewer doses of filgrastim.
“We believe [motixafortide] will play a critical role in addressing unmet needs and introduce a new treatment paradigm for” patients with multiple myeloma, CEO Philip Serlin said in the release.
The drug approval was based on the GENESIS trial, which randomized 122 patients to either motixafortide plus filgrastim or placebo plus filgrastim.
BioLineRx said the trial included patients considered representative of the typical multiple myeloma population undergoing ASCT, with a median age of 63 years and with about 70% of patients in both arms of the trial receiving lenalidomide-containing induction therapy.
Motixafortide plus filgrastim enabled 67.5% of patients to achieve the stem cell collection goal of 6 million or more CD34+ cells/kg within two apheresis sessions, versus 9.5% of patients receiving the placebo plus filgrastim regimen. Additionally, 92.5% of patients reached the stem cell collection goal in up to two apheresis sessions in the motixafortide arm and 21.4% in the placebo arm.
However, “the data are descriptive and were not statistically powered nor prespecified. The information should be cautiously interpreted,” the company said.
Serious adverse reactions occurred in 5.4% of patients in the motixafortide arm, including vomiting, injection-site reaction, hypersensitivity reaction, injection-site cellulitis, hypokalemia, and hypoxia. The most common adverse reactions, occurring in more than 20% of patients, were injection site reactions (pain, erythema, and pruritus), pruritus, flushing, and back pain.
Labeling for the subcutaneous injection is available online.
A version of this article first appeared on Medscape.com.
The success of autologous stem cell transplantation (ASCT) depends on adequate mobilization of stem cells during the treatment process. Collection of a sufficient number of stem cells to perform two transplantations is recommended. However, in up to 47% of patients, collecting target numbers of hematopoietic stem cells for ASCT after one apheresis session remains a challenge, BioLineRx explained in a press release today, announcing the approval.
The goal of combining motixafortide with filgrastim is to mobilize stem cells more reliably than filgrastim can alone, with fewer days of apheresis sessions and fewer doses of filgrastim.
“We believe [motixafortide] will play a critical role in addressing unmet needs and introduce a new treatment paradigm for” patients with multiple myeloma, CEO Philip Serlin said in the release.
The drug approval was based on the GENESIS trial, which randomized 122 patients to either motixafortide plus filgrastim or placebo plus filgrastim.
BioLineRx said the trial included patients considered representative of the typical multiple myeloma population undergoing ASCT, with a median age of 63 years and with about 70% of patients in both arms of the trial receiving lenalidomide-containing induction therapy.
Motixafortide plus filgrastim enabled 67.5% of patients to achieve the stem cell collection goal of 6 million or more CD34+ cells/kg within two apheresis sessions, versus 9.5% of patients receiving the placebo plus filgrastim regimen. Additionally, 92.5% of patients reached the stem cell collection goal in up to two apheresis sessions in the motixafortide arm and 21.4% in the placebo arm.
However, “the data are descriptive and were not statistically powered nor prespecified. The information should be cautiously interpreted,” the company said.
Serious adverse reactions occurred in 5.4% of patients in the motixafortide arm, including vomiting, injection-site reaction, hypersensitivity reaction, injection-site cellulitis, hypokalemia, and hypoxia. The most common adverse reactions, occurring in more than 20% of patients, were injection site reactions (pain, erythema, and pruritus), pruritus, flushing, and back pain.
Labeling for the subcutaneous injection is available online.
A version of this article first appeared on Medscape.com.
Dusty, but still cool
When I was 16, keeping my car shiny was a priority. I washed it every weekend and waxed it once a month. I was pretty good at it and got paid to do the occasional job for a neighbor, too.
In college I was busier, and my car was back at the house, so it didn’t need to be washed as much.
In medical school I think I washed the car once a year. Residency was probably the same.
Today I realized I couldn’t remember when I last had it washed (at my age I don’t have time to do it myself). So I looked it up in Quicken: Nov. 14, 2018.
Really? I’ve gone almost 5 years without washing my car? I can’t even blame that on the pandemic.
I mean, I still like my car. It’s comfortable, has good air conditioning (in Phoenix that’s critical), and gets me where I want to go. At my age those things are what’s really important. It’s hard to believe that 40 years ago, keeping a polished car was the center of my existence. Of course, it probably still is for most guys that age.
It’s a reminder of how much things change as life goes by.
Here in my little corner of neurology, multiple sclerosis has gone from steroids for relapses, to a few injections of mild benefit, to a bunch of drugs that are, literally, game-changing for many patients. And the Big Four epilepsy drugs (Dilantin, Tegretol, Depakote, and Phenobarb) are slowly fading into the background.
But back to changing priorities – it’s the way life rewrites our plans at each step. From a freshly waxed car to good grades to mortgages to kids – and then watching as they wax their cars.
Suddenly my car looks dusty. Am I the same way? I’m certainly not 16 anymore. Realistically, the majority of my life and career are behind me now. That doesn’t mean I’m not still having fun, it’s just the truth. I try not to think about it that much, as doing so won’t change anything.
In all honesty, neither did washing my car. I mean, the car looked good, but did it make me one of the cool kids? Or get me a girlfriend? Or invited to THE parties? Not at all. Like so many things about appearances, very few of them really matter. There’s only so far that style will get you, compared with substance.
Which doesn’t change the fact that I need to wash my car. But procrastination is for another column.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
When I was 16, keeping my car shiny was a priority. I washed it every weekend and waxed it once a month. I was pretty good at it and got paid to do the occasional job for a neighbor, too.
In college I was busier, and my car was back at the house, so it didn’t need to be washed as much.
In medical school I think I washed the car once a year. Residency was probably the same.
Today I realized I couldn’t remember when I last had it washed (at my age I don’t have time to do it myself). So I looked it up in Quicken: Nov. 14, 2018.
Really? I’ve gone almost 5 years without washing my car? I can’t even blame that on the pandemic.
I mean, I still like my car. It’s comfortable, has good air conditioning (in Phoenix that’s critical), and gets me where I want to go. At my age those things are what’s really important. It’s hard to believe that 40 years ago, keeping a polished car was the center of my existence. Of course, it probably still is for most guys that age.
It’s a reminder of how much things change as life goes by.
Here in my little corner of neurology, multiple sclerosis has gone from steroids for relapses, to a few injections of mild benefit, to a bunch of drugs that are, literally, game-changing for many patients. And the Big Four epilepsy drugs (Dilantin, Tegretol, Depakote, and Phenobarb) are slowly fading into the background.
But back to changing priorities – it’s the way life rewrites our plans at each step. From a freshly waxed car to good grades to mortgages to kids – and then watching as they wax their cars.
Suddenly my car looks dusty. Am I the same way? I’m certainly not 16 anymore. Realistically, the majority of my life and career are behind me now. That doesn’t mean I’m not still having fun, it’s just the truth. I try not to think about it that much, as doing so won’t change anything.
In all honesty, neither did washing my car. I mean, the car looked good, but did it make me one of the cool kids? Or get me a girlfriend? Or invited to THE parties? Not at all. Like so many things about appearances, very few of them really matter. There’s only so far that style will get you, compared with substance.
Which doesn’t change the fact that I need to wash my car. But procrastination is for another column.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
When I was 16, keeping my car shiny was a priority. I washed it every weekend and waxed it once a month. I was pretty good at it and got paid to do the occasional job for a neighbor, too.
In college I was busier, and my car was back at the house, so it didn’t need to be washed as much.
In medical school I think I washed the car once a year. Residency was probably the same.
Today I realized I couldn’t remember when I last had it washed (at my age I don’t have time to do it myself). So I looked it up in Quicken: Nov. 14, 2018.
Really? I’ve gone almost 5 years without washing my car? I can’t even blame that on the pandemic.
I mean, I still like my car. It’s comfortable, has good air conditioning (in Phoenix that’s critical), and gets me where I want to go. At my age those things are what’s really important. It’s hard to believe that 40 years ago, keeping a polished car was the center of my existence. Of course, it probably still is for most guys that age.
It’s a reminder of how much things change as life goes by.
Here in my little corner of neurology, multiple sclerosis has gone from steroids for relapses, to a few injections of mild benefit, to a bunch of drugs that are, literally, game-changing for many patients. And the Big Four epilepsy drugs (Dilantin, Tegretol, Depakote, and Phenobarb) are slowly fading into the background.
But back to changing priorities – it’s the way life rewrites our plans at each step. From a freshly waxed car to good grades to mortgages to kids – and then watching as they wax their cars.
Suddenly my car looks dusty. Am I the same way? I’m certainly not 16 anymore. Realistically, the majority of my life and career are behind me now. That doesn’t mean I’m not still having fun, it’s just the truth. I try not to think about it that much, as doing so won’t change anything.
In all honesty, neither did washing my car. I mean, the car looked good, but did it make me one of the cool kids? Or get me a girlfriend? Or invited to THE parties? Not at all. Like so many things about appearances, very few of them really matter. There’s only so far that style will get you, compared with substance.
Which doesn’t change the fact that I need to wash my car. But procrastination is for another column.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
Probing the link between GERD and anxiety and depression
Anxiety and depression are prevalent in patients with gastroesophageal reflux disease (GERD), according to a large-scale analysis that also found evidence for a possible underlying causal link between GERD and the common psychiatric conditions.
In a systematic review and meta-analysis, researchers found that anxiety and depressive symptoms were more than twice as likely in patients with GERD than in healthy controls. Among individuals with GERD, up to one-third and one-fourth were affected by anxiety and depressive symptoms, respectively.
Moreover, a Mendelian randomization analysis suggested that a genetic risk for GERD increased the risk of developing depression or anxiety and vice versa.
A genetic predisposition seems to underlie GERD and anxiety or depression, said Nicholas J. Talley, MD, PhD, School of Medicine and Public Health, University of Newcastle, Australia, and corresponding author of the review, published online in the American Journal of Gastroenterology.
Scientifically, the possible underlying link “suggests there are different disease subsets within reflux that probably have a different pathogenesis, and these relationships are an expression of whatever the underlying causal pathways are,” Dr. Talley told this news organization.
Clinically, “if you’ve got a patient with reflux, you should be asking one or two questions about anxiety and particularly depression ... because it may impact on how well patients respond to therapy, and how well they do in the longer term,” Dr. Talley said.
A deeper look
In the review, the authors note that GERD affects about 15% of the general population worldwide and has a negative impact on the health-related quality of life.
The pathogenesis is complex and may be associated with psychological distress alongside the well-known predisposing anatomical factors, aggravated by lifestyle factors such as obesity and smoking.
Individual studies examining the association between GERD and the prevalence of anxiety or depressive symptoms have had mixed results, “perhaps limited by heterogeneous study design, the severity of disease included, and diagnostic criteria for depression and anxiety,” the researchers wrote.
They therefore set out to perform a systematic review and meta-analysis to assess the issue in greater depth. They looked at three primary outcomes – the prevalence of symptoms of anxiety and depression in individuals with GERD, the epidemiological risk of developing GERD in people with anxiety or depression and vice versa, and the cause-and-effect relationship between anxiety or depression and GERD.
They searched the Embase, PubMed, Scopus, and Web of Science databases, and found 36 eligible studies published between 2003 and 2023.
Of the total, 30 were observational studies that examined the prevalence of anxiety and/or depressive symptoms using validated questionnaires in ≥ 100 individuals aged 18 years or older.
These included 26 studies of anxiety symptoms among 10,378 individuals with GERD, 30 that looked at depressive symptoms among 14,030 subjects with GERD, and four that reported the prevalence of simultaneous anxiety and depressive symptoms in 3,878 patients with GERD. Some studies were population based and others were hospital based. The extracted data were combined using a random-effects model.
The overall pooled analyses revealed a prevalence of anxiety symptoms of 34.4% among individuals with GERD, at an adjusted odds ratio versus healthy controls of 4.46 (95% confidence interval [CI], 1.94-10.25).
The pooled prevalence of depressive symptoms among those with GERD was 24.2%, at an adjusted odds ratio compared with healthy controls of 2.56 (95% CI, 1.11-5.87).
The pooled prevalence of simultaneous anxiety and depression in people with GERD was 22.3% (95% CI, 7.0-37.7).
Next, the team added three cohort studies that explored the risk for GERD in individuals with anxiety or depression in the vice versa scenario, as well as three Mendelian randomization studies that assessed the cause-and-effect relationship between anxiety or depression and GERD.
The cohort studies could not be pooled, but individuals with depression had a significantly increased risk for GERD versus those without depression, at an adjusted odds ratio in one study of 2.01 (95% CI, 1.96-2.07) and an adjusted hazard ratio in another study of 1.72 (95% CI, 1.60-1.85). A similar pattern was seen for anxiety.
The Mendelian randomization studies suggested that a genetic risk for depression was associated with an increased risk of developing GERD, at an odds ratio of 1.36 (95% CI, 1.21-1.51), and that a genetic risk for GERD was linked to an increased risk of developing depression, at an odds ratio of 1.30 (95% CI, 1.17-1.43).
One study reported a similar effect of a genetic risk for GERD on the risk for anxiety.
“We don’t really know what causes reflux disease. We know the pathophysiology, but we don’t really know what the etiology is, and depression is the same,” Dr. Talley noted. “It’s obviously thought to be a brain disease, but sometimes it might be coming from the gut as well, for all we know, and there is certainly evidence for that.”
The ‘chicken and egg’ question
Experts welcomed the findings, saying they supported their observations and validated the idea of looking outside the gut for complicating factors in patients with GERD, but some questioned the clinical relevance of the Mendelian randomization.
Approached for comment, Amir E. Masoud, MD, medical codirector, Hartford HealthCare Neurogastroenterology and Motility Center, Fairfield, Conn., said the study’s most important contribution is increased recognition of the connection between GERD and anxiety or depression.
“There is a psychological component” to GERD, he said, and it is an “important area for investigation and possible intervention for patients who don’t respond to traditional therapies.”
Dr. Masoud singled out the study design. The researchers “tried to tee this up with something we’ve always thought about,” he told this news organization, referring to the “chicken and egg” question about the relationship between GERD and anxiety or depressive symptoms.
As such, it provides evidence for a common observation in the clinic, he said. “A lot of times, when a therapy that should work does not work for something like GERD, for example, we tend to think of psychological factors that could be playing a role.”
No longer a taboo topic
Rena Yadlapati, MD, medical director, Center for Esophageal Diseases, UC San Diego Health, told this news organization that the study is “validating, in terms of the conversations with patients and some of the nontraditional methods we are trying to explore in their management.”
She noted the bidirectional relationship revealed by the Mendelian randomization, but cautioned that the approach relied on a small number of studies and hasn’t been utilized in gastroenterology as much as in other fields.
“We need to understand that this is hypothesis generating, and there are probably some important limitations,” she added, even if the results “certainly make sense.”
From a clinical perspective, the idea of anxiety or depression being linked to GERD has been a “taboo topic” for a long time, Dr. Yadlapati said.
“But if we can effectively communicate that relationship between the brain and gut and this whole cyclical process [to patients], there’s power to gain patient insight and engagement.”
Another key aspect is to consider a psychological referral for patients, as well as engaging them with “simple things like relaxation strategies and diaphragmatic breathing and referring them to people who can do cognitive behavioral therapy.”
Dr. Yadlapati emphasized that “it doesn’t necessarily have to be a psychologist. There are a lot of other ways to provide that access to patients.”
Practical clinical implications
Philip O. Katz, MD, director of motility laboratories, division of gastroenterology, Weill Cornell Medicine, New York, said that the study, like many of its kind, reinforces that there are multiple cofactors associated with managing patients with reflux disease.
As for the bidirectional relationship identified through the Mendelian randomization, he was skeptical about its clinical value and pointed out that the odds ratios are “relatively small.”
“It is commonplace for people to look for genetic predispositions to a lot of disease,” Katz told Medscape Medical News, but, “in clinical practice, I don’t believe that it’s particularly meaningful, to be honest.”
However, there is no doubt that GERD symptoms are augmented by times of what is generically called “stress,” he added.
“Regardless of those findings, it doesn’t change the way I view the importance of this article, which is when someone’s not responding to medication to look for reasons other than the original diagnosis,” Dr. Katz said.
“There is so much overlap between true GERD and symptoms that sound like GERD” that clinicians need “to be aware that both anxiety and depression are cofactors,” he said.
Dr. Talley is supported by funding from the National Health and Medical Research Council (NHMRC) to the Centre of Research Excellence in Digestive Health, and he holds an NHMRC Investigator grant. He declared relationships with Norgine, Bristol Myers Squibb, Allakos, Bayer, Planet Innovation, twoXAR, Viscera Labs, Dr Falk Pharma, Sanofi, Glutagen, ISOVive, BluMaiden, Rose Pharma, Intrinsic Medicine, Comvita Manuka Honey, GlaxoSmithKline Australia, and AstraZeneca; and holds numerous patents. Dr. Masoud declared no relevant relationships. Dr. Yadlapati declared relationships with Medtronic, Phathom Pharmaceuticals, Ironwood Pharmaceuticals, and RJS Mediagnostix. Dr. Katz declared relationships with Phathom Pharma, Sebella, and Syneos.
A version of this article first appeared on Medscape.com.
Anxiety and depression are prevalent in patients with gastroesophageal reflux disease (GERD), according to a large-scale analysis that also found evidence for a possible underlying causal link between GERD and the common psychiatric conditions.
In a systematic review and meta-analysis, researchers found that anxiety and depressive symptoms were more than twice as likely in patients with GERD than in healthy controls. Among individuals with GERD, up to one-third and one-fourth were affected by anxiety and depressive symptoms, respectively.
Moreover, a Mendelian randomization analysis suggested that a genetic risk for GERD increased the risk of developing depression or anxiety and vice versa.
A genetic predisposition seems to underlie GERD and anxiety or depression, said Nicholas J. Talley, MD, PhD, School of Medicine and Public Health, University of Newcastle, Australia, and corresponding author of the review, published online in the American Journal of Gastroenterology.
Scientifically, the possible underlying link “suggests there are different disease subsets within reflux that probably have a different pathogenesis, and these relationships are an expression of whatever the underlying causal pathways are,” Dr. Talley told this news organization.
Clinically, “if you’ve got a patient with reflux, you should be asking one or two questions about anxiety and particularly depression ... because it may impact on how well patients respond to therapy, and how well they do in the longer term,” Dr. Talley said.
A deeper look
In the review, the authors note that GERD affects about 15% of the general population worldwide and has a negative impact on the health-related quality of life.
The pathogenesis is complex and may be associated with psychological distress alongside the well-known predisposing anatomical factors, aggravated by lifestyle factors such as obesity and smoking.
Individual studies examining the association between GERD and the prevalence of anxiety or depressive symptoms have had mixed results, “perhaps limited by heterogeneous study design, the severity of disease included, and diagnostic criteria for depression and anxiety,” the researchers wrote.
They therefore set out to perform a systematic review and meta-analysis to assess the issue in greater depth. They looked at three primary outcomes – the prevalence of symptoms of anxiety and depression in individuals with GERD, the epidemiological risk of developing GERD in people with anxiety or depression and vice versa, and the cause-and-effect relationship between anxiety or depression and GERD.
They searched the Embase, PubMed, Scopus, and Web of Science databases, and found 36 eligible studies published between 2003 and 2023.
Of the total, 30 were observational studies that examined the prevalence of anxiety and/or depressive symptoms using validated questionnaires in ≥ 100 individuals aged 18 years or older.
These included 26 studies of anxiety symptoms among 10,378 individuals with GERD, 30 that looked at depressive symptoms among 14,030 subjects with GERD, and four that reported the prevalence of simultaneous anxiety and depressive symptoms in 3,878 patients with GERD. Some studies were population based and others were hospital based. The extracted data were combined using a random-effects model.
The overall pooled analyses revealed a prevalence of anxiety symptoms of 34.4% among individuals with GERD, at an adjusted odds ratio versus healthy controls of 4.46 (95% confidence interval [CI], 1.94-10.25).
The pooled prevalence of depressive symptoms among those with GERD was 24.2%, at an adjusted odds ratio compared with healthy controls of 2.56 (95% CI, 1.11-5.87).
The pooled prevalence of simultaneous anxiety and depression in people with GERD was 22.3% (95% CI, 7.0-37.7).
Next, the team added three cohort studies that explored the risk for GERD in individuals with anxiety or depression in the vice versa scenario, as well as three Mendelian randomization studies that assessed the cause-and-effect relationship between anxiety or depression and GERD.
The cohort studies could not be pooled, but individuals with depression had a significantly increased risk for GERD versus those without depression, at an adjusted odds ratio in one study of 2.01 (95% CI, 1.96-2.07) and an adjusted hazard ratio in another study of 1.72 (95% CI, 1.60-1.85). A similar pattern was seen for anxiety.
The Mendelian randomization studies suggested that a genetic risk for depression was associated with an increased risk of developing GERD, at an odds ratio of 1.36 (95% CI, 1.21-1.51), and that a genetic risk for GERD was linked to an increased risk of developing depression, at an odds ratio of 1.30 (95% CI, 1.17-1.43).
One study reported a similar effect of a genetic risk for GERD on the risk for anxiety.
“We don’t really know what causes reflux disease. We know the pathophysiology, but we don’t really know what the etiology is, and depression is the same,” Dr. Talley noted. “It’s obviously thought to be a brain disease, but sometimes it might be coming from the gut as well, for all we know, and there is certainly evidence for that.”
The ‘chicken and egg’ question
Experts welcomed the findings, saying they supported their observations and validated the idea of looking outside the gut for complicating factors in patients with GERD, but some questioned the clinical relevance of the Mendelian randomization.
Approached for comment, Amir E. Masoud, MD, medical codirector, Hartford HealthCare Neurogastroenterology and Motility Center, Fairfield, Conn., said the study’s most important contribution is increased recognition of the connection between GERD and anxiety or depression.
“There is a psychological component” to GERD, he said, and it is an “important area for investigation and possible intervention for patients who don’t respond to traditional therapies.”
Dr. Masoud singled out the study design. The researchers “tried to tee this up with something we’ve always thought about,” he told this news organization, referring to the “chicken and egg” question about the relationship between GERD and anxiety or depressive symptoms.
As such, it provides evidence for a common observation in the clinic, he said. “A lot of times, when a therapy that should work does not work for something like GERD, for example, we tend to think of psychological factors that could be playing a role.”
No longer a taboo topic
Rena Yadlapati, MD, medical director, Center for Esophageal Diseases, UC San Diego Health, told this news organization that the study is “validating, in terms of the conversations with patients and some of the nontraditional methods we are trying to explore in their management.”
She noted the bidirectional relationship revealed by the Mendelian randomization, but cautioned that the approach relied on a small number of studies and hasn’t been utilized in gastroenterology as much as in other fields.
“We need to understand that this is hypothesis generating, and there are probably some important limitations,” she added, even if the results “certainly make sense.”
From a clinical perspective, the idea of anxiety or depression being linked to GERD has been a “taboo topic” for a long time, Dr. Yadlapati said.
“But if we can effectively communicate that relationship between the brain and gut and this whole cyclical process [to patients], there’s power to gain patient insight and engagement.”
Another key aspect is to consider a psychological referral for patients, as well as engaging them with “simple things like relaxation strategies and diaphragmatic breathing and referring them to people who can do cognitive behavioral therapy.”
Dr. Yadlapati emphasized that “it doesn’t necessarily have to be a psychologist. There are a lot of other ways to provide that access to patients.”
Practical clinical implications
Philip O. Katz, MD, director of motility laboratories, division of gastroenterology, Weill Cornell Medicine, New York, said that the study, like many of its kind, reinforces that there are multiple cofactors associated with managing patients with reflux disease.
As for the bidirectional relationship identified through the Mendelian randomization, he was skeptical about its clinical value and pointed out that the odds ratios are “relatively small.”
“It is commonplace for people to look for genetic predispositions to a lot of disease,” Katz told Medscape Medical News, but, “in clinical practice, I don’t believe that it’s particularly meaningful, to be honest.”
However, there is no doubt that GERD symptoms are augmented by times of what is generically called “stress,” he added.
“Regardless of those findings, it doesn’t change the way I view the importance of this article, which is when someone’s not responding to medication to look for reasons other than the original diagnosis,” Dr. Katz said.
“There is so much overlap between true GERD and symptoms that sound like GERD” that clinicians need “to be aware that both anxiety and depression are cofactors,” he said.
Dr. Talley is supported by funding from the National Health and Medical Research Council (NHMRC) to the Centre of Research Excellence in Digestive Health, and he holds an NHMRC Investigator grant. He declared relationships with Norgine, Bristol Myers Squibb, Allakos, Bayer, Planet Innovation, twoXAR, Viscera Labs, Dr Falk Pharma, Sanofi, Glutagen, ISOVive, BluMaiden, Rose Pharma, Intrinsic Medicine, Comvita Manuka Honey, GlaxoSmithKline Australia, and AstraZeneca; and holds numerous patents. Dr. Masoud declared no relevant relationships. Dr. Yadlapati declared relationships with Medtronic, Phathom Pharmaceuticals, Ironwood Pharmaceuticals, and RJS Mediagnostix. Dr. Katz declared relationships with Phathom Pharma, Sebella, and Syneos.
A version of this article first appeared on Medscape.com.
Anxiety and depression are prevalent in patients with gastroesophageal reflux disease (GERD), according to a large-scale analysis that also found evidence for a possible underlying causal link between GERD and the common psychiatric conditions.
In a systematic review and meta-analysis, researchers found that anxiety and depressive symptoms were more than twice as likely in patients with GERD than in healthy controls. Among individuals with GERD, up to one-third and one-fourth were affected by anxiety and depressive symptoms, respectively.
Moreover, a Mendelian randomization analysis suggested that a genetic risk for GERD increased the risk of developing depression or anxiety and vice versa.
A genetic predisposition seems to underlie GERD and anxiety or depression, said Nicholas J. Talley, MD, PhD, School of Medicine and Public Health, University of Newcastle, Australia, and corresponding author of the review, published online in the American Journal of Gastroenterology.
Scientifically, the possible underlying link “suggests there are different disease subsets within reflux that probably have a different pathogenesis, and these relationships are an expression of whatever the underlying causal pathways are,” Dr. Talley told this news organization.
Clinically, “if you’ve got a patient with reflux, you should be asking one or two questions about anxiety and particularly depression ... because it may impact on how well patients respond to therapy, and how well they do in the longer term,” Dr. Talley said.
A deeper look
In the review, the authors note that GERD affects about 15% of the general population worldwide and has a negative impact on the health-related quality of life.
The pathogenesis is complex and may be associated with psychological distress alongside the well-known predisposing anatomical factors, aggravated by lifestyle factors such as obesity and smoking.
Individual studies examining the association between GERD and the prevalence of anxiety or depressive symptoms have had mixed results, “perhaps limited by heterogeneous study design, the severity of disease included, and diagnostic criteria for depression and anxiety,” the researchers wrote.
They therefore set out to perform a systematic review and meta-analysis to assess the issue in greater depth. They looked at three primary outcomes – the prevalence of symptoms of anxiety and depression in individuals with GERD, the epidemiological risk of developing GERD in people with anxiety or depression and vice versa, and the cause-and-effect relationship between anxiety or depression and GERD.
They searched the Embase, PubMed, Scopus, and Web of Science databases, and found 36 eligible studies published between 2003 and 2023.
Of the total, 30 were observational studies that examined the prevalence of anxiety and/or depressive symptoms using validated questionnaires in ≥ 100 individuals aged 18 years or older.
These included 26 studies of anxiety symptoms among 10,378 individuals with GERD, 30 that looked at depressive symptoms among 14,030 subjects with GERD, and four that reported the prevalence of simultaneous anxiety and depressive symptoms in 3,878 patients with GERD. Some studies were population based and others were hospital based. The extracted data were combined using a random-effects model.
The overall pooled analyses revealed a prevalence of anxiety symptoms of 34.4% among individuals with GERD, at an adjusted odds ratio versus healthy controls of 4.46 (95% confidence interval [CI], 1.94-10.25).
The pooled prevalence of depressive symptoms among those with GERD was 24.2%, at an adjusted odds ratio compared with healthy controls of 2.56 (95% CI, 1.11-5.87).
The pooled prevalence of simultaneous anxiety and depression in people with GERD was 22.3% (95% CI, 7.0-37.7).
Next, the team added three cohort studies that explored the risk for GERD in individuals with anxiety or depression in the vice versa scenario, as well as three Mendelian randomization studies that assessed the cause-and-effect relationship between anxiety or depression and GERD.
The cohort studies could not be pooled, but individuals with depression had a significantly increased risk for GERD versus those without depression, at an adjusted odds ratio in one study of 2.01 (95% CI, 1.96-2.07) and an adjusted hazard ratio in another study of 1.72 (95% CI, 1.60-1.85). A similar pattern was seen for anxiety.
The Mendelian randomization studies suggested that a genetic risk for depression was associated with an increased risk of developing GERD, at an odds ratio of 1.36 (95% CI, 1.21-1.51), and that a genetic risk for GERD was linked to an increased risk of developing depression, at an odds ratio of 1.30 (95% CI, 1.17-1.43).
One study reported a similar effect of a genetic risk for GERD on the risk for anxiety.
“We don’t really know what causes reflux disease. We know the pathophysiology, but we don’t really know what the etiology is, and depression is the same,” Dr. Talley noted. “It’s obviously thought to be a brain disease, but sometimes it might be coming from the gut as well, for all we know, and there is certainly evidence for that.”
The ‘chicken and egg’ question
Experts welcomed the findings, saying they supported their observations and validated the idea of looking outside the gut for complicating factors in patients with GERD, but some questioned the clinical relevance of the Mendelian randomization.
Approached for comment, Amir E. Masoud, MD, medical codirector, Hartford HealthCare Neurogastroenterology and Motility Center, Fairfield, Conn., said the study’s most important contribution is increased recognition of the connection between GERD and anxiety or depression.
“There is a psychological component” to GERD, he said, and it is an “important area for investigation and possible intervention for patients who don’t respond to traditional therapies.”
Dr. Masoud singled out the study design. The researchers “tried to tee this up with something we’ve always thought about,” he told this news organization, referring to the “chicken and egg” question about the relationship between GERD and anxiety or depressive symptoms.
As such, it provides evidence for a common observation in the clinic, he said. “A lot of times, when a therapy that should work does not work for something like GERD, for example, we tend to think of psychological factors that could be playing a role.”
No longer a taboo topic
Rena Yadlapati, MD, medical director, Center for Esophageal Diseases, UC San Diego Health, told this news organization that the study is “validating, in terms of the conversations with patients and some of the nontraditional methods we are trying to explore in their management.”
She noted the bidirectional relationship revealed by the Mendelian randomization, but cautioned that the approach relied on a small number of studies and hasn’t been utilized in gastroenterology as much as in other fields.
“We need to understand that this is hypothesis generating, and there are probably some important limitations,” she added, even if the results “certainly make sense.”
From a clinical perspective, the idea of anxiety or depression being linked to GERD has been a “taboo topic” for a long time, Dr. Yadlapati said.
“But if we can effectively communicate that relationship between the brain and gut and this whole cyclical process [to patients], there’s power to gain patient insight and engagement.”
Another key aspect is to consider a psychological referral for patients, as well as engaging them with “simple things like relaxation strategies and diaphragmatic breathing and referring them to people who can do cognitive behavioral therapy.”
Dr. Yadlapati emphasized that “it doesn’t necessarily have to be a psychologist. There are a lot of other ways to provide that access to patients.”
Practical clinical implications
Philip O. Katz, MD, director of motility laboratories, division of gastroenterology, Weill Cornell Medicine, New York, said that the study, like many of its kind, reinforces that there are multiple cofactors associated with managing patients with reflux disease.
As for the bidirectional relationship identified through the Mendelian randomization, he was skeptical about its clinical value and pointed out that the odds ratios are “relatively small.”
“It is commonplace for people to look for genetic predispositions to a lot of disease,” Katz told Medscape Medical News, but, “in clinical practice, I don’t believe that it’s particularly meaningful, to be honest.”
However, there is no doubt that GERD symptoms are augmented by times of what is generically called “stress,” he added.
“Regardless of those findings, it doesn’t change the way I view the importance of this article, which is when someone’s not responding to medication to look for reasons other than the original diagnosis,” Dr. Katz said.
“There is so much overlap between true GERD and symptoms that sound like GERD” that clinicians need “to be aware that both anxiety and depression are cofactors,” he said.
Dr. Talley is supported by funding from the National Health and Medical Research Council (NHMRC) to the Centre of Research Excellence in Digestive Health, and he holds an NHMRC Investigator grant. He declared relationships with Norgine, Bristol Myers Squibb, Allakos, Bayer, Planet Innovation, twoXAR, Viscera Labs, Dr Falk Pharma, Sanofi, Glutagen, ISOVive, BluMaiden, Rose Pharma, Intrinsic Medicine, Comvita Manuka Honey, GlaxoSmithKline Australia, and AstraZeneca; and holds numerous patents. Dr. Masoud declared no relevant relationships. Dr. Yadlapati declared relationships with Medtronic, Phathom Pharmaceuticals, Ironwood Pharmaceuticals, and RJS Mediagnostix. Dr. Katz declared relationships with Phathom Pharma, Sebella, and Syneos.
A version of this article first appeared on Medscape.com.
Comorbidities, CV risk factors common in early PsA
TOPLINE:
Patients with early psoriatic arthritis (PsA) were significantly more likely to have multiple comorbidities and cardiovascular risk factors than controls.
METHODOLOGY:
- The study population included 67 adults with early PsA and 61 healthy matched controls with mean ages of 47.9 years and 45 years, respectively.
- Early PsA was defined as symptom duration of less than 2 years; patients with conditions including active infection, malignancy, or other rheumatic or systemic disease were excluded.
- The researchers examined the prevalence of comorbidities and cardiovascular risk factors in treatment-naive, newly diagnosed patients with PsA at baseline and after 1 year.
TAKEAWAY:
- , compared with healthy controls (odds ratios, 1.9 and 2.1, respectively).
- Dyslipidemia was the most prevalent comorbidity among patients with PsA and was more prevalent than in controls (64.2% vs. 39.3%; OR, 1.7).
- Obesity was more common in patients with PsA, compared with controls (40.3% vs. 18.3%, respectively), and more patients with PsA had cardiovascular disease at baseline than did controls (20.9% vs. 6.6%; OR, 3.2).
- Disease activity scores improved after 1 year, but the proportion of patients with comorbidities and CV risk factors remained stable.
IN PRACTICE:
The results support the early assessment of patients with PsA for comorbidities to inform treatment and suggest that comorbidities and CV risk factors are more than a consequence of long-term PsA and chronic systemic inflammation.
SOURCE:
The study was conducted by Alla Ishchenko, MD, and colleagues in the division of rheumatology at University Hospitals Leuven, Belgium. The study was published online in Arthritis Care & Research.
LIMITATIONS:
The study was exploratory in nature, with a short follow-up period and a relatively small sample size.
DISCLOSURES:
Dr. Ishchenko disclosed support from PARTNER, an international fellowship program to study disease mechanisms in psoriatic arthritis, as well as grants from Lilly and from the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis.
A version of this article first appeared on Medscape.com.
TOPLINE:
Patients with early psoriatic arthritis (PsA) were significantly more likely to have multiple comorbidities and cardiovascular risk factors than controls.
METHODOLOGY:
- The study population included 67 adults with early PsA and 61 healthy matched controls with mean ages of 47.9 years and 45 years, respectively.
- Early PsA was defined as symptom duration of less than 2 years; patients with conditions including active infection, malignancy, or other rheumatic or systemic disease were excluded.
- The researchers examined the prevalence of comorbidities and cardiovascular risk factors in treatment-naive, newly diagnosed patients with PsA at baseline and after 1 year.
TAKEAWAY:
- , compared with healthy controls (odds ratios, 1.9 and 2.1, respectively).
- Dyslipidemia was the most prevalent comorbidity among patients with PsA and was more prevalent than in controls (64.2% vs. 39.3%; OR, 1.7).
- Obesity was more common in patients with PsA, compared with controls (40.3% vs. 18.3%, respectively), and more patients with PsA had cardiovascular disease at baseline than did controls (20.9% vs. 6.6%; OR, 3.2).
- Disease activity scores improved after 1 year, but the proportion of patients with comorbidities and CV risk factors remained stable.
IN PRACTICE:
The results support the early assessment of patients with PsA for comorbidities to inform treatment and suggest that comorbidities and CV risk factors are more than a consequence of long-term PsA and chronic systemic inflammation.
SOURCE:
The study was conducted by Alla Ishchenko, MD, and colleagues in the division of rheumatology at University Hospitals Leuven, Belgium. The study was published online in Arthritis Care & Research.
LIMITATIONS:
The study was exploratory in nature, with a short follow-up period and a relatively small sample size.
DISCLOSURES:
Dr. Ishchenko disclosed support from PARTNER, an international fellowship program to study disease mechanisms in psoriatic arthritis, as well as grants from Lilly and from the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis.
A version of this article first appeared on Medscape.com.
TOPLINE:
Patients with early psoriatic arthritis (PsA) were significantly more likely to have multiple comorbidities and cardiovascular risk factors than controls.
METHODOLOGY:
- The study population included 67 adults with early PsA and 61 healthy matched controls with mean ages of 47.9 years and 45 years, respectively.
- Early PsA was defined as symptom duration of less than 2 years; patients with conditions including active infection, malignancy, or other rheumatic or systemic disease were excluded.
- The researchers examined the prevalence of comorbidities and cardiovascular risk factors in treatment-naive, newly diagnosed patients with PsA at baseline and after 1 year.
TAKEAWAY:
- , compared with healthy controls (odds ratios, 1.9 and 2.1, respectively).
- Dyslipidemia was the most prevalent comorbidity among patients with PsA and was more prevalent than in controls (64.2% vs. 39.3%; OR, 1.7).
- Obesity was more common in patients with PsA, compared with controls (40.3% vs. 18.3%, respectively), and more patients with PsA had cardiovascular disease at baseline than did controls (20.9% vs. 6.6%; OR, 3.2).
- Disease activity scores improved after 1 year, but the proportion of patients with comorbidities and CV risk factors remained stable.
IN PRACTICE:
The results support the early assessment of patients with PsA for comorbidities to inform treatment and suggest that comorbidities and CV risk factors are more than a consequence of long-term PsA and chronic systemic inflammation.
SOURCE:
The study was conducted by Alla Ishchenko, MD, and colleagues in the division of rheumatology at University Hospitals Leuven, Belgium. The study was published online in Arthritis Care & Research.
LIMITATIONS:
The study was exploratory in nature, with a short follow-up period and a relatively small sample size.
DISCLOSURES:
Dr. Ishchenko disclosed support from PARTNER, an international fellowship program to study disease mechanisms in psoriatic arthritis, as well as grants from Lilly and from the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis.
A version of this article first appeared on Medscape.com.
Should people who play sports pay higher medical insurance premiums?
This transcript has been edited for clarity.
If you’re anywhere near Seattle, anywhere near Florida, or anywhere where it might be not oppressively hot outside but encouraging some people who might want to go out and get a little exercise, you’ve undoubtedly seen or heard of pickleball.
This took off, I think, out of Bainbridge Island, Wash. It was meant as a gentlemanly game where people didn’t exert themselves too much. The joke is you could play it while holding a drink in one hand. It’s gotten more popular and more competitive. It’s kind of a miniature version of tennis, with a smaller court, a plastic ball, and a wooden paddle. The ball can go back and forth rapidly, but you’re always playing doubles and it doesn’t take as much energy, exertion, and, if you will, fitness as a game like singles tennis.
The upside is it’s gotten many people outdoors getting some exercise and socializing. That’s all to the good. But a recent study suggested that there are about $500 million worth of injuries coming into the health care system associated with pickleball. There have been leg sprains, broken bones, people getting hit in the eye, hamstring pulls, and many other problems. I’ve been told that many of the spectators who show up for pickleball matches are there with a cast or have some kind of a wrap on because they were injured.
Well, many people have argued in the past about what we are going to do about health care costs. Some suggest if you voluntarily incur health care damage, you ought to pay for that yourself and you ought to have a big copay.
If you decide you’re going to do cross-country skiing or downhill skiing and you injure yourself, you chose to do it, so you pay. If you’re not going to maintain your weight, you’re going to smoke, or you’re going to ride around without a helmet, that’s your choice. You ought to pay.
I think the pickleball example is really a good challenge to these views. You obviously want people to go out and get some exercise. Here, we’re talking about a population that’s a little older and oftentimes doesn’t get out there as much as doctors would like to get the exercise that’s still important that they need, and yet it does incur injuries and problems.
My suggestion would be to make the game a little safer. Let’s try to encourage people to warm up more before they get out there and jump out of the car and engage in their pickleball battles. Goggles might be important to prevent the eye injuries in a game that’s played up close. Maybe we want to make sure that people look out for one another out there. If they think they’re getting dehydrated or tired, they should say, “Let’s sit down.”
I’m not willing to put a tax or a copay on the pickleball players of America. I know they choose to do it. It’s got an upside and benefits, as many things like skiing and other behaviors that have some risk do, but I think we want to be encouraging, not discouraging, of it.
I don’t like a society where anybody who tries to do something that takes risk winds up bearing extra cost for doing that. I understand that that gets people irritated when it comes to dangerous, hyper-risky behavior like smoking and not wearing a motorcycle helmet. I think the way to engage is not to call out the sinner or to try and punish those who are trying to do things that bring them enjoyment, reward, or in some of these cases, physical fitness, but to try to make things safer and try to gradually improve and get rid of the risk side to capture the full benefit side.
I’m not sure I’ve come up with all the best ways to make pickleball safer, but I think that’s where our thinking in health care should go. My view is to get out there and play pickleball. If you do pull your hamstring, raise my insurance premium a little bit. I’ll help to pay for it. Better you get some enjoyment and some exercise.
I get the downside, but come on, folks, we ought to be, as a community, somewhat supportive of the fun and recreation that our fellow citizens engage in.
Dr. Caplan is director, division of medical ethics, New York University Langone Medical Center. He disclosed serving as a director, officer, partner, employee, adviser, consultant, or trustee for Johnson & Johnson’s Panel for Compassionate Drug Use (unpaid position); and as a contributing author and adviser for Medscape.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
If you’re anywhere near Seattle, anywhere near Florida, or anywhere where it might be not oppressively hot outside but encouraging some people who might want to go out and get a little exercise, you’ve undoubtedly seen or heard of pickleball.
This took off, I think, out of Bainbridge Island, Wash. It was meant as a gentlemanly game where people didn’t exert themselves too much. The joke is you could play it while holding a drink in one hand. It’s gotten more popular and more competitive. It’s kind of a miniature version of tennis, with a smaller court, a plastic ball, and a wooden paddle. The ball can go back and forth rapidly, but you’re always playing doubles and it doesn’t take as much energy, exertion, and, if you will, fitness as a game like singles tennis.
The upside is it’s gotten many people outdoors getting some exercise and socializing. That’s all to the good. But a recent study suggested that there are about $500 million worth of injuries coming into the health care system associated with pickleball. There have been leg sprains, broken bones, people getting hit in the eye, hamstring pulls, and many other problems. I’ve been told that many of the spectators who show up for pickleball matches are there with a cast or have some kind of a wrap on because they were injured.
Well, many people have argued in the past about what we are going to do about health care costs. Some suggest if you voluntarily incur health care damage, you ought to pay for that yourself and you ought to have a big copay.
If you decide you’re going to do cross-country skiing or downhill skiing and you injure yourself, you chose to do it, so you pay. If you’re not going to maintain your weight, you’re going to smoke, or you’re going to ride around without a helmet, that’s your choice. You ought to pay.
I think the pickleball example is really a good challenge to these views. You obviously want people to go out and get some exercise. Here, we’re talking about a population that’s a little older and oftentimes doesn’t get out there as much as doctors would like to get the exercise that’s still important that they need, and yet it does incur injuries and problems.
My suggestion would be to make the game a little safer. Let’s try to encourage people to warm up more before they get out there and jump out of the car and engage in their pickleball battles. Goggles might be important to prevent the eye injuries in a game that’s played up close. Maybe we want to make sure that people look out for one another out there. If they think they’re getting dehydrated or tired, they should say, “Let’s sit down.”
I’m not willing to put a tax or a copay on the pickleball players of America. I know they choose to do it. It’s got an upside and benefits, as many things like skiing and other behaviors that have some risk do, but I think we want to be encouraging, not discouraging, of it.
I don’t like a society where anybody who tries to do something that takes risk winds up bearing extra cost for doing that. I understand that that gets people irritated when it comes to dangerous, hyper-risky behavior like smoking and not wearing a motorcycle helmet. I think the way to engage is not to call out the sinner or to try and punish those who are trying to do things that bring them enjoyment, reward, or in some of these cases, physical fitness, but to try to make things safer and try to gradually improve and get rid of the risk side to capture the full benefit side.
I’m not sure I’ve come up with all the best ways to make pickleball safer, but I think that’s where our thinking in health care should go. My view is to get out there and play pickleball. If you do pull your hamstring, raise my insurance premium a little bit. I’ll help to pay for it. Better you get some enjoyment and some exercise.
I get the downside, but come on, folks, we ought to be, as a community, somewhat supportive of the fun and recreation that our fellow citizens engage in.
Dr. Caplan is director, division of medical ethics, New York University Langone Medical Center. He disclosed serving as a director, officer, partner, employee, adviser, consultant, or trustee for Johnson & Johnson’s Panel for Compassionate Drug Use (unpaid position); and as a contributing author and adviser for Medscape.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
If you’re anywhere near Seattle, anywhere near Florida, or anywhere where it might be not oppressively hot outside but encouraging some people who might want to go out and get a little exercise, you’ve undoubtedly seen or heard of pickleball.
This took off, I think, out of Bainbridge Island, Wash. It was meant as a gentlemanly game where people didn’t exert themselves too much. The joke is you could play it while holding a drink in one hand. It’s gotten more popular and more competitive. It’s kind of a miniature version of tennis, with a smaller court, a plastic ball, and a wooden paddle. The ball can go back and forth rapidly, but you’re always playing doubles and it doesn’t take as much energy, exertion, and, if you will, fitness as a game like singles tennis.
The upside is it’s gotten many people outdoors getting some exercise and socializing. That’s all to the good. But a recent study suggested that there are about $500 million worth of injuries coming into the health care system associated with pickleball. There have been leg sprains, broken bones, people getting hit in the eye, hamstring pulls, and many other problems. I’ve been told that many of the spectators who show up for pickleball matches are there with a cast or have some kind of a wrap on because they were injured.
Well, many people have argued in the past about what we are going to do about health care costs. Some suggest if you voluntarily incur health care damage, you ought to pay for that yourself and you ought to have a big copay.
If you decide you’re going to do cross-country skiing or downhill skiing and you injure yourself, you chose to do it, so you pay. If you’re not going to maintain your weight, you’re going to smoke, or you’re going to ride around without a helmet, that’s your choice. You ought to pay.
I think the pickleball example is really a good challenge to these views. You obviously want people to go out and get some exercise. Here, we’re talking about a population that’s a little older and oftentimes doesn’t get out there as much as doctors would like to get the exercise that’s still important that they need, and yet it does incur injuries and problems.
My suggestion would be to make the game a little safer. Let’s try to encourage people to warm up more before they get out there and jump out of the car and engage in their pickleball battles. Goggles might be important to prevent the eye injuries in a game that’s played up close. Maybe we want to make sure that people look out for one another out there. If they think they’re getting dehydrated or tired, they should say, “Let’s sit down.”
I’m not willing to put a tax or a copay on the pickleball players of America. I know they choose to do it. It’s got an upside and benefits, as many things like skiing and other behaviors that have some risk do, but I think we want to be encouraging, not discouraging, of it.
I don’t like a society where anybody who tries to do something that takes risk winds up bearing extra cost for doing that. I understand that that gets people irritated when it comes to dangerous, hyper-risky behavior like smoking and not wearing a motorcycle helmet. I think the way to engage is not to call out the sinner or to try and punish those who are trying to do things that bring them enjoyment, reward, or in some of these cases, physical fitness, but to try to make things safer and try to gradually improve and get rid of the risk side to capture the full benefit side.
I’m not sure I’ve come up with all the best ways to make pickleball safer, but I think that’s where our thinking in health care should go. My view is to get out there and play pickleball. If you do pull your hamstring, raise my insurance premium a little bit. I’ll help to pay for it. Better you get some enjoyment and some exercise.
I get the downside, but come on, folks, we ought to be, as a community, somewhat supportive of the fun and recreation that our fellow citizens engage in.
Dr. Caplan is director, division of medical ethics, New York University Langone Medical Center. He disclosed serving as a director, officer, partner, employee, adviser, consultant, or trustee for Johnson & Johnson’s Panel for Compassionate Drug Use (unpaid position); and as a contributing author and adviser for Medscape.
A version of this article appeared on Medscape.com.
Colchicine still needed to prevent gout flares in allopurinol ‘start-low, go-slow’ strategy
The use of colchicine prophylaxis during the process of up-titrating allopurinol slowly to reach target serum urate levels for patients with gout proved necessary to reduce the risk of flares in a randomized, double-blind, placebo-controlled noninferiority trial conducted by researchers in New Zealand.
Use of colchicine led to fewer flares during the first 6 months after initiating treatment with allopurinol, but when colchicine and placebo were stopped at 6 months, the number of flares rose, and there was no overall difference in the average number of flares between the placebo and colchicine groups over 12 months.
“Gout flares are common when starting urate-lowering therapy, although may be less frequent using the newer allopurinol ‘start-low, go-slow’ dose escalation strategy,” Lisa Stamp, MBChB, PhD, of the University of Otago, Christchurch, New Zealand, and colleagues write in Annals of the Rheumatic Diseases. “This study was undertaken to determine whether colchicine prophylaxis is required with the more gradual dose escalation of allopurinol to achieve target serum urate.”
The study was published online in the BMJ.
The researchers randomly assigned 200 participants to receive colchicine 0.5 mg daily or placebo. At the same time, allopurinol 50 mg daily was initially given to those with an estimated glomerular filtration rate (eGFR) less than 60 mL/min/1.73 m2, and 100 mg daily was given to those with eGFR greater than or equal to 60 mL/min/1.73 m2; doses were increased monthly by 50 mg daily for those with eGFR less than 60 and by 100 mg daily for those with eGFR greater than or equal to 60 until the target urate level of less than 0.36 mmol/L (about < 6 mg/dL) was achieved. In total, 93% of patients were male, and 69% were White. Patients were enrolled from February 2019 through December 2021. The final study visit was in January 2023. Researchers tracked the mean number of patient-reported gout flares per month with a prespecified noninferiority margin of 0.12 gout flares/month. After the first 6 months, colchicine and placebo were stopped, and researchers continued to track outcomes up to month 12.
From baseline to 6 months, there were more gout flares per month in the placebo group (0.61) than in the colchicine group (0.35). From 6 to 12 months, there were more gout flares among patients who previously received colchicine, compared with those who received placebo. While 22.8% of patients in the colchicine group experienced flares at month 6, the following month – after stopping the drug – 41.2% experienced flares. For comparison, 30.8% of the placebo group experienced flares at month 6, and in month 7, 23.5% experienced flares. The spike in flares in the colchicine group began to decline after month 9.
Among the colchicine and placebo groups, reductions in serum urate at 6 and 12 months were similar, and mean serum urate levels declined below 0.36 mmol/L at 4 months and thereafter. Both groups reached a mean allopurinol dose of 280 mg/d at 6 months.
It is not clear what caused this rise in flares in the colchicine group, Stamp noted in an email to this news organization. Overall, there was no difference in the average number of gout flares between either group over the entire 12-month study period.
The study findings make a case for continuing colchicine prophylaxis considerably beyond the point at which the target urate serum level is reached, said Robert Terkeltaub, MD, professor emeritus of medicine in the division of rheumatology, allergy, and immunology at the University of California, San Diego. He was not involved with the research.
“The flare burden with this really conservative allopurinol dosing regimen is really stunning,” he said in an interview. Despite this “start low and go slow” strategy, flares remained a problem, he noted. “It argues that you need at least a year to get out of the woods and have a lower flare burden, even if colchicine prophylaxis is used for the first 6 months.”
The current standard of care in rheumatology for gout treatment includes flare prophylaxis when starting urate-lowering therapy. In daily practice, “this can (and often does) mean continuing flare prophylaxis for longer than the recommended minimum duration from [established] gout treatment guidelines for an individual patient,” explained Elizabeth R. Graef, DO, assistant professor of medicine at Boston University and a rheumatologist at Boston Medical Center.
There are many different factors to consider when determining a patient’s flare risk beyond serum urate levels and date of last flare, she noted. Flare severity, concurrent diuretic use, dose changes, tophus count, and additional health conditions such as diabetes can all contribute to individual patient outcomes.
“Gout is an erosive arthritis, and every time someone flares, there’s a risk of permanent erosive damage, so I tend to be more aggressive with prophylaxis,” she said.
The study was funded by the Health Research Council of New Zealand. Dr. Graef disclosed no relevant financial relationships. Dr. Terkeltaub consults for LG Chem, Fortress/Urica, Selecta Biosciences, Horizon Therapeutics, Atom Biosciences, Acquist Therapeutics, Generate Biomedicines, AstraZeneca, and Synlogic. Dr. Terkeltaub serves as the nonsalaried president of the Gout, Hyperuricemia, and Crystal-Associated Disease Network, which annually receives unrestricted arms-length grant support from pharma donors.
A version of this article first appeared on Medscape.com.
The use of colchicine prophylaxis during the process of up-titrating allopurinol slowly to reach target serum urate levels for patients with gout proved necessary to reduce the risk of flares in a randomized, double-blind, placebo-controlled noninferiority trial conducted by researchers in New Zealand.
Use of colchicine led to fewer flares during the first 6 months after initiating treatment with allopurinol, but when colchicine and placebo were stopped at 6 months, the number of flares rose, and there was no overall difference in the average number of flares between the placebo and colchicine groups over 12 months.
“Gout flares are common when starting urate-lowering therapy, although may be less frequent using the newer allopurinol ‘start-low, go-slow’ dose escalation strategy,” Lisa Stamp, MBChB, PhD, of the University of Otago, Christchurch, New Zealand, and colleagues write in Annals of the Rheumatic Diseases. “This study was undertaken to determine whether colchicine prophylaxis is required with the more gradual dose escalation of allopurinol to achieve target serum urate.”
The study was published online in the BMJ.
The researchers randomly assigned 200 participants to receive colchicine 0.5 mg daily or placebo. At the same time, allopurinol 50 mg daily was initially given to those with an estimated glomerular filtration rate (eGFR) less than 60 mL/min/1.73 m2, and 100 mg daily was given to those with eGFR greater than or equal to 60 mL/min/1.73 m2; doses were increased monthly by 50 mg daily for those with eGFR less than 60 and by 100 mg daily for those with eGFR greater than or equal to 60 until the target urate level of less than 0.36 mmol/L (about < 6 mg/dL) was achieved. In total, 93% of patients were male, and 69% were White. Patients were enrolled from February 2019 through December 2021. The final study visit was in January 2023. Researchers tracked the mean number of patient-reported gout flares per month with a prespecified noninferiority margin of 0.12 gout flares/month. After the first 6 months, colchicine and placebo were stopped, and researchers continued to track outcomes up to month 12.
From baseline to 6 months, there were more gout flares per month in the placebo group (0.61) than in the colchicine group (0.35). From 6 to 12 months, there were more gout flares among patients who previously received colchicine, compared with those who received placebo. While 22.8% of patients in the colchicine group experienced flares at month 6, the following month – after stopping the drug – 41.2% experienced flares. For comparison, 30.8% of the placebo group experienced flares at month 6, and in month 7, 23.5% experienced flares. The spike in flares in the colchicine group began to decline after month 9.
Among the colchicine and placebo groups, reductions in serum urate at 6 and 12 months were similar, and mean serum urate levels declined below 0.36 mmol/L at 4 months and thereafter. Both groups reached a mean allopurinol dose of 280 mg/d at 6 months.
It is not clear what caused this rise in flares in the colchicine group, Stamp noted in an email to this news organization. Overall, there was no difference in the average number of gout flares between either group over the entire 12-month study period.
The study findings make a case for continuing colchicine prophylaxis considerably beyond the point at which the target urate serum level is reached, said Robert Terkeltaub, MD, professor emeritus of medicine in the division of rheumatology, allergy, and immunology at the University of California, San Diego. He was not involved with the research.
“The flare burden with this really conservative allopurinol dosing regimen is really stunning,” he said in an interview. Despite this “start low and go slow” strategy, flares remained a problem, he noted. “It argues that you need at least a year to get out of the woods and have a lower flare burden, even if colchicine prophylaxis is used for the first 6 months.”
The current standard of care in rheumatology for gout treatment includes flare prophylaxis when starting urate-lowering therapy. In daily practice, “this can (and often does) mean continuing flare prophylaxis for longer than the recommended minimum duration from [established] gout treatment guidelines for an individual patient,” explained Elizabeth R. Graef, DO, assistant professor of medicine at Boston University and a rheumatologist at Boston Medical Center.
There are many different factors to consider when determining a patient’s flare risk beyond serum urate levels and date of last flare, she noted. Flare severity, concurrent diuretic use, dose changes, tophus count, and additional health conditions such as diabetes can all contribute to individual patient outcomes.
“Gout is an erosive arthritis, and every time someone flares, there’s a risk of permanent erosive damage, so I tend to be more aggressive with prophylaxis,” she said.
The study was funded by the Health Research Council of New Zealand. Dr. Graef disclosed no relevant financial relationships. Dr. Terkeltaub consults for LG Chem, Fortress/Urica, Selecta Biosciences, Horizon Therapeutics, Atom Biosciences, Acquist Therapeutics, Generate Biomedicines, AstraZeneca, and Synlogic. Dr. Terkeltaub serves as the nonsalaried president of the Gout, Hyperuricemia, and Crystal-Associated Disease Network, which annually receives unrestricted arms-length grant support from pharma donors.
A version of this article first appeared on Medscape.com.
The use of colchicine prophylaxis during the process of up-titrating allopurinol slowly to reach target serum urate levels for patients with gout proved necessary to reduce the risk of flares in a randomized, double-blind, placebo-controlled noninferiority trial conducted by researchers in New Zealand.
Use of colchicine led to fewer flares during the first 6 months after initiating treatment with allopurinol, but when colchicine and placebo were stopped at 6 months, the number of flares rose, and there was no overall difference in the average number of flares between the placebo and colchicine groups over 12 months.
“Gout flares are common when starting urate-lowering therapy, although may be less frequent using the newer allopurinol ‘start-low, go-slow’ dose escalation strategy,” Lisa Stamp, MBChB, PhD, of the University of Otago, Christchurch, New Zealand, and colleagues write in Annals of the Rheumatic Diseases. “This study was undertaken to determine whether colchicine prophylaxis is required with the more gradual dose escalation of allopurinol to achieve target serum urate.”
The study was published online in the BMJ.
The researchers randomly assigned 200 participants to receive colchicine 0.5 mg daily or placebo. At the same time, allopurinol 50 mg daily was initially given to those with an estimated glomerular filtration rate (eGFR) less than 60 mL/min/1.73 m2, and 100 mg daily was given to those with eGFR greater than or equal to 60 mL/min/1.73 m2; doses were increased monthly by 50 mg daily for those with eGFR less than 60 and by 100 mg daily for those with eGFR greater than or equal to 60 until the target urate level of less than 0.36 mmol/L (about < 6 mg/dL) was achieved. In total, 93% of patients were male, and 69% were White. Patients were enrolled from February 2019 through December 2021. The final study visit was in January 2023. Researchers tracked the mean number of patient-reported gout flares per month with a prespecified noninferiority margin of 0.12 gout flares/month. After the first 6 months, colchicine and placebo were stopped, and researchers continued to track outcomes up to month 12.
From baseline to 6 months, there were more gout flares per month in the placebo group (0.61) than in the colchicine group (0.35). From 6 to 12 months, there were more gout flares among patients who previously received colchicine, compared with those who received placebo. While 22.8% of patients in the colchicine group experienced flares at month 6, the following month – after stopping the drug – 41.2% experienced flares. For comparison, 30.8% of the placebo group experienced flares at month 6, and in month 7, 23.5% experienced flares. The spike in flares in the colchicine group began to decline after month 9.
Among the colchicine and placebo groups, reductions in serum urate at 6 and 12 months were similar, and mean serum urate levels declined below 0.36 mmol/L at 4 months and thereafter. Both groups reached a mean allopurinol dose of 280 mg/d at 6 months.
It is not clear what caused this rise in flares in the colchicine group, Stamp noted in an email to this news organization. Overall, there was no difference in the average number of gout flares between either group over the entire 12-month study period.
The study findings make a case for continuing colchicine prophylaxis considerably beyond the point at which the target urate serum level is reached, said Robert Terkeltaub, MD, professor emeritus of medicine in the division of rheumatology, allergy, and immunology at the University of California, San Diego. He was not involved with the research.
“The flare burden with this really conservative allopurinol dosing regimen is really stunning,” he said in an interview. Despite this “start low and go slow” strategy, flares remained a problem, he noted. “It argues that you need at least a year to get out of the woods and have a lower flare burden, even if colchicine prophylaxis is used for the first 6 months.”
The current standard of care in rheumatology for gout treatment includes flare prophylaxis when starting urate-lowering therapy. In daily practice, “this can (and often does) mean continuing flare prophylaxis for longer than the recommended minimum duration from [established] gout treatment guidelines for an individual patient,” explained Elizabeth R. Graef, DO, assistant professor of medicine at Boston University and a rheumatologist at Boston Medical Center.
There are many different factors to consider when determining a patient’s flare risk beyond serum urate levels and date of last flare, she noted. Flare severity, concurrent diuretic use, dose changes, tophus count, and additional health conditions such as diabetes can all contribute to individual patient outcomes.
“Gout is an erosive arthritis, and every time someone flares, there’s a risk of permanent erosive damage, so I tend to be more aggressive with prophylaxis,” she said.
The study was funded by the Health Research Council of New Zealand. Dr. Graef disclosed no relevant financial relationships. Dr. Terkeltaub consults for LG Chem, Fortress/Urica, Selecta Biosciences, Horizon Therapeutics, Atom Biosciences, Acquist Therapeutics, Generate Biomedicines, AstraZeneca, and Synlogic. Dr. Terkeltaub serves as the nonsalaried president of the Gout, Hyperuricemia, and Crystal-Associated Disease Network, which annually receives unrestricted arms-length grant support from pharma donors.
A version of this article first appeared on Medscape.com.
Navigating chronic cough in primary care
Chronic cough took center stage at the European Respiratory Society Congress session titled “Conditions We Are Just Dealing With the Tip of the Iceberg in Primary Care: Frequently Mismanaged Conditions in Primary Health Care.”
“When it comes to chronic cough, general practitioners often feel lost,” Miguel Román Rodríguez, family doctor and an associate professor of family medicine at the University of the Balearic Islands, Palma, Mallorca, Spain, and one of the chairs of the session, said to this news organization.
“GPs are central in diagnosing conditions like chronic cough. We bring something that the specialists don’t bring: the knowledge of the context, of the family, the longitudinal history,” echoed the second chair of the session, Hilary Pinnock, family physician and professor of primary care respiratory medicine at the University of Edinburgh.
Understanding the multifaceted nature of chronic cough
Imran Satia, an assistant professor at McMaster University, Hamilton, Ont., guided attendees at the Milan, Italy, meeting through a comprehensive exploration of chronic cough. The first issue he addressed was the definition of the condition, emphasizing that it is defined by its duration, with chronic cough typically lasting for more than 8 weeks. Prof. Satia pointed out common associations of chronic cough, including asthma, nasal disease, and reflux disease.
Delving into epidemiology, he cited a meta-analysis indicating a global prevalence of approximately 10% in the adult population, with significant regional variability: from 18.1% in Australia to 2.3% in Africa. Notably, the Canadian Longitudinal Study on Aging found an overall prevalence of 16% at baseline. “The most common risk factor was smoke, but even in nonsmokers the prevalence reached 10%,” Prof. Satia added, highlighting that it increased with age and changed depending on location. “The most common associated comorbidities were heart failure and hypertension, but also conditions related to chronic pain, mood, and anxiety,” he explained.
Mental health was identified as a crucial factor in chronic cough, with psychological distress and depressive symptoms emerging as risk factors for developing chronic cough over the next 3 years, contributing to a 20% increased risk.
Effective management strategies
Prof. Satia proposed the use of algorithms to aid in the management of patients with chronic cough in primary care. He introduced a Canadian algorithm that offers specific recommendations for both primary and secondary care.
The algorithm’s primary care assessment, step 1, includes a comprehensive evaluation of the cough history (duration, severity, triggers, nature, location); cardiorespiratory, gastrointestinal, and nasal symptoms; and use of angiotensin-converting enzyme inhibitors and smoking status. Essential diagnostic tests, such as chest radiography (to check for structural disease), complete blood cell count, and spirometry (with or without bronchodilator reversibility), were emphasized. Urgent referral criteria encompassed symptoms like hemoptysis, weight loss, fever, or abnormal chest radiography findings.
“When checking for cough history, GPs should always consider factors like the presence of dry or productive cough, mental health, presence of chronic pain, stroke, and swallowing,” said Prof. Satia, stressing the importance of documenting the impact of chronic cough on quality of life, work life, social life, and family life. “This is something that doctors sometimes do not ask about. They may think that these are not major problems, but acknowledging their importance can help the patient,” he added.
Step 2 of the algorithm focuses on treatment options tailored to specific diagnoses, such as asthma or chronic obstructive pulmonary disease. Prof. Satia urged caution, emphasizing that treatment should only be initiated when evidence of these conditions is present. Additionally, he encouraged early consideration of cough hypersensitivity syndrome when patients exhibit coughing in response to low levels of mechanical stimulation.
Current treatments and future prospects
Prof. Satia presented an overview of existing treatments for chronic cough, outlining their respective advantages and disadvantages. For instance, speech therapy is a patient-led approach with no side effects but entails challenges related to access, costs, and patient motivation. On the other hand, low-dose morphine offers rapid relief but is associated with issues like nausea, stigma, and constipation.
Looking ahead, Prof. Satia shared the results of COUGH-1 and COUGH-2, pivotal phase 3 trials evaluating the oral, peripherally acting P2X3-receptor antagonist gefapixant. This drug, currently approved in Switzerland and Japan, demonstrated a significant reduction in cough frequency, compared with placebo, with rapid and sustained effects. “The estimated relative reduction for 45 mg was 18.45% in COUGH-1 (12 weeks) and 14.64% in COUGH-2 (24 weeks). Of note, cough reduction is very quick and sustained with gefapixant, but a 40% reduction is observed in the placebo arm,” commented Prof. Satia.
Experts unanimously stressed the importance for specialists and GPs of effective communication in managing chronic cough, involving both patients and their families.
“As GPs, we are crucial to manage the common problems, but we are also crucial to spot the needle in the haystack: this is extremely difficult and challenging, and we need support from our colleagues,” Dr. Pinnock concluded.
Prof. Satia reported funding from Merck MSD, AstraZeneca, and GSK; consulting fees from Merck MSD, Genentech, and Respiplus; and speaker fees from AstraZeneca, GSK, and Merck MSD.
A version of this article first appeared on Medscape.com.
Chronic cough took center stage at the European Respiratory Society Congress session titled “Conditions We Are Just Dealing With the Tip of the Iceberg in Primary Care: Frequently Mismanaged Conditions in Primary Health Care.”
“When it comes to chronic cough, general practitioners often feel lost,” Miguel Román Rodríguez, family doctor and an associate professor of family medicine at the University of the Balearic Islands, Palma, Mallorca, Spain, and one of the chairs of the session, said to this news organization.
“GPs are central in diagnosing conditions like chronic cough. We bring something that the specialists don’t bring: the knowledge of the context, of the family, the longitudinal history,” echoed the second chair of the session, Hilary Pinnock, family physician and professor of primary care respiratory medicine at the University of Edinburgh.
Understanding the multifaceted nature of chronic cough
Imran Satia, an assistant professor at McMaster University, Hamilton, Ont., guided attendees at the Milan, Italy, meeting through a comprehensive exploration of chronic cough. The first issue he addressed was the definition of the condition, emphasizing that it is defined by its duration, with chronic cough typically lasting for more than 8 weeks. Prof. Satia pointed out common associations of chronic cough, including asthma, nasal disease, and reflux disease.
Delving into epidemiology, he cited a meta-analysis indicating a global prevalence of approximately 10% in the adult population, with significant regional variability: from 18.1% in Australia to 2.3% in Africa. Notably, the Canadian Longitudinal Study on Aging found an overall prevalence of 16% at baseline. “The most common risk factor was smoke, but even in nonsmokers the prevalence reached 10%,” Prof. Satia added, highlighting that it increased with age and changed depending on location. “The most common associated comorbidities were heart failure and hypertension, but also conditions related to chronic pain, mood, and anxiety,” he explained.
Mental health was identified as a crucial factor in chronic cough, with psychological distress and depressive symptoms emerging as risk factors for developing chronic cough over the next 3 years, contributing to a 20% increased risk.
Effective management strategies
Prof. Satia proposed the use of algorithms to aid in the management of patients with chronic cough in primary care. He introduced a Canadian algorithm that offers specific recommendations for both primary and secondary care.
The algorithm’s primary care assessment, step 1, includes a comprehensive evaluation of the cough history (duration, severity, triggers, nature, location); cardiorespiratory, gastrointestinal, and nasal symptoms; and use of angiotensin-converting enzyme inhibitors and smoking status. Essential diagnostic tests, such as chest radiography (to check for structural disease), complete blood cell count, and spirometry (with or without bronchodilator reversibility), were emphasized. Urgent referral criteria encompassed symptoms like hemoptysis, weight loss, fever, or abnormal chest radiography findings.
“When checking for cough history, GPs should always consider factors like the presence of dry or productive cough, mental health, presence of chronic pain, stroke, and swallowing,” said Prof. Satia, stressing the importance of documenting the impact of chronic cough on quality of life, work life, social life, and family life. “This is something that doctors sometimes do not ask about. They may think that these are not major problems, but acknowledging their importance can help the patient,” he added.
Step 2 of the algorithm focuses on treatment options tailored to specific diagnoses, such as asthma or chronic obstructive pulmonary disease. Prof. Satia urged caution, emphasizing that treatment should only be initiated when evidence of these conditions is present. Additionally, he encouraged early consideration of cough hypersensitivity syndrome when patients exhibit coughing in response to low levels of mechanical stimulation.
Current treatments and future prospects
Prof. Satia presented an overview of existing treatments for chronic cough, outlining their respective advantages and disadvantages. For instance, speech therapy is a patient-led approach with no side effects but entails challenges related to access, costs, and patient motivation. On the other hand, low-dose morphine offers rapid relief but is associated with issues like nausea, stigma, and constipation.
Looking ahead, Prof. Satia shared the results of COUGH-1 and COUGH-2, pivotal phase 3 trials evaluating the oral, peripherally acting P2X3-receptor antagonist gefapixant. This drug, currently approved in Switzerland and Japan, demonstrated a significant reduction in cough frequency, compared with placebo, with rapid and sustained effects. “The estimated relative reduction for 45 mg was 18.45% in COUGH-1 (12 weeks) and 14.64% in COUGH-2 (24 weeks). Of note, cough reduction is very quick and sustained with gefapixant, but a 40% reduction is observed in the placebo arm,” commented Prof. Satia.
Experts unanimously stressed the importance for specialists and GPs of effective communication in managing chronic cough, involving both patients and their families.
“As GPs, we are crucial to manage the common problems, but we are also crucial to spot the needle in the haystack: this is extremely difficult and challenging, and we need support from our colleagues,” Dr. Pinnock concluded.
Prof. Satia reported funding from Merck MSD, AstraZeneca, and GSK; consulting fees from Merck MSD, Genentech, and Respiplus; and speaker fees from AstraZeneca, GSK, and Merck MSD.
A version of this article first appeared on Medscape.com.
Chronic cough took center stage at the European Respiratory Society Congress session titled “Conditions We Are Just Dealing With the Tip of the Iceberg in Primary Care: Frequently Mismanaged Conditions in Primary Health Care.”
“When it comes to chronic cough, general practitioners often feel lost,” Miguel Román Rodríguez, family doctor and an associate professor of family medicine at the University of the Balearic Islands, Palma, Mallorca, Spain, and one of the chairs of the session, said to this news organization.
“GPs are central in diagnosing conditions like chronic cough. We bring something that the specialists don’t bring: the knowledge of the context, of the family, the longitudinal history,” echoed the second chair of the session, Hilary Pinnock, family physician and professor of primary care respiratory medicine at the University of Edinburgh.
Understanding the multifaceted nature of chronic cough
Imran Satia, an assistant professor at McMaster University, Hamilton, Ont., guided attendees at the Milan, Italy, meeting through a comprehensive exploration of chronic cough. The first issue he addressed was the definition of the condition, emphasizing that it is defined by its duration, with chronic cough typically lasting for more than 8 weeks. Prof. Satia pointed out common associations of chronic cough, including asthma, nasal disease, and reflux disease.
Delving into epidemiology, he cited a meta-analysis indicating a global prevalence of approximately 10% in the adult population, with significant regional variability: from 18.1% in Australia to 2.3% in Africa. Notably, the Canadian Longitudinal Study on Aging found an overall prevalence of 16% at baseline. “The most common risk factor was smoke, but even in nonsmokers the prevalence reached 10%,” Prof. Satia added, highlighting that it increased with age and changed depending on location. “The most common associated comorbidities were heart failure and hypertension, but also conditions related to chronic pain, mood, and anxiety,” he explained.
Mental health was identified as a crucial factor in chronic cough, with psychological distress and depressive symptoms emerging as risk factors for developing chronic cough over the next 3 years, contributing to a 20% increased risk.
Effective management strategies
Prof. Satia proposed the use of algorithms to aid in the management of patients with chronic cough in primary care. He introduced a Canadian algorithm that offers specific recommendations for both primary and secondary care.
The algorithm’s primary care assessment, step 1, includes a comprehensive evaluation of the cough history (duration, severity, triggers, nature, location); cardiorespiratory, gastrointestinal, and nasal symptoms; and use of angiotensin-converting enzyme inhibitors and smoking status. Essential diagnostic tests, such as chest radiography (to check for structural disease), complete blood cell count, and spirometry (with or without bronchodilator reversibility), were emphasized. Urgent referral criteria encompassed symptoms like hemoptysis, weight loss, fever, or abnormal chest radiography findings.
“When checking for cough history, GPs should always consider factors like the presence of dry or productive cough, mental health, presence of chronic pain, stroke, and swallowing,” said Prof. Satia, stressing the importance of documenting the impact of chronic cough on quality of life, work life, social life, and family life. “This is something that doctors sometimes do not ask about. They may think that these are not major problems, but acknowledging their importance can help the patient,” he added.
Step 2 of the algorithm focuses on treatment options tailored to specific diagnoses, such as asthma or chronic obstructive pulmonary disease. Prof. Satia urged caution, emphasizing that treatment should only be initiated when evidence of these conditions is present. Additionally, he encouraged early consideration of cough hypersensitivity syndrome when patients exhibit coughing in response to low levels of mechanical stimulation.
Current treatments and future prospects
Prof. Satia presented an overview of existing treatments for chronic cough, outlining their respective advantages and disadvantages. For instance, speech therapy is a patient-led approach with no side effects but entails challenges related to access, costs, and patient motivation. On the other hand, low-dose morphine offers rapid relief but is associated with issues like nausea, stigma, and constipation.
Looking ahead, Prof. Satia shared the results of COUGH-1 and COUGH-2, pivotal phase 3 trials evaluating the oral, peripherally acting P2X3-receptor antagonist gefapixant. This drug, currently approved in Switzerland and Japan, demonstrated a significant reduction in cough frequency, compared with placebo, with rapid and sustained effects. “The estimated relative reduction for 45 mg was 18.45% in COUGH-1 (12 weeks) and 14.64% in COUGH-2 (24 weeks). Of note, cough reduction is very quick and sustained with gefapixant, but a 40% reduction is observed in the placebo arm,” commented Prof. Satia.
Experts unanimously stressed the importance for specialists and GPs of effective communication in managing chronic cough, involving both patients and their families.
“As GPs, we are crucial to manage the common problems, but we are also crucial to spot the needle in the haystack: this is extremely difficult and challenging, and we need support from our colleagues,” Dr. Pinnock concluded.
Prof. Satia reported funding from Merck MSD, AstraZeneca, and GSK; consulting fees from Merck MSD, Genentech, and Respiplus; and speaker fees from AstraZeneca, GSK, and Merck MSD.
A version of this article first appeared on Medscape.com.
FROM ERS 2023
Hepatic presentations of celiac disease
Liver biopsy findings may include variable degrees of steatosis, inflammation, and fibrosis.
In one case we have seen, the patient presented with unexplained ascites and features suggestive of Budd-Chiari syndrome. The serum ascites albumin gradient was 2.3 with a total protein of 0.8 g/dL, and albumin 0.5 g/dL, with an ascitic WBC count of 88/mm3.
Echocardiography showed an ejection fraction of 80%. Transjugular liver biopsy revealed a normal hepatic venous pressure gradient but marked sinusoidal dilatation and congestion with hepatocyte atrophy and focal necrosis suggestive of vascular outlet obstruction (Figure 1).
Hepatic venography, however, showed no evidence of Budd-Chiari syndrome. When seen in consultation, pertinent observations included Irish ancestry, a history of occasional diarrhea, short stature, osteoporosis, and an atrophic spleen on computed tomography. An IgA transglutaminase antibody was positive, and a small-bowel biopsy confirmed celiac disease (Figure 2).
On a gluten-free diet, the patient’s symptoms resolved, with clinical and laboratory abnormalities returning to normal. She lived another 20 years before dying of primary pulmonary hypertension. Recognition of an unusual hepatic manifestation of celiac disease led to effective management.
Dr. Friedman is the Anton R. Fried, MD, Chair of the department of medicine at Newton-Wellesley Hospital in Newton, Mass., and assistant chief of medicine at Massachusetts General Hospital, and a professor of medicine at Harvard Medical School and Tufts University School of Medicine, all in Boston. Dr. Martin is chief of the division of digestive health and liver diseases at the Miller School of Medicine, University of Miami, where he is the Mandel Chair of Gastroenterology. The authors disclose no conflicts.
Previously published in Gastro Hep Advances. 2023. doi: 10.1016/j.gastha.2023.03.018.
Liver biopsy findings may include variable degrees of steatosis, inflammation, and fibrosis.
In one case we have seen, the patient presented with unexplained ascites and features suggestive of Budd-Chiari syndrome. The serum ascites albumin gradient was 2.3 with a total protein of 0.8 g/dL, and albumin 0.5 g/dL, with an ascitic WBC count of 88/mm3.
Echocardiography showed an ejection fraction of 80%. Transjugular liver biopsy revealed a normal hepatic venous pressure gradient but marked sinusoidal dilatation and congestion with hepatocyte atrophy and focal necrosis suggestive of vascular outlet obstruction (Figure 1).
Hepatic venography, however, showed no evidence of Budd-Chiari syndrome. When seen in consultation, pertinent observations included Irish ancestry, a history of occasional diarrhea, short stature, osteoporosis, and an atrophic spleen on computed tomography. An IgA transglutaminase antibody was positive, and a small-bowel biopsy confirmed celiac disease (Figure 2).
On a gluten-free diet, the patient’s symptoms resolved, with clinical and laboratory abnormalities returning to normal. She lived another 20 years before dying of primary pulmonary hypertension. Recognition of an unusual hepatic manifestation of celiac disease led to effective management.
Dr. Friedman is the Anton R. Fried, MD, Chair of the department of medicine at Newton-Wellesley Hospital in Newton, Mass., and assistant chief of medicine at Massachusetts General Hospital, and a professor of medicine at Harvard Medical School and Tufts University School of Medicine, all in Boston. Dr. Martin is chief of the division of digestive health and liver diseases at the Miller School of Medicine, University of Miami, where he is the Mandel Chair of Gastroenterology. The authors disclose no conflicts.
Previously published in Gastro Hep Advances. 2023. doi: 10.1016/j.gastha.2023.03.018.
Liver biopsy findings may include variable degrees of steatosis, inflammation, and fibrosis.
In one case we have seen, the patient presented with unexplained ascites and features suggestive of Budd-Chiari syndrome. The serum ascites albumin gradient was 2.3 with a total protein of 0.8 g/dL, and albumin 0.5 g/dL, with an ascitic WBC count of 88/mm3.
Echocardiography showed an ejection fraction of 80%. Transjugular liver biopsy revealed a normal hepatic venous pressure gradient but marked sinusoidal dilatation and congestion with hepatocyte atrophy and focal necrosis suggestive of vascular outlet obstruction (Figure 1).
Hepatic venography, however, showed no evidence of Budd-Chiari syndrome. When seen in consultation, pertinent observations included Irish ancestry, a history of occasional diarrhea, short stature, osteoporosis, and an atrophic spleen on computed tomography. An IgA transglutaminase antibody was positive, and a small-bowel biopsy confirmed celiac disease (Figure 2).
On a gluten-free diet, the patient’s symptoms resolved, with clinical and laboratory abnormalities returning to normal. She lived another 20 years before dying of primary pulmonary hypertension. Recognition of an unusual hepatic manifestation of celiac disease led to effective management.
Dr. Friedman is the Anton R. Fried, MD, Chair of the department of medicine at Newton-Wellesley Hospital in Newton, Mass., and assistant chief of medicine at Massachusetts General Hospital, and a professor of medicine at Harvard Medical School and Tufts University School of Medicine, all in Boston. Dr. Martin is chief of the division of digestive health and liver diseases at the Miller School of Medicine, University of Miami, where he is the Mandel Chair of Gastroenterology. The authors disclose no conflicts.
Previously published in Gastro Hep Advances. 2023. doi: 10.1016/j.gastha.2023.03.018.
Is additional treatment needed, pretransplant, for r/r AML?
This critically important question was debated at the annual meeting of the Society of Hematologic Oncology, held in Houston and online.
Johannes Schetelig, MD, argued in favor of proceeding to transplant, even without a complete remission.
“In the past, I’ve told many patients with relapsed or refractory AML that we do need to induce a [complete remission] prior to transplantation,” said Dr. Schetelig, from the Clinical Trials Unit at DKMS in Dresden, Germany. “But is it true?”
According to findings from a recent randomized trial, it may not be. The trial, led by Dr. Schetelig, found that patients with AML who received immediate allogeneic transplant without first having achieved a complete response following induction therapy did just as well as those who received intensive salvage induction therapy to establish remission before transplant.
If this finding holds, it “completely upends” how experts have traditionally approached patients with AML, Mikkael A. Sekeres, MD, of the University of Miami said at a conference press briefing last year.
The phase 3 ASAP trial, presented at last year’s American Society of Hematology meeting, included patients with AML who had had a poor response or who had experienced a relapse after first induction therapy. Patients were randomly assigned to a remission-induction strategy prior to allogeneic stem cell transplant (alloHCT) or a disease-control approach of watchful waiting followed by sequential conditioning and alloHCT. The primary endpoint was treatment success, defined as a complete response at day 56 following alloHCT.
In an intention-to-treat analysis, 83.5% of patients in the disease-control group and 81% in the remission-induction group achieved treatment success. Similarly, in the per-protocol analysis, 84.1% and 81.3%, respectively, achieved a complete response at day 56 after alloHCT. After a median follow-up of 4 years, there were no differences in leukemia-free survival or overall survival between the two groups.
Another advantage to forgoing an intensive salvage induction regimen: Patients in the disease-control arm experienced significantly fewer severe adverse events (23% vs. 64% in the remission induction arm) and spent a mean of 27 fewer days in the hospital prior to transplantation.
At last year’s press briefing, Dr. Schetelig said his team did not expect that a complete response on day 56 after transplantation would translate into “equal long-term benefit” for these groups. “This is what I was really astonished about,” he said.
Delving further into the findings, Dr. Schetelig explained that in the remission-induction arm patients who had had a complete response prior to transplantation demonstrated significantly better overall survival at 4 years than those who had not had a complete response at that point: 60% vs. 40%.
The study also revealed that in the disease-control arm, for patients under watchful waiting who did not need low-dose cytarabine and mitoxantrone for disease control, overall survival outcomes were similar to those of patients in the remission-induction arm who achieved a complete response.
These findings suggest that patients who can be bridged with watchful waiting may have a more favorable disease biology, and chemosensitivity could just be a biomarker for disease biology. In other words, “AML biology matters for transplant outcome and not tumor load,” Dr. Schetelig explained.
A recent study that found that having minimal residual disease (MRD) prior to transplant “had no independent effect on leukemia-free survival” supports this idea, he added.
Overall, Dr. Schetelig concluded that data from the ASAP trial suggest that watchful waiting prior to alloHCT represents “an alternative” for some patients.
Counterpoint: Aim for complete remission
Ronald B. Walter, MD, PhD, argued the counterpoint: that residual disease before transplantation is associated with worse posttransplant outcomes and represents a meaningful pretransplant therapeutic target.
The goal of intensifying treatment for patients with residual disease is to erase disease vestiges prior to transplantation.
“The idea is that by doing so you might optimize the benefit-to-risk ratio and ultimately improve outcomes,” said Dr. Walter, of the translational science and therapeutics division at the Fred Hutchinson Cancer Research Center in Seattle.
Several reports support this view that patients who are MRD negative at the time of transplant have significantly better survival outcomes than patients with residual disease who undergo transplant.
A 2016 study from Dr. Walter and colleagues at Fred Hutchinson, for instance, found that 3-year overall survival was significantly higher among patients with no MRD who underwent myeloablative alloHCT: 73% vs. 26% of those in MRD-positive morphologic remission and 23% of patients with active AML.
Another study, published the year before by a different research team, also revealed that “adult patients with AML in morphologic [complete remission] but with detectable MRD who undergo alloHCT have poor outcomes, which approximates those who undergo transplantation with active disease,” the authors of the 2015 study wrote in a commentary highlighting findings from both studies.
Still, providing intensive therapy prior to transplant comes with drawbacks, Dr. Walter noted. These downsides include potential toxicity from more intense therapy, which may prevent further therapy with curative intent, as well as the possibility that deintensifying therapy could lead to difficult-to-treat relapse.
It may, however, be possible to reduce the intensity of therapy before transplant and still achieve good outcomes after transplant, though the data remain mixed.
One trial found that a reduced-intensity conditioning regimen was associated with a greater risk of relapse post transplant and worse overall survival, compared with standard myeloablative conditioning.
However, another recent trial in which patients with AML or high-risk myelodysplastic syndrome were randomly assigned to either a reduced-intensity conditioning regimen or an intensified version of that regimen prior to transplant demonstrated no difference in relapse rates and overall survival, regardless of patients’ MRD status prior to transplant.
“To me, it’s still key to go into transplant with as little disease as possible,” Dr. Walter said. How much value there is in targeted treatment to further reduce disease burden prior to transplant “will really require further careful study,” he said.
The ASAP trial was sponsored by DKMS. Dr. Schetelig has received honoraria from BeiGene, BMS, Janssen, AstraZeneca, AbbVie, and DKMS. Dr. Walter reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
This critically important question was debated at the annual meeting of the Society of Hematologic Oncology, held in Houston and online.
Johannes Schetelig, MD, argued in favor of proceeding to transplant, even without a complete remission.
“In the past, I’ve told many patients with relapsed or refractory AML that we do need to induce a [complete remission] prior to transplantation,” said Dr. Schetelig, from the Clinical Trials Unit at DKMS in Dresden, Germany. “But is it true?”
According to findings from a recent randomized trial, it may not be. The trial, led by Dr. Schetelig, found that patients with AML who received immediate allogeneic transplant without first having achieved a complete response following induction therapy did just as well as those who received intensive salvage induction therapy to establish remission before transplant.
If this finding holds, it “completely upends” how experts have traditionally approached patients with AML, Mikkael A. Sekeres, MD, of the University of Miami said at a conference press briefing last year.
The phase 3 ASAP trial, presented at last year’s American Society of Hematology meeting, included patients with AML who had had a poor response or who had experienced a relapse after first induction therapy. Patients were randomly assigned to a remission-induction strategy prior to allogeneic stem cell transplant (alloHCT) or a disease-control approach of watchful waiting followed by sequential conditioning and alloHCT. The primary endpoint was treatment success, defined as a complete response at day 56 following alloHCT.
In an intention-to-treat analysis, 83.5% of patients in the disease-control group and 81% in the remission-induction group achieved treatment success. Similarly, in the per-protocol analysis, 84.1% and 81.3%, respectively, achieved a complete response at day 56 after alloHCT. After a median follow-up of 4 years, there were no differences in leukemia-free survival or overall survival between the two groups.
Another advantage to forgoing an intensive salvage induction regimen: Patients in the disease-control arm experienced significantly fewer severe adverse events (23% vs. 64% in the remission induction arm) and spent a mean of 27 fewer days in the hospital prior to transplantation.
At last year’s press briefing, Dr. Schetelig said his team did not expect that a complete response on day 56 after transplantation would translate into “equal long-term benefit” for these groups. “This is what I was really astonished about,” he said.
Delving further into the findings, Dr. Schetelig explained that in the remission-induction arm patients who had had a complete response prior to transplantation demonstrated significantly better overall survival at 4 years than those who had not had a complete response at that point: 60% vs. 40%.
The study also revealed that in the disease-control arm, for patients under watchful waiting who did not need low-dose cytarabine and mitoxantrone for disease control, overall survival outcomes were similar to those of patients in the remission-induction arm who achieved a complete response.
These findings suggest that patients who can be bridged with watchful waiting may have a more favorable disease biology, and chemosensitivity could just be a biomarker for disease biology. In other words, “AML biology matters for transplant outcome and not tumor load,” Dr. Schetelig explained.
A recent study that found that having minimal residual disease (MRD) prior to transplant “had no independent effect on leukemia-free survival” supports this idea, he added.
Overall, Dr. Schetelig concluded that data from the ASAP trial suggest that watchful waiting prior to alloHCT represents “an alternative” for some patients.
Counterpoint: Aim for complete remission
Ronald B. Walter, MD, PhD, argued the counterpoint: that residual disease before transplantation is associated with worse posttransplant outcomes and represents a meaningful pretransplant therapeutic target.
The goal of intensifying treatment for patients with residual disease is to erase disease vestiges prior to transplantation.
“The idea is that by doing so you might optimize the benefit-to-risk ratio and ultimately improve outcomes,” said Dr. Walter, of the translational science and therapeutics division at the Fred Hutchinson Cancer Research Center in Seattle.
Several reports support this view that patients who are MRD negative at the time of transplant have significantly better survival outcomes than patients with residual disease who undergo transplant.
A 2016 study from Dr. Walter and colleagues at Fred Hutchinson, for instance, found that 3-year overall survival was significantly higher among patients with no MRD who underwent myeloablative alloHCT: 73% vs. 26% of those in MRD-positive morphologic remission and 23% of patients with active AML.
Another study, published the year before by a different research team, also revealed that “adult patients with AML in morphologic [complete remission] but with detectable MRD who undergo alloHCT have poor outcomes, which approximates those who undergo transplantation with active disease,” the authors of the 2015 study wrote in a commentary highlighting findings from both studies.
Still, providing intensive therapy prior to transplant comes with drawbacks, Dr. Walter noted. These downsides include potential toxicity from more intense therapy, which may prevent further therapy with curative intent, as well as the possibility that deintensifying therapy could lead to difficult-to-treat relapse.
It may, however, be possible to reduce the intensity of therapy before transplant and still achieve good outcomes after transplant, though the data remain mixed.
One trial found that a reduced-intensity conditioning regimen was associated with a greater risk of relapse post transplant and worse overall survival, compared with standard myeloablative conditioning.
However, another recent trial in which patients with AML or high-risk myelodysplastic syndrome were randomly assigned to either a reduced-intensity conditioning regimen or an intensified version of that regimen prior to transplant demonstrated no difference in relapse rates and overall survival, regardless of patients’ MRD status prior to transplant.
“To me, it’s still key to go into transplant with as little disease as possible,” Dr. Walter said. How much value there is in targeted treatment to further reduce disease burden prior to transplant “will really require further careful study,” he said.
The ASAP trial was sponsored by DKMS. Dr. Schetelig has received honoraria from BeiGene, BMS, Janssen, AstraZeneca, AbbVie, and DKMS. Dr. Walter reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
This critically important question was debated at the annual meeting of the Society of Hematologic Oncology, held in Houston and online.
Johannes Schetelig, MD, argued in favor of proceeding to transplant, even without a complete remission.
“In the past, I’ve told many patients with relapsed or refractory AML that we do need to induce a [complete remission] prior to transplantation,” said Dr. Schetelig, from the Clinical Trials Unit at DKMS in Dresden, Germany. “But is it true?”
According to findings from a recent randomized trial, it may not be. The trial, led by Dr. Schetelig, found that patients with AML who received immediate allogeneic transplant without first having achieved a complete response following induction therapy did just as well as those who received intensive salvage induction therapy to establish remission before transplant.
If this finding holds, it “completely upends” how experts have traditionally approached patients with AML, Mikkael A. Sekeres, MD, of the University of Miami said at a conference press briefing last year.
The phase 3 ASAP trial, presented at last year’s American Society of Hematology meeting, included patients with AML who had had a poor response or who had experienced a relapse after first induction therapy. Patients were randomly assigned to a remission-induction strategy prior to allogeneic stem cell transplant (alloHCT) or a disease-control approach of watchful waiting followed by sequential conditioning and alloHCT. The primary endpoint was treatment success, defined as a complete response at day 56 following alloHCT.
In an intention-to-treat analysis, 83.5% of patients in the disease-control group and 81% in the remission-induction group achieved treatment success. Similarly, in the per-protocol analysis, 84.1% and 81.3%, respectively, achieved a complete response at day 56 after alloHCT. After a median follow-up of 4 years, there were no differences in leukemia-free survival or overall survival between the two groups.
Another advantage to forgoing an intensive salvage induction regimen: Patients in the disease-control arm experienced significantly fewer severe adverse events (23% vs. 64% in the remission induction arm) and spent a mean of 27 fewer days in the hospital prior to transplantation.
At last year’s press briefing, Dr. Schetelig said his team did not expect that a complete response on day 56 after transplantation would translate into “equal long-term benefit” for these groups. “This is what I was really astonished about,” he said.
Delving further into the findings, Dr. Schetelig explained that in the remission-induction arm patients who had had a complete response prior to transplantation demonstrated significantly better overall survival at 4 years than those who had not had a complete response at that point: 60% vs. 40%.
The study also revealed that in the disease-control arm, for patients under watchful waiting who did not need low-dose cytarabine and mitoxantrone for disease control, overall survival outcomes were similar to those of patients in the remission-induction arm who achieved a complete response.
These findings suggest that patients who can be bridged with watchful waiting may have a more favorable disease biology, and chemosensitivity could just be a biomarker for disease biology. In other words, “AML biology matters for transplant outcome and not tumor load,” Dr. Schetelig explained.
A recent study that found that having minimal residual disease (MRD) prior to transplant “had no independent effect on leukemia-free survival” supports this idea, he added.
Overall, Dr. Schetelig concluded that data from the ASAP trial suggest that watchful waiting prior to alloHCT represents “an alternative” for some patients.
Counterpoint: Aim for complete remission
Ronald B. Walter, MD, PhD, argued the counterpoint: that residual disease before transplantation is associated with worse posttransplant outcomes and represents a meaningful pretransplant therapeutic target.
The goal of intensifying treatment for patients with residual disease is to erase disease vestiges prior to transplantation.
“The idea is that by doing so you might optimize the benefit-to-risk ratio and ultimately improve outcomes,” said Dr. Walter, of the translational science and therapeutics division at the Fred Hutchinson Cancer Research Center in Seattle.
Several reports support this view that patients who are MRD negative at the time of transplant have significantly better survival outcomes than patients with residual disease who undergo transplant.
A 2016 study from Dr. Walter and colleagues at Fred Hutchinson, for instance, found that 3-year overall survival was significantly higher among patients with no MRD who underwent myeloablative alloHCT: 73% vs. 26% of those in MRD-positive morphologic remission and 23% of patients with active AML.
Another study, published the year before by a different research team, also revealed that “adult patients with AML in morphologic [complete remission] but with detectable MRD who undergo alloHCT have poor outcomes, which approximates those who undergo transplantation with active disease,” the authors of the 2015 study wrote in a commentary highlighting findings from both studies.
Still, providing intensive therapy prior to transplant comes with drawbacks, Dr. Walter noted. These downsides include potential toxicity from more intense therapy, which may prevent further therapy with curative intent, as well as the possibility that deintensifying therapy could lead to difficult-to-treat relapse.
It may, however, be possible to reduce the intensity of therapy before transplant and still achieve good outcomes after transplant, though the data remain mixed.
One trial found that a reduced-intensity conditioning regimen was associated with a greater risk of relapse post transplant and worse overall survival, compared with standard myeloablative conditioning.
However, another recent trial in which patients with AML or high-risk myelodysplastic syndrome were randomly assigned to either a reduced-intensity conditioning regimen or an intensified version of that regimen prior to transplant demonstrated no difference in relapse rates and overall survival, regardless of patients’ MRD status prior to transplant.
“To me, it’s still key to go into transplant with as little disease as possible,” Dr. Walter said. How much value there is in targeted treatment to further reduce disease burden prior to transplant “will really require further careful study,” he said.
The ASAP trial was sponsored by DKMS. Dr. Schetelig has received honoraria from BeiGene, BMS, Janssen, AstraZeneca, AbbVie, and DKMS. Dr. Walter reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
FROM SOHO 2023