User login
FDA approves neratinib for extended adjuvant treatment of HER2+ breast cancer
The Food and Drug Administration has approved neratinib, an oral tyrosine kinase inhibitor, for the extended adjuvant treatment of patients with early-stage, HER2-positive breast cancer who have previously been treated with trastuzumab.
Approval was based on improved invasive disease-free survival (iDFS) in the phase 3 ExteNET trial of 2,840 women with early-stage HER2-positive breast cancer who were within 2 years of completing adjuvant trastuzumab. Patients were randomized to receive either neratinib or placebo daily for 1 year. After 2 years of follow-up, iDFS was 94.2% in patients treated with neratinib, compared with 91.9% in those receiving placebo (hazard ratio, 0.66; 95% confidence interval, 0.49, 0.90; P = .008), according to the FDA statement.
The most common adverse reactions to neratinib in ExteNET were diarrhea, nausea, abdominal pain, fatigue, vomiting, rash, stomatitis, decreased appetite, muscle spasms, dyspepsia, AST or ALT increase, nail disorder, dry skin, abdominal distention, weight loss, and urinary tract infection. Diarrhea was observed in 16.8% of neratinib-treated patients.
The recommended dose of of neratinib is 240 mg (six 40 mg tablets) given orally once daily with food, continuously, for 1 year. Patients should be given antidiarrheal prophylaxis for the first 56 days of treatment with neratinib and as needed thereafter to help manage diarrhea, the FDA said.
The Food and Drug Administration has approved neratinib, an oral tyrosine kinase inhibitor, for the extended adjuvant treatment of patients with early-stage, HER2-positive breast cancer who have previously been treated with trastuzumab.
Approval was based on improved invasive disease-free survival (iDFS) in the phase 3 ExteNET trial of 2,840 women with early-stage HER2-positive breast cancer who were within 2 years of completing adjuvant trastuzumab. Patients were randomized to receive either neratinib or placebo daily for 1 year. After 2 years of follow-up, iDFS was 94.2% in patients treated with neratinib, compared with 91.9% in those receiving placebo (hazard ratio, 0.66; 95% confidence interval, 0.49, 0.90; P = .008), according to the FDA statement.
The most common adverse reactions to neratinib in ExteNET were diarrhea, nausea, abdominal pain, fatigue, vomiting, rash, stomatitis, decreased appetite, muscle spasms, dyspepsia, AST or ALT increase, nail disorder, dry skin, abdominal distention, weight loss, and urinary tract infection. Diarrhea was observed in 16.8% of neratinib-treated patients.
The recommended dose of of neratinib is 240 mg (six 40 mg tablets) given orally once daily with food, continuously, for 1 year. Patients should be given antidiarrheal prophylaxis for the first 56 days of treatment with neratinib and as needed thereafter to help manage diarrhea, the FDA said.
The Food and Drug Administration has approved neratinib, an oral tyrosine kinase inhibitor, for the extended adjuvant treatment of patients with early-stage, HER2-positive breast cancer who have previously been treated with trastuzumab.
Approval was based on improved invasive disease-free survival (iDFS) in the phase 3 ExteNET trial of 2,840 women with early-stage HER2-positive breast cancer who were within 2 years of completing adjuvant trastuzumab. Patients were randomized to receive either neratinib or placebo daily for 1 year. After 2 years of follow-up, iDFS was 94.2% in patients treated with neratinib, compared with 91.9% in those receiving placebo (hazard ratio, 0.66; 95% confidence interval, 0.49, 0.90; P = .008), according to the FDA statement.
The most common adverse reactions to neratinib in ExteNET were diarrhea, nausea, abdominal pain, fatigue, vomiting, rash, stomatitis, decreased appetite, muscle spasms, dyspepsia, AST or ALT increase, nail disorder, dry skin, abdominal distention, weight loss, and urinary tract infection. Diarrhea was observed in 16.8% of neratinib-treated patients.
The recommended dose of of neratinib is 240 mg (six 40 mg tablets) given orally once daily with food, continuously, for 1 year. Patients should be given antidiarrheal prophylaxis for the first 56 days of treatment with neratinib and as needed thereafter to help manage diarrhea, the FDA said.
Use of Pap smears, mammography on the decline
Use of Pap smears and mammography has slowly but steadily declined since both peaked in the year 2000, according to the National Center for Health Statistics.
The age-adjusted rate of women aged 18 years and over who reported having had a Pap smear in the past 3 years dropped from 81.3% in 2000 to 70.2% in 2015. Over that same time period, the age-adjusted rate of women aged 40 years and over who had a mammogram over the previous 2 years declined from 70.4% in 2000 to 64% in 2015, the NCHS reported in “Health, United States, 2016.”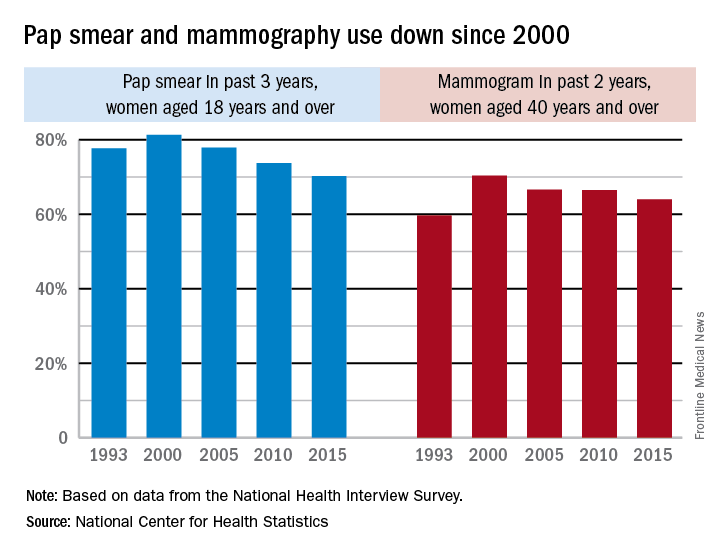
The pattern for mammography use, however, was somewhat different. Declines in use were consistent by age group, but not by race. White women were less likely to get a mammogram in 2015 (65.3%), compared with 2000 (71.4%), while Hispanic women experienced a small drop from 61.2% to 60.9%, but black, American Indian/Alaska Native, and Asian women all increased their use of mammography over that time period, the NCHS reported.
Use of Pap smears and mammography has slowly but steadily declined since both peaked in the year 2000, according to the National Center for Health Statistics.
The age-adjusted rate of women aged 18 years and over who reported having had a Pap smear in the past 3 years dropped from 81.3% in 2000 to 70.2% in 2015. Over that same time period, the age-adjusted rate of women aged 40 years and over who had a mammogram over the previous 2 years declined from 70.4% in 2000 to 64% in 2015, the NCHS reported in “Health, United States, 2016.”
The pattern for mammography use, however, was somewhat different. Declines in use were consistent by age group, but not by race. White women were less likely to get a mammogram in 2015 (65.3%), compared with 2000 (71.4%), while Hispanic women experienced a small drop from 61.2% to 60.9%, but black, American Indian/Alaska Native, and Asian women all increased their use of mammography over that time period, the NCHS reported.
Use of Pap smears and mammography has slowly but steadily declined since both peaked in the year 2000, according to the National Center for Health Statistics.
The age-adjusted rate of women aged 18 years and over who reported having had a Pap smear in the past 3 years dropped from 81.3% in 2000 to 70.2% in 2015. Over that same time period, the age-adjusted rate of women aged 40 years and over who had a mammogram over the previous 2 years declined from 70.4% in 2000 to 64% in 2015, the NCHS reported in “Health, United States, 2016.”
The pattern for mammography use, however, was somewhat different. Declines in use were consistent by age group, but not by race. White women were less likely to get a mammogram in 2015 (65.3%), compared with 2000 (71.4%), while Hispanic women experienced a small drop from 61.2% to 60.9%, but black, American Indian/Alaska Native, and Asian women all increased their use of mammography over that time period, the NCHS reported.
Temporary tissue expanders optimize radiotherapy after mastectomy
LAS VEGAS – Radiation oncologists at the University of Texas MD Anderson Cancer Center, Houston, were able to complete 98% of their radiotherapy plans when women received temporary tissue expanders, instead of immediate reconstructions, at the time of skin-sparing mastectomy, in a series of 384 women, most with stage 2-3 breast cancer.
The expanders – saline-filled bags commonly used in plastic surgery to create new skin – were kept in place but deflated for radiotherapy, which allowed for optimal access to treatment fields; the final reconstruction, successful in 90% of women, came a median of 7 months following radiation.
“The shape and volume of the reconstruction” – and the need to avoid damaging the new breast – “got in the way of putting radiation where we wanted it to be. We ended up having bad radiotherapy plans, patients not getting skin-sparing mastectomies, and high probabilities of radiation complications to the reconstruction,” said investigator Eric Strom, MD, professor of radiation oncology at MD Anderson.
Radiologists and plastic and oncologic surgeons collaborated to try tissue expanders instead. “We wanted the advantage of skin-sparing mastectomy without the disadvantages” of immediate reconstruction, Dr. Strom said at the American Society of Breast Surgeons annual meeting.
With the new approach, “radiotherapy is superior. We don’t have to compromise our plans. I can put radiation everywhere it needs to be, without frying the heart” and almost completely avoiding the lungs, he said.
The 5-year rates of locoregional control, disease-free survival, and overall survival were 99.2%, 86.1%, and 92.4%, respectively, which “is extraordinary” in patients with stage 2-3 breast cancer, and likely due at least in part to optimal radiotherapy, he said.
Tissue expanders also keep the skin envelope open so it’s able to receive a graft at final reconstruction; abdominal skin doesn’t have to brought up to recreate the breast.
“This approach lessens negative interactions between breast reconstruction and [radiotherapy] and offers patients what they most desire: a high probability of freedom from cancer and optimal final aesthetic outcome,” said Zeina Ayoub, MD, a radiation oncology fellow at Anderson who presented the findings.
The median age of the women was 44 years, and almost all were node positive. Radiation was delivered to the chest wall and regional lymphatics, including the internal mammary chain.
Fifty women (13.0%) required explantation after radiation but before reconstruction, most commonly because of cellulitis; even so, more than half went on to final reconstruction.
Abdominal autologous reconstruction was the most common type, followed by latissimus dorsi–based reconstruction, and exchange of the tissue expander with an implant.
Dr. Ayoub and Dr. Strom had no relevant disclosures.
LAS VEGAS – Radiation oncologists at the University of Texas MD Anderson Cancer Center, Houston, were able to complete 98% of their radiotherapy plans when women received temporary tissue expanders, instead of immediate reconstructions, at the time of skin-sparing mastectomy, in a series of 384 women, most with stage 2-3 breast cancer.
The expanders – saline-filled bags commonly used in plastic surgery to create new skin – were kept in place but deflated for radiotherapy, which allowed for optimal access to treatment fields; the final reconstruction, successful in 90% of women, came a median of 7 months following radiation.
“The shape and volume of the reconstruction” – and the need to avoid damaging the new breast – “got in the way of putting radiation where we wanted it to be. We ended up having bad radiotherapy plans, patients not getting skin-sparing mastectomies, and high probabilities of radiation complications to the reconstruction,” said investigator Eric Strom, MD, professor of radiation oncology at MD Anderson.
Radiologists and plastic and oncologic surgeons collaborated to try tissue expanders instead. “We wanted the advantage of skin-sparing mastectomy without the disadvantages” of immediate reconstruction, Dr. Strom said at the American Society of Breast Surgeons annual meeting.
With the new approach, “radiotherapy is superior. We don’t have to compromise our plans. I can put radiation everywhere it needs to be, without frying the heart” and almost completely avoiding the lungs, he said.
The 5-year rates of locoregional control, disease-free survival, and overall survival were 99.2%, 86.1%, and 92.4%, respectively, which “is extraordinary” in patients with stage 2-3 breast cancer, and likely due at least in part to optimal radiotherapy, he said.
Tissue expanders also keep the skin envelope open so it’s able to receive a graft at final reconstruction; abdominal skin doesn’t have to brought up to recreate the breast.
“This approach lessens negative interactions between breast reconstruction and [radiotherapy] and offers patients what they most desire: a high probability of freedom from cancer and optimal final aesthetic outcome,” said Zeina Ayoub, MD, a radiation oncology fellow at Anderson who presented the findings.
The median age of the women was 44 years, and almost all were node positive. Radiation was delivered to the chest wall and regional lymphatics, including the internal mammary chain.
Fifty women (13.0%) required explantation after radiation but before reconstruction, most commonly because of cellulitis; even so, more than half went on to final reconstruction.
Abdominal autologous reconstruction was the most common type, followed by latissimus dorsi–based reconstruction, and exchange of the tissue expander with an implant.
Dr. Ayoub and Dr. Strom had no relevant disclosures.
LAS VEGAS – Radiation oncologists at the University of Texas MD Anderson Cancer Center, Houston, were able to complete 98% of their radiotherapy plans when women received temporary tissue expanders, instead of immediate reconstructions, at the time of skin-sparing mastectomy, in a series of 384 women, most with stage 2-3 breast cancer.
The expanders – saline-filled bags commonly used in plastic surgery to create new skin – were kept in place but deflated for radiotherapy, which allowed for optimal access to treatment fields; the final reconstruction, successful in 90% of women, came a median of 7 months following radiation.
“The shape and volume of the reconstruction” – and the need to avoid damaging the new breast – “got in the way of putting radiation where we wanted it to be. We ended up having bad radiotherapy plans, patients not getting skin-sparing mastectomies, and high probabilities of radiation complications to the reconstruction,” said investigator Eric Strom, MD, professor of radiation oncology at MD Anderson.
Radiologists and plastic and oncologic surgeons collaborated to try tissue expanders instead. “We wanted the advantage of skin-sparing mastectomy without the disadvantages” of immediate reconstruction, Dr. Strom said at the American Society of Breast Surgeons annual meeting.
With the new approach, “radiotherapy is superior. We don’t have to compromise our plans. I can put radiation everywhere it needs to be, without frying the heart” and almost completely avoiding the lungs, he said.
The 5-year rates of locoregional control, disease-free survival, and overall survival were 99.2%, 86.1%, and 92.4%, respectively, which “is extraordinary” in patients with stage 2-3 breast cancer, and likely due at least in part to optimal radiotherapy, he said.
Tissue expanders also keep the skin envelope open so it’s able to receive a graft at final reconstruction; abdominal skin doesn’t have to brought up to recreate the breast.
“This approach lessens negative interactions between breast reconstruction and [radiotherapy] and offers patients what they most desire: a high probability of freedom from cancer and optimal final aesthetic outcome,” said Zeina Ayoub, MD, a radiation oncology fellow at Anderson who presented the findings.
The median age of the women was 44 years, and almost all were node positive. Radiation was delivered to the chest wall and regional lymphatics, including the internal mammary chain.
Fifty women (13.0%) required explantation after radiation but before reconstruction, most commonly because of cellulitis; even so, more than half went on to final reconstruction.
Abdominal autologous reconstruction was the most common type, followed by latissimus dorsi–based reconstruction, and exchange of the tissue expander with an implant.
Dr. Ayoub and Dr. Strom had no relevant disclosures.
AT ASBS 2017
Key clinical point:
Major finding: The 5-year rates of locoregional control, disease-free survival, and overall survival were 99.2%, 86.1%, and 92.4%, respectively, likely due at least in part to optimal radiotherapy.
Data source: Review of 384 patients.
Disclosures: The investigators said they had no relevant disclosures.
Consequence of change? Medicaid disenrollment delayed breast cancer diagnosis
A spike in later-stage breast cancers is a potential byproduct of the Republican-proposed Medicaid reductions, according to Wafa Tarazi, PhD, and her colleagues.
The team looked at breast cancer stage at diagnosis following the 2005 Medicaid disenrollment of nearly 170,000 nonelderly adults in Tennessee that occurred because of state financial issues.
“Overall, nonelderly women in Tennessee were diagnosed at later stages of disease and experienced more delays in treatment in the period after disenrollment,” wrote Dr. Tarazi of Virginia Commonwealth University, Richmond, and her colleagues. “Disenrollment was found to be associated with a 3.3 percentage point increase in late stage of disease at the time of diagnosis” (Cancer 2016 June 26. doi: 10.1002/cncr.30771).
The investigators offered a few explanations for why this could be the case.
While disenrollment was associated with later stage of breast cancer at diagnosis, it also was associated with a 1.9 percentage-point decrease in a 60-day–plus delay in surgical treatment and a 1.4 percentage-point decrease in a greater-than-90-day–plus delay in treatment for women living in low-income ZIP codes, compared with women living in high-income ZIP codes.
Contractions “in the availability of Medicaid coverage have important health consequences for low-income women, and may increase income-related disparities, morbidity, and mortality for those diagnosed with breast cancer,” the authors wrote. “These negative health consequences should be considered by policymakers who weigh the costs and benefits of implementing or discontinuing expanded Medicaid coverage under the Patient Protection and Affordable Care Act and future federal and state policies.”
The Susan G. Komen Breast Cancer Foundation funded the study. The authors reported no conflicts of interest.
A spike in later-stage breast cancers is a potential byproduct of the Republican-proposed Medicaid reductions, according to Wafa Tarazi, PhD, and her colleagues.
The team looked at breast cancer stage at diagnosis following the 2005 Medicaid disenrollment of nearly 170,000 nonelderly adults in Tennessee that occurred because of state financial issues.
“Overall, nonelderly women in Tennessee were diagnosed at later stages of disease and experienced more delays in treatment in the period after disenrollment,” wrote Dr. Tarazi of Virginia Commonwealth University, Richmond, and her colleagues. “Disenrollment was found to be associated with a 3.3 percentage point increase in late stage of disease at the time of diagnosis” (Cancer 2016 June 26. doi: 10.1002/cncr.30771).
The investigators offered a few explanations for why this could be the case.
While disenrollment was associated with later stage of breast cancer at diagnosis, it also was associated with a 1.9 percentage-point decrease in a 60-day–plus delay in surgical treatment and a 1.4 percentage-point decrease in a greater-than-90-day–plus delay in treatment for women living in low-income ZIP codes, compared with women living in high-income ZIP codes.
Contractions “in the availability of Medicaid coverage have important health consequences for low-income women, and may increase income-related disparities, morbidity, and mortality for those diagnosed with breast cancer,” the authors wrote. “These negative health consequences should be considered by policymakers who weigh the costs and benefits of implementing or discontinuing expanded Medicaid coverage under the Patient Protection and Affordable Care Act and future federal and state policies.”
The Susan G. Komen Breast Cancer Foundation funded the study. The authors reported no conflicts of interest.
A spike in later-stage breast cancers is a potential byproduct of the Republican-proposed Medicaid reductions, according to Wafa Tarazi, PhD, and her colleagues.
The team looked at breast cancer stage at diagnosis following the 2005 Medicaid disenrollment of nearly 170,000 nonelderly adults in Tennessee that occurred because of state financial issues.
“Overall, nonelderly women in Tennessee were diagnosed at later stages of disease and experienced more delays in treatment in the period after disenrollment,” wrote Dr. Tarazi of Virginia Commonwealth University, Richmond, and her colleagues. “Disenrollment was found to be associated with a 3.3 percentage point increase in late stage of disease at the time of diagnosis” (Cancer 2016 June 26. doi: 10.1002/cncr.30771).
The investigators offered a few explanations for why this could be the case.
While disenrollment was associated with later stage of breast cancer at diagnosis, it also was associated with a 1.9 percentage-point decrease in a 60-day–plus delay in surgical treatment and a 1.4 percentage-point decrease in a greater-than-90-day–plus delay in treatment for women living in low-income ZIP codes, compared with women living in high-income ZIP codes.
Contractions “in the availability of Medicaid coverage have important health consequences for low-income women, and may increase income-related disparities, morbidity, and mortality for those diagnosed with breast cancer,” the authors wrote. “These negative health consequences should be considered by policymakers who weigh the costs and benefits of implementing or discontinuing expanded Medicaid coverage under the Patient Protection and Affordable Care Act and future federal and state policies.”
The Susan G. Komen Breast Cancer Foundation funded the study. The authors reported no conflicts of interest.
FROM CANCER
Some adjuvant endocrine therapies better than others for young breast cancer patients
Young women treated for early hormone receptor–positive, HER2-negative breast cancer fare better if their adjuvant endocrine therapy includes ovarian function suppression (OFS), according to a new analysis published online.
Senior author Gini F. Fleming, MD, director of the medical oncology breast program at the University of Chicago Medical Center, and her colleagues analyzed data from a pair of international phase III randomized adjuvant trials: the Suppression of Ovarian Function Trial (SOFT) and the Tamoxifen and Exemestane Trial (TEXT).
Main analyses were based on a respective 240 and 145 women younger than 35 years who had undergone surgery for early hormone receptor–positive, HER2-negative early breast cancer, received chemotherapy, and were randomly assigned to 5 years of various adjuvant endocrine therapies.
In SOFT, the 5-year breast cancer–free interval was 67.1% (95% CI, 54.6%-76.9%) with tamoxifen (Nolvadex) alone, 75.9% (64.0%-84.4%) with tamoxifen plus OFS, and 83.2% (72.7%-90.0%) with exemestane (Aromasin) plus OFS (J Clin Oncol. 2017 June 27 doi: 10.1200/JCO.2016.72.0946). In TEXT, it was 79.2% (66.2%-87.7%) with tamoxifen plus OFS and 81.6% (69.8%-89.2%) with exemestane plus OFS.
In a quality of life analysis among women receiving OFS, vasomotor symptoms (hot flushes and sweats) showed greatest increase from baseline (roughly 30-40 points) in the first 6 months of therapy. Loss of sexual interest and difficulties in becoming aroused were also noteworthy (8 points or greater). However, scores for global quality of life (physical well-being, mood, coping effort, and health perception) showed little change from baseline and were essentially the same as those seen among premenopausal women aged 35 or older in the same trials.
Overall, 19.8% of the young women in SOFT and TEXT stopped all protocol-assigned endocrine therapy early. The proportion rose with time and was higher than that among the older premenopausal group.
“There was a meaningful clinical benefit in breast cancer outcomes with the addition of OFS to tamoxifen and some additional benefit from use of an aromatase inhibitor with OFS. Longer follow-up is critical to clarify potential survival benefits,” the investigators wrote. “There were substantial adverse effects from these combined endocrine treatments, but they were not different in the younger and older than 35 years populations. Despite this, rates of nonadherence were slightly higher in women younger than 35 years.
“Availability of these age-specific data regarding risks and benefits of combined endocrine therapy will support shared decision making regarding OFS among young women at high risk for recurrence and death from breast cancer and, it is hoped, improve adherence among those who select OFS,” they concluded.
Young women treated for early hormone receptor–positive, HER2-negative breast cancer fare better if their adjuvant endocrine therapy includes ovarian function suppression (OFS), according to a new analysis published online.
Senior author Gini F. Fleming, MD, director of the medical oncology breast program at the University of Chicago Medical Center, and her colleagues analyzed data from a pair of international phase III randomized adjuvant trials: the Suppression of Ovarian Function Trial (SOFT) and the Tamoxifen and Exemestane Trial (TEXT).
Main analyses were based on a respective 240 and 145 women younger than 35 years who had undergone surgery for early hormone receptor–positive, HER2-negative early breast cancer, received chemotherapy, and were randomly assigned to 5 years of various adjuvant endocrine therapies.
In SOFT, the 5-year breast cancer–free interval was 67.1% (95% CI, 54.6%-76.9%) with tamoxifen (Nolvadex) alone, 75.9% (64.0%-84.4%) with tamoxifen plus OFS, and 83.2% (72.7%-90.0%) with exemestane (Aromasin) plus OFS (J Clin Oncol. 2017 June 27 doi: 10.1200/JCO.2016.72.0946). In TEXT, it was 79.2% (66.2%-87.7%) with tamoxifen plus OFS and 81.6% (69.8%-89.2%) with exemestane plus OFS.
In a quality of life analysis among women receiving OFS, vasomotor symptoms (hot flushes and sweats) showed greatest increase from baseline (roughly 30-40 points) in the first 6 months of therapy. Loss of sexual interest and difficulties in becoming aroused were also noteworthy (8 points or greater). However, scores for global quality of life (physical well-being, mood, coping effort, and health perception) showed little change from baseline and were essentially the same as those seen among premenopausal women aged 35 or older in the same trials.
Overall, 19.8% of the young women in SOFT and TEXT stopped all protocol-assigned endocrine therapy early. The proportion rose with time and was higher than that among the older premenopausal group.
“There was a meaningful clinical benefit in breast cancer outcomes with the addition of OFS to tamoxifen and some additional benefit from use of an aromatase inhibitor with OFS. Longer follow-up is critical to clarify potential survival benefits,” the investigators wrote. “There were substantial adverse effects from these combined endocrine treatments, but they were not different in the younger and older than 35 years populations. Despite this, rates of nonadherence were slightly higher in women younger than 35 years.
“Availability of these age-specific data regarding risks and benefits of combined endocrine therapy will support shared decision making regarding OFS among young women at high risk for recurrence and death from breast cancer and, it is hoped, improve adherence among those who select OFS,” they concluded.
Young women treated for early hormone receptor–positive, HER2-negative breast cancer fare better if their adjuvant endocrine therapy includes ovarian function suppression (OFS), according to a new analysis published online.
Senior author Gini F. Fleming, MD, director of the medical oncology breast program at the University of Chicago Medical Center, and her colleagues analyzed data from a pair of international phase III randomized adjuvant trials: the Suppression of Ovarian Function Trial (SOFT) and the Tamoxifen and Exemestane Trial (TEXT).
Main analyses were based on a respective 240 and 145 women younger than 35 years who had undergone surgery for early hormone receptor–positive, HER2-negative early breast cancer, received chemotherapy, and were randomly assigned to 5 years of various adjuvant endocrine therapies.
In SOFT, the 5-year breast cancer–free interval was 67.1% (95% CI, 54.6%-76.9%) with tamoxifen (Nolvadex) alone, 75.9% (64.0%-84.4%) with tamoxifen plus OFS, and 83.2% (72.7%-90.0%) with exemestane (Aromasin) plus OFS (J Clin Oncol. 2017 June 27 doi: 10.1200/JCO.2016.72.0946). In TEXT, it was 79.2% (66.2%-87.7%) with tamoxifen plus OFS and 81.6% (69.8%-89.2%) with exemestane plus OFS.
In a quality of life analysis among women receiving OFS, vasomotor symptoms (hot flushes and sweats) showed greatest increase from baseline (roughly 30-40 points) in the first 6 months of therapy. Loss of sexual interest and difficulties in becoming aroused were also noteworthy (8 points or greater). However, scores for global quality of life (physical well-being, mood, coping effort, and health perception) showed little change from baseline and were essentially the same as those seen among premenopausal women aged 35 or older in the same trials.
Overall, 19.8% of the young women in SOFT and TEXT stopped all protocol-assigned endocrine therapy early. The proportion rose with time and was higher than that among the older premenopausal group.
“There was a meaningful clinical benefit in breast cancer outcomes with the addition of OFS to tamoxifen and some additional benefit from use of an aromatase inhibitor with OFS. Longer follow-up is critical to clarify potential survival benefits,” the investigators wrote. “There were substantial adverse effects from these combined endocrine treatments, but they were not different in the younger and older than 35 years populations. Despite this, rates of nonadherence were slightly higher in women younger than 35 years.
“Availability of these age-specific data regarding risks and benefits of combined endocrine therapy will support shared decision making regarding OFS among young women at high risk for recurrence and death from breast cancer and, it is hoped, improve adherence among those who select OFS,” they concluded.
FROM JOURNAL OF CLINICAL ONCOLOGY
Key clinical point:
Major finding: In SOFT, the 5-year breast cancer–free interval was 67.1% with tamoxifen alone, 75.9% with tamoxifen plus OFS, and 83.2% with exemestane plus OFS. In TEXT, it was 79.2% with tamoxifen plus OFS and 81.6% with exemestane plus OFS.
Data source: An analysis of women younger than 35 years treated for early hormone receptor–positive, HER2-negative breast cancer and given adjuvant endocrine therapy in the phase III randomized SOFT trial (240 women) or TEXT trial (145 women).
Disclosures: Dr. Fleming disclosed that she receives research funding from Corcept Therapeutics (institutional) and has a relationship with Aeterna Zentaris.
Low-fat diet reduces risk of death if breast cancer is diagnosed
Women can reduce their risk of dying should they receive a breast cancer diagnosis by following a low-fat diet, suggests an analysis from the phase III multicenter randomized Women’s Health Initiative Dietary Modification trial.
Investigators led by Rowan T. Chlebowski, MD, PhD, formerly of the Los Angeles Biomedical Research Institute at Harbor-UCLA Medical Center, Torrance, California, and now at City of Hope National Medical Center, Duarte, California, analyzed data from 48,835 postmenopausal women who had never had breast cancer and had normal mammograms. The women were randomly assigned 2:3 to a diet aimed at reducing fat intake to 20% of energy and increasing intake of fruits, vegetables, and grains or to a usual diet.
However, the rate of deaths after breast cancer from any cause was 0.025% per year in the former group and 0.038% per year in the latter group, a difference that translated to a more than one-third reduction in risk (hazard ratio, 0.65; P = .02).
Similarly, during the median 16.1-year total follow-up, the rate of deaths attributed to breast cancer was 0.035% per year in the low-fat diet group and 0.039% per year in the usual diet group, a difference that was not significant (P =. 41). However, the rate of deaths after breast cancer from any cause was 0.085% per year in the former group and 0.11% per year in the latter group, a difference that translated to a nearly one-fifth reduction in the risk of death (HR, 0.82; P = .01).
In subgroup analyses, there were significant interactions whereby benefit was greater for women who had a baseline waist circumference of at least 88 cm and increased with the baseline percentage of total energy from fat.
“The lower risk of poor prognosis, ER+, PR– breast cancers … in the dietary group contributed to the favorable dietary effect on death after breast cancer,” the investigators noted. “An additional factor that potentially influenced deaths after breast cancer could be a favorable dietary influence on mortality as a result of other causes, including cardiovascular disease.”
“Future studies of other lifestyle interventions on breast cancer incidence and outcome could incorporate some form of a low-fat dietary pattern as a base,” they concluded.
Women can reduce their risk of dying should they receive a breast cancer diagnosis by following a low-fat diet, suggests an analysis from the phase III multicenter randomized Women’s Health Initiative Dietary Modification trial.
Investigators led by Rowan T. Chlebowski, MD, PhD, formerly of the Los Angeles Biomedical Research Institute at Harbor-UCLA Medical Center, Torrance, California, and now at City of Hope National Medical Center, Duarte, California, analyzed data from 48,835 postmenopausal women who had never had breast cancer and had normal mammograms. The women were randomly assigned 2:3 to a diet aimed at reducing fat intake to 20% of energy and increasing intake of fruits, vegetables, and grains or to a usual diet.
However, the rate of deaths after breast cancer from any cause was 0.025% per year in the former group and 0.038% per year in the latter group, a difference that translated to a more than one-third reduction in risk (hazard ratio, 0.65; P = .02).
Similarly, during the median 16.1-year total follow-up, the rate of deaths attributed to breast cancer was 0.035% per year in the low-fat diet group and 0.039% per year in the usual diet group, a difference that was not significant (P =. 41). However, the rate of deaths after breast cancer from any cause was 0.085% per year in the former group and 0.11% per year in the latter group, a difference that translated to a nearly one-fifth reduction in the risk of death (HR, 0.82; P = .01).
In subgroup analyses, there were significant interactions whereby benefit was greater for women who had a baseline waist circumference of at least 88 cm and increased with the baseline percentage of total energy from fat.
“The lower risk of poor prognosis, ER+, PR– breast cancers … in the dietary group contributed to the favorable dietary effect on death after breast cancer,” the investigators noted. “An additional factor that potentially influenced deaths after breast cancer could be a favorable dietary influence on mortality as a result of other causes, including cardiovascular disease.”
“Future studies of other lifestyle interventions on breast cancer incidence and outcome could incorporate some form of a low-fat dietary pattern as a base,” they concluded.
Women can reduce their risk of dying should they receive a breast cancer diagnosis by following a low-fat diet, suggests an analysis from the phase III multicenter randomized Women’s Health Initiative Dietary Modification trial.
Investigators led by Rowan T. Chlebowski, MD, PhD, formerly of the Los Angeles Biomedical Research Institute at Harbor-UCLA Medical Center, Torrance, California, and now at City of Hope National Medical Center, Duarte, California, analyzed data from 48,835 postmenopausal women who had never had breast cancer and had normal mammograms. The women were randomly assigned 2:3 to a diet aimed at reducing fat intake to 20% of energy and increasing intake of fruits, vegetables, and grains or to a usual diet.
However, the rate of deaths after breast cancer from any cause was 0.025% per year in the former group and 0.038% per year in the latter group, a difference that translated to a more than one-third reduction in risk (hazard ratio, 0.65; P = .02).
Similarly, during the median 16.1-year total follow-up, the rate of deaths attributed to breast cancer was 0.035% per year in the low-fat diet group and 0.039% per year in the usual diet group, a difference that was not significant (P =. 41). However, the rate of deaths after breast cancer from any cause was 0.085% per year in the former group and 0.11% per year in the latter group, a difference that translated to a nearly one-fifth reduction in the risk of death (HR, 0.82; P = .01).
In subgroup analyses, there were significant interactions whereby benefit was greater for women who had a baseline waist circumference of at least 88 cm and increased with the baseline percentage of total energy from fat.
“The lower risk of poor prognosis, ER+, PR– breast cancers … in the dietary group contributed to the favorable dietary effect on death after breast cancer,” the investigators noted. “An additional factor that potentially influenced deaths after breast cancer could be a favorable dietary influence on mortality as a result of other causes, including cardiovascular disease.”
“Future studies of other lifestyle interventions on breast cancer incidence and outcome could incorporate some form of a low-fat dietary pattern as a base,” they concluded.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Key clinical point:
Major finding: Compared with peers assigned to a usual diet, women assigned to a low-fat diet were less likely to die after breast cancer diagnosis from any cause during both the 8.5-year intervention (HR 0.65) and the 16.1-year total follow-up (HR, 0.82).
Data source: A posthoc analysis of a phase III randomized controlled trial among 48,835 postmenopausal women who had never had breast cancer and had normal mammograms (Women’s Health Initiative Dietary Modification trial).
Disclosures: Dr. Chlebowski disclosed consulting or advisory roles with Novartis, Genentech, Amgen, Pfizer, and AstraZeneca. He is also on the Speakers’ Bureau for Novartis and Genentech.
Quality of life preserved with ribociclib + letrozole for advanced breast cancer
CHICAGO – Patients with advanced breast cancer whose aromatase inhibitor therapy was supplemented with a cycline-dependent kinase inhibitor had better progression-free survival with no drop in quality of life. Health-related quality of life for patients on the combination therapy was equivalent to that of patients on monotherapy in most aspects, but patients receiving both therapies had a sustained and clinically meaningful decrease in pain.
Also, the time to definitive deterioration by 10% or more of the global health status/quality of life scale score was similar between treatment arms (hazard ratio [HR] 0.944; 95% confidence interval [CI] 0.720-1.237).
The MONALEESA-2 trial had previously shown that the CDK 4/6 inhibitor ribociclib, when added to the aromatase inhibitor letrozole, significantly improved progression-free survival for postmenopausal patients with hormone receptor-positive, HER2 negative advanced breast cancer, when compared to letrozole in combination with placebo.
Sunil Verma, MD, reported on health-related quality of life and symptoms in the two arms of MONALEESA-2, reporting change from baseline, time to a definitive 10% deterioration, and the mean scores for on-treatment time compared to end of treatment on the global health status/quality of life subscale of the European Organization for Research and Treatment of Cancer 30-item core quality of life questionnaire (EORTC QLQ-30).
“During treatment, overall health-related quality of life was maintained from baseline and was similar in both arms,” said Dr. Verma, the study’s first author. Changes during treatment were not statistically significant, and did not reach the predetermined threshold for a clinically meaningful difference. The effect of such key symptoms as fatigue, nausea, and vomiting on quality of life was similar regardless of whether patients received ribociclib or placebo, he said; though symptom scores were slightly higher for patients in the active arm of the study, the results were not clinically significant.
Reporting validated, cancer-specific patient-reported outcomes from the trial, Dr. Verma, professor and head of the department of oncology at the University of Calgary, (Alta.), sought to “highlight the patient experience with a focus on health-related quality of life and symptoms,” he said during his presentation at the annual meeting of the American Society of Clinical Oncology.
“A clinically meaningful – more than 5-point – improvement from baseline in pain score was maintained up to and including cycle 15 in the ribociclib plus letrozole arm,” said Dr. Verma. The placebo arm had a mild, clinically insignificant improvement at most assessment points. For both treatment arms, pain scores increased a bit above baseline levels at the time of disease progression or end of therapy, he said.
Patients completed the EORTC 30-item core quality of life questionnaire at their screening visit, and then every 8 weeks for the first 18 months. Then, they completed the questionnaires every 12 weeks until they experienced disease progression, died, were lost to follow-up or withdrew from the study, or stopped treatment.
Statistical analysis of the questionnaire results took into account the patients’ baseline scores, treatments received, and how they were stratified. Investigators assessed both statistical significance and the clinical meaningfulness of changes, defined as a change of 5-10 points.
In MONALEESA-2, 334 patients each were allocated to the ribociclib + letrozole arm and the placebo + letrozole arm. Patients in both arms, said Dr. Verma, were very compliant with questionnaire completion. Over 90% of patients who were eligible completed questionnaire through cycle 19 of ribociclib or placebo. He explained that the overall numbers completing the questionnaire declined with time, as more patients had disease progression.
It’s important to include these measures, he said, because patients and their families care about “the quality of the time gained,” so patient-reported outcomes should be a part of risk-benefit assessments for new cancer therapies. “While delaying disease progression may help to maintain patient quality of life, the addition of novel treatments to existing therapies can add toxicities, which may diminish quality of life,” said Dr. Verma.
Dr. Verma reported financial relationships with multiple pharmaceutical companies, including Novartis, which funded the study.
[email protected]
On Twitter @karioakes
CHICAGO – Patients with advanced breast cancer whose aromatase inhibitor therapy was supplemented with a cycline-dependent kinase inhibitor had better progression-free survival with no drop in quality of life. Health-related quality of life for patients on the combination therapy was equivalent to that of patients on monotherapy in most aspects, but patients receiving both therapies had a sustained and clinically meaningful decrease in pain.
Also, the time to definitive deterioration by 10% or more of the global health status/quality of life scale score was similar between treatment arms (hazard ratio [HR] 0.944; 95% confidence interval [CI] 0.720-1.237).
The MONALEESA-2 trial had previously shown that the CDK 4/6 inhibitor ribociclib, when added to the aromatase inhibitor letrozole, significantly improved progression-free survival for postmenopausal patients with hormone receptor-positive, HER2 negative advanced breast cancer, when compared to letrozole in combination with placebo.
Sunil Verma, MD, reported on health-related quality of life and symptoms in the two arms of MONALEESA-2, reporting change from baseline, time to a definitive 10% deterioration, and the mean scores for on-treatment time compared to end of treatment on the global health status/quality of life subscale of the European Organization for Research and Treatment of Cancer 30-item core quality of life questionnaire (EORTC QLQ-30).
“During treatment, overall health-related quality of life was maintained from baseline and was similar in both arms,” said Dr. Verma, the study’s first author. Changes during treatment were not statistically significant, and did not reach the predetermined threshold for a clinically meaningful difference. The effect of such key symptoms as fatigue, nausea, and vomiting on quality of life was similar regardless of whether patients received ribociclib or placebo, he said; though symptom scores were slightly higher for patients in the active arm of the study, the results were not clinically significant.
Reporting validated, cancer-specific patient-reported outcomes from the trial, Dr. Verma, professor and head of the department of oncology at the University of Calgary, (Alta.), sought to “highlight the patient experience with a focus on health-related quality of life and symptoms,” he said during his presentation at the annual meeting of the American Society of Clinical Oncology.
“A clinically meaningful – more than 5-point – improvement from baseline in pain score was maintained up to and including cycle 15 in the ribociclib plus letrozole arm,” said Dr. Verma. The placebo arm had a mild, clinically insignificant improvement at most assessment points. For both treatment arms, pain scores increased a bit above baseline levels at the time of disease progression or end of therapy, he said.
Patients completed the EORTC 30-item core quality of life questionnaire at their screening visit, and then every 8 weeks for the first 18 months. Then, they completed the questionnaires every 12 weeks until they experienced disease progression, died, were lost to follow-up or withdrew from the study, or stopped treatment.
Statistical analysis of the questionnaire results took into account the patients’ baseline scores, treatments received, and how they were stratified. Investigators assessed both statistical significance and the clinical meaningfulness of changes, defined as a change of 5-10 points.
In MONALEESA-2, 334 patients each were allocated to the ribociclib + letrozole arm and the placebo + letrozole arm. Patients in both arms, said Dr. Verma, were very compliant with questionnaire completion. Over 90% of patients who were eligible completed questionnaire through cycle 19 of ribociclib or placebo. He explained that the overall numbers completing the questionnaire declined with time, as more patients had disease progression.
It’s important to include these measures, he said, because patients and their families care about “the quality of the time gained,” so patient-reported outcomes should be a part of risk-benefit assessments for new cancer therapies. “While delaying disease progression may help to maintain patient quality of life, the addition of novel treatments to existing therapies can add toxicities, which may diminish quality of life,” said Dr. Verma.
Dr. Verma reported financial relationships with multiple pharmaceutical companies, including Novartis, which funded the study.
[email protected]
On Twitter @karioakes
CHICAGO – Patients with advanced breast cancer whose aromatase inhibitor therapy was supplemented with a cycline-dependent kinase inhibitor had better progression-free survival with no drop in quality of life. Health-related quality of life for patients on the combination therapy was equivalent to that of patients on monotherapy in most aspects, but patients receiving both therapies had a sustained and clinically meaningful decrease in pain.
Also, the time to definitive deterioration by 10% or more of the global health status/quality of life scale score was similar between treatment arms (hazard ratio [HR] 0.944; 95% confidence interval [CI] 0.720-1.237).
The MONALEESA-2 trial had previously shown that the CDK 4/6 inhibitor ribociclib, when added to the aromatase inhibitor letrozole, significantly improved progression-free survival for postmenopausal patients with hormone receptor-positive, HER2 negative advanced breast cancer, when compared to letrozole in combination with placebo.
Sunil Verma, MD, reported on health-related quality of life and symptoms in the two arms of MONALEESA-2, reporting change from baseline, time to a definitive 10% deterioration, and the mean scores for on-treatment time compared to end of treatment on the global health status/quality of life subscale of the European Organization for Research and Treatment of Cancer 30-item core quality of life questionnaire (EORTC QLQ-30).
“During treatment, overall health-related quality of life was maintained from baseline and was similar in both arms,” said Dr. Verma, the study’s first author. Changes during treatment were not statistically significant, and did not reach the predetermined threshold for a clinically meaningful difference. The effect of such key symptoms as fatigue, nausea, and vomiting on quality of life was similar regardless of whether patients received ribociclib or placebo, he said; though symptom scores were slightly higher for patients in the active arm of the study, the results were not clinically significant.
Reporting validated, cancer-specific patient-reported outcomes from the trial, Dr. Verma, professor and head of the department of oncology at the University of Calgary, (Alta.), sought to “highlight the patient experience with a focus on health-related quality of life and symptoms,” he said during his presentation at the annual meeting of the American Society of Clinical Oncology.
“A clinically meaningful – more than 5-point – improvement from baseline in pain score was maintained up to and including cycle 15 in the ribociclib plus letrozole arm,” said Dr. Verma. The placebo arm had a mild, clinically insignificant improvement at most assessment points. For both treatment arms, pain scores increased a bit above baseline levels at the time of disease progression or end of therapy, he said.
Patients completed the EORTC 30-item core quality of life questionnaire at their screening visit, and then every 8 weeks for the first 18 months. Then, they completed the questionnaires every 12 weeks until they experienced disease progression, died, were lost to follow-up or withdrew from the study, or stopped treatment.
Statistical analysis of the questionnaire results took into account the patients’ baseline scores, treatments received, and how they were stratified. Investigators assessed both statistical significance and the clinical meaningfulness of changes, defined as a change of 5-10 points.
In MONALEESA-2, 334 patients each were allocated to the ribociclib + letrozole arm and the placebo + letrozole arm. Patients in both arms, said Dr. Verma, were very compliant with questionnaire completion. Over 90% of patients who were eligible completed questionnaire through cycle 19 of ribociclib or placebo. He explained that the overall numbers completing the questionnaire declined with time, as more patients had disease progression.
It’s important to include these measures, he said, because patients and their families care about “the quality of the time gained,” so patient-reported outcomes should be a part of risk-benefit assessments for new cancer therapies. “While delaying disease progression may help to maintain patient quality of life, the addition of novel treatments to existing therapies can add toxicities, which may diminish quality of life,” said Dr. Verma.
Dr. Verma reported financial relationships with multiple pharmaceutical companies, including Novartis, which funded the study.
[email protected]
On Twitter @karioakes
AT ASCO 2017
Key clinical point:
Major finding: Quality of life was sustained and pain scores decreased when ribociclib was added to letrozole for patients with advanced breast cancer.
Data source: Double-blind, placebo-controlled phase III trial of letrozole plus ribociclib compared to letrozole plus placebo in 668 patients with advanced hormone receptor-positive, HER-2 negative breast cancer.
Disclosures: Dr. Verma reported financial relationships with multiple companies, including Novartis, which markets ribociclib.
SOLE trial: Intermittent letrozole may provide benefit for some breast cancer patients
CHICAGO – Five years of extended intermittent letrozole provided no overall improvement in disease-free survival (DFS) when compared with continuous treatment with the aromatase inhibitor among postmenopausal women who received 4-6 years of endocrine therapy for hormone receptor–positive, lymph node–positive early breast cancer in the randomized phase III Study of Letrozole Extension (SOLE).
However, the similarities in DFS and adverse events incidence in the intermittent and continuous treatment groups – and improvements in a number of quality of life measures with intermittent treatment – suggest that temporary treatment breaks are acceptable in those who could benefit from them, Marco Colleoni, MD, reported at the annual meeting of the American Society of Clinical Oncology.
The DFS among 2,425 women who received 5 years of intermittent letrozole after completing 4-6 years of adjuvant endocrine therapy was 85.8%, compared with 87.5% among 2,426 women who received 5 years of continuous letrozole after adjuvant therapy (hazard ratio, 1.08) said Dr. Colleoni of the European Institute of Oncology, Milan, Italy.
Similar outcomes were observed for breast cancer–free interval (HR, 0.98), distant recurrence-free interval (HR, 0.88), and overall survival (HR, 0.85), he said.
Of note, trends toward greater improvement on both the primary and secondary endpoints were seen with continuous therapy among those who received prior selective estrogen receptor modulator (SERM) vs. aromatase inhibitor (AI) therapy, but the differences did not reach statistical significance, he said.
Adjuvant extended endocrine therapy with letrozole has been recommended after initial tamoxifen for postmenopausal women with hormone receptor–positive (HR+) breast cancer, but the magnitude of the beneficial effect of extended letrozole in women who previously received an aromatase inhibitor has been limited, Dr. Colleoni said.
“Therefore, based on available data, the use of an AI for 10 years should be discussed on an individualized basis,” he said.
However, there is rationale for the intermittent use of letrozole as a therapeutic option to prolong sensitivity to endocrine therapies,” he noted.
“Breast cancer cells, in fact, can develop resistance following antihormonal therapy. In particular, cells that are maintained estrogen-free for years start to grow spontaneously. It has been shown ... that minimal concentrations of estrogen, similar to those achievable through interruption of treatment with aromatase inhibitors, can produce a cytocidal effect on cells that are deprived of estrogen,” he explained. “Moreover, in breast cancer cells transplanted into animal models, a response to a 6-week withdrawal of letrozole was observed when the treatment was started again.”
Thus, the SOLE study was designed to look at the value of extended adjuvant therapy in patients with HR+ breast cancer, the role of intermittent administration of letrozole in patients previously treated with AIs, the impact of intermittent vs. continuous administration on the side effect profile and quality of life and adherence to intermittent administration, he said.
Study participants were women with a median age of 60 years who were enrolled from 240 centers in 22 countries between November 2007 and July 2012. They had HR+ invasive breast cancer, any HER2 status, and disease confined to the breast and axillary lymph nodes. All had received SERM, AI, or combination SERM and AI treatment for 4-6 years but must have discontinued that therapy within 1 year prior to randomization.
Women in the intermittent treatment group were treated for the first 9 months of years 1-4 and for 12 months in year 5. Those in the continuous treatment group received 2.5 mg of letrozole daily for 5 years.
The adverse events were as expected, with 36.2% and 34.5% of patients in the intermittent and continuous treatment groups experiencing grade 3 or 5 events, respectively, and were similar, Dr. Colleoni said.
Treatment was discontinued for DFS events in 8% of patients and for other reasons in 24% of patients. Treatment was completed by 39% of patients and currently is ongoing in 29%, he said.
“When we designed the SOLE study, we were worried about ... the treatment gap. We’re delighted to observe that more than 90% of the patients regularly resumed [treatment] after interruption, ... reassuring [us of] the feasibility of a treatment gap during extended adjuvant letrozole,” he said.
Additionally, a quality of life substudy of 955 patients from the SOLE trial, who were assessed at baseline and at 6, 12, 18, and 24 months, showed a consistent pattern favoring intermittent therapy on patient-reported quality of life measures. Specifically, there was significantly less worsening from baseline with respect to vaginal problems, musculoskeletal pain, sleep disturbance, physical wellbeing, and mood at 12 months in the intermittent vs. continuous letrozole group and significantly greater improvement in hot flashes at 24 months in the intermittent vs. continuous group, he said.
“The results ... provide clinically relevant information on intermittent administration of extended letrozole for patients who could benefit from temporary treatment breaks,” he concluded, noting that translational studies are ongoing, such as measurement of estradiol levels during treatment gaps.
The SOLE trial was supported by Novartis. Dr. Colleoni reported receiving honoraria from Novartis, and serving as a consultant or advisor for AstraZeneca, Celldex, OBI Pharma, Pfizer, Pierre Fabre, and Puma Biotechnology.
CHICAGO – Five years of extended intermittent letrozole provided no overall improvement in disease-free survival (DFS) when compared with continuous treatment with the aromatase inhibitor among postmenopausal women who received 4-6 years of endocrine therapy for hormone receptor–positive, lymph node–positive early breast cancer in the randomized phase III Study of Letrozole Extension (SOLE).
However, the similarities in DFS and adverse events incidence in the intermittent and continuous treatment groups – and improvements in a number of quality of life measures with intermittent treatment – suggest that temporary treatment breaks are acceptable in those who could benefit from them, Marco Colleoni, MD, reported at the annual meeting of the American Society of Clinical Oncology.
The DFS among 2,425 women who received 5 years of intermittent letrozole after completing 4-6 years of adjuvant endocrine therapy was 85.8%, compared with 87.5% among 2,426 women who received 5 years of continuous letrozole after adjuvant therapy (hazard ratio, 1.08) said Dr. Colleoni of the European Institute of Oncology, Milan, Italy.
Similar outcomes were observed for breast cancer–free interval (HR, 0.98), distant recurrence-free interval (HR, 0.88), and overall survival (HR, 0.85), he said.
Of note, trends toward greater improvement on both the primary and secondary endpoints were seen with continuous therapy among those who received prior selective estrogen receptor modulator (SERM) vs. aromatase inhibitor (AI) therapy, but the differences did not reach statistical significance, he said.
Adjuvant extended endocrine therapy with letrozole has been recommended after initial tamoxifen for postmenopausal women with hormone receptor–positive (HR+) breast cancer, but the magnitude of the beneficial effect of extended letrozole in women who previously received an aromatase inhibitor has been limited, Dr. Colleoni said.
“Therefore, based on available data, the use of an AI for 10 years should be discussed on an individualized basis,” he said.
However, there is rationale for the intermittent use of letrozole as a therapeutic option to prolong sensitivity to endocrine therapies,” he noted.
“Breast cancer cells, in fact, can develop resistance following antihormonal therapy. In particular, cells that are maintained estrogen-free for years start to grow spontaneously. It has been shown ... that minimal concentrations of estrogen, similar to those achievable through interruption of treatment with aromatase inhibitors, can produce a cytocidal effect on cells that are deprived of estrogen,” he explained. “Moreover, in breast cancer cells transplanted into animal models, a response to a 6-week withdrawal of letrozole was observed when the treatment was started again.”
Thus, the SOLE study was designed to look at the value of extended adjuvant therapy in patients with HR+ breast cancer, the role of intermittent administration of letrozole in patients previously treated with AIs, the impact of intermittent vs. continuous administration on the side effect profile and quality of life and adherence to intermittent administration, he said.
Study participants were women with a median age of 60 years who were enrolled from 240 centers in 22 countries between November 2007 and July 2012. They had HR+ invasive breast cancer, any HER2 status, and disease confined to the breast and axillary lymph nodes. All had received SERM, AI, or combination SERM and AI treatment for 4-6 years but must have discontinued that therapy within 1 year prior to randomization.
Women in the intermittent treatment group were treated for the first 9 months of years 1-4 and for 12 months in year 5. Those in the continuous treatment group received 2.5 mg of letrozole daily for 5 years.
The adverse events were as expected, with 36.2% and 34.5% of patients in the intermittent and continuous treatment groups experiencing grade 3 or 5 events, respectively, and were similar, Dr. Colleoni said.
Treatment was discontinued for DFS events in 8% of patients and for other reasons in 24% of patients. Treatment was completed by 39% of patients and currently is ongoing in 29%, he said.
“When we designed the SOLE study, we were worried about ... the treatment gap. We’re delighted to observe that more than 90% of the patients regularly resumed [treatment] after interruption, ... reassuring [us of] the feasibility of a treatment gap during extended adjuvant letrozole,” he said.
Additionally, a quality of life substudy of 955 patients from the SOLE trial, who were assessed at baseline and at 6, 12, 18, and 24 months, showed a consistent pattern favoring intermittent therapy on patient-reported quality of life measures. Specifically, there was significantly less worsening from baseline with respect to vaginal problems, musculoskeletal pain, sleep disturbance, physical wellbeing, and mood at 12 months in the intermittent vs. continuous letrozole group and significantly greater improvement in hot flashes at 24 months in the intermittent vs. continuous group, he said.
“The results ... provide clinically relevant information on intermittent administration of extended letrozole for patients who could benefit from temporary treatment breaks,” he concluded, noting that translational studies are ongoing, such as measurement of estradiol levels during treatment gaps.
The SOLE trial was supported by Novartis. Dr. Colleoni reported receiving honoraria from Novartis, and serving as a consultant or advisor for AstraZeneca, Celldex, OBI Pharma, Pfizer, Pierre Fabre, and Puma Biotechnology.
CHICAGO – Five years of extended intermittent letrozole provided no overall improvement in disease-free survival (DFS) when compared with continuous treatment with the aromatase inhibitor among postmenopausal women who received 4-6 years of endocrine therapy for hormone receptor–positive, lymph node–positive early breast cancer in the randomized phase III Study of Letrozole Extension (SOLE).
However, the similarities in DFS and adverse events incidence in the intermittent and continuous treatment groups – and improvements in a number of quality of life measures with intermittent treatment – suggest that temporary treatment breaks are acceptable in those who could benefit from them, Marco Colleoni, MD, reported at the annual meeting of the American Society of Clinical Oncology.
The DFS among 2,425 women who received 5 years of intermittent letrozole after completing 4-6 years of adjuvant endocrine therapy was 85.8%, compared with 87.5% among 2,426 women who received 5 years of continuous letrozole after adjuvant therapy (hazard ratio, 1.08) said Dr. Colleoni of the European Institute of Oncology, Milan, Italy.
Similar outcomes were observed for breast cancer–free interval (HR, 0.98), distant recurrence-free interval (HR, 0.88), and overall survival (HR, 0.85), he said.
Of note, trends toward greater improvement on both the primary and secondary endpoints were seen with continuous therapy among those who received prior selective estrogen receptor modulator (SERM) vs. aromatase inhibitor (AI) therapy, but the differences did not reach statistical significance, he said.
Adjuvant extended endocrine therapy with letrozole has been recommended after initial tamoxifen for postmenopausal women with hormone receptor–positive (HR+) breast cancer, but the magnitude of the beneficial effect of extended letrozole in women who previously received an aromatase inhibitor has been limited, Dr. Colleoni said.
“Therefore, based on available data, the use of an AI for 10 years should be discussed on an individualized basis,” he said.
However, there is rationale for the intermittent use of letrozole as a therapeutic option to prolong sensitivity to endocrine therapies,” he noted.
“Breast cancer cells, in fact, can develop resistance following antihormonal therapy. In particular, cells that are maintained estrogen-free for years start to grow spontaneously. It has been shown ... that minimal concentrations of estrogen, similar to those achievable through interruption of treatment with aromatase inhibitors, can produce a cytocidal effect on cells that are deprived of estrogen,” he explained. “Moreover, in breast cancer cells transplanted into animal models, a response to a 6-week withdrawal of letrozole was observed when the treatment was started again.”
Thus, the SOLE study was designed to look at the value of extended adjuvant therapy in patients with HR+ breast cancer, the role of intermittent administration of letrozole in patients previously treated with AIs, the impact of intermittent vs. continuous administration on the side effect profile and quality of life and adherence to intermittent administration, he said.
Study participants were women with a median age of 60 years who were enrolled from 240 centers in 22 countries between November 2007 and July 2012. They had HR+ invasive breast cancer, any HER2 status, and disease confined to the breast and axillary lymph nodes. All had received SERM, AI, or combination SERM and AI treatment for 4-6 years but must have discontinued that therapy within 1 year prior to randomization.
Women in the intermittent treatment group were treated for the first 9 months of years 1-4 and for 12 months in year 5. Those in the continuous treatment group received 2.5 mg of letrozole daily for 5 years.
The adverse events were as expected, with 36.2% and 34.5% of patients in the intermittent and continuous treatment groups experiencing grade 3 or 5 events, respectively, and were similar, Dr. Colleoni said.
Treatment was discontinued for DFS events in 8% of patients and for other reasons in 24% of patients. Treatment was completed by 39% of patients and currently is ongoing in 29%, he said.
“When we designed the SOLE study, we were worried about ... the treatment gap. We’re delighted to observe that more than 90% of the patients regularly resumed [treatment] after interruption, ... reassuring [us of] the feasibility of a treatment gap during extended adjuvant letrozole,” he said.
Additionally, a quality of life substudy of 955 patients from the SOLE trial, who were assessed at baseline and at 6, 12, 18, and 24 months, showed a consistent pattern favoring intermittent therapy on patient-reported quality of life measures. Specifically, there was significantly less worsening from baseline with respect to vaginal problems, musculoskeletal pain, sleep disturbance, physical wellbeing, and mood at 12 months in the intermittent vs. continuous letrozole group and significantly greater improvement in hot flashes at 24 months in the intermittent vs. continuous group, he said.
“The results ... provide clinically relevant information on intermittent administration of extended letrozole for patients who could benefit from temporary treatment breaks,” he concluded, noting that translational studies are ongoing, such as measurement of estradiol levels during treatment gaps.
The SOLE trial was supported by Novartis. Dr. Colleoni reported receiving honoraria from Novartis, and serving as a consultant or advisor for AstraZeneca, Celldex, OBI Pharma, Pfizer, Pierre Fabre, and Puma Biotechnology.
AT ASCO 2017
Key clinical point:
Major finding: Disease-free survival was 85.8% and 87.5% with intermittent and continuous letrozole, respectively (hazard ratio, 1.08).
Data source: The phase III SOLE study of 4,851 women.
Disclosures: The SOLE trial was supported by Novartis. Dr. Colleoni reported receiving honoraria from Novartis and serving as a consultant or advisor for AstraZeneca, Celldex, OBI Pharma, Pfizer, Pierre Fabre, and Puma Biotechnology.
ALTTO follow-up: Dual HER2 blockade may benefit HER2+/HR- tumors
CHICAGO – HER2-positive/HR-negative breast tumors may have a different biology than HER2+/HR+ tumors and may derive more benefit from dual HER2 blockade, according to 5-year results from the phase III ALTTO trial comparing 1 year of adjuvant anti-HER2 therapy with lapatinib and trastuzumab alone, sequentially, or in combination in patients with HER2-positive early breast cancer.
The hazard ratios for this preplanned updated analysis at a median of 6.9 years of follow-up (when all patients had reached 5 years of follow-up) are similar to those from the primary analysis reported at the 2014 ASCO annual meeting and published in the Journal of Clinical Oncology in 2015 (J Clin Oncol. 2015;34:1034-42) at a median clinical follow-up of 4.5 years, Alvaro Moreno-Aspitia, MD, reported at the annual meeting of the American Society of Clinical Oncology.
In the current analysis, the rate of disease-free survival was 85% with combined lapatinib and trastuzumab (L+T), 84% for sequential trastuzumab and lapatinib (T–L), and 82% with T (hazard ratio for disease-free survival was 0.86 for L+T vs. T and 0.93 for T–L vs. T alone,) said Dr. Moreno-Aspitia of Mayo Clinic, Jacksonville, Fla.
“So, at this time, as noted in the primary analysis, there’s no benefit on the primary endpoint in regard to dual HER2 blockade as provided in this clinical trial,” he said.
There also were no significant differences seen in disease-free survival based on chemotherapy timing, he said.
Notably, disease-free survival was similar across treatment groups among patients with HER2+/HR+ tumors (85% for L+T, 85% to T–L, and 83% for T; HRs, 0.91 and 0.90 vs. T, respectively), but, among those with HER2+/HR– tumors, the 6-year disease-free survival difference for L+T (84%) vs. T (80%) was slightly greater (HR, 0.80).
“This is a hypothesis generating observation that also holds that possibly these patients with hormone receptor–negative tumors may derive benefit from dual HER2 blockade,” Dr. Moreno-Aspitia said, noting that this has also been observed in several other trials.
The 6-year overall survival in ALTTO was 93%, 92%, and 91% for L+T, T–L, and T, respectively (HRs for overall survival, 0.86 for L+T vs. T and 0.88 for T–L).
“At this time, there is absolutely no [overall survival] benefit on dual HER2 blockade over single agent trastuzumab. However, it is very rewarding to see that over 90% of the patients who participated in this trial are alive at this longer-term follow-up,” he said.
As for cardiac events, the numbers remained low as in the primary ALTTO analysis, and no new safety signals had emerged.
ALTTO study subjects were 8,381 patients randomized from 946 sites in 44 countries between June 2007 and July 2011. Those in the L+T group received both agents together for 52 weeks; those in the sequential treatment group received T for 12 weeks followed by a 6-week wash-out period, followed by lapatinib for 34 weeks; and those in the trastuzumab arm (the control arm) and the lapatinib arm each received treatment for 52 weeks. In 2011 the lapatinib arm was closed because of futility, and results for that arm are not included in this analysis.
Long-term follow up of participants continues, a large number of correlative studies are ongoing, and final results will be reported when all patients have been followed for at least 10 years, Dr. Moreno-Aspitia said.
The ALTTO trial was sponsored by GlaxoSmithKline and Novartis. Dr. Moreno-Aspitia reported having no disclosures.
CHICAGO – HER2-positive/HR-negative breast tumors may have a different biology than HER2+/HR+ tumors and may derive more benefit from dual HER2 blockade, according to 5-year results from the phase III ALTTO trial comparing 1 year of adjuvant anti-HER2 therapy with lapatinib and trastuzumab alone, sequentially, or in combination in patients with HER2-positive early breast cancer.
The hazard ratios for this preplanned updated analysis at a median of 6.9 years of follow-up (when all patients had reached 5 years of follow-up) are similar to those from the primary analysis reported at the 2014 ASCO annual meeting and published in the Journal of Clinical Oncology in 2015 (J Clin Oncol. 2015;34:1034-42) at a median clinical follow-up of 4.5 years, Alvaro Moreno-Aspitia, MD, reported at the annual meeting of the American Society of Clinical Oncology.
In the current analysis, the rate of disease-free survival was 85% with combined lapatinib and trastuzumab (L+T), 84% for sequential trastuzumab and lapatinib (T–L), and 82% with T (hazard ratio for disease-free survival was 0.86 for L+T vs. T and 0.93 for T–L vs. T alone,) said Dr. Moreno-Aspitia of Mayo Clinic, Jacksonville, Fla.
“So, at this time, as noted in the primary analysis, there’s no benefit on the primary endpoint in regard to dual HER2 blockade as provided in this clinical trial,” he said.
There also were no significant differences seen in disease-free survival based on chemotherapy timing, he said.
Notably, disease-free survival was similar across treatment groups among patients with HER2+/HR+ tumors (85% for L+T, 85% to T–L, and 83% for T; HRs, 0.91 and 0.90 vs. T, respectively), but, among those with HER2+/HR– tumors, the 6-year disease-free survival difference for L+T (84%) vs. T (80%) was slightly greater (HR, 0.80).
“This is a hypothesis generating observation that also holds that possibly these patients with hormone receptor–negative tumors may derive benefit from dual HER2 blockade,” Dr. Moreno-Aspitia said, noting that this has also been observed in several other trials.
The 6-year overall survival in ALTTO was 93%, 92%, and 91% for L+T, T–L, and T, respectively (HRs for overall survival, 0.86 for L+T vs. T and 0.88 for T–L).
“At this time, there is absolutely no [overall survival] benefit on dual HER2 blockade over single agent trastuzumab. However, it is very rewarding to see that over 90% of the patients who participated in this trial are alive at this longer-term follow-up,” he said.
As for cardiac events, the numbers remained low as in the primary ALTTO analysis, and no new safety signals had emerged.
ALTTO study subjects were 8,381 patients randomized from 946 sites in 44 countries between June 2007 and July 2011. Those in the L+T group received both agents together for 52 weeks; those in the sequential treatment group received T for 12 weeks followed by a 6-week wash-out period, followed by lapatinib for 34 weeks; and those in the trastuzumab arm (the control arm) and the lapatinib arm each received treatment for 52 weeks. In 2011 the lapatinib arm was closed because of futility, and results for that arm are not included in this analysis.
Long-term follow up of participants continues, a large number of correlative studies are ongoing, and final results will be reported when all patients have been followed for at least 10 years, Dr. Moreno-Aspitia said.
The ALTTO trial was sponsored by GlaxoSmithKline and Novartis. Dr. Moreno-Aspitia reported having no disclosures.
CHICAGO – HER2-positive/HR-negative breast tumors may have a different biology than HER2+/HR+ tumors and may derive more benefit from dual HER2 blockade, according to 5-year results from the phase III ALTTO trial comparing 1 year of adjuvant anti-HER2 therapy with lapatinib and trastuzumab alone, sequentially, or in combination in patients with HER2-positive early breast cancer.
The hazard ratios for this preplanned updated analysis at a median of 6.9 years of follow-up (when all patients had reached 5 years of follow-up) are similar to those from the primary analysis reported at the 2014 ASCO annual meeting and published in the Journal of Clinical Oncology in 2015 (J Clin Oncol. 2015;34:1034-42) at a median clinical follow-up of 4.5 years, Alvaro Moreno-Aspitia, MD, reported at the annual meeting of the American Society of Clinical Oncology.
In the current analysis, the rate of disease-free survival was 85% with combined lapatinib and trastuzumab (L+T), 84% for sequential trastuzumab and lapatinib (T–L), and 82% with T (hazard ratio for disease-free survival was 0.86 for L+T vs. T and 0.93 for T–L vs. T alone,) said Dr. Moreno-Aspitia of Mayo Clinic, Jacksonville, Fla.
“So, at this time, as noted in the primary analysis, there’s no benefit on the primary endpoint in regard to dual HER2 blockade as provided in this clinical trial,” he said.
There also were no significant differences seen in disease-free survival based on chemotherapy timing, he said.
Notably, disease-free survival was similar across treatment groups among patients with HER2+/HR+ tumors (85% for L+T, 85% to T–L, and 83% for T; HRs, 0.91 and 0.90 vs. T, respectively), but, among those with HER2+/HR– tumors, the 6-year disease-free survival difference for L+T (84%) vs. T (80%) was slightly greater (HR, 0.80).
“This is a hypothesis generating observation that also holds that possibly these patients with hormone receptor–negative tumors may derive benefit from dual HER2 blockade,” Dr. Moreno-Aspitia said, noting that this has also been observed in several other trials.
The 6-year overall survival in ALTTO was 93%, 92%, and 91% for L+T, T–L, and T, respectively (HRs for overall survival, 0.86 for L+T vs. T and 0.88 for T–L).
“At this time, there is absolutely no [overall survival] benefit on dual HER2 blockade over single agent trastuzumab. However, it is very rewarding to see that over 90% of the patients who participated in this trial are alive at this longer-term follow-up,” he said.
As for cardiac events, the numbers remained low as in the primary ALTTO analysis, and no new safety signals had emerged.
ALTTO study subjects were 8,381 patients randomized from 946 sites in 44 countries between June 2007 and July 2011. Those in the L+T group received both agents together for 52 weeks; those in the sequential treatment group received T for 12 weeks followed by a 6-week wash-out period, followed by lapatinib for 34 weeks; and those in the trastuzumab arm (the control arm) and the lapatinib arm each received treatment for 52 weeks. In 2011 the lapatinib arm was closed because of futility, and results for that arm are not included in this analysis.
Long-term follow up of participants continues, a large number of correlative studies are ongoing, and final results will be reported when all patients have been followed for at least 10 years, Dr. Moreno-Aspitia said.
The ALTTO trial was sponsored by GlaxoSmithKline and Novartis. Dr. Moreno-Aspitia reported having no disclosures.
AT ASCO 2017
Key clinical point:
Major finding: DFS differed slightly between lapatinib plus trastuzumab and trastuzumab alone (84% vs. 80%; HR, 0.80) among those with HER2+/HR-negative tumors but not among those with HER2+/HR+ tumors.
Data source: The phase III ALTTO trial of 8,381 patients.
Disclosures: The ALTTO trial was sponsored by GlaxoSmithKline and Novartis. Dr. Moreno-Aspitia reported having no disclosures.
ACOG recommends women start mammography between ages 40 and 50 years
Woman at average risk for breast cancer should be offered their first screening mammogram at age 40, with initiation of screening beginning no later than age 50, according to new recommendations from the American College of Obstetricians and Gynecologists.
ACOG suggested a screening interval of 1-2 years based on patient values and preferences.
Screening should continue until at least age 75 years. Past that age, continuation should be considered in terms of the patient’s overall health and expected lifespan, according to the Practice Bulletin.
That’s a welcome approach, according to Ritu Salani, MD, a gynecologic oncologist at the Ohio State University, Columbus. “I do believe the ACOG guidelines have the most flexibility and are probably the most comprehensive for average-risk patients,” said Dr. Salani, MD, who was not involved in the drafting of the guidelines.
The recommendations are based in part on a review of existing recommendations from the U.S. Preventive Services Task Force, the American Cancer Society, and the National Comprehensive Cancer Network.
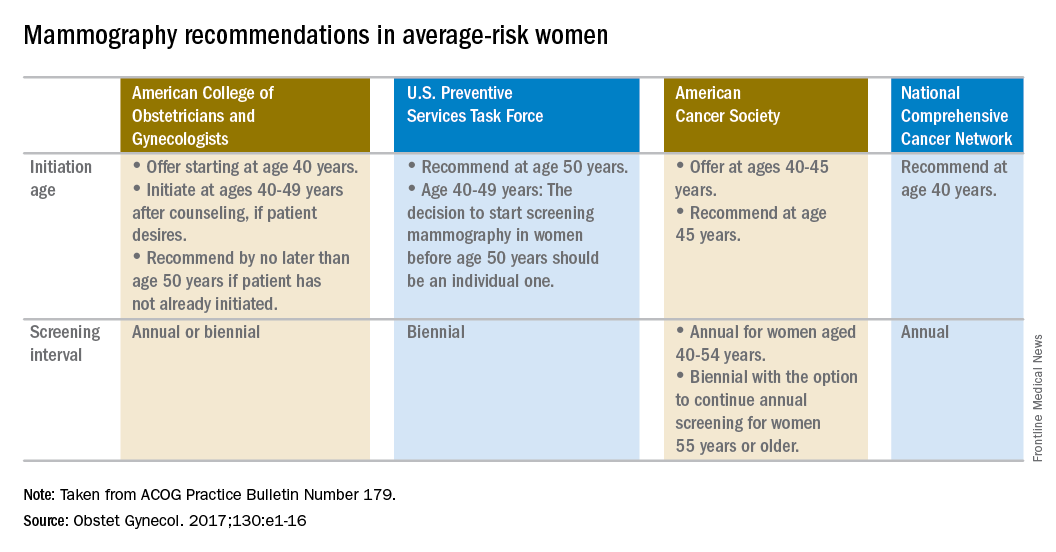
“For patients anxious about developing cancer, maybe every year would be appropriate. When you talk to patients, you have a sense of what would be beneficial for them,” said Dr. Salani, who applauded the flexibility built into the new ACOG guidelines, which says the screening interval can be annual or biennial. “It caters to shared decision-making, and that’s a nice way to frame it.”
The ACOG guidelines recommend against breast self examination because this practice carries the risk of false positives and lacks supporting evidence of benefit. But, ACOG encourages “breast self-awareness,” which means being “attuned to noticing a change or potential problem with her breasts” but without the routine examination.
Women who are asymptomatic and at average risk can be offered clinical breast examination as a screening tool, but clinicians should explain the “uncertainty of additional benefits and the possibility of adverse consequences of clinical breast examination beyond screening mammography.” If performed, ACOG recommends intervals of 1-3 years for women aged 25-39 years and annually for women aged 40 years and over.
The recommendations apply to average-risk women. To help determine risk, the Practice Bulletin also lays out a series of breast cancer risk factors that should be considered, including family history of cancer, known germline mutations, prior biopsy findings, early menarche, late menopause, breast density, and a range of lifestyle and other factors.
Woman at average risk for breast cancer should be offered their first screening mammogram at age 40, with initiation of screening beginning no later than age 50, according to new recommendations from the American College of Obstetricians and Gynecologists.
ACOG suggested a screening interval of 1-2 years based on patient values and preferences.
Screening should continue until at least age 75 years. Past that age, continuation should be considered in terms of the patient’s overall health and expected lifespan, according to the Practice Bulletin.
That’s a welcome approach, according to Ritu Salani, MD, a gynecologic oncologist at the Ohio State University, Columbus. “I do believe the ACOG guidelines have the most flexibility and are probably the most comprehensive for average-risk patients,” said Dr. Salani, MD, who was not involved in the drafting of the guidelines.
The recommendations are based in part on a review of existing recommendations from the U.S. Preventive Services Task Force, the American Cancer Society, and the National Comprehensive Cancer Network.

“For patients anxious about developing cancer, maybe every year would be appropriate. When you talk to patients, you have a sense of what would be beneficial for them,” said Dr. Salani, who applauded the flexibility built into the new ACOG guidelines, which says the screening interval can be annual or biennial. “It caters to shared decision-making, and that’s a nice way to frame it.”
The ACOG guidelines recommend against breast self examination because this practice carries the risk of false positives and lacks supporting evidence of benefit. But, ACOG encourages “breast self-awareness,” which means being “attuned to noticing a change or potential problem with her breasts” but without the routine examination.
Women who are asymptomatic and at average risk can be offered clinical breast examination as a screening tool, but clinicians should explain the “uncertainty of additional benefits and the possibility of adverse consequences of clinical breast examination beyond screening mammography.” If performed, ACOG recommends intervals of 1-3 years for women aged 25-39 years and annually for women aged 40 years and over.
The recommendations apply to average-risk women. To help determine risk, the Practice Bulletin also lays out a series of breast cancer risk factors that should be considered, including family history of cancer, known germline mutations, prior biopsy findings, early menarche, late menopause, breast density, and a range of lifestyle and other factors.
Woman at average risk for breast cancer should be offered their first screening mammogram at age 40, with initiation of screening beginning no later than age 50, according to new recommendations from the American College of Obstetricians and Gynecologists.
ACOG suggested a screening interval of 1-2 years based on patient values and preferences.
Screening should continue until at least age 75 years. Past that age, continuation should be considered in terms of the patient’s overall health and expected lifespan, according to the Practice Bulletin.
That’s a welcome approach, according to Ritu Salani, MD, a gynecologic oncologist at the Ohio State University, Columbus. “I do believe the ACOG guidelines have the most flexibility and are probably the most comprehensive for average-risk patients,” said Dr. Salani, MD, who was not involved in the drafting of the guidelines.
The recommendations are based in part on a review of existing recommendations from the U.S. Preventive Services Task Force, the American Cancer Society, and the National Comprehensive Cancer Network.

“For patients anxious about developing cancer, maybe every year would be appropriate. When you talk to patients, you have a sense of what would be beneficial for them,” said Dr. Salani, who applauded the flexibility built into the new ACOG guidelines, which says the screening interval can be annual or biennial. “It caters to shared decision-making, and that’s a nice way to frame it.”
The ACOG guidelines recommend against breast self examination because this practice carries the risk of false positives and lacks supporting evidence of benefit. But, ACOG encourages “breast self-awareness,” which means being “attuned to noticing a change or potential problem with her breasts” but without the routine examination.
Women who are asymptomatic and at average risk can be offered clinical breast examination as a screening tool, but clinicians should explain the “uncertainty of additional benefits and the possibility of adverse consequences of clinical breast examination beyond screening mammography.” If performed, ACOG recommends intervals of 1-3 years for women aged 25-39 years and annually for women aged 40 years and over.
The recommendations apply to average-risk women. To help determine risk, the Practice Bulletin also lays out a series of breast cancer risk factors that should be considered, including family history of cancer, known germline mutations, prior biopsy findings, early menarche, late menopause, breast density, and a range of lifestyle and other factors.