User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
‘In the presence of kindness’: humanitarian Paul Farmer dies
Renowned infectious disease specialist, humanitarian, and healthcare champion for many of the world’s most vulnerable patient populations, Paul Edward Farmer, MD, died suddenly in his sleep from an acute cardiac event on Feb. 21 in Rwanda, where he had been teaching. He was 62.
Dr. Farmer cofounded the Boston-based global nonprofit Partners In Health and spent decades providing healthcare to impoverished communities worldwide, fighting on the frontline to protect underserved communities against deadly pandemics.
Dr. Farmer was the Kolokotrones University Professor and chair of the department of global health and social medicine in the Blavatnik Institute at Harvard Medical School, Boston. He served as chief of the division of global health equity at Brigham and Women’s Hospital, also in Boston.
“Paul dedicated his life to improving human health and advocating for health equity and social justice on a global scale,” said HMS dean George Q. Daley in a letter to the HMS community. “I am particularly shaken by his passing because he was not only a consummate colleague and a beloved mentor, but a close friend. To me, Paul represented the heart and soul of Harvard Medical School.”
He was also chancellor and cofounder of the University of Global Health Equity in Kigali, Rwanda. Before his death, he spent several weeks teaching at the university.
“Paul Farmer’s loss is devastating, but his vision for the world will live on through Partners In Health,” said Partners In Health CEO Sheila Davis in a statement. “Paul taught all those around him the power of accompaniment, love for one another, and solidarity. Our deepest sympathies are with his family.”
Dr. Farmer was born in North Adams, Mass., and grew up in Florida with his parents and five siblings. He attended Duke University on a Benjamin N. Duke Scholarship and received his medical degree in 1988, followed by his PhD in 1990 from Harvard University.
His humanitarian work began when he was a college student volunteering in Haiti in 1983 working with dispossessed farmers. In 1987, he cofounded Partners In Health with the goal of helping patients in poverty-stricken corners of the world.
Under Dr. Farmer’s leadership, the nonprofit tackled major public health crises: Haiti’s devastating 2010 earthquake, drug-resistant tuberculosis in Peru and other countries, and an Ebola outbreak that tore through West Africa.
Dr. Farmer documented his 2014-2015 experience treating Africa’s Ebola patients in a book called “Fevers, Feuds, and Diamonds: Ebola and the Ravages of History.”
He wrote that by the time he arrived, “western Sierra Leone was ground zero of the epidemic, and Upper West Africa was just about the worst place in the world to be critically ill or injured.”
One of his greatest qualities was his ability to connect with patients – to treat them “not like ones who suffered, but like a pal you’d joke with,” said Pardis Sabeti, MD, PhD, a Harvard University geneticist who also spent time in Africa and famously sequenced samples of the Ebola virus’ genome.
Dr. Sabeti and Dr. Farmer bonded over their love for Sierra Leone, and their grief over losing a close colleague to Ebola, Sheik Humarr Khan, who was one of the area’s leading infectious disease experts.
Dr. Sabeti first met Dr. Farmer years earlier as a first-year Harvard medical student when she enrolled in one of his courses. She said students introduced themselves, one by one, each veering into heartfelt testimonies about what Dr. Farmer’s work had meant to them.
Dr. Farmer and Dr. Sabeti were just texting on Feb. 19, and the two were “goofing around in our usual way, and scheming about how to make the world better, as we always did.”
Dr. Farmer was funny, mischievous, and above all, exactly what you would expect upon meeting him, Dr. Sabeti said.
“It’s cliché, but the energetic kick you get from just being in his presence, it’s almost otherworldly,” she said. “It’s not even otherworldly in the sense of: ‘I just came across – greatness.’ It’s more: ‘I just came across kindness.’ ”
Dr. Farmer’s work has been widely distributed in publications including Bulletin of the World Health Organization, The Lancet, the New England Journal of Medicine, Clinical Infectious Diseases, and Social Science & Medicine.
He was awarded the 2020 Berggruen Prize for Philosophy & Culture, the Margaret Mead Award from the American Anthropological Association, the American Medical Association’s Outstanding International Physician (Nathan Davis) Award, and, with his Partners In Health colleagues, the Hilton Humanitarian Prize.
He is survived by his wife, Didi Bertrand Farmer, and their three children.
A verison of this article first appeared on Medscape.com.
Renowned infectious disease specialist, humanitarian, and healthcare champion for many of the world’s most vulnerable patient populations, Paul Edward Farmer, MD, died suddenly in his sleep from an acute cardiac event on Feb. 21 in Rwanda, where he had been teaching. He was 62.
Dr. Farmer cofounded the Boston-based global nonprofit Partners In Health and spent decades providing healthcare to impoverished communities worldwide, fighting on the frontline to protect underserved communities against deadly pandemics.
Dr. Farmer was the Kolokotrones University Professor and chair of the department of global health and social medicine in the Blavatnik Institute at Harvard Medical School, Boston. He served as chief of the division of global health equity at Brigham and Women’s Hospital, also in Boston.
“Paul dedicated his life to improving human health and advocating for health equity and social justice on a global scale,” said HMS dean George Q. Daley in a letter to the HMS community. “I am particularly shaken by his passing because he was not only a consummate colleague and a beloved mentor, but a close friend. To me, Paul represented the heart and soul of Harvard Medical School.”
He was also chancellor and cofounder of the University of Global Health Equity in Kigali, Rwanda. Before his death, he spent several weeks teaching at the university.
“Paul Farmer’s loss is devastating, but his vision for the world will live on through Partners In Health,” said Partners In Health CEO Sheila Davis in a statement. “Paul taught all those around him the power of accompaniment, love for one another, and solidarity. Our deepest sympathies are with his family.”
Dr. Farmer was born in North Adams, Mass., and grew up in Florida with his parents and five siblings. He attended Duke University on a Benjamin N. Duke Scholarship and received his medical degree in 1988, followed by his PhD in 1990 from Harvard University.
His humanitarian work began when he was a college student volunteering in Haiti in 1983 working with dispossessed farmers. In 1987, he cofounded Partners In Health with the goal of helping patients in poverty-stricken corners of the world.
Under Dr. Farmer’s leadership, the nonprofit tackled major public health crises: Haiti’s devastating 2010 earthquake, drug-resistant tuberculosis in Peru and other countries, and an Ebola outbreak that tore through West Africa.
Dr. Farmer documented his 2014-2015 experience treating Africa’s Ebola patients in a book called “Fevers, Feuds, and Diamonds: Ebola and the Ravages of History.”
He wrote that by the time he arrived, “western Sierra Leone was ground zero of the epidemic, and Upper West Africa was just about the worst place in the world to be critically ill or injured.”
One of his greatest qualities was his ability to connect with patients – to treat them “not like ones who suffered, but like a pal you’d joke with,” said Pardis Sabeti, MD, PhD, a Harvard University geneticist who also spent time in Africa and famously sequenced samples of the Ebola virus’ genome.
Dr. Sabeti and Dr. Farmer bonded over their love for Sierra Leone, and their grief over losing a close colleague to Ebola, Sheik Humarr Khan, who was one of the area’s leading infectious disease experts.
Dr. Sabeti first met Dr. Farmer years earlier as a first-year Harvard medical student when she enrolled in one of his courses. She said students introduced themselves, one by one, each veering into heartfelt testimonies about what Dr. Farmer’s work had meant to them.
Dr. Farmer and Dr. Sabeti were just texting on Feb. 19, and the two were “goofing around in our usual way, and scheming about how to make the world better, as we always did.”
Dr. Farmer was funny, mischievous, and above all, exactly what you would expect upon meeting him, Dr. Sabeti said.
“It’s cliché, but the energetic kick you get from just being in his presence, it’s almost otherworldly,” she said. “It’s not even otherworldly in the sense of: ‘I just came across – greatness.’ It’s more: ‘I just came across kindness.’ ”
Dr. Farmer’s work has been widely distributed in publications including Bulletin of the World Health Organization, The Lancet, the New England Journal of Medicine, Clinical Infectious Diseases, and Social Science & Medicine.
He was awarded the 2020 Berggruen Prize for Philosophy & Culture, the Margaret Mead Award from the American Anthropological Association, the American Medical Association’s Outstanding International Physician (Nathan Davis) Award, and, with his Partners In Health colleagues, the Hilton Humanitarian Prize.
He is survived by his wife, Didi Bertrand Farmer, and their three children.
A verison of this article first appeared on Medscape.com.
Renowned infectious disease specialist, humanitarian, and healthcare champion for many of the world’s most vulnerable patient populations, Paul Edward Farmer, MD, died suddenly in his sleep from an acute cardiac event on Feb. 21 in Rwanda, where he had been teaching. He was 62.
Dr. Farmer cofounded the Boston-based global nonprofit Partners In Health and spent decades providing healthcare to impoverished communities worldwide, fighting on the frontline to protect underserved communities against deadly pandemics.
Dr. Farmer was the Kolokotrones University Professor and chair of the department of global health and social medicine in the Blavatnik Institute at Harvard Medical School, Boston. He served as chief of the division of global health equity at Brigham and Women’s Hospital, also in Boston.
“Paul dedicated his life to improving human health and advocating for health equity and social justice on a global scale,” said HMS dean George Q. Daley in a letter to the HMS community. “I am particularly shaken by his passing because he was not only a consummate colleague and a beloved mentor, but a close friend. To me, Paul represented the heart and soul of Harvard Medical School.”
He was also chancellor and cofounder of the University of Global Health Equity in Kigali, Rwanda. Before his death, he spent several weeks teaching at the university.
“Paul Farmer’s loss is devastating, but his vision for the world will live on through Partners In Health,” said Partners In Health CEO Sheila Davis in a statement. “Paul taught all those around him the power of accompaniment, love for one another, and solidarity. Our deepest sympathies are with his family.”
Dr. Farmer was born in North Adams, Mass., and grew up in Florida with his parents and five siblings. He attended Duke University on a Benjamin N. Duke Scholarship and received his medical degree in 1988, followed by his PhD in 1990 from Harvard University.
His humanitarian work began when he was a college student volunteering in Haiti in 1983 working with dispossessed farmers. In 1987, he cofounded Partners In Health with the goal of helping patients in poverty-stricken corners of the world.
Under Dr. Farmer’s leadership, the nonprofit tackled major public health crises: Haiti’s devastating 2010 earthquake, drug-resistant tuberculosis in Peru and other countries, and an Ebola outbreak that tore through West Africa.
Dr. Farmer documented his 2014-2015 experience treating Africa’s Ebola patients in a book called “Fevers, Feuds, and Diamonds: Ebola and the Ravages of History.”
He wrote that by the time he arrived, “western Sierra Leone was ground zero of the epidemic, and Upper West Africa was just about the worst place in the world to be critically ill or injured.”
One of his greatest qualities was his ability to connect with patients – to treat them “not like ones who suffered, but like a pal you’d joke with,” said Pardis Sabeti, MD, PhD, a Harvard University geneticist who also spent time in Africa and famously sequenced samples of the Ebola virus’ genome.
Dr. Sabeti and Dr. Farmer bonded over their love for Sierra Leone, and their grief over losing a close colleague to Ebola, Sheik Humarr Khan, who was one of the area’s leading infectious disease experts.
Dr. Sabeti first met Dr. Farmer years earlier as a first-year Harvard medical student when she enrolled in one of his courses. She said students introduced themselves, one by one, each veering into heartfelt testimonies about what Dr. Farmer’s work had meant to them.
Dr. Farmer and Dr. Sabeti were just texting on Feb. 19, and the two were “goofing around in our usual way, and scheming about how to make the world better, as we always did.”
Dr. Farmer was funny, mischievous, and above all, exactly what you would expect upon meeting him, Dr. Sabeti said.
“It’s cliché, but the energetic kick you get from just being in his presence, it’s almost otherworldly,” she said. “It’s not even otherworldly in the sense of: ‘I just came across – greatness.’ It’s more: ‘I just came across kindness.’ ”
Dr. Farmer’s work has been widely distributed in publications including Bulletin of the World Health Organization, The Lancet, the New England Journal of Medicine, Clinical Infectious Diseases, and Social Science & Medicine.
He was awarded the 2020 Berggruen Prize for Philosophy & Culture, the Margaret Mead Award from the American Anthropological Association, the American Medical Association’s Outstanding International Physician (Nathan Davis) Award, and, with his Partners In Health colleagues, the Hilton Humanitarian Prize.
He is survived by his wife, Didi Bertrand Farmer, and their three children.
A verison of this article first appeared on Medscape.com.
Children and COVID: The Omicron surge has become a retreat
The Omicron decline continued for a fourth consecutive week as new cases of COVID-19 in children fell by 42% from the week before, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
That 42% represents a drop from the 299,000 new cases reported for Feb. 4-10 down to 174,000 for the most recent week, Feb. 11-17. 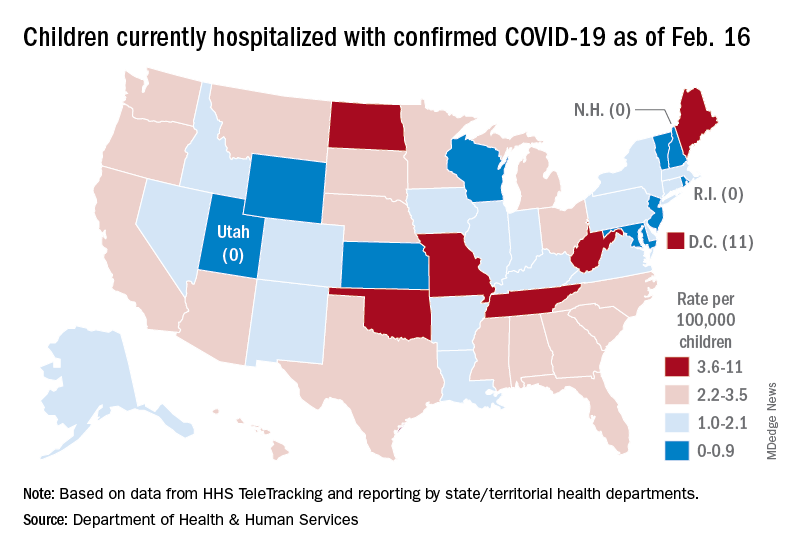
The overall count of COVID-19 cases in children is 12.5 million over the course of the pandemic, and that represents 19% of cases reported among all ages, the AAP and CHA said based on data collected from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
Hospital admissions also continued to fall, with the rate for children aged 0-17 at 0.43 per 100,000 population as of Feb. 20, down by almost 66% from the peak of 1.25 per 100,000 reached on Jan. 16, the Centers for Disease Control and Prevention reported.
A snapshot of the hospitalization situation shows that 1,687 children were occupying inpatient beds on Feb. 16, compared with 4,070 on Jan. 19, which appears to be the peak of the Omicron surge, according to data from the Department of Health & Human Services.
The state with the highest rate – 5.6 per 100,000 children – on Feb. 16 was North Dakota, although the District of Columbia came in at 11.0 per 100,000. They were followed by Oklahoma (5.3), Missouri (5.2), and West Virginia (4.1). There were three states – New Hampshire, Rhode Island, and Utah – with no children in the hospital on that date, the HHS said.
New vaccinations in children aged 5-11 years, which declined in mid- and late January, even as Omicron surged, continued to decline, as did vaccine completions. Vaccinations also fell among children aged 12-17 for the latest reporting week, Feb. 10-16, the AAP said in a separate report.
As more states and school districts drop mask mandates, data from the CDC indicate that 32.5% of 5- to 11-year olds and 67.4% of 12- to 17-year-olds have gotten at least one dose of the COVID-19 vaccine and that 25.1% and 57.3%, respectively, are fully vaccinated. Meanwhile, 20.5% of those fully vaccinated 12- to 17-year-olds have gotten a booster dose, the CDC said.
The Omicron decline continued for a fourth consecutive week as new cases of COVID-19 in children fell by 42% from the week before, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
That 42% represents a drop from the 299,000 new cases reported for Feb. 4-10 down to 174,000 for the most recent week, Feb. 11-17. 
The overall count of COVID-19 cases in children is 12.5 million over the course of the pandemic, and that represents 19% of cases reported among all ages, the AAP and CHA said based on data collected from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
Hospital admissions also continued to fall, with the rate for children aged 0-17 at 0.43 per 100,000 population as of Feb. 20, down by almost 66% from the peak of 1.25 per 100,000 reached on Jan. 16, the Centers for Disease Control and Prevention reported.
A snapshot of the hospitalization situation shows that 1,687 children were occupying inpatient beds on Feb. 16, compared with 4,070 on Jan. 19, which appears to be the peak of the Omicron surge, according to data from the Department of Health & Human Services.
The state with the highest rate – 5.6 per 100,000 children – on Feb. 16 was North Dakota, although the District of Columbia came in at 11.0 per 100,000. They were followed by Oklahoma (5.3), Missouri (5.2), and West Virginia (4.1). There were three states – New Hampshire, Rhode Island, and Utah – with no children in the hospital on that date, the HHS said.
New vaccinations in children aged 5-11 years, which declined in mid- and late January, even as Omicron surged, continued to decline, as did vaccine completions. Vaccinations also fell among children aged 12-17 for the latest reporting week, Feb. 10-16, the AAP said in a separate report.
As more states and school districts drop mask mandates, data from the CDC indicate that 32.5% of 5- to 11-year olds and 67.4% of 12- to 17-year-olds have gotten at least one dose of the COVID-19 vaccine and that 25.1% and 57.3%, respectively, are fully vaccinated. Meanwhile, 20.5% of those fully vaccinated 12- to 17-year-olds have gotten a booster dose, the CDC said.
The Omicron decline continued for a fourth consecutive week as new cases of COVID-19 in children fell by 42% from the week before, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
That 42% represents a drop from the 299,000 new cases reported for Feb. 4-10 down to 174,000 for the most recent week, Feb. 11-17. 
The overall count of COVID-19 cases in children is 12.5 million over the course of the pandemic, and that represents 19% of cases reported among all ages, the AAP and CHA said based on data collected from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
Hospital admissions also continued to fall, with the rate for children aged 0-17 at 0.43 per 100,000 population as of Feb. 20, down by almost 66% from the peak of 1.25 per 100,000 reached on Jan. 16, the Centers for Disease Control and Prevention reported.
A snapshot of the hospitalization situation shows that 1,687 children were occupying inpatient beds on Feb. 16, compared with 4,070 on Jan. 19, which appears to be the peak of the Omicron surge, according to data from the Department of Health & Human Services.
The state with the highest rate – 5.6 per 100,000 children – on Feb. 16 was North Dakota, although the District of Columbia came in at 11.0 per 100,000. They were followed by Oklahoma (5.3), Missouri (5.2), and West Virginia (4.1). There were three states – New Hampshire, Rhode Island, and Utah – with no children in the hospital on that date, the HHS said.
New vaccinations in children aged 5-11 years, which declined in mid- and late January, even as Omicron surged, continued to decline, as did vaccine completions. Vaccinations also fell among children aged 12-17 for the latest reporting week, Feb. 10-16, the AAP said in a separate report.
As more states and school districts drop mask mandates, data from the CDC indicate that 32.5% of 5- to 11-year olds and 67.4% of 12- to 17-year-olds have gotten at least one dose of the COVID-19 vaccine and that 25.1% and 57.3%, respectively, are fully vaccinated. Meanwhile, 20.5% of those fully vaccinated 12- to 17-year-olds have gotten a booster dose, the CDC said.
Subvariant may be more dangerous than original Omicron strain
, a lab study from Japan says.
“Our multiscale investigations suggest that the risk of BA.2 for global health is potentially higher than that of BA.1,” the researchers said in the study published on the preprint server bioRxiv. The study has not been peer-reviewed.
The researchers infected hamsters with BA.1 and BA.2. The hamsters infected with BA.2 got sicker, with more lung damage and loss of body weight. Results were similar when mice were infected with BA.1 and BA.2.
“Infection experiments using hamsters show that BA.2 is more pathogenic than BA.1,” the study said.
BA.1 and BA.2 both appear to evade immunity created by COVID-19 vaccines, the study said. But a booster shot makes illness after infection 74% less likely, CNN said.
What’s more, therapeutic monoclonal antibodies used to treat people infected with COVID didn’t have much effect on BA.2.
BA.2 was “almost completely resistant” to casirivimab and imdevimab and was 35 times more resistant to sotrovimab, compared to the original B.1.1 virus, the researchers wrote.
“In summary, our data suggest the possibility that BA.2 would be the most concerned variant to global health,” the researchers wrote. “Currently, both BA.2 and BA.1 are recognised together as Omicron and these are almost undistinguishable. Based on our findings, we propose that BA.2 should be recognised as a unique variant of concern, and this SARS-CoV-2 variant should be monitored in depth.”
If the World Health Organization recognized BA.2 as a “unique variant of concern,” it would be given its own Greek letter.
But some scientists noted that findings in the lab don’t always reflect what’s happening in the real world of people.
“I think it’s always hard to translate differences in animal and cell culture models to what’s going on with regards to human disease,” Jeremy Kamil, PhD, an associate professor of microbiology and immunology at Louisiana State University Health Shreveport, told Newsweek. “That said, the differences do look real.”
“It might be, from a human’s perspective, a worse virus than BA.1 and might be able to transmit better and cause worse disease,” Daniel Rhoads, MD, section head of microbiology at the Cleveland Clinic in Ohio, told CNN. He reviewed the Japanese study but was not involved in it.
Another scientist who reviewed the study but was not involved in the research noted that human immune systems are evolving along with the COVID variants.
“One of the caveats that we have to think about, as we get new variants that might seem more dangerous, is the fact that there’s two sides to the story,” Deborah Fuller, PhD, a virologist at the University of Washington School of Medicine, told CNN. “Our immune system is evolving as well. And so that’s pushing back on things.”
Scientists have already established that BA.2 is more transmissible than BA.1. The Omicron subvariant has been detected in 74 countries and 47 U.S. states, according to CNN. About 4% of Americans with COVID were infected with BA.2, the outlet reported, citing the CDC, but it’s now the dominant strain in other nations.
It’s not clear yet if BA.2 causes more severe illness in people. While BA.2 spreads faster than BA.1, there’s no evidence the subvariant makes people any sicker, an official with the World Health Organization said, according to CNBC.
A version of this article first appeared on WebMD.com.
, a lab study from Japan says.
“Our multiscale investigations suggest that the risk of BA.2 for global health is potentially higher than that of BA.1,” the researchers said in the study published on the preprint server bioRxiv. The study has not been peer-reviewed.
The researchers infected hamsters with BA.1 and BA.2. The hamsters infected with BA.2 got sicker, with more lung damage and loss of body weight. Results were similar when mice were infected with BA.1 and BA.2.
“Infection experiments using hamsters show that BA.2 is more pathogenic than BA.1,” the study said.
BA.1 and BA.2 both appear to evade immunity created by COVID-19 vaccines, the study said. But a booster shot makes illness after infection 74% less likely, CNN said.
What’s more, therapeutic monoclonal antibodies used to treat people infected with COVID didn’t have much effect on BA.2.
BA.2 was “almost completely resistant” to casirivimab and imdevimab and was 35 times more resistant to sotrovimab, compared to the original B.1.1 virus, the researchers wrote.
“In summary, our data suggest the possibility that BA.2 would be the most concerned variant to global health,” the researchers wrote. “Currently, both BA.2 and BA.1 are recognised together as Omicron and these are almost undistinguishable. Based on our findings, we propose that BA.2 should be recognised as a unique variant of concern, and this SARS-CoV-2 variant should be monitored in depth.”
If the World Health Organization recognized BA.2 as a “unique variant of concern,” it would be given its own Greek letter.
But some scientists noted that findings in the lab don’t always reflect what’s happening in the real world of people.
“I think it’s always hard to translate differences in animal and cell culture models to what’s going on with regards to human disease,” Jeremy Kamil, PhD, an associate professor of microbiology and immunology at Louisiana State University Health Shreveport, told Newsweek. “That said, the differences do look real.”
“It might be, from a human’s perspective, a worse virus than BA.1 and might be able to transmit better and cause worse disease,” Daniel Rhoads, MD, section head of microbiology at the Cleveland Clinic in Ohio, told CNN. He reviewed the Japanese study but was not involved in it.
Another scientist who reviewed the study but was not involved in the research noted that human immune systems are evolving along with the COVID variants.
“One of the caveats that we have to think about, as we get new variants that might seem more dangerous, is the fact that there’s two sides to the story,” Deborah Fuller, PhD, a virologist at the University of Washington School of Medicine, told CNN. “Our immune system is evolving as well. And so that’s pushing back on things.”
Scientists have already established that BA.2 is more transmissible than BA.1. The Omicron subvariant has been detected in 74 countries and 47 U.S. states, according to CNN. About 4% of Americans with COVID were infected with BA.2, the outlet reported, citing the CDC, but it’s now the dominant strain in other nations.
It’s not clear yet if BA.2 causes more severe illness in people. While BA.2 spreads faster than BA.1, there’s no evidence the subvariant makes people any sicker, an official with the World Health Organization said, according to CNBC.
A version of this article first appeared on WebMD.com.
, a lab study from Japan says.
“Our multiscale investigations suggest that the risk of BA.2 for global health is potentially higher than that of BA.1,” the researchers said in the study published on the preprint server bioRxiv. The study has not been peer-reviewed.
The researchers infected hamsters with BA.1 and BA.2. The hamsters infected with BA.2 got sicker, with more lung damage and loss of body weight. Results were similar when mice were infected with BA.1 and BA.2.
“Infection experiments using hamsters show that BA.2 is more pathogenic than BA.1,” the study said.
BA.1 and BA.2 both appear to evade immunity created by COVID-19 vaccines, the study said. But a booster shot makes illness after infection 74% less likely, CNN said.
What’s more, therapeutic monoclonal antibodies used to treat people infected with COVID didn’t have much effect on BA.2.
BA.2 was “almost completely resistant” to casirivimab and imdevimab and was 35 times more resistant to sotrovimab, compared to the original B.1.1 virus, the researchers wrote.
“In summary, our data suggest the possibility that BA.2 would be the most concerned variant to global health,” the researchers wrote. “Currently, both BA.2 and BA.1 are recognised together as Omicron and these are almost undistinguishable. Based on our findings, we propose that BA.2 should be recognised as a unique variant of concern, and this SARS-CoV-2 variant should be monitored in depth.”
If the World Health Organization recognized BA.2 as a “unique variant of concern,” it would be given its own Greek letter.
But some scientists noted that findings in the lab don’t always reflect what’s happening in the real world of people.
“I think it’s always hard to translate differences in animal and cell culture models to what’s going on with regards to human disease,” Jeremy Kamil, PhD, an associate professor of microbiology and immunology at Louisiana State University Health Shreveport, told Newsweek. “That said, the differences do look real.”
“It might be, from a human’s perspective, a worse virus than BA.1 and might be able to transmit better and cause worse disease,” Daniel Rhoads, MD, section head of microbiology at the Cleveland Clinic in Ohio, told CNN. He reviewed the Japanese study but was not involved in it.
Another scientist who reviewed the study but was not involved in the research noted that human immune systems are evolving along with the COVID variants.
“One of the caveats that we have to think about, as we get new variants that might seem more dangerous, is the fact that there’s two sides to the story,” Deborah Fuller, PhD, a virologist at the University of Washington School of Medicine, told CNN. “Our immune system is evolving as well. And so that’s pushing back on things.”
Scientists have already established that BA.2 is more transmissible than BA.1. The Omicron subvariant has been detected in 74 countries and 47 U.S. states, according to CNN. About 4% of Americans with COVID were infected with BA.2, the outlet reported, citing the CDC, but it’s now the dominant strain in other nations.
It’s not clear yet if BA.2 causes more severe illness in people. While BA.2 spreads faster than BA.1, there’s no evidence the subvariant makes people any sicker, an official with the World Health Organization said, according to CNBC.
A version of this article first appeared on WebMD.com.
Malawi declares polio outbreak after girl, 3, paralyzed
Health authorities in Malawi have declared an outbreak of wild poliovirus type 1 after a case was confirmed in a 3-year-old girl in the capital, Lilongwe. It was the first case in Africa in 5 years, according to the World Health Organization.
Globally, there were only five cases of wild poliovirus in 2021, the WHO states.
“As long as wild polio exists anywhere in the world all countries remain at risk of importation of the virus,” Matshidiso Moeti, MBBS, WHO regional director for Africa, said in the statement.
Girl paralyzed in November
The Global Polio Eradication Initiative (GPEI) said in a statement that the 3-year-old girl experienced paralysis in November, and stool specimens were collected. Sequencing of the virus was conducted in February, 2022, by the National Institute for Communicable Diseases in South Africa, and the Centers for Disease Control and Prevention confirmed the case as WPV1.
According to the WHO announcement, laboratory analysis shows that the strain identified in Malawi is linked to one circulating in Sindh Province in Pakistan. Polio remains endemic only in Afghanistan and Pakistan.
Kacey C. Ernst, PhD, MPH, professor and infectious disease epidemiologist at the University of Arizona’s Zuckerman College of Public Health in Tucson, pointed out that what is not clear from the press release is whether the girl had traveled to Pakistan or was infected in Malawi.
“This is a very significant detail that would indicate whether or not transmission was actively occurring in Malawi. Until that information is released, it is hard to judge the extent of the possible outbreak,” she said in an interview. “The good news is that this case was in fact detected. The surveillance systems are in place and they were able to identify wild-type cases.”
Dr. Ernst said that although there is cause for concern, it is “not a reason to panic. Malawi has very high polio vaccination rates and it is quite possible that this will be a very small defined outbreak that will be well contained.”
She added that the medical community should be alerted that this case has been identified so travelers who have been to affected areas who have any symptoms can be appropriately screened.
The WHO said it is helping Malawi health authorities in the response, including increasing immunizations.
However, a vaccination campaign comes at a time of health system upheaval in Malawi.
“Malawi, like countries all over the world, has seen an interruption in services due to COVID,” Joia S. Mukherjee, MD, MPH, chief medical officer with Partners in Health and associate professor with the division of global health equity at Brigham and Women’s Hospital and in the department of global health and social medicine at Harvard Medical School, Boston, said in an interview. “In addition, Malawi is currently dealing with the aftermath of a cyclone – where nearly a million people were displaced. Vaccination campaigns work best if there is solid infrastructure. Both COVID and the impact of climate change have shaken the health system.”
UN health agencies warned last year that millions of children who have not received immunizations during the pandemic, especially in Africa, “are now at risk from life-threatening diseases such as measles, polio, yellow fever, and diphtheria,” Reuters reported.
Africa was certified as wild poliovirus free on Aug. 25, 2020. The CDC had served as the lead partner over 3 decades in helping Africa reach the milestone. Africa will retain that status, the WHO stated, because the strain originated in Pakistan.
Five of six WHO regions have been certified polio free. The Americas received eradication certification in 1994.
There is no cure for polio, which can cause irreversible paralysis within hours, but the disease has been largely eradicated globally with an effective vaccine.
GPEI sending teams
The GPEI is sending a team to Malawi to support emergency operations, communications, and surveillance. Partner organizations will also send teams to support operations and innovative vaccination campaign solutions.
GPEI was launched in 1988 with the combined efforts of national governments, WHO, Rotary International, the CDC, and UNICEF. The GPEI partnership has included the Bill & Melinda Gates Foundation and, in recent years, Gavi, the Vaccine Alliance.
The CDC states, “[G]lobal incidence of polio has decreased by 99.9% since GPEI’s foundation. An estimated 16 million people today are walking who would otherwise have been paralyzed by the disease, and more than 1.5 million people are alive, whose lives would otherwise have been lost. Now the task remains to tackle polio in its last few strongholds and get rid of the final 0.1% of polio cases.”
Three wild poliovirus strains
There are three wild poliovirus strains: type 1 (WPV1), type 2 (WPV2), and type 3 (WPV3).
“Symptomatically, all three strains are identical, in that they cause irreversible paralysis or even death. But there are genetic and virologic differences which make these three strains three separate viruses that must each be eradicated individually,” according to WHO.
WPV3 is the second strain to be wiped out, following the certification of the eradication of WPV2 in 2015.
A version of this article first appeared on Medscape.com.
Health authorities in Malawi have declared an outbreak of wild poliovirus type 1 after a case was confirmed in a 3-year-old girl in the capital, Lilongwe. It was the first case in Africa in 5 years, according to the World Health Organization.
Globally, there were only five cases of wild poliovirus in 2021, the WHO states.
“As long as wild polio exists anywhere in the world all countries remain at risk of importation of the virus,” Matshidiso Moeti, MBBS, WHO regional director for Africa, said in the statement.
Girl paralyzed in November
The Global Polio Eradication Initiative (GPEI) said in a statement that the 3-year-old girl experienced paralysis in November, and stool specimens were collected. Sequencing of the virus was conducted in February, 2022, by the National Institute for Communicable Diseases in South Africa, and the Centers for Disease Control and Prevention confirmed the case as WPV1.
According to the WHO announcement, laboratory analysis shows that the strain identified in Malawi is linked to one circulating in Sindh Province in Pakistan. Polio remains endemic only in Afghanistan and Pakistan.
Kacey C. Ernst, PhD, MPH, professor and infectious disease epidemiologist at the University of Arizona’s Zuckerman College of Public Health in Tucson, pointed out that what is not clear from the press release is whether the girl had traveled to Pakistan or was infected in Malawi.
“This is a very significant detail that would indicate whether or not transmission was actively occurring in Malawi. Until that information is released, it is hard to judge the extent of the possible outbreak,” she said in an interview. “The good news is that this case was in fact detected. The surveillance systems are in place and they were able to identify wild-type cases.”
Dr. Ernst said that although there is cause for concern, it is “not a reason to panic. Malawi has very high polio vaccination rates and it is quite possible that this will be a very small defined outbreak that will be well contained.”
She added that the medical community should be alerted that this case has been identified so travelers who have been to affected areas who have any symptoms can be appropriately screened.
The WHO said it is helping Malawi health authorities in the response, including increasing immunizations.
However, a vaccination campaign comes at a time of health system upheaval in Malawi.
“Malawi, like countries all over the world, has seen an interruption in services due to COVID,” Joia S. Mukherjee, MD, MPH, chief medical officer with Partners in Health and associate professor with the division of global health equity at Brigham and Women’s Hospital and in the department of global health and social medicine at Harvard Medical School, Boston, said in an interview. “In addition, Malawi is currently dealing with the aftermath of a cyclone – where nearly a million people were displaced. Vaccination campaigns work best if there is solid infrastructure. Both COVID and the impact of climate change have shaken the health system.”
UN health agencies warned last year that millions of children who have not received immunizations during the pandemic, especially in Africa, “are now at risk from life-threatening diseases such as measles, polio, yellow fever, and diphtheria,” Reuters reported.
Africa was certified as wild poliovirus free on Aug. 25, 2020. The CDC had served as the lead partner over 3 decades in helping Africa reach the milestone. Africa will retain that status, the WHO stated, because the strain originated in Pakistan.
Five of six WHO regions have been certified polio free. The Americas received eradication certification in 1994.
There is no cure for polio, which can cause irreversible paralysis within hours, but the disease has been largely eradicated globally with an effective vaccine.
GPEI sending teams
The GPEI is sending a team to Malawi to support emergency operations, communications, and surveillance. Partner organizations will also send teams to support operations and innovative vaccination campaign solutions.
GPEI was launched in 1988 with the combined efforts of national governments, WHO, Rotary International, the CDC, and UNICEF. The GPEI partnership has included the Bill & Melinda Gates Foundation and, in recent years, Gavi, the Vaccine Alliance.
The CDC states, “[G]lobal incidence of polio has decreased by 99.9% since GPEI’s foundation. An estimated 16 million people today are walking who would otherwise have been paralyzed by the disease, and more than 1.5 million people are alive, whose lives would otherwise have been lost. Now the task remains to tackle polio in its last few strongholds and get rid of the final 0.1% of polio cases.”
Three wild poliovirus strains
There are three wild poliovirus strains: type 1 (WPV1), type 2 (WPV2), and type 3 (WPV3).
“Symptomatically, all three strains are identical, in that they cause irreversible paralysis or even death. But there are genetic and virologic differences which make these three strains three separate viruses that must each be eradicated individually,” according to WHO.
WPV3 is the second strain to be wiped out, following the certification of the eradication of WPV2 in 2015.
A version of this article first appeared on Medscape.com.
Health authorities in Malawi have declared an outbreak of wild poliovirus type 1 after a case was confirmed in a 3-year-old girl in the capital, Lilongwe. It was the first case in Africa in 5 years, according to the World Health Organization.
Globally, there were only five cases of wild poliovirus in 2021, the WHO states.
“As long as wild polio exists anywhere in the world all countries remain at risk of importation of the virus,” Matshidiso Moeti, MBBS, WHO regional director for Africa, said in the statement.
Girl paralyzed in November
The Global Polio Eradication Initiative (GPEI) said in a statement that the 3-year-old girl experienced paralysis in November, and stool specimens were collected. Sequencing of the virus was conducted in February, 2022, by the National Institute for Communicable Diseases in South Africa, and the Centers for Disease Control and Prevention confirmed the case as WPV1.
According to the WHO announcement, laboratory analysis shows that the strain identified in Malawi is linked to one circulating in Sindh Province in Pakistan. Polio remains endemic only in Afghanistan and Pakistan.
Kacey C. Ernst, PhD, MPH, professor and infectious disease epidemiologist at the University of Arizona’s Zuckerman College of Public Health in Tucson, pointed out that what is not clear from the press release is whether the girl had traveled to Pakistan or was infected in Malawi.
“This is a very significant detail that would indicate whether or not transmission was actively occurring in Malawi. Until that information is released, it is hard to judge the extent of the possible outbreak,” she said in an interview. “The good news is that this case was in fact detected. The surveillance systems are in place and they were able to identify wild-type cases.”
Dr. Ernst said that although there is cause for concern, it is “not a reason to panic. Malawi has very high polio vaccination rates and it is quite possible that this will be a very small defined outbreak that will be well contained.”
She added that the medical community should be alerted that this case has been identified so travelers who have been to affected areas who have any symptoms can be appropriately screened.
The WHO said it is helping Malawi health authorities in the response, including increasing immunizations.
However, a vaccination campaign comes at a time of health system upheaval in Malawi.
“Malawi, like countries all over the world, has seen an interruption in services due to COVID,” Joia S. Mukherjee, MD, MPH, chief medical officer with Partners in Health and associate professor with the division of global health equity at Brigham and Women’s Hospital and in the department of global health and social medicine at Harvard Medical School, Boston, said in an interview. “In addition, Malawi is currently dealing with the aftermath of a cyclone – where nearly a million people were displaced. Vaccination campaigns work best if there is solid infrastructure. Both COVID and the impact of climate change have shaken the health system.”
UN health agencies warned last year that millions of children who have not received immunizations during the pandemic, especially in Africa, “are now at risk from life-threatening diseases such as measles, polio, yellow fever, and diphtheria,” Reuters reported.
Africa was certified as wild poliovirus free on Aug. 25, 2020. The CDC had served as the lead partner over 3 decades in helping Africa reach the milestone. Africa will retain that status, the WHO stated, because the strain originated in Pakistan.
Five of six WHO regions have been certified polio free. The Americas received eradication certification in 1994.
There is no cure for polio, which can cause irreversible paralysis within hours, but the disease has been largely eradicated globally with an effective vaccine.
GPEI sending teams
The GPEI is sending a team to Malawi to support emergency operations, communications, and surveillance. Partner organizations will also send teams to support operations and innovative vaccination campaign solutions.
GPEI was launched in 1988 with the combined efforts of national governments, WHO, Rotary International, the CDC, and UNICEF. The GPEI partnership has included the Bill & Melinda Gates Foundation and, in recent years, Gavi, the Vaccine Alliance.
The CDC states, “[G]lobal incidence of polio has decreased by 99.9% since GPEI’s foundation. An estimated 16 million people today are walking who would otherwise have been paralyzed by the disease, and more than 1.5 million people are alive, whose lives would otherwise have been lost. Now the task remains to tackle polio in its last few strongholds and get rid of the final 0.1% of polio cases.”
Three wild poliovirus strains
There are three wild poliovirus strains: type 1 (WPV1), type 2 (WPV2), and type 3 (WPV3).
“Symptomatically, all three strains are identical, in that they cause irreversible paralysis or even death. But there are genetic and virologic differences which make these three strains three separate viruses that must each be eradicated individually,” according to WHO.
WPV3 is the second strain to be wiped out, following the certification of the eradication of WPV2 in 2015.
A version of this article first appeared on Medscape.com.
Two factors linked to higher risk of long COVID in IBD
Two features are significantly associated with a higher risk for developing long COVID symptoms among people with inflammatory bowel disease (IBD), according to a large Danish population study.
People with Crohn’s disease (CD) who experienced adverse acute COVID-19, defined as requiring hospitalization, were nearly three times more likely to report persistent symptoms 12 weeks after acute infection.
“Long-term, persisting symptoms following COVID-19 is a frequently occurring problem, which is probably underappreciated. IBD specialists should therefore be aware of any of these symptoms and actively ask patients whether they have these problems,” lead author Mohamed Attauabi, MD, PhD, said in an interview.
Dr. Attauabi and colleagues also found that people with ulcerative colitis (UC) who discontinued immunosuppressive agents because of COVID-19 were 1.5 times more likely to experience long COVID symptoms, a result that surprised the researchers.
“This has not been shown before and remains to be confirmed,” said Dr. Attauabi, a fellow in the department of gastroenterology at Herlev Hospital at the University of Copenhagen.
Attauabi presented the results as a digital oral presentation at the 17th congress of the European Crohn’s and Colitis Organisation.
A closer look at IBD and COVID-19
Large, hospital-based studies of symptoms consistent with long COVID reveal a high prevalence of fatigue, sleep difficulties, and anxiety at 12 weeks or more post acute infection. However, these were not specific to people with CD or UC, Dr. Attauabi said.
“In patients with IBD, the risk of long-term sequelae of COVID-19 remains to be investigated,” he said.
Dr. Attauabi and colleagues studied 197 people with CD and 319 with UC, all of whom had polymerase chain reaction–confirmed COVID-19. Participants were prospectively enrolled in the population-based Danish IBD-COVID registry from January 28, 2020 to April 1, 2021. At a median of 5.1 months, a subset of 85 people with CD and 137 with UC agreed to report any post-COVID symptoms.
Older age, smoking, IBD disease activity, and presence of comorbidities were not associated with a significantly elevated risk of long COVID.
In a multivariate analysis, hospitalization for COVID-19 among people with CD was significantly associated with long COVID (odds ratio, 2.76; 95% confidence interval, 1.05-3.90; P = .04).
Furthermore, people with UC who stopped taking immunosuppressive agents also had a significantly higher risk (OR, 1.50; 95% CI, 1.07-10.22; P = .01).
“However, IBD medications such as systemic steroids were not associated with this outcome,” Dr. Attauabi said.
Fatigue most common long COVID symptom
Fatigue was the most common long COVID symptom, reported by 37% of patients with CD and 36% with UC.
Anosmia and ageusia were also common, reported by 29% and 28% of patients with CD, and 27% and 19% of those with UC, respectively.
“In our cohort of patients with UC or CD who developed COVID-19, the long-term health effects of COVID-19 did not appear to differ among patients with UC or CD nor according to IBD medications,” Dr. Attauabi said.
That is a “great study,” said session cochair Torsten Kucharzik, MD, PhD, head of internal medicine and gastroenterology at Lueneburg (Germany) Hospital.
When Dr. Kucharzik asked about smoking, Dr. Attauabi responded that they collected information on current and previous smoking, but they chose not to include the data because it was not statistically significant.
Dr. Attauabi has reported no relevant financial relationships. Dr. Kucharzik has reported receiving grants from Takeda and personal fees from companies including MSD/Essex, AbbVie, Falk Foundation, Biogen, Bristol-Myers Squibb, Arena, Celgene, Celltrion, Ferring, Janssen, Galapagos, Olympus, Mundipharma, Takeda, Amgen, Pfizer, Roche, and Vifor Pharma.
A version of this article first appeared on Medscape.com.
Two features are significantly associated with a higher risk for developing long COVID symptoms among people with inflammatory bowel disease (IBD), according to a large Danish population study.
People with Crohn’s disease (CD) who experienced adverse acute COVID-19, defined as requiring hospitalization, were nearly three times more likely to report persistent symptoms 12 weeks after acute infection.
“Long-term, persisting symptoms following COVID-19 is a frequently occurring problem, which is probably underappreciated. IBD specialists should therefore be aware of any of these symptoms and actively ask patients whether they have these problems,” lead author Mohamed Attauabi, MD, PhD, said in an interview.
Dr. Attauabi and colleagues also found that people with ulcerative colitis (UC) who discontinued immunosuppressive agents because of COVID-19 were 1.5 times more likely to experience long COVID symptoms, a result that surprised the researchers.
“This has not been shown before and remains to be confirmed,” said Dr. Attauabi, a fellow in the department of gastroenterology at Herlev Hospital at the University of Copenhagen.
Attauabi presented the results as a digital oral presentation at the 17th congress of the European Crohn’s and Colitis Organisation.
A closer look at IBD and COVID-19
Large, hospital-based studies of symptoms consistent with long COVID reveal a high prevalence of fatigue, sleep difficulties, and anxiety at 12 weeks or more post acute infection. However, these were not specific to people with CD or UC, Dr. Attauabi said.
“In patients with IBD, the risk of long-term sequelae of COVID-19 remains to be investigated,” he said.
Dr. Attauabi and colleagues studied 197 people with CD and 319 with UC, all of whom had polymerase chain reaction–confirmed COVID-19. Participants were prospectively enrolled in the population-based Danish IBD-COVID registry from January 28, 2020 to April 1, 2021. At a median of 5.1 months, a subset of 85 people with CD and 137 with UC agreed to report any post-COVID symptoms.
Older age, smoking, IBD disease activity, and presence of comorbidities were not associated with a significantly elevated risk of long COVID.
In a multivariate analysis, hospitalization for COVID-19 among people with CD was significantly associated with long COVID (odds ratio, 2.76; 95% confidence interval, 1.05-3.90; P = .04).
Furthermore, people with UC who stopped taking immunosuppressive agents also had a significantly higher risk (OR, 1.50; 95% CI, 1.07-10.22; P = .01).
“However, IBD medications such as systemic steroids were not associated with this outcome,” Dr. Attauabi said.
Fatigue most common long COVID symptom
Fatigue was the most common long COVID symptom, reported by 37% of patients with CD and 36% with UC.
Anosmia and ageusia were also common, reported by 29% and 28% of patients with CD, and 27% and 19% of those with UC, respectively.
“In our cohort of patients with UC or CD who developed COVID-19, the long-term health effects of COVID-19 did not appear to differ among patients with UC or CD nor according to IBD medications,” Dr. Attauabi said.
That is a “great study,” said session cochair Torsten Kucharzik, MD, PhD, head of internal medicine and gastroenterology at Lueneburg (Germany) Hospital.
When Dr. Kucharzik asked about smoking, Dr. Attauabi responded that they collected information on current and previous smoking, but they chose not to include the data because it was not statistically significant.
Dr. Attauabi has reported no relevant financial relationships. Dr. Kucharzik has reported receiving grants from Takeda and personal fees from companies including MSD/Essex, AbbVie, Falk Foundation, Biogen, Bristol-Myers Squibb, Arena, Celgene, Celltrion, Ferring, Janssen, Galapagos, Olympus, Mundipharma, Takeda, Amgen, Pfizer, Roche, and Vifor Pharma.
A version of this article first appeared on Medscape.com.
Two features are significantly associated with a higher risk for developing long COVID symptoms among people with inflammatory bowel disease (IBD), according to a large Danish population study.
People with Crohn’s disease (CD) who experienced adverse acute COVID-19, defined as requiring hospitalization, were nearly three times more likely to report persistent symptoms 12 weeks after acute infection.
“Long-term, persisting symptoms following COVID-19 is a frequently occurring problem, which is probably underappreciated. IBD specialists should therefore be aware of any of these symptoms and actively ask patients whether they have these problems,” lead author Mohamed Attauabi, MD, PhD, said in an interview.
Dr. Attauabi and colleagues also found that people with ulcerative colitis (UC) who discontinued immunosuppressive agents because of COVID-19 were 1.5 times more likely to experience long COVID symptoms, a result that surprised the researchers.
“This has not been shown before and remains to be confirmed,” said Dr. Attauabi, a fellow in the department of gastroenterology at Herlev Hospital at the University of Copenhagen.
Attauabi presented the results as a digital oral presentation at the 17th congress of the European Crohn’s and Colitis Organisation.
A closer look at IBD and COVID-19
Large, hospital-based studies of symptoms consistent with long COVID reveal a high prevalence of fatigue, sleep difficulties, and anxiety at 12 weeks or more post acute infection. However, these were not specific to people with CD or UC, Dr. Attauabi said.
“In patients with IBD, the risk of long-term sequelae of COVID-19 remains to be investigated,” he said.
Dr. Attauabi and colleagues studied 197 people with CD and 319 with UC, all of whom had polymerase chain reaction–confirmed COVID-19. Participants were prospectively enrolled in the population-based Danish IBD-COVID registry from January 28, 2020 to April 1, 2021. At a median of 5.1 months, a subset of 85 people with CD and 137 with UC agreed to report any post-COVID symptoms.
Older age, smoking, IBD disease activity, and presence of comorbidities were not associated with a significantly elevated risk of long COVID.
In a multivariate analysis, hospitalization for COVID-19 among people with CD was significantly associated with long COVID (odds ratio, 2.76; 95% confidence interval, 1.05-3.90; P = .04).
Furthermore, people with UC who stopped taking immunosuppressive agents also had a significantly higher risk (OR, 1.50; 95% CI, 1.07-10.22; P = .01).
“However, IBD medications such as systemic steroids were not associated with this outcome,” Dr. Attauabi said.
Fatigue most common long COVID symptom
Fatigue was the most common long COVID symptom, reported by 37% of patients with CD and 36% with UC.
Anosmia and ageusia were also common, reported by 29% and 28% of patients with CD, and 27% and 19% of those with UC, respectively.
“In our cohort of patients with UC or CD who developed COVID-19, the long-term health effects of COVID-19 did not appear to differ among patients with UC or CD nor according to IBD medications,” Dr. Attauabi said.
That is a “great study,” said session cochair Torsten Kucharzik, MD, PhD, head of internal medicine and gastroenterology at Lueneburg (Germany) Hospital.
When Dr. Kucharzik asked about smoking, Dr. Attauabi responded that they collected information on current and previous smoking, but they chose not to include the data because it was not statistically significant.
Dr. Attauabi has reported no relevant financial relationships. Dr. Kucharzik has reported receiving grants from Takeda and personal fees from companies including MSD/Essex, AbbVie, Falk Foundation, Biogen, Bristol-Myers Squibb, Arena, Celgene, Celltrion, Ferring, Janssen, Galapagos, Olympus, Mundipharma, Takeda, Amgen, Pfizer, Roche, and Vifor Pharma.
A version of this article first appeared on Medscape.com.
FROM ECCO 2022
Ivermectin does not stop progression to severe COVID: randomized trial
Ivermectin treatment given to high-risk patients with mild-to-moderate COVID-19 during the first week of illness did not prevent progression to severe disease, according to results from a randomized clinical trial.
“The study findings do not support the use of ivermectin for patients with COVID-19,” researchers conclude in the paper published online in JAMA Internal Medicine.
The open-label trial was conducted at 20 public hospitals and a COVID-19 quarantine center in Malaysia between May 31 and Oct. 25, 2021. It was led by Steven Chee Loon Lim, MRCP, department of medicine, Raja Permaisuri Bainun Hospital, Perak, Malaysia.
Among 490 patients in the primary analysis, 52 of 241 patients (21.6%) in the ivermectin group and 43 of 249 patients (17.3%) in the control group progressed to severe disease (relative risk, 1.25; 95% confidence interval, 0.87-1.80; P = .25). All major ethnic groups in Malaysia were well represented, the researchers write.
Participants (average age 62.5 and 54.5% women) were randomly assigned 1:1 to receive either a 5-day course of oral ivermectin (0.4 mg/kg body weight daily for 5 days) plus standard of care (n = 241) or standard of care alone (n = 249). Standard of care included symptomatic therapy and monitoring for early deterioration based on clinical findings, laboratory tests, and chest imaging.
Secondary outcomes
Secondary outcomes included rates of mechanical ventilation, intensive care unit (ICU) admission, 28-day in-hospital mortality, and side effects.
In all the secondary outcomes, there were no significant differences between groups.
Mechanical ventilation occurred in four patients on the ivermectin protocol (1.7%) versus 10 patients in the control group (4.0%) (RR, 0.41; 95% CI, 0.13-1.30; P = .17); ICU admission occurred in six (2.4%) versus eight (3.2%) (RR, 0.78; 95% CI, 0.27-2.20; P = .79); and 28-day in-hospital death occurred in three (1.2%) versus 10 (4.0%) (RR, 0.31; 95% CI, 0.09-1.11; P = .09).
The most common adverse event was diarrhea, reported by 5.8% in the ivermectin group and 1.6% in the control group.
No difference by vaccine status
The researchers conducted a subgroup analysis to evaluate any differences in whether participants were vaccinated. They said that analysis was “unremarkable.”
Just more than half of participants (51.8%) were fully vaccinated, with two doses of COVID-19 vaccines. Among the fully vaccinated patients, 17.7% in the ivermectin group and 9.2% in the control group developed severe disease (RR, 1.92; 95% CI, 0.99-3.71; P = .06).
Ivermectin, an inexpensive and widely available antiparasitic drug, is prescribed to treat COVID-19 but has not been approved by the U.S. Food and Drug Administration for that purpose. Evidence-based data for or against use has been sparse.
The authors write that “although some early clinical studies suggested the potential efficacy of ivermectin in the treatment and prevention of COVID-19, these studies had methodologic weaknesses.”
Dr. Lim and colleagues point out that their findings are consistent with those of the IVERCOR-COVID19 trial, which found ivermectin ineffective in reducing hospitalization risk.
Previous randomized trials of ivermectin for COVID-19 patients that have included at least 400 patients have focused on outpatients.
In the current study, the authors note, patients were hospitalized, which allowed investigators to observe administration of ivermectin with a high adherence rate. Additionally, the researchers used clearly defined criteria for determining progression to severe disease.
Limitations of the current study include that the open-label design might lead to under-reporting of adverse events in the control group while overestimating the drug effects of ivermectin. The study was also not designed to assess the effects of ivermectin on mortality from COVID-19.
A version of this article first appeared on Medscape.com.
Ivermectin treatment given to high-risk patients with mild-to-moderate COVID-19 during the first week of illness did not prevent progression to severe disease, according to results from a randomized clinical trial.
“The study findings do not support the use of ivermectin for patients with COVID-19,” researchers conclude in the paper published online in JAMA Internal Medicine.
The open-label trial was conducted at 20 public hospitals and a COVID-19 quarantine center in Malaysia between May 31 and Oct. 25, 2021. It was led by Steven Chee Loon Lim, MRCP, department of medicine, Raja Permaisuri Bainun Hospital, Perak, Malaysia.
Among 490 patients in the primary analysis, 52 of 241 patients (21.6%) in the ivermectin group and 43 of 249 patients (17.3%) in the control group progressed to severe disease (relative risk, 1.25; 95% confidence interval, 0.87-1.80; P = .25). All major ethnic groups in Malaysia were well represented, the researchers write.
Participants (average age 62.5 and 54.5% women) were randomly assigned 1:1 to receive either a 5-day course of oral ivermectin (0.4 mg/kg body weight daily for 5 days) plus standard of care (n = 241) or standard of care alone (n = 249). Standard of care included symptomatic therapy and monitoring for early deterioration based on clinical findings, laboratory tests, and chest imaging.
Secondary outcomes
Secondary outcomes included rates of mechanical ventilation, intensive care unit (ICU) admission, 28-day in-hospital mortality, and side effects.
In all the secondary outcomes, there were no significant differences between groups.
Mechanical ventilation occurred in four patients on the ivermectin protocol (1.7%) versus 10 patients in the control group (4.0%) (RR, 0.41; 95% CI, 0.13-1.30; P = .17); ICU admission occurred in six (2.4%) versus eight (3.2%) (RR, 0.78; 95% CI, 0.27-2.20; P = .79); and 28-day in-hospital death occurred in three (1.2%) versus 10 (4.0%) (RR, 0.31; 95% CI, 0.09-1.11; P = .09).
The most common adverse event was diarrhea, reported by 5.8% in the ivermectin group and 1.6% in the control group.
No difference by vaccine status
The researchers conducted a subgroup analysis to evaluate any differences in whether participants were vaccinated. They said that analysis was “unremarkable.”
Just more than half of participants (51.8%) were fully vaccinated, with two doses of COVID-19 vaccines. Among the fully vaccinated patients, 17.7% in the ivermectin group and 9.2% in the control group developed severe disease (RR, 1.92; 95% CI, 0.99-3.71; P = .06).
Ivermectin, an inexpensive and widely available antiparasitic drug, is prescribed to treat COVID-19 but has not been approved by the U.S. Food and Drug Administration for that purpose. Evidence-based data for or against use has been sparse.
The authors write that “although some early clinical studies suggested the potential efficacy of ivermectin in the treatment and prevention of COVID-19, these studies had methodologic weaknesses.”
Dr. Lim and colleagues point out that their findings are consistent with those of the IVERCOR-COVID19 trial, which found ivermectin ineffective in reducing hospitalization risk.
Previous randomized trials of ivermectin for COVID-19 patients that have included at least 400 patients have focused on outpatients.
In the current study, the authors note, patients were hospitalized, which allowed investigators to observe administration of ivermectin with a high adherence rate. Additionally, the researchers used clearly defined criteria for determining progression to severe disease.
Limitations of the current study include that the open-label design might lead to under-reporting of adverse events in the control group while overestimating the drug effects of ivermectin. The study was also not designed to assess the effects of ivermectin on mortality from COVID-19.
A version of this article first appeared on Medscape.com.
Ivermectin treatment given to high-risk patients with mild-to-moderate COVID-19 during the first week of illness did not prevent progression to severe disease, according to results from a randomized clinical trial.
“The study findings do not support the use of ivermectin for patients with COVID-19,” researchers conclude in the paper published online in JAMA Internal Medicine.
The open-label trial was conducted at 20 public hospitals and a COVID-19 quarantine center in Malaysia between May 31 and Oct. 25, 2021. It was led by Steven Chee Loon Lim, MRCP, department of medicine, Raja Permaisuri Bainun Hospital, Perak, Malaysia.
Among 490 patients in the primary analysis, 52 of 241 patients (21.6%) in the ivermectin group and 43 of 249 patients (17.3%) in the control group progressed to severe disease (relative risk, 1.25; 95% confidence interval, 0.87-1.80; P = .25). All major ethnic groups in Malaysia were well represented, the researchers write.
Participants (average age 62.5 and 54.5% women) were randomly assigned 1:1 to receive either a 5-day course of oral ivermectin (0.4 mg/kg body weight daily for 5 days) plus standard of care (n = 241) or standard of care alone (n = 249). Standard of care included symptomatic therapy and monitoring for early deterioration based on clinical findings, laboratory tests, and chest imaging.
Secondary outcomes
Secondary outcomes included rates of mechanical ventilation, intensive care unit (ICU) admission, 28-day in-hospital mortality, and side effects.
In all the secondary outcomes, there were no significant differences between groups.
Mechanical ventilation occurred in four patients on the ivermectin protocol (1.7%) versus 10 patients in the control group (4.0%) (RR, 0.41; 95% CI, 0.13-1.30; P = .17); ICU admission occurred in six (2.4%) versus eight (3.2%) (RR, 0.78; 95% CI, 0.27-2.20; P = .79); and 28-day in-hospital death occurred in three (1.2%) versus 10 (4.0%) (RR, 0.31; 95% CI, 0.09-1.11; P = .09).
The most common adverse event was diarrhea, reported by 5.8% in the ivermectin group and 1.6% in the control group.
No difference by vaccine status
The researchers conducted a subgroup analysis to evaluate any differences in whether participants were vaccinated. They said that analysis was “unremarkable.”
Just more than half of participants (51.8%) were fully vaccinated, with two doses of COVID-19 vaccines. Among the fully vaccinated patients, 17.7% in the ivermectin group and 9.2% in the control group developed severe disease (RR, 1.92; 95% CI, 0.99-3.71; P = .06).
Ivermectin, an inexpensive and widely available antiparasitic drug, is prescribed to treat COVID-19 but has not been approved by the U.S. Food and Drug Administration for that purpose. Evidence-based data for or against use has been sparse.
The authors write that “although some early clinical studies suggested the potential efficacy of ivermectin in the treatment and prevention of COVID-19, these studies had methodologic weaknesses.”
Dr. Lim and colleagues point out that their findings are consistent with those of the IVERCOR-COVID19 trial, which found ivermectin ineffective in reducing hospitalization risk.
Previous randomized trials of ivermectin for COVID-19 patients that have included at least 400 patients have focused on outpatients.
In the current study, the authors note, patients were hospitalized, which allowed investigators to observe administration of ivermectin with a high adherence rate. Additionally, the researchers used clearly defined criteria for determining progression to severe disease.
Limitations of the current study include that the open-label design might lead to under-reporting of adverse events in the control group while overestimating the drug effects of ivermectin. The study was also not designed to assess the effects of ivermectin on mortality from COVID-19.
A version of this article first appeared on Medscape.com.
FROM JAMA INTERNAL MEDICNE
Early in career, female academic docs earn less than males: study
Worse still, the earning potential of women in most specialties is $214,440 (or 10%) less than their male colleagues over the course of the first 10 years of their careers in academic medicine.
Among the vast majority of subspecialties, women’s starting salaries and their salaries 10 years into their careers were lower than their male colleagues in academic medicine, per the study in JAMA Network Open.
Eva Catenaccio, MD, an epilepsy fellow at Children’s Hospital of Philadelphia and the lead author of the study, told this news organization that the gender disparities in earning potential are “pervasive in academic medicine.” These earnings disparities, which occur in nearly all subspecialties and can reach hundreds of thousands of dollars in the first 10 years of an academic physician’s career, “are largely the result of gender differences in annual salary that start immediately after training,” she said.
Changing the timing of academic promotion and equalizing starting salary and salary growth can help close the salary gap, said Dr. Catenaccio.
The study also reveals that women could face a 1-year delay in promotion from assistant to associate professor, compared with men. This delay could reduce female physicians’ earning potential by a 10-year median of $26,042 (or 2%), whereas failure to be promoted at all could decrease the 10-year earning potential by a median of $218,724 (or 13%).
Across medicine more broadly, male physicians continue to earn 35% more than their female colleagues, according to the 2021 Medscape Physician Compensation Report. The biggest differences in take-home pay exist between male and female specialists, per the report. On average, male physicians earn $376,000, while women’s take-home pay is $283,000.
Medical schools and hospital leaders have a role to play
The earning potential during the first 10 years of post-training employment by gender was the most dramatic in neurosurgery, orthopedic surgery, and cardiology, per the study. Three subspecialties where women and men have similar earning potential include pediatric nephrology, pediatric neurology, and pediatric rheumatology.
The coauthors note that it’s commonly understood that women don’t negotiate as often or as successfully as their male colleagues. A 2019 study in JAMA Surgery of 606 male and female surgery residents revealed that while residents of both genders shared similar career goals, women had lower future salary expectations and a significantly more negative view of the salary negotiation process.
Dr. Catenaccio and her coauthors acknowledge that negotiation skills and financial literacy should be taught during medical school and postgraduate training. “However, the onus for ensuring salary equity should not fall on the individual candidate alone; rather, departmental and hospital leadership should take responsibility to ensure uniform starting salaries and prevent gender-based inequalities,” they wrote in the study.
“We hope that this study encourages academic medical institutions to increase transparency and equity around compensation, particularly for junior faculty,” Dr. Catenaccio said in an interview. “This will require both ensuring equal starting salaries and providing periodic adjustments throughout individuals’ careers to prevent divergence in earning potential by gender or any other individual characteristics.”
Harold Simon, MD, MBA, vice chair for faculty for the department of pediatrics and professor of pediatrics and emergency medicine at Emory University, Atlanta, told this news organization that “[i]ncreased transparency around compensation can enable women to advocate for equitable pay. However, the burden for ensuring equity should not fall on individuals but instead must be the primary responsibility of academic institutions.”
Specifically, Dr. Simon advocates for hospital leaders to “ensure equity among providers including compensation [as] a crucial part of maintaining a diverse workforce and, ultimately, providing balanced access to health care for patients.”
In addition, the authors call for periodic compensation evaluations and adjustments to help prevent gender-based salary differences among female and male physicians in academia. “This is absolutely necessary, both to develop future compensation plans and to address any pre-existing gender-based salary inequities for those women currently well into their careers,” they wrote in the study.
Data analysis was conducted from March to May 2021. Researchers used models to estimate the impacts of promotion timing and potential interventions, which include equalizing starting salaries and annual salary rates.
The study included compensation data for 24,593 female and 29,886 male academic physicians across 45 subspecialties. It relied on publicly available data from the Association of American Medical Colleges’ annual Medical School Faculty Salary Survey report.
A version of this article first appeared on Medscape.com.
Worse still, the earning potential of women in most specialties is $214,440 (or 10%) less than their male colleagues over the course of the first 10 years of their careers in academic medicine.
Among the vast majority of subspecialties, women’s starting salaries and their salaries 10 years into their careers were lower than their male colleagues in academic medicine, per the study in JAMA Network Open.
Eva Catenaccio, MD, an epilepsy fellow at Children’s Hospital of Philadelphia and the lead author of the study, told this news organization that the gender disparities in earning potential are “pervasive in academic medicine.” These earnings disparities, which occur in nearly all subspecialties and can reach hundreds of thousands of dollars in the first 10 years of an academic physician’s career, “are largely the result of gender differences in annual salary that start immediately after training,” she said.
Changing the timing of academic promotion and equalizing starting salary and salary growth can help close the salary gap, said Dr. Catenaccio.
The study also reveals that women could face a 1-year delay in promotion from assistant to associate professor, compared with men. This delay could reduce female physicians’ earning potential by a 10-year median of $26,042 (or 2%), whereas failure to be promoted at all could decrease the 10-year earning potential by a median of $218,724 (or 13%).
Across medicine more broadly, male physicians continue to earn 35% more than their female colleagues, according to the 2021 Medscape Physician Compensation Report. The biggest differences in take-home pay exist between male and female specialists, per the report. On average, male physicians earn $376,000, while women’s take-home pay is $283,000.
Medical schools and hospital leaders have a role to play
The earning potential during the first 10 years of post-training employment by gender was the most dramatic in neurosurgery, orthopedic surgery, and cardiology, per the study. Three subspecialties where women and men have similar earning potential include pediatric nephrology, pediatric neurology, and pediatric rheumatology.
The coauthors note that it’s commonly understood that women don’t negotiate as often or as successfully as their male colleagues. A 2019 study in JAMA Surgery of 606 male and female surgery residents revealed that while residents of both genders shared similar career goals, women had lower future salary expectations and a significantly more negative view of the salary negotiation process.
Dr. Catenaccio and her coauthors acknowledge that negotiation skills and financial literacy should be taught during medical school and postgraduate training. “However, the onus for ensuring salary equity should not fall on the individual candidate alone; rather, departmental and hospital leadership should take responsibility to ensure uniform starting salaries and prevent gender-based inequalities,” they wrote in the study.
“We hope that this study encourages academic medical institutions to increase transparency and equity around compensation, particularly for junior faculty,” Dr. Catenaccio said in an interview. “This will require both ensuring equal starting salaries and providing periodic adjustments throughout individuals’ careers to prevent divergence in earning potential by gender or any other individual characteristics.”
Harold Simon, MD, MBA, vice chair for faculty for the department of pediatrics and professor of pediatrics and emergency medicine at Emory University, Atlanta, told this news organization that “[i]ncreased transparency around compensation can enable women to advocate for equitable pay. However, the burden for ensuring equity should not fall on individuals but instead must be the primary responsibility of academic institutions.”
Specifically, Dr. Simon advocates for hospital leaders to “ensure equity among providers including compensation [as] a crucial part of maintaining a diverse workforce and, ultimately, providing balanced access to health care for patients.”
In addition, the authors call for periodic compensation evaluations and adjustments to help prevent gender-based salary differences among female and male physicians in academia. “This is absolutely necessary, both to develop future compensation plans and to address any pre-existing gender-based salary inequities for those women currently well into their careers,” they wrote in the study.
Data analysis was conducted from March to May 2021. Researchers used models to estimate the impacts of promotion timing and potential interventions, which include equalizing starting salaries and annual salary rates.
The study included compensation data for 24,593 female and 29,886 male academic physicians across 45 subspecialties. It relied on publicly available data from the Association of American Medical Colleges’ annual Medical School Faculty Salary Survey report.
A version of this article first appeared on Medscape.com.
Worse still, the earning potential of women in most specialties is $214,440 (or 10%) less than their male colleagues over the course of the first 10 years of their careers in academic medicine.
Among the vast majority of subspecialties, women’s starting salaries and their salaries 10 years into their careers were lower than their male colleagues in academic medicine, per the study in JAMA Network Open.
Eva Catenaccio, MD, an epilepsy fellow at Children’s Hospital of Philadelphia and the lead author of the study, told this news organization that the gender disparities in earning potential are “pervasive in academic medicine.” These earnings disparities, which occur in nearly all subspecialties and can reach hundreds of thousands of dollars in the first 10 years of an academic physician’s career, “are largely the result of gender differences in annual salary that start immediately after training,” she said.
Changing the timing of academic promotion and equalizing starting salary and salary growth can help close the salary gap, said Dr. Catenaccio.
The study also reveals that women could face a 1-year delay in promotion from assistant to associate professor, compared with men. This delay could reduce female physicians’ earning potential by a 10-year median of $26,042 (or 2%), whereas failure to be promoted at all could decrease the 10-year earning potential by a median of $218,724 (or 13%).
Across medicine more broadly, male physicians continue to earn 35% more than their female colleagues, according to the 2021 Medscape Physician Compensation Report. The biggest differences in take-home pay exist between male and female specialists, per the report. On average, male physicians earn $376,000, while women’s take-home pay is $283,000.
Medical schools and hospital leaders have a role to play
The earning potential during the first 10 years of post-training employment by gender was the most dramatic in neurosurgery, orthopedic surgery, and cardiology, per the study. Three subspecialties where women and men have similar earning potential include pediatric nephrology, pediatric neurology, and pediatric rheumatology.
The coauthors note that it’s commonly understood that women don’t negotiate as often or as successfully as their male colleagues. A 2019 study in JAMA Surgery of 606 male and female surgery residents revealed that while residents of both genders shared similar career goals, women had lower future salary expectations and a significantly more negative view of the salary negotiation process.
Dr. Catenaccio and her coauthors acknowledge that negotiation skills and financial literacy should be taught during medical school and postgraduate training. “However, the onus for ensuring salary equity should not fall on the individual candidate alone; rather, departmental and hospital leadership should take responsibility to ensure uniform starting salaries and prevent gender-based inequalities,” they wrote in the study.
“We hope that this study encourages academic medical institutions to increase transparency and equity around compensation, particularly for junior faculty,” Dr. Catenaccio said in an interview. “This will require both ensuring equal starting salaries and providing periodic adjustments throughout individuals’ careers to prevent divergence in earning potential by gender or any other individual characteristics.”
Harold Simon, MD, MBA, vice chair for faculty for the department of pediatrics and professor of pediatrics and emergency medicine at Emory University, Atlanta, told this news organization that “[i]ncreased transparency around compensation can enable women to advocate for equitable pay. However, the burden for ensuring equity should not fall on individuals but instead must be the primary responsibility of academic institutions.”
Specifically, Dr. Simon advocates for hospital leaders to “ensure equity among providers including compensation [as] a crucial part of maintaining a diverse workforce and, ultimately, providing balanced access to health care for patients.”
In addition, the authors call for periodic compensation evaluations and adjustments to help prevent gender-based salary differences among female and male physicians in academia. “This is absolutely necessary, both to develop future compensation plans and to address any pre-existing gender-based salary inequities for those women currently well into their careers,” they wrote in the study.
Data analysis was conducted from March to May 2021. Researchers used models to estimate the impacts of promotion timing and potential interventions, which include equalizing starting salaries and annual salary rates.
The study included compensation data for 24,593 female and 29,886 male academic physicians across 45 subspecialties. It relied on publicly available data from the Association of American Medical Colleges’ annual Medical School Faculty Salary Survey report.
A version of this article first appeared on Medscape.com.
HIV: Dual therapy with twice-yearly injections on the horizon
The findings, presented at the Conference on Retroviruses and Opportunistic Infections, also showed the drug was well tolerated, with 2 of 182 people developing drug-resistant mutations to lenacapavir and one person developing a nodule at the injection site.
Laura Waters, MD, consulting physician in HIV and sexual health at Central and Northwest London NHS Trust, who was not involved in the trial, called the findings “hugely exciting,” especially given its unique mode of administration. Right now it’s formulated as a subcutaneous injection, not an intramuscular injection like cabotegravir and rilpivirine (Cabenuva, ViiV Healthcare).
“Clearly it’s incredibly exciting to have the option for a subcutaneous drug that could be given at home every 6 months,” Dr. Waters said in an interview. With phase 3 trials and long-term follow-up still to come, she said she’s looking forward to how the treatment evolves.
CALIBRATE is a phase 2, four-arm, open-label, active-control study. Of the 182 participants, 25 were randomized to the active control arm, in which participants took daily oral bictegravir, emtricitabine, and tenofovir alafenamide (Biktarvy, Gilead Sciences) as standard of care. The rest of the 182 participants were divided evenly between three arms. In one of those, treatment group three, participants took a daily oral version of 50-mg LEN with emtricitabine and tenofovir alafenamide (F/TAF, Descovy, Gilead Sciences).
The other two arms, treatment groups one and two, were the arms in which Samir K. Gupta, MD, of Indiana University, Indianapolis, and colleagues tried the twice-yearly subcutaneous shots of LEN (LEN SC). Participants in those arms underwent a 2-week lead-in period in which they took 600 mg of LEN orally on days 1 and 2 and then a 300-mg pill on day 8 before starting the shots on day 15. As currently formulated, LEN SC shots can be given into the stomach and are designed to be simple enough to administer at home.
“It’s like doing an insulin shot,” Dr. Gupta said in an interview.
In addition to LEN SC, participants in treatment group one took a daily Descovy lead-in and then engaged in dual therapy of LEN plus TAF. In group two, they took a daily Descovy lead-in and then switched to LEN SC plus bictegravir (BIC) daily at 28 weeks.
Study results presented at the meeting were the results at 54 weeks. Researchers had previously presented 28-week results for CALIBRATE at the International AIDS Society Conference on HIV Science 2021.
Of the participants, 7% were cisgender women, 52% were Black, and 45% were Latinx. A total of 15% of participants had baseline viral loads of more than 100,000 copies, and median CD4 counts were 437. None of the participants in either the active-control arm or the LEN SC plus Descovy group had a history of AIDS, defined as fewer than 200 cells/mcL. In the LEN SC plus Biktarvy group, two did, and there were six people with a history of AIDS in the oral LEN plus Descovy group.
At 54 weeks, 88% of participants in groups one and two – the LEN SC arms – had undetectable viral loads (viral loads below 50 copies/mL). Specifically, 90% of those in the LEN SC plus TAF arm had viral loads below 50 copies. Those taking LEN SC plus BIC had an 85% viral suppression rate. The best performing of all was the control arm, in which 92% had undetectable viral loads. The protocol didn’t allow for analysis of statistical significance between the arms, so it’s unclear if any of the treatments really surpassed the others, Dr. Gupta said.
When they looked only at people whose viral loads became undetectable early, by week 28, the results were slightly better, with 93% of people between the two subcutaneous arms having undetectable viral loads at week 54. Again, the results were slightly higher in the LEN SC plus TAF arm (94%) than the LEN SC plus BIC (92%). People taking daily oral LEN plus Descovy had the lowest rate of viral suppression, but it was still at 90% if they were virally suppressed early. Again, it’s unclear whether those differences were statistically significant.
Well tolerated, with a chance of a nodule
Regarding safety, LEN was well tolerated across treatment arms, though the oral LEN group had as many people develop resistance mutations as the other three groups combined. Still, that was only three people, with one each in the two subcutaneous arms and one in the control arm.
Two people developed resistance to LEN – one in the LEN SC plus BIC arm and one in the oral LEN plus Descovy arm. In the LEN SC plus BIC arm, the mutations Q67H and K70R emerged at week 10, following a mutation that’s common when people don’t take integrase inhibitors as they’re supposed to (M184M/I). This led Dr. Gupta to conclude that lack of adherence to the oral part of the regimen may have contributed to the development of resistance, rather than an issue with LEN SC itself.
In the oral LEN plus Descovy arm, mutation Q67H didn’t emerge until week 54, when pill counts and drug levels revealed that that participant hadn’t been taking his Descovy as prescribed. In both cases, viral loads in those participants returned to undetectable after switching to an integrase inhibitor–based three-drug regimen.
Most safety concerns were mild and included nausea, diarrhea, and vomiting. But that was for non–injection-site reactions. There were more side effects of that sort, with most of them being pain, hardening of the site, and swelling. But 11% of participants developed more serious side effects, including nodules. Only one nodule was considered a grade 3 reaction; that person didn’t leave the trial because of it.
“These phase 2 data from the ongoing CALIBRATE trial support the further evaluation of lenacapavir for treatment and prevention of HIV-1,” he said.
Finding the right partner
In short, the findings are promising, Dr. Gupta told this news organization . But the question now, as Gilead prepares to begin a phase 3 efficacy trial, is what would be a good combination with LEN SC?
“Lenacapavir in a two-drug regimen should work,” he said. “The question now, though, is you have to find a suitable, potent, second agent to pair it with.”
Indeed, the long-acting HIV treatment pipeline sustained a blow in December 2021 when the U.S. Food and Drug Administration halted all trials for Merck’s investigational, long-acting drug islatravir. This led Dr. Waters to tweet: “Is it just me or are the islatravir abstracts at #CROI2022 quite sad to read?” She ended the tweet with, “I just hope it recovers!”
Indeed, Merck and Gilead have entered into an agreement to codevelop lenacapavir and islatravir as a weekly oral tablet. Islatravir seemed like the obvious choice as a partner to lenacapavir if phase 3 trials are successful, said Dr. Gupta. But now, it’s anyone’s guess as to what will happen. And while it’s too soon to say that lenacapavir is a success, it does leave the field wondering about how to use LEN SC without another, equally long-acting agent.
“When I talk about LEN, I show a picture of a man standing solo at a party, leaning against the wall looking a bit lonely, with ‘lenacapavir’ written over his head,” she said in an interview. “Right now, lenacapavir is the only drug at the 6-monthly party. It’s going to need some other guests if that party is going to rock.”
The study was funded by Gilead Sciences. Dr. Gupta has received research funding from ViiV Healthcare and advisory board fees from Gilead Sciences and ViiV Healthcare. Dr. Waters has received speaker or advisory fees from Gilead Sciences, ViiV Healthcare, Merck, Janssen, Theratech, Sipla, and Mylan.
A version of this article first appeared on Medscape.com.
The findings, presented at the Conference on Retroviruses and Opportunistic Infections, also showed the drug was well tolerated, with 2 of 182 people developing drug-resistant mutations to lenacapavir and one person developing a nodule at the injection site.
Laura Waters, MD, consulting physician in HIV and sexual health at Central and Northwest London NHS Trust, who was not involved in the trial, called the findings “hugely exciting,” especially given its unique mode of administration. Right now it’s formulated as a subcutaneous injection, not an intramuscular injection like cabotegravir and rilpivirine (Cabenuva, ViiV Healthcare).
“Clearly it’s incredibly exciting to have the option for a subcutaneous drug that could be given at home every 6 months,” Dr. Waters said in an interview. With phase 3 trials and long-term follow-up still to come, she said she’s looking forward to how the treatment evolves.
CALIBRATE is a phase 2, four-arm, open-label, active-control study. Of the 182 participants, 25 were randomized to the active control arm, in which participants took daily oral bictegravir, emtricitabine, and tenofovir alafenamide (Biktarvy, Gilead Sciences) as standard of care. The rest of the 182 participants were divided evenly between three arms. In one of those, treatment group three, participants took a daily oral version of 50-mg LEN with emtricitabine and tenofovir alafenamide (F/TAF, Descovy, Gilead Sciences).
The other two arms, treatment groups one and two, were the arms in which Samir K. Gupta, MD, of Indiana University, Indianapolis, and colleagues tried the twice-yearly subcutaneous shots of LEN (LEN SC). Participants in those arms underwent a 2-week lead-in period in which they took 600 mg of LEN orally on days 1 and 2 and then a 300-mg pill on day 8 before starting the shots on day 15. As currently formulated, LEN SC shots can be given into the stomach and are designed to be simple enough to administer at home.
“It’s like doing an insulin shot,” Dr. Gupta said in an interview.
In addition to LEN SC, participants in treatment group one took a daily Descovy lead-in and then engaged in dual therapy of LEN plus TAF. In group two, they took a daily Descovy lead-in and then switched to LEN SC plus bictegravir (BIC) daily at 28 weeks.
Study results presented at the meeting were the results at 54 weeks. Researchers had previously presented 28-week results for CALIBRATE at the International AIDS Society Conference on HIV Science 2021.
Of the participants, 7% were cisgender women, 52% were Black, and 45% were Latinx. A total of 15% of participants had baseline viral loads of more than 100,000 copies, and median CD4 counts were 437. None of the participants in either the active-control arm or the LEN SC plus Descovy group had a history of AIDS, defined as fewer than 200 cells/mcL. In the LEN SC plus Biktarvy group, two did, and there were six people with a history of AIDS in the oral LEN plus Descovy group.
At 54 weeks, 88% of participants in groups one and two – the LEN SC arms – had undetectable viral loads (viral loads below 50 copies/mL). Specifically, 90% of those in the LEN SC plus TAF arm had viral loads below 50 copies. Those taking LEN SC plus BIC had an 85% viral suppression rate. The best performing of all was the control arm, in which 92% had undetectable viral loads. The protocol didn’t allow for analysis of statistical significance between the arms, so it’s unclear if any of the treatments really surpassed the others, Dr. Gupta said.
When they looked only at people whose viral loads became undetectable early, by week 28, the results were slightly better, with 93% of people between the two subcutaneous arms having undetectable viral loads at week 54. Again, the results were slightly higher in the LEN SC plus TAF arm (94%) than the LEN SC plus BIC (92%). People taking daily oral LEN plus Descovy had the lowest rate of viral suppression, but it was still at 90% if they were virally suppressed early. Again, it’s unclear whether those differences were statistically significant.
Well tolerated, with a chance of a nodule
Regarding safety, LEN was well tolerated across treatment arms, though the oral LEN group had as many people develop resistance mutations as the other three groups combined. Still, that was only three people, with one each in the two subcutaneous arms and one in the control arm.
Two people developed resistance to LEN – one in the LEN SC plus BIC arm and one in the oral LEN plus Descovy arm. In the LEN SC plus BIC arm, the mutations Q67H and K70R emerged at week 10, following a mutation that’s common when people don’t take integrase inhibitors as they’re supposed to (M184M/I). This led Dr. Gupta to conclude that lack of adherence to the oral part of the regimen may have contributed to the development of resistance, rather than an issue with LEN SC itself.
In the oral LEN plus Descovy arm, mutation Q67H didn’t emerge until week 54, when pill counts and drug levels revealed that that participant hadn’t been taking his Descovy as prescribed. In both cases, viral loads in those participants returned to undetectable after switching to an integrase inhibitor–based three-drug regimen.
Most safety concerns were mild and included nausea, diarrhea, and vomiting. But that was for non–injection-site reactions. There were more side effects of that sort, with most of them being pain, hardening of the site, and swelling. But 11% of participants developed more serious side effects, including nodules. Only one nodule was considered a grade 3 reaction; that person didn’t leave the trial because of it.
“These phase 2 data from the ongoing CALIBRATE trial support the further evaluation of lenacapavir for treatment and prevention of HIV-1,” he said.
Finding the right partner
In short, the findings are promising, Dr. Gupta told this news organization . But the question now, as Gilead prepares to begin a phase 3 efficacy trial, is what would be a good combination with LEN SC?
“Lenacapavir in a two-drug regimen should work,” he said. “The question now, though, is you have to find a suitable, potent, second agent to pair it with.”
Indeed, the long-acting HIV treatment pipeline sustained a blow in December 2021 when the U.S. Food and Drug Administration halted all trials for Merck’s investigational, long-acting drug islatravir. This led Dr. Waters to tweet: “Is it just me or are the islatravir abstracts at #CROI2022 quite sad to read?” She ended the tweet with, “I just hope it recovers!”
Indeed, Merck and Gilead have entered into an agreement to codevelop lenacapavir and islatravir as a weekly oral tablet. Islatravir seemed like the obvious choice as a partner to lenacapavir if phase 3 trials are successful, said Dr. Gupta. But now, it’s anyone’s guess as to what will happen. And while it’s too soon to say that lenacapavir is a success, it does leave the field wondering about how to use LEN SC without another, equally long-acting agent.
“When I talk about LEN, I show a picture of a man standing solo at a party, leaning against the wall looking a bit lonely, with ‘lenacapavir’ written over his head,” she said in an interview. “Right now, lenacapavir is the only drug at the 6-monthly party. It’s going to need some other guests if that party is going to rock.”
The study was funded by Gilead Sciences. Dr. Gupta has received research funding from ViiV Healthcare and advisory board fees from Gilead Sciences and ViiV Healthcare. Dr. Waters has received speaker or advisory fees from Gilead Sciences, ViiV Healthcare, Merck, Janssen, Theratech, Sipla, and Mylan.
A version of this article first appeared on Medscape.com.
The findings, presented at the Conference on Retroviruses and Opportunistic Infections, also showed the drug was well tolerated, with 2 of 182 people developing drug-resistant mutations to lenacapavir and one person developing a nodule at the injection site.
Laura Waters, MD, consulting physician in HIV and sexual health at Central and Northwest London NHS Trust, who was not involved in the trial, called the findings “hugely exciting,” especially given its unique mode of administration. Right now it’s formulated as a subcutaneous injection, not an intramuscular injection like cabotegravir and rilpivirine (Cabenuva, ViiV Healthcare).
“Clearly it’s incredibly exciting to have the option for a subcutaneous drug that could be given at home every 6 months,” Dr. Waters said in an interview. With phase 3 trials and long-term follow-up still to come, she said she’s looking forward to how the treatment evolves.
CALIBRATE is a phase 2, four-arm, open-label, active-control study. Of the 182 participants, 25 were randomized to the active control arm, in which participants took daily oral bictegravir, emtricitabine, and tenofovir alafenamide (Biktarvy, Gilead Sciences) as standard of care. The rest of the 182 participants were divided evenly between three arms. In one of those, treatment group three, participants took a daily oral version of 50-mg LEN with emtricitabine and tenofovir alafenamide (F/TAF, Descovy, Gilead Sciences).
The other two arms, treatment groups one and two, were the arms in which Samir K. Gupta, MD, of Indiana University, Indianapolis, and colleagues tried the twice-yearly subcutaneous shots of LEN (LEN SC). Participants in those arms underwent a 2-week lead-in period in which they took 600 mg of LEN orally on days 1 and 2 and then a 300-mg pill on day 8 before starting the shots on day 15. As currently formulated, LEN SC shots can be given into the stomach and are designed to be simple enough to administer at home.
“It’s like doing an insulin shot,” Dr. Gupta said in an interview.
In addition to LEN SC, participants in treatment group one took a daily Descovy lead-in and then engaged in dual therapy of LEN plus TAF. In group two, they took a daily Descovy lead-in and then switched to LEN SC plus bictegravir (BIC) daily at 28 weeks.
Study results presented at the meeting were the results at 54 weeks. Researchers had previously presented 28-week results for CALIBRATE at the International AIDS Society Conference on HIV Science 2021.
Of the participants, 7% were cisgender women, 52% were Black, and 45% were Latinx. A total of 15% of participants had baseline viral loads of more than 100,000 copies, and median CD4 counts were 437. None of the participants in either the active-control arm or the LEN SC plus Descovy group had a history of AIDS, defined as fewer than 200 cells/mcL. In the LEN SC plus Biktarvy group, two did, and there were six people with a history of AIDS in the oral LEN plus Descovy group.
At 54 weeks, 88% of participants in groups one and two – the LEN SC arms – had undetectable viral loads (viral loads below 50 copies/mL). Specifically, 90% of those in the LEN SC plus TAF arm had viral loads below 50 copies. Those taking LEN SC plus BIC had an 85% viral suppression rate. The best performing of all was the control arm, in which 92% had undetectable viral loads. The protocol didn’t allow for analysis of statistical significance between the arms, so it’s unclear if any of the treatments really surpassed the others, Dr. Gupta said.
When they looked only at people whose viral loads became undetectable early, by week 28, the results were slightly better, with 93% of people between the two subcutaneous arms having undetectable viral loads at week 54. Again, the results were slightly higher in the LEN SC plus TAF arm (94%) than the LEN SC plus BIC (92%). People taking daily oral LEN plus Descovy had the lowest rate of viral suppression, but it was still at 90% if they were virally suppressed early. Again, it’s unclear whether those differences were statistically significant.
Well tolerated, with a chance of a nodule
Regarding safety, LEN was well tolerated across treatment arms, though the oral LEN group had as many people develop resistance mutations as the other three groups combined. Still, that was only three people, with one each in the two subcutaneous arms and one in the control arm.
Two people developed resistance to LEN – one in the LEN SC plus BIC arm and one in the oral LEN plus Descovy arm. In the LEN SC plus BIC arm, the mutations Q67H and K70R emerged at week 10, following a mutation that’s common when people don’t take integrase inhibitors as they’re supposed to (M184M/I). This led Dr. Gupta to conclude that lack of adherence to the oral part of the regimen may have contributed to the development of resistance, rather than an issue with LEN SC itself.
In the oral LEN plus Descovy arm, mutation Q67H didn’t emerge until week 54, when pill counts and drug levels revealed that that participant hadn’t been taking his Descovy as prescribed. In both cases, viral loads in those participants returned to undetectable after switching to an integrase inhibitor–based three-drug regimen.
Most safety concerns were mild and included nausea, diarrhea, and vomiting. But that was for non–injection-site reactions. There were more side effects of that sort, with most of them being pain, hardening of the site, and swelling. But 11% of participants developed more serious side effects, including nodules. Only one nodule was considered a grade 3 reaction; that person didn’t leave the trial because of it.
“These phase 2 data from the ongoing CALIBRATE trial support the further evaluation of lenacapavir for treatment and prevention of HIV-1,” he said.
Finding the right partner
In short, the findings are promising, Dr. Gupta told this news organization . But the question now, as Gilead prepares to begin a phase 3 efficacy trial, is what would be a good combination with LEN SC?
“Lenacapavir in a two-drug regimen should work,” he said. “The question now, though, is you have to find a suitable, potent, second agent to pair it with.”
Indeed, the long-acting HIV treatment pipeline sustained a blow in December 2021 when the U.S. Food and Drug Administration halted all trials for Merck’s investigational, long-acting drug islatravir. This led Dr. Waters to tweet: “Is it just me or are the islatravir abstracts at #CROI2022 quite sad to read?” She ended the tweet with, “I just hope it recovers!”
Indeed, Merck and Gilead have entered into an agreement to codevelop lenacapavir and islatravir as a weekly oral tablet. Islatravir seemed like the obvious choice as a partner to lenacapavir if phase 3 trials are successful, said Dr. Gupta. But now, it’s anyone’s guess as to what will happen. And while it’s too soon to say that lenacapavir is a success, it does leave the field wondering about how to use LEN SC without another, equally long-acting agent.
“When I talk about LEN, I show a picture of a man standing solo at a party, leaning against the wall looking a bit lonely, with ‘lenacapavir’ written over his head,” she said in an interview. “Right now, lenacapavir is the only drug at the 6-monthly party. It’s going to need some other guests if that party is going to rock.”
The study was funded by Gilead Sciences. Dr. Gupta has received research funding from ViiV Healthcare and advisory board fees from Gilead Sciences and ViiV Healthcare. Dr. Waters has received speaker or advisory fees from Gilead Sciences, ViiV Healthcare, Merck, Janssen, Theratech, Sipla, and Mylan.
A version of this article first appeared on Medscape.com.
FROM CROI 22
New ivermectin, HCQ scripts highest in GOP-dominated counties
New prescriptions of hydroxychloroquine (HCQ) and ivermectin increased in 2020, driven particularly by rates in counties with the highest proportion of Republican votes in the 2020 U.S. presidential election, according to a cross-sectional study published in JAMA Internal Medicine.
“Our findings are consistent with the hypothesis that U.S. prescribing of hydroxychloroquine and ivermectin during the COVID-19 pandemic may have been influenced by political affiliation,” wrote Michael L. Barnett, MD, of the Harvard T.H. Chan School of Public Health in Boston and colleagues.
The researchers used data from the OptumLabs Data Warehouse to analyze commercial and Medicare Advantage medical claims from January 2019 through December 2020 for more than 18.5 million adults living in counties with at least 50 enrollees.
Using U.S. Census data and 2020 presidential election results, the researchers classified counties according to their proportion of Republican voters and then examined whether those proportions were associated with that county’s rates of new prescriptions for HCQ, ivermectin, methotrexate sodium, and albendazole. Methotrexate is prescribed for similar conditions and indications as HCQ, and albendazole is prescribed for similar reasons as ivermectin, although neither of the comparison drugs has been considered for COVID-19 treatment.
The Food and Drug Administration issued an emergency use authorization (EUA) for HCQ as a COVID-19 treatment on March 28, 2020, but the agency revoked the EUA 3 months later on June 15. Ivermectin never received an EUA for COVID treatment, but an in vitro study published April 3, 2020 claimed it had an antiviral effect.
The National Institutes of Health recommended against using ivermectin as a COVID-19 treatment on Aug. 1, 2020, but a few months later, on Nov. 13, a flawed clinical trial – later retracted – claimed ivermectin was 90% effective in treating COVID-19. Despite the lack of evidence for ivermectin’s efficacy, a Senate committee meeting on Dec. 8, 2020, included testimony from a physician who promoted its use.
In comparing ivermectin and HCQ prescription rates with counties’ political composition, the researchers adjusted their findings to account for differences in the counties’ racial composition and COVID-19 incidence as well as enrollees’ age, sex, insurance type, income, comorbidity burden, and home in a rural or urban area.
The results showed an average of 20 new HCQ prescriptions per 100,000 enrollees in 2019, but 2020 saw a sharp increase and drop in new HCQ prescriptions in March-April 2020, independent of counties’ breakdown of political affiliation.
“However, after June 2020, coinciding with the revocation of the U.S. Food and Drug Administration’s emergency use authorization for hydroxychloroquine, prescribing volume was significantly higher in the highest vs. lowest Republican vote share counties,” the authors report. The gradual increase from June through December 2020 averaged to 42 new prescriptions per 100,000, a 146% increase over 2019 rates that was driven largely by the 25% of counties with the highest proportion of Republican voters.
Similarly, rates of new ivermectin prescriptions in December 2020 were more than nine times higher in counties with the highest Republican vote share, compared with new prescriptions throughout 2019. The researchers found no differences in new prescriptions for methotrexate or albendazole in 2020 based on counties’ proportion of Republican votes.
Since the study is an ecological, observational one, it cannot show causation or shed light on what role patients, physicians, or other factors might have played in prescribing patterns. Nevertheless, the authors noted the potentially negative implications of their findings.
“Because political affiliation should not be a factor in clinical treatment decisions, our findings raise concerns for public trust in a nonpartisan health care system,” the authors write.
Coauthor Ateev Mehrotra, MD, MPH, reported personal fees from Sanofi-Aventis, and coauthor Anupam B. Jena, MD, PhD, reported personal fees from Bioverativ, Merck, Janssen, Edwards Lifesciences, Novartis, Amgen, Eisai, Otsuka, Vertex, Celgene, Sanofi-Aventis, Precision Health Economics (now PRECISIONheor), Analysis Group, and Doubleday and hosting the podcast Freakonomics, M.D. The other coauthors have disclosed no relevant financial relationships. No external funding source was noted.
A version of this article first appeared on Medscape.com.
New prescriptions of hydroxychloroquine (HCQ) and ivermectin increased in 2020, driven particularly by rates in counties with the highest proportion of Republican votes in the 2020 U.S. presidential election, according to a cross-sectional study published in JAMA Internal Medicine.
“Our findings are consistent with the hypothesis that U.S. prescribing of hydroxychloroquine and ivermectin during the COVID-19 pandemic may have been influenced by political affiliation,” wrote Michael L. Barnett, MD, of the Harvard T.H. Chan School of Public Health in Boston and colleagues.
The researchers used data from the OptumLabs Data Warehouse to analyze commercial and Medicare Advantage medical claims from January 2019 through December 2020 for more than 18.5 million adults living in counties with at least 50 enrollees.
Using U.S. Census data and 2020 presidential election results, the researchers classified counties according to their proportion of Republican voters and then examined whether those proportions were associated with that county’s rates of new prescriptions for HCQ, ivermectin, methotrexate sodium, and albendazole. Methotrexate is prescribed for similar conditions and indications as HCQ, and albendazole is prescribed for similar reasons as ivermectin, although neither of the comparison drugs has been considered for COVID-19 treatment.
The Food and Drug Administration issued an emergency use authorization (EUA) for HCQ as a COVID-19 treatment on March 28, 2020, but the agency revoked the EUA 3 months later on June 15. Ivermectin never received an EUA for COVID treatment, but an in vitro study published April 3, 2020 claimed it had an antiviral effect.
The National Institutes of Health recommended against using ivermectin as a COVID-19 treatment on Aug. 1, 2020, but a few months later, on Nov. 13, a flawed clinical trial – later retracted – claimed ivermectin was 90% effective in treating COVID-19. Despite the lack of evidence for ivermectin’s efficacy, a Senate committee meeting on Dec. 8, 2020, included testimony from a physician who promoted its use.
In comparing ivermectin and HCQ prescription rates with counties’ political composition, the researchers adjusted their findings to account for differences in the counties’ racial composition and COVID-19 incidence as well as enrollees’ age, sex, insurance type, income, comorbidity burden, and home in a rural or urban area.
The results showed an average of 20 new HCQ prescriptions per 100,000 enrollees in 2019, but 2020 saw a sharp increase and drop in new HCQ prescriptions in March-April 2020, independent of counties’ breakdown of political affiliation.
“However, after June 2020, coinciding with the revocation of the U.S. Food and Drug Administration’s emergency use authorization for hydroxychloroquine, prescribing volume was significantly higher in the highest vs. lowest Republican vote share counties,” the authors report. The gradual increase from June through December 2020 averaged to 42 new prescriptions per 100,000, a 146% increase over 2019 rates that was driven largely by the 25% of counties with the highest proportion of Republican voters.
Similarly, rates of new ivermectin prescriptions in December 2020 were more than nine times higher in counties with the highest Republican vote share, compared with new prescriptions throughout 2019. The researchers found no differences in new prescriptions for methotrexate or albendazole in 2020 based on counties’ proportion of Republican votes.
Since the study is an ecological, observational one, it cannot show causation or shed light on what role patients, physicians, or other factors might have played in prescribing patterns. Nevertheless, the authors noted the potentially negative implications of their findings.
“Because political affiliation should not be a factor in clinical treatment decisions, our findings raise concerns for public trust in a nonpartisan health care system,” the authors write.
Coauthor Ateev Mehrotra, MD, MPH, reported personal fees from Sanofi-Aventis, and coauthor Anupam B. Jena, MD, PhD, reported personal fees from Bioverativ, Merck, Janssen, Edwards Lifesciences, Novartis, Amgen, Eisai, Otsuka, Vertex, Celgene, Sanofi-Aventis, Precision Health Economics (now PRECISIONheor), Analysis Group, and Doubleday and hosting the podcast Freakonomics, M.D. The other coauthors have disclosed no relevant financial relationships. No external funding source was noted.
A version of this article first appeared on Medscape.com.
New prescriptions of hydroxychloroquine (HCQ) and ivermectin increased in 2020, driven particularly by rates in counties with the highest proportion of Republican votes in the 2020 U.S. presidential election, according to a cross-sectional study published in JAMA Internal Medicine.
“Our findings are consistent with the hypothesis that U.S. prescribing of hydroxychloroquine and ivermectin during the COVID-19 pandemic may have been influenced by political affiliation,” wrote Michael L. Barnett, MD, of the Harvard T.H. Chan School of Public Health in Boston and colleagues.
The researchers used data from the OptumLabs Data Warehouse to analyze commercial and Medicare Advantage medical claims from January 2019 through December 2020 for more than 18.5 million adults living in counties with at least 50 enrollees.
Using U.S. Census data and 2020 presidential election results, the researchers classified counties according to their proportion of Republican voters and then examined whether those proportions were associated with that county’s rates of new prescriptions for HCQ, ivermectin, methotrexate sodium, and albendazole. Methotrexate is prescribed for similar conditions and indications as HCQ, and albendazole is prescribed for similar reasons as ivermectin, although neither of the comparison drugs has been considered for COVID-19 treatment.
The Food and Drug Administration issued an emergency use authorization (EUA) for HCQ as a COVID-19 treatment on March 28, 2020, but the agency revoked the EUA 3 months later on June 15. Ivermectin never received an EUA for COVID treatment, but an in vitro study published April 3, 2020 claimed it had an antiviral effect.
The National Institutes of Health recommended against using ivermectin as a COVID-19 treatment on Aug. 1, 2020, but a few months later, on Nov. 13, a flawed clinical trial – later retracted – claimed ivermectin was 90% effective in treating COVID-19. Despite the lack of evidence for ivermectin’s efficacy, a Senate committee meeting on Dec. 8, 2020, included testimony from a physician who promoted its use.
In comparing ivermectin and HCQ prescription rates with counties’ political composition, the researchers adjusted their findings to account for differences in the counties’ racial composition and COVID-19 incidence as well as enrollees’ age, sex, insurance type, income, comorbidity burden, and home in a rural or urban area.
The results showed an average of 20 new HCQ prescriptions per 100,000 enrollees in 2019, but 2020 saw a sharp increase and drop in new HCQ prescriptions in March-April 2020, independent of counties’ breakdown of political affiliation.
“However, after June 2020, coinciding with the revocation of the U.S. Food and Drug Administration’s emergency use authorization for hydroxychloroquine, prescribing volume was significantly higher in the highest vs. lowest Republican vote share counties,” the authors report. The gradual increase from June through December 2020 averaged to 42 new prescriptions per 100,000, a 146% increase over 2019 rates that was driven largely by the 25% of counties with the highest proportion of Republican voters.
Similarly, rates of new ivermectin prescriptions in December 2020 were more than nine times higher in counties with the highest Republican vote share, compared with new prescriptions throughout 2019. The researchers found no differences in new prescriptions for methotrexate or albendazole in 2020 based on counties’ proportion of Republican votes.
Since the study is an ecological, observational one, it cannot show causation or shed light on what role patients, physicians, or other factors might have played in prescribing patterns. Nevertheless, the authors noted the potentially negative implications of their findings.
“Because political affiliation should not be a factor in clinical treatment decisions, our findings raise concerns for public trust in a nonpartisan health care system,” the authors write.
Coauthor Ateev Mehrotra, MD, MPH, reported personal fees from Sanofi-Aventis, and coauthor Anupam B. Jena, MD, PhD, reported personal fees from Bioverativ, Merck, Janssen, Edwards Lifesciences, Novartis, Amgen, Eisai, Otsuka, Vertex, Celgene, Sanofi-Aventis, Precision Health Economics (now PRECISIONheor), Analysis Group, and Doubleday and hosting the podcast Freakonomics, M.D. The other coauthors have disclosed no relevant financial relationships. No external funding source was noted.
A version of this article first appeared on Medscape.com.
FROM JAMA INTERNAL MEDICINE
Treatment for BV, trichomoniasis approved for adolescents
The antimicrobial agent, marketed as Solosec, was first approved in 2017 as a treatment for BV in adult women. In 2021, it was approved for the treatment of trichomoniasis in adult men and women.
Lupin Pharmaceuticals, which manufactures the drug, announced the expanded approval for adolescents in a news release.
The medication is meant to be taken as a single dose. It comes in a packet that should be sprinkled onto applesauce, yogurt, or pudding and consumed without chewing or crunching.
The treatment option may help “address gaps in care related to adherence,” said Tom Merriam, an executive director with Lupin.
Bacterial vaginosis is a common vaginal infection. Trichomoniasis is the most common nonviral, curable STI in the United States. Sexual partners of patients with trichomoniasis can be treated at the same time.
Vulvovaginal candidiasis is one of the possible side effects of secnidazole treatment, the drug’s label notes.
The antimicrobial agent, marketed as Solosec, was first approved in 2017 as a treatment for BV in adult women. In 2021, it was approved for the treatment of trichomoniasis in adult men and women.
Lupin Pharmaceuticals, which manufactures the drug, announced the expanded approval for adolescents in a news release.
The medication is meant to be taken as a single dose. It comes in a packet that should be sprinkled onto applesauce, yogurt, or pudding and consumed without chewing or crunching.
The treatment option may help “address gaps in care related to adherence,” said Tom Merriam, an executive director with Lupin.
Bacterial vaginosis is a common vaginal infection. Trichomoniasis is the most common nonviral, curable STI in the United States. Sexual partners of patients with trichomoniasis can be treated at the same time.
Vulvovaginal candidiasis is one of the possible side effects of secnidazole treatment, the drug’s label notes.
The antimicrobial agent, marketed as Solosec, was first approved in 2017 as a treatment for BV in adult women. In 2021, it was approved for the treatment of trichomoniasis in adult men and women.
Lupin Pharmaceuticals, which manufactures the drug, announced the expanded approval for adolescents in a news release.
The medication is meant to be taken as a single dose. It comes in a packet that should be sprinkled onto applesauce, yogurt, or pudding and consumed without chewing or crunching.
The treatment option may help “address gaps in care related to adherence,” said Tom Merriam, an executive director with Lupin.
Bacterial vaginosis is a common vaginal infection. Trichomoniasis is the most common nonviral, curable STI in the United States. Sexual partners of patients with trichomoniasis can be treated at the same time.
Vulvovaginal candidiasis is one of the possible side effects of secnidazole treatment, the drug’s label notes.