User login
POSH study: BRCA mutations did not influence survival in young onset breast cancer
For women with young-onset breast cancer, presence of a BRCA mutation did not significantly impact survival, according to results of the Prospective Outcomes in Sporadic versus Hereditary breast cancer (POSH) study.
BRCA-positive and BRCA-negative women had similar overall survival at 2 years, 5 years, and 10 years after diagnosis, according to lead author Ellen R. Copson, MD, a senior lecturer in medical oncology in the cancer sciences division, University of Southampton (England) and her study coauthors.
Currently, young women with breast cancer and a BRCA mutation frequently are offered bilateral mastectomy, the authors noted.
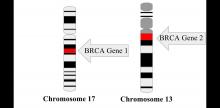
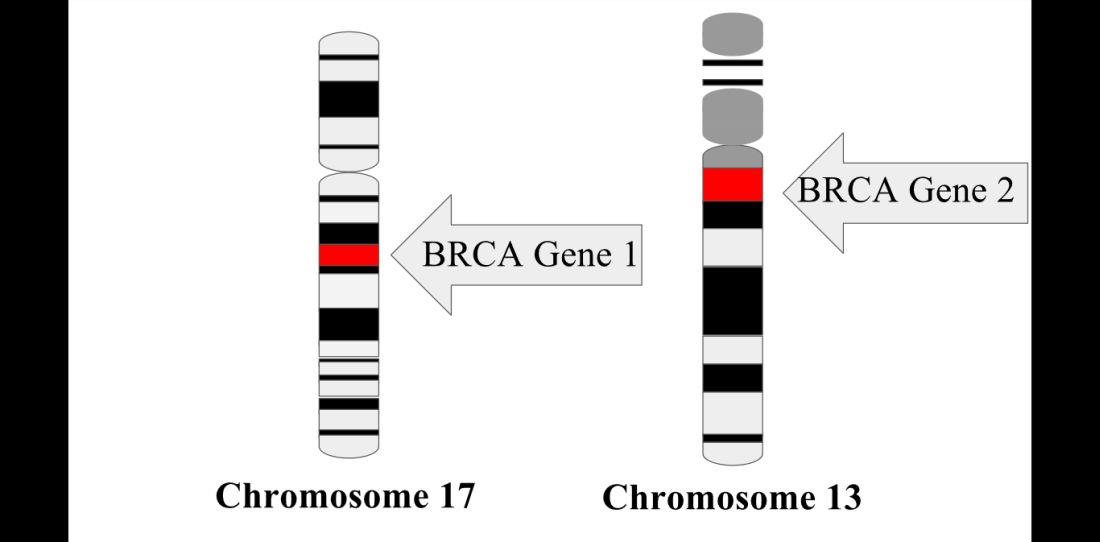
The prospective cohort study by Dr. Copson and her colleagues included 2,733 women with breast cancer who were aged 40 years or younger at first diagnosis. Of those patients, 338 (12%) had either a BRCA1 or BRCA2 mutation, according to investigators.
At 2 years after diagnosis, overall survival was 97.0% and 96.6% for BRCA-positive and BRCA-negative patients, respectively, the report said. Similarly, overall survival was 83.8% and 85.0% for the two groups at 5 years after diagnosis, and 73.4% vs. 70.1% at 10 years.
Multivariable analysis accounting for known prognostic factors including ethnicity and body mass index showed there was no significant difference between groups (hazard ratio, 0.96; 95% confidence interval, 0.76-1.22; P = .76), the authors wrote.
Triple-negative breast cancer patients with a BRCA mutation might have a survival advantage in the first few years following diagnosis,compared with non-BRCA carriers, the POSH study also found. Researchers reported a significant difference at 2 years (95% for BRCA-positive vs. 91% for BRCA-negative patients; P = .047), but there was no significant difference between arms at 5 or 10 years.
POSH is believed to be the largest prospective cohort study to compare breast cancer outcomes for patients with BRCA mutations to those with sporadic breast cancer. Previous studies, primarily retrospective, have suggested “better, worse, or similar outcomes” for BRCA-positive versus BRCA-negative patients, the authors wrote. Dr. Copson reported receiving honoraria from Roche, while her coauthors reported honoraria from GSK, Pfizer, AstraZeneca, and Pierre Fabre. Funding for the study was provided by the Wessex Cancer Trust, Cancer Research UK, and Breast Cancer Now.
SOURCE: Copson et al. Lancet Oncol. 2018 Jan 11 doi: 10.1016/S1470-2045(17)30891-4.
The POSH prospective cohort study, which showed no significant difference in survival for BRCA-positive versus BRCA-negative young onset breast cancer patients, has contributed to the understanding of this patient population, providing “comprehensive data about patient, tumor, and treatment characteristics, along with extensive follow-up data,” wrote Peter A. Fasching, MD, in accompanying editorial.
“Understanding prognosis in young patients is important because patients with BRCA mutations are at increased risk of developing specific conditions, such as secondary cancers,” Dr. Fasching said. “These risks determine treatment, and knowing that BRCA1 or BRCA2 mutations do not result in a different prognosis might change the therapeutic approach for these risks.”
Moreover, in retrospective analyses, bilateral mastectomy conferred an overall survival benefit for BRCA mutation carriers: “This important topic needs more prospective research, as preventive surgical measures might have an effect on what might be a very long life after a diagnosis of breast cancer at a young age,” said Dr. Fasching. “The data from POSH deliver a rationale for prospective studies to address these questions.”
Dr. Peter A. Fasching is with Friedrich-Alexander University Erlangen-Nuremberg, Germany. These comments are based on his editorial appearing in Lancet Oncology (2018 Jan 11. doi: 10.1016/S1470-2045(18)30008-1). Dr. Fasching declared grants from Novartis, along with personal fees from Novartis, Pfizer, Roche, Teva, and Amgen.
The POSH prospective cohort study, which showed no significant difference in survival for BRCA-positive versus BRCA-negative young onset breast cancer patients, has contributed to the understanding of this patient population, providing “comprehensive data about patient, tumor, and treatment characteristics, along with extensive follow-up data,” wrote Peter A. Fasching, MD, in accompanying editorial.
“Understanding prognosis in young patients is important because patients with BRCA mutations are at increased risk of developing specific conditions, such as secondary cancers,” Dr. Fasching said. “These risks determine treatment, and knowing that BRCA1 or BRCA2 mutations do not result in a different prognosis might change the therapeutic approach for these risks.”
Moreover, in retrospective analyses, bilateral mastectomy conferred an overall survival benefit for BRCA mutation carriers: “This important topic needs more prospective research, as preventive surgical measures might have an effect on what might be a very long life after a diagnosis of breast cancer at a young age,” said Dr. Fasching. “The data from POSH deliver a rationale for prospective studies to address these questions.”
Dr. Peter A. Fasching is with Friedrich-Alexander University Erlangen-Nuremberg, Germany. These comments are based on his editorial appearing in Lancet Oncology (2018 Jan 11. doi: 10.1016/S1470-2045(18)30008-1). Dr. Fasching declared grants from Novartis, along with personal fees from Novartis, Pfizer, Roche, Teva, and Amgen.
The POSH prospective cohort study, which showed no significant difference in survival for BRCA-positive versus BRCA-negative young onset breast cancer patients, has contributed to the understanding of this patient population, providing “comprehensive data about patient, tumor, and treatment characteristics, along with extensive follow-up data,” wrote Peter A. Fasching, MD, in accompanying editorial.
“Understanding prognosis in young patients is important because patients with BRCA mutations are at increased risk of developing specific conditions, such as secondary cancers,” Dr. Fasching said. “These risks determine treatment, and knowing that BRCA1 or BRCA2 mutations do not result in a different prognosis might change the therapeutic approach for these risks.”
Moreover, in retrospective analyses, bilateral mastectomy conferred an overall survival benefit for BRCA mutation carriers: “This important topic needs more prospective research, as preventive surgical measures might have an effect on what might be a very long life after a diagnosis of breast cancer at a young age,” said Dr. Fasching. “The data from POSH deliver a rationale for prospective studies to address these questions.”
Dr. Peter A. Fasching is with Friedrich-Alexander University Erlangen-Nuremberg, Germany. These comments are based on his editorial appearing in Lancet Oncology (2018 Jan 11. doi: 10.1016/S1470-2045(18)30008-1). Dr. Fasching declared grants from Novartis, along with personal fees from Novartis, Pfizer, Roche, Teva, and Amgen.
For women with young-onset breast cancer, presence of a BRCA mutation did not significantly impact survival, according to results of the Prospective Outcomes in Sporadic versus Hereditary breast cancer (POSH) study.
BRCA-positive and BRCA-negative women had similar overall survival at 2 years, 5 years, and 10 years after diagnosis, according to lead author Ellen R. Copson, MD, a senior lecturer in medical oncology in the cancer sciences division, University of Southampton (England) and her study coauthors.
Currently, young women with breast cancer and a BRCA mutation frequently are offered bilateral mastectomy, the authors noted.
The prospective cohort study by Dr. Copson and her colleagues included 2,733 women with breast cancer who were aged 40 years or younger at first diagnosis. Of those patients, 338 (12%) had either a BRCA1 or BRCA2 mutation, according to investigators.
At 2 years after diagnosis, overall survival was 97.0% and 96.6% for BRCA-positive and BRCA-negative patients, respectively, the report said. Similarly, overall survival was 83.8% and 85.0% for the two groups at 5 years after diagnosis, and 73.4% vs. 70.1% at 10 years.
Multivariable analysis accounting for known prognostic factors including ethnicity and body mass index showed there was no significant difference between groups (hazard ratio, 0.96; 95% confidence interval, 0.76-1.22; P = .76), the authors wrote.
Triple-negative breast cancer patients with a BRCA mutation might have a survival advantage in the first few years following diagnosis,compared with non-BRCA carriers, the POSH study also found. Researchers reported a significant difference at 2 years (95% for BRCA-positive vs. 91% for BRCA-negative patients; P = .047), but there was no significant difference between arms at 5 or 10 years.
POSH is believed to be the largest prospective cohort study to compare breast cancer outcomes for patients with BRCA mutations to those with sporadic breast cancer. Previous studies, primarily retrospective, have suggested “better, worse, or similar outcomes” for BRCA-positive versus BRCA-negative patients, the authors wrote. Dr. Copson reported receiving honoraria from Roche, while her coauthors reported honoraria from GSK, Pfizer, AstraZeneca, and Pierre Fabre. Funding for the study was provided by the Wessex Cancer Trust, Cancer Research UK, and Breast Cancer Now.
SOURCE: Copson et al. Lancet Oncol. 2018 Jan 11 doi: 10.1016/S1470-2045(17)30891-4.
For women with young-onset breast cancer, presence of a BRCA mutation did not significantly impact survival, according to results of the Prospective Outcomes in Sporadic versus Hereditary breast cancer (POSH) study.
BRCA-positive and BRCA-negative women had similar overall survival at 2 years, 5 years, and 10 years after diagnosis, according to lead author Ellen R. Copson, MD, a senior lecturer in medical oncology in the cancer sciences division, University of Southampton (England) and her study coauthors.
Currently, young women with breast cancer and a BRCA mutation frequently are offered bilateral mastectomy, the authors noted.
The prospective cohort study by Dr. Copson and her colleagues included 2,733 women with breast cancer who were aged 40 years or younger at first diagnosis. Of those patients, 338 (12%) had either a BRCA1 or BRCA2 mutation, according to investigators.
At 2 years after diagnosis, overall survival was 97.0% and 96.6% for BRCA-positive and BRCA-negative patients, respectively, the report said. Similarly, overall survival was 83.8% and 85.0% for the two groups at 5 years after diagnosis, and 73.4% vs. 70.1% at 10 years.
Multivariable analysis accounting for known prognostic factors including ethnicity and body mass index showed there was no significant difference between groups (hazard ratio, 0.96; 95% confidence interval, 0.76-1.22; P = .76), the authors wrote.
Triple-negative breast cancer patients with a BRCA mutation might have a survival advantage in the first few years following diagnosis,compared with non-BRCA carriers, the POSH study also found. Researchers reported a significant difference at 2 years (95% for BRCA-positive vs. 91% for BRCA-negative patients; P = .047), but there was no significant difference between arms at 5 or 10 years.
POSH is believed to be the largest prospective cohort study to compare breast cancer outcomes for patients with BRCA mutations to those with sporadic breast cancer. Previous studies, primarily retrospective, have suggested “better, worse, or similar outcomes” for BRCA-positive versus BRCA-negative patients, the authors wrote. Dr. Copson reported receiving honoraria from Roche, while her coauthors reported honoraria from GSK, Pfizer, AstraZeneca, and Pierre Fabre. Funding for the study was provided by the Wessex Cancer Trust, Cancer Research UK, and Breast Cancer Now.
SOURCE: Copson et al. Lancet Oncol. 2018 Jan 11 doi: 10.1016/S1470-2045(17)30891-4.
FROM LANCET ONCOLOGY
Key clinical point: Presence of BRCA1 or BRCA2 germline mutations did not significantly affect overall survival in women with young onset breast cancer.
Major finding: At 2 years, overall survival was 97.0% for BRCA mutation carriers and 96.6% for non-carriers, with similar results reported at 5 and 10 years.
Data source: A prospective cohort study including 2,733 women with breast cancer who were aged 40 years or younger at first diagnosis.
Disclosures: Funding for the study was provided by the Wessex Cancer Trust, Cancer Research UK, and Breast Cancer Now. Study authors declared honoraria from Roche, GSK, Pfizer, AstraZeneca, and Pierre Fabre.
Source: Copson ER et al. Lancet Oncol. 2018 Jan 11. doi: 10.1016/S1470-2045(17)30891-4.
FDA approves PARP inhibitor for BRCA+ advanced breast cancer
The Food and Drug Administration has approved the PARP inhibitor olaparib for the treatment of patients with germline BRCA-positive, HER2-negative metastatic breast cancer who have previously received chemotherapy.
This is the first PARP inhibitor approved to treat breast cancer and the first approval for treatment of patients with metastatic breast cancer who have a BRCA gene mutation, the FDA said in a press statement.
The FDA also expanded approval of the companion diagnostic, BRACAnalysis CDx, to include the detection of BRCA mutations in blood samples from patients with breast cancer.
A capsule form of olaparib (Lynparza) was first approved in 2014 for the treatment of patients with deleterious or suspected deleterious germline BRCA-mutated advanced ovarian cancer who have been treated with three or more prior lines of chemotherapy. In August 2017, the FDA granted regular approval to olaparib tablets for the maintenance treatment of adult patients with recurrent epithelial ovarian, fallopian tube, or primary peritoneal cancer, who are in a complete or partial response to platinum-based chemotherapy. Olaparib tablets and capsules are not interchangeable. Olaparib capsules are being phased out of the U.S. market and will be available only through the Lynparza Specialty Pharmacy Network, the FDA said.
Approval for the treatment of breast cancer was based on a 2.8 month improvement in progression-free survival with olaparib vs standard chemotherapy in the phase 3 OlympiAD trial of 302 previously treated patients with BRCA-positive, HER2-negative breast cancer. Results of the trial were presented at ASCO 2017 and simultaneously published in the New England Journal of Medicine (N Engl J Med. 2017 Jun 4. doi: 10.1056/NEJMoa1706450).
Common side effects of olaparib include anemia, neutropenia, leukopenia, nausea, fatigue, vomiting, nasopharyngitis, respiratory tract infection, influenza, diarrhea, arthralgia/myalgia, dysgeusia, headache, dyspepsia, decreased appetite, constipation and stomatitis.
Severe side effects include development of myelodysplastic syndrome/acute myeloid leukemia and pneumonitis. Women should be advised of the potential risk to the fetus and to use effective contraception, the FDA said.
The FDA granted the approval of olaparib to AstraZeneca Pharmaceuticals LP and the approval of the BRACAnalysis CDx to Myriad Genetic Laboratories, Inc.
The Food and Drug Administration has approved the PARP inhibitor olaparib for the treatment of patients with germline BRCA-positive, HER2-negative metastatic breast cancer who have previously received chemotherapy.
This is the first PARP inhibitor approved to treat breast cancer and the first approval for treatment of patients with metastatic breast cancer who have a BRCA gene mutation, the FDA said in a press statement.
The FDA also expanded approval of the companion diagnostic, BRACAnalysis CDx, to include the detection of BRCA mutations in blood samples from patients with breast cancer.
A capsule form of olaparib (Lynparza) was first approved in 2014 for the treatment of patients with deleterious or suspected deleterious germline BRCA-mutated advanced ovarian cancer who have been treated with three or more prior lines of chemotherapy. In August 2017, the FDA granted regular approval to olaparib tablets for the maintenance treatment of adult patients with recurrent epithelial ovarian, fallopian tube, or primary peritoneal cancer, who are in a complete or partial response to platinum-based chemotherapy. Olaparib tablets and capsules are not interchangeable. Olaparib capsules are being phased out of the U.S. market and will be available only through the Lynparza Specialty Pharmacy Network, the FDA said.
Approval for the treatment of breast cancer was based on a 2.8 month improvement in progression-free survival with olaparib vs standard chemotherapy in the phase 3 OlympiAD trial of 302 previously treated patients with BRCA-positive, HER2-negative breast cancer. Results of the trial were presented at ASCO 2017 and simultaneously published in the New England Journal of Medicine (N Engl J Med. 2017 Jun 4. doi: 10.1056/NEJMoa1706450).
Common side effects of olaparib include anemia, neutropenia, leukopenia, nausea, fatigue, vomiting, nasopharyngitis, respiratory tract infection, influenza, diarrhea, arthralgia/myalgia, dysgeusia, headache, dyspepsia, decreased appetite, constipation and stomatitis.
Severe side effects include development of myelodysplastic syndrome/acute myeloid leukemia and pneumonitis. Women should be advised of the potential risk to the fetus and to use effective contraception, the FDA said.
The FDA granted the approval of olaparib to AstraZeneca Pharmaceuticals LP and the approval of the BRACAnalysis CDx to Myriad Genetic Laboratories, Inc.
The Food and Drug Administration has approved the PARP inhibitor olaparib for the treatment of patients with germline BRCA-positive, HER2-negative metastatic breast cancer who have previously received chemotherapy.
This is the first PARP inhibitor approved to treat breast cancer and the first approval for treatment of patients with metastatic breast cancer who have a BRCA gene mutation, the FDA said in a press statement.
The FDA also expanded approval of the companion diagnostic, BRACAnalysis CDx, to include the detection of BRCA mutations in blood samples from patients with breast cancer.
A capsule form of olaparib (Lynparza) was first approved in 2014 for the treatment of patients with deleterious or suspected deleterious germline BRCA-mutated advanced ovarian cancer who have been treated with three or more prior lines of chemotherapy. In August 2017, the FDA granted regular approval to olaparib tablets for the maintenance treatment of adult patients with recurrent epithelial ovarian, fallopian tube, or primary peritoneal cancer, who are in a complete or partial response to platinum-based chemotherapy. Olaparib tablets and capsules are not interchangeable. Olaparib capsules are being phased out of the U.S. market and will be available only through the Lynparza Specialty Pharmacy Network, the FDA said.
Approval for the treatment of breast cancer was based on a 2.8 month improvement in progression-free survival with olaparib vs standard chemotherapy in the phase 3 OlympiAD trial of 302 previously treated patients with BRCA-positive, HER2-negative breast cancer. Results of the trial were presented at ASCO 2017 and simultaneously published in the New England Journal of Medicine (N Engl J Med. 2017 Jun 4. doi: 10.1056/NEJMoa1706450).
Common side effects of olaparib include anemia, neutropenia, leukopenia, nausea, fatigue, vomiting, nasopharyngitis, respiratory tract infection, influenza, diarrhea, arthralgia/myalgia, dysgeusia, headache, dyspepsia, decreased appetite, constipation and stomatitis.
Severe side effects include development of myelodysplastic syndrome/acute myeloid leukemia and pneumonitis. Women should be advised of the potential risk to the fetus and to use effective contraception, the FDA said.
The FDA granted the approval of olaparib to AstraZeneca Pharmaceuticals LP and the approval of the BRACAnalysis CDx to Myriad Genetic Laboratories, Inc.
Pain after breast surgery may not be caused by the operation
, according to a study of almost 2,000 women recruited from the Mastectomy Reconstructive Outcomes Consortium (MROC).
In the February issue of The Breast, investigators from the University of Michigan, Ann Arbor and Memorial Sloan Kettering Cancer Center, New York, wrote that almost half of the study subjects had some level of pain before their operations and that, at 2 years afterward, their pain had increased but not in a clinically meaningful way. This finding is consistent with earlier research, which investigators noted found that “one-fourth to one-half of women who undergo postmastectomy report persistent pain months and years after surgery.”
“Average clinical pain severity was strikingly similar for preoperative and postoperative assessments,” said lead author Randy S. Roth, PhD, of the University of Michigan, and his coauthors. “Postoperative levels of pain, acute postoperative pain and (marginally) level of depression held consistent relationship at 2-year follow-up with all outcome measures.”
The prospective, multicenter cohort study of 1,996 women was undertaken over 5 years. Most patients had immediate (92.7%) and bilateral (53.8%) reconstruction; 47.6% had sentinel lymph node biopsy and 25.9% had axillary lymph node dissection. Most had no adjuvant therapy: 70.3% received no radiation and 52.7% no chemotherapy.
At 2 years, the Numerical Pain Rating Scale (NPRS) measured what Dr. Roth and his coauthors called a “significant increase in pain intensity” – from an average rating of 1.1 to 1.2, an increase of 9%. However, the absolute change and standard deviation (1.7 for both intervals) “suggest that this was not a clinically meaningful change.” The researchers also recorded more complaints of bodily discomfort after 2 years, “but the statistical parameters again indicate little clinically meaningful differences from preoperative status.”
Pain ratings measured with the McGill Pain Questionnaire showed a significant decrease in the MPQ affective pain rating, from 1.6 preoperatively to 0.8 at 2 years (P less than .001), and virtually no change in the MPQ sensory rating, from 3.2 to 3.1.
The researchers drew some conclusions about demographic profiles and pain after breast reconstruction. Older age was associated with more severe pain on NPRS, and higher body mass index was linked with chronic postsurgical pain for the MPQ sensory rating, NPRS score, and body discomfort scores.
Treatment characteristics associated with chronic postsurgical pain (CPSP) include radiation therapy during or after reconstruction and chemotherapy before reconstruction. Chemotherapy during or after reconstruction was associated with higher MPQ affective rating scores at 2 years (P = .011), as was chemotherapy both before and during or after reconstruction (P = .001). The latter also was linked to higher NPRS scores (P = .0015).
The type of surgery also was a factor in CPSP, the researchers wrote. Both MPQ sensory and affective ratings were higher in women who had free transverse flap surgery, or deep or superficial inferior epigastric perforator surgery than in women who had tissue expander/implant reconstruction. Lymph node status and timing of surgery had no impact on chronic pain.
One noteworthy finding, Dr. Roth and his coauthors wrote, is that “careful examination of our data suggests that CPSP following breast reconstruction may be of less clinical concern as a direct consequence of breast reconstruction than suggested by previous investigations of major surgery, including mastectomy and breast reconstruction.” Future studies of chronic postsurgical pain in breast reconstruction “will require greater methodological rigor” to reach more sound conclusions to use in patient counseling.
Dr. Roth and his coauthors had no financial relationships to disclose.
SOURCE: Roth RS et al. Breast 2018;37:119-25.
, according to a study of almost 2,000 women recruited from the Mastectomy Reconstructive Outcomes Consortium (MROC).
In the February issue of The Breast, investigators from the University of Michigan, Ann Arbor and Memorial Sloan Kettering Cancer Center, New York, wrote that almost half of the study subjects had some level of pain before their operations and that, at 2 years afterward, their pain had increased but not in a clinically meaningful way. This finding is consistent with earlier research, which investigators noted found that “one-fourth to one-half of women who undergo postmastectomy report persistent pain months and years after surgery.”
“Average clinical pain severity was strikingly similar for preoperative and postoperative assessments,” said lead author Randy S. Roth, PhD, of the University of Michigan, and his coauthors. “Postoperative levels of pain, acute postoperative pain and (marginally) level of depression held consistent relationship at 2-year follow-up with all outcome measures.”
The prospective, multicenter cohort study of 1,996 women was undertaken over 5 years. Most patients had immediate (92.7%) and bilateral (53.8%) reconstruction; 47.6% had sentinel lymph node biopsy and 25.9% had axillary lymph node dissection. Most had no adjuvant therapy: 70.3% received no radiation and 52.7% no chemotherapy.
At 2 years, the Numerical Pain Rating Scale (NPRS) measured what Dr. Roth and his coauthors called a “significant increase in pain intensity” – from an average rating of 1.1 to 1.2, an increase of 9%. However, the absolute change and standard deviation (1.7 for both intervals) “suggest that this was not a clinically meaningful change.” The researchers also recorded more complaints of bodily discomfort after 2 years, “but the statistical parameters again indicate little clinically meaningful differences from preoperative status.”
Pain ratings measured with the McGill Pain Questionnaire showed a significant decrease in the MPQ affective pain rating, from 1.6 preoperatively to 0.8 at 2 years (P less than .001), and virtually no change in the MPQ sensory rating, from 3.2 to 3.1.
The researchers drew some conclusions about demographic profiles and pain after breast reconstruction. Older age was associated with more severe pain on NPRS, and higher body mass index was linked with chronic postsurgical pain for the MPQ sensory rating, NPRS score, and body discomfort scores.
Treatment characteristics associated with chronic postsurgical pain (CPSP) include radiation therapy during or after reconstruction and chemotherapy before reconstruction. Chemotherapy during or after reconstruction was associated with higher MPQ affective rating scores at 2 years (P = .011), as was chemotherapy both before and during or after reconstruction (P = .001). The latter also was linked to higher NPRS scores (P = .0015).
The type of surgery also was a factor in CPSP, the researchers wrote. Both MPQ sensory and affective ratings were higher in women who had free transverse flap surgery, or deep or superficial inferior epigastric perforator surgery than in women who had tissue expander/implant reconstruction. Lymph node status and timing of surgery had no impact on chronic pain.
One noteworthy finding, Dr. Roth and his coauthors wrote, is that “careful examination of our data suggests that CPSP following breast reconstruction may be of less clinical concern as a direct consequence of breast reconstruction than suggested by previous investigations of major surgery, including mastectomy and breast reconstruction.” Future studies of chronic postsurgical pain in breast reconstruction “will require greater methodological rigor” to reach more sound conclusions to use in patient counseling.
Dr. Roth and his coauthors had no financial relationships to disclose.
SOURCE: Roth RS et al. Breast 2018;37:119-25.
, according to a study of almost 2,000 women recruited from the Mastectomy Reconstructive Outcomes Consortium (MROC).
In the February issue of The Breast, investigators from the University of Michigan, Ann Arbor and Memorial Sloan Kettering Cancer Center, New York, wrote that almost half of the study subjects had some level of pain before their operations and that, at 2 years afterward, their pain had increased but not in a clinically meaningful way. This finding is consistent with earlier research, which investigators noted found that “one-fourth to one-half of women who undergo postmastectomy report persistent pain months and years after surgery.”
“Average clinical pain severity was strikingly similar for preoperative and postoperative assessments,” said lead author Randy S. Roth, PhD, of the University of Michigan, and his coauthors. “Postoperative levels of pain, acute postoperative pain and (marginally) level of depression held consistent relationship at 2-year follow-up with all outcome measures.”
The prospective, multicenter cohort study of 1,996 women was undertaken over 5 years. Most patients had immediate (92.7%) and bilateral (53.8%) reconstruction; 47.6% had sentinel lymph node biopsy and 25.9% had axillary lymph node dissection. Most had no adjuvant therapy: 70.3% received no radiation and 52.7% no chemotherapy.
At 2 years, the Numerical Pain Rating Scale (NPRS) measured what Dr. Roth and his coauthors called a “significant increase in pain intensity” – from an average rating of 1.1 to 1.2, an increase of 9%. However, the absolute change and standard deviation (1.7 for both intervals) “suggest that this was not a clinically meaningful change.” The researchers also recorded more complaints of bodily discomfort after 2 years, “but the statistical parameters again indicate little clinically meaningful differences from preoperative status.”
Pain ratings measured with the McGill Pain Questionnaire showed a significant decrease in the MPQ affective pain rating, from 1.6 preoperatively to 0.8 at 2 years (P less than .001), and virtually no change in the MPQ sensory rating, from 3.2 to 3.1.
The researchers drew some conclusions about demographic profiles and pain after breast reconstruction. Older age was associated with more severe pain on NPRS, and higher body mass index was linked with chronic postsurgical pain for the MPQ sensory rating, NPRS score, and body discomfort scores.
Treatment characteristics associated with chronic postsurgical pain (CPSP) include radiation therapy during or after reconstruction and chemotherapy before reconstruction. Chemotherapy during or after reconstruction was associated with higher MPQ affective rating scores at 2 years (P = .011), as was chemotherapy both before and during or after reconstruction (P = .001). The latter also was linked to higher NPRS scores (P = .0015).
The type of surgery also was a factor in CPSP, the researchers wrote. Both MPQ sensory and affective ratings were higher in women who had free transverse flap surgery, or deep or superficial inferior epigastric perforator surgery than in women who had tissue expander/implant reconstruction. Lymph node status and timing of surgery had no impact on chronic pain.
One noteworthy finding, Dr. Roth and his coauthors wrote, is that “careful examination of our data suggests that CPSP following breast reconstruction may be of less clinical concern as a direct consequence of breast reconstruction than suggested by previous investigations of major surgery, including mastectomy and breast reconstruction.” Future studies of chronic postsurgical pain in breast reconstruction “will require greater methodological rigor” to reach more sound conclusions to use in patient counseling.
Dr. Roth and his coauthors had no financial relationships to disclose.
SOURCE: Roth RS et al. Breast 2018;37:119-25.
FROM THE BREAST
Key clinical point: Breast reconstruction surgery may not be the cause of persistent pain afterward.
Major finding: McGill Pain Questionnaire affective pain rating decreased from 1.6 preoperatively to 0.8 at 2 years.
Data source: Five-year prospective multicenter cohort study of 1,966 women recruited for the Mastectomy Reconstruction Outcomes Consortium.
Disclosures: Dr. Roth and his coauthors reported having no financial disclosures.
Source: Roth RS et al. Breast 2018;37:119-25.
Modeling study: Screening, treatment effects on breast cancer mortality vary by subtype
, according to a simulation modeling study.
The estimated rate of reduction in overall breast cancer mortality in 2000 was 37% from an estimated baseline rate of 64 deaths per 100,000 women, with 44% and 56% of that associated with screening and treatment, respectively. In 2012 the estimated reduction was 49% from an estimated baseline rate of 63 per 100,000 women, with 37% and 63% associated with screening and treatment, respectively (estimated 12% difference in 2012 vs. 2000), Sylvia K. Plevritis, PhD, of Stanford (Calif.) University and her colleagues reported in JAMA.
Screening and treatment were estimated to contribute to the reductions at varying rates. For example, the relative contributions of screening vs. treatment were 36% vs. 64% for ER+/ERBB2– disease; 31% vs. 69% for ER+/ERBB2+ disease; 40% vs. 60% for ER–/ERBB2+ disease; and 48% vs. 52% for ER–/ERBB2– disease.
The model-based analysis provides clinically relevant insights about the contributions of screening and treatment to reductions in breast cancer mortality by molecular subtype, showing a greater relative contribution of treatment in 2012 overall and for all subtypes except ER–/ERBB2– disease, the authors said.
“Because ER+ cancers are the most prevalent and this group is expected to increase with time, additional advances for this subtype could have the largest effect on reducing the overall population burden of breast cancer,” they noted.
This study was supported by grants from the National Cancer Institute and the American Cancer Society. Dr. Plevritis reported consulting for GRAIL.
SOURCE: Plevritis S et al. JAMA. 2018 Jan 9;319(2):154-64. doi: 10.1001/jama.2017.19130.
.
, according to a simulation modeling study.
The estimated rate of reduction in overall breast cancer mortality in 2000 was 37% from an estimated baseline rate of 64 deaths per 100,000 women, with 44% and 56% of that associated with screening and treatment, respectively. In 2012 the estimated reduction was 49% from an estimated baseline rate of 63 per 100,000 women, with 37% and 63% associated with screening and treatment, respectively (estimated 12% difference in 2012 vs. 2000), Sylvia K. Plevritis, PhD, of Stanford (Calif.) University and her colleagues reported in JAMA.
Screening and treatment were estimated to contribute to the reductions at varying rates. For example, the relative contributions of screening vs. treatment were 36% vs. 64% for ER+/ERBB2– disease; 31% vs. 69% for ER+/ERBB2+ disease; 40% vs. 60% for ER–/ERBB2+ disease; and 48% vs. 52% for ER–/ERBB2– disease.
The model-based analysis provides clinically relevant insights about the contributions of screening and treatment to reductions in breast cancer mortality by molecular subtype, showing a greater relative contribution of treatment in 2012 overall and for all subtypes except ER–/ERBB2– disease, the authors said.
“Because ER+ cancers are the most prevalent and this group is expected to increase with time, additional advances for this subtype could have the largest effect on reducing the overall population burden of breast cancer,” they noted.
This study was supported by grants from the National Cancer Institute and the American Cancer Society. Dr. Plevritis reported consulting for GRAIL.
SOURCE: Plevritis S et al. JAMA. 2018 Jan 9;319(2):154-64. doi: 10.1001/jama.2017.19130.
.
, according to a simulation modeling study.
The estimated rate of reduction in overall breast cancer mortality in 2000 was 37% from an estimated baseline rate of 64 deaths per 100,000 women, with 44% and 56% of that associated with screening and treatment, respectively. In 2012 the estimated reduction was 49% from an estimated baseline rate of 63 per 100,000 women, with 37% and 63% associated with screening and treatment, respectively (estimated 12% difference in 2012 vs. 2000), Sylvia K. Plevritis, PhD, of Stanford (Calif.) University and her colleagues reported in JAMA.
Screening and treatment were estimated to contribute to the reductions at varying rates. For example, the relative contributions of screening vs. treatment were 36% vs. 64% for ER+/ERBB2– disease; 31% vs. 69% for ER+/ERBB2+ disease; 40% vs. 60% for ER–/ERBB2+ disease; and 48% vs. 52% for ER–/ERBB2– disease.
The model-based analysis provides clinically relevant insights about the contributions of screening and treatment to reductions in breast cancer mortality by molecular subtype, showing a greater relative contribution of treatment in 2012 overall and for all subtypes except ER–/ERBB2– disease, the authors said.
“Because ER+ cancers are the most prevalent and this group is expected to increase with time, additional advances for this subtype could have the largest effect on reducing the overall population burden of breast cancer,” they noted.
This study was supported by grants from the National Cancer Institute and the American Cancer Society. Dr. Plevritis reported consulting for GRAIL.
SOURCE: Plevritis S et al. JAMA. 2018 Jan 9;319(2):154-64. doi: 10.1001/jama.2017.19130.
.
FROM JAMA
Key clinical point: Effects of treatment and screening advances on breast cancer mortality vary by molecular subtype.
Major finding: The estimated rate of reduction on mortality was 12% greater in 2012.
Study details: A simulation study using six models and national breast cancer data.
Disclosures: This study was supported by grants from the National Cancer Institute and the American Cancer Society. Dr. Plevritis reported consulting for GRAIL.
Source: Plevritis S et al. JAMA. 2018 Jan 9;319(2):154-64.
Trial updates will help tailor endocrine therapy for premenopausal breast cancer
SAN ANTONIO – Adjuvant endocrine therapies improve outcomes of premenopausal breast cancer in the long term, with absolute benefit varying somewhat by therapy and by patient and disease characteristics, according to planned updates of a pair of pivotal phase 3 trials.
The trials – TEXT (Tamoxifen and Exemestane Trial) and SOFT (Suppression of Ovarian Function Trial) – are coordinated by the International Breast Cancer Study Group and together randomized more than 5,000 premenopausal women with early hormone receptor–positive breast cancer to 5 years of various types of adjuvant endocrine therapy. Their initial results, reported several years ago, form part of treatment guidelines that are used worldwide.
Relative benefits for various outcomes were generally similar across subgroups, but absolute benefits were greater for women having certain features increasing risk for poor outcomes.
Clinical implications
These updates, along with other emerging data, can be used to optimize endocrine therapy for younger women with breast cancer, according to invited discussant Ann H. Partridge, MD, of Dana Farber Cancer Institute in Boston.
“For higher-risk disease, we should be considering OFS. At this point in time, I don’t think HER2 status alone should drive this decision,” she commented. “If you are getting OFS, what do we do, AI versus tamoxifen? Well, we do see a large improvement in disease-free survival [with AIs], so many women will want to use AIs. Yet tamoxifen is still reasonable, especially in light of the survival data.”
Data on switch strategies and extended-duration therapy are generally lacking at present for the premenopausal population, Dr. Partridge noted. “That’s something that we still need to extrapolate from data that’s predominantly in postmenopausal women.”
Another compelling question is whether OFS can be used instead of chemo for some patients. “We are increasingly recognizing that women with higher-risk anatomy and lower-risk biology having endocrine-responsive tumors may get more bang for the buck from the optimizing of hormonal therapy, and chemo may not add much,” she said.
Both short- and long-term toxicities of the various endocrine therapies and, for aromatase inhibitors, the potential for breakthrough (return of estradiol levels to premenopausal levels) also need to be considered, Dr. Partridge stressed. “And ultimately, patient preference and tolerance are key. After all, the best treatment is the one the patient will take.”
“We need to follow these women on TEXT and SOFT very long term. It would be a crime not to follow these women further out,” she maintained. “We need to conduct real-world comparative effectiveness research to understand the risks and benefits of OFS more fully in our survivors. Then, as we start to suppress more ovaries in more women with breast cancer, we need to be aware clinically of these risks, and we need to share this awareness with their primary care providers because we need to optimize in particular their cardiovascular risk factors, and screen and treat for potential comorbidities that they may be at higher risk for.”
Joint TEXT and SOFT update
Initial results of the joint TEXT and SOFT analysis, reported after a median follow-up of 5.7 years, showed that exemestane plus OFS was superior to tamoxifen plus OFS for the primary outcome, providing a significant 3.8% absolute gain in 5-year disease-free survival (N Engl J Med. 2014;371:107-18).
The updated joint analysis, now with a median follow-up of 9 years and based on data from 4,690 women, showed that the 8-year rate of disease-free survival was 86.8% with exemestane plus OFS versus 82.8% with tamoxifen plus OFS (hazard ratio, 0.77; P = .0006), for a similar absolute benefit of 4.0%, reported Prudence Francis, MD, of the University of Melbourne, head of Medical Oncology in the Breast Service at the Peter MacCallum Cancer Centre, Melbourne.
In stratified analysis, absolute benefit tended to be greater among women in TEXT who received chemotherapy (6.0%); intermediate among women in TEXT who did not receive chemotherapy (3.7%) and women in SOFT who received prior chemotherapy (3.7%); and less among women in SOFT who did not receive chemotherapy (1.9%).
Exemestane plus OFS was also superior to tamoxifen plus OFS in terms of breast cancer–free interval, with an absolute 4.1% benefit (P = .0002), and distant recurrence–free interval, with an absolute 2.1% benefit (P = .02). Overall survival did not differ significantly between arms.
Among the 86% of patients with HER2-negative disease, exemestane plus OFS netted an absolute disease-free survival gain of 5.4% and an absolute distant recurrence–free interval gain of 3.4%. There was a consistent relative treatment benefit across subgroups, but larger absolute benefit, on the order of 5%-9%, in women given chemotherapy and in those younger than 35 years.
“Results for the HER2-positive subgroup require further investigation,” Dr. Francis said. “The trials enrolled both before and after the routine use of adjuvant trastuzumab, and a significant proportion of the patients with HER2-positive breast cancer did not receive adjuvant HER2-targeted therapy.”
In the entire joint-analysis population, exemestane plus OFS was associated with higher rates of musculoskeletal events of grade 3 or 4 (11% vs. 6%) and osteoporosis of grade 2-4 (15% vs. 7%), while tamoxifen plus OFS was associated with a higher rate of thrombosis/embolism of grade 2-4 (2.3% vs. 1.2%) and more cases of endometrial cancer (9 vs. 4 cases). At 4 years, early discontinuation of oral endocrine therapy was greater for exemestane than for tamoxifen (25% vs. 19%).
“After longer follow-up, with a median of 9 years, the combined analysis results confirm a statistically significant improvement in disease outcomes with exemestane plus ovarian suppression. As is critical given the long natural history of estrogen receptor–positive breast cancer, follow-up in these trials is currently continuing,” Dr. Francis summarized.
“To optimally translate the observed absolute trial improvements into clinical practice, oncologists need to discuss and weigh the potential benefits and toxicities in each individual patient who is premenopausal with hormone receptor–positive breast cancer,” she recommended.
Session attendee Hope S. Rugo, MD, of the University of California, San Francisco, Helen Diller Family Comprehensive Cancer Center, noted that exemestane had superior benefit despite the 25% rate of early discontinuation. “I wonder if one of the interpretations of that, given the toxicity of this therapy for very young women, is that we need some but maybe not so much. Maybe they don’t need 5 years altogether,” she said.
“The fact that they stopped their assigned endocrine therapy doesn’t mean that they didn’t continue any therapy. They may have switched over to tamoxifen or they may have decided they wanted to have a baby or there may have been many other things,” Dr. Francis replied, noting that analyses sorting out the reasons for early discontinuation are planned.
Session attendee Mark E. Sherman, MD, of the Mayo Clinic, Jacksonville, Fla., asked, “Do you have any ability to test for tamoxifen metabolites? It’s possible that a third to a half of patients got reduced benefit from that drug.”
Banked samples are available and a substudy is planned, according to Dr. Francis. “We haven’t got data on that yet, but yes, we are analyzing that.”
SOFT update
Initial results of the SOFT trial, reported after a median follow-up of 5.6 years, showed that adding OFS to tamoxifen did not significantly improve disease-free survival over tamoxifen alone in the entire trial population (N Engl J Med. 2015;372:436-46). However, there was benefit for women who received chemotherapy and remained premenopausal.
The updated SOFT analysis, now with a median follow-up of 8 years, focused mainly on the 1,018 women given tamoxifen alone and the 1,015 women given tamoxifen plus OFS. (Another 1,014 women were given exemestane plus OFS.)
“SOFT is now positive for its primary endpoint,” reported first author Gini Fleming, MD, director of the Medical Oncology Breast Program and medical oncology director of Gynecologic Oncology at University of Chicago Medicine. The 8-year disease-free survival rate was 83.2% with tamoxifen plus OFS, compared with 78.9% with tamoxifen alone (HR, 0.76; P = .009), corresponding to a 4.2% gain in this outcome. The relative benefit was identical whether patients had received chemotherapy or not, but absolute benefit was greater for those who had (5.3%), as well as for patients younger than 35 years (8.7%).
In addition, exemestane plus OFS was superior to tamoxifen alone (85.9% vs. 78.9%; HR, 0.65), with an absolute benefit of 7.0%. Again, absolute benefit was more pronounced among women who had received prior chemotherapy (9.0%) or were younger than 35 years (13.1%).
The relative disease-free survival benefit of tamoxifen plus OFS over tamoxifen alone was similar across most subgroups stratified by disease characteristics, but patients with HER2-positive disease derived greater relative benefit from the combination as compared with their HER2-negative counterparts (P = .04 for interaction). “When we look at the combination of exemestane plus OFS versus tamoxifen, this heterogeneity is no longer seen,” Dr. Fleming noted.
In the entire trial population, there was no significant benefit of tamoxifen plus OFS over tamoxifen alone for distant recurrence-free interval and a small, significant absolute 1.9% gain in overall survival.
“The cohort who had elected to receive no prior chemotherapy did exceedingly well regardless of therapy,” she said, with little difference in overall survival across the three arms. “There were only 24 deaths total in this cohort, and 12 of those deaths were in the setting of no distant recurrence.”
On the other hand, among the women who received chemotherapy, there were significant absolute overall survival benefits of 4.3% with tamoxifen plus OFS and 2.1% with exemestane plus OFS, over tamoxifen alone. “This late emergence of an overall survival benefit is consistent with the time course of events in estrogen receptor–positive breast cancer,” Dr. Fleming commented.
The proportion of patients who stopped their oral endocrine therapy early was 22.5% with tamoxifen alone and 18.5% with tamoxifen plus OFS. (It was 27.8% with exemestane plus OFS.) “Almost a quarter of the patients on either tamoxifen arm were using extended oral endocrine therapy at 6 years or later prior to any disease progression. Only about 12% of patients in the exemestane group were doing so,” she noted.
There were more cases of endometrial cancer with tamoxifen alone than with tamoxifen plus OFS (7 vs. 4 cases). Thrombosis/embolism of grade 2-4 occurred in 2.2% of each group (and 0.9% of the exemestane plus OFS group). Musculoskeletal symptoms of grade 3 or 4 occurred in 6.7% of patients with tamoxifen alone and 5.9% with tamoxifen plus OFS, but 12.0% with exemestane plus OFS. Respective rates of osteoporosis grade 2-4 were 3.9%, 6.1%, and 11.9%.
“The addition of OFS to tamoxifen significantly improves disease-free survival at 8 years’ median follow-up, and disease-free survival benefits are further improved by the use of exemestane plus OFS,” Dr. Fleming summarized. “Follow-up, which is critically important given the long natural history of ER-positive disease, continues.”
Session attendee Matthew P. Goetz, MD, of the Mayo Clinic, Rochester, Minn., commented, “For the primary endpoint, I was looking at the tail for tamoxifen. It seemed that there was a relatively rapid drop-off between year 5 and this 8-year follow-up. Have you looked carefully to see whether there is a difference between those who stayed on their therapy versus those who went off it per protocol? That is, extended versus not extended? The question is whether there is a carry-over effect, if you will, that is different in those with OFS versus those not.”
“The percent who went on to extended therapy between the tamoxifen and the tamoxifen plus OFS was fairly similar,” Dr. Fleming replied. “But the answer is no, we have not yet done any sort of per protocol analysis.”
Session attendee Steven Vogl, MD, of Montefiore Medical Center, New York, commented, “I worry about your control group. I’m worried, first, how many of your tamoxifen patients lost their menses and became postmenopausal in those 5 years? And of those, why didn’t they switch to an aromatase inhibitor? Only 25% of the patients continued after the 5 years according to your slide, and all of those patients should either have stayed on tamoxifen or switched to an aromatase inhibitor, now probably for 2 years at least.”
“We have not yet looked at data for who became amenorrheic during treatment, although we have it. However, it’s certainly possible to become amenorrheic on tamoxifen and not be postmenopausal, and we didn’t regularly collect estradiol levels on any but the very small subset of women in the SOFT-S trial. So I don’t know that we have exactly the data that you’re looking for,” Dr. Fleming said. “Many of these women are obviously at very, very low risk and have done well with 5 years of tamoxifen alone, and I don’t know, even given current guidelines, that extended tamoxifen would add a lot to that.”
Finally, session attendee Richard Gray, professor of medical statistics at the University of Oxford (England), wondered, “What is the certainty that follow-up will happen? Because obviously, prolonged follow-up is expensive and there are controversies about that. But this would be the one study you would really want to have 15- and 20-year data on.”
“We are working very, very hard on that,” Dr. Fleming replied. “NCI granted additional funds to institutions for prolonging follow-up, and IBCSG has been ceaselessly working to look for funding to continue it. So I think it’s relatively certain that it will happen.”
Dr. Francis disclosed that she has received fees for non-CME services from AstraZeneca and has given an overseas lecture for Pfizer. Dr. Fleming disclosed that she had no relevant financial relationships with commercial interests. The trials received financial support from Pfizer and Ipsen.
SOURCES: Francis et al. SABCS Abstract GS4-02; Fleming et al. SABCS Abstract GS4-03
SAN ANTONIO – Adjuvant endocrine therapies improve outcomes of premenopausal breast cancer in the long term, with absolute benefit varying somewhat by therapy and by patient and disease characteristics, according to planned updates of a pair of pivotal phase 3 trials.
The trials – TEXT (Tamoxifen and Exemestane Trial) and SOFT (Suppression of Ovarian Function Trial) – are coordinated by the International Breast Cancer Study Group and together randomized more than 5,000 premenopausal women with early hormone receptor–positive breast cancer to 5 years of various types of adjuvant endocrine therapy. Their initial results, reported several years ago, form part of treatment guidelines that are used worldwide.
Relative benefits for various outcomes were generally similar across subgroups, but absolute benefits were greater for women having certain features increasing risk for poor outcomes.
Clinical implications
These updates, along with other emerging data, can be used to optimize endocrine therapy for younger women with breast cancer, according to invited discussant Ann H. Partridge, MD, of Dana Farber Cancer Institute in Boston.
“For higher-risk disease, we should be considering OFS. At this point in time, I don’t think HER2 status alone should drive this decision,” she commented. “If you are getting OFS, what do we do, AI versus tamoxifen? Well, we do see a large improvement in disease-free survival [with AIs], so many women will want to use AIs. Yet tamoxifen is still reasonable, especially in light of the survival data.”
Data on switch strategies and extended-duration therapy are generally lacking at present for the premenopausal population, Dr. Partridge noted. “That’s something that we still need to extrapolate from data that’s predominantly in postmenopausal women.”
Another compelling question is whether OFS can be used instead of chemo for some patients. “We are increasingly recognizing that women with higher-risk anatomy and lower-risk biology having endocrine-responsive tumors may get more bang for the buck from the optimizing of hormonal therapy, and chemo may not add much,” she said.
Both short- and long-term toxicities of the various endocrine therapies and, for aromatase inhibitors, the potential for breakthrough (return of estradiol levels to premenopausal levels) also need to be considered, Dr. Partridge stressed. “And ultimately, patient preference and tolerance are key. After all, the best treatment is the one the patient will take.”
“We need to follow these women on TEXT and SOFT very long term. It would be a crime not to follow these women further out,” she maintained. “We need to conduct real-world comparative effectiveness research to understand the risks and benefits of OFS more fully in our survivors. Then, as we start to suppress more ovaries in more women with breast cancer, we need to be aware clinically of these risks, and we need to share this awareness with their primary care providers because we need to optimize in particular their cardiovascular risk factors, and screen and treat for potential comorbidities that they may be at higher risk for.”
Joint TEXT and SOFT update
Initial results of the joint TEXT and SOFT analysis, reported after a median follow-up of 5.7 years, showed that exemestane plus OFS was superior to tamoxifen plus OFS for the primary outcome, providing a significant 3.8% absolute gain in 5-year disease-free survival (N Engl J Med. 2014;371:107-18).
The updated joint analysis, now with a median follow-up of 9 years and based on data from 4,690 women, showed that the 8-year rate of disease-free survival was 86.8% with exemestane plus OFS versus 82.8% with tamoxifen plus OFS (hazard ratio, 0.77; P = .0006), for a similar absolute benefit of 4.0%, reported Prudence Francis, MD, of the University of Melbourne, head of Medical Oncology in the Breast Service at the Peter MacCallum Cancer Centre, Melbourne.
In stratified analysis, absolute benefit tended to be greater among women in TEXT who received chemotherapy (6.0%); intermediate among women in TEXT who did not receive chemotherapy (3.7%) and women in SOFT who received prior chemotherapy (3.7%); and less among women in SOFT who did not receive chemotherapy (1.9%).
Exemestane plus OFS was also superior to tamoxifen plus OFS in terms of breast cancer–free interval, with an absolute 4.1% benefit (P = .0002), and distant recurrence–free interval, with an absolute 2.1% benefit (P = .02). Overall survival did not differ significantly between arms.
Among the 86% of patients with HER2-negative disease, exemestane plus OFS netted an absolute disease-free survival gain of 5.4% and an absolute distant recurrence–free interval gain of 3.4%. There was a consistent relative treatment benefit across subgroups, but larger absolute benefit, on the order of 5%-9%, in women given chemotherapy and in those younger than 35 years.
“Results for the HER2-positive subgroup require further investigation,” Dr. Francis said. “The trials enrolled both before and after the routine use of adjuvant trastuzumab, and a significant proportion of the patients with HER2-positive breast cancer did not receive adjuvant HER2-targeted therapy.”
In the entire joint-analysis population, exemestane plus OFS was associated with higher rates of musculoskeletal events of grade 3 or 4 (11% vs. 6%) and osteoporosis of grade 2-4 (15% vs. 7%), while tamoxifen plus OFS was associated with a higher rate of thrombosis/embolism of grade 2-4 (2.3% vs. 1.2%) and more cases of endometrial cancer (9 vs. 4 cases). At 4 years, early discontinuation of oral endocrine therapy was greater for exemestane than for tamoxifen (25% vs. 19%).
“After longer follow-up, with a median of 9 years, the combined analysis results confirm a statistically significant improvement in disease outcomes with exemestane plus ovarian suppression. As is critical given the long natural history of estrogen receptor–positive breast cancer, follow-up in these trials is currently continuing,” Dr. Francis summarized.
“To optimally translate the observed absolute trial improvements into clinical practice, oncologists need to discuss and weigh the potential benefits and toxicities in each individual patient who is premenopausal with hormone receptor–positive breast cancer,” she recommended.
Session attendee Hope S. Rugo, MD, of the University of California, San Francisco, Helen Diller Family Comprehensive Cancer Center, noted that exemestane had superior benefit despite the 25% rate of early discontinuation. “I wonder if one of the interpretations of that, given the toxicity of this therapy for very young women, is that we need some but maybe not so much. Maybe they don’t need 5 years altogether,” she said.
“The fact that they stopped their assigned endocrine therapy doesn’t mean that they didn’t continue any therapy. They may have switched over to tamoxifen or they may have decided they wanted to have a baby or there may have been many other things,” Dr. Francis replied, noting that analyses sorting out the reasons for early discontinuation are planned.
Session attendee Mark E. Sherman, MD, of the Mayo Clinic, Jacksonville, Fla., asked, “Do you have any ability to test for tamoxifen metabolites? It’s possible that a third to a half of patients got reduced benefit from that drug.”
Banked samples are available and a substudy is planned, according to Dr. Francis. “We haven’t got data on that yet, but yes, we are analyzing that.”
SOFT update
Initial results of the SOFT trial, reported after a median follow-up of 5.6 years, showed that adding OFS to tamoxifen did not significantly improve disease-free survival over tamoxifen alone in the entire trial population (N Engl J Med. 2015;372:436-46). However, there was benefit for women who received chemotherapy and remained premenopausal.
The updated SOFT analysis, now with a median follow-up of 8 years, focused mainly on the 1,018 women given tamoxifen alone and the 1,015 women given tamoxifen plus OFS. (Another 1,014 women were given exemestane plus OFS.)
“SOFT is now positive for its primary endpoint,” reported first author Gini Fleming, MD, director of the Medical Oncology Breast Program and medical oncology director of Gynecologic Oncology at University of Chicago Medicine. The 8-year disease-free survival rate was 83.2% with tamoxifen plus OFS, compared with 78.9% with tamoxifen alone (HR, 0.76; P = .009), corresponding to a 4.2% gain in this outcome. The relative benefit was identical whether patients had received chemotherapy or not, but absolute benefit was greater for those who had (5.3%), as well as for patients younger than 35 years (8.7%).
In addition, exemestane plus OFS was superior to tamoxifen alone (85.9% vs. 78.9%; HR, 0.65), with an absolute benefit of 7.0%. Again, absolute benefit was more pronounced among women who had received prior chemotherapy (9.0%) or were younger than 35 years (13.1%).
The relative disease-free survival benefit of tamoxifen plus OFS over tamoxifen alone was similar across most subgroups stratified by disease characteristics, but patients with HER2-positive disease derived greater relative benefit from the combination as compared with their HER2-negative counterparts (P = .04 for interaction). “When we look at the combination of exemestane plus OFS versus tamoxifen, this heterogeneity is no longer seen,” Dr. Fleming noted.
In the entire trial population, there was no significant benefit of tamoxifen plus OFS over tamoxifen alone for distant recurrence-free interval and a small, significant absolute 1.9% gain in overall survival.
“The cohort who had elected to receive no prior chemotherapy did exceedingly well regardless of therapy,” she said, with little difference in overall survival across the three arms. “There were only 24 deaths total in this cohort, and 12 of those deaths were in the setting of no distant recurrence.”
On the other hand, among the women who received chemotherapy, there were significant absolute overall survival benefits of 4.3% with tamoxifen plus OFS and 2.1% with exemestane plus OFS, over tamoxifen alone. “This late emergence of an overall survival benefit is consistent with the time course of events in estrogen receptor–positive breast cancer,” Dr. Fleming commented.
The proportion of patients who stopped their oral endocrine therapy early was 22.5% with tamoxifen alone and 18.5% with tamoxifen plus OFS. (It was 27.8% with exemestane plus OFS.) “Almost a quarter of the patients on either tamoxifen arm were using extended oral endocrine therapy at 6 years or later prior to any disease progression. Only about 12% of patients in the exemestane group were doing so,” she noted.
There were more cases of endometrial cancer with tamoxifen alone than with tamoxifen plus OFS (7 vs. 4 cases). Thrombosis/embolism of grade 2-4 occurred in 2.2% of each group (and 0.9% of the exemestane plus OFS group). Musculoskeletal symptoms of grade 3 or 4 occurred in 6.7% of patients with tamoxifen alone and 5.9% with tamoxifen plus OFS, but 12.0% with exemestane plus OFS. Respective rates of osteoporosis grade 2-4 were 3.9%, 6.1%, and 11.9%.
“The addition of OFS to tamoxifen significantly improves disease-free survival at 8 years’ median follow-up, and disease-free survival benefits are further improved by the use of exemestane plus OFS,” Dr. Fleming summarized. “Follow-up, which is critically important given the long natural history of ER-positive disease, continues.”
Session attendee Matthew P. Goetz, MD, of the Mayo Clinic, Rochester, Minn., commented, “For the primary endpoint, I was looking at the tail for tamoxifen. It seemed that there was a relatively rapid drop-off between year 5 and this 8-year follow-up. Have you looked carefully to see whether there is a difference between those who stayed on their therapy versus those who went off it per protocol? That is, extended versus not extended? The question is whether there is a carry-over effect, if you will, that is different in those with OFS versus those not.”
“The percent who went on to extended therapy between the tamoxifen and the tamoxifen plus OFS was fairly similar,” Dr. Fleming replied. “But the answer is no, we have not yet done any sort of per protocol analysis.”
Session attendee Steven Vogl, MD, of Montefiore Medical Center, New York, commented, “I worry about your control group. I’m worried, first, how many of your tamoxifen patients lost their menses and became postmenopausal in those 5 years? And of those, why didn’t they switch to an aromatase inhibitor? Only 25% of the patients continued after the 5 years according to your slide, and all of those patients should either have stayed on tamoxifen or switched to an aromatase inhibitor, now probably for 2 years at least.”
“We have not yet looked at data for who became amenorrheic during treatment, although we have it. However, it’s certainly possible to become amenorrheic on tamoxifen and not be postmenopausal, and we didn’t regularly collect estradiol levels on any but the very small subset of women in the SOFT-S trial. So I don’t know that we have exactly the data that you’re looking for,” Dr. Fleming said. “Many of these women are obviously at very, very low risk and have done well with 5 years of tamoxifen alone, and I don’t know, even given current guidelines, that extended tamoxifen would add a lot to that.”
Finally, session attendee Richard Gray, professor of medical statistics at the University of Oxford (England), wondered, “What is the certainty that follow-up will happen? Because obviously, prolonged follow-up is expensive and there are controversies about that. But this would be the one study you would really want to have 15- and 20-year data on.”
“We are working very, very hard on that,” Dr. Fleming replied. “NCI granted additional funds to institutions for prolonging follow-up, and IBCSG has been ceaselessly working to look for funding to continue it. So I think it’s relatively certain that it will happen.”
Dr. Francis disclosed that she has received fees for non-CME services from AstraZeneca and has given an overseas lecture for Pfizer. Dr. Fleming disclosed that she had no relevant financial relationships with commercial interests. The trials received financial support from Pfizer and Ipsen.
SOURCES: Francis et al. SABCS Abstract GS4-02; Fleming et al. SABCS Abstract GS4-03
SAN ANTONIO – Adjuvant endocrine therapies improve outcomes of premenopausal breast cancer in the long term, with absolute benefit varying somewhat by therapy and by patient and disease characteristics, according to planned updates of a pair of pivotal phase 3 trials.
The trials – TEXT (Tamoxifen and Exemestane Trial) and SOFT (Suppression of Ovarian Function Trial) – are coordinated by the International Breast Cancer Study Group and together randomized more than 5,000 premenopausal women with early hormone receptor–positive breast cancer to 5 years of various types of adjuvant endocrine therapy. Their initial results, reported several years ago, form part of treatment guidelines that are used worldwide.
Relative benefits for various outcomes were generally similar across subgroups, but absolute benefits were greater for women having certain features increasing risk for poor outcomes.
Clinical implications
These updates, along with other emerging data, can be used to optimize endocrine therapy for younger women with breast cancer, according to invited discussant Ann H. Partridge, MD, of Dana Farber Cancer Institute in Boston.
“For higher-risk disease, we should be considering OFS. At this point in time, I don’t think HER2 status alone should drive this decision,” she commented. “If you are getting OFS, what do we do, AI versus tamoxifen? Well, we do see a large improvement in disease-free survival [with AIs], so many women will want to use AIs. Yet tamoxifen is still reasonable, especially in light of the survival data.”
Data on switch strategies and extended-duration therapy are generally lacking at present for the premenopausal population, Dr. Partridge noted. “That’s something that we still need to extrapolate from data that’s predominantly in postmenopausal women.”
Another compelling question is whether OFS can be used instead of chemo for some patients. “We are increasingly recognizing that women with higher-risk anatomy and lower-risk biology having endocrine-responsive tumors may get more bang for the buck from the optimizing of hormonal therapy, and chemo may not add much,” she said.
Both short- and long-term toxicities of the various endocrine therapies and, for aromatase inhibitors, the potential for breakthrough (return of estradiol levels to premenopausal levels) also need to be considered, Dr. Partridge stressed. “And ultimately, patient preference and tolerance are key. After all, the best treatment is the one the patient will take.”
“We need to follow these women on TEXT and SOFT very long term. It would be a crime not to follow these women further out,” she maintained. “We need to conduct real-world comparative effectiveness research to understand the risks and benefits of OFS more fully in our survivors. Then, as we start to suppress more ovaries in more women with breast cancer, we need to be aware clinically of these risks, and we need to share this awareness with their primary care providers because we need to optimize in particular their cardiovascular risk factors, and screen and treat for potential comorbidities that they may be at higher risk for.”
Joint TEXT and SOFT update
Initial results of the joint TEXT and SOFT analysis, reported after a median follow-up of 5.7 years, showed that exemestane plus OFS was superior to tamoxifen plus OFS for the primary outcome, providing a significant 3.8% absolute gain in 5-year disease-free survival (N Engl J Med. 2014;371:107-18).
The updated joint analysis, now with a median follow-up of 9 years and based on data from 4,690 women, showed that the 8-year rate of disease-free survival was 86.8% with exemestane plus OFS versus 82.8% with tamoxifen plus OFS (hazard ratio, 0.77; P = .0006), for a similar absolute benefit of 4.0%, reported Prudence Francis, MD, of the University of Melbourne, head of Medical Oncology in the Breast Service at the Peter MacCallum Cancer Centre, Melbourne.
In stratified analysis, absolute benefit tended to be greater among women in TEXT who received chemotherapy (6.0%); intermediate among women in TEXT who did not receive chemotherapy (3.7%) and women in SOFT who received prior chemotherapy (3.7%); and less among women in SOFT who did not receive chemotherapy (1.9%).
Exemestane plus OFS was also superior to tamoxifen plus OFS in terms of breast cancer–free interval, with an absolute 4.1% benefit (P = .0002), and distant recurrence–free interval, with an absolute 2.1% benefit (P = .02). Overall survival did not differ significantly between arms.
Among the 86% of patients with HER2-negative disease, exemestane plus OFS netted an absolute disease-free survival gain of 5.4% and an absolute distant recurrence–free interval gain of 3.4%. There was a consistent relative treatment benefit across subgroups, but larger absolute benefit, on the order of 5%-9%, in women given chemotherapy and in those younger than 35 years.
“Results for the HER2-positive subgroup require further investigation,” Dr. Francis said. “The trials enrolled both before and after the routine use of adjuvant trastuzumab, and a significant proportion of the patients with HER2-positive breast cancer did not receive adjuvant HER2-targeted therapy.”
In the entire joint-analysis population, exemestane plus OFS was associated with higher rates of musculoskeletal events of grade 3 or 4 (11% vs. 6%) and osteoporosis of grade 2-4 (15% vs. 7%), while tamoxifen plus OFS was associated with a higher rate of thrombosis/embolism of grade 2-4 (2.3% vs. 1.2%) and more cases of endometrial cancer (9 vs. 4 cases). At 4 years, early discontinuation of oral endocrine therapy was greater for exemestane than for tamoxifen (25% vs. 19%).
“After longer follow-up, with a median of 9 years, the combined analysis results confirm a statistically significant improvement in disease outcomes with exemestane plus ovarian suppression. As is critical given the long natural history of estrogen receptor–positive breast cancer, follow-up in these trials is currently continuing,” Dr. Francis summarized.
“To optimally translate the observed absolute trial improvements into clinical practice, oncologists need to discuss and weigh the potential benefits and toxicities in each individual patient who is premenopausal with hormone receptor–positive breast cancer,” she recommended.
Session attendee Hope S. Rugo, MD, of the University of California, San Francisco, Helen Diller Family Comprehensive Cancer Center, noted that exemestane had superior benefit despite the 25% rate of early discontinuation. “I wonder if one of the interpretations of that, given the toxicity of this therapy for very young women, is that we need some but maybe not so much. Maybe they don’t need 5 years altogether,” she said.
“The fact that they stopped their assigned endocrine therapy doesn’t mean that they didn’t continue any therapy. They may have switched over to tamoxifen or they may have decided they wanted to have a baby or there may have been many other things,” Dr. Francis replied, noting that analyses sorting out the reasons for early discontinuation are planned.
Session attendee Mark E. Sherman, MD, of the Mayo Clinic, Jacksonville, Fla., asked, “Do you have any ability to test for tamoxifen metabolites? It’s possible that a third to a half of patients got reduced benefit from that drug.”
Banked samples are available and a substudy is planned, according to Dr. Francis. “We haven’t got data on that yet, but yes, we are analyzing that.”
SOFT update
Initial results of the SOFT trial, reported after a median follow-up of 5.6 years, showed that adding OFS to tamoxifen did not significantly improve disease-free survival over tamoxifen alone in the entire trial population (N Engl J Med. 2015;372:436-46). However, there was benefit for women who received chemotherapy and remained premenopausal.
The updated SOFT analysis, now with a median follow-up of 8 years, focused mainly on the 1,018 women given tamoxifen alone and the 1,015 women given tamoxifen plus OFS. (Another 1,014 women were given exemestane plus OFS.)
“SOFT is now positive for its primary endpoint,” reported first author Gini Fleming, MD, director of the Medical Oncology Breast Program and medical oncology director of Gynecologic Oncology at University of Chicago Medicine. The 8-year disease-free survival rate was 83.2% with tamoxifen plus OFS, compared with 78.9% with tamoxifen alone (HR, 0.76; P = .009), corresponding to a 4.2% gain in this outcome. The relative benefit was identical whether patients had received chemotherapy or not, but absolute benefit was greater for those who had (5.3%), as well as for patients younger than 35 years (8.7%).
In addition, exemestane plus OFS was superior to tamoxifen alone (85.9% vs. 78.9%; HR, 0.65), with an absolute benefit of 7.0%. Again, absolute benefit was more pronounced among women who had received prior chemotherapy (9.0%) or were younger than 35 years (13.1%).
The relative disease-free survival benefit of tamoxifen plus OFS over tamoxifen alone was similar across most subgroups stratified by disease characteristics, but patients with HER2-positive disease derived greater relative benefit from the combination as compared with their HER2-negative counterparts (P = .04 for interaction). “When we look at the combination of exemestane plus OFS versus tamoxifen, this heterogeneity is no longer seen,” Dr. Fleming noted.
In the entire trial population, there was no significant benefit of tamoxifen plus OFS over tamoxifen alone for distant recurrence-free interval and a small, significant absolute 1.9% gain in overall survival.
“The cohort who had elected to receive no prior chemotherapy did exceedingly well regardless of therapy,” she said, with little difference in overall survival across the three arms. “There were only 24 deaths total in this cohort, and 12 of those deaths were in the setting of no distant recurrence.”
On the other hand, among the women who received chemotherapy, there were significant absolute overall survival benefits of 4.3% with tamoxifen plus OFS and 2.1% with exemestane plus OFS, over tamoxifen alone. “This late emergence of an overall survival benefit is consistent with the time course of events in estrogen receptor–positive breast cancer,” Dr. Fleming commented.
The proportion of patients who stopped their oral endocrine therapy early was 22.5% with tamoxifen alone and 18.5% with tamoxifen plus OFS. (It was 27.8% with exemestane plus OFS.) “Almost a quarter of the patients on either tamoxifen arm were using extended oral endocrine therapy at 6 years or later prior to any disease progression. Only about 12% of patients in the exemestane group were doing so,” she noted.
There were more cases of endometrial cancer with tamoxifen alone than with tamoxifen plus OFS (7 vs. 4 cases). Thrombosis/embolism of grade 2-4 occurred in 2.2% of each group (and 0.9% of the exemestane plus OFS group). Musculoskeletal symptoms of grade 3 or 4 occurred in 6.7% of patients with tamoxifen alone and 5.9% with tamoxifen plus OFS, but 12.0% with exemestane plus OFS. Respective rates of osteoporosis grade 2-4 were 3.9%, 6.1%, and 11.9%.
“The addition of OFS to tamoxifen significantly improves disease-free survival at 8 years’ median follow-up, and disease-free survival benefits are further improved by the use of exemestane plus OFS,” Dr. Fleming summarized. “Follow-up, which is critically important given the long natural history of ER-positive disease, continues.”
Session attendee Matthew P. Goetz, MD, of the Mayo Clinic, Rochester, Minn., commented, “For the primary endpoint, I was looking at the tail for tamoxifen. It seemed that there was a relatively rapid drop-off between year 5 and this 8-year follow-up. Have you looked carefully to see whether there is a difference between those who stayed on their therapy versus those who went off it per protocol? That is, extended versus not extended? The question is whether there is a carry-over effect, if you will, that is different in those with OFS versus those not.”
“The percent who went on to extended therapy between the tamoxifen and the tamoxifen plus OFS was fairly similar,” Dr. Fleming replied. “But the answer is no, we have not yet done any sort of per protocol analysis.”
Session attendee Steven Vogl, MD, of Montefiore Medical Center, New York, commented, “I worry about your control group. I’m worried, first, how many of your tamoxifen patients lost their menses and became postmenopausal in those 5 years? And of those, why didn’t they switch to an aromatase inhibitor? Only 25% of the patients continued after the 5 years according to your slide, and all of those patients should either have stayed on tamoxifen or switched to an aromatase inhibitor, now probably for 2 years at least.”
“We have not yet looked at data for who became amenorrheic during treatment, although we have it. However, it’s certainly possible to become amenorrheic on tamoxifen and not be postmenopausal, and we didn’t regularly collect estradiol levels on any but the very small subset of women in the SOFT-S trial. So I don’t know that we have exactly the data that you’re looking for,” Dr. Fleming said. “Many of these women are obviously at very, very low risk and have done well with 5 years of tamoxifen alone, and I don’t know, even given current guidelines, that extended tamoxifen would add a lot to that.”
Finally, session attendee Richard Gray, professor of medical statistics at the University of Oxford (England), wondered, “What is the certainty that follow-up will happen? Because obviously, prolonged follow-up is expensive and there are controversies about that. But this would be the one study you would really want to have 15- and 20-year data on.”
“We are working very, very hard on that,” Dr. Fleming replied. “NCI granted additional funds to institutions for prolonging follow-up, and IBCSG has been ceaselessly working to look for funding to continue it. So I think it’s relatively certain that it will happen.”
Dr. Francis disclosed that she has received fees for non-CME services from AstraZeneca and has given an overseas lecture for Pfizer. Dr. Fleming disclosed that she had no relevant financial relationships with commercial interests. The trials received financial support from Pfizer and Ipsen.
SOURCES: Francis et al. SABCS Abstract GS4-02; Fleming et al. SABCS Abstract GS4-03
REPORTING FROM SABCS 2017
Key clinical point:
Major finding: In the joint TEXT-SOFT update, 8-year disease-free survival was superior with exemestane plus OFS versus tamoxifen plus OFS (86.8% vs. 82.8%; P = .0006). In the SOFT update, 8-year disease-free survival was superior with tamoxifen plus OFS versus tamoxifen alone (83.2% vs. 78.9%; P = .009).
Data source: Updated analyses of phase 3 trials among premenopausal women with HR-positive breast cancer: TEXT and SOFT joint analysis (n = 4,690; median 9-year follow-up) and SOFT analysis (n = 3,047; median 8-year follow-up).
Disclosures: Dr. Francis disclosed that she has received fees for non-CME services from AstraZeneca and has given an overseas lecture for Pfizer. Dr. Fleming disclosed that she had no relevant financial relationships with commercial interests. The trials received financial support from Pfizer and Ipsen.
Source: Francis et al. SABCS Abstract GS4-02; Fleming et al. SABCS Abstract GS4-03
New tool predicts late distant recurrence of postmenopausal ER+ breast cancer
SAN ANTONIO – A new prognostic tool that uses four clinical and pathological variables may help to guide decisions about extending adjuvant endocrine therapy for postmenopausal women with estrogen receptor–positive (ER+) breast cancer, according to a study reported at the San Antonio Breast Cancer Symposium.
ER+ breast cancer is well known for recurring long after endocrine therapy stops, but the risk varies widely, ranging from 10% to 40% (N Engl J Med. 2017;377:1836-46), noted lead investigator Ivana Sestak, MS, PhD, a lecturer in medical statistics at the Queen Mary University of London. “A few trials have shown that extended endocrine therapy can reduce the risk of recurrence, but careful assessment of potential side effects and actual risk of developing a late distant recurrence is essential,” she said.
The investigators used the CTS5 to stratify patients into a low-risk group (risk of late distant recurrence less than 5%), an intermediate-risk group (risk between 5% and 10%), and a high-risk group (risk more than 10%). The observed rates of distant recurrence between years 5 and 10 were about 3% for the low-risk group, 7% for the intermediate-risk group, and 19% for the high risk-group. In addition, the CTS5 outperformed the original Clinical Treatment Score (CTS0), which was developed to predict recurrence between 0 and 10 years (J Clin Oncol. 2011;29:4273-8).
“We have developed a simple prognostic tool for the prediction of late distant recurrences which will help clinicians and their patients in the decision-making process about extended endocrine therapy,” Dr. Sestak commented. “The CTS5 was highly prognostic for the prediction of late distant recurrences and identified a large proportion of women, 42%, as low risk, where the value of extended endocrine therapy is limited. The CTS5 was also more prognostic than the already published CTS0 and should be used in this context for the prediction of late distant recurrence.”
“We aim to make the CTS5 algorithm and risk curve, with a read-out table, available to clinicians, and it will also be published in our manuscript,” she added.
Session attendee Frankie Ann Holmes, MD, of the Texas Oncology/US Oncology Network in Houston commented, “Just identifying high risk doesn’t necessarily translate into benefit, which is what we see with the Breast Cancer Index: You get the high risk, but then you learn if there is actually benefit to the extended therapy. Does your assay have a benefit portion to it?”
“No, we can’t look at the predictive benefit [with the CTS5]. This assay is purely a prognostic tool to predict late distant recurrences,” Dr. Sestak replied. “In these two trials, we do not have information on how many patients actually went on to extended endocrine therapy. You have to remember, these are old trials – they finished in about 2007-2008 – so not many women would have been given extended endocrine therapy at that time point.”
Session attendee Laura J. van’t Veer, PhD, of the University of California, San Francisco, asked, “How do you feel this will translate for risk up to 20 years, for which the question of extended endocrine therapy might also be very relevant?”
“For the purpose of this analysis, we only looked at out to 10 years. But I agree, it’s also important if we could apply a prognostic tool out to 20 years,” Dr. Sestak replied. “We have longer follow-up on some of the ATAC women, and we might look into that to see if we see any benefit of using a prognostic tool in the prediction of late distant recurrences.”
Study details
The investigators developed and trained the new tool using data from 4,735 women from the ATAC trial. They then validated the tool using data from 6,711 women from the BIG 1-98 trial.
The final CTS5 model contained four clinical variables, Dr. Sestak reported: number of involved nodes, size of the tumor, grade of the tumor, and age of the patient.
In the ATAC population, the CTS5 model did a better job than the original CTS0 model of predicting late distant recurrence. CTS5 improved the prediction of late distant recurrence by a factor of 2.47, whereas CTS0 improved the predictive value by a factor of 2.04. The CTS5 model performed similarly well regardless of whether patients had received chemotherapy.
In the BIG 1-98 population, the findings were much the same: The CTS5 model improved prediction of late distant recurrence by 2.07, while the CTS0 model improved prediction of late distant recurrence by 1.84. Performance of the CTS5 model was again similarly good regardless of whether patients had received chemotherapy.
Observed rates of distant recurrence between years 5 and 10 were similar in the ATAC and BIG 1-98 populations for the CTS5-defined low-risk group (2.5% and 3.0%, respectively), intermediate-risk group (7.7% and 6.9%), and the high-risk group (20.3% and 17.3%).
When the two trials’ populations were combined, the observed rate was 3.0% in the CTS5-defined low-risk group, 7.3% in the intermediate-risk group, and 18.9% in the high-risk group.
In addition, the main results held up among all node-negative women combined and among all women who had between one and three positive nodes combined. “For women with four or more positive lymph nodes, the CTS5 was not informative and categorized virtually all women into the high-risk group,” Dr. Sestak noted.
The investigators did not look at whether local or regional recurrences modulated the risk of late distant recurrence, she said. However, women who had experienced isolated local recurrence during the first 5 years would have been included in analysis.
“A strength of our study is that we used clinicopathological parameters that are measured in all breast cancer patients, and there is no need for further testing,” noted Dr. Sestak, who disclosed that she has received fees for advisory boards and lectures from Myriad Genetics.
On the other hand, it is unclear how the CTS5 would perform among premenopausal women and among women with HER2-positive disease given that two trials took place before routine HER2 testing and HER2-directed therapy were used.
SOURCE: Sestak I et al. SABCS 2017 Abstract GS6-01.
SAN ANTONIO – A new prognostic tool that uses four clinical and pathological variables may help to guide decisions about extending adjuvant endocrine therapy for postmenopausal women with estrogen receptor–positive (ER+) breast cancer, according to a study reported at the San Antonio Breast Cancer Symposium.
ER+ breast cancer is well known for recurring long after endocrine therapy stops, but the risk varies widely, ranging from 10% to 40% (N Engl J Med. 2017;377:1836-46), noted lead investigator Ivana Sestak, MS, PhD, a lecturer in medical statistics at the Queen Mary University of London. “A few trials have shown that extended endocrine therapy can reduce the risk of recurrence, but careful assessment of potential side effects and actual risk of developing a late distant recurrence is essential,” she said.
The investigators used the CTS5 to stratify patients into a low-risk group (risk of late distant recurrence less than 5%), an intermediate-risk group (risk between 5% and 10%), and a high-risk group (risk more than 10%). The observed rates of distant recurrence between years 5 and 10 were about 3% for the low-risk group, 7% for the intermediate-risk group, and 19% for the high risk-group. In addition, the CTS5 outperformed the original Clinical Treatment Score (CTS0), which was developed to predict recurrence between 0 and 10 years (J Clin Oncol. 2011;29:4273-8).
“We have developed a simple prognostic tool for the prediction of late distant recurrences which will help clinicians and their patients in the decision-making process about extended endocrine therapy,” Dr. Sestak commented. “The CTS5 was highly prognostic for the prediction of late distant recurrences and identified a large proportion of women, 42%, as low risk, where the value of extended endocrine therapy is limited. The CTS5 was also more prognostic than the already published CTS0 and should be used in this context for the prediction of late distant recurrence.”
“We aim to make the CTS5 algorithm and risk curve, with a read-out table, available to clinicians, and it will also be published in our manuscript,” she added.
Session attendee Frankie Ann Holmes, MD, of the Texas Oncology/US Oncology Network in Houston commented, “Just identifying high risk doesn’t necessarily translate into benefit, which is what we see with the Breast Cancer Index: You get the high risk, but then you learn if there is actually benefit to the extended therapy. Does your assay have a benefit portion to it?”
“No, we can’t look at the predictive benefit [with the CTS5]. This assay is purely a prognostic tool to predict late distant recurrences,” Dr. Sestak replied. “In these two trials, we do not have information on how many patients actually went on to extended endocrine therapy. You have to remember, these are old trials – they finished in about 2007-2008 – so not many women would have been given extended endocrine therapy at that time point.”
Session attendee Laura J. van’t Veer, PhD, of the University of California, San Francisco, asked, “How do you feel this will translate for risk up to 20 years, for which the question of extended endocrine therapy might also be very relevant?”
“For the purpose of this analysis, we only looked at out to 10 years. But I agree, it’s also important if we could apply a prognostic tool out to 20 years,” Dr. Sestak replied. “We have longer follow-up on some of the ATAC women, and we might look into that to see if we see any benefit of using a prognostic tool in the prediction of late distant recurrences.”
Study details
The investigators developed and trained the new tool using data from 4,735 women from the ATAC trial. They then validated the tool using data from 6,711 women from the BIG 1-98 trial.
The final CTS5 model contained four clinical variables, Dr. Sestak reported: number of involved nodes, size of the tumor, grade of the tumor, and age of the patient.
In the ATAC population, the CTS5 model did a better job than the original CTS0 model of predicting late distant recurrence. CTS5 improved the prediction of late distant recurrence by a factor of 2.47, whereas CTS0 improved the predictive value by a factor of 2.04. The CTS5 model performed similarly well regardless of whether patients had received chemotherapy.
In the BIG 1-98 population, the findings were much the same: The CTS5 model improved prediction of late distant recurrence by 2.07, while the CTS0 model improved prediction of late distant recurrence by 1.84. Performance of the CTS5 model was again similarly good regardless of whether patients had received chemotherapy.
Observed rates of distant recurrence between years 5 and 10 were similar in the ATAC and BIG 1-98 populations for the CTS5-defined low-risk group (2.5% and 3.0%, respectively), intermediate-risk group (7.7% and 6.9%), and the high-risk group (20.3% and 17.3%).
When the two trials’ populations were combined, the observed rate was 3.0% in the CTS5-defined low-risk group, 7.3% in the intermediate-risk group, and 18.9% in the high-risk group.
In addition, the main results held up among all node-negative women combined and among all women who had between one and three positive nodes combined. “For women with four or more positive lymph nodes, the CTS5 was not informative and categorized virtually all women into the high-risk group,” Dr. Sestak noted.
The investigators did not look at whether local or regional recurrences modulated the risk of late distant recurrence, she said. However, women who had experienced isolated local recurrence during the first 5 years would have been included in analysis.
“A strength of our study is that we used clinicopathological parameters that are measured in all breast cancer patients, and there is no need for further testing,” noted Dr. Sestak, who disclosed that she has received fees for advisory boards and lectures from Myriad Genetics.
On the other hand, it is unclear how the CTS5 would perform among premenopausal women and among women with HER2-positive disease given that two trials took place before routine HER2 testing and HER2-directed therapy were used.
SOURCE: Sestak I et al. SABCS 2017 Abstract GS6-01.
SAN ANTONIO – A new prognostic tool that uses four clinical and pathological variables may help to guide decisions about extending adjuvant endocrine therapy for postmenopausal women with estrogen receptor–positive (ER+) breast cancer, according to a study reported at the San Antonio Breast Cancer Symposium.
ER+ breast cancer is well known for recurring long after endocrine therapy stops, but the risk varies widely, ranging from 10% to 40% (N Engl J Med. 2017;377:1836-46), noted lead investigator Ivana Sestak, MS, PhD, a lecturer in medical statistics at the Queen Mary University of London. “A few trials have shown that extended endocrine therapy can reduce the risk of recurrence, but careful assessment of potential side effects and actual risk of developing a late distant recurrence is essential,” she said.
The investigators used the CTS5 to stratify patients into a low-risk group (risk of late distant recurrence less than 5%), an intermediate-risk group (risk between 5% and 10%), and a high-risk group (risk more than 10%). The observed rates of distant recurrence between years 5 and 10 were about 3% for the low-risk group, 7% for the intermediate-risk group, and 19% for the high risk-group. In addition, the CTS5 outperformed the original Clinical Treatment Score (CTS0), which was developed to predict recurrence between 0 and 10 years (J Clin Oncol. 2011;29:4273-8).
“We have developed a simple prognostic tool for the prediction of late distant recurrences which will help clinicians and their patients in the decision-making process about extended endocrine therapy,” Dr. Sestak commented. “The CTS5 was highly prognostic for the prediction of late distant recurrences and identified a large proportion of women, 42%, as low risk, where the value of extended endocrine therapy is limited. The CTS5 was also more prognostic than the already published CTS0 and should be used in this context for the prediction of late distant recurrence.”
“We aim to make the CTS5 algorithm and risk curve, with a read-out table, available to clinicians, and it will also be published in our manuscript,” she added.
Session attendee Frankie Ann Holmes, MD, of the Texas Oncology/US Oncology Network in Houston commented, “Just identifying high risk doesn’t necessarily translate into benefit, which is what we see with the Breast Cancer Index: You get the high risk, but then you learn if there is actually benefit to the extended therapy. Does your assay have a benefit portion to it?”
“No, we can’t look at the predictive benefit [with the CTS5]. This assay is purely a prognostic tool to predict late distant recurrences,” Dr. Sestak replied. “In these two trials, we do not have information on how many patients actually went on to extended endocrine therapy. You have to remember, these are old trials – they finished in about 2007-2008 – so not many women would have been given extended endocrine therapy at that time point.”
Session attendee Laura J. van’t Veer, PhD, of the University of California, San Francisco, asked, “How do you feel this will translate for risk up to 20 years, for which the question of extended endocrine therapy might also be very relevant?”
“For the purpose of this analysis, we only looked at out to 10 years. But I agree, it’s also important if we could apply a prognostic tool out to 20 years,” Dr. Sestak replied. “We have longer follow-up on some of the ATAC women, and we might look into that to see if we see any benefit of using a prognostic tool in the prediction of late distant recurrences.”
Study details
The investigators developed and trained the new tool using data from 4,735 women from the ATAC trial. They then validated the tool using data from 6,711 women from the BIG 1-98 trial.
The final CTS5 model contained four clinical variables, Dr. Sestak reported: number of involved nodes, size of the tumor, grade of the tumor, and age of the patient.
In the ATAC population, the CTS5 model did a better job than the original CTS0 model of predicting late distant recurrence. CTS5 improved the prediction of late distant recurrence by a factor of 2.47, whereas CTS0 improved the predictive value by a factor of 2.04. The CTS5 model performed similarly well regardless of whether patients had received chemotherapy.
In the BIG 1-98 population, the findings were much the same: The CTS5 model improved prediction of late distant recurrence by 2.07, while the CTS0 model improved prediction of late distant recurrence by 1.84. Performance of the CTS5 model was again similarly good regardless of whether patients had received chemotherapy.
Observed rates of distant recurrence between years 5 and 10 were similar in the ATAC and BIG 1-98 populations for the CTS5-defined low-risk group (2.5% and 3.0%, respectively), intermediate-risk group (7.7% and 6.9%), and the high-risk group (20.3% and 17.3%).
When the two trials’ populations were combined, the observed rate was 3.0% in the CTS5-defined low-risk group, 7.3% in the intermediate-risk group, and 18.9% in the high-risk group.
In addition, the main results held up among all node-negative women combined and among all women who had between one and three positive nodes combined. “For women with four or more positive lymph nodes, the CTS5 was not informative and categorized virtually all women into the high-risk group,” Dr. Sestak noted.
The investigators did not look at whether local or regional recurrences modulated the risk of late distant recurrence, she said. However, women who had experienced isolated local recurrence during the first 5 years would have been included in analysis.
“A strength of our study is that we used clinicopathological parameters that are measured in all breast cancer patients, and there is no need for further testing,” noted Dr. Sestak, who disclosed that she has received fees for advisory boards and lectures from Myriad Genetics.
On the other hand, it is unclear how the CTS5 would perform among premenopausal women and among women with HER2-positive disease given that two trials took place before routine HER2 testing and HER2-directed therapy were used.
SOURCE: Sestak I et al. SABCS 2017 Abstract GS6-01.
REPORTING FROM SABCS 2017
Key clinical point:
Major finding: The tool stratified patients for risk of distant recurrence between years 5 and 10 as low risk (less than 5% risk), intermediate risk (5%-10% risk), and high risk (more than 10% risk).
Data source: A cohort study of 11,446 postmenopausal women with early-stage breast cancer who were free of distant recurrence after 5 years of adjuvant endocrine therapy.
Disclosures: Dr. Sestak disclosed that she has received fees for advisory boards and lectures from Myriad Genetics.
Source: Sestak I et al. SABCS 2017 Abstract GS6-01.
EndoPredict results reflected tumor response to neoadjuvant therapy
SAN ANTONIO – The results of the EndoPredict test appear to predict tumor response in patients with early hormone receptor–positive, HER2-negative breast cancer given neoadjuvant therapy, based on results of a study conducted by the Austrian Breast & Colorectal Cancer Study Group (ABCSG).
“Very good tumor shrinkage in estrogen receptor–positive, HER2-negative disease is going to happen only in a minority of patients, and biomarkers that would predict excellent tumor shrinkage are an unmet medical need,” commented lead investigator Peter Dubsky, MD, PhD, who is head of the Breast Center at Hirslanden Klinik St. Anna, Lucerne, Switzerland. “As a surgeon, that would help me to predict breast conservation at diagnosis, but as a surgical oncologist, I would also recognize that tumor response is an important component of future survival.”
The ABCSG findings suggest expanded utility for EndoPredict. The test’s molecular score is currently used along with tumor size and nodal status to predict the 10-year distant recurrence rate, and whether patients may safely forgo chemotherapy or are at high risk and may need adjuvant chemotherapy in addition to endocrine therapy.
Dr. Dubsky and his coinvestigators assessed performance of the EndoPredict test among 217 patients treated on ABCSG 34, a randomized phase 2 neoadjuvant trial. Findings showed that among patients given neoadjuvant endocrine therapy because they had less aggressive disease features, an EndoPredict high-risk result was associated with poor response (negative predictive value of 92%), defined as a residual cancer burden (RCB) of II or III, he reported at the San Antonio Breast Cancer Symposium.
On the other hand, among patients given neoadjuvant chemotherapy because they had more aggressive disease features, a low-risk result was associated with poor response (negative predictive value of 100%).
“Clinicians really gave us two distinct cohorts within ABCSG 34. In the luminal A–type patients who were treated with neoendocrine therapy, a high EndoPredict score predicted a low chance of tumor shrinkage. In the more aggressive ER-positive tumors, so-called luminal B type, treated with neoadjuvant chemotherapy, there was absolutely no excellent response in the low-risk group,” Dr. Dubsky summarized. “We believe that this molecular score may contribute to patient selection for biomarker-driven studies, especially in the neoadjuvant setting.”
Session attendee Steven Vogl, MD, a medical oncologist with the Montefiore Medical Center in New York, commented, “I have trouble correlating an RCB of 0 or I with what you as a surgeon do for the patient, because you are talking about pathologic complete response or just a few cells there. That’s not what determines how much breast you take off: It’s determined by the total size of the tumor and the size of the breast. So if it’s less than a few centimeters, I’m sure you can do a lumpectomy in every patient. Tell me why I should care that you are getting an RCB of 0 or I in these endocrine patients.”
“Because it’s more likely that these patients will have a smaller tumor and better tumor shrinkage,” Dr. Dubsky replied. “You are of course right, RCB 0 or I was not designed to help surgeons. But it helps me as a translational scientist to have a surrogate and an exact classification for good tumor shrinkage. That’s how I used it.”
C. Kent Osborne, MD, codirector of SABCS and director of the Dan L. Duncan Cancer Center at Baylor College of Medicine in Houston, asked, “We see it in the clinic, and I’m sure you have as well, patients whose tumor doesn’t shrink very much, but the Ki-67 really drops. And that may or may not be a better factor than the actual tumor shrinkage. So how many patients who had tumors that didn’t shrink, which was your endpoint, had a reduction in Ki-67 that was, say, 5%?”
“We haven’t looked at that specifically, but we will do so as we carry on with the follow-up of these patients. Then we can learn more about the prognosis,” Dr. Dubsky replied.
Study details
ABCSG 34 was a randomized phase 2 trial testing addition of the cancer vaccine tecemotide (Stimuvax) to neoadjuvant standard of care among patients with HER2-negative early breast cancer.
Dr. Dubsky and coinvestigators restricted analyses to patients with hormone receptor–positive disease who, depending on clinical and pathologic factors, received neoadjuvant chemotherapy (eight cycles of epirubicin-cyclophosphamide and docetaxel) or neoadjuvant endocrine therapy (6 months of letrozole [Femara]) as standard of care. They were then randomized to additionally receive tecemotide or not before undergoing surgery.
Overall, 25% of the 134 patients in the neoadjuvant chemotherapy group had a good tumor response, defined as pathologic complete response in both breast and nodes (RCB of 0) or minimal residual disease (RCB of I).
Higher EndoPredict score was associated with greater likelihood of good response to chemotherapy. EndoPredict risk group (high vs. low) had a negative predictive value of 100%, a positive predictive value of 26.4%, a true-positive rate of 100%, and a true-negative rate of 8.9% for predicting response (P = .112).
Area under the receiver operating characteristic curve was 0.736.
In a multivariate model, EndoPredict score as a continuous variable was not an independent predictor of response. “The good response was largely driven by covariates that included cell proliferation, and it was Ki-67 that was significant,” Dr. Dubsky noted.
Overall, 18% of the 83 patients in the neoadjuvant endocrine therapy group had a good tumor response (RCB of 0 or I). Here, lower EndoPredict score was associated with greater likelihood of good response. EndoPredict risk group (high vs. low) had a negative predictive value of 92.3%, a positive predictive value of 27.3%, a true-positive rate of 80.0%, and a true-negative rate of 52.9% for predicting response (P = .024). Area under the curve was 0.726.
In a multivariate model here, EndoPredict score as a continuous variable, its estrogen receptor–signaling/differentiation component, and Ki-67 did not independently predict response. “It was maybe a bit surprising that T stage was the strongest factor, possibly indicating that we should have simply treated those women longer than 6 months,” Dr. Dubsky commented. The EndoPredict proliferation component was also a significant predictor.
“Possibly, the very narrow distribution of Ki-67 [among patients given neoendocrine therapy] may have prevented this factor from playing a bigger role in this particular model,” he speculated.
Dr. Dubsky disclosed that he receives consulting fees from Myriad, the maker of EndoPredict, and from Cepheid, Nanostring, and Amgen.
SOURCE: Dubsky P et al. SABCS 2017 Abstract GS6-04.
SAN ANTONIO – The results of the EndoPredict test appear to predict tumor response in patients with early hormone receptor–positive, HER2-negative breast cancer given neoadjuvant therapy, based on results of a study conducted by the Austrian Breast & Colorectal Cancer Study Group (ABCSG).
“Very good tumor shrinkage in estrogen receptor–positive, HER2-negative disease is going to happen only in a minority of patients, and biomarkers that would predict excellent tumor shrinkage are an unmet medical need,” commented lead investigator Peter Dubsky, MD, PhD, who is head of the Breast Center at Hirslanden Klinik St. Anna, Lucerne, Switzerland. “As a surgeon, that would help me to predict breast conservation at diagnosis, but as a surgical oncologist, I would also recognize that tumor response is an important component of future survival.”
The ABCSG findings suggest expanded utility for EndoPredict. The test’s molecular score is currently used along with tumor size and nodal status to predict the 10-year distant recurrence rate, and whether patients may safely forgo chemotherapy or are at high risk and may need adjuvant chemotherapy in addition to endocrine therapy.
Dr. Dubsky and his coinvestigators assessed performance of the EndoPredict test among 217 patients treated on ABCSG 34, a randomized phase 2 neoadjuvant trial. Findings showed that among patients given neoadjuvant endocrine therapy because they had less aggressive disease features, an EndoPredict high-risk result was associated with poor response (negative predictive value of 92%), defined as a residual cancer burden (RCB) of II or III, he reported at the San Antonio Breast Cancer Symposium.
On the other hand, among patients given neoadjuvant chemotherapy because they had more aggressive disease features, a low-risk result was associated with poor response (negative predictive value of 100%).
“Clinicians really gave us two distinct cohorts within ABCSG 34. In the luminal A–type patients who were treated with neoendocrine therapy, a high EndoPredict score predicted a low chance of tumor shrinkage. In the more aggressive ER-positive tumors, so-called luminal B type, treated with neoadjuvant chemotherapy, there was absolutely no excellent response in the low-risk group,” Dr. Dubsky summarized. “We believe that this molecular score may contribute to patient selection for biomarker-driven studies, especially in the neoadjuvant setting.”
Session attendee Steven Vogl, MD, a medical oncologist with the Montefiore Medical Center in New York, commented, “I have trouble correlating an RCB of 0 or I with what you as a surgeon do for the patient, because you are talking about pathologic complete response or just a few cells there. That’s not what determines how much breast you take off: It’s determined by the total size of the tumor and the size of the breast. So if it’s less than a few centimeters, I’m sure you can do a lumpectomy in every patient. Tell me why I should care that you are getting an RCB of 0 or I in these endocrine patients.”
“Because it’s more likely that these patients will have a smaller tumor and better tumor shrinkage,” Dr. Dubsky replied. “You are of course right, RCB 0 or I was not designed to help surgeons. But it helps me as a translational scientist to have a surrogate and an exact classification for good tumor shrinkage. That’s how I used it.”
C. Kent Osborne, MD, codirector of SABCS and director of the Dan L. Duncan Cancer Center at Baylor College of Medicine in Houston, asked, “We see it in the clinic, and I’m sure you have as well, patients whose tumor doesn’t shrink very much, but the Ki-67 really drops. And that may or may not be a better factor than the actual tumor shrinkage. So how many patients who had tumors that didn’t shrink, which was your endpoint, had a reduction in Ki-67 that was, say, 5%?”
“We haven’t looked at that specifically, but we will do so as we carry on with the follow-up of these patients. Then we can learn more about the prognosis,” Dr. Dubsky replied.
Study details
ABCSG 34 was a randomized phase 2 trial testing addition of the cancer vaccine tecemotide (Stimuvax) to neoadjuvant standard of care among patients with HER2-negative early breast cancer.
Dr. Dubsky and coinvestigators restricted analyses to patients with hormone receptor–positive disease who, depending on clinical and pathologic factors, received neoadjuvant chemotherapy (eight cycles of epirubicin-cyclophosphamide and docetaxel) or neoadjuvant endocrine therapy (6 months of letrozole [Femara]) as standard of care. They were then randomized to additionally receive tecemotide or not before undergoing surgery.
Overall, 25% of the 134 patients in the neoadjuvant chemotherapy group had a good tumor response, defined as pathologic complete response in both breast and nodes (RCB of 0) or minimal residual disease (RCB of I).
Higher EndoPredict score was associated with greater likelihood of good response to chemotherapy. EndoPredict risk group (high vs. low) had a negative predictive value of 100%, a positive predictive value of 26.4%, a true-positive rate of 100%, and a true-negative rate of 8.9% for predicting response (P = .112).
Area under the receiver operating characteristic curve was 0.736.
In a multivariate model, EndoPredict score as a continuous variable was not an independent predictor of response. “The good response was largely driven by covariates that included cell proliferation, and it was Ki-67 that was significant,” Dr. Dubsky noted.
Overall, 18% of the 83 patients in the neoadjuvant endocrine therapy group had a good tumor response (RCB of 0 or I). Here, lower EndoPredict score was associated with greater likelihood of good response. EndoPredict risk group (high vs. low) had a negative predictive value of 92.3%, a positive predictive value of 27.3%, a true-positive rate of 80.0%, and a true-negative rate of 52.9% for predicting response (P = .024). Area under the curve was 0.726.
In a multivariate model here, EndoPredict score as a continuous variable, its estrogen receptor–signaling/differentiation component, and Ki-67 did not independently predict response. “It was maybe a bit surprising that T stage was the strongest factor, possibly indicating that we should have simply treated those women longer than 6 months,” Dr. Dubsky commented. The EndoPredict proliferation component was also a significant predictor.
“Possibly, the very narrow distribution of Ki-67 [among patients given neoendocrine therapy] may have prevented this factor from playing a bigger role in this particular model,” he speculated.
Dr. Dubsky disclosed that he receives consulting fees from Myriad, the maker of EndoPredict, and from Cepheid, Nanostring, and Amgen.
SOURCE: Dubsky P et al. SABCS 2017 Abstract GS6-04.
SAN ANTONIO – The results of the EndoPredict test appear to predict tumor response in patients with early hormone receptor–positive, HER2-negative breast cancer given neoadjuvant therapy, based on results of a study conducted by the Austrian Breast & Colorectal Cancer Study Group (ABCSG).
“Very good tumor shrinkage in estrogen receptor–positive, HER2-negative disease is going to happen only in a minority of patients, and biomarkers that would predict excellent tumor shrinkage are an unmet medical need,” commented lead investigator Peter Dubsky, MD, PhD, who is head of the Breast Center at Hirslanden Klinik St. Anna, Lucerne, Switzerland. “As a surgeon, that would help me to predict breast conservation at diagnosis, but as a surgical oncologist, I would also recognize that tumor response is an important component of future survival.”
The ABCSG findings suggest expanded utility for EndoPredict. The test’s molecular score is currently used along with tumor size and nodal status to predict the 10-year distant recurrence rate, and whether patients may safely forgo chemotherapy or are at high risk and may need adjuvant chemotherapy in addition to endocrine therapy.
Dr. Dubsky and his coinvestigators assessed performance of the EndoPredict test among 217 patients treated on ABCSG 34, a randomized phase 2 neoadjuvant trial. Findings showed that among patients given neoadjuvant endocrine therapy because they had less aggressive disease features, an EndoPredict high-risk result was associated with poor response (negative predictive value of 92%), defined as a residual cancer burden (RCB) of II or III, he reported at the San Antonio Breast Cancer Symposium.
On the other hand, among patients given neoadjuvant chemotherapy because they had more aggressive disease features, a low-risk result was associated with poor response (negative predictive value of 100%).
“Clinicians really gave us two distinct cohorts within ABCSG 34. In the luminal A–type patients who were treated with neoendocrine therapy, a high EndoPredict score predicted a low chance of tumor shrinkage. In the more aggressive ER-positive tumors, so-called luminal B type, treated with neoadjuvant chemotherapy, there was absolutely no excellent response in the low-risk group,” Dr. Dubsky summarized. “We believe that this molecular score may contribute to patient selection for biomarker-driven studies, especially in the neoadjuvant setting.”
Session attendee Steven Vogl, MD, a medical oncologist with the Montefiore Medical Center in New York, commented, “I have trouble correlating an RCB of 0 or I with what you as a surgeon do for the patient, because you are talking about pathologic complete response or just a few cells there. That’s not what determines how much breast you take off: It’s determined by the total size of the tumor and the size of the breast. So if it’s less than a few centimeters, I’m sure you can do a lumpectomy in every patient. Tell me why I should care that you are getting an RCB of 0 or I in these endocrine patients.”
“Because it’s more likely that these patients will have a smaller tumor and better tumor shrinkage,” Dr. Dubsky replied. “You are of course right, RCB 0 or I was not designed to help surgeons. But it helps me as a translational scientist to have a surrogate and an exact classification for good tumor shrinkage. That’s how I used it.”
C. Kent Osborne, MD, codirector of SABCS and director of the Dan L. Duncan Cancer Center at Baylor College of Medicine in Houston, asked, “We see it in the clinic, and I’m sure you have as well, patients whose tumor doesn’t shrink very much, but the Ki-67 really drops. And that may or may not be a better factor than the actual tumor shrinkage. So how many patients who had tumors that didn’t shrink, which was your endpoint, had a reduction in Ki-67 that was, say, 5%?”
“We haven’t looked at that specifically, but we will do so as we carry on with the follow-up of these patients. Then we can learn more about the prognosis,” Dr. Dubsky replied.
Study details
ABCSG 34 was a randomized phase 2 trial testing addition of the cancer vaccine tecemotide (Stimuvax) to neoadjuvant standard of care among patients with HER2-negative early breast cancer.
Dr. Dubsky and coinvestigators restricted analyses to patients with hormone receptor–positive disease who, depending on clinical and pathologic factors, received neoadjuvant chemotherapy (eight cycles of epirubicin-cyclophosphamide and docetaxel) or neoadjuvant endocrine therapy (6 months of letrozole [Femara]) as standard of care. They were then randomized to additionally receive tecemotide or not before undergoing surgery.
Overall, 25% of the 134 patients in the neoadjuvant chemotherapy group had a good tumor response, defined as pathologic complete response in both breast and nodes (RCB of 0) or minimal residual disease (RCB of I).
Higher EndoPredict score was associated with greater likelihood of good response to chemotherapy. EndoPredict risk group (high vs. low) had a negative predictive value of 100%, a positive predictive value of 26.4%, a true-positive rate of 100%, and a true-negative rate of 8.9% for predicting response (P = .112).
Area under the receiver operating characteristic curve was 0.736.
In a multivariate model, EndoPredict score as a continuous variable was not an independent predictor of response. “The good response was largely driven by covariates that included cell proliferation, and it was Ki-67 that was significant,” Dr. Dubsky noted.
Overall, 18% of the 83 patients in the neoadjuvant endocrine therapy group had a good tumor response (RCB of 0 or I). Here, lower EndoPredict score was associated with greater likelihood of good response. EndoPredict risk group (high vs. low) had a negative predictive value of 92.3%, a positive predictive value of 27.3%, a true-positive rate of 80.0%, and a true-negative rate of 52.9% for predicting response (P = .024). Area under the curve was 0.726.
In a multivariate model here, EndoPredict score as a continuous variable, its estrogen receptor–signaling/differentiation component, and Ki-67 did not independently predict response. “It was maybe a bit surprising that T stage was the strongest factor, possibly indicating that we should have simply treated those women longer than 6 months,” Dr. Dubsky commented. The EndoPredict proliferation component was also a significant predictor.
“Possibly, the very narrow distribution of Ki-67 [among patients given neoendocrine therapy] may have prevented this factor from playing a bigger role in this particular model,” he speculated.
Dr. Dubsky disclosed that he receives consulting fees from Myriad, the maker of EndoPredict, and from Cepheid, Nanostring, and Amgen.
SOURCE: Dubsky P et al. SABCS 2017 Abstract GS6-04.
REPORTING FROM SABCS 2017
Key clinical point:
Major finding: EndoPredict predicted poor tumor shrinkage in patients given neoadjuvant endocrine therapy (high-risk test result NPV, 92%) or neoadjuvant chemotherapy (low-risk test result NPV, 100%).
Data source: A cohort study of 217 patients with HR–positive, HER2-negative breast cancer enrolled in a phase 2 trial of neoadjuvant therapy (ABCSG 34).
Disclosures: Dr. Dubsky disclosed that he receives consulting fees from Cepheid, Myriad, Nanostring, and Amgen.
Wider margins may reduce recurrence risk in early breast cancer
SAN ANTONIO – A margin width beyond ‘no tumor on ink’ may reduce local recurrence in certain subsets of patients undergoing breast-conserving treatment for early-stage breast cancer, according to findings from a meta-analysis.
“Further prospective studies are required to validate the appropriate margin width,” study author Frank A. Vicini, MD, of 21st Century Oncology/Michigan Healthcare Professionals in Farmington Hills, Michigan, said at the San Antonio Breast Cancer Symposium.
Dr. Vicini pointed out that the medical community has been addressing the question of “what is the appropriate margin for breast conserving therapy” for about 3 or 4 decades. A meta-analysis of 33 studies (Ann Surg Oncol. 2014 Mar;21[3]:717-30. doi: 10.1245/s10434-014-3480-5) showed that wider margins are unlikely to have a substantial benefit over “no tumor on ink.”
The results of the meta-analysis led to guidelines published by the Society of Surgical Oncology–American Society for Radiation Oncology that supported those conclusions.
“So the question now is that with additional patients and additional modeling, is this conclusion still correct?” Dr. Vicini said.
The 2014 meta-analysis had significant limitations, and to address them, Dr. Vicini and his colleagues conducted an updated analysis of all available data that included 31 of the initial studies. However, stricter criteria were used for inclusion into their review. The authors reviewed all studies published between 1995 and 2016 that had a minimum follow-up of 50 months, and that included explicit pathologic definitions of margin status and local recurrence in relation to margin status.
The final meta-analysis included 55,302 patients (74% T1 tumors; 72% node-negative disease) from a total of 38 studies, and the median follow up was 7.2 years.
The analysis used three generalized linear mixed models for the outcome of local recurrence, and random effects for study and fixed effects for various patient and study characteristics.
In model 1, all patients with at or equal margin width were compared with those who had wider margins. Model 2 examined the impact of margin width “range” as opposed to a set margin width of 0 mm to 2 mm, 2 mm to 5 mm, or greater than 5 mm. Model 3 divided margin distance by cut points of –1 mm, 2 mm, and 5 mm.
The crude rate of local recurrence was 10.3% for patients with positive margins versus 3.8% for those with negative margins that were defined as no tumor on ink or wider (P less than .001). Local recurrence rates declined as the margin distance increased: 7.2% for patients with margins 0-2 mm, 3.6% for margins of 2-5 mm, and 3.2% for margins wider than 5 mm (P less than .001 for each). The use of endocrine therapy and increasing median study year were associated with decreased rates of local recurrence in univariate models but not in multivariable analyses.
For local recurrence in model 1, a benefit for wider margins was observed, with the greatest benefit seen at 1 mm. The odds ratio (OR) for local recurrence for negative vs. close/positive margins was 0.46 (95% confidence interval, 0.4-0.53) for margins larger than 0 mm, 0.43 (95% CI, 0.36-0.51) for those greater than 1 mm, 0.49 (95% CI, 0.42-0.55) for those larger than 2 mm, and 0.53 (95% CI, 0.43-0.66) for greater than 5 mm. Upon multivariate analysis, the only significant predictive variable was margin status.
For model 2, margin width turned out to be the significant variable, and only wider margins were associated with a decrease in local recurrence.
Finally, in model 3, both margin status and margin width were significantly associated with local recurrence. When modeling as negative, close, or positive margins, the lowest rates were at 2 mm (negative, 3.6%; close, 5.5%; positive, 9.5%) and 5 mm (negative, 2.9%; close, 4.1%; positive, 12.8%).
“So the initial question is the same – should we achieve a 1- to 2-mm margin for our patients as compared to a no tumor on ink, taking into account the potential for local control benefits versus morbidity and time, and cost?” said Dr. Vicini.
“The real question in my mind, is which patients with no tumor on ink require additional surgery,” he concluded. “In my opinion, it may not only be related to the margin status itself, but to the volume of disease near the margin but these meta-analyses were not designed to look at that point.”
SOURCE: Vicini et al. SABCS Abstract GS5-01
SAN ANTONIO – A margin width beyond ‘no tumor on ink’ may reduce local recurrence in certain subsets of patients undergoing breast-conserving treatment for early-stage breast cancer, according to findings from a meta-analysis.
“Further prospective studies are required to validate the appropriate margin width,” study author Frank A. Vicini, MD, of 21st Century Oncology/Michigan Healthcare Professionals in Farmington Hills, Michigan, said at the San Antonio Breast Cancer Symposium.
Dr. Vicini pointed out that the medical community has been addressing the question of “what is the appropriate margin for breast conserving therapy” for about 3 or 4 decades. A meta-analysis of 33 studies (Ann Surg Oncol. 2014 Mar;21[3]:717-30. doi: 10.1245/s10434-014-3480-5) showed that wider margins are unlikely to have a substantial benefit over “no tumor on ink.”
The results of the meta-analysis led to guidelines published by the Society of Surgical Oncology–American Society for Radiation Oncology that supported those conclusions.
“So the question now is that with additional patients and additional modeling, is this conclusion still correct?” Dr. Vicini said.
The 2014 meta-analysis had significant limitations, and to address them, Dr. Vicini and his colleagues conducted an updated analysis of all available data that included 31 of the initial studies. However, stricter criteria were used for inclusion into their review. The authors reviewed all studies published between 1995 and 2016 that had a minimum follow-up of 50 months, and that included explicit pathologic definitions of margin status and local recurrence in relation to margin status.
The final meta-analysis included 55,302 patients (74% T1 tumors; 72% node-negative disease) from a total of 38 studies, and the median follow up was 7.2 years.
The analysis used three generalized linear mixed models for the outcome of local recurrence, and random effects for study and fixed effects for various patient and study characteristics.
In model 1, all patients with at or equal margin width were compared with those who had wider margins. Model 2 examined the impact of margin width “range” as opposed to a set margin width of 0 mm to 2 mm, 2 mm to 5 mm, or greater than 5 mm. Model 3 divided margin distance by cut points of –1 mm, 2 mm, and 5 mm.
The crude rate of local recurrence was 10.3% for patients with positive margins versus 3.8% for those with negative margins that were defined as no tumor on ink or wider (P less than .001). Local recurrence rates declined as the margin distance increased: 7.2% for patients with margins 0-2 mm, 3.6% for margins of 2-5 mm, and 3.2% for margins wider than 5 mm (P less than .001 for each). The use of endocrine therapy and increasing median study year were associated with decreased rates of local recurrence in univariate models but not in multivariable analyses.
For local recurrence in model 1, a benefit for wider margins was observed, with the greatest benefit seen at 1 mm. The odds ratio (OR) for local recurrence for negative vs. close/positive margins was 0.46 (95% confidence interval, 0.4-0.53) for margins larger than 0 mm, 0.43 (95% CI, 0.36-0.51) for those greater than 1 mm, 0.49 (95% CI, 0.42-0.55) for those larger than 2 mm, and 0.53 (95% CI, 0.43-0.66) for greater than 5 mm. Upon multivariate analysis, the only significant predictive variable was margin status.
For model 2, margin width turned out to be the significant variable, and only wider margins were associated with a decrease in local recurrence.
Finally, in model 3, both margin status and margin width were significantly associated with local recurrence. When modeling as negative, close, or positive margins, the lowest rates were at 2 mm (negative, 3.6%; close, 5.5%; positive, 9.5%) and 5 mm (negative, 2.9%; close, 4.1%; positive, 12.8%).
“So the initial question is the same – should we achieve a 1- to 2-mm margin for our patients as compared to a no tumor on ink, taking into account the potential for local control benefits versus morbidity and time, and cost?” said Dr. Vicini.
“The real question in my mind, is which patients with no tumor on ink require additional surgery,” he concluded. “In my opinion, it may not only be related to the margin status itself, but to the volume of disease near the margin but these meta-analyses were not designed to look at that point.”
SOURCE: Vicini et al. SABCS Abstract GS5-01
SAN ANTONIO – A margin width beyond ‘no tumor on ink’ may reduce local recurrence in certain subsets of patients undergoing breast-conserving treatment for early-stage breast cancer, according to findings from a meta-analysis.
“Further prospective studies are required to validate the appropriate margin width,” study author Frank A. Vicini, MD, of 21st Century Oncology/Michigan Healthcare Professionals in Farmington Hills, Michigan, said at the San Antonio Breast Cancer Symposium.
Dr. Vicini pointed out that the medical community has been addressing the question of “what is the appropriate margin for breast conserving therapy” for about 3 or 4 decades. A meta-analysis of 33 studies (Ann Surg Oncol. 2014 Mar;21[3]:717-30. doi: 10.1245/s10434-014-3480-5) showed that wider margins are unlikely to have a substantial benefit over “no tumor on ink.”
The results of the meta-analysis led to guidelines published by the Society of Surgical Oncology–American Society for Radiation Oncology that supported those conclusions.
“So the question now is that with additional patients and additional modeling, is this conclusion still correct?” Dr. Vicini said.
The 2014 meta-analysis had significant limitations, and to address them, Dr. Vicini and his colleagues conducted an updated analysis of all available data that included 31 of the initial studies. However, stricter criteria were used for inclusion into their review. The authors reviewed all studies published between 1995 and 2016 that had a minimum follow-up of 50 months, and that included explicit pathologic definitions of margin status and local recurrence in relation to margin status.
The final meta-analysis included 55,302 patients (74% T1 tumors; 72% node-negative disease) from a total of 38 studies, and the median follow up was 7.2 years.
The analysis used three generalized linear mixed models for the outcome of local recurrence, and random effects for study and fixed effects for various patient and study characteristics.
In model 1, all patients with at or equal margin width were compared with those who had wider margins. Model 2 examined the impact of margin width “range” as opposed to a set margin width of 0 mm to 2 mm, 2 mm to 5 mm, or greater than 5 mm. Model 3 divided margin distance by cut points of –1 mm, 2 mm, and 5 mm.
The crude rate of local recurrence was 10.3% for patients with positive margins versus 3.8% for those with negative margins that were defined as no tumor on ink or wider (P less than .001). Local recurrence rates declined as the margin distance increased: 7.2% for patients with margins 0-2 mm, 3.6% for margins of 2-5 mm, and 3.2% for margins wider than 5 mm (P less than .001 for each). The use of endocrine therapy and increasing median study year were associated with decreased rates of local recurrence in univariate models but not in multivariable analyses.
For local recurrence in model 1, a benefit for wider margins was observed, with the greatest benefit seen at 1 mm. The odds ratio (OR) for local recurrence for negative vs. close/positive margins was 0.46 (95% confidence interval, 0.4-0.53) for margins larger than 0 mm, 0.43 (95% CI, 0.36-0.51) for those greater than 1 mm, 0.49 (95% CI, 0.42-0.55) for those larger than 2 mm, and 0.53 (95% CI, 0.43-0.66) for greater than 5 mm. Upon multivariate analysis, the only significant predictive variable was margin status.
For model 2, margin width turned out to be the significant variable, and only wider margins were associated with a decrease in local recurrence.
Finally, in model 3, both margin status and margin width were significantly associated with local recurrence. When modeling as negative, close, or positive margins, the lowest rates were at 2 mm (negative, 3.6%; close, 5.5%; positive, 9.5%) and 5 mm (negative, 2.9%; close, 4.1%; positive, 12.8%).
“So the initial question is the same – should we achieve a 1- to 2-mm margin for our patients as compared to a no tumor on ink, taking into account the potential for local control benefits versus morbidity and time, and cost?” said Dr. Vicini.
“The real question in my mind, is which patients with no tumor on ink require additional surgery,” he concluded. “In my opinion, it may not only be related to the margin status itself, but to the volume of disease near the margin but these meta-analyses were not designed to look at that point.”
SOURCE: Vicini et al. SABCS Abstract GS5-01
REPORTING FROM SABCS 2017
Key clinical point: Margin widths of at least 2 mm are associated with a reduced risk of ipsilateral breast tumor recurrence versus narrower but uninvolved margins.
Major finding: Crude rates of local recurrence decreased as the margin distance increased: 7.2% for patients with margins 0-2 mm, 3.6% for margins of 2-5 mm, and 3.2% for margins wider than 5 mm (P less than .001 for each).
Data source: Meta-analysis of 38 studies with a total cohort of 55,302 patients treated during 1968-2010.
Disclosures: Study funding was not disclosed. The authors have no relevant disclosures.
Source: Vicini et al. SABCS Abstract GS5-01
Enzalutamide plus exemestane improves PFS in HR+ breast cancer subset
SAN ANTONIO – Enzalutamide added to exemestane improved progression-free survival (PFS) in patients with hormone receptor (HR)-positive advanced breast cancer, investigators reported.
Specifically, it improved outcomes in patients who had not received any prior endocrine therapy and who were positive for a gene signature-based biomarker indicating androgen receptor (AR) signaling.
Patients in this subset who were treated with combination enzalutamide and exemestane achieved a median PFS of 16.5 months, which was significantly higher than the 4 months observed with placebo and exemestane.
However, the addition of enzalutamide had no effect on PFS in the overall cohort or among patients who were biomarker positive but who had received prior endocrine therapy.
“The study met its primary endpoint in improving PFS in the enzalutamide plus exemestane-treated patients who were biomarker positive and HR positive with no prior endocrine therapy for advanced disease as compared [with] exemestane alone,” Denise A. Yardley, MD, of Tennessee Oncology, Nashville, said at the San Antonio Breast Cancer Symposium.
“The role of the AR in HR-positive breast cancer and the predictive value of the identified biomarker are still unclear and will require further studies and validation,” said Dr. Yardley.
Targeting AR is an active area of breast cancer research, as a majority of HR-positive tumors express the AR, as do a moderate number of HER2-positive tumors and almost a third of triple-negative breast cancers. AR signaling has also been associated with resistance to endocrine therapy. Aromatase inhibitors divert estrogen precursors to androgens and data from preclinical models have shown that enzalutamide blocked both estrogen- and androgen-mediated growth of HR+ cells.
Enzalutamide is an inhibitor of AR signaling that is currently used to treat patients with castration-resistant prostate cancer, and has demonstrated clinical activity and was well tolerated in patients with AR-positive advanced triple negative breast cancer, explained Dr. Yardley.
In this study, Dr. Yardley and her colleagues conducted a placebo-controlled phase 2 randomized trial that included 247 patients with HR+/HER2-normal advanced/metastatic breast cancer who were assigned to either 25 mg exemestane plus placebo or 50 mg exemestane and 160 mg enzalutamide daily.
The patients were divided into two parallel cohorts: those with no prior endocrine therapy (C1; n = 127) or those who had received one prior endocrine therapy for metastatic disease (C2; n = 120).
The primary endpoint was PFS in the intent-to-treat population and in the biomarker subgroup of each cohort. Secondary endpoints included the clinical benefit rate at 24 weeks, best overall response, and safety.
The authors found that the PFS in the intent-to-treat population did not significantly differ between those randomized to enzalutamide or placebo in either cohort. In cohort 1, the median PFS was 11.8 months in the enzalutamide arm and 5.8 months in the placebo arm (hazard ratio, 0.82; P = .3631), and in cohort 2, 3.6 months and 3.9 months, respectively (HR, 1.02; P = .9212).
However, statistically significant improvements in median PFS and clinical benefit rate at 24 weeks were observed only in the group with a positive biomarker who had not received any prior endocrine therapy. In cohort 1, the median PFS was 16.5 months in the enzalutamide arm vs. 4.3 months in the placebo arm (HR, 0.44, P = .0335). In cohort 2, median PFS did not significantly differ between groups (6.0 vs. 5.3 months; HR, 0.55; P = .1936).
The clinical response rate in cohort 1 of the biomarker positive group was 83% in the enzalutamide arm versus 38% in the placebo arm (P = .0012).
Adverse events with enzalutamide was similar to those previously reported, and the most common were nausea (39%) in cohort 1 and fatigue (37%) in cohort 2. Dose interruptions due to adverse events occurred in 21.0% and 25.0% of patients randomized to enzalutamide in cohorts 1 and 2 vs. 20.6% and 15.0% in the placebo group.
Dr. Yardley explained that the biomarker used in the study was identified on PAM50. “It was exploratory and proprietary,” she noted, adding that she is unable to share any further information about it at this time.
SOURCE: Yardley et al. SABCS Abstract GS4-07
SAN ANTONIO – Enzalutamide added to exemestane improved progression-free survival (PFS) in patients with hormone receptor (HR)-positive advanced breast cancer, investigators reported.
Specifically, it improved outcomes in patients who had not received any prior endocrine therapy and who were positive for a gene signature-based biomarker indicating androgen receptor (AR) signaling.
Patients in this subset who were treated with combination enzalutamide and exemestane achieved a median PFS of 16.5 months, which was significantly higher than the 4 months observed with placebo and exemestane.
However, the addition of enzalutamide had no effect on PFS in the overall cohort or among patients who were biomarker positive but who had received prior endocrine therapy.
“The study met its primary endpoint in improving PFS in the enzalutamide plus exemestane-treated patients who were biomarker positive and HR positive with no prior endocrine therapy for advanced disease as compared [with] exemestane alone,” Denise A. Yardley, MD, of Tennessee Oncology, Nashville, said at the San Antonio Breast Cancer Symposium.
“The role of the AR in HR-positive breast cancer and the predictive value of the identified biomarker are still unclear and will require further studies and validation,” said Dr. Yardley.
Targeting AR is an active area of breast cancer research, as a majority of HR-positive tumors express the AR, as do a moderate number of HER2-positive tumors and almost a third of triple-negative breast cancers. AR signaling has also been associated with resistance to endocrine therapy. Aromatase inhibitors divert estrogen precursors to androgens and data from preclinical models have shown that enzalutamide blocked both estrogen- and androgen-mediated growth of HR+ cells.
Enzalutamide is an inhibitor of AR signaling that is currently used to treat patients with castration-resistant prostate cancer, and has demonstrated clinical activity and was well tolerated in patients with AR-positive advanced triple negative breast cancer, explained Dr. Yardley.
In this study, Dr. Yardley and her colleagues conducted a placebo-controlled phase 2 randomized trial that included 247 patients with HR+/HER2-normal advanced/metastatic breast cancer who were assigned to either 25 mg exemestane plus placebo or 50 mg exemestane and 160 mg enzalutamide daily.
The patients were divided into two parallel cohorts: those with no prior endocrine therapy (C1; n = 127) or those who had received one prior endocrine therapy for metastatic disease (C2; n = 120).
The primary endpoint was PFS in the intent-to-treat population and in the biomarker subgroup of each cohort. Secondary endpoints included the clinical benefit rate at 24 weeks, best overall response, and safety.
The authors found that the PFS in the intent-to-treat population did not significantly differ between those randomized to enzalutamide or placebo in either cohort. In cohort 1, the median PFS was 11.8 months in the enzalutamide arm and 5.8 months in the placebo arm (hazard ratio, 0.82; P = .3631), and in cohort 2, 3.6 months and 3.9 months, respectively (HR, 1.02; P = .9212).
However, statistically significant improvements in median PFS and clinical benefit rate at 24 weeks were observed only in the group with a positive biomarker who had not received any prior endocrine therapy. In cohort 1, the median PFS was 16.5 months in the enzalutamide arm vs. 4.3 months in the placebo arm (HR, 0.44, P = .0335). In cohort 2, median PFS did not significantly differ between groups (6.0 vs. 5.3 months; HR, 0.55; P = .1936).
The clinical response rate in cohort 1 of the biomarker positive group was 83% in the enzalutamide arm versus 38% in the placebo arm (P = .0012).
Adverse events with enzalutamide was similar to those previously reported, and the most common were nausea (39%) in cohort 1 and fatigue (37%) in cohort 2. Dose interruptions due to adverse events occurred in 21.0% and 25.0% of patients randomized to enzalutamide in cohorts 1 and 2 vs. 20.6% and 15.0% in the placebo group.
Dr. Yardley explained that the biomarker used in the study was identified on PAM50. “It was exploratory and proprietary,” she noted, adding that she is unable to share any further information about it at this time.
SOURCE: Yardley et al. SABCS Abstract GS4-07
SAN ANTONIO – Enzalutamide added to exemestane improved progression-free survival (PFS) in patients with hormone receptor (HR)-positive advanced breast cancer, investigators reported.
Specifically, it improved outcomes in patients who had not received any prior endocrine therapy and who were positive for a gene signature-based biomarker indicating androgen receptor (AR) signaling.
Patients in this subset who were treated with combination enzalutamide and exemestane achieved a median PFS of 16.5 months, which was significantly higher than the 4 months observed with placebo and exemestane.
However, the addition of enzalutamide had no effect on PFS in the overall cohort or among patients who were biomarker positive but who had received prior endocrine therapy.
“The study met its primary endpoint in improving PFS in the enzalutamide plus exemestane-treated patients who were biomarker positive and HR positive with no prior endocrine therapy for advanced disease as compared [with] exemestane alone,” Denise A. Yardley, MD, of Tennessee Oncology, Nashville, said at the San Antonio Breast Cancer Symposium.
“The role of the AR in HR-positive breast cancer and the predictive value of the identified biomarker are still unclear and will require further studies and validation,” said Dr. Yardley.
Targeting AR is an active area of breast cancer research, as a majority of HR-positive tumors express the AR, as do a moderate number of HER2-positive tumors and almost a third of triple-negative breast cancers. AR signaling has also been associated with resistance to endocrine therapy. Aromatase inhibitors divert estrogen precursors to androgens and data from preclinical models have shown that enzalutamide blocked both estrogen- and androgen-mediated growth of HR+ cells.
Enzalutamide is an inhibitor of AR signaling that is currently used to treat patients with castration-resistant prostate cancer, and has demonstrated clinical activity and was well tolerated in patients with AR-positive advanced triple negative breast cancer, explained Dr. Yardley.
In this study, Dr. Yardley and her colleagues conducted a placebo-controlled phase 2 randomized trial that included 247 patients with HR+/HER2-normal advanced/metastatic breast cancer who were assigned to either 25 mg exemestane plus placebo or 50 mg exemestane and 160 mg enzalutamide daily.
The patients were divided into two parallel cohorts: those with no prior endocrine therapy (C1; n = 127) or those who had received one prior endocrine therapy for metastatic disease (C2; n = 120).
The primary endpoint was PFS in the intent-to-treat population and in the biomarker subgroup of each cohort. Secondary endpoints included the clinical benefit rate at 24 weeks, best overall response, and safety.
The authors found that the PFS in the intent-to-treat population did not significantly differ between those randomized to enzalutamide or placebo in either cohort. In cohort 1, the median PFS was 11.8 months in the enzalutamide arm and 5.8 months in the placebo arm (hazard ratio, 0.82; P = .3631), and in cohort 2, 3.6 months and 3.9 months, respectively (HR, 1.02; P = .9212).
However, statistically significant improvements in median PFS and clinical benefit rate at 24 weeks were observed only in the group with a positive biomarker who had not received any prior endocrine therapy. In cohort 1, the median PFS was 16.5 months in the enzalutamide arm vs. 4.3 months in the placebo arm (HR, 0.44, P = .0335). In cohort 2, median PFS did not significantly differ between groups (6.0 vs. 5.3 months; HR, 0.55; P = .1936).
The clinical response rate in cohort 1 of the biomarker positive group was 83% in the enzalutamide arm versus 38% in the placebo arm (P = .0012).
Adverse events with enzalutamide was similar to those previously reported, and the most common were nausea (39%) in cohort 1 and fatigue (37%) in cohort 2. Dose interruptions due to adverse events occurred in 21.0% and 25.0% of patients randomized to enzalutamide in cohorts 1 and 2 vs. 20.6% and 15.0% in the placebo group.
Dr. Yardley explained that the biomarker used in the study was identified on PAM50. “It was exploratory and proprietary,” she noted, adding that she is unable to share any further information about it at this time.
SOURCE: Yardley et al. SABCS Abstract GS4-07
REPORTING FROM SABCS 2017
Key clinical point: Enzalutamide added to exemestane improves progression-free survival in hormone receptor–positive advanced breast cancer patients with a biomarker indicating androgen receptor signaling.
Major finding: In this subset of patients, combination therapy improved PFS: 16.5 months vs. 4.3 months for the placebo arm (HR 0.44, P = .0335).
Data source: A placebo-controlled phase 2 randomized trial comprising 247 patients with HR+/HER2-normal advanced/metastatic breast cancer.
Disclosures: Study funding was not disclosed.
Source: Yardley et al. SABCS Abstract GS4-07.
Pertuzumab approved for HER2-positive breast cancer
, according to the Food and Drug Administration.
The approval was based on results from the APHINITY trial, which included 4,804 patients who had HER2-positive early breast cancers that were excised prior to the study. After a median follow-up period of 45.4 months, an invasive disease event occurred in 7.1% of all patients who received pertuzumab (Perjeta) and in 8.7% of patients who received placebo. In patients with hormone receptor–negative disease, invasive events occurred in 8.2% of the pertuzumab group and in 10.6% of the placebo group. In patients with node-positive disease, the invasive event rate was 9.2% in the pertuzumab group and 12.1% in the placebo group.
“The initial pertuzumab dose is 840 mg administered as a 60-minute intravenous infusion, followed every 3 weeks thereafter by 420 mg administered as a 30- to 60-minute intravenous infusion,” the FDA said in the statement.
Find the full statement on the FDA website.
, according to the Food and Drug Administration.
The approval was based on results from the APHINITY trial, which included 4,804 patients who had HER2-positive early breast cancers that were excised prior to the study. After a median follow-up period of 45.4 months, an invasive disease event occurred in 7.1% of all patients who received pertuzumab (Perjeta) and in 8.7% of patients who received placebo. In patients with hormone receptor–negative disease, invasive events occurred in 8.2% of the pertuzumab group and in 10.6% of the placebo group. In patients with node-positive disease, the invasive event rate was 9.2% in the pertuzumab group and 12.1% in the placebo group.
“The initial pertuzumab dose is 840 mg administered as a 60-minute intravenous infusion, followed every 3 weeks thereafter by 420 mg administered as a 30- to 60-minute intravenous infusion,” the FDA said in the statement.
Find the full statement on the FDA website.
, according to the Food and Drug Administration.
The approval was based on results from the APHINITY trial, which included 4,804 patients who had HER2-positive early breast cancers that were excised prior to the study. After a median follow-up period of 45.4 months, an invasive disease event occurred in 7.1% of all patients who received pertuzumab (Perjeta) and in 8.7% of patients who received placebo. In patients with hormone receptor–negative disease, invasive events occurred in 8.2% of the pertuzumab group and in 10.6% of the placebo group. In patients with node-positive disease, the invasive event rate was 9.2% in the pertuzumab group and 12.1% in the placebo group.
“The initial pertuzumab dose is 840 mg administered as a 60-minute intravenous infusion, followed every 3 weeks thereafter by 420 mg administered as a 30- to 60-minute intravenous infusion,” the FDA said in the statement.
Find the full statement on the FDA website.