User login
Novel PARP inhibitor boosts PFS in HER2- breast cancer with BRCA mutations
SAN ANTONIO – In women with advanced HER2-negative breast cancer with germline BRCA mutations, an investigational oral PARP inhibitor talazoparib was associated with a near doubling in progression-free survival (PFS) when compared with single-agent chemotherapy, results of the phase 3 EMBRACA trial show.
After a median follow-up of 11.2 months, the median PFS by blinded central review – the primary endpoint – was 8.6 months for patients assigned to receive talazoparib, compared with 5.6 months for patients randomized to receive the physician’s choice of either capecitabine, eribulin, gemcitabine, or vinorelbine, reported Jennifer K. Litton, MD, from the University of Texas MD Anderson Cancer Center in Houston.
“Patients who were assigned to talazoparib had an improvement in their global health status versus patients who had deterioration when randomized.”
Talazoparib is an oral inhibitor of poly ADP-ribose polymerase (PARP) with a dual mechanism of action: It both inhibits the PARP enzyme directly and traps PARP on single-stranded DNA breaks, preventing repair of DNA damage and leading to the death of malignant cells.
In the phase 2 ABRAZO trial, the PARP inhibitor showed “encouraging” efficacy and safety in patients with germline BRCA1/BRCA2 mutations who had received platinum-based chemotherapy or at least three prior cytotoxic regimens.
Dr. Litton reported results of the EMBRACA trial, a phase 3 study in patients with locally advanced or metastatic HER2 negative breast cancer a germline BRCA1 or BRCA2 mutation. Patients were stratified by number of prior chemotherapy regimens, by having triple-negative breast cancer or hormone receptor-positive breast cancer, and by having a history of either central nervous system metastases or no CNS metastases; they were then randomized on a 2:1 basis to either oral talazoparib 1 mg daily (287 patients) or to the physician’s choice of therapy with one of the agents noted before.
The patient characteristics were generally well balanced, although there was a higher percentage of patients aged younger than 50 years in the talazoparib group than in the group treated with other agents (63.4% vs. 46.5%, respectively), slightly more CNS metastases (15% vs. 13.9%), and a higher percentage of patients with a disease-free interval (time from initial diagnosis to advanced breast cancer) shorter than 12 months (37.6% vs. 29.2%).
The primary endpoint of PFS by blinded central review showed the aforementioned significant benefit of talazoparib. A PFS by subgroup analysis showed that talazoparib was significantly better in all parameters except for patients who had previously received platinum-based therapy.
The trial was also powered to show overall survival as a secondary endpoint, but the data are not mature, Dr. Litton said. An interim OS analysis showed an apparent trend favoring the PARP inhibitor, with a median of 22.3 months, compared with 19.5 months with physician’s choice of treatment.
The 24- and 36-month probabilities of survival were 45% and 34% respectively for patients treated with talazoparib, compared with 37% and 0% for patients treated with other agents.
The objective response rate by investigator rating was 62.6% with talazoparib, compared with 27.2% for other drugs (odds ratio, 4.99; P less than .0001).
Anemia was the most common hematologic adverse event, with grade 3 or greater occurring in 39.2% of patients on the PARP inhibitor, compared with 4.8% of patients treated with other agents.
Talazoparib, unlike other PARP inhibitors, was also associated with grade 1 or 2 alopecia, which occurred in 25.2% of those patients, compared with 27.8% of those receiving the physician’s choice of treatment.
Grade 3 or 4 serious adverse events occurred in about 25.5% of patients in each study arm. Events leading to permanent drug discontinuation were more common with physician’s choice agents at 9.5%, compared with 7.7% of patients treated with talazoparib.
Kent Osborne, MD, the director of the Dan L. Duncan Cancer Center at Baylor College of Medicine, Houston, who moderated a briefing where Dr. Litton presented the data, commented that patients may not be as enthusiastic about the results as investigators seem to be.
“I’ve heard doctors like you and I say ‘This is really great, we’ve got some activity from a PARP inhibitor;’ patients look at it and say ‘Gee, a few more responses and a 3-month prolongation on average of my time to progression is not a very big advantage,’ ” he said to Dr. Litton.
“So what’s the next step in the development of these drugs? Are they going to be used in combinations? Are we going to come up with a mechanism of resistance that we can then overcome to extend the duration of their benefit?” he asked.
Dr. Litton replied that she was encouraged by fact that the tails of the survival curves appear to be separating and that some patients have complete responses and some have relatively durable responses.
“One of the things that we’re going to be looking at are the correlatives, trying to identify who these extraordinary responders are and the mechanisms of resistance as best we can,” she said.
This study was funded by Pfizer, which developed the inhibitor. Dr. Litton has disclosed research funding with EMD Serono, AstraZeneca, Pfizer, Genentech, and GlaxoSmithKline, and serves on advisory boards for Pfizer and AstraZeneca, all uncompensated.
SOURCE: Litton et al. SABCS 2017 Abstract GS6-07.
SAN ANTONIO – In women with advanced HER2-negative breast cancer with germline BRCA mutations, an investigational oral PARP inhibitor talazoparib was associated with a near doubling in progression-free survival (PFS) when compared with single-agent chemotherapy, results of the phase 3 EMBRACA trial show.
After a median follow-up of 11.2 months, the median PFS by blinded central review – the primary endpoint – was 8.6 months for patients assigned to receive talazoparib, compared with 5.6 months for patients randomized to receive the physician’s choice of either capecitabine, eribulin, gemcitabine, or vinorelbine, reported Jennifer K. Litton, MD, from the University of Texas MD Anderson Cancer Center in Houston.
“Patients who were assigned to talazoparib had an improvement in their global health status versus patients who had deterioration when randomized.”
Talazoparib is an oral inhibitor of poly ADP-ribose polymerase (PARP) with a dual mechanism of action: It both inhibits the PARP enzyme directly and traps PARP on single-stranded DNA breaks, preventing repair of DNA damage and leading to the death of malignant cells.
In the phase 2 ABRAZO trial, the PARP inhibitor showed “encouraging” efficacy and safety in patients with germline BRCA1/BRCA2 mutations who had received platinum-based chemotherapy or at least three prior cytotoxic regimens.
Dr. Litton reported results of the EMBRACA trial, a phase 3 study in patients with locally advanced or metastatic HER2 negative breast cancer a germline BRCA1 or BRCA2 mutation. Patients were stratified by number of prior chemotherapy regimens, by having triple-negative breast cancer or hormone receptor-positive breast cancer, and by having a history of either central nervous system metastases or no CNS metastases; they were then randomized on a 2:1 basis to either oral talazoparib 1 mg daily (287 patients) or to the physician’s choice of therapy with one of the agents noted before.
The patient characteristics were generally well balanced, although there was a higher percentage of patients aged younger than 50 years in the talazoparib group than in the group treated with other agents (63.4% vs. 46.5%, respectively), slightly more CNS metastases (15% vs. 13.9%), and a higher percentage of patients with a disease-free interval (time from initial diagnosis to advanced breast cancer) shorter than 12 months (37.6% vs. 29.2%).
The primary endpoint of PFS by blinded central review showed the aforementioned significant benefit of talazoparib. A PFS by subgroup analysis showed that talazoparib was significantly better in all parameters except for patients who had previously received platinum-based therapy.
The trial was also powered to show overall survival as a secondary endpoint, but the data are not mature, Dr. Litton said. An interim OS analysis showed an apparent trend favoring the PARP inhibitor, with a median of 22.3 months, compared with 19.5 months with physician’s choice of treatment.
The 24- and 36-month probabilities of survival were 45% and 34% respectively for patients treated with talazoparib, compared with 37% and 0% for patients treated with other agents.
The objective response rate by investigator rating was 62.6% with talazoparib, compared with 27.2% for other drugs (odds ratio, 4.99; P less than .0001).
Anemia was the most common hematologic adverse event, with grade 3 or greater occurring in 39.2% of patients on the PARP inhibitor, compared with 4.8% of patients treated with other agents.
Talazoparib, unlike other PARP inhibitors, was also associated with grade 1 or 2 alopecia, which occurred in 25.2% of those patients, compared with 27.8% of those receiving the physician’s choice of treatment.
Grade 3 or 4 serious adverse events occurred in about 25.5% of patients in each study arm. Events leading to permanent drug discontinuation were more common with physician’s choice agents at 9.5%, compared with 7.7% of patients treated with talazoparib.
Kent Osborne, MD, the director of the Dan L. Duncan Cancer Center at Baylor College of Medicine, Houston, who moderated a briefing where Dr. Litton presented the data, commented that patients may not be as enthusiastic about the results as investigators seem to be.
“I’ve heard doctors like you and I say ‘This is really great, we’ve got some activity from a PARP inhibitor;’ patients look at it and say ‘Gee, a few more responses and a 3-month prolongation on average of my time to progression is not a very big advantage,’ ” he said to Dr. Litton.
“So what’s the next step in the development of these drugs? Are they going to be used in combinations? Are we going to come up with a mechanism of resistance that we can then overcome to extend the duration of their benefit?” he asked.
Dr. Litton replied that she was encouraged by fact that the tails of the survival curves appear to be separating and that some patients have complete responses and some have relatively durable responses.
“One of the things that we’re going to be looking at are the correlatives, trying to identify who these extraordinary responders are and the mechanisms of resistance as best we can,” she said.
This study was funded by Pfizer, which developed the inhibitor. Dr. Litton has disclosed research funding with EMD Serono, AstraZeneca, Pfizer, Genentech, and GlaxoSmithKline, and serves on advisory boards for Pfizer and AstraZeneca, all uncompensated.
SOURCE: Litton et al. SABCS 2017 Abstract GS6-07.
SAN ANTONIO – In women with advanced HER2-negative breast cancer with germline BRCA mutations, an investigational oral PARP inhibitor talazoparib was associated with a near doubling in progression-free survival (PFS) when compared with single-agent chemotherapy, results of the phase 3 EMBRACA trial show.
After a median follow-up of 11.2 months, the median PFS by blinded central review – the primary endpoint – was 8.6 months for patients assigned to receive talazoparib, compared with 5.6 months for patients randomized to receive the physician’s choice of either capecitabine, eribulin, gemcitabine, or vinorelbine, reported Jennifer K. Litton, MD, from the University of Texas MD Anderson Cancer Center in Houston.
“Patients who were assigned to talazoparib had an improvement in their global health status versus patients who had deterioration when randomized.”
Talazoparib is an oral inhibitor of poly ADP-ribose polymerase (PARP) with a dual mechanism of action: It both inhibits the PARP enzyme directly and traps PARP on single-stranded DNA breaks, preventing repair of DNA damage and leading to the death of malignant cells.
In the phase 2 ABRAZO trial, the PARP inhibitor showed “encouraging” efficacy and safety in patients with germline BRCA1/BRCA2 mutations who had received platinum-based chemotherapy or at least three prior cytotoxic regimens.
Dr. Litton reported results of the EMBRACA trial, a phase 3 study in patients with locally advanced or metastatic HER2 negative breast cancer a germline BRCA1 or BRCA2 mutation. Patients were stratified by number of prior chemotherapy regimens, by having triple-negative breast cancer or hormone receptor-positive breast cancer, and by having a history of either central nervous system metastases or no CNS metastases; they were then randomized on a 2:1 basis to either oral talazoparib 1 mg daily (287 patients) or to the physician’s choice of therapy with one of the agents noted before.
The patient characteristics were generally well balanced, although there was a higher percentage of patients aged younger than 50 years in the talazoparib group than in the group treated with other agents (63.4% vs. 46.5%, respectively), slightly more CNS metastases (15% vs. 13.9%), and a higher percentage of patients with a disease-free interval (time from initial diagnosis to advanced breast cancer) shorter than 12 months (37.6% vs. 29.2%).
The primary endpoint of PFS by blinded central review showed the aforementioned significant benefit of talazoparib. A PFS by subgroup analysis showed that talazoparib was significantly better in all parameters except for patients who had previously received platinum-based therapy.
The trial was also powered to show overall survival as a secondary endpoint, but the data are not mature, Dr. Litton said. An interim OS analysis showed an apparent trend favoring the PARP inhibitor, with a median of 22.3 months, compared with 19.5 months with physician’s choice of treatment.
The 24- and 36-month probabilities of survival were 45% and 34% respectively for patients treated with talazoparib, compared with 37% and 0% for patients treated with other agents.
The objective response rate by investigator rating was 62.6% with talazoparib, compared with 27.2% for other drugs (odds ratio, 4.99; P less than .0001).
Anemia was the most common hematologic adverse event, with grade 3 or greater occurring in 39.2% of patients on the PARP inhibitor, compared with 4.8% of patients treated with other agents.
Talazoparib, unlike other PARP inhibitors, was also associated with grade 1 or 2 alopecia, which occurred in 25.2% of those patients, compared with 27.8% of those receiving the physician’s choice of treatment.
Grade 3 or 4 serious adverse events occurred in about 25.5% of patients in each study arm. Events leading to permanent drug discontinuation were more common with physician’s choice agents at 9.5%, compared with 7.7% of patients treated with talazoparib.
Kent Osborne, MD, the director of the Dan L. Duncan Cancer Center at Baylor College of Medicine, Houston, who moderated a briefing where Dr. Litton presented the data, commented that patients may not be as enthusiastic about the results as investigators seem to be.
“I’ve heard doctors like you and I say ‘This is really great, we’ve got some activity from a PARP inhibitor;’ patients look at it and say ‘Gee, a few more responses and a 3-month prolongation on average of my time to progression is not a very big advantage,’ ” he said to Dr. Litton.
“So what’s the next step in the development of these drugs? Are they going to be used in combinations? Are we going to come up with a mechanism of resistance that we can then overcome to extend the duration of their benefit?” he asked.
Dr. Litton replied that she was encouraged by fact that the tails of the survival curves appear to be separating and that some patients have complete responses and some have relatively durable responses.
“One of the things that we’re going to be looking at are the correlatives, trying to identify who these extraordinary responders are and the mechanisms of resistance as best we can,” she said.
This study was funded by Pfizer, which developed the inhibitor. Dr. Litton has disclosed research funding with EMD Serono, AstraZeneca, Pfizer, Genentech, and GlaxoSmithKline, and serves on advisory boards for Pfizer and AstraZeneca, all uncompensated.
SOURCE: Litton et al. SABCS 2017 Abstract GS6-07.
REPORTING FROM SABCS 2017
Key clinical point: The investigational PARP inhibitor talazoparib extended progression-free survival of advanced HER2-negative breast cancer with germline BRCA mutations.
Major finding: Talazoparib was associated with a 46% reduction in risk for progression when compared with standard single agent therapies.
Data source: Randomized clinical trial in 431 patients with advanced, previously treated breast cancer with germline BRCA1 and BRCA2 mutations.
Disclosures: This study was funded by Pfizer, which developed the inhibitor. Dr. Litton disclosed that she has received research funding from EMD Serono, AstraZeneca, Pfizer, Genentech, and GlaxoSmithKline and that she serves on advisory boards for Pfizer and AstraZeneca, all uncompensated.
Source: Litton J et al. SABCS 2017 Abstract GS6-07.
Extra years of adjuvant bisphosphonate not needed in early breast cancer
SAN ANTONIO – When it comes to adjuvant bisphosphonate therapy following adjuvant chemotherapy for high-risk early breast cancer, more is not better than less, phase 3 data from the randomized SUCCESS A study suggest.
Among 3,421 patients randomized to adjuvant bisphosphonate therapy following chemotherapy, there was barely a speck of difference in either disease-free survival (DFS) or overall survival (OS) between patients randomized to either 2 years or 5 years of adjuvant bisphosphonate therapy with zoledronate, reported Wolfgang Janni, MD, from University Hospital Ulm (Germany).
“We conclude 5 years of adjuvant zoledronate treatment should not be considered currently in these patients in the absence of decreased bone density,” he said at the San Antonio Breast Cancer Symposium.
Adjuvant bisphosphonate therapy in patients with early breast cancer is associated with improved breast cancer–specific survival and reduced rates of breast cancer recurrence in bone, especially for postmenopausal patients, as shown in a meta-analysis from the Early Breast Cancer Trialists’ Collaborative Group, Dr. Janni noted.
German breast cancer guidelines state that postmenopausal women should be offered bisphosphonates as part of their adjuvant systemic therapy, but the optimal duration of therapy is uncertain, prompting the investigators to examine the issue in a randomized trial.
SUCCESS A was a multicenter, phase 3, randomized trial with a multifactorial 2 x 2 design, in patients with high-risk node-negative and node-positive disease. Patients were randomized to FEC100 chemotherapy followed by docetaxel with or without gemcitabine. Chemotherapy was followed by endocrine therapy with 2 years of tamoxifen followed by 3 years of anastrozole (Arimidex). At the start of endocrine therapy, patients were further randomized to receive either 2 or 5 years of adjuvant zoledronate, 4 mg intravenously every 3 months for 2 years, or the same schedule over 2 years, followed by 4 mg every 6 months for 3 years.
A total of 2,987 of the 3,421 patients randomized to a zoledronate schedule were available for inclusion in the analysis.
As noted, adapted DFS and OS, measured starting from 2 years after the start of zoledronate with a maximum observation time of 48 months, were virtually identical between the two treatment groups, with respective P values of .827 and .713. Similarly, in a multivariate regression analysis model adjusted for age, body mass index, menopausal status, tumor size, nodal stage, histological grade and type, hormone receptor status, HER2 status, surgery type, and chemotherapy regimen, the hazard ratio for 5 vs. 2 years was 0.97 for DFS and 0.98 for OS. Neither endpoint was significantly different between the groups.
Similarly, there was no significant differences in the number of bone recurrences as first distant recurrences or in premenopausal vs. postmenopausal women.
Adverse events of any grade were significantly higher with 5 years of bisphosphonate therapy (46.2% vs. 27.2%, P less than .001), including significantly higher grade 3 or greater adverse events (7.6% vs. 5.1%, P = .006).
Following presentation of the data in an oral session, moderator Sibylle Loibl, MD, PhD, of the German Breast Group in Neu-Isenburg, Germany, questioned whether the follow-up was long enough to detect a clinically meaningful difference.
“The negative result of this study might be due to the small observation time,” Dr. Janni conceded.”We have a quite intensive drug regimen for the first 2 years, so this might also be a contributing factor [as to why] we did not see any difference.”
The SUCCESS A study was supported by AstraZeneca, Chugai, Janssen Diagnostics, Lilly, Novartis, and Sanofi-Aventis. Dr. Janni has reported financial relationships with AstraZeneca, Chugai, Janssen, Lilly, Novartis, and Sanofi.
SOURCE: Janni et al. SABCS 2017 Abstract GS1-06
SAN ANTONIO – When it comes to adjuvant bisphosphonate therapy following adjuvant chemotherapy for high-risk early breast cancer, more is not better than less, phase 3 data from the randomized SUCCESS A study suggest.
Among 3,421 patients randomized to adjuvant bisphosphonate therapy following chemotherapy, there was barely a speck of difference in either disease-free survival (DFS) or overall survival (OS) between patients randomized to either 2 years or 5 years of adjuvant bisphosphonate therapy with zoledronate, reported Wolfgang Janni, MD, from University Hospital Ulm (Germany).
“We conclude 5 years of adjuvant zoledronate treatment should not be considered currently in these patients in the absence of decreased bone density,” he said at the San Antonio Breast Cancer Symposium.
Adjuvant bisphosphonate therapy in patients with early breast cancer is associated with improved breast cancer–specific survival and reduced rates of breast cancer recurrence in bone, especially for postmenopausal patients, as shown in a meta-analysis from the Early Breast Cancer Trialists’ Collaborative Group, Dr. Janni noted.
German breast cancer guidelines state that postmenopausal women should be offered bisphosphonates as part of their adjuvant systemic therapy, but the optimal duration of therapy is uncertain, prompting the investigators to examine the issue in a randomized trial.
SUCCESS A was a multicenter, phase 3, randomized trial with a multifactorial 2 x 2 design, in patients with high-risk node-negative and node-positive disease. Patients were randomized to FEC100 chemotherapy followed by docetaxel with or without gemcitabine. Chemotherapy was followed by endocrine therapy with 2 years of tamoxifen followed by 3 years of anastrozole (Arimidex). At the start of endocrine therapy, patients were further randomized to receive either 2 or 5 years of adjuvant zoledronate, 4 mg intravenously every 3 months for 2 years, or the same schedule over 2 years, followed by 4 mg every 6 months for 3 years.
A total of 2,987 of the 3,421 patients randomized to a zoledronate schedule were available for inclusion in the analysis.
As noted, adapted DFS and OS, measured starting from 2 years after the start of zoledronate with a maximum observation time of 48 months, were virtually identical between the two treatment groups, with respective P values of .827 and .713. Similarly, in a multivariate regression analysis model adjusted for age, body mass index, menopausal status, tumor size, nodal stage, histological grade and type, hormone receptor status, HER2 status, surgery type, and chemotherapy regimen, the hazard ratio for 5 vs. 2 years was 0.97 for DFS and 0.98 for OS. Neither endpoint was significantly different between the groups.
Similarly, there was no significant differences in the number of bone recurrences as first distant recurrences or in premenopausal vs. postmenopausal women.
Adverse events of any grade were significantly higher with 5 years of bisphosphonate therapy (46.2% vs. 27.2%, P less than .001), including significantly higher grade 3 or greater adverse events (7.6% vs. 5.1%, P = .006).
Following presentation of the data in an oral session, moderator Sibylle Loibl, MD, PhD, of the German Breast Group in Neu-Isenburg, Germany, questioned whether the follow-up was long enough to detect a clinically meaningful difference.
“The negative result of this study might be due to the small observation time,” Dr. Janni conceded.”We have a quite intensive drug regimen for the first 2 years, so this might also be a contributing factor [as to why] we did not see any difference.”
The SUCCESS A study was supported by AstraZeneca, Chugai, Janssen Diagnostics, Lilly, Novartis, and Sanofi-Aventis. Dr. Janni has reported financial relationships with AstraZeneca, Chugai, Janssen, Lilly, Novartis, and Sanofi.
SOURCE: Janni et al. SABCS 2017 Abstract GS1-06
SAN ANTONIO – When it comes to adjuvant bisphosphonate therapy following adjuvant chemotherapy for high-risk early breast cancer, more is not better than less, phase 3 data from the randomized SUCCESS A study suggest.
Among 3,421 patients randomized to adjuvant bisphosphonate therapy following chemotherapy, there was barely a speck of difference in either disease-free survival (DFS) or overall survival (OS) between patients randomized to either 2 years or 5 years of adjuvant bisphosphonate therapy with zoledronate, reported Wolfgang Janni, MD, from University Hospital Ulm (Germany).
“We conclude 5 years of adjuvant zoledronate treatment should not be considered currently in these patients in the absence of decreased bone density,” he said at the San Antonio Breast Cancer Symposium.
Adjuvant bisphosphonate therapy in patients with early breast cancer is associated with improved breast cancer–specific survival and reduced rates of breast cancer recurrence in bone, especially for postmenopausal patients, as shown in a meta-analysis from the Early Breast Cancer Trialists’ Collaborative Group, Dr. Janni noted.
German breast cancer guidelines state that postmenopausal women should be offered bisphosphonates as part of their adjuvant systemic therapy, but the optimal duration of therapy is uncertain, prompting the investigators to examine the issue in a randomized trial.
SUCCESS A was a multicenter, phase 3, randomized trial with a multifactorial 2 x 2 design, in patients with high-risk node-negative and node-positive disease. Patients were randomized to FEC100 chemotherapy followed by docetaxel with or without gemcitabine. Chemotherapy was followed by endocrine therapy with 2 years of tamoxifen followed by 3 years of anastrozole (Arimidex). At the start of endocrine therapy, patients were further randomized to receive either 2 or 5 years of adjuvant zoledronate, 4 mg intravenously every 3 months for 2 years, or the same schedule over 2 years, followed by 4 mg every 6 months for 3 years.
A total of 2,987 of the 3,421 patients randomized to a zoledronate schedule were available for inclusion in the analysis.
As noted, adapted DFS and OS, measured starting from 2 years after the start of zoledronate with a maximum observation time of 48 months, were virtually identical between the two treatment groups, with respective P values of .827 and .713. Similarly, in a multivariate regression analysis model adjusted for age, body mass index, menopausal status, tumor size, nodal stage, histological grade and type, hormone receptor status, HER2 status, surgery type, and chemotherapy regimen, the hazard ratio for 5 vs. 2 years was 0.97 for DFS and 0.98 for OS. Neither endpoint was significantly different between the groups.
Similarly, there was no significant differences in the number of bone recurrences as first distant recurrences or in premenopausal vs. postmenopausal women.
Adverse events of any grade were significantly higher with 5 years of bisphosphonate therapy (46.2% vs. 27.2%, P less than .001), including significantly higher grade 3 or greater adverse events (7.6% vs. 5.1%, P = .006).
Following presentation of the data in an oral session, moderator Sibylle Loibl, MD, PhD, of the German Breast Group in Neu-Isenburg, Germany, questioned whether the follow-up was long enough to detect a clinically meaningful difference.
“The negative result of this study might be due to the small observation time,” Dr. Janni conceded.”We have a quite intensive drug regimen for the first 2 years, so this might also be a contributing factor [as to why] we did not see any difference.”
The SUCCESS A study was supported by AstraZeneca, Chugai, Janssen Diagnostics, Lilly, Novartis, and Sanofi-Aventis. Dr. Janni has reported financial relationships with AstraZeneca, Chugai, Janssen, Lilly, Novartis, and Sanofi.
SOURCE: Janni et al. SABCS 2017 Abstract GS1-06
REPORTING FROM SABCS 2017
Key clinical point: Five years of adjuvant bisphosphonate therapy offered no survival advantages over 2 years of therapy for women with early breast cancers.
Major finding: Neither adapted disease-free survival nor overall survival were significantly better with 3 extra years of zoledronate therapy.
Data source: Randomized phase 3 trial.
Disclosures: The SUCCESS A study was supported by AstraZeneca, Chugai, Janssen Diagnostics, Lilly, Novartis, and Sanofi-Aventis. Dr. Janni has reported financial relationships with AstraZeneca, Chugai, Janssen, Lilly, Novartis, and Sanofi.
Source: Janni et al., SABCS 2017 abstract GS1-06
VIDEO: CTCs may identify asymptomatic late breast cancer recurrences
SAN ANTONIO – Although the initial ardor over the use of circulating tumor cells (CTCs) in cancer diagnosis has cooled, new research suggests that they may play a role in identifying late breast cancer recurrences in otherwise asymptomatic patients, according to members of the ECOG-ACRIN cancer research group.
In this video interview from the the San Antonio Breast Cancer Symposium, Joseph A. Sparano, MD, of Montefiore Medical Center and Albert Einstein College of Medicine in New York, describes ECOG-ACRIN’s experiments showing that patients with hormone receptor–positive disease and HER2-negative breast cancer have a significantly elevated risk for recurrence, supporting CTCs as prognostic biomarkers for late recurrences.
If the findings can be replicated in prospective clinical trials, CTC assay results could help clinicians choose treatments for patients who are at risk for late recurrence.
ECOG-ACRIN received funding for this study from the Breast Cancer Research Foundation, Susan G. Komen, and the National Cancer Institute. Dr. Sparano declared no conflicts of interest.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SAN ANTONIO – Although the initial ardor over the use of circulating tumor cells (CTCs) in cancer diagnosis has cooled, new research suggests that they may play a role in identifying late breast cancer recurrences in otherwise asymptomatic patients, according to members of the ECOG-ACRIN cancer research group.
In this video interview from the the San Antonio Breast Cancer Symposium, Joseph A. Sparano, MD, of Montefiore Medical Center and Albert Einstein College of Medicine in New York, describes ECOG-ACRIN’s experiments showing that patients with hormone receptor–positive disease and HER2-negative breast cancer have a significantly elevated risk for recurrence, supporting CTCs as prognostic biomarkers for late recurrences.
If the findings can be replicated in prospective clinical trials, CTC assay results could help clinicians choose treatments for patients who are at risk for late recurrence.
ECOG-ACRIN received funding for this study from the Breast Cancer Research Foundation, Susan G. Komen, and the National Cancer Institute. Dr. Sparano declared no conflicts of interest.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SAN ANTONIO – Although the initial ardor over the use of circulating tumor cells (CTCs) in cancer diagnosis has cooled, new research suggests that they may play a role in identifying late breast cancer recurrences in otherwise asymptomatic patients, according to members of the ECOG-ACRIN cancer research group.
In this video interview from the the San Antonio Breast Cancer Symposium, Joseph A. Sparano, MD, of Montefiore Medical Center and Albert Einstein College of Medicine in New York, describes ECOG-ACRIN’s experiments showing that patients with hormone receptor–positive disease and HER2-negative breast cancer have a significantly elevated risk for recurrence, supporting CTCs as prognostic biomarkers for late recurrences.
If the findings can be replicated in prospective clinical trials, CTC assay results could help clinicians choose treatments for patients who are at risk for late recurrence.
ECOG-ACRIN received funding for this study from the Breast Cancer Research Foundation, Susan G. Komen, and the National Cancer Institute. Dr. Sparano declared no conflicts of interest.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
REPORTING FROM SABCS 2017
Hallmark tumor metabolism becomes a validated therapeutic target
Altered cell metabolism has long been recognized as a distinctive feature of malignant cells but, until recently, research efforts had focused on a single aspect. It has become increasingly evident that many metabolic pathways are altered in cancer cells. Improved understanding has yielded the first regulatory approval in this new class of drugs. Here, we discuss the latest developments in the therapeutic targeting of the cancer metabolism hallmark.
A cancer cell’s sweet tooth
The metabolism of cancer cells differs from that of normal cells, an observation that has spawned a dedicated field of research and new targeted drug development. The German physiologist Otto Warburg is credited as the father of the field with his observations about the way in which cancer cells derive energy from glucose.1
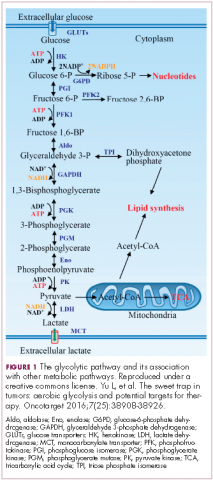
In normal cells, glucose is converted into pyruvate in the cytoplasm, which is then, most often, fed to the mitochondria that use oxidative phosphorylation to produce energy in the form of adenosine triphosphate (ATP). Cancer cells seem instead to favor using the pyruvate to produce lactate through glycolysis (Figure 1).
Glycolysis is usually reserved for conditions of poor oxygen availability, but although the tumor microenvironment is often hypoxic, cancer cells have been shown to use glycolysis even when oxygen is plentiful. As a result, the phenomenon is known as aerobic glycolysis, although it is most often referred to as the Warburg effect.2
Glycolysis is much less efficient than oxidative phosphorylation at producing energy, yielding only 2 ATP. In order to meet their energy demands in this way, cancer cells ramp up their glucose intake, an effect that has been exploited for the detection of cancer with positron-emission tomography.
Warburg postulated that this metabolic shift was a result of mitochondrial damage and defective oxidative phosphorylation, even going so far as to suggest that cancer was a mitochondrial disease. It has subsequently been shown that the mitochondria are mostly intact in cancer cells and that oxidative phosphorylation can still occur.3
The Warburg effect has been the subject of significant investigative efforts as researchers have attempted to better understand how this phenomenon comes about. Studies have shown that it is driven in large part by the transcription factors hypoxia inducible factor 1 alpha (HIF-1α) and c-Myc. In addition, numerous other signaling pathways, including the phosphatidylinositol 3-kinase (PI3K)-Akt-mammalian target of rapamycin (mTOR) pathway, and the activation of oncogenes and inactivation of tumor suppressors, are thought to play a central role.
HIF-1α is an oxygen-sensing transcription factor that coordinates cellular responses to reduced oxygen levels by binding to specific regions, known as hypoxia response elements, on target genes in the nucleus and regulating their subsequent expression. Oxygen levels and metabolism are tightly linked, and HIF-1α sits at the intersection of the 2 since many of its target genes are involved in metabolic pathways, including many glycolytic enzymes, but it also directly inhibits oxidative phosphorylation by suppressing key enzymes in this metabolic pathway.
The expression of HIF-1α and numerous glycolytic enzymes, including lactate dehydrogenase (LDH), phosphofructokinase (PFK), hexokinase II (HKII), and pyruvate dehydrogenase kinase (PDK) is increased in many tumor types. Other molecules that are associated with glucose uptake and metabolism are also dysregulated, such as the GLUT-1 glucose transporter.2,4-6
Targeting glycolysis and glucose uptake
According to one study, glucose transporters and glycolytic enzymes are overexpressed in 24 different types of cancer, representing more than 70% of all cancer cases.7 This enables cancer cells to respond metabolically as though they are experiencing hypoxia, even when oxygen is plentiful and, indeed, when hypoxia is a concern, to mount a faster response. It also provides a tempting avenue for anticancer drug design by exploiting the dependency of cancer cells on glycolysis to survive and thrive.
Inhibitors of HKII, LDH, PFK, PDK, and GLUT-1 have been and continue to be developed. For example, 2-deoxy-D-glucose is a glucose molecule in which the 2-hydroxyl group has been replaced by hydrogen, preventing further glycolysis; it acts as a competitive inhibitor of HKII. Dichloroacetate (DCA) activates the pyruvate dehydrogenase complex and inhibits the actions of the PDKs. Although development of DCA itself was unsuccessful, DCA derivatives continue to be pursued. WZB117 and STF-31 are novel small-molecule inhibitors of GLUT-1-mediated glucose transport. To date, where inhibitors of glycolysis have progressed into clinical trials, they have not proved successful, often limited by off-target effects and low potency.8-11
A variety of cell signaling pathways are implicated in metabolism by tightly regulating the ability of cells to gain access to and use nutrients. Through aberrations in these pathways, cancer cells can essentially go rogue, ignoring regulatory signals and taking up nutrients in an autonomous manner. One of the most frequently altered signaling pathways in human cancer, the PI3K-Akt-mTOR pathway, is also an important regulator of metabolism, coordinating the uptake of multiple nutrients, including glucose.
Akt in particular is thought to have a critical role in glucose metabolism and increased Akt pathway signaling has been shown to correlate with increased rates of glycolysis in cancer cells. Thus, Akt inhibitors could double as glycolytic or glucose transport inhibitors.12,13
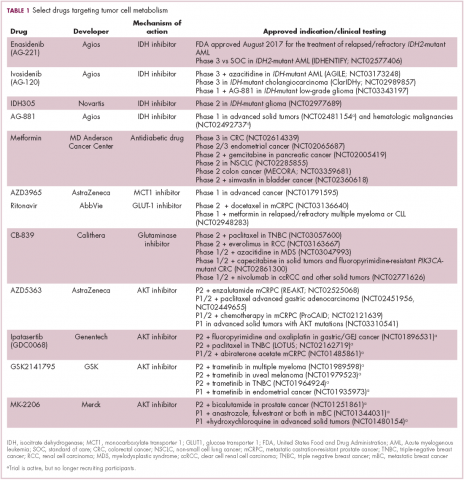
A number of Akt inhibitors are being evaluated in clinical trials (Table) and results from the phase 2 LOTUS trial of ipatasertib (GDC-0068) were recently published.
Among 124 patients randomly assigned to paclitaxel in combination with either ipatasertib or placebo, there was a modest improvement in progression-free survival (PFS) in the ipatasertib arm in patients with triple-negative breast cancer (TNBC; 6 months vs 4.2 months, respectively; hazard ratio [HR], 0.60; P = .037). The effect was more pronounced, though not statistically significant, in patients with phosphatase and tensin homolog (PTEN)-low tumors (6.2 months vs 3.7 months; HR, 0.59; P = .18). The most common grade 3 and higher adverse events (AEs) were diarrhea, reduced neutrophil count, and neutropenia.14
The Warburg paradox
Although the molecular mechanisms underlying the Warburg effect have been revealed to some extent, why cancer cells would choose to use such an energy-inefficient process when they have such high energy demands, remains something of a paradox. It’s still not entirely clear, but several explanations that are not necessarily mutually exclusive have been proposed and relate to the inherent benefits of glycolysis and might explain why cancer cells favor this pathway despite its poor energy yield. First, ATP is produced much more rapidly through glycolysis than oxidative phosphorylation, up to 100 times faster. Thus, using glycolysis is a trade-off, between making less energy and making it more quickly.
Second, cancer cells require more than just ATP to meet their metabolic demands. They need amino acids for protein synthesis; nucleotides for DNA replication; lipids for cell membrane synthesis; nicotinamide adenine dinucleotide phosphate (NADPH), which helps the cancer cell deal with oxidative stress; and various other metabolites. Glycolysis branches off into other metabolic pathways that generate many of these metabolites. Among these branched pathways is the pentose phosphate pathway (PPP), which is required for the generation of ribonucleotides and is a major source for NADPH. Cancer cells have been shown to upregulate the flux of glucose into the PPP to meet their anabolic demands and counter oxidative stress.
Third, the lactic acid produced through glycolysis is actively exported from tumor cells by monocarboxylate transporters (MCTs). This creates a highly acidic tumor microenvironment, which can promote several cancer-related processes and also plays a role in tumor-induced immunosuppression, by inhibiting the activity of tumor-infiltrating T cells, reducing dendritic cell maturation, and promoting the transformation of macrophages to a protumorigenic form.2,4,6
Beyond the Warburg effect
Although the focus has been on glucose metabolism and glycolysis, it has been increasingly recognized that many different metabolic pathways are altered. Fundamental changes to the metabolism of all 4 major classes of macromolecules – carbohydrates, lipids, proteins, and nucleic acids – have been observed, encompassing all aspects of cellular metabolism and enabling cancer cells to meet their complete metabolic requirements. There is also evidence that cancer cells are able to switch between different metabolic pathways depending on the availability of oxygen, their energetic needs, environmental stresses, and many other factors. Certainly, there is significant heterogeneity in the metabolic changes that occur in tumors, which vary from tumor to tumor and even within the same tumor and across the lifespan of a tumor as it progresses from an early stage to more advanced or metastatic disease.
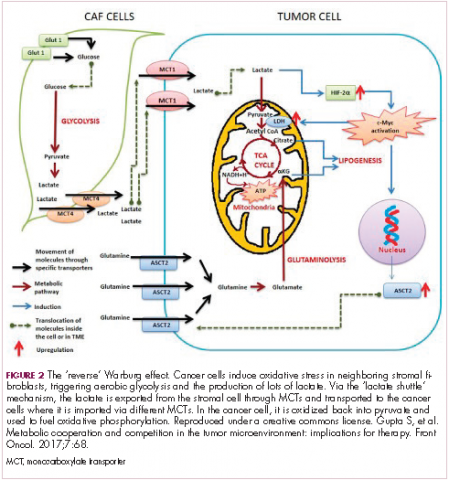
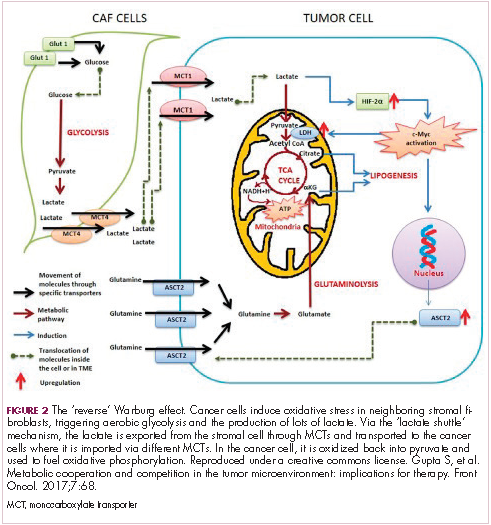
The notion of the Warburg effect as a universal phenomenon in cancer cells is now being widely disregarded. Many tumors continue to use oxidative phosphorylation, particularly slower growing tumors, to meet their energy needs. More recently a “reverse” Warburg effect was described, whereby cancer cells are thought to influence the metabolism of the surrounding stromal fibroblasts and essentially outsource aerobic glycolysis to these cells, while performing energy-efficient oxidative phosphorylation themselves (Figure 2).5,15,16
There is thought to be a “lactate shuttle” between the stromal and cancer cells. The stromal cells express high levels of efflux MCTs so that they can remove the subsequently high levels of lactate from the cytoplasm and avoid pickling themselves. The lactate is then shuttled to the cancer cells that have MCTs on their surface that are involved in lactate uptake. The cancer cells oxidize the lactate back into pyruvate, which can then be used in the tricarboxylic acid (TCA) cycle to feed oxidative phosphorylation for efficient ATP production. This hypothesis reflects a broader appreciation of the role of the microenvironment in contributing to cancer metabolism.17,18
An improved holistic understanding of cancer cell metabolism has led to the recognition of altered cancer metabolism as one of the hallmark abilities required for transformation of a normal cell into a cancerous one. It is categorized as “deregulation of bioenergetics” in the most up to date review of the cancer hallmarks.19 It has also begun to shape the therapeutic landscape as new drug targets have emerged.
IDH inhibitors first to market
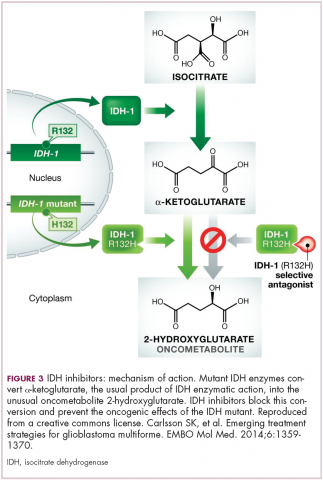
A number of new metabolically-targeted treatment strategies are being developed. Most promising are small molecule inhibitors of the isocitrate dehydrogenase (IDH) enzymes. These enzymes play an essential role in the TCA cycle, catalyzing the conversion of isocitrate to alpha-ketoglutarate, generating carbon dioxide and NADPH. Recurrent mutations in the IDH1 and IDH2 genes have been observed in several different types of cancer, including glioma, acute myeloid leukemia (AML), and cholangiocarcinoma.
IDH mutations are known as neomorphic mutations because they confer a new function on the altered gene product. In this case, the mutant IDH enzyme converts alpha-ketoglutarate further into D-2-hydroxyglutarate (D-2HG). This molecule has a number of different effects that promote tumorigenesis, including fostering defective DNA repair (Figure 3).20,21
Intriguing research presented at the American Association of Cancer Research Annual Meeting revealed that IDH mutations may make cancer cells more vulnerable to poly (ADP-ribose) polymerase (PARP) inhibition, likely as a result of defects in homologous recombination pathways of DNA repair.22The pursuit of IDH as a potential therapeutic target has yielded the first regulatory approval for a metabolically targeted anticancer therapy. In August 2017, the United States Food and Drug Administration (FDA) approved enasidenib, an IDH2 inhibitor, for the treatment of relapsed or refractory AML with an IDH2 mutation. It was approved in combination with a companion diagnostic, the RealTime IDH2 Assay, which is used to detect IDH2 mutations.
The approval was based on a single-arm trial in which responses occurred in almost a quarter of the 199 patients treated with 100 mg oral enasidenib daily. After a median follow-up of 6.6 months, 23% of the patients experienced a complete response or a complete response with partial hematologic recovery lasting a median of 8.2 months. The most common AEs were nausea, vomiting, diarrhea, elevated bilirubin levels, and reduced appetite.23
Several other IDH inhibitors are also showing encouraging efficacy. Ivosidenib is an IDH1 inhibitor and the results of a phase 1 study in patients with cholangiocarcinoma were recently presented at a leading conference. Escalating doses of ivosidenib (100 mg twice daily to 1,200 mg once daily) were administered to 73 patients (as of December 2016). The confirmed partial response (PR) rate was 6%, the rate of stable disease was 56%, and PFS at 6 months was 40%. There were no dose-limiting toxicities (DLTs) and treatment-emergent AEs included fatigue, nausea, vomiting, diarrhea, decreased appetite, dysgeusia, and QT prolongation.24
Another study of ivosidenib was presented at the 2017 annual meeting of the Society for Neuro-Oncology. In that study, patients with glioma received daily doses of ivosidenib ranging from 300 mg to 900 mg. Two patients had a minor response, 83% had stable disease, and the median PFS was 13 months. There were no DLTs and most AEs were mild to moderate and included, most commonly, headache, nausea, diarrhea, and vomiting.25
Pursuing alternative targets and repurposing drugs
Other metabolic targets that are being pursued include glutaminase, given the observation of significantly enhanced glutamine uptake in cancer cells. CB-839 is a glutaminase inhibitor that is currently being evaluated in phase 1 and 2 clinical trials. Updated clinical trial data from a phase 1 trial of CB-839 in combination with paclitaxel in patients with advanced/metastatic TNBC were presented at the San Antonio Breast Cancer Symposium last year.26
As of October 2017, 49 patients had been treated with 400 mg, 600 mg, or 800 mg CB-839 twice daily in combination with 80 mg/m2 intravenous paclitaxel weekly. Among the 44 patients evaluable for response, the rate of PR was 22% and of disease control, 59%. The one DLT was grade 3 neutropenia at the 400 mg dose. Overall AEs were mostly low grade and reversible.
In recent years, lactate has emerged as more than just a by-product of altered cancer cell metabolism. It is responsible, at least in part, for the highly acidic tumor microenvironment that fosters many of the other hallmarks of cancer. In addition, lactate promotes angiogenesis by upregulating HIF-1α in endothelial cells. Depriving tumor cells of the ability to export lactate is a potentially promising therapeutic strategy. An MCT-1 inhibitor, AZD3965, is being evaluated in early stage clinical trials.
Finally, several drugs that are renowned for their use in other disease settings are being repurposed for cancer therapy because of their potential effects on cancer cell metabolism. Ritonavir, an antiretroviral drug used in the treatment of HIV, is an inhibitor of GLUT-1 and is being evaluated in phase 1 and 2 clinical trials. Meanwhile, long-term studies of metformin, a drug that has revolutionized the treatment of diabetes, have revealed a reduction in the emergence of new cancers in diabetic patients treated who are treated with it, and the drug has been shown to improve breast cancer survival rates. Its precise anticancer effects are somewhat unclear, but it is thought to act in part by inhibiting oxidative phosphorylation. Numerous clinical trials of metformin in different types of cancer are ongoing.27,2
1. Warburg O. On respiratory impairment in cancer cells. Science. 1956;124(3215):269-270.
2. Yu L, Chen X, Wang L, Chen S. The sweet trap in tumors: aerobic glycolysis and potential targets for therapy. Oncotarget. 2016;7(25):38908-38926.
3. Warburg O. On the origin of cancer cells. Science. 1956;123(3191):309-314.
4. Chen XS, Li LY, Guan YD, Yang JM, Cheng Y. Anticancer strategies based on the metabolic profile of tumor cells: therapeutic targeting of the Warburg effect. Acta Pharmacol Sin. 2016;37(8):1013-1019.
5. Gupta S, Roy A, Dwarakanath BS. Metabolic cooperation and competition in the tumor microenvironment: implications for therapy. Front Oncol. 2017;7:68.
6. Marchiq I, Pouyssegur J. Hypoxia, cancer metabolism and the therapeutic benefit of targeting lactate/H(+) symporters. J Mol Med (Berl). 2016;94(2):155-171.
7. Altenberg B, Greulich KO. Genes of glycolysis are ubiquitously overexpressed in 24 cancer classes. Genomics. 2004;84(6):1014-1020.
8. Yu L, Chen X, Sun X, Wang L, Chen S. The glycolytic switch in tumors: how many players are involved? J Cancer. 2017;8(17):3430-3440.
9. Zhang W, Zhang SL, Hu X, Tam KY. Targeting tumor metabolism for cancer treatment: is pyruvate dehydrogenase kinases (PDKs) a viable anticancer target? Int J Biol Sci. 2015;11(12):1390-1400.
10. Talekar M, Boreddy SR, Singh A, Amiji M. Tumor aerobic glycolysis: new insights into therapeutic strategies with targeted delivery. Expert Opin Biol Ther. 2014;14(8):1145-1159.
11. Ganapathy-Kanniappan S, Geschwind JF. Tumor glycolysis as a target for cancer therapy: progress and prospects. Mol Cancer. 2013;12:152.
12. Lien EC, Lyssiotis CA, Cantley LC. Metabolic reprogramming by the PI3K-Akt-mTOR pathway in cancer. In: Cramer T, Schmitt CA, eds. Metabolism in Cancer. Cham, Switzerland: Springer International Publishing; 2016:39-72.
13. Simons AL, Orcutt KP, Madsen JM, Scarbrough PM, Spitz DR. The role of Akt pathway signaling in glucose metabolism and metabolic oxidative stress. In: Spitz DR, Dornfeld KJ, Krishnan K, Gius D (eds). Oxidative stress in cancer biology and therapy. Humana Press (copyright holder, Springer Science+Business Media, LLC); 2012:21-46.
14. Kim S-B, Dent R, Im S-A, et al. Ipatasertib plus paclitaxel versus placebo plus paclitaxel as first-line therapy for metastatic triple-negative breast cancer (LOTUS): a multicentre, randomised, double-blind, placebo-controlled, phase 2 trial. Lancet Oncol. 2017;18(10):1360-1372.
15. Fu Y, Liu S, Yin S, et al. The reverse Warburg effect is likely to be an Achilles’ heel of cancer that can be exploited for cancer therapy. Oncotarget. 2017;8(34):57813-57825.
16. Wilde L, Roche M, Domingo-Vidal M, et al. Metabolic coupling and the reverse Warburg effect in cancer: implications for novel biomarker and anticancer agent development. Semin Oncol. 2017;44(3):198-203.
17. Brooks GA. Cell–cell and intracellular lactate shuttles. Journal Physiol. 2009;587(23):5591-5600.
18. Chiarugi P, Cirri P. Metabolic exchanges within tumor microenvironment. Cancer Lett. 2016;380(1):272-280.
19. Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144(5):646-674.
20. Fujii T, Khawaja MR, DiNardo CD, Atkins JT, Janku F. Targeting isocitrate dehydrogenase (IDH) in cancer. Discov Med. 2016;21(117):373-380.
21. Carlsson SK, Brothers SP, Wahlestedt C. Emerging treatment strategies for glioblastoma multiforme. EMBO Mol Med. 2014;6(11):1359-1370.
22. Lu Y, Kwintkiewicz J, Liu Y, et al. Chemosensitivity of IDH1-mutated gliomas due to an impairment in PARP1-mediated DNA repair. Cancer Res. 2017;77(7):1709-1718.
23. Stein EM, DiNardo CD, Pollyea DA, et al. Enasidenib in mutant IDH2 relapsed or refractory acute myeloid leukemia. Blood. 2017;130(6):722-731.
24. Lowery MA, Abou-Alfa GK, Burris HA, et al. Phase I study of AG-120, an IDH1 mutant enzyme inhibitor: results from the cholangiocarcinoma dose escalation and expansion cohorts. J Clin Oncol. 2017;35(15_suppl):4015-4015.
25. Mellinghoff IK, Touat M, Maher E, et al. ACTR-46. AG-120, a first-in-class mutant IDH1 inhibitor in patients with recurrent or progressive IDH1 mutant glioma: updated results from the phase 1 non-enhancing glioma population. Neuro Oncol. 2017;19(suppl_6):vi10-vi11.
26. Kalinsky K, Harding J, DeMichele A, et al. Phase 1 study of CB-839, a first-in-class oral inhibitor of glutaminase, in combination with paclitaxel in patients with advanced triple negative breast cancer. Paper presented at San Antonio Breast Cancer Symposium; December 5-9, 2017; San Antonio, Texas.
27. Hatoum D, McGowan EM. Recent advances in the use of metformin: can treating diabetes prevent breast cancer? Biomed Res Int. 2015;2015:548436.
28. Leone A, Di Gennaro E, Bruzzese F, Avallone A, Budillon A. New perspective for an old antidiabetic drug: metformin as anticancer agent. Cancer Treat Res. 2014;159:355-376.
Altered cell metabolism has long been recognized as a distinctive feature of malignant cells but, until recently, research efforts had focused on a single aspect. It has become increasingly evident that many metabolic pathways are altered in cancer cells. Improved understanding has yielded the first regulatory approval in this new class of drugs. Here, we discuss the latest developments in the therapeutic targeting of the cancer metabolism hallmark.
A cancer cell’s sweet tooth
The metabolism of cancer cells differs from that of normal cells, an observation that has spawned a dedicated field of research and new targeted drug development. The German physiologist Otto Warburg is credited as the father of the field with his observations about the way in which cancer cells derive energy from glucose.1
In normal cells, glucose is converted into pyruvate in the cytoplasm, which is then, most often, fed to the mitochondria that use oxidative phosphorylation to produce energy in the form of adenosine triphosphate (ATP). Cancer cells seem instead to favor using the pyruvate to produce lactate through glycolysis (Figure 1).
Glycolysis is usually reserved for conditions of poor oxygen availability, but although the tumor microenvironment is often hypoxic, cancer cells have been shown to use glycolysis even when oxygen is plentiful. As a result, the phenomenon is known as aerobic glycolysis, although it is most often referred to as the Warburg effect.2
Glycolysis is much less efficient than oxidative phosphorylation at producing energy, yielding only 2 ATP. In order to meet their energy demands in this way, cancer cells ramp up their glucose intake, an effect that has been exploited for the detection of cancer with positron-emission tomography.
Warburg postulated that this metabolic shift was a result of mitochondrial damage and defective oxidative phosphorylation, even going so far as to suggest that cancer was a mitochondrial disease. It has subsequently been shown that the mitochondria are mostly intact in cancer cells and that oxidative phosphorylation can still occur.3
The Warburg effect has been the subject of significant investigative efforts as researchers have attempted to better understand how this phenomenon comes about. Studies have shown that it is driven in large part by the transcription factors hypoxia inducible factor 1 alpha (HIF-1α) and c-Myc. In addition, numerous other signaling pathways, including the phosphatidylinositol 3-kinase (PI3K)-Akt-mammalian target of rapamycin (mTOR) pathway, and the activation of oncogenes and inactivation of tumor suppressors, are thought to play a central role.
HIF-1α is an oxygen-sensing transcription factor that coordinates cellular responses to reduced oxygen levels by binding to specific regions, known as hypoxia response elements, on target genes in the nucleus and regulating their subsequent expression. Oxygen levels and metabolism are tightly linked, and HIF-1α sits at the intersection of the 2 since many of its target genes are involved in metabolic pathways, including many glycolytic enzymes, but it also directly inhibits oxidative phosphorylation by suppressing key enzymes in this metabolic pathway.
The expression of HIF-1α and numerous glycolytic enzymes, including lactate dehydrogenase (LDH), phosphofructokinase (PFK), hexokinase II (HKII), and pyruvate dehydrogenase kinase (PDK) is increased in many tumor types. Other molecules that are associated with glucose uptake and metabolism are also dysregulated, such as the GLUT-1 glucose transporter.2,4-6
Targeting glycolysis and glucose uptake
According to one study, glucose transporters and glycolytic enzymes are overexpressed in 24 different types of cancer, representing more than 70% of all cancer cases.7 This enables cancer cells to respond metabolically as though they are experiencing hypoxia, even when oxygen is plentiful and, indeed, when hypoxia is a concern, to mount a faster response. It also provides a tempting avenue for anticancer drug design by exploiting the dependency of cancer cells on glycolysis to survive and thrive.
Inhibitors of HKII, LDH, PFK, PDK, and GLUT-1 have been and continue to be developed. For example, 2-deoxy-D-glucose is a glucose molecule in which the 2-hydroxyl group has been replaced by hydrogen, preventing further glycolysis; it acts as a competitive inhibitor of HKII. Dichloroacetate (DCA) activates the pyruvate dehydrogenase complex and inhibits the actions of the PDKs. Although development of DCA itself was unsuccessful, DCA derivatives continue to be pursued. WZB117 and STF-31 are novel small-molecule inhibitors of GLUT-1-mediated glucose transport. To date, where inhibitors of glycolysis have progressed into clinical trials, they have not proved successful, often limited by off-target effects and low potency.8-11
A variety of cell signaling pathways are implicated in metabolism by tightly regulating the ability of cells to gain access to and use nutrients. Through aberrations in these pathways, cancer cells can essentially go rogue, ignoring regulatory signals and taking up nutrients in an autonomous manner. One of the most frequently altered signaling pathways in human cancer, the PI3K-Akt-mTOR pathway, is also an important regulator of metabolism, coordinating the uptake of multiple nutrients, including glucose.
Akt in particular is thought to have a critical role in glucose metabolism and increased Akt pathway signaling has been shown to correlate with increased rates of glycolysis in cancer cells. Thus, Akt inhibitors could double as glycolytic or glucose transport inhibitors.12,13
A number of Akt inhibitors are being evaluated in clinical trials (Table) and results from the phase 2 LOTUS trial of ipatasertib (GDC-0068) were recently published.
Among 124 patients randomly assigned to paclitaxel in combination with either ipatasertib or placebo, there was a modest improvement in progression-free survival (PFS) in the ipatasertib arm in patients with triple-negative breast cancer (TNBC; 6 months vs 4.2 months, respectively; hazard ratio [HR], 0.60; P = .037). The effect was more pronounced, though not statistically significant, in patients with phosphatase and tensin homolog (PTEN)-low tumors (6.2 months vs 3.7 months; HR, 0.59; P = .18). The most common grade 3 and higher adverse events (AEs) were diarrhea, reduced neutrophil count, and neutropenia.14
The Warburg paradox
Although the molecular mechanisms underlying the Warburg effect have been revealed to some extent, why cancer cells would choose to use such an energy-inefficient process when they have such high energy demands, remains something of a paradox. It’s still not entirely clear, but several explanations that are not necessarily mutually exclusive have been proposed and relate to the inherent benefits of glycolysis and might explain why cancer cells favor this pathway despite its poor energy yield. First, ATP is produced much more rapidly through glycolysis than oxidative phosphorylation, up to 100 times faster. Thus, using glycolysis is a trade-off, between making less energy and making it more quickly.
Second, cancer cells require more than just ATP to meet their metabolic demands. They need amino acids for protein synthesis; nucleotides for DNA replication; lipids for cell membrane synthesis; nicotinamide adenine dinucleotide phosphate (NADPH), which helps the cancer cell deal with oxidative stress; and various other metabolites. Glycolysis branches off into other metabolic pathways that generate many of these metabolites. Among these branched pathways is the pentose phosphate pathway (PPP), which is required for the generation of ribonucleotides and is a major source for NADPH. Cancer cells have been shown to upregulate the flux of glucose into the PPP to meet their anabolic demands and counter oxidative stress.
Third, the lactic acid produced through glycolysis is actively exported from tumor cells by monocarboxylate transporters (MCTs). This creates a highly acidic tumor microenvironment, which can promote several cancer-related processes and also plays a role in tumor-induced immunosuppression, by inhibiting the activity of tumor-infiltrating T cells, reducing dendritic cell maturation, and promoting the transformation of macrophages to a protumorigenic form.2,4,6
Beyond the Warburg effect
Although the focus has been on glucose metabolism and glycolysis, it has been increasingly recognized that many different metabolic pathways are altered. Fundamental changes to the metabolism of all 4 major classes of macromolecules – carbohydrates, lipids, proteins, and nucleic acids – have been observed, encompassing all aspects of cellular metabolism and enabling cancer cells to meet their complete metabolic requirements. There is also evidence that cancer cells are able to switch between different metabolic pathways depending on the availability of oxygen, their energetic needs, environmental stresses, and many other factors. Certainly, there is significant heterogeneity in the metabolic changes that occur in tumors, which vary from tumor to tumor and even within the same tumor and across the lifespan of a tumor as it progresses from an early stage to more advanced or metastatic disease.
The notion of the Warburg effect as a universal phenomenon in cancer cells is now being widely disregarded. Many tumors continue to use oxidative phosphorylation, particularly slower growing tumors, to meet their energy needs. More recently a “reverse” Warburg effect was described, whereby cancer cells are thought to influence the metabolism of the surrounding stromal fibroblasts and essentially outsource aerobic glycolysis to these cells, while performing energy-efficient oxidative phosphorylation themselves (Figure 2).5,15,16
There is thought to be a “lactate shuttle” between the stromal and cancer cells. The stromal cells express high levels of efflux MCTs so that they can remove the subsequently high levels of lactate from the cytoplasm and avoid pickling themselves. The lactate is then shuttled to the cancer cells that have MCTs on their surface that are involved in lactate uptake. The cancer cells oxidize the lactate back into pyruvate, which can then be used in the tricarboxylic acid (TCA) cycle to feed oxidative phosphorylation for efficient ATP production. This hypothesis reflects a broader appreciation of the role of the microenvironment in contributing to cancer metabolism.17,18
An improved holistic understanding of cancer cell metabolism has led to the recognition of altered cancer metabolism as one of the hallmark abilities required for transformation of a normal cell into a cancerous one. It is categorized as “deregulation of bioenergetics” in the most up to date review of the cancer hallmarks.19 It has also begun to shape the therapeutic landscape as new drug targets have emerged.
IDH inhibitors first to market
A number of new metabolically-targeted treatment strategies are being developed. Most promising are small molecule inhibitors of the isocitrate dehydrogenase (IDH) enzymes. These enzymes play an essential role in the TCA cycle, catalyzing the conversion of isocitrate to alpha-ketoglutarate, generating carbon dioxide and NADPH. Recurrent mutations in the IDH1 and IDH2 genes have been observed in several different types of cancer, including glioma, acute myeloid leukemia (AML), and cholangiocarcinoma.
IDH mutations are known as neomorphic mutations because they confer a new function on the altered gene product. In this case, the mutant IDH enzyme converts alpha-ketoglutarate further into D-2-hydroxyglutarate (D-2HG). This molecule has a number of different effects that promote tumorigenesis, including fostering defective DNA repair (Figure 3).20,21
Intriguing research presented at the American Association of Cancer Research Annual Meeting revealed that IDH mutations may make cancer cells more vulnerable to poly (ADP-ribose) polymerase (PARP) inhibition, likely as a result of defects in homologous recombination pathways of DNA repair.22The pursuit of IDH as a potential therapeutic target has yielded the first regulatory approval for a metabolically targeted anticancer therapy. In August 2017, the United States Food and Drug Administration (FDA) approved enasidenib, an IDH2 inhibitor, for the treatment of relapsed or refractory AML with an IDH2 mutation. It was approved in combination with a companion diagnostic, the RealTime IDH2 Assay, which is used to detect IDH2 mutations.
The approval was based on a single-arm trial in which responses occurred in almost a quarter of the 199 patients treated with 100 mg oral enasidenib daily. After a median follow-up of 6.6 months, 23% of the patients experienced a complete response or a complete response with partial hematologic recovery lasting a median of 8.2 months. The most common AEs were nausea, vomiting, diarrhea, elevated bilirubin levels, and reduced appetite.23
Several other IDH inhibitors are also showing encouraging efficacy. Ivosidenib is an IDH1 inhibitor and the results of a phase 1 study in patients with cholangiocarcinoma were recently presented at a leading conference. Escalating doses of ivosidenib (100 mg twice daily to 1,200 mg once daily) were administered to 73 patients (as of December 2016). The confirmed partial response (PR) rate was 6%, the rate of stable disease was 56%, and PFS at 6 months was 40%. There were no dose-limiting toxicities (DLTs) and treatment-emergent AEs included fatigue, nausea, vomiting, diarrhea, decreased appetite, dysgeusia, and QT prolongation.24
Another study of ivosidenib was presented at the 2017 annual meeting of the Society for Neuro-Oncology. In that study, patients with glioma received daily doses of ivosidenib ranging from 300 mg to 900 mg. Two patients had a minor response, 83% had stable disease, and the median PFS was 13 months. There were no DLTs and most AEs were mild to moderate and included, most commonly, headache, nausea, diarrhea, and vomiting.25
Pursuing alternative targets and repurposing drugs
Other metabolic targets that are being pursued include glutaminase, given the observation of significantly enhanced glutamine uptake in cancer cells. CB-839 is a glutaminase inhibitor that is currently being evaluated in phase 1 and 2 clinical trials. Updated clinical trial data from a phase 1 trial of CB-839 in combination with paclitaxel in patients with advanced/metastatic TNBC were presented at the San Antonio Breast Cancer Symposium last year.26
As of October 2017, 49 patients had been treated with 400 mg, 600 mg, or 800 mg CB-839 twice daily in combination with 80 mg/m2 intravenous paclitaxel weekly. Among the 44 patients evaluable for response, the rate of PR was 22% and of disease control, 59%. The one DLT was grade 3 neutropenia at the 400 mg dose. Overall AEs were mostly low grade and reversible.
In recent years, lactate has emerged as more than just a by-product of altered cancer cell metabolism. It is responsible, at least in part, for the highly acidic tumor microenvironment that fosters many of the other hallmarks of cancer. In addition, lactate promotes angiogenesis by upregulating HIF-1α in endothelial cells. Depriving tumor cells of the ability to export lactate is a potentially promising therapeutic strategy. An MCT-1 inhibitor, AZD3965, is being evaluated in early stage clinical trials.
Finally, several drugs that are renowned for their use in other disease settings are being repurposed for cancer therapy because of their potential effects on cancer cell metabolism. Ritonavir, an antiretroviral drug used in the treatment of HIV, is an inhibitor of GLUT-1 and is being evaluated in phase 1 and 2 clinical trials. Meanwhile, long-term studies of metformin, a drug that has revolutionized the treatment of diabetes, have revealed a reduction in the emergence of new cancers in diabetic patients treated who are treated with it, and the drug has been shown to improve breast cancer survival rates. Its precise anticancer effects are somewhat unclear, but it is thought to act in part by inhibiting oxidative phosphorylation. Numerous clinical trials of metformin in different types of cancer are ongoing.27,2
Altered cell metabolism has long been recognized as a distinctive feature of malignant cells but, until recently, research efforts had focused on a single aspect. It has become increasingly evident that many metabolic pathways are altered in cancer cells. Improved understanding has yielded the first regulatory approval in this new class of drugs. Here, we discuss the latest developments in the therapeutic targeting of the cancer metabolism hallmark.
A cancer cell’s sweet tooth
The metabolism of cancer cells differs from that of normal cells, an observation that has spawned a dedicated field of research and new targeted drug development. The German physiologist Otto Warburg is credited as the father of the field with his observations about the way in which cancer cells derive energy from glucose.1
In normal cells, glucose is converted into pyruvate in the cytoplasm, which is then, most often, fed to the mitochondria that use oxidative phosphorylation to produce energy in the form of adenosine triphosphate (ATP). Cancer cells seem instead to favor using the pyruvate to produce lactate through glycolysis (Figure 1).
Glycolysis is usually reserved for conditions of poor oxygen availability, but although the tumor microenvironment is often hypoxic, cancer cells have been shown to use glycolysis even when oxygen is plentiful. As a result, the phenomenon is known as aerobic glycolysis, although it is most often referred to as the Warburg effect.2
Glycolysis is much less efficient than oxidative phosphorylation at producing energy, yielding only 2 ATP. In order to meet their energy demands in this way, cancer cells ramp up their glucose intake, an effect that has been exploited for the detection of cancer with positron-emission tomography.
Warburg postulated that this metabolic shift was a result of mitochondrial damage and defective oxidative phosphorylation, even going so far as to suggest that cancer was a mitochondrial disease. It has subsequently been shown that the mitochondria are mostly intact in cancer cells and that oxidative phosphorylation can still occur.3
The Warburg effect has been the subject of significant investigative efforts as researchers have attempted to better understand how this phenomenon comes about. Studies have shown that it is driven in large part by the transcription factors hypoxia inducible factor 1 alpha (HIF-1α) and c-Myc. In addition, numerous other signaling pathways, including the phosphatidylinositol 3-kinase (PI3K)-Akt-mammalian target of rapamycin (mTOR) pathway, and the activation of oncogenes and inactivation of tumor suppressors, are thought to play a central role.
HIF-1α is an oxygen-sensing transcription factor that coordinates cellular responses to reduced oxygen levels by binding to specific regions, known as hypoxia response elements, on target genes in the nucleus and regulating their subsequent expression. Oxygen levels and metabolism are tightly linked, and HIF-1α sits at the intersection of the 2 since many of its target genes are involved in metabolic pathways, including many glycolytic enzymes, but it also directly inhibits oxidative phosphorylation by suppressing key enzymes in this metabolic pathway.
The expression of HIF-1α and numerous glycolytic enzymes, including lactate dehydrogenase (LDH), phosphofructokinase (PFK), hexokinase II (HKII), and pyruvate dehydrogenase kinase (PDK) is increased in many tumor types. Other molecules that are associated with glucose uptake and metabolism are also dysregulated, such as the GLUT-1 glucose transporter.2,4-6
Targeting glycolysis and glucose uptake
According to one study, glucose transporters and glycolytic enzymes are overexpressed in 24 different types of cancer, representing more than 70% of all cancer cases.7 This enables cancer cells to respond metabolically as though they are experiencing hypoxia, even when oxygen is plentiful and, indeed, when hypoxia is a concern, to mount a faster response. It also provides a tempting avenue for anticancer drug design by exploiting the dependency of cancer cells on glycolysis to survive and thrive.
Inhibitors of HKII, LDH, PFK, PDK, and GLUT-1 have been and continue to be developed. For example, 2-deoxy-D-glucose is a glucose molecule in which the 2-hydroxyl group has been replaced by hydrogen, preventing further glycolysis; it acts as a competitive inhibitor of HKII. Dichloroacetate (DCA) activates the pyruvate dehydrogenase complex and inhibits the actions of the PDKs. Although development of DCA itself was unsuccessful, DCA derivatives continue to be pursued. WZB117 and STF-31 are novel small-molecule inhibitors of GLUT-1-mediated glucose transport. To date, where inhibitors of glycolysis have progressed into clinical trials, they have not proved successful, often limited by off-target effects and low potency.8-11
A variety of cell signaling pathways are implicated in metabolism by tightly regulating the ability of cells to gain access to and use nutrients. Through aberrations in these pathways, cancer cells can essentially go rogue, ignoring regulatory signals and taking up nutrients in an autonomous manner. One of the most frequently altered signaling pathways in human cancer, the PI3K-Akt-mTOR pathway, is also an important regulator of metabolism, coordinating the uptake of multiple nutrients, including glucose.
Akt in particular is thought to have a critical role in glucose metabolism and increased Akt pathway signaling has been shown to correlate with increased rates of glycolysis in cancer cells. Thus, Akt inhibitors could double as glycolytic or glucose transport inhibitors.12,13
A number of Akt inhibitors are being evaluated in clinical trials (Table) and results from the phase 2 LOTUS trial of ipatasertib (GDC-0068) were recently published.
Among 124 patients randomly assigned to paclitaxel in combination with either ipatasertib or placebo, there was a modest improvement in progression-free survival (PFS) in the ipatasertib arm in patients with triple-negative breast cancer (TNBC; 6 months vs 4.2 months, respectively; hazard ratio [HR], 0.60; P = .037). The effect was more pronounced, though not statistically significant, in patients with phosphatase and tensin homolog (PTEN)-low tumors (6.2 months vs 3.7 months; HR, 0.59; P = .18). The most common grade 3 and higher adverse events (AEs) were diarrhea, reduced neutrophil count, and neutropenia.14
The Warburg paradox
Although the molecular mechanisms underlying the Warburg effect have been revealed to some extent, why cancer cells would choose to use such an energy-inefficient process when they have such high energy demands, remains something of a paradox. It’s still not entirely clear, but several explanations that are not necessarily mutually exclusive have been proposed and relate to the inherent benefits of glycolysis and might explain why cancer cells favor this pathway despite its poor energy yield. First, ATP is produced much more rapidly through glycolysis than oxidative phosphorylation, up to 100 times faster. Thus, using glycolysis is a trade-off, between making less energy and making it more quickly.
Second, cancer cells require more than just ATP to meet their metabolic demands. They need amino acids for protein synthesis; nucleotides for DNA replication; lipids for cell membrane synthesis; nicotinamide adenine dinucleotide phosphate (NADPH), which helps the cancer cell deal with oxidative stress; and various other metabolites. Glycolysis branches off into other metabolic pathways that generate many of these metabolites. Among these branched pathways is the pentose phosphate pathway (PPP), which is required for the generation of ribonucleotides and is a major source for NADPH. Cancer cells have been shown to upregulate the flux of glucose into the PPP to meet their anabolic demands and counter oxidative stress.
Third, the lactic acid produced through glycolysis is actively exported from tumor cells by monocarboxylate transporters (MCTs). This creates a highly acidic tumor microenvironment, which can promote several cancer-related processes and also plays a role in tumor-induced immunosuppression, by inhibiting the activity of tumor-infiltrating T cells, reducing dendritic cell maturation, and promoting the transformation of macrophages to a protumorigenic form.2,4,6
Beyond the Warburg effect
Although the focus has been on glucose metabolism and glycolysis, it has been increasingly recognized that many different metabolic pathways are altered. Fundamental changes to the metabolism of all 4 major classes of macromolecules – carbohydrates, lipids, proteins, and nucleic acids – have been observed, encompassing all aspects of cellular metabolism and enabling cancer cells to meet their complete metabolic requirements. There is also evidence that cancer cells are able to switch between different metabolic pathways depending on the availability of oxygen, their energetic needs, environmental stresses, and many other factors. Certainly, there is significant heterogeneity in the metabolic changes that occur in tumors, which vary from tumor to tumor and even within the same tumor and across the lifespan of a tumor as it progresses from an early stage to more advanced or metastatic disease.
The notion of the Warburg effect as a universal phenomenon in cancer cells is now being widely disregarded. Many tumors continue to use oxidative phosphorylation, particularly slower growing tumors, to meet their energy needs. More recently a “reverse” Warburg effect was described, whereby cancer cells are thought to influence the metabolism of the surrounding stromal fibroblasts and essentially outsource aerobic glycolysis to these cells, while performing energy-efficient oxidative phosphorylation themselves (Figure 2).5,15,16
There is thought to be a “lactate shuttle” between the stromal and cancer cells. The stromal cells express high levels of efflux MCTs so that they can remove the subsequently high levels of lactate from the cytoplasm and avoid pickling themselves. The lactate is then shuttled to the cancer cells that have MCTs on their surface that are involved in lactate uptake. The cancer cells oxidize the lactate back into pyruvate, which can then be used in the tricarboxylic acid (TCA) cycle to feed oxidative phosphorylation for efficient ATP production. This hypothesis reflects a broader appreciation of the role of the microenvironment in contributing to cancer metabolism.17,18
An improved holistic understanding of cancer cell metabolism has led to the recognition of altered cancer metabolism as one of the hallmark abilities required for transformation of a normal cell into a cancerous one. It is categorized as “deregulation of bioenergetics” in the most up to date review of the cancer hallmarks.19 It has also begun to shape the therapeutic landscape as new drug targets have emerged.
IDH inhibitors first to market
A number of new metabolically-targeted treatment strategies are being developed. Most promising are small molecule inhibitors of the isocitrate dehydrogenase (IDH) enzymes. These enzymes play an essential role in the TCA cycle, catalyzing the conversion of isocitrate to alpha-ketoglutarate, generating carbon dioxide and NADPH. Recurrent mutations in the IDH1 and IDH2 genes have been observed in several different types of cancer, including glioma, acute myeloid leukemia (AML), and cholangiocarcinoma.
IDH mutations are known as neomorphic mutations because they confer a new function on the altered gene product. In this case, the mutant IDH enzyme converts alpha-ketoglutarate further into D-2-hydroxyglutarate (D-2HG). This molecule has a number of different effects that promote tumorigenesis, including fostering defective DNA repair (Figure 3).20,21
Intriguing research presented at the American Association of Cancer Research Annual Meeting revealed that IDH mutations may make cancer cells more vulnerable to poly (ADP-ribose) polymerase (PARP) inhibition, likely as a result of defects in homologous recombination pathways of DNA repair.22The pursuit of IDH as a potential therapeutic target has yielded the first regulatory approval for a metabolically targeted anticancer therapy. In August 2017, the United States Food and Drug Administration (FDA) approved enasidenib, an IDH2 inhibitor, for the treatment of relapsed or refractory AML with an IDH2 mutation. It was approved in combination with a companion diagnostic, the RealTime IDH2 Assay, which is used to detect IDH2 mutations.
The approval was based on a single-arm trial in which responses occurred in almost a quarter of the 199 patients treated with 100 mg oral enasidenib daily. After a median follow-up of 6.6 months, 23% of the patients experienced a complete response or a complete response with partial hematologic recovery lasting a median of 8.2 months. The most common AEs were nausea, vomiting, diarrhea, elevated bilirubin levels, and reduced appetite.23
Several other IDH inhibitors are also showing encouraging efficacy. Ivosidenib is an IDH1 inhibitor and the results of a phase 1 study in patients with cholangiocarcinoma were recently presented at a leading conference. Escalating doses of ivosidenib (100 mg twice daily to 1,200 mg once daily) were administered to 73 patients (as of December 2016). The confirmed partial response (PR) rate was 6%, the rate of stable disease was 56%, and PFS at 6 months was 40%. There were no dose-limiting toxicities (DLTs) and treatment-emergent AEs included fatigue, nausea, vomiting, diarrhea, decreased appetite, dysgeusia, and QT prolongation.24
Another study of ivosidenib was presented at the 2017 annual meeting of the Society for Neuro-Oncology. In that study, patients with glioma received daily doses of ivosidenib ranging from 300 mg to 900 mg. Two patients had a minor response, 83% had stable disease, and the median PFS was 13 months. There were no DLTs and most AEs were mild to moderate and included, most commonly, headache, nausea, diarrhea, and vomiting.25
Pursuing alternative targets and repurposing drugs
Other metabolic targets that are being pursued include glutaminase, given the observation of significantly enhanced glutamine uptake in cancer cells. CB-839 is a glutaminase inhibitor that is currently being evaluated in phase 1 and 2 clinical trials. Updated clinical trial data from a phase 1 trial of CB-839 in combination with paclitaxel in patients with advanced/metastatic TNBC were presented at the San Antonio Breast Cancer Symposium last year.26
As of October 2017, 49 patients had been treated with 400 mg, 600 mg, or 800 mg CB-839 twice daily in combination with 80 mg/m2 intravenous paclitaxel weekly. Among the 44 patients evaluable for response, the rate of PR was 22% and of disease control, 59%. The one DLT was grade 3 neutropenia at the 400 mg dose. Overall AEs were mostly low grade and reversible.
In recent years, lactate has emerged as more than just a by-product of altered cancer cell metabolism. It is responsible, at least in part, for the highly acidic tumor microenvironment that fosters many of the other hallmarks of cancer. In addition, lactate promotes angiogenesis by upregulating HIF-1α in endothelial cells. Depriving tumor cells of the ability to export lactate is a potentially promising therapeutic strategy. An MCT-1 inhibitor, AZD3965, is being evaluated in early stage clinical trials.
Finally, several drugs that are renowned for their use in other disease settings are being repurposed for cancer therapy because of their potential effects on cancer cell metabolism. Ritonavir, an antiretroviral drug used in the treatment of HIV, is an inhibitor of GLUT-1 and is being evaluated in phase 1 and 2 clinical trials. Meanwhile, long-term studies of metformin, a drug that has revolutionized the treatment of diabetes, have revealed a reduction in the emergence of new cancers in diabetic patients treated who are treated with it, and the drug has been shown to improve breast cancer survival rates. Its precise anticancer effects are somewhat unclear, but it is thought to act in part by inhibiting oxidative phosphorylation. Numerous clinical trials of metformin in different types of cancer are ongoing.27,2
1. Warburg O. On respiratory impairment in cancer cells. Science. 1956;124(3215):269-270.
2. Yu L, Chen X, Wang L, Chen S. The sweet trap in tumors: aerobic glycolysis and potential targets for therapy. Oncotarget. 2016;7(25):38908-38926.
3. Warburg O. On the origin of cancer cells. Science. 1956;123(3191):309-314.
4. Chen XS, Li LY, Guan YD, Yang JM, Cheng Y. Anticancer strategies based on the metabolic profile of tumor cells: therapeutic targeting of the Warburg effect. Acta Pharmacol Sin. 2016;37(8):1013-1019.
5. Gupta S, Roy A, Dwarakanath BS. Metabolic cooperation and competition in the tumor microenvironment: implications for therapy. Front Oncol. 2017;7:68.
6. Marchiq I, Pouyssegur J. Hypoxia, cancer metabolism and the therapeutic benefit of targeting lactate/H(+) symporters. J Mol Med (Berl). 2016;94(2):155-171.
7. Altenberg B, Greulich KO. Genes of glycolysis are ubiquitously overexpressed in 24 cancer classes. Genomics. 2004;84(6):1014-1020.
8. Yu L, Chen X, Sun X, Wang L, Chen S. The glycolytic switch in tumors: how many players are involved? J Cancer. 2017;8(17):3430-3440.
9. Zhang W, Zhang SL, Hu X, Tam KY. Targeting tumor metabolism for cancer treatment: is pyruvate dehydrogenase kinases (PDKs) a viable anticancer target? Int J Biol Sci. 2015;11(12):1390-1400.
10. Talekar M, Boreddy SR, Singh A, Amiji M. Tumor aerobic glycolysis: new insights into therapeutic strategies with targeted delivery. Expert Opin Biol Ther. 2014;14(8):1145-1159.
11. Ganapathy-Kanniappan S, Geschwind JF. Tumor glycolysis as a target for cancer therapy: progress and prospects. Mol Cancer. 2013;12:152.
12. Lien EC, Lyssiotis CA, Cantley LC. Metabolic reprogramming by the PI3K-Akt-mTOR pathway in cancer. In: Cramer T, Schmitt CA, eds. Metabolism in Cancer. Cham, Switzerland: Springer International Publishing; 2016:39-72.
13. Simons AL, Orcutt KP, Madsen JM, Scarbrough PM, Spitz DR. The role of Akt pathway signaling in glucose metabolism and metabolic oxidative stress. In: Spitz DR, Dornfeld KJ, Krishnan K, Gius D (eds). Oxidative stress in cancer biology and therapy. Humana Press (copyright holder, Springer Science+Business Media, LLC); 2012:21-46.
14. Kim S-B, Dent R, Im S-A, et al. Ipatasertib plus paclitaxel versus placebo plus paclitaxel as first-line therapy for metastatic triple-negative breast cancer (LOTUS): a multicentre, randomised, double-blind, placebo-controlled, phase 2 trial. Lancet Oncol. 2017;18(10):1360-1372.
15. Fu Y, Liu S, Yin S, et al. The reverse Warburg effect is likely to be an Achilles’ heel of cancer that can be exploited for cancer therapy. Oncotarget. 2017;8(34):57813-57825.
16. Wilde L, Roche M, Domingo-Vidal M, et al. Metabolic coupling and the reverse Warburg effect in cancer: implications for novel biomarker and anticancer agent development. Semin Oncol. 2017;44(3):198-203.
17. Brooks GA. Cell–cell and intracellular lactate shuttles. Journal Physiol. 2009;587(23):5591-5600.
18. Chiarugi P, Cirri P. Metabolic exchanges within tumor microenvironment. Cancer Lett. 2016;380(1):272-280.
19. Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144(5):646-674.
20. Fujii T, Khawaja MR, DiNardo CD, Atkins JT, Janku F. Targeting isocitrate dehydrogenase (IDH) in cancer. Discov Med. 2016;21(117):373-380.
21. Carlsson SK, Brothers SP, Wahlestedt C. Emerging treatment strategies for glioblastoma multiforme. EMBO Mol Med. 2014;6(11):1359-1370.
22. Lu Y, Kwintkiewicz J, Liu Y, et al. Chemosensitivity of IDH1-mutated gliomas due to an impairment in PARP1-mediated DNA repair. Cancer Res. 2017;77(7):1709-1718.
23. Stein EM, DiNardo CD, Pollyea DA, et al. Enasidenib in mutant IDH2 relapsed or refractory acute myeloid leukemia. Blood. 2017;130(6):722-731.
24. Lowery MA, Abou-Alfa GK, Burris HA, et al. Phase I study of AG-120, an IDH1 mutant enzyme inhibitor: results from the cholangiocarcinoma dose escalation and expansion cohorts. J Clin Oncol. 2017;35(15_suppl):4015-4015.
25. Mellinghoff IK, Touat M, Maher E, et al. ACTR-46. AG-120, a first-in-class mutant IDH1 inhibitor in patients with recurrent or progressive IDH1 mutant glioma: updated results from the phase 1 non-enhancing glioma population. Neuro Oncol. 2017;19(suppl_6):vi10-vi11.
26. Kalinsky K, Harding J, DeMichele A, et al. Phase 1 study of CB-839, a first-in-class oral inhibitor of glutaminase, in combination with paclitaxel in patients with advanced triple negative breast cancer. Paper presented at San Antonio Breast Cancer Symposium; December 5-9, 2017; San Antonio, Texas.
27. Hatoum D, McGowan EM. Recent advances in the use of metformin: can treating diabetes prevent breast cancer? Biomed Res Int. 2015;2015:548436.
28. Leone A, Di Gennaro E, Bruzzese F, Avallone A, Budillon A. New perspective for an old antidiabetic drug: metformin as anticancer agent. Cancer Treat Res. 2014;159:355-376.
1. Warburg O. On respiratory impairment in cancer cells. Science. 1956;124(3215):269-270.
2. Yu L, Chen X, Wang L, Chen S. The sweet trap in tumors: aerobic glycolysis and potential targets for therapy. Oncotarget. 2016;7(25):38908-38926.
3. Warburg O. On the origin of cancer cells. Science. 1956;123(3191):309-314.
4. Chen XS, Li LY, Guan YD, Yang JM, Cheng Y. Anticancer strategies based on the metabolic profile of tumor cells: therapeutic targeting of the Warburg effect. Acta Pharmacol Sin. 2016;37(8):1013-1019.
5. Gupta S, Roy A, Dwarakanath BS. Metabolic cooperation and competition in the tumor microenvironment: implications for therapy. Front Oncol. 2017;7:68.
6. Marchiq I, Pouyssegur J. Hypoxia, cancer metabolism and the therapeutic benefit of targeting lactate/H(+) symporters. J Mol Med (Berl). 2016;94(2):155-171.
7. Altenberg B, Greulich KO. Genes of glycolysis are ubiquitously overexpressed in 24 cancer classes. Genomics. 2004;84(6):1014-1020.
8. Yu L, Chen X, Sun X, Wang L, Chen S. The glycolytic switch in tumors: how many players are involved? J Cancer. 2017;8(17):3430-3440.
9. Zhang W, Zhang SL, Hu X, Tam KY. Targeting tumor metabolism for cancer treatment: is pyruvate dehydrogenase kinases (PDKs) a viable anticancer target? Int J Biol Sci. 2015;11(12):1390-1400.
10. Talekar M, Boreddy SR, Singh A, Amiji M. Tumor aerobic glycolysis: new insights into therapeutic strategies with targeted delivery. Expert Opin Biol Ther. 2014;14(8):1145-1159.
11. Ganapathy-Kanniappan S, Geschwind JF. Tumor glycolysis as a target for cancer therapy: progress and prospects. Mol Cancer. 2013;12:152.
12. Lien EC, Lyssiotis CA, Cantley LC. Metabolic reprogramming by the PI3K-Akt-mTOR pathway in cancer. In: Cramer T, Schmitt CA, eds. Metabolism in Cancer. Cham, Switzerland: Springer International Publishing; 2016:39-72.
13. Simons AL, Orcutt KP, Madsen JM, Scarbrough PM, Spitz DR. The role of Akt pathway signaling in glucose metabolism and metabolic oxidative stress. In: Spitz DR, Dornfeld KJ, Krishnan K, Gius D (eds). Oxidative stress in cancer biology and therapy. Humana Press (copyright holder, Springer Science+Business Media, LLC); 2012:21-46.
14. Kim S-B, Dent R, Im S-A, et al. Ipatasertib plus paclitaxel versus placebo plus paclitaxel as first-line therapy for metastatic triple-negative breast cancer (LOTUS): a multicentre, randomised, double-blind, placebo-controlled, phase 2 trial. Lancet Oncol. 2017;18(10):1360-1372.
15. Fu Y, Liu S, Yin S, et al. The reverse Warburg effect is likely to be an Achilles’ heel of cancer that can be exploited for cancer therapy. Oncotarget. 2017;8(34):57813-57825.
16. Wilde L, Roche M, Domingo-Vidal M, et al. Metabolic coupling and the reverse Warburg effect in cancer: implications for novel biomarker and anticancer agent development. Semin Oncol. 2017;44(3):198-203.
17. Brooks GA. Cell–cell and intracellular lactate shuttles. Journal Physiol. 2009;587(23):5591-5600.
18. Chiarugi P, Cirri P. Metabolic exchanges within tumor microenvironment. Cancer Lett. 2016;380(1):272-280.
19. Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144(5):646-674.
20. Fujii T, Khawaja MR, DiNardo CD, Atkins JT, Janku F. Targeting isocitrate dehydrogenase (IDH) in cancer. Discov Med. 2016;21(117):373-380.
21. Carlsson SK, Brothers SP, Wahlestedt C. Emerging treatment strategies for glioblastoma multiforme. EMBO Mol Med. 2014;6(11):1359-1370.
22. Lu Y, Kwintkiewicz J, Liu Y, et al. Chemosensitivity of IDH1-mutated gliomas due to an impairment in PARP1-mediated DNA repair. Cancer Res. 2017;77(7):1709-1718.
23. Stein EM, DiNardo CD, Pollyea DA, et al. Enasidenib in mutant IDH2 relapsed or refractory acute myeloid leukemia. Blood. 2017;130(6):722-731.
24. Lowery MA, Abou-Alfa GK, Burris HA, et al. Phase I study of AG-120, an IDH1 mutant enzyme inhibitor: results from the cholangiocarcinoma dose escalation and expansion cohorts. J Clin Oncol. 2017;35(15_suppl):4015-4015.
25. Mellinghoff IK, Touat M, Maher E, et al. ACTR-46. AG-120, a first-in-class mutant IDH1 inhibitor in patients with recurrent or progressive IDH1 mutant glioma: updated results from the phase 1 non-enhancing glioma population. Neuro Oncol. 2017;19(suppl_6):vi10-vi11.
26. Kalinsky K, Harding J, DeMichele A, et al. Phase 1 study of CB-839, a first-in-class oral inhibitor of glutaminase, in combination with paclitaxel in patients with advanced triple negative breast cancer. Paper presented at San Antonio Breast Cancer Symposium; December 5-9, 2017; San Antonio, Texas.
27. Hatoum D, McGowan EM. Recent advances in the use of metformin: can treating diabetes prevent breast cancer? Biomed Res Int. 2015;2015:548436.
28. Leone A, Di Gennaro E, Bruzzese F, Avallone A, Budillon A. New perspective for an old antidiabetic drug: metformin as anticancer agent. Cancer Treat Res. 2014;159:355-376.
VIDEO: Novel PARP inhibitor boosts PFS in HER2– breast cancer with BRCA mutations
SAN ANTONIO – In women with advanced HER2-negative breast cancer with germline BRCA mutations, talazoparib, an investigational oral PARP inhibitor, was associated with a near doubling in progression-free survival, compared with single-agent chemotherapy in the phase 3 EMBRACA trial.
After a median follow-up of 11.2 months, median progression-free survival by blinded central review was 8.6 months for patients assigned to receive talazoparib, compared with 5.6 months for patients randomized to receive the physician’s choice of either capecitabine, eribulin, gemcitabine, or vinorelbine.
In this video interview from the San Antonio Breast Cancer Symposium, Jennifer K. Litton, MD, from the University of Texas MD Anderson Cancer Center in Houston discusses the comparative efficacy of the drug relative to standard chemotherapy agents in this population, and the association of the PARP inhibitor with improved patient-reported quality of life outcomes.
The EMBRACA study was funded by Pfizer. Dr. Litton has disclosed research funding with EMD Serono, AstraZeneca, Pfizer, Genentech, and GlaxoSmithKline, and serves on advisory boards for Pfizer and AstraZeneca, all uncompensated.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SAN ANTONIO – In women with advanced HER2-negative breast cancer with germline BRCA mutations, talazoparib, an investigational oral PARP inhibitor, was associated with a near doubling in progression-free survival, compared with single-agent chemotherapy in the phase 3 EMBRACA trial.
After a median follow-up of 11.2 months, median progression-free survival by blinded central review was 8.6 months for patients assigned to receive talazoparib, compared with 5.6 months for patients randomized to receive the physician’s choice of either capecitabine, eribulin, gemcitabine, or vinorelbine.
In this video interview from the San Antonio Breast Cancer Symposium, Jennifer K. Litton, MD, from the University of Texas MD Anderson Cancer Center in Houston discusses the comparative efficacy of the drug relative to standard chemotherapy agents in this population, and the association of the PARP inhibitor with improved patient-reported quality of life outcomes.
The EMBRACA study was funded by Pfizer. Dr. Litton has disclosed research funding with EMD Serono, AstraZeneca, Pfizer, Genentech, and GlaxoSmithKline, and serves on advisory boards for Pfizer and AstraZeneca, all uncompensated.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SAN ANTONIO – In women with advanced HER2-negative breast cancer with germline BRCA mutations, talazoparib, an investigational oral PARP inhibitor, was associated with a near doubling in progression-free survival, compared with single-agent chemotherapy in the phase 3 EMBRACA trial.
After a median follow-up of 11.2 months, median progression-free survival by blinded central review was 8.6 months for patients assigned to receive talazoparib, compared with 5.6 months for patients randomized to receive the physician’s choice of either capecitabine, eribulin, gemcitabine, or vinorelbine.
In this video interview from the San Antonio Breast Cancer Symposium, Jennifer K. Litton, MD, from the University of Texas MD Anderson Cancer Center in Houston discusses the comparative efficacy of the drug relative to standard chemotherapy agents in this population, and the association of the PARP inhibitor with improved patient-reported quality of life outcomes.
The EMBRACA study was funded by Pfizer. Dr. Litton has disclosed research funding with EMD Serono, AstraZeneca, Pfizer, Genentech, and GlaxoSmithKline, and serves on advisory boards for Pfizer and AstraZeneca, all uncompensated.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
REPORTING FROM SABCS 2017
Ovarian suppression during breast cancer chemo helps stave off early menopause, preserve fertility
SAN ANTONIO – Premenopausal women with early breast cancer should be offered temporary ovarian suppression during chemotherapy if they wish to remain fertile or avoid early menopause, suggests a meta-analysis of five randomized controlled trials among 873 women reported at the San Antonio Breast Cancer Symposium.
“Oocyte and embryo cryopreservation are standard strategies for fertility preservation in these patients, meaning increasing the chance of pregnancy after the end of treatment,” said lead author Matteo Lambertini, MD, medical oncologist and ESMO fellow at the Institut Jules Bordet in Brussels. “However, they do not prevent the risk of developing chemotherapy-induced premature ovarian insufficiency, and so patients are still at risk of developing early menopause.”
Data from individual trials of ovarian suppression have been mixed, and its use remains controversial, he further noted. As a result, guidelines from ASCO and ESMO for fertility preservation in cancer patients still consider ovarian suppression to be investigational.
Results of the new meta-analysis, reported in a press briefing and session, showed that, compared with control peers, premenopausal women given a gonadotropin-releasing hormone analog (GnRHa) to suppress ovarian function during breast cancer chemotherapy had a more than one-half reduction in odds of premature ovarian insufficiency and were almost twice as likely to become pregnant after completing their treatment.
“We believe that this strategy should now be considered a standard option to reduce the likelihood of chemotherapy-induced premature ovarian insufficiency and potentially improve future fertility in premenopausal early breast cancer patients who undergo adjuvant or neoadjuvant chemotherapy,” Dr. Lambertini maintained. The analysis and trial participants “remind us that there is a life after cancer and to cure the disease should not be considered enough any more.”
These new data are sufficient to put the controversy to rest, for several reasons, he contended. “First, this is an individual-patient meta-analysis of the five major randomized controlled trials in this setting, so it’s kind of the highest level of evidence that we can reach. Second, it’s very unlikely that we will have new randomized, controlled trials in this setting, and I would say also that, based on the results, it would probably be unethical to run randomized, controlled trials in this setting.”
Press briefing moderator Carlos L. Arteaga, MD, director of the Harold C. Simmons Comprehensive Cancer Center at UT Southwestern Medical Center, Dallas, wondered how trials were selected for inclusion in the meta-analysis.
Thirteen trials have been conducted on ovarian suppression in premenopausal patients with breast cancer, and the investigators included the five for which they were able to obtain patient-level data, a subset that contained both positive and negative trials, Dr. Lambertini replied. But findings would likely be the same had all trials been included, given that a 2015 analysis using trial-level data from the 12 trials completed at that time showed very similar results (Ann Oncol. 2015 Dec;26:2408-19).
“Societally, this is a hugely significant issue, but the difference you are showing I have to admit is rather modest,” commented Dr. Arteaga, who is also SABCS codirector and AACR past president. “So what kind of conversation do you have with the patient? Who are the ones who would be the best candidates for this approach?”
“The main message is that giving a GnRHa does not avoid the risk of early menopause in all patients, but still, it decreases significantly the number of patients who have this side effect,” Dr. Lambertini replied.
Two groups are optimal candidates for ovarian suppression, he proposed. “First, the patients who are concerned about developing early menopause and its related side effects, who are not interested per se in having a baby after the end of treatment, but may be preserving ovarian function. [Second], for patients interested in having a baby, so interested in fertility preservation, this strategy can be used after cryopreservation procedures or in patients who have no access, for different reasons, to cryopreservation strategies.”
Another point of view
In the session, attendee Kutluk Oktay, MD, PhD, professor of obstetrics & gynecology and reproductive sciences at Yale University, New Haven, and cochair of ASCO’s guideline committee on fertility preservation said, “I cannot agree with your conclusions based on what you presented to us.”
In particular, he took issue with the exclusion of additional trials in breast cancer as well as trials among patients with other types of cancers. “I’m wondering what the rationale is to limit this to breast cancer because chemotherapy is chemotherapy and ovary is ovary, so underlying disease should not matter. By limiting it to breast cancer, you are leaving out three important studies, all in hematological cancer, with better designs … three negative studies,” he commented.
From a clinical point of view, patients with lymphoma and patients with breast cancer differ greatly, Dr. Lambertini countered: The former are about 20 years younger, on average, and often receive less-granulotoxic chemotherapy. “For these reasons, I don’t believe that mixing these two populations would have been [appropriate] for analysis,” he said. “From a methodological point of view, the studies you have mentioned include overall [fewer] than 150 patients, so it’s a very small proportion in comparison to the data we have in breast cancer.”
Study details
For their meta-analysis, Dr. Lambertini and coinvestigators pooled individual patient data from five trials (PROMISE-GIM6, POEMS/SWOG S0230, Anglo-Celtic Group OPTION, GBG-37 ZORO, and a trial led by the Moffitt Cancer Center) that randomized premenopausal women with early breast cancer to adjuvant or neoadjuvant chemotherapy either with or without concurrent GnRHa therapy.
Two of the trials restricted enrollment to women with estrogen receptor (ER)-negative disease. The GnRHa agents used were triptorelin (Trelstar, Triptodur)and goserelin(Zoladex).
Main results showed that the rate of premature ovarian insufficiency, defined differently across trials, was 14.1% among women given a GnRHa and 30.9% among control women (adjusted odds ratio, 0.38; P less than .001), Dr. Lambertini reported. Findings were similar in subgroups stratified by age, ER status, and type and duration of chemotherapy.
The rate of amenorrhea, used as a standardized definition of premature ovarian insufficiency, was similar in the GnRHa and control groups at 1 year (36.8% and 40.4%) but sharply lower in the former at 2 years (18.2% vs. 30.0%; adjusted odds ratio, 0.51; P = .009).
Overall, 10.3% of women in the GnRHa group and 5.5% in the control group had at least one pregnancy after completing their breast cancer treatment (incidence rate ratio, 1.83; P = .030). “All of the randomized trials except for the POEMS study actually did not have fertility outcomes as a preplanned endpoint, and so the patients’ wish to have a pregnancy was not collected,” he noted; therefore, it was not possible to calculate pregnancy rates in the subset who actually wanted to conceive.
All pregnancies occurred among women aged 40 years or younger, and 86% occurred among women who had had ER-negative disease, likely reflecting use of adjuvant endocrine therapy in patients with ER-positive disease, he said. Of the 57 total pregnancies, 50 resulted in live births, and none of the infants had malformations; the other pregnancies ended in spontaneous or induced abortion.
With a median follow-up of 5 years, the groups did not differ significantly on rates of disease-free survival and overall survival, suggesting that ovarian suppression was safe, according to Dr. Lambertini. Findings were similar when patients were stratified by ER status.
“What I think researchers should do in the next year is to better understand how this strategy [of ovarian suppression] works because this is probably the main controversy right now, because it’s still not very clear how this strategy actually works,” he concluded.
SOURCE: Lambertini M et al., SABCS 2017 Abstract GS4-01.
SAN ANTONIO – Premenopausal women with early breast cancer should be offered temporary ovarian suppression during chemotherapy if they wish to remain fertile or avoid early menopause, suggests a meta-analysis of five randomized controlled trials among 873 women reported at the San Antonio Breast Cancer Symposium.
“Oocyte and embryo cryopreservation are standard strategies for fertility preservation in these patients, meaning increasing the chance of pregnancy after the end of treatment,” said lead author Matteo Lambertini, MD, medical oncologist and ESMO fellow at the Institut Jules Bordet in Brussels. “However, they do not prevent the risk of developing chemotherapy-induced premature ovarian insufficiency, and so patients are still at risk of developing early menopause.”
Data from individual trials of ovarian suppression have been mixed, and its use remains controversial, he further noted. As a result, guidelines from ASCO and ESMO for fertility preservation in cancer patients still consider ovarian suppression to be investigational.
Results of the new meta-analysis, reported in a press briefing and session, showed that, compared with control peers, premenopausal women given a gonadotropin-releasing hormone analog (GnRHa) to suppress ovarian function during breast cancer chemotherapy had a more than one-half reduction in odds of premature ovarian insufficiency and were almost twice as likely to become pregnant after completing their treatment.
“We believe that this strategy should now be considered a standard option to reduce the likelihood of chemotherapy-induced premature ovarian insufficiency and potentially improve future fertility in premenopausal early breast cancer patients who undergo adjuvant or neoadjuvant chemotherapy,” Dr. Lambertini maintained. The analysis and trial participants “remind us that there is a life after cancer and to cure the disease should not be considered enough any more.”
These new data are sufficient to put the controversy to rest, for several reasons, he contended. “First, this is an individual-patient meta-analysis of the five major randomized controlled trials in this setting, so it’s kind of the highest level of evidence that we can reach. Second, it’s very unlikely that we will have new randomized, controlled trials in this setting, and I would say also that, based on the results, it would probably be unethical to run randomized, controlled trials in this setting.”
Press briefing moderator Carlos L. Arteaga, MD, director of the Harold C. Simmons Comprehensive Cancer Center at UT Southwestern Medical Center, Dallas, wondered how trials were selected for inclusion in the meta-analysis.
Thirteen trials have been conducted on ovarian suppression in premenopausal patients with breast cancer, and the investigators included the five for which they were able to obtain patient-level data, a subset that contained both positive and negative trials, Dr. Lambertini replied. But findings would likely be the same had all trials been included, given that a 2015 analysis using trial-level data from the 12 trials completed at that time showed very similar results (Ann Oncol. 2015 Dec;26:2408-19).
“Societally, this is a hugely significant issue, but the difference you are showing I have to admit is rather modest,” commented Dr. Arteaga, who is also SABCS codirector and AACR past president. “So what kind of conversation do you have with the patient? Who are the ones who would be the best candidates for this approach?”
“The main message is that giving a GnRHa does not avoid the risk of early menopause in all patients, but still, it decreases significantly the number of patients who have this side effect,” Dr. Lambertini replied.
Two groups are optimal candidates for ovarian suppression, he proposed. “First, the patients who are concerned about developing early menopause and its related side effects, who are not interested per se in having a baby after the end of treatment, but may be preserving ovarian function. [Second], for patients interested in having a baby, so interested in fertility preservation, this strategy can be used after cryopreservation procedures or in patients who have no access, for different reasons, to cryopreservation strategies.”
Another point of view
In the session, attendee Kutluk Oktay, MD, PhD, professor of obstetrics & gynecology and reproductive sciences at Yale University, New Haven, and cochair of ASCO’s guideline committee on fertility preservation said, “I cannot agree with your conclusions based on what you presented to us.”
In particular, he took issue with the exclusion of additional trials in breast cancer as well as trials among patients with other types of cancers. “I’m wondering what the rationale is to limit this to breast cancer because chemotherapy is chemotherapy and ovary is ovary, so underlying disease should not matter. By limiting it to breast cancer, you are leaving out three important studies, all in hematological cancer, with better designs … three negative studies,” he commented.
From a clinical point of view, patients with lymphoma and patients with breast cancer differ greatly, Dr. Lambertini countered: The former are about 20 years younger, on average, and often receive less-granulotoxic chemotherapy. “For these reasons, I don’t believe that mixing these two populations would have been [appropriate] for analysis,” he said. “From a methodological point of view, the studies you have mentioned include overall [fewer] than 150 patients, so it’s a very small proportion in comparison to the data we have in breast cancer.”
Study details
For their meta-analysis, Dr. Lambertini and coinvestigators pooled individual patient data from five trials (PROMISE-GIM6, POEMS/SWOG S0230, Anglo-Celtic Group OPTION, GBG-37 ZORO, and a trial led by the Moffitt Cancer Center) that randomized premenopausal women with early breast cancer to adjuvant or neoadjuvant chemotherapy either with or without concurrent GnRHa therapy.
Two of the trials restricted enrollment to women with estrogen receptor (ER)-negative disease. The GnRHa agents used were triptorelin (Trelstar, Triptodur)and goserelin(Zoladex).
Main results showed that the rate of premature ovarian insufficiency, defined differently across trials, was 14.1% among women given a GnRHa and 30.9% among control women (adjusted odds ratio, 0.38; P less than .001), Dr. Lambertini reported. Findings were similar in subgroups stratified by age, ER status, and type and duration of chemotherapy.
The rate of amenorrhea, used as a standardized definition of premature ovarian insufficiency, was similar in the GnRHa and control groups at 1 year (36.8% and 40.4%) but sharply lower in the former at 2 years (18.2% vs. 30.0%; adjusted odds ratio, 0.51; P = .009).
Overall, 10.3% of women in the GnRHa group and 5.5% in the control group had at least one pregnancy after completing their breast cancer treatment (incidence rate ratio, 1.83; P = .030). “All of the randomized trials except for the POEMS study actually did not have fertility outcomes as a preplanned endpoint, and so the patients’ wish to have a pregnancy was not collected,” he noted; therefore, it was not possible to calculate pregnancy rates in the subset who actually wanted to conceive.
All pregnancies occurred among women aged 40 years or younger, and 86% occurred among women who had had ER-negative disease, likely reflecting use of adjuvant endocrine therapy in patients with ER-positive disease, he said. Of the 57 total pregnancies, 50 resulted in live births, and none of the infants had malformations; the other pregnancies ended in spontaneous or induced abortion.
With a median follow-up of 5 years, the groups did not differ significantly on rates of disease-free survival and overall survival, suggesting that ovarian suppression was safe, according to Dr. Lambertini. Findings were similar when patients were stratified by ER status.
“What I think researchers should do in the next year is to better understand how this strategy [of ovarian suppression] works because this is probably the main controversy right now, because it’s still not very clear how this strategy actually works,” he concluded.
SOURCE: Lambertini M et al., SABCS 2017 Abstract GS4-01.
SAN ANTONIO – Premenopausal women with early breast cancer should be offered temporary ovarian suppression during chemotherapy if they wish to remain fertile or avoid early menopause, suggests a meta-analysis of five randomized controlled trials among 873 women reported at the San Antonio Breast Cancer Symposium.
“Oocyte and embryo cryopreservation are standard strategies for fertility preservation in these patients, meaning increasing the chance of pregnancy after the end of treatment,” said lead author Matteo Lambertini, MD, medical oncologist and ESMO fellow at the Institut Jules Bordet in Brussels. “However, they do not prevent the risk of developing chemotherapy-induced premature ovarian insufficiency, and so patients are still at risk of developing early menopause.”
Data from individual trials of ovarian suppression have been mixed, and its use remains controversial, he further noted. As a result, guidelines from ASCO and ESMO for fertility preservation in cancer patients still consider ovarian suppression to be investigational.
Results of the new meta-analysis, reported in a press briefing and session, showed that, compared with control peers, premenopausal women given a gonadotropin-releasing hormone analog (GnRHa) to suppress ovarian function during breast cancer chemotherapy had a more than one-half reduction in odds of premature ovarian insufficiency and were almost twice as likely to become pregnant after completing their treatment.
“We believe that this strategy should now be considered a standard option to reduce the likelihood of chemotherapy-induced premature ovarian insufficiency and potentially improve future fertility in premenopausal early breast cancer patients who undergo adjuvant or neoadjuvant chemotherapy,” Dr. Lambertini maintained. The analysis and trial participants “remind us that there is a life after cancer and to cure the disease should not be considered enough any more.”
These new data are sufficient to put the controversy to rest, for several reasons, he contended. “First, this is an individual-patient meta-analysis of the five major randomized controlled trials in this setting, so it’s kind of the highest level of evidence that we can reach. Second, it’s very unlikely that we will have new randomized, controlled trials in this setting, and I would say also that, based on the results, it would probably be unethical to run randomized, controlled trials in this setting.”
Press briefing moderator Carlos L. Arteaga, MD, director of the Harold C. Simmons Comprehensive Cancer Center at UT Southwestern Medical Center, Dallas, wondered how trials were selected for inclusion in the meta-analysis.
Thirteen trials have been conducted on ovarian suppression in premenopausal patients with breast cancer, and the investigators included the five for which they were able to obtain patient-level data, a subset that contained both positive and negative trials, Dr. Lambertini replied. But findings would likely be the same had all trials been included, given that a 2015 analysis using trial-level data from the 12 trials completed at that time showed very similar results (Ann Oncol. 2015 Dec;26:2408-19).
“Societally, this is a hugely significant issue, but the difference you are showing I have to admit is rather modest,” commented Dr. Arteaga, who is also SABCS codirector and AACR past president. “So what kind of conversation do you have with the patient? Who are the ones who would be the best candidates for this approach?”
“The main message is that giving a GnRHa does not avoid the risk of early menopause in all patients, but still, it decreases significantly the number of patients who have this side effect,” Dr. Lambertini replied.
Two groups are optimal candidates for ovarian suppression, he proposed. “First, the patients who are concerned about developing early menopause and its related side effects, who are not interested per se in having a baby after the end of treatment, but may be preserving ovarian function. [Second], for patients interested in having a baby, so interested in fertility preservation, this strategy can be used after cryopreservation procedures or in patients who have no access, for different reasons, to cryopreservation strategies.”
Another point of view
In the session, attendee Kutluk Oktay, MD, PhD, professor of obstetrics & gynecology and reproductive sciences at Yale University, New Haven, and cochair of ASCO’s guideline committee on fertility preservation said, “I cannot agree with your conclusions based on what you presented to us.”
In particular, he took issue with the exclusion of additional trials in breast cancer as well as trials among patients with other types of cancers. “I’m wondering what the rationale is to limit this to breast cancer because chemotherapy is chemotherapy and ovary is ovary, so underlying disease should not matter. By limiting it to breast cancer, you are leaving out three important studies, all in hematological cancer, with better designs … three negative studies,” he commented.
From a clinical point of view, patients with lymphoma and patients with breast cancer differ greatly, Dr. Lambertini countered: The former are about 20 years younger, on average, and often receive less-granulotoxic chemotherapy. “For these reasons, I don’t believe that mixing these two populations would have been [appropriate] for analysis,” he said. “From a methodological point of view, the studies you have mentioned include overall [fewer] than 150 patients, so it’s a very small proportion in comparison to the data we have in breast cancer.”
Study details
For their meta-analysis, Dr. Lambertini and coinvestigators pooled individual patient data from five trials (PROMISE-GIM6, POEMS/SWOG S0230, Anglo-Celtic Group OPTION, GBG-37 ZORO, and a trial led by the Moffitt Cancer Center) that randomized premenopausal women with early breast cancer to adjuvant or neoadjuvant chemotherapy either with or without concurrent GnRHa therapy.
Two of the trials restricted enrollment to women with estrogen receptor (ER)-negative disease. The GnRHa agents used were triptorelin (Trelstar, Triptodur)and goserelin(Zoladex).
Main results showed that the rate of premature ovarian insufficiency, defined differently across trials, was 14.1% among women given a GnRHa and 30.9% among control women (adjusted odds ratio, 0.38; P less than .001), Dr. Lambertini reported. Findings were similar in subgroups stratified by age, ER status, and type and duration of chemotherapy.
The rate of amenorrhea, used as a standardized definition of premature ovarian insufficiency, was similar in the GnRHa and control groups at 1 year (36.8% and 40.4%) but sharply lower in the former at 2 years (18.2% vs. 30.0%; adjusted odds ratio, 0.51; P = .009).
Overall, 10.3% of women in the GnRHa group and 5.5% in the control group had at least one pregnancy after completing their breast cancer treatment (incidence rate ratio, 1.83; P = .030). “All of the randomized trials except for the POEMS study actually did not have fertility outcomes as a preplanned endpoint, and so the patients’ wish to have a pregnancy was not collected,” he noted; therefore, it was not possible to calculate pregnancy rates in the subset who actually wanted to conceive.
All pregnancies occurred among women aged 40 years or younger, and 86% occurred among women who had had ER-negative disease, likely reflecting use of adjuvant endocrine therapy in patients with ER-positive disease, he said. Of the 57 total pregnancies, 50 resulted in live births, and none of the infants had malformations; the other pregnancies ended in spontaneous or induced abortion.
With a median follow-up of 5 years, the groups did not differ significantly on rates of disease-free survival and overall survival, suggesting that ovarian suppression was safe, according to Dr. Lambertini. Findings were similar when patients were stratified by ER status.
“What I think researchers should do in the next year is to better understand how this strategy [of ovarian suppression] works because this is probably the main controversy right now, because it’s still not very clear how this strategy actually works,” he concluded.
SOURCE: Lambertini M et al., SABCS 2017 Abstract GS4-01.
REPORTING FROM SABCS 2017
Key clinical point:
Major finding: Compared with controls, women given a GnRHa during chemotherapy were less likely to develop premature ovarian insufficiency (adjusted OR, 0.38; P less than .001) and more likely to become pregnant after treatment (IRR, 1.83; P = .030).
Data source: A meta-analysis of individual patient data from five trials among 873 premenopausal women with early breast cancer.
Disclosures: Dr. Lambertini disclosed that he had no relevant conflicts of interest.
Source: Lambertini M et al., SABCS 2017 Abstract GS4-01.
VIDEO: Weight loss cut risk of breast cancer
SAN ANTONIO – A new analysis from the Women’s Health Initiative Observational Study gives postmenopausal women yet another reason to mind their weight. Results showed that women losing at least 5% of their body weight had a significant 12% reduction in adjusted breast cancer risk relative to peers who maintained a stable weight, reported lead author Rowan Chlebowski, MD, PhD, research professor in the department of medical oncology and therapeutics research at City of Hope in Duarte, Calif. Findings were much the same regardless of whether women were of normal weight, overweight, or obese at baseline. Dr. Chlebowski discussed the implications for patient counseling and insurance coverage of weight loss interventions, as well as planned research that will assess the physiologic mechanisms at play in a video interview.
SAN ANTONIO – A new analysis from the Women’s Health Initiative Observational Study gives postmenopausal women yet another reason to mind their weight. Results showed that women losing at least 5% of their body weight had a significant 12% reduction in adjusted breast cancer risk relative to peers who maintained a stable weight, reported lead author Rowan Chlebowski, MD, PhD, research professor in the department of medical oncology and therapeutics research at City of Hope in Duarte, Calif. Findings were much the same regardless of whether women were of normal weight, overweight, or obese at baseline. Dr. Chlebowski discussed the implications for patient counseling and insurance coverage of weight loss interventions, as well as planned research that will assess the physiologic mechanisms at play in a video interview.
SAN ANTONIO – A new analysis from the Women’s Health Initiative Observational Study gives postmenopausal women yet another reason to mind their weight. Results showed that women losing at least 5% of their body weight had a significant 12% reduction in adjusted breast cancer risk relative to peers who maintained a stable weight, reported lead author Rowan Chlebowski, MD, PhD, research professor in the department of medical oncology and therapeutics research at City of Hope in Duarte, Calif. Findings were much the same regardless of whether women were of normal weight, overweight, or obese at baseline. Dr. Chlebowski discussed the implications for patient counseling and insurance coverage of weight loss interventions, as well as planned research that will assess the physiologic mechanisms at play in a video interview.
REPORTING FROM SABCS 2017
Adjuvant trastuzumab for 1 year remains standard of care for early HER2+ breast cancer
SAN ANTONIO – The phase 3 SOLD trial failed to show a 9-week course of trastuzumab as noninferior to the standard 12-month course when combined with chemotherapy for women with early-stage HER2-positive breast cancer.
Disease-free survival (DFS) with a 9-week course of adjuvant trastuzumab (Herceptin)did not pass muster as noninferior to the standard 12 months, reported Dr. Heikki Joensuu, professor in the department of oncology at the Helsinki University Hospital and University of Helsinki.
“The non-inferiority of 9 week administration of trastuzumab plus docetaxel could not be demonstrated in terms of disease free survival, as compared to chemotherapy and one year duration of adjuvant trastuzumab.”
Based on these findings, he noted that chemotherapy plus one year of anti-HER2 therapy should remain the standard of care.
However, the dose of docetaxel administered along with trastuzumab could influence survival outcomes. Patients who received a higher dose of docetaxel in the 9 week cohort had similar DFS as those who received 12 months of trastuzumab.
Dr. Joensuu explained that the regimen of giving trastuzumab for one year is an arbitrary one and is not based on research data. In the four large randomized trials that established the current standard treatment for this population of breast cancer patients, trastuzumab was administered for one year and that eventually became the standard of care, he said at the San Antonio Breast Cancer Symposium.
But in two randomized trials that had relatively limited statistical power, a statistically significant difference in DFS or overall survival was not observed between patients who received only nine weeks of trastuzumab compared with those who received the one year regimen.
In addition, the one year treatment course is costly and has been associated with cardiac-related adverse events, although it is relatively uncommon (less than 3% of patients).
The phase 3 SOLD (Synergism or Long Duration) clinical trial was conducted by Dr. Joensuu and his colleagues to compare DFS between women treated with 9 weeks of concomitant trastuzumab plus docetaxel followed by 5-FU, epirubicin, and cyclophosphamide (FEC) to that of women treated with the same regimen followed by single-agent trastuzumab for a one year duration.
The cohort included 2,176 patients with early-stage HER2-positive breast cancer who were randomized to either the 9-week or the 12-month trastuzumab arm. In addition, cardiac function among women enrolled in the trial had to be normal, Dr. Joensuu said, as determined by left ventricular ejection fraction of 50% or greater.
Patients in both groups received three cycles of docetaxel (80 mg/m2 or 100 mg/m2) and trastuzumab three times a week, followed by three cycles of FEC. The dose of docetaxel was determined by the individual center. Patients in the 12-month arm continued to receive trastuzumab every 3 weeks for 14 cycles and in both groups, those with estrogen receptor–positive cancer received appropriate endocrine treatment per guidelines.
“SOLD was initially designed as a superiority trial,” he said, “But, we redesigned the study and the size during the study because we noticed that the assumptions we made for 5-year disease-free survival were too low.”
“We also realized that the shorter arm cannot be better than the longer arm because less drug is given,” said Dr. Joensuu. “So we changed it from a superiority trial to a noninferiority design, where the shorter arm would be at least equal to the longer arm.”
When looking at results, the DFS favored the longer arm: 90.5% in the 12-month arm vs. 88% in the 9-week arm (hazard ratio, 1.39; 90% confidence interval, 1.12-1.72). However, there were no substantial differences in secondary endpoints. Five-year distant DFS was 93.2% in the 9-week arm vs. 94.2% in the 12-month arm (HR, 1.24; 90% CI, 0.93-1.65), and similarly, 5-year overall survival was 94.7% vs. 95.9% (HR, 1.39; 90% CI, 0.98-1.89).
There was an interaction between the dose of docetaxel given concomitantly with trastuzumab. For patients who received 100 mg/m2 docetaxel, DFS was similar in both groups: 92.2% (long arm) vs. 87.8% (short arm) (HR 0.71, 90% CI 0,44-1.14). But among those who received 80 mg/m2 docetaxel, DFS was superior in the group who received 12 months of trastuzumab, compared with the 9-week group (91.3% vs. 86.8%; HR, 1.66; 90% CI, 1.30-2.11).
Less cardiac toxicity was observed in the 9-week group, for any protocol-defined cardiac adverse event (n = 22 [2%] vs. n = 42 [3.9%]; P = .012) and for heart failure (n = 21 [1.9%] vs. n = 36 [3.3%]; P = .046).
“Docetaxel dosing with trastuzumab requires further study,” Dr. Joensuu said.
SOURCE: Joensuu H et al., Abstract GS3-04
SAN ANTONIO – The phase 3 SOLD trial failed to show a 9-week course of trastuzumab as noninferior to the standard 12-month course when combined with chemotherapy for women with early-stage HER2-positive breast cancer.
Disease-free survival (DFS) with a 9-week course of adjuvant trastuzumab (Herceptin)did not pass muster as noninferior to the standard 12 months, reported Dr. Heikki Joensuu, professor in the department of oncology at the Helsinki University Hospital and University of Helsinki.
“The non-inferiority of 9 week administration of trastuzumab plus docetaxel could not be demonstrated in terms of disease free survival, as compared to chemotherapy and one year duration of adjuvant trastuzumab.”
Based on these findings, he noted that chemotherapy plus one year of anti-HER2 therapy should remain the standard of care.
However, the dose of docetaxel administered along with trastuzumab could influence survival outcomes. Patients who received a higher dose of docetaxel in the 9 week cohort had similar DFS as those who received 12 months of trastuzumab.
Dr. Joensuu explained that the regimen of giving trastuzumab for one year is an arbitrary one and is not based on research data. In the four large randomized trials that established the current standard treatment for this population of breast cancer patients, trastuzumab was administered for one year and that eventually became the standard of care, he said at the San Antonio Breast Cancer Symposium.
But in two randomized trials that had relatively limited statistical power, a statistically significant difference in DFS or overall survival was not observed between patients who received only nine weeks of trastuzumab compared with those who received the one year regimen.
In addition, the one year treatment course is costly and has been associated with cardiac-related adverse events, although it is relatively uncommon (less than 3% of patients).
The phase 3 SOLD (Synergism or Long Duration) clinical trial was conducted by Dr. Joensuu and his colleagues to compare DFS between women treated with 9 weeks of concomitant trastuzumab plus docetaxel followed by 5-FU, epirubicin, and cyclophosphamide (FEC) to that of women treated with the same regimen followed by single-agent trastuzumab for a one year duration.
The cohort included 2,176 patients with early-stage HER2-positive breast cancer who were randomized to either the 9-week or the 12-month trastuzumab arm. In addition, cardiac function among women enrolled in the trial had to be normal, Dr. Joensuu said, as determined by left ventricular ejection fraction of 50% or greater.
Patients in both groups received three cycles of docetaxel (80 mg/m2 or 100 mg/m2) and trastuzumab three times a week, followed by three cycles of FEC. The dose of docetaxel was determined by the individual center. Patients in the 12-month arm continued to receive trastuzumab every 3 weeks for 14 cycles and in both groups, those with estrogen receptor–positive cancer received appropriate endocrine treatment per guidelines.
“SOLD was initially designed as a superiority trial,” he said, “But, we redesigned the study and the size during the study because we noticed that the assumptions we made for 5-year disease-free survival were too low.”
“We also realized that the shorter arm cannot be better than the longer arm because less drug is given,” said Dr. Joensuu. “So we changed it from a superiority trial to a noninferiority design, where the shorter arm would be at least equal to the longer arm.”
When looking at results, the DFS favored the longer arm: 90.5% in the 12-month arm vs. 88% in the 9-week arm (hazard ratio, 1.39; 90% confidence interval, 1.12-1.72). However, there were no substantial differences in secondary endpoints. Five-year distant DFS was 93.2% in the 9-week arm vs. 94.2% in the 12-month arm (HR, 1.24; 90% CI, 0.93-1.65), and similarly, 5-year overall survival was 94.7% vs. 95.9% (HR, 1.39; 90% CI, 0.98-1.89).
There was an interaction between the dose of docetaxel given concomitantly with trastuzumab. For patients who received 100 mg/m2 docetaxel, DFS was similar in both groups: 92.2% (long arm) vs. 87.8% (short arm) (HR 0.71, 90% CI 0,44-1.14). But among those who received 80 mg/m2 docetaxel, DFS was superior in the group who received 12 months of trastuzumab, compared with the 9-week group (91.3% vs. 86.8%; HR, 1.66; 90% CI, 1.30-2.11).
Less cardiac toxicity was observed in the 9-week group, for any protocol-defined cardiac adverse event (n = 22 [2%] vs. n = 42 [3.9%]; P = .012) and for heart failure (n = 21 [1.9%] vs. n = 36 [3.3%]; P = .046).
“Docetaxel dosing with trastuzumab requires further study,” Dr. Joensuu said.
SOURCE: Joensuu H et al., Abstract GS3-04
SAN ANTONIO – The phase 3 SOLD trial failed to show a 9-week course of trastuzumab as noninferior to the standard 12-month course when combined with chemotherapy for women with early-stage HER2-positive breast cancer.
Disease-free survival (DFS) with a 9-week course of adjuvant trastuzumab (Herceptin)did not pass muster as noninferior to the standard 12 months, reported Dr. Heikki Joensuu, professor in the department of oncology at the Helsinki University Hospital and University of Helsinki.
“The non-inferiority of 9 week administration of trastuzumab plus docetaxel could not be demonstrated in terms of disease free survival, as compared to chemotherapy and one year duration of adjuvant trastuzumab.”
Based on these findings, he noted that chemotherapy plus one year of anti-HER2 therapy should remain the standard of care.
However, the dose of docetaxel administered along with trastuzumab could influence survival outcomes. Patients who received a higher dose of docetaxel in the 9 week cohort had similar DFS as those who received 12 months of trastuzumab.
Dr. Joensuu explained that the regimen of giving trastuzumab for one year is an arbitrary one and is not based on research data. In the four large randomized trials that established the current standard treatment for this population of breast cancer patients, trastuzumab was administered for one year and that eventually became the standard of care, he said at the San Antonio Breast Cancer Symposium.
But in two randomized trials that had relatively limited statistical power, a statistically significant difference in DFS or overall survival was not observed between patients who received only nine weeks of trastuzumab compared with those who received the one year regimen.
In addition, the one year treatment course is costly and has been associated with cardiac-related adverse events, although it is relatively uncommon (less than 3% of patients).
The phase 3 SOLD (Synergism or Long Duration) clinical trial was conducted by Dr. Joensuu and his colleagues to compare DFS between women treated with 9 weeks of concomitant trastuzumab plus docetaxel followed by 5-FU, epirubicin, and cyclophosphamide (FEC) to that of women treated with the same regimen followed by single-agent trastuzumab for a one year duration.
The cohort included 2,176 patients with early-stage HER2-positive breast cancer who were randomized to either the 9-week or the 12-month trastuzumab arm. In addition, cardiac function among women enrolled in the trial had to be normal, Dr. Joensuu said, as determined by left ventricular ejection fraction of 50% or greater.
Patients in both groups received three cycles of docetaxel (80 mg/m2 or 100 mg/m2) and trastuzumab three times a week, followed by three cycles of FEC. The dose of docetaxel was determined by the individual center. Patients in the 12-month arm continued to receive trastuzumab every 3 weeks for 14 cycles and in both groups, those with estrogen receptor–positive cancer received appropriate endocrine treatment per guidelines.
“SOLD was initially designed as a superiority trial,” he said, “But, we redesigned the study and the size during the study because we noticed that the assumptions we made for 5-year disease-free survival were too low.”
“We also realized that the shorter arm cannot be better than the longer arm because less drug is given,” said Dr. Joensuu. “So we changed it from a superiority trial to a noninferiority design, where the shorter arm would be at least equal to the longer arm.”
When looking at results, the DFS favored the longer arm: 90.5% in the 12-month arm vs. 88% in the 9-week arm (hazard ratio, 1.39; 90% confidence interval, 1.12-1.72). However, there were no substantial differences in secondary endpoints. Five-year distant DFS was 93.2% in the 9-week arm vs. 94.2% in the 12-month arm (HR, 1.24; 90% CI, 0.93-1.65), and similarly, 5-year overall survival was 94.7% vs. 95.9% (HR, 1.39; 90% CI, 0.98-1.89).
There was an interaction between the dose of docetaxel given concomitantly with trastuzumab. For patients who received 100 mg/m2 docetaxel, DFS was similar in both groups: 92.2% (long arm) vs. 87.8% (short arm) (HR 0.71, 90% CI 0,44-1.14). But among those who received 80 mg/m2 docetaxel, DFS was superior in the group who received 12 months of trastuzumab, compared with the 9-week group (91.3% vs. 86.8%; HR, 1.66; 90% CI, 1.30-2.11).
Less cardiac toxicity was observed in the 9-week group, for any protocol-defined cardiac adverse event (n = 22 [2%] vs. n = 42 [3.9%]; P = .012) and for heart failure (n = 21 [1.9%] vs. n = 36 [3.3%]; P = .046).
“Docetaxel dosing with trastuzumab requires further study,” Dr. Joensuu said.
SOURCE: Joensuu H et al., Abstract GS3-04
REPORTING FROM SABCS 2017
Key clinical point: Disease-free survival with a 9-week course of adjuvant trastuzumab did not pass muster as noninferior to the standard 12 months.
Major finding: DFS favored the longer arm: 90.5% in the 12 month arm vs. 88% in the 9-week arm (HR, 1.39; 90% CI 1.12-1.72), but no substantial differences were observed in secondary endpoints.
Data source: Randomized phase 3 trial that included 2,176 patients with early-stage HER2-positive breast cancer
Disclosures:. The study was funded by Pharmac, Sanofi, Novartis, the Academy of Finland, the Cancer Society of Finland, Helsinki University Hospital research funds, Sigrid Juselius Foundation, and Jane and Aatos Erkko Foundation. Dr. Joensuu is a scientific adviser for Neutron Therapeutics, has received consultation fees from Orion Pharma, and has stock ownership interest in Orion, Faron Pharmaceuticals, and Sartar Therapeutics.
Source: Joensuu H et al., GS3-04
2 = 5 for additional AI therapy for postmenopausal HR+ breast cancer
SAN ANTONIO – Five years of additional therapy with anastrozole (Arimidex) was no more effective than 2 additional years following the standard 5 years of initial endocrine therapy in postmenopausal women with hormone receptor–positive (HR+) breast cancer, Austrian investigators reported.
In fact, the only thing that the additional years of the aromatase inhibitor (AI) anastrozole seemed to add was increased risk for fracture, said Michael Gnant, MD, from the Medical University of Vienna, on behalf of colleagues in the ABCSG-16 trial.
Previous trials have convincingly demonstrated the benefit of giving patients an AI for 5 years after 5 years of tamoxifen, but the optimal duration of extended adjuvant AI therapy is not known, Dr. Gnant said.
The ABCSG trialists recruited 3,484 postmenopausal women from with HR+, stage T1-3, node-negative or -positive, nonmetastatic breast cancer who had completed 4-6 years of endocrine therapy with either tamoxifen, an AI, or tamoxifen followed by an AI. The patients were randomly assigned at the end of initial endocrine therapy to either 2 years or 5 years of anastrozole.
As noted before, disease-free survival (DFS), the primary endpoint, was virtually identical between the treatment arms. The DFS rate at a median of 8.75 years after randomization – that is, approximately 14 years after diagnosis – was 71.1% among patients treated for 2 additional years, vs. 70.3% for patients treated for 5 extra years, translating into a hazard ratio of 1.007 and making the contest a statistical dead heat.
Similarly, there was no difference by anastrozole duration in the secondary endpoint of overall survival at 10 years, with respective rates of 85.3% vs. 84.9%, with a hazard ratio identical to that in the DFS analysis.
Where the 5-year schedule surpassed the 2-year schedule, however, was in apparent risk for fractures, which was 6.3% after 5 years of additional therapy, compared with 4.7% at 5 years among patients who received just 2 additional years of anastrozole. The hazard ratio associated with the difference was 1.353, but because the lower end of the 95% confidence interval was 1.00, the finding was of borderline significance (P = .053), Dr. Gnant acknowledged.
There are several ongoing translational studies that may help to identify specific molecular characteristics that could predict benefit from prolonged extended therapy in a given patient, “but for now we can conclude that 7 years are good enough for almost every patient with luminal breast cancer,” Dr. Gnant said at a briefing prior to his presentation of the study in an oral session.
“I do believe that for us as clinical scientists a negative trial is always disappointing, but I think the clinical take-home message can actually help to avoid unnecessary side effects for many, many women,” he added.
Asked at the briefing whether, given the identical survival curves between the two trial arms, additional therapy beyond 5 years was needed, Dr. Gnant replied “that was addressed by other trials. I think that the trials after tamoxifen are very clear: We have hazard ratios around 0.6 after tamoxifen, so some type of extension for adding aromatase inhibitors should be the standard of care.”
He noted that the optimal duration of additional therapy with an AI has not been known, because the trial that could have answered that question, the MA-17 trial, was halted and unblinded after just 2.5 years when an interim analysis showed superior survival with letrozole (Femara), compared with placebo.
More than 60% of patients in the placebo group in that trial were crossed over to letrozole, further muddying long-term follow-up results.
Carlos Arteaga, MD, director of the Harold C. Simmons Comprehensive Cancer Center at University of Texas Southwestern Medical Center in Dallas, who moderated the briefing, agreed with Dr. Gnant that this ostensibly negative trial had good results for patients.
“I hope that we continue to see more de-escalation studies. I hope that as we combine AIs with CDK4/6 inhibitors, we may make therapy even shorter. I think we should do better than just extending and extending and extending. We have to come up with better ideas,” he said in an interview.
The ABCSG-16 study was supported by AstraZeneca. Dr. Gnant disclosed research funding, honoraria, and travel funding from that company and others. Dr. Arteaga disclosed consulting fees from AstraZeneca and other companies.
SOURCE: Gnant et al. SABCS 2017 Abstract GS3-01
SAN ANTONIO – Five years of additional therapy with anastrozole (Arimidex) was no more effective than 2 additional years following the standard 5 years of initial endocrine therapy in postmenopausal women with hormone receptor–positive (HR+) breast cancer, Austrian investigators reported.
In fact, the only thing that the additional years of the aromatase inhibitor (AI) anastrozole seemed to add was increased risk for fracture, said Michael Gnant, MD, from the Medical University of Vienna, on behalf of colleagues in the ABCSG-16 trial.
Previous trials have convincingly demonstrated the benefit of giving patients an AI for 5 years after 5 years of tamoxifen, but the optimal duration of extended adjuvant AI therapy is not known, Dr. Gnant said.
The ABCSG trialists recruited 3,484 postmenopausal women from with HR+, stage T1-3, node-negative or -positive, nonmetastatic breast cancer who had completed 4-6 years of endocrine therapy with either tamoxifen, an AI, or tamoxifen followed by an AI. The patients were randomly assigned at the end of initial endocrine therapy to either 2 years or 5 years of anastrozole.
As noted before, disease-free survival (DFS), the primary endpoint, was virtually identical between the treatment arms. The DFS rate at a median of 8.75 years after randomization – that is, approximately 14 years after diagnosis – was 71.1% among patients treated for 2 additional years, vs. 70.3% for patients treated for 5 extra years, translating into a hazard ratio of 1.007 and making the contest a statistical dead heat.
Similarly, there was no difference by anastrozole duration in the secondary endpoint of overall survival at 10 years, with respective rates of 85.3% vs. 84.9%, with a hazard ratio identical to that in the DFS analysis.
Where the 5-year schedule surpassed the 2-year schedule, however, was in apparent risk for fractures, which was 6.3% after 5 years of additional therapy, compared with 4.7% at 5 years among patients who received just 2 additional years of anastrozole. The hazard ratio associated with the difference was 1.353, but because the lower end of the 95% confidence interval was 1.00, the finding was of borderline significance (P = .053), Dr. Gnant acknowledged.
There are several ongoing translational studies that may help to identify specific molecular characteristics that could predict benefit from prolonged extended therapy in a given patient, “but for now we can conclude that 7 years are good enough for almost every patient with luminal breast cancer,” Dr. Gnant said at a briefing prior to his presentation of the study in an oral session.
“I do believe that for us as clinical scientists a negative trial is always disappointing, but I think the clinical take-home message can actually help to avoid unnecessary side effects for many, many women,” he added.
Asked at the briefing whether, given the identical survival curves between the two trial arms, additional therapy beyond 5 years was needed, Dr. Gnant replied “that was addressed by other trials. I think that the trials after tamoxifen are very clear: We have hazard ratios around 0.6 after tamoxifen, so some type of extension for adding aromatase inhibitors should be the standard of care.”
He noted that the optimal duration of additional therapy with an AI has not been known, because the trial that could have answered that question, the MA-17 trial, was halted and unblinded after just 2.5 years when an interim analysis showed superior survival with letrozole (Femara), compared with placebo.
More than 60% of patients in the placebo group in that trial were crossed over to letrozole, further muddying long-term follow-up results.
Carlos Arteaga, MD, director of the Harold C. Simmons Comprehensive Cancer Center at University of Texas Southwestern Medical Center in Dallas, who moderated the briefing, agreed with Dr. Gnant that this ostensibly negative trial had good results for patients.
“I hope that we continue to see more de-escalation studies. I hope that as we combine AIs with CDK4/6 inhibitors, we may make therapy even shorter. I think we should do better than just extending and extending and extending. We have to come up with better ideas,” he said in an interview.
The ABCSG-16 study was supported by AstraZeneca. Dr. Gnant disclosed research funding, honoraria, and travel funding from that company and others. Dr. Arteaga disclosed consulting fees from AstraZeneca and other companies.
SOURCE: Gnant et al. SABCS 2017 Abstract GS3-01
SAN ANTONIO – Five years of additional therapy with anastrozole (Arimidex) was no more effective than 2 additional years following the standard 5 years of initial endocrine therapy in postmenopausal women with hormone receptor–positive (HR+) breast cancer, Austrian investigators reported.
In fact, the only thing that the additional years of the aromatase inhibitor (AI) anastrozole seemed to add was increased risk for fracture, said Michael Gnant, MD, from the Medical University of Vienna, on behalf of colleagues in the ABCSG-16 trial.
Previous trials have convincingly demonstrated the benefit of giving patients an AI for 5 years after 5 years of tamoxifen, but the optimal duration of extended adjuvant AI therapy is not known, Dr. Gnant said.
The ABCSG trialists recruited 3,484 postmenopausal women from with HR+, stage T1-3, node-negative or -positive, nonmetastatic breast cancer who had completed 4-6 years of endocrine therapy with either tamoxifen, an AI, or tamoxifen followed by an AI. The patients were randomly assigned at the end of initial endocrine therapy to either 2 years or 5 years of anastrozole.
As noted before, disease-free survival (DFS), the primary endpoint, was virtually identical between the treatment arms. The DFS rate at a median of 8.75 years after randomization – that is, approximately 14 years after diagnosis – was 71.1% among patients treated for 2 additional years, vs. 70.3% for patients treated for 5 extra years, translating into a hazard ratio of 1.007 and making the contest a statistical dead heat.
Similarly, there was no difference by anastrozole duration in the secondary endpoint of overall survival at 10 years, with respective rates of 85.3% vs. 84.9%, with a hazard ratio identical to that in the DFS analysis.
Where the 5-year schedule surpassed the 2-year schedule, however, was in apparent risk for fractures, which was 6.3% after 5 years of additional therapy, compared with 4.7% at 5 years among patients who received just 2 additional years of anastrozole. The hazard ratio associated with the difference was 1.353, but because the lower end of the 95% confidence interval was 1.00, the finding was of borderline significance (P = .053), Dr. Gnant acknowledged.
There are several ongoing translational studies that may help to identify specific molecular characteristics that could predict benefit from prolonged extended therapy in a given patient, “but for now we can conclude that 7 years are good enough for almost every patient with luminal breast cancer,” Dr. Gnant said at a briefing prior to his presentation of the study in an oral session.
“I do believe that for us as clinical scientists a negative trial is always disappointing, but I think the clinical take-home message can actually help to avoid unnecessary side effects for many, many women,” he added.
Asked at the briefing whether, given the identical survival curves between the two trial arms, additional therapy beyond 5 years was needed, Dr. Gnant replied “that was addressed by other trials. I think that the trials after tamoxifen are very clear: We have hazard ratios around 0.6 after tamoxifen, so some type of extension for adding aromatase inhibitors should be the standard of care.”
He noted that the optimal duration of additional therapy with an AI has not been known, because the trial that could have answered that question, the MA-17 trial, was halted and unblinded after just 2.5 years when an interim analysis showed superior survival with letrozole (Femara), compared with placebo.
More than 60% of patients in the placebo group in that trial were crossed over to letrozole, further muddying long-term follow-up results.
Carlos Arteaga, MD, director of the Harold C. Simmons Comprehensive Cancer Center at University of Texas Southwestern Medical Center in Dallas, who moderated the briefing, agreed with Dr. Gnant that this ostensibly negative trial had good results for patients.
“I hope that we continue to see more de-escalation studies. I hope that as we combine AIs with CDK4/6 inhibitors, we may make therapy even shorter. I think we should do better than just extending and extending and extending. We have to come up with better ideas,” he said in an interview.
The ABCSG-16 study was supported by AstraZeneca. Dr. Gnant disclosed research funding, honoraria, and travel funding from that company and others. Dr. Arteaga disclosed consulting fees from AstraZeneca and other companies.
SOURCE: Gnant et al. SABCS 2017 Abstract GS3-01
REPORTING FROM SABCS 2017
Key clinical point: Disease-free and overall survival were no different for women treated with 2 or 5 additional years of aromatase inhibitor therapy following 4-6 years of initial endocrine therapy.
Major finding: The hazard ratio for both DFS and OS with 5 additional years of anastrozole, compared with 2 years, was 1.007 and was not statistically significant.
Data source: Randomized phase 3 trial in 3,484 postmenopausal women with hormone receptor–positive breast cancer.
Disclosures: The ABCSG-16 study was supported by AstraZeneca. Dr. Gnant disclosed research funding, honoraria, and travel funding from that company and others. Dr. Arteaga disclosed consulting fees from AstraZeneca and other companies.
Source: Gnant et al. SABCS 2017 Abstract GS3-01
Shaping practice: Z1071 continues to redefine axillary management
SAN DIEGO – A 2013 breast cancer trial is changing the way lymph nodes are managed in women with node-positive disease who have an axillary pathologic complete response to neoadjuvant chemotherapy.
Emerging additional data support the initial theory of the American College of Surgeons Oncology Group (ACOSOG) Z1071 trial, said Judy C. Boughey, MD, FACS, at the American College of Surgeons Clinical Congress: Performing sentinel lymph node surgery after chemotherapy is an acceptable alternative for some women. This change in practice could bestow a profound long-term benefit on the approximately 40% of patients, who have an axillary pathologic complete response after neoadjuvant chemotherapy (NAC) – patients who otherwise might undergo an unnecessary axillary node exploration, which can lead to higher risk of lymphedema, said Dr. Boughey, head of surgical research at the Mayo Clinic, Rochester, Minn.
“About 20% of patients who are treated with chemotherapy for their breast cancer receive the chemotherapy prior to surgery. Of those who do receive neoadjuvant chemotherapy, probably half could benefit from this approach,” she said. “Lymphedema after axillary dissection is one of the situations patients are most concerned about. This approach is a great one when patients have a good chemotherapy response, and we want to reliably reassure ourselves that there’s no disease left in the axilla without automatically removing all the nodes. Of course, if there is any remaining disease in any of the lymph nodes, the current standard is still to remove all the nodes. This approach, however, optimizes management for patients who have the best response to chemotherapy.”
Neoadjuvant therapy success
Prechemotherapy nodal exploration was routine a decade or so ago and is what many surgeons were most comfortable with, Dr. Boughey said. “We know the false-negative rate, and chemotherapy doesn’t interfere with axillary staging. However, it means patients have to go through two surgeries, and, although the chemotherapy does not interfere with the procedure, if any of the sentinel nodes are positive and an axillary dissection is performed at the same setting, then systemic therapy will be delayed. However, most importantly, when the sentinel node is removed prior to chemotherapy, we lose the ability to assess axillary response to chemotherapy – which correlates with survival.”
The biggest drawback of axillary dissection is its potential for lifelong morbidity from lymphedema. “Women know about this. They worry about this, and they want to avoid it if at all possible,” Dr. Boughey said.
More effective, targeted chemotherapeutic agents have resulted in higher rates of eradication of disease with neoadjuvant treatment. So this leads to the question: Why not reassess nodes after treatment, when these drugs have had a chance to work? Doing so reduces the one-size-fits-all prescription of axillary dissection and, thus, the number of women with lasting adverse events.
Some early data supported this theory
In 2009, researchers at the MD Anderson Center reported that sentinel node surgery after chemotherapy in patients with node-negative breast cancer resulted in fewer positive sentinel nodes and decreased unnecessary axillary dissections. Node identification rates were about 98% whether the surgery came before or after treatment. The false-negative rate hovered around 5%. And there were significantly fewer axillary dissections with posttreatment surgery: 20% vs. 36% in women with T2 disease and 30% vs. 51% in those with T3 disease. Importantly, holding off on the surgery didn’t lead to higher local-regional failure rates or survival among the 3,746 women treated during 1994-2007.
The American College of Surgeons Oncology Group Z1071 trial was designed to explore this question in patients with node-positive breast cancer. The Z1071 trial enrolled 756 women who had clinical T0-T4, N1-N2, M0 breast cancer and received neoadjuvant chemotherapy. Patients underwent both sentinel lymph node surgery and axillary lymph node dissection following chemotherapy. The primary endpoint was the false-negative rate of sentinel lymph node surgery after chemotherapy in women who presented with cN1 disease and had at least two sentinel nodes resected; a rate of 10% lower was considered acceptable and would justify the approach.
Of the entire cohort, 40% had a complete pathologic nodal response rate. The sentinel node identification rate was nearly 93%. The false-negative rate among 525 women with two or more positive sentinel nodes, however, was 12.6% – short of the 10% rate investigators needed to deem the study a success, Dr. Boughey said.
But there were some positive findings in subgroup analyses. Among women who had nodes identified with a dual tracer (both dye and radioactive clipping), the false-negative rate dipped to 10.8%. It was just 9% in those who had more than two sentinel nodes identified.
A recent subanalysis of the Z1071 trial further refined these data. It looked at 170 of the patients with cN1 disease (32%) who had had a clip placed in the positive lymph node at the time of percutaneous biopsy and compared false-negative rates among them with rates in the 355 patients who were not clipped.
“When we looked at them, if the clipped node came out during the sentinel node surgery, then the false-negative rate dropped down to about 7%,” Dr. Boughey said. The comparator group pointed out the value of using a clip. The false-negative rate was 13% in patients who didn’t have a clip placed and 19% in the patients whose clip wasn’t retrieved until axillary dissection.
The results of Z1071 and its subanalyses have popularized nodal clipping, Dr. Boughey said. “When we ran Z1071, clipping wasn’t commonly being performed, but there has been a huge uptake in it now.”
Confirmatory data
Other recent studies confirm the feasibility of this approach in women who have clinically negative nodes after NAC.
In 2013, the German study SENTINA (sentinel-lymph-node biopsy in patients with breast cancer before and after neoadjuvant chemotherapy) explored the false-negative rate in women who had sentinel node biopsy before or after neoadjuvant chemotherapy. Overall, it found an unacceptably high false-negative rate of 14% in women with node positive disease who converted to clinically negative nodal status. However, when the analysis was limited to those cases with at least two sentinel nodes, the false-negative rate was less than 10%, once more suggesting a potential role for sentinel node surgery after neoadjuvant chemotherapy.
In 2015, the Sentinel Node Biopsy Following Neoadjuvant Chemotherapy (SN FNAC) study highlighted the potential effect of sentinel node surgery after NAC. The prospective study showed not only that the strategy was safe, with a false-negative rate around 8%, but also that it could have eliminated complete axillary dissection in about 30% of the cohort.
The study enrolled 153 women with biopsy-proven node-positive breast cancer (T0-3, N1-2) who underwent both sentinel node surgery and complete nodal dissection. Immunohistochemistry of the retrieved sentinel nodes was mandatory, and the presence of any tumor cells in the sentinel node rendered it positive.
The sentinel node retrieval rate was 88%, and the false-negative rate, 8.4%. The study also employed dual tracers of isotope and blue dye in a majority of patients; this was associated with a threefold decrease in the false-negative rate in those patients, dropping it to around 5%. “By using sentinel node biopsy after NAC, axillary node dissection could potentially be avoided in at least 30% of patients who present with node-positive breast cancer,” the study’s team concluded.
Long-term consequences?
It’s increasingly clear that for carefully selected patients, with robust NAC response, a postchemotherapy assessment can accurately assess nodal disease – especially if dual tracers are employed, several sentinel nodes examined, and the biopsy-proven positive node is resected. What isn’t clear yet is the long-term effect of this strategy, Dr. Boughey said.
“Five years ago, when Z1071 was first being reported, I would discuss it in terms of the controversy, and give the pros and cons,” she said. “But now that we have more information about this strategy under our belts, I feel much more confident. However, we still do not have information on patients with node-positive disease who have been treated with sentinel node only after neoadjuvant chemotherapy and followed for 5 or 10 years. That’s the piece we just can’t have, without time.”
Dr. Boughey had no relevant financial disclosures.
SOURCE: Boughey JC. Session PS108.
SAN DIEGO – A 2013 breast cancer trial is changing the way lymph nodes are managed in women with node-positive disease who have an axillary pathologic complete response to neoadjuvant chemotherapy.
Emerging additional data support the initial theory of the American College of Surgeons Oncology Group (ACOSOG) Z1071 trial, said Judy C. Boughey, MD, FACS, at the American College of Surgeons Clinical Congress: Performing sentinel lymph node surgery after chemotherapy is an acceptable alternative for some women. This change in practice could bestow a profound long-term benefit on the approximately 40% of patients, who have an axillary pathologic complete response after neoadjuvant chemotherapy (NAC) – patients who otherwise might undergo an unnecessary axillary node exploration, which can lead to higher risk of lymphedema, said Dr. Boughey, head of surgical research at the Mayo Clinic, Rochester, Minn.
“About 20% of patients who are treated with chemotherapy for their breast cancer receive the chemotherapy prior to surgery. Of those who do receive neoadjuvant chemotherapy, probably half could benefit from this approach,” she said. “Lymphedema after axillary dissection is one of the situations patients are most concerned about. This approach is a great one when patients have a good chemotherapy response, and we want to reliably reassure ourselves that there’s no disease left in the axilla without automatically removing all the nodes. Of course, if there is any remaining disease in any of the lymph nodes, the current standard is still to remove all the nodes. This approach, however, optimizes management for patients who have the best response to chemotherapy.”
Neoadjuvant therapy success
Prechemotherapy nodal exploration was routine a decade or so ago and is what many surgeons were most comfortable with, Dr. Boughey said. “We know the false-negative rate, and chemotherapy doesn’t interfere with axillary staging. However, it means patients have to go through two surgeries, and, although the chemotherapy does not interfere with the procedure, if any of the sentinel nodes are positive and an axillary dissection is performed at the same setting, then systemic therapy will be delayed. However, most importantly, when the sentinel node is removed prior to chemotherapy, we lose the ability to assess axillary response to chemotherapy – which correlates with survival.”
The biggest drawback of axillary dissection is its potential for lifelong morbidity from lymphedema. “Women know about this. They worry about this, and they want to avoid it if at all possible,” Dr. Boughey said.
More effective, targeted chemotherapeutic agents have resulted in higher rates of eradication of disease with neoadjuvant treatment. So this leads to the question: Why not reassess nodes after treatment, when these drugs have had a chance to work? Doing so reduces the one-size-fits-all prescription of axillary dissection and, thus, the number of women with lasting adverse events.
Some early data supported this theory
In 2009, researchers at the MD Anderson Center reported that sentinel node surgery after chemotherapy in patients with node-negative breast cancer resulted in fewer positive sentinel nodes and decreased unnecessary axillary dissections. Node identification rates were about 98% whether the surgery came before or after treatment. The false-negative rate hovered around 5%. And there were significantly fewer axillary dissections with posttreatment surgery: 20% vs. 36% in women with T2 disease and 30% vs. 51% in those with T3 disease. Importantly, holding off on the surgery didn’t lead to higher local-regional failure rates or survival among the 3,746 women treated during 1994-2007.
The American College of Surgeons Oncology Group Z1071 trial was designed to explore this question in patients with node-positive breast cancer. The Z1071 trial enrolled 756 women who had clinical T0-T4, N1-N2, M0 breast cancer and received neoadjuvant chemotherapy. Patients underwent both sentinel lymph node surgery and axillary lymph node dissection following chemotherapy. The primary endpoint was the false-negative rate of sentinel lymph node surgery after chemotherapy in women who presented with cN1 disease and had at least two sentinel nodes resected; a rate of 10% lower was considered acceptable and would justify the approach.
Of the entire cohort, 40% had a complete pathologic nodal response rate. The sentinel node identification rate was nearly 93%. The false-negative rate among 525 women with two or more positive sentinel nodes, however, was 12.6% – short of the 10% rate investigators needed to deem the study a success, Dr. Boughey said.
But there were some positive findings in subgroup analyses. Among women who had nodes identified with a dual tracer (both dye and radioactive clipping), the false-negative rate dipped to 10.8%. It was just 9% in those who had more than two sentinel nodes identified.
A recent subanalysis of the Z1071 trial further refined these data. It looked at 170 of the patients with cN1 disease (32%) who had had a clip placed in the positive lymph node at the time of percutaneous biopsy and compared false-negative rates among them with rates in the 355 patients who were not clipped.
“When we looked at them, if the clipped node came out during the sentinel node surgery, then the false-negative rate dropped down to about 7%,” Dr. Boughey said. The comparator group pointed out the value of using a clip. The false-negative rate was 13% in patients who didn’t have a clip placed and 19% in the patients whose clip wasn’t retrieved until axillary dissection.
The results of Z1071 and its subanalyses have popularized nodal clipping, Dr. Boughey said. “When we ran Z1071, clipping wasn’t commonly being performed, but there has been a huge uptake in it now.”
Confirmatory data
Other recent studies confirm the feasibility of this approach in women who have clinically negative nodes after NAC.
In 2013, the German study SENTINA (sentinel-lymph-node biopsy in patients with breast cancer before and after neoadjuvant chemotherapy) explored the false-negative rate in women who had sentinel node biopsy before or after neoadjuvant chemotherapy. Overall, it found an unacceptably high false-negative rate of 14% in women with node positive disease who converted to clinically negative nodal status. However, when the analysis was limited to those cases with at least two sentinel nodes, the false-negative rate was less than 10%, once more suggesting a potential role for sentinel node surgery after neoadjuvant chemotherapy.
In 2015, the Sentinel Node Biopsy Following Neoadjuvant Chemotherapy (SN FNAC) study highlighted the potential effect of sentinel node surgery after NAC. The prospective study showed not only that the strategy was safe, with a false-negative rate around 8%, but also that it could have eliminated complete axillary dissection in about 30% of the cohort.
The study enrolled 153 women with biopsy-proven node-positive breast cancer (T0-3, N1-2) who underwent both sentinel node surgery and complete nodal dissection. Immunohistochemistry of the retrieved sentinel nodes was mandatory, and the presence of any tumor cells in the sentinel node rendered it positive.
The sentinel node retrieval rate was 88%, and the false-negative rate, 8.4%. The study also employed dual tracers of isotope and blue dye in a majority of patients; this was associated with a threefold decrease in the false-negative rate in those patients, dropping it to around 5%. “By using sentinel node biopsy after NAC, axillary node dissection could potentially be avoided in at least 30% of patients who present with node-positive breast cancer,” the study’s team concluded.
Long-term consequences?
It’s increasingly clear that for carefully selected patients, with robust NAC response, a postchemotherapy assessment can accurately assess nodal disease – especially if dual tracers are employed, several sentinel nodes examined, and the biopsy-proven positive node is resected. What isn’t clear yet is the long-term effect of this strategy, Dr. Boughey said.
“Five years ago, when Z1071 was first being reported, I would discuss it in terms of the controversy, and give the pros and cons,” she said. “But now that we have more information about this strategy under our belts, I feel much more confident. However, we still do not have information on patients with node-positive disease who have been treated with sentinel node only after neoadjuvant chemotherapy and followed for 5 or 10 years. That’s the piece we just can’t have, without time.”
Dr. Boughey had no relevant financial disclosures.
SOURCE: Boughey JC. Session PS108.
SAN DIEGO – A 2013 breast cancer trial is changing the way lymph nodes are managed in women with node-positive disease who have an axillary pathologic complete response to neoadjuvant chemotherapy.
Emerging additional data support the initial theory of the American College of Surgeons Oncology Group (ACOSOG) Z1071 trial, said Judy C. Boughey, MD, FACS, at the American College of Surgeons Clinical Congress: Performing sentinel lymph node surgery after chemotherapy is an acceptable alternative for some women. This change in practice could bestow a profound long-term benefit on the approximately 40% of patients, who have an axillary pathologic complete response after neoadjuvant chemotherapy (NAC) – patients who otherwise might undergo an unnecessary axillary node exploration, which can lead to higher risk of lymphedema, said Dr. Boughey, head of surgical research at the Mayo Clinic, Rochester, Minn.
“About 20% of patients who are treated with chemotherapy for their breast cancer receive the chemotherapy prior to surgery. Of those who do receive neoadjuvant chemotherapy, probably half could benefit from this approach,” she said. “Lymphedema after axillary dissection is one of the situations patients are most concerned about. This approach is a great one when patients have a good chemotherapy response, and we want to reliably reassure ourselves that there’s no disease left in the axilla without automatically removing all the nodes. Of course, if there is any remaining disease in any of the lymph nodes, the current standard is still to remove all the nodes. This approach, however, optimizes management for patients who have the best response to chemotherapy.”
Neoadjuvant therapy success
Prechemotherapy nodal exploration was routine a decade or so ago and is what many surgeons were most comfortable with, Dr. Boughey said. “We know the false-negative rate, and chemotherapy doesn’t interfere with axillary staging. However, it means patients have to go through two surgeries, and, although the chemotherapy does not interfere with the procedure, if any of the sentinel nodes are positive and an axillary dissection is performed at the same setting, then systemic therapy will be delayed. However, most importantly, when the sentinel node is removed prior to chemotherapy, we lose the ability to assess axillary response to chemotherapy – which correlates with survival.”
The biggest drawback of axillary dissection is its potential for lifelong morbidity from lymphedema. “Women know about this. They worry about this, and they want to avoid it if at all possible,” Dr. Boughey said.
More effective, targeted chemotherapeutic agents have resulted in higher rates of eradication of disease with neoadjuvant treatment. So this leads to the question: Why not reassess nodes after treatment, when these drugs have had a chance to work? Doing so reduces the one-size-fits-all prescription of axillary dissection and, thus, the number of women with lasting adverse events.
Some early data supported this theory
In 2009, researchers at the MD Anderson Center reported that sentinel node surgery after chemotherapy in patients with node-negative breast cancer resulted in fewer positive sentinel nodes and decreased unnecessary axillary dissections. Node identification rates were about 98% whether the surgery came before or after treatment. The false-negative rate hovered around 5%. And there were significantly fewer axillary dissections with posttreatment surgery: 20% vs. 36% in women with T2 disease and 30% vs. 51% in those with T3 disease. Importantly, holding off on the surgery didn’t lead to higher local-regional failure rates or survival among the 3,746 women treated during 1994-2007.
The American College of Surgeons Oncology Group Z1071 trial was designed to explore this question in patients with node-positive breast cancer. The Z1071 trial enrolled 756 women who had clinical T0-T4, N1-N2, M0 breast cancer and received neoadjuvant chemotherapy. Patients underwent both sentinel lymph node surgery and axillary lymph node dissection following chemotherapy. The primary endpoint was the false-negative rate of sentinel lymph node surgery after chemotherapy in women who presented with cN1 disease and had at least two sentinel nodes resected; a rate of 10% lower was considered acceptable and would justify the approach.
Of the entire cohort, 40% had a complete pathologic nodal response rate. The sentinel node identification rate was nearly 93%. The false-negative rate among 525 women with two or more positive sentinel nodes, however, was 12.6% – short of the 10% rate investigators needed to deem the study a success, Dr. Boughey said.
But there were some positive findings in subgroup analyses. Among women who had nodes identified with a dual tracer (both dye and radioactive clipping), the false-negative rate dipped to 10.8%. It was just 9% in those who had more than two sentinel nodes identified.
A recent subanalysis of the Z1071 trial further refined these data. It looked at 170 of the patients with cN1 disease (32%) who had had a clip placed in the positive lymph node at the time of percutaneous biopsy and compared false-negative rates among them with rates in the 355 patients who were not clipped.
“When we looked at them, if the clipped node came out during the sentinel node surgery, then the false-negative rate dropped down to about 7%,” Dr. Boughey said. The comparator group pointed out the value of using a clip. The false-negative rate was 13% in patients who didn’t have a clip placed and 19% in the patients whose clip wasn’t retrieved until axillary dissection.
The results of Z1071 and its subanalyses have popularized nodal clipping, Dr. Boughey said. “When we ran Z1071, clipping wasn’t commonly being performed, but there has been a huge uptake in it now.”
Confirmatory data
Other recent studies confirm the feasibility of this approach in women who have clinically negative nodes after NAC.
In 2013, the German study SENTINA (sentinel-lymph-node biopsy in patients with breast cancer before and after neoadjuvant chemotherapy) explored the false-negative rate in women who had sentinel node biopsy before or after neoadjuvant chemotherapy. Overall, it found an unacceptably high false-negative rate of 14% in women with node positive disease who converted to clinically negative nodal status. However, when the analysis was limited to those cases with at least two sentinel nodes, the false-negative rate was less than 10%, once more suggesting a potential role for sentinel node surgery after neoadjuvant chemotherapy.
In 2015, the Sentinel Node Biopsy Following Neoadjuvant Chemotherapy (SN FNAC) study highlighted the potential effect of sentinel node surgery after NAC. The prospective study showed not only that the strategy was safe, with a false-negative rate around 8%, but also that it could have eliminated complete axillary dissection in about 30% of the cohort.
The study enrolled 153 women with biopsy-proven node-positive breast cancer (T0-3, N1-2) who underwent both sentinel node surgery and complete nodal dissection. Immunohistochemistry of the retrieved sentinel nodes was mandatory, and the presence of any tumor cells in the sentinel node rendered it positive.
The sentinel node retrieval rate was 88%, and the false-negative rate, 8.4%. The study also employed dual tracers of isotope and blue dye in a majority of patients; this was associated with a threefold decrease in the false-negative rate in those patients, dropping it to around 5%. “By using sentinel node biopsy after NAC, axillary node dissection could potentially be avoided in at least 30% of patients who present with node-positive breast cancer,” the study’s team concluded.
Long-term consequences?
It’s increasingly clear that for carefully selected patients, with robust NAC response, a postchemotherapy assessment can accurately assess nodal disease – especially if dual tracers are employed, several sentinel nodes examined, and the biopsy-proven positive node is resected. What isn’t clear yet is the long-term effect of this strategy, Dr. Boughey said.
“Five years ago, when Z1071 was first being reported, I would discuss it in terms of the controversy, and give the pros and cons,” she said. “But now that we have more information about this strategy under our belts, I feel much more confident. However, we still do not have information on patients with node-positive disease who have been treated with sentinel node only after neoadjuvant chemotherapy and followed for 5 or 10 years. That’s the piece we just can’t have, without time.”
Dr. Boughey had no relevant financial disclosures.
SOURCE: Boughey JC. Session PS108.
EXPERT ANALYSIS FROM THE ACS CLINICAL CONGRESS