User login
Final ASCEND study data: Acalabrutinib beat standard of care for r/r CLL
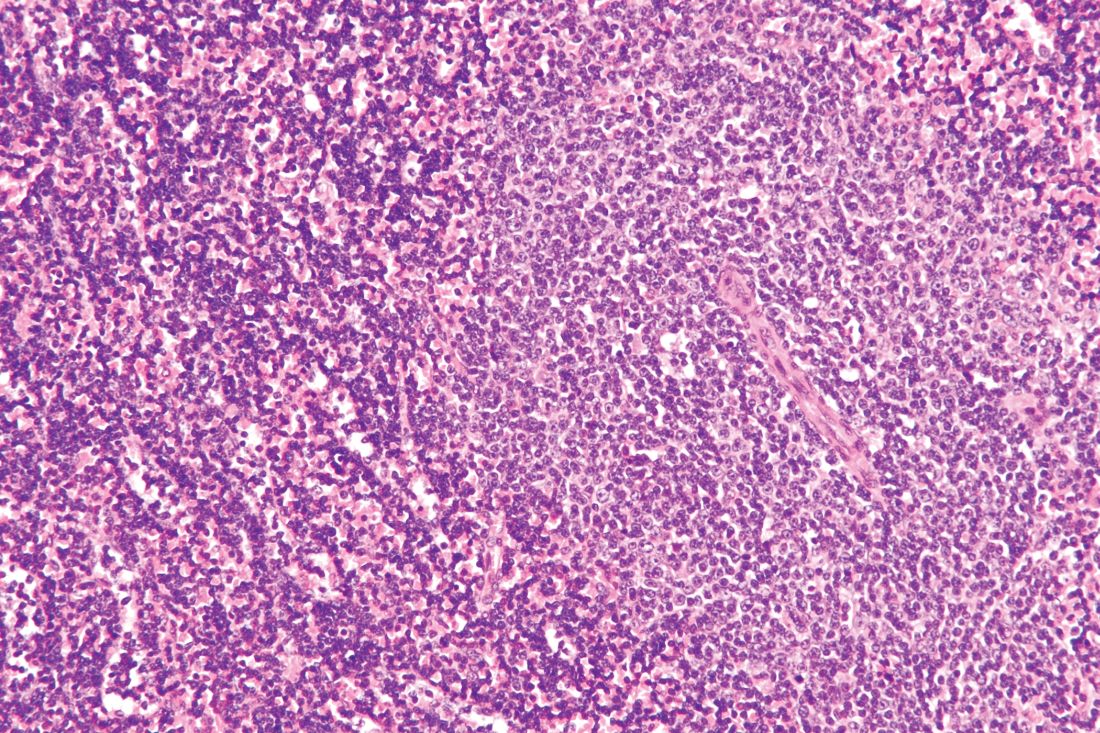
Acalabrutinib, a next-generation Bruton tyrosine kinase inhibitor, provides prolonged progression-free survival and better tolerability, compared with standard-of-care regimens for relapsed or refractory chronic lymphocytic leukemia (CLL), according to final results from the phase 3 ASCEND study.
The estimated 18-month progression-free survival (PFS) at a median of 22 months was 82% in 155 patients treated with acalabrutinib, compared with 48% in 155 treated with investigator’s choice of either idelalisib-rituximab (IdR) or bendamustine-rituximab (BR), which were given in 119 and 36 patients, respectively, Paolo Ghia, MD, PhD, reported at the Society of Hematologic Oncology virtual meeting.
The benefits of acalabrutinib were apparent regardless of high-risk genetic characteristics: Those with and without both del(17p) and TP53 mutations had similarly good PFS outcomes with acalabrutinib versus IdR/BR (HRs, 0.11 and 0.29, respectively), as did those with versus without unmutated IgVH (HRs, 0.28 and 0.30, respectively), said Dr. Ghia, professor of medical oncology at the Università Vita-Salute San Raffaele and IRCCS Ospedale San Raffaele, Milan.
The median overall survival was not reached in either arm, but estimated 18-month OS was 88% in both groups, likely because of the crossover being allowed for nonresponders in the IdR/BR groups, he noted.
Overall responses
The investigator-assessed overall response rates, including partial response or better, were also similar in the groups at 80% and 84%, respectively, and ORR, including partial response with lymphocytosis, was 92% versus 88%.
The duration of response was not reached in the acalabrutinib arm versus 18 months with IdR/BR, and estimated duration of response was 85% versus 49%.
The median drug exposure with acalabrutinib was approximately double that with IdR and about four times that of BR, Dr. Ghia said, noting that the difference between acalabrutinib and BR is explained by the short 6-month duration of treatment with BR, but the difference between acalabrutinib and IdR is because of adverse events (AEs).
Adverse events
AEs were the most common reason for treatment discontinuation in all three groups, but they led to discontinuation in only 16% with acalabrutinib versus 56% with IdR, he added.
The rates of AEs and AEs of clinical interest were generally similar to those reported at the interim analysis as presented in 2019 at the European Hematology Association annual meeting and published in the Journal of Clinical Oncology, despite the additional 6 months of follow up, he said.
Additionally, the incidence of grade 3 or higher AEs, serious AEs, and treatment-related AEs were all greater with IdR than with acalabrutinib or BR. The most common AEs with acalabrutinib were headache, neutropenia, diarrhea, and upper-respiratory infection, which were mostly grade 1 or 2. The most common grade 3 or higher AEs were neutropenia, anemia, and pneumonia, which were reported in 12%, 17%, and 7% of patients.
Confirmatory results
“The final results from the ASCEND study confirm the findings at the interim analysis and support the favorable efficacy and safety of acalabrutinib versus standard-of-care regimens ... in patients with relapsed/refractory CLL,” Dr. Ghia said.
“Overall, these final results from ASCENT support the use of acalabrutinib in patients with relapsed/refractory CLL, including those with high-risk genetic features.”
This study was sponsored by Acerta Pharma. Dr. Ghia reported consulting or advisory roles, grant or research funding, and/or honoraria from Abbvie, BeiGene, Janssen, Gilead Sciences, Sunesis Pharmaceuticals, Juno Therapeutics, ArQule, Adaptive Biotechnologies, Dynamo Therapeutics, MEI Pharma, and Novartis.
SOURCE: Ghia P et al. SOHO 2020, Abstract CLL-091.
Acalabrutinib, a next-generation Bruton tyrosine kinase inhibitor, provides prolonged progression-free survival and better tolerability, compared with standard-of-care regimens for relapsed or refractory chronic lymphocytic leukemia (CLL), according to final results from the phase 3 ASCEND study.
The estimated 18-month progression-free survival (PFS) at a median of 22 months was 82% in 155 patients treated with acalabrutinib, compared with 48% in 155 treated with investigator’s choice of either idelalisib-rituximab (IdR) or bendamustine-rituximab (BR), which were given in 119 and 36 patients, respectively, Paolo Ghia, MD, PhD, reported at the Society of Hematologic Oncology virtual meeting.
The benefits of acalabrutinib were apparent regardless of high-risk genetic characteristics: Those with and without both del(17p) and TP53 mutations had similarly good PFS outcomes with acalabrutinib versus IdR/BR (HRs, 0.11 and 0.29, respectively), as did those with versus without unmutated IgVH (HRs, 0.28 and 0.30, respectively), said Dr. Ghia, professor of medical oncology at the Università Vita-Salute San Raffaele and IRCCS Ospedale San Raffaele, Milan.
The median overall survival was not reached in either arm, but estimated 18-month OS was 88% in both groups, likely because of the crossover being allowed for nonresponders in the IdR/BR groups, he noted.
Overall responses
The investigator-assessed overall response rates, including partial response or better, were also similar in the groups at 80% and 84%, respectively, and ORR, including partial response with lymphocytosis, was 92% versus 88%.
The duration of response was not reached in the acalabrutinib arm versus 18 months with IdR/BR, and estimated duration of response was 85% versus 49%.
The median drug exposure with acalabrutinib was approximately double that with IdR and about four times that of BR, Dr. Ghia said, noting that the difference between acalabrutinib and BR is explained by the short 6-month duration of treatment with BR, but the difference between acalabrutinib and IdR is because of adverse events (AEs).
Adverse events
AEs were the most common reason for treatment discontinuation in all three groups, but they led to discontinuation in only 16% with acalabrutinib versus 56% with IdR, he added.
The rates of AEs and AEs of clinical interest were generally similar to those reported at the interim analysis as presented in 2019 at the European Hematology Association annual meeting and published in the Journal of Clinical Oncology, despite the additional 6 months of follow up, he said.
Additionally, the incidence of grade 3 or higher AEs, serious AEs, and treatment-related AEs were all greater with IdR than with acalabrutinib or BR. The most common AEs with acalabrutinib were headache, neutropenia, diarrhea, and upper-respiratory infection, which were mostly grade 1 or 2. The most common grade 3 or higher AEs were neutropenia, anemia, and pneumonia, which were reported in 12%, 17%, and 7% of patients.
Confirmatory results
“The final results from the ASCEND study confirm the findings at the interim analysis and support the favorable efficacy and safety of acalabrutinib versus standard-of-care regimens ... in patients with relapsed/refractory CLL,” Dr. Ghia said.
“Overall, these final results from ASCENT support the use of acalabrutinib in patients with relapsed/refractory CLL, including those with high-risk genetic features.”
This study was sponsored by Acerta Pharma. Dr. Ghia reported consulting or advisory roles, grant or research funding, and/or honoraria from Abbvie, BeiGene, Janssen, Gilead Sciences, Sunesis Pharmaceuticals, Juno Therapeutics, ArQule, Adaptive Biotechnologies, Dynamo Therapeutics, MEI Pharma, and Novartis.
SOURCE: Ghia P et al. SOHO 2020, Abstract CLL-091.
Acalabrutinib, a next-generation Bruton tyrosine kinase inhibitor, provides prolonged progression-free survival and better tolerability, compared with standard-of-care regimens for relapsed or refractory chronic lymphocytic leukemia (CLL), according to final results from the phase 3 ASCEND study.
The estimated 18-month progression-free survival (PFS) at a median of 22 months was 82% in 155 patients treated with acalabrutinib, compared with 48% in 155 treated with investigator’s choice of either idelalisib-rituximab (IdR) or bendamustine-rituximab (BR), which were given in 119 and 36 patients, respectively, Paolo Ghia, MD, PhD, reported at the Society of Hematologic Oncology virtual meeting.
The benefits of acalabrutinib were apparent regardless of high-risk genetic characteristics: Those with and without both del(17p) and TP53 mutations had similarly good PFS outcomes with acalabrutinib versus IdR/BR (HRs, 0.11 and 0.29, respectively), as did those with versus without unmutated IgVH (HRs, 0.28 and 0.30, respectively), said Dr. Ghia, professor of medical oncology at the Università Vita-Salute San Raffaele and IRCCS Ospedale San Raffaele, Milan.
The median overall survival was not reached in either arm, but estimated 18-month OS was 88% in both groups, likely because of the crossover being allowed for nonresponders in the IdR/BR groups, he noted.
Overall responses
The investigator-assessed overall response rates, including partial response or better, were also similar in the groups at 80% and 84%, respectively, and ORR, including partial response with lymphocytosis, was 92% versus 88%.
The duration of response was not reached in the acalabrutinib arm versus 18 months with IdR/BR, and estimated duration of response was 85% versus 49%.
The median drug exposure with acalabrutinib was approximately double that with IdR and about four times that of BR, Dr. Ghia said, noting that the difference between acalabrutinib and BR is explained by the short 6-month duration of treatment with BR, but the difference between acalabrutinib and IdR is because of adverse events (AEs).
Adverse events
AEs were the most common reason for treatment discontinuation in all three groups, but they led to discontinuation in only 16% with acalabrutinib versus 56% with IdR, he added.
The rates of AEs and AEs of clinical interest were generally similar to those reported at the interim analysis as presented in 2019 at the European Hematology Association annual meeting and published in the Journal of Clinical Oncology, despite the additional 6 months of follow up, he said.
Additionally, the incidence of grade 3 or higher AEs, serious AEs, and treatment-related AEs were all greater with IdR than with acalabrutinib or BR. The most common AEs with acalabrutinib were headache, neutropenia, diarrhea, and upper-respiratory infection, which were mostly grade 1 or 2. The most common grade 3 or higher AEs were neutropenia, anemia, and pneumonia, which were reported in 12%, 17%, and 7% of patients.
Confirmatory results
“The final results from the ASCEND study confirm the findings at the interim analysis and support the favorable efficacy and safety of acalabrutinib versus standard-of-care regimens ... in patients with relapsed/refractory CLL,” Dr. Ghia said.
“Overall, these final results from ASCENT support the use of acalabrutinib in patients with relapsed/refractory CLL, including those with high-risk genetic features.”
This study was sponsored by Acerta Pharma. Dr. Ghia reported consulting or advisory roles, grant or research funding, and/or honoraria from Abbvie, BeiGene, Janssen, Gilead Sciences, Sunesis Pharmaceuticals, Juno Therapeutics, ArQule, Adaptive Biotechnologies, Dynamo Therapeutics, MEI Pharma, and Novartis.
SOURCE: Ghia P et al. SOHO 2020, Abstract CLL-091.
FROM SOHO 2020
New data and trial outcomes clarify path to TFR in CML
The rate of reduction in BCR-ABL1 value during the first 3 months of tyrosine kinase inhibitor therapy for chronic myeloid leukemia (CML) independently predicts the likelihood of sustained treatment-free remission (TFR) in eligible patients, a recent study shows.
The findings, along with the 10-year outcomes data from the phase 3 ENESTnd trial reported in 2019, can help with complex TFR decision-making, lead author Timothy P. Hughes, MD, said at the Society of Hematologic Oncology virtual meeting.
In 115 chronic-phase CML patients who were eligible and attempted TFR and had at least 12 months of follow-up, the probability of sustained TFR, defined as major molecular response off tyrosine kinase inhibitor therapy for 12 continuous months, was 55%. Sustained TFR occurred in 80% of those in the first quartile of response time (halving time of less than 9.4 days), compared with 4% of those in the last quartile (halving time of more than 21.9 days), said Dr. Hughes of the South Australian Health and Medical Research Institute, Adelaide.
The model assumes molecular response of 4.5 status duration for 3 years – not just achievement of MR4.5.
“So that’s the other variable in this equation,’ he said.
The findings, which were published online Sept. 1 in Blood, were validated in an independent dataset.
Dr. Hughes and colleagues concluded that the data “support the critical importance of the initial kinetics of BCR-ABL1 decline for long-term outcomes.”
As an example of how the findings, along with those from ENESTnd, can help with TFR decision-making, Dr. Hughes presented a case involving a 59-year-old man with chronic-phase CML diagnosed 5 years prior with intermediate EUTOS long-term survival score (ELTS) and Sokal scores and a low Framingham Risk Score at diagnosis.
The patient was treated with frontline nilotinib at a standard dose of 300 mg twice daily and he responded well, achieving an MR4 molecular response after 18 months, and MR4.5 score at 2.5 years, which was maintained at 5 years.
“That’s a BCR-ABL level of less than 0.01% on the International Scale,” he said, noting that the patient’s BCR-ABL level started at 290% and had “a very, very steep fall to 0.26% at 3 months.”
Cardiovascular risk a factor
The patient was interested in attempting TFR when eligible, but had some vascular toxicity risks; he was being treated for hypertension and hypercholesterolemia and also had a family history of coronary artery disease.
Hypercholesterolemia is a recognized effect of nilotinib therapy, but both where being treated and were under control, Dr. Hughes noted.
The patient’s Framingham Risk Score had increased from 9 (low risk) to 16 (intermediate risk).
In determining whether to attempt TFR and closely monitor the patient or delay the attempt and perhaps either change to imatinib therapy or reduce the nilotinib dose, Dr. Hughes said it was important to consider the cardiovascular event risks as elucidated in ENESTnd.
It was hoped that the increased cardiovascular event risk demonstrated in years 0-5 of the study would diminish in the later years, but the 10-year finding actually showed persistent risk with nilotinib treatment: In years 0-5, 7.2%, 11.9% and 1.8% of patients in study arms receiving nilotinib 300 mg twice daily, nilotinib 400 mg twice daily, and imatinib 400 mg once daily, respectively, experienced a cardiovascular event. In years 5-10, the corresponding rates were 9.3%, 11.9%, and 1.8%.
“I think it’s an important message that the risk is there, at about the same rate, in the second 5 years,” said Dr. Hughes, the first author of the study.
The ENESTnd data also show how the Framingham Risk Score, which is based mainly on age, cholesterol levels, blood pressure, smoking history, and diabetes history, is associated with cardiovascular event rates in the treatment arms.
Patients with a low Framingham score who were receiving nilotinib had no greater risk of a cardiovascular event than did those receiving nilotinib during years 0-5.
“I think that makes it an attractive option in patients where you’re focusing on early achievement of deep molecular response and eligibility for treatment-free remission,” he said, adding that it’s a different story for those with intermediate or high Framingham scores, who have “ a really quite substantial” risk in the first 5 years.
The 5- to 10-year ENESTnd data, however, show that this lack of risk in low Framingham scores did not hold true. Even in those with a low-risk Framingham score, the overall 10-year event rate was 7.3% with nilotinib versus 1.1% with imatinib.
“This is an important message that it’s probably not appropriate to assume that your patient with low Framingham Risk Score at diagnosis is not having a higher risk of cardiovascular events in the period after 5 years out to 10 years,” Dr. Hughes said.
Of note, the case patient was considered eligible for TFR under all of the mandatory requirements of both the 2020 European LeukemiaNET recommendations and the National Comprehensive Cancer Network 2020 guideline for CML, which have slight differences but are “generally in accord.”
Based on those recommendations, the patient would be “eligible and probably recommended,” for TFR, he said.
The 10-year ENESTnd findings and the findings by Dr. Hughes and colleagues with respect to the tempo of early tyrosine kinase inhibitor response provide further confirmation of the patient’s eligibility.
“I would feel very happy to say to this patient: ‘You’ve got an excellent chance of achieving treatment-free remission today; going on with therapy is probably not in your interest given the risk of a cardiovascular event, so I’d recommend stopping,’ ” he said. “If the patient was not keen to stop, then I’d recommend switching to imatinib, because I don’t think we’re getting any great benefit from pushing on with nilotinib if the plan is not to attempt treatment-free remission.”
However, if the patient preferred another year of treatment before attempting TFR, it might be worth considering reducing the dose or switching to low-dose dasatinib, he noted, concluding that “the vascular risk profile and the prospect of treatment-free remission need to be carefully considered in every patient, particularly patients on second-generation drugs, before deciding whether to recommend treatment-free remission or extending therapy longer and whether it’s appropriate to just reduce the dose or switch.”
Dr. Hughes has received grant or research support and honoraria from Novartis and Bristol-Myers Squibb, and has been a paid consultant and advisory committee or review panel member for both companies.
The rate of reduction in BCR-ABL1 value during the first 3 months of tyrosine kinase inhibitor therapy for chronic myeloid leukemia (CML) independently predicts the likelihood of sustained treatment-free remission (TFR) in eligible patients, a recent study shows.
The findings, along with the 10-year outcomes data from the phase 3 ENESTnd trial reported in 2019, can help with complex TFR decision-making, lead author Timothy P. Hughes, MD, said at the Society of Hematologic Oncology virtual meeting.
In 115 chronic-phase CML patients who were eligible and attempted TFR and had at least 12 months of follow-up, the probability of sustained TFR, defined as major molecular response off tyrosine kinase inhibitor therapy for 12 continuous months, was 55%. Sustained TFR occurred in 80% of those in the first quartile of response time (halving time of less than 9.4 days), compared with 4% of those in the last quartile (halving time of more than 21.9 days), said Dr. Hughes of the South Australian Health and Medical Research Institute, Adelaide.
The model assumes molecular response of 4.5 status duration for 3 years – not just achievement of MR4.5.
“So that’s the other variable in this equation,’ he said.
The findings, which were published online Sept. 1 in Blood, were validated in an independent dataset.
Dr. Hughes and colleagues concluded that the data “support the critical importance of the initial kinetics of BCR-ABL1 decline for long-term outcomes.”
As an example of how the findings, along with those from ENESTnd, can help with TFR decision-making, Dr. Hughes presented a case involving a 59-year-old man with chronic-phase CML diagnosed 5 years prior with intermediate EUTOS long-term survival score (ELTS) and Sokal scores and a low Framingham Risk Score at diagnosis.
The patient was treated with frontline nilotinib at a standard dose of 300 mg twice daily and he responded well, achieving an MR4 molecular response after 18 months, and MR4.5 score at 2.5 years, which was maintained at 5 years.
“That’s a BCR-ABL level of less than 0.01% on the International Scale,” he said, noting that the patient’s BCR-ABL level started at 290% and had “a very, very steep fall to 0.26% at 3 months.”
Cardiovascular risk a factor
The patient was interested in attempting TFR when eligible, but had some vascular toxicity risks; he was being treated for hypertension and hypercholesterolemia and also had a family history of coronary artery disease.
Hypercholesterolemia is a recognized effect of nilotinib therapy, but both where being treated and were under control, Dr. Hughes noted.
The patient’s Framingham Risk Score had increased from 9 (low risk) to 16 (intermediate risk).
In determining whether to attempt TFR and closely monitor the patient or delay the attempt and perhaps either change to imatinib therapy or reduce the nilotinib dose, Dr. Hughes said it was important to consider the cardiovascular event risks as elucidated in ENESTnd.
It was hoped that the increased cardiovascular event risk demonstrated in years 0-5 of the study would diminish in the later years, but the 10-year finding actually showed persistent risk with nilotinib treatment: In years 0-5, 7.2%, 11.9% and 1.8% of patients in study arms receiving nilotinib 300 mg twice daily, nilotinib 400 mg twice daily, and imatinib 400 mg once daily, respectively, experienced a cardiovascular event. In years 5-10, the corresponding rates were 9.3%, 11.9%, and 1.8%.
“I think it’s an important message that the risk is there, at about the same rate, in the second 5 years,” said Dr. Hughes, the first author of the study.
The ENESTnd data also show how the Framingham Risk Score, which is based mainly on age, cholesterol levels, blood pressure, smoking history, and diabetes history, is associated with cardiovascular event rates in the treatment arms.
Patients with a low Framingham score who were receiving nilotinib had no greater risk of a cardiovascular event than did those receiving nilotinib during years 0-5.
“I think that makes it an attractive option in patients where you’re focusing on early achievement of deep molecular response and eligibility for treatment-free remission,” he said, adding that it’s a different story for those with intermediate or high Framingham scores, who have “ a really quite substantial” risk in the first 5 years.
The 5- to 10-year ENESTnd data, however, show that this lack of risk in low Framingham scores did not hold true. Even in those with a low-risk Framingham score, the overall 10-year event rate was 7.3% with nilotinib versus 1.1% with imatinib.
“This is an important message that it’s probably not appropriate to assume that your patient with low Framingham Risk Score at diagnosis is not having a higher risk of cardiovascular events in the period after 5 years out to 10 years,” Dr. Hughes said.
Of note, the case patient was considered eligible for TFR under all of the mandatory requirements of both the 2020 European LeukemiaNET recommendations and the National Comprehensive Cancer Network 2020 guideline for CML, which have slight differences but are “generally in accord.”
Based on those recommendations, the patient would be “eligible and probably recommended,” for TFR, he said.
The 10-year ENESTnd findings and the findings by Dr. Hughes and colleagues with respect to the tempo of early tyrosine kinase inhibitor response provide further confirmation of the patient’s eligibility.
“I would feel very happy to say to this patient: ‘You’ve got an excellent chance of achieving treatment-free remission today; going on with therapy is probably not in your interest given the risk of a cardiovascular event, so I’d recommend stopping,’ ” he said. “If the patient was not keen to stop, then I’d recommend switching to imatinib, because I don’t think we’re getting any great benefit from pushing on with nilotinib if the plan is not to attempt treatment-free remission.”
However, if the patient preferred another year of treatment before attempting TFR, it might be worth considering reducing the dose or switching to low-dose dasatinib, he noted, concluding that “the vascular risk profile and the prospect of treatment-free remission need to be carefully considered in every patient, particularly patients on second-generation drugs, before deciding whether to recommend treatment-free remission or extending therapy longer and whether it’s appropriate to just reduce the dose or switch.”
Dr. Hughes has received grant or research support and honoraria from Novartis and Bristol-Myers Squibb, and has been a paid consultant and advisory committee or review panel member for both companies.
The rate of reduction in BCR-ABL1 value during the first 3 months of tyrosine kinase inhibitor therapy for chronic myeloid leukemia (CML) independently predicts the likelihood of sustained treatment-free remission (TFR) in eligible patients, a recent study shows.
The findings, along with the 10-year outcomes data from the phase 3 ENESTnd trial reported in 2019, can help with complex TFR decision-making, lead author Timothy P. Hughes, MD, said at the Society of Hematologic Oncology virtual meeting.
In 115 chronic-phase CML patients who were eligible and attempted TFR and had at least 12 months of follow-up, the probability of sustained TFR, defined as major molecular response off tyrosine kinase inhibitor therapy for 12 continuous months, was 55%. Sustained TFR occurred in 80% of those in the first quartile of response time (halving time of less than 9.4 days), compared with 4% of those in the last quartile (halving time of more than 21.9 days), said Dr. Hughes of the South Australian Health and Medical Research Institute, Adelaide.
The model assumes molecular response of 4.5 status duration for 3 years – not just achievement of MR4.5.
“So that’s the other variable in this equation,’ he said.
The findings, which were published online Sept. 1 in Blood, were validated in an independent dataset.
Dr. Hughes and colleagues concluded that the data “support the critical importance of the initial kinetics of BCR-ABL1 decline for long-term outcomes.”
As an example of how the findings, along with those from ENESTnd, can help with TFR decision-making, Dr. Hughes presented a case involving a 59-year-old man with chronic-phase CML diagnosed 5 years prior with intermediate EUTOS long-term survival score (ELTS) and Sokal scores and a low Framingham Risk Score at diagnosis.
The patient was treated with frontline nilotinib at a standard dose of 300 mg twice daily and he responded well, achieving an MR4 molecular response after 18 months, and MR4.5 score at 2.5 years, which was maintained at 5 years.
“That’s a BCR-ABL level of less than 0.01% on the International Scale,” he said, noting that the patient’s BCR-ABL level started at 290% and had “a very, very steep fall to 0.26% at 3 months.”
Cardiovascular risk a factor
The patient was interested in attempting TFR when eligible, but had some vascular toxicity risks; he was being treated for hypertension and hypercholesterolemia and also had a family history of coronary artery disease.
Hypercholesterolemia is a recognized effect of nilotinib therapy, but both where being treated and were under control, Dr. Hughes noted.
The patient’s Framingham Risk Score had increased from 9 (low risk) to 16 (intermediate risk).
In determining whether to attempt TFR and closely monitor the patient or delay the attempt and perhaps either change to imatinib therapy or reduce the nilotinib dose, Dr. Hughes said it was important to consider the cardiovascular event risks as elucidated in ENESTnd.
It was hoped that the increased cardiovascular event risk demonstrated in years 0-5 of the study would diminish in the later years, but the 10-year finding actually showed persistent risk with nilotinib treatment: In years 0-5, 7.2%, 11.9% and 1.8% of patients in study arms receiving nilotinib 300 mg twice daily, nilotinib 400 mg twice daily, and imatinib 400 mg once daily, respectively, experienced a cardiovascular event. In years 5-10, the corresponding rates were 9.3%, 11.9%, and 1.8%.
“I think it’s an important message that the risk is there, at about the same rate, in the second 5 years,” said Dr. Hughes, the first author of the study.
The ENESTnd data also show how the Framingham Risk Score, which is based mainly on age, cholesterol levels, blood pressure, smoking history, and diabetes history, is associated with cardiovascular event rates in the treatment arms.
Patients with a low Framingham score who were receiving nilotinib had no greater risk of a cardiovascular event than did those receiving nilotinib during years 0-5.
“I think that makes it an attractive option in patients where you’re focusing on early achievement of deep molecular response and eligibility for treatment-free remission,” he said, adding that it’s a different story for those with intermediate or high Framingham scores, who have “ a really quite substantial” risk in the first 5 years.
The 5- to 10-year ENESTnd data, however, show that this lack of risk in low Framingham scores did not hold true. Even in those with a low-risk Framingham score, the overall 10-year event rate was 7.3% with nilotinib versus 1.1% with imatinib.
“This is an important message that it’s probably not appropriate to assume that your patient with low Framingham Risk Score at diagnosis is not having a higher risk of cardiovascular events in the period after 5 years out to 10 years,” Dr. Hughes said.
Of note, the case patient was considered eligible for TFR under all of the mandatory requirements of both the 2020 European LeukemiaNET recommendations and the National Comprehensive Cancer Network 2020 guideline for CML, which have slight differences but are “generally in accord.”
Based on those recommendations, the patient would be “eligible and probably recommended,” for TFR, he said.
The 10-year ENESTnd findings and the findings by Dr. Hughes and colleagues with respect to the tempo of early tyrosine kinase inhibitor response provide further confirmation of the patient’s eligibility.
“I would feel very happy to say to this patient: ‘You’ve got an excellent chance of achieving treatment-free remission today; going on with therapy is probably not in your interest given the risk of a cardiovascular event, so I’d recommend stopping,’ ” he said. “If the patient was not keen to stop, then I’d recommend switching to imatinib, because I don’t think we’re getting any great benefit from pushing on with nilotinib if the plan is not to attempt treatment-free remission.”
However, if the patient preferred another year of treatment before attempting TFR, it might be worth considering reducing the dose or switching to low-dose dasatinib, he noted, concluding that “the vascular risk profile and the prospect of treatment-free remission need to be carefully considered in every patient, particularly patients on second-generation drugs, before deciding whether to recommend treatment-free remission or extending therapy longer and whether it’s appropriate to just reduce the dose or switch.”
Dr. Hughes has received grant or research support and honoraria from Novartis and Bristol-Myers Squibb, and has been a paid consultant and advisory committee or review panel member for both companies.
FROM SOHO 2020
CML: New TKIs and combos show promise for resistant, intolerant disease
Most patients with chronic myeloid leukemia (CML) have a normal life expectancy thanks to dramatic improvements in treatments and outcomes over the past few decades, but new treatment approaches are needed for the subset who fail to respond or who develop resistance to existing treatments, according to Jorge Cortes, MD, director of the Georgia Cancer Center, Augusta.
Several novel tyrosine kinase inhibitors (TKIs) and combination therapies show promise in early studies, he said at the Society of Hematologic Oncology virtual meeting.
Asciminib
The allosteric inhibitor asciminib (ABL-001), for example, has completed phase 1/2 trials evaluating its use as a single agent and in combination with other therapies in the first-line setting, and a pivotal phase 3 study comparing it with bosutinib in the third-line setting is underway, Dr. Cortes said.
The rate of major cytogenetic response (MCyr) to asciminib in heavily pretreated patients in a phase 1/2 study published the New England Journal of Medicine was “very good” at 77%.
“And almost half [48%] of the patients had a major molecular response by 12 months,” he said, noting that even after excluding those who had a prior response but were enrolled because they couldn’t tolerate prior treatments, the MCyr and major molecular response (MMR) rates were 60% and 36%, respectively.
Asciminib also showed activity in patients with T315I mutations: The MCyr rate was 55% and the MMR rate at 12 months was 24%.
“Now, it is important to recognize that the doses that are required for inhibition – for getting these responses in [patients with] T315I – are higher than we need for the patients that do not have T315I, so it needs higher concentrations in vitro and it needs higher doses in vivo,” he said.
Also of note, the response rates were good both in those with two or fewer prior lines of therapy and in those with three or more (12-month MMR rates were 47% and 34%, respectively). For the latter, that’s “a very good rate, even though we’re only talking about 12 months of therapy,” Dr. Cortes said.
“And even in the patients who had been resistant or intolerant to ponatinib, 40% achieved a major molecular response, so very good results regardless of the number or type of tyrosine kinase inhibitors the patient had received, ” he added. The numbers in the group with T315I mutations are small, so further exploration is needed in subsequent studies, he noted.
The emergence of resistance is a concern with asciminib, but in a xenograft model, combining it with nilotinib appeared to prevent resistance. Therefore, the combination of asciminib and various TKIs has been explored in the clinic.
In a phase 1 study of asciminib and imatinib presented by Dr. Cortes at the European Hematology Association meeting in 2019, the complete cytogenetic response and MMR rates at 48 weeks were 50% and 42%, respectively.
“Now, this is a different type of population – perhaps a little more heavily pretreated than the ones who received single-agent asciminib, but it does show the potential for synergy, and importantly it was not associated with increased toxicity,” he said.
PF-114
Another agent in development is PF-114, a third-generation BCR-ABL inhibitor. It is a structural analogue of ponatinib that is modified to avoid inhibiting the VEGFR receptor in an effort to prevent “arterial occlusive and particularly hypertension, adverse events that we see with ponatinib,” he said.
In a phase 1 study of 51 patients with CML who failed at least two prior TKIs or had T315I mutation, the MCyr rate was 50% and the MMR rate was 36%. The drug was very well tolerated: The dose-limiting toxicity was skin toxicity involving psoriasiform lesions, which were manageable, he noted.
“Importantly ... there was no cardiovascular toxicity,” he added.
Those findings were presented at ASH 2018. The drug is now moving to a phase 2 study.
HQP1351 (GZD824)
The orally active, small-molecule BCR-ABL inhibitor HQP1351 is a third-generation TKI with activity against a broad spectrum of BCR-ABL mutations.
A phase 1 study of patients who were resistant to prior TKIs is complete, and results presented at ASH 2019 showed that most patients (67%) had only one or two prior therapies and 63% had T315I mutation. Response rates were better in the patients with T315I mutations (MCyr, 78% vs. 34%; MMR, 52% vs. 15% in 101 chronic phase patients).
The treatment was well tolerated, with grade 3 toxicity involving only hypertriglyceridemia, pyrexia, and proteinuria. No arterial occlusive events were reported.
K0706
K0706 is a selective inhibitor of BCR-ABL1 designed to inhibit enzymatic activity of BCR-ABL. The agent was efficacious and well tolerated with limited off-target activity in preclinical models. It can inhibit wild-type and mutant forms of BCR-ABL, but does not have activity against T315I.
Results of a phase 1 study presented at ASH in 2019 by Dr. Cortes showed that all the patients who received a dose of 174 mg or greater achieved or maintained a cytogenetic response at 6 months, and 50% achieved or maintained an MMR.
“This is a very good response rate in this heavily pretreated population,” he said.
Patients who received prior ponatinib had a somewhat lower response, but still, nearly 45% achieved an MCyr.
“So very good response rates, no arterial occlusive events, and phase 2 studies will be starting at the dose of 174 mg,” he said.
Additional combinations
As for combining TKIs with other agents, efforts are underway around the world to find ways to eradicate minimal residual disease. Examples include TKIs and imatinib, TKIs and azacitidine, and asciminib plus another TKI, to name a few.
One study from Germany showed that adding interferon leads to earlier achievement of MMR, but ultimately the responses were similar, Dr. Cortes said.
Adding venetoclax has shown some activity in the preclinical setting, and studies of that combination will be starting soon in the clinic, he noted.
Implications
The current survival probability in CML patients is 92% when considering CML-related deaths (68% when considering all-cause mortality), compared with 8% in the 1980s and 35%-43% in the early 1990s.
But the current benefits don’t extend to all patients, Dr. Cortes said.
“There are patients who actually end up having worse prognosis than we would expect,” he said, explaining that some CML-related deaths are attributable to lack of access to therapy and good care, but some are related to true poor prognosis, often caused by resistance or inability to tolerate treatments.
In fact, data from studies of various treatments show that almost 40% of patients on dasatinib or nilotinib change therapy by 5 years, and by 10 years, half of those randomized to nilotinib have changed therapy.
“So it is not uncommon that patients have to change therapy for one reason or another,” he said, adding that, as resistance persists through additional treatment options, the prognosis worsens significantly.
“It is important that we have new therapeutic options to be able to help these patients who are going to be in need of additional therapies,” he said.
Dr. Cortes has received grant or research support from Novartis, Pfizer, Takeda, and Sun Pharma, and he is a paid consultant for Pfizer, Novartis, and Takeda.
Most patients with chronic myeloid leukemia (CML) have a normal life expectancy thanks to dramatic improvements in treatments and outcomes over the past few decades, but new treatment approaches are needed for the subset who fail to respond or who develop resistance to existing treatments, according to Jorge Cortes, MD, director of the Georgia Cancer Center, Augusta.
Several novel tyrosine kinase inhibitors (TKIs) and combination therapies show promise in early studies, he said at the Society of Hematologic Oncology virtual meeting.
Asciminib
The allosteric inhibitor asciminib (ABL-001), for example, has completed phase 1/2 trials evaluating its use as a single agent and in combination with other therapies in the first-line setting, and a pivotal phase 3 study comparing it with bosutinib in the third-line setting is underway, Dr. Cortes said.
The rate of major cytogenetic response (MCyr) to asciminib in heavily pretreated patients in a phase 1/2 study published the New England Journal of Medicine was “very good” at 77%.
“And almost half [48%] of the patients had a major molecular response by 12 months,” he said, noting that even after excluding those who had a prior response but were enrolled because they couldn’t tolerate prior treatments, the MCyr and major molecular response (MMR) rates were 60% and 36%, respectively.
Asciminib also showed activity in patients with T315I mutations: The MCyr rate was 55% and the MMR rate at 12 months was 24%.
“Now, it is important to recognize that the doses that are required for inhibition – for getting these responses in [patients with] T315I – are higher than we need for the patients that do not have T315I, so it needs higher concentrations in vitro and it needs higher doses in vivo,” he said.
Also of note, the response rates were good both in those with two or fewer prior lines of therapy and in those with three or more (12-month MMR rates were 47% and 34%, respectively). For the latter, that’s “a very good rate, even though we’re only talking about 12 months of therapy,” Dr. Cortes said.
“And even in the patients who had been resistant or intolerant to ponatinib, 40% achieved a major molecular response, so very good results regardless of the number or type of tyrosine kinase inhibitors the patient had received, ” he added. The numbers in the group with T315I mutations are small, so further exploration is needed in subsequent studies, he noted.
The emergence of resistance is a concern with asciminib, but in a xenograft model, combining it with nilotinib appeared to prevent resistance. Therefore, the combination of asciminib and various TKIs has been explored in the clinic.
In a phase 1 study of asciminib and imatinib presented by Dr. Cortes at the European Hematology Association meeting in 2019, the complete cytogenetic response and MMR rates at 48 weeks were 50% and 42%, respectively.
“Now, this is a different type of population – perhaps a little more heavily pretreated than the ones who received single-agent asciminib, but it does show the potential for synergy, and importantly it was not associated with increased toxicity,” he said.
PF-114
Another agent in development is PF-114, a third-generation BCR-ABL inhibitor. It is a structural analogue of ponatinib that is modified to avoid inhibiting the VEGFR receptor in an effort to prevent “arterial occlusive and particularly hypertension, adverse events that we see with ponatinib,” he said.
In a phase 1 study of 51 patients with CML who failed at least two prior TKIs or had T315I mutation, the MCyr rate was 50% and the MMR rate was 36%. The drug was very well tolerated: The dose-limiting toxicity was skin toxicity involving psoriasiform lesions, which were manageable, he noted.
“Importantly ... there was no cardiovascular toxicity,” he added.
Those findings were presented at ASH 2018. The drug is now moving to a phase 2 study.
HQP1351 (GZD824)
The orally active, small-molecule BCR-ABL inhibitor HQP1351 is a third-generation TKI with activity against a broad spectrum of BCR-ABL mutations.
A phase 1 study of patients who were resistant to prior TKIs is complete, and results presented at ASH 2019 showed that most patients (67%) had only one or two prior therapies and 63% had T315I mutation. Response rates were better in the patients with T315I mutations (MCyr, 78% vs. 34%; MMR, 52% vs. 15% in 101 chronic phase patients).
The treatment was well tolerated, with grade 3 toxicity involving only hypertriglyceridemia, pyrexia, and proteinuria. No arterial occlusive events were reported.
K0706
K0706 is a selective inhibitor of BCR-ABL1 designed to inhibit enzymatic activity of BCR-ABL. The agent was efficacious and well tolerated with limited off-target activity in preclinical models. It can inhibit wild-type and mutant forms of BCR-ABL, but does not have activity against T315I.
Results of a phase 1 study presented at ASH in 2019 by Dr. Cortes showed that all the patients who received a dose of 174 mg or greater achieved or maintained a cytogenetic response at 6 months, and 50% achieved or maintained an MMR.
“This is a very good response rate in this heavily pretreated population,” he said.
Patients who received prior ponatinib had a somewhat lower response, but still, nearly 45% achieved an MCyr.
“So very good response rates, no arterial occlusive events, and phase 2 studies will be starting at the dose of 174 mg,” he said.
Additional combinations
As for combining TKIs with other agents, efforts are underway around the world to find ways to eradicate minimal residual disease. Examples include TKIs and imatinib, TKIs and azacitidine, and asciminib plus another TKI, to name a few.
One study from Germany showed that adding interferon leads to earlier achievement of MMR, but ultimately the responses were similar, Dr. Cortes said.
Adding venetoclax has shown some activity in the preclinical setting, and studies of that combination will be starting soon in the clinic, he noted.
Implications
The current survival probability in CML patients is 92% when considering CML-related deaths (68% when considering all-cause mortality), compared with 8% in the 1980s and 35%-43% in the early 1990s.
But the current benefits don’t extend to all patients, Dr. Cortes said.
“There are patients who actually end up having worse prognosis than we would expect,” he said, explaining that some CML-related deaths are attributable to lack of access to therapy and good care, but some are related to true poor prognosis, often caused by resistance or inability to tolerate treatments.
In fact, data from studies of various treatments show that almost 40% of patients on dasatinib or nilotinib change therapy by 5 years, and by 10 years, half of those randomized to nilotinib have changed therapy.
“So it is not uncommon that patients have to change therapy for one reason or another,” he said, adding that, as resistance persists through additional treatment options, the prognosis worsens significantly.
“It is important that we have new therapeutic options to be able to help these patients who are going to be in need of additional therapies,” he said.
Dr. Cortes has received grant or research support from Novartis, Pfizer, Takeda, and Sun Pharma, and he is a paid consultant for Pfizer, Novartis, and Takeda.
Most patients with chronic myeloid leukemia (CML) have a normal life expectancy thanks to dramatic improvements in treatments and outcomes over the past few decades, but new treatment approaches are needed for the subset who fail to respond or who develop resistance to existing treatments, according to Jorge Cortes, MD, director of the Georgia Cancer Center, Augusta.
Several novel tyrosine kinase inhibitors (TKIs) and combination therapies show promise in early studies, he said at the Society of Hematologic Oncology virtual meeting.
Asciminib
The allosteric inhibitor asciminib (ABL-001), for example, has completed phase 1/2 trials evaluating its use as a single agent and in combination with other therapies in the first-line setting, and a pivotal phase 3 study comparing it with bosutinib in the third-line setting is underway, Dr. Cortes said.
The rate of major cytogenetic response (MCyr) to asciminib in heavily pretreated patients in a phase 1/2 study published the New England Journal of Medicine was “very good” at 77%.
“And almost half [48%] of the patients had a major molecular response by 12 months,” he said, noting that even after excluding those who had a prior response but were enrolled because they couldn’t tolerate prior treatments, the MCyr and major molecular response (MMR) rates were 60% and 36%, respectively.
Asciminib also showed activity in patients with T315I mutations: The MCyr rate was 55% and the MMR rate at 12 months was 24%.
“Now, it is important to recognize that the doses that are required for inhibition – for getting these responses in [patients with] T315I – are higher than we need for the patients that do not have T315I, so it needs higher concentrations in vitro and it needs higher doses in vivo,” he said.
Also of note, the response rates were good both in those with two or fewer prior lines of therapy and in those with three or more (12-month MMR rates were 47% and 34%, respectively). For the latter, that’s “a very good rate, even though we’re only talking about 12 months of therapy,” Dr. Cortes said.
“And even in the patients who had been resistant or intolerant to ponatinib, 40% achieved a major molecular response, so very good results regardless of the number or type of tyrosine kinase inhibitors the patient had received, ” he added. The numbers in the group with T315I mutations are small, so further exploration is needed in subsequent studies, he noted.
The emergence of resistance is a concern with asciminib, but in a xenograft model, combining it with nilotinib appeared to prevent resistance. Therefore, the combination of asciminib and various TKIs has been explored in the clinic.
In a phase 1 study of asciminib and imatinib presented by Dr. Cortes at the European Hematology Association meeting in 2019, the complete cytogenetic response and MMR rates at 48 weeks were 50% and 42%, respectively.
“Now, this is a different type of population – perhaps a little more heavily pretreated than the ones who received single-agent asciminib, but it does show the potential for synergy, and importantly it was not associated with increased toxicity,” he said.
PF-114
Another agent in development is PF-114, a third-generation BCR-ABL inhibitor. It is a structural analogue of ponatinib that is modified to avoid inhibiting the VEGFR receptor in an effort to prevent “arterial occlusive and particularly hypertension, adverse events that we see with ponatinib,” he said.
In a phase 1 study of 51 patients with CML who failed at least two prior TKIs or had T315I mutation, the MCyr rate was 50% and the MMR rate was 36%. The drug was very well tolerated: The dose-limiting toxicity was skin toxicity involving psoriasiform lesions, which were manageable, he noted.
“Importantly ... there was no cardiovascular toxicity,” he added.
Those findings were presented at ASH 2018. The drug is now moving to a phase 2 study.
HQP1351 (GZD824)
The orally active, small-molecule BCR-ABL inhibitor HQP1351 is a third-generation TKI with activity against a broad spectrum of BCR-ABL mutations.
A phase 1 study of patients who were resistant to prior TKIs is complete, and results presented at ASH 2019 showed that most patients (67%) had only one or two prior therapies and 63% had T315I mutation. Response rates were better in the patients with T315I mutations (MCyr, 78% vs. 34%; MMR, 52% vs. 15% in 101 chronic phase patients).
The treatment was well tolerated, with grade 3 toxicity involving only hypertriglyceridemia, pyrexia, and proteinuria. No arterial occlusive events were reported.
K0706
K0706 is a selective inhibitor of BCR-ABL1 designed to inhibit enzymatic activity of BCR-ABL. The agent was efficacious and well tolerated with limited off-target activity in preclinical models. It can inhibit wild-type and mutant forms of BCR-ABL, but does not have activity against T315I.
Results of a phase 1 study presented at ASH in 2019 by Dr. Cortes showed that all the patients who received a dose of 174 mg or greater achieved or maintained a cytogenetic response at 6 months, and 50% achieved or maintained an MMR.
“This is a very good response rate in this heavily pretreated population,” he said.
Patients who received prior ponatinib had a somewhat lower response, but still, nearly 45% achieved an MCyr.
“So very good response rates, no arterial occlusive events, and phase 2 studies will be starting at the dose of 174 mg,” he said.
Additional combinations
As for combining TKIs with other agents, efforts are underway around the world to find ways to eradicate minimal residual disease. Examples include TKIs and imatinib, TKIs and azacitidine, and asciminib plus another TKI, to name a few.
One study from Germany showed that adding interferon leads to earlier achievement of MMR, but ultimately the responses were similar, Dr. Cortes said.
Adding venetoclax has shown some activity in the preclinical setting, and studies of that combination will be starting soon in the clinic, he noted.
Implications
The current survival probability in CML patients is 92% when considering CML-related deaths (68% when considering all-cause mortality), compared with 8% in the 1980s and 35%-43% in the early 1990s.
But the current benefits don’t extend to all patients, Dr. Cortes said.
“There are patients who actually end up having worse prognosis than we would expect,” he said, explaining that some CML-related deaths are attributable to lack of access to therapy and good care, but some are related to true poor prognosis, often caused by resistance or inability to tolerate treatments.
In fact, data from studies of various treatments show that almost 40% of patients on dasatinib or nilotinib change therapy by 5 years, and by 10 years, half of those randomized to nilotinib have changed therapy.
“So it is not uncommon that patients have to change therapy for one reason or another,” he said, adding that, as resistance persists through additional treatment options, the prognosis worsens significantly.
“It is important that we have new therapeutic options to be able to help these patients who are going to be in need of additional therapies,” he said.
Dr. Cortes has received grant or research support from Novartis, Pfizer, Takeda, and Sun Pharma, and he is a paid consultant for Pfizer, Novartis, and Takeda.
FROM SOHO 2020
Baseline gene expression predicts TKI response in CML
Baseline gene expression in patients with chronic myeloid leukemia (CML) who received tyrosine kinase inhibitor (TKI) therapy in the phase 3 ENESTnd trial differentiated those who achieved a good response from those with a poor response at 5 years in an exploratory analysis.
The investigators developed gene-expression models based on RNA sequencing of whole blood samples collected prior to treatment with nilotinib or imatinib in study participants who completed at least 5 years of therapy, including both good responders – those who achieved a major molecular response (MMR), defined as BCR-ABL1IS (a gene sequence found in an abnormal chromosome 22) less than 0.01% by 12 months and sustained deep molecular response (DMR) by 5 years, and poor responders – those without MMR by 12 months or with BCR-ABL1IS greater than 10% at 3 months.
A model based on the comparison of gene signatures from 47 patients who achieved a molecular response of 4.5 (MR4.5) on the International Scale (BCR-ABL1S less than 0.00032%), compared with 23 patients with a poor response, best predicted 5-year responder status (area under the receiver operating characteristic curve, 0.76), Jerald P. Radich, MD, reported during the Society of Hematologic Oncology virtual meeting.
“For this kind of work, that’s really quite good,” said Dr. Radich of the clinical research division at Fred Hutchinson Cancer Research Center, Seattle.
Notably, the differences in patient responses observed by 12 months in ENESTnd persisted for up to 10 years, he said.
The findings have potential implications for drug development and facilitation of DMR in patients on TKI therapy – a prerequisite for attempting treatment-free remission, he said.
Dr. Radich and colleagues assessed 24 clinical factors – such as Sokal risk score, TKI therapy type, age, and sex – according to responder status, and applied penalized regression to the clinical variables, to expression of 13,575 genes, and to a combination of the clinical variables and gene expression.
Clinical variables didn’t predict response in the trial, and including the clinical variables in the gene-expression model in the exploratory analysis did not improve it’s performance (AUC, 0.75). However, both the MR4.5 plus clinical variables model and the MR4.5-only model outperformed the clinical variables–only model (AUC, 0.59), he noted, adding: “So gene expression seems to be highly correlated with response.”
Of note, 458 genes were differentially expressed; those found in responders were most often associated with immune response, whereas those in poor responders were more likely to be associated with drug catabolism, WNT signaling, and cell cycle.
This suggests that good responders, compared with poor responders, have an activated immune system that is better able to engage after TKI therapy is administered to “cull through the heard, so to speak,” Dr. Radich said.
The findings were validated in an independent dataset of 19 good responders and 25 poor responders (AUC, 0.67 for the MR4.5 vs. poor-responder model).
A comparison of the expression of immune cell marker genes in good responders and poor responders further showed that T cells – particularly CD8 T cells, B cells, natural killer cells, and aggregate cytotoxic lymphocytes were expressed at significantly higher levels in good responders.
This was true in both the ENESTnd cohort and the validation dataset, he said.
The ENESTnd study is a randomized, open-label study comparing nilotinib and imatinib in adults with newly diagnosed Philadelphia chromosome–positive chronic-phase CML. A 5-year study update published in 2016 showed that 54% and 52% of patients in nilotinib 300- and 400-mg twice-daily arms, respectively, achieved MR4.5, compared with 31% of those in an imatinib 400-mg once-daily arm. In the current exploratory analysis, the gene expression model differentiated between good and poor responders regardless of the TKI used, Dr. Radich said.
The findings are of note because achieving sustained deep molecular response is necessary before CML patients can attempt treatment-free remission and because biomarkers for predicting DMR have been lacking, he explained.
“These findings could really be used, potentially, for a couple of things: One is to predict response, and that could drive patient goals, expectations, and maybe drug choice,” he said.
The findings could also be used to inform clinical trials to investigate how to best treat poor responders to improve their response, he added.
“I think there’s a lot of work to be done and a lot things to chew over, and we’re hoping that we’ll have more to talk to you about in the future,” he said.
The study was sponsored by Novartis. Dr. Radich is a paid consultant for Genentech, Cepheid, Bristol-Myers Squibb, Takeda, and Novartis.
SOURCE: Radich JP et al. SOHO 2020, Abstract CML-109.
Baseline gene expression in patients with chronic myeloid leukemia (CML) who received tyrosine kinase inhibitor (TKI) therapy in the phase 3 ENESTnd trial differentiated those who achieved a good response from those with a poor response at 5 years in an exploratory analysis.
The investigators developed gene-expression models based on RNA sequencing of whole blood samples collected prior to treatment with nilotinib or imatinib in study participants who completed at least 5 years of therapy, including both good responders – those who achieved a major molecular response (MMR), defined as BCR-ABL1IS (a gene sequence found in an abnormal chromosome 22) less than 0.01% by 12 months and sustained deep molecular response (DMR) by 5 years, and poor responders – those without MMR by 12 months or with BCR-ABL1IS greater than 10% at 3 months.
A model based on the comparison of gene signatures from 47 patients who achieved a molecular response of 4.5 (MR4.5) on the International Scale (BCR-ABL1S less than 0.00032%), compared with 23 patients with a poor response, best predicted 5-year responder status (area under the receiver operating characteristic curve, 0.76), Jerald P. Radich, MD, reported during the Society of Hematologic Oncology virtual meeting.
“For this kind of work, that’s really quite good,” said Dr. Radich of the clinical research division at Fred Hutchinson Cancer Research Center, Seattle.
Notably, the differences in patient responses observed by 12 months in ENESTnd persisted for up to 10 years, he said.
The findings have potential implications for drug development and facilitation of DMR in patients on TKI therapy – a prerequisite for attempting treatment-free remission, he said.
Dr. Radich and colleagues assessed 24 clinical factors – such as Sokal risk score, TKI therapy type, age, and sex – according to responder status, and applied penalized regression to the clinical variables, to expression of 13,575 genes, and to a combination of the clinical variables and gene expression.
Clinical variables didn’t predict response in the trial, and including the clinical variables in the gene-expression model in the exploratory analysis did not improve it’s performance (AUC, 0.75). However, both the MR4.5 plus clinical variables model and the MR4.5-only model outperformed the clinical variables–only model (AUC, 0.59), he noted, adding: “So gene expression seems to be highly correlated with response.”
Of note, 458 genes were differentially expressed; those found in responders were most often associated with immune response, whereas those in poor responders were more likely to be associated with drug catabolism, WNT signaling, and cell cycle.
This suggests that good responders, compared with poor responders, have an activated immune system that is better able to engage after TKI therapy is administered to “cull through the heard, so to speak,” Dr. Radich said.
The findings were validated in an independent dataset of 19 good responders and 25 poor responders (AUC, 0.67 for the MR4.5 vs. poor-responder model).
A comparison of the expression of immune cell marker genes in good responders and poor responders further showed that T cells – particularly CD8 T cells, B cells, natural killer cells, and aggregate cytotoxic lymphocytes were expressed at significantly higher levels in good responders.
This was true in both the ENESTnd cohort and the validation dataset, he said.
The ENESTnd study is a randomized, open-label study comparing nilotinib and imatinib in adults with newly diagnosed Philadelphia chromosome–positive chronic-phase CML. A 5-year study update published in 2016 showed that 54% and 52% of patients in nilotinib 300- and 400-mg twice-daily arms, respectively, achieved MR4.5, compared with 31% of those in an imatinib 400-mg once-daily arm. In the current exploratory analysis, the gene expression model differentiated between good and poor responders regardless of the TKI used, Dr. Radich said.
The findings are of note because achieving sustained deep molecular response is necessary before CML patients can attempt treatment-free remission and because biomarkers for predicting DMR have been lacking, he explained.
“These findings could really be used, potentially, for a couple of things: One is to predict response, and that could drive patient goals, expectations, and maybe drug choice,” he said.
The findings could also be used to inform clinical trials to investigate how to best treat poor responders to improve their response, he added.
“I think there’s a lot of work to be done and a lot things to chew over, and we’re hoping that we’ll have more to talk to you about in the future,” he said.
The study was sponsored by Novartis. Dr. Radich is a paid consultant for Genentech, Cepheid, Bristol-Myers Squibb, Takeda, and Novartis.
SOURCE: Radich JP et al. SOHO 2020, Abstract CML-109.
Baseline gene expression in patients with chronic myeloid leukemia (CML) who received tyrosine kinase inhibitor (TKI) therapy in the phase 3 ENESTnd trial differentiated those who achieved a good response from those with a poor response at 5 years in an exploratory analysis.
The investigators developed gene-expression models based on RNA sequencing of whole blood samples collected prior to treatment with nilotinib or imatinib in study participants who completed at least 5 years of therapy, including both good responders – those who achieved a major molecular response (MMR), defined as BCR-ABL1IS (a gene sequence found in an abnormal chromosome 22) less than 0.01% by 12 months and sustained deep molecular response (DMR) by 5 years, and poor responders – those without MMR by 12 months or with BCR-ABL1IS greater than 10% at 3 months.
A model based on the comparison of gene signatures from 47 patients who achieved a molecular response of 4.5 (MR4.5) on the International Scale (BCR-ABL1S less than 0.00032%), compared with 23 patients with a poor response, best predicted 5-year responder status (area under the receiver operating characteristic curve, 0.76), Jerald P. Radich, MD, reported during the Society of Hematologic Oncology virtual meeting.
“For this kind of work, that’s really quite good,” said Dr. Radich of the clinical research division at Fred Hutchinson Cancer Research Center, Seattle.
Notably, the differences in patient responses observed by 12 months in ENESTnd persisted for up to 10 years, he said.
The findings have potential implications for drug development and facilitation of DMR in patients on TKI therapy – a prerequisite for attempting treatment-free remission, he said.
Dr. Radich and colleagues assessed 24 clinical factors – such as Sokal risk score, TKI therapy type, age, and sex – according to responder status, and applied penalized regression to the clinical variables, to expression of 13,575 genes, and to a combination of the clinical variables and gene expression.
Clinical variables didn’t predict response in the trial, and including the clinical variables in the gene-expression model in the exploratory analysis did not improve it’s performance (AUC, 0.75). However, both the MR4.5 plus clinical variables model and the MR4.5-only model outperformed the clinical variables–only model (AUC, 0.59), he noted, adding: “So gene expression seems to be highly correlated with response.”
Of note, 458 genes were differentially expressed; those found in responders were most often associated with immune response, whereas those in poor responders were more likely to be associated with drug catabolism, WNT signaling, and cell cycle.
This suggests that good responders, compared with poor responders, have an activated immune system that is better able to engage after TKI therapy is administered to “cull through the heard, so to speak,” Dr. Radich said.
The findings were validated in an independent dataset of 19 good responders and 25 poor responders (AUC, 0.67 for the MR4.5 vs. poor-responder model).
A comparison of the expression of immune cell marker genes in good responders and poor responders further showed that T cells – particularly CD8 T cells, B cells, natural killer cells, and aggregate cytotoxic lymphocytes were expressed at significantly higher levels in good responders.
This was true in both the ENESTnd cohort and the validation dataset, he said.
The ENESTnd study is a randomized, open-label study comparing nilotinib and imatinib in adults with newly diagnosed Philadelphia chromosome–positive chronic-phase CML. A 5-year study update published in 2016 showed that 54% and 52% of patients in nilotinib 300- and 400-mg twice-daily arms, respectively, achieved MR4.5, compared with 31% of those in an imatinib 400-mg once-daily arm. In the current exploratory analysis, the gene expression model differentiated between good and poor responders regardless of the TKI used, Dr. Radich said.
The findings are of note because achieving sustained deep molecular response is necessary before CML patients can attempt treatment-free remission and because biomarkers for predicting DMR have been lacking, he explained.
“These findings could really be used, potentially, for a couple of things: One is to predict response, and that could drive patient goals, expectations, and maybe drug choice,” he said.
The findings could also be used to inform clinical trials to investigate how to best treat poor responders to improve their response, he added.
“I think there’s a lot of work to be done and a lot things to chew over, and we’re hoping that we’ll have more to talk to you about in the future,” he said.
The study was sponsored by Novartis. Dr. Radich is a paid consultant for Genentech, Cepheid, Bristol-Myers Squibb, Takeda, and Novartis.
SOURCE: Radich JP et al. SOHO 2020, Abstract CML-109.
FROM SOHO 2020