User login
Adding lymphopenia component ‘improves’ FLIPI
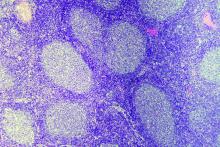
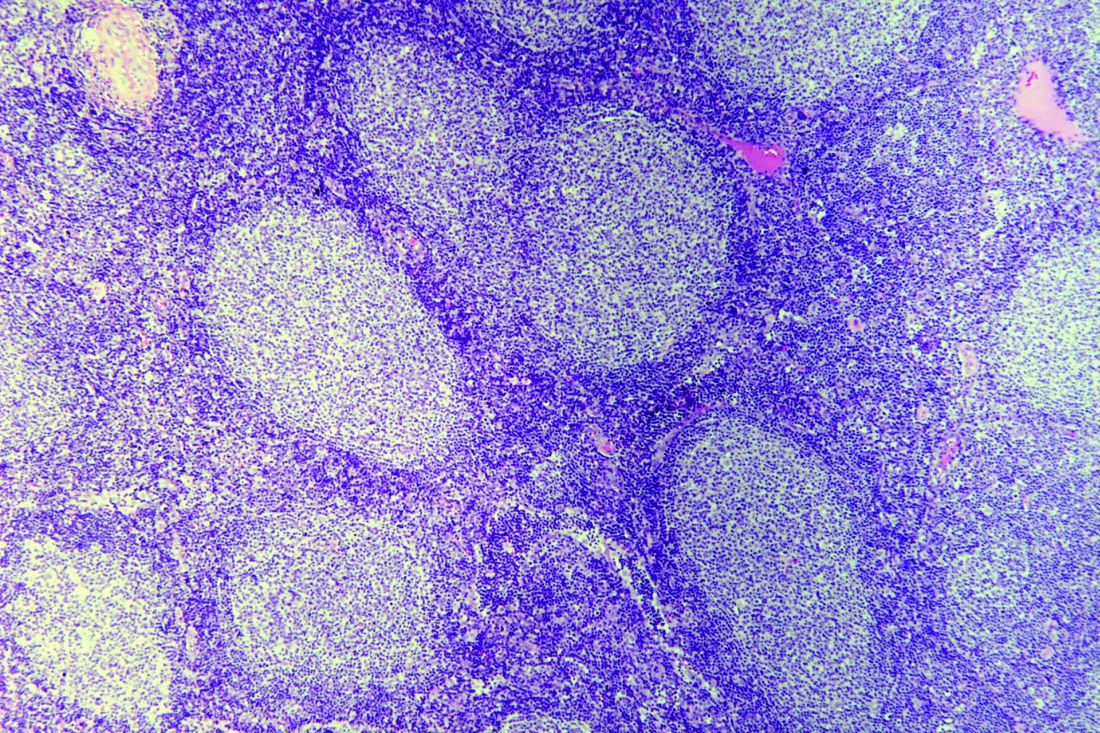
Incorporating lymphopenia into the Follicular Lymphoma International Prognostic Index (FLIPI) can improve prognostication, according to researchers.
The team added lymphopenia as a point in a revised FLIPI scoring system, called FLIPI-L, and found the new system could better predict overall survival (OS), progression-free survival, and histologic transformation in patients with follicular lymphoma.
George Yang, MD, of Moffitt Cancer Center in Tampa, Fla., and his colleagues described results with the FLIPI-L in a letter published in Blood Cancer Journal.
“Prior studies have demonstrated that lymphopenia was associated with worsened OS in [follicular lymphoma],” Dr. Yang and his colleagues wrote. “Therefore, we hypothesized that lymphopenia may be integrated with existing FLIPI to better stratify long-term survival outcomes and predict for transformation.”
The researchers tested this theory in 736 follicular lymphoma patients who were followed for a median of 72 months (range, 2-211 months). The 5-year OS in this cohort was 81.3%, the 10-year OS was 67.3%, and 18% of patients experienced transformation to high-grade lymphoma.
The researchers defined absolute lymphopenia as less than 1.0 × 109 lymphocytes per liter. In multivariate analyses, lymphopenia was an independent predictor of OS (hazard ratio, 1.74; P less than .01) and transformation (odds ratio, 2.1; P less than .01).
To incorporate lymphopenia into the FLIPI, the researchers created a model in which 1 point was given for each of the standard FLIPI components (age, Ann Arbor stage, number of nodal areas, lactate dehydrogenase, and hemoglobin level), and one point was given for the presence of lymphopenia. Patients in the low-risk FLIPI-L category had 0-1 points, those in the intermediate-risk category had 2-3 points, and patients in the high-risk FLIPI-L category had 4-6 points.
Using the original FLIPI, the 5-year OS was 91% in the low-risk group (0-1), 82.7% in the intermediate-risk group (2), and 66% in the high-risk group (3-5). The 10-year OS was 80.4%, 66%, and 45.8%, respectively.
Using the FLIPI-L, the 5-year OS was 94.5% in the low-risk group (0-1), 89% in the intermediate-risk group (2-3), and 61% in the high-risk group (4-6). The 10-year OS was 83.9%, 68.5%, and 34.5%, respectively.
In a univariate Cox regression analysis of OS, each point increase in FLIPI-L score was associated with a significant increase in hazard ratio. For example, the hazard ratio was 3.4 for patients with a FLIPI-L score of 1 and 30.9 for those with a FLIPI-L score of 6 (P less than .02 for all FLIPI-L scores). Conversely, increases in hazard ratio were not significant with the original FLIPI (P greater than .05 for all FLIPI scores).
The FLIPI-L was prognostic for OS in different treatment groups. In patients who received rituximab alone, radiation alone, or rituximab plus chemotherapy, the scoring system differentiated low-, intermediate-, and high-risk groups (P less than .04). In patients under observation, the FLIPI-L distinguished low/intermediate-risk and high-risk groups (P less than .01).
For patients who progressed within 24 months, the FLIPI-L was more predictive of progression-free survival (P = .05) than was the original FLIPI (P = .11).
Increasing FLIPI-L was an independent predictor of transformation, both when assessed as a continuous variable (P less than .01) and stepwise for FLIPI-L 3-5 (P = .004-.01). The original FLIPI, on the other hand, was not an independent predictor of transformation.
“Our analysis of a lymphopenia cutoff as an addition to the original FLIPI is simple yet improves risk stratification to differentiate between prognostic groups and, importantly, to predict transformation,” Dr. Yang and his colleagues wrote.
The authors reported having no conflicts of interest.
SOURCE: Yang G et al. Blood Cancer J. 2020 Jan 2;9(12):104. doi: 10.1038/s41408-019-0269-6.
Incorporating lymphopenia into the Follicular Lymphoma International Prognostic Index (FLIPI) can improve prognostication, according to researchers.
The team added lymphopenia as a point in a revised FLIPI scoring system, called FLIPI-L, and found the new system could better predict overall survival (OS), progression-free survival, and histologic transformation in patients with follicular lymphoma.
George Yang, MD, of Moffitt Cancer Center in Tampa, Fla., and his colleagues described results with the FLIPI-L in a letter published in Blood Cancer Journal.
“Prior studies have demonstrated that lymphopenia was associated with worsened OS in [follicular lymphoma],” Dr. Yang and his colleagues wrote. “Therefore, we hypothesized that lymphopenia may be integrated with existing FLIPI to better stratify long-term survival outcomes and predict for transformation.”
The researchers tested this theory in 736 follicular lymphoma patients who were followed for a median of 72 months (range, 2-211 months). The 5-year OS in this cohort was 81.3%, the 10-year OS was 67.3%, and 18% of patients experienced transformation to high-grade lymphoma.
The researchers defined absolute lymphopenia as less than 1.0 × 109 lymphocytes per liter. In multivariate analyses, lymphopenia was an independent predictor of OS (hazard ratio, 1.74; P less than .01) and transformation (odds ratio, 2.1; P less than .01).
To incorporate lymphopenia into the FLIPI, the researchers created a model in which 1 point was given for each of the standard FLIPI components (age, Ann Arbor stage, number of nodal areas, lactate dehydrogenase, and hemoglobin level), and one point was given for the presence of lymphopenia. Patients in the low-risk FLIPI-L category had 0-1 points, those in the intermediate-risk category had 2-3 points, and patients in the high-risk FLIPI-L category had 4-6 points.
Using the original FLIPI, the 5-year OS was 91% in the low-risk group (0-1), 82.7% in the intermediate-risk group (2), and 66% in the high-risk group (3-5). The 10-year OS was 80.4%, 66%, and 45.8%, respectively.
Using the FLIPI-L, the 5-year OS was 94.5% in the low-risk group (0-1), 89% in the intermediate-risk group (2-3), and 61% in the high-risk group (4-6). The 10-year OS was 83.9%, 68.5%, and 34.5%, respectively.
In a univariate Cox regression analysis of OS, each point increase in FLIPI-L score was associated with a significant increase in hazard ratio. For example, the hazard ratio was 3.4 for patients with a FLIPI-L score of 1 and 30.9 for those with a FLIPI-L score of 6 (P less than .02 for all FLIPI-L scores). Conversely, increases in hazard ratio were not significant with the original FLIPI (P greater than .05 for all FLIPI scores).
The FLIPI-L was prognostic for OS in different treatment groups. In patients who received rituximab alone, radiation alone, or rituximab plus chemotherapy, the scoring system differentiated low-, intermediate-, and high-risk groups (P less than .04). In patients under observation, the FLIPI-L distinguished low/intermediate-risk and high-risk groups (P less than .01).
For patients who progressed within 24 months, the FLIPI-L was more predictive of progression-free survival (P = .05) than was the original FLIPI (P = .11).
Increasing FLIPI-L was an independent predictor of transformation, both when assessed as a continuous variable (P less than .01) and stepwise for FLIPI-L 3-5 (P = .004-.01). The original FLIPI, on the other hand, was not an independent predictor of transformation.
“Our analysis of a lymphopenia cutoff as an addition to the original FLIPI is simple yet improves risk stratification to differentiate between prognostic groups and, importantly, to predict transformation,” Dr. Yang and his colleagues wrote.
The authors reported having no conflicts of interest.
SOURCE: Yang G et al. Blood Cancer J. 2020 Jan 2;9(12):104. doi: 10.1038/s41408-019-0269-6.
Incorporating lymphopenia into the Follicular Lymphoma International Prognostic Index (FLIPI) can improve prognostication, according to researchers.
The team added lymphopenia as a point in a revised FLIPI scoring system, called FLIPI-L, and found the new system could better predict overall survival (OS), progression-free survival, and histologic transformation in patients with follicular lymphoma.
George Yang, MD, of Moffitt Cancer Center in Tampa, Fla., and his colleagues described results with the FLIPI-L in a letter published in Blood Cancer Journal.
“Prior studies have demonstrated that lymphopenia was associated with worsened OS in [follicular lymphoma],” Dr. Yang and his colleagues wrote. “Therefore, we hypothesized that lymphopenia may be integrated with existing FLIPI to better stratify long-term survival outcomes and predict for transformation.”
The researchers tested this theory in 736 follicular lymphoma patients who were followed for a median of 72 months (range, 2-211 months). The 5-year OS in this cohort was 81.3%, the 10-year OS was 67.3%, and 18% of patients experienced transformation to high-grade lymphoma.
The researchers defined absolute lymphopenia as less than 1.0 × 109 lymphocytes per liter. In multivariate analyses, lymphopenia was an independent predictor of OS (hazard ratio, 1.74; P less than .01) and transformation (odds ratio, 2.1; P less than .01).
To incorporate lymphopenia into the FLIPI, the researchers created a model in which 1 point was given for each of the standard FLIPI components (age, Ann Arbor stage, number of nodal areas, lactate dehydrogenase, and hemoglobin level), and one point was given for the presence of lymphopenia. Patients in the low-risk FLIPI-L category had 0-1 points, those in the intermediate-risk category had 2-3 points, and patients in the high-risk FLIPI-L category had 4-6 points.
Using the original FLIPI, the 5-year OS was 91% in the low-risk group (0-1), 82.7% in the intermediate-risk group (2), and 66% in the high-risk group (3-5). The 10-year OS was 80.4%, 66%, and 45.8%, respectively.
Using the FLIPI-L, the 5-year OS was 94.5% in the low-risk group (0-1), 89% in the intermediate-risk group (2-3), and 61% in the high-risk group (4-6). The 10-year OS was 83.9%, 68.5%, and 34.5%, respectively.
In a univariate Cox regression analysis of OS, each point increase in FLIPI-L score was associated with a significant increase in hazard ratio. For example, the hazard ratio was 3.4 for patients with a FLIPI-L score of 1 and 30.9 for those with a FLIPI-L score of 6 (P less than .02 for all FLIPI-L scores). Conversely, increases in hazard ratio were not significant with the original FLIPI (P greater than .05 for all FLIPI scores).
The FLIPI-L was prognostic for OS in different treatment groups. In patients who received rituximab alone, radiation alone, or rituximab plus chemotherapy, the scoring system differentiated low-, intermediate-, and high-risk groups (P less than .04). In patients under observation, the FLIPI-L distinguished low/intermediate-risk and high-risk groups (P less than .01).
For patients who progressed within 24 months, the FLIPI-L was more predictive of progression-free survival (P = .05) than was the original FLIPI (P = .11).
Increasing FLIPI-L was an independent predictor of transformation, both when assessed as a continuous variable (P less than .01) and stepwise for FLIPI-L 3-5 (P = .004-.01). The original FLIPI, on the other hand, was not an independent predictor of transformation.
“Our analysis of a lymphopenia cutoff as an addition to the original FLIPI is simple yet improves risk stratification to differentiate between prognostic groups and, importantly, to predict transformation,” Dr. Yang and his colleagues wrote.
The authors reported having no conflicts of interest.
SOURCE: Yang G et al. Blood Cancer J. 2020 Jan 2;9(12):104. doi: 10.1038/s41408-019-0269-6.
FROM BLOOD CANCER JOURNAL
Experts break down latest CAR T-cell advances in lymphoma
ORLANDO – There’s now mature data surrounding the use of chimeric antigen receptor (CAR) T-cell therapy in lymphoma, and the annual meeting of the American Society of Hematology brought forth additional information from real-world studies, insights about what is driving relapse, and promising data on mantle cell lymphoma.

The roundtable participants included Brian Hill, MD, of the Cleveland Clinic Taussig Cancer Center; Frederick L. Locke, MD, of the Moffit Cancer Center in Tampa, Fla.; and Peter Riedell, MD, of the University of Chicago.
Among the studies highlighted by the panel was the Transcend NHL 001 study (Abstract 241), which looked at third-line use of lisocabtagene maraleucel (liso-cel) in patients with diffuse large B-cell lymphoma, transformed follicular lymphoma, and other indolent non-Hodgkin lymphoma subtypes. More than 300 patients were enrolled, and liso-cel met all primary and secondary efficacy endpoints, with an overall response rate of more than 70%. The notable take-home point from the study was the safety profile, Dr. Riedell noted. Liso-cel was associated with a lower rate of cytokine release syndrome and neurologic toxicity, compared with the currently approved products.
Since patients in the study had a lower incidence and later onset of cytokine release syndrome, liso-cel could be a candidate for outpatient administration, Dr. Locke said. However, doing that would require “significant infrastructure” in hospitals and clinics to properly support patients, especially given that the treatment-related mortality on the study was similar to approved CAR T-cell products at about 3%. “You have to be ready to admit the patient to the hospital very rapidly, and you have to have the providers and the nurses who are vigilant when the patient is not in the hospital,” he said.
Another notable study presented at ASH examined the characteristics and outcomes of patients receiving bridging therapy while awaiting treatment with axicabtagene ciloleucel (Abstract 245). This real-world study adds interesting information to the field because, in some of the studies that were pivotal to the approval of CAR T-cell therapy, bridging therapy was not allowed, Dr. Locke said.
In this analysis, researchers found that the overall survival was worse among patients who received bridging. This finding suggests that patients who received bridging therapy had a different biology or that the therapy itself may have had an effect on the host or tumor microenvironment that affected the efficacy of the CAR T-cell therapy, the researchers reported.
The panel also highlighted the Zuma-2 study, which looked at KTE-X19, an anti-CD19 CAR T-cell therapy, among more than 70 patients with relapsed/refractory mantle cell lymphoma who had failed treatment with a Bruton’s tyrosine kinase inhibitor (Abstract 754). “This was, I thought, kind of a sleeper study at ASH,” said Dr. Hill, who was one of the authors of the study.
The overall response rate was 93% with about two-thirds of patients achieving a complete response. Researchers found that the response was consistent across subgroups, including Ki-67 and patients with prior use of steroids or bridging therapy. Dr. Locke, who was also a study author, said the results are a “game changer.”
“I’m very excited about it,” Dr. Riedell said, noting that these are patients without a lot of treatment options.
The panel also discussed other studies from ASH, including an analysis of tumor tissue samples from patients in the ZUMA-1 trial who had responded and subsequently relapsed (Abstract 203); a multicenter prospective analysis of circulating tumor DNA in diffuse large B-cell lymphoma patients who had relapsed after treatment with axicabtagene ciloleucel (Abstract 884); and the early use of corticosteroids to prevent toxicities in patients in cohort 4 of the ZUMA-1 trial (Abstract 243).
Dr. Hill reported consulting with Juno/Celgene/BMS and Novartis and research and consulting for Kite/Gilead. Dr. Locke reported consulting for Cellular Biomedicine Group and being a scientific adviser to Kite/Gilead, Novartis, Celgene/BMS, GammaDelta Therapeutics, Calibr, and Allogene. Dr. Riedell reported consulting for Bayer and Verastem, consulting for and research funding from Novartis and BMS/Celgene, and consulting for, research funding from, and speaking for Kite.
ORLANDO – There’s now mature data surrounding the use of chimeric antigen receptor (CAR) T-cell therapy in lymphoma, and the annual meeting of the American Society of Hematology brought forth additional information from real-world studies, insights about what is driving relapse, and promising data on mantle cell lymphoma.

The roundtable participants included Brian Hill, MD, of the Cleveland Clinic Taussig Cancer Center; Frederick L. Locke, MD, of the Moffit Cancer Center in Tampa, Fla.; and Peter Riedell, MD, of the University of Chicago.
Among the studies highlighted by the panel was the Transcend NHL 001 study (Abstract 241), which looked at third-line use of lisocabtagene maraleucel (liso-cel) in patients with diffuse large B-cell lymphoma, transformed follicular lymphoma, and other indolent non-Hodgkin lymphoma subtypes. More than 300 patients were enrolled, and liso-cel met all primary and secondary efficacy endpoints, with an overall response rate of more than 70%. The notable take-home point from the study was the safety profile, Dr. Riedell noted. Liso-cel was associated with a lower rate of cytokine release syndrome and neurologic toxicity, compared with the currently approved products.
Since patients in the study had a lower incidence and later onset of cytokine release syndrome, liso-cel could be a candidate for outpatient administration, Dr. Locke said. However, doing that would require “significant infrastructure” in hospitals and clinics to properly support patients, especially given that the treatment-related mortality on the study was similar to approved CAR T-cell products at about 3%. “You have to be ready to admit the patient to the hospital very rapidly, and you have to have the providers and the nurses who are vigilant when the patient is not in the hospital,” he said.
Another notable study presented at ASH examined the characteristics and outcomes of patients receiving bridging therapy while awaiting treatment with axicabtagene ciloleucel (Abstract 245). This real-world study adds interesting information to the field because, in some of the studies that were pivotal to the approval of CAR T-cell therapy, bridging therapy was not allowed, Dr. Locke said.
In this analysis, researchers found that the overall survival was worse among patients who received bridging. This finding suggests that patients who received bridging therapy had a different biology or that the therapy itself may have had an effect on the host or tumor microenvironment that affected the efficacy of the CAR T-cell therapy, the researchers reported.
The panel also highlighted the Zuma-2 study, which looked at KTE-X19, an anti-CD19 CAR T-cell therapy, among more than 70 patients with relapsed/refractory mantle cell lymphoma who had failed treatment with a Bruton’s tyrosine kinase inhibitor (Abstract 754). “This was, I thought, kind of a sleeper study at ASH,” said Dr. Hill, who was one of the authors of the study.
The overall response rate was 93% with about two-thirds of patients achieving a complete response. Researchers found that the response was consistent across subgroups, including Ki-67 and patients with prior use of steroids or bridging therapy. Dr. Locke, who was also a study author, said the results are a “game changer.”
“I’m very excited about it,” Dr. Riedell said, noting that these are patients without a lot of treatment options.
The panel also discussed other studies from ASH, including an analysis of tumor tissue samples from patients in the ZUMA-1 trial who had responded and subsequently relapsed (Abstract 203); a multicenter prospective analysis of circulating tumor DNA in diffuse large B-cell lymphoma patients who had relapsed after treatment with axicabtagene ciloleucel (Abstract 884); and the early use of corticosteroids to prevent toxicities in patients in cohort 4 of the ZUMA-1 trial (Abstract 243).
Dr. Hill reported consulting with Juno/Celgene/BMS and Novartis and research and consulting for Kite/Gilead. Dr. Locke reported consulting for Cellular Biomedicine Group and being a scientific adviser to Kite/Gilead, Novartis, Celgene/BMS, GammaDelta Therapeutics, Calibr, and Allogene. Dr. Riedell reported consulting for Bayer and Verastem, consulting for and research funding from Novartis and BMS/Celgene, and consulting for, research funding from, and speaking for Kite.
ORLANDO – There’s now mature data surrounding the use of chimeric antigen receptor (CAR) T-cell therapy in lymphoma, and the annual meeting of the American Society of Hematology brought forth additional information from real-world studies, insights about what is driving relapse, and promising data on mantle cell lymphoma.

The roundtable participants included Brian Hill, MD, of the Cleveland Clinic Taussig Cancer Center; Frederick L. Locke, MD, of the Moffit Cancer Center in Tampa, Fla.; and Peter Riedell, MD, of the University of Chicago.
Among the studies highlighted by the panel was the Transcend NHL 001 study (Abstract 241), which looked at third-line use of lisocabtagene maraleucel (liso-cel) in patients with diffuse large B-cell lymphoma, transformed follicular lymphoma, and other indolent non-Hodgkin lymphoma subtypes. More than 300 patients were enrolled, and liso-cel met all primary and secondary efficacy endpoints, with an overall response rate of more than 70%. The notable take-home point from the study was the safety profile, Dr. Riedell noted. Liso-cel was associated with a lower rate of cytokine release syndrome and neurologic toxicity, compared with the currently approved products.
Since patients in the study had a lower incidence and later onset of cytokine release syndrome, liso-cel could be a candidate for outpatient administration, Dr. Locke said. However, doing that would require “significant infrastructure” in hospitals and clinics to properly support patients, especially given that the treatment-related mortality on the study was similar to approved CAR T-cell products at about 3%. “You have to be ready to admit the patient to the hospital very rapidly, and you have to have the providers and the nurses who are vigilant when the patient is not in the hospital,” he said.
Another notable study presented at ASH examined the characteristics and outcomes of patients receiving bridging therapy while awaiting treatment with axicabtagene ciloleucel (Abstract 245). This real-world study adds interesting information to the field because, in some of the studies that were pivotal to the approval of CAR T-cell therapy, bridging therapy was not allowed, Dr. Locke said.
In this analysis, researchers found that the overall survival was worse among patients who received bridging. This finding suggests that patients who received bridging therapy had a different biology or that the therapy itself may have had an effect on the host or tumor microenvironment that affected the efficacy of the CAR T-cell therapy, the researchers reported.
The panel also highlighted the Zuma-2 study, which looked at KTE-X19, an anti-CD19 CAR T-cell therapy, among more than 70 patients with relapsed/refractory mantle cell lymphoma who had failed treatment with a Bruton’s tyrosine kinase inhibitor (Abstract 754). “This was, I thought, kind of a sleeper study at ASH,” said Dr. Hill, who was one of the authors of the study.
The overall response rate was 93% with about two-thirds of patients achieving a complete response. Researchers found that the response was consistent across subgroups, including Ki-67 and patients with prior use of steroids or bridging therapy. Dr. Locke, who was also a study author, said the results are a “game changer.”
“I’m very excited about it,” Dr. Riedell said, noting that these are patients without a lot of treatment options.
The panel also discussed other studies from ASH, including an analysis of tumor tissue samples from patients in the ZUMA-1 trial who had responded and subsequently relapsed (Abstract 203); a multicenter prospective analysis of circulating tumor DNA in diffuse large B-cell lymphoma patients who had relapsed after treatment with axicabtagene ciloleucel (Abstract 884); and the early use of corticosteroids to prevent toxicities in patients in cohort 4 of the ZUMA-1 trial (Abstract 243).
Dr. Hill reported consulting with Juno/Celgene/BMS and Novartis and research and consulting for Kite/Gilead. Dr. Locke reported consulting for Cellular Biomedicine Group and being a scientific adviser to Kite/Gilead, Novartis, Celgene/BMS, GammaDelta Therapeutics, Calibr, and Allogene. Dr. Riedell reported consulting for Bayer and Verastem, consulting for and research funding from Novartis and BMS/Celgene, and consulting for, research funding from, and speaking for Kite.
EXPERT ANALYSIS FROM ASH 2019
REGN1979 shows good activity in pretreated aggressive B-NHL
ORLANDO – A novel bispecific antibody directed against CD20 and CD3 was associated in a phase 1 trial with a high overall response rate among patients with relapsed or refractory B-cell non-Hodgkin lymphomas in early clinical trials, including patients with diffuse large B-cell lymphoma (DLBCL) that had progressed following chimeric antigen receptor (CAR) T-cell therapy.
Among 22 patients previously treated for relapsed/refractory follicular lymphoma of grade 1-3a, there were 21 responses (95%), including 17 complete responses (CR) and 4 partial responses (PR), with the remaining patient having stable disease at 12 weeks of follow-up, reported Rajat Bannerji, MD, PhD, from the Rutgers Cancer Institute of New Jersey in New Brunswick.
“We had activity that was fairly robust in this heavily pretreated population with follicular lymphoma, large-cell patients who had not received CAR T and large-cell patients who had received CAR T, mantle cell, and marginal zone [lymphoma],” he said at the annual meeting of the American Society of Hematology.
REGN1979 is an anti-CD20 and anti-CD3 bispecific IgG4 antibody. It is designed to cross-link and activate CD3-expressing T cells on contact with CD20-positive B cells to kill CD20-positive tumor cells independent of T-cell receptor recognition.
The antibody is administered via an escalating dose schedule consisting of initial, intermediate, and step-up doses.
In addition to the follicular lymphoma response rates noted before, patients with heavily pretreated DLBCL who received the antibody at a dose of 80 mg or higher had an overall response rate of 57.9% (11 patients) including 42.1% CR (8 patients), and 15.8% PR (3 patients). Two patients had stable disease at the 12-week assessment, three had disease progression, and three were not available for assessment.
Among seven patients with DLBCL treated at 80 mg or above who had not received CAR T therapy, five had a CR, one had stable disease, and one had disease progression. Of 12 patients with prior CAR T exposure, 3 had complete responses, 3 had partial responses, 1 had stable disease, 2 had progressive disease, and 3 were not available for assessment.
Among six patients with mantle cell lymphoma and six with marginal zone lymphoma treated across all disease levels, the ORR in each cohort was 67%, with two of six patients in each cohort having a complete response, and two having a partial response.
The safety analysis of all 110 patients enrolled showed that no patients experience a dose-limiting toxicity during the escalation phase, and no maximum tolerated doses were identified.
The most common treatment-related adverse events (AEs) were pyrexia in 88 patients, cytokine release syndrome in 65, chills in 56, fatigue in 40, and anemia in 39.
The most common grade 3-4 AEs were anemia in 24, and hypophosphatemia, lymphopenia, and neutropenia in 21 patients each.
Neurologic AEs were transient and did not require treatment discontinuation, and there were no grade 4 neurologic AEs or deaths from neurologic side effects.
Six patients discontinued the study drug because of treatment-related AEs that included cytomegalovirus infection, grade 3 hemolysis, fatigue, pneumonia, and toxoplasmosis.
A total of 15 patients died during the study, 10 of which were caused by progressive disease, with other deaths caused by gastric perforation, cardiac arrest, lung infection, pneumonia, and 1 from fungal pneumonia 7 months after treatment discontinuation. In addition, after the data cutoff, one patient with mantle cell lymphoma blastoid variant with bone-marrow involvement and bulky disease who was enrolled in an expansion cohort died from tumor lysis syndrome.
The dose-escalation portion of the trial has been completed and expansion cohorts are being enrolled. In addition, REGN1979 is being investigated in a phase 2 global multiarm trial.
The study is supported by Regeneron Pharmaceuticals. Dr. Bannerji reported research funding, travel support, and consulting fees from Regeneron and others.
SOURCE: Bannerji R et al. ASH 2019, Abstract 762.
ORLANDO – A novel bispecific antibody directed against CD20 and CD3 was associated in a phase 1 trial with a high overall response rate among patients with relapsed or refractory B-cell non-Hodgkin lymphomas in early clinical trials, including patients with diffuse large B-cell lymphoma (DLBCL) that had progressed following chimeric antigen receptor (CAR) T-cell therapy.
Among 22 patients previously treated for relapsed/refractory follicular lymphoma of grade 1-3a, there were 21 responses (95%), including 17 complete responses (CR) and 4 partial responses (PR), with the remaining patient having stable disease at 12 weeks of follow-up, reported Rajat Bannerji, MD, PhD, from the Rutgers Cancer Institute of New Jersey in New Brunswick.
“We had activity that was fairly robust in this heavily pretreated population with follicular lymphoma, large-cell patients who had not received CAR T and large-cell patients who had received CAR T, mantle cell, and marginal zone [lymphoma],” he said at the annual meeting of the American Society of Hematology.
REGN1979 is an anti-CD20 and anti-CD3 bispecific IgG4 antibody. It is designed to cross-link and activate CD3-expressing T cells on contact with CD20-positive B cells to kill CD20-positive tumor cells independent of T-cell receptor recognition.
The antibody is administered via an escalating dose schedule consisting of initial, intermediate, and step-up doses.
In addition to the follicular lymphoma response rates noted before, patients with heavily pretreated DLBCL who received the antibody at a dose of 80 mg or higher had an overall response rate of 57.9% (11 patients) including 42.1% CR (8 patients), and 15.8% PR (3 patients). Two patients had stable disease at the 12-week assessment, three had disease progression, and three were not available for assessment.
Among seven patients with DLBCL treated at 80 mg or above who had not received CAR T therapy, five had a CR, one had stable disease, and one had disease progression. Of 12 patients with prior CAR T exposure, 3 had complete responses, 3 had partial responses, 1 had stable disease, 2 had progressive disease, and 3 were not available for assessment.
Among six patients with mantle cell lymphoma and six with marginal zone lymphoma treated across all disease levels, the ORR in each cohort was 67%, with two of six patients in each cohort having a complete response, and two having a partial response.
The safety analysis of all 110 patients enrolled showed that no patients experience a dose-limiting toxicity during the escalation phase, and no maximum tolerated doses were identified.
The most common treatment-related adverse events (AEs) were pyrexia in 88 patients, cytokine release syndrome in 65, chills in 56, fatigue in 40, and anemia in 39.
The most common grade 3-4 AEs were anemia in 24, and hypophosphatemia, lymphopenia, and neutropenia in 21 patients each.
Neurologic AEs were transient and did not require treatment discontinuation, and there were no grade 4 neurologic AEs or deaths from neurologic side effects.
Six patients discontinued the study drug because of treatment-related AEs that included cytomegalovirus infection, grade 3 hemolysis, fatigue, pneumonia, and toxoplasmosis.
A total of 15 patients died during the study, 10 of which were caused by progressive disease, with other deaths caused by gastric perforation, cardiac arrest, lung infection, pneumonia, and 1 from fungal pneumonia 7 months after treatment discontinuation. In addition, after the data cutoff, one patient with mantle cell lymphoma blastoid variant with bone-marrow involvement and bulky disease who was enrolled in an expansion cohort died from tumor lysis syndrome.
The dose-escalation portion of the trial has been completed and expansion cohorts are being enrolled. In addition, REGN1979 is being investigated in a phase 2 global multiarm trial.
The study is supported by Regeneron Pharmaceuticals. Dr. Bannerji reported research funding, travel support, and consulting fees from Regeneron and others.
SOURCE: Bannerji R et al. ASH 2019, Abstract 762.
ORLANDO – A novel bispecific antibody directed against CD20 and CD3 was associated in a phase 1 trial with a high overall response rate among patients with relapsed or refractory B-cell non-Hodgkin lymphomas in early clinical trials, including patients with diffuse large B-cell lymphoma (DLBCL) that had progressed following chimeric antigen receptor (CAR) T-cell therapy.
Among 22 patients previously treated for relapsed/refractory follicular lymphoma of grade 1-3a, there were 21 responses (95%), including 17 complete responses (CR) and 4 partial responses (PR), with the remaining patient having stable disease at 12 weeks of follow-up, reported Rajat Bannerji, MD, PhD, from the Rutgers Cancer Institute of New Jersey in New Brunswick.
“We had activity that was fairly robust in this heavily pretreated population with follicular lymphoma, large-cell patients who had not received CAR T and large-cell patients who had received CAR T, mantle cell, and marginal zone [lymphoma],” he said at the annual meeting of the American Society of Hematology.
REGN1979 is an anti-CD20 and anti-CD3 bispecific IgG4 antibody. It is designed to cross-link and activate CD3-expressing T cells on contact with CD20-positive B cells to kill CD20-positive tumor cells independent of T-cell receptor recognition.
The antibody is administered via an escalating dose schedule consisting of initial, intermediate, and step-up doses.
In addition to the follicular lymphoma response rates noted before, patients with heavily pretreated DLBCL who received the antibody at a dose of 80 mg or higher had an overall response rate of 57.9% (11 patients) including 42.1% CR (8 patients), and 15.8% PR (3 patients). Two patients had stable disease at the 12-week assessment, three had disease progression, and three were not available for assessment.
Among seven patients with DLBCL treated at 80 mg or above who had not received CAR T therapy, five had a CR, one had stable disease, and one had disease progression. Of 12 patients with prior CAR T exposure, 3 had complete responses, 3 had partial responses, 1 had stable disease, 2 had progressive disease, and 3 were not available for assessment.
Among six patients with mantle cell lymphoma and six with marginal zone lymphoma treated across all disease levels, the ORR in each cohort was 67%, with two of six patients in each cohort having a complete response, and two having a partial response.
The safety analysis of all 110 patients enrolled showed that no patients experience a dose-limiting toxicity during the escalation phase, and no maximum tolerated doses were identified.
The most common treatment-related adverse events (AEs) were pyrexia in 88 patients, cytokine release syndrome in 65, chills in 56, fatigue in 40, and anemia in 39.
The most common grade 3-4 AEs were anemia in 24, and hypophosphatemia, lymphopenia, and neutropenia in 21 patients each.
Neurologic AEs were transient and did not require treatment discontinuation, and there were no grade 4 neurologic AEs or deaths from neurologic side effects.
Six patients discontinued the study drug because of treatment-related AEs that included cytomegalovirus infection, grade 3 hemolysis, fatigue, pneumonia, and toxoplasmosis.
A total of 15 patients died during the study, 10 of which were caused by progressive disease, with other deaths caused by gastric perforation, cardiac arrest, lung infection, pneumonia, and 1 from fungal pneumonia 7 months after treatment discontinuation. In addition, after the data cutoff, one patient with mantle cell lymphoma blastoid variant with bone-marrow involvement and bulky disease who was enrolled in an expansion cohort died from tumor lysis syndrome.
The dose-escalation portion of the trial has been completed and expansion cohorts are being enrolled. In addition, REGN1979 is being investigated in a phase 2 global multiarm trial.
The study is supported by Regeneron Pharmaceuticals. Dr. Bannerji reported research funding, travel support, and consulting fees from Regeneron and others.
SOURCE: Bannerji R et al. ASH 2019, Abstract 762.
REPORTING FROM ASH 2019
NCCN guidelines highlight ‘complicated’ treatment for pediatric lymphomas
The National Comprehensive Cancer Network (NCCN) has released its first set of guidelines for managing pediatric aggressive mature B-cell lymphomas.
The guidelines highlight the complexities of treating pediatric Burkitt lymphoma (BL) and diffuse large B-cell lymphoma (DLBCL), as recommendations include a range of multiagent regimens for different patient groups at various time points.
“The treatment of this disease is relatively complicated,” said Kimberly J. Davies, MD, a pediatric hematologist/oncologist at Dana-Farber Cancer Institute in Boston and chair of the guidelines panel. “The chemotherapy regimens have a lot of drugs, a lot of nuances to how they’re supposed to be given. These guidelines delineate that treatment and help the provider … make sure they are delivering the treatment a patient needs.”
The guidelines recommend different regimens according to a patient’s risk group, but the same treatment approach should be used for patients with BL and those with DLBCL.
“The biggest difference between pediatric and adult patients is that pediatric patients are more uniformly treated, regardless of what type of aggressive B-cell lymphoma they have,” said Matthew Barth, MD, a pediatric hematologist/oncologist at Roswell Park Comprehensive Cancer Center in Buffalo, N.Y., and vice chair of the NCCN guidelines panel.
“Adults with diffuse large B-cell lymphoma and Burkitt lymphoma are generally treated with different chemotherapy regimens, but, in pediatrics, we use the same treatment regimens for both diffuse large B-cell lymphoma and Burkitt lymphoma,” he added.
As an example, the new guidelines recommend that pediatric patients with low-risk BL/DLBCL receive the POG9219 regimen (N Engl J Med. 1997 Oct 30;337[18]:1259-66) or FAB/LMB96 regimen A (Br J Haematol. 2008 Jun;141[6]:840-7) as induction, or they should be enrolled in a clinical trial.
On the other hand, induction for high-risk pediatric BL/DLBCL patients should consist of rituximab and a chemotherapy regimen used in the COG ANHL1131 trial. The recommendation to incorporate rituximab in high-risk pediatric patients is based on results from that trial (J Clin Oncol. 2016 May 20. doi: 10.1200/JCO.2016.34.15_suppl.10507).
“Until recent clinical trial data was available, we weren’t really sure how to incorporate rituximab into the treatment of pediatric patients with mature B-cell lymphomas,” Dr. Barth said. “We now have evidence that rituximab is clearly beneficial for patients who are in higher-risk groups.”
Dr. Barth and Dr. Davies both noted that pediatric BL and DLBCL have high cure rates. Long-term survival rates range from about 80% to more than 90%, according to the American Cancer Society. However, the patients who do relapse or progress can be difficult to treat.
“We have quite good cure rates at this point in time, which is a great success, but that means that a very small population of patients don’t respond to initial therapy, and … it’s hard to know what the best treatment for those patients is,” Dr. Davies said.
She noted that studies are underway to determine if immunotherapies, including chimeric antigen receptor T-cell therapy, might improve outcomes in patients with relapsed or refractory disease.
For now, the NCCN guidelines recommend clinical trial enrollment for relapsed/refractory patients. Alternatively, these patients can receive additional chemotherapy, and responders can proceed to transplant. Patients who don’t achieve at least a partial response may go on to a clinical trial or receive best supportive care.
Dr. Davies and Dr. Barth reported having no conflicts of interest.
The National Comprehensive Cancer Network (NCCN) has released its first set of guidelines for managing pediatric aggressive mature B-cell lymphomas.
The guidelines highlight the complexities of treating pediatric Burkitt lymphoma (BL) and diffuse large B-cell lymphoma (DLBCL), as recommendations include a range of multiagent regimens for different patient groups at various time points.
“The treatment of this disease is relatively complicated,” said Kimberly J. Davies, MD, a pediatric hematologist/oncologist at Dana-Farber Cancer Institute in Boston and chair of the guidelines panel. “The chemotherapy regimens have a lot of drugs, a lot of nuances to how they’re supposed to be given. These guidelines delineate that treatment and help the provider … make sure they are delivering the treatment a patient needs.”
The guidelines recommend different regimens according to a patient’s risk group, but the same treatment approach should be used for patients with BL and those with DLBCL.
“The biggest difference between pediatric and adult patients is that pediatric patients are more uniformly treated, regardless of what type of aggressive B-cell lymphoma they have,” said Matthew Barth, MD, a pediatric hematologist/oncologist at Roswell Park Comprehensive Cancer Center in Buffalo, N.Y., and vice chair of the NCCN guidelines panel.
“Adults with diffuse large B-cell lymphoma and Burkitt lymphoma are generally treated with different chemotherapy regimens, but, in pediatrics, we use the same treatment regimens for both diffuse large B-cell lymphoma and Burkitt lymphoma,” he added.
As an example, the new guidelines recommend that pediatric patients with low-risk BL/DLBCL receive the POG9219 regimen (N Engl J Med. 1997 Oct 30;337[18]:1259-66) or FAB/LMB96 regimen A (Br J Haematol. 2008 Jun;141[6]:840-7) as induction, or they should be enrolled in a clinical trial.
On the other hand, induction for high-risk pediatric BL/DLBCL patients should consist of rituximab and a chemotherapy regimen used in the COG ANHL1131 trial. The recommendation to incorporate rituximab in high-risk pediatric patients is based on results from that trial (J Clin Oncol. 2016 May 20. doi: 10.1200/JCO.2016.34.15_suppl.10507).
“Until recent clinical trial data was available, we weren’t really sure how to incorporate rituximab into the treatment of pediatric patients with mature B-cell lymphomas,” Dr. Barth said. “We now have evidence that rituximab is clearly beneficial for patients who are in higher-risk groups.”
Dr. Barth and Dr. Davies both noted that pediatric BL and DLBCL have high cure rates. Long-term survival rates range from about 80% to more than 90%, according to the American Cancer Society. However, the patients who do relapse or progress can be difficult to treat.
“We have quite good cure rates at this point in time, which is a great success, but that means that a very small population of patients don’t respond to initial therapy, and … it’s hard to know what the best treatment for those patients is,” Dr. Davies said.
She noted that studies are underway to determine if immunotherapies, including chimeric antigen receptor T-cell therapy, might improve outcomes in patients with relapsed or refractory disease.
For now, the NCCN guidelines recommend clinical trial enrollment for relapsed/refractory patients. Alternatively, these patients can receive additional chemotherapy, and responders can proceed to transplant. Patients who don’t achieve at least a partial response may go on to a clinical trial or receive best supportive care.
Dr. Davies and Dr. Barth reported having no conflicts of interest.
The National Comprehensive Cancer Network (NCCN) has released its first set of guidelines for managing pediatric aggressive mature B-cell lymphomas.
The guidelines highlight the complexities of treating pediatric Burkitt lymphoma (BL) and diffuse large B-cell lymphoma (DLBCL), as recommendations include a range of multiagent regimens for different patient groups at various time points.
“The treatment of this disease is relatively complicated,” said Kimberly J. Davies, MD, a pediatric hematologist/oncologist at Dana-Farber Cancer Institute in Boston and chair of the guidelines panel. “The chemotherapy regimens have a lot of drugs, a lot of nuances to how they’re supposed to be given. These guidelines delineate that treatment and help the provider … make sure they are delivering the treatment a patient needs.”
The guidelines recommend different regimens according to a patient’s risk group, but the same treatment approach should be used for patients with BL and those with DLBCL.
“The biggest difference between pediatric and adult patients is that pediatric patients are more uniformly treated, regardless of what type of aggressive B-cell lymphoma they have,” said Matthew Barth, MD, a pediatric hematologist/oncologist at Roswell Park Comprehensive Cancer Center in Buffalo, N.Y., and vice chair of the NCCN guidelines panel.
“Adults with diffuse large B-cell lymphoma and Burkitt lymphoma are generally treated with different chemotherapy regimens, but, in pediatrics, we use the same treatment regimens for both diffuse large B-cell lymphoma and Burkitt lymphoma,” he added.
As an example, the new guidelines recommend that pediatric patients with low-risk BL/DLBCL receive the POG9219 regimen (N Engl J Med. 1997 Oct 30;337[18]:1259-66) or FAB/LMB96 regimen A (Br J Haematol. 2008 Jun;141[6]:840-7) as induction, or they should be enrolled in a clinical trial.
On the other hand, induction for high-risk pediatric BL/DLBCL patients should consist of rituximab and a chemotherapy regimen used in the COG ANHL1131 trial. The recommendation to incorporate rituximab in high-risk pediatric patients is based on results from that trial (J Clin Oncol. 2016 May 20. doi: 10.1200/JCO.2016.34.15_suppl.10507).
“Until recent clinical trial data was available, we weren’t really sure how to incorporate rituximab into the treatment of pediatric patients with mature B-cell lymphomas,” Dr. Barth said. “We now have evidence that rituximab is clearly beneficial for patients who are in higher-risk groups.”
Dr. Barth and Dr. Davies both noted that pediatric BL and DLBCL have high cure rates. Long-term survival rates range from about 80% to more than 90%, according to the American Cancer Society. However, the patients who do relapse or progress can be difficult to treat.
“We have quite good cure rates at this point in time, which is a great success, but that means that a very small population of patients don’t respond to initial therapy, and … it’s hard to know what the best treatment for those patients is,” Dr. Davies said.
She noted that studies are underway to determine if immunotherapies, including chimeric antigen receptor T-cell therapy, might improve outcomes in patients with relapsed or refractory disease.
For now, the NCCN guidelines recommend clinical trial enrollment for relapsed/refractory patients. Alternatively, these patients can receive additional chemotherapy, and responders can proceed to transplant. Patients who don’t achieve at least a partial response may go on to a clinical trial or receive best supportive care.
Dr. Davies and Dr. Barth reported having no conflicts of interest.
CAR T-cell therapy advances in CLL
ORLANDO – Lisocabtagene maraleucel (liso-cel), a CD19-directed chimeric antigen receptor (CAR) T-cell therapy, has demonstrated manageable toxicity and promising clinical activity in the phase 1 portion of a trial enrolling heavily pretreated patients with chronic lymphocytic leukemia/small lymphocytic lymphoma, according to an investigator.
The overall response rate exceeded 80%, and most patients in response at 6 months had maintained that response at the 9-month mark, said Tanya Siddiqi, MD, of City of Hope National Medical Center, Duarte, Calif.
“Clinical responses were rapid, improved with time, and were deep and durable,” Dr. Siddiqi said at the annual meeting of the American Society of Hematology.
These findings have provided justification for conducting the phase 2 portion of the study, which is currently enrolling at the higher of two dose levels evaluated in phase 1, she added.
Dr. Siddiqi reported on a total of 23 patients enrolled in the study, known as TRANSCEND CLL 004. All patients had relapsed/refractory chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL), and had received at least two prior therapies, including ibrutinib, while about one-third had failed venetoclax as well.
The median patient age was 66 years, and 83% had high-risk features, according to Dr. Siddiqi, who said patients had received a median of five prior lines of therapy.
Nine patients were treated at dose level 1, or 50 x 106 CAR+ T cells, while 14 were treated at dose level 2, or 100 x 106 CAR+ T cells. Two patients experienced grade 3 or 4 dose-limiting toxicities at the second level, including hypertension in one patient, and encephalopathy, muscle weakness, and tumor lysis syndrome (TLS) in the other.
Cytokine release syndrome (CRS) occurred in 17 patients, though only two cases reached grade 3. Neurologic adverse events were seen in nine patients, of which five were grade 3 or 4.
Partial or complete responses were noted in 81.5%, or 18 of 22 evaluable patients, including 10 (45.5%) who had complete remission. In the subset of nine patients who had failed both ibrutinib and venetoclax, that overall response rate was a “very impressive” 89% (eight of nine patients), said Dr. Siddiqi, including 67% complete remissions (six patients).
Undetectable minimal residual disease (MRD) was reported in 65% and 75% of patients, depending on the method used to evaluate it.
About two-thirds of the patients had responses by day 30 evaluation, and responses deepened over time in about one-quarter, according to Dr. Siddiqi. Of 12 patients with a response at 6 months, 10 (83%) were still in response at 9 months, and 8 patients have been in response for 12 months or longer, she reported.
Neurologic adverse events seen in the CLL/SLL patients in this study were associated with higher lymph node tumor burden, and increased levels of interleukin(IL)-16 or tumor necrosis factor (TNF), according to further analysis presented by Dr. Siddiqi.
That raises the possibility that IL-16 or TNF may be a “good predictive biomarker” for neurotoxicity, which seems to be driven at least in part by lymphadenopathy. “If there was a way that we could combine the CAR T-cell with something like a novel agent that can shrink the tumor burden quickly, then maybe we can have even less toxicities with these CAR T cells,” Dr. Siddiqi said.
Dr. Siddiqi reported disclosures related to Kite, TG Therapeutics, Celgene, Janssen, Seattle Genetics, AstraZeneca, PCYC, Juno Therapeutics, and BeiGene.
SOURCE: Siddiqi T et al. ASH 2019, Abstract 503.
ORLANDO – Lisocabtagene maraleucel (liso-cel), a CD19-directed chimeric antigen receptor (CAR) T-cell therapy, has demonstrated manageable toxicity and promising clinical activity in the phase 1 portion of a trial enrolling heavily pretreated patients with chronic lymphocytic leukemia/small lymphocytic lymphoma, according to an investigator.
The overall response rate exceeded 80%, and most patients in response at 6 months had maintained that response at the 9-month mark, said Tanya Siddiqi, MD, of City of Hope National Medical Center, Duarte, Calif.
“Clinical responses were rapid, improved with time, and were deep and durable,” Dr. Siddiqi said at the annual meeting of the American Society of Hematology.
These findings have provided justification for conducting the phase 2 portion of the study, which is currently enrolling at the higher of two dose levels evaluated in phase 1, she added.
Dr. Siddiqi reported on a total of 23 patients enrolled in the study, known as TRANSCEND CLL 004. All patients had relapsed/refractory chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL), and had received at least two prior therapies, including ibrutinib, while about one-third had failed venetoclax as well.
The median patient age was 66 years, and 83% had high-risk features, according to Dr. Siddiqi, who said patients had received a median of five prior lines of therapy.
Nine patients were treated at dose level 1, or 50 x 106 CAR+ T cells, while 14 were treated at dose level 2, or 100 x 106 CAR+ T cells. Two patients experienced grade 3 or 4 dose-limiting toxicities at the second level, including hypertension in one patient, and encephalopathy, muscle weakness, and tumor lysis syndrome (TLS) in the other.
Cytokine release syndrome (CRS) occurred in 17 patients, though only two cases reached grade 3. Neurologic adverse events were seen in nine patients, of which five were grade 3 or 4.
Partial or complete responses were noted in 81.5%, or 18 of 22 evaluable patients, including 10 (45.5%) who had complete remission. In the subset of nine patients who had failed both ibrutinib and venetoclax, that overall response rate was a “very impressive” 89% (eight of nine patients), said Dr. Siddiqi, including 67% complete remissions (six patients).
Undetectable minimal residual disease (MRD) was reported in 65% and 75% of patients, depending on the method used to evaluate it.
About two-thirds of the patients had responses by day 30 evaluation, and responses deepened over time in about one-quarter, according to Dr. Siddiqi. Of 12 patients with a response at 6 months, 10 (83%) were still in response at 9 months, and 8 patients have been in response for 12 months or longer, she reported.
Neurologic adverse events seen in the CLL/SLL patients in this study were associated with higher lymph node tumor burden, and increased levels of interleukin(IL)-16 or tumor necrosis factor (TNF), according to further analysis presented by Dr. Siddiqi.
That raises the possibility that IL-16 or TNF may be a “good predictive biomarker” for neurotoxicity, which seems to be driven at least in part by lymphadenopathy. “If there was a way that we could combine the CAR T-cell with something like a novel agent that can shrink the tumor burden quickly, then maybe we can have even less toxicities with these CAR T cells,” Dr. Siddiqi said.
Dr. Siddiqi reported disclosures related to Kite, TG Therapeutics, Celgene, Janssen, Seattle Genetics, AstraZeneca, PCYC, Juno Therapeutics, and BeiGene.
SOURCE: Siddiqi T et al. ASH 2019, Abstract 503.
ORLANDO – Lisocabtagene maraleucel (liso-cel), a CD19-directed chimeric antigen receptor (CAR) T-cell therapy, has demonstrated manageable toxicity and promising clinical activity in the phase 1 portion of a trial enrolling heavily pretreated patients with chronic lymphocytic leukemia/small lymphocytic lymphoma, according to an investigator.
The overall response rate exceeded 80%, and most patients in response at 6 months had maintained that response at the 9-month mark, said Tanya Siddiqi, MD, of City of Hope National Medical Center, Duarte, Calif.
“Clinical responses were rapid, improved with time, and were deep and durable,” Dr. Siddiqi said at the annual meeting of the American Society of Hematology.
These findings have provided justification for conducting the phase 2 portion of the study, which is currently enrolling at the higher of two dose levels evaluated in phase 1, she added.
Dr. Siddiqi reported on a total of 23 patients enrolled in the study, known as TRANSCEND CLL 004. All patients had relapsed/refractory chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL), and had received at least two prior therapies, including ibrutinib, while about one-third had failed venetoclax as well.
The median patient age was 66 years, and 83% had high-risk features, according to Dr. Siddiqi, who said patients had received a median of five prior lines of therapy.
Nine patients were treated at dose level 1, or 50 x 106 CAR+ T cells, while 14 were treated at dose level 2, or 100 x 106 CAR+ T cells. Two patients experienced grade 3 or 4 dose-limiting toxicities at the second level, including hypertension in one patient, and encephalopathy, muscle weakness, and tumor lysis syndrome (TLS) in the other.
Cytokine release syndrome (CRS) occurred in 17 patients, though only two cases reached grade 3. Neurologic adverse events were seen in nine patients, of which five were grade 3 or 4.
Partial or complete responses were noted in 81.5%, or 18 of 22 evaluable patients, including 10 (45.5%) who had complete remission. In the subset of nine patients who had failed both ibrutinib and venetoclax, that overall response rate was a “very impressive” 89% (eight of nine patients), said Dr. Siddiqi, including 67% complete remissions (six patients).
Undetectable minimal residual disease (MRD) was reported in 65% and 75% of patients, depending on the method used to evaluate it.
About two-thirds of the patients had responses by day 30 evaluation, and responses deepened over time in about one-quarter, according to Dr. Siddiqi. Of 12 patients with a response at 6 months, 10 (83%) were still in response at 9 months, and 8 patients have been in response for 12 months or longer, she reported.
Neurologic adverse events seen in the CLL/SLL patients in this study were associated with higher lymph node tumor burden, and increased levels of interleukin(IL)-16 or tumor necrosis factor (TNF), according to further analysis presented by Dr. Siddiqi.
That raises the possibility that IL-16 or TNF may be a “good predictive biomarker” for neurotoxicity, which seems to be driven at least in part by lymphadenopathy. “If there was a way that we could combine the CAR T-cell with something like a novel agent that can shrink the tumor burden quickly, then maybe we can have even less toxicities with these CAR T cells,” Dr. Siddiqi said.
Dr. Siddiqi reported disclosures related to Kite, TG Therapeutics, Celgene, Janssen, Seattle Genetics, AstraZeneca, PCYC, Juno Therapeutics, and BeiGene.
SOURCE: Siddiqi T et al. ASH 2019, Abstract 503.
REPORTING FROM ASH 2019
LOXO-305: Next-gen BTK inhibitor safe and effective in B-cell malignancies
ORLANDO – A phase 1 trial of the next-generation Bruton tyrosine kinase (BTK) inhibitor LOXO-305 has demonstrated safety and provided evidence of its efficacy in heavily pretreated patients with B-cell malignancies, including some with acquired resistance to other BTK inhibitors and venetoclax, according to an investigator.
The antitumor activity of this highly selective investigational oral BTK inhibitor was significant in patients with chronic lymphocytic leukemia (CLL) and mantle cell lymphoma (MCL), with a rapid onset of action and resolution of lymphocytosis “consistent with effective BTK target inhibition,” said Anthony R. Mato, MD, of the Center for CLL at Memorial Sloan Kettering Cancer Center in New York.
Responses were also seen in patients with BTK C481 mutations, the primary cause of progressive CLL after BTK inhibitor use, Dr. Mato said at the annual meeting of the American Society of Hematology.
The safety and tolerability profile of LOXO-305 is “consistent with highly selective drug design,” with no evidence of off-target effects, he said. “Collectively, these data demonstrate that BTK remains a highly actionable target despite progression on covalent BTK inhibitors.”
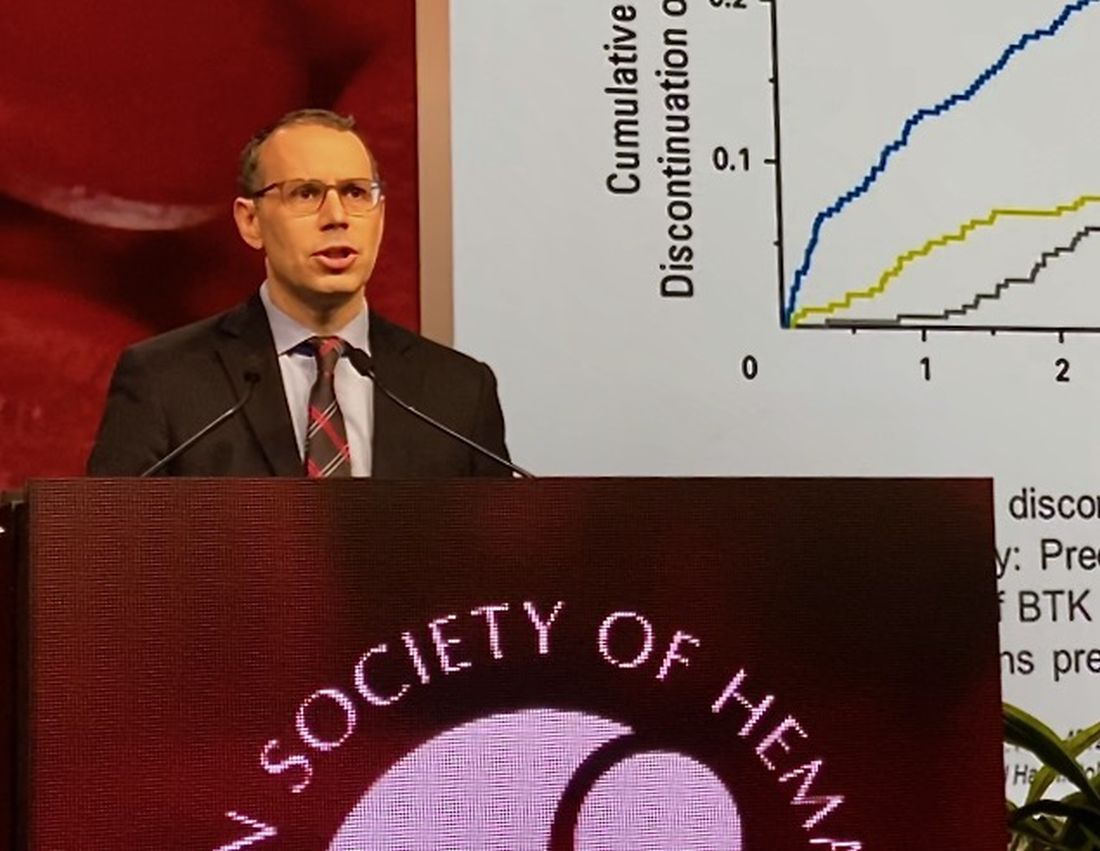
While BTK inhibitors have transformed treatment of B-cell malignancies, resistance remains a major problem, said Dr. Mato, citing 5-year ibrutinib discontinuation rates of 41% in the front line setting and 53.7% in the relapsed/refractory setting.
Key reasons for discontinuation are intolerance, events such as atrial fibrillation and major bleeding, progression of disease, and the appearance of BTK C481 mutations, which prevent covalent BTK inhibitors from achieving effective target inhibition, he said. In contrast, LOXO-305 is designed to non-covalently bind to BTK, regardless of C481 status.
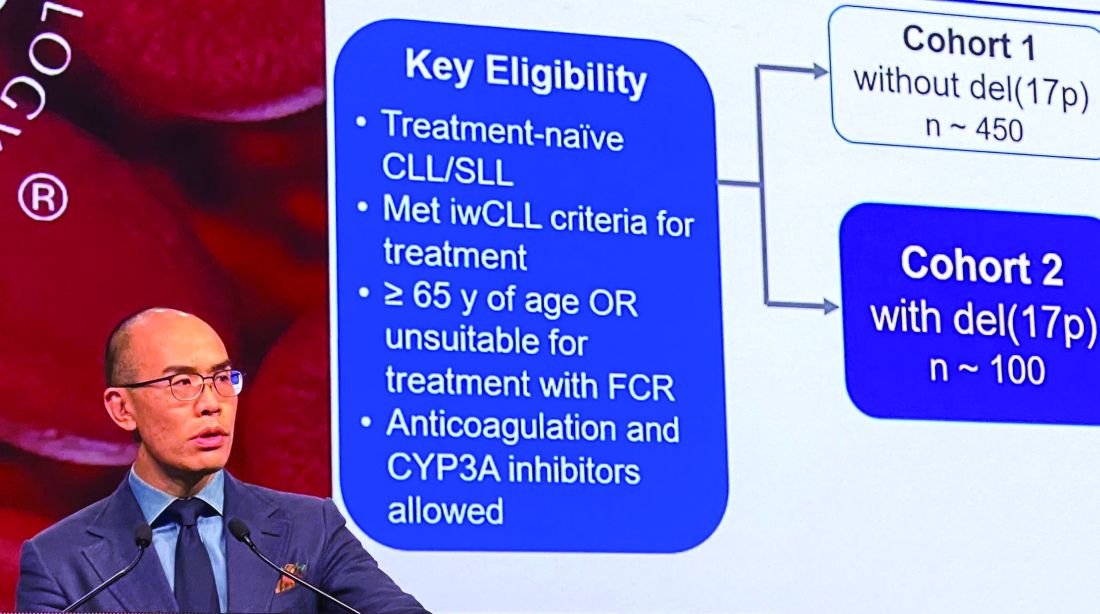
Dr. Mato described results of the phase 1 BRUIN trial, in which 28 adult patients with CLL or B-cell non-Hodgkin lymphomas received once daily oral LOXO-305 at doses ranging from 25 mg to 200 mg. All patients had received at least two lines of prior therapy and had active disease in need of treatment.
For 13 evaluable CLL patients, the overall response rate was 77% (10 patients), Dr. Mato reported. Overall response rates for MCL and other B-cell malignancies were 50%, or three out of six MCL patients and two of four patients with Waldenström macroglobulinemia, diffuse large B-cell lymphoma, or marginal zone lymphoma.
Though only a small subset of CLL patients have had multiple response assessments, the available data suggest that responses “deepen over time” with continued LOXO-305 treatment, Dr. Mato said.
With the median follow-up of 2.7 months, 24 of 28 patients remain on therapy, including all responders. “Some of the responses appear to be quite durable,” Dr. Mato said.
There have been no dose-limiting toxicities, the maximum tolerated dose has not been reached, and there have been no notable adverse events characteristic of covalent BTK inhibitors – namely atrial fibrillation or major bleeding – despite frequent monitoring, according to Dr. Mato.
There were two grade 3 events (leukocytosis and neutropenia), but the remaining treatment-emergent adverse events have been grade 1-2. “Having managed many of these patients, I can tell you that these adverse events were quite manageable,” Dr. Mato told attendees.
BRUIN is a global trial that continues to enroll patients at 18 sites in 3 countries, with a plan in 2020 to incorporate “rational combinations” of agents, according to the investigator.
Dr. Mato reported disclosures related to Gilead, Pharmacyclics, AstraZeneca, AbbVie, Sunesis, Johnson & Johnson, TG Therapeutics, LOXO, DTRM Biopharma, Genentech, Janssen, Acerta, Pharmacyclics, and Celgene.
SOURCE: Mato AR et al. ASH 2019, Abstract 501.
ORLANDO – A phase 1 trial of the next-generation Bruton tyrosine kinase (BTK) inhibitor LOXO-305 has demonstrated safety and provided evidence of its efficacy in heavily pretreated patients with B-cell malignancies, including some with acquired resistance to other BTK inhibitors and venetoclax, according to an investigator.
The antitumor activity of this highly selective investigational oral BTK inhibitor was significant in patients with chronic lymphocytic leukemia (CLL) and mantle cell lymphoma (MCL), with a rapid onset of action and resolution of lymphocytosis “consistent with effective BTK target inhibition,” said Anthony R. Mato, MD, of the Center for CLL at Memorial Sloan Kettering Cancer Center in New York.
Responses were also seen in patients with BTK C481 mutations, the primary cause of progressive CLL after BTK inhibitor use, Dr. Mato said at the annual meeting of the American Society of Hematology.
The safety and tolerability profile of LOXO-305 is “consistent with highly selective drug design,” with no evidence of off-target effects, he said. “Collectively, these data demonstrate that BTK remains a highly actionable target despite progression on covalent BTK inhibitors.”
While BTK inhibitors have transformed treatment of B-cell malignancies, resistance remains a major problem, said Dr. Mato, citing 5-year ibrutinib discontinuation rates of 41% in the front line setting and 53.7% in the relapsed/refractory setting.
Key reasons for discontinuation are intolerance, events such as atrial fibrillation and major bleeding, progression of disease, and the appearance of BTK C481 mutations, which prevent covalent BTK inhibitors from achieving effective target inhibition, he said. In contrast, LOXO-305 is designed to non-covalently bind to BTK, regardless of C481 status.
Dr. Mato described results of the phase 1 BRUIN trial, in which 28 adult patients with CLL or B-cell non-Hodgkin lymphomas received once daily oral LOXO-305 at doses ranging from 25 mg to 200 mg. All patients had received at least two lines of prior therapy and had active disease in need of treatment.
For 13 evaluable CLL patients, the overall response rate was 77% (10 patients), Dr. Mato reported. Overall response rates for MCL and other B-cell malignancies were 50%, or three out of six MCL patients and two of four patients with Waldenström macroglobulinemia, diffuse large B-cell lymphoma, or marginal zone lymphoma.
Though only a small subset of CLL patients have had multiple response assessments, the available data suggest that responses “deepen over time” with continued LOXO-305 treatment, Dr. Mato said.
With the median follow-up of 2.7 months, 24 of 28 patients remain on therapy, including all responders. “Some of the responses appear to be quite durable,” Dr. Mato said.
There have been no dose-limiting toxicities, the maximum tolerated dose has not been reached, and there have been no notable adverse events characteristic of covalent BTK inhibitors – namely atrial fibrillation or major bleeding – despite frequent monitoring, according to Dr. Mato.
There were two grade 3 events (leukocytosis and neutropenia), but the remaining treatment-emergent adverse events have been grade 1-2. “Having managed many of these patients, I can tell you that these adverse events were quite manageable,” Dr. Mato told attendees.
BRUIN is a global trial that continues to enroll patients at 18 sites in 3 countries, with a plan in 2020 to incorporate “rational combinations” of agents, according to the investigator.
Dr. Mato reported disclosures related to Gilead, Pharmacyclics, AstraZeneca, AbbVie, Sunesis, Johnson & Johnson, TG Therapeutics, LOXO, DTRM Biopharma, Genentech, Janssen, Acerta, Pharmacyclics, and Celgene.
SOURCE: Mato AR et al. ASH 2019, Abstract 501.
ORLANDO – A phase 1 trial of the next-generation Bruton tyrosine kinase (BTK) inhibitor LOXO-305 has demonstrated safety and provided evidence of its efficacy in heavily pretreated patients with B-cell malignancies, including some with acquired resistance to other BTK inhibitors and venetoclax, according to an investigator.
The antitumor activity of this highly selective investigational oral BTK inhibitor was significant in patients with chronic lymphocytic leukemia (CLL) and mantle cell lymphoma (MCL), with a rapid onset of action and resolution of lymphocytosis “consistent with effective BTK target inhibition,” said Anthony R. Mato, MD, of the Center for CLL at Memorial Sloan Kettering Cancer Center in New York.
Responses were also seen in patients with BTK C481 mutations, the primary cause of progressive CLL after BTK inhibitor use, Dr. Mato said at the annual meeting of the American Society of Hematology.
The safety and tolerability profile of LOXO-305 is “consistent with highly selective drug design,” with no evidence of off-target effects, he said. “Collectively, these data demonstrate that BTK remains a highly actionable target despite progression on covalent BTK inhibitors.”
While BTK inhibitors have transformed treatment of B-cell malignancies, resistance remains a major problem, said Dr. Mato, citing 5-year ibrutinib discontinuation rates of 41% in the front line setting and 53.7% in the relapsed/refractory setting.
Key reasons for discontinuation are intolerance, events such as atrial fibrillation and major bleeding, progression of disease, and the appearance of BTK C481 mutations, which prevent covalent BTK inhibitors from achieving effective target inhibition, he said. In contrast, LOXO-305 is designed to non-covalently bind to BTK, regardless of C481 status.
Dr. Mato described results of the phase 1 BRUIN trial, in which 28 adult patients with CLL or B-cell non-Hodgkin lymphomas received once daily oral LOXO-305 at doses ranging from 25 mg to 200 mg. All patients had received at least two lines of prior therapy and had active disease in need of treatment.
For 13 evaluable CLL patients, the overall response rate was 77% (10 patients), Dr. Mato reported. Overall response rates for MCL and other B-cell malignancies were 50%, or three out of six MCL patients and two of four patients with Waldenström macroglobulinemia, diffuse large B-cell lymphoma, or marginal zone lymphoma.
Though only a small subset of CLL patients have had multiple response assessments, the available data suggest that responses “deepen over time” with continued LOXO-305 treatment, Dr. Mato said.
With the median follow-up of 2.7 months, 24 of 28 patients remain on therapy, including all responders. “Some of the responses appear to be quite durable,” Dr. Mato said.
There have been no dose-limiting toxicities, the maximum tolerated dose has not been reached, and there have been no notable adverse events characteristic of covalent BTK inhibitors – namely atrial fibrillation or major bleeding – despite frequent monitoring, according to Dr. Mato.
There were two grade 3 events (leukocytosis and neutropenia), but the remaining treatment-emergent adverse events have been grade 1-2. “Having managed many of these patients, I can tell you that these adverse events were quite manageable,” Dr. Mato told attendees.
BRUIN is a global trial that continues to enroll patients at 18 sites in 3 countries, with a plan in 2020 to incorporate “rational combinations” of agents, according to the investigator.
Dr. Mato reported disclosures related to Gilead, Pharmacyclics, AstraZeneca, AbbVie, Sunesis, Johnson & Johnson, TG Therapeutics, LOXO, DTRM Biopharma, Genentech, Janssen, Acerta, Pharmacyclics, and Celgene.
SOURCE: Mato AR et al. ASH 2019, Abstract 501.
REPORTING FROM ASH 2019
Survival data reported from largest CAR T trial in B-cell lymphoma
ORLANDO – Updated results from the TRANSCEND NHL trial include survival data with lisocabtagene maraleucel (liso-cel), an anti-CD19 chimeric antigen receptor (CAR) T-cell therapy, in patients with relapsed/refractory B-cell lymphomas.
The median progression-free survival (PFS) was 6.8 months, and the median overall survival was 21.1 months. PFS results were best among complete responders and among patients with primary mediastinal large B-cell lymphoma or transformed follicular lymphoma.
Jeremy S. Abramson, MD, of Massachusetts General Hospital in Boston, presented these results at the annual meeting of the American Society of Hematology.
“TRANSCEND NHL is the largest clinical study to date of CD19-directed CAR T cells in patients with relapsed/refractory aggressive B-cell lymphoma,” Dr. Abramson said.
The phase 1 trial (NCT02631044) includes 269 patients who received liso-cel. They were diagnosed with transformed follicular lymphoma (22%) or other indolent lymphoma (7%), high-grade B-cell lymphoma (13%), primary mediastinal large B-cell lymphoma (6%), grade 3B follicular lymphoma (1%), or diffuse large B-cell lymphoma not otherwise specified (51%).
At baseline, patients had received a median of three prior systemic therapies (range, one to eight). Some patients had received autologous (33%) or allogeneic (3%) transplant. Many patients were chemotherapy refractory (67%) or had never achieved a complete response to prior therapy (44%).
More than half of patients (59%) received bridging therapy during liso-cel manufacturing. All patients received lymphodepletion with fludarabine and cyclophosphamide, followed by liso-cel at 50 x 106 CAR T cells, 100 x 106 CAR T cells, or 150 x 106 CAR T cells.
Response and survival
The median follow-up was 12.0 months. The overall response rate was 73%, and the complete response rate was 53%.
“Remissions were rapid, with a median of 1 month from CAR T-cell infusion, and durable, with a median duration of response that has not been reached and 55% of patients remaining in response at 1 year,” Dr. Abramson said.
The median PFS was 6.8 months overall, not reached for patients who achieved a complete response, 2.8 months for patients with a partial response, and 1.1 months for patients with stable disease or progressive disease.
The median PFS was not reached for patients with primary mediastinal large B-cell lymphoma or transformed follicular lymphoma, 5.0 months for high-grade B-cell lymphoma, 3.0 months for diffuse large B-cell lymphoma not otherwise specified, and 2.9 months in transformed indolent non-Hodgkin lymphoma.
The median overall survival was 21.1 months overall, not reached for patients who achieved a complete response, 9.0 months for patients who had a partial response, and 5.1 months for patients with stable disease or progressive disease.
Safety
Common treatment-emergent adverse events were neutropenia (63%), anemia (48%), fatigue (44%), nausea (33%), thrombocytopenia (31%), headache (30%), decreased appetite (28%), and diarrhea (26%).
Cytokine release syndrome (CRS) occurred in 42% of patients, and neurologic events occurred in 30%. Grade 3-4 CRS occurred in 2% of patients, and grade 3-4 neurologic events occurred in 10%. There were no cases of grade 5 CRS or neurologic events.
The median time to CRS onset was 5 days, and the median time to onset of neurologic events was 9 days. The median time to resolution of CRS and neurologic events was 5 days and 11 days, respectively.
“The low incidence of severe CRS and neurologic events and their late time of onset support using this product in a large range of patients and in the outpatient setting,” Dr. Abramson said.
There were seven grade 5 treatment-related adverse events, including diffuse alveolar damage, pulmonary hemorrhage, multiple organ dysfunction syndrome, cardiomyopathy, fludarabine leukoencephalopathy, septic shock, and progressive multifocal leukoencephalopathy.
This trial is sponsored by Bristol-Myers Squibb. Dr. Abramson reported relationships with Juno Therapeutics and Celgene, now owned by Bristol-Myers Squibb, and a range of other companies.
SOURCE: Abramson JS et al. ASH 2019, Abstract 241.
ORLANDO – Updated results from the TRANSCEND NHL trial include survival data with lisocabtagene maraleucel (liso-cel), an anti-CD19 chimeric antigen receptor (CAR) T-cell therapy, in patients with relapsed/refractory B-cell lymphomas.
The median progression-free survival (PFS) was 6.8 months, and the median overall survival was 21.1 months. PFS results were best among complete responders and among patients with primary mediastinal large B-cell lymphoma or transformed follicular lymphoma.
Jeremy S. Abramson, MD, of Massachusetts General Hospital in Boston, presented these results at the annual meeting of the American Society of Hematology.
“TRANSCEND NHL is the largest clinical study to date of CD19-directed CAR T cells in patients with relapsed/refractory aggressive B-cell lymphoma,” Dr. Abramson said.
The phase 1 trial (NCT02631044) includes 269 patients who received liso-cel. They were diagnosed with transformed follicular lymphoma (22%) or other indolent lymphoma (7%), high-grade B-cell lymphoma (13%), primary mediastinal large B-cell lymphoma (6%), grade 3B follicular lymphoma (1%), or diffuse large B-cell lymphoma not otherwise specified (51%).
At baseline, patients had received a median of three prior systemic therapies (range, one to eight). Some patients had received autologous (33%) or allogeneic (3%) transplant. Many patients were chemotherapy refractory (67%) or had never achieved a complete response to prior therapy (44%).
More than half of patients (59%) received bridging therapy during liso-cel manufacturing. All patients received lymphodepletion with fludarabine and cyclophosphamide, followed by liso-cel at 50 x 106 CAR T cells, 100 x 106 CAR T cells, or 150 x 106 CAR T cells.
Response and survival
The median follow-up was 12.0 months. The overall response rate was 73%, and the complete response rate was 53%.
“Remissions were rapid, with a median of 1 month from CAR T-cell infusion, and durable, with a median duration of response that has not been reached and 55% of patients remaining in response at 1 year,” Dr. Abramson said.
The median PFS was 6.8 months overall, not reached for patients who achieved a complete response, 2.8 months for patients with a partial response, and 1.1 months for patients with stable disease or progressive disease.
The median PFS was not reached for patients with primary mediastinal large B-cell lymphoma or transformed follicular lymphoma, 5.0 months for high-grade B-cell lymphoma, 3.0 months for diffuse large B-cell lymphoma not otherwise specified, and 2.9 months in transformed indolent non-Hodgkin lymphoma.
The median overall survival was 21.1 months overall, not reached for patients who achieved a complete response, 9.0 months for patients who had a partial response, and 5.1 months for patients with stable disease or progressive disease.
Safety
Common treatment-emergent adverse events were neutropenia (63%), anemia (48%), fatigue (44%), nausea (33%), thrombocytopenia (31%), headache (30%), decreased appetite (28%), and diarrhea (26%).
Cytokine release syndrome (CRS) occurred in 42% of patients, and neurologic events occurred in 30%. Grade 3-4 CRS occurred in 2% of patients, and grade 3-4 neurologic events occurred in 10%. There were no cases of grade 5 CRS or neurologic events.
The median time to CRS onset was 5 days, and the median time to onset of neurologic events was 9 days. The median time to resolution of CRS and neurologic events was 5 days and 11 days, respectively.
“The low incidence of severe CRS and neurologic events and their late time of onset support using this product in a large range of patients and in the outpatient setting,” Dr. Abramson said.
There were seven grade 5 treatment-related adverse events, including diffuse alveolar damage, pulmonary hemorrhage, multiple organ dysfunction syndrome, cardiomyopathy, fludarabine leukoencephalopathy, septic shock, and progressive multifocal leukoencephalopathy.
This trial is sponsored by Bristol-Myers Squibb. Dr. Abramson reported relationships with Juno Therapeutics and Celgene, now owned by Bristol-Myers Squibb, and a range of other companies.
SOURCE: Abramson JS et al. ASH 2019, Abstract 241.
ORLANDO – Updated results from the TRANSCEND NHL trial include survival data with lisocabtagene maraleucel (liso-cel), an anti-CD19 chimeric antigen receptor (CAR) T-cell therapy, in patients with relapsed/refractory B-cell lymphomas.
The median progression-free survival (PFS) was 6.8 months, and the median overall survival was 21.1 months. PFS results were best among complete responders and among patients with primary mediastinal large B-cell lymphoma or transformed follicular lymphoma.
Jeremy S. Abramson, MD, of Massachusetts General Hospital in Boston, presented these results at the annual meeting of the American Society of Hematology.
“TRANSCEND NHL is the largest clinical study to date of CD19-directed CAR T cells in patients with relapsed/refractory aggressive B-cell lymphoma,” Dr. Abramson said.
The phase 1 trial (NCT02631044) includes 269 patients who received liso-cel. They were diagnosed with transformed follicular lymphoma (22%) or other indolent lymphoma (7%), high-grade B-cell lymphoma (13%), primary mediastinal large B-cell lymphoma (6%), grade 3B follicular lymphoma (1%), or diffuse large B-cell lymphoma not otherwise specified (51%).
At baseline, patients had received a median of three prior systemic therapies (range, one to eight). Some patients had received autologous (33%) or allogeneic (3%) transplant. Many patients were chemotherapy refractory (67%) or had never achieved a complete response to prior therapy (44%).
More than half of patients (59%) received bridging therapy during liso-cel manufacturing. All patients received lymphodepletion with fludarabine and cyclophosphamide, followed by liso-cel at 50 x 106 CAR T cells, 100 x 106 CAR T cells, or 150 x 106 CAR T cells.
Response and survival
The median follow-up was 12.0 months. The overall response rate was 73%, and the complete response rate was 53%.
“Remissions were rapid, with a median of 1 month from CAR T-cell infusion, and durable, with a median duration of response that has not been reached and 55% of patients remaining in response at 1 year,” Dr. Abramson said.
The median PFS was 6.8 months overall, not reached for patients who achieved a complete response, 2.8 months for patients with a partial response, and 1.1 months for patients with stable disease or progressive disease.
The median PFS was not reached for patients with primary mediastinal large B-cell lymphoma or transformed follicular lymphoma, 5.0 months for high-grade B-cell lymphoma, 3.0 months for diffuse large B-cell lymphoma not otherwise specified, and 2.9 months in transformed indolent non-Hodgkin lymphoma.
The median overall survival was 21.1 months overall, not reached for patients who achieved a complete response, 9.0 months for patients who had a partial response, and 5.1 months for patients with stable disease or progressive disease.
Safety
Common treatment-emergent adverse events were neutropenia (63%), anemia (48%), fatigue (44%), nausea (33%), thrombocytopenia (31%), headache (30%), decreased appetite (28%), and diarrhea (26%).
Cytokine release syndrome (CRS) occurred in 42% of patients, and neurologic events occurred in 30%. Grade 3-4 CRS occurred in 2% of patients, and grade 3-4 neurologic events occurred in 10%. There were no cases of grade 5 CRS or neurologic events.
The median time to CRS onset was 5 days, and the median time to onset of neurologic events was 9 days. The median time to resolution of CRS and neurologic events was 5 days and 11 days, respectively.
“The low incidence of severe CRS and neurologic events and their late time of onset support using this product in a large range of patients and in the outpatient setting,” Dr. Abramson said.
There were seven grade 5 treatment-related adverse events, including diffuse alveolar damage, pulmonary hemorrhage, multiple organ dysfunction syndrome, cardiomyopathy, fludarabine leukoencephalopathy, septic shock, and progressive multifocal leukoencephalopathy.
This trial is sponsored by Bristol-Myers Squibb. Dr. Abramson reported relationships with Juno Therapeutics and Celgene, now owned by Bristol-Myers Squibb, and a range of other companies.
SOURCE: Abramson JS et al. ASH 2019, Abstract 241.
REPORTING FROM ASH 2019
Zanubrutinib achieved high response rate in del(17p) CLL cohort
ORLANDO – Zanubrutinib has produced a high overall response rate in one the largest cohorts of patients with treatment-naive 17p-deletion chronic lymphocytic leukemia (CLL) studied to date.
An overall response rate of nearly 93% was seen in this 109-patient, high-risk cohort, enrolled as part of the phase 3 SEQUOIA study (BGB-3111-304), said Constantine S. Tam, MBBS, MD, of St. Vincent’s Hospital and Peter MacCallum Cancer Centre in Melbourne.
Tolerability of zanubrutinib was essentially consistent with previous reports of the agent as used in other B-cell malignancies, Dr. Tam said in an oral presentation of the results at the annual meeting of the American Society of Hematology.
Deletion of chromosome 17p13.1, or del(17p), is a marker of poor prognosis and poor response to chemotherapy in patients with CLL or small lymphocytic lymphoma (SLL). For patients with del(17p) CLL, the first-generation Bruton tyrosine kinase (BTK) inhibitor ibrutinib has become a standard of care, Dr. Tam said.
Zanubrutinib, a next-generation BTK inhibitor, was developed to improve BTK occupancy and minimize off-target inhibition of TEC and epidermal growth factor receptor kinases. “What this effectively means is that we are able to dose this drug at levels much higher than that achievable with ibrutinib, and not get intolerable side effects,” Dr. Tam said.
Zanubrutinib has been approved in the United States for previously treated mantle cell lymphoma, and generated durable responses among CLL/SLL patients with or without del(17p) in a phase 1/2 study, according to Dr. Tam.
In the present study, which exclusively enrolled patients with del(17p) CLL/SLL, patients received 160 mg twice daily of zanubrutinib, Dr. Tam said. Out of 109 patients enrolled, 10 (9.2%) had SLL. All patients were aged at least 65 years or were deemed unsuitable for treatment with the combination of fludarabine, cyclophosphamide, and rituximab.
Of 109 patients enrolled, 104 received on-study treatment. The median age was 70 years, Dr. Tam reported, and a number of patients had other high-risk markers beyond del(17p), including unmutated IgVH status in 61.5% of patients.
With a median follow-up of 10 months, the overall response rate was 92.7%, including 1.9% complete responses and 78.9% partial responses. “Only one patient had primary progressive disease after starting this drug,” Dr. Tam said.
Time to response was rapid, according to the investigator, at about 2.8 months; after 6 months, 95% of responders remained in response.
Further analysis showed that the response rate was consistent across subgroups. “There was not a single group that did not respond with a high response rate, including poor prognostic groups,” Dr. Tam said.
Most adverse events were grade 1-2 in severity, and the most common events included confusion and upper respiratory tract infection. The only common grade 3 event, according to Dr. Tam, was neutropenia. Rates of grade 3 major bleeding were low, he said, and the rate of grade 3 atrial fibrillation was 0.9%. One patient died due to pneumonia.
The ongoing SEQUOIA study, designed to compare zanubrutinib to the combination of bendamustine and rituximab in patients with previously untreated CLL or SLL, is sponsored by BeiGene. Dr. Tam reported disclosures related to Novartis, Pharmacyclics, AbbVie, BeiGene, Janssen, and Roche.
SOURCE: Tam C et al. ASH 2019, Abstract 499.
ORLANDO – Zanubrutinib has produced a high overall response rate in one the largest cohorts of patients with treatment-naive 17p-deletion chronic lymphocytic leukemia (CLL) studied to date.
An overall response rate of nearly 93% was seen in this 109-patient, high-risk cohort, enrolled as part of the phase 3 SEQUOIA study (BGB-3111-304), said Constantine S. Tam, MBBS, MD, of St. Vincent’s Hospital and Peter MacCallum Cancer Centre in Melbourne.
Tolerability of zanubrutinib was essentially consistent with previous reports of the agent as used in other B-cell malignancies, Dr. Tam said in an oral presentation of the results at the annual meeting of the American Society of Hematology.
Deletion of chromosome 17p13.1, or del(17p), is a marker of poor prognosis and poor response to chemotherapy in patients with CLL or small lymphocytic lymphoma (SLL). For patients with del(17p) CLL, the first-generation Bruton tyrosine kinase (BTK) inhibitor ibrutinib has become a standard of care, Dr. Tam said.
Zanubrutinib, a next-generation BTK inhibitor, was developed to improve BTK occupancy and minimize off-target inhibition of TEC and epidermal growth factor receptor kinases. “What this effectively means is that we are able to dose this drug at levels much higher than that achievable with ibrutinib, and not get intolerable side effects,” Dr. Tam said.
Zanubrutinib has been approved in the United States for previously treated mantle cell lymphoma, and generated durable responses among CLL/SLL patients with or without del(17p) in a phase 1/2 study, according to Dr. Tam.
In the present study, which exclusively enrolled patients with del(17p) CLL/SLL, patients received 160 mg twice daily of zanubrutinib, Dr. Tam said. Out of 109 patients enrolled, 10 (9.2%) had SLL. All patients were aged at least 65 years or were deemed unsuitable for treatment with the combination of fludarabine, cyclophosphamide, and rituximab.
Of 109 patients enrolled, 104 received on-study treatment. The median age was 70 years, Dr. Tam reported, and a number of patients had other high-risk markers beyond del(17p), including unmutated IgVH status in 61.5% of patients.
With a median follow-up of 10 months, the overall response rate was 92.7%, including 1.9% complete responses and 78.9% partial responses. “Only one patient had primary progressive disease after starting this drug,” Dr. Tam said.
Time to response was rapid, according to the investigator, at about 2.8 months; after 6 months, 95% of responders remained in response.
Further analysis showed that the response rate was consistent across subgroups. “There was not a single group that did not respond with a high response rate, including poor prognostic groups,” Dr. Tam said.
Most adverse events were grade 1-2 in severity, and the most common events included confusion and upper respiratory tract infection. The only common grade 3 event, according to Dr. Tam, was neutropenia. Rates of grade 3 major bleeding were low, he said, and the rate of grade 3 atrial fibrillation was 0.9%. One patient died due to pneumonia.
The ongoing SEQUOIA study, designed to compare zanubrutinib to the combination of bendamustine and rituximab in patients with previously untreated CLL or SLL, is sponsored by BeiGene. Dr. Tam reported disclosures related to Novartis, Pharmacyclics, AbbVie, BeiGene, Janssen, and Roche.
SOURCE: Tam C et al. ASH 2019, Abstract 499.
ORLANDO – Zanubrutinib has produced a high overall response rate in one the largest cohorts of patients with treatment-naive 17p-deletion chronic lymphocytic leukemia (CLL) studied to date.
An overall response rate of nearly 93% was seen in this 109-patient, high-risk cohort, enrolled as part of the phase 3 SEQUOIA study (BGB-3111-304), said Constantine S. Tam, MBBS, MD, of St. Vincent’s Hospital and Peter MacCallum Cancer Centre in Melbourne.
Tolerability of zanubrutinib was essentially consistent with previous reports of the agent as used in other B-cell malignancies, Dr. Tam said in an oral presentation of the results at the annual meeting of the American Society of Hematology.
Deletion of chromosome 17p13.1, or del(17p), is a marker of poor prognosis and poor response to chemotherapy in patients with CLL or small lymphocytic lymphoma (SLL). For patients with del(17p) CLL, the first-generation Bruton tyrosine kinase (BTK) inhibitor ibrutinib has become a standard of care, Dr. Tam said.
Zanubrutinib, a next-generation BTK inhibitor, was developed to improve BTK occupancy and minimize off-target inhibition of TEC and epidermal growth factor receptor kinases. “What this effectively means is that we are able to dose this drug at levels much higher than that achievable with ibrutinib, and not get intolerable side effects,” Dr. Tam said.
Zanubrutinib has been approved in the United States for previously treated mantle cell lymphoma, and generated durable responses among CLL/SLL patients with or without del(17p) in a phase 1/2 study, according to Dr. Tam.
In the present study, which exclusively enrolled patients with del(17p) CLL/SLL, patients received 160 mg twice daily of zanubrutinib, Dr. Tam said. Out of 109 patients enrolled, 10 (9.2%) had SLL. All patients were aged at least 65 years or were deemed unsuitable for treatment with the combination of fludarabine, cyclophosphamide, and rituximab.
Of 109 patients enrolled, 104 received on-study treatment. The median age was 70 years, Dr. Tam reported, and a number of patients had other high-risk markers beyond del(17p), including unmutated IgVH status in 61.5% of patients.
With a median follow-up of 10 months, the overall response rate was 92.7%, including 1.9% complete responses and 78.9% partial responses. “Only one patient had primary progressive disease after starting this drug,” Dr. Tam said.
Time to response was rapid, according to the investigator, at about 2.8 months; after 6 months, 95% of responders remained in response.
Further analysis showed that the response rate was consistent across subgroups. “There was not a single group that did not respond with a high response rate, including poor prognostic groups,” Dr. Tam said.
Most adverse events were grade 1-2 in severity, and the most common events included confusion and upper respiratory tract infection. The only common grade 3 event, according to Dr. Tam, was neutropenia. Rates of grade 3 major bleeding were low, he said, and the rate of grade 3 atrial fibrillation was 0.9%. One patient died due to pneumonia.
The ongoing SEQUOIA study, designed to compare zanubrutinib to the combination of bendamustine and rituximab in patients with previously untreated CLL or SLL, is sponsored by BeiGene. Dr. Tam reported disclosures related to Novartis, Pharmacyclics, AbbVie, BeiGene, Janssen, and Roche.
SOURCE: Tam C et al. ASH 2019, Abstract 499.
REPORTING FROM ASH 2019
Some MCL patients can safely stop venetoclax-ibrutinib, study suggests
ORLANDO – Updated trial results have revealed durable responses with venetoclax and ibrutinib in patients with mantle cell lymphoma (MCL), allowing some patients to stop treatment.
Five of 24 patients were able to stop treatment after achieving minimal residual disease (MRD)-negative complete responses (CRs). Four of these patients remain in CR at up to 18 months off treatment, although one patient ultimately progressed and died.
“Treatment cessation was feasible for patients in MRD-negative complete responses, raising the prospect of limited-duration, targeted-agent therapy in the management of relapsed/refractory mantle cell lymphoma,” said Sasanka M. Handunnetti, MBBS, of Peter MacCallum Cancer Centre in Melbourne. Dr. Handunnetti presented these results, from the AIM trial, at the annual meeting of the American Society of Hematology.
The phase 2 trial enrolled 24 patients. At baseline, patients had a median age of 68 years (range, 47-81 years), and 88% were men. One patient was treatment-naive, but the rest had relapsed/refractory MCL. These patients had received a median of two prior therapies (range, 1-6).
The patients received venetoclax at 400 mg daily and ibrutinib at 560 mg daily.
In the primary analysis, the CR rate was 62% at week 16 and 71% overall, according to positron-emission tomography/computed tomography. MRD negativity was achieved by 67% of patients according to flow cytometry and 38% according to allele-specific oligonucleotide polymerase chain reaction (N Engl J Med. 2018 Mar 29;378[13]:1211-23).
Response and survival
For the current analysis, the median follow up was 37.5 months (range, 1.4-45.3 months). The median duration of response has not been reached, the median progression-free survival is 29 months, and the median overall survival is 32 months.
Thirteen patients have died, 8 of them due to progressive disease. The remaining 11 patients are still alive, and 9 of them are still in CR. One patient is still in partial response, and one has not responded but remains on ibrutinib and venetoclax.
Dr. Handunnetti pointed out that 12 patients had TP53 aberrations, and 8 of them died, but 4 remain alive and in CR. All four patients with SMARCA4 aberrations died.
Treatment status
Five patients are still receiving treatment with ibrutinib and venetoclax, and one patient is receiving only venetoclax. One patient went off study treatment due to a diagnosis of myelodysplastic syndrome, but that patient’s MCL is still in CR.
Five patients were able to stop treatment after achieving MRD-negative CR and were placed under “stringent surveillance,” Dr. Handunnetti said.
One of the five patients who stopped treatment progressed at 7 months and died. The remaining four patients are still alive and in CR at 6 months, 13 months, 17 months, and 18 months off treatment.
Safety update
Within the first 56 weeks of treatment, 15 patients required dose adjustments. Twelve patients required an adjustment to ibrutinib, seven to venetoclax, and four to both drugs. After 56 weeks, there were no dose adjustments.
Two patients developed therapy-related myelodysplastic syndrome. One patient had previously received FCR (fludarabine, cyclophosphamide, and rituximab) and BR (bendamustine and rituximab). The other patient had received R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone).
This investigator-initiated trial was funded by Janssen and Abbvie. Dr. Handunnetti reported relationships with Abbvie and Gilead.
SOURCE: Handunnetti S et al. ASH 2019. Abstract 756.
ORLANDO – Updated trial results have revealed durable responses with venetoclax and ibrutinib in patients with mantle cell lymphoma (MCL), allowing some patients to stop treatment.
Five of 24 patients were able to stop treatment after achieving minimal residual disease (MRD)-negative complete responses (CRs). Four of these patients remain in CR at up to 18 months off treatment, although one patient ultimately progressed and died.
“Treatment cessation was feasible for patients in MRD-negative complete responses, raising the prospect of limited-duration, targeted-agent therapy in the management of relapsed/refractory mantle cell lymphoma,” said Sasanka M. Handunnetti, MBBS, of Peter MacCallum Cancer Centre in Melbourne. Dr. Handunnetti presented these results, from the AIM trial, at the annual meeting of the American Society of Hematology.
The phase 2 trial enrolled 24 patients. At baseline, patients had a median age of 68 years (range, 47-81 years), and 88% were men. One patient was treatment-naive, but the rest had relapsed/refractory MCL. These patients had received a median of two prior therapies (range, 1-6).
The patients received venetoclax at 400 mg daily and ibrutinib at 560 mg daily.
In the primary analysis, the CR rate was 62% at week 16 and 71% overall, according to positron-emission tomography/computed tomography. MRD negativity was achieved by 67% of patients according to flow cytometry and 38% according to allele-specific oligonucleotide polymerase chain reaction (N Engl J Med. 2018 Mar 29;378[13]:1211-23).
Response and survival
For the current analysis, the median follow up was 37.5 months (range, 1.4-45.3 months). The median duration of response has not been reached, the median progression-free survival is 29 months, and the median overall survival is 32 months.
Thirteen patients have died, 8 of them due to progressive disease. The remaining 11 patients are still alive, and 9 of them are still in CR. One patient is still in partial response, and one has not responded but remains on ibrutinib and venetoclax.
Dr. Handunnetti pointed out that 12 patients had TP53 aberrations, and 8 of them died, but 4 remain alive and in CR. All four patients with SMARCA4 aberrations died.
Treatment status
Five patients are still receiving treatment with ibrutinib and venetoclax, and one patient is receiving only venetoclax. One patient went off study treatment due to a diagnosis of myelodysplastic syndrome, but that patient’s MCL is still in CR.
Five patients were able to stop treatment after achieving MRD-negative CR and were placed under “stringent surveillance,” Dr. Handunnetti said.
One of the five patients who stopped treatment progressed at 7 months and died. The remaining four patients are still alive and in CR at 6 months, 13 months, 17 months, and 18 months off treatment.
Safety update
Within the first 56 weeks of treatment, 15 patients required dose adjustments. Twelve patients required an adjustment to ibrutinib, seven to venetoclax, and four to both drugs. After 56 weeks, there were no dose adjustments.
Two patients developed therapy-related myelodysplastic syndrome. One patient had previously received FCR (fludarabine, cyclophosphamide, and rituximab) and BR (bendamustine and rituximab). The other patient had received R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone).
This investigator-initiated trial was funded by Janssen and Abbvie. Dr. Handunnetti reported relationships with Abbvie and Gilead.
SOURCE: Handunnetti S et al. ASH 2019. Abstract 756.
ORLANDO – Updated trial results have revealed durable responses with venetoclax and ibrutinib in patients with mantle cell lymphoma (MCL), allowing some patients to stop treatment.
Five of 24 patients were able to stop treatment after achieving minimal residual disease (MRD)-negative complete responses (CRs). Four of these patients remain in CR at up to 18 months off treatment, although one patient ultimately progressed and died.
“Treatment cessation was feasible for patients in MRD-negative complete responses, raising the prospect of limited-duration, targeted-agent therapy in the management of relapsed/refractory mantle cell lymphoma,” said Sasanka M. Handunnetti, MBBS, of Peter MacCallum Cancer Centre in Melbourne. Dr. Handunnetti presented these results, from the AIM trial, at the annual meeting of the American Society of Hematology.
The phase 2 trial enrolled 24 patients. At baseline, patients had a median age of 68 years (range, 47-81 years), and 88% were men. One patient was treatment-naive, but the rest had relapsed/refractory MCL. These patients had received a median of two prior therapies (range, 1-6).
The patients received venetoclax at 400 mg daily and ibrutinib at 560 mg daily.
In the primary analysis, the CR rate was 62% at week 16 and 71% overall, according to positron-emission tomography/computed tomography. MRD negativity was achieved by 67% of patients according to flow cytometry and 38% according to allele-specific oligonucleotide polymerase chain reaction (N Engl J Med. 2018 Mar 29;378[13]:1211-23).
Response and survival
For the current analysis, the median follow up was 37.5 months (range, 1.4-45.3 months). The median duration of response has not been reached, the median progression-free survival is 29 months, and the median overall survival is 32 months.
Thirteen patients have died, 8 of them due to progressive disease. The remaining 11 patients are still alive, and 9 of them are still in CR. One patient is still in partial response, and one has not responded but remains on ibrutinib and venetoclax.
Dr. Handunnetti pointed out that 12 patients had TP53 aberrations, and 8 of them died, but 4 remain alive and in CR. All four patients with SMARCA4 aberrations died.
Treatment status
Five patients are still receiving treatment with ibrutinib and venetoclax, and one patient is receiving only venetoclax. One patient went off study treatment due to a diagnosis of myelodysplastic syndrome, but that patient’s MCL is still in CR.
Five patients were able to stop treatment after achieving MRD-negative CR and were placed under “stringent surveillance,” Dr. Handunnetti said.
One of the five patients who stopped treatment progressed at 7 months and died. The remaining four patients are still alive and in CR at 6 months, 13 months, 17 months, and 18 months off treatment.
Safety update
Within the first 56 weeks of treatment, 15 patients required dose adjustments. Twelve patients required an adjustment to ibrutinib, seven to venetoclax, and four to both drugs. After 56 weeks, there were no dose adjustments.
Two patients developed therapy-related myelodysplastic syndrome. One patient had previously received FCR (fludarabine, cyclophosphamide, and rituximab) and BR (bendamustine and rituximab). The other patient had received R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone).
This investigator-initiated trial was funded by Janssen and Abbvie. Dr. Handunnetti reported relationships with Abbvie and Gilead.
SOURCE: Handunnetti S et al. ASH 2019. Abstract 756.
REPORTING FROM ASH 2019
KTE-X19 produces highest response rate in MCL subgroup
ORLANDO – KTE-X19, an anti-CD19 chimeric antigen receptor (CAR) T-cell therapy, demonstrated unprecedented efficacy in the ZUMA-2 trial, according to an investigator involved in the study.
KTE-X19 produced a 93% overall response rate in patients with relapsed/refractory mantle cell lymphoma (MCL). This is the highest reported response rate in patients who have failed treatment with a Bruton’s tyrosine kinase (BTK) inhibitor, said Michael L. Wang, MD, of the University of Texas MD Anderson Cancer Center in Houston.
Dr. Wang presented results from ZUMA-2 at the annual meeting of the American Society of Hematology.
“Patients with relapsed/refractory MCL have very poor outcomes,” Dr. Wang noted. “In patients who progress after BTK inhibition therapy, the overall response rate is only between 25% and 42%, and the overall survival is only between 6 and 10 months. Few patients proceed to allogeneic transplantation.”
The phase 2 ZUMA-2 trial was designed to test KTE-X19 in these patients. KTE-X19 is an anti-CD19 CAR T-cell therapy containing a CD3-zeta T-cell activation domain and a CD28 signaling domain. KTE-X19 is distinct from axicabtagene ciloleucel (KTE-C19) because the manufacturing process for KTE-X19 removes circulating tumor cells.
The trial enrolled 74 patients, and 68 of them received KTE-X19. Manufacturing failed for three patients, two patients died of progressive disease before they could receive KTE-X19, and one patient was found to be ineligible for treatment.
The 68 patients had a median age of 65 years (range, 38-79 years), and 84% were men. A majority of patients (85%) had stage IV disease and classical (59%) or blastoid (25%) morphology. Most patients (69%) had a Ki-67 proliferation index of 50% or greater, and most (56%) were intermediate- or high-risk according to the Mantle Cell Lymphoma International Prognostic Index (MIPI).
Patients had received a median of three prior therapies (range, one to five). All had been treated with a BTK inhibitor, with 85% receiving ibrutinib, 24% receiving acalabrutinib, and 9% receiving both. Most patients (68%) were refractory to BTK inhibition, and 32% relapsed on or after BTK inhibitor therapy.
In this study, patients could receive bridging therapy to keep their disease stable while KTE-X19 was being manufactured. There were 25 patients who received bridging therapy, which consisted of ibrutinib (n = 14), acalabrutinib (n = 5), dexamethasone (n = 12), and/or methylprednisolone (n = 2). Six patients received both BTK inhibitors and steroids.
All patients received conditioning with fludarabine and cyclophosphamide, followed by a single infusion of KTE-X19 at 2x106.
Efficacy
Sixty patients were evaluable for efficacy, and the median follow-up was 12.3 months (range, 7.0-32.3 months).
The overall response rate was 93%, with 67% of patients achieving a complete response and 27% achieving a partial response. Three percent of patients had stable disease, and 3% had progressive disease.
“The overall response rate was consistent across key subgroups, without any statistical difference,” Dr. Wang said. “This includes Ki-67, MIPI, and prior use of either steroids or bridging therapy.”
The median time to response was 1.0 month, and the median time to complete response was 3.0 months. Responses deepened over time, with 35% of patients converting from a partial response to a complete response, and 5% converting from stable disease to complete response.
The median duration of response has not been reached. At last follow-up, 57% of all patients and 78% of complete responders were still in response.
The median progression-free and overall survival have not been reached. At 12 months, the progression-free survival rate was 61%, and the overall survival rate was 83%.
Safety
All 68 patients were evaluable for safety. The most common adverse events were pyrexia (94%), neutropenia (87%), thrombocytopenia (74%), anemia (68%), and hypotension (51%).
Grade 3/4 adverse events included pyrexia (13%), neutropenia (85%), thrombocytopenia (51%), anemia (50%), hypotension (22%), hypoxia (21%), hypophosphatemia (22%), fatigue (1%), and headache (1%).
There were two grade 5 treatment-related adverse events – organizing pneumonia on day 37 and staphylococcal bacteremia on day 134.
Cytokine release syndrome (CRS) occurred in 91% of patients, with 15% experiencing grade 3 or higher CRS. Patients were treated with tocilizumab or corticosteroids, and all CRS events resolved.
Neurologic adverse events occurred in 63% of patients, with grade 3 or higher events occurring in 31%. Neurologic events were treated with tocilizumab or corticosteroids, and 86% of neurologic events resolved.
This trial was sponsored by Kite, a Gilead company. Dr. Wang reported financial relationships with Kite and other companies.
SOURCE: Wang M et al. ASH 2019. Abstract 754.
ORLANDO – KTE-X19, an anti-CD19 chimeric antigen receptor (CAR) T-cell therapy, demonstrated unprecedented efficacy in the ZUMA-2 trial, according to an investigator involved in the study.
KTE-X19 produced a 93% overall response rate in patients with relapsed/refractory mantle cell lymphoma (MCL). This is the highest reported response rate in patients who have failed treatment with a Bruton’s tyrosine kinase (BTK) inhibitor, said Michael L. Wang, MD, of the University of Texas MD Anderson Cancer Center in Houston.
Dr. Wang presented results from ZUMA-2 at the annual meeting of the American Society of Hematology.
“Patients with relapsed/refractory MCL have very poor outcomes,” Dr. Wang noted. “In patients who progress after BTK inhibition therapy, the overall response rate is only between 25% and 42%, and the overall survival is only between 6 and 10 months. Few patients proceed to allogeneic transplantation.”
The phase 2 ZUMA-2 trial was designed to test KTE-X19 in these patients. KTE-X19 is an anti-CD19 CAR T-cell therapy containing a CD3-zeta T-cell activation domain and a CD28 signaling domain. KTE-X19 is distinct from axicabtagene ciloleucel (KTE-C19) because the manufacturing process for KTE-X19 removes circulating tumor cells.
The trial enrolled 74 patients, and 68 of them received KTE-X19. Manufacturing failed for three patients, two patients died of progressive disease before they could receive KTE-X19, and one patient was found to be ineligible for treatment.
The 68 patients had a median age of 65 years (range, 38-79 years), and 84% were men. A majority of patients (85%) had stage IV disease and classical (59%) or blastoid (25%) morphology. Most patients (69%) had a Ki-67 proliferation index of 50% or greater, and most (56%) were intermediate- or high-risk according to the Mantle Cell Lymphoma International Prognostic Index (MIPI).
Patients had received a median of three prior therapies (range, one to five). All had been treated with a BTK inhibitor, with 85% receiving ibrutinib, 24% receiving acalabrutinib, and 9% receiving both. Most patients (68%) were refractory to BTK inhibition, and 32% relapsed on or after BTK inhibitor therapy.
In this study, patients could receive bridging therapy to keep their disease stable while KTE-X19 was being manufactured. There were 25 patients who received bridging therapy, which consisted of ibrutinib (n = 14), acalabrutinib (n = 5), dexamethasone (n = 12), and/or methylprednisolone (n = 2). Six patients received both BTK inhibitors and steroids.
All patients received conditioning with fludarabine and cyclophosphamide, followed by a single infusion of KTE-X19 at 2x106.
Efficacy
Sixty patients were evaluable for efficacy, and the median follow-up was 12.3 months (range, 7.0-32.3 months).
The overall response rate was 93%, with 67% of patients achieving a complete response and 27% achieving a partial response. Three percent of patients had stable disease, and 3% had progressive disease.
“The overall response rate was consistent across key subgroups, without any statistical difference,” Dr. Wang said. “This includes Ki-67, MIPI, and prior use of either steroids or bridging therapy.”
The median time to response was 1.0 month, and the median time to complete response was 3.0 months. Responses deepened over time, with 35% of patients converting from a partial response to a complete response, and 5% converting from stable disease to complete response.
The median duration of response has not been reached. At last follow-up, 57% of all patients and 78% of complete responders were still in response.
The median progression-free and overall survival have not been reached. At 12 months, the progression-free survival rate was 61%, and the overall survival rate was 83%.
Safety
All 68 patients were evaluable for safety. The most common adverse events were pyrexia (94%), neutropenia (87%), thrombocytopenia (74%), anemia (68%), and hypotension (51%).
Grade 3/4 adverse events included pyrexia (13%), neutropenia (85%), thrombocytopenia (51%), anemia (50%), hypotension (22%), hypoxia (21%), hypophosphatemia (22%), fatigue (1%), and headache (1%).
There were two grade 5 treatment-related adverse events – organizing pneumonia on day 37 and staphylococcal bacteremia on day 134.
Cytokine release syndrome (CRS) occurred in 91% of patients, with 15% experiencing grade 3 or higher CRS. Patients were treated with tocilizumab or corticosteroids, and all CRS events resolved.
Neurologic adverse events occurred in 63% of patients, with grade 3 or higher events occurring in 31%. Neurologic events were treated with tocilizumab or corticosteroids, and 86% of neurologic events resolved.
This trial was sponsored by Kite, a Gilead company. Dr. Wang reported financial relationships with Kite and other companies.
SOURCE: Wang M et al. ASH 2019. Abstract 754.
ORLANDO – KTE-X19, an anti-CD19 chimeric antigen receptor (CAR) T-cell therapy, demonstrated unprecedented efficacy in the ZUMA-2 trial, according to an investigator involved in the study.
KTE-X19 produced a 93% overall response rate in patients with relapsed/refractory mantle cell lymphoma (MCL). This is the highest reported response rate in patients who have failed treatment with a Bruton’s tyrosine kinase (BTK) inhibitor, said Michael L. Wang, MD, of the University of Texas MD Anderson Cancer Center in Houston.
Dr. Wang presented results from ZUMA-2 at the annual meeting of the American Society of Hematology.
“Patients with relapsed/refractory MCL have very poor outcomes,” Dr. Wang noted. “In patients who progress after BTK inhibition therapy, the overall response rate is only between 25% and 42%, and the overall survival is only between 6 and 10 months. Few patients proceed to allogeneic transplantation.”
The phase 2 ZUMA-2 trial was designed to test KTE-X19 in these patients. KTE-X19 is an anti-CD19 CAR T-cell therapy containing a CD3-zeta T-cell activation domain and a CD28 signaling domain. KTE-X19 is distinct from axicabtagene ciloleucel (KTE-C19) because the manufacturing process for KTE-X19 removes circulating tumor cells.
The trial enrolled 74 patients, and 68 of them received KTE-X19. Manufacturing failed for three patients, two patients died of progressive disease before they could receive KTE-X19, and one patient was found to be ineligible for treatment.
The 68 patients had a median age of 65 years (range, 38-79 years), and 84% were men. A majority of patients (85%) had stage IV disease and classical (59%) or blastoid (25%) morphology. Most patients (69%) had a Ki-67 proliferation index of 50% or greater, and most (56%) were intermediate- or high-risk according to the Mantle Cell Lymphoma International Prognostic Index (MIPI).
Patients had received a median of three prior therapies (range, one to five). All had been treated with a BTK inhibitor, with 85% receiving ibrutinib, 24% receiving acalabrutinib, and 9% receiving both. Most patients (68%) were refractory to BTK inhibition, and 32% relapsed on or after BTK inhibitor therapy.
In this study, patients could receive bridging therapy to keep their disease stable while KTE-X19 was being manufactured. There were 25 patients who received bridging therapy, which consisted of ibrutinib (n = 14), acalabrutinib (n = 5), dexamethasone (n = 12), and/or methylprednisolone (n = 2). Six patients received both BTK inhibitors and steroids.
All patients received conditioning with fludarabine and cyclophosphamide, followed by a single infusion of KTE-X19 at 2x106.
Efficacy
Sixty patients were evaluable for efficacy, and the median follow-up was 12.3 months (range, 7.0-32.3 months).
The overall response rate was 93%, with 67% of patients achieving a complete response and 27% achieving a partial response. Three percent of patients had stable disease, and 3% had progressive disease.
“The overall response rate was consistent across key subgroups, without any statistical difference,” Dr. Wang said. “This includes Ki-67, MIPI, and prior use of either steroids or bridging therapy.”
The median time to response was 1.0 month, and the median time to complete response was 3.0 months. Responses deepened over time, with 35% of patients converting from a partial response to a complete response, and 5% converting from stable disease to complete response.
The median duration of response has not been reached. At last follow-up, 57% of all patients and 78% of complete responders were still in response.
The median progression-free and overall survival have not been reached. At 12 months, the progression-free survival rate was 61%, and the overall survival rate was 83%.
Safety
All 68 patients were evaluable for safety. The most common adverse events were pyrexia (94%), neutropenia (87%), thrombocytopenia (74%), anemia (68%), and hypotension (51%).
Grade 3/4 adverse events included pyrexia (13%), neutropenia (85%), thrombocytopenia (51%), anemia (50%), hypotension (22%), hypoxia (21%), hypophosphatemia (22%), fatigue (1%), and headache (1%).
There were two grade 5 treatment-related adverse events – organizing pneumonia on day 37 and staphylococcal bacteremia on day 134.
Cytokine release syndrome (CRS) occurred in 91% of patients, with 15% experiencing grade 3 or higher CRS. Patients were treated with tocilizumab or corticosteroids, and all CRS events resolved.
Neurologic adverse events occurred in 63% of patients, with grade 3 or higher events occurring in 31%. Neurologic events were treated with tocilizumab or corticosteroids, and 86% of neurologic events resolved.
This trial was sponsored by Kite, a Gilead company. Dr. Wang reported financial relationships with Kite and other companies.
SOURCE: Wang M et al. ASH 2019. Abstract 754.
REPORTING FROM ASH 2019