User login
Hodgkin lymphoma survival has nearly tripled since the 1950s
Five-year relative survival for Hodgkin lymphoma increased 189% over the approximately 60 years from the early 1950s to 2013, according to investigators looking at data from the Surveillance, Epidemiology, and End Results Program.
During 1950-1954, the 5-year relative survival rate for Hodgkin lymphoma was 30%, compared with 86.6% in 2008-2013, said Ali H. Mokdad, PhD, and his associates at the Institute for Health Metrics and Evaluation at the University of Washington, Seattle.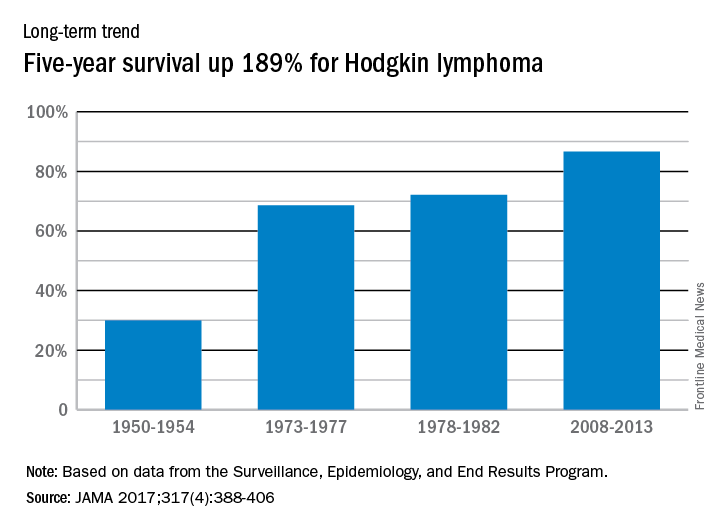
In 2014, mortality for Hodgkin lymphoma was 0.4 per 100,000 population, which put it 27th among the 29 included cancers, with about 36,000 years of life lost, which was 26th of the 29 cancers, Dr. Mokdad and his associates said. This part of their study used deidentified death records from the National Center for Health Statistics and population counts from the Census Bureau, the NCHS, and the Human Mortality Database.
Five-year relative survival for Hodgkin lymphoma increased 189% over the approximately 60 years from the early 1950s to 2013, according to investigators looking at data from the Surveillance, Epidemiology, and End Results Program.
During 1950-1954, the 5-year relative survival rate for Hodgkin lymphoma was 30%, compared with 86.6% in 2008-2013, said Ali H. Mokdad, PhD, and his associates at the Institute for Health Metrics and Evaluation at the University of Washington, Seattle.
In 2014, mortality for Hodgkin lymphoma was 0.4 per 100,000 population, which put it 27th among the 29 included cancers, with about 36,000 years of life lost, which was 26th of the 29 cancers, Dr. Mokdad and his associates said. This part of their study used deidentified death records from the National Center for Health Statistics and population counts from the Census Bureau, the NCHS, and the Human Mortality Database.
Five-year relative survival for Hodgkin lymphoma increased 189% over the approximately 60 years from the early 1950s to 2013, according to investigators looking at data from the Surveillance, Epidemiology, and End Results Program.
During 1950-1954, the 5-year relative survival rate for Hodgkin lymphoma was 30%, compared with 86.6% in 2008-2013, said Ali H. Mokdad, PhD, and his associates at the Institute for Health Metrics and Evaluation at the University of Washington, Seattle.
In 2014, mortality for Hodgkin lymphoma was 0.4 per 100,000 population, which put it 27th among the 29 included cancers, with about 36,000 years of life lost, which was 26th of the 29 cancers, Dr. Mokdad and his associates said. This part of their study used deidentified death records from the National Center for Health Statistics and population counts from the Census Bureau, the NCHS, and the Human Mortality Database.
FROM JAMA
Circulating DNA catches lymphoma relapse early
A newer technique aimed at detect circulating tumor DNA in the blood – cancer personalized profiling by deep sequencing (CAPP-Seq) – detected recurrence of diffuse large B cell lymphoma more than 6 months earlier than radiographic findings in a study at Stanford (Calif.) University, where the technique was invented.
The findings signal another win for “liquid biopsy,” the measurement of tumor DNA circulating in the blood, which is rapidly emerging as a quick and powerful tool for the diagnosis of a range of cancers and tumor subtypes, and prediction of tumor behavior and treatment response. Investigators at Stanford and elsewhere are studying liquid biopsy not only for lymphoma, but also for colorectal, thyroid, breast, prostate, and most other cancers. The Stanford team recently reported that its circulating DNA-detecting CAPP-Seq technique also helps in lung cancer.
In the new study, Stanford used CAPP-Seq (Cancer Personalized Profiling by deep Sequencing), which it called “an ultrasensitive capture-based targeted sequencing method” to analyze 166 plasma and 118 tissue samples from 92 patients with diffuse large B cell lymphoma (DLBCL) at diagnosis and various point afterward. The team compared the results to radiologic, and other standard diagnostic and monitoring techniques (Sci Transl Med. 2016 Nov 9;8[364]:364ra155).
At diagnosis, the amount of circulating DNA (ctDNA) correlated strongly with clinical indices and was independently predictive of patient outcomes; “whereas 100% of pretreatment samples had detectable ctDNA, only 37% of samples had abnormally high serum” lactate dehydrogenase, currently the most commonly used biomarker for DLBCL, said investigators, led by research fellow Florian Scherer, MD.
The group detected ctDNA in 73% of patients (8/11) who eventually relapsed a mean of 188 days before relapse was detected by standard-of-care radiologic techniques.
CAPP-Seq identified nine patients with a particular type of activated B cell-like tumor, for whom ibrutinib (Imbruvica) is particularly effective; ctDNA also predicted the transformation of indolent follicular lymphoma to DLBCL “with high sensitivity and specificity,” the group reported.
Stanford anticipates “ctDNA will have broad utility for dissecting tumor heterogeneity within and between patients with lymphomas and other cancer types, with applications for the identification of adverse risk groups, the discovery of resistance mechanisms to diverse therapies, and the development of risk-adapted therapeutics.”
The team said its approach “outperformed immunoglobulin sequencing and radiographic imaging for the detection of minimal residual disease and facilitated noninvasive identification of emergent resistance mutations to targeted therapies.” Meanwhile, while biomarkers hold “great promise for risk stratification and therapeutic targeting,” they are “currently difficult to measure in clinical settings,” the investigators said.
Roche bought the rights to CAPP-Seq from Stanford in 2015. Several authors are coinventors on patent applications for CAPP-Seq and also Roche consultants. Two are employees. Dr. Scherer had no disclosures. The work was funded by Stanford, the American Society of Hematology, the National Cancer Institute, and others.
A newer technique aimed at detect circulating tumor DNA in the blood – cancer personalized profiling by deep sequencing (CAPP-Seq) – detected recurrence of diffuse large B cell lymphoma more than 6 months earlier than radiographic findings in a study at Stanford (Calif.) University, where the technique was invented.
The findings signal another win for “liquid biopsy,” the measurement of tumor DNA circulating in the blood, which is rapidly emerging as a quick and powerful tool for the diagnosis of a range of cancers and tumor subtypes, and prediction of tumor behavior and treatment response. Investigators at Stanford and elsewhere are studying liquid biopsy not only for lymphoma, but also for colorectal, thyroid, breast, prostate, and most other cancers. The Stanford team recently reported that its circulating DNA-detecting CAPP-Seq technique also helps in lung cancer.
In the new study, Stanford used CAPP-Seq (Cancer Personalized Profiling by deep Sequencing), which it called “an ultrasensitive capture-based targeted sequencing method” to analyze 166 plasma and 118 tissue samples from 92 patients with diffuse large B cell lymphoma (DLBCL) at diagnosis and various point afterward. The team compared the results to radiologic, and other standard diagnostic and monitoring techniques (Sci Transl Med. 2016 Nov 9;8[364]:364ra155).
At diagnosis, the amount of circulating DNA (ctDNA) correlated strongly with clinical indices and was independently predictive of patient outcomes; “whereas 100% of pretreatment samples had detectable ctDNA, only 37% of samples had abnormally high serum” lactate dehydrogenase, currently the most commonly used biomarker for DLBCL, said investigators, led by research fellow Florian Scherer, MD.
The group detected ctDNA in 73% of patients (8/11) who eventually relapsed a mean of 188 days before relapse was detected by standard-of-care radiologic techniques.
CAPP-Seq identified nine patients with a particular type of activated B cell-like tumor, for whom ibrutinib (Imbruvica) is particularly effective; ctDNA also predicted the transformation of indolent follicular lymphoma to DLBCL “with high sensitivity and specificity,” the group reported.
Stanford anticipates “ctDNA will have broad utility for dissecting tumor heterogeneity within and between patients with lymphomas and other cancer types, with applications for the identification of adverse risk groups, the discovery of resistance mechanisms to diverse therapies, and the development of risk-adapted therapeutics.”
The team said its approach “outperformed immunoglobulin sequencing and radiographic imaging for the detection of minimal residual disease and facilitated noninvasive identification of emergent resistance mutations to targeted therapies.” Meanwhile, while biomarkers hold “great promise for risk stratification and therapeutic targeting,” they are “currently difficult to measure in clinical settings,” the investigators said.
Roche bought the rights to CAPP-Seq from Stanford in 2015. Several authors are coinventors on patent applications for CAPP-Seq and also Roche consultants. Two are employees. Dr. Scherer had no disclosures. The work was funded by Stanford, the American Society of Hematology, the National Cancer Institute, and others.
A newer technique aimed at detect circulating tumor DNA in the blood – cancer personalized profiling by deep sequencing (CAPP-Seq) – detected recurrence of diffuse large B cell lymphoma more than 6 months earlier than radiographic findings in a study at Stanford (Calif.) University, where the technique was invented.
The findings signal another win for “liquid biopsy,” the measurement of tumor DNA circulating in the blood, which is rapidly emerging as a quick and powerful tool for the diagnosis of a range of cancers and tumor subtypes, and prediction of tumor behavior and treatment response. Investigators at Stanford and elsewhere are studying liquid biopsy not only for lymphoma, but also for colorectal, thyroid, breast, prostate, and most other cancers. The Stanford team recently reported that its circulating DNA-detecting CAPP-Seq technique also helps in lung cancer.
In the new study, Stanford used CAPP-Seq (Cancer Personalized Profiling by deep Sequencing), which it called “an ultrasensitive capture-based targeted sequencing method” to analyze 166 plasma and 118 tissue samples from 92 patients with diffuse large B cell lymphoma (DLBCL) at diagnosis and various point afterward. The team compared the results to radiologic, and other standard diagnostic and monitoring techniques (Sci Transl Med. 2016 Nov 9;8[364]:364ra155).
At diagnosis, the amount of circulating DNA (ctDNA) correlated strongly with clinical indices and was independently predictive of patient outcomes; “whereas 100% of pretreatment samples had detectable ctDNA, only 37% of samples had abnormally high serum” lactate dehydrogenase, currently the most commonly used biomarker for DLBCL, said investigators, led by research fellow Florian Scherer, MD.
The group detected ctDNA in 73% of patients (8/11) who eventually relapsed a mean of 188 days before relapse was detected by standard-of-care radiologic techniques.
CAPP-Seq identified nine patients with a particular type of activated B cell-like tumor, for whom ibrutinib (Imbruvica) is particularly effective; ctDNA also predicted the transformation of indolent follicular lymphoma to DLBCL “with high sensitivity and specificity,” the group reported.
Stanford anticipates “ctDNA will have broad utility for dissecting tumor heterogeneity within and between patients with lymphomas and other cancer types, with applications for the identification of adverse risk groups, the discovery of resistance mechanisms to diverse therapies, and the development of risk-adapted therapeutics.”
The team said its approach “outperformed immunoglobulin sequencing and radiographic imaging for the detection of minimal residual disease and facilitated noninvasive identification of emergent resistance mutations to targeted therapies.” Meanwhile, while biomarkers hold “great promise for risk stratification and therapeutic targeting,” they are “currently difficult to measure in clinical settings,” the investigators said.
Roche bought the rights to CAPP-Seq from Stanford in 2015. Several authors are coinventors on patent applications for CAPP-Seq and also Roche consultants. Two are employees. Dr. Scherer had no disclosures. The work was funded by Stanford, the American Society of Hematology, the National Cancer Institute, and others.
FROM SCIENCE TRANSLATIONAL MEDICINE
Key clinical point: Major finding: Circulating tumor DNA was found in 73% of relapse patients a mean of 188 days before relapse was detected by standard-of-care radiologic techniques. Circulating tumor DNA was found in the plasma of 100% of patients at diagnosis, but only 37% had abnormally high serum lactate dehydrogenase, currently the most commonly used biomarker.
Data source: Analysis of 166 plasma and 118 tissue samples from 92 patients with diffuse large B cell lymphoma.
Disclosures: Roche bought the rights to CAPP-Seq from Stanford (Calif.) University in 2015. Several authors are coinventors on patent applications for CAPP-Seq and also Roche consults. Two are employees. The work was funded by Stanford, the American Society of Hematology, the National Cancer Institute, and others.
Study provides new insight into B-cell metabolism
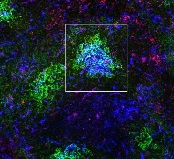
the spleen of a mouse, showing
inactivated GSK3 (magenta)
in B cells (blue) near follicular
dendritic cells (green).
Image from the lab of
Robert Rickert, PhD
Research published in Nature Immunology helps explain how B-cell metabolism adapts to different environments.
The study suggests the protein GSK3 acts as a metabolic checkpoint regulator in B cells, promoting the survival of circulating B cells while limiting the growth and proliferation of B cells in germinal centers.
“Our research shows that the protein GSK3 plays a crucial role in helping B cells meet the energy needs of their distinct states,” said study author Robert Rickert, PhD, of Sanford Burnham Prebys Medical Discovery Institute in La Jolla, California.
“The findings are particularly relevant for certain B-cell pathologies, including lymphoma subtypes, where there is an increased demand for energy to support the hyperproliferation of cells in a microenvironment that may be limited in nutrients.”
Dr Rickert and his colleagues noted that B cells predominate in a quiescent state until they encounter an antigen, which prompts the cells to grow, proliferate, and differentiate.
The team’s new study showed that GSK3 adjusts B-cell metabolism to match the needs of these different cell states.
In circulating B cells, GSK3 limits overall metabolic activity. In proliferating B cells in germinal centers, GSK3 slows glycolysis and the production of mitochondria.
In fact, GSK3 function is essential for B-cell survival in germinal centers. To understand why, the researchers looked at how B cells in these regions generate energy.
The team found that because these B cells are so metabolically active, they consume nearly all available glucose. That switches on glycolysis.
High glycolytic activity leads to an accumulation of toxic reactive oxygen species, as does rapid manufacture of mitochondria, which tend to leak the same chemicals.
Thus, by restraining the metabolism in specific ways, GSK3 prevents cell death induced by reactive oxygen species.
“Our results were really surprising,” Dr Rickert said. “Until now, we would have thought that slowing metabolism would only be important for preventing B cells from becoming cancerous, which it indeed may be. These studies provide insight into the dynamic nature of B-cell metabolism that literally ‘fuels’ differentiation in the germinal center to produce an effective antibody response.”
“It’s not yet clear whether or how GSK3 might be a target for future therapies for B cell-related diseases, but this research opens a lot of doors for further studies. To start with, we plan to investigate how GSK3 is regulated in lymphoma and how that relates to changes in metabolism. That research could lead to new approaches to treating lymphoma.”
This research was performed in collaboration with scientists at Eli Lilly and the Lunenfeld-Tanenbaum Research Institute at the University of Toronto. Funding was provided by the National Institutes of Health, the Lilly Research Award Program, the Arthritis National Research Foundation, and the Canadian Institutes of Health Research. ![]()

the spleen of a mouse, showing
inactivated GSK3 (magenta)
in B cells (blue) near follicular
dendritic cells (green).
Image from the lab of
Robert Rickert, PhD
Research published in Nature Immunology helps explain how B-cell metabolism adapts to different environments.
The study suggests the protein GSK3 acts as a metabolic checkpoint regulator in B cells, promoting the survival of circulating B cells while limiting the growth and proliferation of B cells in germinal centers.
“Our research shows that the protein GSK3 plays a crucial role in helping B cells meet the energy needs of their distinct states,” said study author Robert Rickert, PhD, of Sanford Burnham Prebys Medical Discovery Institute in La Jolla, California.
“The findings are particularly relevant for certain B-cell pathologies, including lymphoma subtypes, where there is an increased demand for energy to support the hyperproliferation of cells in a microenvironment that may be limited in nutrients.”
Dr Rickert and his colleagues noted that B cells predominate in a quiescent state until they encounter an antigen, which prompts the cells to grow, proliferate, and differentiate.
The team’s new study showed that GSK3 adjusts B-cell metabolism to match the needs of these different cell states.
In circulating B cells, GSK3 limits overall metabolic activity. In proliferating B cells in germinal centers, GSK3 slows glycolysis and the production of mitochondria.
In fact, GSK3 function is essential for B-cell survival in germinal centers. To understand why, the researchers looked at how B cells in these regions generate energy.
The team found that because these B cells are so metabolically active, they consume nearly all available glucose. That switches on glycolysis.
High glycolytic activity leads to an accumulation of toxic reactive oxygen species, as does rapid manufacture of mitochondria, which tend to leak the same chemicals.
Thus, by restraining the metabolism in specific ways, GSK3 prevents cell death induced by reactive oxygen species.
“Our results were really surprising,” Dr Rickert said. “Until now, we would have thought that slowing metabolism would only be important for preventing B cells from becoming cancerous, which it indeed may be. These studies provide insight into the dynamic nature of B-cell metabolism that literally ‘fuels’ differentiation in the germinal center to produce an effective antibody response.”
“It’s not yet clear whether or how GSK3 might be a target for future therapies for B cell-related diseases, but this research opens a lot of doors for further studies. To start with, we plan to investigate how GSK3 is regulated in lymphoma and how that relates to changes in metabolism. That research could lead to new approaches to treating lymphoma.”
This research was performed in collaboration with scientists at Eli Lilly and the Lunenfeld-Tanenbaum Research Institute at the University of Toronto. Funding was provided by the National Institutes of Health, the Lilly Research Award Program, the Arthritis National Research Foundation, and the Canadian Institutes of Health Research. ![]()

the spleen of a mouse, showing
inactivated GSK3 (magenta)
in B cells (blue) near follicular
dendritic cells (green).
Image from the lab of
Robert Rickert, PhD
Research published in Nature Immunology helps explain how B-cell metabolism adapts to different environments.
The study suggests the protein GSK3 acts as a metabolic checkpoint regulator in B cells, promoting the survival of circulating B cells while limiting the growth and proliferation of B cells in germinal centers.
“Our research shows that the protein GSK3 plays a crucial role in helping B cells meet the energy needs of their distinct states,” said study author Robert Rickert, PhD, of Sanford Burnham Prebys Medical Discovery Institute in La Jolla, California.
“The findings are particularly relevant for certain B-cell pathologies, including lymphoma subtypes, where there is an increased demand for energy to support the hyperproliferation of cells in a microenvironment that may be limited in nutrients.”
Dr Rickert and his colleagues noted that B cells predominate in a quiescent state until they encounter an antigen, which prompts the cells to grow, proliferate, and differentiate.
The team’s new study showed that GSK3 adjusts B-cell metabolism to match the needs of these different cell states.
In circulating B cells, GSK3 limits overall metabolic activity. In proliferating B cells in germinal centers, GSK3 slows glycolysis and the production of mitochondria.
In fact, GSK3 function is essential for B-cell survival in germinal centers. To understand why, the researchers looked at how B cells in these regions generate energy.
The team found that because these B cells are so metabolically active, they consume nearly all available glucose. That switches on glycolysis.
High glycolytic activity leads to an accumulation of toxic reactive oxygen species, as does rapid manufacture of mitochondria, which tend to leak the same chemicals.
Thus, by restraining the metabolism in specific ways, GSK3 prevents cell death induced by reactive oxygen species.
“Our results were really surprising,” Dr Rickert said. “Until now, we would have thought that slowing metabolism would only be important for preventing B cells from becoming cancerous, which it indeed may be. These studies provide insight into the dynamic nature of B-cell metabolism that literally ‘fuels’ differentiation in the germinal center to produce an effective antibody response.”
“It’s not yet clear whether or how GSK3 might be a target for future therapies for B cell-related diseases, but this research opens a lot of doors for further studies. To start with, we plan to investigate how GSK3 is regulated in lymphoma and how that relates to changes in metabolism. That research could lead to new approaches to treating lymphoma.”
This research was performed in collaboration with scientists at Eli Lilly and the Lunenfeld-Tanenbaum Research Institute at the University of Toronto. Funding was provided by the National Institutes of Health, the Lilly Research Award Program, the Arthritis National Research Foundation, and the Canadian Institutes of Health Research. ![]()
Study quantifies 5-year survival rates for blood cancers

chemotherapy
Photo by Rhoda Baer
A new study shows that 5-year survival rates for US patients with hematologic malignancies have increased greatly since the 1950s, but there is still room for improvement, particularly for patients with acute myeloid leukemia (AML).
Researchers found the absolute difference in improvement for 5-year survival from 1950-1954 to 2008-2013 ranged from 38.2% for non-Hodgkin lymphoma (NHL) to 56.6% for Hodgkin lymphoma.
And although the 5-year survival rate for Hodgkin lymphoma patients reached 86.6% for 2008-2013, the 5-year survival rate for patients with AML only reached 27.4%.
This study also revealed large disparities in overall cancer mortality rates between different counties across the country.
Ali H. Mokdad, PhD, of the Institute for Health Metrics and Evaluation in Seattle, Washington, and his colleagues reported these findings in JAMA.
Overall cancer deaths
The researchers found there were 19,511,910 cancer deaths recorded in the US between 1980 and 2014. Cancer mortality decreased by 20.1% between 1980 and 2014, from 240.2 deaths per 100,000 people to 192.0 deaths per 100,000 people.
In 1980, cancer mortality ranged from 130.6 per 100,000 in Summit County, Colorado, to 386.9 per 100,000 in North Slope Borough, Alaska.
In 2014, cancer mortality ranged from 70.7 per 100,000 in Summit County, Colorado, to 503.1 per 100,000 in Union County, Florida.
“Such significant disparities among US counties is unacceptable,” Dr Mokdad said. “Every person should have access to early screenings for cancer, as well as adequate treatment.”
Mortality rates for hematologic malignancies
In 2014, the mortality rates, per 100,000 people, for hematologic malignancies were:
- 0.4 for Hodgkin lymphoma (rank out of all cancers, 27)
- 8.3 for NHL (rank, 7)
- 3.9 for multiple myeloma (rank, 16)
- 9.0 for all leukemias (rank, 6)
- 0.7 for acute lymphoid leukemia (ALL)
- 2.6 for chronic lymphoid leukemia (CLL)
- 5.1 for AML
- 0.6 for chronic myeloid leukemia (CML).
The leukemia subtypes were not assigned a rank.
5-year survival rates for hematologic malignancies
Hodgkin lymphoma
- 30% for 1950-54
- 68.6% for 1973-77
- 72.1% for 1978-82
- 86.6% for 2008-2013
- Absolute difference (between the first and latest year of data), 56.6%.
NHL
- 33% for 1950-54
- 45.3% for 1973-77
- 48.7% for 1978-82
- 71.2% for 2008-2013
- Absolute difference, 38.2%.
Multiple myeloma
- 6% for 1950-54
- 23.4% for 1973-77
- 26.6% for 1978-82
- 49.8% for 2008-2013
- Absolute difference, 43.8%.
Leukemia
- 10% for 1950-54
- 34% for 1973-77
- 36.3% for 1978-82
- 60.1% for 2008-2013
- Absolute difference, 50.1%.
ALL
- 39.2% for 1973-77
- 50.5% for 1978-82
- 68.1% for 2008-2013
- Absolute difference, 28.9%.
CLL
- 67% for 1973-77
- 66.3% for 1978-82
- 82.5% for 2008-2013
- Absolute difference, 15.5%.
AML
- 6.2% for 1973-77
- 7.9% for 1978-82
- 27.4% for 2008-2013
- Absolute difference, 21.2%.
CML
- 21.1% for 1973-77
- 25.8% for 1978-82
- 66.4% for 2008-2013
- Absolute difference, 45.3%.
For the leukemia subtypes, there was no data for 1950 to 1954. ![]()

chemotherapy
Photo by Rhoda Baer
A new study shows that 5-year survival rates for US patients with hematologic malignancies have increased greatly since the 1950s, but there is still room for improvement, particularly for patients with acute myeloid leukemia (AML).
Researchers found the absolute difference in improvement for 5-year survival from 1950-1954 to 2008-2013 ranged from 38.2% for non-Hodgkin lymphoma (NHL) to 56.6% for Hodgkin lymphoma.
And although the 5-year survival rate for Hodgkin lymphoma patients reached 86.6% for 2008-2013, the 5-year survival rate for patients with AML only reached 27.4%.
This study also revealed large disparities in overall cancer mortality rates between different counties across the country.
Ali H. Mokdad, PhD, of the Institute for Health Metrics and Evaluation in Seattle, Washington, and his colleagues reported these findings in JAMA.
Overall cancer deaths
The researchers found there were 19,511,910 cancer deaths recorded in the US between 1980 and 2014. Cancer mortality decreased by 20.1% between 1980 and 2014, from 240.2 deaths per 100,000 people to 192.0 deaths per 100,000 people.
In 1980, cancer mortality ranged from 130.6 per 100,000 in Summit County, Colorado, to 386.9 per 100,000 in North Slope Borough, Alaska.
In 2014, cancer mortality ranged from 70.7 per 100,000 in Summit County, Colorado, to 503.1 per 100,000 in Union County, Florida.
“Such significant disparities among US counties is unacceptable,” Dr Mokdad said. “Every person should have access to early screenings for cancer, as well as adequate treatment.”
Mortality rates for hematologic malignancies
In 2014, the mortality rates, per 100,000 people, for hematologic malignancies were:
- 0.4 for Hodgkin lymphoma (rank out of all cancers, 27)
- 8.3 for NHL (rank, 7)
- 3.9 for multiple myeloma (rank, 16)
- 9.0 for all leukemias (rank, 6)
- 0.7 for acute lymphoid leukemia (ALL)
- 2.6 for chronic lymphoid leukemia (CLL)
- 5.1 for AML
- 0.6 for chronic myeloid leukemia (CML).
The leukemia subtypes were not assigned a rank.
5-year survival rates for hematologic malignancies
Hodgkin lymphoma
- 30% for 1950-54
- 68.6% for 1973-77
- 72.1% for 1978-82
- 86.6% for 2008-2013
- Absolute difference (between the first and latest year of data), 56.6%.
NHL
- 33% for 1950-54
- 45.3% for 1973-77
- 48.7% for 1978-82
- 71.2% for 2008-2013
- Absolute difference, 38.2%.
Multiple myeloma
- 6% for 1950-54
- 23.4% for 1973-77
- 26.6% for 1978-82
- 49.8% for 2008-2013
- Absolute difference, 43.8%.
Leukemia
- 10% for 1950-54
- 34% for 1973-77
- 36.3% for 1978-82
- 60.1% for 2008-2013
- Absolute difference, 50.1%.
ALL
- 39.2% for 1973-77
- 50.5% for 1978-82
- 68.1% for 2008-2013
- Absolute difference, 28.9%.
CLL
- 67% for 1973-77
- 66.3% for 1978-82
- 82.5% for 2008-2013
- Absolute difference, 15.5%.
AML
- 6.2% for 1973-77
- 7.9% for 1978-82
- 27.4% for 2008-2013
- Absolute difference, 21.2%.
CML
- 21.1% for 1973-77
- 25.8% for 1978-82
- 66.4% for 2008-2013
- Absolute difference, 45.3%.
For the leukemia subtypes, there was no data for 1950 to 1954. ![]()

chemotherapy
Photo by Rhoda Baer
A new study shows that 5-year survival rates for US patients with hematologic malignancies have increased greatly since the 1950s, but there is still room for improvement, particularly for patients with acute myeloid leukemia (AML).
Researchers found the absolute difference in improvement for 5-year survival from 1950-1954 to 2008-2013 ranged from 38.2% for non-Hodgkin lymphoma (NHL) to 56.6% for Hodgkin lymphoma.
And although the 5-year survival rate for Hodgkin lymphoma patients reached 86.6% for 2008-2013, the 5-year survival rate for patients with AML only reached 27.4%.
This study also revealed large disparities in overall cancer mortality rates between different counties across the country.
Ali H. Mokdad, PhD, of the Institute for Health Metrics and Evaluation in Seattle, Washington, and his colleagues reported these findings in JAMA.
Overall cancer deaths
The researchers found there were 19,511,910 cancer deaths recorded in the US between 1980 and 2014. Cancer mortality decreased by 20.1% between 1980 and 2014, from 240.2 deaths per 100,000 people to 192.0 deaths per 100,000 people.
In 1980, cancer mortality ranged from 130.6 per 100,000 in Summit County, Colorado, to 386.9 per 100,000 in North Slope Borough, Alaska.
In 2014, cancer mortality ranged from 70.7 per 100,000 in Summit County, Colorado, to 503.1 per 100,000 in Union County, Florida.
“Such significant disparities among US counties is unacceptable,” Dr Mokdad said. “Every person should have access to early screenings for cancer, as well as adequate treatment.”
Mortality rates for hematologic malignancies
In 2014, the mortality rates, per 100,000 people, for hematologic malignancies were:
- 0.4 for Hodgkin lymphoma (rank out of all cancers, 27)
- 8.3 for NHL (rank, 7)
- 3.9 for multiple myeloma (rank, 16)
- 9.0 for all leukemias (rank, 6)
- 0.7 for acute lymphoid leukemia (ALL)
- 2.6 for chronic lymphoid leukemia (CLL)
- 5.1 for AML
- 0.6 for chronic myeloid leukemia (CML).
The leukemia subtypes were not assigned a rank.
5-year survival rates for hematologic malignancies
Hodgkin lymphoma
- 30% for 1950-54
- 68.6% for 1973-77
- 72.1% for 1978-82
- 86.6% for 2008-2013
- Absolute difference (between the first and latest year of data), 56.6%.
NHL
- 33% for 1950-54
- 45.3% for 1973-77
- 48.7% for 1978-82
- 71.2% for 2008-2013
- Absolute difference, 38.2%.
Multiple myeloma
- 6% for 1950-54
- 23.4% for 1973-77
- 26.6% for 1978-82
- 49.8% for 2008-2013
- Absolute difference, 43.8%.
Leukemia
- 10% for 1950-54
- 34% for 1973-77
- 36.3% for 1978-82
- 60.1% for 2008-2013
- Absolute difference, 50.1%.
ALL
- 39.2% for 1973-77
- 50.5% for 1978-82
- 68.1% for 2008-2013
- Absolute difference, 28.9%.
CLL
- 67% for 1973-77
- 66.3% for 1978-82
- 82.5% for 2008-2013
- Absolute difference, 15.5%.
AML
- 6.2% for 1973-77
- 7.9% for 1978-82
- 27.4% for 2008-2013
- Absolute difference, 21.2%.
CML
- 21.1% for 1973-77
- 25.8% for 1978-82
- 66.4% for 2008-2013
- Absolute difference, 45.3%.
For the leukemia subtypes, there was no data for 1950 to 1954. ![]()
Improving the efficacy of obinutuzumab

Preclinical research suggests that immune stimulation through Toll-like receptor 7 (TLR7) agonism can enhance the efficacy of obinutuzumab in lymphoma.
Researchers found that combining the anti-CD20 monoclonal antibody obinutuzumab with the TLR7 agonist R848 improved survival in lab mice with lymphoma.
The combination also demonstrated efficacy against chronic lymphocytic leukemia (CLL) cells in vitro.
Tim Illidge, PhD, MBBS, of the University of Manchester in the UK, and his colleagues reported these findings in the journal Leukemia.
The research was funded by the Kay Kendall Leukaemia Fund and Cancer Research UK in collaboration with Roche Pharmaceutical Research and Early Development.
The researchers said they initially found that R848 activates immune cells in vivo and enhances obinutuzumab-mediated antitumor effector mechanisms in vitro.
The team therefore went on to test R848 and obinutuzumab in C57Bl/6 mice bearing human CD20+ lymphoma (EL4). The mice received obinutuzumab modified to express the murine glycoengineered IgG2a Fc region (m2a) starting 1 day after tumor inoculation and systemic R848 once weekly for 4 weeks.
The researchers found that monotherapy with either obinutuzumab or R848 significantly improved survival compared to control (P<0.0001), but only 8% to 15% of mice that received monotherapy were long-term survivors (living more than 90 days).
Mice that received obinutuzumab in combination with R848 had significantly better survival than mice that received either monotherapy (P<0.0001). And about 70% of mice receiving the combination remained tumor-free out to 95 days.
Furthermore, long-term survivors that had received the combination treatment were protected from tumor re-challenge.
The researchers also tested the combination in a second model—human CD20 transgenic mice, which express the human CD20 antigen on normal B cells. The team said this model is more akin to the clinical situation.
The mice received treatment 7 days after the inoculation of EL4hCD20 cells. Mice that received obinutuzumab monotherapy had significantly better survival than control mice (P=0.02), but there were no long-term survivors. For mice that received R848 monotherapy, survival was not significantly different from that of controls.
Mice that received R848 in combination with obinutuzumab had significantly better survival than mice that received obinutuzumab alone (P=0.003).
In fact, 6 of the 12 mice that received the combination were long-term survivors. And 5 of these mice rejected tumor re-challenge.
“We were excited when we discovered that combining obinutuzumab with TLR7 activation significantly enhanced survival of animals with lymphoma by effectively eradicating tumors,” Dr Illidge said. “Clearly, more work needs to be done to assess the impact of this combination on humans, but this study is, nevertheless, very promising.”
The researchers said the primary antitumor activity of the combination is dependent on natural killer cells and CD4 helper T cells but not on CD8 killer T cells.
“While the combination therapy was highly effective, CD8 killer T cells did not play a major role in the therapy,” said Eleanor Cheadle, PhD, also of the University of Manchester.
“Given the important role that killer T cells can play in long-term protection from tumor regrowth, we are looking at ways to enhance activation of these cells after obinutuzumab therapy.”
The researchers also found that, in vitro, R848 significantly enhanced natural killer cell-mediated antibody-dependent cellular cytotoxicity against obinutuzumab-opsonized CLL cells and significantly increased non-specific, antibody-independent killing of CLL cells. ![]()

Preclinical research suggests that immune stimulation through Toll-like receptor 7 (TLR7) agonism can enhance the efficacy of obinutuzumab in lymphoma.
Researchers found that combining the anti-CD20 monoclonal antibody obinutuzumab with the TLR7 agonist R848 improved survival in lab mice with lymphoma.
The combination also demonstrated efficacy against chronic lymphocytic leukemia (CLL) cells in vitro.
Tim Illidge, PhD, MBBS, of the University of Manchester in the UK, and his colleagues reported these findings in the journal Leukemia.
The research was funded by the Kay Kendall Leukaemia Fund and Cancer Research UK in collaboration with Roche Pharmaceutical Research and Early Development.
The researchers said they initially found that R848 activates immune cells in vivo and enhances obinutuzumab-mediated antitumor effector mechanisms in vitro.
The team therefore went on to test R848 and obinutuzumab in C57Bl/6 mice bearing human CD20+ lymphoma (EL4). The mice received obinutuzumab modified to express the murine glycoengineered IgG2a Fc region (m2a) starting 1 day after tumor inoculation and systemic R848 once weekly for 4 weeks.
The researchers found that monotherapy with either obinutuzumab or R848 significantly improved survival compared to control (P<0.0001), but only 8% to 15% of mice that received monotherapy were long-term survivors (living more than 90 days).
Mice that received obinutuzumab in combination with R848 had significantly better survival than mice that received either monotherapy (P<0.0001). And about 70% of mice receiving the combination remained tumor-free out to 95 days.
Furthermore, long-term survivors that had received the combination treatment were protected from tumor re-challenge.
The researchers also tested the combination in a second model—human CD20 transgenic mice, which express the human CD20 antigen on normal B cells. The team said this model is more akin to the clinical situation.
The mice received treatment 7 days after the inoculation of EL4hCD20 cells. Mice that received obinutuzumab monotherapy had significantly better survival than control mice (P=0.02), but there were no long-term survivors. For mice that received R848 monotherapy, survival was not significantly different from that of controls.
Mice that received R848 in combination with obinutuzumab had significantly better survival than mice that received obinutuzumab alone (P=0.003).
In fact, 6 of the 12 mice that received the combination were long-term survivors. And 5 of these mice rejected tumor re-challenge.
“We were excited when we discovered that combining obinutuzumab with TLR7 activation significantly enhanced survival of animals with lymphoma by effectively eradicating tumors,” Dr Illidge said. “Clearly, more work needs to be done to assess the impact of this combination on humans, but this study is, nevertheless, very promising.”
The researchers said the primary antitumor activity of the combination is dependent on natural killer cells and CD4 helper T cells but not on CD8 killer T cells.
“While the combination therapy was highly effective, CD8 killer T cells did not play a major role in the therapy,” said Eleanor Cheadle, PhD, also of the University of Manchester.
“Given the important role that killer T cells can play in long-term protection from tumor regrowth, we are looking at ways to enhance activation of these cells after obinutuzumab therapy.”
The researchers also found that, in vitro, R848 significantly enhanced natural killer cell-mediated antibody-dependent cellular cytotoxicity against obinutuzumab-opsonized CLL cells and significantly increased non-specific, antibody-independent killing of CLL cells. ![]()

Preclinical research suggests that immune stimulation through Toll-like receptor 7 (TLR7) agonism can enhance the efficacy of obinutuzumab in lymphoma.
Researchers found that combining the anti-CD20 monoclonal antibody obinutuzumab with the TLR7 agonist R848 improved survival in lab mice with lymphoma.
The combination also demonstrated efficacy against chronic lymphocytic leukemia (CLL) cells in vitro.
Tim Illidge, PhD, MBBS, of the University of Manchester in the UK, and his colleagues reported these findings in the journal Leukemia.
The research was funded by the Kay Kendall Leukaemia Fund and Cancer Research UK in collaboration with Roche Pharmaceutical Research and Early Development.
The researchers said they initially found that R848 activates immune cells in vivo and enhances obinutuzumab-mediated antitumor effector mechanisms in vitro.
The team therefore went on to test R848 and obinutuzumab in C57Bl/6 mice bearing human CD20+ lymphoma (EL4). The mice received obinutuzumab modified to express the murine glycoengineered IgG2a Fc region (m2a) starting 1 day after tumor inoculation and systemic R848 once weekly for 4 weeks.
The researchers found that monotherapy with either obinutuzumab or R848 significantly improved survival compared to control (P<0.0001), but only 8% to 15% of mice that received monotherapy were long-term survivors (living more than 90 days).
Mice that received obinutuzumab in combination with R848 had significantly better survival than mice that received either monotherapy (P<0.0001). And about 70% of mice receiving the combination remained tumor-free out to 95 days.
Furthermore, long-term survivors that had received the combination treatment were protected from tumor re-challenge.
The researchers also tested the combination in a second model—human CD20 transgenic mice, which express the human CD20 antigen on normal B cells. The team said this model is more akin to the clinical situation.
The mice received treatment 7 days after the inoculation of EL4hCD20 cells. Mice that received obinutuzumab monotherapy had significantly better survival than control mice (P=0.02), but there were no long-term survivors. For mice that received R848 monotherapy, survival was not significantly different from that of controls.
Mice that received R848 in combination with obinutuzumab had significantly better survival than mice that received obinutuzumab alone (P=0.003).
In fact, 6 of the 12 mice that received the combination were long-term survivors. And 5 of these mice rejected tumor re-challenge.
“We were excited when we discovered that combining obinutuzumab with TLR7 activation significantly enhanced survival of animals with lymphoma by effectively eradicating tumors,” Dr Illidge said. “Clearly, more work needs to be done to assess the impact of this combination on humans, but this study is, nevertheless, very promising.”
The researchers said the primary antitumor activity of the combination is dependent on natural killer cells and CD4 helper T cells but not on CD8 killer T cells.
“While the combination therapy was highly effective, CD8 killer T cells did not play a major role in the therapy,” said Eleanor Cheadle, PhD, also of the University of Manchester.
“Given the important role that killer T cells can play in long-term protection from tumor regrowth, we are looking at ways to enhance activation of these cells after obinutuzumab therapy.”
The researchers also found that, in vitro, R848 significantly enhanced natural killer cell-mediated antibody-dependent cellular cytotoxicity against obinutuzumab-opsonized CLL cells and significantly increased non-specific, antibody-independent killing of CLL cells. ![]()
Overcoming glucocorticoid resistance in lymphoma

Image by Ed Uthman
Targeting RUNX1 could combat glucocorticoid resistance in patients with lymphoma, according to research published in the Journal of Cellular Biochemistry.
Researchers found an over activity of RUNX1 in lymphoma cells interfered with sphingolipids and caused cells to become resistant to dexamethasone.
Dexamethasone works, in part, through the control of sphingolipid enzymes, which play a role in instructing cells to live or die.
Specifically, the researchers said they found that ectopic expression of RUNX1 in lymphoma cells consistently perturbs the sphingolipid rheostat and confers increased resistance to glucocorticoid-mediated apoptosis.
The team also described the mechanism of cross-talk between glucocorticoid and sphingolipid metabolism through the enzyme Sgpp1.
The researchers said dexamethasone induces expression of Sgpp1 in T-lymphoma cells and drives cell death, which is reduced by partial knockdown of Sgpp1 with short hairpin RNA or direct transcriptional repression of Sgpp1 by ectopic RUNX1.
These findings suggest that drugs targeting RUNX1 may be able to reverse glucocorticoid resistance in lymphoma patients.
“The possibility of making existing therapies more active and specific by combining [them] with drugs that inhibit RUNX is a new and exciting prospect,” said study author James Neil, of The University of Glasgow in Scotland.
“Our collaborators in the US have recently developed drugs that inhibit RUNX, and we plan to test these with existing therapies in blood cancers where MYC and RUNX are both implicated, including multiple myeloma and Burkitt lymphoma.”
An earlier study by Dr Neil and his colleagues suggested that RUNX1 was a potential therapeutic target in MYC-driven lymphomas. ![]()

Image by Ed Uthman
Targeting RUNX1 could combat glucocorticoid resistance in patients with lymphoma, according to research published in the Journal of Cellular Biochemistry.
Researchers found an over activity of RUNX1 in lymphoma cells interfered with sphingolipids and caused cells to become resistant to dexamethasone.
Dexamethasone works, in part, through the control of sphingolipid enzymes, which play a role in instructing cells to live or die.
Specifically, the researchers said they found that ectopic expression of RUNX1 in lymphoma cells consistently perturbs the sphingolipid rheostat and confers increased resistance to glucocorticoid-mediated apoptosis.
The team also described the mechanism of cross-talk between glucocorticoid and sphingolipid metabolism through the enzyme Sgpp1.
The researchers said dexamethasone induces expression of Sgpp1 in T-lymphoma cells and drives cell death, which is reduced by partial knockdown of Sgpp1 with short hairpin RNA or direct transcriptional repression of Sgpp1 by ectopic RUNX1.
These findings suggest that drugs targeting RUNX1 may be able to reverse glucocorticoid resistance in lymphoma patients.
“The possibility of making existing therapies more active and specific by combining [them] with drugs that inhibit RUNX is a new and exciting prospect,” said study author James Neil, of The University of Glasgow in Scotland.
“Our collaborators in the US have recently developed drugs that inhibit RUNX, and we plan to test these with existing therapies in blood cancers where MYC and RUNX are both implicated, including multiple myeloma and Burkitt lymphoma.”
An earlier study by Dr Neil and his colleagues suggested that RUNX1 was a potential therapeutic target in MYC-driven lymphomas. ![]()

Image by Ed Uthman
Targeting RUNX1 could combat glucocorticoid resistance in patients with lymphoma, according to research published in the Journal of Cellular Biochemistry.
Researchers found an over activity of RUNX1 in lymphoma cells interfered with sphingolipids and caused cells to become resistant to dexamethasone.
Dexamethasone works, in part, through the control of sphingolipid enzymes, which play a role in instructing cells to live or die.
Specifically, the researchers said they found that ectopic expression of RUNX1 in lymphoma cells consistently perturbs the sphingolipid rheostat and confers increased resistance to glucocorticoid-mediated apoptosis.
The team also described the mechanism of cross-talk between glucocorticoid and sphingolipid metabolism through the enzyme Sgpp1.
The researchers said dexamethasone induces expression of Sgpp1 in T-lymphoma cells and drives cell death, which is reduced by partial knockdown of Sgpp1 with short hairpin RNA or direct transcriptional repression of Sgpp1 by ectopic RUNX1.
These findings suggest that drugs targeting RUNX1 may be able to reverse glucocorticoid resistance in lymphoma patients.
“The possibility of making existing therapies more active and specific by combining [them] with drugs that inhibit RUNX is a new and exciting prospect,” said study author James Neil, of The University of Glasgow in Scotland.
“Our collaborators in the US have recently developed drugs that inhibit RUNX, and we plan to test these with existing therapies in blood cancers where MYC and RUNX are both implicated, including multiple myeloma and Burkitt lymphoma.”
An earlier study by Dr Neil and his colleagues suggested that RUNX1 was a potential therapeutic target in MYC-driven lymphomas. ![]()
FDA approves ibrutinib to treat rel/ref MZL

Photo courtesy of
Janssen Biotech, Inc.
The US Food and Drug Administration (FDA) has approved the Bruton’s tyrosine kinase inhibitor ibrutinib (Imbruvica®) for the treatment of marginal zone lymphoma (MZL).
The drug is now approved to treat patients with relapsed/refractory MZL who require systemic therapy and have received at least 1 prior anti-CD20-based therapy.
Ibrutinib has accelerated approval for this indication, based on the overall response rate the drug produced in a phase 2 trial.
Continued approval of ibrutinib as a treatment for MZL may be contingent upon verification and description of clinical benefit in a confirmatory trial.
The FDA’s approval of ibrutinib for MZL makes it the first treatment approved specifically for patients with this disease. It also marks the seventh FDA approval and fifth disease indication for ibrutinib since the drug was first approved in 2013.
Ibrutinib is also FDA-approved to treat chronic lymphocytic leukemia/small lymphocytic lymphoma, patients with mantle cell lymphoma who have received at least 1 prior therapy, and patients with Waldenström’s macroglobulinemia. The approval for mantle cell lymphoma is an accelerated approval.
Ibrutinib is jointly developed and commercialized by Pharmacyclics LLC, an AbbVie company, and Janssen Biotech, Inc.
Phase 2 trial
The FDA’s approval of ibrutinib for MZL is based on data from the phase 2, single-arm PCYC-1121 study, in which researchers evaluated the drug in MZL patients who required systemic therapy and had received at least 1 prior anti-CD20-based therapy.
Results from this study were presented at the 2016 ASH Annual Meeting (abstract 1213).
The efficacy analysis included 63 patients with 3 subtypes of MZL: mucosa-associated lymphoid tissue (n=32), nodal (n=17), and splenic (n=14).
The overall response rate was 46%, with a partial response rate of 42.9% and a complete response rate of 3.2%. Responses were observed across all 3 MZL subtypes.
The median time to response was 4.5 months (range, 2.3-16.4 months). And the median duration of response was not reached (range, 16.7 months to not reached).
Overall, the safety data from this study was consistent with the known safety profile of ibrutinib in B-cell malignancies.
The most common adverse events of all grades (occurring in >20% of patients) were thrombocytopenia (49%), fatigue (44%), anemia (43%), diarrhea (43%), bruising (41%), musculoskeletal pain (40%), hemorrhage (30%), rash (29%), nausea (25%), peripheral edema (24%), arthralgia (24%), neutropenia (22%), cough (22%), dyspnea (21%), and upper respiratory tract infection (21%).
The most common (>10%) grade 3 or 4 events were decreases in hemoglobin and neutrophils (13% each) and pneumonia (10%).
The risks associated with ibrutinib as listed in the Warnings and Precautions section of the prescribing information are hemorrhage, infections, cytopenias, atrial fibrillation, hypertension, secondary primary malignancies, tumor lysis syndrome, and embryo fetal toxicities. ![]()

Photo courtesy of
Janssen Biotech, Inc.
The US Food and Drug Administration (FDA) has approved the Bruton’s tyrosine kinase inhibitor ibrutinib (Imbruvica®) for the treatment of marginal zone lymphoma (MZL).
The drug is now approved to treat patients with relapsed/refractory MZL who require systemic therapy and have received at least 1 prior anti-CD20-based therapy.
Ibrutinib has accelerated approval for this indication, based on the overall response rate the drug produced in a phase 2 trial.
Continued approval of ibrutinib as a treatment for MZL may be contingent upon verification and description of clinical benefit in a confirmatory trial.
The FDA’s approval of ibrutinib for MZL makes it the first treatment approved specifically for patients with this disease. It also marks the seventh FDA approval and fifth disease indication for ibrutinib since the drug was first approved in 2013.
Ibrutinib is also FDA-approved to treat chronic lymphocytic leukemia/small lymphocytic lymphoma, patients with mantle cell lymphoma who have received at least 1 prior therapy, and patients with Waldenström’s macroglobulinemia. The approval for mantle cell lymphoma is an accelerated approval.
Ibrutinib is jointly developed and commercialized by Pharmacyclics LLC, an AbbVie company, and Janssen Biotech, Inc.
Phase 2 trial
The FDA’s approval of ibrutinib for MZL is based on data from the phase 2, single-arm PCYC-1121 study, in which researchers evaluated the drug in MZL patients who required systemic therapy and had received at least 1 prior anti-CD20-based therapy.
Results from this study were presented at the 2016 ASH Annual Meeting (abstract 1213).
The efficacy analysis included 63 patients with 3 subtypes of MZL: mucosa-associated lymphoid tissue (n=32), nodal (n=17), and splenic (n=14).
The overall response rate was 46%, with a partial response rate of 42.9% and a complete response rate of 3.2%. Responses were observed across all 3 MZL subtypes.
The median time to response was 4.5 months (range, 2.3-16.4 months). And the median duration of response was not reached (range, 16.7 months to not reached).
Overall, the safety data from this study was consistent with the known safety profile of ibrutinib in B-cell malignancies.
The most common adverse events of all grades (occurring in >20% of patients) were thrombocytopenia (49%), fatigue (44%), anemia (43%), diarrhea (43%), bruising (41%), musculoskeletal pain (40%), hemorrhage (30%), rash (29%), nausea (25%), peripheral edema (24%), arthralgia (24%), neutropenia (22%), cough (22%), dyspnea (21%), and upper respiratory tract infection (21%).
The most common (>10%) grade 3 or 4 events were decreases in hemoglobin and neutrophils (13% each) and pneumonia (10%).
The risks associated with ibrutinib as listed in the Warnings and Precautions section of the prescribing information are hemorrhage, infections, cytopenias, atrial fibrillation, hypertension, secondary primary malignancies, tumor lysis syndrome, and embryo fetal toxicities. ![]()

Photo courtesy of
Janssen Biotech, Inc.
The US Food and Drug Administration (FDA) has approved the Bruton’s tyrosine kinase inhibitor ibrutinib (Imbruvica®) for the treatment of marginal zone lymphoma (MZL).
The drug is now approved to treat patients with relapsed/refractory MZL who require systemic therapy and have received at least 1 prior anti-CD20-based therapy.
Ibrutinib has accelerated approval for this indication, based on the overall response rate the drug produced in a phase 2 trial.
Continued approval of ibrutinib as a treatment for MZL may be contingent upon verification and description of clinical benefit in a confirmatory trial.
The FDA’s approval of ibrutinib for MZL makes it the first treatment approved specifically for patients with this disease. It also marks the seventh FDA approval and fifth disease indication for ibrutinib since the drug was first approved in 2013.
Ibrutinib is also FDA-approved to treat chronic lymphocytic leukemia/small lymphocytic lymphoma, patients with mantle cell lymphoma who have received at least 1 prior therapy, and patients with Waldenström’s macroglobulinemia. The approval for mantle cell lymphoma is an accelerated approval.
Ibrutinib is jointly developed and commercialized by Pharmacyclics LLC, an AbbVie company, and Janssen Biotech, Inc.
Phase 2 trial
The FDA’s approval of ibrutinib for MZL is based on data from the phase 2, single-arm PCYC-1121 study, in which researchers evaluated the drug in MZL patients who required systemic therapy and had received at least 1 prior anti-CD20-based therapy.
Results from this study were presented at the 2016 ASH Annual Meeting (abstract 1213).
The efficacy analysis included 63 patients with 3 subtypes of MZL: mucosa-associated lymphoid tissue (n=32), nodal (n=17), and splenic (n=14).
The overall response rate was 46%, with a partial response rate of 42.9% and a complete response rate of 3.2%. Responses were observed across all 3 MZL subtypes.
The median time to response was 4.5 months (range, 2.3-16.4 months). And the median duration of response was not reached (range, 16.7 months to not reached).
Overall, the safety data from this study was consistent with the known safety profile of ibrutinib in B-cell malignancies.
The most common adverse events of all grades (occurring in >20% of patients) were thrombocytopenia (49%), fatigue (44%), anemia (43%), diarrhea (43%), bruising (41%), musculoskeletal pain (40%), hemorrhage (30%), rash (29%), nausea (25%), peripheral edema (24%), arthralgia (24%), neutropenia (22%), cough (22%), dyspnea (21%), and upper respiratory tract infection (21%).
The most common (>10%) grade 3 or 4 events were decreases in hemoglobin and neutrophils (13% each) and pneumonia (10%).
The risks associated with ibrutinib as listed in the Warnings and Precautions section of the prescribing information are hemorrhage, infections, cytopenias, atrial fibrillation, hypertension, secondary primary malignancies, tumor lysis syndrome, and embryo fetal toxicities. ![]()
VIDEO: First multicenter trial of CAR T cells shows response in DLBCL
SAN DIEGO – Aggressive, refractory non-Hodgkin lymphomas responded to anti-CD19 chimeric antigen receptor T cells in ZUMA-1, the first multicenter trial of the cellular immunotherapy, based on early data reported at the annual meeting of the American Society of Hematology.
In an interim analysis of 51 patients with diffuse large B-cell lymphomas, 47% had complete remissions and 29% had partial remissions. But the remission rate declined to 33% complete remissions and 6% partial remissions after 3 months.
There have really been no new treatments in the last 20 years for patients with non-Hodgkin lymphoma that does not respond to chemotherapy or recurs after autologous stem cell transplant. With median overall survival of 6 months, and about 8% complete remissions with existing therapies, CAR T cells might be a solution for these patients, said ZUMA-1 investigator Sattva S. Neelapu, MD, of the University of Texas MD Anderson Cancer Center in Houston.
In our video interview at the meeting, Dr. Neelapu discussed initial results in the real-world setting of 22 participating centers, most of which had no previous experience with CAR T-cell therapy. With an efficient production and logistics plan, 91% of 110 patients were able to receive the investigational product, known as KTE-C19.
ZUMA-1 is funded by Kite, which makes KTE-C19, and the Leukemia & Lymphoma Society Therapy Acceleration Program. Dr. Neelapu receives research support from and is an advisor to Kite.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
[email protected]
On Twitter @maryjodales
SAN DIEGO – Aggressive, refractory non-Hodgkin lymphomas responded to anti-CD19 chimeric antigen receptor T cells in ZUMA-1, the first multicenter trial of the cellular immunotherapy, based on early data reported at the annual meeting of the American Society of Hematology.
In an interim analysis of 51 patients with diffuse large B-cell lymphomas, 47% had complete remissions and 29% had partial remissions. But the remission rate declined to 33% complete remissions and 6% partial remissions after 3 months.
There have really been no new treatments in the last 20 years for patients with non-Hodgkin lymphoma that does not respond to chemotherapy or recurs after autologous stem cell transplant. With median overall survival of 6 months, and about 8% complete remissions with existing therapies, CAR T cells might be a solution for these patients, said ZUMA-1 investigator Sattva S. Neelapu, MD, of the University of Texas MD Anderson Cancer Center in Houston.
In our video interview at the meeting, Dr. Neelapu discussed initial results in the real-world setting of 22 participating centers, most of which had no previous experience with CAR T-cell therapy. With an efficient production and logistics plan, 91% of 110 patients were able to receive the investigational product, known as KTE-C19.
ZUMA-1 is funded by Kite, which makes KTE-C19, and the Leukemia & Lymphoma Society Therapy Acceleration Program. Dr. Neelapu receives research support from and is an advisor to Kite.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
[email protected]
On Twitter @maryjodales
SAN DIEGO – Aggressive, refractory non-Hodgkin lymphomas responded to anti-CD19 chimeric antigen receptor T cells in ZUMA-1, the first multicenter trial of the cellular immunotherapy, based on early data reported at the annual meeting of the American Society of Hematology.
In an interim analysis of 51 patients with diffuse large B-cell lymphomas, 47% had complete remissions and 29% had partial remissions. But the remission rate declined to 33% complete remissions and 6% partial remissions after 3 months.
There have really been no new treatments in the last 20 years for patients with non-Hodgkin lymphoma that does not respond to chemotherapy or recurs after autologous stem cell transplant. With median overall survival of 6 months, and about 8% complete remissions with existing therapies, CAR T cells might be a solution for these patients, said ZUMA-1 investigator Sattva S. Neelapu, MD, of the University of Texas MD Anderson Cancer Center in Houston.
In our video interview at the meeting, Dr. Neelapu discussed initial results in the real-world setting of 22 participating centers, most of which had no previous experience with CAR T-cell therapy. With an efficient production and logistics plan, 91% of 110 patients were able to receive the investigational product, known as KTE-C19.
ZUMA-1 is funded by Kite, which makes KTE-C19, and the Leukemia & Lymphoma Society Therapy Acceleration Program. Dr. Neelapu receives research support from and is an advisor to Kite.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
[email protected]
On Twitter @maryjodales
Prolonged work-related stress linked to NHL, other cancers in men

burning building in Quebec
Photo by Sylvain Pedneault
New research suggests that prolonged exposure to work-related stress may increase a man’s risk of several cancers.
The study showed a significant association between work-related stress lasting 15 years or more and non-Hodgkin lymphoma (NHL) as well as lung, colon, rectal, and stomach cancers.
Men who had worked as firefighters, engineers, mechanics, and repair workers were most likely to report work-related stress.
Marie-Élise Parent, PhD, of Institut national de la recherche scientifique (INRS) in Laval, Quebec, Canada, and her colleagues conducted this research and published
the results in Preventive Medicine.
The researchers studied 3103 men with 11 different types of cancer who were diagnosed from 1979 to 1985. The team compared these men to 512 control subjects from the general population.
Both cases and controls were interviewed and asked to describe each job they had during their lifetime, including the occurrence of stress related to a job and the reason for that stress.
The researchers then calculated odds ratios (OR) for the association between perceived workplace stress and its duration, and each cancer site. The analyses were adjusted for lifestyle and occupational factors.
The team found that having at least one stressful job in a lifetime was associated with increased odds of 5 cancers:
- Lung—OR=1.33
- Colon—OR=1.51
- Bladder—OR=1.37
- Rectal—OR=1.52
- Stomach—OR=1.53.
When the researchers looked at the duration of stress, they found no significant association between any of the cancers and work-related stress lasting less than 15 years.
However, there were significant associations for several cancers and work-related stress lasting 15 to 30 years or more than 30 years. These included:
- NHL—15-30 years, OR=1.47; >30 years, OR=1.69 (P=0.02)
- Lung cancer—15-30 years, OR=1.47; >30 years, OR=1.51 (P=0.01)
- Colon cancer—15-30 years, OR=1.32; >30 years, OR=1.64 (P<0.01)
- Rectal cancer—15-30 years, OR=1.84; >30 years, OR=1.48 (P=0.01)
- Stomach cancer—15-30 years, OR=2.15; >30 years, OR=1.48 (P=0.01).
The occupations with the highest prevalence of work-related stress were firefighter (40% of firefighting jobs reported as stressful), industrial and aerospace engineer (31%), and motor vehicle and rail transport mechanic/repair worker (28%).
For the same individual, stress varied depending on the job held.
The study also showed that perceived stress was not limited to high work load and time constraints. Customer service, sales commissions, responsibilities, having an anxious temperament, job insecurity, financial problems, challenging or dangerous work conditions, employee supervision, interpersonal conflict, and a difficult commute were all sources of stress listed by study participants.
The researchers said one of the biggest flaws in previous studies of this kind is that none of them assessed work-related stress over a full working lifetime. The team said this made it impossible to determine how the duration of exposure to work-related stress affects cancer development.
This study, on the other hand, shows the importance of measuring stress at different points in an individual’s working life, the researchers said. They added that their results raise the question of whether chronic psychological stress should be viewed as a public health issue.
However, the team also pointed out that these results are unsubstantiated because they are based on a summary assessment of work-related stress for a given job. There is a need for epidemiological studies based on reliable stress measurements, repeated over time, and that take all sources of stress into account. ![]()

burning building in Quebec
Photo by Sylvain Pedneault
New research suggests that prolonged exposure to work-related stress may increase a man’s risk of several cancers.
The study showed a significant association between work-related stress lasting 15 years or more and non-Hodgkin lymphoma (NHL) as well as lung, colon, rectal, and stomach cancers.
Men who had worked as firefighters, engineers, mechanics, and repair workers were most likely to report work-related stress.
Marie-Élise Parent, PhD, of Institut national de la recherche scientifique (INRS) in Laval, Quebec, Canada, and her colleagues conducted this research and published
the results in Preventive Medicine.
The researchers studied 3103 men with 11 different types of cancer who were diagnosed from 1979 to 1985. The team compared these men to 512 control subjects from the general population.
Both cases and controls were interviewed and asked to describe each job they had during their lifetime, including the occurrence of stress related to a job and the reason for that stress.
The researchers then calculated odds ratios (OR) for the association between perceived workplace stress and its duration, and each cancer site. The analyses were adjusted for lifestyle and occupational factors.
The team found that having at least one stressful job in a lifetime was associated with increased odds of 5 cancers:
- Lung—OR=1.33
- Colon—OR=1.51
- Bladder—OR=1.37
- Rectal—OR=1.52
- Stomach—OR=1.53.
When the researchers looked at the duration of stress, they found no significant association between any of the cancers and work-related stress lasting less than 15 years.
However, there were significant associations for several cancers and work-related stress lasting 15 to 30 years or more than 30 years. These included:
- NHL—15-30 years, OR=1.47; >30 years, OR=1.69 (P=0.02)
- Lung cancer—15-30 years, OR=1.47; >30 years, OR=1.51 (P=0.01)
- Colon cancer—15-30 years, OR=1.32; >30 years, OR=1.64 (P<0.01)
- Rectal cancer—15-30 years, OR=1.84; >30 years, OR=1.48 (P=0.01)
- Stomach cancer—15-30 years, OR=2.15; >30 years, OR=1.48 (P=0.01).
The occupations with the highest prevalence of work-related stress were firefighter (40% of firefighting jobs reported as stressful), industrial and aerospace engineer (31%), and motor vehicle and rail transport mechanic/repair worker (28%).
For the same individual, stress varied depending on the job held.
The study also showed that perceived stress was not limited to high work load and time constraints. Customer service, sales commissions, responsibilities, having an anxious temperament, job insecurity, financial problems, challenging or dangerous work conditions, employee supervision, interpersonal conflict, and a difficult commute were all sources of stress listed by study participants.
The researchers said one of the biggest flaws in previous studies of this kind is that none of them assessed work-related stress over a full working lifetime. The team said this made it impossible to determine how the duration of exposure to work-related stress affects cancer development.
This study, on the other hand, shows the importance of measuring stress at different points in an individual’s working life, the researchers said. They added that their results raise the question of whether chronic psychological stress should be viewed as a public health issue.
However, the team also pointed out that these results are unsubstantiated because they are based on a summary assessment of work-related stress for a given job. There is a need for epidemiological studies based on reliable stress measurements, repeated over time, and that take all sources of stress into account. ![]()

burning building in Quebec
Photo by Sylvain Pedneault
New research suggests that prolonged exposure to work-related stress may increase a man’s risk of several cancers.
The study showed a significant association between work-related stress lasting 15 years or more and non-Hodgkin lymphoma (NHL) as well as lung, colon, rectal, and stomach cancers.
Men who had worked as firefighters, engineers, mechanics, and repair workers were most likely to report work-related stress.
Marie-Élise Parent, PhD, of Institut national de la recherche scientifique (INRS) in Laval, Quebec, Canada, and her colleagues conducted this research and published
the results in Preventive Medicine.
The researchers studied 3103 men with 11 different types of cancer who were diagnosed from 1979 to 1985. The team compared these men to 512 control subjects from the general population.
Both cases and controls were interviewed and asked to describe each job they had during their lifetime, including the occurrence of stress related to a job and the reason for that stress.
The researchers then calculated odds ratios (OR) for the association between perceived workplace stress and its duration, and each cancer site. The analyses were adjusted for lifestyle and occupational factors.
The team found that having at least one stressful job in a lifetime was associated with increased odds of 5 cancers:
- Lung—OR=1.33
- Colon—OR=1.51
- Bladder—OR=1.37
- Rectal—OR=1.52
- Stomach—OR=1.53.
When the researchers looked at the duration of stress, they found no significant association between any of the cancers and work-related stress lasting less than 15 years.
However, there were significant associations for several cancers and work-related stress lasting 15 to 30 years or more than 30 years. These included:
- NHL—15-30 years, OR=1.47; >30 years, OR=1.69 (P=0.02)
- Lung cancer—15-30 years, OR=1.47; >30 years, OR=1.51 (P=0.01)
- Colon cancer—15-30 years, OR=1.32; >30 years, OR=1.64 (P<0.01)
- Rectal cancer—15-30 years, OR=1.84; >30 years, OR=1.48 (P=0.01)
- Stomach cancer—15-30 years, OR=2.15; >30 years, OR=1.48 (P=0.01).
The occupations with the highest prevalence of work-related stress were firefighter (40% of firefighting jobs reported as stressful), industrial and aerospace engineer (31%), and motor vehicle and rail transport mechanic/repair worker (28%).
For the same individual, stress varied depending on the job held.
The study also showed that perceived stress was not limited to high work load and time constraints. Customer service, sales commissions, responsibilities, having an anxious temperament, job insecurity, financial problems, challenging or dangerous work conditions, employee supervision, interpersonal conflict, and a difficult commute were all sources of stress listed by study participants.
The researchers said one of the biggest flaws in previous studies of this kind is that none of them assessed work-related stress over a full working lifetime. The team said this made it impossible to determine how the duration of exposure to work-related stress affects cancer development.
This study, on the other hand, shows the importance of measuring stress at different points in an individual’s working life, the researchers said. They added that their results raise the question of whether chronic psychological stress should be viewed as a public health issue.
However, the team also pointed out that these results are unsubstantiated because they are based on a summary assessment of work-related stress for a given job. There is a need for epidemiological studies based on reliable stress measurements, repeated over time, and that take all sources of stress into account. ![]()
Venetoclax approved to treat CLL in Australia

venetoclax (US version)
Photo courtesy of Abbvie
The Australian Therapeutic Goods Administration (TGA) has approved the BCL-2 inhibitor venetoclax (Venclexta™, formerly ABT-199) for use in certain patients with chronic lymphocytic leukemia (CLL).
The drug is now approved to treat Australian patients with relapsed or refractory CLL who have 17p deletion or no other treatment options.
Venetoclax is being developed by AbbVie and Genentech, a member of the Roche Group. The drug is jointly commercialized by the companies in the US and by AbbVie outside of the US.
Now that venetoclax has been approved by the TGA, it can be registered on the Australian Register of Therapeutic Goods and legally marketed and sold in Australia.
To make the drug affordable to the Australian public, the manufacturer can apply to the Pharmaceutical Benefits Advisory Committee to have the cost of the drug subsidized by the Australian government on the Pharmaceutical Benefits Scheme (PBS).
Venetoclax is not listed on the PBS. Historically, the delay between TGA approval and PBS listing ranges from 14 months to 31 months for cancer drugs.
Phase 2 trials
Venetoclax has produced high objective response rates (ORR) in two phase 2 trials of CLL patients.
In one of these trials, researchers tested venetoclax in 107 patients with previously treated CLL and 17p deletion. The results were published in The Lancet Oncology in June 2016.
The ORR in this trial was 79%. At the time of analysis, the median duration of response had not been reached. The same was true for progression-free survival and overall survival.
The progression-free survival estimate for 12 months was 72%, and the overall survival estimate was 87%.
The incidence of treatment-emergent adverse events was 96%, and the incidence of serious adverse events was 55%.
Grade 3 laboratory tumor lysis syndrome (TLS) was reported in 5 patients. Three of these patients continued on venetoclax, but 2 patients required a dose interruption of 1 day each.
In the second trial, researchers tested venetoclax in 64 patients with CLL who had failed treatment with ibrutinib and/or idelalisib. Results from this trial were presented at the 2016 ASH Annual Meeting.
The ORR was 67%. At 11.8 months of follow-up, the median duration of response, progression-free survival, and overall survival had not been reached. The estimated 12-month progression-free survival was 80%.
The incidence of adverse events was 100%, and the incidence of serious adverse events was 53%. No clinical TLS was observed, but 1 patient met Howard criteria for laboratory TLS.
In the past, TLS has caused deaths in patients receiving venetoclax. In response, AbbVie stopped dose-escalation in patients receiving the drug and suspended enrollment in phase 1 trials.
However, researchers subsequently found that a modified dosing schedule, prophylaxis, and patient monitoring can reduce the risk of TLS. ![]()

venetoclax (US version)
Photo courtesy of Abbvie
The Australian Therapeutic Goods Administration (TGA) has approved the BCL-2 inhibitor venetoclax (Venclexta™, formerly ABT-199) for use in certain patients with chronic lymphocytic leukemia (CLL).
The drug is now approved to treat Australian patients with relapsed or refractory CLL who have 17p deletion or no other treatment options.
Venetoclax is being developed by AbbVie and Genentech, a member of the Roche Group. The drug is jointly commercialized by the companies in the US and by AbbVie outside of the US.
Now that venetoclax has been approved by the TGA, it can be registered on the Australian Register of Therapeutic Goods and legally marketed and sold in Australia.
To make the drug affordable to the Australian public, the manufacturer can apply to the Pharmaceutical Benefits Advisory Committee to have the cost of the drug subsidized by the Australian government on the Pharmaceutical Benefits Scheme (PBS).
Venetoclax is not listed on the PBS. Historically, the delay between TGA approval and PBS listing ranges from 14 months to 31 months for cancer drugs.
Phase 2 trials
Venetoclax has produced high objective response rates (ORR) in two phase 2 trials of CLL patients.
In one of these trials, researchers tested venetoclax in 107 patients with previously treated CLL and 17p deletion. The results were published in The Lancet Oncology in June 2016.
The ORR in this trial was 79%. At the time of analysis, the median duration of response had not been reached. The same was true for progression-free survival and overall survival.
The progression-free survival estimate for 12 months was 72%, and the overall survival estimate was 87%.
The incidence of treatment-emergent adverse events was 96%, and the incidence of serious adverse events was 55%.
Grade 3 laboratory tumor lysis syndrome (TLS) was reported in 5 patients. Three of these patients continued on venetoclax, but 2 patients required a dose interruption of 1 day each.
In the second trial, researchers tested venetoclax in 64 patients with CLL who had failed treatment with ibrutinib and/or idelalisib. Results from this trial were presented at the 2016 ASH Annual Meeting.
The ORR was 67%. At 11.8 months of follow-up, the median duration of response, progression-free survival, and overall survival had not been reached. The estimated 12-month progression-free survival was 80%.
The incidence of adverse events was 100%, and the incidence of serious adverse events was 53%. No clinical TLS was observed, but 1 patient met Howard criteria for laboratory TLS.
In the past, TLS has caused deaths in patients receiving venetoclax. In response, AbbVie stopped dose-escalation in patients receiving the drug and suspended enrollment in phase 1 trials.
However, researchers subsequently found that a modified dosing schedule, prophylaxis, and patient monitoring can reduce the risk of TLS. ![]()

venetoclax (US version)
Photo courtesy of Abbvie
The Australian Therapeutic Goods Administration (TGA) has approved the BCL-2 inhibitor venetoclax (Venclexta™, formerly ABT-199) for use in certain patients with chronic lymphocytic leukemia (CLL).
The drug is now approved to treat Australian patients with relapsed or refractory CLL who have 17p deletion or no other treatment options.
Venetoclax is being developed by AbbVie and Genentech, a member of the Roche Group. The drug is jointly commercialized by the companies in the US and by AbbVie outside of the US.
Now that venetoclax has been approved by the TGA, it can be registered on the Australian Register of Therapeutic Goods and legally marketed and sold in Australia.
To make the drug affordable to the Australian public, the manufacturer can apply to the Pharmaceutical Benefits Advisory Committee to have the cost of the drug subsidized by the Australian government on the Pharmaceutical Benefits Scheme (PBS).
Venetoclax is not listed on the PBS. Historically, the delay between TGA approval and PBS listing ranges from 14 months to 31 months for cancer drugs.
Phase 2 trials
Venetoclax has produced high objective response rates (ORR) in two phase 2 trials of CLL patients.
In one of these trials, researchers tested venetoclax in 107 patients with previously treated CLL and 17p deletion. The results were published in The Lancet Oncology in June 2016.
The ORR in this trial was 79%. At the time of analysis, the median duration of response had not been reached. The same was true for progression-free survival and overall survival.
The progression-free survival estimate for 12 months was 72%, and the overall survival estimate was 87%.
The incidence of treatment-emergent adverse events was 96%, and the incidence of serious adverse events was 55%.
Grade 3 laboratory tumor lysis syndrome (TLS) was reported in 5 patients. Three of these patients continued on venetoclax, but 2 patients required a dose interruption of 1 day each.
In the second trial, researchers tested venetoclax in 64 patients with CLL who had failed treatment with ibrutinib and/or idelalisib. Results from this trial were presented at the 2016 ASH Annual Meeting.
The ORR was 67%. At 11.8 months of follow-up, the median duration of response, progression-free survival, and overall survival had not been reached. The estimated 12-month progression-free survival was 80%.
The incidence of adverse events was 100%, and the incidence of serious adverse events was 53%. No clinical TLS was observed, but 1 patient met Howard criteria for laboratory TLS.
In the past, TLS has caused deaths in patients receiving venetoclax. In response, AbbVie stopped dose-escalation in patients receiving the drug and suspended enrollment in phase 1 trials.
However, researchers subsequently found that a modified dosing schedule, prophylaxis, and patient monitoring can reduce the risk of TLS.
