User login
Clinical Progress Note: Decision-making for Tracheostomy Placement in Children With Neurological Impairment
Children with complex medical conditions are living longer, many with the help of interventions and technology, such as gastrostomy tubes, tracheostomies, ventilator support, and parenteral nutrition. Children with medical complexity and technology account for over 80% of hospital days in pediatric academic centers.1
Hospitalists need communication skills and clinical information to guide discussions with patients and families about whether to pursue these measures. Tracheostomy discussions can be particularly challenging. Over 4,000 infants and children undergo tracheostomy each year, with related hospital charges of more than $2 billion, a 30-day readmission rate of 24.9%, and a median length of stay for pneumonia or tracheitis of 4 days.2 There is limited research on prognosis, outcomes, decision-making, and effects on quality of life, especially in the population of children who have significant neurological impairment (NI) and/or progressive or deteriorating neurological conditions. Physician biases may also influence this discussion.
This article will examine the question: How can a hospitalist guide decision-making discussions with families about tracheostomy placement for children with NI? A literature search was performed on Medline and Web of Science using the key terms tracheostomy, prognosis, neurologically impaired children, and decision-making. Articles included were relevant to the clinical question and published in the last 5 years. One article was included outside this timeframe given the scarcity of data.
INDICATIONS FOR TRACHEOSTOMY
Indications for tracheostomy include airway obstruction and the need for prolonged ventilation support.3 The number of tracheostomies placed has been increasing over the last 30 years, especially at tertiary care centers.3 Primary indications for tracheostomy include prolonged ventilation particularly in the context of underlying conditions such as congenital or acquired respiratory disease, congenital or acquired neurologic disease, cardiopulmonary disease, and primary anatomic airway obstruction.3,4 Children who undergo tracheostomy often have multiple medical conditions that impact their overall health and prognosis, with 41% having three or more complex chronic health conditions.5 This article will focus on children who have a primary indication of NI and in whom tracheostomy is often used as a life-prolonging measure.
PROGNOSIS
Discussions about tracheostomy should include information about risks, benefits, and prognosis. Prognosis discussions can be challenging given that many children for whom this intervention is being considered have multiple and complex medical conditions with uncertain or even known poor prognoses. Mortality rates ranging from 3% to 11% have been reported during the initial tracheostomy admission, with NI increasing the risk for mortality during the tracheostomy admission.5,6 Children with NI also have higher mortality beyond the initial hospital stay, lower decannulation rates, and more frequent admissions with longer lengths of stay than do children receiving a tracheostomy for upper airway obstruction (Table 1).6,7

For most children in this population, prognosis is related more to the underlying disease process than to the risk of the surgery for tracheostomy placement itself. Discussions with families should include the anticipated prognosis of the underlying disease, as well as current available data on outcomes for children with neurological impairment who have undergone tracheostomy placement. Most patients who receive a tracheostomy are children with complex medical conditions who have an acute event that leads to airway compromise and respiratory failure underscoring the importance of advance care planning.5
GOALS OF CARE DISCUSSIONS
Clinicians face challenges when initiating advance care planning discussions, including prognostic uncertainty, the perception that families may not want to engage in these discussions, and the complexity and time these discussions can take. In one study of more than 300 chronically ill children, only 17% of parents had discussed advance directives, although 49% reported they would like to create one for their child.9 A small study found that, although parents find these discussions difficult, they also find them important. They value a step by step approach with consideration for hope and nonmedical concerns.10 Advance care planning discussions should be viewed as a time out to clarify what the family sees as the best path forward before initiation of a tracheostomy discussion and decision.
Determining goals of care is a cornerstone of any discussion about tracheostomy placement, especially when a child has a condition that is life limiting. The decision to pursue tracheostomy should involve shared decision-making. This decision-making process is the preferred communication model when multiple options could be pursued, each with its own risks and benefits.10
In this model of decision-making, the family’s goals and values should be determined in the context of the medical intervention that is being pursued. Medical information such as prognosis, risk, benefits, and impact of the intervention on quality of life should all be shared with the family. Ideally, shared decision-making allows the practitioner and family to make a decision together that matches the family’s goals and values with the best option available. If the family’s goal is to prolong life and they feel their child has good quality of life, tracheostomy placement may be the most appropriate option. However, it is also possible that the family’s goals may align more with less invasive treatment options or a transition to comfort care.
Discussions regarding goals of care can be challenging, and involving an interdisciplinary team and a Palliative Care consultant can be helpful.
WHAT PROVIDERS SAY, WHAT FAMILIES NEED TO HEAR
Research on what parents find helpful in discussions about tracheostomy is limited. One study of 56 caregivers found that parents did not feel they could make a “free choice” because the alternative to tracheostomy was death.11 In interviews with caregivers following tracheostomy, this same study found several themes in caregiver perspectives on their decision for tracheostomy (Table 2); caregivers saw a benefit to “health and well-being” from tracheostomy even though they reported feeling unprepared for the caregiving aspect at home or the potential negative side effects. Half the children in this study had a neurologic diagnosis, and only families who chose tracheostomy placement were included. To this author’s knowledge, there are currently no studies that look at decisional themes, satisfaction, or outcomes for families that chose to not pursue tracheostomy.

There is limited literature about how providers discuss tracheostomy. One single-center study of practitioners found that providers focused more often on the benefits of tracheostomy rather than burdens (72% vs 28%).12 A common benefit theme was the provider “suggesting life with a tracheostomy might not be as difficult as families fear in that the child may have the ability to regain speech, engage in normal activities, and have the tracheostomy reversed once the child’s health improved.” However, decannulation rates and recovery trajectories for children with NI do not support this general expectation (Table 1). These provider communication themes may help to explain the family’s perspective that they feel unprepared for the burdens of a tracheostomy or the intensity of home caregiving. Given the limited data, it is difficult to generalize. Comparing communication and decision-making themes side by side does draw attention to how providers might better communicate with families about this decision (Table 2).
The difficult aspects of caregiving deserve special attention. A study of 25 parents showed reduced parental quality of life after their child’s tracheostomy placement related to overwhelming medical care, fear of death of the child requiring constant vigilance, and financial and psychological stressors.13 Most (72%) families in this study reported decisional regret at 3 months.Resources and support for a child with this level of care varies based on the child’s community. Exploration and discussion of what is available for each family, including home nursing, respite, and/or a skilled nursing facility, should be completed prior to tracheostomy placement. Honest discussions about the potential effects of this intervention on the family’s life can help inform their decision.
Decision-making tools for tracheostomy could be valuable for both families and clinicians. These tools allow for a more systematic approach to the decision-making process that takes into account the multidimensional aspects of this decision. The “Child Tracheostomy Decision Guide,” published by the Winnipeg Regional Health Authority, is one available tool.14 This tool guides families through the factors that may affect their decision-making and includes thoughts about goals of care, quality of life, prognosis, care at home, and other options such as comfort care. The Courageous Parents Network has also developed parent videos giving the perspective of parents who have chosen or not chosen tracheostomy.15 Currently, there are no studies that examine the usefulness of decision-making tools.
GAPS IN LITERATURE
A common theme throughout the literature is the lack of a unifying classification system for reporting outcomes data. Each study utilizes different primary indications for tracheostomy and often different definitions for NI. There is very little literature that focuses specifically on outcomes for children with NI who receive tracheostomy as a life-prolonging measure. These gaps present challenges for obtaining meaningful prognosis data to share with families. Outcomes data for children who do not receive tracheostomy is also lacking. Additional studies on how families make this decision and their decisional satisfaction could help inform the decision-making process for both parents and clinicians. Research regarding the helpfulness and outcomes with decision-making tools would be useful.
CONCLUSIONS
Although there are limited data on outcomes specific to the children with NI and tracheostomy, existing literature shows a higher mortality, lower decannulation rate, higher hospitalization rate, and longer length of stay than that for children who receive tracheostomy for other indications. Tracheostomy is often a life-prolonging measure for children with NI. Shared decision-making should be the preferred communication process and include defining goals of care, as well as anticipated prognosis with balanced information about risks and benefits. Further research about the decision-making process and communication would be beneficial.
DISCLOSURE
Dr Shaw has nothing to disclose.
1. Children’s Hospital Association. Spend for children with dominant chronic diseases – The CARE award. Historical spending: 2012-2014. https://www.childrenshospitals.org/Care/Children-With-Medical-Complexity 2018
2. Russel CJ, Mack WJ, Schrager SM, Wu S. Care variations, length of stay and readmissions in children hospitalized for bacterial tracheostomy-associated respiratory infections. Hosp Pediatr. 2017;7(1):16-23. https://doi.org/10.1542/hpeds.2016-0104
3. McPherson ML, Shekerdemian L, Goldsworthy M, et al. A decade of pediatric tracheostomies: indications, outcomes, and long-term prognosis. Pediatr Pulmonol. 2017;52(7):946-953. https://doi.org/10.1002/ppul.23657
4. Gergin O, Adil EA, Kawai K, Watters K, Moritz E, Rahbar R. Indications of pediatric tracheostomy over the last 30 years: has anything changed? Int J Pediatr Otorhinolaryngol. 2016;87:144-147. https://doi.org/10.1016/j.ijporl.2016.06.018
5. Edwards J, Houtrow A, Lucas A, et al. Children and young adults who receive tracheostomies or were initiated on long-term ventilation in PICUs. Pediatr Crit Care Med. 2016;17(8):e324-334. https://doi.org/10.1097/pcc.0000000000000844
6. Berry JG, Graham DA, Graham RJ, et al. Predictors of clinical outcomes and hospital resource use of children after tracheotomy. Pediatrics. 2009;124(2):563-572. https://doi.org/10.1542/peds.2008-3491
7. Tsuboi N, Ide K, Nishimura N, Nakagawa S, Morimoto N. Pediatric tracheostomy: survival and long-term outcomes. Int J Pediatr Otorhinolaryngol. 2016;89:81-85. https://doi.org/10.1016/j.ijporl.2016.07.033
8. Liberman DB, Pham PK, Nager AL. Pediatric advance directives: parents’ knowledge, experience, and preferences. Pediatrics. 2014;134(2):e436-e443. https://doi.org/10.1542/peds.2013-3124
9. Lotz JD, Daxer M, Jox RJ, Borasio GD, Führer M. “Hope for the best, prepare for the worst”: a qualitative interview study on parents’ needs and fears in pediatric advance care planning. Palliat Med. 2017;31(8):764-771. https://doi.org/10.1177/0269216316679913
10. Nelson KE, Mahant S. Shared decision-making about assistive technology for the child with severe neurologic impairment. Pediatr Clin North Am. 2014;61(4):641-652. https://doi.org/10.1016/j.pcl.2014.04.001
11. Nageswaran S, Golden SL, Gower WA, King NMP. Caregiver perceptions about their decision to pursue tracheostomy for children with medical complexity. J Pediatr. 2018;203:354-360.e1. https://doi.org/10.1016/j.jpeds.2018.07.045
12. Hebert LM, Watson AC, Madrigal V, October TW. Discussing benefits and risks of tracheostomy: what physicians actually say. Pediatr Crit Care Med. 2017;18(12):e592-e597. https://doi.org/10.1097/PCC.0000000000001341
13. October T, Jones A, Michals H, Hebert L, Jiang J, Wang J. Parental conflict, regret, and short-term impact on quality of life in tracheostomy decision making. Pediatr Crit Care Med. 2020;21(2):136-142. https://doi.org/10.1097/PCC.0000000000002109
14. Winnipeg Regional Health Authority. Childhood Tracheostomy Decision Guide. Accessed August 15, 2019. https://www.wrha.mb.ca/extranet/eipt/files/EIPT-023-001.pdf
15. Courageous Parents Network. Tracheostomy Decision Making Videos. Accessed August 20, 2019. https://courageousparentsnetwork.org/video-library/decision-making/tracheostomy
Children with complex medical conditions are living longer, many with the help of interventions and technology, such as gastrostomy tubes, tracheostomies, ventilator support, and parenteral nutrition. Children with medical complexity and technology account for over 80% of hospital days in pediatric academic centers.1
Hospitalists need communication skills and clinical information to guide discussions with patients and families about whether to pursue these measures. Tracheostomy discussions can be particularly challenging. Over 4,000 infants and children undergo tracheostomy each year, with related hospital charges of more than $2 billion, a 30-day readmission rate of 24.9%, and a median length of stay for pneumonia or tracheitis of 4 days.2 There is limited research on prognosis, outcomes, decision-making, and effects on quality of life, especially in the population of children who have significant neurological impairment (NI) and/or progressive or deteriorating neurological conditions. Physician biases may also influence this discussion.
This article will examine the question: How can a hospitalist guide decision-making discussions with families about tracheostomy placement for children with NI? A literature search was performed on Medline and Web of Science using the key terms tracheostomy, prognosis, neurologically impaired children, and decision-making. Articles included were relevant to the clinical question and published in the last 5 years. One article was included outside this timeframe given the scarcity of data.
INDICATIONS FOR TRACHEOSTOMY
Indications for tracheostomy include airway obstruction and the need for prolonged ventilation support.3 The number of tracheostomies placed has been increasing over the last 30 years, especially at tertiary care centers.3 Primary indications for tracheostomy include prolonged ventilation particularly in the context of underlying conditions such as congenital or acquired respiratory disease, congenital or acquired neurologic disease, cardiopulmonary disease, and primary anatomic airway obstruction.3,4 Children who undergo tracheostomy often have multiple medical conditions that impact their overall health and prognosis, with 41% having three or more complex chronic health conditions.5 This article will focus on children who have a primary indication of NI and in whom tracheostomy is often used as a life-prolonging measure.
PROGNOSIS
Discussions about tracheostomy should include information about risks, benefits, and prognosis. Prognosis discussions can be challenging given that many children for whom this intervention is being considered have multiple and complex medical conditions with uncertain or even known poor prognoses. Mortality rates ranging from 3% to 11% have been reported during the initial tracheostomy admission, with NI increasing the risk for mortality during the tracheostomy admission.5,6 Children with NI also have higher mortality beyond the initial hospital stay, lower decannulation rates, and more frequent admissions with longer lengths of stay than do children receiving a tracheostomy for upper airway obstruction (Table 1).6,7

For most children in this population, prognosis is related more to the underlying disease process than to the risk of the surgery for tracheostomy placement itself. Discussions with families should include the anticipated prognosis of the underlying disease, as well as current available data on outcomes for children with neurological impairment who have undergone tracheostomy placement. Most patients who receive a tracheostomy are children with complex medical conditions who have an acute event that leads to airway compromise and respiratory failure underscoring the importance of advance care planning.5
GOALS OF CARE DISCUSSIONS
Clinicians face challenges when initiating advance care planning discussions, including prognostic uncertainty, the perception that families may not want to engage in these discussions, and the complexity and time these discussions can take. In one study of more than 300 chronically ill children, only 17% of parents had discussed advance directives, although 49% reported they would like to create one for their child.9 A small study found that, although parents find these discussions difficult, they also find them important. They value a step by step approach with consideration for hope and nonmedical concerns.10 Advance care planning discussions should be viewed as a time out to clarify what the family sees as the best path forward before initiation of a tracheostomy discussion and decision.
Determining goals of care is a cornerstone of any discussion about tracheostomy placement, especially when a child has a condition that is life limiting. The decision to pursue tracheostomy should involve shared decision-making. This decision-making process is the preferred communication model when multiple options could be pursued, each with its own risks and benefits.10
In this model of decision-making, the family’s goals and values should be determined in the context of the medical intervention that is being pursued. Medical information such as prognosis, risk, benefits, and impact of the intervention on quality of life should all be shared with the family. Ideally, shared decision-making allows the practitioner and family to make a decision together that matches the family’s goals and values with the best option available. If the family’s goal is to prolong life and they feel their child has good quality of life, tracheostomy placement may be the most appropriate option. However, it is also possible that the family’s goals may align more with less invasive treatment options or a transition to comfort care.
Discussions regarding goals of care can be challenging, and involving an interdisciplinary team and a Palliative Care consultant can be helpful.
WHAT PROVIDERS SAY, WHAT FAMILIES NEED TO HEAR
Research on what parents find helpful in discussions about tracheostomy is limited. One study of 56 caregivers found that parents did not feel they could make a “free choice” because the alternative to tracheostomy was death.11 In interviews with caregivers following tracheostomy, this same study found several themes in caregiver perspectives on their decision for tracheostomy (Table 2); caregivers saw a benefit to “health and well-being” from tracheostomy even though they reported feeling unprepared for the caregiving aspect at home or the potential negative side effects. Half the children in this study had a neurologic diagnosis, and only families who chose tracheostomy placement were included. To this author’s knowledge, there are currently no studies that look at decisional themes, satisfaction, or outcomes for families that chose to not pursue tracheostomy.

There is limited literature about how providers discuss tracheostomy. One single-center study of practitioners found that providers focused more often on the benefits of tracheostomy rather than burdens (72% vs 28%).12 A common benefit theme was the provider “suggesting life with a tracheostomy might not be as difficult as families fear in that the child may have the ability to regain speech, engage in normal activities, and have the tracheostomy reversed once the child’s health improved.” However, decannulation rates and recovery trajectories for children with NI do not support this general expectation (Table 1). These provider communication themes may help to explain the family’s perspective that they feel unprepared for the burdens of a tracheostomy or the intensity of home caregiving. Given the limited data, it is difficult to generalize. Comparing communication and decision-making themes side by side does draw attention to how providers might better communicate with families about this decision (Table 2).
The difficult aspects of caregiving deserve special attention. A study of 25 parents showed reduced parental quality of life after their child’s tracheostomy placement related to overwhelming medical care, fear of death of the child requiring constant vigilance, and financial and psychological stressors.13 Most (72%) families in this study reported decisional regret at 3 months.Resources and support for a child with this level of care varies based on the child’s community. Exploration and discussion of what is available for each family, including home nursing, respite, and/or a skilled nursing facility, should be completed prior to tracheostomy placement. Honest discussions about the potential effects of this intervention on the family’s life can help inform their decision.
Decision-making tools for tracheostomy could be valuable for both families and clinicians. These tools allow for a more systematic approach to the decision-making process that takes into account the multidimensional aspects of this decision. The “Child Tracheostomy Decision Guide,” published by the Winnipeg Regional Health Authority, is one available tool.14 This tool guides families through the factors that may affect their decision-making and includes thoughts about goals of care, quality of life, prognosis, care at home, and other options such as comfort care. The Courageous Parents Network has also developed parent videos giving the perspective of parents who have chosen or not chosen tracheostomy.15 Currently, there are no studies that examine the usefulness of decision-making tools.
GAPS IN LITERATURE
A common theme throughout the literature is the lack of a unifying classification system for reporting outcomes data. Each study utilizes different primary indications for tracheostomy and often different definitions for NI. There is very little literature that focuses specifically on outcomes for children with NI who receive tracheostomy as a life-prolonging measure. These gaps present challenges for obtaining meaningful prognosis data to share with families. Outcomes data for children who do not receive tracheostomy is also lacking. Additional studies on how families make this decision and their decisional satisfaction could help inform the decision-making process for both parents and clinicians. Research regarding the helpfulness and outcomes with decision-making tools would be useful.
CONCLUSIONS
Although there are limited data on outcomes specific to the children with NI and tracheostomy, existing literature shows a higher mortality, lower decannulation rate, higher hospitalization rate, and longer length of stay than that for children who receive tracheostomy for other indications. Tracheostomy is often a life-prolonging measure for children with NI. Shared decision-making should be the preferred communication process and include defining goals of care, as well as anticipated prognosis with balanced information about risks and benefits. Further research about the decision-making process and communication would be beneficial.
DISCLOSURE
Dr Shaw has nothing to disclose.
Children with complex medical conditions are living longer, many with the help of interventions and technology, such as gastrostomy tubes, tracheostomies, ventilator support, and parenteral nutrition. Children with medical complexity and technology account for over 80% of hospital days in pediatric academic centers.1
Hospitalists need communication skills and clinical information to guide discussions with patients and families about whether to pursue these measures. Tracheostomy discussions can be particularly challenging. Over 4,000 infants and children undergo tracheostomy each year, with related hospital charges of more than $2 billion, a 30-day readmission rate of 24.9%, and a median length of stay for pneumonia or tracheitis of 4 days.2 There is limited research on prognosis, outcomes, decision-making, and effects on quality of life, especially in the population of children who have significant neurological impairment (NI) and/or progressive or deteriorating neurological conditions. Physician biases may also influence this discussion.
This article will examine the question: How can a hospitalist guide decision-making discussions with families about tracheostomy placement for children with NI? A literature search was performed on Medline and Web of Science using the key terms tracheostomy, prognosis, neurologically impaired children, and decision-making. Articles included were relevant to the clinical question and published in the last 5 years. One article was included outside this timeframe given the scarcity of data.
INDICATIONS FOR TRACHEOSTOMY
Indications for tracheostomy include airway obstruction and the need for prolonged ventilation support.3 The number of tracheostomies placed has been increasing over the last 30 years, especially at tertiary care centers.3 Primary indications for tracheostomy include prolonged ventilation particularly in the context of underlying conditions such as congenital or acquired respiratory disease, congenital or acquired neurologic disease, cardiopulmonary disease, and primary anatomic airway obstruction.3,4 Children who undergo tracheostomy often have multiple medical conditions that impact their overall health and prognosis, with 41% having three or more complex chronic health conditions.5 This article will focus on children who have a primary indication of NI and in whom tracheostomy is often used as a life-prolonging measure.
PROGNOSIS
Discussions about tracheostomy should include information about risks, benefits, and prognosis. Prognosis discussions can be challenging given that many children for whom this intervention is being considered have multiple and complex medical conditions with uncertain or even known poor prognoses. Mortality rates ranging from 3% to 11% have been reported during the initial tracheostomy admission, with NI increasing the risk for mortality during the tracheostomy admission.5,6 Children with NI also have higher mortality beyond the initial hospital stay, lower decannulation rates, and more frequent admissions with longer lengths of stay than do children receiving a tracheostomy for upper airway obstruction (Table 1).6,7

For most children in this population, prognosis is related more to the underlying disease process than to the risk of the surgery for tracheostomy placement itself. Discussions with families should include the anticipated prognosis of the underlying disease, as well as current available data on outcomes for children with neurological impairment who have undergone tracheostomy placement. Most patients who receive a tracheostomy are children with complex medical conditions who have an acute event that leads to airway compromise and respiratory failure underscoring the importance of advance care planning.5
GOALS OF CARE DISCUSSIONS
Clinicians face challenges when initiating advance care planning discussions, including prognostic uncertainty, the perception that families may not want to engage in these discussions, and the complexity and time these discussions can take. In one study of more than 300 chronically ill children, only 17% of parents had discussed advance directives, although 49% reported they would like to create one for their child.9 A small study found that, although parents find these discussions difficult, they also find them important. They value a step by step approach with consideration for hope and nonmedical concerns.10 Advance care planning discussions should be viewed as a time out to clarify what the family sees as the best path forward before initiation of a tracheostomy discussion and decision.
Determining goals of care is a cornerstone of any discussion about tracheostomy placement, especially when a child has a condition that is life limiting. The decision to pursue tracheostomy should involve shared decision-making. This decision-making process is the preferred communication model when multiple options could be pursued, each with its own risks and benefits.10
In this model of decision-making, the family’s goals and values should be determined in the context of the medical intervention that is being pursued. Medical information such as prognosis, risk, benefits, and impact of the intervention on quality of life should all be shared with the family. Ideally, shared decision-making allows the practitioner and family to make a decision together that matches the family’s goals and values with the best option available. If the family’s goal is to prolong life and they feel their child has good quality of life, tracheostomy placement may be the most appropriate option. However, it is also possible that the family’s goals may align more with less invasive treatment options or a transition to comfort care.
Discussions regarding goals of care can be challenging, and involving an interdisciplinary team and a Palliative Care consultant can be helpful.
WHAT PROVIDERS SAY, WHAT FAMILIES NEED TO HEAR
Research on what parents find helpful in discussions about tracheostomy is limited. One study of 56 caregivers found that parents did not feel they could make a “free choice” because the alternative to tracheostomy was death.11 In interviews with caregivers following tracheostomy, this same study found several themes in caregiver perspectives on their decision for tracheostomy (Table 2); caregivers saw a benefit to “health and well-being” from tracheostomy even though they reported feeling unprepared for the caregiving aspect at home or the potential negative side effects. Half the children in this study had a neurologic diagnosis, and only families who chose tracheostomy placement were included. To this author’s knowledge, there are currently no studies that look at decisional themes, satisfaction, or outcomes for families that chose to not pursue tracheostomy.

There is limited literature about how providers discuss tracheostomy. One single-center study of practitioners found that providers focused more often on the benefits of tracheostomy rather than burdens (72% vs 28%).12 A common benefit theme was the provider “suggesting life with a tracheostomy might not be as difficult as families fear in that the child may have the ability to regain speech, engage in normal activities, and have the tracheostomy reversed once the child’s health improved.” However, decannulation rates and recovery trajectories for children with NI do not support this general expectation (Table 1). These provider communication themes may help to explain the family’s perspective that they feel unprepared for the burdens of a tracheostomy or the intensity of home caregiving. Given the limited data, it is difficult to generalize. Comparing communication and decision-making themes side by side does draw attention to how providers might better communicate with families about this decision (Table 2).
The difficult aspects of caregiving deserve special attention. A study of 25 parents showed reduced parental quality of life after their child’s tracheostomy placement related to overwhelming medical care, fear of death of the child requiring constant vigilance, and financial and psychological stressors.13 Most (72%) families in this study reported decisional regret at 3 months.Resources and support for a child with this level of care varies based on the child’s community. Exploration and discussion of what is available for each family, including home nursing, respite, and/or a skilled nursing facility, should be completed prior to tracheostomy placement. Honest discussions about the potential effects of this intervention on the family’s life can help inform their decision.
Decision-making tools for tracheostomy could be valuable for both families and clinicians. These tools allow for a more systematic approach to the decision-making process that takes into account the multidimensional aspects of this decision. The “Child Tracheostomy Decision Guide,” published by the Winnipeg Regional Health Authority, is one available tool.14 This tool guides families through the factors that may affect their decision-making and includes thoughts about goals of care, quality of life, prognosis, care at home, and other options such as comfort care. The Courageous Parents Network has also developed parent videos giving the perspective of parents who have chosen or not chosen tracheostomy.15 Currently, there are no studies that examine the usefulness of decision-making tools.
GAPS IN LITERATURE
A common theme throughout the literature is the lack of a unifying classification system for reporting outcomes data. Each study utilizes different primary indications for tracheostomy and often different definitions for NI. There is very little literature that focuses specifically on outcomes for children with NI who receive tracheostomy as a life-prolonging measure. These gaps present challenges for obtaining meaningful prognosis data to share with families. Outcomes data for children who do not receive tracheostomy is also lacking. Additional studies on how families make this decision and their decisional satisfaction could help inform the decision-making process for both parents and clinicians. Research regarding the helpfulness and outcomes with decision-making tools would be useful.
CONCLUSIONS
Although there are limited data on outcomes specific to the children with NI and tracheostomy, existing literature shows a higher mortality, lower decannulation rate, higher hospitalization rate, and longer length of stay than that for children who receive tracheostomy for other indications. Tracheostomy is often a life-prolonging measure for children with NI. Shared decision-making should be the preferred communication process and include defining goals of care, as well as anticipated prognosis with balanced information about risks and benefits. Further research about the decision-making process and communication would be beneficial.
DISCLOSURE
Dr Shaw has nothing to disclose.
1. Children’s Hospital Association. Spend for children with dominant chronic diseases – The CARE award. Historical spending: 2012-2014. https://www.childrenshospitals.org/Care/Children-With-Medical-Complexity 2018
2. Russel CJ, Mack WJ, Schrager SM, Wu S. Care variations, length of stay and readmissions in children hospitalized for bacterial tracheostomy-associated respiratory infections. Hosp Pediatr. 2017;7(1):16-23. https://doi.org/10.1542/hpeds.2016-0104
3. McPherson ML, Shekerdemian L, Goldsworthy M, et al. A decade of pediatric tracheostomies: indications, outcomes, and long-term prognosis. Pediatr Pulmonol. 2017;52(7):946-953. https://doi.org/10.1002/ppul.23657
4. Gergin O, Adil EA, Kawai K, Watters K, Moritz E, Rahbar R. Indications of pediatric tracheostomy over the last 30 years: has anything changed? Int J Pediatr Otorhinolaryngol. 2016;87:144-147. https://doi.org/10.1016/j.ijporl.2016.06.018
5. Edwards J, Houtrow A, Lucas A, et al. Children and young adults who receive tracheostomies or were initiated on long-term ventilation in PICUs. Pediatr Crit Care Med. 2016;17(8):e324-334. https://doi.org/10.1097/pcc.0000000000000844
6. Berry JG, Graham DA, Graham RJ, et al. Predictors of clinical outcomes and hospital resource use of children after tracheotomy. Pediatrics. 2009;124(2):563-572. https://doi.org/10.1542/peds.2008-3491
7. Tsuboi N, Ide K, Nishimura N, Nakagawa S, Morimoto N. Pediatric tracheostomy: survival and long-term outcomes. Int J Pediatr Otorhinolaryngol. 2016;89:81-85. https://doi.org/10.1016/j.ijporl.2016.07.033
8. Liberman DB, Pham PK, Nager AL. Pediatric advance directives: parents’ knowledge, experience, and preferences. Pediatrics. 2014;134(2):e436-e443. https://doi.org/10.1542/peds.2013-3124
9. Lotz JD, Daxer M, Jox RJ, Borasio GD, Führer M. “Hope for the best, prepare for the worst”: a qualitative interview study on parents’ needs and fears in pediatric advance care planning. Palliat Med. 2017;31(8):764-771. https://doi.org/10.1177/0269216316679913
10. Nelson KE, Mahant S. Shared decision-making about assistive technology for the child with severe neurologic impairment. Pediatr Clin North Am. 2014;61(4):641-652. https://doi.org/10.1016/j.pcl.2014.04.001
11. Nageswaran S, Golden SL, Gower WA, King NMP. Caregiver perceptions about their decision to pursue tracheostomy for children with medical complexity. J Pediatr. 2018;203:354-360.e1. https://doi.org/10.1016/j.jpeds.2018.07.045
12. Hebert LM, Watson AC, Madrigal V, October TW. Discussing benefits and risks of tracheostomy: what physicians actually say. Pediatr Crit Care Med. 2017;18(12):e592-e597. https://doi.org/10.1097/PCC.0000000000001341
13. October T, Jones A, Michals H, Hebert L, Jiang J, Wang J. Parental conflict, regret, and short-term impact on quality of life in tracheostomy decision making. Pediatr Crit Care Med. 2020;21(2):136-142. https://doi.org/10.1097/PCC.0000000000002109
14. Winnipeg Regional Health Authority. Childhood Tracheostomy Decision Guide. Accessed August 15, 2019. https://www.wrha.mb.ca/extranet/eipt/files/EIPT-023-001.pdf
15. Courageous Parents Network. Tracheostomy Decision Making Videos. Accessed August 20, 2019. https://courageousparentsnetwork.org/video-library/decision-making/tracheostomy
1. Children’s Hospital Association. Spend for children with dominant chronic diseases – The CARE award. Historical spending: 2012-2014. https://www.childrenshospitals.org/Care/Children-With-Medical-Complexity 2018
2. Russel CJ, Mack WJ, Schrager SM, Wu S. Care variations, length of stay and readmissions in children hospitalized for bacterial tracheostomy-associated respiratory infections. Hosp Pediatr. 2017;7(1):16-23. https://doi.org/10.1542/hpeds.2016-0104
3. McPherson ML, Shekerdemian L, Goldsworthy M, et al. A decade of pediatric tracheostomies: indications, outcomes, and long-term prognosis. Pediatr Pulmonol. 2017;52(7):946-953. https://doi.org/10.1002/ppul.23657
4. Gergin O, Adil EA, Kawai K, Watters K, Moritz E, Rahbar R. Indications of pediatric tracheostomy over the last 30 years: has anything changed? Int J Pediatr Otorhinolaryngol. 2016;87:144-147. https://doi.org/10.1016/j.ijporl.2016.06.018
5. Edwards J, Houtrow A, Lucas A, et al. Children and young adults who receive tracheostomies or were initiated on long-term ventilation in PICUs. Pediatr Crit Care Med. 2016;17(8):e324-334. https://doi.org/10.1097/pcc.0000000000000844
6. Berry JG, Graham DA, Graham RJ, et al. Predictors of clinical outcomes and hospital resource use of children after tracheotomy. Pediatrics. 2009;124(2):563-572. https://doi.org/10.1542/peds.2008-3491
7. Tsuboi N, Ide K, Nishimura N, Nakagawa S, Morimoto N. Pediatric tracheostomy: survival and long-term outcomes. Int J Pediatr Otorhinolaryngol. 2016;89:81-85. https://doi.org/10.1016/j.ijporl.2016.07.033
8. Liberman DB, Pham PK, Nager AL. Pediatric advance directives: parents’ knowledge, experience, and preferences. Pediatrics. 2014;134(2):e436-e443. https://doi.org/10.1542/peds.2013-3124
9. Lotz JD, Daxer M, Jox RJ, Borasio GD, Führer M. “Hope for the best, prepare for the worst”: a qualitative interview study on parents’ needs and fears in pediatric advance care planning. Palliat Med. 2017;31(8):764-771. https://doi.org/10.1177/0269216316679913
10. Nelson KE, Mahant S. Shared decision-making about assistive technology for the child with severe neurologic impairment. Pediatr Clin North Am. 2014;61(4):641-652. https://doi.org/10.1016/j.pcl.2014.04.001
11. Nageswaran S, Golden SL, Gower WA, King NMP. Caregiver perceptions about their decision to pursue tracheostomy for children with medical complexity. J Pediatr. 2018;203:354-360.e1. https://doi.org/10.1016/j.jpeds.2018.07.045
12. Hebert LM, Watson AC, Madrigal V, October TW. Discussing benefits and risks of tracheostomy: what physicians actually say. Pediatr Crit Care Med. 2017;18(12):e592-e597. https://doi.org/10.1097/PCC.0000000000001341
13. October T, Jones A, Michals H, Hebert L, Jiang J, Wang J. Parental conflict, regret, and short-term impact on quality of life in tracheostomy decision making. Pediatr Crit Care Med. 2020;21(2):136-142. https://doi.org/10.1097/PCC.0000000000002109
14. Winnipeg Regional Health Authority. Childhood Tracheostomy Decision Guide. Accessed August 15, 2019. https://www.wrha.mb.ca/extranet/eipt/files/EIPT-023-001.pdf
15. Courageous Parents Network. Tracheostomy Decision Making Videos. Accessed August 20, 2019. https://courageousparentsnetwork.org/video-library/decision-making/tracheostomy
© 2020 Society of Hospital Medicine
Clinical Progress Note: Procalcitonin in the Identification of Invasive Bacterial Infections in Febrile Young Infants
Febrile infants 60 days of age or younger pose a significant diagnostic challenge for clinicians. Most of these infants are well appearing and do not have localizing signs or symptoms of infection, yet they may have serious bacterial infections (SBI) such as urinary tract infection (UTI), bacteremia, and meningitis. While urinalysis is highly sensitive for predicting UTI,1 older clinical decision rules and biomarkers such as white blood cell (WBC) count, absolute neutrophil count (ANC), and C-reactive protein (CRP) lack both appropriate sensitivity and specificity for identifying bacteremia and meningitis (ie, invasive bacterial infection [IBI]),2,3 which affect approximately 2.4% and 0.9% of febrile infants during the first 2 months of life, respectively.4 The lack of accurate diagnostic markers can drive overuse of laboratory testing, antibiotics, and hospitalization despite the low rates of these infections. As a result, procalcitonin (PCT) has generated interest because of its potential to serve as a more accurate biomarker for bacterial infections. This review summarizes recent literature on the diagnostic utility of PCT in the identification of IBI in febrile young infants 60 days or younger.
MECHANISM OF PROCALCITONIN
Procalcitonin is undetectable in noninflammatory states but can be detected in the blood within 4 to 6 hours after initial bacterial infection.5 Its production is stimulated throughout various tissues of the body by cytokines such as interleukin-6 and tumor necrosis factor, which are produced in response to bacterial infections. Interferon-γ, which is produced in response to viral infections, attenuates PCT production. While these characteristics suggest promise for PCT as a more specific screening test for underlying bacterial infection, there are caveats. PCT levels are physiologically elevated in the first 48 hours of life and vary with gestational age, factors that should be considered when interpreting results.6 Additionally, PCT levels can rise in other inflammatory states such as autoimmune conditions and certain malignancies,5 though these states are unlikely to confound the evaluation of febrile young infants.
DIAGNOSTIC ACCURACY OF PROCALCITONIN
Because of PCT’s potential to be more specific than other commonly used biomarkers, multiple studies have evaluated its performance characteristics in febrile young infants. Gomez et al retrospectively evaluated 1,112 well-appearing infants younger than 3 months with fever without a source in seven European emergency departments (EDs).7 Overall, 23 infants (2.1%) had IBI (1 with meningitis). A PCT level of 0.5 ng/mL or greater was the only independent risk factor for IBI (adjusted odds ratio, 21.69; 95% CI, 7.93-59.28). Four infants with IBI had a PCT level less than 0.5 ng/mL, and none of these four had meningitis. PCT was superior to CRP, ANC, and WBC in detecting IBI (area under the curve [AUC], 0.825; 95% CI, 0.698-0.952). PCT was the also the best marker for identifying IBI among 451 infants with a normal urine dipstick and fever detected ≤6 hours before presentation (AUC, 0.819; 95% CI, 0.551-1.087).
In the largest prospective study to date evaluating the diagnostic accuracy of PCT in febrile young infants, Milcent et al studied 2,047 previously healthy infants aged 7-91 days admitted for fever from 15 French EDs.8 In total, 21 (1%) had an IBI (8 with meningitis). PCT performed better than CRP, ANC, and WBC for the detection of IBI with an AUC of 0.91 (95% CI, 0.83-0.99). In a multivariable model, a PCT level of 0.3 ng/mL or greater was the only independent risk factor for IBI with an adjusted odds ratio of 40.3 (95% CI, 5.0-332). Only one infant with IBI had a PCT level less than 0.3 ng/mL. This infant was 83 days old, had 4 hours of fever, and became afebrile spontaneously prior to the blood culture revealing Streptococcus pneumoniae. PCT also performed better than CRP in the detection of IBI in infants 7-30 days of age and those with fever for less than 6 hours, though both subgroups had small numbers of infants with IBI. The authors determined that a PCT level of 0.3 ng/mL was the optimal cutoff for ruling out IBI; this cutoff had a sensitivity of 90% and negative likelihood ratio (LR) of 0.1 (Table). In contrast, the more commonly studied PCT cutoff of 0.5 ng/mL increased the negative LR to 0.2. The authors suggested that PCT, when used in the context of history, exam, and tests such as urinalysis, could identify infants at low risk of IBI.
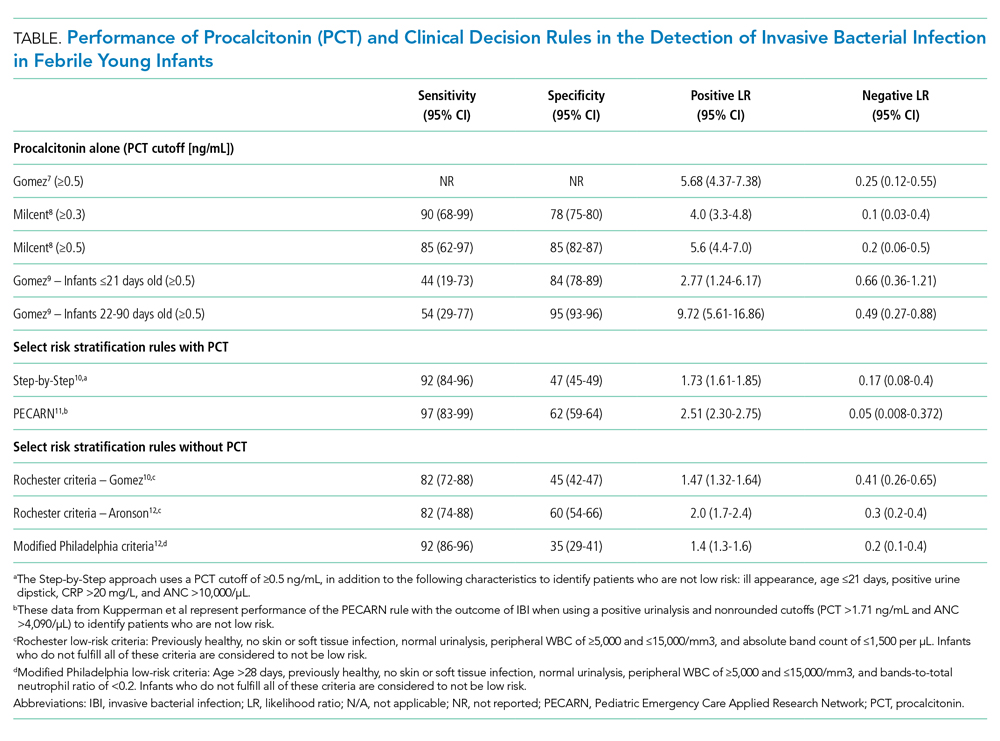
Gomez et al conducted a prospective, single-center study of well-appearing infants with fever without a source and negative urine dipsticks.9 They identified IBI in 9 of 196 infants (4.5%) 21 days or younger and 13 of 1,331 infants (1.0%) 22-90 days old. PCT was superior to CRP and ANC for IBI detection in both age groups. However, in infants 21 days or younger, both the positive and negative LRs for PCT levels of 0.5 ng/mL or greater were poor (Table). Differences in results from the prior two studies7,8 may be related to smaller sample size and differences in patient population because this study included infants younger than 7 days and a higher proportion of infants presenting within 6 hours of fever.
CLINICAL DECISION RULES
PCT has also been incorporated into clinical decision rules for febrile young infants, primarily to identify those at low risk of either IBI or SBI. The Step-by-Step approach10 classified well-appearing febrile infants 90 days or younger as having a high risk of IBI if they were ill appearing, younger than 21 days old, had a positive urine dipstick or a PCT level of 0.5 ng/mL or greater, and classified them as intermediate risk if they had a CRP level greater than 20 mg/L or ANC level greater than 10,000/µL. The remaining infants were classified as low risk and could be managed as outpatients without lumbar puncture or empiric antibiotics. Of note, derivation of this rule excluded patients with respiratory signs or symptoms. In a prospective validation study with 2,185 infants from 11 European EDs, 87 (4.0%) had an IBI (10 with bacterial meningitis). Sequentially identifying patients as high risk using general appearance, age, and urine dipstick alone identified 80% of infants with IBI and 90% of those with bacterial meningitis. The remaining case of meningitis would have been detected by an elevated PCT. A total of 7 of 991 infants (0.7%) classified as low risk had an IBI and none had meningitis. Six of these infants had a fever duration of less than 2 hours, which would not be enough time for PCT to rise. The Step-by-Step approach, with a sensitivity of 92% and negative LR of 0.17, performed well in the ability to rule out IBI.
A clinical prediction rule developed by the Pediatric Emergency Care Applied Research Network (PECARN) found that urinalysis, ANC, and PCT performed well in identifying infants 60 days or younger at low risk for SBI and IBI.11 This prospective observational study of 1,821 infants 60 days or younger in 26 US EDs found 170 (9.3%) with SBI and 30 (1.6%) with IBI; 10 had bacterial meningitis. Only one patient with IBI was classified as low risk, a 30-day-old whose blood culture grew Enterobacter cloacae and who had a negative repeat blood culture prior to antibiotic treatment. Together, a negative urinalysis, ANC of 4,090/µL or less, and PCT level of 1.71 ng/mL or less were excellent in predicting infants at low risk for both SBI and IBI, with a sensitivity of 97% and negative LR of 0.05 for the outcome of IBI. When applying these variables with “rounded cutoffs” of PCT levels less than 0.5 ng/mL (chosen by the authors because it is a more commonly used cutoff) and ANC of 4,000/µL or less to identify infants at low risk for SBI, their performance was similar to nonrounded cutoffs. Data for the rule with rounded cutoffs in identifying infants at low risk for IBI were not presented. The PECARN study was limited by the small numbers of infants with IBIs, and the authors recommended caution when applying the rule to infants 28 days or younger.
Older clinical decision rules without PCT, such as the Rochester and modified Philadelphia criteria, use clinical and laboratory features to assess risk of IBI.3 Recent studies have evaluated these criteria in cohorts with larger numbers of infants with IBI since the derivation studies included mostly infants with SBI and small numbers with IBI.3 Gomez et al demonstrated that the Rochester criteria had lower sensitivity and higher negative LR than the Step-by-Step approach in IBI detection.10 In a case-control study of 135 cases of IBI with 249 matched controls, Aronson et al reported that the modified Philadelphia criteria had higher sensitivity but lower specificity than the Rochester criteria for IBI detection.12 The ability of the Rochester and modified Philadelphia criteria to rule out IBI, as demonstrated by the negative LR (range 0.2-0.4), was inferior to the negative LRs documented by Milcent et al8 (PCT cutoff value of 0.3 ng/mL), the Step-by-Step approach,10 and the PECARN rule11 (range 0.05-0.17; Table). However, clinical decision rules with and without PCT suffer similar limitations in having poor specificity in identifying infants likely to have IBI.
GAPS IN THE LITERATURE
Several key knowledge gaps around PCT use for diagnosing neonatal infections exist. First, the optimal use of PCT in context with other biomarkers and clinical decision rules remains uncertain. A meta-analysis of 28 studies involving over 2,600 infants that compared PCT level (with and without CRP) with isolated CRP and presepsin levels found that PCT in combination with CRP had greater diagnostic accuracy than either PCT or CRP alone, which highlights a potential opportunity for prospective study.13 Second, more data are needed on the use of PCT in the ≤ 28-day age group given the increased risk of both IBI and neonatal herpes simplex virus infection (HSV), compared with that in the second month of life. Neonatal HSV poses diagnostic challenges because half of infants will initially present as afebrile,14 and delays in initiating antiviral treatment dramatically increase the risk of permanent disability or death.15 There have been no prospective studies evaluating PCT use as part of neonatal HSV evaluations.
CLINICAL APPLICATIONS AND CONCLUSIONS
In summary, PCT can play an important adjunctive diagnostic role in the evaluation of febrile young infants, especially during the second month of life when outpatient management is more likely to be considered. PCT is superior to other inflammatory markers in identifying IBI, though the optimal cutoffs to maximize sensitivity and specificity are uncertain. Its performance characteristics, both alone and within clinical decision rules, can help clinicians better identify children at low risk for IBI when compared with clinical decision rules without PCT. PCT measurement can help clinicians miss fewer infants with IBI and identify infants for whom safely doing less is an appropriate option, which can ultimately reduce costs and hospitalizations. PCT may be particularly helpful when the clinical history is difficult to assess or when other diagnostic test results are missing or give conflicting results. Centers that use PCT will need to ensure that results are available within a short turnaround time (a few hours) in order to meaningfully affect care. Future studies of PCT in febrile infant evaluations should focus on identifying optimal strategies for incorporating this biomarker into risk assessments that present information to parents in a way that enables them to understand their child’s risk of a serious infection.
1. Tzimenatos L, Mahajan P, Dayan PS, et al. Accuracy of the urinalysis for urinary tract infections in febrile infants 60 days and younger. Pediatrics. 2018;141(2):e20173068. https://doi.org/10.1542/peds.2017-3068
2. Cruz AT, Mahajan P, Bonsu BK, et al. Accuracy of complete blood cell counts to identify febrile infants 60 days or younger with invasive bacterial infections. JAMA Pediatr. 2017;171(11):e172927. https://doi.org/10.1001/jamapediatrics.2017.2927
3. Hui C, Neto G, Tsertsvadze A, et al. Diagnosis and management of febrile infants (0-3 months). Evid Rep Technol Assess (Full Rep). 2012;(205):1-297.
4. Biondi EA, Lee B, Ralston SL, et al. Prevalence of bacteremia and bacterial meningitis in febrile neonates and infants in the second month of life: a systematic review and meta-analysis. JAMA Netw Open. 2019;2(3):e190874. https://doi.org/10.1001/jamanetworkopen.2019.0874
5. Fontela PS, Lacroix J. Procalcitonin: is this the promised biomarker for critically ill patients? J Pediatr Intensive Care. 2016;5(4):162-171. https://doi.org/10.1055/s-0036-1583279
6. Chiesa C, Natale F, Pascone R, et al. C reactive protein and procalcitonin: reference intervals for preterm and term newborns during the early neonatal period. Clin Chim Acta. 2011;412(11-12):1053-1059. https://doi.org/10.1016/j.cca.2011.02.020
7. Gomez B, Bressan S, Mintegi S, et al. Diagnostic value of procalcitonin in well-appearing young febrile infants. Pediatrics. 2012;130(5):815-822. https://doi.org/10.1542/peds.2011-3575
8. Milcent K, Faesch S, Gras-Le Guen C, et al. Use of procalcitonin assays to predict serious bacterial infection in young febrile infants. JAMA Pediatr. 2016;170(1):62-69. https://doi.org/10.1001/jamapediatrics.2015.3210
9. Gomez B, Diaz H, Carro A, Benito J, Mintegi S. Performance of blood biomarkers to rule out invasive bacterial infection in febrile infants under 21 days old. Arch Dis Child. 2019;104(6):547-551. https://doi.org/10.1136/archdischild-2018-315397
10. Gomez B, Mintegi S, Bressan S, et al. Validation of the “step-by-step” approach in the management of young febrile infants. Pediatrics. 2016;138(2):e20154381. https://doi.org/10.1542/peds.2015-4381
11. Kuppermann N, Dayan PS, Levine DA, et al. A clinical prediction rule to identify febrile infants 60 days and younger at low risk for serious bacterial infections. JAMA Pediatr. 2019;173(4):342-351. https://doi.org/10.1001/jamapediatrics.2018.5501
12. Aronson PL, Wang ME, Shapiro ED, et al. Risk stratification of febrile infants ≤60 days old without routine lumbar puncture. Pediatrics. 2018;142(6):e20181879. https://doi.org/10.1542/peds.2018-1879
13. Ruan L, Chen GY, Liu Z, et al. The combination of procalcitonin and C-reactive protein or presepsin alone improves the accuracy of diagnosis of neonatal sepsis: a meta-analysis and systematic review. Crit Care. 2018;22(1):316. https://doi.org/10.1186/s13054-018-2236-1
14. Brower L, Schondelmeyer A, Wilson P, Shah SS. Testing and empiric treatment for neonatal herpes simplex virus: challenges and opportunities for improving the value of care. Hosp Pediatr. 2016;6(2):108-111. https://doi.org/10.1542/hpeds.2015-0166
15. Long SS. Delayed acyclovir therapy in neonates with herpes simplex virus infection is associated with an increased odds of death compared with early therapy. Evid Based Med. 2013;18(2):e20. https://doi.org/10.1136/eb-2012-100674
Febrile infants 60 days of age or younger pose a significant diagnostic challenge for clinicians. Most of these infants are well appearing and do not have localizing signs or symptoms of infection, yet they may have serious bacterial infections (SBI) such as urinary tract infection (UTI), bacteremia, and meningitis. While urinalysis is highly sensitive for predicting UTI,1 older clinical decision rules and biomarkers such as white blood cell (WBC) count, absolute neutrophil count (ANC), and C-reactive protein (CRP) lack both appropriate sensitivity and specificity for identifying bacteremia and meningitis (ie, invasive bacterial infection [IBI]),2,3 which affect approximately 2.4% and 0.9% of febrile infants during the first 2 months of life, respectively.4 The lack of accurate diagnostic markers can drive overuse of laboratory testing, antibiotics, and hospitalization despite the low rates of these infections. As a result, procalcitonin (PCT) has generated interest because of its potential to serve as a more accurate biomarker for bacterial infections. This review summarizes recent literature on the diagnostic utility of PCT in the identification of IBI in febrile young infants 60 days or younger.
MECHANISM OF PROCALCITONIN
Procalcitonin is undetectable in noninflammatory states but can be detected in the blood within 4 to 6 hours after initial bacterial infection.5 Its production is stimulated throughout various tissues of the body by cytokines such as interleukin-6 and tumor necrosis factor, which are produced in response to bacterial infections. Interferon-γ, which is produced in response to viral infections, attenuates PCT production. While these characteristics suggest promise for PCT as a more specific screening test for underlying bacterial infection, there are caveats. PCT levels are physiologically elevated in the first 48 hours of life and vary with gestational age, factors that should be considered when interpreting results.6 Additionally, PCT levels can rise in other inflammatory states such as autoimmune conditions and certain malignancies,5 though these states are unlikely to confound the evaluation of febrile young infants.
DIAGNOSTIC ACCURACY OF PROCALCITONIN
Because of PCT’s potential to be more specific than other commonly used biomarkers, multiple studies have evaluated its performance characteristics in febrile young infants. Gomez et al retrospectively evaluated 1,112 well-appearing infants younger than 3 months with fever without a source in seven European emergency departments (EDs).7 Overall, 23 infants (2.1%) had IBI (1 with meningitis). A PCT level of 0.5 ng/mL or greater was the only independent risk factor for IBI (adjusted odds ratio, 21.69; 95% CI, 7.93-59.28). Four infants with IBI had a PCT level less than 0.5 ng/mL, and none of these four had meningitis. PCT was superior to CRP, ANC, and WBC in detecting IBI (area under the curve [AUC], 0.825; 95% CI, 0.698-0.952). PCT was the also the best marker for identifying IBI among 451 infants with a normal urine dipstick and fever detected ≤6 hours before presentation (AUC, 0.819; 95% CI, 0.551-1.087).
In the largest prospective study to date evaluating the diagnostic accuracy of PCT in febrile young infants, Milcent et al studied 2,047 previously healthy infants aged 7-91 days admitted for fever from 15 French EDs.8 In total, 21 (1%) had an IBI (8 with meningitis). PCT performed better than CRP, ANC, and WBC for the detection of IBI with an AUC of 0.91 (95% CI, 0.83-0.99). In a multivariable model, a PCT level of 0.3 ng/mL or greater was the only independent risk factor for IBI with an adjusted odds ratio of 40.3 (95% CI, 5.0-332). Only one infant with IBI had a PCT level less than 0.3 ng/mL. This infant was 83 days old, had 4 hours of fever, and became afebrile spontaneously prior to the blood culture revealing Streptococcus pneumoniae. PCT also performed better than CRP in the detection of IBI in infants 7-30 days of age and those with fever for less than 6 hours, though both subgroups had small numbers of infants with IBI. The authors determined that a PCT level of 0.3 ng/mL was the optimal cutoff for ruling out IBI; this cutoff had a sensitivity of 90% and negative likelihood ratio (LR) of 0.1 (Table). In contrast, the more commonly studied PCT cutoff of 0.5 ng/mL increased the negative LR to 0.2. The authors suggested that PCT, when used in the context of history, exam, and tests such as urinalysis, could identify infants at low risk of IBI.

Gomez et al conducted a prospective, single-center study of well-appearing infants with fever without a source and negative urine dipsticks.9 They identified IBI in 9 of 196 infants (4.5%) 21 days or younger and 13 of 1,331 infants (1.0%) 22-90 days old. PCT was superior to CRP and ANC for IBI detection in both age groups. However, in infants 21 days or younger, both the positive and negative LRs for PCT levels of 0.5 ng/mL or greater were poor (Table). Differences in results from the prior two studies7,8 may be related to smaller sample size and differences in patient population because this study included infants younger than 7 days and a higher proportion of infants presenting within 6 hours of fever.
CLINICAL DECISION RULES
PCT has also been incorporated into clinical decision rules for febrile young infants, primarily to identify those at low risk of either IBI or SBI. The Step-by-Step approach10 classified well-appearing febrile infants 90 days or younger as having a high risk of IBI if they were ill appearing, younger than 21 days old, had a positive urine dipstick or a PCT level of 0.5 ng/mL or greater, and classified them as intermediate risk if they had a CRP level greater than 20 mg/L or ANC level greater than 10,000/µL. The remaining infants were classified as low risk and could be managed as outpatients without lumbar puncture or empiric antibiotics. Of note, derivation of this rule excluded patients with respiratory signs or symptoms. In a prospective validation study with 2,185 infants from 11 European EDs, 87 (4.0%) had an IBI (10 with bacterial meningitis). Sequentially identifying patients as high risk using general appearance, age, and urine dipstick alone identified 80% of infants with IBI and 90% of those with bacterial meningitis. The remaining case of meningitis would have been detected by an elevated PCT. A total of 7 of 991 infants (0.7%) classified as low risk had an IBI and none had meningitis. Six of these infants had a fever duration of less than 2 hours, which would not be enough time for PCT to rise. The Step-by-Step approach, with a sensitivity of 92% and negative LR of 0.17, performed well in the ability to rule out IBI.
A clinical prediction rule developed by the Pediatric Emergency Care Applied Research Network (PECARN) found that urinalysis, ANC, and PCT performed well in identifying infants 60 days or younger at low risk for SBI and IBI.11 This prospective observational study of 1,821 infants 60 days or younger in 26 US EDs found 170 (9.3%) with SBI and 30 (1.6%) with IBI; 10 had bacterial meningitis. Only one patient with IBI was classified as low risk, a 30-day-old whose blood culture grew Enterobacter cloacae and who had a negative repeat blood culture prior to antibiotic treatment. Together, a negative urinalysis, ANC of 4,090/µL or less, and PCT level of 1.71 ng/mL or less were excellent in predicting infants at low risk for both SBI and IBI, with a sensitivity of 97% and negative LR of 0.05 for the outcome of IBI. When applying these variables with “rounded cutoffs” of PCT levels less than 0.5 ng/mL (chosen by the authors because it is a more commonly used cutoff) and ANC of 4,000/µL or less to identify infants at low risk for SBI, their performance was similar to nonrounded cutoffs. Data for the rule with rounded cutoffs in identifying infants at low risk for IBI were not presented. The PECARN study was limited by the small numbers of infants with IBIs, and the authors recommended caution when applying the rule to infants 28 days or younger.
Older clinical decision rules without PCT, such as the Rochester and modified Philadelphia criteria, use clinical and laboratory features to assess risk of IBI.3 Recent studies have evaluated these criteria in cohorts with larger numbers of infants with IBI since the derivation studies included mostly infants with SBI and small numbers with IBI.3 Gomez et al demonstrated that the Rochester criteria had lower sensitivity and higher negative LR than the Step-by-Step approach in IBI detection.10 In a case-control study of 135 cases of IBI with 249 matched controls, Aronson et al reported that the modified Philadelphia criteria had higher sensitivity but lower specificity than the Rochester criteria for IBI detection.12 The ability of the Rochester and modified Philadelphia criteria to rule out IBI, as demonstrated by the negative LR (range 0.2-0.4), was inferior to the negative LRs documented by Milcent et al8 (PCT cutoff value of 0.3 ng/mL), the Step-by-Step approach,10 and the PECARN rule11 (range 0.05-0.17; Table). However, clinical decision rules with and without PCT suffer similar limitations in having poor specificity in identifying infants likely to have IBI.
GAPS IN THE LITERATURE
Several key knowledge gaps around PCT use for diagnosing neonatal infections exist. First, the optimal use of PCT in context with other biomarkers and clinical decision rules remains uncertain. A meta-analysis of 28 studies involving over 2,600 infants that compared PCT level (with and without CRP) with isolated CRP and presepsin levels found that PCT in combination with CRP had greater diagnostic accuracy than either PCT or CRP alone, which highlights a potential opportunity for prospective study.13 Second, more data are needed on the use of PCT in the ≤ 28-day age group given the increased risk of both IBI and neonatal herpes simplex virus infection (HSV), compared with that in the second month of life. Neonatal HSV poses diagnostic challenges because half of infants will initially present as afebrile,14 and delays in initiating antiviral treatment dramatically increase the risk of permanent disability or death.15 There have been no prospective studies evaluating PCT use as part of neonatal HSV evaluations.
CLINICAL APPLICATIONS AND CONCLUSIONS
In summary, PCT can play an important adjunctive diagnostic role in the evaluation of febrile young infants, especially during the second month of life when outpatient management is more likely to be considered. PCT is superior to other inflammatory markers in identifying IBI, though the optimal cutoffs to maximize sensitivity and specificity are uncertain. Its performance characteristics, both alone and within clinical decision rules, can help clinicians better identify children at low risk for IBI when compared with clinical decision rules without PCT. PCT measurement can help clinicians miss fewer infants with IBI and identify infants for whom safely doing less is an appropriate option, which can ultimately reduce costs and hospitalizations. PCT may be particularly helpful when the clinical history is difficult to assess or when other diagnostic test results are missing or give conflicting results. Centers that use PCT will need to ensure that results are available within a short turnaround time (a few hours) in order to meaningfully affect care. Future studies of PCT in febrile infant evaluations should focus on identifying optimal strategies for incorporating this biomarker into risk assessments that present information to parents in a way that enables them to understand their child’s risk of a serious infection.
Febrile infants 60 days of age or younger pose a significant diagnostic challenge for clinicians. Most of these infants are well appearing and do not have localizing signs or symptoms of infection, yet they may have serious bacterial infections (SBI) such as urinary tract infection (UTI), bacteremia, and meningitis. While urinalysis is highly sensitive for predicting UTI,1 older clinical decision rules and biomarkers such as white blood cell (WBC) count, absolute neutrophil count (ANC), and C-reactive protein (CRP) lack both appropriate sensitivity and specificity for identifying bacteremia and meningitis (ie, invasive bacterial infection [IBI]),2,3 which affect approximately 2.4% and 0.9% of febrile infants during the first 2 months of life, respectively.4 The lack of accurate diagnostic markers can drive overuse of laboratory testing, antibiotics, and hospitalization despite the low rates of these infections. As a result, procalcitonin (PCT) has generated interest because of its potential to serve as a more accurate biomarker for bacterial infections. This review summarizes recent literature on the diagnostic utility of PCT in the identification of IBI in febrile young infants 60 days or younger.
MECHANISM OF PROCALCITONIN
Procalcitonin is undetectable in noninflammatory states but can be detected in the blood within 4 to 6 hours after initial bacterial infection.5 Its production is stimulated throughout various tissues of the body by cytokines such as interleukin-6 and tumor necrosis factor, which are produced in response to bacterial infections. Interferon-γ, which is produced in response to viral infections, attenuates PCT production. While these characteristics suggest promise for PCT as a more specific screening test for underlying bacterial infection, there are caveats. PCT levels are physiologically elevated in the first 48 hours of life and vary with gestational age, factors that should be considered when interpreting results.6 Additionally, PCT levels can rise in other inflammatory states such as autoimmune conditions and certain malignancies,5 though these states are unlikely to confound the evaluation of febrile young infants.
DIAGNOSTIC ACCURACY OF PROCALCITONIN
Because of PCT’s potential to be more specific than other commonly used biomarkers, multiple studies have evaluated its performance characteristics in febrile young infants. Gomez et al retrospectively evaluated 1,112 well-appearing infants younger than 3 months with fever without a source in seven European emergency departments (EDs).7 Overall, 23 infants (2.1%) had IBI (1 with meningitis). A PCT level of 0.5 ng/mL or greater was the only independent risk factor for IBI (adjusted odds ratio, 21.69; 95% CI, 7.93-59.28). Four infants with IBI had a PCT level less than 0.5 ng/mL, and none of these four had meningitis. PCT was superior to CRP, ANC, and WBC in detecting IBI (area under the curve [AUC], 0.825; 95% CI, 0.698-0.952). PCT was the also the best marker for identifying IBI among 451 infants with a normal urine dipstick and fever detected ≤6 hours before presentation (AUC, 0.819; 95% CI, 0.551-1.087).
In the largest prospective study to date evaluating the diagnostic accuracy of PCT in febrile young infants, Milcent et al studied 2,047 previously healthy infants aged 7-91 days admitted for fever from 15 French EDs.8 In total, 21 (1%) had an IBI (8 with meningitis). PCT performed better than CRP, ANC, and WBC for the detection of IBI with an AUC of 0.91 (95% CI, 0.83-0.99). In a multivariable model, a PCT level of 0.3 ng/mL or greater was the only independent risk factor for IBI with an adjusted odds ratio of 40.3 (95% CI, 5.0-332). Only one infant with IBI had a PCT level less than 0.3 ng/mL. This infant was 83 days old, had 4 hours of fever, and became afebrile spontaneously prior to the blood culture revealing Streptococcus pneumoniae. PCT also performed better than CRP in the detection of IBI in infants 7-30 days of age and those with fever for less than 6 hours, though both subgroups had small numbers of infants with IBI. The authors determined that a PCT level of 0.3 ng/mL was the optimal cutoff for ruling out IBI; this cutoff had a sensitivity of 90% and negative likelihood ratio (LR) of 0.1 (Table). In contrast, the more commonly studied PCT cutoff of 0.5 ng/mL increased the negative LR to 0.2. The authors suggested that PCT, when used in the context of history, exam, and tests such as urinalysis, could identify infants at low risk of IBI.

Gomez et al conducted a prospective, single-center study of well-appearing infants with fever without a source and negative urine dipsticks.9 They identified IBI in 9 of 196 infants (4.5%) 21 days or younger and 13 of 1,331 infants (1.0%) 22-90 days old. PCT was superior to CRP and ANC for IBI detection in both age groups. However, in infants 21 days or younger, both the positive and negative LRs for PCT levels of 0.5 ng/mL or greater were poor (Table). Differences in results from the prior two studies7,8 may be related to smaller sample size and differences in patient population because this study included infants younger than 7 days and a higher proportion of infants presenting within 6 hours of fever.
CLINICAL DECISION RULES
PCT has also been incorporated into clinical decision rules for febrile young infants, primarily to identify those at low risk of either IBI or SBI. The Step-by-Step approach10 classified well-appearing febrile infants 90 days or younger as having a high risk of IBI if they were ill appearing, younger than 21 days old, had a positive urine dipstick or a PCT level of 0.5 ng/mL or greater, and classified them as intermediate risk if they had a CRP level greater than 20 mg/L or ANC level greater than 10,000/µL. The remaining infants were classified as low risk and could be managed as outpatients without lumbar puncture or empiric antibiotics. Of note, derivation of this rule excluded patients with respiratory signs or symptoms. In a prospective validation study with 2,185 infants from 11 European EDs, 87 (4.0%) had an IBI (10 with bacterial meningitis). Sequentially identifying patients as high risk using general appearance, age, and urine dipstick alone identified 80% of infants with IBI and 90% of those with bacterial meningitis. The remaining case of meningitis would have been detected by an elevated PCT. A total of 7 of 991 infants (0.7%) classified as low risk had an IBI and none had meningitis. Six of these infants had a fever duration of less than 2 hours, which would not be enough time for PCT to rise. The Step-by-Step approach, with a sensitivity of 92% and negative LR of 0.17, performed well in the ability to rule out IBI.
A clinical prediction rule developed by the Pediatric Emergency Care Applied Research Network (PECARN) found that urinalysis, ANC, and PCT performed well in identifying infants 60 days or younger at low risk for SBI and IBI.11 This prospective observational study of 1,821 infants 60 days or younger in 26 US EDs found 170 (9.3%) with SBI and 30 (1.6%) with IBI; 10 had bacterial meningitis. Only one patient with IBI was classified as low risk, a 30-day-old whose blood culture grew Enterobacter cloacae and who had a negative repeat blood culture prior to antibiotic treatment. Together, a negative urinalysis, ANC of 4,090/µL or less, and PCT level of 1.71 ng/mL or less were excellent in predicting infants at low risk for both SBI and IBI, with a sensitivity of 97% and negative LR of 0.05 for the outcome of IBI. When applying these variables with “rounded cutoffs” of PCT levels less than 0.5 ng/mL (chosen by the authors because it is a more commonly used cutoff) and ANC of 4,000/µL or less to identify infants at low risk for SBI, their performance was similar to nonrounded cutoffs. Data for the rule with rounded cutoffs in identifying infants at low risk for IBI were not presented. The PECARN study was limited by the small numbers of infants with IBIs, and the authors recommended caution when applying the rule to infants 28 days or younger.
Older clinical decision rules without PCT, such as the Rochester and modified Philadelphia criteria, use clinical and laboratory features to assess risk of IBI.3 Recent studies have evaluated these criteria in cohorts with larger numbers of infants with IBI since the derivation studies included mostly infants with SBI and small numbers with IBI.3 Gomez et al demonstrated that the Rochester criteria had lower sensitivity and higher negative LR than the Step-by-Step approach in IBI detection.10 In a case-control study of 135 cases of IBI with 249 matched controls, Aronson et al reported that the modified Philadelphia criteria had higher sensitivity but lower specificity than the Rochester criteria for IBI detection.12 The ability of the Rochester and modified Philadelphia criteria to rule out IBI, as demonstrated by the negative LR (range 0.2-0.4), was inferior to the negative LRs documented by Milcent et al8 (PCT cutoff value of 0.3 ng/mL), the Step-by-Step approach,10 and the PECARN rule11 (range 0.05-0.17; Table). However, clinical decision rules with and without PCT suffer similar limitations in having poor specificity in identifying infants likely to have IBI.
GAPS IN THE LITERATURE
Several key knowledge gaps around PCT use for diagnosing neonatal infections exist. First, the optimal use of PCT in context with other biomarkers and clinical decision rules remains uncertain. A meta-analysis of 28 studies involving over 2,600 infants that compared PCT level (with and without CRP) with isolated CRP and presepsin levels found that PCT in combination with CRP had greater diagnostic accuracy than either PCT or CRP alone, which highlights a potential opportunity for prospective study.13 Second, more data are needed on the use of PCT in the ≤ 28-day age group given the increased risk of both IBI and neonatal herpes simplex virus infection (HSV), compared with that in the second month of life. Neonatal HSV poses diagnostic challenges because half of infants will initially present as afebrile,14 and delays in initiating antiviral treatment dramatically increase the risk of permanent disability or death.15 There have been no prospective studies evaluating PCT use as part of neonatal HSV evaluations.
CLINICAL APPLICATIONS AND CONCLUSIONS
In summary, PCT can play an important adjunctive diagnostic role in the evaluation of febrile young infants, especially during the second month of life when outpatient management is more likely to be considered. PCT is superior to other inflammatory markers in identifying IBI, though the optimal cutoffs to maximize sensitivity and specificity are uncertain. Its performance characteristics, both alone and within clinical decision rules, can help clinicians better identify children at low risk for IBI when compared with clinical decision rules without PCT. PCT measurement can help clinicians miss fewer infants with IBI and identify infants for whom safely doing less is an appropriate option, which can ultimately reduce costs and hospitalizations. PCT may be particularly helpful when the clinical history is difficult to assess or when other diagnostic test results are missing or give conflicting results. Centers that use PCT will need to ensure that results are available within a short turnaround time (a few hours) in order to meaningfully affect care. Future studies of PCT in febrile infant evaluations should focus on identifying optimal strategies for incorporating this biomarker into risk assessments that present information to parents in a way that enables them to understand their child’s risk of a serious infection.
1. Tzimenatos L, Mahajan P, Dayan PS, et al. Accuracy of the urinalysis for urinary tract infections in febrile infants 60 days and younger. Pediatrics. 2018;141(2):e20173068. https://doi.org/10.1542/peds.2017-3068
2. Cruz AT, Mahajan P, Bonsu BK, et al. Accuracy of complete blood cell counts to identify febrile infants 60 days or younger with invasive bacterial infections. JAMA Pediatr. 2017;171(11):e172927. https://doi.org/10.1001/jamapediatrics.2017.2927
3. Hui C, Neto G, Tsertsvadze A, et al. Diagnosis and management of febrile infants (0-3 months). Evid Rep Technol Assess (Full Rep). 2012;(205):1-297.
4. Biondi EA, Lee B, Ralston SL, et al. Prevalence of bacteremia and bacterial meningitis in febrile neonates and infants in the second month of life: a systematic review and meta-analysis. JAMA Netw Open. 2019;2(3):e190874. https://doi.org/10.1001/jamanetworkopen.2019.0874
5. Fontela PS, Lacroix J. Procalcitonin: is this the promised biomarker for critically ill patients? J Pediatr Intensive Care. 2016;5(4):162-171. https://doi.org/10.1055/s-0036-1583279
6. Chiesa C, Natale F, Pascone R, et al. C reactive protein and procalcitonin: reference intervals for preterm and term newborns during the early neonatal period. Clin Chim Acta. 2011;412(11-12):1053-1059. https://doi.org/10.1016/j.cca.2011.02.020
7. Gomez B, Bressan S, Mintegi S, et al. Diagnostic value of procalcitonin in well-appearing young febrile infants. Pediatrics. 2012;130(5):815-822. https://doi.org/10.1542/peds.2011-3575
8. Milcent K, Faesch S, Gras-Le Guen C, et al. Use of procalcitonin assays to predict serious bacterial infection in young febrile infants. JAMA Pediatr. 2016;170(1):62-69. https://doi.org/10.1001/jamapediatrics.2015.3210
9. Gomez B, Diaz H, Carro A, Benito J, Mintegi S. Performance of blood biomarkers to rule out invasive bacterial infection in febrile infants under 21 days old. Arch Dis Child. 2019;104(6):547-551. https://doi.org/10.1136/archdischild-2018-315397
10. Gomez B, Mintegi S, Bressan S, et al. Validation of the “step-by-step” approach in the management of young febrile infants. Pediatrics. 2016;138(2):e20154381. https://doi.org/10.1542/peds.2015-4381
11. Kuppermann N, Dayan PS, Levine DA, et al. A clinical prediction rule to identify febrile infants 60 days and younger at low risk for serious bacterial infections. JAMA Pediatr. 2019;173(4):342-351. https://doi.org/10.1001/jamapediatrics.2018.5501
12. Aronson PL, Wang ME, Shapiro ED, et al. Risk stratification of febrile infants ≤60 days old without routine lumbar puncture. Pediatrics. 2018;142(6):e20181879. https://doi.org/10.1542/peds.2018-1879
13. Ruan L, Chen GY, Liu Z, et al. The combination of procalcitonin and C-reactive protein or presepsin alone improves the accuracy of diagnosis of neonatal sepsis: a meta-analysis and systematic review. Crit Care. 2018;22(1):316. https://doi.org/10.1186/s13054-018-2236-1
14. Brower L, Schondelmeyer A, Wilson P, Shah SS. Testing and empiric treatment for neonatal herpes simplex virus: challenges and opportunities for improving the value of care. Hosp Pediatr. 2016;6(2):108-111. https://doi.org/10.1542/hpeds.2015-0166
15. Long SS. Delayed acyclovir therapy in neonates with herpes simplex virus infection is associated with an increased odds of death compared with early therapy. Evid Based Med. 2013;18(2):e20. https://doi.org/10.1136/eb-2012-100674
1. Tzimenatos L, Mahajan P, Dayan PS, et al. Accuracy of the urinalysis for urinary tract infections in febrile infants 60 days and younger. Pediatrics. 2018;141(2):e20173068. https://doi.org/10.1542/peds.2017-3068
2. Cruz AT, Mahajan P, Bonsu BK, et al. Accuracy of complete blood cell counts to identify febrile infants 60 days or younger with invasive bacterial infections. JAMA Pediatr. 2017;171(11):e172927. https://doi.org/10.1001/jamapediatrics.2017.2927
3. Hui C, Neto G, Tsertsvadze A, et al. Diagnosis and management of febrile infants (0-3 months). Evid Rep Technol Assess (Full Rep). 2012;(205):1-297.
4. Biondi EA, Lee B, Ralston SL, et al. Prevalence of bacteremia and bacterial meningitis in febrile neonates and infants in the second month of life: a systematic review and meta-analysis. JAMA Netw Open. 2019;2(3):e190874. https://doi.org/10.1001/jamanetworkopen.2019.0874
5. Fontela PS, Lacroix J. Procalcitonin: is this the promised biomarker for critically ill patients? J Pediatr Intensive Care. 2016;5(4):162-171. https://doi.org/10.1055/s-0036-1583279
6. Chiesa C, Natale F, Pascone R, et al. C reactive protein and procalcitonin: reference intervals for preterm and term newborns during the early neonatal period. Clin Chim Acta. 2011;412(11-12):1053-1059. https://doi.org/10.1016/j.cca.2011.02.020
7. Gomez B, Bressan S, Mintegi S, et al. Diagnostic value of procalcitonin in well-appearing young febrile infants. Pediatrics. 2012;130(5):815-822. https://doi.org/10.1542/peds.2011-3575
8. Milcent K, Faesch S, Gras-Le Guen C, et al. Use of procalcitonin assays to predict serious bacterial infection in young febrile infants. JAMA Pediatr. 2016;170(1):62-69. https://doi.org/10.1001/jamapediatrics.2015.3210
9. Gomez B, Diaz H, Carro A, Benito J, Mintegi S. Performance of blood biomarkers to rule out invasive bacterial infection in febrile infants under 21 days old. Arch Dis Child. 2019;104(6):547-551. https://doi.org/10.1136/archdischild-2018-315397
10. Gomez B, Mintegi S, Bressan S, et al. Validation of the “step-by-step” approach in the management of young febrile infants. Pediatrics. 2016;138(2):e20154381. https://doi.org/10.1542/peds.2015-4381
11. Kuppermann N, Dayan PS, Levine DA, et al. A clinical prediction rule to identify febrile infants 60 days and younger at low risk for serious bacterial infections. JAMA Pediatr. 2019;173(4):342-351. https://doi.org/10.1001/jamapediatrics.2018.5501
12. Aronson PL, Wang ME, Shapiro ED, et al. Risk stratification of febrile infants ≤60 days old without routine lumbar puncture. Pediatrics. 2018;142(6):e20181879. https://doi.org/10.1542/peds.2018-1879
13. Ruan L, Chen GY, Liu Z, et al. The combination of procalcitonin and C-reactive protein or presepsin alone improves the accuracy of diagnosis of neonatal sepsis: a meta-analysis and systematic review. Crit Care. 2018;22(1):316. https://doi.org/10.1186/s13054-018-2236-1
14. Brower L, Schondelmeyer A, Wilson P, Shah SS. Testing and empiric treatment for neonatal herpes simplex virus: challenges and opportunities for improving the value of care. Hosp Pediatr. 2016;6(2):108-111. https://doi.org/10.1542/hpeds.2015-0166
15. Long SS. Delayed acyclovir therapy in neonates with herpes simplex virus infection is associated with an increased odds of death compared with early therapy. Evid Based Med. 2013;18(2):e20. https://doi.org/10.1136/eb-2012-100674
© 2020 Society of Hospital Medicine
Clinical Progress Note: Goal-Directed N-acetylcysteine Treatment of Acetaminophen Toxicity
According to the most recent Annual Report of the American Association of Poison Control Centers’ National Poison Data System, there were 51,081 patient exposures to acetaminophen or acetaminophen-containing products treated in a healthcare facility in 2018.1 The toxicity of acetaminophen is mediated by its metabolism into the electrophile N-acetyl-p-benzoquinone imine (NAPQI).2 If the amount of acetaminophen ingested is higher than recommended dosages, glutathione stores may become depleted as the liver tries to detoxify NAPQI and may no longer able to keep up with the demand. As the patient progresses 24 to 36 hours after ingestion, the onset of hepatic injury becomes apparent with elevations in aspartate aminotransferase (AST). AST elevations, should they occur, are almost always present by 36 hours.2 Maximum hepatic dysfunction, as well as elevations in prothrombin time, bilirubin, and serum creatinine, occurs 72 to 96 hours after a toxic ingestion. As a result of marked age-associated differences in the process of conjugating acetaminophen metabolites, infants and young children may be less susceptible to acetaminophen-associated hepatotoxicity, compared with their adult counterparts.3,4
This clinical progress note addresses acetaminophen toxicity treatment, focusing on N-acetylcysteine (NAC) dosing protocols and the 2017 American College of Medical Toxicology (ACMT) position statement, Duration of Intravenous Acetylcysteine Therapy Following Acetaminophen Overdose.5 We conducted a literature search via the PubMed database. The authors began by using the following Medical Subject Headings terms “acetaminophen overdose [title],” which yielded 299 articles, “acetaminophen hepatotoxicity [title],” which yielded 283 articles, and “acetaminophen N-acetylcysteine [title],” which yielded 335 titles. Variations of these terms were used to ensure exhaustive search results. The search results were reviewed for applicability to acetaminophen poisoning and especially goal-directed N-acetylcysteine treatment strategies.
INITIAL TREATMENT
Clarifying a patient’s history of ingestion is a crucial first step to the diagnosis and risk evaluation of acetaminophen-poisoned patients. The initial laboratory workup should include, at minimum, measurement (at 4 or more hours after ingestion) of serum acetaminophen level, AST and alanine aminotransferase (ALT), bilirubin, alkaline phosphatase, creatinine, prothrombin time and international normalized ratio (PT/INR), lactate, and ethanol levels. Gastric decontamination is not appropriate in most cases because of the rapid absorption of acetaminophen.2 Activated charcoal is most effective when given within the first 2 hours following ingestion. For acetaminophen-poisoned patients with severe hepatic toxicity, treating clinicians should consider consultation with a medical toxicologist, an individual with expertise, or the local Poison Control Center.5
The Rumack-Matthew nomogram provides guidance on the initiation of antidotal treatment for patients with acetaminophen overdose. This nomogram was developed to exhibit high sensitivity in patients at risk for hepatic damage from acetaminophen overdose.6 The mainstay for treatment of acetaminophen poisonings is NAC. Early administration of NAC can replenish depleted hepatic glutathione stores, which restores the liver’s ability to detoxify the toxic metabolite NAPQI. When administered within 6 to 8 hours following ingestion, NAC is nearly universally effective at preventing the subsequent hepatic damage from NAPQI.7 Consideration of individualized factors should lead the treating clinician to use the Rumack-Matthew nomogram within the appropriate contexts. Factors such as an unreliable timeline of ingestion by patients, concomitantly ingested medications, and patient-specific factors that might alter the absorption of acetaminophen or impact serum acetaminophen concentrations should be considered. While this nomogram is helpful for decision-making pertaining to the initiation of NAC, further management and continuation of treatment must be patient specific and determined by the treating clinician.
In the United States, intravenous and oral formulations of NAC are available for treatment of acetaminophen toxicity. These formulations are equally efficacious in treating acetaminophen toxicity, with the exception of established hepatic failure, for which only the intravenous route has been studied.8 Oral administration results in vomiting in approximately 20% of patients, while intravenous administration may uncommonly result in severe anaphylactoid reactions. Three scenarios exist in which the intravenous route is preferred over the oral route: acetaminophen toxicity in pregnant women, acetaminophen-induced hepatic failure, and intractable vomiting preventing treatment with oral therapy.2 The intravenous formulation is Food and Drug Administration–approved for a 21-hour treatment duration, while the oral formulation is traditionally administered for a 72-hour duration.9,10
LIMITATIONS OF CURRENT TREATMENT ALGORITHMS
Treatment of acetaminophen poisoning must be tailored on an individual basis and should not follow predetermined durations of treatment. Not only might the FDA-approved 21-hour course of intravenous NAC be inadequate, but the standard 72-hour oral regimen duration may be excessive for many patients. Both the 21-hour and 72-hour protocols were developed based on the half-life and expected elimination of acetaminophen from the body. The traditional 72-hour oral protocol used in the United States was approved in 1985 and was based on the observation that, in patients who developed acetaminophen-induced fatal hepatotoxicity, the acetaminophen half-life was 12 hours, requiring five half-lives to eliminate the drug, for a total of 60 hours.11 The FDA then added an additional 12 hours to provide an additional margin of safety. Researchers in the United Kingdom developed an intravenous treatment protocol based on a shorter 4-hour acetaminophen half-life, calculated from observations of a larger pool of acetaminophen-poisoned patients and not just those with fatal hepatotoxicity.12 This 21-hour protocol—slightly longer than the five 4-hour half-lives needed—was subsequently approved in the United States as the intravenous formulation’s duration in 2004.
Case reports of hepatic failure have described treatment failures of intravenous NAC after the completion of the standard 21 hour treatment regimen.13 In a case report by Smith et al, a patient with significant single ingestion is described who ultimately developed hepatotoxicity despite receiving the FDA-approved 21-hour infusion.13 This patient ingested approximately 96 immediate-release, 500-mg acetaminophen tablets over approximately 1 hour. The initial acetaminophen concentration was 264 mcg/mL at 2.25 hours after ingestion. At 21 hours after ingestion, the patient’s acetaminophen levels remained elevated at 116 mcg/mL, but therapy with NAC was discontinued per protocol because AST and ALT levels were normal. A second peak in serum acetaminophen levels occurred after discontinuation of NAC, with levels reaching 228 mcg/mL approximately 48 hours after ingestion. Liver function worsened over the next few days with elevations in AST of greater than 4,000 U/L, PT of 51.4 seconds, and ammonia of 165 mcg/dL. Intravenous NAC was restarted and hepatic function improved gradually. The patient was ultimately discharged from the hospital. This case and others highlight the possible pitfalls of applying a protocolized, fixed treatment duration of NAC to all patients.
GOAL-DIRECTED TREATMENT RECOMMENDATIONS
Based on individual ingestion timing, quantities, and pharmacokinetics, patients should have pertinent laboratory markers monitored at least every 24 hours from presentation. The clinician may then consider the need to continue or discontinue NAC treatment every 24 hours from the initiation of NAC. Although notable limitations exist for both intravenous and oral treatment regimens, the literature uniformly recommends treating until serum acetaminophen levels are undetectable, regardless of NAC administration route. Based on the metabolism of acetaminophen, as long as serum concentrations of acetaminophen persist, further metabolism to the toxic NAPQI metabolite is possible. The ACMT addressed these concerns with the 2017 position paper, which strongly recommends all of the following criteria to be present prior to discontinuation of intravenous NAC: undetectable acetaminophen concentration, improving hepatic aminotransferases, and improving prognostic markers, including creatinine, lactate, pH, PT/INR, and phosphate.5 In cases in which laboratory and clinical parameters are abnormal, continued treatment beyond the established standard NAC treatment durations is warranted. However, once NAC treatment is extended beyond established protocol lengths, the subsequent dosing regimens that should be employed in these patients are not well studied. The ACMT Position Statement states the administration of an additional NAC bolus or extending the duration of the 6.25 mg/kg per hour maintenance infusion may be appropriate in this subset of patients requiring continued treatment.
Based on the positive outcomes in most patients receiving the 21-hour intravenous NAC protocol, the necessity of an additional 2 days of NAC treatment with the 72-hour oral regimen is uncertain. The ACMT Position Statement states “evidence supports using shorter oral NAC courses, provided that liver enzymes and synthetic function are normal or improving, and plasma acetaminophen concentration is undetectable.”5 Based on this and available literature, patients with a lower risk for developing hepatotoxicity may have oral NAC therapy discontinued after 24 to 48 hours of treatment if specific lab and patient parameters are within defined criteria.
The ACMT does not differentiate between adult and pediatric recommendations. However, the data used to develop the ACMT goal-directed treatment recommendations included both adult and pediatric patients.14,15 The same goal-directed treatment principles may be applied to pediatric patients, though studies have not specifically addressed pediatric goal-directed treatment of acetaminophen poisoning.
CONCLUSION
Acetaminophen-poisoned patients require evaluation not just prior to initiation of treatment, but also when deciding to discontinue NAC therapy. New literature supports reconsideration of standardized 21- and 72-hour NAC treatment protocols, as well as the notion that treatment duration should be guided by laboratory parameters rather than by route of NAC administration. Goal-directed or patient-tailored treatment is also supported by the ACMT.5 Treatment with NAC should continue in patients displaying hepatic toxicity or persistent elevations in acetaminophen levels. In these cases, the optimal treatment regimen in these cases remains unknown and further research into this area is merited.
1. Gummin D, Mowry J, Spyker D, et al. 2018 Annual Report of the American Association of Poison Control Centers’ National Poison Data System (NPDS): 36th Annual Report. Clin Toxicol (Phila). 2019;57(12):1220‐1413. https://doi.org/10.1080/15563650.2019.1677022
2. Hoffman RS, Howland MA, Lewin NA, Nelson LS, Goldfrank LR, eds. Goldfrank’s Toxicologic Emergencies, Tenth Edition. McGraw Hill Education; 2015.
3. Miller RP, Roberts RJ, Fischer LJ. Acetaminophen elimination kinetics in neonates, children, and adults. Clin Pharmacol Ther. 1976;19:284-294. https://doi.org/10.1002/cpt1976193284
4. Rumack BH. Acetaminophen overdose in young children: treatment and effects of alcohol and other additional ingestants in 417 cases. Am J Dis Child. 1984;138(5):428-433. https://doi.org/10.1001/archpedi.1984.02140430006003
5. American College of Medical Toxicology. ACMT position statement: duration of intravenous acetylcysteine therapy following acetaminophen overdose. J Med Toxicol. 2017;13(1):126-127. https://doi.org/10.1007/s13181-016-0542-z
6. Matthew H. Acute acetaminophen poisoning. Clin Toxicol. 1973;6(1):9-11. https://doi.org/10.3109/15563657308991037
7. Smilkstein MJ, Knapp GL, Kulig KW, Rumack BH. Efficacy of oral N-acetylcysteine in the treatment of acetaminophen overdose: analysis of the national multicenter study (1976 to 1985). N Engl J Med. 1988;319(24):1557-1562. https://doi.org/10.1056/nejm198812153192401
8. Keays R, Harrison PM, Wendon JA, et al. Intravenous acetylcysteine in paracetamol induced fulminant hepatic failure: a prospective controlled trial. BMJ. 1991;303(6809):1026-1029. https://doi.org/10.1136/bmj.303.6809.1026
9. Acetadote ® (acetylcysteine injection). Package Insert. Cumberland Pharmaceuticals; 2004.
10. Acetylcysteine Solution. Package insert. Roxanne Laboratories; 2007.
11. Wong A, Graudins A. Risk prediction of hepatotoxicity in paracetamol poisoning. Clin Toxicol (Phila). 2017;55(8):879-892. https://doi.org/10.1080/15563650.2017.1317349
12. Prescott LF. Kinetics and metabolism of paracetamol and phenacetin. Br J Clin Pharmacol. 1980;10(Suppl 2):291S-298S. https://doi.org/10.1111/j.1365-2125.1980.tb01812.x
13. Smith SW, Howland MA, Hoffman RS, Nelson LS. Acetaminophen overdose with altered acetaminophen pharmacokinetics and hepatotoxicity associated with premature cessation of intravenous N-acetylcysteine therapy. Ann Pharmacother. 2008;42(9):1333-1339. https://doi.org/10.1345/aph.1k680
14. Woo OF, Mueller PD, Olsen KR, Anderson B, Kim SY. Shorter duration of oral N-Acetylcysteine therapy for acute acetaminophen overdose. Ann Emerg Med. 2000; 35(4):363-368. https://doi.org/10.1016/S0196-0644(00)70055-2
15. Betten D, Cantrell F, Thomas S, Williams S, Clark R. A prospective evaluation of shortened course oral N-acetylcysteine for the treatment of acute acetaminophen poisoning. Ann Emerg Med. 2007;50(3):272-279. https://doi.org/10.1016/j.annemergmed.2006.11.010
According to the most recent Annual Report of the American Association of Poison Control Centers’ National Poison Data System, there were 51,081 patient exposures to acetaminophen or acetaminophen-containing products treated in a healthcare facility in 2018.1 The toxicity of acetaminophen is mediated by its metabolism into the electrophile N-acetyl-p-benzoquinone imine (NAPQI).2 If the amount of acetaminophen ingested is higher than recommended dosages, glutathione stores may become depleted as the liver tries to detoxify NAPQI and may no longer able to keep up with the demand. As the patient progresses 24 to 36 hours after ingestion, the onset of hepatic injury becomes apparent with elevations in aspartate aminotransferase (AST). AST elevations, should they occur, are almost always present by 36 hours.2 Maximum hepatic dysfunction, as well as elevations in prothrombin time, bilirubin, and serum creatinine, occurs 72 to 96 hours after a toxic ingestion. As a result of marked age-associated differences in the process of conjugating acetaminophen metabolites, infants and young children may be less susceptible to acetaminophen-associated hepatotoxicity, compared with their adult counterparts.3,4
This clinical progress note addresses acetaminophen toxicity treatment, focusing on N-acetylcysteine (NAC) dosing protocols and the 2017 American College of Medical Toxicology (ACMT) position statement, Duration of Intravenous Acetylcysteine Therapy Following Acetaminophen Overdose.5 We conducted a literature search via the PubMed database. The authors began by using the following Medical Subject Headings terms “acetaminophen overdose [title],” which yielded 299 articles, “acetaminophen hepatotoxicity [title],” which yielded 283 articles, and “acetaminophen N-acetylcysteine [title],” which yielded 335 titles. Variations of these terms were used to ensure exhaustive search results. The search results were reviewed for applicability to acetaminophen poisoning and especially goal-directed N-acetylcysteine treatment strategies.
INITIAL TREATMENT
Clarifying a patient’s history of ingestion is a crucial first step to the diagnosis and risk evaluation of acetaminophen-poisoned patients. The initial laboratory workup should include, at minimum, measurement (at 4 or more hours after ingestion) of serum acetaminophen level, AST and alanine aminotransferase (ALT), bilirubin, alkaline phosphatase, creatinine, prothrombin time and international normalized ratio (PT/INR), lactate, and ethanol levels. Gastric decontamination is not appropriate in most cases because of the rapid absorption of acetaminophen.2 Activated charcoal is most effective when given within the first 2 hours following ingestion. For acetaminophen-poisoned patients with severe hepatic toxicity, treating clinicians should consider consultation with a medical toxicologist, an individual with expertise, or the local Poison Control Center.5
The Rumack-Matthew nomogram provides guidance on the initiation of antidotal treatment for patients with acetaminophen overdose. This nomogram was developed to exhibit high sensitivity in patients at risk for hepatic damage from acetaminophen overdose.6 The mainstay for treatment of acetaminophen poisonings is NAC. Early administration of NAC can replenish depleted hepatic glutathione stores, which restores the liver’s ability to detoxify the toxic metabolite NAPQI. When administered within 6 to 8 hours following ingestion, NAC is nearly universally effective at preventing the subsequent hepatic damage from NAPQI.7 Consideration of individualized factors should lead the treating clinician to use the Rumack-Matthew nomogram within the appropriate contexts. Factors such as an unreliable timeline of ingestion by patients, concomitantly ingested medications, and patient-specific factors that might alter the absorption of acetaminophen or impact serum acetaminophen concentrations should be considered. While this nomogram is helpful for decision-making pertaining to the initiation of NAC, further management and continuation of treatment must be patient specific and determined by the treating clinician.
In the United States, intravenous and oral formulations of NAC are available for treatment of acetaminophen toxicity. These formulations are equally efficacious in treating acetaminophen toxicity, with the exception of established hepatic failure, for which only the intravenous route has been studied.8 Oral administration results in vomiting in approximately 20% of patients, while intravenous administration may uncommonly result in severe anaphylactoid reactions. Three scenarios exist in which the intravenous route is preferred over the oral route: acetaminophen toxicity in pregnant women, acetaminophen-induced hepatic failure, and intractable vomiting preventing treatment with oral therapy.2 The intravenous formulation is Food and Drug Administration–approved for a 21-hour treatment duration, while the oral formulation is traditionally administered for a 72-hour duration.9,10
LIMITATIONS OF CURRENT TREATMENT ALGORITHMS
Treatment of acetaminophen poisoning must be tailored on an individual basis and should not follow predetermined durations of treatment. Not only might the FDA-approved 21-hour course of intravenous NAC be inadequate, but the standard 72-hour oral regimen duration may be excessive for many patients. Both the 21-hour and 72-hour protocols were developed based on the half-life and expected elimination of acetaminophen from the body. The traditional 72-hour oral protocol used in the United States was approved in 1985 and was based on the observation that, in patients who developed acetaminophen-induced fatal hepatotoxicity, the acetaminophen half-life was 12 hours, requiring five half-lives to eliminate the drug, for a total of 60 hours.11 The FDA then added an additional 12 hours to provide an additional margin of safety. Researchers in the United Kingdom developed an intravenous treatment protocol based on a shorter 4-hour acetaminophen half-life, calculated from observations of a larger pool of acetaminophen-poisoned patients and not just those with fatal hepatotoxicity.12 This 21-hour protocol—slightly longer than the five 4-hour half-lives needed—was subsequently approved in the United States as the intravenous formulation’s duration in 2004.
Case reports of hepatic failure have described treatment failures of intravenous NAC after the completion of the standard 21 hour treatment regimen.13 In a case report by Smith et al, a patient with significant single ingestion is described who ultimately developed hepatotoxicity despite receiving the FDA-approved 21-hour infusion.13 This patient ingested approximately 96 immediate-release, 500-mg acetaminophen tablets over approximately 1 hour. The initial acetaminophen concentration was 264 mcg/mL at 2.25 hours after ingestion. At 21 hours after ingestion, the patient’s acetaminophen levels remained elevated at 116 mcg/mL, but therapy with NAC was discontinued per protocol because AST and ALT levels were normal. A second peak in serum acetaminophen levels occurred after discontinuation of NAC, with levels reaching 228 mcg/mL approximately 48 hours after ingestion. Liver function worsened over the next few days with elevations in AST of greater than 4,000 U/L, PT of 51.4 seconds, and ammonia of 165 mcg/dL. Intravenous NAC was restarted and hepatic function improved gradually. The patient was ultimately discharged from the hospital. This case and others highlight the possible pitfalls of applying a protocolized, fixed treatment duration of NAC to all patients.
GOAL-DIRECTED TREATMENT RECOMMENDATIONS
Based on individual ingestion timing, quantities, and pharmacokinetics, patients should have pertinent laboratory markers monitored at least every 24 hours from presentation. The clinician may then consider the need to continue or discontinue NAC treatment every 24 hours from the initiation of NAC. Although notable limitations exist for both intravenous and oral treatment regimens, the literature uniformly recommends treating until serum acetaminophen levels are undetectable, regardless of NAC administration route. Based on the metabolism of acetaminophen, as long as serum concentrations of acetaminophen persist, further metabolism to the toxic NAPQI metabolite is possible. The ACMT addressed these concerns with the 2017 position paper, which strongly recommends all of the following criteria to be present prior to discontinuation of intravenous NAC: undetectable acetaminophen concentration, improving hepatic aminotransferases, and improving prognostic markers, including creatinine, lactate, pH, PT/INR, and phosphate.5 In cases in which laboratory and clinical parameters are abnormal, continued treatment beyond the established standard NAC treatment durations is warranted. However, once NAC treatment is extended beyond established protocol lengths, the subsequent dosing regimens that should be employed in these patients are not well studied. The ACMT Position Statement states the administration of an additional NAC bolus or extending the duration of the 6.25 mg/kg per hour maintenance infusion may be appropriate in this subset of patients requiring continued treatment.
Based on the positive outcomes in most patients receiving the 21-hour intravenous NAC protocol, the necessity of an additional 2 days of NAC treatment with the 72-hour oral regimen is uncertain. The ACMT Position Statement states “evidence supports using shorter oral NAC courses, provided that liver enzymes and synthetic function are normal or improving, and plasma acetaminophen concentration is undetectable.”5 Based on this and available literature, patients with a lower risk for developing hepatotoxicity may have oral NAC therapy discontinued after 24 to 48 hours of treatment if specific lab and patient parameters are within defined criteria.
The ACMT does not differentiate between adult and pediatric recommendations. However, the data used to develop the ACMT goal-directed treatment recommendations included both adult and pediatric patients.14,15 The same goal-directed treatment principles may be applied to pediatric patients, though studies have not specifically addressed pediatric goal-directed treatment of acetaminophen poisoning.
CONCLUSION
Acetaminophen-poisoned patients require evaluation not just prior to initiation of treatment, but also when deciding to discontinue NAC therapy. New literature supports reconsideration of standardized 21- and 72-hour NAC treatment protocols, as well as the notion that treatment duration should be guided by laboratory parameters rather than by route of NAC administration. Goal-directed or patient-tailored treatment is also supported by the ACMT.5 Treatment with NAC should continue in patients displaying hepatic toxicity or persistent elevations in acetaminophen levels. In these cases, the optimal treatment regimen in these cases remains unknown and further research into this area is merited.
According to the most recent Annual Report of the American Association of Poison Control Centers’ National Poison Data System, there were 51,081 patient exposures to acetaminophen or acetaminophen-containing products treated in a healthcare facility in 2018.1 The toxicity of acetaminophen is mediated by its metabolism into the electrophile N-acetyl-p-benzoquinone imine (NAPQI).2 If the amount of acetaminophen ingested is higher than recommended dosages, glutathione stores may become depleted as the liver tries to detoxify NAPQI and may no longer able to keep up with the demand. As the patient progresses 24 to 36 hours after ingestion, the onset of hepatic injury becomes apparent with elevations in aspartate aminotransferase (AST). AST elevations, should they occur, are almost always present by 36 hours.2 Maximum hepatic dysfunction, as well as elevations in prothrombin time, bilirubin, and serum creatinine, occurs 72 to 96 hours after a toxic ingestion. As a result of marked age-associated differences in the process of conjugating acetaminophen metabolites, infants and young children may be less susceptible to acetaminophen-associated hepatotoxicity, compared with their adult counterparts.3,4
This clinical progress note addresses acetaminophen toxicity treatment, focusing on N-acetylcysteine (NAC) dosing protocols and the 2017 American College of Medical Toxicology (ACMT) position statement, Duration of Intravenous Acetylcysteine Therapy Following Acetaminophen Overdose.5 We conducted a literature search via the PubMed database. The authors began by using the following Medical Subject Headings terms “acetaminophen overdose [title],” which yielded 299 articles, “acetaminophen hepatotoxicity [title],” which yielded 283 articles, and “acetaminophen N-acetylcysteine [title],” which yielded 335 titles. Variations of these terms were used to ensure exhaustive search results. The search results were reviewed for applicability to acetaminophen poisoning and especially goal-directed N-acetylcysteine treatment strategies.
INITIAL TREATMENT
Clarifying a patient’s history of ingestion is a crucial first step to the diagnosis and risk evaluation of acetaminophen-poisoned patients. The initial laboratory workup should include, at minimum, measurement (at 4 or more hours after ingestion) of serum acetaminophen level, AST and alanine aminotransferase (ALT), bilirubin, alkaline phosphatase, creatinine, prothrombin time and international normalized ratio (PT/INR), lactate, and ethanol levels. Gastric decontamination is not appropriate in most cases because of the rapid absorption of acetaminophen.2 Activated charcoal is most effective when given within the first 2 hours following ingestion. For acetaminophen-poisoned patients with severe hepatic toxicity, treating clinicians should consider consultation with a medical toxicologist, an individual with expertise, or the local Poison Control Center.5
The Rumack-Matthew nomogram provides guidance on the initiation of antidotal treatment for patients with acetaminophen overdose. This nomogram was developed to exhibit high sensitivity in patients at risk for hepatic damage from acetaminophen overdose.6 The mainstay for treatment of acetaminophen poisonings is NAC. Early administration of NAC can replenish depleted hepatic glutathione stores, which restores the liver’s ability to detoxify the toxic metabolite NAPQI. When administered within 6 to 8 hours following ingestion, NAC is nearly universally effective at preventing the subsequent hepatic damage from NAPQI.7 Consideration of individualized factors should lead the treating clinician to use the Rumack-Matthew nomogram within the appropriate contexts. Factors such as an unreliable timeline of ingestion by patients, concomitantly ingested medications, and patient-specific factors that might alter the absorption of acetaminophen or impact serum acetaminophen concentrations should be considered. While this nomogram is helpful for decision-making pertaining to the initiation of NAC, further management and continuation of treatment must be patient specific and determined by the treating clinician.
In the United States, intravenous and oral formulations of NAC are available for treatment of acetaminophen toxicity. These formulations are equally efficacious in treating acetaminophen toxicity, with the exception of established hepatic failure, for which only the intravenous route has been studied.8 Oral administration results in vomiting in approximately 20% of patients, while intravenous administration may uncommonly result in severe anaphylactoid reactions. Three scenarios exist in which the intravenous route is preferred over the oral route: acetaminophen toxicity in pregnant women, acetaminophen-induced hepatic failure, and intractable vomiting preventing treatment with oral therapy.2 The intravenous formulation is Food and Drug Administration–approved for a 21-hour treatment duration, while the oral formulation is traditionally administered for a 72-hour duration.9,10
LIMITATIONS OF CURRENT TREATMENT ALGORITHMS
Treatment of acetaminophen poisoning must be tailored on an individual basis and should not follow predetermined durations of treatment. Not only might the FDA-approved 21-hour course of intravenous NAC be inadequate, but the standard 72-hour oral regimen duration may be excessive for many patients. Both the 21-hour and 72-hour protocols were developed based on the half-life and expected elimination of acetaminophen from the body. The traditional 72-hour oral protocol used in the United States was approved in 1985 and was based on the observation that, in patients who developed acetaminophen-induced fatal hepatotoxicity, the acetaminophen half-life was 12 hours, requiring five half-lives to eliminate the drug, for a total of 60 hours.11 The FDA then added an additional 12 hours to provide an additional margin of safety. Researchers in the United Kingdom developed an intravenous treatment protocol based on a shorter 4-hour acetaminophen half-life, calculated from observations of a larger pool of acetaminophen-poisoned patients and not just those with fatal hepatotoxicity.12 This 21-hour protocol—slightly longer than the five 4-hour half-lives needed—was subsequently approved in the United States as the intravenous formulation’s duration in 2004.
Case reports of hepatic failure have described treatment failures of intravenous NAC after the completion of the standard 21 hour treatment regimen.13 In a case report by Smith et al, a patient with significant single ingestion is described who ultimately developed hepatotoxicity despite receiving the FDA-approved 21-hour infusion.13 This patient ingested approximately 96 immediate-release, 500-mg acetaminophen tablets over approximately 1 hour. The initial acetaminophen concentration was 264 mcg/mL at 2.25 hours after ingestion. At 21 hours after ingestion, the patient’s acetaminophen levels remained elevated at 116 mcg/mL, but therapy with NAC was discontinued per protocol because AST and ALT levels were normal. A second peak in serum acetaminophen levels occurred after discontinuation of NAC, with levels reaching 228 mcg/mL approximately 48 hours after ingestion. Liver function worsened over the next few days with elevations in AST of greater than 4,000 U/L, PT of 51.4 seconds, and ammonia of 165 mcg/dL. Intravenous NAC was restarted and hepatic function improved gradually. The patient was ultimately discharged from the hospital. This case and others highlight the possible pitfalls of applying a protocolized, fixed treatment duration of NAC to all patients.
GOAL-DIRECTED TREATMENT RECOMMENDATIONS
Based on individual ingestion timing, quantities, and pharmacokinetics, patients should have pertinent laboratory markers monitored at least every 24 hours from presentation. The clinician may then consider the need to continue or discontinue NAC treatment every 24 hours from the initiation of NAC. Although notable limitations exist for both intravenous and oral treatment regimens, the literature uniformly recommends treating until serum acetaminophen levels are undetectable, regardless of NAC administration route. Based on the metabolism of acetaminophen, as long as serum concentrations of acetaminophen persist, further metabolism to the toxic NAPQI metabolite is possible. The ACMT addressed these concerns with the 2017 position paper, which strongly recommends all of the following criteria to be present prior to discontinuation of intravenous NAC: undetectable acetaminophen concentration, improving hepatic aminotransferases, and improving prognostic markers, including creatinine, lactate, pH, PT/INR, and phosphate.5 In cases in which laboratory and clinical parameters are abnormal, continued treatment beyond the established standard NAC treatment durations is warranted. However, once NAC treatment is extended beyond established protocol lengths, the subsequent dosing regimens that should be employed in these patients are not well studied. The ACMT Position Statement states the administration of an additional NAC bolus or extending the duration of the 6.25 mg/kg per hour maintenance infusion may be appropriate in this subset of patients requiring continued treatment.
Based on the positive outcomes in most patients receiving the 21-hour intravenous NAC protocol, the necessity of an additional 2 days of NAC treatment with the 72-hour oral regimen is uncertain. The ACMT Position Statement states “evidence supports using shorter oral NAC courses, provided that liver enzymes and synthetic function are normal or improving, and plasma acetaminophen concentration is undetectable.”5 Based on this and available literature, patients with a lower risk for developing hepatotoxicity may have oral NAC therapy discontinued after 24 to 48 hours of treatment if specific lab and patient parameters are within defined criteria.
The ACMT does not differentiate between adult and pediatric recommendations. However, the data used to develop the ACMT goal-directed treatment recommendations included both adult and pediatric patients.14,15 The same goal-directed treatment principles may be applied to pediatric patients, though studies have not specifically addressed pediatric goal-directed treatment of acetaminophen poisoning.
CONCLUSION
Acetaminophen-poisoned patients require evaluation not just prior to initiation of treatment, but also when deciding to discontinue NAC therapy. New literature supports reconsideration of standardized 21- and 72-hour NAC treatment protocols, as well as the notion that treatment duration should be guided by laboratory parameters rather than by route of NAC administration. Goal-directed or patient-tailored treatment is also supported by the ACMT.5 Treatment with NAC should continue in patients displaying hepatic toxicity or persistent elevations in acetaminophen levels. In these cases, the optimal treatment regimen in these cases remains unknown and further research into this area is merited.
1. Gummin D, Mowry J, Spyker D, et al. 2018 Annual Report of the American Association of Poison Control Centers’ National Poison Data System (NPDS): 36th Annual Report. Clin Toxicol (Phila). 2019;57(12):1220‐1413. https://doi.org/10.1080/15563650.2019.1677022
2. Hoffman RS, Howland MA, Lewin NA, Nelson LS, Goldfrank LR, eds. Goldfrank’s Toxicologic Emergencies, Tenth Edition. McGraw Hill Education; 2015.
3. Miller RP, Roberts RJ, Fischer LJ. Acetaminophen elimination kinetics in neonates, children, and adults. Clin Pharmacol Ther. 1976;19:284-294. https://doi.org/10.1002/cpt1976193284
4. Rumack BH. Acetaminophen overdose in young children: treatment and effects of alcohol and other additional ingestants in 417 cases. Am J Dis Child. 1984;138(5):428-433. https://doi.org/10.1001/archpedi.1984.02140430006003
5. American College of Medical Toxicology. ACMT position statement: duration of intravenous acetylcysteine therapy following acetaminophen overdose. J Med Toxicol. 2017;13(1):126-127. https://doi.org/10.1007/s13181-016-0542-z
6. Matthew H. Acute acetaminophen poisoning. Clin Toxicol. 1973;6(1):9-11. https://doi.org/10.3109/15563657308991037
7. Smilkstein MJ, Knapp GL, Kulig KW, Rumack BH. Efficacy of oral N-acetylcysteine in the treatment of acetaminophen overdose: analysis of the national multicenter study (1976 to 1985). N Engl J Med. 1988;319(24):1557-1562. https://doi.org/10.1056/nejm198812153192401
8. Keays R, Harrison PM, Wendon JA, et al. Intravenous acetylcysteine in paracetamol induced fulminant hepatic failure: a prospective controlled trial. BMJ. 1991;303(6809):1026-1029. https://doi.org/10.1136/bmj.303.6809.1026
9. Acetadote ® (acetylcysteine injection). Package Insert. Cumberland Pharmaceuticals; 2004.
10. Acetylcysteine Solution. Package insert. Roxanne Laboratories; 2007.
11. Wong A, Graudins A. Risk prediction of hepatotoxicity in paracetamol poisoning. Clin Toxicol (Phila). 2017;55(8):879-892. https://doi.org/10.1080/15563650.2017.1317349
12. Prescott LF. Kinetics and metabolism of paracetamol and phenacetin. Br J Clin Pharmacol. 1980;10(Suppl 2):291S-298S. https://doi.org/10.1111/j.1365-2125.1980.tb01812.x
13. Smith SW, Howland MA, Hoffman RS, Nelson LS. Acetaminophen overdose with altered acetaminophen pharmacokinetics and hepatotoxicity associated with premature cessation of intravenous N-acetylcysteine therapy. Ann Pharmacother. 2008;42(9):1333-1339. https://doi.org/10.1345/aph.1k680
14. Woo OF, Mueller PD, Olsen KR, Anderson B, Kim SY. Shorter duration of oral N-Acetylcysteine therapy for acute acetaminophen overdose. Ann Emerg Med. 2000; 35(4):363-368. https://doi.org/10.1016/S0196-0644(00)70055-2
15. Betten D, Cantrell F, Thomas S, Williams S, Clark R. A prospective evaluation of shortened course oral N-acetylcysteine for the treatment of acute acetaminophen poisoning. Ann Emerg Med. 2007;50(3):272-279. https://doi.org/10.1016/j.annemergmed.2006.11.010
1. Gummin D, Mowry J, Spyker D, et al. 2018 Annual Report of the American Association of Poison Control Centers’ National Poison Data System (NPDS): 36th Annual Report. Clin Toxicol (Phila). 2019;57(12):1220‐1413. https://doi.org/10.1080/15563650.2019.1677022
2. Hoffman RS, Howland MA, Lewin NA, Nelson LS, Goldfrank LR, eds. Goldfrank’s Toxicologic Emergencies, Tenth Edition. McGraw Hill Education; 2015.
3. Miller RP, Roberts RJ, Fischer LJ. Acetaminophen elimination kinetics in neonates, children, and adults. Clin Pharmacol Ther. 1976;19:284-294. https://doi.org/10.1002/cpt1976193284
4. Rumack BH. Acetaminophen overdose in young children: treatment and effects of alcohol and other additional ingestants in 417 cases. Am J Dis Child. 1984;138(5):428-433. https://doi.org/10.1001/archpedi.1984.02140430006003
5. American College of Medical Toxicology. ACMT position statement: duration of intravenous acetylcysteine therapy following acetaminophen overdose. J Med Toxicol. 2017;13(1):126-127. https://doi.org/10.1007/s13181-016-0542-z
6. Matthew H. Acute acetaminophen poisoning. Clin Toxicol. 1973;6(1):9-11. https://doi.org/10.3109/15563657308991037
7. Smilkstein MJ, Knapp GL, Kulig KW, Rumack BH. Efficacy of oral N-acetylcysteine in the treatment of acetaminophen overdose: analysis of the national multicenter study (1976 to 1985). N Engl J Med. 1988;319(24):1557-1562. https://doi.org/10.1056/nejm198812153192401
8. Keays R, Harrison PM, Wendon JA, et al. Intravenous acetylcysteine in paracetamol induced fulminant hepatic failure: a prospective controlled trial. BMJ. 1991;303(6809):1026-1029. https://doi.org/10.1136/bmj.303.6809.1026
9. Acetadote ® (acetylcysteine injection). Package Insert. Cumberland Pharmaceuticals; 2004.
10. Acetylcysteine Solution. Package insert. Roxanne Laboratories; 2007.
11. Wong A, Graudins A. Risk prediction of hepatotoxicity in paracetamol poisoning. Clin Toxicol (Phila). 2017;55(8):879-892. https://doi.org/10.1080/15563650.2017.1317349
12. Prescott LF. Kinetics and metabolism of paracetamol and phenacetin. Br J Clin Pharmacol. 1980;10(Suppl 2):291S-298S. https://doi.org/10.1111/j.1365-2125.1980.tb01812.x
13. Smith SW, Howland MA, Hoffman RS, Nelson LS. Acetaminophen overdose with altered acetaminophen pharmacokinetics and hepatotoxicity associated with premature cessation of intravenous N-acetylcysteine therapy. Ann Pharmacother. 2008;42(9):1333-1339. https://doi.org/10.1345/aph.1k680
14. Woo OF, Mueller PD, Olsen KR, Anderson B, Kim SY. Shorter duration of oral N-Acetylcysteine therapy for acute acetaminophen overdose. Ann Emerg Med. 2000; 35(4):363-368. https://doi.org/10.1016/S0196-0644(00)70055-2
15. Betten D, Cantrell F, Thomas S, Williams S, Clark R. A prospective evaluation of shortened course oral N-acetylcysteine for the treatment of acute acetaminophen poisoning. Ann Emerg Med. 2007;50(3):272-279. https://doi.org/10.1016/j.annemergmed.2006.11.010
© 2020 Society of Hospital Medicine